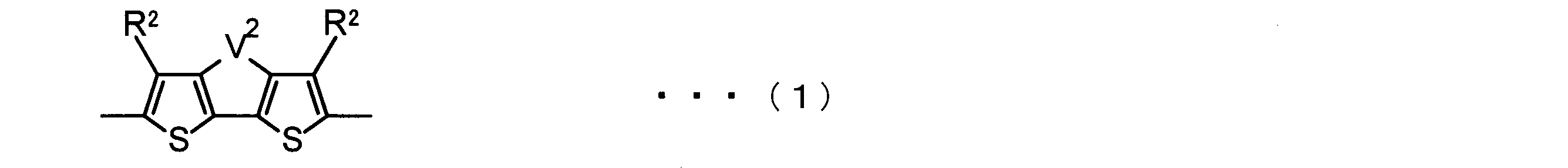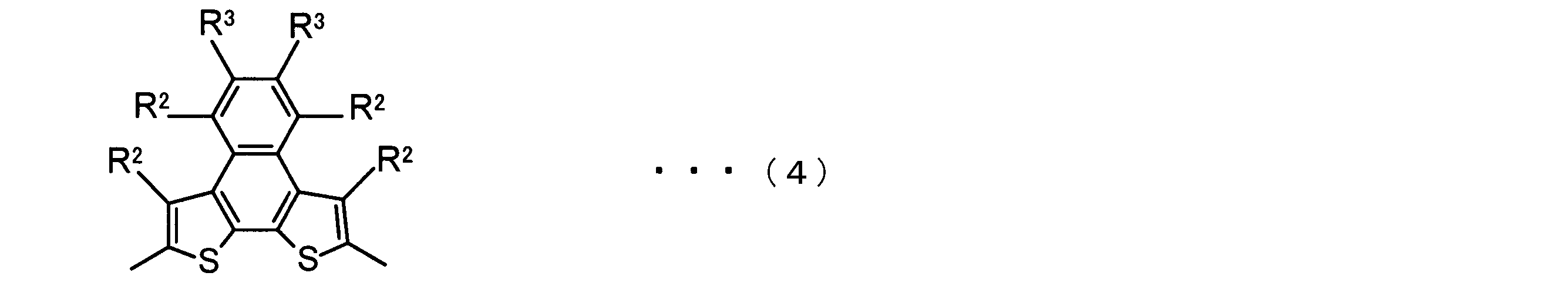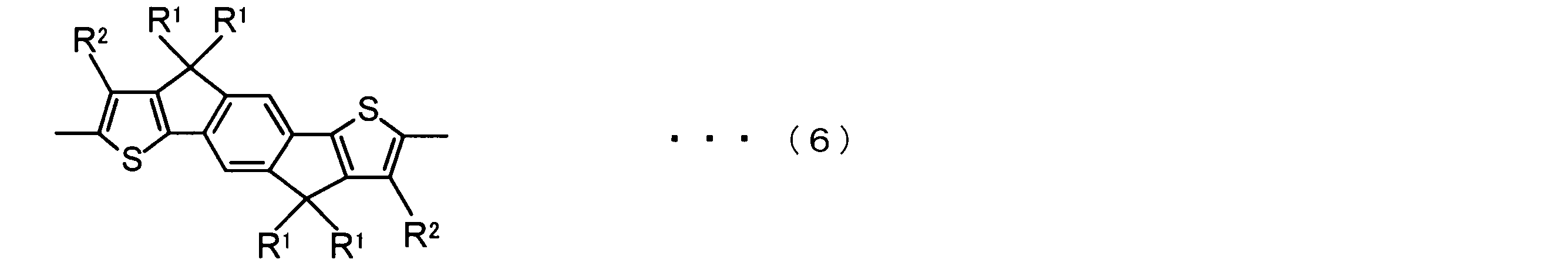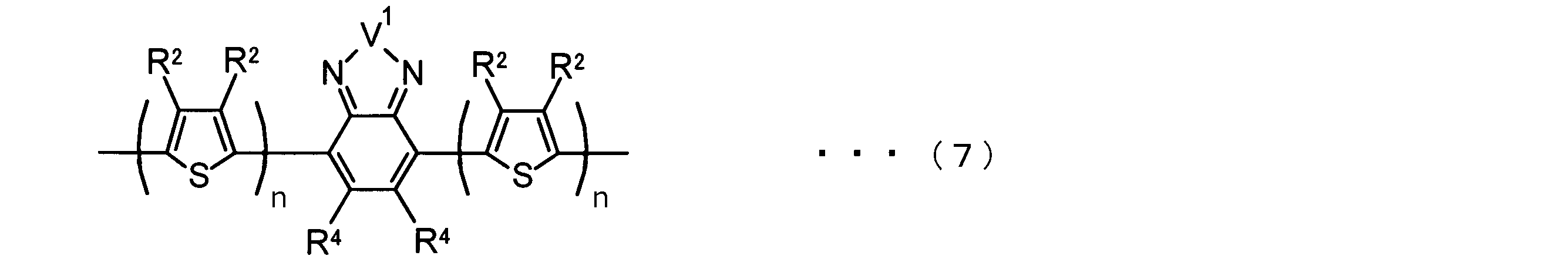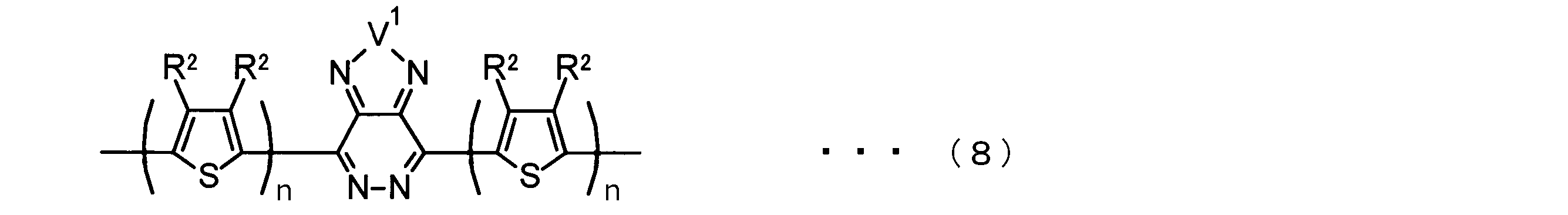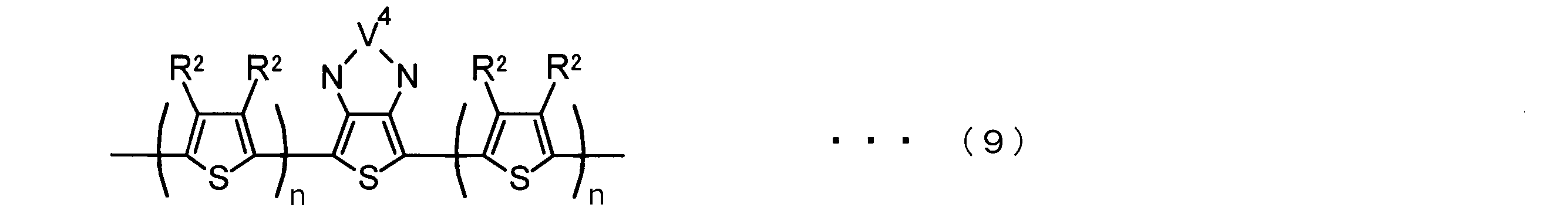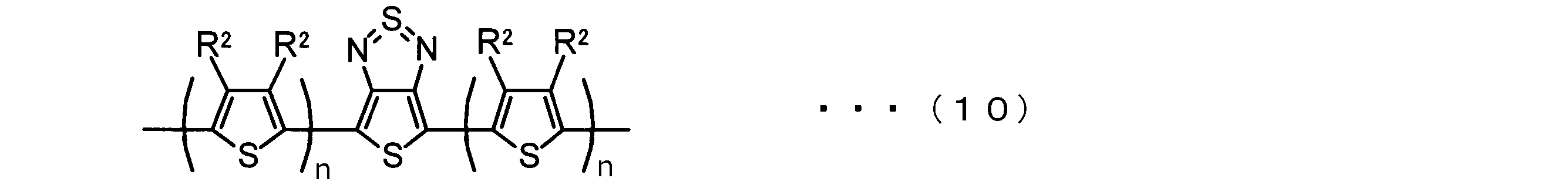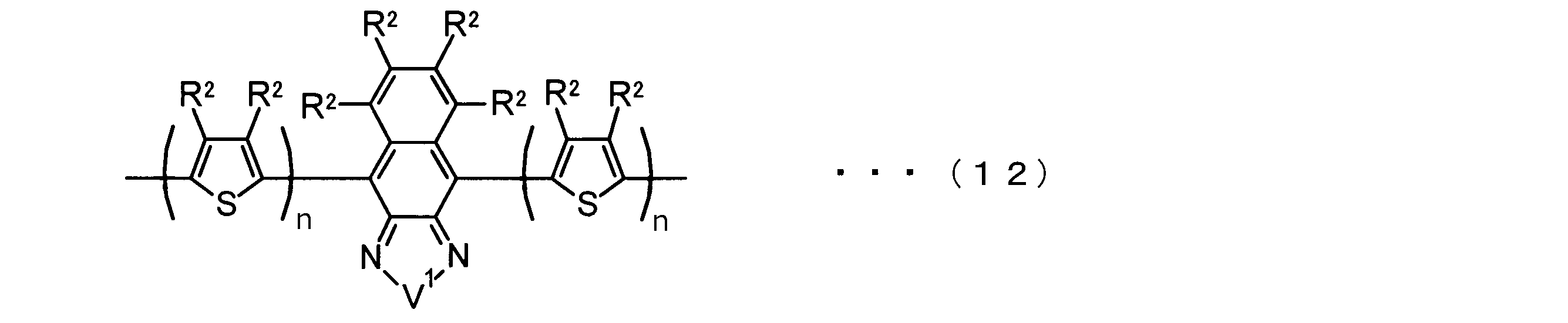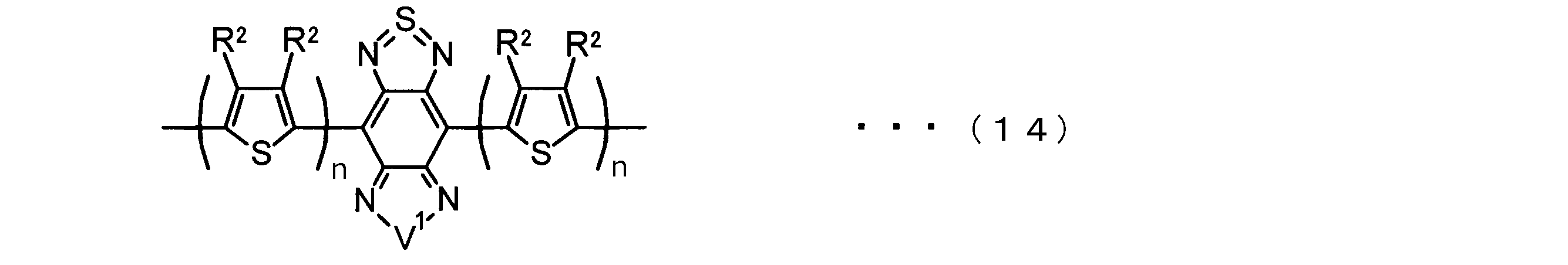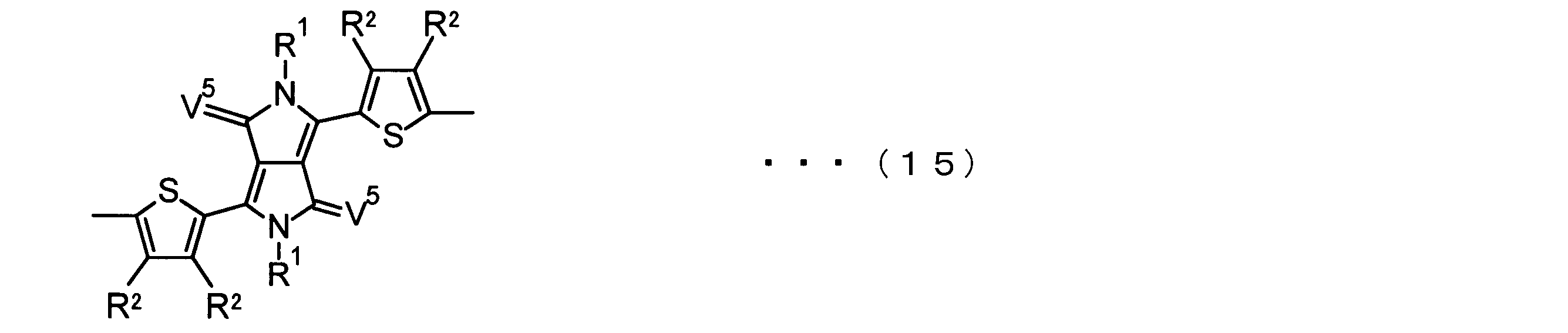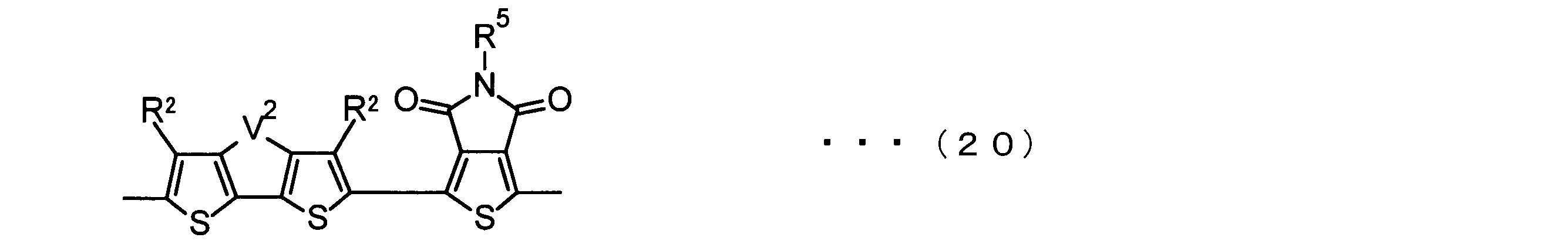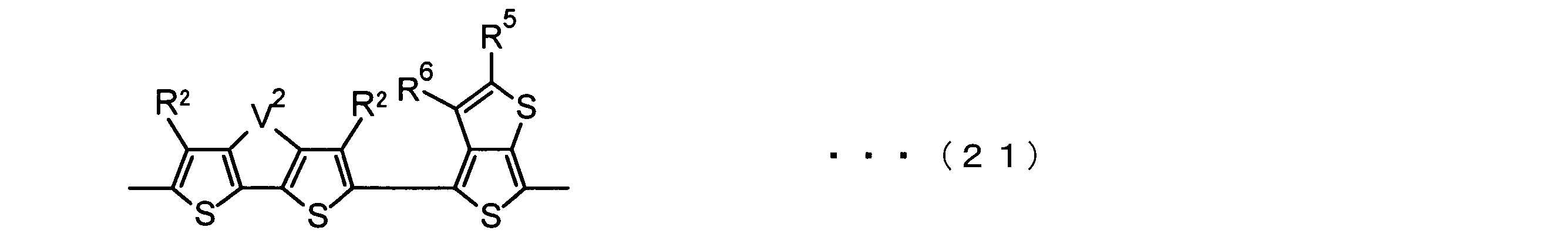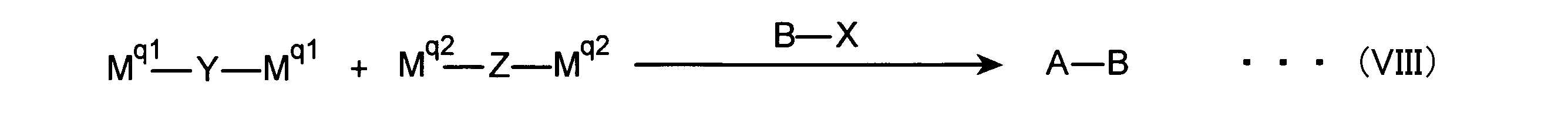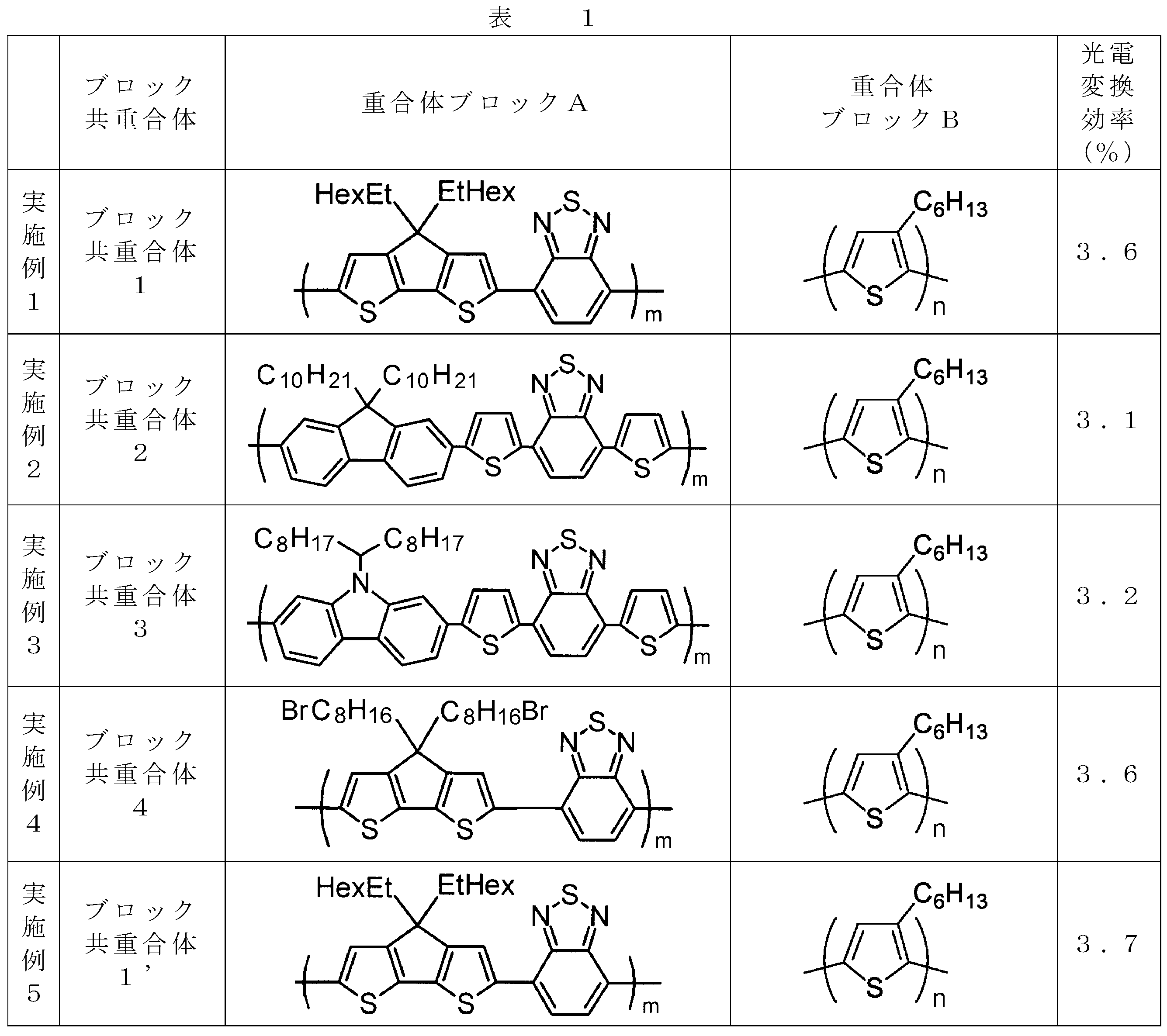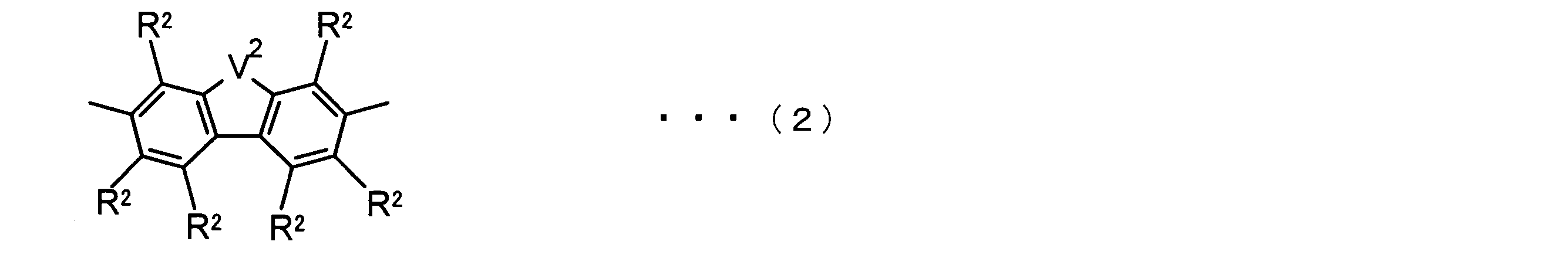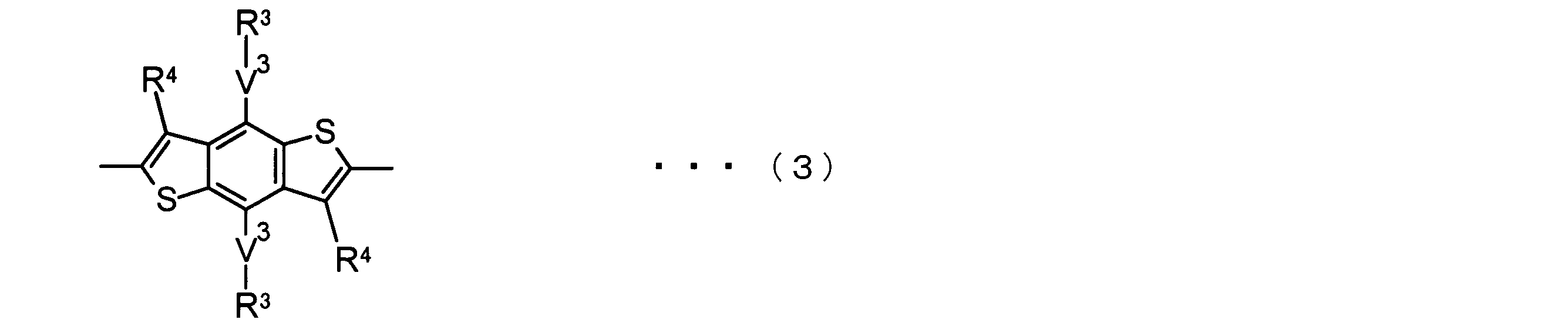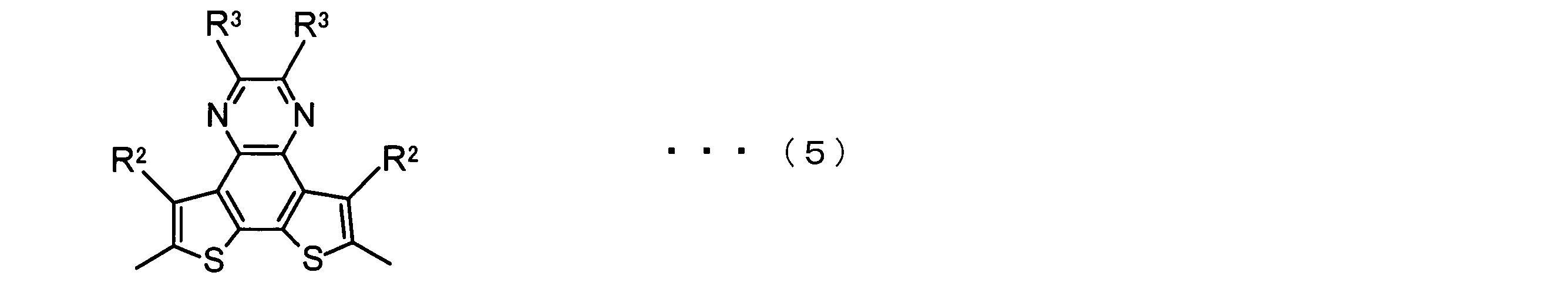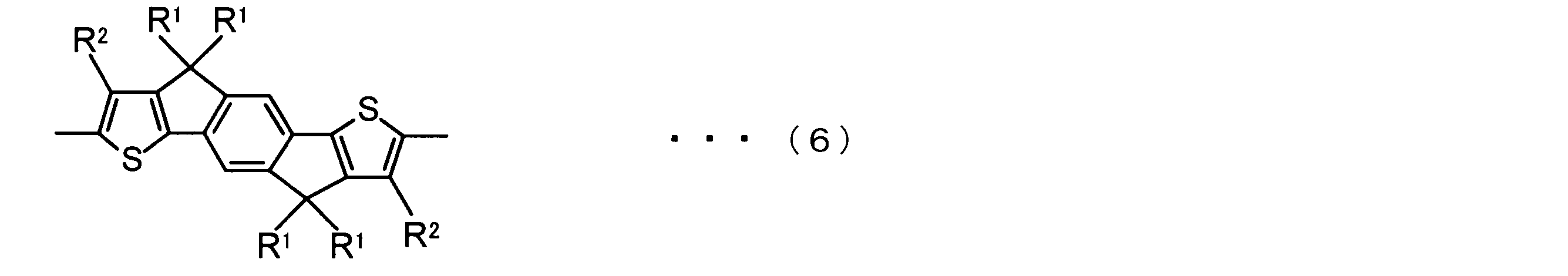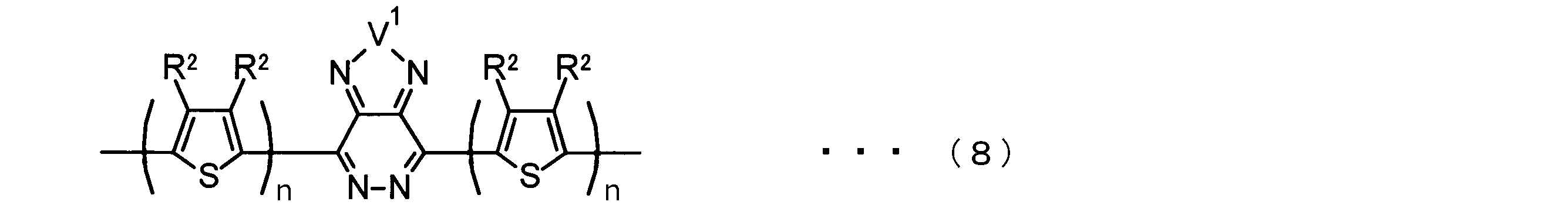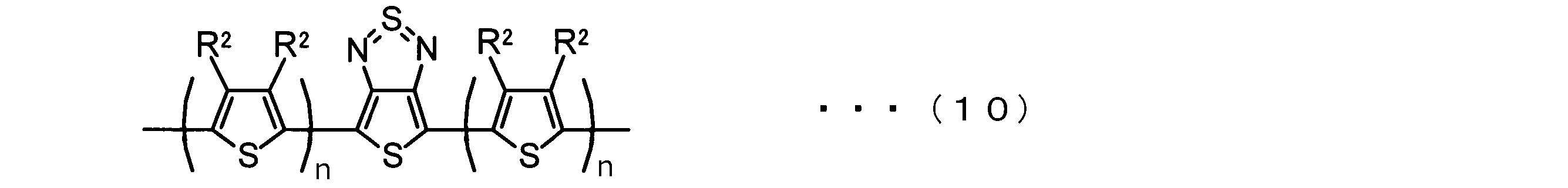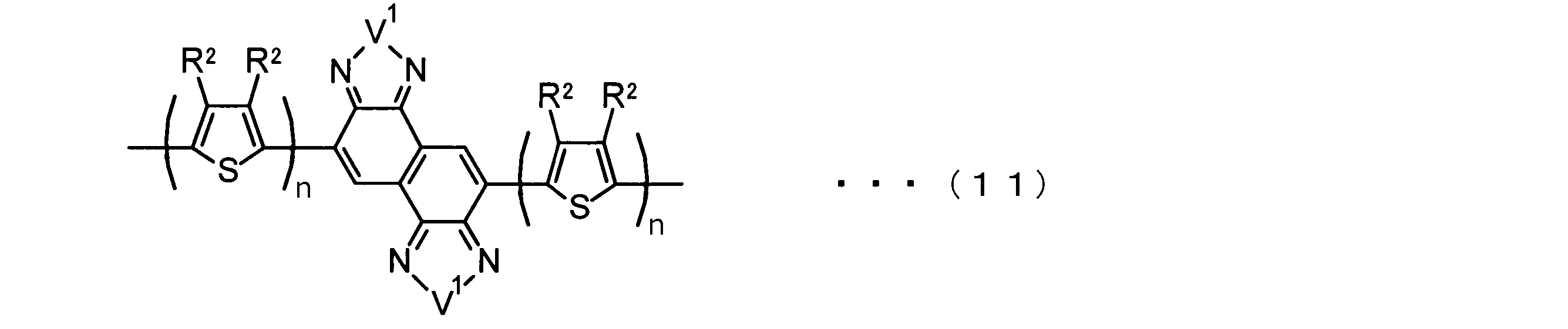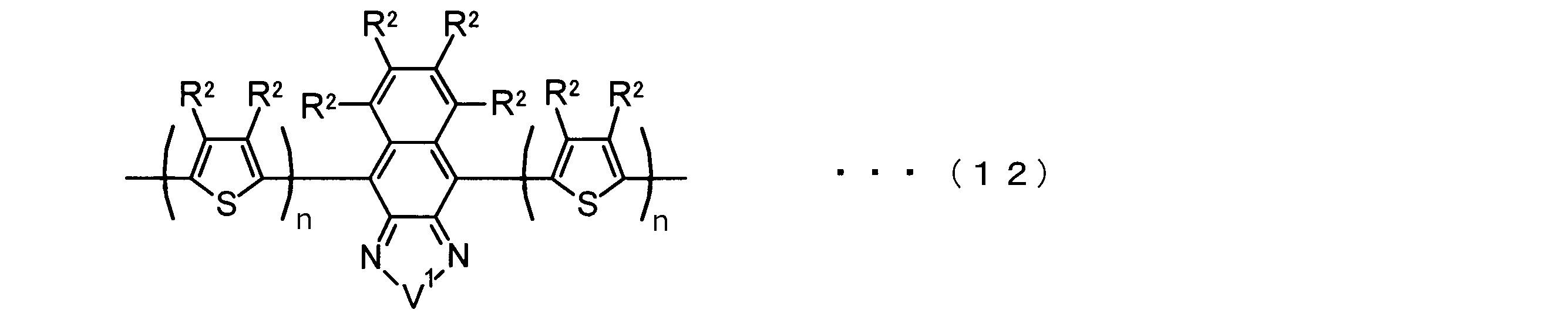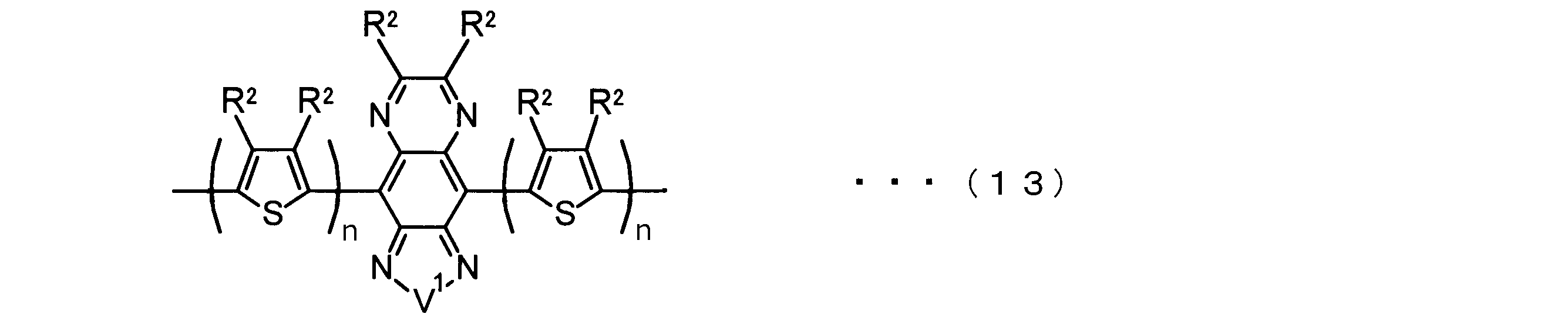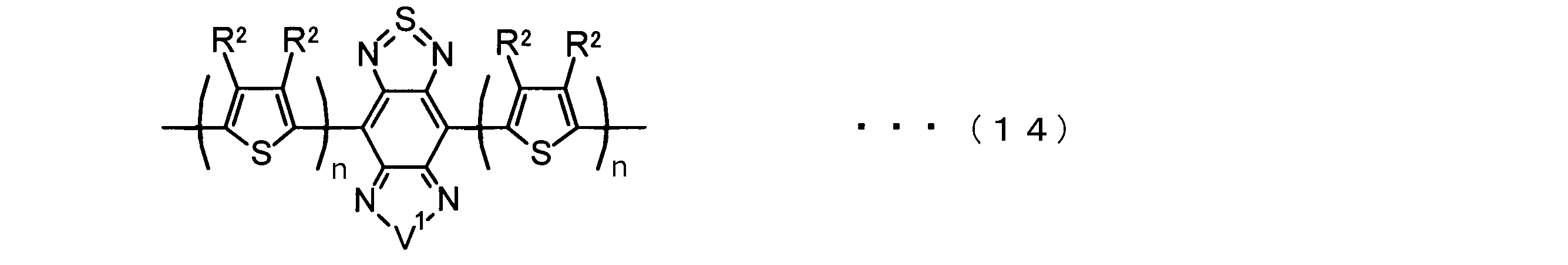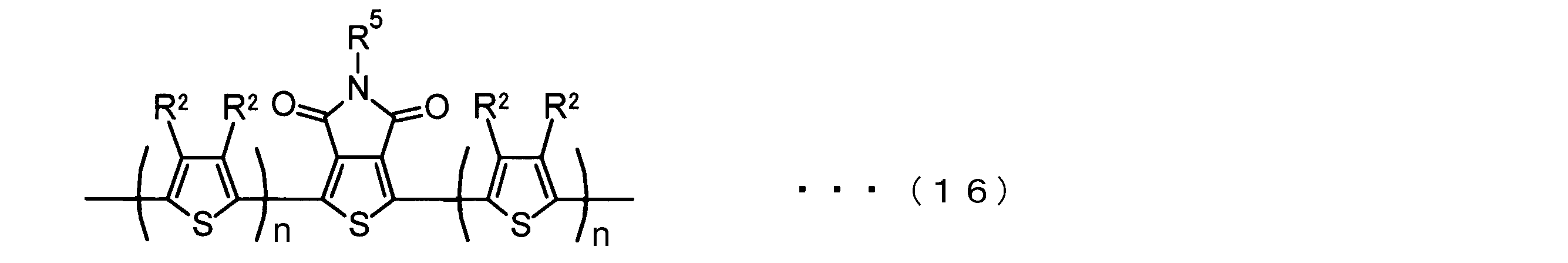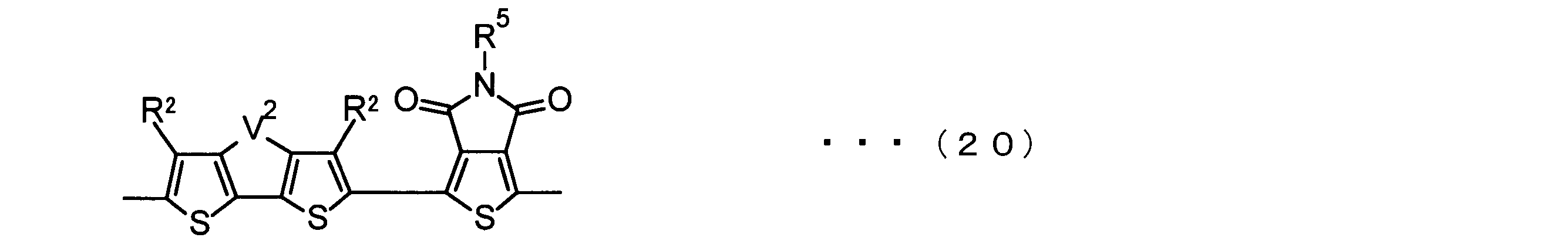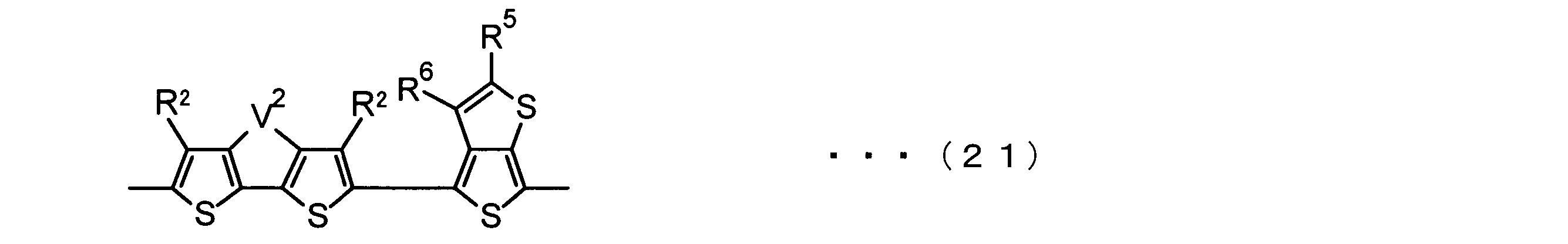WO2012133793A1 - ブロック共重合体および光電変換素子 - Google Patents
ブロック共重合体および光電変換素子 Download PDFInfo
- Publication number
- WO2012133793A1 WO2012133793A1 PCT/JP2012/058617 JP2012058617W WO2012133793A1 WO 2012133793 A1 WO2012133793 A1 WO 2012133793A1 JP 2012058617 W JP2012058617 W JP 2012058617W WO 2012133793 A1 WO2012133793 A1 WO 2012133793A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- polymer block
- block copolymer
- polymer
- electron
- Prior art date
Links
- 229920001400 block copolymer Polymers 0.000 title claims abstract description 109
- 238000006243 chemical reaction Methods 0.000 title claims abstract description 103
- 229920000642 polymer Polymers 0.000 claims abstract description 191
- 239000000178 monomer Substances 0.000 claims abstract description 80
- -1 thiophene-2, 5-diyl group Chemical group 0.000 claims abstract description 77
- 239000000463 material Substances 0.000 claims abstract description 43
- 239000000203 mixture Substances 0.000 claims abstract description 40
- 125000001424 substituent group Chemical group 0.000 claims abstract description 31
- 125000001072 heteroaryl group Chemical group 0.000 claims abstract description 19
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 claims abstract description 15
- UJOBWOGCFQCDNV-UHFFFAOYSA-N 9H-carbazole Chemical compound C1=CC=C2C3=CC=CC=C3NC2=C1 UJOBWOGCFQCDNV-UHFFFAOYSA-N 0.000 claims abstract description 14
- 239000000126 substance Substances 0.000 claims abstract description 9
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 claims abstract description 7
- 125000004432 carbon atom Chemical group C* 0.000 claims description 23
- 125000005843 halogen group Chemical group 0.000 claims description 20
- 125000000217 alkyl group Chemical group 0.000 claims description 19
- 125000003545 alkoxy group Chemical group 0.000 claims description 17
- 125000003118 aryl group Chemical group 0.000 claims description 17
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 16
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 12
- XMWRBQBLMFGWIX-UHFFFAOYSA-N C60 fullerene Chemical compound C12=C3C(C4=C56)=C7C8=C5C5=C9C%10=C6C6=C4C1=C1C4=C6C6=C%10C%10=C9C9=C%11C5=C8C5=C8C7=C3C3=C7C2=C1C1=C2C4=C6C4=C%10C6=C9C9=C%11C5=C5C8=C3C3=C7C1=C1C2=C4C6=C2C9=C5C3=C12 XMWRBQBLMFGWIX-UHFFFAOYSA-N 0.000 claims description 10
- 229910052757 nitrogen Inorganic materials 0.000 claims description 10
- 125000000547 substituted alkyl group Chemical group 0.000 claims description 10
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 8
- 229910052799 carbon Inorganic materials 0.000 claims description 7
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 6
- 239000001301 oxygen Substances 0.000 claims description 6
- 229910052760 oxygen Inorganic materials 0.000 claims description 6
- 229920005604 random copolymer Polymers 0.000 claims description 5
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 4
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 4
- 229910052732 germanium Inorganic materials 0.000 claims description 4
- GNPVGFCGXDBREM-UHFFFAOYSA-N germanium atom Chemical compound [Ge] GNPVGFCGXDBREM-UHFFFAOYSA-N 0.000 claims description 4
- 229910052710 silicon Inorganic materials 0.000 claims description 4
- 239000010703 silicon Substances 0.000 claims description 4
- 229910052717 sulfur Inorganic materials 0.000 claims description 4
- 239000011593 sulfur Substances 0.000 claims description 4
- 125000004453 alkoxycarbonyl group Chemical group 0.000 claims description 3
- 125000004448 alkyl carbonyl group Chemical group 0.000 claims description 3
- 229910003472 fullerene Inorganic materials 0.000 claims description 2
- 230000003287 optical effect Effects 0.000 abstract description 7
- 238000010521 absorption reaction Methods 0.000 abstract description 5
- 238000000034 method Methods 0.000 description 70
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 60
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 42
- 239000002904 solvent Substances 0.000 description 42
- 238000006116 polymerization reaction Methods 0.000 description 36
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 34
- 239000000243 solution Substances 0.000 description 34
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 32
- 239000010409 thin film Substances 0.000 description 31
- 239000010410 layer Substances 0.000 description 30
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 29
- 239000007787 solid Substances 0.000 description 29
- 150000001875 compounds Chemical class 0.000 description 24
- 238000005160 1H NMR spectroscopy Methods 0.000 description 23
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 18
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 17
- 230000000052 comparative effect Effects 0.000 description 17
- 239000012043 crude product Substances 0.000 description 17
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 14
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 description 14
- 238000005859 coupling reaction Methods 0.000 description 14
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 14
- 229920000547 conjugated polymer Polymers 0.000 description 13
- 238000001914 filtration Methods 0.000 description 13
- 229920003046 tetrablock copolymer Polymers 0.000 description 13
- 238000000576 coating method Methods 0.000 description 12
- 239000012299 nitrogen atmosphere Substances 0.000 description 12
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 12
- 0 C*c1c(*)c(*)c(C2=C3N=C(*)C(*)=NC3=C(*c3c(*)c(*)c(C)[s]3)C3=NC*N=C23)[s]1 Chemical compound C*c1c(*)c(*)c(C2=C3N=C(*)C(*)=NC3=C(*c3c(*)c(*)c(C)[s]3)C3=NC*N=C23)[s]1 0.000 description 11
- 229920001577 copolymer Polymers 0.000 description 11
- 238000004519 manufacturing process Methods 0.000 description 11
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 10
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 9
- 235000002597 Solanum melongena Nutrition 0.000 description 9
- MCEWYIDBDVPMES-UHFFFAOYSA-N [60]pcbm Chemical compound C123C(C4=C5C6=C7C8=C9C%10=C%11C%12=C%13C%14=C%15C%16=C%17C%18=C(C=%19C=%20C%18=C%18C%16=C%13C%13=C%11C9=C9C7=C(C=%20C9=C%13%18)C(C7=%19)=C96)C6=C%11C%17=C%15C%13=C%15C%14=C%12C%12=C%10C%10=C85)=C9C7=C6C2=C%11C%13=C2C%15=C%12C%10=C4C23C1(CCCC(=O)OC)C1=CC=CC=C1 MCEWYIDBDVPMES-UHFFFAOYSA-N 0.000 description 9
- 239000010408 film Substances 0.000 description 9
- 238000005227 gel permeation chromatography Methods 0.000 description 9
- 239000000758 substrate Substances 0.000 description 9
- 125000003396 thiol group Chemical group [H]S* 0.000 description 9
- 229920000428 triblock copolymer Polymers 0.000 description 9
- RFFLAFLAYFXFSW-UHFFFAOYSA-N 1,2-dichlorobenzene Chemical compound ClC1=CC=CC=C1Cl RFFLAFLAYFXFSW-UHFFFAOYSA-N 0.000 description 8
- 125000003277 amino group Chemical group 0.000 description 8
- 230000015572 biosynthetic process Effects 0.000 description 8
- 239000003054 catalyst Substances 0.000 description 8
- 229920000359 diblock copolymer Polymers 0.000 description 8
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 8
- 238000003786 synthesis reaction Methods 0.000 description 8
- 125000000524 functional group Chemical group 0.000 description 7
- 238000005259 measurement Methods 0.000 description 7
- 229910052751 metal Inorganic materials 0.000 description 7
- 239000002184 metal Substances 0.000 description 7
- 238000003756 stirring Methods 0.000 description 7
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 6
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 6
- 239000000654 additive Substances 0.000 description 6
- 230000000996 additive effect Effects 0.000 description 6
- 125000004185 ester group Chemical group 0.000 description 6
- 238000010438 heat treatment Methods 0.000 description 6
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Chemical compound [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 6
- 239000004065 semiconductor Substances 0.000 description 6
- DHWADSHFLSDMBJ-UHFFFAOYSA-N 4,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-2,1,3-benzothiadiazole Chemical compound O1C(C)(C)C(C)(C)OB1C(C1=NSN=C11)=CC=C1B1OC(C)(C)C(C)(C)O1 DHWADSHFLSDMBJ-UHFFFAOYSA-N 0.000 description 5
- 238000009835 boiling Methods 0.000 description 5
- 239000011248 coating agent Substances 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 230000005525 hole transport Effects 0.000 description 5
- 230000031700 light absorption Effects 0.000 description 5
- 239000012044 organic layer Substances 0.000 description 5
- 150000002901 organomagnesium compounds Chemical class 0.000 description 5
- 238000000746 purification Methods 0.000 description 5
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 5
- COIOYMYWGDAQPM-UHFFFAOYSA-N tris(2-methylphenyl)phosphane Chemical compound CC1=CC=CC=C1P(C=1C(=CC=CC=1)C)C1=CC=CC=C1C COIOYMYWGDAQPM-UHFFFAOYSA-N 0.000 description 5
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 4
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 4
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 4
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 4
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 4
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 4
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 4
- 125000002723 alicyclic group Chemical group 0.000 description 4
- 229920005603 alternating copolymer Polymers 0.000 description 4
- RDOXTESZEPMUJZ-UHFFFAOYSA-N anisole Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 4
- QARVLSVVCXYDNA-UHFFFAOYSA-N bromobenzene Chemical compound BrC1=CC=CC=C1 QARVLSVVCXYDNA-UHFFFAOYSA-N 0.000 description 4
- XJHCXCQVJFPJIK-UHFFFAOYSA-M caesium fluoride Chemical compound [F-].[Cs+] XJHCXCQVJFPJIK-UHFFFAOYSA-M 0.000 description 4
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical compound C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 4
- 238000001035 drying Methods 0.000 description 4
- 239000011259 mixed solution Substances 0.000 description 4
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 4
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 4
- 239000011368 organic material Substances 0.000 description 4
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical compound N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 4
- 229920000553 poly(phenylenevinylene) Polymers 0.000 description 4
- 238000004528 spin coating Methods 0.000 description 4
- LVEYOSJUKRVCCF-UHFFFAOYSA-N 1,3-Bis(diphenylphosphino)propane Substances C=1C=CC=CC=1P(C=1C=CC=CC=1)CCCP(C=1C=CC=CC=1)C1=CC=CC=C1 LVEYOSJUKRVCCF-UHFFFAOYSA-N 0.000 description 3
- OCJBOOLMMGQPQU-UHFFFAOYSA-N 1,4-dichlorobenzene Chemical compound ClC1=CC=C(Cl)C=C1 OCJBOOLMMGQPQU-UHFFFAOYSA-N 0.000 description 3
- JEDHEMYZURJGRQ-UHFFFAOYSA-N 3-hexylthiophene Chemical compound CCCCCCC=1C=CSC=1 JEDHEMYZURJGRQ-UHFFFAOYSA-N 0.000 description 3
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- DAXYAZKTXFDONJ-UHFFFAOYSA-N CCCCCCc1c(C(C)C)[s]c(C(C)(C)c([s]2)cc(C)c2Br)c1 Chemical compound CCCCCCc1c(C(C)C)[s]c(C(C)(C)c([s]2)cc(C)c2Br)c1 DAXYAZKTXFDONJ-UHFFFAOYSA-N 0.000 description 3
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- UUIQMZJEGPQKFD-UHFFFAOYSA-N Methyl butyrate Chemical compound CCCC(=O)OC UUIQMZJEGPQKFD-UHFFFAOYSA-N 0.000 description 3
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 3
- 238000000137 annealing Methods 0.000 description 3
- 229910052786 argon Inorganic materials 0.000 description 3
- 125000000732 arylene group Chemical group 0.000 description 3
- 239000002981 blocking agent Substances 0.000 description 3
- ZADPBFCGQRWHPN-UHFFFAOYSA-N boronic acid Chemical compound OBO ZADPBFCGQRWHPN-UHFFFAOYSA-N 0.000 description 3
- 125000005620 boronic acid group Chemical group 0.000 description 3
- 239000002041 carbon nanotube Substances 0.000 description 3
- 229910021393 carbon nanotube Inorganic materials 0.000 description 3
- 238000010168 coupling process Methods 0.000 description 3
- RJVPHPXQVBVPLV-UHFFFAOYSA-N cyclopenta[2,1-b:3,4-b']dithiophene Chemical compound S1C=CC2=CC3=CCSC3=C21 RJVPHPXQVBVPLV-UHFFFAOYSA-N 0.000 description 3
- 229940117389 dichlorobenzene Drugs 0.000 description 3
- 230000001747 exhibiting effect Effects 0.000 description 3
- RMBPEFMHABBEKP-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2C3=C[CH]C=CC3=CC2=C1 RMBPEFMHABBEKP-UHFFFAOYSA-N 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 238000002290 gas chromatography-mass spectrometry Methods 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 230000001771 impaired effect Effects 0.000 description 3
- 229910052740 iodine Inorganic materials 0.000 description 3
- 229920002521 macromolecule Polymers 0.000 description 3
- XKBGEWXEAPTVCK-UHFFFAOYSA-M methyltrioctylammonium chloride Chemical compound [Cl-].CCCCCCCC[N+](C)(CCCCCCCC)CCCCCCCC XKBGEWXEAPTVCK-UHFFFAOYSA-M 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 125000002950 monocyclic group Chemical group 0.000 description 3
- YTVNOVQHSGMMOV-UHFFFAOYSA-N naphthalenetetracarboxylic dianhydride Chemical compound C1=CC(C(=O)OC2=O)=C3C2=CC=C2C(=O)OC(=O)C1=C32 YTVNOVQHSGMMOV-UHFFFAOYSA-N 0.000 description 3
- 238000005191 phase separation Methods 0.000 description 3
- CLYVDMAATCIVBF-UHFFFAOYSA-N pigment red 224 Chemical compound C=12C3=CC=C(C(OC4=O)=O)C2=C4C=CC=1C1=CC=C2C(=O)OC(=O)C4=CC=C3C1=C42 CLYVDMAATCIVBF-UHFFFAOYSA-N 0.000 description 3
- 229920000301 poly(3-hexylthiophene-2,5-diyl) polymer Polymers 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 229930192474 thiophene Natural products 0.000 description 3
- 238000001771 vacuum deposition Methods 0.000 description 3
- CYPYTURSJDMMMP-WVCUSYJESA-N (1e,4e)-1,5-diphenylpenta-1,4-dien-3-one;palladium Chemical compound [Pd].[Pd].C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1 CYPYTURSJDMMMP-WVCUSYJESA-N 0.000 description 2
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 2
- FNQJDLTXOVEEFB-UHFFFAOYSA-N 1,2,3-benzothiadiazole Chemical compound C1=CC=C2SN=NC2=C1 FNQJDLTXOVEEFB-UHFFFAOYSA-N 0.000 description 2
- RELMFMZEBKVZJC-UHFFFAOYSA-N 1,2,3-trichlorobenzene Chemical compound ClC1=CC=CC(Cl)=C1Cl RELMFMZEBKVZJC-UHFFFAOYSA-N 0.000 description 2
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 2
- SGHBPXOHQQYDQZ-UHFFFAOYSA-N 1-bromo-8-iodooctane Chemical compound BrCCCCCCCCI SGHBPXOHQQYDQZ-UHFFFAOYSA-N 0.000 description 2
- DURPTKYDGMDSBL-UHFFFAOYSA-N 1-butoxybutane Chemical compound CCCCOCCCC DURPTKYDGMDSBL-UHFFFAOYSA-N 0.000 description 2
- CXBDYQVECUFKRK-UHFFFAOYSA-N 1-methoxybutane Chemical compound CCCCOC CXBDYQVECUFKRK-UHFFFAOYSA-N 0.000 description 2
- KBVDUUXRXJTAJC-UHFFFAOYSA-N 2,5-dibromothiophene Chemical compound BrC1=CC=C(Br)S1 KBVDUUXRXJTAJC-UHFFFAOYSA-N 0.000 description 2
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 2
- SDMKEQYHISDGKT-UHFFFAOYSA-N 2-bromo-3-hexyl-5-iodothiophene Chemical compound CCCCCCC=1C=C(I)SC=1Br SDMKEQYHISDGKT-UHFFFAOYSA-N 0.000 description 2
- JWUJQDFVADABEY-UHFFFAOYSA-N 2-methyltetrahydrofuran Chemical compound CC1CCCO1 JWUJQDFVADABEY-UHFFFAOYSA-N 0.000 description 2
- KPOCSQCZXMATFR-UHFFFAOYSA-N 3-butylthiophene Chemical compound CCCCC=1C=CSC=1 KPOCSQCZXMATFR-UHFFFAOYSA-N 0.000 description 2
- WQYWXQCOYRZFAV-UHFFFAOYSA-N 3-octylthiophene Chemical compound CCCCCCCCC=1C=CSC=1 WQYWXQCOYRZFAV-UHFFFAOYSA-N 0.000 description 2
- KKLCYBZPQDOFQK-UHFFFAOYSA-N 4,4,5,5-tetramethyl-2-phenyl-1,3,2-dioxaborolane Chemical compound O1C(C)(C)C(C)(C)OB1C1=CC=CC=C1 KKLCYBZPQDOFQK-UHFFFAOYSA-N 0.000 description 2
- 239000005964 Acibenzolar-S-methyl Substances 0.000 description 2
- 239000007818 Grignard reagent Substances 0.000 description 2
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 2
- XOBKSJJDNFUZPF-UHFFFAOYSA-N Methoxyethane Chemical compound CCOC XOBKSJJDNFUZPF-UHFFFAOYSA-N 0.000 description 2
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- 238000005481 NMR spectroscopy Methods 0.000 description 2
- XYFCBTPGUUZFHI-UHFFFAOYSA-N Phosphine Chemical compound P XYFCBTPGUUZFHI-UHFFFAOYSA-N 0.000 description 2
- 239000004793 Polystyrene Substances 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 2
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- 239000000443 aerosol Substances 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 150000001350 alkyl halides Chemical class 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 2
- YCOXTKKNXUZSKD-UHFFFAOYSA-N as-o-xylenol Natural products CC1=CC=C(O)C=C1C YCOXTKKNXUZSKD-UHFFFAOYSA-N 0.000 description 2
- 238000007611 bar coating method Methods 0.000 description 2
- 239000011324 bead Substances 0.000 description 2
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 2
- 229910052794 bromium Inorganic materials 0.000 description 2
- DIKBFYAXUHHXCS-UHFFFAOYSA-N bromoform Chemical compound BrC(Br)Br DIKBFYAXUHHXCS-UHFFFAOYSA-N 0.000 description 2
- 229910052804 chromium Inorganic materials 0.000 description 2
- 239000011651 chromium Substances 0.000 description 2
- 229920001940 conductive polymer Polymers 0.000 description 2
- 230000008878 coupling Effects 0.000 description 2
- 238000007766 curtain coating Methods 0.000 description 2
- 125000002933 cyclohexyloxy group Chemical group C1(CCCCC1)O* 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 230000018044 dehydration Effects 0.000 description 2
- 238000006297 dehydration reaction Methods 0.000 description 2
- 238000003618 dip coating Methods 0.000 description 2
- POLCUAVZOMRGSN-UHFFFAOYSA-N dipropyl ether Chemical compound CCCOCCC POLCUAVZOMRGSN-UHFFFAOYSA-N 0.000 description 2
- 150000002170 ethers Chemical class 0.000 description 2
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 150000004795 grignard reagents Chemical class 0.000 description 2
- XAPCMTMQBXLDBB-UHFFFAOYSA-N hexyl butyrate Chemical compound CCCCCCOC(=O)CCC XAPCMTMQBXLDBB-UHFFFAOYSA-N 0.000 description 2
- 229920001519 homopolymer Polymers 0.000 description 2
- 239000011630 iodine Substances 0.000 description 2
- SNHMUERNLJLMHN-UHFFFAOYSA-N iodobenzene Chemical compound IC1=CC=CC=C1 SNHMUERNLJLMHN-UHFFFAOYSA-N 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- 229910044991 metal oxide Inorganic materials 0.000 description 2
- 150000004706 metal oxides Chemical class 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- UZKWTJUDCOPSNM-UHFFFAOYSA-N methoxybenzene Substances CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 2
- SKTCDJAMAYNROS-UHFFFAOYSA-N methoxycyclopentane Chemical compound COC1CCCC1 SKTCDJAMAYNROS-UHFFFAOYSA-N 0.000 description 2
- 125000006606 n-butoxy group Chemical group 0.000 description 2
- 125000006610 n-decyloxy group Chemical group 0.000 description 2
- 125000006608 n-octyloxy group Chemical group 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- FDPIMTJIUBPUKL-UHFFFAOYSA-N pentan-3-one Chemical compound CCC(=O)CC FDPIMTJIUBPUKL-UHFFFAOYSA-N 0.000 description 2
- 230000000737 periodic effect Effects 0.000 description 2
- 150000002978 peroxides Chemical class 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 229920000767 polyaniline Polymers 0.000 description 2
- 229920002223 polystyrene Polymers 0.000 description 2
- 239000004810 polytetrafluoroethylene Substances 0.000 description 2
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 2
- 150000004033 porphyrin derivatives Chemical class 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- NROKBHXJSPEDAR-UHFFFAOYSA-M potassium fluoride Chemical compound [F-].[K+] NROKBHXJSPEDAR-UHFFFAOYSA-M 0.000 description 2
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 2
- 229920005989 resin Polymers 0.000 description 2
- 239000011347 resin Substances 0.000 description 2
- 238000007761 roller coating Methods 0.000 description 2
- 229930195734 saturated hydrocarbon Natural products 0.000 description 2
- 238000010898 silica gel chromatography Methods 0.000 description 2
- PUZPDOWCWNUUKD-UHFFFAOYSA-M sodium fluoride Chemical compound [F-].[Na+] PUZPDOWCWNUUKD-UHFFFAOYSA-M 0.000 description 2
- 229910052938 sodium sulfate Inorganic materials 0.000 description 2
- 235000011152 sodium sulphate Nutrition 0.000 description 2
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 2
- 238000005507 spraying Methods 0.000 description 2
- 238000004544 sputter deposition Methods 0.000 description 2
- 230000002194 synthesizing effect Effects 0.000 description 2
- 229910052718 tin Inorganic materials 0.000 description 2
- 229910052723 transition metal Inorganic materials 0.000 description 2
- 150000003624 transition metals Chemical class 0.000 description 2
- 238000002834 transmittance Methods 0.000 description 2
- YWWDBCBWQNCYNR-UHFFFAOYSA-N trimethylphosphine Chemical compound CP(C)C YWWDBCBWQNCYNR-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- BWHDROKFUHTORW-UHFFFAOYSA-N tritert-butylphosphane Chemical compound CC(C)(C)P(C(C)(C)C)C(C)(C)C BWHDROKFUHTORW-UHFFFAOYSA-N 0.000 description 2
- 238000007740 vapor deposition Methods 0.000 description 2
- 239000008096 xylene Substances 0.000 description 2
- JHPBVORIWHFCDS-UHFFFAOYSA-N (3-diphenylphosphanyl-2,2-dimethylpropyl)-diphenylphosphane Chemical compound C=1C=CC=CC=1P(C=1C=CC=CC=1)CC(C)(C)CP(C=1C=CC=CC=1)C1=CC=CC=C1 JHPBVORIWHFCDS-UHFFFAOYSA-N 0.000 description 1
- BODYVHJTUHHINQ-UHFFFAOYSA-N (4-boronophenyl)boronic acid Chemical compound OB(O)C1=CC=C(B(O)O)C=C1 BODYVHJTUHHINQ-UHFFFAOYSA-N 0.000 description 1
- SKFDVFMUXZWWOW-UHFFFAOYSA-N (5-boronothiophen-2-yl)boronic acid Chemical compound OB(O)C1=CC=C(B(O)O)S1 SKFDVFMUXZWWOW-UHFFFAOYSA-N 0.000 description 1
- GMRJIOFCAMKRNJ-UHFFFAOYSA-N 1$l^{2}-germole Chemical compound [Ge]1C=CC=C1 GMRJIOFCAMKRNJ-UHFFFAOYSA-N 0.000 description 1
- XGCDBGRZEKYHNV-UHFFFAOYSA-N 1,1-bis(diphenylphosphino)methane Chemical compound C=1C=CC=CC=1P(C=1C=CC=CC=1)CP(C=1C=CC=CC=1)C1=CC=CC=C1 XGCDBGRZEKYHNV-UHFFFAOYSA-N 0.000 description 1
- STBMZSJLFYGOJU-UHFFFAOYSA-N 1,1-dibromooctane Chemical compound CCCCCCCC(Br)Br STBMZSJLFYGOJU-UHFFFAOYSA-N 0.000 description 1
- DKLWRIQKXIBVIS-UHFFFAOYSA-N 1,1-diiodooctane Chemical compound CCCCCCCC(I)I DKLWRIQKXIBVIS-UHFFFAOYSA-N 0.000 description 1
- UDGKZGLPXCRRAM-UHFFFAOYSA-N 1,2,5-thiadiazole Chemical compound C=1C=NSN=1 UDGKZGLPXCRRAM-UHFFFAOYSA-N 0.000 description 1
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 1
- QFMZQPDHXULLKC-UHFFFAOYSA-N 1,2-bis(diphenylphosphino)ethane Chemical compound C=1C=CC=CC=1P(C=1C=CC=CC=1)CCP(C=1C=CC=CC=1)C1=CC=CC=C1 QFMZQPDHXULLKC-UHFFFAOYSA-N 0.000 description 1
- SWJPEBQEEAHIGZ-UHFFFAOYSA-N 1,4-dibromobenzene Chemical compound BrC1=CC=C(Br)C=C1 SWJPEBQEEAHIGZ-UHFFFAOYSA-N 0.000 description 1
- DAMSFPUDFJGSHP-UHFFFAOYSA-N 1-(4-phenylphenyl)triazole Chemical class C1(=CC=C(C=C1)N1N=NC=C1)C1=CC=CC=C1 DAMSFPUDFJGSHP-UHFFFAOYSA-N 0.000 description 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 1
- OHZAHWOAMVVGEL-UHFFFAOYSA-N 2,2'-bithiophene Chemical compound C1=CSC(C=2SC=CC=2)=C1 OHZAHWOAMVVGEL-UHFFFAOYSA-N 0.000 description 1
- KXSFECAJUBPPFE-UHFFFAOYSA-N 2,2':5',2''-terthiophene Chemical compound C1=CSC(C=2SC(=CC=2)C=2SC=CC=2)=C1 KXSFECAJUBPPFE-UHFFFAOYSA-N 0.000 description 1
- RMBYPCXALOMPTA-UHFFFAOYSA-N 2,5-dibromo-3-(2-hexyldecyl)thiophene Chemical compound CCCCCCCCC(CCCCCC)CC=1C=C(Br)SC=1Br RMBYPCXALOMPTA-UHFFFAOYSA-N 0.000 description 1
- MUNFOTHAFHGRIM-UHFFFAOYSA-N 2,5-dinaphthalen-1-yl-1,3,4-oxadiazole Chemical compound C1=CC=C2C(C3=NN=C(O3)C=3C4=CC=CC=C4C=CC=3)=CC=CC2=C1 MUNFOTHAFHGRIM-UHFFFAOYSA-N 0.000 description 1
- CBSSXAWGKPSJNX-UHFFFAOYSA-N 2-(2-ethylhexyl)pyrrolo[3,2-b]pyrrole-5,6-dione Chemical compound C(C)C(CC=1N=C2C(C=1)=NC(C2=O)=O)CCCC CBSSXAWGKPSJNX-UHFFFAOYSA-N 0.000 description 1
- JACCFQMSOHCQFN-UHFFFAOYSA-N 2-(2-ethylhexyl)thiophene Chemical compound CCCCC(CC)CC1=CC=CS1 JACCFQMSOHCQFN-UHFFFAOYSA-N 0.000 description 1
- QZVHYFUVMQIGGM-UHFFFAOYSA-N 2-Hexylthiophene Chemical compound CCCCCCC1=CC=CS1 QZVHYFUVMQIGGM-UHFFFAOYSA-N 0.000 description 1
- QUDFKKSCXOANSC-UHFFFAOYSA-N 2-ethyl-1-thieno[2,3-c]thiophen-2-ylhexan-1-one Chemical compound S1C=C2SC(C(=O)C(CC)CCCC)=CC2=C1 QUDFKKSCXOANSC-UHFFFAOYSA-N 0.000 description 1
- NSMJMUQZRGZMQC-UHFFFAOYSA-N 2-naphthalen-1-yl-1H-imidazo[4,5-f][1,10]phenanthroline Chemical compound C12=CC=CN=C2C2=NC=CC=C2C2=C1NC(C=1C3=CC=CC=C3C=CC=1)=N2 NSMJMUQZRGZMQC-UHFFFAOYSA-N 0.000 description 1
- 125000001622 2-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C(*)C([H])=C([H])C2=C1[H] 0.000 description 1
- 125000000175 2-thienyl group Chemical group S1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- YIZCFNBOVXMTMO-UHFFFAOYSA-N 3-(4-tert-butylphenyl)-4-phenyl-1,2,4-triazole Chemical compound C1=CC(C(C)(C)C)=CC=C1C1=NN=CN1C1=CC=CC=C1 YIZCFNBOVXMTMO-UHFFFAOYSA-N 0.000 description 1
- HKQPCIWNHQRHGQ-UHFFFAOYSA-N 3-(phenoxymethyl)thiophene Chemical compound C1=CSC=C1COC1=CC=CC=C1 HKQPCIWNHQRHGQ-UHFFFAOYSA-N 0.000 description 1
- XCMISAPCWHTVNG-UHFFFAOYSA-N 3-bromothiophene Chemical compound BrC=1C=CSC=1 XCMISAPCWHTVNG-UHFFFAOYSA-N 0.000 description 1
- FGHYZEFIPLFAOC-UHFFFAOYSA-N 3-cyclohexylthiophene Chemical compound C1CCCCC1C1=CSC=C1 FGHYZEFIPLFAOC-UHFFFAOYSA-N 0.000 description 1
- UOJCDDLTVQJPGH-UHFFFAOYSA-N 4,4,5,5-tetramethyl-2-[4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-1,3,2-dioxaborolane Chemical compound O1C(C)(C)C(C)(C)OB1C1=CC=C(B2OC(C)(C)C(C)(C)O2)C=C1 UOJCDDLTVQJPGH-UHFFFAOYSA-N 0.000 description 1
- AOJXAKMKFDBHHT-UHFFFAOYSA-N 4,4,5,5-tetramethyl-2-[5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)thiophen-2-yl]-1,3,2-dioxaborolane Chemical compound O1C(C)(C)C(C)(C)OB1C1=CC=C(B2OC(C)(C)C(C)(C)O2)S1 AOJXAKMKFDBHHT-UHFFFAOYSA-N 0.000 description 1
- FEXAKNVHUAEACM-UHFFFAOYSA-N 4,6-dibromothieno[2,3-c]thiophene Chemical compound S1C=CC2=C(Br)SC(Br)=C21 FEXAKNVHUAEACM-UHFFFAOYSA-N 0.000 description 1
- HATOWNJGYIVNBU-UHFFFAOYSA-N 4,8-bis(2-ethylhexoxy)thieno[2,3-f][1]benzothiole Chemical compound CCCCC(CC)COC1=C2C=CSC2=C(OCC(CC)CCCC)C2=C1SC=C2 HATOWNJGYIVNBU-UHFFFAOYSA-N 0.000 description 1
- ZJIRFPOFCZNBAC-UHFFFAOYSA-N 4-amino-2-(2-amino-2-carboxyethyl)sulfanylbutanoic acid Chemical compound NCCC(C(O)=O)SCC(N)C(O)=O ZJIRFPOFCZNBAC-UHFFFAOYSA-N 0.000 description 1
- 108010020212 4-amino-2-(S-cysteinyl)butyric acid Proteins 0.000 description 1
- BKYWEUVIGUEMFX-UHFFFAOYSA-N 4h-dithieno[3,2-a:2',3'-d]pyrrole Chemical compound S1C=CC2=C1NC1=C2SC=C1 BKYWEUVIGUEMFX-UHFFFAOYSA-N 0.000 description 1
- GNBBEOGRQXANFQ-UHFFFAOYSA-N 9,9-didecylfluorene Chemical compound C1=CC=C2C(CCCCCCCCCC)(CCCCCCCCCC)C3=CC=CC=C3C2=C1 GNBBEOGRQXANFQ-UHFFFAOYSA-N 0.000 description 1
- 102100030840 AT-rich interactive domain-containing protein 4B Human genes 0.000 description 1
- ROFVEXUMMXZLPA-UHFFFAOYSA-N Bipyridyl Chemical compound N1=CC=CC=C1C1=CC=CC=N1 ROFVEXUMMXZLPA-UHFFFAOYSA-N 0.000 description 1
- IFRKIUCUEJKAEN-UHFFFAOYSA-N BrC(CC=1SC=CC1)C(CC(C)I)CC Chemical compound BrC(CC=1SC=CC1)C(CC(C)I)CC IFRKIUCUEJKAEN-UHFFFAOYSA-N 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- XDWYGQPKYBRBPF-UHFFFAOYSA-N CCC1C([BrH]C)=C(C)C=C1C Chemical compound CCC1C([BrH]C)=C(C)C=C1C XDWYGQPKYBRBPF-UHFFFAOYSA-N 0.000 description 1
- UEXGEENLNDOPOW-UHFFFAOYSA-N CCC1C([BrH]C)=C(C)C=C1I Chemical compound CCC1C([BrH]C)=C(C)C=C1I UEXGEENLNDOPOW-UHFFFAOYSA-N 0.000 description 1
- DKUHTLRFEWWLCF-UHFFFAOYSA-N CCCCC(CC)CC1C=C2C(=C3C=CSC3=[GeH]2)S1 Chemical compound CCCCC(CC)CC1C=C2C(=C3C=CSC3=[GeH]2)S1 DKUHTLRFEWWLCF-UHFFFAOYSA-N 0.000 description 1
- OVJCJWFVLAWOJC-UHFFFAOYSA-N CCCCC(CC)CN(C=C(C1=C(C(S2)=CC=C2Br)N2CC(CC)CCCC)C2=O)C1=O Chemical compound CCCCC(CC)CN(C=C(C1=C(C(S2)=CC=C2Br)N2CC(CC)CCCC)C2=O)C1=O OVJCJWFVLAWOJC-UHFFFAOYSA-N 0.000 description 1
- YPNQMBPCRALKAJ-UHFFFAOYSA-N CCCCCCC(C=C(C)C1CC)=C1[BrH]C Chemical compound CCCCCCC(C=C(C)C1CC)=C1[BrH]C YPNQMBPCRALKAJ-UHFFFAOYSA-N 0.000 description 1
- GCYFQUALVDRTGN-UHFFFAOYSA-N CCCCCCC(C=C(C1CC)I)=C1[BrH]C Chemical compound CCCCCCC(C=C(C1CC)I)=C1[BrH]C GCYFQUALVDRTGN-UHFFFAOYSA-N 0.000 description 1
- NXHDPTBJFSSSLO-UHFFFAOYSA-N CCCCCCCCC(C)[n]1c2cc(-c3ccc(-c4ccc(-c5ccc(C(c6ccccc6)=C)[s]5)c5n[s]nc45)[s]3)ccc2c2c1cc(C(B1OC(C)(C)C(C)(C)O1)C=C)cc2 Chemical compound CCCCCCCCC(C)[n]1c2cc(-c3ccc(-c4ccc(-c5ccc(C(c6ccccc6)=C)[s]5)c5n[s]nc45)[s]3)ccc2c2c1cc(C(B1OC(C)(C)C(C)(C)O1)C=C)cc2 NXHDPTBJFSSSLO-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- 229940126062 Compound A Drugs 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- JEPXJYKYHMEESP-UHFFFAOYSA-N Dodecyl butyrate Chemical compound CCCCCCCCCCCCOC(=O)CCC JEPXJYKYHMEESP-UHFFFAOYSA-N 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- 208000033962 Fontaine progeroid syndrome Diseases 0.000 description 1
- NLDMNSXOCDLTTB-UHFFFAOYSA-N Heterophylliin A Natural products O1C2COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC2C(OC(=O)C=2C=C(O)C(O)=C(O)C=2)C(O)C1OC(=O)C1=CC(O)=C(O)C(O)=C1 NLDMNSXOCDLTTB-UHFFFAOYSA-N 0.000 description 1
- 101000792935 Homo sapiens AT-rich interactive domain-containing protein 4B Proteins 0.000 description 1
- 238000004566 IR spectroscopy Methods 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- KWYHDKDOAIKMQN-UHFFFAOYSA-N N,N,N',N'-tetramethylethylenediamine Chemical compound CN(C)CCN(C)C KWYHDKDOAIKMQN-UHFFFAOYSA-N 0.000 description 1
- SUAKHGWARZSWIH-UHFFFAOYSA-N N,N‐diethylformamide Chemical compound CCN(CC)C=O SUAKHGWARZSWIH-UHFFFAOYSA-N 0.000 description 1
- 101100438536 Nicotiana plumbaginifolia CABC gene Proteins 0.000 description 1
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 1
- 229920000144 PEDOT:PSS Polymers 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- 239000004734 Polyphenylene sulfide Substances 0.000 description 1
- 238000001069 Raman spectroscopy Methods 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical group [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- XXMOZDBOAIICDA-UHFFFAOYSA-N [4,8-bis(2-ethylhexoxy)-2-trimethylstannylthieno[2,3-f][1]benzothiol-6-yl]-trimethylstannane Chemical compound CCCCC(CC)COC1=C2C=C([Sn](C)(C)C)SC2=C(OCC(CC)CCCC)C2=C1SC([Sn](C)(C)C)=C2 XXMOZDBOAIICDA-UHFFFAOYSA-N 0.000 description 1
- XHCLAFWTIXFWPH-UHFFFAOYSA-N [O-2].[O-2].[O-2].[O-2].[O-2].[V+5].[V+5] Chemical compound [O-2].[O-2].[O-2].[O-2].[O-2].[V+5].[V+5] XHCLAFWTIXFWPH-UHFFFAOYSA-N 0.000 description 1
- 125000003668 acetyloxy group Chemical group [H]C([H])([H])C(=O)O[*] 0.000 description 1
- 125000004414 alkyl thio group Chemical group 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 229910021417 amorphous silicon Inorganic materials 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- ULKGULQGPBMIJU-UHFFFAOYSA-N benzene;hydron;bromide Chemical compound Br.C1=CC=CC=C1 ULKGULQGPBMIJU-UHFFFAOYSA-N 0.000 description 1
- 125000005605 benzo group Chemical group 0.000 description 1
- 125000001231 benzoyloxy group Chemical group C(C1=CC=CC=C1)(=O)O* 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 230000031709 bromination Effects 0.000 description 1
- 238000005893 bromination reaction Methods 0.000 description 1
- 229950005228 bromoform Drugs 0.000 description 1
- XUPYJHCZDLZNFP-UHFFFAOYSA-N butyl butanoate Chemical compound CCCCOC(=O)CCC XUPYJHCZDLZNFP-UHFFFAOYSA-N 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- WUKWITHWXAAZEY-UHFFFAOYSA-L calcium difluoride Chemical compound [F-].[F-].[Ca+2] WUKWITHWXAAZEY-UHFFFAOYSA-L 0.000 description 1
- 229910001634 calcium fluoride Inorganic materials 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 238000004581 coalescence Methods 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 239000004020 conductor Substances 0.000 description 1
- 150000004696 coordination complex Chemical class 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 125000004093 cyano group Chemical group *C#N 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- CVYZQFMDVPRSNE-UHFFFAOYSA-N dicyclohexyl(2-dicyclohexylphosphanylpropan-2-yl)phosphane Chemical compound C1CCCCC1P(C1CCCCC1)C(C)(C)P(C1CCCCC1)C1CCCCC1 CVYZQFMDVPRSNE-UHFFFAOYSA-N 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 239000007772 electrode material Substances 0.000 description 1
- 239000012156 elution solvent Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 239000003822 epoxy resin Substances 0.000 description 1
- 239000004210 ether based solvent Substances 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 125000000031 ethylamino group Chemical group [H]C([H])([H])C([H])([H])N([H])[*] 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- RBTKNAXYKSUFRK-UHFFFAOYSA-N heliogen blue Chemical compound [Cu].[N-]1C2=C(C=CC=C3)C3=C1N=C([N-]1)C3=CC=CC=C3C1=NC([N-]1)=C(C=CC=C3)C3=C1N=C([N-]1)C3=CC=CC=C3C1=N2 RBTKNAXYKSUFRK-UHFFFAOYSA-N 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- 229910052738 indium Inorganic materials 0.000 description 1
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 1
- AMGQUBHHOARCQH-UHFFFAOYSA-N indium;oxotin Chemical compound [In].[Sn]=O AMGQUBHHOARCQH-UHFFFAOYSA-N 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 239000012442 inert solvent Substances 0.000 description 1
- 239000003999 initiator Substances 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 230000026045 iodination Effects 0.000 description 1
- 238000006192 iodination reaction Methods 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 238000007733 ion plating Methods 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- RGFNRWTWDWVHDD-UHFFFAOYSA-N isobutyl butyrate Chemical compound CCCC(=O)OCC(C)C RGFNRWTWDWVHDD-UHFFFAOYSA-N 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000005928 isopropyloxycarbonyl group Chemical group [H]C([H])([H])C([H])(OC(*)=O)C([H])([H])[H] 0.000 description 1
- 239000005453 ketone based solvent Substances 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910001629 magnesium chloride Inorganic materials 0.000 description 1
- ORUIBWPALBXDOA-UHFFFAOYSA-L magnesium fluoride Chemical compound [F-].[F-].[Mg+2] ORUIBWPALBXDOA-UHFFFAOYSA-L 0.000 description 1
- 229910001635 magnesium fluoride Inorganic materials 0.000 description 1
- CQRPUKWAZPZXTO-UHFFFAOYSA-M magnesium;2-methylpropane;chloride Chemical compound [Mg+2].[Cl-].C[C-](C)C CQRPUKWAZPZXTO-UHFFFAOYSA-M 0.000 description 1
- LVKCSZQWLOVUGB-UHFFFAOYSA-M magnesium;propane;bromide Chemical compound [Mg+2].[Br-].C[CH-]C LVKCSZQWLOVUGB-UHFFFAOYSA-M 0.000 description 1
- IUYHWZFSGMZEOG-UHFFFAOYSA-M magnesium;propane;chloride Chemical compound [Mg+2].[Cl-].C[CH-]C IUYHWZFSGMZEOG-UHFFFAOYSA-M 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 230000015654 memory Effects 0.000 description 1
- 229910001512 metal fluoride Inorganic materials 0.000 description 1
- 238000005649 metathesis reaction Methods 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- UJNZOIKQAUQOCN-UHFFFAOYSA-N methyl(diphenyl)phosphane Chemical compound C=1C=CC=CC=1P(C)C1=CC=CC=C1 UJNZOIKQAUQOCN-UHFFFAOYSA-N 0.000 description 1
- 125000000250 methylamino group Chemical group [H]N(*)C([H])([H])[H] 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 239000002808 molecular sieve Substances 0.000 description 1
- 229910000476 molybdenum oxide Inorganic materials 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 229940078552 o-xylene Drugs 0.000 description 1
- WKVAXZCSIOTXBT-UHFFFAOYSA-N octane-1,1-dithiol Chemical compound CCCCCCCC(S)S WKVAXZCSIOTXBT-UHFFFAOYSA-N 0.000 description 1
- 125000005447 octyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 150000007978 oxazole derivatives Chemical class 0.000 description 1
- PQQKPALAQIIWST-UHFFFAOYSA-N oxomolybdenum Chemical compound [Mo]=O PQQKPALAQIIWST-UHFFFAOYSA-N 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 125000005010 perfluoroalkyl group Chemical group 0.000 description 1
- 150000005041 phenanthrolines Chemical class 0.000 description 1
- 125000006678 phenoxycarbonyl group Chemical group 0.000 description 1
- VBXSERPGINMTDE-UHFFFAOYSA-N phenyl(2-phenylphosphanylethyl)phosphane Chemical compound C=1C=CC=CC=1PCCPC1=CC=CC=C1 VBXSERPGINMTDE-UHFFFAOYSA-N 0.000 description 1
- AVNRJUHUOZDFKS-UHFFFAOYSA-N phenyl(3-phenylphosphanylpropyl)phosphane Chemical compound C=1C=CC=CC=1PCCCPC1=CC=CC=C1 AVNRJUHUOZDFKS-UHFFFAOYSA-N 0.000 description 1
- LRYYUQJFQWSHNC-UHFFFAOYSA-N phenyl(4-phenylphosphanylbutyl)phosphane Chemical compound C=1C=CC=CC=1PCCCCPC1=CC=CC=C1 LRYYUQJFQWSHNC-UHFFFAOYSA-N 0.000 description 1
- 150000003003 phosphines Chemical class 0.000 description 1
- 229910000073 phosphorus hydride Inorganic materials 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 238000007747 plating Methods 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229910021420 polycrystalline silicon Inorganic materials 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 229920000647 polyepoxide Polymers 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920002098 polyfluorene Polymers 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 239000002685 polymerization catalyst Substances 0.000 description 1
- 230000000379 polymerizing effect Effects 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 229920000069 polyphenylene sulfide Polymers 0.000 description 1
- 229920000123 polythiophene Polymers 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 239000011698 potassium fluoride Substances 0.000 description 1
- 235000003270 potassium fluoride Nutrition 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 238000002953 preparative HPLC Methods 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 125000004742 propyloxycarbonyl group Chemical group 0.000 description 1
- KOUKXHPPRFNWPP-UHFFFAOYSA-N pyrazine-2,5-dicarboxylic acid;hydrate Chemical group O.OC(=O)C1=CN=C(C(O)=O)C=N1 KOUKXHPPRFNWPP-UHFFFAOYSA-N 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- FYNROBRQIVCIQF-UHFFFAOYSA-N pyrrolo[3,2-b]pyrrole-5,6-dione Chemical compound C1=CN=C2C(=O)C(=O)N=C21 FYNROBRQIVCIQF-UHFFFAOYSA-N 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000004064 recycling Methods 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 238000001226 reprecipitation Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000000467 secondary amino group Chemical group [H]N([*:1])[*:2] 0.000 description 1
- 238000001004 secondary ion mass spectrometry Methods 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 1
- 239000011775 sodium fluoride Substances 0.000 description 1
- 235000013024 sodium fluoride Nutrition 0.000 description 1
- XHFLOLLMZOTPSM-UHFFFAOYSA-M sodium;hydrogen carbonate;hydrate Chemical compound [OH-].[Na+].OC(O)=O XHFLOLLMZOTPSM-UHFFFAOYSA-M 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 238000006557 surface reaction Methods 0.000 description 1
- 238000004381 surface treatment Methods 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 229940042055 systemic antimycotics triazole derivative Drugs 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229940073455 tetraethylammonium hydroxide Drugs 0.000 description 1
- LRGJRHZIDJQFCL-UHFFFAOYSA-M tetraethylazanium;hydroxide Chemical compound [OH-].CC[N+](CC)(CC)CC LRGJRHZIDJQFCL-UHFFFAOYSA-M 0.000 description 1
- 125000001412 tetrahydropyranyl group Chemical group 0.000 description 1
- URMVZUQDPPDABD-UHFFFAOYSA-N thieno[2,3-f][1]benzothiole Chemical compound C1=C2SC=CC2=CC2=C1C=CS2 URMVZUQDPPDABD-UHFFFAOYSA-N 0.000 description 1
- CRUIOQJBPNKOJG-UHFFFAOYSA-N thieno[3,2-e][1]benzothiole Chemical compound C1=C2SC=CC2=C2C=CSC2=C1 CRUIOQJBPNKOJG-UHFFFAOYSA-N 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 125000005556 thienylene group Chemical group 0.000 description 1
- 239000011135 tin Substances 0.000 description 1
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 1
- 229910001887 tin oxide Inorganic materials 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- WLPUWLXVBWGYMZ-UHFFFAOYSA-N tricyclohexylphosphine Chemical compound C1CCCCC1P(C1CCCCC1)C1CCCCC1 WLPUWLXVBWGYMZ-UHFFFAOYSA-N 0.000 description 1
- RXJKFRMDXUJTEX-UHFFFAOYSA-N triethylphosphine Chemical compound CCP(CC)CC RXJKFRMDXUJTEX-UHFFFAOYSA-N 0.000 description 1
- IGNTWNVBGLNYDV-UHFFFAOYSA-N triisopropylphosphine Chemical compound CC(C)P(C(C)C)C(C)C IGNTWNVBGLNYDV-UHFFFAOYSA-N 0.000 description 1
- 125000000025 triisopropylsilyl group Chemical group C(C)(C)[Si](C(C)C)(C(C)C)* 0.000 description 1
- GCCNTKCAHLLRQK-UHFFFAOYSA-N trimethyl-(2-thiophen-2-yl-4-trimethylstannylthiophen-3-yl)stannane Chemical compound C[Sn](C)(C)C1=CSC(C=2SC=CC=2)=C1[Sn](C)(C)C GCCNTKCAHLLRQK-UHFFFAOYSA-N 0.000 description 1
- PWYVVBKROXXHEB-UHFFFAOYSA-M trimethyl-[3-(1-methyl-2,3,4,5-tetraphenylsilol-1-yl)propyl]azanium;iodide Chemical compound [I-].C[N+](C)(C)CCC[Si]1(C)C(C=2C=CC=CC=2)=C(C=2C=CC=CC=2)C(C=2C=CC=CC=2)=C1C1=CC=CC=C1 PWYVVBKROXXHEB-UHFFFAOYSA-M 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 238000001291 vacuum drying Methods 0.000 description 1
- 229910052720 vanadium Inorganic materials 0.000 description 1
- 229910001935 vanadium oxide Inorganic materials 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- YVTHLONGBIQYBO-UHFFFAOYSA-N zinc indium(3+) oxygen(2-) Chemical compound [O--].[Zn++].[In+3] YVTHLONGBIQYBO-UHFFFAOYSA-N 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
Classifications
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/10—Organic polymers or oligomers
- H10K85/111—Organic polymers or oligomers comprising aromatic, heteroaromatic, or aryl chains, e.g. polyaniline, polyphenylene or polyphenylene vinylene
- H10K85/113—Heteroaromatic compounds comprising sulfur or selene, e.g. polythiophene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G61/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G61/12—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule
- C08G61/122—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule derived from five- or six-membered heterocyclic compounds, other than imides
- C08G61/123—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule derived from five- or six-membered heterocyclic compounds, other than imides derived from five-membered heterocyclic compounds
- C08G61/124—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule derived from five- or six-membered heterocyclic compounds, other than imides derived from five-membered heterocyclic compounds with a five-membered ring containing one nitrogen atom in the ring
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G61/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G61/12—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule
- C08G61/122—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule derived from five- or six-membered heterocyclic compounds, other than imides
- C08G61/123—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule derived from five- or six-membered heterocyclic compounds, other than imides derived from five-membered heterocyclic compounds
- C08G61/126—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule derived from five- or six-membered heterocyclic compounds, other than imides derived from five-membered heterocyclic compounds with a five-membered ring containing one sulfur atom in the ring
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/02—Elements
- C08K3/04—Carbon
- C08K3/045—Fullerenes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/10—Organic polymers or oligomers
- H10K85/151—Copolymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/10—Definition of the polymer structure
- C08G2261/12—Copolymers
- C08G2261/126—Copolymers block
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/32—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain
- C08G2261/322—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain non-condensed
- C08G2261/3223—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain non-condensed containing one or more sulfur atoms as the only heteroatom, e.g. thiophene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/32—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain
- C08G2261/324—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed
- C08G2261/3241—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed containing one or more nitrogen atoms as the only heteroatom, e.g. carbazole
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/32—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain
- C08G2261/324—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed
- C08G2261/3243—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed containing one or more sulfur atoms as the only heteroatom, e.g. benzothiophene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/32—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain
- C08G2261/324—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed
- C08G2261/3246—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed containing nitrogen and sulfur as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/34—Monomer units or repeat units incorporating structural elements in the main chain incorporating partially-aromatic structural elements in the main chain
- C08G2261/344—Monomer units or repeat units incorporating structural elements in the main chain incorporating partially-aromatic structural elements in the main chain containing heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/90—Applications
- C08G2261/91—Photovoltaic applications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/02—Elements
- C08K3/04—Carbon
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K30/00—Organic devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation
- H10K30/30—Organic devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation comprising bulk heterojunctions, e.g. interpenetrating networks of donor and acceptor material domains
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K30/00—Organic devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation
- H10K30/50—Photovoltaic [PV] devices
Definitions
- the present invention relates to a novel ⁇ -electron conjugated block copolymer and a photoelectric conversion element using the same.
- Organic thin-film solar cells that can be produced by a coating method using a polymer material that is soluble in a solvent are manufactured at a lower cost than inorganic solar cells such as polycrystalline silicon, amorphous silicon, and compound semiconductors that are currently mainstream solar cells. It is said that it can be done and is attracting a lot of attention.
- Organic thin-film solar cells mainly have a bulk heterojunction structure in which a conjugated polymer and an electron-accepting material are mixed as a photoelectric conversion active layer.
- photoelectric conversion activity obtained by mixing poly (3-hexylthiophene) as a conjugated polymer and [6,6] -phenyl C 61 butyric acid methyl ester (PCBM) as a fullerene derivative as an electron accepting material.
- PCBM poly (6-hexylthiophene)
- PCBM -phenyl C 61 butyric acid methyl ester
- There is an organic thin film solar cell having a layer Non-Patent Document 1.
- a bulk heterojunction structure In a bulk heterojunction structure, light incident from a transparent electrode is absorbed by a conjugated polymer and / or an electron-accepting material to generate excitons in which electrons and holes are combined. The generated exciton moves to the heterojunction interface where the conjugated polymer and the electron-accepting material are adjacent to each other, and charges are separated into holes and electrons. Holes and electrons are transported through the conjugated polymer phase and the electron-accepting material phase, respectively, and are extracted from the electrode.
- the poly (3-hexylthiophene) has a light absorption in the visible light region, but may be abbreviated as a narrow band gap polymer (hereinafter referred to as a narrow band gap polymer) having an absorption up to a further longer wavelength region (near infrared region). )
- a narrow band gap polymer having an absorption up to a further longer wavelength region (near infrared region).
- Non-Patent Documents 2 and 3 have already been proposed (Non-Patent Documents 2 and 3).
- the narrow bandgap polymers since light absorption in the visible light region is reduced, without using very expensive C 70 fullerene derivative having a light absorption in the visible light region as an electron-accepting material has a problem that not .
- Non-Patent Document 4 a bulk heterojunction type photoelectric conversion active layer in which an electron-accepting material is mixed with a conjugated polymer blend of poly (3-hexylthiophene) and a narrow band gap polymer has also been proposed.
- Non-patent Document 5 a diblock copolymer of 3-hexylthiophene and 3- (2-ethylhexylthiophene)
- Non-patent Document 6 a diblock copolymer of 3-hexylthiophene and 3-phenoxymethylthiophene
- Non-patent Documents 7 and 8 A diblock copolymer of 3-butylthiophene and 3-octylthiophene
- Non-patent Document 9 Organic thin-film solar cells using copolymers and fullerene derivatives as electron-accepting materials have been reported.
- an organic thin film solar cell element using a conjugated block copolymer a diblock copolymer of 3-hexylthiophene and 3- (2-ethylhexylthiophene)
- the present invention has been made to solve such problems, and is a conjugated block copolymer capable of increasing the amount of light absorption of the photoelectric conversion active layer and controlling the morphology and exhibiting excellent photoelectric conversion efficiency. It is another object of the present invention to provide a photoelectric conversion element comprising a composition containing an electron-accepting material using such a conjugated block polymer.
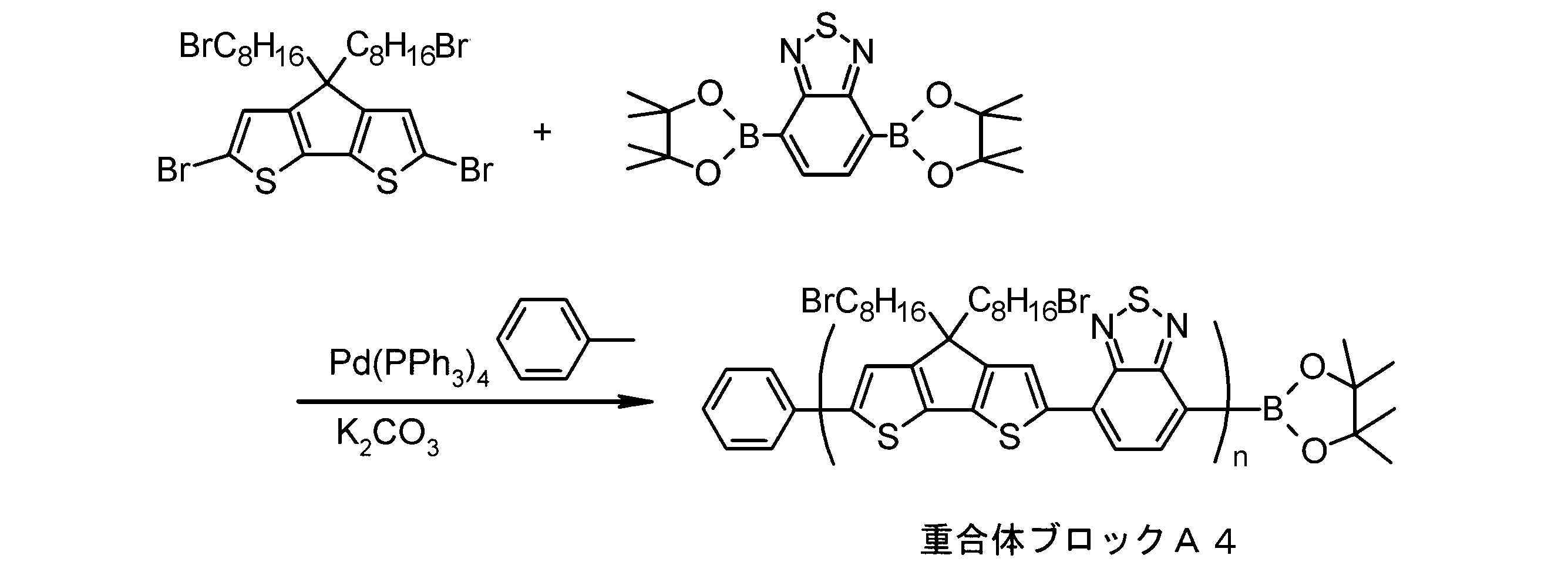
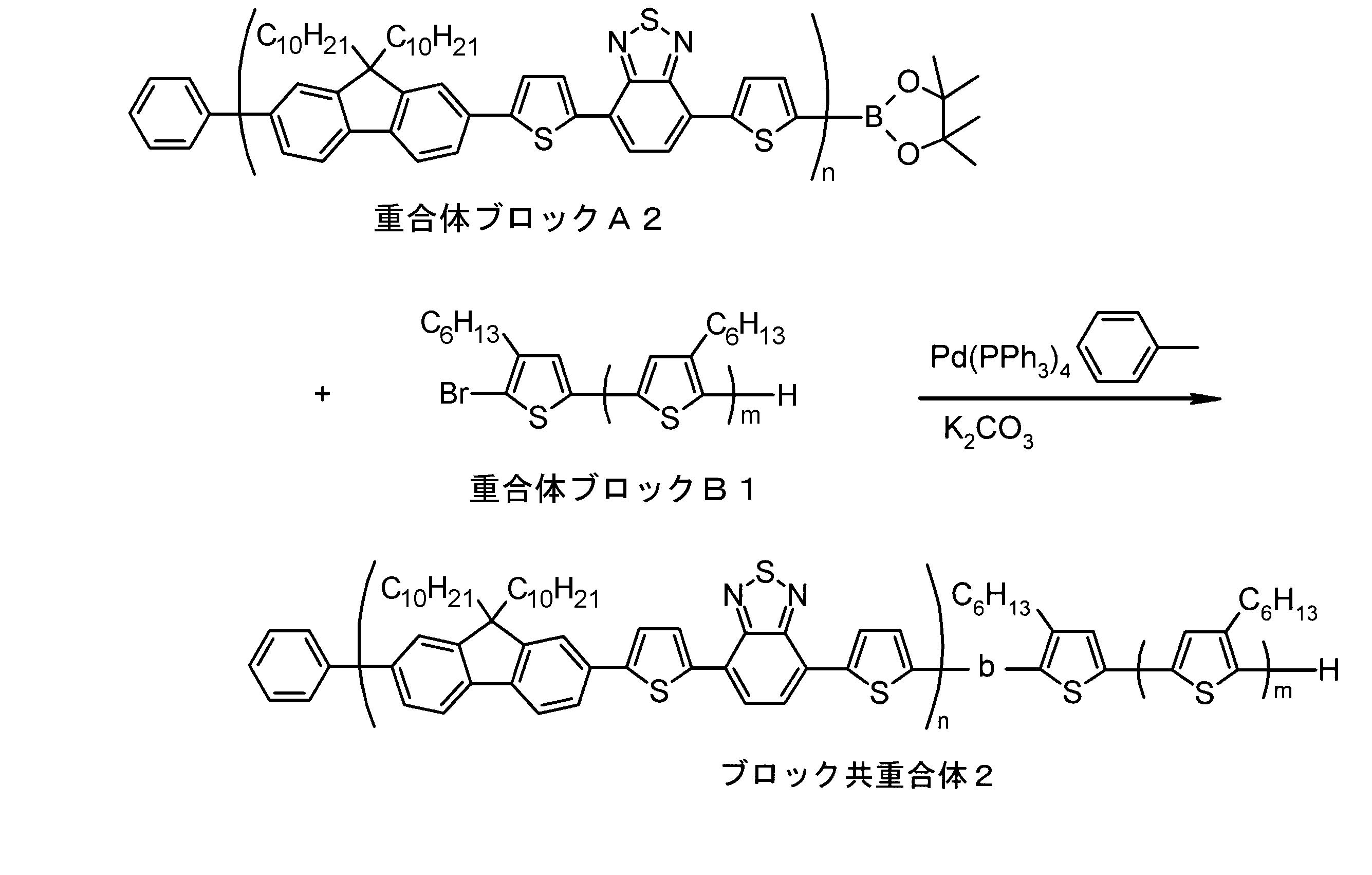
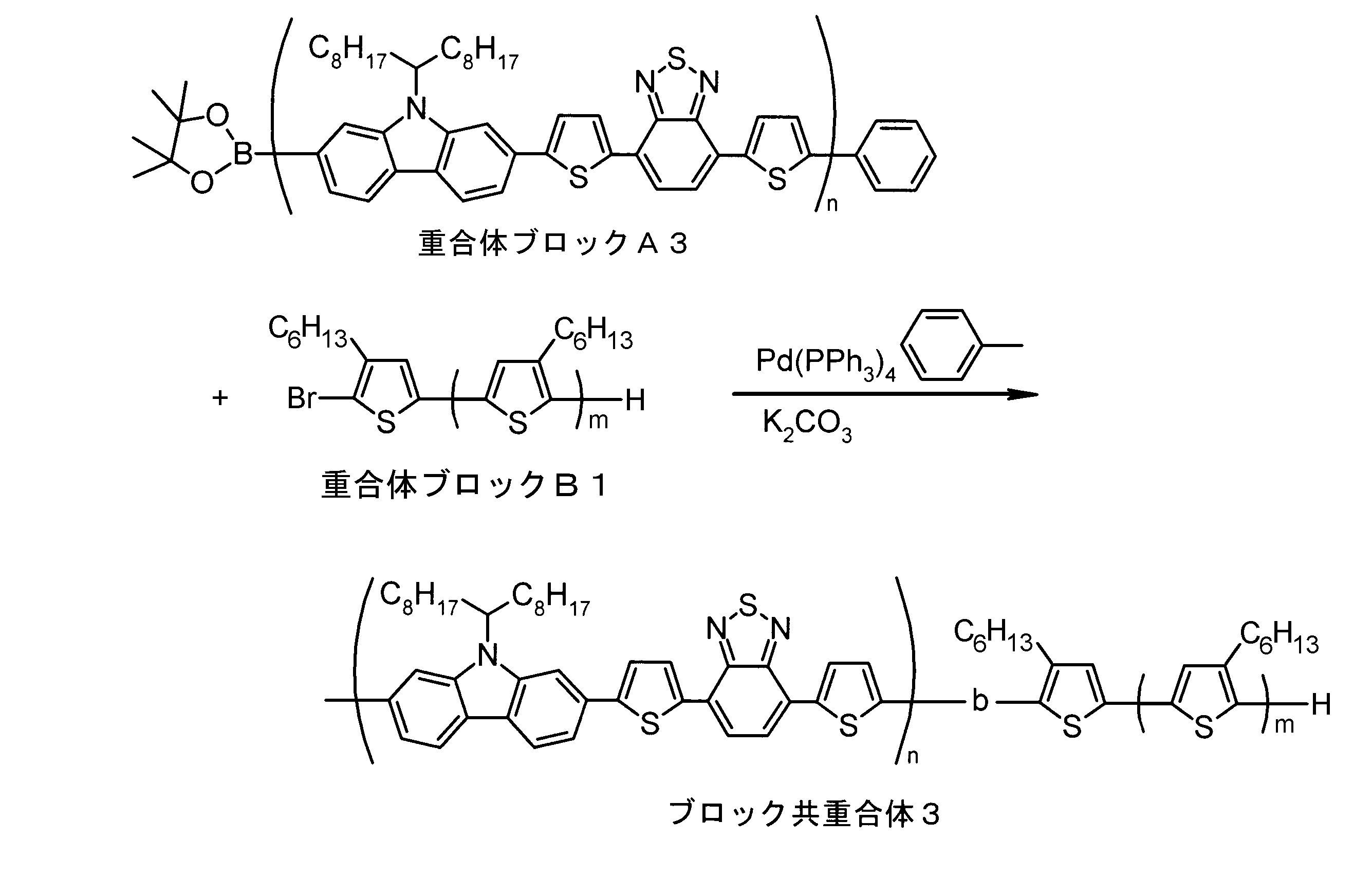
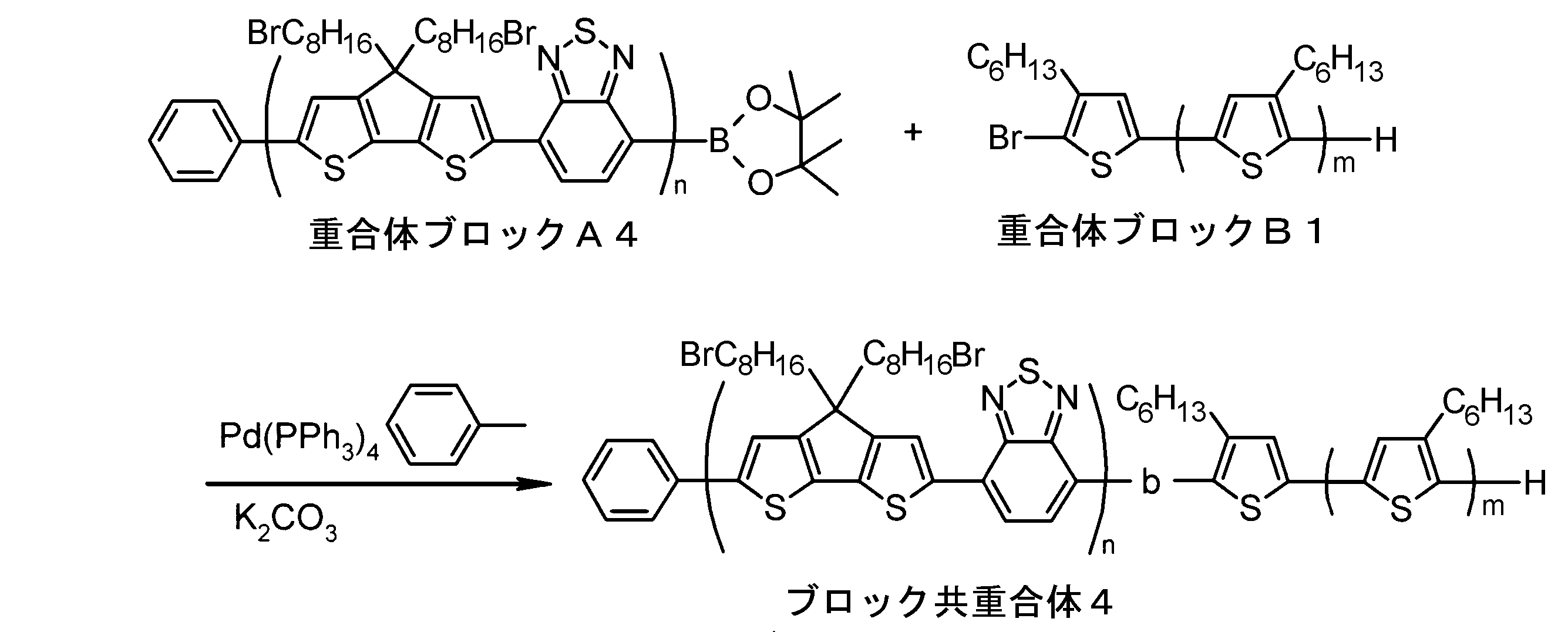
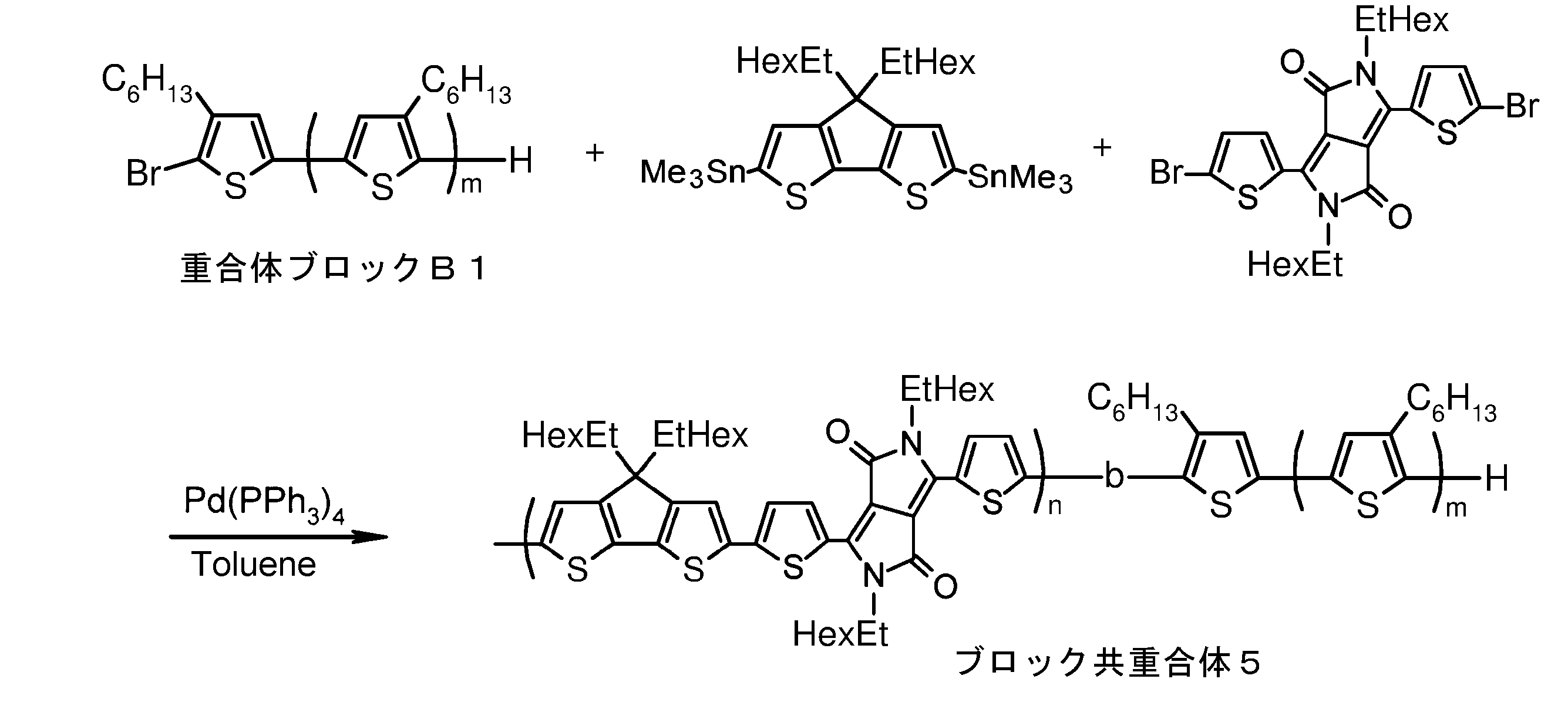
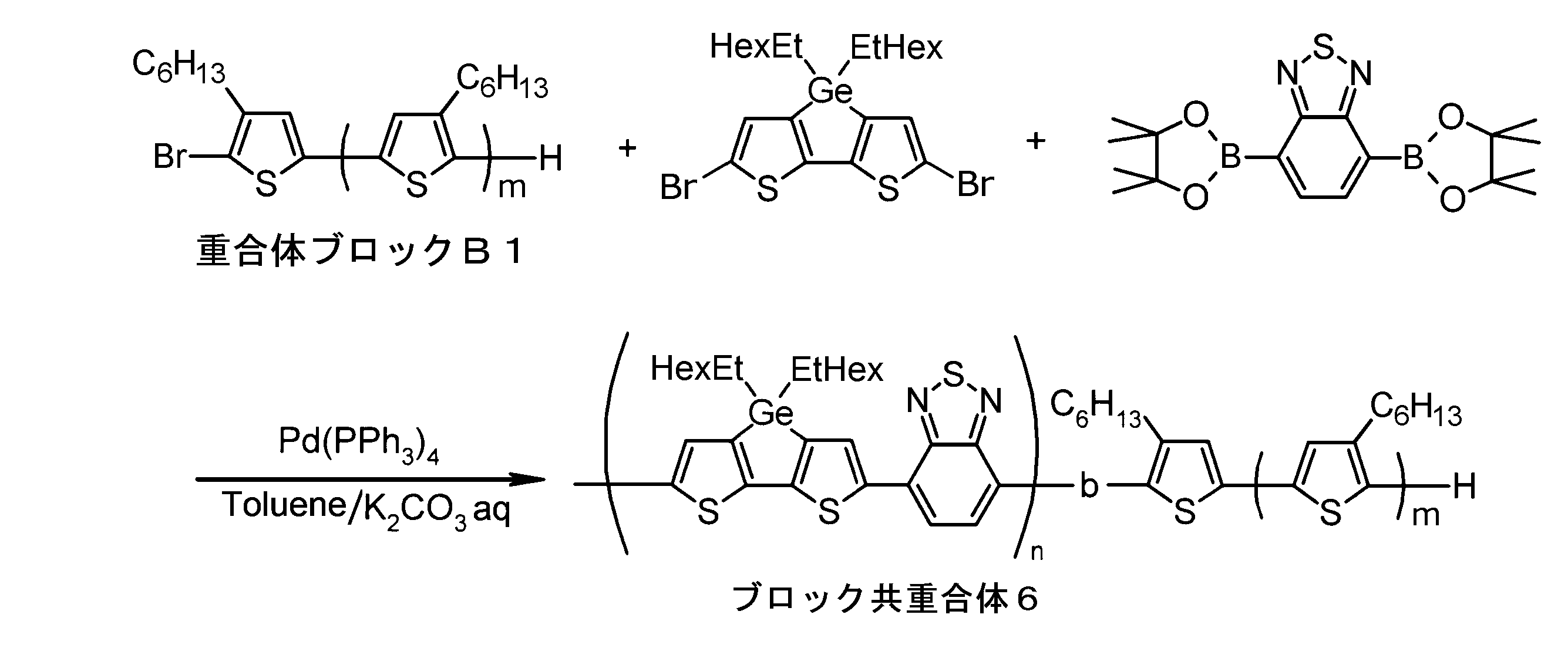
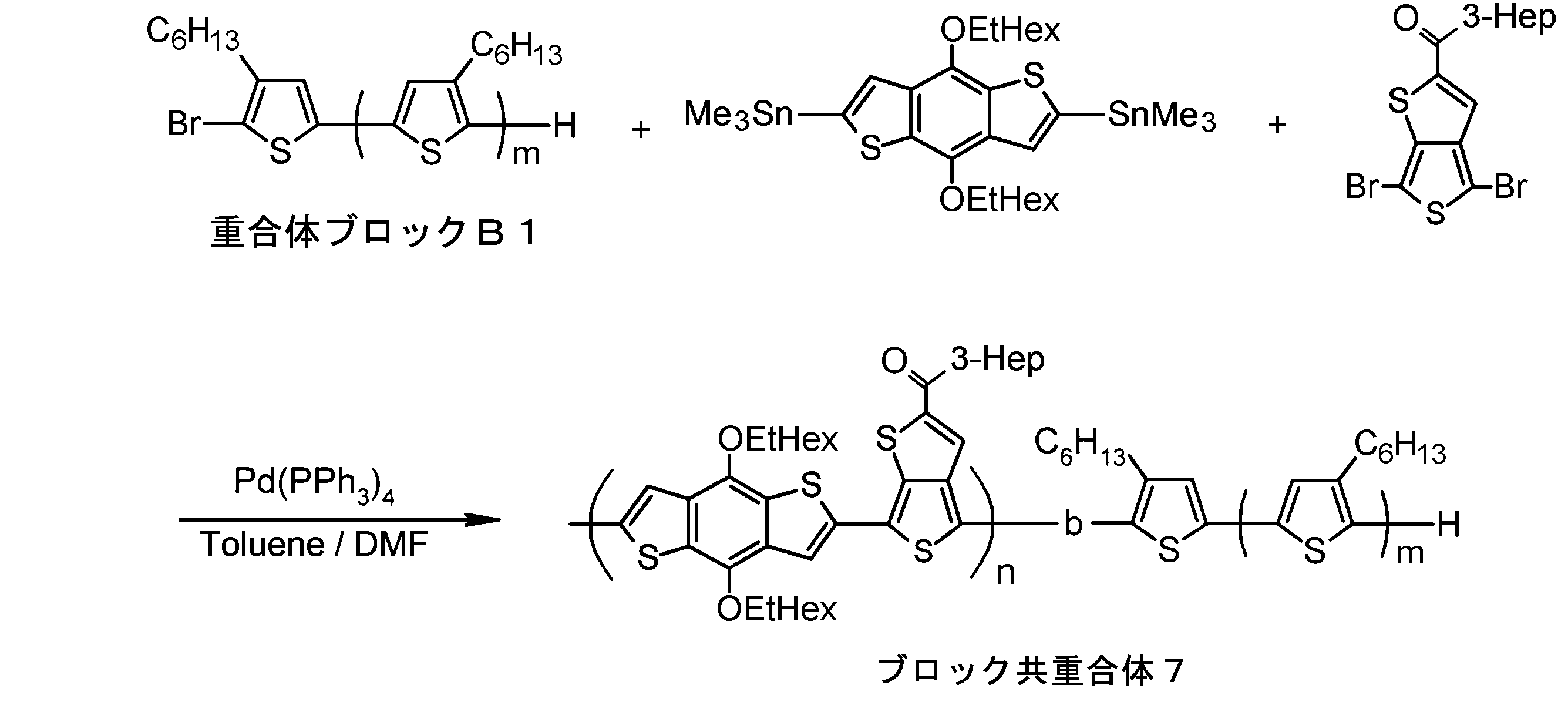
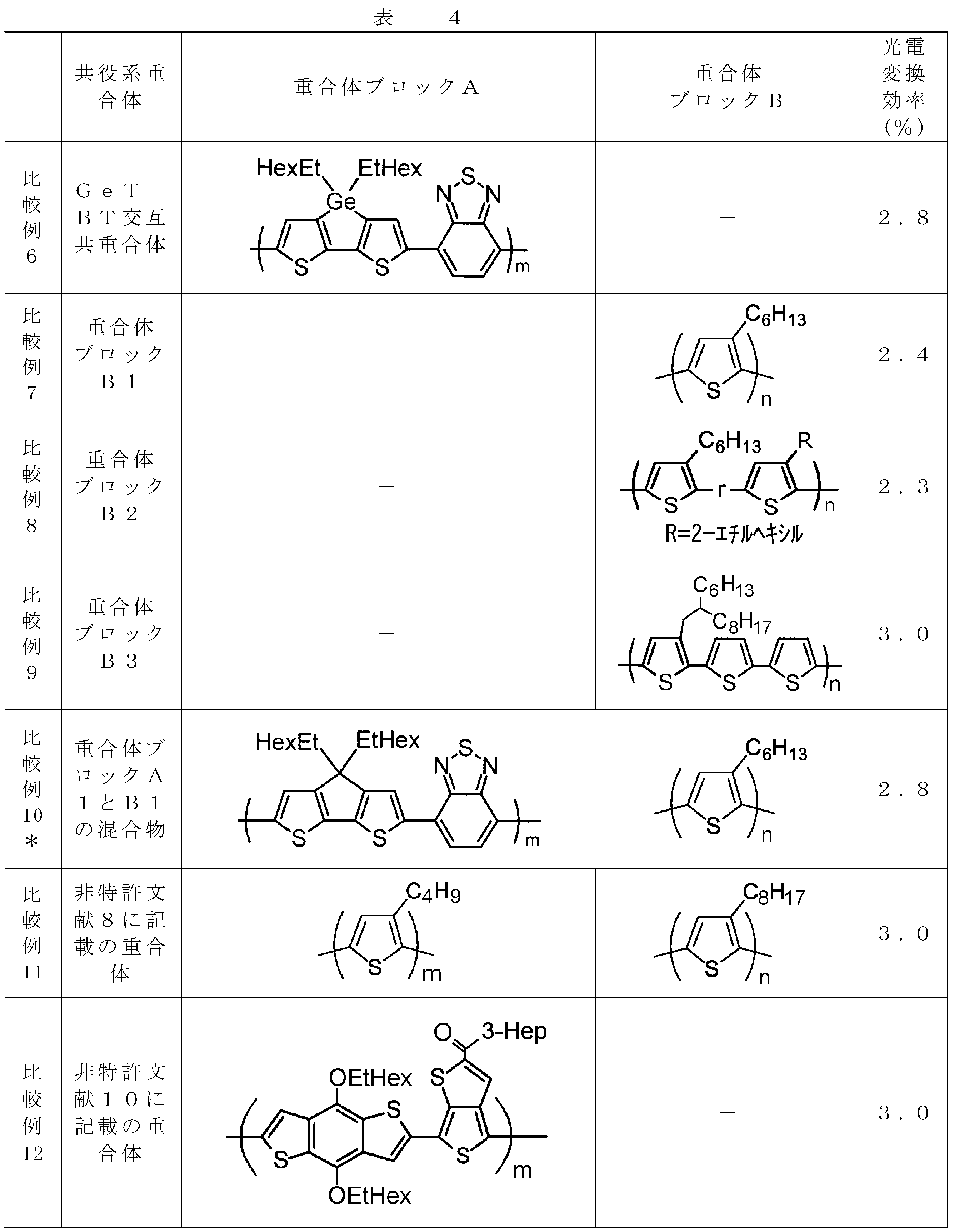
- the invention according to claim 1, which has been made to achieve the above object, includes a condensed ⁇ -conjugated skeleton containing at least one thiophene ring as part of a chemical structure, fluorene, carbazole, dibenzosilole, and A polymer block A comprising a monomer unit having at least one heteroaryl skeleton selected from the group consisting of dibenzogermole, and A ⁇ -electron conjugated block copolymer comprising a polymer block B having a thiophene-2,5-diyl group having a substituent at least at the 3-position as a monomer unit.
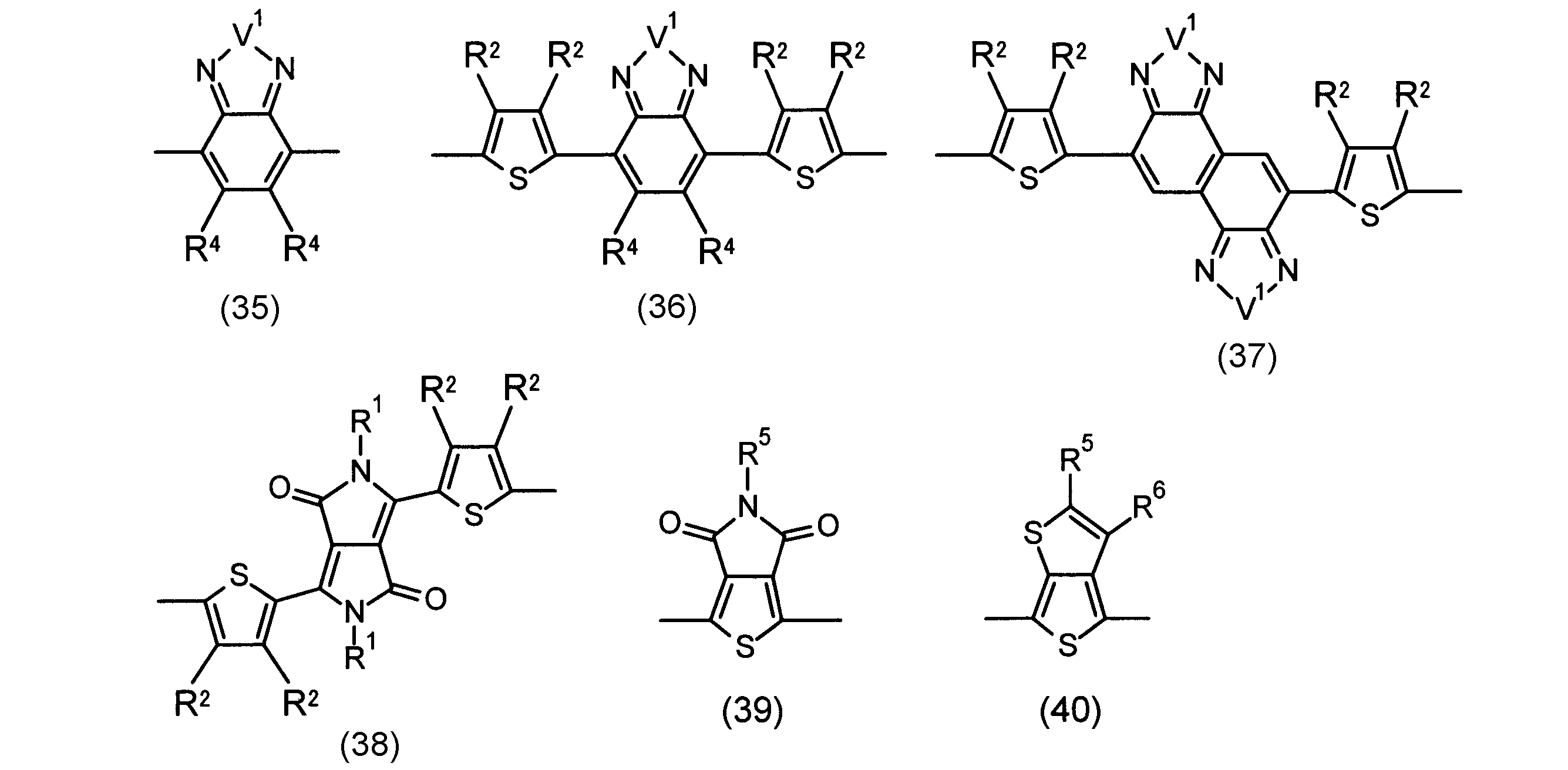
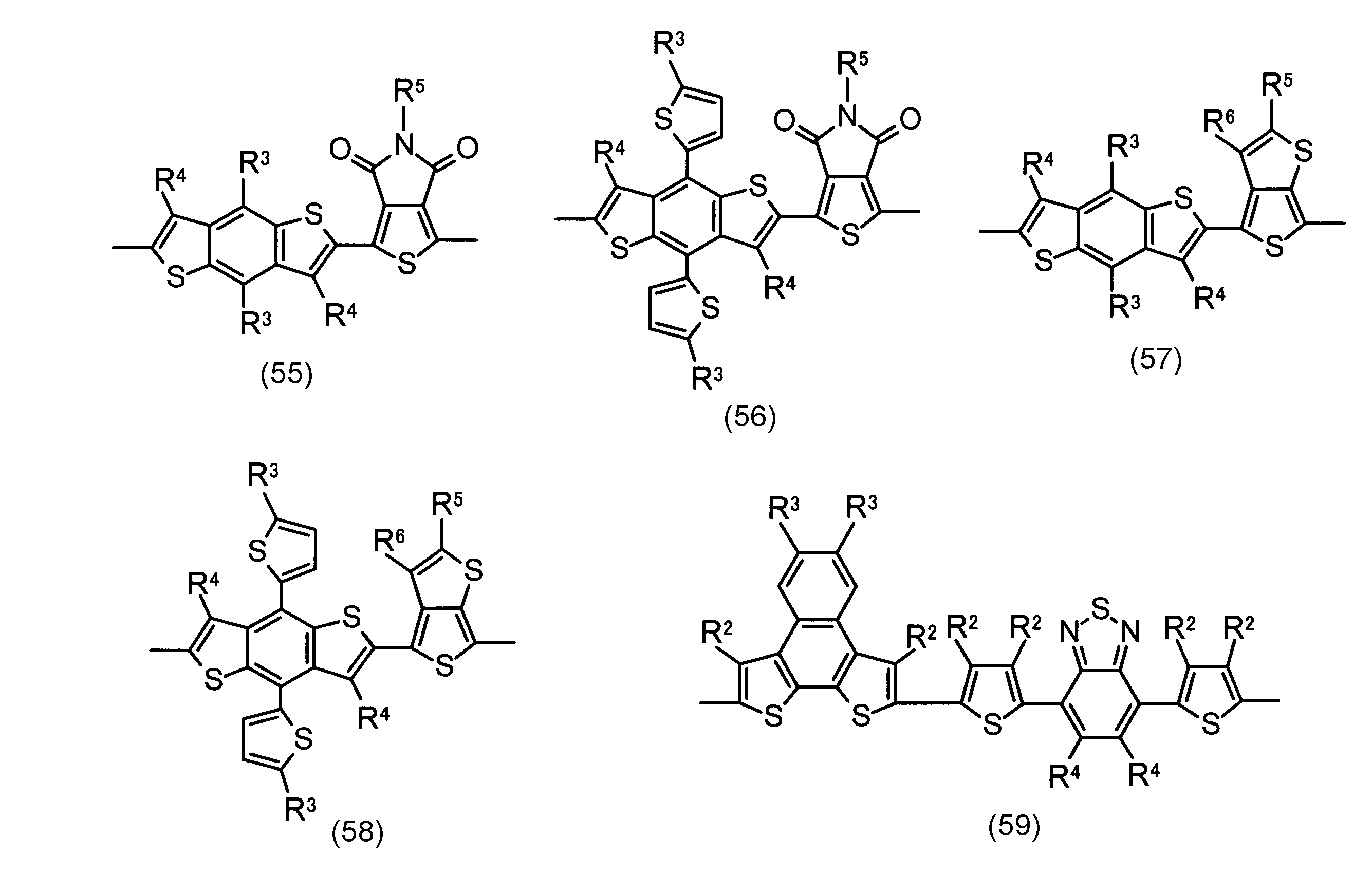
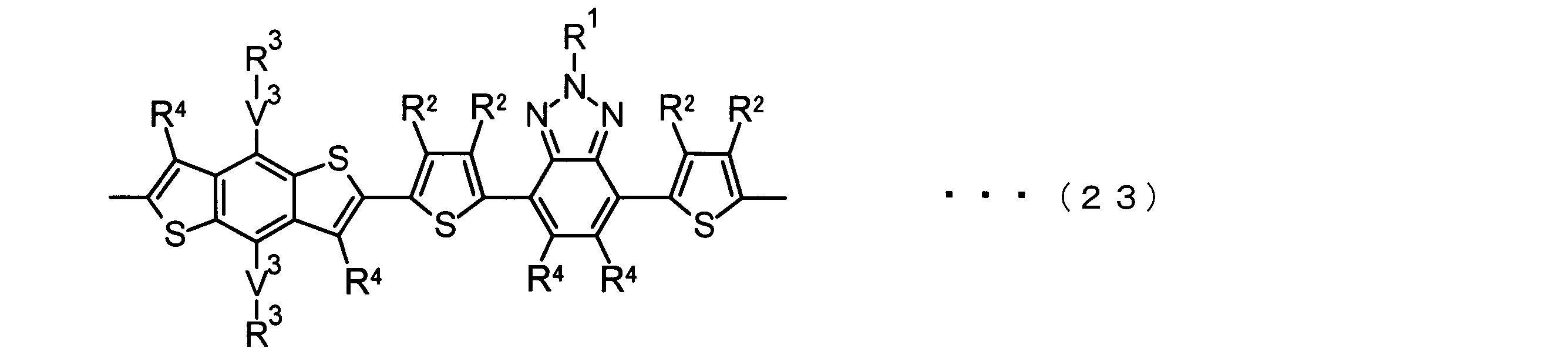
- the invention according to claim 2 is the ⁇ -electron conjugated block copolymer according to claim 1, wherein the polymer block A includes a monomer unit -ab-, Formulas (1) to (6) Having any group represented by -B- represents the following formulas (7) to (17) It is a monomer unit which has either group represented by these.
- V 1 is nitrogen (—NR 1 —), oxygen (—O—) or sulfur (—S—)
- V 2 is carbon (—CR 1 2 —), Nitrogen (—NR 1 —), Silicon (—SiR 1 2 —) or Germanium (—GeR 1 2 —)
- V 3 is an aryl group or heteroaryl group represented by — (Ar) n —
- V 4 is nitrogen (—NR 1 —), oxygen (—O—) or —CR 2 ⁇ CR 2 —
- V 5 is oxygen (O) or sulfur (S).
- R 1 is independently an alkyl group having 1 to 18 carbon atoms which may be independently substituted;
- R 2 is independently a hydrogen atom or an optionally substituted alkyl group having 1 to 18 carbon atoms; 3 is an alkyl group or alkoxy group having 1 to 18 carbon atoms which may be independently substituted, and
- R 4 is each independently a hydrogen atom, halogen atom or optionally substituted alkyl having 1 to 18 carbon atoms.
- R 5 is an optionally substituted alkyl group having 1 to 18 carbon atoms, an aryl group, an alkylcarbonyl group or an alkyloxycarbonyl group, and
- R 6 is a hydrogen atom or a halogen atom.
- m represents an integer of 1 to 3
- n represents an integer of 0 to 3.
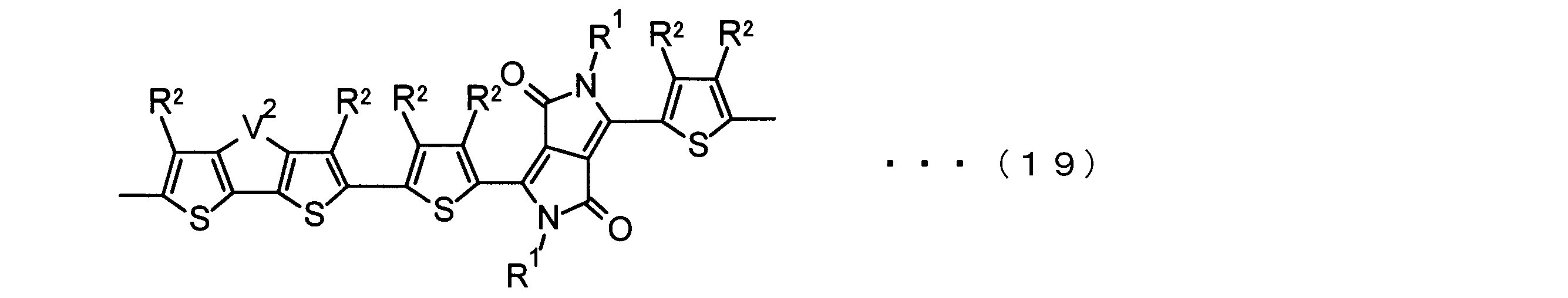
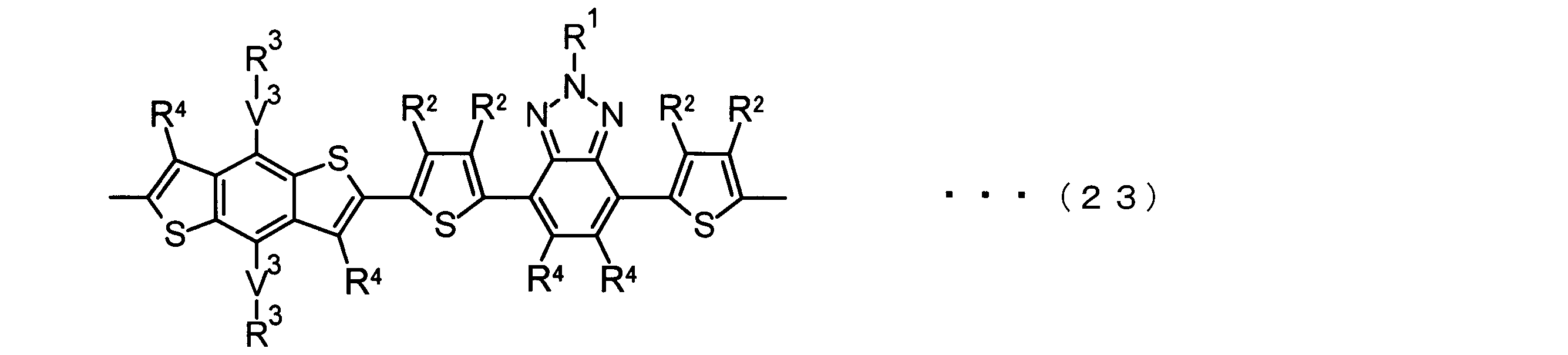
- the invention according to claim 3 is the ⁇ -electron conjugated block copolymer according to claim 2, wherein the monomer unit -ab- is represented by the following formulas (18) to (28): Any monomer unit selected from In the above formulas (18) to (28), V 2 is carbon (—CR 1 2 —), nitrogen (—NR 1 —), silicon (—SiR 1 2 —) or germanium (—GeR 1 2 —). , V 3 is an aryl group or heteroaryl group represented by — (Ar) n —. R 1 , R 2 , R 3 , R 4 , R 5 and R 6 are as defined above. n represents an integer of 0 to 3.
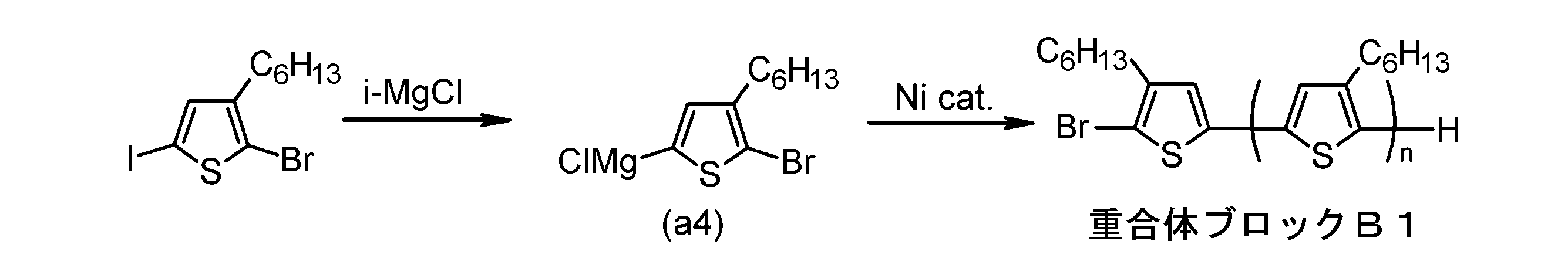
- the invention according to claim 4 is the ⁇ electron conjugated block copolymer according to claim 1, wherein the polymer block B has a thiophene-2,5-diyl group having a substituent at least at the 3-position. It is a polymer block composed of monomer units in which 2 to 5 thiophene-2,5-diyl groups are linked.
- the invention according to claim 5 is the ⁇ -electron conjugated block copolymer according to claim 1, wherein the polymer block B has a thiophene-2,5-diyl group having a substituent at least at the 3-position. It is a random copolymer block having a plurality of types.
- the invention according to claim 6 is the ⁇ -electron conjugated block copolymer according to any one of claims 1 to 5, wherein the number average molecular weight is 1,000 to 200,000 g / mol.
- the invention according to claim 7 of the present invention which has been made to achieve the above object, includes an electron-accepting material and a ⁇ -electron conjugate according to any one of claims 1 to 6. It is a composition containing a system block copolymer.
- the invention according to claim 8 is a photoelectric conversion element having a layer made of the composition according to claim 7.
- the invention according to claim 9 is the photoelectric conversion element according to claim 8, wherein the electron-accepting material is fullerene or a derivative thereof.
- the ⁇ -electron conjugated block copolymer of the present invention When the ⁇ -electron conjugated block copolymer of the present invention is used in a photoelectric conversion element together with an electron-accepting material, the current value increases due to the absorption expansion to a long wavelength region, and the resistance decreases, and the performance is greatly improved. To do.
- polymer block A and polymer block B are linked, and the main chain skeleton of the monomer unit is a ⁇ -electron conjugated divalent group.
- the monomer unit of the polymer block A has a heteroaryl skeleton selected from the group consisting of a condensed ⁇ -conjugated skeleton containing at least one thiophene ring in a chemical structure, fluorene, carbazole, dibenzosilole, and dibenzogermole. Have at least one.
- examples of the condensed ⁇ -conjugated skeleton including at least one thiophene ring as a part of the chemical structure include cyclopentadithiophene, dithienopyrrole, dithienosilole, dithienogermol, benzodithiophene, naphthodithiophene, and the like.
- the monomer unit constituting the polymer block A includes a monomer unit -ab-, where -a- is any group represented by the formulas (1) to (6) (donor type).
- -B- is a monomer unit having any of the groups represented by the above formulas (7) to (17) (a group consisting of a unit having a ring serving as an acceptor). Is preferred.
- V 3 is a monocyclic or polycyclic aryl group or heteroaryl group (n is an integer of 0 to 3) represented by — (Ar) n —, and is a monomer unit. Part of the main chain skeleton. V 3 is particularly preferably a thiophene ring.
- -A- is more preferably one having any of the groups represented by the above formulas (1), (2), (3), and -b- includes the above formulas (7), ( 11) and a monomer unit having any group represented by (15) to (17) is more preferable.
- the monomer unit constituting -ab- include monomer units represented by the above formulas (18) to (28). That is, the monomer unit represented by the formula (1)-(7)-is the formula (18), and the monomer unit represented by the formula (1)-(15)-is the formula (19). The monomer unit represented by the formula (1)-(16)-is represented by the formula (20), and the monomer unit represented by the formula (1)-(17)-is represented by the formula (21). The monomer unit represented by the formula (2)-(7)-is represented by the formula (22), and the monomer unit represented by the formula (3)-(7)-is represented by the formula (23).
- the monomer unit in the present invention has a structure in which a plurality of heteroaryls composed of thiophene, fluorene, carbazole, dibenzosilol, or dibenzogermole are linked (for example, a single monomer) as long as the polymer has a plurality of constant repeating structures.
- the body unit -ab- one unit. That is, the completely alternating copolymer block of the monomer unit -a- and the monomer unit -b- is different from the homopolymer block of the monomer unit -ab- as long as the substituent is the same. Shall be deemed.
- the total number of carbon atoms constituting only the ring structure excluding the substituent R n in one kind of monomer unit including the embodiment of the monomer unit -ab- Is preferably 6-30.
- the ⁇ -electron conjugated block copolymer of the present invention desirably has a light absorption as long as possible in order to increase the amount of current.
- the polymer block B of the block copolymer has a thiophene-2,5-diyl group having a substituent at least at the 3-position as a monomer unit, and this usually has absorption in the visible light region.
- the polymer block A is composed of monomer units having absorption at least on the longer wavelength side than the polymer block B.
- the polymer block A When the polymer block A is a structure in which the polymer has high planarity and is difficult to form a twisted structure, it absorbs light having a long wavelength and increases the photoelectric conversion efficiency. From this point of view, it may be composed of a monomer unit having at least one heteroaryl skeleton selected from the group consisting of a condensed ⁇ -conjugated skeleton containing at least one thiophene ring as a part of the chemical structure, carbazole, and dibenzogermol. Preferably, it is more preferably composed of a monomer unit having at least one condensed ⁇ -conjugated skeleton containing at least one thiophene ring as part of the chemical structure.
- -a- represented by the above formulas (1) to (6) are not particularly limited, and examples thereof include groups represented by the following formulas (29) to (34). It is done. (Wherein V 2 , R 2 , R 3 and R 4 are as defined above, and V 6 is independently a hydrogen atom or R 3 )
- -b- represented by the above formulas (7) to (17) are not particularly limited, and examples thereof include groups represented by the following formulas (35) to (40). It is done. (Wherein V 1 , R 2 , R 4 , R 5 and R 6 are as defined above.)
- -ab- represented by the above formulas (18) to (28) are not particularly limited. For example, they are represented by the following formulas (41) to (59). Group. (Wherein R 1 to R 6 are as defined above.)
- the polymer block A may have a substituent represented by R 1 to R 6 .
- R 1 is independently an alkyl group having 1 to 18 carbon atoms which may be independently substituted;
- R 2 is independently a hydrogen atom or an optionally substituted alkyl group having 1 to 18 carbon atoms;
- 3 is an alkyl group or alkoxy group having 1 to 18 carbon atoms which may be independently substituted, and
- R 4 is each independently a hydrogen atom, halogen atom or optionally substituted alkyl having 1 to 18 carbon atoms.
- R 5 is an optionally substituted alkyl group having 1 to 18 carbon atoms, an aryl group, an alkylcarbonyl group or an alkyloxycarbonyl group, and R 6 is a hydrogen atom or a halogen atom.
- R 1 to R 5 may be further substituted with an alkyl group, an alkoxy group, a halogen atom, a hydroxyl group, an amino group, a thiol group, a silyl group, an ester group, an aryl group, or a heteroaryl group.
- the alkyl group or alkoxy group may be linear, branched or alicyclic.
- the thiophene-2,5-diyl group which is a monomer unit of the polymer block B, has an alkyl group having 1 to 18 carbon atoms, an alkoxy group, or an aryl group as a substituent at least at the 3-position.
- the substituent may be substituted with an alkyl group, an alkoxy group, a halogen atom, a hydroxyl group, an amino group, a thiol group, a silyl group, an ester group, an aryl group, or a heteroaryl group.
- the alkyl group or alkoxy group that is a substituent of the polymer block B may be linear, branched or alicyclic.
- the polymer block B is a monomer unit in which a plurality of thiophene-2,5-diyl groups are linked, at least one of the thiophene-2,5-diyl groups is a substituent at the 3-position.
- a group in which 2 to 5 thiophene-2,5-diyl groups are linked is preferable. Examples thereof include bithiophene, terthiophene and the like containing at least one thiophene-2,5-diyl group having a substituent at the 3-position.
- the polymer block B may be a random copolymer having a plurality of types of thiophene-2,5-diyl groups having a substituent at least at the 3-position. From the viewpoint of photoelectric conversion characteristics, the polymer block B is preferably a random copolymer.
- alkyl group having 1 to 18 carbon atoms examples include methyl group, ethyl group, n-propyl group, isopropyl group, n-butyl group, isobutyl group, sec-butyl group, tert-butyl group, n-pentyl group, Isopentyl, neopentyl, tert-pentyl, n-hexyl, isohexyl, 2-ethylhexyl, n-heptyl, n-octyl, n-nonyl, n-decyl, cyclopropyl, cyclopentyl , A cyclohexyl group, a cyclooctyl group, and the like.
- alkoxy group having 1 to 18 carbon atoms examples include methoxy group, ethoxy group, n-propyloxy group, isopropyloxy group, n-butoxy group, n-hexyl group, cyclohexyloxy group, n-octyloxy group, n -Decyloxy group, n-dodecyloxy group and the like.
- Examples of the alkoxy group which may be substituted on the substituent of the polymer block A and the polymer block B include, for example, a methoxy group, an ethoxy group, an n-propyloxy group, an isopropyloxy group, an n-butoxy group, and an n-hexyl group. And an alkoxy group such as a cyclohexyloxy group, an n-octyloxy group, an n-decyloxy group, and an n-dodecyloxy group.
- Examples of the halogen atom include a fluorine atom, a chlorine atom, a bromine atom, and an iodine atom.
- Examples of the alkyl group substituted with a halogen atom include an ⁇ -bromoalkyl group and a perfluoroalkyl group.
- Examples of the amino group include primary or secondary amino groups such as a dimethylamino group, a diphenylamino group, a methylphenylamino group, a methylamino group, and an ethylamino group.
- Examples of the thiol group include a mercapto group and an alkylthio group.
- Examples of the silyl group include a trimethylsilyl group, a triethylsilyl group, a tripropylsilyl group, a triisopropylsilyl group, a dimethylisopropylsilyl group, and a dimethyl tert-butylsilyl group.
- ester group examples include a methoxycarbonyl group, an ethoxycarbonyl group, a propoxycarbonyl group, an isopropoxycarbonyl group, a tert-butoxycarbonyl group, a phenoxycarbonyl group, an acetyloxy group, and a benzoyloxy group.
- aryl group examples include a phenyl group, a 1-naphthyl group, a 2-naphthyl group, a 1-anthracenyl group, a 2-anthracenyl group, a 9-anthracenyl group, and the like. These further include substituents such as an alkyl group and an alkoxy group. You may have.
- heteroaryl group examples include a pyridyl group, a thienyl group, a furyl group, and a pyrrolyl group.
- the monomer unit contained in the polymer block A or the polymer block B only the same monomer including the structure of the substituent may be used, or another unit amount as long as the effect of the present invention is not impaired.
- the body may be included.
- connection structure of the polymer block A and the polymer block B contained in the ⁇ -electron conjugated block copolymer of the present invention is not particularly limited.
- Examples of the continuous connection structure include an AB type diblock copolymer. Polymer or BA type diblock copolymer, ABA type triblock copolymer or BAB type triblock copolymer, ABAB type tetrablock copolymer Alternatively, a B-A-B-A type tetrablock copolymer, an A-B-A-B-A, or a B-A-B-A-B type penta-block copolymer may be used. These block copolymers may be used alone or in combination of two or more.
- the weight ratio of the polymer block A and the polymer block B is preferably 99: 1 to 1:99, and more preferably 95: 5 to 5:95. If the weight ratio of the polymer block A or B is too small, the photoelectric conversion efficiency may not be sufficiently increased. Therefore, it is more preferably 90:10 to 10:90, and further preferably 70:30 to 30:70. .
- the number average molecular weight of the ⁇ -electron conjugated block copolymer is preferably 1,000 to 200,000 g / mol, and preferably 5,000 to 100,000 g / mol from the viewpoints of processability, crystallinity, solubility, photoelectric conversion characteristics, and the like.
- the range is more preferably in the range of 10,000 to 100,000 g / mol, and particularly preferably in the range of 20,000 to 100,000 g / mol. If the number average molecular weight is too high, the solubility is lowered and the workability of the thin film is lowered. If the number average molecular weight is too low, the crystallinity, film stability, photoelectric conversion characteristics and the like are lowered.
- the number average molecular weight means a molecular weight in terms of polystyrene by gel permeation chromatography (hereinafter sometimes referred to as GPC).
- GPC gel permeation chromatography
- the number average molecular weight of the ⁇ electron conjugated block copolymer of the present invention can be determined by ordinary GPC using a solvent such as tetrahydrofuran (THF), chloroform, dimethylformamide (DMF) and the like.
- THF tetrahydrofuran
- DMF dimethylformamide
- some of the ⁇ -electron conjugated block copolymers of the present invention have low solubility around room temperature. In such cases, the number average molecular weight is determined by high-temperature GPC chromatography using dichlorobenzene or trichlorobenzene as a solvent. May be.
- the ⁇ electron conjugated block copolymer may contain an arbitrary polymer block C different from the polymer block A and the polymer block B.
- Examples of such other polymer block C include, for example, a single block of a single unit of -a-unit, a single block of -b-unit, or other component containing a monocyclic or condensed (hetero) arylene group. Examples of blocks are body units.
- Examples of the structure in the case where the ⁇ -electron conjugated block copolymer contains the polymer block C can be exemplified below.
- Examples of the structure in which the polymer block A and the polymer block B are continuously connected include, for example, an ABC type triblock copolymer, a BAC type triblock copolymer, and a CAB type.
- Type triblock copolymer CBA type triblock copolymer, ABCA type tetrablock copolymer, BABC type tetrablock copolymer, CA -BA type tetrablock copolymer, CBABA type tetrablock copolymer, CABC type tetrablock copolymer, CBAC type tetrablock copolymer Polymer, CABAC type pentablock copolymer, CBABC type pentablock copolymer, ABABC type pentablock copolymer , BBABAC type pentablock copolymer, CBABABA type pentablock Polymers, like C-A-B-A-B-type pentablock copolymer.
- the structure in which the polymer block A and the polymer block B are connected discontinuously refers to a structure in which the polymer block C is inserted between the polymer block A and the polymer block B.
- Polymer block A, polymer block B and polymer block C may be further linked to the above triblock copolymer, tetrablock copolymer and pentablock copolymer.
- the proportion of the polymer block C in the block copolymer is preferably 40% by mass or less.
- the polymer block C is a non- ⁇ electron conjugated polymer and does not contribute to photoelectric conversion, it is more preferably 30% by mass or less, and still more preferably 20% by mass or less.
- Examples of the analysis method of the polymer structure of the ⁇ -electron conjugated block copolymer include measurement by solution NMR, infrared spectroscopy, and Raman spectroscopy. Analysis by secondary ion mass spectrometry or decomposition GCMS is also possible.
- linking method a method in which polymer block A and polymer block B are synthesized separately and then linked together. There is).
- the second method there is a method of polymerizing the polymer block B or the polymer block A in the presence of the polymer block A or the polymer block B, respectively (hereinafter sometimes referred to as “macroinitiator method”). is there.
- macroinitiator method an optimum method can be used depending on the ⁇ -electron conjugated block copolymer to be synthesized.
- the compound A-M p having a polymer block A in advance prepared, previously prepared polymer block It can be produced by performing a coupling reaction with a compound BX having B in the presence of a catalyst.
- X and Mp are terminal functional groups of the polymer block.
- a and B represent a polymer block
- X is a halogen atom
- M p is a boronic acid, boronic acid ester, —MgX, —ZnX, —SiX 3 or —SnRa 3 (where Ra is the number of carbon atoms) 1 to 4 linear alkyl groups).
- a method for producing the polymer block A will be described.
- the following reaction formula in accordance with (III), in the presence of a catalyst by reacting the M q1 -Y-M q1 and M q2 -Z-M q2 is a monomer, a compound A containing a polymer block A by the coupling reaction it can be prepared -X or compound a-M p.
- Y and Z each represent a heteroaryl skeleton constituting at least a part of the monomer unit of the polymer block A.
- M q1 and M q2 are functional groups involved in the coupling reaction.
- M q1 and M q2 are not the same and are each independently a halogen atom, or a boronic acid, boronic acid ester, —MgX, —ZnX, —SiX 3 or —SnRa 3 (where Ra is carbon number 1) represents a 1-4 straight-chain alkyl group), M p represents boronic acid, boronate ester, -MgX, -ZnX, the -SiX 3 or -SnRa 3.
- a and X are as defined above.
- M q1 is a halogen atom
- M q2 is boronic acid, boronic acid ester, —MgX, —ZnX, —SiX 3 or —SnRa 3
- M q1 is boronic acid, Boronate ester, —MgX, —ZnX, —SiX 3 or —SnRa 3 is obtained.
- Polymer block A compound M q1 -Y-M q1 and compound M q2 -Z-M q2 the compound represented by the formula Ar-M r (hereinafter sometimes abbreviated as terminal blocking agent) and using It can also be produced by carrying out a coupling reaction.
- Ar is an aryl group
- M r represents M p or X
- M p and X are as defined above.
- Compound A-X in this way, it becomes easy to introduce functional groups for coupling only one of the end groups of the polymer block A as Compound A-M p.
- Compound A-X even functional group X or M p with the A-M p derived from M q1 -Y-M q1 and M q2 -Z-M q2 monomeric polymer block A it may, or may be a functional group derived from a different linker compound M r -Q-M r is the monomer of the polymer block a.
- Q is an arylene group
- M r represents M p or X
- M p and X are as defined above.
- Q is preferably a monocyclic divalent arylene from the viewpoint of reactivity and availability, and more preferably divalent thiophene or benzene which may have a substituent.
- Specific examples include 2,5-dibromothiophene, 2,5-bis (trimethyltin) thiophene, 2,5-thiophene diboronic acid, 2,5-bis (3,3,4,4-tetramethyl -2,5,1-dioxaborolan-1-yl) thiophene, p-dibromobenzene, p-bis (trimethyltin) benzene, p-benzenediboronic acid, p-bis (3,3,4,4-tetramethyl -2,5,1-dioxaborolan-1-yl) benzene and the like.
- M q1 -Y- M q1 and M q2 end by excessively charged either one of the -Z-M q2 is a monomer It is possible to preferentially introduce either X or M p as a functional group. However, the M q1 for excessively put too polymerization either one of -Y-M q1 and M q2 -Z-M q2 are not proceed, M q1 -Y-M q1 and M q2 -Z-M q2
- the molar ratio is preferably in the range of 0.5 to 1.5, more preferably 0.7 to 1.3.
- a monomer M q1 -Y-M q1 and M q2 polymerization early, middle or linker compound late in the -Z-M q2 M r -Q- M r A linker compound can be preferentially introduced at the end of the polymer block A.
- the input amount of the linker compound is 1.5 times equivalent or more, preferably 2 times equivalent or more, more preferably 5 times equivalent or more of the terminal functional group amount calculated from the number average molecular weight (Mn) of the polymer block A during polymerization. It is.
- the polymer block B in which the monomer unit is a thiophene-2,5-diyl group having a substituent at least at the 3-position can be synthesized, for example, as follows.
- the obtained organomagnesium compound of the formula (VI) is subjected to a coupling reaction in a solvent in the presence of a metal complex catalyst to obtain a ⁇ -conjugated polymer.
- a series of reactions is shown in Reaction Formula (VII).
- W, B and X are as defined above, and two Xs may be the same or different.
- the compound represented by the formula (IV) is a polymer precursor necessary for producing the polymer block B, and more specifically is represented by the following formula (1-1).
- R 2 has the same meaning as described above.
- X 1 and X 2 are bromine or iodine.
- X 1 is iodine
- X 2 is a bromine
- monomer units thiophene ring 4-position is a hydrogen atom 3-bromothiophene and bromohexyl can be coupled, and then synthesized by the bromination with N-bromosuccinimide and the iodination with iodine.
- the polymer block B is not limited to the method described above, in accordance with the polymer block A in the same manner as the formula (III), in the presence of a catalyst which is a monomer M q1 -Y-M q1 and M q2 -Z-M q2 taken by a coupling reaction with, it is possible to produce a compound B-X or the compound B-M p terminal functional groups attached to the polymer block B.
- Y and Z in this case represent a skeleton containing at least one thiophene-2,5-diyl group having a substituent at the 3-position which is a monomer unit of the polymer block B.
- a random copolymer block having a plurality of types of thiophene-2,5-diyl groups having a substituent at least at the 3-position, or a plurality of thiophene-2,5- A polymer block having a monomer unit as a group to which a diyl group is linked can be obtained.
- terminal blocker as needed.
- Macroinitiator method the polymerization initial compound A-X or A-M p having a polymer block A polymer block B, or polymerization medium term to coexist is a technique to carry out the polymerization of the polymer block B.
- the polymerization initial compound B-X or B-M p a polymer block A having a polymer block B, or polymerization medium term to coexist may be carried out polymerization of the polymer block A, [pi electrons of interest The optimum order can be selected depending on the conjugated block copolymer.
- the control easiness of polymerization, the polymerization initial compound B-X or B-M p a polymer block A having a polymer block B, or a method of performing the polymerization polymerization medium term to coexist in the polymer block A is more preferable. From the viewpoint of reducing the number of synthesis steps, a method in which the polymer block A is polymerized by allowing the compound BX having the polymer block B to coexist in the early stage of polymerization of the polymer block A or in the middle of the polymerization is further preferred.
- reaction formula (VIII) in the presence of a catalyst, a compound BX having a polymer block B and a monomer of the polymer block A M q1 -YM q1 and And / or M q2 -ZM q2 is reacted, and the terminal of the polymer block B and the monomer of the polymer block A or the polymer block A are bonded during the polymerization by a coupling reaction.
- a ⁇ -electron conjugated block copolymer of the present invention comprising A and polymer block B can be obtained.
- Polymer block B has a thiophene-2,5-diyl group having a substituent at least at the 3-position as a monomer unit.
- the substituent is an alkoxy group, a halogen atom, a hydroxyl group, an amino group, a thiol group, a silyl group, an ester group, an aryl group or a heteroaryl group, an alkyl group having 1 to 18 carbon atoms, an alkoxy group or an aryl group. It can be synthesized by the method described above using monomer units each having a substituted alkyl group having 1 to 18 carbon atoms, an alkoxy group, or an aryl group.
- the desired ⁇ -electron conjugated block copolymer can be obtained by appropriately protecting the hydroxyl group, amino group, thiol group, and heteroaryl group, or by post-modification after the coupling reaction.
- the hydroxyl group can be introduced into the polymer block B by protecting the hydroxyl group with a benzyl group or tetrahydropyranyl group, the amino group with a benzyloxycarbonyl group or t-butyloxycarbonyl group, and deprotecting after the coupling reaction.
- a benzyl group or tetrahydropyranyl group for example, a monomer having an alkyl group or an alkoxy group substituted with a halogen atom (especially a bromine atom is preferable) is first coupled, and then the halogen atom is substituted with thioacetic acid and hydrolyzed.
- a halogen atom especially a bromine atom is preferable
- transition metal complexes belonging to Group 3 to Group 10 of the periodic table Group 18 long-period type periodic table, particularly Group 8 to Group 10 are listed. Specific examples include known complexes such as Ni, Pd, Ti, Zr, V, Cr, Co, and Fe. Of these, Ni complexes and Pd complexes are more preferable.
- the ligand of the complex to be used is a monodentate phosphine coordination such as trimethylphosphine, triethylphosphine, triisopropylphosphine, tri-t-butylphosphine, tricyclohexylphosphine, triphenylphosphine, tris (2-methylphenyl) phosphine, etc.
- Diphenylphosphinomethane (dppm), 1,2-diphenylphosphinoethane (dppe), 1,3-diphenylphosphinopropane (dppp), 1,4-diphenylphosphinobutane (pdbb), 1,3- Bidentate phosphine compounds such as bis (dicyclohexylphosphino) propane (dcpp), 1,1′-bis (diphenylphosphino) ferrocene (dppf), 2,2-dimethyl-1,3-bis (diphenylphosphino) propane Lico; tetramethyl Ethylenediamine, bipyridine, it is preferable that such a nitrogen-containing ligands such as acetonitrile is contained.
- a nitrogen-containing ligands such as acetonitrile
- the amount of the complex used varies depending on the kind of ⁇ -electron conjugated block copolymer produced by both the linking method and the macroinitiator method, but is preferably 0.001 to 0.1 mol based on the monomer. If the amount of the catalyst is too large, it will cause a decrease in the molecular weight of the resulting polymer, which is also disadvantageous economically. On the other hand, if the amount is too small, the reaction rate becomes slow and stable production becomes difficult.
- the ⁇ -electron conjugated block copolymer of the present invention is preferably produced in the presence of a solvent.
- Solvents that can be used for the production need to be selected depending on the type of ⁇ -electron conjugated block copolymer to be produced, but a commercially available solvent can be selected for both the linking method and the macroinitiator method. .
- ethers such as tetrahydrofuran, 2-methyltetrahydrofuran, 1,4-dioxane, dimethyl ether, ethyl methyl ether, diethyl ether, dipropyl ether, butyl methyl ether, t-butyl methyl ether, dibutyl ether, cyclopentyl methyl ether, diphenyl ether Solvent, aliphatic or alicyclic saturated hydrocarbon solvent such as pentane, hexane, heptane, cyclohexane, aromatic hydrocarbon solvent such as benzene, toluene, xylene, alkyl halide solvent such as dichloromethane, chloroform, chlorobenzene, Aromatic aryl halide solvents such as dichlorobenzene, amide solvents such as dimethylformamide, diethylformamide, N-methylpyrrolidone, water, and these And mixtures thereof.
- the amount of the organic solvent used is preferably in the range of 1 to 1000 times by weight with respect to the monomer of the ⁇ -electron conjugated block copolymer to be produced, and the solubility of the resulting conjugate and the stirring efficiency of the reaction solution From the viewpoint of the above, it is preferably 10 times by weight or more, and from the viewpoint of the reaction rate, it is preferably 100 times by weight or less.
- the polymerization temperature varies depending on the type of ⁇ -electron conjugated block copolymer to be produced. Both the linking method and the macroinitiator method are usually carried out in the range of ⁇ 80 to 200 ° C.
- the pressure of the reaction system is not particularly limited, but is preferably 0.1 to 10 atm. The reaction is usually carried out at around 1 atm.
- the reaction time varies depending on the ⁇ -electron conjugated block copolymer to be produced, but is usually 20 minutes to 100 hours.
- the ⁇ -electron conjugated block copolymer obtained by the linking method and the macroinitiator method is, for example, reprecipitation, solvent removal under heating, solvent removal under reduced pressure, solvent removal with steam (steam stripping), etc.
- the ⁇ -electron conjugated block copolymer can be separated and obtained from the reaction mixture and by-products by a normal operation when isolating from the solution.
- the obtained crude product can be purified by washing or extracting with a commercially available solvent using a Soxhlet extractor.
- ethers such as tetrahydrofuran, 2-methyltetrahydrofuran, 1,4-dioxane, dimethyl ether, ethyl methyl ether, diethyl ether, dipropyl ether, butyl methyl ether, t-butyl methyl ether, dibutyl ether, cyclopentyl methyl ether, diphenyl ether
- Solvents aliphatic or alicyclic saturated hydrocarbon solvents such as pentane, hexane, heptane, cyclohexane, aromatic hydrocarbon solvents such as benzene, toluene, xylene, ketone solvents such as acetone, ethyl methyl ketone, and diethyl ketone
- Alkyl halide solvents such as dichloromethane and chloroform, aromatic aryl halide solvents such as chlorobenzene and dichlorobenzene, dimethyl
- the above-mentioned ⁇ -electron conjugated block copolymer of the present invention has a terminal group in which a coupling residue such as a halogen atom, a trialkyltin group, a boronic acid group, or a boronic ester group, or an atom or group thereof is removed.
- the terminal structure may have a separated hydrogen atom, and these end groups are substituted with an end capping agent composed of an aromatic halide such as benzene bromide or an aromatic boronic acid compound. Also good.
- homopolymers or random polymer components such as polymer block A and polymer block B may remain in the ⁇ -electron conjugated block copolymer produced by the above method.
- the residual component is preferably 70% or less.
- the ⁇ -electron conjugated block copolymer of the present invention can be used for a photoelectric conversion active layer of a photoelectric conversion element by forming a composition with an electron accepting material.
- the electron-accepting material constituting the composition is not particularly limited as long as it is an organic material exhibiting n-type semiconductor characteristics.
- NTCDA 1,4,5,8-naphthalenetetracarboxylic dianhydride
- PTCDA 9,10-perylenetetracarboxylic dianhydride
- NTCDIC8H 9,10-perylenetetracarboxylic dianhydride
- 2- (4-biphenylyl) -5- (4 -Oxazole derivatives such as t-butylphenyl) -1,3,4-oxadiazole and 2,5-di (1-naphthyl) -1,3,4-oxadiazole
- 3- (4-biphenylyl)- Triazole derivatives such as 4-phenyl-5- (4-t-butylphenyl) -1,2,4-triazole, phenanthroline derivative Body, fullerene derivatives such as C 60 or C 70, carbon nanotube (CNT), poly -p- phenylene vinylen
- Fullerene derivatives suitably used as the electron-accepting material used in the present invention include unsubstituted ones such as C 60 , C 70 , C 76 , C 78 , C 82 , C 84 , C 90 , and C 94 , [6,6] - phenyl C 61 butyric acid methyl ester ([6,6] -C 61 -PCBM) , [5,6] - phenyl C 61 butyric acid methyl ester, [6,6] - phenyl C 61 butyric acid n-butyl ester, [6,6] -phenyl C 61 butyric acid i-butyl ester, [6,6] -phenyl C 61 butyric acid hexyl ester, [6,6] -phenyl C 61 Butyric acid dodecyl ester, [6,6] -diphenyl C 62 bis (butyric acid methyl ester) ([
- the above fullerene derivatives can be used alone or as a mixture thereof.
- [6,6] -C 61 -PCBM, [6,6] -C 62 -bis. -PCBM), [6,6] -C 71 -PCBM is preferably used.
- the photoelectric conversion element of the present invention is composed of a composition containing the above-mentioned ⁇ -electron conjugated block copolymer and an electron-accepting material, and can generate electric power when the composition functions as an organic photoelectric conversion layer.
- composition of the present invention may contain other components such as a surfactant, a binder resin, and a filler as long as the object of the present invention is not impaired.
- the ratio of the electron-accepting material in the composition of the present invention is preferably 10 to 1000 parts by weight and preferably 50 to 500 parts by weight with respect to 100 parts by weight of the ⁇ -electron conjugated block copolymer. More preferred.
- the mixing method of the ⁇ -electron conjugated block copolymer and the electron-accepting material is not particularly limited, but after adding to the solvent at a desired ratio, one method such as heating, stirring, and ultrasonic irradiation is used. Alternatively, a method may be mentioned in which a plurality of types are combined and dissolved in a solvent to form a solution.
- the solvent whose solubility in 20 degreeC is 1 mg / mL or more about each of (pi) electron conjugated block copolymer and an electron-accepting material.
- the solubility is less than 1 mg / mL, it is difficult to produce a homogeneous organic thin film, and the composition of the present invention cannot be obtained.
- the boiling point of these solvents is preferably in the range of room temperature to 200 ° C. from the viewpoint of film forming properties and the manufacturing process described later.
- solvent examples include tetrahydrofuran, 1,2-dichloroethane, cyclohexane, chloroform, bromoform, benzene, toluene, o-xylene, chlorobenzene, bromobenzene, iodobenzene, o-dichlorobenzene, anisole, methoxybenzene, trichlorobenzene, pyridine. Etc. These solvents may be used singly or as a mixture of two or more kinds, but o-dichlorobenzene, chlorobenzene, bromobenzene having particularly high solubility of the ⁇ -electron conjugated block copolymer and the electron-accepting material. , Iodobenzene, chloroform and mixtures thereof are preferred. More preferably, o-dichlorobenzene, chlorobenzene and a mixture thereof are used.
- the solution may contain an additive having a boiling point higher than that of the solvent.
- an additive having a boiling point higher than that of the solvent.
- the additive include octanedithiol (boiling point: 270 ° C.), dibromooctane (boiling point: 272 ° C.), diiodooctane (boiling point: 327 ° C.), and the like.
- the addition amount of the additive used in the present invention is not particularly limited as long as the ⁇ -electron conjugated block copolymer and the electron-accepting material do not precipitate and give a uniform solution, but the volume fraction with respect to the solvent
- the content is preferably 0.1 to 20%.
- the additive amount is less than 0.1%, a sufficient effect cannot be obtained to form a fine and continuous phase separation structure.
- the additive amount is more than 20%, The drying speed becomes slow and it becomes difficult to obtain a homogeneous organic thin film. More preferably, it is in the range of 0.5 to 10%.
- the film thickness of the organic photoelectric conversion layer is usually 1 nm to 1 ⁇ m, preferably 2 to 1000 nm, more preferably 5 to 500 nm, and further preferably 20 to 300 nm. If the film thickness is too thin, the light is not sufficiently absorbed, and conversely if it is too thick, the carriers are difficult to reach the electrode.
- the method for coating a solution containing a ⁇ -electron conjugated block copolymer and an electron-accepting material on a substrate or a support is not particularly limited, and a conventionally known coating using a liquid coating material. Any of the methods can be employed. For example, dip coating method, spray coating method, inkjet method, aerosol jet method, spin coating method, bead coating method, wire bar coating method, blade coating method, roller coating method, curtain coating method, slit die coater method, gravure coater method , Slit reverse coater method, micro gravure method, comma coater method, etc. can be adopted, and the coating method can be selected according to the coating film properties to be obtained, such as coating thickness control and orientation control. That's fine.
- the organic photoelectric conversion layer may be further annealed as necessary.
- Thermal annealing is performed by holding the substrate on which the organic thin film of the present invention is formed at a desired temperature.
- Thermal annealing may be performed under reduced pressure or in an inert gas atmosphere, and a preferable temperature is 40 to 300 ° C., more preferably 70 to 200 ° C. If the temperature is low, a sufficient effect cannot be obtained, and if the temperature is too high, the organic thin film is oxidized and / or decomposed, and sufficient photoelectric conversion characteristics cannot be obtained.
- the substrate on which the photoelectric conversion element of the present invention is formed may be any substrate that does not change when an electrode or an organic photoelectric conversion layer is formed.
- inorganic materials such as alkali-free glass and quartz glass, metal films such as aluminum, and any organic material such as polyester, polycarbonate, polyolefin, polyamide, polyimide, polyphenylene sulfide, polyparaxylene, epoxy resin and fluorine resin Films and plates produced by the method can be used.
- the opposite electrode that is, the electrode far from the substrate
- the film thickness of the substrate is not particularly limited, but is usually in the range of 1 ⁇ m to 10 mm.
- the positive electrode or the negative electrode have light transmittance.
- the optical transparency of the electrode is not particularly limited as long as incident light reaches the organic photoelectric conversion layer and an electromotive force is generated.
- the thickness of the electrode may be in a range having light transmittance and conductivity, and is preferably 20 to 300 nm although it varies depending on the electrode material.
- the other electrode is not necessarily light-transmitting as long as it has conductivity, and the thickness is not particularly limited.
- the positive electrode included in the photoelectric conversion element is a metal such as lithium, magnesium, calcium, tin, gold, platinum, silver, copper, chromium, or nickel
- the transparent electrode is a metal oxide such as indium or tin, or a composite metal.
- Oxides indium tin oxide (ITO), indium zinc oxide (IZO), fluorine-doped tin oxide (FTO), etc.
- ITO indium tin oxide
- IZO indium zinc oxide
- FTO fluorine-doped tin oxide
- Examples of the method for producing a grid electrode, polyaniline and derivatives thereof in which a metal is made into a mesh shape include vacuum deposition, sputtering, ion plating, and plating.
- a positive electrode can also be produced by a coating method using a metal ink, a metal paste, a low melting point metal, an organic conductor ink, or the like.
- Metal fluorides such as lithium fluoride, sodium fluoride, potassium fluoride, magnesium fluoride, calcium fluoride, cesium fluoride, more preferably lithium fluoride, cesium fluoride are provided between the negative electrode and the organic photoelectric conversion layer. By introducing it, it is possible to improve the extraction current.
- a hole transport layer may be provided between the positive electrode and the organic photoelectric conversion layer.
- the material for forming the hole transport layer is not particularly limited as long as it has p-type semiconductor characteristics.
- polythiophene polymers, polyaniline polymers, poly-p-phenylene vinylene polymers, polyfluorene polymers are not particularly limited.
- Low molecular organic compounds exhibiting p-type semiconductor properties such as conductive polymers such as coalescence, phthalocyanine (Pc), phthalocyanine derivatives (such as copper phthalocyanine, zinc phthalocyanine), porphyrin derivatives, molybdenum oxide, zinc oxide, vanadium oxide, etc.
- the metal oxide is preferably used.
- the thickness of the hole transport layer is preferably 1 to 600 nm, more preferably 20 to 300 nm.
- an electron transport layer may be provided between the negative electrode and the active layer as necessary.
- the material for forming the electron transport layer is not particularly limited as long as it has n-type semiconductor characteristics, but the above-described electron-accepting organic materials (NTCDA, PTCDA, NTCDI-C8H, oxazole derivatives, triazole derivatives, phenanthroline derivatives, Fullerene derivatives, CNT, CN-PPV, etc.) are preferably used.
- the thickness of the electron transport layer is preferably 1 to 600 nm, more preferably 5 to 100 nm.
- a dip coating method, a spray coating method, an ink jet method, an aerosol jet method, a spin coating method, a bead It can be applied by coating method, wire bar coating method, blade coating method, roller coating method, curtain coating method, slit die coater method, gravure coater method, slit reverse coater method, micro gravure method, comma coater method and the like.
- a low molecular weight organic material such as a phthalocyanine derivative or a porphyrin derivative
- the electron transport layer can be similarly produced.
- the photoelectric conversion element of the present invention can be applied to various photoelectric conversion devices using a photoelectric conversion function, an optical rectification function, and the like.
- a photoelectric conversion function such as solar cells
- electronic elements such as optical sensors, optical switches, and phototransistors
- optical recording materials such as optical memories
- the number average molecular weight and the weight average molecular weight are both determined in terms of polystyrene based on measurement by gel permeation chromatography (GPC).
- GPC gel permeation chromatography
- an HLC-8020 (product number) manufactured by Tosoh Corp. was used as the GPC apparatus, and a TSKgel Multipore HZ manufactured by Tosoh Corp. was connected in series as the column.
- the solvent was chloroform and the measurement temperature was 40 ° C.
- the polymer was purified using a preparative GPC column.
- a device for purification Recycling Preparative HPLC LC-908 manufactured by Japan Analytical Industry was used.
- the type of the column is one in which two styrene polymer columns 2H-40 and 2.5H-40 manufactured by Nippon Analytical Industries, Ltd. are connected in series. In addition, chloroform was used as an elution solvent.
- the solvent is dehydrated tetrahydrofuran (without stabilizer) manufactured by Wako Pure Chemical Industries, Ltd. and purified by distillation in the presence of metallic sodium, and then contacted with Molecular Sieves 5A manufactured by Wako Pure Chemical Industries, Ltd. for more than a day. Purification.
- cyclopenta [2,1-b: 3,4-b ′] dithiophene (0.36 g, 2.0 mmol) and 30 mL of tetrahydrofuran were added to a 100 mL three-necked flask and cooled to 0 ° C. or lower.
- a 1.6M n-butyllithium hexane solution (1.38 mL, 2.2 mmol) was slowly added dropwise, and the mixture was warmed to room temperature and stirred for 1 hour.
- the mixture was cooled again to 0 ° C. or lower, and 8-bromo-1-iodo-octane (0.64 g, 2.0 mmol) was added, followed by stirring for 1 hour.
- Polymer block A1 was synthesized according to the following reaction formula.
- the crude product was washed with acetone (200 mL) and hexane (200 mL) using a Soxhlet extractor and then extracted with chloroform (200 mL).
- the obtained solution was concentrated, poured into methanol (2 L), and the precipitated solid was collected by filtration and dried under reduced pressure to obtain polymer block A1 (1.04 g, 73%) as a black purple solid.
- the resulting polymer block A1 had a weight average molecular weight (Mw) of 45,500, a number average molecular weight (Mn) of 19,600, and a polydispersity of 2.32.
- Polymer block A2 was synthesized according to the following reaction formula.
- the crude product was washed with acetone (200 mL) and hexane (200 mL) using a Soxhlet extractor and then extracted with chloroform (200 mL).
- the obtained solution was concentrated, poured into methanol (2 L), and the precipitated solid was collected by filtration and dried under reduced pressure to obtain polymer block A2 (1.28 g, 64%) as a black purple solid.
- the resulting polymer block A2 had a weight average molecular weight (Mw) of 47,400, a number average molecular weight (Mn) of 18,900, and a polydispersity of 2.51.
- Polymer block A3 was synthesized according to the following reaction formula.
- the resulting polymer block A3 had a weight average molecular weight (Mw) of 36,000, a number average molecular weight (Mn) of 17,400, and a polydispersity of 2.07.
- Mw weight average molecular weight
- Mn number average molecular weight
- Polymer block A4 was synthesized according to the following reaction formula.
- the resulting polymer block A4 had a weight average molecular weight (Mw) of 40,800, a number average molecular weight (Mn) of 17,500, and a polydispersity of 2.33.
- Mw weight average molecular weight
- Mn number average molecular weight
- 1 H-NMR (270 MHz): ⁇ 8.10-7.95 (m, 2H), 7.82-7. 60 (m, 2H), 3.18-2.97 (m, 4H), 2.32-2.07 (m, 4H), 1.62-1.34 (m, 8H), 1.36- 1.09 (m, 16H)
- Polymer block B1 was synthesized according to the following reaction formula.
- the resulting black purple solid was dissolved in chloroform (30 mL), reprecipitated in methanol (300 mL), and sufficiently dried, and purified using a preparative GPC column to form a polymer block.
- B1 (0.69 g, 83%) was obtained.
- the resulting polymer block B1 had a weight average molecular weight of 24,200, a number average molecular weight of 21,000, and a polydispersity of 1.15.
- Polymer block B2 was synthesized according to the following reaction formula.
- polymer block B2 (0.58 g, 56%) was obtained in the same manner as in Polymerization Example 5.
- the weight average molecular weight of the obtained polymer block B2 was 27,800, several flats. Molecular weight 12,800 and a polydispersity of 2.16.
- Polymer block B3 was synthesized according to the following reaction formula.
- the crude product was washed with acetone (100 mL) and hexane (100 mL) using a Soxhlet extractor, and then extracted with chloroform (100 mL).
- the obtained solution was concentrated, poured into methanol (200 mL), and the precipitated solid was collected by filtration and dried under reduced pressure to obtain polymer block B3 (0.35 g, 85%).
- the resulting polymer block B3 had a weight average molecular weight of 33,000, a number average molecular weight of 15,000, and a polydispersity of 2.20.
- Example 1 The block copolymer 1 was synthesized according to the following reaction formula.
- polymer block A1 (0.80 g, 1.50 mmol)
- polymer block B1 (0.25 g, 1.50 mmol)
- toluene (20 mL)
- 2M aqueous potassium carbonate solution 10 mL, 20 mmol
- tetrakis (triphenylphosphine) palladium (0) (20.5 mg, 17.7 ⁇ mol)
- aliquat 336 0.8 mg, 1.98 ⁇ mol
- the reaction solution was poured into methanol (200 mL), the precipitated solid was collected by filtration, washed with water (20 mL) and methanol (20 mL), and the resulting solid was dried under reduced pressure to obtain a crude product. It was.
- the crude product was washed with acetone (100 mL) and hexane (100 mL) using a Soxhlet extractor and then extracted with chloroform (100 mL).
- the obtained solution was concentrated, poured into methanol (200 mL), and the precipitated solid was collected by filtration and dried under reduced pressure to obtain block copolymer 1 (0.32 g, 31%) as a black purple solid.
- the obtained block copolymer 1 had a weight average molecular weight (Mw) of 86,100, a number average molecular weight (Mn) of 41,000, and a polydispersity of 2.10.
- Mw weight average molecular weight
- Mn number average molecular weight
- Example 2 The block copolymer 2 was synthesized according to the following reaction formula.
- Example 3 The block copolymer 3 was synthesized according to the following reaction formula.
- Synthesis was performed in the same manner as in Example 1 except that the polymer block A3 (1.05 g, 1.50 mmol) and the polymer block B1 (0.25 g, 1.50 mmol) were used, and the block copolymer 3 ( 0.43 g, 33%).
- the obtained block copolymer 3 had a weight average molecular weight (Mw) of 76,800, a number average molecular weight (Mn) of 39,200, and a polydispersity of 1.96.
- Example 4 The block copolymer 4 was synthesized according to the following reaction formula.
- Synthesis was performed in the same manner as in Example 1 except that the polymer block A4 (1.04 g, 1.50 mmol) and the polymer block B1 (0.25 g, 1.50 mmol) were used, and the block copolymer 4 ( 1.32 g, 32%).
- the resulting block copolymer 4 had a weight average molecular weight (Mw) of 83,600, a number average molecular weight (Mn) of 39,800, and a polydispersity of 2.10.
- Example 5 A block copolymer 1 ′ was synthesized according to the following reaction formula.
- polymer block B1 (0.15 g, 0.90 mmol) and 2,6-dibromo-4,4′-bis (2 -Ethylhexyl) -cyclopenta [2,1-b: 3,4-b ′] dithiophene (0.51 g, 0.90 mmol) and 4,7-bis (3,3,4,4-tetramethyl-2,5 , 1-Dioxaborolan-1-yl) benzo [c] [1,2,5] thiadiazole (0.35 g, 0.90 mmol) was added, and toluene (17 mL) and 2M aqueous potassium carbonate solution (8.5 mL, 17 mmol) were added.
- the obtained solution was concentrated, poured into methanol (200 mL), and the precipitated solid was collected by filtration and then dried under reduced pressure to obtain block copolymer 1 ′ (0.36 g, 75%) as a black purple solid. .
- the resulting block copolymer 1 ′ had a weight average molecular weight (Mw) of 78,800, a number average molecular weight (Mn) of 33,700, and a polydispersity of 2.34.
- Example 6 The block copolymer 5 was synthesized according to the following reaction formula.
- polymer block B1 (0.15 g, 0.9 mmol) and 4,4′-bis (2-ethylhexyl) -2, which is a monomer constituting polymer block A, 6-bis (trimethyltin) -cyclopenta [2,1-b: 3,4-b ′] dithiophene (0.65 g, 0.90 mmol) and 3.6-bis (5-bromothiophen-2-yl)- 2,5-bis (2-ethylhexyl) pyrrolo [3,4-c] pyrrole-1,4 (2H, 5H) -dione (0.61 g, 0.90 mmol) was added, and toluene (15 mL) was further added.
- block copolymer 5 (0.58 g, 70%).
- the obtained block copolymer 5 had a weight average molecular weight (Mw) of 86,400, a number average molecular weight (Mn) of 43,200, and a polydispersity of 2.00.
- Mw weight average molecular weight
- Mn number average molecular weight
- Example 7 The block copolymer 6 was synthesized according to the following reaction formula.
- polymer block B1 (0.15 g, 0.90 mmol) and 2,6-dibromo-4,4′-bis (2- Ethylhexyl) -dithieno [3,2-b: 2 ′, 3′-d] germole (0.56 g, 0.90 mmol) and 4,7-bis (3,3,4,4-tetramethyl-2,5 , 1-dioxaborolan-1-yl) benzo [c] [1,2,5] thiadiazole (0.35 g, 0.90 mmol) was synthesized in the same manner as in Example 5 to obtain a block copolymer.
- Example 8 The block copolymer 7 was synthesized according to the following reaction formula.
- polymer block B1 (0.15 g, 0.90 mmol) and 1- (4,6-dibromothieno [3,4-b] thiophene as a monomer constituting polymer block A -2-yl) -2-ethylhexane-1-one (0.38 g, 0.90 mmol) and 2,6-bis (trimethyltin) -4,8-bis (2-ethylhexyloxy) benzo [1,2 -B: 4,5-b '] dithiophene (0.70 g, 0.90 mmol) and DMF (3.8 mL) were used in the same manner as in Example 6 except that block polymer 7 ( 0.79 g, quant.) Was obtained.
- the obtained block copolymer 7 had a weight average molecular weight (Mw) of 219,000, a number average molecular weight (Mn) of 37,000, and a polydispersity of 6.00.
- Mw weight average molecular weight
- Mn number average molecular weight
- polydispersity 6.00.
- Polymer block B2 (0.16 g, 0.90 mmol) and 2,5-bis (2-ethylhexyl-3,6-di (5-bromothiophen-2-yl) which is a monomer constituting polymer block A ) Pyrrolo [3,4-c] -2,5-dihydro-1,4-dione (0.61 g, 0.90 mmol) and 2,6-bis (trimethyltin) -4,8-bis (octyloxy) benzo Synthesis was performed in the same manner as in Example 6 except that [1,2-b: 4,5-b ′] dithiophene (0.70 g, 0.90 mmol) was used, and the block polymer 8 (0.56 g, The resulting block copolymer 8 had a weight average molecular weight (Mw) of 77,000, a number average molecular weight (Mn) of 35,000, and a polydispersity of 2.20.
- Mw weight average molecular weight
- Example 10 The block copolymer 9 was synthesized according to the following reaction formula.
- polymer block B3 (0.42 g, 0.90 mmol) and 4,4-bis (2-ethylhexyl) -2, a monomer constituting polymer block A, 6-bis (trimethyltin) -dithieno [3,2-b: 2 ′, 3′-d ′] silole (0.67 g, 0.90 mmol) and 3-dibromo-5-octylthieno [3,4-c] Pyrrole-4,6-dione (0.38 g, 0.90 mmol) was added, and DMF (3.8 mL), toluene (15 mL), and tetrakis (triphenylphosphine) palladium (0) (60 mg, 0.
- the resulting block copolymer 9 had a weight average molecular weight (Mw) of 189,000, a number average molecular weight (Mn) of 65,000, and a polydispersity of 2.90.
- Mw weight average molecular weight
- Mn number average molecular weight
- Example 11 The block copolymer 10 was synthesized according to the following reaction formula.
- the resulting block copolymer 10 had a weight average molecular weight of 232,000, a number average molecular weight of 61,000, and a polydispersity of 3.80.
- Block copolymer 1 (16.0 mg) obtained in Example 1, PCBM (E100H manufactured by Frontier Carbon Co.) (12.8 mg) as an electron-accepting material, and chlorobenzene (1 mL) as a solvent at 40 ° C. For 6 hours. Then, it cooled to room temperature 20 degreeC, and filtered with the PTFE filter with the hole diameter of 0.45 micrometer, and manufactured the solution containing a block copolymer and PCBM. For each block copolymer obtained in Examples 2 to 11, solutions containing PCBM were produced in the same manner.
- a glass substrate provided with an ITO film (resistance value 10 ⁇ / ⁇ ) with a thickness of 150 nm by sputtering was subjected to surface treatment by ozone UV treatment for 15 minutes.
- a PEDOT: PSS aqueous solution (manufactured by HC Starck Co., Ltd .: CLEVIOS PH500) serving as a hole transport layer was formed on the substrate to a thickness of 40 nm by spin coating. After heating and drying at 140 ° C.
- Comparative Example 6 According to the method described in Example 7, polymerization was performed without adding the polymer block B1, whereby 4,4′-bis (2-ethylhexyl) -dithienogermol (GeT) and benzothiadiazole (BT) An alternating copolymer was synthesized, and an organic thin-film solar cell of Comparative Example 6 was prototyped in the same manner as described above.
- the organic thin-film solar cell formed using the ⁇ -electron conjugated block copolymer of the example includes the conjugated polymer of the comparative example and the ⁇ -electron conjugated block copolymer. High photoelectric conversion efficiency was shown compared with the organic thin-film solar cell made by trial use.
- novel ⁇ -electron conjugated block copolymer according to the present invention can be used as a photoelectric conversion layer of a photoelectric conversion element, and the photoelectric conversion element comprising the copolymer is used as various photosensors including solar cells. There are uses.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Polyoxymethylene Polymers And Polymers With Carbon-To-Carbon Bonds (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Abstract
Description
少なくとも3位に置換基を有するチオフェン-2,5-ジイル基を単量体単位とする重合体ブロックBとからなるπ電子共役系ブロック共重合体である。
-b-は下記式(7)~(17)
上記式(1)~(17)中、V1は窒素(-NR1-)、酸素(-O-)または硫黄(-S-)であり、V2は炭素(-CR1 2-)、窒素(-NR1-)、ケイ素(-SiR1 2-)またはゲルマニウム(-GeR1 2-)であり、V3は-(Ar)n-で表されるアリール基またはヘテロアリール基であり、V4は窒素(-NR1-)、酸素(-O-)または-CR2=CR2-であり、V5は酸素(O)または硫黄(S)である。
R1はそれぞれ独立に置換されていてもよい炭素数1~18のアルキル基であり、R2はそれぞれ独立に水素原子または置換されていてもよい炭素数1~18のアルキル基であり、R3はそれぞれ独立に置換されていてもよい炭素数1~18のアルキル基またはアルコキシ基であり、R4はそれぞれ独立に水素原子、ハロゲン原子または置換されていてもよい炭素数1~18のアルキル基もしくはアリール基であり、R5は置換されていてもよい炭素数1~18のアルキル基、アリール基、アルキルカルボニル基またはアルキルオキシカルボニル基であり、R6は水素原子またはハロゲン原子である。
mは1~3の整数、nは0~3の整数を表す。
上記式(18)~(28)中、V2は炭素(-CR1 2-)、窒素(-NR1-)、ケイ素(-SiR1 2-)またはゲルマニウム(-GeR1 2-)であり、V3は-(Ar)n-で表されるアリール基またはヘテロアリール基である。
R1、R2、R3、R4、R5およびR6は前記定義の通りである。
nは0~3の整数を表す。
本発明に用いられる重合体ブロックAにおいて、単量体単位-a-b-の態様まで含む1種類の単量体単位中の置換基Rnを除く環構造のみを構成する炭素原子の合計数は、6~30であるのが好ましい。
ハロゲン原子としては、フッ素原子、塩素原子、臭素原子、ヨウ素原子が挙げられる。ハロゲン原子で置換されたアルキル基としては、例えばω-ブロモアルキル基、パーフルオロアルキル基などが挙げられる。
チオール基としては、例えばメルカプト基、アルキルチオ基が挙げられる。
シリル基としては、例えばトリメチルシリル基、トリエチルシリル基、トリプロピルシリル基、トリイソプロピルシリル基、ジメチルイソプロピルシリル基、ジメチルtert-ブチルシリル基などが挙げられる。
アリール基としては、例えばフェニル基、1-ナフチル基、2-ナフチル基、1-アントラセニル基、2-アントラセニル基、9-アントラセニル基などが例示され、これらはさらにアルキル基、アルコキシ基などの置換基を有していてもよい。
ヘテロアリール基としては、例えばピリジル基、チエニル基、フリル基、ピロリル基などが挙げられる。
こうすることで化合物A-X、化合物A-Mpのように重合体ブロックAの片方の末端基のみに連結のための官能基を導入しやすくなる。
X-W-X ・・・ (IV)
(式(IV)中、Wは少なくとも3位に置換基を有する2価のチエニレン基、Xはハロゲン原子、2つのXは同一であっても異なっていてもよい)で示される単量体と、グリニャール試薬である下記式(V)
R’-MgX ・・・ (V)
(式(V)中、R’は炭素数1~10アルキル基、Xは前記と同義である)で示される有機マグネシウムハロゲン化合物との交換反応であるグリニャールメタセシス反応により、下記式(VI)
X-W-MgX ・・・ (VI)
(式(VI)中、WおよびXは前記と同義であり、2つのXは同一または異なる)で示される有機マグネシウム化合物を得る。
数平均分子量および重量平均分子量は、何れも、ゲル・パーミエーション・クロマトグラフィー(GPC)による測定に基づき、ポリスチレン換算値で求められたものである。ここでは、GPC装置として、東ソー(株)製のHLC-8020(品番)を用い、カラムとして、東ソー(株)製のTSKgel Multipore HZの2本を直列に繋いだものを用いた。溶媒はクロロホルムを用い、測定温度は40℃とした。
重合体の精製には分取用のGPCカラムを用いて精製を行った。精製用の装置は、Japan Analytical Industry(株)製のRecycling Preparative HPLC LC-908を用いた。なお、カラムの種類は、日本分析工業(株)製のスチレン系ポリマーカラム 2H-40および2.5H-40を2本直列に接続したものである。また、溶出溶媒はクロロホルムを用いた。
溶媒は、和光純薬工業(株)製の脱水テトラヒドロフラン(安定剤不含)を、金属ナトリウム存在下蒸留精製を行った後、和光純薬工業(株)製のモレキュラーシーブス5Aに一日以上接触させることで、精製を行った。
1H-NMR測定には日本電子株式会社製JNM-EX270FT-NMR装置を用いた。なお、本明細書中、特に記載がなければ1H-NMRは270MHz、溶媒はクロロホルム(CDCl3)であり、室温下で、測定したものである。
1H-NMR(270MHz):δ=7.12(d、J=4.9Hz、2H)、7.07(d、J=4.9Hz、2H)、3.15(t、J=7.0Hz、4H)、2.33-2.10(m、4H)、1.52-1.41(m、12H)、1.38-1.09(m、16H)
MS(GC-MS)m/z=560(M+)
1H-NMR(270MHz):δ=6.94(s、2H)、3.15(t、J=7.0Hz、4H)、2.35-2.12(m、4H)、1.54-1.40(m、12H)、1.37-1.06(m、16H)
MS(GC-MS)m/z=718
1H-NMR(270MHz):δ=8.10-7.95(m、2H)、7.80-7.61(m、2H)、2.35-2.12(m、4H)、1.60-1.32(m、18H)、1.18-0.82(m、12H)
1H-NMR(270MHz):δ=8.11(d、J=3.5Hz、2H)、7.45(d、J=3.6Hz、2H)、2.32-2.11(m、4H)、1.61-1.34(m、28H)、1.32-1.20(m、4H)1.15-0.82(m、6H)
1H-NMR(270MHz):δ=8.11(d、J=3.5Hz、2H)、8.02(d、J=7.0Hz、2H)、7.94(br、2H)、7.75(br、2H)、7.56(d、J=3.8Hz、2H)、7.45(d、J=3.5Hz、2H)、4.73(br、1H)、2.42(br、2H)、2.05(br、2H)、1.25-1.18(m、8H)、0.75-0,65(m、3H)
1H-NMR(270MHz):δ=8.10-7.95(m、2H)、7.82-7.
60(m、2H)、3.18-2.97(m、4H)、2.32-2.07(m、4H)、1.62-1.34(m、8H)、1.36-1.09(m、16H)
乾燥させたアルゴン置換したナスフラスコBに、脱水および過酸化物除去処理を行ったTHF(25mL)とNiCl2(dppp)(27mg,0.05mmol)を加えて35℃に加熱した後、有機マグネシウム化合物溶液(a4)を添加した。35℃で1.5時間加熱攪拌した後、5M塩酸(50mL)を加えて室温で1時間攪拌した。この反応液をクロロホルム(450mL)で抽出した後、有機層を重曹水(100mL)、蒸留水(100mL)の順で洗浄し、有機層を無水硫酸ナトリウムで乾燥後、濃縮乾固した。得られた黒紫色の固体を、クロロホルム(30mL)に溶かして、メタノ-ル(300mL)に再沈殿し、充分に乾燥したものを、分取用GPCカラムを用いて精製することにより重合体ブロックB1(0.69g,83%)を得た。得られた重合体ブロックB1の重量平均分子量は24,200、数平均分子量は21,000、多分散度は1.15であった。
1H-NMR(270MHz):δ=6.97(s、1H)、2.80(t、J=8.0Hz、2H)、1.89-1.27(m、10H)、0.91(t、J=6.8Hz、3H)
1H-NMR(270MHz):δ=6.97(s、1H)、6.94(s、0.4H)、2.80(t、J=8.0Hz、2.8H)、1.70-1.25(m、11.6H)、0.94-0.89(m、5.4H)
1H-NMR(270MHz):δ=7.05-6.95(m、5H)、2.80(t、J=8.0Hz、2H)、1.70-1.20(m、25H)、1.19-0.83(m、6H)
1H-NMR(270MHz):δ=8.12-7.94(m、2H)、7.80-7.61(m、2H)、6.97(s、1H)、2.80(t、J=8.0Hz、2H)、2.35-2.12(m、4H)、1.62-1.31(m、26H)、1.19-0.83(m、15H)
1H-NMR(270MHz):δ=8.11(d、J=3.5Hz、2H)、δ=8.08-7.93(m、8H)、7.45(d、J=3.6Hz、2H)、6.97(s、1H)、3.70-3.60(m、2H)、2.86-2.72(m、4H)、2.33-2.12(m、4H)、1.70-1.32(m、38H)、1.19-0.82(m、9H)
1H-NMR(270MHz):δ=8.11(d、J=3.5Hz、2H)、8.02(d、J=7.0Hz、2H)、7.94(m、2H)、7.75(m、2H)、7.56(d、J=3.8Hz、2H)、7.45(d、J=3.5Hz、2H)、6.97(s、1H)、4.73(m、1H)、2.80(t、J=8.0Hz、2H)、2.42(m、2H)、2.05-1.20(m、20H)、1.20-0.84(m、9H)
1H-NMR(270MHz):δ=8.13-7.98(m、2H)、7.90-7.12(m、2H)、6.97(s、1H)、3.17-2.97(m、4H)、2.80(t、J=8.0Hz、2H)、2.33-2.09(m、4H)、1.61-1.39(m、20H)、1.35-1.20(m、12H)1.18-0.82(m、3H)
1H-NMR(270MHz):δ=8.20-8.12(m、2H)、8.00-7.80(m、2H)、6.97(s、1H)、2.91-2.72(m、2H)、1.62-1.31(m、26H)、1.19-0.83(m、15H)、2.80(t、J=8.0Hz、2H)、2.35-2.12(m、4H)、1.62-1.31(m、26H)、1.19-0.83(m、15H)
1H-NMR(270MHz):δ=8.91-8.77(m、2H)7.21-7.02(m、4H)、6.97(s、1H)、4.15-4.05(t、J=8.0Hz、4H)、2.80(t、J=8.0Hz、2H)、2.35-2.11(m、4H)、1.61-1.34(m、44H)、1.18-0.60(m、27H)
1H-NMR(270MHz):δ=8.21-7.94(m、2H)、7.92-7.13(m、2H)、6.97(s、1H)、2.80(t、J=8.0Hz、2H)、2.35-2.11(m、4H)、1.61-1.31(m、32H)、1.19-0.83(m、15H)
1H-NMR(270MHz):δ=7.60‐7.30(br,3H)、6.97(s、1H)、4.30-4.00(br,4H),3.20-3.00(br,1H)、2.80(t、J=8.0Hz、2H)、2.00-0.60(br,55H)
1H-NMR(270MHz):δ=8.91-8.77(m、2H)7.21-7.02(m、4H)、6.97(s、0.7H)、6.94(s、0.3H)、4.15-4.05(t、J=8.0Hz、4H)、2.80(m、2H)、2.35-2.11(m、4H)、1.70-1.25(m、44.3H)、1.18-0.60(m、27.9H)
1H-NMR(270MHz):δ=8.59(s、1H)、7.52(s、1H)、7.05-6.95(m、5H)、3.64(br、2H)、2.80(t、J=8.0Hz、2H)、2.10-1.20(m、55H)、1.19-0.60(m、6H)
1H-NMR(270MHz):δ=7.94(br,2H)、7.84-7.82(br,2H)、7.51(br,2H)、7.33(br,2H)、7.05-6.95(m、5H)、2.80(t、J=8.0Hz、2H)、2.71-2.54(br,12H)、1.70-1.20(m、37H)、1.19-0.70(m、92H)
実施例1により得られたブロック共重合体1(16.0mg)と、電子受容性材料としてPCBM(フロンティアカーボン社製 E100H)(12.8mg)と、溶媒としてクロロベンゼン(1mL)とを40℃にて6時間かけて混合した。その後、室温20℃に冷却し、孔径0.45μmのPTFEフィルターで濾過してブロック共重合体とPCBMを含む溶液を製造した。
実施例2~11により得られた各ブロック共重合体についても同じ方法により、PCBMを含む溶液を製造した。
スパッタ法により150nmの厚みでITO膜(抵抗値10Ω/□)を付けたガラス基板を15分間オゾンUV処理して表面処理を行った。基板上に正孔輸送層となるPEDOT:PSS水溶液(H.C.Starck社製:CLEVIOS PH500)をスピンコート法により40nmの厚さに成膜した。ホットプレートにより140℃で20分間加熱乾燥した後、次にスピンコートにより上記により製造した各ブロック共重合体とPCBMとを含む溶液を塗布し、有機薄膜太陽電池の有機光電変換層(膜厚約100nm)を得た。3時間真空乾燥し、120℃で30分間熱アニールした後、真空蒸着法により、真空蒸着機を用いてフッ化リチウムを膜厚1nmで蒸着し、次いでAlを膜厚100nmで蒸着した。これにより実施例1~11で得られたブロック共重合体を含む組成物からなる層を有する光電変換素子である有機薄膜太陽電池が得られた。有機薄膜太陽電池の形状は5×5mmの正四角形であった。
重合例1で得られた重合体ブロックA1を用いて、上記と同様に電子受容性材料との混合溶液を製造し、その混合溶媒を用いて比較例1の有機薄膜太陽電池を試作した。
比較例2~4については重合例2~4で得られた各重合体ブロックA2~A4、比較例7~9については重合例5~7で得られた重合体ブロックB1~B3を用いて上記と同じ方法により有機薄膜太陽電池を試作した。
実施例6に記載した方法に従い、重合体ブロックB1を加えずに重合を行うことで、4,4’-ビス(2-エチルヘキシル)-シクロペンタジチオフェン(CT)と、3.6-ビス(チオフェン-2-イル)-2,5-ビス(2-エチルヘキシル)ジケトピロロピロール(DPP)との交互共重合体を合成し、上記と同様にして比較例5の有機薄膜太陽電池を試作した。
実施例7に記載した方法に従い、重合体ブロックB1を加えずに重合を行うことで4,4’-ビス(2-エチルヘキシル)-ジチエノゲルモール(GeT)と、ベンゾチアジアゾール(BT)との交互共重合体を合成し、上記と同様にして比較例6の有機薄膜太陽電池を試作した。
共役系重合体として重合体ブロックA1(3mg)と、重合体ブロックB1(13mg)と、電子受容性材料としてPCBM(フロンティアカーボン社製E100H)(16mg)と、溶媒としてオルトジクロロベンゼン(1mL)とを40℃にて6時間かけて混合した。その後、室温20℃に冷却し、孔径0.45μmのPTFEフィルターで濾過して共役系重合体ブレンドとPCBMを含む溶液を製造した。この溶媒を用いて上記と同じ方法により、比較例8の有機薄膜太陽電池を試作した。
3-ブチルチオフェンと3-オクチルチオフェンのモル比が1:1のジブロック共重合体を合成し(尚、合成の詳細な手順はChem.Mater.,22,2020(2010)(非特許文献8)に記載されている)、上記と同様に電子受容性材料との混合溶液を製造し、その混合溶液を用いて比較例9の有機薄膜太陽電池を試作した。
4,8-ビス(2-エチルヘキシルオキシ)ベンゾ[1,2-b:4,5-b’]ジチオフェンと1-(チエノ[3,4-b]チオフェン-2-イル)-2-エチルヘキサン-1-オンの交互共重合体を合成し(尚、当該ジブロック共重合体の合成の詳細な手順はJ.Am.Chem.Soc.,131,15586(2009)(非特許文献10)に記載されている)、上記実施例、比較例と同様にして比較例10の有機薄膜太陽電池を試作した。
得られた各実施例、比較例の有機薄膜太陽電池の光電変換効率を300Wのソーラシミュレーター(ペクセルテクノロジー社製、商品名PEC L11:AM1.5Gフィルター、放射照度100mW/cm2)で測定した。測定結果を表1ないし表4に示す。
Claims (9)
- 少なくとも一つのチオフェン環を化学構造の一部に含む縮環π共役骨格、フルオレン、カルバゾール、ジベンゾシロールおよびジベンゾゲルモールからなる群から選ばれるヘテロアリール骨格を、少なくとも一つ有する単量体単位からなる重合体ブロックAと、
少なくとも3位に置換基を有するチオフェン-2,5-ジイル基を単量体単位とする重合体ブロックBとからなるπ電子共役系ブロック共重合体。 - 前記重合体ブロックAは単量体単位-a-b-を含み、
-a-は下記式(1)~(6)
-b-は下記式(7)~(17)
R1はそれぞれ独立に置換されていてもよい炭素数1~18のアルキル基であり、R2はそれぞれ独立に水素原子または置換されていてもよい炭素数1~18のアルキル基であり、R3はそれぞれ独立に置換されていてもよい炭素数1~18のアルキル基またはアルコキシ基であり、R4はそれぞれ独立に水素原子、ハロゲン原子または置換されていてもよい炭素数1~18のアルキル基もしくはアリール基であり、R5は置換されていてもよい炭素数1~18のアルキル基、アリール基、アルキルカルボニル基またはアルキルオキシカルボニル基であり、R6は水素原子またはハロゲン原子である。
mは1~3の整数、nは0~3の整数を表す。)。 - 重合体ブロックBが、少なくとも3位に置換基を有するチオフェン-2,5-ジイル基を含む2~5個のチオフェン-2,5-ジイル基が連結した単量体単位からなる重合体ブロックである請求項1に記載されたπ電子共役系ブロック共重合体。
- 重合体ブロックBが、少なくとも3位に置換基を有するチオフェン-2,5-ジイル基を複数種類有するランダム共重合体ブロックである請求項1に記載されたπ電子共役系ブロック共重合体。
- 数平均分子量が1,000~200,000g/モルである請求項1~5のいずれかの請求項に記載されたπ電子共役系ブロック共重合体。
- 電子受容性材料、および請求項1~6のいずれかの請求項に記載されたπ電子共役系ブロック共重合体を含む組成物。
- 請求項7に記載の組成物からなる層を有する光電変換素子。
- 前記電子受容性材料が、フラーレンまたはその誘導体である請求項8に記載の光電変換素子。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013507799A JP5779233B2 (ja) | 2011-03-31 | 2012-03-30 | ブロック共重合体および光電変換素子 |
| US14/008,654 US20140166942A1 (en) | 2011-03-31 | 2012-03-30 | Block copolymer and photoelectric conversion element |
| CN2012800166627A CN103443163A (zh) | 2011-03-31 | 2012-03-30 | 嵌段共聚物及光电转换元件 |
| EP12762740.4A EP2692760A4 (en) | 2011-03-31 | 2012-03-30 | BLOCK COPOLYMER AND PHOTOELECTRIC CONVERSION ELEMENT |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011-080310 | 2011-03-31 | ||
| JP2011080310 | 2011-03-31 | ||
| JP2011-184629 | 2011-08-26 | ||
| JP2011184629 | 2011-08-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012133793A1 true WO2012133793A1 (ja) | 2012-10-04 |
Family
ID=46931488
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/058617 WO2012133793A1 (ja) | 2011-03-31 | 2012-03-30 | ブロック共重合体および光電変換素子 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20140166942A1 (ja) |
| EP (1) | EP2692760A4 (ja) |
| JP (1) | JP5779233B2 (ja) |
| CN (1) | CN103443163A (ja) |
| TW (1) | TW201245268A (ja) |
| WO (1) | WO2012133793A1 (ja) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012241017A (ja) * | 2011-05-13 | 2012-12-10 | Mitsubishi Chemicals Corp | ポリマー、有機半導体材料、並びにこれを用いた有機電子デバイス、光電変換素子及び太陽電池モジュール |
| WO2013073581A1 (ja) * | 2011-11-15 | 2013-05-23 | コニカミノルタ株式会社 | 有機光電変換素子、ならびにそれを用いた太陽電池及び光センサアレイ |
| JP2013220994A (ja) * | 2012-04-13 | 2013-10-28 | Sumitomo Chemical Co Ltd | 化合物及びそれを用いた電子素子 |
| JP2014531482A (ja) * | 2011-09-23 | 2014-11-27 | オーシャンズ キング ライティング サイエンス アンド テクノロジー シーオー.,エルティーディー | ジフルオロベンゾトリアゾリル有機物半導体材料、調合法、およびその使用方法 |
| JP2014531493A (ja) * | 2011-09-23 | 2014-11-27 | オーシャンズ キング ライティング サイエンスアンドテクノロジー カンパニー リミテッド | ジフルオロベンゾトリアゾリル太陽電池ポリマー材料、並びにその調製方法及び使用 |
| CN104211917A (zh) * | 2013-05-29 | 2014-12-17 | 海洋王照明科技股份有限公司 | 含苯并三唑单元的聚合物及其制备方法和太阳能电池器件 |
| JP2015013989A (ja) * | 2013-06-05 | 2015-01-22 | 三菱化学株式会社 | コポリマー、半導体層形成用組成物、有機電子デバイス及び太陽電池モジュール |
| WO2015092909A1 (ja) * | 2013-12-19 | 2015-06-25 | リンテック株式会社 | 高分子化合物、有機光電変換素子、及び該素子の製造方法 |
| WO2015128891A1 (en) * | 2014-02-28 | 2015-09-03 | Eni S.P.A. | Copolymers containing naphthodithiophene units and diketopyrrolopyrrole units and process for their preparation |
| JP2016017117A (ja) * | 2014-07-07 | 2016-02-01 | 国立研究開発法人理化学研究所 | 高分子化合物、有機半導体材料、光電変換素子及びトランジスタ |
| JP2016531171A (ja) * | 2013-07-15 | 2016-10-06 | エルジー・ケム・リミテッド | 共重合体およびこれを含む有機太陽電池 |
| WO2017047808A1 (ja) * | 2015-09-18 | 2017-03-23 | 三菱化学株式会社 | コポリマー、光電変換素子、太陽電池及び太陽電池モジュール |
| JP2019507215A (ja) * | 2016-01-27 | 2019-03-14 | エルジー・ケム・リミテッド | 共重合体およびこれを含む有機太陽電池 |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2496565B (en) * | 2010-09-04 | 2017-05-17 | Merck Patent Gmbh | Conjugated polymers based on BTZ dicarboxylic acid bis-ester monomers and analogues thereof |
| EP2706077A4 (en) * | 2011-03-11 | 2014-09-17 | Kuraray Co | PI ELECTRON CONJUGATED BLOCK COPOLYMER AND PHOTOELECTRIC CONVERSION ELEMENT |
| CN105473635B (zh) * | 2014-02-14 | 2017-11-28 | 株式会社Lg化学 | 共聚物和包含其的有机太阳能电池 |
| KR101636687B1 (ko) * | 2014-07-21 | 2016-07-06 | 한국과학기술연구원 | 낮은 밴드갭을 갖는 고분자 화합물, 그 제조 방법 및 이를 포함하는 유기태양전지 |
| CN107207534B (zh) * | 2015-02-17 | 2019-10-22 | 株式会社Lg化学 | 杂环化合物和包含其的有机太阳能电池 |
| EP3464415B1 (en) | 2016-05-25 | 2020-08-19 | Basf Se | Semiconductors |
| US10787538B2 (en) * | 2018-01-29 | 2020-09-29 | Phillips 66 Company | Polymers for organic photovoltaics |
| CN116520637A (zh) * | 2018-10-05 | 2023-08-01 | Jsr株式会社 | 光学传感器用组合物 |
| KR102228274B1 (ko) * | 2019-02-21 | 2021-03-17 | 한국과학기술연구원 | 저온공정을 위한 공액 고분자 및 이를 이용한 유기태양전지 |
| CN112812277B (zh) * | 2020-12-30 | 2023-06-09 | 汕头大学 | 噻吩稠合萘单酰亚胺聚合物及其制备方法和应用 |
| CN114479018B (zh) * | 2021-12-28 | 2023-12-19 | 香港理工大学深圳研究院 | 含有二茂铁的环戊二噻吩型金属有机聚合物及其制备方法与应用、复合热电薄膜 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080006324A1 (en) | 2005-07-14 | 2008-01-10 | Konarka Technologies, Inc. | Tandem Photovoltaic Cells |
| JP2008515194A (ja) * | 2004-09-24 | 2008-05-08 | プレックストロニクス インコーポレーティッド | エレクトロルミネセンス装置におけるヘテロ原子位置規則性ポリ(3−置換チオフェン) |
| WO2008093822A1 (ja) * | 2007-02-01 | 2008-08-07 | Sumitomo Chemical Company, Limited | ブロック共重合体及び高分子発光素子 |
| JP2008534751A (ja) * | 2005-04-01 | 2008-08-28 | カーネギー メロン ユニバーシティ | レジオレギュラーポリマー、ポリチオフェンおよびブロックコポリマーを含む導電性ポリマーのリビング合成法 |
| JP2008266459A (ja) | 2007-04-20 | 2008-11-06 | Sumitomo Chemical Co Ltd | 共重合体およびそれを用いた有機光電変換素子 |
| WO2009104781A1 (ja) * | 2008-02-18 | 2009-08-27 | 住友化学株式会社 | 組成物およびそれを用いた有機光電変換素子 |
| JP2010043217A (ja) * | 2008-08-18 | 2010-02-25 | Tokyo Institute Of Technology | π共役系高分子及びその製造方法、並びに電荷輸送材料及び有機電子デバイス |
| WO2011011545A1 (en) | 2009-07-24 | 2011-01-27 | Solarmer Energy, Inc. | Conjugated polymers with carbonyl substituted thieno [3,4-b] thiophene units for polymer solar cell active layer materials |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ATE328050T1 (de) * | 2002-04-24 | 2006-06-15 | Merck Patent Gmbh | Reaktive mesogene benzodithiophene |
| US7781673B2 (en) * | 2005-07-14 | 2010-08-24 | Konarka Technologies, Inc. | Polymers with low band gaps and high charge mobility |
| US8008421B2 (en) * | 2006-10-11 | 2011-08-30 | Konarka Technologies, Inc. | Photovoltaic cell with silole-containing polymer |
| WO2008088595A2 (en) * | 2006-10-11 | 2008-07-24 | Konarka Technologies, Inc. | Photovoltaic cell with silole-containing polymer |
| JP5480510B2 (ja) * | 2008-03-31 | 2014-04-23 | 住友化学株式会社 | 有機半導体組成物、並びに有機薄膜及びこれを備える有機薄膜素子 |
| WO2011085004A2 (en) * | 2010-01-05 | 2011-07-14 | Konarka Technologies, Inc. | Photovoltaic cell with benzodithiophene-containing polymer |
| JP2013533911A (ja) * | 2010-07-08 | 2013-08-29 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | 半導性ポリマー |
| WO2012030942A1 (en) * | 2010-09-02 | 2012-03-08 | Konarka Technologies, Inc. | Photovoltaic cell containing novel photoactive polymer |
| EP2706077A4 (en) * | 2011-03-11 | 2014-09-17 | Kuraray Co | PI ELECTRON CONJUGATED BLOCK COPOLYMER AND PHOTOELECTRIC CONVERSION ELEMENT |
| KR101853395B1 (ko) * | 2011-05-23 | 2018-04-30 | 삼성전자주식회사 | 전자 공여체 고분자 및 이를 포함하는 태양 전지 |
| TWI635111B (zh) * | 2012-03-16 | 2018-09-11 | 馬克專利公司 | 共軛聚合物 |
| WO2013182264A1 (en) * | 2012-06-05 | 2013-12-12 | Merck Patent Gmbh | Small molecules and their use as organic semiconductors |
| JP6465801B2 (ja) * | 2012-10-05 | 2019-02-06 | メルク パテント ゲーエムベーハー | 有機半導体 |
| KR101889920B1 (ko) * | 2012-12-21 | 2018-08-21 | 삼성전자주식회사 | 박막 형성 방법, 전자 소자 및 그 제조 방법 |
-
2012
- 2012-03-30 TW TW101111625A patent/TW201245268A/zh unknown
- 2012-03-30 US US14/008,654 patent/US20140166942A1/en not_active Abandoned
- 2012-03-30 CN CN2012800166627A patent/CN103443163A/zh active Pending
- 2012-03-30 JP JP2013507799A patent/JP5779233B2/ja not_active Expired - Fee Related
- 2012-03-30 WO PCT/JP2012/058617 patent/WO2012133793A1/ja active Application Filing
- 2012-03-30 EP EP12762740.4A patent/EP2692760A4/en not_active Withdrawn
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008515194A (ja) * | 2004-09-24 | 2008-05-08 | プレックストロニクス インコーポレーティッド | エレクトロルミネセンス装置におけるヘテロ原子位置規則性ポリ(3−置換チオフェン) |
| JP2008534751A (ja) * | 2005-04-01 | 2008-08-28 | カーネギー メロン ユニバーシティ | レジオレギュラーポリマー、ポリチオフェンおよびブロックコポリマーを含む導電性ポリマーのリビング合成法 |
| US20080006324A1 (en) | 2005-07-14 | 2008-01-10 | Konarka Technologies, Inc. | Tandem Photovoltaic Cells |
| WO2008093822A1 (ja) * | 2007-02-01 | 2008-08-07 | Sumitomo Chemical Company, Limited | ブロック共重合体及び高分子発光素子 |
| JP2008266459A (ja) | 2007-04-20 | 2008-11-06 | Sumitomo Chemical Co Ltd | 共重合体およびそれを用いた有機光電変換素子 |
| WO2009104781A1 (ja) * | 2008-02-18 | 2009-08-27 | 住友化学株式会社 | 組成物およびそれを用いた有機光電変換素子 |
| JP2010043217A (ja) * | 2008-08-18 | 2010-02-25 | Tokyo Institute Of Technology | π共役系高分子及びその製造方法、並びに電荷輸送材料及び有機電子デバイス |
| WO2011011545A1 (en) | 2009-07-24 | 2011-01-27 | Solarmer Energy, Inc. | Conjugated polymers with carbonyl substituted thieno [3,4-b] thiophene units for polymer solar cell active layer materials |
Non-Patent Citations (20)
| Title |
|---|
| ADV. FUNCT. MATER., vol. 19, 2009, pages 3262 |
| ADV. FUNCT. MATER., vol. 20, 2010, pages 338 |
| ADV. MATER., vol. 15, 2003, pages 988 |
| ADV. MATER., vol. 22, 2010, pages 3839 |
| ADV. MATER., vol. 22, 2010, pages E6 |
| ANGEW., CHEM. INT. ED, vol. 47, 2008, pages 58 |
| CHEM. MATER., vol. 22, 2010, pages 2020 |
| J. AM. CHEM. SCI., vol. 133, 2011, pages 4250 |
| J. AM. CHEM. SCI., vol. 133, 2011, pages 9638 |
| J. AM. CHEM. SOC., vol. 130, 2008, pages 7812 |
| J. AM. CHEM. SOC., vol. 131, 2009, pages 15586 |
| J. AM. CHEM. SOC., vol. 131, 2009, pages 7792 |
| J. PHYS. CHEM. B, vol. 114, 2010, pages 3095 |
| J. POLY. CHEM. PART A., vol. 49, 2011, pages 4368 |
| J. POLYM. SCI. PART A: POLYM. CHEM., vol. 48, 2010, pages 614 |
| MACROMOLECULES, vol. 42, 2009, pages 2317 |
| MACROMOLECULES, vol. 42, 2009, pages 9217 |
| ORGANIC ELECTRONICS, vol. 10, 2009, pages 1541 |
| ORGANOMETALLICS, vol. 30, 2011, pages 3233 |
| See also references of EP2692760A4 |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012241017A (ja) * | 2011-05-13 | 2012-12-10 | Mitsubishi Chemicals Corp | ポリマー、有機半導体材料、並びにこれを用いた有機電子デバイス、光電変換素子及び太陽電池モジュール |
| JP2014531482A (ja) * | 2011-09-23 | 2014-11-27 | オーシャンズ キング ライティング サイエンス アンド テクノロジー シーオー.,エルティーディー | ジフルオロベンゾトリアゾリル有機物半導体材料、調合法、およびその使用方法 |
| JP2014531493A (ja) * | 2011-09-23 | 2014-11-27 | オーシャンズ キング ライティング サイエンスアンドテクノロジー カンパニー リミテッド | ジフルオロベンゾトリアゾリル太陽電池ポリマー材料、並びにその調製方法及び使用 |
| US9318707B2 (en) | 2011-11-15 | 2016-04-19 | Konica Minolta, Inc. | Organic photoelectric conversion element, and solar cell and optical sensor array each using same |
| WO2013073581A1 (ja) * | 2011-11-15 | 2013-05-23 | コニカミノルタ株式会社 | 有機光電変換素子、ならびにそれを用いた太陽電池及び光センサアレイ |
| JPWO2013073581A1 (ja) * | 2011-11-15 | 2015-04-02 | コニカミノルタ株式会社 | 有機光電変換素子、ならびにそれを用いた太陽電池及び光センサアレイ |
| JP2013220994A (ja) * | 2012-04-13 | 2013-10-28 | Sumitomo Chemical Co Ltd | 化合物及びそれを用いた電子素子 |
| CN104211917A (zh) * | 2013-05-29 | 2014-12-17 | 海洋王照明科技股份有限公司 | 含苯并三唑单元的聚合物及其制备方法和太阳能电池器件 |
| JP2015013989A (ja) * | 2013-06-05 | 2015-01-22 | 三菱化学株式会社 | コポリマー、半導体層形成用組成物、有機電子デバイス及び太陽電池モジュール |
| JP2016531171A (ja) * | 2013-07-15 | 2016-10-06 | エルジー・ケム・リミテッド | 共重合体およびこれを含む有機太陽電池 |
| US9748489B2 (en) | 2013-07-15 | 2017-08-29 | Lg Chem, Ltd. | Copolymer and organic solar cell comprising same |
| WO2015092909A1 (ja) * | 2013-12-19 | 2015-06-25 | リンテック株式会社 | 高分子化合物、有機光電変換素子、及び該素子の製造方法 |
| JPWO2015092909A1 (ja) * | 2013-12-19 | 2017-03-16 | リンテック株式会社 | 高分子化合物、有機光電変換素子、及び該素子の製造方法 |
| WO2015128891A1 (en) * | 2014-02-28 | 2015-09-03 | Eni S.P.A. | Copolymers containing naphthodithiophene units and diketopyrrolopyrrole units and process for their preparation |
| JP2016017117A (ja) * | 2014-07-07 | 2016-02-01 | 国立研究開発法人理化学研究所 | 高分子化合物、有機半導体材料、光電変換素子及びトランジスタ |
| WO2017047808A1 (ja) * | 2015-09-18 | 2017-03-23 | 三菱化学株式会社 | コポリマー、光電変換素子、太陽電池及び太陽電池モジュール |
| JP2019507215A (ja) * | 2016-01-27 | 2019-03-14 | エルジー・ケム・リミテッド | 共重合体およびこれを含む有機太陽電池 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20140166942A1 (en) | 2014-06-19 |
| EP2692760A4 (en) | 2014-09-24 |
| JP5779233B2 (ja) | 2015-09-16 |
| EP2692760A1 (en) | 2014-02-05 |
| JPWO2012133793A1 (ja) | 2014-07-28 |
| CN103443163A (zh) | 2013-12-11 |
| TW201245268A (en) | 2012-11-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5779233B2 (ja) | ブロック共重合体および光電変換素子 | |
| JP5779231B2 (ja) | π電子共役系ブロック共重合体および光電変換素子 | |
| JP5779234B2 (ja) | ブロック共重合体および光電変換素子 | |
| JP5810818B2 (ja) | 高分子化合物及びそれを用いた有機光電変換素子 | |
| JP5991324B2 (ja) | 高分子化合物及び有機光電変換素子 | |
| JP2013189602A (ja) | π電子共役重合体および光電変換素子 | |
| WO2012165420A1 (ja) | 有機半導体用組成物及びそれを用いた光電変換素子 | |
| JP5747789B2 (ja) | 高分子化合物及びそれを用いた有機光電変換素子 | |
| JP5834819B2 (ja) | 高分子化合物及びそれを用いた有機光電変換素子 | |
| JP5736456B2 (ja) | 共役ブロック共重合体及びそれを用いた光電変換素子 | |
| WO2014038526A1 (ja) | ブロック共重合体およびそれを用いた光電変換素子 | |
| JP2013221074A (ja) | π電子共役系ブロック共重合体および光電変換素子 | |
| WO2013035710A1 (ja) | 高分子化合物及び有機光電変換素子 | |
| JP2012151468A (ja) | 光電変換素子及びそれに用いられる組成物 | |
| JP2014019781A (ja) | 高分子化合物及びそれを用いた有機光電変換素子 | |
| JP2014114265A (ja) | ジチオフェン化合物及びそのジチオフェン基を有するπ電子共役重合体、並びにその重合体を用いた有機半導体デバイス | |
| JP2014051556A (ja) | π電子共役ランダム共重合体およびそれを用いた光電変換素子 | |
| JP2014234390A (ja) | 高い溶解性を示すπ電子共役共重合体 | |
| JP5874302B2 (ja) | 高分子化合物及びそれを用いた有機光電変換素子 | |
| JP2015017052A (ja) | オキシアルキレン基を含む置換基を有する複素芳香族化合物及び該化合物からなるポリマー | |
| JP2014051583A (ja) | 共役ブロック共重合体及びそれを用いた光電変換素子 | |
| JP2015078262A (ja) | π電子共役共重合体組成物およびそれを用いた光電変換素子 | |
| JP2014055197A (ja) | 共役ブロック共重合体及びそれを用いた光電変換素子 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12762740 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2013507799 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012762740 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14008654 Country of ref document: US |