WO2013122016A1 - 水性顔料分散液及びそれを用いたインクジェット用水性顔料インク - Google Patents
水性顔料分散液及びそれを用いたインクジェット用水性顔料インク Download PDFInfo
- Publication number
- WO2013122016A1 WO2013122016A1 PCT/JP2013/053136 JP2013053136W WO2013122016A1 WO 2013122016 A1 WO2013122016 A1 WO 2013122016A1 JP 2013053136 W JP2013053136 W JP 2013053136W WO 2013122016 A1 WO2013122016 A1 WO 2013122016A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- pigment
- parts
- polymer chain
- meth
- acrylate
- Prior art date
Links
- 239000000049 pigment Substances 0.000 title claims abstract description 336
- 239000006185 dispersion Substances 0.000 title claims abstract description 87
- 239000007788 liquid Substances 0.000 title abstract description 8
- 229920000642 polymer Polymers 0.000 claims abstract description 261
- 239000002270 dispersing agent Substances 0.000 claims abstract description 92
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 claims abstract description 68
- 229920001400 block copolymer Polymers 0.000 claims abstract description 45
- 239000000178 monomer Substances 0.000 claims abstract description 41
- 125000000753 cycloalkyl group Chemical group 0.000 claims abstract description 40
- 229920000578 graft copolymer Polymers 0.000 claims abstract description 36
- 125000003118 aryl group Chemical group 0.000 claims abstract description 13
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 claims abstract description 12
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims abstract description 8
- 229920002554 vinyl polymer Polymers 0.000 claims abstract description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 74
- -1 glycol monoalkyl ether Chemical class 0.000 claims description 52
- 238000000034 method Methods 0.000 claims description 49
- 239000003960 organic solvent Substances 0.000 claims description 24
- LYCAIKOWRPUZTN-UHFFFAOYSA-N ethylene glycol Natural products OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 22
- 239000002904 solvent Substances 0.000 claims description 21
- 125000002947 alkylene group Chemical group 0.000 claims description 16
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims description 14
- 239000003513 alkali Substances 0.000 claims description 10
- 239000002253 acid Substances 0.000 claims description 9
- 238000002156 mixing Methods 0.000 claims description 9
- 150000001346 alkyl aryl ethers Chemical class 0.000 claims description 6
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 5
- ATVJXMYDOSMEPO-UHFFFAOYSA-N 3-prop-2-enoxyprop-1-ene Chemical compound C=CCOCC=C ATVJXMYDOSMEPO-UHFFFAOYSA-N 0.000 claims description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 4
- 150000001408 amides Chemical class 0.000 claims description 4
- 239000003795 chemical substances by application Substances 0.000 claims description 4
- 229920005862 polyol Polymers 0.000 claims description 4
- 239000011164 primary particle Substances 0.000 claims description 4
- 238000004898 kneading Methods 0.000 claims description 3
- 230000001376 precipitating effect Effects 0.000 claims description 2
- 239000000470 constituent Substances 0.000 abstract description 9
- 239000000243 solution Substances 0.000 description 129
- 238000006243 chemical reaction Methods 0.000 description 92
- 238000006116 polymerization reaction Methods 0.000 description 82
- 230000015572 biosynthetic process Effects 0.000 description 68
- 239000000976 ink Substances 0.000 description 68
- 238000003786 synthesis reaction Methods 0.000 description 68
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 58
- 239000007787 solid Substances 0.000 description 42
- 229920001577 copolymer Polymers 0.000 description 34
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 32
- 239000002245 particle Substances 0.000 description 22
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical class COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 20
- 238000006386 neutralization reaction Methods 0.000 description 20
- 230000000052 comparative effect Effects 0.000 description 19
- PFHOSZAOXCYAGJ-UHFFFAOYSA-N 2-[(2-cyano-4-methoxy-4-methylpentan-2-yl)diazenyl]-4-methoxy-2,4-dimethylpentanenitrile Chemical compound COC(C)(C)CC(C)(C#N)N=NC(C)(C#N)CC(C)(C)OC PFHOSZAOXCYAGJ-UHFFFAOYSA-N 0.000 description 16
- 229910052757 nitrogen Inorganic materials 0.000 description 16
- 239000007874 V-70 Substances 0.000 description 15
- 230000005587 bubbling Effects 0.000 description 15
- 239000007877 V-601 Substances 0.000 description 14
- 238000003860 storage Methods 0.000 description 14
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 12
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 12
- 125000000524 functional group Chemical group 0.000 description 12
- 238000010526 radical polymerization reaction Methods 0.000 description 11
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 9
- 230000002209 hydrophobic effect Effects 0.000 description 9
- 239000000047 product Substances 0.000 description 9
- OZAIFHULBGXAKX-UHFFFAOYSA-N 2-(2-cyanopropan-2-yldiazenyl)-2-methylpropanenitrile Chemical compound N#CC(C)(C)N=NC(C)(C)C#N OZAIFHULBGXAKX-UHFFFAOYSA-N 0.000 description 8
- WOBHKFSMXKNTIM-UHFFFAOYSA-N Hydroxyethyl methacrylate Chemical compound CC(=C)C(=O)OCCO WOBHKFSMXKNTIM-UHFFFAOYSA-N 0.000 description 8
- 150000001875 compounds Chemical class 0.000 description 8
- 239000000203 mixture Substances 0.000 description 8
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 7
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 7
- 238000011161 development Methods 0.000 description 7
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 7
- 238000007639 printing Methods 0.000 description 7
- 102100026735 Coagulation factor VIII Human genes 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- 101000911390 Homo sapiens Coagulation factor VIII Proteins 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 6
- 230000007423 decrease Effects 0.000 description 6
- 230000000977 initiatory effect Effects 0.000 description 6
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 6
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 5
- SOGAXMICEFXMKE-UHFFFAOYSA-N Butylmethacrylate Chemical compound CCCCOC(=O)C(C)=C SOGAXMICEFXMKE-UHFFFAOYSA-N 0.000 description 5
- 239000002585 base Substances 0.000 description 5
- 230000003287 optical effect Effects 0.000 description 5
- 230000000379 polymerizing effect Effects 0.000 description 5
- 239000004094 surface-active agent Substances 0.000 description 5
- OZAIFHULBGXAKX-VAWYXSNFSA-N AIBN Substances N#CC(C)(C)\N=N\C(C)(C)C#N OZAIFHULBGXAKX-VAWYXSNFSA-N 0.000 description 4
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 239000012528 membrane Substances 0.000 description 4
- 229920005604 random copolymer Polymers 0.000 description 4
- ZUHZGEOKBKGPSW-UHFFFAOYSA-N tetraglyme Chemical compound COCCOCCOCCOCCOC ZUHZGEOKBKGPSW-UHFFFAOYSA-N 0.000 description 4
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 4
- 229940015975 1,2-hexanediol Drugs 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 3
- 150000003973 alkyl amines Chemical class 0.000 description 3
- 239000006229 carbon black Substances 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 235000014113 dietary fatty acids Nutrition 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 239000000194 fatty acid Substances 0.000 description 3
- 229930195729 fatty acid Natural products 0.000 description 3
- 238000009472 formulation Methods 0.000 description 3
- 238000010528 free radical solution polymerization reaction Methods 0.000 description 3
- 238000005227 gel permeation chromatography Methods 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- FHKSXSQHXQEMOK-UHFFFAOYSA-N hexane-1,2-diol Chemical compound CCCCC(O)CO FHKSXSQHXQEMOK-UHFFFAOYSA-N 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 3
- 239000002244 precipitate Substances 0.000 description 3
- 238000001556 precipitation Methods 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 238000001308 synthesis method Methods 0.000 description 3
- 230000002194 synthesizing effect Effects 0.000 description 3
- 229920003169 water-soluble polymer Polymers 0.000 description 3
- 239000001052 yellow pigment Substances 0.000 description 3
- QGKMIGUHVLGJBR-UHFFFAOYSA-M (4z)-1-(3-methylbutyl)-4-[[1-(3-methylbutyl)quinolin-1-ium-4-yl]methylidene]quinoline;iodide Chemical compound [I-].C12=CC=CC=C2N(CCC(C)C)C=CC1=CC1=CC=[N+](CCC(C)C)C2=CC=CC=C12 QGKMIGUHVLGJBR-UHFFFAOYSA-M 0.000 description 2
- 229920002818 (Hydroxyethyl)methacrylate Polymers 0.000 description 2
- WAEVWDZKMBQDEJ-UHFFFAOYSA-N 2-[2-(2-methoxypropoxy)propoxy]propan-1-ol Chemical compound COC(C)COC(C)COC(C)CO WAEVWDZKMBQDEJ-UHFFFAOYSA-N 0.000 description 2
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 2
- NRCMAYZCPIVABH-UHFFFAOYSA-N Quinacridone Chemical compound N1C2=CC=CC=C2C(=O)C2=C1C=C1C(=O)C3=CC=CC=C3NC1=C2 NRCMAYZCPIVABH-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 238000004220 aggregation Methods 0.000 description 2
- 230000002776 aggregation Effects 0.000 description 2
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 229910021529 ammonia Inorganic materials 0.000 description 2
- 239000002280 amphoteric surfactant Substances 0.000 description 2
- 238000010539 anionic addition polymerization reaction Methods 0.000 description 2
- 239000003945 anionic surfactant Substances 0.000 description 2
- 239000012736 aqueous medium Substances 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000011324 bead Substances 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 239000003093 cationic surfactant Substances 0.000 description 2
- 239000012986 chain transfer agent Substances 0.000 description 2
- 238000004140 cleaning Methods 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- 238000003795 desorption Methods 0.000 description 2
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 2
- 229940028356 diethylene glycol monobutyl ether Drugs 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 239000002612 dispersion medium Substances 0.000 description 2
- SUPCQIBBMFXVTL-UHFFFAOYSA-N ethyl 2-methylprop-2-enoate Chemical compound CCOC(=O)C(C)=C SUPCQIBBMFXVTL-UHFFFAOYSA-N 0.000 description 2
- 238000000227 grinding Methods 0.000 description 2
- RBTKNAXYKSUFRK-UHFFFAOYSA-N heliogen blue Chemical compound [Cu].[N-]1C2=C(C=CC=C3)C3=C1N=C([N-]1)C3=CC=CC=C3C1=NC([N-]1)=C(C=CC=C3)C3=C1N=C([N-]1)C3=CC=CC=C3C1=N2 RBTKNAXYKSUFRK-UHFFFAOYSA-N 0.000 description 2
- 150000004679 hydroxides Chemical class 0.000 description 2
- 229910052740 iodine Inorganic materials 0.000 description 2
- 239000011630 iodine Substances 0.000 description 2
- 238000005342 ion exchange Methods 0.000 description 2
- 238000010552 living cationic polymerization reaction Methods 0.000 description 2
- 239000002736 nonionic surfactant Substances 0.000 description 2
- JCGNDDUYTRNOFT-UHFFFAOYSA-N oxolane-2,4-dione Chemical compound O=C1COC(=O)C1 JCGNDDUYTRNOFT-UHFFFAOYSA-N 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 238000010298 pulverizing process Methods 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 230000009257 reactivity Effects 0.000 description 2
- 239000001054 red pigment Substances 0.000 description 2
- 230000003335 steric effect Effects 0.000 description 2
- DTGKSKDOIYIVQL-WEDXCCLWSA-N (+)-borneol Chemical group C1C[C@@]2(C)[C@@H](O)C[C@@H]1C2(C)C DTGKSKDOIYIVQL-WEDXCCLWSA-N 0.000 description 1
- FVQMJJQUGGVLEP-UHFFFAOYSA-N (2-methylpropan-2-yl)oxy 2-ethylhexaneperoxoate Chemical compound CCCCC(CC)C(=O)OOOC(C)(C)C FVQMJJQUGGVLEP-UHFFFAOYSA-N 0.000 description 1
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical compound OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 1
- RRQYJINTUHWNHW-UHFFFAOYSA-N 1-ethoxy-2-(2-ethoxyethoxy)ethane Chemical compound CCOCCOCCOCC RRQYJINTUHWNHW-UHFFFAOYSA-N 0.000 description 1
- IGGDKDTUCAWDAN-UHFFFAOYSA-N 1-vinylnaphthalene Chemical compound C1=CC=C2C(C=C)=CC=CC2=C1 IGGDKDTUCAWDAN-UHFFFAOYSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- LBLYYCQCTBFVLH-UHFFFAOYSA-N 2-Methylbenzenesulfonic acid Chemical compound CC1=CC=CC=C1S(O)(=O)=O LBLYYCQCTBFVLH-UHFFFAOYSA-N 0.000 description 1
- DABQKEQFLJIRHU-UHFFFAOYSA-N 2-Propenoic acid, 2-methyl-, 3,3,5-trimethylcyclohexyl ester Chemical compound CC1CC(OC(=O)C(C)=C)CC(C)(C)C1 DABQKEQFLJIRHU-UHFFFAOYSA-N 0.000 description 1
- COBPKKZHLDDMTB-UHFFFAOYSA-N 2-[2-(2-butoxyethoxy)ethoxy]ethanol Chemical compound CCCCOCCOCCOCCO COBPKKZHLDDMTB-UHFFFAOYSA-N 0.000 description 1
- JDSQBDGCMUXRBM-UHFFFAOYSA-N 2-[2-(2-butoxypropoxy)propoxy]propan-1-ol Chemical compound CCCCOC(C)COC(C)COC(C)CO JDSQBDGCMUXRBM-UHFFFAOYSA-N 0.000 description 1
- ZHCHRKVXJJJZFX-UHFFFAOYSA-N 2-[2-(2-hydroxyethoxy)ethoxy]-1-phenoxyethanol Chemical compound OCCOCCOCC(O)OC1=CC=CC=C1 ZHCHRKVXJJJZFX-UHFFFAOYSA-N 0.000 description 1
- MXVMODFDROLTFD-UHFFFAOYSA-N 2-[2-[2-(2-butoxyethoxy)ethoxy]ethoxy]ethanol Chemical compound CCCCOCCOCCOCCOCCO MXVMODFDROLTFD-UHFFFAOYSA-N 0.000 description 1
- POAOYUHQDCAZBD-UHFFFAOYSA-N 2-butoxyethanol Chemical compound CCCCOCCO POAOYUHQDCAZBD-UHFFFAOYSA-N 0.000 description 1
- WDQMWEYDKDCEHT-UHFFFAOYSA-N 2-ethylhexyl 2-methylprop-2-enoate Chemical compound CCCCC(CC)COC(=O)C(C)=C WDQMWEYDKDCEHT-UHFFFAOYSA-N 0.000 description 1
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 1
- QCAHUFWKIQLBNB-UHFFFAOYSA-N 3-(3-methoxypropoxy)propan-1-ol Chemical compound COCCCOCCCO QCAHUFWKIQLBNB-UHFFFAOYSA-N 0.000 description 1
- MFKRHJVUCZRDTF-UHFFFAOYSA-N 3-methoxy-3-methylbutan-1-ol Chemical compound COC(C)(C)CCO MFKRHJVUCZRDTF-UHFFFAOYSA-N 0.000 description 1
- XPFCZYUVICHKDS-UHFFFAOYSA-N 3-methylbutane-1,3-diol Chemical compound CC(C)(O)CCO XPFCZYUVICHKDS-UHFFFAOYSA-N 0.000 description 1
- 101710141544 Allatotropin-related peptide Proteins 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- 241000284156 Clerodendrum quadriloculare Species 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 description 1
- 125000002877 alkyl aryl group Chemical group 0.000 description 1
- 150000008055 alkyl aryl sulfonates Chemical class 0.000 description 1
- 150000005215 alkyl ethers Chemical class 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 238000010560 atom transfer radical polymerization reaction Methods 0.000 description 1
- MYONAGGJKCJOBT-UHFFFAOYSA-N benzimidazol-2-one Chemical compound C1=CC=CC2=NC(=O)N=C21 MYONAGGJKCJOBT-UHFFFAOYSA-N 0.000 description 1
- AOJOEFVRHOZDFN-UHFFFAOYSA-N benzyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC1=CC=CC=C1 AOJOEFVRHOZDFN-UHFFFAOYSA-N 0.000 description 1
- 238000012662 bulk polymerization Methods 0.000 description 1
- CQEYYJKEWSMYFG-UHFFFAOYSA-N butyl acrylate Chemical compound CCCCOC(=O)C=C CQEYYJKEWSMYFG-UHFFFAOYSA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000001722 carbon compounds Chemical class 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 238000012711 chain transfer polymerization Methods 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000012295 chemical reaction liquid Substances 0.000 description 1
- 238000003776 cleavage reaction Methods 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 150000004696 coordination complex Chemical class 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- XCJYREBRNVKWGJ-UHFFFAOYSA-N copper(II) phthalocyanine Chemical compound [Cu+2].C12=CC=CC=C2C(N=C2[N-]C(C3=CC=CC=C32)=N2)=NC1=NC([C]1C=CC=CC1=1)=NC=1N=C1[C]3C=CC=CC3=C2[N-]1 XCJYREBRNVKWGJ-UHFFFAOYSA-N 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 238000000151 deposition Methods 0.000 description 1
- 229940019778 diethylene glycol diethyl ether Drugs 0.000 description 1
- SBZXBUIDTXKZTM-UHFFFAOYSA-N diglyme Chemical compound COCCOCCOC SBZXBUIDTXKZTM-UHFFFAOYSA-N 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- CZZYITDELCSZES-UHFFFAOYSA-N diphenylmethane Chemical compound C=1C=CC=CC=1CC1=CC=CC=C1 CZZYITDELCSZES-UHFFFAOYSA-N 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000007720 emulsion polymerization reaction Methods 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- TUEYHEWXYWCDHA-UHFFFAOYSA-N ethyl 5-methylthiadiazole-4-carboxylate Chemical compound CCOC(=O)C=1N=NSC=1C TUEYHEWXYWCDHA-UHFFFAOYSA-N 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- FPVGTPBMTFTMRT-NSKUCRDLSA-L fast yellow Chemical compound [Na+].[Na+].C1=C(S([O-])(=O)=O)C(N)=CC=C1\N=N\C1=CC=C(S([O-])(=O)=O)C=C1 FPVGTPBMTFTMRT-NSKUCRDLSA-L 0.000 description 1
- 235000019233 fast yellow AB Nutrition 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- NVVZQXQBYZPMLJ-UHFFFAOYSA-N formaldehyde;naphthalene-1-sulfonic acid Chemical compound O=C.C1=CC=C2C(S(=O)(=O)O)=CC=CC2=C1 NVVZQXQBYZPMLJ-UHFFFAOYSA-N 0.000 description 1
- 238000001879 gelation Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 239000003906 humectant Substances 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 229920001600 hydrophobic polymer Polymers 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 238000007641 inkjet printing Methods 0.000 description 1
- 239000001023 inorganic pigment Substances 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 238000011835 investigation Methods 0.000 description 1
- 150000002497 iodine compounds Chemical class 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 230000002427 irreversible effect Effects 0.000 description 1
- PXZQEOJJUGGUIB-UHFFFAOYSA-N isoindolin-1-one Chemical compound C1=CC=C2C(=O)NCC2=C1 PXZQEOJJUGGUIB-UHFFFAOYSA-N 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 238000003801 milling Methods 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 229910017464 nitrogen compound Inorganic materials 0.000 description 1
- 150000002830 nitrogen compounds Chemical class 0.000 description 1
- AEIJTFQOBWATKX-UHFFFAOYSA-N octane-1,2-diol Chemical compound CCCCCCC(O)CO AEIJTFQOBWATKX-UHFFFAOYSA-N 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000012860 organic pigment Substances 0.000 description 1
- 150000002927 oxygen compounds Chemical class 0.000 description 1
- QBDSZLJBMIMQRS-UHFFFAOYSA-N p-Cumylphenol Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=CC=C1 QBDSZLJBMIMQRS-UHFFFAOYSA-N 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical compound N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 1
- 229920002503 polyoxyethylene-polyoxypropylene Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- FZYCEURIEDTWNS-UHFFFAOYSA-N prop-1-en-2-ylbenzene Chemical class CC(=C)C1=CC=CC=C1.CC(=C)C1=CC=CC=C1 FZYCEURIEDTWNS-UHFFFAOYSA-N 0.000 description 1
- HJWLCRVIBGQPNF-UHFFFAOYSA-N prop-2-enylbenzene Chemical compound C=CCC1=CC=CC=C1 HJWLCRVIBGQPNF-UHFFFAOYSA-N 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 1
- HNJBEVLQSNELDL-UHFFFAOYSA-N pyrrolidin-2-one Chemical compound O=C1CCCN1 HNJBEVLQSNELDL-UHFFFAOYSA-N 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 229910052707 ruthenium Inorganic materials 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000007086 side reaction Methods 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 230000006641 stabilisation Effects 0.000 description 1
- 238000011105 stabilization Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 238000010557 suspension polymerization reaction Methods 0.000 description 1
- 150000003498 tellurium compounds Chemical class 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 229920003176 water-insoluble polymer Polymers 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
- 239000012991 xanthate Substances 0.000 description 1
- 229910052845 zircon Inorganic materials 0.000 description 1
- GFQYVLUOOAAOGM-UHFFFAOYSA-N zirconium(iv) silicate Chemical compound [Zr+4].[O-][Si]([O-])([O-])[O-] GFQYVLUOOAAOGM-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/34—Heterocyclic compounds having nitrogen in the ring
- C08K5/3412—Heterocyclic compounds having nitrogen in the ring having one nitrogen atom in the ring
- C08K5/3432—Six-membered rings
- C08K5/3437—Six-membered rings condensed with carbocyclic rings
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/05—Alcohols; Metal alcoholates
- C08K5/053—Polyhydroxylic alcohols
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/06—Ethers; Acetals; Ketals; Ortho-esters
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/22—Compounds containing nitrogen bound to another nitrogen atom
- C08K5/23—Azo-compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/34—Heterocyclic compounds having nitrogen in the ring
- C08K5/3412—Heterocyclic compounds having nitrogen in the ring having one nitrogen atom in the ring
- C08K5/3415—Five-membered rings
- C08K5/3417—Five-membered rings condensed with carbocyclic rings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0001—Post-treatment of organic pigments or dyes
- C09B67/0004—Coated particulate pigments or dyes
- C09B67/0008—Coated particulate pigments or dyes with organic coatings
- C09B67/0013—Coated particulate pigments or dyes with organic coatings with polymeric coatings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
- C09B67/0041—Blends of pigments; Mixtured crystals; Solid solutions mixtures containing one azo dye
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0071—Process features in the making of dyestuff preparations; Dehydrating agents; Dispersing agents; Dustfree compositions
- C09B67/0084—Dispersions of dyes
- C09B67/0085—Non common dispersing agents
- C09B67/009—Non common dispersing agents polymeric dispersing agent
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D11/00—Inks
- C09D11/02—Printing inks
- C09D11/10—Printing inks based on artificial resins
- C09D11/106—Printing inks based on artificial resins containing macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D11/00—Inks
- C09D11/02—Printing inks
- C09D11/10—Printing inks based on artificial resins
- C09D11/106—Printing inks based on artificial resins containing macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- C09D11/107—Printing inks based on artificial resins containing macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds from unsaturated acids or derivatives thereof
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D11/00—Inks
- C09D11/30—Inkjet printing inks
- C09D11/32—Inkjet printing inks characterised by colouring agents
- C09D11/324—Inkjet printing inks characterised by colouring agents containing carbon black
- C09D11/326—Inkjet printing inks characterised by colouring agents containing carbon black characterised by the pigment dispersant
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/02—Elements
- C08K3/04—Carbon
Definitions
- the present invention relates to a pigment dispersion containing a pigment as a colorant used in an inkjet aqueous pigment ink, and an inkjet aqueous pigment ink obtained using the pigment dispersion.
- Inkjet printers have a wide range of uses, such as personal use, office use, business use, recording use, color display use, and color photography use due to their high functionality. Further, in order to cope with higher speed and higher image quality, the size of ejected droplets (ink droplets) has been miniaturized by improving the apparatus. In order to make the discharged droplets minute, it is necessary to make the pigment (particles) in the ink finer and to finely disperse the finer pigment in the dispersion medium.
- the sharpness, color tone, color density, etc. of printed matter are improving.
- the saturation which is a color value serving as an index indicating the color tone
- the gloss value is improved.
- the ink containing the finer pigment easily penetrates into the paper, the color developability that is the color density tends to decrease.
- the present invention has been made in view of such problems of the prior art, and the object of the present invention is that it can record printed matter with high color development, high saturation, and high gloss, and is also storable.
- Aqueous pigment dispersion capable of preparing an excellent inkjet aqueous pigment ink, an aqueous inkjet pigment ink obtained using the aqueous pigment dispersion, and a dispersant suitable for obtaining the aqueous pigment dispersion It is to provide a method for preparing a coated pigment.
- the present inventors have used the graft copolymer or block copolymer having a predetermined structure obtained by using a specific monomer component as a constituent component, thereby achieving the above-described problems.
- the present inventors have found that this can be solved and have completed the present invention.
- aqueous pigment dispersion for inkjets containing 5 to 35% by weight of a pigment, 0.5 to 25% by weight of a pigment dispersant, 5 to 30% by weight of an aqueous organic solvent, and 20 to 80% by weight of water.
- the pigment dispersant is at least one of a graft copolymer in which a polymer chain A is grafted to a polymer chain B, and a block copolymer in which one end of the polymer chain A and one end of the polymer chain B are bonded, and the polymer
- the chain A is composed of 20 to 60% by mass of the structural unit derived from the first cycloalkyl group-containing (meth) acrylate, 10 to 35% by mass of the structural unit derived from (meth) acrylic acid, and other (meth) acrylates.
- the pigment has a color index number (CI) pigment blue of 15: 3, 15: 4, C.I. I.
- aqueous pigment ink for inkjet comprising the aqueous pigment dispersion according to any one of [1] to [5], wherein the content of the pigment is 4 to 10% by mass.
- a method for preparing a dispersant-coated pigment coated with a pigment dispersant comprising: (i) a pigment dispersion obtained by mixing a pigment, a pigment dispersant, an aqueous organic solvent, water, and an alkali agent.
- the pigment dispersing agent comprises at least a graft copolymer in which a polymer chain A is grafted to a polymer chain B, and a block copolymer in which one end of the polymer chain A and one end of the polymer chain B are bonded.
- the aqueous pigment dispersion of the present invention contains at least one of a graft copolymer having a water-soluble polymer chain A into which a cycloalkyl group is introduced and a block copolymer as a pigment dispersant. Since these graft copolymers and block copolymers have a hydrophobic cycloalkyl group, they have properties such as high glass transition point (Tg), high light resistance and high refractive index. Therefore, by using the aqueous pigment dispersion of the present invention using these graft copolymers and block copolymers as pigment dispersants, it is possible to record printed matter with high color development, high saturation and high gloss, and excellent storage stability. An aqueous pigment ink for ink jet can be prepared.
- the polymer chain B since the polymer chain B has an aromatic ring and / or a cycloalkyl group, it is easily adsorbed on the surface of the hydrophobic pigment and sterically repels with the polymer chain A dissolved in water. This contributes to the stabilization of pigment dispersion.
- the water-insoluble polymer chain B forms particles without dissolving in the ink, thereby reducing the viscosity of the ink and improving the ejection stability because the viscosity of the ink also exhibits Newtonian properties. .
- the water-soluble polymer chain A has an alkali-neutralized carboxyl group, for example, even when the ink is dried in the ink head, it can be easily re-dissolved and re-dispersed, and also has an ejection property. It is good.
- the aqueous pigment ink for inkjet using the aqueous pigment dispersion of the present invention contributes to speeding up printing and improving the image quality of printed matter in inkjet printing.
- the aqueous pigment dispersion of the present invention is an aqueous pigment dispersion for inkjets containing a pigment, a pigment dispersant, an aqueous organic solvent, and water. The details will be described below.
- the pigment dispersant is at least one of a graft copolymer in which the polymer chain A is grafted to the polymer chain B and a block copolymer in which one end of the polymer chain A and one end of the polymer chain B are bonded.
- a graft copolymer in which the polymer chain A is grafted to the polymer chain B and a block copolymer in which one end of the polymer chain A and one end of the polymer chain B are bonded.
- the graft copolymer one or more polymer chains A are bonded (branched) to a polymer chain B which is a main chain.
- the number of bonds of the polymer chain A with respect to one polymer chain B is not limited.
- the polymer chain A is composed of 20 to 60% by mass of structural units derived from the first cycloalkyl group-containing (meth) acrylate, 10 to 35% by mass of structural units derived from (meth) acrylic acid, and other (meth) acrylates. 5 to 70% by mass derived from the structural unit.
- the carboxyl group contained in the structural unit derived from (meth) acrylic acid is ionized by being neutralized with an alkali. For this reason, the polymer chain A containing the structural unit derived from (meth) acrylic acid is a polymer chain having a property of being dissolved in water.
- the polymer chain B includes at least one of a structural unit derived from the second cycloalkyl group-containing (meth) acrylate and a structural unit derived from a vinyl monomer having an aromatic ring, and other (meta) used as necessary. And a structural unit derived from acrylate.
- This polymer chain B is a polymer chain insoluble in water, adsorbs to the pigment by hydrophobic interaction, and deposits to coat (encapsulate) the pigment.
- the pigment dispersant having the polymer chain A and the polymer chain B having such different properties the pigment can be dispersed in a good state.
- the first cycloalkyl group-containing (meth) acrylate constituting the polymer chain A and the second cycloalkyl group-containing (meth) acrylate constituting the polymer chain B may be the same or different.
- the term “cycloalkyl group-containing (meth) acrylate” simply means both “first cycloalkyl group-containing (meth) acrylate” and “second cycloalkyl group-containing (meth) acrylate”. .
- the polymer chain B forms particles, and the polymer chain A is dissolved and stabilized in the aqueous medium in the ink. For this reason, since this pigment dispersant forms particles having high stability and lowers the viscosity, it does not hinder the dispersion stability of the pigment and the ink dischargeability. Furthermore, since the amount of the carboxyl group in the polymer chain A is appropriately controlled, the pigment dispersant has high solubility in water. For this reason, even when the ink head is dried, it can be easily redissolved and redispersed with another aqueous medium such as a cleaning liquid.
- Polymer chain A contains a cycloalkyl group.
- a pigment dispersant containing a polymer chain A having a cycloalkyl group it is possible to prepare an aqueous pigment ink capable of recording a printed matter with high color development, high saturation, and high gloss.
- first cycloalkyl group-containing (meth) acrylate examples include cyclohexyl (meth) acrylate, methylcyclohexyl (meth) acrylate, 3,3,5-trimethylcyclohexyl (meth) acrylate, t-butylcyclohexyl (meth) Examples include acrylate, cyclohexyloxyethyl (meth) acrylate, tricyclodecyl (meth) acrylate, and isobornyl (meth) acrylate. Of these, cyclohexyl (meth) acrylate and 3,3,5-trimethylcyclohexyl (meth) acrylate are preferable.
- the cycloalkyl group preferably has 6 to 9 carbon atoms. This is because if the cycloalkyl group has 6 to 9 carbon atoms, even if it is introduced in a large amount, the water solubility is not hindered so much and it is easy to obtain.
- the proportion of the structural unit derived from the first cycloalkyl (meth) acrylate contained in the polymer chain A is less than 20% by mass, the effect is not exhibited. On the other hand, if it exceeds 60% by mass, water solubility may be significantly reduced.
- the proportion of structural units derived from the first cycloalkyl (meth) acrylate contained in the polymer chain A is preferably 30 to 50% by mass.
- the polymer chain A contains a structural unit derived from (meth) acrylic acid.
- the carboxyl group in this structural unit is neutralized and ionized, and the polymer chain A is dissolved in water.
- the proportion of the structural unit derived from (meth) acrylic acid contained in the polymer chain A is less than 10% by mass, the polymer chain A may not dissolve in water. On the other hand, if it exceeds 35% by mass, the hydrophilicity of the polymer chain A becomes too high, and the water resistance of the resulting printed matter may be significantly reduced.
- the proportion of structural units derived from (meth) acrylic acid contained in the polymer chain A is preferably 15 to 25% by mass.
- Polymer chain A includes “structural units derived from other (meth) acrylates”.
- other (meth) acrylates include aliphatic alkyl (meth) acrylates such as methyl (meth) acrylate, butyl (meth) acrylate, and dodecyl (meth) acrylate; phenyl (meth) acrylate, benzyl (meth) acrylate Aromatic (meth) acrylates such as; hydroxyl group-containing (meth) acrylates such as 2-hydroxyethyl (meth) acrylate and 2-hydroxypropyl (meth) acrylate; ethers such as (poly) ethylene glycol monoalkyl ether (meth) acrylate Examples thereof include a group or chain-containing (meth) acrylate; an amino group-containing (meth) acrylate such as dimethylaminoethyl (meth) acrylate.
- other (meth) acrylates can be used alone or in combination of
- the number average molecular weight of the polymer chain A is 1,000 to 10,000, preferably 2,000 to 7,000. When the number average molecular weight of the polymer chain A is less than 1,000, the performance as a polymer is not exhibited. On the other hand, if the number average molecular weight of the polymer chain A is more than 10,000, the proportion of the hydrophilic chain in the pigment dispersant is too large, and the desorption of the polymer chain B from the pigment is promoted, and the dispersion stability of the pigment is increased. May decrease.
- the number average molecular weight of the polymer chain or polymer in the present specification is a molecular weight in terms of polystyrene by gel permeation chromatography (hereinafter also referred to as “GPC”).
- the polymer chain B is a polymer chain insoluble in water, and has an adsorptivity to the pigment. For this reason, the polymer chain B is adsorbed on the pigment and deposited on the surface to coat (encapsulate) the pigment.
- Specific examples of the second cycloalkyl group-containing (meth) acrylate include the same as those listed as specific examples of the first cycloalkyl group-containing (meth) acrylate.
- the proportion of the structural unit derived from the second cycloalkyl (meth) acrylate contained in the polymer chain B is preferably 30 to 70% by mass, and more preferably 40 to 60% by mass.
- the vinyl monomer having an aromatic ring examples include styrene, vinyl toluene, vinyl naphthalene and the like.
- Specific examples of the (meth) acrylate having an aromatic ring include phenyl (meth) acrylate, naphthoxy (meth) acrylate, benzyl (meth) acrylate, phenoxyethyl (meth) acrylate, paracumylphenol ethylene oxide modified (meth) An acrylate etc. can be mentioned.
- the proportion of the structural unit derived from the vinyl monomer having an aromatic ring or (meth) acrylate contained in the polymer chain B is preferably 30 to 70% by mass, and more preferably 40 to 60% by mass. .
- the polymer chain B includes the above-mentioned “other structural units derived from (meth) acrylate” in order to soften the polymer chain B or introduce a functional group such as a hydroxyl group. Is preferred.
- the number average molecular weights of the graft copolymer and the block copolymer used as the pigment dispersant are both 2,000 to 20,000, preferably 5,000 to 15,000, and 7,000 to 12,000. More preferably it is.
- the number average molecular weight is less than 2,000, the function as a pigment dispersant is lowered and dispersion stability is not maintained.
- the number average molecular weight is more than 20,000, the viscosity of the aqueous pigment dispersion may increase, or a single molecular chain may be adsorbed on a plurality of pigment particles and dispersion may not proceed.
- the ratio of the polymer chain A which is a hydrophilic chain contained in the graft copolymer or block copolymer is too small, the pigment dispersant becomes insoluble or precipitates in water.
- the ratio of the polymer chain A is too large, the water resistance of the printed matter to be recorded decreases, or the adsorptivity to the pigment decreases.
- the ratio of the polymer chain B which is a hydrophobic chain contained in the graft copolymer or block copolymer is too small, the pigment dispersant is not stably adsorbed on the pigment.
- the graft copolymer can be synthesized according to a conventionally known method.
- Specific examples of the synthesis method include: (i) a method of polymerizing a macromonomer (polymer chain A) having an unsaturated bond that undergoes radical polymerization at one end thereof and a monomer that is a constituent component of the polymer chain B (macro).
- Monomer method (ii) a method of polymerizing a monomer that is a constituent of polymer chain A in the presence of polymer chain B to which a polymerization initiating group is bonded (side chain polymerization method); (iii) reactivity at one end thereof Polymer having functional group “Y” in the side chain obtained by polymerizing monomer having functional group “Y” capable of reacting with reactive chain “X” with polymer chain A introduced with group “X”
- polymer reaction method There is a method of preparing the chain B and reacting the polymer chain A and the polymer chain B (polymer reaction method).
- the graft copolymer can be synthesized by any synthesis method, but the macromonomer method is particularly preferable.
- the side chain polymerization method may cause gelation when a coupling reaction, which is a side reaction of radical polymerization, occurs.
- the polymer reaction method is a reaction between polymers, and since the concentration of reactive groups is low, the reaction rate is low and the polymer chain A and the polymer chain B may remain alone.
- the polymer reaction method may be preferable because the terminal reactivity becomes uniform and the graft copolymer is easily obtained by aligning the molecular weight by the living radical polymerization method described later.
- the macromonomer can be synthesized according to a conventionally known method. Specifically, (i) a method of introducing an unsaturated bond at the terminal by polymerizing (meth) acrylate or the like with depolymerization at high temperature and high pressure; (ii) functional groups such as thiol groups and hydroxyl groups.
- a polymer is obtained using a polymerization initiating compound having a functional group such as a hydroxyl group or halogen, and a compound having an unsaturated bond that can react with the functional group is obtained. And pressurized, there is a method to obtain a macromonomer by introducing an unsaturated bond at the terminal.
- a graft copolymer can be obtained.
- a block copolymer is difficult to obtain by a conventionally known radical polymerization method.
- a polymer chain A having a functional group “X” introduced at one end thereof and a polymer chain B having a functional group “Y” capable of reacting with the functional group “X” introduced at one end thereof are prepared.
- By reacting functional groups “X” and “Y”, a block copolymer can be obtained.
- the reaction rate is low, and a large amount of polymer chain A and polymer chain B may remain.
- Preferable methods for synthesizing block copolymers include living cationic polymerization, living anion polymerization, and living radical polymerization.
- the living radical polymerization method is particularly preferable as a method for synthesizing the block copolymer.
- the living radical polymerization method include (i) a method using a compound capable of generating a nitroxide radical (NMP method); (ii) polymerization of a halogenated compound using a metal complex such as copper or ruthenium.
- a method of livingly polymerizing from the polymerization initiating compound as an initiating compound (ATRP method); (iii) a method using a dithiocarboxylic acid ester or a xanthate compound (RAFT method); (iv) an organic tellurium compound and a polymerization initiating compound (V) a method (RTCP method) obtained by using an iodine compound as a polymerization initiating compound and using a phosphorus compound, a nitrogen compound, a carbon compound, an oxygen compound or the like as a catalyst.
- living radical polymerization methods can be carried out under conventionally known polymerization conditions.
- living radical polymerization may be performed under conditions such as bulk polymerization, suspension polymerization, emulsion polymerization, and solution polymerization.
- the reaction liquid after polymerization may be used as it is as a pigment dispersant solution
- the solvent used for polymerization may be replaced with another solvent, or once in a poor solvent. Only the block copolymer may be removed by precipitation.
- polymerization Thereby, it can be easily used as a pigment dispersant simply by adding an alkali to the reaction solution after polymerization to neutralize the block copolymer.
- a pigment dispersant can be obtained.
- the alkali include ammonia; alkylamines such as trimethylamine and triethylamine; glycol amines such as diethanolamine and triethanolamine; cyclic amines such as morpholine and pyridine; hydroxides such as sodium hydroxide and potassium hydroxide.
- the amount of alkali used is preferably equal to or more than that of the carboxyl groups contained in the graft copolymer or block copolymer.
- the pigment one or more of organic pigments and inorganic pigments can be used.
- the pigment include carbon black, quinacridone pigment, phthalocyanine pigment, benzimidazolone pigment, isoindolinone pigment, and azo pigment. More specifically, from the viewpoint of color developability, dispersibility, weather resistance, and the like, the color index number (CI) indicates C.I. I. Pigment Blue-15: 3, 15: 4, C.I. I. Pigment red-122, 269, C.I. I. Pigment violet-19, C.I. I. Pigment yellow-74, 155, 180, C.I. I. Pigment Green-36, 58, C.I. I. Pigment orange-43, and C.I. I. It is preferably at least one selected from the group consisting of CI Pigment Black-7.
- the number average primary particle diameter of the pigment is preferably less than 150 nm.
- a pigment having a number average primary particle size of less than 150 nm it is possible to improve the optical density, saturation, color development, and print quality of the printed matter to be recorded, and moderate precipitation of the pigment in the ink. Can be suppressed.
- the aqueous organic solvent is an organic solvent having miscibility with water.
- Use at least one selected from the group consisting of glycol monoallyl ether, poly (n 1 or more) alkylene (C2-3) glycol dialkyl ether, alkylene diol, alkylene monool monoalkyl ether, alkylene polyol, and amide solvents. It is preferable.
- aqueous organic solvent examples include ethylene glycol monobutyl ether, diethylene glycol monobutyl ether, tripropylene glycol monobutyl ether, tetraethylene glycol monomethyl ether, tetraethylene glycol monobutyl ether, dipropylene glycol monomethyl ether, and tripropylene glycol monomethyl ether.
- the aqueous organic solvent is an essential component, and by containing the aqueous organic solvent, it is possible to prevent the head from drying and the curling of the printed paper.
- another organic solvent can also be used as needed.
- organic solvents include methanol, ethanol, ethylene carbonate, propylene carbonate, and the like.
- the proportion of the pigment contained in the aqueous pigment dispersion of the present invention is 5 to 35% by mass, preferably 5 to 30% by mass, and more preferably 7 to 20% by mass.
- the content ratio of the pigment dispersant is 0.5 to 25% by mass.
- the content of the aqueous organic solvent is 5 to 30% by mass, preferably 5 to 20% by mass.
- the water content is 20 to 80% by mass.
- the blending ratio of each component is appropriately adjusted in consideration of required quality and cost. For example, it is preferable that the pigment content is 7 to 20% by mass because the chroma, color developability and stability are remarkably improved. If the pigment content is too small, the print density cannot be ensured.
- the content ratio of the pigment is too large, the ink is thickened and the ejection stability of the ink from the inkjet head tends to be lowered.
- the pigment content is 5 to 30% by mass
- the pigment dispersant content is 0.5 to 20% by mass
- the aqueous organic solvent content is 10 to 30% by mass, and water is contained.
- the ratio is preferably 50 to 70% by mass.
- the content of the pigment dispersant with respect to 100 parts by mass of the pigment is preferably 10 to 200 parts by mass, and more preferably 15 to 60 parts by mass.
- the content of the pigment dispersant with respect to 100 parts by mass of the pigment is less than 10 parts by mass, the dispersion stability tends to decrease.
- the content of the pigment dispersant with respect to 100 parts by mass of the pigment is more than 200 parts by mass, the viscosity of the aqueous pigment dispersion may increase excessively.
- the aqueous pigment dispersion of the present invention can be prepared, for example, by mixing and dispersing a pigment, a pigment dispersant, an aqueous organic solvent, and water by a conventionally known method.
- a dispersing machine may be used for mixing and dispersing each component.
- Specific examples of the disperser include kneaders, two rolls, three rolls, trade name “Miracle KCK” (manufactured by Asada Steel Corporation), ultrasonic dispersers, high pressure homogenizers (trade name “Microfluidizer”).
- the size of the grinding media of the disperser is reduced, the filling rate of the grinding media is increased, the processing time is increased, and the discharge is performed.
- Methods such as slowing down the speed, classifying with a filter or centrifuge after pulverization, and the like are used.
- a pigment finely divided in advance by a conventionally known method such as a salt milling method.
- a dispersant-coated pigment that has been coated with a pigment dispersant, that is, a dispersant-coated pigment in which the pigment dispersant is deposited on the surface thereof to coat (encapsulate) the pigment.
- a dispersant-coated pigment By using such a dispersant-coated pigment, the dispersion stability of the pigment can be further increased. That is, by coating the pigment with the pigment dispersant, even if a large amount of the organic solvent is mixed, the pigment dispersant is not easily detached from the surface of the pigment, and furthermore, the polymer chain A is dissolved in water. The dispersion stability of the pigment can be further increased.
- the dispersant-coated pigment includes, for example, (i) a step of adding an acid to a pigment dispersion obtained by mixing a pigment, a pigment dispersant, an aqueous organic solvent, water, and an alkali agent to precipitate the pigment dispersant, Or (ii) It can prepare by the preparation method including the process of adding the kneaded material obtained by knead
- each component is mixed by a conventionally known method to obtain a pigment dispersion in which the pigment is dispersed.
- the alkali agent include ammonia; alkylamines such as trimethylamine and triethylamine; glycol-based amines such as diethanolamine and triethanolamine; cyclic amines such as morpholine and pyridine; hydroxides such as sodium hydroxide and potassium hydroxide. it can.
- the obtained pigment dispersion is stirred using a stirrer capable of stirring at a high speed such as a dissolver, and the acid is gradually added.
- the pigment dispersant can be deposited on the surface of the pigment, and the pigment can be coated (encapsulated) with the polymer chain B which is a hydrophobic chain.
- the acid inorganic acids such as hydrochloric acid, sulfuric acid and nitric acid; organic acids such as acetic acid, propionic acid and toluenesulfonic acid can be used.
- the acid may be added as it is, but it is preferably added in an aqueous solution of 10% by mass or less. Further, the amount of the acid added is preferably equimolar or more, more preferably 1.1 times or more moles of the alkali neutralizing the carboxyl group of the polymer chain A of the pigment dispersant.
- the kneaded product obtained by kneading the pigment and the pigment dispersant is added to the poor solvent for the pigment dispersant.
- the pigment dispersant By adding to the poor solvent of the pigment dispersant, the pigment dispersant can be deposited on the surface of the pigment, and the pigment can be coated (encapsulated) with the polymer chain B which is a hydrophobic chain.
- the poor solvent a solvent that does not dissolve the polymer is used although it depends on the properties derived from the composition of the pigment dispersant.
- Specific examples of such a poor solvent include hydrocarbon solvents such as hexane, polyhydric alcohols such as ethylene glycol, and methanol.
- the formed dispersant-coated pigment (precipitate) is filtered.
- a dispersant-coated pigment (precipitate) can be obtained in a water paste state.
- the water paste may be dried and pulverized, but it is also preferable to use the water paste as it is.
- the water paste By using the water paste as it is, there is no need to fuse the pigment dispersant due to drying, and there is no need for pulverization. Therefore, the number average particle diameter of the pigment can be maintained as it is during dispersion.
- after precipitation of a pigment dispersant it may be heated as necessary to aggregate the dispersant-coated pigment and facilitate filtration.
- the aqueous pigment ink for ink-jet recording of the present invention contains the aforementioned aqueous pigment dispersion, and the pigment content is 4 to 10% by mass.
- the aqueous pigment ink for inkjet according to the present invention contains, for example, a vehicle component as a component other than the aqueous pigment dispersion.
- vehicle component include surfactants, organic solvents, and humectants.
- the surface tension of the aqueous pigment ink for inkjet is preferably 20 to 40 mN / m.
- the surface tension of the aqueous pigment ink for inkjet can be adjusted by adding a surfactant.
- a surfactant an anionic surfactant, a nonionic surfactant, a cationic surfactant, and an amphoteric surfactant can be used.
- anionic surfactants include alkyl sulfate salts, alkyl aryl sulfate salts, alkyl aryl sulfonates, alkyl naphthalene sulfonates, polyoxyethylene alkyl ether sulfonates, polyoxyethylene alkyl aryl ether sulfones.
- examples thereof include acid salts, naphthalenesulfonic acid formalin condensates, polyoxyethylene alkyl phosphoric acid ester salts, polyoxyethylene alkyl aryl phosphoric acid ester salts, and the like.
- nonionic surfactant examples include polyoxyethylene alkyl ether, polyoxyethylene alkyl aryl ether, polyoxyethylene polyoxypropylene block polymer, sorbitan fatty acid ester, polyoxyethylene alkylamine ether, fatty acid diethanoldiamide, sorbitan Examples include fatty acid esters, acetylene alcohols, and acetylene glycols.
- Specific examples of the cationic surfactant include alkylamine salts and quaternary ammonium salts.
- amphoteric surfactants include alkyl betaines and amine oxides.
- the content ratio of the surfactant in the aqueous pigment ink for inkjet is preferably 0.01 to 5% by mass, and more preferably 0.1 to 2% by mass. If the content ratio of the surfactant is too large, the dispersion stability of the pigment may be impaired.
- Mn number average molecular weight (hereinafter referred to as “Mn”) of the macromonomer MM-1 measured using a GPC differential refractometer (hereinafter referred to as “RI”) is 6,800, and the weight average molecular weight (hereinafter referred to as “Mn”).
- Mw was 10,900, and the degree of dispersion (Mw / Mn) (hereinafter referred to as “PDI”) was 1.60. Further, in the ultraviolet absorption detector (wavelength 254 nm) (hereinafter referred to as “UV detector”), almost no peak was observed.
- UV detector ultraviolet absorption detector
- the resulting polymer solution was poured into a large amount of water to precipitate a polymer, and then filtered and washed. After dissolving in THF, it was again poured into a large amount of water to precipitate a polymer, and then filtered and washed.
- the polymer was obtained by drying with a dryer at 50 ° C. for 24 hours.
- 1H-NMR of the polymer obtained using a nuclear magnetic resonance apparatus was measured, a monomer peak and an EBMA-derived unsaturated bond proton peak were observed at 6 ppm and 6.4 ppm, respectively. For this reason, the obtained polymer is considered to be a macromonomer having an unsaturated bond at the terminal. Also in the following synthesis examples, it was confirmed that the polymer obtained by performing the same measurement was a macromonomer.
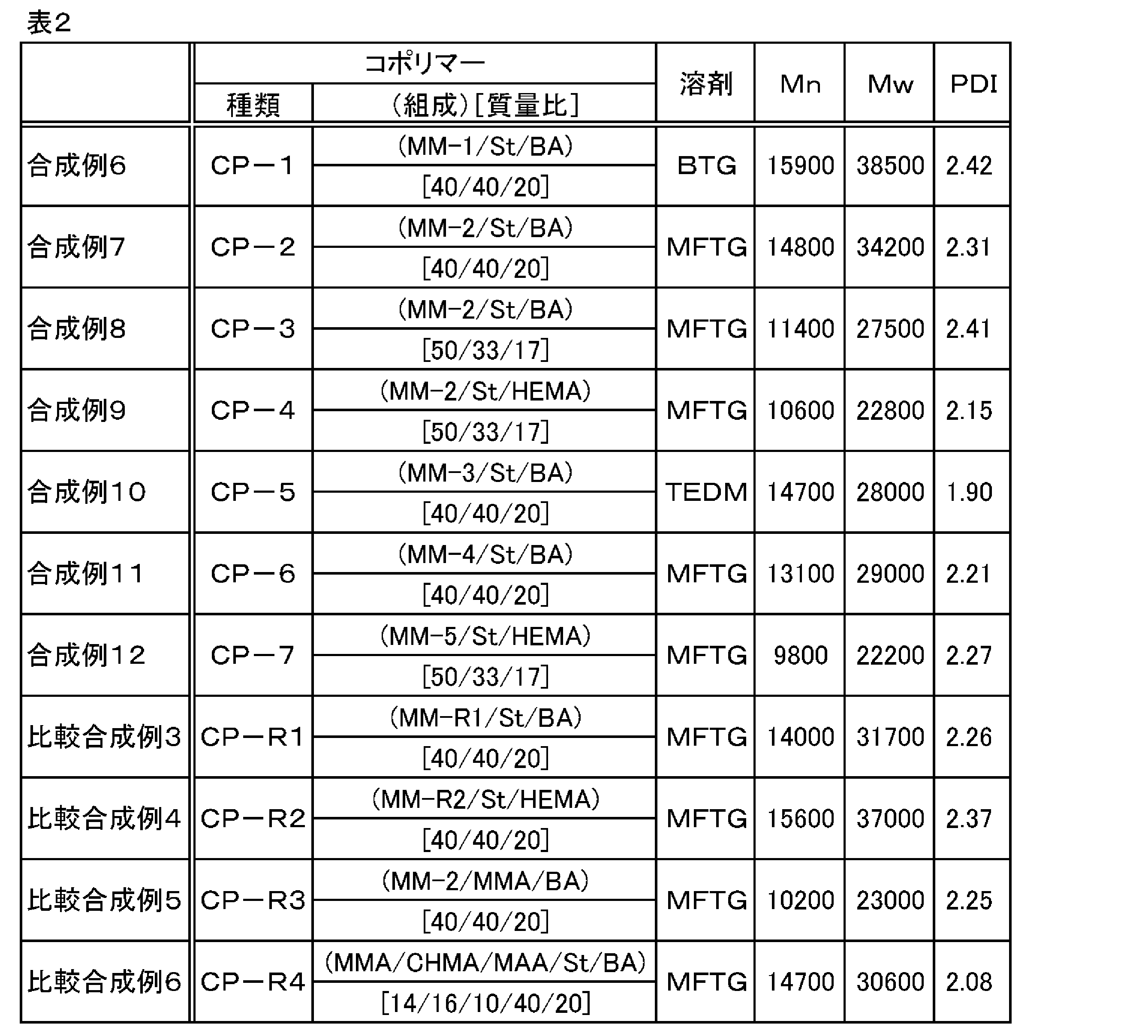
- Synthesis Example 2 In a reaction vessel similar to that used in Synthesis Example 1, 250 parts of tripropylene glycol monomethyl ether (hereinafter referred to as “MFTG”), 30 parts of MMA, 40 parts of CHMA, 30 parts of MAA, 2.5 parts of EBMA, and V-601 were added. One copy was loaded. After polymerization for 3 hours at 75 ° C. with nitrogen bubbling, 0.5 part of V-601 was added. Polymerization was further performed for 4.5 hours to obtain a polymer solution containing a polymer (macromonomer MM-2). The obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content. In addition, Mn of the macromonomer MM-2 was 6,400, Mw was 10,200, and PDI was 1.59.
- MFTG tripropylene glycol monomethyl ether
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- Mn of the macromonomer MM-3 was 6,500
- Mw was 10,400
- PDI was 1.60.
- Synthesis Example 4 In a reaction vessel similar to that used in Synthesis Example 1, 250 parts of MFTG, 36 parts of MMA, 40 parts of CHMA, 24 parts of MAA, 3.5 parts of EBMA, and 1 part of V-601 were charged. After polymerization for 3 hours at 75 ° C. with nitrogen bubbling, 0.5 part of V-601 was added. Polymerization was further performed for 4.5 hours to obtain a polymer solution containing a polymer (macromonomer MM-4). The obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content. In addition, Mn of the macromonomer MM-4 was 5,400, Mw was 8,500, and PDI was 1.57.
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content. Further, Mn of the macromonomer MM-5 was 5,700, Mw was 9,700, and PDI was 1.70.
- This macromonomer MM-R2 is a macromonomer having no cycloalkyl group.
- Synthesis Example 7 In a reaction vessel A similar to that used in Synthesis Example 1, 50 parts of MFTG and 300 parts of a solution of macromonomer MM-2 were charged and heated to 80 ° C. Further, St 100 parts, 50 parts of BA, and 2.5 parts of PBO were charged into another reaction vessel and stirred well to prepare a monomer solution. After adding 1 ⁇ 2 of this monomer solution to the reaction vessel A, the remaining 1 ⁇ 2 was slowly added dropwise over 1 hour. After completion of dropping, polymerization was performed for 3 hours. 1.25 parts of PBO was added, heated to 85 ° C., and further polymerized for 4 hours.
- Synthesis Example 8 A reaction vessel A similar to that used in Synthesis Example 1 was charged with 300 parts of the macromonomer MM-2 solution and heated to 80 ° C. Moreover, St67 parts, 33 parts of BA, and 2 parts of PBO were charged into another reaction vessel and stirred well to prepare a monomer solution. After adding 1 ⁇ 2 of this monomer solution to the reaction vessel A, the remaining 1 ⁇ 2 was slowly added dropwise over 1 hour. After completion of dropping, polymerization was performed for 3 hours. One part of PBO was added, heated to 85 ° C., and further polymerized for 4 hours. 16.1 parts of KOH and 183.9 parts of water were added for neutralization to obtain a polymer solution containing a polymer (copolymer CP-3).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- Mn of copolymer CP-3 was 11,400, Mw was 27,500, and PDI was 2.41.
- Synthesis Example 9 A reaction vessel A similar to that used in Synthesis Example 1 was charged with 300 parts of the macromonomer MM-2 solution and heated to 80 ° C. Moreover, St67 part, 33 parts of HEMA, and 2 parts of PBO were charged in another reaction container, and it stirred well, and the monomer liquid was prepared. After adding 1 ⁇ 2 of this monomer solution to the reaction vessel A, the remaining 1 ⁇ 2 was slowly added dropwise over 1 hour. After completion of dropping, polymerization was performed for 3 hours. One part of PBO was added, heated to 85 ° C., and further polymerized for 4 hours. 16.1 parts of KOH and 183.9 parts of water were added for neutralization to obtain a polymer solution containing a polymer (copolymer CP-4).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- Mn of copolymer CP-4 was 10,600, Mw was 22,800, and PDI was 2.15.
- Synthesis Example 10 In a reaction vessel A similar to that used in Synthesis Example 1, 50 parts of TEDM and 300 parts of a solution of macromonomer MM-3 were charged and heated to 80 ° C. Further, St 100 parts, 50 parts of BA, and 2.5 parts of PBO were charged into another reaction vessel and stirred well to prepare a monomer solution. After adding 1 ⁇ 2 of this monomer solution to the reaction vessel A, the remaining 1 ⁇ 2 was slowly added dropwise over 1 hour. After completion of dropping, polymerization was performed for 3 hours. 1.25 parts of PBO was added, heated to 85 ° C., and further polymerized for 4 hours.
- Synthesis Example 11 In a reaction vessel A similar to that used in Synthesis Example 1, 50 parts of MFTG and 300 parts of a solution of macromonomer MM-4 were charged and heated to 80 ° C. Further, St 100 parts, 50 parts of BA, and 2.5 parts of PBO were charged into another reaction vessel and stirred well to prepare a monomer solution. After adding 1 ⁇ 2 of this monomer solution to the reaction vessel A, the remaining 1 ⁇ 2 was slowly added dropwise over 1 hour. After completion of dropping, polymerization was performed for 3 hours. 1.25 parts of PBO was added, heated to 85 ° C., and further polymerized for 4 hours.
- copolymer CP-6 16.2 parts of KOH and 233.8 parts of water were added for neutralization to obtain a polymer solution containing a polymer (copolymer CP-6). The obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content. In addition, Mn of copolymer CP-6 was 13,100, Mw was 29,000, and PDI was 2.21.
- Synthesis Example 12 In a reaction vessel A similar to that used in Synthesis Example 1, 50 parts of MFTG and 300 parts of a solution of macromonomer MM-5 were charged and heated to 80 ° C. Moreover, St67 part, 33 parts of HEMA, and 2 parts of PBO were charged in another reaction container, and it stirred well, and the monomer liquid was prepared. After adding 1 ⁇ 2 of this monomer solution to the reaction vessel A, the remaining 1 ⁇ 2 was slowly added dropwise over 1 hour. After completion of dropping, polymerization was performed for 3 hours. One part of PBO was added, heated to 85 ° C., and further polymerized for 4 hours.
- copolymer CP-7 16.1 parts of KOH and 183.9 parts of water were added for neutralization to obtain a polymer solution containing a polymer (copolymer CP-7). The obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- Mn of copolymer CP-7 was 9,800, Mw was 22,200, and PDI was 2.27.
- copolymer CP-R1 16.2 parts of KOH and 233.8 parts of water were added for neutralization to obtain a polymer solution containing a polymer (copolymer CP-R1). The obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content. In addition, Mn of copolymer CP-R1 was 14,000, Mw was 31,700, and PDI was 2.26. This copolymer CP-R1 is a graft copolymer that does not have a cycloalkyl group in the grafted polymer.
- copolymer CP-R2 16.2 parts of KOH and 233.8 parts of water were added for neutralization to obtain a polymer solution containing a polymer (copolymer CP-R2).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- Mn of copolymer CP-R2 was 15,600, Mw was 37,000, and PDI was 2.37.
- the copolymer CP-R2 is a graft copolymer having no cycloalkyl group in the grafted polymer (polymer chain A).
- copolymer CP-R3 16.2 parts of KOH and 233.8 parts of water were added for neutralization to obtain a polymer solution containing a polymer (copolymer CP-R3).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted from the nonvolatile content to be 85%.
- Mn of copolymer CP-R3 was 10,200, Mw was 23,000, and PDI was 2.25.
- This copolymer CP-R3 is a graft copolymer having no aromatic ring or cycloalkyl group in the main chain (polymer chain B).
- copolymer CP-R4 16.2 parts of KOH and 233.8 parts of water were added for neutralization to obtain a polymer solution containing a polymer (copolymer CP-R4).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- the copolymer CP-R4 had an Mn of 14,700, an Mw of 30,600, and a PDI of 2.08. This copolymer CP-R4 is a random copolymer.
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted from the nonvolatile content to be 86%.
- the polymer contained in this polymer solution had an Mn of 5,000 and a PDI of 1.19.
- the polymer solution was cooled to 40 ° C., and 16.8 parts of CHMA, 20 parts of MMA, 12.9 parts of MAA, and 1.5 parts of V-70 were added and polymerized for 3.5 hours. 8.4 parts of KOH and 49.2 parts of water were added for neutralization to obtain a polymer solution containing a polymer (block copolymer BP-1).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content. Further, the Mn of the block polymer BP-1 was 10,300, and the PDI was 1.30.
- ion-exchange water was added to the obtained polymer solution, and solid content concentration was adjusted to 30%. In the following synthesis examples, the solid concentration was similarly adjusted to 30%.
- the polymer solution was cooled to 40 ° C., and 16.8 parts of TMCHMA, 20 parts of MMA, 12.9 parts of MAA, and 1.5 parts of V-70 were added and polymerized for 3.5 hours. 8.4 parts of KOH and 49.2 parts of water were added for neutralization to obtain a polymer solution containing a polymer (block copolymer BP-2).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- the block copolymer BP-2 had an Mn of 10,300 and a PDI of 1.31.
- the polymer solution was cooled to 40 ° C., 8.4 parts of CHMA, 25 parts of MMA, 12.9 parts of MAA, and 1.4 parts of V-70 were added and polymerized for 3.5 hours. 8.4 parts of KOH and 47.6 parts of water were added for neutralization to obtain a polymer solution containing a polymer (block copolymer BP-3).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- the block copolymer BP-3 had a Mn of 9,100 and a PDI of 1.31.
- the polymer solution was cooled to 40 ° C., 16.8 parts of CHMA, 20 parts of MMA, 12.9 parts of MAA, and 1.5 parts of V-70 were added and polymerized for 3.5 hours. 8.4 parts of KOH and 47.6 parts of water were added for neutralization to obtain a polymer solution containing a polymer (block copolymer BP-4).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted from the nonvolatile content to be 100%.
- the block copolymer BP-4 had an Mn of 9,600 and a PDI of 1.33.
- Synthesis Example 17 A reaction vessel similar to that used in Synthesis Example 1 was charged with 173 parts of BTG, 1.0 part of iodine, 3.7 parts of V-70, 42 parts of CHMA, 17.6 parts of BzMA, and 0.17 part of DPM. Polymerization was carried out at 45 ° C. for 5.5 hours with nitrogen bubbling to obtain a polymer solution. The obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion was converted from the nonvolatile content to be 89%. Further, the polymer contained in this polymer solution had Mn of 6,000 and PDI of 1.18.
- the polymer solution was cooled to 40 ° C., 16.8 parts of CHMA, 20 parts of MMA, 12.9 parts of MAA, and 1.5 parts of V-70 were added and polymerized for 3.5 hours. 6.0 parts of NaOH and 51.6 parts of water were added for neutralization to obtain a polymer solution containing a polymer (block copolymer BP-5).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- the block copolymer BP-5 had an Mn of 11,100 and a PDI of 1.29.
- Synthesis Example 18 A reaction vessel similar to that used in Synthesis Example 1 was charged with 172 parts of TEDM, 1.0 part of iodine, 3.7 parts of V-70, 58.8 parts of CHMA, and 0.17 part of DPM. Polymerization was carried out at 45 ° C. for 5.5 hours with nitrogen bubbling to obtain a polymer solution. The obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion was converted from the nonvolatile content to be 84%. Further, the Mn of the polymer contained in this polymer solution was 5,200, and the PDI was 1.19.
- the polymer solution was cooled to 40 ° C., 16.8 parts of CHMA, 20 parts of MMA, 12.9 parts of MAA, and 1.5 parts of V-70 were added and polymerized for 3.5 hours. 9.1 parts of 28% aqueous ammonia and 48.2 parts of water were added for neutralization to obtain a polymer solution containing a polymer (block copolymer BP-6).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- the block copolymer BP-6 had an Mn of 10,000 and a PDI of 1.31.
- the polymer solution was cooled to 40 ° C., 16.8 parts of CHMA, 20 parts of MMA, 12.9 parts of MAA, and 1.5 parts of V-70 were added and polymerized for 3.5 hours. 8.4 parts of KOH and 47.6 parts of water were added for neutralization to obtain a polymer solution containing a polymer (block copolymer BP-7).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was converted to 100% from the nonvolatile content.
- the block copolymer BP-7 had a Mn of 9,500 and a PDI of 1.29.
- the polymer solution was cooled to 40 ° C., 20.8 parts of MMA, 40.8 parts of BMA, 15.0 parts of MAA, and 2.3 parts of V-70 were added and polymerized for 3.5 hours. 9.8 parts of KOH and 32.8 parts of water were added for neutralization to obtain a polymer solution containing a polymer (block copolymer BP-R1).
- the obtained polymer solution was sampled, the solid content concentration was measured, and the polymerization conversion rate was calculated from the nonvolatile content, which was 100%.
- the block copolymer BP-R1 had an Mn of 9,200 and a PDI of 1.57.
- the random copolymer RP-R1 had an Mn of 12,100 and a PDI of 2.28.
- Example 1 Aqueous pigment dispersion (Example 1) 233.3 parts of a polymer solution containing the copolymer CP-1 obtained in Synthesis Example 6, 70 parts of diethylene glycol monobutyl ether, and 311.7 parts of water were mixed to obtain a translucent solution having a slight turbidity. To this solution, 350 parts of azo yellow pigment PY-74 (trade name “Seikafast Yellow 2016G”, manufactured by Dainichi Seika Kogyo Co., Ltd.) was added and stirred for 30 minutes using a disper to prepare a mill base.
- azo yellow pigment PY-74 trade name “Seikafast Yellow 2016G”, manufactured by Dainichi Seika Kogyo Co., Ltd.
- a horizontal media disperser (trade name “Dynomill 0.6 liter ECM type”, manufactured by Shinmaru Enterprises Co., Ltd., zirconia bead diameter 0.5 mm), dispersed at a peripheral speed of 10 m / s, and pigment in the mill base was sufficiently dispersed. Thereafter, 316 parts of water were added to make the pigment concentration 18%.
- the mill base taken out from the disperser was centrifuged (7500 rpm, 20 minutes), and then filtered through a 10 ⁇ m membrane filter. Dilution with water gave an aqueous pigment dispersion 1 for inkjet having a pigment concentration of 14%.
- the number average particle size of the pigment contained in the obtained aqueous pigment dispersion 1 was 122 nm as measured with a particle size measuring device (trade name “NICOMP 380ZLS-S”, manufactured by International Business Corporation), and the pigment was finely dispersed. Confirmed that it has been. Further, the viscosity of the aqueous pigment dispersion 1 was 3.1 mPa ⁇ s, and the pH was 8.9. When this aqueous pigment dispersion 1 was stored at 70 ° C. for 1 week, the number average particle size of the pigment was 122 nm and the viscosity was 3.0 mPa ⁇ s, and it was confirmed that the storage stability was very good.
- Examples 2 to 14, Comparative Examples 1 to 6 Aqueous pigment dispersions 2 to 20 were obtained in the same manner as in Example 1 except that the pigment dispersant shown in Table 4 was used. The evaluation results of the obtained aqueous pigment dispersions 2 to 20 are shown in Table 4.
- aqueous pigment dispersions 2 to 16 and 19 obtained in Examples 2 to 14 and Comparative Examples 1, 2 and 5 the pigment is finely dispersed in the same manner as the aqueous pigment dispersions obtained in Example 1.
- the storage stability was good.
- the aqueous pigment dispersions 15, 16, and 19 obtained in Comparative Examples 1, 2, and 5 are all the same pigments as those used in the examples except that the structure does not contain a cycloalkyl group. Since a dispersant is used, it is considered that dispersibility and storage stability are improved.
- the aqueous pigment dispersion of Comparative Example 3 using the copolymer CP-R3 having no aromatic ring or cycloalkyl group in the polymer chain B (main chain) has a high viscosity during dispersion and is difficult to take out. It was necessary to dilute with water to lower the pigment concentration. Further, the dispersibility was low, the number average particle diameter of the pigment was large, and the storage stability was not good. It is presumed that this is because the polymer chain B (main chain) has a poor pigment adsorptivity.
- the aqueous pigment dispersions of Comparative Examples 4 and 6 using the copolymer CP-R4 and the random copolymer RP-R1 had good dispersibility, but the pigment particles aggregated during storage and the fluidity was remarkable. Declined. This is because the use of a copolymer having a random structure in which the adsorbed portion is randomly present in the molecular chain as the pigment dispersant causes one molecule of the pigment dispersant to be adsorbed on a plurality of pigment particles, and It is considered that the dispersion did not proceed well and the heating was performed, so that the influence of the solvent became remarkable, and the pigment dispersant was not adsorbed on the pigment and the pigment was aggregated.
- the molecular structure of the pigment dispersant used in the examples is that the adsorption part to the pigment and the solvent-soluble part are clearly separated in block units, so that the dispersion of the pigment proceeds in a good state and the solvent dissolution It is considered that the aggregation of the pigment was suppressed by the steric effect of the sexing part, and the storage stability was improved.
- copper phthalocyanine pigment PB-15: 3 (trade name “Cyanine Blue A220JC, manufactured by Dainichi Seika Kogyo Co., Ltd.), quinacridone pigment PR-122 (trade name“ CFR130P ”, large Nissei Kagaku Kogyo Co., Ltd.) and carbon black pigment PB-7 (trade name “S170”, manufactured by Degussa) were used in the same manner as in Examples 1 to 14 and Comparative Examples 1 to 6 described above.
- a blue aqueous pigment dispersion, a red aqueous pigment dispersion, and a black aqueous pigment dispersion were prepared.
- the aqueous pigment dispersions obtained using the copolymers CP-1 to CP-7, block copolymers BP-1 to BP-7, CP-R1, CP-R2, and BP-R1 are all yellow aqueous pigments. Similar to the dispersion, the dispersibility and storage stability were good.
- Example 15 Using the aqueous pigment dispersion 1 prepared in Example 1, an aqueous pigment ink for inkjet was prepared according to the following formulation.
- Aqueous pigment dispersion 1 40 parts Water 42.2 parts 1,2-hexanediol 5 parts Glycerin 10 parts Product name “Surfinol 465” (manufactured by Air Products) 1 part
- an aqueous pigment ink 1 After thoroughly mixing the composition of the above formulation, it was filtered through a membrane filter having a pore size of 10 ⁇ m to prepare an aqueous pigment ink 1.
- the number average particle size of the pigment contained in the aqueous pigment ink 1 was measured and found to be 119 nm. Moreover, the viscosity of the aqueous pigment ink 1 was 2.9 mPa ⁇ s.
- this aqueous pigment ink 1 was stored at 70 ° C. for 1 week, the number average particle diameter of the pigment was 118 nm and the viscosity was 2.8 mPa ⁇ s, and it was confirmed that the storage stability was very good. This is presumed that the storage stability was improved because the hydrophobic adsorbing portion of the pigment dispersant was adsorbed on the pigment without desorption.
- Example 16 to 28 Comparative Examples 7 to 9
- Aqueous pigment inks 2 to 16 and 19 were obtained in the same manner as in Example 15 except that the pigment dispersion shown in Table 5 was used.
- aqueous pigment inks of respective colors were prepared using the above-described blue aqueous pigment dispersion, red aqueous pigment dispersion, and black aqueous pigment dispersion, respectively. It was confirmed that any of the aqueous pigment inks had good storage stability.
- Each of the aqueous pigment inks 1 to 16 and 19 is filled in a cartridge, and an ink jet printer (trade name “EM930C”, manufactured by Seiko Epson Corporation) is used, (i) dedicated photographic glossy paper (PGPP), (ii) plain paper (Trade name “4024”, manufactured by Xerox Co., Ltd.) (iii) A printed matter was obtained by printing on a dedicated photo matte paper in a photo 720 dpi print mode. As a result, it was confirmed that any of the aqueous pigment inks can be ejected from the inkjet nozzles without any problem.
- optical densitometer (trade name “Macbeth RD-914”, manufactured by Macbeth Co., Ltd.) was used to evaluate the obtained printed matter.
- the optical density OD value, saturation C *, 20 degree gloss, and 60 degree gloss were measured for the dedicated photographic glossy paper (PGPP).
- PGPP dedicated photographic glossy paper
- the optical density OD value and the saturation C * were measured five times, and the average values were obtained. Table 5 shows the measurement results.
- Example 29 A uniform solution was obtained by mixing 164 parts of the polymer solution containing the block copolymer BP-1 obtained in Synthesis Example 13, 80 parts of BDG, and 356 parts of water. To the resulting solution, 200 parts of a red pigment (CI Pigment Red 122 (dimethylquinacridone pigment, manufactured by Dainichi Seika Kogyo Co., Ltd.)) was added and peptized using a disper to prepare a mill base. To 800 parts of the obtained mill base, 3200 parts of water was added so that the pigment content was 5%, and 5% acetic acid was added dropwise with stirring to precipitate the pigment dispersant. The pH before acetic acid dropping (initial) was 9.5, and the pH after acetic acid dropping was 4.5. Filtration and washing with water gave a dispersant-coated pigment paste (solid content concentration: 32.0%).
- CI Pigment Red 122 dimethylquinacridone pigment, manufactured by Dainichi Seika Kogyo Co., Ltd.
- a solution obtained by dissolving 667 parts of the obtained paste, 9.4 parts of BDG, and 1.15 parts of sodium hydroxide in 62.2 parts of water was mixed and stirred. Subsequently, it was dispersed again using a horizontal media disperser. Furthermore, an ultra-high pressure homogenizer (trade name “Microfluidizer”, manufactured by Microfluidics Co., Ltd.) was used and dispersed in three passes at a pressure of 150 MPa. After centrifugal separation (7500 rpm, 20 minutes), the mixture was filtered through a 10 ⁇ m membrane filter, and then ion-exchanged water was added to obtain a red aqueous pigment dispersion 1 having a pigment concentration of 14%.
- an ultra-high pressure homogenizer trade name “Microfluidizer”, manufactured by Microfluidics Co., Ltd.
- red aqueous pigment dispersion liquid 1 It was 108 nm when the number average particle diameter of the pigment contained in the obtained red aqueous pigment dispersion liquid 1 was measured. Further, the viscosity of the red aqueous pigment dispersion 1 was 2.22 mPa ⁇ s. When this red aqueous pigment dispersion 1 was stored at 70 ° C. for 1 week, no change was observed in the particle diameter and viscosity of the pigment, and it was confirmed that the storage stability was good.
- red aqueous pigment dispersion 1 For 40 parts of red aqueous pigment dispersion 1, 1.8 parts of BDG, 5 parts of 1,2-hexanediol, 10 parts of glycerin, 1 part of “Surfinol 465” (trade name, manufactured by Air Products), and water 42 . 60 parts of 2 parts of the mixture were added. After sufficiently stirring, the mixture was filtered through a membrane filter having a pore size of 10 ⁇ m to obtain a red aqueous pigment ink for inkjet. The number average particle diameter of the pigment particles contained in the obtained red aqueous pigment ink was 114 nm. The viscosity of the red aqueous pigment ink was 3.06 mPa ⁇ s.
- Example 30 to 32 instead of the red pigment, (i) azo yellow pigment PY-74 (trade name “Seika Fast Yellow 2016G”, manufactured by Dainichi Seika Kogyo Co., Ltd.), (ii) copper phthalocyanine pigment PB-15: 3 (trade name “cyanine” Blue A220JC ”(manufactured by Dainichi Seika Kogyo Co., Ltd.), (iii) Carbon black pigment PB-7 (trade name“ S170 ”, manufactured by Degussa Co., Ltd.) was used, respectively, and the ink formulation was as shown in Table 6. Except for this, each color aqueous pigment dispersion and each color aqueous pigment ink for inkjet were obtained in the same manner as in Example 29 described above.
- azo yellow pigment PY-74 trade name “Seika Fast Yellow 2016G”, manufactured by Dainichi Seika Kogyo Co., Ltd.
- copper phthalocyanine pigment PB-15: 3 trade name “cyanine” Blue A
- Table 7 shows the measurement results of the number average particle diameter and viscosity (initially and after storage at 70 ° C. for 1 week) of the pigments of the respective color aqueous pigment inks obtained in Examples 29 to 32.
- Each color aqueous pigment ink is filled in a cartridge, and an inkjet printer (trade name “EM930C”, manufactured by Seiko Epson Corporation) is used on plain paper (trade name “4024”, manufactured by Xerox Corporation) in a print mode of 720 dpi
- a printed matter was obtained by printing.
- an optical densitometer (trade name “Macbeth RD-914”, manufactured by Macbeth Co., Ltd.), the print density of the obtained printed matter was measured five times to calculate an average value. The results are shown in Table 7.
- the pigment coated (encapsulated) with the pigment dispersant also showed good dispersibility and storage stability. This is because the hydrophobic polymer chain B coats the pigment so that the pigment dispersant is not peeled off even by the solvent of the ink, and the water-soluble polymer chain A dissolves in water and prevents aggregation due to steric effects. This is presumed to be due to this. It was also confirmed that printing was possible at a high printing density. This is considered to be because the pigment coated (encapsulated) with the pigment dispersant hardly permeates the paper and remains on the paper surface.
- the inkjet ink head was dried at 45 ° C. for 24 hours to make it impossible to discharge, and then the head cleaning operation was performed once.
- the ink could be ejected without any problem. That is, it is clear that once dried, the dried product can be dissolved and dispersed again, that is, the ink has good redissolvability and redispersibility. This is considered to be because the polymer chain A containing a carboxyl group forms ions and is easily dissolved in a liquid medium such as water even when dried.
- the aqueous pigment dispersion of the present invention is used, it is excellent in dischargeability and long-term stability, can be easily redispersed and re-dissolved even when dried with a head, and has a high level of saturation and gloss. Thus, it is possible to provide an aqueous pigment ink for ink jet recording capable of recording a printed matter having improved color development and light fastness.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Materials Engineering (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Dispersion Chemistry (AREA)
- Inks, Pencil-Leads, Or Crayons (AREA)
- Ink Jet (AREA)
- Ink Jet Recording Methods And Recording Media Thereof (AREA)
- Pigments, Carbon Blacks, Or Wood Stains (AREA)
Abstract
Description
[1]顔料5~35質量%、顔料分散剤0.5~25質量%、水性有機溶剤5~30質量%、及び水20~80質量%を含有するインクジェット用の水性顔料分散液であって、前記顔料分散剤が、ポリマー鎖Aがポリマー鎖Bにグラフトしたグラフトコポリマーと、前記ポリマー鎖Aの片末端と前記ポリマー鎖Bの片末端が結合したブロックコポリマーの少なくともいずれかであり、前記ポリマー鎖Aが、第一のシクロアルキル基含有(メタ)アクリレートに由来する構成単位20~60質量%、(メタ)アクリル酸に由来する構成単位10~35質量%、及びその他の(メタ)アクリレートに由来する構成単位5~70質量%を含むとともに、その数平均分子量が1,000~10,000であり、前記ポリマー鎖Bが、第二のシクロアルキル基含有(メタ)アクリレートに由来する構成単位と、芳香環を有するビニル系モノマー又は(メタ)アクリレートに由来する構成単位の少なくともいずれかを含み、前記ポリマー鎖Aと前記ポリマー鎖Bの質量比が、A:B=30~70:70~30であり、前記グラフトコポリマー及び前記ブロックコポリマーの数平均分子量が2,000~20,000である水性顔料分散液。
[2]前記顔料が、カラーインデックスナンバー(C.I.)ピグメントブルー-15:3、15:4、C.I.ピグメントレッド-122、269、C.I.ピグメントバイオレット-19、C.I.ピグメントイエロ-74、155、180、C.I.ピグメントグリーン-36、58、C.I.ピグメントオレンジ-43、及びC.I.ピグメントブラック-7からなる群より選択される少なくとも一種であるとともに、その数平均一次粒子径が150nm未満である前記[1]に記載の水性顔料分散液。
[3]前記水性有機溶剤が、水に対する25℃における溶解度が20質量%以上の、ポリ(n=1以上)アルキレン(C2~3)グリコールモノアルキルエーテル、ポリ(n=1以上)アルキレン(C2~3)グリコールモノアリルエーテル、ポリ(n=1以上)アルキレン(C2~3)グリコールジアルキルエーテル、アルキレンジオール、アルキレンモノオールモノアルキルエーテル、アルキレンポリオール、及びアミド系溶剤からなる群より選択される少なくとも一種である前記[1]又は[2]に記載の水性顔料分散液。
[4]前記顔料が、前記顔料分散剤で被覆処理されている前記[1]~[3]のいずれかに記載の水性顔料分散液。
[5]前記第一のシクロアルキル基含有(メタ)アクリレートが、シクロヘキシル(メタ)アクリレートと3,3,5-トリメチルシクロヘキシル(メタ)アクリレートの少なくともいずれかである前記[1]~[4]のいずれかに記載の水性顔料分散液。
[6]前記[1]~[5]のいずれかに記載の水性顔料分散液を含有し、前記顔料の含有割合が4~10質量%であるインクジェット用水性顔料インク。
[7]顔料分散剤で被覆処理された分散剤被覆顔料の調製方法であって、(i)顔料、顔料分散剤、水性有機溶剤、水、及びアルカリ剤を混合して得た顔料分散液に酸を添加して、前記顔料分散剤を析出させる工程、又は(ii)前記顔料と前記顔料分散剤を混練して得た混練物を前記顔料分散剤の貧溶媒に添加して、前記顔料分散剤を析出させる工程を含み、前記顔料分散剤が、ポリマー鎖Aがポリマー鎖Bにグラフトしたグラフトコポリマーと、前記ポリマー鎖Aの片末端と前記ポリマー鎖Bの片末端が結合したブロックコポリマーの少なくともいずれかであり、前記ポリマー鎖Aが、第一のシクロアルキル基含有(メタ)アクリレートに由来する構成単位20~60質量%、(メタ)アクリル酸に由来する構成単位10~35質量%、及びその他の(メタ)アクリレートに由来する構成単位5~70質量%を含むとともに、その数平均分子量が1,000~10,000であり、前記ポリマー鎖Bが、第二のシクロアルキル基含有(メタ)アクリレートに由来する構成単位と、芳香環を有するビニル系モノマー又は(メタ)アクリレートに由来する構成単位の少なくともいずれかを含み、前記ポリマー鎖Aと前記ポリマー鎖Bの質量比が、A:B=30~70:70~30であり、前記グラフトコポリマー及び前記ブロックコポリマーの数平均分子量が2,000~20,000である分散剤被覆顔料の調製方法。
顔料分散剤は、ポリマー鎖Aがポリマー鎖Bにグラフトしたグラフトコポリマーと、ポリマー鎖Aの片末端とポリマー鎖Bの片末端が結合したブロックコポリマーの少なくともいずれかである。グラフトコポリマーは、主鎖であるポリマー鎖Bに対して、1本以上のポリマー鎖Aが結合(分岐)している。なお、一のポリマー鎖Bに対するポリマー鎖Aの結合本数は限定されない。ポリマー鎖Aは、第一のシクロアルキル基含有(メタ)アクリレートに由来する構成単位20~60質量%、(メタ)アクリル酸に由来する構成単位10~35質量%、及びその他の(メタ)アクリレートに由来する構成単位5~70質量%を含む。(メタ)アクリル酸に由来する構成単位に含まれるカルボキシル基は、アルカリで中和されることによってイオン化する。このため、(メタ)アクリル酸に由来する構成単位を含むポリマー鎖Aは、水に溶解する性質を有するポリマー鎖である。
ポリマー鎖Aにはシクロアルキル基が含まれる。シクロアルキル基を有するポリマー鎖Aを含む顔料分散剤を用いることにより、高発色、高彩度、及び高グロスな印字物を記録可能な水性顔料インクを調製可能となる。第一のシクロアルキル基含有(メタ)アクリレートの具体例としては、シクロヘキシル(メタ)アクリレート、メチルシクロヘキシル(メタ)アクリレート、3,3,5-トリメチルシクロヘキシル(メタ)アクリレート、t-ブチルシクロヘキシル(メタ)アクリレート、シクロヘキシロキシエチル(メタ)アクリレート、トリシクロデシル(メタ)アクリレート、イソボロニル(メタ)アクリレートなどを挙げることができる。なかでも、シクロヘキシル(メタ)アクリレート、3,3,5-トリメチルシクロヘキシル(メタ)アクリレートが好ましい。また、シクロアルキル基の炭素数は6~9個であることが好ましい。炭素数6~9個のシクロアルキル基であれば、多く導入した場合であっても水溶解性があまり阻害されないとともに、入手も容易だからである。
ポリマー鎖Bは水に不溶のポリマー鎖であり、顔料に対する吸着性を有する。このため、ポリマー鎖Bは顔料に吸着し、表面上に堆積して顔料を被覆(カプセル化)する。第二のシクロアルキル基含有(メタ)アクリレートの具体例としては、前述の第一のシクロアルキル基含有(メタ)アクリレートの具体例として列挙したものと同様のものを挙げることができる。ポリマー鎖Bに含まれる第二のシクロアルキル(メタ)アクリレートに由来する構成単位の割合は、30~70質量%であることが好ましく、40~60質量%であることがさらに好ましい。
グラフトコポリマーは、従来公知の方法に準じて合成することができる。合成方法の具体例としては、(i)その片末端にラジカル重合する不飽和結合が導入されたマクロモノマー(ポリマー鎖A)と、ポリマー鎖Bの構成成分であるモノマーとを重合する方法(マクロモノマー法);(ii)重合開始基を結合させたポリマー鎖Bの存在下、ポリマー鎖Aの構成成分であるモノマーを重合する方法(側鎖重合法);(iii)その片末端に反応性基「X」が導入されたポリマー鎖Aと、反応性基「X」と反応しうる官能基「Y」を有するモノマーを重合して得られる、その側鎖に官能基「Y」を有するポリマー鎖Bとを調製し、これらポリマー鎖Aとポリマー鎖Bとを反応させる方法(高分子反応法)などがある。いずれの合成方法であってもグラフトコポリマーを合成することができるが、なかでもマクロモノマー法が好ましい。
ブロックコポリマーは、従来公知のラジカル重合法では得ることが困難である。その片末端に官能基「X」を導入したポリマー鎖Aと、その片末端に官能基「X」と反応しうる官能基「Y」を導入したポリマー鎖Bとを用意し、これらのポリマー鎖の官能基「X」と「Y」とを反応させることでブロックコポリマーを得ることができる。しかしながら、反応率が低く、ポリマー鎖Aやポリマー鎖Bが多く残存してしまう場合がある。ブロックコポリマーの好ましい合成方法としては、リビングカチオン重合法、リビングアニオン重合法、リビングラジカル重合法などがある。ただし、リビングカチオン重合法やリビングアニオン重合法では、(メタ)アクリレートが重合しないなどの不都合が生ずる場合がある。このため、ブロックコポリマーの合成方法としてはリビングラジカル重合法が特に好ましい。
顔料としては、有機顔料や無機顔料などを一種又は二種以上用いることができる。顔料の具体例としては、カーボンブラック、キナクリドン系顔料、フタロシアニン系顔料、ベンズイミダゾロン系顔料、イソインドリノン系顔料、アゾ系顔料などを挙げることができる。より具体的には、発色性、分散性、及び耐候性などの観点から、カラーインデックスナンバー(C.I.)で示すと、C.I.ピグメントブルー-15:3、15:4、C.I.ピグメントレッド-122、269、C.I.ピグメントバイオレット-19、C.I.ピグメントイエロ-74、155、180、C.I.ピグメントグリーン-36、58、C.I.ピグメントオレンジ-43、及びC.I.ピグメントブラック-7からなる群より選択される少なくとも一種であることが好ましい。
水性有機溶剤は、水に対する混和性を有する有機溶剤である。水性有機溶剤としては、水に対する溶解度が25℃において20質量%以上の、ポリ(n=1以上)アルキレン(C2~3)グリコールモノアルキルエーテル、ポリ(n=1以上)アルキレン(C2~3)グリコールモノアリルエーテル、ポリ(n=1以上)アルキレン(C2~3)グリコールジアルキルエーテル、アルキレンジオール、アルキレンモノオールモノアルキルエーテル、アルキレンポリオール、及びアミド系溶剤からなる群より選択される少なくとも一種を用いることが好ましい。
本発明の水性顔料分散液に含有される顔料の割合は5~35質量%であり、5~30質量%であることが好ましく、7~20質量%であることがさらに好ましい。また、顔料分散剤の含有割合は0.5~25質量%である。さらに、水性有機溶剤の含有割合は5~30質量%であり、5~20質量%であることが好ましい。また、水の含有割合は20~80質量%である。各成分の配合割合は、要求される品質やコストなどを考慮して適宜調整される。例えば、顔料の含有割合が7~20質量%であると、彩度、発色性、安定性が顕著に向上するために好ましい。なお、顔料の含有割合が少なすぎると、印字濃度が確保できなくなる。一方、顔料の含有割合が多すぎるとインクが増粘してしまい、インクジェットヘッドからのインクの吐出安定性が低下する傾向にある。また、顔料の含有割合が5~30質量%である場合には、顔料分散剤の含有割合が0.5~20質量%、水性有機溶剤の含有割合が10~30質量%、及び水の含有割合が50~70質量%であることが好ましい。
本発明の水性顔料分散液は、例えば、顔料、顔料分散剤、水性有機溶剤、及び水を従来公知の方法で混合して分散させることで調製することができる。各成分の混合及び分散に際しては、分散機を用いればよい。分散機の具体例としては、ニーダー、二本ロール、三本ロール、商品名「ミラクルKCK」(浅田鉄鋼社製)などの混練機;超音波分散機;高圧ホモジナイザー(商品名「マイクロフルイダイザー」(みずほ工業社製)、商品名「ナノマイザー」(吉田機械興業社製)、商品名「スターバースト」(スギノマシン社製)、商品名「G-スマッシャー」(リックス社))などを挙げることができる。また、ガラスやジルコンなどのビーズメディアを用いるボールミル、サンドミル、横型メディアミル分散機、コロイドミルなどを使用することもできる。具体的な分散方法は特に限定されない。
本発明においては、顔料分散剤で被覆処理された分散剤被覆顔料、すなわち、その表面に顔料分散剤を堆積させて顔料を被覆(カプセル化)した分散剤被覆顔料を用いることが好ましい。このような分散剤被覆顔料を用いることで、顔料の分散安定性をより高めることができる。すなわち、顔料分散剤によって顔料を被覆することで、有機溶剤が大量に混入しても顔料分散剤が顔料の表面上から脱離しにくくなり、さらには、ポリマー鎖Aが水に溶解しているので顔料の分散安定性をより高めることができる。
本発明のインクジェット用水性顔料インクは、前述の水性顔料分散液を含有するものであり、顔料の含有割合が4~10質量%である。本発明のインクジェット用水性顔料インクには、水性顔料分散液以外の成分として、例えばビヒクル成分が含有される。ビヒクル成分の具体例としては、界面活性剤、有機溶剤、及び保湿剤などを挙げることができる。なお、インクジェットで印字するドット径を最適な幅に広げるという観点から、インクジェット用水性顔料インクの表面張力は20~40mN/mであることが好ましい。インクジェット用水性顔料インクの表面張力は、界面活性剤を添加することで調製することができる。界面活性剤としては、アニオン性界面活性剤、非イオン性界面活性剤、カチオン性界面活性剤、及び両性界面活性剤を用いることができる。
(合成例1)
撹拌機、逆流コンデンサー、温度計、及び窒素導入管を取り付けた反応容器に、ブチルトリグリコール(以下、「BTG」と記す)500部、メチルメタクリレート(以下、「MMA」と記す)72部、シクロヘキシルメタクリレート(以下、「CHMA」と記す)80部、メタクリル酸(以下、「MAA」と記す)48部、エチル-2-(α-ブロモメチル)アクリレート(以下、「EBMA」と記す)5部、及び2,2’-アゾビス(イソ酪酸)ジメチル(以下、「V-601」と記す)2部を仕込んだ。窒素バブリングしながら75℃で3時間重合後、V-601を1部添加した。さらに4.5時間重合させて、ポリマー(マクロモノマーMM-1)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、GPCの示差屈折計(以下、「RI」と記す)を用いて測定したマクロモノマーMM-1の数平均分子量(以下、「Mn」と記す)は6,800であり、重量平均分子量(以下、「Mw」と記す)は10,900であり、分散度(Mw/Mn)(以下、「PDI」と記す)は1.60であった。また、紫外線吸収検出器(波長254nm)(以下、「UV検出器」と記す)では、ピークがほとんど観測されなかった。
合成例1で用いたものと同様の反応容器に、トリプロピレングリコールモノメチルエーテル(以下、「MFTG」と記す)250部、MMA30部、CHMA40部、MAA30部、EBMA2.5部、及びV-601を1部仕込んだ。窒素バブリングしながら75℃で3時間重合後、V-601を0.5部添加した。さらに4.5時間重合させて、ポリマー(マクロモノマーMM-2)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、マクロモノマーMM-2のMnは6,400であり、Mwは10,200であり、PDIは1.59であった。
合成例1で用いたものと同様の反応容器に、テトラエチレングリコールジメチルエーテル(以下、「TEDM」と記す)250部、MMA36部、3,3,5-トリメチルシクロヘキシルメタクリレート(以下、「TMCHMA」と記す)40部、MAA24部、EBMA2.5部、及びV-601を1部仕込んだ。窒素バブリングしながら75℃で3時間重合後、V-601を0.5部添加した。さらに4.5時間重合させて、ポリマー(マクロモノマーMM-3)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、マクロモノマーMM-3のMnは6,500であり、Mwは10,400であり、PDIは1.60であった。
合成例1で用いたものと同様の反応容器に、MFTG250部、MMA36部、CHMA40部、MAA24部、EBMA3.5部、及びV-601を1部仕込んだ。窒素バブリングしながら75℃で3時間重合後、V-601を0.5部添加した。さらに4.5時間重合させて、ポリマー(マクロモノマーMM-4)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、マクロモノマーMM-4のMnは5,400であり、Mwは8,500であり、PDIは1.57であった。
合成例1で用いたものと同様の反応容器に、MFTG250部、MMA20部、エチルメタクリレート(以下、「EMA」と記す)15部、ヒドロキシエチルメタクリレート(以下HEMA)5部、CHMA40部、MAA20部、EBMA3.5部、及びV-601を1部仕込んだ。窒素バブリングしながら75℃で3時間重合後、V-601を0.5部添加した。さらに4.5時間重合させて、ポリマー(マクロモノマーMM-5)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、マクロモノマーMM-5のMnは5,700であり、Mwは9,700であり、PDIは1.70であった。
合成例1で用いたものと同様の反応容器に、MFTG250部、MMA36部、ブチルメタクリレート(以下、「BMA」と記す)40部、MAA24部、EBMA2.5部、及びV-601を1部仕込んだ。窒素バブリングしながら75℃で3時間重合後、V-601を0.5部添加した。さらに4.5時間重合させて、ポリマー(マクロモノマーMM-R1)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、マクロモノマーMM-R1のMnは6,300であり、Mwは10,000であり、PDIは1.59であった。なお、このマクロモノマーMM-R1は、シクロアルキル基を有しないマクロモノマーである。
合成例1で用いたものと同様の反応容器に、MFTG250部、MMA36部、エチルメタクリレート(以下、「EMA」と記す)10部、2-エチルヘキシルメタクリレート(以下、「2EHMA」と記す)30部、MAA24部、EBMA2.5部、及びV-601を1部仕込んだ。窒素バブリングしながら75℃で3時間重合後、V-601を0.5部添加した。さらに4.5時間重合させて、ポリマー(マクロモノマーMM-R2)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、このマクロモノマーMM-R2のMnは7,400であり、Mwは11,000であり、PDIは1.49であった。このマクロモノマーMM-R2は、シクロアルキル基を有しないマクロモノマーである。
(合成例6)
合成例1で用いたものと同様の反応容器Aに、BTG100部、及びマクロモノマーMM-1の溶液600部を仕込んで80℃に加熱した。また、別の反応容器にスチレン(以下、「St」と記す)200部、ブチルアクリレート(以下、「BA」と記す)100部、及びt-ブチルパーオキシ-2-エチルヘキサノアート(以下、「PBO」と記す)5部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。PBOを2.5部添加して85℃に加熱し、さらに4時間重合させた。水酸化カリウム(KOH)32.3部、及び水467.7部を添加して中和し、ポリマー(コポリマーCP-1)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、コポリマーCP-1のMnは15,900であり、Mwは38,500であり、PDIは2.42であった。なお、マクロモノマー由来の分子量のピークは見られなかった。また、UV検出器を使用して分子量を測定したところ、Mnは15,600、Mw39,100、PDIは2.51であった。これは、ポリマー鎖Bを構成するモノマー成分が芳香環を有するものであり、大きな吸収が観測されたためであると考えられる。なお、マクロモノマーMM-1がポリマー鎖Bを構成するモノマー成分と重合して分子量が増大し、グラフトコポリマーが得られたと考えられる。以下の合成例においても、同様の測定を行って得られたコポリマーCP-1がグラフトコポリマーとなっていることを確認した。また、固形分濃度を測定した結果に基づき、得られたポリマー溶液にイオン交換水を加え、固形分濃度を30%に調整した。以下の合成例においても、同様にして固形分濃度を30%に調整した。
合成例1で用いたものと同様の反応容器Aに、MFTG50部、及びマクロモノマーMM-2の溶液300部を仕込んで80℃に加熱した。また、別の反応容器にSt100部、BA50部、及びPBO2.5部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。PBOを1.25部添加して85℃に加熱し、さらに4時間重合させた。KOH16.2部、及び水233.8部を添加して中和し、ポリマー(コポリマーCP-2)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を算出したところ100%であった。また、コポリマーCP-1のMnは14,800であり、Mwは34,200であり、PDIは2.31であった。
合成例1で用いたものと同様の反応容器Aに、マクロモノマーMM-2の溶液300部を仕込んで80℃に加熱した。また、別の反応容器にSt67部、BA33部、及びPBO2部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。PBOを1部添加して85℃に加熱し、さらに4時間重合させた。KOH16.1部、及び水183.9部を添加して中和し、ポリマー(コポリマーCP-3)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、コポリマーCP-3のMnは11,400であり、Mwは27,500であり、PDIは2.41であった。
合成例1で用いたものと同様の反応容器Aに、マクロモノマーMM-2の溶液300部を仕込んで80℃に加熱した。また、別の反応容器にSt67部、HEMA33部、及びPBO2部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。PBOを1部添加して85℃に加熱し、さらに4時間重合させた。KOH16.1部、及び水183.9部を添加して中和し、ポリマー(コポリマーCP-4)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、コポリマーCP-4のMnは10,600であり、Mwは22,800であり、PDIは2.15であった。
合成例1で用いたものと同様の反応容器Aに、TEDM50部、及びマクロモノマーMM-3の溶液300部を仕込んで80℃に加熱した。また、別の反応容器にSt100部、BA50部、及びPBO2.5部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。PBOを1.25部添加して85℃に加熱し、さらに4時間重合させた。KOH16.2部、及び水233.8部を添加して中和し、ポリマー(コポリマーCP-5)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、コポリマーCP-5のMnは14,700であり、Mwは28,000であり、PDIは1.90であった。
合成例1で用いたものと同様の反応容器Aに、MFTG50部、マクロモノマーMM-4の溶液300部を仕込んで80℃に加熱した。また、別の反応容器にSt100部、BA50部、及びPBO2.5部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。PBOを1.25部添加して85℃に加熱し、さらに4時間重合させた。KOH16.2部、及び水233.8部を添加して中和し、ポリマー(コポリマーCP-6)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、コポリマーCP-6のMnは13,100であり、Mwは29,000であり、PDIは2.21であった。
合成例1で用いたものと同様の反応容器Aに、MFTG50部、マクロモノマーMM-5の溶液300部を仕込んで80℃に加熱した。また、別の反応容器にSt67部、HEMA33部、及びPBO2部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。PBOを1部添加して85℃に加熱し、さらに4時間重合させた。KOH16.1部、及び水183.9部を添加して中和し、ポリマー(コポリマーCP-7)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、コポリマーCP-7のMnは9,800であり、Mwは22,200であり、PDIは2.27であった。
合成例1で用いたものと同様の反応容器Aに、MFTG50部、マクロモノマーMM-R1の溶液300部を仕込んで80℃に加熱した。また、別の反応容器にSt100部、BA50部、及びPBO2.5部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。PBOを1.25部添加して85℃に加熱し、さらに4時間重合させた。KOH16.2部、及び水233.8部を添加して中和し、ポリマー(コポリマーCP-R1)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、コポリマーCP-R1のMnは14,000であり、Mwは31,700であり、PDIは2.26であった。このコポリマーCP-R1は、グラフトしているポリマーにシクロアルキル基を有しないグラフトコポリマーである。
合成例1で用いたものと同様の反応容器Aに、MFTG50部、マクロモノマーMM-R2の溶液300部を仕込んで80℃に加熱した。また、別の反応容器にSt100部、HEMA50部、及びPBO2.5部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。PBOを1.25部添加して85℃に加熱し、さらに4時間重合させた。KOH16.2部、及び水233.8部を添加して中和し、ポリマー(コポリマーCP-R2)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、コポリマーCP-R2のMnは15,600であり、Mwは37,000であり、PDIは2.37であった。このコポリマーCP-R2は、グラフトしているポリマー(ポリマー鎖A)にシクロアルキル基を有しないグラフトコポリマーである。
合成例1で用いたものと同様の反応容器Aに、MFTG50部、マクロモノマーMM-2の溶液300部を仕込んで80℃に加熱した。また、別の反応容器にMMA100部、BA50部、及びPBO2.5部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。PBOを1.25部添加して85℃に加熱し、さらに4時間重合させた。KOH16.2部、及び水233.8部を添加して中和し、ポリマー(コポリマーCP-R3)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ85%であった。また、コポリマーCP-R3のMnは10,200、Mwは23,000であり、PDIは2.25であった。このコポリマーCP-R3は、主鎖(ポリマー鎖B)に芳香環やシクロアルキル基を有しないグラフトコポリマーである。
合成例1で用いたものと同様の反応容器Aに、MFTG250部を仕込んで80℃に加熱した。また、別の反応容器にMMA36部、CHMA40部、MAA24部、St100部、BA50部、及びアゾビスイソブチロニトリル(以下、「AIBN」と記す)7.5部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。AIBNを1.25部添加して85℃に加熱し、さらに4時間重合させた。KOH16.2部、及び水233.8部を添加して中和し、ポリマー(コポリマーCP-R4)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、コポリマーCP-R4のMnは14,700であり、Mwは30,600であり、PDIは2.08であった。このコポリマーCP-R4はランダムコポリマーである。
(合成例13)
合成例1で用いたものと同様の反応容器に、MFTG173部、ヨウ素1.0部、2,2’-アゾビス(4-メトキシ-2,4-ジメチルバレロニトリル)(以下、「V-70」と記す)3.7部、CHMA42部、ベンジルメタクリレート(以下、「BzMA」と記す)17.6部、及びジフェニルメタン(以下、「DPM」と記す)0.17部を仕込んだ。窒素バブリングしながら45℃で5.5時間重合させてポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ86%であった。このポリマー溶液に含有されるポリマーのMnは5,000であり、PDIは1.19であった。次に、ポリマー溶液を40℃に冷却し、CHMA16.8部、MMA20部、MAA12.9部、及びV-70を1.5部加えて3.5時間重合させた。KOH8.4部、及び水49.2部を添加して中和し、ポリマー(ブロックコポリマーBP-1)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、ブロックポリマーBP-1のMnは10,300であり、PDIは1.30であった。なお、固形分濃度を測定した結果に基づき、得られたポリマー溶液にイオン交換水を加え、固形分濃度を30%に調整した。以下の合成例においても、同様にして固形分濃度を30%に調整した。
合成例1で用いたものと同様の反応容器に、MFTG174部、ヨウ素1.0部、V-70を3.7部、CHMA29.4部、BzMA30.8部、及びDPM0.17部を仕込んだ。窒素バブリングしながら45℃で5.5時間重合させてポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ82%であった。また、このポリマー溶液に含有されるポリマーのMnは5,700であり、PDIは1.20であった。次に、ポリマー溶液を40℃に冷却し、TMCHMA16.8部、MMA20部、MAA12.9部、及びV-70を1.5部加えて3.5時間重合させた。KOH8.4部、及び水49.2部を添加して中和し、ポリマー(ブロックコポリマーBP-2)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、ブロックコポリマーBP-2のMnは10,300であり、PDIは1.31であった。
合成例1で用いたものと同様の反応容器に、MFTG168部、ヨウ素1.0部、V-70を3.7部、CHMA42部、BzMA17.6部、及びDPM0.17部を仕込んだ。窒素バブリングしながら45℃で5.5時間重合させてポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ80%であった。また、このポリマー溶液に含有されるポリマーのMnは5,000であり、PDIは1.17であった。次に、ポリマー溶液を40℃に冷却し、CHMA8.4部、MMA25部、MAA12.9部、及びV-70を1.4部加え、3.5時間重合させた。KOH8.4部、及び水47.6部を添加して中和し、ポリマー(ブロックコポリマーBP-3)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、ブロックコポリマーBP-3のMnは9,100であり、PDIは1.31であった。
合成例1で用いたものと同様の反応容器に、MFTG168部、ヨウ素1.0部、V-70を3.7部、CHMA42部、ヒドロキシエチルメタクリレート(以下、「HEMA」と記す)13部、及びDPM0.17部を仕込んだ。窒素バブリングしながら45℃で5.5時間重合させてポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ81%であった。また、このポリマー溶液に含有されるポリマーのMnは5,100であり、PDIは1.22であった。次に、ポリマー溶液を40℃に冷却し、CHMA16.8部、MMA20部、MAA12.9部、及びV-70を1.5部加え、3.5時間重合させた。KOH8.4部、及び水47.6部を添加して中和し、ポリマー(ブロックコポリマーBP-4)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、ブロックコポリマーBP-4のMnは9,600であり、PDIは1.33であった。
合成例1で用いたものと同様の反応容器に、BTG173部、ヨウ素1.0部、V-70を3.7部、CHMA42部、BzMA17.6部、及びDPM0.17部を仕込んだ。窒素バブリングしながら45℃で5.5時間重合させてポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ89%であった。また、このポリマー溶液に含有されるポリマーのMnは6,000であり、PDIは1.18であった。次に、ポリマー溶液を40℃に冷却し、CHMA16.8部、MMA20部、MAA12.9部、及びV-70を1.5部加え、3.5時間重合させた。NaOH6.0部、及び水51.6部を添加して中和し、ポリマー(ブロックコポリマーBP-5)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、ブロックコポリマーBP-5のMnは11,100であり、PDIは1.29であった。
合成例1で用いたものと同様の反応容器に、TEDM172部、ヨウ素1.0部、V-70を3.7部、CHMA58.8部、及びDPM0.17部を仕込んだ。窒素バブリングしながら45℃で5.5時間重合させてポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ84%であった。また、このポリマー溶液に含有されるポリマーのMnは5,200であり、PDIは1.19であった。次に、ポリマー溶液を40℃に冷却し、CHMA16.8部、MMA20部、MAA12.9部、及びV-70を1.5部加え、3.5時間重合させた。28%アンモニア水9.1部、及び水48.2部を添加して中和し、ポリマー(ブロックコポリマーBP-6)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、ブロックコポリマーBP-6のMnは10,000であり、PDIは1.31であった。
合成例1で用いたものと同様の反応容器に、3-メトキシ-3-メチル-1-ブタノール(以下、「MMB」と記す)169部、ヨウ素1.0部、V-70を3.7部、CHMA42部、BMA14.2部、及びDPM0.17部を仕込んだ。窒素バブリングしながら45℃で5.5時間重合させてポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ88%であった。また、このポリマー溶液に含有されるポリマーのMnは4,800であり、PDIは1.16であった。次に、ポリマー溶液を40℃に冷却し、CHMA16.8部、MMA20部、MAA12.9部、及びV-70を1.5部加え、3.5時間重合させた。KOH8.4部、及び水47.6部を添加して中和し、ポリマー(ブロックコポリマーBP-7)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、ブロックコポリマーBP-7のMnは9,500であり、PDIは1.29であった。
合成例1で用いたものと同様の反応容器に、MFTG128部、ヨウ素1.0部、V-703.7部、BzMA52.2部、HEMA9.8部、及びDPM0.17部を仕込んだ。窒素バブリングしながら45℃で5.5時間重合させてポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ80%であった。また、このポリマー溶液に含有されるポリマーのMnは4,900であり、PDIは1.26であった。次に、ポリマー溶液を40℃に冷却し、MMA20.8部、BMA40.8部、MAA15.0部、及びV-70を2.3部加え、3.5時間重合させた。KOH9.8部、及び水32.8部を添加して中和し、ポリマー(ブロックコポリマーBP-R1)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を算出したところ100%であった。また、ブロックコポリマーBP-R1のMnは9,200であり、PDIは1.57であった。
合成例1で用いたものと同様の反応容器AにMFTG375部を仕込んで80℃に加熱した。また、別の容器にCHMA110部、BzMA70部、MMA40部、MAA30部、及びアゾビスイソブチロニトリル(以下、「AIBN」と記す)12部を仕込んでよく撹拌し、モノマー液を調製した。このモノマー液を反応容器Aに1/2添加した後、残り1/2を1時間かけてゆっくりと滴下した。滴下終了後、3時間重合させた。AIBNを1.5部添加して85℃に加熱し、さらに4時間重合させた。KOH19.6部、及び水105.4部を添加して中和し、ポリマー(ランダムコポリマーRP-R1)を含有するポリマー溶液を得た。得られたポリマー溶液をサンプリングして固形分濃度を測定し、不揮発分から重合転化率を換算したところ100%であった。また、ランダムコポリマーRP-R1のMnは12,100であり、PDIは2.28であった。
(実施例1)
合成例6で得たコポリマーCP-1を含有するポリマー溶液233.3部、ジエチレングリコールモノブチルエーテル70部、及び水311.7部を混合して若干濁りのある半透明の溶液を得た。この溶液にアゾ系黄色顔料PY-74(商品名「セイカファストイエロー2016G」、大日精化工業社製)350部を添加し、ディスパーを使用して30分撹拌してミルベースを調製した。横型媒体分散機(商品名「ダイノミル0.6リットルECM型」、シンマルエンタープライゼス社製、ジルコニア製ビーズ 径0.5mm)を使用し、周速10m/sで分散処理し、ミルベース中に顔料を十分に分散させた。その後、水316部を添加して顔料濃度を18%とした。分散機から取り出したミルベースを遠心分離処理(7500回転、20分間)した後、10μmのメンブレンフィルターでろ過した。水で希釈して、顔料濃度14%のインクジェット用の水性顔料分散液1を得た。
表4に示す顔料分散剤を用いたこと以外は、上述の実施例1と同様にして水性顔料分散液2~20を得た。得られた水性顔料分散液2~20の評価結果を表4に示す。
(実施例15)
実施例1で調製した水性顔料分散液1を用いて、以下に示す処方でインクジェット用の水性顔料インクを調製した。
水性顔料分散液1 40部
水 42.2部
1,2-ヘキサンジオール 5部
グリセリン 10部
商品名「サーフィノール465」(エアープロダクト社製) 1部
表5に示す顔料分散液を用いたこと以外は、上述の実施例15と同様にして水性顔料インク2~16及び19を得た。また、前述の青色水性顔料分散液、赤色水性顔料分散液、及び黒色水性顔料分散液をそれぞれ用いて各色の水性顔料インクを調製した。いずれの水性顔料インクについても、保存安定性が良好であることを確認した。
(実施例29)
合成例13で得たブロックコポリマーBP-1を含有するポリマー溶液164部、BDG80部、及び水356部を混合して均一な溶液を得た。得られた溶液に赤色顔料(C.I.ピグメントレッド122(ジメチルキナクリドン顔料、大日精化工業社製))200部を添加し、ディスパーを使用して解膠し、ミルベースを調製した。得られたミルベース800部に対して、顔料分5%となるよう水3200部を加えた後、撹拌しながら5%酢酸を滴下して顔料分散剤を析出させた。酢酸滴下前(初期)のpHは9.5であり、酢酸滴下後のpHは4.5であった。ろ過及び水で洗浄して、分散剤被覆顔料のペースト(固形分濃度:32.0%)を得た。
赤色顔料に代えて、(i)アゾ系黄色顔料PY-74(商品名「セイカファストイエロー2016G」、大日精化工業社製)、(ii)銅フタロシアニン顔料PB-15:3(商品名「シアニンブルーA220JC」、大日精化工業社製)、(iii)カーボンブラック顔料PB-7(商品名「S170」、デグザ社製)をそれぞれ用いたこと、並びにインクの合処方を表6に示すようにしたこと以外は、前述の実施例29と同様にして各色水性顔料分散液、及びインクジェット用の各色水性顔料インクを得た。
Claims (7)
- 顔料5~35質量%、顔料分散剤0.5~25質量%、水性有機溶剤5~30質量%、及び水20~80質量%を含有するインクジェット用の水性顔料分散液であって、
前記顔料分散剤が、ポリマー鎖Aがポリマー鎖Bにグラフトしたグラフトコポリマーと、前記ポリマー鎖Aの片末端と前記ポリマー鎖Bの片末端が結合したブロックコポリマーの少なくともいずれかであり、
前記ポリマー鎖Aが、第一のシクロアルキル基含有(メタ)アクリレートに由来する構成単位20~60質量%、(メタ)アクリル酸に由来する構成単位10~35質量%、及びその他の(メタ)アクリレートに由来する構成単位5~70質量%を含むとともに、その数平均分子量が1,000~10,000であり、
前記ポリマー鎖Bが、第二のシクロアルキル基含有(メタ)アクリレートに由来する構成単位と、芳香環を有するビニル系モノマー又は(メタ)アクリレートに由来する構成単位の少なくともいずれかを含み、
前記ポリマー鎖Aと前記ポリマー鎖Bの質量比が、A:B=30~70:70~30であり、
前記グラフトコポリマー及び前記ブロックコポリマーの数平均分子量が2,000~20,000である水性顔料分散液。 - 前記顔料が、カラーインデックスナンバー(C.I.)ピグメントブルー-15:3、15:4、C.I.ピグメントレッド-122、269、C.I.ピグメントバイオレット-19、C.I.ピグメントイエロ-74、155、180、C.I.ピグメントグリーン-36、58、C.I.ピグメントオレンジ-43、及びC.I.ピグメントブラック-7からなる群より選択される少なくとも一種であるとともに、その数平均一次粒子径が150nm未満である請求項1に記載の水性顔料分散液。
- 前記水性有機溶剤が、水に対する25℃における溶解度が20質量%以上の、ポリ(n=1以上)アルキレン(C2~3)グリコールモノアルキルエーテル、ポリ(n=1以上)アルキレン(C2~3)グリコールモノアリルエーテル、ポリ(n=1以上)アルキレン(C2~3)グリコールジアルキルエーテル、アルキレンジオール、アルキレンモノオールモノアルキルエーテル、アルキレンポリオール、及びアミド系溶剤からなる群より選択される少なくとも一種である請求項1又は2に記載の水性顔料分散液。
- 前記顔料が、前記顔料分散剤で被覆処理されている請求項1~3いずれか一項に記載の水性顔料分散液。
- 前記第一のシクロアルキル基含有(メタ)アクリレートが、シクロヘキシル(メタ)アクリレートと3,3,5-トリメチルシクロヘキシル(メタ)アクリレートの少なくともいずれかである請求項1~4いずれか一項に記載の水性顔料分散液。
- 請求項1~5のいずれか一項に記載の水性顔料分散液を含有し、
前記顔料の含有割合が4~10質量%であるインクジェット用水性顔料インク。 - 顔料分散剤で被覆処理された分散剤被覆顔料の調製方法であって、
(i)顔料、顔料分散剤、水性有機溶剤、水、及びアルカリ剤を混合して得た顔料分散液に酸を添加して、前記顔料分散剤を析出させる工程、又は
(ii)前記顔料と前記顔料分散剤を混練して得た混練物を前記顔料分散剤の貧溶媒に添加して、前記顔料分散剤を析出させる工程を含み、
前記顔料分散剤が、ポリマー鎖Aがポリマー鎖Bにグラフトしたグラフトコポリマーと、前記ポリマー鎖Aの片末端と前記ポリマー鎖Bの片末端が結合したブロックコポリマーの少なくともいずれかであり、
前記ポリマー鎖Aが、第一のシクロアルキル基含有(メタ)アクリレートに由来する構成単位20~60質量%、(メタ)アクリル酸に由来する構成単位10~35質量%、及びその他の(メタ)アクリレートに由来する構成単位5~70質量%を含むとともに、その数平均分子量が1,000~10,000であり、
前記ポリマー鎖Bが、第二のシクロアルキル基含有(メタ)アクリレートに由来する構成単位と、芳香環を有するビニル系モノマー又は(メタ)アクリレートに由来する構成単位の少なくともいずれかを含み、
前記ポリマー鎖Aと前記ポリマー鎖Bの質量比が、A:B=30~70:70~30であり、
前記グラフトコポリマー及び前記ブロックコポリマーの数平均分子量が2,000~20,000である分散剤被覆顔料の調製方法。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP13748957.1A EP2816088B1 (en) | 2012-02-16 | 2013-02-08 | Aqueous pigment dispersion liquid and aqueous pigment inkjet ink using same |
| US14/374,318 US9260589B2 (en) | 2012-02-16 | 2013-02-08 | Aqueous pigment dispersion liquid and aqueous pigment inkjet ink using same |
| CA2864384A CA2864384C (en) | 2012-02-16 | 2013-02-08 | Aqueous pigment dispersion liquid and aqueous pigment inkjet ink using same |
| KR1020147025830A KR101623678B1 (ko) | 2012-02-16 | 2013-02-08 | 수성 안료 분산액 및 그것을 이용한 잉크젯용 수성 안료 잉크 |
| CN201380009508.1A CN104114654B (zh) | 2012-02-16 | 2013-02-08 | 水性颜料分散液和使用了其的喷墨用水性颜料墨 |
| AU2013219442A AU2013219442C1 (en) | 2012-02-16 | 2013-02-08 | Aqueous pigment dispersion liquid and aqueous pigment inkjet ink using same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012031222A JP5782394B2 (ja) | 2012-02-16 | 2012-02-16 | 水性顔料分散液及びそれを用いたインクジェット用水性顔料インク |
| JP2012-031222 | 2012-02-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013122016A1 true WO2013122016A1 (ja) | 2013-08-22 |
Family
ID=48984128
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/053136 WO2013122016A1 (ja) | 2012-02-16 | 2013-02-08 | 水性顔料分散液及びそれを用いたインクジェット用水性顔料インク |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US9260589B2 (ja) |
| EP (1) | EP2816088B1 (ja) |
| JP (1) | JP5782394B2 (ja) |
| KR (1) | KR101623678B1 (ja) |
| CN (1) | CN104114654B (ja) |
| AU (1) | AU2013219442C1 (ja) |
| CA (1) | CA2864384C (ja) |
| TW (1) | TWI535744B (ja) |
| WO (1) | WO2013122016A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2990447A1 (en) * | 2014-08-28 | 2016-03-02 | Seiko Epson Corporation | Ink composition and ink set |
| JP7122482B1 (ja) | 2022-01-13 | 2022-08-19 | 大日精化工業株式会社 | バインダー成分、エマルジョン、エマルジョンの製造方法、及び水性インク |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI727366B (zh) | 2013-08-09 | 2021-05-11 | 日商半導體能源研究所股份有限公司 | 發光元件、顯示模組、照明模組、發光裝置、顯示裝置、電子裝置、及照明裝置 |
| EP3070132B1 (en) * | 2013-11-14 | 2019-01-09 | DIC Corporation | Aqueous pigment dispersion and aqueous ink for inkjet recording use |
| JP6468087B2 (ja) | 2014-06-23 | 2019-02-13 | セイコーエプソン株式会社 | 液体収容容器 |
| JP6624845B2 (ja) * | 2015-08-17 | 2019-12-25 | キヤノン株式会社 | 樹脂組成物の製造方法、及び樹脂粒子の製造方法 |
| JP6373813B2 (ja) * | 2015-09-18 | 2018-08-15 | 大日精化工業株式会社 | カラーフィルター用着色剤組成物及び顔料分散剤の製造方法 |
| CN108699201B (zh) * | 2016-02-03 | 2020-05-05 | 大塚化学株式会社 | Aba型嵌段共聚物、分散剂和颜料分散组合物 |
| US9828514B2 (en) | 2016-04-07 | 2017-11-28 | Eastman Kodak Company | Preparation of aqueous green dispersions |
| WO2017176471A1 (en) * | 2016-04-07 | 2017-10-12 | Eastman Kodak Company | Aqueous green pigment dispersions and inkjet compositions |
| US9828513B2 (en) | 2016-04-07 | 2017-11-28 | Eastman Kodak Company | Aqueous green pigment dispersions and inkjet compositions |
| JP6704461B2 (ja) * | 2016-09-30 | 2020-06-03 | 富士フイルム株式会社 | 水性顔料分散物、及び、水性インク組成物 |
| JP6977262B2 (ja) | 2016-12-29 | 2021-12-08 | セイコーエプソン株式会社 | インク組成物、インクセット |
| CN110366586A (zh) * | 2017-03-14 | 2019-10-22 | Dic株式会社 | 水性颜料分散体和水性颜料分散体的制造方法 |
| JP2018154790A (ja) * | 2017-03-21 | 2018-10-04 | セイコーエプソン株式会社 | 分散剤及び捺染インク組成物 |
| US11999810B2 (en) * | 2018-07-05 | 2024-06-04 | Basf Se | Process for producing an aqueous polymer dispersion |
| JP7153623B2 (ja) * | 2019-09-18 | 2022-10-14 | 大日精化工業株式会社 | 顔料分散液組成物及びカラーフィルター用着色剤 |
| JPWO2022025166A1 (ja) * | 2020-07-30 | 2022-02-03 | ||
| JP6886062B1 (ja) * | 2020-08-25 | 2021-06-16 | 大日精化工業株式会社 | A−bブロックコポリマー、ポリマーエマルジョン、及び水性インクジェットインク |
| CN112759981A (zh) * | 2021-01-22 | 2021-05-07 | 传美讯电子科技(珠海)有限公司 | 一种高附着力玻璃用水性颜料墨水 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004051971A (ja) | 2002-05-31 | 2004-02-19 | Dainichiseika Color & Chem Mfg Co Ltd | 水性顔料インク、画像記録方法および画像記録装置 |
| JP2008231130A (ja) | 2007-03-16 | 2008-10-02 | Seiko Epson Corp | インクジェット記録用インク |
| JP2009149912A (ja) | 2004-01-19 | 2009-07-09 | Dainichiseika Color & Chem Mfg Co Ltd | 優れた顔料分散性を有するグラフトコポリマー |
| WO2010013651A1 (ja) * | 2008-07-28 | 2010-02-04 | 大日精化工業株式会社 | 水性顔料分散液、および使用 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006063277A (ja) | 2004-08-30 | 2006-03-09 | Fuji Photo Film Co Ltd | インクジェット記録用インク組成物およびインクジェット記録方法 |
| WO2006082158A1 (en) * | 2005-02-04 | 2006-08-10 | Agfa Graphics Nv | Stable pigment dispersions comprising a block copolymer consisting of ionic aromatic monomers |
| JP5221843B2 (ja) * | 2005-06-10 | 2013-06-26 | Dic株式会社 | インクジェットインク用水性顔料分散体およびインクジェット記録用水性インク |
| JP5263793B2 (ja) | 2010-05-14 | 2013-08-14 | 大日精化工業株式会社 | A−bブロックコポリマー、その製造方法及び顔料分散体 |
| JP5717134B2 (ja) | 2011-03-15 | 2015-05-13 | 大日精化工業株式会社 | エマルジョンバインダー及びそれを含有するインクジェット用水性顔料インク、並びにエマルジョンバインダーの製造方法 |
-
2012
- 2012-02-16 JP JP2012031222A patent/JP5782394B2/ja active Active
-
2013
- 2013-02-08 WO PCT/JP2013/053136 patent/WO2013122016A1/ja active Application Filing
- 2013-02-08 AU AU2013219442A patent/AU2013219442C1/en active Active
- 2013-02-08 EP EP13748957.1A patent/EP2816088B1/en active Active
- 2013-02-08 US US14/374,318 patent/US9260589B2/en active Active
- 2013-02-08 CN CN201380009508.1A patent/CN104114654B/zh active Active
- 2013-02-08 CA CA2864384A patent/CA2864384C/en active Active
- 2013-02-08 KR KR1020147025830A patent/KR101623678B1/ko active IP Right Grant
- 2013-02-08 TW TW102105177A patent/TWI535744B/zh active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004051971A (ja) | 2002-05-31 | 2004-02-19 | Dainichiseika Color & Chem Mfg Co Ltd | 水性顔料インク、画像記録方法および画像記録装置 |
| JP2009149912A (ja) | 2004-01-19 | 2009-07-09 | Dainichiseika Color & Chem Mfg Co Ltd | 優れた顔料分散性を有するグラフトコポリマー |
| JP2008231130A (ja) | 2007-03-16 | 2008-10-02 | Seiko Epson Corp | インクジェット記録用インク |
| WO2010013651A1 (ja) * | 2008-07-28 | 2010-02-04 | 大日精化工業株式会社 | 水性顔料分散液、および使用 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2816088A4 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2990447A1 (en) * | 2014-08-28 | 2016-03-02 | Seiko Epson Corporation | Ink composition and ink set |
| JP7122482B1 (ja) | 2022-01-13 | 2022-08-19 | 大日精化工業株式会社 | バインダー成分、エマルジョン、エマルジョンの製造方法、及び水性インク |
| JP2023103032A (ja) * | 2022-01-13 | 2023-07-26 | 大日精化工業株式会社 | バインダー成分、エマルジョン、エマルジョンの製造方法、及び水性インク |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5782394B2 (ja) | 2015-09-24 |
| US20150011687A1 (en) | 2015-01-08 |
| EP2816088A1 (en) | 2014-12-24 |
| AU2013219442A1 (en) | 2014-08-14 |
| EP2816088B1 (en) | 2018-04-11 |
| CA2864384C (en) | 2016-07-19 |
| CA2864384A1 (en) | 2013-08-22 |
| AU2013219442C1 (en) | 2016-05-26 |
| CN104114654B (zh) | 2016-08-24 |
| TW201336878A (zh) | 2013-09-16 |
| JP2013166867A (ja) | 2013-08-29 |
| TWI535744B (zh) | 2016-06-01 |
| KR20140126382A (ko) | 2014-10-30 |
| CN104114654A (zh) | 2014-10-22 |
| EP2816088A4 (en) | 2015-08-19 |
| US9260589B2 (en) | 2016-02-16 |
| KR101623678B1 (ko) | 2016-05-23 |
| AU2013219442B2 (en) | 2015-09-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5782394B2 (ja) | 水性顔料分散液及びそれを用いたインクジェット用水性顔料インク | |
| EP2687561B1 (en) | Emulsion binder, aqueous pigment ink for inkjet containing same, and method for producing emulsion binder | |
| CN108350298B (zh) | 水基颜料分散体 | |
| JP6049027B2 (ja) | 水性顔料分散液の製造方法 | |
| JP5636580B2 (ja) | インクジェットインク用水性顔料分散液およびインクジェット用水性顔料インク | |
| US8710117B2 (en) | Crosslinked core/shell polymer particles | |
| JP6948285B2 (ja) | 顔料分散剤及びその製造方法、水性顔料分散液、並びに水性インクジェットインク | |
| WO2010071177A1 (ja) | インクジェット記録用水系インク | |
| JP7216694B2 (ja) | 顔料分散剤及びその製造方法、インクジェットインク用顔料分散液、及び水性インクジェットインク | |
| JP6482025B2 (ja) | ポリマー水分散体、その製造方法、水性顔料分散液及びインクジェット記録用インク | |
| JP6870261B2 (ja) | インクジェット記録用水性インク及びインクカートリッジ | |
| AU2015264862B2 (en) | Aqueous pigment dispersion liquid and aqueous pigment inkjet ink using same | |
| JP7316470B1 (ja) | 顔料分散液 | |
| JP6697405B2 (ja) | 顔料分散液、水性インクジェットインク、及び顔料分散液の製造方法 | |
| JP2006176623A (ja) | インクジェット記録用水系インク | |
| JP4769446B2 (ja) | インクジェット記録用顔料水分散体の製造法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13748957 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013748957 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14374318 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 2864384 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2013219442 Country of ref document: AU Date of ref document: 20130208 Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20147025830 Country of ref document: KR Kind code of ref document: A |