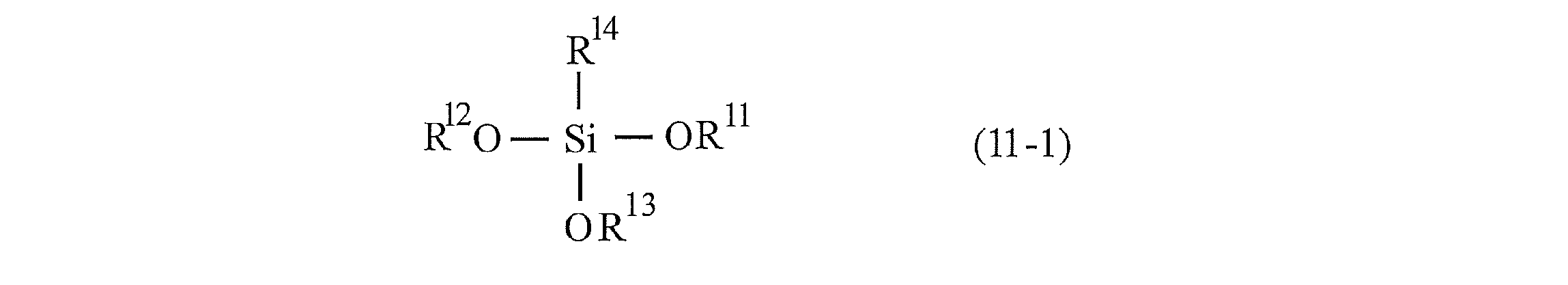WO2020246501A1 - 電池用電解液及びリチウムイオン電池 - Google Patents
電池用電解液及びリチウムイオン電池 Download PDFInfo
- Publication number
- WO2020246501A1 WO2020246501A1 PCT/JP2020/021926 JP2020021926W WO2020246501A1 WO 2020246501 A1 WO2020246501 A1 WO 2020246501A1 JP 2020021926 W JP2020021926 W JP 2020021926W WO 2020246501 A1 WO2020246501 A1 WO 2020246501A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- battery
- electrolytic solution
- chain
- particles
- Prior art date
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0568—Liquid materials characterised by the solutes
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B32/00—Carbon; Compounds thereof
- C01B32/15—Nano-sized carbon materials
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B32/00—Carbon; Compounds thereof
- C01B32/25—Diamond
- C01B32/28—After-treatment, e.g. purification, irradiation, separation or recovery
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0567—Liquid materials characterised by the additives
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0569—Liquid materials characterised by the solvents
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M6/00—Primary cells; Manufacture thereof
- H01M6/14—Cells with non-aqueous electrolyte
- H01M6/16—Cells with non-aqueous electrolyte with organic electrolyte
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/80—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70
- C01P2002/82—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70 by IR- or Raman-data
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/80—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70
- C01P2002/86—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70 by NMR- or ESR-data
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/60—Particles characterised by their size
- C01P2004/64—Nanometer sized, i.e. from 1-100 nanometer
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/002—Inorganic electrolyte
- H01M2300/0022—Room temperature molten salts
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0025—Organic electrolyte
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0025—Organic electrolyte
- H01M2300/0028—Organic electrolyte characterised by the solvent
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Definitions
- the present disclosure relates to an electrolytic solution for a battery and a lithium ion battery. More specifically, the present disclosure relates to an electrolytic solution for a battery in which a nanocarbon material is dispersed, and a lithium ion battery using the electrolytic solution for a battery.
- the present application claims the priority of Japanese Patent Application No. 2019-105066 filed in Japan on June 5, 2019, the contents of which are incorporated herein by reference.
- nano-sized fine substances have new properties that cannot be expressed in the bulk state.
- nanodiamond particles generally have a large proportion of surface atoms, the sum of van der Waals forces that can act between the surface atoms of adjacent particles is large, and agglutination is likely to occur.
- a phenomenon called agglutination can occur in which the Coulomb interaction between crystal planes of adjacent crystal faces contributes and very strongly aggregates.
- Non-Patent Document 1 when LiPF 6 is used as an electrolyte and an equal amount mixture of ethylene carbonate and diethyl carbonate is used as a solvent and an electrolytic solution in which organically modified nanodiamonds are dispersed is used for charging and discharging, a lithium layer is formed on the electrodes. Is described as having grown in a smooth state.
- an object of the present disclosure is to provide an electrolytic solution for a battery in which a nanocarbon material is highly dispersed and a lithium ion battery using the electrolytic solution for the battery.
- the present disclosure provides a battery electrolyte solution containing a dispersion medium, an electrolyte dissolved in the dispersion medium, and a nanocarbon material dispersed in the dispersion medium with an average dispersed particle diameter of 500 nm or less.
- the above battery electrolyte preferably contains a nanocarbon material in a proportion of 10 to 100,000 mass ppm.
- nanocarbon materials nanodiamond, fullerene, graphene, graphene oxide, nanographite, carbon nanotube, carbon nanofilament, onion-like carbon, diamond-like carbon, amorphous carbon, carbon black, carbon nanohorn, and It is preferable to use one or more selected from the group consisting of carbon nanocoils.
- nanodiamond particles having a primary particle diameter of 10 nm or less as the nanocarbon material.
- nanodiamonds containing nanodiamond particles and surface-modifying groups having polyoxyalkylene chains that surface-modify the nanodiamond particles as the nanocarbon material.
- polyoxyalkylene chain it is preferable to use one or more selected from the group consisting of a polyethylene glycol chain, a polypropylene glycol chain, and a polybutylene glycol chain.
- the surface-modified nanodiamond preferably has a structure in which the end of the polyoxyalkylene chain is sealed with an aliphatic hydrocarbon group.
- the surface modifying group preferably contains a silicon atom.
- nanodiamond particles and a polyglycerin chain that surface-modifies the nanodiamond particles and at least a part of the hydrogen atoms of the hydroxyl group in the polyglycerin chain are used. It is preferable to use surface-modified nanodiamonds containing a surface-modifying group substituted with a monovalent organic group.
- the surface-modified nano diamonds may have respective absorption peaks at around 1510 ⁇ 1540 cm -1 and around 1700 ⁇ 1730 cm -1.
- the surface-modified nanodiamond may have an absorption peak in the vicinity of 1730 to 1750 cm -1 in the FT-IR spectrum.
- the surface-modified nanodiamond may have peaks at around 5 to 45 ppm, around 60 to 85 ppm, and around 140 to 200 ppm, respectively, in the 13 C-NMR spectrum.
- the electrolytic solution for a battery preferably contains nanodiamond particles as a nanocarbon material and a dispersant having a mass average molecular weight of 500 or more and an amine value of 15 mgKOH / g or more.
- At least one selected from the group consisting of ethylene carbonate, propylene carbonate, ⁇ -butyrolactone, dimethyl carbonate, diethyl carbonate, and ethyl methyl carbonate is preferable to use at least one selected from the group consisting of ethylene carbonate, propylene carbonate, ⁇ -butyrolactone, dimethyl carbonate, diethyl carbonate, and ethyl methyl carbonate as the dispersion medium.
- LiPF 6 LiBF 4 , LiN (SO 2 CF 3 ) 2 , LiNC F 3 SO 3 , and LiN (SO 2 F) 2 shall be used as the electrolyte. Is preferable.
- the present disclosure also provides a lithium ion battery using the above-mentioned electrolyte for a battery.
- the battery electrolytic solution of the present disclosure it is possible to provide a battery electrolytic solution in which the nanocarbon material is highly dispersed. Further, according to the lithium ion battery of the present disclosure, since the lithium layer is formed smoothly even when charging / discharging is repeated or overcharged, the formation of dendrite can be suppressed, and the dendrite breaks through the separator. Can be suppressed.
- the battery electrolyte solution according to the embodiment of the present disclosure contains at least a dispersion medium, an electrolyte dissolved in the dispersion medium, and a nanocarbon material dispersed in the dispersion medium.
- the average dispersed particle size (D50, median diameter) of the nanocarbon material in the battery electrolyte is 500 nm or less, preferably 400 nm or less, more preferably 300 nm or less, still more preferably 200 nm or less, and particularly preferably 150 nm or less. Is.
- the lower limit of the average dispersed particle size is, for example, 5 nm or more, preferably 10 nm or more.
- the average dispersed particle size can be measured by a dynamic light scattering method.
- the nanocarbon material may be particles having a primary particle diameter of 10 nm or less (for example, 1 to 10 nm), preferably 8 nm or less (for example, 2 to 8 nm), and more preferably 6 nm or less (for example, 4 to 6 nm). preferable.
- nanocarbon material known or commonly used nanocarbon materials can be used, for example, nanodiamond, fullerene, graphene, graphene oxide, nanographite, carbon nanotube, carbon nanofilament, onion-like carbon, diamond-like carbon. , Amorphous carbon, carbon black, carbon nanohorns, carbon nanocoils and the like.
- nanodiamonds nanodiamonds (nanodiamond particles) are preferable.
- the nanocarbon material only one kind may be used, or two or more kinds may be used.
- nanodiamonds may be referred to as "ND".
- the nanodiamond particles are not particularly limited, and known or commonly used nanodiamond particles can be used.
- the ND particles may be surface-modified ND (surface-modified ND) particles or may be surface-modified ND particles.
- the surface-modified ND particles have a hydroxyl group (-OH) on the surface.
- ND particles only one kind may be used, or two or more kinds may be used.
- Examples of the compound or functional group that surface-modifies the ND particles in the surface-modified ND include a silane compound, a carboxyl group (-COOH), a phosphonate ion or a phosphonic acid residue, a surface modifying group having a vinyl group at the end, and an amide.
- Examples thereof include a group, a cation of a cationic surfactant, a group containing a polyoxyalkylene chain, a group containing a polyglycerin chain, and a silane compound.
- a group containing a polyoxyalkylene chain (polyoxyalkylene chain-containing surface modifying group) and a group containing a polyglycerin chain (from the viewpoint of being more excellent in dispersibility in a battery electrolytic solution).
- Polyglycerin chain-containing surface modifying group is preferred.
- the surface-modifying compound or functional group only one kind may be used, or two or more kinds may be used.
- the surface-modified ND containing the ND particles and the polyoxyalkylene chain-containing surface modifying group that surface-modifies the ND particles may be referred to as "polyoxyalkylene chain surface-modified ND”. Further, the surface-modified ND containing the ND particles and the polyglycerin chain-containing surface modifying group that surface-modifies the ND particles may be referred to as "polyglycerin chain surface-modified ND”.
- Zirconia may adhere (fix) to the surface-modified ND.
- a substance in which zirconia is attached to the surface-modified ND may be referred to as a "surface-modified nanodiamond composite". That is, the surface-modified ND complex contains surface-modified ND and zirconia attached to the surface-modified ND.
- the adhesion state of zirconia may be physical adhesion (adhesion, adhesion, etc.) or chemical adhesion (covalent bond with ND particles or surface modifying groups, bond by intermolecular force, hydrogen bond, ionic bond). Etc.), or both.
- zirconia only one kind may be used, or two or more kinds may be used.
- the ND particles constituting the surface-modified ND preferably include primary particles of nanodiamond. In addition, it may contain secondary particles in which several to several tens of the primary particles are aggregated.
- ND particles for example, a detonation method ND (that is, an ND generated by the detonation method) or a high temperature and high pressure method ND (that is, an ND generated by the high temperature and high pressure method) can be used.
- the detonation method ND is preferable in that the dispersibility in the electrolytic solution for a battery is more excellent, that is, the particle size of the primary particles is a single digit nanometer.
- the detonation method ND includes an air-cooled detonation method ND (that is, an ND generated by the air-cooled detonation method) and a water-cooled detonation method ND (that is, an ND generated by the water-cooled detonation method). .. Above all, the air-cooled detonation method ND is preferable in that the primary particles are smaller than the water-cooled detonation method ND.
- polyoxyalkylene chain in the above-mentioned polyoxyalkylene chain-containing surface modifying group examples include polyethylene glycol chain, polypropylene glycol chain, polytetramethylene glycol chain, polybutylene glycol chain and the like. Of these, polyethylene glycol chains, polypropylene glycol chains, and polybutylene glycol chains are preferable. Further, in each polyoxyalkylene chain-containing surface modifying group, the polyoxyalkylene chain may be composed of only one type of oxyalkylene or may be composed of two or more types of oxyalkylene. .. Examples of the polyoxyalkylene chain composed of two or more types of oxyalkylene include polyethylene glycol-polypropylene glycol chain. When the polyoxyalkylene chain is composed of two or more kinds of oxyalkylenes, the bonding form of the two or more kinds of oxyalkylenes may be random, alternating, or block.
- the polyoxyalkylene chain-containing surface modifying group preferably has a structure in which the end of the polyoxyalkylene chain is sealed with an aliphatic hydrocarbon group. That is, it is preferable that the hydrogen atom in the hydroxyl group at one end of the polyoxyalkylene chain is replaced with an aliphatic hydrocarbon group.
- the aliphatic hydrocarbon group that seals the end of the polyoxyalkylene chain is preferably an aliphatic hydrocarbon group having 1 to 10 carbon atoms, and more preferably an aliphatic hydrocarbon group having 1 to 6 carbon atoms.
- the aliphatic hydrocarbon group having 1 to 10 carbon atoms includes a methyl group, an ethyl group, a propyl group, an isopropyl group, a butyl group, an isobutyl group, an s-butyl group, a t-butyl group, a pentyl group, a hexyl group and a heptyl group.
- a linear or branched alkyl group such as an octyl group, a 2-ethylhexyl group or a decyl group; a linear or branched alkenyl group such as a vinyl or allyl group; an alkynyl group such as an ethynyl group or a propynyl group. Be done. Of these, linear or branched alkyl groups are preferable.
- the polyoxyalkylene chain has a number average molecular weight as a polyoxyalkylene, preferably 100 or more (for example, 100 to 50,000), more preferably 200 or more (for example, 200 to 20,000), and further preferably 500 or more (for example, for example). 1000 to 10000), particularly preferably 1000 or more (for example, 1000 to 6000).
- the average degree of polymerization (number average degree of polymerization) of the polyoxyalkylene chain is preferably 2 to 50, more preferably 4 to 45, and even more preferably 6 to 40.
- the average degree of polymerization is 2 or more, the steric hindrance between the surface modifying groups becomes sufficient and it is easy to disperse in the dispersion medium.
- n is 50 or less, the surface modifying groups are suppressed from being entangled with each other, and it is easy to disperse in the dispersion medium.
- the characteristics as a nanodiamond material are not easily impaired.
- the polyoxyalkylene chain-containing surface modifying group preferably contains a silicon atom.
- the silicon atom is preferably present between the polyoxyalkylene chain and the surface-modified ND particles.
- the silicon atom is preferably present in the surface modifying group as a Si—O bond.
- the polyoxyalkylene chain surface-modified ND preferably has a structure in which a silicon atom in the surface modifying group is bonded to ND particles via an oxygen atom.
- the polyoxyalkylene chain-containing surface modifying group is selected from the group consisting of a group represented by the following formula (1), a group represented by the following formula (2), and a group represented by the following formula (3). It is preferably one or more groups.
- the wavy bond in the following formulas (1) to (3) binds to the surface of the nanodiamond particles.
- R 1 represents a hydrogen atom or an aliphatic hydrocarbon group.
- the aliphatic hydrocarbon group is preferably an aliphatic hydrocarbon group having 1 to 10 carbon atoms, and more preferably an aliphatic hydrocarbon group having 1 to 6 carbon atoms.
- the aliphatic hydrocarbon group having 1 to 10 carbon atoms includes a methyl group, an ethyl group, a propyl group, an isopropyl group, a butyl group, an isobutyl group, an s-butyl group, a t-butyl group, a pentyl group, a hexyl group and a heptyl group.
- a linear or branched alkyl group such as an octyl group, a 2-ethylhexyl group or a decyl group; a linear or branched alkenyl group such as a vinyl group or an allyl group; an alkynyl group such as an ethynyl group or a propynyl group.
- linear or branched alkyl groups are preferable.
- R 2 represents an alkylene group, preferably an alkylene group having 1 to 4 carbon atoms.
- alkylene group having 1 to 4 carbon atoms include an ethylene group, an isopropylene group, an isobutylene group, and a tetramethylene group. Of these, an ethylene group and an isopropylene group are preferable.
- the plurality of R 2s may be the same or different. When the plurality of R 2s are different, the binding form of the oxyalkylene chain (—OR 2 ) may be random, alternating, or block.
- n is the average degree of polymerization (number average degree of polymerization) of the oxyalkylene chain (—OR 2 ), and represents a value of 2 or more.
- n is preferably 2 to 50, more preferably 4 to 45, and even more preferably 6 to 40.
- n is 2 or more, the steric hindrance between the surface modifying groups becomes sufficient and it is easy to disperse in the dispersion medium.
- n is 50 or less, the surface modifying groups are suppressed from being entangled with each other, and it is easy to disperse in the dispersion medium.
- the characteristics as a nanodiamond material are not easily impaired.
- X represents a single bond or a linking group (a divalent group having one or more atoms).
- the linking group include a divalent hydrocarbon group, a urethane bond, a carbonyl group, an ether bond, an ester bond, a carbonate group, an amide group, and a group in which a plurality of these groups are linked.
- Examples of the divalent hydrocarbon group include a linear or branched alkylene group having 1 to 18 carbon atoms, a divalent alicyclic hydrocarbon group, and the like.
- Examples of the linear or branched alkylene group having 1 to 18 carbon atoms include a methylene group, a methylmethylene group, a dimethylmethylene group, an ethylene group, a propylene group and a trimethylene group.
- Examples of the divalent alicyclic hydrocarbon group include 1,2-cyclopentylene group, 1,3-cyclopentylene group, cyclopentylidene group, 1,2-cyclohexylene group and 1,3-.
- Examples thereof include a cycloalkylene group (including a cycloalkylidene group) such as a cyclohexylene group, a 1,4-cyclohexylene group and a cyclohexylidene group.
- the number of carbon atoms in the divalent hydrocarbon group is preferably 1 to 10, and more preferably 2 to 6.
- linking group in X it is particularly preferable to contain a urethane bond, and a group in which a divalent hydrocarbon group (particularly, a linear or branched alkylene group) and a urethane bond are linked is more preferable. Further, it is preferable that the silicon atom in the above formulas (1) to (3) and the divalent hydrocarbon group in X (particularly, a linear or branched alkylene group) are directly bonded.
- k represents an integer of 1 to 18, preferably an integer of 1 to 6, and more preferably an integer of 1 to 3.
- R 3 and R 4 represent the same or different hydrogen atom, an aliphatic hydrocarbon group having 1 to 3 carbon atoms, or a group represented by the following formula (a).
- the aliphatic hydrocarbon group having 1 to 3 carbon atoms include a linear or branched alkyl group such as a methyl group, an ethyl group, a propyl group and an isopropyl group; and a linear chain such as a vinyl group and an allyl group.
- a branched chain alkenyl group; an alkynyl group such as an ethynyl group or a propynyl group can be used.
- linear or branched alkyl groups are preferable.
- R 3 in the formula (2) is the same as R 3 in the formula (1).
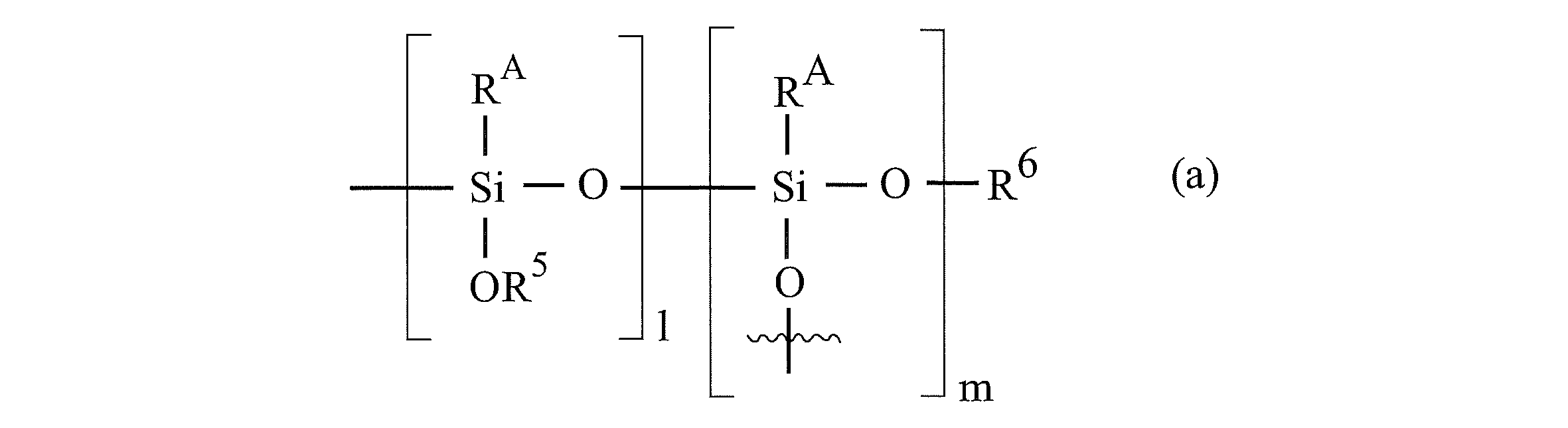
- RA represents [-X- (OR 2 ) n-OR 1 ].
- X, R 1 , R 2 , and n in RA are the same as those exemplified and described in the above formulas (1) to (3), respectively, and the preferred embodiments are also the same.
- the binding form when having two or more types of (OR 2 ) is also as described above.
- the bond extending to the left from the silicon atom bonds to the oxygen atom.
- the wavy bond in the formula binds to the surface of the nanodiamond particles.
- R 5 and R 6 represent hydrogen atoms or aliphatic hydrocarbon groups having 1 to 3 carbon atoms, which are the same or different.
- the aliphatic hydrocarbon group having 1 to 3 carbon atoms include a linear or branched alkyl group such as a methyl group, an ethyl group, a propyl group and an isopropyl group; and a linear chain such as a vinyl group and an allyl group.
- a branched chain alkenyl group; an alkynyl group such as an ethynyl group or a propynyl group can be used. Of these, linear or branched alkyl groups are preferable.
- l and m represent integers of 0 or more, which are the same or different.
- the order of joining the structural unit with l and the structural unit with m is not particularly limited. That is, the silicon atom in the formula (a) that bonds with the oxygen atom in the formula (1) or (2) may be the silicon atom in the structural unit with l, and the structural unit with m. It may be a silicon atom in.
- the oxygen atom in the formula (a) that binds to R 6 in the formula (a) may be the oxygen atom in the structural unit with l, and the oxygen atom in the structural unit with m. It may be.
- the method of combining the structural unit with l and the structural unit with m is random, alternate, or block. It may be any of.
- the polyoxyalkylene chain-containing surface-modified ND has a plurality of R 1 to R 6 , X, l, m, or n
- the plurality of R 1 to R 6 , X, l, m, and n are respectively. It may be the same or different.
- R 3 and R 4 are the groups represented by the above formula (a) is the unreacted alkoxysilyl group in the polyoxyalkylene chain-containing surface modifying group bonded to the ND particles and the unreacted alkoxysilyl group described later. It shows a structure in which an alkoxysilyl group in another polyoxyalkylene chain-containing surface modifying group bonded to the polyoxyalkylene chain-containing silane coupling agent and / or ND particles by dehydration condensation has reacted.
- the surface-modified ND having the polyoxyalkylene chain-containing surface modifying group can be produced, for example, through a modification step of reacting ND particles with a polyoxyalkylene chain-containing silane coupling agent.
- the ND particles to be reacted with the silane coupling agent preferably have a hydroxyl group on the surface.
- the surface-modified ND particles usually have a hydroxyl group (-OH) on the surface.
- -OH hydroxyl group
- the polyoxyalkylene chain-containing silane coupling agent is preferably a compound represented by the following formula (1').
- the polyoxyalkylene chain-containing silane coupling agent only one kind may be used, or two or more kinds may be used.
- R 1 , R 2 , X, and n are the same as those exemplified and described as those in the above formulas (1) to (3), respectively, and the preferred embodiments are also the same. Is. When two or more compounds represented by the above formula (1') are used, the plurality of R 1 , R 2 , X, and n may be the same or different.
- R 7 represents an aliphatic hydrocarbon group having 1 to 3 carbon atoms, which is the same or different.
- the aliphatic hydrocarbon group having 1 to 3 carbon atoms include a linear or branched alkyl group such as a methyl group, an ethyl group, a propyl group and an isopropyl group; and a linear chain such as a vinyl group and an allyl group.
- a branched chain alkenyl group; an alkynyl group such as an ethynyl group or a propynyl group can be used. Of these, linear or branched alkyl groups are preferable.
- the modification step is preferably performed in a solvent.
- the solvent include those exemplified and described as the dispersion medium described later and other organic solvents.
- the organic solvent include aliphatic hydrocarbons such as hexane, heptane and octane (particularly linear saturated aliphatic hydrocarbons); aromatic hydrocarbons such as benzene, toluene and xylene; cyclohexane and methylcyclohexane.
- Aliphatic hydrocarbons such as dimethylformamide (DMF), dimethylacetamide, N-methylpyrrolidone, dimethylsulfoxide; halogenation of chloroform, dichloromethane, dichloroethane, carbon tetrachloride, chlorobenzene, trifluoromethylbenzene, etc.
- Hydrocarbons chain or cyclic ethers such as diethyl ether, diisopropyl ether, dimethoxyethane, tetrahydrofuran (THF), dioxane; esters such as ethyl acetate and butyl acetate.
- cyclic ethers eg THF
- chain ketones eg MEK, MIBK
- the solvent only one kind may be used, or two or more kinds may be used.
- the modification step is performed by stirring a mixed solution containing dried nanodiamond, a silane coupling agent, and a solvent in a reaction vessel.
- the ND particles when the ND particles contain ND particle aggregates (adherent bodies) in which the ND particles adhere to form secondary particles, the ND particles are combined with the polyoxyalkylene chain-containing silane coupling agent.
- the reaction with the ND particles may be carried out while crushing or dispersing the ND particles.
- the ND particle agglomerates can be crushed into primary particles, the surface of the ND primary particles can be modified, and the dispersibility of the ND particles can be improved.
- Examples of the method for crushing or dispersing ND particles include a method of treating with a high shear mixer, a high shear mixer, a homomixer, a ball mill, a bead mill, a high-pressure homogenizer, an ultrasonic homogenizer, a colloid mill, a jet mill and the like. .. Above all, it is preferable to perform ultrasonic treatment in the presence of crushed media (for example, zirconia beads). By using the zirconia beads as the crushing medium, as a result, a surface-modified ND composite to which zirconia is attached and an electrolytic solution for a battery containing zirconia can be obtained.
- crushed media for example, zirconia beads
- Cavitation is generated by sonication, and the jet jet generated when the cavitation collapses causes the crushed media to obtain extremely large kinetic energy, and the crushed media collides with ND aggregates to give impact energy.
- the MD particles are disintegrated (crushed) from the ND aggregate, and the silane coupling agent acts on the ND particles in the dissociated state to bind them.
- the ND particles include an ND cohesive body to which the ND particles are adhered, the ND cohesive body can be crushed to the primary particles, the surface of the ND primary particles can be modified, and the dispersibility This is because an excellent surface-modified ND can be obtained.
- the diameter of the crushed media is, for example, 15 to 500 ⁇ m, preferably 15 to 300 ⁇ m, and particularly preferably 15 to 100 ⁇ m.
- the ratio of the ND particles to be subjected to the reaction to the silane coupling agent is, for example, 2: 1 to 1:80.
- the concentration of the ND particles in the solvent is, for example, 0.5 to 10% by mass, and the concentration of the silane coupling agent in the solvent is, for example, 5 to 60% by mass.
- the reaction time between the ND particles and the silane coupling agent is, for example, 4 to 20 hours. Further, it is preferable that the reaction is carried out while cooling the generated heat with ice water or the like.
- the polyglycerin chain-containing surface modifying group preferably contains a polyglycerin chain in which at least a part of the hydrogen atoms of the hydroxyl groups are replaced with monovalent organic groups.
- the polyglycerin chain-containing surface modifying group may be directly bonded to the ND particles or may be bonded via a divalent group.
- the divalent group include a divalent group exemplified as Y in the formula (4-2) described later.
- Examples of the monovalent organic group include a substituted or unsubstituted hydrocarbon group (monovalent hydrocarbon group), a substituted or unsubstituted heterocyclic group (monovalent heterocyclic group), and 2 of these. Examples thereof include a group bonded as described above and a group to which a bonding group is bonded to these. The bonded group may be directly bonded or may be bonded via a linking group. Examples of the linking group include amino group, ether bond, ester bond, phosphinic acid group, sulfide bond, carbonyl group, organic group substituted amide group, organic group substituted urethane bond, organic group substituted imide bond, thiocarbonyl group and siloxane.
- Examples include a bond, a group in which two or more of these are bonded, and the like.
- the bonding group is a group that bonds to an oxygen atom derived from a hydroxyl group at the end of a polyglycerin chain.
- a group that bonds with the oxygen atom to form an ester group, a urethane bond (carbamoyl group, etc.) is formed.
- Examples thereof include groups to be formed.
- hydrocarbon group in the monovalent organic group examples include an aliphatic hydrocarbon group, an alicyclic hydrocarbon group, an aromatic hydrocarbon group, and a group in which two or more of these are bonded.
- Examples of the aliphatic hydrocarbon group include an alkyl group, an alkenyl group, and an alkynyl group.
- Examples of the alkyl group include a C 1-20 alkyl group (preferably C 1-) such as a methyl group, an ethyl group, a propyl group, an isopropyl group, a butyl group, a hexyl group, an octyl group, an isooctyl group, a decyl group and a dodecyl group. 10 alkyl groups, more preferably C 1-4 alkyl groups) and the like.
- alkenyl group examples include a vinyl group, an allyl group, a metalyl group, a 1-propenyl group, an isopropenyl group, a 1-butenyl group, a 2-butenyl group, a 3-butenyl group, a 1-pentenyl group and a 2-pentenyl group.
- Examples thereof include a C 2-20 alkenyl group (preferably a C 2-10 alkenyl group, more preferably a C 2-4 alkenyl group) such as a 3-pentenyl group, a 4-pentenyl group and a 5-hexenyl group.
- alkynyl group examples include a C 2-20 alkynyl group such as an ethynyl group and a propynyl group (preferably a C 2-10 alkynyl group, more preferably a C 2-4 alkynyl group).
- Examples of the alicyclic hydrocarbon group include a C 3-12 cycloalkyl group such as a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group and a cyclododecyl group; and a C 3-12 cycloalkenyl group such as a cyclohexenyl group.
- Examples include C 4-15 crosslinked cyclic hydrocarbon groups such as bicycloheptanyl group and bicycloheptenyl group.
- aromatic hydrocarbon group examples include a C 6-14 aryl group such as a phenyl group and a naphthyl group (particularly, a C 6-10 aryl group).
- heterocycle forming the above heterocyclic group examples include aromatic heterocycles and non-aromatic heterocycles.
- a heterocycle is a 3- to 10-membered ring (preferably a 4- to 6-membered ring) having a carbon atom and at least one heteroatom (for example, an oxygen atom, a sulfur atom, a nitrogen atom, etc.) in the atoms constituting the ring. ), These fused rings can be mentioned.
- a heterocycle containing an oxygen atom as a heteroatom for example, a 3-membered ring such as an oxyran ring; a 4-membered ring such as an oxetane ring; a furan ring, a tetrahydrofuran ring, an oxazole ring, an isooxazole ring, a ⁇ -butyrolactone ring).
- 5-membered rings such as 4-oxo-4H-pyran ring, tetrahydropyran ring, morpholin ring and the like; benzofuran ring, isobenzofuran ring, 4-oxo-4H-chromen ring, chroman ring, isochroman ring and the like Heterocyclic ring; 3-oxatricyclo [4.3.1.1 4,8 ] undecane-2-one ring, 3-oxatricyclo [4.2.1.0 4,8 ] nonane-2-one ring , Etc.), heterocycles containing sulfur atoms as heteroatoms (eg, 5-membered rings such as thiophene ring, thiazole ring, isothiazole ring, thiazizole ring; 6-membered ring such as 4-oxo-4H-thiopyran ring A 5-membered ring such as a fused ring such as a benzothiophene ring) or a heterocycle
- 6-membered rings such as a ring, a pyrimidine ring, a pyrazine ring, a piperidine ring, a piperazine ring; an indole ring, an indolin ring, a quinoline ring, an acridin ring, a naphthylidine ring, a quinazoline ring, a fused ring such as a purine ring, etc.
- Examples of the group in which the aliphatic hydrocarbon group and the alicyclic hydrocarbon group are bonded include a cyclohexylmethyl group and a methylcyclohexyl group.
- Examples of the group in which the aliphatic hydrocarbon group and the aromatic hydrocarbon group are bonded include a C 7-18 aralkyl group (particularly, C 7-10 aralkyl group) such as a benzyl group and a phenethyl group, and a C such as a cinnamyl group.
- Examples thereof include a C 1-4 alkyl-substituted aryl group such as a 6-10 aryl-C 2-6 alkenyl group and a tolyl group, and a C 2-4 alkenyl-substituted aryl group such as a styryl group.
- Examples of the group in which two or more of the monovalent hydrocarbon group and / or the monovalent heterocyclic group are bonded via a linking group include the monovalent hydrocarbon group and / or the monovalent heterocycle.
- the monovalent organic group may have a substituent.
- substituents include halogen atoms such as fluorine atom, chlorine atom, bromine atom and iodine atom; hydroxyl group; mercapto group; carboxy group; amino group; oxo group and the like.
- Examples of the monovalent organic group in R 8 include a substituted or unsubstituted hydrocarbon group (monovalent hydrocarbon group), a substituted or unsubstituted heterocyclic group (monovalent heterocyclic group), and the like. Examples thereof include a group in which two or more of these are bonded.
- the bonded group may be directly bonded or may be bonded via a linking group.
- Examples of the linking group include amino group, ether bond, ester bond, phosphinic acid group, sulfide bond, carbonyl group, organic group substituted amide group, organic group substituted urethane bond, organic group substituted imide bond, thiocarbonyl group and siloxane.
- Examples include a bond, a group in which two or more of these are bonded, and the like.
- the monovalent hydrocarbon group, the monovalent heterocyclic group, and the group in which two or more of these are bonded are the monovalent organic group (polyglycerin chain terminal hydroxyl) in the above-mentioned polyglycerin chain-containing surface modifying group, respectively. Examples and explanations thereof include monovalent organic groups bonded to oxygen atoms derived from groups).
- the R 8 is preferably a substituted or unsubstituted hydrocarbon group, more preferably an alkyl group, further preferably an alkyl group having 1 to 18 carbon atoms, still more preferably an alkyl group having 1 to 6 carbon atoms, and further.
- An alkyl group having 1 to 4 carbon atoms is preferable, and an ethyl group or a butyl group is particularly preferable.
- R 8 in the groups represented by the above formula (4-1) is the same as each other. It may be present or it may be different.
- the polyglycerin chain-containing surface modifying group in which at least a part of the hydrogen atom of the hydroxyl group in the polyglycerin chain is replaced with a monovalent organic group has a polyglycerin chain represented by the following formula (4-2). Is preferable.
- the polyglycerin chain includes a linear structure, a branched chain structure, and a cyclic polyglycerin chain. -Y- (C 3 H 6 O 2 ) p- (C 3 H 5 O 2 R 9 ) q-OR 10 (4-2)
- [C 3 H 6 O 2 ] to which p in the above formula (4-2) is attached has one or more of the structures represented by the following formulas (5) to (7).
- [C 3 H 5 O 2 R 9 ] to which q of the above formula (4-2) is attached has one or more of the structures represented by the following formulas (8) to (10).
- p and q each indicate the average degree of polymerization (number average degree of polymerization) of the repeating unit in parentheses.
- p is a value of 0 or more
- q is a value of 0 or more
- p + q is a value of 1 or more.
- R 10 is a hydrogen atom
- q indicates a value exceeding 0.
- p + q is preferably 1 to 100, more preferably 2 to 40, and even more preferably 3 to 30.
- p + q is 1 or more, the steric hindrance between the surface modifying groups becomes sufficient and it is easy to disperse in the dispersion medium.
- [Q / (p + q)] is preferably 0.6 or more (for example, 0.8 to 1.0).
- the average degree of polymerization is defined by the number of glycidol units constituting the polyglycerin chain bonded to the surface functional group 1 of the raw material nanodiamond, and the number of surface functional groups of the raw material nanodiamond is an element analysis value of the raw material nanodiamond. It can be determined by measurement, measurement of acid value, or a combination of both.
- R 9 represents a monovalent organic group, preferably a group represented by the above formula (4-1).
- the monovalent organic group in R 9 for example, as a monovalent organic group in the above-mentioned polyglycerin chain-containing surface modifying group (a monovalent organic group bonded to an oxygen atom derived from the hydroxyl group at the terminal of the polyglycerin chain). Examples and explanations are given.
- [-OR 10 ] indicates the terminal of the polyglycerin chain
- R 10 represents a hydrogen atom or a monovalent organic group, and is preferably represented by the above formula (4-1). Indicates a group.
- R 10 is a monovalent organic group
- the monovalent organic group in R 10 for example, as a monovalent organic group in the above-mentioned polyglycerin chain-containing surface modifying group (a monovalent organic group bonded to an oxygen atom derived from the hydroxyl group at the terminal of the polyglycerin chain). Examples and explanations are given.
- Y represents a single bond or a divalent group, and the bond extending to the left from Y bonds to the ND particle.
- divalent hydrocarbon group A group in which two or more of these are bonded, and the like.
- divalent hydrocarbon group include a residue obtained by removing one hydrogen atom from the monovalent hydrocarbon group.
- the divalent group may have a substituent. Examples of the substituent include those exemplified and described as the substituent that the monovalent organic group may have.
- the joining order of the structural unit with p and the structural unit with q is not particularly limited. That is, the oxygen atom bonded to Y in the formula (4-2) may be an oxygen atom in the structural unit with p, or may be an oxygen atom in the structural unit with q.
- the carbon atom in the formula (4-2) that bonds with OR 10 in the formula (4-2) may be the carbon atom in the structural unit with p, and the configuration with q is added. It may be a carbon atom in the unit.
- the method of combining the structural unit with p and the structural unit with q is random, alternating, or block.
- the plurality of R 9s may be the same or different.
- the binding form of the structural unit with q may be random, alternating, or block.
- the mass ratio of ND to the polyglycerin chain-containing surface modifying group [ND / polyglycerin chain-containing surface modifying group] in the above polyglycerin chain surface modifying ND is not particularly limited, but is preferably 0.5 to 1.0. It is preferably 0.6 to 0.8. When the mass ratio is 0.5 or more (particularly 0.6 or more), the characteristics as a nanodiamond material are not easily impaired. When the mass ratio is 1.0 or less (particularly 0.8 or less), the degree of modification of the polyglycerin chain-containing surface modifying group is sufficient, and the dispersibility in the battery electrolyte is excellent.
- the mass ratio is determined based on the weight loss rate measured by thermogravimetric analysis, and the reduced weight is determined as the mass of the polyglycerin chain-containing surface modifying group.
- the absorption peak may fluctuate slightly depending on R 8 and the like in the above equation (4-1).
- the polyglycerin chain-containing surface-modified ND preferably has a peak in the vicinity of 140 to 200 ppm in the 13 C-NMR spectrum.
- the polyglycerin chain-containing surface-modified ND preferably has a peak in the vicinity of 60 to 85 ppm in the 13 C-NMR spectrum. The above peak is presumed to be a peak derived from polyglycerin.
- the polyglycerin chain-containing surface-modified ND preferably has a peak in the vicinity of 5 to 45 ppm in the 13 C-NMR spectrum. It is presumed that the polyglycerin chain-containing surface-modified ND having such a peak corresponds to the polyglycerin chain-containing surface-modified ND in the above formula (4-1) in which R contains a monovalent aliphatic hydrocarbon group. The peak, may vary slightly depending on the R 8 in the formula (4-1).
- the polyglycerin chain-containing surface-modified ND has a median diameter (D50) of 5 to 100 nm when the surface-modified ND is dispersed in at least one electrolytic solution so that the content ratio is 0.1% by mass. Is preferable, more preferably 5 to 80 nm, still more preferably 5 to 50 nm. When the median diameter is within the above range, the dispersibility in the electrolytic solution is more excellent.
- the polyglycerin chain-containing surface-modified ND can be obtained by ring-opening polymerization of glycidol directly on the ND particles.
- ND particles originally have carboxyl groups and hydroxyl groups generated in the manufacturing process, and the surface of ND can be modified by a polyglycerin chain by reacting these functional groups with glycidol.
- the reaction between the ND particles and glycidol can be carried out, for example, by adding glycidol and a catalyst to the ND particles in an inert gas atmosphere and heating to 50 to 100 ° C.
- a catalyst either an acidic catalyst or a basic catalyst can be used.
- the acidic catalyst include trifluoroboron etherate, acetic acid, and phosphoric acid.
- the basic catalyst include triethylamine, pyridine, dimethylaminopyridine, triphenylphosphine and the like.
- the polyglycerin chain surface-modified ND can also be obtained by ring-opening polymerization of glycidol on ND particles having a functional group containing active hydrogen introduced on the surface.
- the functional group containing active hydrogen is not particularly limited, and examples thereof include an amino group, a hydroxyl group, a carboxyl group, a mercapto group (thiol group), and a phosphinic acid group.
- JP-A-2012-82103, JP-A-2010-248023 and the like can be referred to.
- the ring-opening polymerization of glycidol on the ND particles in which a functional group containing active hydrogen is introduced on the surface can be carried out in the same manner as the ring-opening polymerization of glycidol on the ND particles described above.
- a surface modifying group in which the hydrogen atom of the hydroxyl group in at least a part of the polyglycerin chain is replaced with a monovalent organic group can be used, for example, in the polyglycerin chain surface modifying ND obtained by ring-opening polymerization of glycidol as described above. , Alcohol, isocyanic acid ester, carboxylated product, etc. are reacted. These compounds used in the reaction can be appropriately selected depending on the type of surface modifying group to be obtained. As the alcohol, isocyanic acid ester, carboxylated acid chloride and the like, only one kind may be used, or two or more kinds may be used.
- the polyglycerin chain-containing surface-modified ND is obtained by dehydrating and condensing by the reaction of the surface-modified ND into which the polyglycerin chain is introduced and alcohol to form an ether bond.
- an isocyanic acid ester used, the polyglycerin chain-containing surface-modified ND is added with an isocyanate group and a hydroxyl group in the isocyanic acid ester by the reaction between the surface-modified ND into which the polyglycerin chain is introduced and the isocyanate ester. It is obtained by forming a urethane bond (carbamoyl group).
- the polyglycerin chain-containing surface-modified ND is obtained by forming an ester bond by reacting the surface-modified ND into which the polyglycerin chain has been introduced with the carboxylated product.
- the alcohol, the isocyanate, and the carboxyl chloride can be appropriately selected according to the obtained polyglycerin chain-containing surface-modified ND.
- the organic group bonded to the hydroxyl group in the alcohol, the organic group on the nitrogen atom in the isocyanate, and the organic group bonded to the carbonyl group in the carboxylated product correspond to R 8 in the above formula (4-1), respectively.
- the monovalent organic group exemplified and described as R 8 described above can be mentioned.
- the alcohol is preferably C 1-18 alcohol, more preferably ethanol, n-butanol, or n-hexanol.
- the isocyanic acid ester is preferably alkyl isocyanate, more preferably C 1-18 alkyl isocyanate, still more preferably ethyl isocyanate, n-butyl isocyanate, n-hexyl isocyanate.
- acyl chloride is preferable, and the number of carbon atoms in the acyl group is preferably C 1-19 alkyl, more preferably valeryl chloride.
- the reaction with the alcohol, the isocyanate, or the carboxyl chloride may be carried out in a solvent.
- the solvent include the above-mentioned organic solvent.
- the reaction can be carried out, for example, by adding a catalyst to the ND particles under an inert gas atmosphere and stirring at room temperature.
- a basic catalyst is preferable.
- the basic catalyst include tertiary amines such as triethylamine, pyridine, dimethylaminopyridine, and triphenylphosphine.
- the basic catalyst is a liquid, the basic catalyst may be used as the reaction solvent.
- the ratio (former: latter, mass ratio) of the polyglycerin chain surface-modified ND particles to be subjected to the reaction to the alcohol, the isocyanate, and / or the carboxyl chloride is, for example, 2: 1 to 1:80. ..
- the concentration of the polyglycerin chain surface-modified ND particles in the solvent is, for example, 0.5 to 10% by mass, and the concentration of the isocyanic acid ester in the solvent is, for example, 5 to 60% by mass.
- the reaction time between the polyglycerin chain surface-modified ND particles and the alcohol, the isocyanate ester, and / or the carboxyl chloride is, for example, 4 to 24 hours.
- the electrolyte As the electrolyte, a known or commonly used electrolyte used in a battery electrolyte can be used.
- the electrolyte is preferably an electrolyte used in a non-aqueous electrolyte solution, and preferably a lithium salt.
- RI to R VII represent perfluoroalkyl groups having 1 to 8 carbon atoms, which are the same or different from each other.
- LiPF 6 , LiBF 4 , LiN (SO 2 CF 3 ) 2 , LiNC F 3 SO 3 , and LiN (SO 2 F) 2 are preferable from the viewpoint of being more excellent in dispersibility of the nanocarbon material.
- electrolyte only one kind may be used, or two or more kinds may be used.
- the dispersion medium functions as a solvent for the electrolyte and a dispersion medium for the nanocarbon material in the electrolytic solution for batteries.
- a known or commonly used solvent used for an electrolytic solution for a battery can be used.
- the dispersion medium is preferably a non-aqueous solvent, and examples thereof include carbonate compounds (saturated cyclic carbonates, chain carbonates, etc.), cyclic carboxylic acid esters, cyclic ethers, and sulfone compounds.
- the dispersion medium only one kind may be used, or two or more kinds may be used.
- saturated cyclic carbonate examples include those having an alkylene group having 2 to 4 carbon atoms, and a saturated cyclic carbonate having 2 to 3 carbon atoms is preferable.
- saturated cyclic carbonate examples include ethylene carbonate, propylene carbonate, butylene carbonate and the like.
- chain carbonate for example, one having 3 to 7 carbon atoms is used, and a chain carbonate having 3 to 5 carbon atoms is preferable.
- chain carbonate include dimethyl carbonate, diethyl carbonate, di-n-propyl carbonate, diisopropyl carbonate, n-propylisopropyl carbonate, ethylmethyl carbonate, methyl-n-propyl carbonate, n-butylmethyl carbonate, isobutylmethyl carbonate, and the like.
- Examples thereof include t-butyl methyl carbonate, ethyl-n-propyl carbonate, n-butyl ethyl carbonate, isobutyl ethyl carbonate and t-butyl ethyl carbonate.
- a chain carbonate having a fluorine atom (hereinafter, may be referred to as "fluorinated chain carbonate”) can also be mentioned.
- the number of fluorine atoms contained in the fluorinated chain carbonate is 1 or more.
- the upper limit of the number of fluorine atoms is, for example, 6, preferably 4.
- the fluorinated chain carbonate has a plurality of fluorine atoms, the plurality of fluorine atoms may be bonded to the same carbon or may be bonded to different carbons.
- the fluorinated chain carbonate include a fluorinated dimethyl carbonate derivative, a fluorinated ethyl methyl carbonate derivative, and a fluorinated diethyl carbonate derivative.
- fluorinated dimethyl carbonate derivative examples include fluoromethylmethyl carbonate, difluoromethylmethyl carbonate, trifluoromethylmethyl carbonate, bis (fluoromethyl) carbonate, bis (difluoromethyl) carbonate, bis (trifluoromethyl) carbonate and the like. ..
- fluorinated ethyl methyl carbonate derivative examples include 2-fluoroethyl methyl carbonate, ethyl fluoromethyl carbonate, 2,2-difluoroethyl methyl carbonate, 2-fluoroethyl fluoromethyl carbonate, ethyl difluoromethyl carbonate, 2,2,2-.
- fluorinated ethyl methyl carbonate derivative examples include 2-fluoroethyl methyl carbonate, ethyl fluoromethyl carbonate, 2,2-difluoroethyl methyl carbonate, 2-fluoroethyl difluoromethyl carbonate and ethyl trifluoromethyl carbonate.
- fluorinated diethyl carbonate derivative examples include ethyl- (2-fluoroethyl) carbonate, ethyl- (2,2-difluoroethyl) carbonate, bis (2-fluoroethyl) carbonate, and ethyl- (2,2,2-tri).
- Fluoroethyl) carbonate 2,2-difluoroethyl-2'-fluoroethyl carbonate, bis (2,2-difluoroethyl) carbonate, 2,2,2-trifluoroethyl-2'-fluoroethyl carbonate, 2,2 , 2-Trifluoroethyl-2', 2'-difluoroethyl carbonate, bis (2,2,2-trifluoroethyl) carbonate and the like.
- Examples of the cyclic carboxylic acid ester include a cyclic carboxylic acid ester having 3 to 12 carbon atoms, preferably a cyclic carboxylic acid ester having 3 to 10 carbon atoms, and more preferably a cyclic carboxylic acid ester having 3 to 8 carbon atoms.
- Examples of the cyclic carboxylic acid ester include ⁇ -butyrolactone, ⁇ -valerolactone, ⁇ -caprolactone, and epsilon caprolactone.
- Examples of the cyclic ether include cyclic ethers having 3 to 6 carbon atoms, and cyclic ethers having 3 to 5 carbon atoms are preferable.
- Examples of the cyclic ether include tetrahydrofuran, 2-methyltetrahydrofuran, 3-methyltetrahydrofuran, 1,3-dioxane, 2-methyl-1,3-dioxane, 4-methyl-1,3-dioxane, 1,4-dioxane, and the like. And these fluorinated compounds and the like.
- the sulfone compound examples include cyclic sulfone and chain sulfone.
- the cyclic sulfone has, for example, 3 to 6, preferably 3 to 5.
- the chain sulfone has, for example, 2 to 6, preferably 2 to 5.
- the number of sulfonyl groups in one molecule of the sulfone compound is not particularly limited, but is usually 1 or 2.
- cyclic sulfone examples include monosulfone compounds such as trimethylene sulfone, tetramethylene sulfone, and hexamethylene sulfone; and disulfone compounds such as trimethylene disulfone, tetramethylene disulfone, and hexamethylene disulfone.
- tetramethyl sulfolanes examples include sulfolane, 2-methyl sulfolane, 3-methyl sulfolane, 2-fluoro sulfolane, 3-fluoro sulfolane, 2,2-difluoro sulfolane, 2,3-difluoro sulfolane, and 2, 4-Difluorosulfolane, 2,5-difluorosulfolane, 3,4-difluorosulfolane, 2-fluoro-3-methylsulfolane, 2-fluoro-2-methylsulfolane, 3-fluoro-3-methylsulfolane, 3-fluoro- 2-Methyl sulfolane, 4-fluoro-3-methyl sulfolane, 4-fluoro-2-methyl sulfolane, 5-fluoro-3-methyl sulfolane, 5-fluoro-2-methyl sulfolane, 2-fluoro
- chain sulfone examples include dimethyl sulfone, ethyl methyl sulfone, diethyl sulfone, n-propyl methyl sulfone, n-propyl ethyl sulfone, di-n-propyl sulfone, isopropyl methyl sulfone, isopropyl ethyl sulfone, diisopropyl sulfone, and n-butyl.
- saturated cyclic carbonate, chain carbonate, and cyclic carboxylic acid ester are preferable, and ethylene carbonate, propylene carbonate, ⁇ -butyrolactone, and dimethyl carbonate are more preferable from the viewpoint of being more excellent in dispersibility of the nanocarbon material. Diethyl carbonate and ethyl methyl carbonate.
- the above battery electrolyte solution may further contain a dispersant.
- a dispersant having a mass average molecular weight of 500 or more and an amine value of 15 mgKOH / g or more (sometimes referred to as "dispersant (A)"). Is preferable.
- the dispersant By using such a dispersant, the dispersibility of the ND particles in the battery electrolyte is particularly excellent.
- the dispersant only one kind may be used, or two or more kinds may be used.
- the mass average molecular weight of the dispersant (A) is 500 or more, preferably 650 or more, and more preferably 950 or more.
- the mass average molecular weight is preferably 20,000 or less, more preferably 10,000 or less.
- the mass average molecular weight is a standard polystyrene-equivalent molecular weight measured by gel permeation chromatography (GPC).
- the amine value of the dispersant (A) is 15 mgKOH / g or more, preferably 18 mgKOH / g or more, more preferably 20 mgKOH / g or more, still more preferably 30 mgKOH / g or more.
- the amine value is preferably 100 mgKOH / g or less, more preferably 90 mgKOH / g or less, and further preferably 60 mgKOH / g or less.
- Examples of the dispersant (A) include a compound having a structure derived from polyalkylene glycol monoalkyl ether (particularly, a structure derived from polyethylene glycol monoalkyl ether or a structure derived from polypropylene glycol monoalkyl ether), and a compound having a carbamate structure.
- a compound having a structure derived from polycaprolactone is preferable.
- Each of the above compounds may contain only one kind, or may contain two or more kinds. Further, one compound may independently have one or more of the above structures.
- dispersant (A) a commercially available product can also be used.
- examples of commercially available products of the dispersant (A) include the product name "DISPERBYK-2008", the product name “BYK-9076", the product name “BYK-9077”, and the product name "ANTI-TERRA-U” (above, BIC). ⁇ Chemie), etc.
- the nanocarbon material is preferably a surface-modified ND in which a silane compound is bonded to the surface.
- the silane compound preferably has a hydrolyzable group and an aliphatic hydrocarbon group.
- the silane compound used for surface modification of ND particles may be only one kind or two or more kinds.
- the silane compound preferably contains at least a compound represented by the following formula (11-1).
- R 11 , R 12 , and R 13 represent the same or different aliphatic hydrocarbon groups having 1 to 3 carbon atoms.
- R 14 represents an aliphatic hydrocarbon group having 1 or more carbon atoms.
- Examples of the aliphatic hydrocarbon group having 1 to 3 carbon atoms in R 11 , R 12 , and R 13 include a linear or branched alkyl group such as a methyl group, an ethyl group, a propyl group, and an isopropyl group; vinyl.
- Examples include linear or branched alkenyl groups such as groups and allyl groups; alkynyl groups such as ethynyl groups and propynyl groups. Of these, linear or branched alkyl groups are preferable.
- R 14 is an aliphatic hydrocarbon group having 1 or more carbon atoms, and is, for example, a methyl group, an ethyl group, a propyl group, an isopropyl group, a butyl group, an isobutyl group, an s-butyl group, a t-butyl group, a pentyl group, and the like.
- 11-Dodecenyl group oleyl group and other linear or branched alkenyl groups; examples thereof include ethynyl group, propynyl group, decynyl group, pentadecynyl group, octadecynyl group and the like linear or branched alkynyl group.
- R 14 is preferably an aliphatic hydrocarbon group having 4 or more carbon atoms, and is particularly preferable, because it is excellent in the aggregation suppressing effect because it can cause a larger steric hindrance and can impart a higher degree of dispersibility. It is an aliphatic hydrocarbon group having 6 or more carbon atoms. The upper limit of the number of carbon atoms of the aliphatic hydrocarbon group is, for example, 25, preferably 20, and more preferably 12. Further, as the aliphatic hydrocarbon group, a linear or branched alkyl group or an alkenyl group is preferable, and a linear or branched alkyl group is particularly preferable.
- R 14 When R 14 is an aliphatic hydrocarbon group having 4 or more carbon atoms, it exhibits an affinity for an organic dispersion medium and can cause a larger steric hindrance, so that it has an excellent aggregation inhibitory effect and is a group containing an oxygen atom.
- R 14 exhibits an affinity for the dispersion medium, excellent affinity for the dispersion medium, exerts a more excellent dispersibility in the electrolytic solution for a battery be able to.
- examples of the ND particles surface-modified with the silane compound include ND particles having a structure surface-modified with a group represented by the following formula (11).
- R 14 represents an aliphatic hydrocarbon group having 1 or more carbon atoms.
- R 11 ', R 12' is a group the same or different and each represents a hydrogen atom, represented by aliphatic hydrocarbon group having 1 to 3 carbon atoms, or the following formula (b).
- the wavy bond in the formula binds to the surface of the nanodiamond particles.
- R 14 represents an aliphatic hydrocarbon group having 1 or more carbon atoms.
- R 13 and R 15 are the same or different, and represent a hydrogen atom or an aliphatic hydrocarbon group having 1 to 3 carbon atoms.
- r and s are the same or different and indicate integers of 0 or more.
- the bond extending to the left from the silicon atom bonds to the oxygen atom.
- the wavy bond is bonded to the surface of the nanodiamond particles.
- R 14 in the formula (11) in corresponds to R 14 in the formula (11-1).
- Examples of the aliphatic hydrocarbon group having 1 to 3 carbon atoms in R 11 ', R 12 ', R 13 and R 15 in the above formula (11) include a methyl group, an ethyl group, a propyl group and an isopropyl group.
- Examples thereof include a linear or branched alkyl group; a linear or branched alkenyl group such as a vinyl group and an allyl group; and an alkynyl group such as an ethynyl group and a propynyl group. Of these, linear or branched alkyl groups are preferable.
- R and s are the number of structural units shown in parentheses, and indicate the same or different integers of 0 or more. When r and s are 2 or more, the method of combining the two or more structural units may be random, alternating, or block.
- the silane compound surface-modified ND particle has, for example, a group represented by the following formula (11') and other surface functional groups (for example, amino group, hydroxyl group, carboxyl). It may have other functional groups such as groups).
- the other functional groups may be only one kind or two or more kinds.
- R 11 the formula (11)', R 14 are as defined above.
- the wavy bond in the formula binds to the surface of the ND particle.
- the said compound is, for example, OR 11 groups and OR 12 groups in the above formula (11-1).
- OR 13 groups and other hydrolyzable alkoxysilyl groups are easily hydrolyzed to form silanol groups. Therefore, for example, one of the silanol groups is covalently bonded to the hydroxyl group existing on the surface of the ND particles.
- the silanol groups of other silane compounds can be condensed with the remaining two silanol groups to form a siloxane bond (Si—O—Si), giving the ND particles an affinity for the dispersion medium. It can be imparted, and even more excellent dispersibility can be exhibited in the electrolytic solution for a battery.
- the electrolytic solution for a battery may be composed of only an electrolyte, a nanocarbon material, and a dispersion medium, may further contain a dispersant, and may be overcharged within a range that does not impair the above-mentioned effects. It may contain a known or commonly used additive contained in a battery electrolyte such as an inhibitor.
- the content ratio of the nanocarbon material in the electrolytic solution for a battery is, for example, 1 to 100,000 mass ppm, preferably 10 to 10000 mass ppm, and more preferably 100 to 1000 mass ppm.
- the battery electrolyte is particularly excellent in dispersibility of the nanocarbon material in the content ratio in the above range.
- the content ratio of the electrolyte in the battery electrolyte is, for example, 5 to 30% by mass, preferably 8 to 18% by mass, and more preferably 10 to 15% by mass. When the content ratio is within the above range, the conductivity becomes appropriate.
- the content ratio of the dispersion medium in the electrolytic solution for batteries is, for example, 75 to 94% by mass.
- the content ratio of the non-aqueous solvent in the total amount of the dispersion medium is, for example, 60% by mass or more, preferably 70% by mass or more, more preferably 80% by mass or more, and particularly preferably 90% by mass or more.
- the content ratio of the dispersant (for example, the dispersant (A)) in the battery electrolyte is, for example, 0.1 to 10000 parts by mass, preferably 10 to 1000 parts by mass with respect to 100 parts by mass of the total amount of the nanocarbon material. , More preferably 50 to 500 parts by mass.
- the content ratio of the dispersant is within the above range, the dispersibility of the nanocarbon material in the electrolytic solution for a battery is more excellent.
- the battery electrolyte solution preferably has a haze value of 5 or less, more preferably 3 or less, and further preferably 1 or less. Since the battery electrolyte has excellent dispersibility of the nanocarbon material, it is possible to obtain an electrolyte having the haze value.
- the haze value can be measured based on JIS K 7136.
- the viscosity of the battery electrolyte solution at 25 ° C. is preferably 0.1 to 100 mPa ⁇ s, more preferably 0.5 to 50 mPa ⁇ s, and even more preferably 1.0 to 30 mPa ⁇ s.
- the rotor and the rotation speed of the rotor at the time of measuring the viscosity are appropriately selected according to the measured values.
- the viscosity can be measured using, for example, an EMS viscometer (trade name "EMS1000", manufactured by Kyoto Denshi Kogyo Co., Ltd.).
- the electrolytic solution for a battery may be an electrolytic solution used for either a primary battery or a secondary battery.
- the electrolytic solution is for a secondary battery.
- it is preferably an electrolytic solution for a lithium ion battery because it is particularly excellent in dispersibility in an electrolytic solution used for a lithium ion battery.
- the shape of the battery is not particularly limited, and may be any of a cylindrical type, a square type, a laminated type, a coin type, a large size, and the like.
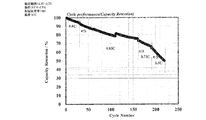
- the electrolytic solution for a battery can be practically used for a battery (particularly, a lithium ion battery), and in a battery provided with a separator, the nanocarbon material passes through the through hole of the separator and is free between the positive electrode and the negative electrode. You can move to. Further, even when used for a battery such as a lithium ion secondary battery in which dendrites are likely to be formed when repeatedly charged and discharged or overcharged, the lithium layer is formed smoothly, so that dendrites are formed. Can be suppressed, and dendrites can be suppressed from breaking through the separator. Therefore, by using the above-mentioned electrolytic solution for batteries, it is not necessary to use a separator having high strength, and the options for usable separators are expanded.
- Example 1 Surface-modified ND particles and an electrolytic solution were produced through the following steps.
- a molded explosive equipped with an electric detonator was installed inside a pressure-resistant container for detonation, and the container was sealed.
- the container is made of iron and the volume of the container is 15 m 3 .
- As the explosive 0.50 kg of a mixture of TNT and RDX was used.
- the mass ratio of TNT to RDX (TNT / RDX) in this explosive is 50/50.
- the electric detonator was detonated and the explosive was detonated in the container (generation of ND by the detonation method).
- the temperature of the container and its inside was lowered by leaving it at room temperature for 24 hours.
- the ND crude product adhering to the inner wall of the container (including the cohesive body of the ND particles and soot generated by the above detonation method) is scraped off with a spatula, and the ND crude product is scraped off.
- the ND crude product adhering to the inner wall of the container (including the cohesive body of the ND particles and soot generated by the above detonation method) is scraped off with a spatula, and the ND crude product is scraped off. was recovered.
- an acid treatment step was performed on the ND crude product obtained by performing the above-mentioned production step a plurality of times. Specifically, the slurry obtained by adding 6 L of 10% by mass hydrochloric acid to 200 g of the crude ND product was heat-treated for 1 hour under reflux under normal pressure conditions. The heating temperature in this acid treatment is 85 to 100 ° C. Next, after cooling, the solid content (including the ND adherent and soot) was washed with water by decantation. The solid content was repeatedly washed with water by decantation until the pH of the precipitate was from the low pH side to 2.
- an oxidation treatment step was performed. Specifically, 6 L of 98% by mass sulfuric acid and 1 L of 69% by mass nitric acid are added to a precipitate (including an ND adhering body) obtained through decantation after acid treatment to form a slurry, which is then added.
- the slurry was heat-treated for 48 hours under reflux under normal pressure conditions. The heating temperature in this oxidation treatment is 140 to 160 ° C.
- the solid content (including the ND adherent) was washed with water by decantation. The supernatant liquid at the beginning of washing with water was colored, and the solid content was repeatedly washed with water by decantation until the supernatant liquid became visually transparent.
- the precipitate liquid (liquid containing the ND cohesive body) obtained through the above-mentioned water washing treatment was subjected to a drying step to obtain a dry powder (ND cohesive body).
- a drying step evaporative drying performed using an evaporator was adopted.
- the ND particles were reacted with a polyoxyalkylene chain-containing silane coupling agent by ultrasonic treatment. It was gray at first, but gradually became smaller in particle size and dispersed, and finally became a uniform and black liquid. This is because the ND particles are sequentially disintegrated (crushed) from the ND cohesive body, and the polyoxyalkylene chain-containing silane coupling agent acts on the dissociated ND particles to bond them, resulting in surface-modified ND particles. This is probably because the particles are dispersed and stabilized in a toluene solvent.
- the obtained dispersion was distilled under reduced pressure using an evaporator to distill off methyl ethyl ketone, and further allowed to stand in a vacuum dryer at 100 ° C. for 24 hours to obtain a dry solid content.
- a transparent ND-dispersed electrolyte was obtained by stirring with an ultrasonic cleaner for 30 minutes.
- the obtained electrolyte was centrifuged at 6900 ⁇ g for 15 minutes to obtain a supernatant (transparent ND-dispersed electrolyte).
- the median diameter (particle size D50) of the surface-modified ND particles obtained in this step was 150 nm.
- Example 2 In the reaction between the ND particles and the silane coupling agent, 1.2 g of hexyltrimethoxysilane was used instead of 8 g of the polyoxyalkylene chain-containing silane coupling agent used in Example 1, and instead of 15 cc of methyl ethyl ketone.
- the ND particles were reacted with an alkyl chain-containing silane coupling agent (hexyltrimethoxysilane) by ultrasonic treatment for 15 hours in the same manner as in Example 1 except that 15 cc of methyl isobutyl ketone was used.
- an alkyl chain-containing silane coupling agent hexyltrimethoxysilane
- the median diameter (particle size D50) of the surface-modified ND particles obtained in this step was 25 nm.
- 15 cc of methyl isobutyl ketone obtained by dissolving a dispersant "BYK-9077" (manufactured by Big Chemie Japan Co., Ltd.) having a mass 100 times the mass of a separately prepared ND solid content is mixed with the above ND dispersion solution.
- Methyl isobutyl ketone was distilled off by vacuum distillation using an evaporator, and the mixture was allowed to stand at 100 ° C. for 24 hours in a vacuum dryer to obtain a dry solid content.
- EC DEC (1: 1 v / v%) (manufactured by Kishida Chemical Co., Ltd.) in which 1 M of LiBF4 was dissolved with respect to the dry solid content was added to the vial so that the ND content was 0.05% by mass.
- a transparent ND-dispersed electrolyte was obtained by stirring with an ultrasonic cleaner for 30 minutes. The supernatant obtained after standing for 20 days was centrifuged at 6900 ⁇ g for 15 minutes to obtain a supernatant (transparent ND dispersion electrolyte).
- the median diameter (particle size D50) of the surface-modified ND particles obtained in this step was 35.8 nm.
- Example 3 4.5 g of the dry powder (ND cohesive body) obtained through the drying step in Example 1 was placed in the core tube of a gas atmosphere furnace (trade name "gas atmosphere tube furnace KTF045N1", manufactured by Koyo Thermo System Co., Ltd.). After standing still and allowing nitrogen gas to flow through the core tube at a flow rate of 1 L / min for 30 minutes, the flow gas is switched from nitrogen to a mixed gas of oxygen and nitrogen, and the mixed gas is passed through the core tube at a flow rate of 1 L / min. Continued to flow through the core tube. The oxygen concentration in the mixed gas is 4% by volume. After switching to the mixed gas, the temperature inside the furnace was raised to 400 ° C., which is the set heating temperature.
- the rate of temperature rise was 10 ° C./min up to 380 ° C., which is 20 ° C. lower than the set heating temperature, and 1 ° C./min from 380 ° C. to 400 ° C. thereafter. Then, while maintaining the temperature condition in the furnace at 400 ° C., the ND powder in the furnace was subjected to oxygen oxidation treatment. The processing time was 3 hours.
- the hydrogenation process was carried out using the above-mentioned gas atmosphere furnace. Specifically, after the nitrogen gas is continuously passed through the gas atmosphere furnace in which the ND powder that has undergone the oxygen oxidation step is arranged at a flow rate of 1 L / min for 30 minutes, the flowing gas is changed from nitrogen to hydrogen. The gas was switched to a mixed gas of nitrogen and nitrogen, and the mixed gas was continuously passed through the core tube at a flow rate of 1 L / min. The hydrogen concentration in the mixed gas is 2% by volume. After switching to the mixed gas, the temperature inside the furnace was raised to the set heating temperature of 600 ° C. The heating rate was 10 ° C./min. Then, while maintaining the temperature condition in the furnace at 600 ° C., the ND powder in the furnace was hydrogenated. The processing time was 5 hours. As described above, the hydrogenated ND powder was obtained.
- the crushing process was performed. Specifically, first, 0.9 g of ND powder and 29.1 ml of pure water that had undergone the above-mentioned hydrogenation step were added to a 50 ml sample bottle and mixed to obtain about 30 ml of slurry. After adjusting the pH to 4 with 1N hydrochloric acid, the slurry was sonicated. In the ultrasonic treatment, an ultrasonic irradiator (trade name "ultrasonic cleaner AS-3", manufactured by AS ONE) was used to irradiate the slurry with ultrasonic waves for 2 hours. ..
- an ultrasonic irradiator trade name "ultrasonic cleaner AS-3", manufactured by AS ONE
- bead milling was performed using a bead milling device (trade name "parallel four-cylinder sand grinder LSG-4U-2L type", manufactured by IMEX Co., Ltd.). Specifically, 30 ml of the slurry after ultrasonic irradiation and zirconia beads having a diameter of 30 ⁇ m are put into a 100 ml mill container, Vessel (manufactured by Imex Co., Ltd.) and sealed, and the device is driven to perform bead milling. Executed. In this bead milling, the input amount of zirconia beads is, for example, 33% by volume with respect to the volume of the mill container, the rotation speed of the mill container is 2570 rpm, and the milling time is 2 hours.
- a bead milling device trade name "parallel four-cylinder sand grinder LSG-4U-2L type", manufactured by IMEX Co., Ltd.
- the slurry that had undergone the above-mentioned crushing step was centrifuged using a centrifuge device (classification operation).
- the centrifugal force in this centrifugation treatment was 20000 ⁇ g, and the centrifugation time was 10 minutes.
- 10 ml of the supernatant of the ND-containing solution that had undergone the centrifugation treatment was collected.
- the solid content concentration of this nanodiamond dispersion was 2.1% by mass, and the pH was 5.40.
- the median diameter (particle size D50) of the ND dispersion obtained as described above was 35.8 nm.
- the ND particle aqueous dispersion obtained above was dried using an evaporator to obtain a black dry powder.
- the obtained dry powder (100 mg) was added to 12 mL of glycidol placed in a glass reactor, and used in an ultrasonic cleaner (trade name "BRANSON2510", manufactured by Marshall Scientific) at room temperature. It was sonicated for hours to dissolve. This was reacted at 140 ° C. for 20 hours while stirring under a nitrogen atmosphere. After cooling the reaction mixture, 120 mL of methanol was added, the mixture was sonicated, and then centrifuged at 50400 ⁇ g for 2 hours to obtain a precipitate.
- Pyridine was added to the PG-ND gray powder, and the concentration was adjusted to 4 g / L based on the mass of the ND particles to obtain a PG-ND particle pyridine dispersion.
- a PG-ND particle pyridine dispersion To 20 ml of the obtained PG-ND particle pyridine dispersion, 4 g of methyl isocyanate was added, and the mixture was stirred at room temperature for 30 minutes. Subsequently, 40 ml of hexane was added, and the mixture was centrifuged at 20000 ⁇ g for 10 minutes to obtain a precipitate. This washing operation was repeated 3 times, and after removing pyridine, the mixture was allowed to stand at 100 ° C. for 24 hours in a vacuum dryer to obtain a dry solid content.
- EC DEC (1: 1 v / v%) (manufactured by Kishida Chemical Co., Ltd.) in which 1 M of LiBF 4 was dissolved with respect to the dry solid content was added to the vial so that the ND content was 0.05% by mass.
- a transparent ND-dispersed electrolyte was obtained by stirring with an ultrasonic cleaner for 30 minutes. The obtained electrolyte was centrifuged at 6900 ⁇ g for 15 minutes to obtain a supernatant (transparent ND-dispersed electrolyte).
- the median diameter (particle size D50) of the surface-modified nanodiamond particles obtained in this step was 60.3 nm, and the mass ratio [ND / polyglycerin chain-containing surface modifying group] determined by thermogravimetric analysis was 0.60. ..
- Example 4 In the modification step of Example 3, 4 g of butyl isocyanate was used instead of 4 g of methyl isocyanate, and EC: DEC (1: 1 v / v%) in which 1 M of LiBF 4 was dissolved (Kishida Chemistry). In the same manner as in Example 3 except that EC: DEC (1: 1 v / v%) (manufactured by Kishida Chemical Co., Ltd.) in which 1 M of LiPF 6 was dissolved was used instead of (manufactured by), the mixture was stirred with an ultrasonic cleaner for 30 minutes. A transparent ND dispersion electrolytic solution was obtained.
- the obtained electrolyte was centrifuged at 6900 ⁇ g for 15 minutes to obtain a supernatant (transparent ND-dispersed electrolyte).
- the median diameter (particle size D50) of the surface-modified nanodiamond particles obtained in this step was 78.8 nm, and the mass ratio [ND / polyglycerin chain-containing surface modifying group] determined by thermogravimetric analysis was 0.74. ..
- ⁇ Diameter D50> The median diameter (particle size D50) of the ND particles in the ND dispersion electrolytic solution of the example obtained as described above was measured from the particle size distribution of nanodiamonds obtained by the dynamic light scattering method. Specifically, the particle size distribution is measured by a dynamic light scattering method (non-contact backscattering method) using a device manufactured by Malvern (trade name "Zetasizer Nano ZS"). did.
- Example 4 The surface-modified nanodiamonds obtained in Example 4 were subjected to 13 C-NMR analysis. Specifically, the dry powder of surface-modified nanodiamond obtained in Example 4 and hydrophilic ND particles (PG-ND) modified with polyglycerin were converted to heavy DMSO (0.3% tetramethylsilane). After dispersion using an ultrasonic cleaner, 13 C-NMR spectrum measurements were performed using Bruker AVANCE (600 MHz). The measurement was carried out under the conditions of 25 ° C. and 200,000 times of integration.
- PG-ND hydrophilic ND particles
- Example 4 The 13 C-NMR spectrum of the surface-modified nanodiamond obtained in Example 4 is shown in FIG.
- a peak a near 13 to 14 ppm and a peak b near 19 to 20 ppm and 31 to 32 ppm were observed, respectively.
- These peaks are derived from the hydroxyl groups on the polyglycerin chain being sealed with an alkyl chain.
- Example 5 About the surface-modified nanodiamond (dry solid content) obtained in Example 4, 50 g of a solution (1M) in which LiPF 6 was dissolved in a mixed solvent (EC: EMC, 3: 7v / v%) (manufactured by Kishida Chemical Co., Ltd.). An electrolytic solution was prepared by adding nanodiamond so that the component concentration of nanodiamond was 500 ppm. For comparison, the above solution to which nanodiamond was not added was used as an electrolytic solution. As the evaluation single-layer laminated cell, NMC111 was used for the positive electrode and graphitized MCMB was used for the negative electrode.
- the initial capacity of the positive electrode NMC is 150 mAh / g class (initial efficiency 85%), and the initial capacity of the negative electrode MCMB is 340 mAh / g class (initial efficiency 93%).
- electrodes punched into standard sizes of 30 mm ⁇ 50 mm for the positive electrode and 32 mm ⁇ 52 mm for the negative electrode were vacuum-dried at 170 ° C. for 10 hours for both the positive electrode and the negative electrode.
- the dried electrodes were opposed to each other via a PE microporous membrane separator vacuum-dried at 70 ° C. for 3 hours, inserted into an Al laminate, each electrolytic solution for evaluation was injected, and then vacuum sealing was performed.
- the capacity changes of the single-layer laminated cell thus produced were compared while gradually increasing the measurement rate based on 0 ° C. and a negative electrode load factor of 90% (charging voltage 4.3 V).
- Graphs plotting the capacity retention rate with respect to the number of cycles are shown in FIGS. 2 and 3.
- the battery using the ND dispersion electrolytic solution maintains a capacity of 50% after the 220 cycle test.
- the capacity of the battery to which ND was not added decreased to 36%.
- [Appendix 2] The electrolytic solution for a battery according to Appendix 1, which contains a nanocarbon material at a ratio of 10 to 100,000 mass ppm (preferably 10 to 10000 mass ppm, more preferably 100 to 1000 mass ppm).
- Nanocarbon materials include nanodiamond, fullerene, graphene, graphene oxide, nanographite, carbon nanotube, carbon nanofilament, onion-like carbon, diamond-like carbon, amorphous carbon, carbon black, carbon nanohorn, and carbon nanocoil.
- the electrolytic solution for a battery according to Appendix 1 or 2 which uses one or more selected from the group consisting of.
- nanodiamonds having a primary particle diameter of 10 nm or less (for example, 1 to 10 nm) (preferably 8 nm or less (for example, 2 to 8 nm), more preferably 6 nm or less (for example, 4 to 6 nm)).
- the electrolytic solution for a battery according to any one of Appendix 1 to 4 which uses diamond particles.
- Appendix 6 Any one of Appendix 1 to 5 using surface-modified nanodiamonds containing nanodiamond particles and a surface-modifying group having a polyoxyalkylene chain that surface-modifies the nanodiamond particles as the nanocarbon material.
- Appendix 7 The electrolytic solution for a battery according to Appendix 6, wherein the polyoxyalkylene chain is one or more selected from the group consisting of a polyethylene glycol chain, a polypropylene glycol chain, and a polybutylene glycol chain.
- [Appendix 8] The electrolytic solution for a battery according to Appendix 6 or 7, wherein the surface-modified nanodiamond has a structure in which the end of a polyoxyalkylene chain is sealed with an aliphatic hydrocarbon group.
- [Appendix 9] The electrolytic solution for a battery according to Appendix 8, wherein the aliphatic hydrocarbon group is an aliphatic hydrocarbon group having 1 to 10 carbon atoms (preferably an aliphatic hydrocarbon group having 1 to 6 carbon atoms).
- [Appendix 10] The electrolytic solution for a battery according to Appendix 8 or 9, wherein the aliphatic hydrocarbon group is a linear or branched alkyl group.
- the polyoxyalkylene chain has a number average molecular weight as a polyoxyalkylene, preferably 100 or more (for example, 100 to 50,000) (preferably 200 or more (for example, 200 to 20000), and more preferably 500 or more. (For example, 1000 to 10000), more preferably 1000 or more (for example, 1000 to 6000)).
- Supplementary Note 12 The electrolytic solution for a battery according to any one of Supplementary notes 6 to 11, wherein the polyoxyalkylene chain has a number average degree of polymerization of 2 to 50 (preferably 4 to 45, more preferably 6 to 40). ..
- [Appendix 13] The electrolytic solution for a battery according to any one of Annex 6 to 12, wherein the surface modifying group contains a silicon atom.
- Appendix 14 The battery electrolyte solution according to Appendix 13, wherein the silicon atom is present between the polyoxyalkylene chain and the surface-modified nanodiamond particles.
- Appendix 15 The electrolytic solution for a battery according to Appendix 13 or 14, wherein the silicon atom is present in the surface modifying group as a Si—O bond.
- R 1 is a hydrogen atom or an aliphatic hydrocarbon group (preferably an aliphatic hydrocarbon group having 1 to 10 carbon atoms, more preferably an aliphatic hydrocarbon group having 1 to 6 carbon atoms. Hydrocarbon group) is shown.
- R 2 represents an alkylene group (preferably an alkylene group having 1 to 4 carbon atoms).
- R 3 and R 4 are the same or different, and represent a hydrogen atom, an aliphatic hydrocarbon group having 1 to 3 carbon atoms, or a group represented by the following formula (a).
- n is the number average degree of polymerization of the oxyalkylene chain (—OR 2 ), and represents a value of 2 or more (preferably 2 to 50, more preferably 4 to 45, still more preferably 6 to 40).
- X indicates a single bond or a linking group. The wavy bond in the formula binds to the surface of the nanodiamond particles.
- RA represents [-X- (OR 2 ) n-OR 1 ]
- X, R 1 , R 2 , and n in RA are the above formulas (A), respectively. It is the same as that in 1) to (3).
- R 5 and R 6 represent hydrogen atoms or aliphatic hydrocarbon groups having 1 to 3 carbon atoms, which are the same or different from each other. l and m indicate the same or different integers of 0 or more. The bond extending to the left from the silicon atom bonds to the oxygen atom. The wavy bond in the formula binds to the surface of the nanodiamond particles. ]] [Appendix 19] The battery electrolyte solution according to Appendix 18, wherein X contains a urethane bond.
- the nanocarbon material has nanodiamond particles and a polyglycerin chain that surface-modifies the nanodiamond particles, and at least a part of the hydroxyl group hydrogen atoms in the polyglycerin chain are monovalent organic.
- R 8 is a monovalent organic group (preferably a substituted or unsubstituted hydrocarbon group, more preferably an alkyl group, still more preferably an alkyl group having 1 to 18 carbon atoms, still more preferably an alkyl group having 1 to 6 carbon atoms. More preferably, it exhibits an alkyl group having 1 to 4 carbon atoms, particularly preferably an ethyl group or a butyl group), and the atom bonded to X 1 is a carbon atom.
- R 9 represents a monovalent organic group.
- [-OR 10 ] indicates the end of the polyglycerin chain, and R 10 indicates a hydrogen atom or a monovalent organic group. However, when R 10 is a hydrogen atom, q indicates a value exceeding 0.
- the oxygen atom bonded to Y in the formula (4-2) may be an oxygen atom in the structural unit with p, or an oxygen atom in the structural unit with q, and may be an oxygen atom in the structural unit with q.
- the carbon atom in the formula (4-2) that bonds with OR 10 in 4-2) may be the carbon atom in the structural unit with p, or the carbon atom in the structural unit with q. There may be.
- [Appendix 24] The electrolytic solution for a battery according to Appendix 23, wherein [q / (p + q)] in the above formula (4-2) is 0.6 or more (preferably 0.8 to 1.0).
- Appendix 31 At least one organic dispersion of the surface-modified nanodiamond having a solubility parameter ( ⁇ ) of Hildebrandt of 10 (cal / cm 3 ) 1/2 or less so that the content ratio is 0.1% by mass.
- Appendix 32 The electrolytic solution for a battery according to any one of Annex 4 to 31, wherein the nanodiamond particles include detonation nanodiamonds (preferably air-cooled detonation nanodiamonds).
- [Supplementary Note 33] The electrolytic solution for a battery according to any one of Supplementary notes 1 to 32, which comprises nanodiamond particles as the nanocarbon material and a dispersant.
- Appendix 34 The electrolytic solution for a battery according to Appendix 33, wherein the dispersant has a mass average molecular weight of 500 or more (preferably 650 or more, more preferably 950 or more).
- Appendix 35 The electrolytic solution for a battery according to Appendix 33 or 34, wherein the mass average molecular weight of the dispersant is 20000 or less (preferably 10000 or less).
- the dispersant is a compound having a structure derived from polyalkylene glycol monoalkyl ether (particularly, a structure derived from polyethylene glycol monoalkyl ether or a structure derived from polypropylene glycol monoalkyl ether), a compound having a carbamate structure, and the like.
- the content ratio of the dispersant in the battery electrolyte is 0.1 to 10000 parts by mass (preferably 10 to 1000 parts by mass, more preferably 50 parts by mass) with respect to 100 parts by mass of the total amount of the nanocarbon material.
- the electrolytic solution for a battery according to any one of Appendix 33 to 38 (up to 500 parts by mass).
- Appendix 40 As the dispersion medium, one or more selected from the group consisting of non-aqueous solvents (preferably carbonate compounds, cyclic carboxylic acid esters, cyclic ethers, and cyclic compounds, more preferably saturated cyclic carbonates, chain carbonates, etc. And one or more selected from the group consisting of cyclic carboxylic acid esters, more preferably one or more selected from the group consisting of ethylene carbonate, propylene carbonate, ⁇ -butyrolactone, dimethyl carbonate, diethyl carbonate, and ethyl methyl carbonate).
- the electrolytes preferably lithium salts, more preferably LiPF 6 , LiBF 4 , LiN (SO 2 CF 3 ) 2 , LiNC F 3 SO 3 , and LiN (SO 2 F) used in the non-aqueous electrolyte solution.
- Appendix 42 Any one of Appendix 1 to 41, wherein the content ratio of the electrolyte in the battery electrolyte is 5 to 30% by mass (preferably 8 to 18% by mass, more preferably 10 to 15% by mass). Battery electrolyte according to.
- [Appendix 43] The battery electrolyte solution according to any one of Supplementary notes 1 to 42, wherein the content ratio of the dispersion medium in the battery electrolyte solution is 75 to 94% by mass.
- the content ratio of the non-aqueous solvent in the total amount of the dispersion medium is 60% by mass or more (preferably 70% by mass or more, more preferably 80% by mass or more, still more preferably 90% by mass or more).
- [Appendix 45] The battery electrolyte according to any one of Supplements 1 to 44, wherein the haze value of the battery electrolyte is 5 or less (preferably 3 or less, more preferably 1 or less).
- the viscosity of the battery electrolyte solution at 25 ° C. is 0.1 to 100 mPa ⁇ s (preferably 0.5 to 50 mPa ⁇ s, more preferably 1.0 to 30 mPa ⁇ s).
- Appendix 47 A lithium ion battery using the battery electrolyte solution according to any one of Supplementary notes 1 to 46.
Abstract
Description
-X1-R8 (4-1)
[上記式(4-1)中、X1は、単結合、-C(=O)-、又は-C(=O)-NH-を示す。R8は一価の有機基を示し、X1と結合する原子が炭素原子である。]
-Y-(C3H6O2)p-(C3H5O2R9)q-OR10 (4-2)
-OCH2-CHOH-CH2- (5)
-OCH(CH2OH)-CH2- (6)
-OCH2-CH(CH2OH)- (7)
-OCH2-CH(OR9)-CH2- (8)
-OCH(CH2OR9)-CH2- (9)
-OCH2-CH(CH2OR9)- (10)
下記工程を経て、表面修飾ND粒子及び電解液を製造した。
まず、爆轟法によるNDの生成工程を行った。本工程では、まず、成形された爆薬に電気雷管が装着されたものを爆轟用の耐圧性容器の内部に設置して容器を密閉した。容器は鉄製で、容器の容積は15m3である。爆薬としては、TNTとRDXとの混合物0.50kgを使用した。この爆薬におけるTNTとRDXの質量比(TNT/RDX)は、50/50である。次に、電気雷管を起爆させ、容器内で爆薬を爆轟させた(爆轟法によるNDの生成)。次に、室温での24時間の放置により、容器及びその内部を降温させた。この放冷の後、容器の内壁に付着しているND粗生成物(上記爆轟法で生成したND粒子の凝着体と煤を含む)をヘラで掻き取る作業を行い、ND粗生成物を回収した。
ND粒子とシランカップリング剤の反応において、実施例1で用いたポリオキシアルキレン鎖含有シランカップリング剤8gの代わりに、ヘキシルトリメトキシシラン1.2gを用いたこと、及び、メチルエチルケトン15ccの代わりにメチルイソブチルケトン15ccを用いたこと以外は実施例1と同様にして、15時間超音波処理してND粒子とアルキル鎖含有シランカップリング剤(ヘキシルトリメトキシシラン)を反応させた。15時間超音波処理後、得られた体積比で2倍量のメタノールを加えた後、13000×g、15分の条件で遠心分離を行い、上澄み液を取り除いた。得られた沈殿物にメチルイソブチルケトン15ccを加えて、再び超音波分散機(株式会社エスエムテー製、型式「UH-600S」)を用い、超音波分散機の振動子の先端を反応容器内の溶液に浸けた状態で30分間超音波処理して、未反応シランカップリング剤を取り除いた分散液を得た。この工程で得られた表面修飾ND粒子のメディアン径(粒径D50)は、25nmであった。続いて、別途調製したND固形分に対して100倍質量の分散剤「BYK-9077」(ビックケミー・ジャパン社製)を溶解させたメチルイソブチルケトン15ccと、上記ND分散液を混ぜ合わせた後、エバポレーターによる減圧蒸留でメチルイソブチルケトンを留去し、さらに真空乾燥機内で100℃24時間静置することで乾燥固形分を得た。バイアル瓶中にこのND含有量が0.05質量%になるように乾燥固形分に対して1MのLiBF4を溶かしたEC:DEC(1:1v/v%)(キシダ化学株式会社製)を加え、超音波洗浄器で30分間攪拌することで透明なND分散電解液を得た。20日静置後に得られた上澄み液に対して6900×g、15分の条件で遠心分離を行い、上澄み液(透明なND分散電解液)を得た。この工程で得られた表面修飾ND粒子のメディアン径(粒径D50)は、35.8nmであった。
実施例1で乾燥工程を経て得られた乾燥粉体(ND凝着体)4.5gについて、ガス雰囲気炉(商品名「ガス雰囲気チューブ炉 KTF045N1」、光洋サーモシステム株式会社製)の炉心管内に静置し、炉心管に窒素ガスを流速1L/分で30分間通流させ続けた後、通流ガスを窒素から酸素と窒素との混合ガスへと切り替えて当該混合ガスを流速1L/分で炉心管に通流させ続けた。混合ガス中の酸素濃度は4体積%である。混合ガスへの切り替えの後、炉内を加熱設定温度である400℃まで昇温させた。昇温速度については、加熱設定温度より20℃低い380℃までは10℃/分とし、その後の380℃から400℃までは1℃/分とした。そして、炉内の温度条件を400℃に維持しつつ、炉内のND粉体について酸素酸化処理を行った。処理時間は3時間とした。
上記で得られたND粒子水分散液を、エバポレーターを使用して乾燥させ、黒色の乾燥粉体を得た。得られた乾燥粉体(100mg)を、ガラス製反応器に入れた12mLのグリシドール中に添加し、超音波洗浄器(商品名「BRANSON2510」、マーシャルサイエンティフィック社製)にて、室温で2時間、超音波処理して溶解させた。これを窒素雰囲気下で撹拌しつつ、140℃で20時間反応させた。反応混合液を冷却後、120mLのメタノールを加え、超音波処理した後、50400×gで2時間遠心分離し、沈殿物を得た。この沈殿物に対して、120mLのメタノールを加え、同様に洗浄-遠心分離工程を5回繰り返し、最後に沈殿物に対して透析膜(Spectra/Prodialysis membrane, MWCO: 12-14 kDa)を用いて純水透析を行い、残留メタノールを水に置換して凍結乾燥し、ポリグリセリンで修飾された親水性ND粒子(PG-ND粒子)の灰色粉体を得た。TG-DTA熱分析により、ND粒子と表面修飾基の比率を測定した結果、ND粒子:表面修飾基=1:0.7であった。
実施例3の上記修飾化工程において、イソシアン酸メチル4gの代わりに、イソシアン酸ブチル4gを用いたこと、及び、1MのLiBF4を溶かしたEC:DEC(1:1v/v%)(キシダ化学製)の代わりに1MのLiPF6を溶かしたEC:DEC(1:1v/v%)(キシダ化学製)を用いたこと以外は実施例3と同様にして、超音波洗浄器で30分間撹拌することで透明なND分散電解液を得た。得られた電解液に対して6900×g、15分の条件で遠心分離を行い、上澄み液(透明なND分散電解液)を得た。この工程で得られた表面修飾ナノダイヤモンド粒子のメディアン径(粒径D50)は78.8nm、熱重量分析により求められる質量比[ND/ポリグリセリン鎖含有表面修飾基]は0.74であった。
上述のようにして得られた実施例のND分散電解液におけるND粒子のメディアン径(粒径D50)は、動的光散乱法によって得られたナノダイヤモンドの粒度分布から測定した。上記粒度分布は、具体的には、Malvern社製の装置(商品名「ゼータサイザー ナノZS」)を使用して、ナノダイヤモンドの粒度分布を動的光散乱法(非接触後方散乱法)によって測定した。
TG/DTA(熱重量測定・示差熱分析)装置(商品名「EXSTAR6300」、エスアイアイナノテクノロジー社製)を用い、試料(約3mg)を、空気雰囲気下、昇温速度20℃/分にて加熱して重量減少を測定した。なお、基準物質には、アルミナを用いた。
実施例4で得られた表面修飾ナノダイヤモンドについて13C-NMR分析を行った。具体的には、実施例4で得られた表面修飾ナノダイヤモンドの乾燥粉体とポリグリセリンで修飾された親水性ND粒子(PG-ND)について、重DMSO(0.3%テトラメチルシラン)に超音波洗浄機を使って分散させた後に、Brucker AVANCE(600MHz)を用いて13C-NMRスペクトル測定を行った。なお、測定は25℃、積算回数200000回の条件で実施した。
実施例4で得られた表面修飾ナノダイヤモンド(乾燥固形分)について、LiPF6を混合溶媒(EC:EMC、3:7v/v%)(キシダ化学株式会社製)に溶かした溶液(1M)50gに対してナノダイヤモンドの成分濃度が500ppmとなるように添加し、電解液を作製した。また、比較用としてナノダイヤモンド未添加の上記溶液を電解液とした。評価用単層ラミネートセルとして、正極にNMC111と負極に黒鉛化MCMBを使用した。正極NMC初期容量は150mAh/g級(初期効率85%)、負極MCMB初期容量は340mAh/g級(初期効率93%)である。セルの試作は、正極30mm×50mm、負極32mm×52mmの定型サイズにそれぞれ打ち抜いた電極を正極、負極ともに170℃で10時間の条件にて真空乾燥したものを使用した。乾燥した電極を70℃3時間の条件で真空乾燥したPE微多孔膜セパレータを介して対向させ、Alラミネート内に挿入し、評価用の各電解液を注入後、真空シールを実施した。このように作製した単層ラミネートセルについて、0℃、負極負荷率90%(充電電圧4.3V)を基本として測定レートを徐々に増加させつつ容量変化を比較した。サイクル数に対する容量保持率をプロットしたグラフを図2および図3に示す。図2に示されるように、ND分散電解液を使用した電池は220サイクル試験後の容量は50%を維持している。一方、図3に示されるように、NDを添加していない電池の容量は36%まで低下した。
[付記1]分散媒と、前記分散媒に溶解している電解質と、前記分散媒中に平均分散粒子径500nm以下(好ましくは400nm以下、より好ましくは300nm以下、さらに好ましくは200nm以下、特に好ましくは150nm以下)で分散しているナノ炭素材料とを含む電池用電解液。
[付記2]ナノ炭素材料を、10~100000質量ppm(好ましくは10~10000質量ppm、より好ましくは100~1000質量ppm)の割合で含有する付記1に記載の電池用電解液。
[付記3]ナノ炭素材料として、ナノダイヤモンド、フラーレン、グラフェン、酸化グラフェン、ナノグラファイト、カーボンナノチューブ、カーボンナノフィラメント、オニオンライクカーボン、ダイヤモンドライクカーボン、アモルファスカーボン、カーボンブラック、カーボンナノホーン、及びカーボンナノコイルからなる群より選択される一種以上を用いた付記1又は2に記載の電池用電解液。
[付記4]ナノ炭素材料はナノダイヤモンド粒子を含む付記1~3のいずれか1つに記載の電池用電解液。
[付記5]ナノ炭素材料として、一次粒子径が10nm以下(例えば、1~10nm)(好ましくは8nm以下(例えば、2~8nm)、より好ましくは6nm以下(例えば、4~6nm))のナノダイヤモンド粒子を用いた付記1~4のいずれか1つに記載の電池用電解液。
[付記6]ナノ炭素材料として、ナノダイヤモンド粒子と、前記ナノダイヤモンド粒子を表面修飾するポリオキシアルキレン鎖を有する表面修飾基とを含む表面修飾ナノダイヤモンドを用いた付記1~5のいずれか1つに記載の電池用電解液。
[付記7]前記ポリオキシアルキレン鎖として、ポリエチレングリコール鎖、ポリプロピレングリコール鎖、及びポリブチレングリコール鎖からなる群より選択される一種以上を用いた付記6に記載の電池用電解液。
[付記8]前記表面修飾ナノダイヤモンドが、ポリオキシアルキレン鎖の末端が脂肪族炭化水素基で封止された構造を有する付記6又は7に記載の電池用電解液。
[付記9]前記脂肪族炭化水素基は炭素数1~10の脂肪族炭化水素基(好ましくは炭素数1~6の脂肪族炭化水素基)である付記8に記載の電池用電解液。
[付記10]前記脂肪族炭化水素基は直鎖状又は分岐鎖状アルキル基である付記8又は9に記載の電池用電解液。
[付記12]前記ポリオキシアルキレン鎖の数平均重合度は2~50(好ましくは4~45、より好ましくは6~40)である付記6~11のいずれか1つに記載の電池用電解液。
[付記13]前記表面修飾基がケイ素原子を含む付記6~12のいずれか1つに記載の電池用電解液。
[付記14]前記ケイ素原子は、前記ポリオキシアルキレン鎖と表面修飾されるナノダイヤモンド粒子との間に存在する付記13に記載の電池用電解液。
[付記15]前記ケイ素原子は、Si-O結合として前記表面修飾基に存在する付記13又は14に記載の電池用電解液。
[付記16]前記表面修飾ナノダイヤモンド、前記表面修飾基におけるケイ素原子が酸素原子を介して前記ナノダイヤモンド粒子と結合した構造を有する付記13~15のいずれか1つに記載の電池用電解液。
[付記17]前記表面修飾基中、前記ケイ素原子と前記ポリオキシアルキレン鎖とはウレタン結合を介して結合している、付記13~16のいずれか1つに記載の電池用電解液。
[付記18]前記表面修飾基は、下記式(1)で表される基、下記式(2)で表される基、及び下記式(3)で表される基からなる群より選択される1以上の基である、付記6~17のいずれか1つに記載の電池用電解液。
[付記19]前記Xはウレタン結合を含む付記18に記載の電池用電解液。
[付記20]前記Xは二価の炭化水素基(特に、直鎖又は分岐鎖状のアルキレン基)とウレタン結合が連結した基(好ましくは、-(CH2)k-NH-C(=O)-(左端のCはケイ素原子に、右端のCはポリオキシアルキレン鎖中のOにそれぞれ結合)であり、前記式中、kは1~18の整数(好ましくは1~6の整数、より好ましくは1~3の整数)を示す)である、付記19に記載の電池用電解液。
[付記22]前記一価の有機基は下記式(4-1)で表される基である付記21に記載の電池用電解液。
-X1-R8 (4-1)
[前記式(4-1)中、X1は、単結合、-C(=O)-、又は-C(=O)-NH-を示す。R8は一価の有機基(好ましくは置換又は無置換の炭化水素基、より好ましくはアルキル基、さらに好ましくは炭素数1~18のアルキル基、さらに好ましくは炭素数1~6のアルキル基、さらに好ましくは炭素数1~4のアルキル基、特に好ましくはエチル基、ブチル基)を示し、X1と結合する原子が炭素原子である。]
[付記23]前記表面修飾基が下記式(4-2)で表されるポリグリセリン鎖含有表面修飾基である、付記21又は22に記載の電池用電解液。
-Y-(C3H6O2)p-(C3H5O2R9)q-OR10 (4-2)
[式(4-2)中、p及びqは、それぞれ、括弧内の繰り返し単位の平均重合度を示し、pは0以上の値、qは0以上の値、p+qは1以上(好ましくは1~100、より好ましくは2~40、さらに好ましくは3~30)の値である。R9は、一価の有機基を示す。[-OR10]は、ポリグリセリン鎖の末端を示し、R10は水素原子又は一価の有機基を示す。但し、R10が水素原子である場合、qは0を超える値を示す。Yは、単結合又は二価の基(好ましくは、単結合、-NH-、-O-、-C(=O)O-、-PH(=O)O-、-S-、より好ましくは単結合)を示し、Yから左に伸びる結合手はナノダイヤモンド粒子に結合する。式(4-2)中のYと結合する酸素原子は、pが付された構成単位における酸素原子であってもよく、qが付された構成単位における酸素原子であってもよく、式(4-2)中のOR10と結合する式(4-2)中の炭素原子は、pが付された構成単位における炭素原子であってもよく、qが付された構成単位における炭素原子であってもよい。]
[付記24]前記式(4-2)における[q/(p+q)]は0.6以上(好ましくは0.8~1.0)である、付記23に記載の電池用電解液。
[付記25]前記表面修飾ナノダイヤモンドにおける、前記表面修飾基に対するナノダイヤモンド粒子の質量比[ナノダイヤモンド粒子/表面修飾基]が、0.5~1.0(好ましくは0.6~0.8)である、付記21~24のいずれか1つに記載の電池用電解液。
[付記26]前記表面修飾ナノダイヤモンドは、FT-IRスペクトルにおいて、1510~1540cm-1付近と1700~1730cm-1付近とにそれぞれ吸収ピークを有する、付記21~25のいずれか1つに記載の電池用電解液。
[付記27]前記表面修飾ナノダイヤモンドは、FT-IRスペクトルにおいて、1730~1750cm-1付近に吸収ピークを有する、付記21~25のいずれか1つに記載の電池用電解液。
[付記28]前記表面修飾ナノダイヤモンドは、13C-NMRスペクトルにおいて、60~85ppm付近にピークを有する、付記21~27のいずれか1つに記載の電池用電解液。
[付記29]前記表面修飾ナノダイヤモンドは、13C-NMRスペクトルにおいて、140~200ppm付近にピークを有する、付記21~28のいずれか1つに記載の電池用電解液。
[付記30]前記表面修飾ナノダイヤモンドは、13C-NMRスペクトルにおいて、5~45ppm付近にピークを有する、付記21~29のいずれか1つに記載の電池用電解液。
[付記32]前記ナノダイヤモンド粒子は爆轟法ナノダイヤモンド(好ましくは空冷式爆轟法ナノダイヤモンド)を含む付記4~31のいずれか1つに記載の電池用電解液。
[付記33]前記ナノ炭素材料としてのナノダイヤモンド粒子と、分散剤とを含む、付記1~32のいずれか1つに記載の電池用電解液。
[付記34]前記分散剤の質量平均分子量は500以上(好ましくは650以上、より好ましくは950以上)である付記33に記載の電池用電解液。
[付記35]前記分散剤の質量平均分子量は20000以下(好ましくは10000以下)である付記33又は34に記載の電池用電解液。
[付記36]前記分散剤のアミン価は15mgKOH/g以上(好ましくは18mgKOH/g以上、より好ましくは20mgKOH/g以上、さらに好ましくは30mgKOH/g以上)である付記33~35のいずれか1つに記載の電池用電解液。
[付記37]前記分散剤のアミン価は100mgKOH/g以下(好ましくは90mgKOH/g以下、より好ましくは60mgKOH/g以下)である付記33~36のいずれか1つに記載の電池用電解液。
[付記38]前記分散剤は、ポリアルキレングリコールモノアルキルエーテル由来の構造(特に、ポリエチレングリコールモノアルキルエーテル由来の構造、又はポリプロピレングリコールモノアルキルエーテル由来の構造)を有する化合物、カーバメート構造を有する化合物、又はポリカプロラクトン由来の構造を有する化合物を含む付記33~37のいずれか1つに記載の電池用電解液。
[付記39]前記電池用電解液中の分散剤の含有割合は、ナノ炭素材料の総量100質量部に対して、0.1~10000質量部(好ましくは10~1000質量部、より好ましくは50~500質量部)である付記33~38のいずれか1つに記載の電池用電解液。
[付記40]分散媒として、非水系溶媒(好ましくはカーボネート化合物、環状カルボン酸エステル、環状エーテル、及びスルホン系化合物からなる群より選択される一種以上、より好ましくは飽和環状カーボネート、鎖状カーボネート、及び環状カルボン酸エステルからなる群より選択される一種以上、さらに好ましくはエチレンカーボネート、プロピレンカーボネート、γ-ブチロラクトン、ジメチルカーボネート、ジエチルカーボネート、及びエチルメチルカーボネートからなる群より選択される一種以上)を用いた付記1~39のいずれか1つに記載の電池用電解液。
[付記42]前記電池用電解液中の電解質の含有割合は5~30質量%(好ましくは8~18質量%、より好ましくは10~15質量%)である付記1~41のいずれか1つに記載の電池用電解液。
[付記43]前記電池用電解液中の分散媒の含有割合は75~94質量%である付記1~42のいずれか1つに記載の電池用電解液。
[付記44]前記分散媒の総量における非水系溶媒の含有割合は60質量%以上(好ましくは70質量%以上、より好ましくは80質量%以上、さらに好ましくは90質量%以上)である付記1~43のいずれか1つに記載の電池用電解液。
[付記45]前記電池用電解液のヘイズ値は5以下(好ましくは3以下、より好ましくは1以下)である付記1~44のいずれか1つに記載の電池用電解液。
[付記46]前記電池用電解液の25℃における粘度は0.1~100mPa・s(好ましくは0.5~50mPa・s、より好ましくは1.0~30mPa・s)である付記1~45のいずれか1つに記載の電池用電解液。
[付記47]付記1~46のいずれか1つに記載の電池用電解液を用いたリチウムイオン電池。
Claims (16)
- 分散媒と、前記分散媒に溶解している電b解質と、前記分散媒中に平均分散粒子径500nm以下で分散しているナノ炭素材料とを含む電池用電解液。
- ナノ炭素材料を、10~100000質量ppmの割合で含有する請求項1に記載の電池用電解液。
- ナノ炭素材料として、ナノダイヤモンド、フラーレン、グラフェン、酸化グラフェン、ナノグラファイト、カーボンナノチューブ、カーボンナノフィラメント、オニオンライクカーボン、ダイヤモンドライクカーボン、アモルファスカーボン、カーボンブラック、カーボンナノホーン、及びカーボンナノコイルからなる群より選択される一種以上を用いた請求項1又は2に記載の電池用電解液。
- ナノ炭素材料として、一次粒子径が10nm以下のナノダイヤモンド粒子を用いた請求項1~3のいずれか1項に記載の電池用電解液。
- ナノ炭素材料として、ナノダイヤモンド粒子と、前記ナノダイヤモンド粒子を表面修飾するポリオキシアルキレン鎖を有する表面修飾基とを含む表面修飾ナノダイヤモンドを用いた請求項1~4のいずれか1項に記載の電池用電解液。
- ポリオキシアルキレン鎖として、ポリエチレングリコール鎖、ポリプロピレングリコール鎖、及びポリブチレングリコール鎖からなる群より選択される一種以上を用いた請求項5に記載の電池用電解液。
- 前記表面修飾ナノダイヤモンドが、ポリオキシアルキレン鎖の末端が脂肪族炭化水素基で封止された構造を有する請求項5又は6に記載の電池用電解液。
- 前記表面修飾基がケイ素原子を含む請求項5~7のいずれか1項に記載の電池用電解液。
- 前記ナノ炭素材料が、ナノダイヤモンド粒子と、前記ナノダイヤモンド粒子を表面修飾する、ポリグリセリン鎖を有し且つ前記ポリグリセリン鎖における少なくとも一部のヒドロキシル基の水素原子が一価の有機基に置換されている表面修飾基とを含む表面修飾ナノダイヤモンドである、請求項8に記載の電池用電解液。
- 前記表面修飾ナノダイヤモンドは、FT-IRスペクトルにおいて、1510~1540cm-1付近と1700~1730cm-1付近とにそれぞれ吸収ピークを有する、請求項9に記載の電池用電解液。
- 前記表面修飾ナノダイヤモンドは、FT-IRスペクトルにおいて、1730~1750cm-1付近に吸収ピークを有する、請求項9に記載の電池用電解液。
- 前記表面修飾ナノダイヤモンドは、13C-NMRスペクトルにおいて、60~85ppm付近と140~200ppm付近とにそれぞれピークを有する、請求項9~11のいずれか1項に記載の電池用電解液。
- 前記ナノ炭素材料としてのナノダイヤモンド粒子と、質量平均分子量が500以上であり、且つアミン価が15mgKOH/g以上である分散剤とを含む、請求項1~4のいずれか1項に記載の電池用電解液。
- 分散媒として、エチレンカーボネート、プロピレンカーボネート、γ-ブチロラクトン、ジメチルカーボネート、ジエチルカーボネート、及びエチルメチルカーボネートからなる群より選択される一種以上を用いた請求項1~13のいずれか1項に記載の電池用電解液。
- 電解質として、LiPF6、LiBF4、LiN(SO2CF3)2、LiNCF3SO3、及びLiN(SO2F)2からなる群より選択される一種以上を用いた請求項1~14のいずれか1項に記載の電池用電解液。
- 請求項1~15のいずれか1項に記載の電池用電解液を用いたリチウムイオン電池。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021524878A JPWO2020246501A1 (ja) | 2019-06-05 | 2020-06-03 | |
| KR1020217043298A KR20220015470A (ko) | 2019-06-05 | 2020-06-03 | 전지용 전해액 및 리튬 이온 전지 |
| US17/612,021 US20220263134A1 (en) | 2019-06-05 | 2020-06-03 | Battery electrolytic solution and lithium ion battery |
| CN202080041626.0A CN113924678A (zh) | 2019-06-05 | 2020-06-03 | 电池用电解液及锂离子电池 |
| EP20819563.6A EP3982446A4 (en) | 2019-06-05 | 2020-06-03 | BATTERY ELECTROLYTIC SOLUTION AND LITHIUM-ION BATTERY |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2019105066 | 2019-06-05 | ||
| JP2019-105066 | 2019-06-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020246501A1 true WO2020246501A1 (ja) | 2020-12-10 |
Family
ID=73653204
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2020/021926 WO2020246501A1 (ja) | 2019-06-05 | 2020-06-03 | 電池用電解液及びリチウムイオン電池 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20220263134A1 (ja) |
| EP (1) | EP3982446A4 (ja) |
| JP (1) | JPWO2020246501A1 (ja) |
| KR (1) | KR20220015470A (ja) |
| CN (1) | CN113924678A (ja) |
| TW (1) | TWI729840B (ja) |
| WO (1) | WO2020246501A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114156482A (zh) * | 2021-12-02 | 2022-03-08 | 吉林大学 | 一种纳米金刚石电解液和纳米金刚石固体电解质界面的制备方法 |
| WO2023162595A1 (ja) * | 2022-02-28 | 2023-08-31 | 株式会社ダイセル | 表面修飾微粒子、及びその製造方法 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114583261B (zh) * | 2022-03-10 | 2023-11-10 | 山东天润新能源材料有限公司 | 一种含氧化石墨烯的钠离子二次电池电解液的制备方法 |
| CN115395001A (zh) * | 2022-10-11 | 2022-11-25 | 吉林大学 | 一种有效提升钠离子电池性能的方法 |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000340233A (ja) * | 1999-05-26 | 2000-12-08 | Tokai Carbon Co Ltd | 鉛蓄電池用添加剤 |
| JP2008074639A (ja) * | 2006-09-19 | 2008-04-03 | Toppan Printing Co Ltd | スルホン酸基導入無定形炭素含有組成物 |
| JP2009224141A (ja) * | 2008-03-14 | 2009-10-01 | Toyota Central R&D Labs Inc | スラリー利用型二次電池 |
| JP2010202458A (ja) * | 2009-03-03 | 2010-09-16 | Daicel Chem Ind Ltd | 表面修飾ナノダイヤモンド及びその製造法 |
| JP2010248023A (ja) | 2009-04-13 | 2010-11-04 | Daicel Chem Ind Ltd | 表面修飾ナノダイヤモンド及びその製造法 |
| JP2012082103A (ja) | 2010-10-12 | 2012-04-26 | Shiga Univ Of Medical Science | サイズ制御された表面修飾ナノダイヤモンド |
| WO2017203763A1 (ja) * | 2016-05-23 | 2017-11-30 | 株式会社ダイセル | ナノダイヤモンド有機溶媒分散液製造方法およびナノダイヤモンド有機溶媒分散液 |
| JP2018526531A (ja) * | 2015-07-06 | 2018-09-13 | カルボデオン リミティド オサケユイチア | 金属コーティング及びその製造方法 |
| WO2018186382A1 (ja) * | 2017-04-07 | 2018-10-11 | 株式会社ダイセル | 表面修飾ナノダイヤモンド、表面修飾ナノダイヤモンド分散液、及び樹脂分散体 |
| JP2019105066A (ja) | 2017-12-12 | 2019-06-27 | 三菱重工機械システム株式会社 | 縦列型機械式駐車装置及び縦列型機械式駐車装置の制御方法 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1440087A (zh) * | 2003-03-03 | 2003-09-03 | 王祥西 | 铅酸蓄电池纳米碳活化剂 |
| TW200810175A (en) * | 2006-08-11 | 2008-02-16 | Advanced Nanopower Inc | Electrolyte containing carbon nano tube |
| US8486562B2 (en) * | 2009-02-25 | 2013-07-16 | Applied Materials, Inc. | Thin film electrochemical energy storage device with three-dimensional anodic structure |
| TW201034276A (en) * | 2009-02-09 | 2010-09-16 | Applied Materials Inc | Mesoporous carbon material for energy storage |
| CN101924198B (zh) * | 2010-06-12 | 2011-12-07 | 河北金力新能源材料科技有限公司 | 一种锂离子电池正极材料的制备方法 |
| CN104218256A (zh) * | 2013-05-29 | 2014-12-17 | 中国科学院苏州纳米技术与纳米仿生研究所 | 锂离子电池的高电压电解液添加剂、电解液及其制备方法、锂离子二次电池 |
| CN104701029A (zh) * | 2015-01-06 | 2015-06-10 | 宁波南车新能源科技有限公司 | 一种含无机纳米颗粒的超级电容器有机电解液 |
| WO2017204028A1 (ja) * | 2016-05-23 | 2017-11-30 | 富士フイルム株式会社 | 固体電解質組成物、固体電解質含有シートおよび全固体二次電池ならびに固体電解質含有シートおよび全固体二次電池の製造方法 |
| EP3643679A4 (en) * | 2017-06-19 | 2021-04-07 | Daicel Corporation | SURFACE MODIFIED NANODIAMANT, LIQUID DISPERSION CONTAINING SURFACE MODIFIED NANODIAMANT, AND RESIN DISPERSION |
| CN107482251A (zh) * | 2017-07-21 | 2017-12-15 | 昆山正国新能源动力电池有限公司 | 高温锂电池电解液及其制备方法 |
| US10873107B2 (en) * | 2017-08-01 | 2020-12-22 | Drexel University | Additives for suppressing dendritic growth in batteries |
-
2020
- 2020-06-03 CN CN202080041626.0A patent/CN113924678A/zh active Pending
- 2020-06-03 US US17/612,021 patent/US20220263134A1/en active Pending
- 2020-06-03 EP EP20819563.6A patent/EP3982446A4/en active Pending
- 2020-06-03 JP JP2021524878A patent/JPWO2020246501A1/ja active Pending
- 2020-06-03 WO PCT/JP2020/021926 patent/WO2020246501A1/ja unknown
- 2020-06-03 KR KR1020217043298A patent/KR20220015470A/ko unknown
- 2020-06-05 TW TW109118958A patent/TWI729840B/zh active
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000340233A (ja) * | 1999-05-26 | 2000-12-08 | Tokai Carbon Co Ltd | 鉛蓄電池用添加剤 |
| JP2008074639A (ja) * | 2006-09-19 | 2008-04-03 | Toppan Printing Co Ltd | スルホン酸基導入無定形炭素含有組成物 |
| JP2009224141A (ja) * | 2008-03-14 | 2009-10-01 | Toyota Central R&D Labs Inc | スラリー利用型二次電池 |
| JP2010202458A (ja) * | 2009-03-03 | 2010-09-16 | Daicel Chem Ind Ltd | 表面修飾ナノダイヤモンド及びその製造法 |
| JP2010248023A (ja) | 2009-04-13 | 2010-11-04 | Daicel Chem Ind Ltd | 表面修飾ナノダイヤモンド及びその製造法 |
| JP2012082103A (ja) | 2010-10-12 | 2012-04-26 | Shiga Univ Of Medical Science | サイズ制御された表面修飾ナノダイヤモンド |
| JP2018526531A (ja) * | 2015-07-06 | 2018-09-13 | カルボデオン リミティド オサケユイチア | 金属コーティング及びその製造方法 |
| WO2017203763A1 (ja) * | 2016-05-23 | 2017-11-30 | 株式会社ダイセル | ナノダイヤモンド有機溶媒分散液製造方法およびナノダイヤモンド有機溶媒分散液 |
| WO2018186382A1 (ja) * | 2017-04-07 | 2018-10-11 | 株式会社ダイセル | 表面修飾ナノダイヤモンド、表面修飾ナノダイヤモンド分散液、及び樹脂分散体 |
| JP2019105066A (ja) | 2017-12-12 | 2019-06-27 | 三菱重工機械システム株式会社 | 縦列型機械式駐車装置及び縦列型機械式駐車装置の制御方法 |
Non-Patent Citations (6)
| Title |
|---|
| CHENG XIN-BING, ZHAO MENG-QIANG, CHEN CHI, PENTECOST AMANDA, MALESKI KATHLEEN, MATHIS TYLER, ZHANG XUE-QIANG, ZHANG QIANG, JIANG J: "Nanodiamonds suppress the growth of lithium dendrites", NATURE COMMUNICATIONS, vol. 8, no. 336, 2017, pages 1 - 9, XP055770309 * |
| E. J. VANDERBERG, J. POLYM. SCI., POLYM. CHEM. ED., vol. 23, 1985, pages 915 |
| G. R. NEWCOME ET AL.: "Dendritic Macromolecules: Concepts, Syntheses, Perspectives", 1996, VCH, WEINHEIM |
| S. R. SANDLER ET AL., J. POLYM. SCI., POLYM. CHEM. ED., vol. 4, 1966, pages 1253 |
| See also references of EP3982446A4 |
| XIN-BING, CHANG ET AL., NATURE COMMUNICATIONS, vol. 8, 2017, pages 337 - 345 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114156482A (zh) * | 2021-12-02 | 2022-03-08 | 吉林大学 | 一种纳米金刚石电解液和纳米金刚石固体电解质界面的制备方法 |
| CN114156482B (zh) * | 2021-12-02 | 2023-11-17 | 吉林大学 | 一种纳米金刚石电解液和纳米金刚石固体电解质界面的制备方法 |
| WO2023162595A1 (ja) * | 2022-02-28 | 2023-08-31 | 株式会社ダイセル | 表面修飾微粒子、及びその製造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2020246501A1 (ja) | 2020-12-10 |
| EP3982446A4 (en) | 2023-07-05 |
| KR20220015470A (ko) | 2022-02-08 |
| TWI729840B (zh) | 2021-06-01 |
| EP3982446A1 (en) | 2022-04-13 |
| TW202105819A (zh) | 2021-02-01 |
| US20220263134A1 (en) | 2022-08-18 |
| CN113924678A (zh) | 2022-01-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2020246501A1 (ja) | 電池用電解液及びリチウムイオン電池 | |
| EP3981740A1 (en) | Surface-modified nanodiamond and method for producing surface-modified nanodiamond | |
| US20240116762A1 (en) | Surface-modified nanodiamonds and method for producing surface-modified nano carbon particles | |
| CN105899522A (zh) | 硅烷基化的环状膦酰胺 | |
| WO2020179370A1 (ja) | 表面修飾ナノダイヤモンド、ナノダイヤモンド分散組成物、及び表面修飾ナノダイヤモンドの製造方法 | |
| JP5301322B2 (ja) | ホスホニウム基含有ゲル状ナノコンポジット、その製造方法及びイオン伝導体 | |
| US20220259048A1 (en) | Nanodiamond dispersion composition | |
| WO2020095581A1 (ja) | ナノダイヤモンド分散組成物 | |
| US11939222B2 (en) | Nanodiamond particle dispersion | |
| WO2023022238A1 (ja) | ナノダイヤモンド水分散液 | |
| JP2024027659A (ja) | リチウム金属電池用電解液、リチウム金属電池、及び電気機器 | |
| KR20230145168A (ko) | 나노 탄소 재료 분산 조성물 | |
| WO2022138986A1 (ja) | ナノダイヤモンド分散組成物の製造方法およびナノダイヤモンド分散組成物 | |
| RU2780325C1 (ru) | Поверхностно-модифицированный наноалмаз, дисперсная композиция наноалмаза и способ производства поверхностно-модифицированного наноалмаза | |
| JP2023131428A (ja) | 表面修飾ナノ炭素粒子及びその製造方法、並びに表面修飾ナノ炭素粒子を含む分散液、複合材料 | |
| WO2022091725A1 (ja) | ナノダイヤモンド分散組成物 | |
| KR20220032577A (ko) | 표면 수식 나노 다이아몬드, 나노 다이아몬드 분산 조성물 및 표면 수식 나노 탄소 입자의 제조 방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20819563 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2021524878 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20217043298 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2020819563 Country of ref document: EP Effective date: 20220105 |