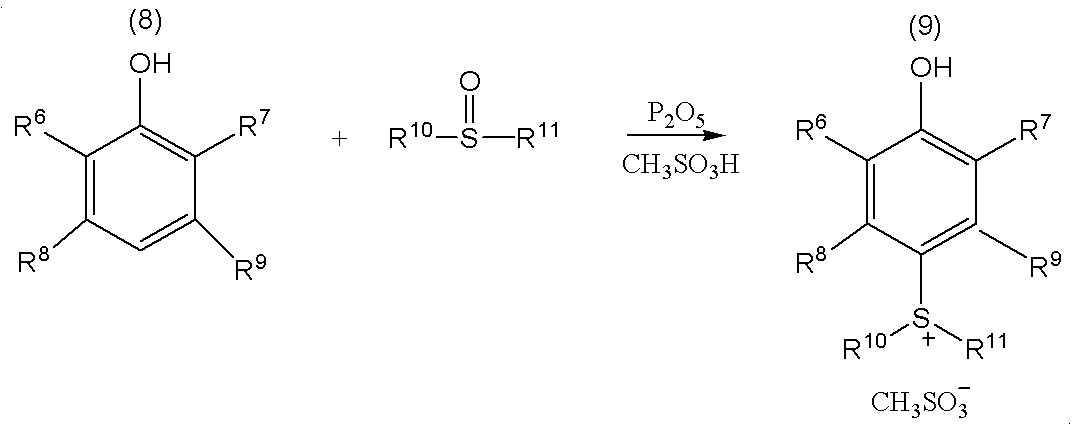JP5116312B2 - スルホニウム塩 - Google Patents
スルホニウム塩 Download PDFInfo
- Publication number
- JP5116312B2 JP5116312B2 JP2007025961A JP2007025961A JP5116312B2 JP 5116312 B2 JP5116312 B2 JP 5116312B2 JP 2007025961 A JP2007025961 A JP 2007025961A JP 2007025961 A JP2007025961 A JP 2007025961A JP 5116312 B2 JP5116312 B2 JP 5116312B2
- Authority
- JP
- Japan
- Prior art keywords
- group
- formula
- represented
- ion
- integer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical class S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 title claims description 37
- -1 t- butylphenyl group Chemical group 0.000 claims description 101
- 125000004432 carbon atom Chemical group C* 0.000 claims description 18
- 150000001450 anions Chemical class 0.000 claims description 17
- 125000000217 alkyl group Chemical group 0.000 claims description 14
- 125000003118 aryl group Chemical group 0.000 claims description 13
- 150000002500 ions Chemical class 0.000 claims description 10
- 229940077388 benzenesulfonate Drugs 0.000 claims description 6
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 6
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 6
- 125000004122 cyclic group Chemical group 0.000 claims description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 4
- 229910017008 AsF 6 Inorganic materials 0.000 claims description 3
- 229910018286 SbF 6 Inorganic materials 0.000 claims description 3
- 150000004996 alkyl benzenes Chemical class 0.000 claims description 3
- 125000003944 tolyl group Chemical group 0.000 claims description 3
- 125000002947 alkylene group Chemical group 0.000 claims description 2
- 150000001875 compounds Chemical class 0.000 description 32
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 30
- 239000002253 acid Substances 0.000 description 24
- 239000002904 solvent Substances 0.000 description 24
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 24
- 238000006243 chemical reaction Methods 0.000 description 22
- 239000010410 layer Substances 0.000 description 19
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 15
- 125000000962 organic group Chemical group 0.000 description 14
- 230000015572 biosynthetic process Effects 0.000 description 13
- 238000003786 synthesis reaction Methods 0.000 description 13
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 12
- 239000000203 mixture Substances 0.000 description 11
- 229920002120 photoresistant polymer Polymers 0.000 description 11
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 10
- 239000003054 catalyst Substances 0.000 description 10
- 239000012044 organic layer Substances 0.000 description 10
- 238000005160 1H NMR spectroscopy Methods 0.000 description 9
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 9
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 8
- 239000000243 solution Substances 0.000 description 8
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 description 6
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 6
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 6
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 6
- 125000002723 alicyclic group Chemical group 0.000 description 6
- QGZKDVFQNNGYKY-UHFFFAOYSA-N ammonia Natural products N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 6
- 235000011114 ammonium hydroxide Nutrition 0.000 description 6
- 230000005855 radiation Effects 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 125000001424 substituent group Chemical group 0.000 description 6
- 125000002837 carbocyclic group Chemical group 0.000 description 5
- 229940098779 methanesulfonic acid Drugs 0.000 description 5
- 239000003960 organic solvent Substances 0.000 description 5
- 229920000642 polymer Polymers 0.000 description 5
- 150000003839 salts Chemical group 0.000 description 5
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- 230000002378 acidificating effect Effects 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 4
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 4
- 229910000027 potassium carbonate Inorganic materials 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- GHMLBKRAJCXXBS-UHFFFAOYSA-N resorcinol Chemical compound OC1=CC=CC(O)=C1 GHMLBKRAJCXXBS-UHFFFAOYSA-N 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- 238000005406 washing Methods 0.000 description 4
- JGTNAGYHADQMCM-UHFFFAOYSA-M 1,1,2,2,3,3,4,4,4-nonafluorobutane-1-sulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F JGTNAGYHADQMCM-UHFFFAOYSA-M 0.000 description 3
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonium chloride Substances [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 3
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 3
- KWYHDKDOAIKMQN-UHFFFAOYSA-N N,N,N',N'-tetramethylethylenediamine Chemical compound CN(C)CCN(C)C KWYHDKDOAIKMQN-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- FFPGPHSOELVMNF-UHFFFAOYSA-N [2-(4-ethenoxyoctoxy)phenyl]-diphenylsulfanium Chemical compound CCCCC(OC=C)CCCOC1=CC=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 FFPGPHSOELVMNF-UHFFFAOYSA-N 0.000 description 3
- MBSCDFBXUGQIGB-UHFFFAOYSA-N [4-(2-ethenoxyethoxy)phenyl]-diphenylsulfanium Chemical compound C1=CC(OCCOC=C)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 MBSCDFBXUGQIGB-UHFFFAOYSA-N 0.000 description 3
- 125000005073 adamantyl group Chemical group C12(CC3CC(CC(C1)C3)C2)* 0.000 description 3
- 150000001299 aldehydes Chemical class 0.000 description 3
- 125000003545 alkoxy group Chemical group 0.000 description 3
- 239000012141 concentrate Substances 0.000 description 3
- 239000013078 crystal Substances 0.000 description 3
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 3
- 238000004821 distillation Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 3
- 238000001914 filtration Methods 0.000 description 3
- 238000004255 ion exchange chromatography Methods 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 238000000034 method Methods 0.000 description 3
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 3
- 125000001624 naphthyl group Chemical group 0.000 description 3
- 239000012299 nitrogen atmosphere Substances 0.000 description 3
- NLKNQRATVPKPDG-UHFFFAOYSA-M potassium iodide Chemical compound [K+].[I-] NLKNQRATVPKPDG-UHFFFAOYSA-M 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 229960001755 resorcinol Drugs 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 239000000741 silica gel Substances 0.000 description 3
- 229910002027 silica gel Inorganic materials 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- DLYUQMMRRRQYAE-UHFFFAOYSA-N tetraphosphorus decaoxide Chemical compound O1P(O2)(=O)OP3(=O)OP1(=O)OP2(=O)O3 DLYUQMMRRRQYAE-UHFFFAOYSA-N 0.000 description 3
- NAWXUBYGYWOOIX-SFHVURJKSA-N (2s)-2-[[4-[2-(2,4-diaminoquinazolin-6-yl)ethyl]benzoyl]amino]-4-methylidenepentanedioic acid Chemical compound C1=CC2=NC(N)=NC(N)=C2C=C1CCC1=CC=C(C(=O)N[C@@H](CC(=C)C(O)=O)C(O)=O)C=C1 NAWXUBYGYWOOIX-SFHVURJKSA-N 0.000 description 2
- RCBARGVNZTUGHE-UHFFFAOYSA-O (4-hydroxyphenyl)-diphenylsulfanium Chemical compound C1=CC(O)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 RCBARGVNZTUGHE-UHFFFAOYSA-O 0.000 description 2
- JHIGBMVXTRJDTO-UHFFFAOYSA-N (4-hydroxyphenyl)-diphenylsulfanium;1,1,2,2,3,3,4,4,4-nonafluorobutane-1-sulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F.C1=CC(O)=CC=C1[S+](C=1C=CC=CC=1)C1=CC=CC=C1 JHIGBMVXTRJDTO-UHFFFAOYSA-N 0.000 description 2
- DNJRKFKAFWSXSE-UHFFFAOYSA-N 1-chloro-2-ethenoxyethane Chemical compound ClCCOC=C DNJRKFKAFWSXSE-UHFFFAOYSA-N 0.000 description 2
- JHEBBEQGBZDHDA-UHFFFAOYSA-N 1-chloro-8-ethenoxyoctane Chemical compound ClCCCCCCCCOC=C JHEBBEQGBZDHDA-UHFFFAOYSA-N 0.000 description 2
- NXXYKOUNUYWIHA-UHFFFAOYSA-N 2,6-Dimethylphenol Chemical compound CC1=CC=CC(C)=C1O NXXYKOUNUYWIHA-UHFFFAOYSA-N 0.000 description 2
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- JJHHIJFTHRNPIK-UHFFFAOYSA-N Diphenyl sulfoxide Chemical compound C=1C=CC=CC=1S(=O)C1=CC=CC=C1 JJHHIJFTHRNPIK-UHFFFAOYSA-N 0.000 description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 2
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 2
- UIHCLUNTQKBZGK-UHFFFAOYSA-N Methyl isobutyl ketone Natural products CCC(C)C(C)=O UIHCLUNTQKBZGK-UHFFFAOYSA-N 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- 235000011054 acetic acid Nutrition 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- FMLKCNMSYUQGPP-UHFFFAOYSA-O bis(4-tert-butylphenyl)-(4-hydroxy-3,5-dimethylphenyl)sulfanium Chemical compound CC1=C(O)C(C)=CC([S+](C=2C=CC(=CC=2)C(C)(C)C)C=2C=CC(=CC=2)C(C)(C)C)=C1 FMLKCNMSYUQGPP-UHFFFAOYSA-O 0.000 description 2
- HSQVKUIFOMPJII-UHFFFAOYSA-N bis(4-tert-butylphenyl)-[4-(2-ethenoxyethoxy)-3,5-dimethylphenyl]sulfanium Chemical compound CC1=C(OCCOC=C)C(C)=CC([S+](C=2C=CC(=CC=2)C(C)(C)C)C=2C=CC(=CC=2)C(C)(C)C)=C1 HSQVKUIFOMPJII-UHFFFAOYSA-N 0.000 description 2
- UOCJDOLVGGIYIQ-PBFPGSCMSA-N cefatrizine Chemical group S([C@@H]1[C@@H](C(N1C=1C(O)=O)=O)NC(=O)[C@H](N)C=2C=CC(O)=CC=2)CC=1CSC=1C=NNN=1 UOCJDOLVGGIYIQ-PBFPGSCMSA-N 0.000 description 2
- 239000007810 chemical reaction solvent Substances 0.000 description 2
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 2
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 230000018044 dehydration Effects 0.000 description 2
- 238000006297 dehydration reaction Methods 0.000 description 2
- 238000010494 dissociation reaction Methods 0.000 description 2
- 230000005593 dissociations Effects 0.000 description 2
- 238000010894 electron beam technology Methods 0.000 description 2
- FJKIXWOMBXYWOQ-UHFFFAOYSA-N ethenoxyethane Chemical compound CCOC=C FJKIXWOMBXYWOQ-UHFFFAOYSA-N 0.000 description 2
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 2
- 235000019253 formic acid Nutrition 0.000 description 2
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- XMBWDFGMSWQBCA-UHFFFAOYSA-M iodide Chemical compound [I-] XMBWDFGMSWQBCA-UHFFFAOYSA-M 0.000 description 2
- 125000002510 isobutoxy group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])O* 0.000 description 2
- 125000003253 isopropoxy group Chemical group [H]C([H])([H])C([H])(O*)C([H])([H])[H] 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- DNIAPMSPPWPWGF-UHFFFAOYSA-N monopropylene glycol Natural products CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 2
- 125000006606 n-butoxy group Chemical group 0.000 description 2
- 125000001298 n-hexoxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 2
- 125000006608 n-octyloxy group Chemical group 0.000 description 2
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 235000006408 oxalic acid Nutrition 0.000 description 2
- 125000001820 oxy group Chemical group [*:1]O[*:2] 0.000 description 2
- 229960004065 perflutren Drugs 0.000 description 2
- 150000002989 phenols Chemical class 0.000 description 2
- 238000000206 photolithography Methods 0.000 description 2
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 2
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- LLHKCFNBLRBOGN-UHFFFAOYSA-N propylene glycol methyl ether acetate Chemical compound COCC(C)OC(C)=O LLHKCFNBLRBOGN-UHFFFAOYSA-N 0.000 description 2
- 125000005920 sec-butoxy group Chemical group 0.000 description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 238000004904 shortening Methods 0.000 description 2
- 150000003462 sulfoxides Chemical class 0.000 description 2
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- WGTYBPLFGIVFAS-UHFFFAOYSA-M tetramethylammonium hydroxide Chemical compound [OH-].C[N+](C)(C)C WGTYBPLFGIVFAS-UHFFFAOYSA-M 0.000 description 2
- 229910052724 xenon Inorganic materials 0.000 description 2
- FHNFHKCVQCLJFQ-UHFFFAOYSA-N xenon atom Chemical compound [Xe] FHNFHKCVQCLJFQ-UHFFFAOYSA-N 0.000 description 2
- 125000003837 (C1-C20) alkyl group Chemical group 0.000 description 1
- KBHBUUBXEQUIMV-UHFFFAOYSA-N 1,1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8-heptadecafluorooctane Chemical compound FC(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F KBHBUUBXEQUIMV-UHFFFAOYSA-N 0.000 description 1
- VDFVNEFVBPFDSB-UHFFFAOYSA-N 1,3-dioxane Chemical compound C1COCOC1 VDFVNEFVBPFDSB-UHFFFAOYSA-N 0.000 description 1
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 1
- CNQLAJSJIFZPCF-UHFFFAOYSA-N 1-tert-butyl-4-(4-tert-butylphenyl)sulfinylbenzene Chemical compound C1=CC(C(C)(C)C)=CC=C1S(=O)C1=CC=C(C(C)(C)C)C=C1 CNQLAJSJIFZPCF-UHFFFAOYSA-N 0.000 description 1
- YDFAJMDFCCJZSI-UHFFFAOYSA-N 8-chlorooctan-1-ol Chemical compound OCCCCCCCCCl YDFAJMDFCCJZSI-UHFFFAOYSA-N 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 1
- QYKIQEUNHZKYBP-UHFFFAOYSA-N Vinyl ether Chemical compound C=COC=C QYKIQEUNHZKYBP-UHFFFAOYSA-N 0.000 description 1
- 239000003377 acid catalyst Substances 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 238000007259 addition reaction Methods 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 229940045714 alkyl sulfonate alkylating agent Drugs 0.000 description 1
- 150000001449 anionic compounds Chemical class 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 238000006704 dehydrohalogenation reaction Methods 0.000 description 1
- 125000005982 diphenylmethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])(*)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 229910001412 inorganic anion Inorganic materials 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 229940006461 iodide ion Drugs 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 125000004817 pentamethylene group Chemical group [H]C([H])([*:2])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[*:1] 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 239000004810 polytetrafluoroethylene Substances 0.000 description 1
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- RLUCXJBHKHIDSP-UHFFFAOYSA-N propane-1,2-diol;propanoic acid Chemical compound CCC(O)=O.CC(O)CO RLUCXJBHKHIDSP-UHFFFAOYSA-N 0.000 description 1
- 239000012264 purified product Substances 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 239000004065 semiconductor Substances 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 125000005415 substituted alkoxy group Chemical group 0.000 description 1
- 150000003459 sulfonic acid esters Chemical class 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 125000000383 tetramethylene group Chemical group [H]C([H])([*:1])C([H])([H])C([H])([H])C([H])([H])[*:2] 0.000 description 1
- 230000002463 transducing effect Effects 0.000 description 1
Landscapes
- Materials For Photolithography (AREA)
- Nitrogen- Or Sulfur-Containing Heterocyclic Ring Compounds With Rings Of Six Or More Members (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Description
本発明のスルホニウム塩は、上記式(1)で表される化合物である。式(1)において、R1〜R4は炭素数2〜9のアルキル基を表す。特に好ましい有機基として、エチル基、n−ブチル基及びn−ヘキシル基が挙げられる。また、D1〜D8は互いに独立で、少なくとも1個は上記式(2)で表される基であり、残りは水素原子、上記式(3)で表される基又は上記式(4)で表される基である。式(2)で表される基は、活性放射線の露光により酸を発生する光酸発生剤としての機能を有する構造と、この酸発生剤から発生した酸で解離しうる基(酸解離基)とを有するため、式(1)で表されるスルホニウム塩は、有機溶媒に溶解させることにより化学増幅型の感光性組成物とすることができる。式(1)で表されるスルホニウム塩は、式(3)で表される基や式(4)で表される基が導入されていなくてもよいが、式(3)で表される基や式(4)で表される基を導入することにより、溶解度を調整することができる。式(3)で表される基や式(4)で表される基を導入する割合は、式(1)で表されるスルホニウム塩に対してそれぞれ1〜3当量程度が好ましい。
下記式で表される化合物(4−ビニロキシエトキシフェニルジフェニルスルホニウム パーフルオロブタンスルホン酸塩)の合成:
1H−NMR(400MHz,CDCl3)δ4.05−4.08(m,3H),4.24(d,J=7.4,2.4Hz,1H),4.31−4.33(m,2H),6.49(dd,J=14.4,7.4Hz,1H),7.24(d,J=6.8Hz,2H),7.64−7.74(m,12H)
下記式で表される化合物(4−ビニロキシエトキシ−3,5−ジメチルフェニルジ(4−t−ブチルフェニル)スルホニウム シクロ(1,3−パーフルオロプロパンジスルホン)イミド塩)の合成:
1H−NMR(400MHz,CDCl3)δ1.35(s,18H),2.36(s,6H),4.02−4.08(m,3H),4.12−4.14(m,2H),4.25(d,J=14.3,6.1Hz,1H),6.50(dd,J=14.3,6.6Hz,1H),7.35(s,2H),7.59−7.75(m,8H)
下記式で表される化合物(4−ビニロキシオクトキシフェニルジフェニルスルホニウム ビス(パーフルオロメタンスルホン)イミド塩)の合成:
1H−NMR(400MHz,CDCl3) δ1.36−1.47(m,8H),1.64−1.67(m,2H),1.78−1.83(m,2H),3.67(t,J=6.6 Hz,2H),3.96(dd,J=6.8,2.0Hz,1H),4.04(t,J=6.6Hz,2H).4.16(dd,J=14.4,2.0Hz,1H),6.46(dd,J=14.4,6.8Hz,1H),7.16−7.19(m,2H),7.65−7.76(m,12H)
上記の実施例1〜3で得たスルホニウム塩100重量部とトリエタノールアミン3重量部とをプロピレングリコールモノメチルアセテート500重量部に溶解し、フィルター(PTFEフィルター)でろ過した。この溶液を、スピナーを用いて、シリコンウエハ(直径:4インチ)に塗布し、120℃で90秒間プレベークし、膜厚300nmのレジスト膜を得た。この膜に、キセノンランプ(波長:248nm)により露光し、次いで100℃で60秒間ポストベークを行った。その後、23℃で現像液(2.38重量%のテトラメチルアンモニウムハイドロキサイドの水溶液)を用いて、ブレークスルータイム(一定のエネルギーを照射した後、現像により残膜が皆無になる秒数)を測定した。
この結果、実施例1〜3で得たスルホニウム塩のブレークスルータイムが、100mJではすべて1秒以下であった。このことから、実施例1〜3で得られた感光性化合物は、キセノンランプによる露光で、本発明のスルホニウム塩から由来した構造部分から酸が発生し、この酸によりそのスルホニウム塩における式(2)で表される基、式(3)で表される基及び式(4)で表される基が分解し、現像液に対して難溶解性から可溶性になったことが判った。
実施例1〜3のスルホニウム塩は、フェノール化合物を母体とし、任意に式(3)、式(4)の基を導入できるため、従来のスルホニウム塩よりもレジスト溶媒への溶解性が高いという特徴が挙げられる。実施例1〜3のスルホニウム塩は、プロピレングリコールモノメチルエーテルアセテートに20重量%以上溶解することが確認された。
Claims (5)
- 下記式(1)で表されることを特徴とするスルホニウム塩。
- 前記X-で表される陰イオンが、下記式(5)で表される陰イオンであることを特徴とする請求項1に記載のスルホニウム塩。
- 前記X-で表される陰イオンが、Cl-、Br-、I-、BF4 -、AsF6 -、SbF6 -又はPF6 -であることを特徴とする請求項1に記載のスルホニウム塩。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2007025961A JP5116312B2 (ja) | 2007-02-05 | 2007-02-05 | スルホニウム塩 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2007025961A JP5116312B2 (ja) | 2007-02-05 | 2007-02-05 | スルホニウム塩 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2008189602A JP2008189602A (ja) | 2008-08-21 |
| JP5116312B2 true JP5116312B2 (ja) | 2013-01-09 |
Family
ID=39750056
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2007025961A Expired - Fee Related JP5116312B2 (ja) | 2007-02-05 | 2007-02-05 | スルホニウム塩 |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP5116312B2 (ja) |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5084289B2 (ja) * | 2007-02-05 | 2012-11-28 | 東洋合成工業株式会社 | スルホニウム塩 |
| JP5084288B2 (ja) * | 2007-02-05 | 2012-11-28 | 東洋合成工業株式会社 | スルホニウム塩 |
| JP4854023B2 (ja) * | 2007-02-05 | 2012-01-11 | 兵庫県 | 感光性組成物 |
| JP5262631B2 (ja) * | 2008-12-01 | 2013-08-14 | Jsr株式会社 | 感放射線性組成物 |
| JP5292216B2 (ja) * | 2009-07-31 | 2013-09-18 | 富士フイルム株式会社 | 感活性光線性又は感放射線性組成物及び該組成物を用いたパターン形成方法 |
| JP5707249B2 (ja) * | 2011-06-22 | 2015-04-22 | 富士フイルム株式会社 | 化学増幅型レジスト組成物、並びに、それを用いたレジスト膜、レジスト塗布マスクブランクス、レジストパターン形成方法、及び、フォトマスク |
| JP2013100261A (ja) * | 2011-10-14 | 2013-05-23 | Idemitsu Kosan Co Ltd | 環状化合物及びその組成物 |
| JP5812914B2 (ja) * | 2012-03-27 | 2015-11-17 | Jsr株式会社 | レジスト組成物 |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5100115B2 (ja) * | 2006-03-16 | 2012-12-19 | 東洋合成工業株式会社 | スルホニウム塩及び酸発生剤 |
| JP4854023B2 (ja) * | 2007-02-05 | 2012-01-11 | 兵庫県 | 感光性組成物 |
-
2007
- 2007-02-05 JP JP2007025961A patent/JP5116312B2/ja not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| JP2008189602A (ja) | 2008-08-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5116311B2 (ja) | スルホニウム塩 | |
| US7396960B2 (en) | Sulfonium salts | |
| JP4854023B2 (ja) | 感光性組成物 | |
| JP5116312B2 (ja) | スルホニウム塩 | |
| KR101799325B1 (ko) | 술폰산 유도체 및 광산발생제 | |
| JP6472097B2 (ja) | スルホン酸誘導体、それを用いた光酸発生剤、レジスト組成物及びデバイスの製造方法 | |
| US7858287B2 (en) | Photosensitive resin, and photosensitive composition | |
| JP5116310B2 (ja) | スルホニウム塩 | |
| JP5728916B2 (ja) | スルホンアミドオニウム塩の製造方法 | |
| KR20010051364A (ko) | 술포늄 염 화합물, 포토레지스트 조성물 및 이를 사용한패턴화 방법 | |
| JP5088933B2 (ja) | スルホニウム塩 | |
| TWI423948B (zh) | 光酸產生劑及其製造方法,以及含有該光酸產生劑的光阻組合物 | |
| TWI591048B (zh) | 新穎鋶鹽、其製造方法及光酸產生劑 | |
| JP5084288B2 (ja) | スルホニウム塩 | |
| JP5084289B2 (ja) | スルホニウム塩 | |
| JP4921160B2 (ja) | 感光性樹脂及び感光性組成物 | |
| JP4122175B2 (ja) | 新規スルホン酸誘導体及び光酸発生剤 | |
| JP6215229B2 (ja) | 新規なスルホニウム塩化合物、その製造方法及び光酸発生剤 | |
| JP6530957B2 (ja) | レジスト組成物、スルホン酸誘導体、該スルホン酸誘導体の製造方法及びデバイスの製造方法 | |
| JP2002080420A (ja) | アリールエステル残基を有する化合物の光変換方法 | |
| JPH08208597A (ja) | ベンジルスルホニウムトリフルオロメタンスルホン酸塩 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20100126 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120704 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120823 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20121010 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20121016 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20171026 Year of fee payment: 5 |
|
| LAPS | Cancellation because of no payment of annual fees |