WO2012132069A1 - 銀含有組成物用洗浄剤、銀含有組成物の除去方法、および銀の回収方法 - Google Patents
銀含有組成物用洗浄剤、銀含有組成物の除去方法、および銀の回収方法 Download PDFInfo
- Publication number
- WO2012132069A1 WO2012132069A1 PCT/JP2011/072629 JP2011072629W WO2012132069A1 WO 2012132069 A1 WO2012132069 A1 WO 2012132069A1 JP 2011072629 W JP2011072629 W JP 2011072629W WO 2012132069 A1 WO2012132069 A1 WO 2012132069A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- silver
- containing composition
- cleaning
- iron
- cleaning agent
- Prior art date
Links
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 title claims abstract description 120
- 229910052709 silver Inorganic materials 0.000 title claims abstract description 119
- 239000004332 silver Substances 0.000 title claims abstract description 119
- 239000000203 mixture Substances 0.000 title claims abstract description 62
- 239000012459 cleaning agent Substances 0.000 title claims abstract description 33
- 238000000034 method Methods 0.000 title claims description 49
- MVFCKEFYUDZOCX-UHFFFAOYSA-N iron(2+);dinitrate Chemical compound [Fe+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O MVFCKEFYUDZOCX-UHFFFAOYSA-N 0.000 claims abstract description 36
- 239000002245 particle Substances 0.000 claims abstract description 32
- FOIXSVOLVBLSDH-UHFFFAOYSA-N Silver ion Chemical compound [Ag+] FOIXSVOLVBLSDH-UHFFFAOYSA-N 0.000 claims abstract description 28
- 239000002105 nanoparticle Substances 0.000 claims abstract description 24
- 238000004140 cleaning Methods 0.000 claims description 66
- 238000007639 printing Methods 0.000 claims description 41
- VCJMYUPGQJHHFU-UHFFFAOYSA-N iron(3+);trinitrate Chemical group [Fe+3].[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O VCJMYUPGQJHHFU-UHFFFAOYSA-N 0.000 claims description 38
- 239000000243 solution Substances 0.000 claims description 36
- 239000007788 liquid Substances 0.000 claims description 33
- 239000007864 aqueous solution Substances 0.000 claims description 28
- 238000005406 washing Methods 0.000 claims description 24
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims description 20
- 239000000126 substance Substances 0.000 claims description 12
- 238000011084 recovery Methods 0.000 claims description 9
- 239000002699 waste material Substances 0.000 claims description 9
- 229910021607 Silver chloride Inorganic materials 0.000 claims description 8
- HKZLPVFGJNLROG-UHFFFAOYSA-M silver monochloride Chemical compound [Cl-].[Ag+] HKZLPVFGJNLROG-UHFFFAOYSA-M 0.000 claims description 8
- 239000002244 precipitate Substances 0.000 claims description 6
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 claims description 3
- 238000007774 anilox coating Methods 0.000 description 31
- 239000000976 ink Substances 0.000 description 24
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 24
- 238000011156 evaluation Methods 0.000 description 14
- 239000002904 solvent Substances 0.000 description 13
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 10
- 230000000694 effects Effects 0.000 description 10
- 239000011164 primary particle Substances 0.000 description 10
- 229910052742 iron Inorganic materials 0.000 description 9
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 238000004364 calculation method Methods 0.000 description 6
- 238000001035 drying Methods 0.000 description 6
- 229920000139 polyethylene terephthalate Polymers 0.000 description 6
- 239000005020 polyethylene terephthalate Substances 0.000 description 6
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 5
- 239000002253 acid Substances 0.000 description 5
- 238000011049 filling Methods 0.000 description 5
- 238000007646 gravure printing Methods 0.000 description 5
- 235000014413 iron hydroxide Nutrition 0.000 description 5
- NCNCGGDMXMBVIA-UHFFFAOYSA-L iron(ii) hydroxide Chemical compound [OH-].[OH-].[Fe+2] NCNCGGDMXMBVIA-UHFFFAOYSA-L 0.000 description 5
- 229910017604 nitric acid Inorganic materials 0.000 description 5
- 239000008399 tap water Substances 0.000 description 5
- 235000020679 tap water Nutrition 0.000 description 5
- -1 Polyethylene terephthalate Polymers 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000011148 porous material Substances 0.000 description 4
- 238000007650 screen-printing Methods 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- 229910001316 Ag alloy Inorganic materials 0.000 description 3
- 229910002554 Fe(NO3)3·9H2O Inorganic materials 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 3
- 239000002738 chelating agent Substances 0.000 description 3
- 238000004090 dissolution Methods 0.000 description 3
- 230000007613 environmental effect Effects 0.000 description 3
- SZQUEWJRBJDHSM-UHFFFAOYSA-N iron(3+);trinitrate;nonahydrate Chemical compound O.O.O.O.O.O.O.O.O.[Fe+3].[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O SZQUEWJRBJDHSM-UHFFFAOYSA-N 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 238000003917 TEM image Methods 0.000 description 2
- 239000004809 Teflon Substances 0.000 description 2
- 229920006362 Teflon® Polymers 0.000 description 2
- UMEAURNTRYCPNR-UHFFFAOYSA-N azane;iron(2+) Chemical compound N.[Fe+2] UMEAURNTRYCPNR-UHFFFAOYSA-N 0.000 description 2
- 230000005540 biological transmission Effects 0.000 description 2
- 238000004061 bleaching Methods 0.000 description 2
- 238000005524 ceramic coating Methods 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 239000008235 industrial water Substances 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- JVXHQHGWBAHSSF-UHFFFAOYSA-L 2-[2-[bis(carboxylatomethyl)amino]ethyl-(carboxylatomethyl)amino]acetate;hydron;iron(2+) Chemical compound [H+].[H+].[Fe+2].[O-]C(=O)CN(CC([O-])=O)CCN(CC([O-])=O)CC([O-])=O JVXHQHGWBAHSSF-UHFFFAOYSA-L 0.000 description 1
- XNCSCQSQSGDGES-UHFFFAOYSA-N 2-[2-[bis(carboxymethyl)amino]propyl-(carboxymethyl)amino]acetic acid Chemical compound OC(=O)CN(CC(O)=O)C(C)CN(CC(O)=O)CC(O)=O XNCSCQSQSGDGES-UHFFFAOYSA-N 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- VTLYFUHAOXGGBS-UHFFFAOYSA-N Fe3+ Chemical compound [Fe+3] VTLYFUHAOXGGBS-UHFFFAOYSA-N 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 230000001133 acceleration Effects 0.000 description 1
- IENXJNLJEDMNTE-UHFFFAOYSA-N acetic acid;ethane-1,2-diamine Chemical compound CC(O)=O.NCCN IENXJNLJEDMNTE-UHFFFAOYSA-N 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 239000007844 bleaching agent Substances 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 239000004020 conductor Substances 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 238000004851 dishwashing Methods 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 235000020188 drinking water Nutrition 0.000 description 1
- 239000003651 drinking water Substances 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 238000005530 etching Methods 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- MSNWSDPPULHLDL-UHFFFAOYSA-K ferric hydroxide Chemical compound [OH-].[OH-].[OH-].[Fe+3] MSNWSDPPULHLDL-UHFFFAOYSA-K 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 150000002506 iron compounds Chemical class 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000002082 metal nanoparticle Substances 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- YPJKMVATUPSWOH-UHFFFAOYSA-N nitrooxidanyl Chemical compound [O][N+]([O-])=O YPJKMVATUPSWOH-UHFFFAOYSA-N 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 239000005416 organic matter Substances 0.000 description 1
- 239000003002 pH adjusting agent Substances 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- 239000012466 permeate Substances 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 238000004064 recycling Methods 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 238000000518 rheometry Methods 0.000 description 1
- 230000001953 sensory effect Effects 0.000 description 1
- 229940100890 silver compound Drugs 0.000 description 1
- 150000003379 silver compounds Chemical class 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- GGCZERPQGJTIQP-UHFFFAOYSA-N sodium;9,10-dioxoanthracene-2-sulfonic acid Chemical compound [Na+].C1=CC=C2C(=O)C3=CC(S(=O)(=O)O)=CC=C3C(=O)C2=C1 GGCZERPQGJTIQP-UHFFFAOYSA-N 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D7/00—Compositions of detergents based essentially on non-surface-active compounds
- C11D7/02—Inorganic compounds
- C11D7/04—Water-soluble compounds
- C11D7/10—Salts
- C11D7/105—Nitrates; Nitrites
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D2111/00—Cleaning compositions characterised by the objects to be cleaned; Cleaning compositions characterised by non-standard cleaning or washing processes
- C11D2111/10—Objects to be cleaned
- C11D2111/14—Hard surfaces
- C11D2111/20—Industrial or commercial equipment, e.g. reactors, tubes or engines
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D2111/00—Cleaning compositions characterised by the objects to be cleaned; Cleaning compositions characterised by non-standard cleaning or washing processes
- C11D2111/10—Objects to be cleaned
- C11D2111/14—Hard surfaces
- C11D2111/22—Electronic devices, e.g. PCBs or semiconductors
Definitions
- the present invention relates to a cleaning agent for a silver-containing composition used when removing a silver component derived from a silver nanoparticle composition from a cleaning object to which a silver-containing composition derived from the silver nanoparticle-containing composition is attached,
- the present invention relates to a cleaning method using such a cleaning agent and a silver recovery method.
- Print electronics uses a composition in which the metal species and particle diameter are changed according to the object. It is known that silver particles having an average primary particle size of 100 nm or less (hereinafter also referred to as “silver nanoparticles”) are sintered at a low temperature and exhibit excellent conductivity. Therefore, a printing method using a silver nanoparticle-containing composition containing silver nanoparticles is adopted even on a substrate having low heat resistance such as PET (Polyethylene terephthalate) film and paper, which has been considered difficult conventionally. It is possible to form conductive wiring.
- PET Polyethylene terephthalate
- the transition from solvent-based printing inks that use organic solvents that were originally used to so-called aqueous printing inks that use solvents centered on water has been promoted. It has become like this.
- the solvent is mainly an organic solvent, but dispersed in water or the like.
- the silver nanoparticle containing composition which uses the silver nanoparticle which makes water as a solvent is examined (for example, refer patent document 1 or 2).
- a printed matter can be obtained from a silver nanoparticle-containing composition typified by an aqueous ink containing silver nanoparticles as described in Patent Document 1 and the like.
- the aqueous ink remaining in the member used for the printing may have a component containing silver nanoparticles strongly adhered to the member used for the printing due to drying of the solvent or the like. It has been found that such strongly adhered silver nanoparticle-derived components are difficult to remove even when using solvents or diluents used in water-based inks or commercially available cleaning liquids.
- an anilox roll for flexographic printing, a flexographic plate, a gravure plate, a screen plate, and the like may use a porous member.
- a porous member In this case, once the silver nanoparticles permeate the fine pores and adhere strongly, Removal by washing becomes very difficult.
- the silver particles having a relatively large particle diameter have such fine pores even if the paste is left on the plate. There is little room for entry into the portion, and removal was relatively easy if a cleaning operation was performed using a dedicated cleaning agent or brush.
- a cleaning operation was performed using a dedicated cleaning agent or brush.
- the ink using silver particles with a particle size suitable for printed electronics which is nano-order
- the fine pores formed in the plate or anilox roll Silver nanoparticles reach easily. Therefore, the solvent is dried after being left for a short time, and the component containing the silver nanoparticles is strongly attached. Therefore, it is difficult to remove such fine holes with a normal cleaning agent or brush.
- the materials such as printing members constituting the printing apparatus are limited to those having high acid resistance, and if the cleaning after the removal is not sufficiently performed, there is a possibility that the next printing operation may be affected. There is a problem that there is. Furthermore, depending on the acid used, a gas having a bad odor or corrosiveness may be generated, which may not be suitable in terms of work.
- bleaching mainly composed of a chelating agent such as EDTA (ethylenediamine acetic acid) iron ammonium or PDTA (propandeminetic acetic acid) iron ammonium used for silver salt photography and the like. It is also conceivable to use a solution (also referred to as a bleach solution), a bleach-fixing solution (also referred to as a bleach-fix solution) or an etching solution.
- a chelating agent such as EDTA (ethylenediamine acetic acid) iron ammonium or PDTA (propandeminetic acetic acid) iron ammonium used for silver salt photography and the like.
- a solution also referred to as a bleach solution
- a bleach-fixing solution also referred to as a bleach-fix solution
- an etching solution an etching solution.
- the chelating agents themselves used in these productions are inherently expensive and can be used for photographic development, etc., which requires a small amount, but they can be used in large articles such
- the above-mentioned bleaching component mainly composed of EDTA iron ammonium is mainly composed of organic matter. Although such a component is stable as a substance, it is difficult to use it in a large amount in consideration of an adverse effect on the environment at the time of disposal.
- An object of the present invention is a silver-containing composition derived from silver nanoparticles that is excellent in workability, has a low environmental load, and can easily remove components containing silver nanoparticles attached to an object to be cleaned.
- a cleaning agent and a method for removing a silver-containing composition using the cleaning agent are provided, and a silver recovery method for separating and recovering silver from the cleaning waste liquid is also provided.
- the cleaning agent for the composition derived from silver nanoparticles according to the present invention removes the silver component when the silver-containing composition derived from silver nanoparticles having an average particle diameter of 1 to 100 nm adheres to the object to be cleaned. And contains an aqueous iron nitrate solution.
- the silver nanoparticles that are the basis of the silver-containing composition to be acted are, for example, in the form of ink or paste, and are silver particles or silver alloy particles having an average particle diameter of 1 to 100 nm.
- a printing plate or printing member can be used for the object to be cleaned.
- the member in which the cleaning agent according to the present invention is used more effectively include a printing member having fine holes such as an anilox roll for flexographic printing, a flexographic plate, a gravure plate, and a screen plate.
- iron nitrate valence of iron is trivalent (III) (Formula Fe (NO 3) 3) and more preferable to be.
- the method for removing a silver-containing composition derived from silver nanoparticles removes a silver component when a composition derived from silver nanoparticles having an average particle diameter of 1 to 100 nm adheres to an object to be cleaned.
- a method for removing a silver-containing composition is provided, which includes a step of removing a silver-containing composition adhering to an object to be cleaned using a cleaning liquid containing an aqueous iron nitrate solution.
- a silver recovery method that further includes a step of recovering silver by adding chloride from the waste liquid generated by the above removal method is adopted.
- the cleaning performed using the cleaning agent according to the present invention is easily separated from the cleaning product by dissolution of the silver-containing composition adhering to the object to be cleaned, in particular the silver-containing composition penetrating fine details. It is possible. In addition, since no organic substance such as a chelating agent is used, the environmental load is reduced, and since the acid itself is not used, the workability is excellent.
- the content of iron nitrate contained in the aqueous iron nitrate solution used in the present invention is 50% by mass or less, preferably 1 to 50% by mass, more preferably 5 to 50% by mass, and still more preferably 10 to 50% by mass. It is. In order to obtain higher removal capability, the content of iron nitrate may be 20 to 50% by mass. If it exceeds 50% by mass, iron nitrate may be precipitated depending on the liquid temperature, which is not preferable. Here, when the liquid temperature is raised, precipitation can be suppressed, but insoluble iron compounds such as iron oxide and iron hydroxide are easily generated in the aqueous solution.
- the aqueous solution when the aqueous solution is stored at room temperature, it is appropriate to set it to 50% by mass or less.
- the content ratio of iron nitrate when it is less than 1% by mass, although there is a cleaning effect, a long time is required until the action appears.
- iron (III) nitrate also known as ferric nitrate
- Fe (NO 3 ) 3 is particularly preferable
- Fe (NO 3 ) is particularly preferable.
- solvent The solvent for dissolving iron nitrate is effective if the main component (mixing ratio is half or more) is water.
- a solvent for dissolving these components for example, an alcohol or a polyol, may be added for the purpose of enhancing the cleaning effect.
- the boiling point of these solvents is higher than that of water, the drying rate of the aqueous solution can be reduced, and the drying of the solution on the printing member can be controlled, so that the cleaning effect can be further enhanced.
- the water used for the solvent is preferably distilled water or ion exchange water. If chlorine remains in the liquid, care must be taken because the silver once eluted may form insoluble silver chloride. However, since there is no problem in actual use as long as the chlorine concentration is in an appropriate range as drinking water, there is no problem in the use of general industrial water or tap water.
- deaeration treatment is performed to remove oxygen in the liquid.
- nitrogen which is an inert gas, is circulated to remove oxygen in the liquid.
- An iron nitrate aqueous solution is prepared by dissolving the iron nitrate in a solvent and, if necessary, adding additional components. After adjustment, in order to suppress the precipitation of insoluble oxides and hydroxides, it is appropriate to store in a cool and dark place, but even if some insoluble precipitates are generated, the cleaning effect is not greatly affected. .
- the iron nitrate aqueous solution prepared in this way is suppressed in volatility, it is excellent in improving the use environment during use.
- the corrosiveness is low even if the object to be cleaned is a metal, which contributes to the extension of the life of printing members and the like.
- washing method When the object to be cleaned is a relatively small member, a method of immersing the member as it is in a cleaning liquid as a cleaning agent and then cleaning with a take-out brush is recommended. In some cases, an ultrasonic wave may be applied from the outside of the cleaning liquid container to remove the silver-containing composition present in the details. After washing with a brush, wash thoroughly with water. If the aqueous iron nitrate solution is washed before drying, the cleaning effect can be further enhanced. In order to increase the cleaning efficiency, cleaning with a brush may be performed while washing with water. Of course, it may be washed while applying ultrasonic waves during washing with water.
- the conditions for the washing and washing treatment can be appropriately adjusted according to factors such as the contamination status, size and shape of the member.
- the content ratio of iron nitrate is high, and specifically, it is preferably about 25% by mass or more.
- the content rate of iron nitrate should be about 25% by mass or less depending on the size of the member and the cleaning time, such as being immersed in a cleaning solution.
- the temperature can be adjusted.
- the silver-containing composition to be removed by washing that is, the silver-containing composition derived from the silver nanoparticle-containing composition is composed of particles of a single silver or silver alloy having an average primary particle diameter of 1 to 100 nm.
- What is supplied in the form of a configured ink or paste is metallized.
- a paste or ink having metal nanoparticles having such an average particle diameter range as a constituent component is effective for forming a conductive material because the formed wiring is metallized at a low temperature.
- the paste penetrates into the details of the printing member and becomes metalized during drying, making it difficult to remove.
- the particle diameter of such particles is confirmed by the following method, for example.
- the average primary particle diameter which is an average value of primary particle diameters from a TEM (Transmission Electron Microscope) image
- 2 parts by mass of the silver nanoparticle composition was added to a mixed solution of 96 parts by mass of cyclohexane and 2 parts by mass of oleic acid, and dispersed by ultrasonic waves.
- the dispersion solution was dropped on a Cu microgrid with a support film and dried to obtain a TEM sample.
- the created microgrid was observed using a transmission electron microscope (JEM-100CXMark-II type manufactured by JEOL Ltd.) at an acceleration voltage of 100 kV and an image of particles observed in a bright field at a magnification of 300,000 times. .
- image analysis software (A Image-kun (registered trademark) manufactured by Asahi Kasei Engineering Co., Ltd.) was used. This image analysis software identifies individual particles based on color shading. For a 300,000-fold TEM image, the “particle brightness” is “dark”, the “noise removal filter” is “present”, “ The circular particle analysis is performed under the conditions of “20” for the “round threshold” and “50” for the “overlap degree”, the primary particles are measured for 200 or more particles, and the number average diameter is obtained. It was. In addition, when there were many condensed particles and irregular shaped particles in the TEM image, it was determined that measurement was impossible.
- silver or silver alloy particles exceeding 100 nm (hereinafter also simply referred to as “silver powder”) can be added for the purpose of adjusting rheology.
- silver powder can be added for the purpose of adjusting rheology.
- the addition amount is too large, the low-temperature sinterability is impaired, so silver nanoparticles need to be the main component.
- This main component means that the silver nanoparticles are 50% or more by mass ratio of the metal component.
- composition containing the above-mentioned silver nanoparticles can be made excellent in the natural environment and the working environment by mainly using water.
- composition containing silver nanoparticles may contain additives such as a pH adjuster and a viscosity adjuster in a timely manner according to the printing method.
- the waste liquid generated in the present invention that is, the cleaning liquid after cleaning with the above-described cleaning agent for the silver-containing composition is composed of iron, silver, nitrate radical, and water, and the components are clarified. . Since methods for recovering iron and silver from the liquid have been established, it is also preferable to separate and recover the silver in the liquid and reuse it in combination with these methods.
- iron ions may be precipitated as iron oxide or iron hydroxide.
- the aqueous solution after dissolution of silver is filtered once to remove iron oxide or iron hydroxide, and then a substance having chloride ions (hydrochloric acid, NaCl aqueous solution, KCl aqueous solution, etc.) is added to the obtained aqueous solution.
- a substance having chloride ions hydroochloric acid, NaCl aqueous solution, KCl aqueous solution, etc.
- recovering iron as a hydroxide can be illustrated. It is a feature of the present invention that silver recovery and waste liquid treatment are easy.
- a specific example of silver recovery is as follows.
- An iron (III) nitrate aqueous solution is applied to a Teflon (registered trademark) filter having an aperture ratio of 0.1 ⁇ m to remove foreign matters in the liquid.
- Teflon registered trademark
- the silver dissolves, and depending on the solution environment, the iron ions precipitate as iron oxide or iron hydroxide instead of dissolving the silver. Therefore, first, these foreign substances are removed by a filter.
- a 3.3 mol / l KCl aqueous solution is added to the aqueous solution from which foreign substances have been removed by filtering.
- the addition amount is the same mol amount as iron (III) in the aqueous solution. If the dissolution amount of silver can be estimated, about 10 times the estimated silver amount is added. The precipitate that forms is silver chloride. (Iii) After the addition, the mixture is stirred for several minutes, and silver chloride is recovered with a Teflon filter having an aperture ratio of 0.1 ⁇ m. After recovery, the amount of silver is calculated from the weight of silver chloride. When NaOH is added to the filtrate, iron (III) hydroxide precipitates, so that the iron component can be easily recovered from the solution.
- both silver and iron components can be recovered. If the recovered iron is dissolved in nitric acid, it can be reused in the cleaning method.
- the cell volume of the anilox roll is measured with a laser microscope. This measurement result is defined as the volume after filling of the anilox roll ink.
- Volume after washing After washing by a predetermined method, the anilox roll is sufficiently dried, and after confirming that the anilox roll is in the range of 20 to 25 ° C., the cell volume of the anilox roll is measured with a laser microscope. This measurement result is taken as the volume after washing of the anilox roll.
- the removal rate is defined as follows.
- Removal rate (%) [ ⁇ (Initial volume ⁇ volume after filling) ⁇ (initial volume ⁇ volume after washing) ⁇ / (initial volume ⁇ volume after filling)] ⁇ 100
- the case where the silver removal rate by washing is 100 to 90% is “good”, the case where it is 90 to 80% is “normal”, and the case where it is less than 80% is “inferior”.
- the gravure printing can be exemplified by the following method, and the following method can be used for evaluating the cleaning state of the gravure plate which is one of the members.
- the gravure ink As the gravure ink, PGI-700 type manufactured by PChem Associates, Inc., silver concentration of about 60% by mass was used.
- the gravure cell volume after nitric acid cleaning is the “initial volume”
- the volume after drying after printing is “the volume after filling”
- the volume after the cleaning is “washing”, as described above. Calculation was performed using a similar evaluation apparatus and equation. Similarly, at this time, the case where the removal rate of silver by washing is 100 to 90% is “good”, the case where it is 90 to 80% is “normal”, and the case where it is less than 80% is “poor”.
- the screen printing can be exemplified by the following method, and the following method can be used for evaluating the cleaning condition of the screen plate which is one of the members.
- Screen ink As a screen ink, PSI-111 type manufactured by PChem Associates, Inc., silver concentration of about 40% by mass was used.
- Example 1 Iron (III) nitrate nonahydrate (chemical formula: Fe (NO 3) 3 ⁇ 9H 2 O) was dissolved in tap water, to create the iron (III) nitrate 50 wt% of iron (III) nitrate solution in terms of A “cleaning solution” was obtained. The aqueous solution at this time was confirmed to have a slight odor due to nitric acid (the evaluation at this time was “normal”).
- a composition containing silver nanoparticles (manufactured by PChem: PFI-700 type, silver concentration 60 mass%, average particle size (average primary particle size) 22 nm) is applied to a flexographic printing machine (anilox roll is 2.6 BCM, or 13 .5BCM, each having a ceramic coating) and flexographically printed. Then, after the printing operation was suspended for 24 hours, the above-described cleaning liquid was applied to the roll using a brush, and the roll was washed with water while being rubbed with a brush after 10 minutes. Applying the cleaning liquid to the roll using a brush is a cleaning method that assumes a large printing press that is difficult to remove. Then, when the cleaning property was evaluated by the method using the above-mentioned digital microscope, the silver nanoparticle-containing composition was completely removed in both cases of 2.6 BCM and 13.5 BCM. It was an evaluation.
- Example 2 The evaluation of Example 1 was repeated except that a 25 mass% iron nitrate (III) aqueous solution in terms of iron nitrate (III) was prepared. A slight odor was confirmed from this solution (the evaluation at this time is “normal”). The detergency was evaluated as “good” because the silver nanoparticle-containing composition was completely removed in both cases of 2.6 BCM and 13.5 BCM.
- Example 3 A 10 mass% iron nitrate (III) aqueous solution in terms of iron nitrate (III) was prepared to obtain a cleaning liquid. The solution at this time confirmed a slight odor. Then, a silver nanoparticle-containing composition (PChem: PFI-700 type, silver concentration 60% by mass) is used with a flexographic printing machine (anilox roll is 2.6 BCM or 13.5 BCM, respectively, and the surface is Flexographic printing was performed using a ceramic coating. Then, after stopping the printing operation for 24 hours, the anilox roll was immersed in the cleaning liquid. After 6 hours, it was removed from the cleaning solution and washed with water while rubbing with a brush. This cleaning method assumes cleaning of a small and disassembled printing press. The detergency was evaluated as “good” because the silver nanoparticle-containing composition was almost completely removed in both cases of 2.6 BCM and 13.5 BCM.
- the iron nitrate (III) aqueous solution is divided into two parts, one is quantified by ICP emission analysis and the other is quantified by the technique of measuring the weight of silver chloride obtained by the silver recovery process described above. Confirmed the effectiveness of the process.
- the silver concentration in the iron (III) nitrate aqueous solution obtained by ICP emission analysis is 269 ppm, and the silver concentration obtained from the silver chloride gravimetric method is almost equal to 271 ppm. According to the above recovery process, It was shown that almost all silver can be recovered.
- Example 4 The evaluation of Example 2 was repeated in the same manner except that the concentration of iron (III) nitrate in the cleaning liquid was changed to 5% (Example 4) and 1% (Example 5), respectively. Evaluation by odor and evaluation of detergency were performed in the same manner as in Example 1. In Examples 4 and 5, no odor was observed. When the iron (III) nitrate concentration is 5%, the removal rate after 6 hours is 2.6 BCM and 13.5 BCM, and the silver nanoparticle-containing composition is almost completely removed and is “good”. It was an evaluation. The obtained results are also shown in Table 1.
- Iron (III) nitrate nonahydrate (chemical formula: Fe (NO 3) 3 ⁇ 9H 2 O) was dissolved in tap water, to create the iron (III) nitrate 10 wt% of iron (III) nitrate solution in terms of A “cleaning solution” was obtained. The solution at this time confirmed a slight odor. Then, a silver nanoparticle composition (manufactured by PChem: PGI-700 type, silver concentration 60% by mass, average particle size (average primary particle size) 24 nm) is applied to a gravure printing machine (a gravure plate has 300 and 100 lines). Were used, and gravure printing was performed.
- PChem PGI-700 type, silver concentration 60% by mass, average particle size (average primary particle size) 24 nm
- the gravure plate was immersed in a container filled with the cleaning agent. After 6 hours, it was removed from the cleaning solution and washed with water while rubbing with a brush. When the detergency was evaluated, the silver nanoparticle-containing composition was almost completely removed and evaluated as “good” in either case of 300 wires or 100 wires.
- Example 7 Iron (III) nitrate nonahydrate (chemical formula: Fe (NO 3) 3 ⁇ 9H 2 O) was dissolved in tap water, to create the iron (III) nitrate 10 wt% of iron (III) nitrate solution in terms of A “cleaning solution” was obtained. The solution at this time confirmed a slight odor. Then, a silver nanoparticle composition (manufactured by PChem: PSI-111 type, silver concentration 40 mass%, average particle size (average primary particle size) 21 nm) is applied to a screen printing machine (screen plate: PET mesh, 420 lines / inch). And a screen diameter of 23 ⁇ m). After printing, excess ink was removed with a rubber spatula.
- PChem PSI-111 type, silver concentration 40 mass%, average particle size (average primary particle size) 21 nm
- the screen plate was immersed in a container filled with the cleaning agent. After 6 hours, it was removed from the cleaning solution and washed with water while rubbing with a brush. When the detergency was evaluated, the silver nanoparticle-containing composition was almost completely removed and was evaluated as “good”.
- Example 1 Ceram Clean II manufactured by HARPER, which is a ceramic abrasive-containing cleaning agent that is usually used as a physical cleaning agent for anilox roll, was used instead of the aqueous iron (III) nitrate solution.
- Example 1 was repeated, except that the cleaning solution was soaked in the waste and rubbed for 2 minutes for cleaning. In the evaluation of 2.6 BCM, the removal efficiency was about 83%, but 13.5 BCM could not be removed at all. In the case of 13.5 BCM, it seems that the cleaning effect could not be obtained because the capacity was larger and the cell was deeper. Moreover, when Example 1 was repeated except having changed the said washing
- Example 2 (Comparative Example 2) In Example 1, a glycol ether type cleaning agent (ANILOX ROLL CLEANER (manufactured by Anilox Roll Services)), which is usually used as a chemical cleaning agent for anilox roll, was used instead of the iron (III) nitrate aqueous solution. Example 1 was repeated except that the cleaning agent was soaked in a sponge and washed by rubbing for 2 minutes. The removal rate was 40% or less in both cases of 2.6 BCM and 13.5 BCM. Moreover, when Example 1 was repeated except having changed the said washing
- ANILOX ROLL CLEANER manufactured by Anilox Roll Services
- Example 3 (Comparative Example 3)
- the product name Palmolive manufactured by Colgate-Palmolive Company was used as a commercially available dishwashing detergent instead of the iron (III) nitrate aqueous solution.
- Example 1 was repeated except that the cleaning agent was soaked in a sponge sufficiently containing water and washed by rubbing for 2 minutes. The removal rate was 40% or less in both cases of 2.6 BCM and 13.5 BCM. Moreover, when Example 1 was repeated except having changed the said washing
- Example 3 was repeated in the same manner except that nitric acid (10%) was used instead of iron nitrate (III).
- the evaluation results are also shown in Table 1. Although it exhibits high detergency, it was confirmed that it had a strong odor and was difficult to use continuously.
Landscapes
- Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Detergent Compositions (AREA)
- Inking, Control Or Cleaning Of Printing Machines (AREA)
- Cleaning By Liquid Or Steam (AREA)
Abstract
Description
本発明に使用される硝酸鉄水溶液に含まれる硝酸鉄の含有割合は50質量%以下、好ましくは1~50質量%、更に好ましくは5~50質量%であり、一層好ましくは10~50質量%である。より高い除去能力を得るためには、硝酸鉄の含有割合を20~50質量%とすればよい。50質量%を超える場合には、液温によっては硝酸鉄が析出するおそれがあり好ましくない。ここで、液温を高めると析出を抑制できるが、液中で酸化鉄や水酸化鉄など不溶性の鉄化合物が水溶液中で生成しやすくなる。したがって、室温で水溶液を保存する場合には、50質量%以下とすることが適切である。一方、硝酸鉄の含有割合が少なすぎる場合、例えば、1質量%未満である場合には、洗浄効果はあるものの、その作用が現れるまでに長い時間が必要となる。
そして、硝酸鉄を溶解する溶媒は、主成分(混合割合が半分以上)を水とすれば効果的である。ただし、銀ナノ粒子組成物内に樹脂やバインダを含んでいた場合はより洗浄効果を高める目的でそれらの成分を溶解するための溶剤、例えばアルコールやポリオールを添加しても構わない。また、これら溶剤の沸点が水より高い場合は、水溶液の乾燥速度を遅くでき、印刷部材での溶液の乾燥などを制御できることから、より洗浄効果を高めることができる。
上記硝酸鉄を溶媒に溶解、さらに必要であれば添加成分を加えて、硝酸鉄水溶液を調製する。調整後は、不溶性の酸化物および水酸化物の析出を抑制する目的で、冷暗所で保管する方が適当であるが、多少の不溶性の析出物が発生しても、洗浄効果に大きな影響はない。
(洗浄方法)
洗浄対象物が比較的小物の部材である場合には、洗浄剤としての洗浄液に対してそのまま部材を浸漬して、その後取り出しブラシで洗浄する方法が推奨される。場合によっては、洗浄液の容器の外部から超音波をあてて、細部に存在する銀含有組成物を除去するようにしても構わない。ブラシで洗浄した後に、十分に水洗を実施する。硝酸鉄水溶液が乾燥するよりも前に水洗すれば、より洗浄効果を高めることができる。洗浄効率を高めるため、ブラシによる洗浄を水洗中に行っても構わない。当然、水洗の際にも超音波を与えながら洗浄しても構わない。
本発明において洗浄による除去対象となる銀含有組成物、すなわち、銀ナノ粒子含有組成物に由来する銀含有組成物は、平均一次粒子径が1~100nmである銀単体もしくは銀合金からなる粒子から構成されるインクもしくはペーストの形態により供給されるものが金属化したものである。このような平均粒子径の範囲を有する金属ナノ粒子を構成成分に有するペーストやインクは、形成される配線は低温で金属化するため、導電性材料の形成に効果的ではあるが、そのインクやペーストは印刷部材の細部にまで浸透し、乾燥時に金属化してしまい除去が難しくなる。こうした粒子の粒子径はたとえば、下記のような手法で確認する。
本明細書で銀ナノ粒子の直径をいう場合は、TEM(Transmission Electron Microscope)像からの一次粒子径の平均値である一次粒子平均径をいい、以下のようにして測定した。銀ナノ粒子組成物2質量部をシクロヘキサン96質量部とオレイン酸2質量部との混合溶液に添加し、超音波によって分散させた。分散溶液を支持膜付きCuマイクログリッドに滴下し、乾燥させることでTEM試料とした。作成したマイクログリッドを透過型電子顕微鏡(日本電子株式会社製JEM-100CXMark-II型)を使用し、100kVの加速電圧で、明視野で粒子を観察した像を、倍率300,000倍で撮影した。
本発明で発生する廃液、すなわち、銀含有組成物に対して上述した洗浄剤による洗浄後の洗浄液については、鉄、銀、硝酸根、水からなるものであり、含有成分が明確化されている。鉄や銀に関しては液中から回収するための手法も確立されていることから、これらの方法と組み合わせることによって、液中の銀を分離回収し、再利用することも好ましい構成である。
(i)硝酸鉄(III)水溶液を開口率0.1μmのテフロン(登録商標)フィルタにかけ、液中の異物を取り除く。硝酸鉄(III)水溶液に銀および銀化合物を浸漬すると銀は溶解し、溶液環境によっては銀が溶解する代わりに、鉄イオンが酸化鉄もしくは水酸化鉄として析出する。よってまず、これらの異物をフィルタにより取り除く。
(ii)フィルタリングにより異物を取り除いた水溶液に3.3mol/l濃度のKCl水溶液を添加する。添加量は、水溶液の鉄(III)と同じmol量である。仮に銀の溶解量が推定できる場合は、推定銀量の10倍程度添加する。生成する沈殿物は塩化銀である。
(iii)添加後、数分攪拌し、開口率0.1μmのテフロンフィルターで塩化銀を回収する。回収後塩化銀の重量から銀量を算出する。なお、ろ液にNaOHを添加すると、水酸化鉄(III)が沈殿するので、溶液中から鉄成分の回収が容易となる。
除去操作前後のアニロックスロール等の被洗浄部材における洗浄効果は、デジタルマイクロスコープを用いて溝の中に存在する銀の状態を確認し、銀の除去が十分になされているか否かについて確認した。
フレキソ印刷方法としては、以下に示す方法が例示でき、またその部材の一つであるアニロックスロールの洗浄状況の評価は、下記に示す方法が利用できる。
(フレキソインク)
フレキソインクとしては、PChem Associates, Inc社製PFI-700型、銀濃度約60質量%を用いた。
フレキソプルーフ(製造元:RK Print Coat Instruments、型式:ESI12、アニロックスロール:容量2.6BCM(600線)、13.5BCM(120線))を使用した。
フレキソプルーフの設定として、アニロックスロールとゴム版の圧力調整を行う。圧力調整は、両端の調整用つまみを用いてアニロックスロールとゴム版がちょうど接触する位置からさらに、0.05~0.10mm押込む。次にアニロックスロール上に組成物を約1ml滴下し、約1秒でPET(ポリエチレンテレフタレート)フィルム(デュポンテイジンフイルム社製のメリネックス(Melinex:登録商標)545)上に塗布を行った。
(初期容積)
アニロックスロールを30質量%の硝酸水溶液に30分浸漬後、十分に水洗、乾燥を行う。アニロックスロールが、20~25℃の範囲にあることを確認後、キーエンス社製レーザー顕微鏡VK9700を使ってアニロックスロールのセルの容積の測定を実施する。この測定結果をアニロックスロールの初期容積とする。単位は、BCM(平方インチ当り10億立方ミクロン)である。
(充填後容積)
上述のフレキソ印刷手順で印刷を実施後、アニロックスロールを室温で24時間乾燥させる。アニロックスロールが、20~25℃の範囲にあることを確認後、アニロックスロールのセル容積をレーザー顕微鏡にて測定する。この測定結果をアニロックスロールのインクの充填後容積とする。
(洗浄後容積)
所定の方法で洗浄した後、アニロックスロールを十分乾燥させ、アニロックスロールが、20~25℃の範囲にあることを確認後、アニロックスロールのセル容積をレーザー顕微鏡にて測定する。この測定結果をアニロックスロールの洗浄後容積とする。
(除去率)
除去率は以下の様に定義する。
除去率(%)=
[{(初期容積-充填後容積)-(初期容積-洗浄後容積)}/(初期容積-充填後容積)]×100
このとき、洗浄により銀の除去率が100~90%である場合を「良い」、90~80%の場合を「普通」、80%未満であれば「劣る」としている。
グラビア印刷としては、以下に示す方法が例示でき、またその部材の一つであるグラビア版の洗浄状況の評価は、以下に示す方法が利用できる。
(グラビアインク)
グラビアインクとしては、PChem Associates, Inc社製PGI-700型、銀濃度約60質量%を用いた。
KPrintingプルーファー(製造元:RK Print Coat Instruments、グラビア版:300線数、100線数)を使用した。
除去率の算出については、上述のものと同じく、硝酸洗浄後のグラビア版のセル容積を「初期容積」、印刷後に乾燥させた後の容積を「充填後容積」、洗浄後の容積を「洗浄後容積」とし、同様の評価装置および式を使って算出を行った。同様にこのとき、洗浄により銀の除去率が100~90%である場合を「良い」、90~80%の場合を「普通」、80%未満であれば「劣る」としている。
スクリーン印刷としては、以下に示す方法が例示でき、またその部材の一つであるスクリーン版の洗浄状況の評価は、以下に示す方法が利用できる。
(スクリーンインク)
スクリーンインクとしては、PChem Associates, Inc社製PSI-111型、銀濃度約40質量%を用いた。
スクリーン印刷機(製造元:C.W.プライス社、型式:224型、スクリーン:PETメッシュ、420線/インチ、線径23μm)を用いた。
上述のインクおよび印刷条件で印刷後、不要なインクをゴムのスキージで除去後、24時間乾燥した。その後、マイクロスコープでメッシュを観察するとメッシュの孔はほぼ100%インクで詰まっていた。このスクリーン版を硝酸鉄(III)水溶液で洗浄後に同様にマイクロスコープで観察し、5mm角内の完全に洗浄されているメッシュの孔の個数/5mm角内の全メッシュの孔個数を除去率とした。このとき、洗浄により銀の除去率が100~90%である場合を「良い」、90~80%の場合を「普通」、80%未満であれば「劣る」としている。
洗浄液を調製した後に、官能法により液の臭気を確認した。判断基準としては、臭気が確認できない場合を「大変良い」、僅かに臭気が確認される場合を「良い」、若干臭気が確認される場合を「普通」、明らかに臭気があることが確認される場合を「劣る」として評価した。
硝酸鉄(III)九水和物(化学式:Fe(NO3)3・9H2O)を水道水に溶解し、硝酸鉄(III)換算で50質量%硝酸鉄(III)水溶液を作成し、「洗浄液」を得た。この時の水溶液は、僅かに硝酸起因の臭気が確認された(このときの評価は「普通」となる)。そして、銀ナノ粒子含有組成物(PChem社製:PFI-700型、銀濃度60質量%、平均粒子径(平均一次粒径)22nm)を、フレキソ印刷機(アニロックスロールは2.6BCM、もしくは13.5BCMのものをそれぞれ用いて、表面はセラミックコーティングされている)を用いて、フレキソ印刷を施した。そして、印刷操作を24時間休止後、刷毛を用いてロールに上記洗浄液の塗布を行い、ロールを10分後にブラシで擦りながら水洗を実施した。刷毛を用いてロールに洗浄液を塗布するのは、取り外しが困難な大型の印刷機を想定した洗浄方法である。そして、洗浄性の評価を上述のデジタルマイクロスコープを用いた方法により行ったところ、2.6BCM、13.5BCMのいずれの場合でも、銀ナノ粒子含有組成物は、完全に除去されており「良い」の評価となるものであった。
硝酸鉄(III)換算で25質量%の硝酸鉄(III)水溶液を調整する以外は、実施例1の評価を繰り返した。この溶液からは僅かに臭気を確認した(このときの評価は「普通」となる)。洗浄性は2.6BCM、13.5BCMのいずれの場合でも、銀ナノ粒子含有組成物は、完全に除去されており「良い」の評価となるものであった。
硝酸鉄(III)換算で10質量%の硝酸鉄(III)水溶液を作成し、洗浄液を得た。この時の溶液は、僅かに臭気を確認した。そして、銀ナノ粒子含有組成物(PChem社製:PFI-700型、銀濃度60質量%)を、フレキソ印刷機(アニロックスロールは2.6BCM、もしくは13.5BCMのものをそれぞれ用いて、表面はセラミックコーティングされている)を用いて、フレキソ印刷を施した。そして、印刷操作を24時間休止後、アニロックスロールを洗浄液に浸漬させた。6時間後に洗浄液から取り出しブラシで擦りながら水洗を実施した。この洗浄方法は小型で分解可能な印刷機の洗浄を想定したものである。洗浄性は2.6BCM、13.5BCMのいずれの場合でも、銀ナノ粒子含有組成物は、ほぼ完全に除去されており「良い」の評価となるものであった。
洗浄液中の硝酸鉄(III)濃度を5%(実施例4)、1%(実施例5)にそれぞれ変化させた以外は同様にして実施例2の評価を繰り返した。臭気による評価、および洗浄性の評価を実施例1と同様に行った。実施例4、5ともに臭気は確認されなかった。硝酸鉄(III)濃度が5%の場合は6時間での除去率は2.6BCM、13.5BCMのいずれの場合でも、銀ナノ粒子含有組成物は、ほぼ完全に除去されており「良い」の評価となるものであった。得られた結果について、表1に併せて示す。
硝酸鉄(III)濃度が1%の場合は6時間での除去率が、2.6BCM、13.5BCMのいずれの場合でも、除去率は60%以下であり洗浄性が不十分であった。しかし、浸漬後24時間についても洗浄性を評価すると、2.6BCMが「良い」で、13.5BCMが「普通」と、適切な洗浄性を有することを確認した。
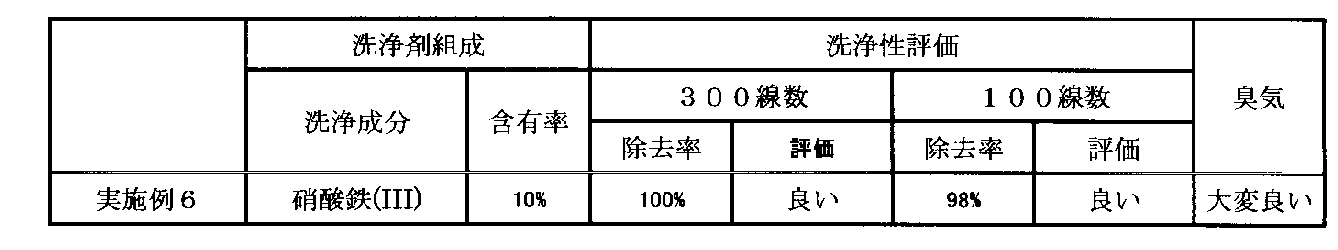
硝酸鉄(III)九水和物(化学式:Fe(NO3)3・9H2O)を水道水に溶解し、硝酸鉄(III)換算で10質量%硝酸鉄(III)水溶液を作成し、「洗浄液」を得た。この時の溶液は、僅かに臭気を確認した。そして、銀ナノ粒子組成物(PChem社製:PGI-700型、銀濃度60質量%、平均粒子径(平均一次粒径)24nm)を、グラビア印刷機(グラビア版は300線数と100線数のものをそれぞれ用いる。)を用いて、グラビア印刷を施した。そして、印刷操作を24時間休止後、上記洗浄剤を満たした容器内にグラビア版を浸漬された。6時間後に洗浄液から取り出しブラシで擦りながら水洗を実施した。洗浄性を評価すると、300線数、100線数のいずれの場合でも、銀ナノ粒子含有組成物は、ほぼ完全に除去されており「良い」の評価となるものであった。
硝酸鉄(III)九水和物(化学式:Fe(NO3)3・9H2O)を水道水に溶解し、硝酸鉄(III)換算で10質量%硝酸鉄(III)水溶液を作成し、「洗浄液」を得た。この時の溶液は、僅かに臭気を確認した。そして、銀ナノ粒子組成物(PChem社製:PSI-111型、銀濃度40質量%、平均粒子径(平均一次粒径)21nm)を、スクリーン印刷機(スクリーン版:PETメッシュ、420線/インチ、線径23μm)を用いて、スクリーン印刷を施した。
印刷後、余分なインクをゴムヘラで除去した。そして、印刷操作を24時間休止後、上記洗浄剤を満たした容器内にスクリーン版を浸漬させた。6時間後に洗浄液から取り出しブラシで擦りながら水洗を実施した。洗浄性を評価すると、銀ナノ粒子含有組成物は、ほぼ完全に除去されており「良い」の評価となるものであった。
実施例1において、硝酸鉄(III)水溶液の代わりに通常アニロックスロールの物理的洗浄剤として使用されているセラミックス系研磨剤含有の洗浄剤であるHARPER社製Ceram CleanIIを使用した。ウエスに該洗浄液を染み込ませ、2分間擦過させて洗浄した以外は実施例1を繰り返した。2.6BCMの評価では83%程度の除去効率であったが、13.5BCMでは全く除去できていなかった。これは13.5BCMの場合は容量が大きくよりセルが深いため洗浄効果が得られなかったと思われる。また、前記の洗浄剤の変更をした以外は実施例1を繰り返した場合には、除去率が更に低下する結果となった。
実施例1において、硝酸鉄(III)水溶液の代わりに通常アニロックスロールの化学的洗浄剤として使用されているグリコールエーテル系の洗浄剤(ANILOX ROLL CLEANER(Anilox Roll Services社製))を使用した。スポンジに該洗浄剤を染み込ませ、2分間擦過させて洗浄した以外は実施例1を繰り返した。除去率は2.6BCM、13.5BCMのいずれの場合でも40%以下であった。また、前記の洗浄剤の変更をした以外は実施例1を繰り返した場合には、除去率が更に低下する結果となった。
実施例1において、硝酸鉄(III)水溶液の代わりに市販の食器用洗剤としてColgate-Palmolive Company社製の製品名Palmoliveを使用した。水を十分に含んだスポンジに該洗浄剤を染み込ませ、2分間擦過させて洗浄した以外は実施例1を繰り返した。除去率は2.6BCM、13.5BCMのいずれの場合でも40%以下であった。また、前記の洗浄剤の変更をした以外は実施例1を繰り返した場合には、除去率が更に低下する結果となった。
実施例3において、硝酸鉄(III)に変えて、硝酸(10%)を用いた以外には同様にして、実施例3を繰り返した。評価結果を表1に併せて示す。高い洗浄性を示すが、臭気が強く連続使用には難があることを確認した。
Claims (9)
- 平均粒子径が1~100nmである銀ナノ粒子を含有する銀ナノ粒子含有組成物が洗浄対象物に付着して形成された銀成分の除去に用いられる、硝酸鉄水溶液を含有する、銀含有組成物用洗浄剤。
- 前記硝酸鉄水溶液中の硝酸鉄は、1~50質量%の範囲である、請求項1に記載の銀含有組成物用洗浄剤。
- 前記硝酸鉄は、硝酸鉄(III)(化学式Fe(NO3)3)である、請求項1に記載の銀含有組成物用洗浄剤。
- 前記洗浄対象物は、印刷用の版または印刷部材である、請求項1に記載の銀含有組成物用洗浄剤。
- 平均粒子径が1~100nmである銀ナノ粒子を含有する銀ナノ粒子含有組成物が洗浄対象物に付着して形成された銀成分の除去を行う銀含有組成物の除去方法であって、
硝酸鉄水溶液を含有する洗浄液を用い、除去対象物に付着した銀含有組成物を除去する工程を備える、銀含有組成物の除去方法。 - 前記硝酸鉄水溶液中の硝酸鉄は、1~50質量%の範囲である、請求項5に記載の銀含有組成物の除去方法。
- 前記除去対象物が、印刷用の版もしくは印刷部材である、請求項5に記載の銀含有組成物の除去方法。
- 請求項5に記載の銀含有組成物の除去方法により生じた銀含有洗浄廃液から、銀を分離回収する工程を備える、銀の回収方法。
- 前記銀を分離回収する工程は、前記洗浄後の銀含有洗浄廃液中に塩化物イオンを有する物質を添加し、銀成分を塩化銀として沈降させる、請求項8に記載の銀の回収方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013507040A JP5758483B2 (ja) | 2011-03-25 | 2011-09-30 | 銀含有組成物用洗浄剤、銀含有組成物の除去方法、および銀の回収方法 |
| CN201180066398.3A CN103339246B (zh) | 2011-03-25 | 2011-09-30 | 含银组合物用清洗剂、含银组合物的去除方法、以及银的回收方法 |
| EP11862362.8A EP2662437A4 (en) | 2011-03-25 | 2011-09-30 | CLEANING AGENT FOR SILVER-CONTAINING COMPOSITIONS, PROCESS FOR REMOVING COMPOSITION CONTAINING SILVER, AND METHOD FOR RECOVERING SILVER |
| HK13114325.2A HK1186750A1 (en) | 2011-03-25 | 2013-12-27 | Cleaning agent for silver-containing compositions, method for removing silver-containing composition, and method for recovering silver |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/071,865 | 2011-03-25 | ||
| US13/071,865 US20120244050A1 (en) | 2011-03-25 | 2011-03-25 | Cleaning agent for silver-containing composition, method for removing silver-containing composition, and method for recovering silver |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012132069A1 true WO2012132069A1 (ja) | 2012-10-04 |
Family
ID=46877512
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2011/072629 WO2012132069A1 (ja) | 2011-03-25 | 2011-09-30 | 銀含有組成物用洗浄剤、銀含有組成物の除去方法、および銀の回収方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20120244050A1 (ja) |
| EP (1) | EP2662437A4 (ja) |
| JP (1) | JP5758483B2 (ja) |
| CN (1) | CN103339246B (ja) |
| HK (1) | HK1186750A1 (ja) |
| TW (1) | TW201239083A (ja) |
| WO (1) | WO2012132069A1 (ja) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104409398A (zh) * | 2014-12-02 | 2015-03-11 | 昆山国显光电有限公司 | 刻蚀装置 |
| CN106701361B (zh) * | 2016-11-30 | 2020-07-31 | 江苏本川智能电路科技股份有限公司 | 一种用于清洗定影缸的药水及其使用方法 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH02217431A (ja) * | 1988-12-15 | 1990-08-30 | Imasa Ltd | 銅素地から錫、鉛、または錫・鉛合金析出物を剥離する方法と剥離用組成物 |
| JPH03505349A (ja) * | 1989-04-07 | 1991-11-21 | マクダーミツド インコーポレーテツド | アルカンスルホン酸と硝酸第2鉄を含む、銅表面からスズまたはスズ―鉛合金を剥離するための組成物、およびスズまたはスズ―鉛合金を剥離する方法 |
| JP2001011681A (ja) * | 1999-07-02 | 2001-01-16 | Mitsubishi Kakoki Kaisha Ltd | 廃液からの銀の回収方法 |
| JP2004002946A (ja) * | 2001-07-23 | 2004-01-08 | Sony Corp | エッチング方法及びエッチング液 |
| JP2005530885A (ja) * | 2002-06-25 | 2005-10-13 | サムスン エレクトロニクス カンパニー リミテッド | 配線用エッチング液とこれを用いる配線の製造方法とその配線を含む薄膜トランジスタアレイ基板及びその製造方法 |
| JP2005310703A (ja) | 2004-04-26 | 2005-11-04 | Bando Chem Ind Ltd | 導電性インクおよびそれを用いた導電性被膜 |
| JP2006164961A (ja) * | 2004-11-09 | 2006-06-22 | Ulvac Seimaku Kk | 積層型透明電極層の製造方法及びこの方法に使用する積層型透明電極形成用の積層体 |
| WO2008048316A2 (en) | 2005-12-20 | 2008-04-24 | Pchem Associates, Inc. | Synthesis of metallic nanoparticle dispersions |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2904432A (en) * | 1954-09-29 | 1959-09-15 | Corning Glass Works | Method of producing a photograph in glass |
| US3785818A (en) * | 1969-08-04 | 1974-01-15 | Du Pont | Method of making lithographic printing plates |
| JPS5112582B1 (ja) * | 1970-10-17 | 1976-04-20 | ||
| US3860423A (en) * | 1973-08-24 | 1975-01-14 | Rca Corp | Etching solution for silver |
| US5244539A (en) * | 1992-01-27 | 1993-09-14 | Ardrox, Inc. | Composition and method for stripping films from printed circuit boards |
| US5234542A (en) * | 1992-03-04 | 1993-08-10 | Macdermid, Incorporated | Composition and process for stripping tin from copper surfaces |
| US5512201A (en) * | 1995-02-13 | 1996-04-30 | Applied Chemical Technologies, Inc. | Solder and tin stripper composition |
| US6156433A (en) * | 1996-01-26 | 2000-12-05 | Dai Nippon Printing Co., Ltd. | Electrode for plasma display panel and process for producing the same |
| US6258294B1 (en) * | 1997-10-01 | 2001-07-10 | Morton International, Inc. | Composition for stripping solder and tin from printed circuit boards |
| US6531213B1 (en) * | 1999-01-13 | 2003-03-11 | Minolta Co, Ltd. | Recyclable image-recording medium, surface of which has specified saturated swelling amount |
| TWI243008B (en) * | 1999-12-22 | 2005-11-01 | Toyo Kohan Co Ltd | Multi-layer printed circuit board and its manufacturing method |
| US6165911A (en) * | 1999-12-29 | 2000-12-26 | Calveley; Peter Braden | Method of patterning a metal layer |
| US20050225413A1 (en) * | 2001-10-26 | 2005-10-13 | Kozicki Michael N | Microelectromechanical structures, devices including the structures, and methods of forming and tuning same |
| CN1312034C (zh) * | 2005-05-20 | 2007-04-25 | 清华大学 | 单一轴向排布的单晶硅纳米线阵列制备方法 |
| KR100713660B1 (ko) * | 2005-10-07 | 2007-05-02 | 한국지질자원연구원 | 은 스크랩으로부터 고순도 은의 정제방법 |
| CA2663688C (en) * | 2006-09-22 | 2015-11-17 | Fry's Metals, Inc. | Solvent systems for metals and inks |
| EP2244283A4 (en) * | 2008-02-15 | 2012-01-04 | Lion Corp | CLEANING COMPOSITION AND METHOD FOR CLEANING SUBSTRATE FOR ELECTRONIC DEVICE |
-
2011
- 2011-03-25 US US13/071,865 patent/US20120244050A1/en not_active Abandoned
- 2011-09-30 CN CN201180066398.3A patent/CN103339246B/zh not_active Expired - Fee Related
- 2011-09-30 EP EP11862362.8A patent/EP2662437A4/en not_active Withdrawn
- 2011-09-30 JP JP2013507040A patent/JP5758483B2/ja not_active Expired - Fee Related
- 2011-09-30 WO PCT/JP2011/072629 patent/WO2012132069A1/ja active Application Filing
- 2011-10-11 TW TW100136705A patent/TW201239083A/zh unknown
-
2013
- 2013-12-27 HK HK13114325.2A patent/HK1186750A1/xx not_active IP Right Cessation
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH02217431A (ja) * | 1988-12-15 | 1990-08-30 | Imasa Ltd | 銅素地から錫、鉛、または錫・鉛合金析出物を剥離する方法と剥離用組成物 |
| JPH03505349A (ja) * | 1989-04-07 | 1991-11-21 | マクダーミツド インコーポレーテツド | アルカンスルホン酸と硝酸第2鉄を含む、銅表面からスズまたはスズ―鉛合金を剥離するための組成物、およびスズまたはスズ―鉛合金を剥離する方法 |
| JP2001011681A (ja) * | 1999-07-02 | 2001-01-16 | Mitsubishi Kakoki Kaisha Ltd | 廃液からの銀の回収方法 |
| JP2004002946A (ja) * | 2001-07-23 | 2004-01-08 | Sony Corp | エッチング方法及びエッチング液 |
| JP2005530885A (ja) * | 2002-06-25 | 2005-10-13 | サムスン エレクトロニクス カンパニー リミテッド | 配線用エッチング液とこれを用いる配線の製造方法とその配線を含む薄膜トランジスタアレイ基板及びその製造方法 |
| JP2005310703A (ja) | 2004-04-26 | 2005-11-04 | Bando Chem Ind Ltd | 導電性インクおよびそれを用いた導電性被膜 |
| JP2006164961A (ja) * | 2004-11-09 | 2006-06-22 | Ulvac Seimaku Kk | 積層型透明電極層の製造方法及びこの方法に使用する積層型透明電極形成用の積層体 |
| WO2008048316A2 (en) | 2005-12-20 | 2008-04-24 | Pchem Associates, Inc. | Synthesis of metallic nanoparticle dispersions |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2662437A4 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20120244050A1 (en) | 2012-09-27 |
| EP2662437A4 (en) | 2015-06-03 |
| JPWO2012132069A1 (ja) | 2014-07-24 |
| CN103339246A (zh) | 2013-10-02 |
| HK1186750A1 (en) | 2014-03-21 |
| CN103339246B (zh) | 2015-09-02 |
| TW201239083A (en) | 2012-10-01 |
| EP2662437A1 (en) | 2013-11-13 |
| JP5758483B2 (ja) | 2015-08-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5631910B2 (ja) | 銀被覆銅粉 | |
| US6569252B1 (en) | Semi-aqueous solvent cleaning of paste processing residue from substrates | |
| JP2009235438A (ja) | エッチング液、該エッチング液を用いたエッチング方法および被エッチング基板 | |
| JP5388150B2 (ja) | 導電性金属インク組成物および導電性パターンの形成方法 | |
| CN107708893B (zh) | 覆银铜粉及使用其的铜膏、导电性涂料、导电性片以及覆银铜粉的制造方法 | |
| JP2010077495A (ja) | 銀被覆銅微粒子とその分散液及びその製造方法 | |
| JP5631841B2 (ja) | 銀被覆銅粉 | |
| JP5876971B2 (ja) | 銅粉 | |
| JP5424545B2 (ja) | 銅微粒子及びその製造方法、並びに銅微粒子分散液 | |
| JP5758483B2 (ja) | 銀含有組成物用洗浄剤、銀含有組成物の除去方法、および銀の回収方法 | |
| JP2007200775A (ja) | 金属微粒子分散体および金属微粒子分散体を利用した導電材料 | |
| JP6348241B1 (ja) | グラビアオフセット印刷用導電性ペースト、導電性パターンの形成方法、及び、導電性基板の製造方法 | |
| TWI754708B (zh) | 凹版轉印用導電性糊劑、導電性圖案的形成方法以及導電性基板的製造方法 | |
| JP2014159646A (ja) | 銀被覆銅粉 | |
| JP4939800B2 (ja) | スクリーン印刷ステンシルキャリヤーを親水化するための方法、ならびにスクリーン印刷ステンシルキャリヤーからステンシル材料を除去するための方法およびそのためのコーティング除去流体 | |
| JP2006219688A (ja) | 金属超微粉の精製方法 | |
| JP2005146386A (ja) | 金属粉スラリーの製造方法及びその製造方法で得られたニッケル粉スラリー | |
| CN104246017A (zh) | 抗蚀剂密合性提高剂和铜配线制造方法 | |
| JP7486294B2 (ja) | 洗浄液、洗浄液の製造方法及び設備の洗浄方法 | |
| JP3794965B2 (ja) | 銅板のレジストインク密着性改善方法 | |
| WO2023017749A1 (ja) | 導電性ペースト用洗浄剤および導電性ペーストの洗浄方法 | |
| JP6776973B2 (ja) | 表面被覆銅フィラー中間体の製造方法 | |
| JP2020070489A (ja) | 導電性微粒子分散体、導電性パターンの形成方法及び導電性基板の製造方法 | |
| JP2014173092A (ja) | ニッケルナノ粒子とその製造方法およびニッケルペースト | |
| JP2024005977A (ja) | ルテニウム酸鉛微粉末の製造方法、並びにルテニウム酸鉛微粉末作製用のルテニウム酸鉛の水酸化物及び水和物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11862362 Country of ref document: EP Kind code of ref document: A1 |
|
| REEP | Request for entry into the european phase |
Ref document number: 2011862362 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011862362 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2013507040 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |