WO2022209894A1 - 非水電解質二次電池用正極活物質および非水電解質二次電池 - Google Patents
非水電解質二次電池用正極活物質および非水電解質二次電池 Download PDFInfo
- Publication number
- WO2022209894A1 WO2022209894A1 PCT/JP2022/011852 JP2022011852W WO2022209894A1 WO 2022209894 A1 WO2022209894 A1 WO 2022209894A1 JP 2022011852 W JP2022011852 W JP 2022011852W WO 2022209894 A1 WO2022209894 A1 WO 2022209894A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- positive electrode
- active material
- electrode active
- composite oxide
- aqueous electrolyte
- Prior art date
Links
- 239000007774 positive electrode material Substances 0.000 title claims abstract description 51
- 239000011255 nonaqueous electrolyte Substances 0.000 title claims abstract description 30
- 150000001875 compounds Chemical class 0.000 claims abstract description 60
- 239000011163 secondary particle Substances 0.000 claims abstract description 29
- 229910052744 lithium Inorganic materials 0.000 claims abstract description 26
- 239000011164 primary particle Substances 0.000 claims abstract description 26
- 229910052760 oxygen Inorganic materials 0.000 claims abstract description 24
- 229910052723 transition metal Inorganic materials 0.000 claims abstract description 23
- 239000002905 metal composite material Substances 0.000 claims abstract description 19
- -1 lithium transition metal Chemical class 0.000 claims abstract description 14
- 229910052721 tungsten Inorganic materials 0.000 claims abstract description 12
- 229910052791 calcium Inorganic materials 0.000 claims abstract description 9
- 229910052750 molybdenum Inorganic materials 0.000 claims abstract description 9
- 229910052710 silicon Inorganic materials 0.000 claims abstract description 9
- 229910052719 titanium Inorganic materials 0.000 claims abstract description 9
- 229910052758 niobium Inorganic materials 0.000 claims abstract description 8
- 229910052712 strontium Inorganic materials 0.000 claims abstract description 8
- 229910052726 zirconium Inorganic materials 0.000 claims abstract description 8
- 229910052796 boron Inorganic materials 0.000 claims abstract description 6
- 230000002776 aggregation Effects 0.000 claims abstract description 5
- 229910052748 manganese Inorganic materials 0.000 claims description 10
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 10
- 229910052782 aluminium Inorganic materials 0.000 claims description 8
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 claims description 6
- 238000005054 agglomeration Methods 0.000 claims description 4
- 239000003792 electrolyte Substances 0.000 claims description 3
- 229910052742 iron Inorganic materials 0.000 claims description 3
- 238000004220 aggregation Methods 0.000 abstract 1
- 239000002131 composite material Substances 0.000 description 52
- 239000000203 mixture Substances 0.000 description 36
- 238000012360 testing method Methods 0.000 description 33
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 32
- 239000010410 layer Substances 0.000 description 26
- 230000015572 biosynthetic process Effects 0.000 description 18
- 230000000052 comparative effect Effects 0.000 description 18
- 238000003786 synthesis reaction Methods 0.000 description 18
- 238000010304 firing Methods 0.000 description 17
- 230000014759 maintenance of location Effects 0.000 description 14
- 238000007789 sealing Methods 0.000 description 12
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 11
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 11
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 11
- 239000000920 calcium hydroxide Substances 0.000 description 11
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 11
- 239000001301 oxygen Substances 0.000 description 11
- 239000002245 particle Substances 0.000 description 11
- 229910052759 nickel Inorganic materials 0.000 description 10
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 9
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 9
- 239000011575 calcium Substances 0.000 description 9
- QGLKJKCYBOYXKC-UHFFFAOYSA-N nonaoxidotritungsten Chemical compound O=[W]1(=O)O[W](=O)(=O)O[W](=O)(=O)O1 QGLKJKCYBOYXKC-UHFFFAOYSA-N 0.000 description 9
- UUCCCPNEFXQJEL-UHFFFAOYSA-L strontium dihydroxide Chemical compound [OH-].[OH-].[Sr+2] UUCCCPNEFXQJEL-UHFFFAOYSA-L 0.000 description 9
- 229910001866 strontium hydroxide Inorganic materials 0.000 description 9
- 229910001930 tungsten oxide Inorganic materials 0.000 description 9
- 230000007423 decrease Effects 0.000 description 8
- 229910000476 molybdenum oxide Inorganic materials 0.000 description 8
- PQQKPALAQIIWST-UHFFFAOYSA-N oxomolybdenum Chemical compound [Mo]=O PQQKPALAQIIWST-UHFFFAOYSA-N 0.000 description 8
- 238000007600 charging Methods 0.000 description 7
- 238000010438 heat treatment Methods 0.000 description 7
- 229910052751 metal Inorganic materials 0.000 description 7
- 239000002184 metal Substances 0.000 description 7
- 150000003839 salts Chemical class 0.000 description 7
- 239000011230 binding agent Substances 0.000 description 6
- 238000007599 discharging Methods 0.000 description 6
- 238000001035 drying Methods 0.000 description 6
- 239000010936 titanium Substances 0.000 description 6
- 239000011149 active material Substances 0.000 description 5
- 239000003125 aqueous solvent Substances 0.000 description 5
- 239000006258 conductive agent Substances 0.000 description 5
- 239000010408 film Substances 0.000 description 5
- 229910002804 graphite Inorganic materials 0.000 description 5
- 239000010439 graphite Substances 0.000 description 5
- 239000012535 impurity Substances 0.000 description 5
- 238000004519 manufacturing process Methods 0.000 description 5
- 238000000034 method Methods 0.000 description 5
- 239000007773 negative electrode material Substances 0.000 description 5
- LLZRNZOLAXHGLL-UHFFFAOYSA-J titanic acid Chemical compound O[Ti](O)(O)O LLZRNZOLAXHGLL-UHFFFAOYSA-J 0.000 description 5
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 4
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 4
- 239000003575 carbonaceous material Substances 0.000 description 4
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 4
- 239000011888 foil Substances 0.000 description 4
- 150000002642 lithium compounds Chemical class 0.000 description 4
- 239000002002 slurry Substances 0.000 description 4
- 238000003466 welding Methods 0.000 description 4
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 3
- 239000002033 PVDF binder Substances 0.000 description 3
- 239000004642 Polyimide Substances 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000012046 mixed solvent Substances 0.000 description 3
- 229920002239 polyacrylonitrile Polymers 0.000 description 3
- 229920001721 polyimide Polymers 0.000 description 3
- 229920000098 polyolefin Polymers 0.000 description 3
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 229920005989 resin Polymers 0.000 description 3
- 239000011347 resin Substances 0.000 description 3
- 239000002344 surface layer Substances 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- 229920000178 Acrylic resin Polymers 0.000 description 2
- 239000004925 Acrylic resin Substances 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 229910013870 LiPF 6 Inorganic materials 0.000 description 2
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 description 2
- 229920002125 Sokalan® Polymers 0.000 description 2
- 229910003514 Sr(OH) Inorganic materials 0.000 description 2
- 239000006230 acetylene black Substances 0.000 description 2
- 229910021383 artificial graphite Inorganic materials 0.000 description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 238000000975 co-precipitation Methods 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- IEJIGPNLZYLLBP-UHFFFAOYSA-N dimethyl carbonate Chemical compound COC(=O)OC IEJIGPNLZYLLBP-UHFFFAOYSA-N 0.000 description 2
- 239000002612 dispersion medium Substances 0.000 description 2
- IIPYXGDZVMZOAP-UHFFFAOYSA-N lithium nitrate Chemical compound [Li+].[O-][N+]([O-])=O IIPYXGDZVMZOAP-UHFFFAOYSA-N 0.000 description 2
- INHCSSUBVCNVSK-UHFFFAOYSA-L lithium sulfate Chemical compound [Li+].[Li+].[O-]S([O-])(=O)=O INHCSSUBVCNVSK-UHFFFAOYSA-L 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 description 2
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 2
- 239000004810 polytetrafluoroethylene Substances 0.000 description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 239000002210 silicon-based material Substances 0.000 description 2
- 229920003048 styrene butadiene rubber Polymers 0.000 description 2
- 229910052718 tin Inorganic materials 0.000 description 2
- 150000003624 transition metals Chemical class 0.000 description 2
- 229910001928 zirconium oxide Inorganic materials 0.000 description 2
- 229910000838 Al alloy Inorganic materials 0.000 description 1
- 238000004438 BET method Methods 0.000 description 1
- 229910004648 CaMoO3 Inorganic materials 0.000 description 1
- 229910004647 CaMoO4 Inorganic materials 0.000 description 1
- 229910002971 CaTiO3 Inorganic materials 0.000 description 1
- 229910004829 CaWO4 Inorganic materials 0.000 description 1
- 229910002976 CaZrO3 Inorganic materials 0.000 description 1
- 229910052684 Cerium Inorganic materials 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229910000881 Cu alloy Inorganic materials 0.000 description 1
- 229910052692 Dysprosium Inorganic materials 0.000 description 1
- 229910052691 Erbium Inorganic materials 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 229910052693 Europium Inorganic materials 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 229910052688 Gadolinium Inorganic materials 0.000 description 1
- 229910052689 Holmium Inorganic materials 0.000 description 1
- FUJCRWPEOMXPAD-UHFFFAOYSA-N Li2O Inorganic materials [Li+].[Li+].[O-2] FUJCRWPEOMXPAD-UHFFFAOYSA-N 0.000 description 1
- 229910001323 Li2O2 Inorganic materials 0.000 description 1
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 1
- 229910052779 Neodymium Inorganic materials 0.000 description 1
- BPQQTUXANYXVAA-UHFFFAOYSA-N Orthosilicate Chemical compound [O-][Si]([O-])([O-])[O-] BPQQTUXANYXVAA-UHFFFAOYSA-N 0.000 description 1
- 239000004962 Polyamide-imide Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 229910052777 Praseodymium Inorganic materials 0.000 description 1
- 229910052772 Samarium Inorganic materials 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 229910002412 SrMoO4 Inorganic materials 0.000 description 1
- 229910003383 SrSiO3 Inorganic materials 0.000 description 1
- 229910002370 SrTiO3 Inorganic materials 0.000 description 1
- 229910004415 SrWO4 Inorganic materials 0.000 description 1
- 239000002174 Styrene-butadiene Substances 0.000 description 1
- 229910052771 Terbium Inorganic materials 0.000 description 1
- 229910052775 Thulium Inorganic materials 0.000 description 1
- 229910052769 Ytterbium Inorganic materials 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 230000004931 aggregating effect Effects 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 239000012670 alkaline solution Substances 0.000 description 1
- 229910045601 alloy Inorganic materials 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- IZJSTXINDUKPRP-UHFFFAOYSA-N aluminum lead Chemical compound [Al].[Pb] IZJSTXINDUKPRP-UHFFFAOYSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 229910052787 antimony Inorganic materials 0.000 description 1
- 239000004760 aramid Substances 0.000 description 1
- 229920003235 aromatic polyamide Polymers 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- 229910000171 calcio olivine Inorganic materials 0.000 description 1
- 229910052918 calcium silicate Inorganic materials 0.000 description 1
- 235000012241 calcium silicate Nutrition 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 229910021393 carbon nanotube Inorganic materials 0.000 description 1
- 239000002041 carbon nanotube Substances 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 238000010280 constant potential charging Methods 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- XUCJHNOBJLKZNU-UHFFFAOYSA-M dilithium;hydroxide Chemical compound [Li+].[Li+].[OH-] XUCJHNOBJLKZNU-UHFFFAOYSA-M 0.000 description 1
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 1
- 230000003628 erosive effect Effects 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910052733 gallium Inorganic materials 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 229910052735 hafnium Inorganic materials 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- 230000020169 heat generation Effects 0.000 description 1
- 229920006015 heat resistant resin Polymers 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 229910052738 indium Inorganic materials 0.000 description 1
- 239000010954 inorganic particle Substances 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- 230000010220 ion permeability Effects 0.000 description 1
- 229910052741 iridium Inorganic materials 0.000 description 1
- 239000003273 ketjen black Substances 0.000 description 1
- 229910052746 lanthanum Inorganic materials 0.000 description 1
- 229910052745 lead Inorganic materials 0.000 description 1
- HEPLMSKRHVKCAQ-UHFFFAOYSA-N lead nickel Chemical compound [Ni].[Pb] HEPLMSKRHVKCAQ-UHFFFAOYSA-N 0.000 description 1
- 239000011244 liquid electrolyte Substances 0.000 description 1
- XGZVUEUWXADBQD-UHFFFAOYSA-L lithium carbonate Chemical compound [Li+].[Li+].[O-]C([O-])=O XGZVUEUWXADBQD-UHFFFAOYSA-L 0.000 description 1
- 229910052808 lithium carbonate Inorganic materials 0.000 description 1
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Chemical compound [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 1
- SIAPCJWMELPYOE-UHFFFAOYSA-N lithium hydride Chemical compound [LiH] SIAPCJWMELPYOE-UHFFFAOYSA-N 0.000 description 1
- 229910001416 lithium ion Inorganic materials 0.000 description 1
- PAZHGORSDKKUPI-UHFFFAOYSA-N lithium metasilicate Chemical compound [Li+].[Li+].[O-][Si]([O-])=O PAZHGORSDKKUPI-UHFFFAOYSA-N 0.000 description 1
- 229910003002 lithium salt Inorganic materials 0.000 description 1
- 159000000002 lithium salts Chemical class 0.000 description 1
- 229910052912 lithium silicate Inorganic materials 0.000 description 1
- GLXDVVHUTZTUQK-UHFFFAOYSA-M lithium;hydroxide;hydrate Chemical compound [Li+].O.[OH-] GLXDVVHUTZTUQK-UHFFFAOYSA-M 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 239000011325 microbead Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 229910021382 natural graphite Inorganic materials 0.000 description 1
- 150000002823 nitrates Chemical class 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 239000004745 nonwoven fabric Substances 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000004584 polyacrylic acid Substances 0.000 description 1
- 229920002312 polyamide-imide Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 229910052702 rhenium Inorganic materials 0.000 description 1
- 229910052703 rhodium Inorganic materials 0.000 description 1
- 229910052707 ruthenium Inorganic materials 0.000 description 1
- 239000012266 salt solution Substances 0.000 description 1
- 229910052706 scandium Inorganic materials 0.000 description 1
- 229910052814 silicon oxide Inorganic materials 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000007784 solid electrolyte Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 229910014031 strontium zirconium oxide Inorganic materials 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 238000002216 synchrotron radiation X-ray diffraction Methods 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 229910052720 vanadium Inorganic materials 0.000 description 1
- 229910052882 wollastonite Inorganic materials 0.000 description 1
- 239000002759 woven fabric Substances 0.000 description 1
- 229910052727 yttrium Inorganic materials 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000004711 α-olefin Substances 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/366—Composites as layered products
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/485—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of mixed oxides or hydroxides for inserting or intercalating light metals, e.g. LiTi2O4 or LiTi2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Definitions
- the present disclosure relates to a positive electrode active material for nonaqueous electrolyte secondary batteries and a nonaqueous electrolyte secondary battery using the active material.
- Patent Document 1 discloses a positive electrode in which an alkaline earth metal and W are present on the surface of secondary particles of a lithium-transition metal composite oxide containing one or more selected from Mn, Ni, and Co as a transition metal element. Active materials are disclosed.
- Patent Document 2 discloses a positive electrode active material containing one or more selected from Mn, Ni, and Co as a transition metal element, and further containing a lithium-transition metal composite oxide in which Ca and W are solid-dissolved. ing.
- Lithium-transition metal composite oxides with a high Ni content are known as high-capacity positive electrode active materials. There is a problem that the material deteriorates and the capacity decreases.
- An object of the present disclosure is to suppress the capacity decrease due to charging and discharging and improve cycle characteristics in a non-aqueous electrolyte secondary battery using a positive electrode active material with a high Ni content.
- a positive electrode active material for a non-aqueous electrolyte secondary battery according to the present disclosure has a layered structure and contains 75 mol% or more of Ni with respect to the total molar amount of elements excluding Li and O.
- Lithium transition metal composite oxide and the lithium -transition metal composite oxide is a secondary particle formed by agglomeration of primary particles. , 1 ⁇ x ⁇ 2, 1 ⁇ y ⁇ 5, 4 ⁇ z ⁇ 9, A is at least one selected from Ca and Sr, B is selected from W, Mo, Ti, Si, Nb, and Zr At least one compound represented by ) is fixed.
- a non-aqueous electrolyte secondary battery includes a positive electrode containing the positive electrode active material, a negative electrode, and a non-aqueous electrolyte.
- the positive electrode active material according to the present disclosure it is possible to improve the cycle characteristics of the non-aqueous electrolyte secondary battery.
- the non-aqueous electrolyte secondary battery according to the present disclosure has a small decrease in capacity due to charging and discharging, and is excellent in cycle characteristics.
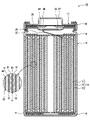
- FIG. 1 is a cross-sectional view of a non-aqueous electrolyte secondary battery that is an example of an embodiment
- the present inventors fixed the A x B y O z compound to the interface between the primary particles inside the secondary particles of the composite oxide in a high-capacity lithium-transition metal composite oxide with a high Ni content. As a result, the inventors have found that it is possible to effectively suppress the decrease in capacity that accompanies charging and discharging of the battery. Lithium - transition metal composite oxides with a high Ni content have an unstable surface layer structure and are easily eroded, as described above. It is believed that the surface is effectively protected and such erosion is inhibited.
- the AxByOz compound may exist on the surface of the secondary particles, but as in Comparative Example 5 described later, the AxByOz compound exists only on the surface of the secondary particles, and the two If it does not exist on the surface of the primary particles inside the secondary particles, the effect of improving cycle characteristics cannot be obtained. In addition, only one of the elements A and B cannot effectively protect the surfaces of the primary particles inside the secondary particles, and the effect of improving the cycle characteristics cannot be obtained. That is, only when the A x B y O z compound is present at the interface between the primary particles inside the secondary particles, the decrease in capacity due to charging and discharging is specifically suppressed, and the cycle characteristics are improved.
- a cylindrical battery in which the wound electrode body 14 is housed in a bottomed cylindrical outer can 16 will be exemplified. It may be a prismatic battery), a coin-shaped outer can (coin-shaped battery), or an outer body (laminate battery) composed of a laminate sheet including a metal layer and a resin layer. Further, the electrode assembly is not limited to the wound type, and may be a laminated electrode assembly in which a plurality of positive electrodes and a plurality of negative electrodes are alternately laminated with separators interposed therebetween.
- FIG. 1 is a cross-sectional view of a non-aqueous electrolyte secondary battery 10 that is an example of an embodiment.
- the non-aqueous electrolyte secondary battery 10 includes a wound electrode body 14, a non-aqueous electrolyte, and an outer can 16 that accommodates the electrode body 14 and the non-aqueous electrolyte.
- the electrode body 14 has a positive electrode 11, a negative electrode 12, and a separator 13, and has a wound structure in which the positive electrode 11 and the negative electrode 12 are spirally wound with the separator 13 interposed therebetween.
- the outer can 16 is a bottomed cylindrical metal container that is open on one side in the axial direction. In the following description, for convenience of explanation, the side of the sealing member 17 of the battery will be referred to as the upper side, and the bottom side of the outer can 16 will be referred to as the lower side.
- the non-aqueous electrolyte contains a non-aqueous solvent and an electrolyte salt dissolved in the non-aqueous solvent.
- non-aqueous solvents include esters, ethers, nitriles, amides, and mixed solvents of two or more thereof.
- the non-aqueous solvent may contain a halogen-substituted product obtained by substituting at least part of the hydrogen atoms of these solvents with halogen atoms such as fluorine.
- non-aqueous solvents include ethylene carbonate (EC), ethylmethyl carbonate (EMC), dimethyl carbonate (DMC), mixed solvents thereof, and the like.
- a lithium salt such as LiPF 6 is used as the electrolyte salt.
- the non-aqueous electrolyte is not limited to a liquid electrolyte, and may be a solid electrolyte.
- the positive electrode 11, the negative electrode 12, and the separator 13, which constitute the electrode assembly 14, are all strip-shaped elongated bodies, and are alternately laminated in the radial direction of the electrode assembly 14 by being spirally wound.
- the negative electrode 12 is formed with a size one size larger than that of the positive electrode 11 in order to prevent deposition of lithium. That is, the negative electrode 12 is formed longer than the positive electrode 11 in the longitudinal direction and the width direction (transverse direction).
- the separator 13 is at least one size larger than the positive electrode 11, and two separators 13 are arranged so as to sandwich the positive electrode 11, for example.
- the electrode body 14 has a positive electrode lead 20 connected to the positive electrode 11 by welding or the like, and a negative electrode lead 21 connected to the negative electrode 12 by welding or the like.
- Insulating plates 18 and 19 are arranged above and below the electrode body 14, respectively.
- the positive electrode lead 20 extends through the through hole of the insulating plate 18 toward the sealing member 17
- the negative electrode lead 21 extends through the outside of the insulating plate 19 toward the bottom of the outer can 16 .
- the positive electrode lead 20 is connected to the lower surface of the internal terminal plate 23 of the sealing body 17 by welding or the like, and the cap 27, which is the top plate of the sealing body 17 electrically connected to the internal terminal plate 23, serves as the positive electrode terminal.
- the negative electrode lead 21 is connected to the inner surface of the bottom of the outer can 16 by welding or the like, and the outer can 16 serves as a negative electrode terminal.
- a gasket 28 is provided between the outer can 16 and the sealing body 17 to ensure hermeticity inside the battery.
- the outer can 16 is formed with a grooved portion 22 that supports the sealing member 17 and has a portion of the side surface projecting inward.
- the grooved portion 22 is preferably annularly formed along the circumferential direction of the outer can 16 and supports the sealing member 17 on its upper surface.
- the sealing member 17 is fixed to the upper portion of the outer can 16 by the grooved portion 22 and the open end of the outer can 16 that is crimped to the sealing member 17 .
- the sealing body 17 has a structure in which an internal terminal plate 23, a lower valve body 24, an insulating member 25, an upper valve body 26, and a cap 27 are layered in order from the electrode body 14 side.
- Each member constituting the sealing member 17 has, for example, a disk shape or a ring shape, and each member except for the insulating member 25 is electrically connected to each other.
- the lower valve body 24 and the upper valve body 26 are connected at their central portions, and an insulating member 25 is interposed between their peripheral edge portions.
- the positive electrode 11, the negative electrode 12, and the separator 13 that make up the electrode body 14 will be described in detail below, particularly the positive electrode active material that makes up the positive electrode 11.
- the positive electrode 11 includes a positive electrode core 30 and a positive electrode mixture layer 31 provided on the surface of the positive electrode core 30 .
- a foil of a metal such as aluminum or an aluminum alloy that is stable in the potential range of the positive electrode 11, a film having the metal on the surface layer, or the like can be used.
- the positive electrode mixture layer 31 contains a positive electrode active material, a binder, and a conductive agent, and is preferably provided on both surfaces of the positive electrode core 30 .
- the positive electrode 11 can be produced, for example, by applying slurry of a positive electrode mixture onto the positive electrode core 30 , drying the coating film, and then compressing it to form the positive electrode mixture layers 31 on both sides of the positive electrode core 30 . .
- binder contained in the positive electrode mixture layer 31 examples include fluororesins such as polytetrafluoroethylene (PTFE) and polyvinylidene fluoride (PVdF), polyacrylonitrile (PAN), polyimide, acrylic resins, and polyolefins. These resins may be used in combination with cellulose derivatives such as carboxymethyl cellulose (CMC) or salts thereof, polyethylene oxide (PEO), and the like.
- the content of the binder is, for example, 0.5 to 2 mass % with respect to the mass of the positive electrode mixture layer 31 .
- Examples of the conductive agent contained in the positive electrode mixture layer 31 include carbon materials such as carbon black, acetylene black, ketjen black, graphite, and carbon nanotubes.
- the content of the conductive agent is, for example, 0.5 to 10 mass % with respect to the mass of the positive electrode mixture layer 31 .
- the positive electrode active material contains a lithium transition metal composite oxide containing 75 mol% or more of Ni with respect to the total molar amount of elements excluding Li and O.
- the lithium-transition metal composite oxide has a layered crystal structure. Specific examples include a layered structure belonging to the space group R-3m or a layered structure belonging to the space group C2/m.
- the lithium-transition metal composite oxide is a secondary particle formed by agglomeration of a plurality of primary particles.
- the particle size of the primary particles is, for example, 0.05 ⁇ m to 1 ⁇ m. The particle size of primary particles is measured as the diameter of the circumscribed circle in a particle image observed by a scanning electron microscope (SEM).
- the positive electrode active material contains a composite oxide (Z) as a main component.
- the main component means the component with the highest mass ratio among the constituent components of the positive electrode active material.
- a composite oxide other than the composite oxide (Z) may be used in combination as the positive electrode active material in the positive electrode mixture layer 31, but the content of the composite oxide (Z) is preferably 50% by mass or more. Preferably, it may be substantially 100% by mass.
- the volume-based median diameter (D50) of the composite oxide (Z) is, for example, 3 ⁇ m to 30 ⁇ m, preferably 5 ⁇ m to 25 ⁇ m. Since the composite oxide (Z) is secondary particles formed by agglomeration of primary particles, the D50 of the composite oxide (Z) means the D50 of the secondary particles. D50 means a particle size at which the cumulative frequency is 50% from the smaller particle size in the volume-based particle size distribution, and is also called median diameter.
- the particle size distribution of the composite oxide (Z) can be measured using a laser diffraction particle size distribution analyzer (eg MT3000II manufactured by Microtrack Bell Co., Ltd.) using water as a dispersion medium.
- the BET specific surface area of the composite oxide (Z) is preferably 0.5-3.5 m 2 /g. If the BET specific surface area is within this range, the cycle characteristics can be improved without reducing the discharge capacity. If the BET specific surface area is smaller than this range, the reaction area decreases, which may result in a decrease in discharge capacity. On the other hand, when the BET specific surface area is larger than the above range, the surface cannot be sufficiently coated with the AxByOz compound alone , so that the effect of improving the cycle characteristics is reduced.
- the BET specific surface area is measured according to the BET method (nitrogen adsorption method) described in JIS R1626.
- the composite oxide (Z) contains 75 mol% Ni with respect to the total number of moles of the elements excluding Li and O.
- a high energy density battery can be obtained by setting the Ni content to 75 mol % or more.
- the upper limit of the Ni content is preferably 95 mol%. When the Ni content exceeds 95 mol %, it becomes difficult to ensure the stability of the layered structure of the composite oxide (Z), and cycle characteristics may deteriorate.
- An example of a suitable Ni content range is 80-95 mol %, or 85-95 mol %.
- Elements contained in the composite oxide (Z) include Li, O, Ni, Co, Mn, Al, Na, K, Mg, Ca, Sr, Ba, Sc, Y, La, Ce, Pr , Nd, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, Lu, Ge, Sn, Pb, Sc, Ti, Si, V, Cr, Fe, Cu, Zn, Ru, Rh, Re , Pd, Ir, Ag, Bi, Sb, B, Ga, In, P, Zr, Hf, Nb, Mo, W, and the like.
- the content of element M is preferably 5 to 25 mol % with respect to the total molar amount of elements excluding Li and O.
- the a which indicates the ratio of Li in the composite oxide (Z), more preferably satisfies 0.95 ⁇ a ⁇ 1.05, and particularly preferably satisfies 0.97 ⁇ a ⁇ 1.03.
- a is less than 0.95, the capacity may be lower than when a satisfies the above range.
- a 1.05 or more, a larger amount of Li compound is added than when a satisfies the above range, which may not be economical from the viewpoint of production cost.
- Co since Co is expensive, it is preferable to reduce the Co content in consideration of manufacturing costs.
- Al When Al is contained in the composite oxide (Z), e, which indicates the proportion of Al, more preferably satisfies 0.02 ⁇ e ⁇ 0.07. Since Al does not change its oxidation number during charging and discharging, it is considered that the structure of the transition metal layer is stabilized by being contained in the transition metal layer. On the other hand, if the Al content is too high, it leads to a decrease in capacity. Al, for example, may be uniformly dispersed in the layered structure of the lithium-transition metal composite oxide, or may be partially present in the layered structure.
- a x B y O z (wherein 1 ⁇ x ⁇ 2, 1 ⁇ y ⁇ 5, 4 ⁇ z ⁇ 9 , A is at least one selected from Ca and Sr, and B is at least one selected from W, Mo, Ti, Si, Nb, and Zr) (hereinafter, “A x B y O z compound”) is adhered.
- a x B y O z compound is present on the surface of the primary particles inside the secondary particles of the composite oxide (Z), corrosion and deterioration of the composite oxide (Z) are effectively suppressed, and the battery cycle characteristics are specifically improved.
- the presence of the A x B y O z compound can be confirmed by measuring the cross section of the secondary particles using TEM-EDX (transmission microscope-energy dispersive X-ray spectroscopy).
- the A x B y O z compound may, for example, be scattered on the surface of the primary particles, or may be present in a layer so as to widely cover the surface of the primary particles.
- the AxByOz compound may further exist on the surface of the secondary particles of the composite oxide ( Z ). That is, the A x B y O z compound is widely present on the surface of the primary particles inside and on the surface of the secondary particles.
- the secondary particles of the composite oxide (Z) are formed, for example, by aggregating five or more primary particles, and the surface area of the primary particles is larger inside than on the surface of the secondary particles.
- the A x B y O z compound is contained more inside than on the surface of the secondary particles.
- AxByOz compounds include CaWO4 , CaMoO3 , CaMoO4 , CaTiO3 , Ca2TiO4 , CaSiO3 , Ca2SiO4 , CaNbO3 , CaNb2O6 , CaZrO3 , CaZr . 4O9 , SrWO4 , SrMoO3 , SrMoO4 , SrTiO3 , Sr2TiO4 , SrSiO3 , Sr2SiO4 , SrNbO3 , SrNb2O6 , SrZrO3 , SrZr4O9 .
- the content of the element A in the AxByOz compound is 3 mol% or less with respect to the total molar amount of the elements excluding Li and O in the composite oxide ( Z ) and the AxByOz compound .
- cycle characteristics can be efficiently improved without problems such as increased resistance.
- the content of the element B in the AxByOz compound is 3 mol with respect to the total molar amount of the elements excluding Li and O in the composite oxide ( Z ) and the AxByOz compound . % or less.
- the content of the elements A and B in the AxByOz compound is, for example, 0.1 mol % or more. Part of the elements A and B added to form the A x B y O z compound may be dissolved in the composite oxide (Z).
- the manufacturing process of the composite oxide (Z) includes, for example, a first step of obtaining a composite oxide containing Ni or the like, a second step of mixing the composite oxide and a lithium compound to obtain a mixture, A third step of baking the mixture and a fourth step of washing the baked product with water and drying by heating are included.
- a x B y O z compound is obtained by adding a compound containing element A and a compound containing element B during the manufacturing process of composite oxide (Z), thereby forming a secondary It can be adhered to the interface between primary particles in the interior of the particles.
- a compound containing element B is added in the second step or the fourth step.
- a compound containing element A may likewise be added in the fourth step, but is preferably added in the second step.
- an alkaline solution such as sodium hydroxide is added dropwise to adjust the pH to the alkaline side (eg, 8.5 to 12.5).
- a composite hydroxide containing Ni and the element M is precipitated (coprecipitated).
- the firing temperature is not particularly limited, it is 300°C to 600°C as an example.
- the composite oxide obtained in the first step a lithium compound, a compound containing element A, and a compound containing element B are mixed.
- a compound containing element B may be added in the fourth step, as described above.
- lithium compounds include Li2CO3 , LiOH , Li2O2 , Li2O , LiNO3 , LiNO2 , Li2SO4 , LiOH.H2O , LiH, and LiF.
- the composite oxide and the lithium compound are preferably mixed at a molar ratio of 1:0.98 to 1:1.12 between the total amount of Ni and the element M and Li.
- Examples of compounds containing element A include Ca(OH) 2 , CaO, CaCO 3 , CaSO 4 , Ca(NO 3 ) 2 , Sr(OH) 2 , Sr(OH) 2.8H 2 O , Sr( OH) 2.H 2 O, SrO, SrCO 3 , SrSO 4 , Sr(NO 3 ) 2 and the like. good. Further, these compounds may be pulverized to a particle size of 0.1 to 20 ⁇ m. Compounds containing element B also include hydroxides, oxides, carbonates, sulfates, nitrates, etc. of element B, but in order to reduce the amount of water generated during firing, they are dried and dehydrated. It can be used after Further, these compounds may be pulverized to a particle size of 0.1 to 20 ⁇ m.
- the compound containing the complex oxide and the element A can be mixed at a ratio such that the molar ratio of the total amount of Ni and the element M to the element A is 1:0.0005 to 1:0.03. preferable.
- a suitable mixing ratio with the composite oxide is the same for the compound containing the element B.
- Elements A and B are also preferably mixed according to the stoichiometric ratio of the A x B y O z compound described in paragraph 0036.
- the firing step of the mixture in the third step includes, for example, a first firing step of firing at 450 ° C. to 680 ° C. under an oxygen stream, and a fired product obtained by the first firing step under an oxygen stream at a temperature exceeding 680 ° C. It is a multi-stage firing process including at least a second firing process of firing at a temperature.
- the temperature is raised to a first set temperature of 680° C. or lower at a first temperature elevation rate of 0.2° C./min to 5.5° C./min.
- the second firing step the temperature is raised to a second set temperature of 900° C. or less at a second temperature increase rate of 0.1° C./min to 3.5° C./min and slower than the first temperature increase rate. .
- a plurality of the first and second heating rates may be set for each predetermined temperature range within the above range.
- the retention time of the first set temperature in the first firing step is preferably 5 hours or less, more preferably 3 hours or less.
- the holding time of the first set temperature is the time for maintaining the first set temperature after reaching the first set temperature, and the holding time may be zero.
- the holding time of the second set temperature in the second firing step is preferably 1 hour to 10 hours, more preferably 1 hour to 5 hours.
- the holding time of the second set temperature is the time for maintaining the second set temperature after reaching the second set temperature. Firing of the mixture is performed, for example, in an oxygen stream with an oxygen concentration of 60% or more, and the flow rate of the oxygen stream is 0.2 mL/min to 4 mL/min per 10 cm 3 of the firing furnace and 0.3 L/min or more per 1 kg of the mixture. and
- the baked product obtained in the third step is washed with water to remove impurities, and the washed baked product is dried by heating. If necessary, the fired product is pulverized, classified, etc., and the D50 of the positive electrode active material is adjusted to the desired range. If the compound containing the element B is not added in the second step, for example, the compound containing the element B is added to and mixed with the water-washed baked product. Drying of the baked product after washing with water may be performed at a temperature of less than 100°C, but when adding a compound containing element B in the fourth step, a mixture of the compound containing element B and the baked product is , preferably at a temperature of 100° C. or higher. An example of a suitable temperature range in this case is 150°C to 250°C. Drying may be performed under vacuum or in the atmosphere. An example of the drying treatment time is 1 hour to 5 hours.
- the A x B y O z compound In order to fix the A x B y O z compound to the interface between the primary particles inside the secondary particles of the composite oxide (Z), a compound containing the element A and a compound containing the element B must be added respectively. That is, even if a compound containing both elements A and B is used, the AxByOz compound cannot exist on the surface of the primary particles inside the secondary particles.
- the A x B y O z compound is considered to be produced by melting the element A and incorporating the element B, and in the presence of the compound containing the element A and the compound containing the element B, The A x B y O z compound cannot exist on the surfaces of the primary particles inside the secondary particles unless the heat treatment is performed at a temperature of .
- the negative electrode 12 includes a negative electrode core 40 and a negative electrode mixture layer 41 provided on the surface of the negative electrode core 40 .
- a foil of a metal such as copper or a copper alloy that is stable in the potential range of the negative electrode 12, a film having the metal on the surface layer, or the like can be used.
- the negative electrode mixture layer 41 contains a negative electrode active material and a binder, and is preferably provided on both surfaces of the negative electrode core 40 .
- a negative electrode mixture slurry containing a negative electrode active material, a binder, and the like is applied to the surface of the negative electrode core 40, the coating film is dried, and then the negative electrode mixture layer 41 is compressed to form the negative electrode core. It can be made by forming on both sides of the body 40 .
- the negative electrode mixture layer 41 may contain the same conductive agent as in the case of the positive electrode 11 .
- the negative electrode mixture layer 41 contains, as a negative electrode active material, for example, a carbon material that reversibly absorbs and releases lithium ions.
- a carbon material that reversibly absorbs and releases lithium ions.
- a suitable example of the carbon material is natural graphite such as flake graphite, massive graphite, and earthy graphite, artificial graphite such as massive artificial graphite (MAG), and graphitized mesophase carbon microbeads (MCMB).
- an active material containing at least one of an element that alloys with Li, such as Si and Sn, and a compound containing the element may be used.
- a preferred example of the active material is a silicon material in which Si fine particles are dispersed in a silicon oxide phase or a silicate phase such as lithium silicate.
- a carbon material such as graphite and a silicon material are used together.
- the binder contained in the negative electrode mixture layer 41 fluororesin, PAN, polyimide, acrylic resin, polyolefin, etc. can be used as in the case of the positive electrode 11, but styrene-butadiene rubber (SBR) is used. is preferred.
- the negative electrode mixture layer 41 preferably further contains CMC or its salt, polyacrylic acid (PAA) or its salt, polyvinyl alcohol (PVA), or the like. Among them, it is preferable to use SBR together with CMC or its salt or PAA or its salt.
- the negative electrode mixture layer 41 may contain a conductive agent.
- a porous sheet having ion permeability and insulation is used for the separator 13 .
- porous sheets include microporous thin films, woven fabrics, and non-woven fabrics.
- Suitable materials for the separator 13 include polyolefins such as polyethylene, polypropylene, copolymers of ethylene and ⁇ -olefin, and cellulose.
- the separator 13 may have either a single layer structure or a laminated structure.
- a heat-resistant layer containing inorganic particles, a heat-resistant layer made of a highly heat-resistant resin such as aramid resin, polyimide, polyamideimide, or the like may be formed on the surface of the separator 13 .
- the positive electrode active material, acetylene black, and polyvinylidene fluoride are mixed at a mass ratio of 91:7:2, and N-methyl-2-pyrrolidone (NMP) is used as a dispersion medium to prepare a positive electrode mixture slurry.
- NMP N-methyl-2-pyrrolidone
- the positive electrode material mixture slurry is applied onto a positive electrode core made of aluminum foil, the coating film is dried and compressed, and then the positive electrode core is cut into a predetermined electrode size.
- a positive electrode having an agent layer formed thereon was obtained.
- an exposed portion where the surface of the positive electrode core was exposed was provided on a part of the positive electrode.
- Ethylene carbonate (EC), methyl ethyl carbonate (MEC) and dimethyl carbonate (DMC) were mixed in a volume ratio of 3:3:4 (25° C.).
- a non-aqueous electrolyte was prepared by dissolving LiPF 6 in the mixed solvent so as to have a concentration of 1.2 mol/liter.
- test cell An aluminum lead is attached to the exposed portion of the positive electrode, and a nickel lead is attached to the lithium metal foil as the negative electrode, respectively. A wound electrode body was produced. This electrode assembly was housed in an exterior made of an aluminum laminate sheet, and after the non-aqueous electrolyte was injected thereinto, the opening of the exterior was sealed to obtain a test cell Al.
- Example 2 A test cell A2 was produced in the same manner as in Example 1, except that strontium hydroxide and titanium hydroxide were added instead of calcium hydroxide, molybdenum oxide and tungsten oxide in the synthesis of the positive electrode active material.
- Example 3 In the synthesis of the positive electrode active material, a composite hydroxide represented by [Ni 0.86 Mn 0.14 ](OH) 2 is used, and a composite oxide obtained by firing the composite hydroxide, Lithium, calcium hydroxide, and tungsten oxide were added so that the molar ratio of Li, the sum of Ni and Mn, Ca, and W was 1.03:1.00:0.005:0.005. mixed into The mixture is heated from room temperature to 650° C. at a heating rate of 3.0° C./min under an oxygen stream with an oxygen concentration of 95% (flow rate of 2 mL/min per 10 cm 3 and 5 L/min per 1 kg of the mixture) and fired. After that, the temperature was raised to 780° C.
- a composite hydroxide represented by [Ni 0.86 Mn 0.14 ](OH) 2 is used, and a composite oxide obtained by firing the composite hydroxide, Lithium, calcium hydroxide, and tungsten oxide were added so that the molar ratio of Li, the sum
- a test cell A3 was produced in the same manner as in Example 1, except that the baked product was washed with water to remove impurities, and vacuum-dried at 200° C. for 2 hours to obtain a positive electrode active material.
- Example 4 A test cell A4 was produced in the same manner as in Example 3, except that zirconium oxide was added instead of tungsten oxide in the synthesis of the positive electrode active material.
- Example 5 In the synthesis of the positive electrode active material, a composite hydroxide represented by [Ni 0.91 Mn 0.09 ](OH) 2 is used, and a composite oxide obtained by baking the composite hydroxide, and a hydroxide The molar ratio of lithium, strontium hydroxide, and titanium hydroxide to Li, the total amount of Ni and Mn, Sr, and Ti is 1.05:1.00:0.025:0.025.
- a test cell A5 was prepared in the same manner as in Example 1, except that the mixture was mixed as follows.
- Example 6 A test cell A6 was prepared in the same manner as in Example 5, except that calcium hydroxide and molybdenum oxide were added instead of strontium hydroxide and titanium hydroxide in the synthesis of the positive electrode active material.
- Example 7 A test cell A7 was prepared in the same manner as in Example 5, except that calcium hydroxide was added instead of strontium hydroxide in the synthesis of the positive electrode active material.
- Example 8 A test cell A8 was produced in the same manner as in Example 5, except that calcium hydroxide and tungsten oxide were added instead of strontium hydroxide and titanium hydroxide in the synthesis of the positive electrode active material.
- Example 9 A composite hydroxide represented by [Ni 0.9 Co 0.05 Al 0.05 ](OH) 2 obtained by the coprecipitation method was fired at 500° C. for 8 hours to obtain a composite oxide.
- the composite oxide, lithium hydroxide, and calcium hydroxide were added so that the molar ratio of Li, the total amount of Ni, Co, and Al, and Ca was 1.03:1.00:0.005.
- mixed into The mixture is heated from room temperature to 650° C. at a heating rate of 3.0° C./min under an oxygen stream with an oxygen concentration of 95% (flow rate of 2 mL/min per 10 cm 3 and 5 L/min per 1 kg of the mixture) and fired. After that, the temperature was raised to 730° C.
- a test cell A9 was produced in the same manner as in Example 1, except that the positive electrode active material was used for the positive electrode.
- Example 10 In the synthesis of the positive electrode active material, a composite hydroxide represented by [Ni 0.93 Mn 0.07 ](OH) 2 is used, and a composite oxide obtained by baking the composite hydroxide, and a hydroxide Lithium, strontium hydroxide, and molybdenum oxide were added so that the molar ratio of Li, the sum of Ni and Mn, Sr, and Mo was 1.05:1.00:0.005:0.005.
- a test cell A10 was prepared in the same manner as in Example 1, except that the mixture was mixed in.
- Example 11 A test cell A11 was produced in the same manner as in Example 10, except that calcium hydroxide and zirconium oxide were added instead of strontium hydroxide and molybdenum oxide in the synthesis of the positive electrode active material.
- test cell B1 was prepared in the same manner as in Example 1, except that calcium hydroxide, molybdenum oxide, and tungsten oxide were not added in the synthesis of the positive electrode active material.
- test cell B2 was prepared in the same manner as in Example 3, except that calcium hydroxide and tungsten oxide were not added in the synthesis of the positive electrode active material.
- test cell B3 was prepared in the same manner as in Example 5, except that strontium hydroxide and titanium hydroxide were not added in the synthesis of the positive electrode active material.
- test cell B4 was prepared in the same manner as in Example 9, except that strontium hydroxide and molybdenum oxide were not added in the synthesis of the positive electrode active material.
- test cell B6 was prepared in the same manner as in Example 3, except that calcium hydroxide was not added in the synthesis of the positive electrode active material.
- Example 7 A test cell B7 was produced in the same manner as in Example 9, except that molybdenum oxide was not added in the synthesis of the positive electrode active material.
- Capacity retention rate (%) (discharge capacity at 30th cycle/discharge capacity at 1st cycle) x 100 ⁇ Cycle test>
- Test cells A1 to 4, B1, 2, 5, and 6 are charged at a constant current of 0.2 It in a temperature environment of 25° C. until the battery voltage reaches 4.4 V, and the current value is 4.4 V. Constant voltage charging was performed until the voltage reached 1/100 It.
- Test cells A5-9, B3, 4, and 7 were charged at a constant current of 0.2 It until the battery voltage reached 4.3 V, and then charged at a constant current of 4.3 V until the current reached 1/100 It.
- Test cells A1 to 4, B1, 2, 5, and 6 were performed in the same manner, except that voltage charging was performed.
- Tables 1 to 4 show the calculated capacity retention rates. Note that the capacity retention rate shown in Table 1 is a relative value when the capacity retention rate of the test cell B1 of Comparative Example 1 is set to 100.
- the capacity retention rates shown in Table 2 are relative values when the capacity retention rate of the test cell B2 of Comparative Example 2 is set to 100.
- the capacity retention rate shown in Table 3 is a relative value when the capacity retention rate of the test cell B3 of Comparative Example 3 is set to 100.
- the capacity retention rate shown in Table 4 is a relative value when the capacity retention rate of the test cell B4 of Comparative Example 4 is set to 100.
Abstract
Description
正極11は、正極芯体30と、正極芯体30の表面に設けられた正極合剤層31とを備える。正極芯体30には、アルミニウム、アルミニウム合金など正極11の電位範囲で安定な金属の箔、当該金属を表層に配置したフィルム等を用いることができる。正極合剤層31は、正極活物質、結着剤、および導電剤を含み、正極芯体30の両面に設けられることが好ましい。正極11は、例えば正極芯体30上に正極合剤のスラリーを塗布し、塗膜を乾燥させた後、圧縮して正極合剤層31を正極芯体30の両面に形成することにより作製できる。
負極12は、負極芯体40と、負極芯体40の表面に設けられた負極合剤層41とを備える。負極芯体40には、銅、銅合金などの負極12の電位範囲で安定な金属の箔、当該金属を表層に配置したフィルム等を用いることができる。負極合剤層41は、負極活物質および結着剤を含み、負極芯体40の両面に設けられることが好ましい。負極12は、例えば、負極芯体40の表面に負極活物質および結着剤等を含む負極合剤スラリーを塗布し、塗膜を乾燥させた後、圧縮して負極合剤層41を負極芯体40の両面に形成することにより作製できる。負極合剤層41には、正極11の場合と同様の導電剤が含まれていてもよい。
セパレータ13には、イオン透過性および絶縁性を有する多孔性シートが用いられる。多孔性シートの具体例としては、微多孔薄膜、織布、不織布等が挙げられる。セパレータ13の材質としては、ポリエチレン、ポリプロピレン、エチレンとαオレフィンの共重合体等のポリオレフィン、セルロースなどが好適である。セパレータ13は、単層構造、積層構造のいずれであってもよい。セパレータ13の表面には、無機粒子を含む耐熱層、アラミド樹脂、ポリイミド、ポリアミドイミド等の耐熱性の高い樹脂で構成される耐熱層などが形成されていてもよい。
[正極活物質の合成]
共沈法により得られた[Ni0.8Mn0.1Co0.1](OH)2で表される複合水酸化物を、500℃で8時間焼成して複合酸化物を得た。当該複合酸化物と、水酸化リチウムと、水酸化カルシウムと、酸化モリブデンと、酸化タングステンとを、Liと、Ni、Mn、およびCoの総量と、Caと、Moと、Wとのモル比が1.05:1.00:0.01:0.01:0.01となるように混合した。当該混合物を、酸素濃度95%の酸素気流下(10cm3あたり2mL/minおよび混合物1kgあたり5L/minの流量)、昇温速度2.0℃/minで室温から650℃まで昇温して焼成した後、昇温速度0.5℃/minで750℃まで昇温して焼成した。この焼成物を水洗して不純物を除去し、180℃で2時間真空乾燥することにより正極活物質を得た。
上記正極活物質と、アセチレンブラックと、ポリフッ化ビニリデンとを、91:7:2の質量比で混合し、分散媒としてN-メチル-2-ピロリドン(NMP)を用いて、正極合剤スラリーを調製した。次に、アルミニウム箔からなる正極芯体上に正極合剤スラリーを塗布し、塗膜を乾燥、圧縮した後、正極芯体を所定の電極サイズに切断して、正極芯体の両面に正極合剤層が形成された正極を得た。なお、正極の一部に正極芯体の表面が露出した露出部を設けた。
エチレンカーボネート(EC)と、メチルエチルカーボネート(MEC)と、ジメチルカーボネート(DMC)とを、3:3:4の体積比(25℃)で混合した。当該混合溶媒にLiPF6を1.2モル/リットルの濃度となるように溶解させて、非水電解液を調製した。
上記正極の露出部にアルミニウムリードを、負極としてリチウム金属箔にニッケルリードをそれぞれ取り付け、ポリオレフィン製のセパレータを介して正極と負極を渦巻き状に巻回した後、径方向にプレス成形して扁平状の巻回型電極体を作製した。この電極体をアルミラミネートシートで構成される外装体内に収容し、上記非水電解液を注入した後、外装体の開口部を封止して試験セルAlを得た。
正極活物質の合成において、水酸化カルシウム、酸化モリブデンおよび酸化タングステンの代わりに、水酸化ストロンチウムおよび水酸化チタンを添加したこと以外は、実施例1と同様にして試験セルA2を作製した。
正極活物質の合成において、[Ni0.86Mn0.14](OH)2で表される複合水酸化物を用い、当該複合水酸化物を焼成して得られる複合酸化物と、水酸化リチウムと、水酸化カルシウムと、酸化タングステンとを、Liと、NiおよびMnの総量と、Caと、Wとのモル比が1.03:1.00:0.005:0.005となるように混合した。当該混合物を、酸素濃度95%の酸素気流下(10cm3あたり2mL/minおよび混合物1kgあたり5L/minの流量)、昇温速度3.0℃/minで室温から650℃まで昇温して焼成した後、昇温速度1.0℃/minで780℃まで昇温して焼成した。この焼成物を水洗して不純物を除去し、200℃で2時間、真空乾燥することにより正極活物質を得たこと以外は、実施例1と同様にして試験セルA3を作製した。
正極活物質の合成において、酸化タングステンの代わりに、酸化ジルコニウムを添加したこと以外は、実施例3と同様にして試験セルA4を作製した。
正極活物質の合成において、[Ni0.91Mn0.09](OH)2で表される複合水酸化物を用い、当該複合水酸化物を焼成して得られる複合酸化物と、水酸化リチウムと、水酸化ストロンチウムと、水酸化チタンとを、Liと、NiおよびMnの総量と、Srと、Tiとのモル比が1.05:1.00:0.025:0.025となるように混合したこと以外は、実施例1と同様にして試験セルA5を作製した。
正極活物質の合成において、水酸化ストロンチウムおよび水酸化チタンの代わりに、水酸化カルシウムおよび酸化モリブデンを添加したこと以外は、実施例5と同様にして試験セルA6を作製した。
正極活物質の合成において、水酸化ストロンチウムの代わりに、水酸化カルシウムを添加したこと以外は、実施例5と同様にして試験セルA7を作製した。
正極活物質の合成において、水酸化ストロンチウムおよび水酸化チタンの代わりに、水酸化カルシウムおよび酸化タングステンを添加したこと以外は、実施例5と同様にして試験セルA8を作製した。
共沈法により得られた[Ni0.9Co0.05Al0.05](OH)2で表される複合水酸化物を、500℃で8時間焼成して複合酸化物を得た。当該複合酸化物と、水酸化リチウムと、水酸化カルシウムとを、Liと、Ni、Co、およびAlの総量と、Caとのモル比が1.03:1.00:0.005となるように混合した。当該混合物を、酸素濃度95%の酸素気流下(10cm3あたり2mL/minおよび混合物1kgあたり5L/minの流量)、昇温速度3.0℃/minで室温から650℃まで昇温して焼成した後、昇温速度0.5℃/minで730℃まで昇温して焼成した。この焼成物を水洗して不純物を除去し、所定量の酸化タングステンを添加してから、180℃で2時間真空乾燥することにより正極活物質を得た。正極に当該正極活物質を用いたこと以外は、実施例1と同様にして試験セルA9を作製した。
正極活物質の合成において、[Ni0.93Mn0.07](OH)2で表される複合水酸化物を用い、当該複合水酸化物を焼成して得られる複合酸化物と、水酸化リチウムと、水酸化ストロンチウムと、酸化モリブデンとを、Liと、NiおよびMnの総量と、Srと、Moとのモル比が1.05:1.00:0.005:0.005となるように混合したこと以外は、実施例1と同様にして試験セルA10を作製した。
正極活物質の合成において、水酸化ストロンチウムおよび酸化モリブデンの代わりに、水酸化カルシウムおよび酸化ジルコニウムを添加したこと以外は、実施例10と同様にして試験セルA11を作製した。
正極活物質の合成において、水酸化カルシウム、酸化モリブデン、および酸化タングステンを添加しなかったこと以外は、実施例1と同様にして試験セルB1を作製した。
正極活物質の合成において、水酸化カルシウムおよび酸化タングステンを添加しなかったこと以外は、実施例3と同様にして試験セルB2を作製した。
正極活物質の合成において、水酸化ストロンチウムおよび水酸化チタンを添加しなかったこと以外は、実施例5と同様にして試験セルB3を作製した。
正極活物質の合成において、水酸化ストロンチウムおよび酸化モリブデンを添加しなかったこと以外は、実施例9と同様にして試験セルB4を作製した。
正極活物質の合成において、酸化タングステンの添加のタイミングを焼成物の水洗後に変更し、水洗後の乾燥を大気圧下、80℃の条件で行ったこと以外は、実施例3と同様にして試験セルB5を作製した。比較例5の正極活物質は、CaWO4が二次粒子の表面のみに存在し、二次粒子の内部における一次粒子同士の界面にCaWO4は確認されなかった。
正極活物質の合成において、水酸化カルシウムを添加しなかったこと以外は、実施例3と同様にして試験セルB6を作製した。
正極活物質の合成において、酸化モリブデンを添加しなかったこと以外は、実施例9と同様にして試験セルB7を作製した。
実施例および比較例の各試験セルについて、サイクル試験を行なった。サイクル試験の1サイクル目の放電容量と、30サイクル目の放電容量を求め、下記式により容量維持率を算出した。
容量維持率(%)=(30サイクル目放電容量÷1サイクル目放電容量)×100
<サイクル試験>
試験セルA1~4、B1,2,5,6を、25℃の温度環境下、0.2Itの定電流で電池電圧が4.4Vになるまで定電流充電を行い、4.4Vで電流値が1/100Itになるまで定電圧充電を行った。その後、0.2Itの定電流で電池電圧が2.5Vになるまで定電流放電を行った。この充放電サイクルを30サイクル繰り返した。試験セルA5~9、B3,4,7については、0.2Itの定電流で電池電圧が4.3Vになるまで定電流充電を行い、4.3Vで電流値が1/100Itになるまで定電圧充電を行った以外は、試験セルA1~4、B1,2,5,6と同様にして行った。
Claims (5)
- 層状構造を有し、Li、Oを除く元素の総モル量に対して75モル%以上のNiを含有するリチウム遷移金属複合酸化物を含み、
前記リチウム遷移金属複合酸化物は、一次粒子が凝集してなる二次粒子であり、少なくとも前記二次粒子の内部における前記一次粒子同士の界面には、一般式AxByOz(式中、1≦x≦2、1≦y≦5、4≦z≦9、AはCaおよびSrから選択される少なくとも1種、BはW、Mo、Ti、Si、Nb、およびZrから選択される少なくとも1種)で表される化合物が固着している、非水電解質二次電池用正極活物質。 - 前記化合物中の元素Aの含有量は、前記リチウム遷移金属複合酸化物および前記化合物中のLi、Oを除く元素の総モル量に対して3モル%以下である、請求項1に記載の非水電解質二次電池用正極活物質。
- 前記リチウム遷移金属複合酸化物は、Mn、Al、Co、Fe、Ti、Si、Nb、Mo、W、およびZrから選択される少なくとも1種である元素Mを含有し、
元素Mの含有量は、Li、Oを除く元素の総モル量に対して5~25モル%である、請求項1または2に記載の非水電解質二次電池用正極活物質。 - 前記リチウム遷移金属複合酸化物は、BET比表面積が0.5~3.5m2/gである、請求項1~3のいずれか一項に記載の非水電解質二次電池用正極活物質。
- 請求項1~4のいずれか一項に記載の正極活物質を含む正極と、負極と、非水電解質とを備える、非水電解質二次電池。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202280023447.3A CN117043990A (zh) | 2021-04-01 | 2022-03-16 | 非水电解质二次电池用正极活性物质和非水电解质二次电池 |
| JP2023510910A JPWO2022209894A1 (ja) | 2021-04-01 | 2022-03-16 | |
| EP22780114.9A EP4318664A1 (en) | 2021-04-01 | 2022-03-16 | Positive electrode active material for non-aqueous electrolyte secondary batteries, and non-aqueous electrolyte secondary battery |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021-062506 | 2021-04-01 | ||
| JP2021062506 | 2021-04-01 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022209894A1 true WO2022209894A1 (ja) | 2022-10-06 |
Family
ID=83459015
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/011852 WO2022209894A1 (ja) | 2021-04-01 | 2022-03-16 | 非水電解質二次電池用正極活物質および非水電解質二次電池 |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP4318664A1 (ja) |
| JP (1) | JPWO2022209894A1 (ja) |
| CN (1) | CN117043990A (ja) |
| WO (1) | WO2022209894A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024070385A1 (ja) * | 2022-09-30 | 2024-04-04 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006351378A (ja) * | 2005-06-16 | 2006-12-28 | Matsushita Electric Ind Co Ltd | リチウムイオン二次電池 |
| WO2012035664A1 (ja) | 2010-09-17 | 2012-03-22 | トヨタ自動車株式会社 | リチウムイオン二次電池 |
| JP2012252807A (ja) * | 2011-05-31 | 2012-12-20 | Toyota Motor Corp | リチウム二次電池 |
| JP2017188428A (ja) * | 2016-03-30 | 2017-10-12 | Basf戸田バッテリーマテリアルズ合同会社 | 非水電解質二次電池用の正極活物質及びその製造方法、並びにそれを用いた非水電解質二次電池 |
| JP2018129221A (ja) | 2017-02-09 | 2018-08-16 | 株式会社Gsユアサ | 非水電解質二次電池用正極活物質、その製造方法、非水電解質二次電池用正極、及び非水電解質二次電池 |
| JP2018185884A (ja) * | 2015-09-29 | 2018-11-22 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池 |
| CN110931738A (zh) * | 2019-11-20 | 2020-03-27 | 广东邦普循环科技有限公司 | 一种复相高压正极材料及其制备方法 |
| JP2020515010A (ja) * | 2017-11-22 | 2020-05-21 | エルジー・ケム・リミテッド | リチウム二次電池用正極活物質およびその製造方法 |
| WO2021241075A1 (ja) * | 2020-05-29 | 2021-12-02 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池用正極活物質、及び非水電解質二次電池 |
-
2022
- 2022-03-16 EP EP22780114.9A patent/EP4318664A1/en active Pending
- 2022-03-16 JP JP2023510910A patent/JPWO2022209894A1/ja active Pending
- 2022-03-16 CN CN202280023447.3A patent/CN117043990A/zh active Pending
- 2022-03-16 WO PCT/JP2022/011852 patent/WO2022209894A1/ja active Application Filing
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006351378A (ja) * | 2005-06-16 | 2006-12-28 | Matsushita Electric Ind Co Ltd | リチウムイオン二次電池 |
| WO2012035664A1 (ja) | 2010-09-17 | 2012-03-22 | トヨタ自動車株式会社 | リチウムイオン二次電池 |
| JP2012252807A (ja) * | 2011-05-31 | 2012-12-20 | Toyota Motor Corp | リチウム二次電池 |
| JP2018185884A (ja) * | 2015-09-29 | 2018-11-22 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池 |
| JP2017188428A (ja) * | 2016-03-30 | 2017-10-12 | Basf戸田バッテリーマテリアルズ合同会社 | 非水電解質二次電池用の正極活物質及びその製造方法、並びにそれを用いた非水電解質二次電池 |
| JP2018129221A (ja) | 2017-02-09 | 2018-08-16 | 株式会社Gsユアサ | 非水電解質二次電池用正極活物質、その製造方法、非水電解質二次電池用正極、及び非水電解質二次電池 |
| JP2020515010A (ja) * | 2017-11-22 | 2020-05-21 | エルジー・ケム・リミテッド | リチウム二次電池用正極活物質およびその製造方法 |
| CN110931738A (zh) * | 2019-11-20 | 2020-03-27 | 广东邦普循环科技有限公司 | 一种复相高压正极材料及其制备方法 |
| WO2021241075A1 (ja) * | 2020-05-29 | 2021-12-02 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池用正極活物質、及び非水電解質二次電池 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024070385A1 (ja) * | 2022-09-30 | 2024-04-04 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4318664A1 (en) | 2024-02-07 |
| JPWO2022209894A1 (ja) | 2022-10-06 |
| CN117043990A (zh) | 2023-11-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2022092182A1 (ja) | 非水電解質二次電池 | |
| CN113597409A (zh) | 二次电池用的正极活性物质和二次电池 | |
| WO2023054041A1 (ja) | 非水電解質二次電池用正極活物質および非水電解質二次電池 | |
| WO2021039751A1 (ja) | 非水電解質二次電池 | |
| JP7324120B2 (ja) | 非水電解質二次電池用正極活物質、及び非水電解質二次電池 | |
| WO2021059728A1 (ja) | 非水電解質二次電池用正極活物質、及び非水電解質二次電池 | |
| WO2022209894A1 (ja) | 非水電解質二次電池用正極活物質および非水電解質二次電池 | |
| JP7324119B2 (ja) | 非水電解質二次電池用正極活物質、及び非水電解質二次電池 | |
| WO2021241027A1 (ja) | 非水電解質二次電池用正極活物質および非水電解質二次電池 | |
| WO2021220875A1 (ja) | 非水電解質二次電池用正極活物質、及び非水電解質二次電池 | |
| WO2021049198A1 (ja) | 非水電解質二次電池 | |
| WO2021024789A1 (ja) | 非水電解質二次電池用正極活物質、及び非水電解質二次電池 | |
| WO2021019943A1 (ja) | 非水電解質二次電池 | |
| WO2024004578A1 (ja) | 非水電解質二次電池 | |
| WO2023189507A1 (ja) | 非水電解質二次電池用正極活物質および非水電解質二次電池 | |
| WO2022070648A1 (ja) | 非水電解質二次電池 | |
| WO2024062866A1 (ja) | 二次電池用正極活物質および二次電池 | |
| WO2023204077A1 (ja) | 非水電解質二次電池用正極活物質および非水電解質二次電池 | |
| WO2024062848A1 (ja) | 二次電池用正極活物質および二次電池 | |
| WO2021039750A1 (ja) | 非水電解質二次電池用正極活物質、及び非水電解質二次電池 | |
| WO2022181264A1 (ja) | 非水電解質二次電池用正極活物質および非水電解質二次電池 | |
| WO2024024364A1 (ja) | 非水電解質二次電池用正極活物質および非水電解質二次電池 | |
| WO2022114231A1 (ja) | 非水電解質二次電池 | |
| WO2023127290A1 (ja) | 非水電解質二次電池用正極活物質 | |
| WO2024057933A1 (ja) | 非水電解質二次電池 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22780114 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023510910 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18282606 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202280023447.3 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022780114 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2022780114 Country of ref document: EP Effective date: 20231102 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |