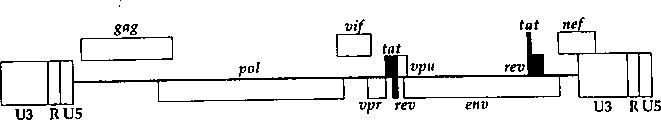
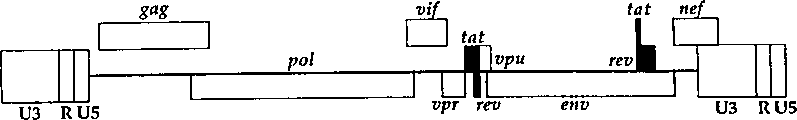
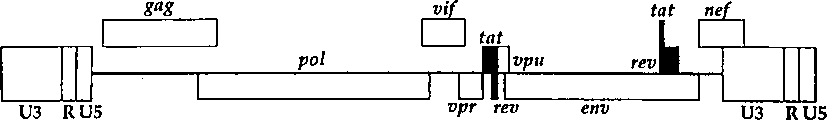
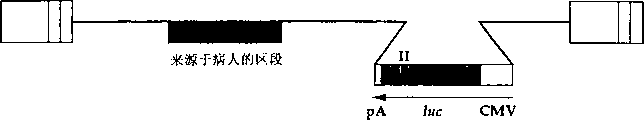
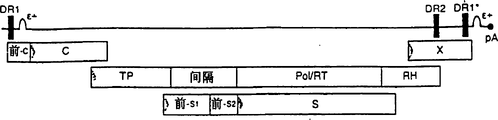
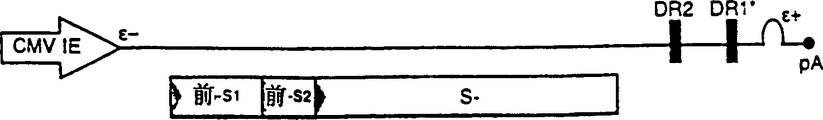
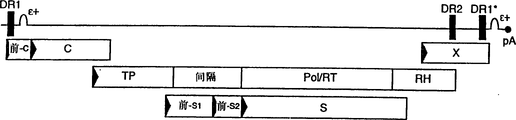
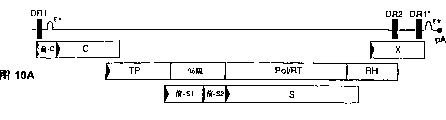
CN1263867C - 用于确定抗病毒药物敏感性和抗性和筛选抗病毒药物的组合物和方法 - Google Patents
用于确定抗病毒药物敏感性和抗性和筛选抗病毒药物的组合物和方法 Download PDFInfo
- Publication number
- CN1263867C CN1263867C CNB971929327A CN97192932A CN1263867C CN 1263867 C CN1263867 C CN 1263867C CN B971929327 A CNB971929327 A CN B971929327A CN 97192932 A CN97192932 A CN 97192932A CN 1263867 C CN1263867 C CN 1263867C
- Authority
- CN
- China
- Prior art keywords
- resistance test
- patient
- host cell
- gene
- viral
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/502—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics for testing non-proliferative effects
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/02—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving viable microorganisms
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/64—General methods for preparing the vector, for introducing it into the cell or for selecting the vector-containing host
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6897—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids involving reporter genes operably linked to promoters
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/5044—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics involving specific cell types
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/5044—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics involving specific cell types
- G01N33/5047—Cells of the immune system
- G01N33/505—Cells of the immune system involving T-cells
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/5044—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics involving specific cell types
- G01N33/5067—Liver cells
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2800/00—Detection or diagnosis of diseases
- G01N2800/52—Predicting or monitoring the response to treatment, e.g. for selection of therapy based on assay results in personalised medicine; Prognosis
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Biomedical Technology (AREA)
- Immunology (AREA)
- Molecular Biology (AREA)
- Biotechnology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Cell Biology (AREA)
- Hematology (AREA)
- Organic Chemistry (AREA)
- Urology & Nephrology (AREA)
- Biochemistry (AREA)
- Microbiology (AREA)
- General Health & Medical Sciences (AREA)
- Physics & Mathematics (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Analytical Chemistry (AREA)
- General Engineering & Computer Science (AREA)
- Tropical Medicine & Parasitology (AREA)
- Pathology (AREA)
- Food Science & Technology (AREA)
- General Physics & Mathematics (AREA)
- Medicinal Chemistry (AREA)
- Toxicology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biophysics (AREA)
- Plant Pathology (AREA)
- Gastroenterology & Hepatology (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Investigating Or Analysing Biological Materials (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US59300996A | 1996-01-29 | 1996-01-29 | |
| US08/593,009 | 1996-01-29 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1213407A CN1213407A (zh) | 1999-04-07 |
| CN1263867C true CN1263867C (zh) | 2006-07-12 |
Family
ID=24372979
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB971929327A Expired - Fee Related CN1263867C (zh) | 1996-01-29 | 1997-01-29 | 用于确定抗病毒药物敏感性和抗性和筛选抗病毒药物的组合物和方法 |
Country Status (18)
| Country | Link |
|---|---|
| EP (2) | EP0852626B1 (enExample) |
| JP (1) | JP4183749B2 (enExample) |
| KR (1) | KR100537153B1 (enExample) |
| CN (1) | CN1263867C (enExample) |
| AP (1) | AP9801360A0 (enExample) |
| AT (1) | ATE447621T1 (enExample) |
| AU (1) | AU732255B2 (enExample) |
| CA (1) | CA2216126C (enExample) |
| DE (2) | DE69711584T2 (enExample) |
| EA (1) | EA005426B1 (enExample) |
| ES (1) | ES2175355T3 (enExample) |
| HU (1) | HUP9900388A3 (enExample) |
| IL (1) | IL125464A (enExample) |
| NO (1) | NO983421L (enExample) |
| NZ (1) | NZ331376A (enExample) |
| PL (1) | PL328068A1 (enExample) |
| RO (1) | RO118887B1 (enExample) |
| WO (1) | WO1997027319A1 (enExample) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109722465A (zh) * | 2019-01-07 | 2019-05-07 | 福建省疾病预防控制中心(福建省健康教育促进中心、福建省卫生检验检测中心) | 一种hiv耐药检测载体和构建方法 |
Families Citing this family (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2123065T3 (es) | 1992-08-25 | 1999-01-01 | Searle & Co | Hidroxietilamino-sulfonamidas utiles como inhibidores de proteasas retroviricas. |
| KR100495690B1 (ko) | 1996-01-26 | 2005-11-08 | 비르코 비브이비에이 | 인체 hiv 계통의 표현형적 약제 감수성에 기초한 하나 이상의 최적한 hiv 저해제를 선발하는 방법 |
| US6242187B1 (en) | 1996-01-29 | 2001-06-05 | Virologic, Inc. | Compositions and methods for determining anti-viral drug susceptibility and resistance and anti-viral drug screening |
| CA2298102A1 (en) * | 1997-07-30 | 1999-02-11 | Virologic, Inc. | Compositions and methods for determining anti-viral drug susceptibility and resistance and anti-viral drug screening |
| WO1999051776A1 (en) * | 1998-04-07 | 1999-10-14 | Southern Research Institute | High-throughput method for screening for antiviral activity |
| US7037644B1 (en) | 1998-05-26 | 2006-05-02 | Virologic, Inc. | Means and methods for monitoring non-nucleoside reverse transcriptase inhibitor antiretroviral therapy and guiding therapeutic decisions in the treatment of HIV/AIDS |
| NZ508834A (en) * | 1998-05-26 | 2004-06-25 | Virologic Inc | Means and methods for monitoring non-nucleoside reverse transcriptase inhibitor antiretroviral therapy |
| US20030148307A1 (en) | 1998-06-19 | 2003-08-07 | Department Of Health & Human Services, C/O Centers Of Disease Control And Prevention | Method and kit for detecting resistance to antiviral drugs |
| US6787126B1 (en) | 1998-06-19 | 2004-09-07 | The United States Of America As Represented By The Department Of Health And Human Services | Method and kit for detecting resistance to antiviral drugs |
| CA2331260C (en) * | 1998-06-19 | 2008-03-18 | The Government Of The United States Of America As Represented By The Secretary Of The Department Of Health And Human Services | Method and kit for detecting resistance to antiviral drugs |
| BR9912209A (pt) * | 1998-06-24 | 2002-11-05 | Virologic Inc | Recursos e métodos para monitoração de terapia anti-retroviral inibidora de transcriptase reversa de nucleosìdeo e guia de decisões terapêuticas no tratamento de hiv/aids |
| ES2246859T3 (es) | 1999-05-28 | 2006-03-01 | Virco Bvba | Nuevos perfiles mutacionales en la transcriptasa inversa del vih-1 correlacionadas con una resistencia fenotipica a farmacos. |
| EP1326640A4 (en) * | 2000-09-15 | 2004-12-15 | Virologic Inc | NON-NUCLEOSIDIC REVERSE-TRANSCRIPTASE INHIBITOR AS ANTIRETROVIRAL THERAPY |
| EP1356082A2 (en) | 2000-10-20 | 2003-10-29 | Virco Bvba | Mutational profiles in hiv-1 reverse transcriptase correlated with phenotypic drug resistance |
| JP4264256B2 (ja) * | 2000-10-20 | 2009-05-13 | ビルコ・ビーブイビーエイ | 治療に対する耐性を予測するための生物学的カット−オフ値の確立 |
| US7247439B1 (en) | 2001-06-04 | 2007-07-24 | Monogram Biosciences, Inc. | Method of evaluating a patient antibody response to envelope proteins of a virus |
| US7097970B2 (en) | 2001-06-04 | 2006-08-29 | Monogram Biosciences, Inc. | Methods of evaluating viral entry inhibitors using patient derived envelope protein constructs |
| WO2003040390A2 (en) | 2001-11-08 | 2003-05-15 | Tibotec Pharmaceuticals Ltd. | Protease assay for therapeutic drug monitoring |
| EP1520247B1 (en) | 2002-07-01 | 2009-05-27 | Tibotec Pharmaceuticals Ltd. | Mutational profiles in hiv-1 protease correlated with phenotypic drug resistance |
| DE60321466D1 (de) | 2002-07-01 | 2008-07-17 | Tibotec Pharm Ltd | Mit phänotypischer medikamentenresistenz korrelierte mutationsprofile in hiv-1-reverse-transkriptase |
| DE20218008U1 (de) | 2002-11-21 | 2003-03-06 | FESTO AG & Co., 73734 Esslingen | Anordnung von mehreren Handhabungsvorrichtungen |
| US8603736B2 (en) | 2004-06-07 | 2013-12-10 | Monogram Biosciences, Inc. | Compositions and methods for determining resistance to inhibitors of virus entry using recombinant virus assays |
| US8178291B2 (en) | 2005-02-18 | 2012-05-15 | Monogram Biosciences, Inc. | Methods and compositions for determining hypersusceptibility of HIV-1 to non-nucleoside reverse transcriptase inhibitors |
| EP2522757A1 (en) | 2005-02-18 | 2012-11-14 | Monogram BioSciences, Inc. | Methods and compositions for determining anti-HIV drug susceptibility and replication capacity of HIV |
| CA2609910A1 (en) | 2005-05-27 | 2006-12-07 | Monogram Biosciences, Inc. | Methods and compositions for determining resistance of hiv-1 to protease inhibitors |
| WO2006133267A2 (en) | 2005-06-06 | 2006-12-14 | Monogram Biosciences, Inc. | Methods and compositions for determining altered susceptibility of hiv-1 to anti-hiv drugs |
| WO2006133266A2 (en) | 2005-06-06 | 2006-12-14 | Monogram Biosciences, Inc. | Methods for determining resistance or susceptibility to hiv entry inhibitors |
| WO2007146336A2 (en) | 2006-06-13 | 2007-12-21 | Monogram Biosciences, Inc. | Molecular determinants associated with enhanced ability to enter cells expressing cxcr4 |
| EP2126137B1 (en) | 2007-01-23 | 2017-06-07 | Janssen Pharmaceutica NV | Method for designing a drug regime for hiv-infected patients |
| WO2008106115A2 (en) | 2007-02-26 | 2008-09-04 | Monogram Biosciences, Inc. | Compositions and methods for determining whether a subject would benefit from co-receptor inhibitor therapy |
| JPWO2018030534A1 (ja) * | 2016-08-10 | 2019-06-13 | 国立研究開発法人理化学研究所 | B型肝炎治療用組成物、及びb型肝炎ウイルスの複製活性の評価方法 |
| RU2646472C1 (ru) * | 2016-10-07 | 2018-03-05 | Федеральное государственное бюджетное образовательное учреждение высшего образования "Пермский государственный медицинский университет имени академика Е.А. Вагнера" Министерства здравоохранения Российской Федерации | Способ оценки эффективности терапии инфекции вызванной вирусом герпеса человека 6 типа у детей |
| CN110382009A (zh) * | 2016-12-29 | 2019-10-25 | 富荣吉有限责任公司 | Retro-HBV和Lenti-HBV的混合载体及构建体 |
| CN113832169B (zh) * | 2020-06-24 | 2025-10-14 | 厦门大学 | 靶向HBV cccDNA的药物筛选模型及方法 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0291893A1 (en) * | 1987-05-19 | 1988-11-23 | The Du Pont Merck Pharmaceutical Company | Stable human cell lines expressing an indicator gene product under virus-specific genetic controls |
| ATE118247T1 (de) * | 1988-09-27 | 1995-02-15 | Dana Farber Cancer Inst Inc | Vektor, der ein replikationskompetentes hiv-i- provirus und ein heterologes gen enthält. |
| HU209835B (en) * | 1988-12-07 | 1994-11-28 | Univ Osaka Res Found | Method for producing of retrovirus protease, reverse transcriptase and integrase |
| ATE161041T1 (de) * | 1989-08-25 | 1997-12-15 | Chiron Corp | Verfahren zur hcv-züchtung in zell-linien aus b- oder t-lymphozyten |
| GB8923123D0 (en) * | 1989-10-13 | 1989-11-29 | Connaught Lab | A vaccine for human immunodeficiency virus |
| FR2700169B1 (fr) * | 1993-01-04 | 1995-03-24 | Transgene Sa | Nouveaux variants trans-dominants TAT du virus de l'immunodéficience humaine. |
| US5645982A (en) * | 1993-08-19 | 1997-07-08 | Systemix, Inc. | Method for screening potential therapeutically effective antiviral agents |
| WO1995022622A1 (en) * | 1994-02-18 | 1995-08-24 | University Of Washington | Methods and compositions for screening for anti-aids drugs |
| US5733543A (en) * | 1994-04-29 | 1998-03-31 | Nabel; Gary J. | Introduction of HIV-protective genes into cells by particle-mediated gene transfer |
| DE69535669T2 (de) * | 1994-05-09 | 2008-12-04 | Oxford Biomedica (Uk) Ltd. | Retrovirale vektoren mit verminderter rekombinationsrate |
| US5837464A (en) * | 1996-01-29 | 1998-11-17 | Virologic, Inc. | Compositions and methods for determining anti-viral drug susceptibility and resistance and anti-viral drug screening |
| CA2298102A1 (en) * | 1997-07-30 | 1999-02-11 | Virologic, Inc. | Compositions and methods for determining anti-viral drug susceptibility and resistance and anti-viral drug screening |
-
1997
- 1997-01-29 EP EP97907549A patent/EP0852626B1/en not_active Expired - Lifetime
- 1997-01-29 EA EA199800669A patent/EA005426B1/ru not_active IP Right Cessation
- 1997-01-29 DE DE69711584T patent/DE69711584T2/de not_active Expired - Lifetime
- 1997-01-29 AP APAP/P/1998/001360A patent/AP9801360A0/en unknown
- 1997-01-29 CN CNB971929327A patent/CN1263867C/zh not_active Expired - Fee Related
- 1997-01-29 EP EP01117002A patent/EP1170380B1/en not_active Expired - Lifetime
- 1997-01-29 AT AT01117002T patent/ATE447621T1/de not_active IP Right Cessation
- 1997-01-29 KR KR1019980705851A patent/KR100537153B1/ko not_active Expired - Fee Related
- 1997-01-29 WO PCT/US1997/001609 patent/WO1997027319A1/en not_active Ceased
- 1997-01-29 NZ NZ331376A patent/NZ331376A/xx not_active IP Right Cessation
- 1997-01-29 JP JP52712397A patent/JP4183749B2/ja not_active Expired - Fee Related
- 1997-01-29 DE DE69739645T patent/DE69739645D1/de not_active Expired - Lifetime
- 1997-01-29 PL PL97328068A patent/PL328068A1/xx not_active IP Right Cessation
- 1997-01-29 CA CA002216126A patent/CA2216126C/en not_active Expired - Lifetime
- 1997-01-29 RO RO98-01216A patent/RO118887B1/ro unknown
- 1997-01-29 AU AU19528/97A patent/AU732255B2/en not_active Ceased
- 1997-01-29 ES ES97907549T patent/ES2175355T3/es not_active Expired - Lifetime
- 1997-01-29 HU HU9900388A patent/HUP9900388A3/hu unknown
- 1997-01-29 IL IL125464A patent/IL125464A/en not_active IP Right Cessation
-
1998
- 1998-07-24 NO NO983421A patent/NO983421L/no unknown
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109722465A (zh) * | 2019-01-07 | 2019-05-07 | 福建省疾病预防控制中心(福建省健康教育促进中心、福建省卫生检验检测中心) | 一种hiv耐药检测载体和构建方法 |
| CN109722465B (zh) * | 2019-01-07 | 2022-02-01 | 福建省疾病预防控制中心(福建省健康教育促进中心、福建省卫生检验检测中心) | 一种hiv耐药检测载体和构建方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| IL125464A (en) | 2007-12-03 |
| NO983421D0 (no) | 1998-07-24 |
| EP0852626B1 (en) | 2002-04-03 |
| AU732255B2 (en) | 2001-04-12 |
| NO983421L (no) | 1998-09-25 |
| DE69711584T2 (de) | 2002-11-07 |
| AU1952897A (en) | 1997-08-20 |
| EP0852626A1 (en) | 1998-07-15 |
| CA2216126A1 (en) | 1997-07-31 |
| EA199800669A1 (ru) | 1999-02-25 |
| WO1997027319A1 (en) | 1997-07-31 |
| EP0852626A4 (en) | 1999-08-25 |
| CA2216126C (en) | 2009-03-31 |
| JP4183749B2 (ja) | 2008-11-19 |
| EA005426B1 (ru) | 2005-02-24 |
| ES2175355T3 (es) | 2002-11-16 |
| AP9801360A0 (en) | 1998-12-31 |
| CN1213407A (zh) | 1999-04-07 |
| NZ331376A (en) | 2000-03-27 |
| PL328068A1 (en) | 1999-01-04 |
| KR100537153B1 (ko) | 2006-12-15 |
| DE69711584D1 (de) | 2002-05-08 |
| HUP9900388A2 (hu) | 1999-05-28 |
| ATE447621T1 (de) | 2009-11-15 |
| KR19990082129A (ko) | 1999-11-15 |
| JP2000503849A (ja) | 2000-04-04 |
| EP1170380B1 (en) | 2009-11-04 |
| DE69739645D1 (de) | 2009-12-17 |
| RO118887B1 (ro) | 2003-12-30 |
| EP1170380A2 (en) | 2002-01-09 |
| IL125464A0 (en) | 1999-03-12 |
| EP1170380A3 (en) | 2003-05-02 |
| HUP9900388A3 (en) | 2005-03-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1263867C (zh) | 用于确定抗病毒药物敏感性和抗性和筛选抗病毒药物的组合物和方法 | |
| US6242187B1 (en) | Compositions and methods for determining anti-viral drug susceptibility and resistance and anti-viral drug screening | |
| CN1452661A (zh) | 用于监测hiv药物抗性的病毒载体 | |
| CN1833024A (zh) | 含有衍生自基因型2a丙肝病毒(HCV)基因组的核酸的核酸构建体和具有转移入其中的这样的核酸构建体的细胞 | |
| CN1836042A (zh) | 用于基因沉默的hbv和hcv保守序列 | |
| CN1991365A (zh) | 根据人类hiv病毒株的表型药物敏感性控制hiv阳性病人化疗的方法 | |
| CN1117876C (zh) | 刺激促生长素抑制素和胰岛素产生的化合物的筛选方法 | |
| CN1942585A (zh) | 含人丙型肝炎病毒全长基因组的核酸构建物、核酸构建物转入其中的重组全长病毒基因组复制型细胞和生产丙型肝炎病毒颗粒的方法 | |
| CN1646706A (zh) | 应用重组病毒分析试验评价病毒受体/共受体用途以及病毒侵入抑制剂的组合物和方法 | |
| CN1646704A (zh) | 确定致病性病毒对蛋白酶抑制剂敏感性的组合物和方法 | |
| CN1451042A (zh) | 监测人免疫缺陷病毒药物抗性的方法 | |
| CN1125467A (zh) | 一种对白细胞介素-5受体依赖细胞信息传递通路有调整作用的物质筛选方法 | |
| CN1820081A (zh) | 分析基因导入位点的方法 | |
| CN1545550A (zh) | 含泡沫逆转录病毒包膜蛋白的感染性嗜肝dna病毒颗粒的制备及其使用方法 | |
| JP2001512036A (ja) | 抗ウイルス薬剤感受性および耐性を測定するための組成物および方法、並びに抗ウイルス薬剤をスクリーニングするための組成物および方法 | |
| CN1529758A (zh) | 新的表达载体及其应用 | |
| CN1886417A (zh) | Hiv-依赖型表现构筑体及其用途 | |
| HK1019077A (en) | Compositions and methods for determining anti-viral drug susceptibility and resistance and anti-viral drug screening | |
| CN1969048A (zh) | 研究hiv的遗传和功能变异性的方法以及用于该方法的试剂盒 | |
| CN1547618A (zh) | 利用重组病毒分析法评价病毒受体/辅助受体使用和病毒侵入抑制剂的组合物和方法 | |
| Yu et al. | An RNA-based system to study hepatitis B virus replication and select drug-resistance mutations | |
| CN1292069C (zh) | Hiv-1病毒tat-蛋白突变体 | |
| AU776535B2 (en) | Compositions and methods for determining anti-viral drug susceptibility and resistance and anti-viral drug screening | |
| CN1827768A (zh) | 增强有用基因表达的核酸序列和方法 | |
| CN1671729A (zh) | 人Corin基因的控制序列 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: WD Ref document number: 1019077 Country of ref document: HK |
|
| C19 | Lapse of patent right due to non-payment of the annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |