WO2012173097A1 - 癌細胞接着性向上剤 - Google Patents
癌細胞接着性向上剤 Download PDFInfo
- Publication number
- WO2012173097A1 WO2012173097A1 PCT/JP2012/064932 JP2012064932W WO2012173097A1 WO 2012173097 A1 WO2012173097 A1 WO 2012173097A1 JP 2012064932 W JP2012064932 W JP 2012064932W WO 2012173097 A1 WO2012173097 A1 WO 2012173097A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cancer cell
- cells
- substrate
- cancer
- polymer
- Prior art date
Links
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N1/00—Sampling; Preparing specimens for investigation
- G01N1/28—Preparing specimens for investigation including physical details of (bio-)chemical methods covered elsewhere, e.g. G01N33/50, C12Q
- G01N1/40—Concentrating samples
- G01N1/405—Concentrating samples by adsorption or absorption
Definitions
- the present invention relates to a cancer cell adhesion improver. More specifically, the present invention relates to a cancer cell adhesion improver, a cancer cell concentration filter, and a cancer cell inspection method.
- CTC cancer Tumor Cell
- Non-Patent Documents 1 and 2 As another technique for separating and concentrating CTC, a method using a polycarbonate filter has been proposed (for example, see Non-Patent Documents 1 and 2). In addition, techniques for separating and concentrating CTC using the difference in size have been proposed (see, for example, Non-Patent Documents 3 to 5 and Patent Document 1).
- Cancer cells such as CTC are slightly larger in size than blood cells in blood, such as red blood cells, white blood cells, or platelets. Therefore, theoretically, it is possible to apply these mechanical filtration methods to remove these blood cell components and concentrate the cancer cells.
- the membrane filters of Non-Patent Documents 1 and 2 include randomly distributed pores and the pore diameter is not uniform, the cancer cell recovery rate may be poor. Further, even in the filters of Non-Patent Documents 3 to 5 and Patent Document 1 in which the pore size and thickness are accurately controlled, it may be difficult to concentrate cancer cells with an accuracy that can be accurately analyzed.
- the inventors of the present invention have improved the adhesion of cancer cells to the surface of the material coated with a polymer containing a structural unit represented by the following general formula (1), while the adhesion of blood cell components decreases. It was found for the first time and the present invention was completed.
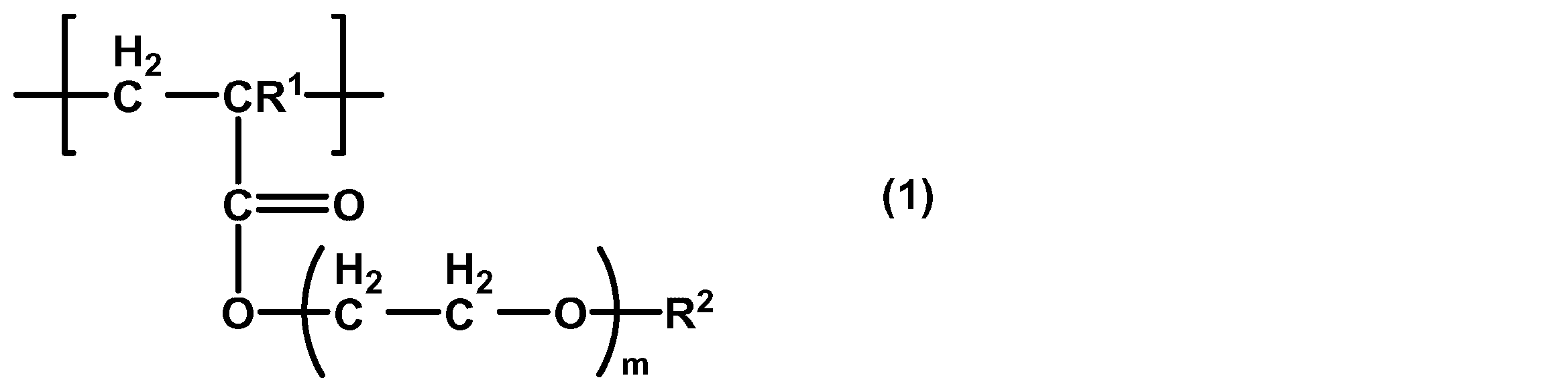
- the present invention provides a cancer cell adhesion improver comprising a polymer containing a structural unit represented by the following general formula (1).
- R 1 is a hydrogen atom or a methyl group
- R 2 is a methyl group or an ethyl group
- m is 1 to 3.
- the cancer cell adhesion improving agent of the present invention By applying the cancer cell adhesion improving agent of the present invention to the surface of a cancer cell concentration filter, it is possible to increase the adhesion of cancer cells to the filter surface and improve the concentration rate of cancer cells.
- the cancer cell adhesion improver of the present invention can simultaneously satisfy the characteristics in a trade-off relationship between the non-adhesiveness of blood cell components and the adhesion of cancer cells.
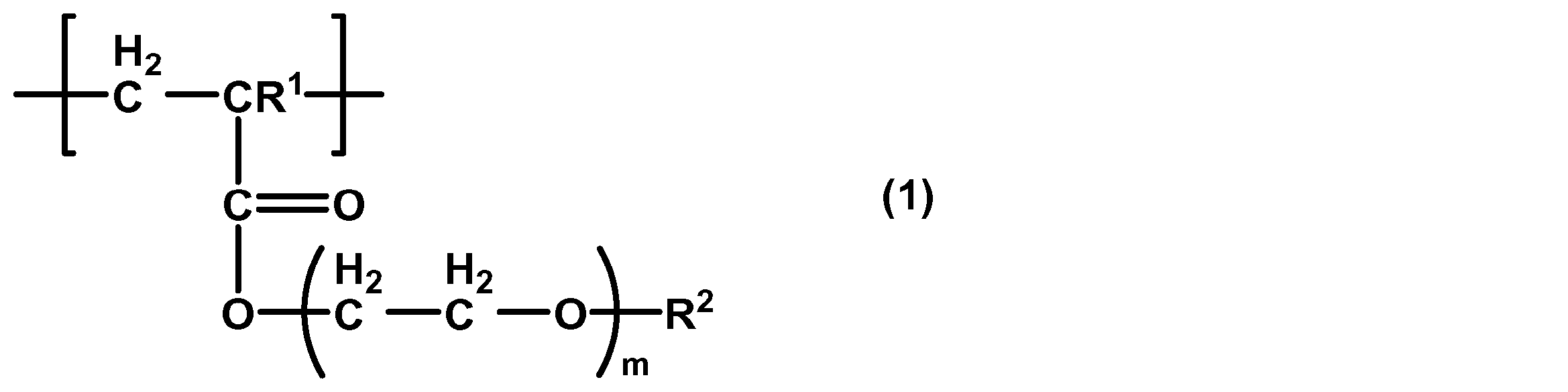
- the present invention also provides a method for improving the adhesion of cancer cells to a substrate, comprising a step of coating the substrate with a polymer containing a structural unit represented by the following general formula (1).
- R 1 is a hydrogen atom or a methyl group
- R 2 is a methyl group or an ethyl group
- m is 1 to 3.
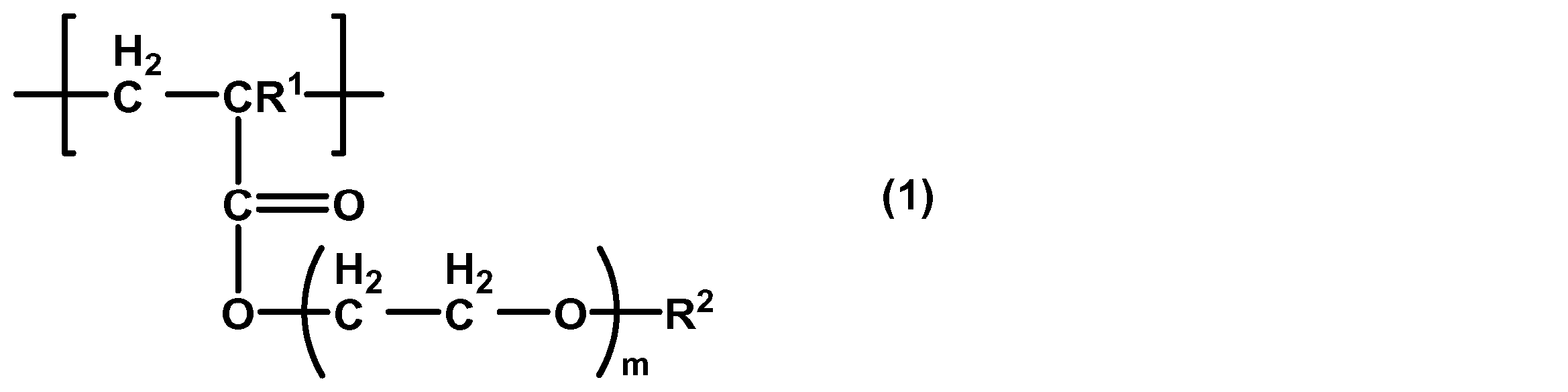
- the present invention also provides use of a polymer containing a structural unit represented by the following general formula (1) for improving cancer cell adhesion.
- R 1 is a hydrogen atom or a methyl group
- R 2 is a methyl group or an ethyl group
- m is 1 to 3.
- the present invention also provides application of a polymer containing a structural unit represented by the following general formula (1) for improving cancer cell adhesion.
- R 1 is a hydrogen atom or a methyl group
- R 2 is a methyl group or an ethyl group
- m is 1 to 3.
- the present invention also provides an application of a polymer containing a structural unit represented by the following general formula (1) for producing a cancer cell adhesion improver.
- R 1 is a hydrogen atom or a methyl group
- R 2 is a methyl group or an ethyl group
- m is 1 to 3.
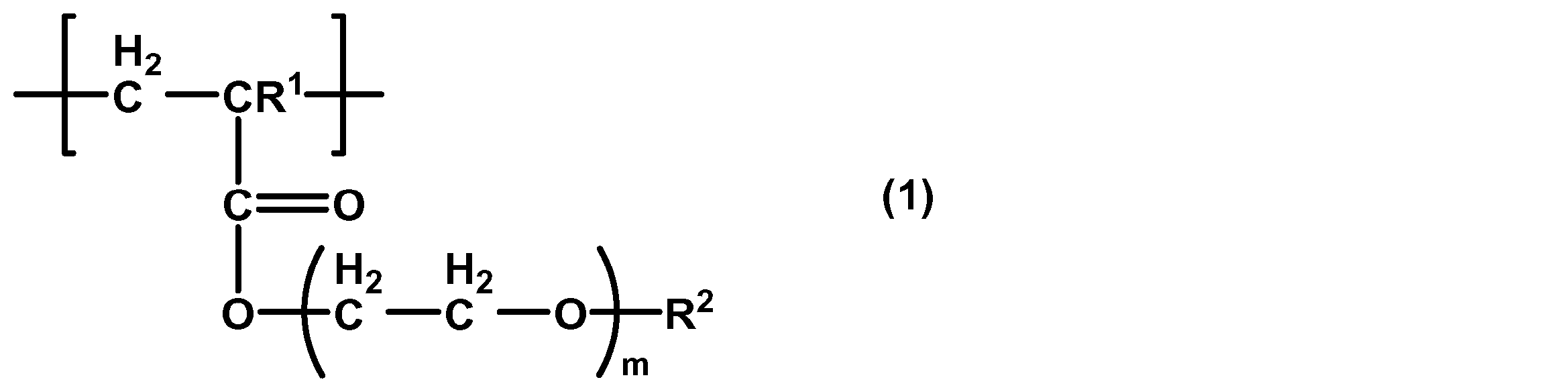
- the present invention also provides a cancer cell concentration filter comprising a substrate having a plurality of through-holes, wherein at least a part of the substrate is coated with a polymer containing a structural unit represented by the following general formula (1). To do.
- R 1 is a hydrogen atom or a methyl group
- R 2 is a methyl or ethyl group
- m is 1-3.
- the polymer may be composed of a structural unit represented by the general formula (1).
- R 1 is preferably a hydrogen atom
- R 2 is a methyl group
- m is preferably 1.
- R 1 is a hydrogen atom
- R 2 is an ethyl group
- m may be 2
- R 1 is a methyl group
- R 2 is a methyl group
- m is 2. Good.
- the polymer preferably has a number average molecular weight of 10,000 to 300,000.
- Such a polymer can further improve the adhesion of cancer cells.
- the present invention also provides a cancer cell concentration filter comprising a substrate having a plurality of through-holes, wherein at least a part of the substrate is coated with the polymer.
- cancer cells in a blood sample can be concentrated at a high concentration rate.
- the above through holes preferably have an average pore diameter of 5 ⁇ m or more and less than 30 ⁇ m, an average aperture ratio of 5% or more and less than 50%, and are preferably formed by electroforming.
- the substrate is preferably made of metal. Further, the metal is preferably selected from the group consisting of copper, nickel, an alloy of copper and nickel, and those whose surfaces are plated with gold.
- cancer cells in a blood sample can be concentrated at a higher concentration rate.
- the present invention also provides a method for detecting the presence of cancer cells, comprising a filtration step of filtering peripheral blood with the above-described cancer cell concentration filter.
- the presence of cancer cells such as CTC in peripheral blood collected from a patient can be easily detected.
- the inspection method may further include a step of analyzing the gene of the cells concentrated in the filtration step, and may further include a step of culturing the cells concentrated in the filtration step.
- the method for detecting the presence of cancer cells of the present invention can more accurately detect the presence of cancer cells such as CTC in peripheral blood.
- a cancer cell adhesion improving agent a cancer cell concentration filter whose surface is coated with a cancer cell adhesion improving agent, and a method for examining cancer cells comprising a step of filtering peripheral blood with the cancer cell concentration filter.
- FIG. 1 is a schematic view showing an embodiment of a cancer cell concentration filter.
- FIG. 2 is a graph showing the results of Experimental Example I-3. A: As a result of applying the polymer solution once, B: As a result of applying the polymer solution twice, C: Positive control.
- FIG. 3 is a graph showing the results of Experimental Example I-4.
- FIG. 4 is a graph showing the results of Experimental Example I-5.
- FIG. 5 is a graph showing the results of Experimental Example II-4.
- FIG. 6 is a graph showing the results of Experimental Example III-4.
- the cancer cell adhesion improver comprises a polymer containing a structural unit represented by the following general formula (1).
- R 1 is a hydrogen atom or a methyl group
- R 2 is a methyl group or an ethyl group
- m is 1 to 3.
- the number average molecular weight of the polymer is preferably 10,000 to 300,000. When the number average molecular weight is 10,000 or less, the above polymer becomes liquid and may be difficult to handle. Moreover, it may be difficult to make the number average molecular weight of the above-mentioned polymer 300,000 or more by ordinary radical polymerization.
- the above-mentioned polymer can be typically produced by adding an appropriate initiator to a monomer solution represented by the following general formula (2) and polymerizing by a general method.
- the temperature for carrying out the polymerization reaction is preferably 40 ° C. to 100 ° C., more preferably 60 ° C. to 90 ° C., still more preferably 70 ° C. to 80 ° C.
- the pressure for conducting the polymerization reaction is preferably normal pressure.
- R 3 is a hydrogen atom or a methyl group
- R 4 is a methyl or ethyl radical
- n is 1-3.
- a solvent that can dissolve the monomer represented by the general formula (2) can be used as the solvent.
- an aliphatic or aromatic organic solvent more specifically, ether solvents such as dioxane, tetrahydrofuran, diethyl ether, halogenated aromatic hydrocarbons such as o-dichlorobenzene, N, N-dimethylformamide, etc.
- the cancer cell adhesion improver is made of a polymer composed of a structural unit represented by the general formula (1).
- R 1 , R 2 and m in the general formula (1) are preferably a combination of any of the following (a) to (h).
- R 1 is a methyl group, R 2 is a methyl group, and m is 3.
- G R 1 is a methyl group, R 2 is an ethyl group, and m is 1 to 2.
- R 1 is a methyl group, R 2 is an ethyl group, and m is 3.
- R 1 , R 2 and m are the above combinations, the adhesion of blood cells to the substrate coated with the cancer cell adhesion improver is lowered and the effect of improving the adhesion of cancer cells is further enhanced. be able to.
- the cancer cell adhesion improver is a random copolymer, a block copolymer, or a graft copolymer of the monomer represented by the general formula (2) and another polymerizable monomer.
- Monomers that can be polymerized with the monomer represented by the general formula (2) include alkyl acrylamides such as acrylamide, t-butyl acrylamide, n-butyl acrylamide, i-butyl acrylamide, hexyl acrylamide, heptyl acrylamide, N, N- N, N-dialkylacrylamides such as dimethylacrylamide and N, N-diethylacrylamide, aminoalkyl acrylates such as aminomethyl acrylate, aminoethyl acrylate and aminoisopropyl acrylate, diaminoalkyls such as diaminomethyl acrylate, diaminoethyl acrylate and diaminobutyl acrylate N, N-dials such as acrylate, methacrylamide, N, N-dimethylmethacrylamide, N, N-diethylmethacrylamide Aminomethacrylate such as methacrylamide, aminomethyl methacrylate, aminoethyl me
- the monomer to be polymerized with the monomer represented by the general formula (2) is at least one selected from the group consisting of alkyl acrylate, alkyl methacrylate, alkoxy (meth) acrylate, alkoxyalkyl (meth) acrylate, glycidyl methacrylate and propylene. It may be a monomer.
- the monomer represented by the general formula (2) and the copolymer with the polymerizable monomer may be any of a random copolymer, a block copolymer, and a graft copolymer. Random polymerization, ionic polymerization, polymerization using a macromer, etc. Can be manufactured.
- the monomer represented by the general formula (2) is copolymerized with the polymerizable monomer, the monomer represented by the general formula (2) is 30 to 99% by mass in the copolymer. It is preferably 50 to 99% by mass.
- the polymer containing the structural unit represented by the general formula (1) is typically an initiator suitable for a solution of the monomer represented by the general formula (2) and, optionally, the above-described polymerizable.
- One or more monomers can be added and polymerized by a method such as random polymerization, ionic polymerization, photopolymerization, or polymerization using a macromer.
- the temperature for carrying out the polymerization reaction is preferably 40 ° C. to 100 ° C., more preferably 60 ° C. to 90 ° C., still more preferably 70 ° C. to 80 ° C.
- the pressure for conducting the polymerization reaction is preferably normal pressure.
- a solvent that can dissolve the monomer represented by the general formula (2) and the polymerizable monomer can be used as the solvent.
- a solvent is, for example, an aliphatic or aromatic organic solvent, and more specifically, an ether solvent such as dioxane, tetrahydrofuran or diethyl ether, a halogenated aromatic hydrocarbon such as o-dichlorobenzene, N Amides such as N, dimethylformamide, sulfoxides such as dimethyl sulfoxide, aromatic hydrocarbons such as benzene and toluene, and aliphatic hydrocarbons such as hexane and pentane, and ether solvents such as dioxane are preferred.
- a cancer cell adhesion improver When a cancer cell adhesion improver is applied to the surface of a substrate such as a cancer cell concentration filter, blood compatibility is imparted, so that activation of blood components can be suppressed during blood contact, and adhesion of blood cell components to the filter is prevented. It becomes possible to reduce. Moreover, since the cancer cell concentration filter to which the cancer cell adhesion improving agent is applied has good adhesion to various cancer cells, it can concentrate cancer cells efficiently.
- the polymer containing the structural unit represented by the general formula (1) has an ether bond and an ester bond as polar groups in the molecule, and these polar groups include a nitrogen atom (amino group, imino group), a carboxyl group, and the like. Unlike the biological component, it does not have a strong electrostatic interaction. Moreover, since it does not have a large hydrophobic group, the hydrophobic interaction is small. For this reason, it is estimated that the polymer containing the structural unit represented by the general formula (1) has low activity against blood and exhibits excellent blood compatibility.
- the protein is not adsorbed and denatured or activated.
- hydrophobic interaction which is a large interaction between substances, is desirable. It is considered useful to reduce the action and electrostatic interaction.
- the surface to which the cancer cell adhesion improving agent is applied can have a suitable surface structure.
- the surface to which the cancer cell adhesion improving agent is applied has moderate hydrophilicity, even when it comes into contact with blood, the adhesion of platelets is slight and expresses excellent blood compatibility. Moreover, it is thought that the interaction with the biological component by the hydrogen bond resulting from the hydroxyl group, denaturation of the adsorbed protein, and the like are also suppressed.
- a cancer cell adhesion improver By applying a cancer cell adhesion improver to the surface of a substrate such as a cancer cell concentration filter, adhesion of blood cell components such as red blood cells, white blood cells, and platelets to the filter is reduced, and at the same time, cancer cells are selectively and efficiently captured. It becomes possible to do. Although details about the mechanism of this function have not been clarified, the inventors presume that it can be explained by the concept of intermediate water.
- the water in contact with the substrate to which the cancer cell adhesion improver is applied includes (1) free water that weakly interacts with the cancer cell adhesion improver and melts at 0 ° C., (2) Antifreeze water that has strong interaction with cancer cell adhesion improver and does not freeze even at -100 °C, (3) Interplay between free water and antifreeze water, and freezes below 0 °C Intermediate water is thought to exist.
- Normal blood cells have a hydration shell and antifreeze water, intermediate water, free water, etc. that are in contact with the cancer cell adhesion improver, and are stabilized by this hydration structure.
- the antifreeze water that strongly reflects the structure of the cell adhesion improver is camouflaged and adhesion to the substrate surface is suppressed.
- cancer cells since the expression of sugar chains on the cell surface is different from that of normal cells, cancer cells are considered to have a disordered hydration structure compared to normal blood cells. Thereby, it is considered that the structure of the intermediate water on the substrate surface is disturbed and the adhesion to the substrate surface is improved.
- the cancer cell adhesion improving agent can be applied to all types of cancer cells, but is preferably applied to cancer derived from epithelial cells that easily cause blood vessel invasion, and among them, CTC is considered to be large. It is particularly preferred to apply to digestive organ cancers such as lung cancer, colon cancer, stomach cancer or esophageal cancer, breast cancer and prostate cancer.
- the present invention provides a cancer cell concentration filter comprising a substrate having a plurality of through-holes, wherein at least a part of the substrate is coated with a cancer cell adhesion improver.
- This filter can concentrate cancer cells such as CTC in blood.
- the opening shape of the through hole include a circle, an ellipse, a rectangle, a rounded rectangle, and a polygon.
- a rounded rectangle is a shape composed of two long sides of equal length and two semicircles. From the viewpoint of efficiently capturing cancer cells, a circle, rectangle, or rounded rectangle is preferable. In addition, a rounded rectangle is particularly preferable from the viewpoint of preventing clogging of the filter.
- FIG. 1 is a schematic view showing one embodiment of a cancer cell concentration filter.
- the filter 100 includes a substrate 20 in which a plurality of through holes 10 are formed.
- the opening shape of the through hole 10 is a rounded rectangle.
- CTC is captured on the surface 30 of the substrate 20.
- At least a portion of the surface 30 is coated with a cancer cell adhesion improver.
- the cancer cell adhesion improver preferably covers the entire surface 30. Part or all of the surface opposite to the surface 30 may be coated with a cancer cell adhesion improver.
- Coating is the most common method for applying a cancer cell adhesion improver to the surface of a substrate such as a cancer cell concentration filter.
- the coating is performed by adhering a polymer solution containing the structural unit represented by the general formula (1) to the substrate surface by dipping, spraying, spin coating, or the like, and then removing (drying) the solvent. Done.
- the film thickness after drying is preferably 0.01 ⁇ m to 1.0 mm, more preferably 0.1 to 100 ⁇ m, still more preferably 0.5 to 50 ⁇ m. If the film thickness is less than 0.01 ⁇ m, non-adhesiveness with blood cell components and adhesiveness with cancer cells may not be sufficiently developed. Further, when the film thickness exceeds 1.0 mm, the balance of these adhesive properties may be lost.
- crosslinking it is possible to exemplify adding a crosslinking monomer to a polymer material in advance.
- electron beam, ⁇ -ray, or light irradiation may be used.
- a polymer layer containing the structural unit represented by the general formula (1) on the surface of the substrate by plasma graft polymerization about 1.3 ⁇ 10 ⁇ 1 Pa, preferably 1.3 to 133.3 Pa.
- the monomer represented by the general formula (2) is supplied. Then, plasma-initiated polymerization may be performed.
- the material and shape of the substrate are not particularly limited, and for example, a porous body, a fiber, a nonwoven fabric, a film, a sheet, and a tube can be used.
- Substrate materials include natural polymers such as cotton and linen, nylon, polyester, polyacrylonitrile, polyolefin, halogenated polyolefin, polyurethane, polyamide, polysulfone, polyethersulfone, poly (meth) acrylate, and halogenated polyolefin ethylene-polyvinyl. Examples thereof include synthetic polymers such as alcohol copolymers and butadiene-acrylonitrile copolymers, or mixtures thereof.
- a metal, ceramics, these composite materials, etc. can be illustrated and may be comprised from the some board
- substrate can be illustrated and may be comprised from the some board
- the metal examples include, but are not limited to, noble metals such as gold and silver, base metals such as copper, aluminum, tungsten, nickel and chromium, and alloys of these metals.
- the metal may be used alone, or may be used as an alloy with another metal or an oxide of a metal in order to impart functionality. From the viewpoint of price and availability, it is preferable to use nickel, copper, and metals containing these as main components.
- the main component refers to a component occupying 50% by weight or more of the material forming the substrate.
- the general size of CTC is 10 ⁇ m or more in diameter.
- the diameter of a cell means the length of the longest straight line connecting two arbitrary points on the outline of the cell when observed with a microscope.
- the cancer cell concentration filter preferably has an average pore diameter of 5 ⁇ m or more and less than 30 ⁇ m and an average aperture ratio of 5% or more and less than 50%. Further, the average pore diameter is 5 ⁇ m or more and less than 15 ⁇ m, the average aperture ratio is more preferably 10% or more and less than 40%, the average pore diameter is 5 ⁇ m or more and less than 10 ⁇ m, and the average aperture ratio is 20% or more and less than 40%. It is particularly preferred.
- the aperture ratio refers to the area occupied by the through holes with respect to the entire area of the filter.
- the average aperture ratio is preferably as large as possible from the viewpoint of prevention of clogging. However, if the average opening ratio exceeds the above upper limit, the strength of the filter may be lowered or processing may be difficult. On the other hand, if it is less than 5%, the cancer cell concentration performance of the filter may decrease.
- the hole diameter when the opening shape is other than a circle such as an ellipse, rectangle, or polygon is the maximum value of the diameter of a sphere that can pass through each through hole.
- the diameter of the through hole is, for example, the length of the short side of the rectangle when the opening shape is a rectangle, and the diameter of the inscribed circle of the polygon when the opening shape is a polygon.
- the thickness of the filter substrate is preferably 3 to 100 ⁇ m, more preferably 5 to 50 ⁇ m, and particularly preferably 10 to 30 ⁇ m. If the thickness of the substrate is less than 3 ⁇ m, the strength of the filter may be reduced and handling may be difficult. On the other hand, if the thickness of the substrate exceeds 100 ⁇ m, more materials than necessary are consumed and processing takes a long time, which may be disadvantageous in cost and may make precision processing itself difficult.
- the manufacturing method of the filter of this embodiment is not specifically limited, For example, it forms by the electrocasting method (electroforming method).
- the electrocasting method is a method of peeling after performing thick electroplating on the mother die.
- a photosensitive resist film photosensitive layer
- a mask having a pattern of the opening shape of the through hole of the filter is fixed on the photosensitive layer.
- light active light is irradiated from above the mask.
- the photosensitive layer After light irradiation, if there is a support on the photosensitive layer, it is removed and the unexposed part is removed by wet development with a developer such as an alkaline aqueous solution, aqueous developer and organic solvent, or dry development. Development is performed to form a resist pattern. Subsequently, plating is performed on the exposed substrate without being masked using the developed resist pattern as a mask. Examples of the plating method include copper plating, solder plating, nickel plating, and gold plating. After the plating treatment, the plating layer is obtained by peeling the plating layer from the support and the photosensitive layer. By coating at least part of this plating layer with a cancer cell adhesion improver by the above method, a cancer cell concentration filter can be obtained.
- a cancer cell concentration filter can be obtained.
- the present invention provides a method for detecting the presence of cancer cells, comprising a filtration step of filtering a sample with the above-described cancer cell concentration filter.
- a sample for concentrating cancer cells such as CTC
- blood pooled in bone marrow, spleen, liver, etc., lymph, tissue fluid, umbilical cord blood, etc. can be used, but peripheral blood circulating in the body Is the simplest to use. Detecting the presence of CTC in peripheral blood is a useful means of determining the progression of cancer pathology.
- the method for detecting the presence of cancer cells includes, for example, incorporating the above-described cancer cell concentration filter into a flow channel, concentrating cells containing CTC by introducing peripheral blood into the flow channel, and concentrated cells. It can be carried out by confirming whether or not CTC exists in.
- Examples of the introduction of blood into the flow path include a method using pressurization from the flow path entrance direction, a method using pressure reduction from the flow path exit direction, and a method using a peristaltic pump.
- the area of the filter to be used is suitably 1 to 10 cm 2 when, for example, CTC is concentrated from 1 mL of blood.
- CTC When CTC is concentrated by the above method, not only CTC but also blood cells such as leukocytes are concentrated at the same time. For this reason, it is necessary to confirm whether or not cancer cells are contained in the collected cells. For example, after enriching CTC by the above method, it can be confirmed that it is a cancer cell by staining with an antibody against a fluorescently labeled cancer marker.
- antibodies against cancer markers include anti-EpCAM antibodies.
- the cells are cancer cells by analyzing the genes of the cells concentrated by the above method. For example, mutations in genes such as p53, K-RAS, H-RAS, N-RAS, BRAF, and APC can be analyzed to confirm that they are cancer cells. In addition, the results of these gene analyzes can also be used for the subsequent determination of the patient's treatment policy. Or it can also confirm that it is a cancer cell by measuring the telomerase activity etc. of the cell concentrated by said method.
- the concentrated cells can be cultured for further detailed analysis.
- the product was dissolved in tetrahydrofuran, further purified twice using n-hexane, and dried under reduced pressure overnight. A colorless, transparent and highly viscous polymer was obtained. The yield was 76%.
- GPC gel permeation chromatography
- the molecular weight of the polymer was calculated as a standard polystyrene equivalent molecular weight under the following GPC measurement conditions.
- Pump PU Intelligent HPLC Pump (manufactured by Jasco)
- Column GPC K804 (Showa Denko, Shodex)
- Eluent Chloroform Measurement temperature: Room temperature Flow rate: 1.0 mL / min
- Detector Jasco RI-1530 RI (manufactured by Jasco)
- Example I-2 (Production of nickel substrate) A commercially available titanium plate is immersed in an electrolytic bath for electrolytic nickel plating (nickel sulfamate 450 g / L, nickel chloride 5 g / L, boric acid 30 g / L, 55 ° C.) using the titanium plate as a cathode, and the anode is It was immersed in the same electrolytic bath. Nickel plating was performed by applying a voltage to both electrodes to a thickness of 10 ⁇ m, thereby preparing a nickel substrate.
- electrolytic nickel plating nickel sulfamate 450 g / L, nickel chloride 5 g / L, boric acid 30 g / L, 55 ° C.
- Example I-3 (Nickel substrate coating and confirmation)
- the polymer synthesized in Experimental Example I-1 was dissolved in chloroform to obtain a plurality of polymer solutions having different concentrations in the range of 0 to 5% by mass.
- Each polymer solution was applied (cast) onto the nickel substrate prepared in Experimental Example I-2, and the solvent was dried to coat the surface of the nickel substrate.
- a substrate on which the polymer was applied once and a substrate on which the polymer had been applied twice were prepared.
- a positive control a polyethylene terephthalate substrate coated with 40 ⁇ L of a polymer solution was used. The contact angle of water was measured on these substrates, and it was confirmed that they were coated with a polymer.
- FIG. 2 is a graph showing the results of Experimental Example I-3.
- the horizontal axis indicates the polymer concentration, and the vertical axis indicates the contact angle of water.
- A is the result of applying the polymer solution once
- B is the result of applying the polymer solution twice
- C is a positive control.
- Example I-4 Preparation of substrate
- the polymer synthesized in Experimental Example I-1 was dissolved in chloroform to obtain a 4% by mass solution. This solution was applied onto the nickel substrate prepared in Experimental Example I-2, the solvent was dried, and the surface of the nickel substrate was coated with a polymer, whereby the nickel substrate of Example I-1 was prepared. The polymer solution was applied twice. The nickel peak derived from the nickel substrate was not observed by X-ray photoelectron spectroscopy (Shimadzu Corporation, ESCA-1000), and the carbon and oxygen peaks derived from the polymer were detected. Therefore, the nickel substrate was coated with the polymer. It was confirmed. Further, the nickel substrate prepared in Experimental Example I-2 was used as the nickel substrate of Comparative Example I-1.
- Example I-1 Adhesion test of cancer cells
- 1.0 mL of a cancer cell suspension adjusted to 10,000 cells / mL with a medium supplemented with 10% serum was dropped with a pipette, and the temperature was 37 ° C. For 60 minutes.
- the human fibrosarcoma cell line HT-1080 was used as the cancer cell.
- the cells were rinsed with physiological buffer saline and the number of cells adhered to the substrate was counted.
- cells were fixed with formaldehyde, and then cell nuclei were stained with 4,6-diamino-2-phenylidole (DAPI). The number of cell nuclei was counted using a confocal laser microscope (Olympus FV-1000) to obtain the number of cells.
- DAPI 4,6-diamino-2-phenylidole
- FIG. 3 is a graph showing the results of Experimental Example I-4. The experiment was performed 5 times, and the results were shown as mean ⁇ standard deviation. The number of adhering cancer cells was improved in the nickel substrate of Example I-1 as compared with the nickel substrate of Comparative Example I-1. Similar results were confirmed in many other cancer cells.
- Example I-5 (Preparation of substrate) The experiment was conducted using the nickel substrates of Example I-1 and Comparative Example I-1 as in Experimental Example I-4.
- FIG. 4 is a graph showing the results of Experimental Example I-5. The experiment was performed 5 times, and the results were shown as mean ⁇ standard deviation. The nickel substrate of Example I-1 was found to have fewer adhered platelets than the nickel substrate of Comparative Example I-1.
- Example I-1 has low adhesion of blood cell components despite the improved adhesion of cancer cells. It was. Therefore, by applying the cancer cell adhesion improving agent of the present invention to the surface of a cancer cell concentration filter, it is possible to enhance the adhesion of cancer cells to the filter and improve the concentration rate of cancer cells.
- the product was isolated by dropping into n-hexane and precipitation.
- the product was dissolved in tetrahydrofuran and further purified twice using n-hexane.
- the purified product was dried under reduced pressure overnight.
- a colorless and transparent syrupy polymer was obtained.
- the yield (yield) was 11.4 g (76.0%).
- the obtained polymer structure was confirmed by 1H-NMR.
- the number average molecular weight (Mn) was 12,000 and the molecular weight distribution (Mw / Mn) was 3.9.
- the molecular weight of the polymer was calculated as a standard polystyrene equivalent molecular weight under the same GPC measurement conditions as in Experimental Example I-1.
- the product was isolated by dropping into n-hexane and precipitation.
- the product was dissolved in tetrahydrofuran and further purified twice using n-hexane.
- the purified product was dried under reduced pressure overnight.
- a colorless and transparent syrupy polymer was obtained.
- the yield (yield) was 8.2 g (82.0%).
- the obtained polymer structure was confirmed by 1H-NMR.
- the number average molecular weight (Mn) was 104,000, and the molecular weight distribution (Mw / Mn) was 4.6.
- the molecular weight of the polymer was calculated as a standard polystyrene equivalent molecular weight under the same GPC measurement conditions as in Experimental Example I-1.
- Poly (2-methacryloyloxyethyl phosphorylcholine-co-butyl methacrylate) (Synthesis of poly (2-methacryloyloxyethyl phosphorylcholine-co-butyl methacrylate))
- X and Y are 1 to 10,000, and X: Y is 40:60 mol% to 1:99 mol%.
- Example II-2 (Production of nickel substrate) The same treatment as in Experimental Example I-2 was performed to produce a nickel substrate.
- Example II-4 Preparation of substrate
- the four polymers synthesized in Experimental Example I-1 and Experimental Example II-1 were each dissolved in chloroform to obtain a 4 mass% solution. These solutions were applied onto the nickel substrate prepared in Experimental Example II-2, the solvent was dried, and the surface of the nickel substrate was coated with a polymer.
- Example II-1 polymethoxyethyl acrylate
- Example II-2 poly [2- (2-ethoxyethoxy) ethyl acrylate]
- Example II-3 poly [2- (2-methoxyethoxy) ethyl methacrylate]
- Comparative Example II-1 poly ( A nickel substrate of 2-methacryloyloxyethyl phosphorylcholine-co-butyl methacrylate)) was prepared. The polymer solution was applied twice. The nickel peak derived from the nickel substrate was not observed by X-ray photoelectron spectroscopy (Shimadzu Corporation, ESCA-1000), and the carbon and oxygen peaks derived from the polymer were detected. Therefore, the nickel substrate was coated with the polymer. It was confirmed.
- FIG. 5 is a graph showing the results of Experimental Example II-4. The experiment was performed 5 times, and the results were shown as mean ⁇ standard deviation.
- the nickel substrate of Example II-1 captured 2000 or more cancer cells.
- the nickel substrate of Example II-2 captured over 1500 cancer cells.
- the nickel substrate of Example II-3 captured 500 or more cancer cells. Compared to these, the nickel substrate of Comparative Example II-1 was able to capture only about 140 cancer cells.
- Initiator azobisisobutyronitrile (15.1 mg, 9.2 ⁇ 10 ⁇ 2 mmol) was dissolved in a small amount of 1,4-dioxane and polymerized at 75 ° C. for 2 hours and 10 minutes in a nitrogen atmosphere. Purification was performed using hexane. The reaction solution was dropped into 1500 mL of hexane, which is a poor solvent, and the solvent was removed by decantation. About 50 mL of tetrahydrofuran (THF) was added to the crude polymer to dissolve it, and the solution was dropped again into 1000 mL of hexane, and then the solvent was removed by decantation.
- THF tetrahydrofuran
- Example III-2 (Production of nickel substrate) The same treatment as in Experimental Example I-2 was performed to produce a nickel substrate.
- Example III-4 (Preparation of substrate) Five types of polymers synthesized in Experimental Example I-1, Experimental Example II-1 and Experimental Example III-1 were dissolved in chloroform to obtain a 4 mass% solution. These solutions were applied onto the nickel substrate prepared in Experimental Example III-2, the solvent was dried, and the surface of the nickel substrate was coated with a polymer.
- Example III-1 (polymethoxyethyl acrylate), Example III-2 (poly [2- (2-ethoxyethoxy) ethyl acrylate]), Example III-3 (poly [2- (2-methoxyethoxy) ethyl methacrylate]), Comparative Example III-1 (poly (2- A nickel substrate of methacryloyloxyethyl phosphorylcholine-co-butyl methacrylate)) and Example III-4 (poly [2- (2-ethoxyethoxy) ethyl methacrylate]) was prepared. The polymer solution was applied twice.
- the nickel peak derived from the nickel substrate was not observed by X-ray photoelectron spectroscopy (Shimadzu Corporation, ESCA-1000), and the carbon and oxygen peaks derived from the polymer were detected. Therefore, the nickel substrate was coated with the polymer. It was confirmed.
- FIG. 6 is a graph showing the results of Experimental Example III-4. The experiment was performed 5 times, and the results were shown as mean ⁇ standard deviation.
- the nickel substrates of Example III-1 and Example III-4 captured 6000 cells / cm 2 or more of cancer cells.
- the nickel substrate of Example III-2 captured 5000 cells / cm 2 or more of cancer cells.
- the nickel substrate of Example III-3 captured 2000 cells / cm 2 or more of cancer cells.
- the nickel substrate of Comparative Example III-1 was able to capture cancer cells only at about 130 cells / cm 2 .
Landscapes
- Physics & Mathematics (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Immunology (AREA)
- Pathology (AREA)
- Apparatus Associated With Microorganisms And Enzymes (AREA)
- Investigating Or Analysing Biological Materials (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
(a)R1が水素原子であり、R2がメチル基であり、mが1~2である。
(b)R1が水素原子であり、R2がメチル基であり、mが3である。
(c)R1が水素原子であり、R2がエチル基であり、mが1~2である。
(d)R1が水素原子であり、R2がエチル基であり、mが3である。
(e)R1がメチル基であり、R2がメチル基であり、mが1~2である。
(f)R1がメチル基であり、R2がメチル基であり、mが3である。
(g)R1がメチル基であり、R2がエチル基であり、mが1~2である。
(h)R1がメチル基であり、R2がエチル基であり、mが3である。
(ポリメトキシエチルアクリレートの合成)
上記一般式(1)において、R1が水素であり、R2がメチル基であり、mが1である、ポリメトキシエチルアクリレートを合成した。具体的には、メトキシエチルアクリレート15gを1,4-ジオキサン60g中でアゾビスイソブチロニトリル(0.1質量%)を開始剤として、窒素バブリングしながら75℃で10時間重合を行った。重合反応終了後、n-ヘキサンに滴下し沈殿させ、生成物を単離した。生成物をテトラヒドロフランに溶解し、さらにn-ヘキサンを用いて2回精製を行い、一昼夜減圧乾燥した。無色透明で高粘度のポリマーが得られた。収率は76%であった。得られたポリマーをゲル浸透クロマトグラフィー(GPC)により解析した結果、数平均分子量が15,000で、分子量分布(Mw/Mn)が3.4であった。
ポンプ:PU Intelligent HPLC Pump (Jasco製)
カラム:GPC K804(昭和電工製、Shodex)
溶離液:クロロホルム
測定温度:室温
流量:1.0mL/分
検出器:Jasco RI-1530型RI(Jasco製)
(ニッケル基板の作製)
市販のチタン板上に、チタン板を陰極として電解ニッケルめっき用の電解浴(スルファミン酸ニッケル450g/L、塩化ニッケル5g/L、ホウ酸30g/Lの水溶液、55℃)中に浸し、陽極を同電解浴中に浸した。両極に電圧をかけて10μm厚になるようにニッケルめっきを行い、ニッケル基板を作製した。
(ニッケル基板の被覆及び確認)
実験例I-1で合成したポリマーをクロロホルムに溶解し、0~5質量%の範囲で濃度の異なる複数のポリマー溶液を得た。各ポリマー溶液を、実験例I-2で作製したニッケル基板上に塗布(キャスト)し、溶媒を乾燥させて、ニッケル基板の表面を被覆した。ポリマーの塗布を1回行った基板と、2回行った基板を作製した。陽性対照として、ポリエチレンテレフタレート基板上に、40μLのポリマー溶液を塗布したものを使用した。これらの基板上で水の接触角を測定し、ポリマーで被覆されていることを確認した。接触角の測定には、接触角測定装置(エルマ、MODEL G-1-100)を使用した。純水2μlを各基板の表面に滴下し、30秒後の静的接触角を測定した。図2は、実験例I-3の結果を示すグラフである。横軸はポリマーの濃度を示し、縦軸は水の接触角を示す。図2中、Aはポリマー溶液を1回塗布した結果であり、Bはポリマー溶液を2回塗布した結果であり、Cは陽性対照である。
(基板の準備)
実験例I-1で合成したポリマーを、クロロホルムに溶解し4質量%溶液を得た。この溶液を、実験例I-2で作製したニッケル基板上に塗布し、溶媒を乾燥させて、ニッケル基板の表面をポリマーで被覆し、実施例I-1のニッケル基板を作製した。ポリマー溶液の塗布は2回行った。X線光電子分光(島津製作所、ESCA-1000)により、ニッケル基板由来のニッケルのピークが観測されず、ポリマー由来の炭素と酸素のピークが検出されたことから、ニッケル基板がポリマーで被覆されていることを確認した。また、実験例I-2で作製したニッケル基板を比較例I-1のニッケル基板として使用した。
実施例I-1及び比較例I-1のニッケル基板上に、血清を10%添加した培地で10,000個/mLに調整した癌細胞懸濁液1.0mLをピペットで滴下し、37℃で60分間静置した。癌細胞として、ヒト線維肉腫細胞株HT-1080を使用した。続いて、生理緩衝食塩水を用いてリンスし、基板に接着した細胞の数をカウントした。カウントを容易にするために、ホルムアルデヒドで細胞を固定化後、4,6-diamino-2-phenylidole(DAPI)で細胞核を染色した。細胞核の数を、共焦点レーザー顕微鏡(オリンパスFV-1000)を用いてカウントし、細胞の数とした。
(基板の準備)
実験例I-4と同様に、実施例I-1及び比較例I-1のニッケル基板を使用して実験を行った。
実施例I-1及び比較例I-1のニッケル基板上に、クエン酸ナトリウムで抗凝固したヒト新鮮多血小板血漿0.2mLをピペットで滴下し、37℃で60分間静置した。続いてリン酸緩衝溶液でリンスし、グルタルアルデヒドで固定した後、基板を走査型電子顕微鏡で観察し、1×104μm2の面積に接着した血小板数をカウントした。
(ポリ[2-(2-エトキシエトキシ)エチルアクリレート]の合成)
上記一般式(1)において、R1が水素であり、R2がエチル基であり、mが2である、ポリ[2-(2-エトキシエトキシ)エチルアクリレート]を合成した。具体的には、2-(2-エトキシエトキシ)エチルアクリレート15gを1,4-ジオキサン60g中でアゾビスイソブチロニトリル(0.1質量%)を開始剤として、窒素バブリングしながら75℃で10時間重合を行った。重合反応終了後、n-ヘキサンに滴下し沈殿させ、生成物を単離した。生成物をテトラヒドロフランに溶解し、さらに2回n-ヘキサンを用いて精製を行った。精製物を一昼夜減圧乾燥した。無色透明で水飴状のポリマーが得られた。収量(収率)は11.4g(76.0%)であった。得られたポリマー構造は、1H-NMRによって確認した。GPCの分子量分析の結果、数平均分子量(Mn)が12,000であり分子量分布(Mw/Mn)が3.9であった。ポリマーの分子量は、実験例I-1と同様のGPCの測定条件にて標準ポリスチレン換算分子量で算出した。
上記一般式(1)において、R1がメチル基であり、R2がメチル基であり、mが2である、ポリ[2-(2-メトキシエトキシ)エチルメタクリレート]を合成した。具体的には、2-(2-メトキシエトキシ)エチルメタクリレート10gを1,4-ジオキサン50g中でアゾビスイソブチロニトリル(0.1質量%)を開始剤として、窒素バブリングしながら80℃で8時間重合した。重合反応終了後、n-ヘキサンに滴下し沈殿させ、生成物を単離した。生成物をテトラヒドロフランに溶解し、さらに2回n-ヘキサンを用いて精製を行った。精製物を一昼夜減圧乾燥した。無色透明で水飴状のポリマーが得られた。収量(収率)は8.2g(82.0%)であった。得られたポリマー構造は、1H-NMRによって確認した。GPCの分子量分析の結果、数平均分子量(Mn)が104,000であり分子量分布(Mw/Mn)が4.6であった。ポリマーの分子量は、実験例I-1と同様のGPCの測定条件にて標準ポリスチレン換算分子量で算出した。
下記一般式(3)で表される、ポリ(2-メタクリロイルオキシエチルホスホリルコリン-コ-ブチルメタクリレート)を合成した。ポリ(2-メタクリロイルオキシエチルホスホリルコリン-コ-ブチルメタクリレート)は、2-メタクリロイルオキシエチルホスホリルコリン(MPC)とブチルメタクリレート(BMA)の共重合体であり、MPC:BMA=40:60モル%~1:99モル%であった。
(ニッケル基板の作製)
実験例I-2と同様の処理を行ってニッケル基板を作製した。
(実験例I-3)と同様の処理を行ってニッケル基板にポリマーを被覆したときの水の静的接触角を測定した。その結果、ポリ[2-(2-エトキシエトキシ)エチルアクリレート]については、0.2wt/vol%以上で接触角が25°でほぼ安定した。また、ポリ[2-(2-メトキシエトキシ)エチルメタクリレート]については、0.5wt/vol%以上で接触角が38°でほぼ安定した。また、ポリ(2-メタクリロイルオキシエチルホスホリルコリン-コ-ブチルメタクリレート)については、0.1wt/vol%以上で接触角が104°でほぼ安定した。
(基板の準備)
実験例I-1及び実験例II-1で合成したポリマー4種を、それぞれクロロホルムに溶解し4質量%溶液を得た。これらの溶液を、実験例II-2で作製したニッケル基板上に塗布し、溶媒を乾燥させて、ニッケル基板の表面をポリマーで被覆し、それぞれ、実施例II-1(ポリメトキシエチルアクリレート)、実施例II-2(ポリ[2-(2-エトキシエトキシ)エチルアクリレート])、実施例II-3(ポリ[2-(2-メトキシエトキシ)エチルメタクリレート])及び比較例II-1(ポリ(2-メタクリロイルオキシエチルホスホリルコリン-コ-ブチルメタクリレート))のニッケル基板を作製した。ポリマー溶液の塗布は2回行った。X線光電子分光(島津製作所、ESCA-1000)により、ニッケル基板由来のニッケルのピークが観測されず、ポリマー由来の炭素と酸素のピークが検出されたことから、ニッケル基板がポリマーで被覆されていることを確認した。
実施例II-1~3及び比較例II-1のニッケル基板上に、血清を10%添加した培地で10,000個/mLに調整した癌細胞懸濁液1.0mLをピペットで滴下し、37℃で60分間静置した。癌細胞として、ヒト乳腺癌細胞株MDA-MB-231を使用した。続いて、生理緩衝食塩水を用いてリンスし、基板に接着した細胞の数をカウントした。カウントを容易にするために、ホルムアルデヒドで細胞を固定化後、4,6-diamino-2-phenylidole(DAPI)で細胞核を染色した。細胞核の数を、共焦点レーザー顕微鏡(オリンパスFV-1000)を用いてカウントし、細胞の数とした。
(ポリ[2-(2-エトキシエトキシ)エチルメタクリレート]の合成)
上記一般式(1)において、R1がメチル基であり、R2がエチル基であり、mが2である、ポリ[2-(2-エトキシエトキシ)エチルメタクリレート]を合成した。具体的には、2-(2-エトキシエトキシ)エチルメタクリレート15.0g(7.4×10-2mol)を1,4-ジオキサン58.2mLに溶解し、2時間N2パージを行った。開始剤アゾビスイソブチロニトリル15.1mg(9.2×10-2mmol)を少量の1,4-ジオキサンに溶解して加え、窒素雰囲気下75℃で2時間10分重合した。精製はヘキサンを用いて行った。反応液を貧溶媒であるヘキサン1500mL中に滴下し、デカンテーションによって溶媒を除いた。この粗ポリマーにテトラヒドロフラン(THF)約50mLを加えて溶解し、再びヘキサン1000mLに滴下して析出させたあと、デカンテーションによって溶媒を除いた。この操作をもう一度繰り返し、ポリマー中に含まれるモノマー及び開始剤を完全に除去した。一晩減圧乾燥し、質量を測定した。無色透明・水飴状のポリマー、ポリ[2-(2-エトキシエトキシ)エチルメタクリレート]が得られ、収量は5.36g、収率は35.7%であった。GPCの分子量分析の結果から、数平均分子量(Mn)が142,000であり分子量分布(Mw/Mn)が6.06であった。ポリマーの分子量は、実験例I-1と同様のGPCの測定条件にて標準ポリスチレン換算分子量で算出した。
(ニッケル基板の作製)
実験例I-2と同様の処理を行ってニッケル基板を作製した。
(実験例I-3)と同様の処理を行ってニッケル基板にポリマーを被覆したときの水の静的接触角を測定した結果、ポリ[2-(2-エトキシエトキシ)エチルメタクリレート]については、0.5wt/vol%以上で接触角が78°でほぼ安定した。
(基板の準備)
実験例I-1、実験例II-1及び実験例III-1で合成したポリマー5種を、クロロホルムに溶解し4質量%溶液を得た。これらの溶液を、実験例III-2で作製したニッケル基板上に塗布し、溶媒を乾燥させて、ニッケル基板の表面をポリマーで被覆し、実施例III-1(ポリメトキシエチルアクリレート)、実施例III-2(ポリ[2-(2-エトキシエトキシ)エチルアクリレート])、実施例III-3(ポリ[2-(2-メトキシエトキシ)エチルメタクリレート])、比較例III-1(ポリ(2-メタクリロイルオキシエチルホスホリルコリン-コ-ブチルメタクリレート))及び実施例III-4(ポリ[2-(2-エトキシエトキシ)エチルメタクリレート])のニッケル基板を作製した。ポリマー溶液の塗布は2回行った。X線光電子分光(島津製作所、ESCA-1000)により、ニッケル基板由来のニッケルのピークが観測されず、ポリマー由来の炭素と酸素のピークが検出されたことから、ニッケル基板がポリマーで被覆されていることを確認した。
実施例III-1~4及び比較例III-1のニッケル基板上に、血清を10%添加した培地で10,000個/mLに調整した癌細胞懸濁液1.0mLをピペットで滴下し、37℃で60分間静置した。癌細胞として、肺癌細胞株A549を使用した。続いて、生理緩衝食塩水を用いてリンスし、基板に接着した細胞の数をカウントした。カウントを容易にするために、ホルムアルデヒドで細胞を固定化後、4,6-diamino-2-phenylidole(DAPI)で細胞核を染色した。細胞核の数を、共焦点レーザー顕微鏡(オリンパスFV-1000)を用いてカウントし、細胞の数とした。
Claims (14)
- 前記一般式(1)で表される構造単位からなるポリマーからなる、請求項1に記載の癌細胞接着性向上剤。
- R1が水素原子であり、R2がメチル基であり、mが1である、請求項1又は2に記載の癌細胞接着性向上剤。
- R1が水素原子であり、R2がエチル基であり、mが2である、請求項1又は2に記載の癌細胞接着性向上剤。
- R1がメチル基であり、R2がメチル基であり、mが2である、請求項1又は2に記載の癌細胞接着性向上剤。
- 数平均分子量が10,000~300,000である、請求項1~5のいずれか一項に記載の癌細胞接着性向上剤。
- 複数の貫通孔が形成された基板からなり、当該基板の少なくとも一部が請求項1~6のいずれか一項に記載の癌細胞接着性向上剤で被覆されている、癌細胞濃縮フィルター。
- 前記貫通孔は、平均孔径が5μm以上30μm未満であり、平均開口率が5%以上50%未満である、請求項7に記載の癌細胞濃縮フィルター。
- 前記基板は金属からなる、請求項7又は8に記載の癌細胞濃縮フィルター。
- 前記金属は、銅、ニッケル、銅及びニッケルの合金並びにこれらの表面が金めっきされたものからなる群より選択されるものである、請求項9に記載の癌細胞濃縮フィルター。
- 前記貫通孔は、電気鋳造法によって形成されたものである、請求項9又は10に記載の癌細胞濃縮フィルター。
- 請求項7~11のいずれか一項に記載の癌細胞濃縮フィルターで末梢血を濾過する濾過工程を含む、癌細胞の存在を検出する方法。
- 前記濾過工程で濃縮された細胞の遺伝子を解析する工程を更に含む、請求項12に記載の方法。
- 前記濾過工程で濃縮された細胞を培養する工程を更に含む、請求項12又は13に記載の方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013520549A JP5975532B2 (ja) | 2011-06-13 | 2012-06-11 | 癌細胞接着性向上剤 |
| EP12801028.7A EP2720039B1 (en) | 2011-06-13 | 2012-06-11 | Agent for improving cancer cell adhesiveness |
| CN201280029309.2A CN103635801A (zh) | 2011-06-13 | 2012-06-11 | 癌细胞粘附性提高剂 |
| US14/126,049 US9372136B2 (en) | 2011-06-13 | 2012-06-11 | Agent for improving cancer cell adhesiveness |
| CA2839313A CA2839313C (en) | 2011-06-13 | 2012-06-11 | Agent for improving cancer cell adhesiveness |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011131241 | 2011-06-13 | ||
| JP2011-131241 | 2011-06-13 | ||
| JP2012123228 | 2012-05-30 | ||
| JP2012-123228 | 2012-05-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012173097A1 true WO2012173097A1 (ja) | 2012-12-20 |
Family
ID=47357084
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/064932 WO2012173097A1 (ja) | 2011-06-13 | 2012-06-11 | 癌細胞接着性向上剤 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US9372136B2 (ja) |
| EP (1) | EP2720039B1 (ja) |
| JP (1) | JP5975532B2 (ja) |
| CN (1) | CN103635801A (ja) |
| CA (1) | CA2839313C (ja) |
| TW (1) | TWI516294B (ja) |
| WO (1) | WO2012173097A1 (ja) |
Cited By (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014162810A1 (ja) * | 2013-04-04 | 2014-10-09 | 日立化成株式会社 | 生体物質捕獲用のフィルター |
| USD746467S1 (en) | 2014-04-04 | 2015-12-29 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| USD747487S1 (en) | 2014-04-04 | 2016-01-12 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| USD748267S1 (en) | 2014-04-04 | 2016-01-26 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| USD748801S1 (en) | 2014-04-04 | 2016-02-02 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| USD750785S1 (en) | 2014-04-04 | 2016-03-01 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| WO2016035772A1 (ja) * | 2014-09-03 | 2016-03-10 | 日立化成株式会社 | 生体物質捕獲用フィルター及び生体物質捕獲システム |
| JP2016052300A (ja) * | 2014-09-03 | 2016-04-14 | 日立化成株式会社 | 生体物質捕獲システム |
| US20160169781A1 (en) * | 2014-12-10 | 2016-06-16 | Hitachi Chemical Company, Ltd. | Cell-trapping system |
| JP2016131561A (ja) * | 2015-01-22 | 2016-07-25 | 国立大学法人山形大学 | 細胞を回収する方法、及びそれに用いられるポリマー |
| JP2017036511A (ja) * | 2011-10-14 | 2017-02-16 | 日立化成株式会社 | 金属フィルター、血液中の癌細胞を濃縮する方法 |
| JP2017042225A (ja) * | 2015-08-24 | 2017-03-02 | 旭化成メディカル株式会社 | 医療用部材 |
| JP2017102038A (ja) * | 2015-12-02 | 2017-06-08 | 国立大学法人山形大学 | 癌細胞接着剤及び癌細胞の検出方法 |
| JP2018050548A (ja) * | 2016-09-29 | 2018-04-05 | 住友ゴム工業株式会社 | がん細胞捕捉方法 |
| WO2018221496A1 (ja) * | 2017-06-02 | 2018-12-06 | 日立化成株式会社 | 血液試料中の希少細胞を分離する方法及び該細胞の遺伝子を解析する方法 |
| JP2019129836A (ja) * | 2019-03-27 | 2019-08-08 | 住友ゴム工業株式会社 | がん細胞捕捉方法 |
| JP2019138839A (ja) * | 2018-02-14 | 2019-08-22 | 住友ゴム工業株式会社 | 特定細胞捕捉方法 |
| JP2019138841A (ja) * | 2018-02-14 | 2019-08-22 | 住友ゴム工業株式会社 | 特定細胞捕捉方法 |
| JP2019138838A (ja) * | 2018-02-14 | 2019-08-22 | 住友ゴム工業株式会社 | 特定細胞捕捉方法 |
| WO2020153364A1 (ja) * | 2019-01-23 | 2020-07-30 | 富士フイルム株式会社 | 細胞付着用シート |
| JP2020118661A (ja) * | 2019-01-24 | 2020-08-06 | 住友ゴム工業株式会社 | 特定細胞の分画方法及び捕捉方法 |
| WO2022009968A1 (ja) * | 2020-07-09 | 2022-01-13 | 丸善石油化学株式会社 | 癌細胞接着用組成物、癌細胞捕集フィルター、及び癌細胞の検出方法 |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20150111293A1 (en) * | 2012-05-14 | 2015-04-23 | Hitachi Chemical Company, Ltd. | Cancer cell-trapping metal filter, cancer cell-trapping metal filter sheet, cancer cell-trapping device, and manufacturing methods therefor |
| US9739783B1 (en) * | 2016-03-15 | 2017-08-22 | Anixa Diagnostics Corporation | Convolutional neural networks for cancer diagnosis |
| EP3244208A1 (en) | 2016-05-09 | 2017-11-15 | Sumitomo Rubber Industries, Ltd. | Medical analysis device and cell analysis method |
| JP6779483B2 (ja) * | 2016-09-29 | 2020-11-04 | 住友ゴム工業株式会社 | 医療用検査装置及び細胞検査方法 |
| US9934364B1 (en) | 2017-02-28 | 2018-04-03 | Anixa Diagnostics Corporation | Methods for using artificial neural network analysis on flow cytometry data for cancer diagnosis |
| US11164082B2 (en) | 2017-02-28 | 2021-11-02 | Anixa Diagnostics Corporation | Methods for using artificial neural network analysis on flow cytometry data for cancer diagnosis |
| US10360499B2 (en) | 2017-02-28 | 2019-07-23 | Anixa Diagnostics Corporation | Methods for using artificial neural network analysis on flow cytometry data for cancer diagnosis |
| US11614440B2 (en) | 2019-01-24 | 2023-03-28 | Sumitomo Rubber Industries, Ltd. | Specific cell fractionating and capturing methods |
| CN112870852B (zh) * | 2020-12-30 | 2022-11-04 | 新疆浩天能环保科技有限公司 | 一种三元净化细孔陶瓷滤芯 |
| TWI827188B (zh) * | 2022-08-04 | 2023-12-21 | 國立清華大學 | 用於去除全血中紅血球的晶片及方法 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004087228A1 (ja) * | 2003-03-28 | 2004-10-14 | Japan Science And Technology Agency | 生体適合性と温度応答性を併せ持つ高分子 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1059979A4 (en) | 1997-11-07 | 2001-05-30 | California Inst Of Techn | MICRO-MADE PARTICLE FILTER MEMBRANE WITH PARYLENE REINFORCEMENT |
| AU4645299A (en) | 1998-07-08 | 2000-02-01 | Advanced Biocompatible Coatings Inc. | Biocompatible metallic stents with hydroxy methacrylate coating |
| WO2005043121A2 (en) * | 2003-10-31 | 2005-05-12 | Vitatex, Inc. | Blood test prototypes and methods for the detection of circulating tumor and endothelial cells |
| WO2006116327A1 (en) * | 2005-04-21 | 2006-11-02 | California Institute Of Technology | Uses of parylene membrane filters |
| CN101583722A (zh) | 2006-07-14 | 2009-11-18 | 阿维瓦生物科学股份有限公司 | 从生物学样品检测稀有细胞的方法和组合物 |
| EP2097119A4 (en) | 2006-11-21 | 2012-10-17 | Abbott Lab | USE OF A TERPOLYMER OF TETRAFLUOROETHYLENE, HEXAFLUORPROPYLENE AND VINYLIDENE FLUORIDE IN MEDICAMENTAL COATINGS |
| CN101769930A (zh) | 2010-01-08 | 2010-07-07 | 卢小明 | 氟代化合物在鉴定循环肿瘤细胞中的应用方法 |
-
2012
- 2012-06-11 US US14/126,049 patent/US9372136B2/en not_active Expired - Fee Related
- 2012-06-11 CA CA2839313A patent/CA2839313C/en not_active Expired - Fee Related
- 2012-06-11 WO PCT/JP2012/064932 patent/WO2012173097A1/ja active Application Filing
- 2012-06-11 EP EP12801028.7A patent/EP2720039B1/en not_active Not-in-force
- 2012-06-11 CN CN201280029309.2A patent/CN103635801A/zh active Pending
- 2012-06-11 JP JP2013520549A patent/JP5975532B2/ja not_active Expired - Fee Related
- 2012-06-13 TW TW101121020A patent/TWI516294B/zh not_active IP Right Cessation
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004087228A1 (ja) * | 2003-03-28 | 2004-10-14 | Japan Science And Technology Agency | 生体適合性と温度応答性を併せ持つ高分子 |
Non-Patent Citations (7)
| Title |
|---|
| MASATO HOSOKAWA ET AL.: "Development of microfluidic device for rapid detection of circulating tumor cells", CHEM. SENS., vol. 26, no. SUPP.B, 2 September 2010 (2010-09-02), pages 40 - 42, XP008173145 * |
| MASATO HOSOKAWA ET AL.: "Zenketsu kara no Junkan Shuyo Saibo Kokoritsu Kenshutsu Device no Kaihatsu", CSJ, vol. 90, no. 3, 12 March 2010 (2010-03-12), pages 659, XP008172941 * |
| SATOMI YAGI ET AL.: "Kekkyu Saibo no Hi Secchakusei to Gan Saibo no Secchakusei o Awasemotsu Kobunshi Hyomen no Sosei", REGENERATIVE MEDICINE, vol. 10, 1 February 2011 (2011-02-01), pages 264, XP008172556 * |
| SATOMI YAGI ET AL.: "Ketsueki Tekigosei Kobunshi Hyomenjo deno Hito Gan Saibo no Sentakuteki Secchaku", POLYMER PREPRINTS, JAPAN, vol. 60, no. 1, 10 May 2011 (2011-05-10), pages 1884, XP008173098 * |
| SATOMI YAGI ET AL.: "Poly (2-methoxyethylacrylate) Hyomenjo deno Hito Gan Saibo no Sentakuteki Secchaku", THE ANNUAL MEETING OF THE JAPANESE SOCIETY FOR BIOMATERIALS YOKOSHU, vol. 33, 21 November 2011 (2011-11-21), pages 306, XP008173130 * |
| TAKASHI HOSHIBA ET AL.: "Ketsueki Tekigosei Kobunshi Kibanjo eno Gan Saibo no Secchaku Kiko no Kaiseki", POLYMER PREPRINTS, vol. 61, no. 1, 15 May 2012 (2012-05-15), JAPAN, pages 1689, XP008173097 * |
| ZHENG S. ET AL.: "3D microfilter device for viable circulating tumor cell (CTC)enrichment from blood", BIOMED. MICRODEVICES, vol. 13, no. 1, February 2011 (2011-02-01), pages 203 - 213, XP019877255 * |
Cited By (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017036511A (ja) * | 2011-10-14 | 2017-02-16 | 日立化成株式会社 | 金属フィルター、血液中の癌細胞を濃縮する方法 |
| EP2982743A4 (en) * | 2013-04-04 | 2016-11-30 | Hitachi Chemical Co Ltd | FILTER FOR DETECTING A BIOLOGICAL SUBSTANCE |
| CN105102606A (zh) * | 2013-04-04 | 2015-11-25 | 日立化成株式会社 | 生物体物质捕获用的过滤器 |
| JPWO2014162810A1 (ja) * | 2013-04-04 | 2017-02-16 | 日立化成株式会社 | 生体物質捕獲用のフィルター |
| WO2014162810A1 (ja) * | 2013-04-04 | 2014-10-09 | 日立化成株式会社 | 生体物質捕獲用のフィルター |
| JP2018088932A (ja) * | 2013-04-04 | 2018-06-14 | 日立化成株式会社 | 生体物質捕獲用のフィルター |
| USD750785S1 (en) | 2014-04-04 | 2016-03-01 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| USD751189S1 (en) | 2014-04-04 | 2016-03-08 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| USD748801S1 (en) | 2014-04-04 | 2016-02-02 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| USD752227S1 (en) | 2014-04-04 | 2016-03-22 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| USD748267S1 (en) | 2014-04-04 | 2016-01-26 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| USD747487S1 (en) | 2014-04-04 | 2016-01-12 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| USD746467S1 (en) | 2014-04-04 | 2015-12-29 | Hitachi Chemical Company, Ltd. | Filter sheet for a cell collecting cartridge |
| WO2016035772A1 (ja) * | 2014-09-03 | 2016-03-10 | 日立化成株式会社 | 生体物質捕獲用フィルター及び生体物質捕獲システム |
| JP2016052300A (ja) * | 2014-09-03 | 2016-04-14 | 日立化成株式会社 | 生体物質捕獲システム |
| JPWO2016035772A1 (ja) * | 2014-09-03 | 2017-05-25 | 日立化成株式会社 | 生体物質捕獲用フィルター及び生体物質捕獲システム |
| US20160169781A1 (en) * | 2014-12-10 | 2016-06-16 | Hitachi Chemical Company, Ltd. | Cell-trapping system |
| JP2016131561A (ja) * | 2015-01-22 | 2016-07-25 | 国立大学法人山形大学 | 細胞を回収する方法、及びそれに用いられるポリマー |
| JP2017042225A (ja) * | 2015-08-24 | 2017-03-02 | 旭化成メディカル株式会社 | 医療用部材 |
| JP2017102038A (ja) * | 2015-12-02 | 2017-06-08 | 国立大学法人山形大学 | 癌細胞接着剤及び癌細胞の検出方法 |
| JP2018050548A (ja) * | 2016-09-29 | 2018-04-05 | 住友ゴム工業株式会社 | がん細胞捕捉方法 |
| WO2018221496A1 (ja) * | 2017-06-02 | 2018-12-06 | 日立化成株式会社 | 血液試料中の希少細胞を分離する方法及び該細胞の遺伝子を解析する方法 |
| JP7170254B2 (ja) | 2018-02-14 | 2022-11-14 | 住友ゴム工業株式会社 | 特定細胞捕捉方法 |
| JP2019138839A (ja) * | 2018-02-14 | 2019-08-22 | 住友ゴム工業株式会社 | 特定細胞捕捉方法 |
| JP2019138841A (ja) * | 2018-02-14 | 2019-08-22 | 住友ゴム工業株式会社 | 特定細胞捕捉方法 |
| JP2019138838A (ja) * | 2018-02-14 | 2019-08-22 | 住友ゴム工業株式会社 | 特定細胞捕捉方法 |
| JP7158671B2 (ja) | 2018-02-14 | 2022-10-24 | 住友ゴム工業株式会社 | 特定細胞捕捉方法 |
| JP7109719B2 (ja) | 2018-02-14 | 2022-08-01 | 住友ゴム工業株式会社 | 特定細胞捕捉方法 |
| JPWO2020153364A1 (ja) * | 2019-01-23 | 2021-11-18 | 富士フイルム株式会社 | 細胞付着用シート |
| WO2020153364A1 (ja) * | 2019-01-23 | 2020-07-30 | 富士フイルム株式会社 | 細胞付着用シート |
| JP7199065B2 (ja) | 2019-01-23 | 2023-01-05 | 富士フイルム株式会社 | 細胞付着用シート |
| JP2020118661A (ja) * | 2019-01-24 | 2020-08-06 | 住友ゴム工業株式会社 | 特定細胞の分画方法及び捕捉方法 |
| JP2019129836A (ja) * | 2019-03-27 | 2019-08-08 | 住友ゴム工業株式会社 | がん細胞捕捉方法 |
| WO2022009968A1 (ja) * | 2020-07-09 | 2022-01-13 | 丸善石油化学株式会社 | 癌細胞接着用組成物、癌細胞捕集フィルター、及び癌細胞の検出方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2720039A4 (en) | 2015-01-28 |
| CN103635801A (zh) | 2014-03-12 |
| TWI516294B (zh) | 2016-01-11 |
| US20140178890A1 (en) | 2014-06-26 |
| TW201302252A (zh) | 2013-01-16 |
| CA2839313C (en) | 2018-03-13 |
| JPWO2012173097A1 (ja) | 2015-02-23 |
| CA2839313A1 (en) | 2012-12-20 |
| EP2720039A1 (en) | 2014-04-16 |
| JP5975532B2 (ja) | 2016-08-23 |
| US9372136B2 (en) | 2016-06-21 |
| EP2720039B1 (en) | 2017-07-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5975532B2 (ja) | 癌細胞接着性向上剤 | |
| JP5961889B2 (ja) | 末梢循環腫瘍細胞分離用デバイス、希少細胞分離用デバイス、末梢循環腫瘍細胞分離方法及び希少細胞分離方法 | |
| US7793787B2 (en) | Polymers useful as medical materials | |
| NL1038359C2 (en) | Device and method for separation of circulating tumor cells. | |
| US8136676B2 (en) | Polymers useful as medical materials | |
| JP6518985B2 (ja) | がん細胞捕捉方法 | |
| US10317406B2 (en) | System for detecting rare cells | |
| JP2016131561A (ja) | 細胞を回収する方法、及びそれに用いられるポリマー | |
| US20140120537A1 (en) | Collection and Concentration System for Biologic Substance of Interest and Use Thereof | |
| EP1473310A1 (en) | Coating material for leukocyte removal filter and the filter material | |
| US11360078B2 (en) | Medical analysis device and cell analysis method | |
| JP7158671B2 (ja) | 特定細胞捕捉方法 | |
| JP6601768B2 (ja) | 癌細胞接着剤及び癌細胞の検出方法 | |
| JP4838594B2 (ja) | セルアレイソータ、その製造方法及びそれを用いた細胞ソート方法 | |
| JP6080035B2 (ja) | 膜、基体及び細胞の採取方法 | |
| EP3490696A2 (en) | Flow capture device and method for removing cells from blood | |
| JP5262708B2 (ja) | mRNA捕捉用担体及びmRNAの精製方法 | |
| WO2023013410A1 (ja) | 高分子組成物 | |
| JP6886667B2 (ja) | がん細胞捕捉方法 | |
| US20240293784A1 (en) | Thermo-induced stimuli-responsive membrane for leukocyte enrichment and application thereof | |
| Fu et al. | Thermo-responsive bioseparation engineered for human leukocyte enrichment process driven by functionalized polypropylene bio-separators |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12801028 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2013520549 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2839313 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2012801028 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14126049 Country of ref document: US |