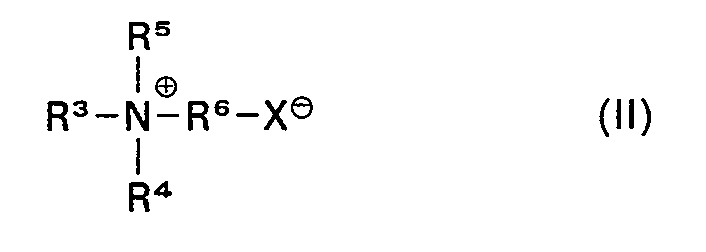EP0019734B1 - Agent de lavage pour textiles - Google Patents
Agent de lavage pour textiles Download PDFInfo
- Publication number
- EP0019734B1 EP0019734B1 EP80102334A EP80102334A EP0019734B1 EP 0019734 B1 EP0019734 B1 EP 0019734B1 EP 80102334 A EP80102334 A EP 80102334A EP 80102334 A EP80102334 A EP 80102334A EP 0019734 B1 EP0019734 B1 EP 0019734B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- weight
- radical
- detergent
- alkyl
- light
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired
Links
- 239000004753 textile Substances 0.000 title claims abstract description 8
- 238000005406 washing Methods 0.000 title description 22
- 239000003599 detergent Substances 0.000 claims abstract description 32
- 150000004665 fatty acids Chemical class 0.000 claims abstract description 26
- 235000014113 dietary fatty acids Nutrition 0.000 claims abstract description 23
- 239000000194 fatty acid Substances 0.000 claims abstract description 23
- 229930195729 fatty acid Natural products 0.000 claims abstract description 23
- 239000000203 mixture Substances 0.000 claims abstract description 19
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims abstract description 17
- 125000004432 carbon atom Chemical group C* 0.000 claims abstract description 17
- 229920000151 polyglycol Polymers 0.000 claims abstract description 16
- 239000010695 polyglycol Substances 0.000 claims abstract description 16
- 239000004094 surface-active agent Substances 0.000 claims abstract description 13
- 150000002170 ethers Chemical class 0.000 claims abstract description 10
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical class N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 claims abstract description 9
- 239000004615 ingredient Substances 0.000 claims abstract description 7
- 150000003242 quaternary ammonium salts Chemical class 0.000 claims abstract description 7
- 239000003945 anionic surfactant Substances 0.000 claims abstract description 6
- 239000000126 substance Substances 0.000 claims abstract description 5
- 125000001931 aliphatic group Chemical group 0.000 claims abstract description 4
- 125000002091 cationic group Chemical group 0.000 claims abstract description 4
- 239000001257 hydrogen Substances 0.000 claims abstract description 4
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract description 4
- MTNDZQHUAFNZQY-UHFFFAOYSA-N imidazoline Chemical compound C1CN=CN1 MTNDZQHUAFNZQY-UHFFFAOYSA-N 0.000 claims abstract description 4
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 4
- 125000004433 nitrogen atom Chemical group N* 0.000 claims abstract description 4
- 239000004721 Polyphenylene oxide Substances 0.000 claims abstract description 3
- 125000003342 alkenyl group Chemical group 0.000 claims abstract description 3
- 125000002947 alkylene group Chemical group 0.000 claims abstract description 3
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims abstract description 3
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 claims abstract description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 3
- 229920000570 polyether Polymers 0.000 claims abstract description 3
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims abstract 2
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 claims description 23
- 150000002191 fatty alcohols Chemical class 0.000 claims description 16
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 claims description 9
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 claims description 9
- 150000001450 anions Chemical class 0.000 claims description 5
- JZMJDSHXVKJFKW-UHFFFAOYSA-M methyl sulfate(1-) Chemical compound COS([O-])(=O)=O JZMJDSHXVKJFKW-UHFFFAOYSA-M 0.000 claims description 5
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 claims description 4
- 229960000541 cetyl alcohol Drugs 0.000 claims description 4
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 claims description 4
- 229940055577 oleyl alcohol Drugs 0.000 claims description 4
- XMLQWXUVTXCDDL-UHFFFAOYSA-N oleyl alcohol Natural products CCCCCCC=CCCCCCCCCCCO XMLQWXUVTXCDDL-UHFFFAOYSA-N 0.000 claims description 4
- 125000001117 oleyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])/C([H])=C([H])\C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 4
- LBLYYCQCTBFVLH-UHFFFAOYSA-M 2-methylbenzenesulfonate Chemical compound CC1=CC=CC=C1S([O-])(=O)=O LBLYYCQCTBFVLH-UHFFFAOYSA-M 0.000 claims description 3
- KIWBPDUYBMNFTB-UHFFFAOYSA-N Ethyl hydrogen sulfate Chemical compound CCOS(O)(=O)=O KIWBPDUYBMNFTB-UHFFFAOYSA-N 0.000 claims description 3
- OTMSDBZUPAUEDD-UHFFFAOYSA-N Ethane Chemical compound CC OTMSDBZUPAUEDD-UHFFFAOYSA-N 0.000 claims description 2
- 229910021529 ammonia Inorganic materials 0.000 claims description 2
- 235000019864 coconut oil Nutrition 0.000 claims 2
- 239000003240 coconut oil Substances 0.000 claims 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 abstract description 4
- 239000000047 product Substances 0.000 description 15
- 239000003795 chemical substances by application Substances 0.000 description 14
- -1 imidazolinium compound Chemical class 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 12
- 239000004435 Oxo alcohol Substances 0.000 description 11
- 235000013162 Cocos nucifera Nutrition 0.000 description 10
- 244000060011 Cocos nucifera Species 0.000 description 10
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 7
- 150000003856 quaternary ammonium compounds Chemical class 0.000 description 7
- REZZEXDLIUJMMS-UHFFFAOYSA-M dimethyldioctadecylammonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCCCC[N+](C)(C)CCCCCCCCCCCCCCCCCC REZZEXDLIUJMMS-UHFFFAOYSA-M 0.000 description 6
- 239000002736 nonionic surfactant Substances 0.000 description 6
- 239000003760 tallow Substances 0.000 description 6
- JSPLKZUTYZBBKA-UHFFFAOYSA-N trioxidane Chemical compound OOO JSPLKZUTYZBBKA-UHFFFAOYSA-N 0.000 description 6
- 230000000694 effects Effects 0.000 description 5
- 239000004744 fabric Substances 0.000 description 5
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 4
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 239000004480 active ingredient Substances 0.000 description 4
- 150000001298 alcohols Chemical class 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 238000005187 foaming Methods 0.000 description 4
- 210000002268 wool Anatomy 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- 239000000654 additive Substances 0.000 description 3
- 150000001412 amines Chemical class 0.000 description 3
- 239000000470 constituent Substances 0.000 description 3
- 239000004664 distearyldimethylammonium chloride (DHTDMAC) Substances 0.000 description 3
- 239000006260 foam Substances 0.000 description 3
- 238000000034 method Methods 0.000 description 3
- 229920002239 polyacrylonitrile Polymers 0.000 description 3
- 229920002647 polyamide Polymers 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- NTHWMYGWWRZVTN-UHFFFAOYSA-N sodium silicate Chemical compound [Na+].[Na+].[O-][Si]([O-])=O NTHWMYGWWRZVTN-UHFFFAOYSA-N 0.000 description 3
- 229910052938 sodium sulfate Inorganic materials 0.000 description 3
- 235000011152 sodium sulphate Nutrition 0.000 description 3
- 235000019832 sodium triphosphate Nutrition 0.000 description 3
- 238000001694 spray drying Methods 0.000 description 3
- 150000003512 tertiary amines Chemical class 0.000 description 3
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 2
- KXDHJXZQYSOELW-UHFFFAOYSA-N Carbamic acid Chemical class NC(O)=O KXDHJXZQYSOELW-UHFFFAOYSA-N 0.000 description 2
- IGFHQQFPSIBGKE-UHFFFAOYSA-N Nonylphenol Natural products CCCCCCCCCC1=CC=C(O)C=C1 IGFHQQFPSIBGKE-UHFFFAOYSA-N 0.000 description 2
- 239000004952 Polyamide Substances 0.000 description 2
- 229910004298 SiO 2 Inorganic materials 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- 239000004115 Sodium Silicate Substances 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 239000003513 alkali Substances 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- 230000029936 alkylation Effects 0.000 description 2
- 238000005804 alkylation reaction Methods 0.000 description 2
- 150000001735 carboxylic acids Chemical class 0.000 description 2
- 239000013065 commercial product Substances 0.000 description 2
- 230000000536 complexating effect Effects 0.000 description 2
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 239000002979 fabric softener Substances 0.000 description 2
- 239000003205 fragrance Substances 0.000 description 2
- 229910052740 iodine Inorganic materials 0.000 description 2
- 239000011630 iodine Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- SNQQPOLDUKLAAF-UHFFFAOYSA-N nonylphenol Chemical compound CCCCCCCCCC1=CC=CC=C1O SNQQPOLDUKLAAF-UHFFFAOYSA-N 0.000 description 2
- 150000002894 organic compounds Chemical class 0.000 description 2
- 239000004014 plasticizer Substances 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 150000003335 secondary amines Chemical class 0.000 description 2
- 229910052911 sodium silicate Inorganic materials 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 229920002994 synthetic fiber Polymers 0.000 description 2
- 239000012209 synthetic fiber Substances 0.000 description 2
- ALSTYHKOOCGGFT-KTKRTIGZSA-N (9Z)-octadecen-1-ol Chemical compound CCCCCCCC\C=C/CCCCCCCCO ALSTYHKOOCGGFT-KTKRTIGZSA-N 0.000 description 1
- PSBDWGZCVUAZQS-UHFFFAOYSA-N (dimethylsulfonio)acetate Chemical compound C[S+](C)CC([O-])=O PSBDWGZCVUAZQS-UHFFFAOYSA-N 0.000 description 1
- FSSPGSAQUIYDCN-UHFFFAOYSA-N 1,3-Propane sultone Chemical compound O=S1(=O)CCCO1 FSSPGSAQUIYDCN-UHFFFAOYSA-N 0.000 description 1
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N 2-Propenoic acid Natural products OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical class OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- UGFAIRIUMAVXCW-UHFFFAOYSA-N Carbon monoxide Chemical compound [O+]#[C-] UGFAIRIUMAVXCW-UHFFFAOYSA-N 0.000 description 1
- 229920000742 Cotton Polymers 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical class S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 229930186657 Lat Natural products 0.000 description 1
- 229920000388 Polyphosphate Polymers 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 150000001335 aliphatic alkanes Chemical group 0.000 description 1
- 150000001447 alkali salts Chemical class 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 150000001642 boronic acid derivatives Chemical class 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 229910002091 carbon monoxide Inorganic materials 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical class OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- FOCAUTSVDIKZOP-UHFFFAOYSA-N chloroacetic acid Chemical compound OC(=O)CCl FOCAUTSVDIKZOP-UHFFFAOYSA-N 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 229910052681 coesite Inorganic materials 0.000 description 1
- 229910052906 cristobalite Inorganic materials 0.000 description 1
- XPPKVPWEQAFLFU-UHFFFAOYSA-J diphosphate(4-) Chemical compound [O-]P([O-])(=O)OP([O-])([O-])=O XPPKVPWEQAFLFU-UHFFFAOYSA-J 0.000 description 1
- 235000011180 diphosphates Nutrition 0.000 description 1
- LQZZUXJYWNFBMV-UHFFFAOYSA-N dodecan-1-ol Chemical compound CCCCCCCCCCCCO LQZZUXJYWNFBMV-UHFFFAOYSA-N 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- MEKDPHXPVMKCON-UHFFFAOYSA-N ethane;methane Chemical compound C.CC MEKDPHXPVMKCON-UHFFFAOYSA-N 0.000 description 1
- WBJINCZRORDGAQ-UHFFFAOYSA-N ethyl formate Chemical compound CCOC=O WBJINCZRORDGAQ-UHFFFAOYSA-N 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 230000002070 germicidal effect Effects 0.000 description 1
- 150000003977 halocarboxylic acids Chemical class 0.000 description 1
- IPCSVZSSVZVIGE-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- 238000007037 hydroformylation reaction Methods 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- 150000002462 imidazolines Chemical class 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 125000005341 metaphosphate group Chemical group 0.000 description 1
- JZMJDSHXVKJFKW-UHFFFAOYSA-N methyl sulfate Chemical compound COS(O)(=O)=O JZMJDSHXVKJFKW-UHFFFAOYSA-N 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 235000011837 pasties Nutrition 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 150000003009 phosphonic acids Chemical class 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 239000001205 polyphosphate Substances 0.000 description 1
- 235000011176 polyphosphates Nutrition 0.000 description 1
- 235000019353 potassium silicate Nutrition 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- KCXFHTAICRTXLI-UHFFFAOYSA-N propane-1-sulfonic acid Chemical compound CCCS(O)(=O)=O KCXFHTAICRTXLI-UHFFFAOYSA-N 0.000 description 1
- ROSDSFDQCJNGOL-UHFFFAOYSA-N protonated dimethyl amine Natural products CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 1
- 125000001453 quaternary ammonium group Chemical group 0.000 description 1
- 238000005956 quaternization reaction Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 229910052682 stishovite Inorganic materials 0.000 description 1
- 229940117986 sulfobetaine Drugs 0.000 description 1
- DIORMHZUUKOISG-UHFFFAOYSA-N sulfoformic acid Chemical class OC(=O)S(O)(=O)=O DIORMHZUUKOISG-UHFFFAOYSA-N 0.000 description 1
- 150000003460 sulfonic acids Chemical class 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 150000008053 sultones Chemical class 0.000 description 1
- 229910052905 tridymite Inorganic materials 0.000 description 1
- 235000011178 triphosphate Nutrition 0.000 description 1
- 239000001226 triphosphate Substances 0.000 description 1
- UNXRWKVEANCORM-UHFFFAOYSA-N triphosphoric acid Chemical compound OP(O)(=O)OP(O)(=O)OP(O)(O)=O UNXRWKVEANCORM-UHFFFAOYSA-N 0.000 description 1
- 239000010457 zeolite Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/66—Non-ionic compounds
- C11D1/72—Ethers of polyoxyalkylene glycols
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/38—Cationic compounds
- C11D1/52—Carboxylic amides, alkylolamides or imides or their condensation products with alkylene oxides
- C11D1/523—Carboxylic alkylolamides, or dialkylolamides, or hydroxycarboxylic amides (R1-CO-NR2R3), where R1, R2 or R3 contain one hydroxy group per alkyl group
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/38—Cationic compounds
- C11D1/62—Quaternary ammonium compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/88—Ampholytes; Electroneutral compounds
- C11D1/94—Mixtures with anionic, cationic or non-ionic compounds
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/0005—Other compounding ingredients characterised by their effect
- C11D3/001—Softening compositions
Definitions
- the invention relates to a powdery or granular detergent with a fabric softening effect for the simultaneous washing and care of delicates in the washing machine or by hand.
- the washing effect of such combinations is particularly good if the ratio of the nonionic surfactant to the quaternary ammonium compound in a ratio of 4: 1 to 1: 1 lies.
- Powdered detergents for machine washing based on nonionic adducts of ethylene oxide with certain alkylphenols, fatty alcohols, mercaptans, fatty acid monoethanolamides or fatty amines as nonionic surfactant components, which contain cationic ammonium compounds to reduce foam, are described in DE-AS No. 1220956.
- DE-OS No. 2828619 discloses a detergent with nonionic polyoxyalkylene surfactants, a small proportion of zwitterionic or semipolar surfactants, small amounts of a cationic surface-active substance and 50 to 95% by weight of other customary detergent additives, which can also be understood as alkanolamides. According to the teaching of this patent application, the small proportions of zwitterionic or semipolar surfactants and cationic surface-active substances bring about a reduction in color transfer while washing colored and white textiles. US Patent No.
- 3351557 describes emulsions which can contain about 1-15% by weight of polyoxyalkylene surfactants, 2-10% by weight of a sulfobetaine or amine oxide and 0.1-0.5% by weight of quaternary ammonium salt as a germicide .
- the invention was therefore based on the object of providing a powdery or granular detergent for mild detergents which has optimum washing power, textile-softening properties and also foaming behavior and which can be used both in the washing machine and for washing by hand.
- Another object of the invention is to provide a detergent formulation which, when produced by spray drying, provides a detergent with a reduced bulk density.
- the polyglycol ethers of the formula I which are suitable according to the invention as constituent a are derived from natural or synthetic primary fatty alcohols or oxo alcohols.
- Oxo alcohols are produced by reacting linear olefins with carbon monoxide and hydrogen according to the known oxo process by hydroformylation and subsequent hydrogenation.
- Commercially available oxo alcohol mixtures which are suitable for the preparation of the surfactant component a are, for example, the oxo alcohols available under the trade name Dobanol® from the German Shell Chemie Deutschen, which have about 25% by weight of 2-alkyl branched alcohols; other suitable oxo alcohols are available under the name Synprol® from Imperial Chemical Industries Ltd.
- oxo alcohols available with about 50-70% by weight of 2-alkyl branched alcohols;
- suitable products based on oxo alcohols are, for example, various types of Lutensol from BASF with approx. 30-35% by weight of branched alcohols and some Lial® types from Liquichimica SpA with approx. 60% by weight of branched alcohols.
- Typical preferred products are, for example, Dobanol 45-7 s , which consists of at least 95% C 14 or C 15 oxo alcohol with an average of 7 mol of ethylene oxide; also Lutensol AO-8, which is a C 13 to. C 15 oxo alcohol ethoxylate with an average of 8 mol of ethylene oxide and Lutensol ON-70®, which is a C 9 to C 11 oxo alcohol ethoxylate with an average of 7 mol of ethylene oxide.
- Typical surfactants derived from fatty alcohols are based, for example, on tallow fatty alcohol or the commercial products Lorol® with 8-18 carbon atoms from Henkel.
- Typical preferred products are, for example, Dehydol TA 5 and Dehydol TA 14® (Henkel), which are tallow fatty alcohol polyglycol ethers with 5 or 14 mol ethylene oxide / mol alcohol.
- Another preferred product is, for example, the commercial product Marlipal KF® (Chemische Werke Huls), a C 10 to C 12 fatty alcohol ethoxylate with an average of 6 mol of ethylene oxide.
- polyglycol ethers are the addition products derived from oleyl alcohol or cetyl alcohol with 5-10 mol of ethylene oxide or technical mixtures of predominantly oleyl and cetyl alcohol with 5-10 mol of ethylene oxide, the iodine number of which is between approximately 40 and approximately 60. Also preferred is the commercial product Dehydol LS 4® (Henkel), a C 12 to C 14 fatty alcohol ethoxylate with approx. 4 mol ethylene oxide. These polyglycol ethers, individually or in a mixture, form component a of the agents according to the invention.
- the finished agent are contained in an amount of 5-20% by weight, preferably 7-15% by weight, and can be wholly or partly by the C s to C 15 alkylaryl polyglycol ethers known as nonionic surfactants, such as nonylphenol with approx 15 mol of ethylene oxide are replaced.
- nonionic surfactants such as nonylphenol with approx 15 mol of ethylene oxide
- Fatty acid mono- or diethanolamides with a fatty acid residue with 12-18 carbon atoms are suitable as component b of the agents according to the invention. Both the compounds with a uniform number of carbon atoms in the fatty acid residue and mixtures of fatty acid ethanolamides with differently long fatty acid residues can be used.
- a preferred product is derived from coconut fatty acid with a proportion of more than 50% by weight of C 12 fatty acid, approx. 20% by weight C 14 fatty acid, approx. 10% by weight C 16 fatty acid and a further proportion of saturated and unsaturated C 18 fatty acid.
- Such a product is commercially available under the name Comperlan 100 ® (Henkel).
- Another suitable product is the commercially available coconut fatty acid diethanolamide as Comperlan KD ® (Henkel).
- the fatty acid ethanolamides are contained in the agents according to the invention to 1-10% by weight, preferably to 2-5% by weight.
- component c of the agents according to the invention are N-alkyl-N, N-dimethylammonium acetate, N-alkyl-N-methyl-N-benzylammonium acetate, N-alkyl-N-bis (2-hydroxyethyl) ammonium acetate, N-alkyl- N-diethylammonium propionate, 2-alkyl-1- (2-hydroxyethyl) -.
- Imidazolinium-1-acetate 3- (N-alkyl-N-dimethyl-ammonium) -2-hydroxypropyl sulfate, 3- (N-alkyl-N- dimethylammonium) propanesulfonate and 1- (3-trimethylammonium) alkanesulfonate, in which the alkyl or alkane groups are preferably straight-chain hydrocarbon residues with 10-22, preferably 12 to 18 carbon atoms and are derived, for example, from natural fat residues, such as coconut or tallow fat can.
- tertiary amines of the formula R 3 R 4 R 5 N are obtainable by reacting tertiary amines of the formula R 3 R 4 R 5 N with halocarboxylic acids, for example chloro- or bromoacetic acid or a-chloropropionic acid, or by quaternizing aminocarboxylic acids or reaction products of secondary amines with acrylic acid in a suitable manner.
- Sulfate betaines can be obtained in an analogous manner by reacting the tertiary amines with haloalkylsulfuric acid esters or their salts.
- Sulfobetaines are obtained by reacting the tertiary amines with haloalkylsulfonic acids or sultones, in particular with propane sultone.
- the betaines are contained in the agents according to the invention in an amount of 1-10% by weight, preferably 2-6% by weight.
- the quaternary ammonium compounds with preferably two long-chain, preferably saturated aliphatic radicals are suitable as textile-softening active ingredients (component d) each with 16-20 carbon atoms and at least one quaternary nitrogen atom in the molecule.
- the long-chain aliphatic radicals can be straight-chain or branched and can accordingly be derived from fatty acids or from fatty amines, Guerbetamines, or from the alkylamines obtainable by reduction of nitroparaffins.
- quaternary ammonium compounds are, in particular, derivatives of ammonia, ie the quaternary salts obtainable by alkylation of long-chain secondary amines, such as, for example, the compounds distearyldimethylammonium chloride or ditalgalkyldimethylammonium chloride, or the reaction of 1 mol of an aminoalkylethylenediamine or hydroxyalkylethylenediamine with 2 mol a long-chain, C 14 to C 26 fatty acid or its ester obtainable imidazoline compounds which are subsequently converted into the quaternary imidazolinium compounds by alkylation.
- long-chain secondary amines such as, for example, the compounds distearyldimethylammonium chloride or ditalgalkyldimethylammonium chloride, or the reaction of 1 mol of an aminoalkylethylenediamine or hydroxyalkylethylenediamine with 2 mol a long-chain, C 14 to C 26 fatty acid or its ester
- Typical preferred textile plasticizers are, for example, ditallow alkyldimethylammonium chloride (for example the commercial products refpagen WK K and refpagen WKT®, Hoechst, Adogen 442®, Ashland) or distearyldimethylammonium chloride (for example Arosurf TA 100 9 , Ashland) and 2-heptadecyl-1-methyl-1-oleoylamidium methyl sulfate.
- ditallow alkyldimethylammonium chloride for example the commercial products recuperpagen WK K and Reechst, Adogen 442®, Ashland
- distearyldimethylammonium chloride for example Arosurf TA 100 9 , Ashland
- 2-heptadecyl-1-methyl-1-oleoylamidium methyl sulfate for example, 2-heptadecyl-1-methyl-1-oleoylamidium methyl sulfate.
- the textile-softening active ingredients are contained in the agents according to the invention to 2-10% by weight, preferably 3-7% by weight, the textile-softening active ingredient d preferably being more than 25 - in particular more than 30% by weight - based on the total weight of the product Polyglycol ether a, the fatty acid ethanol amide b and the fabric softening active ingredient d is included.
- the agents according to the invention contain 50-91% by weight of additives usually present in detergents as further constituents. These are, for example, builders, fragrances, dyes and dirt carriers, enzymes, optical brighteners or small amounts of water.
- the builders include calcium-precipitating or complexing inorganic or organic compounds.
- Inorganic precipitating or complexing substances are, for example, pyrophosphate, triphosphate, higher polyphosphates and metaphosphates and the cation-exchanging alkali alumosilicates, as described, for example, in DE-AS No. 2412837, in particular the zeolites A and X.
- Suitable organic compounds are, for example, polycarboxylic acids, hydroxycarboxylic acids , Amino carboxylic acids, carboxyalkyl ether, polyanionic polymeric carboxylic acids, phosphonic acids.
- Other builders are weakly acidic, neutral or alkaline inorganic salts, such as bicarbonates, carbonates, borates or silicates of alkalis, alkali sulfates, and the alkali salts of organic, non-capillary, 1 to 8 carbon atoms containing sulfonic acids, carboxylic acids and sulfocarboxylic acids.
- the agents according to the invention preferably contain about 10-50% by weight sodium tripolyphosphate, 2-10% by weight water glass with a Na 2 0 / Si0 2 ratio of, for example, 1: 3.4 and sodium sulfate as builders.
- Anionic surfactants are not contained in the agents according to the invention.
- the agents according to the invention are in powder form. These are fine-grained to granular or spherical products that are easy to pour. They can be produced by all methods known in the art. So you can e.g. mix the powdered ingredients together and oily or pasty ingredients such as e.g. spray non-ionic surfactants on the product. Another possibility for production consists in pasting the constituents of the agents with water to a paste which can be obtained by evaporating the water, e.g. on rollers or in atomizing towers, converted into a powder. After hot drying, heat-sensitive or volatile substances such as fragrances can be incorporated. Products according to the invention produced by spray drying have a bulk density of approximately 150 g / l to approximately 300 g / l.
- the detergents according to the invention can be used for washing and simultaneously softening fine laundry articles made of wool, synthetic fibers such as polyester, polyacrylonitrile, polyamide and mixtures of wool and synthetic fibers.
- the washing and softening process can take place either in the washing machine or by hand at washing temperatures up to about 60 ° C, preferably up to about 30 ° C.
- the application concentration of the agents is generally 1 to 10 g / l of wash liquor, preferably 2 to 5 g / l.
- the detergents are characterized by the fact that they clean the textiles perfectly and at the same time give them a pleasant handle and anti-electrostatic properties. When washing by hand in the washbasin, they form a dense, pleasant foam, and no foaming is observed when washing in the washing machine. The foam can also be easily rinsed out.
- An agent of the above composition is a detergent according to the invention containing conventional additives in conventional amounts. It was produced by spray drying and has a bulk density of approximately 250 g / l.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Detergent Compositions (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AT80102334T ATE2226T1 (de) | 1979-05-07 | 1980-04-30 | Waschmittel fuer textilien. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19792918363 DE2918363A1 (de) | 1979-05-07 | 1979-05-07 | Waschmittel fuer textilien |
| DE2918363 | 1979-05-07 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0019734A1 EP0019734A1 (fr) | 1980-12-10 |
| EP0019734B1 true EP0019734B1 (fr) | 1983-01-12 |
Family
ID=6070129
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP80102334A Expired EP0019734B1 (fr) | 1979-05-07 | 1980-04-30 | Agent de lavage pour textiles |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP0019734B1 (fr) |
| AT (1) | ATE2226T1 (fr) |
| BR (1) | BR8002781A (fr) |
| DE (2) | DE2918363A1 (fr) |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3026090A1 (de) * | 1980-07-10 | 1982-02-18 | Henkel KGaA, 4000 Düsseldorf | Fluessiges waschmittel mit einem gehalt an verfaerbungsinhibierenden zusaetzen |
| ZA856296B (en) * | 1984-08-31 | 1987-03-25 | Colgate Palmolive Co | Hot water wash cycle detergent-softener compositions |
| GB2163771B (en) * | 1984-08-31 | 1989-01-18 | Colgate Palmolive Co | Wash cycle detergent-softener compositions |
| GB8609884D0 (en) * | 1986-04-23 | 1986-05-29 | Procter & Gamble | Softening detergent compositions |
| EP0258923B1 (fr) * | 1986-09-02 | 1993-10-06 | Akzo Nobel N.V. | Composition adoucissante pour le linge et détergent la contenant |
| DE3726911A1 (de) * | 1987-08-13 | 1989-02-23 | Henkel Kgaa | Waessrige zubereitungen ionischer tenside mit erhoehter viskositaet |
| DE3928601A1 (de) * | 1989-08-30 | 1991-03-07 | Henkel Kgaa | Verwendung ausgewaehlter mischungen von polyalkylendiolethern als schaumdrueckende zusaetze fuer schaumarme reinigungsmittel |
| GB9410678D0 (en) * | 1994-05-27 | 1994-07-13 | Unilever Plc | Surfactant composition and cleaning composition comprising the same |
| DE19624637A1 (de) * | 1996-06-21 | 1998-01-02 | Henkel Kgaa | Waschverfahren für Textilien mit verbesserten Pflegeeigenschaften |
| DE19939806A1 (de) * | 1999-08-21 | 2001-02-22 | Cognis Deutschland Gmbh | Schaumkontrollierte feste Waschmittel |
| EP1397475A2 (fr) * | 2001-05-31 | 2004-03-17 | Huntsman International Llc | Poudres compactables |
| WO2014154234A1 (fr) * | 2013-03-25 | 2014-10-02 | Rhodia Operations | Produit assouplissant |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5914077B2 (ja) * | 1976-10-01 | 1984-04-03 | 花王株式会社 | 液体軽質洗剤組成物 |
| IT1097290B (it) * | 1977-06-29 | 1985-08-31 | Procter & Gamble | Composizioni detergenti per lavanderia |
| CA1115618A (fr) * | 1977-07-01 | 1982-01-05 | Unilever Limited | Detergents |
-
1979
- 1979-05-07 DE DE19792918363 patent/DE2918363A1/de not_active Withdrawn
-
1980
- 1980-04-30 DE DE8080102334T patent/DE3061591D1/de not_active Expired
- 1980-04-30 EP EP80102334A patent/EP0019734B1/fr not_active Expired
- 1980-04-30 AT AT80102334T patent/ATE2226T1/de not_active IP Right Cessation
- 1980-05-06 BR BR8002781A patent/BR8002781A/pt unknown
Also Published As
| Publication number | Publication date |
|---|---|
| DE2918363A1 (de) | 1980-11-27 |
| DE3061591D1 (en) | 1983-02-17 |
| BR8002781A (pt) | 1980-12-16 |
| EP0019734A1 (fr) | 1980-12-10 |
| ATE2226T1 (de) | 1983-01-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE2646995C2 (fr) | ||
| EP0019734B1 (fr) | Agent de lavage pour textiles | |
| DE2817834C2 (de) | Flüssiges Waschmittel | |
| EP1064349A2 (fr) | Detergent liquide polyphase | |
| DE2407708A1 (de) | Textilweichmachungsmittel und dessen verwendung | |
| EP0044003B1 (fr) | Agent de lavage liquide avec une teneur en additifs inhibant le changement de couleur | |
| CH672138A5 (fr) | ||
| DE1220956B (de) | Maschinenwaschmittel | |
| EP0131138B1 (fr) | Utilisation de cyanamides d'acides gras comme agents tensio-actifs pour le nettoyage de surfaces dures | |
| WO1994010279A1 (fr) | Procede de preparation de solutions aqueuses de tensioactifs anioniques ayant une resistance aux basses temperatures amelioree | |
| DE3615544A1 (de) | Verfahren zur vorbehandlung verschmutzter textilien | |
| DE1669103A1 (de) | Scheuermittel | |
| DE19830267A1 (de) | Handgeschirrspülmittel | |
| EP0005850B1 (fr) | Agent de lavage liquide | |
| DE2060849A1 (de) | Waschmittel zum gleichzeitigen Reinigen und Weichmachen | |
| DE3686917T2 (de) | Dispergierbare waescheweichmacher. | |
| DE1617116C3 (de) | Schwachschäumende Wasch- und Reinigungsmittel | |
| DE2825218A1 (de) | Fluessiges wasch- bzw. reinigungsmittel | |
| EP0018630A1 (fr) | Agent de lavage pour textiles | |
| DE2544605A1 (de) | Verfahren zur reinigung textiler flaechenbelaege | |
| DE2025944A1 (en) | Detergent compsns for textiles - with combined cleaning - and softening actions,contng quaternary ammonium salts of unsatd carb | |
| DE2153164C2 (de) | Flüssiges Grobwaschmittel | |
| DE19535260A1 (de) | Wäßrige Tensidzusammensetzung | |
| DE2700640C2 (de) | Für die Kaltwäsche geeignetes Waschmittel | |
| DE1467657A1 (de) | Detergenzusammensetzungen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH DE FR IT NL |
|
| 17P | Request for examination filed |
Effective date: 19810404 |
|
| ITF | It: translation for a ep patent filed | ||
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH DE FR IT LI NL |
|
| REF | Corresponds to: |
Ref document number: 2226 Country of ref document: AT Date of ref document: 19830115 Kind code of ref document: T |
|
| REF | Corresponds to: |
Ref document number: 3061591 Country of ref document: DE Date of ref document: 19830217 |
|
| ET | Fr: translation filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 19890323 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 19890330 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 19890405 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19890412 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 19890428 Year of fee payment: 10 |
|
| ITTA | It: last paid annual fee | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19890430 Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Effective date: 19900430 Ref country code: CH Effective date: 19900430 Ref country code: BE Effective date: 19900430 Ref country code: AT Effective date: 19900430 |
|
| BERE | Be: lapsed |
Owner name: HENKEL G.M.B.H. Effective date: 19900430 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19901101 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Effective date: 19901228 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Effective date: 19910101 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |