KR20220083861A - Pcv2 orf2 단백질 변이체 및 이로 이루어진 바이러스 유사 입자 - Google Patents
Pcv2 orf2 단백질 변이체 및 이로 이루어진 바이러스 유사 입자 Download PDFInfo
- Publication number
- KR20220083861A KR20220083861A KR1020227019654A KR20227019654A KR20220083861A KR 20220083861 A KR20220083861 A KR 20220083861A KR 1020227019654 A KR1020227019654 A KR 1020227019654A KR 20227019654 A KR20227019654 A KR 20227019654A KR 20220083861 A KR20220083861 A KR 20220083861A
- Authority
- KR
- South Korea
- Prior art keywords
- pcv2
- amino acid
- polypeptide
- residue
- infection
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 108090000623 proteins and genes Proteins 0.000 title claims abstract description 24
- 102000004169 proteins and genes Human genes 0.000 title claims abstract description 21
- 239000002245 particle Substances 0.000 title claims description 7
- 241000700605 Viruses Species 0.000 title description 17
- 241001673669 Porcine circovirus 2 Species 0.000 claims abstract description 240
- 208000015181 infectious disease Diseases 0.000 claims abstract description 135
- 239000000203 mixture Substances 0.000 claims abstract description 131
- 230000002163 immunogen Effects 0.000 claims abstract description 92
- 101100382437 Porcine circovirus 2 Cap gene Proteins 0.000 claims abstract description 73
- 238000000034 method Methods 0.000 claims abstract description 69
- 230000035772 mutation Effects 0.000 claims abstract description 57
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 20
- 101710167800 Capsid assembly scaffolding protein Proteins 0.000 claims abstract description 19
- 101710113540 ORF2 protein Proteins 0.000 claims abstract description 19
- 101710090523 Putative movement protein Proteins 0.000 claims abstract description 19
- 201000010099 disease Diseases 0.000 claims abstract description 18
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 260
- 229920001184 polypeptide Polymers 0.000 claims description 258
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 258
- 235000001014 amino acid Nutrition 0.000 claims description 248
- 125000003275 alpha amino acid group Chemical group 0.000 claims description 213
- 241001465754 Metazoa Species 0.000 claims description 129
- 229940024606 amino acid Drugs 0.000 claims description 65
- 125000000539 amino acid group Chemical group 0.000 claims description 65
- 150000001413 amino acids Chemical class 0.000 claims description 65
- 241000701447 unidentified baculovirus Species 0.000 claims description 58
- 101000977023 Azospirillum brasilense Uncharacterized 17.8 kDa protein in nodG 5'region Proteins 0.000 claims description 53
- 101000961984 Bacillus thuringiensis Uncharacterized 30.3 kDa protein Proteins 0.000 claims description 53
- 101000644901 Drosophila melanogaster Putative 115 kDa protein in type-1 retrotransposable element R1DM Proteins 0.000 claims description 53
- 101000747702 Enterobacteria phage N4 Uncharacterized protein Gp2 Proteins 0.000 claims description 53
- 101000758599 Escherichia coli Uncharacterized 14.7 kDa protein Proteins 0.000 claims description 53
- 101000768930 Lactococcus lactis subsp. cremoris Uncharacterized protein in pepC 5'region Proteins 0.000 claims description 53
- 101000976302 Leptospira interrogans Uncharacterized protein in sph 3'region Proteins 0.000 claims description 53
- 101000778886 Leptospira interrogans serogroup Icterohaemorrhagiae serovar Lai (strain 56601) Uncharacterized protein LA_2151 Proteins 0.000 claims description 53
- 101001121571 Rice tungro bacilliform virus (isolate Philippines) Protein P2 Proteins 0.000 claims description 53
- 101000818098 Spirochaeta aurantia Uncharacterized protein in trpE 3'region Proteins 0.000 claims description 53
- 101001026590 Streptomyces cinnamonensis Putative polyketide beta-ketoacyl synthase 2 Proteins 0.000 claims description 53
- 101000750896 Synechococcus elongatus (strain PCC 7942 / FACHB-805) Uncharacterized protein Synpcc7942_2318 Proteins 0.000 claims description 53
- 101000916321 Xenopus laevis Transposon TX1 uncharacterized 149 kDa protein Proteins 0.000 claims description 53
- 101000760088 Zymomonas mobilis subsp. mobilis (strain ATCC 10988 / DSM 424 / LMG 404 / NCIMB 8938 / NRRL B-806 / ZM1) 20.9 kDa protein Proteins 0.000 claims description 53
- 125000000637 arginyl group Chemical group N[C@@H](CCCNC(N)=N)C(=O)* 0.000 claims description 52
- 238000011282 treatment Methods 0.000 claims description 50
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 claims description 49
- 206010058874 Viraemia Diseases 0.000 claims description 38
- 239000000427 antigen Substances 0.000 claims description 32
- 102000036639 antigens Human genes 0.000 claims description 32
- 108091007433 antigens Proteins 0.000 claims description 32
- 108091033319 polynucleotide Proteins 0.000 claims description 31
- 102000040430 polynucleotide Human genes 0.000 claims description 31
- 239000002157 polynucleotide Substances 0.000 claims description 31
- 230000002829 reductive effect Effects 0.000 claims description 27
- 229960005486 vaccine Drugs 0.000 claims description 26
- 238000006467 substitution reaction Methods 0.000 claims description 25
- 230000028993 immune response Effects 0.000 claims description 19
- 235000018102 proteins Nutrition 0.000 claims description 18
- 230000009467 reduction Effects 0.000 claims description 18
- 125000000341 threoninyl group Chemical group [H]OC([H])(C([H])([H])[H])C([H])(N([H])[H])C(*)=O 0.000 claims description 18
- 230000002265 prevention Effects 0.000 claims description 17
- 210000001519 tissue Anatomy 0.000 claims description 13
- 125000000741 isoleucyl group Chemical group [H]N([H])C(C(C([H])([H])[H])C([H])([H])C([H])([H])[H])C(=O)O* 0.000 claims description 12
- COLNVLDHVKWLRT-QMMMGPOBSA-N phenylalanine group Chemical group N[C@@H](CC1=CC=CC=C1)C(=O)O COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 claims description 12
- 238000011321 prophylaxis Methods 0.000 claims description 12
- 125000000613 asparagine group Chemical group N[C@@H](CC(N)=O)C(=O)* 0.000 claims description 11
- 230000006698 induction Effects 0.000 claims description 11
- 239000013612 plasmid Substances 0.000 claims description 11
- 206010061218 Inflammation Diseases 0.000 claims description 10
- 125000003295 alanine group Chemical group N[C@@H](C)C(=O)* 0.000 claims description 10
- 230000004054 inflammatory process Effects 0.000 claims description 10
- 210000003563 lymphoid tissue Anatomy 0.000 claims description 10
- 238000004519 manufacturing process Methods 0.000 claims description 10
- 125000003607 serino group Chemical group [H]N([H])[C@]([H])(C(=O)[*])C(O[H])([H])[H] 0.000 claims description 9
- 125000001500 prolyl group Chemical group [H]N1C([H])(C(=O)[*])C([H])([H])C([H])([H])C1([H])[H] 0.000 claims description 8
- 208000003322 Coinfection Diseases 0.000 claims description 7
- 241000238631 Hexapoda Species 0.000 claims description 7
- 206010035664 Pneumonia Diseases 0.000 claims description 7
- 206010037660 Pyrexia Diseases 0.000 claims description 7
- 239000013604 expression vector Substances 0.000 claims description 7
- 210000004072 lung Anatomy 0.000 claims description 7
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 claims description 6
- 101000768804 Micromonospora olivasterospora Uncharacterized 10.9 kDa protein in fmrO 5'region Proteins 0.000 claims description 6
- 235000021051 daily weight gain Nutrition 0.000 claims description 6
- 125000000404 glutamine group Chemical group N[C@@H](CCC(N)=O)C(=O)* 0.000 claims description 6
- 125000003630 glycyl group Chemical group [H]N([H])C([H])([H])C(*)=O 0.000 claims description 6
- 230000002209 hydrophobic effect Effects 0.000 claims description 6
- 125000001909 leucine group Chemical group [H]N(*)C(C(*)=O)C([H])([H])C(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 6
- 238000002360 preparation method Methods 0.000 claims description 6
- 125000000430 tryptophan group Chemical group [H]N([H])C(C(=O)O*)C([H])([H])C1=C([H])N([H])C2=C([H])C([H])=C([H])C([H])=C12 0.000 claims description 6
- 125000001493 tyrosinyl group Chemical group [H]OC1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 claims description 6
- 125000002987 valine group Chemical group [H]N([H])C([H])(C(*)=O)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 6
- 239000003814 drug Substances 0.000 claims description 5
- 230000001926 lymphatic effect Effects 0.000 claims description 5
- 238000012217 deletion Methods 0.000 claims description 4
- 230000037430 deletion Effects 0.000 claims description 4
- 230000002458 infectious effect Effects 0.000 claims description 4
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 claims description 2
- 235000004279 alanine Nutrition 0.000 claims description 2
- 230000003247 decreasing effect Effects 0.000 claims description 2
- 235000021052 average daily weight gain Nutrition 0.000 claims 1
- 238000002255 vaccination Methods 0.000 abstract description 22
- 101710159752 Poly(3-hydroxyalkanoate) polymerase subunit PhaE Proteins 0.000 description 67
- 101710130262 Probable Vpr-like protein Proteins 0.000 description 67
- 241000202347 Porcine circovirus Species 0.000 description 46
- 210000004027 cell Anatomy 0.000 description 40
- KERCOYANYUPLHJ-XGEHTFHBSA-N Thr-Pro-Ser Chemical compound C[C@@H](O)[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(O)=O KERCOYANYUPLHJ-XGEHTFHBSA-N 0.000 description 36
- 241000282887 Suidae Species 0.000 description 31
- 239000002773 nucleotide Substances 0.000 description 24
- 125000003729 nucleotide group Chemical group 0.000 description 24
- PLDJDCJLRCYPJB-VOAKCMCISA-N Lys-Lys-Thr Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(O)=O PLDJDCJLRCYPJB-VOAKCMCISA-N 0.000 description 22
- 239000002671 adjuvant Substances 0.000 description 22
- SWWCDAGDQHTKIE-RHYQMDGZSA-N Lys-Arg-Thr Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(O)=O SWWCDAGDQHTKIE-RHYQMDGZSA-N 0.000 description 21
- 238000006722 reduction reaction Methods 0.000 description 16
- 239000003795 chemical substances by application Substances 0.000 description 15
- YBIAYFFIVAZXPK-AVGNSLFASA-N Arg-His-Arg Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O YBIAYFFIVAZXPK-AVGNSLFASA-N 0.000 description 13
- 108020004414 DNA Proteins 0.000 description 11
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 11
- 239000013598 vector Substances 0.000 description 11
- 239000013603 viral vector Substances 0.000 description 11
- XPSGESXVBSQZPL-SRVKXCTJSA-N Arg-Arg-Arg Chemical compound NC(N)=NCCC[C@H](N)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O XPSGESXVBSQZPL-SRVKXCTJSA-N 0.000 description 10
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 10
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 10
- 108010062796 arginyllysine Proteins 0.000 description 10
- MEFILNJXAVSUTO-JXUBOQSCSA-N Ala-Leu-Thr Chemical compound C[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O MEFILNJXAVSUTO-JXUBOQSCSA-N 0.000 description 9
- LYILPUNCKACNGF-NAKRPEOUSA-N Ala-Val-Ile Chemical compound CC[C@H](C)[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@H](C)N LYILPUNCKACNGF-NAKRPEOUSA-N 0.000 description 9
- OVVUNXXROOFSIM-SDDRHHMPSA-N Arg-Arg-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CCCN=C(N)N)N)C(=O)O OVVUNXXROOFSIM-SDDRHHMPSA-N 0.000 description 9
- CPSHGRGUPZBMOK-CIUDSAMLSA-N Arg-Asn-Gln Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O CPSHGRGUPZBMOK-CIUDSAMLSA-N 0.000 description 9
- NKNILFJYKKHBKE-WPRPVWTQSA-N Arg-Gly-Val Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)NCC(=O)N[C@@H](C(C)C)C(O)=O NKNILFJYKKHBKE-WPRPVWTQSA-N 0.000 description 9
- NVUIWHJLPSZZQC-CYDGBPFRSA-N Arg-Ile-Arg Chemical compound NC(N)=NCCC[C@H](N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O NVUIWHJLPSZZQC-CYDGBPFRSA-N 0.000 description 9
- GMFAGHNRXPSSJS-SRVKXCTJSA-N Arg-Leu-Gln Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O GMFAGHNRXPSSJS-SRVKXCTJSA-N 0.000 description 9
- APHUDFFMXFYRKP-CIUDSAMLSA-N Asn-Asn-Lys Chemical compound C(CCN)C[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)N)NC(=O)[C@H](CC(=O)N)N APHUDFFMXFYRKP-CIUDSAMLSA-N 0.000 description 9
- 108091026890 Coding region Proteins 0.000 description 9
- NSNUZSPSADIMJQ-WDSKDSINSA-N Gln-Gly-Asp Chemical compound NC(=O)CC[C@H](N)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(O)=O NSNUZSPSADIMJQ-WDSKDSINSA-N 0.000 description 9
- FFJQHWKSGAWSTJ-BFHQHQDPSA-N Gly-Thr-Ala Chemical compound [H]NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(O)=O FFJQHWKSGAWSTJ-BFHQHQDPSA-N 0.000 description 9
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 9
- FHKZHRMERJUXRJ-DCAQKATOSA-N His-Ser-Arg Chemical compound NC(N)=NCCC[C@@H](C(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC1=CN=CN1 FHKZHRMERJUXRJ-DCAQKATOSA-N 0.000 description 9
- XVZJRZQIHJMUBG-TUBUOCAGSA-N His-Thr-Ile Chemical compound CC[C@H](C)[C@@H](C(=O)O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC1=CN=CN1)N XVZJRZQIHJMUBG-TUBUOCAGSA-N 0.000 description 9
- DLFAACQHIRSQGG-CIUDSAMLSA-N Leu-Asp-Asp Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(O)=O)C(O)=O DLFAACQHIRSQGG-CIUDSAMLSA-N 0.000 description 9
- IDGZVZJLYFTXSL-DCAQKATOSA-N Leu-Ser-Arg Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CCCN=C(N)N IDGZVZJLYFTXSL-DCAQKATOSA-N 0.000 description 9
- FPFOYSCDUWTZBF-IHPCNDPISA-N Leu-Trp-Leu Chemical compound C1=CC=C2C(C[C@H](NC(=O)[C@@H]([NH3+])CC(C)C)C(=O)N[C@@H](CC(C)C)C([O-])=O)=CNC2=C1 FPFOYSCDUWTZBF-IHPCNDPISA-N 0.000 description 9
- YSPZCHGIWAQVKQ-AVGNSLFASA-N Lys-Pro-Val Chemical compound CC(C)[C@@H](C(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)CCCCN YSPZCHGIWAQVKQ-AVGNSLFASA-N 0.000 description 9
- NYTDJEZBAAFLLG-IHRRRGAJSA-N Lys-Val-Lys Chemical compound NCCCC[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(O)=O NYTDJEZBAAFLLG-IHRRRGAJSA-N 0.000 description 9
- NSMXRFMGZYTFEX-KJEVXHAQSA-N Met-Thr-Tyr Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)NC(=O)[C@H](CCSC)N)O NSMXRFMGZYTFEX-KJEVXHAQSA-N 0.000 description 9
- FMMIYCMOVGXZIP-AVGNSLFASA-N Phe-Glu-Asn Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O FMMIYCMOVGXZIP-AVGNSLFASA-N 0.000 description 9
- XNMYNGDKJNOKHH-BZSNNMDCSA-N Phe-Ser-Tyr Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O XNMYNGDKJNOKHH-BZSNNMDCSA-N 0.000 description 9
- SHUFSZDAIPLZLF-BEAPCOKYSA-N Phe-Thr-Pro Chemical compound C[C@H]([C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CC2=CC=CC=C2)N)O SHUFSZDAIPLZLF-BEAPCOKYSA-N 0.000 description 9
- MWQXFDIQXIXPMS-UNQGMJICSA-N Phe-Val-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](C(C)C)NC(=O)[C@H](CC1=CC=CC=C1)N)O MWQXFDIQXIXPMS-UNQGMJICSA-N 0.000 description 9
- LNLNHXIQPGKRJQ-SRVKXCTJSA-N Pro-Arg-Arg Chemical compound NC(N)=NCCC[C@@H](C(O)=O)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H]1CCCN1 LNLNHXIQPGKRJQ-SRVKXCTJSA-N 0.000 description 9
- XSXABUHLKPUVLX-JYJNAYRXSA-N Pro-Ser-Trp Chemical compound C1C[C@H](NC1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC2=CNC3=CC=CC=C32)C(=O)O XSXABUHLKPUVLX-JYJNAYRXSA-N 0.000 description 9
- GZGFSPWOMUKKCV-NAKRPEOUSA-N Ser-Pro-Ile Chemical compound CC[C@H](C)[C@@H](C(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](N)CO GZGFSPWOMUKKCV-NAKRPEOUSA-N 0.000 description 9
- KQNDIKOYWZTZIX-FXQIFTODSA-N Ser-Ser-Arg Chemical compound OC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CCCNC(N)=N KQNDIKOYWZTZIX-FXQIFTODSA-N 0.000 description 9
- CRZNCABIJLRFKZ-IUKAMOBKSA-N Thr-Ile-Asp Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)O)NC(=O)[C@H]([C@@H](C)O)N CRZNCABIJLRFKZ-IUKAMOBKSA-N 0.000 description 9
- WMBFONUKQXGLMU-WDSOQIARSA-N Trp-Leu-Val Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](C(C)C)C(=O)O)NC(=O)[C@H](CC1=CNC2=CC=CC=C21)N WMBFONUKQXGLMU-WDSOQIARSA-N 0.000 description 9
- KXIQQAWIPDDVOE-BPUTZDHNSA-N Trp-Pro-Cys Chemical compound O=C([C@H](CC=1C2=CC=CC=C2NC=1)N)N1CCC[C@H]1C(=O)N[C@@H](CS)C(O)=O KXIQQAWIPDDVOE-BPUTZDHNSA-N 0.000 description 9
- YLHFIMLKNPJRGY-BVSLBCMMSA-N Tyr-Arg-Trp Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(O)=O YLHFIMLKNPJRGY-BVSLBCMMSA-N 0.000 description 9
- FDKDGFGTHGJKNV-FHWLQOOXSA-N Tyr-Phe-Gln Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](CC2=CC=C(C=C2)O)N FDKDGFGTHGJKNV-FHWLQOOXSA-N 0.000 description 9
- AEOFMCAKYIQQFY-YDHLFZDLSA-N Tyr-Val-Asn Chemical compound NC(=O)C[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 AEOFMCAKYIQQFY-YDHLFZDLSA-N 0.000 description 9
- ZQGPWORGSNRQLN-NHCYSSNCSA-N Val-Asp-His Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)N ZQGPWORGSNRQLN-NHCYSSNCSA-N 0.000 description 9
- FOADDSDHGRFUOC-DZKIICNBSA-N Val-Glu-Phe Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)O)N FOADDSDHGRFUOC-DZKIICNBSA-N 0.000 description 9
- URIRWLJVWHYLET-ONGXEEELSA-N Val-Gly-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)CNC(=O)[C@@H](N)C(C)C URIRWLJVWHYLET-ONGXEEELSA-N 0.000 description 9
- 108010068265 aspartyltyrosine Proteins 0.000 description 9
- 150000002148 esters Chemical class 0.000 description 9
- 238000011156 evaluation Methods 0.000 description 9
- XKUKSGPZAADMRA-UHFFFAOYSA-N glycyl-glycyl-glycine Natural products NCC(=O)NCC(=O)NCC(O)=O XKUKSGPZAADMRA-UHFFFAOYSA-N 0.000 description 9
- 108010085325 histidylproline Proteins 0.000 description 9
- 125000001360 methionine group Chemical group N[C@@H](CCSC)C(=O)* 0.000 description 9
- 108010083476 phenylalanyltryptophan Proteins 0.000 description 9
- 108010071635 tyrosyl-prolyl-arginine Proteins 0.000 description 9
- 230000003612 virological effect Effects 0.000 description 9
- HJVGMOYJDDXLMI-AVGNSLFASA-N Arg-Arg-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CCCNC(N)=N HJVGMOYJDDXLMI-AVGNSLFASA-N 0.000 description 8
- PBSQFBAJKPLRJY-BYULHYEWSA-N Asn-Gly-Ile Chemical compound CC[C@H](C)[C@@H](C(=O)O)NC(=O)CNC(=O)[C@H](CC(=O)N)N PBSQFBAJKPLRJY-BYULHYEWSA-N 0.000 description 8
- YVXRYLVELQYAEQ-SRVKXCTJSA-N Asn-Leu-Lys Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CC(=O)N)N YVXRYLVELQYAEQ-SRVKXCTJSA-N 0.000 description 8
- BKOIIURTQAJHAT-GUBZILKMSA-N Asp-Pro-Pro Chemical compound OC(=O)C[C@H](N)C(=O)N1CCC[C@H]1C(=O)N1[C@H](C(O)=O)CCC1 BKOIIURTQAJHAT-GUBZILKMSA-N 0.000 description 8
- BPQYBFAXRGMGGY-LAEOZQHASA-N Gly-Gln-Ile Chemical compound CC[C@H](C)[C@@H](C(=O)O)NC(=O)[C@H](CCC(=O)N)NC(=O)CN BPQYBFAXRGMGGY-LAEOZQHASA-N 0.000 description 8
- HTKNPQZCMLBOTQ-XVSYOHENSA-N Phe-Asn-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC(=O)N)NC(=O)[C@H](CC1=CC=CC=C1)N)O HTKNPQZCMLBOTQ-XVSYOHENSA-N 0.000 description 8
- LEIKGVHQTKHOLM-IUCAKERBSA-N Pro-Pro-Gly Chemical compound OC(=O)CNC(=O)[C@@H]1CCCN1C(=O)[C@H]1NCCC1 LEIKGVHQTKHOLM-IUCAKERBSA-N 0.000 description 8
- CICQXRWZNVXFCU-SRVKXCTJSA-N Ser-His-Leu Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CC(C)C)C(O)=O CICQXRWZNVXFCU-SRVKXCTJSA-N 0.000 description 8
- MQQBBLVOUUJKLH-HJPIBITLSA-N Ser-Ile-Tyr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O MQQBBLVOUUJKLH-HJPIBITLSA-N 0.000 description 8
- 229920002125 Sokalan® Polymers 0.000 description 8
- DIPIPFHFLPTCLK-LOKLDPHHSA-N Thr-Gln-Pro Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N1CCC[C@@H]1C(=O)O)N)O DIPIPFHFLPTCLK-LOKLDPHHSA-N 0.000 description 8
- NRFTYDWKWGJLAR-MELADBBJSA-N Tyr-Asp-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CC(=O)O)NC(=O)[C@H](CC2=CC=C(C=C2)O)N)C(=O)O NRFTYDWKWGJLAR-MELADBBJSA-N 0.000 description 8
- 108010068380 arginylarginine Proteins 0.000 description 8
- 238000004113 cell culture Methods 0.000 description 8
- 108010050848 glycylleucine Proteins 0.000 description 8
- 238000003306 harvesting Methods 0.000 description 8
- 230000000415 inactivating effect Effects 0.000 description 8
- 230000003902 lesion Effects 0.000 description 8
- 108010087846 prolyl-prolyl-glycine Proteins 0.000 description 8
- 102200132330 rs1555538151 Human genes 0.000 description 8
- 238000012546 transfer Methods 0.000 description 8
- 239000004475 Arginine Substances 0.000 description 7
- YWAQATDNEKZFFK-BYPYZUCNSA-N Gly-Gly-Ser Chemical compound NCC(=O)NCC(=O)N[C@@H](CO)C(O)=O YWAQATDNEKZFFK-BYPYZUCNSA-N 0.000 description 7
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 7
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 7
- 230000000694 effects Effects 0.000 description 7
- 108010045069 keyhole-limpet hemocyanin Proteins 0.000 description 7
- 210000002966 serum Anatomy 0.000 description 7
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 6
- 206010023126 Jaundice Diseases 0.000 description 6
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 6
- 208000008771 Lymphadenopathy Diseases 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 6
- 239000002269 analeptic agent Substances 0.000 description 6
- 108010077245 asparaginyl-proline Proteins 0.000 description 6
- 230000036039 immunity Effects 0.000 description 6
- 208000018555 lymphatic system disease Diseases 0.000 description 6
- 239000000463 material Substances 0.000 description 6
- 239000008188 pellet Substances 0.000 description 6
- 208000028489 postweaning multisystemic wasting syndrome Diseases 0.000 description 6
- 230000001850 reproductive effect Effects 0.000 description 6
- 239000006228 supernatant Substances 0.000 description 6
- OLISTMZJGQUOGS-GMOBBJLQSA-N Asn-Ile-Arg Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CCCN=C(N)N)C(=O)O)NC(=O)[C@H](CC(=O)N)N OLISTMZJGQUOGS-GMOBBJLQSA-N 0.000 description 5
- LSYFGBRDBIQYAQ-FHWLQOOXSA-N Glu-Tyr-Tyr Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O LSYFGBRDBIQYAQ-FHWLQOOXSA-N 0.000 description 5
- GBYYQVBXFVDJPJ-WLTAIBSBSA-N Gly-Tyr-Thr Chemical compound C[C@H]([C@@H](C(=O)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)NC(=O)CN)O GBYYQVBXFVDJPJ-WLTAIBSBSA-N 0.000 description 5
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 5
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 5
- 125000000174 L-prolyl group Chemical group [H]N1C([H])([H])C([H])([H])C([H])([H])[C@@]1([H])C(*)=O 0.000 description 5
- 125000000510 L-tryptophano group Chemical group [H]C1=C([H])C([H])=C2N([H])C([H])=C(C([H])([H])[C@@]([H])(C(O[H])=O)N([H])[*])C2=C1[H] 0.000 description 5
- OXIWIYOJVNOKOV-SRVKXCTJSA-N Met-Met-Arg Chemical compound CSCC[C@H](N)C(=O)N[C@@H](CCSC)C(=O)N[C@H](C(O)=O)CCCNC(N)=N OXIWIYOJVNOKOV-SRVKXCTJSA-N 0.000 description 5
- PESQCPHRXOFIPX-UHFFFAOYSA-N N-L-methionyl-L-tyrosine Natural products CSCCC(N)C(=O)NC(C(O)=O)CC1=CC=C(O)C=C1 PESQCPHRXOFIPX-UHFFFAOYSA-N 0.000 description 5
- XMBSYZWANAQXEV-UHFFFAOYSA-N N-alpha-L-glutamyl-L-phenylalanine Natural products OC(=O)CCC(N)C(=O)NC(C(O)=O)CC1=CC=CC=C1 XMBSYZWANAQXEV-UHFFFAOYSA-N 0.000 description 5
- 206010033546 Pallor Diseases 0.000 description 5
- MPGJIHFJCXTVEX-KKUMJFAQSA-N Phe-Arg-Glu Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(O)=O MPGJIHFJCXTVEX-KKUMJFAQSA-N 0.000 description 5
- KIEPQOIQHFKQLK-PCBIJLKTSA-N Phe-Asn-Ile Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O KIEPQOIQHFKQLK-PCBIJLKTSA-N 0.000 description 5
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 5
- 229930006000 Sucrose Natural products 0.000 description 5
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 5
- 241000282898 Sus scrofa Species 0.000 description 5
- KLOZTPOXVVRVAQ-DZKIICNBSA-N Tyr-Val-Gln Chemical compound NC(=O)CC[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 KLOZTPOXVVRVAQ-DZKIICNBSA-N 0.000 description 5
- QIVPZSWBBHRNBA-JYJNAYRXSA-N Val-Pro-Phe Chemical compound CC(C)[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1ccccc1)C(O)=O QIVPZSWBBHRNBA-JYJNAYRXSA-N 0.000 description 5
- 239000000839 emulsion Substances 0.000 description 5
- 239000000499 gel Substances 0.000 description 5
- 201000008298 histiocytosis Diseases 0.000 description 5
- 230000001965 increasing effect Effects 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- 108010012581 phenylalanylglutamate Proteins 0.000 description 5
- 229910052698 phosphorus Inorganic materials 0.000 description 5
- 108010090894 prolylleucine Proteins 0.000 description 5
- 230000000069 prophylactic effect Effects 0.000 description 5
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 5
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 5
- 235000019345 sodium thiosulphate Nutrition 0.000 description 5
- 239000005720 sucrose Substances 0.000 description 5
- 208000011580 syndromic disease Diseases 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- 206010000234 Abortion spontaneous Diseases 0.000 description 4
- SKTGPBFTMNLIHQ-KKUMJFAQSA-N Arg-Glu-Phe Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O SKTGPBFTMNLIHQ-KKUMJFAQSA-N 0.000 description 4
- PTSDPWIHOYMRGR-UGYAYLCHSA-N Asn-Ile-Asn Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(N)=O)C(O)=O PTSDPWIHOYMRGR-UGYAYLCHSA-N 0.000 description 4
- LTCKTLYKRMCFOC-KKUMJFAQSA-N Asp-Phe-Leu Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CC(C)C)C(O)=O LTCKTLYKRMCFOC-KKUMJFAQSA-N 0.000 description 4
- 108020004705 Codon Proteins 0.000 description 4
- 102220527016 Cytochrome b5 reductase 4_R63K_mutation Human genes 0.000 description 4
- 201000004624 Dermatitis Diseases 0.000 description 4
- OFPWCBGRYAOLMU-AVGNSLFASA-N Gln-Asp-Tyr Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CCC(=O)N)N)O OFPWCBGRYAOLMU-AVGNSLFASA-N 0.000 description 4
- FUESBOMYALLFNI-VKHMYHEASA-N Gly-Asn Chemical compound NCC(=O)N[C@H](C(O)=O)CC(N)=O FUESBOMYALLFNI-VKHMYHEASA-N 0.000 description 4
- LCRDMSSAKLTKBU-ZDLURKLDSA-N Gly-Ser-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)[C@H](CO)NC(=O)CN LCRDMSSAKLTKBU-ZDLURKLDSA-N 0.000 description 4
- PROLDOGUBQJNPG-RWMBFGLXSA-N His-Arg-Pro Chemical compound C1C[C@@H](N(C1)C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC2=CN=CN2)N)C(=O)O PROLDOGUBQJNPG-RWMBFGLXSA-N 0.000 description 4
- 208000029523 Interstitial Lung disease Diseases 0.000 description 4
- 241001148567 Lawsonia intracellularis Species 0.000 description 4
- WGNOPSQMIQERPK-GARJFASQSA-N Leu-Asn-Pro Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)N1CCC[C@@H]1C(=O)O)N WGNOPSQMIQERPK-GARJFASQSA-N 0.000 description 4
- WGNOPSQMIQERPK-UHFFFAOYSA-N Leu-Asn-Pro Natural products CC(C)CC(N)C(=O)NC(CC(=O)N)C(=O)N1CCCC1C(=O)O WGNOPSQMIQERPK-UHFFFAOYSA-N 0.000 description 4
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 4
- OBVHKUFUDCPZDW-JYJNAYRXSA-N Met-Arg-Phe Chemical compound CSCC[C@H](N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 OBVHKUFUDCPZDW-JYJNAYRXSA-N 0.000 description 4
- 101150102680 ORF2b gene Proteins 0.000 description 4
- 206010030113 Oedema Diseases 0.000 description 4
- UAMFZRNCIFFMLE-FHWLQOOXSA-N Phe-Glu-Tyr Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CC2=CC=C(C=C2)O)C(=O)O)N UAMFZRNCIFFMLE-FHWLQOOXSA-N 0.000 description 4
- 241001135989 Porcine reproductive and respiratory syndrome virus Species 0.000 description 4
- 208000007107 Stomach Ulcer Diseases 0.000 description 4
- FDQXPJCLVPFKJW-KJEVXHAQSA-N Thr-Met-Tyr Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)O)N)O FDQXPJCLVPFKJW-KJEVXHAQSA-N 0.000 description 4
- QNXZCKMXHPULME-ZNSHCXBVSA-N Thr-Val-Pro Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](C(C)C)C(=O)N1CCC[C@@H]1C(=O)O)N)O QNXZCKMXHPULME-ZNSHCXBVSA-N 0.000 description 4
- KZTLZZQTJMCGIP-ZJDVBMNYSA-N Thr-Val-Thr Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O KZTLZZQTJMCGIP-ZJDVBMNYSA-N 0.000 description 4
- MTEQZJFSEMXXRK-CFMVVWHZSA-N Tyr-Asn-Ile Chemical compound CC[C@H](C)[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)N)NC(=O)[C@H](CC1=CC=C(C=C1)O)N MTEQZJFSEMXXRK-CFMVVWHZSA-N 0.000 description 4
- ZSZFTYVFQLUWBF-QXEWZRGKSA-N Val-Asp-Met Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CCSC)C(=O)O)N ZSZFTYVFQLUWBF-QXEWZRGKSA-N 0.000 description 4
- JXGWQYWDUOWQHA-DZKIICNBSA-N Val-Gln-Phe Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)O)N JXGWQYWDUOWQHA-DZKIICNBSA-N 0.000 description 4
- 239000003242 anti bacterial agent Substances 0.000 description 4
- 108010060035 arginylproline Proteins 0.000 description 4
- 238000003556 assay Methods 0.000 description 4
- 230000002238 attenuated effect Effects 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 239000003085 diluting agent Substances 0.000 description 4
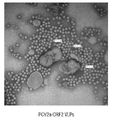
- 238000001493 electron microscopy Methods 0.000 description 4
- 201000005917 gastric ulcer Diseases 0.000 description 4
- XBGGUPMXALFZOT-UHFFFAOYSA-N glycyl-L-tyrosine hemihydrate Natural products NCC(=O)NC(C(O)=O)CC1=CC=C(O)C=C1 XBGGUPMXALFZOT-UHFFFAOYSA-N 0.000 description 4
- 108010087823 glycyltyrosine Proteins 0.000 description 4
- 230000002779 inactivation Effects 0.000 description 4
- 239000007951 isotonicity adjuster Substances 0.000 description 4
- 208000017169 kidney disease Diseases 0.000 description 4
- 210000004185 liver Anatomy 0.000 description 4
- 230000008774 maternal effect Effects 0.000 description 4
- 239000002609 medium Substances 0.000 description 4
- 108010085203 methionylmethionine Proteins 0.000 description 4
- 208000015994 miscarriage Diseases 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 239000013642 negative control Substances 0.000 description 4
- 201000008383 nephritis Diseases 0.000 description 4
- 230000003472 neutralizing effect Effects 0.000 description 4
- 230000001717 pathogenic effect Effects 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- 208000023504 respiratory system disease Diseases 0.000 description 4
- 239000011780 sodium chloride Substances 0.000 description 4
- 208000000995 spontaneous abortion Diseases 0.000 description 4
- 239000003381 stabilizer Substances 0.000 description 4
- 208000002254 stillbirth Diseases 0.000 description 4
- 231100000537 stillbirth Toxicity 0.000 description 4
- 229940031626 subunit vaccine Drugs 0.000 description 4
- 108010003137 tyrosyltyrosine Proteins 0.000 description 4
- 235000019786 weight gain Nutrition 0.000 description 4
- 230000004584 weight gain Effects 0.000 description 4
- 230000004580 weight loss Effects 0.000 description 4
- NIXOWILDQLNWCW-UHFFFAOYSA-N 2-Propenoic acid Natural products OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- INXWADWANGLMPJ-JYJNAYRXSA-N Arg-Phe-Arg Chemical compound NC(=N)NCCC[C@H](N)C(=O)N[C@H](C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)CC1=CC=CC=C1 INXWADWANGLMPJ-JYJNAYRXSA-N 0.000 description 3
- JZRLLSOWDYUKOK-SRVKXCTJSA-N Asn-Asp-Phe Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)O)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CC(=O)N)N JZRLLSOWDYUKOK-SRVKXCTJSA-N 0.000 description 3
- JZLFYAAGGYMRIK-BYULHYEWSA-N Asn-Val-Asp Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(O)=O)C(O)=O JZLFYAAGGYMRIK-BYULHYEWSA-N 0.000 description 3
- 241001533384 Circovirus Species 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- JJQQGCMKLOEGAV-OSUNSFLBSA-N Ile-Thr-Met Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCSC)C(=O)O)N JJQQGCMKLOEGAV-OSUNSFLBSA-N 0.000 description 3
- YKNBJXOJTURHCU-DCAQKATOSA-N Leu-Asp-Arg Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C(O)=O)CCCN=C(N)N YKNBJXOJTURHCU-DCAQKATOSA-N 0.000 description 3
- MPOHDJKRBLVGCT-CIUDSAMLSA-N Lys-Ala-Asn Chemical compound C[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](CCCCN)N MPOHDJKRBLVGCT-CIUDSAMLSA-N 0.000 description 3
- KNKHAVVBVXKOGX-JXUBOQSCSA-N Lys-Ala-Thr Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O KNKHAVVBVXKOGX-JXUBOQSCSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 241001661006 Pepper cryptic virus 2 Species 0.000 description 3
- 108020004511 Recombinant DNA Proteins 0.000 description 3
- 238000011053 TCID50 method Methods 0.000 description 3
- UQCNIMDPYICBTR-KYNKHSRBSA-N Thr-Thr-Gly Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(O)=O UQCNIMDPYICBTR-KYNKHSRBSA-N 0.000 description 3
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 3
- 239000004473 Threonine Substances 0.000 description 3
- JHDZONWZTCKTJR-KJEVXHAQSA-N Tyr-Thr-Val Chemical compound CC(C)[C@@H](C(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 JHDZONWZTCKTJR-KJEVXHAQSA-N 0.000 description 3
- CKLJMWTZIZZHCS-REOHCLBHSA-L aspartate group Chemical group N[C@@H](CC(=O)[O-])C(=O)[O-] CKLJMWTZIZZHCS-REOHCLBHSA-L 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 210000000234 capsid Anatomy 0.000 description 3
- 238000005119 centrifugation Methods 0.000 description 3
- -1 coatings Substances 0.000 description 3
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- WHUUTDBJXJRKMK-VKHMYHEASA-L glutamate group Chemical group N[C@@H](CCC(=O)[O-])C(=O)[O-] WHUUTDBJXJRKMK-VKHMYHEASA-L 0.000 description 3
- 125000000487 histidyl group Chemical group [H]N([H])C(C(=O)O*)C([H])([H])C1=C([H])N([H])C([H])=N1 0.000 description 3
- 230000000670 limiting effect Effects 0.000 description 3
- 230000001404 mediated effect Effects 0.000 description 3
- 239000003921 oil Substances 0.000 description 3
- 235000019198 oils Nutrition 0.000 description 3
- 244000052769 pathogen Species 0.000 description 3
- 108010082795 phenylalanyl-arginyl-arginine Proteins 0.000 description 3
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 3
- 239000013641 positive control Substances 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 108010089164 prolyl-leucyl-asparagyl-prolyl-lysine Proteins 0.000 description 3
- 238000011002 quantification Methods 0.000 description 3
- 238000005070 sampling Methods 0.000 description 3
- 238000013207 serial dilution Methods 0.000 description 3
- 238000004904 shortening Methods 0.000 description 3
- 238000002741 site-directed mutagenesis Methods 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 241000894007 species Species 0.000 description 3
- PRAKJMSDJKAYCZ-UHFFFAOYSA-N squalane Chemical compound CC(C)CCCC(C)CCCC(C)CCCCC(C)CCCC(C)CCCC(C)C PRAKJMSDJKAYCZ-UHFFFAOYSA-N 0.000 description 3
- 108010073969 valyllysine Proteins 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- 238000001262 western blot Methods 0.000 description 3
- XDOFQFKRPWOURC-UHFFFAOYSA-N 16-methylheptadecanoic acid Chemical compound CC(C)CCCCCCCCCCCCCCC(O)=O XDOFQFKRPWOURC-UHFFFAOYSA-N 0.000 description 2
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 2
- JVTIXNMXDLQEJE-UHFFFAOYSA-N 2-decanoyloxypropyl decanoate 2-octanoyloxypropyl octanoate Chemical compound C(CCCCCCC)(=O)OCC(C)OC(CCCCCCC)=O.C(=O)(CCCCCCCCC)OCC(C)OC(=O)CCCCCCCCC JVTIXNMXDLQEJE-UHFFFAOYSA-N 0.000 description 2
- BVLPIIBTWIYOML-ZKWXMUAHSA-N Ala-Val-Asp Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(O)=O)C(O)=O BVLPIIBTWIYOML-ZKWXMUAHSA-N 0.000 description 2
- QMQZYILAWUOLPV-JYJNAYRXSA-N Arg-Tyr-Arg Chemical compound NC(N)=NCCC[C@H](N)C(=O)N[C@H](C(=O)N[C@@H](CCCN=C(N)N)C(O)=O)CC1=CC=C(O)C=C1 QMQZYILAWUOLPV-JYJNAYRXSA-N 0.000 description 2
- BYLSYQASFJJBCL-DCAQKATOSA-N Asn-Pro-Leu Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(O)=O BYLSYQASFJJBCL-DCAQKATOSA-N 0.000 description 2
- 102000053602 DNA Human genes 0.000 description 2
- 208000004232 Enteritis Diseases 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- 239000004471 Glycine Substances 0.000 description 2
- 241001662043 Icterus Species 0.000 description 2
- VQTUBCCKSQIDNK-UHFFFAOYSA-N Isobutene Chemical compound CC(C)=C VQTUBCCKSQIDNK-UHFFFAOYSA-N 0.000 description 2
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 2
- PVMPDMIKUVNOBD-CIUDSAMLSA-N Leu-Asp-Ser Chemical compound CC(C)C[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(O)=O PVMPDMIKUVNOBD-CIUDSAMLSA-N 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 2
- 101150009852 ORF2 gene Proteins 0.000 description 2
- 208000005342 Porcine Reproductive and Respiratory Syndrome Diseases 0.000 description 2
- 241000928435 Porcine circovirus 1 Species 0.000 description 2
- 238000012300 Sequence Analysis Methods 0.000 description 2
- 210000001744 T-lymphocyte Anatomy 0.000 description 2
- URPSJRMWHQTARR-MBLNEYKQSA-N Thr-Ile-Gly Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(O)=O URPSJRMWHQTARR-MBLNEYKQSA-N 0.000 description 2
- BIBYEFRASCNLAA-CDMKHQONSA-N Thr-Phe-Gly Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@H](C(=O)NCC(O)=O)CC1=CC=CC=C1 BIBYEFRASCNLAA-CDMKHQONSA-N 0.000 description 2
- OGOYMQWIWHGTGH-KZVJFYERSA-N Thr-Val-Ala Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C)C(O)=O OGOYMQWIWHGTGH-KZVJFYERSA-N 0.000 description 2
- BKIOKSLLAAZYTC-KKHAAJSZSA-N Thr-Val-Asn Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(N)=O)C(O)=O BKIOKSLLAAZYTC-KKHAAJSZSA-N 0.000 description 2
- FYBFTPLPAXZBOY-KKHAAJSZSA-N Thr-Val-Asp Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(O)=O)C(O)=O FYBFTPLPAXZBOY-KKHAAJSZSA-N 0.000 description 2
- AXEJRUGTOJPZKG-XGEHTFHBSA-N Thr-Val-Cys Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CS)C(=O)O)N)O AXEJRUGTOJPZKG-XGEHTFHBSA-N 0.000 description 2
- QGVBFDIREUUSHX-IFFSRLJSSA-N Thr-Val-Gln Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O QGVBFDIREUUSHX-IFFSRLJSSA-N 0.000 description 2
- KPMIQCXJDVKWKO-IFFSRLJSSA-N Thr-Val-Glu Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(O)=O KPMIQCXJDVKWKO-IFFSRLJSSA-N 0.000 description 2
- AKHDFZHUPGVFEJ-YEPSODPASA-N Thr-Val-Gly Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)NCC(O)=O AKHDFZHUPGVFEJ-YEPSODPASA-N 0.000 description 2
- CURFABYITJVKEW-QTKMDUPCSA-N Thr-Val-His Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC1=CN=CN1)C(=O)O)N)O CURFABYITJVKEW-QTKMDUPCSA-N 0.000 description 2
- ILUOMMDDGREELW-OSUNSFLBSA-N Thr-Val-Ile Chemical compound CC[C@H](C)[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](N)[C@@H](C)O ILUOMMDDGREELW-OSUNSFLBSA-N 0.000 description 2
- BKVICMPZWRNWOC-RHYQMDGZSA-N Thr-Val-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](N)[C@@H](C)O BKVICMPZWRNWOC-RHYQMDGZSA-N 0.000 description 2
- SBYQHZCMVSPQCS-RCWTZXSCSA-N Thr-Val-Met Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCSC)C(O)=O SBYQHZCMVSPQCS-RCWTZXSCSA-N 0.000 description 2
- SPIFGZFZMVLPHN-UNQGMJICSA-N Thr-Val-Phe Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC1=CC=CC=C1)C(O)=O SPIFGZFZMVLPHN-UNQGMJICSA-N 0.000 description 2
- MNYNCKZAEIAONY-XGEHTFHBSA-N Thr-Val-Ser Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CO)C(O)=O MNYNCKZAEIAONY-XGEHTFHBSA-N 0.000 description 2
- BTAJAOWZCWOHBU-HSHDSVGOSA-N Thr-Val-Trp Chemical compound C1=CC=C2C(C[C@H](NC(=O)[C@@H](NC(=O)[C@@H](N)[C@@H](C)O)C(C)C)C(O)=O)=CNC2=C1 BTAJAOWZCWOHBU-HSHDSVGOSA-N 0.000 description 2
- BPGDJSUFQKWUBK-KJEVXHAQSA-N Thr-Val-Tyr Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 BPGDJSUFQKWUBK-KJEVXHAQSA-N 0.000 description 2
- VYVBSMCZNHOZGD-RCWTZXSCSA-N Thr-Val-Val Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C(C)C)C(O)=O VYVBSMCZNHOZGD-RCWTZXSCSA-N 0.000 description 2
- USXYVSTVPHELAF-RCWTZXSCSA-N Val-Thr-Met Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCSC)C(=O)O)NC(=O)[C@H](C(C)C)N)O USXYVSTVPHELAF-RCWTZXSCSA-N 0.000 description 2
- 239000003070 absorption delaying agent Substances 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 238000007792 addition Methods 0.000 description 2
- 150000001298 alcohols Chemical class 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 230000000844 anti-bacterial effect Effects 0.000 description 2
- 239000003429 antifungal agent Substances 0.000 description 2
- 229940121375 antifungal agent Drugs 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 108010059459 arginyl-threonyl-phenylalanine Proteins 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- 230000003115 biocidal effect Effects 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 238000003776 cleavage reaction Methods 0.000 description 2
- 238000010367 cloning Methods 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- GHVNFZFCNZKVNT-UHFFFAOYSA-M decanoate Chemical compound CCCCCCCCCC([O-])=O GHVNFZFCNZKVNT-UHFFFAOYSA-M 0.000 description 2
- 238000000326 densiometry Methods 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 239000008121 dextrose Substances 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- 239000002612 dispersion medium Substances 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 230000001747 exhibiting effect Effects 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 239000000834 fixative Substances 0.000 description 2
- 238000003205 genotyping method Methods 0.000 description 2
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 2
- 108010089804 glycyl-threonine Proteins 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 208000009326 ileitis Diseases 0.000 description 2
- 230000001976 improved effect Effects 0.000 description 2
- 230000001939 inductive effect Effects 0.000 description 2
- 230000000977 initiatory effect Effects 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 230000000527 lymphocytic effect Effects 0.000 description 2
- 102000039446 nucleic acids Human genes 0.000 description 2
- 108020004707 nucleic acids Proteins 0.000 description 2
- 150000007523 nucleic acids Chemical class 0.000 description 2
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 2
- 238000006384 oligomerization reaction Methods 0.000 description 2
- 210000002741 palatine tonsil Anatomy 0.000 description 2
- 239000008194 pharmaceutical composition Substances 0.000 description 2
- 239000000902 placebo Substances 0.000 description 2
- 229940068196 placebo Drugs 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 230000002028 premature Effects 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 208000017443 reproductive system disease Diseases 0.000 description 2
- 230000007017 scission Effects 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 238000010186 staining Methods 0.000 description 2
- 230000014616 translation Effects 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- JNYAEWCLZODPBN-KVTDHHQDSA-N (2r,3r,4r)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical compound OC[C@@H](O)[C@H]1OC[C@@H](O)[C@H]1O JNYAEWCLZODPBN-KVTDHHQDSA-N 0.000 description 1
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical compound OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 1
- YYGNTYWPHWGJRM-UHFFFAOYSA-N (6E,10E,14E,18E)-2,6,10,15,19,23-hexamethyltetracosa-2,6,10,14,18,22-hexaene Chemical compound CC(C)=CCCC(C)=CCCC(C)=CCCC=C(C)CCC=C(C)CCC=C(C)C YYGNTYWPHWGJRM-UHFFFAOYSA-N 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 1
- ULQISTXYYBZJSJ-UHFFFAOYSA-N 12-hydroxyoctadecanoic acid Chemical compound CCCCCCC(O)CCCCCCCCCCC(O)=O ULQISTXYYBZJSJ-UHFFFAOYSA-N 0.000 description 1
- RFIMISVNSAUMBU-UHFFFAOYSA-N 2-(hydroxymethyl)-2-(prop-2-enoxymethyl)propane-1,3-diol Chemical compound OCC(CO)(CO)COCC=C RFIMISVNSAUMBU-UHFFFAOYSA-N 0.000 description 1
- KIHBGTRZFAVZRV-UHFFFAOYSA-N 2-Hydroxyoctadecanoic acid Natural products CCCCCCCCCCCCCCCCC(O)C(O)=O KIHBGTRZFAVZRV-UHFFFAOYSA-N 0.000 description 1
- UMHYVXGZRGOICM-AUYXYSRISA-N 2-[(z)-octadec-9-enoyl]oxypropyl (z)-octadec-9-enoate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC(C)OC(=O)CCCCCCC\C=C/CCCCCCCC UMHYVXGZRGOICM-AUYXYSRISA-N 0.000 description 1
- WLAMNBDJUVNPJU-UHFFFAOYSA-N 2-methylbutyric acid Chemical compound CCC(C)C(O)=O WLAMNBDJUVNPJU-UHFFFAOYSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- QWVRTSZDKPRPDF-UHFFFAOYSA-N 5-(piperidin-1-ylmethyl)-3-pyridin-3-yl-5,6-dihydro-2h-1,2,4-oxadiazine Chemical compound C1CCCCN1CC(N=1)CONC=1C1=CC=CN=C1 QWVRTSZDKPRPDF-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- 102220553675 APC membrane recruitment protein 1_K59A_mutation Human genes 0.000 description 1
- 108010042708 Acetylmuramyl-Alanyl-Isoglutamine Proteins 0.000 description 1
- 102000009027 Albumins Human genes 0.000 description 1
- 108010088751 Albumins Proteins 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- ATABBWFGOHKROJ-GUBZILKMSA-N Arg-Pro-Ser Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(O)=O ATABBWFGOHKROJ-GUBZILKMSA-N 0.000 description 1
- JWKDQOORUCYUIW-ZPFDUUQYSA-N Asn-Lys-Ile Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)CC)C(O)=O JWKDQOORUCYUIW-ZPFDUUQYSA-N 0.000 description 1
- GADKFYNESXNRLC-WDSKDSINSA-N Asn-Pro Chemical compound NC(=O)C[C@H](N)C(=O)N1CCC[C@H]1C(O)=O GADKFYNESXNRLC-WDSKDSINSA-N 0.000 description 1
- QXOPPIDJKPEKCW-GUBZILKMSA-N Asn-Pro-Arg Chemical compound C1C[C@H](N(C1)C(=O)[C@H](CC(=O)N)N)C(=O)N[C@@H](CCCN=C(N)N)C(=O)O QXOPPIDJKPEKCW-GUBZILKMSA-N 0.000 description 1
- SVFOIXMRMLROHO-SRVKXCTJSA-N Asp-Asp-Phe Chemical compound OC(=O)C[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 SVFOIXMRMLROHO-SRVKXCTJSA-N 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 1
- NOWKCMXCCJGMRR-UHFFFAOYSA-N Aziridine Chemical compound C1CN1 NOWKCMXCCJGMRR-UHFFFAOYSA-N 0.000 description 1
- 102000004506 Blood Proteins Human genes 0.000 description 1
- 108010017384 Blood Proteins Proteins 0.000 description 1
- 201000004813 Bronchopneumonia Diseases 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 108090000565 Capsid Proteins Proteins 0.000 description 1
- 102100023321 Ceruloplasmin Human genes 0.000 description 1
- 241001227713 Chiron Species 0.000 description 1
- 102000009016 Cholera Toxin Human genes 0.000 description 1
- 108010049048 Cholera Toxin Proteins 0.000 description 1
- 108020004638 Circular DNA Proteins 0.000 description 1
- 102220527008 Cytochrome b5 reductase 4_R63Q_mutation Human genes 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 206010012735 Diarrhoea Diseases 0.000 description 1
- 206010013883 Dwarfism Diseases 0.000 description 1
- 208000000059 Dyspnea Diseases 0.000 description 1
- 206010013975 Dyspnoeas Diseases 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical class OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- LVGKNOAMLMIIKO-UHFFFAOYSA-N Elaidinsaeure-aethylester Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC LVGKNOAMLMIIKO-UHFFFAOYSA-N 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- CEAZRRDELHUEMR-URQXQFDESA-N Gentamicin Chemical compound O1[C@H](C(C)NC)CC[C@@H](N)[C@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](NC)[C@@](C)(O)CO2)O)[C@H](N)C[C@@H]1N CEAZRRDELHUEMR-URQXQFDESA-N 0.000 description 1
- 229930182566 Gentamicin Natural products 0.000 description 1
- JHPFPROFOAJRFN-IHRRRGAJSA-N Gln-Glu-Tyr Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)O)NC(=O)[C@H](CCC(=O)O)NC(=O)[C@H](CCC(=O)N)N)O JHPFPROFOAJRFN-IHRRRGAJSA-N 0.000 description 1
- FTIJVMLAGRAYMJ-MNXVOIDGSA-N Gln-Ile-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H]([C@@H](C)CC)NC(=O)[C@@H](N)CCC(N)=O FTIJVMLAGRAYMJ-MNXVOIDGSA-N 0.000 description 1
- UQJNXZSSGQIPIQ-FBCQKBJTSA-N Gly-Gly-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)CNC(=O)CN UQJNXZSSGQIPIQ-FBCQKBJTSA-N 0.000 description 1
- IALQAMYQJBZNSK-WHFBIAKZSA-N Gly-Ser-Asn Chemical compound [H]NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(O)=O IALQAMYQJBZNSK-WHFBIAKZSA-N 0.000 description 1
- WCORRBXVISTKQL-WHFBIAKZSA-N Gly-Ser-Ser Chemical compound NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(O)=O WCORRBXVISTKQL-WHFBIAKZSA-N 0.000 description 1
- AIPUZFXMXAHZKY-QWRGUYRKSA-N His-Leu-Gly Chemical compound [H]N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CC(C)C)C(=O)NCC(O)=O AIPUZFXMXAHZKY-QWRGUYRKSA-N 0.000 description 1
- 206010020565 Hyperaemia Diseases 0.000 description 1
- OTSVBELRDMSPKY-PCBIJLKTSA-N Ile-Phe-Asn Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CC(=O)N)C(=O)O)N OTSVBELRDMSPKY-PCBIJLKTSA-N 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- 102100027612 Kallikrein-11 Human genes 0.000 description 1
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 1
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 1
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- OGUUKPXUTHOIAV-SDDRHHMPSA-N Leu-Glu-Pro Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CCC(=O)O)C(=O)N1CCC[C@@H]1C(=O)O)N OGUUKPXUTHOIAV-SDDRHHMPSA-N 0.000 description 1
- RZXLZBIUTDQHJQ-SRVKXCTJSA-N Leu-Lys-Asp Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(O)=O RZXLZBIUTDQHJQ-SRVKXCTJSA-N 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- HQVDJTYKCMIWJP-YUMQZZPRSA-N Lys-Asn-Gly Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(O)=O HQVDJTYKCMIWJP-YUMQZZPRSA-N 0.000 description 1
- 101710141347 Major envelope glycoprotein Proteins 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 206010027256 Meningitis streptococcal Diseases 0.000 description 1
- 241000204045 Mycoplasma hyopneumoniae Species 0.000 description 1
- 125000001429 N-terminal alpha-amino-acid group Chemical group 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 239000005662 Paraffin oil Substances 0.000 description 1
- 102000035195 Peptidases Human genes 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- SKICPQLTOXGWGO-GARJFASQSA-N Pro-Gln-Pro Chemical compound C1C[C@H](NC1)C(=O)N[C@@H](CCC(=O)N)C(=O)N2CCC[C@@H]2C(=O)O SKICPQLTOXGWGO-GARJFASQSA-N 0.000 description 1
- HAAQQNHQZBOWFO-LURJTMIESA-N Pro-Gly-Gly Chemical compound OC(=O)CNC(=O)CNC(=O)[C@@H]1CCCN1 HAAQQNHQZBOWFO-LURJTMIESA-N 0.000 description 1
- VTFXTWDFPTWNJY-RHYQMDGZSA-N Pro-Leu-Thr Chemical compound [H]N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(O)=O VTFXTWDFPTWNJY-RHYQMDGZSA-N 0.000 description 1
- CGSOWZUPLOKYOR-AVGNSLFASA-N Pro-Pro-Leu Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H]1NCCC1 CGSOWZUPLOKYOR-AVGNSLFASA-N 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 208000006311 Pyoderma Diseases 0.000 description 1
- 238000011529 RT qPCR Methods 0.000 description 1
- 108010073443 Ribi adjuvant Proteins 0.000 description 1
- 206010039438 Salmonella Infections Diseases 0.000 description 1
- MOINZPRHJGTCHZ-MMWGEVLESA-N Ser-Ile-Pro Chemical compound CC[C@H](C)[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CO)N MOINZPRHJGTCHZ-MMWGEVLESA-N 0.000 description 1
- QJKPECIAWNNKIT-KKUMJFAQSA-N Ser-Lys-Tyr Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O QJKPECIAWNNKIT-KKUMJFAQSA-N 0.000 description 1
- ANOQEBQWIAYIMV-AEJSXWLSSA-N Ser-Val-Pro Chemical compound CC(C)[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CO)N ANOQEBQWIAYIMV-AEJSXWLSSA-N 0.000 description 1
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 1
- 241000256251 Spodoptera frugiperda Species 0.000 description 1
- 230000024932 T cell mediated immunity Effects 0.000 description 1
- BHEOSNUKNHRBNM-UHFFFAOYSA-N Tetramethylsqualene Natural products CC(=C)C(C)CCC(=C)C(C)CCC(C)=CCCC=C(C)CCC(C)C(=C)CCC(C)C(C)=C BHEOSNUKNHRBNM-UHFFFAOYSA-N 0.000 description 1
- TYVAWPFQYFPSBR-BFHQHQDPSA-N Thr-Ala-Gly Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)NCC(O)=O TYVAWPFQYFPSBR-BFHQHQDPSA-N 0.000 description 1
- PRTHQBSMXILLPC-XGEHTFHBSA-N Thr-Ser-Arg Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O PRTHQBSMXILLPC-XGEHTFHBSA-N 0.000 description 1
- LECUEEHKUFYOOV-ZJDVBMNYSA-N Thr-Thr-Val Chemical compound CC(C)[C@@H](C(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@@H](N)[C@@H](C)O LECUEEHKUFYOOV-ZJDVBMNYSA-N 0.000 description 1
- XGFYGMKZKFRGAI-RCWTZXSCSA-N Thr-Val-Arg Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@H](C(O)=O)CCCN=C(N)N XGFYGMKZKFRGAI-RCWTZXSCSA-N 0.000 description 1
- 101710152431 Trypsin-like protease Proteins 0.000 description 1
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 1
- NZBSVMQZQMEUHI-WZLNRYEVSA-N Tyr-Thr-Ile Chemical compound CC[C@H](C)[C@@H](C(=O)O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC1=CC=C(C=C1)O)N NZBSVMQZQMEUHI-WZLNRYEVSA-N 0.000 description 1
- YMTOEGGOCHVGEH-IHRRRGAJSA-N Val-Lys-Lys Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(O)=O YMTOEGGOCHVGEH-IHRRRGAJSA-N 0.000 description 1
- UZQJVUCHXGYFLQ-AYDHOLPZSA-N [(2s,3r,4s,5r,6r)-4-[(2s,3r,4s,5r,6r)-4-[(2r,3r,4s,5r,6r)-4-[(2s,3r,4s,5r,6r)-3,5-dihydroxy-6-(hydroxymethyl)-4-[(2s,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoxan-2-yl]oxy-3,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-3,5-dihydroxy-6-(hy Chemical compound O([C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O)O[C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O)O[C@H]1CC[C@]2(C)[C@H]3CC=C4[C@@]([C@@]3(CC[C@H]2[C@@]1(C=O)C)C)(C)CC(O)[C@]1(CCC(CC14)(C)C)C(=O)O[C@H]1[C@@H]([C@@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O[C@H]4[C@@H]([C@@H](O[C@H]5[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O5)O)[C@H](O)[C@@H](CO)O4)O)[C@H](O)[C@@H](CO)O3)O)[C@H](O)[C@@H](CO)O2)O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O UZQJVUCHXGYFLQ-AYDHOLPZSA-N 0.000 description 1
- 230000021736 acetylation Effects 0.000 description 1
- 238000006640 acetylation reaction Methods 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 238000007605 air drying Methods 0.000 description 1
- 150000001447 alkali salts Chemical class 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 229940086737 allyl sucrose Drugs 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- ILRRQNADMUWWFW-UHFFFAOYSA-K aluminium phosphate Chemical compound O1[Al]2OP1(=O)O2 ILRRQNADMUWWFW-UHFFFAOYSA-K 0.000 description 1
- 230000000845 anti-microbial effect Effects 0.000 description 1
- 230000000890 antigenic effect Effects 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 229960001230 asparagine Drugs 0.000 description 1
- 235000009582 asparagine Nutrition 0.000 description 1
- 229940009098 aspartate Drugs 0.000 description 1
- 108010093581 aspartyl-proline Proteins 0.000 description 1
- 238000011888 autopsy Methods 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 229960001631 carbomer Drugs 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 230000003833 cell viability Effects 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000012761 co-transfection Methods 0.000 description 1
- 230000035071 co-translational protein modification Effects 0.000 description 1
- 229940001442 combination vaccine Drugs 0.000 description 1
- 238000004590 computer program Methods 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- 210000002808 connective tissue Anatomy 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 239000012228 culture supernatant Substances 0.000 description 1
- 230000009089 cytolysis Effects 0.000 description 1
- 210000001151 cytotoxic T lymphocyte Anatomy 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 239000002158 endotoxin Substances 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 210000000981 epithelium Anatomy 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- LVGKNOAMLMIIKO-QXMHVHEDSA-N ethyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC LVGKNOAMLMIIKO-QXMHVHEDSA-N 0.000 description 1
- 229940093471 ethyl oleate Drugs 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000013613 expression plasmid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- MHMNJMPURVTYEJ-UHFFFAOYSA-N fluorescein-5-isothiocyanate Chemical compound O1C(=O)C2=CC(N=C=S)=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 MHMNJMPURVTYEJ-UHFFFAOYSA-N 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 229960002518 gentamicin Drugs 0.000 description 1
- 125000003976 glyceryl group Chemical group [H]C([*])([H])C(O[H])([H])C(O[H])([H])[H] 0.000 description 1
- 150000002334 glycols Chemical class 0.000 description 1
- 230000013595 glycosylation Effects 0.000 description 1
- 238000006206 glycosylation reaction Methods 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000002744 homologous recombination Methods 0.000 description 1
- 230000006801 homologous recombination Effects 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 230000002519 immonomodulatory effect Effects 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 238000003018 immunoassay Methods 0.000 description 1
- 238000010166 immunofluorescence Methods 0.000 description 1
- 230000017555 immunoglobulin mediated immune response Effects 0.000 description 1
- 230000003308 immunostimulating effect Effects 0.000 description 1
- 230000001506 immunosuppresive effect Effects 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 229940047124 interferons Drugs 0.000 description 1
- 229940047122 interleukins Drugs 0.000 description 1
- 238000001361 intraarterial administration Methods 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000007913 intrathecal administration Methods 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 229960000310 isoleucine Drugs 0.000 description 1
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 230000000366 juvenile effect Effects 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 235000020997 lean meat Nutrition 0.000 description 1
- 229940057995 liquid paraffin Drugs 0.000 description 1
- 208000019423 liver disease Diseases 0.000 description 1
- 210000005210 lymphoid organ Anatomy 0.000 description 1
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 238000000386 microscopy Methods 0.000 description 1
- 238000007479 molecular analysis Methods 0.000 description 1
- 238000010369 molecular cloning Methods 0.000 description 1
- 229940035032 monophosphoryl lipid a Drugs 0.000 description 1
- BSOQXXWZTUDTEL-ZUYCGGNHSA-N muramyl dipeptide Chemical compound OC(=O)CC[C@H](C(N)=O)NC(=O)[C@H](C)NC(=O)[C@@H](C)O[C@H]1[C@H](O)[C@@H](CO)O[C@@H](O)[C@@H]1NC(C)=O BSOQXXWZTUDTEL-ZUYCGGNHSA-N 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- JXTPJDDICSTXJX-UHFFFAOYSA-N n-Triacontane Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCC JXTPJDDICSTXJX-UHFFFAOYSA-N 0.000 description 1
- AFFLGGQVNFXPEV-UHFFFAOYSA-N n-decene Natural products CCCCCCCCC=C AFFLGGQVNFXPEV-UHFFFAOYSA-N 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- 239000007764 o/w emulsion Substances 0.000 description 1
- 229940049964 oleate Drugs 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 230000009340 pathogen transmission Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 230000002688 persistence Effects 0.000 description 1
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 1
- IYDGMDWEHDFVQI-UHFFFAOYSA-N phosphoric acid;trioxotungsten Chemical compound O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.OP(O)(O)=O IYDGMDWEHDFVQI-UHFFFAOYSA-N 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 229920001983 poloxamer Polymers 0.000 description 1
- 229920000058 polyacrylate Polymers 0.000 description 1
- 229920000223 polyglycerol Polymers 0.000 description 1
- 230000004481 post-translational protein modification Effects 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 108010020755 prolyl-glycyl-glycine Proteins 0.000 description 1
- 229940010310 propylene glycol dioleate Drugs 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 230000020978 protein processing Effects 0.000 description 1
- 239000012460 protein solution Substances 0.000 description 1
- 238000001243 protein synthesis Methods 0.000 description 1
- 208000009305 pseudorabies Diseases 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 239000013014 purified material Substances 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000003259 recombinant expression Methods 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- WBHHMMIMDMUBKC-XLNAKTSKSA-N ricinelaidic acid Chemical compound CCCCCC[C@@H](O)C\C=C\CCCCCCCC(O)=O WBHHMMIMDMUBKC-XLNAKTSKSA-N 0.000 description 1
- 229960003656 ricinoleic acid Drugs 0.000 description 1
- FEUQNCSVHBHROZ-UHFFFAOYSA-N ricinoleic acid Natural products CCCCCCC(O[Si](C)(C)C)CC=CCCCCCCCC(=O)OC FEUQNCSVHBHROZ-UHFFFAOYSA-N 0.000 description 1
- 102200104785 rs864309491 Human genes 0.000 description 1
- 206010039447 salmonellosis Diseases 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 229930182490 saponin Natural products 0.000 description 1
- 150000007949 saponins Chemical class 0.000 description 1
- 235000017709 saponins Nutrition 0.000 description 1
- 238000001338 self-assembly Methods 0.000 description 1
- 230000000405 serological effect Effects 0.000 description 1
- 108010048818 seryl-histidine Proteins 0.000 description 1
- 230000001568 sexual effect Effects 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 238000003307 slaughter Methods 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 229940032094 squalane Drugs 0.000 description 1
- 229940031439 squalene Drugs 0.000 description 1
- TUHBEKDERLKLEC-UHFFFAOYSA-N squalene Natural products CC(=CCCC(=CCCC(=CCCC=C(/C)CCC=C(/C)CC=C(C)C)C)C)C TUHBEKDERLKLEC-UHFFFAOYSA-N 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 239000008174 sterile solution Substances 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 201000001739 streptococcal meningitis Diseases 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 150000003505 terpenes Chemical class 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- RTKIYNMVFMVABJ-UHFFFAOYSA-L thimerosal Chemical compound [Na+].CC[Hg]SC1=CC=CC=C1C([O-])=O RTKIYNMVFMVABJ-UHFFFAOYSA-L 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 238000001890 transfection Methods 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 239000004474 valine Substances 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 230000007485 viral shedding Effects 0.000 description 1
- 239000007762 w/o emulsion Substances 0.000 description 1
- 239000011534 wash buffer Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/005—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/12—Viral antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/20—Antivirals for DNA viruses
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N7/00—Viruses; Bacteriophages; Compositions thereof; Preparation or purification thereof
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P21/00—Preparation of peptides or proteins
- C12P21/02—Preparation of peptides or proteins having a known sequence of two or more amino acids, e.g. glutathione
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/525—Virus
- A61K2039/5256—Virus expressing foreign proteins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/525—Virus
- A61K2039/5258—Virus-like particles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/53—DNA (RNA) vaccination
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2710/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA dsDNA viruses
- C12N2710/00011—Details
- C12N2710/14011—Baculoviridae
- C12N2710/14041—Use of virus, viral particle or viral elements as a vector
- C12N2710/14043—Use of virus, viral particle or viral elements as a vector viral genome or elements thereof as genetic vectore
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/10011—Circoviridae
- C12N2750/10021—Viruses as such, e.g. new isolates, mutants or their genomic sequences
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/10011—Circoviridae
- C12N2750/10022—New viral proteins or individual genes, new structural or functional aspects of known viral proteins or genes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/10011—Circoviridae
- C12N2750/10023—Virus like particles [VLP]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/10011—Circoviridae
- C12N2750/10034—Use of virus or viral component as vaccine, e.g. live-attenuated or inactivated virus, VLP, viral protein
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/10011—Circoviridae
- C12N2750/10051—Methods of production or purification of viral material
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2799/00—Uses of viruses
- C12N2799/02—Uses of viruses as vector
- C12N2799/021—Uses of viruses as vector for the expression of a heterologous nucleic acid
- C12N2799/026—Uses of viruses as vector for the expression of a heterologous nucleic acid where the vector is derived from a baculovirus
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Virology (AREA)
- Medicinal Chemistry (AREA)
- Genetics & Genomics (AREA)
- Engineering & Computer Science (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Immunology (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biotechnology (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- General Engineering & Computer Science (AREA)
- Epidemiology (AREA)
- Mycology (AREA)
- Biophysics (AREA)
- Gastroenterology & Hepatology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biomedical Technology (AREA)
- Communicable Diseases (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Oncology (AREA)
- Peptides Or Proteins (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361885871P | 2013-10-02 | 2013-10-02 | |
| US61/885,871 | 2013-10-02 | ||
| KR1020167011660A KR102409183B1 (ko) | 2013-10-02 | 2014-10-02 | Pcv2 orf2 단백질 변이체 및 이로 이루어진 바이러스 유사 입자 |
| PCT/US2014/058793 WO2015051099A1 (en) | 2013-10-02 | 2014-10-02 | Pcv2 orf2 protein variant and virus like particles composed thereof |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020167011660A Division KR102409183B1 (ko) | 2013-10-02 | 2014-10-02 | Pcv2 orf2 단백질 변이체 및 이로 이루어진 바이러스 유사 입자 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20220083861A true KR20220083861A (ko) | 2022-06-20 |
Family
ID=51795768
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020227019654A Ceased KR20220083861A (ko) | 2013-10-02 | 2014-10-02 | Pcv2 orf2 단백질 변이체 및 이로 이루어진 바이러스 유사 입자 |
| KR1020167011660A Active KR102409183B1 (ko) | 2013-10-02 | 2014-10-02 | Pcv2 orf2 단백질 변이체 및 이로 이루어진 바이러스 유사 입자 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020167011660A Active KR102409183B1 (ko) | 2013-10-02 | 2014-10-02 | Pcv2 orf2 단백질 변이체 및 이로 이루어진 바이러스 유사 입자 |
Country Status (12)
| Country | Link |
|---|---|
| US (6) | US9505808B2 (enExample) |
| EP (1) | EP3052516B1 (enExample) |
| JP (1) | JP6778104B2 (enExample) |
| KR (2) | KR20220083861A (enExample) |
| CN (1) | CN105658660A (enExample) |
| AU (1) | AU2014329524B2 (enExample) |
| CA (1) | CA2924228C (enExample) |
| DK (1) | DK3052516T3 (enExample) |
| EA (1) | EA035265B1 (enExample) |
| ES (1) | ES2778425T3 (enExample) |
| MX (1) | MX373884B (enExample) |
| WO (1) | WO2015051099A1 (enExample) |
Families Citing this family (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| UA95602C2 (ru) | 2004-12-30 | 2011-08-25 | Берингер Ингельхейм Ветмедика, Инк. | Иммуногенная композиция цвс2 и способы приготовления такой композиции |
| DK2371383T3 (en) | 2005-12-29 | 2015-11-30 | Boehringer Ingelheim Vetmed | Use of a PCV2 immunogenic composition for reduction of clinical symptoms in pigs |
| RU2488407C2 (ru) | 2005-12-29 | 2013-07-27 | Берингер Ингельхайм Ветмедика, Инк. | Поливалентные иммуногенные композиции pcv2 и способы получения таких композиций |
| US20100129397A1 (en) | 2006-12-11 | 2010-05-27 | Boehringer Ingelheim Vetmedica, Inc. | Effective method of treatment of porcine circovirus and lawsonia intracellularis infections |
| EP1941903A1 (en) | 2007-01-03 | 2008-07-09 | Boehringer Ingelheim Vetmedica Gmbh | Prophylaxis and treatment of PRDC |
| EP1958644A1 (en) | 2007-02-13 | 2008-08-20 | Boehringer Ingelheim Vetmedica Gmbh | Prevention and treatment of sub-clinical pcvd |
| EP2242511A4 (en) | 2008-01-23 | 2012-10-24 | Boehringer Ingelheim Vetmed | IMMUNOGENIC COMPOSITIONS OF MYCOPLASMA HYOPNEUMONIAE PCV2, AND METHODS FOR PRODUCING SUCH COMPOSITIONS |
| CN105658660A (zh) | 2013-10-02 | 2016-06-08 | 勃林格殷格翰动物保健公司 | Pcv2 orf2蛋白变体和由其组成的病毒样颗粒 |
| CN107531760A (zh) * | 2015-03-30 | 2018-01-02 | 勃林格殷格翰动物保健公司 | Pcv2 orf2载体平台 |
| HK1252427A1 (zh) | 2015-05-14 | 2019-05-24 | Boehringer Ingelheim Animal Health USA Inc. | 猪环状病毒生产方法和pcv2疫苗 |
| CN104984335A (zh) * | 2015-07-14 | 2015-10-21 | 浙江诺倍威生物技术有限公司 | 猪圆环病毒双亚型orf2共表达载体构建及疫苗制备 |
| WO2017187277A2 (en) * | 2016-03-07 | 2017-11-02 | Virginia Tech Intellectual Properties, Inc. | Chimeric porcine circovirus type 2 (pcv2) vaccines |
| US10431889B2 (en) * | 2016-04-22 | 2019-10-01 | The Charles Stark Draper Laboratory, Inc. | Low-loss compact transmit impedance matching tuning technique |
| CN106399350B (zh) * | 2016-09-07 | 2020-02-21 | 复旦大学 | 一种猪圆环病毒ii型病毒样颗粒疫苗及其制备方法 |
| CN106399138B (zh) * | 2016-09-07 | 2019-11-12 | 复旦大学 | 猪圆环病毒ii型口服病毒样颗粒疫苗及其制备方法和应用 |
| CN109803678B (zh) * | 2016-11-03 | 2023-08-22 | 勃林格殷格翰动物保健有限公司 | 抗猪细小病毒的疫苗 |
| CN106834242B (zh) * | 2017-03-02 | 2021-02-23 | 扬州大学 | 嵌合型猪圆环病毒活疫苗c1-233株及其构建方法 |
| CN106834241B (zh) * | 2017-03-02 | 2021-01-19 | 扬州大学 | 嵌合型猪圆环病毒活疫苗c1-402株及其构建方法 |
| CN107245477B (zh) * | 2017-06-06 | 2020-06-30 | 广东海大畜牧兽医研究院有限公司 | 一种猪圆环病毒2型病毒样颗粒的制备方法及通过该方法获得的病毒样颗粒 |
| PL3697804T3 (pl) * | 2017-10-17 | 2024-01-03 | Intervet International B.V. | Rekombinowana ekspresja białka orf2 pcv2b w komórkach owadzich |
| CN108484725A (zh) * | 2018-03-16 | 2018-09-04 | 广东海大畜牧兽医研究院有限公司 | 一种区分检测pcv2野毒抗体与疫苗抗体的多肽及其在elisa检测试剂盒中的应用 |
| CN110358742B (zh) * | 2019-07-05 | 2020-12-22 | 武汉科前生物股份有限公司 | 猪圆环病毒2b型和2d型的二价亚单位疫苗及其制备方法 |
| CN111415356B (zh) * | 2020-03-17 | 2020-12-29 | 推想医疗科技股份有限公司 | 肺炎征象的分割方法、装置、介质及电子设备 |
| CN112010949B (zh) * | 2020-08-03 | 2021-07-27 | 山东绿都生物科技有限公司 | 一种重组pcv2病毒样颗粒蜂胶疫苗及其制备方法 |
| CN114250259B (zh) * | 2020-09-21 | 2024-07-19 | 成都史纪生物制药有限公司 | 一种猪圆环2型病毒Cap蛋白病毒样颗粒的发酵培养基 |
| TWI867524B (zh) * | 2022-05-09 | 2024-12-21 | 福又達生物科技股份有限公司 | 豬環狀病毒第二型及豬瘟病毒次單位雙價疫苗 |
| AU2024265654A1 (en) | 2023-05-03 | 2025-12-04 | Boehringer Ingelheim Vetmedica Gmbh | Method for inducing an immune response |
| CN121013728A (zh) | 2023-05-03 | 2025-11-25 | 勃林格殷格翰动物保健有限公司 | 一种可用于猪自我施用的免疫原性组合物 |
| CN118063562B (zh) * | 2024-03-11 | 2025-04-01 | 天康生物制药有限公司 | 优化的猪圆环病毒2型Cap蛋白及其病毒样颗粒和应用 |
Family Cites Families (98)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2909462A (en) | 1955-12-08 | 1959-10-20 | Bristol Myers Co | Acrylic acid polymer laxative compositions |
| EP0283085B1 (en) | 1987-03-17 | 1992-11-11 | Akzo N.V. | Adjuvant mixture |
| US5069901A (en) | 1988-02-03 | 1991-12-03 | Jones Elaine V | Preparation of a recombinant subunit vaccine against pseudorabies infection |
| DE69027112T2 (de) | 1989-01-23 | 1997-01-09 | Auspharm International Ltd 4Th | Impfstoffzusammensetzung |
| US5155037A (en) | 1989-08-04 | 1992-10-13 | The Texas A&M University System | Insect signal sequences useful to improve the efficiency of processing and secretion of foreign genes in insect systems |
| US5202430A (en) | 1990-01-16 | 1993-04-13 | University Of Tennessee | Transmissible gastroenteritis virus genes |
| JP3187419B2 (ja) | 1990-05-29 | 2001-07-11 | アメリカン・サイアナミド・カンパニー | ブタ肺炎ワクチンおよびその製造方法 |
| US5565205A (en) | 1990-08-16 | 1996-10-15 | Solvay Animal Health, Inc. | Inactivated Mycoplasma hypopneumoniae bacterin and method of use thereof |
| GB9023111D0 (en) | 1990-10-24 | 1990-12-05 | Wellcome Found | Expression system |
| WO1992007934A1 (en) | 1990-11-01 | 1992-05-14 | Iowa State University Research Foundation, Inc. | Bacterial attenuation method and vaccine |
| MX9204885A (es) | 1991-08-26 | 1994-05-31 | Boehringer Animal Health Inc | Composicion de vacuna que comprende virus del sindrome de infertilidad y respiratorio de los cerdos. |
| US6042830A (en) | 1992-08-05 | 2000-03-28 | Boehringer Ingelheim Vetmedica, Inc. | Viral agent associated with mystery swine disease |
| US5580557A (en) | 1991-10-09 | 1996-12-03 | Iowa State University Research Foundation, Inc. | Live, avirulent salmonella choleraesuis vaccine used for inducing an immune response in animals |
| US5338543A (en) | 1992-02-27 | 1994-08-16 | Ambico, Inc. | Thimerosal inactivated mycoplasma hyopneumoniae vaccine |
| CN1268393C (zh) | 1994-05-10 | 2006-08-09 | 惠氏公司 | 经改良修饰的brsv或疫苗 |
| US5885823A (en) | 1995-06-05 | 1999-03-23 | Nobl Laboratories, Inc. | Lawsonia intracellularis cultivation, anti-Lawsonia intracellularis vaccines and diagnostic agents |
| WO1998006835A2 (en) | 1996-08-16 | 1998-02-19 | The Texas A & M University System | Modifying insect cell gylcosylation pathways with baculovirus expression vectors |
| DK0835930T3 (da) | 1996-10-09 | 2001-06-18 | Akzo Nobel Nv | Europæiske vaccinestammer af porcint reproduktions- og respirationssyndrom-virus. (PRRSV) |
| US6517843B1 (en) | 1999-08-31 | 2003-02-11 | Merial | Reduction of porcine circovirus-2 viral load with inactivated PCV-2 |
| FR2781159B1 (fr) | 1998-07-06 | 2000-10-06 | Merial Sas | Vaccin circovirus et parvovirus porcin |
| US7192594B2 (en) | 1997-10-03 | 2007-03-20 | Merial Limited | Postweaning multisystemic wasting syndrome and porcine circovirus from pigs |
| UA78180C2 (uk) | 1997-10-03 | 2007-03-15 | Меріаль | Кільцевий вірус свині типу ii, вакцини та діагностичні реагенти |
| US6391314B1 (en) | 1997-10-03 | 2002-05-21 | Merial | Porcine circoviruses vaccines diagnostic reagents |
| US7211379B2 (en) | 1997-10-03 | 2007-05-01 | Merial Sas | Prevention of myocarditis, abortion and intrauterine infection associated with porcine circovirus-2 |
| US20060029617A1 (en) | 1997-10-03 | 2006-02-09 | Charreyre Catherine E | Porcine circovirus and Helicobacter combination vaccines and methods of use |
| FR2772047B1 (fr) | 1997-12-05 | 2004-04-09 | Ct Nat D Etudes Veterinaires E | Sequence genomique et polypeptides de circovirus associe a la maladie de l'amaigrissement du porcelet (map), applications au diagnostic et a la prevention et/ou au traitement de l'infection |
| US20040062775A1 (en) | 1997-12-05 | 2004-04-01 | Agence Francaise De Securite Sanitaire Des Aliments | Circovirus sequences associated with piglet weight loss disease (PWD) |
| ES2289796T3 (es) | 1997-12-11 | 2008-02-01 | University Of Saskatchewan | Virus del sindrome del desmedro post-destete procedente de cerdos. |
| EP1064024A4 (en) | 1998-03-13 | 2005-04-06 | Univ Georgia Res Found | VACCINES AGAINST CIRCOVIRUS INFECTIONS |
| EP0953574A1 (en) * | 1998-04-30 | 1999-11-03 | Innogenetics N.V. | Immunodiagnostic reagent for the detection of Maedi-Visna virus and CAEV infection |
| US6294176B1 (en) | 1998-07-10 | 2001-09-25 | Schering-Plough Veterinary Corp. | Recombinant raccoonpox virus and uses thereof as a vaccine in mammalian and avian species |
| DE69933647T2 (de) | 1998-11-19 | 2007-08-23 | Azwell Inc. | Rekombinante lysophosphatidsäure - phosphatase. |
| FR2789695B1 (fr) | 1999-02-11 | 2003-03-07 | Merial Sas | Vecteurs et vaccins viraux a base d'adenovirus porcins recombines et replicatifs |
| US6943152B1 (en) | 1999-06-10 | 2005-09-13 | Merial | DNA vaccine-PCV |
| US6497883B1 (en) | 1999-06-10 | 2002-12-24 | Merial | Porcine circovirus recombinant poxvirus vaccine |
| WO2001017556A1 (fr) | 1999-09-07 | 2001-03-15 | Shionogi & Co., Ltd. | Préparations vaccinales administrables par les muqueuses |
| US6656478B1 (en) | 1999-11-12 | 2003-12-02 | Samuel D. Charles | Cross-protective salmonella vaccines |
| ATE385808T1 (de) | 1999-12-21 | 2008-03-15 | Merial Sas | Mindestens ein cryptosporidium parvum-antigen und mindestens ein antigen eines anderen darmkrankheitserregers enthaltende zusammensetzungen und impfstoffe |
| US6808900B2 (en) | 2000-06-15 | 2004-10-26 | Manitoba, University Of | Cryptosporidium parvum antigens, antibodies thereto and diagnostic and therapeutic compositions thereof |
| MXPA02012352A (es) | 2000-06-15 | 2004-09-06 | Purdue Research Foundation | Vacuna para temblores congenitos en cerdos. |
| MY129765A (en) | 2000-12-19 | 2007-04-30 | Wyeth Corp | Improved mycoplasma hyopneumoniae bacterin vaccine |
| US7018638B2 (en) | 2000-12-19 | 2006-03-28 | Wyeth | Mycoplasma hyopneumoniae bacterin vaccine |
| JP2002247979A (ja) | 2001-02-23 | 2002-09-03 | Nippon Biologicals Inc | マレック病ワクチンおよびその製造方法 |
| SK287969B6 (sk) | 2001-03-27 | 2012-08-06 | University Of Saskatchewan | Method to culture circovirus |
| US20030096377A1 (en) | 2001-06-28 | 2003-05-22 | Virginia Tech Intellectual Properties, Inc. | Differential PCR-RFLP assay for detecting and distinguishing between nonpathogenic PCV-1 and pathogenic PCV-2 |
| CN102872456B (zh) | 2001-07-02 | 2016-01-20 | 硕腾服务有限责任公司 | 单剂量猪肺炎支原体疫苗 |
| US7361352B2 (en) | 2001-08-15 | 2008-04-22 | Acambis, Inc. | Influenza immunogen and vaccine |
| US7279166B2 (en) | 2001-12-12 | 2007-10-09 | Virginia Tech Intellectual Properties, Inc. | Chimeric infectious DNA clones, chimeric porcine circoviruses and uses thereof |
| US7276353B2 (en) | 2001-12-12 | 2007-10-02 | Virginia Tech Intellectual Properties, Inc. | Chimeric infectious DNA clones, chimeric porcine circoviruses and uses thereof |
| US6841364B2 (en) | 2002-01-22 | 2005-01-11 | Protatek International, Inc. | Infectious cDNA clones of porcine reproductive and respiratory syndrome virus and expression vectors thereof |
| AU2003217413A1 (en) | 2002-02-14 | 2003-09-04 | Novavax, Inc. | Method for isolation and purification of expressed gene products in vitro |
| AU2003220240A1 (en) | 2002-03-13 | 2003-09-29 | Biophysica, Inc. | Boswellin compositions enhanced with 3-beta-acety1-11-keto-beta-boswellic acid (akba), industrial manufacture and their uses |
| US20030215455A1 (en) | 2002-05-14 | 2003-11-20 | Bailey Reynolds | Vaccine stabilizer and method of use |
| AU2002951692A0 (en) | 2002-09-23 | 2002-10-17 | Vital Biotech (Hong Kong) Limited | Improvements in or relating to vaccines |
| MXPA05008252A (es) | 2003-02-03 | 2006-05-22 | Cerebus Biolog Inc | Metodos para tratar, prevenir y detectar una infeccion con helicobacter. |
| US7563449B2 (en) | 2003-04-21 | 2009-07-21 | Pfizer Inc, | Methods for reducing cattle reproductive diseases |
| CN1458167A (zh) | 2003-06-02 | 2003-11-26 | 中国农业科学院哈尔滨兽医研究所 | 截短表达的猪圆环病毒ⅱ型衣壳蛋白抗原及其应用 |
| US7335361B2 (en) | 2003-06-09 | 2008-02-26 | Animal Technology Institute Taiwan | Fusion antigen used as vaccine |
| UA84024C2 (ru) | 2003-07-24 | 2008-09-10 | Мериал Лимитед | Новые вакцинные композиции, которые содержат эмульсию типа "масло в воде" |
| JP2006528882A (ja) | 2003-07-25 | 2006-12-28 | ベーリンガー インゲルハイム フェトメディカ インコーポレイテッド | 欧州起源のローソニア・イントラセルラリス並びにそのワクチン、診断薬及び使用方法 |
| FR2861731B1 (fr) | 2003-10-30 | 2006-02-24 | Centre Nat Rech Scient | Nouvelle proteine de fixation du phosphate, compositions pharmaceutiques la contenant et ses utilisations |
| WO2005112995A1 (en) | 2004-04-26 | 2005-12-01 | Choongang Vaccine Laboratory Co., Ltd. | Inactivated mixed vaccine for porcine respiratory disease and the method of manufacturing thereof |
| CN1579553A (zh) | 2004-05-18 | 2005-02-16 | 浙江大学 | Ii型猪圆环病毒核酸疫苗的制备方法及其应用 |
| AU2005319716A1 (en) | 2004-06-30 | 2006-06-29 | Id Biomedical Corporation Of Quebec | Vaccine compositions for treating coronavirus infection |
| US7700285B1 (en) | 2005-12-29 | 2010-04-20 | Boehringer Ingelheim Vetmedica, Inc. | PCV2 immunogenic compositions and methods of producing such compositions |
| US7833707B2 (en) | 2004-12-30 | 2010-11-16 | Boehringer Ingelheim Vetmedica, Inc. | Methods of overexpression and recovery of porcine circovirus type 2 ORF2 |
| EP1838343B1 (en) | 2005-01-13 | 2010-03-31 | Boehringer Ingelheim Vetmedica Gmbh | Prrs vaccines |
| US7300785B2 (en) | 2005-02-03 | 2007-11-27 | Universiteit Ghent | Culturing circular ssDNA viruses for the production of vaccines |
| US8834891B2 (en) | 2005-03-14 | 2014-09-16 | Boehringer Ingelheim Vetmedica, Inc. | Immunogenic compositions comprising Lawsonia intracellularis |
| PT1869471T (pt) | 2005-04-13 | 2016-08-16 | Merial Inc | Ensaio para a produção de circovírus suíno |
| US7691368B2 (en) | 2005-04-15 | 2010-04-06 | Merial Limited | Vaccine formulations |
| RU2389506C2 (ru) | 2005-09-09 | 2010-05-20 | Интервет Интернэшнл Б.В. | Вакцина против рсv-2 |
| RU2488407C2 (ru) | 2005-12-29 | 2013-07-27 | Берингер Ингельхайм Ветмедика, Инк. | Поливалентные иммуногенные композиции pcv2 и способы получения таких композиций |
| DK2371383T3 (en) | 2005-12-29 | 2015-11-30 | Boehringer Ingelheim Vetmed | Use of a PCV2 immunogenic composition for reduction of clinical symptoms in pigs |
| EP2099927A4 (en) | 2006-11-22 | 2010-05-05 | Boehringer Ingelheim Vetmed | Methods of reducing porcine circovirus-associated disease outbreaks |
| US20100129397A1 (en) | 2006-12-11 | 2010-05-27 | Boehringer Ingelheim Vetmedica, Inc. | Effective method of treatment of porcine circovirus and lawsonia intracellularis infections |
| DK2481420T3 (da) * | 2006-12-15 | 2019-05-06 | Boehringer Ingelheim Animal Health Usa Inc | Enkeltdosis-anti-PCV2-svinevaccine |
| EP1941903A1 (en) | 2007-01-03 | 2008-07-09 | Boehringer Ingelheim Vetmedica Gmbh | Prophylaxis and treatment of PRDC |
| EP1958644A1 (en) | 2007-02-13 | 2008-08-20 | Boehringer Ingelheim Vetmedica Gmbh | Prevention and treatment of sub-clinical pcvd |
| GB0712160D0 (en) * | 2007-06-22 | 2007-08-01 | Univ Ghent | Methods and compositions in the treatment of procine circoviral infection |
| US20090017064A1 (en) | 2007-07-10 | 2009-01-15 | Wyeth | Methods and Compositions for Immunizing Pigs Against Porcine Circovirus |
| US7829274B2 (en) | 2007-09-04 | 2010-11-09 | Boehringer Ingelheim Vetmedica, Inc. | Reduction of concomitant infections in pigs by the use of PCV2 antigen |
| AU2008343172B2 (en) | 2007-12-21 | 2015-02-19 | Zoetis Services Llc | Methods and compositions for immunizing pigs against porcine circovirus |
| EP2242511A4 (en) | 2008-01-23 | 2012-10-24 | Boehringer Ingelheim Vetmed | IMMUNOGENIC COMPOSITIONS OF MYCOPLASMA HYOPNEUMONIAE PCV2, AND METHODS FOR PRODUCING SUCH COMPOSITIONS |
| WO2009103037A1 (en) | 2008-02-15 | 2009-08-20 | Boehringer Ingelheim Vetmedica, Inc. | Methods and compositions for reducing the impact of enteric diseases |
| US8444989B1 (en) | 2008-04-18 | 2013-05-21 | Boehringer Ingelheim Vetmedica Gmbh | One dose vaccination against mycoplasma infections of pigs |
| TWI627281B (zh) | 2009-09-02 | 2018-06-21 | 百靈佳殷格翰家畜藥品公司 | 降低pcv-2組合物殺病毒活性之方法及具有改良免疫原性之pcv-2組合物 |
| SI2547770T1 (sl) | 2010-03-16 | 2020-07-31 | Virginia Tech Intellectual Properties, Inc. | Vakcina atenuiranega živega himernega prašičjega virusa |
| CN102859875B (zh) | 2010-04-22 | 2016-10-05 | 皇家飞利浦电子股份有限公司 | 皮肤接触检测器 |
| AU2010357208A1 (en) | 2010-07-08 | 2013-02-21 | United Biomedical, Inc. | Designer peptide-based PCV2 vaccine |
| US9580474B2 (en) | 2010-09-08 | 2017-02-28 | The Johns Hopkins University | Polyionic papilloma virus-like particle (VLP) vaccines |
| EP2564869A1 (en) | 2011-09-02 | 2013-03-06 | Ceva Sante Animale | Synthetic capsid proteins and uses thereof |
| CN103122352B (zh) | 2012-09-27 | 2015-02-11 | 华中农业大学 | 一种猪圆环病毒2型重组杆状病毒及制备方法和应用 |
| ES2734374T3 (es) * | 2013-03-01 | 2019-12-05 | Boehringer Ingelheim Animal Health Usa Inc | Cuantificación de composiciones de vacuna |
| CN105246505A (zh) | 2013-04-30 | 2016-01-13 | 勃林格殷格翰动物保健公司 | PCV2a亚型(PCV2a)的ORF2蛋白用于交叉保护中的用途 |
| CN105658660A (zh) | 2013-10-02 | 2016-06-08 | 勃林格殷格翰动物保健公司 | Pcv2 orf2蛋白变体和由其组成的病毒样颗粒 |
| WO2015169732A1 (en) | 2014-05-07 | 2015-11-12 | Boehringer Ingelheim International Gmbh | Container, nebulizer and use |
| CN107531760A (zh) | 2015-03-30 | 2018-01-02 | 勃林格殷格翰动物保健公司 | Pcv2 orf2载体平台 |
-
2014
- 2014-10-02 CN CN201480054488.4A patent/CN105658660A/zh active Pending
- 2014-10-02 US US14/504,839 patent/US9505808B2/en active Active
- 2014-10-02 MX MX2016004063A patent/MX373884B/es active IP Right Grant
- 2014-10-02 CA CA2924228A patent/CA2924228C/en active Active
- 2014-10-02 EP EP14789683.1A patent/EP3052516B1/en active Active
- 2014-10-02 DK DK14789683.1T patent/DK3052516T3/da active
- 2014-10-02 ES ES14789683T patent/ES2778425T3/es active Active
- 2014-10-02 KR KR1020227019654A patent/KR20220083861A/ko not_active Ceased
- 2014-10-02 JP JP2016519871A patent/JP6778104B2/ja active Active
- 2014-10-02 AU AU2014329524A patent/AU2014329524B2/en active Active
- 2014-10-02 KR KR1020167011660A patent/KR102409183B1/ko active Active
- 2014-10-02 WO PCT/US2014/058793 patent/WO2015051099A1/en not_active Ceased
- 2014-10-02 EA EA201600305A patent/EA035265B1/ru not_active IP Right Cessation
-
2016
- 2016-10-17 US US15/295,053 patent/US10131696B2/en active Active
-
2018
- 2018-10-08 US US16/154,294 patent/US10961281B2/en active Active
-
2021
- 2021-03-29 US US17/215,636 patent/US11858963B2/en active Active
-
2023
- 2023-11-21 US US18/516,237 patent/US12247051B2/en active Active
-
2025
- 2025-02-05 US US19/046,205 patent/US20250243246A1/en active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| CN105658660A (zh) | 2016-06-08 |
| US9505808B2 (en) | 2016-11-29 |
| US11858963B2 (en) | 2024-01-02 |
| EA201600305A1 (ru) | 2016-09-30 |
| US20190023747A1 (en) | 2019-01-24 |
| JP2016534034A (ja) | 2016-11-04 |
| CA2924228C (en) | 2024-01-16 |
| US12247051B2 (en) | 2025-03-11 |
| JP6778104B2 (ja) | 2020-10-28 |
| KR102409183B1 (ko) | 2022-06-15 |
| KR20160058958A (ko) | 2016-05-25 |
| US20150093404A1 (en) | 2015-04-02 |
| AU2014329524A1 (en) | 2016-03-17 |
| US20210221852A1 (en) | 2021-07-22 |
| DK3052516T3 (da) | 2020-03-23 |
| CA2924228A1 (en) | 2015-04-09 |
| EP3052516A1 (en) | 2016-08-10 |
| US20250243246A1 (en) | 2025-07-31 |
| WO2015051099A1 (en) | 2015-04-09 |
| US20170029471A1 (en) | 2017-02-02 |
| US10131696B2 (en) | 2018-11-20 |
| US20240076321A1 (en) | 2024-03-07 |
| ES2778425T3 (es) | 2020-08-10 |
| MX2016004063A (es) | 2016-06-06 |
| US10961281B2 (en) | 2021-03-30 |
| EP3052516B1 (en) | 2020-01-01 |
| EA035265B1 (ru) | 2020-05-21 |
| MX373884B (es) | 2020-03-26 |
| AU2014329524B2 (en) | 2019-01-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12247051B2 (en) | PCV2 ORF2 protein variant and virus like particles composed thereof | |
| EP2114447B1 (en) | Prevention and treatment of sub-clinical pcvd | |
| EP3035958B1 (en) | Porcine circovirus type 2 (pcv2) subunit vaccine | |
| US20200237894A1 (en) | Pcv2 orf2 carrier platform | |
| US12433942B2 (en) | Porcine circovirus type 3 (PCV3) vaccines, and production and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A107 | Divisional application of patent | ||
| PA0104 | Divisional application for international application |
Comment text: Divisional Application for International Patent Patent event code: PA01041R01D Patent event date: 20220610 Application number text: 1020167011660 Filing date: 20160502 |
|
| PG1501 | Laying open of application | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20220708 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20230316 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20230922 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20230316 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |