JP5020463B2 - ドナリエラ属(Dunaliella)の粉末の治療的使用 - Google Patents
ドナリエラ属(Dunaliella)の粉末の治療的使用 Download PDFInfo
- Publication number
- JP5020463B2 JP5020463B2 JP2004212054A JP2004212054A JP5020463B2 JP 5020463 B2 JP5020463 B2 JP 5020463B2 JP 2004212054 A JP2004212054 A JP 2004212054A JP 2004212054 A JP2004212054 A JP 2004212054A JP 5020463 B2 JP5020463 B2 JP 5020463B2
- Authority
- JP
- Japan
- Prior art keywords
- riera
- dunaliella
- donna
- genus
- powder
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/427—Thiazoles not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/192—Carboxylic acids, e.g. valproic acid having aromatic groups, e.g. sulindac, 2-aryl-propionic acids, ethacrynic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/216—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acids having aromatic rings, e.g. benactizyne, clofibrate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/426—1,3-Thiazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/02—Algae
- A61K36/05—Chlorophycota or chlorophyta (green algae), e.g. Chlorella
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
Description
本発明は、高いトリグリセリドおよび低い高−密度リポタンパク質(HDL)コレステロールレベル、アテローム硬化症および糖尿病を含む多数の状態の処置における粗製ドナリエラ バーダウィル(Dunaliella bardawil)粉末の使用に関する。
参考文献
以下の参考文献リストは、本発明の背景として直接関係があると考えられる:
1.非特許文献1
2.1980年3月18日に発効された特許文献1
3.1980年4月29日に発効された特許文献2
4.非特許文献2
5.非特許文献3
6.非特許文献4
ドナリエラ属(Dunaliella)の2種であるドナリエラ サリーナ(Dunaliella salina)Teod.およびドナリエラ バーダウィル(Dunaliella bardawil)は、単細胞で2本の鞭毛を持ち(biflagellate)、壁が無い緑藻で、大変大量のβ−カロテンを生産することができる(1)。D.バーダウィル(bardawil)は、β−カロテン含量が約50%の全トランス−β−カロテンから構成され、そして残りのほとんどが9−シスβ−カロテンおよび幾らかの他のβ−カロテン異性体からなる耐塩性の藻である(2)。最高約5重量%のβ−カロテンを含む藻を得るために、D.バーダウィル(bardawil)を培養する方法が記載された(3)。後にこの方法を開発して、この割合が8%以上に増加した。藻であるドナリエラ バーダウィル(Dunaliella bardawil)に蓄積するβ−カロテンの天然の異性体混合物は、合成の全トランス−β−カロテンを与えることにより観察されるよりも約10倍高い程度で、ラットおよびニワトリの脂肪組織に蓄積することが示された(4)。
本発明の目的は糖尿病、低血漿HDLおよび/または高血漿TGおよび/またはアテローム硬化症に罹患している患者の処置法を提供することである。
発明の態様の詳細な説明
方法および材料
以下に記載するすべてのヒトの実験には、以下のように調製したドナリエラ属(Dunaliella)の粉末を含むカプセルを使用した。
1.対照、西洋食のみ(対照)
2.9−シスレチノイン酸(RA)
3.ロシグリタゾン(Rosi)
4.ドナリエラ属(Dunaliella)の粉末8%(Dun)
結果
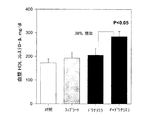
ドナリエラ属(Dunaliella)の処置は、ロシグリタゾンおよび対照群に比べて血漿グルコースおよびインスリンレベルの両方を下げた(図7および8)。さらにドナリエラ属(Dunaliella)の処置は、ロシグリタゾン群に比べて血漿コレステロールおよびTGレベルの両方を有意に下げた(図9および10)。他方、9−シスレチノイン酸は血漿インスリンおよびグルコースレベルに影響を与えず、そしてドナリエラ属(Dunaliella)群のそれらのレベルは有意に低かった。結果はドナリエラ属(Dunaliella)の粉末が、LDLR−/−マウスモデルにおいて9−シスレチノイン酸またはロシグリタゾンよりも効果的に血漿インスリンおよびグルコースレベルを下げることができることを証明する。
Claims (18)
- 有効量の実質的に粗製のドナリエラ属(Dunaliella)の藻類調製物を含んでなる、2型糖尿病を治療するための薬剤。
- 核内受容体の1以上のアクチベーターを更に含んでなる、請求項1に記載の薬剤。
- 核内受容体のアクチベーターがペルオキシソーム増殖因子活性化受容体αまたはγ(PPARαまたはPPARγ)アゴニストである、請求項2に記載の薬剤。
- PPARαまたはPPARγアゴニストがフィブラートおよびチアゾリジンジオンから選択される、請求項3に記載の薬剤。
- フィブラートがクロフィブラート、フェノフィブラート、ベザフィブラート、シプロフィブラート、ベクロフィブラートおよびゲムフィブロジルから選択される、請求項4に記載の薬剤。
- チアゾリジンジオンがアバンディア、トログリタゾン、BRL49653、ピオグリタゾン、シグリタゾン、WAY−120,744、エングリタゾン、AD5075、ダルグリタゾンおよびロシグリタゾンから選択される、請求項4に記載の薬剤。
- 上記の粗製のドナリエラ属(Dunaliella)の粉末が経口的に投与される、請求項1に記載の薬剤。
- 上記藻類がドナリエラ バーダウィル(Dunaliella bardawil)である請求項1に記載の薬剤。
- 上記粉末がカプセル化されている、請求項1に記載の薬剤。
- 2型糖尿病を治療するための製薬学的組成物の調製における、有効量の実質的に粗製のドナリエラ属(Dunaliella)の藻類調製物の使用。
- 製薬学的組成物が核内受容体の1以上のアクチベーターを更に含んでなる、請求項10に記載の使用。
- 核内受容体のアクチベーターが、ペルオキシソーム増殖因子活性化受容体αまたはγ(PPARαまたはPPARγ)アゴニストである、請求項11に記載の使用。
- PPARαまたはPPARγアゴニストがフィブラートおよびチアゾリジンジオンから選択される、請求項12に記載の使用。
- フィブラートがクロフィブラート、フェノフィブラート、ベザフィブラート、シプロフィブラート、ベクロフィブラートおよびゲムフィブロジルから選択される、請求項13に記載の使用。
- チアゾリジンジオンがアバンディア、トログリタゾン、BRL49653、ピオグリタゾン、シグリタゾン、WAY−120,744、エングリタゾン、AD5075、ダルグリタゾンおよびロシグリタゾンから選択される、請求項13に記載の使用。
- 上記の粗製のドナリエラ属(Dunaliella)の粉末が経口的に投与される、請求項10に記載の使用。
- 上記藻類がドナリエラ バーダウィル(Dunaliella bardawil)である、請求項10に記載の使用。
- 上記粉末がカプセル化されている、請求項10に記載の使用。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/668,601 US7264813B2 (en) | 2003-09-24 | 2003-09-24 | Therapeutic uses of Dunaliella powder |
| US10/668601 | 2003-09-24 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009187809A Division JP5356946B2 (ja) | 2003-09-24 | 2009-08-13 | ドナリエラ属(Dunaliella)の粉末の治療的使用 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2005097255A JP2005097255A (ja) | 2005-04-14 |
| JP2005097255A5 JP2005097255A5 (ja) | 2005-09-29 |
| JP5020463B2 true JP5020463B2 (ja) | 2012-09-05 |
Family
ID=34313520
Family Applications (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2004212054A Active JP5020463B2 (ja) | 2003-09-24 | 2004-07-20 | ドナリエラ属(Dunaliella)の粉末の治療的使用 |
| JP2009187809A Active JP5356946B2 (ja) | 2003-09-24 | 2009-08-13 | ドナリエラ属(Dunaliella)の粉末の治療的使用 |
| JP2013126599A Withdrawn JP2013177457A (ja) | 2003-09-24 | 2013-06-17 | ドナリエラ属(Dunaliella)の粉末の治療的使用 |
| JP2016104166A Active JP6321074B2 (ja) | 2003-09-24 | 2016-05-25 | ドナリエラ属(Dunaliella)の粉末の治療的使用 |
| JP2017235555A Active JP6490782B2 (ja) | 2003-09-24 | 2017-12-07 | ドナリエラ属(Dunaliella)の粉末の治療的使用 |
Family Applications After (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009187809A Active JP5356946B2 (ja) | 2003-09-24 | 2009-08-13 | ドナリエラ属(Dunaliella)の粉末の治療的使用 |
| JP2013126599A Withdrawn JP2013177457A (ja) | 2003-09-24 | 2013-06-17 | ドナリエラ属(Dunaliella)の粉末の治療的使用 |
| JP2016104166A Active JP6321074B2 (ja) | 2003-09-24 | 2016-05-25 | ドナリエラ属(Dunaliella)の粉末の治療的使用 |
| JP2017235555A Active JP6490782B2 (ja) | 2003-09-24 | 2017-12-07 | ドナリエラ属(Dunaliella)の粉末の治療的使用 |
Country Status (6)
| Country | Link |
|---|---|
| US (3) | US7264813B2 (ja) |
| EP (2) | EP1522310B1 (ja) |
| JP (5) | JP5020463B2 (ja) |
| AT (1) | ATE548046T1 (ja) |
| HK (1) | HK1071845A1 (ja) |
| IL (1) | IL162987A (ja) |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007210917A (ja) * | 2006-02-08 | 2007-08-23 | Kochi Univ | 脂肪細胞縮小化剤、医薬品及び飲食品 |
| AU2006341188A1 (en) * | 2006-03-24 | 2007-10-04 | Ian Simon Tracton | Stable packaged dosage form and process therefor |
| JP2008222555A (ja) * | 2007-03-08 | 2008-09-25 | Nagasakiken Koritsu Daigaku Hojin | アディポネクチン受容体発現増加剤 |
| WO2009105048A2 (fr) * | 2008-02-19 | 2009-08-27 | Rached Smida | Nouvelles applications du hdl reconstitué |
| JP5443470B2 (ja) | 2008-04-29 | 2014-03-19 | 株式会社日健総本社 | 眼障害の治療方法 |
| JP5733592B2 (ja) * | 2008-10-20 | 2015-06-10 | 株式会社日健総本社 | 脂質異常治療薬の副作用防止物質 |
| US8747834B2 (en) | 2009-03-03 | 2014-06-10 | Solazyme, Inc. | Methods of treating impaired glucose metabolism via administration of algal biomass |
| US9180152B2 (en) | 2009-12-10 | 2015-11-10 | Nikken Sohonsha Corporation | Method for treating psoriasis |
| US9577089B2 (en) | 2010-03-02 | 2017-02-21 | Vishay-Siliconix | Structures and methods of fabricating dual gate devices |
| US10098371B2 (en) | 2013-01-28 | 2018-10-16 | Solazyme Roquette Nutritionals, LLC | Microalgal flour |
| FR3009619B1 (fr) | 2013-08-07 | 2017-12-29 | Roquette Freres | Compositions de biomasse de microalgues riches en proteines de qualite sensorielle optimisee |
| GB201718822D0 (en) | 2017-11-14 | 2017-12-27 | Univ Greenwich | Production of dunaliella |
| JP6947665B2 (ja) * | 2018-03-13 | 2021-10-13 | アイカ工業株式会社 | 光硬化性粘着樹脂組成物 |
| CN113905750A (zh) * | 2019-04-01 | 2022-01-07 | 戴尔哈修墨医学研究内结构和服务有限公司 | 用于预防及/或治疗神经退行性疾病、与蛋白质错误折叠关联的疾病及认知能力下降的杜氏藻属藻类制剂 |
Family Cites Families (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4199895A (en) | 1977-05-25 | 1980-04-29 | Yeda Research And Development Co. Ltd. | Production of glycerol, carotenes and algae meal |
| USPP4511P (en) | 1978-06-26 | 1980-03-18 | Yeda Research & Development Co. Ltd. | Alga strain |
| DE3729209A1 (de) | 1987-09-01 | 1989-03-09 | Boehringer Mannheim Gmbh | Verwendung von bezafibrat zur behandlung von diabetes |
| IL87260A (en) * | 1988-02-25 | 1991-12-15 | Tanaka Yoshio | Encapsulated food containing dunaliella algae and its production |
| US5190970A (en) * | 1990-10-19 | 1993-03-02 | E. R. Squibb & Sons, Inc. | Method for preventing onset of or treating Type II diabetes employing a cholesterol lowering drug alone or in combination with an ace inhibitor |
| US5219888A (en) | 1992-03-31 | 1993-06-15 | American Cyanamid Company | Use of retinoids for the treatment of coronary artery disease |
| DE69327100T2 (de) | 1992-06-04 | 2000-04-13 | Betatene Pty Ltd | Gemisch mit hohem gehalt an cis beta-carotin |
| US6262109B1 (en) * | 1995-12-22 | 2001-07-17 | Henkel Corporation | Methods of preventing and/or treating high serum levels of cholesterol and/or lipids |
| AU725998B2 (en) | 1995-09-18 | 2000-10-26 | Eisai R&D Management Co., Ltd. | Treating NIDDM with RXR agonists |
| WO1997010813A1 (en) * | 1995-09-18 | 1997-03-27 | Ligand Pharmaceuticals Incorporated | Ppar gamma antagonists for treating obesity |
| IL119224A (en) | 1996-09-09 | 2002-11-10 | Hadasit Med Res Service | Medicament for protection from effects of irradiation |
| JP3037628B2 (ja) * | 1997-03-06 | 2000-04-24 | マイクロアルジェコーポレーション株式会社 | 健康補助食品用軟カプセルの製法 |
| GB9712866D0 (en) * | 1997-06-18 | 1997-08-20 | Smithkline Beecham Plc | Novel method of treatment |
| US6083497A (en) * | 1997-11-05 | 2000-07-04 | Geltex Pharmaceuticals, Inc. | Method for treating hypercholesterolemia with unsubstituted polydiallylamine polymers |
| CA2324060A1 (en) | 1998-03-30 | 1999-10-07 | The Regents Of The University Of California | Methods and compounds for modulating nuclear receptor activity |
| JP2000080047A (ja) * | 1998-06-30 | 2000-03-21 | Takeda Chem Ind Ltd | 医 薬 |
| ES2255316T3 (es) | 1998-06-30 | 2006-06-16 | Takeda Pharmaceutical Company Limited | Composicion farmaceutica para el tratamiento de diabetes. |
| IL126447A (en) * | 1998-10-04 | 2004-09-27 | Vascular Biogenics Ltd | An immune preparation that confers tolerance in oral administration and its use in the prevention and / or treatment of atherosclerosis |
| EA004231B1 (ru) | 1998-12-23 | 2004-02-26 | Джи.Ди.Сирл Ллс | Сочетание ингибиторов белка, переносящего эфир холестерила, и производных фибриновой кислоты для сердечно-сосудистых показаний |
| US6914073B2 (en) * | 1999-03-18 | 2005-07-05 | Bristol Myers Squibb Company | Vitamin formulation for cardiovascular health |
| FR2795961B1 (fr) | 1999-07-09 | 2004-05-28 | Ethypharm Lab Prod Ethiques | Composition pharmaceutique contenant du fenofibrate micronise, un tensioactif et un derive cellulosique liant et procede de preparation |
| AU7586600A (en) | 1999-09-14 | 2001-04-17 | Ligand Pharmaceuticals Incorporated | Rxr modulators with improved pharmacologic profile |
| US6680387B2 (en) * | 2000-04-24 | 2004-01-20 | Aryx Therapeutics | Materials and methods for the treatment of diabetes, hyperlipidemia, hypercholesterolemia, and atherosclerosis |
| GB0019226D0 (en) * | 2000-08-04 | 2000-09-27 | Smithkline Beecham Plc | Novel pharmaceutical |
| JP2005511516A (ja) | 2001-09-25 | 2005-04-28 | スミスクライン ビーチャム コーポレーション | 選択的rxrリガンド |
| IN189624B (ja) * | 2001-10-23 | 2003-03-29 | Ajanta Pharma Ltd | |
| JP3978582B2 (ja) * | 2001-12-19 | 2007-09-19 | 株式会社日健総本社 | カロチノイドの製造方法 |
| US20030212138A1 (en) * | 2002-01-14 | 2003-11-13 | Pharmacia Corporation | Combinations of peroxisome proliferator-activated receptor-alpha agonists and cyclooxygenase-2 selective inhibitors and therapeutic uses therefor |
-
2003
- 2003-09-24 US US10/668,601 patent/US7264813B2/en not_active Expired - Lifetime
-
2004
- 2004-07-13 IL IL162987A patent/IL162987A/en active IP Right Grant
- 2004-07-20 JP JP2004212054A patent/JP5020463B2/ja active Active
- 2004-08-12 EP EP04019206A patent/EP1522310B1/en active Active
- 2004-08-12 AT AT04019206T patent/ATE548046T1/de active
- 2004-08-12 EP EP10010882A patent/EP2269620A1/en not_active Withdrawn
-
2005
- 2005-05-09 HK HK05103861.5A patent/HK1071845A1/xx unknown
-
2007
- 2007-07-10 US US11/822,786 patent/US7763255B2/en not_active Expired - Lifetime
-
2009
- 2009-08-13 JP JP2009187809A patent/JP5356946B2/ja active Active
-
2010
- 2010-05-11 US US12/777,757 patent/US8343549B2/en not_active Expired - Lifetime
-
2013
- 2013-06-17 JP JP2013126599A patent/JP2013177457A/ja not_active Withdrawn
-
2016
- 2016-05-25 JP JP2016104166A patent/JP6321074B2/ja active Active
-
2017
- 2017-12-07 JP JP2017235555A patent/JP6490782B2/ja active Active
Also Published As
| Publication number | Publication date |
|---|---|
| US20080019997A1 (en) | 2008-01-24 |
| JP2018035200A (ja) | 2018-03-08 |
| US8343549B2 (en) | 2013-01-01 |
| JP6321074B2 (ja) | 2018-05-09 |
| JP2016147916A (ja) | 2016-08-18 |
| EP1522310A1 (en) | 2005-04-13 |
| US20100221348A1 (en) | 2010-09-02 |
| EP1522310B1 (en) | 2012-03-07 |
| JP2013177457A (ja) | 2013-09-09 |
| JP5356946B2 (ja) | 2013-12-04 |
| JP2009256392A (ja) | 2009-11-05 |
| HK1071845A1 (en) | 2005-08-05 |
| EP2269620A1 (en) | 2011-01-05 |
| US7763255B2 (en) | 2010-07-27 |
| US20050063991A1 (en) | 2005-03-24 |
| JP2005097255A (ja) | 2005-04-14 |
| ATE548046T1 (de) | 2012-03-15 |
| IL162987A (en) | 2013-06-27 |
| US7264813B2 (en) | 2007-09-04 |
| JP6490782B2 (ja) | 2019-03-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6490782B2 (ja) | ドナリエラ属(Dunaliella)の粉末の治療的使用 | |
| JP2009256392A5 (ja) | ||
| JP2755559B2 (ja) | 前脂肪細胞の分化を刺激する組成物と方法、およびこれに関連する治療処置方法 | |
| AU2001286576B2 (en) | Composition and method for treatment of hypertriglyceridemia | |
| EA011637B1 (ru) | Лечение с использованием омега-3 жирных кислот и агониста и/или антагониста ppar и их комбинированный продукт | |
| MX2007001009A (es) | Composicion que contiene estatinas y acidos grasos omega-3. | |
| AU2001286576A1 (en) | Composition and method for treatment of hypertriglyceridemia | |
| JP2008521863A (ja) | Hiv感染患者における抗レトロウイルス治療がひき起こす高コレステロール血症の治療のためのオメガ−3脂肪酸の利用 | |
| JP5810166B2 (ja) | 糖尿病性心血管合併症の予防・治療剤 | |
| AU2015279905A1 (en) | Treatment of severe hypertriglyceridemia | |
| US8026281B2 (en) | Treating metabolic syndrome with fenofibrate | |
| JPS6299323A (ja) | 高脂血症剤 | |
| CA2963980A1 (en) | Monounsaturated fatty acid compositions and use for treating atherosclerosis | |
| US6890941B1 (en) | Compositions containing HMG Co-A reductase inhibitors and policosanol | |
| JP2002104964A (ja) | 糖尿病処置のためのフィタン酸の使用 | |
| JP2020503388A (ja) | 悪液質を予防及び/又は処置するためのオメガ−3脂肪酸組成物 | |
| WO1993009773A1 (en) | Preventive or curative for dysmenorrhea and food having function of preventing dysmenorrhea | |
| ZA200301525B (en) | Composition and method for treatment of hypertriglyceridemia. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20050720 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20050720 |
|
| RD03 | Notification of appointment of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7423 Effective date: 20081028 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20081203 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090107 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20090414 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20090714 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20090717 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090813 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20090813 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20090915 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20100115 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20100217 |
|
| A912 | Re-examination (zenchi) completed and case transferred to appeal board |
Free format text: JAPANESE INTERMEDIATE CODE: A912 Effective date: 20100423 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20111017 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20111020 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20120202 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20120613 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 Ref document number: 5020463 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20150622 Year of fee payment: 3 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |