WO2019197631A1 - Feststoff-formulierung insektizider mischungen - Google Patents
Feststoff-formulierung insektizider mischungen Download PDFInfo
- Publication number
- WO2019197631A1 WO2019197631A1 PCT/EP2019/059471 EP2019059471W WO2019197631A1 WO 2019197631 A1 WO2019197631 A1 WO 2019197631A1 EP 2019059471 W EP2019059471 W EP 2019059471W WO 2019197631 A1 WO2019197631 A1 WO 2019197631A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- acid
- group
- composition according
- weight
- spp
- Prior art date
Links
- 0 BC(*)(C(O)=C1c2c(*)cc(*)cc2*)NC1OC Chemical compound BC(*)(C(O)=C1c2c(*)cc(*)cc2*)NC1OC 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N25/00—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests
- A01N25/08—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests containing solids as carriers or diluents
- A01N25/10—Macromolecular compounds
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N25/00—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests
- A01N25/12—Powders or granules
- A01N25/14—Powders or granules wettable
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N25/00—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests
- A01N25/30—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests characterised by the surfactants
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/04—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom
- A01N43/06—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings
- A01N43/12—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings condensed with a carbocyclic ring
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/24—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with two or more hetero atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/24—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with two or more hetero atoms
- A01N43/32—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with two or more hetero atoms six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/36—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom five-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/36—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom five-membered rings
- A01N43/38—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom five-membered rings condensed with carbocyclic rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N57/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic phosphorus compounds
- A01N57/10—Biocides, pest repellants or attractants, or plant growth regulators containing organic phosphorus compounds having phosphorus-to-oxygen bonds or phosphorus-to-sulfur bonds
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N59/00—Biocides, pest repellants or attractants, or plant growth regulators containing elements or inorganic compounds
- A01N59/26—Phosphorus; Compounds thereof
Definitions
- the invention relates to solid formulations (in particular water-dispersible granules) of tetramic acid derivatives and mixtures of these tetramic acid derivatives, a process for their preparation and their use for the application of the active ingredients contained.
- the preparation of WG formulations with the formulation auxiliaries mentioned in the prior art leads to formulations which have undesired properties.
- the low melting point of the drug or drug mixture leads to the formation of large amounts of Nasssiebguriquen that lead to clogging of the spray nozzles in the subsequent application of the spray mixture.
- the dispersed active ingredient or the dispersed active ingredients is often available only in low concentration in solution and / or the uptake or penetration of the active ingredient of the active ingredients through the Kutikel is insufficient.
- the present formulations have particularly advantageous properties.
- wet sieve residues in their production, only very small amounts of wet sieve residues, they can be well extruded, have particularly high suspension stability, have a good suspensibility and high bioavailability of the drug, or a good penetration through the Kutikel.
- formulations of the present invention are further distinguished by the fact that they have a good suspensibility even under high salt load, especially after storage.
- the present invention relates to a process for the preparation of these water-dispersible granules and to their use for application.
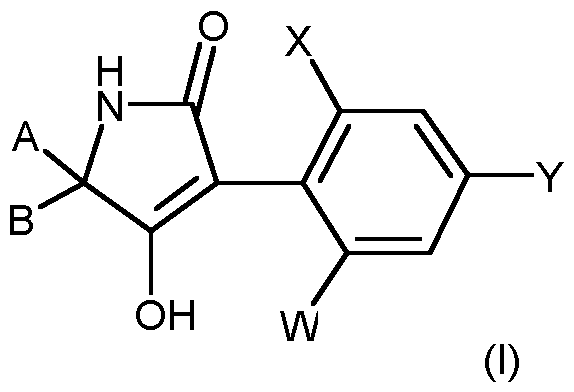
- the invention therefore provides insecticidal solid compositions in the form of water-dispersible granules (solid formulations) preferably comprising: a. as component a tetramic acid derivatives of the formula (I)
- W and Y independently of one another represent hydrogen, C 1 -C 4 -alkyl, chlorine, bromine, iodine or fluorine,
- X is Cl-C4-alkyl, Cl-C4-alkoxy, chlorine, bromine or iodine,
- A, B and the carbon atom to which they are attached represent C 3 -C 6 -cycloalkyl which is represented by an optionally substituted by C 1 -C 4 -alkyl or C 1 -C 4 -alkoxy-C 1 -C 2 -alkyl-substituted alkylenedioxy group which forms a 5-ring or 6-ring ketal with the carbon atom to which it is attached,
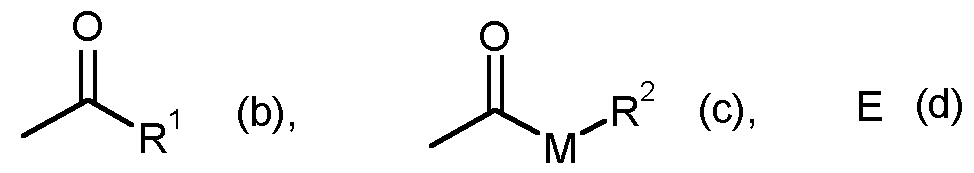
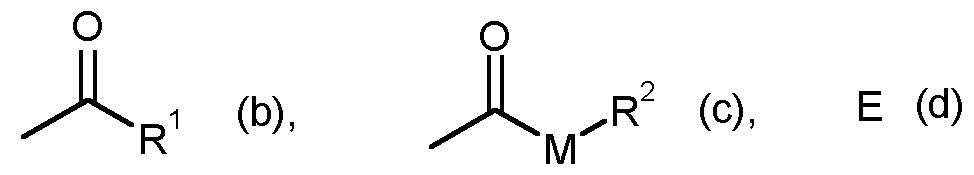
- G is hydrogen (a) or one of the groups stand in which
- E is a metal ion or an ammonium ion
- M is oxygen or sulfur
- R1 is straight-chain or branched C1-C6-alkyl
- R2 represents straight-chain or branched C1-C6-alkyl.
- unsubstituted or substituted radicals may, unless stated otherwise, be monosubstituted or polysubstituted, where in the case of multiple substitutions the substituents may be identical or different.
- the various preferred levels are to be understood that they can be permutierend combined with each other, but in each case the same preference levels and in particular the most preferred embodiment / preferred level to combine with each other and as such combination also disclosed.
- compositions as described above which consist only of the essential components (non-optional components), are to be considered as disclosed.
- Percentages are by weight unless otherwise stated, with the weight percent of the compositions adding up to 100%.
- basic in the sense of the present invention means a pH in aqueous solution with pH> 7.
- the formulations according to the invention have a pH of> 7 in aqueous dispersion.
- the formulations contain at least one defoamer (i), preferably solid, preferably silicone oil or wax base, more preferably a solid support absorbed PDMS.
- the preferred antifoaming agent is: Rhodorsil Antimousse EP6703 (absorbed polydimethylsiloxane) from Solvay a) Active substance
- component a) is in the erfmdungshielen
- W is methyl
- X is chlorine or methyl
- Y is chlorine, bromine or methyl
- A, B and the carbon atom to which they are attached represent saturated C 6 -cycloalkyl which is substituted with an alkylenedioxy group which forms a 5-ring or 6-ring ketal with the carbon atom to which it is attached .
- G is hydrogen (a) or one of the groups
- E is a metal ion equivalent or an ammonium ion
- R.2 for straight-chain or branched C
- component a) in the compositions according to the invention is a compound of the formula (I)
- component a) is a compound of the formula
- Component b) is preferably selected from the group comprising inorganic basic ammonium salts. Further preferred is b) selected from the group comprising ammonium carbonate, ammonium bicarbonate and diammonium hydrogen phosphate (DAHP).
- DAHP diammonium hydrogen phosphate
- Suitable dispersants in the present invention are dispersants of the polycarboxylate type, for example those such as hydrophobically modified comb-like polymers such as polyacrylic acid, polymethacrylic acid, polymaleic acid, polymaleic anhydride, a copolymer of maleic acid or maleic anhydride with an olefin (such as isobutylene or diisobutylene), a copolymer of acrylic acid and itaconic acid, a copolymer of methacrylic acid and itaconic acid, a copolymer of maleic acid or maleic anhydride and styrene, a copolymer of acrylic acid and methacrylic acid, a copolymer of acrylic acid and methacrylate, a copolymer of acrylic acid and vinyl acetate, a copolymer of styrene and methacrylic acid , modified copolymers of styrene and methacrylic acid, a copolymer
- dispersants selected from the group consisting of sodium salts of the copolymers of maleic acid and olefins (e.g., Geropon T / 36 / Solvay; Duramax D-305 / Dow); and sodium salts of copolymers of methacrylic acid and styrene (Tersperse 2700 / Huntsman, Atlox Metasperse 500S / Croda).
- the dispersant is a sodium salt of copolymers of methacrylic acid and styrene.
- Suitable nonionic surfactants are all substances of this type conventionally usable in agrochemical compositions.
- polyethylene oxide-polypropylene oxide block copolymers polyethylene glycol ethers of linear alcohols, reaction products of fatty acids with ethylene oxide and / or propylene oxide, furthermore polyvinyl alcohol, polyvinylpyrrolidone, copolymers of polyvinyl alcohol and polyvinylpyrrolidone and copolymers of (Meth) acrylic acid and (meth) acrylic acid esters, furthermore alkyl ethoxylates and alkylaryl ethoxylates, which may optionally be phosphated and optionally neutralized with bases, wherein sorbitol ethoxylates may be mentioned by way of example, and polyoxyalkyleneamine derivatives.
- anionic surfactants are all commonly used in agrochemical agents sub stances of this type in question.
- wetting agents are anionic surfactants selected from the group consisting of salts of polystyrenesulfonic acids, salts of polyvinylsulfonic acids, salts of naphthalenesulfonic acid-formaldehyde condensation products, salts of condensation products of naphthalenesulfonic acid, phenolsulfonic acid and formaldehyde and salts of ligninsulfonic acid and sodium salts of alkylated naphthalenesulfonates, e.g. ®Morwet EFW, and the sodium salts of dioctylsulfosuccinic acid, e.g. ⁇ Aerosol OTB, and block polymer of propylene oxide and ethylene oxide on ethylenediamine, e.g. ⁇ Synperonic T 905, included.
- anionic surfactants selected from the group consisting of salts of polystyrenesulfonic acids, salts of polyvinylsulfonic acids,
- suitable wetting agents (d) for the purposes of the present invention are wetting agents selected from the group consisting of the sodium salts of alkylated naphthalenesulfonates, such as, for example, ⁇ Morwet EFW, and the sodium salts of dioctylsulfosuccinic acid, e.g. Aerosol OTB, and most preferably consisting of the sodium salts of alkylated naphthalenesulfonates, preferably ®Morwet EFW.
- filler selected from the group consisting of the sodium salts of alkylated naphthalenesulfonates, such as, for example, ⁇ Morwet EFW, and the sodium salts of dioctylsulfosuccinic acid, e.g. Aerosol OTB, and most preferably consisting of the sodium salts of alkylated naphthalenesulfonates, preferably ®Morwet EFW.
- filler selected from
- Suitable inert fillers are all substances customarily usable for this purpose in agrochemical compositions. Preference is given to inorganic particles such as carbonates, silicates and oxides, as well as organic substances such as urea-formaldehyde condensates and celluloses (for example Ulmer Stamms 6AL, Celite 209, Argirec B21 and B22, Bentone EW, talc Luz2, Etiquette Violette). Examples include kaolin, rutile, silicon dioxide, so-called highly disperse silica, silica gels, and natural and synthetic silicates, as well as talc.
- the filler is selected from the group comprising kaolin, rutile and silica. Particularly preferred is kaolin. f) structuring agent
- the formulations of the invention may further comprise a structuring agent.
- a structuring agent especially polyacrylic acid and its salts and in particular cross-linked polyacrylates and polyurethanes and derivatized polyurethanes and their salts are suitable.
- suitable Acrylates are: (poly) methacrylate, (poly) methylmethacrylate, polyacrylamide,
- Pergopak® M is a polymethyl urea resin from Albemarle Corporation, Baton Rouge, LA, USA.
- the structuring agent is particularly preferably a polymethylurea resin, in particular Pergopak®M g) complexing agent
- complexing agent is selected from the group comprising ethylenediaminetetraacetic acid, gluconic acid, nitrilotriacetic acid, diethylenediaminepentaacetic acid, and salts and / or hydrates of the abovementioned or combinations thereof.
- Na4EDTA tetrasodium ethylenediaminetetraacetate
- Trilon® B Powder from BASF
- the formulation may in principle contain one or more other agrochemical active substances, preferably selected from the group of insecticides, herbicides, fungicides and host defense inducers.
- one or more active ingredients selected from the group of insecticides, still more preferably from the group comprising imidacloprid, nitenpyram, acetamiprid, thiacloprid, thiamethoxam, clothianidin, cyantraniliprole, chlorantraniliprole, flubendiamide, tetraniliprole, cyclaniliprole; Spirodiclofen, spiromesifen, spirotetramat, abamectin, acrinathrin, chlorfenapyr, emamectin, ethiprole, fipronil, flonicamid, flupyradifuron, indoxacarb, metaflumizone, methoxyfenozide, milbemycin, pyridaben, pyridalyl, silafluofen, spinosad, sulfoxaflor, triflumuron;
- One or more other active ingredients are particularly preferably selected from the group comprising ethiproles and flupyradifurones. i) adjuvants and formulation auxiliaries.
- Suitable foam-inhibiting substances are all substances customarily usable for this purpose in agrochemical compositions. Preference is given to defoamers based on silicone oil and magnesium stearate, Particularly preferred is polydimethylsiloxane absorbed on a solid substrate. An example is Rhodorsil Antimousse EP 6703.
- Suitable preservatives are all substances customarily usable for this purpose in agrochemical compositions of this type. Examples which may be mentioned are Preventol® (Fa. Fanxess AG) and Proxel®.
- antioxidants are all commonly used for this purpose in agrochemical agents substances into consideration. Preference is butylhydroxytoluene.
- Suitable dyes are all substances customarily usable for this purpose in agrochemical compositions. Examples include titanium dioxide, carbon black, zinc oxide and blue pigments as well as permanent red FGR.
- the subject of the invention is an insecticidal solid composition in the form of water-dispersible granules comprising: a. Compounds of the formula (I)
- X is chlorine or methyl
- Y is chlorine, bromine or methyl
- A, B and the carbon atom to which they are attached represent saturated C 6 -cycloalkyl which is substituted with an alkylenedioxy group which forms a 5-ring or 6-ring ketal with the carbon atom to which it is attached .
- G is hydrogen (a) or one of the groups E (d),
- E is a metal ion equivalent or an ammonium ion
- R.1 is straight-chain or branched C
- At least one inorganic basic ammonium salt b. at least one inorganic basic ammonium salt, c. at least one dispersant of the polycarboxylate type d. at least one anionic wetting agent, e. at least one inert filler, f. at least one structuring agent g. at least one complexing agent h. optionally further insecticidal active ingredients, i. optionally further adjuvants.
- the object of the invention is an insecticidal solid compositions in the form of water-dispersible granules comprising: a.
- Maleic anhydride and styrene and modified copolymers of maleic acid or maleic anhydride and styrene a copolymer of acrylic acid and methacrylic acid, a copolymer of styrene and methacrylic acid, modified copolymers of styrene and methacrylic acid, a copolymer of acrylic acid and methacrylate, a copolymer of acrylic acid and vinyl acetate, a copolymer of Maleic acid or maleic anhydride and
- Acrylic acid an N-methyl fatty acid (eg Cs cisj sarcosinate, a carboxylic acid such as a rosin acid or a fatty acid (eg Cs-Cis) or a salt of such a carboxylic acid, as well as the salts of these copolymers, i.e. at least one anionic surfactant selected from the group consisting of the sodium salts of alkylated naphthalenesulfonates and the sodium salts of dioctylsulfosuccinic acid, e.
- anionic surfactant selected from the group consisting of the sodium salts of alkylated naphthalenesulfonates and the sodium salts of dioctylsulfosuccinic acid, e.
- At least one inert filler selected from the group comprising carbonates, silicates and oxides, and urea-formaldehyde condensates and celluloses
- f Structuring agents selected from the group comprising polyacrylic acid and its salts, cross-linked polyacrylates and polyureas, polyurethanes and derivatized polyureas and polyurethanes, g.
- At least one complexing agent selected from the group consisting of
- Ethylenediaminetetraacetic acid EDTA
- gluconic acid gluconic acid
- nitrilotriacetic acid diethylenediaminepentaacetic acid
- salts and / or hydrates of these compounds h. optionally further insecticidal active ingredients, i. optionally further adjuvants.
- the subject of the invention is an insecticidal solid compositions in the form of water-dispersible granules containing: a. Compound of the formula (1-2) having the following structure:
- DAHP diammonium hydrogens
- another insecticidal active ingredient (g) is mandatory in the abovementioned embodiments, preferably from the group comprising imidacloprid, nitenpyram, acetamiprid, thiacloprid, thiamethoxam, clothianidin, cyantraniliproles, chlorantraniliproles, flubendiamide, tetraniliproles, cyclaniliproles; Spirodiclofen, spiromesifen, spirotetramat, abamectin, acrinathrin, chlorfenapyr, emamectin, ethiprole, fipronil, flonicamid, flupyradifuron, indoxacarb, metaflumizone, methoxyfenozide, milbemycin, pyridaben, pyridalyl, silafluofen, spinosad, sulfoxaflor, triflumuron; particularly
- the proportion of the active ingredient (component a / compounds of formula I / 1-2) in the compositions according to the invention with only one active ingredient is preferably 0, 1-25 wt .-%, more preferably 2- 10 wt .-%, and particularly preferably 4-8% by weight.
- Active ingredient is preferably 20-75% by weight, more preferably 50-68% by weight, and particularly preferably 60-66% by weight.
- the proportion of the dispersant (component c) in the compositions according to the invention with only one active ingredient is preferably 1-20% by weight, more preferably 5-15% by weight, and particularly preferably 8-12% by weight.
- the proportion of wetting agent (component d) in the compositions according to the invention with only one active ingredient is preferably 0.5-15% by weight, more preferably 1-10% by weight, and particularly preferably 1.5-2.5% by weight. -%.
- the proportion of the filler (component e) in the compositions according to the invention with only one active ingredient is preferably 0.5-70% by weight, more preferably 5-25% by weight, and particularly preferably 10-22% by weight.
- the proportion of the structuring agent (component f) in the compositions according to the invention having only one active ingredient is preferably 0.5 to 15% by weight, more preferably 1 to 10% by weight, and particularly preferably 3 to 8% by weight.
- the proportion of the complexing agent (component g) in the compositions according to the invention with only one active ingredient is preferably 0-10% by weight, more preferably 0-1-2% by weight, and particularly preferably 0.1-1% by weight. be.
- the proportion of further adjuvants (component i), if present, in the compositions according to the invention is preferably 0-10% by weight, more preferably 0-8% by weight, and particularly preferably 0-5% by weight.
- component i) according to the alternative embodiment is mandatory, e.g. when
- compositions comprising components a) 2-10% by weight
- compositions comprising components a) 4-8% by weight
- a solid composition is preferably containing components in a) 4-8% by weight.
- the proportion of the active ingredient (component a / compounds of the formula I / 1-2) in the inventive compositions is preferably 1-10% by weight, more preferably 1-7% by weight, and particularly preferably 1.5-5% by weight .-%.
- the proportion of the salt (component b) in the compositions according to the invention is preferably 20-75% by weight, more preferably 30-60% by weight, and particularly preferably 30-50% by weight.
- the proportion of the dispersant (component c) in the compositions according to the invention is preferably 1-20% by weight, more preferably 5-15% by weight, and particularly preferably 8-12% by weight.
- the proportion of wetting agent (component d) in the inventive compositions is preferably 0.5 to 15 wt .-%, more preferably 1 to 10 wt .-%, and particularly preferably 1.5-2.5% by weight.
- the proportion of the filler (component e) in the inventive compositions is preferably 0.5 to 70 wt .-%, more preferably 5 to 25 wt .-%, and particularly preferably 10 to 22 wt .-% amount.
- the proportion of the structuring agent (component f) in the compositions according to the invention is preferably 0.5 to 15% by weight, more preferably 1 to 10% by weight, and particularly preferably 3 to 8% by weight.
- the proportion of the complexing agent (component g) in the compositions according to the invention is preferably 0 to 10% by weight, more preferably 0 to 1 to 2% by weight, and particularly preferably 0.1 to 1% by weight.
- the proportion of further active ingredients (component h), if present, in the compositions according to the invention is preferably 1-60% by weight, more preferably 10-40% by weight, and particularly preferably 10-35% by weight.
- the proportion of further adjuvants (component i), if present, in the compositions according to the invention is preferably 0 to 10% by weight, more preferably 0 to 8% by weight, and particularly preferably 0 to 5 wt .-% amount.
- component i) according to the alternative embodiment is mandatory, e.g. when
- the compound 1-2 is preferably used in the form of its thermodynamically most stable polymorphic structure. This crystal structure and other physical data were determined as follows:
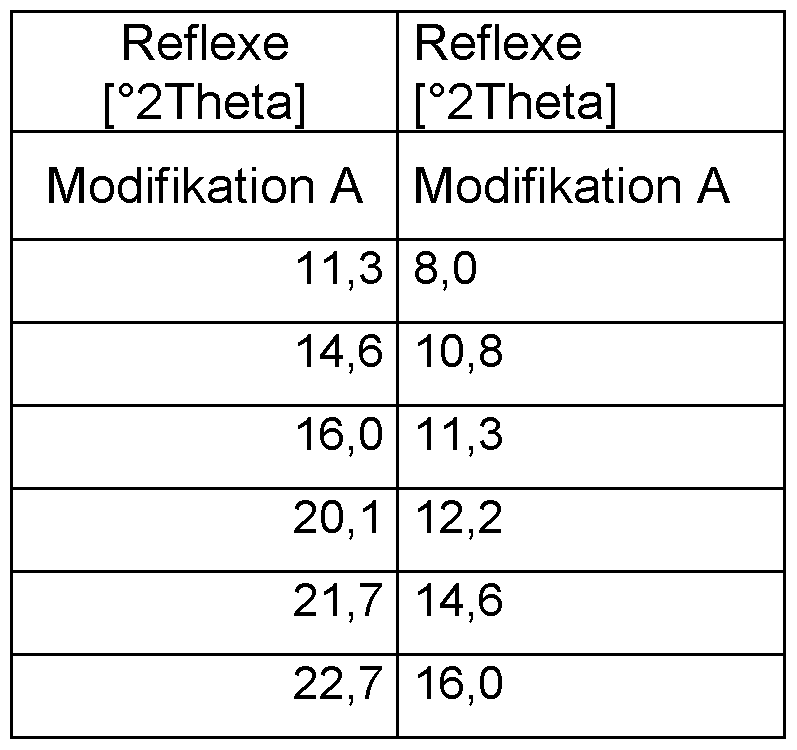
- Modification A of 1-2 can be characterized by X-ray powder diffractometry based on the corresponding diffraction patterns taken at 25 ° C and with Cu-Ka 1 radiation (1.5406 ⁇ ) (FIGURE 1).
- Modification A exhibits at least 3, preferably at least 5, more preferably at least 7, even more preferably at least 10, and most preferably all reflections as shown in FIGURE 1 and Table 2a) Reflections, modification A:
- Modification A according to the present invention is further characterized by the X-ray diffraction pattern illustrated in FIG.
- the polymorphic form of modification A of 1-1 can be determined by IR spectroscopy on the appropriate spectrum recorded at 25 ° C using a diamond ATR instrument at a resolution of 4 cm -1 (FIGURE 2).
- Modification A of the present invention exhibits at least 3, preferably at least 5, more preferably at least 7, and most preferably all bands as seen in Figure 2 and described in Table 2c.
- Compound 1-2 can be present in other polymorphic forms as well as pseudopolymorphic forms (hydrates, solvates, etc.).
- the application rate of the formulations according to the invention can be varied within a relatively wide range. It depends on the particular active ingredients and their content in the compositions.
- the insecticidal active substance mixtures can be applied in a particularly advantageous manner to plants and / or their habitat.
- plants and plant parts can be treated with the compositions according to the invention.
- plants are understood as meaning all plants and plant populations, such as desired and undesired wild plants or crop plants (including naturally occurring crop plants).
- Crop plants can be plants which can be obtained by conventional breeding and optimization methods or by biotechnological and genetic engineering methods or combinations of these methods, including the transgenic plants and including the plant varieties which can or can not be protected by plant breeders' rights.
- Plant parts are to be understood as meaning all aboveground and subterranean parts and organs of the plants, such as shoot, leaf, flower and root, examples of which include leaves, needles, stems, stems, flowers, fruiting bodies, fruits and seeds, and roots, tubers and rhizomes.
- the plant parts also include emmentals and vegetative and generative propagation material, such as cuttings, tubers, rhizomes, offshoots and seeds.
- the compounds of the formula (I) are preferably employed in the formulations according to the invention by spray application, but also after casting (drench), dip, or by drip application against animal pests from the following pest families:
- bladder lice Pemphigidae: Eriosoma spp., Pemphigus spp., In cultures such as e.g. Citrus, pome fruit, stone fruit, leafy vegetables, root and tuber vegetables and ornamental plants.
- Diaspididae quadraspidiotus spp., Aonidiella spp., Lepidosaphes spp., Aspidiotus spp., Aspis spp., Diaspis spp., Parlatoria spp., Pseudaulacaspis spp., Unaspis spp., Pinnaspis spp. , Selenaspidus spp., In cultures such as Citrus, pome fruit, stone fruit, almonds, pistachios, nuts, olives, tea, ornamentals, wine, tropical crops.
- mothback lice family (Aleyrodidae): Bemisia tabaci, Bemisia argentifolii, Trialeurodes vaporariorum, Aleurothrixus floccosus, Aleurodes spp., Dialeurodes spp., Parabemisia myricae in crops such as e.g. Vegetables, melons, potatoes, tobacco, berry fruits, citrus, ornamental plants, cotton, soybeans and tropical crops.
- crops such as e.g. Vegetables, melons, potatoes, tobacco, berry fruits, citrus, ornamental plants, cotton, soybeans and tropical crops.
- Rhodobium porosum in strawberries Nasonovia ribisnigri in leafy vegetables
- Phorodon humuli in hops Brevicoryne brassicae in leafy vegetables
- Toxoptera spp. In citrus, stone fruits, almonds, nuts, spices,
- thrips Thripidae: Anaphothrips spp., Basothrips spp., Caliothrips spp., Frankliniella spp., Heliothrips spp., Hercinothrips spp., Rhipiphorothrips spp., Scirtothrips spp., Kakothrips spp., Selenothrips spp , and Thrips spp., in cultures such as e.g. Fruit, cotton, wine, tea, nuts, tropical crops, ornamental plants, conifers, tobacco, spices, vegetables, berries, melons, citrus and potatoes.
- cultures such as e.g. Fruit, cotton, wine, tea, nuts, tropical crops, ornamental plants, conifers, tobacco, spices, vegetables, berries, melons, citrus and potatoes.
- Agromyza spp. Agromyza spp., Amauromyza spp., Atherigona spp., Chlorops spp., Liriomyza spp., Oscinella spp., Pegomyia spp. in cultures such as e.g. Vegetables, melons, potatoes, nuts, ornamental plants.
- Caloptilia spp. Gracillaria spp., Lithocolletis spp., Leucoptera spp., Phtorimaea spp., Phyllocnistis spp. in crops such as pome fruit, stone fruit, wine, nuts, citrus, conifers, potatoes, coffee.
- Anastrepha spp. Ceratitis spp.
- Dacus spp. Rhagoletis spp. in crops such as vegetables, berries, melons, stone fruit and stone fruits, ornamental plants, potatoes, wine, tropical crops, citrus, olives.
- Tetranychus spp. Tetranychus spp., Panonychus spp., Aculops spp. in crops such as vegetables, potatoes, ornamental plants, citrus, wine, conifers.
- the treatment according to the invention of the plants and plant parts with the compositions according to the invention is carried out directly or by acting on their environment, Febensraum or Fagerraum according to the usual treatment methods, e.g. by drenching, dipping, spraying, vaporizing, atomizing, spreading, brushing and, in the case of propagation material, in particular in seeds, further by single or multi-layer coating.
- the application of the active ingredient is preferably carried out by Spriztap rates.
- the application of the active ingredient is carried out by casting (drench), droplet application (Drip) or by dip application.
- the plant to be treated is selected from the group consisting of cotton, soybean, tobacco, vegetables, spices, ornamentals, conifers, citrus plants, fruits, tropical crops, nuts and wine.
- composition of the invention against pests of the families of the bladder lice, root lice, leaf fleas, Napfschildläuse, Lupus, Louse, Licecomb, mealybugs, moth sign lice, louse, thrips, dwarf cicadas, Homzikaden, miner flies, gall bladder, fruit flies, leafminers, spider mites, gall mites.
- compositions of the invention can be prepared by moistening a powdery mixture consisting of the active ingredients) and the formulation auxiliaries, then granulated by means of low-pressure extrusion and the moist granules are then dried.
- Relevant equipment for humidification, extrusion and drying are known in the art.
- the temperatures to which the product is exposed are kept below 60 ° C at all process stages, and preferably lower than 50 ° C. This method is also the subject of the invention.
- the treatment according to the invention of the plants and plant parts with the compositions according to the invention is carried out directly or by acting on their environment, habitat or storage space according to the usual treatment methods, e.g. by dipping, spraying, vaporizing, atomizing, spreading, spreading and in propagation material, in particular in seeds, further by single or multi-layer wrapping.
- a 1% suspension in water (CIPAC C) is dispersed in a 250 ml graduated cylinder. The mixture is equilibrated for 30 minutes. Subsequently, the upper 9/10 of the suspension are removed. The remaining 1/10 are dried and the residue is determined gravimetrically.
- the suspensibility is defined as: 10/9 100 (sample quantity [g] * proportion ai [%] / 100 - residue [g]) / (sample quantity [g] * proportion ai [%] / 100)
- a value of 100% corresponds to a uniform distribution of all insoluble ones Components over the sample volume.
- a good visual rating means no sediment and no flocculation. l.b) Suspensibility and Stability Solo & Mixture Formulation
- a mixture of COMPOUND 1-2, a second active ingredient, Morwet EFW, Pergopak M, optionally Trilon B Powder, Tersperse 2700, DAHP and optionally kaolin W, as known to those skilled in the art, is dissolved in a 4 "air jet mill Hosokawa 100 AS ( Injector air 5.5 bar, grinding air 4.5 bar, throughput 100 g / min) ground, moistened with water (about 14 wt .-%) and on a Fuji Paudal dome extruder type (die size 0.8 or 1 mm) extruded to a WG and dried in a fluidized bed dryer.
- Example 22 shows that with an active ingredient combination the optimum content of filler must be adjusted. ) Solubility of the active substance
- sample 19 6 samples of sample 19 (see example 2b) were dispersed in 50 g of water so that concentrations of 0.12 and 0.24 g of active ingredient / L were achieved. The mixtures were stirred and after each 30 minutes, 2 h or 5 h completely centrifuged and determined by HPLC, the proportion of dissolved active ingredient in the supernatant.
- Sheets were used, which were cut in the fully developed state of apple trees of the variety Golden Delicious. The isolation of the cuticles took place in such a way that
- the cuticles were placed with tweezers in the middle of the coated with silicone grease edges of the diffusion cells and sealed with a likewise greased ring.
- the arrangement was chosen so that the morphological outside of the cuticles was directed outwards, ie towards the air, while the original inner side was facing the inside of the diffusion cell.
- the Difenüsionszellen were filled with a 30% ethylene glycol / water solution. To determine the penetration, in each case 10 ⁇ l of the spray mixture of the following composition was applied to the outside of the cuticle. The preparation of the spray mixture is carried out with local tap water medium water hardness.
- the water was allowed to evaporate, inverting the chambers and placed in thermostated tubs in which the temperature and humidity above the cuticle was adjustable by a slight air flow onto the cuticle with the spray coating (20 ° C, 60%). rh). At regular intervals aliquots were taken from an autosampler and the active ingredient content was determined by HPLC.
- test results are shown in the following table.
- the figures given are average values from 8 to 10 measurements.
- Sample 1 and a control sample (sample 25) with the same concentration of ammonium dihydrogen phosphate (ADHP) as DAHP were measured.
- Kraftsperse ® EDF-350, ® DD-5, Kersperse ® DD-8, Kersperse ® DW-8, Kersperse ® EDF-450, Reax ® 88B and Reax ® 907 are lignosulfonates from Ingevity, North Charleston, SC, USA.
- Geropon® TA / 72, T / 36 is a polycarboxylic acid sodium salt from Solvay.
- Oparyl® MT 804 is a di-butyl-naphthalenesulfonic acid sodium salt from Giovanni Bozzetto S.p.A., Filago, Italy.
- Oparyl® DT 530 is a naphthalenesulfonic acid-formaldehyde condensate, sodium salt from Giovanni Bozzetto S.p.A., Filago, Italy.
- Pergopak® M is a polymethylurea resin from Albemarle Corporation, Baton Rouge, FA, USA.
- Rhodorsil® EP 6703 is a polydimethylsiloxane on starch from Rhodia, Boulogne, France.
- Celite® 209 S is a diatomaceous earth from Fehmann & Voss, Hamburg, Germany.
- Talcum Fuzenac 2 is a magnesium hydrosilicate from Fuzenac Europe, Paris, France.
- Kaolin W is an aluminum hydrosilicate from Erbslöh Fohrheim GmbH & Co. KG, Fohrheim, Germany.
- Borresperse ® Na and Ufoxane ® 3 A are Figninsulfonate the company Borregaard, Sarpsborg, Norway.
- Morwet® D 425 is a naphthalenesulfonic acid-formaldehyde condensate, sodium salt from Akzo Nobel, Stenungsund, Sweden.
- Tersperse® is a naphthalenesulfonic acid-formaldehyde condensate, sodium salt from Huntsman, The Woodlands, Texas, USA.
- Baykanol® SU is an alkyl aryl sulfonate from Fanxess, Feverkusen, Germany.
- Rhodorsil Antimousse EP 6703® is an absorbed polydimethylsiloxane, Solvay, PAGE INTENTIONALLY LEFT BLANK
Landscapes
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Health & Medical Sciences (AREA)
- Dentistry (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Engineering & Computer Science (AREA)
- Agronomy & Crop Science (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Toxicology (AREA)
- Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Catching Or Destruction (AREA)
Abstract
Description
Claims
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2019250613A AU2019250613A1 (en) | 2018-04-13 | 2019-04-12 | Solid formulation of insecticidal mixtures |
| BR112020017813-6A BR112020017813A2 (pt) | 2018-04-13 | 2019-04-12 | Formulação sólida de misturas inseticidas |
| MX2020010834A MX2020010834A (es) | 2018-04-13 | 2019-04-12 | Formulacion solida de mezclas insecticidas. |
| EP19716900.6A EP3772955A1 (de) | 2018-04-13 | 2019-04-12 | Feststoff-formulierung insektizider mischungen |
| PE2020001564A PE20210399A1 (es) | 2018-04-13 | 2019-04-12 | Formulacion solida de mezclas insecticidas |
| KR1020207026906A KR20200142501A (ko) | 2018-04-13 | 2019-04-12 | 살곤충 혼합물의 고형 제제 |
| CN201980025594.2A CN111935979B (zh) | 2018-04-13 | 2019-04-12 | 杀虫剂混合物的固体制剂 |
| JP2020555855A JP2021521196A (ja) | 2018-04-13 | 2019-04-12 | 殺虫混合物の固体製剤 |
| US17/046,477 US20210161128A1 (en) | 2018-04-13 | 2019-04-12 | Solid formulation of insecticidal mixtures |
| IL277679A IL277679B1 (en) | 2018-04-13 | 2019-04-12 | Solid formulation of insecticidal mixtures |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP18167281 | 2018-04-13 | ||
| EP18167281.7 | 2018-04-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019197631A1 true WO2019197631A1 (de) | 2019-10-17 |
Family
ID=62001992
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2019/059471 WO2019197631A1 (de) | 2018-04-13 | 2019-04-12 | Feststoff-formulierung insektizider mischungen |
Country Status (16)
| Country | Link |
|---|---|
| US (1) | US20210161128A1 (de) |
| EP (1) | EP3772955A1 (de) |
| JP (1) | JP2021521196A (de) |
| KR (1) | KR20200142501A (de) |
| CN (1) | CN111935979B (de) |
| AR (1) | AR114487A1 (de) |
| AU (1) | AU2019250613A1 (de) |
| BR (1) | BR112020017813A2 (de) |
| CL (1) | CL2020002614A1 (de) |
| EC (1) | ECSP20063701A (de) |
| IL (1) | IL277679B1 (de) |
| MA (1) | MA52187A (de) |
| MX (1) | MX2020010834A (de) |
| PE (1) | PE20210399A1 (de) |
| TW (1) | TWI820115B (de) |
| WO (1) | WO2019197631A1 (de) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021062338A1 (en) * | 2019-09-27 | 2021-04-01 | Fmc Corporation | Anthranilamide composition |
| EP4265110A1 (de) | 2022-04-20 | 2023-10-25 | Bayer AG | Wasserdispergierbare granulate mit durch extrusion hergestellten niedrigschmelzenden wirkstoffen |
| WO2024008807A1 (de) | 2022-07-08 | 2024-01-11 | Bayer Aktiengesellschaft | Feststoff-formulierung insektizider mischungen mit besonders guten dispergiereigenschaften |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114636768B (zh) * | 2022-03-23 | 2023-11-07 | 广西-东盟食品检验检测中心 | 一种茉莉花中噻虫嗪残留的检测方法 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008036865A2 (en) | 2006-09-21 | 2008-03-27 | Medtronic, Inc. | Radiofrequency (rf)-shunted sleeve head and use in electrical stimulation leads |
| US20150259345A1 (en) * | 2008-12-12 | 2015-09-17 | Syngenta Limited | Spiroheterocyclic n-oxypiperidines as pesticides |
| US20160081342A1 (en) * | 2013-05-23 | 2016-03-24 | Syngenta Participations Ag | Tank-mix formulations |
| US20160106095A1 (en) * | 2013-05-23 | 2016-04-21 | Syngenta Participations Ag | Tank-mix formulations |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2100339A1 (en) * | 1991-01-24 | 1992-07-25 | Erhard John Prill | Glyphosate formulations |
| WO2003101197A1 (en) * | 2002-05-31 | 2003-12-11 | Cjb Industries, Inc. | Adjuvant for pesticides |
| US7537778B2 (en) * | 2002-09-26 | 2009-05-26 | W. Neudorff Gmbh Kg | Pesticidal compositions and methods |
| DE102005008021A1 (de) * | 2005-02-22 | 2006-08-24 | Bayer Cropscience Ag | Spiroketal-substituierte cyclische Ketoenole |
| JP5449914B2 (ja) * | 2008-08-07 | 2014-03-19 | 石原産業株式会社 | 水田用農薬組成物 |
| CN103492367B (zh) * | 2011-02-17 | 2015-04-01 | 拜耳知识产权有限责任公司 | 用于治疗的取代的3-(联苯-3-基)-8,8-二氟-4-羟基-1-氮杂螺[4.5]癸-3-烯-2-酮 |
| EP2925144A2 (de) * | 2012-12-03 | 2015-10-07 | Bayer CropScience AG | Zusammensetzung aus einem biologischen bekämpfungsmittel und einem insektizid |
| WO2014096130A1 (en) * | 2012-12-20 | 2014-06-26 | Bayer Cropscience Nv | Use of tetramic acid derivatives on plants to control flowering |
| CN104995193A (zh) * | 2012-12-20 | 2015-10-21 | 拜耳作物科学股份公司 | 作为杀螨剂和杀虫剂的芳基硫化物和芳基亚砜衍生物 |
| US9751862B2 (en) * | 2014-03-20 | 2017-09-05 | Syngenta Participations Ag | Pesticidally active substituted pyridyl carboxamides |
| ES2762787T3 (es) * | 2014-03-27 | 2020-05-25 | Bayer Cropscience Ag | Combinaciones de principios activos insecticidas y nematocidas |
| MY190137A (en) * | 2014-12-22 | 2022-03-30 | Nihon Nohyaku Co Ltd | Noxious organism control agent composition for agricultural and horticultural applications, and method for using said composition |
| EP3494109A1 (de) * | 2016-08-04 | 2019-06-12 | Bayer CropScience Aktiengesellschaft | Verfahren zur herstellung von spiroketal-substituierten cyclischen ketoenolen |
| JP2018039745A (ja) * | 2016-09-06 | 2018-03-15 | 日本農薬株式会社 | 農園芸用殺虫・殺ダニ剤組成物及びその使用方法 |
-
2019
- 2019-04-12 JP JP2020555855A patent/JP2021521196A/ja active Pending
- 2019-04-12 AR ARP190100975A patent/AR114487A1/es unknown
- 2019-04-12 KR KR1020207026906A patent/KR20200142501A/ko active Search and Examination
- 2019-04-12 PE PE2020001564A patent/PE20210399A1/es unknown
- 2019-04-12 US US17/046,477 patent/US20210161128A1/en active Pending
- 2019-04-12 MX MX2020010834A patent/MX2020010834A/es unknown
- 2019-04-12 IL IL277679A patent/IL277679B1/en unknown
- 2019-04-12 MA MA052187A patent/MA52187A/fr unknown
- 2019-04-12 AU AU2019250613A patent/AU2019250613A1/en active Pending
- 2019-04-12 BR BR112020017813-6A patent/BR112020017813A2/pt unknown
- 2019-04-12 CN CN201980025594.2A patent/CN111935979B/zh active Active
- 2019-04-12 TW TW108112907A patent/TWI820115B/zh active
- 2019-04-12 EP EP19716900.6A patent/EP3772955A1/de not_active Withdrawn
- 2019-04-12 WO PCT/EP2019/059471 patent/WO2019197631A1/de active Application Filing
-
2020
- 2020-10-07 EC ECSENADI202063701A patent/ECSP20063701A/es unknown
- 2020-10-09 CL CL2020002614A patent/CL2020002614A1/es unknown
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008036865A2 (en) | 2006-09-21 | 2008-03-27 | Medtronic, Inc. | Radiofrequency (rf)-shunted sleeve head and use in electrical stimulation leads |
| US20150259345A1 (en) * | 2008-12-12 | 2015-09-17 | Syngenta Limited | Spiroheterocyclic n-oxypiperidines as pesticides |
| US20160081342A1 (en) * | 2013-05-23 | 2016-03-24 | Syngenta Participations Ag | Tank-mix formulations |
| US20160106095A1 (en) * | 2013-05-23 | 2016-04-21 | Syngenta Participations Ag | Tank-mix formulations |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2021062338A1 (en) * | 2019-09-27 | 2021-04-01 | Fmc Corporation | Anthranilamide composition |
| CN114430657A (zh) * | 2019-09-27 | 2022-05-03 | Fmc公司 | 邻氨基苯甲酰胺组合物 |
| EP4265110A1 (de) | 2022-04-20 | 2023-10-25 | Bayer AG | Wasserdispergierbare granulate mit durch extrusion hergestellten niedrigschmelzenden wirkstoffen |
| WO2023203009A1 (en) | 2022-04-20 | 2023-10-26 | Bayer Aktiengesellschaft | Water dispersible granules with low melting active ingredients prepared by extrusion |
| WO2024008807A1 (de) | 2022-07-08 | 2024-01-11 | Bayer Aktiengesellschaft | Feststoff-formulierung insektizider mischungen mit besonders guten dispergiereigenschaften |
Also Published As
| Publication number | Publication date |
|---|---|
| PE20210399A1 (es) | 2021-03-02 |
| BR112020017813A2 (pt) | 2020-12-29 |
| ECSP20063701A (es) | 2021-01-29 |
| AR114487A1 (es) | 2020-09-09 |
| JP2021521196A (ja) | 2021-08-26 |
| IL277679B1 (en) | 2024-02-01 |
| CN111935979A (zh) | 2020-11-13 |
| EP3772955A1 (de) | 2021-02-17 |
| MA52187A (fr) | 2021-02-17 |
| US20210161128A1 (en) | 2021-06-03 |
| IL277679A (en) | 2020-11-30 |
| TW202002788A (zh) | 2020-01-16 |
| CN111935979B (zh) | 2022-12-09 |
| KR20200142501A (ko) | 2020-12-22 |
| AU2019250613A1 (en) | 2020-10-22 |
| CL2020002614A1 (es) | 2021-01-29 |
| TWI820115B (zh) | 2023-11-01 |
| MX2020010834A (es) | 2020-11-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2019197631A1 (de) | Feststoff-formulierung insektizider mischungen | |
| EP2001296B1 (de) | Herbizide mittel als dispersionen enthaltend diflufenican und flurtamone | |
| BR112020022770A2 (pt) | composição de suspensão, composição granular, processos de preparação da composição e método para melhorar a saúde ou o rendimento das plantas | |
| JP2007529435A (ja) | 種子処理のための殺虫組成物及び殺虫方法 | |
| EP1926370B1 (de) | Verfahren zum schutz vor phytopathogenen mit kiralaxyl, entsprechende verwendung und mittel hierzu | |
| WO2019197635A1 (de) | Formulierung insektizider mischungen mit propylencarbonat | |
| EP0818144B1 (de) | Mikrobizide | |
| EP1642498A2 (de) | Wässrige Dispersionen enthaltend Diflufenican | |
| EP3402332B1 (de) | Wässrige dispersionen enthaltend aclonifen und flufenacet | |
| WO2019197634A1 (de) | Feststoff-formulierung insektizider mischungen | |
| CN105557993B (zh) | 咯菌腈及其组合物在防治甘薯储藏期病害上的应用 | |
| CN115299449B (zh) | 一种农药组合物及其应用 | |
| JP2017529351A (ja) | 軟体動物忌避のためのヘテロ環式フルオロアルケニルスルホン化合物の使用 | |
| DE60222438T2 (de) | Zusammensetzung zur bekämpfung von pflanzenpathogenen bakterien und verfahren zur bekämpfung von pflanzenpathogenen bakterien | |
| EP4275492A1 (de) | Formulierung von insektiziden mit propylencarbonat | |
| WO2024008807A1 (de) | Feststoff-formulierung insektizider mischungen mit besonders guten dispergiereigenschaften | |
| WO2019197637A1 (de) | Hochbeladene formulierungen mit insektiziden aus der ketoenol-klasse zur verwendung bei drip- und drench- applikationen | |
| DE60209040T2 (de) | Pyridin-3-sulfonylverbindungen als pestizide mittel | |
| CN115067348A (zh) | 一种复配组合物及其在农业中的应用 | |
| WO2019197652A1 (de) | Feststoff-formulierung insektizider mischungen | |
| EP2493884A1 (de) | Co-kristall von 4- { [ (6-chlorpyrid-3-yl) methyl](2,2-difluorethyl) amino} furan-2 (5h) -on mit salizylsäure und dessen verwendung als pestizid | |
| WO2019197636A1 (de) | Feststoff-formulierung insektizider mischungen | |
| CN111357748A (zh) | 含有Pyriofenone和氟苯醚酰胺或氯苯醚酰胺的杀菌组合物 | |
| CN115039784B (zh) | 一种组合物及其在农业中的应用 | |
| EP4275493A1 (de) | Formulierung insektizider mischungen mit glykoletherlösungsmitteln |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 19716900 Country of ref document: EP Kind code of ref document: A1 |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112020017813 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 2020555855 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2019250613 Country of ref document: AU Date of ref document: 20190412 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2019716900 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 112020017813 Country of ref document: BR Kind code of ref document: A2 Effective date: 20200831 |