WO2023112209A1 - 水溶性組成物及びその製造方法 - Google Patents
水溶性組成物及びその製造方法 Download PDFInfo
- Publication number
- WO2023112209A1 WO2023112209A1 PCT/JP2021/046291 JP2021046291W WO2023112209A1 WO 2023112209 A1 WO2023112209 A1 WO 2023112209A1 JP 2021046291 W JP2021046291 W JP 2021046291W WO 2023112209 A1 WO2023112209 A1 WO 2023112209A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- magnesium
- water
- sodium
- lactoferrin
- soluble composition
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 78
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 10
- 239000011777 magnesium Substances 0.000 claims abstract description 96
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 claims abstract description 83
- 229910052749 magnesium Inorganic materials 0.000 claims abstract description 83
- 239000011734 sodium Substances 0.000 claims abstract description 57
- 235000021242 lactoferrin Nutrition 0.000 claims abstract description 49
- 102000010445 Lactoferrin Human genes 0.000 claims abstract description 47
- 108010063045 Lactoferrin Proteins 0.000 claims abstract description 47
- CSSYQJWUGATIHM-IKGCZBKSSA-N l-phenylalanyl-l-lysyl-l-cysteinyl-l-arginyl-l-arginyl-l-tryptophyl-l-glutaminyl-l-tryptophyl-l-arginyl-l-methionyl-l-lysyl-l-lysyl-l-leucylglycyl-l-alanyl-l-prolyl-l-seryl-l-isoleucyl-l-threonyl-l-cysteinyl-l-valyl-l-arginyl-l-arginyl-l-alanyl-l-phenylal Chemical compound C([C@H](N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(O)=O)C1=CC=CC=C1 CSSYQJWUGATIHM-IKGCZBKSSA-N 0.000 claims abstract description 47
- 229940078795 lactoferrin Drugs 0.000 claims abstract description 47
- 229910052708 sodium Inorganic materials 0.000 claims abstract description 44
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 claims abstract description 43
- 235000018102 proteins Nutrition 0.000 claims abstract description 7
- 102000004169 proteins and genes Human genes 0.000 claims abstract description 7
- 108090000623 proteins and genes Proteins 0.000 claims abstract description 7
- 239000000243 solution Substances 0.000 claims description 28
- 239000011259 mixed solution Substances 0.000 claims description 6
- 238000002156 mixing Methods 0.000 claims description 6
- 230000001413 cellular effect Effects 0.000 abstract description 11
- 239000000523 sample Substances 0.000 description 52
- 241000894006 Bacteria Species 0.000 description 27
- 238000012360 testing method Methods 0.000 description 24
- 238000011156 evaluation Methods 0.000 description 23
- 230000006872 improvement Effects 0.000 description 23
- 208000005888 Periodontal Pocket Diseases 0.000 description 21
- 239000000606 toothpaste Substances 0.000 description 21
- 229940034610 toothpaste Drugs 0.000 description 20
- 230000000694 effects Effects 0.000 description 19
- 208000028169 periodontal disease Diseases 0.000 description 16
- 208000032843 Hemorrhage Diseases 0.000 description 14
- 208000034158 bleeding Diseases 0.000 description 14
- 230000000740 bleeding effect Effects 0.000 description 14
- 238000007689 inspection Methods 0.000 description 14
- 210000000214 mouth Anatomy 0.000 description 13
- -1 pH adjusters Substances 0.000 description 11
- 206010061218 Inflammation Diseases 0.000 description 10
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 10
- 230000004054 inflammatory process Effects 0.000 description 10
- 238000002360 preparation method Methods 0.000 description 10
- 208000024891 symptom Diseases 0.000 description 10
- 239000003921 oil Substances 0.000 description 9
- 235000019198 oils Nutrition 0.000 description 9
- 230000000052 comparative effect Effects 0.000 description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 8
- 241001131796 Botaurus stellaris Species 0.000 description 7
- 230000001680 brushing effect Effects 0.000 description 7
- 230000008859 change Effects 0.000 description 6
- 239000000551 dentifrice Substances 0.000 description 6
- 235000014113 dietary fatty acids Nutrition 0.000 description 6
- 239000000194 fatty acid Substances 0.000 description 6
- 229930195729 fatty acid Natural products 0.000 description 6
- 239000000796 flavoring agent Substances 0.000 description 6
- 238000005187 foaming Methods 0.000 description 6
- 239000004615 ingredient Substances 0.000 description 6
- 210000003491 skin Anatomy 0.000 description 6
- 210000001035 gastrointestinal tract Anatomy 0.000 description 5
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 5
- 238000005259 measurement Methods 0.000 description 5
- 238000000034 method Methods 0.000 description 5
- 239000011780 sodium chloride Substances 0.000 description 5
- 206010010774 Constipation Diseases 0.000 description 4
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 4
- 208000002193 Pain Diseases 0.000 description 4
- 239000003082 abrasive agent Substances 0.000 description 4
- 239000004480 active ingredient Substances 0.000 description 4
- 239000004088 foaming agent Substances 0.000 description 4
- 239000011521 glass Substances 0.000 description 4
- 230000004899 motility Effects 0.000 description 4
- 210000004400 mucous membrane Anatomy 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- 206010006326 Breath odour Diseases 0.000 description 3
- 229920000858 Cyclodextrin Polymers 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 230000001580 bacterial effect Effects 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 239000006196 drop Substances 0.000 description 3
- 235000019634 flavors Nutrition 0.000 description 3
- 235000013355 food flavoring agent Nutrition 0.000 description 3
- 235000003599 food sweetener Nutrition 0.000 description 3
- 239000004310 lactic acid Substances 0.000 description 3
- 235000014655 lactic acid Nutrition 0.000 description 3
- 230000003239 periodontal effect Effects 0.000 description 3
- 239000008213 purified water Substances 0.000 description 3
- 239000013535 sea water Substances 0.000 description 3
- 239000003765 sweetening agent Substances 0.000 description 3
- 239000002562 thickening agent Substances 0.000 description 3
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 2
- MPDGHEJMBKOTSU-YKLVYJNSSA-N 18beta-glycyrrhetic acid Chemical compound C([C@H]1C2=CC(=O)[C@H]34)[C@@](C)(C(O)=O)CC[C@]1(C)CC[C@@]2(C)[C@]4(C)CC[C@@H]1[C@]3(C)CC[C@H](O)C1(C)C MPDGHEJMBKOTSU-YKLVYJNSSA-N 0.000 description 2
- 241000186000 Bifidobacterium Species 0.000 description 2
- QFOHBWFCKVYLES-UHFFFAOYSA-N Butylparaben Chemical compound CCCCOC(=O)C1=CC=C(O)C=C1 QFOHBWFCKVYLES-UHFFFAOYSA-N 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- 235000014653 Carica parviflora Nutrition 0.000 description 2
- 244000132059 Carica parviflora Species 0.000 description 2
- 241000178435 Eliokarmos dubius Species 0.000 description 2
- 241000186660 Lactobacillus Species 0.000 description 2
- 235000011203 Origanum Nutrition 0.000 description 2
- 241000282320 Panthera leo Species 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 229910021536 Zeolite Inorganic materials 0.000 description 2
- POJWUDADGALRAB-UHFFFAOYSA-N allantoin Chemical compound NC(=O)NC1NC(=O)NC1=O POJWUDADGALRAB-UHFFFAOYSA-N 0.000 description 2
- 230000000844 anti-bacterial effect Effects 0.000 description 2
- 230000003110 anti-inflammatory effect Effects 0.000 description 2
- 230000002421 anti-septic effect Effects 0.000 description 2
- CUFNKYGDVFVPHO-UHFFFAOYSA-N azulene Chemical compound C1=CC=CC2=CC=CC2=C1 CUFNKYGDVFVPHO-UHFFFAOYSA-N 0.000 description 2
- 230000003385 bacteriostatic effect Effects 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- 239000006059 cover glass Substances 0.000 description 2
- 229940097362 cyclodextrins Drugs 0.000 description 2
- RBLGLDWTCZMLRW-UHFFFAOYSA-K dicalcium;phosphate;dihydrate Chemical compound O.O.[Ca+2].[Ca+2].[O-]P([O-])([O-])=O RBLGLDWTCZMLRW-UHFFFAOYSA-K 0.000 description 2
- 238000007865 diluting Methods 0.000 description 2
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 210000000613 ear canal Anatomy 0.000 description 2
- 239000000284 extract Substances 0.000 description 2
- 239000003889 eye drop Substances 0.000 description 2
- 229940012356 eye drops Drugs 0.000 description 2
- 235000011187 glycerol Nutrition 0.000 description 2
- 208000014617 hemorrhoid Diseases 0.000 description 2
- 239000003906 humectant Substances 0.000 description 2
- 229940039696 lactobacillus Drugs 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 229910001629 magnesium chloride Inorganic materials 0.000 description 2
- 229910001425 magnesium ion Inorganic materials 0.000 description 2
- 239000001525 mentha piperita l. herb oil Substances 0.000 description 2
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 2
- 238000001000 micrograph Methods 0.000 description 2
- 235000015097 nutrients Nutrition 0.000 description 2
- 235000019477 peppermint oil Nutrition 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- 201000009890 sinusitis Diseases 0.000 description 2
- 208000003265 stomatitis Diseases 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 2
- 239000010457 zeolite Substances 0.000 description 2
- FTLYMKDSHNWQKD-UHFFFAOYSA-N (2,4,5-trichlorophenyl)boronic acid Chemical compound OB(O)C1=CC(Cl)=C(Cl)C=C1Cl FTLYMKDSHNWQKD-UHFFFAOYSA-N 0.000 description 1
- QYIXCDOBOSTCEI-QCYZZNICSA-N (5alpha)-cholestan-3beta-ol Chemical compound C([C@@H]1CC2)[C@@H](O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@H](C)CCCC(C)C)[C@@]2(C)CC1 QYIXCDOBOSTCEI-QCYZZNICSA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- 229940058015 1,3-butylene glycol Drugs 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- QCDWFXQBSFUVSP-UHFFFAOYSA-N 2-phenoxyethanol Chemical compound OCCOC1=CC=CC=C1 QCDWFXQBSFUVSP-UHFFFAOYSA-N 0.000 description 1
- SLXKOJJOQWFEFD-UHFFFAOYSA-N 6-aminohexanoic acid Chemical compound NCCCCCC(O)=O SLXKOJJOQWFEFD-UHFFFAOYSA-N 0.000 description 1
- POJWUDADGALRAB-PVQJCKRUSA-N Allantoin Natural products NC(=O)N[C@@H]1NC(=O)NC1=O POJWUDADGALRAB-PVQJCKRUSA-N 0.000 description 1
- 244000061520 Angelica archangelica Species 0.000 description 1
- 244000063299 Bacillus subtilis Species 0.000 description 1
- 235000014469 Bacillus subtilis Nutrition 0.000 description 1
- 208000035143 Bacterial infection Diseases 0.000 description 1
- 241000186018 Bifidobacterium adolescentis Species 0.000 description 1
- 241000901050 Bifidobacterium animalis subsp. lactis Species 0.000 description 1
- 241000186016 Bifidobacterium bifidum Species 0.000 description 1
- 241000186012 Bifidobacterium breve Species 0.000 description 1
- 241001608472 Bifidobacterium longum Species 0.000 description 1
- 241000186015 Bifidobacterium longum subsp. infantis Species 0.000 description 1
- 241000186148 Bifidobacterium pseudolongum Species 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 235000005747 Carum carvi Nutrition 0.000 description 1
- 240000000467 Carum carvi Species 0.000 description 1
- 244000037364 Cinnamomum aromaticum Species 0.000 description 1
- 235000014489 Cinnamomum aromaticum Nutrition 0.000 description 1
- 241000951471 Citrus junos Species 0.000 description 1
- 235000019499 Citrus oil Nutrition 0.000 description 1
- 206010009900 Colitis ulcerative Diseases 0.000 description 1
- 208000035473 Communicable disease Diseases 0.000 description 1
- 241000412611 Consul Species 0.000 description 1
- 229920000742 Cotton Polymers 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 208000001840 Dandruff Diseases 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- QXNVGIXVLWOKEQ-UHFFFAOYSA-N Disodium Chemical compound [Na][Na] QXNVGIXVLWOKEQ-UHFFFAOYSA-N 0.000 description 1
- 208000003556 Dry Eye Syndromes Diseases 0.000 description 1
- 206010013774 Dry eye Diseases 0.000 description 1
- 240000002943 Elettaria cardamomum Species 0.000 description 1
- MPDGHEJMBKOTSU-UHFFFAOYSA-N Glycyrrhetinsaeure Natural products C12C(=O)C=C3C4CC(C)(C(O)=O)CCC4(C)CCC3(C)C1(C)CCC1C2(C)CCC(O)C1(C)C MPDGHEJMBKOTSU-UHFFFAOYSA-N 0.000 description 1
- 229920002907 Guar gum Polymers 0.000 description 1
- 235000001287 Guettarda speciosa Nutrition 0.000 description 1
- 229920000569 Gum karaya Polymers 0.000 description 1
- 206010019233 Headaches Diseases 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 240000001046 Lactobacillus acidophilus Species 0.000 description 1
- 235000013956 Lactobacillus acidophilus Nutrition 0.000 description 1
- 244000199866 Lactobacillus casei Species 0.000 description 1
- 235000013958 Lactobacillus casei Nutrition 0.000 description 1
- 241000186606 Lactobacillus gasseri Species 0.000 description 1
- 241000186605 Lactobacillus paracasei Species 0.000 description 1
- 240000006024 Lactobacillus plantarum Species 0.000 description 1
- 235000013965 Lactobacillus plantarum Nutrition 0.000 description 1
- 241000186604 Lactobacillus reuteri Species 0.000 description 1
- 241000218588 Lactobacillus rhamnosus Species 0.000 description 1
- 241000186869 Lactobacillus salivarius Species 0.000 description 1
- 241000194036 Lactococcus Species 0.000 description 1
- 235000017858 Laurus nobilis Nutrition 0.000 description 1
- 235000019501 Lemon oil Nutrition 0.000 description 1
- VTAJIXDZFCRWBR-UHFFFAOYSA-N Licoricesaponin B2 Natural products C1C(C2C(C3(CCC4(C)CCC(C)(CC4C3=CC2)C(O)=O)C)(C)CC2)(C)C2C(C)(C)CC1OC1OC(C(O)=O)C(O)C(O)C1OC1OC(C(O)=O)C(O)C(O)C1O VTAJIXDZFCRWBR-UHFFFAOYSA-N 0.000 description 1
- 206010024561 Lip pain Diseases 0.000 description 1
- 206010024570 Lip swelling Diseases 0.000 description 1
- 208000008167 Magnesium Deficiency Diseases 0.000 description 1
- JLVVSXFLKOJNIY-UHFFFAOYSA-N Magnesium ion Chemical compound [Mg+2] JLVVSXFLKOJNIY-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 206010049816 Muscle tightness Diseases 0.000 description 1
- AOMUHOFOVNGZAN-UHFFFAOYSA-N N,N-bis(2-hydroxyethyl)dodecanamide Chemical compound CCCCCCCCCCCC(=O)N(CCO)CCO AOMUHOFOVNGZAN-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 235000019502 Orange oil Nutrition 0.000 description 1
- 241001529744 Origanum Species 0.000 description 1
- 240000000783 Origanum majorana Species 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 235000008331 Pinus X rigitaeda Nutrition 0.000 description 1
- 235000011613 Pinus brutia Nutrition 0.000 description 1
- 241000018646 Pinus brutia Species 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- 229920001214 Polysorbate 60 Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 208000003251 Pruritus Diseases 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 208000005392 Spasm Diseases 0.000 description 1
- 241000934878 Sterculia Species 0.000 description 1
- UEDUENGHJMELGK-HYDKPPNVSA-N Stevioside Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O UEDUENGHJMELGK-HYDKPPNVSA-N 0.000 description 1
- 241000194020 Streptococcus thermophilus Species 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- 206010042496 Sunburn Diseases 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- XOAAWQZATWQOTB-UHFFFAOYSA-N Taurine Natural products NCCS(O)(=O)=O XOAAWQZATWQOTB-UHFFFAOYSA-N 0.000 description 1
- 235000005212 Terminalia tomentosa Nutrition 0.000 description 1
- 244000125380 Terminalia tomentosa Species 0.000 description 1
- 201000006704 Ulcerative Colitis Diseases 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- 238000005299 abrasion Methods 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 229960000458 allantoin Drugs 0.000 description 1
- 230000007815 allergy Effects 0.000 description 1
- QYIXCDOBOSTCEI-UHFFFAOYSA-N alpha-cholestanol Natural products C1CC2CC(O)CCC2(C)C2C1C1CCC(C(C)CCCC(C)C)C1(C)CC2 QYIXCDOBOSTCEI-UHFFFAOYSA-N 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- SNAAJJQQZSMGQD-UHFFFAOYSA-N aluminum magnesium Chemical compound [Mg].[Al] SNAAJJQQZSMGQD-UHFFFAOYSA-N 0.000 description 1
- 229960002684 aminocaproic acid Drugs 0.000 description 1
- 150000008064 anhydrides Chemical class 0.000 description 1
- 239000003945 anionic surfactant Substances 0.000 description 1
- 239000010617 anise oil Substances 0.000 description 1
- 230000002272 anti-calculus Effects 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 230000002882 anti-plaque Effects 0.000 description 1
- 229940064004 antiseptic throat preparations Drugs 0.000 description 1
- 210000000436 anus Anatomy 0.000 description 1
- 229940076292 aquafresh Drugs 0.000 description 1
- 239000000607 artificial tear Substances 0.000 description 1
- 239000012752 auxiliary agent Substances 0.000 description 1
- 208000022362 bacterial infectious disease Diseases 0.000 description 1
- 239000010620 bay oil Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- WHGYBXFWUBPSRW-FOUAGVGXSA-N beta-cyclodextrin Chemical compound OC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)CO)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1CO WHGYBXFWUBPSRW-FOUAGVGXSA-N 0.000 description 1
- 229940002008 bifidobacterium bifidum Drugs 0.000 description 1
- 229940004120 bifidobacterium infantis Drugs 0.000 description 1
- 229940009289 bifidobacterium lactis Drugs 0.000 description 1
- 229940009291 bifidobacterium longum Drugs 0.000 description 1
- 235000019658 bitter taste Nutrition 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 235000019437 butane-1,3-diol Nutrition 0.000 description 1
- 229940067596 butylparaben Drugs 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 244000309466 calf Species 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 229940105329 carboxymethylcellulose Drugs 0.000 description 1
- 235000005300 cardamomo Nutrition 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 229960001927 cetylpyridinium chloride Drugs 0.000 description 1
- YMKDRGPMQRFJGP-UHFFFAOYSA-M cetylpyridinium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCC[N+]1=CC=CC=C1 YMKDRGPMQRFJGP-UHFFFAOYSA-M 0.000 description 1
- 239000010628 chamomile oil Substances 0.000 description 1
- 235000019480 chamomile oil Nutrition 0.000 description 1
- 239000013522 chelant Substances 0.000 description 1
- 229930002875 chlorophyll Natural products 0.000 description 1
- 235000019804 chlorophyll Nutrition 0.000 description 1
- ATNHDLDRLWWWCB-AENOIHSZSA-M chlorophyll a Chemical compound C1([C@@H](C(=O)OC)C(=O)C2=C3C)=C2N2C3=CC(C(CC)=C3C)=[N+]4C3=CC3=C(C=C)C(C)=C5N3[Mg-2]42[N+]2=C1[C@@H](CCC(=O)OC\C=C(/C)CCC[C@H](C)CCC[C@H](C)CCCC(C)C)[C@H](C)C2=C5 ATNHDLDRLWWWCB-AENOIHSZSA-M 0.000 description 1
- 239000001279 citrus aurantifolia swingle expressed oil Substances 0.000 description 1
- 239000001524 citrus aurantium oil Substances 0.000 description 1
- 239000010500 citrus oil Substances 0.000 description 1
- 239000001071 citrus reticulata blanco var. mandarin Substances 0.000 description 1
- 239000010634 clove oil Substances 0.000 description 1
- 239000010636 coriander oil Substances 0.000 description 1
- 239000001941 cymbopogon citratus dc and cymbopogon flexuosus oil Substances 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000013872 defecation Effects 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- MHJAJDCZWVHCPF-UHFFFAOYSA-L dimagnesium phosphate Chemical compound [Mg+2].OP([O-])([O-])=O MHJAJDCZWVHCPF-UHFFFAOYSA-L 0.000 description 1
- 229910000395 dimagnesium phosphate Inorganic materials 0.000 description 1
- 235000019791 dimagnesium phosphate Nutrition 0.000 description 1
- XPPKVPWEQAFLFU-UHFFFAOYSA-N diphosphoric acid Chemical compound OP(O)(=O)OP(O)(O)=O XPPKVPWEQAFLFU-UHFFFAOYSA-N 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 206010013781 dry mouth Diseases 0.000 description 1
- 210000000883 ear external Anatomy 0.000 description 1
- 229960003720 enoxolone Drugs 0.000 description 1
- AEUTYOVWOVBAKS-UWVGGRQHSA-N ethambutol Natural products CC[C@@H](CO)NCCN[C@@H](CC)CO AEUTYOVWOVBAKS-UWVGGRQHSA-N 0.000 description 1
- 229960001617 ethyl hydroxybenzoate Drugs 0.000 description 1
- 239000004403 ethyl p-hydroxybenzoate Substances 0.000 description 1
- 235000010228 ethyl p-hydroxybenzoate Nutrition 0.000 description 1
- NUVBSKCKDOMJSU-UHFFFAOYSA-N ethylparaben Chemical compound CCOC(=O)C1=CC=C(O)C=C1 NUVBSKCKDOMJSU-UHFFFAOYSA-N 0.000 description 1
- 239000010642 eucalyptus oil Substances 0.000 description 1
- 229940044949 eucalyptus oil Drugs 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 206010015915 eye discharge Diseases 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 210000004195 gingiva Anatomy 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- LPLVUJXQOOQHMX-UHFFFAOYSA-N glycyrrhetinic acid glycoside Natural products C1CC(C2C(C3(CCC4(C)CCC(C)(CC4C3=CC2=O)C(O)=O)C)(C)CC2)(C)C2C(C)(C)C1OC1OC(C(O)=O)C(O)C(O)C1OC1OC(C(O)=O)C(O)C(O)C1O LPLVUJXQOOQHMX-UHFFFAOYSA-N 0.000 description 1
- 239000001685 glycyrrhizic acid Substances 0.000 description 1
- 229960004949 glycyrrhizic acid Drugs 0.000 description 1
- UYRUBYNTXSDKQT-UHFFFAOYSA-N glycyrrhizic acid Natural products CC1(C)C(CCC2(C)C1CCC3(C)C2C(=O)C=C4C5CC(C)(CCC5(C)CCC34C)C(=O)O)OC6OC(C(O)C(O)C6OC7OC(O)C(O)C(O)C7C(=O)O)C(=O)O UYRUBYNTXSDKQT-UHFFFAOYSA-N 0.000 description 1
- 235000019410 glycyrrhizin Nutrition 0.000 description 1
- LPLVUJXQOOQHMX-QWBHMCJMSA-N glycyrrhizinic acid Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@H](O[C@@H]1O[C@@H]1C([C@H]2[C@]([C@@H]3[C@@]([C@@]4(CC[C@@]5(C)CC[C@@](C)(C[C@H]5C4=CC3=O)C(O)=O)C)(C)CC2)(C)CC1)(C)C)C(O)=O)[C@@H]1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O LPLVUJXQOOQHMX-QWBHMCJMSA-N 0.000 description 1
- 239000010651 grapefruit oil Substances 0.000 description 1
- 239000000665 guar gum Substances 0.000 description 1
- 235000010417 guar gum Nutrition 0.000 description 1
- 229960002154 guar gum Drugs 0.000 description 1
- 231100000869 headache Toxicity 0.000 description 1
- 229910052588 hydroxylapatite Inorganic materials 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 208000027866 inflammatory disease Diseases 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 210000000936 intestine Anatomy 0.000 description 1
- 230000007803 itching Effects 0.000 description 1
- 239000010656 jasmine oil Substances 0.000 description 1
- 239000000231 karaya gum Substances 0.000 description 1
- 235000010494 karaya gum Nutrition 0.000 description 1
- 229940039371 karaya gum Drugs 0.000 description 1
- 239000000832 lactitol Substances 0.000 description 1
- 235000010448 lactitol Nutrition 0.000 description 1
- 229960003451 lactitol Drugs 0.000 description 1
- 229940039695 lactobacillus acidophilus Drugs 0.000 description 1
- 229940017800 lactobacillus casei Drugs 0.000 description 1
- 229940072205 lactobacillus plantarum Drugs 0.000 description 1
- 229940001882 lactobacillus reuteri Drugs 0.000 description 1
- 229940031957 lauric acid diethanolamide Drugs 0.000 description 1
- 239000000171 lavandula angustifolia l. flower oil Substances 0.000 description 1
- 239000010501 lemon oil Substances 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- HZZOEADXZLYIHG-UHFFFAOYSA-N magnesiomagnesium Chemical compound [Mg][Mg] HZZOEADXZLYIHG-UHFFFAOYSA-N 0.000 description 1
- UEGPKNKPLBYCNK-UHFFFAOYSA-L magnesium acetate Chemical compound [Mg+2].CC([O-])=O.CC([O-])=O UEGPKNKPLBYCNK-UHFFFAOYSA-L 0.000 description 1
- 239000011654 magnesium acetate Substances 0.000 description 1
- 235000011285 magnesium acetate Nutrition 0.000 description 1
- 229940069446 magnesium acetate Drugs 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 235000004764 magnesium deficiency Nutrition 0.000 description 1
- 239000004137 magnesium phosphate Substances 0.000 description 1
- 239000002075 main ingredient Substances 0.000 description 1
- 239000011738 major mineral Substances 0.000 description 1
- 235000011963 major mineral Nutrition 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 239000000845 maltitol Substances 0.000 description 1
- 235000010449 maltitol Nutrition 0.000 description 1
- 229940035436 maltitol Drugs 0.000 description 1
- 239000001683 mentha spicata herb oil Substances 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- OSWPMRLSEDHDFF-UHFFFAOYSA-N methyl salicylate Chemical compound COC(=O)C1=CC=CC=C1O OSWPMRLSEDHDFF-UHFFFAOYSA-N 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- 230000003020 moisturizing effect Effects 0.000 description 1
- 239000002324 mouth wash Substances 0.000 description 1
- 210000003928 nasal cavity Anatomy 0.000 description 1
- 239000007923 nasal drop Substances 0.000 description 1
- 229940100662 nasal drops Drugs 0.000 description 1
- 210000002850 nasal mucosa Anatomy 0.000 description 1
- 210000002445 nipple Anatomy 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 239000010502 orange oil Substances 0.000 description 1
- 239000006072 paste Substances 0.000 description 1
- XYJRXVWERLGGKC-UHFFFAOYSA-D pentacalcium;hydroxide;triphosphate Chemical compound [OH-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O XYJRXVWERLGGKC-UHFFFAOYSA-D 0.000 description 1
- KFHHGNBIPJDZPH-UHFFFAOYSA-D pentamagnesium [oxido(phosphonatooxy)phosphoryl] phosphate Chemical compound [Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[O-]P([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O.[O-]P([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O KFHHGNBIPJDZPH-UHFFFAOYSA-D 0.000 description 1
- 229960005323 phenoxyethanol Drugs 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920000259 polyoxyethylene lauryl ether Polymers 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 239000006041 probiotic Substances 0.000 description 1
- 235000018291 probiotics Nutrition 0.000 description 1
- ULWHHBHJGPPBCO-UHFFFAOYSA-N propane-1,1-diol Chemical compound CCC(O)O ULWHHBHJGPPBCO-UHFFFAOYSA-N 0.000 description 1
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- 229940005657 pyrophosphoric acid Drugs 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 239000010666 rose oil Substances 0.000 description 1
- 235000019719 rose oil Nutrition 0.000 description 1
- 239000010668 rosemary oil Substances 0.000 description 1
- 229940058206 rosemary oil Drugs 0.000 description 1
- 229940085605 saccharin sodium Drugs 0.000 description 1
- 239000010670 sage oil Substances 0.000 description 1
- 210000003296 saliva Anatomy 0.000 description 1
- 235000019643 salty taste Nutrition 0.000 description 1
- 210000004761 scalp Anatomy 0.000 description 1
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 description 1
- 210000002374 sebum Anatomy 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- WXMKPNITSTVMEF-UHFFFAOYSA-M sodium benzoate Chemical compound [Na+].[O-]C(=O)C1=CC=CC=C1 WXMKPNITSTVMEF-UHFFFAOYSA-M 0.000 description 1
- 239000004299 sodium benzoate Substances 0.000 description 1
- 235000010234 sodium benzoate Nutrition 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 235000019721 spearmint oil Nutrition 0.000 description 1
- 229940013618 stevioside Drugs 0.000 description 1
- OHHNJQXIOPOJSC-UHFFFAOYSA-N stevioside Natural products CC1(CCCC2(C)C3(C)CCC4(CC3(CCC12C)CC4=C)OC5OC(CO)C(O)C(O)C5OC6OC(CO)C(O)C(O)C6O)C(=O)OC7OC(CO)C(O)C(O)C7O OHHNJQXIOPOJSC-UHFFFAOYSA-N 0.000 description 1
- 235000019202 steviosides Nutrition 0.000 description 1
- 210000000434 stratum corneum Anatomy 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 1
- 230000009469 supplementation Effects 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 229920003002 synthetic resin Polymers 0.000 description 1
- 239000000057 synthetic resin Substances 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 229960003080 taurine Drugs 0.000 description 1
- 239000010678 thyme oil Substances 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- GYDJEQRTZSCIOI-LJGSYFOKSA-N tranexamic acid Chemical compound NC[C@H]1CC[C@H](C(O)=O)CC1 GYDJEQRTZSCIOI-LJGSYFOKSA-N 0.000 description 1
- 229960000401 tranexamic acid Drugs 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 229940078499 tricalcium phosphate Drugs 0.000 description 1
- 229910000391 tricalcium phosphate Inorganic materials 0.000 description 1
- 235000019731 tricalcium phosphate Nutrition 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 230000002087 whitening effect Effects 0.000 description 1
- 239000009637 wintergreen oil Substances 0.000 description 1
- 239000000230 xanthan gum Substances 0.000 description 1
- 235000010493 xanthan gum Nutrition 0.000 description 1
- 229920001285 xanthan gum Polymers 0.000 description 1
- 229940082509 xanthan gum Drugs 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
- 239000004711 α-olefin Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K33/00—Medicinal preparations containing inorganic active ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K33/00—Medicinal preparations containing inorganic active ingredients
- A61K33/06—Aluminium, calcium or magnesium; Compounds thereof, e.g. clay
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/40—Transferrins, e.g. lactoferrins, ovotransferrins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/02—Inorganic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/64—Proteins; Peptides; Derivatives or degradation products thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0031—Rectum, anus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/51—Nanocapsules; Nanoparticles
- A61K9/5107—Excipients; Inactive ingredients
- A61K9/5115—Inorganic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/10—Laxatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q11/00—Preparations for care of the teeth, of the oral cavity or of dentures; Dentifrices, e.g. toothpastes; Mouth rinses
Definitions
- the present disclosure relates to a water-soluble composition and a method for producing the same.
- periodontal disease is recognized as a bacterial infection. Therefore, various oral compositions having a bactericidal effect have been proposed as means for improving periodontal disease.
- Patent Document 1 discloses an oral composition containing a cationic antiseptic such as cetylpyridinium chloride.
- an object of the present disclosure is to provide a water-soluble composition that improves the cell environment and a method for producing the same.
- the water-soluble composition is containing magnesium, sodium and protein,
- the protein is lactoferrin
- the content of magnesium is 2.0 to 12.0 w / v%
- the content of lactoferrin is 3.0 to 10.0 w/v%.
- nutrients are absorbed from the mucosal tissue and supplied to the cells, thereby improving the cell environment and providing various effects.
- an anti-inflammatory effect against bacterial inflammation is obtained.
- the water-soluble composition of the present disclosure When the water-soluble composition of the present disclosure is used in oral compositions such as dentifrices and mouthwashes, it reduces the motility of bad bacteria in the mouth and improves the cellular environment in the gums, leading to inflammation and bleeding in the gums. can be suppressed to prevent periodontal disease.
- the water-soluble composition of the present disclosure when the water-soluble composition of the present disclosure is orally ingested, it reduces the motility of bad bacteria in the gastrointestinal tract and improves the cell environment in the gastrointestinal tract. Constipation caused by can be improved.
- the water-soluble composition of the present disclosure when the water-soluble composition of the present disclosure is applied to a wounded or burned skin, it can promote improvement of inflammation.
- FIG. 4 is an image of a phase-contrast microscope of Comparative Example 1.
- FIG. 4 is a phase-contrast microscope image of Example 1.
- FIG. It is an example of filling in a subjective evaluation check sheet used for evaluation of the water-soluble composition, and shows the evaluation results of subject 007.
- composition [Composition, action, use]
- the water-soluble composition of the present disclosure (hereinafter also simply referred to as the “composition”) contains high concentrations of magnesium, sodium, and lactoferrin as a protein, and is absorbed from mucosal tissue to supply nutrients to cells. As a result, the cell environment is improved, and various effects can be obtained.
- Magnesium ions are abundant in cells and are one of the major minerals essential for life support. Deficiency of magnesium ions in cells is thought to deteriorate the cellular environment and cause various diseases. The composition improves the cellular environment by allowing magnesium to be absorbed through mucosal tissue, making it possible to obtain various effects.
- the water-soluble composition preferably has a magnesium content of 2.0 to 12.0 w/v%, more preferably a magnesium content of 8.0 to 12.0 w/v%, more preferably 8.5 to 12.0 w/v%, particularly preferably 9.0 to 12.0 w/v%, most preferably 10.5 to 12.0 w/v%.
- a magnesium content of 2.0 to 12.0 w/v% more preferably a magnesium content of 8.0 to 12.0 w/v%, more preferably 8.5 to 12.0 w/v%, particularly preferably 9.0 to 12.0 w/v%, most preferably 10.5 to 12.0 w/v%.
- "-" is used to mean that the numerical values before and after it are included as lower and upper limits.
- magnesium deficiency is one of the factors that deteriorate the periodontal cell environment. Supplementation with magnesium is believed to be effective in alleviating various symptoms such as bleeding from gums and dissolution of alveolar bone caused by periodontal disease in which the periodontal cell environment deteriorates. It is considered that a high concentration of magnesium of 9.0 w/v% or more acts more effectively for periodontal disease symptoms, especially bleeding from the gums.
- a naturally occurring high-concentration magnesium solution can be a readily available commercial solution, such as a 12.0 w/v % magnesium solution produced from the water of Great Salt Lake, Utah, USA. .
- a magnesium content of 10.5 to 12.0 w/v% is suitable.
- [sodium] Sodium can promote the effect of magnesium by being included in the magnesium solution.
- the content ratio of magnesium and sodium (hereinafter also referred to as "Mg:Na ratio”) is considered to be important.
- the content ratio of magnesium to sodium is preferably 16 times or more, particularly preferably 40 times or more, relative to sodium.
- the Mg:Na ratio of the water-soluble composition is preferably 16:1 to 73:1.
- a sufficient amount of magnesium can be supplied to the mucosal tissue even in an environment where the composition is diluted.
- bittern An example of a magnesium solution is bittern, but the content ratio of magnesium and sodium in naturally derived bittern is not considered appropriate for improving the cell environment.
- Lactoferrin can impart foaming properties to the water-soluble composition. By containing lactoferrin, the composition stays in the mouth for a long time during brushing or gargling, making it possible to more effectively enhance magnesium absorption.
- the water-soluble composition preferably has a lactoferrin content of 3.0 to 10.0 w/v%.
- the lactoferrin is preferably lactoferrin that does not form a chelate structure, and more preferably lactoferrin that can form a complex.
- Preferred lactoferrins include, for example, lactoferrins with hollow lobes.
- FIG. 1 shows an image of the lactoferrin 2.0 w/v% preparation after 200 brushings.
- the preparation liquid containing 1.0 to 2.0 w/v % of lactoferrin hardly foamed.
- FIG. 2 shows an image of the lactoferrin 3.0 w/v % preparation after 200 brushings.
- lactoferrin it is preferable to use naturally-derived lactoferrin.
- By blending 3.0 to 10.0 w/v% of lactoferrin in the composition it is possible to form a composition with good foaming properties using only naturally-derived ingredients without containing foaming agents such as surfactants. can.
- the water-soluble composition can obtain various anti-inflammatory effects against bacterial inflammation by appropriately blending known ingredients within a range that does not interfere with the effects of the present disclosure and adopting various dosage forms. Therefore, the composition can also act as a dentifrice adjuvant.
- the water-soluble composition when used as an oral composition, it reduces the motility of bad bacteria in the mouth, improves the cellular environment in the gums, suppresses inflammation and bleeding in the gums, and prevents periodontal disease. be able to.
- the composition when the composition is orally ingested, it reduces the motility of bad bacteria in the gastrointestinal tract, improves the cell environment in the gastrointestinal tract, and improves constipation caused by an imbalance between good and bad bacteria in the intestine. be able to.
- the water-soluble composition when the water-soluble composition is applied to a wounded or burned skin, it can promote improvement of inflammation.
- a water-soluble composition can be applied to applications other than those mentioned above, but the composition is particularly suitable for use as an oral composition.
- oral compositions when employed as, for example, a dentifrice, flavoring agents, abrasives, humectants, foaming agents, thickeners, flavors, sweeteners, coloring agents, preservatives, pH adjusters, active ingredients, etc. It may be blended or used as an auxiliary agent for dentifrices.
- the water-soluble composition When used as an oral composition, it can contain a flavoring agent to mask the bitter taste of magnesium.
- a flavoring agent to mask the bitter taste of magnesium.
- preferred flavoring agents include cyclodextrins. Cyclodextrins include ⁇ -, ⁇ -, and ⁇ -forms, and each of them can be blended alone or in combination of two or more. In addition, other corrigents may be blended together with the cyclodextrin.
- abrasives examples include silica-based abrasives such as silica gel, dicalcium phosphate dihydrate and anhydride, tricalcium phosphate, calcium carbonate, aluminum hydroxide, alumina, magnesium carbonate, dimagnesium phosphate, One or two or more selected from magnesium triphosphate, magnesium acetate, zeolite, hydroxyapatite, pentonite, synthetic resins, and the like.
- silica-based abrasives such as silica gel, dicalcium phosphate dihydrate and anhydride, tricalcium phosphate, calcium carbonate, aluminum hydroxide, alumina, magnesium carbonate, dimagnesium phosphate, One or two or more selected from magnesium triphosphate, magnesium acetate, zeolite, hydroxyapatite, pentonite, synthetic resins, and the like.
- humectants include one or more selected from glycerin, sorbitol, ethylene glycol, propanediol, polyethylene glycol, 1,3-butylene glycol, propylene glycol, xylit, maltite and the like.
- foaming agents examples include anionic surfactants such as sodium lauryl sulfate, sodium ⁇ -olefin sulfonate, sodium N-methyl-N-acyl taurine, and sodium N-methyl-N-acylalanine, and sucrose fatty acid esters.
- anionic surfactants such as sodium lauryl sulfate, sodium ⁇ -olefin sulfonate, sodium N-methyl-N-acyl taurine, and sodium N-methyl-N-acylalanine, and sucrose fatty acid esters.
- maltose fatty acid ester maltitol fatty acid ester, lactitol fatty acid ester, sorbitan fatty acid ester, glycerin fatty acid ester, hexaglyceryl monolaurate, hexaglyceryl monomyristate, decaglyceryl monolaurate, polyoxyethylene sorbitan monolaurate, polyoxyethylene sorbitan
- nonionic surfactants such as monostearate, polyoxyethylene hydrogenated castor oil, polyoxyethylene lauryl ether, and lauric acid diethanolamide can be used.
- thickeners include one or more selected from guar gum, xanthan gum, karaya gum, carboxymethylcellulose, hydroxylmethylcellulose, colloidal magnesium aluminum silicate, and the like.
- Flavors include, for example, peppermint oil, spearmint oil, anise oil, eucalyptus oil, wintergreen oil, cassia oil, clove oil, thyme oil, sage oil, lemon oil, lime oil, orange oil, citrus oil, peppermint oil, and cardamom. oil, coriander oil, mandarin oil, lavender oil, rosemary oil, laurel oil, chamomile oil, caraway oil, marjoram oil, bay oil, lemongrass oil, origanum oil, pine needle oil, neroli oil, rose oil, jasmine oil , grapefruit oil, sweetie oil, and yuzu oil.
- Sweeteners include, for example, xylitol, saccharin sodium, and stevioside.
- antiseptics examples include methylparaben, ethylparaben, propylparaben, butylparaben, paraoxybenzoate, sodium benzoate, phenoxyethanol, and the like.
- pH adjusters examples include citric acid, phosphoric acid, malic acid, pyrophosphoric acid, lactic acid, tartaric acid, acetic acid, and nitric acid.
- active ingredients include anti-inflammatory agents such as tranexamic acid, epsilon aminocaproic acid, allantoin, glycyrrhetinic acid, and glycyrrhizic acid; zeolite, azulene, dihydrocholesterol, chlorophyll, angelica soft extract; Extracts, vitamins, anti-calculus agents, anti-plaque agents and the like.
- anti-inflammatory agents such as tranexamic acid, epsilon aminocaproic acid, allantoin, glycyrrhetinic acid, and glycyrrhizic acid
- zeolite azulene, dihydrocholesterol, chlorophyll, angelica soft extract
- Extracts vitamins, anti-calculus agents, anti-plaque agents and the like.
- the water-soluble composition may contain good bacteria such as lactic acid bacteria, natto bacteria, saccharifying bacteria, and butyric acid bacteria as useful ingredients.
- beneficial bacteria include bacteria of the genus Lactobacillus and Lactococcus.
- Useful ingredients include, for example, Bifidobacterium bifidum, Bifidobacterium longum, Bifidobacterium adolescentis, Bifidobacterium breve, Bifidobacterium infantis, Bifidobacterium ⁇ Bifidobacteria such as animalis, Bifidobacterium pseudolongum, Bifidobacterium lactis, Lactobacillus casei, Lactobacillus paracasei, Lactobacillus acidophilus, Lactobacillus reuteri, Lactobacillus gasseri, Lactobacillus - Lactic acid bacteria such as bulgaricus, Lactobacillus salivarius, Lactobacillus rhamnos
- the sodium-containing magnesium solution used as a raw material it is preferable to use a naturally derived magnesium solution produced from seawater or salt lake water.
- lactoferrin is mixed with the sodium-containing magnesium solution.
- the resulting mixed solution is prepared to have a magnesium content of 2.0 to 12.0 w/v% and a lactoferrin content of 3.0 to 10.0 w/v%.
- other ingredients such as abrasives, wetting agents, foaming agents, thickeners, flavors, sweeteners, corrigents, coloring agents, preservatives, and active ingredients may be added together. may be mixed into
- the mixed solution foamed by the effect of lactoferrin is defoamed.
- a method of standing at normal temperature and pressure for a certain period of time for example, 3 days
- a method of keeping the state cooled to 0 to 5 ° C. at normal pressure for a certain period of time for example, 3 days
- pulling to 0 atm at room temperature There is a method of keeping the pressed state for a certain period of time (5 to 24 hours).
- the method for producing the water-soluble composition preferably does not further include a step of adjusting the amount of sodium. Specifically, it is more preferable that the method for producing the water-soluble composition does not further include the step of adding sodium and/or the step of removing sodium. That is, the Mg:Na ratio in the water-soluble composition is defined in step S1. In other words, the first step S1 includes a step of determining the content ratio of magnesium and sodium.
- Comparative example 1 First, the above plaque alone was applied to a slide glass, and a cover glass was placed on the slide glass, which was then observed under a phase-contrast microscope. A phase-contrast microscope image is shown in FIG. In Comparative Example 1, many bacteria were observed to move actively.
- Example 1 A water-soluble composition having a magnesium content of 10.5 w/v%, a sodium content of 0.25 w/v%, a potassium content of 1.18 w/v%, and a lactoferrin content of 4.5 w/v% (hereinafter referred to as sample 1 ) was prepared. Specifically, first, a sodium-containing magnesium solution having a magnesium content of 10.5 w/v% and a Mg:Na ratio of 42.7:1 (manufactured by Meitia, Magneforce (registered trademark)) prepared. Then, 0.45 g of lactoferrin was mixed with 10 mL of the sodium-containing magnesium solution to obtain a mixed solution as a precursor of sample 1.
- FIG. 5 shows an image obtained by a phase-contrast microscope when this sample 1 was dropped onto the plaque.
- the bacteria did not move at all, confirming that Sample 1 has the effect of suppressing the action of bacteria.
- Example 2 Same as in the preparation of sample 1 above, except that 0.1 g of sodium chloride was added to the sodium-containing magnesium solution (Magneforce (registered trademark), manufactured by Meitia) to make the Mg:Na ratio 16.4: 1. to prepare sample 1A.
- This sample 1A was dropped onto the plaque and observed with a phase-contrast microscope. As in Example 1, the bacteria did not move at all. rice field.
- Example 2 14 mL of purified water was added to 16 mL of a sodium-containing magnesium solution (manufactured by Meitia, Magneforce (registered trademark)) having a magnesium content of 10.5 w/v% and a Mg:Na ratio of 42.7:1. In addition, it was diluted and 0.11 g of sodium chloride was added thereto. Sample 2A was prepared with a magnesium content of 5.6 w/v% and a Mg:Na ratio of 20.3:1. This sample 2A was dropped onto the plaque and observed with a phase-contrast microscope.
- a sodium-containing magnesium solution manufactured by Meitia, Magneforce (registered trademark)
- Sample 2C was prepared by mixing 0.6 g of lactoferrin with 20 mL of commercially available bittern (manufactured by Hakumatsu Co., Ltd., trade name “Hamamishio seawater bittern”). Sample 2C has a magnesium content of 5.6 w/v%, a lactoferrin content of 3.0 w/v% and a Mg:Na ratio of 1.3:1. This sample 2C was dropped onto the plaque and observed with a phase-contrast microscope.
- Sample 2D was prepared by mixing 0.75 g of lactoferrin with a solution obtained by diluting 10 mL of commercially available bittern (manufactured by Hakumatsu Co., Ltd., trade name "Hamamishio seawater bittern") with 15 mL of purified water.
- Sample 2D has a magnesium content of 2.2 w/v%, a lactoferrin content of 3.0 w/v% and a Mg:Na ratio of 1.3:1. This sample 2D was dropped onto the plaque and observed with a phase-contrast microscope.
- Example 3 Using a sodium-containing magnesium solution (Magneforce (registered trademark) manufactured by Meitia) and lactoferrin, the magnesium content was 8.8 w/v%, the lactoferrin content was 3.9 w/v%, and the Mg:Na ratio was 47.7. Sample 3A was prepared which was :1. This sample 3A was dropped onto the plaque and observed with a phase contrast microscope.
- Magneticforce registered trademark manufactured by Meitia
- Example 4 Using a sodium-containing magnesium solution (Magneforce (registered trademark) manufactured by Meitia) and lactoferrin, the magnesium content was 8.0 w/v%, the lactoferrin content was 3.5 w/v%, and the Mg:Na ratio was 47.4. Sample 3B was prepared which was :1. This sample 3B was dropped onto the plaque and observed with a phase-contrast microscope. In Example 4, the state of bacteriostasis was not as clear as in Examples 1 to 3, but compared with Comparative Examples 1 to 5, the movement of bacteria was weakened, indicating a tendency toward bacteriostasis. I was able to say
- Table 1 shows the results of phase contrast observation using samples 1 and 1A, samples 2A to 2D, and samples 3A and 3B. As described above, it was confirmed that Sample 1, Sample 1A, Sample 3A and Sample 3B had a bacteriostatic effect.
- Subject evaluation Seventeen subjects were recruited to use the water-soluble composition of the present disclosure as a toothpaste for three months. The subjects were instructed to swallow as much of the water-soluble composition as possible without spitting out the gargling water when brushing their teeth. Subjects evaluated the following 10 subjective evaluation items with integers from 0 to 10 before the start of the study and after 3 months. For example, for each item, 10 points are given for "strongly feel” or “strongly agree”, and 0 points are given for "do not feel at all” or “do not agree at all”. ⁇ I'm concerned about bad breath ⁇ Stickiness ⁇ Slimy ⁇ Loose teeth 6 shows the evaluation results of subject 007 as an example of filling out a subjective evaluation check sheet. For some items, the scores after 3 months had decreased compared to before the start of the test, and the symptoms had improved.
- Table 2 shows the evaluation results of subjects 001 to 017.
- Toothpaste I Magnesium Toothpaste, manufactured by New Science. Contains magnesium chloride (MgCl.6H 2 O). The magnesium content calculated from the magnesium content is 1.8 w/v%. Contains no sodium or lactoferrin. Toothpaste II: Clear Clean (registered trademark) premium whitening manufactured by Kao Corporation. Toothpaste III: New Aquafresh® ZF3, manufactured by GlaxoSmithKline. Toothpaste IV: Sunstar Ora2® Premium Stain Clear.
- Toothpaste V Kamtect (registered trademark) Complete Care EX manufactured by GlaxoSmithKline.
- Toothpaste VI Shabondama Soap (registered trademark) toothpaste manufactured by Shabondama Soap Co., Ltd.
- Toothpaste VII Clear Clean (registered trademark) RR manufactured by Kao Corporation.
- Toothpaste VIII Medicated GUM (registered trademark) dental paste GS manufactured by Sunstar.
- Toothpaste IX crude drug Hc manufactured by Kobayashi Pharmaceutical Co., Ltd.; Dentifrices II-IX do not contain magnesium and lactoferrin.
- Toothpaste X B+ manufactured by AT-MARK CONSUL. The main ingredient is fossil coral.
- the content of magnesium in natural fossil coral is less than 1%, so it is considered that the content of magnesium in this toothpaste is also less than 1 w/v%. Does not contain lactoferrin. Toothpaste XI: Lion Corporation, Dentor Systema® EXW. Does not contain magnesium and lactoferrin.
- Subject 001 used a water-soluble composition having a magnesium content of 2.0 w/v%, a lactoferrin content of 3.2 w/v%, and a Mg:Na ratio of 42.8:1 (hereinafter referred to as sample 4). .
- Test subjects 002 to 017 used Sample 1 of Example 1 above.
- subject 001 used toothpaste I, which contained 1.8 w/v% magnesium and did not contain lactoferrin and sodium. After 3 months from the start of the test, subject 001 responded that symptoms such as bad breath, sticky/slimy gums, swollen and bleeding gums, and stomatitis were greatly improved. The results indicated that the combination of magnesium with sodium and lactoferrin improved the cellular environment in the mouth.
- Figures 7 and 8 are graphs visually showing changes in scores before the start of the test and after 3 months, regarding the subjective evaluation results of the 17 subjects.
- Table 2 an improvement of 3 points or more was marked as A, but it can be seen from FIGS. 7 and 8 that there are many subjects who felt a significant improvement of 8 to 10 points.
- Table 3 shows the results of the subjective evaluation of 17 subjects, the improvement rate calculated from the transition of points before the start of the test and after 3 months, and the improvement from the transition of the number of people who evaluated 5 points or less. The result of calculating the increase rate of the number of people who felt it is shown.
- the improvement rate and the rate of increase in the number of people who felt improvement also showed that the water-soluble composition of the present disclosure has the effect of adjusting the cellular environment in the mouth.
- the inspection table shown in Figures 9 and 10 was used for the probing inspection.
- the notation of "PD" in FIGS. 9 and 10 is the periodontal pocket depth (Probing depth).
- a periodontal pocket probe was inserted into the gingival sulcus on the front and back of all teeth, and the depth of insertion of the probe when the tip reached the bottom of the gingival sulcus was visually confirmed to measure the depth of the periodontal pocket.
- Numerals in FIGS. 9 and 10 indicate the measurement results of the periodontal pocket depth (unit: mm).
- the condition of the gums is considered to be good. 3 mm is diagnosed as mild periodontal disease, 4-5 mm as moderate periodontal disease, and 6 mm or more as severe periodontal disease. As shown in FIGS. 9 and 10, in the examination table, the numbers indicating the measurement results of the periodontal pocket depth are color-coded according to mild, moderate, and severe symptoms.
- the notation "BoP” indicates an inspection for the presence or absence of bleeding (Bleeding on Probing) during the probing inspection. , the squares are shown in color. Measurement of periodontal pocket depth is an important clinical examination, and if bleeding is observed from the measurement site during periodontal pocket depth measurement, it means that inflammation exists at that site. do. After measuring the depth of the periodontal pockets on the front and back of all teeth, the periodontal pocket probe was pulled out and it was visually determined whether there was bleeding from the gingival sulcus. As shown in FIGS. 9 and 10, in the inspection table, the locations where bleeding has been confirmed are colored. In other words, it is considered that the colored portion of the inspection table indicates that the gingiva is inflamed.
- BOP rate The number of test sites where bleeding was observed during the probing test, divided by the total number of test sites, and expressed as a percentage is called the "BOP rate.”
- Table 4 shows the results of the probing examinations performed before the start of the study and after 3 months had passed for 7 of the 17 subjects.
- the "improvement rate of periodontal pocket depth” is the number of inspection locations where the periodontal pocket depth became shallow after 3 months compared to before the start of the test, divided by the total number of inspection locations. It is a numerical value expressed as a percentage.
- "improved BOP rate” is the value obtained by subtracting the BOP rate after 3 months from the BOP rate before the start of the test. For both “improvement rate of periodontal pocket depth” and “improvement of BOP rate”, the larger the numerical value, the better the improvement.
- Subjects 016 and 017 answered that they did not feel any improvement in the subjective evaluation, but they did see improvement in the objective evaluation. Specifically, subject 016 showed significant improvement in both periodontal pocket depth and BOP ratio, and subject 017 showed improvement in BOP ratio. Subject 016 had mild to moderate periodontal disease, since the BOP rate before the start of the test was 92.9% and there were many places with periodontal pocket depths of 4 mm or more. Therefore, it is considered that Subject 017 showed a remarkable improvement effect. Subject 017 had a good oral environment, with a BOP rate of 5.4% before the start of the test, and no areas with a periodontal pocket depth of 4 mm or more. Therefore, although there was no remarkable change, it is considered that the oral environment was further improved.
- the periodontal pocket depth can be improved when the magnesium content is 2.0 w/v%. Do you get it. In addition, when the magnesium content was 10.5 w/v% or more, it was thought that the periodontal pocket depth and BOP ratio could be improved more effectively.
- Sample 1 As a toothpaste for 3 months. After 3 months from the start of the test, a total of 26 subjects were asked to freely state their impressions of using the water-soluble composition of the present disclosure, not only changes in the oral cavity, but also changes in their physical condition.
- Subject X instilled one drop of sample 1 into each of his left and right eyes.
- subject Y two drops of sample 1 were added to 2.5 ml of water to prepare a solution, and the eye was washed with the obtained solution.
- Subjects X and Y experienced no discomfort due to eye discharge or dry eye after eye drops or washing.
- Subject Y impregnated a cotton swab with several drops of Sample 1 and applied it to the ear canal. After 48 hours, the inflammation caused by abrasion of the outer ear subsided, suggesting that the bacterial balance in the ear canal was improved.
- Subject Y applied the solution obtained by dropping 0.2 ml of sample 1 into about 1.5 ml of commercially available lotion on the skin. After 8 hours, the condition of the sebum film and/or stratum corneum of the skin became smooth.
- Subject X applied an excess amount of the solution obtained by diluting Sample 1 to double the amount on the sural spasm area and percutaneously absorbed it. After 8 hours, the muscle tension in the calf was relaxed and the pain was improved.
- Sample 1 can also be packed in a capsule and used as a suppository, and can be expected to induce defecation, improve bleeding around the anus, improve symptoms of hemorrhoids, and improve the balance of bacteria in the rectum.
- subject Z used a suppository containing Sample 1 in a capsule, bleeding due to ulcerative colitis was suppressed.
- Sample 1 can be diluted and used as a nasal wash or nasal drops, and can be expected to improve inflammation of the nasal mucosa, improve sinusitis, and improve the balance of bacteria in the nasal cavity.
- the water-soluble composition When used as a toothpaste, it can improve stomatitis, reduce pain in areas covered by teeth, prevent dry mouth, improve bad breath, improve lip sores, improve lip swelling, It can also be expected to be effective in alleviating headaches.
- the water-soluble composition when used as an external preparation, is effective in improving sinusitis, improving skin itching, moisturizing the skin, preventing dandruff on the scalp, improving sunburn, and improving nipple sores. can be expected.
- the water-soluble composition is expected to be effective in improving hemorrhoids when used as an internal medicine.
- this water-soluble composition is particularly useful as an oral composition, but it has also been shown to be useful for other uses.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Inorganic Chemistry (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Immunology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Birds (AREA)
- Nanotechnology (AREA)
- Optics & Photonics (AREA)
- Biomedical Technology (AREA)
- Physics & Mathematics (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Cosmetics (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
- Biological Depolymerization Polymers (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Jellies, Jams, And Syrups (AREA)
- Food Preservation Except Freezing, Refrigeration, And Drying (AREA)
Abstract
Description
マグネシウムと、ナトリウムと、タンパク質とを含み、
マグネシウムとナトリウムとの含有比が、マグネシウム:ナトリウム=16:1~73:1であり、
タンパク質は、ラクトフェリンであり、
マグネシウムの含有率が、2.0~12.0w/v%であり、
ラクトフェリンの含有率が、3.0~10.0w/v%である。
本開示の水溶性組成物(以下、単に「組成物」ともいう。)は、高濃度のマグネシウムと、ナトリウムと、タンパク質としてのラクトフェリンを含み、粘膜組織から吸収されて細胞へ栄養素が補給されることにより、細胞環境が改善し、様々な効能を得ることができる。
マグネシウムイオンは、細胞中に多く含まれており、生命維持に欠かせない主要なミネラルの1つである。細胞中のマグネシウムイオンが欠乏すると、細胞環境が悪化し、様々な疾病を引き起こすと考えられる。組成物によって、マグネシウムが粘膜組織から吸収されることにより、細胞環境が改善し、様々な効能を得ることが可能となる。
ナトリウムは、マグネシウム溶液に含まれることにより、マグネシウムの効果を促進させることができる。しかしながら、細胞環境を改善するという上記マグネシウムの効果を相乗的に高めるためには、マグネシウムとナトリウムとの含有比(以下、「Mg:Na比」ともいう。)が重要であると考えられる。細胞環境を改善するという効果を得るためには、マグネシウムとナトリウムとの含有比は、ナトリウムに対してマグネシウムが16倍以上含まれることが好ましく、40倍以上含まれることが特に好ましい。現在入手可能なマグネシウム溶液として、マグネシウム:ナトリウム=73:1のマグネシウム溶液(ニュー・サイエンス社製、商品名「超高濃度マグネシウム」)がある。
ラクトフェリンは、水溶性組成物に泡立ち性を付与することができる。ラクトフェリンを含むことで、ブラッシング時または含嗽時に、組成物が口内に長く留まり、マグネシウムの吸収をより効果的に高めることが可能となる。水溶性組成物は、ラクトフェリンの含有率が、3.0~10.0w/v%であることが好ましい。
精製水に所定量のラクトフェリンを溶解させ、ラクトフェリン濃度を1.0w/v%ずつ増やして1.0~10.0w/v%の10種類の調製液を調製した。各調製液を歯ブラシ(ライオン社製、クリニカ(登録商標)アドバンテージNEXT STAGE ハブラシ)に所定量付け、ふるいの網目上で200回ブラッシングした。なお、上記歯ブラシは、ブラッシングの際の圧力が、一定の圧力を超えると音と振動で知らせるものである。200回ブラッシング後、ふるいの表裏を観察して泡立ち性を評価した。
水溶性組成物は、公知成分を適宜本開示の効果を妨げない範囲で配合し、様々な剤形を採用することにより、細菌性の炎症に対する多様な抗炎症効果を得ることが可能である。したがって、組成物が歯磨剤の助剤としても振る舞いうる。
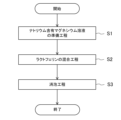
水溶性組成物の製造方法は、図3に示す手順で行う。
水溶性組成物が細菌にどのような影響を与えるかを、位相差顕微鏡を用いて評価した。具体的な評価方法は以下である。歯科受診患者から得られたプラークをスライドガラスに所定量分取し、そこへ下記表1に示す組成でそれぞれ調製した溶液を滴下した。スライドガラス上の試料にカバーガラスを載置して、各サンプルを得た。得られた各サンプルを、位相差顕微鏡(ピーテック社製、P-SCOPE Pro)を用いて3分間観察した。
まず、上記プラークのみをスライドガラスに塗布し、カバーガラスを置いて載置したものを、位相差顕微鏡で観察した。位相差顕微鏡の画像を図4に示す。この比較例1では、多数の細菌が活発に動く様子が観察された。
マグネシウム含有率10.5w/v%、ナトリウム含有率0.25w/v%、カリウム含有率1.18w/v%及びラクトフェリン含有率4.5w/v%の水溶性組成物(以下、試料1ともいう。)を調製した。具体的には、まず、マグネシウム含有率が10.5w/v%であり、かつ、Mg:Na比が42.7:1であるナトリウム含有マグネシウム溶液(メイティア社製、マグネフォース(登録商標))を準備した。そして、ナトリウム含有マグネシウム溶液10mLにラクトフェリン0.45gを混合し、試料1の前駆物質である混合溶液を得た。その後、この混合溶液を常温常圧で3日間静置し、消泡させることにより、試料1を調製した。この試料1をプラークへ滴下した際の位相差顕微鏡による画像を図5に示す。この実施例1では、細菌は全く動いておらず、試料1に細菌の働きを抑制する効果があることが確認できた。
上記試料1の調製において、ナトリウム含有マグネシウム溶液(メイティア社製、マグネフォース(登録商標))にさらに塩化ナトリウムを0.1g添加して、Mg:Na比を16.4:1とした以外は同様にして、試料1Aを調製した。この試料1Aをプラークへ滴下して位相差顕微鏡による観察をおこなったところ、実施例1と同様に、細菌は全く動いておらず、試料1Aに細菌の働きを抑制する効果があることが確認できた。
マグネシウム含有率が10.5w/v%であり、かつ、Mg:Na比が42.7:1であるナトリウム含有マグネシウム溶液(メイティア社製、マグネフォース(登録商標))16mLに、精製水14mLを加えて希釈し、そこへ塩化ナトリウム0.11gを添加した。マグネシウムの含有率5.6w/v%、Mg:Na比20.3:1である試料2Aを調製した。この試料2Aをプラークへ滴下して位相差顕微鏡による観察をおこなったところ、比較例1と同様に、多数の細菌が活発に動く様子が観察された。
上記試料2Aの調製において、塩化ナトリウム0.11g添加したことに代えて、塩化ナトリウムを0.15g添加したこと以外は同様にして、Mg:Na比10.7:1である試料2Bを調製した。この試料2Bをプラークへ滴下して位相差顕微鏡による観察をおこなったところ、比較例1と同様に、多数の細菌が活発に動く様子が観察された。
市販されるにがり(白松社製、商品名「浜御塩の海水にがり」)20mLにラクトフェリン0.6gを混合し、試料2Cを調製した。試料2Cは、マグネシウム含有率5.6w/v%、ラクトフェリン含有率3.0w/v%、Mg:Na比1.3:1である。この試料2Cをプラークへ滴下して位相差顕微鏡による観察をおこなったところ、比較例1と同様に、多数の細菌が活発に動く様子が観察された。
市販されるにがり(白松社製、商品名「浜御塩の海水にがり」)10mLを精製水15mLで希釈した溶液に、ラクトフェリン0.75gを混合し、試料2Dを調製した。試料2Dは、マグネシウム含有率2.2w/v%、ラクトフェリン含有率3.0w/v%、Mg:Na比1.3:1である。この試料2Dをプラークへ滴下して位相差顕微鏡による観察をおこなったところ、比較例1と同様に、多数の細菌が活発に動く様子が観察された。
ナトリウム含有マグネシウム溶液(メイティア社製、マグネフォース(登録商標))とラクトフェリンとを用いて、マグネシウム含有率8.8w/v%、ラクトフェリン含有率3.9w/v%、Mg:Na比47.7:1である試料3Aを調製した。この試料3Aをプラークへ滴下して位相差顕微鏡による観察をおこなったところ、実施例1と同様に、細菌はほとんど動いておらず、細菌が静菌された様子を観察できた。
ナトリウム含有マグネシウム溶液(メイティア社製、マグネフォース(登録商標))とラクトフェリンとを用いて、マグネシウム含有率8.0w/v%、ラクトフェリン含有率3.5w/v%、Mg:Na比47.4:1である試料3Bを調製した。この試料3Bをプラークへ滴下して位相差顕微鏡による観察をおこなった。実施例4では、実施例1から3ほど明確な静菌の様子は観察されなかったが、比較例1から比較例5と比較すると、菌の動きは弱まっており、静菌の傾向にあると言えた。
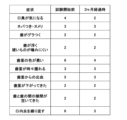
17名の被験者を募り、3ヶ月間、本開示の水溶性組成物を歯磨き剤として使用させた。被験者には、可能な限り、歯磨き時のうがいの水は吐き出さず、可能な限り水溶性組成物を飲み込むように指示した。被験者には、試験開始前と3ヶ月経過時において、以下10の自覚的評価項目を0~10の整数で評価させた。例えば、各項目について、「強く感じる」又は「とても当てはまる」場合は10点であり、「全く感じない」又は「全く当てはまらない」場合は0点である。
・口臭が気になる
・ネバつき・ヌメり
・歯がぐらつく
・歯が浮く、硬いものが噛みにくい
・歯茎の色が悪い
・歯茎が時々腫れる
・歯茎からの出血
・歯茎が下がってきた
・歯と歯の隙間が空いてきた
・口内炎を繰り返す
自覚的評価チェックシートの記入例として、図6に被験者007の評価結果を示す。いくつかの項目について、試験開始前と比して3ヶ月経過時の点数が小さくなっており、症状が改善されていた。
歯磨き剤I:ニュー・サイエンス社製、マグネシウム歯磨きペースト。塩化マグネシウム(MgCl・6H2O)を含有する。マグネシウム含有量から換算したマグネシウム含有率は、1.8w/v%である。ナトリウム、ラクトフェリンを含有しない。
歯磨き剤II:花王社製、クリアクリーン(登録商標)プレミアム美白。
歯磨き剤III:グラクソ・スミスクライン社製、ニューアクアフレッシュ(登録商標)ZF3。
歯磨き剤IV:サンスター社製、Ora2(登録商標)プレミアムステインクリア。
歯磨き剤V:グラクソ・スミスクライン社製、カムテクト(登録商標)コンプリートケアEX。
歯磨き剤VI:シャボン玉石けん社製、シャボン玉せっけん(登録商標)ハミガキ。
歯磨き剤VII:花王社製、クリアクリーン(登録商標)RR。
歯磨き剤VIII:サンスター社製、薬用G・U・M(登録商標)デンタルペーストGS。
歯磨き剤IX:小林製薬社製、生薬Hc。
歯磨き剤II~IXは、マグネシウム及びラクトフェリンを含有しない。
歯磨き剤X:AT-MARK CONSUL.社製、B+。化石サンゴを主成分とする。公知の分析例によれば、天然化石サンゴのマグネシウム含有率は1%未満であることから、この歯磨き剤に含まれるマグネシウム含有率も1w/v%未満であると考えられる。ラクトフェリンを含有しない。
歯磨き剤XI:ライオン社製、デンターシステマ(登録商標)EXW。マグネシウム及びラクトフェリンを含有しない。
上記17名の被験者のうち、一部の被験者に、歯科にて、試験前後で同一の施術者による他覚的評価を行わせた。具体的には、歯周ポケット深さと出血の有無の確認する、プロービング検査を行った。
Claims (4)
- マグネシウムと、ナトリウムと、タンパク質とを含み、
マグネシウムとナトリウムとの含有比が、マグネシウム:ナトリウム=16:1~73:1であり、
タンパク質は、ラクトフェリンであり、
マグネシウムの含有率が、2.0~12.0w/v%であり、
ラクトフェリンの含有率が、3.0~10.0w/v%である、水溶性組成物。 - マグネシウムの含有率が、8.0~12.0w/v%である、請求項1に記載の水溶性組成物。
- マグネシウムの含有率が、10.5~12.0w/v%であり、
口腔用である、請求項1に記載の水溶性組成物。 - 下記工程を含む、請求項1に記載の水溶性組成物の製造方法:
マグネシウムとナトリウムとの含有比が、マグネシウム:ナトリウム=16:1~73:1であるナトリウム含有マグネシウム溶液を準備する工程、
ラクトフェリン及びナトリウム含有マグネシウム溶液を混合し、マグネシウムの含有率が2.0~12.0w/v%であり、ラクトフェリンの含有率が3.0~10.0w/v%である混合溶液を得る工程、および
前記混合溶液を消泡する工程。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP2021/046291 WO2023112209A1 (ja) | 2021-12-15 | 2021-12-15 | 水溶性組成物及びその製造方法 |
| AU2021478651A AU2021478651B2 (en) | 2021-12-15 | 2021-12-15 | Water-soluble composition and production method thereof |
| JP2022548951A JP7177568B1 (ja) | 2021-12-15 | 2021-12-15 | 水溶性組成物及びその製造方法 |
| EP21967440.5A EP4331591A4 (en) | 2021-12-15 | 2021-12-15 | WATER-SOLUBLE COMPOSITION AND METHOD FOR PRODUCING SAME |
| TW111147892A TWI805533B (zh) | 2021-12-15 | 2022-12-14 | 水溶性組成物及其製造方法 |
| US18/525,147 US11957738B1 (en) | 2021-12-15 | 2023-11-30 | Water soluble composition and production method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP2021/046291 WO2023112209A1 (ja) | 2021-12-15 | 2021-12-15 | 水溶性組成物及びその製造方法 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US18/525,147 Continuation US11957738B1 (en) | 2021-12-15 | 2023-11-30 | Water soluble composition and production method thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023112209A1 true WO2023112209A1 (ja) | 2023-06-22 |
Family
ID=84178883
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2021/046291 WO2023112209A1 (ja) | 2021-12-15 | 2021-12-15 | 水溶性組成物及びその製造方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US11957738B1 (ja) |
| EP (1) | EP4331591A4 (ja) |
| JP (1) | JP7177568B1 (ja) |
| AU (1) | AU2021478651B2 (ja) |
| TW (1) | TWI805533B (ja) |
| WO (1) | WO2023112209A1 (ja) |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5898037A (en) * | 1992-11-13 | 1999-04-27 | Marx; Alvin J. | Formulations of magnesium compounds for local application and methods of treatment using the same |
| JP2003246738A (ja) * | 2001-07-02 | 2003-09-02 | Meiteia:Kk | マグネシウム強化液並びにそれを用いた保湿剤、化粧品及び飲料 |
| JP2007223975A (ja) * | 2006-02-24 | 2007-09-06 | Sunstar Inc | ラクトフェリン内包リポソームを含有する口腔用組成物 |
| CN102112101A (zh) * | 2008-07-07 | 2011-06-29 | 《Wds》公司 | 用于口腔护理的治疗性和预防性制剂 |
| JP2013538199A (ja) * | 2010-08-07 | 2013-10-10 | ザ・リサーチ・フアウンデーシヨン・フオー・ザ・ステート・ユニバーシテイ・オブ・ニユー・ヨーク | 亜鉛化合物および抗微生物剤を含む口腔用組成物 |
| JP2016175901A (ja) * | 2015-03-18 | 2016-10-06 | 第一三共ヘルスケア株式会社 | L−アスパラギン酸またはその塩、及びニコチンアミドを含有する口腔ケア用組成物 |
| JP2016210758A (ja) * | 2015-05-07 | 2016-12-15 | ジャパンモード株式会社 | 口腔ケア組成物、錠剤、顆粒状薬剤 |
| JP2018016553A (ja) * | 2016-07-25 | 2018-02-01 | 森永乳業株式会社 | 口腔内細菌叢改善剤 |
| JP2018043982A (ja) | 2016-09-07 | 2018-03-22 | 第一三共ヘルスケア株式会社 | カチオン性殺菌剤、未修飾シリカ、及びベタイン系界面活性剤を含む口腔用組成物 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20030003059A1 (en) * | 2001-04-03 | 2003-01-02 | Frederic Dana | Dentifrice compositions |
| WO2016174861A1 (ja) * | 2015-04-27 | 2016-11-03 | ジャパンモード株式会社 | 口腔ケア組成物 |
| US20180117070A1 (en) | 2015-04-27 | 2018-05-03 | Japan Mode Co. Ltd. | Oral care composition |
-
2021
- 2021-12-15 WO PCT/JP2021/046291 patent/WO2023112209A1/ja active Application Filing
- 2021-12-15 JP JP2022548951A patent/JP7177568B1/ja active Active
- 2021-12-15 EP EP21967440.5A patent/EP4331591A4/en active Pending
- 2021-12-15 AU AU2021478651A patent/AU2021478651B2/en active Active
-
2022
- 2022-12-14 TW TW111147892A patent/TWI805533B/zh active
-
2023
- 2023-11-30 US US18/525,147 patent/US11957738B1/en active Active
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5898037A (en) * | 1992-11-13 | 1999-04-27 | Marx; Alvin J. | Formulations of magnesium compounds for local application and methods of treatment using the same |
| JP2003246738A (ja) * | 2001-07-02 | 2003-09-02 | Meiteia:Kk | マグネシウム強化液並びにそれを用いた保湿剤、化粧品及び飲料 |
| JP2007223975A (ja) * | 2006-02-24 | 2007-09-06 | Sunstar Inc | ラクトフェリン内包リポソームを含有する口腔用組成物 |
| CN102112101A (zh) * | 2008-07-07 | 2011-06-29 | 《Wds》公司 | 用于口腔护理的治疗性和预防性制剂 |
| JP2013538199A (ja) * | 2010-08-07 | 2013-10-10 | ザ・リサーチ・フアウンデーシヨン・フオー・ザ・ステート・ユニバーシテイ・オブ・ニユー・ヨーク | 亜鉛化合物および抗微生物剤を含む口腔用組成物 |
| JP2016175901A (ja) * | 2015-03-18 | 2016-10-06 | 第一三共ヘルスケア株式会社 | L−アスパラギン酸またはその塩、及びニコチンアミドを含有する口腔ケア用組成物 |
| JP2016210758A (ja) * | 2015-05-07 | 2016-12-15 | ジャパンモード株式会社 | 口腔ケア組成物、錠剤、顆粒状薬剤 |
| JP2018016553A (ja) * | 2016-07-25 | 2018-02-01 | 森永乳業株式会社 | 口腔内細菌叢改善剤 |
| JP2018043982A (ja) | 2016-09-07 | 2018-03-22 | 第一三共ヘルスケア株式会社 | カチオン性殺菌剤、未修飾シリカ、及びベタイン系界面活性剤を含む口腔用組成物 |
Non-Patent Citations (4)
| Title |
|---|
| KOBAYASHI TETSUO: "Effects of lactoferrin intake in patients with periodontal disease", LACTOFERRIN NEWS, vol. 3, 1 September 2011 (2011-09-01), pages 1 - 14, XP093072059 * |
| KONDO ICHIRO, KOBAYASHI TETSUO, WAKABAYASHI HIROYUKI, YAMAUCHI KOJI, IWATSUKI KEIJI, YOSHE HIROMASA: "Effects of Oral Administration of Bovine Lactoferrin on Periodontitis Patients", THE JAPANESE JOURNAL OF CONSERVATIVE DENTISTRY, vol. 51, no. 3, 1 January 2008 (2008-01-01), pages 281 - 291, XP093072056 * |
| PRESENTFUTURE: "Bittern Industry", FOCUS ON BROMINE, BULLETIN OF THE SOCIETY OF SEA WATER SCIENCE, JAPAN, vol. 34, no. 4, 1980, pages 217 - 23 |
| See also references of EP4331591A4 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2023112209A1 (ja) | 2023-06-22 |
| JP7177568B1 (ja) | 2022-11-24 |
| US11957738B1 (en) | 2024-04-16 |
| TW202325318A (zh) | 2023-07-01 |
| AU2021478651B2 (en) | 2024-01-25 |
| EP4331591A4 (en) | 2024-06-26 |
| EP4331591A1 (en) | 2024-03-06 |
| TWI805533B (zh) | 2023-06-11 |
| US20240100130A1 (en) | 2024-03-28 |
| AU2021478651A1 (en) | 2023-12-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4697497B2 (ja) | 湿潤させる製剤 | |
| WO2007061328A1 (fr) | Composition de soins et de prevention pour la cavite buccale | |
| JP2007169181A (ja) | 歯牙貼付用製品及び口腔ケア方法 | |
| JP2008143825A (ja) | 歯磨組成物 | |
| KR20190055561A (ko) | 잇몸질환 예방 및 개선용 치약 조성물 | |
| US20120288451A1 (en) | Means for removing tobacco tars and composition containing same | |
| JP4826790B2 (ja) | 加水分解シルクを含有する口腔用組成物 | |
| JP5527219B2 (ja) | 練歯磨組成物 | |
| CN104288466B (zh) | 一种医用生物氟防龋敷料及其制备方法 | |
| JP2007197393A (ja) | 歯磨 | |
| JP2006182705A (ja) | 歯牙貼付用製品及び口腔ケア物質の供給方法 | |
| RU2457829C2 (ru) | Минерально-витаминный комплекс для укрепления эмали зубов, композиция для гигиены полости рта и зубная паста | |
| US11723847B2 (en) | Dual component oral composition for enhancing remineralization of teeth | |
| JP7177568B1 (ja) | 水溶性組成物及びその製造方法 | |
| US11564878B2 (en) | Oral care product formulation | |
| KR980008210A (ko) | 구강위생 증진용 조성물 | |
| JPH01287015A (ja) | 歯牙口腔液体洗滌剤 | |
| KR0170108B1 (ko) | 유창목 수지 및 발색 조성물을 함유한 치약 조성물 | |
| AU2021225354B2 (en) | Composition for the prevention and treatment of dysbiosis | |
| US20240148619A1 (en) | Dual component oral composition for enhancing remineralization of teeth | |
| RU2455975C2 (ru) | Зубная паста | |
| RU2453300C2 (ru) | Зубные пасты на основе металлокомплекса цинка с производными 1-алкенилимидазола | |
| RU2457824C2 (ru) | Лечебно-профилактическая зубная паста "stomatol" | |
| KR20140146301A (ko) | 구강청결제 조성물과 이를 이용한 구강용 제품 | |
| JP2005298346A (ja) | 口腔用組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022548951 Country of ref document: JP |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21967440 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2021478651 Country of ref document: AU Ref document number: 2021967440 Country of ref document: EP Ref document number: AU2021478651 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: 2021967440 Country of ref document: EP Effective date: 20231129 |
|
| ENP | Entry into the national phase |
Ref document number: 2021478651 Country of ref document: AU Date of ref document: 20211215 Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |