WO2016027709A1 - オーステナイト系ステンレス鋼板およびメタルガスケット - Google Patents
オーステナイト系ステンレス鋼板およびメタルガスケット Download PDFInfo
- Publication number
- WO2016027709A1 WO2016027709A1 PCT/JP2015/072605 JP2015072605W WO2016027709A1 WO 2016027709 A1 WO2016027709 A1 WO 2016027709A1 JP 2015072605 W JP2015072605 W JP 2015072605W WO 2016027709 A1 WO2016027709 A1 WO 2016027709A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- steel sheet
- mass
- stainless steel
- austenitic stainless
- cold
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/42—Ferrous alloys, e.g. steel alloys containing chromium with nickel with copper
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D9/00—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor
- C21D9/40—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor for rings; for bearing races
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B21—MECHANICAL METAL-WORKING WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21B—ROLLING OF METAL
- B21B1/00—Metal-rolling methods or mills for making semi-finished products of solid or profiled cross-section; Sequence of operations in milling trains; Layout of rolling-mill plant, e.g. grouping of stands; Succession of passes or of sectional pass alternations
- B21B1/22—Metal-rolling methods or mills for making semi-finished products of solid or profiled cross-section; Sequence of operations in milling trains; Layout of rolling-mill plant, e.g. grouping of stands; Succession of passes or of sectional pass alternations for rolling plates, strips, bands or sheets of indefinite length
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B21—MECHANICAL METAL-WORKING WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21D—WORKING OR PROCESSING OF SHEET METAL OR METAL TUBES, RODS OR PROFILES WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21D22/00—Shaping without cutting, by stamping, spinning, or deep-drawing
- B21D22/02—Stamping using rigid devices or tools
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0221—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the working steps
- C21D8/0226—Hot rolling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/02—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips
- C21D8/0221—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of plates or strips characterised by the working steps
- C21D8/0236—Cold rolling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D9/00—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor
- C21D9/46—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor for sheet metals
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/001—Ferrous alloys, e.g. steel alloys containing N
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/02—Ferrous alloys, e.g. steel alloys containing silicon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/04—Ferrous alloys, e.g. steel alloys containing manganese
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/06—Ferrous alloys, e.g. steel alloys containing aluminium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/34—Ferrous alloys, e.g. steel alloys containing chromium with more than 1.5% by weight of silicon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/46—Ferrous alloys, e.g. steel alloys containing chromium with nickel with vanadium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/48—Ferrous alloys, e.g. steel alloys containing chromium with nickel with niobium or tantalum
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/50—Ferrous alloys, e.g. steel alloys containing chromium with nickel with titanium or zirconium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/54—Ferrous alloys, e.g. steel alloys containing chromium with nickel with boron
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/40—Ferrous alloys, e.g. steel alloys containing chromium with nickel
- C22C38/58—Ferrous alloys, e.g. steel alloys containing chromium with nickel with more than 1.5% by weight of manganese
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F16—ENGINEERING ELEMENTS AND UNITS; GENERAL MEASURES FOR PRODUCING AND MAINTAINING EFFECTIVE FUNCTIONING OF MACHINES OR INSTALLATIONS; THERMAL INSULATION IN GENERAL
- F16J—PISTONS; CYLINDERS; SEALINGS
- F16J15/00—Sealings
- F16J15/02—Sealings between relatively-stationary surfaces
- F16J15/06—Sealings between relatively-stationary surfaces with solid packing compressed between sealing surfaces
- F16J15/08—Sealings between relatively-stationary surfaces with solid packing compressed between sealing surfaces with exclusively metal packing
- F16J15/0806—Sealings between relatively-stationary surfaces with solid packing compressed between sealing surfaces with exclusively metal packing characterised by material or surface treatment
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F16—ENGINEERING ELEMENTS AND UNITS; GENERAL MEASURES FOR PRODUCING AND MAINTAINING EFFECTIVE FUNCTIONING OF MACHINES OR INSTALLATIONS; THERMAL INSULATION IN GENERAL
- F16J—PISTONS; CYLINDERS; SEALINGS
- F16J15/00—Sealings
- F16J15/02—Sealings between relatively-stationary surfaces
- F16J15/06—Sealings between relatively-stationary surfaces with solid packing compressed between sealing surfaces
- F16J15/08—Sealings between relatively-stationary surfaces with solid packing compressed between sealing surfaces with exclusively metal packing
- F16J15/0818—Flat gaskets
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B21—MECHANICAL METAL-WORKING WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21B—ROLLING OF METAL
- B21B1/00—Metal-rolling methods or mills for making semi-finished products of solid or profiled cross-section; Sequence of operations in milling trains; Layout of rolling-mill plant, e.g. grouping of stands; Succession of passes or of sectional pass alternations
- B21B1/22—Metal-rolling methods or mills for making semi-finished products of solid or profiled cross-section; Sequence of operations in milling trains; Layout of rolling-mill plant, e.g. grouping of stands; Succession of passes or of sectional pass alternations for rolling plates, strips, bands or sheets of indefinite length
- B21B2001/225—Metal-rolling methods or mills for making semi-finished products of solid or profiled cross-section; Sequence of operations in milling trains; Layout of rolling-mill plant, e.g. grouping of stands; Succession of passes or of sectional pass alternations for rolling plates, strips, bands or sheets of indefinite length by hot-rolling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D2211/00—Microstructure comprising significant phases
- C21D2211/001—Austenite
Definitions
- the present invention relates to a stainless steel plate for a metal gasket excellent in strength, fatigue characteristics and high temperature softening resistance, and a metal gasket using the same.
- Engine cylinder head gaskets and exhaust manifold gaskets in automobiles, motorcycles, etc. are exposed to repeated pressure fluctuations under engine-specific high temperatures, high pressures and high vibrations.
- a cylinder gasket of an automobile engine is subjected to a high pressure during compression, it needs to be in contact with both contact counterparts with a high contact pressure (surface pressure) in order to maintain the sealing performance.
- a metal gasket used for an engine or an exhaust gas path is generally formed with a bead (continuous raised portion) having a certain height by bead molding by a press. This type of metal gasket ensures high sealing performance by using the top of the bead convex part (hereinafter referred to as “bead head”) by pressing it against the contact material.
- bead head the top of the bead convex part
- Patent Documents 1 and 2 disclose metastable austenitic stainless steels having these characteristics improved.
- Patent Document 3 describes that a martensitic steel type is applied to a gasket.
- metastable austenitic stainless steel is intended to increase strength by cold rolling.
- anisotropy occurs in bending workability and fatigue characteristics in the rolling parallel direction (L direction) and the rolling perpendicular direction (C direction) of the plate material.
- Such anisotropy when used as a metal gasket, becomes an impediment to maintaining a uniform contact surface pressure between the bead head and the contact partner material, and is a cause of performance degradation of the metal gasket.
- martensitic stainless steel there is no need to impart a high cold rolling rate and work hardening, so that the problem of anisotropy associated with work hardening hardly occurs.
- the present invention discloses a technique for reducing “anisotropy” of workability and fatigue resistance due to oxide inclusions in an austenitic stainless steel sheet.
- a technique for imparting “high temperature softening resistance” capable of maintaining high durability in applications of metal gaskets used at high temperatures and preventing “hot rolling cracks” in hot rolling is disclosed.
- the mass% value of the element in the steel composition is substituted for the location of the element symbol in the formulas (1) and (2), and the oxide system is substituted for the location of MnO and SiO 2 in the formula (3).
- the converted mass% values of MnO and SiO 2 in the average composition of inclusions are substituted.
- the Al content of the steel component element is the total Al content.
- Mass ratio conversion of Al 2 O 3 , SiO 2 and MnO means the content ratio of Al, Si and Mn of oxide inclusions in the mass ratio of single oxides of Al 2 O 3 , SiO 2 and MnO, respectively. It means to convert to.
- an austenitic stainless steel cold-rolled steel sheet derived from the above hot-rolled steel sheet can be exemplified.
- the austenitic stainless steel cold-rolled steel sheet has a Vickers hardness of the plate surface (rolled surface) adjusted to 400 to 500 HV, for example.
- the plate thickness can be set to 0.05 to 0.5 mm, for example, and may be controlled to 0.1 to 0.3 mm.
- the present invention provides a metal gasket formed from the cold-rolled steel sheet described above, having a bead formed by press molding, and pressing the top of the bead against a contact partner material. After bead molding, an aging treatment is performed, for example, at 100 to 500 ° C. as necessary.
- Bead head top means the top of the bead convex portion in contact with the contact partner material.
- the oxide inclusions present in the material have a low melting point and are softened, the oxide inclusions are a steel substrate (matrix) during hot rolling and subsequent cold rolling. In this way, it is avoided that coarse particles remain in the cold-rolled steel sheet that has been stretched in the rolling direction and thinned following the deformation. As a result, the workability and fatigue resistance starting from oxide inclusions are significantly improved.
- oxide-based inclusions have coarse particles that are partly divided by hot rolling and are distributed close to the rolling direction, which deteriorates the bending workability and fatigue resistance such that the bending ridge line is in the rolling direction. This is a factor causing anisotropy in workability and fatigue resistance.
- the cold-rolled steel sheet according to the present invention such anisotropy is reduced, and a gasket with high dimensional accuracy is obtained after bead forming. Further, even when the gasket is used, since the fatigue resistance anisotropy is small, the contact surface pressure applied to the top of the bead is maintained evenly. As a result, a metal gasket excellent in leak resistance is realized. Furthermore, since the cold-rolled steel sheet of the present invention is excellent in high temperature softening resistance, it is extremely useful for metal gaskets used at high temperatures.
- Al 2 O 3, and SiO 2, MnO ternary oxide composition shows the relationship between the wrought of oxide inclusions.
- the oxide inclusions are actually considered to be complex oxides mainly composed of Al, Si, and Mn. According to the inventors' detailed examination, when the oxide inclusions Al, Si, Mn content is expressed as a composition converted to a single oxide of Al 2 O 3 , SiO 2 , MnO, the oxide It was found that it was possible to specify an effective inclusion composition range for imparting extensibility to the system inclusions.
- the composition range generally coincided with the range having a relatively low melting point in the Al 2 O 3 , SiO 2 , and MnO ternary oxide equilibrium diagram.
- FIG. 1 shows the relationship between the ternary oxide composition of Al 2 O 3 , SiO 2 and MnO and the extensibility of oxide inclusions.
- the plot in the figure shows the results of evaluating the extension state of the oxide inclusions in a cross section (L cross section) parallel to the rolling direction and the plate thickness direction of a cold steel plate on a certain basis for a number of stainless steels. It is displayed. Specifically, the case where the individual oxide inclusion particles are crushed by cold rolling and clearly stretched in the rolling direction is indicated by ⁇ (extensibility; present). Coordinates of each plot, of oxide inclusions Al, the "average composition of oxide inclusions" when converted to the content of Si and Mn to the mass ratio of Al 2 O 3, SiO 2 and MnO, respectively To express.
- Oxide inclusions have extensibility. MnO ⁇ ⁇ 3SiO 2 +110 (3) As shown in the examples below, when the composition of oxide inclusions is in this region, the anisotropy of bending workability and fatigue resistance is remarkably improved, and it is particularly suitable for metal gaskets that require high performance. A material steel plate is obtained.
- the composition of oxide inclusions can be controlled mainly by the steel composition and steelmaking conditions. In particular, it is effective to ensure a sufficient Mn content in the steel composition and to limit the Al content. In addition, it is extremely effective to use deoxidation in steelmaking as Si deoxidation instead of Al deoxidation.
- FIG. 2 illustrates an optical micrograph of oxide inclusions observed in the L cross section at the stage where the hot-rolled annealed steel sheet is cold-rolled at a rolling rate of 40% to a thickness of 0.8 mm.
- 2A is an example of Comparative Example No. 22 described later
- FIG. 2B is an example of Invention Example No. 1 described later.
- oxide inclusions found in an austenitic stainless steel sheet are hard and exist in the steel sheet without being crushed so much by cold rolling as in (a). As the plate thickness decreases, the ratio of the diameter of inclusion particles to the plate thickness increases, which tends to hinder workability and fatigue resistance.
- the austenitic stainless steel sheet according to the present invention is adjusted to have a soft composition of oxide inclusions, and is crushed by rolling as shown in (b), following the metal flow of the steel substrate in the rolling direction. Expand. As the plate thickness decreases, the degree of extension of oxide inclusions also increases, and the adverse effects on bending workability and fatigue resistance properties become very small.
- the maximum diameter in the plate thickness direction of the oxide inclusions is preferably 5.0 ⁇ m or less in observation of the L cross section of the steel sheet to be formed, and is preferably 3.0 ⁇ m or less. More preferred. Further, it is more effective that the maximum diameter in the plate thickness direction is extended to 1.0% or less of the plate thickness.
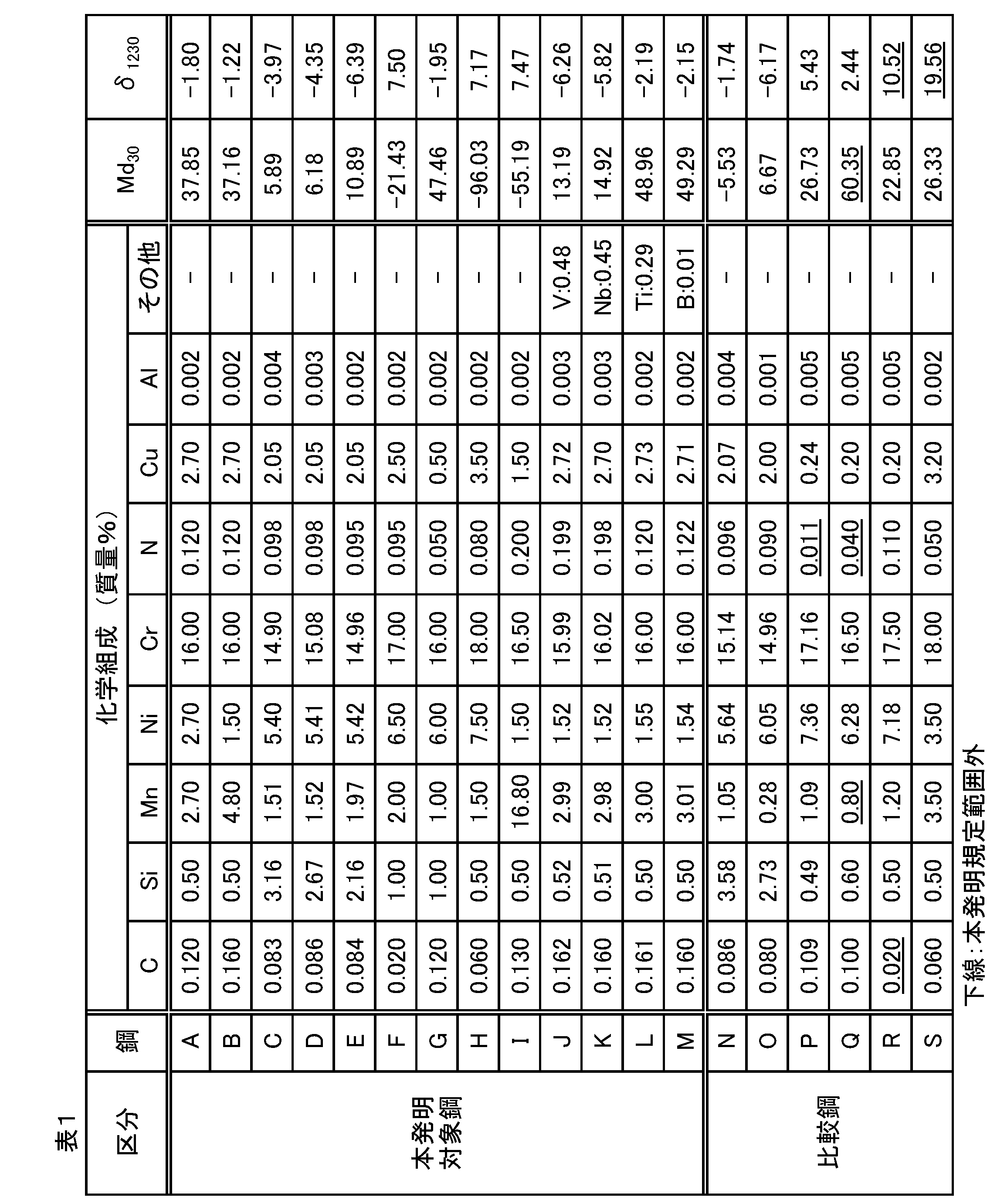
- Step composition The chemical composition (steel composition) of the steel sheet that is the subject of the present invention will be described.
- “%” in the steel composition means “% by mass” unless otherwise specified.
- C is an austenite-forming element and is an element effective for strengthening the austenite phase and the work-induced martensite phase. If the C content is too small, the above-described reinforcing action cannot be sufficiently exhibited. As a result of various studies, the C content needs to be 0.030% or more, and more preferably 0.060% or more. You may manage to C content exceeding 0.100%. However, excessive C content tends to cause grain boundary precipitation of Cr-based carbides during the cooling process, which causes a decrease in corrosion resistance. C content is adjusted in the range of 0.300% or less.
- Si is added as a deoxidizer during steelmaking. According to the study by the inventors, deoxidation with Si is extremely effective in controlling the composition of oxide inclusions in a soft region. It is necessary to add Si so that the Si content is 0.30% or more. Si has a large effect of hardening the austenite phase and the work-induced martensite phase, and this hardening effect is effective for increasing the strength of the gasket. However, excessive hardening is a factor that decreases workability and toughness. The Si content is limited to a range of 3.20% or less, and may be controlled to 3.00% or less.
- Mn is an austenite forming element and an important element for softening oxide inclusions. Moreover, when Mn content was raised, it turned out that the improvement effect of the high temperature softening-proof characteristic by ensuring N content enough can be enjoyed. This is because when Mn content is high, among C and N accumulated at the edge (blade part) of the edge dislocation by heating after cold working, N maintains the action of fixing the dislocation to a high temperature. It is inferred that this effect is caused by the fact that it becomes easier. That is, the combined action of Mn and N is considered to make it difficult for the “strain aging” structure to collapse even when heated at high temperatures. This is because, for example, Comparative Example No.
- the Mn content can also be controlled within a range exceeding 1.00%. However, when the Mn content increases, the burden on steelmaking increases, and depending on the application, corrosion resistance is insufficient. As a result of various studies, the Mn content is allowed up to 17.00%, but is preferably 8.00% or less, and may be controlled within a range of 5.00% or less.
- Ni is an austenite-generating element, and in the present invention, the Ni content is 1.00% or more. If the Ni content is less than that, it is difficult to adjust the components to obtain an austenite single phase structure in the state after annealing. In the present invention, since Mn is contained as described above, the Ni content can be saved thereby.
- the Ni content may be set within a range of 8.00% or less.
- Cr is an essential element for imparting the necessary corrosion resistance as stainless steel. For metal gasket applications, it is desirable to ensure a Cr content of 14.00% or more. However, since Cr is a ferrite-forming element, if the Cr content increases, the amount of austenite-forming elements necessary for obtaining an austenite single-phase structure after annealing also increases, leading to an increase in steel material cost. As a result of various studies, the Cr content is preferably set to a range of 19.00% or less.
- Cu is an austenite-forming element and is considered to be effective for improving the high temperature softening resistance similarly to Mn.
- steel with a Cu content of 0.50% or more is targeted.
- the Cu—Mn phase is likely to precipitate during heating before hot rolling, which causes cracks in hot rolling.
- N is an austenite-forming element and, like C, is effective for strengthening the austenite phase and the work-induced martensite phase.
- addition of N is important in order to sufficiently obtain the effect of improving the high temperature softening resistance, which is considered to be a combined effect with Mn as described above.
- the N content is set to 0.045% or more. If it is less than that, it will be difficult to improve the high temperature softening resistance in a stable manner. It is more effective to set the N content to 0.085% or more. Excessive N content causes nitrides to form during the cooling process after annealing, which causes a decrease in corrosion resistance and fatigue resistance. N content is limited to 0.250% or less.
- Al is an element having a strong deoxidizing action.
- the method of refining to contain Al so that the total amount of Al in steel is 0.0001% or more, and Si is mainly deoxidized, rather than deoxidizing Al alone.
- Increasing the Al content may adversely affect toughness.
- the total Al content in the steel is limited to 0.0300% or less.
- V, Nb, Ti, and B are effective elements for improving manufacturability, strength, fatigue resistance, and the like. One or more of these may be added as necessary.
- V is 0.50% or less
- Nb is 0.50% or less
- Ti is 0.30% or less
- B is 0.020% or less. More effective content ranges are: V: 0.01 to 0.50%, Nb: 0.01 to 0.50%, Ti: 0.01 to 0.30%, B: 0.0005 to 0.5. 020%.
- Md 30 value determined by equation adjusting the content of each element so that 50.0 or less.
- Md 30 551-462 (C + N) -9.2Si-8.1Mn-29 (Ni + Cu) -13.7Cr (1)
- the value of mass% of the element is substituted for the element symbol in the formula (1).
- Md 30 is an austenite stability index defined as the temperature (° C.) at which 50% of the structure transforms into martensite when a tensile strain of 30% is applied to single-phase austenite.
- the content of each component element is balanced so that the Md 30 value is 50.0 or less so that the work-induced martensite phase is not excessively induced.
- ⁇ 1230 -101.5-78.6C + 3.1Si + 0.4Mn-2.4Ni + 7.7Cr-1.5Cu-51.4N (2)
- ⁇ 1230 is an index representing the amount (volume%) of ⁇ ferrite phase at the center of the slab cross section after heating the slab obtained by continuous casting at 1230 ° C. for 120 min.
- the Cu—Mn phase is likely to precipitate during heating of the slab before hot rolling, and cracks occur during hot rolling. It becomes a factor to cause.
- the effect of the Cu—Mn phase on hot rolling cracks correlates with the abundance of the ⁇ ferrite phase, and in the steel type in which the total content of Mn and Cu is 1.4% by mass or more as in the steel of the present invention, ⁇ By adjusting the 1230 value to 8.0 or less, hot rolling cracks can be remarkably prevented.
- this ⁇ 1230 is a general austenitic stainless steel hot rolling condition (slab heating temperature: 1100 to 1350 ° C.) for an austenitic steel type in which the total content of Mn and Cu is 1.4% by mass or more. ) Is an index that leads to a stable technique for preventing hot rolling cracks when applied, and does not mean that the slab heating temperature before hot rolling should be 1230 ° C.
- ⁇ Production method ⁇ A typical manufacturing method is illustrated below.
- the steel adjusted to the above-mentioned chemical composition is melted by normal stainless steel making equipment to obtain a slab. No special treatment is required for high cleanliness.
- the deoxidation method is preferably Si deoxidation rather than Al single deoxidation. It is more effective to use Al in combination.
- hot rolling is performed in the same manner as in the production of a normal austenitic stainless steel sheet to obtain a hot rolled steel sheet.
- the slab heating temperature before hot rolling may be in the range of 1100 to 1350 ° C.
- the oxide inclusions present in the hot-rolled steel sheet are in the above-described composition range and are softened.
- cold rolling is performed to reduce the sheet thickness.
- intermediate annealing is performed during cold rolling.
- the oxide inclusions that have been softened are crushed by the reduction in cold rolling, and extend in the rolling direction following the metal flow of the steel substrate. What is necessary is just to give temper rolling after finishing annealing, and to make final product board thickness.
- the plate thickness is, for example, 0.05 to 0.5 mm. In this way, a cold rolled steel sheet derived from the hot rolled steel sheet is obtained.
- pickling is normally performed after annealing of each said process.
- the obtained cold-rolled steel sheet has solved the problem of anisotropy in conventional materials due to the presence of coarse oxide inclusions in the rolling direction, including metal gaskets. Suitable for various press working applications.
- a bead having a certain height is formed by bead press molding.
- the obtained pressed product can be subjected to an aging treatment at 100 to 500 ° C. as necessary.
- the above-mentioned “strain aging” effect is obtained, and the material is strengthened. Even when the aging treatment is not performed, the effect of strain aging can be obtained if the material is heated to a high temperature during use as a metal gasket. Since the metal gasket according to the present invention has improved high-temperature softening resistance as described above, it is less likely to cause strength reduction in high-temperature applications, and exhibits excellent durability in combination with the effect of reducing anisotropy of fatigue resistance. .
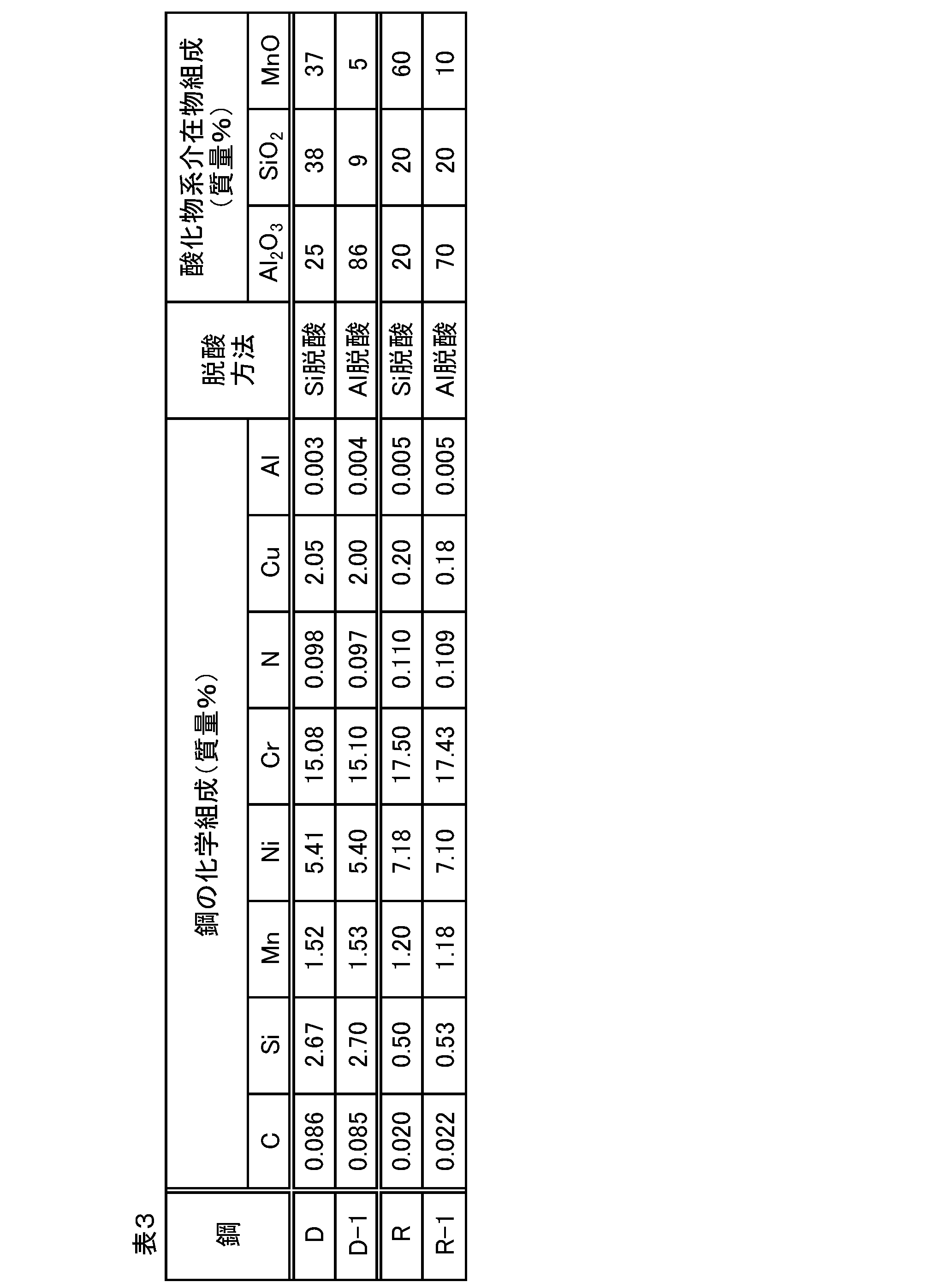
- composition analysis of oxide inclusions SEM observation is performed on the cross section (L cross section) parallel to the rolling direction and the plate thickness direction of the sample cut out from each hot rolled steel sheet, and 30 particles are randomly selected from the oxide inclusion particles existing in the L cross section. Was selected, and composition analysis was performed by EDX (energy dispersive X-ray analysis). The content of Al, Si and Mn of each inclusion is converted into the mass ratio of the single oxides Al 2 O 3 , SiO 2 and MnO, respectively, and the value of the mass ratio is averaged for 30 oxide inclusions. The average composition of oxide inclusions in the steel sheet was determined.
- each hot-rolled steel sheet was subjected to a heat treatment of 1100 ° C. ⁇ soaking 60 seconds, and then the sheet thickness was reduced by cold rolling.
- intermediate annealing with a soaking temperature of 60 sec in the range of 900 to 1100 ° C. is performed one or more times, and the hardness of the plate surface (rolled surface) becomes 430 to 460 HV at the final plate thickness of 0.2 mm.
- the temper rolling ratio was set and temper rolling was performed to obtain a cold-rolled steel sheet having a thickness of 0.2 mm.
- the cold-rolled steel sheet having the temper rolled finish thus obtained was subjected to the following test as a test material.
- the ratio of the minimum bending radius R and thickness t at which defects such as cracks did not occur outside the curved portion was defined as “bending limit R / t”.
- the bending limit R / t is 1.5 or less in both the L direction and the C direction, and the ratio of [the bending limit R / t value in the C direction] / [the bending limit R / t value in the L direction] is 1.3 or less.
- 3B and 3C are drawn with exaggerated dimensions in the thickness direction.
- a fatigue test was performed in which a swing stress was applied to the simulated bead portion, and a fatigue limit (fatigue limit; N / mm 2 ) at a repetition rate of 10 7 times was obtained.
- L direction when the both C direction fatigue limit is 300N / mm 2 or more and the difference between the fatigue limit of the L direction and C direction as is 30 N / mm 2 or less, excellent in metal gasket having a bead press formed part It can be evaluated that it exhibits fatigue resistance.
- FIG. 4 illustrates measurement results for some of the test materials.
- 4A shows Comparative Example No. 23, and
- FIG. 4B shows Example No. 1 of the present invention (No. is described in Table 2).
- a heating time of 120 h an increase in hardness due to strain aging is observed at heating temperatures of 300 ° C. and 400 ° C.
- the heating temperature becomes high, the material softens. Comparing the softening behavior in the high temperature region of 600 ° C.
- the softening is remarkably suppressed and the hardness of about 350 HV is maintained even when heated at 800 ° C. ⁇ 120 h. It can be seen that is significantly improved.
- the heating temperature at which the hardness decreases to 2/3 with respect to the hardness H 0 (HV) of the temper rolled material that is, the heating temperature at which 2 / 3H 0 (HV) is 800 ° C. or higher.
- HV hardness H 0
- the metal gasket formed with a bead has excellent high temperature softening resistance. Therefore, the high-temperature oxidation resistance was evaluated at a temperature of 2 / 3H 0 here.
- the composition of the oxide inclusions is in the above-described soft range, the anisotropy of bending workability and fatigue resistance is small, and good characteristics suitable for metal gaskets.
- the oxide inclusions were crushed by rolling and expanded in the rolling direction, and the maximum diameter in the thickness direction was 2 ⁇ m or less.
- the inventive examples also had good high temperature softening resistance.
- Comparative Examples No. 21 to 24 all had large inclusion work composition and anisotropy in bending workability and fatigue characteristics because the composition was out of the soft region. Since No. 22 had a low Mn content, the high temperature softening resistance was poor even though the N content was sufficiently secured. No. 23 had a low N content, and No. 24 had a low Mn content and a low N content, so these steel sheets also had poor high temperature softening resistance. No. 25 had a low C content, so the temper rolling ratio required for increasing the strength was high and the anisotropy was poor. No. 26 had a ⁇ 1230 value that was too high, and cracking occurred during hot rolling.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- General Engineering & Computer Science (AREA)
- Heat Treatment Of Sheet Steel (AREA)
- Gasket Seals (AREA)
Abstract
Description
Md30=551-462(C+N)-9.2Si-8.1Mn-29(Ni+Cu)-13.7Cr …(1)
δ1230=-101.5-78.6C+3.1Si+0.4Mn-2.4Ni+7.7Cr-1.5Cu-51.4N …(2)
MnO≧-3SiO2+110 …(3)
ここで、(1)式および(2)式の元素記号の箇所には鋼組成における当該元素の質量%値が代入され、(3)式のMnOおよびSiO2の箇所にはそれぞれ前記酸化物系介在物の平均組成におけるMnOおよびSiO2の換算質量%値が代入される。
鋼中に存在する介在物は、高延伸性タイプと難変形性タイプに大別される。前者は主として硫化物系、後者は主として酸化物系である。このうち、難変形性タイプの酸化物系介在物は、冷間圧延時にも展伸されにくく、粗大な粒子として鋼板中に残存する。粗大な酸化物系介在物粒子は加工性や耐疲労特性を劣化させる要因となる。通常、製鋼段階では介在物量の低減(高清浄度化)や小径化を意図した精錬や鋳造が行われる。しかし、過度な高清浄度化は製鋼工程の負荷を増大させ製品コストの増大を招く。そこで本発明では、一般的な清浄度レベルのオーステナイト系ステンレス鋼の溶製において実現可能な技術として、酸化物系介在物をできるだけ低融点化・軟質化させる手法を採用する。
MnO≧-3SiO2+110 …(3)
後述実施例に示すように、酸化物系介在物の組成がこの領域にあるとき、曲げ加工性や耐疲労特性の異方性が顕著に改善され、特に高性能が要求されるメタルガスケットに適した素材鋼板が得られる。
本発明の対象となる鋼板の化学組成(鋼組成)について説明する。以下、鋼組成における「%」は特に断らない限り「質量%」を意味する。
Cは、オーステナイト生成元素であり、オーステナイト相および加工誘起マルテンサイト相の強化に有効な元素である。C含有量が少なすぎると上記の強化作用が十分に発揮されない。種々検討の結果、C含有量は0.030%以上とする必要があり、0.060%以上とすることがより好ましい。0.100%を超えるC含有量に管理してもよい。ただし、過剰のC含有は冷却過程でCr系炭化物の粒界析出を招きやすく、耐食性低下の要因となる。C含有量は0.300%以下の範囲で調整する。
Md30=551-462(C+N)-9.2Si-8.1Mn-29(Ni+Cu)-13.7Cr …(1)
ここで、(1)式の元素記号の箇所には当該元素の質量%の値が代入される。Md30は、単相オーステナイトに対して30%の引張り歪を与えた時に、組織の50%がマルテンサイトに変態する温度(℃)として定義されるオーステナイト安定度の指標である。本発明ではMd30値が50.0以下となるように各成分元素の含有量をバランスさせ、加工誘起マルテンサイト相を過度に誘起させないようにする。
δ1230=-101.5-78.6C+3.1Si+0.4Mn-2.4Ni+7.7Cr-1.5Cu-51.4N …(2)
ここで、(2)式の元素記号の箇所には当該元素の質量%の値が代入される。δ1230は、連続鋳造によって得られた鋳片を1230℃×120min加熱した後の鋳片断面中央部におけるδフェライト相の量(体積%)を表す指標である。発明者らの検討によれば、オーステナイト系ステンレス鋼においてMn含有量とCu含有量を同時に増大させると熱間圧延前の鋳片加熱時にCu-Mn相が析出しやすくなり、熱間圧延時に割れを生じる要因となる。Cu-Mn相の熱間圧延割れに及ぼす影響はδフェライト相の存在量と相関があり、本発明対象鋼のようにMnとCuの合計含有量が1.4質量%以上となる鋼種ではδ1230値を8.0以下に調整することによって熱間圧延割れを顕著に防止できる。
代表的な製造方法を以下に例示する。上述の化学組成に調整された鋼を通常のステンレス鋼の製鋼設備によって溶製し、鋳片を得る。高清浄度化のための特殊な処理は必要としない。ただし、脱酸方法はAl単独脱酸よりもSi脱酸とすることが望ましい。Al添加も併用することがより効果的である。鋳片に対しては通常のオーステナイト系ステンレス鋼板の製造と同様に熱間圧延を行い、熱延鋼板を得る。熱間圧延前の鋳片加熱温度は1100~1350℃の範囲とすればよい。熱延鋼板中に存在する酸化物系介在物は上述の組成範囲にあり、軟質化されている。
各熱延鋼板から切り出した試料の圧延方向と板厚方向に平行な断面(L断面)についてSEM観察を行い、L断面内に存在する酸化物系介在物の粒子から無作為に30個の粒子を選択してEDX(エネルギー分散型X線分析)により組成分析を行った。個々の介在物のAl、SiおよびMnの含有率をそれぞれ単独酸化物Al2O3、SiO2およびMnOの質量割合に換算し、その質量割合の値を30個の酸化物系介在物について平均することによって当該鋼板における酸化物系介在物の平均組成を求めた。
供試材の板面(圧延面)についてビッカース硬さを測定した。
〔曲げ加工性〕
調質圧延仕上の冷延鋼板である供試材について、JIS Z2248:2006のVブロック法にて曲げ試験を行った。試験片の長手方向が圧延平行方向となるものをL方向、圧延直角方向となるものをC方向と表示する。L方向の曲げ試験片では曲げ稜線が圧延直角方向となり、C方向の曲げ試験片では曲げ稜線が圧延平行方向となる。湾曲部の外側に裂けきずなどの欠陥が生じない最小の曲げ半径Rと板厚tの比を「曲げ限界R/t」とした。試験数n=3にて曲げ試験を行い、3回のうち最も悪い結果をその試験での成績として採用した。L方向、C方向とも曲げ限界R/tが1.5以下であり、かつ[C方向の曲げ限界R/t値]/[L方向の曲げ限界R/t値]の比が1.3以下であるものは、ビードプレス成形に供するメタルガスケット用の素材鋼板として良好な曲げ加工性を有すると評価できる。
供試材から長手方向がL方向およびC方向の短冊状試料(幅8mm)をそれぞれ採取し、ビードプレス成形を行って図3(a)、(b)に示す形状の「初期ビード」を有する試験片に加工した。初期ビードの溝幅は約3mm、初期ビード高さは約0.4mmである。この初期ビード部に、メタルガスケットの初期締め相当の圧縮を加えて、図3(c)に示すように残存ビード高さが約0.1mmとなる模擬ビードを有する疲労試験片を作製した。なお、図3(b)、(c)に模式的に示す断面形状は板厚方向の寸法を誇張して描いてある。この疲労試験片を用いて模擬ビード部に両振り応力を付与する疲労試験を行い、繰返し数107回における疲労限(疲れ限度;N/mm2)を求めた。L方向、C方向とも疲労限が300N/mm2以上であり、かつL方向とC方向の疲労限の差が30N/mm2以下であるものは、ビードプレス成形部を有するメタルガスケットにおいて優れた耐疲労特性を呈すると評価できる。
調質圧延ままの各供試材について、300~800℃の範囲における100℃刻みの各温度で120h保持する加熱処理を施し、加熱処理後の板面(圧延面)の硬さを測定した。図4に、一部の供試材についての測定結果を例示する。図4(a)は比較例No.23、(b)は本発明例No.1である(No.は表2に記載のもの)。加熱時間120hの条件ではいずれも300℃、400℃の加熱温度でひずみ時効に起因する硬さの上昇が見られる。加熱温度が高温になると材料が軟化する。600℃以上の高温領域での軟化挙動を比べると、本発明例No.1では軟化が顕著に抑制されて800℃×120h加熱においても350HV程度の硬さを維持しており、耐高温軟化特性が顕著に改善されていることがわかる。この加熱試験において、調質圧延材の硬さH0(HV)に対して2/3まで硬さが低下する加熱温度、すなわち2/3H0(HV)となる加熱温度が800℃以上であれば、ビード成形されたメタルガスケットとして優れた耐高温軟化特性を有すると判断できる。そこで、ここでは2/3H0となる温度によって耐高温酸化特性を評価した。
これらの結果を表2に示す。なお、表2中に記載の「仕上焼鈍」は調質圧延前に行った最終焼鈍を意味する。
Claims (7)
- 質量%で、C:0.030~0.300%、Si:0.30~3.20%、Mn:0.90~17.00%、Ni:1.00~8.00%、Cr:14.00~19.00%、Cu:0.50~3.50%、N:0.045~0.250%、Al:0.0001~0.0300%、V:0~0.50%、Nb:0~0.50%、Ti:0~0.30%、B:0~0.010%、残部Feおよび不可避的不純物からなり、下記(1)式により定まるMd30値が50.0以下、下記(2)式により定まるδ1230値が8.0以下である鋼組成を有し、金属組織中に観察される酸化物系介在物の平均組成がAl2O3、SiO2およびMnOの質量割合換算でAl2O3:30質量%以下、SiO2:60質量%以下、MnO:15質量%以上、かつ下記(3)式を満たすオーステナイト系ステンレス鋼熱延鋼板。
Md30=551-462(C+N)-9.2Si-8.1Mn-29(Ni+Cu)-13.7Cr …(1)
δ1230=-101.5-78.6C+3.1Si+0.4Mn-2.4Ni+7.7Cr-1.5Cu-51.4N …(2)
MnO≧-3SiO2+110 …(3)
ここで、(1)式および(2)式の元素記号の箇所には鋼組成における当該元素の質量%値が代入され、(3)式のMnOおよびSiO2の箇所にはそれぞれ前記酸化物系介在物の平均組成におけるMnOおよびSiO2の換算質量%値が代入される。 - 請求項1に記載の熱延鋼板に由来するオーステナイト系ステンレス鋼冷延鋼板。
- 板面(圧延面)のビッカース硬さが400~500HVである請求項2に記載のオーステナイト系ステンレス鋼冷延鋼板。
- 板厚が0.05~0.5mmである請求項2に記載のオーステナイト系ステンレス鋼冷延鋼板。
- 板面(圧延面)のビッカース硬さが400~500HVであり、板厚が0.05~0.5mmである請求項2に記載のオーステナイト系ステンレス鋼冷延鋼板。
- 請求項2~5のいずれか1項に記載の冷延鋼板を成形したメタルガスケットであって、プレス成形によるビードを有し、ビード頭頂部を接触相手材に押し当てて使用するメタルガスケット。
- 請求項2~5のいずれか1項に記載の冷延鋼板を成形したのち100~500℃で時効処理したメタルガスケットであって、プレス成形によるビードを有し、ビード頭頂部を接触相手材に押し当てて使用するメタルガスケット。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/503,434 US10060003B2 (en) | 2014-08-19 | 2015-08-10 | Austenitic stainless steel sheet and metal gasket |
| EP15834321.0A EP3184662A4 (en) | 2014-08-19 | 2015-08-10 | Austenitic stainless steel sheet and metal gasket |
| CA2957391A CA2957391C (en) | 2014-08-19 | 2015-08-10 | Austenitic stainless steel sheet and metal gasket |
| KR1020177007431A KR102377582B1 (ko) | 2014-08-19 | 2015-08-10 | 오스테나이트계 스테인리스 강판 및 메탈 가스켓 |
| MX2017001943A MX2017001943A (es) | 2014-08-19 | 2015-08-10 | Lamina de acero inoxidable austenitico y junta metalica. |
| CN201580044095.XA CN106687612B (zh) | 2014-08-19 | 2015-08-10 | 奥氏体系不锈钢板和金属密封垫 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014166523A JP6095619B2 (ja) | 2014-08-19 | 2014-08-19 | オーステナイト系ステンレス鋼板およびメタルガスケット |
| JP2014-166523 | 2014-08-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016027709A1 true WO2016027709A1 (ja) | 2016-02-25 |
Family
ID=55350654
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/072605 WO2016027709A1 (ja) | 2014-08-19 | 2015-08-10 | オーステナイト系ステンレス鋼板およびメタルガスケット |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US10060003B2 (ja) |
| EP (1) | EP3184662A4 (ja) |
| JP (1) | JP6095619B2 (ja) |
| KR (1) | KR102377582B1 (ja) |
| CN (1) | CN106687612B (ja) |
| CA (1) | CA2957391C (ja) |
| MX (1) | MX2017001943A (ja) |
| TW (1) | TWI657152B (ja) |
| WO (1) | WO2016027709A1 (ja) |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6077693B1 (ja) * | 2016-03-09 | 2017-02-08 | 日新製鋼株式会社 | メタルガスケット用ステンレス鋼 |
| JP6791711B2 (ja) * | 2016-10-04 | 2020-11-25 | 日本冶金工業株式会社 | Fe−Cr−Ni合金およびその製造方法 |
| CN109112430A (zh) * | 2017-06-26 | 2019-01-01 | 宝钢不锈钢有限公司 | 一种低成本高强度节镍奥氏体不锈钢及制造方法 |
| CN107604262A (zh) * | 2017-08-09 | 2018-01-19 | 宁波市镇海甬鼎紧固件制造有限公司 | 一种高强度304不锈钢及其制备方法 |
| CN110373615B (zh) * | 2018-04-13 | 2022-04-01 | 宝钢德盛不锈钢有限公司 | 一种经济型细晶奥氏体不锈钢及其制造方法 |
| KR102385472B1 (ko) * | 2020-04-22 | 2022-04-13 | 주식회사 포스코 | 고강도, 고성형의 저원가 오스테나이트계 스테인리스강 및 그 제조방법 |
| EP4166680A4 (en) * | 2020-06-15 | 2023-07-26 | NIPPON STEEL Stainless Steel Corporation | PRECIPITATION-HARDENING TYPE MARTENSITIC STAINLESS STEEL SHEET HAVING EXCELLENT FATIGUE RESISTANCE |
| KR20220143758A (ko) * | 2020-07-17 | 2022-10-25 | 히타치 긴조쿠 가부시키가이샤 | 스테인리스 강박, 스위치용 스프링, 플렉시블 디스플레이용 기판 및 스테인리스 강박의 제조 방법 |
| CN111961989B (zh) * | 2020-08-05 | 2022-06-03 | 广西柳钢中金不锈钢有限公司 | 高氮低镍无铜奥氏体不锈钢的制造方法 |
| CN112063936B (zh) * | 2020-08-05 | 2022-06-03 | 广西柳钢中金不锈钢有限公司 | 高氮低镍无铜奥氏体不锈钢 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04202643A (ja) * | 1990-11-30 | 1992-07-23 | Nkk Corp | 高強度、高靭性ステンレス鋼およびその製造方法 |
| JPH07150304A (ja) * | 1993-11-29 | 1995-06-13 | Nkk Corp | 耐破断性に優れたidブレード基板用ステンレス鋼薄板およびその製造方法 |
| JPH08134595A (ja) * | 1994-11-11 | 1996-05-28 | Nippon Steel Corp | 耐応力腐食割れ特性に優れた高強度ステンレス鋼板 |
| JP2002371339A (ja) * | 2001-04-12 | 2002-12-26 | Nisshin Steel Co Ltd | 加工性,冷間鍛造性に優れた軟質ステンレス鋼板 |
| JP2003082441A (ja) * | 2001-09-10 | 2003-03-19 | Nisshin Steel Co Ltd | メタルガスケット用高強度オーステナイト系ステンレス鋼 |
| JP2011252208A (ja) * | 2010-06-03 | 2011-12-15 | Nisshin Steel Co Ltd | メタルガスケット用耐熱オーステナイト系ステンレス鋼 |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH02282424A (ja) * | 1989-04-20 | 1990-11-20 | Uchiyama Mfg Corp | メタルガスケットの製造方法 |
| DE4406052A1 (de) * | 1993-11-30 | 1995-06-01 | Nippon Kokan Kk | Rostfreies Stahlblech und Verfahren zu dessen Herstellung |
| JP2000109957A (ja) | 1998-10-05 | 2000-04-18 | Sumitomo Metal Ind Ltd | ガスケット用ステンレス鋼およびその製造方法 |
| US20040156737A1 (en) * | 2003-02-06 | 2004-08-12 | Rakowski James M. | Austenitic stainless steels including molybdenum |
| JP2002173742A (ja) * | 2000-12-04 | 2002-06-21 | Nisshin Steel Co Ltd | 形状平坦度に優れた高強度オーステナイト系ステンレス鋼帯およびその製造方法 |
| JP4321066B2 (ja) * | 2001-04-27 | 2009-08-26 | 住友金属工業株式会社 | 金属ガスケットとその素材およびそれらの製造方法 |
| JP4379804B2 (ja) * | 2004-08-13 | 2009-12-09 | 大同特殊鋼株式会社 | 高窒素オーステナイト系ステンレス鋼 |
| JP4859356B2 (ja) * | 2004-08-24 | 2012-01-25 | 日本リークレス工業株式会社 | メタルガスケットの製造方法 |
| KR101466044B1 (ko) * | 2007-03-09 | 2014-11-27 | 페더럴-모걸 코오포레이숀 | 금속 개스킷 |
| JP5177747B2 (ja) * | 2008-08-06 | 2013-04-10 | 独立行政法人産業技術総合研究所 | オーステナイト系ステンレス鋼、及びその水素添加方法 |
-
2014
- 2014-08-19 JP JP2014166523A patent/JP6095619B2/ja active Active
-
2015
- 2015-08-10 KR KR1020177007431A patent/KR102377582B1/ko active IP Right Grant
- 2015-08-10 CN CN201580044095.XA patent/CN106687612B/zh active Active
- 2015-08-10 MX MX2017001943A patent/MX2017001943A/es unknown
- 2015-08-10 US US15/503,434 patent/US10060003B2/en active Active
- 2015-08-10 EP EP15834321.0A patent/EP3184662A4/en not_active Withdrawn
- 2015-08-10 CA CA2957391A patent/CA2957391C/en active Active
- 2015-08-10 WO PCT/JP2015/072605 patent/WO2016027709A1/ja active Application Filing
- 2015-08-17 TW TW104126682A patent/TWI657152B/zh active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04202643A (ja) * | 1990-11-30 | 1992-07-23 | Nkk Corp | 高強度、高靭性ステンレス鋼およびその製造方法 |
| JPH07150304A (ja) * | 1993-11-29 | 1995-06-13 | Nkk Corp | 耐破断性に優れたidブレード基板用ステンレス鋼薄板およびその製造方法 |
| JPH08134595A (ja) * | 1994-11-11 | 1996-05-28 | Nippon Steel Corp | 耐応力腐食割れ特性に優れた高強度ステンレス鋼板 |
| JP2002371339A (ja) * | 2001-04-12 | 2002-12-26 | Nisshin Steel Co Ltd | 加工性,冷間鍛造性に優れた軟質ステンレス鋼板 |
| JP2003082441A (ja) * | 2001-09-10 | 2003-03-19 | Nisshin Steel Co Ltd | メタルガスケット用高強度オーステナイト系ステンレス鋼 |
| JP2011252208A (ja) * | 2010-06-03 | 2011-12-15 | Nisshin Steel Co Ltd | メタルガスケット用耐熱オーステナイト系ステンレス鋼 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3184662A4 * |
Also Published As
| Publication number | Publication date |
|---|---|
| MX2017001943A (es) | 2017-05-04 |
| CA2957391C (en) | 2022-08-30 |
| US10060003B2 (en) | 2018-08-28 |
| CA2957391A1 (en) | 2016-02-25 |
| EP3184662A1 (en) | 2017-06-28 |
| CN106687612A (zh) | 2017-05-17 |
| EP3184662A4 (en) | 2018-03-21 |
| JP2016041843A (ja) | 2016-03-31 |
| CN106687612B (zh) | 2019-08-13 |
| US20170233845A1 (en) | 2017-08-17 |
| KR20170054410A (ko) | 2017-05-17 |
| TW201615863A (zh) | 2016-05-01 |
| KR102377582B1 (ko) | 2022-03-24 |
| TWI657152B (zh) | 2019-04-21 |
| JP6095619B2 (ja) | 2017-03-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6095619B2 (ja) | オーステナイト系ステンレス鋼板およびメタルガスケット | |
| JP6124930B2 (ja) | マルテンサイト系ステンレス鋼板およびメタルガスケット | |
| US6488786B2 (en) | High-strength, high-toughness martensitic stainless steel sheet | |
| KR102244174B1 (ko) | 마텐자이트계 스테인리스 강판 | |
| JP6128291B2 (ja) | マルテンサイト系ステンレス鋼 | |
| JPH0820843A (ja) | 深絞り成形性と耐二次加工脆性に優れるクロム鋼板およびその製造方法 | |
| JP2010189719A (ja) | 時効硬化型ばね用ステンレス鋼板 | |
| JP4606113B2 (ja) | 比例限界応力の高いオーステナイト系ステンレス鋼材および製造法 | |
| JP2002030346A (ja) | 成形性に優れたCr含有耐熱耐食鋼板の製造方法 | |
| JP3420373B2 (ja) | 成形加工性に優れるクロム鋼板 | |
| JP2002332543A (ja) | 疲労特性及び耐高温ヘタリ性に優れたメタルガスケット用高強度ステンレス鋼及びその製造方法 | |
| JP6077693B1 (ja) | メタルガスケット用ステンレス鋼 | |
| JP4314962B2 (ja) | 疲労特性に優れた複合組織鋼板およびその製造方法 | |
| JP4315049B2 (ja) | 強度,疲労強度,耐食性及び耐磨耗性に優れた薄鋼帯板及びその製造方法 | |
| JP6095822B1 (ja) | マルテンサイト系ステンレス鋼板およびメタルガスケット製造法 | |
| JP4419572B2 (ja) | 疲労特性に優れた複合組織鋼板の製造方法 | |
| JP2005320612A (ja) | 無段変速機ベルトの金属帯リング用薄鋼帯板及びその製造方法 | |
| JPH05279812A (ja) | 耐応力腐食割れ特性に優れた高強度ばね用ステンレス鋼およびその製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15834321 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2957391 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2017/001943 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15503434 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015834321 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015834321 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20177007431 Country of ref document: KR Kind code of ref document: A |