EP1124502B1 - Dispositif de traitement de l'incontinence - Google Patents
Dispositif de traitement de l'incontinence Download PDFInfo
- Publication number
- EP1124502B1 EP1124502B1 EP99947825A EP99947825A EP1124502B1 EP 1124502 B1 EP1124502 B1 EP 1124502B1 EP 99947825 A EP99947825 A EP 99947825A EP 99947825 A EP99947825 A EP 99947825A EP 1124502 B1 EP1124502 B1 EP 1124502B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- signals
- processor
- patient
- control unit
- electrode
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/0004—Closure means for urethra or rectum, i.e. anti-incontinence devices or support slings against pelvic prolapse
- A61F2/0031—Closure means for urethra or rectum, i.e. anti-incontinence devices or support slings against pelvic prolapse for constricting the lumen; Support slings for the urethra
- A61F2/0036—Closure means for urethra or rectum, i.e. anti-incontinence devices or support slings against pelvic prolapse for constricting the lumen; Support slings for the urethra implantable
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2250/00—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2250/0001—Means for transferring electromagnetic energy to implants
Definitions
- the present invention relates generally to medical electronic devices, and specifically to implantable electrical muscle stimulators.
- Urinary stress incontinence affects millions of people, causing discomfort and embarrassment, sometimes to the point of social isolation. Stress incontinence results from weakness or inability of pelvic muscles to hold back urinary flow from the bladder when abdominal pressure increases due to everyday events such as coughing, laughing or mild physical exertion. Muscles involved in controlling the urinary flow include primarily the urethral sphincter and the levator ani, with the cooperation of fibromuscular extensions along the urethra and other muscles in the general region of the pelvic diaphragm. In the United States, it is estimated that 10-13 million patients seek medical care for incontinence each year, of whom about 35% suffer from stress-type incontinence.
- Stress incontinence is typically associated with either or both of the following anatomical conditions:
- bladder neck suspension primarily to control urethral hypermobility by elevating the bladder neck and urethra. These procedures typically use bone anchors and sutures or slings to support the bladder neck.
- the success rates for bladder neck suspension surgery in controlling urinary leakage are typically in the 60-80% range, depending on the patient's condition, the surgeon and the procedure that is used.
- the disadvantages of surgery are its high cost, need for hospitalization and long recovery period, and high frequency of complications.
- the AMS 800 urinary sphincter produced by American Medical Systems Inc., of Minnetonka, Minnesota, includes a periurethral inflatable cuff, used to overcome urinary incontinence when the function of the natural sphincter is impaired.
- the cuff is coupled to a manually-operated pump and a pressure regulator chamber, which are implanted in a patient's body together with the cuff.
- the cuff is maintained at a constant pressure of 60-80 cm of water, which is generally higher than the bladder pressure.
- This artificial sphincter has several shortcomings, however.
- the constant concentric pressure that the periurethral cuff exerts on the urethra results in impaired blood supply to tissue in the area, leading to tissue atrophy, urethral erosion and infection.
- the constant pressure in the cuff is not always sufficient to overcome transient increases in bladder pressure that may result from straining, coughing, laughing or contraction of the detrusor muscle, for example. In such cases, urine leakage may result.
- Interstim for treatment of urge incontinence, which is a different disorder from stress incontinence.
- urge incontinence a sudden, urgent need to pass urine causes involuntary urination, before the patient can get to a toilet.
- the condition may be caused by damage to nerve pathways from the brain to the bladder or by psychosomatic factors, leading to involuntary bladder contraction.
- Interstim uses an implantable pulse generator, which is surgically implanted in the lower abdomen and wired to nerves near the sacrum (the bone at the base of the spine) in a major surgical procedure under general anesthesia. Electrical impulses are then transmitted continuously to a sacral nerve that controls urinary voiding. The continuous electrical stimulation of the nerve has been found to reduce or eliminate urge incontinence in some patients.
- Exercise and behavioral training are also effective in some cases in rehabilitating pelvic muscles and thus reducing or resolving incontinence.
- Patients are taught to perform Kegel exercises to strengthen their pelvic muscles, which may be combined with electrical stimulation of the pelvic floor.
- Electromyographic biofeedback may also be provided to give the patients an indication as to the effectiveness of their muscular exertions. Retraining muscles is not possible or fully effective for most patients, however, particularly when there may be neurological damage or other pathologies involved.
- EMG electromyogram
- U.S. Patent 5,562,717 describes electrodes that are placed on the body surface, typically in the areas of the perineum and the sacrum, and are electrically actuated to control incontinence.
- U.S. Patent 4,785,828 describes a vaginal plug having electrodes on an outer surface thereof. A pulse generator in the plug applies electrical pulses to the electrodes so as to constrict the pelvic muscles and prevent urine flow.
- U.S. Patent 4,153,059 describes an intra-anal electrode, to which repetitive electrical pulses are applied in order to control urinary incontinence.
- U.S. Patent 5,562,717 describes electrodes that are placed on the body surface, typically in the areas of the perineum and the sacrum, and are electrically actuated to control incontinence.
- U.S. Patent 4,785,828 describes a vaginal plug having electrodes on an outer surface thereof. A pulse generator in the plug applies electrical pulses to the electrodes so as to constrict the pelvic muscles and prevent urine flow.
- Patent 4,106,511 similarly describes an electrical stimulator in the form of a plug for insertion into the vagina or the anus.
- U.S. Patent 3,866,613 describes a pessary ring having two electrodes thereon, which are energized to control incontinence. The disclosures of all of the above-mentioned patents are incorporated herein by reference.
- U.S. Patent 4,580,578 whose disclosure is also incorporated herein by reference, describes a device for stimulating the sphincter muscles controlling the bladder.
- a supporting body is fitted into the patient's vulva between the labia, so that two electrodes attached to the supporting body contact the epidermal surface on either side of the external urethral orifice. Electrical impulses are applied to the electrodes to stimulate the region of the sphincter.
- US-A-3628538 discloses apparatus for stimulating a muscle using an EMG signal sensed in the muscle. If the EMG signal is greater than a threshold value a mono-stable circuit to which the signal is applied enters its quasi-stable state and a stimulator circuit applies a voltage to electrodes adjacent the muscle. The apparatus reverts to a sensing mode when the mono-stable circuit is in its stable state.
- a portable electrical stimulation device comprising one or more electrodes for applying electrical stimulation signals to a patient's body, a signal generator to generate the stimulation signals and a storage device for imparting a set of instructions to the signal generator.
- the invention provides a device for treatment of urinary stress incontinence, comprising at least one electrode for implanting in a pelvic muscle of a patient; and a control unit, for receiving signals indicative of abdominal stress in the patient and responsive thereto the control unit is adapted to apply an electrical waveform to the electrode which stimulates the muscle to contract, so as to inhibit involuntary urine flow through the patient's urethra due to the stress, characterised in that the control unit is adapted to receive signals indicative of a fill level of the patient's bladder and further adapted in response to said signals to prevent application of the electrical waveform when the fill level of the bladder is low, even when signals are received indicative of abdominal stress.
- an implantable device for treatment of urinary stress incontinence comprises a control unit and one or more electrodes coupled to the control unit.
- the electrode or electrodes are preferably implanted in the genital region of a patient so as to contact one or more of the muscles that are used in regulating urine flow from the bladder.
- the control unit is preferably implanted under the skin of the abdomen or genital region. Motion of or pressure on or in the area of the bladder generates an electromyographic (EMG) signal in the muscles, which is sensed by the one or more electrodes and analyzed by the control unit. Alternatively or additionally, non-electromyographic signals are received and analyzed by the control unit, as described hereinbelow.
- EMG electromyographic
- control unit determines that the signals are indicative of a condition, such as an increase in abdominal or intravesical pressure, that is likely to cause involuntary urine flow from the bladder, it applies an electrical waveform to the electrode or electrodes, stimulating the contacted muscle to contract and thus to inhibit the urine flow.
- a condition such as an increase in abdominal or intravesical pressure
- the device also includes one or more other physiological sensors, which generate signals responsive to motion or to intravesical or abdominal pressure, or to urine volume in the bladder. These signals are thus indicative of possible incontinence that may occur due to coughing, laughing, or other strain or motion of the abdominal muscles. On the other hand, when the urine volume in the bladder is low, there will be no urine flow even when the abdominal pressure does increase.
- the control unit processes the signals from the other sensors and uses them to determine when the electrical stimulation should be applied to the muscles.
- control unit comprises a processor, which is programmed to distinguish between signals indicative of possible incontinence and other signals that do not warrant stimulation of the muscles.
- the processor is preferably programmed to recognize signal patterns indicative of normal voiding, and does not stimulate the muscles when such patterns occur, so that the patient can pass urine normally.
- the processor analyzes both long-term and short-term variations in the signals, as well as rates and patterns of change in the signals.
- the processor in response to the analysis, the processor: (a) makes an assessment of the patient's physiological condition, such as of the patient's bladder fill level, (b) responsive to the assessment, adjusts a time-varying threshold level associated with an aspect of the EMG signal (e.g., magnitude and/or rate) that varies over time, and (c) applies the stimulation only when a transient variation in the aspect of the EMG signal exceeds the threshold.
- a time-varying threshold level associated with an aspect of the EMG signal (e.g., magnitude and/or rate) that varies over time
- the control unit comprises a low-power, low-speed processor, which monitors the EMG signals continuously, and a high-speed processor, which turns on only when the low-speed processor detects an increase in EMG activity.
- the high-speed processor performs an accurate analysis of the signals to determine whether stimulation is actually warranted. The inventors have found that the signals must generally be analyzed at a sample rate greater than 1000 Hz in order to accurately forecast whether or not involuntary urine loss is about to occur.
- the electrodes are implanted (unlike electrical muscle stimulators known in the art) and generally apply electrical stimulation directly into the muscle only when contraction is actually required, preferably as indicated by intrinsic physiological signals. At other times, stimulation is not applied, and the muscles are allowed to relax. Implantation of the device provides reliable, typically long-term control of muscle function, and relieves incontinence in a manner that is unobtrusive and minimizes inconvenience and discomfort of the patient. The stimulation mimics the natural function of the muscles in maintaining urinary continence. Repeated stimulation using these embodiments of the present invention also tends to exercise and strengthen the muscles, thus enhancing their inherent capability to control urine flow. Direct stimulation of the muscles, in accordance with the principles of these embodiments of the present invention, is believed to be effective against urine loss due to substantially all common types of stress incontinence.
- a variation not within the scope of the claims, provides a device for treatment of urinary stress incontinence, including:
- the signals include electromyographic signals received from the at least one electrode
- the device includes a switch between the electrode and an input of the control unit, which switch is opened when the electrical waveform is applied so as to prevent feedback from the electrode to the input.
- control unit includes a processor, which analyzes the signals so as to determine when an involuntary urine flow is likely, whereupon the waveform is applied. Further preferably, the processor distinguishes between signals indicative of an involuntary urine flow and signals indicative of voluntary voiding by the patient.
- the processor is programmable to vary one or more parameters associated with the application of the waveform, and the device includes a wireless receiver, which receives data for programming the processor from a programming unit outside the patient's body.
- the processor's analysis is performed on substantially non-rectified data.
- the processor analyzes the signals using spectral analysis.
- the spectral analysis is performed by the processor on substantially non-rectified data.
- the at least one electrode includes a single unipolar electrode or, alternatively or additionally, a pair of bipolar electrodes. Further preferably, the at least one electrode includes a flexible intra-muscular electrode.
- the device includes a physiological sensor coupled to the patient's bladder, which sensor provides at least some of the signals to the control unit.
- the sensor includes a pressure sensor or, alternatively or additionally, an acceleration sensor.
- the at least one electrode and the control unit are implanted in the body of the patient, and the control unit includes a rechargeable power source.
- the power source is recharged by inductive energy transfer, substantially without electrical contact between the control unit and any object outside the patient's body.
- the pelvic muscle includes the levator ani muscle or, alternatively or additionally, the urethral sphincter muscle or another muscle adjacent to the urethral sphincter muscle.
- a device for treatment of urinary incontinence in a patient including:
- control unit receives a further signal indicative of abdominal stress and applies the stimulation to the pelvic muscle responsive to the stress except when the fill level of the bladder is below the threshold level.
- the sensor includes an electrode, which is placed in electrical contact with the pelvic muscle of the patient to receive an electromyogram signal therefrom indicative of the stress and of the fill level.
- the device includes an electrode, which is placed in electrical contact with the pelvic muscle of the patient, and the control unit applies an electrical waveform to the electrode so as to stimulates the muscle to contract, thereby inhibiting the involuntary flow of urine.
- the senor includes a pressure sensor or, alternatively or additionally, an ultrasound transducer.
- a device for treatment of urinary stress incontinence including:
- the threshold signal level varies over time responsive to temporal variation of an average value of the electromyogram signals. Additionally or alternatively, the threshold signal level increases responsive to time elapsed since the patient last passed urine or responsive to an increase in a fill level of the patient's bladder.
- a device for treatment of urinary stress incontinence including:
- control unit when the rate of change is below a threshold rate, the control unit withholds the waveform so as to allow voluntary voiding.
- a device for treatment of urinary stress incontinence including:
- control unit distinguishes between the signals indicative of an involuntary urine flow and the signals indicative of voluntary voiding, substantially without application of an input to the control unit from outside the patient's body.
- a device for treatment of urinary stress incontinence including:
- a device for treatment of urinary stress incontinence including:
- an electronic muscle stimulator device is implanted in the genital region of a patient and stimulates one or more of the muscles in the region, so as to control and treat urinary stress incontinence.
- motion of or pressure on or in the area of the patient's urinary bladder generates an electromyographic (EMG) signal in the muscles, which is sensed by one or more electrodes and is analyzed by a control unit of the device.
- EMG electromyographic
- non-electromyographic signals are received and analyzed by the control unit.
- control unit determines that the signals are indicative of a condition, such as an increase in abdominal or intravesical pressure, that is likely to cause involuntary urine flow from the bladder, it applies an electrical waveform to the one or more electrodes, stimulating the contacted muscle to contract and thus to inhibit the urine flow.
- a condition such as an increase in abdominal or intravesical pressure
- the device preferably also comprises one or more other physiological sensors, described hereinbelow with reference to Figs. 2-4, which generate signals responsive to motion, to intravesical or abdominal pressure, or to urine volume in the bladder. These signals are indicative of possible incontinence that may occur due to coughing, laughing, or other strain or motion of the abdominal muscles. Typically, when the urine volume in the bladder is low, there will be no urine flow even when the abdominal pressure does increase.
- the control unit preferably processes the signals from the other sensors and uses them to determine when the electrical stimulation should be applied to the muscles.
- the control unit comprises a processor, e.g., as described with reference to Figs. 3 and 4, which is programmed to distinguish between signals indicative of possible incontinence and other signals that do not warrant stimulation of the muscles.
- the processor is preferably programmed to recognize signal patterns indicative of normal voiding, and does not stimulate the muscles when such patterns occur, so that the patient can pass urine normally. Detection of normal voiding is described in more detail with reference to Fig. 7.
- the processor analyzes both long-term and short-term variations in the signals, as well as rates, spectral patterns, and patterns of change in the signals.
- the processor sets a threshold of an aspect of the EMG signal that varies over time responsive to an assessment of the patient's physiological condition, and applies the stimulation only when a transient variation in the aspect of the EMG signal exceeds the threshold.
- a threshold is to be understood as comprising substantially any appropriate time-varying detection parameters that a person skilled in the art, having read the disclosure of the present patent application, would consider useful in applying the principles of the present invention.
- these time-varying detection parameters may include magnitude, rate, or other aspects of the EMG signal, and quantitative ultrasound, pressure, or acceleration measurements, as described herein.
- control unit preferably comprises a low-power, low-speed processor, which monitors the EMG signals continuously, and a high-speed processor, which turns on only when the low-speed processor detects an increase in EMG activity.
- a low-power, low-speed processor which monitors the EMG signals continuously
- a high-speed processor which turns on only when the low-speed processor detects an increase in EMG activity.
- Use of the two processors has been shown to significantly reduce consumption of electrical power.
- the high-speed processor performs an accurate analysis of the signals to determine whether stimulation is actually warranted.
- FIG. 1 is a schematic, pictorial illustration of an implantable electronic muscle stimulator device 20, in accordance with a preferred embodiment of the present invention.
- Device 20 is preferably implanted in the genital region of a patient, as described further hereinbelow, for use in stimulating one or more of the muscles in the region, so as to control and treat urinary stress incontinence.
- Device 20 comprises a control unit 22 and electrodes 27 and 29, mutually coupled by an electrical lead 24.
- the electrodes are preferably flexible intramuscular-type wire electrodes, about 1-5 mm long and 50-100 microns in diameter, thus designed to minimize patient discomfort. They are typically formed in the shape of a spiral or hook, as is known in the art, so that they can be easily and permanently anchored in the muscle.
- the wire from which the electrodes are made comprises a suitable conductive material, preferably a biocompatible metal such as silver, a platinum/iridium alloy (90/10) or a nickel/chromium alloy.
- Lead 24 is preferably 5-10 cm long and has an insulating jacket 26 preferably comprising nylon, polyurethane, Teflon or other flexible, biocompatible insulating material.
- An optional additional wire 28 inside jacket 26 serves as an antenna for the purpose of wireless communications with device 20, as described further hereinbelow.
- Control unit 22 contains circuitry, described further hereinbelow with references to Figs. 3, 4 and 5, for receiving electrical signals from and applying a waveform to electrodes 27 and 29 via lead 24.
- the circuitry is preferably contained in a case 25, made of stainless steel or other suitable biocompatible metal, and is preferably about 20 mm in diameter and 4 mm thick.
- the stainless steel case serves as a ground electrode for electrodes 27 and 29 when they are sensing or stimulating in a bipolar mode.
- the case may be made of a plastic material which is coated with a layer of biocompatible plastic, such as polymethyl methacrylate (PMMA) or silicone.
- PMMA polymethyl methacrylate
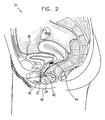
- Fig. 2 is a schematic, partly sectional illustration showing the genitourinary anatomy of a female patient 31 in whom device 20 is implanted, in accordance with a preferred embodiment of the present invention. It will be appreciated that whereas preferred embodiments of the present invention are described with respect to female patients, the principles of the present invention can also be applied to male patients, mutatis mutandis. Electrodes 27 and 29 (not shown) are preferably inserted into a muscle 32, in a vicinity of urethra 34 and bladder 36.
- the electrodes are inserted into the patient's levator ani muscle, which supports and reinforces the operation of the urethral sphincter and can generally compensate for lost function of the sphincter in controlling urine flow from the bladder, such as may occur in cases of stress incontinence.
- the electrodes are preferably inserted through an incision made in the wall of vagina 42, and control unit 22 may likewise be implanted through this incision.
- another suitable approach may be chosen for ease of access and minimization of tissue trauma.
- the levator ani and sphincter cooperate with fibromuscular extensions along urethra 34 and with other muscles in the general vicinity of the pelvic diaphragm.
- one or both of the electrodes may, alternatively or additionally, be inserted into one of these other muscles, such as the puborectalis, pubococcygeus, bulbospongiosus or the urethral sphincter itself
- the precise placement of the electrodes is not essential, particularly since electrical signals tend to pass among the different muscles in the region.
- any placement of the electrode in or on one or more of the pelvic muscles suitable for exercising urine control is considered to be within the scope of this embodiment of the present invention.
- Control unit 22 is preferably implanted under the skin in the genitopelvic region of patient 31. Most preferably, the control unit is implanted inside the patient's labia minora 38 or in the labia majora 40, near muscle 32. Alternatively, the control unit is not implanted in the patient's body, but is instead maintained outside the body, connected by lead 24 to the electrodes. This configuration is convenient particularly for an initial test period, during which the effectiveness of device 20 in treating a given patient is evaluated before permanent implantation.
- a miniaturized ultrasound transducer 44 is implanted in proximity to bladder 36 and is coupled to control unit 22. Signals from the transducer are analyzed to estimate the urine volume within the bladder. When the bladder is empty, there is no need to actuate electrodes 27 and 29, even when a transient increase in the electromyogram (EMG) signal, as described hereinbelow, indicates an increase in abdominal pressure.
- EMG electromyogram
- the EMG signal itself is analyzed to gain an indication of the urine volume in the bladder, since when the bladder is full, the average EMG activity typically increases.
- Fig. 3 is a schematic block diagram showing circuitry used in control unit 22 to receive signals from and apply electrical waveforms to electrode 27, in accordance with a preferred embodiment of the present invention.
- device 20 is described as operating in a unipolar mode, the principles described hereinbelow are applicable to bipolar operation, as well, in which both electrodes 27 and 29 are active.
- electrode 27 When stress is applied to the abdomen of patient 31, electrode 27 receives EMG signals from muscle 32. These signals are conveyed via a switch 46, which is normally closed, to the input of an amplifier 48, preferably a low-noise operational amplifier. Amplified signals output from amplifier 48 are digitized by an analog/digital (A/D) converter 50 and conveyed to a central processing unit (CPU) 52, preferably a microprocessor. Preferably, although not necessarily, the amplified signals are not rectified prior to being digitized, to allow various forms of analysis, for example, spectral analysis, to be performed on the raw data, without the distortion imparted by rectification.
- A/D analog/digital
- CPU central processing unit
- CPU 52 preferably analyzes these signals and/or signals from other physiological sensors, such as ultrasound, pressure, and acceleration sensors described hereinbelow, to determine whether they fit a pattern indicating that incontinence, i.e., involuntary urine flow from bladder 36, is likely to result from the stress.
- the pattern may correspond to coughing, laughing, or other strain or motion of the abdominal muscles.
- the analysis preferably comprises a spectral analysis and an analysis of EMG signal magnitude and rate.
- the CPU is programmed to distinguish between incontinence-related patterns and other signal patterns not associated with incontinence, such as signals generated when patient 31 wishes to pass urine voluntarily.
- the CPU gathers long-term statistical information regarding the EMG and analyzes the information to "learn" common signal patterns that are characteristic of patient 31.
- the learned patterns are used in refining decision criteria used by the CPU in determining whether or not to apply waveforms to the electrodes.
- Fig. 6 is a graph that schematically illustrates results of a simulation experiment, in accordance with a preferred embodiment of the present invention, including a simulated EMG signal 100 of a woman suffering from stress incontinence.
- a variable, adaptive threshold level 102 is marked on the graph. Over the course of several hours, as the woman's bladder fill level increases, the average level of EMG signal 100 increases accordingly.
- threshold level 102 is computed so as to increase as a function of the average EMG.
- threshold level 102 and a plurality of other time-varying detection parameters are calculated as functions of other features of the EMG signal or of other aspects of the woman's condition (particularly as measured by sensors 44, 76 and 78 (Fig.
- Adaptive threshold level 102 enables five possible incidents of incontinence, marked by excursions 104 of signal 100 over level 102, to be detected reliably, with a low false alarm rate. On the other hand, if a fixed threshold level 106 is used, as is known in the art, a number of EMG excursions 104 are missed, and the false alarm rate is high.
- Fig. 7 includes graphs 110 and 112 that schematically illustrate experimental measurements made before, during and after voluntary voiding of urine, in accordance with a preferred embodiment of the present invention.
- Graph 112 is a continuation in time of graph 110.
- the upper trace in both graphs illustrates urine flow, wherein the beginning and end of voluntary flow are marked by arrows.
- the lower trace illustrates measured EMG signals.
- an EMG signal 114 shows substantial high-frequency activity, which is generally indicative of a full bladder. As illustrated by the graphs in the preceding figures, high-frequency spikes in signal 114 (of which none appear in Fig. 9) would be interpreted by CPU 52 as signs of imminent incontinence, leading to actuation of pulse generator 54.
- voluntary voiding is preceded by an EMG signal 116, in which there is a large but gradual increase in the signal level.
- Signal 116 is associated with voluntary activation of the pelvic floor muscles for the purpose of passing urine from the bladder, as is a later signal 118 during voiding.
- CPU 52 analyzes not only the level of the EMG signals, but also a rate of change of the signals, in order to distinguish between voluntary and involuntary contractions of the pelvic muscles.
- rate of change is characteristic of voluntary voiding, no stimulation is applied by pulse generator 54.
- CPU 52 opens switch 46 and commands a pulse generator 54 to apply a suitable electrical waveform to electrode 27 so as to stimulate muscle 32 to contract.
- Switch 46 is opened in order to avoid feedback of the stimulation waveform to amplifier 48, and is closed again after the waveform is terminated.
- the waveform is applied to the electrode in a unipolar mode, wherein case 25 of control unit 22 serves as the return (ground) electrode. (This mode can be used only when case 25 comprises a conductive material.
- control unit 22 has a plastic case, at least two electrodes are generally needed, in order to administer bipolar stimulation.
- the waveform is terminated and switch 46 is closed after a predetermined period of time, typically about 5 sec, has passed. If possible incontinence is again detected at this point, the waveform is re-applied.
- the waveform may be uniphasic or biphasic and may have a range of amplitudes, duty cycles and/or frequencies. It has been found generally that pulse frequencies in the range between 5 and 200 Hz are effective in engendering contraction of the levator ani and other pelvic muscles, but it may also be possible to use frequencies outside this range.
- the waveform comprises a bipolar square wave having the following characteristics:
- the waveform may comprise a decaying square wave, sinusoid or sawtooth or have any other shape found to be suitable. Further alternatively or additionally, the waveform may comprise one or more bursts of short pulses, each pulse preferably less than I ms in duration. Generally, appropriate waveforms and parameters thereof are determined during the initial test period.
- a battery 56 which may comprise a primary battery (non-rechargeable) and/or a rechargeable battery.
- a super-capacitor may be used to store and provide the electrical power.
- a rechargeable battery or super-capacitor it is preferably recharged via an inductive coil 58 or antenna, which receives energy by magnetic induction from an external magnetic field charging source (not shown) held in proximity to the pelvis of patient 31.
- the magnetic field causes a current to flow in coil 58, which is rectified by a rectifier 60 and furnished to charge battery 56.
- Wire 28 may also be used for this purpose.
- battery 56 comprises a standard battery, such as a lithium battery, having a nominal output of 3 volts.
- pulse generator 54 comprises a DC/DC converter, as is known in the art, and a capacitor, which is charged by the DC/DC converter to a constant, stepped-up voltage level regardless of the precise battery voltage, which may vary between 3.5 and 1.8 volts.
- the same DC/DC converter, or another similar device, preferably supplies power to other circuit components of control unit 22.
- An inductive arrangement using wire 28 coupled to CPU 52, is preferably used to program the CPU, using an external programming device (not shown) with a suitable antenna.
- the programming device generates a modulated magnetic field to communicate with a receiver inside case 25, which preferably senses the field using a Hall effect transducer.
- Such programming may be used, for example, to set an amplitude or duration of the stimulation waveform applied by pulse generator 54, or to set a threshold level or other parameters, according to which the CPU distinguishes between electromyographic or other signals that are indicative of impending incontinence and those that are not (e.g., those that indicate voluntary voiding).
- Such programming may be carried out by medical personnel or by the patient herself, who can similarly turn the implanted control unit on and off as desired by passing a suitable magnet over the area of her pelvis.
- control unit 22 Although the circuit blocks in control unit 22 are shown as discrete elements, some or all of these blocks are preferably embodied in a custom or semi-custom integrated circuit device, as is known in the art.
- Fig. 4 is a schematic block diagram illustrating a muscle stimulator device 70, in accordance with an alternative embodiment of the present invention.
- Device 70 is substantially similar to device 20, except for features described hereinbelow.
- Device 70 comprises a control unit 74, which is coupled to electrodes 27 and 29. Electrode 29 also serves as the sensing electrode, furnishing electromyographic signals via switch 46 to amplifier 48, as described hereinabove. Alternatively, electrodes 27 and 29 may be coupled as differential inputs to amplifier 48.
- Pulse generator 54 applies the stimulation waveforms between electrodes 27 and 29 in a bipolar mode.
- CPU 52 preferably receives additional signals from other physiological sensors, such as ultrasound transducer 44 (shown in Fig. 2), a pressure sensor 76 and/or an acceleration sensor 78, or other types of strain and motion measurement devices, as are known in the art.
- Pressure sensor 76 is preferably implanted on or in bladder 36, so as to detect increases in abdominal or intravesical pressure that may lead to involuntary urine loss.
- acceleration sensor 78 is preferably implanted so as to detect bladder motion associated with hypermobility, which is similarly associated with urine loss.
- the additional signals from these sensors are preferably analyzed by the CPU together with the electromyographic signals in order to improve the accuracy and reliability of detection of impending stress incontinence.
- An impedance sensor 79 is used to measure the tissue impedance between leads 27 and 29, using physiological impedance measurement techniques known in the art. During long-term use of device 70 (or other such devices), fibrosis in the area of the implanted electrodes tends to cause the impedance to increase, so that the stimulating current for a given applied voltage decreases. The impedance measured by sensor 79 is used as a feedback signal instructing CPU 52 to increase the voltage, so that a generally constant level of stimulation current is maintained.
- Fig. 5 is a schematic block diagram showing details of signal processing circuitry 80 for use in device 20 or 70, in accordance with a preferred embodiment of the present invention.
- A/D converter 50 In order to detect impending incontinence with adequate reliability, A/D converter 50 must typically sample the EMG signals from the electrodes at 1000-5000 Hz, and CPU 52 must perform a detailed analysis of the sample stream. Systems for incontinence control known in the art, operating at sample rates below 1000 Hz, cannot adequately distinguish between signals that may be indicative of incontinence and those that are not.
- CPU 52 preferably comprises a low-power, software-programmable processor. If A/D converter 50 and CPU 52 were to operate continuously, however, battery 56 would rapidly run down.
- circuitry 80 comprises a low-power, low-resolution A/D converter 84 and hard-coded processing logic 86, which operate continuously at a low sampling rate, preferably at about 100-200 Hz.
- Input from amplifier 48 to A/D converter 84 is preferably rectified by a rectifier 82.
- A/D converter 50 and CPU 52 are normally maintained in a standby state, in which their power consumption is negligible.
- logic 86 operating at the low sampling rate, detects EMG signals that may be a precursor to incontinence, it signals A/D converter 50 to begin sampling at the high rate.
- signals from A/D converter 84 are preferably stored in a cyclic (or first-in first-out) queue 88, such as a delay line. The entire sequence of signal detection and processing is estimated to take between 5 and 20 ms, up to the point at which CPU 52 reaches a decision as to whether or not to actuate pulse generator 54.
- Pulse generation takes between 1 and 20 ms, with the result that contraction of the pelvic muscles begins within 15-50 ms of the onset of increased EMG activity indicating impending urine loss. Thus, urethra 34 is substantially closed off before any significant amount of urine can leak out.
- EMG inputs from electrodes 27 and 29 are preferably amplified before processing in a dual-differential configuration. Electrode 27 and 29 are coupled to respective differential preamplifiers 87 and 89. The outputs of the preamplifiers are differentially amplified by amplifier 48. This configuration, which affords enhanced sensitivity and reduced noise in device 70, is shown in Fig. 4.
Landscapes
- Health & Medical Sciences (AREA)
- Urology & Nephrology (AREA)
- Cardiology (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Transplantation (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Vascular Medicine (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Electrotherapy Devices (AREA)
- Orthopedics, Nursing, And Contraception (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
- Massaging Devices (AREA)
- Electrical Control Of Ignition Timing (AREA)
- Ultra Sonic Daignosis Equipment (AREA)
- Control Of Eletrric Generators (AREA)
Claims (23)
- Dispositif pour le traitement de l'incontinence urinaire liée au stress, comprenant :au moins une électrode (27) à implanter dans un muscle pelvien d'un patient (31) ; etune unité de commande (22) adaptée pour recevoir des signaux indiquant un stress abdominal chez le patient (31) et en réponse à ceux-ci, l'unité de commande (22) est adaptée pour appliquer une forme d'onde électrique à l'électrode (27) qui stimule le muscle afin qu'il se contracte, de manière à inhiber un flux d'urine involontaire à travers l'urètre (34) du patient à cause du stress, etdans lequel l'unité de commande (22) est adaptée pour recevoir des signaux indiquant un niveau de remplissage de la vessie (36) du patient,
caractérisé en ce que l'unité de commande est en outre adaptée pour, en réponse aux dits signaux, empêcher une application de la forme d'onde électrique quand le niveau de remplissage de la vessie (36) est bas, même quand des signaux sont reçus indiquant un stress abdominal. - Dispositif selon la revendication 1, dans lequel la au moins une électrode (27) comprend une seule électrode unipolaire, une paire d'électrodes bipolaires (27, 29) ou une électrode intramusculaire flexible.
- Dispositif selon la revendication 1, dans lequel ledit muscle pelvien est le muscle sphincter de l'urètre ou est adjacent au muscle sphincter de l'urètre.
- Dispositif selon la revendication 1, dans lequel les signaux comprennent des signaux électromyographiques reçus depuis la au moins une électrode (27).
- Dispositif selon la revendication 4, et comprenant un commutateur (46) entre l'électrode (27) et une entrée de l'unité de commande (22), lequel commutateur (46) est ouvert quand la forme d'onde électrique est appliquée de manière à empêcher une rétroaction de l'électrode (27) vers l'entrée.
- Dispositif selon l'une quelconque des revendications précédentes, dans lequel la au moins une électrode (27) et l'unité de commande (22) sont implantées dans le corps du patient (31).
- Dispositif selon la revendication 6, dans lequel l'unité de commande (22) comprend une source d'alimentation rechargeable (56).
- Dispositif selon la revendication 7, dans lequel la source d'alimentation (56) est rechargée par un transfert d'énergie inductif, sensiblement sans contact électrique entre l'unité de commande (22) et un quelconque objet à l'extérieur du corps du patient.
- Dispositif selon l'une quelconque des revendications 1 à 5, et comprenant un capteur physiologique (44; 76 ; 78) couplé à la vessie du patient (36), lequel capteur (44; 76; 78) fournit au moins certains des signaux vers l'unité de commande (22).
- Dispositif selon la revendication 9, dans lequel le capteur comprend un capteur de pression (76), un capteur d'accélération (78) et un capteur ultrasonore (44).
- Dispositif selon l'une quelconque des revendications 1 à 5, dans lequel l'unité de commande (22) comprend un processeur (52), lequel analyse les signaux de manière à déterminer quand un flux d'urine involontaire est probable, après quoi la forme d'onde est appliquée.
- Dispositif selon la revendication 11, dans lequel le processeur (52) analyse les signaux à une fréquence d'échantillonnage sensiblement supérieure à 1 000 Hz.
- Dispositif selon la revendication 11, dans lequel l'analyse du processeur est réalisée sur des données sensiblement non rectifiées.
- Dispositif selon la revendication 11, dans lequel le processeur (52) analyse les signaux en utilisant une analyse spectrale.
- Dispositif selon la revendication 14, dans lequel l'analyse spectrale est réalisée par le processeur (52) sur des données sensiblement non rectifiées.
- Dispositif selon la revendication 11, dans lequel le processeur (52) peut être programmé pour varier un ou plusieurs paramètres associés à l'application de la forme d'onde.
- Dispositif selon la revendication 16, et comprenant un récepteur sans fil (28), lequel reçoit des données pour programmer le processeur (52) depuis une unité de programmation à l'extérieur du corps du patient.
- Dispositif selon la revendication 11, dans lequel le processeur (52) comprend un premier processeur, lequel analyse les signaux de manière sensiblement continue à une cadence d'analyse des données basse, et un deuxième processeur, lequel est actionné par le premier processeur pour analyser les signaux à une cadence d'analyse des données élevée quand le premier processeur détermine qu'un flux d'urine involontaire va probablement se produire.
- Dispositif selon la revendication 18, et comprenant une file d'attente, dans laquelle les signaux sont stockés avant que le deuxième processeur ne soit actionné, et depuis laquelle des signaux reçus par l'unité de commande (22) avant l'actionnement du deuxième processeur sont transférés vers le deuxième processeur pour une analyse.
- Dispositif selon la revendication 11, dans lequel le processeur (52) fait la distinction entre des signaux indiquant un flux d'urine involontaire et des signaux indiquant une évacuation volontaire par le patient (31).
- Dispositif selon la revendication 20, dans lequel le processeur (52) fait la distinction entre les signaux indiquant un flux d'urine involontaire et les signaux indiquant une évacuation volontaire par le patient (31) en réponse à un taux de variation des signaux.
- Dispositif selon la revendication 20, dans lequel le processeur (52) rassemble des informations concernant les signaux sur une période prolongée et analyse les informations pour trouver un schéma caractéristique du patient (31), à utiliser pour déterminer quand un flux d'urine involontaire est probable.
- Dispositif selon la revendication 22, dans lequel le schéma comprend un seuil variant au cours du temps auquel un niveau des signaux est comparé.
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US16724498A | 1998-10-06 | 1998-10-06 | |
| US167244 | 1998-10-06 | ||
| IL12748198 | 1998-12-09 | ||
| IL12748198A IL127481A (en) | 1998-10-06 | 1998-12-09 | Urine excretion prevention device |
| PCT/IL1999/000529 WO2000019940A1 (fr) | 1998-10-06 | 1999-10-05 | Dispositif de traitement de l'incontinence |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1124502A1 EP1124502A1 (fr) | 2001-08-22 |
| EP1124502A4 EP1124502A4 (fr) | 2005-07-20 |
| EP1124502B1 true EP1124502B1 (fr) | 2007-04-04 |
Family
ID=26323757
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP99947825A Expired - Lifetime EP1124502B1 (fr) | 1998-10-06 | 1999-10-05 | Dispositif de traitement de l'incontinence |
| EP99947824A Expired - Lifetime EP1119314B1 (fr) | 1998-10-06 | 1999-10-05 | Controle de l'incontinence instante |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP99947824A Expired - Lifetime EP1119314B1 (fr) | 1998-10-06 | 1999-10-05 | Controle de l'incontinence instante |
Country Status (6)
| Country | Link |
|---|---|
| US (4) | US6652449B1 (fr) |
| EP (2) | EP1124502B1 (fr) |
| AT (2) | ATE328548T1 (fr) |
| AU (2) | AU6118699A (fr) |
| DE (2) | DE69931797T2 (fr) |
| WO (1) | WO2000019939A1 (fr) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9457186B2 (en) | 2010-11-15 | 2016-10-04 | Bluewind Medical Ltd. | Bilateral feedback |
Families Citing this family (222)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6941171B2 (en) | 1998-07-06 | 2005-09-06 | Advanced Bionics Corporation | Implantable stimulator methods for treatment of incontinence and pain |
| US6735474B1 (en) | 1998-07-06 | 2004-05-11 | Advanced Bionics Corporation | Implantable stimulator system and method for treatment of incontinence and pain |
| US7890176B2 (en) | 1998-07-06 | 2011-02-15 | Boston Scientific Neuromodulation Corporation | Methods and systems for treating chronic pelvic pain |
| IL127481A (en) * | 1998-10-06 | 2004-05-12 | Bio Control Medical Ltd | Urine excretion prevention device |
| ATE328548T1 (de) * | 1998-10-06 | 2006-06-15 | Bio Control Medical Ltd | Kontrolle von drängender inkontinenz |
| AU6465399A (en) * | 1998-10-30 | 2000-05-22 | Aalborg Universitet | A method to control an overactive bladder |
| ES2161648B1 (es) * | 2000-05-26 | 2002-07-01 | Consejo Superior Investigacion | Sistema de control de miccion, defecacion y ereccion en pacientes con neuropatias. |
| US7054689B1 (en) * | 2000-08-18 | 2006-05-30 | Advanced Bionics Corporation | Fully implantable neurostimulator for autonomic nerve fiber stimulation as a therapy for urinary and bowel dysfunction |
| AU2002247449B2 (en) | 2001-03-30 | 2006-10-12 | Case Western Reserve University | Systems and methods for selectively stimulating components in, on, or near the pudendal nerve or its branches to achieve selective physiologic responses |
| US7047078B2 (en) * | 2001-03-30 | 2006-05-16 | Case Western Reserve University | Methods for stimulating components in, on, or near the pudendal nerve or its branches to achieve selective physiologic responses |
| SE0102312D0 (sv) | 2001-06-28 | 2001-06-28 | Obtech Medical Ag | Urinary dysfunction treatment apparatus |
| US7499753B2 (en) | 2001-06-28 | 2009-03-03 | Urologica Ag | Urinary Dysfunction Treatment Apparatus |
| SE0102313D0 (sv) * | 2001-06-28 | 2001-06-28 | Obtech Medical Ag | Intestine dysfunction treatment apparatus |
| US6754516B2 (en) | 2001-07-19 | 2004-06-22 | Nellcor Puritan Bennett Incorporated | Nuisance alarm reductions in a physiological monitor |
| US20050055063A1 (en) * | 2001-07-20 | 2005-03-10 | Loeb Gerald E. | Method and apparatus for the treatment of urinary tract dysfunction |
| US20030082884A1 (en) * | 2001-10-26 | 2003-05-01 | International Business Machine Corporation And Kabushiki Kaisha Toshiba | Method of forming low-leakage dielectric layer |
| US6862480B2 (en) * | 2001-11-29 | 2005-03-01 | Biocontrol Medical Ltd. | Pelvic disorder treatment device |
| US6712772B2 (en) | 2001-11-29 | 2004-03-30 | Biocontrol Medical Ltd. | Low power consumption implantable pressure sensor |
| WO2004047914A1 (fr) * | 2001-11-29 | 2004-06-10 | Biocontrol Medical Ltd. | Dispositif de traitement de troubles pelviens |
| EP1578247B1 (fr) | 2002-05-07 | 2015-10-28 | AMS Research Corporation | Detecteur de pression implantable a faible consommation de courant |
| US20040068162A1 (en) * | 2002-07-01 | 2004-04-08 | Vaclav Kirsner | Apparatus and method of personal screening for cervical cancer conditions in vivo |
| US7369894B2 (en) | 2002-09-06 | 2008-05-06 | Medtronic, Inc. | Method, system and device for treating disorders of the pelvic floor by electrical stimulation of the sacral and/or pudendal nerves |
| US7328068B2 (en) * | 2003-03-31 | 2008-02-05 | Medtronic, Inc. | Method, system and device for treating disorders of the pelvic floor by means of electrical stimulation of the pudendal and associated nerves, and the optional delivery of drugs in association therewith |
| US7427280B2 (en) | 2002-09-06 | 2008-09-23 | Medtronic, Inc. | Method, system and device for treating disorders of the pelvic floor by delivering drugs to various nerves or tissues |
| US7328069B2 (en) * | 2002-09-06 | 2008-02-05 | Medtronic, Inc. | Method, system and device for treating disorders of the pelvic floor by electrical stimulation of and the delivery of drugs to the left and right pudendal nerves |
| US7276057B2 (en) * | 2002-09-06 | 2007-10-02 | Medtronic, Inc. | Method, system and device for treating disorders of the pelvic floor by drug delivery to the pudendal and sacral nerves |
| EP1549274A4 (fr) * | 2002-09-27 | 2008-06-11 | Us Gov Health & Human Serv | Procedes et dispositifs de stimulation intramusculaire des groupes de muscle des voies aeriennes superieures et de la deglutition |
| US20040089308A1 (en) * | 2002-11-13 | 2004-05-13 | Welch Robert A. | Cervical ring to deliver medication |
| US7584001B2 (en) * | 2003-07-17 | 2009-09-01 | Beck Kent F | Electrical epidermal stimulation device |
| US7343202B2 (en) | 2004-02-12 | 2008-03-11 | Ndi Medical, Llc. | Method for affecting urinary function with electrode implantation in adipose tissue |
| US7381180B2 (en) * | 2003-10-31 | 2008-06-03 | Medtronic, Inc. | Implantable devices and methods for treating fecal incontinence |
| US7585271B2 (en) | 2003-11-01 | 2009-09-08 | Thd Spa | Implantable devices and methods for treating urinary incontinence |
| US7647112B2 (en) * | 2004-02-11 | 2010-01-12 | Ethicon, Inc. | System and method for selectively stimulating different body parts |
| WO2005077276A2 (fr) * | 2004-02-11 | 2005-08-25 | Ethicon, Inc. | Systeme et procede d'evaluation urodynamique mettant en oeuvre un systeme mecanique micro-electronique |
| US7979137B2 (en) | 2004-02-11 | 2011-07-12 | Ethicon, Inc. | System and method for nerve stimulation |
| US8751003B2 (en) | 2004-02-11 | 2014-06-10 | Ethicon, Inc. | Conductive mesh for neurostimulation |
| US8165695B2 (en) | 2004-02-11 | 2012-04-24 | Ethicon, Inc. | System and method for selectively stimulating different body parts |
| US8467875B2 (en) | 2004-02-12 | 2013-06-18 | Medtronic, Inc. | Stimulation of dorsal genital nerves to treat urologic dysfunctions |
| US9205255B2 (en) | 2004-06-10 | 2015-12-08 | Medtronic Urinary Solutions, Inc. | Implantable pulse generator systems and methods for providing functional and/or therapeutic stimulation of muscles and/or nerves and/or central nervous system tissue |
| WO2005123184A2 (fr) * | 2004-06-10 | 2005-12-29 | Ndi Medical, Llc | Structures de conducteurs et d'electrodes dont la taille et la configuration permettent une implantation dans le tissu adipeux et methodes d'implantation associees |
| US7761167B2 (en) | 2004-06-10 | 2010-07-20 | Medtronic Urinary Solutions, Inc. | Systems and methods for clinician control of stimulation systems |
| WO2005123181A2 (fr) | 2004-06-10 | 2005-12-29 | Ndi Medical, Llc | Generateur d'impulsions implantable destine a fournir une stimulation fonctionnelle et/ou therapeutique des muscles et/ou des nerfs et/ou des tissus du systeme nerveux central |
| US9308382B2 (en) | 2004-06-10 | 2016-04-12 | Medtronic Urinary Solutions, Inc. | Implantable pulse generator systems and methods for providing functional and/or therapeutic stimulation of muscles and/or nerves and/or central nervous system tissue |
| US8165692B2 (en) | 2004-06-10 | 2012-04-24 | Medtronic Urinary Solutions, Inc. | Implantable pulse generator power management |
| US8195304B2 (en) * | 2004-06-10 | 2012-06-05 | Medtronic Urinary Solutions, Inc. | Implantable systems and methods for acquisition and processing of electrical signals |
| US20060020225A1 (en) * | 2004-07-20 | 2006-01-26 | Gerber Martin T | Wireless urodynamic monitoring system with automated voiding diary |
| US7729772B2 (en) * | 2005-01-07 | 2010-06-01 | Uroplasty, Inc. | Implantable neuromodulation system and method |
| US7775966B2 (en) | 2005-02-24 | 2010-08-17 | Ethicon Endo-Surgery, Inc. | Non-invasive pressure measurement in a fluid adjustable restrictive device |
| GB2423020A (en) * | 2005-02-14 | 2006-08-16 | Algotec Ltd | Percutaneous electrical stimulation probe for pain relief |
| US7515965B2 (en) * | 2005-02-23 | 2009-04-07 | Medtronic, Inc. | Implantable medical device providing adaptive neurostimulation therapy for incontinence |
| US7927270B2 (en) | 2005-02-24 | 2011-04-19 | Ethicon Endo-Surgery, Inc. | External mechanical pressure sensor for gastric band pressure measurements |
| US7658196B2 (en) | 2005-02-24 | 2010-02-09 | Ethicon Endo-Surgery, Inc. | System and method for determining implanted device orientation |
| US7775215B2 (en) | 2005-02-24 | 2010-08-17 | Ethicon Endo-Surgery, Inc. | System and method for determining implanted device positioning and obtaining pressure data |
| US8016744B2 (en) | 2005-02-24 | 2011-09-13 | Ethicon Endo-Surgery, Inc. | External pressure-based gastric band adjustment system and method |
| US8066629B2 (en) | 2005-02-24 | 2011-11-29 | Ethicon Endo-Surgery, Inc. | Apparatus for adjustment and sensing of gastric band pressure |
| US7699770B2 (en) | 2005-02-24 | 2010-04-20 | Ethicon Endo-Surgery, Inc. | Device for non-invasive measurement of fluid pressure in an adjustable restriction device |
| US7328070B2 (en) * | 2005-04-28 | 2008-02-05 | Medtronic, Inc. | Multi-tube sensor for sensing urinary sphincter and urethral pressure |
| US8068910B2 (en) | 2005-04-28 | 2011-11-29 | Medtronic, Inc. | Flexible tube sensor for sensing urinary sphincter pressure |
| US7623923B2 (en) * | 2005-04-28 | 2009-11-24 | Medtronic, Inc. | Tube sensor for penile tumescence |
| US7610093B2 (en) * | 2005-04-28 | 2009-10-27 | Medtronic, Inc. | Implantable optical pressure sensor for sensing urinary sphincter pressure |
| US8588930B2 (en) * | 2005-06-07 | 2013-11-19 | Ethicon, Inc. | Piezoelectric stimulation device |
| US20070225616A1 (en) * | 2005-06-15 | 2007-09-27 | Alpine Biomed Corp. | Wireless urinary incontinence monitoring system |
| US8388561B2 (en) * | 2005-07-01 | 2013-03-05 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Systems and methods for recovery from motor control via stimulation to a substituted site to an affected area |
| EP1917067A1 (fr) | 2005-07-01 | 2008-05-07 | The Government of the United States of America as represented by The Secretary of the Department of Health and Human Services | Systemes et methodes destines a la recuperation d'une faculte motrice par stimulation d'un site de substitution d'une zone affectee |
| US20070027495A1 (en) * | 2005-07-29 | 2007-02-01 | Medtronic, Inc. | External bladder sensor for sensing bladder condition |
| US7769460B2 (en) * | 2005-07-29 | 2010-08-03 | Medtronic, Inc. | Transmembrane sensing device for sensing bladder condition |
| US9061146B2 (en) * | 2005-10-28 | 2015-06-23 | Medtronic, Inc. | Impedance-based bladder sensing |
| US20070100388A1 (en) * | 2005-10-31 | 2007-05-03 | Medtronic, Inc. | Implantable medical device providing adaptive neurostimulation therapy for incontinence |
| US8195296B2 (en) | 2006-03-03 | 2012-06-05 | Ams Research Corporation | Apparatus for treating stress and urge incontinence |
| US8170686B2 (en) * | 2006-03-14 | 2012-05-01 | Boston Scientific Scimed, Inc. | Heatable sling support for an anatomical location |
| US8449445B2 (en) | 2006-03-30 | 2013-05-28 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Device for volitional swallowing with a substitute sensory system |
| WO2007126632A2 (fr) * | 2006-04-04 | 2007-11-08 | Ams Research Corporation | Systèmes et procédés pour implanter des dispositifs médicaux |
| US8870742B2 (en) | 2006-04-06 | 2014-10-28 | Ethicon Endo-Surgery, Inc. | GUI for an implantable restriction device and a data logger |
| US8152710B2 (en) | 2006-04-06 | 2012-04-10 | Ethicon Endo-Surgery, Inc. | Physiological parameter analysis for an implantable restriction device and a data logger |
| US7522061B2 (en) | 2006-04-28 | 2009-04-21 | Medtronic, Inc. | External voiding sensor system |
| US7855653B2 (en) * | 2006-04-28 | 2010-12-21 | Medtronic, Inc. | External voiding sensor system |
| US20070255176A1 (en) * | 2006-04-28 | 2007-11-01 | Medtronic, Inc. | Voiding detection with learning mode |
| US20070265675A1 (en) * | 2006-05-09 | 2007-11-15 | Ams Research Corporation | Testing Efficacy of Therapeutic Mechanical or Electrical Nerve or Muscle Stimulation |
| US9480846B2 (en) | 2006-05-17 | 2016-11-01 | Medtronic Urinary Solutions, Inc. | Systems and methods for patient control of stimulation systems |
| US8047215B1 (en) * | 2006-06-06 | 2011-11-01 | Larry Sasaki | Laparoscopic lens cleaner |
| US8160710B2 (en) | 2006-07-10 | 2012-04-17 | Ams Research Corporation | Systems and methods for implanting tissue stimulation electrodes in the pelvic region |
| US20080103572A1 (en) | 2006-10-31 | 2008-05-01 | Medtronic, Inc. | Implantable medical lead with threaded fixation |
| US20080132962A1 (en) * | 2006-12-01 | 2008-06-05 | Diubaldi Anthony | System and method for affecting gatric functions |
| US7647113B2 (en) | 2006-12-21 | 2010-01-12 | Ams Research Corporation | Electrode implantation in male external urinary sphincter |
| US20080177398A1 (en) * | 2007-01-24 | 2008-07-24 | Yossi Gross | Treatment of bladder dysfunction |
| US8295933B2 (en) * | 2007-05-30 | 2012-10-23 | Medtronic, Inc. | Implantable medical lead including voiding event sensor |
| US8121691B2 (en) * | 2007-05-30 | 2012-02-21 | Medtronic, Inc. | Voiding event identification based on patient input |
| US9185489B2 (en) | 2007-05-30 | 2015-11-10 | Medtronic, Inc. | Automatic voiding diary |
| US9427573B2 (en) | 2007-07-10 | 2016-08-30 | Astora Women's Health, Llc | Deployable electrode lead anchor |
| US20100049289A1 (en) | 2007-07-10 | 2010-02-25 | Ams Research Corporation | Tissue anchor |
| KR101494190B1 (ko) | 2007-07-27 | 2015-02-17 | 에이엠에스 리서치 코포레이션 | 골반저 치료 관련 장치 및 임플란트 |
| US8352026B2 (en) | 2007-10-03 | 2013-01-08 | Ethicon, Inc. | Implantable pulse generators and methods for selective nerve stimulation |
| HUE055743T2 (hu) * | 2007-10-11 | 2021-12-28 | Implantica Patent Ltd | Készülék petesejtek petevezetékben való áramlásának szabályozására |
| US7918782B2 (en) * | 2007-11-15 | 2011-04-05 | Ams Research Corporation | Prosthesis with bladder that adjusts girth |
| US8187163B2 (en) | 2007-12-10 | 2012-05-29 | Ethicon Endo-Surgery, Inc. | Methods for implanting a gastric restriction device |
| US8100870B2 (en) | 2007-12-14 | 2012-01-24 | Ethicon Endo-Surgery, Inc. | Adjustable height gastric restriction devices and methods |
| US8142452B2 (en) | 2007-12-27 | 2012-03-27 | Ethicon Endo-Surgery, Inc. | Controlling pressure in adjustable restriction devices |
| US8377079B2 (en) | 2007-12-27 | 2013-02-19 | Ethicon Endo-Surgery, Inc. | Constant force mechanisms for regulating restriction devices |
| US8337389B2 (en) | 2008-01-28 | 2012-12-25 | Ethicon Endo-Surgery, Inc. | Methods and devices for diagnosing performance of a gastric restriction system |
| US8192350B2 (en) | 2008-01-28 | 2012-06-05 | Ethicon Endo-Surgery, Inc. | Methods and devices for measuring impedance in a gastric restriction system |
| US8591395B2 (en) | 2008-01-28 | 2013-11-26 | Ethicon Endo-Surgery, Inc. | Gastric restriction device data handling devices and methods |
| US7844342B2 (en) | 2008-02-07 | 2010-11-30 | Ethicon Endo-Surgery, Inc. | Powering implantable restriction systems using light |
| US8221439B2 (en) | 2008-02-07 | 2012-07-17 | Ethicon Endo-Surgery, Inc. | Powering implantable restriction systems using kinetic motion |
| US8114345B2 (en) | 2008-02-08 | 2012-02-14 | Ethicon Endo-Surgery, Inc. | System and method of sterilizing an implantable medical device |
| US8057492B2 (en) | 2008-02-12 | 2011-11-15 | Ethicon Endo-Surgery, Inc. | Automatically adjusting band system with MEMS pump |
| US8591532B2 (en) | 2008-02-12 | 2013-11-26 | Ethicon Endo-Sugery, Inc. | Automatically adjusting band system |
| US8034065B2 (en) | 2008-02-26 | 2011-10-11 | Ethicon Endo-Surgery, Inc. | Controlling pressure in adjustable restriction devices |
| US8187162B2 (en) | 2008-03-06 | 2012-05-29 | Ethicon Endo-Surgery, Inc. | Reorientation port |
| US8233995B2 (en) | 2008-03-06 | 2012-07-31 | Ethicon Endo-Surgery, Inc. | System and method of aligning an implantable antenna |
| WO2009145911A1 (fr) * | 2008-05-29 | 2009-12-03 | Ams Research Corporation | Réparation d'avulsion de muscle releveur à invasion minimale |
| US10603489B2 (en) | 2008-10-09 | 2020-03-31 | Virender K. Sharma | Methods and apparatuses for stimulating blood vessels in order to control, treat, and/or prevent a hemorrhage |
| WO2010042686A1 (fr) | 2008-10-09 | 2010-04-15 | Sharma Virender K | Procédé et appareil pour stimuler le système vasculaire |
| SI2349082T1 (sl) * | 2008-10-10 | 2021-09-30 | Implantica Patent Ltd. | Implantabilna naprava za notranje nadziranje urina |
| US20100160715A1 (en) * | 2008-12-23 | 2010-06-24 | Yiming Deng | Method of minimal invasive tunneling |
| US8545391B2 (en) * | 2008-12-23 | 2013-10-01 | Ams Research Corporation | Penile prosthesis implantation device |
| US20110301663A1 (en) | 2009-02-18 | 2011-12-08 | Ams Research Corporation | Treatment of a pelvic condition through indirect electrical stimulation |
| US20100217340A1 (en) * | 2009-02-23 | 2010-08-26 | Ams Research Corporation | Implantable Medical Device Connector System |
| EP2416842B1 (fr) | 2009-03-17 | 2017-07-05 | AMS Research Corporation | Outil destiné à l'implantation d'électrodes |
| US9539433B1 (en) | 2009-03-18 | 2017-01-10 | Astora Women's Health, Llc | Electrode implantation in a pelvic floor muscular structure |
| US9155885B2 (en) | 2009-04-24 | 2015-10-13 | Medtronic, Inc. | Incontinence therapy |
| WO2011003072A2 (fr) | 2009-07-02 | 2011-01-06 | Ams Research Corporation | Implantation d'électrodes dans le sphincter uréthral |
| EP2295109A3 (fr) | 2009-09-11 | 2011-05-25 | AMS Research Corporation | Ancrage de tissus |
| US8465421B2 (en) * | 2009-12-14 | 2013-06-18 | C2Cure Inc. | Endoscope with an improved working channel |
| US8380312B2 (en) | 2009-12-31 | 2013-02-19 | Ams Research Corporation | Multi-zone stimulation implant system and method |
| US20130053688A1 (en) | 2010-05-13 | 2013-02-28 | Ams Research Corporation | Implantable mechanical support |
| US8805519B2 (en) | 2010-09-30 | 2014-08-12 | Nevro Corporation | Systems and methods for detecting intrathecal penetration |
| US20120277583A1 (en) | 2011-04-29 | 2012-11-01 | Pneumoflex Systems, Llc | System and method for testing the gastric valve |
| US9232917B2 (en) | 2011-04-29 | 2016-01-12 | Pneumoflex Systems, Llc | Urinary catheter system for diagnosing a physiological abnormality such as stress urinary incontinence |
| US9220887B2 (en) | 2011-06-09 | 2015-12-29 | Astora Women's Health LLC | Electrode lead including a deployable tissue anchor |
| US9399135B2 (en) | 2011-07-12 | 2016-07-26 | Astora Women's Health, Llc | Electronic stimulator device pulse generator circuit |
| WO2013025678A1 (fr) | 2011-08-16 | 2013-02-21 | Ams Research Corporation | Ancrage tissulaire à fil d'électrode |
| US9731112B2 (en) | 2011-09-08 | 2017-08-15 | Paul J. Gindele | Implantable electrode assembly |
| WO2013059171A1 (fr) | 2011-10-20 | 2013-04-25 | Ams Research Corporation | Dispositif de stimulation électrique à accès distant |
| EP3295991A1 (fr) | 2011-10-20 | 2018-03-21 | AMS Research Corporation | Dispositif de stimulation électrique comportant plusieurs canaux de stimulation |
| US8630709B2 (en) | 2011-12-07 | 2014-01-14 | Cyberonics, Inc. | Computer-implemented system and method for selecting therapy profiles of electrical stimulation of cervical vagus nerves for treatment of chronic cardiac dysfunction |
| US8918191B2 (en) | 2011-12-07 | 2014-12-23 | Cyberonics, Inc. | Implantable device for providing electrical stimulation of cervical vagus nerves for treatment of chronic cardiac dysfunction with bounded titration |
| US8600505B2 (en) | 2011-12-07 | 2013-12-03 | Cyberonics, Inc. | Implantable device for facilitating control of electrical stimulation of cervical vagus nerves for treatment of chronic cardiac dysfunction |
| US8918190B2 (en) | 2011-12-07 | 2014-12-23 | Cyberonics, Inc. | Implantable device for evaluating autonomic cardiovascular drive in a patient suffering from chronic cardiac dysfunction |
| US8577458B1 (en) | 2011-12-07 | 2013-11-05 | Cyberonics, Inc. | Implantable device for providing electrical stimulation of cervical vagus nerves for treatment of chronic cardiac dysfunction with leadless heart rate monitoring |
| US10188856B1 (en) | 2011-12-07 | 2019-01-29 | Cyberonics, Inc. | Implantable device for providing electrical stimulation of cervical vagus nerves for treatment of chronic cardiac dysfunction |
| EP2795574A4 (fr) | 2011-12-21 | 2015-04-01 | Sca Hygiene Prod Ab | Procédé de mesure de l'absorption d'un fluide dans un produit absorbant |
| EP2793785B1 (fr) | 2011-12-21 | 2019-11-06 | Essity Hygiene and Health Aktiebolag | Procédé et programme informatique pour surveiller l'utilisation d'un produit absorbant |
| BR112014015474B1 (pt) * | 2011-12-21 | 2020-12-22 | Essity Hygiene And Health Aktiebolag | método para monitorar o uso de produtos absorventes e dispositivo móvel para monitorar o uso de um produto absorvente |
| US8700150B2 (en) | 2012-01-17 | 2014-04-15 | Cyberonics, Inc. | Implantable neurostimulator for providing electrical stimulation of cervical vagus nerves for treatment of chronic cardiac dysfunction with bounded titration |
| US8571654B2 (en) | 2012-01-17 | 2013-10-29 | Cyberonics, Inc. | Vagus nerve neurostimulator with multiple patient-selectable modes for treating chronic cardiac dysfunction |
| ES2421055B1 (es) * | 2012-01-24 | 2014-12-17 | Tequir S.L. | Sistema para el tratamiento de la escoliosis idiopática |
| ES2829585T3 (es) | 2012-01-25 | 2021-06-01 | Nevro Corp | Anclajes de cables y sistemas y métodos asociados |
| US9782583B2 (en) | 2012-02-21 | 2017-10-10 | Virender K. Sharma | System and method for electrical stimulation of anorectal structures to treat urinary dysfunction |
| FR2988996B1 (fr) | 2012-04-06 | 2015-01-23 | Uromems | Methode et dispositif de controle d'un dispositif implantable |
| US10576278B2 (en) | 2012-02-21 | 2020-03-03 | Virender K. Sharma | System and method for electrical stimulation of anorectal structures to treat urinary dysfunction |
| US8706234B2 (en) | 2012-02-21 | 2014-04-22 | Virender K. Sharma | System and method for electrical stimulation of anorectal structures to treat anal dysfunction |
| US9089426B2 (en) | 2012-03-21 | 2015-07-28 | Ams Research Corporation | Automated implantable penile prosthesis pump system |
| EP2669537A1 (fr) | 2012-06-01 | 2013-12-04 | Alstom Technology Ltd. | Palier à patins pour turbine |
| US8688212B2 (en) | 2012-07-20 | 2014-04-01 | Cyberonics, Inc. | Implantable neurostimulator-implemented method for managing bradycardia through vagus nerve stimulation |
| WO2014036449A1 (fr) | 2012-08-31 | 2014-03-06 | Alfred E. Mann Foundation For Scientific Research | Dispositif de commande de bobine asservi par rétroaction destiné à permettre un transfert de puissance inductif |
| US9452290B2 (en) | 2012-11-09 | 2016-09-27 | Cyberonics, Inc. | Implantable neurostimulator-implemented method for managing tachyarrhythmia through vagus nerve stimulation |
| US8923964B2 (en) | 2012-11-09 | 2014-12-30 | Cyberonics, Inc. | Implantable neurostimulator-implemented method for enhancing heart failure patient awakening through vagus nerve stimulation |
| US9643008B2 (en) | 2012-11-09 | 2017-05-09 | Cyberonics, Inc. | Implantable neurostimulator-implemented method for enhancing post-exercise recovery through vagus nerve stimulation |
| WO2014113813A1 (fr) | 2013-01-21 | 2014-07-24 | Cala Health, Inc. | Dispositifs et procédés pour contrôler les tremblements |
| US20140276270A1 (en) | 2013-03-13 | 2014-09-18 | Passy-Muir, Inc. | Systems and methods for stimulating swallowing |
| US9643011B2 (en) | 2013-03-14 | 2017-05-09 | Cyberonics, Inc. | Implantable neurostimulator-implemented method for managing tachyarrhythmic risk during sleep through vagus nerve stimulation |
| JP6174781B2 (ja) | 2013-03-15 | 2017-08-02 | アルフレッド イー. マン ファウンデーション フォー サイエンティフィック リサーチ | 高電圧モニタリングを行う逐次比較型アナログ・デジタル変換器 |
| AU2014232252B2 (en) | 2013-03-15 | 2018-01-18 | Alfred E. Mann Foundation For Scientific Research | Current sensing multiple output current stimulators with fast turn on time |
| CN104602760B (zh) | 2013-05-03 | 2017-11-07 | 艾尔弗雷德·E·曼科学研究基金会 | 用于植入式装置的高可靠性导线焊接 |
| JP6842917B2 (ja) | 2013-05-03 | 2021-03-17 | アルフレッド イー. マン ファウンデーション フォー サイエンティフィック リサーチ | インプラント充電器ハンドシェイキングシステム及び方法 |
| JP6513638B2 (ja) | 2013-05-03 | 2019-05-15 | アルフレッド イー. マン ファウンデーション フォー サイエンティフィック リサーチ | 皮下領域刺激のための多枝刺激電極 |
| US9328698B2 (en) | 2013-05-14 | 2016-05-03 | Ford Global Technologies, Llc | Automotive virtual humidity sensor |
| US9265935B2 (en) | 2013-06-28 | 2016-02-23 | Nevro Corporation | Neurological stimulation lead anchors and associated systems and methods |
| CA2919474C (fr) | 2013-07-29 | 2020-05-05 | Alfred E. Mann Foundation For Scientific Research | Circuit d'attaque de classe e commande par microprocesseur |
| JP6503351B2 (ja) | 2013-07-29 | 2019-04-17 | アルフレッド イー. マン ファウンデーション フォー サイエンティフィック リサーチ | 埋め込み可能なデバイス用の高効率磁気リンク |
| CN103479353B (zh) * | 2013-09-05 | 2015-02-25 | 常州大学 | 一种卧床失禁病人无线预警系统及方法 |
| CA2924817C (fr) | 2013-09-16 | 2023-09-19 | The Board Of Trustees Of The Leland Stanford Junior University | Coupleur a elements multiples permettant la production d'une energie electromagnetique |
| US9999773B2 (en) | 2013-10-30 | 2018-06-19 | Cyberonics, Inc. | Implantable neurostimulator-implemented method utilizing multi-modal stimulation parameters |
| US9511228B2 (en) | 2014-01-14 | 2016-12-06 | Cyberonics, Inc. | Implantable neurostimulator-implemented method for managing hypertension through renal denervation and vagus nerve stimulation |
| US9409024B2 (en) | 2014-03-25 | 2016-08-09 | Cyberonics, Inc. | Neurostimulation in a neural fulcrum zone for the treatment of chronic cardiac dysfunction |
| US9713719B2 (en) | 2014-04-17 | 2017-07-25 | Cyberonics, Inc. | Fine resolution identification of a neural fulcrum for the treatment of chronic cardiac dysfunction |
| US9272143B2 (en) | 2014-05-07 | 2016-03-01 | Cyberonics, Inc. | Responsive neurostimulation for the treatment of chronic cardiac dysfunction |
| US9415224B2 (en) | 2014-04-25 | 2016-08-16 | Cyberonics, Inc. | Neurostimulation and recording of physiological response for the treatment of chronic cardiac dysfunction |
| US9950169B2 (en) | 2014-04-25 | 2018-04-24 | Cyberonics, Inc. | Dynamic stimulation adjustment for identification of a neural fulcrum |
| US20160336813A1 (en) | 2015-05-15 | 2016-11-17 | NeuSpera Medical Inc. | Midfield coupler |
| EP3294173B1 (fr) | 2014-05-18 | 2020-07-15 | Neuspera Medical Inc. | Coupleur de champ intermédiaire |
| WO2015187712A1 (fr) | 2014-06-02 | 2015-12-10 | Cala Health, Inc. | Systèmes et procédés de stimulation de nerf périphérique pour traiter un tremblement |
| US9533153B2 (en) | 2014-08-12 | 2017-01-03 | Cyberonics, Inc. | Neurostimulation titration process |
| US9770599B2 (en) | 2014-08-12 | 2017-09-26 | Cyberonics, Inc. | Vagus nerve stimulation and subcutaneous defibrillation system |
| US9737716B2 (en) | 2014-08-12 | 2017-08-22 | Cyberonics, Inc. | Vagus nerve and carotid baroreceptor stimulation system |
| ES2782556T3 (es) | 2014-08-15 | 2020-09-15 | Axonics Modulation Tech Inc | Sistema para configuraciones de electrodos de neuroestimulación basadas en localización neuronal |
| US9700731B2 (en) | 2014-08-15 | 2017-07-11 | Axonics Modulation Technologies, Inc. | Antenna and methods of use for an implantable nerve stimulator |
| US10682521B2 (en) | 2014-08-15 | 2020-06-16 | Axonics Modulation Technologies, Inc. | Attachment devices and associated methods of use with a nerve stimulation charging device |
| AU2015301398B2 (en) | 2014-08-15 | 2020-05-21 | Axonics Modulation Technologies, Inc. | Implantable lead affixation structure for nerve stimulation to alleviate bladder dysfunction and other indications |
| US9802051B2 (en) | 2014-08-15 | 2017-10-31 | Axonics Modulation Technologies, Inc. | External pulse generator device and associated methods for trial nerve stimulation |
| CA2958210C (fr) | 2014-08-15 | 2023-09-26 | Axonics Modulation Technologies, Inc. | Programmateur clinicien electromyographique integre destine a etre utilise avec un neurostimulateur implantable |
| US9555246B2 (en) | 2014-08-15 | 2017-01-31 | Axonics Modulation Technologies, Inc. | Electromyographic lead positioning and stimulation titration in a nerve stimulation system for treatment of overactive bladder |
| US9504832B2 (en) | 2014-11-12 | 2016-11-29 | Cyberonics, Inc. | Neurostimulation titration process via adaptive parametric modification |
| AU2016205050B2 (en) | 2015-01-09 | 2020-02-27 | Axonics Modulation Technologies, Inc. | Attachment devices and associated methods of use with a nerve stimulation charging device |
| CN107427675B (zh) | 2015-01-09 | 2021-10-26 | 艾克索尼克斯股份有限公司 | 患者遥控器及其与神经刺激系统一起使用的相关联方法 |
| CN112914514A (zh) | 2015-06-10 | 2021-06-08 | 卡拉健康公司 | 用于外周神经刺激以利用可拆卸治疗和监测单元治疗震颤的系统和方法 |
| CN110101968B (zh) | 2015-07-10 | 2023-09-08 | 艾克索尼克斯股份有限公司 | 具有无asic的内部电子设备的可植入神经刺激器以及使用方法 |
| FR3038841A1 (fr) * | 2015-07-15 | 2017-01-20 | Akse | Dispositif de sonde pour la gestion de l'incontinence urinaire a l'effort |
| US10456580B2 (en) | 2015-09-04 | 2019-10-29 | Medtronic, Inc. | Managing therapy delivery based on physiological markers |
| WO2017053847A1 (fr) | 2015-09-23 | 2017-03-30 | Cala Health, Inc. | Systèmes et procédés pour la stimulation des nerfs périphériques dans le doigt ou la main pour traiter des tremblements dans la main |
| US10195423B2 (en) | 2016-01-19 | 2019-02-05 | Axonics Modulation Technologies, Inc. | Multichannel clip device and methods of use |
| US9517338B1 (en) | 2016-01-19 | 2016-12-13 | Axonics Modulation Technologies, Inc. | Multichannel clip device and methods of use |
| AU2017211048B2 (en) | 2016-01-21 | 2022-03-10 | Cala Health, Inc. | Systems, methods and devices for peripheral neuromodulation for treating diseases related to overactive bladder |
| CA3012828C (fr) | 2016-01-29 | 2024-04-30 | Axonics Modulation Technologies, Inc. | Procedes et systemes pour ajustement de frequence pour optimiser la charge d'un neurostimulateur implantable |
| ES2883179T3 (es) | 2016-02-12 | 2021-12-07 | Axonics Inc | Dispositivo generador de impulsos externo para estimulación nerviosa de prueba |
| US11045649B2 (en) | 2016-02-19 | 2021-06-29 | Medtronic, Inc. | Incontinence therapy |
| WO2018009680A1 (fr) | 2016-07-08 | 2018-01-11 | Cala Health, Inc. | Systèmes et procédés pour stimuler n nerfs avec exactement n électrodes et électrodes sèches améliorées |
| US11331480B2 (en) | 2017-04-03 | 2022-05-17 | Cala Health, Inc. | Systems, methods and devices for peripheral neuromodulation for treating diseases related to overactive bladder |
| US11896823B2 (en) | 2017-04-04 | 2024-02-13 | Btl Healthcare Technologies A.S. | Method and device for pelvic floor tissue treatment |
| US11857778B2 (en) | 2018-01-17 | 2024-01-02 | Cala Health, Inc. | Systems and methods for treating inflammatory bowel disease through peripheral nerve stimulation |
| CA3090810A1 (fr) | 2018-02-22 | 2019-08-29 | Axonics Modulation Technologies, Inc. | Fils de neurostimulation pour stimulation nerveuse d'essai et procedes d'utilisation |
| WO2020051341A1 (fr) * | 2018-09-05 | 2020-03-12 | University Of Miami | Dispositif personnalisé pour détecter et réduire automatiquement des spasmes musculaires avec des vibrations |
| US11510766B2 (en) | 2019-02-14 | 2022-11-29 | Uromedica, Inc. | Method and apparatus for monitoring implantable device for urinary continence |
| WO2020185902A1 (fr) | 2019-03-11 | 2020-09-17 | Axonics Modulation Technologies, Inc. | Dispositif de charge doté de bobine décentrée |
| US20200330323A1 (en) | 2019-04-19 | 2020-10-22 | Alex Jolly | Vibratory Nerve Exciter |
| US11439829B2 (en) | 2019-05-24 | 2022-09-13 | Axonics, Inc. | Clinician programmer methods and systems for maintaining target operating temperatures |
| US11848090B2 (en) | 2019-05-24 | 2023-12-19 | Axonics, Inc. | Trainer for a neurostimulator programmer and associated methods of use with a neurostimulation system |
| US11890468B1 (en) | 2019-10-03 | 2024-02-06 | Cala Health, Inc. | Neurostimulation systems with event pattern detection and classification |
| EP4048369A1 (fr) | 2019-10-22 | 2022-08-31 | Hologic, Inc. | Traitement transvaginal de l'incontinence urinaire provoquée par le stress |
| WO2021119098A1 (fr) * | 2019-12-11 | 2021-06-17 | Galvani Bioelectronics Limited | Stimulation par salves du nerf pudendal pour contrôler la vessie |
| US12076564B2 (en) | 2020-04-14 | 2024-09-03 | Medtronic, Inc. | Patient specific optimization algorithm |
| DE102022101549A1 (de) | 2022-01-24 | 2023-07-27 | Friedrich-Alexander Universität Erlangen-Nürnberg, Körperschaft des öffentlichen Rechts | Künstlicher Schließmuskel |
Family Cites Families (112)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1227186A (fr) | 1968-09-18 | 1971-04-07 | ||
| US3662758A (en) * | 1969-06-30 | 1972-05-16 | Mentor Corp | Stimulator apparatus for muscular organs with external transmitter and implantable receiver |
| US3640284A (en) * | 1970-01-05 | 1972-02-08 | Philip A De Langis | Apparatus for electrotherapy of the pubococcygeus |
| GB1434524A (en) | 1972-04-27 | 1976-05-05 | Nat Res Dev | Urinary control apparatus |
| GB1452262A (en) | 1972-07-13 | 1976-10-13 | Devices Implants Ltd | Pessary ring electrode system |
| US3941136A (en) | 1973-11-21 | 1976-03-02 | Neuronyx Corporation | Method for artificially inducing urination, defecation, or sexual excitation |
| AT332528B (de) | 1974-10-18 | 1976-10-11 | Nemec Hans | Elektromedizinischer apparat |
| US3926178A (en) | 1975-01-17 | 1975-12-16 | Alvin N Feldzamen | Apparatus for aiding the voluntary exercising of sphincter muscles |
| US3983865A (en) | 1975-02-05 | 1976-10-05 | Shepard Richard S | Method and apparatus for myofunctional biofeedback |
| US3983881A (en) | 1975-05-21 | 1976-10-05 | Telectronics Pty. Limited | Muscle stimulator |
| SE7604553L (sv) | 1976-04-21 | 1977-10-22 | Svenska Utvecklings Ab | Elektrisk stimulator for avhjelpande av inkontinens |
| US4136684A (en) | 1977-02-07 | 1979-01-30 | Scattergood Mark G | Linear electromyographic biofeedback system |
| US4139006A (en) | 1977-03-18 | 1979-02-13 | Corey Arthur E | Female incontinence device |
| US4222377A (en) | 1977-06-27 | 1980-09-16 | American Medical Systems, Inc. | Pressure regulated artificial sphincter systems |
| US4153059A (en) | 1977-10-25 | 1979-05-08 | Minnesota Mining And Manufacturing Company | Urinary incontinence stimulator system |
| US4157087A (en) | 1978-03-06 | 1979-06-05 | Med General, Inc. | Peripheral nerve stimulator |
| US4165750A (en) | 1978-03-18 | 1979-08-28 | Aleev Leonid S | Bioelectrically controlled electric stimulator of human muscles |
| US4177819A (en) | 1978-03-30 | 1979-12-11 | Kofsky Harvey I | Muscle stimulating apparatus |
| US4290420A (en) | 1980-06-09 | 1981-09-22 | Alberto Manetta | Stress incontinence diagnostic and treatment device |
| GB2099304B (en) | 1981-02-06 | 1985-01-30 | Cornwell George Herbert Ivan | Incontinence control devices |
| US4406288A (en) | 1981-04-06 | 1983-09-27 | Hugh P. Cash | Bladder control device and method |
| US4688575A (en) | 1982-03-12 | 1987-08-25 | Duvall Wilbur E | Muscle contraction stimulation |
| US4492233A (en) | 1982-09-14 | 1985-01-08 | Wright State University | Method and apparatus for providing feedback-controlled muscle stimulation |
| US4731083A (en) | 1982-09-21 | 1988-03-15 | The Johns Hopkins University | Manually actuated hydraulic sphincter |
| US4571749A (en) | 1982-09-21 | 1986-02-25 | The Johns Hopkins University | Manually actuated hydraulic sphincter |
| EP0109217A1 (fr) | 1982-11-05 | 1984-05-23 | Craig Medical Products Limited | Dispositif d'incontinence pour femme |
| US4542753A (en) | 1982-12-22 | 1985-09-24 | Biosonics, Inc. | Apparatus and method for stimulating penile erectile tissue |
| US4515167A (en) | 1983-02-28 | 1985-05-07 | Hochman Joel S | Device for the development, training and rehabilitation of the pubococcygeal and related perineal musculature of the female |
| DE3317118C2 (de) | 1983-05-06 | 1986-08-28 | Richard Wolf Gmbh, 7134 Knittlingen | Vorrichtung zur Behandlung der weiblichen Incontinentia Urinae |
| US4585005A (en) | 1984-04-06 | 1986-04-29 | Regents Of University Of California | Method and pacemaker for stimulating penile erection |
| US4607639A (en) | 1984-05-18 | 1986-08-26 | Regents Of The University Of California | Method and system for controlling bladder evacuation |
| US4739764A (en) | 1984-05-18 | 1988-04-26 | The Regents Of The University Of California | Method for stimulating pelvic floor muscles for regulating pelvic viscera |
| US4602624A (en) | 1984-10-11 | 1986-07-29 | Case Western Reserve University | Implantable cuff, method of manufacture, and method of installation |
| US4628942A (en) | 1984-10-11 | 1986-12-16 | Case Western Reserve University | Asymmetric shielded two electrode cuff |
| US4735205A (en) | 1986-02-24 | 1988-04-05 | Medtronic, Inc. | Method and apparatus including a sliding insulation lead for cardiac assistance |
| US4785828A (en) | 1986-10-06 | 1988-11-22 | Empi, Inc. | Vaginal stimulator for controlling urinary incontinence in women |
| US4881526A (en) | 1988-05-27 | 1989-11-21 | Empi, Inc. | Intravaginal electrode and stimulation system for controlling female urinary incontinence |
| US5013292A (en) | 1989-02-24 | 1991-05-07 | R. Laborie Medical Corporation | Surgical correction of female urinary stress incontinence and kit therefor |
| JPH0724668B2 (ja) * | 1990-05-02 | 1995-03-22 | 明夫 山田 | 尿失禁予防モニタ装置 |
| ATE160290T1 (de) | 1990-05-26 | 1997-12-15 | Med El Medical Electronics Ele | Einrichtung zur neuromuskulären elektroreizung |
| US6131575A (en) | 1991-01-10 | 2000-10-17 | The Procter & Gamble Company | Urinary incontinence device |
| US5366493A (en) | 1991-02-04 | 1994-11-22 | Case Western Reserve University | Double helix functional stimulation electrode |
| US5385577A (en) | 1992-11-12 | 1995-01-31 | Empi, Inc. | Electrode for activating pelvic reflexes |
| US5452719A (en) | 1991-07-23 | 1995-09-26 | Eisman; Eugene | Multiple electrode myographic probe and method |
| US5330507A (en) | 1992-04-24 | 1994-07-19 | Medtronic, Inc. | Implantable electrical vagal stimulation for prevention or interruption of life threatening arrhythmias |
| GB9211085D0 (en) | 1992-05-23 | 1992-07-08 | Tippey Keith E | Electrical stimulation |
| US5324323A (en) | 1992-09-09 | 1994-06-28 | Telectronics Pacing Systems, Inc. | Multiple channel cardiosynchronous myoplasty apparatus |
| US5291902A (en) * | 1993-01-11 | 1994-03-08 | Brent Carman | Incontinence treatment |
| US5425751A (en) | 1993-07-30 | 1995-06-20 | Medtronic, Inc. | Method and apparatus for optimum positioning of a muscle stimulating implant |
| US5484445A (en) | 1993-10-12 | 1996-01-16 | Medtronic, Inc. | Sacral lead anchoring system |
| US5400784A (en) | 1993-10-15 | 1995-03-28 | Case Western Reserve University | Slowly penetrating inter-fascicular nerve cuff electrode and method of using |
| US5370670A (en) | 1993-12-13 | 1994-12-06 | Thomas Jefferson University | Detrusor myoplasty and neuromuscular electrical stimulation of the urinary bladder |
| US5423329A (en) | 1994-04-15 | 1995-06-13 | Rehab Centers Of America, Inc. | Method of treatment for urinary incontinence |
| US5833595A (en) | 1994-09-06 | 1998-11-10 | Lin; Vernon Wen-Hau | Treatment of excretory problems |
| US5591217A (en) * | 1995-01-04 | 1997-01-07 | Plexus, Inc. | Implantable stimulator with replenishable, high value capacitive power source and method therefor |
| DE19508591A1 (de) | 1995-03-13 | 1996-10-10 | Osbon Medical Systems Ltd | Therapiegerät zur funktionellen Elektromyostimulation glatter Muskelzellen |
| US5785666A (en) | 1995-10-31 | 1998-07-28 | Ergonomic Technologies Corporation | Portable electronic data collection apparatus for monitoring musculoskeletal stresses |
| EP0855927A1 (fr) * | 1995-11-24 | 1998-08-05 | Advanced Bionics Corporation | Systeme et procede de traitement de la musculature pelvienne au moyen d'un microstimulateur implante |
| US5766229A (en) | 1996-04-15 | 1998-06-16 | Pacesetter, Inc. | Capture verification method and apparatus for implantable pacemaker utilizing heart rhythm stability measurements to minimize the likelihood of fusion |
| US6006134A (en) | 1998-04-30 | 1999-12-21 | Medtronic, Inc. | Method and device for electronically controlling the beating of a heart using venous electrical stimulation of nerve fibers |
| WO1998009679A1 (fr) | 1996-09-05 | 1998-03-12 | The Governors Of The University Of Alberta | Stimulateur electrique gastro-intestinal |
| DE69724386T2 (de) * | 1996-10-30 | 2004-04-08 | Nihon Kohden Corp. | Stimulationsvorrichtung zur Verhinderung der Harninkontinenz |
| US6026326A (en) | 1997-01-13 | 2000-02-15 | Medtronic, Inc. | Apparatus and method for treating chronic constipation |
| US5766073A (en) * | 1997-01-23 | 1998-06-16 | O'kelley; Charles H. | Exhaust header for building ventilator fan |
| US5876353A (en) | 1997-01-31 | 1999-03-02 | Medtronic, Inc. | Impedance monitor for discerning edema through evaluation of respiratory rate |
| JP2001513679A (ja) * | 1997-02-26 | 2001-09-04 | アルフレッド イー マン ファウンデーション フォア サイエンティフィック リサーチ | バッテリ給電式の患者の皮下挿入器具 |
| US5957965A (en) | 1997-03-03 | 1999-09-28 | Medtronic, Inc. | Sacral medical electrical lead |
| US6078840A (en) | 1997-04-30 | 2000-06-20 | Medtronic, Inc. | Medical electrical lead having improved fixation |
| US5836994A (en) | 1997-04-30 | 1998-11-17 | Medtronic, Inc. | Method and apparatus for electrical stimulation of the gastrointestinal tract |
| US6135945A (en) * | 1997-08-04 | 2000-10-24 | Sultan; Hashem | Anti-incontinence device |
| US5824027A (en) | 1997-08-14 | 1998-10-20 | Simon Fraser University | Nerve cuff having one or more isolated chambers |
| US7628795B2 (en) | 1997-09-24 | 2009-12-08 | Atrium Medical Corporation | Tunneling device for use with a graft |
| US6038463A (en) | 1997-09-26 | 2000-03-14 | Medtronic, Inc. | Medical electrical lead |
| US6149636A (en) | 1998-06-29 | 2000-11-21 | The Procter & Gamble Company | Disposable article having proactive sensors |
| US5963097A (en) | 1998-01-27 | 1999-10-05 | Garachtchenko; Alexander Viktorovich | Low-noise amplifier |
| US6321116B1 (en) | 1998-02-08 | 2001-11-20 | Seung Kee Mo | Electrical apparatus medical treatment using EMG envelope signal |
| US5941903A (en) | 1998-04-30 | 1999-08-24 | Cardiac Pacemakers, Inc | Pacemaker for detection of evoked response |
| US6407308B1 (en) | 1998-06-29 | 2002-06-18 | The Procter & Gamble Company | Disposable article having sensor to detect impending elimination of bodily waste |
| US6735474B1 (en) * | 1998-07-06 | 2004-05-11 | Advanced Bionics Corporation | Implantable stimulator system and method for treatment of incontinence and pain |
| EP1100402A4 (fr) | 1998-07-06 | 2008-12-10 | Boston Scient Neuromodulation | Systeme de stimulation implantable et procede de traitement de l'incontinence urinaire |
| US6941171B2 (en) * | 1998-07-06 | 2005-09-06 | Advanced Bionics Corporation | Implantable stimulator methods for treatment of incontinence and pain |
| US6002964A (en) | 1998-07-15 | 1999-12-14 | Feler; Claudio A. | Epidural nerve root stimulation |
| IL127481A (en) | 1998-10-06 | 2004-05-12 | Bio Control Medical Ltd | Urine excretion prevention device |
| ATE328548T1 (de) * | 1998-10-06 | 2006-06-15 | Bio Control Medical Ltd | Kontrolle von drängender inkontinenz |
| US6366814B1 (en) | 1998-10-26 | 2002-04-02 | Birinder R. Boveja | External stimulator for adjunct (add-on) treatment for neurological, neuropsychiatric, and urological disorders |
| AU6465399A (en) | 1998-10-30 | 2000-05-22 | Aalborg Universitet | A method to control an overactive bladder |
| US6341236B1 (en) | 1999-04-30 | 2002-01-22 | Ivan Osorio | Vagal nerve stimulation techniques for treatment of epileptic seizures |
| AUPQ202699A0 (en) | 1999-08-04 | 1999-08-26 | University Of Melbourne, The | Prosthetic device for incontinence |
| US20030028232A1 (en) | 2000-01-20 | 2003-02-06 | Medtronic, Inc. | Method of lmplanting a medical electrical lead |
| US6650943B1 (en) | 2000-04-07 | 2003-11-18 | Advanced Bionics Corporation | Fully implantable neurostimulator for cavernous nerve stimulation as a therapy for erectile dysfunction and other sexual dysfunction |
| US6558297B1 (en) * | 2000-07-28 | 2003-05-06 | Carnegie Mellon University | Energy storage device used in locomotion machine |
| US7054689B1 (en) | 2000-08-18 | 2006-05-30 | Advanced Bionics Corporation | Fully implantable neurostimulator for autonomic nerve fiber stimulation as a therapy for urinary and bowel dysfunction |
| WO2002020086A1 (fr) | 2000-09-07 | 2002-03-14 | Alfred E. Mann Institute For Biomedical Engineering At The University Of Southern California | Methode et appareil de controle de l'activite intestinale |
| US6612977B2 (en) | 2001-01-23 | 2003-09-02 | American Medical Systems Inc. | Sling delivery system and method of use |
| US20020161382A1 (en) | 2001-03-29 | 2002-10-31 | Neisz Johann J. | Implant inserted without bone anchors |
| US6641525B2 (en) | 2001-01-23 | 2003-11-04 | Ams Research Corporation | Sling assembly with secure and convenient attachment |
| EP1416903A2 (fr) * | 2001-07-20 | 2004-05-12 | Alfred E. Mann Institute for Biomedical Engineering at the University of Southern California | Procede et appareil de traitement de dysfonctionnements du tractus urinaire |
| US7407480B2 (en) | 2001-07-27 | 2008-08-05 | Ams Research Corporation | Method and apparatus for correction of urinary and gynecological pathologies, including treatment of incontinence cystocele |
| US6600956B2 (en) | 2001-08-21 | 2003-07-29 | Cyberonics, Inc. | Circumneural electrode assembly |
| US6862480B2 (en) | 2001-11-29 | 2005-03-01 | Biocontrol Medical Ltd. | Pelvic disorder treatment device |
| WO2004047914A1 (fr) | 2001-11-29 | 2004-06-10 | Biocontrol Medical Ltd. | Dispositif de traitement de troubles pelviens |
| US6911003B2 (en) | 2002-03-07 | 2005-06-28 | Ams Research Corporation | Transobturator surgical articles and methods |
| US7860570B2 (en) * | 2002-06-20 | 2010-12-28 | Boston Scientific Neuromodulation Corporation | Implantable microstimulators and methods for unidirectional propagation of action potentials |
| US7292890B2 (en) * | 2002-06-20 | 2007-11-06 | Advanced Bionics Corporation | Vagus nerve stimulation via unidirectional propagation of action potentials |
| US20040015205A1 (en) | 2002-06-20 | 2004-01-22 | Whitehurst Todd K. | Implantable microstimulators with programmable multielectrode configuration and uses thereof |
| US7203548B2 (en) * | 2002-06-20 | 2007-04-10 | Advanced Bionics Corporation | Cavernous nerve stimulation via unidirectional propagation of action potentials |
| CA2491018C (fr) * | 2002-06-28 | 2013-06-18 | Advanced Bionics Corporation | Microstimulateur dote d'une source d'alimentation autonome et systeme de telemetrie bidirectionnelle |
| US7328068B2 (en) * | 2003-03-31 | 2008-02-05 | Medtronic, Inc. | Method, system and device for treating disorders of the pelvic floor by means of electrical stimulation of the pudendal and associated nerves, and the optional delivery of drugs in association therewith |
| AU2005254105B2 (en) | 2004-06-14 | 2012-01-12 | Boston Scientific Limited | Systems, methods and devices relating to implantable supportive slings |
| EP1991133B1 (fr) | 2006-02-16 | 2018-10-24 | Boston Scientific Scimed, Inc. | Articles chirurgicaux pour traiter des affections pelviennes |
| US8195296B2 (en) | 2006-03-03 | 2012-06-05 | Ams Research Corporation | Apparatus for treating stress and urge incontinence |
| CN101460218B (zh) | 2006-06-05 | 2011-10-19 | Ams研究公司 | 治疗粪便失禁和/或盆腔内器官脱垂的装置 |
-
1999
- 1999-10-05 AT AT99947824T patent/ATE328548T1/de not_active IP Right Cessation
- 1999-10-05 EP EP99947825A patent/EP1124502B1/fr not_active Expired - Lifetime
- 1999-10-05 DE DE69931797T patent/DE69931797T2/de not_active Expired - Lifetime
- 1999-10-05 DE DE69935727T patent/DE69935727T2/de not_active Expired - Lifetime
- 1999-10-05 AU AU61186/99A patent/AU6118699A/en not_active Abandoned
- 1999-10-05 AU AU61187/99A patent/AU6118799A/en not_active Abandoned
- 1999-10-05 EP EP99947824A patent/EP1119314B1/fr not_active Expired - Lifetime
- 1999-10-05 US US09/806,970 patent/US6652449B1/en not_active Expired - Lifetime
- 1999-10-05 WO PCT/IL1999/000528 patent/WO2000019939A1/fr active IP Right Grant
- 1999-10-05 AT AT99947825T patent/ATE358454T1/de not_active IP Right Cessation
- 1999-10-06 US US09/413,272 patent/US6354991B1/en not_active Expired - Fee Related
-
2003
- 2003-09-24 US US10/671,144 patent/US7582053B2/en not_active Expired - Fee Related
-
2009
- 2009-06-17 US US12/486,492 patent/US8083663B2/en not_active Expired - Fee Related
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9457186B2 (en) | 2010-11-15 | 2016-10-04 | Bluewind Medical Ltd. | Bilateral feedback |
Also Published As
| Publication number | Publication date |
|---|---|
| AU6118799A (en) | 2000-04-26 |
| US8083663B2 (en) | 2011-12-27 |
| US20050261746A1 (en) | 2005-11-24 |
| EP1124502A1 (fr) | 2001-08-22 |
| EP1119314B1 (fr) | 2006-06-07 |
| EP1119314A4 (fr) | 2004-06-09 |
| US20090254145A1 (en) | 2009-10-08 |
| DE69931797D1 (de) | 2006-07-20 |
| EP1124502A4 (fr) | 2005-07-20 |
| ATE328548T1 (de) | 2006-06-15 |
| AU6118699A (en) | 2000-04-26 |
| EP1119314A1 (fr) | 2001-08-01 |
| DE69935727D1 (de) | 2007-05-16 |
| US6354991B1 (en) | 2002-03-12 |
| DE69931797T2 (de) | 2007-05-24 |
| US6652449B1 (en) | 2003-11-25 |
| WO2000019939A1 (fr) | 2000-04-13 |
| US7582053B2 (en) | 2009-09-01 |
| ATE358454T1 (de) | 2007-04-15 |
| DE69935727T2 (de) | 2007-12-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1124502B1 (fr) | Dispositif de traitement de l'incontinence | |
| US6896651B2 (en) | Mechanical and electrical sensing for incontinence treatment | |
| US7613516B2 (en) | Pelvic disorder treatment device | |
| US6862480B2 (en) | Pelvic disorder treatment device | |
| EP2032203B1 (fr) | Stimulation electrique des muscles pour le traitement de l'incontinence fecale et/ou de l'aptose pelvienne | |
| EP2046444B1 (fr) | Maille conductrice pour neurostimulation | |
| EP1981583B1 (fr) | Système de stimulation sélective de différentes parties du corps | |
| AU2007211151B2 (en) | System and method for selectively stimulating different body parts |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20010504 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: 7A 61N 1/36 B Ipc: 7A 61F 2/00 A |
|
| A4 | Supplementary search report drawn up and despatched |
Effective date: 20050608 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070404 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REF | Corresponds to: |
Ref document number: 69935727 Country of ref document: DE Date of ref document: 20070516 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070704 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070715 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: VOSSIUS & PARTNER |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070404 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070404 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070404 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PCAR Free format text: VOSSIUS & PARTNER;NADELBERG 3;4051 BASEL (CH) |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20080107 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070404 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070705 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20071031 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20071005 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070404 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 732E Free format text: REGISTERED BETWEEN 20090716 AND 20090722 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20071005 |
|
| NLS | Nl: assignments of ep-patents |
Owner name: AMS RESEARCH CORPORATION Effective date: 20090707 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PUE Owner name: AMS RESEARCH CORPORATION Free format text: BIO CONTROL MEDICAL, LTD.#HAHORESH STREET 4#56100 YAHUD (IL) -TRANSFER TO- AMS RESEARCH CORPORATION#10700 BREN ROAD WEST#MINNETONKA, MN 55343 (US) |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: TP |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20070404 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20110923 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20120924 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20120918 Year of fee payment: 14 Ref country code: FR Payment date: 20121115 Year of fee payment: 14 Ref country code: DE Payment date: 20120924 Year of fee payment: 14 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: V1 Effective date: 20140501 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20131005 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131031 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131005 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131031 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 69935727 Country of ref document: DE Effective date: 20140501 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20140630 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20131031 Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140501 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20140501 |