CN102753199A - 癌干细胞标记物及其用途 - Google Patents
癌干细胞标记物及其用途 Download PDFInfo
- Publication number
- CN102753199A CN102753199A CN2011800065922A CN201180006592A CN102753199A CN 102753199 A CN102753199 A CN 102753199A CN 2011800065922 A CN2011800065922 A CN 2011800065922A CN 201180006592 A CN201180006592 A CN 201180006592A CN 102753199 A CN102753199 A CN 102753199A
- Authority
- CN
- China
- Prior art keywords
- cancer
- cells
- tumor
- cancer stem
- cell
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/575—Immunoassay; Biospecific binding assay; Materials therefor for cancer
- G01N33/5756—Immunoassay; Biospecific binding assay; Materials therefor for cancer involving tumour-associated glycolinkage [TAG]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/435—Assays involving biological materials from specific organisms or of a specific nature from animals; from humans
- G01N2333/705—Assays involving receptors, cell surface antigens or cell surface determinants
- G01N2333/70596—Molecules with a "CD"-designation not provided for elsewhere in G01N2333/705
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Biomedical Technology (AREA)
- Molecular Biology (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biotechnology (AREA)
- Pathology (AREA)
- Cell Biology (AREA)
- Oncology (AREA)
- Food Science & Technology (AREA)
- Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- General Physics & Mathematics (AREA)
- Microbiology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Peptides Or Proteins (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Apparatus Associated With Microorganisms And Enzymes (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Investigating Or Analysing Biological Materials (AREA)
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US29673810P | 2010-01-20 | 2010-01-20 | |
| EP10000527.1 | 2010-01-20 | ||
| US61/296,738 | 2010-01-20 | ||
| EP10000527A EP2347769A1 (en) | 2010-01-20 | 2010-01-20 | Cancer stem cell markers and uses thereof |
| PCT/EP2011/000217 WO2011089004A1 (en) | 2010-01-20 | 2011-01-20 | Cancer stem cell markers and uses thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN102753199A true CN102753199A (zh) | 2012-10-24 |
Family
ID=42173400
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2011800065922A Pending CN102753199A (zh) | 2010-01-20 | 2011-01-20 | 癌干细胞标记物及其用途 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20120294859A1 (enExample) |
| EP (2) | EP2347769A1 (enExample) |
| JP (2) | JP2013517487A (enExample) |
| CN (1) | CN102753199A (enExample) |
| AU (1) | AU2011208874B2 (enExample) |
| BR (1) | BR112012018132A2 (enExample) |
| CA (1) | CA2786212A1 (enExample) |
| WO (1) | WO2011089004A1 (enExample) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103969437A (zh) * | 2014-04-22 | 2014-08-06 | 广州恒泰生物科技有限公司 | 用于卵巢癌早中期快速诊断试剂盒的制备方法 |
| CN111363008A (zh) * | 2020-03-18 | 2020-07-03 | 北京鼎成肽源生物技术有限公司 | 一种输卵管癌靶标抗原组合、输卵管癌靶标抗原组合刺激培养的ctl细胞及其应用 |
| CN113804531A (zh) * | 2021-09-10 | 2021-12-17 | 北京大学 | 一种细胞高活性染色方法 |
| CN115916349A (zh) * | 2020-06-30 | 2023-04-04 | 葛莱高托普有限公司 | 针对Lewis Y的人源化抗体 |
| CN116718774A (zh) * | 2023-07-07 | 2023-09-08 | 杭州准星医学科技有限公司 | 一种用于肿瘤体外诊断的试剂盒及其使用方法 |
Families Citing this family (66)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7960139B2 (en) | 2007-03-23 | 2011-06-14 | Academia Sinica | Alkynyl sugar analogs for the labeling and visualization of glycoconjugates in cells |
| EP2318832B1 (en) | 2008-07-15 | 2013-10-09 | Academia Sinica | Glycan arrays on ptfe-like aluminum coated glass slides and related methods |
| US10087236B2 (en) | 2009-12-02 | 2018-10-02 | Academia Sinica | Methods for modifying human antibodies by glycan engineering |
| US11377485B2 (en) | 2009-12-02 | 2022-07-05 | Academia Sinica | Methods for modifying human antibodies by glycan engineering |
| US10338069B2 (en) | 2010-04-12 | 2019-07-02 | Academia Sinica | Glycan arrays for high throughput screening of viruses |
| US9778264B2 (en) | 2010-09-03 | 2017-10-03 | Abbvie Stemcentrx Llc | Identification and enrichment of cell subpopulations |
| CN106434561A (zh) | 2010-09-03 | 2017-02-22 | 艾伯维施特姆森特克斯有限责任公司 | 细胞亚群的鉴定和富集 |
| WO2012039430A1 (ja) * | 2010-09-21 | 2012-03-29 | 株式会社Gpバイオサイエンス | がん幹細胞の単離方法 |
| US10130714B2 (en) | 2012-04-14 | 2018-11-20 | Academia Sinica | Enhanced anti-influenza agents conjugated with anti-inflammatory activity |
| JP6302909B2 (ja) | 2012-08-18 | 2018-03-28 | アカデミア シニカAcademia Sinica | シアリダーゼの同定および画像化のための細胞透過性プローブ |
| US9341619B2 (en) | 2013-03-14 | 2016-05-17 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Hyposialylation disorders |
| US10086054B2 (en) | 2013-06-26 | 2018-10-02 | Academia Sinica | RM2 antigens and use thereof |
| WO2014210564A1 (en) | 2013-06-27 | 2014-12-31 | Academia Sinica | Glycan conjugates and use thereof |
| CA2923579C (en) | 2013-09-06 | 2023-09-05 | Academia Sinica | Human inkt cell activation using glycolipids with altered glycosyl groups |
| SG11201510740YA (en) | 2013-09-17 | 2016-01-28 | Obi Pharma Inc | Compositions of a carbohydrate vaccine for inducing immune responses and uses thereof in cancer treatment |
| US9982041B2 (en) | 2014-01-16 | 2018-05-29 | Academia Sinica | Compositions and methods for treatment and detection of cancers |
| US10150818B2 (en) | 2014-01-16 | 2018-12-11 | Academia Sinica | Compositions and methods for treatment and detection of cancers |
| CN106415244B (zh) | 2014-03-27 | 2020-04-24 | 中央研究院 | 反应性标记化合物及其用途 |
| EP3797793A1 (en) | 2014-04-18 | 2021-03-31 | The Research Foundation of the State University of New York at Buffalo | Humanized anti-tf-antigen antibodies |
| US10023651B2 (en) | 2014-04-18 | 2018-07-17 | The Research Foundation For The State University Of New York | Humanized anti-TF-antigen antibodies |
| CN106661099A (zh) | 2014-05-27 | 2017-05-10 | 中央研究院 | 抗her2醣抗体及其用途 |
| US20150344585A1 (en) | 2014-05-27 | 2015-12-03 | Academia Sinica | Anti-cd20 glycoantibodies and uses thereof |
| US10118969B2 (en) | 2014-05-27 | 2018-11-06 | Academia Sinica | Compositions and methods relating to universal glycoforms for enhanced antibody efficacy |
| EP3904388A1 (en) | 2014-05-27 | 2021-11-03 | Academia Sinica | Fucosidase from bacteroides and methods using the same |
| EP3154582A4 (en) | 2014-05-28 | 2018-01-10 | Academia Sinica | Anti-tnf-alpha glycoantibodies and uses thereof |
| EP3191500A4 (en) | 2014-09-08 | 2018-04-11 | Academia Sinica | HUMAN iNKT CELL ACTIVATION USING GLYCOLIPIDS |
| EP4183806A3 (en) | 2014-11-12 | 2023-08-02 | Seagen Inc. | Glycan-interacting compounds and methods of use |
| US9975965B2 (en) | 2015-01-16 | 2018-05-22 | Academia Sinica | Compositions and methods for treatment and detection of cancers |
| US10495645B2 (en) | 2015-01-16 | 2019-12-03 | Academia Sinica | Cancer markers and methods of use thereof |
| CA2972072A1 (en) | 2015-01-24 | 2016-07-28 | Academia Sinica | Novel glycan conjugates and methods of use thereof |
| CN107430127B (zh) * | 2015-01-24 | 2020-08-28 | 中央研究院 | 癌症标记及其使用方法 |
| CA2979640A1 (en) | 2015-02-25 | 2016-09-01 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Sialylation-increasing therapies for diseases associated with oxidative stress |
| JP2018532990A (ja) | 2015-09-04 | 2018-11-08 | オービーアイ ファーマ,インコーポレイテッド | グリカンアレイおよび使用の方法 |
| CN108463248B (zh) | 2015-11-12 | 2022-10-21 | 西雅图基因公司 | 聚糖相互作用化合物及使用方法 |
| WO2017100796A1 (en) * | 2015-12-11 | 2017-06-15 | SINACA, Academia | Modulation of globoseries glycosphingolipid synthesis and cancer biomarkers |
| KR20180114210A (ko) | 2016-03-08 | 2018-10-17 | 아카데미아 시니카 | N-글리칸의 모듈 합성 방법 및 그의 어레이 |
| US10980894B2 (en) | 2016-03-29 | 2021-04-20 | Obi Pharma, Inc. | Antibodies, pharmaceutical compositions and methods |
| TWI780045B (zh) | 2016-03-29 | 2022-10-11 | 台灣浩鼎生技股份有限公司 | 抗體、醫藥組合物及方法 |
| AU2017252128B2 (en) | 2016-04-22 | 2024-06-06 | Obi Pharma, Inc. | Cancer immunotherapy by immune activation or immune modulation via Globo series antigens |
| KR20230117482A (ko) | 2016-07-27 | 2023-08-08 | 오비아이 파머 인코퍼레이티드 | 면역원성/치료 글리칸 조성물 및 그의 용도 |
| JP7121724B2 (ja) | 2016-07-29 | 2022-08-18 | オービーアイ ファーマ,インコーポレイテッド | ヒト抗体、医薬組成物及び方法 |
| US10538592B2 (en) | 2016-08-22 | 2020-01-21 | Cho Pharma, Inc. | Antibodies, binding fragments, and methods of use |
| WO2018094143A1 (en) | 2016-11-17 | 2018-05-24 | Siamab Therapeutics, Inc. | Glycan-interacting compounds and methods of use |
| SG10201912529SA (en) | 2016-11-18 | 2020-02-27 | Astellas Pharma Inc | Novel anti-human muc1 antibody fab fragment |
| WO2018094414A1 (en) | 2016-11-21 | 2018-05-24 | Obi Pharma, Inc. | Conjugated biological molecules, pharmaceutical compositions and methods |
| WO2018160909A1 (en) | 2017-03-03 | 2018-09-07 | Siamab Therapeutics, Inc. | Glycan-interacting compounds and methods of use |
| KR20190134994A (ko) * | 2017-03-29 | 2019-12-05 | 글리코토페 게엠베하 | Muc1 및 cd3에 결합하는 다중특이적 항체 작제물 |
| TWI795415B (zh) | 2017-07-07 | 2023-03-11 | 日商安斯泰來製藥股份有限公司 | 新穎的抗人類CEACAM5抗體Fab片段 |
| US20190292188A1 (en) | 2018-02-27 | 2019-09-26 | Incyte Corporation | Imidazopyrimidines and triazolopyrimidines as a2a / a2b inhibitors |
| PT110722B (pt) * | 2018-05-03 | 2021-08-17 | Instituto De Patologia E Imunologia Molecular Da Univ Do Porto Ipatimup | Uso de anticorpos para o antigénio thomsen-friedenreich num método e estojo para avaliação de instabilidade de microsatelites |
| MX2020012351A (es) | 2018-05-17 | 2021-01-29 | Astellas Pharma Inc | Complejo que tiene fragmento fab de anticuerpo anti-muc1 de humano, enlazador peptido y/o ligando. |
| MA52940A (fr) | 2018-05-18 | 2021-04-28 | Incyte Corp | Dérivés de pyrimidine fusionnés utilisés en tant qu'inhibiteurs de a2a/a2b |
| HUE067382T2 (hu) | 2018-05-18 | 2024-10-28 | Daiichi Sankyo Co Ltd | Anti-MUC1-exatecan antitest-hatóanyag konjugátum |
| TWI853822B (zh) | 2018-06-27 | 2024-09-01 | 台灣浩鼎生技股份有限公司 | 用於糖蛋白工程的糖苷合成酶變體及其使用方法 |
| CN113166153B (zh) | 2018-07-05 | 2024-11-01 | 因赛特公司 | 作为a2a/a2b抑制剂的稠合吡嗪衍生物 |
| EP3865154A4 (en) | 2018-10-10 | 2022-11-09 | Astellas Pharma Inc. | PHARMACEUTICAL COMPOSITION CONTAINING ANTIBODY FAB FRAGMENT COMPLEX AT LABELED ANTI-HUMAN SITE |
| TWI829857B (zh) | 2019-01-29 | 2024-01-21 | 美商英塞特公司 | 作為a2a / a2b抑制劑之吡唑并吡啶及三唑并吡啶 |
| CN110161241A (zh) * | 2019-04-30 | 2019-08-23 | 丁晓昆 | 一种快速检测生物组织切片cosmc基因突变试剂盒 |
| TWI873265B (zh) | 2020-01-03 | 2025-02-21 | 美商英塞特公司 | 抗cd73抗體及其用途 |
| PH12022551621A1 (en) | 2020-01-03 | 2023-10-23 | Incyte Corp | Cd73 inhibitor and a2a/a2b adenosine receptor inhibitor combination therapy |
| KR20230157940A (ko) | 2020-12-29 | 2023-11-17 | 인사이트 코포레이션 | A2a/a2b 저해제, pd-1/pd-l1 저해제 및 항-cd73 항체를포함하는 병용 요법 |
| JP2024504406A (ja) * | 2021-01-25 | 2024-01-31 | ユニオジェン・オサケユフティオ | 膀胱がんのバイオマーカーとしてのFUCα(1-2)Gal糖鎖構造の使用 |
| AU2022331944A1 (en) * | 2021-07-16 | 2024-02-01 | Université de Montréal | Novel tumor-specific antigens for cancer stem cells and uses thereof |
| CN115960169A (zh) * | 2021-10-09 | 2023-04-14 | 清华大学深圳国际研究生院 | 特异性靶向非小细胞肺癌肿瘤干细胞的多肽及其应用 |
| CN119955728A (zh) * | 2022-08-01 | 2025-05-09 | 首都医科大学附属北京胸科医院 | 恶性表型大细胞肺癌细胞株及其应用 |
| CN121319187B (zh) * | 2025-12-08 | 2026-03-17 | 宁波大学附属第一医院 | 一种与cd176蛋白结合的羊驼源纳米抗体及其应用 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020192223A1 (en) * | 1989-06-30 | 2002-12-19 | Ingegerg Hellstrom | Novel antibody conjugates reactive with human carcinomas |
| US20060251668A1 (en) * | 2002-11-29 | 2006-11-09 | Steffen Goletz | Tumor-specific recognition molecules |
| US7374755B2 (en) * | 2004-07-26 | 2008-05-20 | The Research Foundation Of State University Of New York | Therapeutic use of anti-TF-Antigen antibody |
| US20090041836A1 (en) * | 2006-01-03 | 2009-02-12 | University Of Georgia Research Foundation, Inc. | Glycolipopeptide and uses thereof |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SI1654353T1 (sl) | 2003-08-18 | 2013-09-30 | Glycotope Gmbh | Tumorski celični liniji NM-F9 (DSM ACC2606) in NM-D4 (DSM ACC2605), njihove uporabe |
| US20110190166A1 (en) * | 2006-05-26 | 2011-08-04 | Susan Wong | Erythroid progenitor cells and methods for producing parvovirus b19 therein |
-
2010
- 2010-01-20 EP EP10000527A patent/EP2347769A1/en not_active Withdrawn
-
2011
- 2011-01-20 AU AU2011208874A patent/AU2011208874B2/en not_active Ceased
- 2011-01-20 WO PCT/EP2011/000217 patent/WO2011089004A1/en not_active Ceased
- 2011-01-20 BR BR112012018132A patent/BR112012018132A2/pt active Search and Examination
- 2011-01-20 EP EP11703580A patent/EP2525832A1/en not_active Withdrawn
- 2011-01-20 US US13/574,260 patent/US20120294859A1/en not_active Abandoned
- 2011-01-20 CN CN2011800065922A patent/CN102753199A/zh active Pending
- 2011-01-20 CA CA2786212A patent/CA2786212A1/en not_active Abandoned
- 2011-01-20 JP JP2012549291A patent/JP2013517487A/ja not_active Ceased
-
2015
- 2015-12-25 JP JP2015253064A patent/JP2016047848A/ja not_active Withdrawn
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020192223A1 (en) * | 1989-06-30 | 2002-12-19 | Ingegerg Hellstrom | Novel antibody conjugates reactive with human carcinomas |
| US20060251668A1 (en) * | 2002-11-29 | 2006-11-09 | Steffen Goletz | Tumor-specific recognition molecules |
| US7374755B2 (en) * | 2004-07-26 | 2008-05-20 | The Research Foundation Of State University Of New York | Therapeutic use of anti-TF-Antigen antibody |
| US20090041836A1 (en) * | 2006-01-03 | 2009-02-12 | University Of Georgia Research Foundation, Inc. | Glycolipopeptide and uses thereof |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103969437A (zh) * | 2014-04-22 | 2014-08-06 | 广州恒泰生物科技有限公司 | 用于卵巢癌早中期快速诊断试剂盒的制备方法 |
| CN111363008A (zh) * | 2020-03-18 | 2020-07-03 | 北京鼎成肽源生物技术有限公司 | 一种输卵管癌靶标抗原组合、输卵管癌靶标抗原组合刺激培养的ctl细胞及其应用 |
| CN113388023A (zh) * | 2020-03-18 | 2021-09-14 | 北京鼎成肽源生物技术有限公司 | 一种输卵管癌靶标抗原、输卵管癌靶标抗原刺激培养的ctl细胞及其应用 |
| CN113429473A (zh) * | 2020-03-18 | 2021-09-24 | 北京鼎成肽源生物技术有限公司 | 一种输卵管癌靶标抗原、输卵管癌靶标抗原刺激培养的ctl细胞及其应用 |
| CN111363008B (zh) * | 2020-03-18 | 2021-11-16 | 北京鼎成肽源生物技术有限公司 | 一种输卵管癌靶标抗原组合、输卵管癌靶标抗原组合刺激培养的ctl细胞及其应用 |
| CN113429473B (zh) * | 2020-03-18 | 2022-11-04 | 北京鼎成肽源生物技术有限公司 | 一种输卵管癌靶标抗原、输卵管癌靶标抗原刺激培养的ctl细胞及其应用 |
| CN115916349A (zh) * | 2020-06-30 | 2023-04-04 | 葛莱高托普有限公司 | 针对Lewis Y的人源化抗体 |
| US12415864B2 (en) | 2020-06-30 | 2025-09-16 | Pentixapharm Ag | Humanized antibodies against Lewis Y |
| CN113804531A (zh) * | 2021-09-10 | 2021-12-17 | 北京大学 | 一种细胞高活性染色方法 |
| CN113804531B (zh) * | 2021-09-10 | 2024-02-06 | 北京大学 | 一种细胞高活性染色方法 |
| CN116718774A (zh) * | 2023-07-07 | 2023-09-08 | 杭州准星医学科技有限公司 | 一种用于肿瘤体外诊断的试剂盒及其使用方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2525832A1 (en) | 2012-11-28 |
| AU2011208874A1 (en) | 2012-08-02 |
| BR112012018132A2 (pt) | 2017-06-20 |
| US20120294859A1 (en) | 2012-11-22 |
| WO2011089004A1 (en) | 2011-07-28 |
| JP2013517487A (ja) | 2013-05-16 |
| EP2347769A1 (en) | 2011-07-27 |
| CA2786212A1 (en) | 2011-07-28 |
| JP2016047848A (ja) | 2016-04-07 |
| AU2011208874B2 (en) | 2015-06-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2011208874B2 (en) | Cancer stem cell markers and uses thereof | |
| JP5689133B2 (ja) | 結腸直腸癌におけるpodxlタンパク質 | |
| JP7022191B2 (ja) | 前立腺がんの分析のための組成物及び方法 | |
| US12287337B2 (en) | Autoantibody biomarkers of ovarian cancer | |
| JP6795813B2 (ja) | がん細胞の検出方法、がん細胞内に物質を導入するための試薬、及びがん治療用組成物 | |
| JPWO2008056833A1 (ja) | invivoで抗腫瘍活性を有する抗ヒトDlk−1抗体 | |
| JP2020125352A (ja) | 抗−cd43抗体およびその癌治療用途 | |
| RU2641968C2 (ru) | Композиции и способы анализа рака предстательной железы | |
| US12091457B2 (en) | Use for preventing and treating myeloid-derived suppressor cell-related diseases | |
| CN101553259A (zh) | 显示cd63表面表达的细胞的细胞毒性介导 | |
| JP2019528437A (ja) | 腫瘍関連マクロファージをターゲティングすることによるガン疾患の処置方法 | |
| Wagner et al. | Two birds with one stone: human SIRPα nanobodies for functional modulation and in vivo imaging of myeloid cells | |
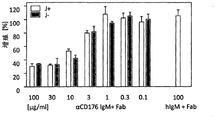
| Lin et al. | Co-expression of CD173 (H2) and CD174 (Lewis Y) with CD44 suggests that fucosylated histo-blood group antigens are markers of breast cancer-initiating cells | |
| US20150132291A1 (en) | Methods for treatment of gastric cancer | |
| JP2022520383A (ja) | 癌細胞上に存在するケラチン14(krt14)の細胞外部分を標的とすることによる癌の治療または予防のための方法 | |
| JP2022530339A (ja) | インテグリンα10および侵攻性癌型 | |
| US9206258B2 (en) | Anti-prostate cancer antibodies and methods of detection and treatment of prostate cancer using the same | |
| WO2025026204A1 (en) | Antibodies against ror1 and uses thereof | |
| WO2020171171A1 (ja) | 抗hla-dr抗体、およびそのがん治療用途 | |
| US20130143231A1 (en) | MST1/STK4 PHOSPHO-THREONINE 120 (pMST-T120) ANTIBODY | |
| BR112018007334B1 (pt) | Anticorpo anti-cd43, conjugado e composição do mesmo para tratamento de câncer | |
| HK1255484B (en) | Compositions and methods for prostate cancer analysis | |
| HK1255484A1 (en) | Compositions and methods for prostate cancer analysis |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20121024 |
|
| RJ01 | Rejection of invention patent application after publication |