WO2013147139A1 - 生分解性ポリエステル樹脂組成物 - Google Patents
生分解性ポリエステル樹脂組成物 Download PDFInfo
- Publication number
- WO2013147139A1 WO2013147139A1 PCT/JP2013/059493 JP2013059493W WO2013147139A1 WO 2013147139 A1 WO2013147139 A1 WO 2013147139A1 JP 2013059493 W JP2013059493 W JP 2013059493W WO 2013147139 A1 WO2013147139 A1 WO 2013147139A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- p3ha
- resin composition
- weight
- film
- polyester resin
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L67/00—Compositions of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Compositions of derivatives of such polymers
- C08L67/02—Polyesters derived from dicarboxylic acids and dihydroxy compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/18—Manufacture of films or sheets
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L67/00—Compositions of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Compositions of derivatives of such polymers
- C08L67/04—Polyesters derived from hydroxycarboxylic acids, e.g. lactones
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2300/00—Characterised by the use of unspecified polymers
- C08J2300/16—Biodegradable polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2367/00—Characterised by the use of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Derivatives of such polymers
- C08J2367/02—Polyesters derived from dicarboxylic acids and dihydroxy compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2367/00—Characterised by the use of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Derivatives of such polymers
- C08J2367/04—Polyesters derived from hydroxy carboxylic acids, e.g. lactones
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2467/00—Characterised by the use of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Derivatives of such polymers
- C08J2467/02—Polyesters derived from dicarboxylic acids and dihydroxy compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2467/00—Characterised by the use of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Derivatives of such polymers
- C08J2467/04—Polyesters derived from hydroxy carboxylic acids, e.g. lactones
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/10—Esters; Ether-esters
- C08K5/101—Esters; Ether-esters of monocarboxylic acids
- C08K5/103—Esters; Ether-esters of monocarboxylic acids with polyalcohols
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L101/00—Compositions of unspecified macromolecular compounds
- C08L101/16—Compositions of unspecified macromolecular compounds the macromolecular compounds being biodegradable
Definitions
- the present invention relates to a biodegradable polyester resin composition and a film or sheet comprising the same.
- biodegradable plastics which are decomposed into water and carbon dioxide by the action of microorganisms after use, are attracting attention as the realization of a recycling-oriented society on a global scale is eagerly desired.
- biodegradable plastics include 1) microbially produced aliphatic polyesters such as polyhydroxyalkanoates (hereinafter referred to as PHA), 2) polylactic acid and polycaprolactone, polybutylene adipate terephthalate, polybutylene succinate adipate, polybutylene. Petroleum-derived resins such as succinates, and 3) natural polymers such as starch and cellulose acetate.
- PHBH poly (3-hydroxybutyrate-co-3-hydroxyhexanoate, abbreviated as PHBH) produced from microorganisms as a film or sheet using a microorganism-producing aliphatic polyester
- Patent Document 1 a sheet comprising a resin composition containing a petroleum-derived resin such as polybutylene adipate terephthalate, polybutylene succinate adipate, polybutylene succinate, polycaproclactone, and PHBH (see Patent Document 2). It is disclosed.
- the present invention aims to improve the tear strength in the MD direction of a film or sheet formed from a resin composition containing a microorganism-producing aliphatic polyester.
- the present inventors examined the microstructure (morphology) of the obtained molded article by a transmission electron microscope analysis-image analysis method (TEM method). It has been found that the film is stretched and oriented in the take-up direction (MD direction). Furthermore, the present inventors presume that the decrease in tear strength in the MD direction of the film or sheet is due to the stretched / orientated state of the aliphatic polyester component, and further studied to adjust the morphology of the molded product. As a result, it was found that the morphology of the resin material in the molded product of film or sheet can be controlled by using a specific amount of modified glycerin in addition to the microbially-produced aliphatic polyester and other biodegradable resin. By controlling the morphology, the first success in increasing the tear strength in the MD direction of molded articles such as films and sheets was achieved, and the present invention was completed.
- TEM method transmission electron microscope analysis-image analysis method
- the first of the present invention is the formula (1): [—CHR—CH 2 —CO—O—] (Wherein, R is an alkyl group represented by C n H 2n + 1, n is an integer of 1 to 15.)
- a biodegradable polyester resin comprising 60 to 400 parts by weight of polybutylene adipate terephthalate (PBAT) and 10 to 50 parts by weight of modified glycerin with respect to 100 parts by weight of an aliphatic polyester (P3HA) having a repeating unit represented by Relates to the composition.
- PBAT polybutylene adipate terephthalate
- P3HA aliphatic polyester
- Relates a repeating unit represented by Relates to the composition.
- the biodegradable polyester resin composition is for film molding or sheet molding.
- the aliphatic polyester (P3HA) is poly (3-hydroxybutyrate-co-3-hydroxyhexanoate) (PHBH), poly (3-hydroxybutyrate) (P3HB), poly (3 -Hydroxybutyrate-co-3-hydroxyvalerate) (PHBV), poly (3-hydroxybutyrate-co-4-hydroxybutyrate) (P3HB4HB), poly (3-hydroxybutyrate-co-3-hydroxy) Octanoate) and at least one selected from the group consisting of poly (3-hydroxybutyrate-co-3-hydroxyoctadecanoate).
- the aliphatic polyester (P3HA) has a melt viscosity at 160 ° C.
- the ratio of the melt viscosity of the aliphatic polyester (P3HA) to the melt viscosity of the polybutylene adipate terephthalate (PBAT) is 0.5 or more.
- PBS polybutylene succinate
- P3HA polybutylene succinate
- the ratio of aliphatic polyester (P3HA) to the total amount of aliphatic polyester (P3HA), polybutylene adipate terephthalate (PBAT) and polybutylene succinate (PBS) is 45% by weight or less.
- the modified glycerin is an acetylated monoglyceride.
- the second of the present invention is formed by molding the biodegradable polyester resin composition
- the present invention relates to a film or sheet in which the maximum value of the major axis of the phase composed of the aliphatic polyester (P3HA) measured by transmission electron microscope analysis-image analysis method (TEM method) is 18 ⁇ m or less and the average value is 8 ⁇ m or less.
- the film or sheet is formed by an inflation method or a T-die extrusion method.
- the take-up speed during molding is 30 m / min or less.
- a biodegradable resin raw material is used, and carbon dioxide on the earth is not increased.
- the proportion of the microbially produced aliphatic polyester that is carbon neutral is high, and the MD direction of the molded product Films and sheets having high tear strength can be provided.
- FIG. 1 is a TEM image of the film obtained in Example 1.
- FIG. FIG. 2 is an image obtained by processing the TEM image of FIG. 1 with image analysis software and determining the major axis of PHBH.
- FIG. 3 is a TEM image of the film obtained in Comparative Example 1.
- the biodegradable polyester resin composition according to the present invention has a resin component represented by the formula (1): [—CHR—CH 2 —CO—O—] (where R is an alkyl represented by C n H 2n + 1).
- the poly (3-hydroxyalkanoate) (abbreviation: P3HA) used in the present invention is produced from a microorganism and has the formula (1): [—CHR—CH 2 —CO—O—] (wherein R is an alkyl group represented by C n H 2n + 1 , and n is an integer of 1 to 15.)).
- the microorganism that produces P3HA is not particularly limited as long as it is a microorganism capable of producing P3HAs.
- a copolymer-producing bacterium of hydroxybutyrate and other hydroxyalkanoate a copolymer having monomer units of 3-hydroxybutyrate and 3-hydroxyvalerate (hereinafter abbreviated as “PHBV”).
- PHBH poly (3-hydroxybutyrate-co-3-hydroxyhexanoate
- Aeromonas caviae poly (3-hydroxybutyrate-co -4-hydroxybutyrate) producing bacteria such as Alcaligenes eutrophus
- PHBH poly (3-hydroxybutyrate-co-3-hydroxyhexanoate
- Aeromonas caviae poly (3-hydroxybutyrate-co -4-hydroxybutyrate) producing bacteria such as Alcaligenes eutrophus
- alkaligenes Utropha Strain AC32 Alcaligenes eutrophus AC32, FERM BP-6038
- J. Bateriol., 179, p4821-4830 (1997) is more preferable, and these microorganisms are cultured under appropriate conditions to accumulate PHBH in the cells
- the microbial cells that have been used are used.
- the weight average molecular weight of P3HA used in the present invention is preferably 50,000 to 3,000,000, more preferably 100,000 to 1,500,000 from the viewpoint of the balance between moldability and physical properties.
- the weight average molecular weight here means what was measured from the polystyrene conversion molecular weight distribution using the gel permeation chromatography (GPC) which used chloroform eluent.
- P3HA examples include poly (3-hydroxybutyrate) (abbreviation: P3HB), poly (3-hydroxybutyrate-co-3-hydroxyhexanoate) (abbreviation: PHBH), poly (3-hydroxybutyrate) Rate-co-3-hydroxyvalerate) (abbreviation: PHBV), poly (3-hydroxybutyrate-co-4-hydroxybutyrate) (abbreviation: P3HB4HB), poly (3-hydroxybutyrate-co-3- Hydroxyoctanoate), poly (3-hydroxybutyrate-co-3-hydroxyoctadecanoate) and the like.
- P3HB, PHBH, PHBV, and P3HB4HB are listed as those that are industrially easy to produce.
- n in the alkyl group (R) is 1 PHBH consisting of a unit and a repeating unit in which n is 3 is preferred.
- Patent Document 2 A specific method for producing PHBH is described in, for example, International Publication No. 2010/013483 (Patent Document 2).
- Patent Document 2 Kaneka Corporation "AONILEX (AONILEX)" (registered trademark) etc. are mentioned.
- the composition ratio of PHBH repeating units is such that the composition ratio of poly (3-hydroxybutyrate) / poly (3-hydroxyhexanoate) is 80/20 to 99/1 from the viewpoint of the balance between flexibility and strength. (Mol / mol) is preferred, and 75/15 to 97/3 (mo1 / mo1) is more preferred. The reason is that 99/1 or less is preferable from the viewpoint of flexibility, and 80/20 or more is preferable in that the resin has an appropriate hardness.
- PHBV also changes its melting point, Young's modulus, etc. depending on the ratio of the 3-hydroxybutyrate (3HB) component and the 3-hydroxyvalerate (3HV) component, but the 3HB component and the 3HV component co-crystallize.
- the degree is as high as 50% or more, and it is more flexible than poly 3-hydroxybutyrate (P3HB), but the elongation at break tends to be as low as 50% or less.
- the P3HA used in the present invention preferably has a melt viscosity at 160 ° C. of 900 to 3,600 Pa ⁇ s. If the melt viscosity is less than 900 Pa ⁇ s, the melt viscosity ratio with respect to polybutylene adipate terephthalate (PBAT) becomes small, and it may be difficult to control the morphology of the resin composition and the molded product such as a film or sheet obtained therefrom. If it is greater than 3,600 Pa ⁇ s, the moldability may be inferior. In the present invention, the melt viscosity can be increased by adding isocyanate to P3HA.
- PBAT polybutylene adipate terephthalate
- P3HA produced by microorganisms has a particularly low crystallization rate among aliphatic polyesters, it is particularly effective to use the processing method as in the present invention.
- P3HA is excellent in biodegradability in both aerobic and anaerobic environments, and does not generate toxic gas during combustion.
- PHBH is preferable in that it does not use petroleum-derived materials as raw materials, uses plant raw materials, and does not increase carbon dioxide on the earth, that is, has an excellent feature of being carbon neutral.
- the present invention has an advantage that a non-biodegradable crystal nucleating agent is not added and the excellent biodegradability of P3HA is not impaired.
- the polybutylene adipate terephthalate (PBAT) used in the present invention refers to a random copolymer of 1,4-butanediol, adipic acid and terephthalic acid, and in particular, Japanese Patent Publication No. 10-508640, etc.
- PBAT polybutylene adipate terephthalate
- B) is a mixture containing butanediol (provided that the molar ratio of (a) to (b) is 0.4: 1 to 1.5: PBAT obtained by the reaction of 1) is preferred.
- Examples of commercially available PBAT include “Ecoflex” (registered trademark) manufactured by BASF.
- the content of the PBAT in the biodegradable polyester of the present invention is preferably 60 to 400 parts by weight, more preferably 100 to 250 parts by weight with respect to 100 parts by weight of the P3HA. If the content is less than 60 parts by weight, P3HA cannot form a discontinuous layer in a dispersed state, so that the tear strength may be lowered, and if it exceeds 400 parts by weight, the moldability may be lowered.
- the morphology of the resin composition and the molded product such as film or sheet obtained therefrom is controlled. be able to.
- modified glycerin acetylated monoglycerides such as glycerin diacetomonolaurate, glycerin diacetomonocaprylate, and glycerin diacetomonodecanoate are preferable.
- commercially available modified glycerin include “Rikemar” (registered trademark) PL series manufactured by Riken Vitamin Co., Ltd.
- the content of the modified glycerin with respect to the biodegradable polyester of the present invention is preferably 10 to 50 parts by weight, more preferably 20 to 40 parts by weight with respect to 100 parts by weight of the P3HA.
- the content is less than 10 parts by weight, the maximum value and the average value of the major axis of the phase made of P3HA become too large, P3HA cannot form a discontinuous layer in a dispersed state, and the tear strength of the film or sheet is lowered.
- it exceeds 50 parts by weight the effect will not change and may cause bleeding out.
- the biodegradable polyester resin composition of the present invention contains polybutylene succinate (PBS) and has a function of lowering the melt viscosity of PBAT, the viscosity ratio of P3HA to PBAT is increased, and P3HA is non-peptidized. This is excellent in that a continuous phase is easily formed and the tear strength is improved.
- PBS is an aliphatic polyester copolymer synthesized by dehydration polycondensation of 1,4-butanediol and succinic acid, and commercially available products include “Bionole” manufactured by Showa Denko KK.
- the content of the PBS in the biodegradable polyester of the present invention is preferably 20 to 150 parts by weight, more preferably 40 to 120 parts by weight with respect to 100 parts by weight of the P3HA, from the viewpoint of not affecting the tear strength.
- the resin composition of the present invention includes fillers used as usual additives, colorants such as pigments and dyes, odor absorbents such as activated carbon and zeolite, vanillin, and dextrin as long as the effects of the present invention are not impaired. Fragrances such as antioxidants, antioxidants, weather resistance improvers, UV absorbers, plasticizers, lubricants, mold release agents, water repellents, antibacterial agents, slidability improvers, and other secondary additives You may add at least 1 sort.
- the resin composition as described above and the film or sheet obtained therefrom are obtained by controlling the morphology.
- the maximum value of the major axis of the phase composed of the P3HA measured by transmission electron microscope analysis-image analysis method (TEM method) is 18 ⁇ m or less, and the average The value is in the range of 8 ⁇ m or less, and is obtained by controlling the morphology so that P3HA forms a discontinuous layer in a dispersed state.
- the TEM method referred to in the present invention is a ruthenium tetroxide (RuO 4) obtained by cutting a slice of about 100 nm parallel to the surface of the film or sheet from the film or sheet using a microtome so that the MD direction can be observed. ) After selectively staining P3HA with osmium tetroxide, phosphotungstic acid, etc., using a transmission electron microscope (JEM-1200EX, manufactured by JEOL Ltd.), the cut surface of the film or sheet was cut at an acceleration voltage of 80 kV.
- RuO 4 ruthenium tetroxide
- the phase state of P3HA when observed from the surface direction was photographed at a magnification of 10,000 times, and a binarized image analysis software for the range of about 18 ⁇ m x about 25 ⁇ m of the image (“Win Roof of Mitani Corporation”) Is a method of automatically measuring the major axis of the phase composed of P3HA and its average value. Further, the major axis of the phase composed of P3HA that cannot be discriminated by the image analysis software is obtained manually.
- the major axis of the phase composed of P3HA indicates the longest diameter of the individual phases composed of P3HA in the microscopically observed image, and the average value indicates the major axis of the phase composed of all P3HA present in the visual field. The average value is shown.
- three visual fields are randomly observed as the visual field to be observed.
- the ratio of the melt viscosity of the aliphatic polyester (P3HA) to the melt viscosity of the polybutylene adipate terephthalate (PBAT) is 0.5 or more, preferably 0.7 or more.
- the take-up speed during molding is 30 m / min or less, preferably 25 m / min or less, more preferably Examples of the method include 20 m / min or less.
- the ratio of melt viscosity (P3HA / PBAT) of 1) is a ratio of melt viscosity at 160 ° C. of P3HA and PBAT.
- the upper limit of the ratio of the melt viscosity of P3HA to the melt viscosity of PBAT is not particularly limited, but if it is too large, the viscosity of the resin composition becomes too high, and molding processability may be deteriorated, and P3HA tends to be finely dispersed. From the viewpoint, 2.0 or less is preferable.
- the ratio of the melt viscosity of P3HA to the melt viscosity of PBAT refers to a value obtained by dividing the melt viscosity of P3HA by the melt viscosity of PBAT.
- the melt viscosity was measured using a high shear viscometer capillary rheometer, with a set temperature of 160 ° C. to 170 ° C., a capillary size of ⁇ 1 mm, a length of 10 mm, a shear rate of 100 (1 / sec) to 2,000 (1). / Sec). In the examples described later, the melt viscosity is measured by this method.
- the ratio of the aliphatic polyester (P3HA) exceeds 45% by weight, PHBH constitutes a continuous phase in the resin, and the tear strength in the MD direction in a molded product such as a molded film or sheet. May decrease too much.
- the lower limit of the ratio of the aliphatic polyester (P3HA) is not particularly limited, but the content ratio of the aliphatic polyester (P3HA) is preferably 1% by weight or more from the viewpoint of increasing the non-petroleum degree, and the aliphatic polyester (P3HA) If the ratio is too small, the effect of increasing the blending ratio of the microorganism-producing aliphatic polyester that is carbon neutral is weakened. Therefore, the ratio of the aliphatic polyester (P3HA) is more preferably 20% by weight or more, More preferably, it is 25% by weight or more.
- the take-up speed at the time of molding of a film or sheet exceeds 30 m / min, the major axis of the phase made of P3HA in the film or sheet becomes too long, and further in the MD direction. In some cases, the tear strength may be greatly reduced.
- the film and sheet of the present invention in which the maximum value of the major axis of the discontinuous phase composed of P3HA is 18 ⁇ m or less and the average value is 8 ⁇ m or less can be obtained.
- the resin composition of the present invention can be produced by a known method such as heating and melting using a single screw extruder, a twin screw extruder, or the like.
- the film or sheet of the present invention can be produced by a known molding method such as an inflation method or a T-die extrusion method. Specific conditions may be set as appropriate.
- the inflation method the pellet is dried by a dehumidifying dryer or the like before inflation molding until the moisture content of the pellets is 500 ppm or less, and the cylinder set temperature is 100 ° C. to 160 ° C. It is preferable to set the adapter and the die to a set temperature of 130 ° C. to 160 ° C.
- a thickness of about 1 to 100 ⁇ m is called a film
- a thickness of over 100 ⁇ m to about 20 mm is called a sheet.
- the film or sheet of the present invention can be suitably used in agriculture, fishery, forestry, horticulture, medicine, hygiene, food industry, clothing, non-clothing, packaging, automobiles, building materials, and other fields. Used for applications such as agricultural multi-films, forestry fumigation sheets, tying tapes including flat yarns, rooting films for plants, diaper back sheets, packaging sheets, shopping bags, garbage bags, draining bags, and other compost bags It is done.
- 3-Hydroxyalkanoate polymer PHBH (raw material A-1) used in this example was produced as follows. KNK-631 strain (see International Publication No. 2009/145164) was used for PHA culture production.
- the composition of the seed medium is 1 w / v% Meat-extract, 1 w / v% Bacto-Triptone, 0.2 w / v% Yeast-extract, 0.9 w / v% Na 2 HPO 4 ⁇ 12H 2 O, 0.15 w / V% KH 2 PO 4 , pH 6.8.
- the composition of the preculture medium is 1.1 w / v% Na 2 HPO 4 ⁇ 12H 2 O, 0.19 w / v% KH 2 PO 4 , 1.29 w / v% (NH 4 ) 2 SO 4 , 0.1 w / v% MgSO 4 .7H 2 O, 0.5 v / v% trace metal salt solution (1.6 W / v% FeCl 3 .6H 2 O in 0.1 N hydrochloric acid, 1 w / v% CaCl 2 .2H 2 O, 0 0.02 w / v% CoCl 2 .6H 2 O, 0.016 w / v% CuSO 4 .5H 2 O, 0.012 w / v% NiCl 2 .6H 2 O).
- the carbon source was palm kernel oil added at a concentration of 10 g / L.
- the composition of the PHA production medium is 0.385 w / v% Na 2 HPO 4 ⁇ 12H 2 O, 0.067 w / v% KH 2 PO 4 , 0.291 w / v% (NH 4 ) 2 SO 4 , 0.1 w / v% MgSO 4 .7H 2 O, 0.5 v / v% trace metal salt solution (1.6 w / v% FeCl 3 .6H 2 O in 0.1 N hydrochloric acid, 1 w / v% CaCl 2 .2H 2 O, 0 0.02 w / v% CoCl 2 ⁇ 6H 2 O, 0.016 w / v% CuSO 4 ⁇ 5H 2 O, 0.012 w / v% NiCl 2 ⁇ 6H 2 O), 0.05 w / v% BIOSPUREX 200K (Antifoamer: manufactured by Cognis Japan).
- a glycerol stock (50 ⁇ L) of KNK-631 strain was inoculated into a seed medium (10 mL) and cultured for 24 hours to perform seed culture.
- 1.0 v / v% of the seed mother culture solution was inoculated into a 3 L jar fermenter (MDL-300 type, manufactured by Maruhishi Bioengine) containing 1.8 L of a preculture medium.
- the operating conditions were a culture temperature of 33 ° C., a stirring speed of 500 rpm, an aeration rate of 1.8 L / min, and the culture was performed for 28 hours while controlling the pH between 6.7 and 6.8.
- a 14% aqueous ammonium hydroxide solution was used for pH control.
- 1.0 V / v% of the preculture solution was inoculated into a 10 L jar fermenter (MDS-1000, manufactured by Maruhishi Bioengine) containing 6 L of production medium.
- the operating conditions were a culture temperature of 28 ° C., a stirring speed of 400 rpm, an aeration rate of 6.0 L / min, and a pH controlled between 6.7 and 6.8.
- a 14% aqueous ammonium hydroxide solution was used for pH control. Palm kernel olein oil was used as the carbon source. Culturing was performed for 64 hours, and after completion of the cultivation, the cells were collected by centrifugation, washed with methanol, freeze-dried, and the weight of the dried cells was measured.
- the gas chromatograph used was Shimadzu GC-17A, and the capillary column used was GL Science's Neutra Bond-1 (column length 25 m, column inner diameter 0.25 mm, liquid film thickness 0.4 ⁇ m). He was used as the carrier gas, the column inlet pressure was set to 100 kPa, and 1 ⁇ L of the sample was injected. As temperature conditions, the temperature was raised from the initial temperature of 100 to 200 ° C. at a rate of 8 ° C./min, and further from 200 to 290 ° C. at the rate of 30 ° C./min. As a result of analysis under the above conditions, it was PHA as shown in the chemical formula (1), poly (3-hydroxybutyrate-co-3-hydroxyhexanoate).
- the 3HH composition was 11.2 mol%.
- PHBH was obtained from the culture solution according to the method described in International Publication No. 2010/066753.
- the weight average molecular weight measured by GPC was 570,000.
- the melt viscosity of PHBH at 160 ° C. was measured and found to be 1,150 Pa ⁇ s.
- the raw material A-1 was obtained by hydrolysis for 72 hours at 80 ° C. and a relative humidity of 95%.
- Raw material A-4 100 parts by weight of raw material A-1 2 parts by weight of an isocyanate compound (manufactured by Nippon Polyurethane Industry Co., Ltd., Millionate MR200) was set by a twin-screw extruder at a set temperature of 100 to 130 ° C. and a screw rotation speed of 100 rpm.
- the raw material A-4 was obtained by melting and kneading at The melt viscosity of the obtained raw material A-4 was 2,350 Pa ⁇ s.
- the raw material A-1 was obtained by hydrolysis for 36 hours at 80 ° C. and a relative humidity of 95%.
- the KNK-005 strain was used in place of the KNK-631 strain, and was obtained in the same manner as in Production Example 1.
- Raw material B-1 PBAT having a melt viscosity of 1,800 Pa ⁇ s at 160 ° C. (manufactured by BASF, “Ecoflex (registered trademark)”).
- Raw material B-2 PBS (manufactured by Showa Denko KK, “Bionore (registered trademark)”). ⁇ Modified glycerin compound>
- Raw material C-1 Acetylated monoglyceride (Riken Vitamin Co., Ltd., “Riquemar (registered trademark)” PL012).
- polyester B-1 (PBAT), 50 parts by weight of polyester B-2 (PBS), modified glycerin compound C-1 (acetyl) with respect to 100 parts by weight of 3-hydroxyalkanoate polymer (raw material A-1)
- the polyester resin composition containing PHBH is obtained by melting and kneading 25 parts by weight of a monoglyceride) with a twin screw extruder (manufactured by Nippon Steel Co., Ltd .: TEX30) at a set temperature of 100 to 130 ° C. and a screw rotation speed of 100 rpm. It was.
- FIG. 1 shows a TEM image of the film obtained in Example 1.
- the vertical direction is the take-up direction (MD direction).
- FIG. 2 shows a state of an image in which the major axis of PHBH is discriminated by the image analysis software.
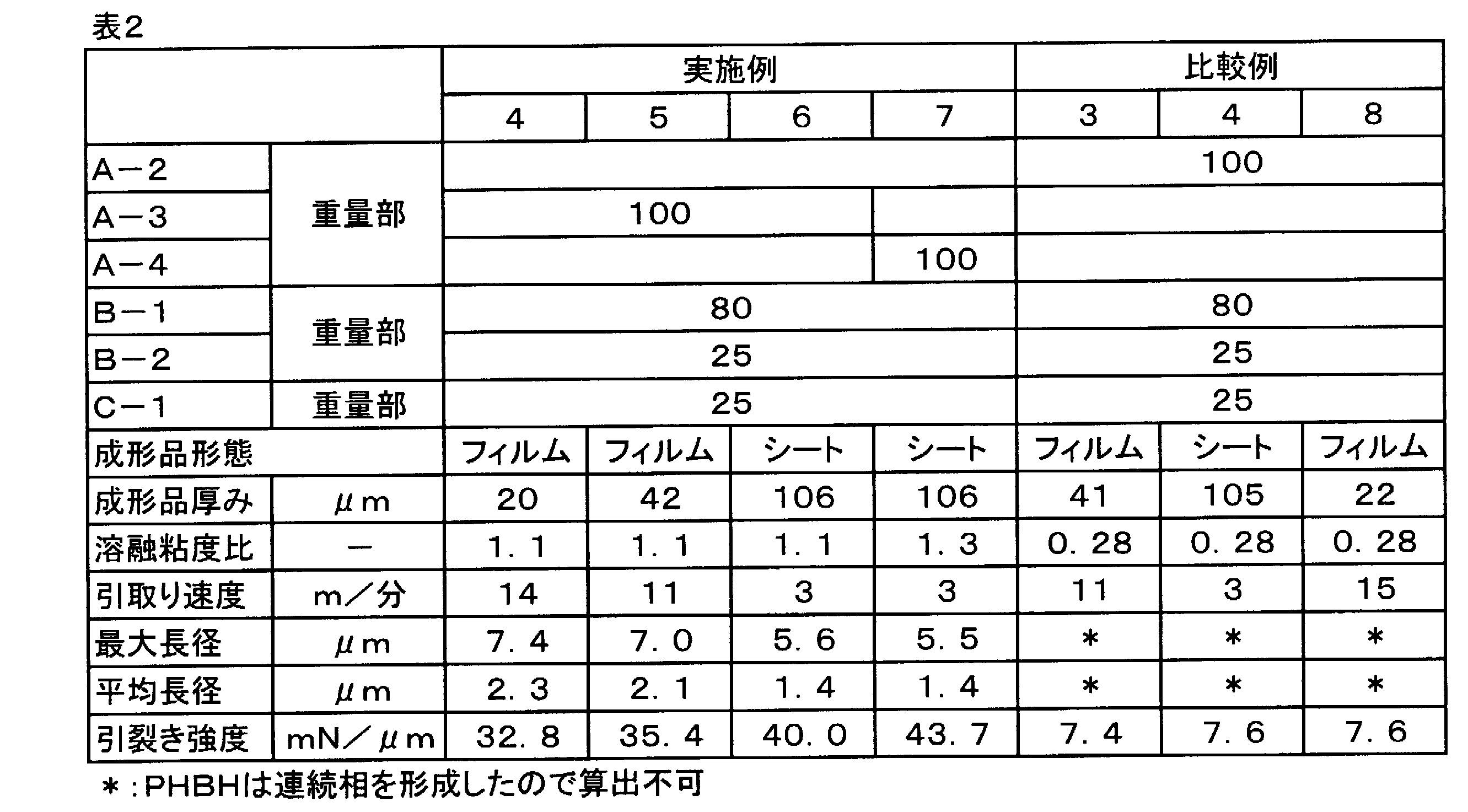
- Example 3 The resin composition obtained in Example 1 was formed into a sheet, and the tear strength and the maximum major axis and average major axis of the phase composed of PHBH were measured in the same manner as in Example 1.
- Example 1 Example 1 was used except that 100 parts by weight of 3-hydroxyalkanoate polymer (raw material A-2) having a low melt viscosity was used instead of 100 parts by weight of 3-hydroxyalkanoate polymer (starting material A-1).
- a resin composition was produced, a film was formed in the same manner as in Example 2, and the tear strength of the film, the maximum major axis and the average major axis of the PHBH phase were calculated.
- FIG. 3 shows a TEM image of the film obtained in Comparative Example 1. In the figure, the vertical direction is the MD direction.
- Comparative Example 2 Using the resin composition obtained in Comparative Example 1, a sheet was molded in the same manner as in Example 3, and the tear strength and the maximum major axis and average major axis of the PHBH phase were measured for the obtained sheet.
- the PHBH phase (white portion in the figure) in the sheet obtained in Example 1 has an elliptical shape slightly stretched in the MD direction as shown in FIG. 1, but is randomly distributed and arranged. As shown in Table 1, the maximum major axis of these PHBH phases was less than 18 ⁇ m, and the average major axis was less than 8 ⁇ m. Further, the PHBH phase of the film obtained in Example 2 and the sheet obtained in Example 3 also formed the same dispersed phase as in Example 1, and the maximum major axis of each PHBH phase was less than 18 ⁇ m. The average major axis was less than 8 ⁇ m. Further, the tear strength in the MD direction was 40 mN / ⁇ m or more in all of Examples 1 to 3, indicating a high value.
- Polyester B-1 80 parts by weight
- polyester B-2 25 parts by weight
- modified glycerin compound C-1 with respect to 100 parts by weight of 3-hydroxyalkanoate polymer raw material A-3)
- Acylated monoglyceride 25 parts by weight
- Example 4 is a method similar to Example 1
- Example 5 is a film formed by the same method as Example 2. For the obtained film, the tear strength, the maximum major axis of PHBH and the average major axis are calculated. The same operation as in Example 1 was performed.
- Example 6 The resin composition obtained in Example 4 was formed into a sheet by the same method as in Example 3, and the tear strength, the maximum major axis of the PHBH phase, and the average major axis were measured for the obtained sheet.
- Example 7 A polyester resin composition was obtained in the same manner as in Example 4 except that the raw material A-4 was used instead of the raw material A-3.
- the obtained polyester resin composition was formed into a sheet by the same method as in Example 3, and the tear strength, the maximum major axis and the average major axis of the PHBH phase were measured for the obtained sheet.
- Comparative Example 4 The resin composition obtained in Comparative Example 3 was formed into a sheet by the same method as in Example 6, and the tear strength, the maximum major axis of the PHBH phase, and the average major axis were measured for the obtained sheet.
- Table 2 shows the results of Examples 4 to 7 and Comparative Examples 3 and 4 described above.
- the PHBH (raw material A-3) in the films obtained in Examples 4 and 5 and the sheet obtained in Example 6 has a high molecular weight and thus has a melt viscosity of 1,910 Pa ⁇ s and is Ecoflex. It was higher than (raw material B-1) ((P3HA / PBAT) was 1.0 or more). Therefore, even if the composition ratio of PHBH exceeded 45% by weight of the total resin and approached 50% by weight, the phase composed of PHBH could form a dispersed state. Furthermore, since the maximum major axis of the PHBH phase in the films and sheets obtained in Examples 4 to 7 was less than 18 ⁇ m and the average major axis was less than 8 ⁇ m, the tear strength in the MD direction was high. showed that. On the other hand, in Comparative Examples 3 and 4, since the polyester raw material A-2 formed a continuous phase and did not form an island phase (dispersed phase), the maximum major axis and the average major axis could not be calculated.
- Examples 8 to 12 A polyester resin composition was produced in the same manner as in Example 1 with the formulation described in Table 3, and a film was molded in the same manner as in Example 1 except that the take-up speed shown in Table 3 was adjusted. With respect to the obtained film, the tear strength, the maximum major axis and the average major axis of the PHBH phase were calculated. The results are shown in Table 3.
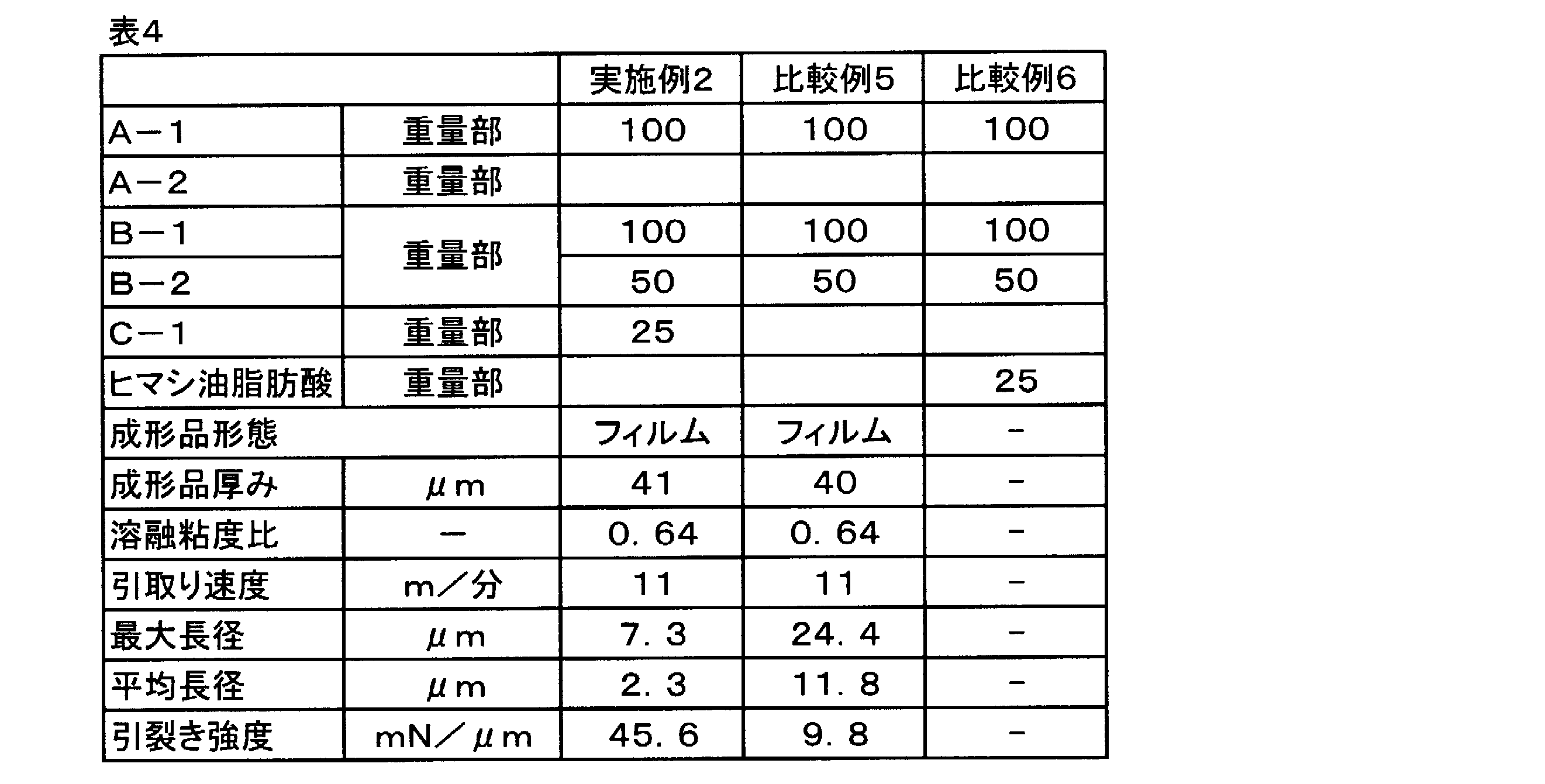
- Example 5 A resin composition was produced in the same manner as in Example 2 except that the modified glycerin compound C-1 was not used. A film was formed from this resin composition, and the obtained film had a tear strength, a maximum major axis of PHBH and The average major axis was calculated. The results are shown in Table 4.
- Comparative Example 5 the maximum major axis and the average major axis are both larger than those in Example 2, and the tear strength is low. From this, without the modified glycerin compound C-1, the discontinuous phase composed of PHBH in the resin composition is not as fine as in Example 1 because its maximum major axis and average major axis are long, and as a result The tear strength is considered to be low.
- Example 7 The same procedure as in Example 1 was conducted except that 100 parts by weight of the 3-hydroxyalkanoate polymer (raw material A-2) having a low melt viscosity was used instead of 100 parts by weight of the 3-hydroxyalkanoate polymer (starting material A-1). A resin composition was manufactured, and a film was formed in the same manner as in Example 1 except that the take-up speed was 15 m / min. The maximum major axis and the average major axis of the PHBH phase were calculated from the tear strength and the TEM image. The results are shown in Table 1. As in Comparative Example 5, the maximum major axis and the average major axis were both larger than those in Example 2, and the tear strength was low.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Materials Engineering (AREA)
- Biological Depolymerization Polymers (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
Abstract
Description
(但し、RはCnH2n+1で表されるアルキル基であり、nは1以上15以下の整数である。)
で示される繰り返し単位を有する脂肪族ポリエステル(P3HA)100重量部に対し、ポリブチレンアジペートテレフタレート(PBAT)を60~400重量部および変性グリセリンを10~50重量部含有してなる生分解性ポリエステル樹脂組成物に関する。
好ましい実施態様では、前記生分解性ポリエステル樹脂組成物は、フィルム成形用またはシート成形用である。
好ましい実施態様では、前記脂肪族ポリエステル(P3HA)は、ポリ(3-ヒドロキシブチレート-コ-3-ヒドロキシヘキサノエート)(PHBH)、ポリ(3-ヒドロキシブチレート)(P3HB)、ポリ(3-ヒドロキシブチレート-コ-3-ヒドロキシバリレート)(PHBV)、ポリ(3-ヒドロキシブチレート-コ-4-ヒドロキシブチレート)(P3HB4HB)、ポリ(3-ヒドロキシブチレート-コ-3-ヒドロキシオクタノエート)、ポリ(3-ヒドロキシブチレート-コ-3-ヒドロキシオクタデカノエート)からなる群から選択される少なくとも1種である。
好ましい実施態様では、脂肪族ポリエステル(P3HA)の160℃における溶融粘度が900~3,600Pa・sである。
好ましい実施態様では、前記ポリブチレンアジペートテレフタレート(PBAT)の溶融粘度に対する脂肪族ポリエステル(P3HA)の溶融粘度の比(P3HA/PBAT)が0.5以上である。
好ましい実施態様では、脂肪族ポリエステル(P3HA)100重量部に対し、さらにポリブチレンサクシネート(PBS)を20~150重量部含有してなる。
好ましい実施態様では、脂肪族ポリエステル(P3HA)、ポリブチレンアジペートテレフタレート(PBAT)およびポリブチレンサクシネート(PBS)の合計量に対する脂肪族ポリエステル(P3HA)の比率が45重量%以下である。
好ましい実施態様では、変性グリセリンが、アセチル化モノグリセライドである。
透過型電子顕微鏡分析-画像解析法(TEM法)によって測定される前記脂肪族ポリエステル(P3HA)からなる相の長径の最大値が18μm以下であり、平均値が8μm以下であるフィルムまたはシートに関する。
好ましい実施態様では、前記フィルムまたはシートはインフレーション法またはTダイ押出法により成形される。
好ましい実施態様では、成形時の引き取り速度が30m/分以下である。
変性グリセリンとしては、グリセリンジアセトモノラウレート、グリセリンジアセトモノカプリレート、グリセリンジアセトモノデカノエートなどのアセチル化モノグリセライドが好ましい。変性グリセリンの市販品としては、理研ビタミン株式会社の「リケマール」(登録商標)PLシリーズなどが挙げられる。
本実施例で使用するPHBH(原料A-1)は、以下のようにして作製した。
PHAの培養生産にはKNK-631株(国際公開第2009/145164号参照)を用いた。
種母培地の組成は1w/v% Meat-extract、1w/v% Bacto-Tryptone、0.2w/v% Yeast-extract、0.9w/v% Na2HPO4・12H2O、0.15w/v% KH2PO4、pH6.8とした。
<3-ヒドロキシアルカノエート重合体>
原料A-2:Mw32万、3HH=11.2モル%、160℃の溶融粘度=510Pa・sのPHBH(カネカ社製)。原料A-1を80℃、相対湿度95%で72時間加水分解して得た。
原料A-3:Mw79万、3HH=10.4モル%、160℃の溶融粘度=1,910Pa・sのPHBH。培養時間を96時間にした以外は、製造例1と同様にして得た。
原料A-4:原料A-1 100重量部に対して2重量部のイソシアネート化合物(日本ポリウレタン工業社製、ミリオネートMR200)を、2軸押出機により、設定温度100~130℃、スクリュー回転数100rpmで溶融混錬して、原料A-4を得た。得られた原料A-4の溶融粘度は2,350Pa・sであった。
原料A-5:Mw48万、3HH=11.2モル%、160℃の溶融粘度940Pa・sのPHBH(カネカ社製)。原料A-1を80℃、相対湿度95%で36時間加水分解して得た。
原料A-6:Mw62万、3HH=5.4モル%、160℃の溶融粘度=1,240Pa・sのPHBH(カネカ社製)。KNK-631株のかわりにKNK-005株を用い、製造例1と同様にして得た。
<ポリエステル>
原料B-1:160℃の溶融粘度1,800Pa・sのPBAT(BASF社製、「エコフレックス(登録商標)」)。
原料B-2:PBS(昭和電工社製、「ビオノーレ(登録商標)」)。
<変性グリセリン化合物>
原料C-1:アセチル化モノグリセライド(理研ビタミン社製、「リケマール(登録商標)」PL012)。
(樹脂組成物の製造)
3-ヒドロキシアルカノエート重合体(原料A-1)100重量部に対して、ポリエステルB-1(PBAT)100重量部、ポリエステルB-2(PBS)50重量部、変性グリセリン化合物C-1(アセチル化モノグリセライド)25重量部を、2軸押出機(日本製鋼社製:TEX30)で、設定温度100~130℃、スクリュー回転数100rpmで溶融混錬して、PHBHを含有するポリエステル樹脂組成物を得た。
得られた樹脂組成物はインフレーションフィルム成形機(北進産業社製)を用い、円形ダイスリップ厚=1mm、円形ダイスリップ直径=100mm、設定温度=120~140℃、表1に記載した引き取り速度でフィルムを成形した。
得られたフィルム成形品またはシート成形品は、エルメンドルフ引き裂き強度測定器(熊谷理器工業社製)を用い、JIS 8116に準拠して引き裂き強度を測定した。
得られたフィルムまたはシート成形品から、ミクロトームを用い、MD方向が観察できるようにフィルムまたはシートの表面に平行に約100nm厚の薄片サンプルを切り取り、RuO4で染色した後、透過型電子顕微鏡(TEM)(日本電子社製、JEM-1200EX)を用い、加速電圧80kVでPHBHの分散状態を観察した。図1に実施例1で得られたフィルムのTEM画像を示す。なお、図中、上下方向が引取り方向(MD方向)である。
画像解析ソフト(三谷商事社製「Win Roof」)を用いて最大長径と平均長径を算出した。値は、1万倍の倍率で撮影したTEM画像の約18μm×約25μmの範囲で算出した。
前記画像はRuO4染色でコントラストをつけているが、このコントラストがはっきりしないと解析ソフトで2値化処理によるPHBHからなる相の判別が困難な場合がある。そのような場合は、TEM写真から手作業で長径を求めた。図2に前記画像解析ソフトでPHBHの長径を判別した画像の様子を示す。
実施例1で得られた樹脂組成物をシート成形して、実施例1と同様に引き裂き強度およびPHBHからなる相の最大長径と平均長径とを測定した。
得られた樹脂組成物はTダイシート成形機(東洋精機製作所社製:ラボプラストミル)を用い、ダイスリップ厚=250μm、ダイスリップ幅=150mm、シリンダー設定温度=120~140℃、ダイス設定温度=150~160℃、表1に記載した引き取り速度でシートを成形した。
3-ヒドロキシアルカノエート重合体(原料A-1)100重量部の代わりに溶融粘度が低い3-ヒドロキシアルカノエート重合体(原料A-2)100重量部を用いた以外は実施例1と同様に樹脂組成物を製造し、実施例2と同様の方法でフィルムを成形し、そのフィルムの引き裂き強度、PHBHの相の最大長径と平均長径を算出した。図3に比較例1で得られたフィルムのTEM画像を示す。図中、上下方向がMD方向である。
比較例1で得られた樹脂組成物を用いて実施例3と同様の方法でシートを成形し、得られたシートについて、引き裂き強度、PHBHの相の最大長径と平均長径を測定した。
また、実施例2で得られたフィルムおよび実施例3で得られたシートのPHBHの相も、実施例1と同様の分散相を形成しており、それぞれのPHBHの相の最大長径は18μm未満であり、平均長径は8μm未満であった。また、MD方向の引き裂き強度は実施例1~3いずれも40mN/μm以上であり、高い値を示した。
これに対し、比較例1および比較例2で使用した原料A-2は分子量が低いので溶融粘度が低く、実施例1または2と同じ条件で樹脂組成物およびフィルムあるいはシート成形しても、図3に示すように、PHBH(図中の白い部分)はDM方向に大きく延伸・配向し、最大長径が18μmより大きく、平均長径が8μmより大きくなり、長径が非常に大きな層状の相になった。そのため、引き裂き強度は実施例1、2に比べて低い値であった。
(樹脂組成物の製造)
3-ヒドロキシアルカノエート重合体(原料A-3)100重量部に対して、ポリエステルB-1(PBAT)=80重量部、ポリエステルB-2(PBS)=25重量部、変性グリセリン化合物C-1(アセチル化モノグリセライド)=25重量部を、2軸押出機で、設定温度100~130℃、スクリュー回転数100rpmで溶融混錬して、PHBHを含有するポリエステル樹脂組成物を得た。
実施例4は実施例1と同様の方法、実施例5は実施例2と同様の方法でフィルムを成形し、得られたフィルムについて、引き裂き強度、PHBHの相の最大長径および平均長径の算出を実施例1と同様に実施した。
実施例4で得られた樹脂組成物を、実施例3と同様の方法でシート成形し、得られたシートについて、引き裂き強度、PHBHの相の最大長径および平均長径を測定した。
原料A-3の代わりに原料A-4を用いた以外は、実施例4と同様の方法でポリエステル樹脂組成物を得た。
得られたポリエステル樹脂組成物を実施例3と同様の方法でシート成形し、得られたシートについて、引き裂き強度、PHBHの相の最大長径および平均長径を測定した。
原料A-3=100重量部の代わりに原料A-2=100重量部を用いた以外は実施例5と同様にポリエステル樹脂組成物を製造した。
得られたポリエステル樹脂組成物を実施例5と同様の方法でフィルムを成形し、得られたフィルムについて、引き裂き強度、PHBHの相の最大長径および平均長径を算出した。
比較例3で得られた樹脂組成物を、実施例6と同様の方法でシート成形し、得られたシートについて、引き裂き強度、PHBHの相の最大長径および平均長径を測定した。
表3に記載した配合で実施例1と同様にしてポリエステル樹脂組成物を製造し、表3に示す引き取り速度に調整した以外は実施例1と同様の方法でフィルムを成形した。得られたフィルムについて、引き裂き強度、PHBHの相の最大長径および平均長径を算出した。その結果を表3に示す。
変性グリセリン化合物C-1を用いない以外は実施例2と同様に樹脂組成物を製造し、この樹脂組成物からフィルムを成形し、得られたフィルムについて、引き裂き強度、PHBHの相の最大長径および平均長径を算出した。その結果を表4に示す。
変性グリセリン化合物C-1の代わりに、変性グリセリンに類似の化合物としてヒマシ油脂肪酸(伊藤製油社製)を用いた以外は実施例1と同様の配合で樹脂組成物の製造を試みた(表4に組成を記載する)。しかし、原料A-1、原料B-1、B-2のような樹脂と混練しても相溶せずにブリードアウトしてしまい、樹脂組成物を得ることができなった。
3-ヒドロキシアルカノエート重合体(原料A-1)100重量部の代わりに溶融粘度が低い3-ヒドロキシアルカノエート重合体(原料A-2)100重量部を用いた以外は実施例1と同様にして樹脂組成物を製造し、引き取り速度を15m/分にした以外は実施例1と同様にしてフィルムを成形し、引き裂き強度とTEM画像からPHBH相の最大長径と平均長径を算出した。結果を表1に示すが、比較例5と同様に、最大長径、平均長径がいずれも実施例2のものと比べて大きく、引き裂き強度は逆に低くなっていた。
原料A-3=100重量部の代わりに原料A-2=100重量部を用いた以外は実施例4と同様にして樹脂組成物を製造し、引き取り速度を15m/分にした以外は実施例1と同様にしてフィルムを成形した。得られたフィルムのTEM画像を確認したところ、PHBH相が連続相を形成していたため、最大長径と平均長径の算出はできなかった。また、引っ張り強度を測定したところ、5.9mN/μmと非常に低いものであった。これらの結果を表2に示す。
Claims (11)
- 式(1):[-CHR-CH2-CO-O-]
(但し、RはCnH2n+1で表されるアルキル基であり、nは1以上15以下の整数である。)
で示される繰り返し単位を有する脂肪族ポリエステル(P3HA)100重量部に対し、ポリブチレンアジペートテレフタレート(PBAT)を60~400重量部および変性グリセリンを10~50重量部含有してなる生分解性ポリエステル樹脂組成物。 - フィルム成形用またはシート成形用である請求項1に記載の生分解性ポリエステル樹脂組成物。
- 脂肪族ポリエステル(P3HA)が、ポリ(3-ヒドロキシブチレート-コ-3-ヒドロキシヘキサノエート)(PHBH)、ポリ(3-ヒドロキシブチレート)(P3HB)、ポリ(3-ヒドロキシブチレート-コ-3-ヒドロキシバリレート)(PHBV)、ポリ(3-ヒドロキシブチレート-コ-4-ヒドロキシブチレート)(P3HB4HB)、ポリ(3-ヒドロキシブチレート-コ-3-ヒドロキシオクタノエート)、ポリ(3-ヒドロキシブチレート-コ-3-ヒドロキシオクタデカノエート)からなる群から選択される少なくとも1種である請求項1または2に記載の生分解性ポリエステル樹脂組成物。
- 脂肪族ポリエステル(P3HA)の160℃における溶融粘度が900~3,600Pa・sである請求項1~3のいずれかに記載の生分解性ポリエステル樹脂組成物。
- ポリブチレンアジペートテレフタレート(PBAT)の溶融粘度に対する脂肪族ポリエステル(P3HA)の溶融粘度の比(P3HA/PBAT)が0.5以上である請求項1~4のいずれかに記載の生分解性ポリエステル樹脂組成物。
- 脂肪族ポリエステル(P3HA)100重量部に対し、さらにポリブチレンサクシネート(PBS)を20~150重量部含有してなる請求項1~5のいずれかに記載の生分解性ポリエステル樹脂組成物。
- 脂肪族ポリエステル(P3HA)、ポリブチレンアジペートテレフタレート(PBAT)およびポリブチレンサクシネート(PBS)の合計量に対する脂肪族ポリエステル(P3HA)の比率が45重量%以下である請求項6に記載の生分解性ポリエステル樹脂組成物。
- 変性グリセリンが、アセチル化モノグリセライドである請求項1~7のいずれかに記載の生分解性ポリエステル樹脂組成物。
- 請求項1~8のいずれかに記載の生分解性ポリエステル樹脂組成物を成形してなり、
透過型電子顕微鏡分析-画像解析法(TEM法)によって測定される前記脂肪族ポリエステル(P3HA)からなる相の長径の最大値が18μm以下であり、平均値が8μm以下であるフィルムまたはシート。 - インフレーション法またはTダイ押出法により成形される請求項9に記載のフィルムまたはシート。
- 成形時の引き取り速度が30m/分以下である請求項9または10に記載のフィルムまたはシート。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201380016206.7A CN104204092A (zh) | 2012-03-30 | 2013-03-29 | 生物降解性聚酯树脂组合物 |
| EP13769826.2A EP2832796B1 (en) | 2012-03-30 | 2013-03-29 | Biodegradable polyester resin composition |
| US14/388,954 US9371445B2 (en) | 2012-03-30 | 2013-03-29 | Biodegradable polyester resin composition |
| JP2014508084A JP6015748B2 (ja) | 2012-03-30 | 2013-03-29 | 生分解性ポリエステル樹脂組成物 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012082310 | 2012-03-30 | ||
| JP2012-082310 | 2012-03-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013147139A1 true WO2013147139A1 (ja) | 2013-10-03 |
Family
ID=49260381
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/059493 WO2013147139A1 (ja) | 2012-03-30 | 2013-03-29 | 生分解性ポリエステル樹脂組成物 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US9371445B2 (ja) |
| EP (1) | EP2832796B1 (ja) |
| JP (1) | JP6015748B2 (ja) |
| CN (1) | CN104204092A (ja) |
| WO (1) | WO2013147139A1 (ja) |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018012583A1 (ja) | 2016-07-14 | 2018-01-18 | 株式会社カネカ | 脂肪族ポリエステル樹脂組成物の製造方法 |
| WO2018181500A1 (ja) | 2017-03-29 | 2018-10-04 | 株式会社カネカ | 生分解性ポリエステルフィルムの製造方法 |
| WO2019022008A1 (ja) | 2017-07-24 | 2019-01-31 | 株式会社カネカ | ポリ(3-ヒドロキシアルカノエート)樹脂組成物 |
| WO2019146555A1 (ja) | 2018-01-26 | 2019-08-01 | 株式会社カネカ | ポリ(3-ヒドロキシアルカノエート)系発泡粒子およびポリ(3-ヒドロキシアルカノエート)系発泡成形体 |
| WO2019189367A1 (ja) * | 2018-03-30 | 2019-10-03 | 三菱ケミカル株式会社 | 成形体、シート及び容器、並びに管状体、ストロー、綿棒及び風船用スティック |
| JP2019178206A (ja) * | 2018-03-30 | 2019-10-17 | 三菱ケミカル株式会社 | 成形体、シート及び容器 |
| WO2019239913A1 (ja) * | 2018-06-14 | 2019-12-19 | 株式会社カネカ | 生分解性積層体の製造方法 |
| JP2020516712A (ja) * | 2017-04-05 | 2020-06-11 | バイオ−テック ビオローギッシュ ナチューフェアパックンゲン ゲーエムベーハー ウント コンパニ カーゲー | 生分解性フィルム |
| WO2020195550A1 (ja) | 2019-03-28 | 2020-10-01 | 株式会社カネカ | ポリヒドロキシアルカノエート系樹脂組成物、その成形体及びフィルム又はシート |
| WO2020202813A1 (ja) * | 2019-03-29 | 2020-10-08 | 株式会社カネカ | ポリエステル系樹脂組成物、その製造方法及び成形体 |
| WO2021002092A1 (ja) | 2019-07-02 | 2021-01-07 | 株式会社カネカ | ポリ(3-ヒドロキシアルカノエート)系発泡粒子およびポリ(3-ヒドロキシアルカノエート)系発泡成形体 |
| WO2022009717A1 (ja) | 2020-07-07 | 2022-01-13 | 株式会社カネカ | 樹脂チューブ |
| WO2022044836A1 (ja) | 2020-08-25 | 2022-03-03 | 株式会社カネカ | 樹脂フィルム、及び、該樹脂フィルムから形成される袋、手袋、結束材 |
| WO2022065182A1 (ja) | 2020-09-28 | 2022-03-31 | 株式会社カネカ | 射出成形用樹脂組成物および射出成形体 |
| WO2022075233A1 (ja) | 2020-10-07 | 2022-04-14 | 株式会社カネカ | 多層フィルム、および包材 |
| WO2022264944A1 (ja) | 2021-06-16 | 2022-12-22 | 株式会社カネカ | 生分解性積層体及び、その製造方法 |
| WO2023054388A1 (ja) | 2021-09-29 | 2023-04-06 | 株式会社カネカ | ブロー成形体およびその製造方法 |
| WO2023085375A1 (ja) | 2021-11-12 | 2023-05-19 | 株式会社カネカ | 積層体、およびその利用 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2015338664B2 (en) * | 2014-10-27 | 2019-11-14 | Tipa Corp. Ltd | Biodegradable sheets |
| CN109535667A (zh) * | 2018-11-01 | 2019-03-29 | 刘辉 | 一种生物基降解餐具 |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5974148A (ja) * | 1983-09-05 | 1984-04-26 | Japan Crown Cork Co Ltd | 軟質樹脂組成物 |
| JPH10508640A (ja) | 1994-11-15 | 1998-08-25 | ビーエーエスエフ アクチェンゲゼルシャフト | 生分解可能なポリマー、その製造及び生分解可能な成形体の製造のためのその使用 |
| JP2006045365A (ja) | 2004-08-05 | 2006-02-16 | Kaneka Corp | ポリ(3−ヒドロキシブチレート−コ−3−ヒドロキシヘキサノエート)からなるフィルム |
| JP2006525136A (ja) * | 2003-05-08 | 2006-11-09 | ザ プロクター アンド ギャンブル カンパニー | ポリヒドロキシアルカノエートコポリマー及び環境分解性である熱可塑性ポリマーを含む成形又は押出成形物品 |
| JP2007145423A (ja) * | 2005-10-27 | 2007-06-14 | Yoshino Kogyosho Co Ltd | 生分解性樹脂製ヒンジキャップ |
| JP2009073498A (ja) * | 2007-09-19 | 2009-04-09 | Asahi Kasei Home Products Kk | 溶断シール袋及びその製造方法 |
| JP2009227882A (ja) * | 2008-03-25 | 2009-10-08 | Campo Tecnico:Kk | 相溶性を高めることにより、柔軟性と強度を兼ね備えた生分解性フィルムの製造方法とその組成物 |
| WO2009145164A1 (ja) | 2008-05-26 | 2009-12-03 | 株式会社カネカ | 改良されたポリヒドロキシアルカノエート生産微生物及びそれを用いたポリヒドロキシアルカノエートの製造方法 |
| WO2010013483A1 (ja) | 2008-08-01 | 2010-02-04 | 株式会社カネカ | 樹脂組成物及びシート |
| WO2010067543A1 (ja) | 2008-12-09 | 2010-06-17 | 株式会社カネカ | ポリ-3-ヒドロキシアルカン酸の製造方法およびその凝集体 |
| WO2011080623A2 (en) * | 2009-12-31 | 2011-07-07 | Kimberly-Clark Worldwide, Inc. | Natural biopolymer thermoplastic films |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5832647A (ja) * | 1981-08-21 | 1983-02-25 | Riken Vitamin Co Ltd | 熱可塑性樹脂組成物 |
| US8053491B2 (en) * | 2006-08-10 | 2011-11-08 | Kaneka Corporation | Biodegradable resin composition and molded article of the same |
| CN102056985B (zh) * | 2008-05-06 | 2014-02-19 | 梅塔玻利克斯公司 | 生物可降解聚酯掺合物 |
| EP2417179B1 (en) * | 2009-04-06 | 2014-07-02 | Metabolix, Inc. | Method of improving film processing and injection molding of polyhydroxyalkanoate polymers |
| SE534029C2 (sv) * | 2009-07-10 | 2011-04-05 | Billeruds Ab | Biologiskt nedbrytbar vidhäftande film |
-
2013
- 2013-03-29 WO PCT/JP2013/059493 patent/WO2013147139A1/ja active Application Filing
- 2013-03-29 JP JP2014508084A patent/JP6015748B2/ja active Active
- 2013-03-29 EP EP13769826.2A patent/EP2832796B1/en active Active
- 2013-03-29 CN CN201380016206.7A patent/CN104204092A/zh active Pending
- 2013-03-29 US US14/388,954 patent/US9371445B2/en active Active
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5974148A (ja) * | 1983-09-05 | 1984-04-26 | Japan Crown Cork Co Ltd | 軟質樹脂組成物 |
| JPH10508640A (ja) | 1994-11-15 | 1998-08-25 | ビーエーエスエフ アクチェンゲゼルシャフト | 生分解可能なポリマー、その製造及び生分解可能な成形体の製造のためのその使用 |
| JP2006525136A (ja) * | 2003-05-08 | 2006-11-09 | ザ プロクター アンド ギャンブル カンパニー | ポリヒドロキシアルカノエートコポリマー及び環境分解性である熱可塑性ポリマーを含む成形又は押出成形物品 |
| JP2006045365A (ja) | 2004-08-05 | 2006-02-16 | Kaneka Corp | ポリ(3−ヒドロキシブチレート−コ−3−ヒドロキシヘキサノエート)からなるフィルム |
| JP2007145423A (ja) * | 2005-10-27 | 2007-06-14 | Yoshino Kogyosho Co Ltd | 生分解性樹脂製ヒンジキャップ |
| JP2009073498A (ja) * | 2007-09-19 | 2009-04-09 | Asahi Kasei Home Products Kk | 溶断シール袋及びその製造方法 |
| JP2009227882A (ja) * | 2008-03-25 | 2009-10-08 | Campo Tecnico:Kk | 相溶性を高めることにより、柔軟性と強度を兼ね備えた生分解性フィルムの製造方法とその組成物 |
| WO2009145164A1 (ja) | 2008-05-26 | 2009-12-03 | 株式会社カネカ | 改良されたポリヒドロキシアルカノエート生産微生物及びそれを用いたポリヒドロキシアルカノエートの製造方法 |
| WO2010013483A1 (ja) | 2008-08-01 | 2010-02-04 | 株式会社カネカ | 樹脂組成物及びシート |
| WO2010067543A1 (ja) | 2008-12-09 | 2010-06-17 | 株式会社カネカ | ポリ-3-ヒドロキシアルカン酸の製造方法およびその凝集体 |
| WO2011080623A2 (en) * | 2009-12-31 | 2011-07-07 | Kimberly-Clark Worldwide, Inc. | Natural biopolymer thermoplastic films |
Non-Patent Citations (2)
| Title |
|---|
| "Raw material C-1: Acetylated monoglyceride", RIKEN VITAMIN CO., LTD., article "RIKEMAL" |
| J. BATERIOL., vol. 179, 1997, pages 4821 - 4830 |
Cited By (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018012583A1 (ja) | 2016-07-14 | 2018-01-18 | 株式会社カネカ | 脂肪族ポリエステル樹脂組成物の製造方法 |
| WO2018181500A1 (ja) | 2017-03-29 | 2018-10-04 | 株式会社カネカ | 生分解性ポリエステルフィルムの製造方法 |
| JP2020516712A (ja) * | 2017-04-05 | 2020-06-11 | バイオ−テック ビオローギッシュ ナチューフェアパックンゲン ゲーエムベーハー ウント コンパニ カーゲー | 生分解性フィルム |
| JP7139351B2 (ja) | 2017-04-05 | 2022-09-20 | バイオ-テック ビオローギッシュ ナチューフェアパックンゲン ゲーエムベーハー ウント コンパニ カーゲー | 生分解性フィルム |
| WO2019022008A1 (ja) | 2017-07-24 | 2019-01-31 | 株式会社カネカ | ポリ(3-ヒドロキシアルカノエート)樹脂組成物 |
| WO2019146555A1 (ja) | 2018-01-26 | 2019-08-01 | 株式会社カネカ | ポリ(3-ヒドロキシアルカノエート)系発泡粒子およびポリ(3-ヒドロキシアルカノエート)系発泡成形体 |
| WO2019189367A1 (ja) * | 2018-03-30 | 2019-10-03 | 三菱ケミカル株式会社 | 成形体、シート及び容器、並びに管状体、ストロー、綿棒及び風船用スティック |
| JP2019178206A (ja) * | 2018-03-30 | 2019-10-17 | 三菱ケミカル株式会社 | 成形体、シート及び容器 |
| JP7106936B2 (ja) | 2018-03-30 | 2022-07-27 | 三菱ケミカル株式会社 | 成形体、シート及び容器 |
| WO2019239913A1 (ja) * | 2018-06-14 | 2019-12-19 | 株式会社カネカ | 生分解性積層体の製造方法 |
| JPWO2019239913A1 (ja) * | 2018-06-14 | 2021-07-08 | 株式会社カネカ | 生分解性積層体の製造方法 |
| JP7353280B2 (ja) | 2018-06-14 | 2023-09-29 | 株式会社カネカ | 生分解性積層体の製造方法 |
| WO2020195550A1 (ja) | 2019-03-28 | 2020-10-01 | 株式会社カネカ | ポリヒドロキシアルカノエート系樹脂組成物、その成形体及びフィルム又はシート |
| WO2020202813A1 (ja) * | 2019-03-29 | 2020-10-08 | 株式会社カネカ | ポリエステル系樹脂組成物、その製造方法及び成形体 |
| JP7360450B2 (ja) | 2019-03-29 | 2023-10-12 | 株式会社カネカ | ポリエステル系樹脂組成物の製造方法 |
| WO2021002092A1 (ja) | 2019-07-02 | 2021-01-07 | 株式会社カネカ | ポリ(3-ヒドロキシアルカノエート)系発泡粒子およびポリ(3-ヒドロキシアルカノエート)系発泡成形体 |
| WO2022009717A1 (ja) | 2020-07-07 | 2022-01-13 | 株式会社カネカ | 樹脂チューブ |
| WO2022044836A1 (ja) | 2020-08-25 | 2022-03-03 | 株式会社カネカ | 樹脂フィルム、及び、該樹脂フィルムから形成される袋、手袋、結束材 |
| WO2022065182A1 (ja) | 2020-09-28 | 2022-03-31 | 株式会社カネカ | 射出成形用樹脂組成物および射出成形体 |
| WO2022075233A1 (ja) | 2020-10-07 | 2022-04-14 | 株式会社カネカ | 多層フィルム、および包材 |
| WO2022264944A1 (ja) | 2021-06-16 | 2022-12-22 | 株式会社カネカ | 生分解性積層体及び、その製造方法 |
| WO2023054388A1 (ja) | 2021-09-29 | 2023-04-06 | 株式会社カネカ | ブロー成形体およびその製造方法 |
| WO2023085375A1 (ja) | 2021-11-12 | 2023-05-19 | 株式会社カネカ | 積層体、およびその利用 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2013147139A1 (ja) | 2015-12-14 |
| EP2832796A4 (en) | 2015-11-11 |
| US20150073079A1 (en) | 2015-03-12 |
| US9371445B2 (en) | 2016-06-21 |
| CN104204092A (zh) | 2014-12-10 |
| EP2832796A1 (en) | 2015-02-04 |
| EP2832796B1 (en) | 2019-10-30 |
| JP6015748B2 (ja) | 2016-10-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6015748B2 (ja) | 生分解性ポリエステル樹脂組成物 | |
| Rajan et al. | Polyhydroxybutyrate (PHB): a standout biopolymer for environmental sustainability | |
| Yu et al. | Polymer blends and composites from renewable resources | |
| Popa et al. | Polyhydroxybutyrate blends: A solution for biodegradable packaging? | |
| CN111801385B (zh) | 成形体、片材及容器,以及管状体、吸管、棉签及气球用杆 | |
| JP6220340B2 (ja) | ポリエステル樹脂組成物およびその製造方法 | |
| Nesic et al. | Bio-based packaging materials | |
| Garcia-Garcia et al. | Innovative solutions and challenges to increase the use of Poly (3-hydroxybutyrate) in food packaging and disposables | |
| JP6291488B2 (ja) | ポリエステル樹脂組成物および該樹脂組成物を含む成形体 | |
| Kushwah et al. | RETRACTED ARTICLE: Towards understanding polyhydroxyalkanoates and their use | |
| JP6666328B2 (ja) | ポリエステル樹脂組成物及び成形体の製造方法、並びにポリエステル樹脂組成物及び成形体 | |
| Arrieta | Influence of plasticizers on the compostability of polylactic acid | |
| US10093799B2 (en) | Polyester resin composition and polyester resin formed article | |
| EP4271745A1 (en) | A polymer composition having inorganic additive and production method thereof | |
| JP3984440B2 (ja) | 樹脂組成物、フィルム及び廃棄方法 | |
| JP2015052045A (ja) | ポリエステル樹脂組成物、ポリエステル樹脂発泡体及びその製造方法 | |
| JP6172795B2 (ja) | ポリエステル樹脂組成物およびその製造方法、並びに該樹脂組成物から形成される成形体 | |
| JP2016094547A (ja) | 樹脂組成物および成型体 | |
| Lu | Processing and characterization of bio-based composites | |
| Maikrang et al. | Preparation and characterization of enzymatically-treated granular cassava starch and poly (butylene adipate-co-terephthalate) blends | |
| JPWO2016114127A1 (ja) | ポリエステル樹脂組成物およびポリエステル樹脂成形体 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13769826 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2014508084 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14388954 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013769826 Country of ref document: EP |