WO2007068355A1 - Insektizide zusammensetzungen mit verbesserter wirkung - Google Patents
Insektizide zusammensetzungen mit verbesserter wirkung Download PDFInfo
- Publication number
- WO2007068355A1 WO2007068355A1 PCT/EP2006/011468 EP2006011468W WO2007068355A1 WO 2007068355 A1 WO2007068355 A1 WO 2007068355A1 EP 2006011468 W EP2006011468 W EP 2006011468W WO 2007068355 A1 WO2007068355 A1 WO 2007068355A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition according
- formula
- hydrogen
- alkyl
- salt
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/50—1,3-Diazoles; Hydrogenated 1,3-diazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N33/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic nitrogen compounds
- A01N33/02—Amines; Quaternary ammonium compounds
- A01N33/12—Quaternary ammonium compounds
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/02—Saturated carboxylic acids or thio analogues thereof; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/02—Saturated carboxylic acids or thio analogues thereof; Derivatives thereof
- A01N37/04—Saturated carboxylic acids or thio analogues thereof; Derivatives thereof polybasic
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/40—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having a double or triple bond to nitrogen, e.g. cyanates, cyanamides
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N51/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds having the sequences of atoms O—N—S, X—O—S, N—N—S, O—N—N or O-halogen, regardless of the number of bonds each atom has and with no atom of these sequences forming part of a heterocyclic ring
Definitions
- the present invention relates to the enhancement of the action of crop protection agents containing inhibitors of the nicotinic acetylcholine receptor (for example neonicotinoids) by the addition of ammonium or phosphonium salts or by the addition of ammonium or phosphonium salts and penetrants, the corresponding agents, processes for their preparation and their use in crop protection.
- nicotinic acetylcholine receptor for example neonicotinoids
- All inhibitors of the nicotinic acetylcholine receptor according to the invention are already known as agents for controlling animal pests, in particular insects, and can be prepared by methods described in the prior art.
- the effectiveness of these compounds is good, but not always fully satisfactory, especially at low application rates and concentrations. Furthermore, the plant tolerance of these compounds is not always sufficient. There is therefore a need for an increase in the effectiveness of the compounds containing plant protection products.
- Neonicotinoids can be represented by the formula (Ia)
- A represents in each case optionally substituted cycloalkyl, heterocyclyl, aryl or hetaryl,
- R 1 is hydrogen or alky 1
- R 2 is hydrogen or alkyl
- E is methyl, OR 3 , SR 3 or NR 4 R 5 ,
- R is in each case optionally substituted alkyl, alkenyl, cycloalkyl, cycloalkylalkyl or alkenyl interrupted by one or more heteroatoms or in each case substituted aryl, hetaryl, arylalkyl or hetarylalkyl, 4 5
- R and R independently of one another represent hydrogen, optionally interrupted by one or more heteroatoms and in each case optionally substituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkylalkyl, alkoxycarbonyl or in each case optionally substituted aryl, hetaryl, arylalkyl or hetarylalkyl,
- R 2 and R 5 together with the nitrogen atom to which they are attached form an optionally interrupted by one or more heteroatoms and optionally substituted ring,
- R 2 and R 3 together with the atoms linking them, form an optionally interrupted by one or more heteroatoms and optionally substituted ring,
- R 2 and R 5 together with the atoms linking them, form an optionally interrupted by one or more heteroatoms and optionally substituted ring,
- neonicotinoids can be replaced by the formula (Ib)
- B is in each case optionally substituted aryl, hetaryl or heterocyclyl,
- R 6 and R 8 are hydrogen or alkyl
- R 7 represents hydrogen or in each case optionally substituted alkyl, alkenyl, alkynyl, phenylalkyl or hetarylalkyl
- R 9 is hydrogen, alkyl or cycloalkyl
- Z stands for cyano or nitro
- neonicotinoids can be represented by the formula (Ic)
- Q is a five- or six-membered carbocyclic or heterocyclic ring, e.g. Phenyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, pyrrolyl, pyrazolyl, imidazolyl, thienyl,
- R 10 , R 11 , R 12 , R 13 and R 14 are each independently
- each Q and each R 10 , R u , R 12 , R 13 and R 14 may have one of the following substituents instead of any hydrogen present:
- Alkylthiocarbonyl, Ci-Ci 0 -Alkoxycarbonothioyl, Ci-Cio-Alkylthiocarbonothioyl, C r Cio-Dialkylphosphonyl, C, -C, 0 -Dialkylphosphatyl or HC ( NH>,
- neonicotinoids can be represented by the formula (Id)
- Ci-C branched or unbranched Ci-C 0 alkyl, Ci-Qo alkoxy, Ci-Cio-alkenyl, Ci-Ci 0 -
- (d) is hydrogen, hydroxyl, mercapto, amino, cyano, formyl, nitro, halogen or aminocarbonyl,
- R 15 and R 16 are each independently
- R 15 and R 16 are optionally linked together directly or via a one to four atom chain, these atoms being carbon, nitrogen, sulfur, phosphorus or oxygen,
- R 17 and R 18 are each independently
- Ci-Cio-alkyl CpCio alkoxy, Cj-Cio-alkenyl, Ci-C 10 - alkynyl, Ci-Ci 0 alkylthio, C r 0 Ci alkylsulfinyl, C r 0 Ci alkylsulfonyl, C 1 -C 10 -alkylcarbonyl, C 1 -C 10 -alkylcarbonothioyl, C 1 -C 10 -alkoxycarbonyl, C 1 -Ci 0 -
- Alkylthiocarbonyl, Cj-Cio-Alkoxycarbonothioyl, Ci-Cio-Alkylthiocarbonothioyl, C r 0 -Dialkylphosphonyl Ci, C, -C] 0 -Dialkylphosphatyl or HC ( NH>,
- R 17 and R 18 are optionally linked together directly or via a one to four atom chain, these atoms being carbon, nitrogen, sulfur, phosphorus or oxygen,
- R 16 and R 17 are optionally linked together directly or via a one to four atom chain, these atoms being carbon, nitrogen, sulfur, phosphorus or oxygen,
- Each Q, Y, and each R 15 , R 16 , R 17 , R 18, and R 19 may have one of the following substituents instead of any hydrogen present:
- neonicotinoids can be represented by the formula (Ie)
- R 20 , R 21 and R 22 are each independently
- R 20 and R 21 are optionally linked together directly or via a one to four atom chain, these atoms being carbon, nitrogen, sulfur, or oxygen,
- Each Q and each of R 20 , R 21 and R 22 may have one of the following substituents instead of any hydrogen present:
- Dialkylphosphonyl, C, -C, 0 -Dialkylphosphatyl or HC ( NH>,
- neonicotinoids can be replaced by the formula (If)
- R 23 is an unsubstituted or substituted 5- or 6-membered aromatic heterocyclic radical containing nitrogen
- R 24 is hydrogen, in each case unsubstituted or substituted alkyl, alkenyl, cycloalkyl, cycloalkenyl, aryl, -OR 25 , -SR 25 , -SO-R 25 , -SO 2 -R 25 , -CO-R 25 or -CO 2 - R 25 is
- R 25 is hydrogen or in each case unsubstituted or substituted alky), alkenyl, alkynyl, cycloalkyl, cycloalkenyl or aryl,
- G, J and D independently of one another each represent an unsubstituted or substituted carbon atom or heteroatom or a single bond
- L is in each case unsubstituted or substituted alkylene or alkylidene
- M is CO or CS
- V is hydrogen or in each case unsubstituted or substituted alkyl, alkenyl, alkynyl or aryl or is nitro, halogen or -WR 26 ,
- W is CO, CO 2 or S (O) n ,
- n 0, 1 or 2 and
- R 26 is each unsubstituted or substituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl or aryl,
- neonicotinoids can be expressed by the formula (Ig)
- n is an integer from 0 to 3
- R 31 and R 32 independently of one another represent hydrogen, methyl, ethyl, fluorine, chlorine or bromine,
- R 33 is nitro, cyano or COOR 37 ,
- R 34 represents a single bond or R 36 , S and R 34 together represent a five- or six-membered ring,
- R 36 is methyl or ethyl
- R 37 is Ci-Cj-alkyl
- a and hr are independently 0 or 1
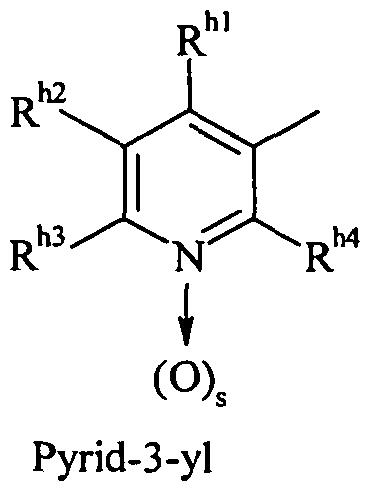
- R h1 , R h2 , R h3 and R h4 are independently selected from hydrogen, halogen, alkyl, alkoxy, haloalkyl and haloalkoxy,
- s is 0 or 1
- R> ha a , i Rjhb, R Tj hc c , and R are independently selected from hydrogen and alkyl
- b and c are 0 or 1 and
- R he, hf R, R and R are h ⁇ 11 * 1 are independently selected from hydrogen and alkyl
- R h is selected from hydrogen, haloalkyl, alkoxyalkyl, alkoxyalkoxyalkyl, cycloalkylalkyl, cyanoalkyl, formyl, alkylcarbonyl, alkoxycarbonyl, alkylsulphonyl, dialkylphosphonato, oxolan-3-ylmethyl, 2H-3,4,5,6-tetrahydropyran-2 -ylmethyl, cyclohex-1-en-3-yl, thien-3-ylmethyl, furan-2-ylmethyl, furan-3-ylmethyl, benzo [b] furan-2-ylmethyl, 2-R h8 -l, 3-thiazol-4-ylmethyl, 5-R h8 -l, 2,4-oxadiazol-3-ylmethyl,
- R is selected from halo, alkyl, aryl and hetaryl, wherein aryl and hetaryl are optionally substituted with at least one substituent selected from halogen, alkyl, haloalkyl, alkoxy and haloalkoxy;
- f 1 or 2
- R h9 , R hl °, R h ⁇ , R hl2 and R M3 are independently selected from hydrogen, halogen, alkyl, haloalkyl, alkoxy, haloalkoxy, alkoxyiminoalkyl, cyano, nitro, 2-alkyl-2H-tetrazol-5-yl, Aryl and aryloxy;
- R hl4 , R hl5 and R hl6 are independently selected from hydrogen, halogen, alkyl and aryl; R> hl7 is selected from hydrogen, alkyl,
- R hl8 , R hl9 , R h20 5 R h21 and R 1 " 22 are independently selected from hydrogen, halogen, alkyl, haloalkyl, alkoxy and haloalkoxy,
- R> h5 is selected from hydrogen, alkyl and
- n 1 or 2
- R h23 s R h24 ⁇ R t as ⁇ R h2 ⁇ and R h2? are independently selected from hydrogen, halogen, alkyl, haloalkyl, alkoxy and haloalkoxy,
- d and e are independently 0 or 1 and
- R h6 is selected from hydrogen, alkyl, cycloalkyl, cycloalkylalkyl, alkoxy, alkoxyalkyl, alkoxyalkoxyalkyl, alkenyl, haloalkenyl and
- R m , R h29 , R h3 °, R h31 and R h32 are independently selected from hydrogen, halogen, alkyl, haloalkyl, alkoxy and haloalkoxy,
- R h7 is selected from Cyano and Nitro
- W is selected from -CR h33 - and -N-,
- X 2 is selected from -CR h34 R h35 -, -O-, -S- and -NR h36 -
- R h33 , R h34 , R h35 and R h36 are independently selected from hydrogen and alkyl
- R h and R h5 together are ethylene to form a piperazine ring
- R h5 and X 2 together may form a cycle -CH 2 (CH 2 ), - or -CH 2 YCH 2 -, wherein q is 1 or 2, Y is selected from O, S and NR h37 , wherein R h37 is hydrogen or Alkyl is and
- X 2 is selected from -CH-, -O-, -S- and -N-, wherein when X is -CH- or -N-, R h6 is selected from hydrogen, alkyl and the above definitions for R h ,
- A is preferably phenyl which is optionally substituted by halogen (fluorine, chlorine, bromine, iodine), cyano, nitro, C 1 -C 4 -alkyl, C 1 -C 4 -haloalkyl, C 1 -C 4 -alkoxy or C 1 -C 4 -haloalkoxy,
- A preferably represents tetrahydrofuryl, which are optionally substituted by halo- gen, Ci-C 3 alkyl or C r C 3 haloalkyl.
- a furthermore preferably represents a saturated C 5 -C 6 -cycloalkyl radical optionally substituted by halogen or C 1 -C 3 -alkyl, in which optionally a methylene group is replaced by O or S.
- A is furthermore particularly preferred for
- R 1 is preferably hydrogen, methyl, ethyl, n-propyl or i-propyl.
- R 1 particularly preferably represents hydrogen or methyl.
- R 2 is preferably hydrogen, methyl, ethyl, n-propyl or i-propyl.
- R 2 particularly preferably represents hydrogen, methyl or ethyl.
- R 3 is preferably in each case optionally interrupted by oxygen or sulfur and optionally substituted by halogen, hydroxy or cyano-substituted straight-chain or branched C 1 -C 6 -alkyl, C 3 -C 6 -alkenyl, C 3 -C 7 -cycloalkyl, C 3 -C 7 -cycloalkyl-C, -C 2 -alkyl, for optionally substituted by Q-Gj-alkyl, Ci-C 2 -haloalkyl, halogen, CpC 4 -alkoxy, Ci-C 2 - haloalkoxy, nitro or cyano-substituted phenyl C r C 2 alkyl, phenyl, pyridyl, thiazolyl, pyrazolyl or pyrimidyl.
- halogen hydroxy or cyano-substituted straight-chain or branched C 1 -C 6 -alkyl
- R 3 particularly preferably represents straight-chain or branched C 1 -C 4 -alkyl, in particular methyl, ethyl, propyl, isopropyl, sec-butyl, tert-butyl, hydroxy-C 1 -C -alkyl, in particular 2-hydroxyethyl, 3-hydroxypropyl, C 3 -C 4 -alkenyl, in particular 2-propenyl, 2-butenyl, C 5 -C 7 -cycloalkyl, in particular cyclopropylmethyl, where appropriate by fluorine, chlorine, bromine,
- R 3 most preferably represents methyl or phenyl.
- R 4 and R 5 are preferably, and independently represent hydrogen, in each case optionally interrupted by oxygen or sulfur, and in each case optionally halogen-substituted straight-chain or branched Ci-C 6 alkyl, C 3 -C 6 alkenyl, C 3 -C 6 - Alkynyl, C 3 -C 7 -cycloalkyl, C 3 -C 7 -cycloalkyl-Ci-C 6 -alkenyl, Ci-C 6 -alkoxycarbonyl or optionally substituted by Ci-C 4 alkyl, Ci-C 2 -haloalkyl or halogen Phenyl, phenyl-Cp C 2 -alkenyl, pyridyl, thiazolyl, pyrid-Ci-C 2 -alkenyl or thiazolyl-C r C 2 -alkenyl.
- R 4 and R 5 are especially preferred and are independently hydrogen, straight or branched Ci-C 4 alkyl, C r G
- R 4 and R 5 are very particularly preferably and independently of one another hydrogen, methyl, ethyl, propyl, isopropyl, trifluoroethyl, 1,1,1-trifluoro-isopropyl, 2-propenyl or 2-butenyl.
- R 4 and R 5 are particularly preferably and independently of one another hydrogen, methyl or ethyl.
- E is preferably OR 3 , SR 3 or NR 4 R 5 .
- E is particularly preferably SR 3 or NR 4 R 5 .
- R 2 and R 3 preferably represent an optionally substituted by C) -C 4 alkyl C 2 -Q-Alkylidendiyl distrinum which may optionally be interrupted by a heteroatom from the series oxygen, sulfur or nitrogen.
- R 2 and R 3 are particularly preferably a C 2 -C 3 -alkyl] idendiyl group, which may optionally be interrupted by a heteroatom from the series oxygen, sulfur or nitrogen.
- R 2 and R 3 are very particularly preferably -CH 2 -CH 2 -, -CH 2 -CH 2 -CH 2 -, -CH 2 -O-CH 2 -, - CH 2 -S-CH 2 -, - CH 2 -NH-CH 2 or -CH 2 -N (CH 3 ) -CH 2 -.
- R 4 and R 5 are preferably and together with the N-atom to which they are attached for a 4-, 5-, 6- or 7-membered ring or for a 7- to 10-membered bicyclic, which may be also interrupted by oxygen, sulfur, sulfoxyl, sulfonyl, carbonyl, NR 6 or by quaternized nitrogen and optionally substituted by Ci-Gi-alkyl.
- R 4 and R 5 particularly preferably represent optionally substituted by methyl or ethyl, optionally interrupted by oxygen, sulfur or NR 6 interrupted C 4 -C 6 alkylidenediyl.
- R 4 and R 5 are very particularly preferably taken together with the N-atom to which they are attached, for pyrrolidino which is optionally substituted by methyl or ethyl, morpholino,
- R 2 and R 5 preferably together with the atoms which link them represent a saturated 5-, 6- or 7-membered ring optionally substituted by C 1 -C 4 -alkyl, R 2 and R 5 particularly preferably together with the atoms bonded to them represent a saturated 5-, 6- or 7-membered ring.
- R 2 and R 5 are most preferred and, together with the atoms linking them, a saturated 5- or 6-membered ring.
- R 1 is hydrogen
- B is preferably optionally substituted by halogen, cyano, nitro, C 1 -C 4 - haloalkyl, Ci-C4-alkoxy or Ci-C4-haloalkoxy.
- B is preferably also pyrazolyl, 1, 2,4-triazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, 1,2,5-thiadiazolyl, pyridyl, pyrazinyl, pyrimidinyl, Tetra- hydrofuryl, which (which is optionally substituted by fluorine and / or chlorine), optionally substituted by fluorine, chlorine, bromine, cyano, nitro, C r C 4 alkyl, Cj-C 4 -alkylthio (which is optionally substituted by fluorine and / or Chlorine is substituted), (which is optionally substituted by fluorine and / or chlorine) are substituted.
- B is particularly preferably thiazolyl or pyridyl, which are optionally substituted by halogen or C r C 3 alkyl.
- B is very particularly preferably thiazolyl or pyridyl, which are optionally substituted by chlorine or methyl.
- B is particularly preferably 2-chloro-pyrid-5-yl or 2-chloro-l, 3-thiazol-5-yl.
- R 6 is preferably hydrogen or methyl, ethyl or n-propyl or iso-propyl.
- R 6 particularly preferably represents hydrogen or methyl.
- R 6 very particularly preferably represents hydrogen.
- R 7 is preferably hydrogen, methyl, ethyl, n-propyl or n-butyl.
- R 7 particularly preferably represents hydrogen, methyl, ethyl or n-propyl.
- R 7 is very particularly preferably methyl.
- R 8 is preferably hydrogen, methyl or ethyl. R 8 particularly preferably represents hydrogen.
- R 9 preferably represents hydrogen, methyl, ethyl, n-propyl, isopropyl, cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
- R 9 particularly preferably represents hydrogen, methyl, ethyl or cyclopropyl.
- R 9 very particularly preferably represents hydrogen.
- Z is preferably cyano or nitro.
- Neonicotinoids called:
- Compound (Ib-2) has the formula
- Compound (Ib-3) has the formula
- Compound (Id-2) has the formula
- Compound (Ie-2) has the formula
- Compound (Ig-2) has the formula
- Compound (Ig-3) has the formula
- Compound (Ig-4) has the formula
- Compound (Ig-5) has the formula
- Inhibitors of the nicotinic acetylcholine receptor have a broad insecticidal effect, but the effect leaves much to be desired in detail.
- the active compounds can be used in the compositions according to the invention in a wide concentration range.
- concentration of the active ingredients in the formulation is usually 0.1-50 wt .-%.
- ammonium sulfate as a formulation aid is described for certain active ingredients and applications (WO 92/16108), but it is there to stabilize the formulation, not to increase the effect.
- the present invention thus relates to the use of ammonium and / or phosphonium salts to increase the efficacy of crop protection agents which contain insecticidally active inhibitors of the nicotinic acetylcholine receptor as the active ingredient.
- the invention also relates to compositions which contain such insecticides and the action-enhancing ammonium and / or phosphonium salts, both formulated active ingredients and ready-to-use agents (spray-broths).
- the subject matter of the invention is the use of these agents for controlling harmful insects.
- Ammonium and phosphonium salts which according to the invention increase the action of crop protection agents containing nicotinic acetylcholine receptor inhibitors are defined by formula (II)
- D is nitrogen or phosphorus
- D is preferably nitrogen
- R 26 , R 27 , R 28 and R 29 independently of one another represent hydrogen or in each case optionally substituted C 1 -C 6 -alkyl or mono- or polyunsaturated, optionally substituted C 1 -C 6 -alkylene, where the substituents are selected from halogen, nitro and cyano can,
- R 26 , R 27 , R 28 and R 29 preferably independently of one another represent hydrogen or in each case optionally substituted (VQ-alkyl, where the substituents can be selected from halogen, nitro and cyano,
- R 26 , R 27 , R 28 and R 29 more preferably independently of one another are hydrogen, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, s-butyl or t-butyl,
- R 26 , R 27 , R 28 and R 29 most preferably represent hydrogen, R 26 , R 27 , R 28 and R 29 furthermore very particularly preferably mean at the same time methyl or simultaneously ethyl,
- n 1, 2, 3 or 4
- n is preferably 1 or 2
- R 30 is an inorganic or organic anion
- R 30 is preferably hydrogencarbonate, tetraborate, fluoride, bromide, iodide, chloride, monohydrogenphosphate, dihydrogenphosphate, hydrogensulphate, tartrate, sulphate, nitrate, thiosulphate, thiocyanate, formate, lactate, acetate, propionate, butyrate, pentanoate, citrate or Oxalate stands,
- R 30 furthermore preferably represents carbonate, pentaborate, sulfite, benzoate, hydrogen oxalate, hydrogen citrate, methyl sulfate or tetrafluoroborate,
- R 30 particularly preferably represents lactate, sulfate, nitrate, thiosulfate, thiocyanate, citrate, oxalate or formate,
- R 30 is also particularly preferred for monohydrogen phosphate or dihydrogen phosphate
- R 30 very particularly preferably represents sulfate.
- the ammonium and phosphonium salts of the formula (II) can be used in a wide concentration range for increasing the effect of crop protection agents containing ketoenols.
- the ammonium or phosphonium salts in the ready-to-use crop protection agent are used in a concentration of 0.5 to 80 mmol / l, preferably 0.75 to 37.5 mmol / l, particularly preferably 1.5 to 25 mmol / l.
- the ammonium and / or phosphonium salt concentration in the formulation is selected to be in the specified general, preferred or most preferred ranges after dilution of the formulation to the desired drug concentration.
- the concentration of the salt in the formulation is usually 1-50% by weight.
- a penetration promoter is added to the crop protection agents to increase the effect. It can be described as completely surprising that even in these cases an even greater increase in activity can be observed.
- the subject matter of the present invention is therefore likewise the use of a combination of penetration promoters and ammonium and / or phosphonium salts to increase the efficacy of plant protection products, which insecticidally effective inhibitors of the nicotinic acetylcholine receptor as an active ingredient.
- the invention also relates to compositions which contain insecticidally active inhibitors of the nicotinic acetylcholine receptor, penetrants and ammonium and / or phosphonium salts, both formulated active ingredients and ready-to-use agents (spray-drying).
- insecticidally active inhibitors of the nicotinic acetylcholine receptor, penetrants and ammonium and / or phosphonium salts both formulated active ingredients and ready-to-use agents (spray-drying).
- the subject matter of the invention is the use of these agents for controlling harmful insects.
- Suitable penetration promoters in the present context are all those substances which are usually used to improve the penetration of agrochemical active substances into plants.
- Penetration promoters are in this context defined by the fact that they can penetrate from the aqueous spray mixture and / or from the spray coating in the cuticle of the plant and thereby increase the material mobility (mobility) of active ingredients in the cuticle. The method described in the literature (Baur et al.), 1997, Pesticide Science 51, 131-152) can be used to determine this property.
- Suitable penetration promoters are, for example, alkanol alkoxylates.
- Inventive penetration promoters are alkanol alkoxylates of the formula
- R is straight-chain or branched alkyl having 4 to 20 carbon atoms
- R 1 is hydrogen, methyl, ethyl, n-propyl, i-propyl, n-butyl, i-butyl, t-butyl, n-pentyl or n-hexyl,
- AO stands for an ethylene oxide radical, a propylene oxide radical, a butylene oxide radical or mixtures of ethylene oxide and propylene oxide radicals or butylene oxide radicals and
- v stands for numbers from 2 to 30.
- a preferred group of penetration enhancers are alkanol alkoxylates of the formula
- R has the meaning given above, R 'has the meaning given above,
- n stands for numbers from 2 to 20.
- Another preferred group of penetration enhancers are alkanol alkoxylates of the formula
- EO stands for -CH 2 -CH 2 -O-
- q stands for numbers from 1 to 10.
- Another preferred group of penetration promoters are alkanol alkoxylates of the formula
- EO stands for -CH 2 -CH 2 -O-
- s stands for numbers from 1 to 10.
- Another preferred group of penetration enhancers are alkanol alkoxylates of the formula
- EO is CH 2 -CH 2 -O-
- q stands for numbers from 1 to 10.
- Another preferred group of penetration enhancers are alkanol alkoxylates of the formula
- EO is CH 2 -CH 2 -O-
- s stands for numbers from 1 to 10.
- Another preferred group of penetration promoters are alkanol alkoxylates of the formula
- t stands for numbers from 8 to 13
- u stands for numbers from 6 to 17.
- R is preferably butyl, i-butyl, n -pentyl, i-pentyl, neopentyl, n-hexyl, i-hexyl, n-octyl, i-octyl, 2-ethylhexyl, nonyl, i-nonyl, decyl, n-dodecyl, i-dodecyl, lauryl, myristyl, i-tridecyl, trimethyl-nonyl, palmityl, stearyl or eicosyl.
- alkanol alkoxylate of the formula (III-c) is 2-ethyl-hexyl alkoxylate of the formula
- EO is -CH 2 -CH 2 -O-
- the numbers 8 and 6 represent average values called.
- the numbers 10, 6 and 2 represent average values called.
- Particularly preferred alkanol alkoxylates of the formula (III-f) are compounds of this formula in which
- alkanol alkoxylate of the formula (III-f-1) very particular preference is given to alkanol alkoxylate of the formula (III-f-1)
- u stands for the average 8.4.
- alkanol alkoxylates are generally defined by the above formulas. These substances are mixtures of substances of the specified type with different chain lengths. For the indices, therefore, average values are calculated, which may also differ from integers.
- alkanol alkoxylates of the formulas given are known and are partly available commercially or can be prepared by known methods (cf., WO 98-35553, WO 00-35278 and EP-A 0 681 865).
- Suitable penetration promoters are substances which promote the solubility of the compounds of the formula (I) in the spray coating. These include, for example, mineral or vegetable oils. Suitable oils are all mineral or vegetable, optionally modified, oils which can usually be used in agrochemical compositions. Examples include sunflower oil, rapeseed oil, olive oil, castor oil, rapeseed oil, corn kernel oil, cottonseed oil and soybean oil or the esters of said oils. Rape oil, sunflower oil and their methyl or ethyl esters are preferred.
- the concentration of penetration promoter can be varied within a wide range in the agents according to the invention.
- a formulated crop protection agent it is generally from 1 to 95% by weight, preferably from 1 to 55% by weight, more preferably from 15 to 40% by weight.
- the concentration is generally between 0.1 and 10 g / l, preferably between 0.5 and 5 g / l.
- Penetration promoter according to test means in that any compound which acts as a penetration enhancer in the test for cuticle penetration (Baur et al., 1997, Pesticide Science 51, 131-152) is suitable.
- Plant protection agents according to the invention may also contain further components, for example surfactants or dispersants or emulsifiers.
- Suitable nonionic surfactants or dispersing agents are all substances of this type which can usually be used in agrochemical compositions.
- Suitable anionic surfactants are all substances of this type conventionally usable in agrochemical compositions. Preference is given to alkali metal and alkaline earth metal salts of alkyl sulfonic acids or alkylaryl sulfonic acids.
- anionic surfactants or dispersing agents are salts of polystyrenesulfonic acids which are sparingly soluble in vegetable oil, salts of polyvinylsulfonic acids, salts of naphthalenesulfonic acid-formaldehyde condensation products, salts of condensation products of naphthalenesulfonic acid, phenolsulfonic acid and formaldehyde and salts of lignosulfonic acid.
- Suitable additives which may be present in the formulations according to the invention are emulsifiers, foam-inhibiting agents, preservatives, antioxidants, dyes and inert fillers.
- Preferred emulsifiers are ethoxylated nonylphenols, reaction products of alkylphenols with ethylene oxide and / or propylene oxide, ethoxylated arylalkylphenols, furthermore ethoxylated and propoxylated arylalkylphenols, as well as sulfated or phosphated arylalkyl ethoxylates or ethoxy-propoxylates, wherein sorbitan derivatives such as polyethylene oxide sorbitan fatty acid esters and sorbitan fatty acid esters may be mentioned as examples.
- Emulsifier 1 part by weight of alkylaryl polyglycol ether
- active compound 1 part by weight of active compound is mixed with the indicated amounts of solvent and emulsifier, and the concentrate is diluted with water to the desired concentration. If the addition of ammonium salts, phosphonium salts or penetration promoter is required, the corresponding amount is in each case pipetted into the finished preparation solution after dilution.
- One-leaved cotton plants (Gossypium hirsutum), which are heavily infested with the cotton aphid (Aphis gossypii), are treated by spraying the top of the leaves (spray volume 600 l / ha) with the preparation of active compound in the desired concentration.
- the kill is determined in%. 100% means that all animals have been killed; 0% means that no animals were killed.
- ammonium or phosphonium salts are able to increase the effect even when applying ready-to-use plant protection products containing penetration enhancers to increase their activity.
- Emulsifier part by weight of alkylaryl polyglycol ether
- active compound 1 part by weight of active compound is mixed with the indicated amounts of solvent and emulsifier, and the concentrate is diluted with water to the desired concentration.
- ammonium salts, phosphonium salts or penetration enhancer the corresponding amount is pipetted after dilution of each of the finished preparation solution.
- One-leaved pepper plants Capsicum an nuurri
- Myzus persicaei are treated by spraying the top of the leaves (spray volume 600 l / ha) with the preparation of active compound in the desired concentration. After the desired time the kill is determined in%. 100% means that all animals have been killed; 0% means that no animals were killed.
- RME rapeseed oil methyl ester (use formulated as 500 EW, concentration in g active substance / 1)
- active compound 1 part by weight of active compound is mixed with the indicated amounts of solvent and emulsifier, and the concentrate is diluted with water to the desired concentration.
- the corresponding amount is pipetted after dilution of each of the finished preparation solution.
- One-leaved cotton plants (Gossypium hirsutum), which are heavily infested with the cotton aphid ⁇ Aphis gossypii), are treated by spraying the leaf top (spray volume 600 l / ha) with the preparation of active compound in the desired concentration.
- the kill is determined in%. 100% means that all animals have been killed; 0% means that no animals were killed.
- RME rapeseed oil methyl ester (use formulated as 500 EW, concentration in g of active ingredient / 1)
- AS ammonium sulfate
- Emulsifier part by weight of alkylaryl polyglycol ether
- active compound 1 part by weight of active compound is mixed with the indicated amounts of solvent and emulsifier, and the concentrate is diluted with emulsifier-containing water to the desired concentration.
- the corresponding amount is pipetted after dilution of each of the finished preparation solution (1000 ppm each).
- Paprika plants (Capsicum annuum) which are heavily infested with the green peach aphid ⁇ Myzus persicaei) are treated by spraying with the preparation of active compound in the desired concentration.
- the kill is determined in%. 100% means that all animals have been killed; 0% means that no animals were killed.
- Emulsifier parts by weight of alkylaryl polyglycol ether
- active compound 1 part by weight of active compound is mixed with the indicated amounts of solvent and emulsifier, and the concentrate is diluted with emulsifier-containing water to the desired concentration. If ammonium salts or ammonium salts and penetration enhancers are required, the corresponding amount is added by pipette to the finished preparation solution after dilution (in each case 1000 ppm).
- Cotton leaves (Gossypium hirsutum), which are heavily infested with the cotton aphid ⁇ Aphis gossypii), are sprayed with a preparation of active compound of the desired concentration.
- Emulsifier part by weight of alkylaryl polyglycol ether
- Active ingredient with the specified amounts of solvent and emulsifier and the concentrate is diluted with water to the desired concentration.
- Ammonium salts or ammonium salts and penetration enhancers will the quantity is pipetted into the finished preparation solution after dilution (in each case 10000 ppm).
- One-leaved paprika plants (Capsicum annuum) which are heavily infested with the green peach aphid ⁇ Myzus persicae ⁇ ) are treated by spraying the underside of the leaf with the preparation of active compound in the desired concentration.
- the defrost is determined in%. 100% means that all animals have been killed; 0% means that no animals were killed.
- Emulsifier part by weight of alkylaryl polyglycol ether
- active compound 1 part by weight of active compound is mixed with the indicated amounts of solvent and emulsifier, and the concentrate is diluted with water to the desired concentration.
- the corresponding amount is pipetted after dilution of each of the finished preparation solution (1000 ppm each).
- One-leaved paprika plants (Capsicum annuum) heavily infested with the green peach aphid ⁇ Myzus persicaei) are treated by spraying the leaf top with the preparation of active compound at the desired concentration. After the desired time the kill is determined in%. 1 OO% means that all animals have been killed; O% means that no animals have been killed.
- Emulsifier 1 part by weight of alkylaryl polyglycol ether To prepare a suitable preparation of active compound, 1 part by weight of active compound is mixed with the stated amounts of solvent and emulsifier, and the concentrate is diluted with water to the desired concentration. When necessary addition of ammonium salts or ammonium salts and Penetrationsforderer the corresponding amount is pipetted after dilution of each of the finished preparation solution (1000 ppm each).
- One-leaved cotton plants (Gossypium hirsutum) heavily infested with the cotton aphid ⁇ Aphis gossypii) are treated by spraying the leaf underside with the preparation of active compound in the desired concentration.
- the kill is determined in%. 100% means that all animals have been killed; 0% means that no animals were killed.
- Emulsifier part by weight of alkylaryl polyglycol ether
- active compound 1 part by weight of active compound is mixed with the indicated amounts of solvent and emulsifier, and the concentrate is diluted with water to the desired concentration.
- the corresponding amount is pipetted after dilution of each of the finished preparation solution (1000 ppm each).
- One-leaved cotton plants (Gossypium hirsutum) heavily infested with the cotton aphid ⁇ Aphis gossypii) are treated by spraying the leaf top with the preparation of the active substance in the desired concentration.
- the kill is determined in%. 100% means that all animals have been killed; 0% means that no animals were killed.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008544797A JP2009519258A (ja) | 2005-12-13 | 2006-11-30 | 改善された活性を有する殺虫組成物 |
| BRPI0619816-3A BRPI0619816A2 (pt) | 2005-12-13 | 2006-11-30 | composições inseticidas com melhor efeito |
| AU2006326728A AU2006326728A1 (en) | 2005-12-13 | 2006-11-30 | Insecticidal compositions with improved effect |
| US12/096,903 US20090105235A1 (en) | 2005-12-13 | 2006-11-30 | Insecticidal compositions with improved effect |
| CA002632904A CA2632904A1 (en) | 2005-12-13 | 2006-11-30 | Insecticidal compositions with improved effect |
| EP06818913A EP1962606A1 (de) | 2005-12-13 | 2006-11-30 | Insektizide zusammensetzungen mit verbesserter wirkung |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102005059468.9 | 2005-12-13 | ||
| DE102005059468A DE102005059468A1 (de) | 2005-12-13 | 2005-12-13 | Insektizide Zusammensetzungen mit verbesserter Wirkung |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2007068355A1 true WO2007068355A1 (de) | 2007-06-21 |
| WO2007068355A8 WO2007068355A8 (de) | 2008-07-03 |
Family
ID=37907919
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2006/011468 WO2007068355A1 (de) | 2005-12-13 | 2006-11-30 | Insektizide zusammensetzungen mit verbesserter wirkung |
Country Status (15)
| Country | Link |
|---|---|
| US (1) | US20090105235A1 (de) |
| EP (1) | EP1962606A1 (de) |
| JP (1) | JP2009519258A (de) |
| KR (1) | KR20080078871A (de) |
| CN (1) | CN101330829A (de) |
| AR (1) | AR058339A1 (de) |
| AU (1) | AU2006326728A1 (de) |
| BR (1) | BRPI0619816A2 (de) |
| CA (1) | CA2632904A1 (de) |
| DE (1) | DE102005059468A1 (de) |
| IN (1) | IN2008DE04758A (de) |
| MX (1) | MX2008007471A (de) |
| TW (1) | TW200803744A (de) |
| WO (1) | WO2007068355A1 (de) |
| ZA (1) | ZA200805039B (de) |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102007045921A1 (de) | 2007-09-26 | 2009-04-02 | Bayer Cropscience Ag | Verfahren zur verbesserten Nutzung des Produktionspotentials transgener Pflanzen |
| EP2044841A1 (de) * | 2007-10-02 | 2009-04-08 | Bayer CropScience AG | Methode zur Verbesserung des Pflanzenwachstums |
| WO2009046837A2 (de) * | 2007-10-02 | 2009-04-16 | Bayer Cropscience Ag | Methoden zur verbesserung des pflanzenwachstums |
| WO2009056229A1 (de) * | 2007-10-30 | 2009-05-07 | Bayer Cropscience Ag | Insektizide zusammensetzungen von 2-cyano(het)-arylsulfonamidverbindungen und ihre isomeren formen mit verbesserter wirkung |
| EP2090168A1 (de) | 2008-02-12 | 2009-08-19 | Bayer CropScience AG | Methode zur Verbesserung des Pflanzenwachstums |
| EP2123159A1 (de) | 2008-05-21 | 2009-11-25 | Bayer CropScience AG | (1,2-Benzisothiazol-3-yl)(thio)carbamate und (1,2-Benzisothiazol-3-yl)(thio)oxamate und deren Oxidationsformen als Pestizide |
| WO2010019453A1 (en) | 2008-08-12 | 2010-02-18 | Dow Agrosciences Llc | Synergistic pesticidal compositions comprising an active compound, an ammonium salt, and a nonionic surfactant |
| DE102008041695A1 (de) | 2008-08-29 | 2010-03-04 | Bayer Cropscience Ag | Methoden zur Verbesserung des Pflanzenwachstums |
| EP2193713A1 (de) | 2008-12-05 | 2010-06-09 | Bayer CropScience AG | Verfahren zur Bekämpfung tierischer Schädlinge ohne Schädigung bestäubender Insekten |
| EP2196461A1 (de) | 2008-12-15 | 2010-06-16 | Bayer CropScience AG | 4-Amino-1,2,3-benzoxathiazin-Derivate als Pestizide |
| EP2198709A1 (de) | 2008-12-19 | 2010-06-23 | Bayer CropScience AG | Verfahren zur Bekämpfung resistenter tierischer Schädlinge |
| EP2201838A1 (de) | 2008-12-05 | 2010-06-30 | Bayer CropScience AG | Wirkstoff-Nützlings-Kombinationen mit insektiziden und akariziden Eigenschaften |
| WO2010083955A2 (de) | 2009-01-23 | 2010-07-29 | Bayer Cropscience Aktiengesellschaft | Verwendung von enaminocarbonylverbindungen zur bekämpfung von durch insekten übertragenen virosen |
| WO2010092032A1 (en) * | 2009-02-11 | 2010-08-19 | Basf Se | Pesticidal mixtures |
| WO2010094418A2 (de) | 2009-02-23 | 2010-08-26 | Bayer Cropscience Ag | Insektizide zusammensetzungen mit verbesserter wirkung |
| EP2223602A1 (de) | 2009-02-23 | 2010-09-01 | Bayer CropScience AG | Verfahren zur verbesserten Nutzung des Produktionspotentials genetisch modifizierter Pflanzen |
| WO2012000902A1 (de) * | 2010-06-29 | 2012-01-05 | Bayer Cropscience Ag | Verbesserte insektizide zusammensetzungen enthaltend cyclische carbonylamidine |
| EP2484676A2 (de) | 2008-12-18 | 2012-08-08 | Bayer CropScience AG | Tetrazolsubstituierte Anthranilsäureamide als Pestizide |
| WO2014170345A2 (en) | 2013-04-19 | 2014-10-23 | Bayer Cropscience Ag | Method for improved utilization of the production potential of transgenic plants |
| WO2016001129A1 (de) * | 2014-07-01 | 2016-01-07 | Bayer Cropscience Aktiengesellschaft | Verbesserte insektizide zusammensetzungen |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102004056626A1 (de) * | 2004-11-24 | 2006-06-01 | Bayer Cropscience Ag | Substituierte Oxyguanidine |
| EP2000027A1 (de) * | 2007-06-06 | 2008-12-10 | Bayer CropScience AG | Insektizide Zusammensetzungen mit verbesserter Wirkung |
| US9763451B2 (en) | 2008-12-29 | 2017-09-19 | Bayer Intellectual Property Gmbh | Method for improved use of the production potential of genetically modified plants |
| WO2018102385A1 (en) * | 2016-11-30 | 2018-06-07 | Mclaughlin Gormley King Company | Mixtures of sabadilla alkaloids and neonicotinoids and uses thereof |
| KR20210073518A (ko) | 2018-10-10 | 2021-06-18 | 구미아이 가가쿠 고교 가부시키가이샤 | 유성 현탁 농약 조성물 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2842476A (en) * | 1953-04-23 | 1958-07-08 | Mclaughlin Gormley King Co | Insecticidal compositions |
| US4020182A (en) * | 1973-04-10 | 1977-04-26 | Leo E. Burt | Pesticidal formaldehyde concentrate |
| US4888049A (en) * | 1986-06-27 | 1989-12-19 | Kao Corporation | Synergist for biocide |
| WO1992016108A1 (en) * | 1991-03-25 | 1992-10-01 | Valent U.S.A. Corporation | Chemically stable granules containing insecticidal phosphoroamidothioates |
| EP0664081A2 (de) * | 1994-01-20 | 1995-07-26 | Hoechst Schering AgrEvo GmbH | Synergistische Kombinationen von Ammoniumsalzen |
| WO2002098230A2 (de) * | 2001-04-11 | 2002-12-12 | Bayer Cropscience Ag | Verwendung von fettalkoholethoxylaten als penetrationsförderer |
| US20050009880A1 (en) * | 2002-09-12 | 2005-01-13 | Cottrell Ian W. | High concentration topical insecticide containing insect growth regulator |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000025586A1 (en) * | 1998-11-04 | 2000-05-11 | Syngenta Participations Ag | Herbicidal composition |
| US7445791B2 (en) * | 2004-07-12 | 2008-11-04 | United Phosphorus, Ltd. | Synergistic insecticidal composition containing Chloronicotynyle and Organosphosphorus compounds |
-
2005
- 2005-12-13 DE DE102005059468A patent/DE102005059468A1/de not_active Withdrawn
-
2006
- 2006-11-30 AU AU2006326728A patent/AU2006326728A1/en not_active Abandoned
- 2006-11-30 KR KR1020087016323A patent/KR20080078871A/ko not_active Application Discontinuation
- 2006-11-30 BR BRPI0619816-3A patent/BRPI0619816A2/pt not_active IP Right Cessation
- 2006-11-30 JP JP2008544797A patent/JP2009519258A/ja not_active Withdrawn
- 2006-11-30 EP EP06818913A patent/EP1962606A1/de not_active Withdrawn
- 2006-11-30 CN CNA2006800467614A patent/CN101330829A/zh active Pending
- 2006-11-30 US US12/096,903 patent/US20090105235A1/en not_active Abandoned
- 2006-11-30 WO PCT/EP2006/011468 patent/WO2007068355A1/de active Application Filing
- 2006-11-30 CA CA002632904A patent/CA2632904A1/en not_active Abandoned
- 2006-12-12 TW TW095146343A patent/TW200803744A/zh unknown
- 2006-12-13 AR ARP060105493A patent/AR058339A1/es not_active Application Discontinuation
-
2008
- 2008-06-03 IN IN4758DE2008 patent/IN2008DE04758A/en unknown
- 2008-06-10 ZA ZA200805039A patent/ZA200805039B/xx unknown
- 2008-06-10 MX MX2008007471A patent/MX2008007471A/es not_active Application Discontinuation
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2842476A (en) * | 1953-04-23 | 1958-07-08 | Mclaughlin Gormley King Co | Insecticidal compositions |
| US4020182A (en) * | 1973-04-10 | 1977-04-26 | Leo E. Burt | Pesticidal formaldehyde concentrate |
| US4888049A (en) * | 1986-06-27 | 1989-12-19 | Kao Corporation | Synergist for biocide |
| WO1992016108A1 (en) * | 1991-03-25 | 1992-10-01 | Valent U.S.A. Corporation | Chemically stable granules containing insecticidal phosphoroamidothioates |
| EP0664081A2 (de) * | 1994-01-20 | 1995-07-26 | Hoechst Schering AgrEvo GmbH | Synergistische Kombinationen von Ammoniumsalzen |
| WO2002098230A2 (de) * | 2001-04-11 | 2002-12-12 | Bayer Cropscience Ag | Verwendung von fettalkoholethoxylaten als penetrationsförderer |
| US20050009880A1 (en) * | 2002-09-12 | 2005-01-13 | Cottrell Ian W. | High concentration topical insecticide containing insect growth regulator |
Cited By (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102007045921A1 (de) | 2007-09-26 | 2009-04-02 | Bayer Cropscience Ag | Verfahren zur verbesserten Nutzung des Produktionspotentials transgener Pflanzen |
| US8481457B2 (en) | 2007-09-26 | 2013-07-09 | Bayer Cropscience Ag | Method for the improved use of the production potential of transgenic plants |
| EP2044841A1 (de) * | 2007-10-02 | 2009-04-08 | Bayer CropScience AG | Methode zur Verbesserung des Pflanzenwachstums |
| WO2009046837A2 (de) * | 2007-10-02 | 2009-04-16 | Bayer Cropscience Ag | Methoden zur verbesserung des pflanzenwachstums |
| JP2010540577A (ja) * | 2007-10-02 | 2010-12-24 | バイエル・クロツプサイエンス・アクチエンゲゼルシヤフト | 植物の生長を向上させる方法 |
| WO2009046837A3 (de) * | 2007-10-02 | 2010-03-25 | Bayer Cropscience Ag | Methoden zur verbesserung des pflanzenwachstums |
| CN101820763B (zh) * | 2007-10-02 | 2014-07-09 | 拜尔农作物科学股份公司 | 改善植物生长的方法 |
| WO2009056229A1 (de) * | 2007-10-30 | 2009-05-07 | Bayer Cropscience Ag | Insektizide zusammensetzungen von 2-cyano(het)-arylsulfonamidverbindungen und ihre isomeren formen mit verbesserter wirkung |
| EP2062476A1 (de) * | 2007-10-30 | 2009-05-27 | Bayer CropScience AG | Insektizide Zusammensetzungen von 2-Cyano(het)-arylsulfonamidverbindungen und ihre isomeren Formen mit verbesserter Wirkung |
| EP2090168A1 (de) | 2008-02-12 | 2009-08-19 | Bayer CropScience AG | Methode zur Verbesserung des Pflanzenwachstums |
| EP2123159A1 (de) | 2008-05-21 | 2009-11-25 | Bayer CropScience AG | (1,2-Benzisothiazol-3-yl)(thio)carbamate und (1,2-Benzisothiazol-3-yl)(thio)oxamate und deren Oxidationsformen als Pestizide |
| KR101292344B1 (ko) * | 2008-08-12 | 2013-07-31 | 다우 아그로사이언시즈 엘엘씨 | 활성 화합물, 암모늄 염 및 비이온성 계면활성제를 포함하는 상승작용적 살충성 조성물 |
| WO2010019453A1 (en) | 2008-08-12 | 2010-02-18 | Dow Agrosciences Llc | Synergistic pesticidal compositions comprising an active compound, an ammonium salt, and a nonionic surfactant |
| AU2009282194B2 (en) * | 2008-08-12 | 2012-04-19 | Dow Agrosciences Llc | Synergistic pesticidal compositions comprising an active compound, an ammonium salt, and a nonionic surfactant |
| JP2011530605A (ja) * | 2008-08-12 | 2011-12-22 | ダウ アグロサイエンシィズ エルエルシー | 活性化合物、アンモニウム塩、および非イオン性界面活性剤を含む相乗的殺虫剤組成物 |
| CN102123592A (zh) * | 2008-08-12 | 2011-07-13 | 陶氏益农公司 | 包含活性化合物、铵盐和非离子型表面活性剂的协同杀虫组合物 |
| US8232301B2 (en) | 2008-08-12 | 2012-07-31 | Dow AgroScience, LLC. | Pesticidal compositions |
| DE102008041695A1 (de) | 2008-08-29 | 2010-03-04 | Bayer Cropscience Ag | Methoden zur Verbesserung des Pflanzenwachstums |
| EP2193713A1 (de) | 2008-12-05 | 2010-06-09 | Bayer CropScience AG | Verfahren zur Bekämpfung tierischer Schädlinge ohne Schädigung bestäubender Insekten |
| EP2201838A1 (de) | 2008-12-05 | 2010-06-30 | Bayer CropScience AG | Wirkstoff-Nützlings-Kombinationen mit insektiziden und akariziden Eigenschaften |
| EP2196461A1 (de) | 2008-12-15 | 2010-06-16 | Bayer CropScience AG | 4-Amino-1,2,3-benzoxathiazin-Derivate als Pestizide |
| US8173641B2 (en) | 2008-12-15 | 2012-05-08 | Bayer Cropscience Ag | 4-amino-1,2,3-benzoxathiazine-derivatives as pesticides |
| EP2484676A2 (de) | 2008-12-18 | 2012-08-08 | Bayer CropScience AG | Tetrazolsubstituierte Anthranilsäureamide als Pestizide |
| EP2198709A1 (de) | 2008-12-19 | 2010-06-23 | Bayer CropScience AG | Verfahren zur Bekämpfung resistenter tierischer Schädlinge |
| EP2227951A1 (de) | 2009-01-23 | 2010-09-15 | Bayer CropScience AG | Verwendung von Enaminocarbonylverbindungen zur Bekämpfung von durch Insekten übertragenen Viren |
| WO2010083955A2 (de) | 2009-01-23 | 2010-07-29 | Bayer Cropscience Aktiengesellschaft | Verwendung von enaminocarbonylverbindungen zur bekämpfung von durch insekten übertragenen virosen |
| WO2010092032A1 (en) * | 2009-02-11 | 2010-08-19 | Basf Se | Pesticidal mixtures |
| WO2010094418A2 (de) | 2009-02-23 | 2010-08-26 | Bayer Cropscience Ag | Insektizide zusammensetzungen mit verbesserter wirkung |
| WO2010094418A3 (de) * | 2009-02-23 | 2011-06-23 | Bayer Cropscience Ag | Insektizide zusammensetzungen mit verbesserter wirkung |
| EP2223598A1 (de) | 2009-02-23 | 2010-09-01 | Bayer CropScience AG | Insektizide Zusammensetzungen mit verbesserter Wirkung |
| EP2223602A1 (de) | 2009-02-23 | 2010-09-01 | Bayer CropScience AG | Verfahren zur verbesserten Nutzung des Produktionspotentials genetisch modifizierter Pflanzen |
| WO2012000902A1 (de) * | 2010-06-29 | 2012-01-05 | Bayer Cropscience Ag | Verbesserte insektizide zusammensetzungen enthaltend cyclische carbonylamidine |
| WO2014170345A2 (en) | 2013-04-19 | 2014-10-23 | Bayer Cropscience Ag | Method for improved utilization of the production potential of transgenic plants |
| WO2016001129A1 (de) * | 2014-07-01 | 2016-01-07 | Bayer Cropscience Aktiengesellschaft | Verbesserte insektizide zusammensetzungen |
Also Published As
| Publication number | Publication date |
|---|---|
| DE102005059468A1 (de) | 2007-06-14 |
| KR20080078871A (ko) | 2008-08-28 |
| TW200803744A (en) | 2008-01-16 |
| WO2007068355A8 (de) | 2008-07-03 |
| BRPI0619816A2 (pt) | 2011-10-18 |
| EP1962606A1 (de) | 2008-09-03 |
| US20090105235A1 (en) | 2009-04-23 |
| MX2008007471A (es) | 2008-06-30 |
| CA2632904A1 (en) | 2007-06-21 |
| IN2008DE04758A (de) | 2008-08-15 |
| AR058339A1 (es) | 2008-01-30 |
| JP2009519258A (ja) | 2009-05-14 |
| ZA200805039B (en) | 2009-12-30 |
| AU2006326728A1 (en) | 2007-06-21 |
| CN101330829A (zh) | 2008-12-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2007068355A1 (de) | Insektizide zusammensetzungen mit verbesserter wirkung | |
| EP0113857B1 (de) | Herbizide Mittel | |
| EP1962603A1 (de) | Insektizide zusammensetzungen mit verbesserter wirkung | |
| EP1962594A1 (de) | Insektizide zusammensetzungen mit verbesserter wirkung | |
| AT506843B1 (de) | Direkt auf die haut aufzubringende lösung zur parasitenbekämpfung bei rindern und schafen | |
| EP1962595A1 (de) | Insektizide zusammensetzungen mit verbesserter wirkung | |
| DE19951328C2 (de) | Kältestabile Konservierungsmittel | |
| DE2641896A1 (de) | Quaternaere ammoniumverbindung und deren verwendung | |
| EP2000027A1 (de) | Insektizide Zusammensetzungen mit verbesserter Wirkung | |
| AT334134B (de) | Bekampfung von pilzschadlingen | |
| WO2010094418A2 (de) | Insektizide zusammensetzungen mit verbesserter wirkung | |
| DD151037A5 (de) | Herbizides und das pflanzenwachstum regulierendes mittel | |
| EP1593307A2 (de) | Mittel zur Bekämpfung von Pflanzenschädlingen | |
| DD262992A5 (de) | Nematizide und insektizide zusammensetzung | |
| DD236868A5 (de) | Mittel zum protahieren der wirkungsdauer und zur erhoehung der selektivitaet von herbiziden zusammensetzungen | |
| DD260644A5 (de) | Fungizide zusammensetzungen und ihre verwendung | |
| WO1997030043A1 (de) | Salze von imidazolin-derivaten und ihre verwendung als schädlingsbekämpfungsmittel | |
| DE2648705A1 (de) | Verfahren zur herstellung eines fungiziden mittels, fungizides mittel und verfahren zur behandlung von pilzinfektionen bei pflanzen | |
| DD279370A7 (de) | Rodenticide verbindungen, diese enthaltende konzentrateund daraus hergestellte mittel | |
| DE1049148B (de) | Nematozides Mittel | |
| DE1668081A1 (de) | Verfahren zur Herstellung von neuen Mucononitrilen und diese enthaltende pesticide Zusammensetzungen sowie neue Muconitrile | |
| DE2244745B2 (de) | Thiocarbaminsäureester, Ihre Herstellung und diese Ester enthaltende pestizide MIttel | |
| DE4014255A1 (de) | Herbizidloesung | |
| DD273571A1 (de) | Insektizide mittel | |
| DD278268A1 (de) | Fungizide mittel |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200680046761.4 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2006818913 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 4758/DELNP/2008 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 08058458 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/a/2008/007471 Country of ref document: MX Ref document number: 2632904 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008544797 Country of ref document: JP Ref document number: 2006326728 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12008501439 Country of ref document: PH |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2006326728 Country of ref document: AU Date of ref document: 20061130 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2006326728 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020087016323 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2006818913 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12096903 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: PI0619816 Country of ref document: BR Kind code of ref document: A2 Effective date: 20080613 |