WO2010041330A1 - フッ素不溶化剤、含有フッ素の溶出を低減させた石膏及びフッ素汚染土壌の処理方法 - Google Patents
フッ素不溶化剤、含有フッ素の溶出を低減させた石膏及びフッ素汚染土壌の処理方法 Download PDFInfo
- Publication number
- WO2010041330A1 WO2010041330A1 PCT/JP2008/068441 JP2008068441W WO2010041330A1 WO 2010041330 A1 WO2010041330 A1 WO 2010041330A1 JP 2008068441 W JP2008068441 W JP 2008068441W WO 2010041330 A1 WO2010041330 A1 WO 2010041330A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- fluorine
- gypsum
- water
- insolubilizing agent
- hydrogen phosphate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B09—DISPOSAL OF SOLID WASTE; RECLAMATION OF CONTAMINATED SOIL
- B09B—DISPOSAL OF SOLID WASTE NOT OTHERWISE PROVIDED FOR
- B09B3/00—Destroying solid waste or transforming solid waste into something useful or harmless
-
- A—HUMAN NECESSITIES
- A62—LIFE-SAVING; FIRE-FIGHTING
- A62D—CHEMICAL MEANS FOR EXTINGUISHING FIRES OR FOR COMBATING OR PROTECTING AGAINST HARMFUL CHEMICAL AGENTS; CHEMICAL MATERIALS FOR USE IN BREATHING APPARATUS
- A62D3/00—Processes for making harmful chemical substances harmless or less harmful, by effecting a chemical change in the substances
- A62D3/30—Processes for making harmful chemical substances harmless or less harmful, by effecting a chemical change in the substances by reacting with chemical agents
- A62D3/33—Processes for making harmful chemical substances harmless or less harmful, by effecting a chemical change in the substances by reacting with chemical agents by chemical fixing the harmful substance, e.g. by chelation or complexation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B09—DISPOSAL OF SOLID WASTE; RECLAMATION OF CONTAMINATED SOIL
- B09C—RECLAMATION OF CONTAMINATED SOIL
- B09C1/00—Reclamation of contaminated soil
- B09C1/02—Extraction using liquids, e.g. washing, leaching, flotation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B09—DISPOSAL OF SOLID WASTE; RECLAMATION OF CONTAMINATED SOIL
- B09C—RECLAMATION OF CONTAMINATED SOIL
- B09C1/00—Reclamation of contaminated soil
- B09C1/08—Reclamation of contaminated soil chemically
-
- A—HUMAN NECESSITIES
- A62—LIFE-SAVING; FIRE-FIGHTING
- A62D—CHEMICAL MEANS FOR EXTINGUISHING FIRES OR FOR COMBATING OR PROTECTING AGAINST HARMFUL CHEMICAL AGENTS; CHEMICAL MATERIALS FOR USE IN BREATHING APPARATUS
- A62D2101/00—Harmful chemical substances made harmless, or less harmful, by effecting chemical change
- A62D2101/40—Inorganic substances
- A62D2101/49—Inorganic substances containing halogen
Definitions
- the present invention relates to a fluorine insolubilizer, gypsum in which elution of contained fluorine is reduced using the same, and a method for treating fluorine-contaminated soil using the same.
- a fluorine insolubilizer gypsum in which elution of contained fluorine is reduced using the same
- a method for treating fluorine-contaminated soil using the same For example, when landfilling gypsum board waste materials used as building materials, from the viewpoint of environmental protection, the amount of fluorine elution from such waste materials is required to be as low as possible. As a guideline, 0.8 mg / L or less is required. Needless to say, the main raw material of gypsum board is gypsum, and there are natural gypsum and artificially produced or by-produced gypsum.
- the elution amount of fluorine from gypsum waste material containing fluorine or soil contaminated with fluorine is one guideline, and 0.8 mg / L or less of the soil environment standard
- the present invention relates to a fluorine insolubilizing agent that can be reduced to a level, gypsum in which elution of contained fluorine is reduced using the same, and a method for treating fluorine-contaminated soil using the same.
- Patent Document 1 the conventional method of adding an aluminum compound such as sodium aluminate as in Patent Document 1 has a problem that it is difficult to handle due to the nature of the drug to be added, and the pH is a constant value as in Patent Document 2.
- the conventional method of adjusting by moving up and down with a width there are problems of many man-hours and complicated operations.
- Non-Patent Document 1 the conventional method of simply adding calcium hydrogen phosphate dihydrate is used. In order to achieve the desired effect, a correspondingly large amount of sodium hydrogenphosphate dihydrate is required, which is uneconomical.
- the problem to be solved by the present invention is that the amount of fluorine eluted from gypsum containing fluorine, soil contaminated with fluorine, etc., as a standard by more economically and efficiently by simple work.
- the present invention provides a fluorine insolubilizing agent that can be reduced to 0.8 mg / L or less, a gypsum in which the elution amount of fluorine contained therein is reduced, and a method for treating fluorine-contaminated soil using the same.
- the present invention for solving the above-mentioned problems is that calcium hydrogen phosphate dihydrate is active for 0.5 minutes or more in water at 40 ° C. or higher where the mass ratio of water / calcium hydrogen phosphate dihydrate is 25 or higher. It relates to a fluorine insolubilizing agent characterized by comprising. The present invention also relates to a gypsum with reduced elution of contained fluorine, characterized in that a fluorine insolubilizing agent is added to gypsum containing fluorine. The present invention further relates to a method for treating fluorine-contaminated soil, characterized by adding a fluorine insolubilizing agent to the fluorine-contaminated soil.
- the fluorine insolubilizing agent according to the present invention includes calcium hydrogen phosphate dihydrate (CaHPO 4 .2H 2 O) in water at a temperature of 40 ° C. or higher where the mass ratio of water / calcium hydrogen phosphate dihydrate is 25 or higher. And activated for 0.5 minutes or more. It is known that calcium hydrogen phosphate dihydrate insolubilizes fluorine as fluorapatite. In addition, when the particles of calcium hydrogen phosphate dihydrate are suspended in water, for example, when stirred in water, many nanometer (nm) scale fine crystals are uniformly deposited on the particle surface.
- CaHPO 4 .2H 2 O calcium hydrogen phosphate dihydrate
- calcium hydrogen phosphate dihydrate is activated by fluorination for at least 0.5 minutes in water at 40 ° C. or higher where the mass ratio of water / calcium hydrogen phosphate dihydrate is 25 or higher. Insolubilizer.
- the temperature has the greatest effect on the water / calcium hydrogen phosphate dihydrate mass ratio, temperature, and time when activating calcium hydrogen phosphate dihydrate in water.
- the calcium hydrogen phosphate dihydrate particles are activated in water at 40 ° C. or higher. From the viewpoint of actual operation and activation, the calcium hydrogen phosphate dihydrate particles are usually 40 It is activated in water at a temperature from 0 ° C. to the boiling point of water, preferably activated in water at 40 to 80 ° C., particularly preferably activated in water at 50 to 80 ° C.
- the temperature has the greatest effect on the activation of calcium hydrogen phosphate dihydrate in water, but the time and concentration also have a corresponding effect.
- the time for activating the calcium hydrogen phosphate dihydrate in water is 0.5 minutes or more, but from the actual operation surface and activation surface, it is 0.5 to 100 minutes. It is preferably 1 to 60 minutes.
- the mass ratio of water / calcium hydrogen phosphate dihydrate related to the concentration is set to 25 or more, but is preferably 25 to 500, and preferably 35 to 100 from the actual operation surface and activation surface. Is particularly preferred.
- the activation in water may be carried out by keeping the particles of calcium hydrogen phosphate dihydrate immersed in water, but it is preferable to stir intermittently or continuously.
- the calcium hydrogen phosphate dihydrate activated in water under a predetermined condition as described above is used as a fluorine insolubilizer.
- This is further dehydrated and dried as a fluorine insolubilizer. It is preferable to do this.
- the dehydration can be carried out by a known means such as decantation, filtration, centrifugation, etc. In addition to drying in the sun, ventilation drying using a heating source, vacuum drying, etc. As such, known means can be used. What was further dehydrated and dried after being activated in water under predetermined conditions has no substantial effect on the degree of activation and is convenient for handling such as storage, transportation and use.
- gypsum (hereinafter, also simply referred to as gypsum of the present invention) in which the elution of contained fluorine according to the present invention is reduced.
- the gypsum of the present invention is obtained by adding the fluorine insolubilizing agent according to the present invention as described above to gypsum containing fluorine.
- the kind of gypsum containing fluorine used here is not particularly limited as long as it contains fluorine. For example, this is a by-product when phosphoric acid is produced from phosphorus ore by a wet phosphoric acid method.
- Gypsum obtained as a by-product when flue gas is desulfurized by the lime method gypsum obtained from waste materials of gypsum board produced using these gypsum, and the like.
- the method itself of adding the fluorine insolubilizing agent according to the present invention to gypsum containing fluorine is not particularly limited. Both can be simply mixed as they are, but it is preferable to make both powdery before mixing. Moreover, before both mixing, during mixing or after mixing, it can be added as needed, and further, after mixing both, it can be appropriately cured as necessary.
- the amount of the fluorine insolubilizing agent according to the present invention to the gypsum containing fluorine is such that the fluorine insolubilizing agent according to the present invention is usually in a ratio of 0.5 parts by mass or more per 100 parts by mass of the gypsum containing fluorine. However, the ratio is preferably 0.5 to 5 parts by mass.
- the amount of the fluorine insolubilizing agent according to the present invention is set to a ratio of less than 0.5 parts by mass per 100 parts by mass of gypsum containing fluorine, the amount of fluorine eluted from the gypsum under such conditions Even if the amount of addition of the fluorine insolubilizing agent according to the present invention is more than 5 parts by mass, it is difficult to achieve a soil environment standard of 0.8 mg / L or less. Expression does not change that much.
- the gypsum of the present invention is a gypsum in which the elution amount of fluorine is reduced to 0.8 mg / L or less of the soil environment standard, and therefore can be disposed as landfill as it is, but gypsum board, plaster, soil solidifying material, etc. Can be reused as raw material.
- the treatment method of the present invention is a method of adding the fluorine insolubilizing agent according to the present invention as described above to fluorine-contaminated soil.
- the method itself of adding the fluorine insolubilizing agent according to the present invention to fluorine-contaminated soil is not particularly limited. Both can be mixed as they are, but before or during mixing, or after mixing, the water can be adjusted by dehydrating or adding water as needed. You can also. In the case of adjusting the water content, it is preferable that the water content after mixing both is 5 to 20% by mass.
- the amount of the fluorine insolubilizing agent according to the present invention added to the fluorine-contaminated soil is such that the fluorine insolubilizing agent according to the present invention is usually in a ratio of 1 part by mass or more per 100 parts by mass of the dried material of the fluorine-contaminated soil. It is preferable that the ratio be ⁇ 20 parts by mass.
- the amount of addition of the fluorine insolubilizing agent according to the present invention is less than 1 part by mass per 100 parts by mass of the dried substance of fluorine-contaminated soil, the amount of fluorine eluted from the fluorine-contaminated soil under such conditions It shows a tendency that it is practically difficult to make the soil environmental standard 0.8 mg / L or less, and conversely, even if the addition amount of the fluorine insolubilizing agent according to the present invention is an amount exceeding 5 parts by mass, the effect The expression of is not so different.
- the amount of fluorine eluted from gypsum containing fluorine, soil contaminated with fluorine, and the like, with a simple work, more economically and efficiently, is 0.8 mg / It can be reduced to L or less.
- the 5000-times scanning electron micrograph which illustrates the particle
- the scanning electron micrograph of 5000 times which illustrates the particle
- the graph which illustrates the influence which the temperature in the activation test for obtaining the fluorine insolubilizer which concerns on this invention has on the fluorine elution amount.
- the graph which illustrates the influence which the time in the activation test for obtaining the fluorine insolubilizer which concerns on this invention has on the amount of elution of fluorine.
- the graph which illustrates the influence which the mass ratio of water / calcium hydrogenphosphate dihydrate has in the fluorine elution amount in the activation test for obtaining the fluorine insolubilizer according to the present invention illustrates the influence which the addition mass part of the fluorine insolubilizing agent which concerns on this invention per 100 mass parts of gypsum has on the fluorine elution amount from gypsum in the gypsum of this invention.
- the graph which illustrates the influence which the addition mass part of the fluorine insolubilizing agent which concerns on this invention per 100 mass parts of dried material of fluorine-contaminated soil gives to the fluorine elution amount from fluorine-contaminated soil in the processing method of this invention.
- Test category 1 The particles of calcium hydrogen phosphate dihydrate were mixed at a temperature of 25 ° C., 30 ° C., 35 ° C., 40 ° C., 45 ° C., 50 ° C., 60 ° C. at which the mass ratio of water / calcium hydrogen phosphate dihydrate was 100. After stirring for 1 hour in water at 70 ° C. or 80 ° C., the particle surface is etched, dehydrated by filtration, and dried by ventilation with hot air at 60 ° C. Obtained.
- FIG. 1 is a 5000 ⁇ scanning electron micrograph illustrating the particle surface of calcium hydrogen phosphate dihydrate used as a raw material
- FIG. 2 is a diagram of calcium hydrogen phosphate dihydrate activated in water at 50 ° C. It is a 5000 times scanning electron micrograph which illustrates the particle
- gypsum containing fluorine As the gypsum containing fluorine, gypsum discharged as a by-product when flue gas desulfurization from a thermal power plant by the lime method is used, and 0.25 g of the fluorine insolubilizing agent in a total of 9 categories and 500 ml of pure water are added to 50 g of this gypsum.
- the supernatant is filtered through a 0.45 nm membrane filter to obtain an eluate, and the fluorine concentration in the eluate is determined by an ion-selective electrode. The fluorine elution amount was obtained (lower limit of quantification was 0.02 mg / L).
- the results are shown in Table 1 and FIG.
- the blank in Table 1 is the fluorine elution amount obtained for the above-mentioned gypsum not added with a fluorine insolubilizing agent.
- the amount of fluorine eluted is drastically reduced to 40 ° C. when the calcium hydrogen phosphate dihydrate particles are activated in water. It is almost unchanged.
- the temperature is usually from 40 ° C. to the boiling point of water, preferably from 40 to 80 ° C., particularly preferably from 50 to 80 ° C. .
- Test category 2 Calcium hydrogen phosphate dihydrate particles in water at a temperature of 60 ° C. where the mass ratio of water / calcium hydrogen phosphate dihydrate is 100, 0.25 minutes, 0.5 minutes, 1 minute, After 5 minutes, 5 minutes, 7.5 minutes, 10 minutes or 60 minutes of stirring, the particle surface is etched, dehydrated by filtration, and dried by ventilation with hot air at 60 ° C., for a total of 7 activated particles That is, a fluorine insolubilizing agent was obtained.
- gypsum containing fluorine As the gypsum containing fluorine, gypsum discharged as a by-product when flue gas desulphurization from a thermal power plant by the lime method is used, and 0.25 g of the fluorine insolubilizing agent and 500 ml of pure water described above are added to 50 g of this gypsum.
- the fluorine elution amount was determined in the same manner as in test category 1 below (lower limit of quantification was 0.02 mg / L). The results are shown in Table 2 and FIG.
- the blank in Table 2 is the amount of fluorine elution determined for the gypsum to which no fluorine insolubilizing agent is added. As is clear from Table 2 and FIG.
- this time is 0.5 minutes or more, preferably 0.5 to 100 minutes, particularly preferably 1 to 60 minutes.
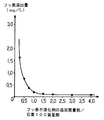
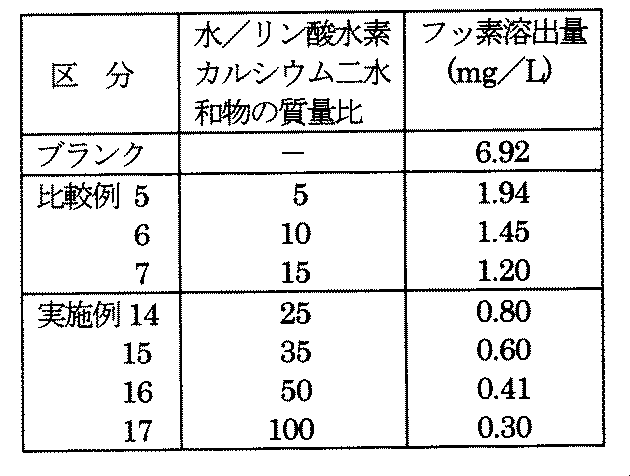
- Test category 3 Calcium hydrogen phosphate dihydrate particles are stirred in water at a temperature of 60 ° C. for which the mass ratio of water / calcium hydrogen phosphate dihydrate is 5, 10, 15, 25, 35, 50 or 100 for 1 hour. Then, after the particle surface was etched, it was dehydrated by filtration and dried by ventilation with warm air at 60 ° C. to obtain a total of 7 activated particles, that is, a fluorine insolubilizing agent.
- gypsum containing fluorine As the gypsum containing fluorine, gypsum discharged as a by-product when flue gas desulphurization from a thermal power plant by the lime method is used, and 0.25 g of the fluorine insolubilizing agent and 500 ml of pure water described above are added to 50 g of this gypsum.
- the fluorine elution amount was determined in the same manner as in test category 1 below (lower limit of quantification was 0.02 mg / L). The results are shown in Table 3 and FIG.
- the blank in Table 3 is the fluorine elution amount obtained for the above-mentioned gypsum not added with a fluorine insolubilizing agent. As apparent from Table 3 and FIG.
- the elution amount of fluorine is determined by the mass ratio of water / calcium hydrogen phosphate dihydrate when the calcium hydrogen phosphate dihydrate particles are activated in water. It decreases rapidly to 25, and hardly changes at 35 or higher.
- the mass ratio is 25 or more, preferably 25 to 500, particularly preferably 35 to 100, from the actual operation surface and activation surface.
- Test category 4 The particles of calcium hydrogen phosphate dihydrate were stirred for 1 hour in water at a temperature of 60 ° C. where the mass ratio of water / calcium hydrogen phosphate dihydrate was 100, and the particle surface was etched, followed by dehydration by filtration. And dried by ventilation with warm air of 60 ° C. to obtain a fluorine insolubilizing agent.
- gypsum containing fluorine As the gypsum containing fluorine, gypsum discharged as a by-product when flue gas is desulfurized from a thermal power plant by the lime method, 0.125 g, 0.25 g, and 0.375 g of the above-described fluorine insolubilizing agent are added to 50 g of this gypsum. , 0.5 g, 0.75 g, 1.0 g, 1.5 g or 2.0 g, and further 500 ml of pure water were added, and the fluorine elution amount was determined in the same manner as in Test Category 1 (the lower limit of quantification was 0.02 mg / L). The results are shown in Table 4 and FIG.
- the blank in Table 4 is the elution amount of fluorine determined for the above-mentioned gypsum not added with a fluorine insolubilizing agent.
- the amount of fluorine eluted from gypsum containing fluorine is abruptly lowered to 0.5 when the added mass part of the fluorine insolubilizing agent according to the present invention per 100 parts by mass of gypsum. It has become.
- the added mass part of the fluorine insolubilizing agent is usually 0.5 or more, preferably 0.5 to 5.
- Test category 5 The particles of calcium hydrogen phosphate dihydrate were stirred for 1 hour in water at a temperature of 60 ° C. where the mass ratio of water / calcium hydrogen phosphate dihydrate was 100, and the particle surface was etched, followed by dehydration by filtration. And dried by ventilation with warm air of 60 ° C. to obtain a fluorine insolubilizing agent.
- fluorine-contaminated soil soil contaminated with fluorine collected from a gypsum factory was used.
- the soil was vacuum dried at a temperature of 50 ° C. and sieved with a 10 mm sieve.
- the amount of fluorine eluted from the fluorine-contaminated soil is abruptly increased up to 1 by mass part of the fluorine insolubilizing agent according to the present invention per 100 parts by mass of the dry matter of the fluorine-contaminated soil. It is low.
- the addition mass part of the fluorine insolubilizing agent is usually 1 or more, preferably 1 to 20.
Landscapes
- Engineering & Computer Science (AREA)
- Environmental & Geological Engineering (AREA)
- Life Sciences & Earth Sciences (AREA)
- Soil Sciences (AREA)
- Toxicology (AREA)
- General Health & Medical Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Business, Economics & Management (AREA)
- Emergency Management (AREA)
- Processing Of Solid Wastes (AREA)
- Soil Conditioners And Soil-Stabilizing Materials (AREA)
- Compounds Of Alkaline-Earth Elements, Aluminum Or Rare-Earth Metals (AREA)
Abstract
Description
リン酸水素カルシウム二水和物の粒子を、水/リン酸水素カルシウム二水和物の質量比が100となる温度25℃、30℃、35℃、40℃、45℃、50℃、60℃、70℃又は80℃の水中で1時間撹拌して粒子表面をエッチングした後、濾過により脱水し、60℃の温風により通風乾燥して、合計9区分の活性化した粒子すなわちフッ素不溶化剤を得た。図1は原料として用いたリン酸水素カルシウム二水和物の粒子表面を例示する5000倍の走査型電子顕微鏡写真、図2は50℃の水中で活性化したリン酸水素カルシウム二水和物の粒子表面を例示する5000倍の走査型電子顕微鏡写真である。前者の粒子表面が滑らかであるのに対し、後者の粒子表面は複雑にエッチングされている。
リン酸水素カルシウム二水和物の粒子を、水/リン酸水素カルシウム二水和物の質量比が100となる温度60℃の水中で0.25分、0.5分、1分、2.5分、5分、7.5分、10分又は60分撹拌して粒子表面をエッチングした後、濾過により脱水し、60℃の温風により通風乾燥して、合計7区分の活性化した粒子すなわちフッ素不溶化剤を得た。
リン酸水素カルシウム二水和物の粒子を、水/リン酸水素カルシウム二水和物の質量比が5、10、15、25、35、50又は100となる温度60℃の水中で1時間撹拌して粒子表面をエッチングした後、濾過により脱水し、60℃の温風により通風乾燥して、合計7区分の活性化した粒子すなわちフッ素不溶化剤を得た。
リン酸水素カルシウム二水和物の粒子を、水/リン酸水素カルシウム二水和物の質量比が100となる温度60℃の水中で1時間撹拌して粒子表面をエッチングした後、濾過により脱水し、60℃の温風により通風乾燥して、フッ素不溶化剤を得た。
リン酸水素カルシウム二水和物の粒子を、水/リン酸水素カルシウム二水和物の質量比が100となる温度60℃の水中で1時間撹拌して粒子表面をエッチングした後、濾過により脱水し、60℃の温風により通風乾燥して、フッ素不溶化剤を得た。
Claims (7)
- リン酸水素カルシウム二水和物を、水/リン酸水素カルシウム二水和物の質量比が25以上となる40℃以上の水中で0.5分以上活性化して成ることを特徴とするフッ素不溶化剤。
- リン酸水素カルシウム二水和物を、水/リン酸水素カルシウム二水和物の質量比が35~100となる50~80℃の水中で1~60分活性化した請求項1記載のフッ素不溶化剤。
- 水中で活性化したものを、更に脱水し、乾燥した請求項1又は2記載のフッ素不溶化剤。
- フッ素を含有する石膏に請求項1~3のいずれか一つの項記載のフッ素不溶化剤を添加して成ることを特徴とする含有フッ素の溶出を低減させた石膏。
- 石膏100質量部当たりフッ素不溶化剤を0.5~5質量部の割合で添加した請求項4記載の含有フッ素の溶出を低減させた石膏。
- フッ素汚染土壌に請求項1~3のいずれか一つの項記載のフッ素不溶化剤を添加することを特徴とするフッ素汚染土壌の処理方法。
- フッ素汚染土壌の乾燥物100質量部当たりフッ素不溶化剤を1~20質量部の割合で添加する請求項6記載のフッ素汚染土壌の処理方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2010532747A JP5582356B2 (ja) | 2008-10-10 | 2008-10-10 | フッ素不溶化剤、含有フッ素の溶出を低減させた石膏及びフッ素汚染土壌の処理方法 |
| EP08877278.5A EP2347835A4 (en) | 2008-10-10 | 2008-10-10 | FLUOR UNSUITABLE MEANS, GIPS WITH THE REDUCED FLUORELUTION CONTAINING THEREIN, AND METHOD FOR THE TREATMENT OF FLUOROMED SUBSTANCE |
| KR1020117010070A KR101481635B1 (ko) | 2008-10-10 | 2008-10-10 | 불소 불용화제, 함유 불소의 용출을 저감시킨 석고 및 불소 오염 토양의 처리방법 |
| CN2008801314668A CN102176983A (zh) | 2008-10-10 | 2008-10-10 | 氟不溶化剂、所含氟的溶出减少的石膏和氟污染土壤的处理方法 |
| PCT/JP2008/068441 WO2010041330A1 (ja) | 2008-10-10 | 2008-10-10 | フッ素不溶化剤、含有フッ素の溶出を低減させた石膏及びフッ素汚染土壌の処理方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP2008/068441 WO2010041330A1 (ja) | 2008-10-10 | 2008-10-10 | フッ素不溶化剤、含有フッ素の溶出を低減させた石膏及びフッ素汚染土壌の処理方法 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010041330A1 true WO2010041330A1 (ja) | 2010-04-15 |
Family
ID=42100295
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2008/068441 Ceased WO2010041330A1 (ja) | 2008-10-10 | 2008-10-10 | フッ素不溶化剤、含有フッ素の溶出を低減させた石膏及びフッ素汚染土壌の処理方法 |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP2347835A4 (ja) |
| JP (1) | JP5582356B2 (ja) |
| KR (1) | KR101481635B1 (ja) |
| CN (1) | CN102176983A (ja) |
| WO (1) | WO2010041330A1 (ja) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2386331A1 (en) * | 2010-05-10 | 2011-11-16 | Institute of National Colleges of Technology, Japan | Fluorine insolubilizers and methods of producing same |
| JP2016150317A (ja) * | 2015-02-18 | 2016-08-22 | 石坂産業株式会社 | 土砂系混合廃棄物から抽出された精選土の再資源化処理方法及び再資源化処理材 |
| JP2017095287A (ja) * | 2015-11-18 | 2017-06-01 | 株式会社トクヤマ | フッ素溶出量を低減した二水石膏の製造方法 |
| JP2020132746A (ja) * | 2019-02-19 | 2020-08-31 | 独立行政法人国立高等専門学校機構 | フッ素不溶化剤、その製造方法、処理石膏、フッ素含有汚染土壌及び汚染水の処理方法 |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6113957B2 (ja) * | 2012-01-13 | 2017-04-12 | 新日鉄住金エンジニアリング株式会社 | スラグ混じり土の処理方法 |
| CN103242110B (zh) * | 2013-05-23 | 2015-03-04 | 四川师范大学 | 降低土壤水溶态氟含量的方法 |
| CN103736716B (zh) * | 2013-12-20 | 2015-11-18 | 中国科学院南京土壤研究所 | 氟化物类复合污染场地土壤的化学-植物联合修复方法 |
| CN105152619A (zh) * | 2015-09-30 | 2015-12-16 | 湖南创元铝业有限公司 | 含氟危废的处理方法及其处理药剂 |
| CN105419804B (zh) * | 2015-10-30 | 2018-09-25 | 深圳市铁汉生态环境股份有限公司 | 一种氟污染土壤的修复剂及一种氟污染土壤的修复方法 |
| KR101937106B1 (ko) | 2018-09-10 | 2019-04-09 | (주)에스지알테크 | 입도별 순차반응을 이용한 불소 오염토양 정화방법 및 정화시스템 |
| CN109731901B (zh) * | 2019-01-14 | 2021-10-19 | 北京泷涛环境修复有限公司 | 一种氟化物污染土壤的稳定化修复药剂及修复方法 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07118005A (ja) * | 1993-09-17 | 1995-05-09 | Fuji Chem Ind Co Ltd | リン酸水素カルシウム及びその製法並びにそれを用い た賦形剤 |
| JP2001253755A (ja) | 2000-03-10 | 2001-09-18 | Natl Inst Of Advanced Industrial Science & Technology Meti | フッ素不溶出性石膏組成物とその製造方法 |
| JP2003206133A (ja) | 2002-01-15 | 2003-07-22 | Shimonoseki Mitsui Chemicals Inc | 溶出するフッ素を低下させた石膏 |
| JP2007106622A (ja) * | 2005-10-12 | 2007-04-26 | Kawasaki Plant Systems Ltd | 石こう硬化体の安定化処理方法および安定化処理装置 |
| JP2007216156A (ja) | 2006-02-17 | 2007-08-30 | Institute Of National Colleges Of Technology Japan | フッ素汚染土壌の処理剤及び処理方法 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1162322C (zh) * | 2000-05-16 | 2004-08-18 | 单光渝 | 脱氟磷酸盐的制造和含氟尾气的净化方法 |
-
2008
- 2008-10-10 CN CN2008801314668A patent/CN102176983A/zh active Pending
- 2008-10-10 JP JP2010532747A patent/JP5582356B2/ja active Active
- 2008-10-10 EP EP08877278.5A patent/EP2347835A4/en not_active Withdrawn
- 2008-10-10 KR KR1020117010070A patent/KR101481635B1/ko active Active
- 2008-10-10 WO PCT/JP2008/068441 patent/WO2010041330A1/ja not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07118005A (ja) * | 1993-09-17 | 1995-05-09 | Fuji Chem Ind Co Ltd | リン酸水素カルシウム及びその製法並びにそれを用い た賦形剤 |
| JP2001253755A (ja) | 2000-03-10 | 2001-09-18 | Natl Inst Of Advanced Industrial Science & Technology Meti | フッ素不溶出性石膏組成物とその製造方法 |
| JP2003206133A (ja) | 2002-01-15 | 2003-07-22 | Shimonoseki Mitsui Chemicals Inc | 溶出するフッ素を低下させた石膏 |
| JP2007106622A (ja) * | 2005-10-12 | 2007-04-26 | Kawasaki Plant Systems Ltd | 石こう硬化体の安定化処理方法および安定化処理装置 |
| JP2007216156A (ja) | 2006-02-17 | 2007-08-30 | Institute Of National Colleges Of Technology Japan | フッ素汚染土壌の処理剤及び処理方法 |
Non-Patent Citations (2)
| Title |
|---|
| JOURNAL OF THE EUROPEAN CERAMIC SOCIETY, vol. 26, 2006, pages 767 - 770 |
| See also references of EP2347835A4 * |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2386331A1 (en) * | 2010-05-10 | 2011-11-16 | Institute of National Colleges of Technology, Japan | Fluorine insolubilizers and methods of producing same |
| JP2011256356A (ja) * | 2010-05-10 | 2011-12-22 | Institute Of National Colleges Of Technology Japan | フッ素不溶化剤及びその製造方法 |
| KR101753219B1 (ko) | 2010-05-10 | 2017-07-04 | 지요다 우테 가부시키가이샤 | 불소 불용화제 및 그의 제조방법 |
| JP2016150317A (ja) * | 2015-02-18 | 2016-08-22 | 石坂産業株式会社 | 土砂系混合廃棄物から抽出された精選土の再資源化処理方法及び再資源化処理材 |
| JP2017095287A (ja) * | 2015-11-18 | 2017-06-01 | 株式会社トクヤマ | フッ素溶出量を低減した二水石膏の製造方法 |
| JP2020132746A (ja) * | 2019-02-19 | 2020-08-31 | 独立行政法人国立高等専門学校機構 | フッ素不溶化剤、その製造方法、処理石膏、フッ素含有汚染土壌及び汚染水の処理方法 |
| JP7315160B2 (ja) | 2019-02-19 | 2023-07-26 | 独立行政法人国立高等専門学校機構 | フッ素不溶化剤、その製造方法、処理石膏、フッ素含有汚染土壌及び汚染水の処理方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102176983A (zh) | 2011-09-07 |
| JPWO2010041330A1 (ja) | 2012-03-01 |
| KR101481635B1 (ko) | 2015-01-12 |
| EP2347835A4 (en) | 2013-08-07 |
| JP5582356B2 (ja) | 2014-09-03 |
| EP2347835A1 (en) | 2011-07-27 |
| KR20110069835A (ko) | 2011-06-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5582356B2 (ja) | フッ素不溶化剤、含有フッ素の溶出を低減させた石膏及びフッ素汚染土壌の処理方法 | |
| JP6438888B2 (ja) | 特定有害物質の不溶化材及びこれを用いた特定有害物質の不溶化方法 | |
| JP2014057949A (ja) | 公害型廃石膏類の処理法ならびに無公害型石膏組成物 | |
| CN103740372A (zh) | 一种修复砷污染土壤固定剂及其制备和应用方法 | |
| JP4434156B2 (ja) | フッ素汚染土壌の処理方法 | |
| JP4209223B2 (ja) | 六価クロムの溶出抑制方法 | |
| JP2002241183A (ja) | 多孔性セラミック粒子、およびその製造方法 | |
| CN104245603B (zh) | 特定有害物质的不溶化材料、使用其的特定有害物质的不溶化方法及土壤的改良方法 | |
| JP4752038B2 (ja) | 含有フッ素の溶出を低減させる石膏の処理方法 | |
| JP2004305833A (ja) | 廃棄物の安定化処理方法 | |
| JP3382202B2 (ja) | フッ素不溶出性石膏組成物とその製造方法 | |
| JP2009028639A (ja) | 汚泥の処理方法 | |
| JP4753182B2 (ja) | フッ素含有排水の処理方法 | |
| JP2016155719A (ja) | 多孔質石膏硬化体の製造方法及び水質浄化材 | |
| JP3786899B2 (ja) | 泥土改質固化安定剤 | |
| JP4014400B2 (ja) | 土壌処理材組成物及びその製造方法 | |
| JP2008029937A (ja) | 副生物の処理方法 | |
| JP2001335778A (ja) | 土質改良剤 | |
| JP2007106622A (ja) | 石こう硬化体の安定化処理方法および安定化処理装置 | |
| JP6227879B2 (ja) | 特定有害物質の不溶化材及びこれを用いた特定有害物質の不溶化方法 | |
| JP2016150317A (ja) | 土砂系混合廃棄物から抽出された精選土の再資源化処理方法及び再資源化処理材 | |
| JP3559905B2 (ja) | 環境浄化型無機質材 | |
| JPH0515731A (ja) | 珪酸石灰系多孔質硬化体による脱臭処理方法 | |
| JPH0669477B2 (ja) | 脱臭剤 | |
| JP4698880B2 (ja) | 汚泥の処理方法および処理システム |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200880131466.8 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 08877278 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2010532747 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20117010070 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008877278 Country of ref document: EP |