WO2017154901A1 - フォトクロミック特性を有する合成樹脂積層体およびそれを用いたレンズ - Google Patents
フォトクロミック特性を有する合成樹脂積層体およびそれを用いたレンズ Download PDFInfo
- Publication number
- WO2017154901A1 WO2017154901A1 PCT/JP2017/009000 JP2017009000W WO2017154901A1 WO 2017154901 A1 WO2017154901 A1 WO 2017154901A1 JP 2017009000 W JP2017009000 W JP 2017009000W WO 2017154901 A1 WO2017154901 A1 WO 2017154901A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- synthetic resin
- layer
- layers
- transparent
- resin laminate
- Prior art date
Links
Images
Classifications
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B5/00—Optical elements other than lenses
- G02B5/20—Filters
- G02B5/22—Absorbing filters
- G02B5/223—Absorbing filters containing organic substances, e.g. dyes, inks or pigments
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29D—PRODUCING PARTICULAR ARTICLES FROM PLASTICS OR FROM SUBSTANCES IN A PLASTIC STATE
- B29D11/00—Producing optical elements, e.g. lenses or prisms
- B29D11/0073—Optical laminates
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B23/00—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose
- B32B23/04—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose comprising such cellulosic plastic substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B23/08—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose comprising such cellulosic plastic substance as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29D—PRODUCING PARTICULAR ARTICLES FROM PLASTICS OR FROM SUBSTANCES IN A PLASTIC STATE
- B29D11/00—Producing optical elements, e.g. lenses or prisms
- B29D11/00009—Production of simple or compound lenses
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B23/00—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose
- B32B23/20—Layered products comprising a layer of cellulosic plastic substances, i.e. substances obtained by chemical modification of cellulose, e.g. cellulose ethers, cellulose esters, viscose comprising esters
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/06—Layered products comprising a layer of synthetic resin as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B27/08—Layered products comprising a layer of synthetic resin as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/18—Layered products comprising a layer of synthetic resin characterised by the use of special additives
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/18—Layered products comprising a layer of synthetic resin characterised by the use of special additives
- B32B27/20—Layered products comprising a layer of synthetic resin characterised by the use of special additives using fillers, pigments, thixotroping agents
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/30—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers
- B32B27/306—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers comprising vinyl acetate or vinyl alcohol (co)polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/30—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers
- B32B27/308—Layered products comprising a layer of synthetic resin comprising vinyl (co)polymers; comprising acrylic (co)polymers comprising acrylic (co)polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/34—Layered products comprising a layer of synthetic resin comprising polyamides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/36—Layered products comprising a layer of synthetic resin comprising polyesters
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/36—Layered products comprising a layer of synthetic resin comprising polyesters
- B32B27/365—Layered products comprising a layer of synthetic resin comprising polyesters comprising polycarbonates
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/40—Layered products comprising a layer of synthetic resin comprising polyurethanes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B38/00—Ancillary operations in connection with laminating processes
- B32B38/0012—Mechanical treatment, e.g. roughening, deforming, stretching
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B7/00—Layered products characterised by the relation between layers; Layered products characterised by the relative orientation of features between layers, or by the relative values of a measurable parameter between layers, i.e. products comprising layers having different physical, chemical or physicochemical properties; Layered products characterised by the interconnection of layers
- B32B7/04—Interconnection of layers
- B32B7/12—Interconnection of layers using interposed adhesives or interposed materials with bonding properties
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B3/00—Simple or compound lenses
- G02B3/02—Simple or compound lenses with non-spherical faces
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B5/00—Optical elements other than lenses
- G02B5/20—Filters
- G02B5/22—Absorbing filters
- G02B5/23—Photochromic filters
-
- G—PHYSICS
- G02—OPTICS
- G02C—SPECTACLES; SUNGLASSES OR GOGGLES INSOFAR AS THEY HAVE THE SAME FEATURES AS SPECTACLES; CONTACT LENSES
- G02C7/00—Optical parts
- G02C7/10—Filters, e.g. for facilitating adaptation of the eyes to the dark; Sunglasses
-
- G—PHYSICS
- G02—OPTICS
- G02C—SPECTACLES; SUNGLASSES OR GOGGLES INSOFAR AS THEY HAVE THE SAME FEATURES AS SPECTACLES; CONTACT LENSES
- G02C7/00—Optical parts
- G02C7/10—Filters, e.g. for facilitating adaptation of the eyes to the dark; Sunglasses
- G02C7/102—Photochromic filters
-
- G—PHYSICS
- G02—OPTICS
- G02C—SPECTACLES; SUNGLASSES OR GOGGLES INSOFAR AS THEY HAVE THE SAME FEATURES AS SPECTACLES; CONTACT LENSES
- G02C7/00—Optical parts
- G02C7/12—Polarisers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2250/00—Layers arrangement
- B32B2250/24—All layers being polymeric
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/10—Coating on the layer surface on synthetic resin layer or on natural or synthetic rubber layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/26—Polymeric coating
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/40—Properties of the layers or laminate having particular optical properties
- B32B2307/402—Coloured
- B32B2307/4026—Coloured within the layer by addition of a colorant, e.g. pigments, dyes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/40—Properties of the layers or laminate having particular optical properties
- B32B2307/412—Transparent
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/40—Properties of the layers or laminate having particular optical properties
- B32B2307/42—Polarizing, birefringent, filtering
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/732—Dimensional properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2551/00—Optical elements
-
- G—PHYSICS
- G02—OPTICS
- G02C—SPECTACLES; SUNGLASSES OR GOGGLES INSOFAR AS THEY HAVE THE SAME FEATURES AS SPECTACLES; CONTACT LENSES
- G02C2202/00—Generic optical aspects applicable to one or more of the subgroups of G02C7/00
- G02C2202/16—Laminated or compound lenses
Definitions
- the present invention relates to a synthetic resin laminate having a photochromic property that is suitably used for sunglasses, goggles, correction lenses, and the like, and a lens using the same.
- a photochromic organic compound is added to a silicone-based surface-curing paint, applied to one side of the laminate, and cured, or a photochromic dye is added to a urethane-based paint.
- a photochromic dye is added to a urethane-based paint.
- the method of applying a paint containing a photochromic dye on one side of the synthetic resin laminate increases the number of steps of painting on the individual laminates, causes cracks due to the solvent in the coating components, and results in poor appearance.
- Patent Document 2 discloses a method of forming a synthetic resin laminate having a photochromic property in which two transparent resins are bonded with a polyurethane resin to which a photochromic dye is added.
- Patent Document 3 discloses a method of forming a lens by applying or transferring a resin layer having a photochromic property onto a transparent plastic sheet and injection molding a backup resin on the resin layer.
- the thickness of the polyurethane resin layer having photochromic properties is increased in order to obtain sufficient contrast, and control is performed so that the thickness is smooth and uniform. It was difficult. Thickness non-uniformity is undesired in practice because it manifests itself as shading unevenness during color development. Further, there has been a problem that delamination is likely to occur, and the synthetic resin laminate is warped due to cure shrinkage of the polyurethane resin.
- a transparent synthetic resin laminate having a photochromic property excellent in controlling the surface smoothness and the thickness of the coating film of a layer having a good color development and a photochromic property has not been obtained. is the current situation.
- the present invention is intended to solve the problems associated with the prior art as described above, and the object of the present invention is to provide a layer having photochromic characteristics with good color contrast and no in-plane color unevenness.
- An object of the present invention is to provide a transparent synthetic resin laminate having photochromic properties excellent in the smoothness of the coating film surface and the control of the coating film thickness.
- the present invention is as follows. ⁇ 1> Two transparent synthetic resin layers (A), two or more functional layers (B) having photochromic properties interposed between the two synthetic resin layers (A), and the two or more A transparent resin layer (C) interposed between the functional layers (B), wherein the functional layer (B) contains a photochromic dye and is a cured layer obtained from diisocyanate and polyol. And a synthetic resin laminate. ⁇ 2> Polarization characteristics in order from the functional layer (B) side between one of the functional layer (B) and one of the synthetic resin layers (A) on the opposite side of the resin layer (C). It is a synthetic resin laminated body as described in said ⁇ 1> which has the resin layer (D) which has, and an adhesive material layer (E).
- the total thickness of all functional layers (B) interposed between the two transparent synthetic resin layers (A) is 30 ⁇ m or more, and the thickness of each functional layer (B) is 25 ⁇ m.
- At least one of the transparent synthetic resin layers (A) is selected from the group consisting of aromatic polycarbonate, poly (meth) acrylate, acetylcellulose, polyamide, and a mixture of aromatic polycarbonate and alicyclic polyester.
- the transparent resin layer (C) interposed between the functional layers (B) is aromatic polycarbonate, poly (meth) acrylate, acetylcellulose, polyamide, polyester, aromatic polycarbonate, and alicyclic polyester.
- the synthetic resin laminate according to any one of ⁇ 1> to ⁇ 6> including one or more selected from the group consisting of: ⁇ 8>
- ⁇ 9> A lens obtained by bending the synthetic resin laminate according to any one of ⁇ 1> to ⁇ 8> into a spherical surface or an aspherical surface.
- ⁇ 10> A lens having the synthetic resin laminate according to any one of ⁇ 1> to ⁇ 8> bent and having a transparent resin integral injection-molded body on the concave surface side.
- a transparent synthetic resin laminate having a photochromic property excellent in control of the thickness of a coating film of a layer having a good color contrast and in-plane color unevenness and having a photochromic property, and the synthetic resin laminate A photochromic lens using a body can be provided.
- FIG. 1 A schematic cross-sectional view showing one embodiment of the synthetic resin laminate of the present invention is shown in FIG.
- the synthetic resin laminate shown in FIG. 1 has two transparent synthetic resin layers (A) and two functional layers (B) having photochromic properties interposed between the two synthetic resin layers (A). And a transparent resin layer (C) interposed between the two functional layers (B).
- FIG. 2 the schematic sectional drawing which shows another one Embodiment of the synthetic resin laminated body of this invention is shown in FIG.
- the synthetic resin laminate shown in FIG. 2 is different from the synthetic resin laminate shown in FIG. 1 on one side of two functional layers (B) having photochromic properties and on the side opposite to the transparent resin layer (C).
- a resin layer (D) having polarization characteristics and an adhesive layer (E) are sequentially provided from the functional layer (B) side.
- the synthetic resin laminate of the present invention is not limited to the embodiment shown in FIGS.
- the resin used as the transparent synthetic resin layer (A) in the present invention examples include, for example, aromatic polycarbonate, poly (meth) acrylate, acetylcellulose, polyamide, and a mixture of aromatic polycarbonate and alicyclic polyester. More can be selected.
- two transparent synthetic resin layers (A) are used, but each may contain the same resin component or different resin components. From the viewpoint of warping of the synthetic resin laminate and bending workability, the two transparent synthetic resin layers (A) preferably contain the same resin component.
- the transparent synthetic resin layer (A) in the present invention is selected from the group consisting of aromatic polycarbonate, poly (meth) acrylate, acetylcellulose, polyamide, and a mixture of aromatic polycarbonate and alicyclic polyester.
- both of the transparent synthetic resin layers (A) in the present invention are more preferably a single layer containing an aromatic polycarbonate.
- a thermoplastic polycarbonate polymer which may be branched by reacting an aromatic dihydroxy compound or a small amount thereof with a diester of phosgene or carbonic acid is more preferable.
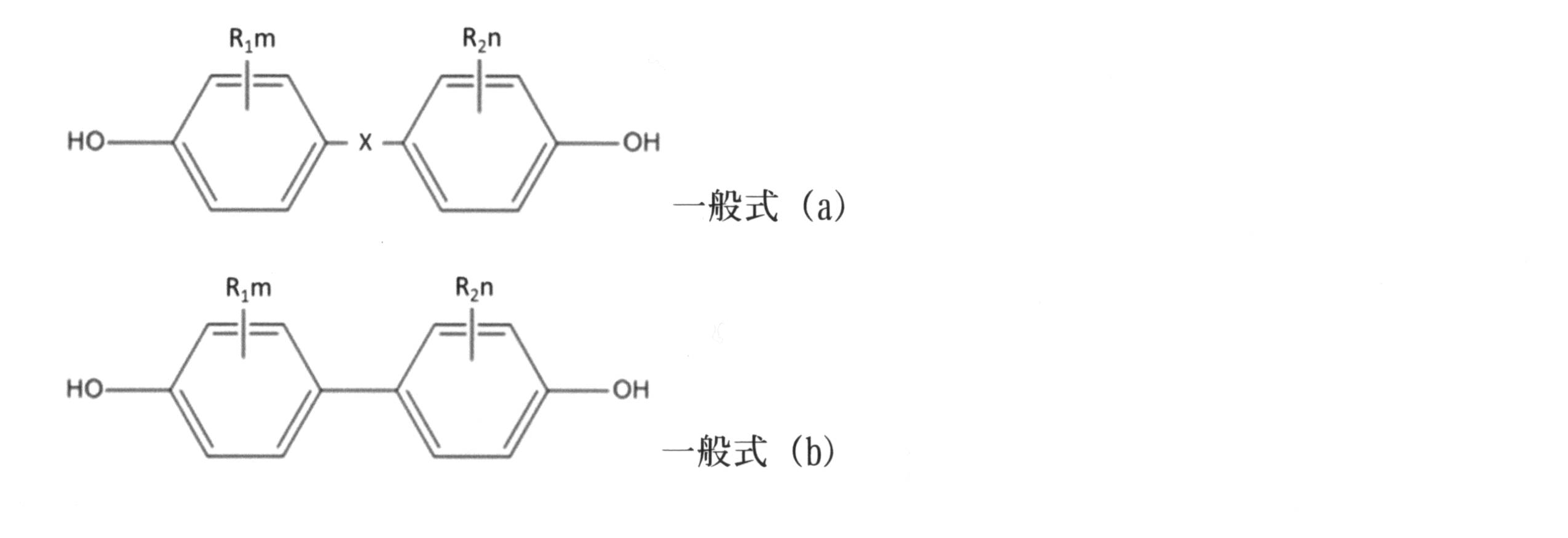
- the aromatic dihydroxy compound is a compound represented by the following general formula (a) or general formula (b).
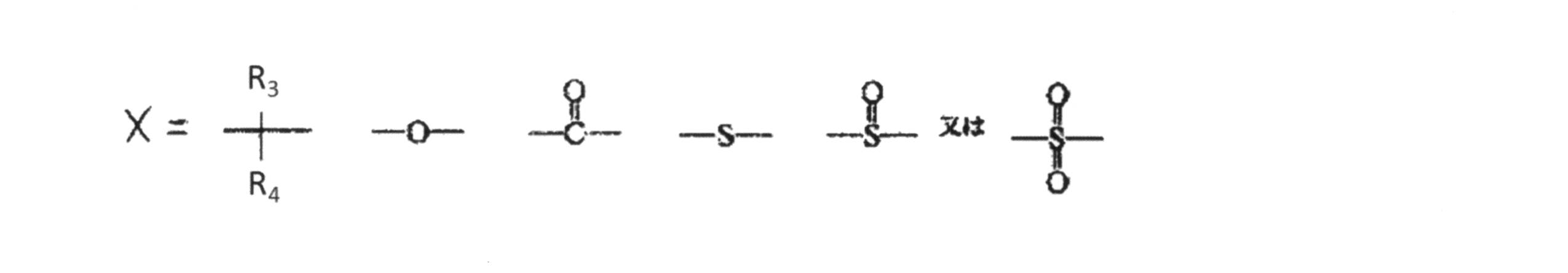
- X is as follows.
- R 1 and R 2 are a hydrogen atom, an alkyl group having 1 to 10 carbon atoms, or halogen, and R 1 and R 2 may be the same or different.
- M and n represent the number of substituents and are integers of 0 to 4.
- R 3 and R 4 are each independently a hydrogen atom, an alkyl group having 1 to 10 carbon atoms, or a phenyl group, and R 3 and R 4 may be bonded to form a ring.
- aromatic dihydroxy compound represented by the general formula (a) or the general formula (b) examples include bis (4-hydroxyphenyl) methane, 1,1-bis (4-hydroxyphenyl) ethane, 2,2 -Bis (4-hydroxyphenyl) propane, 2,2-bis (4-hydroxyphenyl) butane, 2,2-bis (4-hydroxyphenyl) octane, bis (4-hydroxyphenyl) phenylmethane, 2,2- Bis (4-hydroxy-3-methylphenyl) propane, 1,1-bis (4-hydroxy-3-tert-butylphenyl) propane, 2,2-bis (4-hydroxy-3-bromophenyl) propane, , 1-bis (4-hydroxyphenyl) cyclopentane, 1,1-bis (4-hydroxyphenyl) cyclohexane, 4,4′- Hydroxydiphenyl ether, 4,4′-dihydroxy-3,3′-dimethylphenyl ether, 4,4′-dihydroxyphenyl sulfide, 4,4
- 2,2'-bis (4-hydroxyphenyl) propane (bisphenol A: BPA) is particularly preferable from the viewpoint of strength, heat resistance, durability, and bending workability.
- the aromatic dihydroxy compound represented by the general formula (a) can be used in combination of two or more.
- the transparent synthetic resin layer (A) in the present invention can be produced by injection molding, compression molding, extrusion molding, cast molding or the like.
- the thickness of the transparent synthetic resin layer (A) in the present invention is preferably 50 ⁇ m to 1.5 mm, more preferably 100 ⁇ m to 800 ⁇ m.
- the resin layer (A) may be a single layer of the above-described resin, or may be used as a multilayer sheet or film in which two or more different resins are laminated and integrated.
- the laminating method is not particularly limited, but a coextrusion molding method is desirable because it can be laminated with good adhesion. From the viewpoint of heat resistance and impact resistance, it is also preferable to use a multilayer sheet or film obtained by laminating and integrating a layer made of an aromatic polycarbonate resin and a layer made of poly (meth) acrylate from the viewpoint of scratch resistance.
- the resin for the functional layer (B) is such that the layer made of poly (meth) acrylate is located on the surface side of the synthetic resin laminate and the layer made of aromatic polycarbonate resin is located on the functional layer (B) side. It is preferable to arrange the layer (A).
- the synthetic resin laminate of the present invention has two or more functional layers (B) having photochromic properties interposed between the two synthetic resin layers (A), and the functional layer (B) is a photochromic dye. And a cured layer in which diisocyanate and polyol are mixed.
- a functional layer (B) can be formed, for example, by curing a mixture of a polyurethane prepolymer, a curing agent, and a photochromic dye (such as a light stabilizer or an antioxidant if necessary).
- a photochromic dye such as a light stabilizer or an antioxidant if necessary.
- One feature of the present invention is that it has two or more functional layers (B) having photochromic characteristics.
- the number of functional layers (B) having photochromic properties in the present invention is preferably 2 or 3, and more preferably 2.
- the functional layer (B) having photochromic properties in the present invention preferably contains a urethane-based resin containing a photochromic dye.
- the thickness of each functional layer (B) is preferably 5 ⁇ m to 25 ⁇ m, more preferably 10 ⁇ m to 20 ⁇ m from the viewpoint of adhesion, strength, and thickness accuracy.
- the total thickness of all functional layers (B) interposed between the two transparent synthetic resin layers (A) is preferably 30 ⁇ m or more, and more preferably 35 ⁇ m to 50 ⁇ m.
- the urethane resin it is preferable to use a polyurethane prepolymer obtained by reacting a diisocyanate and a polyol at a certain ratio.
- diisocyanate compound diphenylmethane-4,4′-diisocyanate (MDI) is preferred.
- polyol it is preferable to use polypropylene glycol (PPG) having a polymerization degree of 5 to 30.
- the number average molecular weight of the polyurethane prepolymer is preferably 500 to 5000, more preferably 1500 to 4000, and particularly preferably 2000 to 3000.
- the curing agent is not particularly limited as long as it is a compound having two or more hydroxyl groups, and examples thereof include polyurethane polyol, polyether polyol, polyester polyol, acrylic polyol, polybutadiene polyol, and polycarbonate polyol.
- a polyurethane polyol having a hydroxyl group at a terminal obtained from a specific isocyanate and a specific polyol is preferable.
- a polyurethane polyol having hydroxyl groups at least at both terminal groups derived from diisocyanate and polyol is preferable, and tolylene diisocyanate (TDI) is preferably used as the diisocyanate.
- TDI tolylene diisocyanate
- polypropylene glycol PPG
- the number average molecular weight of the curing agent is preferably 500 to 5000, more preferably 1500 to 4000, and particularly preferably 2000 to 3000.
- solvents such as ethyl acetate, tetrahydrofuran and toluene may be added for viscosity adjustment.
- the photochromic dye used in the present invention is not particularly limited as long as it is compatible with the urethane resin, but spiropyran compounds, spirooxazine compounds and naphthopyran compounds are preferable.
- Various additives such as UV absorbers, antioxidants and antifoaming agents can be added to the functional layer (B).
- the stabilizer include hindered amine light stabilizers and antioxidants such as hindered phenols.
- hindered amine light stabilizer examples include bis (2,2,6,6-tetramethyl-4-piperidyl) sebacate and bis (1,2,2,6,6-pentamethyl-4-piperidyl) sebacate.
- [2,2′-thiobis (4-tert-octylphenolate)]-2-ethylhexylamine nickel, nickel complex-3,5-di-t-butyl-4-hydroxybenzyl as a nickel-based UV stabilizer -Phosphoric acid monoethylate, nickel dibutyl-dithiocarbamate, etc. can also be used.
- the hindered amine light stabilizer a hindered amine light stabilizer containing only a tertiary amine is preferable.
- bis (1,2,2,6,6-pentamethyl-4-piperidyl) is preferable.
- Sebacate bis (1,2,2,6,6-pentamethyl-4-piperidyl) -2- (3,5-di-tert-butyl-4-hydroxybenzyl) -2-n-butyl malonate, or A condensate of 1,2,2,6,6-pentamethyl-4-piperidinol / tridecyl alcohol and 1,2,3,4-butanetetracarboxylic acid is preferred.
- Antioxidants such as various hindered phenols can be used as the antioxidant, and among them, phenolic antioxidants, thiol antioxidants, and phosphite antioxidants can be used. preferable.
- phenol-based antioxidant examples include 1,1,3-tris (2-methyl-4-hydroxy-5-tert-butylphenyl) butane, 2,2′-methylenebis (4-ethyl-6-tert).
- -Butylphenol tetrakis- [methylene-3- (3 ′, 5′-di-tert-butyl-4′-hydroxyphenyl) propionate] methane, 2,6-di-tert-butyl-p-cresol, 4, 4'-thiobis (3-methyl-6-tert-butylphenol), 4,4'-butylidenebis (3-methyl-6-tert-butylphenol), 1,3,5-tris (3 ', 5'-di-) tert-butyl-4′-hydroxybenzyl) -S-triazine-2,4,6- (1H, 3H, 5H) trione, stearyl- ⁇ - (3,5- -Tert-butyl-4-hydroxyphenyl) propionate,
- the synthetic resin laminate of the present invention has a transparent resin layer (C) interposed between the two or more functional layers (B) having photochromic properties.
- this transparent resin layer (C) may be 2 or more.
- the resin used for the transparent resin layer (C) in the present invention include aromatic polycarbonate, poly (meth) acrylate, acetylcellulose, polyamide, polyester, and a mixture of aromatic polycarbonate and alicyclic polyester. You can select from a group.
- acetylcellulose is preferable from the viewpoint of transparency, low birefringence, and solvent resistance.
- acetyl cellulose examples include triacetyl cellulose (TAC) and diacetyl cellulose, and triacetyl cellulose (TAC) is particularly preferable.
- the thickness of the transparent resin layer (C) in the present invention is preferably 10 ⁇ m to 300 ⁇ m, more preferably 30 ⁇ m to 100 ⁇ m.
- Various known additives can be added to the transparent resin layer (C) in the present invention according to the purpose within a range not impairing its physical properties.
- heat stabilizer, hydrolysis stabilizer, antioxidant, pigment, dye, reinforcing agent, filler, ultraviolet absorber, lubricant, mold release agent, crystal nucleating agent, plasticizer, fluidity improver, antistatic An agent, an antibacterial agent and the like can be added.
- the transparent resin layer (C) in the present invention may have polarization characteristics.
- polarized light is sequentially applied from one side of the functional layer (B) between one side of the functional layer (B) and one side of the synthetic resin layer (A) existing on the side opposite to the resin layer (C). It is preferable to have a resin layer (D) having properties and an adhesive layer (E). For the resin layer (D) having the polarizing property, it is preferable to use a layer produced by dyeing and stretching a polyvinyl alcohol resin film with a dichroic organic dye.
- polyvinyl alcohols examples include polyvinyl alcohol (PVA), a PVA acetate ester structure that remains in a trace amount, and PVA derivatives or analogs such as polyvinyl formal, polyvinyl acetal, saponified ethylene-vinyl acetate copolymer, and the like.
- PVA is preferred. While this polyvinyl alcohol resin film is stretched in one direction, a dichroic organic dye is impregnated or adsorbed, appropriately fixed, and dried to obtain a polarizing film.
- the weight average molecular weight of PVA is preferably 50,000 to 350,000, more preferably 150,000 to 300,000.
- the thickness of the PVA film before stretching is usually about 100 to 300 ⁇ m, and the PVA polarizing film after stretching is preferably about 10 to 50 ⁇ m.
- the stretching ratio of the PVA film is preferably 2 to 8 times, and is appropriately selected depending on the purpose, but 3 to 6 times is more preferable from the viewpoint of strength after stretching.
- the adhesive layer (E) may be any adhesive as long as it is used for laminating a normal PC film and a polarizing film, but is used for the functional layer (B) having the photochromic characteristics.
- a polyurethane resin is preferably used as an adhesive.
- the thickness of the adhesive layer (E) is preferably in the range of 5 to 100 ⁇ m, more preferably 5 to 50 ⁇ m. When the thickness is less than 5 ⁇ m, it may be difficult to obtain a sufficient adhesive force.
- the adhesive force is sufficient, but it takes time to volatilize the solvent of the adhesive layer, and the productivity and economy may deteriorate. It is also possible to add a UV absorber to this adhesive layer to give the laminate a UV cut ability.
- a method for manufacturing the lens of the present invention After the synthetic resin laminate of the present invention is processed into a shape for each lens by punching or the like, a lens can be produced by bending a spherical surface or an aspherical surface. When used as an injection lens, the synthetic resin laminate of the present invention can be bent and then inserted into a mold, and a transparent resin can be integrally injection-molded on the concave side to produce a lens.
- the transparent resin examples include aromatic polycarbonate, amorphous polyolefin (alicyclic polyolefin), polyacrylate, polysulfone, acetylcellulose, polystyrene, polyester, transparent polyamide, and a composition of aromatic polycarbonate and alicyclic polyester. Or a mixture thereof.
- aromatic polycarbonate is preferred. Among them, bisphenol A type polycarbonate is more preferable.
- the lens of the present invention can be subjected to a hard coat treatment.
- the resin surface is more easily damaged than glass, and in sunglasses or goggles, in order to improve the scratch resistance, it is strongly required to laminate a hard coat layer on the surface.
- the material or processing conditions of the hard coat it is appropriately selected from those having excellent scratch resistance, appearance, adhesion to the lens of the present invention, or adhesion to an inorganic layer such as a mirror coat or an antireflection coat to be subsequently coated. You can choose.
- a coating layer is formed by applying a hard coat solution to a substrate by a known method such as a dip method, a spray method, a roll coating method, a spin coating method, drying, and heating as necessary.
- the hard coat liquid can be prepared by dissolving or dispersing the hard coat composition in a solvent and further diluting with a diluting solvent as necessary.
- Diluent solvents can include alcohols, cellosolves, ketones, esters, ethers, and the like.
- eyewear (sunglasses, goggles, etc.) is fixed to the frame by balling, drilling, screw tightening, etc.
- a resin liquid was prepared using 50 parts by weight of a polyurethane prepolymer and 5 parts by weight of a curing agent and 65 parts by weight of ethyl acetate as a solvent.
- the polyurethane prepolymer is composed of a polyurethane prepolymer (diphenylmethane-4,4′-diisocyanate (MDI) having an NCO group equivalent weight (average molecular weight per functional group) of 1500 and a polypropylene glycol having an average polymerization degree of 15 (Prepared from (PPG)).
- MDI diphenylmethane-4,4′-diisocyanate
- the curing agent is a curing agent having a hydroxyl group equivalent weight of 1050 (prepared from tolylene diisocyanate and polypropylene glycol having an average degree of polymerization of 10).
- the resin solution was mixed with 0.2 parts by weight of Vivimed's “Ruby” as a photochromic dye so as to obtain a photochromic dye-containing resin solution.
- This dyed film was stretched 4 times while being immersed in an aqueous solution containing nickel acetate 2.3 g / L and boric acid 4.4 g / L at 35 ° C. for 120 seconds.
- the film was dried at room temperature for 3 minutes in a tensioned state, and then heat-treated at 110 ° C. for 3 minutes to obtain a polarizing film having a transmittance of 30% and a polarization degree of 99%.
- the synthetic resin laminate was irradiated with ultraviolet light using a UV surface irradiation device LMH133F (manufactured by ITEC System) and visually judged in a state where the photochromic dye was colored.
- Example 1 The photochromic dye-containing resin liquid (for the functional layer (B)) obtained by the method (1) above is placed on a 40 ⁇ m-thick triacetyl cellulose (TAC) film (manufactured by Fuji Film Co., Ltd.) as the resin layer (C). Coating was performed with coater # 42. Thereafter, the solvent is volatilized for 10 minutes under an atmosphere of 70 ° C., and the resin liquid surface and the synthetic resin layer (A) are 320 ⁇ m thick and an aromatic polycarbonate film having a Re of 5500 nm (trade name, manufactured by Mitsubishi Engineering Plastics). : Iupilon E-2000 (hereinafter the same)).
- TAC triacetyl cellulose
- the photochromic dye-containing resin solution (for the functional layer (B)) obtained by the method (1) is applied in the same manner to the TAC film (resin layer (C)) surface of the obtained laminate, and the resin solution The surface was bonded to an aromatic polycarbonate film having a thickness of 320 ⁇ m and Re of 5500 nm, which was a synthetic resin layer (A).
- the obtained synthetic resin laminate was heat-cured at 70 ° C. for 2 days. About the synthetic resin laminated body obtained in this way, the thickness of the functional layer (B) which has a photochromic characteristic was measured, and also the external appearance was observed. Table 1 below shows the evaluation results of the synthetic resin laminate.
- Example 2 In Example 1, instead of the aromatic polycarbonate film which is the synthetic resin layer (A), an integral laminate composed of a poly (meth) acrylate resin having a thickness of 50 ⁇ m and an aromatic polycarbonate resin having a thickness of 250 ⁇ m manufactured by a coextrusion method. A synthetic resin laminate was prepared and evaluated in the same manner as in Example 1 except that a 300 ⁇ m thick film was used and laminated so that the aromatic polycarbonate resin was bonded to the photochromic dye-containing resin solution.
- Example 3 In Example 1, a synthetic resin laminate was prepared and evaluated in the same manner as in Example 1 except that a polyamide film was used instead of the aromatic polycarbonate film as the synthetic resin layer (A).
- Example 4 In the same manner as in Example 1, a TAC film (resin layer (C)) and an aromatic polycarbonate film (synthetic resin layer (A)) were bonded together via a functional layer (B) having photochromic properties (laminate 1). ). Next, a urethane adhesive produced in the same manner as in the method (1) except that no photochromic dye is used in the polarizing film (resin layer (D)) having a thickness of about 30 ⁇ m obtained by the method (2). (Adhesive layer (E)) was applied using a micro gravure coater.
- the photochromic dye-containing resin liquid (for the functional layer (B)) obtained by the method (1) is applied to the polarizing film (resin layer (D)) surface of the laminate 2 in the same manner, and the TAC of the laminate 1 is applied.
- the film (resin layer (C)) surface was bonded.
- the obtained synthetic resin laminate was thermally cured in the same manner as in Example 1 and evaluated in the same manner.
- Example 5 The photochromic dye-containing resin liquid (for the functional layer (B)) obtained by the method (1) was applied to a TAC film (resin layer (C)) having a thickness of 50 ⁇ m with a bar coater # 36. Thereafter, the solvent was volatilized for 10 minutes in an atmosphere at 70 ° C., and the resin liquid surface was bonded to an aromatic polycarbonate film having a thickness of 320 ⁇ m and Re of 5500 nm as the synthetic resin layer (A). Another set of the same was prepared, and the photochromic dye-containing resin liquid (for the functional layer (B)) obtained by the method (1) above was applied to the TAC film (resin layer (C)) surface of one laminate. Was applied in the same manner using a bar coater # 24 and bonded to the TAC film (resin layer (C)) surface of the other laminate. The obtained synthetic resin laminate was thermally cured in the same manner as in Example 1 and evaluated in the same manner.
- Example 6 In Example 1, a synthetic resin laminate was prepared and evaluated in the same manner as in Example 1 except that an aromatic polycarbonate film having a thickness of 300 ⁇ m was used instead of the TAC film as the resin layer (C).
- Comparative Example 2 In Comparative Example 1, a synthetic resin laminate was prepared and evaluated in the same manner as in Comparative Example 1 except that the photochromic dye-containing resin liquid (for functional layer (B)) was applied using a bar coater # 42. did.
- the photochromic dye-containing resin solution (for the functional layer (B)) obtained by the method of (1) above was applied to the polarizing film (resin layer (D)) surface of the obtained laminate with a doctor blade having a gap of 150 ⁇ m.
- the solvent was volatilized for 10 minutes in an atmosphere at 70 ° C., and an aromatic polycarbonate sheet having a thickness of 320 ⁇ m and a Re of 5500 nm as the synthetic resin layer (A) was bonded to the resin liquid surface.
- the obtained synthetic resin laminate was thermally cured in the same manner as in Example 1 and evaluated in the same manner.
- a synthetic resin laminate having a photochromic property with good contrast during color development and no uneven coloring in the surface and sunglasses and goggles using the synthetic resin laminate.
- Lens eyewear can be provided.
Landscapes
- Physics & Mathematics (AREA)
- Health & Medical Sciences (AREA)
- Ophthalmology & Optometry (AREA)
- Optics & Photonics (AREA)
- General Physics & Mathematics (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Mechanical Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Optical Filters (AREA)
- Laminated Bodies (AREA)
- Eyeglasses (AREA)
- Polarising Elements (AREA)
- Non-Silver Salt Photosensitive Materials And Non-Silver Salt Photography (AREA)
Abstract
Description
<1> 2個の透明な合成樹脂層(A)と、該2個の合成樹脂層(A)間に介在する2個以上のフォトクロミック特性を有する機能層(B)と、該2個以上の機能層(B)間に介在する透明な樹脂層(C)とを有し、該機能層(B)が、フォトクロミック色素を含み、かつ、ジイソシアネート及びポリオールから得られた硬化層であることを特徴とする合成樹脂積層体である。
<2> 前記機能層(B)の一方と、前記樹脂層(C)と反対側に存在する前記合成樹脂層(A)の一方との間に、前記機能層(B)側から順に偏光特性を有する樹脂層(D)と接着材層(E)とを有する、上記<1>に記載の合成樹脂積層体である。
<3> 前記樹脂層(C)が偏光特性を有する、上記<1>または<2>に記載の合成樹脂積層体である。
<4> 前記2個の透明な合成樹脂層(A)間に介在する全ての機能層(B)の厚みの合計が30μm以上であり、かつ、個々の該機能層(B)の厚みが25μm以下である、上記<1>から<3>のいずれかに記載の合成樹脂積層体である。
<5> 前記透明な合成樹脂層(A)の少なくとも一方が、芳香族ポリカーボネート、ポリ(メタ)アクリレート、アセチルセルロース、ポリアミド、及び芳香族ポリカーボネートと脂環式ポリエステルとの混合物からなる群より選択される1つを含む単層であるか、あるいはこれらの2つ以上が積層一体化されてなる多層である、上記<1>から<4>のいずれかに記載の合成樹脂積層体である。
<6> 前記透明な合成樹脂層(A)の両方が、芳香族ポリカーボネートを含む単層である、上記<5>に記載の合成樹脂積層体である。
<7> 前記機能層(B)間に介在する前記透明な樹脂層(C)が、芳香族ポリカーボネート、ポリ(メタ)アクリレート、アセチルセルロース、ポリアミド、ポリエステル、及び芳香族ポリカーボネートと脂環式ポリエステルとの混合物からなる群より選択される1つ以上を含む、上記<1>から<6>のいずれかに記載の合成樹脂積層体である。
<8> 前記機能層(B)間に介在する前記透明な樹脂層(C)が、トリアセチルセルロースを含む、上記<7>に記載の合成樹脂積層体である。
<9> 上記<1>から<8>のいずれかに記載の合成樹脂積層体が球面あるいは非球面に曲げ加工されてなるレンズである。
<10> 上記<1>から<8>のいずれかに記載の合成樹脂積層体が曲げ加工されてなり、凹面側に透明樹脂の一体射出成形体を有するレンズである。
<11> 上記<9>または<10>に記載のレンズを用いたアイウェアである。
本発明の合成樹脂積層体の一実施形態を示す概略断面図を図1に示す。図1に記載の合成樹脂積層体は、2個の透明な合成樹脂層(A)と、該2個の合成樹脂層(A)間に介在する2個のフォトクロミック特性を有する機能層(B)と、該2個の機能層(B)間に介在する透明な樹脂層(C)とを有する。
次に、本発明の合成樹脂積層体の別の一実施形態を示す概略断面図を図2に示す。図2に記載の合成樹脂積層体は、図1に記載の合成樹脂積層体に対し、2個のフォトクロミック特性を有する機能層(B)の一方と、透明な樹脂層(C)と反対側に存在する前記合成樹脂層(A)の一方との間に、前記機能層(B)側から順に偏光特性を有する樹脂層(D)と接着材層(E)とを有する。
本発明の合成樹脂積層体は、図1および図2に示される実施形態に何ら限定されるものではない。
上記の樹脂成分の中でも、芳香族ポリカーボネートが好ましく、本発明における透明な合成樹脂層(A)の両方が、芳香族ポリカーボネートを含む単層であることがより好ましい。
中でも芳香族ジヒドロキシ化合物又はこれと少量のポリヒドロキシ化合物を、ホスゲン又は炭酸のジエステルと反応させることによって作られる分岐しても良い熱可塑性ポリカーボネート重合体がより好ましい。芳香族ジヒドロキシ化合物とは、下記一般式(a)または一般式(b)で表される化合物である。
本発明では、フォトクロミック特性を有する機能層(B)を2個以上有することが特徴の一つである。このような構成により、先行技術における問題点、即ち、フォトクロミック色素の低溶解性により、十分なコントラストを得るためにフォトクロミック特性を有するポリウレタン樹脂層の厚みが大きくなり、平滑で厚さが均一になるよう制御することが困難であったという問題点を解決することができる。
本発明におけるフォトクロミック特性を有する機能層(B)は、2個または3個であることが好ましく、2個であることがより好ましい。
前記ウレタン系樹脂としては、ジイソシアネートとポリオールとを一定割合で反応させたポリウレタンプレポリマーを用いることが好ましい。すなわち、ジイソシアネートとポリオールから得られる両末端がイソシアネート基を有する化合物である。ジイソシアネート化合物としては、ジフェニルメタン-4,4’-ジイソシアネート(MDI)が好ましい。また、ポリオールとしては、5~30の重合度を有するポリプロピレングリコール(PPG)を使用することが好ましい。ポリウレタンプレポリマーの数平均分子量は、好ましくは500~5000であり、より好ましくは1500~4000であり、特に好ましくは2000~3000である。
本発明における透明な樹脂層(C)に用いられる樹脂としては、例えば、芳香族ポリカーボネート、ポリ(メタ)アクリレート、アセチルセルロース、ポリアミド、ポリエステル、及び芳香族ポリカーボネートと脂環式ポリエステルとの混合物からなる群より選択することができる。上記樹脂の中でも、透明性、低複屈折率、耐溶剤性の点から、アセチルセルロースが好ましい。アセチルセルロースとしては、トリアセチルセルロース(TAC)、ジアセチルセルロースなどが挙げられ、中でもトリアセチルセルロース(TAC)が好ましい。本発明における透明な樹脂層(C)の厚みは、好ましくは10μmから300μmであり、より好ましくは30μmから100μmである。
本発明における透明な樹脂層(C)は、偏光特性を有するものであってもよい。
前記偏光特性を有する樹脂層(D)には、ポリビニルアルコール系樹脂のフィルムを二色性の有機染料で染色および延伸して製造されたものを用いることが好ましい。このポリビニルアルコール類としては、ポリビニルアルコール(PVA)、PVAの酢酸エステル構造を微量残したもの及びPVA誘導体または類縁体であるポリビニルホルマール、ポリビニルアセタール、エチレン-酢酸ビニル共重合体ケン化物等が例示され、特にPVAが好ましい。このポリビニルアルコール系樹脂のフィルムを一方向に延伸させつつ、2色性の有機染料を含浸もしくは吸着させ、適宜、固定し、乾燥して偏光性のフィルムとする。
PVAの重量平均分子量は、好ましくは50,000~350,000であり、より好ましくは150,000~300,000である。延伸前のPVAフィルムの厚みは、通常100~300μm程度であり、延伸後のPVA偏光フィルムは10~50μm程度が好ましい。
PVAフィルムの延伸倍率は2~8倍が好ましく、目的に応じて適宜選択されるが、延伸後の強度の点から3~6倍がより好ましい。
ポリウレタンプレポリマー50重量部に対し、硬化剤5重量部、溶媒として酢酸エチル65重量部を使用して樹脂液を調製した。前記ポリウレタンプレポリマーは、NCO基当量重量(当量重量とは官能基1個当たりの平均分子量)が1500のポリウレタンプレポリマー(ジフェニルメタン-4,4’-ジイソシアネート(MDI)と平均重合度15のポリプロピレングリコール(PPG)から調製したもの)である。また、前記硬化剤は、水酸基当量重量が1050である硬化剤(トリレンジイソシアネートと平均重合度が10のポリプロピレングリコールから調製したもの)である。前記樹脂液にフォトクロミック色素としてVivimed社の「Ruby」0.2重量部を均一になるよう混合し、フォトクロミック色素含有樹脂液とした。
ポリビニルアルコール(クラレ株式会社製、商品名:VF-PS#7500)を35℃の水中で270秒間膨潤しつつ、2倍に延伸した。
引き続いて、カヤラススプラブルーBWL、スミライトレッド4B、スミライトスプラオレンジ2GL、および10g/Lの無水硫酸ナトリウムを含む35℃の水溶液中で染色しつつ、3倍に延伸した。
この染色フィルムを酢酸ニッケル2.3g/Lおよびホウ酸4.4g/Lを含む水溶液中35℃で120秒間浸漬しつつ、4倍に延伸した。そのフィルムを緊張状態が保持された状態で室温にて3分乾燥を行った後、110℃で3分間加熱処理し、透過率30%、偏光度99%の偏光フィルムを得た。
(フォトクロミック性能評価)
恒温槽23℃下でソーラーシミュレーターHAL‐320(朝日分光製)を用いて照射強度50000ルクスの疑似太陽光を照射した。照射開始15分後の透過率(T1)を分光光度計MCPD-7700(大塚電子製)によって測定した。同様に無照射時の透過率(T2)を測定し、下記(式1)で表されるコントラストを評価した。ここで言う透過率とは、JIS Z-8701で規格される、C光源を用いた2度視野における視感透過率である。
コントラスト=T2/T1 (式1)
(外観評価)
合成樹脂積層体にUV面照射装置LMH133F(アイテックシステム製)を用いて紫外光を照射し、フォトクロミック色素を発色させた状態で目視判断した。
上記(1)の方法で得られたフォトクロミック色素含有樹脂液(機能層(B)用)を、樹脂層(C)である厚み40μmのトリアセチルセルロース(TAC)フィルム(富士フィルム社製)にバーコーター#42で塗布した。その後、70℃の雰囲気下で10分間溶剤を揮発させ、該樹脂液面と、合成樹脂層(A)である厚み320μmでRe が5500nmの芳香族ポリカーボネートフィルム(三菱エンジニアリングプラスチックス社製、商品名:ユーピロンE-2000(以下同じ))とを貼り合わせた。
得られた積層体のTACフィルム(樹脂層(C))面に、上記(1)の方法で得られたフォトクロミック色素含有樹脂液(機能層(B)用)を同様に塗布し、該樹脂液面と、合成樹脂層(A)である厚み320μmでRe が5500nmの芳香族ポリカーボネートフィルムとを貼り合わせた。
得られた合成樹脂積層体を70℃で2日間加熱硬化させた。こうして得られた合成樹脂積層体について、フォトクロミック特性を有する機能層(B)の厚みを測定し、さらに外観を観察した。下記表1に合成樹脂積層体の評価結果を示した。
実施例1において、合成樹脂層(A)である芳香族ポリカーボネートフィルムの代わりに、共押出法により製造された厚み50μmのポリ(メタ)アクリレート樹脂と厚み250μmの芳香族ポリカーボネート樹脂とからなる一体積層化された厚み300μmのフィルムを使用し、該芳香族ポリカーボネート樹脂がフォトクロミック色素含有樹脂液と貼り合わさるよう積層した以外は、実施例1と同様に合成樹脂積層体を作製し、同様に評価した。
実施例1において、合成樹脂層(A)である芳香族ポリカーボネートフィルムの代わりに、ポリアミドフィルムを使用した以外は、実施例1と同様に合成樹脂積層体を作製し、同様に評価した。
実施例1と同様に、フォトクロミック特性を有する機能層(B)を介してTACフィルム(樹脂層(C))と芳香族ポリカーボネートフィルム(合成樹脂層(A))とを貼り合わせた(積層体1)。ついで上記(2)の方法で得られた厚み約30μmの偏光フィルム(樹脂層(D))に、上記(1)の方法において、フォトクロミック色素を使用しないこと以外は同様に作製されたウレタン接着剤(接着剤層(E))を、マイクログラビアコーターを使用して塗布した。それから、70℃の雰囲気下で10分間溶剤を揮発させ、接着剤層(E)と合成樹脂層(A)である厚み320μmでReが5500nmの芳香族ポリカーボネートシートとを貼り合わせた(積層体2)。
積層体2の偏光フィルム(樹脂層(D))面に、上記(1)の方法で得られたフォトクロミック色素含有樹脂液(機能層(B)用)を同様に塗布し、積層体1のTACフィルム(樹脂層(C))面と貼り合わせた。得られた合成樹脂積層体を実施例1と同様に熱硬化させ、同様に評価した。
上記(1)の方法で得られたフォトクロミック色素含有樹脂液(機能層(B)用)を厚み50μmのTACフィルム(樹脂層(C))にバーコーター#36で塗布した。その後、70℃の雰囲気下で10分間溶剤を揮発させ、該樹脂液面と、合成樹脂層(A)である厚み320μmでReが5500nmの芳香族ポリカーボネートフィルムとを貼り合わせた。同様のものをもう一組作製し、片方の積層体のTACフィルム(樹脂層(C))面に、上記(1)の方法で得られたフォトクロミック色素含有樹脂液(機能層(B)用)をバーコーター#24を用いて同様に塗布し、もう片方の積層体のTACフィルム(樹脂層(C))面と貼り合せた。得られた合成樹脂積層体を実施例1と同様に熱硬化させ、同様に評価した。
実施例1において、樹脂層(C)としてTACフィルムの代わりに、厚み300μmの芳香族ポリカーボネートフィルムを用いること以外は、実施例1と同様に合成樹脂積層体を作製し、同様に評価した。
合成樹脂層(A)である厚み320μmでReが5500nmの芳香族ポリカーボネートシートに、上記(1)の方法で得られたフォトクロミック色素含有樹脂液(機能層(B)用)をギャップ150μmのドクターブレードで塗布し、70℃の雰囲気下で10分間溶剤を揮発させ、該樹脂液面に合成樹脂層(A)である厚み320μmでReが5500nmの芳香族ポリカーボネートシートを貼り合わせた。この合成樹脂積層体を70℃で2日間加熱硬化させた。得られた合成樹脂積層体の評価は実施例1と同様に行った。
比較例1において、フォトクロミック色素含有樹脂液(機能層(B)用)をバーコーター#42を使用して塗布したこと以外は、比較例1と同様に合成樹脂積層体を作製し、同様に評価した。
上記(2)の方法で得られた厚み約30μmの偏光フィルム(樹脂層(D))に、上記(1)の方法において、フォトクロミック色素を使用しないこと以外は同様に作製されたウレタン接着剤(接着剤層(E))を、マイクログラビアコーターを使用して塗布した。それから、70℃の雰囲気下で10分間溶剤を揮発させ、接着剤層(E)と合成樹脂層(A)である厚み320μmでReが5500nmの芳香族ポリカーボネートシートとを貼り合わせた。
得られた積層体の偏光フィルム(樹脂層(D))面に、上記(1)の方法で得られたフォトクロミック色素含有樹脂液(機能層(B)用)をギャップ150μmのドクターブレードで塗布し、70℃の雰囲気下で10分間溶剤を揮発させ、該樹脂液面に合成樹脂層(A)である厚み320μmでReが5500nmの芳香族ポリカーボネートシートを貼り合わせた。得られた合成樹脂積層体を実施例1と同様に熱硬化させ、同様に評価した。
B:フォトクロミック特性を有する機能層(B)
C:透明な樹脂層(C)
D:偏光特性を有する樹脂層(D)
E:接着材層(E)
Claims (11)
- 2個の透明な合成樹脂層(A)と、該2個の合成樹脂層(A)間に介在する2個以上のフォトクロミック特性を有する機能層(B)と、該2個以上の機能層(B)間に介在する透明な樹脂層(C)とを有し、該機能層(B)が、フォトクロミック色素を含み、かつ、ジイソシアネート及びポリオールをから得られた硬化層であることを特徴とする合成樹脂積層体。
- 前記機能層(B)の一方と、前記樹脂層(C)と反対側に存在する前記合成樹脂層(A)の一方との間に、前記機能層(B)側から順に偏光特性を有する樹脂層(D)と接着材層(E)とを有する、請求項1に記載の合成樹脂積層体。
- 前記樹脂層(C)が偏光特性を有する、請求項1または2に記載の合成樹脂積層体。
- 前記2個の透明な合成樹脂層(A)間に介在する全ての機能層(B)の厚みの合計が30μm以上であり、かつ、個々の該機能層(B)の厚みが25μm以下である、請求項1から3のいずれかに記載の合成樹脂積層体。
- 前記透明な合成樹脂層(A)の少なくとも一方が、芳香族ポリカーボネート、ポリ(メタ)アクリレート、アセチルセルロース、ポリアミド、及び芳香族ポリカーボネートと脂環式ポリエステルとの混合物からなる群より選択される1つを含む単層であるか、あるいはこれらの2つ以上が積層一体化されてなる多層である、請求項1から4のいずれかに記載の合成樹脂積層体。
- 前記透明な合成樹脂層(A)の両方が、芳香族ポリカーボネートを含む単層である、請求項5に記載の合成樹脂積層体。
- 前記機能層(B)間に介在する前記透明な樹脂層(C)が、芳香族ポリカーボネート、ポリ(メタ)アクリレート、アセチルセルロース、ポリアミド、ポリエステル、及び芳香族ポリカーボネートと脂環式ポリエステルとの混合物からなる群より選択される1つ以上を含む、請求項1から6のいずれかに記載の合成樹脂積層体。
- 前記機能層(B)間に介在する前記透明な樹脂層(C)が、トリアセチルセルロースを含む、請求項7に記載の合成樹脂積層体。
- 請求項1から8のいずれかに記載の合成樹脂積層体が球面あるいは非球面に曲げ加工されてなるレンズ。
- 請求項1から8のいずれかに記載の合成樹脂積層体が曲げ加工されてなり、凹面側に透明樹脂の一体射出成形体を有するレンズ。
- 請求項9または10に記載のレンズを用いたアイウェア。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018504509A JP6900364B2 (ja) | 2016-03-10 | 2017-03-07 | フォトクロミック特性を有する合成樹脂積層体およびそれを用いたレンズ |
| EP17763241.1A EP3428698A4 (en) | 2016-03-10 | 2017-03-07 | SYNTHETIC RESIN LAMINATE HAVING PHOTOCHROMIC PROPERTIES USING THE SAME |
| CN201780016104.3A CN108700689A (zh) | 2016-03-10 | 2017-03-07 | 具有光致变色特性的合成树脂叠层体和使用其的透镜 |
| KR1020187028699A KR102263213B1 (ko) | 2016-03-10 | 2017-03-07 | 포토크로믹 특성을 갖는 합성 수지 적층체 및 그것을 이용한 렌즈 |
| US16/082,116 US10737471B2 (en) | 2016-03-10 | 2017-03-07 | Synthetic resin laminate having photochromic properties and lens using same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016-046844 | 2016-03-10 | ||
| JP2016046844 | 2016-03-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017154901A1 true WO2017154901A1 (ja) | 2017-09-14 |
Family
ID=59789390
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/009000 WO2017154901A1 (ja) | 2016-03-10 | 2017-03-07 | フォトクロミック特性を有する合成樹脂積層体およびそれを用いたレンズ |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US10737471B2 (ja) |
| EP (1) | EP3428698A4 (ja) |
| JP (1) | JP6900364B2 (ja) |
| KR (1) | KR102263213B1 (ja) |
| CN (1) | CN108700689A (ja) |
| TW (1) | TWI780043B (ja) |
| WO (1) | WO2017154901A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2018164062A1 (ja) * | 2017-03-06 | 2019-11-07 | 日東電工株式会社 | 光学機能層付偏光フィルムおよび液晶表示装置 |
| WO2020204176A1 (ja) * | 2019-04-03 | 2020-10-08 | 株式会社トクヤマ | フォトクロミック光学物品およびその製造方法 |
| JP7344112B2 (ja) | 2019-12-26 | 2023-09-13 | 株式会社トクヤマ | フォトクロミック性接着組成物およびフォトクロミック光学物品 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109648956B (zh) * | 2018-12-29 | 2021-03-05 | 温州富奇士光电材料有限公司 | 光致变色材料及其制备方法、应用 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002099513A1 (fr) * | 2001-06-01 | 2002-12-12 | Mitsubishi Gas Chemical Company, Inc. | Produit en plastique moule a caracteristiques photochromiques et/ou polarisantes |
| JP2005500925A (ja) * | 2001-08-30 | 2005-01-13 | キム,ス−ジン | 光可逆性変色度数偏光サングラスレンズ及び製造方法 |
| JP2007017636A (ja) * | 2005-07-06 | 2007-01-25 | Ricoh Co Ltd | 静電潜像担持体、並びに画像形成方法、画像形成装置、及びプロセスカートリッジ |
| JP2007025609A (ja) * | 2005-07-21 | 2007-02-01 | Mgc Filsheet Co Ltd | 偏光調光特性を有する光制御プラスチックレンズおよびその製造方法 |
| JP2012048113A (ja) * | 2010-08-30 | 2012-03-08 | Hoya Corp | 偏光レンズ |
| JP2014032273A (ja) * | 2012-08-02 | 2014-02-20 | Talex Optical Co Ltd | フォトクロミックレンズ |
| JP2015535946A (ja) * | 2012-09-14 | 2015-12-17 | トランジションズ オプティカル, インコーポレイテッド | 2層の少なくとも部分的に交差したフォトクロミック二色層を有するフォトクロミック物品 |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS63178193A (ja) | 1987-01-20 | 1988-07-22 | Nippon Sheet Glass Co Ltd | フオトクロミツク性を有する被覆用組成物 |
| JP4586953B2 (ja) | 2000-06-09 | 2010-11-24 | 三菱瓦斯化学株式会社 | 偏光性とフォトクロミック性を併せ持つ合成樹脂積層体 |
| AU2003254253A1 (en) * | 2002-07-31 | 2004-02-16 | Bmc Industries, Inc. | Polyurethane laminates for photochromic lenses |
| CN100408590C (zh) * | 2003-08-12 | 2008-08-06 | 上海迪赛诺医药科技开发有限公司 | 螺吡喃和螺噁嗪类光致变色化合物修饰核苷的方法 |
| US7273659B2 (en) * | 2003-12-18 | 2007-09-25 | Lintec Corporation | Photochromic film material |
| TWI323254B (en) * | 2004-02-02 | 2010-04-11 | Mgc Filsheet Co Ltd | Light control plastic lens, coated sheet-like light control element for the lens production, and a production method for light control plastic lens |
| CN100432067C (zh) * | 2006-06-05 | 2008-11-12 | 南开大学 | 二苯并、二萘并吡喃和二苯并、二萘并螺吡喃类化合物及制备 |
| CN101215282A (zh) * | 2007-12-27 | 2008-07-09 | 上海交通大学 | 光致变色萘并吡喃类化合物及其合成方法 |
| US7884992B1 (en) * | 2009-08-13 | 2011-02-08 | Darwin Optical Co., Ltd. | Photochromic optical article |
| JP5559497B2 (ja) | 2009-08-18 | 2014-07-23 | 山本光学株式会社 | 光学物品 |
| JPWO2011049108A1 (ja) * | 2009-10-21 | 2013-03-14 | 三菱瓦斯化学株式会社 | 機能性シートおよびそれを用いたレンズ |
| CN103097131A (zh) * | 2010-08-26 | 2013-05-08 | 三菱瓦斯化学株式会社 | 合成树脂叠层体 |
| TWI559024B (zh) * | 2011-10-03 | 2016-11-21 | Mitsubishi Gas Chemical Co | Polarized glasses |
| TWI592714B (zh) | 2012-08-14 | 2017-07-21 | Mitsubishi Gas Chemical Co | Special polycarbonate polarized glasses |
| CN102875822B (zh) * | 2012-10-30 | 2014-04-16 | 天津科技大学 | 一种光致变色胶乳乳液的制备方法 |
| CN103254453B (zh) * | 2013-04-24 | 2015-09-09 | 华中科技大学 | 一种聚合物分散有机凝胶光致变色薄膜的制备方法 |
| CN104016996B (zh) * | 2014-06-04 | 2017-01-11 | 上海甘田光学材料有限公司 | 苯并吡喃类光致变色衍生物 |
-
2017
- 2017-03-07 US US16/082,116 patent/US10737471B2/en active Active
- 2017-03-07 EP EP17763241.1A patent/EP3428698A4/en not_active Withdrawn
- 2017-03-07 JP JP2018504509A patent/JP6900364B2/ja active Active
- 2017-03-07 WO PCT/JP2017/009000 patent/WO2017154901A1/ja active Application Filing
- 2017-03-07 KR KR1020187028699A patent/KR102263213B1/ko active IP Right Grant
- 2017-03-07 TW TW106107408A patent/TWI780043B/zh active
- 2017-03-07 CN CN201780016104.3A patent/CN108700689A/zh active Pending
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002099513A1 (fr) * | 2001-06-01 | 2002-12-12 | Mitsubishi Gas Chemical Company, Inc. | Produit en plastique moule a caracteristiques photochromiques et/ou polarisantes |
| JP2005500925A (ja) * | 2001-08-30 | 2005-01-13 | キム,ス−ジン | 光可逆性変色度数偏光サングラスレンズ及び製造方法 |
| JP2007017636A (ja) * | 2005-07-06 | 2007-01-25 | Ricoh Co Ltd | 静電潜像担持体、並びに画像形成方法、画像形成装置、及びプロセスカートリッジ |
| JP2007025609A (ja) * | 2005-07-21 | 2007-02-01 | Mgc Filsheet Co Ltd | 偏光調光特性を有する光制御プラスチックレンズおよびその製造方法 |
| JP2012048113A (ja) * | 2010-08-30 | 2012-03-08 | Hoya Corp | 偏光レンズ |
| JP2014032273A (ja) * | 2012-08-02 | 2014-02-20 | Talex Optical Co Ltd | フォトクロミックレンズ |
| JP2015535946A (ja) * | 2012-09-14 | 2015-12-17 | トランジションズ オプティカル, インコーポレイテッド | 2層の少なくとも部分的に交差したフォトクロミック二色層を有するフォトクロミック物品 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3428698A4 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2018164062A1 (ja) * | 2017-03-06 | 2019-11-07 | 日東電工株式会社 | 光学機能層付偏光フィルムおよび液晶表示装置 |
| WO2020204176A1 (ja) * | 2019-04-03 | 2020-10-08 | 株式会社トクヤマ | フォトクロミック光学物品およびその製造方法 |
| JP7344112B2 (ja) | 2019-12-26 | 2023-09-13 | 株式会社トクヤマ | フォトクロミック性接着組成物およびフォトクロミック光学物品 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20180120736A (ko) | 2018-11-06 |
| JPWO2017154901A1 (ja) | 2019-01-17 |
| CN108700689A (zh) | 2018-10-23 |
| EP3428698A1 (en) | 2019-01-16 |
| US20190084278A1 (en) | 2019-03-21 |
| TWI780043B (zh) | 2022-10-11 |
| TW201808633A (zh) | 2018-03-16 |
| KR102263213B1 (ko) | 2021-06-10 |
| JP6900364B2 (ja) | 2021-07-07 |
| US10737471B2 (en) | 2020-08-11 |
| EP3428698A4 (en) | 2019-09-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2017154901A1 (ja) | フォトクロミック特性を有する合成樹脂積層体およびそれを用いたレンズ | |
| US9739910B2 (en) | Polarizing plate and liquid crystal display device | |
| US7035010B2 (en) | Polarized lenses with variable transmission | |
| US10444547B2 (en) | Functional laminated spectacle lens having functional layer formed by spin-coating | |
| WO2019163728A1 (ja) | 機能性積層体、及び機能性積層体を用いた機能性レンズ | |
| US20170031072A1 (en) | Polarizing plate and liquid crystal display device | |
| KR101972191B1 (ko) | 방향족 폴리카보네이트제 편광 렌즈 | |
| JP2006264109A (ja) | プラスチックレンズの製造方法 | |
| WO2021235121A1 (ja) | インサート成形用機能性偏光素子および機能性偏光レンズ | |
| US10816699B2 (en) | Functional sheet including polyester resin, and lens using functional sheet | |
| US11648742B2 (en) | Methods for preparing functional optical films | |
| TW202041365A (zh) | 偏光板 | |
| JP2007316617A (ja) | 偏光フィルム | |
| EP3871866B1 (en) | Primer coating composition for polarizing ophthalmic lens | |
| JPWO2019004337A1 (ja) | 眼鏡レンズ、組成物、眼鏡レンズの製造方法 | |
| JP2004246157A (ja) | 紫外線から目を保護する眼鏡 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2018504509 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20187028699 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2017763241 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2017763241 Country of ref document: EP Effective date: 20181010 |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17763241 Country of ref document: EP Kind code of ref document: A1 |