JP2005298625A - ポリアミド酸およびポリイミド - Google Patents
ポリアミド酸およびポリイミド Download PDFInfo
- Publication number
- JP2005298625A JP2005298625A JP2004115197A JP2004115197A JP2005298625A JP 2005298625 A JP2005298625 A JP 2005298625A JP 2004115197 A JP2004115197 A JP 2004115197A JP 2004115197 A JP2004115197 A JP 2004115197A JP 2005298625 A JP2005298625 A JP 2005298625A
- Authority
- JP
- Japan
- Prior art keywords
- group
- polyimide
- polyamic acid
- substituted
- unsubstituted
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 229920001721 polyimide Polymers 0.000 title claims abstract description 69
- 239000004642 Polyimide Substances 0.000 title claims abstract description 63
- 229920005575 poly(amic acid) Polymers 0.000 title claims abstract description 55
- -1 polycyclic hydrocarbon Chemical class 0.000 claims description 15
- 125000002950 monocyclic group Chemical group 0.000 claims description 13
- 125000003545 alkoxy group Chemical group 0.000 claims description 6
- 125000000217 alkyl group Chemical group 0.000 claims description 6
- 125000000623 heterocyclic group Chemical group 0.000 claims description 6
- 229920000642 polymer Polymers 0.000 claims description 6
- KPPVNWGJXFMGAM-UUILKARUSA-N (e)-2-methyl-1-(6-methyl-3,4-dihydro-2h-quinolin-1-yl)but-2-en-1-one Chemical compound CC1=CC=C2N(C(=O)C(/C)=C/C)CCCC2=C1 KPPVNWGJXFMGAM-UUILKARUSA-N 0.000 claims description 5
- 125000003118 aryl group Chemical group 0.000 claims description 5
- 125000000732 arylene group Chemical group 0.000 claims description 5
- 125000002993 cycloalkylene group Chemical group 0.000 claims description 5
- 125000005843 halogen group Chemical group 0.000 claims description 5
- CREMABGTGYGIQB-UHFFFAOYSA-N carbon carbon Chemical compound C.C CREMABGTGYGIQB-UHFFFAOYSA-N 0.000 claims description 4
- 239000011203 carbon fibre reinforced carbon Substances 0.000 claims description 4
- 229930195733 hydrocarbon Natural products 0.000 claims description 4
- 238000006467 substitution reaction Methods 0.000 claims description 4
- 239000004215 Carbon black (E152) Substances 0.000 claims description 3
- 239000002243 precursor Substances 0.000 abstract description 4
- 150000001875 compounds Chemical class 0.000 description 25
- 239000010408 film Substances 0.000 description 14
- 239000002904 solvent Substances 0.000 description 14
- 238000000034 method Methods 0.000 description 12
- 150000000000 tetracarboxylic acids Chemical class 0.000 description 12
- 150000004985 diamines Chemical class 0.000 description 11
- 230000015572 biosynthetic process Effects 0.000 description 9
- 230000000052 comparative effect Effects 0.000 description 9
- CYIDZMCFTVVTJO-UHFFFAOYSA-N pyromellitic acid Chemical compound OC(=O)C1=CC(C(O)=O)=C(C(O)=O)C=C1C(O)=O CYIDZMCFTVVTJO-UHFFFAOYSA-N 0.000 description 9
- 238000003786 synthesis reaction Methods 0.000 description 9
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 8
- 125000004432 carbon atom Chemical group C* 0.000 description 8
- 239000011248 coating agent Substances 0.000 description 8
- 238000000576 coating method Methods 0.000 description 8
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 7
- 239000002253 acid Substances 0.000 description 7
- 239000000758 substrate Substances 0.000 description 7
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 6
- 239000000835 fiber Substances 0.000 description 6
- 238000010438 heat treatment Methods 0.000 description 6
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 6
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 5
- 238000006243 chemical reaction Methods 0.000 description 5
- 238000005259 measurement Methods 0.000 description 5
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- 239000011521 glass Substances 0.000 description 4
- 238000002329 infrared spectrum Methods 0.000 description 4
- 239000000377 silicon dioxide Substances 0.000 description 4
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 3
- 239000004952 Polyamide Substances 0.000 description 3
- 150000008065 acid anhydrides Chemical class 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- 230000009477 glass transition Effects 0.000 description 3
- 239000011229 interlayer Substances 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 239000012299 nitrogen atmosphere Substances 0.000 description 3
- 239000011368 organic material Substances 0.000 description 3
- 230000003071 parasitic effect Effects 0.000 description 3
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 3
- 229920002647 polyamide Polymers 0.000 description 3
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 2
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 2
- NUIURNJTPRWVAP-UHFFFAOYSA-N 3,3'-Dimethylbenzidine Chemical compound C1=C(N)C(C)=CC(C=2C=C(C)C(N)=CC=2)=C1 NUIURNJTPRWVAP-UHFFFAOYSA-N 0.000 description 2
- QQWWWAQUMVHHQN-UHFFFAOYSA-N 4-(4-amino-4-phenylcyclohexa-1,5-dien-1-yl)aniline Chemical group C1=CC(N)=CC=C1C1=CCC(N)(C=2C=CC=CC=2)C=C1 QQWWWAQUMVHHQN-UHFFFAOYSA-N 0.000 description 2
- CRXDDVBCCDDHPX-UHFFFAOYSA-N 4-ethynyl-5-phenyl-2-benzofuran-1,3-dione Chemical compound O=C1OC(=O)C(C=2C#C)=C1C=CC=2C1=CC=CC=C1 CRXDDVBCCDDHPX-UHFFFAOYSA-N 0.000 description 2
- VVJKKWFAADXIJK-UHFFFAOYSA-N Allylamine Chemical compound NCC=C VVJKKWFAADXIJK-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- 229920000049 Carbon (fiber) Polymers 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- BHHGXPLMPWCGHP-UHFFFAOYSA-N Phenethylamine Chemical compound NCCC1=CC=CC=C1 BHHGXPLMPWCGHP-UHFFFAOYSA-N 0.000 description 2
- LGRFSURHDFAFJT-UHFFFAOYSA-N Phthalic anhydride Natural products C1=CC=C2C(=O)OC(=O)C2=C1 LGRFSURHDFAFJT-UHFFFAOYSA-N 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- 239000004721 Polyphenylene oxide Substances 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 2
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 2
- ADCOVFLJGNWWNZ-UHFFFAOYSA-N antimony trioxide Chemical compound O=[Sb]O[Sb]=O ADCOVFLJGNWWNZ-UHFFFAOYSA-N 0.000 description 2
- 239000010425 asbestos Substances 0.000 description 2
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 2
- WGQKYBSKWIADBV-UHFFFAOYSA-N benzylamine Chemical compound NCC1=CC=CC=C1 WGQKYBSKWIADBV-UHFFFAOYSA-N 0.000 description 2
- JHIWVOJDXOSYLW-UHFFFAOYSA-N butyl 2,2-difluorocyclopropane-1-carboxylate Chemical compound CCCCOC(=O)C1CC1(F)F JHIWVOJDXOSYLW-UHFFFAOYSA-N 0.000 description 2
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 2
- 239000004917 carbon fiber Substances 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 238000004040 coloring Methods 0.000 description 2
- ARUKYTASOALXFG-UHFFFAOYSA-N cycloheptylcycloheptane Chemical compound C1CCCCCC1C1CCCCCC1 ARUKYTASOALXFG-UHFFFAOYSA-N 0.000 description 2
- NLUNLVTVUDIHFE-UHFFFAOYSA-N cyclooctylcyclooctane Chemical compound C1CCCCCCC1C1CCCCCCC1 NLUNLVTVUDIHFE-UHFFFAOYSA-N 0.000 description 2
- 239000012024 dehydrating agents Substances 0.000 description 2
- 150000001991 dicarboxylic acids Chemical class 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 239000010439 graphite Substances 0.000 description 2
- 229910002804 graphite Inorganic materials 0.000 description 2
- 239000011810 insulating material Substances 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- RBYAGNRAPXMZGZ-UHFFFAOYSA-N n-(2-phenylethynyl)aniline Chemical compound C=1C=CC=CC=1NC#CC1=CC=CC=C1 RBYAGNRAPXMZGZ-UHFFFAOYSA-N 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- YNPNZTXNASCQKK-UHFFFAOYSA-N phenanthrene Chemical compound C1=CC=C2C3=CC=CC=C3C=CC2=C1 YNPNZTXNASCQKK-UHFFFAOYSA-N 0.000 description 2
- 125000003367 polycyclic group Chemical group 0.000 description 2
- 239000009719 polyimide resin Substances 0.000 description 2
- 238000007639 printing Methods 0.000 description 2
- 125000003373 pyrazinyl group Chemical group 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 229920005989 resin Polymers 0.000 description 2
- 239000011347 resin Substances 0.000 description 2
- 229910052895 riebeckite Inorganic materials 0.000 description 2
- 239000004065 semiconductor Substances 0.000 description 2
- 229910010271 silicon carbide Inorganic materials 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 238000005979 thermal decomposition reaction Methods 0.000 description 2
- FBZXHOSXGAGXNJ-UHFFFAOYSA-N (2-phenylbenzoyl) 2-phenylbenzoate Chemical compound C=1C=CC=C(C=2C=CC=CC=2)C=1C(=O)OC(=O)C1=CC=CC=C1C1=CC=CC=C1 FBZXHOSXGAGXNJ-UHFFFAOYSA-N 0.000 description 1
- KXBLYKUKRIUXKV-UHFFFAOYSA-N 1,2,3,4-tetrakis(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=C(C(F)(F)F)C(C(F)(F)F)=C1C(F)(F)F KXBLYKUKRIUXKV-UHFFFAOYSA-N 0.000 description 1
- FKASFBLJDCHBNZ-UHFFFAOYSA-N 1,3,4-oxadiazole Chemical compound C1=NN=CO1 FKASFBLJDCHBNZ-UHFFFAOYSA-N 0.000 description 1
- 125000005655 1,3-cyclohexylene group Chemical group [H]C1([H])C([H])([H])C([H])([*:1])C([H])([H])C([H])([*:2])C1([H])[H] 0.000 description 1
- 125000001989 1,3-phenylene group Chemical group [H]C1=C([H])C([*:1])=C([H])C([*:2])=C1[H] 0.000 description 1
- 125000004955 1,4-cyclohexylene group Chemical group [H]C1([H])C([H])([H])C([H])([*:1])C([H])([H])C([H])([H])C1([H])[*:2] 0.000 description 1
- 125000001140 1,4-phenylene group Chemical group [H]C1=C([H])C([*:2])=C([H])C([H])=C1[*:1] 0.000 description 1
- CBCKQZAAMUWICA-UHFFFAOYSA-N 1,4-phenylenediamine Chemical compound NC1=CC=C(N)C=C1 CBCKQZAAMUWICA-UHFFFAOYSA-N 0.000 description 1
- IANQTJSKSUMEQM-UHFFFAOYSA-N 1-benzofuran Chemical group C1=CC=C2OC=CC2=C1 IANQTJSKSUMEQM-UHFFFAOYSA-N 0.000 description 1
- VLDPXPPHXDGHEW-UHFFFAOYSA-N 1-chloro-2-dichlorophosphoryloxybenzene Chemical compound ClC1=CC=CC=C1OP(Cl)(Cl)=O VLDPXPPHXDGHEW-UHFFFAOYSA-N 0.000 description 1
- 125000004825 2,2-dimethylpropylene group Chemical group [H]C([H])([H])C(C([H])([H])[H])(C([H])([H])[*:1])C([H])([H])[*:2] 0.000 description 1
- CDULGHZNHURECF-UHFFFAOYSA-N 2,3-dimethylaniline 2,4-dimethylaniline 2,5-dimethylaniline 2,6-dimethylaniline 3,4-dimethylaniline 3,5-dimethylaniline Chemical group CC1=CC=C(N)C(C)=C1.CC1=CC=C(C)C(N)=C1.CC1=CC(C)=CC(N)=C1.CC1=CC=C(N)C=C1C.CC1=CC=CC(N)=C1C.CC1=CC=CC(C)=C1N CDULGHZNHURECF-UHFFFAOYSA-N 0.000 description 1
- 125000004959 2,6-naphthylene group Chemical group [H]C1=C([H])C2=C([H])C([*:1])=C([H])C([H])=C2C([H])=C1[*:2] 0.000 description 1
- SBASXUCJHJRPEV-UHFFFAOYSA-N 2-(2-methoxyethoxy)ethanol Chemical compound COCCOCCO SBASXUCJHJRPEV-UHFFFAOYSA-N 0.000 description 1
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical compound ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 description 1
- HLCPWBZNUKCSBN-UHFFFAOYSA-N 2-aminobenzonitrile Chemical compound NC1=CC=CC=C1C#N HLCPWBZNUKCSBN-UHFFFAOYSA-N 0.000 description 1
- CDAWCLOXVUBKRW-UHFFFAOYSA-N 2-aminophenol Chemical compound NC1=CC=CC=C1O CDAWCLOXVUBKRW-UHFFFAOYSA-N 0.000 description 1
- TWBPWBPGNQWFSJ-UHFFFAOYSA-N 2-phenylaniline Chemical group NC1=CC=CC=C1C1=CC=CC=C1 TWBPWBPGNQWFSJ-UHFFFAOYSA-N 0.000 description 1
- JRBJSXQPQWSCCF-UHFFFAOYSA-N 3,3'-Dimethoxybenzidine Chemical compound C1=C(N)C(OC)=CC(C=2C=C(OC)C(N)=CC=2)=C1 JRBJSXQPQWSCCF-UHFFFAOYSA-N 0.000 description 1
- NBAUUNCGSMAPFM-UHFFFAOYSA-N 3-(3,4-dicarboxyphenyl)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C1=CC=CC(C(O)=O)=C1C(O)=O NBAUUNCGSMAPFM-UHFFFAOYSA-N 0.000 description 1
- LXJLFVRAWOOQDR-UHFFFAOYSA-N 3-(3-aminophenoxy)aniline Chemical compound NC1=CC=CC(OC=2C=C(N)C=CC=2)=C1 LXJLFVRAWOOQDR-UHFFFAOYSA-N 0.000 description 1
- NDXGRHCEHPFUSU-UHFFFAOYSA-N 3-(3-aminophenyl)aniline Chemical group NC1=CC=CC(C=2C=C(N)C=CC=2)=C1 NDXGRHCEHPFUSU-UHFFFAOYSA-N 0.000 description 1
- GIDZGEJVGCDPLV-UHFFFAOYSA-N 3-[2-(2,3-dicarboxyphenoxy)-3,4-bis(trifluoromethyl)phenoxy]phthalic acid Chemical compound OC(=O)C1=CC=CC(OC=2C(=C(C(=CC=2)C(F)(F)F)C(F)(F)F)OC=2C(=C(C(O)=O)C=CC=2)C(O)=O)=C1C(O)=O GIDZGEJVGCDPLV-UHFFFAOYSA-N 0.000 description 1
- ICNFHJVPAJKPHW-UHFFFAOYSA-N 4,4'-Thiodianiline Chemical compound C1=CC(N)=CC=C1SC1=CC=C(N)C=C1 ICNFHJVPAJKPHW-UHFFFAOYSA-N 0.000 description 1
- YBRVSVVVWCFQMG-UHFFFAOYSA-N 4,4'-diaminodiphenylmethane Chemical compound C1=CC(N)=CC=C1CC1=CC=C(N)C=C1 YBRVSVVVWCFQMG-UHFFFAOYSA-N 0.000 description 1
- AXMANIZPMQZKTG-UHFFFAOYSA-N 4-(2-phenylethynyl)-2-benzofuran-1,3-dione Chemical compound O=C1OC(=O)C2=C1C=CC=C2C#CC1=CC=CC=C1 AXMANIZPMQZKTG-UHFFFAOYSA-N 0.000 description 1
- UITKHKNFVCYWNG-UHFFFAOYSA-N 4-(3,4-dicarboxybenzoyl)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C(=O)C1=CC=C(C(O)=O)C(C(O)=O)=C1 UITKHKNFVCYWNG-UHFFFAOYSA-N 0.000 description 1
- LFBALUPVVFCEPA-UHFFFAOYSA-N 4-(3,4-dicarboxyphenyl)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C1=CC=C(C(O)=O)C(C(O)=O)=C1 LFBALUPVVFCEPA-UHFFFAOYSA-N 0.000 description 1
- FWOLORXQTIGHFX-UHFFFAOYSA-N 4-(4-amino-2,3,5,6-tetrafluorophenyl)-2,3,5,6-tetrafluoroaniline Chemical compound FC1=C(F)C(N)=C(F)C(F)=C1C1=C(F)C(F)=C(N)C(F)=C1F FWOLORXQTIGHFX-UHFFFAOYSA-N 0.000 description 1
- QYIMZXITLDTULQ-UHFFFAOYSA-N 4-(4-amino-2-methylphenyl)-3-methylaniline Chemical compound CC1=CC(N)=CC=C1C1=CC=C(N)C=C1C QYIMZXITLDTULQ-UHFFFAOYSA-N 0.000 description 1
- HLBLWEWZXPIGSM-UHFFFAOYSA-N 4-Aminophenyl ether Chemical compound C1=CC(N)=CC=C1OC1=CC=C(N)C=C1 HLBLWEWZXPIGSM-UHFFFAOYSA-N 0.000 description 1
- HNHQPIBXQALMMN-UHFFFAOYSA-N 4-[(3,4-dicarboxyphenyl)-dimethylsilyl]phthalic acid Chemical compound C=1C=C(C(O)=O)C(C(O)=O)=CC=1[Si](C)(C)C1=CC=C(C(O)=O)C(C(O)=O)=C1 HNHQPIBXQALMMN-UHFFFAOYSA-N 0.000 description 1
- DZIHTWJGPDVSGE-UHFFFAOYSA-N 4-[(4-aminocyclohexyl)methyl]cyclohexan-1-amine Chemical compound C1CC(N)CCC1CC1CCC(N)CC1 DZIHTWJGPDVSGE-UHFFFAOYSA-N 0.000 description 1
- APXJLYIVOFARRM-UHFFFAOYSA-N 4-[2-(3,4-dicarboxyphenyl)-1,1,1,3,3,3-hexafluoropropan-2-yl]phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C(C(F)(F)F)(C(F)(F)F)C1=CC=C(C(O)=O)C(C(O)=O)=C1 APXJLYIVOFARRM-UHFFFAOYSA-N 0.000 description 1
- GEYAGBVEAJGCFB-UHFFFAOYSA-N 4-[2-(3,4-dicarboxyphenyl)propan-2-yl]phthalic acid Chemical compound C=1C=C(C(O)=O)C(C(O)=O)=CC=1C(C)(C)C1=CC=C(C(O)=O)C(C(O)=O)=C1 GEYAGBVEAJGCFB-UHFFFAOYSA-N 0.000 description 1
- CQHNNQBNJNKIGT-UHFFFAOYSA-N 4-[3,4-dicarboxy-2,5-bis(trifluoromethyl)phenyl]-3,6-bis(trifluoromethyl)phthalic acid Chemical group C1=C(C(F)(F)F)C(C(=O)O)=C(C(O)=O)C(C(F)(F)F)=C1C1=CC(C(F)(F)F)=C(C(O)=O)C(C(O)=O)=C1C(F)(F)F CQHNNQBNJNKIGT-UHFFFAOYSA-N 0.000 description 1
- JEDMLZGYTDOQTD-UHFFFAOYSA-N 4-[4-(3,4-dicarboxy-2,5,6-trifluorophenoxy)-2,3,5,6-tetrafluoro-4-(2,3,4,5-tetrafluorophenyl)cyclohexa-1,5-dien-1-yl]oxy-3,5,6-trifluorophthalic acid Chemical group FC1=C(C(O)=O)C(C(=O)O)=C(F)C(F)=C1OC(C(=C1F)F)=C(F)C(F)C1(C=1C(=C(F)C(F)=C(F)C=1)F)OC1=C(F)C(F)=C(C(O)=O)C(C(O)=O)=C1F JEDMLZGYTDOQTD-UHFFFAOYSA-N 0.000 description 1
- DXPDSWSYCBNHTO-UHFFFAOYSA-N 4-[4-(3,4-dicarboxy-2,5,6-trifluorophenoxy)-2,3,5,6-tetrafluorophenoxy]-3,5,6-trifluorophthalic acid Chemical compound FC1=C(C(O)=O)C(C(=O)O)=C(F)C(F)=C1OC(C(=C1F)F)=C(F)C(F)=C1OC1=C(F)C(F)=C(C(O)=O)C(C(O)=O)=C1F DXPDSWSYCBNHTO-UHFFFAOYSA-N 0.000 description 1
- IOUVQFAYPGDXFG-UHFFFAOYSA-N 4-[4-[2-[4-(3,4-dicarboxyphenoxy)phenyl]-1,1,1,3,3,3-hexafluoropropan-2-yl]phenoxy]phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1OC1=CC=C(C(C=2C=CC(OC=3C=C(C(C(O)=O)=CC=3)C(O)=O)=CC=2)(C(F)(F)F)C(F)(F)F)C=C1 IOUVQFAYPGDXFG-UHFFFAOYSA-N 0.000 description 1
- KJLPSBMDOIVXSN-UHFFFAOYSA-N 4-[4-[2-[4-(3,4-dicarboxyphenoxy)phenyl]propan-2-yl]phenoxy]phthalic acid Chemical compound C=1C=C(OC=2C=C(C(C(O)=O)=CC=2)C(O)=O)C=CC=1C(C)(C)C(C=C1)=CC=C1OC1=CC=C(C(O)=O)C(C(O)=O)=C1 KJLPSBMDOIVXSN-UHFFFAOYSA-N 0.000 description 1
- AUPIFOPXTAGGSW-UHFFFAOYSA-N 4-[4-[9-[4-(4-aminophenoxy)phenyl]fluoren-9-yl]phenoxy]aniline Chemical compound C1=CC(N)=CC=C1OC1=CC=C(C2(C3=CC=CC=C3C3=CC=CC=C32)C=2C=CC(OC=3C=CC(N)=CC=3)=CC=2)C=C1 AUPIFOPXTAGGSW-UHFFFAOYSA-N 0.000 description 1
- XZOQPRNOAGCWNT-UHFFFAOYSA-N 4-[[(3,4-dicarboxyphenyl)-dimethylsilyl]oxy-dimethylsilyl]phthalic acid Chemical compound C=1C=C(C(O)=O)C(C(O)=O)=CC=1[Si](C)(C)O[Si](C)(C)C1=CC=C(C(O)=O)C(C(O)=O)=C1 XZOQPRNOAGCWNT-UHFFFAOYSA-N 0.000 description 1
- DOWVFPAIJRADGF-UHFFFAOYSA-N 4-ethenyl-2-benzofuran-1,3-dione Chemical compound C=CC1=CC=CC2=C1C(=O)OC2=O DOWVFPAIJRADGF-UHFFFAOYSA-N 0.000 description 1
- VCJUSEFXUWAMHH-UHFFFAOYSA-N 4-ethynyl-2-benzofuran-1,3-dione Chemical compound C1=CC=C(C#C)C2=C1C(=O)OC2=O VCJUSEFXUWAMHH-UHFFFAOYSA-N 0.000 description 1
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 1
- VQVIHDPBMFABCQ-UHFFFAOYSA-N 5-(1,3-dioxo-2-benzofuran-5-carbonyl)-2-benzofuran-1,3-dione Chemical compound C1=C2C(=O)OC(=O)C2=CC(C(C=2C=C3C(=O)OC(=O)C3=CC=2)=O)=C1 VQVIHDPBMFABCQ-UHFFFAOYSA-N 0.000 description 1
- ZHBXLZQQVCDGPA-UHFFFAOYSA-N 5-[(1,3-dioxo-2-benzofuran-5-yl)sulfonyl]-2-benzofuran-1,3-dione Chemical compound C1=C2C(=O)OC(=O)C2=CC(S(=O)(=O)C=2C=C3C(=O)OC(C3=CC=2)=O)=C1 ZHBXLZQQVCDGPA-UHFFFAOYSA-N 0.000 description 1
- YSKGYNLAQHUZHZ-UHFFFAOYSA-N 5-[3,4-dicarboxy-5-(trifluoromethyl)phenyl]-3-(trifluoromethyl)phthalic acid Chemical group FC(F)(F)C1=C(C(O)=O)C(C(=O)O)=CC(C=2C=C(C(C(O)=O)=C(C(O)=O)C=2)C(F)(F)F)=C1 YSKGYNLAQHUZHZ-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 1
- IEAWJWWOUCYCAO-UHFFFAOYSA-N C(=O)(O)C1=CC=C(C2=C(C(=CC=C12)C(=O)O)C(=O)O)C(=O)O.C(=O)(O)C1=CC=C(C2=C(C=C(C=C12)C(=O)O)C(=O)O)C(=O)O Chemical compound C(=O)(O)C1=CC=C(C2=C(C(=CC=C12)C(=O)O)C(=O)O)C(=O)O.C(=O)(O)C1=CC=C(C2=C(C=C(C=C12)C(=O)O)C(=O)O)C(=O)O IEAWJWWOUCYCAO-UHFFFAOYSA-N 0.000 description 1
- CDJHSRCGUYUQLZ-UHFFFAOYSA-N C(=O)(O)C=1C=C(C=CC1C(=O)O)C(=O)NC1=CC=C(C=C1)C1=C2C(=C3CC4=CC=CC=C4C3=C1)C(=O)OC2=O Chemical compound C(=O)(O)C=1C=C(C=CC1C(=O)O)C(=O)NC1=CC=C(C=C1)C1=C2C(=C3CC4=CC=CC=C4C3=C1)C(=O)OC2=O CDJHSRCGUYUQLZ-UHFFFAOYSA-N 0.000 description 1
- 0 CC*(CC)N(C(c1c2cc(*c3ccc(C4(c5ccc(*c6cc(C)c(C)c(C(*(**7(CC)CC7)=O)=O)c6)cc5)c5ccccc5CC=CC=C[C@@]4C)cc3)cc1)=*)C2=O Chemical compound CC*(CC)N(C(c1c2cc(*c3ccc(C4(c5ccc(*c6cc(C)c(C)c(C(*(**7(CC)CC7)=O)=O)c6)cc5)c5ccccc5CC=CC=C[C@@]4C)cc3)cc1)=*)C2=O 0.000 description 1
- FVULUUNGIFGTJH-UHFFFAOYSA-N Cc1cc(C2(c3ccccc3-c3ccccc23)c(cc2)cc(C)c2OCCOc(cc2C(O3)=O)ccc2C3=O)ccc1OCCOc(cc1C(O2)=O)ccc1C2=O Chemical compound Cc1cc(C2(c3ccccc3-c3ccccc23)c(cc2)cc(C)c2OCCOc(cc2C(O3)=O)ccc2C3=O)ccc1OCCOc(cc1C(O2)=O)ccc1C2=O FVULUUNGIFGTJH-UHFFFAOYSA-N 0.000 description 1
- 241000195493 Cryptophyta Species 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- MQJKPEGWNLWLTK-UHFFFAOYSA-N Dapsone Chemical compound C1=CC(N)=CC=C1S(=O)(=O)C1=CC=C(N)C=C1 MQJKPEGWNLWLTK-UHFFFAOYSA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- GELSXJZLUISGEW-UHFFFAOYSA-N FC(F)(F)C1=C(C(=C(OC2=C(C=CC=C2)OC2=C(C(=C(C=C2)C(F)(F)F)C(=O)O)C(=O)O)C=C1)C(=O)O)C(=O)O.FC(C=1C(=C(C=C(C(=O)C2=CC(=C(C(=C2)C(F)(F)F)C(=O)O)C(=O)O)C1)C(=O)O)C(=O)O)(F)F Chemical compound FC(F)(F)C1=C(C(=C(OC2=C(C=CC=C2)OC2=C(C(=C(C=C2)C(F)(F)F)C(=O)O)C(=O)O)C=C1)C(=O)O)C(=O)O.FC(C=1C(=C(C=C(C(=O)C2=CC(=C(C(=C2)C(F)(F)F)C(=O)O)C(=O)O)C1)C(=O)O)C(=O)O)(F)F GELSXJZLUISGEW-UHFFFAOYSA-N 0.000 description 1
- DLEPYXFUDLQGDW-UHFFFAOYSA-N FC(F)(F)NC1=CC=C(C2=CC=C(NC(F)(F)F)C=C2)C=C1 Chemical compound FC(F)(F)NC1=CC=C(C2=CC=C(NC(F)(F)F)C=C2)C=C1 DLEPYXFUDLQGDW-UHFFFAOYSA-N 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 1
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical group C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 1
- 239000004962 Polyamide-imide Substances 0.000 description 1
- 239000004695 Polyether sulfone Substances 0.000 description 1
- 239000004697 Polyetherimide Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000004734 Polyphenylene sulfide Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- GTDPSWPPOUPBNX-UHFFFAOYSA-N ac1mqpva Chemical compound CC12C(=O)OC(=O)C1(C)C1(C)C2(C)C(=O)OC1=O GTDPSWPPOUPBNX-UHFFFAOYSA-N 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 239000002313 adhesive film Substances 0.000 description 1
- 239000002390 adhesive tape Substances 0.000 description 1
- 230000005260 alpha ray Effects 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- YUENFNPLGJCNRB-UHFFFAOYSA-N anthracen-1-amine Chemical compound C1=CC=C2C=C3C(N)=CC=CC3=CC2=C1 YUENFNPLGJCNRB-UHFFFAOYSA-N 0.000 description 1
- 150000004984 aromatic diamines Chemical class 0.000 description 1
- 239000011805 ball Substances 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- HFACYLZERDEVSX-UHFFFAOYSA-N benzidine Chemical compound C1=CC(N)=CC=C1C1=CC=C(N)C=C1 HFACYLZERDEVSX-UHFFFAOYSA-N 0.000 description 1
- IZJDCINIYIMFGX-UHFFFAOYSA-N benzo[f][2]benzofuran-1,3-dione Chemical compound C1=CC=C2C=C3C(=O)OC(=O)C3=CC2=C1 IZJDCINIYIMFGX-UHFFFAOYSA-N 0.000 description 1
- 125000002529 biphenylenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C12)* 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 229910052796 boron Inorganic materials 0.000 description 1
- WKDNYTOXBCRNPV-UHFFFAOYSA-N bpda Chemical compound C1=C2C(=O)OC(=O)C2=CC(C=2C=C3C(=O)OC(C3=CC=2)=O)=C1 WKDNYTOXBCRNPV-UHFFFAOYSA-N 0.000 description 1
- 229910052918 calcium silicate Inorganic materials 0.000 description 1
- 235000012241 calcium silicate Nutrition 0.000 description 1
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 description 1
- 239000003990 capacitor Substances 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 150000001244 carboxylic acid anhydrides Chemical class 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 229910052570 clay Inorganic materials 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 239000012787 coverlay film Substances 0.000 description 1
- CURBACXRQKTCKZ-UHFFFAOYSA-N cyclobutane-1,2,3,4-tetracarboxylic acid Chemical compound OC(=O)C1C(C(O)=O)C(C(O)=O)C1C(O)=O CURBACXRQKTCKZ-UHFFFAOYSA-N 0.000 description 1
- ILUAAIDVFMVTAU-UHFFFAOYSA-N cyclohex-4-ene-1,2-dicarboxylic acid Chemical compound OC(=O)C1CC=CCC1C(O)=O ILUAAIDVFMVTAU-UHFFFAOYSA-N 0.000 description 1
- VKIRRGRTJUUZHS-UHFFFAOYSA-N cyclohexane-1,4-diamine Chemical compound NC1CCC(N)CC1 VKIRRGRTJUUZHS-UHFFFAOYSA-N 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- STZIXLPVKZUAMV-UHFFFAOYSA-N cyclopentane-1,1,2,2-tetracarboxylic acid Chemical compound OC(=O)C1(C(O)=O)CCCC1(C(O)=O)C(O)=O STZIXLPVKZUAMV-UHFFFAOYSA-N 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 238000010292 electrical insulation Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000003063 flame retardant Substances 0.000 description 1
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 239000003365 glass fiber Substances 0.000 description 1
- 125000006343 heptafluoro propyl group Chemical group 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- RTWNYYOXLSILQN-UHFFFAOYSA-N methanediamine Chemical compound NCN RTWNYYOXLSILQN-UHFFFAOYSA-N 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 229910052618 mica group Inorganic materials 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 239000012778 molding material Substances 0.000 description 1
- CWQXQMHSOZUFJS-UHFFFAOYSA-N molybdenum disulfide Chemical compound S=[Mo]=S CWQXQMHSOZUFJS-UHFFFAOYSA-N 0.000 description 1
- 229910052982 molybdenum disulfide Inorganic materials 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- KUDPGZONDFORKU-UHFFFAOYSA-N n-chloroaniline Chemical compound ClNC1=CC=CC=C1 KUDPGZONDFORKU-UHFFFAOYSA-N 0.000 description 1
- VBEGHXKAFSLLGE-UHFFFAOYSA-N n-phenylnitramide Chemical compound [O-][N+](=O)NC1=CC=CC=C1 VBEGHXKAFSLLGE-UHFFFAOYSA-N 0.000 description 1
- KQSABULTKYLFEV-UHFFFAOYSA-N naphthalene-1,5-diamine Chemical compound C1=CC=C2C(N)=CC=CC2=C1N KQSABULTKYLFEV-UHFFFAOYSA-N 0.000 description 1
- DOBFTMLCEYUAQC-UHFFFAOYSA-N naphthalene-2,3,6,7-tetracarboxylic acid Chemical compound OC(=O)C1=C(C(O)=O)C=C2C=C(C(O)=O)C(C(=O)O)=CC2=C1 DOBFTMLCEYUAQC-UHFFFAOYSA-N 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- BHAAPTBBJKJZER-UHFFFAOYSA-N p-anisidine Chemical compound COC1=CC=C(N)C=C1 BHAAPTBBJKJZER-UHFFFAOYSA-N 0.000 description 1
- 238000002161 passivation Methods 0.000 description 1
- FVDOBFPYBSDRKH-UHFFFAOYSA-N perylene-3,4,9,10-tetracarboxylic acid Chemical group C=12C3=CC=C(C(O)=O)C2=C(C(O)=O)C=CC=1C1=CC=C(C(O)=O)C2=C1C3=CC=C2C(=O)O FVDOBFPYBSDRKH-UHFFFAOYSA-N 0.000 description 1
- 229940117803 phenethylamine Drugs 0.000 description 1
- 229920001643 poly(ether ketone) Polymers 0.000 description 1
- 229920002492 poly(sulfone) Polymers 0.000 description 1
- 229920002312 polyamide-imide Polymers 0.000 description 1
- 229920001230 polyarylate Polymers 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 125000004585 polycyclic heterocycle group Chemical group 0.000 description 1
- 229920000570 polyether Polymers 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 229920001601 polyetherimide Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920006380 polyphenylene oxide Polymers 0.000 description 1
- 229920000069 polyphenylene sulfide Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 1
- 239000004810 polytetrafluoroethylene Substances 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- VHNQIURBCCNWDN-UHFFFAOYSA-N pyridine-2,6-diamine Chemical compound NC1=CC=CC(N)=N1 VHNQIURBCCNWDN-UHFFFAOYSA-N 0.000 description 1
- YKWDNEXDHDSTCU-UHFFFAOYSA-N pyrrolidine-2,3,4,5-tetracarboxylic acid Chemical compound OC(=O)C1NC(C(O)=O)C(C(O)=O)C1C(O)=O YKWDNEXDHDSTCU-UHFFFAOYSA-N 0.000 description 1
- 239000012779 reinforcing material Substances 0.000 description 1
- 239000011342 resin composition Substances 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 description 1
- 238000004528 spin coating Methods 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 239000004575 stone Substances 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 229940014800 succinic anhydride Drugs 0.000 description 1
- 150000003457 sulfones Chemical class 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 125000006158 tetracarboxylic acid group Chemical group 0.000 description 1
- 229920005992 thermoplastic resin Polymers 0.000 description 1
- LUEGQDUCMILDOJ-UHFFFAOYSA-N thiophene-2,3,4,5-tetracarboxylic acid Chemical compound OC(=O)C=1SC(C(O)=O)=C(C(O)=O)C=1C(O)=O LUEGQDUCMILDOJ-UHFFFAOYSA-N 0.000 description 1
- 150000004992 toluidines Chemical class 0.000 description 1
- 238000005292 vacuum distillation Methods 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Landscapes
- Macromolecular Compounds Obtained By Forming Nitrogen-Containing Linkages In General (AREA)
- Macromonomer-Based Addition Polymer (AREA)
Abstract
Description
すなわち、本発明の課題は、下記一般式(1)で表される繰り返し単位を有するポリアミド酸により達成される。
以下、下記一般式(1)で表される繰り返し単位を有するポリアミド酸について説明する。
(トリフルオロメチル)ピロメリット酸、ジ(トリフルオロメチル)ピロメリット酸、ジ(ヘプタフルオロプロピル)ピロメリット酸、ペンタフルオロエチルピロメリット酸、ビス〔3、5−ジ(トリフルオロメチル)フェノキシ〕ピロメリット酸、2,3,3’,4’−ビフェニルテトラカルボン酸、3,3’,4,4’−ビフェニルテトラカルボン酸、3,3’,4,4’−テトラカルボキシジフェニルエーテル、2,3’,3,4’−テトラカルボキシジフェニルエーテル、3,3’,4,4’−ベンゾフェノンテトラカルボン酸、2,3,6,7−テトラカルボキシナフタレン、1,4,5,7−テトラカルボキシナフタレン、1,4,5,6−テトラカルボキシナフタレン、3,3’,4,4’−テトラカルボキシジフェニルメタン、3,3’,4,4’−テトラカルボキシジフェニルスルホン、2,2−ビス(3,4−ジカルボキシフェニル)プロパン、2,2−ビス(3,4−ジカルボキシフェニル)ヘキサフルオロプロパン、5,5’−ビス(トリフルオロメチル)−3,3’,4,4’−テトラカルボキシビフェニル、2,2’,5,5’−テトラキス(トリフルオロメチル)−3,3’,4,4’−テトラカルボキシビフェニル、5,5’−ビス(トリフルオロメチル)−3,3’,4,4’−テトラカルボキシジフェニルエーテル、5,5’−ビス(トリフルオロメチル)−3,3’,4,4’−テトラカルボキシベンゾフェノン、ビス〔(トリフルオロメチル)ジカルボキシフェノキシ〕ベンゼン、ビス(ジカルボキシフェノキシ)ビス(トリフルオロメチル)ベンゼン、ビス(ジカルボキシフェノキシ)テトラキス(トリフルオロメチル)ベンゼン、3,4,9,10−テトラカルボキシペリレン、2,2−ビス〔4−(3,4−ジカルボキシフェノキシ)フェニル〕プロパン、シクロブタンテトラカルボン酸、シクロペンタンテトラカルボン酸、2,2−ビス(4−(3,4−ジカルボキシフェノキシ)フェニル)ヘキサフルオロプロパン、ビス(3,4−ジカルボキシフェニル)ジメチルシラン、1,3−ビス(3,4−ジカルボキシフェニル)テトラメチルジシロキサン、ジフルオロピロメリット酸、1,4−ビス(3,4−ジカルボキシトリフルオロフェノキシ)テトラフルオロベンゼン、1,4−ビス(3,4−ジカルボキシトリフルオロフェノキシ)オクタフルオロビフェニル、ピラジン−2,3,5,6−テトラカルボン酸、ピロリジン−2,3,4,5−テトラカルボン酸、チオフェン−2,3,4,5−テトラカルボン酸、ビシクロ[2.2.1]ヘプタ−2,3,5,6−テトラカルボン酸、ビシクロ[2.2.2]オクタ−2,3,5,6−テトラカルボン酸など。
p−フェニレンジアミン、ベンジジン、o−トリジン、m−トリジン、ビス(トリフルオロメチル)ベンジジン、オクタフルオロベンジジン、4,4’−ジアミノ−p−テルフェニル、4,4’−ジアミノジフェニルメタン、4,4’−ジアミノジフェニルスルフィド、3,3’−ジアミノジフェニルスルフィド、4,4’−ジアミノジフェニルスルホン、4,4’−ジアミノジフェニルエーテル、3,3’−ジアミノジフェニルエーテル、3,3’−ジアミノビフェニル、3,3’−ジメチル−4,4’−ジアミノビフェニル、3,3’−ジメトキシベンジジン、4,4’−ジアミノ−p−テルフェニル、ビス(4−アミノシクロヘキシル)メタン、1,5−ジアミノナフタレン、2,6−ジアミノナフタレン、2,6−ジアミノピリジン、2,5−ジアミノ−1,3,4−オキサジアゾール、1,4−ジアミノシクロヘキサン、ピペラジン、メチレンジアミン、エチレンジアミン、2,2−ジメチルプロピレンジアミン、9,9’−ビス(4−アミノフェニル)フルオレンなど。
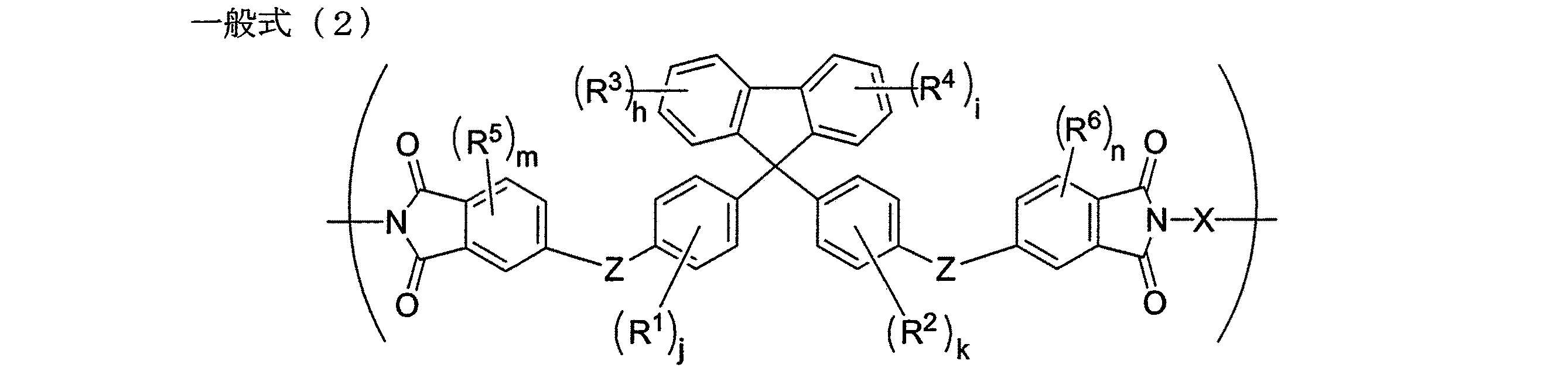
本発明のポリイミドは、下記一般式(2)で表される繰り返し単位を有する。
(1)ポリアミド酸を有機溶剤で溶解してポリアミド酸溶液を調製した後、前記溶剤を減圧蒸留等の手法を用いて低温下で除去するか、ポリアミド酸溶液を貧溶剤に排出する方法でポリアミド酸を単離した後、これを加熱してイミド化を行ってポリイミドを得る方法。
(2)前記ポリアミド酸溶液を調製した後、無水酢酸に代表される脱水剤を加え、さらに必要に応じて触媒を加えて化学的にイミド化を行った後、公知の方法によりポリイミドを単離し、必要に応じて洗浄、乾燥を行う方法。
(3)ポリアミド酸溶液を調製した後、減圧または加熱処理により溶剤を除去すると同時に熱的にイミド化を行う方法。
(4)有機溶剤中にポリアミド酸の原料を混入した後、加熱してポリアミド酸の合成とイミド化反応を同時に行い、必要に応じて触媒や共沸剤、脱水剤を共存させる方法。
本発明のポリイミドフィルムは、ポリイミド溶液を用いる場合、ポリイミド溶液を基体上に塗工し、剥離することにより得られる。また、ポリアミド酸溶液を用いる場合、ポリアミド酸溶液を基体上に塗工し、加熱してイミド化すると、ポリイミド塗膜が得られ、さらにポリイミド塗膜を基体から剥離するとポリイミドフィルムが得られる。例えば、ポリアミド酸溶液を従来公知のスピンコート法、スプレイコート法等を用い、あるいはスリット状ノズルから押し出し、またはバーコーター等により基体上に塗工し、乾燥して溶媒をある程度除去し、剥離可能になった状態で、膜を基体から剥離し、さらに加熱することでポリイミドフィルムが得られる。この際の加熱条件の最大温度は200〜400℃であることが好ましく、250〜350℃であることがより好ましい。加熱温度が200〜400℃の範囲であれば、イミド化が行いやすく、さらには熱による塗膜の変形および劣化をきたしにくい。
(1)例示化合物P−5の合成
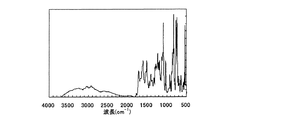
窒素雰囲気下で、9,9’−ビス(4−アミノフェニル)フルオレン24.9gをジメチルアセトアミド88gに溶解した後、15℃で9,9’−ビス〔4−(3,4−ジカルボキシフェニルカルボニルアミノ)フェニル〕フルオレン二酸無水物47.1gを徐々に添加した。20℃で1時間、40℃で5時間反応させた後に、フェニルエチニルフタル酸無水物を0.3g加えて更に、40℃で2時間反応させたところ、透明な溶液が得られた。さらにこの溶液をフィルムアプリケーターを用いてガラス板上に170μmの厚さで流延し、窒素雰囲気下、100℃で2時間、150℃で1時間、200℃で1時間、250℃で1時間、350℃で1時間それぞれ加熱してイミド化を行った。得られたポリイミド塗膜をガラス板上から剥離して例示化合物P−5を得た。例示化合物P−5のIRスペクトルを図1に示す。
図1より波長1640cm-1のピークが減少し、波長1720cm-1にピークが見られるため、例示化合物P−5がポリイミドであることが分かる。
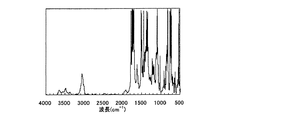
例示化合物P−1と同様の方法により、例示化合物P−12を作製した。得られた例示化合物P−12のIRスペクトルを図2に示す。
図2より波長1660cm-1のピークが消失し、波長1750cm-1のピークが見られるため、例示化合物P−12がポリイミドであることが分かる。
特開平11−116675号公報の実施例2に従い、比較化合物P−41を作製した。
特開平11−116675号公報の比較例に従い、比較化合物P−42を作製した。
(1)ガラス転移温度(Tg)の測定
セイコー(株)製、DSC6200を用いてDSC(窒素中、昇温温度10℃/分)により例示化合物および比較化合物のTgを測定した。結果を表1に示す。
(2)比誘電率(ε)の測定
例示化合物および比較化合物の1MHzにおける静電容量を、横河ヒユーレットパッカード社製のLCRメーター4284Aを用いて測定し、下記式により比誘電率(ε)を求めた。結果を表1に示す。
ε= C・d/(ε0・S)
但し、Cは静電容量、dは試料膜厚、ε0は真空中の誘電率、Sは上部電極面積である。
Claims (4)
- 下記一般式(1)で表される繰り返し単位を有するポリアミド酸。
- 下記一般式(2)で表される繰り返し単位を有するポリイミド。
- ポリマーの末端付近に架橋反応性の炭素−炭素不飽和結合を有する請求項1に記載のポリアミド酸。
- 請求項3に記載のポリアミド酸から誘導されるポリイミド。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004115197A JP4485240B2 (ja) | 2004-04-09 | 2004-04-09 | ポリアミド酸およびポリイミド |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004115197A JP4485240B2 (ja) | 2004-04-09 | 2004-04-09 | ポリアミド酸およびポリイミド |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2005298625A true JP2005298625A (ja) | 2005-10-27 |
| JP4485240B2 JP4485240B2 (ja) | 2010-06-16 |
Family
ID=35330563
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2004115197A Expired - Lifetime JP4485240B2 (ja) | 2004-04-09 | 2004-04-09 | ポリアミド酸およびポリイミド |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP4485240B2 (ja) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007314583A (ja) * | 2006-05-23 | 2007-12-06 | Nissan Chem Ind Ltd | 脂環式ポリイミド前駆体、ポジ型感光性樹脂組成物およびその硬化膜 |
| JP2015155385A (ja) * | 2014-02-20 | 2015-08-27 | 田岡化学工業株式会社 | フルオレン骨格を有するテトラカルボン酸二無水物、ポリアミック酸、及びポリイミド。 |
| CN109207170A (zh) * | 2017-06-30 | 2019-01-15 | 奇美实业股份有限公司 | 液晶配向剂、液晶配向膜及液晶显示元件 |
| JP2019218337A (ja) * | 2018-06-20 | 2019-12-26 | 信越化学工業株式会社 | 化合物及び有機膜形成用組成物 |
| JP2020132532A (ja) * | 2019-02-14 | 2020-08-31 | 田岡化学工業株式会社 | テトラカルボン酸二無水物、ポリアミック酸及びポリイミド |
| US20210040267A1 (en) * | 2017-09-12 | 2021-02-11 | Sk Innovation Co., Ltd. | Polyamic Acid Resin and Polyamideimide Film |
| CN114415472A (zh) * | 2021-12-24 | 2022-04-29 | 上海玟昕科技有限公司 | 含有聚酰亚胺前驱体的感光性树脂组合物 |
| CN114442431A (zh) * | 2021-12-24 | 2022-05-06 | 上海玟昕科技有限公司 | 含有聚酰亚胺前驱体的感光性树脂组合物 |
| CN117229506A (zh) * | 2023-11-16 | 2023-12-15 | 山东华夏神舟新材料有限公司 | 一种聚酰胺酸、聚酰亚胺、聚酰亚胺薄膜及制备方法 |
| JP2023552372A (ja) * | 2020-12-04 | 2023-12-15 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | カルド/スピロ部分を含有するビスマレイミドに基づく誘電材料 |
| EP4438657A1 (en) | 2023-03-27 | 2024-10-02 | Tamura Corporation | Polyamic acid, polyamic acid composition, polyimide, polyimide film, and printed circuit board |
| EP4438656A1 (en) | 2023-03-27 | 2024-10-02 | Tamura Corporation | Polyamic acid, polyamic acid composition, polyimide, polyimide film, and printed circuit board |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11116675A (ja) * | 1997-10-15 | 1999-04-27 | Jsr Corp | 芳香族ビス(エーテル酸無水物)、ポリアミック酸およびポリイミド |
| JPH11292968A (ja) * | 1998-04-15 | 1999-10-26 | Jsr Corp | 電子部品およびその製造方法 |
| JP2000053767A (ja) * | 1997-07-11 | 2000-02-22 | Jsr Corp | 可溶性ポリイミドおよびその製造方法 |
| JP2000063518A (ja) * | 1998-08-18 | 2000-02-29 | Mitsui Chemicals Inc | 架橋基を含有する低誘電性ポリイミド及びその製造方法 |
| JP2005262529A (ja) * | 2004-03-17 | 2005-09-29 | Fuji Photo Film Co Ltd | ガスバリアフィルムおよび該フィルムを用いた有機エレクトロルミネッセンス素子 |
-
2004
- 2004-04-09 JP JP2004115197A patent/JP4485240B2/ja not_active Expired - Lifetime
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000053767A (ja) * | 1997-07-11 | 2000-02-22 | Jsr Corp | 可溶性ポリイミドおよびその製造方法 |
| JPH11116675A (ja) * | 1997-10-15 | 1999-04-27 | Jsr Corp | 芳香族ビス(エーテル酸無水物)、ポリアミック酸およびポリイミド |
| JPH11292968A (ja) * | 1998-04-15 | 1999-10-26 | Jsr Corp | 電子部品およびその製造方法 |
| JP2000063518A (ja) * | 1998-08-18 | 2000-02-29 | Mitsui Chemicals Inc | 架橋基を含有する低誘電性ポリイミド及びその製造方法 |
| JP2005262529A (ja) * | 2004-03-17 | 2005-09-29 | Fuji Photo Film Co Ltd | ガスバリアフィルムおよび該フィルムを用いた有機エレクトロルミネッセンス素子 |
Cited By (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007314583A (ja) * | 2006-05-23 | 2007-12-06 | Nissan Chem Ind Ltd | 脂環式ポリイミド前駆体、ポジ型感光性樹脂組成物およびその硬化膜 |
| JP2015155385A (ja) * | 2014-02-20 | 2015-08-27 | 田岡化学工業株式会社 | フルオレン骨格を有するテトラカルボン酸二無水物、ポリアミック酸、及びポリイミド。 |
| CN109207170A (zh) * | 2017-06-30 | 2019-01-15 | 奇美实业股份有限公司 | 液晶配向剂、液晶配向膜及液晶显示元件 |
| CN109207170B (zh) * | 2017-06-30 | 2021-12-10 | 奇美实业股份有限公司 | 液晶配向剂、液晶配向膜及液晶显示元件 |
| US20210040267A1 (en) * | 2017-09-12 | 2021-02-11 | Sk Innovation Co., Ltd. | Polyamic Acid Resin and Polyamideimide Film |
| US11970579B2 (en) * | 2017-09-12 | 2024-04-30 | Sk Innovation Co., Ltd. | Polyamic acid resin and polyamideimide film |
| JP7361499B2 (ja) | 2018-06-20 | 2023-10-16 | 信越化学工業株式会社 | 化合物及び有機膜形成用組成物 |
| JP2019218337A (ja) * | 2018-06-20 | 2019-12-26 | 信越化学工業株式会社 | 化合物及び有機膜形成用組成物 |
| JP7622155B2 (ja) | 2018-06-20 | 2025-01-27 | 信越化学工業株式会社 | 化合物 |
| JP2023162191A (ja) * | 2018-06-20 | 2023-11-08 | 信越化学工業株式会社 | 化合物 |
| JP2020132532A (ja) * | 2019-02-14 | 2020-08-31 | 田岡化学工業株式会社 | テトラカルボン酸二無水物、ポリアミック酸及びポリイミド |
| JP7128587B2 (ja) | 2019-02-14 | 2022-08-31 | 田岡化学工業株式会社 | テトラカルボン酸二無水物、ポリアミック酸及びポリイミド |
| JP2023552372A (ja) * | 2020-12-04 | 2023-12-15 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | カルド/スピロ部分を含有するビスマレイミドに基づく誘電材料 |
| CN114442431B (zh) * | 2021-12-24 | 2023-08-04 | 上海玟昕科技有限公司 | 含有聚酰亚胺前驱体的感光性树脂组合物 |
| CN114415472B (zh) * | 2021-12-24 | 2023-08-04 | 上海玟昕科技有限公司 | 含有聚酰亚胺前驱体的感光性树脂组合物 |
| CN114442431A (zh) * | 2021-12-24 | 2022-05-06 | 上海玟昕科技有限公司 | 含有聚酰亚胺前驱体的感光性树脂组合物 |
| CN114415472A (zh) * | 2021-12-24 | 2022-04-29 | 上海玟昕科技有限公司 | 含有聚酰亚胺前驱体的感光性树脂组合物 |
| EP4438657A1 (en) | 2023-03-27 | 2024-10-02 | Tamura Corporation | Polyamic acid, polyamic acid composition, polyimide, polyimide film, and printed circuit board |
| EP4438656A1 (en) | 2023-03-27 | 2024-10-02 | Tamura Corporation | Polyamic acid, polyamic acid composition, polyimide, polyimide film, and printed circuit board |
| CN117229506A (zh) * | 2023-11-16 | 2023-12-15 | 山东华夏神舟新材料有限公司 | 一种聚酰胺酸、聚酰亚胺、聚酰亚胺薄膜及制备方法 |
| CN117229506B (zh) * | 2023-11-16 | 2024-03-15 | 山东华夏神舟新材料有限公司 | 一种聚酰胺酸、聚酰亚胺、聚酰亚胺薄膜及制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP4485240B2 (ja) | 2010-06-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7450488B2 (ja) | ポリアミック酸樹脂、ポリイミド樹脂およびこれらを含む樹脂組成物 | |
| CN101421356B (zh) | 热固性聚酰亚胺树脂组合物及其固化物 | |
| JP4498382B2 (ja) | アミン酸エステルオリゴマー、それを含有するポリイミド樹脂のための前駆体組成物、及び使用 | |
| JP4998040B2 (ja) | ポリアミック酸のイミド化重合体絶縁膜および膜形成組成物とその製造方法 | |
| JP4485240B2 (ja) | ポリアミド酸およびポリイミド | |
| CN113396173B (zh) | 环氧树脂溶液 | |
| JP5804778B2 (ja) | 新規ポリイミドワニス | |
| JP5010357B2 (ja) | 新規ポリアミド酸、ポリイミド並びにその用途 | |
| WO2016093310A1 (ja) | ポリイミド及びそれを含む印刷用組成物 | |
| TWI418577B (zh) | A method for producing a polyimide film, and a polyamic acid solution composition | |
| US6040418A (en) | Fluorinated polyimides, laminated substrates and polyamic acid solutions | |
| JP4691555B2 (ja) | 反応性モノマー、及びそれを含む樹脂組成物 | |
| JP5270865B2 (ja) | 接着剤並びにその用途 | |
| JPH11292968A (ja) | 電子部品およびその製造方法 | |
| JPH0292535A (ja) | 金属とポリイミドの複合成形体 | |
| JP2023130732A (ja) | ビスマレイミド化合物及びその製造方法 | |
| KR20150097439A (ko) | 폴리이미드 수지의 제조 방법, 폴리이미드막의 제조 방법, 폴리아믹산 용액의 제조 방법, 폴리이미드막 및 폴리아믹산 용액 | |
| TW201815891A (zh) | 含金剛烷之聚醯亞胺之製備方法 | |
| JP2022101201A (ja) | ポリアミド酸組成物、ポリイミド組成物、金属張積層板及び回路基板 | |
| JP6103992B2 (ja) | ポリイミド | |
| JP4401893B2 (ja) | ポリイミド前駆体樹脂組成物 | |
| CN115141370A (zh) | 聚酰亚胺、金属包覆层叠板及电路基板 | |
| JP2002060488A (ja) | ポリアミド酸及びポリイミドの製造方法、並びにこれらを用いて得られた接着テープ | |
| JP3649543B2 (ja) | ポリイミドシロキサン樹脂組成物 | |
| JP7567071B2 (ja) | ポリイミド、ポリイミド溶液、コーティング材料および成形材料 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A711 | Notification of change in applicant |
Free format text: JAPANESE INTERMEDIATE CODE: A712 Effective date: 20061212 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20070214 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20090929 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20091006 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20091104 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20100323 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20100324 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4485240 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20130402 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20130402 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20140402 Year of fee payment: 4 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |