WO2017150519A1 - 水性化粧料組成物 - Google Patents
水性化粧料組成物 Download PDFInfo
- Publication number
- WO2017150519A1 WO2017150519A1 PCT/JP2017/007750 JP2017007750W WO2017150519A1 WO 2017150519 A1 WO2017150519 A1 WO 2017150519A1 JP 2017007750 W JP2017007750 W JP 2017007750W WO 2017150519 A1 WO2017150519 A1 WO 2017150519A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- powder
- cosmetic composition
- oil
- mass
- hydrophilic
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/342—Alcohols having more than seven atoms in an unbroken chain
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/0216—Solid or semisolid forms
- A61K8/022—Powders; Compacted Powders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/0241—Containing particulates characterized by their shape and/or structure
- A61K8/025—Explicitly spheroidal or spherical shape
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/04—Dispersions; Emulsions
- A61K8/06—Emulsions
- A61K8/062—Oil-in-water emulsions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/86—Polyethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/89—Polysiloxanes
- A61K8/891—Polysiloxanes saturated, e.g. dimethicone, phenyl trimethicone, C24-C28 methicone or stearyl dimethicone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q1/00—Make-up preparations; Body powders; Preparations for removing make-up
- A61Q1/02—Preparations containing skin colorants, e.g. pigments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/41—Particular ingredients further characterized by their size
- A61K2800/412—Microsized, i.e. having sizes between 0.1 and 100 microns
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/54—Polymers characterized by specific structures/properties
- A61K2800/542—Polymers characterized by specific structures/properties characterized by the charge
- A61K2800/5422—Polymers characterized by specific structures/properties characterized by the charge nonionic
Definitions
- “aesthetic role” that makes the appearance beautiful.
- “aesthetic role” in cosmetics applied to the skin such as creams and foundations includes adjusting the skin tone and correcting irregularities such as pores.
- a component having a high refractive index for correcting the skin tone and a powder component that makes the unevenness of the skin less noticeable due to the light diffusion effect have been blended.
- the conventional cosmetics having the effect of correcting unevenness are often oily cosmetics or water-in-oil emulsified cosmetics, in which case the oil is the first to touch the skin when applied to the skin. Since it was a phase, it was undeniable that the oiliness was felt relatively strongly. In addition, since the amount of oil was large, stickiness was sometimes felt after application.
- Patent Document 3 describes an oil-in-water emulsified cosmetic material that contains ⁇ -monoacryl glyceryl ether, wax, silicone oil, and silicone resin in a specific ratio and that is blended with a hydrophobic treatment powder and has excellent water resistance.
- this cosmetic since the hydrophobized powder is blended in the oil phase, it was necessary to increase the amount of oil and surfactant in order to increase the hydrophobic powder in order to improve the skin correction effect. .
- the present invention (A) one or more of higher alcohols having a linear saturated alkyl chain; (B) a hydrophilic surfactant containing a nonionic surfactant; (C) 4-30% by mass of powder having an average particle size of 1-30 ⁇ m; and (D) containing water,
- the ratio of the number of moles of hydrophilic nonionic surfactant to the total number of moles of (B) hydrophilic surfactant is 0.5 or more, and (C) the total of powders having an average particle size of 1 to 30 ⁇ m
- a cosmetic composition characterized in that the proportion of the amount of the hydrophobic spherical powder occupying in the amount is 50% by mass, and the hydrophobic spherical powder is added to the aqueous phase.
- the cosmetic composition of the present invention has, as essential components, (A) one or more of higher alcohols having a linear saturated alkyl chain, (B) a hydrophilic surfactant, and (C) an average particle size of 1. 4-30% by mass of powder of ⁇ 30 ⁇ m and (D) water.
- A one or more of higher alcohols having a linear saturated alkyl chain
- B a hydrophilic surfactant
- C an average particle size of 1. 4-30% by mass of powder of ⁇ 30 ⁇ m and (D) water.
- the component (A) in the present invention is one or more of higher alcohols having a linear saturated alkyl chain.
- the higher alcohol having a linear saturated alkyl chain is an alcohol having a linear saturated alkyl group having 12 or more carbon atoms, preferably 12 to 22 carbon atoms.
- component (B) hydrophilic surfactant in the present invention comprises a nonionic surfactant as an essential component and may optionally contain an ionic surfactant.
- an HLB value is required to be 10 or more, and a nonionic surfactant having an HLB value of 11 or more, 12 or more, 13 or more, or 14 or more is preferably used. It is done.
- hydrophilic nonionic surfactants it is preferable to use an ether of a linear saturated alkyl chain (preferably having 12 to 22 carbon atoms) and a polyalkylene glycol (polyethylene oxide, polypropylene oxide, polybutylene oxide, etc.).
- the ionic active agent include fatty acids having 12 to 22 carbon atoms such as palmitic acid, stearic acid, and isostearic acid and salts thereof, acyl sarcosine having 12 to 22 carbon atoms such as sodium lauroyl sarcosine, and salts thereof, and the like.
- Alkyl phosphoric acid having 12 to 22 carbon atoms such as sodium cetyl phosphate or a salt thereof, acylmethyl taurine having 12 to 22 carbon atoms such as stearoylmethyl taurine sodium and its salt, and acyl lactic acid having 12 to 22 carbon atoms such as sodium stearoyl lactate And its salts, acyl glutamic acid having 12 to 22 carbon atoms such as sodium stearoyl glutamate and its salts, distearyldimethylammonium chloride, alkyltrimethylammonium chloride, benzalkonium chloride, alkyldimethylaminoacetic acid betaine, etc. .
- (B) the total number of moles of hydrophilic surfactant (that is, the number of moles of hydrophilic nonionic surfactant (B1) and the number of moles of ionic surfactant (B2) It is essential that the ratio [X B1 / (B1 + B2)] of the number of moles (B1) of the hydrophilic nonionic surfactant to the sum of ()) is 0.5 or more.
- This molar ratio (X) is preferably 0.65 or more, more preferably 0.8 or more, and (B) a composition in which the hydrophilic surfactant consists only of a hydrophilic nonionic surfactant (ie, moles).
- the ratio (X) 1) may be used. When the molar ratio is less than 0.5, the stability of the composition is lowered, and the uniformity of the appearance when applied to the skin is also lowered.
- the blending amount of (B) hydrophilic surfactant (total blending amount of nonionic surfactant and ionic surfactant) in the cosmetic composition of the present invention is not particularly limited, but is usually 0.05. To 6% by mass, preferably 0.1 to 5% by mass.
- the component (C) in the cosmetic composition of the present invention is a powder having an average particle diameter of 1 to 30 ⁇ m.
- This average particle diameter can take all values within the range of 1 to 30 ⁇ m.
- the lower limit of the average particle size range can be 1 ⁇ m, 1.5 ⁇ m, 2 ⁇ m, and the upper limit can be 30 ⁇ m, 25 ⁇ m, 20 ⁇ m, 18 ⁇ m, 15 ⁇ m, and the like.
- the “average particle diameter” in this specification means an average value of particle diameters obtained by laser analysis.
- (C) the total amount of powder having an average particle diameter of 1 to 30 ⁇ m (the blending amount of hydrophobic spherical powder (C1) and the blending amount of powder other than hydrophobic spherical powder (C2))
- the surface tension of the acetone aqueous solution is the critical surface tension of the powder. It becomes an index.
- the hydrophobic powder in the present invention is a surface hydrophobic powder having an approximate value of critical surface tension determined as described above of about 60 or less. More preferably, the powder has a value of about 55 or less.
- Examples of powders having an average particle diameter of 1 to 30 ⁇ m that can constitute the component (C) and do not correspond to the hydrophobic spherical powder include silica, starch, cellulose, etc. that are easily dispersed in water (surface hydrophilic) The powder which consists of is mentioned.
- the cosmetic composition of the present invention is also referred to as (E) a lipophilic nonionic surfactant (hereinafter referred to as “lipophilic nonionic surfactant”) in addition to the essential components (A) to (D). Can be further included.
- lipophilic nonionic surfactant hereinafter referred to as “lipophilic nonionic surfactant”
- the lipophilic nonionic surfactant used in the present invention is a nonionic surfactant that is widely used in cosmetics and the like and has an HLB value of 6 or less, preferably 5 or less. Can do. Specific examples include glyceryl monostearate, glyceryl monostearyl ether, PEG-10 glyceryl triisostearate, PEG-15 hydrogenated castor oil triisostearate, PEG-10 glyceryl trioleate, PEG-10 glyceryl tristearate, Examples include, but are not limited to, PEG-10 tristearate, trimethylolpropane tristearate, PEG-5 hydrogenated castor oil, PEG-4 trimethylolpropane distearate, diisopropyl sebacate, and the like.
- a higher alcohol is formed from a lamellar bilayer together with (B) a hydrophilic surfactant and preferably (E) a lipophilic surfactant.
- B a hydrophilic surfactant
- E a lipophilic surfactant.
- ⁇ -Gel can be obtained by dissolving the surfactant and higher alcohol at high temperature and mixing with water or cooling, or by melting higher alcohol at high temperature and mixing with surfactant-dissolved water and cooling. It is the gel structure obtained.
- the cosmetic composition of the present invention may contain a water-soluble polymer such as guar gum, xanthan gum or polyvinyl alcohol.
- a water-soluble polymer such as guar gum, xanthan gum or polyvinyl alcohol.
- the composition can be further stabilized by blending a water-soluble polymer, but since the stickiness may occur when the blending amount increases, the blending amount is preferably less than 0.1% by mass. .
- the cosmetic composition of the present invention is an aqueous composition that does not contain oil or contains 30% by mass or less, 25% by mass or less, 20% by mass or less, or 15% by mass or less of oil.
- the cosmetic composition of the present invention contains an oil
- the oil can be selected from the oils usually used in cosmetics.
- Linseed oil camellia oil, macadamia nut oil, corn oil, mink oil, olive oil, avocado oil, sasanqua oil, castor oil, safflower oil, jojoba oil, sunflower oil, almond oil, rapeseed oil, sesame oil, soybean oil, peanut oil, Liquid oils such as triglycerin, glycerin trioctanoate, glycerin triisopalmitate.
- silica anhydrous silicic acid
- calcium silicate magnesium silicate
- mica talc
- kaolin kaolin
- sericite titanium oxide
- zinc oxide magnesium oxide
- zirconium oxide calcium carbonate
- magnesium carbonate magnesium silicate
- barium sulfate bengara
- Yellow iron oxide black iron oxide, carbon black, manganese violet, glass beads
- zeolite pearl pigment
- pearl pigment Bengara-coated mica, titanium oxide-coated mica, etc.
- vitamin B group vitamin C and derivatives thereof, tranexamic acid, pantothenic acid and derivatives thereof, water-soluble active substances such as vitamins such as biotin, oil-soluble active substances such as vitamin E and ⁇ -carotene, arginine and aspartic acid
- buffering agents such as citric acid, tartaric acid and lactic acid, chelating agents such as EDTA, preservatives, and various dyes.
- the cosmetic composition of the present invention is particularly suitable for use as an aqueous basic cosmetic having a fresh feel and excellent correction effect, for example, a makeup cosmetic such as a foundation or a makeup base, or a skin care cosmetic such as an emulsion or a cosmetic liquid. It is suitable.
- the present invention will be described in more detail with reference to specific examples.
- the examples are representative examples of the present invention and do not limit the technical scope of the present invention.
- the unit of a compounding quantity is the mass%.
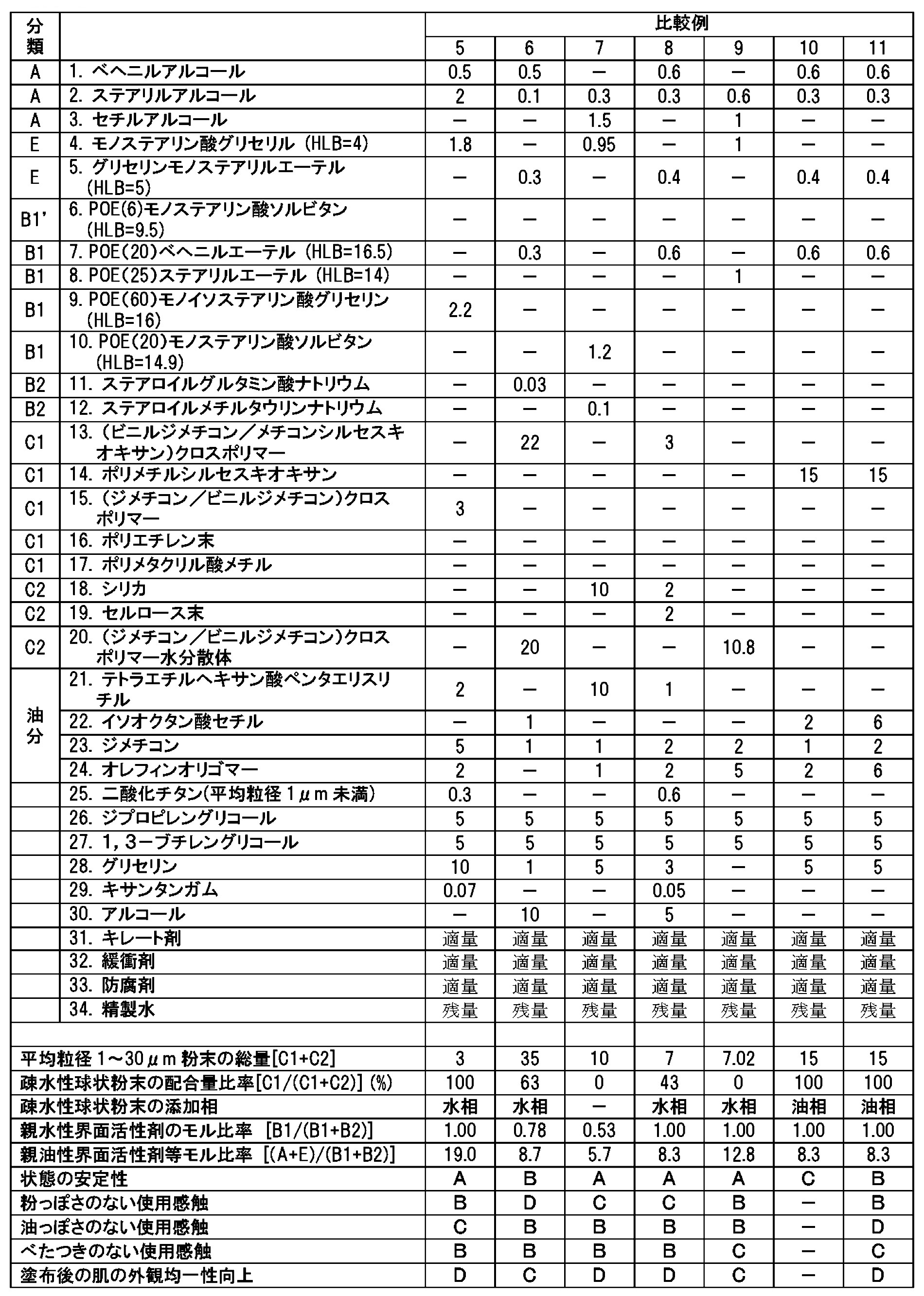
- Comparative Example 1 was an example in which the powder was dispersed only with a surfactant without using a gel formed of a higher alcohol and a surfactant, and the use feeling was sticky. Also, because there is no thickening effect due to the gel formed with higher alcohol and activator, a thickener was added to maintain a uniform dispersion state of the powder, but it was combined with the powder when applied to the skin. The body was formed and the powder did not spread uniformly, and there was no effect of improving the appearance uniformity of the skin.
- Comparative Example 2 only the ionic one was used as the hydrophilic activator, and in Comparative Example 3, the ionic one was used in the molar concentration of the hydrophilic activator more than the nonionic one. A sufficient thickening effect was not obtained, and the dispersion state of the powder was slightly inferior, so that the skin appearance uniformity improving effect was low.
- Comparative Example 7 the powder is only hydrophilic, and in Comparative Example 8, the proportion of the hydrophobic spherical powder in the powder is 43%.
- the familiarity of the powder with the skin was poor, and the effect of improving the appearance of the skin was poor.
- Comparative Example 9 the raw material provided as an aqueous dispersion of hydrophobic spherical powder was used, but the raw material contains an activator and a thickener for stably dispersing the powder. Similarly, stickiness was observed. Further, the powder surface was substantially hydrophilic, and the effect of improving the skin appearance uniformity was low as in Comparative Examples 7 and 8.
- Comparative Examples 10 and 11 were produced by dispersing hydrophobic spherical powder in the oil phase, but the amount of oil in Comparative Example 10 did not satisfy a sufficient amount to disperse the powder, resulting in poor dispersion. It was inferior in the state immediately after manufacture. In Comparative Example 11, when a sufficient amount of oil was used to disperse the powder, an oily feel was obtained, and the adhesion of the powder on the skin was poor.
- Formulation Example 1 Cream amount (% by mass) 1 Behenyl alcohol 2 2 Stearyl alcohol 2 3 Glycerin monostearyl ether 2 4 POE (20) behenyl ether 1.5 5 (Vinyl dimethicone / methicone silsesquioxane) cross polymer 10 6 Silica 2 7 Pentaerythrityl tetraethylhexanoate 5 8 Dimethicone 3 9 Olefin oligomer 2 10 Titanium dioxide 0.1 11 1,3-butylene glycol 10 12 Glycerin 10 13 Ascorbic acid glucoside 2 14 (Acrylic acid / alkyl acrylate (C10-30)) copolymer (* 16) 0.05 15 Xanthan gum 0.1 16 Alcohol 3 17 Chelating agent Appropriate amount 18 Buffering agent Appropriate amount 19 Preservative Appropriate amount 20 Purified water remaining amount * 16) PEMULEN TR
- Manufacturing method Heat the aqueous phase in which 10 to 20 are uniformly mixed and dissolved to 70 ° C, add the oil phase in which 1 to 4 and 7 to 9 are heated and dissolved at 75 ° C, and then add 5, 6 to be uniform Dispersed and cooled to 30 ° C. with stirring to obtain a cream.
- Formulation Example 2 Essence amount (mass%) 1 cetearyl alcohol 0.4 2 Glycerin monostearyl ether 0.3 3 POE (60) hydrogenated castor oil (* 17) 0.5 4 (Vinyl dimethicone / methicone silsesquioxane) cross polymer (* 18) 15 5 (Dimethicone / Vinyl Dimethicone) Cross-polymer aqueous dispersion 20 6 Isohexadecane 10 7 Hydrogenated jojoba oil 2 8 Dipropylene glycol 5 9 Glycerin 5 10 (Acryloyldimethyltauronium ammonium / Beheneth-25 methacrylate) Crosspolymer (* 19) 0.3 11 Chelating agent Appropriate amount 12 Buffering agent Appropriate amount 13 Preservative Appropriate amount 14 Purified water remaining * 17) Nikkor HCO-60 (Nikko Chemicals) * 18) KSP102 (Shin-Etsu Chemical)
- aqueous phase mixed with 8 to 14 is heated to 70 ° C., 1 to 3 and 7 are heated and dissolved at 70 ° C., and 6 is added to form an oil phase, which is then added to the aqueous phase using a homomixer. Emulsified and dispersed. After cooling to 35 ° C., 4 and 5 were added and dispersed with a homomixer to obtain a cosmetic liquid.
- Production method 17 to 25 were mixed to form an aqueous phase and heated to 70 ° C. 1 to 4 and 9 to 11 heated to 70 ° C. were used as an oil phase (1), added to the aqueous phase, emulsified with a homomixer, and then cooled to 30 ° C.
- the oil phase (2) dispersed by adding 12-16 to 7-8 was added thereto, emulsified and dispersed again using a homomixer, and dispersed again by adding 5 and 6 to obtain a foundation.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Chemical & Material Sciences (AREA)
- Emergency Medicine (AREA)
- Dermatology (AREA)
- Dispersion Chemistry (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Cosmetics (AREA)
Abstract
Description
(A)直鎖飽和アルキル鎖を有する高級アルコールの1種または2種以上;
(B)非イオン性界面活性剤を含む親水性界面活性剤;
(C)平均粒径が1~30μmの粉末を4~30質量%;及び
(D)水を含有し、
前記(B)親水性界面活性剤の総モル数に対する親水性非イオン性界面活性剤のモル数の比率が0.5以上であり、前記(C)平均粒径が1~30μmの粉末の合計配合量に占める疎水性球状粉末の配合量の比率が50質量%であり、かつ前記疎水性球状粉末が水相に添加されてなることを特徴とする化粧料組成物を提供する。
以下に各成分について詳細に説明する。
本発明における(A)成分は、直鎖飽和アルキル鎖を有する高級アルコールの1種または2種以上である。直鎖飽和アルキル鎖を有する高級アルコールは、炭素数12以上、好ましくは炭素数12~22の直鎖飽和アルキル基を有するアルコールである。
本発明における成分(B)親水性界面活性剤は、非イオン性界面活性剤を必須成分とし、任意にイオン性界面活性剤を含んでもよいものである。
本発明の化粧料組成物における(C)成分は、平均粒径が1~30μmの粉末である。この平均粒径は、1~30μmの範囲内の全ての値を採ることができる。例えば、平均粒径範囲の下限値は1μm、1.5μm、あるいは2μm等とすることができ、上限値は30μm、25μm、20μm、18μm、あるいは15μm等とすることができる。
なお、本明細書における「平均粒径」とは、レーザー解析によって求めた粒径の平均値を意味する。
流動パラフィン、スクワラン、スクワレン、パラフィン、イソパラフィン、セレシン等の炭化水素油。
ジメチルポリシロキサン、メチルフェニルポリシロキサン、メチルハイドロジェンポリシロキサン等の鎖状シリコーン;オクタメチルシクロテトラシロキサン、デカメチルシクロペンタシロキサン、ドデカメチルシクロヘキサシロキサン等の環状シリコーン等のシリコーン油。
オクタン酸セチル、ラウリン酸ヘキシル、ミリスチン酸イソプロピル、パルミチン酸オクチル、ステアリン酸イソセチル、イソステアリン酸イソプロピル、イソパルミチン酸オクチル、オレイン酸イソデシル、トリ2-エチルヘキサン酸グリセリル、テトラ2-エチルヘキサン酸ペンタエリスリット、コハク酸2-エチルヘキシル、セバシン酸ジエチル等のエステル油。
得られた各例の美容液について、組成物の状態の安定性、使用感触(粉っぽさ、油っぽさ、べたつきに関する)、及び肌に塗布した後に外観を下記に示す基準により評価した。
(1)実施例1、3、4、6、7、比較例1、3、6、7
成分11~12、26~34を溶解・混合し70℃に加熱したものに、成分1~10、21~24を70℃に加熱し均一に溶解・混合したものを添加したのち、攪拌機(T.K.ロボミックス、特殊機化工業製)を用いて7000rpmで乳化した。そこへ成分13~20を添加して再度攪拌機を用いて分散し、35℃まで冷却して美容液を得た。
成分11~12、26~30、成分32、33、成分34のうち10重量%を除いた分を溶解・混合し70℃に加熱したものに、成分1~10、21~24を70℃に加熱し均一に溶解・混合したものを添加したのち、攪拌機を用いて7000rpmで乳化した。そこへ、成分25、31、34の10重量%を混合し攪拌機で分散したものを添加し混合した。さらに成分13~20を添加して再度攪拌機を用いて分散し、35℃まで冷却して美容液を得た。
成分11~12、26~34を溶解・混合し70℃に加熱したものに、成分1~10、21~24を70℃に加熱し均一に溶解・混合したものに成分14を添加して攪拌機で分散したしたものを添加して、攪拌機を用いて7000rpmで乳化し、35℃まで冷却して美容液を得た。
・状態の安定性
上記の製造方法に従って調製したサンプルの1日後(静置)の状態;1日後のサンプルを50ccのガラス管に半量入れ、45回転/分で倒立と正置方向に4時間回転した後の状態;及び1か月保管後の状態を、スライドガラスに各サンプルを適量滴下しカバーガラスをかけた状態で目視観察し、以下の基準で評価した。
A:上記3条件すべてにおいて不均一な凝集物が認められなかった。
B:製造した1日後(静置)は均一だが、他の2条件のいずれかにおいて不均一な凝集物が認められた。
C:製造した1日後(静置)、及び他の2条件の両方において不均一な凝集物が認められた。
官能評価パネル10名により、サンプルを肌に塗布した直後の感触について「粉っぽさ」「油っぽさ」「べたつき」の3項目を「ある:1点」「ややある:2点」「あまりない:3点」「ない:4点」の4段階で評価し、各項目の平均点により以下の通り判定した。
A:3.26~4点
B:2.51~3.25点
C:1.76~2.5点
D:1~1.75点
30代の女性被験者5名に各サンプルを塗布し、それぞれ外観評価パネル5名が塗布後の肌の均一性について「向上した:3点」「やや向上した:2点」「変化なし:1点」の3段階で評価した。被験者5名分の外観評価パネル5名に対する評点を平均し、各サンプルの効果を以下のように判定した。
A:2.6~3点
B:2.1~2.5点
C:1.6~2点
D:1~1.5点
比較例1は、高級アルコールと界面活性剤で形成されるゲルを用いずに、界面活性剤のみで粉末の分散を行った例であり、べたつきのある使用感触となった。また高級アルコールと活性剤で形成されるゲルによる増粘効果がないため、粉末の均一な分散状態を維持するために増粘剤を添加していたが、肌に塗布した際に粉末との複合体ができて粉末が均一に広がらず、肌の外観均一性向上効果はなかった。
比較例5では粉末量が少なく肌の外観均一性向上効果に劣り、比較例6では粉末量が多く感触が粉っぽくなったことに加え、塗布後の肌の外観でも粉っぽさが目立った。
比較例9では、疎水性球状粉末の水分散体として提供された原料を用いたが、原料には粉末を安定に分散させるための活性剤や増粘剤が含まれており、比較例3と同様にべたつきが認められた。また粉末表面が実質的には親水性となっており、比較例7、8と同様に肌の外観均一性向上効果は低くかった。
処方例1:
クリーム 配合量(質量%)
1 ベヘニルアルコール 2
2 ステアリルアルコール 2
3 グリセリンモノステアリルエーテル 2
4 POE(20)ベヘニルエーテル 1.5
5 (ビニルジメチコン/メチコンシルセスキオキサン)クロスポリマー
10
6 シリカ 2
7 テトラエチルヘキサン酸ペンタエリスリチル 5
8 ジメチコン 3
9 オレフィンオリゴマー 2
10 二酸化チタン 0.1
11 1,3-ブチレングリコール 10
12 グリセリン 10
13 アスコルビン酸グルコシド 2
14 (アクリル酸/アクリル酸アルキル(C10-30))コポリマー(※16)
0.05
15 キサンタンガム 0.1
16 アルコール 3
17 キレート剤 適量
18 緩衝剤 適量
19 防腐剤 適量
20 精製水 残量
※16)PEMULEN TR-2(Lubrizol Advanced Materials)
10~20を均一混合・溶解した水相を70℃に加熱し、1~4、7~9を75℃で加熱溶解した油相を添加したのち、5、6、を添加して均一分散し、撹拌しながら30℃に冷却してクリームを得た。
美容液 配合量(質量%)
1 セテアリルアルコール 0.4
2 グリセリンモノステアリルエーテル 0.3
3 POE(60)水添ヒマシ油(※17) 0.5
4 (ビニルジメチコン/メチコンシルセスキオキサン)クロスポリマー(※18)
15
5 (ジメチコン/ビニルジメチコン)クロスポリマー水分散体
20
6 イソヘキサデカン 10
7 水添ホホバ油 2
8 ジプロピレングリコール 5
9 グリセリン 5
10 (アクリロイルジメチルタウリンアンモニウム/メタクリル酸べへネス-25)クロスポリマー(※19)
0.3
11 キレート剤 適量
12 緩衝剤 適量
13 防腐剤 適量
14 精製水 残量
※17)ニッコール HCO-60(日光ケミカルズ)
※18)KSP102(信越化学工業)
※19)ARISTOFLEX HMB(クラリアントジャパン)
8~14を混合した水相を70℃に加熱し、1~3、7を70℃に加熱溶解したところに6を加えて油相とし、水相に添加してホモミキサーを用いて乳化分散した。その後35℃まで冷却したのち、4と5を添加してホモミキサーで分散し、美容液を得た。
ファンデーション 配合量(質量%)
1 ベヘニルアルコール 0.4
2 ステアリルアルコール 0.1
3 モノステアリン酸グリセリル 0.1
4 POE(30)ベヘニルエーテル(※20) 0.3
5 (ジメチコン/ビニルジメチコン)クロスポリマー
5
6 ポリメタクリル酸メチル 5
7 デカメチルシロキサン 6
8 メチルポリシロキサン 3
9 コハク酸ジ2-エチルヘキシル 2
10 パラメトキシケイ皮酸2-エチルヘキシル 5
11 オクトクリレン 2
12 疎水化処理(※21)二酸化チタン 5
13 疎水化処理(※21)ベンガラ 0.1
14 疎水化処理(※21)黄酸化鉄 0.5
15 疎水化処理(※21)黒酸化鉄 0.01
16 アミノエチルアミノプロピルメチルシロキサン・ジメチルシロキサン共重合体(※22)
1
17 1,3-ブチレングリコール 3
18 グリセリン 1
19 キサンタンガム 0.1
20 (アクリル酸ヒドロキシエチル/アクリロイルジメチルタウリンNa)コポリマー(※23)
0.5
21 アルコール 5
22 キレート剤 適量
23 緩衝剤 適量
24 防腐剤 適量
25 精製水 残量
※21)テトラデセン、テトラヒドロテトラメチルシクロテトラシロキサン処理
※22)KF-8004(信越化学工業)
※23)SIMULGEL NS(SEPPIC S.A.)
17~25を混合したものを水相とし、70℃に加熱した。1~4、9~11を70℃に加熱したものを油相(1)とし、水相に添加してホモミキサーで乳化したのち30℃まで冷却した。そこへ、7~8に12~16を添加して分散した油相(2)を加えて再度ホモミキサーを用いて乳化分散し、5、6を加えて再度分散してファンデーションを得た。
Claims (6)
- (A)直鎖飽和アルキル鎖を有する高級アルコールの1種または2種以上;
(B)非イオン性界面活性剤を含む親水性界面活性剤;
(C)平均粒径が1~30μmの粉末を4~30質量%;及び
(D)水を含有し、
前記(B)親水性界面活性剤の総モル数に対する親水性非イオン性界面活性剤のモル数の比率が0.5以上であり、
前記(C)平均粒径が1~30μmの粉末の合計配合量に占める疎水性球状粉末の配合量の比率が50質量%以上であり、かつ前記疎水性球状粉末が水相に添加されてなることを特徴とする化粧料組成物。 - (E)親油性非イオン性界面活性剤を更に含み、前記(B)親水性界面活性剤のモル数に対する(A)高級アルコールと(E)親油性非イオン性界面活性剤の総モル数の比率[(A+E)/B]が3~30の範囲内である、請求項1に記載の化粧料組成物。
- 前記(C)平均粒径が1~30μmの粉末の配合量が5~25質量%である、請求項1又は2に記載の化粧料組成物。
- 0.1質量%未満の水溶性高分子を更に含む、請求項1から3のいずれか一項に記載の化粧料組成物。
- 前記(B)親水性界面活性剤に含まれる非イオン活性剤が、直鎖飽和アルキル鎖とポリアルキレングリコールとのエーテルである、請求項1から4のいずれか一項に記載の化粧料組成物。
- 前記(C)平均粒径が1~30μmの粉末に含まれる疎水性球状粉末が、シリコーンゴム、シリコーンレジン、及びそれらの複合球状粉末からなる群より選ばれる1種または2種以上である、請求項1から5のいずれか一項に記載の化粧料組成物。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020187024474A KR20180117621A (ko) | 2016-02-29 | 2017-02-28 | 수성 화장료 조성물 |
| CN201780012261.7A CN108697625B (zh) | 2016-02-29 | 2017-02-28 | 水性化妆品组合物 |
| JP2018503325A JP6927956B2 (ja) | 2016-02-29 | 2017-02-28 | 水性化粧料組成物 |
| US16/080,693 US20190076342A1 (en) | 2016-02-29 | 2017-02-28 | Aqueous cosmetic composition |
| EP17759985.9A EP3424488A4 (en) | 2016-02-29 | 2017-02-28 | AQUEOUS COSMETIC COMPOSITION |
| HK18114098.2A HK1254987A1 (zh) | 2016-02-29 | 2018-11-05 | 水性化妝品組合物 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016038176 | 2016-02-29 | ||
| JP2016-038176 | 2016-02-29 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017150519A1 true WO2017150519A1 (ja) | 2017-09-08 |
Family
ID=59743024
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/007750 WO2017150519A1 (ja) | 2016-02-29 | 2017-02-28 | 水性化粧料組成物 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20190076342A1 (ja) |
| EP (1) | EP3424488A4 (ja) |
| JP (1) | JP6927956B2 (ja) |
| KR (1) | KR20180117621A (ja) |
| CN (1) | CN108697625B (ja) |
| HK (1) | HK1254987A1 (ja) |
| TW (1) | TW201733563A (ja) |
| WO (1) | WO2017150519A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111757726A (zh) * | 2018-02-28 | 2020-10-09 | 株式会社资生堂 | 水性化妆品 |
| WO2021002285A1 (ja) * | 2019-07-01 | 2021-01-07 | 株式会社 資生堂 | 水中油型乳化化粧料 |
| WO2023042688A1 (ja) * | 2021-09-16 | 2023-03-23 | 株式会社 資生堂 | 水中油型組成物 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP7387640B2 (ja) * | 2018-12-28 | 2023-11-28 | 株式会社 資生堂 | ジェル状化粧料 |
| WO2021020135A1 (ja) * | 2019-07-31 | 2021-02-04 | 三好化成株式会社 | 化粧料の製造方法、及び化粧料 |
| JP2021176826A (ja) * | 2020-05-08 | 2021-11-11 | 株式会社トキワ | 水性化粧料 |
| CN114522109B (zh) * | 2022-03-14 | 2023-04-25 | 广州科妙生物科技有限公司 | 一种美白焕颜安瓶精华微乳液及其制备方法 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006219427A (ja) * | 2005-02-10 | 2006-08-24 | Kao Corp | 皮膚化粧料 |
| JP2010189281A (ja) * | 2009-02-16 | 2010-09-02 | Shiseido Co Ltd | 水中油型乳化組成物 |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000505095A (ja) * | 1996-08-23 | 2000-04-25 | 花王株式会社 | 水系皮膚化粧料 |
| JP2000239139A (ja) * | 1999-02-16 | 2000-09-05 | Shiseido Co Ltd | 皮膚化粧料 |
| TW200637598A (en) * | 2005-01-31 | 2006-11-01 | Kao Corp | Skin cosmetics and manufacturing method thereof |
| JP5571913B2 (ja) * | 2009-07-17 | 2014-08-13 | 株式会社 資生堂 | 水中油型乳化皮膚化粧料 |
| WO2015098675A1 (ja) * | 2013-12-27 | 2015-07-02 | 株式会社コーセー | 皮膚用水中油型乳化化粧料 |
| KR20170018798A (ko) * | 2014-06-30 | 2017-02-20 | 가부시키가이샤 시세이도 | 수성 화장료 |
-
2017
- 2017-02-28 CN CN201780012261.7A patent/CN108697625B/zh active Active
- 2017-02-28 KR KR1020187024474A patent/KR20180117621A/ko unknown
- 2017-02-28 JP JP2018503325A patent/JP6927956B2/ja active Active
- 2017-02-28 EP EP17759985.9A patent/EP3424488A4/en not_active Withdrawn
- 2017-02-28 WO PCT/JP2017/007750 patent/WO2017150519A1/ja active Application Filing
- 2017-02-28 US US16/080,693 patent/US20190076342A1/en not_active Abandoned
- 2017-03-01 TW TW106106723A patent/TW201733563A/zh unknown
-
2018
- 2018-11-05 HK HK18114098.2A patent/HK1254987A1/zh unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006219427A (ja) * | 2005-02-10 | 2006-08-24 | Kao Corp | 皮膚化粧料 |
| JP2010189281A (ja) * | 2009-02-16 | 2010-09-02 | Shiseido Co Ltd | 水中油型乳化組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3424488A4 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111757726A (zh) * | 2018-02-28 | 2020-10-09 | 株式会社资生堂 | 水性化妆品 |
| WO2021002285A1 (ja) * | 2019-07-01 | 2021-01-07 | 株式会社 資生堂 | 水中油型乳化化粧料 |
| WO2023042688A1 (ja) * | 2021-09-16 | 2023-03-23 | 株式会社 資生堂 | 水中油型組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3424488A1 (en) | 2019-01-09 |
| EP3424488A4 (en) | 2019-10-30 |
| TW201733563A (zh) | 2017-10-01 |
| JP6927956B2 (ja) | 2021-09-01 |
| KR20180117621A (ko) | 2018-10-29 |
| JPWO2017150519A1 (ja) | 2018-12-20 |
| CN108697625B (zh) | 2022-06-28 |
| HK1254987A1 (zh) | 2019-08-02 |
| US20190076342A1 (en) | 2019-03-14 |
| CN108697625A (zh) | 2018-10-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6927956B2 (ja) | 水性化粧料組成物 | |
| JP6053922B2 (ja) | 水中油型化粧料 | |
| JP5352116B2 (ja) | 水中油型乳化組成物及びその製造方法 | |
| EP3357488B1 (en) | Cosmetic material | |
| WO2019188842A1 (ja) | 化粧料 | |
| JP5295440B2 (ja) | メーキャップ化粧料 | |
| JP7324217B2 (ja) | 油中水型乳化固形化粧料 | |
| TW201831165A (zh) | 油中水型乳化化妝料 | |
| JP2017501993A (ja) | 皮脂吸い上げフィラー及び多量のモノアルコールを含む組成物 | |
| JP7465055B2 (ja) | 二層状油性化粧料 | |
| JP2004269418A (ja) | 化粧料 | |
| JP2007210895A (ja) | 粉末化粧料およびその製造方法 | |
| JP6831737B2 (ja) | 水中油型化粧料 | |
| EP3626229A1 (en) | Oil-in-water emulsion composition | |
| CN110636826A (zh) | 水包油型乳化化妆品 | |
| EP4082617A1 (en) | Powdery solid cosmetic preparation | |
| CN110868990B (zh) | 彩妆化妆品 | |
| JP2004137226A (ja) | 化粧料 | |
| TW201919578A (zh) | 油中水型乳化化妝料 | |
| JP7354078B2 (ja) | 化粧料 | |
| JP2017109981A (ja) | 化粧料 | |
| WO2021221149A1 (ja) | 水中油型乳化組成物 | |
| US20230052859A1 (en) | Method for producing powdered solid cosmetic | |
| JP6968528B2 (ja) | 脂肪酸系ゲル化剤及び共ゲル化剤を含む固体化粧用組成物 | |
| WO2023032834A1 (ja) | 水中油型乳化化粧料 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 2018503325 Country of ref document: JP |
|
| ENP | Entry into the national phase |
Ref document number: 20187024474 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2017759985 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2017759985 Country of ref document: EP Effective date: 20181001 |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17759985 Country of ref document: EP Kind code of ref document: A1 |