WO2020017629A1 - Htlv-1関連脊髄症(ham)治療又は予防剤、及びhamの治療方法 - Google Patents
Htlv-1関連脊髄症(ham)治療又は予防剤、及びhamの治療方法 Download PDFInfo
- Publication number
- WO2020017629A1 WO2020017629A1 PCT/JP2019/028431 JP2019028431W WO2020017629A1 WO 2020017629 A1 WO2020017629 A1 WO 2020017629A1 JP 2019028431 W JP2019028431 W JP 2019028431W WO 2020017629 A1 WO2020017629 A1 WO 2020017629A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- rgma
- ham
- antibody
- seq
- cells
- Prior art date
Links
- 208000006961 tropical spastic paraparesis Diseases 0.000 title claims abstract description 104
- 238000000034 method Methods 0.000 title claims abstract description 31
- 230000002265 prevention Effects 0.000 title abstract 2
- 239000003112 inhibitor Substances 0.000 claims abstract description 28
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 13
- 230000001225 therapeutic effect Effects 0.000 claims description 5
- 206010028570 Myelopathy Diseases 0.000 claims description 4
- 230000003449 preventive effect Effects 0.000 claims 2
- 241000714260 Human T-lymphotropic virus 1 Species 0.000 claims 1
- 125000003275 alpha amino acid group Chemical group 0.000 description 91
- 210000004027 cell Anatomy 0.000 description 88
- 102100036011 T-cell surface glycoprotein CD4 Human genes 0.000 description 42
- 210000003819 peripheral blood mononuclear cell Anatomy 0.000 description 41
- 210000001744 T-lymphocyte Anatomy 0.000 description 39
- 230000014509 gene expression Effects 0.000 description 32
- 108090000623 proteins and genes Proteins 0.000 description 31
- 108010047041 Complementarity Determining Regions Proteins 0.000 description 22
- 230000000694 effects Effects 0.000 description 22
- 239000012634 fragment Substances 0.000 description 20
- 239000004055 small Interfering RNA Substances 0.000 description 20
- 239000000427 antigen Substances 0.000 description 17
- 102000036639 antigens Human genes 0.000 description 17
- 108091007433 antigens Proteins 0.000 description 17
- 239000002609 medium Substances 0.000 description 17
- YKYOUMDCQGMQQO-UHFFFAOYSA-L cadmium dichloride Chemical compound Cl[Cd]Cl YKYOUMDCQGMQQO-UHFFFAOYSA-L 0.000 description 16
- 210000002569 neuron Anatomy 0.000 description 15
- 108020004414 DNA Proteins 0.000 description 13
- 239000000243 solution Substances 0.000 description 12
- 108010072135 Cell Adhesion Molecule-1 Proteins 0.000 description 11
- 102100024649 Cell adhesion molecule 1 Human genes 0.000 description 11
- 108020004459 Small interfering RNA Proteins 0.000 description 11
- 230000006907 apoptotic process Effects 0.000 description 11
- 102000004169 proteins and genes Human genes 0.000 description 11
- 108091027967 Small hairpin RNA Proteins 0.000 description 10
- 238000004458 analytical method Methods 0.000 description 10
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 9
- 102000017481 Repulsive guidance molecule Human genes 0.000 description 9
- 108050005592 Repulsive guidance molecule Proteins 0.000 description 9
- 241000700605 Viruses Species 0.000 description 9
- 230000001154 acute effect Effects 0.000 description 9
- 238000007792 addition Methods 0.000 description 8
- 150000001413 amino acids Chemical class 0.000 description 8
- 239000002299 complementary DNA Substances 0.000 description 8
- 238000012217 deletion Methods 0.000 description 8
- 230000037430 deletion Effects 0.000 description 8
- OIGNJSKKLXVSLS-VWUMJDOOSA-N prednisolone Chemical compound O=C1C=C[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 OIGNJSKKLXVSLS-VWUMJDOOSA-N 0.000 description 8
- 229960005205 prednisolone Drugs 0.000 description 8
- 238000006467 substitution reaction Methods 0.000 description 8
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 7
- 102100025248 C-X-C motif chemokine 10 Human genes 0.000 description 7
- 101000858088 Homo sapiens C-X-C motif chemokine 10 Proteins 0.000 description 7
- 241000699666 Mus <mouse, genus> Species 0.000 description 7
- 239000000872 buffer Substances 0.000 description 7
- 102000004127 Cytokines Human genes 0.000 description 6
- 108090000695 Cytokines Proteins 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- 239000012980 RPMI-1640 medium Substances 0.000 description 6
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 6
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 6
- -1 infusions Substances 0.000 description 6
- 108090000765 processed proteins & peptides Proteins 0.000 description 6
- 208000024891 symptom Diseases 0.000 description 6
- 241000283707 Capra Species 0.000 description 5
- 238000012413 Fluorescence activated cell sorting analysis Methods 0.000 description 5
- 101100005713 Homo sapiens CD4 gene Proteins 0.000 description 5
- 229910002091 carbon monoxide Inorganic materials 0.000 description 5
- 230000001684 chronic effect Effects 0.000 description 5
- 201000010099 disease Diseases 0.000 description 5
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 5
- 230000006698 induction Effects 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 238000004519 manufacturing process Methods 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 239000013603 viral vector Substances 0.000 description 5
- 241000124008 Mammalia Species 0.000 description 4
- 108091034117 Oligonucleotide Proteins 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 239000000074 antisense oligonucleotide Substances 0.000 description 4
- 238000012230 antisense oligonucleotides Methods 0.000 description 4
- 238000003501 co-culture Methods 0.000 description 4
- 239000012228 culture supernatant Substances 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 239000003814 drug Substances 0.000 description 4
- 238000005516 engineering process Methods 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 238000013467 fragmentation Methods 0.000 description 4
- 238000006062 fragmentation reaction Methods 0.000 description 4
- 238000001802 infusion Methods 0.000 description 4
- 238000003780 insertion Methods 0.000 description 4
- 230000037431 insertion Effects 0.000 description 4
- 239000002502 liposome Substances 0.000 description 4
- 108020004999 messenger RNA Proteins 0.000 description 4
- 208000015122 neurodegenerative disease Diseases 0.000 description 4
- 230000016273 neuron death Effects 0.000 description 4
- 239000002953 phosphate buffered saline Substances 0.000 description 4
- 239000003755 preservative agent Substances 0.000 description 4
- 230000002335 preservative effect Effects 0.000 description 4
- 238000003753 real-time PCR Methods 0.000 description 4
- 102000005962 receptors Human genes 0.000 description 4
- 108020003175 receptors Proteins 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- 230000005353 IP-10 production Effects 0.000 description 3
- 108060003951 Immunoglobulin Proteins 0.000 description 3
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 3
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 3
- 108090000174 Interleukin-10 Proteins 0.000 description 3
- 206010033799 Paralysis Diseases 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 208000030886 Traumatic Brain injury Diseases 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 239000011324 bead Substances 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 210000003169 central nervous system Anatomy 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 229960004926 chlorobutanol Drugs 0.000 description 3
- 210000004408 hybridoma Anatomy 0.000 description 3
- 230000002163 immunogen Effects 0.000 description 3
- 102000018358 immunoglobulin Human genes 0.000 description 3
- 230000001939 inductive effect Effects 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 238000002493 microarray Methods 0.000 description 3
- 201000006417 multiple sclerosis Diseases 0.000 description 3
- 210000005036 nerve Anatomy 0.000 description 3
- 230000004770 neurodegeneration Effects 0.000 description 3
- 230000003472 neutralizing effect Effects 0.000 description 3
- 239000002773 nucleotide Substances 0.000 description 3
- 125000003729 nucleotide group Chemical group 0.000 description 3
- 230000036961 partial effect Effects 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 229940104230 thymidine Drugs 0.000 description 3
- 239000013598 vector Substances 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- 108020004463 18S ribosomal RNA Proteins 0.000 description 2
- 208000016683 Adult T-cell leukemia/lymphoma Diseases 0.000 description 2
- 208000024827 Alzheimer disease Diseases 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 101001046686 Homo sapiens Integrin alpha-M Proteins 0.000 description 2
- 208000023105 Huntington disease Diseases 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 102100022338 Integrin alpha-M Human genes 0.000 description 2
- 108010002350 Interleukin-2 Proteins 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 241000699670 Mus sp. Species 0.000 description 2
- 102100031900 Neogenin Human genes 0.000 description 2
- 108091028043 Nucleic acid sequence Proteins 0.000 description 2
- 241000283973 Oryctolagus cuniculus Species 0.000 description 2
- 229930040373 Paraformaldehyde Natural products 0.000 description 2
- 208000018737 Parkinson disease Diseases 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- 241000700159 Rattus Species 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 208000000389 T-cell leukemia Diseases 0.000 description 2
- 208000028530 T-cell lymphoblastic leukemia/lymphoma Diseases 0.000 description 2
- 230000000996 additive effect Effects 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 201000006966 adult T-cell leukemia Diseases 0.000 description 2
- 229960000686 benzalkonium chloride Drugs 0.000 description 2
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 2
- 239000004327 boric acid Substances 0.000 description 2
- 235000010338 boric acid Nutrition 0.000 description 2
- 230000030833 cell death Effects 0.000 description 2
- 238000010835 comparative analysis Methods 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 238000012258 culturing Methods 0.000 description 2
- 230000016396 cytokine production Effects 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 230000007850 degeneration Effects 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 238000010195 expression analysis Methods 0.000 description 2
- 239000000796 flavoring agent Substances 0.000 description 2
- 239000008103 glucose Substances 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- 230000003053 immunization Effects 0.000 description 2
- 230000002757 inflammatory effect Effects 0.000 description 2
- 230000028709 inflammatory response Effects 0.000 description 2
- 238000002372 labelling Methods 0.000 description 2
- 210000002540 macrophage Anatomy 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 230000035772 mutation Effects 0.000 description 2
- 108010076969 neogenin Proteins 0.000 description 2
- 108020004707 nucleic acids Proteins 0.000 description 2
- 102000039446 nucleic acids Human genes 0.000 description 2
- 150000007523 nucleic acids Chemical class 0.000 description 2
- 239000003002 pH adjusting agent Substances 0.000 description 2
- 208000021090 palsy Diseases 0.000 description 2
- 229920002866 paraformaldehyde Polymers 0.000 description 2
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 2
- 239000002504 physiological saline solution Substances 0.000 description 2
- 102000004196 processed proteins & peptides Human genes 0.000 description 2
- 230000000069 prophylactic effect Effects 0.000 description 2
- 235000013772 propylene glycol Nutrition 0.000 description 2
- 238000011002 quantification Methods 0.000 description 2
- 230000006798 recombination Effects 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 235000010356 sorbitol Nutrition 0.000 description 2
- 230000001148 spastic effect Effects 0.000 description 2
- 210000000278 spinal cord Anatomy 0.000 description 2
- 208000020431 spinal cord injury Diseases 0.000 description 2
- 210000004989 spleen cell Anatomy 0.000 description 2
- 230000002269 spontaneous effect Effects 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 238000010186 staining Methods 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 108010038751 tax Gene Products Proteins 0.000 description 2
- UEUXEKPTXMALOB-UHFFFAOYSA-J tetrasodium;2-[2-[bis(carboxylatomethyl)amino]ethyl-(carboxylatomethyl)amino]acetate Chemical compound [Na+].[Na+].[Na+].[Na+].[O-]C(=O)CN(CC([O-])=O)CCN(CC([O-])=O)CC([O-])=O UEUXEKPTXMALOB-UHFFFAOYSA-J 0.000 description 2
- 238000013518 transcription Methods 0.000 description 2
- 230000035897 transcription Effects 0.000 description 2
- 230000009529 traumatic brain injury Effects 0.000 description 2
- 241000701161 unidentified adenovirus Species 0.000 description 2
- 241001430294 unidentified retrovirus Species 0.000 description 2
- 238000011144 upstream manufacturing Methods 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- NFGXHKASABOEEW-UHFFFAOYSA-N 1-methylethyl 11-methoxy-3,7,11-trimethyl-2,4-dodecadienoate Chemical compound COC(C)(C)CCCC(C)CC=CC(C)=CC(=O)OC(C)C NFGXHKASABOEEW-UHFFFAOYSA-N 0.000 description 1
- SPSPIUSUWPLVKD-UHFFFAOYSA-N 2,3-dibutyl-6-methylphenol Chemical compound CCCCC1=CC=C(C)C(O)=C1CCCC SPSPIUSUWPLVKD-UHFFFAOYSA-N 0.000 description 1
- 238000010600 3H thymidine incorporation assay Methods 0.000 description 1
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 1
- 208000030090 Acute Disease Diseases 0.000 description 1
- 108010088751 Albumins Proteins 0.000 description 1
- 102000009027 Albumins Human genes 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- 102100022548 Beta-hexosaminidase subunit alpha Human genes 0.000 description 1
- 108010049931 Bone Morphogenetic Protein 2 Proteins 0.000 description 1
- 108010049955 Bone Morphogenetic Protein 4 Proteins 0.000 description 1
- 108010007726 Bone Morphogenetic Proteins Proteins 0.000 description 1
- 102000007350 Bone Morphogenetic Proteins Human genes 0.000 description 1
- 102100024506 Bone morphogenetic protein 2 Human genes 0.000 description 1
- 102100024505 Bone morphogenetic protein 4 Human genes 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- 206010010774 Constipation Diseases 0.000 description 1
- 208000016192 Demyelinating disease Diseases 0.000 description 1
- 241000702421 Dependoparvovirus Species 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- 206010012689 Diabetic retinopathy Diseases 0.000 description 1
- 241000991587 Enterovirus C Species 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 206010016717 Fistula Diseases 0.000 description 1
- 208000015872 Gaucher disease Diseases 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 208000010412 Glaucoma Diseases 0.000 description 1
- 102000006395 Globulins Human genes 0.000 description 1
- 108010044091 Globulins Proteins 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 101000946889 Homo sapiens Monocyte differentiation antigen CD14 Proteins 0.000 description 1
- 101100468540 Homo sapiens RGMA gene Proteins 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 102100037850 Interferon gamma Human genes 0.000 description 1
- 108010074328 Interferon-gamma Proteins 0.000 description 1
- 208000032382 Ischaemic stroke Diseases 0.000 description 1
- 102100035877 Monocyte differentiation antigen CD14 Human genes 0.000 description 1
- 206010056886 Mucopolysaccharidosis I Diseases 0.000 description 1
- 241000711408 Murine respirovirus Species 0.000 description 1
- 108010083674 Myelin Proteins Proteins 0.000 description 1
- 102000006386 Myelin Proteins Human genes 0.000 description 1
- 208000003926 Myelitis Diseases 0.000 description 1
- 108091007491 NSP3 Papain-like protease domains Proteins 0.000 description 1
- 208000029067 Neuromyelitis optica spectrum disease Diseases 0.000 description 1
- 208000014060 Niemann-Pick disease Diseases 0.000 description 1
- 208000003435 Optic Neuritis Diseases 0.000 description 1
- 206010069350 Osmotic demyelination syndrome Diseases 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 241000978776 Senegalia senegal Species 0.000 description 1
- 241000710960 Sindbis virus Species 0.000 description 1
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 1
- 239000004288 Sodium dehydroacetate Substances 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 208000006011 Stroke Diseases 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 230000006044 T cell activation Effects 0.000 description 1
- 101710137500 T7 RNA polymerase Proteins 0.000 description 1
- 208000022292 Tay-Sachs disease Diseases 0.000 description 1
- 208000026062 Tissue disease Diseases 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- 108091023040 Transcription factor Proteins 0.000 description 1
- 102000040945 Transcription factor Human genes 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 241000700618 Vaccinia virus Species 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 239000008351 acetate buffer Substances 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 206010064930 age-related macular degeneration Diseases 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 206010002026 amyotrophic lateral sclerosis Diseases 0.000 description 1
- 230000000692 anti-sense effect Effects 0.000 description 1
- 239000003125 aqueous solvent Substances 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 230000002238 attenuated effect Effects 0.000 description 1
- 230000004009 axon guidance Effects 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 229940112869 bone morphogenetic protein Drugs 0.000 description 1
- 229910021538 borax Inorganic materials 0.000 description 1
- 229960002645 boric acid Drugs 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000010354 butylated hydroxytoluene Nutrition 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 230000021164 cell adhesion Effects 0.000 description 1
- 230000005779 cell damage Effects 0.000 description 1
- 208000037887 cell injury Diseases 0.000 description 1
- 208000009885 central pontine myelinolysis Diseases 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 208000037976 chronic inflammation Diseases 0.000 description 1
- 230000006020 chronic inflammation Effects 0.000 description 1
- 239000007979 citrate buffer Substances 0.000 description 1
- 238000010367 cloning Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 239000013065 commercial product Substances 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 210000004748 cultured cell Anatomy 0.000 description 1
- 210000004443 dendritic cell Anatomy 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- UGMCXQCYOVCMTB-UHFFFAOYSA-K dihydroxy(stearato)aluminium Chemical compound CCCCCCCCCCCCCCCCCC(=O)O[Al](O)O UGMCXQCYOVCMTB-UHFFFAOYSA-K 0.000 description 1
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 241001493065 dsRNA viruses Species 0.000 description 1
- 206010013990 dysuria Diseases 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 230000003890 fistula Effects 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 238000003633 gene expression assay Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 235000001727 glucose Nutrition 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 210000001320 hippocampus Anatomy 0.000 description 1
- 229920003063 hydroxymethyl cellulose Polymers 0.000 description 1
- 229940031574 hydroxymethyl cellulose Drugs 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 238000002649 immunization Methods 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 230000031261 interleukin-10 production Effects 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- FZWBNHMXJMCXLU-BLAUPYHCSA-N isomaltotriose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@@H](OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O)O1 FZWBNHMXJMCXLU-BLAUPYHCSA-N 0.000 description 1
- 239000000644 isotonic solution Substances 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 208000036546 leukodystrophy Diseases 0.000 description 1
- 238000001638 lipofection Methods 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 208000002780 macular degeneration Diseases 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000002923 metal particle Substances 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 238000000520 microinjection Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 1
- 238000010172 mouse model Methods 0.000 description 1
- 210000005012 myelin Anatomy 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- 210000003061 neural cell Anatomy 0.000 description 1
- 230000001537 neural effect Effects 0.000 description 1
- 210000000276 neural tube Anatomy 0.000 description 1
- 208000008795 neuromyelitis optica Diseases 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 230000008506 pathogenesis Effects 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 210000005259 peripheral blood Anatomy 0.000 description 1
- 239000011886 peripheral blood Substances 0.000 description 1
- 238000002823 phage display Methods 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920001184 polypeptide Polymers 0.000 description 1
- 229940068968 polysorbate 80 Drugs 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 206010036807 progressive multifocal leukoencephalopathy Diseases 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 210000001525 retina Anatomy 0.000 description 1
- 238000003757 reverse transcription PCR Methods 0.000 description 1
- 101150096543 rpl19 gene Proteins 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 231100000241 scar Toxicity 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 230000011664 signaling Effects 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 235000002639 sodium chloride Nutrition 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 235000019259 sodium dehydroacetate Nutrition 0.000 description 1
- 229940079839 sodium dehydroacetate Drugs 0.000 description 1
- 229940037001 sodium edetate Drugs 0.000 description 1
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 235000010339 sodium tetraborate Nutrition 0.000 description 1
- 239000004328 sodium tetraborate Substances 0.000 description 1
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 1
- 235000019345 sodium thiosulphate Nutrition 0.000 description 1
- DSOWAKKSGYUMTF-GZOLSCHFSA-M sodium;(1e)-1-(6-methyl-2,4-dioxopyran-3-ylidene)ethanolate Chemical compound [Na+].C\C([O-])=C1/C(=O)OC(C)=CC1=O DSOWAKKSGYUMTF-GZOLSCHFSA-M 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 210000001032 spinal nerve Anatomy 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 238000002636 symptomatic treatment Methods 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 230000000451 tissue damage Effects 0.000 description 1
- 231100000827 tissue damage Toxicity 0.000 description 1
- 235000010487 tragacanth Nutrition 0.000 description 1
- 239000000196 tragacanth Substances 0.000 description 1
- 229940116362 tragacanth Drugs 0.000 description 1
- 238000001890 transfection Methods 0.000 description 1
- 230000009261 transgenic effect Effects 0.000 description 1
- 208000009174 transverse myelitis Diseases 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 241001529453 unidentified herpesvirus Species 0.000 description 1
- 208000002670 vitamin B12 deficiency Diseases 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2863—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against receptors for growth factors, growth regulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/22—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against growth factors ; against growth regulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/74—Inducing cell proliferation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
Definitions
- the present invention relates to an agent for treating or preventing HTLV-1-related myelopathy (HAM).
- HAM HTLV-1-related myelopathy
- the invention also relates to a method for treating HAM.
- HTLV-1 Human T-cell Lukemia Virus Type 1
- T cells infected with HTLV-1 form chronic inflammation in the spinal cord, resulting in damage and degeneration of spinal nerve cells and spastic spinal palsy.
- Spastic spinal palsy caused by cells infected with HTLV-1 is called HTLV-1-associated myelopathy (also abbreviated as HAM).
- HAM HTLV-1-associated myelopathy
- Symptoms of HAM include symptoms such as paralysis of both feet, pain, dysuria, and stubborn constipation due to nervous tissue disorders. As these symptoms progress, people become wheelchair-bound and bedridden.
- HAM is one of the diseases designated as intractable disease in Japan.
- Non-Patent Document 1 Non-Patent Document 5
- RGMa protein is a member of the RGM (Repulsive guidance molecule) family of proteins involved in axon guidance of nerve cells of the retina and hippocampus, closure of neural tubes, and the like.
- the function of the RGM is not limited to these, and it is known that the RGM has various functions.
- Patent Document 1 discloses that RGM is expressed in bone marrow-derived dendritic cells (BMDCs), an RGM receptor is expressed on CD4 + T cells and CD11b + macrophages, and RGM is expressed on the RGM receptor. It is disclosed that binding enhances the cell adhesion activity of CD4 + T cells and CD11b + macrophages.
- Patent Document 1 discloses that anti-RGM neutralizing antibodies can reduce both clinical symptoms and tissue lesions of multiple sclerosis model mice, and that spleen cells obtained from the mice are antigen-specific and non-specific. It is disclosed that effective T cell activation was attenuated.
- Patent Documents 2 and 3 disclose neutralizing monoclonal antibodies against RGMa that selectively inhibit the binding of RGMa to RGMa receptor neogenin and bone morphogenetic proteins 2 and 4 (BMP-2, BMP-4). ing.
- BMP-2, BMP-4 bone morphogenetic proteins 2 and 4
- the neutralizing monoclonal antibody the injured and inflamed human central nervous system, specifically, multiple sclerosis, acute spinal cord injury, after brain trauma, Huntington's chorea, Parkinson's disease, Alzheimer's disease, etc.
- neurodegenerative diseases it is possible to promote nerve regeneration and regrowth of disrupted neuronal connections.
- Patent Document 4 discloses that RGMa is localized to central nervous system myelin, fresh foci and mature scar tissue of humans suffering from traumatic brain injury or ischemic stroke, and to diagnose these neurodegenerative diseases. For the purpose, a method for detecting and quantifying RGMa fragments has been disclosed.
- Patent Document 4 discloses, as neurodegenerative diseases and disorders for which RGMa fragments are detected, multiple sclerosis, Parkinson's disease, Alzheimer's disease, Tay-Sachs disease, Niemann-Pick disease, Gaucher disease, Hurler's syndrome, Huntington's disease Disease, amyotrophic lateral sclerosis, idiopathic inflammatory demyelinating disease, vitamin B12 deficiency, central pontine myelinolysis, spinal fistula, transverse myelitis, Devic disease, progressive multifocal leukoencephalopathy, optic neuritis , Spinal cord injury, traumatic brain injury, stroke, glaucoma, diabetic retinopathy, age-related macular degeneration, leukodystrophies.
- Non-patent Document 1 and Patent Document 5 According to the method using the anti-CCR4 antibody disclosed in Non-patent Document 1 and Patent Document 5, the symptoms of HAM can be improved, but the effect of the anti-CCR4 antibody is limited in HAM patients with advanced nerve destruction. It is. Therefore, there is a need for better methods that can treat HAM.
- the present invention has been made in view of the above problems, and has as its object to provide a therapeutic agent and a therapeutic method capable of treating HAM.
- the present inventors have conducted intensive studies to solve the above problems, and as a result, have found that a substance that inhibits RGMa is effective for treating HAM, and have completed the present invention.
- the present invention is as follows.
- An agent for treating or preventing HTLV-1-related myelopathy comprising an RGMa inhibitor.
- a method of treating HAM comprising administering to a HTLV-1-associated myelopathy (HAM) patient in need thereof a pharmacologically effective amount of an RGMa inhibitor.
- the method for treating HAM according to [3], wherein the RGMa inhibitor is an antibody that recognizes RGMa.
- HAM which is an intractable disease can be treated.
- FIG. 4 is a diagram showing, among CD4-positive T cells, CADM1, which is an indicator of HTLV-1-infected cells, and groups P, D, and N classified by CD7.
- Normal T cells (Normal.CD4), CD4 + T cells from HAM patients (HAM.CD4), CD4 + / CADM1 negative / CD7 + T cells (Normal.P) from healthy individuals, HTLV-1 infected CD4 + / CADM1 Negative / CD7 positive T cells (P group), HTLV-1 infected CD4 positive / CADM1 positive / CD7 positive T cells (D group), HTLV-1 infected CD4 positive / CADM1 positive / CD7 negative T cells (N group) , CD4 + / CADM1-positive / CD7-negative T cells (Acute.N), CD4 + T cells (Normal.CD4.1) in healthy ATL patients, PBMC (Smoldering) from smoldering ATL patients, Chronic ATL patients FIG.
- FIG. 2 shows the expression levels of the RGMa gene in PBMC (Chronic) and PBMC (Acute) derived from acute ATL patients. It is a figure which shows the expression level of RGMa in CD4 positive T cells of HAM patients and CD4 positive T cells of healthy persons.
- FIG. 9 is a view showing the results of analyzing the expression level of RGMa between cell types in PBMC of a HAM patient.
- FIG. 4 is a graph showing changes in RGMa expression associated with the expression of HTLV-1 virus in culture of HAM patient PBMC.
- FIG. 3 is a diagram showing the level of H3K27me3 near -2,916 bp upstream from the transcription start point of the RGMa gene.
- FIG. 4 is a diagram showing the level of RGMa gene mRNA when a lentiviral vector into which cDNA encoding HTLV-1 Tax is inserted into a human CD4-positive T-cell leukemia cell line Jurkat.
- FIG. 9 shows the results of analyzing protein expression of Tax and RGMa in the HTLV-1-tax expression-inducing cell line JPX-9.
- FIG. 3 is a view showing the results of the effect of the RGMa antibody on spontaneous growth activity with respect to the effect of the RGMa antibody on PBMC of HAM patients.
- FIG. 9 is a view showing the results of the effect of the RGMa antibody on changes in the amount of HTLV-1 provirus in relation to the effect of the RGMa antibody on PBMCs of HAM patients.
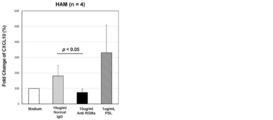
- FIG. 4 is a view showing the results of the action of the RGMa antibody on CXCL10 production with respect to the action of the RGMa antibody on PBMC of a HAM patient.
- FIG. 3 is a view showing the results of the effect of an RGMa antibody on cytokine production of PBMC of a HAM patient with respect to the effect of an RGMa antibody on PBMC of a HAM patient.
- FIG. 4 is a diagram showing that HAM-PBMC caused apoptosis induction of a neuronal cell line.
- FIG. 9 shows the results of the inhibitory effect of RGMa antibody on apoptosis induction of neuronal cell lines by HTLV-1-tax-induced cell lines.
- (a) is a FACS plot when NB-1 cells and JPX-9 cells (unstimulated) were co-cultured.
- (b) is a FACS plot when NB-1 cells and CdCl 2 -stimulated JPX-9 cells (HTLV-1-tax expressing cells) were co-cultured.
- (c) is a FACS plot when a control antibody is added to the condition of (b).
- (d) is a FACS plot when an anti-RGMa antibody was added to the conditions of (b).
- the present invention is an agent for treating or preventing HTLV-1-related myelopathy (HAM), which comprises an RGMa inhibitor.
- the RGMa inhibitor may be a substance that acts on RGMa itself to inhibit the activity of RGMa or a substance that suppresses the expression of RGMa.
- Examples of the RGMa inhibitor include a compound having an activity of inhibiting RGMa, an antibody that recognizes RGMa, and the like.
- RGMa inhibitor examples include substances that suppress the expression of RGMa, such as siRNA (short interfering RNA), shRNA (short hairpin RNA), and antisense oligonucleotide of the gene that expresses RGMa.
- examples of the RGMa gene include, but are not limited to, a human RGMa gene having the nucleotide sequence shown in SEQ ID NO: 1 and an RGMa gene having the nucleotide sequence shown in SEQ ID NO: 2.
- Information on the nucleotide sequences of RGMa genes derived from various organisms can be obtained from databases such as GenBank.
- siRNA is a double-stranded RNA that can suppress the expression of the target RGMa gene.
- the length (base length) of the base sequence in the siRNA is not particularly limited, but is preferably less than about 30 bases, more preferably about 19 to 27 bases, and still more preferably about 21 to 25 bases.
- shRNA has a double-stranded structure in the molecule by partially containing a palindromic base sequence in the single-stranded RNA, and is composed of a short hairpin structure having a short hairpin structure having an overhang at the 3 ′ end. Refers to molecules of base pairs or more. After being introduced into a cell, the shRNA is decomposed into a length of about 20 bases in the cell, and can suppress the expression of the target RGMa gene, similarly to the siRNA.
- SiRNA and shRNA can be artificially chemically synthesized.
- antisense and sense RNA can be synthesized in vitro from template DNA using T7 RNA polymerase and T7 promoter.
- the antisense oligonucleotide may be any nucleotide that is complementary to or hybridizes with a continuous base sequence of less than about 30 bases in the DNA sequence of the RGMa gene, and may be either DNA or RNA. Further, it may be modified as long as the function is not hindered.
- the antisense oligonucleotide can be synthesized by a conventional method, and can be easily synthesized by, for example, a commercially available DNA synthesizer.
- an antibody that recognizes RGMa is preferable.
- an antibody that recognizes RGMa is also referred to as an RGMa antibody.
- the RGMa antibody of the present invention may be any antibody that binds to RGMa and inhibits its activity, such as an antibody that binds to RGMa to prevent RGMa from binding to the RGMa receptor.
- the RGMa antibody in the present invention may be a monoclonal antibody or a polyclonal antibody. Further, the antibody in the present invention may be of any isotype of IgG, IgM, IgA, IgD and IgE.
- the RGMa antibody in the present invention may be, for example, a mouse antibody, a human CDR-grafted antibody, a human chimeric antibody, a humanized antibody, a fully human antibody, or a low-molecular antibody. These antibodies may be used alone or in combination of two or more.
- a human CDR-grafted antibody is an antibody obtained by replacing the CDR of an antibody of a non-human animal with the CDR of a human antibody.
- a human chimeric antibody is an antibody comprising a variable region derived from an antibody of a non-human animal and a constant region derived from a human antibody.
- humanized antibody refers to an antibody of a non-human animal, in which a part derived from a human antibody is incorporated, leaving a part of a highly safe region, a human chimeric antibody, and a humanized antibody. The concept includes CDR-grafted antibodies.
- small antibody refers to an antibody fragment or an antibody fragment obtained by binding any molecule to an antibody fragment and recognizing the same epitope as the original antibody.
- Fab consisting of VL, VH, CL and CH1 regions; F (ab ') 2 in which two Fabs are connected by a disulfide bond at a hinge region; Fv consisting of VL and VH; SdFv, Diabody, and sc (Fv) 2, in addition to scFv, which is a single-chain antibody linked by the above polypeptide linker, but is not limited thereto.
- the RGMa antibody used in the present invention can be prepared by a known method using RGMa or a fragment thereof as an immunogen. The fact that the obtained antibody is an RGMa antibody can be confirmed using RGMa activity as an index.
- RGMa include human RGMa containing the amino acid sequence shown in SEQ ID NO: 3, RGMa containing the amino acid sequence shown in SEQ ID NO: 4, and the like. RGMa from various organisms can be used as the immunogen.
- the amino acid sequence of RGMa can be obtained from a known database such as Protein Data Bank.
- the RGMa antibody used in the present invention is a polyclonal antibody, it can be prepared, for example, as follows. First, RGMa or a fragment thereof is dissolved in phosphate-buffered saline (also referred to as PBS) as an antigen, and a normal adjuvant, for example, Freund's complete adjuvant mixed with an appropriate amount thereof as necessary is used as an immunogen. Immunize mammals such as rats, rabbits, goats and horses.
- the immunization method is not particularly limited, and examples thereof include a method of injecting one time or two or more times at an appropriate interval, subcutaneously or intraperitoneally.
- blood can be collected from the immunized animal, the serum can be separated, and the polyclonal antibody fraction can be obtained by purification.
- the RGMa antibody used in the present invention is a monoclonal antibody
- the monoclonal antibody is obtained by fusing immunocytes obtained from the immunized mammal, for example, spleen cells and myeloma cells to obtain a hybridoma, and culturing the hybridoma. It can be obtained by collecting the antibody from the product.
- the above-mentioned monoclonal antibody can also be obtained by cloning an antibody gene from a hybridoma, incorporating the gene into an appropriate vector, introducing this into a host cell, and producing a recombinant monoclonal antibody using a gene recombination technique. Further, the above-mentioned monoclonal antibody can also be prepared using a phage display method.
- RGMa antibody used in the present invention examples include, for example, Yamashita, T., Mueller, BK & Hata, K. Neogenin and repulsive guidance molecule signaling in the central nervous system.Curr.Opin.Neurobiol. 17, 29-34 (2007 JP-A-2014-138599; JP-A-2016-175897; JP-T-2017-526930; WO 2016/175236; JP-T-2015-508061; Can be.
- the RGMa antibody used in the present invention can be obtained as a commercial product, for example, from Immune Biological Laboratories (IBL) or R & D Systems.
- RGMa antibody as an antigen-binding domain, GTTPDY (SEQ ID NO: 7); FQATHDPLT (SEQ ID NO: 10); ARRNEYYGSSFFDY (SEQ ID NO: 13); LQGYIPPRT (SEQ ID NO: 16); and a modified CDR amino acid sequence having at least 50% sequence identity to one of the sequences; Preferably, it comprises at least one CDR comprising an amino acid sequence selected from the group consisting of: The above sequence identity is preferably 80% or more, more preferably 90% or more.
- amino acids may be represented by a conventional one-letter code or three-letter code.
- the complementarity-determining region refers to a region that forms an antigen-binding site in the variable region of an immunoglobulin molecule, and is also called a hypervariable region, and refers to a portion in which the amino acid sequence changes greatly for each immunoglobulin molecule. .
- There are three CDRs in each of the light and heavy chains (CDR-L1, CDR-L2, CDR-L3, and CDR-H1, CDR-H2, CDR-H3).
- the CDRs of the immunoglobulin molecule are determined according to the Kabat numbering system (Kabat et al., 1987, Sequences of Proteins of Immunological Interest, US Department of Health and Human Services, NIH, USA).
- the amino acid sequence of the CDR is invariable from the predetermined sequence, and other than the CDR may be changed by mutation or the like.
- the homology is preferably 90% or more.
- the RGMa antibody is an amino acid selected from the group consisting of SEQ ID NOs: 5, 6, 8, 9, 11, 12, 14, 15, and a modified CDR amino acid sequence having at least 50% sequence identity to one of the sequences. Preferably, it further comprises at least one CDR comprising the sequence.
- the above sequence identity is preferably 80% or more, more preferably 90% or more.
- the RGMa antibody comprises a set of variable domain CDRs as shown in Table 1, or a modified CDR amino acid sequence wherein at least one of the three CDRs has at least 50%, preferably 80%, more preferably 90% sequence identity with the parent sequence. More preferably, it comprises at least three CDRs selected from the set of variable domains. Further, the RGMa antibody further preferably includes at least two variable domain CDR sets shown in Table 1. Further, the at least two variable domain CDR sets are preferably a combination of a VH5F9 set and a VL5F9 set, or a combination of a VH8D1 set and a VL8D1 set.
- the RGMa antibody may include a framework region.
- the amino acid sequences contained in the framework regions include SEQ ID NOs: 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39 and 40. No. These amino acid sequences may be used alone or in a combination of two or more.
- the RGMa antibody has at least one heavy chain variable domain selected from SEQ ID NOs: 41, 42, 43, 44, 45, 46, 47, 48 and 49 as a heavy chain variable domain and a light chain variable domain; and / or Or it preferably comprises at least one light chain variable domain selected from SEQ ID NOs: 50, 51 and 52.
- the RGMa antibody the binding site when binding to RGMa is a site that causes RGMa inhibition is not particularly limited, for example, in terms of human RGMa, EEVVNAVEDWDSQG (SEQ ID NO: 53) NQQIDFQAFHTNAE (SEQ ID NO: 54) PTAPETFPYET (SEQ ID NO: 55) KLPVEDLYYQA (SEQ ID NO: 56) LYERTRDLPGRAAAGL (SEQ ID NO: 57) It preferably binds to one or more peptides having the amino acid sequence represented by More preferably, the RGMa antibody binds to a peptide having the amino acid sequence represented by SEQ ID NO: 53 and / or SEQ ID NO: 54, and a peptide having the amino acid sequence represented by SEQ ID NO: 53 and / or SEQ ID NO: 54; More preferably, it binds to a peptide having the amino acid sequence represented by SEQ ID NO: 55 and / or SEQ ID NO:
- the RGMa antibody is preferably an antibody that binds to the amino acid sequence of human RGMa at the 250th position or later. More preferably, the RGMa antibody binds to a peptide having the amino acid sequence represented by SEQ ID NO: 53 and SEQ ID NO: 54, and SEQ ID NO: 55 or SEQ ID NO: 56.
- RGMa antibodies are polyclonal antibodies and monoclonal antibodies obtained by immunizing a mammal such as a mouse with the RGMa protein or a partial fragment thereof (eg, a fragment containing one or more of SEQ ID NOS: 53 to 57) as an antigen; Chimeric antibodies and humanized antibodies produced using genetic recombination techniques, human antibodies produced using human antibody-producing transgenic animals, and the like may be used.
- a humanized antibody or a human antibody is desirable from the viewpoint of side effects.
- the RGMa antibody may be used as a partial fragment of a polyclonal antibody and / or a monoclonal antibody that recognizes an RGMa protein or a partial fragment thereof (eg, a fragment containing one or more of SEQ ID NOS: 53 to 57) as an antigen. It may be.
- the complementarity determining region 1 of the light chain (LCDR1), the complementarity determining region 2 of the light chain (LCDR2), the complementarity determining region 3 of the light chain (LCDR3) The respective amino acid sequences of heavy chain complementarity determining region 1 (HCDR1), heavy chain complementarity determining region 2 (HCDR2) and heavy chain complementarity determining region 3 (HCDR3)
- LCDR1 RASQDISSYLN (SEQ ID NO: 58)
- LCDR2 YTSRLHS (SEQ ID NO: 59)
- LCDR1 RSSQSLVHSNGNTYLH (SEQ ID NO: 64) LCDR
- RGMa antibody examples include an antibody having the amino acid sequence of SEQ ID NO: 73 in the light chain and the amino acid sequence of SEQ ID NO: 74 in the heavy chain.
- amino acid sequence represented by these SEQ ID NOs there may be substitution, deletion, addition or insertion of one or several amino acids (1 to 20, 1 to 10, or 1 to 5). Such substitutions, deletions and additions may be introduced into CDRs, but are preferably introduced into regions other than CDRs.
- a mouse / human chimeric antibody having a human-derived constant region may be used.
- the amino acid sequence of SEQ ID NO: 77 (variable region: 1 to 107) is used for the light chain
- the amino acid sequence of SEQ ID NO: 78 is used for the heavy chain.
- An antibody having an amino acid sequence (the variable region is from 1 to 116) is exemplified.
- substitution, deletion, addition or insertion of one or several amino acids (1 to 20, 1 to 10, or 1 to 5).
- substitutions, deletions and additions may be introduced into CDRs, but are preferably introduced into regions other than CDRs.
- a humanized antibody derived from a human other than CDR may be used.
- the heavy chain has any of the amino acid sequences of SEQ ID NOs: 70 to 87 (the variable region is up to 116 residues on the N-terminal side), and the light chain is SEQ ID NOs: 88 to 94 (the variable region is the N-terminal Antibodies having any of the amino acid sequences (from 1 to 107 residues on the side) are exemplified.
- the amino acid sequence represented by these SEQ ID NOs may have substitution, deletion, addition or insertion of one or several amino acids (1 to 20, 1 to 10, or 1 to 5). Such substitutions, deletions and additions may be introduced into CDRs, but are preferably introduced into regions other than CDRs.
- the heavy chain amino acid sequence and the light chain amino acid sequence may be in any combination of these, but are preferably antibodies having the amino acid sequence of SEQ ID NO: 84 in the heavy chain and the amino acid sequence of SEQ ID NO: 88 in the light chain.
- the amino acid sequence corresponding to the heavy chain variable region is represented by SEQ ID NO: 95
- the amino acid sequence corresponding to the light chain variable region is represented by SEQ ID NO: 96.
- the RGMa antibody has a heavy chain variable region (VH) EVQLVESGGGLVQPGRSLRLSCTASGFTFSDAWMDWVRQAPGKGLEWVAEIRSKANNHATYYAESVKGRFTISRDDSKSIVYLQMNSLRTEDTALYYCTRRDGAYWGKGTTVTVSS (SEQ ID NO: 95) Or comprising an amino acid sequence having at least 90% identity to said amino acid sequence, Light chain variable region (VL) DIQMTQSPSSVSASVGDRVTITCRASQDISSYLNWYQQKPGKAPKLLIYYTSRLHSGVPSRFSGSGSGTDFTLTISSLQPEDFASYFCQQLNTLPWTFGGGTKVEME (SEQ ID NO: 96) Alternatively, an isolated RGMa antibody, or an antigen-binding fragment thereof, comprising an amino acid sequence having at least 90% identity to the amino acid sequence is preferred.
- VH heavy chain variable region
- RGMa antibody examples include an antibody having the amino acid sequence of SEQ ID NO: 75 in the light chain and the amino acid sequence of SEQ ID NO: 76 in the heavy chain.
- amino acid sequence represented by these SEQ ID NOs there may be substitution, deletion, addition or insertion of one or several amino acids (1 to 20, 1 to 10, or 1 to 5).
- substitutions, deletions and additions may be introduced into CDRs, but are preferably introduced into regions other than CDRs.
- a mouse / human chimeric antibody having a constant region derived from human may be used, and a humanized antibody derived from a human other than CDR may be used.
- the anti-RGMa antibody may be an isolated RGMa antibody selected from the following (a1) to (h1), or an antigen-binding fragment thereof.
- A1 a light chain variable region including LCDR1 containing the amino acid sequence of SEQ ID NO: 97, LCDR2 containing the amino acid sequence of SEQ ID NO: 98 and LCDR3 containing the amino acid sequence of SEQ ID NO: 99, and SEQ ID NO: 100
- HCDR1 comprising the amino acid sequence of SEQ ID NO: 101
- HCDR2 comprising the amino acid sequence of SEQ ID NO: 101 and an anti-RGMa antibody comprising a heavy chain variable region comprising HCDR3 comprising the amino acid sequence of SEQ ID NO: 102, or an antigen-binding fragment thereof
- B1 a light chain variable region including LCDR1 including the amino acid sequence of SEQ ID NO: 97, LCDR2 including the amino acid sequence of SEQ ID NO: 98, and LCDR3 including the amino acid sequence of SEQ ID NO: 103
- the MaRGMa antibody is an antibody having the above-mentioned amino acid sequence, is preferably a humanized antibody, and preferably has a human IgG constant region.
- HAM can be treated or prevented by the RGMa inhibitor of the present invention.
- cells derived from HAM patients cause neuronal cell death, that is, induction of spinal cord tissue damage and degeneration.
- RGMa is remarkably expressed in CD4-positive T cells, which are the main HTLV-1-infected cells of HAM patients. Expression was found to be involved.
- RGMa was involved in the production of CXCL10, IL-2, and IL-10.
- CXCL10 is a protein produced from HAM-pathogenic HTLV-1-infected T cells in response to IFN- ⁇ , and is known to induce the pathology of HAM (Brain 2013). It is known that there is a strong correlation with the speed of progress (PLoS Negl Trop Dis 2013).
- RGMa antibody was allowed to act on cells of HAM patients, the production of CXCL10 was suppressed.
- IL-10 is one of cytokines and works to suppress inflammation. When RGMa antibody was applied to cells of HAM patients, IL-10 production was significantly increased. In order for HAM to have a true therapeutic effect, it is necessary to suppress nerve cell damage.
- HTLV-1-tax The expression level of HTLV-1-tax is high in HTLV-1-infected cells of HAM patients (Blood 2002), indicating that HTLV-1-tax is important for pathogenesis (J Clin Invest 2014).
- HTLV-1-tax induces RGMa expression
- HAM patient-derived cells with high RGMa expression levels induce neuronal cell death.Importantly, HTLV-1-tax expressing cells
- RGMa inhibitor suppresses neuronal cell death by RGMa.
- RGMa inhibitors not only suppress HAM-specific inflammatory responses, but also suppress neuronal death by HTLV-1-tax-expressing cells, which are pathogenic cells of HAM patients, thus treating or preventing HAM. can do.
- RGMa inhibitors can treat or prevent HAM
- the invention relates to RGMa inhibitors for use in treating HAM; pharmaceutical compositions for use in treating HAM; Use of an RGMa inhibitor in the manufacture of a medicament for the treatment of HAM; use of an RGMa inhibitor for use in the manufacture of a medicament for the treatment of HAM; an effective amount of an RGMa inhibitor in a subject in need thereof Or a method for treating or preventing HAM, comprising administering to a subject.
- the therapeutic or prophylactic agent for HAM of the present invention may contain an RGMa inhibitor, and may be formulated by appropriately mixing a pharmaceutically acceptable carrier and / or additive.
- Formulation forms include, for example, oral preparations such as tablets, coated tablets, pills, powders, granules, capsules, solutions, suspensions, and emulsions; injections, infusions, suppositories, ointments, patches and the like. Parenteral preparations; The mixing ratio of the carrier or the additive may be appropriately set based on the range usually adopted in the pharmaceutical field.
- the carrier is not particularly restricted but includes, for example, water, physiological saline, other aqueous solvents, and aqueous or oily bases.
- the additives are not particularly limited, but include excipients, binders, pH adjusters, disintegrants, absorption enhancers, lubricants, coloring agents, flavoring agents, flavors, and the like.
- the RGMa inhibitor of the present invention is an antibody that recognizes RGMa
- the antibody is administered as a parenteral administration route, for example, intravenously, intramuscularly, as an injection or infusion formulated with a pharmaceutically acceptable carrier. It is preferably administered intradermally, intraperitoneally, subcutaneously or topically.
- the injection or infusion containing the RGMa antibody can be used as a solution, suspension or emulsion.
- the solvent include distilled water for injection, physiological saline, glucose solution, and isotonic solution (for example, a solution of sodium chloride, potassium chloride, glycerin, mannitol, sorbitol, boric acid, borax, propylene glycol, and the like). Etc. can be used. These solvents may be used alone or in combination of two or more.
- the injection or infusion may contain additives such as a stabilizer, a solubilizer, a suspending agent, an emulsifier, a soothing agent, a buffer, a preservative, a preservative, and a pH adjuster.
- a stabilizer for example, albumin, globulin, gelatin, mannitol, glucose, dextran, ethylene glycol, propylene glycol, ascorbic acid, sodium bisulfite, sodium thiosulfate, sodium EDTA, sodium citrate, dibutylhydroxytoluene and the like are used.
- a stabilizer for example, albumin, globulin, gelatin, mannitol, glucose, dextran, ethylene glycol, propylene glycol, ascorbic acid, sodium bisulfite, sodium thiosulfate, sodium EDTA, sodium citrate, dibutylhydroxytoluene and the like are used. Can be.
- solubilizers include alcohols (eg, ethanol, etc.), polyalcohols (eg, propylene glycol, polyethylene glycol, etc.), nonionic surfactants (eg, Polysorbate 80 (registered trademark), HCO-50, etc.) Etc.
- alcohols eg, ethanol, etc.
- polyalcohols eg, propylene glycol, polyethylene glycol, etc.
- nonionic surfactants eg, Polysorbate 80 (registered trademark), HCO-50, etc.
- Etc. can be used.
- the suspending agent for example, glyceryl monostearate, aluminum monostearate, methyl cellulose, carboxymethyl cellulose, hydroxymethyl cellulose, sodium lauryl sulfate and the like can be used.
- emulsifier for example, gum arabic, sodium alginate, tragacanth and the like can be used.
- the soothing agent for example, benzyl alcohol, chlorobutanol, sorbitol and the like can be used.
- the buffer for example, a phosphate buffer, an acetate buffer, a borate buffer, a carbonate buffer, a citrate buffer, a Tris buffer, and the like can be used.
- the preservative for example, methyl paraoxybenzoate, ethyl paraoxybenzoate, propyl paraoxybenzoate, butyl paraoxybenzoate, chlorobutanol, benzyl alcohol, benzalkonium chloride, sodium dehydroacetate, sodium edetate, boric acid, borate Sand or the like can be used.
- preservative for example, benzalkonium chloride, paraoxybenzoic acid, chlorobutanol and the like can be used.
- pH adjuster for example, hydrochloric acid, sodium hydroxide, phosphoric acid, acetic acid and the like can be used.
- the RGMa inhibitor of the present invention is a substance that suppresses the expression of RGMa such as siRNA (short interfering RNA), shRNA (short hairpin RNA), and antisense oligonucleotide of a gene that expresses RGMa, a non-viral vector or It can be administered in the form of a viral vector.
- siRNA short interfering RNA
- shRNA short hairpin RNA
- antisense oligonucleotide of a gene that expresses RGMa a non-viral vector or It can be administered in the form of a viral vector.
- the method of administration is to introduce nucleic acid molecules using liposomes (liposome method, HVJ-liposome method, cationic liposome method, lipofection method, lipofectamine method, etc.)
- liposome method liposome method, HVJ-liposome method, cationic liposome method, lipofection method, lipofectamine method, etc.
- a microinjection method a method of transferring a nucleic acid molecule into a cell together with a carrier (metal particles) by a gene gun (Gene Gun), and the like.
- a viral vector such as a recombinant adenovirus or retrovirus can be used.
- Detoxified retrovirus adenovirus, adeno-associated virus, herpes virus, vaccinia virus, pox virus, polio virus, Sindbis virus, Sendai virus, DNA virus such as SV40 or RNA virus, DNA expressing siRNA or shRNA
- the gene can be introduced into a cell or tissue by introducing the recombinant virus into the cell or tissue.
- the preparation thus obtained can be administered to humans and other mammals such as rats, mice, rabbits, sheep, pigs, cows, cats, dogs, and monkeys by administering an effective amount thereof to, for example, humans.
- HAM can be prevented or treated.
- the dose is appropriately set in consideration of the purpose, the severity of the disease, the patient's age, weight, sex, medical history, type of active ingredient, and the like.
- Example 1 HAM and adult T-cell leukemia / lymphoma (ALT; adult T-cell leukemia / lymphoma) onset mechanism and comprehensive comparative analysis for elucidation of the disease state]
- ALT adult T-cell leukemia / lymphoma
- CADM1 which is an indicator of HTLV-1-infected cells
- mRNA expression analysis was performed.
- HAM was also analyzed from CD4 positive cells and CD4 negative cells enriched with magnetic beads from peripheral blood mononuclear cells from HAM patients in four cases.
- HTLV-1 non-infected healthy subjects Normal were similarly separated.
- PBMC Peripheral blood mononuclear cells
- ATL suppressing, chronic, acute disease types
- PBMC mainly composed of CD4 positive cells was used.
- a one-color microarray gene expression analysis method manufactured by Agilent Technologies, Inc. was performed.
- the RGMa gene level was graphed.
- the Log 2 value of the fluorescence intensity of the array was plotted.
- the HAM patient-derived CD4-positive T cell group was significantly different from all other groups (P ⁇ 0.05). The graph is shown in FIG.
- CD4 positive T cells were separated from PBMCs isolated from peripheral blood of five HAM patients using a human CD4 + isolation kit (Miltenyi Biotec) to obtain a cell population containing a large number of HTLV-1 infected cells.
- CD4-positive T cells were separated from PBMCs of four healthy subjects and used as a control group.
- Total RNA was recovered from the separated CD4-positive T cells, and cDNA was prepared using ReverTra Ace (Toyobo).
- ReverTra Ace Toyobo
- the difference in the expression level of RGMa between CD4 + T cells in HAM patients and CD4 + T cells in healthy subjects was analyzed by Real time PCR. 18srRNA was used for internal control.
- FIG. 3 shows a graph of the result of the analysis.
- HD-CD4 + in the graph refers to the control group
- HAM-CD4 + refers to the group of HAM patients.
- Example 3 Analysis of RGMa-expressing cells in PBMC
- Fc block treatment of HAM patient PBMC using Clear Back (MBL) add anti-CD3-PECy7 (TONBO), CD4-FITC (eBioscience), CD14-PE (eBioscience) antibodies and stain at 4 ° C for 30 minutes. went.
- FACS sorting was performed using AriaIIIu (BD), and CD3-positive CD4-negative cells (CD3 + CD4-), CD3-positive CD4-positive cells (CD3 + CD4 +), CD3-negative CD14-negative cells ( CD3-CD14-) and CD3-negative CD14-positive cells (CD3-CD14 +) were separated and collected.
- FIG. 4 shows a graph of the result of the analysis. In HAM-PBMC, it was found that RGMa expression was highest in CD3-positive CD4-positive cells (CD3 + CD4 +) where many infected cells were present.
- Example 4 RGMa expression change associated with culture of HAM patient PBMC
- the PBMCs of two HAM patients were suspended in a medium (RPMI1640 medium (wako) containing 10% FBS (GIBCO)) and seeded at 1e5 cells per 96-well round bottom plate for 10 wells and cultured for 1, 3, 5, and 7 days .
- Total RNA was extracted from PBMC cultured for each period together with PBMC on Day 0 in which no culture was performed, and cDNA was prepared using ReverTra Ace (Toyobo).
- FIG. 5 is a graph showing the results of the analysis.
- Example 5 Analysis of H3K27me3 level on all promoters
- the H3K27me3 levels near -2,916 bp upstream from the transcription start point of the RGMa gene were graphed. The graph is shown in FIG. In the graph, the Log 2 value of the fluorescence intensity of the array was plotted.
- HAM50 and HAM123 in the figure each indicate a HAM patient who obtained CD4 positive T cells. It was suggested that RGMa gene expression suppression was released in HAM patient-derived CD4-positive T cells.
- Example 6 Quantification of RGMa gene mRNA level
- a human CD4-positive T-cell leukemia cell line, Jurkat was transfected with a lentiviral vector into which cDNA encoding HTLV-1 Tax was inserted, and quantitative RT-PCR was performed for three consecutive days after the transfection. Was measured.
- RPL19 gene mRNA was also measured and used as an internal standard.
- a graph showing the results of the quantification is shown in FIG. It was suggested that HTLV-1 virus caused RGMa expression.
- Example 7 Tax-dependent RGMa expression induction in HTLV-1-tax expression-inducing cell line JPX-9 cells
- JPX9 cells were cultured in a medium (RPMI1640 medium containing 10% FBS) for 24 hours, and cadmium chloride (CdCl2; Nacalai Tesque) for inducing HTLV-1-tax expression was added to a final concentration of 20 ⁇ M. Culture was performed for 2 and 3 days. JACS cells treated with and without cadmium chloride were subjected to FACS analysis for protein expression of Tax and RGMa.
- the cadmium chloride-treated and untreated JPX9 cells were each washed, and subjected to cell permeation treatment using a Foxp3 / Transcription Factor Staining Buffer Kit (eBioscience). Thereafter, an anti-Tax-FITC antibody (Lt-4: provided by Dr. Tanaka, University of the Ryukyus) was added, and the cells were treated at 4 ° C. for 1 hour, thereby staining Tax proteins expressed in the cells. The stained Tax protein was detected by FACS analysis using CantoII.
- JPX9 cells treated with cadmium chloride and untreated were each washed, and an anti-RGMa antibody (manufactured by Immune Biological Laboratories (IBL)) was added and treated at 4 ° C. for 30 minutes. Thereafter, the cells were washed, an anti-mouse IgG-PE antibody (BioLegend) was added, and the mixture was reacted at 4 ° C. for 30 minutes to stain RGMa protein expressed in JPX9. The stained RGMa protein was detected by FACS analysis using CantoII.
- FIG. 8 shows the results of analysis of the protein expression of Tax and RGMa.
- Example 8 Examination of action of RGMa antibody on PBMC of HAM patient] (Effect of RGMa antibody on spontaneous growth activity)
- a medium RPMI1640 medium containing 10% FBS
- RGMa antibody R & D Systems
- the cultured cells were adsorbed to a glass filter (Printed Filtermat A PerkinElmer) using a cell harvester (Tomtec MH3 PerkinElmer), dried, and then impregnated with a solid scintillator Meltilex-A (PerkinElmer), followed by MicroBeta (WALLAC MicroBeta TriLux 1450).
- -021 was used to measure the amount of 3 H-Thymidine incorporated into the cells. Average counts of 3 H-Thymidine in Medium group of each HAM patient PBMC as 100%, calculates the relative value of each group, the average value was obtained for 3 H-Thymidine incorporation rate of HAM four patients.
- FIG. 9 shows the results.
- HAM patient PBMCs were suspended in a medium (RPMI1640 medium containing 10% FBS), seeded on a 96-well round bottom plate at 1e5 cells each, and added with RGMa antibody (R & D Systems) to a final concentration of 10 ⁇ g / ml.
- the cells were cultured in 0.1 ml of a culture solution at 37 ° C. under 5% CO 2 for 7 days.
- HAM-PBMC induces apoptosis of nerve cell line
- the neuronal cell line NB-1 or SK-N-AS was seeded on a 6-well plate and cultured for 24 hours. Then, a healthy person (HD) or a PBMC of a HAM patient was added and co-cultured. 48 hours after the start of the co-culture, the PBMC added together with the medium was removed, and after washing with PBS, the nerve cell line was recovered.
- Each of the recovered neural cell lines was analyzed by the TUNEL method (MEBSTAIN Apoptosis TUNEL Kit Direct (MBL)) for specifically detecting cells that have undergone DNA fragmentation due to apoptosis.
- FIG. 13 shows the analysis result.
- the X-axis in the histogram indicates the intensity of positive DNA fragmentation.
- HAM-derived cells more strongly induced apoptosis in neuronal cell lines than HD-derived cells. Specifically, cell death was analyzed according to the following ⁇ Experimental procedure>.
- ⁇ Experimental procedure> Each of the neuronal cell lines NB-1 and SK-N-AS was seeded on a 6-well plate and cultured for 24 hours. Next, HD or HAM-PBMC was added (double the number of seeded neuronal cell lines) and cultured for 48 hours. Thereafter, cells were fixed with 4% paraformaldehyde, and subjected to cell permeation treatment with 70% ethanol.
- MEBSTAIN Apoptosis TUNEL Kit Direct MBL
- Example 10 Inhibitory effect of RGMa antibody on apoptosis of neuronal cell line induced by T cell line expressing HTLV-1 Tax
- JPX9 (-) unstimulated JPX9
- JPX9 (+ CdCl2) JPX9 (+ CdCl2
- Normal mouse IgG2b (MBL) or RGMa antibody (IBL) was added to the final concentration of 10 ⁇ g / ml in the co-culture of NB-1 cells and (JPX9 (+ CdCl 2 ). 48 hours after the start of the co-culture. The JPX9 added together with the medium was removed, and the nerve cell lines were collected after washing with PBS.For each of the collected nerve cell lines, a TUNEL method (MEBSTAIN Apoptosis) for specifically detecting cells that had undergone DNA fragmentation due to apoptosis was performed. The analysis was performed using TUNEL Kit Direct (MBL), and the analysis results are shown in Fig. 14. The X-axis in the histogram indicates the intensity of positive DNA fragmentation.
- ⁇ Experimental procedure> The nerve cell line (NB-1) was seeded on a 6-well plate and cultured for 24 hours. Next, JPX9 ( ⁇ ) or JPX9 (+ CdCl 2 ) was added to NB-1 cells and co-cultured. The number of JPX9 ( ⁇ ) or JPX9 (+ CdCl 2 ) cells was twice the number of seeded NB-1 cells. JPX9 (+ CdCl 2 ) was washed three times with 10 ml of medium to remove cadmium chloride and added to NB-1 cells.
- NB-1 cells and (JPX9 (+ CdCl 2 ) were co-cultured with Normal Mouse IgG2b (MBL) or anti-RGMa antibody (IBL) to a final concentration of 10 ⁇ g / ml, and cultured for 48 hours. Thereafter, the cells were fixed with 4% paraformaldehyde, subjected to cell permeation treatment with 70% ethanol, and stained with an anti-CD45-V450 antibody.
- MEBSTAIN Apoptosis TUNEL Kit Direct MBL
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Virology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Neurosurgery (AREA)
- Neurology (AREA)
- Biomedical Technology (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- Epidemiology (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
HTLV-1に感染したT細胞は、脊髄に慢性の炎症を形成し、その結果、脊髄神経細胞の障害及び変性を引き起こし、痙性脊髄麻痺を惹起する。HTLV-1に感染した細胞が原因となって引き起こされる痙性脊髄麻痺は、HTLV-1関連脊髄症(HAMとも略記する。)と呼ばれる。
HAMの症状としては、神経の組織障害による、両足の麻痺、痛み、排尿障害、頑固な便秘等の症状等が挙げられる。これらの症状が進行すると、車いす生活、寝たきり生活になる。HAMは日本国の指定難病に指定される疾患の一つである。現在、有効なHAMの治療方法は確立されておらず、専ら対症療法がおこなわれている。
治療方法の一つとして、抗CCR4抗体を用いる治療方法が、HTLV-1感染細胞を減少させ、HAMの脊髄炎症を軽減し、症状改善効果を奏することが証明されている(非特許文献1、特許文献5)。
[1]
RGMa阻害物質を含む、HTLV-1関連脊髄症(HAM)の治療又は予防剤。
[2]
前記RGMa阻害物質が、RGMaを認識する抗体である、[1]に記載のHAMの治療又は予防剤。
[3]
それを必要とするHTLV-1関連脊髄症(HAM)患者に薬理学的に有効量のRGMa阻害物質を投与することを含む、HAMの治療方法。
[4]
前記RGMa阻害物質が、RGMaを認識する抗体である、[3]に記載のHAMの治療方法。
RGMa阻害物質としては、例えば、RGMaを阻害する活性を有する化合物、及びRGMaを認識する抗体等が挙げられる。
RGMa遺伝子としては、例えば配列番号1に示される塩基配列からなるヒトRGMa遺伝子、配列番号2に示される塩基配列からなるRGMa遺伝子等が挙げられるが、これに限定されるものではない。種々の生物由来のRGMa遺伝子の塩基配列の情報は、GenBank等のデータベースから取得することができる。
shRNAは、一本鎖RNA中に部分的に回文状の塩基配列を含むことにより、分子内に二本鎖構造を有し、3’末端に突出部を有する短いヘアピン構造からからなる約20塩基対以上の分子を指す。shRNAは、細胞内に導入された後、細胞内で約20塩基の長さに分解され、siRNAと同様に標的であるRGMa遺伝子の発現を抑制することができる。
本発明におけるRGMa抗体は、例えば、マウス抗体、ヒト型CDR移植抗体、ヒト型キメラ抗体、ヒト化抗体、又は完全ヒト抗体であってもよく、低分子抗体であってもよい。これらの抗体は、1種単独で用いてもよく、2種以上を組み合わせて用いてもよい。
得られた抗体がRGMa抗体であることは、RGMa活性を指標とし確認することができる。
RGMaとしては、例えば配列番号3に示されるアミノ酸配列を含むヒトRGMa、配列番号4に示されるアミノ酸配列を含むRGMa等が挙げられる。種々の生物由来のRGMaを免疫原として用いることができる。RGMaのアミノ酸配列は公知のデータベースであるProtein Data Bank等から取得することができる。
本発明において用いられるRGMa抗体がモノクローナル抗体であるとき、当該モノクローナル抗体は、上記免疫された哺乳動物から得た免疫細胞、例えば脾細胞とミエローマ細胞とを融合させてハイブリドーマを得、当該ハイブリドーマの培養物から抗体を採取することによって得ることができる。また、上記モノクローナル抗体は、抗体遺伝子をハイブリドーマからクローニングし、適当なベクターに組み込んで、これを宿主細胞に導入し、遺伝子組換え技術を用いて組換え型のモノクローナル抗体を産生させることもできる。さらに、上記モノクローナル抗体は、ファージディスプレイ法を用いて作製することもできる。
また、本発明において用いられるRGMa抗体としては、例えば、株式会社免疫生物研究所(IBL)製、R&D Systems社製等の市販品として入手することもできる。
GTTPDY(配列番号7);
FQATHDPLT(配列番号10);
ARRNEYYGSSFFDY(配列番号13);
LQGYIPPRT(配列番号16);及び
該配列の1つと少なくとも50%の配列同一性を有する修飾CDRアミノ酸配列;
からなる群から選択されるアミノ酸配列を含む少なくとも1つのCDRを含むことが好ましい。上記の配列同一性は、好ましくは80%以上であり、より好ましくは90%以上である。なお、本明細書においてアミノ酸は慣用的な一文字表記又は三文字表記で示されることがある。
また、本発明における、軽鎖及び重鎖のアミノ酸配列で規定される抗体においては、CDRのアミノ酸配列が所定の配列から不変であり、CDR以外が変異等によって変化していてもよい。CDR以外に変異等があるとき、相同性は90%以上であることが好ましい。
また、RGMa抗体は、重鎖可変ドメイン及び軽鎖可変ドメインとして、配列番号41、42、43、44、45、46、47、48及び49から選択される少なくとも1つの重鎖可変ドメイン;及び/又は配列番号50、51及び52から選択される少なくとも1つの軽鎖可変ドメインを含むことが好ましい。
EEVVNAVEDWDSQG(配列番号53)
NQQIDFQAFHTNAE(配列番号54)
PTAPETFPYET(配列番号55)
KLPVEDLYYQA(配列番号56)
LYERTRDLPGRAAAGL(配列番号57)
で表されるアミノ酸配列を有する一つ以上のペプチドに結合することが好ましい。
RGMa抗体は、配列番号53及び/又は配列番号54で表されるアミノ酸配列を有するペプチドに結合することがより好ましく、配列番号53及び/又は配列番号54で表されるアミノ酸配列を有するペプチドと、配列番号55及び/又は配列番号56で表されるアミノ酸配列を有するペプチドに結合することがより好ましい。
RGMa抗体は、ヒトRGMaのアミノ酸配列における250番目以降に結合する抗体であることが好ましい。
RGMa抗体は、配列番号53と配列番号54、かつ配列番号55又は配列番号56で表されるアミノ酸配列を有するペプチドに結合することがより好ましい。
本発明において、RGMa抗体は、医薬としてヒトに投与する場合、副作用の観点から、ヒト化抗体またはヒト抗体が望ましい。
RGMa抗体は、RGMaタンパク質またはその部分断片(例えば、配列番号53~57の一つ以上を含む断片)を抗原として認識するポリクローナル抗体及び/又はモノクローナル抗体の部分断片として用いてもよく、低分子抗体であってもよい。
LCDR1:RASQDISSYLN(配列番号58)
LCDR2:YTSRLHS(配列番号59)
LCDR3:QQLNTLP(配列番号60)
HCDR1:DAWMD(配列番号61)
HCDR2:EIRSKANNHATYYAESVKG(配列番号62)及び
HCDR3:RDGAY(配列番号63)を含む、
又は、
LCDR1:RSSQSLVHSNGNTYLH(配列番号64)
LCDR2:KVSNRFS(配列番号65)
LCDR3:SQSTHVP(配列番号66)
HCDR1:TSYYWN(配列番号67)
HCDR2:YISYDGTNNYNPSLKN(配列番号68)及び
HCDR3:SFGを含む、
単離されたRGMa抗体、またはその抗原結合断片であってもよい。
各CDR配列においては、1又は数個のアミノ酸が置換、欠失、及び/または付加されていてもよく、例えば、1個又は2個のアミノ酸が置換、欠失、及び/または付加されていてもよい。
EVQLVESGGGLVQPGRSLRLSCTASGFTFSDAWMDWVRQAPGKGLEWVAEIRSKANNHATYYAESVKGRFTISRDDSKSIVYLQMNSLRTEDTALYYCTRRDGAYWGKGTTVTVSS(配列番号95)
又は該アミノ酸配列に少なくとも90%の同一性のあるアミノ酸配列を含み、
軽鎖可変領域(VL)が
DIQMTQSPSSVSASVGDRVTITCRASQDISSYLNWYQQKPGKAPKLLIYYTSRLHSGVPSRFSGSGSGTDFTLTISSLQPEDFASYFCQQLNTLPWTFGGGTKVEME(配列番号96)
又は該アミノ酸配列に少なくとも90%の同一性のあるアミノ酸配列を含む、単離されたRGMa抗体、又はその抗原結合断片が好ましい。
定常領域をヒト由来としたマウス/ヒトキメラ抗体であってもよく、CDR以外をヒト由来としたヒト化抗体であってもよい。
(a1)配列番号97に記載のアミノ酸配列を含むLCDR1、配列番号98に記載のアミノ酸配列を含むLCDR2及び配列番号99に記載のアミノ酸配列を含むLCDR3を含む軽鎖可変領域、並びに配列番号100に記載のアミノ酸配列を含むHCDR1、配列番号101に記載のアミノ酸配列を含むHCDR2及び配列番号102に記載のアミノ酸配列を含むHCDR3を含む重鎖可変領域を含む抗RGMa抗体、またはその抗原結合断片、
(b1)配列番号97に記載のアミノ酸配列を含むLCDR1、配列番号98に記載のアミノ酸配列を含むLCDR2及び配列番号103に記載のアミノ酸配列を含むLCDR3を含む軽鎖可変領域、並びに配列番号100に記載のアミノ酸配列を含むHCDR1、配列番号101に記載のアミノ酸配列を含むHCDR2及び配列番号102に記載のアミノ酸配列を含むHCDR3を含む重鎖可変領域を含む抗RGMa抗体、またはその抗原結合断片、
(c1)配列番号97に記載のアミノ酸配列を含むLCDR1、配列番号98に記載のアミノ酸配列を含むLCDR2及び配列番号104に記載のアミノ酸配列を含むLCDR3を含む軽鎖可変領域、並びに配列番号100に記載のアミノ酸配列を含むHCDR1、配列番号101に記載のアミノ酸配列を含むHCDR2及び配列番号102に記載のアミノ酸配列を含むHCDR3を含む重鎖可変領域を含む抗RGMa抗体、またはその抗原結合断片、
(d1)配列番号97に記載のアミノ酸配列を含むLCDR1、配列番号98に記載のアミノ酸配列を含むLCDR2及び配列番号105に記載のアミノ酸配列を含むLCDR3を含む軽鎖可変領域、並びに配列番号100に記載のアミノ酸配列を含むHCDR1、配列番号101に記載のアミノ酸配列を含むHCDR2及び配列番号102に記載のアミノ酸配列を含むHCDR3を含む重鎖可変領域を含む抗RGMa抗体、またはその抗原結合断片、
(e1)配列番号97に記載のアミノ酸配列を含むLCDR1、配列番号98に記載のアミノ酸配列を含むLCDR2及び配列番号106に記載のアミノ酸配列を含むLCDR3を含む軽鎖可変領域、並びに配列番号100に記載のアミノ酸配列を含むHCDR1、配列番号101に記載のアミノ酸配列を含むHCDR2及び配列番号102に記載のアミノ酸配列を含むHCDR3を含む重鎖可変領域を含む抗RGMa抗体、またはその抗原結合断片、
(f1)配列番号97に記載のアミノ酸配列を含むLCDR1、配列番号98に記載のアミノ酸配列を含むLCDR2及び配列番号107に記載のアミノ酸配列を含むLCDR3を含む軽鎖可変領域、並びに配列番号100に記載のアミノ酸配列を含むHCDR1、配列番号101に記載のアミノ酸配列を含むHCDR2及び配列番号102に記載のアミノ酸配列を含むHCDR3を含む重鎖可変領域を含む抗RGMa抗体、またはその抗原結合断片、
(g1)配列番号97に記載のアミノ酸配列を含むLCDR1、配列番号98に記載のアミノ酸配列を含むLCDR2及び配列番号108に記載のアミノ酸配列を含むLCDR3を含む軽鎖可変領域、並びに配列番号100に記載のアミノ酸配列を含むHCDR1、配列番号101に記載のアミノ酸配列を含むHCDR2及び配列番号102に記載のアミノ酸配列を含むHCDR3を含む重鎖可変領域を含む抗RGMa抗体、またはその抗原結合断片、及び
(h1)配列番号97に記載のアミノ酸配列を含むLCDR1、配列番号98に記載のアミノ酸配列を含むLCDR2及び配列番号109に記載のアミノ酸配列を含むLCDR3を含む軽鎖可変領域、並びに配列番号100に記載のアミノ酸配列を含むHCDR1、配列番号101に記載のアミノ酸配列を含むHCDR2及び配列番号102に記載のアミノ酸配列を含むHCDR3を含む重鎖可変領域を含む抗RGMa抗体、またはその抗原結合断片。
後述の実施例及び図13に示されるように、HAM患者ではHAM患者由来細胞によって神経細胞死、すなわち脊髄組織の損傷及び変性の誘導が起こっている。
本発明者らが検討した結果、後述する実施例に示したように、HAM患者の主なHTLV-1感染細胞であるCD4陽性T細胞においてRGMaが顕著に発現しており、HAMにはRGMaの発現が関与することがわかった。また、RGMa抗体を用いてRGMaの阻害を行う試験の結果、RGMaがCXCL10、IL-2、及びIL-10の産生に関与することがわかった。
CXCL10は、IFN-γに応答してHAM病原性のHTLV-1感染T細胞から産生されるタンパク質であり、HAMの病態を誘導することが知られており(Brain 2013)、またHAMの症状の進行速度とも強く相関することが知られている(PLoS Negl Trop Dis 2013)。HAM患者の細胞にRGMa抗体を作用させると、このCXCL10の産生が抑えられた。また、IL-10はサイトカインの一つであり、炎症を抑制するようにはたらく。HAM患者の細胞にRGMa抗体を作用させると、IL-10の産生が顕著に増加した。
HAMに真の治療効果をもたらすためには、神経細胞の傷害を抑えることが求められる。HAM患者のHTLV-1感染細胞ではHTLV-1-taxの発現レベルが高く(Blood 2002)、HTLV-1-taxが病態形成に重要であることが示されている(J Clin Invest 2014)。本発明では、HTLV-1-taxがRGMaの発現を誘導し、RGMa発現レベルが高いHAM患者由来細胞が神経細胞死を誘導することが示され、重要なことに、HTLV-1-tax発現細胞による神経細胞死をRGMa阻害物質が抑制することが示された。
以上のように、RGMa阻害物質は、HAMの炎症病態の誘導を抑え、また、HAM患者細胞による炎症反応を抑制することができるため、HAMを治療又は予防することができる。さらにRGMa阻害物質はHAM特有の炎症反応を抑制するのみでなく、HAM患者の病原的な細胞であるHTLV-1-tax発現細胞による神経細胞死を抑制することができるため、HAMを治療又は予防することができる。
製剤化の形態としては、例えば、錠剤、被覆錠剤、丸剤、散剤、顆粒剤、カプセル剤、液剤、懸濁剤、乳剤等の経口剤;注射剤、輸液、坐剤、軟膏、パッチ剤等の非経口剤;が挙げられる。
担体又は添加剤の配合割合については、医薬品分野において通常採用されている範囲に基づいて適宜設定すればよい。
上記担体としては、特に制限されないが、例えば、水、生理食塩水、その他の水性溶媒、及び、水性又は油性基剤等が挙げられる。上記添加剤としては、特に制限されないが、賦形剤、結合剤、pH調整剤、崩壊剤、吸収促進剤、滑沢剤、着色剤、矯味剤、及び香料等が挙げられる。
RGMa抗体を含む注射剤又は輸液は、溶液、懸濁液又は乳濁液として用いることができる。その溶剤としては、例えば、注射用蒸留水、生理食塩水、ブドウ糖溶液、及び等張液(例えば、塩化ナトリウム、塩化カリウム、グリセリン、マンニトール、ソルビトール、ホウ酸、ホウ砂、プロピレングリコール等の溶液)等を用いることができる。これらの溶剤は1種単独で用いてもよく、2種以上を組み合わせて用いてもよい。
安定剤としては、例えば、アルブミン、グロブリン、ゼラチン、マンニトール、グルコース、デキストラン、エチレングリコール、プロピレングリコール、アスコルビン酸、亜硫酸水素ナトリウム、チオ硫酸ナトリウム、EDTAナトリウム、クエン酸ナトリウム、ジブチルヒドロキシトルエン等を用いることができる。
溶解補助剤としては、例えば、アルコール(例えば、エタノール等)、ポリアルコール(例えば、プロピレングリコール、ポリエチレングリコール等)、非イオン性界面活性剤(例えば、ポリソルベート80(登録商標)、HCO-50等)等を用いることができる。
懸濁化剤としては、例えば、モノステアリン酸グリセリン、モノステアリン酸アルミニウム、メチルセルロース、カルボキシメチルセルロース、ヒドロキシメチルセルロース、ラウリル硫酸ナトリウム等を用いることができる。
乳化剤としては、例えば、アラビアゴム、アルギン酸ナトリウム、トラガント等を用いることができる。
無痛化剤としては、例えば、ベンジルアルコール、クロロブタノール、ソルビトール等を用いることができる。
緩衝剤としては、例えば、リン酸緩衝液、酢酸緩衝液、ホウ酸緩衝液、炭酸緩衝液、クエン酸緩衝液、トリス緩衝液、等を用いることができる。
保存剤としては、例えば、パラオキシ安息香酸メチル、パラオキシ安息香酸エチル、パラオキシ安息香酸プロピル、パラオキシ安息香酸ブチル、クロロブタノール、ベンジルアルコール、塩化ベンザルコニウム、デヒドロ酢酸ナトリウム、エデト酸ナトリウム、ホウ酸、ホウ砂等を用いることができる。
防腐剤としては、例えば、塩化ベンザルコニウム、パラオキシ安息香酸、クロロブタノール等を用いることができる。
pH調整剤としては、例えば、塩酸、水酸化ナトリウム、リン酸、酢酸等を用いることができる。
RGMa阻害物質が非ウイルスベクター形態のとき、投与の方法としては、リポソームを用いて核酸分子を導入する方法(リポソーム法、HVJ-リポソーム法、カチオニックリポソーム法、リポフェクション法、リポフェクトアミン法等)、マイクロインジェクション法、遺伝子銃(Gene Gun)でキャリア(金属粒子)とともに核酸分子を細胞に移入する方法等を上げることができる。
siRNA又はshRNAを、ウイルスベクターを用いて生体に投与する場合、組換えアデノウイルス、レトロウイルス等のウイルスベクターを利用することができる。無毒化したレトロウイルス、アデノウイルス、アデノ随伴ウイルス、ヘルペスウイルス、ワクシニアウイルス、ポックスウイルス、ポリオウイルス、シンドビスウイルス、センダイウイルス、SV40等のDNAウイルス又はRNAウイルスに、siRNAまたはshRNAを発現するDNAを導入し、細胞又は組織にこの組換えウイルスを感染させることにより、細胞又は組織内に遺伝子を導入することができる。
投与量は、目的、疾患の重篤度、患者の年齢、体重、性別、既往歴、有効成分の種類などを考慮して、適宜設定される。
図1に示すように、HTLV-1感染未発症者の検体については、CD4陽性細胞のうち、HTLV-1感染細胞の指標であるCADM1と、CD7により分類された群P、D、Nについて、mRNAの発現解析を行った。
またHAMは、HAM患者末梢血単核細胞から、磁気ビーズで濃縮したCD4陽性細胞とCD4陰性細胞も4例ずつ解析した。HTLV-1非感染健常者(Normal)についても同様に分離した。末梢血単核細胞は、PBMCとも記載する。ATL(smodering, chronic, acuteの各病型)は、主にCD4陽性細胞からなるPBMCを用いた。
アジレントテクノロジー株式会社製の一色マイクロアレイ遺伝子発現解析法を行った。
正常T細胞 4例(Normal.CD4)、HAM患者由来CD4陽性T細胞 4例(HAM.CD4)、健常人由来CD4陽性/CADM1陰性/CD7陽性T細胞 3例(Normal.P)、HTLV-1感染者CD4陽性/CADM1陰性/CD7陽性T細胞5例 (P群)、HTLV-1感染者CD4陽性/CADM1陽性/CD7陽性T細胞 5例(D群)、HTLV-1感染者CD4陽性/CADM1陽性/CD7陰性T細胞5例(N群)、急性型ATL患者CD4陽性/CADM1陽性/CD7陰性T細胞 3例(Acute.N)、健常人CD4陽性T細胞 21例(Normal.CD4.1)、くすぶり型ATL患者由来PBMC 3例(Smoldering)、慢性型ATL患者由来PBMC 20例(Chronic)、急性型ATL患者由来PBMC 26例(Acute)について、アジレントテクノロジー株式会社のHuman Gene Expression 4x44K Microarrayを用いて全遺伝子発現データを取得し、中央値で標準化したのち、RGMa遺伝子レベルをグラフ化した。
グラフではアレイの蛍光強度のLog2値をプロットした。HAM患者由来CD4陽性T細胞群は、その他の全ての群と有意差をもって上昇していた(P < 0.05)。グラフを図2に示す。
HAM患者5名の末梢血から分離したPBMCよりhuman CD4+ isolation kit(Miltenyi Biotec)を用いてCD4陽性T細胞を分離し、HTLV-1感染細胞が多数含まれる細胞集団とした。同様に健常者4名のPBMCからCD4陽性T細胞を分離し、コントロール群とした。
分離したCD4陽性T細胞から全RNAを回収し、ReverTra Ace(東洋紡)を用いてcDNAを作製した。作製されたcDNAを用いてHAM患者CD4陽性T細胞と健常者CD4陽性T細胞との間におけるRGMaの発現量の差をReal time PCRにより解析した。インターナルコントロールには18srRNAを用いた。
解析の結果のグラフを図3に示す。グラフにおけるHD-CD4+は、コントロール群を指し、HAM-CD4+は、HAM患者の群を指す。
HAM患者PBMCにClear Back(MBL)を用いてFc block処理した後、抗CD3-PECy7(TONBO), CD4-FITC(eBioscience), CD14-PE(eBioscience)抗体を加えて4℃で30分間染色を行った。
抗体染色されたPBMCを洗浄した後にAriaIIIu(BD)を用いてFACSソーティングを行い、CD3陽性CD4陰性細胞(CD3+CD4-)、CD3陽性CD4陽性細胞(CD3+CD4+)、CD3陰性CD14陰性細胞(CD3-CD14-)およびCD3陰性CD14陽性細胞(CD3-CD14+)の分離回収を行った。
回収された各細胞から全RNAを回収し、ReverTra Ace(東洋紡)を用いてcDNAを作製した。作製されたcDNAを用いて細胞種間におけるRGMaの発現量の差をReal time PCRにより解析した。インターナルコントロールには18s rRNAを用いた。解析の結果のグラフを図4に示す。
HAM-PBMCでは、感染細胞が多く存在するCD3陽性CD4陽性細胞(CD3+CD4+)においてRGMaの発現が最も高いことがわかった。
HAM患者2名のPBMCを培地(10% FBS(GIBCO)を含むRPMI1640培地(wako))に懸濁し1e5 cellsずつ96 well round bottom plateに10well分播種した後に1, 3, 5, 7日間培養した。
培養を行っていないDay0のPBMCとともに各期間培養したPBMCより全RNAを抽出してReverTra Ace(東洋紡)を用いてcDNAを作製した。HAM患者PBMCは培養するとHTLV-1ウイルスが過剰に発現することが知られており、作製されたcDNAを用いてHAM患者PBMCの培養に伴う、すなわち、ウイルスの発現に伴うRGMaの発現変化について、Real time PCRにより解析した。インターナルコントロールには18s rRNAを用いた。解析の結果を示すグラフを図5に示す。
正常T細胞 3例(Normal T cell)、HAM患者由来CD4陽性T細胞 4例(HAM)、急性型ATL患者由来PBMC 3例(ATL)について、Agilent Technologies社のSurePrint G3 Human Promoter 2x400K Microarrayを用いて全プロモーター上のH3K27me3レベルを取得し、標準化したのち、RGMa遺伝子の転写開始点から-2,916bp上流付近のH3K27me3レベルをグラフ化した。
グラフを図6に示す。グラフではアレイの蛍光強度のLog2値をプロットした。なお、図中のHAM50及びHAM123は、それぞれCD4陽性T細胞を得たHAM患者を指す。HAM患者由来のCD4陽性T細胞では、RGMaの遺伝子発現抑制が解除されていることが示唆された。
ヒトCD4陽性T細胞白血病細胞株Jurkatに対し、HTLV-1 TaxをコードするcDNAを挿入するレンチウイルスベクターを導入し、導入後3日間の継時的なRGMa遺伝子mRNAのレベルを定量的RT-PCRで測定した。RPL19遺伝子mRNAも測定し、内部標準として使用した。
定量の結果を表すグラフを図7に示す。HTLV-1ウイルスによってRGMa発現が引き起こされることが示唆された。
JPX9細胞を培地(10% FBSを含むRPMI1640培地)で24時間培養し、HTLV-1-tax発現を誘導する塩化カドミウム(CdCl2;ナカライテスク)を終濃度20μMとなるように添加した後、1、2、及び3日間培養した。塩化カドミウム処理及び未処理のJPX9細胞について、Tax及びRGMaのタンパク質発現についてFACS解析した。
塩化カドミウム処理及び未処理のJPX9細胞をそれぞれ洗浄し、Foxp3/Transcription Factor Staining Buffer Kit (eBioscience)を用いて細胞透過処理を行った。その後、抗Tax-FITC抗体(Lt-4:琉球大学 田中先生より分与)を添加して4℃で1時間処理することにより細胞内に発現するTaxタンパク質を染色した。染色されたTaxタンパク質はCantoIIを用いたFACS解析により検出した。
塩化カドミウム処理及び未処理のJPX9細胞をそれぞれ洗浄し、抗RGMa抗体(株式会社免疫生物研究所(IBL)製)を添加して4℃で30分間処理した。その後、細胞を洗浄し、抗mouse IgG-PE抗体(BioLegend)を添加し4℃で30分間反応させ、JPX9に発現するRGMaタンパク質を染色した。染色されたRGMaタンパク質はCantoIIを用いたFACS解析により検出した。
図8にTax及びRGMaのタンパク質発現の解析結果を示す。
(自発的増殖活性へのRGMa抗体の作用)
4例のHAM患者PBMCを培地(10% FBSを含むRPMI1640培地)に懸濁し、1e5 cellsずつ96 well round bottom plateに播種し、終濃度10μg/mlとなるようRGMa抗体(R&D Systems製)を加え合計0.1mlの培養液において37℃、5% CO2条件下で7日間培養した。
何も添加しなかった群(Medium)、同濃度のNormal Goat IgG (Santa Cruz Biotechnology)添加群(Normal IgG)、及び1μg/ml プレドニゾロン(PSL)(フナコシ)添加群をコントロールとした。
培養開始から6日後、各ウェルに1μ Ci 3H-Thymidineを添加し、37℃、5% CO2条件下で16時間培養を行った。その後、培養細胞をセルハーべスター(Tomtec MH3 PerkinElmer)を用いてガラスフィルター(Printed Filtermat A PerkinElmer)に吸着させ、乾燥させた後に固体シンチレータMeltilex-A(PerkinElmer)を染み込ませ、MicroBeta (WALLAC MicroBeta TriLux 1450-021)を用いて細胞に取り込まれた3H-Thymidine量の測定を行った。各HAM患者PBMCのMedium群における3H-Thymidineのカウントの平均を100%として、各群の相対値を算出し、HAM患者4例の3H-Thymidine取り込み率の平均値を求めた。結果を図9に示す。
4例のHAM患者PBMCを培地(10% FBSを含むRPMI1640培地)に懸濁し1e5 cellsずつ96 well round bottom plateに播種し、終濃度10μg/mlとなるようRGMa抗体(R&D Systems製)を加え合計0.1mlの培養液において37℃、5% CO2条件下で7日間培養した。
何も添加しなかった群(Medium)、同濃度のNormal Goat IgG (Santa Cruz Biotechnology)添加群(Normal IgG)、1μg/ml プレドニゾロン(PSL)(フナコシ)添加群をコントロールとした。
培養開始から7日後、遠心し上清を除いた細胞塊からゲノムDNA抽出を行った。抽出されたゲノムDNAを用いて、リアルタイムPCRによりHTLV-1プロウイルス量(感染細胞率)の測定を行った。
各HAM患者PBMCのMedium群おけるHTLV-1プロウイルス量を100%として、各群におけるHTLV-1プロウイルス量の相対値を算出し、HAM患者4例のHTLV-1プロウイルス量の平均値を求めた。結果を図10に示す。
HAM患者PBMCからのCXCL10産生に対するRGMa抗体の作用について解析するため、4例のHAM患者PBMCを培地(10% FBSを含むRPMI1640培地)に懸濁し1e5 cellsずつ96 well round bottom plateに播種し、終濃度10μg/mlとなるようRGMa抗体(R&D Systems製)を加え合計0.1mlの培養液において37℃、5% CO2条件下で7日間培養した。
何も添加しなかった群(Medium)、同濃度のNormal Goat IgG (Santa Cruz Biotechnology)添加群(Normal IgG)、1μg/ml プレドニゾロン(PSL)(フナコシ)添加群をコントロールとした。
培養開始から7日後、培養液を遠心分離して培養上清のみを回収した。培養上清中のCXCL10濃度はCytokine Beads Array kit(BD Biosciences)を用いて、フローサイトメーターFACSCantoII(BD Biosciences)により測定した。
Medium群の培養液中におけるCXCL10濃度を100%として、各群における各群の培養液中CXCL10濃度の相対値を算出し、HAM患者4例のCXCL10濃度の平均値を求めた。結果を図11に示す。
HAM患者PBMCにおける各種サイトカインの産生に対するRGMa抗体の作用について解析するため、4例のHAM患者PBMCを培地(10% FBSを含むRPMI1640培地)に懸濁し1e5 cellsずつ98 well round bottom plateに播種し、終濃度10μg/mlとなるようRGMa抗体(R&D Systems製)を加え合計0.1mlの培養液において37℃、5% CO2条件下で7日間培養した。
何も添加しなかった群(Medium)、同濃度のNormal Goat IgG (Santa Cruz Biotechnology)添加群(Normal IgG)、1μg/ml プレドニゾロン(PSL)(フナコシ)添加群をコントロールとした。
培養開始から7日後、培養液を遠心分離して培養上清のみを回収した。培養上清中のIFNγ、TNF、IL-2、IL-10濃度はCytokine Beads Array kit(BD Biosciences)を用いて、フローサイトメーターFACSCantoII(BD Biosciences)により測定した。
Medium群の培養液中における各サイトカイン濃度を100%として、各培養条件におけるサイトカイン濃度の相対値を算出し、HAM患者4例の平均値を求めた。
神経細胞株NB-1又はSK-N-ASを6well plateに播種し24時間培養後、健常者(HD)又はHAM患者PBMCを添加し共培養を行った。共培養開始から48時間後に培地とともに添加したPBMCを除去し、PBSで洗浄後に神経細胞株を回収した。
回収された各神経細胞株について、アポトーシスによりDNA断片化を起こしている細胞を特異的に検出するTUNEL法(MEBSTAIN Apoptosis TUNEL Kit Direct(MBL))により解析を行った。図13に解析結果を示す。ヒストグラムにおけるX軸が、DNA断片化陽性の強度を示す。HAM由来細胞はHD由来細胞と比較して、神経細胞株にアポトーシスをより強く誘導した。
具体的には、以下の<実験手順>にしたがって、細胞死の解析を行った。
<実験手順>
神経細胞株NB-1、SK-N-ASをそれぞれ6well plateに播種し、24時間培養した。
次に、HDまたはHAM-PBMCを添加(神経細胞株の播種数の2倍量)し、48時間培養した。
その後、4%パラホルムアルデヒドを用いて細胞固定し、70%エタノールにより細胞透過処理を行った。
DNA nick end labelingを行うため、MEBSTAIN Apoptosis TUNEL Kit Direct(MBL)のTdT solution 20uL(TdT buffer II : TdT : FITC-dUTP = 18 : 1 : 1)に細胞を懸濁させ、37℃で60分間反応させた後、FACS解析を行った。
神経細胞株NB-1を6well plateに播種し24時間培養後、未刺激JPX9(JPX9(-))または20μM塩化カドミウムを24時間添加してTax発現を誘導したJPX9(JPX9(+CdCl2))を加えて共培養を行った。また、NB-1細胞と(JPX9(+CdCl2)の共培養に終濃度10μg/ml になるようにNormal Mouse IgG2b(MBL)またはRGMa抗体(IBL)を加えた。共培養開始から48時間後に培地とともに添加したJPX9を除去し、PBSで洗浄後に神経細胞株を回収した。回収された各神経細胞株について、アポトーシスによりDNA断片化を起こしている細胞を特異的に検出するTUNEL法(MEBSTAIN Apoptosis TUNEL Kit Direct(MBL))により解析を行った。図14に解析結果を示す。ヒストグラムにおけるX軸が、DNA断片化陽性の強度を示す。
具体的には、以下の<実験手順>にしたがって、細胞死の解析を行った。
<実験手順>
神経細胞株(NB-1)を6well plateに播種し、24時間培養した。
次に、JPX9(-)またはJPX9(+CdCl2)をNB-1細胞に添加して共培養した。なお、JPX9(-)またはJPX9(+CdCl2)の細胞数はNB-1細胞の播種数の2倍量とした。JPX9(+CdCl2)については、塩化カドミウムを除去するために培地10mlで3回洗浄してNB-1細胞に加えた。
続いて、NB-1細胞と(JPX9(+CdCl2)の共培養にNormal Mouse IgG2b(MBL)または抗RGMa抗体(IBL)を終濃度10μg/mlとなるように加え、48時間培養した。
その後、4%パラホルムアルデヒドを用いて細胞固定し、70%エタノールにより細胞透過処理を行い、抗CD45-V450抗体を加えて染色した。
DNA nick end labelingを行うため、MEBSTAIN Apoptosis TUNEL Kit Direct(MBL)のTdT solution 20uL(TdT buffer II : TdT : FITC-dUTP = 18 : 1 : 1)に細胞を懸濁し、37℃で60分間反応させた後、FACS解析を行った。
Claims (4)
- RGMa阻害物質を含む、HTLV-1関連脊髄症(HAM)の治療又は予防剤。
- 前記RGMa阻害物質が、RGMaを認識する抗体である、請求項1に記載のHAMの治療又は予防剤。
- それを必要とするHTLV-1関連脊髄症(HAM)患者に薬理学的に有効量のRGMa阻害物質を投与することを含む、HAMの治療方法。
- 前記RGMa阻害物質が、RGMaを認識する抗体である、請求項3に記載のHAMの治療方法。
Priority Applications (18)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202310983277.XA CN116785439A (zh) | 2018-07-19 | 2019-07-19 | Htlv-1相关性脊髓病(ham)治疗或预防剂、及ham的治疗方法 |
| CN202310983220.XA CN116942831A (zh) | 2018-07-19 | 2019-07-19 | Htlv-1相关性脊髓病(ham)治疗或预防剂、及ham的治疗方法 |
| CA3112511A CA3112511A1 (en) | 2018-07-19 | 2019-07-19 | Therapeutic or prophylactic agent for htlv-1-associated myelopathy (ham), and method for treating ham |
| EP19838340.8A EP3824907A4 (en) | 2018-07-19 | 2019-07-19 | AGENT FOR THE TREATMENT OR PREVENTION OF HTLV-1 ASSOCIATED MYELOPATHY (HAM), AND METHOD OF TREATMENT OF HAM |
| JP2020531380A JP7150286B2 (ja) | 2018-07-19 | 2019-07-19 | Htlv-1関連脊髄症(ham)治療又は予防剤、及びhamの治療方法 |
| CN201980048012.2A CN112566664B (zh) | 2018-07-19 | 2019-07-19 | Htlv-1相关性脊髓病(ham)治疗或预防剂、及ham的治疗方法 |
| BR112021000949-3A BR112021000949A2 (pt) | 2018-07-19 | 2019-07-19 | agente terapêutico ou profilático para mielopatia associada a htlv-1 (ham) e método para tratar ham |
| PE2021000080A PE20211376A1 (es) | 2018-07-19 | 2019-07-19 | Agente terapeutico o profilactico para la mielopatia asociada al htlv-1 (ham), y metodo de tratamiento del ham |
| US17/261,480 US20210269535A1 (en) | 2018-07-19 | 2019-07-19 | Therapeutic or prophylactic agent for htlv-1-associated myelopathy (ham), and method for treating ham |
| KR1020217005032A KR20210034055A (ko) | 2018-07-19 | 2019-07-19 | Htlv-1 관련 척수증(ham) 치료 또는 예방제, 및 ham의 치료 방법 |
| CN202310983521.2A CN116850291A (zh) | 2018-07-19 | 2019-07-19 | Htlv-1相关性脊髓病(ham)治疗或预防剂、及ham的治疗方法 |
| MX2021000732A MX2021000732A (es) | 2018-07-19 | 2019-07-19 | Agente terapeutico o profilactico para la mielopatia asociada al htlv-1 (ham), y metodo para tratar ham. |
| SG11202100561QA SG11202100561QA (en) | 2018-07-19 | 2019-07-19 | Therapeutic or prophylactic agent for htlv-1-associated myelopathy (ham), and method for treating ham |
| AU2019306721A AU2019306721A1 (en) | 2018-07-19 | 2019-07-19 | Therapeutic or prophylactic agent for HTLV-1-associated myelopathy (HAM), and method for treating HAM |
| PH12021550134A PH12021550134A1 (en) | 2018-07-19 | 2021-01-18 | Therapeutic or prophylactic agent for htlv-1-associated myelopathy (ham), and method for treating ham |
| IL280257A IL280257A (en) | 2018-07-19 | 2021-01-18 | Therapeutic or prophylactic agent for HTLV-1-associated myelopathy (HAM) and method for treating HAM |
| ZA2021/01113A ZA202101113B (en) | 2018-07-19 | 2021-02-18 | Therapeutic or prophylactic agent of htlv-1-associated myelopathy (ham), and method for treating ham |
| JP2022148180A JP7475011B2 (ja) | 2018-07-19 | 2022-09-16 | Htlv-1関連脊髄症(ham)治療又は予防剤、及びhamの治療方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2018135925 | 2018-07-19 | ||
| JP2018-135925 | 2018-07-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020017629A1 true WO2020017629A1 (ja) | 2020-01-23 |
Family
ID=69163691
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2019/028431 WO2020017629A1 (ja) | 2018-07-19 | 2019-07-19 | Htlv-1関連脊髄症(ham)治療又は予防剤、及びhamの治療方法 |
Country Status (16)
| Country | Link |
|---|---|
| US (1) | US20210269535A1 (ja) |
| EP (1) | EP3824907A4 (ja) |
| JP (2) | JP7150286B2 (ja) |
| KR (1) | KR20210034055A (ja) |
| CN (4) | CN116785439A (ja) |
| AU (1) | AU2019306721A1 (ja) |
| BR (1) | BR112021000949A2 (ja) |
| CA (1) | CA3112511A1 (ja) |
| IL (1) | IL280257A (ja) |
| MX (1) | MX2021000732A (ja) |
| PE (1) | PE20211376A1 (ja) |
| PH (1) | PH12021550134A1 (ja) |
| SG (1) | SG11202100561QA (ja) |
| TW (1) | TWI825131B (ja) |
| WO (1) | WO2020017629A1 (ja) |
| ZA (1) | ZA202101113B (ja) |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005087268A1 (ja) * | 2004-03-11 | 2005-09-22 | Bioclues, Inc | 軸索再生促進剤 |
| JP2006516594A (ja) * | 2003-01-31 | 2006-07-06 | エンスティテュ ナショナル ドゥ ラ サンテ エ ドゥ ラ ルシェルシュ メディカル(イ エヌ エス ウ エール エム) | 抗−cd100抗体の使用 |
| JP2010100578A (ja) | 2008-10-24 | 2010-05-06 | St Marianna Univ School Of Medicine | Htlv−i関連脊髄症を治療または予防するための医薬、およびhtlv−i関連脊髄症の患者に対する抗体療法の効果を予測する方法 |
| WO2011071059A1 (ja) | 2009-12-09 | 2011-06-16 | 国立大学法人大阪大学 | T細胞活性化阻害剤、これを含有する医薬組成物およびt細胞活性化阻害物質のスクリーニング方法 |
| JP2013119531A (ja) * | 2011-12-07 | 2013-06-17 | St Marianna Univ School Of Medicine | Htlv−1関連脊髄症を治療または予防するための医薬および前記医薬を用いた抗体療法の治療効果の確認方法 |
| WO2014007303A1 (ja) * | 2012-07-06 | 2014-01-09 | 学校法人 聖マリアンナ医科大学 | Htlv-1関連脊髄症患者の治療方法および治療剤 |
| JP2014138599A (ja) | 2008-02-29 | 2014-07-31 | Abbott Gmbh & Co Kg | Rgmaタンパク質に対するモノクローナル抗体及びその使用 |
| WO2016175236A1 (ja) | 2015-04-28 | 2016-11-03 | 田辺三菱製薬株式会社 | RGMa結合タンパク質及びその使用 |
| WO2017210278A1 (en) * | 2016-06-01 | 2017-12-07 | Abbvie Inc. | Anti-repulsive guidance molecule a (rgma) antagonistic antibodies for treating spinal cord injury and pain |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101741605B1 (ko) | 2008-09-02 | 2017-06-15 | 도레이 배터리 세퍼레이터 필름 주식회사 | 미세다공성 고분자막, 이 미세다공성 고분자막의 제조방법 및 전지 세퍼레이터 필름으로서의 이 미세다공성 고분자막의 사용 |
| ES2778053T3 (es) * | 2011-01-18 | 2020-08-07 | Bioniz Llc | Composiciones para modular la actividad de la citocina gamma-c |
| JP6879905B2 (ja) | 2014-09-10 | 2021-06-02 | アッヴィ・ドイチュラント・ゲー・エム・ベー・ハー・ウント・コー・カー・ゲー | RGMa断片ベース診断アッセイ |
| EP3448875A4 (en) * | 2016-04-29 | 2020-04-08 | Voyager Therapeutics, Inc. | COMPOSITIONS FOR TREATING A DISEASE |
| JP6923215B2 (ja) * | 2016-08-03 | 2021-08-18 | 国立大学法人 鹿児島大学 | 抗htlv−1剤、htlv−1関連脊髄症(ham/tsp)治療薬 |
-
2019
- 2019-07-19 BR BR112021000949-3A patent/BR112021000949A2/pt unknown
- 2019-07-19 CA CA3112511A patent/CA3112511A1/en active Pending
- 2019-07-19 AU AU2019306721A patent/AU2019306721A1/en active Pending
- 2019-07-19 CN CN202310983277.XA patent/CN116785439A/zh active Pending
- 2019-07-19 CN CN202310983220.XA patent/CN116942831A/zh active Pending
- 2019-07-19 WO PCT/JP2019/028431 patent/WO2020017629A1/ja unknown
- 2019-07-19 MX MX2021000732A patent/MX2021000732A/es unknown
- 2019-07-19 CN CN201980048012.2A patent/CN112566664B/zh active Active
- 2019-07-19 US US17/261,480 patent/US20210269535A1/en active Pending
- 2019-07-19 TW TW108125746A patent/TWI825131B/zh active
- 2019-07-19 KR KR1020217005032A patent/KR20210034055A/ko unknown
- 2019-07-19 PE PE2021000080A patent/PE20211376A1/es unknown
- 2019-07-19 SG SG11202100561QA patent/SG11202100561QA/en unknown
- 2019-07-19 JP JP2020531380A patent/JP7150286B2/ja active Active
- 2019-07-19 CN CN202310983521.2A patent/CN116850291A/zh active Pending
- 2019-07-19 EP EP19838340.8A patent/EP3824907A4/en active Pending
-
2021
- 2021-01-18 PH PH12021550134A patent/PH12021550134A1/en unknown
- 2021-01-18 IL IL280257A patent/IL280257A/en unknown
- 2021-02-18 ZA ZA2021/01113A patent/ZA202101113B/en unknown
-
2022
- 2022-09-16 JP JP2022148180A patent/JP7475011B2/ja active Active
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006516594A (ja) * | 2003-01-31 | 2006-07-06 | エンスティテュ ナショナル ドゥ ラ サンテ エ ドゥ ラ ルシェルシュ メディカル(イ エヌ エス ウ エール エム) | 抗−cd100抗体の使用 |
| WO2005087268A1 (ja) * | 2004-03-11 | 2005-09-22 | Bioclues, Inc | 軸索再生促進剤 |
| JP2014138599A (ja) | 2008-02-29 | 2014-07-31 | Abbott Gmbh & Co Kg | Rgmaタンパク質に対するモノクローナル抗体及びその使用 |
| JP2016175897A (ja) | 2008-02-29 | 2016-10-06 | アッヴィ・ドイチュラント・ゲー・エム・ベー・ハー・ウント・コー・カー・ゲー | Rgm aタンパク質に対するモノクローナル抗体及びその使用 |
| JP2010100578A (ja) | 2008-10-24 | 2010-05-06 | St Marianna Univ School Of Medicine | Htlv−i関連脊髄症を治療または予防するための医薬、およびhtlv−i関連脊髄症の患者に対する抗体療法の効果を予測する方法 |
| WO2011071059A1 (ja) | 2009-12-09 | 2011-06-16 | 国立大学法人大阪大学 | T細胞活性化阻害剤、これを含有する医薬組成物およびt細胞活性化阻害物質のスクリーニング方法 |
| JP2013119531A (ja) * | 2011-12-07 | 2013-06-17 | St Marianna Univ School Of Medicine | Htlv−1関連脊髄症を治療または予防するための医薬および前記医薬を用いた抗体療法の治療効果の確認方法 |
| WO2014007303A1 (ja) * | 2012-07-06 | 2014-01-09 | 学校法人 聖マリアンナ医科大学 | Htlv-1関連脊髄症患者の治療方法および治療剤 |
| WO2016175236A1 (ja) | 2015-04-28 | 2016-11-03 | 田辺三菱製薬株式会社 | RGMa結合タンパク質及びその使用 |
| WO2017210278A1 (en) * | 2016-06-01 | 2017-12-07 | Abbvie Inc. | Anti-repulsive guidance molecule a (rgma) antagonistic antibodies for treating spinal cord injury and pain |
Non-Patent Citations (3)
| Title |
|---|
| KABAT ET AL.: "Sequences of Proteins of Immunological Interest", 1987, US DEPARTMENT OF HEALTH AND HUMAN SERVICES, NIH |
| N ENGL J MED, vol. 378, 2018, pages 529 - 538 |
| YAMASHITA, T.MUELLER, B.K.HATA, K.: "Neogenin and repulsive guidance molecule signaling in the central nervous system", CURR. OPIN. NEUROBIOL., vol. 17, 2007, pages 29 - 34, XP005931698, DOI: 10.1016/j.conb.2006.12.001 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN112566664A (zh) | 2021-03-26 |
| KR20210034055A (ko) | 2021-03-29 |
| TWI825131B (zh) | 2023-12-11 |
| JP7150286B2 (ja) | 2022-10-11 |
| JP7475011B2 (ja) | 2024-04-26 |
| CA3112511A1 (en) | 2020-01-23 |
| EP3824907A1 (en) | 2021-05-26 |
| MX2021000732A (es) | 2021-04-28 |
| PH12021550134A1 (en) | 2021-09-27 |
| US20210269535A1 (en) | 2021-09-02 |
| SG11202100561QA (en) | 2021-02-25 |
| CN116942831A (zh) | 2023-10-27 |
| PE20211376A1 (es) | 2021-07-27 |
| BR112021000949A2 (pt) | 2021-04-27 |
| TW202019482A (zh) | 2020-06-01 |
| CN112566664B (zh) | 2023-08-25 |
| JP2022180487A (ja) | 2022-12-06 |
| IL280257A (en) | 2021-03-25 |
| ZA202101113B (en) | 2022-07-27 |
| AU2019306721A1 (en) | 2021-03-11 |
| CN116785439A (zh) | 2023-09-22 |
| CN116850291A (zh) | 2023-10-10 |
| EP3824907A4 (en) | 2022-05-04 |
| JPWO2020017629A1 (ja) | 2021-08-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5785873B2 (ja) | T細胞活性化阻害剤、これを含有する医薬組成物およびt細胞活性化阻害物質のスクリーニング方法 | |
| JP6875683B2 (ja) | 多発性硬化症(ms)患者の新規治療適用判断方法 | |
| JP5552630B2 (ja) | Htlv−i関連脊髄症を治療または予防するための医薬、およびhtlv−i関連脊髄症の患者に対する抗体療法の効果を試験する方法 | |
| CA2824313A1 (en) | Tlr3 binding agents | |
| JP7475011B2 (ja) | Htlv-1関連脊髄症(ham)治療又は予防剤、及びhamの治療方法 | |
| US10428144B2 (en) | DICAM-specific antibodies and uses thereof | |
| TW202408577A (zh) | Htlv-1關聯性脊髓病(ham)之治療或防預劑、及ham之治療方法 | |
| WO2021085295A1 (ja) | 免疫応答抑制剤 | |
| WO2023171714A1 (ja) | 医薬組成物及び樹状細胞 | |
| WO2022191306A1 (ja) | 重症筋無力症の治療または予防用の医薬組成物 | |
| JP2022553643A (ja) | 細胞傷害性t細胞耐性腫瘍を治療するための組成物および方法 | |
| CN116997570A (zh) | 用于治疗抗体介导的移植排斥的抗cd38抗体 | |
| PRAT et al. | Patent 2971364 Summary | |
| WO2017184734A1 (en) | Functional plasticity of ilc2, immunity, and copd |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19838340 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2020531380 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 3112511 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112021000949 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 20217005032 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2019838340 Country of ref document: EP Effective date: 20210219 |
|
| ENP | Entry into the national phase |
Ref document number: 2019306721 Country of ref document: AU Date of ref document: 20190719 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 112021000949 Country of ref document: BR Kind code of ref document: A2 Effective date: 20210118 |