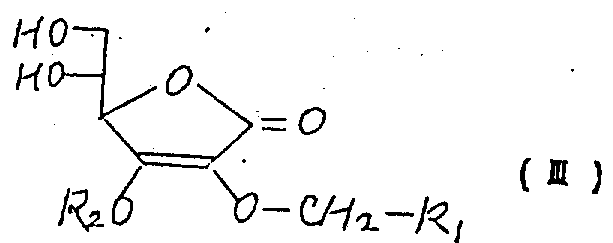CN1009552B - 抗坏血酸衍生物的制备方法 - Google Patents
抗坏血酸衍生物的制备方法Info
- Publication number
- CN1009552B CN1009552B CN85102961.2A CN85102961A CN1009552B CN 1009552 B CN1009552 B CN 1009552B CN 85102961 A CN85102961 A CN 85102961A CN 1009552 B CN1009552 B CN 1009552B
- Authority
- CN
- China
- Prior art keywords
- group
- compound
- reaction
- gram
- substituent
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired
Links
- 238000002360 preparation method Methods 0.000 title claims abstract description 8
- 125000003289 ascorbyl group Chemical class [H]O[C@@]([H])(C([H])([H])O*)[C@@]1([H])OC(=O)C(O*)=C1O* 0.000 title claims abstract 3
- 238000000034 method Methods 0.000 title claims description 19
- 150000001875 compounds Chemical class 0.000 claims abstract description 51
- 125000000217 alkyl group Chemical group 0.000 claims description 23
- 125000003118 aryl group Chemical group 0.000 claims description 23
- 238000006722 reduction reaction Methods 0.000 claims description 8
- 238000006460 hydrolysis reaction Methods 0.000 claims description 6
- 125000001424 substituent group Chemical group 0.000 claims description 6
- 230000007062 hydrolysis Effects 0.000 claims description 4
- 125000003349 3-pyridyl group Chemical group N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 claims description 2
- 125000002252 acyl group Chemical group 0.000 claims description 2
- 125000000051 benzyloxy group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])O* 0.000 claims description 2
- 125000006518 morpholino carbonyl group Chemical group [H]C1([H])OC([H])([H])C([H])([H])N(C(*)=O)C1([H])[H] 0.000 claims description 2
- UIXPTCZPFCVOQF-UHFFFAOYSA-N ubiquinone-0 Chemical compound COC1=C(OC)C(=O)C(C)=CC1=O UIXPTCZPFCVOQF-UHFFFAOYSA-N 0.000 claims 2
- 125000004454 (C1-C6) alkoxycarbonyl group Chemical group 0.000 claims 1
- 125000003358 C2-C20 alkenyl group Chemical group 0.000 claims 1
- 235000013305 food Nutrition 0.000 abstract description 14
- 239000003963 antioxidant agent Substances 0.000 abstract description 5
- 230000003078 antioxidant effect Effects 0.000 abstract description 5
- 235000006708 antioxidants Nutrition 0.000 abstract description 5
- 125000000962 organic group Chemical group 0.000 abstract 1
- 239000002585 base Substances 0.000 description 32
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 30
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 29
- 238000006243 chemical reaction Methods 0.000 description 26
- -1 isobutyl- Chemical group 0.000 description 23
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 17
- 239000000203 mixture Substances 0.000 description 16
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 15
- 239000000243 solution Substances 0.000 description 15
- 229910052799 carbon Inorganic materials 0.000 description 14
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 14
- 150000001721 carbon Chemical group 0.000 description 13
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 10
- 235000019198 oils Nutrition 0.000 description 10
- 230000002829 reductive effect Effects 0.000 description 10
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 9
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 9
- 239000003795 chemical substances by application Substances 0.000 description 8
- 239000012141 concentrate Substances 0.000 description 8
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 8
- 238000001953 recrystallisation Methods 0.000 description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 7
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 6
- 125000003545 alkoxy group Chemical group 0.000 description 6
- 235000010323 ascorbic acid Nutrition 0.000 description 6
- 239000011668 ascorbic acid Substances 0.000 description 6
- 229960005070 ascorbic acid Drugs 0.000 description 6
- WDCDAAMJNUHOIY-UHFFFAOYSA-N ethyl acetate;2-propan-2-yloxypropane Chemical compound CCOC(C)=O.CC(C)OC(C)C WDCDAAMJNUHOIY-UHFFFAOYSA-N 0.000 description 6
- 235000019688 fish Nutrition 0.000 description 6
- 238000009472 formulation Methods 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 239000012046 mixed solvent Substances 0.000 description 6
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Substances [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 6
- 235000015320 potassium carbonate Nutrition 0.000 description 6
- 239000000843 powder Substances 0.000 description 6
- 241000251468 Actinopterygii Species 0.000 description 5
- DHKHKXVYLBGOIT-UHFFFAOYSA-N acetaldehyde Diethyl Acetal Natural products CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 5
- 125000003342 alkenyl group Chemical group 0.000 description 5
- 235000013399 edible fruits Nutrition 0.000 description 5
- 125000001624 naphthyl group Chemical group 0.000 description 5
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- 238000001228 spectrum Methods 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 5
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical compound C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 5
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 4
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 4
- 125000005099 aryl alkyl carbonyl group Chemical group 0.000 description 4
- 125000003710 aryl alkyl group Chemical group 0.000 description 4
- 239000007795 chemical reaction product Substances 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 235000015203 fruit juice Nutrition 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 235000013372 meat Nutrition 0.000 description 4
- 230000003647 oxidation Effects 0.000 description 4
- 238000007254 oxidation reaction Methods 0.000 description 4
- 125000006239 protecting group Chemical group 0.000 description 4
- 230000035484 reaction time Effects 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- 238000005406 washing Methods 0.000 description 4
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 3
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- 239000003377 acid catalyst Substances 0.000 description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 3
- 229910052794 bromium Inorganic materials 0.000 description 3
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 3
- 125000004432 carbon atom Chemical group C* 0.000 description 3
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 3
- 238000010531 catalytic reduction reaction Methods 0.000 description 3
- 229910052801 chlorine Inorganic materials 0.000 description 3
- 239000000460 chlorine Substances 0.000 description 3
- HHEAADYXPMHMCT-UHFFFAOYSA-N dpph Chemical compound [O-][N+](=O)C1=CC([N+](=O)[O-])=CC([N+]([O-])=O)=C1[N]N(C=1C=CC=CC=1)C1=CC=CC=C1 HHEAADYXPMHMCT-UHFFFAOYSA-N 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 125000002541 furyl group Chemical group 0.000 description 3
- 229910052736 halogen Inorganic materials 0.000 description 3
- 150000002367 halogens Chemical class 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 239000011541 reaction mixture Substances 0.000 description 3
- 235000015067 sauces Nutrition 0.000 description 3
- 239000000741 silica gel Substances 0.000 description 3
- 229910002027 silica gel Inorganic materials 0.000 description 3
- 125000001544 thienyl group Chemical group 0.000 description 3
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 2
- ADOBXTDBFNCOBN-UHFFFAOYSA-N 1-heptadecene Chemical compound CCCCCCCCCCCCCCCC=C ADOBXTDBFNCOBN-UHFFFAOYSA-N 0.000 description 2
- JRZJOMJEPLMPRA-UHFFFAOYSA-N 1-nonene Chemical compound CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 2
- HFDVRLIODXPAHB-UHFFFAOYSA-N 1-tetradecene Chemical compound CCCCCCCCCCCCC=C HFDVRLIODXPAHB-UHFFFAOYSA-N 0.000 description 2
- CXDHJGCWMIOAQP-UHFFFAOYSA-N 2-pyridin-3-yl-1,4,5,6-tetrahydropyrimidine;hydrochloride Chemical compound Cl.C1CCNC(C=2C=NC=CC=2)=N1 CXDHJGCWMIOAQP-UHFFFAOYSA-N 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- QWOJMRHUQHTCJG-UHFFFAOYSA-N CC([CH2-])=O Chemical compound CC([CH2-])=O QWOJMRHUQHTCJG-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- XJUZRXYOEPSWMB-UHFFFAOYSA-N Chloromethyl methyl ether Chemical compound COCCl XJUZRXYOEPSWMB-UHFFFAOYSA-N 0.000 description 2
- 239000004375 Dextrin Substances 0.000 description 2
- 229920001353 Dextrin Polymers 0.000 description 2
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- DPDMMXDBJGCCQC-UHFFFAOYSA-N [Na].[Cl] Chemical compound [Na].[Cl] DPDMMXDBJGCCQC-UHFFFAOYSA-N 0.000 description 2
- 239000003513 alkali Substances 0.000 description 2
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 2
- 230000003064 anti-oxidating effect Effects 0.000 description 2
- 125000004104 aryloxy group Chemical group 0.000 description 2
- 125000004152 benzoquinonyl group Chemical group C1(C(=CC(C=C1)=O)*)=O 0.000 description 2
- 239000012267 brine Substances 0.000 description 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 2
- 229940061627 chloromethyl methyl ether Drugs 0.000 description 2
- BGTOWKSIORTVQH-UHFFFAOYSA-N cyclopentanone Chemical compound O=C1CCCC1 BGTOWKSIORTVQH-UHFFFAOYSA-N 0.000 description 2
- 235000019425 dextrin Nutrition 0.000 description 2
- 239000003480 eluent Substances 0.000 description 2
- 238000007429 general method Methods 0.000 description 2
- GNOIPBMMFNIUFM-UHFFFAOYSA-N hexamethylphosphoric triamide Chemical compound CN(C)P(=O)(N(C)C)N(C)C GNOIPBMMFNIUFM-UHFFFAOYSA-N 0.000 description 2
- 125000004435 hydrogen atom Chemical class [H]* 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- UKVIEHSSVKSQBA-UHFFFAOYSA-N methane;palladium Chemical compound C.[Pd] UKVIEHSSVKSQBA-UHFFFAOYSA-N 0.000 description 2
- HZVOZRGWRWCICA-UHFFFAOYSA-N methanediyl Chemical compound [CH2] HZVOZRGWRWCICA-UHFFFAOYSA-N 0.000 description 2
- 235000010755 mineral Nutrition 0.000 description 2
- 239000011707 mineral Substances 0.000 description 2
- 150000002978 peroxides Chemical class 0.000 description 2
- 230000001603 reducing effect Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 235000017550 sodium carbonate Nutrition 0.000 description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 description 2
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 2
- 235000013599 spices Nutrition 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 235000013311 vegetables Nutrition 0.000 description 2
- 239000011709 vitamin E Substances 0.000 description 2
- 235000019165 vitamin E Nutrition 0.000 description 2
- SMDGVPQREIZILS-UHFFFAOYSA-N $l^{1}-oxidanylmethylbenzene Chemical compound [O]CC1=CC=CC=C1 SMDGVPQREIZILS-UHFFFAOYSA-N 0.000 description 1
- MIOPJNTWMNEORI-GMSGAONNSA-N (S)-camphorsulfonic acid Chemical compound C1C[C@@]2(CS(O)(=O)=O)C(=O)C[C@@H]1C2(C)C MIOPJNTWMNEORI-GMSGAONNSA-N 0.000 description 1
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 1
- FRASJONUBLZVQX-UHFFFAOYSA-N 1,4-naphthoquinone Chemical class C1=CC=C2C(=O)C=CC(=O)C2=C1 FRASJONUBLZVQX-UHFFFAOYSA-N 0.000 description 1
- VQOXUMQBYILCKR-UHFFFAOYSA-N 1-Tridecene Chemical group CCCCCCCCCCCC=C VQOXUMQBYILCKR-UHFFFAOYSA-N 0.000 description 1
- QSSXJPIWXQTSIX-UHFFFAOYSA-N 1-bromo-2-methylbenzene Chemical compound CC1=CC=CC=C1Br QSSXJPIWXQTSIX-UHFFFAOYSA-N 0.000 description 1
- GQEZCXVZFLOKMC-UHFFFAOYSA-N 1-hexadecene Chemical compound CCCCCCCCCCCCCCC=C GQEZCXVZFLOKMC-UHFFFAOYSA-N 0.000 description 1
- AIDLAEPHWROGFI-UHFFFAOYSA-N 2-methylbenzene-1,3-dicarboxylic acid Chemical compound CC1=C(C(O)=O)C=CC=C1C(O)=O AIDLAEPHWROGFI-UHFFFAOYSA-N 0.000 description 1
- PNWSHHILERSSLF-UHFFFAOYSA-N 4-methylbenzene-1,3-dicarboxylic acid Chemical compound CC1=CC=C(C(O)=O)C=C1C(O)=O PNWSHHILERSSLF-UHFFFAOYSA-N 0.000 description 1
- CMXCKHYWEJGQIU-UHFFFAOYSA-N CCCCCCCCCCCCCCCCCC[S] Chemical compound CCCCCCCCCCCCCCCCCC[S] CMXCKHYWEJGQIU-UHFFFAOYSA-N 0.000 description 1
- QPQMMFKANPOVIT-UHFFFAOYSA-N COC1C(C(=O)O)=CC(=CC1(C(=O)O)OC)C Chemical compound COC1C(C(=O)O)=CC(=CC1(C(=O)O)OC)C QPQMMFKANPOVIT-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 241000555825 Clupeidae Species 0.000 description 1
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 150000000996 L-ascorbic acids Chemical class 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- UWHZIFQPPBDJPM-FPLPWBNLSA-M Vaccenic acid Natural products CCCCCC\C=C/CCCCCCCCCC([O-])=O UWHZIFQPPBDJPM-FPLPWBNLSA-M 0.000 description 1
- 235000021322 Vaccenic acid Nutrition 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- YKIOKAURTKXMSB-UHFFFAOYSA-N adams's catalyst Chemical compound O=[Pt]=O YKIOKAURTKXMSB-UHFFFAOYSA-N 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- 230000031709 bromination Effects 0.000 description 1
- 238000005893 bromination reaction Methods 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 229920001525 carrageenan Polymers 0.000 description 1
- 235000010418 carrageenan Nutrition 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 239000007810 chemical reaction solvent Substances 0.000 description 1
- XTEGARKTQYYJKE-UHFFFAOYSA-N chloric acid Chemical compound OCl(=O)=O XTEGARKTQYYJKE-UHFFFAOYSA-N 0.000 description 1
- 229940005991 chloric acid Drugs 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 125000005066 dodecenyl group Chemical group C(=CCCCCCCCCCC)* 0.000 description 1
- 239000003657 drainage water Substances 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000005562 fading Methods 0.000 description 1
- 235000019197 fats Nutrition 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 235000019261 food antioxidant Nutrition 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 125000006038 hexenyl group Chemical group 0.000 description 1
- 125000000717 hydrazino group Chemical group [H]N([*])N([H])[H] 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 125000000687 hydroquinonyl group Chemical class C1(O)=C(C=C(O)C=C1)* 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 235000020997 lean meat Nutrition 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 235000013622 meat product Nutrition 0.000 description 1
- 239000000155 melt Substances 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 1
- AFFLGGQVNFXPEV-UHFFFAOYSA-N n-decene Natural products CCCCCCCCC=C AFFLGGQVNFXPEV-UHFFFAOYSA-N 0.000 description 1
- 238000006386 neutralization reaction Methods 0.000 description 1
- 125000004365 octenyl group Chemical group C(=CCCCCCC)* 0.000 description 1
- 125000004043 oxo group Chemical group O=* 0.000 description 1
- CFHIDWOYWUOIHU-UHFFFAOYSA-N oxomethyl Chemical compound O=[CH] CFHIDWOYWUOIHU-UHFFFAOYSA-N 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 235000011837 pasties Nutrition 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- PTMHPRAIXMAOOB-UHFFFAOYSA-L phosphoramidate Chemical compound NP([O-])([O-])=O PTMHPRAIXMAOOB-UHFFFAOYSA-L 0.000 description 1
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 1
- 231100000614 poison Toxicity 0.000 description 1
- 230000007096 poisonous effect Effects 0.000 description 1
- 229920005990 polystyrene resin Polymers 0.000 description 1
- 235000015277 pork Nutrition 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 239000012088 reference solution Substances 0.000 description 1
- 238000005096 rolling process Methods 0.000 description 1
- 235000019512 sardine Nutrition 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 235000015170 shellfish Nutrition 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- APSBXTVYXVQYAB-UHFFFAOYSA-M sodium docusate Chemical compound [Na+].CCCCC(CC)COC(=O)CC(S([O-])(=O)=O)C(=O)OCC(CC)CCCC APSBXTVYXVQYAB-UHFFFAOYSA-M 0.000 description 1
- 235000019830 sodium polyphosphate Nutrition 0.000 description 1
- 230000003595 spectral effect Effects 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000004575 stone Substances 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 229940095068 tetradecene Drugs 0.000 description 1
- YXFVVABEGXRONW-UHFFFAOYSA-N toluene Substances CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- UWHZIFQPPBDJPM-BQYQJAHWSA-N trans-vaccenic acid Chemical compound CCCCCC\C=C\CCCCCCCCCC(O)=O UWHZIFQPPBDJPM-BQYQJAHWSA-N 0.000 description 1
- 125000005065 undecenyl group Chemical group C(=CCCCCCCCCC)* 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 210000001835 viscera Anatomy 0.000 description 1
- 235000019154 vitamin C Nutrition 0.000 description 1
- 239000011718 vitamin C Substances 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 239000003643 water by type Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23B—PRESERVATION OF FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES; CHEMICAL RIPENING OF FRUIT OR VEGETABLES
- A23B2/00—Preservation of foods or foodstuffs, in general
- A23B2/70—Preservation of foods or foodstuffs, in general by treatment with chemicals
- A23B2/725—Preservation of foods or foodstuffs, in general by treatment with chemicals in the form of liquids or solids
- A23B2/729—Organic compounds; Microorganisms; Enzymes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/02—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings
- C07D307/34—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D307/56—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D307/62—Three oxygen atoms, e.g. ascorbic acid
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D407/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00
- C07D407/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00 containing two hetero rings
- C07D407/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Polymers & Plastics (AREA)
- Food Science & Technology (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Plural Heterocyclic Compounds (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Anti-Oxidant Or Stabilizer Compositions (AREA)
- Food Preservation Except Freezing, Refrigeration, And Drying (AREA)
- Furan Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Fats And Perfumes (AREA)
Abstract
制备具有下列分子式的抗坏血酸衍生物其中R1是分子量从58到400的有机基,该衍生物是一个新型化合物,可用作食品的抗氧化剂。
Description
本发明关系到一种抗坏血酸衍生物及其生产和使用。
抗坏血酸具有防止氧化的活力,用于食品中,目的在于防止枯黄,保留香味,保持新鲜等等。
但是,抗坏血酸容易降解,所以要求在较长时斯内能起到上述作用的情况下就会遇到困难。
在此情况下,本发明人抱着以获得一种不太容易降解,而且仍具有十足防止氧化活力的抗坏血酸衍生物的特定目的进行了广泛研究,结果发现,用一个比较大分子量的基团来改变抗坏血酸的第二位置,则可产生能够达到预期目的的化合物,对此发现又进行了深入地研究,最后取得本发明。
本发明的目标是:
(1)具有下列分子式的一种抗坏血酸衍生物
其中R1是一个分子量从58到400的有机基,
(2)一个生产抗坏血酸衍生物(Ⅰ)的方法,包括将下列分子式化合物(Ⅲ)的5,6-缩醛或缩酮衍生物(Ⅱ)水解和/或还原而成
其中R1的定义如前,R2是一个可以用水解或还原作用除去的基,以及
(3)含有抗坏血酸衍生物食品的抗氧化剂制剂。
上面的分子式中,用R1来表示的分子量从58到400的有机基包括,例如,直链或支链的烷基基团,带有取代基或多取代基的直链或支链烷基基团,可以带有取代基或多取代基的链烯基基团,可以带有取代基或多取代基的芳香基基团,可以带有取代基或多取代基的芳烷基基团,可以带有取代基或多取代基的酰基基团,可以带有取代基或多取代基的芳氧基基团,以及可以带有取代基或多取代基的芳烷氧基基团。
上面分子量从58到400的直链或支链烷基基团,以具有5到22个碳原子为宜,最好是具有9到20个碳原子。
这些烷基包括诸如,正-戊基,正-己基,正-庚基,正-辛基,正-壬基,正-癸基,正-十一烷基,正-十二烷基,正-十三烷基,正-十四烷基,
正-十五烷基,正-十六烷基,正-十七烷基,正-十八烷基,正-十九烷基,正-二十烷基,正-二十一烷基,以及正-二十二烷基。
上面分子量从58到400的带有取代基或多取代基的直链或支链烷基基团中的烷基,最好是具有1到10个碳原子。
这些烷基包括诸如,甲基,乙基,正-丙基,异丙基,正-丁基,异丁基,仲丁基,叔丁基,正-戊基,正-己基,正-庚基,正-辛基,正-壬基,和正-癸基。
其取代其包括诸如,低级的烷氧羰基基团,可以带有取代基或多取代基的芳香基基团,可以带有取代基或多取代基的芳烷基基团。可以带有取代基或多取代基的芳香羰氧基基团,可以带有取代基或多取代基的芳烷羰氧基基团,2,3,5-三甲基-1,4-对醌基基团,2,3-二甲氧基-5-甲基-1,4-对醌基基团以及2-甲基-1,4-萘醌基基团。
上述的低级烷氧羰基基团中的烷氧基,最好是具有1至6个碳原子,这些烷氧基包括诸如,甲氧基,乙氧基,正-丙氧基,正-丁氧基,正-戊氧基以及正-己氧基。
上述可以带有取代基或多取代基的芳香基基团和上述可以带有取代基或多取代基的芳香羰氧基基团中的芳香基基团包括诸如,苯基,萘基,噻吩基和呋喃基。在上述可以带有取代基或多取代基的芳烷基基团以及可以带有取代基或多取代基的芳烷羰氧基基团中的芳烷基基团包括诸如,苄基和苯乙基。此等芳香基基团或芳烷基基团的取代基包括诸如,1至5个碳原子的低级烷基(例如,甲基,乙基,正-丙基,异-丙基,正-丁基,仲-丁基,叔-丁基,正-戊基等),1至3个碳原子的低级烷氧基(例如,甲氧基,乙氧基,丙氧基等),卤素(例如,氯,溴,碘,氟等)硝基,氨基,氧代,羟基和苄氧基。
上述分子量从58至400,可以带有取代基或多取代基的链烯基基团中的链烯基,最好是具有2至20个碳原子,此等链烯基基团包括诸如,乙烯基、丙烯基、丁烯基、戊烯基、己烯基、庚烯基、辛烯基、壬烯基、癸烯基、十一碳烯基、十二碳烯基、十三碳烯基、十四碳烯基、十五碳烯基、十六碳烯基、十七碳烯基、十八碳烯基、十九碳烯基和二十碳烯基,此等链烯基基团上的取代基包括诸如芳香基基团(例如,苯基、萘基等),芳烷基(例如,苄基、苯乙基等)和5或6元杂环基(例如,3-吡啶基、噻吩基、呋喃基等)。
上述分子量从58至400可以带有取代基或多取代基的多种芳香基基团中的芳香基基团包括诸如,苯基和萘基。此等芳香基基团上的取代基包括诸如,1至5个碳原子的烷基(例如,甲基、乙基、正-丙基、异-丙基、正-丁基、异丁基、仲丁基、叔丁基、正-戊基等),和甲氧基,亚甲二氧基和羟基基团。
上面所述,分子量从58至400,可以带有取代基或多取代基的多种芳烷基基团中的芳烷基基团包括诸如,苄基和苯乙基,此等芳烷基基团上的取代基包括诸如,1至5个碳原子的烷基基团(例如,甲基、乙基、正-丙基、异丙基,正-丁基、异-丁基、仲-丁基、叔-丁基、正-戊基等),和甲氧基,亚甲二氧基和羟基基团。
上面所述,分子量从58至400,可以带有取代基或多取代基的酰基基团中的酰基,最好是具有1至9个碳原子,此等酰基基团包括诸如,甲酰基、乙酰基、丙酰基、丁酰基、异丁酰基、苯甲酰基、吗啉代羰基、C1-3烷氧基-羰基吡咯烷羰基(例如,2-甲氧羰基吡咯烷羰基,2-丙氧羰基吡咯烷羰基等),C1-3烷氧基-羰基(例如,甲氧羰基、乙氧羰基、丙氧羰基等),可以带有取代基或多取代基的芳香羰基和可以带有取代基或多取代基的芳烷羰基。
上面所述,可以带有取代基或多取代基的芳香羰基中的芳香基包括诸如,苯基、萘基。上述可以带有取代基或多取代基的芳烷羰基中的芳烷基包括诸如,苄基或苯乙基。此等芳香基或芳烷基上的取代基包括诸如,羟基、1至5个碳原子的低级烷基(例如,甲基、乙基、正-丙基、异丙基、正-丁基、异丁基、叔-丁基、正-戊基等)和1至3个碳原子的低级烷氧基(例如甲氧基、乙氧基、丙氧基等)。
上述分子量从58至400,可以带有取代基或多取代基的芳氧基基团中的芳香基包括诸如,苯基、萘基、噻吩基和呋喃基,其取代基包括诸如,羟基基团,1至5个碳原子的低级烷基(例如,甲基、乙基、正-丙基、异丙基、正-丁基、仲-丁基、叔-丁基、正-戊基等)和1至3个碳原子的低
级烷氧基(例如,甲氧基、乙氧基、丙氧基等)。
上述分子量从58至400,可以带有取代基或多取代基的芳烷氧基基团中的芳烷基包括诸如,苄基和苯乙基,其取代基包括诸如,羟基基团、1至5个碳原子的低级烷基(例如,甲基、乙基、正-丙基、异丙基、正-丁基、仲-丁基、叔-丁基、正-戊基等)和1至3个碳原子的低级烷氧基(例如,甲氧基、乙氧基、丙氧基等)。
上述取代基的数量最好是1至3个。
上述分子式中,用R2表示的可用水解或还原作用除去的基团包括诸如,甲氧甲基、苄氧甲基、2-四氢吡喃基、三甲基甲硅烷基、二甲基-叔丁基甲硅烷基、苄基和对-甲氧苄基。
上述乙缩醛包括例如具有下列化学式的基:
(其中R3是氢、甲基、苄基、对-甲氧苄基等)缩酮包括例如下列化学式的基:
(其中R4和R5可以相同也可以不同,每一个表示氢原子,甲基或乙基或者是R4和R5的联合形式-(CH2)a-其中a是4或5)。
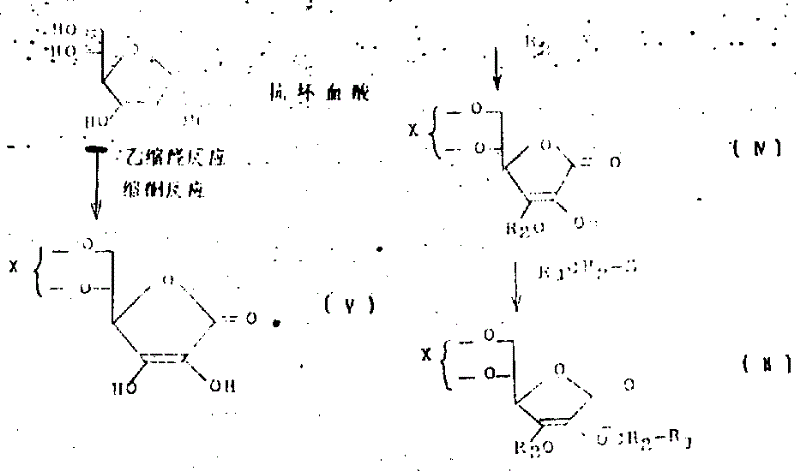
本发明中的化合物(Ⅰ)是把化合物(Ⅱ)进行水解反应和(或)还原反应来生产的。
在化合物(Ⅱ)中,处于第三位置上的保护基R2若是一个可用水解反应除掉的基(例如,甲氧基甲基、苄氧甲基、三甲基甲硅烷基、二甲基-叔-丁基甲硅烷基),则对化合物(Ⅱ)进行水解反应时,除掉处于第三位置上的保护基以后,就可以连续实现除去第5和第6位置上的乙缩醛和缩酮,从而得到化合物(Ⅰ)。
在化合物(Ⅱ)中,处于第三位置上的保护基R2若是可以用还原反应去掉的基(例如,2-四氢吡喃基,苄基,对-甲氧苄基)当把化合物(Ⅱ)进行水解反应时,处于第五和第六位置上的乙缩醛或缩酮基团首先去掉,生成化合物(Ⅲ),然后通过还原反应除去保护基,生成化合物(Ⅰ)。
上述水解反应是将起始化合物与酸催化剂接触下进行的。该酸催化剂包括诸如,盐酸、硫酸、对甲苯磺酸、过氯酸和乙酸。反应是在含水溶剂中进行的,该溶剂包括诸如,甲醇、乙醇、二噁烷、1,2-甲氧基乙烷和四氢呋喃。反应温度范围选自0℃至80℃,反应时间大约10分钟至3小时。
上述还原反应包括例如催化还原。所谓催化还原就是把起始化合物在与催化接触下进行。此种催化剂包括诸如,钯、钯-碳、铂黑和二氧化铂。反应一般在溶剂中进行,此等溶剂包括诸如,甲醇、乙醇、乙酸和乙酸乙酯。反应温度大约10至40℃,反应时间约1至18小时。
如此生产出的抗坏血酸衍生物(Ⅰ)可以用一般的分离和净化方法进行分离和收集(例如用色谱柱法,使用聚苯乙烯树脂,活性碳,逆相系统等,重结晶等)。
本发明过程中,用作原材料的化合物(Ⅱ),可以用下列反应步骤进行生产。
上述分子式中,X是乙缩醛或缩酮基,上述反应包括抗坏血酸的乙缩醛反应或缩酮反应,以生产化合物(Ⅴ)。该反应是用抗坏血酸与酮或醛诸如丙酮、苯甲醛、环戊酮和环己酮进行反应的。所用的反应溶剂可以是四氢呋喃、氯仿、二乙基醚、二氯甲烷和二氯乙烷。反应温度为室温至60℃,反应在酸催化剂下进行。催化剂包括诸如硫酸,对-甲基苯磺酸和樟脑磺酸。反应时间为4至24小时。
其后,化合物(Ⅴ)与化学式R2-Y〔其中R2的定义如前所述;Y是卤素(例如,氯、溴等)〕的化合物(例如,氯甲基甲醚、苄基氧、苄基溴等)在二甲基甲酰胺、二甲亚砚、六甲基磷酰胺或四氢呋喃中,或是单独的,或是呈混合溶剂进行反应,反应在无机碱诸如碳酸钾、碳酸钠、氢氧化钠、氢氧化钾和碳酸氢钠作用下生成化合物(Ⅳ)。反应温度为0-40℃(最好25℃),反应时间要1至18小时。其后,反应生成的化合物(Ⅳ),再与化学式
R1-CH2-Z的化合物〔其中R1的定义如前;Z为卤素(例如氯、溴等)〕反应。反应是在诸如二甲基甲酰胺、二甲亚砜、六甲基磷酰胺和四氢呋喃溶剂,单独的或呈混合溶剂中进行,并在无机碱(例如氢氧化钠、氢氧化钾、碳酸钠、碳酸钾等)作用下,温度10-60℃,通过1至18小时生成化合物(Ⅰ)。
按照本发明的过程所生产的化合物(Ⅱ)或(Ⅲ),用作生产化合物(Ⅰ)时的一个合成中间体。
当把具有抑制氧化活力的化合物(Ⅰ)给老鼠作口试试验时,其LD50值为0.8-10克/公斤,所以是属低毒物,可以用作食品的抗氧化剂。
把化合物(Ⅰ)用作食品抗氧化剂时,就要把它制作成含有化合物(Ⅰ)食品的抗氧化剂制剂,然后加到或接触到食品上去。
在加工或按方配制成例如化合物(Ⅰ)的制剂时,可以用一般方法不加稀释剂制成或是加以稀释后制成。
稀释的方法,例如把载体(例如葡萄糖、淀粉等)与化合物(Ⅰ)混合,再用一般方法把混合物作成粉或是细颗粒。
按照本发明的抗氧化剂制剂所加入到的食品包括诸如水果、果汁、果品类、食用肉、鱼和水生贝壳类动物,和油和脂(例如凉拌菜用油、猪油等)。
按照本发明的食品抗氧化剂制剂的使用方法是,当化合物(Ⅰ)-活性成分,溶于水中时,举其一例,该法是按照本发明把食品抗氧化剂制剂粉熔化,直接加到诸如果汁、果品类和食用肉类制品等食品上去。另一方法是把按照本发明的食品抗氧化剂制剂事先先溶于水中,然后把该溶液加到果汁、果品类或食用肉制品上去,或是把鱼和水生贝壳类浸渍在上述溶液中。
按照本发明,食品抗氧化剂制剂在将之用于食用肉类制品和鱼及水生贝壳类的情况下,可以用一种乳化剂将之制成水质乳化液再进行使用。
当化合物(Ⅰ)一活性组分,溶于油中时,按照本发明的食品抗氧化剂制剂则可直接加到油或脂中去。
按照本发明,食品抗氧化剂制剂的用量,当使用化合物(Ⅰ)时,对果汁和水果约为0.20-0.04%(重量%),对食用肉类制品约为0.02-0.08%(重量%),对于将之混合于鱼类和水生贝壳类时约为0.02-0.08%(重量%),对于制备成浸渍液的溶液浓度约为0.1-1%(重量%),对于油和脂类约为0.002-0.02%(重量%)。
此外,化合物(Ⅰ)还可以用作化妆品的致白美容剂。化合物(Ⅰ)在化妆品的致白美容剂中的用量,例如在洗涤剂中大约为0.1-1%(重量/体积),在雪花膏中大约为0.1-1%(重量%)。
为说明本发明,下面将逐一叙述实验实例,参考实例和实例。
实验1
用一个稳定的官能团来测定抑制氧化的活性。
按照Brois MS的方法[Nature,181,1199,1958],测定了α,α-二苯基-β-苦基肼基(DPPH)的还原活性,将之用作评定抑制氧化活性的尺度。如此则将试验的化合物〔例如,化合物(Ⅰ),其中R1=-(CH2)16CH3〕加到3毫升的0.1毫摩尔DPPH乙醇溶液中,过20分钟以后,用光谱分光光度计在517毫微米波长下测定其吸收系数,其间与参考溶液吸收系数的差距(不大于0.5%二甲基甲酰胺)即作为还原活性。
实验结果见图1。
图1中,-●-代表上述化合物的实验结果,-0-代表维生素E的结果,和-▲-代表维生素C的结果。
结果证实上述试验用的化合物还原DPPH的浓度按照用量不小于10-5摩尔。维生素C和E所显示的活性与试验用的化合物相同。
实验2
把除去内脏和鱼骨后的沙丁鱼绞碎,绞碎的肉一部分不加入化合物(Ⅰ),另一部分加入化合物(Ⅰ),其中R1=(CH2)4CH3,加入量为0.03%和0.05%,并制成鱼丸子。将鱼丸子在-20℃下冰冻保存,在不同的保存时间内测定其过氧化物值(POV)。结果示于表1。
实验3
在一个直径10厘米的石头盘子中,放入25克凉拌菜用油,在60℃下保存于恒温箱中,一部分不加入化合物(Ⅰ),另一部分加入融化的化合物(Ⅰ),其中R1=(CH2)16CH3,加入量为0.03%和0.05%,在不同的保存时间内测定其过氧化物值(POV),结果示于表2。
参考实例1
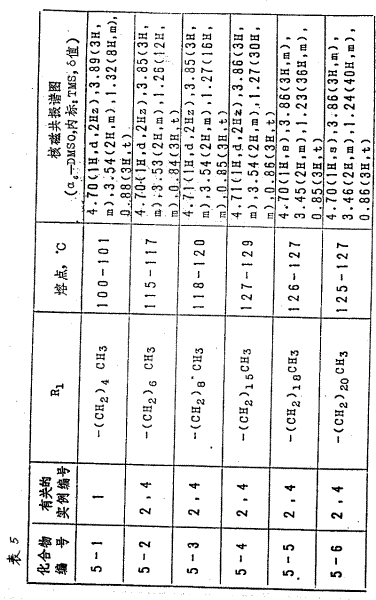
(1)将L-抗坏血酸丙酮化合物(21.6克,0.1摩尔)溶解于二甲基甲酰胺(210毫升)中,溶液用冰冷却。向该溶液中加入碳酸钾(14克,0.1摩尔),继之加入溴化苄基(11.2毫升),在室温下搅拌20小时。反应终结后,向反应溶液中加入水(100毫升),将混合物用2N的盐酸中和,调整到pH5.0,然后用两批乙酸乙酯抽提。有机液层用水洗后进行干燥(用硫酸镁),再在减压下浓缩。将产品用硅胶色谱柱处理,用异丙醚-乙酸乙酯进行洗脱(3∶1)。洗脱液经过浓缩,之后将残留物从异丙醚-乙酸乙酯中重结晶,得到L-5、6-0、0-异丙叉-3-0-苄基抗坏血酸(13克,40%),熔点为105-106℃。
(2)将L-5、6-0、0-异丙叉-3-0-苄基抗坏血酸(3.06克,0.01摩尔)溶解于二甲亚砜(20毫升)和四氢呋喃(15毫升)混合溶剂中,并向溶液中加入碳酸钾(1.5克,0.011摩尔)。然后向混合物中加入十八烷基碘(3.83克),于室温下搅拌18小时。反应结束后,向反应混合物中加入水(100毫升),继之用乙酸乙酯抽提。有机液层用水洗涤、干燥(用硫酸镁)后,于真空下浓缩。将残留物用硅胶色谱栓处理,并用异丙醚-乙酸乙酯洗脱(10∶1),如此得到L-5、6-0、0-异丙叉-3-0-苄基-2-0-十八烷基抗坏血酸(3.8克)。
熔点:44-45℃
核磁共振谱线
〔CDCL3,内标:TMS(四甲基硅烷),δ值〕
7.29(5H,S),5.43(2H,S),4.51(1H,d,2赫兹)。4.08(3H,m),1.38(6H,S),0.88(3H,t)
参考实例2
(1)将L-抗坏血酸丙酮化合物(42克,0.19摩尔)溶于二甲基甲酰胺(100毫升)和六甲基氨基磷酸酯(100毫升)的混合溶剂中,并向溶液中加入碳酸钾(32克、0.23摩尔)用冰冷却。向混合物中历经20分钟逐滴加入氯甲基甲醚(18克,0.22摩尔)在四氢呋喃(25毫升)中的溶液。在室温下搅拌2.5小时以后,向反应混合物中加入水(100毫升),并加入2N的盐酸,调整到pH5.0,继之用乙酸乙酯分四批抽提。有机液层经水洗干燥后,在减压下浓缩,残留物有硅胶色谱柱处理,并用异丙醚-乙酸乙酯洗脱(2∶1)。洗脱液经浓缩,残留物从同一溶剂系统中重结晶,从而得到L-5、6-0、0-异丙叉-3-0-甲基甲基抗坏血酸(46克)。
熔点:93-94℃
对C11H16O7的元素分析
求出值:C,50.84;H,6.05
计算值:C,50.77;H,6.20
(2)将L-5、6-0、0-异丙叉-3-0-甲氧甲基抗坏血酸(1.84克,7.1毫摩尔)溶解于二甲亚砜(10毫升)和十八烷基磺(10毫升)中,并向溶液中加入碳酸钾(1.0克),使反应在60℃下进行6小时。反应终止后,向反应混合物中加入水(50毫升),再用乙酸乙酯抽提反应产物。有机液层经用水洗、干燥并在减压下浓缩,残留物用硅胶色谱柱处理,用异丙醚洗脱,洗脱液经浓缩,残留物从异丙醚-乙酸乙酯中重结晶,从而得到L-5、6-0、0-异丙叉-3-0-甲氧甲基-2-0十八烷基抗坏血酸(0.8克)。
熔点:50至52℃
核磁共振谱图
(CDCl3,内标:TMS,δ值)
5.42(2H,S),4.53(1H,d,2赫兹),4.10(5H,m),3.51(3H,S),1.38(3H,S),1.36(3H,S),1.27(32H,m),0.88(3H,t)
参考实例3
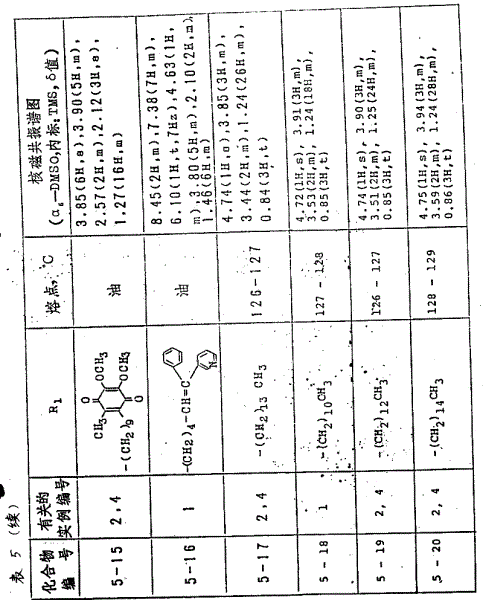
用参考实例1或2的同一方法,得到的化合物(以分子式(Ⅱ)为代表,其中R1和R2值逐一列于表3中)见表3。
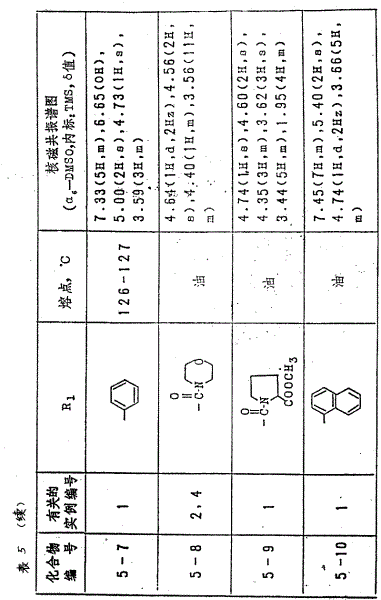
实例1
将L-5,6-0,0-异丙叉-3-0-甲氧甲基-2-0-十八烷基抗坏血酸(1.2克)溶于甲醇(30毫升)和四氢呋喃(10毫升)混合溶剂中,并向溶液中加入2N的盐酸(10毫升),继之于50℃下搅拌6小时。反应溶液在减压下浓缩,反应产物用乙酸乙酯抽提。有机液层经水洗,干燥(用硫酸镁),再在减压下浓缩,所得残留物从异丙醚-乙酸乙酯中重结晶,从而得到2-0-十八烷基抗坏血酸(0.82克)
熔点:127-128℃
核磁共振谱图
(CDCl3,内标:TMS,δ值)
4.69(1H,d),3.88(3H,m),3.53(2H,m),1.25(32H,m),0.87(3H,t)。
实例2
将L-5,6-0,0-异丙叉-3-0-苄基-2-0十八烷基抗坏血酸(3.8克)溶于四氢呋喃(40毫升)和
甲醇(10毫升)混合溶剂中,向溶液中加入2N的盐酸(20毫升),于50℃下搅拌24小时。反应终结以后,反应溶液在减压下浓缩,反应产物用乙酸乙酯抽提。有机液层经水洗,干燥后,在减压下浓缩,所得残余物从异丙醚-乙酸乙酯中重结晶,从而得到3-0-苄基-2-0-十八烷基抗坏血酸(2.6克)。
熔点:75-76℃
核磁共振谱图。
(CDCl3,内标:TMS,δ值)
7.32(5H,S),5.45(2H,S),4.66(1H,d),4.00(5H,m),1.26(32H,m),0.87(3H,t)
实例3
用实例2中的同一方法制得的化合物如表4所示,表中各实例表示前面所述的分子式(Ⅲ)的结构式。
实例4
将3-0-苄基-2-0-十八烷基抗坏血酸(2.1克)溶于乙酸乙酯(25毫升)中,向溶液中加入5%钯-碳(0.5克),在常压下进行催化还原。然后过滤出催化剂,滤液在减压下浓缩。反应产物从异丙醚-乙酸乙酯中重结晶,从而得到2-0-十八烷基抗坏血酸(1.5克)
熔点:127-128℃
核磁共振谱图(d6-DMSO):
4.69(1H,d),3.88(3H,m),3.53(2H,m),1.25(32H,m),0.87(3H,t)
实例5
用实例1或4中的同一方法,得到表5中的各化合物。表中所得到的各化合物,表示前面所述分子式(Ⅰ)的结构式。
从4到15的各化合物,是采用了实例2和3的同一方法,同时把所得的氢醌衍生物用氯化铁进行了氧化。
实例6
在一个微速混合器(日本Takara Koki公司制造)中,用大约2分钟,混合在实例5-12中得到的1.5公斤分子式(Ⅰ)的化合物,其中R1=-COOCH3,和1.5公斤角叉胶,以及1.5公斤糊精,把混合物制成细粉。
把鲸鱼油10秒钟内浸入到上述15克细粉的1立升水溶液中,排出水后,于-20℃下保存6个月。将此产品与未经处理的对照部分进行比较,证实臭味小,而且具有较好的颜色与光泽。
实例7
在一个微速混合器(日本Takara Koki公司制造)中,用大约2分钟时间,混合3公斤在实例5-12中得到的分子式(Ⅰ)的化合物,其中R1=COOCH3,和2公斤糊精,制成细粉。
浸渍盐水的制备是,向8.2公斤冰冷水中加入900克氯化钠,3克亚硫酸钠,150克多磷酸钠,50克L-谷氨酸钠,120克蔗糖,300克精白面粉(Powdered White)以及30克香料(核对部分)或30克上述细粉(本发明部分)。用浸渍盐水各300克,分别注射到1公斤食用的后腿肉中,经过翻滚18个小时后,再把火腿放到空气能够渗透的盒子中,于60℃下干燥30分钟,60℃下烟熏30分钟,再在75℃下用蒸汽沸腾60分钟,从而制得烤火腿。
该产品在5℃下保存过夜,切片,置于有散射光线的屋中3小时,然后检查其外观,本发明部分的外观呈红色,并减轻了褪色反应,比对照的部分要好。
实例8
在混合器中,用实例1方法得到的700克分子式(Ⅰ)的化合物,其中R1=-(CH2)16CH3,与300克棉子凉拌用油相混,制成溶液。
再将700克瘦肉,300克猪油,10克氯化钠,5克L-谷酸钠以及3克香料进行混合,把混合物按每份100克分开制成猪肉馅饼。另一部分把上述混合物混以0.8克上述油溶液,用同样办法制成肉馅饼。经过-20℃冰冻保存3个月后,加入油溶液的部分,比对照部分臭味小。
表1
保存的天数 0 30 60 90
加入量
- 6.9 34.5 54.1 70.3
0.03% 6.9 10.3 15.6 30.5
0.05% 6.9 7.5 10.4 28.7
表2
保存的天数 0 30 60 90
加入量
- 2.3 10 57 83
0.03% 2.3 5 34 51
0.05% 2.3 4 29 45
~12~
Claims (6)
1、一种具下式的抗坏血酸衍生物的制备方法,
其中R1是(A):C5-C22直链或支链烷基,或带有取代基的C1-C10直链或支链烷基,所述取代基包括:(1)C1-C6烷氧羰基;(2)芳基,并且可带有选自C1-C5烷基、C1-C3烷氧基、以及苄氧基的取代基;(3)2,3-二甲氧基-5-甲基-1,4-苯醌基;
(B):可带有取代基的C2-C20链烯基,所述取代基包括芳基、3-吡啶基
(C):一种芳基;
(D):包括以下各种的C1-C9酰基:吗啉代羰基、C1-C3烷氧羰基吡咯烷羰基、C1-C3烷氧羰基;
该方法包括将具下式的化合物的5,6-缩醛或缩酮衍生物经水解反应和/或还原反应而成,
式中R1如上定义,R2是通过水解反应和/或还原反应可以除掉的基团。
2、按权利要求1的方法,其中R1是-(CH2)13CH。
3、按权利要求1的方法,其中R1是-(CH2)13CH。
4、按权利要求1的方法,其中R1是-(CH2)18CH。
5、按权利要求1的方法,其中R1是-(CH2)9COOCH3。
6、按权利要求1的方法,其中R1是
(CH2)7CH3。
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP58240741A JPS60130582A (ja) | 1983-12-19 | 1983-12-19 | 食品用酸化防止剤,アスコルビン酸誘導体およびその製造法 |
| DE8484115337T DE3473448D1 (en) | 1983-12-19 | 1984-12-13 | Ascorbic acid ethers and their production |
| EP84115337A EP0146121B1 (en) | 1983-12-19 | 1984-12-13 | Ascorbic acid ethers and their production |
| CA000470382A CA1236461A (en) | 1983-12-19 | 1984-12-18 | Ascorbic acid ethers and their production |
| AU40391/85A AU576835B2 (en) | 1983-12-19 | 1985-03-26 | Ascorbic acid derivatives |
| IE80185A IE58383B1 (en) | 1983-12-19 | 1985-03-28 | Ascorbic acid ethers and their production |
| HU851176A HU197734B (en) | 1983-12-19 | 1985-03-28 | Antioxidant compositions containing ascorbinic acid derivatives as active component and process for producing ascorbinic acid derivatives |
| JP60079069A JPS61236772A (ja) | 1983-12-19 | 1985-04-12 | 食品用酸化防止剤,アスコルビン酸誘導体およびその製造法 |
| SU853884104A SU1428198A3 (ru) | 1983-12-19 | 1985-04-19 | Способ получени производных аскорбиновой кислоты |
| CN85102961.2A CN1009552B (zh) | 1983-12-19 | 1985-04-20 | 抗坏血酸衍生物的制备方法 |
| US07/149,895 US4780549A (en) | 1983-12-19 | 1988-01-28 | Ascorbic acid derivatives and their production |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP58240741A JPS60130582A (ja) | 1983-12-19 | 1983-12-19 | 食品用酸化防止剤,アスコルビン酸誘導体およびその製造法 |
| HU851176A HU197734B (en) | 1983-12-19 | 1985-03-28 | Antioxidant compositions containing ascorbinic acid derivatives as active component and process for producing ascorbinic acid derivatives |
| JP60079069A JPS61236772A (ja) | 1983-12-19 | 1985-04-12 | 食品用酸化防止剤,アスコルビン酸誘導体およびその製造法 |
| SU853884104A SU1428198A3 (ru) | 1983-12-19 | 1985-04-19 | Способ получени производных аскорбиновой кислоты |
| CN85102961.2A CN1009552B (zh) | 1983-12-19 | 1985-04-20 | 抗坏血酸衍生物的制备方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN85102961A CN85102961A (zh) | 1986-12-24 |
| CN1009552B true CN1009552B (zh) | 1990-09-12 |
Family
ID=36838685
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN85102961.2A Expired CN1009552B (zh) | 1983-12-19 | 1985-04-20 | 抗坏血酸衍生物的制备方法 |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US4780549A (zh) |
| EP (1) | EP0146121B1 (zh) |
| JP (2) | JPS60130582A (zh) |
| CN (1) | CN1009552B (zh) |
| AU (1) | AU576835B2 (zh) |
| CA (1) | CA1236461A (zh) |
| DE (1) | DE3473448D1 (zh) |
| HU (1) | HU197734B (zh) |
| IE (1) | IE58383B1 (zh) |
| SU (1) | SU1428198A3 (zh) |
Families Citing this family (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1986007592A1 (fr) * | 1985-06-18 | 1986-12-31 | Takeda Chemical Industries, Ltd. | Derives d'acide ascorbique, leur procede de preparation, et preparations les contenant |
| ZA852614B (en) * | 1985-05-17 | 1986-10-09 | Takeda Chemical Industries, Ltd. | Ascorbic acid ethers and their production |
| JPS63138678A (ja) * | 1986-12-01 | 1988-06-10 | ヒロセ電機株式会社 | 電気コネクタ |
| JPS62210792A (ja) * | 1986-03-12 | 1987-09-16 | Alps Electric Co Ltd | 有料テレビ放送のスクランブル方式 |
| JPS62221611A (ja) * | 1986-03-20 | 1987-09-29 | Kanebo Ltd | 皮膚化粧料 |
| JPH07112966B2 (ja) * | 1986-03-26 | 1995-12-06 | 鐘紡株式会社 | 皮膚化粧料 |
| JP2515556B2 (ja) * | 1986-08-29 | 1996-07-10 | 武田薬品工業株式会社 | ヒドロキシブテノライド誘導体およびその製造法 |
| JPH0813739B2 (ja) * | 1986-12-03 | 1996-02-14 | 武田薬品工業株式会社 | 白内障治療剤 |
| US4826872A (en) * | 1986-12-03 | 1989-05-02 | Takeda Chemical Industries, Ltd. | Pharmaceutical composition for treatment of cataract |
| DK331388A (da) * | 1987-07-09 | 1989-01-10 | Hoffmann La Roche | Antioxidanter |
| JPH01228978A (ja) * | 1988-03-09 | 1989-09-12 | Nippon Hai Potsukusu:Kk | アスコルビン酸誘導体及びその製造方法並びにアスコルビン酸誘導体からなる抗酸化剤 |
| JPH0730066B2 (ja) * | 1988-03-09 | 1995-04-05 | 株式会社日本ハイポックス | アスコルビン酸誘導体及びアスコルビン酸誘導体からなる抗酸化剤 |
| JP2752080B2 (ja) * | 1988-03-09 | 1998-05-18 | 株式会社日本ハイポックス | アスコルビン酸誘導体及びその製造方法並びにアスコルビン酸誘導体からなる抗酸化剤 |
| JP2830268B2 (ja) * | 1989-01-21 | 1998-12-02 | 武田薬品工業株式会社 | アスコルビン酸誘導体の製造法 |
| ATE123306T1 (de) * | 1989-05-19 | 1995-06-15 | Hayashibara Biochem Lab | Alpha-glycosyl-l-ascorbinsäure und ihre herstellung und verwendungen. |
| US5034543A (en) * | 1989-07-20 | 1991-07-23 | Nippon Hypox Laboratories Incorporated | Ascorbic acid derivative and use as antioxidants |
| US5061812A (en) * | 1989-07-20 | 1991-10-29 | Nippon Hypox Laboratories Incorporated | Ascorbic acid derivative |
| WO1991003471A1 (en) * | 1989-09-11 | 1991-03-21 | Nippon Hypox Laboratories Incorporated | Ascorbic acid derivatives |
| TW325997B (en) * | 1993-02-02 | 1998-02-01 | Senju Pharma Co | Pharmaceutical composition for preventing and treating retinal diseases |
| US5441872A (en) * | 1993-06-04 | 1995-08-15 | Board Of Supervisors Of Louisiana State University And Agricultural And Mechanical College | Method for the analysis of Vitamin C |
| FR2715156B1 (fr) * | 1994-01-20 | 1996-03-01 | Oreal | Mono-esters d'acide cinnamique ou de ses dérivés et de vitamine C, leur procédé de préparation et leur utilisation comme anti-oxydants dans des compositions cosmétiques, pharmaceutiques ou alimentaires. |
| KR100399348B1 (ko) * | 1998-03-27 | 2004-03-22 | 주식회사 엘지생명과학 | 폴리에톡실화비타민c유도체및그의제조방법 |
| CN1295567A (zh) * | 1998-03-27 | 2001-05-16 | 株式会社Lg化学 | 作为新型抗氧剂的聚乙氧基化抗坏血酸衍生物及其制备方法 |
| KR100604068B1 (ko) * | 1999-09-14 | 2006-07-24 | 주식회사 엘지생활건강 | 자유라디칼 소거제 혹은 항산화제로서 폴리에톡실레이티드 비타민 씨 유도체를 함유하는 조성물 |
| FR2807320B1 (fr) * | 2000-04-10 | 2002-05-24 | Oreal | Utilisation de derives d'acide ascorbique pour augmenter la synthese des creramides epidermiques |
| FR2807322B1 (fr) | 2000-04-10 | 2004-02-20 | Oreal | Composition, notamment cosmetique, comprenant de l'acide ascorbique en association avec un derive d'acide ascorbique |
| KR100450274B1 (ko) * | 2001-07-30 | 2004-09-30 | 주식회사 엘지생명과학 | 폴리에톡실화 아스코르브산 유도체를 함유하는 동물용사료첨가제 조성물 |
| JP2003070450A (ja) * | 2001-09-04 | 2003-03-11 | Toshiaki Oshima | 食品用黒変・褐変防止剤 |
| US20080255228A1 (en) * | 2006-08-04 | 2008-10-16 | Bioderm Research | Ascorbic Acid Complexes for Skin Care Including Treatment of Dark Skin & Wrinkles |
| US20060093676A1 (en) * | 2002-12-07 | 2006-05-04 | Hebert Rolland F | Ascorbic acid stability |
| BRPI0816234B1 (pt) * | 2007-08-22 | 2018-01-23 | Seiwa Kasei Co., Ltd. | Derivado de ácido ascórbico ou sal do mesmo, método de produção do mesmo, e cosmético |
| FR2955493B1 (fr) * | 2010-01-28 | 2012-02-24 | Oreal | Utilisation de derives d'acide ascorbique en coloration des fibres keratiniques humaines, composition les comprenant, procede de coloration et dispositif |
| JP5602456B2 (ja) * | 2010-02-24 | 2014-10-08 | 日本精化株式会社 | アスコルビン酸誘導体 |
| FR2988092B1 (fr) * | 2012-03-16 | 2014-04-25 | Centre Nat Rech Scient | Complexes de vitamine c, nanoparticules desdits complexes, procedes pour leur preparation, leurs compositions, leurs utilisations cosmetiques et procede de traitement cosmetique |
| WO2017095704A1 (en) * | 2015-12-03 | 2017-06-08 | 3M Innovative Properties Company | Redox polymerizable composition with photolabile reducing agents |
| CN110709486B (zh) | 2017-05-24 | 2021-04-16 | 3M创新有限公司 | 粘合剂制品及其制造和使用方法 |
| CN115260170B (zh) * | 2021-04-30 | 2024-05-28 | 禾美生物科技(浙江)有限公司 | 抗坏血酸多肽衍生物及其制备方法和应用 |
| CN119331136A (zh) * | 2023-07-21 | 2025-01-21 | 中国石油天然气股份有限公司 | 烯烃聚合催化剂的组分、烯烃聚合催化剂及其应用 |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS585920B2 (ja) * | 1977-01-14 | 1983-02-02 | 三菱化学株式会社 | L−アスコルビン酸誘導体 |
| US4208434A (en) * | 1978-07-11 | 1980-06-17 | The Coca-Cola Company | Color stable food containing anthocyanic pigments and bio-available vitamin C and process for its production |
| EP0057699A4 (en) * | 1980-08-14 | 1982-11-08 | Nat Foundation For Cancer Res | 5,6-O-ISOALKYLIDENE ASCORBIC ACID DERIVATIVES. |
| FI830078L (fi) * | 1982-01-15 | 1983-07-16 | Lilly Co Eli | Askorbinsyraetrar och liknande foereningar |
| US4552888A (en) * | 1982-01-15 | 1985-11-12 | Eli Lilly And Company | Ascorbic acid ethers in angiogene |
| JPH0358345A (ja) * | 1989-07-25 | 1991-03-13 | Nec Corp | ディスク装置の電源制御方式 |
-
1983
- 1983-12-19 JP JP58240741A patent/JPS60130582A/ja active Granted
-
1984
- 1984-12-13 EP EP84115337A patent/EP0146121B1/en not_active Expired
- 1984-12-13 DE DE8484115337T patent/DE3473448D1/de not_active Expired
- 1984-12-18 CA CA000470382A patent/CA1236461A/en not_active Expired
-
1985
- 1985-03-26 AU AU40391/85A patent/AU576835B2/en not_active Ceased
- 1985-03-28 HU HU851176A patent/HU197734B/hu not_active IP Right Cessation
- 1985-03-28 IE IE80185A patent/IE58383B1/en not_active IP Right Cessation
- 1985-04-12 JP JP60079069A patent/JPS61236772A/ja active Pending
- 1985-04-19 SU SU853884104A patent/SU1428198A3/ru active
- 1985-04-20 CN CN85102961.2A patent/CN1009552B/zh not_active Expired
-
1988
- 1988-01-28 US US07/149,895 patent/US4780549A/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| HU197734B (en) | 1989-05-29 |
| EP0146121A3 (en) | 1985-11-13 |
| DE3473448D1 (en) | 1988-09-22 |
| AU576835B2 (en) | 1988-09-08 |
| EP0146121A2 (en) | 1985-06-26 |
| IE58383B1 (en) | 1993-09-08 |
| JPH0358345B2 (zh) | 1991-09-05 |
| CN85102961A (zh) | 1986-12-24 |
| SU1428198A3 (ru) | 1988-09-30 |
| AU4039185A (en) | 1986-10-02 |
| US4780549A (en) | 1988-10-25 |
| HUT38923A (en) | 1986-07-28 |
| JPS60130582A (ja) | 1985-07-12 |
| JPS61236772A (ja) | 1986-10-22 |
| IE850801L (en) | 1986-09-28 |
| EP0146121B1 (en) | 1988-08-17 |
| CA1236461A (en) | 1988-05-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1009552B (zh) | 抗坏血酸衍生物的制备方法 | |
| CN1158255C (zh) | 类胡萝卜素的制备方法 | |
| Campion et al. | A stable. eta. 2-silene complex of iridium:(. eta. 5-C5Me5)(PMe3) Ir (. eta. 2-CH2: SiPh2) | |
| Lehr et al. | A New Method for the Preparation of Non‐Terminal Alkynes: Application to the Total Syntheses of Tulearin A and C | |
| CN1073096C (zh) | 制备2-氯-5-氯甲基-1,3-噻唑的方法 | |
| Saito et al. | Mixed Crossed Aldol Condensation between Conjugated Esters and Aldehydes Using Aluminum Tris (2, 6‐diphenylphenoxide) | |
| DE60123579T2 (de) | Synthese von 3,6-dialkyl-5,6-dihydro-4-hydroxy-pyran-2-one | |
| CN1202070C (zh) | C12-c22脂肪酸单酸甘油酯和甘油二酯的二乙酰酒石酸酯 | |
| CN1135220C (zh) | 顺式构型不饱和酯,其生产方法以及含有它的香料组合物 | |
| FR2534907A1 (fr) | Complexes diamine-platine, un procede pour leur preparation, un procede pour la preparation d'une composition medicinale utilisant un tel complexe diamine platine pour le traitement du cancer ainsi que la composition ainsi formee | |
| CN1267018C (zh) | 风味和香料组合物 | |
| CN1019010B (zh) | 取代的β-二酮的制备方法 | |
| CN1241890C (zh) | 制备2,7-二甲基-2,4,6-辛三烯醛单缩醛的方法 | |
| Muñoz et al. | Hydroxy-and alkoxycyclizations of enynes catalyzed by platinum (II) chloride | |
| CN1248964A (zh) | 一种通过羰基化反应制备醛的方法 | |
| CN1245934C (zh) | 香料和风味组合物 | |
| JP3586441B2 (ja) | 香味料前駆体 | |
| CN1030583A (zh) | 制备6,7-二酰基-7-脱乙酰福斯克林衍生物的新方法 | |
| EP3640255B1 (en) | Method for producing silicon compound, and silicon compound | |
| JP6154818B2 (ja) | オキソビニルイオノール及びそのo保護誘導体の製造方法 | |
| CN113979864A (zh) | 一种新型薄荷醇碳酸酯类凉味剂的一锅法合成方法及其在卷烟中的应用 | |
| CN117624016B (zh) | 一种异吲哚啉酮类化合物的合成方法 | |
| JPH09118893A (ja) | 置換されたラクトン類の芳香物質としての使用 | |
| CN108341803A (zh) | 一种苯醚甲环唑的合成方法 | |
| Krauß et al. | Synthesis and Antimicrobial Activity of Hydroxyalkyl‐and Hydroxyacyl‐phenols and Their Benzyl Ethers |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C13 | Decision | ||
| GR02 | Examined patent application | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C19 | Lapse of patent right due to non-payment of the annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |