EP0216280A2 - Verwendung von Alkenylbernsteinsäurehalbamiden als Korrosionsschutzmittel - Google Patents
Verwendung von Alkenylbernsteinsäurehalbamiden als Korrosionsschutzmittel Download PDFInfo
- Publication number
- EP0216280A2 EP0216280A2 EP86112680A EP86112680A EP0216280A2 EP 0216280 A2 EP0216280 A2 EP 0216280A2 EP 86112680 A EP86112680 A EP 86112680A EP 86112680 A EP86112680 A EP 86112680A EP 0216280 A2 EP0216280 A2 EP 0216280A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- alkenylsuccinic
- alkyl
- corrosion inhibitors
- monoamides
- mol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23F—NON-MECHANICAL REMOVAL OF METALLIC MATERIAL FROM SURFACE; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL; MULTI-STEP PROCESSES FOR SURFACE TREATMENT OF METALLIC MATERIAL INVOLVING AT LEAST ONE PROCESS PROVIDED FOR IN CLASS C23 AND AT LEAST ONE PROCESS COVERED BY SUBCLASS C21D OR C22F OR CLASS C25
- C23F11/00—Inhibiting corrosion of metallic material by applying inhibitors to the surface in danger of corrosion or adding them to the corrosive agent
- C23F11/08—Inhibiting corrosion of metallic material by applying inhibitors to the surface in danger of corrosion or adding them to the corrosive agent in other liquids
- C23F11/10—Inhibiting corrosion of metallic material by applying inhibitors to the surface in danger of corrosion or adding them to the corrosive agent in other liquids using organic inhibitors
- C23F11/14—Nitrogen-containing compounds
- C23F11/145—Amides; N-substituted amides
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M133/00—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing nitrogen
- C10M133/02—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing nitrogen having a carbon chain of less than 30 atoms
- C10M133/16—Amides; Imides
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M173/00—Lubricating compositions containing more than 10% water
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2201/00—Inorganic compounds or elements as ingredients in lubricant compositions
- C10M2201/02—Water
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant Compositions
- C10M2215/08—Amides [having hydrocarbon substituents containing less than thirty carbon atoms]
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant Compositions
- C10M2215/08—Amides [having hydrocarbon substituents containing less than thirty carbon atoms]
- C10M2215/082—Amides [having hydrocarbon substituents containing less than thirty carbon atoms] containing hydroxyl groups; Alkoxylated derivatives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant Compositions
- C10M2215/086—Imides [having hydrocarbon substituents containing less than thirty carbon atoms]
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant Compositions
- C10M2215/12—Partial amides of polycarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant Compositions
- C10M2215/12—Partial amides of polycarboxylic acids
- C10M2215/122—Phtalamic acid
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2010/00—Metal present as such or in compounds
- C10N2010/02—Groups 1 or 11
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/12—Inhibition of corrosion, e.g. anti-rust agents or anti-corrosives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/20—Metal working
- C10N2040/22—Metal working with essential removal of material, e.g. cutting, grinding or drilling
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/20—Metal working
- C10N2040/24—Metal working without essential removal of material, e.g. forming, gorging, drawing, pressing, stamping, rolling or extruding; Punching metal
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/20—Metal working
- C10N2040/241—Manufacturing joint-less pipes
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/20—Metal working
- C10N2040/242—Hot working
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/20—Metal working
- C10N2040/243—Cold working
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/20—Metal working
- C10N2040/244—Metal working of specific metals
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/20—Metal working
- C10N2040/244—Metal working of specific metals
- C10N2040/245—Soft metals, e.g. aluminum
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/20—Metal working
- C10N2040/244—Metal working of specific metals
- C10N2040/246—Iron or steel
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/20—Metal working
- C10N2040/244—Metal working of specific metals
- C10N2040/247—Stainless steel
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2050/00—Form in which the lubricant is applied to the material being lubricated
- C10N2050/01—Emulsions, colloids, or micelles
Definitions
- the alkenylsuccinic acid halamides are prepared in known catfish by heating an alkenylsuccinic anhydride with an amine of the formula NH 2 R 1 in an approximately equimolar ratio to about 70 to 90 ° C. for about 2 hours.
- the compounds obtained can be used directly, that is to say in the form of the free acid, as corrosion inhibitors in metalworking fluids.
- These salts can be prepared in a simple manner by neutralizing the alkenylsuccinic acid halamides, for example by neutralizing with sodium hydroxide solution, mono-, di- or triethanolamine.
- the alkenylsuccinic acid halamides described give clear or milky, emulsion-like liquids which retain their good properties over a longer period of time, since no creams or phase separations occur even after several days.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Lubricants (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Preventing Corrosion Or Incrustation Of Metals (AREA)
Abstract
Description
- Die Verwendung von Salzen der Alkenylbernsteinsäurehalbamide der Formeln
- Es wurde nun gefunden, daß diese Nachteile der bekannten Alkenylbernstelnsäurehalbamide vermieden werden können, wenn die Amidgruppe durch einen C12-C18-Alkylrest substituiert ist. Gegenstand der Erfindung Ist die Verwendung von Alkenylbernsteinsäurehalbamiden der Formeln
- Die Herstellung der Alkenylbernsteinsäurohalbamide erfolgt in bekannter Welse, indem man ein Alkenylbernsteinsäureanhydrid mit einem Amin der Formel NH2R1 im ungefähr äquimolaren Verhältnis ca. 2 Stunden auf 70 bis 90°C erhitzt. Die dabei erhaltenen Verbindungen können direkt, das heißt in Form der freien Säure als Korrosionsschutzmittel in Metallbearbeitungsflüssigkeiten eingesetzt werden. Bevorzugt ist aber die Verwendung dieser Alkenylbernsteinsäurehalbamide in Form ihrer Alkali- oder Alkanolaminsalze. Diese Salze lassen sich in einfacher Weise durch Neutralisation der Alkenylbernsteinsäurehalbamide herstellen, beispielsweise durch Neutralisation mit Natronlauge, Mono-, Di- oder Triethanolamin.
- Die oben beschriebenen Alkenylbernsteinsäurehalbamide können mit besonderem Vorteil als Korrosionsschutzmittel in wäßrigen und mineralölhaltigen Kühlschmiermitteln, insbesondere Bohr-, Schneid- und Walzflüssigkeiten eingesetzt werden. Zur Bereitung dieser Kühlschmiermittel werden die Reaktionsprodukte in die erforderliche Menge Wasser oder Mineralöl eingerührt. Die Anwendungskonzentration in den Bohr-, Schneid- und Walzflüssigkeiten beträgt im allgemeinen etwa 0,1 bis 10 Gew.-%, vorzugsweise 2 bis 5 Gew.-%. Erforderlichenfalls können auch noch weitere, für diesen Anwendungszweck bekannte Wirkstoffe zugegeben werden.
- Je nach der Menge an Mineralöl ergeben die beschriebenen Alkenylbernsteinsäurehalbamide klare oder milchige, emulsionsartige Flüssigkeiten, die über einen längeren Zeitraum ihre guten Gebrauchseigenschaften behalten, da auch nach mehreren Tagen keine Aufrahmungen oder Phasentrennungen auftreten.
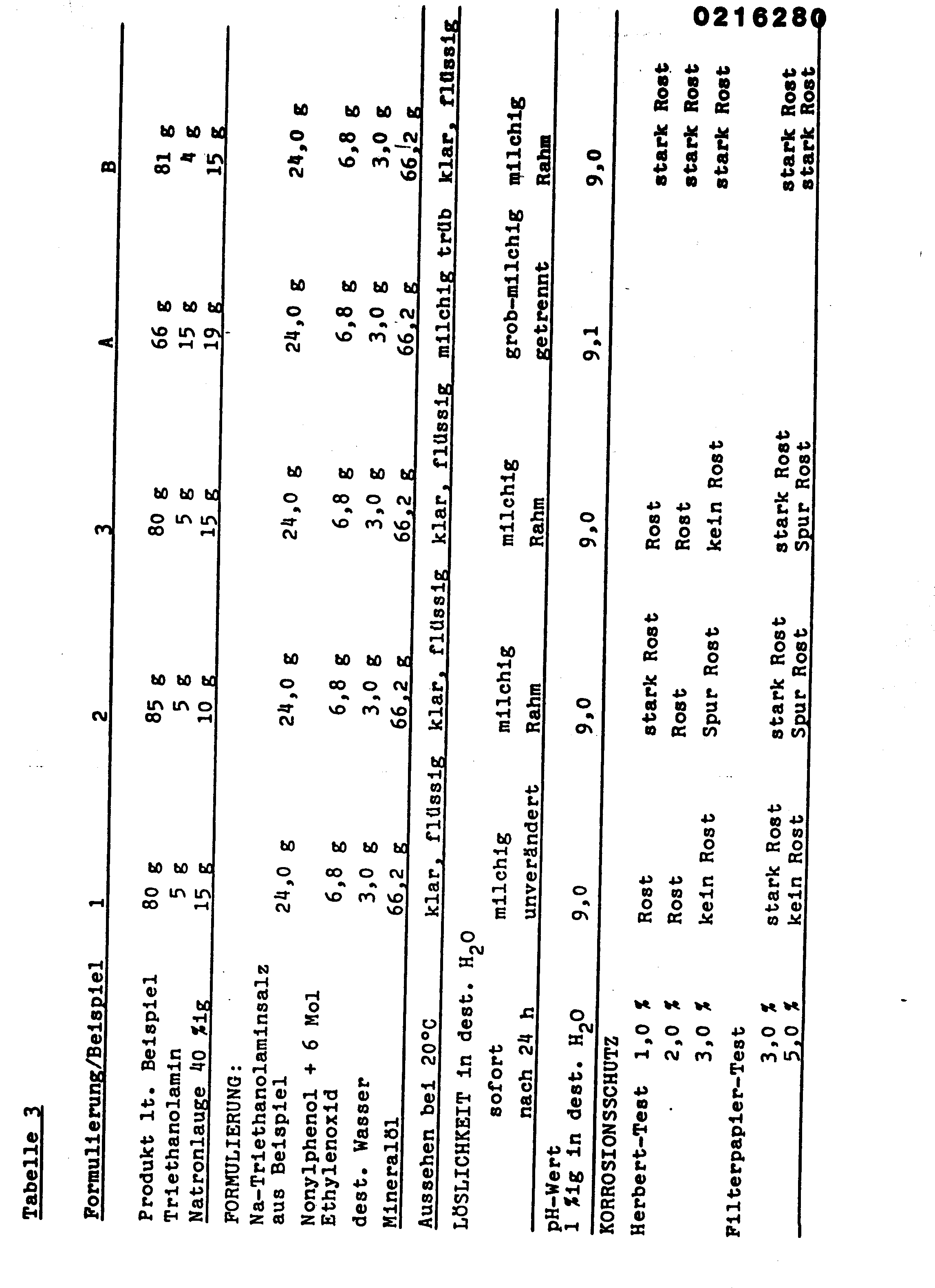
- Die folgenden Beispiele erläutern die Erfindung, die anschließenden Tabellen 1 bis 3 zeigen die hervorragenden Eigenschaften der Produkte gegenüber den Vergleichssubstanzen A und B.
- Beispiel 1
- In einem Reaktionskolben legt man 197 g (1 Mol) Cocosfett- 'amin vor und tropft unter Rühren 224 g (1 Mol) Tripropenyl bernsteinsäureanhydrid zu. Die Temperatur steigt dabei rasch an. Durch Kühlen mit einem Wasserbad hält man die Innentemperatur zwischen 70 und 90°C. Nach beendetem Zutropfen rührt man noch 90 Minuten bei 80°C, dann läßt man auf Raumtemperatur erkalten.
- Man erhält ca. 420 g einer viskosen Flüssigkeit mit einer Säurezahl von ca. 130 (Theorie: 133.3).
- Zu 185 g (1 Mol) Dodecylamin werden 266 g (1 Mol) Tetrapropenylbernsteinsäureanhydrid zugetropft und wie im Beispiel 1 verfahren.
- Man erhält ca. 450 g einer viskosen Flüssigkeit mit einer Säurezahl von ca. 125 (Theorie: 124,4)
- 196 g einer Mischung von Dodecylamin und Tetradecylamin (Mol-Verh. 75:25) werden bei Raumtemperatur vorgelegt, dazu werden unter Rühren 224 g (1 Mol) Tripropenylbernsteinsäureanhydrid zugetropft und weiter wie im Beispiel 1 verfahren. Man erhält ca. 420 g einer viskosen Flüssigkeit, die Säurezahl beträgt ca. 130 (Theorie: 133,6).
- 72 g (1 Mol) n-Butylamin werden vorgelegt und 224 g (1 Mol) Tripropenylbernsteinsäureanhydrid so zugetropft, daß die Innentemperatur 80°C nicht übersteigt. Nach beendetem Zutropfen rührt man noch 30 Minuten bei 80°C nach und läßt dann erkalten. Man erhält ca. 295 g einer viskosen Flüssigkeit, die Säurezahl beträgt ca. 190 (Theorie: 189,5).
- 250 (1 Mol) Di-octylamin werden bei Raumtemperatur vorgelegt. Dazu werden unter Rühren 224 g (1 Mol) Tripropenylbernsteinsäureanhydrid zugetropft, wobei die Temperatur auf 80°C steigt. Nach beendetem Zutropfen rührt man noch 2 1/2 Stunden bei 80°C und läßt dann erkalten.
-
Claims (1)
- Verwendung von Alkenylbernsteinsäurehalbamiden der Formeln
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE3534439 | 1985-09-27 | ||
| DE19853534439 DE3534439A1 (de) | 1985-09-27 | 1985-09-27 | Verwendung von alkenylbernsteinsaeurehalbamiden als korrosionsschutzmittel |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0216280A2 true EP0216280A2 (de) | 1987-04-01 |
| EP0216280A3 EP0216280A3 (en) | 1987-12-09 |
| EP0216280B1 EP0216280B1 (de) | 1995-12-27 |
Family
ID=6282082
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP86112680A Expired - Lifetime EP0216280B1 (de) | 1985-09-27 | 1986-09-13 | Verwendung von Alkenylbernsteinsäurehalbamiden als Korrosionsschutzmittel |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US4724124A (de) |
| EP (1) | EP0216280B1 (de) |
| JP (1) | JPS6274997A (de) |
| DE (2) | DE3534439A1 (de) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0359048A3 (de) * | 1988-09-03 | 1991-10-23 | Hoechst Aktiengesellschaft | Amindoamin-Salze von Alkenylbernsteinsäurederivaten, Verfahren zu deren Herstellung und deren Verwendung als Korrosionsinhibitoren |
| EP0464473A1 (de) * | 1990-06-23 | 1992-01-08 | Hoechst Aktiengesellschaft | Salze von Alkenylbernsteinsäurehalbamiden und deren Verwendung als Korrosionsschutzmittel und Emulgatoren für Metallbearbeitungsöle |
| EP0501368A1 (de) * | 1991-02-26 | 1992-09-02 | Hoechst Aktiengesellschaft | Verwendung von Alkenylbernsteinsäurehalbamiden |
| EP0566956A1 (de) * | 1992-04-22 | 1993-10-27 | Hoechst Aktiengesellschaft | Korrosionsschutzmittel |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5250225A (en) * | 1991-02-04 | 1993-10-05 | Basf Aktiengesellschaft | Ammonium salt of an alkenylsuccinic half-amide and the use thereof as corrosion inhibitor in oil and/or gas production technology |
| US5401428A (en) * | 1993-10-08 | 1995-03-28 | Monsanto Company | Water soluble metal working fluids |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2944969A (en) * | 1957-02-06 | 1960-07-12 | Petrolite Corp | Prevention of rust and corrosion |
| BE568954A (de) * | 1958-01-07 | |||
| US3269946A (en) * | 1961-08-30 | 1966-08-30 | Lubrizol Corp | Stable water-in-oil emulsions |
| US3324033A (en) * | 1966-03-29 | 1967-06-06 | Ethyl Corp | Ester-amides of alkenyl succinic anhydride and diethanolamine as ashless dispersants |
| US3903005A (en) * | 1973-11-05 | 1975-09-02 | Texaco Inc | Corrosion inhibited compositions |
| JPS52144883A (en) * | 1976-05-28 | 1977-12-02 | Sanyo Chemical Ind Ltd | Water soluble cutting oil agent |
| GB1532836A (en) * | 1976-09-09 | 1978-11-22 | Mobil Oil Corp | Lubricant compositions |
| US4289636A (en) * | 1979-10-01 | 1981-09-15 | Mobil Oil Corporation | Aqueous lubricant compositions |
| US4379063A (en) * | 1981-02-20 | 1983-04-05 | Cincinnati Milacron Inc. | Novel functional fluid |
| DE3300874A1 (de) * | 1983-01-13 | 1984-07-19 | Henkel KGaA, 4000 Düsseldorf | Bernsteinsaeurederivate als korrosionsschutzmittel |
| DE3319183A1 (de) * | 1983-05-27 | 1984-11-29 | Hoechst Ag, 6230 Frankfurt | Verwendung von alkenylbernsteinsaeurehalbamiden als korrosionsschutzmittel |
| DE3341013A1 (de) * | 1983-11-12 | 1985-05-23 | Henkel KGaA, 4000 Düsseldorf | Bernsteinsaeure-mono-dialkylamide als wasserloesliche korrosionsschutzmittel |
-
1985
- 1985-09-27 DE DE19853534439 patent/DE3534439A1/de not_active Withdrawn
-
1986
- 1986-09-13 EP EP86112680A patent/EP0216280B1/de not_active Expired - Lifetime
- 1986-09-13 DE DE3650459T patent/DE3650459D1/de not_active Expired - Fee Related
- 1986-09-25 JP JP61225074A patent/JPS6274997A/ja active Pending
- 1986-09-26 US US06/913,128 patent/US4724124A/en not_active Expired - Lifetime
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0359048A3 (de) * | 1988-09-03 | 1991-10-23 | Hoechst Aktiengesellschaft | Amindoamin-Salze von Alkenylbernsteinsäurederivaten, Verfahren zu deren Herstellung und deren Verwendung als Korrosionsinhibitoren |
| EP0464473A1 (de) * | 1990-06-23 | 1992-01-08 | Hoechst Aktiengesellschaft | Salze von Alkenylbernsteinsäurehalbamiden und deren Verwendung als Korrosionsschutzmittel und Emulgatoren für Metallbearbeitungsöle |
| EP0501368A1 (de) * | 1991-02-26 | 1992-09-02 | Hoechst Aktiengesellschaft | Verwendung von Alkenylbernsteinsäurehalbamiden |
| EP0566956A1 (de) * | 1992-04-22 | 1993-10-27 | Hoechst Aktiengesellschaft | Korrosionsschutzmittel |
Also Published As
| Publication number | Publication date |
|---|---|
| DE3534439A1 (de) | 1987-04-02 |
| EP0216280A3 (en) | 1987-12-09 |
| DE3650459D1 (de) | 1996-02-08 |
| EP0216280B1 (de) | 1995-12-27 |
| JPS6274997A (ja) | 1987-04-06 |
| US4724124A (en) | 1988-02-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0088724B1 (de) | Benzotriazolgemische, Verfahren zu deren Herstellung und ihre Verwendung als Metallpassivatoren | |
| EP0222311A2 (de) | Verwendung von Alkoxyhydroxyfettsäuren als Korrosionsinhibitoren in Ölen und Ölhaltigen Emulsionen | |
| EP0127132B1 (de) | Verwendung von Alkenylbernsteinsäurehalbamiden als Korrosionsschutzmittel | |
| EP0144663B1 (de) | Verwendung von Korrosionsinhibitoren in wässrigen Systemen | |
| DE2840112C2 (de) | Wassermischbare Korrosionsschutzmittel und Verfahren zur Verhinderung der Korrosion von Eisenmetallen | |
| DE2722819C2 (de) | Korrosionsinhibierendes Kühl- und Metallbearbeitungsmittel | |
| EP0216280B1 (de) | Verwendung von Alkenylbernsteinsäurehalbamiden als Korrosionsschutzmittel | |
| EP0584711B1 (de) | Alkenylbernsteinsäurederivate als Metallbearbeitungshilfsmittel | |
| EP0160042B1 (de) | Korrosionsinhibitoren für zink | |
| EP0452349B1 (de) | Verfahren zur herstellung von amphoteren grenzflächenaktiven imidazolinderivaten | |
| EP0332897B1 (de) | Reaktionsprodukte aus Borsäure und Alkanoletheraminen und deren Verwendung als Korrosionsschutzmittel | |
| EP0144738B1 (de) | Bernsteinsäure-mono-dialkylamide als wasserlösliche Korrosionsschutzmittel | |
| EP0163107B1 (de) | Verwendung von Benzoylalaninen als Korrosionsinhibitoren für wässrige Systeme | |
| EP0566956A1 (de) | Korrosionsschutzmittel | |
| EP1184486A2 (de) | Verwendung von N-Alkyl-beta-alanin-Derivaten zur Herstellung von reinigenden Korrsionsschutzmitteln | |
| EP0191952B1 (de) | Salze von Alkenylbernsteinsäurehalbamiden, Verfahren zu deren Herstellung und deren Verwendung als Korrosionsinhibitoren | |
| EP0116348B2 (de) | Reaktionsprodukte aus Borsäure, Diethanolamin und Monoethanolaminen und deren Verwendung als Korrosionsschutzmittel | |
| DE1240872B (de) | Verfahren zur Herstellung wasserloeslicher, kapillaraktiver, als Wasch- und Reinigungsmittel und als Schaumstabilisatoren verwendbarer Ampholyte | |
| EP0294687A1 (de) | Verwendung von Salzen von Sulfonamidocarbonsäuren als Korrosionsinhibitoren in wässrigen Systemen | |
| EP0726335B1 (de) | Lactobionsäureamid enthaltende Korrosionsschutzmittel | |
| EP0464473B1 (de) | Salze von Alkenylbernsteinsäurehalbamiden und deren Verwendung als Korrosionsschutzmittel und Emulgatoren für Metallbearbeitungsöle | |
| DE3223794C2 (de) | Addukte derO,O'-Dialkyldithiophosphorsäure mit einem substituierten 2-Oxazolin oder Imidazolin und deren Verwendung als Schmiermittelzusatz | |
| EP0501368B1 (de) | Verwendung von Alkenylbernsteinsäurehalbamiden | |
| DE1916628A1 (de) | Korrosionsinhibitor fuer wasserfuehrende Kuehl- und Heizungssysteme | |
| EP0109549A1 (de) | Korrosionsschutzmittel für wässrige Flüssigkeiten zur Bearbeitung von Metallen und Verfahren zu deren Herstellung |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): DE FR GB IT SE |
|
| XX | Miscellaneous (additional remarks) |
Free format text: EIN ANTRAG AUF BERICHTIGUNG DES ANSPRUCHES GEMAESS REGEL 88 EPU LIEGT VOR. UBER DIESEN ANTRAG WIRD IM LAUFENDES VERFAHRENS VON DER PRUEFUNGSABTEILUNG EINE ENTSCHEIDUNG GETROFFEN WERDEN. |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): DE FR GB IT SE |
|
| 17P | Request for examination filed |
Effective date: 19880526 |
|
| 17Q | First examination report despatched |
Effective date: 19891117 |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): DE FR IT SE |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| XX | Miscellaneous (additional remarks) | ||
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): DE FR IT SE |
|
| REF | Corresponds to: |
Ref document number: 3650459 Country of ref document: DE Date of ref document: 19960208 |
|
| ITF | It: translation for a ep patent filed | ||
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 19980709 Year of fee payment: 13 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 19990929 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: TP |
|
| EUG | Se: european patent has lapsed |
Ref document number: 86112680.3 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20030811 Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20030813 Year of fee payment: 18 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050401 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050531 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20050913 |
|
| APAH | Appeal reference modified |
Free format text: ORIGINAL CODE: EPIDOSCREFNO |