WO1995012592A1 - Neue pyrazincarboxamidderivate, ihre herstellung und ihre verwendung in arzneimitteln - Google Patents
Neue pyrazincarboxamidderivate, ihre herstellung und ihre verwendung in arzneimitteln Download PDFInfo
- Publication number
- WO1995012592A1 WO1995012592A1 PCT/EP1994/003580 EP9403580W WO9512592A1 WO 1995012592 A1 WO1995012592 A1 WO 1995012592A1 EP 9403580 W EP9403580 W EP 9403580W WO 9512592 A1 WO9512592 A1 WO 9512592A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- phenyl
- alkyl
- hydrogen
- compounds
- Prior art date
Links
- 0 C*C1(C)CCN(C)CCC1 Chemical compound C*C1(C)CCN(C)CCC1 0.000 description 5
- SCCRHBSDEKGLDA-UHFFFAOYSA-N CN1C(CCCC2)C2N(C)C1 Chemical compound CN1C(CCCC2)C2N(C)C1 SCCRHBSDEKGLDA-UHFFFAOYSA-N 0.000 description 1
- LGUDBXQVURAVTF-UHFFFAOYSA-N CN1C(CCCC2)C2N(C)CC1 Chemical compound CN1C(CCCC2)C2N(C)CC1 LGUDBXQVURAVTF-UHFFFAOYSA-N 0.000 description 1
- UCEQMZLXVLXLOV-UHFFFAOYSA-N CN1CN(C)C(CCCC2)C2C1 Chemical compound CN1CN(C)C(CCCC2)C2C1 UCEQMZLXVLXLOV-UHFFFAOYSA-N 0.000 description 1
- DRLIPNKQPYZWNG-UHFFFAOYSA-N CN1N(C)C(CCCC2)C2CC1 Chemical compound CN1N(C)C(CCCC2)C2CC1 DRLIPNKQPYZWNG-UHFFFAOYSA-N 0.000 description 1
- OWKIFNQHMYDNTN-UHFFFAOYSA-N CN1N(C)CC(CCCC2)C2C1 Chemical compound CN1N(C)CC(CCCC2)C2C1 OWKIFNQHMYDNTN-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/10—Expectorants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/10—Antioedematous agents; Diuretics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/08—Vasodilators for multiple indications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
Definitions
- the invention relates to new pyrazine carboxamide derivatives, their production by conventional methods and their use in or in the production of medicaments.
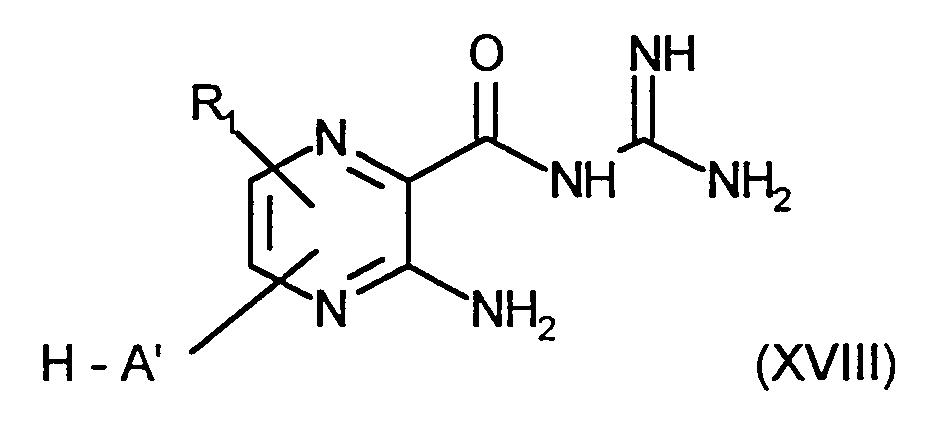
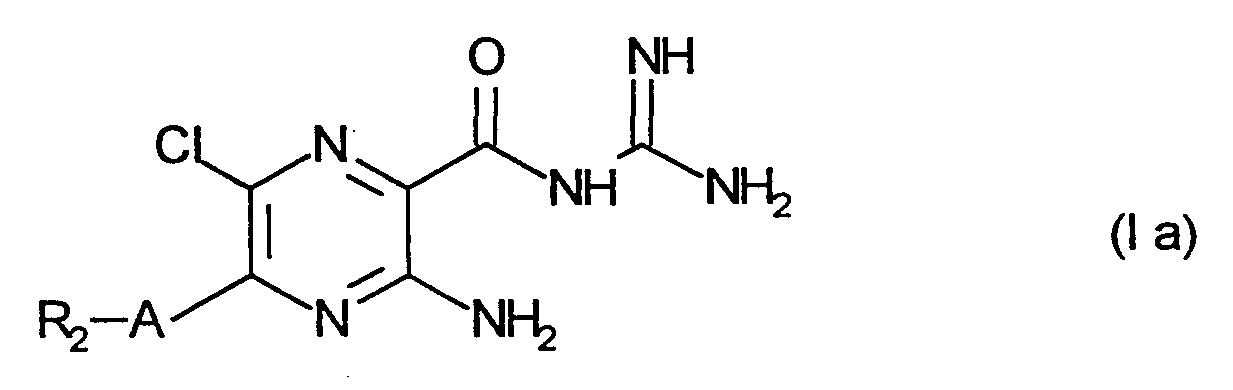
- R 1 for hydrogen, fluorine, chlorine, C 1 -C 4 alkyl, C 1 -C 4 alkoxy, trifluoromethyl
- R 2 for a radical of the formula
- R 3 , R 4 and R 4 ' which may be the same or different, are hydrogen, C 1 -C 4 alkyl, R 4 and R 4 ' are also phenyl, benzyl and C 3 -C 7 cycloalkyl,
- R 5 and R 6 which may be the same or different, are hydrogen, methyl, methoxy, hydroxy or halogen,
- R 7 is hydrogen, C 1 -C 4 alkyl, benzyl or benzyloxy,
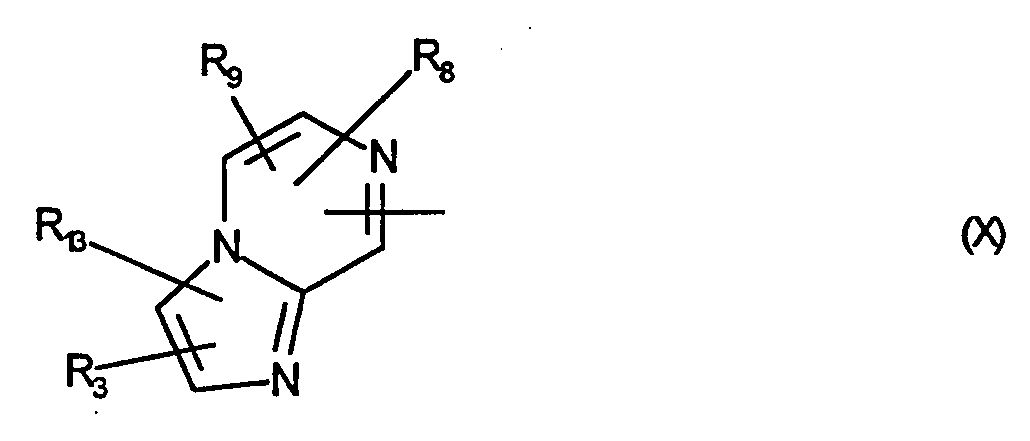
- R 8 and R 9 which may be the same or different, are hydrogen, C 1 -C 4 alkyl, phenyl or halogen,
- R 10 is hydrogen or C 1 -C 6 -alkyl, which can also be substituted by phenyl or methyl, methoxy or halogen-substituted phenyl,
- R 1 1 and R 12 which may be the same or different, are hydrogen, methyl,
- R 1 1 also a fused benzene ring which can have up to three substituents from the group consisting of methyl, methoxy, halogen, CF 3 and CN,
- R 13 is hydrogen, C 1 -C 4 alkyl, which can also be substituted by phenyl, phenoxy or benzyloxy, or halogen, and
- E and G which can be the same or different, N or CH, m 2, 3, 4, 5 or 6,
- p 2, 3 or 4.
- Alkylene groups can be straight-chain or branched. “Lower” groups are those with 1 to 4, in particular 1 to 3, especially 1 to 2, carbon atoms.
- the halogens are fluorine, chlorine and bromine, especially fluorine and chlorine prefers.
- Preferred unsaturated hydrocarbon radicals are allyl and propargyl.
- the index m is preferably 2, 3 or 4, p is 2 or 3.
- the groups R 2 -A- typically have structures like the following:
- reaction is carried out in a polar, water-free solvent or solvent mixture, in particular dimethyl sulfoxide, dimethylformamide, preferably in the presence of a base, for example triethylamine, N-methylpiperidine, pyridine, under hot conditions.
- a base for example triethylamine, N-methylpiperidine, pyridine
- N atom is bound to R 2 , as in IT, IM 'and IV)
- R lower alkyl group, benzyl
- the reaction is not limited to the esters with R in the meanings mentioned, but the person skilled in the art will expediently use an ester which is easy to prepare, for example the methyl or ethyl ester, or an ester whose reaction produces an unproblematic alcohol.
- the alcohol which is also contained in the ester group is preferably used, for example by a methyl ester of formula XX in methanol
- the compounds can be present in stereoisomeric forms, appropriate starting products are used or mixtures formed during the preparation are optionally separated into the components. Unless they are already known, the starting materials can also be obtained by conventional methods. If, for example, the corresponding esters are used instead of the N-amidino-carboxamides of the formula XVI or the formula XVII, the starting materials of the formula XX are obtained.
- the compounds of the formula I can be used as active ingredients in pharmaceuticals or can be used as intermediates for the preparation of such active ingredients.
- the new compounds inhibit Na + / H + and Na + / Li + exchange.
- the active compounds according to the invention can be used as
- Antihypertensives, mucolytics, diuretics and cancerostats are used; they can also be used for diseases related to ischemia (examples: cardiac, cerebral, gastrointestinal, pulmonary, renal ischemia, ischemia of the liver, ischemia of the skeletal muscles).
- diseases related to ischemia include cardiac, cerebral, gastrointestinal, pulmonary, renal ischemia, ischemia of the liver, ischemia of the skeletal muscles.
- Corresponding diseases are, for example, coronary heart disease, angina pectoris, embolism in the
- Renal insufficiency e.g. after blood flow to the brain areas after vascular occlusions have been released, also in combination with t-PA,
- Streptokinases, urokinase, etc. acute and chronic circulatory disorders in the brain.
- ischemic heart is reperfused (e.g. after an attack of angina or a heart attack)
- irreversible damage can occur
- Cardiomyocytes occur in the affected region.
- the compounds according to the invention can be used, inter alia, for cardioprotection in such a case.
- the new compounds are notable for minor side effects, with the substantial absence of an ⁇ 1 and / or 0.2 effect
- the ischemia field of application also includes the prevention of damage to grafts (e.g. as protection of the graft before, during and after implantation), which can occur in connection with transplants.
- Customary formulations such as tablets, dragees, capsules, granules, injection solutions, and possibly also nasally applicable, are suitable for use of the active ingredients Preparations, the amount of the active substance in a single dose generally being 1 to 200 mg, preferably 20-100 mg.
- the content of one capsule consists of 50.0 mg of a compound according to the invention and 150.0 mg of corn starch.
- the purified substance is dissolved in ethanol, with ethereal
- R 2 -A is a radical of formula IV
- R 2 -A is a radical of the formula V ', R 3 is H and
- R 2 -A is a radical of the formula VI '
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Vascular Medicine (AREA)
- Pulmonology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
Abstract
Description
Claims
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| NZ274848A NZ274848A (en) | 1993-11-04 | 1994-10-31 | 2-n-amidinocarbamoyl-3-aminopiperazine derivatives substituted by a phenyl substituted piperazinyl moiety |
| CA002175837A CA2175837A1 (en) | 1993-11-04 | 1994-10-31 | Novel pyrazine carboxamide derivatives, their production and their use in medicaments |
| DE59409094T DE59409094D1 (de) | 1993-11-04 | 1994-10-31 | Neue pyrazincarboxamidderivate, ihre herstellung und ihre verwendung in arzneimitteln |
| AU79936/94A AU690588B2 (en) | 1993-11-04 | 1994-10-31 | Novel pyrazine carboxamide derivatives, their production and their use in medicaments |
| DK94931018T DK0726899T3 (da) | 1993-11-04 | 1994-10-31 | Nye pyrazincarboxamidderivater, deres fremstilling og deres anvendelse i lægemidler |
| PL94314187A PL314187A1 (en) | 1993-11-04 | 1994-10-31 | Novel derivatives of pyrazine carboxamide, method of obtaining them and their application in pharmaceutical composition |
| EP94931018A EP0726899B1 (de) | 1993-11-04 | 1994-10-31 | Neue pyrazincarboxamidderivate, ihre herstellung und ihre verwendung in arzneimitteln |
| JP7513010A JPH09505035A (ja) | 1993-11-04 | 1994-10-31 | 新規ピラジンカルボキシアミド誘導体、その調製及び医薬組成物における使用 |
| AT94931018T ATE188965T1 (de) | 1993-11-04 | 1994-10-31 | Neue pyrazincarboxamidderivate, ihre herstellung und ihre verwendung in arzneimitteln |
| KR1019960702301A KR960705815A (ko) | 1993-11-04 | 1996-05-03 | 신규한 피라진 카복스아미드 유도체, 이의 제조방법 및 약제에 있어서의 이의 용도(Novel pyrazine carboxamide derivatives, their production and their use in medicaments) |
| GR20000400720T GR3033034T3 (en) | 1993-11-04 | 2000-03-22 | Novel pyrazine carboxamide derivatives, their production and their use in medicaments |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE4337609A DE4337609A1 (de) | 1993-11-04 | 1993-11-04 | Neue Pyrazincarboxamidderivate, ihre Herstellung und ihre Verwendung in Arzneimitteln |
| DEP4337609.6 | 1993-11-04 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1995012592A1 true WO1995012592A1 (de) | 1995-05-11 |
Family
ID=6501757
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP1994/003580 WO1995012592A1 (de) | 1993-11-04 | 1994-10-31 | Neue pyrazincarboxamidderivate, ihre herstellung und ihre verwendung in arzneimitteln |
Country Status (17)
| Country | Link |
|---|---|
| EP (1) | EP0726899B1 (de) |
| JP (1) | JPH09505035A (de) |
| KR (1) | KR960705815A (de) |
| CN (1) | CN1134151A (de) |
| AT (1) | ATE188965T1 (de) |
| AU (1) | AU690588B2 (de) |
| CA (1) | CA2175837A1 (de) |
| CO (1) | CO4290432A1 (de) |
| DE (2) | DE4337609A1 (de) |
| DK (1) | DK0726899T3 (de) |
| ES (1) | ES2140565T3 (de) |
| GR (1) | GR3033034T3 (de) |
| NZ (1) | NZ274848A (de) |
| PL (1) | PL314187A1 (de) |
| PT (1) | PT726899E (de) |
| WO (1) | WO1995012592A1 (de) |
| ZA (1) | ZA948669B (de) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6156758A (en) * | 1999-09-08 | 2000-12-05 | Isis Pharmaceuticals, Inc. | Antibacterial quinazoline compounds |
| US7244738B2 (en) | 2003-07-02 | 2007-07-17 | Roche Palo Alto Llc | Arylamine-substituted quinazolinone compounds useful as alpha 1A/B adrenergic receptor antagonists |
| US7652135B2 (en) | 2003-09-23 | 2010-01-26 | Vertex Pharmaceuticals Incorporated | Compositions useful as inhibitors of protein kinases |
| US11384082B2 (en) | 2017-08-25 | 2022-07-12 | Kronos Bio, Inc. | Hydrates of polymorphs of 6-(1H-indazol-6-YL)-N-(4-morpholinophenyl)-2,3-dihydroimidazo[1,2-A]pyrazin-8-amine bisemsylate as Syk inhibitors |
Families Citing this family (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19548812A1 (de) * | 1995-12-27 | 1997-07-03 | Hoechst Ag | Verwendung von Inhibitoren des zellulären Na·+·/H·+·-Exchangers (NHE) zur Herstellung eines Medikaments zur Atemstimulation |
| DE19738604A1 (de) * | 1997-09-04 | 1999-03-11 | Hoechst Marion Roussel De Gmbh | Die Verwendung von Hemmern des Natrium-Wasserstoff-Austauschers zur Herstellung eines Medikaments zur Verminderung der Giftigkeit cardiotoxischer Stoffe |
| EP1182194A4 (de) | 1999-06-03 | 2004-02-11 | Takeda Chemical Industries Ltd | Pernasale zusammensetzungen |
| EP1509526A2 (de) | 2002-04-19 | 2005-03-02 | Cellular Genomics Inc. | Imidazo(1,2-a)pyrazin-8-ylamine, verfahren zu ihrer herstellung und methoden zu ihrer verwendung |
| US7312341B2 (en) | 2002-09-09 | 2007-12-25 | Cgi Pharmaceuticals, Inc. | 6-aryl-imidazo[1,2-a] pyrazin-8-ylamines, method of making, and method of use thereof |
| WO2004072080A1 (en) | 2003-02-10 | 2004-08-26 | Cellular Genomics, Inc. | Certain 8-heteroaryl-6-phenyl-imidazo[1,2-a]pyrazines as modulators of hsp90 complex activity |
| US7405295B2 (en) | 2003-06-04 | 2008-07-29 | Cgi Pharmaceuticals, Inc. | Certain imidazo[1,2-a]pyrazin-8-ylamines and method of inhibition of Bruton's tyrosine kinase by such compounds |
| US7393848B2 (en) | 2003-06-30 | 2008-07-01 | Cgi Pharmaceuticals, Inc. | Certain heterocyclic substituted imidazo[1,2-A]pyrazin-8-ylamines and methods of inhibition of Bruton's tyrosine kinase by such compounds |
| US7259164B2 (en) | 2003-08-11 | 2007-08-21 | Cgi Pharmaceuticals, Inc. | Certain substituted imidazo[1,2-a]pyrazines, as modulators of kinase activity |
| US7777040B2 (en) | 2005-05-03 | 2010-08-17 | Cgi Pharmaceuticals, Inc. | Certain substituted ureas, as modulators of kinase activity |
| JP5496915B2 (ja) | 2008-02-13 | 2014-05-21 | シージーアイ ファーマシューティカルズ,インコーポレーテッド | 6−アリール−イミダゾ[1,2−a]ピラジン誘導体、その製造方法、及びその使用方法 |
| US8450321B2 (en) | 2008-12-08 | 2013-05-28 | Gilead Connecticut, Inc. | 6-(1H-indazol-6-yl)-N-[4-(morpholin-4-yl)phenyl]imidazo-[1,2-A]pyrazin-8-amine, or a pharmaceutically acceptable salt thereof, as a SYK inhibitor |
| CN102307581B (zh) | 2008-12-08 | 2016-08-17 | 吉利德康涅狄格股份有限公司 | 咪唑并哌嗪syk抑制剂 |
| SI2716157T1 (sl) | 2008-12-08 | 2016-10-28 | Gilead Connecticut, Inc., c/o Gilead Sciences, Inc. | Imidazopirazinski Syk-inhibitorji |
| AU2011226689B2 (en) | 2010-03-11 | 2016-09-01 | Kronos Bio, Inc. | Imidazopyridines Syk inhibitors |
| PT2616460E (pt) * | 2010-09-13 | 2015-11-24 | Otsuka Pharma Co Ltd | Compostos heterocíclicos para tratar ou prevenir distúrbios provocados por neurotransmissão reduzida de serotonina, norepinefrina ou dopamina |
| AP2016009007A0 (en) | 2013-07-30 | 2016-01-31 | Gilead Connecticut Inc | Formulation of syk inhibitors |
| HUE052090T2 (hu) | 2013-07-30 | 2021-04-28 | Kronos Bio Inc | A SYK inhibitorok polimorfja |
| CN103467392B (zh) * | 2013-10-09 | 2016-08-17 | 重庆工商大学 | 一种多卤代吡嗪甲酰胺衍生物及其盐类、制备方法和用途 |
| PT3076976T (pt) | 2013-12-04 | 2020-12-07 | Kronos Bio Inc | Métodos para tratar cancros |
| UY35898A (es) | 2013-12-23 | 2015-07-31 | Gilead Sciences Inc | ?compuestos inhibidores de syk y composiciones que los comprenden?. |
| US9290505B2 (en) | 2013-12-23 | 2016-03-22 | Gilead Sciences, Inc. | Substituted imidazo[1,2-a]pyrazines as Syk inhibitors |
| SG11201610551TA (en) | 2014-07-14 | 2017-01-27 | Gilead Sciences Inc | Combinations for treating cancers |
| CA3130848A1 (en) | 2019-02-22 | 2020-08-27 | Kronos Bio, Inc. | Solid forms of condensed pyrazines as syk inhibitors |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3313813A (en) * | 1962-10-30 | 1967-04-11 | Merck & Co Inc | (3-amino-5, 6-disubstituted-pyrazinoyl) guanidines |
| WO1993004048A1 (de) * | 1991-08-16 | 1993-03-04 | Boehringer Ingelheim Kg | Neue pyrazinderivative, ihre herstellung und verwendung |
-
1993
- 1993-11-04 DE DE4337609A patent/DE4337609A1/de not_active Withdrawn
-
1994
- 1994-10-31 PT PT94931018T patent/PT726899E/pt unknown
- 1994-10-31 PL PL94314187A patent/PL314187A1/xx unknown
- 1994-10-31 CN CN94194016A patent/CN1134151A/zh active Pending
- 1994-10-31 DE DE59409094T patent/DE59409094D1/de not_active Expired - Fee Related
- 1994-10-31 NZ NZ274848A patent/NZ274848A/en unknown
- 1994-10-31 CA CA002175837A patent/CA2175837A1/en not_active Abandoned
- 1994-10-31 AT AT94931018T patent/ATE188965T1/de not_active IP Right Cessation
- 1994-10-31 AU AU79936/94A patent/AU690588B2/en not_active Ceased
- 1994-10-31 ES ES94931018T patent/ES2140565T3/es not_active Expired - Lifetime
- 1994-10-31 EP EP94931018A patent/EP0726899B1/de not_active Expired - Lifetime
- 1994-10-31 DK DK94931018T patent/DK0726899T3/da active
- 1994-10-31 WO PCT/EP1994/003580 patent/WO1995012592A1/de active IP Right Grant
- 1994-10-31 JP JP7513010A patent/JPH09505035A/ja active Pending
- 1994-11-02 CO CO94050173A patent/CO4290432A1/es unknown
- 1994-11-03 ZA ZA948669A patent/ZA948669B/xx unknown
-
1996
- 1996-05-03 KR KR1019960702301A patent/KR960705815A/ko not_active Application Discontinuation
-
2000
- 2000-03-22 GR GR20000400720T patent/GR3033034T3/el not_active IP Right Cessation
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3313813A (en) * | 1962-10-30 | 1967-04-11 | Merck & Co Inc | (3-amino-5, 6-disubstituted-pyrazinoyl) guanidines |
| WO1993004048A1 (de) * | 1991-08-16 | 1993-03-04 | Boehringer Ingelheim Kg | Neue pyrazinderivative, ihre herstellung und verwendung |
Non-Patent Citations (2)
| Title |
|---|
| G.KACZOROWSKI: "Inhibition of Na+/Ca2+ exchange in pituitary plasma membrane vesicles by analogues of Amiloride", BIOCHEMISTRY, vol. 24, no. 6, 1985, pages 1394 - 1403 * |
| L.SIMCHOWITZ ET AL: "Inhibition of Chemotactic Factor-activated Na+/H+ exchange in Human Neutrophils by analogues of Amiloride", MOLECULAR PHARMACOLOGY, vol. 30, no. 2, 1986, pages 112 - 120 * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6156758A (en) * | 1999-09-08 | 2000-12-05 | Isis Pharmaceuticals, Inc. | Antibacterial quinazoline compounds |
| US7244738B2 (en) | 2003-07-02 | 2007-07-17 | Roche Palo Alto Llc | Arylamine-substituted quinazolinone compounds useful as alpha 1A/B adrenergic receptor antagonists |
| US7652135B2 (en) | 2003-09-23 | 2010-01-26 | Vertex Pharmaceuticals Incorporated | Compositions useful as inhibitors of protein kinases |
| US8642779B2 (en) | 2003-09-23 | 2014-02-04 | Vertex Pharmaceuticals Incorporated | Compositions useful as inhibitors of protein kinases |
| US11384082B2 (en) | 2017-08-25 | 2022-07-12 | Kronos Bio, Inc. | Hydrates of polymorphs of 6-(1H-indazol-6-YL)-N-(4-morpholinophenyl)-2,3-dihydroimidazo[1,2-A]pyrazin-8-amine bisemsylate as Syk inhibitors |
Also Published As
| Publication number | Publication date |
|---|---|
| PL314187A1 (en) | 1996-09-02 |
| ES2140565T3 (es) | 2000-03-01 |
| GR3033034T3 (en) | 2000-08-31 |
| PT726899E (pt) | 2000-04-28 |
| DK0726899T3 (da) | 2000-06-26 |
| ZA948669B (en) | 1995-07-04 |
| EP0726899A1 (de) | 1996-08-21 |
| KR960705815A (ko) | 1996-11-08 |
| JPH09505035A (ja) | 1997-05-20 |
| ATE188965T1 (de) | 2000-02-15 |
| CO4290432A1 (es) | 1996-04-17 |
| AU7993694A (en) | 1995-05-23 |
| EP0726899B1 (de) | 2000-01-19 |
| CN1134151A (zh) | 1996-10-23 |
| NZ274848A (en) | 1998-03-25 |
| DE4337609A1 (de) | 1995-05-11 |
| CA2175837A1 (en) | 1995-05-11 |
| AU690588B2 (en) | 1998-04-30 |
| DE59409094D1 (de) | 2000-02-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0726899B1 (de) | Neue pyrazincarboxamidderivate, ihre herstellung und ihre verwendung in arzneimitteln | |
| EP0733046B1 (de) | benzoylguanidine, ihre herstellung und ihre verwendung in arzneimitteln | |
| DD202152A5 (de) | Verfahren zur herstellung von phenylpiperazinderivaten | |
| EP0885204B1 (de) | Neue arylglycinamidderivate, verfahren zu ihrer herstellung und diese verbindungen enthaltende pharmazeutische zusammensetzungen | |
| DE2705025A1 (de) | Xanthinderivate, verfahren zu ihrer herstellung und sie enthaltende arzneimittel | |
| EP0598770B1 (de) | Neue pyrazinderivative, ihre herstellung und verwendung | |
| DE3105330A1 (de) | "4-amino-6,7-dimethoxy-2-piperazinylchinazolin-derivate, ihre herstellung und ihre verwendung" | |
| DE2238504A1 (de) | Propanolaminderivate | |
| DE3840380A1 (de) | 3,6-dihydro-1,5(2h)-pyrimidincarbonsaeureester | |
| EP0116360B1 (de) | 1-Phenyl-2-aminocarbonylindol-Verbindungen sowie Verfahren zu ihrer Herstellung und diese Verbindungen enthaltende Arzneimittel | |
| DE69720021T2 (de) | 1,4-disubstituierte piperazine | |
| EP0090369B1 (de) | Salicylsäurederivate, Verfahren zu ihrer Herstellung, pharmazeutische Präparate auf Basis dieser Verbindungen und ihre Verwendung | |
| EP0018360B1 (de) | N-(5-Methoxybentofuran-2-ylcarbonyl)-N'-benzylpiperazin und Verfahren zu dessen Herstellung | |
| DE3014813A1 (de) | 2-hydroxy-5-(1-hydroxy-2-piperazinylethyl)benzoesaeure-derivate | |
| EP0552758B1 (de) | N-5-geschützte 2,5-Diamino-4,6-dichlorpyrimidine und Verfahren zu deren Herstellung | |
| DE3419223C2 (de) | Substituierte Acylpiperazinochinazoline, ein Verfahren zu deren Herstellung und diese enthaltende pharmazeutische Zubereitungen | |
| EP1003728B1 (de) | 2-(3-(4-(2-t-Butyl-6-trifluormethylpyrimidin-4-yl)piperazin-1-yl)propylmercapto)pyrimidin-4-ol-fumarat | |
| DE2725019A1 (de) | Verfahren zur herstellung substituierter aminochinazolinderivate und zwischenprodukte dafuer | |
| EP0663395B1 (de) | 3-(Phenylalkylaminoalkyloxy)-5-phenylpyrazol-Derivate, Verfahren und Zwischenprodukte zu ihrer Herstellung und ihre Verwendung als herzfrequenzsenkendes Mittel | |
| EP0104445A2 (de) | Piperidinochinoxalin-Derivate, ihre Herstellung und Verwendung | |
| DE2711149A1 (de) | (2-pyrimidinyl-thio)alkansaeureamide mit antilypemischer wirkung | |
| EP0816359A1 (de) | 4-Amino-2-ureido-pyrimidin-5-carbonsäureamide, Verfahren zu deren Herstellung, diese Verbindungen enthaltende Arzneimittel und deren Verwendung | |
| CH584695A5 (en) | 3-amino-2-hydroxypropoxy substd. diazines and pyridines - with beta-adrenergic blocking or stimulating activity | |
| EP0318851A2 (de) | Neue Labdanderivate, ein Verfahren zu ihrer Herstellung und ihre Verwendung als Arzneimittel | |
| CH561211A5 (en) | Antihypertensive 3-hydrazino pyrido(4,3-c)pyridazines - prepd. by reacting corresp 3-halo cpd. with hydrazines |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 94194016.0 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AM AU BG CA CN CZ FI GE HU JP KR KZ LT LV NO NZ PL RO RU SI SK UA US UZ VN |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): KE MW SD SZ AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2175837 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1994931018 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 274848 Country of ref document: NZ |
|
| ENP | Entry into the national phase |
Ref document number: 1996 640940 Country of ref document: US Date of ref document: 19960708 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 1994931018 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1994931018 Country of ref document: EP |