WO2013099730A1 - 不溶性アゾ顔料であるc.i.ピグメントイエロー74及びそれを用いた着色組成物 - Google Patents
不溶性アゾ顔料であるc.i.ピグメントイエロー74及びそれを用いた着色組成物 Download PDFInfo
- Publication number
- WO2013099730A1 WO2013099730A1 PCT/JP2012/082899 JP2012082899W WO2013099730A1 WO 2013099730 A1 WO2013099730 A1 WO 2013099730A1 JP 2012082899 W JP2012082899 W JP 2012082899W WO 2013099730 A1 WO2013099730 A1 WO 2013099730A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- pigment
- peak
- particles
- coloring composition
- powder
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B29/00—Monoazo dyes prepared by diazotising and coupling
- C09B29/32—Monoazo dyes prepared by diazotising and coupling from coupling components containing a reactive methylene group
- C09B29/33—Aceto- or benzoylacetylarylides
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B51/00—Nitro or nitroso dyes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B41—PRINTING; LINING MACHINES; TYPEWRITERS; STAMPS
- B41J—TYPEWRITERS; SELECTIVE PRINTING MECHANISMS, i.e. MECHANISMS PRINTING OTHERWISE THAN FROM A FORME; CORRECTION OF TYPOGRAPHICAL ERRORS
- B41J2/00—Typewriters or selective printing mechanisms characterised by the printing or marking process for which they are designed
- B41J2/005—Typewriters or selective printing mechanisms characterised by the printing or marking process for which they are designed characterised by bringing liquid or particles selectively into contact with a printing material
- B41J2/01—Ink jet
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B29/00—Monoazo dyes prepared by diazotising and coupling
- C09B29/10—Monoazo dyes prepared by diazotising and coupling from coupling components containing hydroxy as the only directing group
- C09B29/12—Monoazo dyes prepared by diazotising and coupling from coupling components containing hydroxy as the only directing group of the benzene series
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0001—Post-treatment of organic pigments or dyes
- C09B67/0022—Wet grinding of pigments
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0025—Crystal modifications; Special X-ray patterns
- C09B67/0028—Crystal modifications; Special X-ray patterns of azo compounds
- C09B67/0029—Crystal modifications; Special X-ray patterns of azo compounds of monoazo compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D11/00—Inks
- C09D11/30—Inkjet printing inks
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D11/00—Inks
- C09D11/30—Inkjet printing inks
- C09D11/32—Inkjet printing inks characterised by colouring agents
- C09D11/322—Pigment inks
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/41—Organic pigments; Organic dyes
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03G—ELECTROGRAPHY; ELECTROPHOTOGRAPHY; MAGNETOGRAPHY
- G03G9/00—Developers
- G03G9/08—Developers with toner particles
- G03G9/09—Colouring agents for toner particles
- G03G9/0906—Organic dyes
- G03G9/091—Azo dyes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/29—Coated or structually defined flake, particle, cell, strand, strand portion, rod, filament, macroscopic fiber or mass thereof
- Y10T428/2982—Particulate matter [e.g., sphere, flake, etc.]
Definitions
- the present invention relates to C.I. which is an insoluble azo pigment excellent in weather resistance (light) and the like.
- I. This invention relates to Pigment Yellow 74 and a coloring composition using the same.
- ink jet ink As one of the required performances for ink for ink jet recording (ink jet ink), it is possible to form an image with excellent transparency and sharpness. For this reason, conventionally, a dye capable of forming an image having excellent transparency and sharpness has been used as a color material for an ink used in an OA inkjet printer.
- inkjet inks are used to obtain printed materials installed outdoors, such as posters for outdoor display and printed materials for building materials.
- an image recorded using a dye ink containing a dye as a coloring material has a problem that color fading or color skip is likely to occur particularly when installed outdoors. Under these circumstances, there is an increasing demand for pigment inks containing pigments having excellent weather resistance (light) resistance as color materials even for inkjet recording.
- the colorant for inkjet ink the following pigments such as yellow, magenta, and cyan are used.
- magenta pigments and cyan pigments medium and high-grade pigments having excellent weather resistance (light) properties such as quinacridone pigments and phthalocyanine pigments are mainly used.
- examples of the yellow pigment include C.I., which is an insoluble azo yellow pigment, mainly from the viewpoints of production cost, ease of production, transparency and clarity.
- I. Pigment Yellow 74 is used.
- azo pigments such as C.I. I. Pigment Yellow 74 has a problem that its weather resistance (light) is insufficient as compared with medium to high grade pigments used as magenta pigments and cyan pigments.
- Pigment Yellow 74 can be synthesized, for example, by a coupling reaction in which a diazonium salt obtained by diazotizing an aromatic amine compound and a coupling component are reacted.
- a coupling reaction in which a diazonium salt obtained by diazotizing an aromatic amine compound and a coupling component are reacted.
- a main raw material aromatic amine compound (diazonium salt) or coupling component is added to a diazonium salt solution or a submerged solution at the time of the coupling reaction in the same manner as a conventionally known azo pigment synthesis method.
- an accessory coupling method in which a component having a structure close to that of the component is added and reacted as an accessory.
- the applicant is C.I. I.
- the average particle diameter is already 30 to 150 nm
- the half-width of the maximum peak in the X-ray diffraction spectrum or a peak corresponding thereto is 0.3 to 0.7 °
- the major axis of the particle C. and the minor axis length ratio (average value) is 1 to 1.5.
- I. Pigment Yellow 74 is proposed. (See Patent Document 1).
- This C.I. I. Pigment Yellow 74 is excellent in light resistance and can record an image with good transparency and sharpness.
- the present invention has been made in view of such problems of the prior art, and its purpose is superior to conventional pigments in dispersibility when used as a colorant in a coloring composition, and has a particle size.
- a colored composition having good stability and storage stability can be provided, and by using the colored composition, an image having a high level of sharpness and transparency that could not be achieved by the prior art.
- C. is an insoluble azo pigment that can be formed (recorded) and has excellent weather resistance (light).
- C.I. I. Pigment Yellow 74 has a diffraction intensity ratio (26.6 ° / 11.7) between a peak at 11.7 ° and a peak at 26.6 ° among the peaks measured by powder X-ray diffraction using CuK ⁇ rays.
- the problems of the prior art described above can be solved when the average primary particle diameter is within a specific range. It came.
- the present invention relates to C.I. I. Pigment Yellow 74 is provided.
- Ratio of the diffraction intensity of the peak at 26.6 ° to the diffraction intensity at the peak of 11.7 ° at the Bragg angle (2 ⁇ ⁇ 0.2 °) measured by powder X-ray diffraction using CuK ⁇ rays ( 26.6 ° / 11.7 °) is 0.85 to 1.12, and the insoluble azo pigment is a crystalline particle having an average primary particle diameter of 20 to 130 nm.
- I. Pigment Yellow 74 is provided.
- the following coloring compositions are provided as another embodiment.
- C.I. is an insoluble azo pigment provided by the prior art when used as a colorant in a coloring composition.
- C.I. is an insoluble azo pigment that is clearly superior in dispersibility, better particle size stability of the pigment, and better storage stability of the coloring composition.
- Pigment Yellow 74 is provided.
- the excellent C.I. I. By using a coloring composition containing Pigment Yellow 74, image formation (recording) with a high level of sharpness and transparency that could not be achieved with the prior art can be made possible. The resulting image will be superior in weather resistance (light).
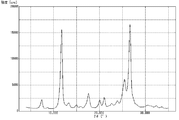
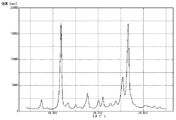
- FIG. 4 is a graph showing measurement results of powder X-ray diffraction of pigment A prepared in Example 1.
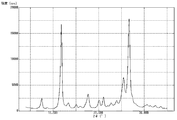
- FIG. 4 is a graph showing measurement results of powder X-ray diffraction of pigment B prepared in Example 2.
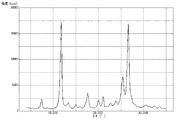
- FIG. 6 is a graph showing measurement results of powder X-ray diffraction of pigment C prepared in Comparative Example 1.
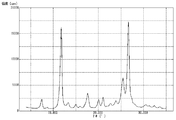
- 4 is a graph showing measurement results of powder X-ray diffraction of pigment D prepared in Example 3. It is a graph which shows the measurement result of the powder X-ray diffraction of the pigment E prepared in the comparative example 2.
- 6 is a graph showing measurement results of powder X-ray diffraction of pigment F prepared in Example 4.
- C.I. I. Pigment Yellow 74 (insoluble azo pigment)
- P.Y.74 P.I. Y. 74
- its crystallinity was measured, and detailed examination was made on the difference between these values and the characteristics when each of the obtained pigments was used as the colorant of the coloring composition.
- the P.I. Y. 74 pigment when it is used as a colorant for a coloring composition, Y.
- the P.I. Y Compared with a colored composition using 74 pigments, it exhibits better particle size stability and storage stability. Together with this, the P.I. Y.
- the conventional P.P. Y. Excellent image formation (recording) showing a high level of clarity and transparency that could not be achieved with 74 pigments becomes possible. Furthermore, the printed matter obtained has less color change when installed outdoors and is more excellent in weather resistance (light). According to the study by the present inventors, the above-mentioned excellent effect is obtained by pigmented P.I. Y. 74 is stably obtained when the following requirements are satisfied. Specifically, the P.I. Y.
- 74 pigment is the ratio of the diffraction intensity of the 26.6 ° peak to the diffraction intensity of the 11.7 ° peak at a Bragg angle (2 ⁇ ⁇ 0.2 °) measured by powder X-ray diffraction using CuK ⁇ rays.
- (26.6 ° / 11.7 °) is a crystal particle having crystallinity in the range of 0.85 to 1.12 and having an average primary particle diameter of 20 to 130 nm. Cost.
- the excellent effect of the present invention is particularly the effect of pigmented P.I. Y. 74, depending on the crystallinity of 74. Y.
- the 74 pigment is stably obtained when the specific crystal particles defined in the present invention are used.
- the 74 pigment has a crystallinity of a 26.6 ° peak relative to the diffraction intensity of the 11.7 ° peak at a Bragg angle (2 ⁇ ⁇ 0.2 °) measured by powder X-ray diffraction using CuK ⁇ rays. Crystal grains having a diffraction intensity ratio (26.6 ° / 11.7 °) of 0.85 to 1.12.
- a preferable range of the above value is 0.87 to 1.10, more preferably 0.89 to 1.00.
- P.I. Y. 74 pigment is a crystal particle in which the ratio of the diffraction intensity of the peak at 26.6 ° to the diffraction intensity of the peak at 11.7 ° (hereinafter also simply referred to as “diffraction intensity ratio”) is within the above numerical range.
- the weather resistance (light) of an image formed by using this as a colorant is remarkably improved.
- a coloring composition as a colorant when it is incorporated in a coloring composition as a colorant, its particle size stability and storage stability are improved, and together with this, an image with excellent sharpness and transparency is formed ( It becomes a coloring composition capable of recording).
- powder X-ray diffraction of 74 pigments should be measured using a powder X-ray diffractometer (trade name “RINT2000”, manufactured by Rigaku Corporation) according to Japanese Industrial Standards JIS K0131 (general rules for X-ray diffraction analysis). Can do.
- the 74 pigment further has a half-width of a peak of 11.7 ° and a half-width of a peak of 26.6 ° at a Bragg angle (2 ⁇ ⁇ 0.2 °) measured by powder X-ray diffraction using CuK ⁇ rays. Any of these is preferably crystal grains within a range of 0.10 to 0.60 °, and more preferably 0.10 to 0.58 °. In particular, it is preferable that these half-value widths are in the range of 0.10 to 0.56 °.
- the “half-width of the peak” means a peak width (angle) at a height (X-ray diffraction intensity) of 1 ⁇ 2 of each peak from the base line in the powder X-ray diffraction spectrum. According to the study by the present inventors, the peak width at 11.7 ° and the peak width at 26.6 ° are both in the above numerical range.
- the 74 pigment has very high crystallinity. That is, P.P. having such high crystallinity. Y. No. 74 shows better weather resistance (light) despite its average primary particle size being very small as described below.
- Y. 74 needs to be the pigment particles (crystal particles) having the specific crystallinity described above having an average primary particle diameter of 20 to 130 nm, and the particle diameter is preferably 50 to 120 nm. More preferably, it is ⁇ 110 nm. According to the study by the present inventors, P.I. Y. When the average primary particle size of the 74 pigment particles is within the above numerical range, when this is used as a colorant, a colored composition (ink) capable of recording an image with more transparency and sharpness is obtained. be able to. P.P. Y.
- the average primary particle size of 74 pigments is, for example, that the pigment particles are photographed using a transmission electron microscope (trade name “H7100”, manufactured by Hitachi, Ltd.), and the primary particles of about 10 arbitrarily selected primary particles. It can be measured and calculated as an average value of the particle size.
- the shape of the 74 pigment crystal particles is preferably not an acicular shape but an oval or true sphere.
- the particle state of the 74 crystal grains is preferably uniform. More specific P.I. Y.
- the average value of the ratio of the major axis to the minor axis is preferably 1 to 1.50, more preferably 1 to 1.30. According to the study by the present inventors, P.I. Y.
- the average value of the ratio of the major axis to the minor axis of 74 crystal grains is within the above numerical range, and when this is used for the coloring composition, the dispersibility in the dispersion medium is further improved. Can be improved.
- the average value of the ratio of the major axis to the minor axis (major axis / minor axis) of 74 crystal grains can be measured in the same manner as in the case of measuring the average primary particle diameter described above. Specifically, for example, pigment particles are photographed using a transmission electron microscope (trade name “H7100”, manufactured by Hitachi, Ltd.). Then, the minor axis and major axis of the primary particles of the pigment are measured, and the ratio of the major axis to the minor axis (major axis / minor axis) is calculated. The average value of the “major axis / minor axis” calculated for the primary particles of about 10 arbitrarily selected pigments is shown in FIG. Y. The ratio of the major axis to the minor axis of 74 (major axis / minor axis) can be taken as an average value.
- Pigment particles of the present invention which are pigment particles (crystal particles) having a specific crystallinity and an average particle size defined in the present invention, which are pigmented.
- P.P. Y. 74 (hereinafter referred to as “pre-treatment pigment” for the sake of convenience) particles may be pigmented by a method such as wet milling.
- the wet grinding of the pre-treatment pigment can be carried out, for example, according to a conventionally known method. *
- wet grinding examples include collision milling such as a ball mill, sand mill, attritor, and vibration mill; shearing force of a horizontal continuous medium disperser, kneader, three rolls, Banbury mixer, etc. The thing etc. which grind
- additives can be used as necessary.
- dispersants that are rosins such as hydrogenated rosin
- polymer dispersants such as surfactants, pigment derivatives, extender pigments, and the like are mixed alone or in combination to perform wet grinding. Is also a preferred method. By doing in this way, the dispersibility of a pigment can be considered and it can use for preparation effective also with respect to aggregation, adhesion suppression, etc.
- additives that can be used in this case include the following.
- polyoxyalkylene alkyl ethers such as polyoxyethylene lauryl ether and polyoxyethylene stearyl ether, polyoxyalkylene alkyl phenyl ether, polyoxyalkylene alkyl amino ether, sorbitan fatty acid ester, glycerin fatty acid ester and the like
- Nonionic surfactants anionic surfactants such as sulfonates such as sodium polyoxyethylene lauryl ether sulfate and sodium lauryl sulfate, and phosphate esters such as potassium alkyl phosphate, or lauryltrimethylammonium Cationic surfactants typified by quaternary ammonium salts such as chloride and stearyltrimethylammonium chloride can be mentioned and can be preferably used.
- pigment derivatives that can be used in preparing 74 pigments include alkyl groups, sulfonic acid groups, sulfonic acid amide groups, sulfonic acid metal bases, halogen groups, and the like for pigments such as anthraquinone, quinacridone, phthalocyanine, and perylene. Examples thereof include those substituted with a phthalimide group or the like.
- extender pigments that can be used include silica, talc, calcium carbonate, barium sulfate, and titanium oxide. These are not particularly limited, and can be appropriately used depending on the purpose.
- a water-soluble organic solvent that moderately promotes crystal growth of the pigment is preferable as a solvent for wet-grinding the untreated pigment.
- water-soluble organic solvents having such effects include diethylene glycol, ethylene glycol monobutyl ether, triethylene glycol monobutyl ether, propylene glycol, polyethylene glycol (# 200, # 300, etc.), glycerin, formamide, and the like.
- a solvent that enhances the crystal growth of the pigment (for example, N-methyl-2-pyrrolidone, etc.) promotes the crystal growth rather than the wet milling effect.
- wet milling of the pre-treatment pigment under heat retention or heating conditions.
- P.D. can satisfy the diffraction intensity ratio and the average primary particle size defined in the present invention.
- Y. Crystal particles of 74 pigments can be easily prepared.
- the wet milling is preferably carried out while controlling the temperature of the object to be treated (pre-treatment pigment) to be 50 to 100 ° C., and more preferably being controlled to be 60 to 95 ° C. preferable.
- the time required for wet grinding is not particularly limited, but is usually about 3 to 24 hours.
- the pre-treatment pigment necessary for obtaining 74 pigments a commercially available product or a synthesized pigment may be used.
- the pre-treatment pigment can be synthesized by a conventionally known azo synthesis method described in, for example, Japanese Patent Publication No. 60-36224 and Japanese Patent No. 3055673. In that case, in order to prevent aggregation of the particles of the obtained pre-treatment pigment, a known rosin, a polymeric dispersant, a surfactant, or a pigment derivative having a polar group introduced therein is used in combination. Is preferably synthesized.
- the accessory coupling method, the heat treatment method, or the method of adding a water-soluble organic solvent or a hydrophobic organic solvent during the heat treatment is used in combination to obtain crystals of the pre-treatment pigment obtained. It is preferable to grow.
- the coloring composition of the present invention is the P.I. Y. 74 crystal grains are contained as a colorant.
- the coloring composition of the present invention includes, for example, various printing inks, sizing colors, binder colors, coating agents, various paints, image recording compositions such as dry or wet toners for electronic copying machines, thermal transfer recording inks, and writing instrument inks. It can be used as an inkjet ink or the like. Among these, it is required at a high level that images having excellent particle size stability and storage stability, and weather resistance (light), sharpness, and transparency can be formed (recorded). It is particularly useful as an image recording composition (toner) and an inkjet ink.
- coloring composition of the present invention P.I. Y.
- components other than 74 what is normally contained in coloring compositions including an inkjet ink can be used.
- components other than 74 pigments include water, water-soluble resins, surfactants, and water-soluble solvents.
- water-soluble resin examples include acrylic resins, acrylic-styrene resins, polyester resins, polyamide resins, polyurethane resins, and the like.
- surfactant include an anionic surfactant, a nonionic surfactant, an amphoteric surfactant, and a cationic surfactant.
- water-soluble solvent examples include alcohols such as methanol, ethanol and isopropyl alcohol; polyhydric alcohols such as ethylene glycol, diethylene glycol, polyethylene glycol and glycerin; polyhydric alcohols such as ethylene glycol monomethyl ether and ethylene glycol monobutyl ether. Ethers; amine solvents such as diethanolamine and ethanolamine; heterocyclic compounds solvents such as N-methylpyrrolidone; sulfoxide solvents; sulfone solvents; acetonitrile and the like.
- the coloring composition of the present invention is an image recording composition (toner), P.I. Y.
- P.I. Y As components other than the 74 pigment, P.I. Y.
- a binder resin for fusing 74 pigment particles is contained. Specific examples of such a resin include polystyrene, styrene- (meth) acrylic acid ester copolymer, styrene-butadiene copolymer, polyester, and epoxy resin.
- the coloring composition of the present invention is a coloring agent such as P.I. Y.
- a coloring agent such as P.I. Y.
- any of dispersion media corresponding to various applications can be used as the dispersion medium.
- P.I. Y The content of the 74 pigment varies depending on the use, but it may be about 1 to 60% by mass with respect to the entire coloring composition.
- the colorant composition of the present invention includes a surface tension adjuster, a viscosity adjuster, a specific resistance adjuster, an antifoaming agent, and an antifungal agent for the purpose of improving physical properties such as storage stability and ejection stability of the colored composition. And the like may be optionally added.
- the coloring composition of the present invention includes, for example, P.I. Y. After mixing 74 pigments and various other components, it can be produced by highly dispersing according to a conventional method.
- powder X-ray diffraction was performed using a powder X-ray diffractometer (trade name “RINT2000”, manufactured by Rigaku Corporation), and the measurement results are shown in FIG.
- the ratio of the diffraction intensity at the peak of 26.6 ° to the diffraction intensity at the peak of 11.7 ° was 1.20.
- the half width of the peak at 11.7 ° was 0.45 °
- the half width of the peak at 26.6 ° was 0.53 °.
- the pigment (particles) before processing was photographed under photographing conditions of 60,000 times.
- the average primary particle diameter of the pre-treatment pigment (particles) obtained above was 160 nm.
- the shape of the pigment (particles) before processing was needle-like, and the average value of the ratio of the long axis to the short axis (long axis / short axis) was 2.03.
- the above-mentioned powder X-ray diffractometer was used also in the X-ray diffraction in another example.
- the peak diffraction intensity ratio (26.6 ° / 11.7 °) measured in the same manner as described above is simply referred to as “(26.6 ° / 11.7 °) diffraction intensity ratio”.
- Example 2 Preparation of pigment (Example 1) 400 parts of the pre-treatment pigment prepared in the synthesis example, 2400 parts of sodium chloride, and 700 parts of propylene glycol were charged into a kneader to make the contents a uniformly wet mass. The cooling device and the heat medium device were adjusted and kneaded and milled for 6 hours while controlling the temperature of the contents to be 60 to 90 ° C. to obtain a milled product. Then, 40000 parts of water was added to the ground product taken out from the kneader and left overnight. This was heated to 60 ° C. and then filtered and washed with water to remove sodium chloride and propylene glycol. Then, after drying at 100 ° C. for 24 hours, the mixture was pulverized to obtain 385 parts of pigment A.
- Powder X-ray diffraction was performed on the pigment A obtained above using a powder X-ray diffractometer, and the measurement results are shown in FIG.
- the (26.6 ° / 11.7 °) diffraction intensity ratio was 0.94.
- the half width of the peak at 11.7 ° was 0.39 °
- the half width of the peak at 26.6 ° was 0.52 °.
- pigment A (particles) was photographed under photographing conditions of 60,000 times.
- the average primary particle diameter of the pigment A (particles) measured by visual observation of the photographed image was 85 nm.
- the shape of the pigment A (particles) was relatively spherical, and the average value of the ratio of the major axis to the minor axis (major axis / minor axis) was 1.05.
- Example 2 400 parts of the pre-treatment pigment prepared in the synthesis example, 3600 parts of sodium chloride, and 900 parts of propylene glycol were charged into a kneader to make the contents a uniformly wet mass.
- the cooling device and the heat medium device were adjusted and kneaded and milled for 6 hours while controlling the temperature of the contents to be 60 to 90 ° C. to obtain a milled product.
- 40000 parts of water was added to the ground product taken out from the kneader and left overnight. This was heated to 60 ° C. and then filtered and washed with water to remove sodium chloride and propylene glycol. Then, after drying at 100 ° C. for 24 hours, pulverization gave 385 parts of pigment B.
- Powder X-ray diffraction was performed on the pigment B obtained above using a powder X-ray diffractometer, and the measurement results are shown in FIG.
- the (26.6 ° / 11.7 °) diffraction intensity ratio was 1.12.
- the half width of the peak at 11.7 ° was 0.40 °
- the half width of the peak at 26.6 ° was 0.52 °.
- pigment B (particles) was photographed under photographing conditions of 60,000 times.
- the average primary particle diameter of the pigment B (particles) measured by visual observation of the photographed image was 105 nm.
- the shape of the pigment B (particles) was relatively spherical, and the average value of the ratio of the major axis to the minor axis (major axis / minor axis) was 1.10.
- Powder X-ray diffraction was performed on the pigment C obtained above using a powder X-ray diffractometer, and the measurement results are shown in FIG.
- the (26.6 ° / 11.7 °) diffraction intensity ratio was 1.05.
- the half width of the peak at 11.7 ° was 0.40 °
- the half width of the peak at 26.6 ° was 0.52 °.
- pigment C (particles) was photographed under photographing conditions of 60,000 times.
- the average primary particle diameter of the pigment C (particles) measured by visual observation of the photographed image was 135 nm.
- the shape of the pigment C (particles) was relatively close to a needle shape, and the average value of the ratio of the major axis to the minor axis (major axis / minor axis) was 1.40.
- Example 3 300 parts of the pre-treatment pigment prepared in the synthesis example, 26 parts of triethylene glycol monobutyl ether, and 6000 parts of zirconia beads having a diameter of 3 mm were charged in a ball mill to obtain a uniformly wet mass. Wet milling was performed at 60 rpm for 20 hours. Then, after adding 2000 parts of water and driving
- Powder X-ray diffraction was performed on the pigment D obtained above using a powder X-ray diffractometer, and the measurement results are shown in FIG.
- the (26.6 ° / 11.7 °) diffraction intensity ratio was 0.98.
- the half width of the peak at 11.7 ° was 0.40 °
- the half width of the peak at 26.6 ° was 0.52 °.
- pigment D (particles) was photographed under photographing conditions of 60,000 times.
- the average primary particle diameter of the pigment D (particles) measured by visual observation of the photographed image was 110 nm.
- the shape of the pigment D (particle) was relatively close to a columnar shape, and the average value of the ratio of the major axis to the minor axis (major axis / minor axis) was 1.25.
- Comparative Example 2 300 parts of the pre-treatment pigment prepared in the synthesis example, 39 parts of N-methyl-2-pyrrolidone, and 6000 parts of zirconia beads having a diameter of 3 mm were charged in a ball mill to make the contents uniformly a wet mass. . Wet milling was performed at 60 rpm for 20 hours. Then, after adding 2000 parts of water and driving
- Powder X-ray diffraction was performed on the pigment E obtained above using a powder X-ray diffractometer, and the measurement results are shown in FIG.
- the (26.6 ° / 11.7 °) diffraction intensity ratio was 1.08.
- the half width of the peak at 11.7 ° was 0.41 °
- the half width of the peak at 26.6 ° was 0.52 °.
- Pigment E (particles) was photographed under photographing conditions of 60,000 times.
- the average primary particle diameter of the pigment E (particles) measured by visual observation of the photographed image was 150 nm.
- the shape of the pigment E (particles) was relatively close to a needle shape, and the average value of the ratio of the long axis to the short axis (long axis / short axis) was 2.01.
- Example 4 100 parts of the pre-treatment pigment prepared in the synthesis example and 75 parts of triethylene glycol monobutyl ether were placed in a beaker and kneaded with a glass rod until the pre-treatment pigment was evenly wet to obtain a kneaded product.
- the obtained kneaded material was dispersed in 4 passes using a three-roll mill, and the dispersion was left in 4000 parts of water overnight to form a slurry.
- the slurry was stirred, filtered, washed with water, dried at 90 ° C. for 24 hours, and then pulverized to obtain 90 parts of the target pigment F.
- the powder F obtained above was subjected to powder X-ray diffraction, and the measurement results are shown in FIG.
- the (26.6 ° / 11.7 °) diffraction intensity ratio was 0.99.
- the half width of the peak at 11.7 ° was 0.40 °
- the half width of the peak at 26.6 ° was 0.52 °.
- the pigment F (particles) was photographed under photographing conditions of 60,000 times.
- the average primary particle diameter of the pigment F (particles) measured by visual observation of the photographed image was 120 nm.
- the shape of the pigment F (particles) was relatively close to a needle shape, and the average value of the ratio of the major axis to the minor axis (major axis / minor axis) was 1.29.
- Example 5 Preparation of inkjet ink (Example 5)
- 10.0 parts of pigment A prepared in Example 1 5.0 parts of a water-soluble styrene acrylic resin (solid content 45%), and 45 parts of ion-exchanged water. 0 parts were added and premixed.
- a dispersion treatment was performed using a paint conditioner to obtain 60.0 parts of a dispersion liquid in which pigment particles having a volume average particle diameter of around 110 nm were dispersed. The time required for the dispersion treatment at this time was 240 minutes.
- the volume average particle diameter in the above was measured during the dispersion treatment using a laser scattering particle size distribution analyzer (trade name “N5 Submicron Particle Size Analyzer”, manufactured by Beckman Coulter, Inc.).
- each of the inkjet inks was prepared in the same manner as in Example 5 except that the pigments prepared in Examples 2 and 3, 4 and Comparative Examples 1 and 2 and Synthesis Example were used. Obtained.
- Reflection density is 85% or more 4: Reflection density is 70% or more and less than 85% 3: Reflection density is 55% or more and less than 70% 2: Reflection density is 40% or more and less than 55% 1: Reflection density is Less than 40%
- the coloring composition of the present invention includes, for example, various printing inks, sizing colors, binder colors, coating agents, various paints, electrophotographic dry or wet toners, thermal transfer recording inks, writing instrument inks, and color filter pixel formations. It is useful as a coating solution, inkjet ink, etc., and its use is expected.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Inks, Pencil-Leads, Or Crayons (AREA)
- Ink Jet Recording Methods And Recording Media Thereof (AREA)
- Ink Jet (AREA)
Abstract
Description
[1]CuKα線を用いた粉末X線回折により測定されるブラッグ角(2θ±0.2°)における、11.7°のピークの回折強度に対する26.6°のピークの回折強度の比(26.6°/11.7°)が0.85~1.12であり、かつ、平均一次粒子径が20~130nmの結晶粒子であることを特徴とする不溶性アゾ顔料であるC.I.ピグメントイエロー74。
[2]前記11.7°のピークの半値幅と前記26.6°のピークの半値幅のいずれもが、0.10~0.60°の範囲内にある前記[1]に記載のC.I.ピグメントイエロー74。
[3]前記結晶粒子の短軸に対する長軸の比(長軸/短軸)の平均値が1~1.50である前記[1]又は[2]に記載のC.I.ピグメントイエロー74。
[4]前記[1]~[3]のいずれかに記載のC.I.ピグメントイエロー74を着色剤として含有してなる着色組成物。
[5]画像記録用組成物又はインクジェット記録用組成物である前記[4]に記載の着色組成物。
本発明者らは、上記従来技術の課題を解決するため、C.I.ピグメントイエロー74(以下、「P.Y.74」とも記す)の顔料化の条件を種々に変更し、それぞれの条件で顔料化したP.Y.74(P.Y.74顔料とも呼ぶ)について、その結晶性を測定し、これらの値と、得られた各顔料を着色組成物の着色剤に利用した場合における特性の違いに関し詳細な検討を行い、本発明を達成した。すなわち、本発明のP.Y.74顔料は、これを着色組成物の着色剤に用いた場合、従来のP.Y.74顔料を用いた着色組成物と比べ、より優れた粒径安定性及び保存安定性を示す。これとともに、本発明のP.Y.74顔料を用いることで、従来のP.Y.74顔料では達成できなかった高いレベルの鮮明性及び透明性を示す優れた画像形成(記録)が可能になる。さらに、得られる印刷物は、屋外に設置した場合における色変化が少ない、より耐候(光)性に優れたものになる。本発明者らの検討によれば、上記の優れた効果は、顔料化したP.Y.74が、下記の要件を満たすものである場合に安定して得られる。具体的には、本発明のP.Y.74顔料は、CuKα線を用いた粉末X線回折により測定されるブラッグ角(2θ±0.2°)における、11.7°のピークの回折強度に対する26.6°のピークの回折強度の比(26.6°/11.7°)が、0.85~1.12の範囲内にある結晶性を有し、さらに、その平均一次粒子径が20~130nmである結晶粒子であることを要する。以下、好ましい実施の形態を挙げて本発明をさらに詳細に説明する。
本発明の着色組成物は、前述の本発明のP.Y.74の結晶粒子を着色剤として含有してなる。本発明の着色組成物は、例えば、各種印刷インキ、サイジングカラー、バインダーカラー、コーティング剤、各種塗料、電子複写機用乾式又は湿式トナー等の画像記録用組成物、熱転写記録用インキ、筆記具用インキ、インクジェットインク等として利用することができる。これらのなかでも、優れた粒径安定性及び保存安定性、並びに耐候(光)性、鮮明性、及び透明性に優れた画像を形成(記録)可能であること等が高いレベルで要求される、画像記録用組成物(トナー)及びインクジェットインクとして特に有用である。
(合成例)
2-メトキシ-4-ニトロアニリンを910部と、2-メトキシアセトアセトアニライドを1220部使用して、従来公知の方法によりアクセサリーカップリング反応させた。さらに、加熱熟成処理を行った後、水洗及び乾燥して、処理前顔料(2-[(2-メトキシ-4-ニトロフェニル)アゾ]-N-(2-メトキシフェニル)-3-オキソブタンアミド)2100部を得た。
(実施例1)
合成例で作製した処理前顔料を400部、塩化ナトリウムを2400部、及びプロピレングリコールを700部、ニーダーに仕込んで、内容物を均一に湿潤された塊とした。冷却装置及び熱媒装置を調整し、内容物の温度が60~90℃になるように管理しながら6時間混練摩砕処理して摩砕物を得た。そして、ニーダーより取り出した摩砕物に水40000部を加えて一晩放置した。これを60℃に加熱した後、ろ過及び水洗して塩化ナトリウムとプロピレングリコールを除去した。その後、100℃で24時間乾燥した後に粉砕して、顔料Aを385部得た。
合成例で作製した処理前顔料を400部、塩化ナトリウムを3600部、及びプロピレングリコールを900部、ニーダーに仕込んで、内容物を均一に湿潤された塊とした。冷却装置及び熱媒装置を調整し、内容物の温度が60~90℃になるように管理しながら6時間混練摩砕処理して摩砕物を得た。そして、ニーダーより取り出した摩砕物に水40000部を加えて一晩放置した。これを60℃に加熱した後、ろ過及び水洗して塩化ナトリウムとプロピレングリコールを除去した。その後、100℃で24時間乾燥した後に粉砕して顔料Bを385部得た。
合成例で作製した処理前顔料を400部、塩化ナトリウムを1200部、及びプロピレングリコールを1000部、ニーダーに仕込んで、内容物を均一に湿潤された塊とした。冷却装置及び熱媒装置を調整し、内容物の温度が40~50℃になるように管理しながら6時間混練摩砕処理して摩砕物を得た。そして、ニーダーより取り出した摩砕物に水40000部を加えて一晩放置した。これを60℃に加熱した後、ろ過及び水洗して塩化ナトリウムとプロピレングリコールを除去した。100℃で24時間乾燥した後に粉砕して顔料Cを385部得た。
合成例で作製した処理前顔料を300部、トリエチレングリコールモノブチルエーテルを26部、及び直径3mmのジルコニアビーズを6000部、ボールミル中に仕込んで、内容物を均一に湿潤された塊とした。60rpmで20時間運転し湿式粉砕を行った。引き続き、水2000部を加えて10時間運転した後、内容物(スラリー)を取り出した。取り出したスラリーをろ過及び水洗し、90℃で24時間乾燥させた後に粉砕処理して、目的の顔料Dを290部得た。
合成例で作製した処理前顔料を300部、N-メチル-2-ピロリドンを39部、及び直径3mmのジルコニアビーズを6000部、ボールミル中に仕込んで、内容物を均一に湿潤された塊とした。60rpmで20時間運転し湿式粉砕を行った。引き続き、水2000部を加えて10時間運転した後、内容物(スラリー)を取り出した。取り出したスラリーを、ろ過及び水洗し、90℃で24時間乾燥させた後に粉砕処理して、目的の顔料E290部を得た。
合成例で作製した処理前顔料を100部、及びトリエチレングリコールモノブチルエーテルを75部、ビーカーに入れ、処理前顔料がまんべんなく濡れるまでガラス棒を用いてよく混練して混練物を得た。得られた混練物を三本ロールミルにて4パス分散させ、分散物を4000部の水中に一晩放置してスラリーとした。このスラリーを撹拌し、ろ過及び水洗した後、90℃で24時間乾燥させた後に粉砕処理して、目的の顔料Fを90部得た。
(実施例5)
ジルコニアビーズを120部入れた250mLガラスビンに、実施例1で調製した顔料Aを10.0部、水溶性スチレンアクリル系樹脂(固形分45%)を5.0部、及びイオン交換水を45.0部加え、プレミキシングした。さらにペイントコンディショナーを使用して分散処理し、体積平均粒子径が110nm前後の顔料粒子が分散してなる分散液60.0部を得た。この際の分散処理に要した時間は240分であった。なお、上記における体積平均粒子径は、レーザー散乱方式粒度分布計(商品名「N5 Submicron Particle Size Analyzer」、ベックマンコールター社製)を使用して分散処理中に測定した。
顔料Aに代えて、実施例2及び3、4、比較例1及び2、並びに合成例で調製した顔料をそれぞれに用いたこと以外は、前述の実施例5と同様にして、それぞれインクジェットインクを得た。
(耐候(光)性)
市販のインクジェット用グロス紙にバーコーター規格3番を用いて、上記で調製したそれぞれのインクジェットインクを展色した。展色後のグロス紙を室温で一日放置した後、促進耐候性試験装置を使用して、照射強度100mW/cm2(照射波長:295~450nmの紫外光)、室温で50時間照射した。次いで、分光濃度計(商品名「500シリーズ」、エックスライト社製)を使用して照射前後の反射濃度を測定し、以下に示した5段階の基準に従って、それぞれ耐候(光)性を評価した。結果を表2にまとめて示した。
[評価基準]
5:反射濃度が、85%以上
4:反射濃度が、70%以上85%未満
3:反射濃度が、55%以上70%未満
2:反射濃度が、40%以上55%未満
1:反射濃度が、40%未満
先に調製した各インクジェットインクを、それぞれ25℃で1時間静置した。その後、先に使用したと同様のレーザー散乱方式粒度分布計を使用して、それぞれのインクジェットインク中の顔料の体積平均粒子径(初期分散粒径φ1)を測定した。次いで、各インクジェットインクを50℃で10日間保持した後、上記と同様にしてインクジェットインク中の顔料の体積平均粒子径(経時分散粒径φ2)をそれぞれ測定した。そして、初期分散粒径φ1に対する経時分散粒径φ2の比(φ2/φ1)を算出し、以下に示した5段階の基準に従って、それぞれのインクについて粒径安定性を評価した。結果を表2にまとめて示した。
[評価基準]
5:「φ2/φ1」が、1.3以下
4:「φ2/φ1」が、1.3超1.5以下
3:「φ2/φ1」が、1.5超1.7以下
2:「φ2/φ1」が、1.7超1.9以下
1:「φ2/φ1」が、1.9超
先に調製した各インクジェットインクを、それぞれ25℃で1時間静置した。その後、E型粘度計(商品名「RE80型」、東京産業社製、50rpm値)を使用して、それぞれのインクジェットインクの粘度(初期粘度粘度η1)を測定した。次いで、各インクジェットインクを50℃で10日間保持した後、上記と同様にしてインクジェットインクの粘度(経時粘度η2)をそれぞれ測定した。そして、初期粘度(η1)に対する経時粘度(η2)の比(η2/η1)を算出し、以下に示した5段階の基準に従って、それぞれのインクについて保存安定性を評価した。結果を表2にまとめて示した。
[評価基準]
5:「η2/η1」が、1.1以下
4:「η2/η1」が、1.1超1.3以下
3:「η2/η1」が、1.3超1.5以下
2:「η2/η1」が、1.5超1.7以下
1:「η2/η1」が、1.7超
先に調製した各インクジェットインクをそれぞれ用いて、ピエゾ振動子を有するオンデマンド型インクジェットプリンターで画像を印刷した。得られた画像を目視観察し、以下に示す基準に従い、それぞれの画像について鮮明性を評価した。結果を表2にまとめて示した。
[評価基準]
◎:良好
〇:実用レベル
△:不明瞭
市販のインクジェット用OHPフィルムに、バーコーター規格3番を用いて、先に調製した各インクジェットインクを展色した。展色後のフィルムを目視観察し、以下に示す基準に従い、それぞれの画像について透明性を評価した。結果を表2にまとめて示した。
[評価基準]
○:良好
△:半透明
×:透明性なし
Claims (5)
- CuKα線を用いた粉末X線回折により測定されるブラッグ角(2θ±0.2°)における、11.7°のピークの回折強度に対する26.6°のピークの回折強度の比(26.6°/11.7°)が0.85~1.12であり、かつ、平均一次粒子径が20~130nmの結晶粒子であることを特徴とする不溶性アゾ顔料であるC.I.ピグメントイエロー74。
- 前記11.7°のピークの半値幅と前記26.6°のピークの半値幅のいずれもが、0.10~0.60°の範囲内にある請求項1に記載のC.I.ピグメントイエロー74。
- 前記結晶粒子の短軸に対する長軸の比(長軸/短軸)の平均値が1.00~1.50である請求項1又は2に記載のC.I.ピグメントイエロー74。
- 請求項1~3のいずれか一項に記載のC.I.ピグメントイエロー74を着色剤として含有してなる着色組成物。
- 画像記録用組成物又はインクジェット記録用組成物である請求項4に記載の着色組成物。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013551648A JP5815744B2 (ja) | 2011-12-28 | 2012-12-19 | 不溶性アゾ顔料であるc.i.ピグメントイエロー74及びそれを用いた着色組成物 |
| KR1020147021180A KR20140116172A (ko) | 2011-12-28 | 2012-12-19 | 불용성 아조 안료인 c.i. 피그먼트 옐로우 74 및 그것을 이용한 착색 조성물 |
| US14/363,180 US9657176B2 (en) | 2011-12-28 | 2012-12-19 | C.I. pigment yellow 74 (insoluble AZO pigment), and coloring composition using same |
| CN201280065137.4A CN104024340A (zh) | 2011-12-28 | 2012-12-19 | 为不溶性偶氮颜料的c.i.颜料黄74和使用其的着色组合物 |
| EP12863374.0A EP2799499B1 (en) | 2011-12-28 | 2012-12-19 | C.i. pigment yellow 74 (insoluble azo pigment), and coloring composition using same |
| IN1191KON2014 IN2014KN01191A (ja) | 2011-12-28 | 2012-12-19 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011288579 | 2011-12-28 | ||
| JP2011-288579 | 2011-12-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013099730A1 true WO2013099730A1 (ja) | 2013-07-04 |
Family
ID=48697221
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/082899 WO2013099730A1 (ja) | 2011-12-28 | 2012-12-19 | 不溶性アゾ顔料であるc.i.ピグメントイエロー74及びそれを用いた着色組成物 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US9657176B2 (ja) |
| EP (1) | EP2799499B1 (ja) |
| JP (1) | JP5815744B2 (ja) |
| KR (1) | KR20140116172A (ja) |
| CN (1) | CN104024340A (ja) |
| IN (1) | IN2014KN01191A (ja) |
| TW (1) | TWI510562B (ja) |
| WO (1) | WO2013099730A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160008820A1 (en) * | 2014-07-10 | 2016-01-14 | Xerox Corporation | Magnetic actuated-milled pigment dispersions and process for making thereof |
| JP2018154739A (ja) * | 2017-03-17 | 2018-10-04 | セイコーエプソン株式会社 | 水系インクジェットインク組成物 |
| WO2020075568A1 (ja) * | 2018-10-10 | 2020-04-16 | 富士フイルム株式会社 | 着色組成物、膜、カラーフィルタの製造方法、カラーフィルタ、固体撮像素子及び画像表示装置 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6036224B2 (ja) | 1982-02-26 | 1985-08-19 | 大日精化工業株式会社 | アゾ顔料の製造方法 |
| JP3055673B2 (ja) | 1997-12-12 | 2000-06-26 | 大日精化工業株式会社 | 画像記録用着色組成物 |
| JP2002138233A (ja) * | 2000-11-06 | 2002-05-14 | Sharp Corp | インク組成物およびそれを用いた記録装置 |
| JP2003012960A (ja) * | 2001-07-03 | 2003-01-15 | Sharp Corp | 擬1次元結晶性イエロー有機顔料からなる着色剤およびそれを含むインク組成物 |
| JP2003012968A (ja) * | 2001-06-27 | 2003-01-15 | Sharp Corp | インク組成物およびそれを用いた記録装置 |
| JP2004026930A (ja) * | 2002-06-24 | 2004-01-29 | Dainichiseika Color & Chem Mfg Co Ltd | 不溶性アゾ顔料及び着色組成物 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2674325B2 (ja) | 1990-12-21 | 1997-11-12 | 三菱化学株式会社 | カラー電子写真用イエロートナー及びその製造方法 |
-
2012
- 2012-12-19 JP JP2013551648A patent/JP5815744B2/ja active Active
- 2012-12-19 WO PCT/JP2012/082899 patent/WO2013099730A1/ja active Application Filing
- 2012-12-19 IN IN1191KON2014 patent/IN2014KN01191A/en unknown
- 2012-12-19 EP EP12863374.0A patent/EP2799499B1/en active Active
- 2012-12-19 KR KR1020147021180A patent/KR20140116172A/ko not_active Application Discontinuation
- 2012-12-19 US US14/363,180 patent/US9657176B2/en active Active
- 2012-12-19 CN CN201280065137.4A patent/CN104024340A/zh active Pending
- 2012-12-28 TW TW101150893A patent/TWI510562B/zh active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6036224B2 (ja) | 1982-02-26 | 1985-08-19 | 大日精化工業株式会社 | アゾ顔料の製造方法 |
| JP3055673B2 (ja) | 1997-12-12 | 2000-06-26 | 大日精化工業株式会社 | 画像記録用着色組成物 |
| JP2002138233A (ja) * | 2000-11-06 | 2002-05-14 | Sharp Corp | インク組成物およびそれを用いた記録装置 |
| JP2003012968A (ja) * | 2001-06-27 | 2003-01-15 | Sharp Corp | インク組成物およびそれを用いた記録装置 |
| JP2003012960A (ja) * | 2001-07-03 | 2003-01-15 | Sharp Corp | 擬1次元結晶性イエロー有機顔料からなる着色剤およびそれを含むインク組成物 |
| JP2004026930A (ja) * | 2002-06-24 | 2004-01-29 | Dainichiseika Color & Chem Mfg Co Ltd | 不溶性アゾ顔料及び着色組成物 |
| JP4190218B2 (ja) | 2002-06-24 | 2008-12-03 | 大日精化工業株式会社 | 不溶性アゾ顔料及び着色組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2799499A4 |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160008820A1 (en) * | 2014-07-10 | 2016-01-14 | Xerox Corporation | Magnetic actuated-milled pigment dispersions and process for making thereof |
| US10066115B2 (en) | 2014-07-10 | 2018-09-04 | Xerox Corporation | Magnetic actuated-milled pigment dispersions and process for making thereof |
| JP2018154739A (ja) * | 2017-03-17 | 2018-10-04 | セイコーエプソン株式会社 | 水系インクジェットインク組成物 |
| JP7087269B2 (ja) | 2017-03-17 | 2022-06-21 | セイコーエプソン株式会社 | 水系インクジェットインク組成物 |
| WO2020075568A1 (ja) * | 2018-10-10 | 2020-04-16 | 富士フイルム株式会社 | 着色組成物、膜、カラーフィルタの製造方法、カラーフィルタ、固体撮像素子及び画像表示装置 |
| JPWO2020075568A1 (ja) * | 2018-10-10 | 2021-09-16 | 富士フイルム株式会社 | 着色組成物、膜、カラーフィルタの製造方法、カラーフィルタ、固体撮像素子及び画像表示装置 |
| JP7284184B2 (ja) | 2018-10-10 | 2023-05-30 | 富士フイルム株式会社 | 着色組成物、膜、カラーフィルタの製造方法、カラーフィルタ、固体撮像素子及び画像表示装置 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20140335358A1 (en) | 2014-11-13 |
| IN2014KN01191A (ja) | 2015-10-16 |
| TWI510562B (zh) | 2015-12-01 |
| EP2799499B1 (en) | 2018-02-21 |
| JPWO2013099730A1 (ja) | 2015-05-07 |
| KR20140116172A (ko) | 2014-10-01 |
| US9657176B2 (en) | 2017-05-23 |
| EP2799499A1 (en) | 2014-11-05 |
| EP2799499A4 (en) | 2015-08-12 |
| CN104024340A (zh) | 2014-09-03 |
| JP5815744B2 (ja) | 2015-11-17 |
| TW201335288A (zh) | 2013-09-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| ES2280957T3 (es) | Formulaciones de agentes colorantes que se basan en agua, para la impresion por chorros de tinta. | |
| WO2009110557A1 (ja) | アゾ顔料組成物、アゾ顔料組成物の製造方法、アゾ顔料組成物を含む分散物、着色組成物及びインクジェット記録用インク | |
| WO2009110558A1 (ja) | アゾ顔料組成物、アゾ顔料組成物の製造方法、アゾ顔料組成物を含む分散物、着色組成物及びインクジェット記録用インク | |
| JP6697571B2 (ja) | キナクリドン固溶体顔料の製造方法、顔料分散液及びインクジェット用インキ | |
| JP5010229B2 (ja) | 顔料分散剤、顔料組成物、顔料着色剤およびカラーフィルター用塗布液 | |
| US6942724B2 (en) | Modified organic colorants and dispersions, and methods for their preparation | |
| JP2007009096A (ja) | 顔料組成物およびそれを用いた顔料分散体 | |
| JP2010202858A (ja) | インク、インクジェット記録方法、インクカートリッジ、及びインクジェット記録装置 | |
| BRPI0613766A2 (pt) | método de preparação de uma composição de pigmento e composição de tinta para jato de tinta | |
| JP2007538117A (ja) | 櫛型分岐コポリマーを含む水性インクジェットインク組成物 | |
| JP5815744B2 (ja) | 不溶性アゾ顔料であるc.i.ピグメントイエロー74及びそれを用いた着色組成物 | |
| JP7066828B2 (ja) | キナクリドン固溶体顔料の製造方法、顔料分散液及びインクジェット用インキ | |
| JP4190218B2 (ja) | 不溶性アゾ顔料及び着色組成物 | |
| JP2014177575A (ja) | モノアゾ顔料組成物の製造方法、トナーおよびインクジェットインキ | |
| TWI772405B (zh) | 喹吖酮固溶體顏料之製造方法、顏料分散液及噴墨用墨水 | |
| JP2005120265A (ja) | 水性顔料分散体及び水性顔料記録液 | |
| JP4391073B2 (ja) | 水性マジェンタインク | |
| JP2002105351A (ja) | キノフタロン(C.I.PigmentYellow138)顔料及び該顔料を含む着色組成物 | |
| JP6644994B2 (ja) | 顔料分散体の製造方法 | |
| JP2022113966A (ja) | 顔料組成物およびその利用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12863374 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2013551648 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14363180 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012863374 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20147021180 Country of ref document: KR Kind code of ref document: A |