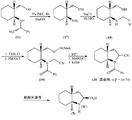
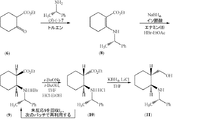
JP2012533563A - トランドラプリル用の中間体である(2S,3aR,7aS)−オクタヒドロ−1H−インドールカルボン酸の合成法 - Google Patents
トランドラプリル用の中間体である(2S,3aR,7aS)−オクタヒドロ−1H−インドールカルボン酸の合成法 Download PDFInfo
- Publication number
- JP2012533563A JP2012533563A JP2012520807A JP2012520807A JP2012533563A JP 2012533563 A JP2012533563 A JP 2012533563A JP 2012520807 A JP2012520807 A JP 2012520807A JP 2012520807 A JP2012520807 A JP 2012520807A JP 2012533563 A JP2012533563 A JP 2012533563A
- Authority
- JP
- Japan
- Prior art keywords
- compound
- ethyl
- phenylethylamino
- indole
- added
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 238000000034 method Methods 0.000 title claims abstract description 95
- CQYBNXGHMBNGCG-CSMHCCOUSA-N (2s,3ar,7as)-2,3,3a,4,5,6,7,7a-octahydro-1h-indol-1-ium-2-carboxylate Chemical compound C1CCC[C@@H]2N[C@H](C(=O)O)C[C@H]21 CQYBNXGHMBNGCG-CSMHCCOUSA-N 0.000 title abstract description 22
- VXFJYXUZANRPDJ-WTNASJBWSA-N Trandopril Chemical compound C([C@@H](C(=O)OCC)N[C@@H](C)C(=O)N1[C@@H](C[C@H]2CCCC[C@@H]21)C(O)=O)CC1=CC=CC=C1 VXFJYXUZANRPDJ-WTNASJBWSA-N 0.000 title abstract description 22
- 229960002051 trandolapril Drugs 0.000 title abstract description 15
- 230000002194 synthesizing effect Effects 0.000 title abstract 2
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims abstract description 120
- PONAUWFRJYNGAC-MWDCIYOWSA-N (2s,3ar,7as)-2,3,3a,4,5,6,7,7a-octahydro-1h-indole-2-carboxylic acid;hydrochloride Chemical compound Cl.C1CCC[C@@H]2N[C@H](C(=O)O)C[C@H]21 PONAUWFRJYNGAC-MWDCIYOWSA-N 0.000 claims abstract description 39
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims abstract description 32
- 238000004519 manufacturing process Methods 0.000 claims abstract description 23
- 150000001875 compounds Chemical class 0.000 description 187
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 105
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 102
- 239000000243 solution Substances 0.000 description 67
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical group CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 65
- 238000006243 chemical reaction Methods 0.000 description 65
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 56
- -1 benzyl ester Chemical class 0.000 description 46
- 239000000203 mixture Substances 0.000 description 46
- LMASLKDDKPUJIF-IMJJTQAJSA-N ethyl (1r,2s)-2-[[(1s)-1-phenylethyl]amino]cyclohexane-1-carboxylate Chemical compound CCOC(=O)[C@@H]1CCCC[C@@H]1N[C@@H](C)C1=CC=CC=C1 LMASLKDDKPUJIF-IMJJTQAJSA-N 0.000 description 39
- YWMXXASKTSNCOG-RANZSIQMSA-N ethyl (2s,3ar,7as)-1-[(1s)-1-phenylethyl]-2,3,3a,4,5,6,7,7a-octahydroindole-2-carboxylate Chemical compound C1([C@H](C)N2[C@H]3CCCC[C@@H]3C[C@H]2C(=O)OCC)=CC=CC=C1 YWMXXASKTSNCOG-RANZSIQMSA-N 0.000 description 37
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 36
- 230000008569 process Effects 0.000 description 34
- QEPMKQQWRZBOMR-GEUPQXMHSA-N [(1s,2s)-2-ethoxycarbonylcyclohexyl]-[(1s)-1-phenylethyl]azanium;chloride Chemical compound Cl.CCOC(=O)[C@H]1CCCC[C@@H]1N[C@@H](C)C1=CC=CC=C1 QEPMKQQWRZBOMR-GEUPQXMHSA-N 0.000 description 32
- 239000010410 layer Substances 0.000 description 32
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 30
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 30
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 30
- 239000012299 nitrogen atmosphere Substances 0.000 description 30
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 30
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 30
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 28
- WOXVBEWLLZBVSX-DSMRVHDJSA-N benzyl (2s,3ar,7as)-2,3,3a,4,5,6,7,7a-octahydro-1h-indole-2-carboxylate;hydrochloride Chemical compound Cl.O=C([C@H]1N[C@H]2CCCC[C@@H]2C1)OCC1=CC=CC=C1 WOXVBEWLLZBVSX-DSMRVHDJSA-N 0.000 description 28
- UYUHUFZUXQFRBI-ZDUSSCGKSA-N ethyl 2-[[(1s)-1-phenylethyl]amino]cyclohexene-1-carboxylate Chemical compound C1CCCC(C(=O)OCC)=C1N[C@@H](C)C1=CC=CC=C1 UYUHUFZUXQFRBI-ZDUSSCGKSA-N 0.000 description 28
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 28
- 239000007787 solid Substances 0.000 description 27
- 239000003054 catalyst Substances 0.000 description 24
- FGSGHBPKHFDJOP-UHFFFAOYSA-N ethyl 2-oxocyclohexane-1-carboxylate Chemical compound CCOC(=O)C1CCCCC1=O FGSGHBPKHFDJOP-UHFFFAOYSA-N 0.000 description 23
- 239000002904 solvent Substances 0.000 description 23
- HQNOCIZWFSGXEE-UHOSZYNNSA-N ethyl 2-[[(1s,2s)-2-(methylsulfonyloxymethyl)cyclohexyl]-[(1s)-1-phenylethyl]amino]acetate Chemical compound C1([C@H](C)N(CC(=O)OCC)[C@@H]2[C@H](CCCC2)COS(C)(=O)=O)=CC=CC=C1 HQNOCIZWFSGXEE-UHOSZYNNSA-N 0.000 description 19
- 239000007788 liquid Substances 0.000 description 19
- 239000012044 organic layer Substances 0.000 description 19
- FHHADLZNYHYSSM-CFVMTHIKSA-N [(1s,2s)-2-[[(1s)-1-phenylethyl]amino]cyclohexyl]methanol Chemical compound N([C@@H](C)C=1C=CC=CC=1)[C@H]1CCCC[C@@H]1CO FHHADLZNYHYSSM-CFVMTHIKSA-N 0.000 description 17
- GCWPGEWXYDEQAY-RQJHMYQMSA-N [(1s,2s)-2-aminocyclohexyl]methanol Chemical compound N[C@H]1CCCC[C@@H]1CO GCWPGEWXYDEQAY-RQJHMYQMSA-N 0.000 description 16
- NHAKCNUGMZOAEA-JQHSSLGASA-N ethyl 2-[[(1s,2s)-2-(hydroxymethyl)cyclohexyl]-[(1s)-1-phenylethyl]amino]acetate Chemical compound C1([C@H](C)N(CC(=O)OCC)[C@@H]2[C@H](CCCC2)CO)=CC=CC=C1 NHAKCNUGMZOAEA-JQHSSLGASA-N 0.000 description 16
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 15
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 15
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 15
- 230000008901 benefit Effects 0.000 description 15
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 14
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 14
- 150000001412 amines Chemical class 0.000 description 14
- KQNPFQTWMSNSAP-UHFFFAOYSA-N isobutyric acid Chemical compound CC(C)C(O)=O KQNPFQTWMSNSAP-UHFFFAOYSA-N 0.000 description 14
- 239000011780 sodium chloride Substances 0.000 description 14
- RQEUFEKYXDPUSK-ZETCQYMHSA-N (1S)-1-phenylethanamine Chemical compound C[C@H](N)C1=CC=CC=C1 RQEUFEKYXDPUSK-ZETCQYMHSA-N 0.000 description 13
- 239000002253 acid Substances 0.000 description 13
- 239000000284 extract Substances 0.000 description 13
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 12
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 12
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 12
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 12
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 12
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 12
- PQJJJMRNHATNKG-UHFFFAOYSA-N ethyl bromoacetate Chemical compound CCOC(=O)CBr PQJJJMRNHATNKG-UHFFFAOYSA-N 0.000 description 12
- 239000012458 free base Substances 0.000 description 12
- 238000004128 high performance liquid chromatography Methods 0.000 description 12
- 239000000047 product Substances 0.000 description 12
- 239000011541 reaction mixture Substances 0.000 description 12
- USFZMSVCRYTOJT-UHFFFAOYSA-N Ammonium acetate Chemical compound N.CC(O)=O USFZMSVCRYTOJT-UHFFFAOYSA-N 0.000 description 11
- 239000005695 Ammonium acetate Substances 0.000 description 11
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical group [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 11
- 235000019257 ammonium acetate Nutrition 0.000 description 11
- 229940043376 ammonium acetate Drugs 0.000 description 11
- 239000000872 buffer Substances 0.000 description 11
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 11
- 238000003756 stirring Methods 0.000 description 11
- MEKOFIRRDATTAG-UHFFFAOYSA-N 2,2,5,8-tetramethyl-3,4-dihydrochromen-6-ol Chemical compound C1CC(C)(C)OC2=C1C(C)=C(O)C=C2C MEKOFIRRDATTAG-UHFFFAOYSA-N 0.000 description 10
- 239000006184 cosolvent Substances 0.000 description 10
- 238000002360 preparation method Methods 0.000 description 10
- 239000002002 slurry Substances 0.000 description 10
- 239000007858 starting material Substances 0.000 description 10
- 239000002585 base Substances 0.000 description 9
- 238000010992 reflux Methods 0.000 description 9
- QCQCHGYLTSGIGX-GHXANHINSA-N 4-[[(3ar,5ar,5br,7ar,9s,11ar,11br,13as)-5a,5b,8,8,11a-pentamethyl-3a-[(5-methylpyridine-3-carbonyl)amino]-2-oxo-1-propan-2-yl-4,5,6,7,7a,9,10,11,11b,12,13,13a-dodecahydro-3h-cyclopenta[a]chrysen-9-yl]oxy]-2,2-dimethyl-4-oxobutanoic acid Chemical compound N([C@@]12CC[C@@]3(C)[C@]4(C)CC[C@H]5C(C)(C)[C@@H](OC(=O)CC(C)(C)C(O)=O)CC[C@]5(C)[C@H]4CC[C@@H]3C1=C(C(C2)=O)C(C)C)C(=O)C1=CN=CC(C)=C1 QCQCHGYLTSGIGX-GHXANHINSA-N 0.000 description 8
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 8
- 239000012043 crude product Substances 0.000 description 8
- LMASLKDDKPUJIF-BPUTZDHNSA-N ethyl (1s,2s)-2-[[(1s)-1-phenylethyl]amino]cyclohexane-1-carboxylate Chemical compound CCOC(=O)[C@H]1CCCC[C@@H]1N[C@@H](C)C1=CC=CC=C1 LMASLKDDKPUJIF-BPUTZDHNSA-N 0.000 description 8
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 8
- NXJCBFBQEVOTOW-UHFFFAOYSA-L palladium(2+);dihydroxide Chemical compound O[Pd]O NXJCBFBQEVOTOW-UHFFFAOYSA-L 0.000 description 8
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 8
- 0 *C(CCCC1)C1=O Chemical compound *C(CCCC1)C1=O 0.000 description 7
- 229930194542 Keto Natural products 0.000 description 7
- 235000019270 ammonium chloride Nutrition 0.000 description 7
- 230000003197 catalytic effect Effects 0.000 description 7
- 239000003153 chemical reaction reagent Substances 0.000 description 7
- 239000001257 hydrogen Substances 0.000 description 7
- 229910052739 hydrogen Inorganic materials 0.000 description 7
- 229910000029 sodium carbonate Inorganic materials 0.000 description 7
- 239000012359 Methanesulfonyl chloride Substances 0.000 description 6
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 6
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 6
- 239000007864 aqueous solution Substances 0.000 description 6
- 125000003118 aryl group Chemical group 0.000 description 6
- 229910052799 carbon Inorganic materials 0.000 description 6
- 238000007796 conventional method Methods 0.000 description 6
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 6
- 125000004185 ester group Chemical group 0.000 description 6
- 125000001072 heteroaryl group Chemical group 0.000 description 6
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 6
- UKVIEHSSVKSQBA-UHFFFAOYSA-N methane;palladium Chemical compound C.[Pd] UKVIEHSSVKSQBA-UHFFFAOYSA-N 0.000 description 6
- AFVFQIVMOAPDHO-UHFFFAOYSA-N methanesulfonic acid Substances CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 6
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 description 6
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 6
- 239000011591 potassium Substances 0.000 description 6
- 229910052700 potassium Inorganic materials 0.000 description 6
- PDELQDSYLBLPQO-UHFFFAOYSA-N 2,3,3a,4,5,6,7,7a-octahydro-1h-indole Chemical group C1CCCC2NCCC21 PDELQDSYLBLPQO-UHFFFAOYSA-N 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical group [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 5
- 235000019445 benzyl alcohol Nutrition 0.000 description 5
- KXZJHVJKXJLBKO-UHFFFAOYSA-N chembl1408157 Chemical compound N=1C2=CC=CC=C2C(C(=O)O)=CC=1C1=CC=C(O)C=C1 KXZJHVJKXJLBKO-UHFFFAOYSA-N 0.000 description 5
- 239000007795 chemical reaction product Substances 0.000 description 5
- 238000003776 cleavage reaction Methods 0.000 description 5
- 238000006345 epimerization reaction Methods 0.000 description 5
- 125000004494 ethyl ester group Chemical group 0.000 description 5
- 125000000524 functional group Chemical group 0.000 description 5
- 238000007327 hydrogenolysis reaction Methods 0.000 description 5
- 238000000746 purification Methods 0.000 description 5
- 230000007017 scission Effects 0.000 description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 4
- 239000012448 Lithium borohydride Substances 0.000 description 4
- NPXOKRUENSOPAO-UHFFFAOYSA-N Raney nickel Chemical compound [Al].[Ni] NPXOKRUENSOPAO-UHFFFAOYSA-N 0.000 description 4
- 125000003342 alkenyl group Chemical group 0.000 description 4
- 125000000304 alkynyl group Chemical group 0.000 description 4
- IJOOHPMOJXWVHK-UHFFFAOYSA-N chlorotrimethylsilane Chemical compound C[Si](C)(C)Cl IJOOHPMOJXWVHK-UHFFFAOYSA-N 0.000 description 4
- 238000004587 chromatography analysis Methods 0.000 description 4
- 238000011097 chromatography purification Methods 0.000 description 4
- 125000000753 cycloalkyl group Chemical group 0.000 description 4
- TWGKMVURZNPDDC-RDTSDOJCSA-N ethyl (2s,3ar,7as)-2,3,3a,4,5,6,7,7a-octahydro-1h-indole-2-carboxylate;hydrochloride Chemical compound Cl.C1CCC[C@@H]2N[C@H](C(=O)OCC)C[C@H]21 TWGKMVURZNPDDC-RDTSDOJCSA-N 0.000 description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 4
- 239000000706 filtrate Substances 0.000 description 4
- 150000003840 hydrochlorides Chemical group 0.000 description 4
- 229910000042 hydrogen bromide Inorganic materials 0.000 description 4
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 4
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 4
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 4
- 238000005984 hydrogenation reaction Methods 0.000 description 4
- 229910052500 inorganic mineral Inorganic materials 0.000 description 4
- ZCSHNCUQKCANBX-UHFFFAOYSA-N lithium diisopropylamide Chemical compound [Li+].CC(C)[N-]C(C)C ZCSHNCUQKCANBX-UHFFFAOYSA-N 0.000 description 4
- 235000010755 mineral Nutrition 0.000 description 4
- 239000011707 mineral Substances 0.000 description 4
- 238000002156 mixing Methods 0.000 description 4
- 238000005191 phase separation Methods 0.000 description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 239000000523 sample Substances 0.000 description 4
- 239000012279 sodium borohydride Substances 0.000 description 4
- 229910000033 sodium borohydride Inorganic materials 0.000 description 4
- 239000008399 tap water Substances 0.000 description 4
- 235000020679 tap water Nutrition 0.000 description 4
- AHZJKOKFZJYCLG-UHFFFAOYSA-K trifluoromethanesulfonate;ytterbium(3+) Chemical compound [Yb+3].[O-]S(=O)(=O)C(F)(F)F.[O-]S(=O)(=O)C(F)(F)F.[O-]S(=O)(=O)C(F)(F)F AHZJKOKFZJYCLG-UHFFFAOYSA-K 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 3
- 238000007126 N-alkylation reaction Methods 0.000 description 3
- 238000013459 approach Methods 0.000 description 3
- 239000000010 aprotic solvent Substances 0.000 description 3
- 239000012298 atmosphere Substances 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 238000002425 crystallisation Methods 0.000 description 3
- 230000008025 crystallization Effects 0.000 description 3
- DZGCGKFAPXFTNM-UHFFFAOYSA-N ethanol;hydron;chloride Chemical compound Cl.CCO DZGCGKFAPXFTNM-UHFFFAOYSA-N 0.000 description 3
- ULGUQTOELKSOAJ-UTLUCORTSA-N ethyl (2s,3ar,7as)-2,3,3a,4,5,6,7,7a-octahydro-1h-indole-2-carboxylate Chemical compound C1CCC[C@@H]2N[C@H](C(=O)OCC)C[C@H]21 ULGUQTOELKSOAJ-UTLUCORTSA-N 0.000 description 3
- 238000001914 filtration Methods 0.000 description 3
- 239000011968 lewis acid catalyst Substances 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 229940098779 methanesulfonic acid Drugs 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- IUGYQRQAERSCNH-UHFFFAOYSA-N pivalic acid Chemical compound CC(C)(C)C(O)=O IUGYQRQAERSCNH-UHFFFAOYSA-N 0.000 description 3
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 3
- 235000019260 propionic acid Nutrition 0.000 description 3
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 230000000717 retained effect Effects 0.000 description 3
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 3
- 235000017557 sodium bicarbonate Nutrition 0.000 description 3
- 238000001308 synthesis method Methods 0.000 description 3
- ITMCEJHCFYSIIV-UHFFFAOYSA-M triflate Chemical compound [O-]S(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-M 0.000 description 3
- DYLIWHYUXAJDOJ-OWOJBTEDSA-N (e)-4-(6-aminopurin-9-yl)but-2-en-1-ol Chemical compound NC1=NC=NC2=C1N=CN2C\C=C\CO DYLIWHYUXAJDOJ-OWOJBTEDSA-N 0.000 description 2
- UUUHXMGGBIUAPW-UHFFFAOYSA-N 1-[1-[2-[[5-amino-2-[[1-[5-(diaminomethylideneamino)-2-[[1-[3-(1h-indol-3-yl)-2-[(5-oxopyrrolidine-2-carbonyl)amino]propanoyl]pyrrolidine-2-carbonyl]amino]pentanoyl]pyrrolidine-2-carbonyl]amino]-5-oxopentanoyl]amino]-3-methylpentanoyl]pyrrolidine-2-carbon Chemical compound C1CCC(C(=O)N2C(CCC2)C(O)=O)N1C(=O)C(C(C)CC)NC(=O)C(CCC(N)=O)NC(=O)C1CCCN1C(=O)C(CCCN=C(N)N)NC(=O)C1CCCN1C(=O)C(CC=1C2=CC=CC=C2NC=1)NC(=O)C1CCC(=O)N1 UUUHXMGGBIUAPW-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- XPDWGBQVDMORPB-UHFFFAOYSA-N Fluoroform Chemical compound FC(F)F XPDWGBQVDMORPB-UHFFFAOYSA-N 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 102000004270 Peptidyl-Dipeptidase A Human genes 0.000 description 2
- 108090000882 Peptidyl-Dipeptidase A Proteins 0.000 description 2
- 239000007868 Raney catalyst Substances 0.000 description 2
- 229910000564 Raney nickel Inorganic materials 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 2
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 2
- 241000282898 Sus scrofa Species 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- GCWPGEWXYDEQAY-NKWVEPMBSA-N [(1r,2r)-2-aminocyclohexyl]methanol Chemical compound N[C@@H]1CCCC[C@H]1CO GCWPGEWXYDEQAY-NKWVEPMBSA-N 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- 125000004644 alkyl sulfinyl group Chemical group 0.000 description 2
- 239000012300 argon atmosphere Substances 0.000 description 2
- 125000005135 aryl sulfinyl group Chemical group 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- PASDCCFISLVPSO-UHFFFAOYSA-N benzoyl chloride Chemical compound ClC(=O)C1=CC=CC=C1 PASDCCFISLVPSO-UHFFFAOYSA-N 0.000 description 2
- 238000009903 catalytic hydrogenation reaction Methods 0.000 description 2
- 239000003638 chemical reducing agent Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 238000010168 coupling process Methods 0.000 description 2
- 238000005859 coupling reaction Methods 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- NZNMSOFKMUBTKW-UHFFFAOYSA-M cyclohexanecarboxylate Chemical compound [O-]C(=O)C1CCCCC1 NZNMSOFKMUBTKW-UHFFFAOYSA-M 0.000 description 2
- PAFZNILMFXTMIY-UHFFFAOYSA-N cyclohexylamine Chemical compound NC1CCCCC1 PAFZNILMFXTMIY-UHFFFAOYSA-N 0.000 description 2
- 238000000354 decomposition reaction Methods 0.000 description 2
- ORMNPSYMZOGSSV-UHFFFAOYSA-N dinitrooxymercury Chemical compound [Hg+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O ORMNPSYMZOGSSV-UHFFFAOYSA-N 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 238000011031 large-scale manufacturing process Methods 0.000 description 2
- YNESATAKKCNGOF-UHFFFAOYSA-N lithium bis(trimethylsilyl)amide Chemical compound [Li+].C[Si](C)(C)[N-][Si](C)(C)C YNESATAKKCNGOF-UHFFFAOYSA-N 0.000 description 2
- 210000004185 liver Anatomy 0.000 description 2
- 150000002825 nitriles Chemical group 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- WLJVXDMOQOGPHL-UHFFFAOYSA-N phenylacetic acid Chemical compound OC(=O)CC1=CC=CC=C1 WLJVXDMOQOGPHL-UHFFFAOYSA-N 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 239000002244 precipitate Substances 0.000 description 2
- 239000013557 residual solvent Substances 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- 239000012312 sodium hydride Substances 0.000 description 2
- 229910000104 sodium hydride Inorganic materials 0.000 description 2
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- AHYHTSYNOHNUSH-HXFGRODQSA-N trandolaprilat Chemical compound C([C@H](N[C@@H](C)C(=O)N1[C@@H](C[C@H]2CCCC[C@@H]21)C(O)=O)C(O)=O)CC1=CC=CC=C1 AHYHTSYNOHNUSH-HXFGRODQSA-N 0.000 description 2
- FWZVVDOFZQMNKT-LZWOXQAQSA-N (1S,2R)-2-ethyl-N-[(1S)-1-phenylethyl]cyclohexan-1-amine Chemical compound C(C)[C@H]1[C@H](CCCC1)N[C@@H](C)C1=CC=CC=C1 FWZVVDOFZQMNKT-LZWOXQAQSA-N 0.000 description 1
- GHOCRGGRRGZNJN-RDBSUJKOSA-N (2S,3aR,7aS)-1-benzoyl-2,3,3a,4,5,6,7,7a-octahydroindole-2-carboxylic acid Chemical group N1([C@H]2CCCC[C@@H]2C[C@H]1C(=O)O)C(=O)C1=CC=CC=C1 GHOCRGGRRGZNJN-RDBSUJKOSA-N 0.000 description 1
- YONLFQNRGZXBBF-ZIAGYGMSSA-N (2r,3r)-2,3-dibenzoyloxybutanedioic acid Chemical compound O([C@@H](C(=O)O)[C@@H](OC(=O)C=1C=CC=CC=1)C(O)=O)C(=O)C1=CC=CC=C1 YONLFQNRGZXBBF-ZIAGYGMSSA-N 0.000 description 1
- CEIWXEQZZZHLDM-AAEUAGOBSA-N (2s)-2-[[(2s)-1-ethoxy-1-oxo-4-phenylbutan-2-yl]amino]propanoic acid Chemical compound CCOC(=O)[C@@H](N[C@@H](C)C(O)=O)CCC1=CC=CC=C1 CEIWXEQZZZHLDM-AAEUAGOBSA-N 0.000 description 1
- SGTNSNPWRIOYBX-UHFFFAOYSA-N 2-(3,4-dimethoxyphenyl)-5-{[2-(3,4-dimethoxyphenyl)ethyl](methyl)amino}-2-(propan-2-yl)pentanenitrile Chemical compound C1=C(OC)C(OC)=CC=C1CCN(C)CCCC(C#N)(C(C)C)C1=CC=C(OC)C(OC)=C1 SGTNSNPWRIOYBX-UHFFFAOYSA-N 0.000 description 1
- 239000005541 ACE inhibitor Substances 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- ZITRDHAWJNAAJZ-VIFPVBQESA-N C[C@@H]1CC2(CC2)C2(CC2)CC1 Chemical compound C[C@@H]1CC2(CC2)C2(CC2)CC1 ZITRDHAWJNAAJZ-VIFPVBQESA-N 0.000 description 1
- QSFGDCFSEOZANA-ZGVCCVRISA-N C[C@H](CCCC1)[C@H]1C(CC#N)N[C@@H](C#C)P Chemical compound C[C@H](CCCC1)[C@H]1C(CC#N)N[C@@H](C#C)P QSFGDCFSEOZANA-ZGVCCVRISA-N 0.000 description 1
- 229940127291 Calcium channel antagonist Drugs 0.000 description 1
- 108090000371 Esterases Proteins 0.000 description 1
- 206010019280 Heart failures Diseases 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- ODZTXUXIYGJLMC-UHFFFAOYSA-N OC(CCCC1)C1=O Chemical compound OC(CCCC1)C1=O ODZTXUXIYGJLMC-UHFFFAOYSA-N 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- SAJUROZXXGFMST-UHFFFAOYSA-L [C].[Pt](O)O Chemical compound [C].[Pt](O)O SAJUROZXXGFMST-UHFFFAOYSA-L 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000012042 active reagent Substances 0.000 description 1
- 229940044094 angiotensin-converting-enzyme inhibitor Drugs 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- ARGCRCXTJMQKNA-ILXRZTDVSA-N benzyl (2s,3ar,7as)-2,3,3a,4,5,6,7,7a-octahydro-1h-indole-2-carboxylate Chemical compound O=C([C@H]1N[C@H]2CCCC[C@@H]2C1)OCC1=CC=CC=C1 ARGCRCXTJMQKNA-ILXRZTDVSA-N 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 239000000480 calcium channel blocker Substances 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical group 0.000 description 1
- 239000002327 cardiovascular agent Substances 0.000 description 1
- 229940125692 cardiovascular agent Drugs 0.000 description 1
- 238000013375 chromatographic separation Methods 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- ATDGTVJJHBUTRL-UHFFFAOYSA-N cyanogen bromide Chemical compound BrC#N ATDGTVJJHBUTRL-UHFFFAOYSA-N 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 238000010511 deprotection reaction Methods 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 239000002934 diuretic Substances 0.000 description 1
- 229940030606 diuretics Drugs 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 150000002081 enamines Chemical class 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 230000007515 enzymatic degradation Effects 0.000 description 1
- VEUUMBGHMNQHGO-UHFFFAOYSA-N ethyl chloroacetate Chemical compound CCOC(=O)CCl VEUUMBGHMNQHGO-UHFFFAOYSA-N 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 230000008570 general process Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000003402 intramolecular cyclocondensation reaction Methods 0.000 description 1
- 238000006317 isomerization reaction Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000002715 modification method Methods 0.000 description 1
- 239000012452 mother liquor Substances 0.000 description 1
- 239000002105 nanoparticle Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 229940126701 oral medication Drugs 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 239000003279 phenylacetic acid Substances 0.000 description 1
- 229960003424 phenylacetic acid Drugs 0.000 description 1
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Substances [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 229940002612 prodrug Drugs 0.000 description 1
- 239000000651 prodrug Substances 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 230000000707 stereoselective effect Effects 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 229960001722 verapamil Drugs 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 150000003747 ytterbium compounds Chemical class 0.000 description 1
- AWSFICBXMUKWSK-UHFFFAOYSA-N ytterbium(3+) Chemical compound [Yb+3] AWSFICBXMUKWSK-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/30—Indoles; Hydrogenated indoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to carbon atoms of the hetero ring
- C07D209/42—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Life Sciences & Earth Sciences (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Indole Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US22603009P | 2009-07-16 | 2009-07-16 | |
| US61/226,030 | 2009-07-16 | ||
| PCT/US2010/042219 WO2011009021A1 (en) | 2009-07-16 | 2010-07-16 | Processes for the synthesis of (2s, 3ar, 7as)-octahydro-1h-indole carboxylic acid as an intermediate for trandolapril |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2012533563A true JP2012533563A (ja) | 2012-12-27 |
Family
ID=42585097
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2012520807A Pending JP2012533563A (ja) | 2009-07-16 | 2010-07-16 | トランドラプリル用の中間体である(2S,3aR,7aS)−オクタヒドロ−1H−インドールカルボン酸の合成法 |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US8288565B2 (enExample) |
| EP (1) | EP2454235A1 (enExample) |
| JP (1) | JP2012533563A (enExample) |
| CN (1) | CN102498098A (enExample) |
| AU (1) | AU2010273259B2 (enExample) |
| CA (1) | CA2768239A1 (enExample) |
| CO (1) | CO6491043A2 (enExample) |
| IN (1) | IN2012DN00453A (enExample) |
| RU (1) | RU2012103753A (enExample) |
| WO (1) | WO2011009021A1 (enExample) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111620788B (zh) * | 2020-04-20 | 2022-09-30 | 广东莱佛士制药技术有限公司 | 一种制备(2s,3s)-3-氨基-二环[2.2.2]辛烷-2-甲酸酯的方法 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002534412A (ja) * | 1999-01-07 | 2002-10-15 | ビーエーエスエフ アクチェンゲゼルシャフト | (2s,4r,9s)−オクタヒドロ−1h−インドール−2−カルボン酸の製法及びその中間体 |
| WO2004092133A1 (fr) * | 2003-04-15 | 2004-10-28 | Les Laboratoires Servier | NOUVEAU PROCEDE DE SYNTHESE DE L'ACIDE (2S, 3aS, 7aS)-PERHYDROINDOLE-2-CARBOXYLIQUE ET DE SES ESTERS, ET APPLICATION A LA SYNTHESE DU PERINDOPRIL |
| WO2005054194A1 (en) * | 2003-11-25 | 2005-06-16 | Texcontor Etablissement | A method for the preparation of (2s, 3ar, 7as)-octahydro-1h-indole-2-carboxylic acid as key intermediate in the preparation of trandolapril by reacting a cyclohexyl aziridine with a dialkyl malonate |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US487932A (en) | 1892-12-13 | George b | ||
| US4350704A (en) | 1980-10-06 | 1982-09-21 | Warner-Lambert Company | Substituted acyl derivatives of octahydro-1H-indole-2-carboxylic acids |
| GR78413B (enExample) | 1981-12-29 | 1984-09-27 | Hoechst Ag | |
| US4490386A (en) | 1982-09-23 | 1984-12-25 | Warner-Lambert Company | Phosphate salts of 1-[2-[(1-alkoxycarbonyl-3-aralkyl)-amino]-1-oxoalkyl]octahydro-1H-indole-2-carboxylic acids, preparation of, and medical compositions thereof |
| US4556655A (en) | 1984-09-24 | 1985-12-03 | Schering Corporation | Antihypertensive compounds having both diuretic and angiotensin converting enzyme inhibitory activity |
| FR2605630B1 (fr) | 1986-10-22 | 1989-06-30 | Roussel Uclaf | Procede de preparation de derives de l'octahydroindole et intermediaires de preparation |
| JPWO2004065368A1 (ja) | 2003-01-21 | 2006-05-18 | 大原薬品工業株式会社 | トランドラプリル合成中間体の製造方法 |
| SI21507A (sl) | 2003-05-16 | 2004-12-31 | LEK farmacevtska dru�ba d.d. | Postopek za pripravo spojin z ace inhibitornim delovanjem |
| JP2007512260A (ja) | 2003-11-28 | 2007-05-17 | アツァト・ファーマシューティカルズ・イングリーディエンツ・アーゲー | {N−[1−(S)−カルボアルコキシ−3−フェニルプロピル]−S−アラニル−2S,3aR,7aS−オクタヒドロインドール−2−カルボン酸}化合物類の製造方法 |
| WO2006014916A2 (en) | 2004-07-26 | 2006-02-09 | Dr. Reddy's Laboratories Ltd. | Preparation of trandolapril |
| AU2005327440A1 (en) | 2005-02-14 | 2006-08-17 | Lupin Limited | Improved process for preparation of highly pure trandolapril |
| ATE386718T1 (de) | 2005-05-06 | 2008-03-15 | Sochinaz Sa | Verfahren zur herstellung von (2s, 3ar, 7as)- octahydroindole-2-carbonsäure und umwandlung zum trandolapril |
| CA2614099A1 (en) | 2005-07-05 | 2007-01-11 | Cipla Limited | Process for the synthesis of the ace inhibitor |
| JP5292400B2 (ja) | 2007-07-24 | 2013-09-18 | ブリストル−マイヤーズ スクイブ カンパニー | ケモカイン受容体活性のモジュレーターとしてのピペリジン誘導体 |
| CN101423490B (zh) * | 2008-06-16 | 2012-07-18 | 重庆南松医药科技股份有限公司 | 群多普利关键中间体(2S,3aR,7as)-八氢-1H-吲哚-2-羧酸的合成方法 |
-
2010
- 2010-07-16 IN IN453DEN2012 patent/IN2012DN00453A/en unknown
- 2010-07-16 WO PCT/US2010/042219 patent/WO2011009021A1/en not_active Ceased
- 2010-07-16 EP EP10737165A patent/EP2454235A1/en not_active Withdrawn
- 2010-07-16 AU AU2010273259A patent/AU2010273259B2/en not_active Ceased
- 2010-07-16 US US12/837,686 patent/US8288565B2/en not_active Expired - Fee Related
- 2010-07-16 CA CA2768239A patent/CA2768239A1/en not_active Abandoned
- 2010-07-16 CN CN2010800413803A patent/CN102498098A/zh active Pending
- 2010-07-16 JP JP2012520807A patent/JP2012533563A/ja active Pending
- 2010-07-16 RU RU2012103753/04A patent/RU2012103753A/ru not_active Application Discontinuation
-
2012
- 2012-01-16 CO CO12005568A patent/CO6491043A2/es active IP Right Grant
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002534412A (ja) * | 1999-01-07 | 2002-10-15 | ビーエーエスエフ アクチェンゲゼルシャフト | (2s,4r,9s)−オクタヒドロ−1h−インドール−2−カルボン酸の製法及びその中間体 |
| WO2004092133A1 (fr) * | 2003-04-15 | 2004-10-28 | Les Laboratoires Servier | NOUVEAU PROCEDE DE SYNTHESE DE L'ACIDE (2S, 3aS, 7aS)-PERHYDROINDOLE-2-CARBOXYLIQUE ET DE SES ESTERS, ET APPLICATION A LA SYNTHESE DU PERINDOPRIL |
| WO2005054194A1 (en) * | 2003-11-25 | 2005-06-16 | Texcontor Etablissement | A method for the preparation of (2s, 3ar, 7as)-octahydro-1h-indole-2-carboxylic acid as key intermediate in the preparation of trandolapril by reacting a cyclohexyl aziridine with a dialkyl malonate |
Non-Patent Citations (3)
| Title |
|---|
| JPN6014028795; HAYASHI, Yujiro, et al.: Journal of the American Chemical Society 118(23), 1996, p. 5502-5503 * |
| JPN6014028799; BLANKLEY, C. J., et al.: Journal of Medicinal Chemistry 30, 1987, p. 992-998 * |
| JPN6014028801; BRION, F., et al.: Tetrahedron Letters 33(34), 1992, p. 4889-4892 * |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2010273259A1 (en) | 2012-02-09 |
| EP2454235A1 (en) | 2012-05-23 |
| US8288565B2 (en) | 2012-10-16 |
| AU2010273259B2 (en) | 2013-03-07 |
| WO2011009021A1 (en) | 2011-01-20 |
| CA2768239A1 (en) | 2011-01-20 |
| CN102498098A (zh) | 2012-06-13 |
| RU2012103753A (ru) | 2013-08-27 |
| US20110065930A1 (en) | 2011-03-17 |
| CO6491043A2 (es) | 2012-07-31 |
| IN2012DN00453A (enExample) | 2015-05-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9938239B2 (en) | Method for preparing silodosin | |
| CN103044418B (zh) | (s,s)-2,8-二氮杂双环[4,3,0]壬烷的不对称合成方法、相关原料及制备方法 | |
| WO2008082508A2 (en) | Process for the preparation of 6,6-dimethyl-3-azabicyclo- [3.1.0]- hexane compounds utilizing bisulfite intermediate | |
| CA2634397A1 (en) | Process for the preparation of 6,6-dimethyl-3-azabicyclo[3.1.0|-hexane compounds and enantiomeric salts thereof | |
| AU2003277353A1 (en) | Process for the preparation of (s,s)-cis-2-benzhydryl-3-benzylaminoquinuclidine | |
| US20100076204A1 (en) | Process for the preparation of levetiracetam | |
| TWI438188B (zh) | 用於合成醫藥品之中間化合物的製造方法 | |
| JP5254836B2 (ja) | ベナゼプリル及びその類似体の生産に有用な中間体の速度論的分離 | |
| US8288565B2 (en) | Process for the synthesis of (2S,3AR,7AS)-octahydro-1H-indole carboxylic acid as an intermediate for trandolapril | |
| CN1047167C (zh) | 用于制备毒扁豆碱的中间体的对映选择性合成方法 | |
| US5380849A (en) | Process for optically pure decahydroisoqiunolines | |
| JP4780437B2 (ja) | 2−オキソイミダゾリジン誘導体の製造方法 | |
| CN1989097A (zh) | 用于制备非对映异构体富集的化合物的方法 | |
| HK1172025A (en) | Processes for the synthesis of (2s, 3ar, 7as)-octahydro-1h-indole carboxylic acid as an intermediate for trandolapril | |
| EP1679072B1 (en) | Process for for synthesis of (2S,3aS,7aS)-1-(S)-alanyl-octahydro-1H-indole-2- carboxylic acid derivatives and use in the synthesis of perindopril | |
| US6531594B2 (en) | Process for producing 1H-3-aminopyrrolidine and derivatives thereof | |
| JP4126921B2 (ja) | 光学活性なβ−フェニルアラニン誘導体の製造方法 | |
| JP3204368B2 (ja) | 光学活性アミド類の製法 | |
| JP4187822B2 (ja) | 光学活性4−ヒドロキシ−2−ピロリドンの製造方法 | |
| JP2009507783A (ja) | 高光学純度を有するキラル3−ヒドロキシピロリジン化合物及びその誘導体の製造方法 | |
| US20080242861A1 (en) | Synthesis of amino-protected cyclohexane-1,4-diyldimethanamine and its derivatives | |
| JP2002316977A (ja) | 光学活性1h−3−アミノピロリジン化合物の製造方法 | |
| JPH11158162A (ja) | 光学活性ピペラジンカルボン酸エステルの製造方法 | |
| SK1092004A3 (en) | Method of production of (R)-N,N-di-isopropyl-3-(2-hydroxy-5- methylphenyl)-3-phenylpropylamine | |
| EP1792896A1 (en) | Process for the preparation of perindopril and salts thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20130527 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20140710 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140715 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20141209 |