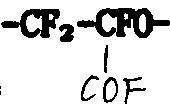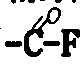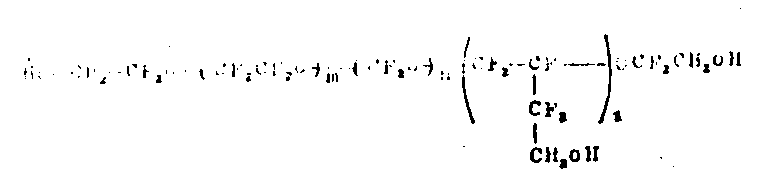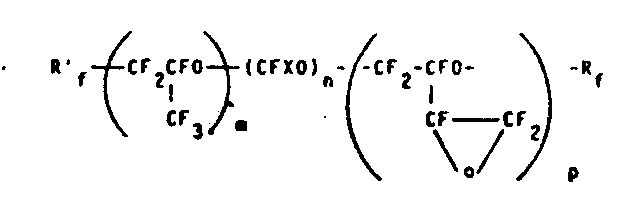CN1012175B - 沿主链含有全氟环氧基而无过基氧的全氟聚醚及其衍生物的制法 - Google Patents
沿主链含有全氟环氧基而无过基氧的全氟聚醚及其衍生物的制法Info
- Publication number
- CN1012175B CN1012175B CN87104043A CN87104043A CN1012175B CN 1012175 B CN1012175 B CN 1012175B CN 87104043 A CN87104043 A CN 87104043A CN 87104043 A CN87104043 A CN 87104043A CN 1012175 B CN1012175 B CN 1012175B
- Authority
- CN
- China
- Prior art keywords
- light
- formula
- peroxy
- product
- ing
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired
Links
- -1 Perfluoro Chemical group 0.000 title description 3
- 238000007254 oxidation reaction Methods 0.000 claims abstract description 19
- 239000000203 mixture Substances 0.000 claims abstract description 18
- 230000003647 oxidation Effects 0.000 claims abstract description 18
- 238000000034 method Methods 0.000 claims abstract description 15
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 claims abstract description 12
- 150000001993 dienes Chemical class 0.000 claims abstract description 11
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims abstract description 8
- 229910001882 dioxygen Inorganic materials 0.000 claims abstract description 8
- 229910052760 oxygen Inorganic materials 0.000 claims abstract description 8
- 239000001301 oxygen Substances 0.000 claims abstract description 8
- 238000007539 photo-oxidation reaction Methods 0.000 claims abstract description 7
- 239000007791 liquid phase Substances 0.000 claims abstract description 6
- 238000002360 preparation method Methods 0.000 claims description 14
- 239000000126 substance Substances 0.000 claims description 8
- 230000000694 effects Effects 0.000 claims description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 5
- 230000003351 photoxidation Effects 0.000 claims description 5
- 239000002994 raw material Substances 0.000 claims description 5
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 4
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 claims description 4
- 229910052794 bromium Inorganic materials 0.000 claims description 4
- 239000003638 chemical reducing agent Substances 0.000 claims description 4
- 238000006722 reduction reaction Methods 0.000 claims description 4
- 238000009826 distribution Methods 0.000 claims description 2
- 229910052731 fluorine Inorganic materials 0.000 claims description 2
- 230000021615 conjugation Effects 0.000 claims 4
- 125000003700 epoxy group Chemical group 0.000 abstract description 41
- 125000000864 peroxy group Chemical group O(O*)* 0.000 abstract description 18
- 125000000524 functional group Chemical group 0.000 abstract description 10
- 239000004593 Epoxy Substances 0.000 abstract description 5
- 239000010702 perfluoropolyether Substances 0.000 abstract description 5
- 238000000354 decomposition reaction Methods 0.000 abstract description 3
- 230000008569 process Effects 0.000 abstract description 3
- 150000005673 monoalkenes Chemical class 0.000 abstract 1
- 239000000047 product Substances 0.000 description 40
- 238000006243 chemical reaction Methods 0.000 description 32
- 150000001875 compounds Chemical class 0.000 description 16
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 9
- 238000010438 heat treatment Methods 0.000 description 8
- 229920000642 polymer Polymers 0.000 description 8
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 7
- 241000786363 Rhampholeon spectrum Species 0.000 description 6
- SBZXBUIDTXKZTM-UHFFFAOYSA-N diglyme Chemical compound COCCOCCOC SBZXBUIDTXKZTM-UHFFFAOYSA-N 0.000 description 6
- 238000010521 absorption reaction Methods 0.000 description 5
- 125000004185 ester group Chemical group 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- 238000001228 spectrum Methods 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- 150000001265 acyl fluorides Chemical class 0.000 description 4
- 125000001246 bromo group Chemical group Br* 0.000 description 4
- 150000002148 esters Chemical group 0.000 description 4
- 238000007540 photo-reduction reaction Methods 0.000 description 4
- 239000000376 reactant Substances 0.000 description 4
- 238000010992 reflux Methods 0.000 description 4
- LGPPATCNSOSOQH-UHFFFAOYSA-N 1,1,2,3,4,4-hexafluorobuta-1,3-diene Chemical compound FC(F)=C(F)C(F)=C(F)F LGPPATCNSOSOQH-UHFFFAOYSA-N 0.000 description 3
- KDSNLYIMUZNERS-UHFFFAOYSA-N 2-methylpropanamine Chemical class CC(C)CN KDSNLYIMUZNERS-UHFFFAOYSA-N 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 239000007795 chemical reaction product Substances 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 239000012530 fluid Substances 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 239000003880 polar aprotic solvent Substances 0.000 description 3
- 239000010453 quartz Substances 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N silicon dioxide Inorganic materials O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- 238000010183 spectrum analysis Methods 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- LOUICXNAWQPGSU-UHFFFAOYSA-N 2,2,3,3-tetrafluorooxirane Chemical compound FC1(F)OC1(F)F LOUICXNAWQPGSU-UHFFFAOYSA-N 0.000 description 2
- 229910010082 LiAlH Inorganic materials 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- 125000002947 alkylene group Chemical group 0.000 description 2
- 125000003368 amide group Chemical group 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000004132 cross linking Methods 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 125000001033 ether group Chemical group 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- HCDGVLDPFQMKDK-UHFFFAOYSA-N hexafluoropropylene Chemical group FC(F)=C(F)C(F)(F)F HCDGVLDPFQMKDK-UHFFFAOYSA-N 0.000 description 2
- 125000000468 ketone group Chemical class 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- 150000002924 oxiranes Chemical class 0.000 description 2
- 238000005502 peroxidation Methods 0.000 description 2
- 150000002978 peroxides Chemical class 0.000 description 2
- 238000006116 polymerization reaction Methods 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 238000005070 sampling Methods 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- PMJHHCWVYXUKFD-SNAWJCMRSA-N (E)-1,3-pentadiene Chemical compound C\C=C\C=C PMJHHCWVYXUKFD-SNAWJCMRSA-N 0.000 description 1
- ZXUJWPHOPHHZLR-UHFFFAOYSA-N 1,1,1-trichloro-2-fluoroethane Chemical compound FCC(Cl)(Cl)Cl ZXUJWPHOPHHZLR-UHFFFAOYSA-N 0.000 description 1
- RRZIJNVZMJUGTK-UHFFFAOYSA-N 1,1,2-trifluoro-2-(1,2,2-trifluoroethenoxy)ethene Chemical compound FC(F)=C(F)OC(F)=C(F)F RRZIJNVZMJUGTK-UHFFFAOYSA-N 0.000 description 1
- AVQQQNCBBIEMEU-UHFFFAOYSA-N 1,1,3,3-tetramethylurea Chemical compound CN(C)C(=O)N(C)C AVQQQNCBBIEMEU-UHFFFAOYSA-N 0.000 description 1
- 229910016569 AlF 3 Inorganic materials 0.000 description 1
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 1
- 239000007818 Grignard reagent Substances 0.000 description 1
- 241001597008 Nomeidae Species 0.000 description 1
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 1
- OUUQCZGPVNCOIJ-UHFFFAOYSA-M Superoxide Chemical compound [O-][O] OUUQCZGPVNCOIJ-UHFFFAOYSA-M 0.000 description 1
- BQODPTQLXVVEJG-UHFFFAOYSA-N [O].C=C Chemical compound [O].C=C BQODPTQLXVVEJG-UHFFFAOYSA-N 0.000 description 1
- 230000021736 acetylation Effects 0.000 description 1
- 238000006640 acetylation reaction Methods 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- MTHSVFCYNBDYFN-UHFFFAOYSA-N anhydrous diethylene glycol Natural products OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 235000011089 carbon dioxide Nutrition 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000003610 charcoal Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 229920006037 cross link polymer Polymers 0.000 description 1
- 239000003431 cross linking reagent Substances 0.000 description 1
- 238000006115 defluorination reaction Methods 0.000 description 1
- 238000007872 degassing Methods 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 230000008034 disappearance Effects 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- NBVXSUQYWXRMNV-UHFFFAOYSA-N fluoromethane Chemical compound FC NBVXSUQYWXRMNV-UHFFFAOYSA-N 0.000 description 1
- 150000004795 grignard reagents Chemical class 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 229960003132 halothane Drugs 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 230000008676 import Effects 0.000 description 1
- 229910052738 indium Inorganic materials 0.000 description 1
- 239000012442 inert solvent Substances 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 150000002576 ketones Chemical group 0.000 description 1
- 239000012263 liquid product Substances 0.000 description 1
- 230000001050 lubricating effect Effects 0.000 description 1
- 238000003760 magnetic stirring Methods 0.000 description 1
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 1
- 229910001512 metal fluoride Inorganic materials 0.000 description 1
- HZVOZRGWRWCICA-UHFFFAOYSA-N methanediyl Chemical compound [CH2] HZVOZRGWRWCICA-UHFFFAOYSA-N 0.000 description 1
- 230000000269 nucleophilic effect Effects 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- CMFNMSMUKZHDEY-UHFFFAOYSA-N peroxynitrous acid Chemical compound OON=O CMFNMSMUKZHDEY-UHFFFAOYSA-N 0.000 description 1
- 238000006552 photochemical reaction Methods 0.000 description 1
- 238000006068 polycondensation reaction Methods 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/02—Polycondensates containing more than one epoxy group per molecule
- C08G59/027—Polycondensates containing more than one epoxy group per molecule obtained by epoxidation of unsaturated precursor, e.g. polymer or monomer
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D303/00—Compounds containing three-membered rings having one oxygen atom as the only ring hetero atom
- C07D303/02—Compounds containing oxirane rings
- C07D303/08—Compounds containing oxirane rings with hydrocarbon radicals, substituted by halogen atoms, nitro radicals or nitroso radicals
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/14—Polycondensates modified by chemical after-treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/20—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the epoxy compounds used
- C08G59/32—Epoxy compounds containing three or more epoxy groups
- C08G59/34—Epoxy compounds containing three or more epoxy groups obtained by epoxidation of an unsaturated polymer
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/002—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from unsaturated compounds
- C08G65/005—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from unsaturated compounds containing halogens
- C08G65/007—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from unsaturated compounds containing halogens containing fluorine
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- General Chemical & Material Sciences (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Polyethers (AREA)
- Epoxy Compounds (AREA)
- Detergent Compositions (AREA)
- Epoxy Resins (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
Abstract
沿主链含有全氟环氧基而无过氧基氧的全氟聚醚,由全氟单烯与共轭全氟双烯的混合物经低温氧化(液相,使用分子氧,在紫外线存在下),随后将光氧化产物中的过氧基进行光化学分解(在0℃~160℃的温度范围内,用紫外光处理含过氧基的产品)而制得。所说的含环氧基全氟聚醚衍生物,其环氧基已被转化为官能团。
Description
本发明是有关沿其主链含有氟代环氧基的全氟聚醚,它从全氟烯烃和全氟双烯烃的混合物开始制备。
众所周知,全氟烯烃在紫外光存在下用分子氧进行的低温(-30℃~-60℃)光氧化作用,最初的反应产物是由一定次序的氧全氟烷撑单元组成的并含有不定数量过氧基(视反应条件而定)的全氟聚醚。除去过氧基以便得到稳定的全氟聚醚,这可采用加热方式来实现,即高温下长时间加热。见美国专利US3,715,378和US3,665,041。
从美国专利US3,451,907中得知:全氟丁二烯,它的单一物或它和全氟烯烃的混合物,在低温、液相和有紫外光存在时,用分子氧进行氧化作用,生成光氧化聚合物。它的特征是同时存在过氧基、氟代环氧基
以及酸性的-COF基。完全消除光氧化产物中的过氧基可按照已知技术中的加热方式来实现,即在至少200℃的温度下加热很长时间(若干小时)。环氧基必然同时分解,大部分转化为酸性的-COF基。
本发明的目的是制备新的全氟聚醚,它由按一定顺序排列的氧全氟烷撑结构单元所构成,其特点是沿主链含有全氟环氧基
而无过氧基。
为得到这样的产物,可先进行全氟烯烃和全氟共轭双烯的混合物的光氧化,随后把过氧基进行光化学分解,或随后对过氧基进行特殊的化学还原反应来分解,下面将予以介绍。
因为使用全氟烯烃作为原料,四氧乙烯和全氟丙烯是特别合适的。全氟双烯中,全氟丁二烯特别合适。
使用C2F4和C4F6作原料时,得到的全氟聚醚具有如下结构:
(Ⅰ)
当采用光化学反应来分解过氧基时,端基Rf和Rf′是-CF2COF或-CF3,它们之中至少有一个是-CF2COF。而当过氧基的化学还原反应是在醇ROH(R=C1~C6烷基)中用HI进行时,则端基Rf和Rf′为-CF2COOR。
m,n和p是非零的整数,m/n比值的范围在0.5~2之间,氧全氟烷撑结构单元沿主链随机分布。 (m+n)/(p) 比值的范围可以从3直到非常高的值,甚至高达40。较为可取的分子量范围在500~15,000之间。当使用C3F6和C4F6时,得到的全氟聚醚的分子式为:
(Ⅱ)
式中,X=F或CF3,m,n和p为非零的整数,Rf′和Rf为上面已经定义的端基。m/n比值的范围在5~40之间,m/p比值的范围在2~50之间,较为可取的分子量范围在500~8,000之间。
如果需要,制备全氟聚醚也可以使用占多数的全氟烯烃(例如C2F4+C3F6)与一种或多种共轭双烯的混合物。
制备的第一步是光氧化,实质上是按已知技术来进行的。全氟烯烃和全氟双烯的混合物溶解在合适的惰性溶剂特别是氯氟化碳(例如CCl2F2)溶剂中,用气态分子氧来进行光氧化,温度为-80℃~+50℃,较为可取的是-60℃-0℃,有紫外线存在,光氧化产物含有过氧基和全氟环氧基。
我们惊奇地发现,在控制的温度下把所说的过氧基进行光化学分解,过氧基可被消除而留下环氧基保持不变。获得无过氧基而保留未变环氧基的产物的合适条件是:采用波长为248~334nm的紫外光,温度控制在0℃~150℃(最为可取的是20℃~50℃)。
通过化学还原反应来消除过氧基并保留未变的环氧基也是可能的,例如在醇ROH(R=C1~C6的烷基)中用HI与之反应。
全氟聚醚主链上的环氧基的出现率是和起始混合物中双烯/单烯的比值成正比的。
作为双烯烃,除全氟丁二烯外,也可以是全氟戊二烯。
本发明的另一目的是制备二溴衍生物,式中Rf和R′f是-CF2Br。
在紫外光存在下和温度大约为100℃时,让溴与之反应,可以得到这些化合物,光化学氧化反应产物含有过氧基和环氧基:溴导入到过氧基的位置上,而环氧基保持不变,如欧洲专利EPO145,946中所述。
本发明还有一个目的,就是制备上面定义的产品的衍生物:将产品中的环氧基和/或端基Rf和R′f进行转化,以便得到多官能衍生物。当环氧基予先被转化为
或-COOR,例如分别为-CF2C-F或-CF2COOR(R=C1~C6烷基),则通过两个端基Rf和R′f以及环氧基的转化而取得的衍生物可以是所有的同一类型的官能团。否则,从Rf和R′f衍生的官能团一般不同于从环氧基衍生的官能团。
所说的官能团是适于通过缩聚或加聚反应来制备聚合物的那种官能团,或者对树脂或聚合物材料起交联剂作用的那种官能团。所得聚合产物的特点是具有下列性质:
-高的热稳定性,
-低的二级有序转变温度,
-憎水性和憎油性,
-低的折光率。
作为更感兴趣的由熟知反应可取得的官能团,我们可以列出下面这些:
-CONHR(R=H或C1~C6烷基或环氧基)
-CN
-CHR′OH(R′=H或-CF3)
-CH2NH2
从上面的主要官能团中,能够通过已知反应而得到许多其它官能团,例如美国专利US3810874;3847978;欧洲专利165649和165650中所描述的官能团。这样得到的产物有同样的用途,其特征是具有前
面讲的聚合衍生物的性质,也具有良好的润滑性能。
不改变端基Rf和R′f而转化环氧基是可能的,例如通过下列反应:
a)在180℃~200℃进行热处理,每个环氧基按下式以气态付产品的形式(成C2F4;C3F6或更高的氟代烯烃)消除一个CF2,
起始产物可以是通式Ⅰ和通式Ⅱ所代表的物质(m,n,p为任何值,Rf和R′f也可以是-CF2Br)。
b)在极性的质子惰性溶剂(例如二乙二醇二甲醚)中,在50℃用KF处理,把环氧基
转化为
基,起始产物可以是通式Ⅰ和通式Ⅱ所代表的物质(m,n,p为任何值,Rf和R′f也可以是-CF2Br)。
c)与化学计量的格氏试剂RMgHal反应,把环氧基-CF-CF2转化为
(Hal是除氟以外的卤素)。
起始化合物为通式Ⅰ和通式Ⅱ的物质,式中Rf和R′f为-CF2COOR。
Rf和R′f所代表的
,或按照反应a),b)及c)由环氧基转化而得到的
基都可以与醇ROH(R=C1~C3烷基)反应,很容易转化成酯基-COOR,这些酯基适用于进行许多进一步的反应,而同时没有游离酰氟基的某些缺点。
d)起始化合物为通式Ⅰ和Ⅱ的物质,其中Rf和R′f是-CF2COOR时,环氧基
转化为酮基-COCF3。在有AlF3存在时于100℃加热就可实现这个反应。该酮基令人感兴趣的下一步反应是该物质用H2(催化剂是炭上的钯)进行还原,把酮基转化为-CHOHCF3基,而端基-CF2COOR保持不变。这样的沿主链引入的羟基-OH可被用于以合适的交联剂(例如二环氧化合物,二异氰酸酯,等等)来进行的交联反应中。
e)Rf和R′f是-CF2Br或-CF2COOR的通式Ⅰ和提通式Ⅱ的化合物中,环氧基的进一步反应是它们在CSF或四甲基脲存在时,在极性的质子惰性溶剂中,环氧基发生聚合反应。得到的聚合物具有全氟聚醚结构,是线型或交联型,并且具有众多的活性基-CF2COOR或-CF2Br。
按上述通过环氧基的转化而变成的-COCF3,
基,可以进一步转变成全氟乙烯醚基。该化合物适于制备含有-CF2Br或-CF2COOR这类端基Rf和Rf′的加成聚合物。通过加入碱金属氟化物CSF和/或KF,随后在极性的质子惰性溶剂中再加入
,即可实现这个转化。如此得到的含有一个酰氟基的衍生物,在120°~220℃,在有碱性物质存在时,生成全氟乙烯醚衍生物。
以上所说的起始化合物是通式Ⅰ和Ⅱ中,Rf和Rf′为-CF2Br或-CF2COOR,且其中的环氧基已予先转化为
酰氟基的化合物。
这样所得的全氟乙烯基醚的特征是分别含有基团-CF2CF2OCF=CF2或-CF2OCF=CF2。
f)环氧基的另一个有意思的转化是按下式变成-Br:
已经说过,本发明的多官能全氟聚醚可被用来制备交联聚合物,或者作为缩合聚合物的交联剂。
在每个全氟聚醚上仅有一个环氧基的通式Ⅰ和Ⅱ的化合物中,通过亲核聚合能得到线型聚合物。
这些线型聚合物可被用作具有多种用途的液膜,这取决于聚合物侧链上的官能团的类型(例如-COOH或-SO3H具有离子交换性质)。
通式为Ⅰ和Ⅱ的化合物或其衍生物一般用来作为涂料,高级润滑剂和边界润滑剂。
下面的实施例用来作为本发明的例证,但并不限于此。
实施例1
A)过氧化合物的制备
采用一个600ml的光化学反应器,光程0.5mm,装上一个温度保持在-80℃的回流冷凝器,用带套管的热电偶检测温度。反应器配有用FC75(R)冷却的石英套管系统以便插入紫外灯(HANAU,TQ150型),其波长范围是248~334nm。
把反应器用干冰-丙酮冷却后加入460mlCF2Cl2,然后将它保温在~50℃,在4.3小时内加入96克O2(3.0摩尔),100克C2F4(1.0摩尔)和14.1克C4F6(0.087摩尔),进入反应器的气态物质都要通过CaCl2阱。反应到达终点后,蒸发掉溶剂,得46克油状物。油状产物含有3.96%(重量)的活性(过氧)氧。根据19F-N.M.R谱和I.R.谱的数据,该产品的结构可表征为由醚链和/或过氧桥链把结构单元-CF2-,-CF2-CF2-按一定顺序连接起来,和表征为仅由醚链把结构单元
在20℃时的粘度为4,300CSt
(厘沲),分子量由G.P.C.测定,经N.M.R.核实)为7,200。环氧基的含量(由19F-N.M.R.计算出)为每个聚合链有2.7个单位。
B)氧化能力为零的化合物的制备
将23克从光合成制得的产物溶解在230ml的CFCl2CF2Cl中,放在300ml反应器内。反应器的光程为0.5mm,配有保持在-10℃的回流冷凝器和套管热电偶以检测温度。该系统配有以FC75(R)冷却的石英套管以便插入紫外灯(HANAU,TQ150型)(FC75(R)是3M公司生产的氟碳化合物),光还原反应在温度为30℃时进行30小时。反应后得出13.9克产品,经碘量法分析,证明(过氧)活性氧含量为零。根据19F-N.M.R.谱和I.R.谱分析,该产品的结构可表示为:结构单元-CF2O-,-CF2-CF2O-,
按一定的顺序连接,端基为-CF2COF-型。可能存在的主链内的-CF-基与环氧基中的(b)-CF2-部分的克分子比保持恒定。事实上,19F-N.M.R、没有检测到主链内有
。
产物的分子量测得为5,400。光还原反应后主链内环氧基的含量为每个聚合链含2.65个单位。
实施例1A(比较实施例)
实施例1中A步反应所得到的产物的O.P.(氧化能力)=3.96%(重量),取其一等分试样来进行热处理,以便降低它的氧化能力。将20克聚合物装入一个配有温度计和搅拌器的50ml烧瓶中,在两小时内温度升到230℃,然后将反应物在230~240℃再保温6小时。在反应终点得到12.1克产品,经碘量法测定,证明氧化能力为零。19F-N.M.R.谱分析没有证实有环氧基的存在,但出现了
基的典型峰值。
〔δ(b)〕=+26.4,δ(a)=-130,-131.5〕
实施例1B(比较实施例)
取1份实施例1中A步反应所得的氧化能力O.P.=3.96%(重量)的油状物进行热处理。将20克油状物放入装有温度计和搅拌器的50ml烧瓶内。烧瓶放在加热浴中,全体反应物恒定在160℃。经52小时加热后,产物经N.M.R.分析,知其氧化能力为2.7%(重量),环氧含量为起始环氧含量的80%。
实施例2
A)过氧化物的制备
往与实施例1相同的装置中,装入460ml的CF2Cl2,然后将它维持在-47℃。在6小时内加入了101.2克O2(3.16摩尔),74.6克C2F4(0.74摩尔)和25.4克C4F6(0.16摩尔)到达反应终点后,蒸发去溶剂,得59.8克油状物。本制法得到的油状物含活性氧3.64%(重量)。据19F-N.M.R.谱和I.R.谱分析,证实本化合物的结构由与实施例1同样的重复结构单元所组成。环氧含量(由19F-N.M.R.谱计算)为每个聚合链含10.4个单位。分子量是7,500。
B)氧化能力为零的化合物的制备
把光合成得到的过氧化产物25克溶解在230ml CF2ClCFCl2中,加至实施例1所述的300ml反应器内。在温度为30℃进行28小时的光还原反应。在反应终点,从反应器中得到15.9克氧化能力为零的产物。产品的分析表明存在-CF2O-,-CF2-CF2O-,
(a)
型的结构。端基为-CF2COF型的结构。19F-N.M.R.分析〔δ(b)=-110,-113;δ(a)=-132.8,-136.5;δ(c)=-147.7)以及I.R.分析〔(
=1540cm-1〕证实了在这种处理过程中环氧基的稳定性。产物的分子量为6200。
实施例2A(比较实施例)
取实施例2中A步制得的氧化能力为3.64%(重量)的油状物20克,参照实施例1的类似方式进行处理。在两小时内,把温度升至230℃,并在230~240℃保温6小时以上。在反应终点,取得12.7克氧化能力为零的产物。19F-N.M.R.谱表明:
基出现了,环氧基被代替了。
实施例3
A)过氧化合物的制备
往实施例1的同样装置中,装入460mlCF2Cl2。反应器维持在-40℃,在紫外光下,在4.4小时内加入95.6克(3摩尔)O2,117.2克(1.17摩尔)C2F4和9.4克(0.058摩尔)C4F6,在反应终点,蒸发掉溶剂,得40.2克油状产物。这种油状物是含有3.17%(重量)活性氧的聚合物。经过19F-N.M.R和I.R.测定,证实它由与实施例1的产品相同的结构单元按一定顺序而组成,存在-CF3和-CF2COF型的端基,二者的比为0.5∶1。产物粘度为3000CSt,分子量为5700。环氧基含量为每条主链含2.1个单位。
B)氧化能力为零的化合物的制备
取20克光合成制得的过氧化产品溶解在230mlCF2ClCFCl2内,并装入一个在实施例1中已描述过的
300ml光化学反应器内。在30℃进行28小时的光还原反应。在反应终点,从反应器得到13.7克氧化能力为零的产物。经分析,证明存在-CF2O-,-CF2-CF2-O-,
,端基是-CF2COF和-CF3型。这些端基间的比例和前面指出的比例是一样的。产品的分子量是4250。
实施例3A(比较实施例)
取15克光合成所得到的氧化物能力为3.17%(重量)的样品,照实施例1的方式进行处理。两小时内把反应物加热至230℃,在230~240℃再继续保温6小时。在反应终点得10.2克无过氧基产品。分析表明,环氧基消失,
基出现。
实施例4
往20ml57%HF,50ml甲醇和150ml.1.1.2-三氟-三氯乙烷制成的混合物中,滴加100克实施例2A得到的产品。将反应混合物在沸腾温度保持8小时。把反应混合物倒进冰水中,分离出重液层,然后将重液层用重量比为1∶1的甲醇/36%盐酸混合物洗涤。重液层用Na2SO4干燥,然后蒸去氟氯化碳溶剂。残留82克油状物。经I.R.分析表明有酯基的特殊吸收谱带(1800cm-1)。经N.M.R.分析知比值m/n=0.9。经测定,分子量为730,酸当量为355,化合物的结构相当于通式Ⅰ(式中的P=1)。
实施例5
取10克实施例4的产品装到50ml玻璃烧瓶内。
加入1克a-AlF3。把此混合物加热到100℃并搅拌。
反应8小时后,将所得的混合物冷却,过滤,回收全部反应产物。N、M、R、分析表明,存在有
基(δ(CF3-
)=-75ppm,对CF-CO-CF3而言),而完全不存在环氧基。
实施例6
在一个100ml玻璃烧瓶中,把0.3克无水KF分散到50ml二乙二醇二甲醚(CH3O-CH2CH2-O-CH2CH2-OCH3)中。
取20克实施例4得到的产品,在50℃逐滴加到上述分散液中。将整个混合物搅拌4小时,然后冷却,过滤。
接着用真空蒸馏分离掉二乙二醇二甲醚,残留物用N、M、R、对19F进行分析,表明环氧基完全消失,-CF2-COF基出现。
用甲醇进行处理,所得产品转化成为相应的三酯。酸当量为240,分子式为
实施例7
先制备4.5克无水KF在150ml无水二乙二醇二甲醚中的悬浮液。取实施例5制备的50克产品于室温在无水气氛下加到悬浮液中;搅拌两小时,在此期间固态KF差不多完全消失。
将20克六氟丙烯环氧化物以气泡状通入混合物中。
盐立刻沉淀出来,其量随环氧化物加入量的增加而变多。
混合物脱气,把过量的全氟丙烯环氧化物去掉,在无水气氛下过滤;分出最重的液层,除掉二乙二醇二甲醚后,I.R.分析表明,有酰氟基(
)谱带(1884cm-1)存在,无酮键谱带(1803cm-1)存在。在二乙二醇二甲醚中用过量的无水Na1CO3对所得的产物进行两次处理,一次为80℃,60分钟;另一次为120~140℃,60分钟。
过滤,分离出的产物表明含有全氟乙烯醚的典型红外谱带(1840cm-1)和酯基的典型红外谱带(1800cm-1)
N.M.R.分析证实了下列结构:
实施例8
在容量1升的四颈烧瓶中放入16克LiAlH4和500ml无水乙醚,回流下在1小时内将实施例6得到的50克三酯一滴一滴地加入。
反应4小时后,加入溶于60ml四氢呋喃中的15ml水以便分解过量的LiAlH4。
搅拌两小时后,加入盐酸(33%)和水的混合物(体积比为1/4)150ml。
然后分离出醚层,用Na2SO4脱水,再蒸去乙醚,余留的液态产品经I.R.分析,知道在-OH区有强吸收,
而在C=O区无吸收。
以乙酰化法测得羟基当量为235。
′H的N.M.R.分析表明,亚甲基CH2存在于-CF2和-OH之间(化学位移大约是4ppm),因此相应于分子式:
N.M.R.19F分析证实了这个结构。
实施例9
将实施例6所得的三酯取50克溶在1.1.2-三氯-1.2.2-三氟乙烷中。
于室温下在一小时内加入10克异丁胺。
一小时后取样,蒸去溶剂,干燥,所得产品经红外分析,表明存在酯基的确凿吸收(1800cm-1),其附近是酰胺基谱带(1712cm-1)。
证实了有过量的异丁胺存在,因此反应要再进行三小时。
这个反应时间以后,反应不再显示酯基的任何痕量的红外吸收。
除去溶剂和过量的胺后,分离出粘性的油状物。
用过量的P2O5于150~170℃将油状物处理3~5小时,再把多磷酸成功地分离掉。观察到了在2277cm-1处有CN基的特征红外吸收,也观察到了酰胺基谱带的消失。
实施例10
500ml光化学反应器,光程0.5cm,装上同轴石英套管(以便插入HANAU,TQ150型汞蒸汽灯),磁力搅拌器,回流冷却器,干冰阱和一套能同时控制反应器及套管温度的系统。将实施例2A中得到的产品取400克放入反应器。
连续地加入10克溴素,把系统加热至100℃。
紫外灯亮后,在15小时的试验中再加入50克溴素。
在反应终点,把反应物倒进烧瓶,真空下蒸馏除去溴素。
所得物平均分子量为700,几乎无氧化能力,重300克,它的端基是-CF2Br,一个全氟聚醚链有两个溴代端基和一个环氧基
。
Claims (5)
1、下面通式(Ⅰ)或(Ⅱ)表示的全氟聚醚的制备方法,
(Ⅰ)
式中:
Rf和R′f是端基-CF2COF或-CF3,至少有一个是-CF2COF,或者Rf和R′f是-CF2COOR,或-CF2Br;
m,n和p是非零的整数,比值m/n的范围在0.5~2之间,氧氟代烷撑结构单元沿主链随机分布;
比值 (m+n)/(p) 的范围从3至40,
或者
式中:
X=F或CF3,m,n和p是非零的整数,R′f和Rf是上面所定义的特定端基;比值m/n的范围从5至40之间,比值m/p的范围在2到50之间,
该方法包括:在紫外光存在下,于-80℃至50℃的温度范围内使含有至少一种全氟烯烃和至少一种共轭全氟二烯的液相混合物在分子氧的作用下进行光氧化,随后将存在于光化学氧化产物中的过氧基团用下述方法分解:
a)在0-160℃用波长为248至334μm的紫外光进行处理;或
b)在化学式为ROH(式中R为C1-C6烷基)的醇中与HI反应。
2、按照权利要求1的所述的制备全氟聚醚的方法,其中步骤(a)在澳存在下进行。
3、按照权利要求1所述的制备全氟聚醚的方法,其中,在起始原料中R′f和Rf是-COF或-CF3,该方法具体包括在紫外光存在下、于-80℃至50℃的温度范围内使含有至少一种全氟烯烃和至少一种共轭全氟二烯的液相混合物在分子氧的作用下进行光化学氧化,随后在0-160℃用波长为248至334μm的紫外光进行处理,使存在于光氧化产物中的过氧基团分解。
4、按照权利要求1所述的制备全氟聚醚的方法,其中,在起始原料中R′f和Rf是-CF2Br,该方法具体包括:在紫外光存在下,于-80℃至50℃的温度范围内使含有至少一种全氟烯烃和至少一种共轭全氟二烯的液相混合物在分子氧的作用下进行光化学氧化,随后在溴存在下,用波长为248至334μm的紫外光进行处理,使存在于光氧化产物中的过氧基团分解。
5、按照权利要求1所述的制备全氟聚醚的方法,其中,在起始原料中R′f和Rf是-CF2COOR,该方法具体包括:在紫外光存在下,于-80至50℃的温度范围内使含有至少一种全氟烯烃和至少一种共轭全氟二烯的液相混合物在分子氧的作用下进行光化学氧化,随后在醇ROH(式中R为C1-C6烷基)中用HI进行化学还原,使存在于光氧化产物中的过氧基团分解。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IT8620346A IT1213071B (it) | 1986-05-07 | 1986-05-07 | Perfluoropolieteri esenti da ossigeno perossidico e contenenti gruppi perfluoroepossidici disposti lungo la catena perfluoropolieterea. |
| IT20346A/86 | 1986-05-07 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN87104043A CN87104043A (zh) | 1988-01-13 |
| CN1012175B true CN1012175B (zh) | 1991-03-27 |
Family
ID=11165912
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN87104043A Expired CN1012175B (zh) | 1986-05-07 | 1987-05-07 | 沿主链含有全氟环氧基而无过基氧的全氟聚醚及其衍生物的制法 |
Country Status (18)
| Country | Link |
|---|---|
| US (2) | US4853097A (zh) |
| EP (1) | EP0244839B1 (zh) |
| JP (1) | JPH0822911B2 (zh) |
| KR (1) | KR960009686B1 (zh) |
| CN (1) | CN1012175B (zh) |
| AT (1) | ATE114686T1 (zh) |
| AU (1) | AU596176B2 (zh) |
| CA (1) | CA1339316C (zh) |
| CS (1) | CS272774B2 (zh) |
| DD (1) | DD263769A5 (zh) |
| DE (1) | DE3750786T2 (zh) |
| ES (1) | ES2065316T3 (zh) |
| FI (1) | FI871983A (zh) |
| IL (1) | IL82423A (zh) |
| IT (1) | IT1213071B (zh) |
| NO (1) | NO167928C (zh) |
| RU (1) | RU1807991C (zh) |
| ZA (1) | ZA873178B (zh) |
Families Citing this family (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS62297478A (ja) * | 1986-06-18 | 1987-12-24 | Nippon Soda Co Ltd | 表面処理剤およびその製造方法 |
| IT1216565B (it) * | 1988-04-08 | 1990-03-08 | Ausimont Spa | Impiego di perfluoropolieteri in forma di microemulsione acquosa per la protezione di materiali lapidei dagli agenti atmosferici. |
| IT1218205B (it) * | 1988-04-08 | 1990-04-12 | Ausimont Spa | Impiego di derivati dei perfluoropolieteri in forma di microemulsione acquosa per la protezione dei materiali lapidei dagli agenti atmosferici |
| IT1218206B (it) * | 1988-04-08 | 1990-04-12 | Ausimont Spa | Impiego di emulsioni di perfluoropolieteri per la protezione di materiali lapidei dagli agenti atmosferici |
| IT1218215B (it) * | 1988-04-11 | 1990-04-12 | Ausimont Spa | Prodotti reticolati a struttura di perfluoropolieteri aventi gruppi funzionali |
| IT1217838B (it) * | 1988-06-17 | 1990-03-30 | Ausimont Spa | Microemulsioni per tipo acqua in olio elettricamente conduttrici,a base di composti perfluorurati usate come catolita in processi elettrochimici |
| IT1217839B (it) * | 1988-06-17 | 1990-03-30 | Ausimont Spa | Microemulsioni del tipo olio in acqua elettricamente conduttrici,a base di composti perfluorurati,usate come catolita in processi elettrochimici |
| IT1229845B (it) * | 1989-04-20 | 1991-09-13 | Ausimont Srl | Procedimento per la preparazione di perfluoropolieteri perossidici. |
| JPH04122412A (ja) * | 1990-09-12 | 1992-04-22 | Hitachi Ltd | 酸素の溶解方法及び酸素溶解装置 |
| IT1265068B1 (it) * | 1993-05-18 | 1996-10-30 | Ausimont Spa | Processo di (co)polimerizzazione radicalica di monomeri olefinici fluorurati in emulsione acquosa |
| IT1264977B1 (it) * | 1993-11-17 | 1996-10-17 | Ausimont Spa | Processo per preparare perfluoropolietri |
| FR2717450B1 (fr) * | 1994-03-21 | 1996-05-15 | Oreal | Conditionnement en matériau plastique composite présentant un effet doux au toucher. |
| IT1270703B (it) * | 1994-11-17 | 1997-05-07 | Ausimont Spa | Microemulsioni di fluoropoliossialchileni in miscela con idrocarburi, e loro uso in processi di (co)polimerizzazione di monomeri fluorurati |
| IT1271075B (it) * | 1994-11-21 | 1997-05-26 | Ausimont Spa | Miscele ternarie di solventi, e loro uso per la rimozione di sostanze oleose |
| IT1276072B1 (it) | 1995-10-31 | 1997-10-24 | Ausimont Spa | Processo di (co)polimerizzazione di monomeri fluorurati per ottenere polimeri contenenti idrogeno |
| IT1284005B1 (it) * | 1996-04-23 | 1998-05-08 | Ausimont Spa | Processo per la praparazione di perfluoropolieteri con terminali bromurati o iodurati |
| IT1286027B1 (it) * | 1996-06-10 | 1998-07-07 | Ausimont Spa | Rivestimenti protettivi a base di perfluoropolieteri funzionalizzati |
| IT1290462B1 (it) * | 1997-04-08 | 1998-12-03 | Ausimont Spa | Polimeri idrogenati modificati |
| WO1999014285A1 (en) | 1997-09-15 | 1999-03-25 | Sofitech N.V. | Electrically conductive non-aqueous wellbore fluids |
| US6405809B2 (en) | 1998-01-08 | 2002-06-18 | M-I Llc | Conductive medium for openhold logging and logging while drilling |
| US6793025B2 (en) * | 1998-01-08 | 2004-09-21 | M-I L. L. C. | Double emulsion based drilling fluids |
| US7320829B2 (en) * | 1998-03-05 | 2008-01-22 | Omnova Solutions Inc. | Fluorinated polymer and amine resin compositions and products formed therefrom |
| IT1313597B1 (it) | 1999-08-04 | 2002-09-09 | Ausimont Spa | Lubrificanti perfluoropolieterei contenenti gruppi solfonilfluoruro |
| US6558803B1 (en) | 2000-07-03 | 2003-05-06 | Adhesives Research Inc. | Ambifunctional perfluorinated polyethers |
| ITMI20010008A1 (it) | 2001-01-03 | 2002-07-03 | Ausimont Spa | Additivi per fluoropolieterei per applicazioni elettromagnetiche |
| US7342080B2 (en) * | 2004-05-07 | 2008-03-11 | 3M Innovative Properties Company | Polymerizable compositions, methods of making the same, and composite articles therefrom |
| US7288619B2 (en) * | 2004-05-07 | 2007-10-30 | 3M Innovative Properties Company | Fluorinated polyether polyamine and method of making the same |
| US8946136B2 (en) * | 2008-08-11 | 2015-02-03 | Solvay Specialty Polymers Italy S.P.A. | Hydrofluoroalcohols with improved thermal and chemical stability |
| JP7345454B2 (ja) | 2017-09-08 | 2023-09-15 | ソルベイ スペシャルティ ポリマーズ イタリー エス.ピー.エー. | フルオロポリマーを製造する方法 |
| US20220033578A1 (en) | 2018-10-01 | 2022-02-03 | Solvay Specialty Polymers Italy S.P.A. | Fluoroelastomer composition |
| WO2023117657A1 (en) | 2021-12-21 | 2023-06-29 | Solvay Specialty Polymers Italy S.P.A. | Perfluoropolyether polymers comprising pendant functional groups |
| TW202419516A (zh) | 2022-09-28 | 2024-05-16 | 義大利商首威專業聚合物義大利公司 | 全氟聚醚聚合物 |
| WO2024068676A1 (en) | 2022-09-28 | 2024-04-04 | Solvay Specialty Polymers Italy S.P.A. | Perfluoropolyether polymers |
| TW202419517A (zh) | 2022-09-28 | 2024-05-16 | 義大利商首威專業聚合物義大利公司 | 全氟聚醚聚合物 |
| WO2024149802A1 (en) | 2023-01-12 | 2024-07-18 | Solvay Specialty Polymers Italy S.P.A. | Perfluoropolyether polymers |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NL6709067A (zh) * | 1964-04-09 | 1968-01-08 | ||
| US3442942A (en) * | 1964-04-09 | 1969-05-06 | Montedison Spa | Fluorinated oxygen containing acyl fluorides |
| FR1499094A (zh) * | 1965-11-18 | 1968-01-11 | ||
| US3715378A (en) * | 1967-02-09 | 1973-02-06 | Montedison Spa | Fluorinated peroxy polyether copolymers and method for preparing them from tetrafluoroethylene |
| US3665041A (en) * | 1967-04-04 | 1972-05-23 | Montedison Spa | Perfluorinated polyethers and process for their preparation |
| US3847978A (en) * | 1968-07-01 | 1974-11-12 | Montedison Spa | Perfluorinated linear polyethers having reactive terminal groups at both ends of the chain and process for the preparation thereof |
| US3810874A (en) * | 1969-03-10 | 1974-05-14 | Minnesota Mining & Mfg | Polymers prepared from poly(perfluoro-alkylene oxide) compounds |
| SE411763B (sv) * | 1971-08-05 | 1980-02-04 | Montedison Spa | Amorf, mycket viskos polymer sampolyeter med hog molekylvikt vars makromolekyler er bildade av monomerenheterna -cf?712cf?712o- och -cf?712o- samt sett for dess framstellning |
| IT1150705B (it) * | 1982-03-19 | 1986-12-17 | Montedison Spa | Processo di inserimento di perfluoroolefine su perfluoropolieteri e prodotti relativi |
| IT1174205B (it) * | 1984-06-19 | 1987-07-01 | Montedison Spa | Fluoroplieteri contenenti gruppi terminali dotati di proprieta' ancoranti |
| US4664766A (en) * | 1985-02-13 | 1987-05-12 | Montedison S.P.A. | Photochemical process for neutralizing perfluoropolyethers |
| IT1185518B (it) * | 1985-02-22 | 1987-11-12 | Montefluos Spa | Ottenimento di perfluoropolieteri a peso molecolare controllato dal prodotto di ossidazione fotochimica del c2f4 |
| KR900007874B1 (ko) * | 1985-02-26 | 1990-10-22 | 몬테디손 에스. 페. 아. | 브롬화된 말단기 및 조절된 분자량을 갖는 이작용성 및 단작용성 과플루오로폴리에테르의 제조방법 |
| IT1200604B (it) * | 1985-04-04 | 1989-01-27 | Montefluos Spa | Processo di fluorurazione in fase liquida di composti insaturi |
-
1986
- 1986-05-07 IT IT8620346A patent/IT1213071B/it active
-
1987
- 1987-05-04 US US07/045,541 patent/US4853097A/en not_active Expired - Lifetime
- 1987-05-04 AU AU72475/87A patent/AU596176B2/en not_active Ceased
- 1987-05-04 ZA ZA873178A patent/ZA873178B/xx unknown
- 1987-05-05 FI FI871983A patent/FI871983A/fi not_active IP Right Cessation
- 1987-05-05 IL IL82423A patent/IL82423A/xx unknown
- 1987-05-05 CS CS315887A patent/CS272774B2/cs unknown
- 1987-05-06 NO NO871882A patent/NO167928C/no unknown
- 1987-05-06 RU SU874202565A patent/RU1807991C/ru active
- 1987-05-06 AT AT87106529T patent/ATE114686T1/de active
- 1987-05-06 ES ES87106529T patent/ES2065316T3/es not_active Expired - Lifetime
- 1987-05-06 EP EP87106529A patent/EP0244839B1/en not_active Expired - Lifetime
- 1987-05-06 JP JP62109058A patent/JPH0822911B2/ja not_active Expired - Lifetime
- 1987-05-06 DE DE3750786T patent/DE3750786T2/de not_active Expired - Lifetime
- 1987-05-06 CA CA000536523A patent/CA1339316C/en not_active Expired - Lifetime
- 1987-05-06 DD DD87302504A patent/DD263769A5/de not_active IP Right Cessation
- 1987-05-07 KR KR87004477A patent/KR960009686B1/ko not_active IP Right Cessation
- 1987-05-07 CN CN87104043A patent/CN1012175B/zh not_active Expired
-
1990
- 1990-07-30 US US07/561,251 patent/US5059700A/en not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| JPH0822911B2 (ja) | 1996-03-06 |
| FI871983A (fi) | 1987-11-08 |
| NO871882L (no) | 1987-11-09 |
| US5059700A (en) | 1991-10-22 |
| IT8620346A0 (it) | 1986-05-07 |
| DE3750786D1 (de) | 1995-01-12 |
| ATE114686T1 (de) | 1994-12-15 |
| JPS6322828A (ja) | 1988-01-30 |
| CS315887A2 (en) | 1990-04-11 |
| KR960009686B1 (en) | 1996-07-23 |
| EP0244839B1 (en) | 1994-11-30 |
| EP0244839A2 (en) | 1987-11-11 |
| IL82423A (en) | 1991-06-10 |
| US4853097A (en) | 1989-08-01 |
| EP0244839A3 (en) | 1988-11-17 |
| NO167928B (no) | 1991-09-16 |
| FI871983A0 (fi) | 1987-05-05 |
| NO871882D0 (no) | 1987-05-06 |
| IT1213071B (it) | 1989-12-07 |
| AU596176B2 (en) | 1990-04-26 |
| CS272774B2 (en) | 1991-02-12 |
| ES2065316T3 (es) | 1995-02-16 |
| NO167928C (no) | 1991-12-27 |
| CN87104043A (zh) | 1988-01-13 |
| DD263769A5 (de) | 1989-01-11 |
| AU7247587A (en) | 1987-11-12 |
| KR870011166A (ko) | 1987-12-21 |
| DE3750786T2 (de) | 1995-04-13 |
| CA1339316C (en) | 1997-08-19 |
| ZA873178B (en) | 1987-10-28 |
| RU1807991C (ru) | 1993-04-07 |
| IL82423A0 (en) | 1987-11-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1012175B (zh) | 沿主链含有全氟环氧基而无过基氧的全氟聚醚及其衍生物的制法 | |
| JP3073497B2 (ja) | フッ素とは異なるハロゲンを含有し、酸末端基を有するペルフルオロポリエーテル | |
| CN1089346C (zh) | 含氟弹性体 | |
| RU2144044C1 (ru) | Фторированные производные бисвинилоксиметана (варианты), полимеры и сополимеры на их основе | |
| CN1031064C (zh) | 制备过氧化全氟聚醚的方法 | |
| CN1224620C (zh) | 生产氟化环氧化物的方法和有关聚合物 | |
| EP0232329B1 (en) | Perfluorinated polyether fluids | |
| CA1339864C (en) | Process for preparing controlled molecular weight perfluoropolyethers having perfluoroalkyl or perfluorochloroalkyl end groups | |
| US5539059A (en) | Perfluorinated polyethers | |
| CN86108018A (zh) | 新功能性全氟聚醚及其制备方法 | |
| EP1524287B1 (en) | Perfluoropolyethers | |
| CN1199928C (zh) | 新型含氟化合物及其制造方法和它的聚合物 | |
| US5506309A (en) | Perfluorinates polyethers | |
| CN1088071C (zh) | 制备具有含溴或碘端基全氟聚醚的方法 | |
| KR0164217B1 (ko) | 퍼할로겐화된 폴리에테르 | |
| US5220076A (en) | Perfluoropolyethers and processes for their preparation | |
| US5177226A (en) | Perfluoropolyethers free from peroxidic oxygen and containing perfluoroepoxy groups positioned along the perfluoropolyether chain, and their derivatives | |
| US5672767A (en) | Process for preparing polyether compounds | |
| EP0638103B1 (en) | Preparation of fluorinated polyethers | |
| US5210233A (en) | Cyclic perfluoropolyether |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| C10 | Entry into substantive examination | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| C13 | Decision | ||
| GR02 | Examined patent application | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C15 | Extension of patent right duration from 15 to 20 years for appl. with date before 31.12.1992 and still valid on 11.12.2001 (patent law change 1993) | ||
| OR01 | Other related matters | ||
| C17 | Cessation of patent right | ||
| CX01 | Expiry of patent term |