EP1431031A2 - Druckform und Verfahren zum Ändern ihrer Benetzungseigenschaften - Google Patents
Druckform und Verfahren zum Ändern ihrer Benetzungseigenschaften Download PDFInfo
- Publication number
- EP1431031A2 EP1431031A2 EP03027825A EP03027825A EP1431031A2 EP 1431031 A2 EP1431031 A2 EP 1431031A2 EP 03027825 A EP03027825 A EP 03027825A EP 03027825 A EP03027825 A EP 03027825A EP 1431031 A2 EP1431031 A2 EP 1431031A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- printing form
- silicon
- groups
- hydrophobic
- end groups
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B41—PRINTING; LINING MACHINES; TYPEWRITERS; STAMPS
- B41C—PROCESSES FOR THE MANUFACTURE OR REPRODUCTION OF PRINTING SURFACES
- B41C1/00—Forme preparation
- B41C1/10—Forme preparation for lithographic printing; Master sheets for transferring a lithographic image to the forme
- B41C1/1041—Forme preparation for lithographic printing; Master sheets for transferring a lithographic image to the forme by modification of the lithographic properties without removal or addition of material, e.g. by the mere generation of a lithographic pattern
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B41—PRINTING; LINING MACHINES; TYPEWRITERS; STAMPS
- B41N—PRINTING PLATES OR FOILS; MATERIALS FOR SURFACES USED IN PRINTING MACHINES FOR PRINTING, INKING, DAMPING, OR THE LIKE; PREPARING SUCH SURFACES FOR USE AND CONSERVING THEM
- B41N1/00—Printing plates or foils; Materials therefor
- B41N1/006—Printing plates or foils; Materials therefor made entirely of inorganic materials other than natural stone or metals, e.g. ceramics, carbide materials, ferroelectric materials
Definitions
- the invention relates to a printing form with a surface which is inorganic has bonded silicon and one of hydrophilic and hydrophobic areas existing pattern carries, the hydrophilic areas a first chemical State and the hydrophobic areas a second, different from the first state chemical state. Furthermore, the invention relates to a method for Changing the wetting properties of a printing form with a surface, which has inorganically bonded silicon, the surface being in a first chemical state with the first wetting property and a subset all areas of the surface into a second chemical state with a second Wetting property by changing the chemical end groups of the surface brought.
- a printing form which with a amorphous semiconductor is coated.
- the disordered amorphous state of the semiconductor can be transformed into a higher order crystalline state with the help of a laser beam change.
- the semiconductor surface is rougher, so that the Rearrangement of the semiconductor surface leads to liquids in the area of the rougher Adhesive surface better than in the amorphous smooth areas.
- This Process made printing form is due to the minimum size of the crystalline areas limited.
- a printing form with a surface which has silicon, in a first chemical state with a first Wetting property and in a second chemical state with a second Wetting property can be brought.
- the local wetting property i.e. the local hydrophilic or hydrophobic wetting property of the printing form
- the local wetting property can via the Change the chemical end groups of the surface with appropriate different electronic properties can be controlled.
- silicon is selected as the semiconductor in document WO 00/21753.
- the surface is first placed in a hydrophobic state, for example with SiH, SiH 2 and / or SiH 3 groups on the surface.
- the hydrophobic end group is then locally replaced by a hydrophilic end group or converted into one, so that, for example, SiOH, SiOSi and / or SiO end groups replace the hydrophobic end groups.
- the object of the present invention is to provide an alternative printing form with areas propose greater hydrophobicity or in a strong hydrophobic state and a method for changing their wetting properties locally and repeatedly specify.
- a printing form according to the invention in particular an offset printing form, comprises a surface which has inorganically bonded silicon and bears a pattern consisting of hydrophilic and hydrophobic areas, the hydrophilic areas having a first chemical state and the hydrophobic areas having a second chemical which is different from the first chemical state Condition.
- the printing form according to the invention is characterized in that in at least one of the hydrophobic regions, preferably in all hydrophobic regions, the surface has silicon atoms to which at least one organic end group is bonded, preferably a number of a certain organic end group is bound in each case.
- the silicon atoms are not only exactly substituted with a CH 3 group or with an OCH 3 group.
- the organic end group preferably not only has exactly one CH 3 group if the sum of the atomic mass of the atoms bonded to the silicon atoms in the end group is below 33.5 u.
- the sum of the atomic mass of the atoms which are bonded to the silicon atoms in the end groups and which are not silicon atoms is preferably greater than 33.5 u, in particular the organic end group can be heavier than 33.5 u.
- the atomic mass is specified in relative atomic mass units u.
- a methyl end group has about 15 u atomic mass units
- a methanol end group has about 31 u
- a fluorine atom has about 19 u.
- An organic end group can, in addition to the carbon derivatives usually referred to by the person skilled in the art as an organic end group in the context of the inventive concept, also be an end group which comprises a non-organic base chain, in particular an Si-O chain, organic substituents, in particular, on valences of the base chain CH 3 groups.
- the printing form according to the invention advantageously exhibits in hydrophobic areas strong hydrophobic end groups.
- so many organic End groups be substituted that the wetting property, the hydrophobicity, the Part of the surface determined by the property of the organic end group is.
- the difference in the wetting properties of the hydrophilic and hydrophobic Areas can be varied with the choice of an organic end group, in particular very high contrast or strong.
- the organic end group can be replaced by a Reactant be generated or emerge from a reactant.
- the surface of the printing form with inorganic silicon can be made of pure Silicon, amorphous, nanocrystalline or polycrystalline or crystalline. Besides pure However, silicon can also be used in silicon nitride ceramics or their layer systems become.
- the silicon is not organically bound as a copolymer with a carbon inacromolecule.
- the silicon nitride ceramics can consist stoichiometrically of Si 3 N 4 or non stoichiometrically of silicon nitride (Si x N y ).
- the silicon nitride ceramics can be amorphous.
- the surface of the printing form can consist of a stoichiometric or non-stoichiometric silicon oxide ceramic (glass ceramic) or of a mixed phase of silicon, oxygen and nitrogen, a silicon oxynitride ceramic (Si x O y N z ).
- the silicon is also inorganic in ceramic form. A large variety in the selection and possible arrangements of organic end groups can advantageously be realized, varied and / or controlled on such surfaces. Silicon nitride ceramics in particular can have greater mechanical strength than pure silicon.
- the surface of the printing form according to the invention is taken up as a thin amorphous film on a metallic carrier, which for example has titanium (preferred), aluminum, chromium or gold.
- the carrier can be a metal sheet, such as titanium sheet.
- the carrier can be plate-shaped, cylindrical or sleeve-shaped.
- the amorphous film is less than 500 micrometers thick, the film thickness is preferably 0.5 to 10 micrometers.
- the printing form can also consist entirely of the amorphous material.
- a hydrophilic state of a surface which has inorganically bonded silicon can be achieved by oxide and / or hydroxide end groups, for example silanol groups (SiOH).
- Such a surface typically has several OH groups per square nanometer (nm 2 ).
- This state of pronounced hydrophilicity can be described by means of wet chemical reactions, as described, for example, in document WO 00/21753, the disclosure of which is incorporated by reference, or by thermal heating or photochemically, for example by means of ozonization in a normal atmosphere with oxygen and / or water molecules or by means of photodissociation.
- a laser light source which emits in the infrared or visible spectral range can preferably be used for the thermal heating.
- a photochemical conversion can be effected by means of UV or VUV radiation (vacuum ultraviolet radiation with a wavelength of less than 200 nm, in particular greater than 100 nm).
- a hydrophobic state of a surface which has inorganic bonded silicon can be brought about by means of hydrogen termination, for example SiH, SiH 2 and / or SiH 3 groups (hydrosilanes).
- the surface which has silicon can be brought into a strongly hydrophobic state by terminating with methyl end groups, in particular -Si (CH 3 ) 3 , -O-Si (CH 3 ) 3 or ⁇ Si-C (CH 3 ) 3 , or a termination with unsubstituted and / or partially or completely halogenated, in particular chlorinated or fluorinated, alkyl end groups, for example CF 3 groups, is carried out.
- silicon atoms can carry several organic end groups on the surface.
- a silicon atom on the surface can be substituted several times.
- the organic end groups in hydrophobic areas can in particular have fewer than 21 carbon atoms.
- the number of carbon atoms can be a natural number from the interval from 1 to 20 (with an interval margin).
- 1 to 6 carbon atoms can preferably be present in a chain of the organic end group. Short chains are preferred due to the shorter time and amount of energy required for removal.
- organic End groups especially ring or chain end groups, by means of an Si-C bond and / or Si-O-C bond and / or an Si-O-Si-C bond.
- a method according to the invention for changing the wetting properties of a printing form with a surface which has inorganically bonded silicon the surface (preferably all areas of the surface which form a printing surface) in a first chemical state with a first Wetting property is brought and a subset, in particular a subset of all areas, the surface is brought into a second chemical state with a second wetting property by changing the chemical end group of the surface.
- the process according to the invention is characterized in that organic end groups are attached to silicon atoms on the surface in such a way that the silicon atoms are not only substituted with a CH 3 group or with an OCH 3 group.
- the sum of the atomic masses of the atoms bonded to the silicon atoms on the surface, which are not silicon atoms, can be greater than 33.5 u.
- the first wetting property can be hydrophilic and the second wetting property can be hydrophobic or the first wetting property can be hydrophobic and the second wetting property can be hydrophilic.
- the process according to the invention can be carried out with particular advantage using a printing form whose surface consists of amorphous, nanocrystalline, polycrystalline or crystalline silicon or is a stoichiometric or non-stoichiometric silicon ceramic which has oxygen and / or nitrogen.
- a printing form whose surface consists of amorphous, nanocrystalline, polycrystalline or crystalline silicon or is a stoichiometric or non-stoichiometric silicon ceramic which has oxygen and / or nitrogen.
- unsubstituted and / or halogenated for example partially and / or completely chlorinated and / or partially fluorinated and / or fully fluorinated, end groups, in particular aryl end groups or alkyl end groups, can be bound as organic end groups in hydrophobic areas of the printing form.
- the organic end groups in the hydrophobic regions can be CH 3 end groups and / or CF 3 end groups.
- the chain molecules can have CH 3 end groups and / or CF 3 end groups
- the inventive method for changing the wetting properties of a Printing form serves to create a structure of hydrophilic and hydrophobic Areas on the printing form, so that in an offset printing process duplications of the structure can be created.
- the method for changing the Wetting properties of the second chemical state through localized processing with a controlled light source such that the second chemical state is so is generated that he an image information to be printed or its negative (not to printing image information).
- a direct attachment of alkyl groups or fluoroalkyl groups to the surface of the printing form via Si-C bonds can be achieved by photoinitiating halosilanes, for example Cl-Si (CH 3 ) 3 , alcohols, alkenes and / or alkynes.
- halosilanes for example Cl-Si (CH 3 ) 3
- alcohols for example alcohols, alkenes and / or alkynes.
- a solution with reactive halogen-containing molecules such as iodoform is possible.
- Alkoxyl monolayers in other words alkyl groups, which are fixed on the surface via Si-OC bonds, can react via alcohols (R-OH), preferably with four or five carbon atoms in a chain, since these substances are not very dangerous for humans and the environment , or aldehydes (R-CHO) with a hydrogen-terminated, halogen-terminated or oxide-terminated surface which has silicon can be obtained.
- R is an unsubstituted alkyl group or aryl group or a partially or fully fluorinated alkyl group or aryl group.
- the hydrocarbon group can be chain or ring-shaped, in particular aromatic, for example a phenyl ring (C 6 H 5 -) or a substituted phenyl ring.
- the reaction can be initiated and / or accelerated by exposure to light, preferably with UV light, for photochemical activation.
- the chain or ring-shaped aromatic unsubstituted or fluorinated carbon end groups can have a different number of carbon atoms, preferably 1 to 6 carbon atoms.
- alkyl groups can be attached via Si-O-Si-C bonds by siloxane chemistry with alkylchlorosilanes, alkylalkoxysilanes, and / or alkylaminosilanes on an oxide-covered surface which has silicon.
- the unsubstituted or fluorinated alkyl group can have a chain of several carbon atoms, preferably 1 to 6 carbon atoms, on which there is one CH 3 or one CF 3 end group or more CH 3 or CF 3 end groups.
- the organic end groups each have a chain of several carbon atoms on which there are CH 3 or CF 3 groups.
- the length of the carbon chain has little influence on the hydrophobic behavior.
- long chains up to 20 carbon atoms
- additional stabilization by lateral van der Waals interactions can advantageously take place; a self-assembling monolayer (self-assembled monolayer, SAM) can form
- SAM self-assembled monolayer
- a short carbon chain and an arrangement in which not every surface atom has an organic end group is sufficient for the printing process.
- the arrangement can have a relatively low surface density of the organic end groups. Typical concentrations are between 10 14 and 10 11 end groups per cm 2 .
- a sufficiently high concentration must be achieved in order to achieve a sufficiently strong hydrophobicity; at the same time, the concentration should be as low as possible or necessary, since subsequent removal of the organic end groups in the case of small end group molecules and / or a low surface density is advantageous is facilitated.
- the surface of a printing form can be reacted with much more reactive, in particular radical, starting molecules can be achieved.
- a organic end group by reaction with iodoform and / or with trimethylene methane derivatives, which occur in a triplet and / or a dipolar singlet state can be connected.
- a stable precursor molecule it is advantageous this is the 1,1-dialkoxy-2-methylene cyclopropane (DMCP). From methylene cyclopropane derivatives can be obtained thermally or by irradiation dipolar trimethylene derivatives be generated.
- the first example relates to the attachment of a hydrophobic layer Alkyl end groups or fluoroalkyl end groups by means of Si-C bonds.
- the Si-C bonds have a relatively high stability.
- Reactive hydrocarbons such as alkenes and / or alkynes
- Photoactivation can be directly attached to silicon with the formation of Si-C bonds ( ⁇ Si-R).
- the A starting point for such a connection can be particularly advantageous be hydrogen-terminated silicon surface.
- a process like this hydrogen-terminated silicon surface can be obtained is in the document WO 00/21753 described. This document is incorporated by reference in the Disclosure content of this presentation added.
- the problem of the relatively slow Course of the reaction in which, under normal conditions, a partial Oxidation of the silicon surface can use pure chemicals and reactive precursor molecules, for example radicals. At the The use of such reactive precursor molecules results in a considerable acceleration of the alkylation process.
- the surface is spatially selective, that is, in Partial areas, are oxidized and thus hydrophilized.
- a deletion the image can be achieved by oxidizing the entire surface and / or again is hydrogen terminated so that the initial state is restored.
- aryl or alkyl end groups in particular methyl or fluoromethyl end groups , depending on the irradiation time, radiation power and wavelength with increasing number of carbon atoms in the organic end group, may not always completely, but only partially oxidized and removed.
- the remaining methylene, methyl or fluoromethyl end groups are oxidized to aldehyde or carboxyl groups and thus also hydrophilic.
- a switch from hydrophobic to hydrophilic is also possible by converting the organic end group, for example CH 3 , of the organic chain without completely removing the organic chain.
- alkyl groups In the case of a surface made of a silicon nitride ceramic, additional silylamine groups can arise. For easy removal, it therefore makes sense to choose the alkyl groups as short-chain as possible. Chain lengths of 1 to 5 carbon atoms are preferred.
- the alkyl groups are completely removed for new imaging. Removal can be achieved photochemically with UV or VUV light sources, in particular lasers, or photothermally with infrared or visible light sources, in particular lasers.
- a second example relates to the attachment of a hydrophobic layer with aryl or Alkyl end groups or fluoroalkyl end groups by means of Si-O-C bonds.
- connection of the aryl radical or alkyl radical takes place on the basis of reactions of primary alcohols (R-OH) and / or secondary alcohols (R- (OH) 2 ) and / or aldehydes (R-CHO) with a hydrogen-terminated, halogen-terminated or oxide-terminated silicon surface or fluoroalkyl residue on the surface via an oxygen bridge to the carbon (Si-OR).
- R-OH primary alcohols
- R- (OH) 2 / or secondary alcohols
- R-CHO aldehydes
- Si-OR oxygen bridge to the carbon
- Alcohols with 3 or 4 carbon atoms are preferred.
- the secondary Under certain conditions, alcohols can have O-bonds between two form organic end groups, giving the modified surface additional stability is awarded. According to the processes described in the first example, the Exit scheduling can be restored.
- the third example relates to binding a hydrophobic layer with aryl or Alkyl end groups or fluoroalkyl end groups by means of Si-O-Si-C bonds.
- the starting point is an oxidized hydrophilic silicon, silicon oxide or silicon nitride surface, which is at least partially covered with silanol and / or silylamine groups. Molecules with hydrophobic alkyl end groups or fluorinated alkyl end groups are chemisorbed on this surface (Si-O-Si-R).
- This hydrophobic surface can be produced with alkyltrimethoxysilanes, for example CH 3 - (CH 2 ) 2 -Si- (OCH 3 ) 3 , or fluoroalkylmethoxisilanes, for example CF 3 - (CH 2 ) 2 -Si- (OCH 3 ) 3 .
- the silicon atoms of the Si-O-Si anchor group can additionally be cross-linked to one another via oxygen bridges.
- halogen atoms or NR 2 , OH or OR groups of mono-, di- or trifunctional alkyldimethylsilanes react, for example to form alkyldimethylsilyl groups (Si-O-Si- (CH 3 ) 2 -R, in particular Si-O- Si (CH 3 ) 3 ).
- the surface density of the anchor or terminating organic end group molecules does not have to correspond to the density of the silicon surface atoms, but can be lower.
- a higher reaction rate for the hydrophobization of the surface can be achieved with unsaturated compounds, such as trimethylene methane derivatives.
- Imaging of the hydrophobic printing form in hydrophilic partial areas or domains can be achieved by means of a laser, as already described in the first example.
- the hydrophilic initial state is retained by light-induced, in particular laser-induced, oxidation of the entire surface.
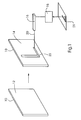
- a printing form 10 is plate-shaped and can be from a printing form cylinder, in particular in a Printing machine to be included.
- the printing form 10 has a surface 12 which has inorganically bonded silicon. This printing form 10 is in the initial state especially after their manufacturing process, usually with a native, few Covered nanometer thick oxide layer.
- the printing form 10 is provided with a defined essentially hydrophobic surface.
- the surface 12 of the Printing form 10 is for this purpose with organic end groups or fluorinated organic end groups terminated.
- the hydrophobic area 14 of the printing form 10 is now in another Process step hydrophilized in some areas. This can be done, for example, with one of the chemical reactions given above, in particular according to Examples 1 to 3, respectively. Two have been chosen for local modification of the hydrophobic surface 14 The method was found to be particularly suitable. As shown by way of example in FIG. 1, Local energy can be supplied by means of a laser 16, so that the chemical Conversion process is triggered. Lasers (in Continuous wave mode or pulsed), which have a small beam cross section, so that the chemical conversion can be carried out in a spatially limited area can. This can be smaller than the beam cross section. For example, creates a Fluor laser VUV light with a wavelength of about 157 nm.
- Light with a short Wavelength in this spectral range can alternatively be used with nonlinear optical Processes can be generated from light with a longer wavelength.
- this laser or one Another shortwave radiation source can be a photochemical Surface modification can be achieved.
- a photothermal modification As mentioned above, a variety of light wavelengths are possible for example, gas lasers (excimer lasers) or solid state lasers (for example frequency-multiplied Nd lasers) or diode lasers.
- the laser 16 is controlled by a control unit 18. It is a means of Generation of a relative movement between laser 16 and printing form 10 in this way provided that the light beam 20 emitted by the laser 16 all points of the surface of the Printing form 10, which represent the printing area, are painted over at least once or can be achieved.
- the printing form 10 on a Printing form cylinder applied or received in a printing press so that by rotating the cylinder around its axis of symmetry and translating the laser 16 essentially parallel to the axis of symmetry of the cylinder, the light beam 20 can paint over the entire surface of the printing form 10.
- the light beam 20 or the Laser 16 is switched on and off or as it passes over the printing form faded in and out so that a pattern 22 to be printed or the negative of the pattern can be introduced as a hydrophilic image in the hydrophobic surface.
- a pattern 22 to be printed or the negative of the pattern can be introduced as a hydrophilic image in the hydrophobic surface.
- the applied pattern 22 to be printed corresponds a template image 21, which can be generated in different ways.
- a template image 21 can be digitized or directly, for example using a graphics program or a digital camera become.
- the template images 21 are usually in a so-called RIP (raster imaging processor) processed and stored.
- the memory can be inside or lie outside the control unit 18. Based on the and determined in the RIP stored data, the light beam 20 is then controlled so that the one to be printed Pattern 22 is applied to the printing form 10.
- a hydrophilic image created in this way in a structured Deleting surface 14 can be done in a further process step in a first Embodiment on all other points of the hydrophobic surface 14 local energy with the help of the laser 16, so that finally the entire surface of the Printing form 14 hydrophilized and thus modified, in particular uniformly hydrophilized or unstructured.
- energy can be broad with a lamp, for example a UV lamp, in particular commercially available Excimer lamps with different UV wavelengths can be supplied.
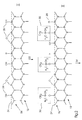
- the starting point is a surface 12 with inorganic silicon, which comprises silicon (di) oxide (partial image I).
- the notation silicon (di) oxide is intended to indicate that in the case of ultra-thin oxide layers, less than 1 nanometer, suboxides SiO x , with x ⁇ 2, are present and silicon dioxide (SiO 2 ) is only present in the case of thicker oxide layers.
- the surface has an oxidized surface layer 26 on the surface line, the thickness of which is typically in the nanometer range. Hydroxyl groups (OH groups) are bonded to valences 28 of the surface 12.
- the surface 12 is hydrophilic.
- Such a surface 12 can be obtained in different ways.
- the surface 12 can be obtained indirectly by forming a native oxide layer (spontaneous surface oxidation) of an amorphous silicon layer deposited on a carrier material.
- a silicon (di) oxide film can be deposited on a carrier material from a liquid phase or gas phase. Silicon oxide can also be used as glass.
- the carrier material or the glass can be shaped as a plate, cylinder or sleeve, in particular for use in a printing press. The steps described below can be carried out in particular in the printing press if the printing form is received in a printing unit.
- the surface 12 is cleaned before the hydrophobic termination. This is preferably done by large-area irradiation with the VUV light of a lamp under a wavelength of 200 nm, preferably 172 nm.
- the process is self-limiting; at room temperature, the surface is covered with a few monolayers of oxide skin.
- cleaning can be carried out by treatment with ozone (O 3 ) or another oxidizing agent, such as, for example, concentrated nitric acid (HNO 3 ), hydrogen peroxide solution (H 2 O 2 ) or the like.
- O 3 ozone
- HNO 3 concentrated nitric acid
- H 2 O 2 hydrogen peroxide solution
- Oxidative cleaning can typically be completed in about 10 minutes.
- the termination that follows in the immediately subsequent step 32 makes the Surface, preferably the entire surface, hydrophobic, i.e. water-repellent.
- the Termination is carried out using trimethylsilyl derivatives, for example hexamethyldisiloxane, Chlorotrimethylsilane, hexamethyldisilazane, ethoxytrimethylsilane or Dimethylaminotrimethylsilane.
- Such a termination reaction can proceed according to a general reaction scheme as follows: Si-OH + Y-Si (CH 3 ) 3 ⁇ Si-O-Si (CH 3 ) 3 + HY, where Y is a suitable leaving group.
- Y may be an OH group, a halogen atom, an NH 2 group or the like.
- the Si atom not carrying methyl groups is located in or on the surface 12.
- a preparation mixture of hexamethyldisiloxane is preferred, one completely second C atom halogenated ethanol and concentrated sulfuric acid (approx 90% strength).
- a mixture of 1.0 to 1.6 g of hexamethyldisiloxane, 4.5 is particularly preferred up to 8.0 g trifluoroethanol and 0.8 to 1.5 g 90% sulfuric acid, in particular 1.3 g Hexamethyldisiloxane, 6.0 g trifluoroethanol and 1.2 g 90% sulfuric acid.
- the trifluoroethanol concentration must be minimized.
- the concentration of Hexamethyldisiloxane and too little of the other components phase separation he follows. If there is too much sulfuric acid, undesirable side reactions can occur.
- the liquid mixture is oxidized for termination on the cleaned Brought silicon surface.
- the mixture can be made with a scraper Plastic can be painted on the surface.
- the surface slowly bathed in the preparation mixture, by one with the preparation mixture filled bathroom. The response time is around 10 seconds.
- the preparation solution automatically drains off from the terminated surface collects into small droplets, which are rinsed off with water or by means of the The principle of wick can be sucked off the surface.
- hexamethyldisilazane As an alternative to the preparation mixture described, it is also particularly advantageous to use hexamethyldisilazane.
- This substance can be used directly without any additional components. It can be supplied as vapor to the surface, but a suction device is advantageous because it releases ammonia gas (NH 3 ). Since hexamethyldisilazane has a high vapor pressure, subsequent rinsing of the surface is not necessary. The response time is also around 10 seconds.
- the result of the termination is a surface 12 which is inorganic bound Silicon and an oxidized surface layer 26 (silicon (di) oxide) on which Hydroxyl groups have been replaced by trimethysiloxy groups to an extent that the surface as a whole has hydrophobic properties.
- 2 are the organic end groups 30 shown roughly schematically in drawing II.
- the hydrophobic termination is carried out Imaging or energy input locally removed, so that at the illustrated areas again A hydrophilic silicon (di) oxide surface is created (structuring step 34, partial image III of Fig. 2).
- Imaging is preferably carried out by means of a laser, IR, NIR, visible or UV radiation can be used.
- Under the silicon (di) oxide layer can be a material, for example a metallic or ceramic layer, with a high absorption coefficients are used so that the radiation in a small Volume is absorbed with high efficiency.
- In the visible or UV Spectral range can (doped) amorphous silicon act as an absorption layer.
- the printing form in one Offset printing process can be printed with conventional printing ink.
- usual detergents or solvents can be removed. For a rough cleaning are about 5 minutes required.
- the structuring can be deleted: During the deletion process, both organic impurities, such as paint residues or solvent residues, as well as the Removed hydrophobic termination of the surface (erase step 36 in Fig. 2).
- the deletion process is used to restore an unstructured hydrophilic Surface.
- sub-picture IV of FIG. 2 corresponds to sub-picture I of FIG. 2, if the organic end groups 30 have been removed over a large area and the surface 12 on the valences 28 again carries OH groups.
- the Si-O portion of the organic can be on the oxidized surface layer 26 of the surface 12 remain so that the surface line 24 is replaced by a new surface line 38 becomes.
- silicon (di) oxide can slowly surface 12 grow without losing the composition in principle or the To change wetting properties.
- the deletion can preferably be done by Energy is applied by means of a laser, in other words a large area Imaging or cleaning using UV light, as already described above carried out.
- a hydrophobic surface 14 of the printing form 10 by local photo-induced Reaction processes in parts in a changed, second chemical state, convert in particular hydrophilic state.
- the surface of the printing form 10 can also over a large area in either the first chemical state or the second chemical state State are set so that a pattern 22 to be printed is removed again and a new structuring can be carried out.
- the printing form 10 can also be used as rewritable printing form or reusable printing form.
- the printing form according to the invention is in particular an offset printing form.
Landscapes
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Chemical & Material Sciences (AREA)
- Ceramic Engineering (AREA)
- Inorganic Chemistry (AREA)
- Printing Plates And Materials Therefor (AREA)
- Materials For Photolithography (AREA)
- Manufacture Or Reproduction Of Printing Formes (AREA)
- Photosensitive Polymer And Photoresist Processing (AREA)
- Emulsifying, Dispersing, Foam-Producing Or Wetting Agents (AREA)
- Coloring (AREA)
Abstract
Description
- Fig. 1
- die schematische Darstellung des erfindungsgemäßen Verfahrens, und
- Fig. 2
- eine schematisches Illustration einer besonders bevorzugten Ausführungsform des Verfahrens.
- 10
- Druckform
- 12
- Oberfläche
- 14
- hydrophober Bereich
- 16
- Laser
- 18
- Steuereinheit
- 20
- Lichtstrahl
- 21
- Vorlage
- 22
- zu druckendes Muster
- 24
- Oberflächenlinie
- 26
- oxidierte Oberflächenschicht
- 28
- Valenz
- 30
- organische Endgruppe
- 32
- Terminierungsschritt
- 34
- Strukturierungsschritt
- 36
- Löschschritt
- 38
- neue Oberflächenlinie
Claims (24)
- Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist und ein aus hydrophilen und hydrophoben Bereichen bestehendes Muster trägt, wobei die hydrophilen Bereiche einen ersten chemischen Zustand und die hydrophoben Bereiche einen zweiten, vom ersten Zustand verschiedenen chemischen Zustand aufweisen,
dadurch gekennzeichnet, dass in wenigstens einem der hydrophoben Bereiche die Oberfläche (12) Siliziumatome aufweist, an denen jeweils wenigstens eine organische Endgruppe angebunden ist, wobei die Siliziumatome nicht nur genau mit einer CH3-Gruppe oder mit einer OCH3-Gruppe substituiert sind. - Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist, gemäß Anspruch 1,
dadurch gekennzeichnet, dass die Oberfläche (12) amorphes, nanokristallines, polykristallines oder kristallines Silizium oder eine stöchiometrische oder nicht stöchiometrische Siliziumkeramik, welche Sauerstoff und/oder Stickstoff aufweist, ist. - Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist, gemäß Anspruch 1 oder 2,
dadurch gekennzeichnet, dass die Oberfläche (12) als dünner amorpher Film auf einem metallischen Träger aufgenommen ist. - Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist, gemäß einem der vorstehenden Ansprüche,
dadurch gekennzeichnet, dass in wenigstens einem der hydrophilen Bereiche die Oberfläche (12) Siliziumatome aufweist, an denen Oxid- und/oder Hydroxy- und/oder Silylaminund/oder Aldehyd- und/oder Carboxyl-Endgruppen angebunden sind. - Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist, gemäß einem der vorstehenden Ansprüche,
dadurch gekennzeichnet, dass in wenigstens einem der hydrophoben Bereiche die organischen Endgruppen unsubstituierte und/oder teilweise chlorierte und/oder vollständig chlorierte und/oder teilweise fluorierte und/oder vollständig fluorierte Endgruppen sind. - Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist, gemäß Anspruch 5,
dadurch gekennzeichnet, dass in wenigstens einem der hydrophoben Bereiche die organischen Endgruppen jeweils eine Kette von mehreren Kohlenstoffatomen aufweisen, an der sich CH3- oder CF3-Gruppen befinden. - Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist, gemäß einem der vorstehenden Ansprüche,
dadurch gekennzeichnet, dass in wenigstens einem der hydrophoben Bereiche die organischen Endgruppen weniger als 21 Kohlenstoffatome aufweisen. - Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist, gemäß einem der vorstehenden Ansprüche,
dadurch gekennzeichnet, dass in wenigstens einem der hydrophoben Bereiche die organischen Endgruppen mittels einer Si-C-Bindung und/oder einer Si-O-C-Bindung und/oder einer Si-O-Si-C-Bindung angebunden sind. - Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist, gemäß einem der vorstehenden Ansprüche,
dadurch gekennzeichnet, dass in wenigstens einem der hydrophoben Bereiche Siliziumatome an der Oberfläche (12) mehrere organische Endgruppen tragen. - Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist, gemäß einem der vorstehenden Ansprüche,
dadurch gekennzeichnet, dass in wenigstens einem der hydrophoben Bereiche Siliziumatome drei Methylgruppen oder eine O-Si-(CH3)3-Gruppe tragen. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) mit einer Oberfläche (12), welche anorganisch gebundenes Silizium aufweist, wobei die Oberfläche (12) in einen ersten chemischen Zustand mit erster Benetzungseigenschaft gebracht wird und eine Teilmenge der Oberfläche (12) in einen zweiten chemischen Zustand mit zweiter Benetzungseigenschaft durch Änderung der chemischen Endgruppen der Oberfläche (12) gebracht wird,
dadurch gekennzeichnet, dass an Siliziumatomen an der hydrophoben Oberfläche (12) organische Endgruppen derart angebunden werden, dass die Siliziumatome nicht nur genau mit einer CH3-Gruppe oder mit einer OCH3-Gruppe substituiert sind. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß Anspruch 11,
dadurch gekennzeichnet, dass entweder die erste Benetzungseigenschaft hydrophil und die zweite Benetzungseigenschaft hydrophob oder die erste Benetzungseigenschaft hydrophob und die zweite Benetzungseigenschaft hydrophil ist. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß Anspruch 11 oder 12,
dadurch gekennzeichnet, dass die Oberfläche (12) amorph ist, nanokristallines, polykristallines oder kristallines Silizium enthält oder eine stöchiometrische oder nicht stöchiometrische Siliziumkeramik, welche Sauerstoff und/oder Stickstoff aufweist, ist. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß einem der vorstehenden Ansprüche 11 bis 13,
dadurch gekennzeichnet, dass in wenigstens einem der hydrophoben Bereiche unsubstituierte und/oder teilweise chlorierte und/oder vollständig chlorierte und/oder teilweise fluorierte und/oder vollständig fluorierte Alkylendgruppen als organische Endgruppen angebunden werden. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß Anspruch 14,
dadurch gekennzeichnet, dass in wenigstens einem der hydrophoben Bereiche Ketten von mehreren Kohlenstoffatomen, an denen sich CH3- oder CF3-Gruppen befinden, als organische Endgruppen angebunden werden. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß einem der vorstehenden Ansprüche 11 bis 15,
dadurch gekennzeichnet, dass der zweite chemische Zustand durch lokalisierte Prozessierung mit einer gesteuerten Lichtquelle derart erfolgt, dass der zweite chemische Zustand so erzeugt wird, dass er einer zu druckenden Bildinformation oder deren Negativ entspricht. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß einem der vorstehenden Ansprüche 11 bis 16,
dadurch gekennzeichnet, dass der chemische Zustand mit hydrophiler Benetzungseigenschaft durch thermische Aufheizung oder photochemisch erreicht wird. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß einem der vorstehenden Ansprüche 11 bis 17,
dadurch gekennzeichnet, dass Arylgruppen und/oder Alkylgruppen und/oder Fluoralkylgruppen und/oder Chlorakylgruppen über eine Si-C-Bindung durch Photoinitiierung mit Halogensilanen, Alkoholen, Alkenen oder Alkinen angebunden werden. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß einem der vorstehenden Ansprüche 11 bis 17,
dadurch gekennzeichnet, dass organische Endgruppen durch Reaktion mit Iodoform und/oder Trimethylenmethan-Derivaten und/oder Methylencyclopropan-Derivaten und/oder 1,1-Dialkoxy-2-Methylencyclopropan (DMCP) und/oder Trimethylsilyl-Derivaten angebunden werden. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß Anspruch 19,
dadurch gekennzeichnet, dass das Trimethysilyl-Derivat Hexamethyldisiloxan oder Hexamethyldisilazan ist. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß einem der vorstehenden Ansprüche 11 bis 17,
dadurch gekennzeichnet, dass Arylgruppen und/oder Alkylgruppen über eine Si-O-C-Bindung durch Reaktion von primären Alkoholen und/oder sekundären Alkoholen und/oder Aldehyden angebunden werden. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß Anspruch 21,
dadurch gekennzeichnet, dass mittels Lichteinwirkung die Reaktion initiiert und/oder beschleunigt wird. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß einem der vorstehenden Ansprüche 11 bis 17,
dadurch gekennzeichnet, dass Alkylgruppen über eine Si-O-Si-C-Bindung durch Reaktion mit Alkylalkoxysilanen, Alkylalkaminosilanen und/oder Alkylchlorsilanen angebunden werden. - Verfahren zum Ändern der Benetzungseigenschaften einer Druckform (10) gemäß Anspruch 23,
dadurch gekennzeichnet, dass Alkylgruppen über eine Si-O-Si-C-Bindung durch Reaktion mit Alkyltrimethoxysilanen und/oder Fluoralkylmethoxysilanen angebunden werden.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE10260114 | 2002-12-19 | ||
| DE10260114 | 2002-12-19 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1431031A2 true EP1431031A2 (de) | 2004-06-23 |
| EP1431031A3 EP1431031A3 (de) | 2004-09-22 |
| EP1431031B1 EP1431031B1 (de) | 2010-11-03 |
Family
ID=32336520
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP03027825A Expired - Lifetime EP1431031B1 (de) | 2002-12-19 | 2003-12-04 | Verfahren zum Ändern der Benetzungseigenschaften einer Druckform |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP1431031B1 (de) |
| JP (1) | JP2004195979A (de) |
| CN (1) | CN1508010B (de) |
| AT (1) | ATE486718T1 (de) |
| DE (2) | DE10356600A1 (de) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1995060A1 (de) * | 2007-05-22 | 2008-11-26 | Ernst-Rudolf Dr. Weidlich | Verfahren zum Beeinflussen der Farbhalte- und/oder Farbabgabefähigkeit von Druckformen und Druckvorrichtung |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006020083A (ja) | 2004-07-01 | 2006-01-19 | Omron Corp | タグ通信用アンテナ、タグ通信装置、タグ通信システム、タグ通信装置のスキャン調整方法、およびスキャン調整プログラム |
| DE102012013302A1 (de) * | 2011-08-11 | 2013-02-14 | Heidelberger Druckmaschinen Ag | Druckform |
| JP6130147B2 (ja) * | 2013-01-11 | 2017-05-17 | 太陽誘電ケミカルテクノロジー株式会社 | 構造体及び構造体の製造方法 |
| TWI797640B (zh) * | 2020-06-18 | 2023-04-01 | 法商液態空氣喬治斯克勞帝方法研究開發股份有限公司 | 基於矽之自組裝單層組成物及使用該組成物之表面製備 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4718340A (en) * | 1982-08-09 | 1988-01-12 | Milliken Research Corporation | Printing method |
| WO1998034795A1 (en) * | 1997-02-06 | 1998-08-13 | Star Micronics Co., Ltd. | Image formation apparatus, image formation method and plate making method |
| EP1376225B1 (de) * | 1997-08-08 | 2010-10-13 | Dai Nippon Printing Co., Ltd. | Struktur zur Musterbildung, Verfahren zur Musterbildung und deren Anwendung |
| JP3384544B2 (ja) * | 1997-08-08 | 2003-03-10 | 大日本印刷株式会社 | パターン形成体およびパターン形成方法 |
| EP1082224B1 (de) * | 1998-10-10 | 2004-02-11 | Heidelberger Druckmaschinen Aktiengesellschaft | Druckform und verfahren zum ändern ihrer benetzungseigenschaften |
| JP2000181071A (ja) * | 1998-12-15 | 2000-06-30 | Fuji Photo Film Co Ltd | 平版印刷版用原版 |
| JP3534697B2 (ja) * | 2000-11-29 | 2004-06-07 | 三菱重工業株式会社 | 印刷用版材の作製方法、再生方法及び印刷機 |
-
2003
- 2003-12-04 EP EP03027825A patent/EP1431031B1/de not_active Expired - Lifetime
- 2003-12-04 DE DE10356600A patent/DE10356600A1/de not_active Withdrawn
- 2003-12-04 AT AT03027825T patent/ATE486718T1/de not_active IP Right Cessation
- 2003-12-04 DE DE50313232T patent/DE50313232D1/de not_active Expired - Lifetime
- 2003-12-15 CN CN200310120627.2A patent/CN1508010B/zh not_active Expired - Fee Related
- 2003-12-16 JP JP2003417824A patent/JP2004195979A/ja active Pending
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1995060A1 (de) * | 2007-05-22 | 2008-11-26 | Ernst-Rudolf Dr. Weidlich | Verfahren zum Beeinflussen der Farbhalte- und/oder Farbabgabefähigkeit von Druckformen und Druckvorrichtung |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2004195979A (ja) | 2004-07-15 |
| EP1431031B1 (de) | 2010-11-03 |
| ATE486718T1 (de) | 2010-11-15 |
| DE10356600A1 (de) | 2004-07-01 |
| DE50313232D1 (de) | 2010-12-16 |
| CN1508010A (zh) | 2004-06-30 |
| EP1431031A3 (de) | 2004-09-22 |
| CN1508010B (zh) | 2010-04-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4679806B2 (ja) | 再使用可能な印刷版 | |
| DE69726634T2 (de) | Entfernung von Kohlenstoff auf Substratenoberflächen | |
| EP1616713B1 (de) | Wiederverwendbare Druckform | |
| DE102020120884B4 (de) | Verfahren und Vorrichtung zum Ätzen einer Lithographiemaske | |
| DE10361257B4 (de) | Verfahren zur Herstellung von feinen Mustern | |
| EP1499490A1 (de) | Verfahren zum erzeugen dreidimensionaler körper oder oberflächen durch laser-bestrahlung | |
| EP1082224B1 (de) | Druckform und verfahren zum ändern ihrer benetzungseigenschaften | |
| EP1431031B1 (de) | Verfahren zum Ändern der Benetzungseigenschaften einer Druckform | |
| DE3038185A1 (de) | Verfahren zur herstellung von aetzmustern | |
| DE102006053074B4 (de) | Strukturierungsverfahren unter Verwendung chemisch verstärkter Fotolacke und Belichtungsvorrichtung | |
| DE4410505C2 (de) | Verfahren zum Herstellen eines Reliefbildes im Submikrometerbereich | |
| US7152530B2 (en) | Printing form and method for modifying its wetting properties | |
| EP0207528A2 (de) | Verfahren zum Herstellung einer Lichtmaske | |
| EP1254768B1 (de) | Bebilderung und Löschung einer Druckform aus Polymermaterial mit Imid-Gruppen | |
| EP1456870A2 (de) | Resistloses lithographieverfahren zur herstellung feiner strukturen | |
| EP0394740B1 (de) | Trockenentwickelbares Resistsystem | |
| EP0163128A2 (de) | Verfahren zur Erzeugung von Strukturen in Resistschichten | |
| WO1998039688A1 (de) | Verfahren zur herstellung einer strukturierten schicht | |
| DE60315824T2 (de) | Verfahren zur regenerierung einer lithographischen druckplatte | |
| DE10057656C1 (de) | Verfahren zur Herstellung von integrierten Abtastnadeln | |
| DE19733520A1 (de) | Verfahren zur Nanostrukturierung von amorphen Kohlenstoffschichten | |
| DE102024101400A1 (de) | Verfahren zum Herstellen einer Druckform | |
| DE102006035955A1 (de) | Wiederbeschreibbare Durckform für den Offsetdruck | |
| DE10226971A1 (de) | Substrat mit strukturierter Oberfläche aus hydrophoben und hydrophilen Bereichen | |
| HK1062662B (en) | A reusable printing form, a printing unit, aprinting machine, amethod for creating an imagable printing area, amethod for imaging a reusable printing form |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK |
|
| 17P | Request for examination filed |
Effective date: 20050322 |
|
| AKX | Designation fees paid |
Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| 17Q | First examination report despatched |
Effective date: 20060324 |
|
| RTI1 | Title (correction) |
Free format text: METHOD FOR MODIFYING THE WETTING CHARACTERISTICS OF A PRINTING FORM |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT RO SE SI SK TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: GERMAN |
|
| REF | Corresponds to: |
Ref document number: 50313232 Country of ref document: DE Date of ref document: 20101216 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20101103 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FD4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110303 Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110203 |
|
| BERE | Be: lapsed |
Owner name: HEIDELBERGER DRUCKMASCHINEN A.G. Effective date: 20101231 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110204 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20101231 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110214 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20101231 |
|
| 26N | No opposition filed |
Effective date: 20110804 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20101231 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20101231 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 50313232 Country of ref document: DE Effective date: 20110804 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20101204 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MM01 Ref document number: 486718 Country of ref document: AT Kind code of ref document: T Effective date: 20101204 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20110504 Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20101204 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20101103 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 13 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20161222 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20161222 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20161231 Year of fee payment: 14 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 50313232 Country of ref document: DE |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20171204 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20180831 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180102 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180703 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20171204 |