WO2018230564A1 - コーティング組成物 - Google Patents
コーティング組成物 Download PDFInfo
- Publication number
- WO2018230564A1 WO2018230564A1 PCT/JP2018/022436 JP2018022436W WO2018230564A1 WO 2018230564 A1 WO2018230564 A1 WO 2018230564A1 JP 2018022436 W JP2018022436 W JP 2018022436W WO 2018230564 A1 WO2018230564 A1 WO 2018230564A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- coating composition
- water
- acid
- composition according
- Prior art date
Links
- 0 CN(*)C(C(*)=C)=O Chemical compound CN(*)C(C(*)=C)=O 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/26—Esters containing oxygen in addition to the carboxy oxygen
- C08F220/28—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety
- C08F220/285—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety and containing a polyether chain in the alcohol moiety
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/52—Amides or imides
- C08F220/54—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide
- C08F220/56—Acrylamide; Methacrylamide
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
- C08J5/245—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs using natural fibres
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
- C08J5/249—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs characterised by the additives used in the prepolymer mixture
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L29/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical; Compositions of hydrolysed polymers of esters of unsaturated alcohols with saturated carboxylic acids; Compositions of derivatives of such polymers
- C08L29/02—Homopolymers or copolymers of unsaturated alcohols
- C08L29/04—Polyvinyl alcohol; Partially hydrolysed homopolymers or copolymers of esters of unsaturated alcohols with saturated carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L29/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical; Compositions of hydrolysed polymers of esters of unsaturated alcohols with saturated carboxylic acids; Compositions of derivatives of such polymers
- C08L29/14—Homopolymers or copolymers of acetals or ketals obtained by polymerisation of unsaturated acetals or ketals or by after-treatment of polymers of unsaturated alcohols
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L33/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L39/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a single or double bond to nitrogen or by a heterocyclic ring containing nitrogen; Compositions of derivatives of such polymers
- C08L39/04—Homopolymers or copolymers of monomers containing heterocyclic rings having nitrogen as ring member
- C08L39/06—Homopolymers or copolymers of N-vinyl-pyrrolidones
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L67/00—Compositions of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Compositions of derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D129/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal, or ketal radical; Coating compositions based on hydrolysed polymers of esters of unsaturated alcohols with saturated carboxylic acids; Coating compositions based on derivatives of such polymers
- C09D129/02—Homopolymers or copolymers of unsaturated alcohols
- C09D129/04—Polyvinyl alcohol; Partially hydrolysed homopolymers or copolymers of esters of unsaturated alcohols with saturated carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D139/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a single or double bond to nitrogen or by a heterocyclic ring containing nitrogen; Coating compositions based on derivatives of such polymers
- C09D139/04—Homopolymers or copolymers of monomers containing heterocyclic rings having nitrogen as ring member
- C09D139/06—Homopolymers or copolymers of N-vinyl-pyrrolidones
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D175/00—Coating compositions based on polyureas or polyurethanes; Coating compositions based on derivatives of such polymers
- C09D175/04—Polyurethanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D4/00—Coating compositions, e.g. paints, varnishes or lacquers, based on organic non-macromolecular compounds having at least one polymerisable carbon-to-carbon unsaturated bond ; Coating compositions, based on monomers of macromolecular compounds of groups C09D183/00 - C09D183/16
- C09D4/06—Organic non-macromolecular compounds having at least one polymerisable carbon-to-carbon unsaturated bond in combination with a macromolecular compound other than an unsaturated polymer of groups C09D159/00 - C09D187/00
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/60—Additives non-macromolecular
- C09D7/63—Additives non-macromolecular organic
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/65—Additives macromolecular
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/0008—Organic ingredients according to more than one of the "one dot" groups of C08K5/01 - C08K5/59
- C08K5/0025—Crosslinking or vulcanising agents; including accelerators
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2201/00—Properties
- C08L2201/10—Transparent films; Clear coatings; Transparent materials
Definitions
- the present invention relates to a coating composition containing a specific water-soluble polymer.
- the coating composition is useful as a conductive film, various optical films, a transfer foil, a gas barrier film, and the like. Furthermore, since a cured pattern can be formed, it is useful as various resist materials.
- Water-soluble polymers are used in various applications such as paints, inks, adhesives, and optical films.
- Patent Documents 1 to 4 disclose various coating materials containing a water-soluble polymer.
- An object of the present invention is to provide a coating composition that forms a cured product having excellent uniformity, durability, and transparency.
- the present invention is a coating composition containing a water-soluble polymer selected from the group consisting of (1) polyvinyl alcohol, a modified product or derivative thereof and (2) polyvinylpyrrolidone, a modified product or derivative thereof.
- the present invention is a cured product of the above coating composition.
- the present invention it is possible to provide a coating composition that forms a cured product having excellent uniformity, durability, and transparency. Since the cured product of the present invention is excellent in uniformity, durability and transparency, it is particularly useful as an antistatic film, a protective film, a conductive film, an optical film and the like.
- the coating composition of the present invention contains a water-soluble polymer selected from the group consisting of the following (1) and (2).
- a water-soluble polymer selected from the group consisting of the following (1) and (2).
- Polyvinyl alcohol, its modified product or its derivative include polyvinyl alcohol obtained by polymerizing vinyl alcohol generally called poval, partially saponified polyvinyl alcohol, fully saponified polyvinyl alcohol, (meth) acrylic group modified polyvinyl Alcohol, (meth) acrylamide group modified polyvinyl alcohol, carboxyl group modified polyvinyl alcohol, acetoacetate ester modified polyvinyl alcohol, methylol group modified polyvinyl alcohol, amino group modified polyvinyl alcohol, ester group modified polyvinyl alcohol, carboxyl group modified polyvinyl alcohol, amide Group-modified polyvinyl alcohol, styrylpyridinium group-modified polyvinyl alcohol, quaternary ammonium base-modified polyvinyl alcohol Allyl group modified polyvinyl alcohol, oxypropylene group modified polyvinyl alcohol, urethane group modified polyvinyl alcohol, ether group modified polyvinyl alcohol, phosphate ester group modified polyvinyl alcohol, acetal
- the modification ratio of polyvinyl alcohol is preferably 0.1 to 10 mol%, preferably 0.5 to 6 mol, based on the amount of hydroxyl group of polyvinyl alcohol. 0.0 mol% is more preferable.
- Examples of the monomer include unsaturated carboxylic acids such as maleic acid, maleic anhydride, fumaric acid, crotonic acid, itaconic acid, acrylic acid and methacrylic acid, and esters thereof, ⁇ -olefins such as ethylene and propylene, Allyl sulfonic acid, methallyl sulfonic acid, allyl sulfonic acid soda, methallyl sulfonic acid soda, sulfonic acid soda, sulfonic acid soda monoalkyl malate, disulfonic acid soda alkyl malate, N-methylol acrylamide, acrylamide alkyl sulfonic acid alkali salt Etc.
- unsaturated carboxylic acids such as maleic acid, maleic anhydride, fumaric acid, crotonic acid, itaconic acid, acrylic acid and methacrylic acid, and esters thereof
- ⁇ -olefins such as ethylene and propylene
- Polyvinyl alcohol, its modified product or its derivative may be used alone or in combination of two or more.
- Polyvinyl alcohol, a modified product thereof, or a derivative thereof has a polystyrene-reduced weight average molecular weight (Mw) of 10,000 to 200,000 by gel permeation chromatography (GPC), and a saponification degree (hydrolysis rate) of 85 to 100.
- Mw polystyrene-reduced weight average molecular weight
- GPC gel permeation chromatography
- saponification degree saponification degree
- the (1) polyvinyl alcohol, modified product or derivative thereof includes one or more structural units selected from the group consisting of the following general formulas (III ⁇ ), (III ⁇ ), (III ⁇ ), (III ⁇ ) and (III ⁇ ).
- Polyvinyl alcohol having a photosensitive group can also be used.
- Y 1 , Y 2 and Y 3 each independently represent a direct bond or a divalent linking group, Q 1 , Q 2 and Q 3 each independently represent a photosensitive group,
- An q ⁇ represents a q-valent anion, q represents 1 or 2
- p represents a coefficient for maintaining a neutral charge, * Means a bond.
- Examples of the photosensitive group represented by Q 1 , Q 2 and Q 3 include stilbazolium group, cinnamoyl group, vinyl group, acrylic group, methacryl group, acrylamide group or methacrylamide group, cinnamyl group, cinnamylidene group, cinnamylidene.
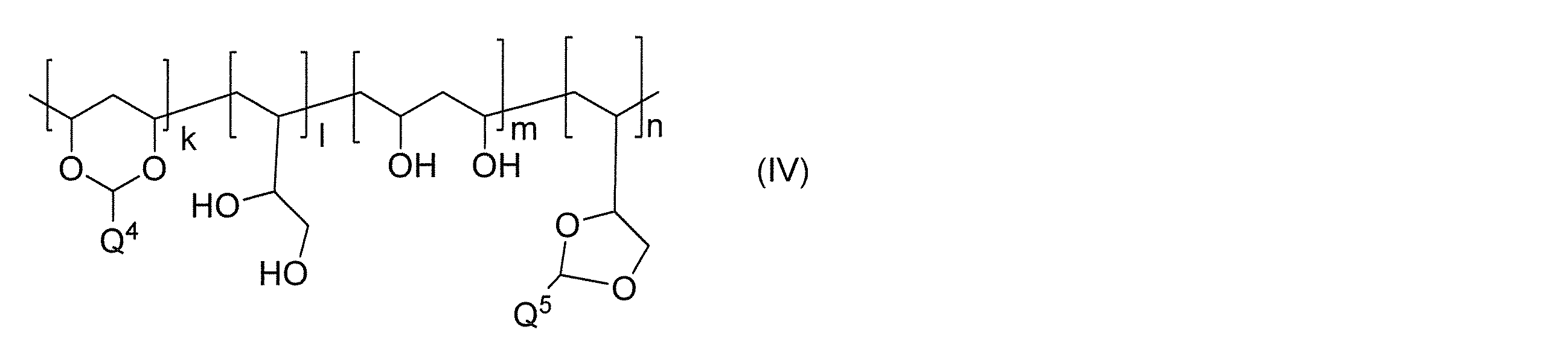
- Q 4 and Q 5 each independently represent a photosensitive group, and 100 ⁇ k + 1 + m + n ⁇ 3000).
- polyvinyl alcohol a modified product thereof or a derivative thereof
- a commercially available product may be used.
- Gohsenol NL-05, NH-18, NH-20, NH-26, NM-14, AH-17 A-300, GM-14L, GL-05, KL-05, GH-23, KH-17 manufactured by Nippon Synthetic Chemical Industry Co., Ltd.
- Gohsenx Z-100, Z-200, Z-300, Z-410 Nippon Synthetic Chemical Industry Co., Ltd.
- Nichigo G-polymer OKS-1081, OKS-1083 Nippon Synthetic Chemical Industry Co., Ltd.
- VF-17, V-S20 (Nippon Vinegar-Poval); Kuraray Poval PVA-103 , PVA-105, PVA-117, PVA-205, PVA-217, PVA-405, PVA-420 (manufactured by Kuraray); Denka Pova Le K-05
- polyvinyl pyrrolidone a modified product thereof or a derivative thereof, in addition to polyvinyl pyrrolidone obtained by polymerizing vinyl pyrrolidone, (meth) acrylic group modified polyvinylpyrrolidone, (meth) acrylamide group modified polyvinylpyrrolidone, carboxyl group modified polyvinylpyrrolidone , Acetoacetate group modified polyvinylpyrrolidone, methylol group modified polyvinylpyrrolidone, amino group modified polyvinylpyrrolidone, ester group modified polyvinylpyrrolidone, carboxyl group modified polyvinylpyrrolidone, amide group modified polyvinylpyrrolidone, styrylpyridinium group modified polyvinylpyrrolidone, quaternary ammonium base Modified polyvinylpyrrolidone, allyl group modified polyvinylpyrrolidone, oxypropylene group modified
- Examples of the monomer include unsaturated carboxylic acids such as maleic acid, maleic anhydride, fumaric acid, crotonic acid, itaconic acid, acrylic acid and methacrylic acid, and esters thereof, ⁇ -olefins such as ethylene and propylene, Allyl sulfonic acid, methallyl sulfonic acid, allyl sulfonic acid soda, methallyl sulfonic acid soda, sulfonic acid soda, sulfonic acid soda monoalkyl malate, disulfonic acid soda alkyl malate, N-methylol acrylamide, acrylamide alkyl sulfonic acid alkali salt Derivatives and the like.
- the above (2) polyvinylpyrrolidone, a modified product thereof or a derivative thereof may be used alone or in combination of two or more at any ratio.
- a conventionally known water-soluble polymer can be used in combination with the coating composition of the present invention.
- Conventional water-soluble polymers include, for example, water-soluble polyesters, oxidized starches, modified starches obtained by etherification / esterification / grafting, cellulose derivatives such as gelatin / casein / carboxymethyl cellulose, and water-soluble polyacrylate resins.
- Water-soluble polycarbonate resin water-soluble polyvinyl acetate resin, water-soluble styrene acrylate resin, water-soluble vinyl toluene acrylate resin, water-soluble polyurethane resin, water-soluble polyamide resin, water-soluble urea resin, water-soluble polycaprolactone resin, water-soluble polystyrene Resin, water-soluble polyvinyl chloride resin, water-soluble polyacrylate resin, water-soluble resin such as water-soluble polyacrylonitrile resin, styrene / butadiene copolymer, acrylate ester copolymer, polyvinyl acetate, ethylene / vinyl acetate copolymer Coalescence Water-soluble polymers, and the like.
- water-soluble polyester those having a carboxyl group and / or a salt of a carboxyl group as a substituent are preferable because they are dissolved in water, emulsified by being dispersed in water, or dissolved in alkaline water.
- a salt, an ammonium salt or an amine salt is more preferable because the effect of the present invention is high.
- the carboxyl group in the water-soluble polyester is preferably adjusted so that the acid value of the produced water-soluble polyester is between 15 and 250 KOHmg / g.
- the acid value is less than 15 KOH mg / g, water dispersion of the polyester (B) becomes difficult and the uniformity is lowered and the film-forming property is lowered.
- the acid value exceeds 250 KOHmg / g, the water resistance becomes poor.
- those having a carboxyl group salt as a substituent can be obtained, for example, by polycondensation of a trifunctional or higher polyvalent carboxylic acid and a compound having one or two hydroxyl groups.
- trifunctional or higher polyvalent carboxylic acid examples include trimellitic acid, trimellitic anhydride, pyromellitic acid, pyromellitic anhydride, 4-methylcyclohexene-1,2,3 tricarboxylic acid anhydride, trimesic acid, etc. Is mentioned. These may be used as a mixture.
- Examples of the compound having one or two hydroxyl groups include aliphatic polyols, polyhydroxy aromatic compounds, polyether diols, polyester diols, polyester polycarbonate diols, polycarbonate diols, polyolefin diols, and compounds of these compounds. Examples thereof include those in which one terminal hydroxyl group is alkoxylated with an alkyl group having 1 to 25 carbon atoms. These may be used as a mixture.
- Examples of the aliphatic polyol include ethylene glycol, 1,2-propanediol, 1,3-propanediol, 2-methyl-1,3-propanediol, and 2-butyl-2-ethyl-1,3-propane.
- polyether diols examples include ethylene oxide adducts such as diethylene glycol and triethylene glycol; propylene oxide adducts such as dipropylene glycol and tripropylene glycol; ethylene oxide and / or propylene oxide adducts of low molecular polyols, Examples include polytetramethylene glycol.
- polyester diols examples include low molecular weight diols, dicarboxylic acids in an amount less than the stoichiometric amount, ester-forming derivatives thereof such as esters, anhydrides, halides, and / or lactones or hydrolyzed derivatives thereof. Examples thereof include those obtained by direct esterification and / or transesterification with a hydroxycarboxylic acid obtained by ring.
- dicarboxylic acid examples include oxalic acid, malonic acid, succinic acid, glutaric acid, adipic acid, azelaic acid, pimelic acid, suberic acid, azelaic acid, sebacic acid, fumaric acid, maleic acid, dodecanedioic acid, 2- Methylsuccinic acid, 2-methyladipic acid, 3-methyladipic acid, 3-methylpentanedioic acid, 2-methyloctanedioic acid, 3,8-dimethyldecanedioic acid, 3,7-dimethyldecanedioic acid, hydrogenated dimer Aliphatic dicarboxylic acids such as acids and dimer acids; phthalic acid, terephthalic acid, isophthalic acid, dimethylmalonic acid, glutaric acid, trimethyladipic acid, 2,2-dimethylglutaric acid, itaconic acid, 1,3-cyclopentanedicarboxylic acid Acid, 1,
- Polycarbonate diols include poly (1,6-hexylene) carbonate and poly (3-methyl-1,5-pentylene) carbonate.
- Polyolefin diols include polybutadiene glycol, hydrogenated polybutadiene glycol, and hydrogenated polyisoprene. Glycol and the like.
- polyether diol and / or polyester diol are particularly preferable.
- the molecular weight of the compound having one or two hydroxyl groups in the same molecule is 300 to 3,000, preferably 500 to 2,000.
- polyhydroxy aromatic compound examples include 4,4-biphenol, 1,1-bis (4-hydroxyphenyl) ethane, 2,2-bis (4-hydroxyphenyl) propane, and 2,2-bis (4 -Hydroxyphenyl) butane, bis (4-hydroxyphenyl) ether, bis (4-hydroxyphenyl) sulfide, bis (4-hydroxyphenyl) sulfone, 4,4 '-(1- ⁇ -methylbenzylidene) bisphenol, 4, 4 '-(1- ⁇ -ethylbenzylidene) bisphenol, 1,1-bis (4-hydroxyphenyl) cyclohexane, 9,9-bis (4-hydroxyphenyl) fluorene, ⁇ , ⁇ '-bis (4-hydroxyphenyl) ) -1,4-diisopropylbenzene, hydrogenated bisphenol compound, resorcinol, Loquinone, 2,5-di-tert-butylhydroquinone, 1,4-dihydroxy
- the water-soluble polyester is obtained by graft polymerization of a polymerizable unsaturated carboxylic acid to a polyester, or as shown in JP-A-62-240318, a glycol or a polyester glycol having a terminal hydroxyl group and a tetracarboxylic acid diester. It can also be obtained by a method in which an anhydride is chain-extended by a selective monoesterification reaction.
- Nichigo Polyester WR-961, WR-1031 manufactured by Nippon Synthetic Chemical Industry
- Pesresin A-680, A-690, A-210, A -695GE manufactured by Takamatsu Yushi Co., Ltd.
- Plus Coat Z-730, Z-760 manufactured by Kyoyo Chemical Co., Ltd.
- Vironal MD-1100, MD-1200, MD-1245, MD-1335, MD-1480, MD-1500 MD-1930, MD-1985, MD-2000 manufactured by Toyobo Co., Ltd.
- the water-soluble polymer contains one or more crosslinking agents selected from the group consisting of zirconium compounds, titanium compounds, bisazide compounds, blocked isocyanates, and epoxy compounds (hereinafter also referred to as “crosslinking agents”).
- crosslinking agents selected from the group consisting of zirconium compounds, titanium compounds, bisazide compounds, blocked isocyanates, and epoxy compounds.
- zirconium compound examples include zirconium halides such as zirconium oxychloride, hydroxyzirconium chloride, zirconium tetrachloride and zirconium bromide; mineral acids such as zirconium sulfate, basic zirconium sulfate, zirconium oxynitrate, zirconium oxyacetate and zirconium oxycarbonate.
- zirconium halides such as zirconium oxychloride, hydroxyzirconium chloride, zirconium tetrachloride and zirconium bromide
- mineral acids such as zirconium sulfate, basic zirconium sulfate, zirconium oxynitrate, zirconium oxyacetate and zirconium oxycarbonate.
- Zirconium salts of organic acids such as zirconium formate, zirconium acetate, zirconium propionate, zirconium caprylate, zirconium stearate, zirconium lactate, zirconium nitrate, zirconium carbonate, zirconium octylate, zirconium citrate, zirconium phosphate; Ammonium zirconium carbonate, sodium zirconium sulfate, ammonium zirconium acetate, ammonium zirconium carbonate, charcoal Zirconium potassium, sodium oxalate, zirconium sodium citrate, zirconium ammonium citrate, other zirconium complex salts such as zirconium lactate ammonium, one or two or more kinds of chelating agents include zirconium chelate complex having a ligand.
- chelating agent examples include hydroxycarboxylic acid or a salt thereof, amino alcohol, aminocarboxylic acid, ⁇ -diketone, dimethylglyoxime, citric acid, tartaric acid, maleic acid, polyhydrazide, and phosphate ester.
- zirconium chelate complex examples include zirconium tetraacetylacetonate, zirconium monoacetylacetonate, zirconium bisacetylacetonate, zirconium monoethylacetoacetate, and zirconium acetate.
- zirconium tetraacetylacetonate zirconium monoacetylacetonate
- zirconium oxychloride zirconium oxynitrate
- zirconium lactate ammonium are preferred because of their high stability, water solubility and reactivity.
- One of these zirconium compounds may be used, or two or more thereof may be used.
- zirconium compound such as zirconium oxychloride, zircozole ZC-2, zircozole ZN, zircozol HA, zircozol AC-7, zircozole ZK-10, zircozole ZN, zircozol ZA-10, zircozol ZA. -20, zirconyl octylate, zirconyl carbonate (manufactured by Daiichi Elemental Chemical Co., Ltd.), ORGATIZ ZB-126, ORGATIZ ZC-700, ORGATIZ ZC-126, ZC-300 (manufactured by Matsumoto Fine Chemical) It is done.
- titanium compound examples include tetramethyl titanate, tetraethyl titanate, tetranormal propyl titanate, tetraisopropyl titanate, tetranormal butyl titanate, tetraisobutyl titanate, tetra-t-butyl titanate, tetraoctyl titanate, tetra (2-ethylhexyl) titanate, Titanium alkoxides such as tetramethyl titanate; oligomers and polymers obtained by hydrolysis reaction of titanium alkoxides such as titanium butyl dimer and titanium butyl tetramer and their derivatives; titanium acetylacetonate, titanium octylene glycolate, titanium tetraacetylacetonate , Titanium chelate complexes such as titanium ethyl acetoacetate, titanium triethanolaluminate, titanium oxalate ; Titanium acylates such as polyhydroxy
- titanium compound for example, ORGATICS TA-10, ORGATICS TC-100, ORGATICS TC-300, ORGATICS TC-310, ORGATICS TC-315 (manufactured by Matsumoto Fine Chemical Co., Ltd.) Etc.
- Examples of the bisazido compound include 4 ′, 4′-diazidostilbene-2-2′-disulfonic acid, 4,4′-diazidobenzalacetophenone-2-sulfonic acid, and 4,4′-diazidostilbene.
- -Water-soluble bisazide compounds such as ⁇ -carboxylic acid and salts thereof such as sodium salt, potassium salt, ammonium salt; p-phenylenebisazide, 4,4'-diazidobenzophenone, 4,4'-diazidostilbene, 4,4′-diazidophenylmethane, 4,4′-diazidobenzalacetophenone, 2,6-di- (4′-azidobenzal) cyclohexanone, 2,6-di- (4′-azidobenzal) -4- Examples thereof include oil-soluble bisazide compounds such as methylcyclohexanone.
- the above-mentioned blocked isocyanate includes a compound obtained by reacting an isocyanate compound with a blocking agent.
- isocyanate compound examples include methyl isocyanate, ethyl isocyanate, propyl isocyanate, n-butyl isocyanate, methyl isocyanate: 2,4-tolylene diisocyanate, 2,6-tolylene diisocyanate, diphenylmethane-4,4′-diisocyanate, p.
- -Aromatic diisocyanates such as phenylene diisocyanate, xylylene diisocyanate, 1,5-naphthylene diisocyanate, 3,3'-dimethyldiphenyl-4,4'-diisocyanate, dianisidine diisocyanate, tetramethylxylylene diisocyanate; isophorone diisocyanate, dicyclohexyl Methane-4,4′-diisocyanate, trans-1,4-cyclohexyl diisocyanate, norbornene diisocyanate Aliphatic diisocyanates such as 1,6-hexamethylene diisocyanate, 2,2,4-trimethylhexamethylene diisocyanate, 2,4,4-trimethylhexamethylene diisocyanate, lysine diisocyanate; Isocyanurate trimers, burette trimers, trimethylolpropane adducts, etc.
- diisocyanates triphenylmethane triisocyanate, 1-methylbenzole-2,4,6-triisocyanate, dimethyltriphenylmethane tetraisocyanate, etc.
- Examples include functional and higher isocyanates.
- carboxylic acid esters such as dimethyl malonate and diethyl malonate: malonic acid, acetylacetone, acetoacetate (methyl acetoacetate, acetoacetate Active methylene compounds such as ethyl acetate; formamide oxime, acetamide oxime, acetoxime, diacetyl monooxime, benzophenone oxime, cyclohexanone oxime, methyl ethyl ketoxime (MEK oxime), methyl isobutyl ketoxime (MIBK oxime), dimethyl ketoxime, diethyl Oxime compounds such as ketoximes; methanol, ethanol, propanol, butanol, 2-ethylhexanol, heptanol, hexanol, octanol, isononyl Monohydric alcohols such as alcohol, stearyl alcohol
- epoxy compound monoepoxy compounds such as aromatic epoxy compounds, alicyclic epoxy compounds, aliphatic epoxy compounds, or polyepoxy compounds can be used.
- Specific examples of the chelate ligand include hydroxycarboxylic acid or a salt thereof, amino alcohol, ⁇ -diketone and the like.
- the blending amount of the cross-linking agent is preferably 0.01 to 3 parts by mass in terms of solid content with respect to 100 parts by mass of the water-soluble polymer, since there is no change in precipitation, thickening, and the like, which is preferable.
- a conventionally known crosslinking agent can be further used in combination.
- Conventionally known crosslinking agents include polyoxazoline compounds, carbodiimide compounds, polyamines, polyols, dicyandiamide derivatives, hydrazine compounds, polyhydrazide compounds (dihydrazide, trihydrazide), aldehydes, methylol compounds, activated vinyl compounds, polyisocyanates. Examples thereof include compounds that react at a heat drying temperature of 100 to 120 ° C., such as a series compound, an alkylene carbonate compound of a phenol type compound, a polyvalent metal salt, and a silane coupling agent.
- the coating composition of the present invention may contain at least one selected from the group consisting of water-soluble polyfunctional (meth) acrylates and water-soluble polyfunctional (meth) acrylamides.
- water-soluble polyfunctional (meth) acrylate examples include polyfunctional (meth) acrylate that dissolves in water, polyfunctional (meth) acrylate that disperses in water and emulsifies, or polyfunctional (meth) acrylate that dissolves in alkaline water.
- the structure includes one having at least one group selected from the group consisting of ethylene oxide group, propylene oxide group, hydroxyl group and carboxyl group, which are hydrophilic functional groups, or ethylene oxide modified, propylene oxide. Denatured ones are mentioned.
- what was described as (meth) acrylate means that either an acrylate or a methacrylate may be sufficient.
- water-soluble polyfunctional (meth) acrylamide examples include polyfunctional (meth) acrylamide that dissolves in water, polyfunctional (meth) acrylamide that disperses in water and emulsifies, or polyfunctional (meth) acrylamide that dissolves in alkaline water.
- the expression (meth) acrylamide means that either acrylamide or methacrylamide may be used.
- water-soluble polyfunctional (meth) acrylate examples include polyethylene glycol diacrylate, trimethylolpropane triacrylate ethylene glycol di (meth) acrylate, 1,4-cyclohexanedimethanol (meth) acrylate, and propylene glycol di (meth).
- Alkylene glycol di (meth) acrylates such as acrylate and butylene glycol di (meth) acrylate; dialkylene glycol di (meth) acrylate such as diethylene glycol di (meth) acrylate, dipropylene glycol di (meth) acrylate and dibutylene glycol di (meth) acrylate ( Meth) acrylate; triethylene glycol di (meth) acrylate, tripropylene glycol di (meth) acrylate and butylene glycol Trialkylene glycol di (meth) acrylate such as (meth) acrylate; tetraalkylene glycol di (meth) such as tetraethylene glycol di (meth) acrylate, tetrapropylene glycol di (meth) acrylate and tetrabutylene glycol di (meth) acrylate Acrylate; Polyalkylene glycol di (meth) acrylate such as polyethylene glycol di (meth) acrylate,
- water-soluble polyfunctional (meth) acrylamide examples include N- [tris (3-acrylamidepropoxymethyl) methyl] (meth) acrylamide, N, N′-diacryloyl-4,7,10-trioxa-1, 13-tridecanediamine, N, N ′, N ′′ -triacryloyldiethylenetriamine, N, N ′- ⁇ [2-acrylamido-2-[(3-acylamidopropoxy) methyl] propane-1,3-diyl] Bis (oxy) ⁇ bis (propane-1,3-diyl) ⁇ diacrylamide, N, N ′, N ′′, N ′ ′′-tetraacryloyltriethylenetetramine and the like.
- water-soluble polyfunctional (meth) acrylates those having a structure represented by the following general formula (I) are preferable because of their high affinity with water.
- R is a hydrogen atom, a methyl group or a halogen atom
- X represents an alkylene group having 1 to 6 carbon atoms
- m is a number of 1 to 30.
- examples of the alkylene group having 1 to 6 carbon atoms include methylene, ethylene, propylene, butylene, isobutylene and the like.
- m is 5 to 20 because of high affinity with water.
- water-soluble polyfunctional (meth) acrylamides those having a structure represented by the following general formula (II) are preferable because of their high affinity with water.
- R is the same as in the general formula (I), and R 11 represents a hydrogen atom or an alkyl group having 1 to 4 carbon atoms.
- water-soluble polyfunctional (meth) acrylate commercially available products can be used.
- hydroxyethyl (meth) acrylate, 1,4-cyclohexanedimethanol (meth) acrylate A-GLY-9E, A- GLY-20E, A-600, A-1000, A-BPE-30, ATM-35E, A-PG5027E, A-PG5054E, 14G, GLY-9E, GLY-20E (manufactured by Shin-Nakamura Chemical Co., Ltd.), PEG400DA-D EBECRYL 11 (manufactured by Daicel Ornex), 9EG-A, 14EG-A (Kyoeisha Chemical), and the like.
- water-soluble polyfunctional (meth) acrylamide examples include hydroxyethyl (meth) acrylamide, (meth) acryloylmorpholine, diethyl (meth) acrylamide, dimethylaminopropyl (meth) acrylamide FFM-2, FFM-3, and FFM-4.
- FFM-5 manufactured by Fuji Film
- organic acid can be further added to the coating composition of the present invention.
- organic acid any weakly acidic compound having a carboxyl group can be used without limitation.
- the blending amount of the organic acid is preferably 0.01 to 1 part by mass in terms of solid content with respect to 100 parts by mass of the water-soluble polymer because there is no change in precipitation, phase separation, thickening and the like.
- the reaction can be controlled and the storage stability can be enhanced by adjusting the pH by adding an organic base.
- organic base include amine compounds and imine compounds.
- the coating composition of the present invention may contain a metal component derived from a cross-linking agent.
- the metal content is 0.01 to 3% by mass in terms of solid content in the coating composition, and preferably is 0.00. It is 1 to 2% by mass.
- a coupling agent, a sensitizer, a surfactant, a photopolymerization initiator and the like can be added to the coating composition of the present invention as necessary.
- Examples of the coupling agent include alkyl functionalities such as dimethyldimethoxysilane, dimethyldiethoxysilane, methylethyldimethoxysilane, methylethyldiethoxysilane, methyltrimethoxysilane, methyltriethoxysilane, ethyltrimethoxysilane, and ethyltrimethoxysilane.
- Alkenyl functional alkoxysilanes such as functional alkoxysilane, vinyltrichlorosilane, vinyltrimethoxysilane, vinyltriethoxysilane, allyltrimethoxysilane, 3-methacryloxypropyltriethoxysilane, 3-methacryloxypropyltrimethoxysilane, 3-methacryloxypropylmethyldiethoxysilane, 3-methacryloxypropylmethyldimethoxysilane, 2-methacryloxypropyltrimethoxysilane, ⁇ -g Epoxy functional alkoxysilanes such as Sidoxypropyltrimethoxysilane, ⁇ -Glycidoxypropylmethyldiethoxysilane, ⁇ - (3,4-epoxycyclohexyl) ethyltrimethoxysilane, N- ⁇ (aminoethyl) - ⁇ - Aminofunctional alkoxysilanes such as aminopropyltrime
- the sensitizer is a compound capable of expanding the wavelength range in which light can be adapted when cured by light irradiation.
- the surfactant examples include fluorine surfactants such as perfluoroalkyl phosphates and perfluoroalkyl carboxylates, anionic surfactants such as higher fatty acid alkali salts, alkyl sulfonates, and alkyl sulfates, and higher amines. Cationic surfactants such as halogenates and quaternary ammonium salts, nonionic surfactants such as polyethylene glycol alkyl ethers, polyethylene glycol fatty acid esters, sorbitan fatty acid esters and fatty acid monoglycerides, amphoteric surfactants, silicone surfactants A surfactant such as an agent can be used. These surfactants may be used in combination.
- Hydrogen abstraction-type photopolymerization initiators such as benzophenone, thioxanthone, 1-chloro-4-propoxythioxanthone, isopropylthioxanthone, diethylthioxanthone, ethyl anthraquinone; phenyl biphenyl ketone, 1-hydroxy-1-benzoylcyclohexane ( ⁇ -hydroxyalkyl) Phenone), benzoin, benzyldimethyl ketal, 1-benzyl-1-dimethylamino-1- (4′-morpholinobenzoyl) propane, 2-morpholy-2- (4′-methylmercapto) benzoylpropane, 4-benzoyl-4 '-Methyldiphenyl sulfide, benzoin butyl ether, 2-hydroxy-2-benzoylpropane, 2-hydroxy-2- (4'-isopropyl) benzoylpropane, 4-butylbenz
- a photodegradable photopolymerization initiator is preferable from the viewpoint of reactivity.
- water-soluble initiators such as IRGACURE 2959, IRGACURE 819DW (manufactured by BASF), ESACURE ONE, ESACURE 1001M, ESACURE KIP 150, ESACURE DP 250 (Lamberti), etc. Is preferred because of its high affinity.
- a thermal polymerization initiator such as a dye, a dye, an antifoaming agent, a thickening agent, a leveling agent, Organic metal coupling agents, thixotropic agents, carbon compounds, metal fine particles, metal oxides, flame retardants, plasticizers, light stabilizers, heat stabilizers, anti-aging agents, elastomer particles, chain transfer agents, polymerization inhibitors, UV absorption
- Various resin additives such as an agent, an antioxidant, an antistatic agent, a mold release agent, a flow regulator, an adhesion promoter, a water-soluble preservative, a conductive substance, and an unsaturated monomer can be added.
- the coating composition of the present invention may contain a solvent.
- a solvent water is preferable, and it is preferable that the solvent is only one liquid because it does not attack the organic material when it is applied on the organic material with a low environmental load.
- the solvent it can be used in combination with an organic solvent.
- organic solvent examples include ketones such as methyl ethyl ketone, methyl amyl ketone, diethyl ketone, acetone, methyl isopropyl ketone, methyl isobutyl ketone, cyclohexanone and 2-heptanone; ethyl ether, dioxane, tetrahydrofuran, 1,2-dimethoxyethane.
- Ether solvents such as 1,2-diethoxyethane and dipropylene glycol dimethyl ether; methyl acetate, ethyl acetate, n-propyl acetate, isopropyl acetate, n-butyl acetate, cyclohexyl acetate, ethyl lactate, dimethyl succinate and texanol
- Ester solvents such as ethylene glycol monomethyl ether and cellosolve solvents such as ethylene glycol monoethyl ether; methanol, ethanol, iso- or n-propanol Alcohol solvents such as iso- or n-butanol and amyl alcohol; ethylene glycol monomethyl acetate, ethylene glycol monoethyl acetate, propylene glycol-1-monomethyl ether (PGM), propylene glycol-1-monomethyl ether-2-acetate (PGMEA) ), Ether ester solvents such as dipropylene glycol monomethyl
- the coating composition of the present invention is applied onto a supporting substrate such as glass, metal, paper, or plastic by a known means such as a spin coater, bar coater, roll coater, curtain coater, various printing or dipping.
- a coating composition of the present invention can be once applied onto a support substrate such as a film and then transferred onto another support substrate. There is no limitation on the application method.
- the material for the transparent support substrate examples include inorganic materials such as glass; cellulose esters such as diacetylcellulose, triacetylcellulose (TAC), propionylcellulose, butyrylcellulose, acetylpropionylcellulose, and nitrocellulose; polyamides; polycarbonates; polyethylenes Polyesters such as terephthalate, polyethylene naphthalate, polybutylene terephthalate, poly-1,4-cyclohexanedimethylene terephthalate, polyethylene-1,2-diphenoxyethane-4,4′-dicarboxylate, polybutylene terephthalate; polystyrene; polyethylene Polyolefins such as polypropylene and polymethylpentene; acrylic resins such as polymethyl methacrylate; polycarbonate; Emissions; polyether sulfone; polyether ketone; polyetherimides; polyoxyethylene, and polymer materials such as norbornene resins.
- the transmittance of the transparent support substrate is
- the irradiation conditions such as the wavelength, intensity and irradiation time of the irradiated light depend on the activity of the photoinitiator and the light used. It is appropriately adjusted depending on the activity of the polymerizable resin.
- the light wavelength is preferably a wavelength peak of 350 to 400 nm, and more preferably a wavelength peak of 360 to 380 nm, in order to allow light to sufficiently enter the inside.
- the light intensity is preferably 1 to 500 mW / cm 2 , more preferably 5 to 300 mW / cm 2
- the irradiation time is preferably 1 to 500 seconds, more preferably 5 to 300 seconds.
- the heating is performed at 50 to 200 ° C., preferably 70 to 150 ° C., for 10 seconds to 1 hour. If it is lower than 50 ° C, the crosslinking reaction may not proceed. On the other hand, when the temperature is higher than 200 ° C., the water-soluble polymer may be decomposed or the transparency of the optical film may be lowered.
- Specific applications of the coating composition of the present invention include glasses, optical materials represented by imaging lenses, antistatic films, optical films, conductive films, protective films, heat ray shielding materials, transfer foils, printing plates, Insulating varnish, insulating sheet, laminated board, printed circuit board, flexible display substrate, touch panel substrate, mask for printing, molding material, putty, building material, nail material, cosmetics, siding, glass fiber impregnating agent, sealing agent, for semiconductors ⁇ Lens sheets such as passivation films for solar cells, interlayer insulating films, protective films, prism lens sheets used for backlights of liquid crystal display devices, Fresnel lens sheets used for screens of projection televisions, lenticular lens sheets, etc. Lens part or a backlight using such a sheet.
- Lens sheets such as passivation films for solar cells, interlayer insulating films, protective films, prism lens sheets used for backlights of liquid crystal display devices, Fresnel lens sheets used for screens of projection televisions, lenticular lens sheets, etc. Lens part or a backlight
- LCD color filter protective films and spacers DNA separation chips, microreactors, nanobio devices, recording materials for hard disks, solid-state imaging devices, solar cell panels, light emitting diodes, organic light emitting devices, electrode protective materials, luminescent films, fluorescence Films, MEMS elements, actuators, holograms, plasmon devices, polarizing plates, polarizing films, alignment films, microlenses and other optical lenses, optical elements, retardation films, optical connectors, optical waveguides, optical molding castings, foods , Beverage containers, food packaging materials, dental materials, sanitary ware, housing equipment such as bathtubs, and the like.
- the substrate that can be applied as the coating agent include metals, wood, rubber, plastics, glass, ceramic products, paper, and cloth.
- the coating composition of the present invention can form a cured product having excellent transparency, it is useful for optical elements represented by retardation films, optical waveguides, optical lenses and the like.
- the coating composition of the present invention can form a cured product having a small retardation, it is useful for a retardation film.
- the coating composition of the present invention is useful for optical waveguides because it can form a cured product with small propagation loss.
- the cloth composition or paper is impregnated with the coating composition of the present invention and cured, thereby imparting water resistance, rigidity, and the like.
- cured material from the coating composition of this invention, it can carry out by the following aspect.
- a layer composed of a plurality of compositions having different solubility in a developing solution is applied to a support substrate, and then repeatedly applied and dried.
- step (2) a predetermined portion of the film is irradiated with an active energy ray for exposure, and in step (3), the film is repeatedly processed with a plurality of developers.
- Another embodiment for producing the patterned cured product is to repeat a series of steps of performing the step (1), the step (2) and the step (3) a plurality of times, and the composition in the step (1) Each flow has a different solubility in the developer, and the developer used for development in step (3) is different for each series of flows.
- a step (1) a lower layer made of the composition is applied to a support substrate and dried, and then a film obtained from the composition is laminated as an upper layer. The composition constituting the upper layer and the lower layer is different in solubility in the developer.
- step (2) a predetermined portion of the film is irradiated with an active energy ray to be exposed, and in step (3), the film is repeatedly processed with a plurality of developers.
- the film is formed as the step (1), it is precured before being exposed as the step (2), or after being developed as the step (3).
- known methods can be used, for example, curtain coating method, extrusion coating method, roll coating method, spin coating method, dip coating method, bar coating method, die coater, curtain coater, spray coating.
- a known means such as various printing methods such as a printing method, a slide coating method, a blade coating method, a gravure coating method, a printing coating method such as screen printing, and an electrostatic coating method, and dipping can be used.
- a film which is applied to another support dried and then peeled off from the support.
- a dry film can be used.
- the support examples include cellulose esters such as diacetylcellulose, triacetylcellulose (TAC), propionylcellulose, butyrylcellulose, acetylpropionylcellulose, and nitrocellulose; polyamide; polycarbonate; polyethylene terephthalate, polyethylene naphthalate, polybutylene terephthalate, poly Polyesters such as 1,4-cyclohexanedimethylene terephthalate, polyethylene-1,2-diphenoxyethane-4,4'-dicarboxylate, polybutylene terephthalate; polystyrene; polyolefins such as polyethylene, polypropylene, polymethylpentene; Acrylic resins such as methyl methacrylate; Polycarbonate; Polysulfone; Polyethersulfone; Poly -Terketone; Polyetherimide; Polymer materials such as polyoxyethylene, norbornene resin, metal, wood, rubber, plastic, soda glass, quartz glass, ceramic products, paper, cloth, canvas, semiconductor substrate,
- the coating composition of the present invention and the cured product obtained from the coating composition will be specifically described with reference to production examples, examples, evaluation examples, and comparative examples.
- A-1 GOHSENOL NL-05 (saponification degree 99) manufactured by Nippon Synthetic Chemical Industry A-2: GOHSENOL GL-05 (degree of saponification 87) manufactured by Nippon Synthetic Chemical Industry A-3: Goseinex Z-200 (saponification degree 98) manufactured by Nippon Synthetic Chemical Industry A-4: GOHSEX Z-300 (saponification degree 98) manufactured by Nippon Synthetic Chemical Industry A-5: Goseinex Z-100 (saponification degree 99) manufactured by Nippon Synthetic Chemical Industry A-6: Goseinex Z-220 (saponification degree 92) manufactured by Nippon Synthetic Chemical Industry PV-1: A-1 aqueous solution PV-2: A-2 aqueous solution PV-3: A-3 aqueous solution PV-4: A-4 aqueous solution PV-5: A-5 aqueous solution PV-6: A -6 aqueous solution
- PV-7 10% by mass aqueous solution of polyvinylpyrrolidone K-90 (manufactured by Nippon Shokubai)
- PV-8 Reaction of Gohsenol GL-05 with N-methylolacrylamide to 3.0 mol% with respect to the amount of hydroxyl group of polyvinyl alcohol
- Aqueous solution adjusted to 10% by mass PV-10 G-Polymer OKS-1083 was reacted with formylstyrylpyridinium at 3.4 mol% with respect to the amount of hydroxyl groups of polyvinyl alcohol to adjust the solid content to 10% by mass.
- Aqueous solution PV-11 Gosenol GL-05 and formylschili An aqueous solution prepared by reacting pyridinium to 3.4 mol% with respect to the amount of hydroxyl group of polyvinyl alcohol to adjust the solid content to 10% by mass
- B-1 Orgatics TA-10 (manufactured by Matsumoto Fine Chemical Co .; tetraisopropyl titanate, Component concentration 99%, Ti content 17%)
- B-2 ORGATICS TC-100 (Matsumoto Fine Chemical Co., Ltd .; titanium acetylacetonate, 2-propanol solution (solvent chelate), component concentration 75%, Ti content 10%)
- B-3 ORGATICS TC-315 (Matsumoto Fine Chemical Co., Ltd .; titanium lactate, aqueous solution (aqueous chelate), component concentration 44%, Ti content 8%)
- B-4 Orugatix ZC-700 (manufactured by Matsumoto Fine Chemical Co .;
- F-2 NK ester A-GLY-20E (water-soluble polyfunctional acrylate) manufactured by Shin-Nakamura Chemical Co., Ltd.
- F-3 Shin Nakamura Chemical Co., Ltd.
- NK Economer A-PG5054E Water-soluble polyfunctional acrylate
- F-4 FFM-3 (water-soluble polyfunctional acrylamide) manufactured by Fuji Film
- F-5 Hydroxyethylacrylamide (HEAA) (water-soluble monofunctional acrylamide) manufactured by KJ Chemicals
- G-1 IRSFACURE 2959 manufactured by BASF
- a Nippon Denshoku haze meter NDH5000 was used for the measurement, and the determination criteria were as follows.
- ⁇ Uniform coating film, less than 1 haze
- ⁇ Uniform coating film, haze 1 or more and less than 3
- ⁇ Whitening or unevenness of the coating film was observed and haze 3 or more
- Part of the coating film was peeled or eluted ⁇ Examples 27 and 28 and Comparative Examples 2 and 5> a2) Preparation of coating film
- Each coating composition was applied on a glass substrate using a spin coater, pre-baked for 5 minutes on a hot plate at 90 ° C., and then using a high-pressure mercury lamp, including a wavelength of 365 nm
- An evaluation substrate was produced by irradiating with light at 500 mJ / cm 2 and further heating at 140 ° C.
- the substrates prepared in the above a1) and a2) were evaluated by haze measurement at the same time as confirming the surface state of the coating film after being left for 24 hours at 85 ° C. and 85% RH.
- a Nippon Denshoku haze meter NDH5000 was used for the measurement, and the determination criteria were as follows. ⁇ : Uniform coating film, less than 1 haze ⁇ : Uniform coating film, haze 1 or more and less than 3 ⁇ : Whitening or unevenness of the coating film was observed and haze 3 or more ⁇ : Part of the coating film was peeled or eluted
- the liquid crystal composition was injected into a TN cell for liquid crystal evaluation (cell thickness 5 ⁇ m, electrode area 8 mm ⁇ 8 mm alignment film JALS2096), and VHR was measured using VHR-1A (manufactured by Toyo Technica).
- the measurement conditions were pulse voltage width: 60 ⁇ s, frame period: 16.7 ms, wave height: ⁇ 5 V, measurement temperature: 25 ° C.
- ⁇ VHR decrease rate is less than 1% ⁇ : VHR decrease rate is 1 to 3%
- X VHR reduction rate exceeds 3%
- the coating composition of the present invention is useful as a coating material for liquid crystal displays and a resist material because it has a low VHR reduction rate and low liquid crystal contamination.
- each coating composition was applied using a spin coater on a glass substrate, after prebaked for 5 minutes on a hot plate at 90 ° C., using a high-pressure mercury lamp, 500 mJ / cm 2 irradiation with light having a wavelength of 365nm Further, the substrate for evaluation was produced by heating at 140 ° C. for 15 minutes.
- the spin coating conditions were adjusted with a stylus profilometer (Dektak 150 manufactured by ULVAC) so that the film thickness was 5.0 to 5.5 ⁇ m.
- the ultraviolet-visible absorption (UV-Vis) spectrum of the evaluation substrate was measured with an ultraviolet-visible absorbance meter V-670 manufactured by JASCO. Using the glass substrate as a reference, the transmittance of the coating film at 400 nm was calculated.
- each coating composition was applied using a spin coater on a glass substrate, after prebaked for 5 minutes on a hot plate at 90 ° C., using a high-pressure mercury lamp, 500 mJ / cm 2 irradiation with light having a wavelength of 365nm Further, the substrate for evaluation was produced by heating at 140 ° C. for 15 minutes.
- the spin coating conditions were adjusted with a stylus profilometer (Dektak 150 manufactured by ULVAC) so that the film thickness was 5.0 to 5.5 ⁇ m.
- the value of the in-plane retardation R0 at 500 nm of the evaluation substrate was measured with RETS-100 manufactured by Otsuka Electronics Co., Ltd.
- the pattern width of the pattern-like cured product was within 50 ⁇ 3 ⁇ m in any substrate produced with any coating composition, and a good pattern was obtained.
- the coating composition of the present invention is excellent in uniformity, durability and transparency, and can form a cured product with low liquid crystal contamination and a small phase difference.
- Examples 32 and 33 and Comparative Example 8 (Production of optical waveguide) A coating composition shown in Table 7 below was laminated on a fired silicon substrate as a clad material at a thickness of 30 ⁇ m by spin coating, irradiated with UV light of 10 mW / cm 2 for 200 seconds, and then heated at 120 ° C. for 15 minutes. To form a cured product. Next, on the formed cured product, a coating composition shown in Table 7 below was laminated as a core material with a thickness of 20 ⁇ m by a spin coating method, and a light amount of 10 mW / cm 2 was used using a negative photomask. Were irradiated for 40 seconds. Development with water and heat curing at 140 ° C.
- the coating composition shown in Table 7 below was laminated as a clad material so as to have a thickness of 30 ⁇ m from the upper surface of the patterned cured product by spin coating, and irradiated with ultraviolet rays having a light amount of 10 mW / cm 2 for 200 seconds, then 140 An optical waveguide was produced on a silicon substrate by heating at 30 ° C. for 30 minutes.
- the optical transmission loss was calculated by subtracting the connection loss from the transmission loss measured by the cutback method using an ASE (Amplified Spontaneous Emission) light source with an isolator having a wavelength of 850 nm. Table 7 shows the measurement results. It can be said that the smaller the optical transmission loss, the more suitable for long-distance transmission.
- the coating composition of the present invention is useful for the production of an optical waveguide because it can be developed with water and can form a cured product with low environmental load and low optical transmission loss.
- UV irradiation means that ultraviolet rays having a light amount of 10 mW / cm 2 were not irradiated in the production of the coated paper. Moreover,-in the table means not evaluated. ⁇ : Two toilet papers were kept in a bonded state. ⁇ : Two toilet papers were peeled but maintained in shape. ⁇ : Two toilet papers were peeled off, and the shape was further changed. could not be maintained
- the coating composition of the present invention improves the water resistance of paper, and is therefore useful as a coating agent for paper.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Wood Science & Technology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Manufacturing & Machinery (AREA)
- Paints Or Removers (AREA)
- Application Of Or Painting With Fluid Materials (AREA)
Abstract
Description
(1)ポリビニルアルコール、その変性体又はその誘導体
(2)ポリビニルピロリドン、その変性体又はその誘導体
Q1、Q2及びQ3は、それぞれ独立に感光性基を表し、
Anq-はq価のアニオンを表し、qは1又は2を表し、pは電荷を中性に保つ係数を表し、
*は結合手を意味する。)
上記光分解型光重合開始剤の中でも、IRGACURE 2959、IRGACURE 819DW(BASF社製)、ESACURE ONE、ESACURE 1001M、ESACURE KIP 150、ESACURE DP 250(Lamberti社)等の水溶性開始剤等が、水への親和性が高いので好ましい。
パターン状硬化物を製造する一つの態様は、工程(1)として、支持基体に、現像液に対する溶解性が互いに異なる複数の組成物からなる層を、一層塗布した後に乾燥することを繰り返して塗布し、続いて工程(2)として、膜の所定部分に活性エネルギー線を照射して露光し、さらに工程(3)として、膜を複数の現像液で繰り返し処理するものである。
パターン状硬化物を製造する別の態様は、工程(1)、工程(2)及び工程(3)を行う一連の流れを複数回繰り返すもので、工程(1)における組成物は、前記一連の流れごとに、現像液に対する溶解性がそれぞれに異なるものであり、工程(3)において現像に用いる現像液は、前記一連の流れごとにそれぞれ異なるものを用いる。
パターン状硬化物を製造する別の態様は、工程(1)として、支持基体に、組成物からなる下層を塗布した後に乾燥し、続いて上層として、組成物から得られるフィルムを積層して膜を形成するもので、上層と下層を構成する組成物は、現像液に対する溶解性が互いに異なるものである。続いて工程(2)として、膜の所定部分に活性エネルギー線を照射して露光し、さらに工程(3)として、膜を複数の現像液で繰り返し処理を行う。
加熱は、ホットプレート上に積層物を載せ昇温したり、サーマルヘッド、クリーン・オーブン等の加熱装置を使用して、プロキシミティ方式や吸着方式、コンベア方式等があり、またそれらを組み合わせることにより行うことができる。
加熱は、50~200℃で数秒~1時間行う。
<水溶性重合体水溶液の調製>
まず、イオン交換水900.0gを室温で撹拌しているところに、下記A-1~A-6いずれかの水溶性重合体100.0gを徐々に添加した。室温で10分間撹拌した後に、内温が85~90℃になるまで加熱し、その温度で1時間撹拌を継続した。溶解を確認した後、室温まで冷却した。調製した水溶液を1μmフィルターでろ過して、下記PV-1~PV-6の水溶性重合体水溶液を得た。
A-1:日本合成化学工業製 ゴーセノールNL-05(けん化度99)
A-2:日本合成化学工業製 ゴーセノールGL-05(けん化度87)
A-3:日本合成化学工業製 ゴーセネックス Z-200(けん化度98)
A-4:日本合成化学工業製 ゴーセネックス Z-300(けん化度98)
A-5:日本合成化学工業製 ゴーセネックス Z-100(けん化度99)
A-6:日本合成化学工業製 ゴーセネックス Z-220(けん化度92)
PV-1:A-1の水溶液
PV-2:A-2の水溶液
PV-3:A-3の水溶液
PV-4:A-4の水溶液
PV-5:A-5の水溶液
PV-6:A-6の水溶液
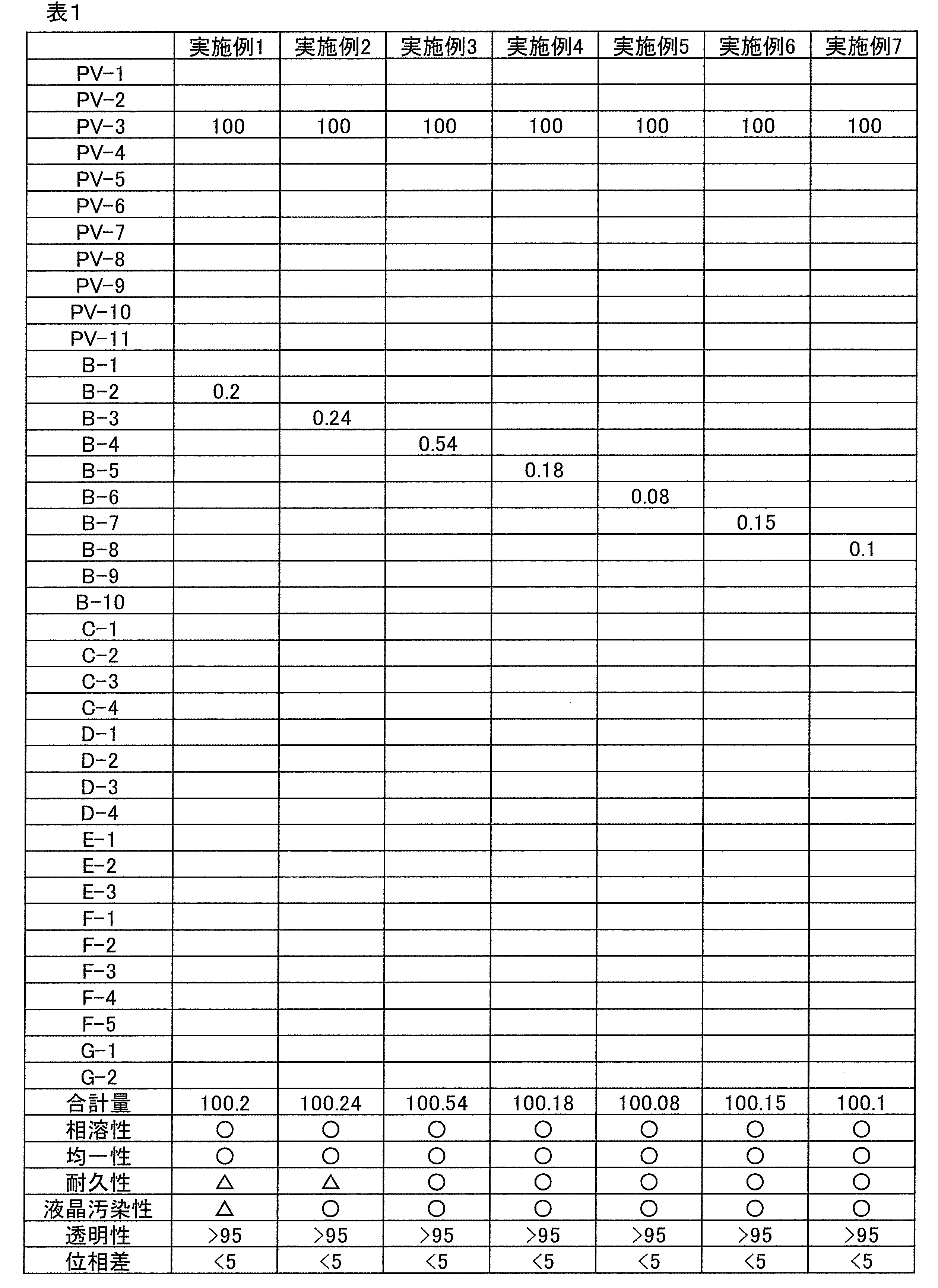
下記の表1~6に示す各成分を混合し、室温で1時間撹拌後、1μmフィルターでろ過し、実施例及び比較例のコーティング組成物を得た。なお、表1~表6中の各成分の詳細は下記の通りである。各成分の配合量及び合計量は質量基準である。ただし、PV-1~PV-11の配合量は、固形分換算した量である。
PV-7:ポリビニルピロリドンK-90(日本触媒製)の10質量%水溶液
PV-8:ゴーセノールGL-05にN-メチロールアクリルアミドをポリビニルアルコールの水酸基量に対して3.0mol%となるように反応させて固形分を10質量%に調整した水溶液
PV-9:G-Polymer OKS-1083にN-メチロールアクリルアミドをポリビニルアルコールの水酸基量に対して2.5mol%となるように反応させて固形分を10質量%に調整した水溶液
PV-10:G-Polymer OKS-1083にホルミルスチリルピリジニウムをポリビニルアルコールの水酸基量に対して3.4mol%となるように反応させて固形分を10質量%に調整した水溶液
PV-11:ゴーセノールGL-05にホルミルスチリルピリジニウムをポリビニルアルコールの水酸基量に対して3.4mol%となるように反応させて固形分を10質量%に調整した水溶液
B-1:オルガチックスTA-10(マツモトファインケミカル社製;テトライソプロピルチタネート、成分濃度99%、Ti含有量17%)
B-2:オルガチックスTC-100(マツモトファインケミカル社製;チタンアセチルアセトネート,2-プロパノール溶液(溶剤系キレート)、成分濃度75%、Ti含有量10%)
B-3:オルガチックスTC-315(マツモトファインケミカル社製;チタンラクテート,水溶液(水系キレート)、成分濃度44%、Ti含有量8%)
B-4:オルガチックスZC-700(マツモトファインケミカル社製;ジルコニウムテトラアセチルアセトネートのトルエン/メタノール溶液(溶剤系キレート)、成分濃度20%、Zr含有量4%)
B-5:オルガチックスZC-126(マツモトファインケミカル社製;オキシ塩化ジルコニウムの水溶液(水系キレート)、成分濃度30%、Zr含有量11%)
B-6:ジルコゾールZN(第一稀元素化学工業社製;オキシ硝酸ジルコニウムの水溶液、成分濃度 46%、Zr含有量25%)
B-7:ジルコゾールAC-7(第一稀元素化学工業社製;炭酸ジルコニウムアンモニウムの水溶液、成分濃度30%、Zr含有量13%)
B-8:ジルコゾールZA-20(第一稀元素化学工業社製;オキシ酢酸ジルコニウムの水溶液、成分濃度35%、Zr含有量20%)
B-9:オルガチックスWS-700(マツモトファインケミカル製;有機チタン変性ポリエチレンイミン、成分濃度15%、Ti含有量5%)
B-10:日本触媒製、エポクロスWS-700(オキサゾリン化合物、固形分25質量%)
C-1:DL-乳酸
C-2:クエン酸
C-3:酢酸
C-4:グリコール酸
D-1:5%アンモニア水
D-2:トリエチルアミン
D-3:2.38%TMAH水溶液
D-4:ポリエチレンイミン70000(純正化学製、30%水溶液)
E-1:ヒドロキシプロピルセルロースNISSO HPC L(日本曹達製)の10%水溶液
E-2:高松油脂製、ペスレジンA-690(親水基:COOH、固形分20質量%、酸価50mgKOH/g)
E-3:日本合成化学工業製、ポリエスターW-1031(親水基:COOH、固形分30質量%、酸価80mgKOH/g)
F-1:新中村化学工業社製 NKエステル A-600(水溶性多官能アクリレート)
F-2:新中村化学工業社製 NKエステル A-GLY-20E(水溶性多官能アクリレート)
F-3:新中村化学工業社製 NKエコノマー A-PG5054E(水溶性多官能アクリレート)
F-4:富士フィルム社製 FFM-3(水溶性多官能アクリルアミド)
F-5:KJケミカルズ社製 ヒドロキシエチルアクリルアミド(HEAA)(水溶性単官能アクリルアミド)
G-1:BASF社製 IRGACURE 2959
G-2:N,N-ビス(2-ヒドロキシエチル)ブタン-1-アミニウム=フェニル(2,4,6-トリメチルベンゾイル)ホスフィネート
上記実施例1~31で得られたコーティング組成物及び比較例1~7で得られた比較コーティング組成物について、下記評価を行った。評価結果を表1~6に示す。
実施例1~31で得られたコーティング組成物及び比較例1~7で得られた比較コーティング組成物の状態を目視で確認した。判定基準は下記の通りである。
○:透明で均一
△:白濁
×:相溶せず(相分離)
<実施例1~26、29~31及び比較例1、3、4、6、7>
a1)塗膜の作製
各コーティング組成物を、ガラス基板上にスピンコーターを用いて塗布し、90℃のホットプレートで5分間のプリベークを行った後、140℃で15分間加熱を行うことで評価用基板を作製した。スピンコートの条件は、触針式形状測定器(アルバック製Dektak150)により膜厚が5.0~5.5μmとなるように調整した。
b1)塗膜の状態
作製した基板上の塗膜について、目視で表面の状態を確認すると同時に、ヘイズ測定によって評価を行った。測定には日本電色製ヘイズメーターNDH5000を使用し、判定基準は下記の通りとした。
○:塗膜が均一、ヘイズ1未満
△:塗膜が均一、ヘイズ1以上3未満
×:塗膜が白化あるいはムラが認められ且つヘイズ3以上
××:塗膜の一部が剥離、あるいは溶出
<実施例27、28及び比較例2、5>
a2)塗膜の作製
各コーティング組成物を、ガラス基板上にスピンコーターを用いて塗布し、90℃のホットプレートで5分間のプリベークを行った後、高圧水銀ランプを用い、365nmの波長を含む光を500mJ/cm2照射し、更に140℃で15分間加熱を行うことで評価用基板を作製した。スピンコートの条件は、触針式形状測定器(アルバック製Dektak150)により膜厚が5.0~5.5μmとなるように調整した。
b2)塗膜の状態
作製した基板上の塗膜について、目視で表面の状態を確認すると同時に、ヘイズ測定によって評価を行った。測定には日本電色製ヘイズメーターNDH5000を使用し、判定基準は下記の通りとした。
○:塗膜が均一、ヘイズ1未満
△:塗膜が均一、ヘイズ1以上3未満
×:塗膜が白化あるいはムラが認められ且つヘイズ3以上
上記a1)及びa2)で作製した基板を85℃、85%RHの条件で24時間放置した後の塗膜の表面状態を確認すると同時に、ヘイズ測定によって評価を行った。測定には日本電色製ヘイズメーターNDH5000を使用し、判定基準は下記の通りとした。
○:塗膜が均一、ヘイズ1未満
△:塗膜が均一、ヘイズ1以上3未満
×:塗膜が白化あるいはムラが認められ且つヘイズ3以上
××:塗膜の一部が剥離、あるいは溶出
上記a1)及びa2)で作製した基板の塗膜上に、下記液晶化合物No.1~No.11よりなる液晶組成物を60℃で24時間接触させた後、液晶組成物を取り出した。接触前後における液晶組成物の電圧保持率(VHR:Voltage Holding Ratio)をそれぞれ測定し、VHRの低下率((接触前のVHR-接触後のVHR)/接触前のVHR×100%)を求め、下記基準により評価を行った。評価は、液晶組成物を液晶評価用TNセル(セル厚5μm、電極面積8mm×8mm配向膜JALS2096)に注入し、VHRをVHR-1A(東陽テクニカ製)を用い測定した。測定条件は、パルス電圧幅:60μs、フレーム周期:16.7ms、波高:±5V、測定温度:25℃とした。
○:VHRの低下率が1%未満
△:VHRの低下率が1~3%
×:VHRの低下率が3%超
各コーティング組成物をガラス基板上にスピンコーターを用いて塗布し、90℃のホットプレートで5分間のプリベークを行った後、高圧水銀ランプを用い、365nmの波長を含む光を500mJ/cm2照射し、更に140℃で15分間加熱を行うことで評価用基板を作製した。スピンコートの条件は、触針式形状測定器(アルバック製Dektak150)により膜厚が5.0~5.5μmとなるように調整した。評価用基板の紫外可視吸収(UV-Vis)スペクトルを日本分光製紫外可視吸光度計V-670にて測定した。ガラス基板をリファレンスとし、塗膜の400nmにおける透過率を算出した。
各コーティング組成物をガラス基板上にスピンコーターを用いて塗布し、90℃のホットプレートで5分間のプリベークを行った後、高圧水銀ランプを用い、365nmの波長を含む光を500mJ/cm2照射し、更に140℃で15分間加熱を行うことで評価用基板を作製した。スピンコートの条件は、触針式形状測定器(アルバック製Dektak150)により膜厚が5.0~5.5μmとなるように調整した。評価用基板の500nmにおける面内位相差R0の値を大塚電子社製 RETS-100にて測定した。
実施例27及び28で得られたコーティング組成物をガラス基板上に、触針式形状測定器(アルバック製Dektak150)により膜厚が5.0~5.5μmとなるように条件を調整したスピンコーターを用いて塗布した後、90℃のホットプレートで10分間のプリベークを行なった。その後、室温まで冷却し、フォトマスク(Line/Space=50μm/50μm)を介し、高圧水銀ランプを用い、365nmの波長を含む光を500mJ/cm2照射し、23℃のイオン交換水に1分間浸漬した後、エアーガンで付着した水を除去し、基板を140℃のオーブン内で30分間乾燥させた。
乾燥後のパターンをレーザー顕微鏡で確認したところ、いずれのコーティング組成物で作製した基板においても、パターン状硬化物のパターン幅が50±3μm以内であり、良好なパターンが得られた。
(光導波路の作製)
焼成シリコン基板に、下記表7に示すコーティング組成物を各クラッド材料としてスピンコート法により30μmの厚さで積層し、光量10mW/cm2の紫外線を200秒照射後、120℃にて15分間加熱して硬化物を形成した。次いで、形成された硬化物上に、下記表7に示すコーティング組成物を各コア材料としてスピンコート法により20μmの厚さで積層し、ネガ型のフォトマスクを使用して、光量10mW/cm2の紫外線を40秒照射した。水で現像し、140℃で30分間加熱硬化させ、線幅20μmのパターン状硬化物を形成した。更に、下記表7に示すコーティング組成物を各クラッド材料としてスピンコート法によりパターン状硬化物の上面から30μmの厚さとなるように積層し、光量10mW/cm2の紫外線を200秒間照射後、140℃にて30分間加熱することで、シリコン基板上に光導波路を作製した。
波長850nmのアイソレータ付ASE(Amplified Spontaneous Emission)光源を用い、カットバック法により測定した伝送損失から接続損失を差し引いて光伝送損失を算出した。測定結果を表7に示す。光伝送損失が小さいほど、長距離伝送に適しているといえる。
(コーティング紙の作製)
トイレットペーパー(春日製紙工業株式会社製 コアユース)に下記表8に示すコーティング組成物を含侵させて、2枚張り合わせ、90℃において1時間乾燥した。乾燥後、光量10mW/cm2の紫外線を200秒照射後、120℃にて15分間加熱し、コーティング紙を得た。5cm×5cmの大きさに裁断したコーティング紙を200mLのイオン交換水に投下し、メカニカルスターラーで5分間攪拌し、コーティング紙の耐水性を評価した。判定基準は下記の通りとした。評価結果を表8に示す。なお、表中のUV照射なしは、上記コーティング紙の作製において、光量10mW/cm2の紫外線を照射しなかったことを意味する。また、表中の-は、未評価を意味する。
〇:2枚のトイレットペーパーが接着された状態を維持していた
△:2枚のトイレットペーパーが剥離したが、形状を維持していた
×:2枚のトイレットペーパーが剥離し、更に、形状を維持できなかった
Claims (16)
- 下記(1)及び(2)からなる群から選ばれる水溶性重合体を含有するコーティング組成物。
(1)ポリビニルアルコール、その変性体又はその誘導体
(2)ポリビニルピロリドン、その変性体又はその誘導体 - ジルコニウム化合物、チタン化合物、ビスアジド化合物、ブロックイソシアネート及びエポキシ化合物からなる群から選ばれる一種以上の架橋剤をさらに含有する請求項1に記載のコーティング組成物。
- 水溶性多官能(メタ)アクリレート及び水溶性多官能(メタ)アクリルアミドからなる群から選ばれる少なくとも1種をさらに含有する請求項1又は2に記載のコーティング組成物。
- カルボキシル基及び/又はカルボキシル基の塩を置換基として有する水溶性ポリエステルをさらに含有する請求項1に記載のコーティング組成物。
- 前記水溶性重合体が、側鎖にアセト酢酸エステル基又は感光性基を有するポリビニルアルコールである請求項1~6のいずれか一項に記載のコーティング組成物。
- 上記一般式(IIIα)、(IIIβ)及び(IIIγ)におけるQ1、Q2及びQ3、並びに上記一般式(IV)におけるQ4及びQ5が、スチルバゾリウム基、シンナモイル基、ビニル基、アクリル基、メタクリル基、アクリルアミド基又はメタアクリルアミド基、シンナミル基、シンナミリデン基、シンナミリデンアセチル基、カルコン基、クマリン基、イソクマリン基、2,5-ジメトキシスチルベン基、マレイミド基、α-フェニルマレイミド基、2-ピロン基、アジド基、チミン基、キノン基、マレイミド基、ウラシル基、ピリミジン基、スチリルピリジニウム基、又はスチリルキノリウム基から選択される請求項8又は9に記載のコーティング組成物。
- 布又は紙に含浸される請求項1~10のいずれか一項に記載のコーティング組成物。
- 光学素子に使用される請求項1~10のいずれか一項に記載のコーティング組成物。
- 請求項1~12のいずれか一項に記載のコーティング組成物の硬化物。
- 請求項1~12のいずれか一項に記載のコーティング組成物を硬化させる工程を有する硬化物の製造方法。
- 請求項1~12のいずれか一項に記載のコーティング組成物のパターン状硬化物。
- 請求項1~12のいずれか一項に記載のコーティング組成物をパターン状に硬化させる工程を有するパターン状硬化物の製造方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2019525461A JPWO2018230564A1 (ja) | 2017-06-16 | 2018-06-12 | コーティング組成物 |
| US16/620,253 US20210079252A1 (en) | 2017-06-16 | 2018-06-12 | Coating composition |
| EP18816510.4A EP3640307A4 (en) | 2017-06-16 | 2018-06-12 | COATING COMPOSITION |
| CN201880039373.6A CN110741053A (zh) | 2017-06-16 | 2018-06-12 | 涂料组合物 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017118438 | 2017-06-16 | ||
| JP2017-118438 | 2017-06-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018230564A1 true WO2018230564A1 (ja) | 2018-12-20 |
Family
ID=64659592
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2018/022436 WO2018230564A1 (ja) | 2017-06-16 | 2018-06-12 | コーティング組成物 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20210079252A1 (ja) |
| EP (1) | EP3640307A4 (ja) |
| JP (1) | JPWO2018230564A1 (ja) |
| CN (1) | CN110741053A (ja) |
| TW (1) | TW201920522A (ja) |
| WO (1) | WO2018230564A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022270460A1 (ja) * | 2021-06-21 | 2022-12-29 | 積水化学工業株式会社 | (メタ)アクリル樹脂組成物、無機微粒子分散スラリー組成物及び無機微粒子分散成形物 |
| EP4059968A4 (en) * | 2019-11-14 | 2023-12-27 | Adeka Corporation | POLYMERIZABLE COMPOSITION, VEHICLE AND HARDENED ARTICLE AND PRODUCTION METHOD THEREOF |
Citations (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS57197289A (en) | 1981-04-09 | 1982-12-03 | Basf Ag | Acylphosphine compound and use |
| JPS62240318A (ja) | 1986-04-10 | 1987-10-21 | Nippon Synthetic Chem Ind Co Ltd:The | カルボキシル基を有するポリエステル樹脂の製法 |
| JPS6429836A (en) * | 1987-06-13 | 1989-01-31 | Basf Ag | Heating type keep plate cross linkable by photopolymerization |
| JPH02118575A (ja) * | 1987-10-17 | 1990-05-02 | Autotype Internatl Ltd | 光二量化性重合体組成物、その製造方法およびスクリーン印刷用フォトステンシル |
| JPH04240644A (ja) * | 1991-01-23 | 1992-08-27 | Tokyo Ohka Kogyo Co Ltd | ネガ型感光性樹脂 |
| JPH06228218A (ja) | 1985-04-03 | 1994-08-16 | Ciba Geigy Ag | 不飽和系の光重合用光開始剤 |
| JPH06239910A (ja) | 1993-02-22 | 1994-08-30 | Nippon Paint Co Ltd | 光重合開始剤 |
| JPH11258797A (ja) * | 1998-03-12 | 1999-09-24 | Goo Chem Ind Co Ltd | 感光性樹脂組成物及びプリント配線板製造用フォトレジストインク |
| JP2000282267A (ja) * | 1999-03-29 | 2000-10-10 | Nippon Paint Co Ltd | アルミニウム合金の防錆・親水化処理剤およびこれを用いた防錆・親水化処理方法 |
| JP3295153B2 (ja) | 1992-12-02 | 2002-06-24 | 日本合成化学工業株式会社 | 紙コーティング方法 |
| JP2002188076A (ja) * | 2000-12-21 | 2002-07-05 | Nippon Shokubai Co Ltd | 親水化処理用組成物及びそれを用いてなる部材 |
| JP2003192712A (ja) | 2001-12-28 | 2003-07-09 | Toagosei Co Ltd | 光重合開始剤 |
| JP2007025723A (ja) * | 2006-10-19 | 2007-02-01 | Kunihiro Ichimura | 感活性エネルギー線樹脂組成物、感活性エネルギー線樹脂フィルム及び該フィルムを用いるパターン形成方法 |
| JP2008050607A (ja) | 2006-08-25 | 2008-03-06 | Dongjin Semichem Co Ltd | 帯電防止用コーティング組成物、それを利用した帯電防止コーティング膜の製造方法及びそのコーティング膜 |
| JP2009102455A (ja) | 2007-10-19 | 2009-05-14 | Canon Inc | 活性エネルギー線重合開始剤、該開始剤を含む硬化型液体組成物、インク、インクジェット記録方法、記録画像、インクカートリッジ、記録ユニット及びインクジェット記録装置 |
| JP2009186776A (ja) | 2008-02-06 | 2009-08-20 | Sumitomo Osaka Cement Co Ltd | ハードコート膜およびその製造方法並びにハードコート膜を備えた光学機能膜および光学レンズ |
| JP2012007071A (ja) | 2010-06-24 | 2012-01-12 | Fujifilm Corp | インク組成物、インクセット及び画像形成方法。 |
| WO2014050551A1 (ja) | 2012-09-28 | 2014-04-03 | 富士フイルム株式会社 | 硬化性樹脂組成物、水溶性インク組成物、インクセットおよび画像形成方法 |
| JP2016510314A (ja) | 2012-12-19 | 2016-04-07 | ビーエーエスエフ ソシエタス・ヨーロピアBasf Se | ビスアシルホスフィン酸の誘導体、その製造および光開始剤としての使用 |
| JP2016520146A (ja) | 2013-05-21 | 2016-07-11 | サン ケミカル コーポレイション | アセタール化したポリビニルアルコールのバリアコーティング材 |
| WO2016136752A1 (ja) * | 2015-02-26 | 2016-09-01 | 株式会社Adeka | パターン形成方法およびこれを用いて製造した電子デバイス |
| JP2016185929A (ja) | 2015-03-27 | 2016-10-27 | 富士フイルム株式会社 | 光重合開始剤、及びそれを用いた、インク組成物、インクセット及び画像形成方法 |
| JP2016193985A (ja) * | 2015-03-31 | 2016-11-17 | 株式会社Adeka | 重合体及び光硬化性組成物 |
| WO2018097147A1 (ja) * | 2016-11-25 | 2018-05-31 | 株式会社Adeka | 硬化性組成物、その硬化物およびその硬化方法 |
| WO2018117138A1 (ja) * | 2016-12-22 | 2018-06-28 | 株式会社Adeka | 水溶性組成物、パターン形成剤、これらを用いた硬化物の製造方法および硬化物 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6178553B2 (ja) * | 2012-08-29 | 2017-08-09 | 株式会社日本触媒 | 光学フィルムおよびその利用 |
| JP7195039B2 (ja) * | 2015-10-16 | 2022-12-23 | 株式会社Adeka | 樹脂組成物およびこれを用いた光学フィルム |
| TWI746467B (zh) * | 2015-10-16 | 2021-11-21 | 日商Adeka股份有限公司 | 樹脂組成物以及使用其之光學薄膜 |
-
2018
- 2018-06-12 WO PCT/JP2018/022436 patent/WO2018230564A1/ja active Application Filing
- 2018-06-12 JP JP2019525461A patent/JPWO2018230564A1/ja active Pending
- 2018-06-12 CN CN201880039373.6A patent/CN110741053A/zh active Pending
- 2018-06-12 US US16/620,253 patent/US20210079252A1/en not_active Abandoned
- 2018-06-12 EP EP18816510.4A patent/EP3640307A4/en not_active Withdrawn
- 2018-06-15 TW TW107120624A patent/TW201920522A/zh unknown
Patent Citations (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS57197289A (en) | 1981-04-09 | 1982-12-03 | Basf Ag | Acylphosphine compound and use |
| JPH06228218A (ja) | 1985-04-03 | 1994-08-16 | Ciba Geigy Ag | 不飽和系の光重合用光開始剤 |
| JPS62240318A (ja) | 1986-04-10 | 1987-10-21 | Nippon Synthetic Chem Ind Co Ltd:The | カルボキシル基を有するポリエステル樹脂の製法 |
| JPS6429836A (en) * | 1987-06-13 | 1989-01-31 | Basf Ag | Heating type keep plate cross linkable by photopolymerization |
| JPH02118575A (ja) * | 1987-10-17 | 1990-05-02 | Autotype Internatl Ltd | 光二量化性重合体組成物、その製造方法およびスクリーン印刷用フォトステンシル |
| JPH04240644A (ja) * | 1991-01-23 | 1992-08-27 | Tokyo Ohka Kogyo Co Ltd | ネガ型感光性樹脂 |
| JP3295153B2 (ja) | 1992-12-02 | 2002-06-24 | 日本合成化学工業株式会社 | 紙コーティング方法 |
| JPH06239910A (ja) | 1993-02-22 | 1994-08-30 | Nippon Paint Co Ltd | 光重合開始剤 |
| JPH11258797A (ja) * | 1998-03-12 | 1999-09-24 | Goo Chem Ind Co Ltd | 感光性樹脂組成物及びプリント配線板製造用フォトレジストインク |
| JP2000282267A (ja) * | 1999-03-29 | 2000-10-10 | Nippon Paint Co Ltd | アルミニウム合金の防錆・親水化処理剤およびこれを用いた防錆・親水化処理方法 |
| JP2002188076A (ja) * | 2000-12-21 | 2002-07-05 | Nippon Shokubai Co Ltd | 親水化処理用組成物及びそれを用いてなる部材 |
| JP2003192712A (ja) | 2001-12-28 | 2003-07-09 | Toagosei Co Ltd | 光重合開始剤 |
| JP2008050607A (ja) | 2006-08-25 | 2008-03-06 | Dongjin Semichem Co Ltd | 帯電防止用コーティング組成物、それを利用した帯電防止コーティング膜の製造方法及びそのコーティング膜 |
| JP2007025723A (ja) * | 2006-10-19 | 2007-02-01 | Kunihiro Ichimura | 感活性エネルギー線樹脂組成物、感活性エネルギー線樹脂フィルム及び該フィルムを用いるパターン形成方法 |
| JP2009102455A (ja) | 2007-10-19 | 2009-05-14 | Canon Inc | 活性エネルギー線重合開始剤、該開始剤を含む硬化型液体組成物、インク、インクジェット記録方法、記録画像、インクカートリッジ、記録ユニット及びインクジェット記録装置 |
| JP2009186776A (ja) | 2008-02-06 | 2009-08-20 | Sumitomo Osaka Cement Co Ltd | ハードコート膜およびその製造方法並びにハードコート膜を備えた光学機能膜および光学レンズ |
| JP2012007071A (ja) | 2010-06-24 | 2012-01-12 | Fujifilm Corp | インク組成物、インクセット及び画像形成方法。 |
| WO2014050551A1 (ja) | 2012-09-28 | 2014-04-03 | 富士フイルム株式会社 | 硬化性樹脂組成物、水溶性インク組成物、インクセットおよび画像形成方法 |
| JP2016510314A (ja) | 2012-12-19 | 2016-04-07 | ビーエーエスエフ ソシエタス・ヨーロピアBasf Se | ビスアシルホスフィン酸の誘導体、その製造および光開始剤としての使用 |
| JP2016520146A (ja) | 2013-05-21 | 2016-07-11 | サン ケミカル コーポレイション | アセタール化したポリビニルアルコールのバリアコーティング材 |
| WO2016136752A1 (ja) * | 2015-02-26 | 2016-09-01 | 株式会社Adeka | パターン形成方法およびこれを用いて製造した電子デバイス |
| JP2016185929A (ja) | 2015-03-27 | 2016-10-27 | 富士フイルム株式会社 | 光重合開始剤、及びそれを用いた、インク組成物、インクセット及び画像形成方法 |
| JP2016193985A (ja) * | 2015-03-31 | 2016-11-17 | 株式会社Adeka | 重合体及び光硬化性組成物 |
| WO2018097147A1 (ja) * | 2016-11-25 | 2018-05-31 | 株式会社Adeka | 硬化性組成物、その硬化物およびその硬化方法 |
| WO2018117138A1 (ja) * | 2016-12-22 | 2018-06-28 | 株式会社Adeka | 水溶性組成物、パターン形成剤、これらを用いた硬化物の製造方法および硬化物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3640307A4 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4059968A4 (en) * | 2019-11-14 | 2023-12-27 | Adeka Corporation | POLYMERIZABLE COMPOSITION, VEHICLE AND HARDENED ARTICLE AND PRODUCTION METHOD THEREOF |
| WO2022270460A1 (ja) * | 2021-06-21 | 2022-12-29 | 積水化学工業株式会社 | (メタ)アクリル樹脂組成物、無機微粒子分散スラリー組成物及び無機微粒子分散成形物 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20210079252A1 (en) | 2021-03-18 |
| EP3640307A1 (en) | 2020-04-22 |
| EP3640307A4 (en) | 2021-02-24 |
| JPWO2018230564A1 (ja) | 2020-04-30 |
| TW201920522A (zh) | 2019-06-01 |
| CN110741053A (zh) | 2020-01-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4801419B2 (ja) | 偏光板 | |
| KR101903888B1 (ko) | 광학 적층체 및 이를 포함하는 플렉서블 디스플레이 | |
| US9857505B2 (en) | Laminate body and manufacturing method thereof | |
| KR101462579B1 (ko) | 편광판 및 이를 포함하는 액정표시장치 | |
| TWI394660B (zh) | 薄膜積層體及其製造方法 | |
| KR102570353B1 (ko) | 수지 조성물 및 이것을 사용한 광학 필름 | |
| JP2008037101A (ja) | フィルム積層体及びその製造方法 | |
| JPWO2019004431A1 (ja) | 接着剤組成物 | |
| WO2018117138A1 (ja) | 水溶性組成物、パターン形成剤、これらを用いた硬化物の製造方法および硬化物 | |
| WO2018230564A1 (ja) | コーティング組成物 | |
| JP6934865B2 (ja) | 水溶性組成物およびこれからなる硬化物 | |
| TWI751228B (zh) | 硬化性組成物、其硬化物及其硬化方法 | |
| WO2017065259A1 (ja) | 樹脂組成物およびこれを用いた光学フィルム | |
| JPWO2018097147A6 (ja) | 硬化性組成物、その硬化物およびその硬化方法 | |
| EP2816085B1 (en) | Optical film using a polyester-based primer composition and polarizing plate comprising the polyester-based primer composition | |
| JP2019001927A (ja) | インク組成物 | |
| JP2020084156A (ja) | 組成物、これを含有するバリア膜形成用組成物、これらの硬化物、それからなるガスバリア膜および硬化物の製造方法 | |
| JP5835055B2 (ja) | プラスチック製フィルム又はシート用活性エネルギー線硬化型接着剤組成物 | |
| JP2011153289A (ja) | 易接着性熱可塑性樹脂フィルム |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18816510 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2019525461 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2018816510 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2018816510 Country of ref document: EP Effective date: 20200116 |