WO2013118220A1 - リチウムイオン二次電池用負極活物質材料およびその製造方法 - Google Patents
リチウムイオン二次電池用負極活物質材料およびその製造方法 Download PDFInfo
- Publication number
- WO2013118220A1 WO2013118220A1 PCT/JP2012/007799 JP2012007799W WO2013118220A1 WO 2013118220 A1 WO2013118220 A1 WO 2013118220A1 JP 2012007799 W JP2012007799 W JP 2012007799W WO 2013118220 A1 WO2013118220 A1 WO 2013118220A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- negative electrode
- ion secondary
- electrode active
- active material
- lithium
- Prior art date
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G23/00—Compounds of titanium
- C01G23/003—Titanates
- C01G23/005—Alkali titanates
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G45/00—Compounds of manganese
- C01G45/12—Manganates manganites or permanganates
- C01G45/1221—Manganates or manganites with a manganese oxidation state of Mn(III), Mn(IV) or mixtures thereof
- C01G45/1292—Manganates or manganites with a manganese oxidation state of Mn(III), Mn(IV) or mixtures thereof of the type [Mn5O12]n-
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G49/00—Compounds of iron
- C01G49/0018—Mixed oxides or hydroxides
- C01G49/0027—Mixed oxides or hydroxides containing one alkali metal
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/485—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of mixed oxides or hydroxides for inserting or intercalating light metals, e.g. LiTi2O4 or LiTi2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/50—Solid solutions
- C01P2002/52—Solid solutions containing elements as dopants
- C01P2002/54—Solid solutions containing elements as dopants one element only
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/70—Crystal-structural characteristics defined by measured X-ray, neutron or electron diffraction data
- C01P2002/72—Crystal-structural characteristics defined by measured X-ray, neutron or electron diffraction data by d-values or two theta-values, e.g. as X-ray diagram
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/60—Particles characterised by their size
- C01P2004/61—Micrometer sized, i.e. from 1-100 micrometer
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/11—Powder tap density
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Definitions
- the present invention relates to an inorganic material used as a negative electrode active material of a lithium ion secondary battery and a method for producing the same.
- lithium ion secondary batteries have been developed.
- carbon materials have been mainly used as negative electrode active materials for lithium ion secondary batteries.
- a new lithium titanium composite oxide material has been developed and attracts attention.
- a lithium ion secondary battery using LiCoO 2 as a positive electrode active material and Li 4 Ti 5 O 12 as a negative electrode active material has already been put into practical use.
- Li 4 Ti 5 O 12 is a material having a spinel crystal structure, and can be repeatedly occluded or released from Li. Therefore, Li 4 Ti 5 O 12 can be used as an active material of a lithium ion secondary battery. Li 4 Ti 5 O 12 occludes or releases Li at a potential of about 1.5 V with reference to the standard oxidation-reduction potential (Li / Li + ) of lithium. For this reason, when Li 4 Ti 5 O 12 is used as a negative electrode active material for a lithium ion secondary battery, lithium metal is not easily deposited on the negative electrode even when a reaction overvoltage occurs due to rapid charging, etc. A secondary battery is expected to be realized. In addition, Li 4 Ti 5 O 12 has a feature that the cycle characteristics are good because the lattice expansion accompanying charge / discharge is very small.
- Li 4/3 Ti 5 / 3-x Fe x O 4 (0 ⁇ x ⁇ 0.2) in which a part of Ti element of Li 4 Ti 5 O 12 is substituted with Fe is Li 4 It discloses that the electron conductivity is improved as compared with Ti 5 O 12 .
- an object of the present invention is to provide a negative electrode active material for a lithium ion secondary battery that can realize a lithium ion secondary battery having a high packing density when processed into an electrode and high input characteristics.
- the present invention (wherein, x is 0 ⁇ x ⁇ 0.3) Li 4 Ti 5-x Fe x O 12, or, in Li 4 Ti 5-y Mn y O 12 ( wherein, y is 0 ⁇ y
- a negative electrode active material for a lithium ion secondary battery comprising a lithium titanium composite oxide having a composition represented by ⁇ 0.3) and having an average primary particle size of 1 ⁇ m or more.
- the negative electrode active material for a lithium ion secondary battery of the present invention includes a lithium titanium composite oxide having an average primary particle size of 1 ⁇ m or more, thereby increasing the packing density in the electrode when processed into an electrode. be able to.
- the lithium titanium composite oxide contained in the negative electrode active material of the present invention is Li 4 Ti 5-x Fe x O 12 (wherein x is 0 ⁇ x ⁇ 0.3) or Li 4 Ti 5
- ⁇ y Mn y O 12 where y is 0 ⁇ y ⁇ 0.3
- high input characteristics can be realized even when the particle size is increased. Therefore, according to the negative electrode active material of the present invention, both high packing density and high input characteristics can be achieved.
- FIG. 3 is a diagram showing compression density and input characteristics of batteries including active materials of Examples 1 to 7 and Comparative Examples 1 to 5.
- the inventors have described conventional negative electrode actives such as Li 4 Ti 5 O 12 and Li 4/3 Ti 5 / 3-x Fe x O 4 synthesized in the conventional process described in the “Background Art” section. It has been found that there are problems in material materials that need to be improved. Specifically, Li 4 Ti 5 O 12 and Li 4/3 Ti 5 / 3-x Fe x O 4 synthesized by a conventional process have very small primary particles. Therefore, these materials have a problem that the packing density in the electrode is low and the energy density as the electrode is small. In order to solve this problem, that is, to achieve a high packing density, for example, when increasing the particle size of Li 4 Ti 5 O 12 , another problem occurs that input characteristics deteriorate as the particle size increases. did.
- the present inventors have repeatedly studied on the above problems, and after that, the negative electrode active material for a lithium ion secondary battery capable of realizing a lithium ion secondary battery having a high packing density when processed into an electrode and high input characteristics. It came to provide material and its manufacturing method.
- Li 4 Ti 5-x Fe x O 12 (wherein x is 0 ⁇ x ⁇ 0.3) or Li 4 Ti 5-y Mn y O 12 (wherein Provided is a negative electrode active material for a lithium ion secondary battery, which includes a lithium titanium composite oxide having a composition represented by 0 ⁇ y ⁇ 0.3) and having an average primary particle size of 1 ⁇ m or more. .
- the negative electrode active material for a lithium ion secondary battery according to the first aspect includes a lithium-titanium composite oxide having an average primary particle size of 1 ⁇ m or more, thereby filling the electrode when processed into an electrode.
- the density can be increased.
- the lithium titanium composite oxide contained in the negative electrode active material according to the first aspect is Li 4 Ti 5-x Fe x O 12 (wherein x is 0 ⁇ x ⁇ 0.3), or Li By having a composition represented by 4 Ti 5-y Mn y O 12 (wherein y is 0 ⁇ y ⁇ 0.3), high input characteristics can be realized even when the particle size increases. Therefore, the negative electrode active material according to the first aspect can achieve both high packing density and high input characteristics.
- the second aspect of the present invention provides a negative electrode active material for a lithium ion secondary battery, wherein, in the first aspect, x satisfies 0 ⁇ x ⁇ 0.2. According to the negative electrode active material according to the second aspect, the electrode capacity density can be improved.
- a third aspect of the present invention provides a negative electrode active material for a lithium ion secondary battery, wherein, in the first aspect, y satisfies 0 ⁇ y ⁇ 0.2. According to the negative electrode active material according to the third aspect, the electrode capacity density can be improved.
- a fourth aspect of the present invention is the lithium ion secondary battery according to any one of the first to third aspects, wherein an average particle diameter of primary particles of the lithium titanium composite oxide is 1 ⁇ m or more and 5 ⁇ m or less.
- a negative electrode active material is provided. According to the negative electrode active material according to the fourth aspect, it is easier to achieve both high packing density and high input characteristics.
- a fifth aspect of the present invention Uniform mixing of at least one lithium source selected from LiOH, LiOH hydrate and Li 2 CO 3 , titanium oxide, and at least one Fe source selected from Fe 2 O 3 and FeOOH And obtaining a mixture, Baking the mixture at a temperature of 900 ° C. or higher and 1000 ° C. or lower; A method for producing a negative electrode active material for a lithium ion secondary battery is provided.
- a negative electrode active material for a lithium ion secondary battery that can realize a lithium ion secondary battery having a high packing density when processed into an electrode and a high input characteristic can be easily obtained. Can be synthesized.
- the sixth aspect of the present invention is: At least one lithium source selected from LiOH, LiOH hydrate, and Li 2 CO 3 , titanium oxide, at least one Fe source selected from Fe 2 O 3 and FeOOH, and LiCl, KCl A step of uniformly mixing at least one molten salt material selected from Li 2 SO 4 and LiClO 4 to obtain a mixture; Baking the mixture at a temperature of 500 ° C. or higher and 1000 ° C. or lower; Washing the obtained fired product to remove the molten salt material; A method for producing a negative electrode active material for a lithium ion secondary battery is provided.
- a negative electrode active material for a lithium ion secondary battery that can realize a lithium ion secondary battery having a high packing density when processed into an electrode and a high input characteristic can be easily obtained. Can be synthesized.
- the seventh aspect of the present invention is At least one lithium source selected from LiOH, LiOH hydrate, and Li 2 CO 3 , titanium oxide, and at least one Mn source selected from MnO 2 and MnCO 3 are uniformly mixed.
- a mixture Baking the mixture at a temperature of 900 ° C. or higher and 1000 ° C. or lower; A method for producing a negative electrode active material for a lithium ion secondary battery is provided.
- a negative electrode active material for a lithium ion secondary battery that can realize a lithium ion secondary battery having a high packing density when processed into an electrode and a high input characteristic can be easily obtained. Can be synthesized.
- the eighth aspect of the present invention is At least one lithium source selected from LiOH, LiOH hydrate and Li 2 CO 3 , titanium oxide, at least one Mn source selected from MnO 2 and MnCO 3 , LiCl, KCl, A step of uniformly mixing at least one molten salt material selected from Li 2 SO 4 and LiClO 4 to obtain a mixture; Baking the mixture at a temperature of 500 ° C. or higher and 1000 ° C. or lower; Washing the obtained fired product to remove the molten salt material; A method for producing a negative electrode active material for a lithium ion secondary battery is provided.
- a negative electrode active material for a lithium ion secondary battery that can realize a lithium ion secondary battery having a high packing density when processed into an electrode and a high input characteristic can be easily obtained. Can be synthesized.
- a ninth aspect of the present invention there is provided a method for producing a negative electrode active material for a lithium ion secondary battery according to any one of the fifth to eighth aspects, wherein the titanium oxide has an anatase type crystal structure. provide. According to the manufacturing method according to the ninth aspect, since the reaction of titanium oxide having an anatase type crystal structure easily proceeds, the negative electrode active material for a lithium ion secondary battery can be synthesized more easily.
- the negative electrode active material for a lithium ion secondary battery of the present embodiment has a composition represented by Li 4 Ti 5-x Fe x O 12 (wherein x is 0 ⁇ x ⁇ 0.3), and primary particles
- the lithium-titanium composite oxide having an average particle size (hereinafter sometimes referred to as a primary particle size) of 1 ⁇ m or more is included.
- This lithium titanium composite oxide is a substance in which a part of Ti element of Li 4 Ti 5 O 12 is substituted with Fe element, and x in the above formula represents the substitution amount (addition amount) of Fe element. .
- Lithium titanium composite oxide having a composition of Li 4 Ti 5 O 12 wherein the lithium titanium composite oxide having a composition of Li 4 Ti 5-x Fe x O 12 (where x is 0 ⁇ x ⁇ 0.3)
- the inventors of the present application have found that the input characteristics are improved as compared with FIG.
- a lithium titanium composite oxide having a composition of Li 4 Ti 5-x Fe x O 12 (where x is 0 ⁇ x ⁇ 0.3) typically has a primary particle size of 1 ⁇ m or less. It was. Therefore, when the electrode is formed using a lithium titanium composite oxide having a composition of Li 4 Ti 5-x Fe x O 12 (where x is 0 ⁇ x ⁇ 0.3), the packing density is increased. There was a problem that it was not possible.
- the inventors of the present application improved the synthesis process to obtain a lithium titanium composite oxide having a composition of Li 4 Ti 5-x Fe x O 12 (where x is 0 ⁇ x ⁇ 0.3). It has been found that the primary particle size of the product can be increased and the filling property of the electrode of the lithium ion secondary battery can be improved. Further, the lithium titanium composite oxide having a composition of Li 4 Ti 5-x Fe x O 12 (where x is 0 ⁇ x ⁇ 0.3) may have a primary particle diameter of 1 ⁇ m or more. The present inventors have found that the input characteristics are larger than those of conventional Li 4 Ti 5 O 12 . Therefore, when the lithium titanium composite oxide of this embodiment is used as a negative electrode material for a lithium ion secondary battery, it is possible to provide a lithium ion secondary battery that can achieve both high packing density of electrodes and high input characteristics. It becomes.
- the lithium titanium composite oxide contained in the negative electrode active material for a lithium ion secondary battery of this embodiment has a spinel crystal structure.
- the crystal structure can be confirmed by X-ray diffraction (XRD).
- the addition amount x of Fe element satisfies 0 ⁇ x ⁇ 0.3.
- x only needs to be larger than 0.
- the dischargeable capacity of the lithium ion secondary battery using the negative electrode active material of the present embodiment tends to decrease, and x is 0.3. It has been found that the discharge capacity is significantly reduced when the value exceeds. From the viewpoint of electrode capacity density, it is more preferable that the addition amount x of Fe element satisfies 0 ⁇ x ⁇ 0.2.
- the average particle diameter d of the primary particles of the lithium titanium composite oxide of the present embodiment is defined as follows. In the SEM photograph in which the negative electrode active material was photographed at an appropriate magnification using a scanning electron microscope (SEM), 30 particles were arbitrarily selected from the lithium titanium composite oxide particles, and the primary particle size of each particle (The length of the long side of the circumscribed rectangle that circumscribes the boundary of the primary particles and has the smallest area) is measured, and the arithmetic average value of the obtained measured values is defined as the average particle diameter d.
- the lithium titanium composite oxide of the present embodiment may be primary particles or may constitute secondary particles in which the primary particles are aggregated, but in either case, the average particle diameter of the primary particles d ( ⁇ m) satisfies 1 ⁇ m or more. Since the input characteristics tend to be slightly deteriorated when the particle size becomes too large, it is preferable that 1 ⁇ d ⁇ 5. However, depending on the application, the average particle size of primary particles of the lithium titanium composite oxide is 5 ⁇ m or more. There may be.
- the primary particle is a particle constituting a state (powder) or an aggregate in which many particles are present, and is a minimum unit particle that exists without breaking bonds between molecules.
- the primary particles may be single crystal, polycrystal, or amorphous.
- the secondary particles are particles formed by aggregating a plurality of primary particles as described above.
- the lithium-titanium composite oxide of this embodiment can be synthesized by mixing and firing a compound containing constituent elements.
- a solid phase reaction method is generally used in the synthesis of lithium-titanium composite oxide. Specifically, Li source, titanium oxide, and Fe source are weighed at a suitable ratio, and the weighed raw materials are combined and mixed uniformly to obtain a mixture, and the mixture is fired. The manufacturing process can be performed. When additional elements other than Fe are added, the additive element raw materials may be further added to the Li source, titanium oxide and Fe source, mixed and fired.
- LiOH, LiOH hydrate, Li 2 CO 3 , Li 2 SO 4 , LiF, and / or Li 2 O can be used as the Li source.
- hydrate of LiOH monohydrate (LiOH.H 2 O) is generally used, but LiOH hydrate having other water content may be used.
- LiOH, a hydrate of LiOH, and / or Li 2 CO 3 titanium oxide having a rutile type or anatase type crystal structure can be used. From the viewpoint of easy progress of the reaction, it is preferable to use an anatase type crystal structure.
- FeO FeO, Fe 2 O 3 , Fe 3 O 4 , FeO 2 , ⁇ -FeOOH, Fe (OH) 3 , FeSO 4 , Fe 2 (SO 4 ) 3 or the like
- Fe 2 O 3 and / or ⁇ -FeOOH it is preferable to use Fe 2 O 3 and / or ⁇ -FeOOH.
- Calcination may be performed in an air atmosphere, an oxygen atmosphere, or an inert gas atmosphere such as nitrogen and argon.
- the firing temperature depends on the Li source, titanium oxide, Fe source, additive element material, and retention time at the firing temperature, but by firing the mixture at a temperature of about 900 ° C. to 1000 ° C. A lithium titanium composite oxide is obtained.
- Lithium-titanium composite oxide synthesized by the solid-phase reaction method is likely to agglomerate or sinter.
- the particle size distribution is adjusted by performing a pulverization (disintegration) step in addition to the above steps. It is possible.
- a pulverization method a pulverization method using apparatuses and instruments such as a jet mill, a bead mill, a ball mill, a masco loader, and a mortar can be considered.
- the pulverization method using a jet mill is useful because the pulverization is performed by causing particles to collide with each other in an air stream, and therefore, there is little possibility of mixing impurities.
- a material having a small primary particle size and a strong cohesive force is pulverized using a jet mill, the material adheres in the jet mill piping and the efficiency is lowered, so that it takes a very long time and the yield is also poor.
- the lithium titanium composite oxide of this embodiment can also be synthesized by a molten salt synthesis method.
- the molten salt synthesis method unlike the solid phase reaction method, it is possible to synthesize a material with less aggregation and no need for a pulverization step.
- Li source, titanium oxide, Fe source, and molten salt material are weighed at a ratio that makes an appropriate ratio, and the weighed raw materials are combined and uniformly mixed to obtain a mixture And a step of firing the mixture and a step of washing the fired product and removing the molten salt.
- the reaction proceeds in the molten salt that is in a liquid state at the firing temperature, and remains in the molten salt after the reaction, so there is little contact with other particles, and there is little aggregation or sintering of the negative electrode active material Can be synthesized.
- LiOH, LiOH hydrate, Li 2 CO 3 , Li 2 SO 4 , LiF, and / or Li 2 O can be used as the Li source.
- hydrate of LiOH monohydrate (LiOH.H 2 O) is generally used, but LiOH hydrate having other water content may be used.
- LiOH, a hydrate of LiOH, and / or Li 2 CO 3 titanium oxide having a rutile type or anatase type crystal structure can be used. From the viewpoint of easy progress of the reaction, it is preferable to use an anatase type crystal structure.
- FeO, Fe 2 O 3 , Fe 3 O 4 , FeO 2 , ⁇ -FeOOH, Fe (OH) 3 , FeSO 4 , Fe 2 (SO 4 ) 3 or the like can be used.
- the molten salt material may be any compound that has a relatively low melting point and can be removed in the washing step.
- a compound containing Li element is preferable.
- LiCl, Li 2 SO 4 , LiClO 4 and the like can be used.
- KCl, a mixture with LiCl, KCl, or the like can also be used.
- Calcination may be performed in an air atmosphere, an oxygen atmosphere, or an inert gas atmosphere such as nitrogen and argon.
- the firing temperature depends on the Li source, titanium oxide, Fe source, and additive element material used, the lithium titanium composite oxide of this embodiment can be obtained by firing the mixture at a temperature equal to or higher than the melting point of the molten salt used. . For example, it can be in the range of 500 ° C. or higher and 1000 ° C. or lower.
- the fired product can be easily washed by using a liquid that dissolves the molten salt.
- a liquid that dissolves the molten salt For example, when LiCl, KCl or a mixture of LiCl and KCl is used as the molten salt, it can be dissolved in water. In order to prevent mixing of impurity elements into the active material, it is preferable to use pure water or ion exchange water.
- the lithium-titanium composite oxide contained in the negative electrode active material for a lithium ion secondary battery according to this embodiment has a large primary particle size, the packing density of the negative electrode active material in the negative electrode can be increased.
- the lithium titanium composite oxide contained in the negative electrode active material for a lithium ion secondary battery of the present embodiment is Li 4 Ti 5-x Fe x O 12 (wherein x is 0 ⁇ x ⁇ 0.3). By having the composition shown by the above, high input characteristics can be realized even when the particle size is large. Therefore, according to the negative electrode active material for a lithium ion secondary battery of the present embodiment, a lithium ion secondary battery that achieves both high packing density of the negative electrode active material in the negative electrode and high input characteristics can be realized.
- the negative electrode active material for a lithium ion secondary battery of the present embodiment has a composition represented by Li 4 Ti 5-y Mn y O 12 (where y is 0 ⁇ y ⁇ 0.3), and primary particles A lithium titanium composite oxide having an average particle size of 1 ⁇ m or more.
- This lithium titanium composite oxide is a substance in which a part of the Ti element of Li 4 Ti 5 O 12 is substituted with Mn element, and y in the above formula represents the substitution amount (addition amount) of the Mn element. .
- the inventors of the present application have found that the input characteristics are improved as compared with FIG.
- a lithium titanium composite oxide having a composition of Li 4 Ti 5-y Mn y O 12 (where y is 0 ⁇ y ⁇ 0.3) typically has a primary particle size of 1 ⁇ m or less. It was. For this reason, there has been a problem that the packing density cannot be increased when an electrode is formed using a lithium titanium composite oxide.
- the inventors of the present application improved the synthesis process to obtain a lithium titanium composite oxide having a composition of Li 4 Ti 5-y Mn y O 12 (where y is 0 ⁇ y ⁇ 0.3). It has been found that the primary particle size of the product can be increased and the filling property of the electrode of the lithium ion secondary battery can be improved. Further, the lithium titanium composite oxide having a composition of Li 4 Ti 5-y Mn y O 12 (where y is 0 ⁇ y ⁇ 0.3) may have a primary particle size of 1 ⁇ m or more. The present inventors have found that the input characteristics are larger than those of conventional Li 4 Ti 5 O 12 . Therefore, when the lithium titanium composite oxide of this embodiment is used as a negative electrode material for a lithium ion secondary battery, it is possible to provide a lithium ion secondary battery that can achieve both high packing density of the electrode and high input characteristics. It becomes.
- the lithium titanium composite oxide contained in the negative electrode active material for a lithium ion secondary battery of this embodiment has a spinel crystal structure.
- the crystal structure can be confirmed by X-ray diffraction (XRD).
- the addition amount y of Mn element satisfies 0 ⁇ y ⁇ 0.3. For this reason, y should just be larger than 0.
- y should just be larger than 0.
- the dischargeable capacity of the lithium ion secondary battery using the negative electrode active material of this embodiment tends to decrease, and y is 0.3. It has been found that the discharge capacity is significantly reduced when the value exceeds. From the viewpoint of electrode capacity density, it is more preferable that the addition amount y of Mn element satisfies 0 ⁇ y ⁇ 0.2.
- the average particle diameter d of the primary particles in the lithium titanium composite oxide of the present embodiment is the same as defined in the first embodiment.
- the lithium titanium composite oxide of the present embodiment may be primary particles or may constitute secondary particles in which the primary particles are aggregated, but in either case, the average particle diameter of the primary particles d ( ⁇ m) satisfies 1 ⁇ m or more.
- the average particle diameter d is larger than 1.5 ⁇ m.
- the average particle size d is preferably 5 ⁇ m or less. That is, d preferably satisfies 1 ⁇ d ⁇ 5, and more preferably satisfies 1.5 ⁇ d ⁇ 5.
- the average particle diameter of the primary particles of the lithium titanium composite oxide may be 5 ⁇ m or more depending on the application.
- the lithium-titanium composite oxide of this embodiment can be synthesized by mixing and firing a compound containing constituent elements.
- a solid phase reaction method is generally used in the synthesis of lithium-titanium composite oxide. Specifically, Li source, titanium oxide, and Mn source are weighed at a suitable ratio, and the weighed raw materials are combined and mixed uniformly to obtain a mixture, and the mixture is fired. The manufacturing process can be performed. When an additional element is added in addition to Mn, the additive element material may be further added to the Li source, titanium oxide and Mn source, mixed and fired.
- LiOH, LiOH hydrate, Li 2 CO 3 , Li 2 SO 4 , LiF, and / or Li 2 O can be used as the Li source.
- hydrate of LiOH monohydrate (LiOH.H 2 O) is generally used, but LiOH hydrate having other water content may be used.
- LiOH, a hydrate of LiOH, and / or Li 2 CO 3 titanium oxide having a rutile type or anatase type crystal structure can be used. From the viewpoint of easy progress of the reaction, it is preferable to use an anatase type crystal structure.
- MnO, Mn 3 O 4 , MnO 2 , Mn (OH) 2 , MnCO 3 , MnSO 4 or the like can be used. From the viewpoint of the reaction temperature, it is preferable to use MnO 2 and / or MnCO 3 .
- Calcination may be performed in an air atmosphere, an oxygen atmosphere, or an inert gas atmosphere such as nitrogen and argon.
- the firing temperature depends on the Li source to be used, titanium oxide, Mn source, additive element material, and the retention time at the firing temperature, but by firing the mixture at a temperature of about 900 ° C. or more and 1000 ° C. or less, A lithium titanium composite oxide is obtained.
- Lithium-titanium composite oxide synthesized by the solid-phase reaction method is likely to agglomerate or sinter.
- the particle size distribution is adjusted by performing a pulverization (disintegration) step in addition to the above steps. It is possible.
- a pulverization method a pulverization method using apparatuses and instruments such as a jet mill, a bead mill, a ball mill, a masco loader, and a mortar can be considered.
- the pulverization method using a jet mill is useful because the pulverization is performed by causing particles to collide with each other in an air stream, and therefore, there is little possibility of mixing impurities.
- a material having a small primary particle size and a strong cohesive force is pulverized using a jet mill, the material adheres in the jet mill piping and the efficiency is lowered, so that it takes a very long time and the yield is also poor.
- the lithium titanium composite oxide of this embodiment can also be synthesized by a molten salt synthesis method.
- the molten salt synthesis method unlike the solid phase reaction method, it is possible to synthesize a material with less aggregation and no need for a pulverization step. Specifically, Li source, titanium oxide, Mn source, and molten salt material are weighed at a suitable ratio, and the weighed raw materials are combined and mixed uniformly to obtain a mixture And a step of firing the mixture and a step of washing the fired product and removing the molten salt.
- the reaction proceeds in the molten salt that is in the liquid state at the firing temperature, and remains in the molten salt after the reaction, so there is little contact with other particles, and lithium titanium composite oxidation with little aggregation or sintering Products can be synthesized.
- LiOH, LiOH hydrate, Li 2 CO 3 , Li 2 SO 4 , LiF, and / or Li 2 O can be used as the Li source.
- hydrate of LiOH monohydrate (LiOH.H 2 O) is generally used, but LiOH hydrate having other water content may be used.
- LiOH, a hydrate of LiOH, and / or Li 2 CO 3 titanium oxide having a rutile type or anatase type crystal structure can be used. From the viewpoint of easy progress of the reaction, it is preferable to use an anatase type crystal structure.
- MnO, Mn 3 O 4 , MnO 2 , Mn (OH) 2 , MnCO 3 , MnSO 4 or the like can be used. From the viewpoint of the reaction temperature, it is preferable to use MnO 2 and / or MnCO 3 .
- the molten salt material may be any compound that has a relatively low melting point and can be removed in the washing step.
- a compound containing Li element is preferable.
- LiCl, Li 2 SO 4 , LiClO 4 and the like can be used.
- KCl, a mixture with LiCl, KCl, or the like can also be used.
- Calcination may be performed in an air atmosphere, an oxygen atmosphere, or an inert gas atmosphere such as nitrogen and argon.
- the firing temperature depends on the Li source used, titanium oxide, Mn source, and additive element material, but the lithium titanium composite oxide of this embodiment can be obtained by firing the mixture at a temperature equal to or higher than the melting point of the molten salt used. .
- the fired product can be easily washed by using a liquid that dissolves the molten salt.
- a liquid that dissolves the molten salt For example, when LiCl, KCl or a mixture of LiCl and KCl is used as the molten salt, it can be dissolved in water. In order to prevent mixing of impurity elements into the active material, it is preferable to use pure water or ion exchange water.
- the lithium-titanium composite oxide contained in the negative electrode active material for a lithium ion secondary battery according to this embodiment has a large primary particle size, the packing density of the negative electrode active material in the negative electrode can be increased.
- the lithium titanium composite oxide contained in the negative electrode active material for a lithium ion secondary battery of the present embodiment is Li 4 Ti 5-x Mn y O 12 (where y is 0 ⁇ y ⁇ 0.3).
- Example 1 The lithium titanium composite oxide of Example 1 was synthesized using a solid phase reaction method.
- a raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 950 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Example 2 The lithium titanium composite oxide of Example 2 was synthesized using a solid phase reaction method.
- Mix in a mortar A raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 950 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Example 3 The lithium titanium composite oxide of Example 3 was synthesized using a solid phase reaction method.
- a raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 900 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Example 4 The lithium titanium composite oxide of Example 4 was synthesized using a solid phase reaction method.
- a raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 900 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Example 5 The lithium titanium composite oxide of Example 5 was synthesized using a solid phase reaction method.
- Mix in a mortar A raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 900 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Example 6 The lithium titanium composite oxide of Example 6 was synthesized using a solid phase reaction method.
- Mix in a mortar A raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 900 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Example 7 The lithium titanium composite oxide of Example 7 was synthesized using a solid phase reaction method.
- Mix in a mortar A raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 900 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Comparative Example 1 The lithium titanium composite oxide of Comparative Example 1 was synthesized using a solid phase reaction method.
- the raw material powders of LiOH.H 2 O and TiO 2 were weighed so that the Li / Ti molar mixing ratio was 4/5, and these were put in a mortar and mixed.
- a raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 850 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Comparative Example 2 The lithium titanium composite oxide of Comparative Example 2 was synthesized using a solid phase reaction method.
- a raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 850 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Comparative Example 3 The lithium titanium composite oxide of Comparative Example 3 was synthesized using a solid phase reaction method.
- Mix in a mortar A raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 850 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Comparative Example 4 The lithium titanium composite oxide of Comparative Example 4 was synthesized using a solid phase reaction method.
- the raw material powders of LiOH.H 2 O and TiO 2 were weighed so that the Li / Ti molar mixing ratio was 4/5, and these were put in a mortar and mixed.
- a raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 950 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
- Comparative Example 5 The lithium titanium composite oxide of Comparative Example 5 was synthesized using a solid phase reaction method.
- the raw material powders of LiOH.H 2 O and TiO 2 were weighed so that the Li / Ti molar mixing ratio was 4/5, and these were put in a mortar and mixed.
- a raw material TiO 2 having an anatase type crystal structure and an average particle diameter of about 0.3 ⁇ m was used.
- the mixed raw material powder was placed in an Al 2 O 3 crucible and fired in an electric furnace in an air atmosphere. The firing temperature was 900 ° C., and the firing temperature was maintained for 12 hours. The fired material was taken out from the crucible and pulverized in a mortar to obtain the target lithium titanium composite oxide.
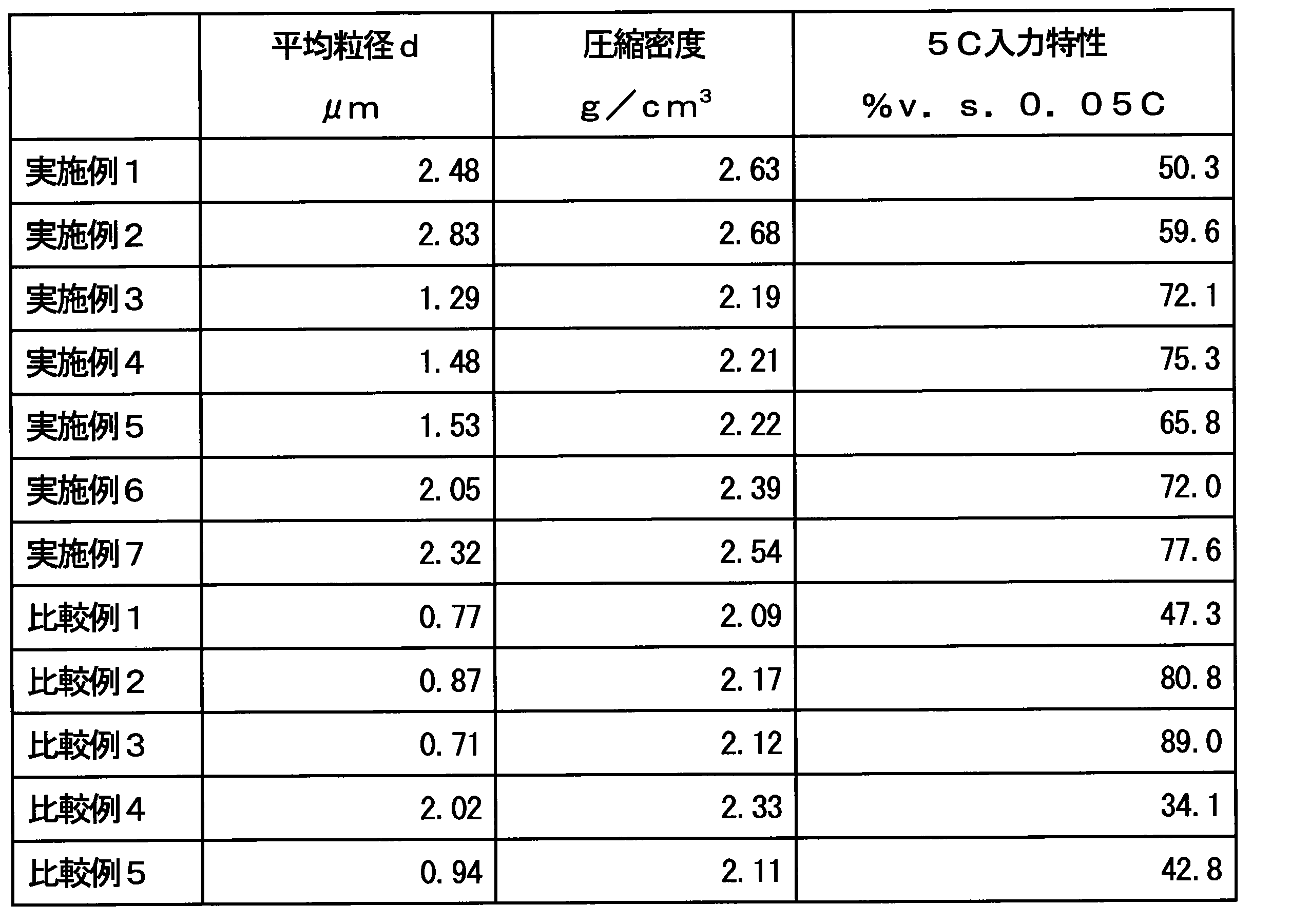
- Table 1 summarizes the addition amount x of Fe and the addition amount y of Mn, and the firing temperature in each Example and Comparative Example.
- “cumulative average particle diameter d 50 ” in particle size distribution measurement is often used as the average particle diameter.
- the particle size distribution measurement measures not the size of primary particles but the size of aggregated particles (secondary particles). There is no correlation between the size of primary particles and the size of secondary particles. Therefore, in order to show the effect of the present invention, “average particle diameter d” which is a unit indicating the size of primary particles is appropriate.
- Table 2 shows the average particle diameters d of the lithium titanium composite oxides of Examples 1 to 7 and Comparative Examples 1 to 5 calculated from SEM photographs.
- the average particle diameter d of the lithium-titanium composite oxide of Comparative Example 2 having a firing temperature of 850 ° C. is The average particle diameter d of the lithium titanium composite oxide of Example 4 having a firing temperature of 900 ° C. is 1.48 ⁇ m and the lithium titanium composite oxide of Example 1 having a firing temperature of 950 ° C. is 0.87 ⁇ m.
- the average particle diameter d was 2.48 ⁇ m.
- the average particle diameter of the lithium titanium composite oxide of Comparative Example 3 having a firing temperature of 850 ° C.
- d is 0.71 ⁇ m
- the average particle diameter d of the lithium titanium composite oxide of Example 7 having a firing temperature of 900 ° C. is 2.32 ⁇ m
- the lithium titanium composite oxide of Example 2 having a firing temperature of 950 ° C.
- the average particle diameter d of the product was 2.83 ⁇ m. From these results, it became clear that increasing the firing temperature has the effect of increasing the primary particle size.
- Table 2 shows the measurement results. From the results of the average particle diameter d and the compression density, it was revealed that those having a large average particle diameter d tend to have a large compression density.
- Electrodes were produced using the lithium titanium composite oxides of Examples 1 to 7 and Comparative Examples 1 to 5 as active materials.
- the active material / conductive material / binder was weighed to a weight ratio of 100/15/5 and kneaded to prepare a paste.
- Acetylene black was used as the conductive material
- PVdF polyvinylidene fluoride
- NMP N-methyl-2-pyrrolidone
- “Awatori Nertaro” manufactured by Shinky Corporation was used.
- the prepared paste was applied onto an aluminum foil current collector and dried in an electric furnace maintained at 100 ° C. After drying, it was rolled with a roller and punched into pellet-shaped electrodes.
- a battery was produced using the above electrode.
- a lithium transition metal composite oxide such as LiCoO 2 (generally containing a transition metal such as Co, Mn, Ni) is used as a positive electrode active material.
- metal Li was used instead of a general positive electrode active material. Such a method is often used to evaluate an active material.
- a coin-shaped battery was produced.
- the above-described electrode produced using this example and comparative example, a separator impregnated with an electrolyte, and a metal Li plate were stacked in this order, placed in a coin-shaped case, and sealed to obtain a battery.
- As the separator two PE microporous films manufactured by Asahi Kasei E-Materials Co., Ltd. were used.
- Batteries produced using the lithium titanium composite oxides of Examples 1 to 7 and Comparative Examples 1 to 5 as active materials are referred to as batteries containing the active materials of Examples 1 to 7 and Comparative Examples 1 to 5, respectively.
- Table 2 shows the measurement results of the input characteristics. From the measurement results of the batteries containing the active materials of Comparative Examples 1 to 3 and 5, it can be seen that the input characteristics are greatly improved when a part of Ti element is replaced with Fe element or Mn element in the conventional particle size. Further, as can be seen from the measurement results of the batteries containing the active materials of Comparative Examples 1, 4, and 5, the input characteristics are deteriorated when the firing temperature is increased. This is because Li 4 Ti 5 O 12 has a small conductivity of electrons or Li ions, so if the particle size increases rapidly, it takes time to diffuse electrons or Li ions in the particles, and the input characteristics under high load are It seems that it is getting worse.
- the negative electrode active material for a lithium ion secondary battery of the present invention can provide a lithium ion secondary battery that can realize a high packing density when processed into an electrode and has excellent input characteristics. Therefore, the negative electrode active material for lithium ion secondary batteries of the present invention is useful as a negative electrode active material for lithium ion secondary batteries for applications such as large batteries and electric vehicles. It can also be applied to mobile applications.
Abstract
Description
LiOH、LiOH水和物およびLi2CO3から選ばれる少なくともいずれか1種のリチウム源と、酸化チタンと、Fe2O3およびFeOOHから選ばれる少なくともいずれか1種のFe源とを均一に混合して混合物を得る工程と、
前記混合物を900℃以上1000℃以下の温度で焼成する工程と、
を包含する、リチウムイオン二次電池用負極活物質材料の製造方法を提供する。
LiOH、LiOH水和物およびLi2CO3から選ばれる少なくともいずれか1種のリチウム源と、酸化チタンと、Fe2O3およびFeOOHから選ばれる少なくともいずれか1種のFe源と、LiCl、KCl、Li2SO4およびLiClO4から選ばれる少なくともいずれか1種の溶融塩材料とを均一に混合して混合物を得る工程と、
前記混合物を500℃以上1000℃以下の温度で焼成する工程と、
得られた焼成物を洗浄して、前記溶融塩材料を除去する工程と、
を包含する、リチウムイオン二次電池用負極活物質材料の製造方法を提供する。
LiOH、LiOH水和物およびLi2CO3から選ばれる少なくともいずれか1種のリチウム源と、酸化チタンと、MnO2およびMnCO3から選ばれる少なくともいずれか1種のMn源とを均一に混合して混合物を得る工程と、
前記混合物を900℃以上1000℃以下の温度で焼成する工程と、
を包含する、リチウムイオン二次電池用負極活物質材料の製造方法を提供する。
LiOH、LiOH水和物およびLi2CO3から選ばれる少なくともいずれか1種のリチウム源と、酸化チタンと、MnO2およびMnCO3から選ばれる少なくともいずれか1種のMn源と、LiCl、KCl、Li2SO4およびLiClO4から選ばれる少なくともいずれか1種の溶融塩材料とを均一に混合して混合物を得る工程と、
前記混合物を500℃以上1000℃以下の温度で焼成する工程と、
得られた焼成物を洗浄して、前記溶融塩材料を除去する工程と、
を包含する、リチウムイオン二次電池用負極活物質材料の製造方法を提供する。
本発明のリチウムイオン二次電池用負極活物質材料の第1の実施形態を説明する。本実施形態のリチウムイオン二次電池用負極活物質材料は、Li4Ti5-xFexO12(式中、xは0<x≦0.3)で示される組成を有し、一次粒子の平均粒径(以下、一次粒子径と記載することがある。)が1μm以上であるリチウムチタン複合酸化物を含む。このリチウムチタン複合酸化物は、Li4Ti5O12のTi元素の一部がFe元素で置換された物質であり、上記式中のxはFe元素の置換量(添加量)を表している。
本発明のリチウムイオン二次電池用負極活物質材料の第2の実施形態を説明する。本実施形態のリチウムイオン二次電池用負極活物質材料は、Li4Ti5-yMnyO12(式中、yは0<y≦0.3)で示される組成を有し、一次粒子の平均粒径が1μm以上であるリチウムチタン複合酸化物を含む。このリチウムチタン複合酸化物は、Li4Ti5O12のTi元素の一部がMn元素で置換された物質であり、上記式中のyはMn元素の置換量(添加量)を表している。
(実施例1)
実施例1のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
実施例2のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
実施例3のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
実施例4のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
実施例5のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
実施例6のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
実施例7のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
比較例1のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
比較例2のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
比較例3のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
比較例4のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
比較例5のリチウムチタン複合酸化物は、固相反応法を用いて合成された。
実施例1~7および比較例1~5のリチウムチタン複合酸化物の結晶構造を確認するため、粉末X線回折(XRD)測定を行った。測定には、リガク社製のXRD測定装置を使用した。
実施例1~7および比較例1~5のリチウムチタン複合酸化物の一次粒子の平均粒径を、以下に定義する「平均粒径d」で評価した。SEM写真から任意に30粒子を選び、それらの粒子の一次粒子の粒径(一次粒子の境界に外接する、面積が最小となる外接長方形の長辺の長さ)を計測し、その平均を算出して、「平均粒径d」を見積もった。
実施例1~7および比較例1~5のリチウムチタン複合酸化物について、電極に加工された際の充填性の指標として、圧縮密度を測定した。測定には三菱化学アナリテック社製の粉体抵抗測定システムを使用した。64MPaの圧力を印加したときの密度を圧縮密度とした。
実施例1~7および比較例1~5のリチウムチタン複合酸化物を活物質として用いて、電極を作製した。活物質/導電材/バインダーを100/15/5の重量比になるよう秤量し、練合してペーストを作製した。導電材にはアセチレンブラック、バインダーにはポリフッ化ビニリデン(PVdF)、溶媒にはN-メチル‐2‐ピロリドン(NMP)を用いた。練合にはシンキー社製の「あわとり練太郎」を用いた。作製したペーストをアルミニウム箔の集電体上に塗工し、100℃に保持した電気炉内で乾燥させた。乾燥後、ローラーで圧延し、打ち抜いてペレット状の電極にした。
リチウムイオン二次電池の負極活物質としての特性を調べるために、上記電極を用いて、電池を作製した。一般にリチウムイオン二次電池では、正極活物質にLiCoO2などのリチウム遷移金属複合酸化物(Co、Mn、Niなどの遷移金属を含むものが一般的である)を用いる。しかしながら、ここでは、正極活物質に依存しない、負極活物質そのものの特性を調べるために、対極に一般の正極活物質ではなく、金属Liを用いた。このような方法は、活物質の評価をするのによく用いられる。
入力特性を調べるために、作製した上記電池を用いて充電レート特性の評価を行った。ソーラトロン社製の電気化学測定システムを使用し、充放電の電圧範囲は1Vから3Vとした。1Cレートでの定電流放電の後に、3Vの定電圧で0.05Cレートの電流値になるまで放電した。放電後の充電時の電流レートは、0.05Cもしくは5Cとして、それぞれ測定した。5Cレートでの充電可能容量を0.05Cレートでの充電可能容量と比較した。
Claims (9)
- Li4Ti5-xFexO12(式中、xは0<x≦0.3)、または、Li4Ti5-yMnyO12(式中、yは0<y≦0.3)で示される組成を有し、一次粒子の平均粒径が1μm以上であるリチウムチタン複合酸化物を含む、
リチウムイオン二次電池用負極活物質材料。 - 前記xは0<x≦0.2を満たす、
請求項1に記載のリチウムイオン二次電池用負極活物質材料。 - 前記yは0<y≦0.2を満たす、
請求項1に記載のリチウムイオン二次電池用負極活物質材料。 - 前記リチウムチタン複合酸化物の一次粒子の平均粒子径は、1μm以上5μm以下である、
請求項1から3のいずれか1項に記載のリチウムイオン二次電池用負極活物質材料。 - LiOH、LiOH水和物およびLi2CO3から選ばれる少なくともいずれか1種のリチウム源と、酸化チタンと、Fe2O3およびFeOOHから選ばれる少なくともいずれか1種のFe源とを均一に混合して混合物を得る工程と、
前記混合物を900℃以上1000℃以下の温度で焼成する工程と、
を包含する、リチウムイオン二次電池用負極活物質材料の製造方法。 - LiOH、LiOH水和物およびLi2CO3から選ばれる少なくともいずれか1種のリチウム源と、酸化チタンと、Fe2O3およびFeOOHから選ばれる少なくともいずれか1種のFe源と、LiCl、KCl、Li2SO4およびLiClO4から選ばれる少なくともいずれか1種の溶融塩材料とを均一に混合して混合物を得る工程と、
前記混合物を500℃以上1000℃以下の温度で焼成する工程と、
得られた焼成物を洗浄して、前記溶融塩材料を除去する工程と、
を包含する、リチウムイオン二次電池用負極活物質材料の製造方法。 - LiOH、LiOH水和物およびLi2CO3から選ばれる少なくともいずれか1種のリチウム源と、酸化チタンと、MnO2およびMnCO3から選ばれる少なくともいずれか1種のMn源とを均一に混合して混合物を得る工程と、
前記混合物を900℃以上1000℃以下の温度で焼成する工程と、
を包含する、リチウムイオン二次電池用負極活物質材料の製造方法。 - LiOH、LiOH水和物およびLi2CO3から選ばれる少なくともいずれか1種のリチウム源と、酸化チタンと、MnO2およびMnCO3から選ばれる少なくともいずれか1種のMn源と、LiCl、KCl、Li2SO4およびLiClO4から選ばれる少なくともいずれか1種の溶融塩材料とを均一に混合して混合物を得る工程と、
前記混合物を500℃以上1000℃以下の温度で焼成する工程と、
得られた焼成物を洗浄して、前記溶融塩材料を除去する工程と、
を包含する、リチウムイオン二次電池用負極活物質材料の製造方法。 - 前記酸化チタンは、アナターゼ型の結晶構造を有する、
請求項5から8のいずれか1項に記載のリチウムイオン二次電池用負極活物質材料の製造方法。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201280017680.7A CN103477474B (zh) | 2012-02-10 | 2012-12-05 | 锂离子二次电池用负极活性物质材料及其制造方法 |
| EP12867857.0A EP2814095B1 (en) | 2012-02-10 | 2012-12-05 | Negative electrode active material for lithium ion secondary batteries and method for producing same |
| JP2013546478A JP5456218B2 (ja) | 2012-02-10 | 2012-12-05 | リチウムイオン二次電池用負極活物質材料 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201261597536P | 2012-02-10 | 2012-02-10 | |
| US61/597,536 | 2012-02-10 | ||
| US201261680916P | 2012-08-08 | 2012-08-08 | |
| US61/680,916 | 2012-08-08 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013118220A1 true WO2013118220A1 (ja) | 2013-08-15 |
Family
ID=48945819
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/007799 WO2013118220A1 (ja) | 2012-02-10 | 2012-12-05 | リチウムイオン二次電池用負極活物質材料およびその製造方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US8962187B2 (ja) |
| EP (1) | EP2814095B1 (ja) |
| JP (1) | JP5456218B2 (ja) |
| CN (1) | CN103477474B (ja) |
| WO (1) | WO2013118220A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015045254A1 (ja) * | 2013-09-25 | 2015-04-02 | 三洋電機株式会社 | リチウムチタン複合酸化物 |
| JP2019079773A (ja) * | 2017-10-27 | 2019-05-23 | 株式会社東芝 | 非水電解質電池及びバッテリーシステム |
| JPWO2019009432A1 (ja) * | 2017-07-07 | 2020-04-09 | 株式会社村田製作所 | 二次電池、電池パック、電動車両、電力貯蔵システム、電動工具および電子機器 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115386947B (zh) * | 2022-08-17 | 2023-11-28 | 甘肃睿斯科锂电材料有限公司 | 一种单晶三元正极材料的制备方法 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000156229A (ja) * | 1998-11-20 | 2000-06-06 | Yuasa Corp | 非水電解質リチウム二次電池 |
| JP2001185141A (ja) | 1999-12-22 | 2001-07-06 | Kyocera Corp | リチウム電池 |
| JP2001196061A (ja) * | 2000-01-11 | 2001-07-19 | Yuasa Corp | 非水電解質リチウム二次電池 |
| JP2002008658A (ja) * | 2000-06-27 | 2002-01-11 | Toyota Central Res & Dev Lab Inc | リチウム二次電池電極活物質用リチウムチタン複合酸化物およびその製造方法 |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2003297433A (ja) * | 2002-03-28 | 2003-10-17 | Hitachi Maxell Ltd | 電気化学素子 |
| JP4061648B2 (ja) * | 2003-04-11 | 2008-03-19 | ソニー株式会社 | 非水電解質二次電池用正極活物質及びそれを用いた非水電解質二次電池 |
| CN101485015A (zh) * | 2006-06-05 | 2009-07-15 | T/J技术公司 | 碱金属钛酸盐及它们的合成方法 |
| CN102362375B (zh) * | 2010-03-15 | 2013-02-27 | 松下电器产业株式会社 | 非水电解质二次电池用电极及含有该电极的非水电解质二次电池 |
| US20120164530A1 (en) * | 2010-06-30 | 2012-06-28 | Hiroshi Temmyo | Negative electrode for nonaqueous electrolyte secondary battery, method for producing same, and nonaqueous electrolyte secondary battery |
| KR20120017991A (ko) * | 2010-08-20 | 2012-02-29 | 삼성에스디아이 주식회사 | 리튬 이차 전지용 음극 활물질, 이의 제조 방법 및 이를 포함하는 리튬 이차 전지 |
| CN102468485B (zh) * | 2010-11-04 | 2014-06-11 | 海洋王照明科技股份有限公司 | 一种钛酸锂复合材料、其制备方法和应用 |
| US9065148B2 (en) * | 2011-02-15 | 2015-06-23 | Panasonic Intellectual Property Management Co., Ltd. | Negative electrode active material for lithium ion secondary battery and method for producing the same |
| US8920976B2 (en) * | 2011-04-20 | 2014-12-30 | Panasonic Corporation | Nonaqueous electrolyte secondary battery |
-
2012
- 2012-12-05 EP EP12867857.0A patent/EP2814095B1/en active Active
- 2012-12-05 CN CN201280017680.7A patent/CN103477474B/zh active Active
- 2012-12-05 JP JP2013546478A patent/JP5456218B2/ja active Active
- 2012-12-05 WO PCT/JP2012/007799 patent/WO2013118220A1/ja active Application Filing
- 2012-12-11 US US13/711,045 patent/US8962187B2/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000156229A (ja) * | 1998-11-20 | 2000-06-06 | Yuasa Corp | 非水電解質リチウム二次電池 |
| JP2001185141A (ja) | 1999-12-22 | 2001-07-06 | Kyocera Corp | リチウム電池 |
| JP2001196061A (ja) * | 2000-01-11 | 2001-07-19 | Yuasa Corp | 非水電解質リチウム二次電池 |
| JP2002008658A (ja) * | 2000-06-27 | 2002-01-11 | Toyota Central Res & Dev Lab Inc | リチウム二次電池電極活物質用リチウムチタン複合酸化物およびその製造方法 |
Non-Patent Citations (3)
| Title |
|---|
| A.D.ROBERTSON ET AL.: "New inorganic spinel oxides for use as negative electrode materials in future lithium-ion batteries", JOURNAL OF POWER SOURCES, vol. 81, no. 82, 1999, pages 352 - 357, XP004363177 * |
| PIERRE KUBIAK ET AL.: "Phase transition in the spinel Li4Ti5012 induced by lithium insertion Influence of the substitutions Ti/V, Ti/Mn, Ti/Fe", JOURNAL OF POWER SOURCES, vol. 119-121, 2003, pages 626 - 630, XP004430243 * |
| See also references of EP2814095A4 |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015045254A1 (ja) * | 2013-09-25 | 2015-04-02 | 三洋電機株式会社 | リチウムチタン複合酸化物 |
| JPWO2019009432A1 (ja) * | 2017-07-07 | 2020-04-09 | 株式会社村田製作所 | 二次電池、電池パック、電動車両、電力貯蔵システム、電動工具および電子機器 |
| JP2019079773A (ja) * | 2017-10-27 | 2019-05-23 | 株式会社東芝 | 非水電解質電池及びバッテリーシステム |
| JP7282482B2 (ja) | 2017-10-27 | 2023-05-29 | 株式会社東芝 | バッテリーシステム |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2814095A1 (en) | 2014-12-17 |
| EP2814095A4 (en) | 2015-07-08 |
| CN103477474B (zh) | 2016-04-20 |
| CN103477474A (zh) | 2013-12-25 |
| US8962187B2 (en) | 2015-02-24 |
| JP5456218B2 (ja) | 2014-03-26 |
| JPWO2013118220A1 (ja) | 2015-05-11 |
| US20130209884A1 (en) | 2013-08-15 |
| EP2814095B1 (en) | 2018-10-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10790511B2 (en) | Lithium cobalt oxide positive electrode material, method for preparing same, and lithium-ion secondary battery | |
| JP5880426B2 (ja) | ニッケル複合水酸化物及びその製造方法、並びに正極活物質の製造方法 | |
| WO2018043669A1 (ja) | 非水系電解質二次電池用正極活物質とその製造方法、および非水系電解質二次電池 | |
| JP5590337B2 (ja) | マンガン複合水酸化物粒子、非水系電解質二次電池用正極活物質、および非水系電解質二次電池と、それらの製造方法 | |
| JP5877817B2 (ja) | 非水系二次電池用正極活物質及びその正極活物質を用いた非水系電解質二次電池 | |
| JP6252384B2 (ja) | ニッケル複合水酸化物及びその製造方法、正極活物質及びその製造方法、並びに非水系電解質二次電池 | |
| WO2012165654A1 (ja) | 非水系二次電池用正極活物質及びその製造方法、並びにその正極活物質を用いた非水系電解質二次電池 | |
| JP5987401B2 (ja) | 非水系電解質二次電池用正極活物質とその製造方法および二次電池 | |
| JP2013147416A (ja) | ニッケル複合水酸化物とその製造方法、非水系電解質二次電池用正極活物質とその製造方法、および非水系電解質二次電池 | |
| JP2012254889A (ja) | ニッケルマンガン複合水酸化物粒子とその製造方法、非水系電解質二次電池用正極活物質とその製造方法、ならびに、非水系電解質二次電池 | |
| JP6346448B2 (ja) | 非水系電解質二次電池用正極活物質、および、非水系電解質二次電池 | |
| JP2005011713A (ja) | リチウム二次電池用正極材およびその製造方法 | |
| JP6201146B2 (ja) | 非水系電解質二次電池用正極活物質の製造方法、非水系電解質二次電池用正極活物質および非水系電解質二次電池 | |
| JP5456218B2 (ja) | リチウムイオン二次電池用負極活物質材料 | |
| JP4628704B2 (ja) | リチウム二次電池用正極材およびその製造方法 | |
| JP5148781B2 (ja) | リチウムイオン二次電池用負極活物質材料およびその製造方法 | |
| JP2014167873A (ja) | リチウムイオン二次電池用負極活物質、その製造方法及びリチウムイオン二次電池 | |
| JP6511761B2 (ja) | 非水系電解質二次電池用正極活物質用の被覆複合酸化物粒子の製造方法及び当該製造方法によって製造される被覆複合酸化物粒子を用いた非水系電解質二次電池 | |
| JP6020700B2 (ja) | 正極活物質及びその製造方法、並びに非水系電解質二次電池 | |
| JP2004186149A (ja) | Liイオン二次電池用正極材料 | |
| JP5070366B2 (ja) | リチウムイオン二次電池用負極活物質材料およびその製造方法 | |
| JP5354112B2 (ja) | 非水系二次電池用正極活物質の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201280017680.7 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12867857 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2013546478 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012867857 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |