JP6952348B2 - 殺菌性ポリマー及びポリマーと細胞との複合体ならびにそれらの合成 - Google Patents
殺菌性ポリマー及びポリマーと細胞との複合体ならびにそれらの合成 Download PDFInfo
- Publication number
- JP6952348B2 JP6952348B2 JP2018503480A JP2018503480A JP6952348B2 JP 6952348 B2 JP6952348 B2 JP 6952348B2 JP 2018503480 A JP2018503480 A JP 2018503480A JP 2018503480 A JP2018503480 A JP 2018503480A JP 6952348 B2 JP6952348 B2 JP 6952348B2
- Authority
- JP
- Japan
- Prior art keywords
- polymer
- days
- mol
- hma
- cells
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/74—Synthetic polymeric materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/74—Synthetic polymeric materials
- A61K31/765—Polymers containing oxygen
- A61K31/78—Polymers containing oxygen of acrylic acid or derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/58—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. poly[meth]acrylate, polyacrylamide, polystyrene, polyvinylpyrrolidone, polyvinylalcohol or polystyrene sulfonic acid resin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/593—Polyesters, e.g. PLGA or polylactide-co-glycolide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/61—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule the organic macromolecular compound being a polysaccharide or a derivative thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/69—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit
- A61K47/6903—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being semi-solid, e.g. an ointment, a gel, a hydrogel or a solidifying gel
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/06—Ointments; Bases therefor; Other semi-solid forms, e.g. creams, sticks, gels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L24/00—Surgical adhesives or cements; Adhesives for colostomy devices
- A61L24/001—Use of materials characterised by their function or physical properties
- A61L24/0031—Hydrogels or hydrocolloids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L24/00—Surgical adhesives or cements; Adhesives for colostomy devices
- A61L24/04—Surgical adhesives or cements; Adhesives for colostomy devices containing macromolecular materials
- A61L24/06—Surgical adhesives or cements; Adhesives for colostomy devices containing macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L26/00—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form
- A61L26/0009—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form containing macromolecular materials
- A61L26/0014—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form containing macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L26/00—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form
- A61L26/0061—Use of materials characterised by their function or physical properties
- A61L26/008—Hydrogels or hydrocolloids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/16—Macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/52—Hydrogels or hydrocolloids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/54—Biologically active materials, e.g. therapeutic substances
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/58—Materials at least partially resorbable by the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P41/00—Drugs used in surgical methods, e.g. surgery adjuvants for preventing adhesion or for vitreum substitution
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/52—Amides or imides
- C08F220/54—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
Description
本明細書に提供される定義に関して、別途明示的に述べられるか、又は文脈から明らかでない限り、定義される用語及び語句は提供された意味を含む。特に断らない限り、又は文脈から明らかでない限り、以下の用語及び語句は、用語又は語句が関連技術分野の当業者によって把握される意味を排除するものではない。本発明の範囲は特許請求の範囲によってのみ限定されるので、定義は具体的な実施形態の記述を補助するために提供され、特許請求の範囲に記載された発明を限定するものではない。文脈によって特に要求されない限り、単数形の用語は複数形を含み、複数形の用語は単数形を含むものとする。
PASポリマーの合成は、いくつかの予期せぬ課題を生じ、その各々は本出願人により本発明の方法で対処された。例えば、5−HMAモノマーの精製の間の、水とメタノールを使用することによるこのモノマーの再結晶化は、2つの溶媒の特定の体積比率(0.5:2、水:メタノール)を必要とし、これにより生成物の完全な溶解及び再結晶を確実にする。
PASポリマーの合成に使用されるモノマーの1つである5−HMAの調製のための合成方法は、SanRoman,et al.[an initial study is shown in Elvira and San Roman,“Synthesis and stereochemistry of isomeric methacrylic polymers derived from 4− and 5−aminosalicylic acids”,Polymer,38,4743−4750,1997;and a follow−up study is disclosed in Elvira,Gallardo,Lacroix,Schacht,San Roman,“Incorporation of salicylic acid derivatives to hydrophilic copolymer systems with biomedical applications”,J Mater Sci Mater Med,12,535−542,2001]に記載された方法から適合できる。
開始剤として0.3モル%の4,4’−アゾビス(4−シアノ吉草酸)を用いて70℃、THF中でNIPAAm(68モル%)、NAS(14モル%)、PLA/HEMA(8モル%)、OEGMA(5モル%)及び5−HMA(5モル%)のフリーラジカル重合によってPASポリマーを合成した。24時間後、得られたポリマー溶液を32℃の水中で沈殿させて、未反応モノマーを除去した。次いで、沈殿したポリマーをエタノールに溶解し、同じ条件下で水中に再沈殿させた。次いで、得られたポリマーを40℃で48時間減圧乾燥した。
OEGMA含量5モル%の合成PASポリマーは水溶性であることが判明した。ポリマーのペプチド反応性を確認するために、GRGDS({GLY}{ARG}{GLY}{ASP}{SER})合成ペプチドをモデルペプチドとして使用した。GRGDSは、細胞外マトリックス中の多くの接着タンパク質の細胞結合部位を模倣し、拡散した細胞を球状にして、剥離を引き起こす。GRGDSペプチドは、骨機能に関与することが最も報告されているインテグリンであるV3及びIIb3に対する中間的な親和性を有する。PAS(150mg/mL)とGRGDS(1mg/mL)を混合し、4℃で8時間反応させた。次いで、得られたPAS−co−GRGDS(PASGel)溶液を、生理学的温度(例えば、37℃)まで温度を上昇させてヒドロゲルに変換した。
組織工学、薬物送達及び創傷被覆剤適用において、生体材料の組織接着性は、組織漏出等の臨床的合併症を防止するために非常に重要である。本出願人は、PASGelのこの特性を評価するために、ブタ皮膚を用いたex vivo ASTMD3164−03(2014)標準法を使用した。この分析のために、100μLのPASGelを用いて、0.5×0.5cm2の領域を充填して、2枚のブタ皮膚を接着させた。
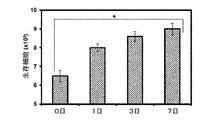
シミュレートされた生理学的条件では、PASGelヒドロゲルがそれぞれ311±47kPa及び781±116kPaの圧縮及び引張り係数を示した。これらの機械的性能パラメータは、以前に開発された又は市販のヒドロゲルと比較して、最も重要である。例えば、軟骨修復のための臨床試験段階にあるNeocartヒドロゲルは、10kPa未満の圧縮弾性率を有する[Ahmed and Hincke,“Strategies for articular cartilage lesion repair and functional restoration”Tissue Eng Part B Rev;16,305−329,2010]、BioGlue[CryoLife:BioGlue(R) Summary of Safety and Effectiveness Surgical Adhesive Indications for Use Contraindications;FDA Rep 2013]、ProGel[ProGel summary of Safety and Effectiveness Data;FDA Rep 2008:1−26]、及びCrosseal[Crosseal Summary of Safety and Effectiveness Data;FDA Rep 2010]のような、外科的シーラントと比較して、PASGelはより弾性的で機械的に堅牢である。これらの機械的性能の両方は、内外の創傷被覆及び組織再生にとって重要である。
PASGelが潜在的に、とりわけ以下のように使用されるかどうかを調べるための試験が行われた:
・内部出血シーラント;深部組織のための外科用シーラント;及び/又は
・欠陥部位の充填又は物理的封じ込めによって血液漏出を防止するために使用されることが意図される物理的凝固剤。
Claims (12)
- ポリ(N−イソプロピルアクリルアミド−co−N−アクリロキシスクシンイミド−co−2−ヒドロキシエチルメタクリレート/ポリラクチド−co−オリゴ(エチレングリコール)モノメチルエーテルメタクリレート−co−サリチル酸)ポリマーをベースとするポリマーであって、
5−HMA、4−HMA、又はそれらの組み合わせから選択されるサリチル酸のメタクリル酸エステル誘導体を含む、少なくとも1つの殺菌性/鎮痛性/抗炎症性モノマー単位;
おおよその相対モル比で、約5モル%未満の親水性エチレングリコール(OEGMA)成分の形態の水溶性モノマー単位;
ポリラクチド−co−2−ヒドロキシ−エチルメチルアクリレート(PLA/HEMA)の形態の機械的強度を付与するモノマー単位;
N−アクリロキシスクシンイミド(NAS)成分の形態のタンパク質/多糖類反応性モノマー単位;及び
N−イソプロピルアクリルアミド(NIPAAm)成分の形態の熱硬化性モノマー単位;
を含む、5元系のポリマー。 - おおよその相対モル比で、NIPAAm(68モル%);NAS(14モル%);PLA/HEMA(8モル%);OEGMA(約5モル%未満);並びに5−HMA及び/又は4−HMA(5モル%)を含む、請求項1に記載のポリマー。
- 前記ポリマーが、天然及び合成のペプチド又は多糖類と結合することができる、請求項1または2に記載のポリマー。
- 生物学的組織の修復を補助するための1以上の細胞を含む、ポリマーと細胞との複合体であって、
前記ポリマーは請求項3に記載のポリマーであって、
前記1以上の細胞が前記ポリマー全体にわたって比較的均一に分布し、
ポリマー中の1以上の細胞の数が約10 2 細胞/mL〜約10 8 細胞/mL、好ましくは約10 3 細胞/mL〜約10 5 細胞/mLである、ポリマーと細胞との複合体。 - 一定のモル量の殺菌性/鎮痛性/抗炎症性モノマー単位と;それぞれ一定のモル量の水溶性モノマー単位;機械的強度を付与するモノマー単位;タンパク質反応性モノマー単位;及び熱硬化性モノマー単位のうちの少なくとも3種との、一定のモル量の開始剤のフリーラジカル重合を含む、請求項1〜4のいずれかに記載のポリマーもしくはポリマーと細胞との複合体の合成方法。
- 約32℃で水中でポリマーを沈殿させて未反応モノマーを除去し、濾過する抽出工程をさらに含み、沈殿したポリマーをエタノールに溶解し、約32℃で水中に再沈殿させる精製工程をさらに含む、請求項5に記載の方法。
- 使用前にポリマーを約40℃の温度で約48時間、減圧下で乾燥させる、請求項6に記載の方法。
- 請求項1〜4のいずれかに記載のポリマーもしくはポリマーと細胞との複合体からなる医薬。
- 約0日〜約270日の期間にわたり、サリチル酸のメタクリル酸エステル誘導体からの殺菌性/鎮痛性/抗炎症性効果のバースト放出を防止するためのビヒクルを提供することによって、手術部位での時間ベースでの感染の発生率を減少させるために使用するための、請求項8に記載の医薬。
- 外科用シーラント、創傷治癒被覆剤、充填剤、又は生体内組織再生又は試験管内細胞増殖足場を必要とする医学的状態の治療における使用のための、請求項8に記載の医薬。
- 癌を含む医学的状態の治療における使用のための、請求項8に記載の医薬。
- 肺癌の治療における使用のための、請求項8に記載の医薬。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2015902943A AU2015902943A0 (en) | 2015-07-24 | Antiseptic polymer and synthesis thereof | |
| AU2015902943 | 2015-07-24 | ||
| PCT/AU2016/050653 WO2017015703A1 (en) | 2015-07-24 | 2016-07-22 | Antiseptic polymer and synthesis thereof |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2018523728A JP2018523728A (ja) | 2018-08-23 |
| JP2018523728A5 JP2018523728A5 (ja) | 2019-07-04 |
| JP6952348B2 true JP6952348B2 (ja) | 2021-10-20 |
Family
ID=57883841
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2018503480A Active JP6952348B2 (ja) | 2015-07-24 | 2016-07-22 | 殺菌性ポリマー及びポリマーと細胞との複合体ならびにそれらの合成 |
Country Status (5)
| Country | Link |
|---|---|
| US (2) | US11219639B2 (ja) |
| EP (1) | EP3325527B1 (ja) |
| JP (1) | JP6952348B2 (ja) |
| AU (2) | AU2016301103B9 (ja) |
| WO (1) | WO2017015703A1 (ja) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2016301103B9 (en) * | 2015-07-24 | 2019-08-08 | Trimph Ip Pty Ltd | Antiseptic polymer and synthesis thereof |
| US20180243469A1 (en) * | 2015-09-01 | 2018-08-30 | The University Of Sydney | Bioactive polymer for bone regeneration |
| US20230039698A1 (en) * | 2019-12-19 | 2023-02-09 | Trimph Ip Pty Ltd | Biocompatible Material |
| GB202001439D0 (en) | 2020-02-03 | 2020-03-18 | Univ Nottingham | Drug formulations |
Family Cites Families (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2624690A (en) * | 1943-02-19 | 1953-01-06 | Portland Plastics Ltd | Adhesive compositions comprising polymerized methyl methacrylate, an ester of salicylic acid and an organic solvent |
| US3290270A (en) | 1963-05-23 | 1966-12-06 | Nat Starch Chem Corp | Salicylic acid derivatives and polymers thereof |
| JPS63268780A (ja) * | 1987-04-28 | 1988-11-07 | G C Dental Ind Corp | 歯質接着性化合物 |
| JPH075680B2 (ja) | 1987-07-03 | 1995-01-25 | 三井石油化学工業株式会社 | 硬化性組成物 |
| FR2629084B1 (fr) | 1988-03-28 | 1990-11-30 | Essilor Int | Polymeres supports de produits actifs relargables, de type hydrogel, compositions correspondantes et leur procede de preparation |
| JP3495099B2 (ja) | 1993-08-02 | 2004-02-09 | サンメディカル株式会社 | 硬化性接着材組成物および接着材キット |
| US5530038A (en) | 1993-08-02 | 1996-06-25 | Sun Medical Co., Ltd. | Primer composition and curable composition |
| JPH08119822A (ja) * | 1994-10-20 | 1996-05-14 | Sogo Shika Iryo Kenkyusho:Kk | 歯科用接着性根管充填材料 |
| CA2427938C (en) | 1999-11-15 | 2010-02-16 | Bio Syntech Canada Inc. | Novel temperature-controlled and ph-dependant self-gelling biopolymeric aqueous solution, composition and preparation thereof |
| US20050233003A1 (en) | 2001-09-28 | 2005-10-20 | Solubest Ltd. | Hydrophilic dispersions of nanoparticles of inclusion complexes of salicylic acid |
| JP2004182661A (ja) * | 2002-12-04 | 2004-07-02 | Gc Corp | 歯科用レジン強化型セメント用前処理剤 |
| US20070224278A1 (en) | 2003-11-12 | 2007-09-27 | Lyons Robert T | Low immunogenicity corticosteroid compositions |
| DE102004018093A1 (de) * | 2004-04-08 | 2005-10-27 | Rohmax Additives Gmbh | Polymere mit H-Brücken bildenden Funktionalitäten |
| US7968085B2 (en) | 2004-07-05 | 2011-06-28 | Ascendis Pharma A/S | Hydrogel formulations |
| US20060062768A1 (en) | 2004-09-23 | 2006-03-23 | Olexander Hnojewyj | Biocompatible hydrogel compositions |
| KR100665672B1 (ko) | 2005-04-13 | 2007-01-09 | 성균관대학교산학협력단 | 새로운 온도 및 pH 민감성 블록 공중합체 및 이를 이용한고분자 하이드로겔 |
| US8828433B2 (en) | 2005-04-19 | 2014-09-09 | Advanced Cardiovascular Systems, Inc. | Hydrogel bioscaffoldings and biomedical device coatings |
| ITTO20050549A1 (it) | 2005-08-03 | 2007-02-04 | Consiglio Nazionale Ricerche | Materiale composito iniettabile atto ad essere utilizzato come sostituto osseo |
| WO2008033847A2 (en) | 2006-09-11 | 2008-03-20 | Emory University | Modified protein polymers |
| US8394782B2 (en) | 2007-11-30 | 2013-03-12 | Allergan, Inc. | Polysaccharide gel formulation having increased longevity |
| EP2323670A4 (en) | 2008-08-05 | 2013-12-25 | Biomimedica Inc | POLYURETHANE-GEPPROPFTE HYDROGELE |
| RU2538677C2 (ru) * | 2009-11-20 | 2015-01-10 | Митсуи Кемикалс, Инк. | Адгезивная композиция для мягких тканей, адгезивная композиция для обработки раны или композиция для раневой повязки |
| WO2011073991A1 (en) | 2009-12-16 | 2011-06-23 | Regentis Biomaterials Ltd. | Scaffolds formed from polymer-protein conjugates, methods of generating same and uses thereof |
| US8741317B2 (en) | 2010-08-19 | 2014-06-03 | Rutgers, The State University Of New Jersey | Slow-degrading polymers comprising salicylic acid for undelayed and sustained drug delivery |
| US8658711B2 (en) | 2010-09-29 | 2014-02-25 | Rutgers, The State University Of New Jersey | Process for the synthesis of methacrylate-derivatized type-1 collagen and derivatives thereof |
| US8853162B2 (en) | 2011-05-11 | 2014-10-07 | Agency For Science, Technology And Research | Interpenetrating polymer network comprising fibrin |
| US9546235B2 (en) | 2011-12-19 | 2017-01-17 | The University Of Sydney | Peptide-hydrogel composite |
| ES2455441B1 (es) | 2012-09-14 | 2015-02-05 | Universidad De Valladolid | Hidrogel útil como soporte inyectable para aplicación en terapia celular y como sistema de liberación controlada de fármacos |
| KR20140093349A (ko) | 2013-01-15 | 2014-07-28 | 주식회사 제닉 | Ow 고형 화장료 조성물 및 그 제조방법 |
| CN105102469A (zh) | 2013-01-28 | 2015-11-25 | 新加坡科技研究局 | 交联的肽水凝胶 |
| EP2956185B1 (en) | 2013-02-13 | 2021-12-29 | Agency For Science, Technology And Research | A polymeric system for release of an active agent |
| SG11201507370TA (en) | 2013-03-14 | 2015-10-29 | Massachusetts Inst Technology | Multi-layered injectable self-assembling peptide scaffold hydrogels for long-term sustained release of human antibodies |
| AU2016301103B9 (en) * | 2015-07-24 | 2019-08-08 | Trimph Ip Pty Ltd | Antiseptic polymer and synthesis thereof |
-
2016
- 2016-07-22 AU AU2016301103A patent/AU2016301103B9/en active Active
- 2016-07-22 WO PCT/AU2016/050653 patent/WO2017015703A1/en unknown
- 2016-07-22 EP EP16829490.8A patent/EP3325527B1/en active Active
- 2016-07-22 JP JP2018503480A patent/JP6952348B2/ja active Active
- 2016-07-22 US US15/746,810 patent/US11219639B2/en active Active
-
2019
- 2019-09-11 AU AU2019229341A patent/AU2019229341A1/en not_active Abandoned
-
2021
- 2021-12-21 US US17/557,108 patent/US11872245B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| EP3325527B1 (en) | 2024-03-27 |
| AU2016301103A1 (en) | 2018-02-15 |
| US11872245B2 (en) | 2024-01-16 |
| AU2016301103B9 (en) | 2019-08-08 |
| JP2018523728A (ja) | 2018-08-23 |
| WO2017015703A1 (en) | 2017-02-02 |
| US11219639B2 (en) | 2022-01-11 |
| AU2019229341A1 (en) | 2019-10-03 |
| EP3325527A4 (en) | 2019-03-20 |
| US20220125828A1 (en) | 2022-04-28 |
| US20200078392A1 (en) | 2020-03-12 |
| AU2016301103B2 (en) | 2019-07-11 |
| EP3325527A1 (en) | 2018-05-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101844878B1 (ko) | 주입형 이중가교 하이드로젤 및 이의 생의학적 용도 | |
| JP6334405B2 (ja) | 生分解性医療用接着剤、その調製法及び使用 | |
| JP6952348B2 (ja) | 殺菌性ポリマー及びポリマーと細胞との複合体ならびにそれらの合成 | |
| US8889791B2 (en) | Thermoresponsive, biodegradable, elastomeric material | |
| KR101301276B1 (ko) | 카테콜기가 결합된 키토산 또는 폴리아민 및 말단에 티올기가 결합된 폴락소머를 포함하는 하이드로젤 및 이의 제조방법 및 이를 이용한 지혈제 | |
| WO2007124132A2 (en) | Polymeric compositions and methods of making and using thereof | |
| KR20100118102A (ko) | 생분해성 단일-상 점착성 히드로겔 | |
| US20120148523A1 (en) | Hydrogel tissue adhesive for medical use | |
| Hao et al. | Injectable self-healing first-aid tissue adhesives with outstanding hemostatic and antibacterial performances for trauma emergency care | |
| US8747870B2 (en) | Polymeric compositions and methods of making and using thereof | |
| WO2015015212A1 (en) | Formulations and materials with cationic polymers | |
| AU2016314146A1 (en) | Bioactive polymer for bone regeneration | |
| Liu et al. | Adhesive, antibacterial and double crosslinked carboxylated polyvinyl alcohol/chitosan hydrogel to enhance dynamic skin wound healing | |
| JP2021504097A (ja) | 止血用組成物及びこれを含む容器 | |
| US8859705B2 (en) | Hydrogel tissue adhesive having decreased gelation time and decreased degradation time | |
| JP2008543922A (ja) | 生体吸収性ヒドロゲル | |
| AU2022353816A1 (en) | Injectable shear-thinning hydrogel containing therapetuic agent for enhanced tumor therapy | |
| JP2022518809A (ja) | 止血用組成物及びこれを含む容器 | |
| Milne et al. | Dual-modified hyaluronic acid for tunable double cross-linked hydrogel adhesives | |
| US20230211043A1 (en) | Medical adhesive and preparation method thereof | |
| US8796242B2 (en) | Hydrogel tissue adhesive for medical use | |
| Lanzalaco et al. | Thermo/Pressure-Sensitive Self-Fixation Surgical Meshes: The Role of Adhesive Hydrogels in Interface Attachment | |
| Guo et al. | Periodontium-Mimicking, Multifunctional Biomass-Based Hydrogel Promotes Full-Course Socket Healing | |
| Sarwan | A hybrid thermo-responsive polymer system for application in wound healing | |
| Kommineni et al. | Historical Developments of Various Adhesives for Biomedical Applications |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A524 | Written submission of copy of amendment under article 19 pct |
Free format text: JAPANESE INTERMEDIATE CODE: A525 Effective date: 20180226 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20180726 |
|
| A711 | Notification of change in applicant |
Free format text: JAPANESE INTERMEDIATE CODE: A711 Effective date: 20190508 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190516 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20190516 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20190508 |
|
| A711 | Notification of change in applicant |
Free format text: JAPANESE INTERMEDIATE CODE: A711 Effective date: 20190701 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20190701 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20200520 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20200721 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20200915 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20210119 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210129 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20210615 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210628 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20210831 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20210917 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6952348 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |