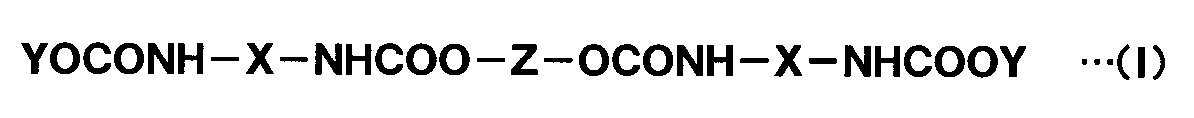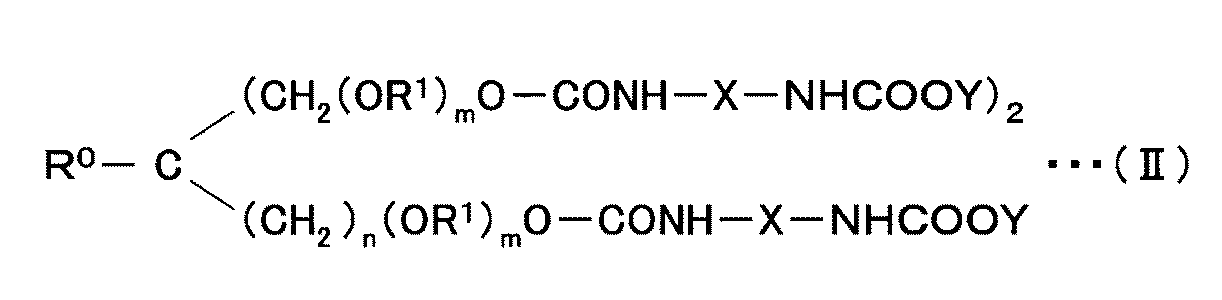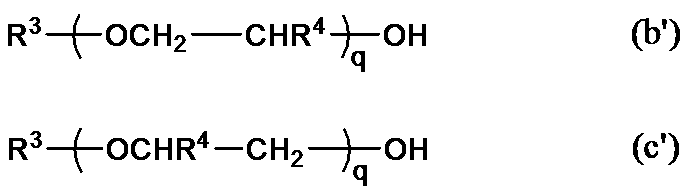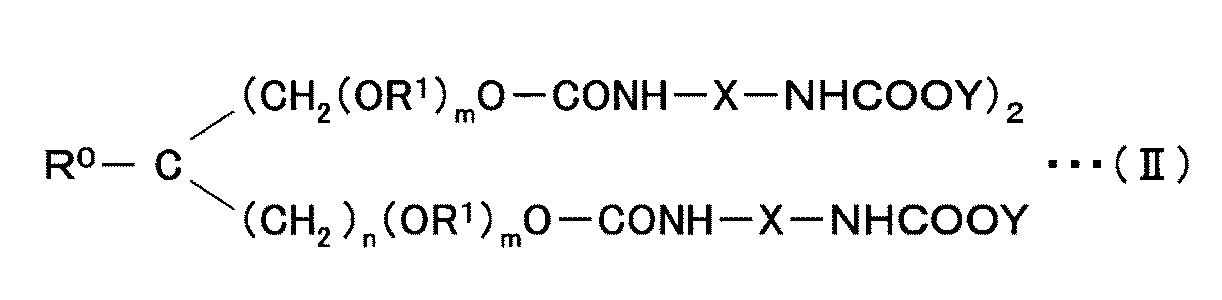WO2017131101A1 - 複層塗膜形成方法 - Google Patents
複層塗膜形成方法 Download PDFInfo
- Publication number
- WO2017131101A1 WO2017131101A1 PCT/JP2017/002778 JP2017002778W WO2017131101A1 WO 2017131101 A1 WO2017131101 A1 WO 2017131101A1 JP 2017002778 W JP2017002778 W JP 2017002778W WO 2017131101 A1 WO2017131101 A1 WO 2017131101A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- aqueous
- coating film
- resin
- coating composition
- group
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/04—Homopolymers or copolymers of esters
- C09D133/06—Homopolymers or copolymers of esters of esters containing only carbon, hydrogen and oxygen, the oxygen atom being present only as part of the carboxyl radical
- C09D133/062—Copolymers with monomers not covered by C09D133/06
- C09D133/066—Copolymers with monomers not covered by C09D133/06 containing -OH groups
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D175/00—Coating compositions based on polyureas or polyurethanes; Coating compositions based on derivatives of such polymers
- C09D175/04—Polyurethanes
- C09D175/14—Polyurethanes having carbon-to-carbon unsaturated bonds
- C09D175/16—Polyurethanes having carbon-to-carbon unsaturated bonds having terminal carbon-to-carbon unsaturated bonds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D—PROCESSES FOR APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D1/00—Processes for applying liquids or other fluent materials
- B05D1/36—Successively applying liquids or other fluent materials, e.g. without intermediate treatment
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D—PROCESSES FOR APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D3/00—Pretreatment of surfaces to which liquids or other fluent materials are to be applied; After-treatment of applied coatings, e.g. intermediate treating of an applied coating preparatory to subsequent applications of liquids or other fluent materials
- B05D3/02—Pretreatment of surfaces to which liquids or other fluent materials are to be applied; After-treatment of applied coatings, e.g. intermediate treating of an applied coating preparatory to subsequent applications of liquids or other fluent materials by baking
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D—PROCESSES FOR APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D7/00—Processes, other than flocking, specially adapted for applying liquids or other fluent materials to particular surfaces or for applying particular liquids or other fluent materials
- B05D7/24—Processes, other than flocking, specially adapted for applying liquids or other fluent materials to particular surfaces or for applying particular liquids or other fluent materials for applying particular liquids or other fluent materials
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
- B32B15/095—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin comprising polyurethanes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/28—Layered products comprising a layer of synthetic resin comprising synthetic resins not wholly covered by any one of the sub-groups B32B27/30 - B32B27/42
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/40—Layered products comprising a layer of synthetic resin comprising polyurethanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D175/00—Coating compositions based on polyureas or polyurethanes; Coating compositions based on derivatives of such polymers
- C09D175/04—Polyurethanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D179/00—Coating compositions based on macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen, with or without oxygen, or carbon only, not provided for in groups C09D161/00 - C09D177/00
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/002—Priming paints
Definitions
- the present invention relates to an aqueous intermediate coating composition and a method for forming a multilayer coating film using an aqueous base coating composition.
- a plurality of coating films having various roles are sequentially formed on the surface of an object such as an automobile body to protect the object to be coated and at the same time impart a beautiful appearance and an excellent design.
- an undercoat coating film such as an electrodeposition coating film is formed on an object having excellent conductivity, and an intermediate coating film is formed thereon.
- a method of sequentially forming a film, a base coating film and a clear coating film is common.
- the crosslinking density of the resulting coating film was lowered, and the coating film performance such as water resistance and chipping resistance was sometimes inferior.
- Patent Document 1 discloses that an intermediate coating film is formed by coating an aqueous intermediate coating composition on a substrate having both a steel plate and a plastic substrate. An aqueous base coating composition is applied onto the coating film to form a base coating film, and then an organic solvent-type clear coating composition is applied to form a clear coating film.
- a method for forming a multilayer coating film in which three layers of a clear coating film are heated and cured wherein the aqueous base coating composition comprises (a) an acrylic resin emulsion, (b) a water-soluble acrylic resin, and (c) melamine It describes about the formation method of the multilayer coating film containing resin and (d) propylene glycol monoalkyl ether.
- the multilayer coating film obtained by this forming method is cured at a temperature of 100 ° C. or lower, for example, there is a possibility that sufficient chipping resistance may not be obtained.
- the present invention solves the above-mentioned conventional problems, and its object is to form a multilayer coating film that can form a multilayer coating film having excellent coating performance even at low temperature curing. It is to provide a method.
- the aqueous intermediate coating composition is An aqueous resin (A1) having a hydroxyl group and a carboxyl group, A polyisocyanate compound (B), and a hydrophilized modified carbodiimide compound (C),
- the aqueous base coating composition is:
- the aqueous resin (A2) having a hydroxyl group and a carboxyl group contained in the aqueous base coating composition has a hydroxyl value of 80 to 200 mgKOH / g in terms of resin solids
- the hydrophilized modified carbodiimide compound (C) is represented by the following general formula (I), (II) or (III), [X is a bifunctional organic group containing at least one carbodiimide group, Y is a structure obtained by removing a hydroxyl group from the same or different polyalkylene glycol monoalkyl ether, and Z is a number average molecular weight of 200. It is a structure in which a hydroxyl group is removed from a bifunctional polyol of ⁇ 5,000.
- [X is a bifunctional organic group containing at least one carbodiimide group
- Y is a structure obtained by removing a hydroxyl group from the same or different polyalkylene glycol monoalkyl ether
- R 0 is a hydrogen or methyl group.
- R 1 is an alkylene group having 4 or less carbon atoms
- n is 0 or 1
- m is 0-60.
- [X is a bifunctional organic group containing at least one carbodiimide group
- Y has a structure in which a hydroxyl group is removed from the same or different polyalkylene glycol monoalkyl ether.
- the aqueous polyurethane resin (E) has a glass transition point (Tg) of ⁇ 50 ° C. or lower,
- the breaking elongation of the cured film of the aqueous polyurethane resin (E) is 400% or more at ⁇ 20 ° C.
- Content of the said water-based polyurethane resin (E) is 10 mass% or more and 50 mass% or less with respect to the resin solid content of the said water-based base coating composition
- the content of the water-dispersible block polyisocyanate compound (D) contained in the aqueous base coating composition is 10 to 25% by mass with respect to the resin solid content of the aqueous base coating composition
- the content of the hydrophilically modified carbodiimide compound (C) contained in the aqueous base coating composition is 1 to 9% by mass with respect to the resin solid content of the aqueous coating composition.
- a method for forming a multilayer coating film [2] The content of the hydrophilized modified carbodiimide compound (C) contained in the aqueous intermediate coating composition is 1 to 8% by mass based on the resin solid content of the aqueous intermediate coating composition. Forming method. [3] Content of the said water-based polyurethane resin (E) is the said multilayer coating-film formation method which is 15 to 30 mass% with respect to the resin solid content of the said aqueous base coating composition.
- the content of the hydrophilized modified carbodiimide compound (C) contained in the aqueous base coating composition is 1.5 to 7% by mass based on the resin solid content of the aqueous base coating composition
- the multilayer coating film forming method, wherein the aqueous base coating composition further includes an aqueous resin (F) having a hydroxyl value of less than 80 mgKOH / g.
- the multilayer coating film forming method wherein the object to be coated includes a steel plate part and a resin part.
- the method for forming a multilayer coating film, wherein the curing step is a step of heating and curing the obtained uncured intermediate coating film, base coating film, and clear coating film.
- the cured reaction proceeds well even under heating conditions under low-temperature conditions (for example, heating conditions of 100 ° C. or less), and has excellent coating film properties.
- low-temperature conditions for example, heating conditions of 100 ° C. or less
- the multilayer coating film forming method of the present invention is, for example, a steel plate part and a resin that require excellent coating film properties (water resistance, chipping resistance, etc.) despite the difficulty of high-temperature heat curing treatment. It can use suitably for the coating of the to-be-coated object which has a part.
- the multilayer coating film forming method of the present invention is An intermediate coating film forming step of forming an uncured intermediate coating film by coating a water-based intermediate coating composition on an object to be coated; An aqueous base coating composition is applied on the obtained uncured intermediate coating film to form an uncured base coating film, and the obtained uncured intermediate coating A curing process for heating and curing the film and the base coating film, Is included.
- the above-mentioned specific aqueous intermediate coating composition and aqueous base coating composition are used in combination, for example, when the aqueous coating composition is baked and cured under low temperature curing conditions.
- a multilayer coating film having good water resistance and chipping resistance can be obtained.
- the coating composition used in each coating film forming step will be described.
- the aqueous resin (A1) having a hydroxyl group and a carboxyl group is a binder component that undergoes a curing reaction with the polyisocyanate compound (B) and the hydrophilized modified carbodiimide compound (C) described later.
- the aqueous resin (A1) having a hydroxyl group and a carboxyl group used in the present invention is: -The hydroxyl value in terms of resin solids is 80 to 200 mg KOH / g, The acid value in terms of resin solids is 10 to 40 mg KOH / g, Is a requirement.
- the hydroxyl value in terms of resin solid content is more preferably from 80 to 160 mgKOH / g, and the acid value in terms of resin solid content is more preferably from 15 to 35 mgKOH / g.
- the aqueous resin (A1) having a hydroxyl group and a carboxyl group used in the present invention has a higher hydroxyl value than the acid value.
- the aqueous resin (A1) may be composed of a single resin that satisfies the above requirements for the hydroxyl value and acid value in terms of resin solids, or a plurality that satisfies the above requirements for the hydroxyl value and acid value. You may be comprised from these resin.
- the aqueous resin (A1) has two types of functional groups, a hydroxyl group and a carboxyl group, as reactive groups involved in curing.
- the hydroxyl group of the aqueous resin (A1) reacts with the polyisocyanate compound (B), and the carboxyl group of the aqueous resin (A1) reacts with the hydrophilized modified carbodiimide compound (C). .
- the type of the aqueous resin (A1) is not particularly limited as long as it satisfies the requirements for the hydroxyl group and carboxyl group, but an acrylic resin and / or a polyester resin are preferable because they are easy to produce and obtain. .
- an acrylic resin alone or a mixture of an acrylic resin and a polyester resin as the aqueous resin (A1).
- an intermediate coating composition it is more preferable to use a mixture of an acrylic resin and a polyester resin as the aqueous resin (A1).
- a top coat base coating composition it is more preferable to use an acrylic resin as the aqueous resin (A1).
- the acrylic resin that can be suitably used as the aqueous resin (A1) includes, for example, a monomer containing an ⁇ , ⁇ -ethylenically unsaturated monomer having a hydroxyl group and an ⁇ , ⁇ -ethylenically unsaturated monomer having a carboxyl group.
- a monomer containing an ⁇ , ⁇ -ethylenically unsaturated monomer having a hydroxyl group and an ⁇ , ⁇ -ethylenically unsaturated monomer having a carboxyl group By subjecting the hydroxyl group and the carboxyl group to acrylic copolymerization in an amount that satisfies the above-mentioned requirements for the hydroxyl value and acid value, the desired resin can be obtained.
- Examples of the ⁇ , ⁇ -ethylenically unsaturated monomer having a hydroxyl group include 2-hydroxyethyl (meth) acrylate, 2-hydroxypropyl (meth) acrylate, 4-hydroxybutyl (meth) acrylate, allyl alcohol, methacryl Mention may be made of adducts of alcohol, hydroxyethyl (meth) acrylate and ⁇ -caprolactone. Among these, preferred are 2-hydroxyethyl (meth) acrylate, 4-hydroxybutyl (meth) acrylate, and an adduct of hydroxyethyl (meth) acrylate and ⁇ -caprolactone.
- “(meth) acryl” means both acrylic and methacrylic.
- ⁇ , ⁇ -ethylenically unsaturated monomers having a carboxyl group include acrylic acid, methacrylic acid, acrylic acid dimer, crotonic acid, 2-acryloyloxyethylphthalic acid, 2-acryloyloxyethyl succinic acid, ⁇ - Carboxy-polycaprolactone mono (meth) acrylate, maleic acid, fumaric acid, itaconic acid and the like can be mentioned. Among these, acrylic acid and methacrylic acid are preferable.
- ⁇ , ⁇ -ethylenically unsaturated monomers can be used as necessary.
- the other ⁇ , ⁇ -ethylenically unsaturated monomers include (meth) acrylic acid esters (for example, methyl (meth) acrylate, ethyl (meth) acrylate, n-propyl (meth) acrylate, (meth) acrylic).
- N-butyl acid isobutyl (meth) acrylate, t-butyl (meth) acrylate, 2-ethylhexyl (meth) acrylate, lauryl methacrylate, phenyl acrylate, isobornyl (meth) acrylate, cyclohexyl methacrylate, ( (Meth) acrylic acid t-butylcyclohexyl, (meth) acrylic acid dicyclopentadienyl, (meth) acrylic acid dihydrodicyclopentadienyl, etc.), polymerizable amide compounds (for example, (meth) acrylamide, N-methylol ( (Meth) acrylamide, N-butoxymethyl (meth) acrylic Amide etc.).
- Examples of the method for obtaining the aqueous resin (A1) include a method of obtaining an acrylic resin after performing solution polymerization and a method of obtaining an emulsion by performing emulsion polymerization in an aqueous medium.
- a crosslinkable monomer can be used as the other ⁇ , ⁇ -ethylenically unsaturated monomer.
- the crosslinkable monomer is a compound having two or more radically polymerizable ethylenically unsaturated groups in the molecule, and examples thereof include divinylbenzene, allyl (meth) acrylate, and ethylene glycol di (meth) acrylate. .
- the solution polymerization is generally carried out by stirring under heating conditions while mixing a mixture of ⁇ , ⁇ -ethylenically unsaturated monomers used as raw materials together with a polymerization initiator into a solvent.
- the conditions for solution polymerization are, for example, a polymerization temperature of 60 to 160 ° C. and a dropping time of 0.5 to 10 hours.
- the ⁇ , ⁇ -ethylenically unsaturated monomer used as the raw material can be polymerized in two stages. In this case, it is only necessary that the ⁇ , ⁇ -ethylenically unsaturated monomer used as a raw material satisfies the requirements for the hydroxyl group and the carboxyl group.
- the polymerization initiator is not particularly limited as long as it is used for normal polymerization, and examples thereof include azo compounds and peroxides. Generally, the amount of the polymerization initiator with respect to 100 parts by mass of the monomer mixture is 0.1 to 18 parts by mass, preferably 0.3 to 12 parts by mass.
- the solvent that can be used here is not particularly limited as long as it does not adversely affect the reaction, and examples thereof include alcohols, ketones, ethers, and hydrocarbon solvents. Furthermore, in order to adjust the molecular weight, a mercaptan such as lauryl mercaptan and a chain transfer agent such as ⁇ -methylstyrene dimer can be used as necessary.
- the number average molecular weight of the acrylic resin thus obtained by solution polymerization is preferably 4,000 to 20,000.
- the number average molecular weight of the acrylic resin obtained by solution polymerization can be measured by gel permeation chromatography (GPC) using a polystyrene standard sample standard.
- the glass transition point (Tg) of the acrylic resin is preferably in the range of ⁇ 20 to 80 ° C.
- the glass transition point of an acrylic resin can be calculated
- DSC differential scanning calorimeter
- the aqueous resin (A1) can be obtained by removing the solvent as necessary from the acrylic resin obtained by the solution polymerization and then adding the basic compound to make it aqueous.
- the basic compound include ammonia, methylamine, ethylamine, dimethylamine, diethylamine, trimethylamine, triethylamine, dimethylethanolamine, diethanolamine, diethylaminoethanol, triethanolamine and the like.
- the amount of the basic compound added is preferably such that the neutralization rate is 60 to 100% with respect to the carboxyl group of the acrylic resin obtained by the solution polymerization. When the neutralization rate is less than 60%, the aqueous solution is not sufficient and the storage stability may be inferior.
- the resin solid content of the aqueous resin (A1) thus obtained is generally 25 to 55% by mass.
- the acrylic resin thus obtained can be used as an acrylic water dispersion.
- Such an acrylic water dispersion preferably has a volume average particle diameter in the range of 0.01 to 1 ⁇ m.
- the volume average particle diameter is in the above range, there is an advantage that the stability of the aqueous dispersion is improved and the appearance of the obtained coating film is improved.
- the acrylic emulsion described later and the volume average particle diameter can be adjusted by adjusting the monomer composition and / or the emulsion polymerization conditions.
- the emulsifier is dissolved in an aqueous medium containing water or an organic solvent such as alcohol as necessary, and heated. Under stirring, a mixture of an ⁇ , ⁇ -ethylenically unsaturated monomer used as a raw material and a polymerization initiator can be added dropwise. A mixture of ⁇ , ⁇ -ethylenically unsaturated monomers used as a raw material may be pre-emulsified with an emulsifier and water.
- Polymerization initiators that can be suitably used for emulsion polymerization include azo oily compounds (for example, azobisisobutyronitrile, 2,2′-azobis (2-methylbutyronitrile), and 2,2′- Azobis (2,4-dimethylvaleronitrile) and the like, and aqueous compounds (eg, anionic 4,4′-azobis (4-cyanovaleric acid), 2,2-azobis (N- (2-carboxyethyl)) -2-methylpropionamidine) and cationic 2,2′-azobis (2-methylpropionamidine)); and redox oily peroxides (eg, benzoyl peroxide, parachlorobenzoyl peroxide, lauroyl peroxide) And t-butyl perbenzoate), and aqueous peroxides (eg, potassium persulfate and hydrogen peroxide) Etc. ammonium) and the like.
- azo oily compounds for example, azo
- emulsifier a general emulsifier usually used by those skilled in the art can be used.
- reactive emulsifiers such as Antox MS-60 (manufactured by Nippon Emulsifier Co., Ltd.), Eleminol JS-2 (manufactured by Sanyo Kasei Kogyo Co., Ltd.), Adekari Soap NE-20 (manufactured by ADEKA) and Aqualon HS -10 (Daiichi Kogyo Seiyaku Co., Ltd.), Latemul PD-104 (Kao Corp.) and the like are particularly preferred.
- a mercaptan such as lauryl mercaptan and a chain transfer agent such as ⁇ -methylstyrene dimer can be used as necessary.
- the reaction temperature is determined by the initiator. For example, it is preferably 60 to 90 ° C. for azo initiators and peroxides, and preferably 30 to 70 ° C. for redox systems. In general, the reaction time is 1 to 8 hours. Generally, the amount of the initiator with respect to 100 parts by mass of the monomer mixture is 0.1 to 5% by mass.
- the emulsion polymerization can be performed in multiple stages, for example, in two stages.
- a part of the mixture of ⁇ , ⁇ -ethylenically unsaturated monomers used as the raw material is subjected to emulsion polymerization, and the remainder of the ⁇ , ⁇ -ethylenically unsaturated monomer mixture is further added to emulsify. Polymerization is performed.
- the above emulsion can be used at a pH of 5 to 10 by neutralizing with a basic compound from the viewpoint of storage stability.
- the basic compound may be the same as that used in making the acrylic resin obtained in the previous solution polymerization aqueous.
- the neutralization is preferably performed by adding the basic compound to the system before or after emulsion polymerization.
- the number average molecular weight is preferably 10,000 to 80,000.
- the acrylic emulsion has a hydroxyl value of 80 to 200 mgKOH / g, an acid value of 10 to 40 mgKOH / g, and a number average molecular weight in the range of 10,000 to 80,000.
- the crosslinking density in the obtained coating film is in a better range.
- the number average molecular weight is in a relatively high range of 10,000 to 80,000, and the acrylic emulsion has a large number of hydroxyl groups within the above range, whereby the aqueous resin (A1) has It is considered that the low-temperature curability of the reacting polyisocyanate compound (B) is ensured, and thereby the crosslink density in the obtained coating film is in a better range.
- the number average molecular weight of the acrylic emulsion can be measured by gel permeation chromatography (GPC) using a polystyrene standard sample standard after removing water by drying under reduced pressure or the like.
- the aqueous resin (A1) may contain a polyester resin.
- the polyester resin that can be used as the aqueous resin (A1) is generally prepared by condensing a polyhydric alcohol component and a polybasic acid component so as to satisfy the requirements for the hydroxyl group and the carboxyl group. Can do.
- polyhydric alcohol component examples include, for example, ethylene glycol, diethylene glycol, propylene glycol, 1,3-propanediol, 1,4-butanediol, 1,5-pentanediol, 1,6-hexanediol, 2, 2-diethyl-1,3-propanediol, neopentyl glycol, 1,9-nonanediol, 1,4-cyclohexanediol, hydroxypivalic acid neopentyl glycol ester, 2-butyl-2-ethyl-1,3-propane Examples thereof include hydroxycarboxylic acid components such as diol, 3-methyl-1,5-pentanediol, and 2,2,4-trimethylpentanediol.
- polybasic acid component examples include, for example, aromatic polyvalent carboxylic acids and acid anhydrides such as phthalic anhydride, isophthalic acid, terephthalic acid, trimellitic anhydride, tetrachlorophthalic anhydride, pyromellitic anhydride; Hexahydrophthalic anhydride, tetrahydrophthalic anhydride, alicyclic polycarboxylic acids and anhydrides such as 1,4- and 1,3-cyclohexanedicarboxylic acid; maleic anhydride, fumaric acid, succinic anhydride, adipic acid, sebacine
- polybasic acid components such as aliphatic polyvalent carboxylic acids such as acids and anhydrides, and anhydrides thereof. If necessary, a monobasic acid such as benzoic acid or t-butylbenzoic acid may be used in combination.
- mono-epoxide compounds such as monohydric alcohol, Cardura E (trade name: manufactured by Ciel Chemical), and lactones ( ⁇ -propiolactone, dimethylpropiolactone, butyrolactone, ⁇ -valerolactone, ⁇ -caprolactone, ⁇ -caprolactone, etc.) may be used in combination.
- fatty acids such as castor oil and dehydrated castor oil, and an oil component that is one or a mixture of two or more of these fatty acids may be added to the acid component and the alcohol component.
- the number average molecular weight of the polyester resin thus obtained is preferably 500 to 20,000, and more preferably 1,500 to 10,000. If the number average molecular weight is less than 500, the storage stability when the polyester resin is dispersed in water may be lowered. On the other hand, when the number average molecular weight exceeds 20,000, the viscosity of the polyester resin is increased, so that the solid content concentration in the case of a coating composition is decreased, and the coating workability may be decreased.
- the glass transition point of the polyester resin is preferably ⁇ 20 to 80 ° C. When the glass transition point is less than ⁇ 20 ° C., the hardness of the resulting coating film may be lowered, and when it exceeds 80 ° C., the base concealability may be lowered.
- the glass transition point is more preferably 0 to 60 ° C.
- the glass transition point of the polyester resin can be obtained by calculation from the kind and amount of the monomer used for preparing the polyester resin, as in the case of the acrylic resin. Moreover, you may measure the glass transition point of a polyester resin with a differential scanning calorimeter (DSC).
- DSC differential scanning calorimeter
- the aqueous resin (A1) can be obtained by neutralizing the polyester resin thus obtained with the basic compounds mentioned above.
- the content of the aqueous resin (A1) contained in the aqueous intermediate coating composition in the present invention is preferably 30 to 80% by mass with respect to the resin solid content of the aqueous intermediate coating composition. More preferably, it is 80 mass%.
- the aqueous coating composition when used as an intermediate coating composition, when a mixture of an acrylic resin and a polyester resin is used as the aqueous resin (A1), the ratio of the acrylic resin to the polyester resin is acrylic resin /
- the polyester resin is preferably in the range of 7/1 to 0.5 / 1, more preferably in the range of 6/1 to 1/1.
- Polyisocyanate compound (B) The aqueous intermediate coating composition in the present invention includes two components, a polyisocyanate compound (B) and a hydrophilized modified carbodiimide compound (C), as components for curing the aqueous resin (A1).
- the polyisocyanate compound (B) may be water-dispersible or hydrophobic. Even if it is hydrophobic, water dispersibility is ensured by the interaction with the hydrophilic modified carbodiimide compound (C) having excellent water dispersibility described later.
- hydrophobic polyisocyanate compounds (B) include tolylene diisocyanate (TDI), 4,4′-diphenylmethane diisocyanate (MDI), xylylene diisocyanate (XDI), metaxylylene diisocyanate (MXDI), and the like.
- Aromatic diisocyanates Aliphatic diisocyanates such as hexamethylene diisocyanate (HDI); Cycloaliphatic diisocyanates such as isophorone diisocyanate (IPDI) and hydrogenated MDI; Compounds in a form in which these diisocyanate compounds are made non-volatile and less toxic; Polyisocyanates such as burettes, uretdiones, isocyanurates or allophanates of diisocyanate compounds; relatively low molecular weight urethane prepolymers; Mention may be made of the door compound.
- HDI hexamethylene diisocyanate
- IPDI isophorone diisocyanate
- MDI hydrogenated MDI
- Compounds in a form in which these diisocyanate compounds are made non-volatile and less toxic Compounds in a form in which these diisocyanate compounds are made non-volatile and less toxic
- Polyisocyanates such as burettes,
- the (B) polyisocyanate compound having water dispersibility those obtained by introducing a hydrophilic group into the polyisocyanate compound and those obtained by mixing and emulsifying a surfactant, so-called self-emulsification Can be mentioned.
- the hydrophilic group examples include an anionic group such as a carboxyl group and a sulfonic acid group, a cationic group such as a tertiary amino group, and a nonionic group such as a polyoxyalkylene group.
- the hydrophilic group is preferably a nonionic group.
- a specific nonionic group a highly hydrophilic polyoxyethylene group is preferred.
- the surfactant suitably used for the preparation of the self-emulsifying polyisocyanate compound obtained by mixing and emulsifying the polyisocyanate compound and the surfactant for example, an anionic interface having an anionic group such as a carboxyl group and a sulfonic acid group
- an anionic interface having an anionic group such as a carboxyl group and a sulfonic acid group
- examples thereof include a surfactant, a cationic surfactant having a cationic group such as a tertiary amino group, and a nonionic surfactant having a nonionic group such as a polyoxyalkylene group.
- a nonionic surfactant it is more preferable to use a nonionic surfactant in consideration of water resistance of the resulting coating film.
- a commercially available product may be used as the polyisocyanate compound (B) having water dispersibility.
- Commercially available products include AQUANATE 100, AQUANATE 110, AQUANATE 200, and AQUANATE 210 (manufactured by Tosoh Corporation), Bihydur TPLS-2032, SBU-isocyanate L801, Bihidul VPLS-2319, Bihjul 3100, VPLS-2336.
- polyisocyanate compound (B) it is more preferable to use a water-dispersible compound as the polyisocyanate compound (B).
- a polyisocyanate compound (B) 1 type may be used independently and 2 or more types may be used in combination.
- the content of the polyisocyanate compound (B) contained in the aqueous intermediate coating composition in the present invention is preferably 5 to 55% by mass with respect to the resin solid content of the aqueous intermediate coating composition. It is more preferable that the content be ⁇ 45% by mass.
- Hydrophilic modified carbodiimide compound (C) The hydrophilized modified carbodiimide compound (C) contained in the aqueous intermediate coating composition in the present invention has, in the molecule, -OCONH-X-NHCOOY [X is a bifunctional organic group containing at least one carbodiimide group, and Y has a structure in which a hydroxyl group is removed from a polyalkylene glycol monoalkyl ether. ]
- One or more structural units represented by having the above structural unit it is considered that both excellent water dispersibility and excellent curability are obtained.
- hydrophilized modified carbodiimide compound (C) there are three types, one having one structural unit, one having two structural units, and one having three structural units. As what has two said structural units, there exists what is represented with the following general formula (I).
- X is a bifunctional organic group containing at least one carbodiimide group
- Y is a structure obtained by removing a hydroxyl group from the same or different polyalkylene glycol monoalkyl ether
- Z is The structure is obtained by removing a hydroxyl group from a bifunctional polyol having a number average molecular weight of 200 to 5,000.
- R 2 is preferably a hydrocarbon group having 6 to 15 carbon atoms.
- Specific examples include a phenylene group, a diphenylenemethyl group, a diphenylene (dimethyl) methyl group, a methylphenylene group, a dimethylphenylene group, a tetramethylxylylene group, a hexylene group, a cyclohexylene group, and a dicyclohexylenemethyl group. be able to. Preference is given to a dicyclohexylenemethyl group.
- the p is 1 to 10.
- p is the number of carbodiimide groups present in the structural unit, preferably 2 or more from the viewpoint of curability, and more preferably 8 or less.
- the number of repetitions is not limited to p, and the number of repetitions is expressed as an average value.
- Y can be represented by the following general formula (b) or (c).
- R 3 is preferably an alkyl group having 1 to 20 carbon atoms. Specific examples include a methyl group, an ethyl group, a butyl group, a hexyl group, an octyl group, a decyl group, a dodecyl group, and a stearyl group.
- R 4 is a hydrogen atom or a methyl group, and is preferably a hydrogen atom. q is 4 to 40. In the general formulas (b) and (c), when R 4 is hydrogen, the general formulas (b) and (c) show the same structure.
- Z is a polymer structure composed of an ether bond, an ester bond, or a carbonate bond, and is difficult to formulate.
- the bifunctional polyol having a number average molecular weight of 200 to 5,000, which will be described later.
- the hydrophilized modified carbodiimide compound (C) having two structural units has a raw material carbodiimide compound containing at least two isocyanate groups in one molecule, a hydroxyl group at the molecular end, and a number average molecular weight of 200 to 5,000.
- a reaction product obtained by reacting the above bifunctional polyol with a molar ratio of the isocyanate group of the raw material carbodiimide compound exceeding the molar amount of the hydroxyl group of the polyol, and further reacting with a polyalkylene glycol monoalkyl ether Can be obtained.
- the raw material carbodiimide compound containing at least two isocyanate groups in the molecule preferably has isocyanate groups at both ends from the viewpoint of reactivity.
- the method for producing a raw material carbodiimide compound having an isocyanate group at both ends is well known by those skilled in the art, and for example, a condensation reaction involving decarbonization of an organic diisocyanate can be used.
- organic diisocyanate examples include aromatic diisocyanates, aliphatic diisocyanates, alicyclic diisocyanates, and mixtures thereof.
- aromatic diisocyanates 4,4- Diphenylmethane diisocyanate, 4,4-diphenyldimethylmethane diisocyanate, 1,3-phenylene diisocyanate, 1,4-phenylene diisocyanate, 2,4-tolylene diisocyanate, 2,6-tolylene diisocyanate, 2,4-tolylene diisocyanate
- isophorone diisocyanate dicyclohexylmeta 4,4-diisocyanate, and the like methylcyclohexane diisocyanate
- a carbodiimidization catalyst is usually used.
- the carbodiimidization catalyst include 1-phenyl-2-phospholene-1-oxide, 3-methyl-2-phospholene-1-oxide, 1-ethyl-2-phospholene-1-oxide, 3- Mention may be made of phospholene oxides such as methyl-1-phenyl-2-phospholene-1-oxide and their 3-phospholene isomers. From the viewpoint of reactivity, 3-methyl-1-phenyl-2-phospholene-1-oxide is preferred.
- the bifunctional polyol having a hydroxyl group at the molecular end is not particularly limited, but the number average molecular weight is preferably 200 to 5,000 from the viewpoint of reaction efficiency.
- Specific examples of the bifunctional polyol having a hydroxyl group at the molecular terminal include polyether diol, polyester diol, and polycarbonate diol, such as polyethylene glycol, polypropylene glycol, polyethylene propylene glycol, polytetramethylene ether glycol, Polyalkylene glycols such as polyhexamethylene ether glycol and polyoctamethylene ether glycol, polyethylene adipate, polybutylene adipate, polyhexamethylene adipate, polyneopentyl adipate, poly-3-methylpentyl adipate, polyethylene / butylene adipate, polyneopentyl / Polyester diol such as hexyl adipate, polycaprolactone diol, poly-3- Poly
- the reaction between the raw material carbodiimide compound containing at least two isocyanate groups in one molecule and a bifunctional polyol having a hydroxyl group at the molecular end and a number average molecular weight of 200 to 5,000 is the reaction of the raw material carbodiimide compound.
- the reaction is carried out at a ratio in which the molar amount of the isocyanate group exceeds the molar amount of the hydroxyl group of the polyol.
- the reaction of the polyalkylene glycol monoalkyl ether described later cannot be performed sufficiently.
- the ratio between the molar amount of the isocyanate group of the raw material carbodiimide compound and the molar amount of the hydroxyl group of the polyol having a hydroxyl group at the molecular end is 1.1: 1.0 to 2.0: from the viewpoint of reaction efficiency and economy. 1.0 is preferable.
- the degree of polymerization of the raw material carbodiimide compound and the bifunctional polyol having a hydroxyl group at the molecular terminal in the reaction product obtained in this step is preferably 1 to 10 from the viewpoint of reaction efficiency.
- reaction product thus obtained is further reacted with a polyalkylene glycol monoalkyl ether to obtain a hydrophilic modified carbodiimide compound (C) having two structural units.
- a polyalkylene glycol monoalkyl ether those represented by the following general formula (b ′) or (c ′) are used.
- R 3 , R 4 , and q the contents described in the general formulas (b) and (c) are applied as they are to R 3 , R 4 , and q.
- the type and q of R 4 in the above unit are appropriately set within the above ranges in consideration of storage stability, water dispersibility, and reactivity after water volatilizes.
- R 3 in the monoalkoxypolyalkylene glycol is preferably a methyl group and R 4 is preferably a hydrogen atom.
- q is preferably from 4 to 20, more preferably from 6 to 12, from the viewpoint of water dispersibility and reactivity after water volatilizes.
- polyalkylene glycol monoalkyl ether a polyalkylene glycol monoalkyl ether having a number average molecular weight of 200 to 5,000 is preferably used.
- the alkyl group of the polyalkylene glycol monoalkyl ether is preferably an alkyl group having 1 to 20 carbon atoms.
- Specific examples of the polyalkylene glycol monoalkyl ether include, for example, those composed of polyethylene glycol, polypropylene glycol or a mixture thereof having one end blocked with an alkyl group having 1 to 20 carbon atoms.
- polyalkylene glycol monoalkyl ethers include, for example, polyethylene glycol monomethyl ether, polyethylene glycol mono-2-ethylhexyl ether, polyethylene glycol monolauryl ether having a number average molecular weight of 200 to 5,000, Examples thereof include polypropylene glycol monomethyl ether, polypropylene glycol mono-2-ethylhexyl ether, and polypropylene glycol monolauryl ether.
- the reaction product and the polyalkylene glycol monoalkyl ether are reacted at a ratio in which the molar amount of the isocyanate group of the reaction product is the same or higher than the molar amount of the hydroxyl group of the polyalkylene glycol monoalkyl ether.
- the molar amount of the isocyanate group is less than the molar amount of the hydroxyl group, the reaction of the polyalkylene glycol monoalkyl ether with the reaction product cannot be sufficiently performed.
- the molar amount of the isocyanate group of the reaction product may be obtained by direct measurement, or may be a value calculated from the charged composition.
- a catalyst can be used in the reaction between the raw material carbodiimide compound and the bifunctional polyol having a hydroxyl group at the molecular end, and the reaction between the reaction product and the polyalkylene glycol monoalkyl ether.
- the temperature during the reaction is not particularly limited, but is preferably 60 to 120 ° C. from the viewpoint of control of the reaction system and reaction efficiency.
- hydrophilized modified carbodiimide compound (C) having two structural units By undergoing such a two-step reaction, a hydrophilized modified carbodiimide compound (C) having two structural units can be obtained.
- the hydrophilized modified carbodiimide compound (C) produced in this way does not have the structure of the general formula (I) shown above, but various other reaction products derived from the raw materials used. It is a mixture containing. However, in general, it may be regarded as having the structure of the general formula (I).
- hydrophilized modified carbodiimide compound (C) there are those represented by the following general formula (II) as those having three structural units.
- R 0 is hydrogen, methyl or ethyl.
- R 1 is an alkylene group having 4 or less carbon atoms, and may be the same or different. Specific examples of the alkylene group include a methylene group, an ethylene group, a propylene group, and a butylene group. n is 0 or 1, and m is 0-60.
- R 0 , R 1 , n, and m are determined by the trifunctional polyol used in producing the hydrophilized modified carbodiimide compound (C).
- the ratio of the hydrophilic part to the hydrophobic part is preferably 2.0 to 6.3.
- the ratio of the hydrophilic part to the hydrophobic part can be determined by dividing the molecular weight of the oxymethylene group or oxyethylene group part present in the carbodiimide compound by the molecular weight of the carbodiimide compound.
- the hydrophilized modified carbodiimide compound (C) having three structural units comprises a raw material carbodiimide compound containing at least two isocyanate groups in one molecule, a polyalkylene glycol monoalkyl ether, and an isocyanate group of the raw material carbodiimide compound.
- the description of the raw material carbodiimide compound of the hydrophilized modified carbodiimide compound (C) having two structural units is applied as it is.
- the reaction between the raw material carbodiimide compound and the polyalkylene glycol monoalkyl ether is further reacted with a trifunctional polyol after the reaction, so that an isocyanate group needs to remain.
- the equivalent of an isocyanate group needs to exceed the equivalent of a hydroxyl group, Preferably, it is preferable that the equivalent ratio of an isocyanate group and a hydroxyl group is 2/1.
- the reaction can usually be carried out under conditions well known to those skilled in the art, and a tin-based catalyst can be used if necessary.
- polyalkylene glycol monoalkyl ether As the polyalkylene glycol monoalkyl ether, the description of the polyalkylene glycol monoalkyl ether of the hydrophilically modified carbodiimide compound (C) having two structural units is applied as it is.
- the trifunctional polyol is reacted with the reaction product thus obtained.
- the amount of the trifunctional polyol used in the reaction is preferably such that the hydroxyl equivalent of the reactant is equal to or greater than the isocyanate equivalent, and more preferably the isocyanate equivalent and the hydroxyl equivalent are equal.
- the isocyanate equivalent of the said reaction product can also be calculated
- the reaction can be carried out in the same manner as the reaction between the raw material carbodiimide compound and the polyalkylene glycol monoalkyl ether.
- the trifunctional polyol is preferably trimethylolpropane, glycerin, or an alkylene oxide adduct thereof from the viewpoint of easy availability.
- the alkylene oxide include ethylene oxide and propylene oxide.
- the alkylene oxide adduct of glycerin is commercially available as a GP series from Sanyo Chemical.
- those having a structure in which an alkylene oxide is added to one hydroxyl group are particularly preferred.
- those having such a structure include GP-250 and GP-3000.
- hydrophilized modified carbodiimide compound (C) having three structural units can be obtained.
- the hydrophilically modified carbodiimide compound (C) thus produced does not have the structure of the general formula (II) alone, but has the structure of the general formula (II). Can be considered as being.
- hydrophilic modified carbodiimide compound (C) examples include those represented by the following general formula (III) having one structural unit.
- X is a bifunctional organic group containing at least one carbodiimide group
- Y has a structure in which a hydroxyl group is removed from the same or different polyalkylene glycol monoalkyl ether.
- X in the general formula (III) is a group that can be represented by the formula (a) in the above general formula (I).
- Y in the general formula (III) has a structure in which a hydroxyl group is removed from the same or different polyalkylene glycol monoalkyl ether. This Y can show the same thing as Y in the above-mentioned general formula (I).
- the hydrophilized modified carbodiimide compound (C) represented by the general formula (III) there is an advantage that the crosslinking density is maintained at a higher level.
- the general formula (I) (II) having a plurality of carbodiimide units has a low reaction efficiency with an acid while the acid value of the aqueous resin is low
- the general formula (III) is a general formula (I) Since it does not have a bulky structure as in (II), it does not hinder the crosslinking of the hydroxyl group and isocyanate of the aqueous resin, so that the hydrophilic modified carbodiimide compound represented by the general formula (III) ( It is considered that the crosslink density of C) has increased.
- Y in the general formula (III) is preferably the following (i) or (ii):
- (ii) a structure having 4 to 60 repeats A structure in which a hydroxyl group is removed from a polypropylene glycol monoalkyl ether in which an alkyl group having 1 to 8 carbon atoms is ether-bonded to the end of a polypropylene oxide unit: More preferably, they are the same or different structures selected from More preferably, the number of repeating (ii) polypropylene oxide units is 15 to 60.
- the hydrophilized modified carbodiimide compound (C) represented by the general formula (III) having the above (i) and (ii) By using the hydrophilized modified carbodiimide compound (C) represented by the general formula (III) having the above (i) and (ii), the water dispersibility is excellent, the stability is improved, and the crosslinking density is higher. There is an advantage of being held at the level.
- the hydrophilized modified carbodiimide compound (C) represented by the general formula (III) is a polyalkylene glycol monoalkyl ether which is the same or different from the raw material carbodiimide compound obtained by the above-described condensation reaction involving decarbonization of the organic diisocyanate. Can be prepared by reacting.
- the polyalkylene glycol monoalkyl ether is A polyethylene glycol monoalkyl ether in which an alkyl group having 1 to 3 carbon atoms is ether-bonded to the terminal of a polyethylene oxide unit having 6 to 20 repeats, or A polypropylene glycol monoalkyl ether in which an alkyl group having 1 to 8 carbon atoms is ether-bonded to the end of a polypropylene oxide unit having 4 to 60 repeats, It is more preferable that In preparing the hydrophilized modified carbodiimide compound (C) represented by the general formula (III), these polyethylene glycol monoalkyl ethers and polypropylene glycol monoalkyl ethers may be used alone or in combination.
- polyethylene glycol monoalkyl ether examples include polyethylene glycol monomethyl ether, polyethylene glycol monoethyl ether, and polyethylene glycol monopropyl ether, with polyethylene glycol monomethyl ether being particularly preferred.
- polypropylene glycol monoalkyl ether examples include polypropylene glycol monomethyl ether, polypropylene glycol monoethyl ether, polypropylene glycol monobutyl ether, polypropylene glycol-2-ethylhexyl ether, and polypropylene glycol monobutyl ether is particularly preferable. It is.
- any one Y is (i), the other Y is (ii), and (i) the number of repetitions 6
- the periphery of the carbodiimide group is somewhat hydrophobic in order to improve water resistance when a coating film is formed.
- the periphery of the carbodiimide group is hydrophobic to some extent and the contact with water molecules is kept low.
- the carbodiimide compound represented by the general formula (III) needs to have a certain amount of polyethylene glycol structure in order to maintain hydrophilicity.
- the hydrophilicity of the carbodiimide compound is secured.
- hydrophobicity can be maintained to some extent around the carbodiimide group.
- the content of the hydrophilized modified carbodiimide compound (C) contained in the aqueous intermediate coating composition is preferably 1 to 8% by mass relative to the resin solid content of the aqueous intermediate coating composition.
- the aqueous intermediate coating composition in the present invention such as the preparation of an aqueous intermediate coating composition, comprises an aqueous resin (A1) having a hydroxyl group and a carboxyl group, a polyisocyanate compound (B), and a hydrophilized modified carbodiimide compound (C). Including.
- the ratio of the equivalent of the carbodiimide group of the hydrophilized modified carbodiimide compound (C) to the equivalent of the isocyanate group of the polyisocyanate compound (B) is in the range of 0.01 to 0.20. Is preferred.
- the present invention is characterized in that the equivalent of the carbodiimide group is very small relative to the equivalent of the isocyanate group.
- the coating ratio obtained while ensuring low-temperature curability is obtained when the ratio of the equivalent of the carbodiimide group to the equivalent of the isocyanate group is in the range of 0.01 to 0.20.
- the equivalent ratio is more preferably in the range of 0.01 to 0.09.
- the aqueous resin (A1) having a hydroxyl group and a carboxyl group used in the present invention has a hydroxyl value of 80 to 200 mgKOH and an acid value of 10 to 40 mgKOH / g in terms of resin solid content. That is, the value of the hydroxyl value is very large compared to the value of the acid value.
- the ratio of the equivalent of the carbodiimide group of the hydrophilized modified carbodiimide compound (C) to the equivalent of the isocyanate group of the polyisocyanate compound (B) is 0.01 to By being in the range of 0.20, there is an advantage that a coating film having a sufficient crosslinking density can be obtained while ensuring the coating stability.
- the ratio of the equivalent of the carbodiimide group of the hydrophilized modified carbodiimide compound (C) to the equivalent of the isocyanate group of the polyisocyanate compound (B) is simply reduced without using the aqueous resin (A1) as described above There is a possibility that the stability of the paint is greatly reduced.
- an aqueous resin (A1) having a very high hydroxyl value relative to the acid value is used.
- the hydroxyl value of the aqueous resin (A1) is so high, a high crosslink density is achieved in the resulting coating film.
- the acid value of the aqueous resin (A1) is low, suppression of undesirable side reactions that can occur between the acid group of the aqueous resin (A1) and the isocyanate group of the polyisocyanate compound (B) is achieved.
- the ratio of the equivalent of the carbodiimide group of the hydrophilized modified carbodiimide compound (C) to the equivalent of the isocyanate group of the polyisocyanate compound (B) is in the range of 0.01 to 0.20, and the amount of the carbodiimide group is extremely small. As a result, there is an advantage that a sufficient crosslink density is achieved even after storage of the coating composition.
- the ratio of the equivalent of the isocyanate group of the polyisocyanate compound (B) to the equivalent of the hydroxyl group contained in the aqueous resin (A1) is in the range of 0.6 to 1.5.
- the hydroxyl group of the aqueous resin (A1) and the isocyanate group of the polyisocyanate compound (B) are groups that react with each other. And when the equivalent ratio of these groups is in the above range, there is an advantage that the curing reaction proceeds well even at a low temperature, whereby a coating film having a desired crosslinking density can be obtained.
- the ratio of the equivalent of the carbodiimide group of the hydrophilized modified carbodiimide compound (C) to the equivalent of the acid group of the aqueous resin (A1) is 0.1 to 1.0. It is preferable that it is the range of these. This equivalent ratio is more preferably in the range of 0.1 to 0.6. In this case, an excess amount of the acid group is present in an equivalent amount of the carbodiimide group and the acid group that react with each other. Thereby, while the carbodiimide group basically does not remain in the cured coating film to be formed, the acid group remains and there is an advantage that the adhesion of the coating film to the object is improved. .
- the above-mentioned aqueous intermediate coating composition may contain, if necessary, a pigment, a curing catalyst, a surface conditioner, an antifoaming agent, a pigment dispersant, a plasticizer, and a film-forming aid. , UV absorbers, antioxidants, solvents (water, organic solvents) and the like. Since the said water-based intermediate coating composition is excellent in the reactivity at low temperature, it is preferable to manufacture at the coating site.
- the aqueous intermediate coating composition can be obtained by mixing the components (A1) to (C).
- the curing agent composition is used even when the water dispersibility of the polyisocyanate compound (B) is not sufficient. By forming the product, the storage stability of the aqueous intermediate coating composition can be enhanced.
- the resin solid content concentration of the above-mentioned aqueous intermediate coating composition varies depending on the coating conditions, but is generally preferably set to 15 to 60% by mass.
- the aqueous base paint composition used in the method of the present invention, an aqueous resin having a hydroxyl group and a carboxyl group (A2), the water-dispersible blocked polyisocyanate compound (D), hydrophilic modified carbodiimide compound (C) And an aqueous polyurethane resin (E).
- Aqueous resin having hydroxyl group and carboxyl group (A2)
- the aqueous resin (A2) contained in the aqueous base coating composition is the same resin as the aqueous resin (A1) contained in the aqueous intermediate coating composition, but the acid value range in the aqueous resin (A1) It is a resin with no regulations. That is, the aqueous resin (A2) contained in the aqueous base coating composition is a resin having a hydroxyl value of 80 to 200 mgKOH / g in terms of resin solid content.
- the aqueous resin (A2) contained in the aqueous base coating composition preferably has an acid value of 10 to 40 mg KOH / g.
- the aqueous resin (A2) contained in the aqueous base coating composition has a hydroxyl value of 80 to 200 mgKOH / g in terms of resin solids, so that the coating stability of the aqueous base coating composition containing the above components can be improved. While ensuring favorable, there exists an advantage that the crosslinking density of the cured coating film obtained becomes a favorable range, and performances such as water resistance are improved. This is because the hydroxyl value of the aqueous resin (A2) is relatively high as in the above range, and in the aqueous base coating composition, a water-dispersible blocked polyisocyanate compound (D), a hydrophilized modified carbodiimide compound (C). Further, it is considered that the low-temperature curability of the coating composition is ensured by further including the water-based polyurethane resin (E), and thereby the crosslinking density in the obtained coating film is in a favorable range.
- the aqueous resin (A2) is preferably contained, for example, within a range of 20 to 60% by mass with respect to the resin solid content of the aqueous base coating composition.
- the above range is more preferably 25 to 50% by mass.
- Hydrophilic modified carbodiimide compound (C) The hydrophilized modified carbodiimide compound (C) contained in the aqueous base coating composition is the hydrophilic modified carbodiimide compound (C) contained in the aqueous intermediate coating composition.
- the hydrophilic modified carbodiimide compound (C) contained in the aqueous base coating composition and the hydrophilic modified carbodiimide compound (C) contained in the aqueous intermediate coating composition are completely the same. Resin may be sufficient and it may be resin which is mutually different within the definition range of the said hydrophilization modified carbodiimide compound (C).
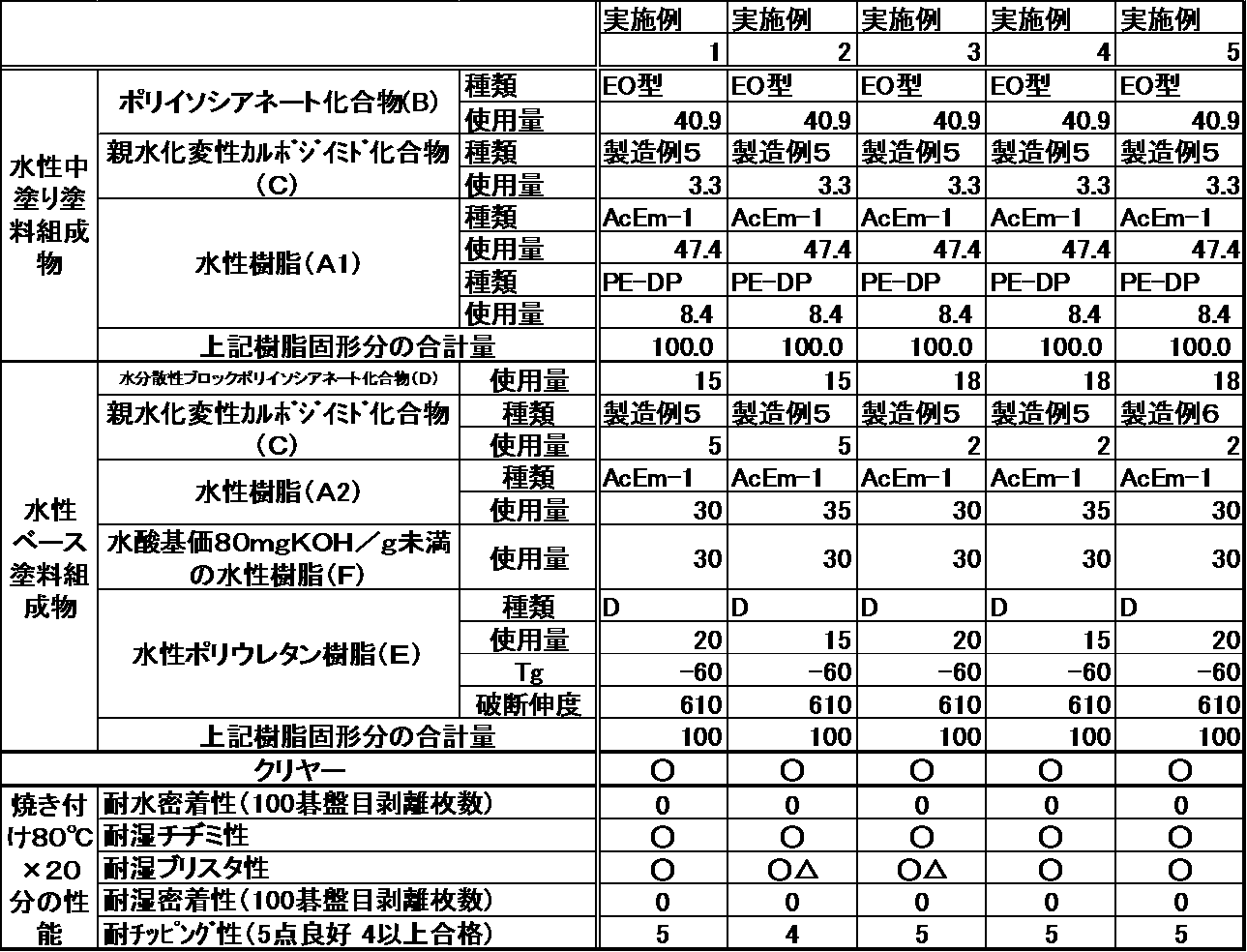
- the content of the hydrophilized modified carbodiimide compound (C) contained in the aqueous base coating composition is preferably 1 to 9% by mass, preferably 1.5 to 7%, based on the resin solid content of the aqueous base coating composition. More preferably, it is mass%.
- the amount of the hydrophilized modified carbodiimide compound (C) contained in the aqueous base coating composition is in the above range, the resulting multilayer coating film has good water resistance and good coating appearance after a moisture resistance test. There are benefits to be gained.
- Water dispersible block polyisocyanate compound (D) The water-dispersible blocked polyisocyanate compound (D) contained in the aqueous base coating composition in the present invention is obtained by reacting the malonic acid diester (D-2) with the polyisocyanate (D-1), and then the resulting reaction product. And an organic amine compound (D-3).
- the polyisocyanate (D-1) is one or more selected from aliphatic polyisocyanates, alicyclic polyisocyanates and aromatic polyisocyanates.
- aliphatic polyisocyanate examples include aliphatic diisocyanate, lysine triisocyanate (hereinafter referred to as LTI), 4-isocyanatomethyl-1,8-octamethylene diisocyanate (trimer triisocyanate: hereinafter referred to as TTI), bis (2 -Isocyanatoethyl) 2-isocyanatoglutarate (glutamate triisocyanate: hereinafter referred to as GTI).
- LTI lysine triisocyanate
- TTI 4-isocyanatomethyl-1,8-octamethylene diisocyanate
- TTI 4-isocyanatomethyl-1,8-octamethylene diisocyanate
- GTI bis (2 -Isocyanatoethyl) 2-isocyanatoglutarate
- aliphatic diisocyanate used in the aliphatic polyisocyanate those having 4 to 30 carbon atoms are preferable.
- HDI is preferred because of its industrial availability.
- the aliphatic diisocyanate only one kind may be used, or two or more kinds may be used in combination.
- the alicyclic polyisocyanate the following alicyclic diisocyanates are mainly used.
- the alicyclic diisocyanate those having 8 to 30 carbon atoms are preferable.
- isophorone diisocyanate hereinafter referred to as IPDI
- 1,3-bis (isocyanatomethyl) -cyclohexane 4,4′-dicyclohexylmethane diisocyanate.
- Norbornene diisocyanate Norbornene diisocyanate
- hydrogenated xylylene diisocyanate and the like.
- IPDI is preferred from the viewpoint of weather resistance and industrial availability. Only 1 type may be used for alicyclic diisocyanate and it may use 2 or more types together.
- aromatic polyisocyanate aromatic diisocyanates shown below are mainly used.
- aromatic diisocyanate examples include 2,4-tolylene diisocyanate, 2,6-tolylene diisocyanate, 4,4'-diphenylmethane diisocyanate, naphthalene diisocyanate, xylylene diisocyanate, and the like.
- Aromatic diisocyanate may use only 1 type and may use 2 or more types together.
- aliphatic polyisocyanates and / or alicyclic polyisocyanates are preferable because of excellent weather resistance. Furthermore, among the aliphatic polyisocyanates, aliphatic diisocyanates are most preferable.
- polyisocyanates examples include triisocyanates such as LTI, TTI, GTI, or derivatives thereof, biuret bonds, urea bonds, isocyanurate bonds, uretdione bonds, urethane bonds, allophanate bonds, oxadiazine trione bonds, etc. And di-isocyanate di- to 20-mer oligomers produced by forming
- the malonic acid diester (D-2) to be reacted with the polyisocyanate (D-1) is a compound represented by the following general formula.
- W 1 and W 2 are each independently an alkyl group having 1 to 8 carbon atoms, a phenyl group or a benzyl group.
- the phenyl group or benzyl group may have 1 to 3 substituents. Examples of the substituent include an alkyl group having 1 to 6 carbon atoms, a halogen atom such as a fluorine atom or a chlorine atom, an amino group, a sulfonyl group, and a hydroxyl group.
- W 1 and W 2 may be the same or different.
- malonic acid diester examples include dimethyl malonate, diethyl malonate, di-n-propyl malonate, diisopropyl malonate, di-n-butyl malonate, diisobutyl malonate, and di-t-butyl malonate.
- it is dimethyl malonate, diethyl malonate, di-n-propyl malonate, diisopropyl malonate, di-n-butyl malonate, diisobutyl malonate, di-t-butyl malonate, methyl t-butyl malonate. More preferred are dimethyl malonate and diethyl malonate, and most preferred is diethyl malonate.
- the malonic acid diester shown above may be used alone or in combination of two or more.
- the amount of malonic acid diester (D-2) used in the reaction is such that malonic acid diester (D-2) is 75 to 150 moles per mole of isocyanate group of polyisocyanate (D-1). preferable.
- This reaction may be performed in the presence of an organic solvent or may be performed without using an organic solvent. When using an organic solvent, it is preferable to use an organic solvent that is inert to the isocyanate group and hardly hydrolyzes.
- Preferable organic solvents include, for example, ether solvents such as propylene glycol dimethyl ether, dipropylene glycol dimethyl ether, diethylene glycol dimethyl ether, and diethylene glycol diethyl ether, and ketone solvents such as methyl ethyl ketone and methyl isobutyl ketone.
- ether solvents such as propylene glycol dimethyl ether, dipropylene glycol dimethyl ether, diethylene glycol dimethyl ether, and diethylene glycol diethyl ether
- ketone solvents such as methyl ethyl ketone and methyl isobutyl ketone.
- a reaction catalyst can be used as necessary.
- the reaction catalyst include organic metal salts such as tin, zinc and lead, metal alcoholates, and tertiary amines.
- the above reaction can be generally carried out at ⁇ 20 to 150 ° C., preferably 0 to 100 ° C., more preferably 40 to 80 ° C. By performing the reaction at 150 ° C. or lower, side reactions can be suppressed, and by performing the reaction at ⁇ 20 ° C. or higher, the reaction rate can be kept high.
- Nonionic hydrophilic compounds include polyethylene glycol compounds having at least three consecutive ethylene oxide groups.
- Polyethylene glycol compounds such as monoalkoxy polyethylene glycol, polyethylene glycol or triol, pluronic type polypropylene glycol or triol obtained by addition polymerization of ethylene oxide at the end of polypropylene glycol, polyoxypropylene polyoxyethylene copolymer diol or triol, polyoxypropylene Examples thereof include polyoxyethylene block polymer diol and triol.
- monoalkoxy polyethylene glycol and polyethylene glycol are preferable, and monoalkoxy polyethylene glycol is more preferable.
- Monoalkoxy polyethylene glycol is obtained by adding an alcohol to one end of polyethylene glycol.
- the monoalcohol that can be used in the monoalkoxy polyethylene glycol preferably has 1 to 8 carbon atoms, more preferably 1 to 6 carbon atoms, and still more preferably 1 to 4 carbon atoms. Most preferred are methanol and ethanol. That is, among monoalkoxy polyethylene glycols, monomethoxy polyethylene glycol and monoethoxy polyethylene glycol are preferable, and monomethoxy polyethylene glycol is most preferable.
- polyethylene glycol compounds polyethylene glycol compounds in which a monoalcohol having 1 to 4 carbon atoms is added to one end having a number average molecular weight of 200 to 2000 are particularly preferable.
- the reaction product thus obtained is reacted with the organic amine compound (D-3) to prepare a water-dispersible block polyisocyanate compound (D).
- the organic amine compound (D-3) include a chain secondary amine compound and a cyclic secondary amine compound containing a nitrogen atom.
- the chain secondary amine compound dialkylamines having two identical or different alkyl groups having 1 to 12 carbon atoms are preferable.
- Preferred examples of the chain secondary amine compound include diisopropylamine, diisobutylamine, di (2-butylamine), di (t-butyl) amine, dicyclohexylamine, Nt-butylcyclohexylamine and the like.
- Cyclic secondary amine compounds containing a nitrogen atom include aziridine, azetidine, pyrrolidine, 2-methylpyrrolidine, piperidine, 2-methylpiperidine, 3-methylpiperidine, 4-methylpiperidine, 4-benzylpiperidine, 2,4-dimethylpiperidine 3,5-dimethylpiperidine, 2,6-dimethylpiperidine, 2,2,6,6-tetramethylpiperidine, 4-piperidinecarboxylic acid methyl ester, 4-piperidinecarboxylic acid ethyl ester, 2,2,6,6 -Tetramethyl-4-piperidone, 4-piperidinopiperidine, piperazine, N-methylpiperazine, N-ethylpiperazine, N-allylpiperazine, N-isobutylpiperazine, N-cyclohexylpiperazine, N-cyclopentylpiperazine, N-phenyl Piperazine, - (2-pyridyl) piperazine, 1- (4-pyrid
- the above reaction may be performed using an organic solvent or may be performed without using an organic solvent.
- an organic solvent it is preferable to use an organic solvent that is inert to the isocyanate group and hardly hydrolyzes.
- organic solvents include, for example, ether solvents such as propylene glycol dimethyl ether, dipropylene glycol dimethyl ether, diethylene glycol dimethyl ether, and diethylene glycol diethyl ether, and ketone solvents such as methyl ethyl ketone and methyl isobutyl ketone.
- the water-dispersible blocked polyisocyanate compound (D) thus obtained can improve storage stability by mixing 10 mol% or more of the basic compound with respect to 1 mol of the blocked isocyanate group of the compound.
- the basic compound include N-allylmorpholine, N-methylmorpholine, N-ethylmorpholine, triethanolamine, 2-methylimidazole and the like.
- a commercially available product may be used as the water-dispersible block polyisocyanate compound (D).
- Examples of commercially available products include WM44-L70G, which is a block polyisocyanate manufactured by Asahi Kasei Corporation.
- the water-dispersible blocked polyisocyanate compound (D) is maintained in reactivity even under heating conditions of 100 ° C. or lower, and further after being stored in a state contained in an aqueous base coating composition. Also has a feature such as high curability retention.
- the content of the water-dispersible block polyisocyanate compound (D) contained in the aqueous base coating composition in the present invention is preferably 10 to 25% by mass with respect to the resin solid content of the aqueous base coating composition. 15 to 22% by mass is more preferable.
- the curing reaction proceeds well even under heating conditions under low temperature conditions (for example, heating conditions of 100 ° C. or less), and a cured coating film having excellent coating film properties is obtained. There are advantages such as being.
- Aqueous polyurethane resin (E) The aqueous base coating composition contains an aqueous polyurethane resin (E) in addition to the above components.
- the aqueous intermediate coating composition and the aqueous base coating composition are applied, and then the aqueous coating composition is subjected to low temperature curing conditions.
- the water-based polyurethane resin can form a tough coating by fusing with itself and other components, so it has excellent adhesion between coatings and water-resistant adhesion. A multilayered multilayer is obtained.
- the aqueous polyurethane resin (E) comprises a polyol compound (E-1), a compound (E-2) having an active hydrogen group and a hydrophilic group in the molecule, an organic polyisocyanate (E-3), and a chain if necessary.
- the polyol compound (E-1) is not particularly limited as long as it is a polyol compound having two or more hydroxyl groups.
- the polyol compound (E-1) is, for example, a polyhydric alcohol such as ethylene glycol, propylene glycol, 1,4-butanediol, 1,6-hexanediol, trimethylolpropane, glycerin, polyethylene glycol, polypropylene glycol, polytetra Polyether polyols such as methylene ether glycol; dicarboxylic acids such as adipic acid, sebacic acid, itaconic acid, maleic anhydride, phthalic acid, isophthalic acid, and ethylene glycol, triethylene glycol, propylene glycol, butylene glycol, tripropylene glycol, Polyester polyols obtained from glycols such as neopentyl glycol; polycaprolactone polyols; polybutadiene polyols; polycarbonate
- the compound (E-2) having an active hydrogen group and a hydrophilic group in the molecule includes an active hydrogen and an anion group ⁇ anion group or anion-forming group (which reacts with a base to form an anion group. Is converted into an anionic group by neutralization with a base before, during or after the urethanization reaction) ⁇ (for example, Japanese Patent Publication No. 42-24192 and Japanese Patent Publication No.
- the organic polyisocyanate (E-3) is not particularly limited as long as it has two or more isocyanate groups in the molecule.
- Aliphatic diisocyanates having 2 to 12 carbon atoms such as hexamethylene diisocyanate, 2,2,4-trimethylhexane diisocyanate, lysine diisocyanate;
- Alicyclic diisocyanates having 4 to 18 carbon atoms such as 1,4-cyclohexane diisocyanate, isophorone diisocyanate, 4,4′-dicyclohexylmethane diisocyanate, methylcyclohexylene diisocyanate, isopropylidenecyclohexyl-4,4′-diisocyanate; 2,4-toluylene diisocyanate, 2,6-toluylene diisocyanate, diphenylmethane-4,4′-diisocyanate,
- these polyisocyanate compounds may be used as dimers and trimers (isocyanurate bonds), or may be used as biurets by reacting with amines.
- polyisocyanates having urethane bonds obtained by reacting these polyisocyanate compounds with polyols can also be used. It is more preferable to use an aliphatic diisocyanate as the organic polyisocyanate (E-3).
- the chain extender that can be used as necessary when preparing the aqueous polyurethane resin (E) is not particularly limited as long as it contains two or more active hydrogen groups.
- a low molecule less than the number average molecular weight of 500
- examples include polyols and polyamines.
- the low molecular polyol include ethylene glycol, propylene glycol, 1,4-butanediol, 3-methylpentanediol, 2-ethyl-1,3-hexanediol, and trimethylolpropane.
- the polyamine include ethylenediamine, hexamethylenediamine, diethylenetriamine, hydrazine, xylylenediamine, and isophoronediamine.
- examples of the polymerization terminator include a compound having one active hydrogen in the molecule, or a monoisocyanate compound.
- Examples of the compound having one active hydrogen in the molecule include monoalcohol (eg, alkyl alcohols such as methanol, butanol and octanol, alkyl alcohol alkylene oxide adducts), or monoamines (eg, butylamine, dibutylamine, etc.) Of the alkylamine).
- monoalcohol eg, alkyl alcohols such as methanol, butanol and octanol, alkyl alcohol alkylene oxide adducts
- monoamines eg, butylamine, dibutylamine, etc.
- Examples of the monoisocyanate compound include methyl isocyanate, ethyl isocyanate, propyl isocyanate, butyl isocyanate, lauryl isocyanate, cyclohexyl isocyanate, phenyl isocyanate, and tolylene isocyanate.
- the reaction method for producing the aqueous polyurethane resin (E) is a one-shot method in which each component is reacted at once or a multistage method in which the components are reacted stepwise ⁇ part of an active hydrogen-containing compound (for example, a polymer polyol) and Any method of producing an NCO-terminated prepolymer by reacting isocyanate and then reacting the remainder of the active hydrogen-containing compound may be used.
- the synthesis reaction of the aqueous polyurethane resin (E) is usually carried out at 40 to 140 ° C, preferably 60 to 120 ° C.
- a tin-based catalyst such as dibutyltin laurate or tin octylate used in a normal urethanization reaction or an amine-based catalyst such as triethylenediamine may be used.
- the above reaction may be performed in an organic solvent inert to isocyanate (for example, acetone, toluene, dimethylformamide, etc.), and a solvent may be added during or after the reaction.
- the aqueous polyurethane resin (E) in the present invention is a known method (in the case of an anion-forming group, a method of neutralizing with a base to form an anion group, and in the case of a cation-forming group, a quaternizing agent is used to form a cation group. It can be prepared by dispersing in water after treatment with a method of forming or a method of forming a cationic group by neutralization with an acid.
- the step of dissolving in water is not particularly limited, and may be after the reaction or in the middle of the multistage method.
- the aqueous polyurethane resin (E) can be obtained by dissolving in water while extending the chain with water and / or polyamine.
- the solvent may be removed after dissolving in water.
- the aqueous polyurethane resin (E) in the present invention has a glass transition point (Tg) of ⁇ 50 ° C. or less, and the breaking elongation of the cured film of the aqueous polyurethane resin (E) is 400% or more at ⁇ 20 ° C. Condition.
- the resulting multilayer coating film is inferior in coating film adhesion, chipping resistance and water resistance.
- the glass transition point (Tg) is more preferably ⁇ 55 ° C. or less, and further preferably ⁇ 58 ° C. or less.
- the glass transition point (Tg) of the aqueous polyurethane resin (E) can be measured by a differential scanning calorimeter.
- the resulting multilayer coating film has poor coating adhesion, chipping resistance and water resistance. Become.
- the breaking elongation is more preferably 500% or more.
- the breaking elongation of the cured film of the aqueous polyurethane resin (E) can be determined according to JIS K7127. Specifically, 95 parts by mass (resin solid content) of the aqueous polyurethane resin (E) and 5 parts by mass (resin solid content) of the hydrophilized modified carbodiimide compound (C) are mixed. The obtained mixture is uniformly coated with a doctor blade so that the dry film thickness becomes 20 ⁇ m. After standing at 20 ° C. for 10 minutes, preheating is performed at 80 ° C. for 3 minutes to volatilize moisture, and baking is performed at 120 ° C. for 30 minutes to prepare a cured film.
- the obtained cured film is subjected to a tensile performance test under a test temperature of ⁇ 20 ° C. according to JIS K7127, the elongation at break is measured, and the obtained elongation is defined as the elongation at break.
- the aqueous polyurethane resin and the carbodiimide compound represented by the general formula (I), (II) or (III) are mixed and baked to form a film. Cross-linking and / or fusion of the resin proceeds, and the breaking elongation of the aqueous polyurethane resin can be evaluated.
- aqueous polyurethane resin E
- Commercially available products include, for example, the NeoRez series, which is an aqueous polyurethane resin sold by Enomoto Kasei Co., Ltd., the HUX series, which is an aqueous polyurethane resin sold by ADEKA, and the U-Coat series, which is an aqueous polyurethane resin sold by Sanyo Kasei. , Permarin series, Upren series and so on.
- the content of the aqueous polyurethane resin (E) is preferably 8% by mass or more, more preferably 10% by mass or more, and more preferably 15% by mass or more with respect to the resin solid content of the aqueous base coating composition. More preferably.
- the upper limit of the content is preferably 50% by mass or less, and more preferably 30% by mass or less.
- the aqueous base coating composition may contain a resin component (other resin) other than the aqueous resin (A2), if necessary.
- a resin component (other resin) other than the aqueous resin (A2) for example, a resin prepared in the same manner as the aqueous resin (A2) and having a hydroxyl value of less than 80 mgKOH / g can be given.
- Other examples of other resins include resins having a hydroxyl group, such as polyether diol and polycarbonate diol.
- Other examples of other resins include, for example, resins having a hydroxyl group, such as polyether diol and polycarbonate diol, and melamine resins having a methylol group.
- Such other resins can be used in any amount provided that the functions (water resistance, chipping resistance, etc.) of the water-based base coating composition are not impaired.
- the resin having a hydroxyl value of less than 80 mgKOH / g is preferably contained, for example, in the range of 15 to 45% by mass with respect to the resin solid content of the aqueous base coating composition.
- the aqueous base coating composition can be prepared by mixing the components constituting the coating composition by commonly used means.
- the above-mentioned aqueous base coating composition may contain pigments, surface conditioners (antifoaming agents, leveling agents, etc.), pigment dispersants, plasticizers, film-forming aids, ultraviolet absorbers, antioxidants, flame retardants as necessary.
- Antistatic agents, electrostatic assistants, heat stabilizers, light stabilizers, solvents (water, organic solvents) and other additives may be contained.
- the pigment content in the case where the water-based base coating composition contains a pigment may be a range that is usually set according to the application.
- the total solid content of the resin and the curing agent in the water-based base coating composition and the mass% of the pigment (PWC: Pigment Weight Concentration) with respect to the total 100 mass parts of the pigment may be 0.1 to 50 mass%. preferable.
- Multi-layer coating film forming method The multi-layer coating film forming method of the present invention, An intermediate coating film forming step of forming an uncured intermediate coating film by coating a water-based intermediate coating composition on an object to be coated; An aqueous base coating composition is applied on the obtained uncured intermediate coating film to form an uncured base coating film, and the obtained uncured intermediate coating A curing process for heating and curing the film and the base coating film, Is included.
- a coating film having excellent coating film properties can be obtained even under low-temperature curing conditions by using the specific aqueous intermediate coating composition and aqueous base coating composition. be able to.
- the heat curing temperature of the coating film in the curing step is not particularly limited, and is preferably 70 to 120 ° C, more preferably 70 to 110 ° C, and further preferably 70 to 100 ° C.
- it can also carry out on low temperature curing conditions of 100 degrees C or less.
- the heat curing temperature of the coating film may further be a curing condition such as 70 to 90 ° C.
- steel sheets such as iron, steel, stainless steel, aluminum, copper, zinc, tin and alloys thereof; polyethylene resins, EVA resins, polyolefin resins (polyethylene resins, polypropylene resins, etc.) ), Vinyl chloride resin, styrene resin, polyester resin (including PET resin, PBT resin, etc.), polycarbonate resin, acrylic resin, acrylonitrile butadiene styrene (ABS) resin, acrylonitrile styrene (AS) resin, polyamide resin, acetal resin, phenol Resin, fluororesin, melamine resin, urethane resin, epoxy resin, polyphenylene oxide (PPO), and the like; and organic-inorganic hybrid materials. These may be in a molded state.
- the steel sheet may be in a state where an electrodeposition coating film is formed after chemical conversion treatment is performed.
- the chemical conversion treatment include zinc phosphate chemical conversion treatment, zirconium chemical conversion treatment, and chromium oxidation chemical conversion treatment.
- the electrodeposition coating film include an electrodeposition coating film obtained by electrodeposition coating using a cationic electrodeposition coating composition or an anion electrodeposition coating composition.
- the above resin may be subjected to steam cleaning using an organic solvent as necessary, or may be cleaned using a neutral detergent. Furthermore, primer coating may be applied as necessary.
- the multilayer coating film forming method of the present invention is characterized in that a coating film having excellent coating film properties can be formed even under low temperature curing conditions. Therefore, as an object to which the method of the present invention can be preferably used, an object to be coated including a steel plate part and a resin part is exemplified. For such an object to be coated, by forming a multilayer coating film by the multilayer coating film forming method of the present invention, the resin portion and It becomes possible to form a multilayer coating film having good physical properties for both of the steel plate portions. By the multilayer coating film forming method of the present invention, different materials such as a resin and a steel plate can be applied using a common coating composition. Thereby, there is an advantage that the hue of the obtained coating film can be matched at a higher level.
- Examples of other objects that are suitable as objects to be coated in the method for forming a multilayer coating film of the present invention include industrial machines and construction machines.
- Industrial machines and construction machines are generally large in size and can withstand strong loads, and therefore have a feature that the constituent base material (steel plate) is thicker than automobile bodies and the like. Therefore, when such an industrial machine or construction machine is an object to be coated, there is a problem that the heat capacity of the object to be coated is large and heat is not sufficiently transferred to the object to be coated in a heating furnace.
- the multi-layer coating film forming method of the present invention is characterized in that it can be cured at a low temperature and that a coating film having a high crosslinking density can be obtained even when cured at a low temperature. Therefore, the multi-layer coating film forming method of the present invention also has a large heat capacity of the object to be coated, and it is difficult to perform high-temperature heat-curing treatment after coating. Can be preferably used.
- the above-mentioned aqueous intermediate coating composition and aqueous base coating composition can be applied by a commonly used coating method.
- a commonly used coating method For example, when the above-mentioned aqueous intermediate coating composition and aqueous base coating composition are applied to an automobile body, in order to enhance the appearance of the obtained coating film, multistage coating by air electrostatic spray coating, preferably two stages, is performed. Paint or combine air electrostatic spray painting with a rotary atomizing electrostatic coating machine called “ ⁇ (micro micro) bell”, “ ⁇ (micro) bell” or “metabell” Coating methods can be used.
- the film thickness of the coating film of the water-based intermediate coating composition can be appropriately selected according to the desired application.
- the film thickness is preferably, for example, 8 to 40 ⁇ m as a dry film thickness, and more preferably 15 to 30 ⁇ m.
- the film thickness of the coating film of the aqueous base coating composition can be appropriately selected according to the desired use.
- the film thickness is preferably, for example, 10 to 30 ⁇ m as a dry film thickness.
- a clear coating composition is further applied to form a clear coating film, and then the uncured multilayer coating film is cured. Including.
- This method can omit the baking and drying furnace, and is preferable from the viewpoint of economy and environmental protection.
- a urethane clear coating composition As an example of a clear coating composition that can be suitably used in the above coating process, a urethane clear coating composition can be mentioned.
- the urethane clear coating composition include a clear coating composition containing a hydroxyl group-containing resin and an isocyanate compound curing agent.
- the isocyanate compound as the curing agent is not particularly limited, and examples thereof include aliphatic isocyanates such as trimethylene diisocyanate, tetramethylene diisocyanate, pentamethylene diisocyanate, hexamethylene diisocyanate (HDI), and trimethylhexamethylene diisocyanate, 1,3-cyclohexane.
- Aliphatic cyclic isocyanates such as pentane diisocyanate, 1,4-cyclohexane diisocyanate, 1,2-cyclohexane diisocyanate, xylylene diisocyanate (XDI), 2,4-tolylene diisocyanate (TDI), 2,6-tolylene diisocyanate, etc.
- Aromatic isocyanates, isophorone diisocyanate (IPDI), norbornane diisocyanate methyl and other alicyclic isocyanates These biuret, etc. multimers and mixtures such as isocyanurate body can be exemplified.
- the hydroxyl value of the hydroxyl group-containing resin is preferably in the range of 20 to 200 mgKOH / g.
- the lower limit is more preferably 30 mgKOH / g
- the upper limit is more preferably 180 mgKOH / g.
- the number average molecular weight of the hydroxyl group-containing resin is preferably in the range of 1000 to 20000. If the number average molecular weight is less than 1000, workability and curability may be insufficient. Moreover, when it exceeds 20000, the non-volatile content at the time of coating will become low, and there exists a possibility that workability
- the lower limit is more preferably 2000, and the upper limit is more preferably 15000.
- the hydroxyl group-containing resin preferably further has an acid value within the range of 2 to 30 mgKOH / g.
- the upper limit is exceeded, the water resistance of the coating film is lowered, and when the lower limit is not reached, the curability of the coating film is lowered.
- the lower limit is more preferably 3 mgKOH / g, and the upper limit is more preferably 25 mgKOH / g.
- the content of the isocyanate compound relative to the hydroxyl group-containing resin can be appropriately selected within the range usually used by those skilled in the art.
- the equivalent ratio (NCO / OH) of isocyanate group (NCO) to hydroxyl group (OH) be used in an amount in the range of 0.5 to 1.7.
- the lower limit is more preferably 0.7, and the upper limit is more preferably 1.5.
- the method for producing the clear coating composition is not particularly limited, and any method known to those skilled in the art can be used. Moreover, a commercial item can also be used as a clear coating composition. Examples of commercially available products include Polyurexel O-1100 Clear, O-1200 Clear (manufactured by Nippon Paint Automotive Coatings Co., Ltd., isocyanate curable clear coating composition).
- a multilayer coating film can be formed by baking and curing at 70 ° C., preferably 70 to 110 ° C., more preferably 70 to 100 ° C. for 10 to 30 minutes. Further low-temperature curing conditions may be curing conditions in which baking curing is performed at 70 to 90 ° C. for 10 to 30 minutes.
- a clear coating composition other than the urethane clear coating composition can be used depending on the material of the object to be coated.
- an acid epoxy curable clear coating composition, an acrylic melamine curable clear coating composition, and the like can also be used.
- these clear coating compositions include, for example, “Mac Flow® O-570 Clear” or “Mack”, which is a clear coating composition containing polyepoxide and polyacid, which is sold by Nippon Paint Automotive Coatings Co., Ltd. “Super Lac® O-100 Clear” (trade name) sold by Nippon Paint Automotive Coatings Co., Ltd., which is a clear coating composition containing an acrylic resin and a melamine curing agent. ) And the like.
- the heat curing conditions when these clear coating compositions are used can be appropriately selected according to the composition of each clear coating composition.
- heat curing conditions when these clear coating compositions are used for example, conditions of heating at 120 to 140 ° C. for 10 to 30 minutes can be mentioned.
- the coating method of the clear coating composition the above-mentioned known coating method can be used, and for example, it can be applied by air spray, electrostatic coating or the like.
- the clear coating composition is preferably applied so that the dry film thickness is generally 10 to 80 ⁇ m, preferably 20 to 50 ⁇ m.
- Production Example 1 Production of acrylic emulsion having a hydroxyl group and a carboxyl group (AcEm-1) 2,000 parts of deionized water were charged into a reaction vessel equipped with a stirrer, nitrogen introduction tube, temperature controller, condenser, and dropping funnel. The temperature was raised to 80 ° C. with stirring in a nitrogen atmosphere.
- Latemul PD-104 (manufactured by Kao Corporation, 20 A pre-emulsion obtained by adding 100 parts of deionized water to 1,000 parts of deionized water was added dropwise over 2 hours together with an initiator aqueous solution in which 3 parts of ammonium persulfate was dissolved in 300 parts of deionized water. After completion of the dropwise addition, the reaction was further continued at 80 ° C.
- Production Example 2 Production of Acrylic Emulsion (AcEm-2) Having Hydroxyl Group and Carboxyl Group
- the amount of monomers in the monomer mixture of Production Example 1 is 90 parts of styrene, 249 parts of n-butyl acrylate, 403 parts of n-butyl methacrylate, Polymerization was carried out in the same manner except for changing to 232 parts of hydroxyethyl acrylate, 26 parts of acrylic acid and 3 parts of dodecyl mercaptan, respectively, and then 8.2 parts of N, N-dimethylaminoethanol was added to give a resin solid content of 30% by mass. An acrylic emulsion was obtained.
- the hydroxyl value in terms of resin solid content of this acrylic emulsion calculated from the monomer composition was 100 mgKOH / g, and the acid value was 20 mgKOH / g.
- the number average molecular weight by GPC measurement after the water removal of the acrylic resin in the obtained acrylic emulsion was 43,000.
- a monomer mixture of 50 parts of styrene, 20 parts of 2-ethylhexyl acrylate, 122 parts of 2-ethylhexyl methacrylate, 426 parts of n-butyl methacrylate, and 182 parts of hydroxyethyl methacrylate was added to Kayaester O (a tert product manufactured by Kayaku Akzo Corporation).
- Kayaester O a tert product manufactured by Kayaku Akzo Corporation
- -Butyl peroxy-2-ethylhexanate was added dropwise over 1.5 hours together with an initiator solution dissolved in 160 parts of 2-methoxy-1-propanol. After completion of the dropwise addition, the mixture was kept at 120 ° C.
- PTMG-1000 a polytetramethylene glycol having a number average molecular weight of 1,000 manufactured by Mitsubishi Chemical Corporation, tetramethylene calculated from the number average molecular weight.
- Oxide repeat unit 13.6 95 parts and dibutyltin dilaurate 0.2 part were added and heated to 85 ° C. and held for 2 hours.
- methyl polyglycol 130 polyethylene glycol monomethyl ether manufactured by Nippon Emulsifier Co., Ltd., ethylene oxide repeat number 9 calculated from a hydroxyl value of 130 mgKOH / g
- the reaction was terminated after confirming that the NCO peak had disappeared by IR measurement, and after cooling to 60 ° C., deionized water was added to the hydrophilized modified carbodiimide compound (1) having a resin solid content of 40% by mass. An aqueous dispersion was obtained.
- the resulting hydrophilized modified carbodiimide compound was a compound represented by the above general formula (I).
- Production Example 6 Production of Hydrophilized Modified Carbodiimide Compound (2)
- 43.2 parts and 0.07 part dibutyltin dilaurate were added and kept at 80 ° C. until there was no NCO absorption by IR.
- deionized water was added to obtain an aqueous dispersion of the hydrophilic modified carbodiimide compound (2) having a resin solid content of 25%.
- the resulting hydrophilized modified carbodiimide compound was a compound represented by the above general formula (III).
- GP-3000 a trivalent polyol having a structure in which propylene oxide was added to three hydroxyl groups of glycerin manufactured by Sanyo Chemical Industries, Ltd. on an average of 17 moles each
- GP-3000 a trivalent polyol having a structure in which propylene oxide was added to three hydroxyl groups of glycerin manufactured by Sanyo Chemical Industries, Ltd. on an average of 17 moles each
- Deionized water was added thereto and stirred to obtain an aqueous dispersion of a hydrophilic modified carbodiimide compound (3) having a resin solid content of 30% by mass.
- the resulting hydrophilized modified carbodiimide compound was a compound represented by the above general formula (II).
- Production Example 8 Production of Colored Pigment Paste After premixing 9.2 parts of a commercially available dispersant “Disperbyk 190” (manufactured by Big Chemie), 17.8 parts of ion-exchanged water, and 73.0 parts of rutile titanium dioxide, a paint conditioner The bead medium was added thereto, and the mixture was dispersed at room temperature until the particle size became 5 ⁇ m or less, and the bead medium was removed by filtration to obtain a colored pigment paste.
- Dispersant “Disperbyk 190” manufactured by Big Chemie
- ion-exchanged water 17.8 parts
- rutile titanium dioxide a paint conditioner
- emulsion resin (aqueous resin) having a hydroxyl value of less than 80 mgKOH / g
- Adekalya soap NE-20 ( ⁇ - [1-[(manufactured by ADEKA) 0.2 part of allyloxy) methyl] -2- (nonylphenoxy) ethyl] - ⁇ -hydroxyoxyethylene, 80% by weight solid content aqueous solution) and Aqualon HS-10 (polyoxyethylene alkylpropenyl phenyl manufactured by Daiichi Kogyo Seiyaku Co., Ltd.) Ether sulphate ester) (0.2 part) was added, and the mixture was heated to 80 ° C.
- Example 1 Production of aqueous intermediate coating composition
- the body (PE-DP) 18.7 parts (resin solid content 45%) was stirred.
- 137.7 parts of the color pigment paste of Production Example 8 was added thereto, pH was adjusted to 8.0 with 0.01 part of dimethylethanolamine (Kishida Chemical Co., Ltd.), and Adecanol UH-814N (urethane associated viscosity agent) , 30 parts by weight of solid content, manufactured by ADEKA, trade name) 1.0 part was mixed and stirred, and stirred until uniform.
- Adecanol UH-814N urethane associated viscosity agent
- Bihijur 305 polyisocyanate compound having an ethylene oxide group manufactured by Sumika Bayer Urethane Co., Ltd., ethylene oxide content: 20% by mass, isocyanate group content: 16% by mass
- a polyisocyanate compound (B) which is a polyisocyanate compound (B). 9 parts was added, and further, 8.3 parts of the hydrophilized modified carbodiimide compound (1) of Production Example 5 (resin solid content: 40% by mass) was added with stirring, followed by stirring to obtain an aqueous intermediate coating composition. It was.
- aqueous base coating composition In a container having a stirrer, 100 parts (resin solid content 30%) of the hydroxyl group-containing acrylic resin emulsion, which is the aqueous resin (A2) obtained in Production Example 1, and Production Example 9 125 parts (resin solid content: 24%) of the acrylic emulsion resin having a hydroxyl value of less than 80 mgKOH / g obtained in the above was stirred and mixed.
- WM44-L70G manufactured by Asahi Kasei Chemical Co., Ltd.
- D water-dispersible block polyisocyanate
- E aqueous polyurethane resin
- Power Nicks 150 (trade name, cationic electrodeposition coating made by Nippon Paint Automotive Coatings Co., Ltd.) is applied to a zinc phosphate-treated dull steel sheet so that the dry coating film has a thickness of 20 ⁇ m.
- the steel plate substrate was prepared by electrodeposition coating, cooling after heating and curing at 160 ° C. for 30 minutes.
- the above-mentioned aqueous intermediate coating composition is applied to the obtained substrate (substrate) with a rotary atomizing electrostatic coating apparatus so that the dry film thickness is 25 ⁇ m, and then the above-mentioned aqueous base coating is rotary atomized.
- the coating film was coated with an electrostatic coating apparatus so that the dry film thickness was 15 ⁇ m, and preheated at 80 ° C. for 3 minutes. Note that a 6-minute interval was provided between the coating of the aqueous intermediate coating composition and the aqueous base coating composition. Furthermore, Polyurexel O-1200 (trade name, manufactured by Nippon Paint Automotive Coatings Co., Ltd., polyisocyanate compound-containing two-component acrylic urethane-based organic solvent-type clear paint) is used as a clear paint on the coated plate. After coating with a coating apparatus so that the dry film thickness was 35 ⁇ m, heat curing was performed at 80 ° C. for 20 minutes to obtain a test piece on which a multilayer coating film was formed.
- Polyurexel O-1200 trade name, manufactured by Nippon Paint Automotive Coatings Co., Ltd., polyisocyanate compound-containing two-component acrylic urethane-based organic solvent-type clear paint
- Examples 2 to 10 In the production of the aqueous intermediate coating composition or the aqueous base coating composition, the aqueous intermediate coating composition was prepared in the same manner as in Example 1 except that the type and amount of each component were changed as described in the following table. And an aqueous base coating composition was prepared. A multilayer coating film was formed in the same manner as in Example 1 using the obtained aqueous intermediate coating composition and aqueous base coating composition. In addition, the usage-amount of each component shown in the following table
- surface is shown with the amount of solid content.
- Comparative Example 1 In the production of the aqueous intermediate coating composition, in the same manner as in Example 1, except that the hydrophilized modified carbodiimide compound (C) was not used and the amount of each component was changed to the amount described in the following table. A coating composition was prepared. A multilayer coating film was formed in the same manner as in Example 1 except that the obtained aqueous intermediate coating composition was used.
- Comparative Examples 2-8 In the production of the aqueous intermediate coating composition or the aqueous base coating composition, the aqueous intermediate coating composition was prepared in the same manner as in Example 1 except that the type and amount of each component were changed as described in the following table. And an aqueous base coating composition was prepared. A multilayer coating film was formed in the same manner as in Example 1 using the obtained aqueous intermediate coating composition and aqueous base coating composition.
- the measurement of the number average molecular weight in an Example is the value measured on the following GPC system measurement conditions.
- the measurement of the elongation at break of the water-based polyurethane resin used in each example and comparative example was performed according to the following procedure. Measurement of elongation at break of aqueous polyurethane resin 95 parts by mass (resin solid content) of aqueous polyurethane resin and 5 parts by mass (resin solid content) of the hydrophilized modified carbodiimide compound (C) described in Production Example 5 Were mixed to be 100 parts by mass in total. In a clean environment free from dust, dust, etc., the resulting mixed solution was uniformly applied on a flat polypropylene plate with a doctor blade so that the dry film thickness was 20 ⁇ m. After standing at 20 ° C. for 10 minutes, preheating was performed at 80 ° C.
- the obtained cured film was subjected to a tensile performance test under a test temperature of ⁇ 20 ° C. according to JIS K7127, and the elongation at break (breaking elongation) was measured. The measurement was performed 20 times, and the average value of 18 times excluding the maximum value and the minimum value was defined as the elongation at break of the sample.
- the obtained test plate was immersed in warm water at 40 ° C. for 240 hours, pulled up, dried at 20 ° C. for 24 hours, and then a multilayer coating film of the test plate was formed in a lattice pattern with a cutter so as to reach the substrate. Incisions were made and 100 goby meshes with a size of 2 mm ⁇ 2 mm were produced. Subsequently, an adhesive cellophane tape (trademark) was attached to the surface, and the remaining number of Gobang eyes coatings after the tape was rapidly peeled off at 20 ° C. was measured. The superiority or inferiority of the coating film can be determined by the number of peeling of the grid. If even one piece of the grid is peeled off, it is judged that practical use is difficult.
- the test plate for evaluating moisture resistance was exposed to an atmosphere of 50 ° C. and 99% humidity for 240 hours and then dried at 20 ° C. for 24 hours.
- the coating film state of the test plate was visually observed, and the change in appearance before and after the test was observed.
- ⁇ There is almost no difference in gloss and smoothness.
- ⁇ ⁇ Slight changes in gloss and smoothness are observed.
- ⁇ Changes in gloss and smoothness are observed, ⁇ ⁇ : Changes in gloss and smoothness are observed, but the change in gloss is particularly remarkable.
- X A remarkable difference can be confirmed in both gloss and smoothness.
- the moisture-proof blister property evaluation test plate was exposed to a temperature of 50 ° C. and 99% humidity for 240 hours, and then dried at 20 ° C. for 24 hours.
- the coating film state of the test plate was visually observed, and the change in appearance before and after the test was observed.
- ⁇ Almost no swelling.
- ⁇ ⁇ Although there is a small bulge of 0.01 mm or less, it is almost eliminated when dried at 20 ° C. for 24 hours.
- ⁇ ⁇ There is a swelling of 0.01 mm or more and 0.05 mm or less, and the swelling does not disappear even when dried at 20 ° C. for 24 hours.
- X There is a bulge of 0.05 mm or more, and the bulge does not disappear even when dried at 20 ° C. for 24 hours.
- the test plate was exposed to a 50 ° C. and 99% humidity atmosphere for 240 hours, then dried at 20 ° C. for 24 hours, and the multilayer coating film of the test plate was cut into a lattice shape with a cutter so as to reach the substrate. 100 gobangs with a size of 2 mm x 2 mm were made. Then, the adhesive cellophane tape was stuck on the surface, and the remaining number of the Gobang eyes coating film after peeling off the tape rapidly at 20 degreeC was measured. The superiority or inferiority of the coating film can be determined by the number of peeling of the grid. If even one piece of the grid is peeled off, it is judged that practical use is difficult.
- the test plate having the laminated coating film obtained in each Example and Comparative Example was subjected to a stepping stone test using a gravel tester KSS-1 (manufactured by Suga Test Instruments Co., Ltd.) under the following conditions. ⁇ Test conditions> Stone size: 6-8mm The amount of stone: 0.7-0.8g / piece distance: 35cm Shot pressure: 0.6 kg / cm 2 Shot angle: 45 ° Test temperature: -20 ° C
- the test plate after the stepping stone test was visually evaluated according to the following criteria. In the following criteria, 4 points or more are judged to be acceptable and usable. 5: Peeling is hardly seen.
- peeling area Although the peeling area is small, peeling at the interface between the electrodeposition coating film and the intermediate coating film is hardly observed.
- Peeling area is slightly large, and peeling is observed at the interface between the electrodeposition coating film and the intermediate coating film.
- 2 The peeling area is large, and peeling is observed at the interface between the electrodeposition coating film and the intermediate coating film.
- 1 The peeling area is large, and the electrodeposition coating film is broken.
- the types of the aqueous polyurethane resin (E) described in the above table are as follows.
- the multilayer coating films obtained in the examples all have excellent water resistance, moisture resistance and chipping resistance even when baked and cured under a low temperature condition of 80 ° C. Was confirmed. On the other hand, it was confirmed that the multilayer coating film obtained by the comparative example was inferior in one or more performances among water resistance, moisture resistance and chipping resistance.
- Comparative Example 1 is an example in which the hydrophilized modified carbodiimide compound (C) is not contained in the aqueous intermediate coating composition. In this case, the water resistance of the obtained multilayer coating film is clearly inferior.
- Comparative Example 2 is an example in which the hydrophilized modified carbodiimide compound (C) is not contained in the aqueous base coating composition. In this case, the resulting multilayer coating film is inferior in moisture resistance and moisture blister resistance.
- Comparative Example 3 the amount of the hydrophilized modified carbodiimide compound (C) contained in the aqueous base coating composition is 10 parts by mass, which is an example exceeding the scope of the present invention. Also in this case, the resulting multilayer coating film is inferior in moisture resistance and moisture blister resistance. Comparative Example 4 is an example in which the amount of the aqueous polyurethane resin (E) contained in the aqueous base coating composition is 5 parts by weight, which is less than the scope of the present invention. In this case, the chipping resistance of the obtained multilayer coating film is greatly inferior. In addition, it has poor moisture resistance and blister resistance.
- Comparative Example 5 is an example in which the amount of the water-dispersible block polyisocyanate compound (D) contained in the aqueous base coating composition is 5 parts by mass, which is less than the scope of the present invention. In this case, the resulting multilayer coating film is inferior in chipping resistance, moisture resistance, moisture blister resistance, and the like.
- Comparative Examples 6 to 8 are examples using an aqueous polyurethane resin outside the scope of the present invention. In this case, the resulting multilayer coating film is inferior in chipping resistance, moisture resistance, moisture blister resistance, and the like.
- a multilayer coating film is formed using a specific aqueous intermediate coating composition and an aqueous base coating composition, and in particular, an aqueous intermediate coating composition containing the specific component is used.
- An uncured coating film is formed, and then an aqueous resin (A2) having a hydroxyl group and a carboxyl group, a hydrophilized modified carbodiimide compound (C), a water-dispersible block polyisocyanate compound (D), and an aqueous polyurethane resin (E)
- A2 aqueous resin having a hydroxyl group and a carboxyl group
- C hydrophilized modified carbodiimide compound
- D water-dispersible block polyisocyanate compound
- E aqueous polyurethane resin
- the cured reaction proceeds well even under heating conditions under low-temperature conditions (for example, heating conditions of 100 ° C. or less), and has excellent coating film properties.
- the multilayer coating film forming method of the present invention can be suitably used, for example, for coating an object having a steel plate portion and a resin portion.
- the multilayer coating film forming method of the present invention is also an effective method as an environmental load reducing means such as energy saving and CO 2 emission reduction.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Application Of Or Painting With Fluid Materials (AREA)
- Paints Or Removers (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
Abstract
Description
[1]
水性中塗り塗料組成物を被塗物に塗装して、未硬化の中塗り塗膜を形成する、中塗り塗膜形成工程、
得られた未硬化の中塗り塗膜の上に、水性ベース塗料組成物を塗装して、未硬化のベース塗膜を形成する、ベース塗膜形成工程、
得られた未硬化の中塗り塗膜およびベース塗膜を加熱して硬化する、硬化工程、
を包含する、複層塗膜形成方法であって、
上記水性中塗り塗料組成物は、
水酸基およびカルボキシル基を有する水性樹脂(A1)、
ポリイソシアネート化合物(B)、および
親水化変性カルボジイミド化合物(C)、
を含む水性中塗り塗料組成物であり、
上記水性ベース塗料組成物は、
水酸基およびカルボキシル基を有する水性樹脂(A2)、
親水化変性カルボジイミド化合物(C)、
水分散性ブロックポリイソシアネート化合物(D)、および
水性ポリウレタン樹脂(E)、
を含む水性ベース塗料組成物であり、
上記水性中塗り塗料組成物中に含まれる、水酸基およびカルボキシル基を有する水性樹脂(A1)は、樹脂固形分換算で、80~200mgKOH/gの水酸基価、および、10~40mgKOH/gの酸価を有し、
上記水性ベース塗料組成物中に含まれる、水酸基およびカルボキシル基を有する水性樹脂(A2)は、樹脂固形分換算で、80~200mgKOH/gの水酸基価を有し、
上記親水化変性カルボジイミド化合物(C)は、下記一般式(I)、(II)または(III)で表されるものであり、
上記水性ポリウレタン樹脂(E)は、ガラス転移点(Tg)が-50℃以下であり、
上記水性ポリウレタン樹脂(E)の硬化膜の破断伸度が-20℃において400%以上であり、
上記水性ポリウレタン樹脂(E)の含有量は、上記水性ベース塗料組成物の樹脂固形分に対して10質量%以上50質量%以下であり、
上記水性ベース塗料組成物中に含まれる水分散性ブロックポリイソシアネート化合物(D)の含有量は、水性ベース塗料組成物の樹脂固形分に対して10~25質量%であり、
上記水性ベース塗料組成物中に含まれる上記親水化変性カルボジイミド化合物(C)の含有量は、水性塗料組成物の樹脂固形分に対して1~9質量%である、
複層塗膜形成方法。
[2]
上記水性中塗り塗料組成物中に含まれる親水化変性カルボジイミド化合物(C)の含有量は、水性中塗り塗料組成物の樹脂固形分に対して1~8質量%である、上記複層塗膜形成方法。
[3]
上記水性ポリウレタン樹脂(E)の含有量は、上記水性ベース塗料組成物の樹脂固形分に対して15質量%以上30質量%以下である、上記複層塗膜形成方法。
[4]
上記水性ベース塗料組成物中に含まれる上記親水化変性カルボジイミド化合物(C)の含有量は、水性ベース塗料組成物の樹脂固形分に対して1.5~7質量%であり、
上記水性ベース塗料組成物中に含まれる水分散性ブロックポリイソシアネート化合物(D)および親水化変性カルボジイミド化合物(C)の含有量の樹脂固形分質量比は、(D):(C)=25:1~1.25:1である、上記複層塗膜形成方法。
[5]
上記水性ベース塗料組成物は、さらに、水酸基価が80mgKOH/g未満である水性樹脂(F)を含む、上記複層塗膜形成方法。
[6]
上記被塗物は、鋼板部および樹脂部を含む、上記複層塗膜形成方法。
[7]
上記ベース塗膜形成工程で得られた未硬化のベース塗膜の上に、クリヤー塗料組成物を塗装して、未硬化のクリヤー塗膜を形成する、クリヤー塗膜形成工程、をさらに包含し、
上記硬化工程は、得られた未硬化の中塗り塗膜、ベース塗膜およびクリヤー塗膜を、加熱して硬化する工程である、上記複層塗膜形成方法。
[8]
上記硬化工程における加熱温度は、70~120℃である、上記複層塗膜形成方法。
水性中塗り塗料組成物を被塗物に塗装して、未硬化の中塗り塗膜を形成する、中塗り塗膜形成工程、
得られた未硬化の中塗り塗膜の上に、水性ベース塗料組成物を塗装して、未硬化のベース塗膜を形成する、ベース塗膜形成工程、および
得られた未硬化の中塗り塗膜およびベース塗膜を加熱して硬化する、硬化工程、
を包含する。本発明の複層塗膜形成方法においては、上記特定の水性中塗り塗料組成物および水性ベース塗料組成物を組み合わせて用いることによって、例えば低温硬化条件で水性塗料組成物を焼き付け硬化させる場合であっても、良好な耐水性および耐チッピング性を有する複層塗膜が得られることを特徴とする。以下、各塗膜形成工程で用いられる塗料組成物について説明する。
本発明の方法で用いられる水性中塗り塗料組成物は、水酸基およびカルボキシル基を有する水性樹脂(A1)、ポリイソシアネート化合物(B)、および、親水化変性カルボジイミド化合物(C)、を含む。
水酸基およびカルボキシル基を有する水性樹脂(A1)は、後述するポリイソシアネート化合物(B)および親水化変性カルボジイミド化合物(C)と硬化反応するバインダー成分である。そして本発明において用いられる水酸基およびカルボキシル基を有する水性樹脂(A1)は、
・樹脂固形分換算での水酸基価が80~200mgKOH/gであり、
・樹脂固形分換算での酸価が10~40mgKOH/gである、
ことを要件とする。
樹脂固形分換算での水酸基価は80~160mgKOH/gがより好ましく、樹脂固形分換算での酸価は15~35mgKOH/gがより好ましい。
本発明における水性中塗り塗料組成物は、上記水性樹脂(A1)を硬化させる成分として、ポリイソシアネート化合物(B)および親水化変性カルボジイミド化合物(C)の2種の成分が含まれる。ここでポリイソシアネート化合物(B)は、水分散性であってもよく、また疎水性であってもよい。疎水性であっても、後述する水分散性に優れた親水化変性カルボジイミド化合物(C)との相互作用により、水分散性が確保される。
本発明における水性中塗り塗料組成物に含まれる親水化変性カルボジイミド化合物(C)は、分子内に、
-OCONH-X-NHCOOY
[Xは少なくとも1個のカルボジイミド基を含有する2官能性有機基であり、Yはポリアルキレングリコールモノアルキルエーテルから水酸基を除いた構造である。]
で表される構造単位を1個または複数個有している。上記構造単位を有することで、優れた水分散性および優れた硬化性の両方の性能が得られると考えられる。
上記構造単位を2個有するものとしては、下記一般式(I)で表されるものがある。
(i)繰り返し数6~20のポリエチレンオキサイドユニットの末端に、炭素数1~3のアルキル基がエーテル結合した、ポリエチレングリコールモノアルキルエーテルから、水酸基を除いた構造
(ii)繰り返し数4~60のポリプロピレンオキサイドユニットの末端に、炭素数1~8のアルキル基がエーテル結合した、ポリプロピレングリコールモノアルキルエーテルから、水酸基を除いた構造:
から選択される、同一または異種の構造であるのがより好ましい。
さらに好ましくは、前記(ii)のポリプロピレンオキサイドユニットの繰り返し数が15~60である。
上記(i)および(ii)を有する、一般式(III)で示される親水化変性カルボジイミド化合物(C)を用いることによって、水分散性に優れ、安定性が向上し、さらに架橋密度がより高いレベルで保持されるという利点がある。
・繰り返し数6~20のポリエチレンオキサイドユニットの末端に、炭素数1~3のアルキル基がエーテル結合した、ポリエチレングリコールモノアルキルエーテル、または、
・繰り返し数4~60のポリプロピレンオキサイドユニットの末端に、炭素数1~8のアルキル基がエーテル結合した、ポリプロピレングリコールモノアルキルエーテル、
であるのがより好ましい。一般式(III)で示される親水化変性カルボジイミド化合物(C)の調製において、これらのポリエチレングリコールモノアルキルエーテルおよびポリプロピレングリコールモノアルキルエーテルは、単独で用いてもよく、併用してもよい。
上記ポリプロピレングリコールモノアルキルエーテルとしては、具体的にはポリプロピレングリコールモノメチルエーテル、ポリプロピレングリコールモノエチルエーテル、ポリプロピレングリコールモノブチルエーテル、ポリプロピレングリコール-2-エチルヘキシルエーテル等を挙げることができ、特にポリプロピレングリコールモノブチルエーテルが好適である。
一般式(III)で示される親水化変性カルボジイミド化合物(C)において、塗膜が形成したときに耐水性を向上させるため、カルボジイミド基の周辺がある程度疎水性となっていることが好ましい。また、水によるカルボジイミドの失活を抑止し、安定性を保持するため、カルボジイミド基の周辺がある程度疎水性となっており、水分子との接触が低い状態に保たれているのが好ましい。その一方で、一般式(III)で示されるカルボジイミド化合物において、親水性を維持するためには、ポリエチレングリコール構造を一定量有することが必要となる。ここで、上記(i)および(ii)の構造が、(i):(ii)=1:0.7~1:8の範囲内で存在する場合において、カルボジイミド化合物の親水性を確保しつつ、一方でカルボジイミド基の周辺においてある程度疎水性を保つことができる。これにより、低温硬化性により優れ、かつ、塗料安定性にもより優れた水性中塗り塗料組成物が得られるという利点がある。なお、上記比率(i):(ii)は、(i):(ii)=1:0.7~1:1.5の範囲内であるのがさらに好ましい。
本発明における水性中塗り塗料組成物は、水酸基およびカルボキシル基を有する水性樹脂(A1)、ポリイソシアネート化合物(B)、および、親水化変性カルボジイミド化合物(C)を含む。
本発明においては、酸価に対して水酸基価が非常に高い水性樹脂(A1)を用いることを特徴としている。水性樹脂(A1)の水酸基価がこのように高いことによって、得られる塗膜において高い架橋密度が達成されることとなる。また、水性樹脂(A1)の酸価が低いことによって、水性樹脂(A1)の酸基とポリイソシアネート化合物(B)のイソシアネート基の間に生じうる、望ましくない副反応の抑制が達成される。そして、ポリイソシアネート化合物(B)が有するイソシアネート基の当量に対する親水化変性カルボジイミド化合物(C)が有するカルボジイミド基の当量の比が0.01~0.20の範囲と、カルボジイミド基の量が極めて少ないことによって、塗料組成物保存後においても十分な架橋密度が達成されるなどの利点がある。
本発明の方法で用いられる水性ベース塗料組成物は、水酸基およびカルボキシル基を有する水性樹脂(A2)、水分散性ブロックポリイソシアネート化合物(D)、親水化変性カルボジイミド化合物(C)、および、水性ポリウレタン樹脂(E)、を含む。
水性ベース塗料組成物中に含まれる水性樹脂(A2)は、水性中塗り塗料組成物中に含まれる上記水性樹脂(A1)と同様の樹脂であるものの、水性樹脂(A1)における酸価の範囲規定を伴わない樹脂である。すなわち、水性ベース塗料組成物中に含まれる水性樹脂(A2)は、樹脂固形分換算で80~200mgKOH/gの水酸基価を有する樹脂である。この水性ベース塗料組成物中に含まれる水性樹脂(A2)は、10~40mgKOH/gの酸価を有するのが好ましい。
水性ベース塗料組成物中に含まれる親水化変性カルボジイミド化合物(C)は、水性中塗り塗料組成物中に含まれる上記親水化変性カルボジイミド化合物(C)である。本発明の方法において、水性ベース塗料組成物中に含まれる親水化変性カルボジイミド化合物(C)と、水性中塗り塗料組成物中に含まれる親水化変性カルボジイミド化合物(C)とは、完全に同一の樹脂であってもよく、また上記親水化変性カルボジイミド化合物(C)の定義範囲内において互いに異なる樹脂であってもよい。
本発明における水性ベース塗料組成物に含まれる水分散性ブロックポリイソシアネート化合物(D)は、ポリイソシアネート(D-1)に、マロン酸ジエステル(D-2)を反応させ、次いで得られた反応物と有機アミン化合物(D-3)とを反応させることによって調製することができる。
上記水性ベース塗料組成物は、上記成分に加えて水性ポリウレタン樹脂(E)を含む。水性ベース塗料組成物中に、特定の水性ポリウレタン樹脂(E)が含まれることによって、上記水性中塗り塗料組成物および水性ベース塗料組成物を塗装し、次いで、低温硬化条件で水性塗料組成物を焼き付け硬化させる場合であっても、水性ポリウレタン樹脂が自己および他の組成分と融着することで強靭な塗膜を形成することが可能なため、塗膜間密着性、耐水密着性などに優れた複層複層が得られることとなる。
ヘキサメチレンジイソシアネート、2,2,4-トリメチルヘキサンジイソシアネート、リジンジイソシアネ-トなどの、炭素数2~12の脂肪族ジイソシアネート;
1,4-シクロヘキサンジイソシアネート、イソホロンジイソシアネート、4,4’-ジシクロヘキシルメタンジイソシアネート、メチルシクロヘキシレンジイソシアネート、イソプロピリデンシクロヘキシル-4,4’-ジイソシアネートなどの、炭素数4~18の脂環族ジイソシアネート;
2,4-トルイレンジイソシアネート、2,6-トルイレンジイソシアネート、ジフェニルメタン-4,4’-ジイソシアネート、1,5’-ナフテンジイソシアネート、トリジンジイソシアネート、ジフェニルメチルメタンジイソシアネート、テトラアルキルジフェニルメタンジイソシアネート、キシリレンジイソシアネート、テトラメチルキシリレンジイソシアネート、4,4’-ジベンジルジイソシアネート、1,3-フェニレンジイソシアネート等の芳香族ジイソシアネート;
リジンエステルトリイソシアネート、トリフェニルメタントリイソシアネート、1,6,11-ウンデカントリイソシアネート、1,8-ジイソシアネート-4,4-イソシアネートメチルオクタン、1,3,6-ヘキサメチレントリイソシアネート、ビシクロヘプタントリイソシアネート等のトリイソシアネート類;
などが挙げられる。
また、これらのポリイソシアネート化合物の、ダイマー、トリマー(イソシアヌレート結合)で用いられてもよく、また、アミンと反応させてビウレットとして用いてもよい。更に、これらのポリイソシアネート化合物と、ポリオールを反応させたウレタン結合を有するポリイソシアネートも用いることができる。
有機ポリイソシアネート(E-3)として、脂肪族ジイソシアネートを用いるのがより好ましい。脂肪族ジイソシアネートを用いて水性ポリウレタン樹脂(E)を調製することによって、得られる塗膜の透水性を適切な範囲に調節することができ、また良好な低温初期耐水性を得ることができるという利点がある。
上記のように、水性ポリウレタン樹脂と、一般式(I)、(II)または(III)で表されるカルボジイミド化合物とを混合し焼き付けてフィルム状にすることで、水性ポリウレタン樹脂とカルボジイミド樹脂の相互の架橋および/または融着が進行し、水性ポリウレタン樹脂の破断伸度を評価できるようになる。
上記水性ベース塗料組成物は、必要に応じて、上記水性樹脂(A2)以外の樹脂成分(その他の樹脂)を含んでもよい。その他の樹脂の1例として、例えば、上記水性樹脂(A2)と同様に調製される樹脂であって、水酸基価が80mgKOH/g未満である樹脂が挙げられる。その他の樹脂の他の1例として、例えば、ポリエーテルジオール、ポリカーボネートジオールなどの、水酸基を有する樹脂などが挙げられる。その他の樹脂の他の1例として、例えば、ポリエーテルジオール、ポリカーボネートジオールなどの、水酸基を有する樹脂、および、メチロール基を有するメラミン樹脂、などが挙げられる。
上記水性ベース塗料組成物は、塗料組成物を構成する各成分を、通常用いられる手段によって混合することによって、調製することができる。上記水性ベース塗料組成物は、必要に応じて、顔料、表面調整剤(消泡剤、レベリング剤等)、顔料分散剤、可塑剤、造膜助剤、紫外線吸収剤、酸化防止剤、難燃剤、帯電防止剤、静電助剤、熱安定剤、光安定剤、溶剤(水、有機溶剤)その他の添加剤を含有してもよい。
本発明の複層塗膜形成方法は、
水性中塗り塗料組成物を被塗物に塗装して、未硬化の中塗り塗膜を形成する、中塗り塗膜形成工程、
得られた未硬化の中塗り塗膜の上に、水性ベース塗料組成物を塗装して、未硬化のベース塗膜を形成する、ベース塗膜形成工程、および
得られた未硬化の中塗り塗膜およびベース塗膜を加熱して硬化する、硬化工程、
を包含する。
本発明の複層塗膜形成方法においては、上記特定の水性中塗り塗料組成物および水性ベース塗料組成物を用いることによって、低温硬化条件であっても、塗膜物性に優れた塗膜を得ることができる。上記硬化工程における塗膜の加熱硬化温度は、特に限定されず、好ましくは70~120℃、より好ましくは70~110℃、さらに好ましくは70~100℃である。本発明の複層塗膜形成方法においては、このように、100℃以下の低温硬化条件で行うこともできる。このような低温硬化条件としては、塗膜の加熱硬化温度はさらに70~90℃といった硬化条件であってよい。
上記方法における被塗物として、鉄、鋼、ステンレス、アルミニウム、銅、亜鉛、スズなどの金属およびこれらの合金などの鋼板;ポリエチレン樹脂、EVA樹脂、ポリオレフィン樹脂(ポリエチレン樹脂、ポリプロピレン樹脂など)、塩化ビニル樹脂、スチロール樹脂、ポリエステル樹脂(PET樹脂、PBT樹脂などを含む)、ポリカーボネート樹脂、アクリル樹脂、アクリロニトリルブタジエンスチレン(ABS)樹脂、アクリロニトリルスチレン(AS)樹脂、ポリアミド樹脂、アセタール樹脂、フェノール樹脂、フッ素樹脂、メラミン樹脂、ウレタン樹脂、エポキシ樹脂、ポリフェニレンオキサイド(PPO)などの樹脂;および、有機-無機ハイブリッド材などが挙げられる。これらは成形された状態であってもよい。
撹拌機、窒素導入管、温度制御装置、コンデンサー、滴下ロートを備えた反応容器に、脱イオン水2,000部を仕込み、窒素雰囲気下で攪拌しながら80℃に昇温した。
スチレン103部、メタクリル酸n-ブチル290部、アクリル酸n-ブチル280部、アクリル酸ヒドロキシエチル302部、アクリル酸26部、ドデシルメルカプタン3部および乳化剤としてのラテムルPD-104(花王社製、20%水溶液)100部を脱イオン水1,000部に加えて乳化したプレエマルションを、過硫酸アンモニウム3部を脱イオン水300部に溶解した開始剤水溶液とともに2時間かけて滴下した。
滴下終了後、さらに80℃ で1時間反応を継続した後冷却し、N、N-ジメチルアミノエタール8.2部を加え、樹脂固形分30質量%のアクリルエマルションを得た。モノマー組成から計算される、このアクリルエマルションの樹脂固形分換算での水酸基価は130mgKOH/g、酸価は20mgKOH/gであった。また、得られたアクリルエマルションにおけるアクリル樹脂の、水分除去後のGPC測定による数平均分子量は、45,000であった。
製造例1のモノマー混合物におけるモノマーの量を、スチレン90部、アクリル酸n-ブチル249部、メタクリル酸n-ブチル403部、アクリル酸ヒドロキシエチル232部、アクリル酸26部、ドデシルメルカプタン3部にそれぞれ変更する以外は同様にして重合した後、N、N-ジメチルアミノエタノール8.2部を添加し樹脂固形分30質量%のアクリルエマルションを得た。モノマー組成から計算される、このアクリルエマルションの樹脂固形分換算での水酸基価は100mgKOH/g、酸価は20mgKOH/gであった。また、得られたアクリルエマルションにおけるアクリル樹脂の、水分除去後のGPC測定による数平均分子量は、43,000であった。
撹拌機、窒素導入管、温度制御装置、コンデンサー、滴下ロートを備えた反応容器に、2-メトキシ-1-プロパノール712部を仕込み、窒素雰囲気下で攪拌しながら120℃に昇温した。
スチレン50部、アクリル酸2-エチルヘキシル20部、メタクリル酸2-エチルヘキシル122部、メタクリル酸n-ブチル426部、メタクリル酸ヒドロキシエチル182部のモノマー混合物を、カヤエステルO(化薬アクゾ社製のtert-ブチルペルオキシ-2-エチルヘキサネート)24部を2-メトキシ-1-プロパノール160部に溶解した開始剤溶液とともに1.5時間かけて滴下した。
滴下終了後、120℃で1時間保ち、次いで、メタクリル酸n-ブチル50部、メタクリル酸ヒドロキシエチル119部、アクリル酸31部のモノマー混合物を、カヤエステルO 6部を2-メトキシ-1-プロパノール40部に溶解した開始剤溶液とともに1時間かけて滴下した。
滴下終了後、120℃で0.5時間保った後、後ショットとして、さらにカヤエステルO 3部を2-メトキシ-1-プロパノール55部に溶解した開始剤溶液を0.5時間かけて滴下し、さらに120℃で1時間攪拌して、樹脂固形分50質量%、GPC測定による数平均分子量6,000のアクリル樹脂を得た。
減圧加熱して溶媒を留去した後、N、N-ジメチルアミノエタノール39部を添加した後、さらに脱イオン水1206部を投入、攪拌することによって、樹脂固形分45質量%のアクリル樹脂水分散体を得た。モノマー組成から計算される、このアクリル水分散体の樹脂固形分換算での水酸基価は130mgKOH/g、酸価は24mgKOH/gであった。
撹拌機、窒素導入管、温度制御装置、コンデンサー、デカンターを備えた反応容器に、トリメチロールプロパン250部、アジピン酸824部、シクロヘキサンジカルボン酸635部を加え、180℃に昇温して、水が留出しなくなるまで縮合反応を行った。60℃まで冷却した後、無水フタル酸120部を加え、140℃まで昇温して、これを60分間保ち、GPC測定による数平均分子量2,000のポリエステル樹脂を得た。ジメチルアミノエタノール59部(樹脂が有する酸価の80%相当(中和率80%))を80℃で加え、さらに脱イオン水1920部を投入、攪拌することによって、樹脂固形分45質量%のポリエステル水分散体を得た。このポリエステル水分散体の樹脂固形分換算での水酸基価は90mgKOH/g、酸価は35mgKOH/gであった。
4,4-ジシクロヘキシルメタンジイソシアネート700部および3-メチル-1-フェニル-2-ホスホレン-1-オキシド7部を170℃で7時間反応させ、上記一般式(a)で表される構造の、1分子にカルボジイミド基を3個有し、両末端にイソシアネート基を有するカルボジイミド化合物を得た。
次に、製造したイソシアネート末端を有する4,4-ジシクロヘキシルメタンカルボジイミド180部に、PTMG-1000(三菱化学社製の数平均分子量1,000のポリテトラメチレングリコール、数平均分子量から計算されるテトラメチレンオキサイドの繰り返し単位13.6)95部およびジブチル錫ジラウレート0.2部を加えて、85℃に加熱し、これを2時間保った。
次いで、メチルポリグリコール130(日本乳化剤社製のポリエチレングリコールモノメチルエーテル、水酸基価130mgKOH/gから計算されるエチレンオキサイドの繰り返し数9)86.4部を加え、85℃で3時間保った。IR測定によりNCOのピークが消失していることを確認して反応を終了し、60℃に冷却した後、脱イオン水を加えて、樹脂固形分40質量%の親水化変性カルボジイミド化合物(1)の水分散体を得た。得られた親水化変性カルボジイミド化合物は、上記一般式(I)で表される化合物であった。
製造例5において製造したイソシアネート末端を有する4,4-ジシクロヘキシルメタンカルボジイミド90部に、繰り返し数が平均19のポリプロピレングリコールモノブチルエーテル120部、メチルポリグリコール130 43.2部およびジブチル錫ジラウレート0.07部を加え、IRでNCOの吸収がなくなるまで80℃で保った。60℃に冷却した後、脱イオン水を加えて樹脂固形分25%の親水化変性カルボジイミド化合物(2)の水分散体を得た。得られた親水化変性カルボジイミド化合物は、上記一般式(III)で表される化合物であった。
また、得られた親水化変性カルボジイミド化合物における、(i)ポリエチレングリコールモノアルキルエーテルから水酸基を除いた構造、および(ii)ポリプロピレングリコールモノアルキルエーテルから水酸基を除いた構造の比率は、(i):(ii)=1.0:1.0であった。
4,4-ジシクロヘキシルメタンジイソシアネート393部および3-メチル-1―フェニル-2-ホスホレン-1-オキシド8部を180℃で16時間反応させ、1分子にカルボジイミド基を4個有し、両末端にイソシアネート基を有するカルボジイミド化合物を得た。ここに、オキシエチレン基の繰り返し数が9であるポリエチレングリコールモノメチルエーテル130部およびジブチル錫ジラウレート0.2部を加え、90℃で2時間加熱して、末端がイソシアネート基および親水性基であるカルボジイミド化合物を得た。さらに、GP-3000(三洋化成工業社製のグリセリンの3つの水酸基に、プロピレンオキサイドを平均で17モルずつ付加した構造を有する3価のポリオール)300部を加え、90℃で6時間反応させた。IR測定によりNCOのピークが消失していることを確認して反応を終了し、親水化変性カルボジイミド化合物(3)を得た。ここに脱イオン水を加えて撹拌し、樹脂固形分30質量%の親水化変性カルボジイミド化合物(3)の水分散体を得た。得られた親水化変性カルボジイミド化合物は、上記一般式(II)で表される化合物であった。
市販の分散剤「Disperbyk 190」(ビックケミー社製)9.2部、イオン交換水17.8部、ルチル型二酸化チタン73.0部を予備混合した後、ペイントコンディショナー中でビーズ媒体を加え、室温で粒度5μm以下となるまで混合分散し、ビーズ媒体を濾過にて取り除いて着色顔料ペーストを得た。
イオン交換水194.1部を仕込んだ反応容器に、アデカリアソープNE-20(ADEKA社製α-[1-[(アリルオキシ)メチル]-2-(ノニルフェノキシ)エチル]-ω-ヒドロキシオキシエチレン、固形分80質量%水溶液)0.2部と、アクアロンHS-10(第一工業製薬社製ポリオキシエチレンアルキルプロペニルフェニルエーテル硫酸エステル)0.2部とを加え、窒素気流中で混合攪拌しながら80℃に昇温した。次いで、第1段目のα,β-エチレン性不飽和モノマー混合物として、アクリル酸メチル18.5部、アクリル酸エチル31.7部、アクリル酸2-ヒドロキシエチル5.8部、スチレン10.0部、アクリルアミド4.0部、アデカリアソープNE-20を0.3部、アクアロンHS-10を0.2部、およびイオン交換水70部からなるモノマー混合物と、過硫酸アンモニウム0.2部、およびイオン交換水7部からなる開始剤溶液とを2時間にわたり並行して反応容器に滴下した。滴下終了後、1時間同温度で熟成を行った。
さらに、80℃で第2段目のα,β-エチレン性不飽和モノマー混合物として、アクリル酸エチル24.5部、アクリル酸2-ヒドロキシエチル2.5部、メタクリル酸3.1部、アクアロンHS-10を0.3部、およびイオン交換水30部からなるモノマー混合物と、過硫酸アンモニウム0.1部、およびイオン交換水3部からなる開始剤溶液とを0.5時間にわたり並行して反応容器に滴下した。滴下終了後、2時間同温度で熟成を行った。
次いで、40℃まで冷却し、400メッシュフィルターで濾過した。さらに10質量%ジメチルアミノエタノール水溶液を加えpH7に調整し、平均粒子径110nm、固形分24質量%、樹脂固形分換算での酸価20mgKOH/g、水酸基価40mgKOH/gのエマルション樹脂を得た。全モノマー組成に基づきガラス転移点を算出したところ、0℃であった。
水性中塗り塗料組成物の製造
製造例1で得られた、水性樹脂(A1)であるアクリルエマルション(AcEm-1)158部(樹脂固形分30%)および製造例4で得られたポリエステル水分散体(PE-DP)18.7部(樹脂固形分45%)を撹拌した。これに製造例8の着色顔料ペーストを137.7部配合し、ジメチルエタノールアミン(キシダ化学社製)0.01部でpHを8.0に調整し、アデカノールUH-814N(ウレタン会合型粘性剤、固形分30質量%、ADEKA社製、商品名)1.0部を混合攪拌し、均一になるまで攪拌した。これに、ポリイソシアネート化合物(B)であるバイヒジュール305(住化バイエルウレタン社製のエチレンオキサイド基を有するポリイソシアネート化合物、エチレンオキサイド含有量:20質量%、イソシアネート基含有量:16質量%)40.9部を加え、さらに、製造例5の親水化変性カルボジイミド化合物(1)8.3部(樹脂固形分40質量%)を攪拌しながら加えて撹拌することにより、水性中塗り塗料組成物を得た。
撹拌機を有する容器に、製造例1で得られた、水性樹脂(A2)である水酸基含有アクリル樹脂エマルションを100部(樹脂固形分30%)、そして、製造例9で得られた水酸基価80mgKOH/g未満であるアクリルエマルション樹脂を125部(樹脂固形分24%)撹拌混合した。得られた混合物に対して、表中に記載の水性ポリウレタン樹脂(E)を66.7部(樹脂固形分30質量%)、水分散性ブロックポリイソシアネート(D)としてWM44-L70G(旭化成ケミカル製 固形分濃度 70.7%)を 21.2部、製造例5で調製した親水化変性カルボジイミド化合物(1)を 12.5部(樹脂固形分40質量%)、光輝性顔料としてアルペーストMH8801(旭化成社製アルミニウム顔料)21部(固形分65質量%)、リン酸基含有アクリル樹脂5部、ラウリルアシッドフォスフェート0.3部を添加し、さらに、2-エチルヘキサノール30部、アデカノールUH-814N3.3部(ADEKA社製増粘剤、固形分30質量%)、ジメチルエタノールアミン(キシダ化学社製)0.01部、そしてイオン交換水 150部を均一分散することにより水性ベース塗料組成物を得た。得られた塗料組成物のPWCは12.0%であった。
被塗物として、リン酸亜鉛処理したダル鋼板に、パワーニックス150(商品名、日本ペイント・オートモーティブコーティングス株式会社製カチオン電着塗料)を、乾燥塗膜が20μmとなるように電着塗装し、160℃で30分間の加熱硬化後冷却して、鋼板基板を準備した。
得られた基板(被塗物)に、上記水性中塗り塗料組成物を回転霧化式静電塗装装置にて乾燥膜厚が25μmとなるように塗装し、ついで上記水性ベース塗料を回転霧化式静電塗装装置にて乾燥膜厚が15μmとなるように塗装し、80℃で3分間プレヒートを行った。なお、水性中塗り塗料組成物と水性ベース塗料組成物との塗装の間に6分間のインターバルを置いた。さらに、その塗板にクリヤー塗料として、ポリウレエクセル O-1200 (商品名、日本ペイント・オートモーティブコーティングス株式会社製、ポリイソシアネート化合物含有2液アクリルウレタン系有機溶剤型クリヤー塗料)を回転霧化式静電塗装装置にて乾燥膜厚が35μmとなるように塗装した後、80℃で20分間の加熱硬化を行い、複層塗膜が形成された試験片を得た。
水性中塗り塗料組成物または水性ベース塗料組成物の製造において、各成分の種類、使用量を下記表に記載の通り変更したこと以外は、実施例1と同様にして、水性中塗り塗料組成物および水性ベース塗料組成物を製造した。
得られた水性中塗り塗料組成物および水性ベース塗料組成物を用いて、実施例1と同様にして、複層塗膜を形成した。
なお、下記表中に示される各成分の使用量は、固形分量で示している。
水性中塗り塗料組成物の製造において、親水化変性カルボジイミド化合物(C)を用いず、各成分の量を下記表に記載の量に変更したこと以外は、実施例1と同様にして、水性中塗り塗料組成物を調製した。
得られた水性中塗り塗料組成物を用いたこと以外は、実施例1と同様にして、複層塗膜を形成した。
水性中塗り塗料組成物または水性ベース塗料組成物の製造において、各成分の種類、使用量を下記表に記載の通り変更したこと以外は、実施例1と同様にして、水性中塗り塗料組成物および水性ベース塗料組成物を製造した。
得られた水性中塗り塗料組成物および水性ベース塗料組成物を用いて、実施例1と同様にして、複層塗膜を形成した。
装置:東ソー社製HLC-8220 GPC
カラム:Shodex KF-606M、KF-603
流速:0.6ml/min
検出器:RI、UV254nm
移動層:テトラヒドロフラン
標準サンプル:TSK STANDARD POLYSTYRENE(東ソー社製)、A-500、A-2500、F-1、F-4、F-20、F-80、F-700、1-フェニルヘキサン(アルドリッチ社製)
水性ポリウレタン樹脂の破断伸度の測定
水性ポリウレタン樹脂95質量部(樹脂固形分量)および製造例5に記載の親水化変性カルボジイミド化合物(C)5質量部(樹脂固形分量)を、2つの樹脂固形分が合計で100質量部となるように混合した。埃、ダストなどがかからないような清浄な環境において、出来上がった混合液を、平坦なポリプロピレン板の上に、乾燥膜厚が20μmとなるように、ドクターブレードで均一に塗装した。20℃で10分間静置した後、80℃で3分間プレヒートを行い、水分を揮発させた後に、120℃で30分間焼き付けて、硬化膜を調製した。得られた硬化膜を、JIS K7127に従い、試験時温度-20℃の条件下で引張性能試験を行い、破断時の伸び率(破断伸度)を測定した。測定は20回行い、最大値と最低値を除いた18回の平均値をそのサンプルの破断伸度とした。
得られた試験板を、40℃の温水に240時間浸漬し、引き上げ、20℃で24時間乾燥した後、試験板の複層塗膜を素地に達するようにカッターで格子状に切り込み、大きさ2mm×2mmのゴバン目を100個作成した。続いて、その表面に粘着セロハンテープ(商標)を貼着し、20℃においてそのテープを急激に剥離した後のゴバン目塗膜の残存枚数を計測した。
碁盤目の剥がれ枚数で塗膜の優劣を判定することができる。碁盤目の剥がれが一枚でも生じた場合は、実用上の使用は困難と判断する。
試験板を温度50℃かつ99%湿度雰囲気に240時間暴露した後、20℃で24時間乾燥させた。試験板の塗膜状態を目視で視認し、試験前後での外観の変化を観察した。下記基準において、○および○△評価の場合は、実用性があると判断する。
○:艶、平滑性にほとんど差が見られない。
○△:艶、平滑性にわずかに変化が見られる。
△:艶、平滑性に変化がみられる
△×:艶、平滑性とも変化がみられるが、特に艶の変化が顕著である。
×:艶、平滑性ともに顕著な差が確認できる。
試験板を温度50℃かつ99%湿度雰囲気に240時間暴露した後、20℃で24時間乾燥させた。試験板の塗膜状態を目視で視認し、試験前後での外観の変化を観察した。下記基準において、○および○△評価の場合は、実用性があると判断する。
○:膨れがほとんどない。
○△:0.01mm以下の小さい膨れがあるが、さらに20℃で24時間乾燥するとほとんどなくなる。
△:0.01mm以下の小さい膨れがあり、さらに20℃で24時間乾燥しても膨れがなくならない。
△×:0.01mm以上0.05mm以下の膨れがあり、さらに20℃で24時間乾燥しても膨れがなくならない。
×:0.05mm以上の膨れがあり、さらに20℃で24時間乾燥しても膨れがなくならない。
試験板を温度50℃かつ99%湿度雰囲気に240時間暴露した後、20℃で24時間乾燥させ、試験板の複層塗膜を素地に達するようにカッターで格子状に切り込み、大きさ2mm×2mmのゴバン目を100個作った。続いて、その表面に粘着セロハンテープを貼着し、20℃においてそのテープを急激に剥離した後のゴバン目塗膜の残存枚数を計測した。
碁盤目の剥がれ枚数で塗膜の優劣を判定することができる。碁盤目の剥がれが一枚でも生じた場合は、実用上の使用は困難と判断する。
各実施例および比較例で得られた積層塗膜を有する試験板を、グラベロ試験機KSS-1(スガ試験機社製)を用い、以下の条件下で飛石試験を行った。
<試験条件>
石の大きさ:6~8mm
石の量:0.7~0.8g/個
距離:35cm
ショット圧:0.6kg/cm2
ショット角度:45°
試験温度:-20℃
飛石試験後の試験板を、下記基準により目視評価した。下記基準において、4点以上を、実用上の使用が可能であり合格と判断する。
5:剥離がほとんど見られない。
4:剥離面積は小さいが、電着塗膜と中塗り塗膜との界面での剥離は殆ど見られない。
3:剥離面積がやや大きく、電着塗膜と中塗り塗膜との界面で剥離が見られる。
2:剥離面積が大きく、電着塗膜と中塗り塗膜との界面で剥離が見られる。
1:剥離面積が大きく、電着塗膜が破壊している。
A:N9603(楠本化成社製)、固形分濃度:34%、Tg:-10℃、破断伸度:12%
B:HUX-232(ADEKA社製)、固形分濃度:30%、Tg:-18℃、破断伸度:130%
C:N800(三洋化成社製)、固形分濃度:38%、Tg:-60℃、破断伸度:312%
D:パーマリンU150(三洋化成社製)、固形分濃度:30%、Tg:-60℃、破断伸度:610%
一方で、比較例により得られた複層塗膜は、耐水性、耐湿性および耐チッピング性のうち1つまたは複数の性能が劣っていることが確認された。
比較例1は、水性中塗り塗料組成物中に親水化変性カルボジイミド化合物(C)が含まれていない例である。この場合は、得られた複層塗膜の耐水性が明らかに劣っている。
比較例2は、水性ベース塗料組成物中に親水化変性カルボジイミド化合物(C)が含まれていない例である。この場合は、得られた複層塗膜の耐湿チヂミ性、耐湿ブリスタ性などが劣っている。
比較例3は、水性ベース塗料組成物中に含まれる親水化変性カルボジイミド化合物(C)の量が10質量部であり、本発明の範囲を超える例である。この場合もまた、得られた複層塗膜の耐湿チヂミ性、耐湿ブリスタ性などが劣っている。
比較例4は、水性ベース塗料組成物中に含まれる水性ポリウレタン樹脂(E)の量が5量部であり、本発明の範囲に満たない例である。この場合は、得られた複層塗膜の耐チッピング性が大きく劣っている。また、耐湿チヂミ性、耐湿ブリスタ性なども劣っている。
比較例5は、水性ベース塗料組成物中に含まれる水分散性ブロックポリイソシアネート化合物(D)の量が5質量部であり、本発明の範囲に満たない例である。この場合は、得られた複層塗膜の耐チッピング性、耐湿チヂミ性、耐湿ブリスタ性などが劣っている。
比較例6~8は、本発明の範囲外の水性ポリウレタン樹脂を用いた例である。この場合は、得られた複層塗膜の耐チッピング性、耐湿チヂミ性、耐湿ブリスタ性などが劣っている。
Claims (8)
- 水性中塗り塗料組成物を被塗物に塗装して、未硬化の中塗り塗膜を形成する、中塗り塗膜形成工程、
得られた未硬化の中塗り塗膜の上に、水性ベース塗料組成物を塗装して、未硬化のベース塗膜を形成する、ベース塗膜形成工程、
得られた未硬化の中塗り塗膜およびベース塗膜を加熱して硬化する、硬化工程、
を包含する、複層塗膜形成方法であって、
前記水性中塗り塗料組成物は、
水酸基およびカルボキシル基を有する水性樹脂(A1)、
ポリイソシアネート化合物(B)、および
親水化変性カルボジイミド化合物(C)、
を含む水性中塗り塗料組成物であり、
前記水性ベース塗料組成物は、
水酸基およびカルボキシル基を有する水性樹脂(A2)、
親水化変性カルボジイミド化合物(C)、
水分散性ブロックポリイソシアネート化合物(D)、および
水性ポリウレタン樹脂(E)、
を含む水性ベース塗料組成物であり、
前記水性中塗り塗料組成物中に含まれる、水酸基およびカルボキシル基を有する水性樹脂(A1)は、樹脂固形分換算で、80~200mgKOH/gの水酸基価、および、10~40mgKOH/gの酸価を有し、
前記水性ベース塗料組成物中に含まれる、水酸基およびカルボキシル基を有する水性樹脂(A2)は、樹脂固形分換算で、80~200mgKOH/gの水酸基価を有し、
前記親水化変性カルボジイミド化合物(C)は、下記一般式(I)、(II)または(III)で表されるものであり、
前記水性ポリウレタン樹脂(E)は、ガラス転移点(Tg)が-50℃以下であり、
前記水性ポリウレタン樹脂(E)の硬化膜の破断伸度が-20℃において400%以上であり、
前記水性ポリウレタン樹脂(E)の含有量は、前記水性ベース塗料組成物の樹脂固形分に対して10質量%以上50質量%以下であり、
前記水性ベース塗料組成物中に含まれる水分散性ブロックポリイソシアネート化合物(D)の含有量は、水性ベース塗料組成物の樹脂固形分に対して10~25質量%であり、
前記水性ベース塗料組成物中に含まれる前記親水化変性カルボジイミド化合物(C)の含有量は、水性塗料組成物の樹脂固形分に対して1~9質量%である、
複層塗膜形成方法。 - 前記水性中塗り塗料組成物中に含まれる親水化変性カルボジイミド化合物(C)の含有量は、水性中塗り塗料組成物の樹脂固形分に対して1~8質量%である、
請求項1記載の複層塗膜形成方法。 - 前記水性ポリウレタン樹脂(E)の含有量は、前記水性ベース塗料組成物の樹脂固形分に対して8質量%以上30質量%以下である、請求項1または2記載の複層塗膜形成方法。
- 前記水性ベース塗料組成物中に含まれる前記親水化変性カルボジイミド化合物(C)の含有量は、水性ベース塗料組成物の樹脂固形分に対して1.5~7質量%であり、
前記水性ベース塗料組成物中に含まれる水分散性ブロックポリイソシアネート化合物(D)および親水化変性カルボジイミド化合物(C)の含有量の樹脂固形分質量比は、(D):(C)=25:1~1.25:1である、
請求項1~3いずれかに記載の複層塗膜形成方法。 - 前記水性ベース塗料組成物は、さらに、水酸基価が80mgKOH/g未満である水性樹脂(F)を含む、請求項1~4いずれかに記載の複層塗膜形成方法。
- 前記被塗物は、鋼板部および樹脂部を含む、請求項1~5いずれかに記載の複層塗膜形成方法。
- 前記ベース塗膜形成工程で得られた未硬化のベース塗膜の上に、クリヤー塗料組成物を塗装して、未硬化のクリヤー塗膜を形成する、クリヤー塗膜形成工程、をさらに包含し、
前記硬化工程は、得られた未硬化の中塗り塗膜、ベース塗膜およびクリヤー塗膜を、加熱して硬化する工程である、
請求項1~6いずれかに記載の複層塗膜形成方法。 - 前記硬化工程における加熱温度は、70~120℃である、請求項1~7いずれかに記載の複層塗膜形成方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017563821A JP6851326B2 (ja) | 2016-01-27 | 2017-01-26 | 複層塗膜形成方法 |
| EP17744331.4A EP3409378B1 (en) | 2016-01-27 | 2017-01-26 | Method for forming multilayer coating film |
| CN201780020008.6A CN108883432B (zh) | 2016-01-27 | 2017-01-26 | 多层涂膜形成方法 |
| US16/072,926 US10696866B2 (en) | 2016-01-27 | 2017-01-26 | Method for forming multilayer coating film |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016013674 | 2016-01-27 | ||
| JP2016-013674 | 2016-01-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017131101A1 true WO2017131101A1 (ja) | 2017-08-03 |
Family
ID=59398140
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/002778 WO2017131101A1 (ja) | 2016-01-27 | 2017-01-26 | 複層塗膜形成方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US10696866B2 (ja) |
| EP (1) | EP3409378B1 (ja) |
| JP (1) | JP6851326B2 (ja) |
| CN (1) | CN108883432B (ja) |
| WO (1) | WO2017131101A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023119905A1 (ja) * | 2021-12-24 | 2023-06-29 | 日本ペイント・オートモーティブコーティングス株式会社 | 水性塗料組成物および塗装物の製造方法 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109235803A (zh) * | 2018-09-12 | 2019-01-18 | 上海市建筑装饰工程集团有限公司 | 历史建筑外墙水泥砂浆或混合砂浆墙面的做旧结构 |
| CN114555735B (zh) * | 2019-11-01 | 2023-03-07 | 关西涂料株式会社 | 水性涂料组合物 |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001009357A (ja) * | 1999-04-30 | 2001-01-16 | Nippon Paint Co Ltd | 塗膜形成方法 |
| JP2001011151A (ja) * | 1999-04-30 | 2001-01-16 | Nippon Paint Co Ltd | 熱硬化性水性塗料組成物およびこれを用いた塗膜形成方法、ならびに、複層塗膜形成方法 |
| JP2001011152A (ja) * | 1999-04-30 | 2001-01-16 | Nippon Paint Co Ltd | 親水化変性ポリカルボジイミド化合物の製造方法および親水化変性ポリカルボジイミド化合物、ならびにこれを含んだ常温硬化型水性塗料組成物 |
| JP2003201442A (ja) * | 2002-01-08 | 2003-07-18 | Nippon Paint Co Ltd | 水性下塗り材組成物 |
| JP2003306476A (ja) * | 2002-04-16 | 2003-10-28 | Nippon Paint Co Ltd | 3鎖型親水性カルボジイミド化合物及び水性塗料組成物 |
| JP2009516638A (ja) * | 2005-11-22 | 2009-04-23 | 日本ペイント株式会社 | カルボジイミド化合物及びそれを含む水性硬化性樹脂組成物 |
| JP2009262002A (ja) * | 2008-04-22 | 2009-11-12 | Nippon Paint Co Ltd | 複層塗膜形成方法 |
| JP2011094102A (ja) * | 2009-09-30 | 2011-05-12 | Nippon Paint Co Ltd | 水性塗料組成物 |
| JP2013060577A (ja) * | 2011-08-22 | 2013-04-04 | Basf Japan Ltd | 水性ベース塗料組成物及びそれを用いた複層塗膜形成方法 |
| JP2013133445A (ja) * | 2011-12-27 | 2013-07-08 | Nippon Paint Co Ltd | 水性塗料組成物 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5232736B2 (ja) * | 2008-09-02 | 2013-07-10 | 日東電工株式会社 | 複合フィルム |
| JP2011131135A (ja) | 2009-12-22 | 2011-07-07 | Nippon Paint Co Ltd | 複層塗膜形成方法 |
| US10870765B2 (en) * | 2015-12-28 | 2020-12-22 | Nippon Paint Automotive Coatings Co., Ltd. | Aqueous coating composition and method for forming a coating film |
-
2017
- 2017-01-26 WO PCT/JP2017/002778 patent/WO2017131101A1/ja active Application Filing
- 2017-01-26 US US16/072,926 patent/US10696866B2/en active Active
- 2017-01-26 CN CN201780020008.6A patent/CN108883432B/zh active Active
- 2017-01-26 JP JP2017563821A patent/JP6851326B2/ja active Active
- 2017-01-26 EP EP17744331.4A patent/EP3409378B1/en active Active
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001009357A (ja) * | 1999-04-30 | 2001-01-16 | Nippon Paint Co Ltd | 塗膜形成方法 |
| JP2001011151A (ja) * | 1999-04-30 | 2001-01-16 | Nippon Paint Co Ltd | 熱硬化性水性塗料組成物およびこれを用いた塗膜形成方法、ならびに、複層塗膜形成方法 |
| JP2001011152A (ja) * | 1999-04-30 | 2001-01-16 | Nippon Paint Co Ltd | 親水化変性ポリカルボジイミド化合物の製造方法および親水化変性ポリカルボジイミド化合物、ならびにこれを含んだ常温硬化型水性塗料組成物 |
| JP2003201442A (ja) * | 2002-01-08 | 2003-07-18 | Nippon Paint Co Ltd | 水性下塗り材組成物 |
| JP2003306476A (ja) * | 2002-04-16 | 2003-10-28 | Nippon Paint Co Ltd | 3鎖型親水性カルボジイミド化合物及び水性塗料組成物 |
| JP2009516638A (ja) * | 2005-11-22 | 2009-04-23 | 日本ペイント株式会社 | カルボジイミド化合物及びそれを含む水性硬化性樹脂組成物 |
| JP2009262002A (ja) * | 2008-04-22 | 2009-11-12 | Nippon Paint Co Ltd | 複層塗膜形成方法 |
| JP2011094102A (ja) * | 2009-09-30 | 2011-05-12 | Nippon Paint Co Ltd | 水性塗料組成物 |
| JP2013060577A (ja) * | 2011-08-22 | 2013-04-04 | Basf Japan Ltd | 水性ベース塗料組成物及びそれを用いた複層塗膜形成方法 |
| JP2013133445A (ja) * | 2011-12-27 | 2013-07-08 | Nippon Paint Co Ltd | 水性塗料組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3409378A4 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023119905A1 (ja) * | 2021-12-24 | 2023-06-29 | 日本ペイント・オートモーティブコーティングス株式会社 | 水性塗料組成物および塗装物の製造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN108883432A (zh) | 2018-11-23 |
| EP3409378A1 (en) | 2018-12-05 |
| JP6851326B2 (ja) | 2021-03-31 |
| JPWO2017131101A1 (ja) | 2018-12-13 |
| EP3409378A4 (en) | 2019-08-21 |
| US20190031915A1 (en) | 2019-01-31 |
| US10696866B2 (en) | 2020-06-30 |
| EP3409378B1 (en) | 2021-01-13 |
| CN108883432B (zh) | 2021-08-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6859267B2 (ja) | 水性塗料組成物および塗膜形成方法 | |
| US6248819B1 (en) | Thermosetting water-borne coating composition, method of forming a coating film using same, and method of forming a multilayer coating film | |
| JP5908276B2 (ja) | 水性塗料組成物 | |
| JP5534998B2 (ja) | 塗料組成物及び塗膜形成方法 | |
| JP6811731B2 (ja) | プライマー塗料組成物および塗膜形成方法 | |
| US20180230327A1 (en) | Urethane resin particles | |
| JP6884754B2 (ja) | 水性塗料組成物 | |
| JP2011094102A (ja) | 水性塗料組成物 | |
| JP2020002244A (ja) | 水性塗料組成物および水性塗料組成物の製造方法 | |
| JP6851326B2 (ja) | 複層塗膜形成方法 | |
| JP6851325B2 (ja) | 複層塗膜形成方法 | |
| CN113227266A (zh) | 水性涂料组合物和涂膜形成方法 | |
| WO2017131102A1 (ja) | 水性塗料組成物 | |
| WO2017131103A1 (ja) | 水性塗料組成物 | |
| WO2021084804A1 (ja) | 水性塗料組成物 | |
| JP6640578B2 (ja) | プライマー塗料組成物および塗膜形成方法 | |
| JP7402712B2 (ja) | 複層塗膜の製造方法 | |
| JP6903248B1 (ja) | 水性塗料組成物 | |
| WO2021084805A1 (ja) | 水性塗料組成物 | |
| WO2023119905A1 (ja) | 水性塗料組成物および塗装物の製造方法 | |
| JP2023088642A (ja) | 塗料組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17744331 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2017563821 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2017744331 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2017744331 Country of ref document: EP Effective date: 20180827 |