WO2023276526A1 - 非水電解質二次電池 - Google Patents
非水電解質二次電池 Download PDFInfo
- Publication number
- WO2023276526A1 WO2023276526A1 PCT/JP2022/022080 JP2022022080W WO2023276526A1 WO 2023276526 A1 WO2023276526 A1 WO 2023276526A1 JP 2022022080 W JP2022022080 W JP 2022022080W WO 2023276526 A1 WO2023276526 A1 WO 2023276526A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- aqueous electrolyte
- composite oxide
- positive electrode
- less
- secondary battery
- Prior art date
Links
- 239000011255 nonaqueous electrolyte Substances 0.000 title claims abstract description 102
- -1 lithium transition metal Chemical class 0.000 claims abstract description 35
- 239000007774 positive electrode material Substances 0.000 claims abstract description 28
- 229910052751 metal Inorganic materials 0.000 claims abstract description 27
- 239000002905 metal composite material Substances 0.000 claims abstract description 24
- 229910052723 transition metal Inorganic materials 0.000 claims abstract description 24
- 125000000217 alkyl group Chemical group 0.000 claims abstract description 22
- 229910052782 aluminium Inorganic materials 0.000 claims abstract description 19
- 229910052744 lithium Inorganic materials 0.000 claims abstract description 19
- 125000004432 carbon atom Chemical group C* 0.000 claims abstract description 18
- 229910052748 manganese Inorganic materials 0.000 claims abstract description 16
- 229910052759 nickel Inorganic materials 0.000 claims abstract description 16
- 150000002903 organophosphorus compounds Chemical class 0.000 claims abstract description 11
- 150000005676 cyclic carbonates Chemical class 0.000 claims description 27
- SBLRHMKNNHXPHG-UHFFFAOYSA-N 4-fluoro-1,3-dioxolan-2-one Chemical compound FC1COC(=O)O1 SBLRHMKNNHXPHG-UHFFFAOYSA-N 0.000 claims description 13
- 229910052760 oxygen Inorganic materials 0.000 claims description 9
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 5
- 229910052791 calcium Inorganic materials 0.000 claims description 5
- 238000000034 method Methods 0.000 claims description 5
- 239000001301 oxygen Substances 0.000 claims description 5
- VVFGLBKYBBUTRO-UHFFFAOYSA-N 1-[difluoromethyl(ethoxy)phosphoryl]oxyethane Chemical compound CCOP(=O)(C(F)F)OCC VVFGLBKYBBUTRO-UHFFFAOYSA-N 0.000 claims description 4
- 229910052749 magnesium Inorganic materials 0.000 claims description 4
- 229910052758 niobium Inorganic materials 0.000 claims description 4
- 229910052710 silicon Inorganic materials 0.000 claims description 4
- 229910052712 strontium Inorganic materials 0.000 claims description 4
- 229910052719 titanium Inorganic materials 0.000 claims description 4
- QFAUKZLHFVELOW-UHFFFAOYSA-N 1-[ethoxy(trifluoromethyl)phosphoryl]oxyethane Chemical compound CCOP(=O)(C(F)(F)F)OCC QFAUKZLHFVELOW-UHFFFAOYSA-N 0.000 claims description 3
- 229910052750 molybdenum Inorganic materials 0.000 claims description 3
- 229910052721 tungsten Inorganic materials 0.000 claims description 3
- 229910052727 yttrium Inorganic materials 0.000 claims description 3
- HIZZGKZEHBXFAQ-UHFFFAOYSA-N 1-[ethoxy(fluoromethyl)phosphoryl]oxyethane Chemical compound CCOP(=O)(CF)OCC HIZZGKZEHBXFAQ-UHFFFAOYSA-N 0.000 claims description 2
- 229910052796 boron Inorganic materials 0.000 claims description 2
- 229910052742 iron Inorganic materials 0.000 claims description 2
- 229910052706 scandium Inorganic materials 0.000 claims description 2
- 229910052725 zinc Inorganic materials 0.000 claims description 2
- 229910052726 zirconium Inorganic materials 0.000 claims description 2
- 125000004429 atom Chemical group 0.000 abstract 4
- 239000002131 composite material Substances 0.000 description 69
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 32
- 229940126062 Compound A Drugs 0.000 description 25
- NLDMNSXOCDLTTB-UHFFFAOYSA-N Heterophylliin A Natural products O1C2COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC2C(OC(=O)C=2C=C(O)C(O)=C(O)C=2)C(O)C1OC(=O)C1=CC(O)=C(O)C(O)=C1 NLDMNSXOCDLTTB-UHFFFAOYSA-N 0.000 description 25
- 239000002245 particle Substances 0.000 description 23
- 239000010408 film Substances 0.000 description 19
- 239000011572 manganese Substances 0.000 description 19
- 239000002184 metal Substances 0.000 description 16
- 239000000203 mixture Substances 0.000 description 16
- 102100028667 C-type lectin domain family 4 member A Human genes 0.000 description 14
- 101000766908 Homo sapiens C-type lectin domain family 4 member A Proteins 0.000 description 14
- 230000000694 effects Effects 0.000 description 14
- 239000000463 material Substances 0.000 description 13
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 10
- 238000007600 charging Methods 0.000 description 10
- 125000001153 fluoro group Chemical group F* 0.000 description 10
- 239000011248 coating agent Substances 0.000 description 9
- 238000000576 coating method Methods 0.000 description 9
- 239000013078 crystal Substances 0.000 description 9
- 238000007599 discharging Methods 0.000 description 9
- 239000011267 electrode slurry Substances 0.000 description 8
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 8
- 229910001416 lithium ion Inorganic materials 0.000 description 8
- 238000004519 manufacturing process Methods 0.000 description 8
- 238000007789 sealing Methods 0.000 description 8
- 238000003860 storage Methods 0.000 description 8
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 7
- 239000007789 gas Substances 0.000 description 7
- 239000007773 negative electrode material Substances 0.000 description 7
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 6
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 239000011230 binding agent Substances 0.000 description 6
- 230000003247 decreasing effect Effects 0.000 description 6
- 238000010438 heat treatment Methods 0.000 description 6
- 230000014759 maintenance of location Effects 0.000 description 6
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 6
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 5
- 150000001875 compounds Chemical class 0.000 description 5
- 239000006258 conductive agent Substances 0.000 description 5
- 229910052731 fluorine Inorganic materials 0.000 description 5
- 229910002804 graphite Inorganic materials 0.000 description 5
- 239000010439 graphite Substances 0.000 description 5
- 229910003002 lithium salt Inorganic materials 0.000 description 5
- 159000000002 lithium salts Chemical class 0.000 description 5
- 239000011163 secondary particle Substances 0.000 description 5
- 239000002562 thickening agent Substances 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 230000000996 additive effect Effects 0.000 description 4
- 239000007864 aqueous solution Substances 0.000 description 4
- 239000003125 aqueous solvent Substances 0.000 description 4
- 239000003575 carbonaceous material Substances 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 230000006866 deterioration Effects 0.000 description 4
- 239000002612 dispersion medium Substances 0.000 description 4
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 4
- 238000010304 firing Methods 0.000 description 4
- 239000011888 foil Substances 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 4
- 150000002739 metals Chemical class 0.000 description 4
- 239000011164 primary particle Substances 0.000 description 4
- 230000009467 reduction Effects 0.000 description 4
- 230000002829 reductive effect Effects 0.000 description 4
- 235000002639 sodium chloride Nutrition 0.000 description 4
- RIUWBIIVUYSTCN-UHFFFAOYSA-N trilithium borate Chemical compound [Li+].[Li+].[Li+].[O-]B([O-])[O-] RIUWBIIVUYSTCN-UHFFFAOYSA-N 0.000 description 4
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 229910013870 LiPF 6 Inorganic materials 0.000 description 3
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 3
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 238000010828 elution Methods 0.000 description 3
- 239000000835 fiber Substances 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 150000002642 lithium compounds Chemical class 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 239000002994 raw material Substances 0.000 description 3
- 229920005989 resin Polymers 0.000 description 3
- 239000011347 resin Substances 0.000 description 3
- 229910052814 silicon oxide Inorganic materials 0.000 description 3
- 230000006641 stabilisation Effects 0.000 description 3
- 238000011105 stabilization Methods 0.000 description 3
- 229920003048 styrene butadiene rubber Polymers 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- 239000010936 titanium Substances 0.000 description 3
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 2
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 2
- OIFBSDVPJOWBCH-UHFFFAOYSA-N Diethyl carbonate Chemical compound CCOC(=O)OCC OIFBSDVPJOWBCH-UHFFFAOYSA-N 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 2
- 239000002174 Styrene-butadiene Substances 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 150000001642 boronic acid derivatives Chemical class 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 150000005678 chain carbonates Chemical class 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 239000011889 copper foil Substances 0.000 description 2
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 2
- 239000008151 electrolyte solution Substances 0.000 description 2
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- FKRCODPIKNYEAC-UHFFFAOYSA-N ethyl propionate Chemical compound CCOC(=O)CC FKRCODPIKNYEAC-UHFFFAOYSA-N 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 2
- GAEKPEKOJKCEMS-UHFFFAOYSA-N gamma-valerolactone Chemical compound CC1CCC(=O)O1 GAEKPEKOJKCEMS-UHFFFAOYSA-N 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- 238000002354 inductively-coupled plasma atomic emission spectroscopy Methods 0.000 description 2
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical compound OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 description 2
- AMXOYNBUYSYVKV-UHFFFAOYSA-M lithium bromide Chemical compound [Li+].[Br-] AMXOYNBUYSYVKV-UHFFFAOYSA-M 0.000 description 2
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 2
- TZIHFWKZFHZASV-UHFFFAOYSA-N methyl formate Chemical compound COC=O TZIHFWKZFHZASV-UHFFFAOYSA-N 0.000 description 2
- 239000012046 mixed solvent Substances 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- LGQLOGILCSXPEA-UHFFFAOYSA-L nickel sulfate Chemical compound [Ni+2].[O-]S([O-])(=O)=O LGQLOGILCSXPEA-UHFFFAOYSA-L 0.000 description 2
- 229910000363 nickel(II) sulfate Inorganic materials 0.000 description 2
- 125000004437 phosphorous atom Chemical group 0.000 description 2
- 229910052698 phosphorus Inorganic materials 0.000 description 2
- 229920000573 polyethylene Polymers 0.000 description 2
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 2
- 239000010703 silicon Substances 0.000 description 2
- LIVNPJMFVYWSIS-UHFFFAOYSA-N silicon monoxide Chemical class [Si-]#[O+] LIVNPJMFVYWSIS-UHFFFAOYSA-N 0.000 description 2
- 239000002002 slurry Substances 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 229910001220 stainless steel Inorganic materials 0.000 description 2
- 239000010935 stainless steel Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 239000010409 thin film Substances 0.000 description 2
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 2
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 2
- YIUFTMLPQFZEFD-UHFFFAOYSA-N 1,1,1-trifluoro-2-[methyl(2,2,2-trifluoroethoxy)phosphoryl]oxyethane Chemical compound FC(F)(F)COP(=O)(C)OCC(F)(F)F YIUFTMLPQFZEFD-UHFFFAOYSA-N 0.000 description 1
- VAYTZRYEBVHVLE-UHFFFAOYSA-N 1,3-dioxol-2-one Chemical compound O=C1OC=CO1 VAYTZRYEBVHVLE-UHFFFAOYSA-N 0.000 description 1
- GEWWCWZGHNIUBW-UHFFFAOYSA-N 1-(4-nitrophenyl)propan-2-one Chemical compound CC(=O)CC1=CC=C([N+]([O-])=O)C=C1 GEWWCWZGHNIUBW-UHFFFAOYSA-N 0.000 description 1
- NYYLZXREFNYPKB-UHFFFAOYSA-N 1-[ethoxy(methyl)phosphoryl]oxyethane Chemical compound CCOP(C)(=O)OCC NYYLZXREFNYPKB-UHFFFAOYSA-N 0.000 description 1
- CQCXMYUCNSJSKG-UHFFFAOYSA-N 1-dimethoxyphosphorylethene Chemical compound COP(=O)(OC)C=C CQCXMYUCNSJSKG-UHFFFAOYSA-N 0.000 description 1
- 125000004778 2,2-difluoroethyl group Chemical group [H]C([H])(*)C([H])(F)F 0.000 description 1
- UHOPWFKONJYLCF-UHFFFAOYSA-N 2-(2-sulfanylethyl)isoindole-1,3-dione Chemical compound C1=CC=C2C(=O)N(CCS)C(=O)C2=C1 UHOPWFKONJYLCF-UHFFFAOYSA-N 0.000 description 1
- 125000004777 2-fluoroethyl group Chemical group [H]C([H])(F)C([H])([H])* 0.000 description 1
- ZTTYKFSKZIRTDP-UHFFFAOYSA-N 4,4-difluoro-1,3-dioxolan-2-one Chemical compound FC1(F)COC(=O)O1 ZTTYKFSKZIRTDP-UHFFFAOYSA-N 0.000 description 1
- DSMUTQTWFHVVGQ-UHFFFAOYSA-N 4,5-difluoro-1,3-dioxolan-2-one Chemical compound FC1OC(=O)OC1F DSMUTQTWFHVVGQ-UHFFFAOYSA-N 0.000 description 1
- RMYFSKOGEWSTQR-UHFFFAOYSA-N 4,5-difluoro-4,5-dimethyl-1,3-dioxolan-2-one Chemical compound CC1(F)OC(=O)OC1(C)F RMYFSKOGEWSTQR-UHFFFAOYSA-N 0.000 description 1
- GKZFQPGIDVGTLZ-UHFFFAOYSA-N 4-(trifluoromethyl)-1,3-dioxolan-2-one Chemical compound FC(F)(F)C1COC(=O)O1 GKZFQPGIDVGTLZ-UHFFFAOYSA-N 0.000 description 1
- BJWMSGRKJIOCNR-UHFFFAOYSA-N 4-ethenyl-1,3-dioxolan-2-one Chemical compound C=CC1COC(=O)O1 BJWMSGRKJIOCNR-UHFFFAOYSA-N 0.000 description 1
- LECKFEZRJJNBNI-UHFFFAOYSA-N 4-fluoro-5-methyl-1,3-dioxolan-2-one Chemical compound CC1OC(=O)OC1F LECKFEZRJJNBNI-UHFFFAOYSA-N 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- 229910000838 Al alloy Inorganic materials 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 1
- 229920000049 Carbon (fiber) Polymers 0.000 description 1
- 229910000881 Cu alloy Inorganic materials 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- MEBALPPDZLDKGS-UHFFFAOYSA-M FC(C(C(C(S(=O)(=O)[O-])(F)F)(F)F)(F)F)(F)F.FC(S(=O)(=O)O)(F)F.[Li+] Chemical compound FC(C(C(C(S(=O)(=O)[O-])(F)F)(F)F)(F)F)(F)F.FC(S(=O)(=O)O)(F)F.[Li+] MEBALPPDZLDKGS-UHFFFAOYSA-M 0.000 description 1
- 229910000733 Li alloy Inorganic materials 0.000 description 1
- 229910010238 LiAlCl 4 Inorganic materials 0.000 description 1
- 229910015015 LiAsF 6 Inorganic materials 0.000 description 1
- 229910013063 LiBF 4 Inorganic materials 0.000 description 1
- 229910013684 LiClO 4 Inorganic materials 0.000 description 1
- 229910012513 LiSbF 6 Inorganic materials 0.000 description 1
- RJUFJBKOKNCXHH-UHFFFAOYSA-N Methyl propionate Chemical compound CCC(=O)OC RJUFJBKOKNCXHH-UHFFFAOYSA-N 0.000 description 1
- 229910000990 Ni alloy Inorganic materials 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 239000002033 PVDF binder Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 229910000676 Si alloy Inorganic materials 0.000 description 1
- 229910004298 SiO 2 Inorganic materials 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- SYRDSFGUUQPYOB-UHFFFAOYSA-N [Li+].[Li+].[Li+].[O-]B([O-])[O-].FC(=O)C(F)=O Chemical compound [Li+].[Li+].[Li+].[O-]B([O-])[O-].FC(=O)C(F)=O SYRDSFGUUQPYOB-UHFFFAOYSA-N 0.000 description 1
- 239000006230 acetylene black Substances 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- DIZPMCHEQGEION-UHFFFAOYSA-H aluminium sulfate (anhydrous) Chemical compound [Al+3].[Al+3].[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O DIZPMCHEQGEION-UHFFFAOYSA-H 0.000 description 1
- 229910021383 artificial graphite Inorganic materials 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- IMHDGJOMLMDPJN-UHFFFAOYSA-N biphenyl-2,2'-diol Chemical compound OC1=CC=CC=C1C1=CC=CC=C1O IMHDGJOMLMDPJN-UHFFFAOYSA-N 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Chemical group BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Chemical group 0.000 description 1
- QHIWVLPBUQWDMQ-UHFFFAOYSA-N butyl prop-2-enoate;methyl 2-methylprop-2-enoate;prop-2-enoic acid Chemical compound OC(=O)C=C.COC(=O)C(C)=C.CCCCOC(=O)C=C QHIWVLPBUQWDMQ-UHFFFAOYSA-N 0.000 description 1
- 238000001354 calcination Methods 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 239000004917 carbon fiber Substances 0.000 description 1
- 239000002041 carbon nanotube Substances 0.000 description 1
- 229910021393 carbon nanotube Inorganic materials 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 150000001733 carboxylic acid esters Chemical class 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 229920003086 cellulose ether Polymers 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 229910000361 cobalt sulfate Inorganic materials 0.000 description 1
- 229940044175 cobalt sulfate Drugs 0.000 description 1
- KTVIXTQDYHMGHF-UHFFFAOYSA-L cobalt(2+) sulfate Chemical compound [Co+2].[O-]S([O-])(=O)=O KTVIXTQDYHMGHF-UHFFFAOYSA-L 0.000 description 1
- 230000002301 combined effect Effects 0.000 description 1
- 238000010280 constant potential charging Methods 0.000 description 1
- 238000010277 constant-current charging Methods 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- 238000009831 deintercalation Methods 0.000 description 1
- 230000002542 deteriorative effect Effects 0.000 description 1
- IJKVHSBPTUYDLN-UHFFFAOYSA-N dihydroxy(oxo)silane Chemical compound O[Si](O)=O IJKVHSBPTUYDLN-UHFFFAOYSA-N 0.000 description 1
- IEJIGPNLZYLLBP-UHFFFAOYSA-N dimethyl carbonate Chemical compound COC(=O)OC IEJIGPNLZYLLBP-UHFFFAOYSA-N 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 239000003792 electrolyte Substances 0.000 description 1
- 229940093499 ethyl acetate Drugs 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- WBJINCZRORDGAQ-UHFFFAOYSA-N formic acid ethyl ester Natural products CCOC=O WBJINCZRORDGAQ-UHFFFAOYSA-N 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 229910021469 graphitizable carbon Inorganic materials 0.000 description 1
- 229910021385 hard carbon Inorganic materials 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- 238000009830 intercalation Methods 0.000 description 1
- 230000010220 ion permeability Effects 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 230000002427 irreversible effect Effects 0.000 description 1
- 239000012948 isocyanate Substances 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000001989 lithium alloy Substances 0.000 description 1
- XGZVUEUWXADBQD-UHFFFAOYSA-L lithium carbonate Chemical compound [Li+].[Li+].[O-]C([O-])=O XGZVUEUWXADBQD-UHFFFAOYSA-L 0.000 description 1
- 229910052808 lithium carbonate Inorganic materials 0.000 description 1
- DEUISMFZZMAAOJ-UHFFFAOYSA-N lithium dihydrogen borate oxalic acid Chemical compound B([O-])(O)O.C(C(=O)O)(=O)O.C(C(=O)O)(=O)O.[Li+] DEUISMFZZMAAOJ-UHFFFAOYSA-N 0.000 description 1
- HSZCZNFXUDYRKD-UHFFFAOYSA-M lithium iodide Inorganic materials [Li+].[I-] HSZCZNFXUDYRKD-UHFFFAOYSA-M 0.000 description 1
- FUJCRWPEOMXPAD-UHFFFAOYSA-N lithium oxide Chemical compound [Li+].[Li+].[O-2] FUJCRWPEOMXPAD-UHFFFAOYSA-N 0.000 description 1
- 229910001947 lithium oxide Inorganic materials 0.000 description 1
- ACFSQHQYDZIPRL-UHFFFAOYSA-N lithium;bis(1,1,2,2,2-pentafluoroethylsulfonyl)azanide Chemical compound [Li+].FC(F)(F)C(F)(F)S(=O)(=O)[N-]S(=O)(=O)C(F)(F)C(F)(F)F ACFSQHQYDZIPRL-UHFFFAOYSA-N 0.000 description 1
- QSZMZKBZAYQGRS-UHFFFAOYSA-N lithium;bis(trifluoromethylsulfonyl)azanide Chemical compound [Li+].FC(F)(F)S(=O)(=O)[N-]S(=O)(=O)C(F)(F)F QSZMZKBZAYQGRS-UHFFFAOYSA-N 0.000 description 1
- 229940099596 manganese sulfate Drugs 0.000 description 1
- 239000011702 manganese sulphate Substances 0.000 description 1
- 235000007079 manganese sulphate Nutrition 0.000 description 1
- SQQMAOCOWKFBNP-UHFFFAOYSA-L manganese(II) sulfate Chemical compound [Mn+2].[O-]S([O-])(=O)=O SQQMAOCOWKFBNP-UHFFFAOYSA-L 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 229940017219 methyl propionate Drugs 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 238000010295 mobile communication Methods 0.000 description 1
- YKYONYBAUNKHLG-UHFFFAOYSA-N n-Propyl acetate Natural products CCCOC(C)=O YKYONYBAUNKHLG-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- JRNGUTKWMSBIBF-UHFFFAOYSA-N naphthalene-2,3-diol Chemical compound C1=CC=C2C=C(O)C(O)=CC2=C1 JRNGUTKWMSBIBF-UHFFFAOYSA-N 0.000 description 1
- 229910021382 natural graphite Inorganic materials 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 229910021470 non-graphitizable carbon Inorganic materials 0.000 description 1
- 239000004745 nonwoven fabric Substances 0.000 description 1
- 125000000962 organic group Chemical group 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 229920006122 polyamide resin Polymers 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 239000009719 polyimide resin Substances 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 229920005672 polyolefin resin Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 229940090181 propyl acetate Drugs 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 238000000790 scattering method Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000007086 side reaction Methods 0.000 description 1
- 150000003377 silicon compounds Chemical class 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 229910021384 soft carbon Inorganic materials 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000002759 woven fabric Substances 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0567—Liquid materials characterised by the additives
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0569—Liquid materials characterised by the solvents
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0025—Organic electrolyte
- H01M2300/0028—Organic electrolyte characterised by the solvent
- H01M2300/0034—Fluorinated solvents
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Definitions
- the present disclosure relates to non-aqueous electrolyte secondary batteries.
- a non-aqueous electrolyte secondary battery represented by a lithium-ion secondary battery includes a positive electrode, a negative electrode, and a non-aqueous electrolyte.
- a positive electrode positive electrode
- a negative electrode negative electrode
- a non-aqueous electrolyte In order to ensure excellent characteristics of non-aqueous electrolyte secondary batteries, attempts have been made to improve battery components.
- Patent Document 1 describes a compound (A) having an organic group having 1 to 20 carbon atoms which may have a substituent on the nitrogen atom of isocyanuric acid, a nitrile compound, an isocyanate compound, a difluorophosphoric acid compound, or a fluoro proposed a non-aqueous electrolyte containing sulfonate and the like.
- Patent Document 2 discloses a lithium-containing composite oxide represented by Formula 1: Li x Ni 1-yz-v-w Co y Al z M1 v M2 w O 2 , wherein element M1 in Formula 1 is at least one selected from the group consisting of Mn, Ti, Y, Nb, Mo and W, element M2 is at least two selected from the group consisting of Mg, Ca, Sr and Ba, and The element M2 contains at least Mg and Ca, and formula 1 satisfies 0.97 ⁇ x ⁇ 1.1, 0.05 ⁇ y ⁇ 0.35, 0.005 ⁇ z ⁇ 0.1, 0.0001 ⁇ Satisfying v ⁇ 0.05 and 0.0001 ⁇ w ⁇ 0.05, the composite oxide has primary particles aggregated to form secondary particles, and the average particle diameter of the primary particles of the composite oxide is , 0.1 ⁇ m or more and 3 ⁇ m or less, and the average particle size of the secondary particles of the composite oxide is 8 ⁇ m or more and 20 ⁇ m or less.
- Formula 1 Li x Ni 1-yz-v-
- JP 2014-194930 A Japanese Patent Application Laid-Open No. 2006-310181
- the positive electrode includes a positive electrode active material
- the positive electrode active material includes a lithium-transition metal composite oxide containing Ni, Mn, and Al, and a metal other than Li contained in the lithium-transition metal composite oxide.
- the proportions of Ni, Mn, and Al in the elements are Ni: 50 atomic % or more, Mn: 10 atomic % or less, and Al: 10 atomic % or less, and the lithium-transition metal composite oxide contains Co
- the ratio of Co to the metal elements other than Li is 1.5 atomic % or less
- the non-aqueous electrolyte has the general formula (1):
- R 1 and R 2 are each independently an alkyl group having 1 to 4 carbon atoms
- R 3 is an alkyl group having 1 to 4 carbon atoms. It relates to a non-aqueous electrolyte secondary battery, which is a fluorinated alkyl group.
- FIG. 1 is a schematic perspective view of a partially cutaway non-aqueous electrolyte secondary battery according to an embodiment of the present disclosure
- any of the illustrated lower limits and any of the illustrated upper limits can be arbitrarily combined as long as the lower limit is not greater than or equal to the upper limit.
- a plurality of materials are exemplified, one of them may be selected and used alone, or two or more may be used in combination.
- the present disclosure encompasses a combination of matters described in two or more claims arbitrarily selected from the multiple claims described in the attached claims. In other words, as long as there is no technical contradiction, the matters described in two or more claims arbitrarily selected from the multiple claims described in the attached claims can be combined.
- Non-aqueous electrolyte secondary batteries include at least lithium ion batteries and lithium metal secondary batteries.
- a nonaqueous electrolyte secondary battery includes a positive electrode, a negative electrode, and a nonaqueous electrolyte.
- the positive electrode contains a positive electrode active material.
- the positive electrode active material contains a lithium transition metal composite oxide containing Ni, Mn and Al.
- the Co content of the lithium-transition metal composite oxide can be reduced and the Ni content can be increased, it is advantageous in terms of cost and can ensure a high capacity. Therefore, in the non-aqueous electrolyte secondary battery according to the present disclosure, the Ni content of the lithium-transition metal composite oxide is increased.
- the lithium-transition metal composite oxide does not contain Co, or the ratio of Co to metal elements other than Li is limited to 1.5 atomic % or less. .
- the lithium-transition metal composite oxide in the non-aqueous electrolyte secondary battery according to the present disclosure is also referred to as “composite oxide NMA”.
- the proportions of Ni, Mn, and Al in the metal elements other than Li contained in the composite oxide NMA are Ni: 50 atomic % or more, Mn: 10 atomic % or less, and Al: 10 atomic % or less, and The composite oxide NMA does not contain Co, or the ratio of Co to metal elements other than Li is 1.5 atomic % or less.
- Mn and Al contribute to stabilization of the crystal structure of the composite oxide NMA with a reduced Co content.
- the composite oxide NMA has a Co content limited to 1.5 atomic % or less and a high Ni content, the crystal structure is likely to be unstable, and the composite oxide NMA has Al, Ni and other metals can be eluted.
- the positive electrode capacity is lowered, and the cycle characteristics (or capacity retention rate) are lowered.
- eluted Ni forms an oxide film having a structure that prevents the absorption and release of Li ions on the particle surface of the composite oxide NMA, which may lead to an increase in internal resistance. .
- the non-aqueous electrolyte is easily oxidatively decomposed, thereby deteriorating cycle characteristics (or capacity retention rate) and increasing resistance.
- the non-aqueous electrolyte secondary battery uses a composite oxide NMA and a non-aqueous electrolyte containing an organic phosphorus compound (hereinafter also referred to as compound A) represented by the following general formula (1).
- a water electrolyte is used.
- R 1 and R 2 are each independently an alkyl group having 1 to 4 carbon atoms.
- R 3 is a fluorinated alkyl group having 1 to 4 carbon atoms.
- the oxidation resistance of the non-aqueous electrolyte is improved, and a good coating derived from the compound A is formed on the surface of the positive electrode active material particles.
- the film has excellent ionic conductivity and stability, and suppresses deterioration of the positive electrode active material (elution of metal from the positive electrode active material) due to contact between the positive electrode active material and the non-aqueous electrolyte. As a result, it is considered that deterioration in cycle characteristics due to deterioration of the positive electrode active material is suppressed.
- the effect of improving the cycle characteristics and the effect of suppressing the increase in internal resistance cannot be significantly obtained.
- the above effect can be remarkably obtained by combining the compound A with the composite oxide NMA.
- the effect of the composite oxide NMA is remarkable because the composite oxide NMA has a large resistance of the composite oxide itself and relatively brittle particles compared to the lithium transition metal composite oxide having a higher Co content. It is considered to be for Particles of the composite oxide NMA are prone to cracking, elution of metal is likely to be significant, and resistance is likely to increase during charging and discharging.
- the range of improvement in the properties due to the film derived from the compound A is large.
- lithium-transition metal composite oxides with a high Co content are superior from this point of view, so it can be said that the need to use compound A is low.
- R 1 and R 2 are each independently an alkyl group having 1 to 4 carbon atoms.
- the alkyl group having 1 to 4 carbon atoms may be linear or branched.
- Examples of alkyl groups having 1 to 4 carbon atoms include methyl group, ethyl group, n-propyl group, isopropyl group, n-butyl group, sec-butyl group and tert-butyl group. Among them, a methyl group and an ethyl group are preferable.
- R 1 and R 2 are fluorinated alkyl group having 1 to 4 carbon atoms, the solubility of the reaction product derived from the organophosphorus compound tends to increase and the ability to form a film tends to decrease.
- R 3 is a fluorinated alkyl group having 1 to 4 carbon atoms.
- the fluorinated alkyl group means a group in which at least one hydrogen atom of an alkyl group is substituted with a fluorine atom.
- the remaining hydrogen atoms not substituted with fluorine atoms may be further substituted with halogen atoms other than fluorine atoms (eg, chlorine atom, bromine atom).
- a fluorinated alkyl group may be linear or branched.
- R 3 is an alkyl group having 1 to 4 carbon atoms and does not contain a fluorine atom, it is considered that a film derived from an organophosphorus compound is unlikely to exist stably. Moreover, even if R 3 is a fluorinated alkyl group having 1 to 4 carbon atoms, if it is bonded to a phosphorus atom via an oxygen atom, the resistance of the film tends to increase.
- Examples of the fluorinated alkyl group having 1 to 4 carbon atoms include a fluoromethyl group, a difluoromethyl group, a trifluoromethyl group, a 2-fluoroethyl group, a 2,2-difluoroethyl group and a 2,2,2-trifluoroethyl group.
- a fluoromethyl group, a difluoromethyl group, and a trifluoromethyl group are preferred.
- a specific example of compound A is preferably at least one selected from the group consisting of diethyl(fluoromethyl)phosphonate, diethyl(difluoromethyl)phosphonate and diethyl(trifluoromethyl)phosphonate. Among them, diethyl (difluoromethyl) phosphonate is more preferable.
- Compound A may be used alone or in combination of two or more.
- the non-aqueous electrolyte may further contain a fluorinated cyclic carbonate.
- the fluorinated cyclic carbonate means a compound in which at least one hydrogen atom of the cyclic carbonate is substituted with a fluorine atom.
- a fluorinated cyclic carbonate e.g., FEC
- FEC fluorinated cyclic carbonate
- Fluorinated cyclic carbonates include, for example, compounds represented by the following general formula (2).
- R 4 to R 7 are each independently a hydrogen atom or a methyl group, and at least one of the hydrogen atom and the hydrogen atom of the methyl group is substituted with a fluorine atom. ing. The remaining hydrogen atoms not substituted with fluorine atoms may be substituted with halogen atoms other than fluorine atoms (for example, chlorine atoms and bromine atoms).
- fluorinated cyclic carbonates include fluoroethylene carbonate (FEC), 1,2-difluoroethylene carbonate, 1,1-difluoroethylene carbonate, 1-fluoropropylene carbonate, 3,3,3-trifluoropropylene carbonate, 2,3-difluoro-2,3-butylene carbonate and the like.
- FEC fluoroethylene carbonate
- 1,2-difluoroethylene carbonate 1,1-difluoroethylene carbonate
- 1-fluoropropylene carbonate 1,3,3-trifluoropropylene carbonate
- 2,3-difluoro-2,3-butylene carbonate 2,3-difluoro-2,3-butylene carbonate and the like.
- Fluorinated cyclic carbonates may be used alone or in combination of two or more.
- the non-aqueous electrolyte secondary battery according to the present disclosure will be specifically described below for each component.
- the positive electrode contains a positive electrode active material.
- a positive electrode generally includes a positive electrode current collector and a layered positive electrode mixture (hereinafter referred to as a positive electrode mixture layer) held by the positive electrode current collector.
- the positive electrode mixture layer can be formed by coating the surface of the positive electrode current collector with a positive electrode slurry in which the components of the positive electrode mixture are dispersed in a dispersion medium, and drying the slurry. The dried coating film may be rolled if necessary.
- the positive electrode mixture contains a positive electrode active material as an essential component, and may contain a binder, a thickener, a conductive agent, etc. as optional components.
- the positive electrode active material contains composite oxide NMA.
- Composite oxide NMA contains Ni, Mn, and Al, and may contain a trace amount of Co, or may contain no Co. From the viewpoint of manufacturing cost reduction, it is desirable that the Co content is as small as possible. The following are more preferable, and Co-free is the most preferable.
- the proportions of Ni, Mn and Al in the metal elements other than Li are Ni: 50 atomic % or more, Mn: 10 atomic % or less, and Al: 10 atomic %. atomic % or less.
- the Ni content in the metal elements other than Li is desirably 80 atomic % or more, more desirably 90 atomic % or more, and may be 92 atomic % or more.
- the Mn content may be 7 atomic % or less, 5 atomic % or less, or 3 atomic % or less.
- the Al content may be 9 atomic % or less, 7 atomic % or less, or 5 atomic % or less.
- Composite oxide NMA has, for example, a layered crystal structure (for example, rock salt type crystal structure).
- the composite oxide NMA is represented, for example, by the formula: Li ⁇ Ni (1-x1-x2-yz) Co x1 Mn x2 Al y M z O 2+ ⁇ .
- Element M is an element other than Li, Ni, Mn, Al, Co and oxygen.
- ⁇ indicating the atomic ratio of lithium is, for example, 0.95 ⁇ 1.05. ⁇ increases and decreases due to charging and discharging. In (2+ ⁇ ) representing the atomic ratio of oxygen, ⁇ satisfies ⁇ 0.05 ⁇ 0.05.
- the valence of Ni in the composite oxide NMA with increased capacity tends to increase.
- the atomic ratio of Ni increases, the atomic ratio of other elements relatively decreases. In this case, especially in a fully charged state, the crystal structure tends to become unstable, metals (such as Ni) tend to leach out, and the crystal structure changes to a crystal structure that makes reversible absorption and release of lithium ions difficult due to repeated charging and discharging. becomes easier to inactivate. As a result, cycle characteristics tend to deteriorate. Therefore, when the composite oxide NMA with a high Ni content is used, the addition of the compound A to the non-aqueous electrolyte significantly improves the cycle characteristics.
- x1 which indicates the atomic ratio of Co, is, for example, 0.015 or less (0 ⁇ x1 ⁇ 0.015), may be 0.01 or less, or may be 0.005 or less.
- x1 0, the case where Co is below the detection limit is included.
- x2 which indicates the atomic ratio of Mn, is, for example, 0.1 or less (0 ⁇ x2 ⁇ 0.1), may be 0.07 or less, may be 0.05 or less, or may be 0.03 It may be below. x2 may be 0.01 or more, or may be 0.02 or more. Mn contributes to stabilization of the crystal structure of the composite oxide NMA, and the composite oxide NMA contains inexpensive Mn, which is advantageous for cost reduction.
- y which indicates the atomic ratio of Al, is, for example, 0.1 or less (0 ⁇ y ⁇ 0.1), may be 0.09 or less, may be 0.07 or less, or may be 0.05 It may be below. y may be 0.01 or more, or 0.02 or more. Al contributes to stabilization of the crystal structure of the composite oxide NMA. Moreover, it is preferable to satisfy 0.05 ⁇ x2+y ⁇ 0.1. In this case, the effect of compound A and the effect of suppressing an increase in internal resistance after repeated charging and discharging are further realized.
- z which indicates the atomic ratio of the element M, is, for example, 0 ⁇ z ⁇ 0.10, may be 0 ⁇ z ⁇ 0.05, or may be 0.001 ⁇ z ⁇ 0.005.
- the element M may be at least one selected from the group consisting of Ti, Zr, Nb, Mo, W, Fe, Zn, B, Si, Mg, Ca, Sr, Sc and Y.
- the surface structure of the composite oxide NMA is stabilized, the resistance is reduced, and the metal is further eluted. considered to be suppressed. It is more effective when the element M is unevenly distributed near the particle surfaces of the composite oxide NMA.
- the content of the elements constituting the composite oxide NMA can be measured using an inductively coupled plasma atomic emission spectroscopy (ICP-AES), an electron probe microanalyzer (EPMA), or an energy dispersive type It can be measured by an X-ray analyzer (Energy dispersive X-ray spectroscopy: EDX) or the like.
- ICP-AES inductively coupled plasma atomic emission spectroscopy
- EPMA electron probe microanalyzer
- EDX X-ray analyzer
- Composite oxide NMA is, for example, secondary particles in which multiple primary particles are aggregated.
- the particle size of the primary particles is generally 0.05 ⁇ m or more and 1 ⁇ m or less.
- the average particle size of the secondary particles of the composite oxide is, for example, 3 ⁇ m or more and 30 ⁇ m or less, and may be 5 ⁇ m or more and 25 ⁇ m or less.
- the average particle size of secondary particles means the particle size (volume average particle size) at which the volume integrated value is 50% in the particle size distribution measured by the laser diffraction scattering method.
- particle size is sometimes referred to as D50.
- LA-750 manufactured by HORIBA, Ltd. can be used as the measuring device.
- Composite oxide NMA can be obtained, for example, by the following procedure. First, a solution containing an alkali such as sodium hydroxide is added dropwise to a solution of a salt containing a metal element constituting the composite oxide NMA under stirring, and the pH is adjusted to the alkaline side (eg, 8.5 to 12.5). , a composite hydroxide containing metal elements (Ni, Mn, Al, optionally Co, and optionally element M) is precipitated. Subsequently, by calcining the composite hydroxide, a composite oxide containing a metal element (hereinafter also referred to as "raw material composite oxide”) is obtained. The firing temperature at this time is not particularly limited, but is, for example, 300°C to 600°C.
- the firing temperature at this time is not particularly limited, but is, for example, 300°C to 600°C.
- the composite oxide NMA can be obtained by mixing the raw material composite oxide, the lithium compound, and, if necessary, a compound containing the element M, and firing the mixture in an oxygen stream.
- the firing temperature at this time is not particularly limited, but is, for example, 450° C. or higher and 800° C. or lower. Each firing may be performed in one step, in multiple steps, or while raising the temperature.
- lithium compound lithium oxide, lithium hydroxide, lithium carbonate, etc.
- the element M By mixing a compound containing the element M when mixing the raw material composite oxide and the lithium compound, the element M can be unevenly distributed in the vicinity of the particle surface of the composite oxide NMA.
- the positive electrode active material can contain a lithium transition metal composite oxide other than the composite oxide NMA, but it is preferable that the ratio of the composite oxide NMA is large.
- the ratio of the composite oxide NMA in the positive electrode active material is, for example, 90% by mass or more, and may be 95% by mass or more.
- the ratio of the composite oxide in the positive electrode active material is 100% by mass or less.
- a resin material is used as the binder.
- binders include fluororesins, polyolefin resins, polyamide resins, polyimide resins, acrylic resins, vinyl resins, and rubber-like materials (eg, styrene-butadiene copolymer (SBR)).
- SBR styrene-butadiene copolymer
- thickeners examples include cellulose derivatives such as cellulose ethers. Examples of cellulose derivatives include carboxymethyl cellulose (CMC) and modified products thereof, methyl cellulose, and the like. A thickener may be used individually by 1 type, and may be used in combination of 2 or more type.
- CMC carboxymethyl cellulose
- Examples of conductive agents include conductive fibers and conductive particles.
- Examples of conductive fibers include carbon fibers, carbon nanotubes, and metal fibers.
- Conductive particles include conductive carbon (carbon black, graphite, etc.), metal powder, and the like. Conductive agents may be used singly or in combination of two or more.
- the dispersion medium used for the positive electrode slurry is not particularly limited, but examples include water, alcohol, N-methyl-2-pyrrolidone (NMP), and mixed solvents thereof.
- a metal foil can be used as the positive electrode current collector.
- the positive electrode current collector may be porous. Examples of porous current collectors include nets, punched sheets, expanded metals, and the like. Examples of the material of the positive electrode current collector include stainless steel, aluminum, aluminum alloys, and titanium.
- the thickness of the positive electrode current collector is not particularly limited, but is, for example, 1 to 50 ⁇ m, and may be 5 to 30 ⁇ m.
- the negative electrode includes at least a negative electrode current collector and may include a negative electrode active material.
- a negative electrode generally includes a negative electrode current collector and a layered negative electrode mixture (hereinafter referred to as a negative electrode mixture layer) held by the negative electrode current collector.
- the negative electrode mixture layer can be formed by coating the surface of the negative electrode current collector with a negative electrode slurry in which the components of the negative electrode mixture are dispersed in a dispersion medium, and drying the slurry. The dried coating film may be rolled if necessary.
- the negative electrode mixture contains a negative electrode active material as an essential component, and may contain a binder, a thickener, a conductive agent, etc. as optional components.
- the negative electrode active material metallic lithium, a lithium alloy, or the like may be used, but a material capable of electrochemically intercalating and deintercalating lithium ions is preferably used. Examples of such materials include carbonaceous materials and Si-containing materials.
- the negative electrode may contain one type of negative electrode active material, or may contain two or more types in combination.
- carbonaceous materials examples include graphite, graphitizable carbon (soft carbon), and non-graphitizable carbon (hard carbon).
- soft carbon graphitizable carbon
- hard carbon non-graphitizable carbon
- graphite is preferable as the carbonaceous material because of its excellent charge-discharge stability and low irreversible capacity.
- examples of graphite include natural graphite, artificial graphite, and graphitized mesophase carbon particles.
- Si-containing materials include simple Si, silicon alloys, silicon compounds (such as silicon oxides), and composite materials in which a silicon phase is dispersed in a lithium ion conductive phase (matrix).
- Silicon oxides include SiOx particles. x is, for example, 0.5 ⁇ x ⁇ 2, and may be 0.8 ⁇ x ⁇ 1.6. At least one selected from the group consisting of SiO 2 phase, silicate phase and carbon phase can be used as the lithium ion conductive phase.
- binder thickener, conductive agent, and dispersion medium used in the negative electrode slurry
- the materials exemplified for the positive electrode can be used.
- a metal foil can be used as the negative electrode current collector.
- the negative electrode current collector may be porous. Examples of materials for the negative electrode current collector include stainless steel, nickel, nickel alloys, copper, copper alloys, and the like.
- the thickness of the negative electrode current collector is not particularly limited, but is, for example, 1 to 50 ⁇ m, and may be 5 to 30 ⁇ m.
- Non-aqueous electrolyte A non-aqueous electrolyte usually contains a non-aqueous solvent, a lithium salt, and an additive.
- the non-aqueous electrolyte contains the above compound A as an additive.
- the content of compound A in the non-aqueous electrolyte may be 2% by mass or less, 1.5% by mass or less, 1% by mass or less, or 0.5% by mass or less.
- the content of compound A is within this range, excessive film formation on the surface of the positive electrode is suppressed, and the effect of suppressing an increase in internal resistance when charging and discharging are repeated can be enhanced.
- the content of Compound A in the non-aqueous electrolyte changes during storage or charge/discharge.
- compound A should remain in the non-aqueous electrolyte collected from the non-aqueous electrolyte secondary battery at a concentration equal to or higher than the detection limit.
- the content of compound A in the non-aqueous electrolyte may be 0.01% by mass or more.
- the content of compound A in the non-aqueous electrolyte used for manufacturing the non-aqueous electrolyte secondary battery may be 0.01% by mass or more, 0.1% by mass or more, or 0.3% by mass or more. may
- the content of compound A in the non-aqueous electrolyte used for manufacturing the non-aqueous electrolyte secondary battery is, for example, 1.5% by mass or less, and may be 1% by mass or less or 0.5% by mass or less. . These lower and upper limits can be combined arbitrarily.
- the nonaqueous electrolyte may further contain the above fluorinated cyclic carbonate as an additive.
- the content of the fluorinated cyclic carbonate in the non-aqueous electrolyte is preferably 1.5% by mass or less, and may be 1% by mass or less or 0.5% by mass or less.
- the content of the fluorinated cyclic carbonate is within this range, excessive film formation on the surface of the positive electrode is suppressed, and the effect of suppressing an increase in internal resistance when charging and discharging are repeated can be enhanced.
- the content of the fluorinated cyclic carbonate in the non-aqueous electrolyte changes during storage or charge/discharge.
- the fluorinated cyclic carbonate remains in the non-aqueous electrolyte collected from the non-aqueous electrolyte secondary battery at a concentration equal to or higher than the detection limit.
- the content of the fluorinated cyclic carbonate in the non-aqueous electrolyte may be 0.01% by mass or more.
- the content of the fluorinated cyclic carbonate in the non-aqueous electrolyte used for manufacturing the non-aqueous electrolyte secondary battery may be 0.01% by mass or more, 0.1% by mass or more, or 0.3% by mass or more. may be The content of the fluorinated cyclic carbonate in the non-aqueous electrolyte used for manufacturing the non-aqueous electrolyte secondary battery is, for example, 1.5% by mass or less, 1% by mass or less, or 0.5% by mass or less. good too. These lower and upper limits can be combined arbitrarily.
- the contents of the compound A and the fluorinated cyclic carbonate in the non-aqueous electrolyte are determined, for example, using gas chromatography under the following conditions.
- the mass ratio of fluorinated cyclic carbonate to compound A may be, for example, 0.5 to 1.5, and 0.8 to 1.2. There may be.
- the mass ratio of both components is within such a range, the composition of the coating formed on the particle surface of the composite oxide NMA is well balanced. That is, a coating film is formed which has excellent ion conductivity and has a large effect of suppressing the elution of metals and an effect of suppressing an increase in internal resistance when charging and discharging are repeated.
- Non-aqueous solvent examples include cyclic carbonates, chain carbonates, cyclic carboxylates, and chain carboxylates.
- Cyclic carbonates include propylene carbonate (PC), ethylene carbonate (EC), and the like.
- Chain carbonates include diethyl carbonate (DEC), ethylmethyl carbonate (EMC), dimethyl carbonate (DMC) and the like.
- Cyclic carboxylic acid esters include ⁇ -butyrolactone (GBL) and ⁇ -valerolactone (GVL).
- Chain carboxylic acid esters include methyl formate, ethyl formate, propyl formate, methyl acetate (MA), ethyl acetate, propyl acetate, methyl propionate, ethyl propionate, and propyl propionate.
- the non-aqueous electrolyte may contain one type of non-aqueous solvent, or may contain two or more types in combination.
- Lithium salts include, for example, LiClO 4 , LiBF 4 , LiPF 6 , LiAlCl 4 , LiSbF 6 , LiSCN, LiCF 3 SO 3 , LiCF 3 CO 2 , LiAsF 6 , LiB 10 Cl 10 , lithium lower aliphatic carboxylate, LiCl , LiBr, LiI, borates, and imide salts.
- Borates include lithium bisoxalate borate, lithium difluorooxalate borate, bis(1,2-benzenediolate(2-)-O,O') lithium borate, bis(2,3-naphthalene diolate (2-)-O,O') lithium borate, bis(2,2'-biphenyldiolate (2-)-O,O') lithium borate, bis(5-fluoro-2-olate- 1-benzenesulfonic acid-O,O') lithium borate and the like.
- the imide salt examples include lithium bisfluorosulfonylimide (LiN(FSO 2 ) 2 ), lithium bistrifluoromethanesulfonimide (LiN(CF 3 SO 2 ) 2 ), lithium trifluoromethanesulfonate nonafluorobutanesulfonate (LiN (CF 3 SO 2 )(C 4 F 9 SO 2 )), lithium bispentafluoroethanesulfonic acid imide (LiN(C 2 F 5 SO 2 ) 2 ), and the like.
- the non-aqueous electrolyte may contain one type of lithium salt, or may contain two or more types in combination.
- the concentration of the lithium salt in the non-aqueous electrolyte is, for example, 0.5 mol/L or more and 2 mol/L or less.
- the nonaqueous electrolyte may further contain, as an additive, at least one selected from the group consisting of vinylene carbonate and vinylethylene carbonate.
- Separator It is desirable to interpose a separator between the positive electrode and the negative electrode.
- the separator has high ion permeability and moderate mechanical strength and insulation.
- a microporous thin film, a woven fabric, a nonwoven fabric, or the like can be used as the separator.
- Polyolefins such as polypropylene and polyethylene are preferable as the material of the separator.
- An example of the structure of a non-aqueous electrolyte secondary battery is a structure in which an electrode group, in which a positive electrode and a negative electrode are wound with a separator interposed therebetween, is accommodated in an exterior body together with a non-aqueous electrolyte.
- an electrode group in which a positive electrode and a negative electrode are wound with a separator interposed therebetween
- a laminated electrode group in which a positive electrode and a negative electrode are laminated with a separator interposed therebetween may be used.
- the shape of the non-aqueous electrolyte secondary battery is not limited, either, and may be, for example, cylindrical, square, coin, button, laminate, or the like.
- the battery includes a prismatic battery case 4 with a bottom, and an electrode group 1 and a non-aqueous electrolyte (not shown) housed in the battery case 4 .
- the electrode group 1 has a long strip-shaped negative electrode, a long strip-shaped positive electrode, and a separator interposed therebetween.
- the negative electrode current collector of the negative electrode is electrically connected to a negative electrode terminal 6 provided on a sealing plate 5 via a negative electrode lead 3 .
- the negative electrode terminal 6 is insulated from the sealing plate 5 by a resin gasket 7 .
- the positive current collector of the positive electrode is electrically connected to the rear surface of the sealing plate 5 via the positive lead 2 . That is, the positive electrode is electrically connected to the battery case 4 which also serves as a positive electrode terminal.
- the peripheral edge of the sealing plate 5 is fitted into the open end of the battery case 4, and the fitted portion is laser-welded.
- the sealing plate 5 has an injection hole for a non-aqueous electrolyte, which is closed by
- a non-aqueous electrolyte secondary battery was produced and evaluated by the following procedure.
- (1) Fabrication of Positive Electrode To 95 parts by mass of positive electrode active material particles, 2.5 parts by mass of acetylene black, 2.5 parts by mass of polyvinylidene fluoride, and an appropriate amount of NMP were added and mixed to obtain a positive electrode slurry. Next, the positive electrode slurry was applied to the surface of the aluminum foil, the coating film was dried, and then rolled to form a positive electrode mixture layer (thickness: 95 ⁇ m, density: 3.6 g/cm 3 ) on both sides of the aluminum foil. , to obtain the positive electrode.
- the positive electrode active material particles were produced by the following procedure.
- An aqueous solution was prepared by dissolving nickel sulfate, aluminum sulfate, and, if necessary, cobalt sulfate or manganese sulfate.
- the concentration of nickel sulfate in the aqueous solution was set to 1 mol/L, and the concentrations of other sulfates were adjusted so that the relationship between the ratios of Ni and each metal element was the value shown in Table 1.
- an aqueous solution containing sodium hydroxide at a concentration of 30% by mass was added dropwise until the pH of the mixture reached 12 to precipitate hydroxide.
- the hydroxide was recovered by filtration, washed with water and dried.
- a composite oxide was obtained by baking the dried product at 500° C. for 8 hours in a nitrogen atmosphere.
- the obtained composite oxide and lithium hydroxide were mixed so that the total atomic ratio of Li and Ni, Co, Mn and Al was 1:1.
- the mixture was fired by heating from room temperature to 650° C. in an oxygen atmosphere at a heating rate of 2.0° C./min using an electric furnace. After that, it was fired by heating from 650° C. to 750° C. at a heating rate of 0.5° C./min.
- Composite oxide NMA positive electrode active material particles
- Negative Electrode A silicon composite material and graphite were mixed at a mass ratio of 5:95 and used as a negative electrode active material.
- the negative electrode slurry was applied to the surface of a copper foil as a negative electrode current collector, the coating film was dried, and then rolled to form negative electrode mixture layers on both sides of the copper foil.
- the voltage value was measured when the battery with an SOC of 50% was discharged for 10 seconds at current values of 0A, 0.1A, 0.5A and 1.0A.
- DCIR initial DCIR was calculated from the absolute value of the slope when the relationship between the discharge current value and the voltage value after 10 seconds was linearly approximated by the method of least squares.
- DCIR increase rate ( ⁇ DCIR) DCIR (DCIR at the 200th cycle) was calculated in the same manner as in (a) above, except that the battery after 200 cycles of charging and discharging in the charge-discharge cycle test (b) above was used. The ratio of the DCIR after 200 cycles to the initial DCIR was calculated as the DCIR increase rate using the following formula.
- DCIR increase rate (%) ⁇ (DCIR at 200th cycle-initial DCIR) / initial DCIR ⁇ x 100
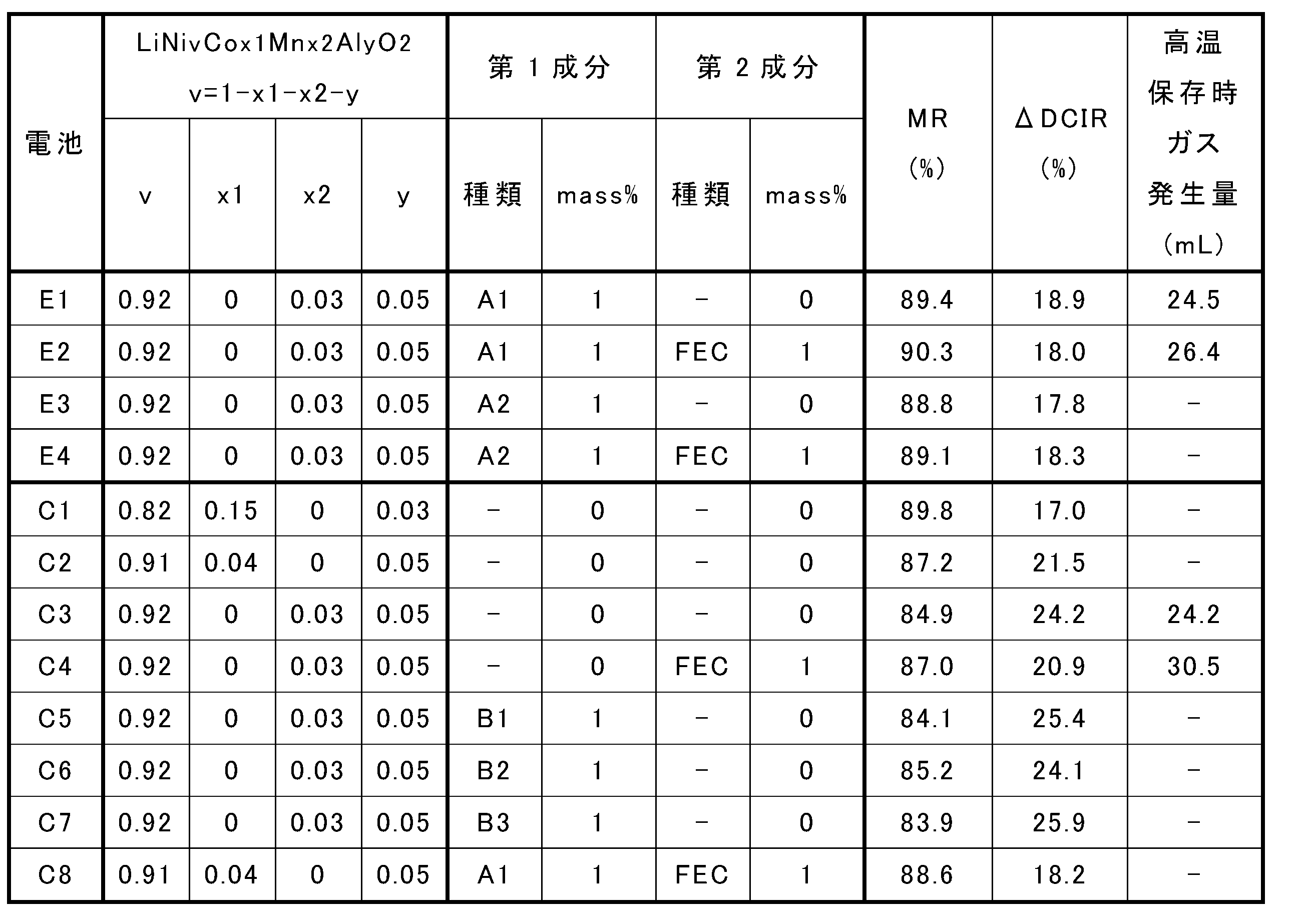
- Table 1 shows the evaluation results.
- A1 is diethyl(difluoromethyl)phosphonate
- A2 is diethyl(trifluoromethyl)phosphonate
- B1 is diethylmethylphosphonate
- B2 is dimethylvinylphosphonate
- B3 is bis(2,2,2-trifluoroethyl)methylphosphonate.
- A1 to A2 are organic phosphorus compounds represented by general formula (1)
- B1 to B3 are not organic phosphorus compounds represented by general formula (1).
- E1 and C3 a composite oxide NMA containing no Co was used as the positive electrode active material, respectively.
- A1 was added to the non-aqueous electrolyte, and in C3, A1 was not added to the non-aqueous electrolyte.
- MR increased significantly by 4.5% (84.9% ⁇ 89.4%), and ⁇ DCIR decreased significantly by 5.3% (24.2% ⁇ 18 .9%).
- C8 and C2 a composite oxide containing a relatively large amount of Co was used as the positive electrode active material.
- A1 was added to the non-aqueous electrolyte, and in C2, A1 was not added to the non-aqueous electrolyte.
- the capacity retention rate (MR) increased by only 1.4% (87.2% ⁇ 88.6%), and the DCIR increase rate ( ⁇ DCIR) was also reduced by only 3.3%. (21.5% ⁇ 18.2%).
- E2 had a further 0.9% increase in MR (89.4% ⁇ 90.3%) and a further 0.9% reduction in ⁇ DCIR (18.9% ⁇ 18.0%) relative to E1. .
- ⁇ DCIR 18.9% ⁇ 18.0%
- E2 and E1 a composite oxide NMA that does not contain Co is used as the positive electrode active material. was not added.
- the amount of gas generated during high-temperature storage increased by only 1.9 mL compared to E1 (24.5 mL ⁇ 26.4 mL), and gas generation due to FEC was greatly suppressed when FEC was added together with A1. rice field.
- R 3 in the general formula (1) is a methyl group.
- R2 in general formula (1) is a vinyl group.
- R 1 and R 2 in general formula (1) are 2,2,2-trifluoroethyl groups, and R 3 is a methyl group.
- a non-aqueous electrolyte secondary battery according to the present disclosure is useful as a main power source for mobile communication devices, portable electronic devices, and the like.
- non-aqueous electrolyte secondary batteries are suitable for in-vehicle use because they have high capacity and excellent cycle characteristics.
- the uses of the non-aqueous electrolyte secondary battery are not limited to these.
Landscapes
- Chemical & Material Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- Engineering & Computer Science (AREA)
- Inorganic Chemistry (AREA)
- Manufacturing & Machinery (AREA)
- Physics & Mathematics (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- General Physics & Mathematics (AREA)
- Materials Engineering (AREA)
- Secondary Cells (AREA)
- Battery Electrode And Active Subsutance (AREA)
Abstract
Description
前記正極は、正極活物質を含み、前記正極活物質は、Niと、Mnと、Alと、を含むリチウム遷移金属複合酸化物を含み、前記リチウム遷移金属複合酸化物に含まれるLi以外の金属元素に占めるNi、MnおよびAlの割合が、それぞれNi:50原子%以上、Mn:10原子%以下、およびAl:10原子%以下であり、前記リチウム遷移金属複合酸化物がCoを含む場合、Li以外の金属元素に占めるCoの割合が1.5原子%以下であり、
前記非水電解質は、一般式(1):
一般式(1)中、R1およびR2は、それぞれ独立して、炭素数1~4のアルキル基である。炭素数1~4のアルキル基は、直鎖状でもよく、分岐鎖状でもよい。炭素数1~4のアルキル基としては、メチル基、エチル基、n-プロピル基、イソプロピル基、n-ブチル基、sec-ブチル基、tert-ブチル基が挙げられる。中でも、メチル基、エチル基が好ましい。
非水電解質は、更に、フッ素化環状カーボネートを含んでもよい。なお、フッ素化環状カーボネートとは、環状カーボネートの水素原子の少なくとも1つがフッ素原子に置換された化合物を意味する。化合物Aとフッ素化環状カーボネートとを併用する場合、化合物Aとフッ素化環状カーボネートとの組み合わせに由来する被膜が形成され、被膜のイオン伝導性が更に高められる。これにより、より優れたサイクル特性を確保できるとともに、内部抵抗の増加が更に抑制される。特に、正極活物質に後述のNMA複合酸化物を用いる場合、化合物Aとフッ素化環状カーボネートとの併用による効果が顕著に得られる。ただし、フッ素化環状カーボネート単独では、被膜のイオン伝導性の面で不利である。
正極は、正極活物質を含む。正極は、通常、正極集電体と、正極集電体に保持された層状の正極合剤(以下、正極合剤層と称する)を備えている。正極合剤層は、正極合剤の構成成分を分散媒に分散させた正極スラリを、正極集電体の表面に塗布し、乾燥させることにより形成できる。乾燥後の塗膜を、必要により圧延してもよい。
正極活物質は、複合酸化物NMAを含む。複合酸化物NMAは、NiとMnとAlとを含み、微量のCoを含んでもよく、Coを含まなくてもよい。製造コスト削減の観点からは、Co含有量は少ないほど望ましく、Li以外の金属元素に占めるCoの割合は1.5原子%以下であり、1.0原子%以下が望ましく、0.5原子%以下がより望ましく、Coを含まないことが最も望ましい。一方、高容量化の観点から、複合酸化物NMAにおいて、Li以外の金属元素に占めるNi、MnおよびAlの割合は、それぞれNi:50原子%以上、Mn:10原子%以下、およびAl:10原子%以下である。Li以外の金属元素に占めるNi含有量は80原子%以上が望ましく、90原子%以上がより望ましく、92原子%以上でもよい。Mn含有量は、7原子%以下でもよく、5原子%以下でもよく、3原子%以下でもよい。Al含有量は、9原子%以下でもよく、7原子%以下でもよく、5原子%以下でもよい。複合酸化物NMAは、例えば、層状の結晶構造(例えば、岩塩型結晶構造)を有する。
結着剤としては、例えば、樹脂材料が用いられる。結着剤としては、例えば、フッ素樹脂、ポリオレフィン樹脂、ポリアミド樹脂、ポリイミド樹脂、アクリル樹脂、ビニル樹脂、ゴム状材料(例えばスチレンブタジエン共重合体(SBR))等が挙げられる。結着剤は、1種を単独で用いてもよく、2種以上を組み合わせて用いてもよい。
負極は、少なくとも負極集電体を含み、負極活物質を含んでもよい。負極は、通常、負極集電体と、負極集電体に保持された層状の負極合剤(以下、負極合剤層と称する)を備えている。負極合剤層は、負極合剤の構成成分を分散媒に分散させた負極スラリを、負極集電体の表面に塗布し、乾燥させることにより形成できる。乾燥後の塗膜を、必要により圧延してもよい。
負極活物質としては、金属リチウム、リチウム合金等を用いてもよいが、電気化学的にリチウムイオンを吸蔵および放出可能な材料が好適に用いられる。このような材料としては、炭素質材料、Si含有材料等が挙げられる。負極は、負極活物質を1種含んでいてもよく、2種以上組み合わせて含んでもよい。
結着剤、増粘剤および導電剤ならびに負極スラリに用いる分散媒としては、例えば、正極で例示した材料を用い得る。
非水電解質は、通常、非水溶媒とリチウム塩と添加剤とを含む。
使用機器:(株)島津製作所製、GC-2010 Plus
カラム:J&W社製、HP-1(膜厚1μm、内径0.32mm、長さ60m)
カラム温度:50℃から昇温速度5℃/minで90℃に昇温し、90℃で15分維持し、次いで、90℃から250℃に昇温速度10℃/minで昇温し、250℃で15分維持
スプリット比:1/50
線速度:30.0cm/sec
注入口温度:270℃
注入量:1μL
検出器:FID 290℃(sens.101)
非水溶媒としては、例えば、環状炭酸エステル、鎖状炭酸エステル、環状カルボン酸エステル、鎖状カルボン酸エステルが挙げられる。環状炭酸エステルとしては、プロピレンカーボネート(PC)、エチレンカーボネート(EC)等が挙げられる。鎖状炭酸エステルとしては、ジエチルカーボネート(DEC)、エチルメチルカーボネート(EMC)、ジメチルカーボネート(DMC)等が挙げられる。環状カルボン酸エステルとしては、γ-ブチロラクトン(GBL)、γ-バレロラクトン(GVL)等が挙げられる。鎖状カルボン酸エステルとしては、ギ酸メチル、ギ酸エチル、ギ酸プロピル、酢酸メチル(MA)、酢酸エチル、酢酸プロピル、プロピオン酸メチル、プロピオン酸エチル、プロピオン酸プロピル等が挙げられる。非水電解質は、非水溶媒を1種含んでもよく、2種以上組み合わせて含んでもよい。
リチウム塩としては、例えば、LiClO4、LiBF4、LiPF6、LiAlCl4、LiSbF6、LiSCN、LiCF3SO3、LiCF3CO2、LiAsF6、LiB10Cl10、低級脂肪族カルボン酸リチウム、LiCl、LiBr、LiI、ホウ酸塩、イミド塩が挙げられる。ホウ酸塩としては、ビスオキサレートホウ酸リチウム、ジフルオロオキサレートホウ酸リチウム、ビス(1,2-ベンゼンジオレート(2-)-O,O’)ホウ酸リチウム、ビス(2,3-ナフタレンジオレート(2-)-O,O’)ホウ酸リチウム、ビス(2,2’-ビフェニルジオレート(2-)-O,O’)ホウ酸リチウム、ビス(5-フルオロ-2-オレート-1-ベンゼンスルホン酸-O,O’)ホウ酸リチウム等が挙げられる。イミド塩としては、ビスフルオロスルホニルイミドリチウム(LiN(FSO2)2)、ビストリフルオロメタンスルホン酸イミドリチウム(LiN(CF3SO2)2)、トリフルオロメタンスルホン酸ノナフルオロブタンスルホン酸イミドリチウム(LiN(CF3SO2)(C4F9SO2))、ビスペンタフルオロエタンスルホン酸イミドリチウム(LiN(C2F5SO2)2)等が挙げられる。非水電解質は、リチウム塩を、1種含んでもよく、2種以上組み合わせて含んでもよい。
正極と負極との間には、セパレータを介在させることが望ましい。セパレータは、イオン透過度が高く、適度な機械的強度および絶縁性を備えている。セパレータとしては、微多孔薄膜、織布、不織布等を用いることができる。セパレータの材質としては、ポリプロピレン、ポリエチレン等のポリオレフィンが好ましい。
下記の手順で、非水電解質二次電池を作製し、評価を行った。
(1)正極の作製
正極活物質粒子95質量部に、アセチレンブラック2.5質量部と、ポリフッ化ビニリデン2.5質量部と、適量のNMPとを加え、混合し、正極スラリを得た。次に、アルミニウム箔の表面に正極スラリを塗布し、塗膜を乾燥させた後、圧延して、アルミニウム箔の両面に正極合剤層(厚み95μm、密度3.6g/cm3)を形成し、正極を得た。
硫酸ニッケル、硫酸アルミニウム、および、必要に応じて、硫酸コバルトまたは硫酸マンガンを溶解させた水溶液を調製した。水溶液中の硫酸ニッケルの濃度を1mol/Lとし、他の硫酸塩の濃度については、Niと各金属元素との比率の関係が表1に示す値となるように調節した。
ケイ素複合材料と黒鉛とを5:95の質量比で混合し、負極活物質として用いた。負極活物質と、CMCのナトリウム塩(CMC-Na)と、SBRと、水とを所定の質量比で混合し、負極スラリを調製した。次に、負極集電体である銅箔の表面に負極スラリを塗布し、塗膜を乾燥させた後、圧延して、銅箔の両面に負極合剤層を形成した。
ECおよびEMCとの混合溶媒(EC:EMC=3:7(体積比))に、LiPF6および必要に応じて表1に示す有機リン化合物(第1成分)およびFEC(第2成分)を溶解させることにより、非水電解質(電解液)を調製した。電解液におけるLiPF6の濃度は1.0mol/Lとした。調製した非水電解質中の第1成分および第2成分の濃度(初期濃度)は、表1中に示す値(質量%)とした。
上記で得られた正極にAl製の正極リードを取り付け、上記で得られた負極にNi製の負極リードを取り付けた。不活性ガス雰囲気中で、正極と負極とをポリエチレン薄膜(セパレータ)を介して渦巻状に捲回し、捲回型の電極群を作製した。電極群を、Al層を備えるラミネートシートで形成される袋状の外装体に収容し、上記非水電解質を注入した後、外装体を封止して非水電解質二次電池を作製した。なお、電極群を外装体に収容する際、正極リードおよび負極リードの一部は、それぞれ、外装体より外部に露出させた。表1中、E1~E4は、実施例1~4であり、C1~C8は、比較例1~8である。
実施例および比較例で得られた非水電解質二次電池について、下記の評価を行った。
(a)初期の直流抵抗値(DCIR)
25℃の環境下において、電池を0.3Itの電流で電圧が4.2Vになるまで定電流充電し、その後、4.2Vの定電圧で電流が0.05Itになるまで定電圧充電した。次いで、0.3Itの定電流で100分間放電し、充電状態(State of charge:SOC)を50%にした。
25℃の環境下において、電池を0.5Itの定電流で電圧が4.2Vになるまで定電流充電し、その後、4.2Vの定電圧で電流が0.02Itになるまで定電圧充電した。次いで、0.5Itの定電流で電圧が3.0Vになるまで定電流放電を行った。この充電および放電を1サイクルとして、200サイクル繰り返した。
上記(b)の充放電サイクル試験で充放電を200サイクル繰り返した後の電池を用いる以外は、上記(a)の場合と同様にして、DCIR(200サイクル目のDCIR)を算出した。初期のDCIRに対する200サイクル後のDCIRの比率をDCIR上昇率として、以下の式により算出した。
DCIR上昇率(%)={(200サイクル目のDCIR-初期のDCIR)/初期のDCIR}×100
上記(b)の充放電サイクル試験において、1サイクル目の放電容量と200サイクル目の放電容量とを測定し、以下の式により容量維持率を求め、サイクル特性の指標とした。
容量維持率(%)=(200サイクル目の放電容量/1サイクル目の放電容量)×100
更に、E1、E2、C3、C4の電池について、以下の高温保存特性の評価を行った。
25℃の環境下において、電池を0.3Itの定電流で電圧が4.2Vになるまで定電流充電し、その後、4.2Vの定電圧で電流が0.02Itになるまで定電圧充電した。充電後の電池を80℃の環境下で72時間保存し、保存時の電池内のガス発生量を調べた。
A1~A2は、一般式(1)で表される有機リン化合物であり、B1~B3は、一般式(1)で表される有機リン化合物ではない。
Claims (7)
- 正極と、負極と、非水電解質と、を備え、
前記正極は、正極活物質を含み、
前記正極活物質は、Niと、Mnと、Alと、を含むリチウム遷移金属複合酸化物を含み、
前記リチウム遷移金属複合酸化物に含まれるLi以外の金属元素に占めるNi、MnおよびAlの割合が、それぞれ
Ni:50原子%以上、
Mn:10原子%以下、および
Al:10原子%以下
であり、
前記リチウム遷移金属複合酸化物がCoを含む場合、Li以外の金属元素に占めるCoの割合が1.5原子%以下であり、
前記非水電解質は、一般式(1):
R1およびR2は、それぞれ独立して、炭素数1~4のアルキル基であり、
R3は、炭素数1~4のフッ素化アルキル基である、非水電解質二次電池。 - 前記有機リン化合物は、ジエチル(フルオロメチル)ホスホネート、ジエチル(ジフルオロメチル)ホスホネートおよびジエチル(トリフルオロメチル)ホスホネートからなる群より選択された少なくとも1種を含む、請求項1に記載の非水電解質二次電池。
- 前記非水電解質中の前記有機リン化合物の含有量は、2質量%以下である、請求項1または2に記載の非水電解質二次電池。
- 更に、フッ素化環状カーボネートを含む、請求項1~3のいずれか1項に記載の非水電解質二次電池。
- 前記フッ素化環状カーボネートは、フルオロエチレンカーボネートを含む、請求項4に記載の非水電解質二次電池。
- 前記リチウム遷移金属複合酸化物は、下記式:
LiαNi(1-x1-x2-y-z)Cox1Mnx2AlyMzO2+β
で表され、式中、
0.95≦α≦1.05、
0.685≦1-x1-x2-y-z≦0.95、
0≦x1≦0.015、
0<x2≦0.1、
0<y≦0.1、
0≦z≦0.1、および
-0.05≦β≦0.05を満たし、
Mは、Li、Ni、Mn、Al、Coおよび酸素以外の元素である、請求項1~5のいずれか1項に記載の非水電解質二次電池。 - 前記元素Mが、Ti、Zr、Nb、Mo、W、Fe、Zn、B、Si、Mg、Ca、Sr、ScおよびYからなる群より選択された少なくとも1種である、請求項6に記載の非水電解質二次電池。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023531722A JPWO2023276526A1 (ja) | 2021-06-30 | 2022-05-31 | |
| CN202280045935.4A CN117597809A (zh) | 2021-06-30 | 2022-05-31 | 非水电解质二次电池 |
| EP22832684.9A EP4366014A1 (en) | 2021-06-30 | 2022-05-31 | Nonaqueous electrolyte secondary battery |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021109544 | 2021-06-30 | ||
| JP2021-109544 | 2021-06-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023276526A1 true WO2023276526A1 (ja) | 2023-01-05 |
Family
ID=84691222
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/022080 WO2023276526A1 (ja) | 2021-06-30 | 2022-05-31 | 非水電解質二次電池 |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP4366014A1 (ja) |
| JP (1) | JPWO2023276526A1 (ja) |
| CN (1) | CN117597809A (ja) |
| WO (1) | WO2023276526A1 (ja) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006310181A (ja) | 2005-04-28 | 2006-11-09 | Matsushita Electric Ind Co Ltd | 非水電解液二次電池 |
| WO2011030686A1 (ja) * | 2009-09-09 | 2011-03-17 | 日本電気株式会社 | 二次電池 |
| WO2013024717A1 (ja) * | 2011-08-12 | 2013-02-21 | 宇部興産株式会社 | 非水電解液及びそれを用いた電気化学素子 |
| JP2014194930A (ja) | 2013-02-27 | 2014-10-09 | Mitsubishi Chemicals Corp | 非水系電解液及びそれを用いた非水系電解液電池 |
| WO2016031316A1 (ja) * | 2014-08-25 | 2016-03-03 | 宇部興産株式会社 | 非水電解液、それを用いた蓄電デバイス、及びそれに用いるリン化合物 |
-
2022
- 2022-05-31 CN CN202280045935.4A patent/CN117597809A/zh active Pending
- 2022-05-31 JP JP2023531722A patent/JPWO2023276526A1/ja active Pending
- 2022-05-31 WO PCT/JP2022/022080 patent/WO2023276526A1/ja active Application Filing
- 2022-05-31 EP EP22832684.9A patent/EP4366014A1/en active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006310181A (ja) | 2005-04-28 | 2006-11-09 | Matsushita Electric Ind Co Ltd | 非水電解液二次電池 |
| WO2011030686A1 (ja) * | 2009-09-09 | 2011-03-17 | 日本電気株式会社 | 二次電池 |
| WO2013024717A1 (ja) * | 2011-08-12 | 2013-02-21 | 宇部興産株式会社 | 非水電解液及びそれを用いた電気化学素子 |
| JP2014194930A (ja) | 2013-02-27 | 2014-10-09 | Mitsubishi Chemicals Corp | 非水系電解液及びそれを用いた非水系電解液電池 |
| WO2016031316A1 (ja) * | 2014-08-25 | 2016-03-03 | 宇部興産株式会社 | 非水電解液、それを用いた蓄電デバイス、及びそれに用いるリン化合物 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4366014A1 (en) | 2024-05-08 |
| JPWO2023276526A1 (ja) | 2023-01-05 |
| CN117597809A (zh) | 2024-02-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2498323B1 (en) | Positive active material, and electrode and lithium battery containing the material | |
| JP6588079B2 (ja) | 非水電解質二次電池 | |
| KR20190131842A (ko) | 이차전지용 양극 활물질, 그 제조방법 및 이를 포함하는 리튬 이차전지 | |
| KR20190059115A (ko) | 리튬 이차전지용 양극재에 포함되는 비가역 첨가제, 이의 제조방법, 및 이 및 포함하는 양극재 | |
| WO2013145846A1 (ja) | 非水電解質二次電池用正極及び非水電解質二次電池 | |
| JP2007188878A (ja) | リチウムイオン二次電池 | |
| JP2011070789A (ja) | 非水電解質二次電池 | |
| KR102595884B1 (ko) | 이차전지용 양극 활물질 전구체, 양극 활물질 및 이를 포함하는 리튬 이차전지 | |
| WO2020026486A1 (ja) | 正極材料および二次電池 | |
| JP7165913B2 (ja) | 非水電解質二次電池 | |
| JPWO2019167581A1 (ja) | 非水電解質二次電池 | |
| JP2022501789A (ja) | 二次電池用正極活物質、その製造方法及びそれを含むリチウム二次電池 | |
| US9847551B2 (en) | Lithium-ion secondary battery | |
| JP7357994B2 (ja) | 二次電池用正極活物質の製造方法 | |
| WO2023276527A1 (ja) | 非水電解質二次電池 | |
| KR20220062974A (ko) | 리튬 이차 전지용 양극 활물질 및 이를 포함하는 리튬 이차 전지 | |
| WO2018096889A1 (ja) | 非水電解液、及びリチウムイオン二次電池 | |
| KR20160023423A (ko) | 리튬이차전지 | |
| JP7458033B2 (ja) | 非水電解質二次電池およびこれに用いる電解液 | |
| WO2023276526A1 (ja) | 非水電解質二次電池 | |
| WO2022091939A1 (ja) | 非水電解質二次電池 | |
| WO2021235131A1 (ja) | 非水電解質二次電池 | |
| KR102567705B1 (ko) | 리튬 이차 전지 | |
| JP7278652B2 (ja) | 二次電池用正極活物質、その製造方法およびこれを含むリチウム二次電池 | |
| WO2023032799A1 (ja) | 非水電解質電池およびこれに用いる非水電解質 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22832684 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023531722 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18569130 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202280045935.4 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022832684 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2022832684 Country of ref document: EP Effective date: 20240130 |