WO2015064710A1 - アジュバント組成物、これを含むアジュバント製剤、及びキット - Google Patents
アジュバント組成物、これを含むアジュバント製剤、及びキット Download PDFInfo
- Publication number
- WO2015064710A1 WO2015064710A1 PCT/JP2014/078958 JP2014078958W WO2015064710A1 WO 2015064710 A1 WO2015064710 A1 WO 2015064710A1 JP 2014078958 W JP2014078958 W JP 2014078958W WO 2015064710 A1 WO2015064710 A1 WO 2015064710A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- adjuvant
- mass
- preparation
- adjuvant composition
- treatment
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/39—Medicinal preparations containing antigens or antibodies characterised by the immunostimulating additives, e.g. chemical adjuvants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0008—Antigens related to auto-immune diseases; Preparations to induce self-tolerance
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0014—Skin, i.e. galenical aspects of topical compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/70—Web, sheet or filament bases ; Films; Fibres of the matrix type containing drug
- A61K9/7023—Transdermal patches and similar drug-containing composite devices, e.g. cataplasms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/54—Medicinal preparations containing antigens or antibodies characterised by the route of administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/555—Medicinal preparations containing antigens or antibodies characterised by a specific combination antigen/adjuvant
- A61K2039/55511—Organic adjuvants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
- A61K9/0021—Intradermal administration, e.g. through microneedle arrays, needleless injectors
Definitions
- the present invention relates to an adjuvant composition, an adjuvant preparation containing the same, and a kit.
- vaccines are shifting from live vaccines to inactivated vaccines (whole particles, components, vaccines, etc.) for the purpose of improving safety.
- inactivated vaccine reduces the risk of infection and the like, but may lead to a decrease in immune response.
- Transdermal administration and transmucosal administration are characterized by being simple and excellent in safety as compared with subcutaneous administration by injection or the like.
- Patent Documents 1 and 2 contain at least one selected from aliphatic alcohols, free fatty acids and fatty acid derivatives, and do not contain a specific diunsaturated carboxylic acid ester, for transdermal or transmucosal administration.
- a number of adjuvants have been described.
- Patent Document 3 discloses an adjuvant for transdermal or transmucosal administration, comprising one or more selected from the group consisting of glycerin, propylene glycol, polyethylene glycol and triacetin, which are polyhydric alcohols or derivatives thereof. Is described.
- the adjuvant When the adjuvant is administered transdermally or transmucosally, it is required to sufficiently suppress skin irritation.
- adjuvants that can be efficiently administered transdermally or transmucosally have sufficiently suppressed skin irritation, and can sufficiently exert an immune response enhancing action, particularly adjuvants composed of small molecules, are still available.
- the current number is small.
- An object of the present invention is to provide an adjuvant composition for transdermal or transmucosal administration capable of sufficiently suppressing skin irritation and exhibiting a sufficient immune response enhancing action.
- the present invention relates to an adjuvant composition containing lauryl alcohol and propylene glycol, wherein the contents of lauryl alcohol and propylene glycol are 0.5 to 25% by mass and 8.0 to An adjuvant composition is provided that is 99.5% by weight, has lauryl alcohol dissolved therein, and is intended for transdermal or transmucosal administration.
- each component contained in this invention is a compound known as an absorption promoter of a transdermal formulation.
- the effect brought about by the combination of the above components in the present adjuvant composition is to enhance the immune response of the subject, and the effect on the mechanism of action and the effect on the absorption enhancer in the transdermal preparation are completely different. Even when the present adjuvant composition is applied after the whole amount of the antigen is administered in the form of an injection, it can be confirmed from the fact that the immune response is enhanced.
- the adjuvant composition of the present invention further contains glycerin, and the contents of lauryl alcohol, propylene glycol and glycerin are 0.5 to 25% by mass and 8.0 to 90% by mass, respectively, based on the total mass of the adjuvant composition. And 1.0 to 90% by mass is preferable.
- the ratio of the propylene glycol content to the lauryl alcohol content is preferably 1.0 to 99.
- the ratio of the propylene glycol content to the glycerin content is preferably 0.4 to 99.
- the present invention also provides an adjuvant preparation containing an adjuvant composition.
- Such an adjuvant preparation contains the above-mentioned adjuvant composition, skin irritation is sufficiently suppressed, and a sufficient immune response enhancing action can be exhibited.
- the content of the adjuvant composition may be 50 to 100% by mass based on the total mass of the adjuvant preparation.
- the content of the adjuvant composition is in such a range, the immune response enhancing action of the adjuvant preparation can be further improved.
- Adjuvant preparations can be ointments, creams, gels, suppositories, poultices, patch preparations, lotions, liquids, impregnations or blisters.
- the patch preparation may be a matrix-type tape preparation, a laminated-type tape preparation, or a reservoir-type patch preparation. According to such a patch preparation, the adjuvant composition can be administered more rapidly and over a long period of time.
- the adjuvant composition may be administered to the skin or mucous membrane by microneedle puncture, needleless injection, skin polishing or mucosal polishing.
- the adjuvant composition may be coated on at least a part of the microneedle and administered to the skin or mucous membrane by puncture.
- the adjuvant composition may be administered by iontophoresis, sonophoresis or electroporation.
- the adjuvant preparation can be administered to intact skin, intact mucosa, skin subjected to physical or chemical treatment, or mucosa subjected to physical or chemical treatment.
- the physical or chemical treatment is at least one treatment selected from the group consisting of heat treatment, ultrasonic treatment, electric field treatment, magnetic field treatment, pressure treatment, alkali treatment, laser irradiation, polishing treatment, and treatment with microneedles. May be.
- the adjuvant preparation may be administered to the skin or mucous membrane before or after the antigen administration.
- the present invention also provides a kit comprising the above adjuvant composition or the above adjuvant preparation.
- the kit may further include an antigen or a device for antigen administration.
- the present invention can also be said to provide an immunostimulation method for individuals who need to promote immune responses.
- the present invention provides an immunostimulation method including transdermally or transmucosally administering the above adjuvant composition or the above adjuvant preparation to a human.
- an adjuvant composition for transdermal or transmucosal administration capable of sufficiently suppressing skin irritation and exhibiting a sufficient immune response enhancing action.
- the present invention can also provide an adjuvant preparation containing an adjuvant composition. Since such an adjuvant preparation contains the adjuvant composition according to the present invention, skin irritation is sufficiently suppressed and a sufficient immune response enhancing action can be exhibited. In addition, since the adjuvant preparation according to the present invention can contain the above-mentioned adjuvant composition at a high concentration, it is excellent in enhancing the immune response when administered to the skin or mucous membrane.
- the present invention can also provide a kit comprising an adjuvant composition or an adjuvant formulation. Moreover, this invention can also provide the immunostimulation method including transdermally or transmucosally administering an adjuvant composition or an adjuvant formulation with respect to the individual who needs promotion of an immune reaction.
- the adjuvant composition for transdermal or transmucosal administration is an adjuvant composition containing lauryl alcohol and propylene glycol, and the content of lauryl alcohol and propylene glycol is based on the total mass of the adjuvant composition. And 0.5 to 25% by mass and 8.0 to 99.5% by mass, respectively, characterized in that lauryl alcohol is dissolved.
- the fact that lauryl alcohol is dissolved means that it is evaluated as A or B, and preferably evaluated as A in Test Example 1 described later.
- the adjuvant composition having the above-described configuration can be transdermally or transmucosally administered, can sufficiently suppress skin irritation, and can exhibit an excellent immune response enhancing effect (immunostimulatory effect). Compared to conventional impregnating agents applied directly to the skin, the skin irritation is further suppressed, and an immunostimulatory effect can be obtained safely. Such an effect can be confirmed by measuring the immune IgG antibody titer.
- the adjuvant composition according to the present embodiment itself can exert an immune response enhancing action, it is not necessary to administer it in a mixture with the antigen, and it can be administered separately from the antigen. It is preferable to administer transdermally or transmucosally independently of the antigen because an excellent immune response enhancing effect can be obtained.
- the timing of administration of the adjuvant composition may be any before the antigen administration, during the antigen administration, or after the antigen administration. It is preferably performed before or after antigen administration.
- the dosage form of the adjuvant composition can also be administered in a dosage form different from the antigen, and may be administered by a route independent of the antigen.
- the adjuvant composition when administering the adjuvant composition, it is not necessary to consider conditions such as the dosage of the antigen, and the dosage, administration time, dosage form, etc. of the adjuvant composition itself can be selected. Since the adjuvant composition according to this embodiment can be administered separately from the antigen, the adjuvant composition can be administered while avoiding swelling and pain at the administration site that occurs during administration of the antigen.
- the content of lauryl alcohol may be 0.5 to 60% by mass, preferably 0.5 to 25% by mass, and 1.0 to 20% by mass based on the total mass of the adjuvant composition. It is more preferable. By setting the content of lauryl alcohol to 60% by mass or less, the skin irritation of the adjuvant composition can be sufficiently suppressed. Further, by setting the content of lauryl alcohol to 0.5% by mass or more, it is possible to sufficiently obtain the immune response enhancing action of the adjuvant composition. Within the above range, the content of lauryl alcohol is 0.5 to 60% by mass, 0.5 to 50% by mass, 0.5 to 45% by mass, 0.5 to 45% by mass based on the total mass of the adjuvant composition.
- the content of lauryl alcohol may be 1.0 to 25% by mass or 1.0 to 20% by mass based on the total mass of the adjuvant composition.
- the content of propylene glycol is 8.0 to 99.5% by mass based on the total amount of the adjuvant composition, but is preferably 40 to 99% by mass, and more preferably 49 to 99% by mass.
- the content of propylene glycol is 8.0% by mass or more, lauryl alcohol can be sufficiently dissolved.
- the propylene glycol content is 8.0 to 99.5% by mass, 40 to 99.5% by mass, or 49 to 99.5% by mass based on the total amount of the adjuvant composition. Also good.
- the propylene glycol content may be 8.0 to 99% by mass, 40 to 99% by mass, or 49 to 99% by mass based on the total amount of the adjuvant composition.
- the adjuvant composition according to this embodiment can further contain glycerin.
- glycerin By containing glycerin, it is possible to further improve the immune response enhancing action of the adjuvant composition.
- the content of propylene glycol is preferably 8.0 to 90% by mass, more preferably 49 to 90% by mass, based on the total mass of the adjuvant composition, More preferably, it is 49 to 79% by mass.
- the content of propylene glycol when the adjuvant composition contains glycerin may be 8.0 to 90% by mass or 49 to 90% by mass based on the total mass of the adjuvant composition.
- the content of propylene glycol when the adjuvant composition contains glycerin is 8.0 to 79% by mass or 49 to 79% by mass based on the total mass of the adjuvant composition. May be.
- the content of glycerin is preferably 1.0 to 90% by mass, more preferably 1.0 to 80% by mass, and 8.0 to 60% by mass based on the total mass of the adjuvant composition. More preferably, the content is 16 to 50% by mass. By setting the glycerin content in such a range, lauryl alcohol can be more sufficiently dissolved. Within the above range, the glycerin content is 1.0 to 90% by mass, 1.0 to 80% by mass, 1.0 to 60% by mass, or 1.0 to 50% based on the total mass of the adjuvant composition. It may be mass%.
- the content of glycerin is 8.0 to 90% by mass, 8.0 to 80% by mass, 8.0 to 60% by mass, or 8.0 based on the total mass of the adjuvant composition. It may be up to 50% by weight. Further, within the above range, the glycerin content may be 16 to 90% by mass, 16 to 80% by mass, 16 to 60% by mass, or 16 to 50% by mass based on the total mass of the adjuvant composition. Good.
- the ratio of the content of propylene glycol to the content of lauryl alcohol is preferably 1.0 to 99, more preferably 2.8 to 89, and even more preferably 2.8 to 79.
- the ratio of the content of propylene glycol to the content of lauryl alcohol may be 1.0 to 99, 1.0 to 89, or 1.0 to 79.
- the ratio of the content of propylene glycol to the content of lauryl alcohol may be 2.8 to 99, 2.8 to 89, or 2.8 to 79.
- the ratio of the propylene glycol content to the glycerin content is preferably 0.4 to 99, more preferably 0.4 to 9.0, and 0.6 to 9.0. Is more preferable, and 1.0 to 4.0 is particularly preferable.
- the ratio of the content of propylene glycol to the content of glycerin may be 0.4 to 99, 0.4 to 9.0, or 0.4 to 4.0.
- the ratio of propylene glycol content to glycerin content may be 0.6 to 99, 0.6 to 9.0, or 0.6 to 4.0.
- the ratio of the propylene glycol content to the glycerin content may be 1.0 to 99, 1.0 to 9.0, or 1.0 to 4.0.
- the contents of lauryl alcohol, propylene glycol and glycerin in the adjuvant composition can be determined by collecting a predetermined amount of the adjuvant composition and quantifying it, for example, by gas chromatography.
- the measurement conditions may be set as follows.
- the adjuvant composition can contain other components as required.
- other components include aliphatic alcohols (excluding lauryl alcohol, glycerin and propylene glycol), fatty acid derivatives, free fatty acids, aliphatic ethers and the like.
- a compound represented by the following general formula (I) can be used as such a component.
- R 1 , R 2 and R 3 each independently represent H, OH, OCOR 4 ; n is 0, 1 or 2, and when n is 2, R 2 R 4 may be the same or different, and R 4 represents a linear or branched alkyl group having 1 to 3 carbon atoms, a linear or branched alkyl group having 2 to 3 carbon atoms, or a carbon number of 2 to Represents an alkynyl group of 3; However, R 1 , R 2 and R 3 are not simultaneously H, and when n is 1, two or more of R 1 , R 2 and R 3 are not simultaneously OH.
- aliphatic alcohol a linear or branched aliphatic alcohol can be used in addition to the aliphatic alcohol represented by the general formula (I), and a linear aliphatic alcohol is preferable.
- the aliphatic alcohol may be saturated or unsaturated, but is preferably a saturated aliphatic alcohol.
- the aliphatic alcohol is not limited in carbon number and molecular weight, but is more preferably an aliphatic alcohol having 8 to 20 carbon atoms. When the carbon number is in such a range, it is preferable from the viewpoint of skin permeability.
- aliphatic alcohol examples include octyldodecanol, oleyl alcohol, isostearyl alcohol, decanol and the like. Among these aliphatic alcohols, octyldodecanol and isostearyl alcohol are preferable.
- a compound containing a fatty acid moiety in addition to the fatty acid derivative represented by the above general formula (I), a compound containing a fatty acid moiety can be used.
- Fatty acid derivatives include fatty acid esters, fatty acid amides, fatty acid halides and the like.
- a fatty acid ester is preferable, and a fatty acid ester having a fatty acid carbon number of 10 to 20 and a fatty acid unsaturation degree of 0 or 1 and a monovalent fatty acid ester are more preferable.
- fatty acid ester examples include triacetin, sorbitan monolaurate, propylene glycol monolaurate, sorbitan monooleate, isopropyl myristate, polyethylene glycol, glycerol monooleate, cetyl palmitate, oleyl oleate and the like. Of these fatty acid esters, sorbitan monolaurate is most preferred.
- the free fatty acid a linear or branched free fatty acid can be used. Moreover, any of a saturated or unsaturated free fatty acid may be sufficient.
- the free fatty acid preferably has 8 to 20 carbon atoms. When the carbon number is in such a range, it is preferable from the viewpoint of skin permeability.
- free fatty acids examples include oleic acid, linoleic acid, ⁇ -linolenic acid, ⁇ -linolenic acid, lauric acid, stearic acid, and palmitic acid. Of these free fatty acids, oleic acid and lauric acid are preferred.
- the aliphatic ether may be any low molecular weight or high molecular weight ether compound.
- ether compounds include polyethylene glycol.
- the polyethylene glycol preferably has an average molecular weight of 200 to 4000, more preferably 200 to 1000.
- Such polyethylene glycol is commercially available, for example, under the trade name of Macrogol 400 (manufactured by Sanyo Chemical Industries, Ltd.).
- aliphatic alcohols fatty acid derivatives, free fatty acids and aliphatic ethers may be used alone or in combination of two or more.
- the above adjuvant composition can be used as an adjuvant preparation containing the same.
- the content of the adjuvant composition in the adjuvant preparation can be adjusted according to the purpose of use and the like, but it is preferable to contain the adjuvant composition at a high concentration.
- the content of the adjuvant composition can be 50 to 100% by mass based on the total mass of the adjuvant preparation, but is preferably 75 to 100% by mass, more preferably 85 to 100% by mass, More preferably, it is 95 to 100% by mass. By setting the content of the adjuvant composition to 50% by mass or more, the immunostimulatory effect of the adjuvant preparation can be further improved.
- the adjuvant preparation can contain other components depending on the purpose of use, the form of the preparation and the like.
- Other ingredients include thickeners, wetting agents, fillers, solubilizers, solubilizers, absorption enhancers, medicinal aids, stabilizers, antioxidants, emulsifiers, surfactants, crosslinkers, and polymerizers.
- the thickener is capable of stably holding water at 30 to 80% and having water retention.
- Thickeners include, for example, guar gum, locust bean gum, carrageenan, alginic acid, sodium alginate, agar, gum arabic (acacia gum), tragacanth gum, karaya gum, pectin, starch and other microbial systems such as xanthan gum, gelatin, collagen Natural polymers such as animal systems, cellulose systems such as methylcellulose, ethylcellulose, hydroxyethylcellulose, sodium carboxymethylcellulose, starch-based semisynthetic polymers such as soluble starch, carboxymethyl starch, and dialdehyde starch, polyvinyl alcohol, polyvinylpyrrolidone , Vinyls such as polyvinyl methacrylate, acrylics such as polyacrylic acid and sodium polyacrylate, polyethylene oxide, methyl vinyl ether / A water-soluble polymer such as a synthetic polymer water maleate
- sodium polyacrylate is preferred.
- Sodium polyacrylate has high gel strength and excellent water retention.
- Sodium polyacrylate having an average degree of polymerization of 20000 to 70000 is more preferred. When the average degree of polymerization is larger than 20000, the thickening effect becomes more sufficient and the gel strength tends to be improved. Moreover, when the average degree of polymerization is less than 70000, it is possible to suppress a decrease in workability due to an excessively thickening effect. Further, by using two or more kinds of the above thickeners in combination, for example, sodium polyacrylate forms a polymer complex with a strong ionic polymer, and an elastic gel with even higher gel strength can be obtained.
- the wetting agent polyhydric alcohols such as sorbitol can be used.
- the filler kaolin, zinc oxide, talc, titanium, bentonite, aluminum silicate, titanium oxide, zinc oxide, aluminum metasilicate, calcium sulfate, calcium phosphate and the like can be used.
- the total amount of the wetting agent and filler is preferably 0.1 to 30% by mass, more preferably 0.1 to 20% by mass, based on the total mass of the adjuvant preparation.
- solubilizer or absorption accelerator propylene carbonate, crotamiton, l-menthol, mint oil, limonene, diisopropyl adipate, and the like can be used.
- methyl salicylate As medicinal aids, use methyl salicylate, glycol salicylate, l-menthol, thymol, mint oil, nonyl acid vanillylamide, red pepper extract, aluminum hydroxide, aluminum phosphate, aluminum chloride, aluminum hydroxyphosphite sulfate, etc. Can do.
- any of a nonionic active agent and an ionic active agent can be used. From the viewpoint of safety, it is desirable to use a nonionic active agent usually used for a pharmaceutical base.
- examples of such surfactants include sugar alcohol fatty acid esters such as sucrose fatty acid esters, sorbitan fatty acid esters, glycerin fatty acid esters, polyglycerin fatty acid esters, propylene glycol fatty acid esters, polyoxyethylene sorbitan fatty acid esters, polyoxyethylene Examples include glycerin fatty acid ester, polyethylene glycol fatty acid ester, polyoxyethylene castor oil, and polyoxyethylene hydrogenated castor oil.
- the adjuvant preparation may contain a crosslinking agent, a polymerization agent and the like. By containing such components, the plaster can be strengthened and water retention can be imparted.
- a crosslinking agent and a polymerization agent are suitably selected according to kinds, such as a thickener.
- polyacrylic acid or polyacrylate in addition to a compound having at least two epoxy groups in the molecule, for example, hydrochlorides such as Ca, Mg, Al, sulfates, Inorganic acid salts such as phosphate and carbonate, organic acid salts such as citrate, tartrate, gluconate and stearate, oxides such as zinc oxide and anhydrous silicic acid, aluminum hydroxide, magnesium hydroxide, etc.
- a polyvalent metal compound such as a hydroxide is preferably used.
- adipic acid When polyvinyl alcohol is used as the thickener, adipic acid, thioglycolic acid, epoxy compound (epichlorohydrin), aldehydes, N-methylol compound, Al, Ti, Zr, Sn, V, Cu, B, Cr Etc. are preferably used.
- methyl vinyl ether / maleic anhydride copolymer methyl vinyl ether / maleic anhydride copolymer, polyacid compound or alkali metal salt thereof (polyacrylic acid, tannic acid and derivatives thereof), etc. are preferably used.
- polyethylene oxide used as the thickener
- peroxide, polysulfone azide and the like are preferably used.
- a methyl vinyl ether / maleic anhydride copolymer when used as the thickener, polyfunctional hydroxy compounds, polyamines, iodine, gelatin, polyvinyl pyrrolidone, iron, mercury, lead salts and the like are preferably used.
- aldehydes such as formaldehyde, glutaraldehyde, and dialdehyde starch
- diepoxides such as glyoxal and butadiene oxide
- diketones such as divinyl ketone
- diisocyanates are preferably used.
- a polyvalent metal salt such as lithium hydroxide, zinc hydroxide, aluminum hydroxide, sodium borate is preferably used as the cross-linking agent.
- zinc salts and aluminum salts are preferred. This is because the crosslinking reaction is promoted.
- the concentration of the polyvalent metal salt added as a crosslinking agent is preferably 0.5 to 1.5 equivalents per 1 equivalent of the thickener (or water-soluble polymer).
- an acrylic polymer or a rubber polymer is preferable.
- the acrylic polymer may be a copolymer containing at least one (meth) acrylic acid derivative typified by 2-ethylhexyl acrylate, methyl acrylate, butyl acrylate, hydroxyethyl acrylate, 2-ethylhexyl methacrylate and the like. Although not particularly limited, those containing 50% or more of 2-ethylhexyl acrylate are preferable.
- Specific examples of adhesives include acrylic acid / octyl acrylate copolymer and 2-ethylhexyl acrylate / vinyl pyrrolidone copolymer listed as adhesives in Pharmaceutical Additives Encyclopedia 2000 (edited by Japan Pharmaceutical Additives Association).
- the included adhesives such as acrylic polymers, DURO-TAK acrylic adhesive series (manufactured by Henkel), Eudragit series (Higuchi Shokai) and the like can be used.
- rubber polymers examples include styrene-isoprene-styrene block copolymer (hereinafter abbreviated as SIS), isoprene rubber, polyisobutylene (hereinafter abbreviated as PIB), styrene-butadiene-styrene block copolymer ( Hereinafter, abbreviated as SBS), styrene-butadiene rubber (hereinafter abbreviated as SBR), polysiloxane, and the like.
- SIS, PIB and polysiloxane are preferred, and SIS and PIB are more preferred.
- These pressure-sensitive adhesives may be used alone or in combination of two or more.
- the compounding amount of the pressure-sensitive adhesive is preferably 5 to 90% by mass, more preferably 10 to 70% by mass based on the total mass of the adjuvant preparation.
- the blending amount of the pressure-sensitive adhesive is within such a range, it is preferable from the viewpoint of permeability of the adjuvant preparation to the skin or mucous membrane, and when forming a patch, it is preferable from the viewpoint of forming the pressure-sensitive adhesive layer.
- tackifier resin examples include rosin derivatives (eg, rosin, glycerin ester of rosin, hydrogenated rosin, glycerin ester of hydrogenated rosin, pentaerythrester ester of rosin), alicyclic saturated hydrocarbon resins (eg, Alcon P100, Arakawa Chemical Industries), aliphatic hydrocarbon resins (for example, Quinton B170, Nippon Zeon), terpene resins (for example, Clearon P-125 Yasuhara Chemical), resin maleate and the like.
- glycerin ester of hydrogenated rosin, alicyclic saturated hydrocarbon resin, aliphatic hydrocarbon resin, and terpene resin are preferable.
- the blending amount of the tackifying resin is preferably 5 to 70% by mass based on the total mass of the adjuvant preparation in consideration of sufficient adhesive strength as a patch and irritation to the skin at the time of peeling. More preferably, it is ⁇ 60% by mass, and further preferably 10 ⁇ 50% by mass.
- Plasticizers include petroleum oils (eg liquid paraffin, paraffinic process oil, naphthenic process oil, aromatic process oil, etc.), squalane, squalene, vegetable oils (eg olive oil, camellia oil, castor oil, tall Oil, peanut oil, etc.), silicon oil, dibasic acid esters (eg, dibutyl phthalate, dioctyl phthalate, etc.), liquid rubber (eg, liquid polybutene, liquid isoprene rubber, etc.), liquid fatty acid esters (isopropyl myristate, lauric acid) Hexyl, diethyl sebacate, diisopropyl sebacate, etc.), diethylene glycol, polyethylene glycol, glycol salicylate, propylene glycol, dipropylene glycol, triacetin, triethyl citrate, crotamito And the like.
- petroleum oils eg liquid paraffin, paraffinic process oil, naphthenic process oil, aromatic process oil,
- plasticizers liquid paraffin, liquid polybutene, isopropyl myristate, hexyl laurate, and diethyl sebacate are preferable, and liquid paraffin, liquid polybutene, and isopropyl myristate are more preferable.
- plasticizers may be used individually by 1 type, or may be used in combination of 2 or more type.
- the blending amount of the plasticizer is preferably 10 to 70% by mass, more preferably 10 to 60% by mass, and further preferably 10 to 50% by mass based on the total mass of the adjuvant preparation.
- the blending amount of the plasticizer is within such a range, it is preferable from the viewpoint of permeability of the adjuvant preparation to the skin or mucous membrane, and when forming a patch, it is preferable because sufficient cohesive force can be maintained.
- Absorption accelerators include, for example, fatty acids having 6 to 20 carbon atoms, fatty alcohols, fatty acid esters, amides, ethers, aromatic organic acids, aromatic alcohols, aromatic organic acid esters or ethers (above saturated, Any of unsaturated, cyclic, linear or branched), lactic acid esters, acetic acid esters, monoterpene compounds, sesquiterpene compounds, Azone, Azone ) Derivatives, pyrothiodecane, glycerin fatty acid esters, propylene glycol fatty acid esters, sorbitan fatty acid esters (Span), polysorbate (Tween), polyethylene glycol fatty acid esters, polyoxyethylene hydrogenated castor oil (HCO), Polyoxyethylene alkyl ethers, ® sugar fatty acid esters, vegetable oils, and the like.
- absorption promoters include, for example, fatty acids having 6 to 20 carbon atoms, fatty alcohols, fatty acid esters, amides,
- the blending amount of the absorption enhancer is 0.01 to 40% by mass based on the total mass of the adjuvant preparation in consideration of sufficient skin permeability and irritation to the skin such as redness and edema. It is preferably 0.05 to 10% by mass, more preferably 0.1 to 5% by mass.
- the adjuvant preparation can also be used as various dosage forms, and can be the same dosage form as a conventionally used transdermal or transmucosal preparation.
- the dosage form of such an adjuvant preparation is preferably such that the adjuvant composition can be administered transcutaneously or transmucosally, and is an ointment, cream, gel, suppository, cataplasm, patch preparation, It is preferably a dosage form such as a lotion agent, liquid agent, impregnating agent or blister agent, more preferably a patch preparation or an impregnating agent dosage form, and even more preferably an impregnating agent dosage form.
- non-invasive administration in the body is possible as an external preparation adjuvant preparation.
- Patch preparations include matrix-type tape preparations, laminated-type tape preparations, and reservoir-type patch preparations. Of these, use as a matrix-type tape formulation and a reservoir-type patch formulation is preferred, and use as a reservoir-type patch formulation is more preferred.
- the matrix type tape preparation means a tape preparation having an adhesive layer in which a pharmacologically active substance is dispersed and contained in a base having adhesiveness.
- the matrix-type tape preparation includes a support on one side of the pressure-sensitive adhesive layer and a release liner on the other side.
- the base essentially comprises a rubbery (glassy) polymer or gel.
- Laminated tape formulation has a plurality of adhesive layers in which a pharmacologically active substance is dispersed and contained in an adhesive base of the tape formulation, and is supported on one side of the adhesive layer The body and the other surface are bonded with a release liner.
- a reservoir type patch preparation has a reservoir for storing a pharmacologically active substance, and has a backing member (support) impermeable to the drug on one side of the reservoir, and a release liner on the other side. Or what has a drug-permeable pressure-sensitive adhesive layer and a release liner.
- the impregnating agent is generally a preparation that covers the pad part with an adhesive cover material in a state where the pad is impregnated with a liquid agent containing an active ingredient.
- a support, an impervious backing member (film) for a liquid agent, an adhesive cover agent, a pad, a liner, and the like can be provided.
- the liquid agent, ointment or gel impregnated in the pad portion can be stably held.
- the liquid agent or the like may be stored in a blister container or the like and impregnated into the pad part at the time of administration to prepare the impregnating agent.
- natural members such as pulp, natural fabric members such as gauze and absorbent cotton, synthetic fiber fabric members such as polyester, polyethylene, and polyvinyl can be used. These members can be combined and processed into a woven fabric or a non-woven fabric.
- the adjuvant preparation may be applied to the skin or mucous membrane as the above-mentioned dosage form, but can also be administered to the skin or mucous membrane by microneedle puncture, needleless injection, skin polishing or mucosal polishing.
- an administration method of the adjuvant preparation a method in which at least a part of the microneedle is coated with the adjuvant preparation and administered by puncture may be used.
- the microneedles can be coated by the methods described in, for example, JP-T-2004-504120, JP-T-2004-528900, and International Publication No. 2005/016440.
- the administration method of the adjuvant preparation may be a method of administration by iontophoresis, sonophoresis or electroporation.
- An antigen is a substance that binds to an antigen receptor on an immune cell and induces an immune response, and the antigen is not particularly limited.
- polynucleotides DNA vaccines, RNA vaccines, protein-based vaccines and the like can be used.
- Such antigens include, for example, cytomegalovirus, hepatitis B virus, hepatitis C virus, human papilloma virus, rubella virus, varicella zoster, etc., attenuated or inactivated viruses such as pertussis, tetanus , Diphtheria, group A streptococcus, Legionella pneumophila, meningococcus, Pseudomonas aeruginosa, Streptococcus pneumoniae, syphilis treponema, cholera, etc., attenuated or inactivated bacteria, proteins, polysaccharides , Oligosaccharides, lipoproteins, and mixtures thereof.
- viruses such as pertussis, tetanus , Diphtheria, group A streptococcus, Legionella pneumophila, meningococcus, Pseudomonas aeruginosa, Streptococcus
- vaccines containing antigenic agents can also be used.
- examples of such vaccines include influenza vaccine, Lyme disease vaccine, rabies vaccine, measles vaccine, epidemic parotitis vaccine, varicella vaccine, smallpox vaccine, hepatitis vaccine, pertussis vaccine, diphtheria vaccine and the like.
- antigens used in vaccine therapy for cancer, arteriosclerosis, neurological disease, Alzheimer and the like can also be used.
- an allergen substance having antigenicity may be used as an antigen.
- allergen materials include a wide variety of metals and chemical substances.
- antigens recognized by inflammatory T cells associated with T cell mediated autoimmune diseases or conditions include a wide variety of T cell mediated autoimmune diseases or conditions.
- the antigen administration method is not particularly limited, and oral, injection (intramuscular, subcutaneous, intradermal), administration methods such as microneedle puncture, transdermal or transmucosal administration methods, and the like can be used.
- a transdermal administration means is selected according to the skin permeability of the antigen and the required dosage. If the antigen can be administered transdermally, a transdermal non-invasive preparation containing the adjuvant composition and the antigen can be formed. If the antigen does not have sufficient transdermal or transmucosal activity, it may be administered non-transdermally or non-mucosally, for example, administration by injection or oral administration is conceivable. In the case of administration by injection, the adjuvant composition and the antigen may be administered simultaneously.
- the administration method of the adjuvant preparation is an adjuvant preparation (particularly preferably an impregnating agent or the like) before or after administration of the antigen non-transcutaneously or non-mucosally (injection, microneedle puncture, etc.). It is preferred to administer a patch formulation). More preferably, the adjuvant preparation is administered after the antigen is administered transdermally or non-mucosally, and the adjuvant preparation is administered immediately after the antigen is administered non-transdermally or non-mucosally. More preferred. In such a case, administration of the adjuvant preparation (preferably administration by sticking) can be continued while administering the antigen. For example, an adjuvant preparation can be separately administered by sticking or the like while the antigen is administered by microneedle puncture or the like.
- the sticking time is not particularly limited as long as the adjuvant preparation can sufficiently permeate the skin or mucous membrane and exert its effect, but it is 0.1 to 96 hours. It is preferably 0.5 to 48 hours, more preferably 2 to 24 hours. Within the above range, the sticking time may be 0.1 to 96 hours, 0.5 to 96 hours, 2 to 96 hours, 4 to 96 hours, or 6 to 96 hours. Further, within the above range, the sticking time may be 0.1 to 48 hours, 0.5 to 48 hours, 2 to 48 hours, 4 to 48 hours, or 6 to 48 hours. Further, within the above range, the sticking time may be 0.1 to 12 hours, 0.5 to 12 hours, 2.0 to 12 hours, or 6 to 12 hours. Of these, the lower limit value of the sticking time is more preferably set longer, and the upper limit value of the sticking time is more preferably set shorter.
- the blending ratio can be appropriately determined depending on the combination of the antigen and the adjuvant. It is preferable to use it in a formulation that results in a high concentration of adjuvant.
- an immunostimulation method or immunomodulation method using the adjuvant composition or adjuvant preparation according to the present embodiment is excellent in increasing the immunogenicity of the antigen and increasing the antibody titer.
- the method includes administering the above-mentioned adjuvant composition or the above-mentioned adjuvant preparation to an individual in need of promoting an immune response, transdermally or transmucosally.
- An immunostimulation method is provided.
- the individual who needs to promote immune response includes, for example, an individual who needs prevention by antigen administration, or an individual who suffers from a disease or disease that needs treatment by antigen administration.
- the individual may be a human.
- the antigen includes viruses, bacteria, and the like that can be used as the antigens described above.
- the immunostimulation method or immunomodulation method can also be referred to as a method for prevention or treatment depending on the subject to which it is applied. For example, when applied to an individual who needs to promote an immune response in order to prevent the onset of a viral infection, the above-described immunostimulation method or immunomodulation method can also be referred to as a viral infection prevention method.
- the adjuvant composition or adjuvant preparation can be administered in an effective amount and an effective time and route for inducing a desired immune response or immune reaction promoting effect.
- an effective amount of an adjuvant composition or adjuvant formulation may be administered as a single dose or divided into multiple doses.
- transdermal or transmucosal administration can be performed 1 to 5 times per day, or 1 to 3 times per day, or 1 to 2 times per day.
- the administration of the adjuvant composition or the adjuvant preparation may be performed simultaneously with the administration of the antigen, or may be performed with a time difference from the administration of the antigen.
- the administration of the adjuvant composition or the adjuvant preparation is preferably performed by a route different from the administration of the antigen.
- the site to which the adjuvant composition or adjuvant preparation is administered may be the same as or different from the site or region to which the antigen is applied.
- the administration form of the adjuvant composition or adjuvant preparation and the administration form of the antigen for example, subcutaneous injection or microneedle puncture administration of the antigen, transdermal or transmucosal of the adjuvant composition, etc.
- a combination with administration is preferred.
- a combination of microneedle puncture administration of an antigen and transdermal administration of an adjuvant composition or the like is more preferable.
- the adjuvant composition of the present invention can be applied while the administration device is applied.
- the adjuvant composition or adjuvant preparation can be administered to intact skin or intact mucous membrane.
- the physical or chemical treatment is, for example, at least selected from the group consisting of heat treatment (thermal treatment), ultrasonic treatment, electric field treatment, magnetic field treatment, pressure treatment, alkali treatment, laser irradiation, polishing treatment, and microneedle.
- heat treatment thermal treatment
- ultrasonic treatment electric field treatment
- magnetic field treatment magnetic field treatment
- pressure treatment alkali treatment
- laser irradiation polishing treatment
- microneedle microneedle.
- One treatment is preferred.
- the response can be completed. It is preferably applied by at least one of lamella structure change, hydration, denaturation, pore formation, exfoliation or bypass formation in the stratum corneum of the skin.
- administration of the adjuvant preparation is accompanied by lamella structure change, hydration, denaturation, pore formation, exfoliation or bypass formation of the stratum corneum, the percutaneous absorption of the adjuvant preparation can be further promoted.
- the adjuvant composition or adjuvant preparation shown above can also be used in a kit for an immunostimulation method.
- a kit may be provided with an adjuvant composition or an adjuvant preparation, and may further include an antigen or a device for administering the antigen.
- the device for antigen administration may be, for example, an administration device such as a microneedle or a syringe.
- kits comprising a microneedle coated with an antigen on at least a part of the surface and a patch preparation impregnated with an adjuvant composition can be used.
- the skin or mucosal Langerhans cells are activated and are present in the lymph node from the skin or mucosa. It is considered that a high immune response is completed by efficiently transmitting a signal to TH cells. This makes it possible to easily evaluate the antigenicity of external medicines, cosmetics or allergens, prevent or treat with vaccines for infectious diseases, cancer, allergies, etc., and treat T cell-mediated autoimmune diseases.
- the adjuvant composition and the adjuvant preparation according to the present embodiment greatly contribute to the development of the pharmaceutical industry and related industries.
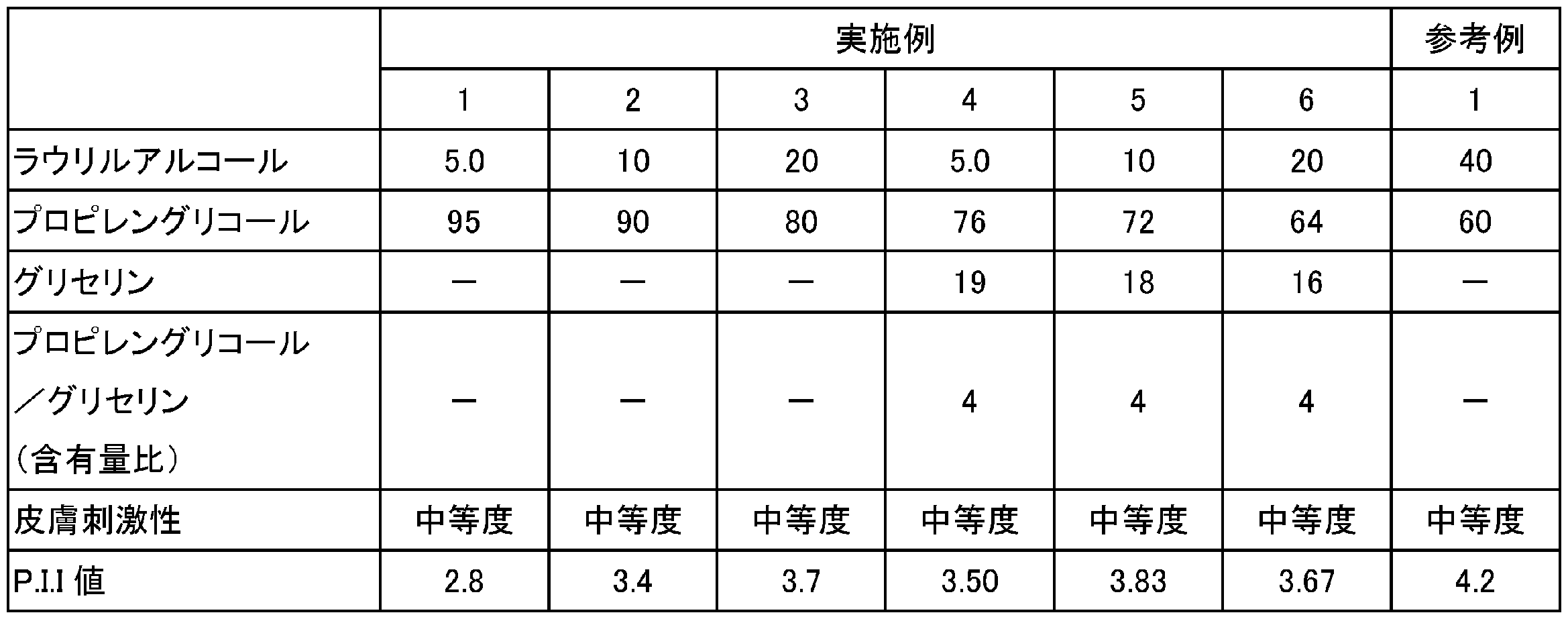
- Example 1 Adjuvant compositions containing lauryl alcohol, propylene glycol and glycerin were prepared at the blending amounts (mass%) shown in Table 2. A sample for skin irritation test was prepared by impregnating 120 ⁇ L of the obtained adjuvant composition into a lint cloth (manufactured by White Cross Co., Ltd., cut into a 2 cm 2 circle).
- the sample for skin irritation test was affixed to the back skin of a rabbit for 24 hours. After peeling the sample, the skin reaction after 0.5 hours, 24 hours and 48 hours was observed and evaluated according to the Draize standard (Draize standard). For the evaluation results, the same sample was tested on 2 to 6 birds, and the average value of the results was used. The evaluation results are shown in Table 2.
- the dreed criterion is a criterion adopted when evaluating skin reaction.
- the skin condition of the test object is visually observed, and the skin condition of the test object is scored based on the following scoring criteria.
- PII value skin primary irritation index
- Example 2 to 6 Reference Example 1 and Comparative Examples 1 to 6
- An adjuvant composition was prepared and an evaluation sample was prepared in the same manner as in Example 1 except that the blending amounts of lauryl alcohol, propylene glycol and glycerin were changed to the blending amounts shown in Table 2 or Table 3.
- the skin irritation test was performed on the obtained evaluation sample in the same manner as in Example 1.
- the results are shown in Tables 2 and 3. From Table 2 and Table 3, the tendency for skin irritation to become intensity
- Adjuvant compositions containing lauryl alcohol and propylene glycol were prepared at the blending amounts (mass%) shown in Table 4.
- a sample for evaluation was prepared by impregnating 120 ⁇ L of the obtained adjuvant composition into an impregnation tape.
- the impregnation tape used was a laminate of lint cloth (manufactured by White Cross Co., Ltd., cut into 2 cm 2 ) and acrylic tape.
- Ovalbumin (OVA) antigen was administered to the abdomen of female 7-week-old BALB-C mice, and then the adjuvant composition was administered transdermally.
- OVA antigen 0.1 mg was administered by intradermal injection.
- the administration was performed by preparing an aqueous solution using physiological saline so as to be 0.1 ⁇ g / 50 ⁇ L.
- the adjuvant composition was administered by applying the sample for evaluation to the skin of a mouse for 6 hours. All the samples for evaluation were fixed with Coban and Skinnagate. In addition to the initial administration, the administration of the antigen and the adjuvant composition was performed after 2 weeks. In order to evaluate the OVA-specific IgG antibody titer, blood was collected after 4 weeks. The OVA specific IgG antibody titer was measured by ELISA. The results are shown in Table 3.
- Example 8 to 19 Reference Example 2, and Comparative Examples 7 to 10.
- An adjuvant composition was prepared and an evaluation sample was prepared in the same manner as in Example 1 except that the blending amounts of lauryl alcohol, propylene glycol and glycerin were changed to the blending amounts shown in Table 4 or Table 5.
- the adjuvant composition of the present invention is superior to the conventional adjuvant composition in terms of safety and immune response enhancement effect.
- the adjuvant composition of the present invention does not require mixed administration with an antigen as is generally done with other adjuvant adjuvant compositions, and can be administered separately from the antigen.
- it is excellent in that the desired effect can be obtained by administering an adjuvant preparation containing a high concentration of the adjuvant composition to the skin or mucous membrane by applying or applying it after the antigen is administered.
- the skin irritation was further suppressed as compared with the conventional impregnating agent, and the increase in the immune IgG antibody titer could be achieved safely.
Abstract
Description
装置:株式会社島津製作所製 GC-2010
検出器:株式会社島津製作所製 水素炎イオン化検出器 FID-2010
カラム:Agilent社製 GCカラム DB-1
キャリアガス:He
CH2R1(CHR2)nCH2R3 (I)
プロピレングリコール単独、グリセリン単独又はプロピレングリコール及びグリセリンの混合溶媒に対するラウリルアルコールの溶解性について評価した。表1に示す配合量となるようにラウリルアルコールを溶媒に添加し、20℃における溶解性を下記基準で評価した。結果を表1に示す。
A:完全に溶解した場合
B:半透明ではあるが均一となっている場合
C:分離している場合
表2に示す配合量(質量%)で、ラウリルアルコール、プロピレングリコール及びグリセリンを含むアジュバント組成物を調製した。得られたアジュバント組成物120μLをリント布(白十字社製、2cm2の円形に裁断)に含浸させたものを皮膚刺激性試験用サンプルとした。
雌性ウサギ(JW)(SPF)の背部に刈毛及び剃毛処置を施した。2週間の馴化期間後、一般状態及び皮膚状態が良好なウサギを選択して試験を実施した。
<紅斑及び痂皮形成について>
0点:紅斑なし。
1点:非常に軽度の紅斑が認められる(かろうじて識別できる)。
2点:はっきりとした紅斑が認められる。
3点:中程度ないし高度の紅斑が認められる。
4点:高度紅斑(ビートの赤色)からわずかな痂皮(深部損傷)の形成が認められる。
<浮腫の形成について>
0点:浮腫なし。
1点:非常に軽度の紅斑が認められる(かろうじて識別できる)。
2点:はっきりとした紅斑が認められる。
3点:中程度ないし高度の紅斑が認められる。
4点:高度紅斑(ビートの赤色)からわずかな痂皮(深部損傷)の形成が認められる。
<ドレイズ基準>
軽度刺激物 :P.I.I.≦2
中等度刺激物:2<P.I.I.≦5
強度刺激物 :5<P.I.I.≦8
ラウリルアルコール、プロピレングリコール及びグリセリンの配合量を表2又は表3に示す配合量に変更した以外は、実施例1と同様にして、アジュバント組成物を調製し、評価用サンプルを作製した。
表4に示す配合量(質量%)で、ラウリルアルコール、及びプロピレングリコールを含むアジュバント組成物を調製した。得られたアジュバント組成物120μLを含浸用テープに含浸させたものを評価用サンプルとした。なお、含浸用テープは、リント布(白十字社製、2cm2に裁断)とアクリルテープとを積層したものを用いた。
得られたアジュバント組成物について、マウス免疫試験を実施した。雌性7週齢のBALB-Cマウスの腹部に、Ovalbumin(OVA)抗原を投与し、その後、アジュバント組成物を経皮投与した。
ラウリルアルコール、プロピレングリコール及びグリセリンの配合量を表4又は表5に示す配合量に変更した以外は、実施例1と同様にして、アジュバント組成物を調製し、評価用サンプルを作製した。
Claims (16)
- ラウリルアルコール及びプロピレングリコールを含有するアジュバント組成物であって、
ラウリルアルコール及びプロピレングリコールの含有量が、アジュバント組成物全質量基準で、それぞれ0.5~25質量%及び8.0~99.5質量%であり、
ラウリルアルコールが溶解しており、
経皮又は経粘膜投与用である、アジュバント組成物。 - グリセリンを更に含有し、
ラウリルアルコール、プロピレングリコール及びグリセリンの含有量が、アジュバント組成物全質量基準で、それぞれ、0.5~25質量%、8.0~90質量%及び1.0~90質量%である、請求項1に記載のアジュバント組成物。 - ラウリルアルコールの含有量に対するプロピレングリコールの含有量の比が、1.0~99である、請求項1又は2に記載のアジュバント組成物。
- グリセリンの含有量に対するプロピレングリコールの含有量の比が、0.4~99である、請求項1~3のいずれか一項に記載のアジュバント組成物。
- 請求項1~4のいずれか一項に記載のアジュバント組成物を含む、アジュバント製剤。
- アジュバント組成物の含有量が、アジュバント製剤全質量基準で50~100質量%である、請求項5に記載のアジュバント製剤。
- 軟膏剤、クリーム剤、ゲル剤、座剤、パップ剤、パッチ製剤、ローション剤、液剤、含浸剤又はブリスター剤である、請求項5又は6に記載のアジュバント製剤。
- パッチ製剤が、マトリックス型のテープ製剤、積層型のテープ製剤又はリザーバー型のパッチ製剤である、請求項7に記載のアジュバント製剤。
- マイクロニードル穿刺、無針注射、皮膚研磨又は粘膜研磨により、皮膚又は粘膜に投与される、請求項5~8のいずれか一項に記載のアジュバント製剤。
- マイクロニードルの少なくとも一部にコーティングされ、穿刺より、皮膚又は粘膜に投与される、請求項5~9のいずれか一項に記載のアジュバント製剤。
- イオントフォレーシス、ソノフォレーシス又はエレクトロポレーションにより投与される、請求項5~8のいずれか一項に記載のアジュバント製剤。
- 無傷の皮膚、無傷の粘膜、物理的若しくは化学的な処理が施された皮膚又は物理的若しくは化学的な処理が施された粘膜に対して投与される、請求項5~11のいずれか一項に記載のアジュバント製剤。
- 物理的若しくは化学的な処理が、熱処理、超音波処理、電場処理、磁場処理、圧力処理、アルカリ処理、レーザー照射、研磨処理及びマイクロニードルによる処理からなる群より選択される少なくとも1つの処理である、請求項12に記載のアジュバント製剤。
- 抗原の投与前又は抗原の投与後に、皮膚又は粘膜に投与される、請求項5~13のいずれか一項に記載のアジュバント製剤。
- 請求項1~4のいずれか一項に記載のアジュバント組成物、又は請求項5~14のいずれか一項に記載のアジュバント製剤を備える、キット。
- 抗原又は抗原投与のための器具を更に備える、請求項15に記載のキット。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP14858381.8A EP3064218B1 (en) | 2013-10-31 | 2014-10-30 | Adjuvant composition |
| US15/032,718 US9993549B2 (en) | 2013-10-31 | 2014-10-30 | Adjuvant composition, adjuvant preparation containing same, and kit |
| ES14858381T ES2744454T3 (es) | 2013-10-31 | 2014-10-30 | Composición de adyuvante |
| JP2015545304A JP6114401B2 (ja) | 2013-10-31 | 2014-10-30 | アジュバント組成物、これを含むアジュバント製剤、及びキット |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013227166 | 2013-10-31 | ||
| JP2013-227166 | 2013-10-31 | ||
| JP2014-008089 | 2014-01-20 | ||
| JP2014008089 | 2014-01-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015064710A1 true WO2015064710A1 (ja) | 2015-05-07 |
Family
ID=53004307
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/078958 WO2015064710A1 (ja) | 2013-10-31 | 2014-10-30 | アジュバント組成物、これを含むアジュバント製剤、及びキット |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US9993549B2 (ja) |
| EP (1) | EP3064218B1 (ja) |
| JP (1) | JP6114401B2 (ja) |
| ES (1) | ES2744454T3 (ja) |
| TW (1) | TWI642443B (ja) |
| WO (1) | WO2015064710A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016222582A (ja) * | 2015-05-29 | 2016-12-28 | ライオン株式会社 | 口腔用組成物 |
| CN108778365A (zh) * | 2016-01-11 | 2018-11-09 | 沃达瑞公司 | 微针组合物及其使用方法 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004504120A (ja) | 2000-07-21 | 2004-02-12 | グラクソスミスクライン バイオロジカルズ ソシエテ アノニム | ワクチン |
| JP2004528900A (ja) | 2001-04-20 | 2004-09-24 | アルザ・コーポレーシヨン | 有益な作用物質を含有するコーティングを有する微小突起アレイ |
| WO2005016440A1 (en) | 2003-08-04 | 2005-02-24 | Alza Corporation | Method and device for enhancing transdermal agent flux |
| WO2007015441A1 (ja) | 2005-08-01 | 2007-02-08 | Hisamitsu Pharmaceutical Co., Inc. | 経皮または経粘膜投与のためのアジュバントおよび製剤 |
| WO2008093772A1 (ja) | 2007-01-31 | 2008-08-07 | Hisamitsu Pharmaceutical Co., Inc. | 経皮または経粘膜投与のためのアジュバントおよびこれを含む医薬製剤 |
| WO2010001671A1 (ja) * | 2008-06-30 | 2010-01-07 | 久光製薬株式会社 | マイクロニードルデバイスおよびマイクロニードルデバイスによるインフルエンザワクチンの奏功性を上昇させる方法 |
| WO2010013601A1 (ja) * | 2008-07-30 | 2010-02-04 | 久光製薬株式会社 | マイクロニードルデバイスおよびマイクロニードルデバイスによる日本脳炎ウイルス抗原の奏功性を上昇させる方法 |
| WO2012115222A1 (ja) | 2011-02-25 | 2012-08-30 | 久光製薬株式会社 | 経皮または経粘膜投与のためのアジュバントおよびこれを含む医薬製剤 |
Family Cites Families (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4590190A (en) | 1983-07-01 | 1986-05-20 | Nitto Electric Industrial Co., Ltd. | Method for percutaneously administering physiologically active agents using an alcohol adjuvant and a solvent |
| US4605670A (en) | 1984-02-01 | 1986-08-12 | Nitto Electric Industrial Co., Ltd. | Method for percutaneously administering metoclopramide |
| DE3587481T2 (de) | 1984-02-27 | 1993-12-16 | Toshiba Kawasaki Kk | Schaltungssubstrat mit hoher Wärmeleitfähigkeit. |
| US4863970A (en) | 1986-11-14 | 1989-09-05 | Theratech, Inc. | Penetration enhancement with binary system of oleic acid, oleins, and oleyl alcohol with lower alcohols |
| WO1988007814A1 (en) * | 1987-04-16 | 1988-10-20 | E.I. Du Pont De Nemours And Company | Fungicide compositions |
| JP2913229B2 (ja) | 1991-10-21 | 1999-06-28 | 塩野義製薬株式会社 | 油性アジュバントワクチン製剤 |
| JPH05255112A (ja) | 1992-03-14 | 1993-10-05 | Kanebo Ltd | アジュバント組成物 |
| ES2137993T3 (es) | 1992-06-11 | 2000-01-01 | Theratech Inc | La utilizacion de glicerina para moderar el aporte transdermico de medicamentos. |
| DK17093D0 (da) | 1993-02-15 | 1993-02-15 | Lyfjathroun H F | Farmaceutisk praeparat til topisk administrering af antigener og/eller vacciner til pattedyr via slimhinder |
| US5376397A (en) * | 1993-05-06 | 1994-12-27 | Kraft General Foods, Inc. | Microemulsions of oil and water |
| WO1995022989A1 (en) | 1994-02-24 | 1995-08-31 | Micro-Pak, Inc. | Vaccines containing paucilamellar lipid vesicles as immunological adjuvants |
| US20010006645A1 (en) | 1996-10-28 | 2001-07-05 | Jane A. Norton | Method for transdermal drug delivery |
| AU6046198A (en) | 1997-01-31 | 1998-08-25 | Kenneth Hoyt | Use of oil from emu or rhea birds as trans-membrane carriers for delivery of drugs, peptides and vaccines |
| US6818222B1 (en) | 1997-03-21 | 2004-11-16 | Chiron Corporation | Detoxified mutants of bacterial ADP-ribosylating toxins as parenteral adjuvants |
| IS4518A (is) | 1997-07-09 | 1999-01-10 | Lyfjathroun Hf, The Icelandic Bio Pharmaceutical Group | Nýtt lyfjaform fyrir bóluefni |
| US20030045492A1 (en) | 1997-08-13 | 2003-03-06 | Tang De-Chu C. | Vaccination by topical application of recombinant vectors |
| AU765260B2 (en) | 1998-04-20 | 2003-09-11 | Torrey Pines Institute For Molecular Studies | Topical immunostimulation to induce langerhans cell migration |
| CN1191872C (zh) | 1999-01-28 | 2005-03-09 | 塞托·帕尔斯科技公司 | 运送大分子进入细胞的装置 |
| MXPA02003067A (es) | 1999-09-24 | 2002-09-30 | Smithkline Beecham Biolog | Uso de combinacion de ester sorbitano de polioxietileno y octoxinol como adjuvante y su uso en vacunas. |
| DE10019171A1 (de) | 2000-04-07 | 2001-10-18 | Schering Ag | Zusammensetzungen zur Verwendung als Penetrationsverstärker in transdermalen Formulierungen für hoch lipophile Wirkstoffe |
| KR100452972B1 (ko) * | 2000-05-16 | 2004-10-14 | 주식회사 삼양사 | 경피투여용 하이드로젤 조성물 |
| US7560484B2 (en) | 2000-08-31 | 2009-07-14 | The Kitasato Institute | Vaccine preparation containing fatty acid as a constituent |
| CA2423610A1 (en) | 2000-10-02 | 2002-04-11 | Glaxosmithkline Biologicals S.A. | Split enveloped virus preparation |
| AU2002252378B2 (en) | 2001-03-19 | 2007-10-18 | Intercell Usa, Inc. | Transcutaneous immunostimulation |
| US20040109869A1 (en) | 2001-03-19 | 2004-06-10 | Iomai Corporation | Transcutaneous immunostimulation |
| IL142536A0 (en) | 2001-04-11 | 2002-03-10 | Yeda Res & Dev | Carriers for therapeutic preparations for treatment of t-cell mediated diseases |
| IL142535A0 (en) | 2001-04-11 | 2002-03-10 | Yeda Res & Dev | Pharmaceutical compositions for the treatment of inflammation |
| US20020193729A1 (en) | 2001-04-20 | 2002-12-19 | Cormier Michel J.N. | Microprojection array immunization patch and method |
| WO2003018051A1 (en) | 2001-08-27 | 2003-03-06 | Vic Jira | Anti-fungal composition |
| WO2003070272A1 (en) | 2002-02-25 | 2003-08-28 | Lyfjathroun Hf, Biopharmaceutical Research | An immunological adjuvant |
| JP2005526063A (ja) | 2002-03-12 | 2005-09-02 | ガルデルマ・リサーチ・アンド・デヴェロップメント・エス・エヌ・セ | 皮膚疾患を治療するためのアダパレンの使用 |
| JP4213432B2 (ja) | 2002-08-28 | 2009-01-21 | 久光製薬株式会社 | 貼付剤 |
| US20070292461A1 (en) * | 2003-08-04 | 2007-12-20 | Foamix Ltd. | Oleaginous pharmaceutical and cosmetic foam |
| AU2004285484A1 (en) | 2003-10-24 | 2005-05-12 | Alza Corporation | Pretreatment method and system for enhancing transdermal drug delivery |
| AU2004292953A1 (en) | 2003-11-21 | 2005-06-09 | Alza Corporation | Ultrasound assisted transdermal vaccine delivery method and system |
| EP1708742A4 (en) | 2003-12-05 | 2008-11-05 | Becton Dickinson Co | METHOD FOR IMPROVING THE IMMUNE RESPONSE IN THE INTRADERMAL COMPARTMENT AND USEFUL LINKS THEREFOR |
| WO2010125470A2 (en) | 2009-04-28 | 2010-11-04 | Foamix Ltd. | Foamable vehicle and pharmaceutical compositions comprising aprotic polar solvents and uses thereof |
| KR101759354B1 (ko) | 2009-06-10 | 2017-07-18 | 히사미쓰 세이야꾸 가부시키가이샤 | 마이크로니들 디바이스 |
| CA2790923C (en) | 2010-02-24 | 2017-10-31 | Toshiyuki Matsudo | Micro-needle device and preparation method |
| US20130323293A1 (en) | 2011-02-24 | 2013-12-05 | Takuya Umemoto | Glp-1 analogue composition for microneedle devices |
-
2014
- 2014-10-30 US US15/032,718 patent/US9993549B2/en active Active
- 2014-10-30 WO PCT/JP2014/078958 patent/WO2015064710A1/ja active Application Filing
- 2014-10-30 EP EP14858381.8A patent/EP3064218B1/en active Active
- 2014-10-30 ES ES14858381T patent/ES2744454T3/es active Active
- 2014-10-30 JP JP2015545304A patent/JP6114401B2/ja active Active
- 2014-10-31 TW TW103137801A patent/TWI642443B/zh active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004504120A (ja) | 2000-07-21 | 2004-02-12 | グラクソスミスクライン バイオロジカルズ ソシエテ アノニム | ワクチン |
| JP2004528900A (ja) | 2001-04-20 | 2004-09-24 | アルザ・コーポレーシヨン | 有益な作用物質を含有するコーティングを有する微小突起アレイ |
| WO2005016440A1 (en) | 2003-08-04 | 2005-02-24 | Alza Corporation | Method and device for enhancing transdermal agent flux |
| WO2007015441A1 (ja) | 2005-08-01 | 2007-02-08 | Hisamitsu Pharmaceutical Co., Inc. | 経皮または経粘膜投与のためのアジュバントおよび製剤 |
| WO2008093772A1 (ja) | 2007-01-31 | 2008-08-07 | Hisamitsu Pharmaceutical Co., Inc. | 経皮または経粘膜投与のためのアジュバントおよびこれを含む医薬製剤 |
| WO2010001671A1 (ja) * | 2008-06-30 | 2010-01-07 | 久光製薬株式会社 | マイクロニードルデバイスおよびマイクロニードルデバイスによるインフルエンザワクチンの奏功性を上昇させる方法 |
| WO2010013601A1 (ja) * | 2008-07-30 | 2010-02-04 | 久光製薬株式会社 | マイクロニードルデバイスおよびマイクロニードルデバイスによる日本脳炎ウイルス抗原の奏功性を上昇させる方法 |
| WO2012115222A1 (ja) | 2011-02-25 | 2012-08-30 | 久光製薬株式会社 | 経皮または経粘膜投与のためのアジュバントおよびこれを含む医薬製剤 |
Non-Patent Citations (2)
| Title |
|---|
| GHUMMAN B. ET AL.: "Chemical chaperones enhance superantigen and conventional antigen presentation by HLA-DM-deficient as well as HLA-DM-sufficient antigen-presenting cells and enhance IgG2a production in vivo.", JOURNAL OF IMMUNOLOGY, vol. 161, no. 7, 1998, pages 3262 - 3270, XP055120048 * |
| STOJANOVIC M. ET AL.: "The context of tetanus toxoid application influences the outcome of antigen-specific and self-directed humoral immune response.", MICROBIOLOGY AND IMMUNOLOGY, vol. 53, no. 2, 2009, pages 89 - 100, XP055120052 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016222582A (ja) * | 2015-05-29 | 2016-12-28 | ライオン株式会社 | 口腔用組成物 |
| CN108778365A (zh) * | 2016-01-11 | 2018-11-09 | 沃达瑞公司 | 微针组合物及其使用方法 |
| CN108778365B (zh) * | 2016-01-11 | 2021-05-25 | 沃达瑞公司 | 微针组合物及其使用方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3064218A1 (en) | 2016-09-07 |
| EP3064218A4 (en) | 2017-04-05 |
| TWI642443B (zh) | 2018-12-01 |
| EP3064218B1 (en) | 2019-06-19 |
| US9993549B2 (en) | 2018-06-12 |
| US20160263215A1 (en) | 2016-09-15 |
| ES2744454T3 (es) | 2020-02-25 |
| JPWO2015064710A1 (ja) | 2017-03-09 |
| TW201609143A (zh) | 2016-03-16 |
| JP6114401B2 (ja) | 2017-04-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5876468B2 (ja) | 経皮または経粘膜投与のためのアジュバントおよびこれを含む医薬製剤 | |
| JP5006196B2 (ja) | 経皮または経粘膜投与のためのアジュバントおよび製剤 | |
| US20090136480A1 (en) | Transcutaneous immunostimulation | |
| JP5275047B2 (ja) | 経皮または経粘膜投与のためのアジュバントおよびこれを含む医薬製剤 | |
| US20070292445A1 (en) | Delivery system for transdermal immunization | |
| JP5403948B2 (ja) | メマンチン含有経皮吸収製剤 | |
| TWI501790B (zh) | Contains percutaneous absorption preparations from Donepezil | |
| JP2004529906A (ja) | 経皮的免疫賦活 | |
| WO2005077364A1 (ja) | ソリフェナシンの経皮投与製剤およびその経皮透過改善方法 | |
| JP6114401B2 (ja) | アジュバント組成物、これを含むアジュバント製剤、及びキット | |
| JP6457486B2 (ja) | 貼付製剤 | |
| JPWO2015025718A1 (ja) | 貼付剤 | |
| EP3178495B1 (en) | Liquid immunity induction-promoting composition and vaccine pharmaceutical composition that include thrombosis treatment drug | |
| JP6352718B2 (ja) | アジュバント組成物及びアジュバント製剤 | |
| JP6980360B2 (ja) | ビスホスホネート剤を含む液性免疫用ワクチン医薬組成物 | |
| WO2015056710A1 (ja) | 経皮免疫製剤 | |
| Seid Jr et al. | Advances in transcutaneous vaccine delivery | |
| JPWO2011096566A1 (ja) | 爪白癬治療用貼付剤 | |
| IL180509A (en) | Skin Transmission Transmission System | |
| JP2010284176A (ja) | 製剤、当該製剤を用いたワクチンの投与方法及びイオントフォレーシス装置 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14858381 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2015545304 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15032718 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2014858381 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014858381 Country of ref document: EP |