WO2017038562A1 - 多能性幹細胞を減少させる方法、多能性幹細胞を減少させた細胞集団の製造方法 - Google Patents
多能性幹細胞を減少させる方法、多能性幹細胞を減少させた細胞集団の製造方法 Download PDFInfo
- Publication number
- WO2017038562A1 WO2017038562A1 PCT/JP2016/074545 JP2016074545W WO2017038562A1 WO 2017038562 A1 WO2017038562 A1 WO 2017038562A1 JP 2016074545 W JP2016074545 W JP 2016074545W WO 2017038562 A1 WO2017038562 A1 WO 2017038562A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cells
- cell
- pluripotent stem
- stem cells
- hours
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0657—Cardiomyocytes; Heart cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0696—Artificially induced pluripotent stem cells, e.g. iPS
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/10—Cells modified by introduction of foreign genetic material
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/998—Proteins not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2502/00—Coculture with; Conditioned medium produced by
- C12N2502/13—Coculture with; Conditioned medium produced by connective tissue cells; generic mesenchyme cells, e.g. so-called "embryonic fibroblasts"
- C12N2502/1329—Cardiomyocytes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2502/00—Coculture with; Conditioned medium produced by
- C12N2502/45—Artificially induced pluripotent stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/45—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from artificially induced pluripotent stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2523/00—Culture process characterised by temperature
Definitions
- the present invention relates to a method for reducing pluripotent stem cells.
- the present invention also relates to a method for producing a cell population in which pluripotent stem cells are reduced.
- cells autologous cells isolated from tissues or blood collected from the patient's own body, cells (autologous cells) isolated from tissue or blood collected from the body of another person, and stocking Cells are used for treatment.
- the cell type is appropriately selected according to the disease site and disease state.
- the problem with cell transplantation is immune rejection.
- MHC major histocompatibility gene complex
- autologous cells since they are originally their own cells, the MHC types match and no rejection occurs. Therefore, from the viewpoint of immune rejection, regenerative medicine using autologous cells is preferable.
- Cardiomyocytes and nerve cells are examples of such cells, and it has been difficult to use these cells for regenerative medicine.
- pluripotent stem cells such as embryonic stem cells (ES cells) and induced pluripotent stem cells (iPS cells) have been discovered, even cells that do not divide and proliferate can be differentiated from pluripotent stem cells. It was possible to induce cardiomyocytes and nerve cells, and even these cells could be supplied.
- Pluripotent stem cells such as ES cells and iPS cells have the property of being able to differentiate into almost all cells constituting a living body.
- ES cells and iPS cells that have maintained the undifferentiated ability before differentiation have a self-replicating ability, and can basically be proliferated indefinitely. Therefore, even when a large amount of cells are required, pluripotent stem cells can theoretically be expanded to the required number of cells. It was possible to secure the necessary number of cells by inducing differentiation of the pluripotent stem cells thus obtained into arbitrary somatic cells.
- ES cells and iPS cells can be proliferated indefinitely and can be induced to differentiate into arbitrary somatic cells.
- cells that have not yet differentiated are transplanted into a living body, their multi-differentiation Because of the ability, there is a risk of forming a teratomas, a kind of tumor. If there are no undifferentiated ES cells or iPS cells remaining in the somatic cell population induced to differentiate from ES cells or iPS cells, there is no problem, but ES cells and iPS cells that remain undifferentiated remain somatic cell populations.
- Non-patent Document 1 a technique for reducing undifferentiated pluripotent stem cells from a cell population induced to differentiate from pluripotent stem cells has been demanded.
- Non-patent Document 2 a method for removing or reducing undifferentiated pluripotent stem cells from a cell population subjected to differentiation induction treatment.
- Methods Non-Patent Document 3
- chemical inhibitors Non-Patent Document 4, Non-Patent Document 5
- the method using a cell sorter a large amount of antibody for recognizing an antigen on the cell surface has to be used, which entails problems such as enormous costs and a long processing time.
- the method of incorporating a suicide gene has a problem in clinical application because the safety of the cell into which the gene has been incorporated is unknown even after transplantation.
- the method using a chemical inhibitor has a subject about the efficiency and safety which remove or reduce a pluripotent stem cell.
- an object of the present invention is to provide a method for removing or reducing pluripotent stem cells and a method for producing a cell population from which pluripotent stem cells have been removed or reduced.
- pluripotent stem cells have been found to induce apoptosis and die by a very simple and inexpensive method of raising the culture temperature for a certain period of time, while being induced to differentiate from somatic cells.
- the resulting pluripotent stem cells were found to be resistant to high temperatures.
- TRPV-1 transient receptor potential vanilloid 1
- it induces apoptosis in pluripotent stem cells, as in the case of high-temperature culture. It has been found that undifferentiated pluripotent stem cells remaining upon differentiation induction can be reduced. That is, the present invention is as follows.
- [1] A method for reducing the pluripotent stem cells contained in the cell population by culturing a cell population containing pluripotent stem cells and differentiated cells derived from pluripotent stem cells at a temperature of 40.5 ° C. or higher.
- the pluripotent stem cell is an induced pluripotent stem cell and / or an embryonic stem cell.
- the differentiated cells include cardiomyocytes, cardiac myoblasts, fibroblasts, mural cells and / or vascular endothelial cells.
- [6] A cell population obtained by reducing the pluripotent stem cells obtained by the method according to any one of [1] to [5].
- Activating TRPV-1 A method comprising the steps of: [8] The method according to [7], wherein the step of activating TRPV-1 is a step of activating TRPV-1 by culturing at a temperature of 40.5 ° C. or higher. [9] The method according to [8], wherein the temperature is 40.5 ° C to 45 ° C. [10] The method according to [8] or [9], wherein the temperature is 41 ° C to 43 ° C.
- the method according to [7], wherein the step of activating TRPV-1 is a step of adding an agonist for TRPV-1.
- the agonist is one or more selected from the group consisting of capsaicin, N-oleoyldopamine (OLDA), albanyl, olbanil, AM404 (anandamide), 2-APB, NADA, PPAHV, anti-TRPV-1 antibody.
- the method according to [12] which is an agonist.
- the pluripotent stem cell is an induced pluripotent stem cell and / or an embryonic stem cell.
- the differentiated cells include cardiomyocytes, cardiac myoblasts, fibroblasts, mural cells and / or vascular endothelial cells.
- a cell population containing pluripotent stem cells and differentiated cells derived from pluripotent stem cells is cultured in a medium supplemented with an agonist for TRPV-1 to reduce the pluripotent stem cells contained in the cell population Method.
- the agonist is one or more selected from the group consisting of capsaicin, N-oleoyldopamine (OLDA), albanyl, olbanil, AM404 (anandamide), 2-APB, NADA, PPAHV, and anti-TRPV-1 antibody.
- LALA N-oleoyldopamine
- AM404 anandamide
- 2-APB 2-APB
- NADA PPAHV
- anti-TRPV-1 antibody anti-TRPV-1 antibody
- [20] A method for producing a second cell population in which the pluripotent stem cells are reduced from a first cell population containing pluripotent stem cells and differentiated cells derived from pluripotent stem cells, Activating TRPV-1 expressed in the pluripotent stem cells contained in the cell population.
- the step of activating TRPV-1 is a step of activating TRPV-1 by culturing at a temperature of 40.5 ° C. or higher.
- the temperature is 41 ° C to 43 ° C.
- the method according to [20], wherein the step of activating TRPV-1 is a step of adding an agonist for TRPV-1.
- the agonist is one or more selected from the group consisting of capsaicin, N-oleoyldopamine (OLDA), albanyl, olbanil, AM404 (anandamide), 2-APB, NADA, PPAHV, anti-TRPV-1 antibody.
- LALA N-oleoyldopamine
- AM404 anandamide
- 2-APB 2-APB
- NADA PPAHV
- anti-TRPV-1 antibody anti-TRPV-1 antibody
- an undifferentiated pluripotent stem cell obtained by inducing differentiation of a pluripotent stem cell and an undifferentiated pluripotent stem cell remaining without being induced to differentiate from a cell population.
- Differentiated pluripotent stem cells can be easily removed or reduced.
- the cell population obtained thereby has a reduced proportion of undifferentiated pluripotent stem cells, and when used as a transplant, the probability of forming a tumor is low.
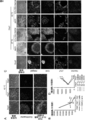
- (C) It is a figure which shows a TUNEL positive cell (green). Cell nuclei are shown in Hoechst (blue).
- the lower row shows a 36 field (6 ⁇ 6) montage image of Oct4 staining.
- A The upper row is a diagram showing a phase difference image, and the bar indicates 100 ⁇ m.
- the middle row is a diagram showing Hoechst staining (blue), and the bar represents 200 ⁇ m.
- the lower row is a diagram showing 36 fields of view (6 ⁇ 6) montage images of Hoechst-stained images.
- (B) It is a figure which shows the change of the cell number with respect to each temperature condition. The total number of cells in 36 fields is shown. It is a figure which shows the influence of 42 degreeC culture
- the upper row is a diagram showing a phase difference image, and the bar indicates 100 ⁇ m.
- the middle row is a diagram showing Oct4 (green) and Hoechst staining (blue), and the bar shows 200 ⁇ m.
- the lower row is a diagram showing an 81 view (9 ⁇ 9) montage image of an Oct4 stained image.
- C It is a figure which shows the result of the coculture experiment of an iPS cell and an iPS cell origin cardiomyocyte. The cell aggregates of iPS cells and iPS cell-derived cells were cultured at 37 ° C. or 42 ° C. in AK03 medium or DMEM containing 10% FBS two days after the start of co-culture. The top row shows a phase difference image, and the bar shows 100 ⁇ m.
- the third row is a diagram showing a fluorescent image of Oct4 (green).
- the fourth row shows a fluorescent image of cTnT (red).
- the bottom row shows cell nuclei stained with Hoechst (blue).
- the bar represents 200 ⁇ m.
- the second column is a merged image of Oct4 fluorescence image, cTnT fluorescence image, and Hoechst fluorescence image.
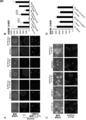
- D It is a figure which shows the change of the number of Oct4 positive cells in 36 visual fields. It is the figure which confirmed the influence of the temperature with respect to an iPS cell origin cardiomyocyte and a fibroblast.
- A It is a figure which shows a myocardial cell.
- the first column is a merged image of fluorescent images of Nkx2.5 (red), cTnT (green), and Hoechst (blue).
- the second row is a montage image of 36 views (6 ⁇ 6) of the cTnT stained image.
- the third row is an Nkx2.5 stained image.
- the 4th column name is a 36 field (6 ⁇ 6) montage image of Nkx2.5 stained image.
- the bar represents 200 ⁇ m.
- C It is a figure which shows a fibroblast.
- the first column shows a phase difference image, and the bar indicates 100 ⁇ m.
- the second row shows vimentin (green) and Hoechst staining (blue).
- the three column names are 36 fields (6 ⁇ 6) montage images of vimentin stained images.
- the names of the four columns are diagrams showing SM22 (red) and Hoechst staining (blue).
- the fifth row is a 36 field (6 ⁇ 6) montage image of the SM22 stained image.
- A It is a figure which shows the expression level of the human iPS cell origin cardiomyocyte mRNA after myocardial differentiation induction in each time of 42 degreeC culture
- cultivation (n 3).
- Y axis is compared with the expression level of GAPDH mRNA.
- Y axis is compared with the expression level of GAPDH mRNA.
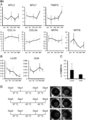
- FIG. 3 shows that culture at 42 ° C. decreases iPS cells via TRPV-1.
- the Y-axis shows the relative TRPV-1 gene expression level corrected with ⁇ -actin. * P ⁇ 0.05 vs. pre. ** p ⁇ 0.01 vs. Pre.
- D shows the results of culturing iPS cells transfected with TRPV-1 siRNA or siRNA at 37 ° C. or 42 ° C. for 1 day.
- the upper row shows the phase difference image, and the bar shows 100 ⁇ m.
- the middle row shows an image of Hoechst staining, and the bar shows 200 ⁇ m.
- the lower row is a 36 field (6 ⁇ 6) montage image of Hoechst stained image.
- A, B The results of culturing iPS cells cultured on laminin E8 fragment with the addition of capsaicin are shown. Vehicle-1 was a control in an experiment with 2 ⁇ 10 ⁇ 4 M capsaicin added, Vehicle-2 was a control in an experiment with 1 ⁇ 10 ⁇ 4 M capsaicin added, and Vehicle-3 was added with 1 ⁇ 10 ⁇ 5 M capsaicin. Represents the control in the experiment.
- the middle row shows a Hoechst-stained image, and the bar shows 200 ⁇ m.
- the lower row shows a 36 field (6 ⁇ 6) montage image of a Hoechst-stained image.
- Vehicle-1 represents a control in an experiment to which 2 ⁇ 10 ⁇ 4 M capsaicin was added
- Vehicle-2 represents a control in an experiment to which 1 ⁇ 10 ⁇ 4 M capsaicin was added.
- (C) The upper part shows a phase difference image, and the bar shows 100 ⁇ m.
- the lower row shows a merged image of Hoechst staining (blue) and Hkx2.5 staining (green), and the bar indicates 200 ⁇ m.
- (D) It is the figure which showed the cell number for 36 visual fields in each conditions. It is a figure which shows about the removal effect of the iPS cell using a TRPV-1 agonist.
- (A, B) The result of culturing iPS cells cultured on laminin E8 fragment with addition of OLDA is shown.
- (A) The upper part shows a phase difference image, and the bar shows 100 ⁇ m.
- the middle row shows Hoechst staining (blue) image, and the bar shows 200 ⁇ m.
- the lower row shows a 36 field (6 ⁇ 6) montage image of a Hoechst-stained image.
- C, D shows the results of culturing iPS cell-derived cardiomyocytes with addition of OLDA.
- C The upper row shows a merged image of cTnT staining (green), Nkx2.5 staining (red), and Hoechst staining (blue), and the bar shows 200 ⁇ m.
- the middle row shows a 36 view (6 ⁇ 6) montage image of the cTnT stained image.
- the lower row shows a 36 field (6 ⁇ 6) montage image of an Hkx2.5 stained image.
- D It is a figure which shows the number of cells (for 36 visual fields) of cTnT positive and Nkx2.5 positive cells after adding OLDA to iPS cell-derived cardiomyocytes and culturing.
- E It is a figure which shows the expression level of Lin28 mRNA after culture
- cultivating an iPS cell origin cardiomyocyte for 2 days at 42 degreeC or OLDA (5 micromol) 37 degreeC (n 3).
- the Y axis shows the relative expression level of Lin28 in iPS cell-derived cardiomyocytes relative to undifferentiated iPS cells cultured on MEF.
- FIG. 1 It is a figure which shows about the removal effect of the iPS cell using a TRPV-1 agonist.
- A, B The result of adding Albanil to iPS cells cultured on laminin E8 fragment and culturing for 1 day is shown.
- A The upper part shows a phase difference image, and the bar shows 100 ⁇ m.
- the middle row shows Hoechst staining (blue) image, and the bar shows 200 ⁇ m.
- the lower row shows a 36 field (6 ⁇ 6) montage image of a Hoechst-stained image.
- B It is the figure which showed the cell number for 36 visual fields in the conditions culture
- cultivated by adding Albanil (n 3).
- the present invention relates to a method for removing or reducing pluripotent stem cells and a method for producing a cell population from which pluripotent stem cells have been removed or reduced.
- the pluripotent stem cell is a cell having self-replicating ability and pluripotency, and means a cell having the ability to form all cells constituting the body (pluripotent).
- Self-replicating ability refers to the ability to make two undifferentiated cells from one cell.
- the pluripotent stem cells used in the present invention include embryonic stem cells (embryonic stem cells: ES cells), embryonic carcinoma cells (embrionic carcinoma cells: EC cells), trophoblast stem cells (TS cells), shrimp.
- Blast stem cell epiblast stem cell: EpiS cell
- embryonic germ cell embrionic germ cell: EG cell
- pluripotent germ cell multipotent stem cell: mGS cell
- induced pluripotent stem cell induced rupted cell
- the pluripotent stem cells used in the present invention are (1) alkaline phosphatase activity in an undifferentiated state, and / or (2) Oct3 / 4 (also referred to as Oct3, Oct4), Nanog or Sox2 which are transcription factors And / or (3) Stage Specific Embrionic Antigen (SSEA) -3, SSEA-4, Tra-1 60, Tra-1 81 or Lin28 protein (these antigens are pluripotent stem cells) And (4) cells having the ability to differentiate into three germ layers derived from endoderm, mesoderm, and ectoderm. Whether or not it has the ability to differentiate into a tissue derived from the three germ layers can be evaluated by whether or not it has the ability to transplant a target cell subcutaneously into a mouse or the like and form a teratoma (deformed species).
- the pluripotent stem cell used in the present invention is a cell that has been confirmed and stocked in advance as a cell (cell population) that satisfies the above definition, such as a public institution (for example, Kyoto University iPS Cell Laboratory, RIKEN, etc.) ) Or a cell (cell population) distributed from a cell bank or the like possessed by a private organization (iPS Academia Japan Co., Ltd.).
- a public institution for example, Kyoto University iPS Cell Laboratory, RIKEN, etc.
- a cell (cell population) distributed from a cell bank or the like possessed by a private organization iPS Academia Japan Co., Ltd.
- the pluripotency markers such as TRA-1, 81, SSEA-3, SSEA-4, and Lin28
- at least one pluripotency marker gene expression level and / or protein expression level is non-pluripotent.
- a method for measuring a pluripotent marker of pluripotent stem cells may be in accordance with a conventional method.
- RT-PCR method measurement method using a flow cytometer
- measurement method using Western blot measurement method using immunohistochemical staining
- a technique such as a microarray method can be used.
- the pluripotent stem cells used in the present invention may be differentiated non-pluripotent stem cells, for example, pluripotent stem cells obtained by reprogramming somatic cells.
- pluripotent stem cells obtained by reprogramming somatic cells.
- a method of removing a nucleus of an unfertilized egg and transplanting a somatic cell nucleus, a method of performing cell fusion between a somatic cell and an ES cell, a specific reprogramming (initialization) factor (Oct3 / 4, (Nanog, Sox2, c-Myc, L-Myc, Lin28, Klf4, etc.) are introduced and initialized, but are not limited thereto.
- the method for introducing the reprogramming factor into the cell is not particularly limited, and a method using a viral vector, a method using a plasmid vector, a method of introducing as mRNA, a method of introducing the reprogramming factor as a protein, etc. may be used. .
- a non-pluripotent stem cell refers to a cell that does not satisfy the above definition of a pluripotent stem cell, specifically, a somatic cell that constitutes a living tissue, and an endoderm, A differentiated cell that has lost the ability to differentiate into either the mesoderm or ectoderm.
- cardiomyocytes myoblasts, mesenchymal stem cells, vascular endothelial cells, vascular endothelial progenitor cells, fibroblasts, bone marrow derived cells, adipose derived cells, hepatocytes, sinusoidal endothelial cells, Kupffer cells, stellate cells, Pit cells, bile duct epithelial cells, kidney cells, granule cells, collecting duct epithelial cells, mural epithelial cells, podocytes, mesangial cells, smooth muscle cells, tubular cells, interstitial cells, glomerular cells, adrenal medullary cells, adrenal glands Cortical cells, spherical layer cells, bundled layer cells, reticulolayer cells, epidermal keratinocytes, melanocytes, napped muscle cells, hair follicle cells, buccal mucosa cells, gastric mucosal cells, intestinal mucosal cells, olfactory epithelial cells
- the origin of the animal species of the cells used in the present invention is not particularly limited, but for example, human, rat, mouse, guinea pig, marmoset, rabbit, dog, cat, sheep, pig, goat, monkey, chimpanzee or Examples thereof include mammal animals such as immunodeficient animals, birds, reptiles, amphibians, amphibians, fish, and insects.
- human cells pigs when used for pig treatment, monkeys when used for monkey treatment, and chimpanzee-derived cells when used for chimpanzee treatment. desirable.
- cells collected from the patient himself autologous cells
- cells collected from other people's cells transgenic cells
- commercially available cell lines may be used. It may be.
- non-pluripotent stem cells obtained by inducing differentiation from pluripotent stem cells are not particularly limited.
- cells obtained by inducing differentiation from pluripotent stem cells include cardiomyocytes, myocardial blasts, myoblasts, mesenchymal stem cells, Examples include vascular endothelial cells, vascular endothelial progenitor cells, fibroblasts, bone marrow-derived cells, fat-derived cells, or a mixture of two or more cells.
- liver parenchyma Any one of cells, sinusoidal endothelial cells, Kupffer cells, stellate cells, pit cells, bile duct epithelial cells, vascular endothelial cells, vascular endothelial progenitor cells, fibroblasts, bone marrow-derived cells, adipose-derived cells, and mesenchymal stem cells Or a mixture of two or more types of cells.
- cells obtained by inducing differentiation from pluripotent stem cells include kidney cells, granule cells Collecting duct epithelial cells, mural epithelial cells, podocytes, mesangial cells, smooth muscle cells, tubular cells, interstitial cells, glomerular cells, vascular endothelial cells, vascular endothelial progenitor cells, fibroblasts, bone marrow derived cells, Examples include any one of adipose-derived cells and mesenchymal stem cells, or a mixture of two or more cells.
- the cells used include adrenal medullary cells, adrenal cortical cells, spherical layer cells, bundled layers Examples include cells, upper layer cells, vascular endothelial cells, vascular endothelial progenitor cells, fibroblasts, bone marrow-derived cells, adipose-derived cells, mesenchymal stem cells, or a mixture of two or more cells. It is done.
- cells obtained by inducing differentiation from pluripotent stem cells include epidermal keratinocytes, melanocytes, napped muscle cells, hair follicle cells, blood vessels
- pluripotent stem cells include epidermal keratinocytes, melanocytes, napped muscle cells, hair follicle cells, blood vessels
- endothelial cells include endothelial cells, vascular endothelial progenitor cells, fibroblasts, bone marrow-derived cells, adipose-derived cells, mesenchymal stem cells, or a mixture of two or more cells.
- cells obtained by inducing differentiation from pluripotent stem cells include buccal mucosa, gastric mucosa, intestinal mucosa, olfactory epithelium, Among the cells of the oral mucosa and uterine mucosa, one or a mixture of two or more types of cells can be used.
- cells obtained by inducing differentiation from pluripotent stem cells include midbrain dopamine neurons, cerebral neurons, retinal cells, cerebellum Among cells and hypothalamic endocrine cells, any one or a mixture of two or more types of cells may be used, but the invention is not particularly limited.
- cells obtained by inducing differentiation from pluripotent stem cells include T cells, B cells, neutrophils, eosinophils, basophils, monocytes, Any one of platelets and erythrocytes, or a mixture of two or more cells may be used, but the invention is not particularly limited.
- medium refers to a cell culture medium for culturing cells, particularly animal cells.
- the medium is used as the same meaning as the cell culture solution. Therefore, the medium used in the present invention refers to a liquid medium.
- the type of the medium can be a commonly used medium and is appropriately determined depending on the type of cells to be cultured.
- the culture method for maintaining the undifferentiation of pluripotent stem cells before differentiation induction may be in accordance with a conventional method, and is not particularly limited.
- a method for inducing differentiation of pluripotent stem cells a differentiation induction method optimized for obtaining a target cell may be used, and it is not particularly limited.
- the culture method for maintaining the undifferentiated nature of pluripotent stem cells and the culture for inducing differentiation may be performed in a flat cell culture dish, or may be performed in a suspension-stirring three-dimensional culture, and are not particularly limited. .
- pluripotent stem cells means a cell population containing pluripotent stem cells and differentiated cells derived from pluripotent stem cells (also referred to herein as “first cell population”). Pluripotency in a cell population by removing all or part of the target pluripotent stem cell from the target, or annihilating or killing part of the target pluripotent stem cell, or stopping or delaying cell proliferation of the target pluripotent stem cell Refers to reducing the relative number of sex stem cells.
- the decrease in pluripotent stem cells in the present invention is caused by a decrease in the relative number of pluripotent stem cells in a cell population due to induction of apoptosis or necrosis, or by arresting or delaying only the cell cycle of pluripotent stem cells. It is.
- “decreasing” pluripotent stem cells means, for example, 5% compared to the case where a cell population containing pluripotent stem cells is cultured by a conventional culture method not using the method of the present invention.
- pluripotent stem cells are reduced.
- a cell population obtained by reducing pluripotent stem cells from a cell population (first cell population) containing pluripotent stem cells and differentiated cells derived from pluripotent stem cells by the method of the present invention is also referred to as “second cell population”.
- pluripotent stem cells and differentiated cells derived from pluripotent stem cells are cultured at a temperature of about 37 ° C., which is the body temperature of a thermostat animal.
- pluripotent stem cells can be removed or reduced by culturing at a temperature higher than the body temperature.
- the culture temperature for removing or reducing pluripotent stem cells may be 40.5 ° C. or higher, for example, 40.5 ° C., 40.6 ° C., 40.7 ° C., 40.
- the culture temperature for removing or reducing the pluripotent stem cells may be 40.5 ° C to 45.0 ° C, 40.7 ° C to 43.7 ° C, or 40.8 ° C to It may be 43.5 ° C, 40.9 ° C to 43.3 ° C, or 41.0 ° C to 43.0 ° C. In particular, when cultured in the range of 41.0 ° C.
- the step of culturing a cell population containing pluripotent stem cells and differentiated cells derived from pluripotent stem cells at a temperature of 40.5 ° C. or higher comprises activating pluripotent stem cells by activating TRPV-1 described later It may also be one that reduces pluripotent stem cells by an action other than activating TRPV-1.
- the time for culturing under conditions higher than the body temperature may be appropriately selected according to the culturing temperature, for example, 6 hours, 7 hours, 8 hours, 9 hours, 10 hours, 11 hours, 12 hours, 13 hours, 14 hours, 15 hours, 16 hours, 17 hours, 18 hours, 19 hours, 20 hours, 21 hours, 22 hours, 23 hours, 24 hours 25 hours, 26 hours, 27 hours, 28 hours, 29 hours, 30 hours, 31 hours, 32 hours, 33 hours, 34 hours, 35 hours, 36 hours, 37 hours, 38 hours, 39 hours, 40 hours, 41 Time, 42 hours, 43 hours, 44 hours, 45 hours, 46 hours, 47 hours, 48 hours, 49 hours, 50 hours, 51 hours, 52 hours, 53 hours, 54 hours, 55 hours, 56 hours, 57 hours, 5 Time, 59 hours, 60 hours, 61 hours, 62 hours, 63 hours, 64 hours, 65 hours, 66 hours, 67 hours, 68 hours, 69 hours, 70 hours, 71 hours,
- it may be 6 to 80 hours, 9 to 75 hours, 10 to 72 hours, 11 to 70 hours, or 12 to 68 hours. It may be time. For example, when culturing at 41.0 ° C. to 43.0 ° C., culturing in the range of 10 hours to 72 hours reduces cell proliferation of pluripotent stem cells and induces differentiation of pluripotent stem cells. Thereafter, the cytotoxic activity of differentiated cells is suppressed, which is preferable.
- a conventional method may be used as a method for evaluating the remaining pluripotent stem cells after induction of differentiation.
- Oct4 also referred to as Oct3 or Oct3 / 4
- Lin28 SSEA-3
- SSEA-4 Tra-1 60
- the evaluation method include, but are not limited to, confirmation by a flow cytometer, confirmation of protein expression by Western blotting, confirmation of mRNA expression using a real-time PCR method, and the like.
- a conventional method may be followed as a method for evaluating the ratio of somatic cells after differentiation induction.
- the ratio of cells expressing proteins and genes expressed in cells constituting the heart such as cardiac troponin T (cTnT) and Nkx2.5 may be evaluated.
- cardiac troponin T (cTnT) and Nkx2.5 may be evaluated.
- the ratio of cells expressing proteins and genes expressed in fibroblasts such as vimentin and SM22 may be evaluated.
- the ratio of other somatic cells the ratio of cells expressing a protein or gene that is specifically expressed by the target cell may be evaluated, and is not particularly limited.
- TRPV-1 transient receptor potential channel subfamily V member 1 refers to a membrane protein cloned as a capsaicin receptor. It is known as a non-selective cation channel that is activated against a wide range of endogenous and exogenous physical and chemical stimuli. It is activated at a temperature higher than 43 ° C., or activated against capsaicin or allyl isothiocyanate. When TRPV-1 is activated, cations flow into the cell and depolarize neurons, resulting in action potential generation from activation of voltage-gated Na + channels, causing pain. It is also activated by low pH. TRPV-1 is expressed mainly in the sensory nervous system, but is also expressed in the central nervous system and other tissues, and is considered to be a molecule mainly involved in the regulation and transmission of pain stimuli It is.
- the present invention has found that TRPV-1 of pluripotent stem cells and differentiated cells derived from pluripotent stem cells after induction of differentiation have a difference in sensitivity to temperature and chemical substances, and by activating TRPV-1
- the present invention relates to a method for removing or reducing pluripotent stem cells from a cell population containing pluripotent stem cells and differentiated cells derived from pluripotent stem cells.
- Examples of agonists (agonists) of TRPV-1 include capsaicin, N-oleoyldopamine (OLDA), alvanyl, olbanil, AM404 (anandamide), 2-APB, NADA, PPAHV, anti-TRPV-1 antibody and the like.
- the present invention is not limited as long as it binds to TRPV-1 and exhibits agonist activity.
- the concentration of capsaicin may be 20 ⁇ M to 100 mM, 30 ⁇ M to 50 mM, 40 ⁇ M to 20 mM, or 50 ⁇ M to 10 mM.
- the concentration of OLDA may be 600 nM to 100 mM, 750 nM to 10 mM, 1 ⁇ M to 1 mM, or 2 ⁇ M to 750 ⁇ M.
- the concentration of albanyl may be 7 ⁇ g / mL (0.016 mM) to 10 mg / mL (22.746 mM), or 10 ⁇ g / mL (0.023 mM) to 5 mg / mL (11.373 mM). It may be 15 ⁇ g / mL (0.034 mM) to 3 mg / mL (6.824 mM).
- a method for removing or reducing pluripotent stem cells from a cell population containing pluripotent stem cells and differentiated cells derived from differentiation-induced pluripotent stem cells is obtained by applying a cell population obtained after differentiation induction treatment to the cell population obtained after differentiation induction treatment.
- the treatment may be performed, or may be performed on a cell tissue after being processed by tissue engineering such as a cell sheet, and may be appropriately selected according to the purpose.
- tissue engineering such as a cell sheet
- it may be used in combination with a conventional method for removing or reducing pluripotent stem cells, and is not limited.
- a method of using the above-described method of causing the agonist of TRPV-1 to be treated at a temperature at which pluripotent stem cells die and at which the differentiated cells derived from pluripotent stem cells do not die may be used.
- the cell population includes those other than cells and is not particularly limited.
- it may be a cell population in which collagen, proteoglycan, laminin, laminin 5, fibronectin, hyaluronic acid, entactin, tenascin, elastin, etc. constituting cells and extracellular matrix are mixed, and cells and cells produced by the cells
- It may be a composition comprising an outer matrix.
- the protein constituting the extracellular matrix may be a gene recombinant protein, or a protein produced from a cell into which a gene encoding it is incorporated or introduced by a vector or the like.
- the cell population may be in a form in which a plurality of layered cells (for example, cell sheets) are laminated, and the cells are suspended in a gel containing an extracellular matrix and poured into a mold. It may also be a
- the cell sheet in the present invention refers to a cell group composed of a single layer or a plurality of sheet-like cell layers obtained by culturing on a cell culture device and peeling from the cell device.
- the method for obtaining the cell sheet is not particularly limited.
- the cells are cultured on a cell culture device coated with a polymer whose molecular structure is changed by stimulation of temperature, pH, light, etc.
- the method of peeling the cells from the surface of the cell culture equipment while maintaining the adhesion state between cells with any cell culture equipment Examples thereof include a method of culturing cells and physically detaching them from the edge of cell culture equipment using tweezers.
- cells are cultured in a temperature range where the hydration power of the polymer is weak on a cell culture support coated with a polymer whose hydration power changes in the temperature range of 0 to 80 ° C. Is cultured by changing the temperature to a temperature at which the hydration power of the polymer is strong, and the cells are detached in a sheet form. At that time, the cells are cultured in a temperature range where the hydration power of the polymer is weak on a cell culture support having a surface coated with a polymer whose hydration power changes in the temperature range of 0 to 80 ° C. Usually, the temperature is preferably 37 ° C., which is a temperature for culturing cells.
- the temperature-responsive polymer used in the present invention may be either a homopolymer or a copolymer. Examples of such a polymer include polymers described in JP-A-2-21865.
- Some cell types are difficult to adhere on the cell culture equipment.
- collagen, laminin, laminin 5, fibronectin, matrigel, etc. alone or a mixture of two or more kinds on the cell culture equipment. It may be coated and cultured.
- These cell adhesion protein coating methods may be in accordance with conventional methods. Usually, an aqueous solution of cell adhesion protein is applied to the surface of the equipment, and then the aqueous solution is removed and rinsed.
- the number of cells to be seeded to produce a cell sheet varies depending on the animal species and cell type of the cells used, but generally 0.3 ⁇ 10 4 to 10 ⁇ 10 6 cells / cm 2 is good. Preferably, 0.5 ⁇ 10 4 to 8 ⁇ 10 6 pieces / cm 2 , more preferably 0.7 ⁇ 10 4 to 5 ⁇ 10 6 pieces / cm 2 .
- the temperature of the culture equipment to which the cultured cells are attached is set to be equal to or higher than the upper critical solution temperature or lower than the lower critical solution temperature. Can be peeled off.
- the culture conditions other than the temperature may be in accordance with conventional methods and are not particularly limited.
- the medium to be used may be a medium to which serum such as known fetal bovine serum (FBS) is added, or a serum-free medium to which such serum is not added.
- FBS fetal bovine serum
- Cardiomyocyte sheets using cardiomyocytes derived from pluripotent stem cells used in the present invention are non-cardiomyocytes such as fibroblasts, wall cells, and vascular endothelial cells induced to differentiate from pluripotent stem cells in addition to cardiomyocytes May be included. Depending on the purpose, cells can be removed using a cell sorter or an antibody, or the necessary cells can be added.
- the “cardiomyocyte sheet” in the examples in this specification includes cells derived from pluripotent stem cells such as fibroblasts, mural cells, vascular endothelial cells, and non-cardiomyocytes, in addition to cardiomyocytes.
- poly (N-isopropylacrylamide) is known as a polymer having a lower critical solution temperature at 31 ° C. In the free state, dehydration occurs in water at a temperature of 31 ° C. or more, and polymer chains aggregate and become cloudy. Conversely, at a temperature of 31 ° C. or lower, the polymer chain is hydrated and dissolved in water. In the present invention, this polymer is coated and fixed on the surface of a device such as a petri dish. Therefore, if the temperature is 31 ° C.
- the polymer on the surface of the culture equipment is similarly dehydrated, but the polymer chain is coated and fixed on the surface of the culture equipment so that the surface of the culture equipment is hydrophobic.
- the polymer on the surface of the culture equipment hydrates, but since the polymer chain is coated and fixed on the surface of the culture equipment, the culture equipment surface becomes hydrophilic.
- the hydrophobic surface is an appropriate surface on which cells can attach and proliferate, and the hydrophilic surface becomes a surface on which cells cannot attach, and the cells or cell sheet in culture are peeled off only by cooling. Will be allowed to.
- the shape of the cell culture device for cell sheet preparation used in the present invention is not particularly limited.
- the shape of a cell insert cultivated on a dish, a multiplate, a flask, a porous membrane, or a flat plate is used.
- a film-like thing etc. are mentioned.
- cells to be cultured are epithelial cells
- the cell culture equipment to be coated include substances that can generally be given form, such as glass, modified glass, polystyrene, polymethyl methacrylate, and the like, which are usually used for cell culture, Examples thereof include molecular compounds and ceramics.
- the cell sheet in the present invention is not damaged by proteolytic enzymes represented by dispase, trypsin and the like during culture. Therefore, the cell sheet peeled from the cell culture equipment has an adhesive protein, and when the cells are peeled into a sheet shape, the cell-cell desmosome structure is maintained. As a result, when the cell sheet is affixed to the affected part of the living body or when the cell sheets are laminated, the cell sheets can be adhered and can be efficiently engrafted.
- dispases which are proteolytic enzymes, are known to be capable of peeling with 10 to 40% of the cell-cell desmosome structure. Since the cell sheet is almost destroyed, the resulting cell sheet is weak.
- the cell sheet of the present invention has a desmosome structure and a basement membrane-like protein remaining in 60% or more, and can obtain various effects as described above.
- the method for producing a cell composition having a plurality of cell layers in the present invention is not particularly limited.
- a cell is seeded on a cell culture device and a protein constituting an extracellular matrix protein thereon.
- a gel containing laminin, collagen, gelatin, cadherin, hyaluronic acid, fibronectin, fibrin, elastin, chitin, chitosan, vitronectin, etc. cells are seeded and stacked to obtain a cell composition having a cell layer It can be obtained by a method, a method in which cultured cells are peeled in a sheet shape, and if necessary, a cultured cell moving jig is used to laminate the cultured cell sheets.
- the temperature of the culture medium is not particularly limited if the polymer coated on the surface of the culture equipment has an upper critical solution temperature or lower, and if the polymer has a lower critical solution temperature or higher, the temperature is particularly limited.
- culturing in a low temperature range for example, 10 ° C. or lower
- a high temperature range for example, 50 ° C. or higher
- the culture conditions other than the temperature may be in accordance with conventional methods and are not particularly limited.
- the medium to be used may be a medium to which serum such as known fetal bovine serum (FBS) is added, or a serum-free medium to which such serum is not added.
- FBS fetal bovine serum
- these materials are usually polyvinylidene difluoride (PVDF), silicon.
- PVDF polyvinylidene difluoride
- Polyvinyl alcohol, urethane, cellulose and derivatives thereof, chitin, chitosan, collagen, gelatin, fibrin glue, etc. are used in contact with the cell sheet in the form of a membrane, porous membrane, nonwoven fabric or woven fabric.
- the antibodies used in the immunocytochemistry and flow cytometry in this example are as follows. Anti-cardiac troponin T (cTnT, Thermo Scientific, Rockford, Illinois, USA) Anti-Tra-1 60 mouse monoclonal antibody (Millipore, Billerica, Massachusetts, USA) Anti-SM22 rabbit polyclonal antibody (Abercam, Cambridge, UK) Anti-myocardial troponin T rabbit polyclonal antibody (Abercam, Cambridge, UK) Anti-Nkx2.5 goat polyclonal antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA) ⁇ Anti-Oct4 goat polyclonal antibody (R & D Systems) Secondary antibodies were purchased from Jackson ImmunoResearch Laboratory (West Grove, Pennsylvania, USA). Unless otherwise specified, reagents purchased from Life Technologies (California, USA) were used.
- Human iPS cells 253G1 and 2017 were purchased from RIKEN (Tsukuba, Japan). Human iPS cell 1231A3 strain was transferred from Kyoto University. In experiments using feeder cells, iPS cells were placed on mouse fibroblasts (MEF) (Reprocell, Tokyo) treated with mitomycin C, and 5 ng / mL basic fibroblast growth factor (bFGF, Reprocell, Japan). ) -Containing primate ES / iPS cell culture medium (Reprocell, Japan), and maintained in a humidified atmosphere of 5% CO 2 and 37 ° C.
- MEF mouse fibroblasts
- bFGF basic fibroblast growth factor
- iPS cells were subcultured as small cell clusters using an ES / iPS cell detachment solution (CTK solution) (Reprocell, Japan) every 3 to 4 days.
- CTK solution ES / iPS cell detachment solution
- StemFit AK03 Alignment AK03
- iPS cells were cultured and adapted on iMatrix 511 (Nippi Co., Tokyo, Japan), followed by maintenance culture.
- iPS cells were passaged every 7-8 days as single cells using TryPLE Select (Life Technologies, CA, USA) (reference: Nakagawa M., et al. A novel efficient feeder-free culture). system for the derivation of human induced pluripotent stem cells. Sci Rep.
- iPS cells were cultured in a humidified atmosphere of 40 ° C., 41 ° C., 42 ° C., and 5% CO 2 . Sample photographs were taken and analyzed with an inverted microscope (Nikon, Tokyo, Japan) and NIS-Elements software (Nikon, Tokyo, Japan).
- a lentiviral vector ( ⁇ -MHC-puro rex-1-neo) containing both a mouse ⁇ -myosin heavy chain ( ⁇ -MHC) promoter-controlled puromycin resistance gene and a rex-1 promoter-controlled neomycin resistance gene is available from Adgene (Cambridge). , Massachusetts, USA).
- Adgene CD34+ ⁇ -MHC-puro rex-1-neo
- the medium containing the vector was removed, and a fresh medium was added for further cultivation.
- the culture supernatant containing the recombinant lentivirus was collected. Centrifuge the culture supernatant at 1700 ⁇ g for 5 minutes to remove large cell debris, and place the supernatant on a 0.45 ⁇ m filter (Merck Millipore, Billerica, Massachusetts, USA) to remove fine debris. I passed. Subsequently, the supernatant was subjected to ultracentrifugation in an ultracentrifuge (CP80 ⁇ , Hitachi Koki Co., Tokyo, Japan) for 1.5 hours under conditions of 87,000 ⁇ g and 4 ° C., and concentrated 100 times. The concentrated virus used to infect human iPS cells was obtained.
- Small cell aggregates were resuspended in 1 mL medium (primate ES / iPS cell medium), transferred to a 15 mL centrifuge tube (Becton Dickinson and Company), and allowed to stand at room temperature for 5 minutes. After removing 500 ⁇ L of the supernatant, 400 ⁇ L of fresh medium containing 8 ⁇ g polybrene (Sigma Aldrich) was added. 100 ⁇ L of concentrated virus supernatant was added, mixed with the cells, and incubated at 37 ° C. for 6 hours. During the 6-hour incubation, the cell and virus suspensions were occasionally agitated.
- neomycin-resistant feeder cells SL10 Reprocell
- SL10 neomycin-resistant feeder cells
- 1 mL of medium was added to the cells, and the virus particles were washed away by changing the medium 36 hours after infection.
- the cells were treated with G418 sulfate (400 ⁇ g / mL) (Invitrogen) for 36 hours. Then, in order to wash away G418 sulfate, it rinsed twice with the phosphate buffered saline (PBS), and also culture
- phosphate buffered saline PBS
- iPS cells After inducing differentiation of iPS cells into cardiomyocytes, they were detached using 0.05% trypsin / EDTA, and cell aggregates were made into single cells using a strainer (BD Biosciences, San Jose, CA, USA) Using DMEM (Sigma Aldrich, St. Louis, Missouri, USA) containing 10% FBS, 2.1 ⁇ 10 5 cells / cm 2 of cells were seeded on the culture dish, and maintained at 37 ° C., 5% CO 2 , humidified. Cultured under atmosphere.
- cardiomyocytes derived from iPS cells were purified by treatment with puromycin (Sigma Aldrich, 1.5 ⁇ g / mL) for 1 day.
- IPS cell-derived fibroblasts were obtained by a technique (pre-plating method) of seeding in a culture dish in advance. That is, after inducing differentiation from iPS cells to myocardium, the cells were seeded and cultured on a non-coated culture dish using 10% FBS-containing DMEM. Thereafter, the cells that did not adhere to the culture dish were discarded, and the adherent cells were washed thoroughly three times with PBS ( ⁇ ) and cultured using DMEM containing 10% FBS. More than 99% of the cells were SM22 positive, and the percentage of cTnT positive was less than 1%. IPS cell-derived fibroblasts that were passaged 2-3 times were used in the experiments.
- iPS cell aggregates and iPS cell-derived cardiomyocytes were co-cultured for 1 day using AK03 medium on a 24-well plate. Subsequently, the cells were cultured for 2 days at 37 ° C. (AK03 medium), 42 ° C. (AK03 medium), 37 ° C. (10% FBS-containing DMEM), or 42 ° C. (10% FBS-containing DMEM).
- IPS cells cultured on MEF were detached as single cells using Accumax (Innovative Cell Technologies, San Diego, CA, USA).
- mTeSR1 Stem Cell Technology, Canada
- Y27632 Stem Cell Technology, Canada
- 2 ⁇ 10 7 iPS cells including MEF
- myocardium 12 days after induction of myocardial differentiation Cells were co-cultured (Day 0).
- the medium was replaced with mTeSR1 medium not containing Y27632.
- the cells were cultured in mTeSR1 medium at 37 ° C. for 48 hours, or at 42 ° C.
- the cells were fixed with 4% paraformaldehyde and analyzed by the method of Matsuura et al. (Matsuura K., et al. Creation of human cardiac cells using pluripotent stem cells. 1 Biochem Biophys Res Commun. 24:25; .) Immunostaining was performed. To perform TUNEL analysis, staining was performed using the Click-iT TUNEL Alexa Fluor 488 Imaging Kit (Life Technologies, CA, USA) according to the instructions. Cell nuclei were stained with Hoechst 33258 (Sigma Aldrich). Stained samples were photographed using ImageXpress (Molecular Devices, Sunnyvale, Calif., USA) and Metaxpress and AcuityXpress software (Molecular Devices).
- RNA extraction and quantitative RT-PCR Total RNA extraction and RT-PCR were performed by Matsuura K. et al. Kodama F .; Luo method (Matsuura K, Kodama F.et al., Elimination of remaining undifferentiated iPS cells in the process of human cardiac cell sheet fabrication using a methionine-free culture condition.Tissue Eng Part C Methods.2014 Sep 23.) performed according to It was.
- the primer pair and Taqman MGB probe used were the above-mentioned Matsuura K, Kodama F. et al. The same method was used. Specific primer information is shown in Table 1. All primers purchased from Life Technologies were used. Quantitative PCR was analyzed using a 7300 real-time PCR system (Applied Biosystems). Relative mRNA expression levels were calculated using a standard curve of GAPDH or ⁇ -actin mRNA expression levels.
- TRPV-1 knockdown iPS cells cultured on iMatrix511 were treated with TRPV-1 siRNA (Life Technologies, catalog number: 4392240) and negative control (Life Technologies) using Lipofectamine® RNAi Max Transfection Reagent (Life Technologies) according to the instructions. Technologies, Silencer (registered trademark) Select Negative Control No. 1 siRNA, product number: 4390843).
- TRPV-1 agonist The following reagents were used as agonists of TRPV-1. ⁇ Capsaicin (Sigma Aldrich) ⁇ Albanil (Wako Pure Chemical Industries) ⁇ OLDA (Tocris) -Solvent (vehicle): ethanol (Wako Pure Chemical Industries, Ltd.)
- Example 1 In order to confirm whether high culture temperature damages iPS cells, first, iPS cells cultured on laminin E8 fragment were cultured at 37 ° C, 40 ° C, 41 ° C, or 42 ° C for 1 day or IPS cells cultured at 37 ° C. and 40 ° C. for 2 days (FIG. 1A, B) had a significant increase in cell number depending on the culture time (37 ° C .; Day 0; 10311 ⁇ 1395, Day 1; 23748 ⁇ 1611, Day2; 41563 ⁇ 760, 40 ° C .; Day1; 21758 ⁇ 3219, Day2; 40706 ⁇ 591). Interestingly, iPS cells cultured at 41 ° C.
- TUNEL positive cells increased significantly in a culture time-dependent manner (Day 0; 3.4 ⁇ 0.2, Day 1; 9.4 ⁇ 4.1, Day 2; 33. 0 ⁇ 5.6) (FIGS. 1C, D).
- a temperature of 42 ° C. is an important temperature at which human iPS cells cause apoptosis. Similar results were observed for these cytotoxic effects at 42 ° C. in different iPS cell lines (1231A3 line) cultured without feeder cells.
- iPS cells were cultured at 42 ° C. for 1 hour or 3 hours, and then cultured at 37 ° C. until 24 hours from the start of the culture.
- the number of cells increased significantly compared to the number of cells of Day 0 at any culture time (before the start of culture; 11897 ⁇ 207, 37 ° C. for 24 hours; 25721 ⁇ 3477, 42 ° C. 1 hour + 37 ° C. 23 hours; 23391 ⁇ 842, 42 ° C. 3 hours + 37 ° C. 21 hours; 20834 ⁇ 1942) (FIG. 3A, B).
- a culture temperature of 42 ° C. is effective for iPS cells cultured on MEF in order to confirm the effect. I checked if there was.
- iPS cells cultured on MEF were cultured at 42 ° C., many cells that formed colonies died by Day 2, but cells around the colonies remained (FIG. 4A). From the results of confocal high content image analysis (Confocal high content image analysis), the number of Oct4-positive cells was significantly decreased depending on the culture time (Day 0; 9931 ⁇ 250, Day 1; 5250 ⁇ 1866, Day 2; 767 ⁇ ).
- iPS cells When iPS cell aggregates were co-cultured with iPS cell-derived cardiomyocytes for 1 day, iPS cells were engrafted with cardiomyocytes (Day 0; 7531 ⁇ 1229) (FIGS. 4C and D).
- iPS cell culture medium AK03
- cardiomyocyte culture medium 10% FBS-containing DMEM
- the cells proliferate remarkably under either condition.
- the size of the colony increased and cardiomyocytes were arranged around the colony (Day2 (AK03); 15065 ⁇ 2166, Day2 (DMEM); 16984 ⁇ 3520) (FIG. 4C, D).
- Example 2 In order to apply the method for reducing iPS cells of the present invention in regenerative medicine applications, what kind of influence the culture conditions at 42 ° C. have on the differentiated cells used in biotechnologically produced tissues It is necessary to confirm. Since human iPS cell-derived cardiomyocyte sheets are mainly composed of cardiomyocytes, fibroblasts, etc., how the 42 ° C culture condition affects iPS cell-derived cardiomyocytes and fibroblasts confirmed.
- iPS cell-derived cardiomyocytes When iPS cell-derived cardiomyocytes were cultured under conditions of 37 ° C. or 42 ° C., the myocardial cells showed self-sustaining pulsation, and many cells remained alive under both conditions cultured at 37 ° C. and 42 ° C. for 2 days. It was. When cultured at 37 ° C. and 42 ° C. for 1 day (Day 1) or 2 days (Day 2), the number of cTnT positive and Nkx2.5 positive cardiomyocytes was almost unchanged (cTnT positive cells: 37 ° C. (Day 1); 11084 ⁇ 1810, 37 ° C. (Day 2); 12625 ⁇ 768, 42 ° C. (Day 1); 12343 ⁇ 105, 42 ° C.
- vimentin-positive or SM22-positive iPS cell-derived fibroblasts significantly increased compared to Day0 (vimentin-positive: Day0; 10451 ⁇ 835, Day1; 12686 ⁇ 593, Day2; 16353 ⁇ 868, SM22 positive: Day0; 10553 ⁇ 876, Day1; 12777 ⁇ 615, Day2; 16752 ⁇ 940) (FIG. 5C, D).
- mRNA expression analysis of myocardial and extracellular matrix genes was performed, and it was examined how the temperature of 42 ° C. affects the gene expression of iPS cell-derived cardiomyocytes and fibroblasts.
- iPS cells including myocardium and fibroblasts were cultured at 42 ° C.
- MYL2, MYL7, Col1A, Col3A, NPPA and NPPB mRNA expression did not change until 48 hours, but the expression level of TNNT2 was significant at 48 hours (Fig. 6A). This suggested that transcriptional activity was maintained even under 42 ° C. culture conditions.
- iPS cell-derived cardiomyocytes Cultivation of iPS cell-derived cardiomyocytes at 42 ° C. has little effect, but to clarify how the 42 ° C. culture method affects the construction of biotechnological tissue used for regenerative medicine is important.
- the iPS cell-derived cardiomyocytes after induction of myocardial differentiation were confirmed to be autonomously pulsating when cultured on a temperature-responsive culture dish at 37 ° C. for 4 days, and when the culture temperature was lowered, a monolayer cell sheet was produced ( FIG. 6D).
- iPS cell-derived cardiomyocytes were cultured at 42 ° C. for 2 days (Day 1 to Day 3) or 3 days (Day 1 to Day 4), and a cell sheet was obtained even when the culture temperature was lowered (FIG.
- TRPV-1 transient receptor potential vanilloid 1
- TRPV-1 was significantly higher in iPS cell-derived cardiomyocytes than in baseline (before 42 ° C. culture) iPS cell-derived cardiomyocytes and iPS cell-derived cardiomyocytes cultured at 42 ° C. for 24 hours. It was high (FIG. 7B).
- iPS cells were transfected with siRNA against TRPV-1. It was confirmed that the increase in the expression level of TRPV-1 accompanying the culture at 42 ° C. was inhibited by TRPV-1 knockdown (FIG. 7C), and in agreement with this, iPS cells after the culture at 42 ° C. The decrease in the number of cells was also inhibited by TRPV-1 siRNA transfection (FIG. 7D, E). These results suggested that TRPV-1 plays an important role in the removal of iPS cells in culture at 42 ° C.
- TRPV-1 pharmacological activity
- TRPV-1 agonists including capsaicin
- OLDA FIG. 9
- albanyl FIG. 10
- the number of cells depended on the concentration when capsaicin was used.
- treatment with other TRPV-1 agonists also showed cytotoxic activity against iPS cells (FIGS. 9 and 10).
- the method shown in the present invention can reduce undifferentiated pluripotent stem cells from a cell population obtained by inducing differentiation of a cell population containing pluripotent stem cells.
- a cell population can reduce the risk of forming a tumor when used as a transplant material for regenerative medicine, and is industrially useful.
- it is useful because it enables simple and inexpensive removal of undifferentiated pluripotent stem cells from a differentiated cell population derived from pluripotent stem cells used in applications such as experiments.
Abstract
Description
[2] 前記温度が、41℃~43℃である、[1]に記載の方法。
[3] 前記温度で培養する時間が、10時間~72時間の範囲である、[1]又は[2]に記載の方法。
[4] 前記多能性幹細胞が、人工多能性幹細胞及び/又は胚性幹細胞である、[1]~[3]のいずれか1項に記載の方法。
[5] 前記分化細胞が、心筋細胞、心筋芽細胞、線維芽細胞、壁細胞及び/又は血管内皮細胞を含む、[1]~[4]のいずれか1項に記載の方法。
[8] 前記TRPV-1を活性化する工程が、40.5℃以上の温度で培養して前記TRPV-1を活性化する工程である[7]に記載の方法。
[9] 前記温度が、40.5℃~45℃である[8]に記載の方法。
[10] 前記温度が、41℃~43℃である[8]又は[9]に記載の方法。
[11] 前記温度で培養して前記TRPV-1を活性化する工程の時間が、10時間~72時間の範囲である[8]~[10]に記載の方法。
[12] 前記TRPV-1を活性化する工程が、TRPV-1に対するアゴニストを添加する工程である[7]に記載の方法。
[13] 前記アゴニストが、カプサイシン、N-oleoyldopamine(OLDA)、アルバニル、オルバニル、AM404(アナンダミド)、2-APB、NADA、PPAHV、抗TRPV-1抗体からなる群から選択された1又は2以上のアゴニストである[12]に記載の方法。
[14] 前記多能性幹細胞が人工多能性幹細胞及び/又は胚性幹細胞である[7]~[13]に記載の方法。
[15] 前記分化細胞が、心筋細胞、心筋芽細胞、線維芽細胞、壁細胞及び/又は血管内皮細胞を含む[7]~[14]に記載の方法。
[21] 前記TRPV-1を活性化する工程が、40.5℃以上の温度で培養してTRPV-1を活性化する工程である[20]に記載の方法。
[22] 前記温度が、41℃~43℃である[20]又は[21]に記載の方法。
[23] 前記TRPV-1を活性化する工程が、TRPV-1に対するアゴニストを添加する工程である[20]に記載の方法。
[24] 前記アゴニストが、カプサイシン、N-oleoyldopamine(OLDA)、アルバニル、オルバニル、AM404(アナンダミド)、2-APB、NADA、PPAHV、抗TRPV-1抗体からなる群から選択された1又は2以上のアゴニストである[23]に記載の方法。
本実施例中の免疫細胞化学、フローサイトメトリーに用いた抗体は以下の通りである。
・抗心筋トロポニンT(cTnT、サーモサイエンティフィック社、ロックフォード、イリノイ州、米国)
・抗Tra-1 60マウス単クローン抗体(ミリポア社、ビレリカ、マサチューセッツ州、米国)
・抗SM22 ウサギポリクローナル抗体(アバカム社、ケンブリッジ、英国)
・抗心筋トロポニンT ウサギポリクローナル抗体(アバカム社、ケンブリッジ、英国)
・抗Nkx2.5 ヤギポリクローナル抗体(サンタクルズ バイオテクノロジー社、サンタクルズ、カリフォルニア州、米国)
・抗Oct4 ヤギポリクローナル抗体(R&Dシステム社)
2次抗体はジャクソン イムノリサーチ ラボラトリー社(ウエスト グローブ社、ペンシルベニア州、米国)から購入した。特に明記しない限り、試薬はライフ テクノロジーズ社(カリフォルニア州、米国)より購入したものを使用した。
ヒトiPS細胞253G1株及び2017株は、国立研究開発法人理化学研究所(筑波、日本)より購入した。ヒトiPS細胞1231A3株は京都大学より譲渡を受けた。フィーダー細胞を用いた実験は、マイトマイシンCで処理したマウス線維芽細胞(MEF)(リプロセル社、東京)上にiPS細胞を載せ、5ng/mL塩基性線維芽細胞増殖因子(bFGF、リプロセル社、日本)を含有した霊長類ES/iPS細胞用培地(リプロセル社、日本)を用いて5%CO2、37℃の加湿雰囲気条件にて維持培養を行った。iPS細胞は3~4日毎にES/iPS細胞剥離液(CTK溶液)(リプロセル社、日本)を用い、小さな細胞塊として継代を行った。フィーダー細胞を用いない実験では、StemFit AK03(味の素社、東京、日本)を培地として用い、iMatrix511(ニッピ社、東京、日本)上でiPS細胞を培養して適応させ、維持培養を行った。iPS細胞は7~8日毎に、TrypLE Select(ライフテクノロジー社、カリフォルニア州、米国)を使用してシングルセルとして継代を行った(参考:Nakagawa M.,et al.A novel efficient feeder-free culture system for the derivation of human induced pluripotent stem cells.Sci Rep.2014;4:3594.)。実験に応じて、iPS細胞は、40℃、41℃、42℃の加湿、5%CO2濃度の雰囲気にて培養を行った。試料の写真は倒立顕微鏡(ニコン社、東京、日本)及びNIS-Elementsソフトウエア(ニコン社、東京、日本)にて撮影して解析した。
マウスα-ミオシン重鎖(α-MHC)プロモーター制御ピューロマイシン耐性遺伝子及びrex-1プロモーター制御ネオマイシン耐性遺伝子の両方を含むレンチウイルスベクター(α-MHC-puro rex-1-neo)はアドジーン社(ケンブリッジ、マサチューセッツ州、米国)より購入した。当該レンチウイルスベクターを用いることで、ヒトiPS細胞を増殖させるときにG418によって未分化状態のiPS細胞だけを選別することができ、また、心筋へと分化誘導された後は、ピューロマイシンによって分化誘導した心筋細胞を選別することができる。遺伝子組み換えiPS細胞は以下の手順により得た。
ベクターの遺伝子導入は、商業的販売されている遺伝子導入キットのリポフェクタミン2000(インビトロジェン社)にて行い、組み換えレンチウイルスベクターの調整はViraPower(商標)レンチウイルスパッケージングミックス(インビトロジェン社)を用いて行った。
100mmポリスチレン培養皿(ベクトンディッキンソン アンド カンパニー社、フランクリンレイク、ニュージャージー州、米国)上に5×104細胞/cm2のHEK293FT細胞を播種した。24時間培養後、リポフェクタミン2000キット及びOPTI-MEM(インビトロジェン社)を用い、ベクター(3μg/培養皿)を遺伝子導入して8時間37℃にて培養した。その後、ベクターを含む培地を除去し、新鮮な培地を加えてさらに培養した。遺伝子導入してから72時間後、遺伝子組み換えレンチウイルスを含む培養上清を回収した。大きな細胞の破片を取り除くために1700×g、5分間、培養上清を遠心分離し、細かい破片を取り除くために上清を0.45μmのフィルター(メルクミリポア社、ビレリカ、マサチューセッツ州、米国)に通した。その後、上清を87,000×g、4℃の条件にて1.5時間、超遠心機(CP80β、日立工機社、東京、日本)にて超遠心処理を行って100倍濃縮を行い、ヒトiPS細胞に感染させるために用いる濃縮ウイルスを得た。
レンチウイルスを感染させる方法についてはNakagawaらの方法(Nakagawa M.,et al.A novel efficient feeder-free culture system for the derivation of human induced pluripotent stem cells.Sci Rep.2014;4:3594.)に改変を加えた手順で行った。以下にその手順を説明する。
ヒトiPS細胞を6ウェル培養皿(ベクトンディッキンソン アンド カンパニー社)の1つのウェルにてコンフルエントになるまで培養し、CTK溶液を用いて剥離した。小さな細胞凝集塊を1mLの培地(霊長類ES/iPS細胞用培地)にて再懸濁し、15mLの遠沈管(ベクトンディッキンソン アンド カンパニー社)に移して5分間室温にて静置した。500μLの上清を除いた後、8μgポリブレン(シグマアルドリッチ社)を含む400μLの新鮮な培地を添加した。100μLの濃縮ウイルス上清を添加し、細胞と混ぜ、37℃で6時間保温した。6時間の保温の間、時々細胞とウイルス懸濁液を撹拌した。6ウェル培養皿の2つのウェルに播種したネオマイシン耐性フィーダー細胞SL10(リプロセル社)の上に、レトロウイルスを感染させた細胞を播種し、37℃で培養した。一晩培養した後、1mLの培地を細胞に加え、感染後36時間後に培地交換を行うことでウイルス粒子を洗い流した。感染4日後、G418硫酸塩(400μg/mL)(インビトロジェン社)にて36時間処理した。その後、G418硫酸塩を洗い流すためにリン酸緩衝生理食塩水(PBS)にて2回リンスし、さらに数日間培養した。必要に応じてG418硫酸塩で再処理を行った。
iPS細胞の心筋分化誘導は、バイオリアクターシステム(エイブル社、日本)を用いて、Matsuuraらの方法(Matsuura K.,et al.Creation of human cardiac cell sheets using pluripotent stem cells.Biochem Biophys Res Commun.2012 Aug 24;425(2):321-7.、特許文献1参照)に従って実施した。細胞を播種する前に、温度応答性培養皿(UpCell(登録商標)、セルシード社、東京、日本)、もしくは細胞培養皿(コーニング社、コーニング、ニューヨーク州、米国)の表面をウシ胎児血清(FBS)にて2時間コーティングした。iPS細胞を心筋細胞へ分化誘導した後、0.05%トリプシン/EDTAを用いて剥離し、細胞凝集塊はストレーナー(BDバイオサイエンス社、サンノゼ、カリフォルニア州、米国)を使用してシングルセルとし、10%FBSを含んだDMEM(シグマアルドリッチ社、セントルイス、ミズーリ州、米国)を用いて2.1×105細胞/cm2の細胞を培養皿に播種し、37℃、5%CO2、加湿雰囲気下で培養した。いくつかの実験では、ピューロマイシン(シグマアルドリッチ社、1.5μg/mL)で1日間処理し、iPS細胞(201B7株、αMHC-puro/Rex1-neo)由来の心筋細胞を純化した。
ヒトiPS細胞から心筋へ分化誘導した後の細胞は、24ウェル培養皿(コーニング社)にて2.1×105細胞/cm2で播種し、10%FBS含有DMEDを用いて37℃、5%CO2、加湿雰囲気下にて2日間培養した。共培養実験を行う1日前、iMatrix511上で培養したiPS細胞をTrypLE Selectで剥離し、細胞凝集塊を形成させるためにY27632(化合物一般名)(10μM)(和光純薬工業社、日本)を含有するAK03培地を用い、シングルセル懸濁液をEZ Sphere(旭硝子社、東京、日本)上で1日間培養した。次の日、24ウェルプレート上でAK03培地を用い、iPS細胞の凝集塊50個とiPS細胞由来の心筋細胞とを1日間、共培養した。続いて、37℃(AK03培地)、42℃(AK03培地)、37℃(10%FBS含有DMEM)、又は42℃(10%FBS含有DMEM)の条件にて、それぞれ2日間培養を行った。
MEF上で培養したiPS細胞はAccumax(イノベーティブセルテクノロジーズ社、サンディエゴ、カリフォルニア州、米国)を用いてシングルセルとして剥離した。100mLバイオリアクターを用い、Y27632(10μM)を含有したmTeSR1(ステムセルテクノロジー社、カナダ)培地を使用して、2×107個のiPS細胞(MEFを含む)と心筋分化誘導後12日目の心筋細胞を共培養した(Day0)。翌日、Y27632を含まないmTeSR1培地へと交換した。共培養開始2日後(Day2)、mTeSR1培地にて37℃48時間、又は42℃6時間培養後に37℃42時間(合計48時間)培養した。培養開始4日後(Day4)、それぞれの条件の細胞をAccumaxで剥離し、FBSをあらかじめコーティングしていた24ウェル培養皿上に2.1×105細胞/cm2で再播種し、10%FBS含有DMEMを用いて37℃で培養し、FACS解析用に4%パラホルムアルデヒドで固定した。培養皿上の細胞は42℃で再び2日間培養し(Day5からDay7)、その後、37℃で1日間培養した(Day7からDay8)。
細胞を4%パラホルムアルデヒドで固定し、Matsuuraらの方法(Matsuura K.,et al.Creation of human cardiac cell sheets using pluripotent stem cells.Biochem Biophys Res Commun.2012 Aug 24;425(2):321-7.)に従い免疫染色を行った。TUNEL解析を行うために、Click-iT TUNEL Alexa Fluor 488イメージングキット(ライフテクノロジー社、カリフォルニア州、米国)を用い、説明書に従って染色を行った。細胞核はHoechst33258(シグマアルドリッチ社)にて染色した。染色した試料は、ImageXpress(モレキュラーデバイス社、サニーベール、カリフォルニア州、米国)及びMetaxpress and AcuityXpressソフトウエア(モレキュラーデバイス社)を用いて撮影した。
全量RNA抽出及びRT-PCRはMatsuura K.、 Kodama F.らの方法(Matsuura K,Kodama F.et al.,Elimination of remaining undifferentiated iPS cells in the process of human cardiac cell sheet fabrication using a methionine-free culture condition.Tissue Eng Part C Methods.2014 Sep 23.)に従い行った。使用したプライマーペアとTaqman MGBプローブは上記Matsuura K、 Kodama F.らの方法と同一のものを使用した。具体的なプライマーの情報は表1に示し、いずれのプライマーもライフ テクノロジーズ社より購入したものを使用した。定量的PCRは7300リアルタイムPCRシステム(アプライドバイオシステムズ社)を用いて解析した。相対的mRNA発現レベルはGAPDH、またはβ-アクチンmRNA発現レベルの標準曲線を用いて計算した。
それぞれの条件の細胞凝集塊は、Accumaxを用いて10分間37℃で剥離し、Matsuuraらの方法(Matsuura K,et al.Creation of human cardiac cell sheets using pluripotent stem cells.Biochem Biophys Res Commun.2012 Aug 24;425(2):321-7.)らの方法に従ってTra-1 60または心筋トロポニンT(cTnT)を染色した。細胞の割合はGallios(ベックマンコールター社、ブレア、カリフォルニア州、米国)及びCell Quest Pro version5.2ソフトウエアを用いて解析した。
iMatrix511上で培養したiPS細胞は、説明書に従ってLipofectamine(登録商標) RNAi Max Transfection Reagent(ライフ テクノロジー社)を使用してTRPV-1 siRNA(ライフ テクノロジーズ社、カタログ番号:4392240)、及びネガティブコントロール(ライフ テクノロジーズ社、Silencer(登録商標) Select Negative Control No.1 siRNA、製品番号:4390843)をトランスフェクトした。
TRPV-1のアゴニストとしては以下の試薬を用いた。
・カプサイシン(シグマアルドリッチ社)
・アルバニル(和光純薬工業社)
・OLDA(トクリス(Tocris)社)
・溶媒(vehicle):エタノール(和光純薬工業社)
データは平均値±標準偏差で表している。2つのグループ間を比較した統計学的解析は、Student’s t-testを用いて行った。複数グループ間の比較統計解析は一元配置分散分析(One-way analysis of variance)を行い、続いてDunnett検定にて比較を行った。p値が0.05より小さいものを統計学的に有意であるとした。
高い培養温度がiPS細胞へ傷害を与えるかどうかを確かめるために、まず初めにラミニンE8フラグメント上で培養したiPS細胞を37℃、40℃、41℃、又は42℃の条件にて、1日間又は2日間培養した(図1A、B)37℃及び40℃で培養したiPS細胞は培養時間に依存して有意に細胞数が増加した(37℃;Day0;10311±1395、Day1;23748±1611、Day2;41563±760、40℃;Day1;21758±3219、Day2;40706±591)。興味深いことに、41℃で培養したiPS細胞はDay1で細胞増殖が停止されており(13228±2037)、Day2では細胞数が減少していた(4278±848)。しかしながら、42℃で培養したiPS細胞は培養時間依存的に細胞数が有意に減少していた(Day1;2459±934、Day2;1216±229)。それに伴い、Oct4を発現するiPS細胞の数は37℃において顕著に増加し(Day0;8834±2545、Day1;17134±5731、Day2;31100±2894)、一方、42℃で培養したOct4を発現するiPS細胞の数は有意に減少した(Day1;2271±1208、Day2;961±26)(図2)。42℃での細胞数の減少に伴い、TUNEL陽性の細胞が培養時間依存的に有意に増加した(Day0;3.4±0.2、Day1;9.4±4.1、Day2;33.0±5.6)(図1C、D)。これらの結果から、42℃という温度はヒトiPS細胞がアポトーシスを引き起こす重要な温度であることを示唆している。42℃におけるこれらの細胞傷害効果は、フィーダー細胞無しで培養した異なったiPS細胞株(1231A3株)においても同様の結果が観察された。
本発明のiPS細胞を減少させる方法を再生医療の用途で適用するには、42℃の培養条件が、生物工学的に作製する組織に用いる分化した細胞に対して、どのような影響を与えるかを確かめる必要がある。ヒトiPS細胞由来の心筋細胞シートは、主に心筋細胞、線維芽細胞等から構成されているので、42℃培養条件が、iPS細胞由来心筋細胞及び線維芽細胞にどのように影響を及ぼすのかを確認した。
(ヒト心筋組織におけるTRPV-1介在型残存iPS細胞の除去)
高温条件は、温度感受性TRPチャンネルであるactivates transient receptor potential vanilloid 1(TRPV-1)を活性化することが知られている。我々は、次にiPS細胞やiPS細胞由来心筋細胞におけるTRPV-1の発現を明らかにした。iPS細胞のTRPV-1 mRNAの発現量は、42℃で9時間培養した場合に有意に上昇し、iPS細胞由来心筋細胞のTRPV-1 mRNAの発現は42℃で12時間培養後に上昇した(図7A)。一方、TRPV-1の発現量は、ベースライン(42℃培養前)のiPS細胞由来心筋細胞及び42℃で24時間培養したiPS細胞由来心筋細胞よりも、iPS細胞の発現量の方が有意に高かった(図7B)。これらの結果は、42℃培養によるTRPV-1の活性の違いが、42℃培養におけるiPS細胞とiPS細胞由来心筋細胞に対する除去効果の違いを説明しているのかもしれない。
Claims (15)
- 多能性幹細胞と多能性幹細胞由来の分化細胞とを含む細胞集団を、40.5℃以上の温度で培養し、前記細胞集団に含まれる前記多能性幹細胞を減少させる方法。
- 前記温度が、41℃~43℃である、請求項1に記載の方法。
- 前記温度で培養する時間が、10時間~72時間の範囲である、請求項1又は2に記載の方法。
- 前記多能性幹細胞が、人工多能性幹細胞及び/又は胚性幹細胞である、請求項1~3のいずれか1項に記載の方法。
- 前記分化細胞が、心筋細胞、心筋芽細胞、線維芽細胞、壁細胞及び/又は血管内皮細胞を含む、請求項1~4のいずれか1項に記載の方法。
- 請求項1~5のいずれか1項に記載の方法によって得られた、前記多能性幹細胞を減少させた細胞集団。
- 多能性幹細胞と、多能性幹細胞由来の分化細胞とを含む細胞集団から、多能性幹細胞を減少させる方法であって、
前記細胞集団に含まれる前記多能性幹細胞に発現するTRPV-1を活性化する工程、を含むことを特徴とする方法。 - 前記TRPV-1を活性化する工程が、40.5℃以上の温度で培養して前記TRPV-1を活性化する工程である請求項7に記載の方法。
- 前記温度が、41℃~43℃である請求項8に記載の方法。
- 前記温度で培養して前記TRPV-1を活性化する工程の時間が、10時間~72時間の範囲である請求項8又は9に記載の方法。
- 前記TRPV-1を活性化する工程が、TRPV-1に対するアゴニストを添加する工程である請求項7に記載の方法。
- 前記アゴニストが、カプサイシン、N-oleoyldopamine(OLDA)、アルバニル、オルバニル、AM404(アナンダミド)、2-APB、NADA、PPAHV、抗TRPV-1抗体のからなる群から選択された1又は2以上のアゴニストである請求項11に記載の方法。
- 前記多能性幹細胞が人工多能性幹細胞及び/又は胚性幹細胞である請求項7~12のいずれか1項に記載の方法。
- 前記分化細胞が、心筋細胞、心筋芽細胞、線維芽細胞、壁細胞及び/又は血管内皮細胞を含む請求項7~13のいずれか1項に記載の方法。
- 請求項7~14のいずれか1項に記載の方法によって得られた、前記多能性幹細胞を減少させた細胞集団。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/755,293 US10711247B2 (en) | 2015-08-31 | 2016-08-23 | Method for reducing pluripotent stem cells, method for producing cell population having reduced pluripotent stem cells |
| JP2017537771A JP6789572B2 (ja) | 2015-08-31 | 2016-08-23 | 多能性幹細胞を減少させる方法、多能性幹細胞を減少させた細胞集団の製造方法 |
| EP16841594.1A EP3346002B1 (en) | 2015-08-31 | 2016-08-23 | Method for reducing pluripotent stem cells, method for producing cell population having reduced pluripotent stem cells |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015171230 | 2015-08-31 | ||
| JP2015-171230 | 2015-08-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017038562A1 true WO2017038562A1 (ja) | 2017-03-09 |
Family
ID=58188057
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/074545 WO2017038562A1 (ja) | 2015-08-31 | 2016-08-23 | 多能性幹細胞を減少させる方法、多能性幹細胞を減少させた細胞集団の製造方法 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US10711247B2 (ja) |
| EP (1) | EP3346002B1 (ja) |
| JP (1) | JP6789572B2 (ja) |
| WO (1) | WO2017038562A1 (ja) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018225705A1 (ja) | 2017-06-05 | 2018-12-13 | テルモ株式会社 | 細胞培養物の製造方法 |
| WO2020027278A1 (ja) * | 2018-08-01 | 2020-02-06 | 国立大学法人大阪大学 | 心筋細胞の製造方法 |
| WO2020067435A1 (ja) * | 2018-09-27 | 2020-04-02 | 国立大学法人大阪大学 | 多能性幹細胞由来細胞のシート化方法 |
| WO2020067438A1 (ja) | 2018-09-27 | 2020-04-02 | 国立大学法人大阪大学 | 多能性幹細胞由来細胞のシート化方法 |
| WO2020067436A1 (ja) * | 2018-09-27 | 2020-04-02 | 国立大学法人大阪大学 | 多能性幹細胞由来細胞の移植片形成方法 |
| JP2020162441A (ja) * | 2019-03-28 | 2020-10-08 | 株式会社ニチレイ | 保存液 |
| US11395863B2 (en) | 2016-11-01 | 2022-07-26 | Terumo Kabushiki Kaisha | Modification method for sheet-shaped cell culture |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012133945A1 (ja) * | 2011-03-30 | 2012-10-04 | 学校法人東京女子医科大学 | 胚性幹細胞から心筋シートを製造する方法 |
| WO2013137491A1 (ja) * | 2012-03-15 | 2013-09-19 | 国立大学法人京都大学 | 人工多能性幹細胞から心筋および血管系混合細胞群を製造する方法 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6791756B2 (ja) * | 2014-01-21 | 2020-11-25 | ザ メディカル カレッジ オブ ウィスコンシン インクThe Medical College Of Wisconsin, Inc. | 多能性幹細胞の選択的な阻害のための方法 |
-
2016
- 2016-08-23 JP JP2017537771A patent/JP6789572B2/ja active Active
- 2016-08-23 US US15/755,293 patent/US10711247B2/en active Active
- 2016-08-23 WO PCT/JP2016/074545 patent/WO2017038562A1/ja active Application Filing
- 2016-08-23 EP EP16841594.1A patent/EP3346002B1/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012133945A1 (ja) * | 2011-03-30 | 2012-10-04 | 学校法人東京女子医科大学 | 胚性幹細胞から心筋シートを製造する方法 |
| WO2013137491A1 (ja) * | 2012-03-15 | 2013-09-19 | 国立大学法人京都大学 | 人工多能性幹細胞から心筋および血管系混合細胞群を製造する方法 |
Non-Patent Citations (3)
| Title |
|---|
| MATSUURA KATSUHISA: "TRPV-1-mediated elimination of residual iPS cells in bioengineered cardiac cell sheet tissues", SCIENTIFIC REPORTS, vol. 6, 18 February 2016 (2016-02-18), pages 21747, XP055367698 * |
| QI YAN ET AL.: "Role of TRPV1 in the differentiation of mouse embryonic stem cells into cardiomyocytes", PLOS ONE, vol. 10, no. 7, 24 July 2015 (2015-07-24), pages e0133211, XP055367697 * |
| See also references of EP3346002A4 * |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11395863B2 (en) | 2016-11-01 | 2022-07-26 | Terumo Kabushiki Kaisha | Modification method for sheet-shaped cell culture |
| WO2018225705A1 (ja) | 2017-06-05 | 2018-12-13 | テルモ株式会社 | 細胞培養物の製造方法 |
| WO2020027278A1 (ja) * | 2018-08-01 | 2020-02-06 | 国立大学法人大阪大学 | 心筋細胞の製造方法 |
| WO2020067435A1 (ja) * | 2018-09-27 | 2020-04-02 | 国立大学法人大阪大学 | 多能性幹細胞由来細胞のシート化方法 |
| WO2020067438A1 (ja) | 2018-09-27 | 2020-04-02 | 国立大学法人大阪大学 | 多能性幹細胞由来細胞のシート化方法 |
| WO2020067436A1 (ja) * | 2018-09-27 | 2020-04-02 | 国立大学法人大阪大学 | 多能性幹細胞由来細胞の移植片形成方法 |
| JPWO2020067438A1 (ja) * | 2018-09-27 | 2021-08-30 | 国立大学法人大阪大学 | 多能性幹細胞由来細胞のシート化方法 |
| JPWO2020067436A1 (ja) * | 2018-09-27 | 2021-08-30 | 国立大学法人大阪大学 | 多能性幹細胞由来細胞の移植片形成方法 |
| JPWO2020067435A1 (ja) * | 2018-09-27 | 2021-09-09 | 国立大学法人大阪大学 | 多能性幹細胞由来細胞のシート化方法 |
| JP2020162441A (ja) * | 2019-03-28 | 2020-10-08 | 株式会社ニチレイ | 保存液 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3346002A1 (en) | 2018-07-11 |
| EP3346002A4 (en) | 2019-02-27 |
| US10711247B2 (en) | 2020-07-14 |
| EP3346002B1 (en) | 2021-09-29 |
| US20180245047A1 (en) | 2018-08-30 |
| JPWO2017038562A1 (ja) | 2018-06-21 |
| JP6789572B2 (ja) | 2020-11-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2017038562A1 (ja) | 多能性幹細胞を減少させる方法、多能性幹細胞を減少させた細胞集団の製造方法 | |
| JP6450311B2 (ja) | 腎前駆細胞の製造方法及び腎前駆細胞を含む医薬 | |
| US11591569B2 (en) | Methods for epicardial differentiation of human pluripotent stem cells | |
| JP5611035B2 (ja) | 胚性幹細胞および人工多能性幹細胞由来からの高心臓形成性前駆細胞および心筋細胞の効率的製造および使用 | |
| DK3183337T3 (en) | Use of Jagged 1 / Frizzled 4 as cell surface marker for isolation of human cardiac ventricular progenitor cells | |
| Shariati et al. | Organoid technology: Current standing and future perspectives | |
| US20100183568A1 (en) | Method for production of hepatic-lobule-like cell cluster from adipose-tissue-derived cell | |
| JP5688970B2 (ja) | 多能性幹細胞の製造方法 | |
| Shafa et al. | Human-induced pluripotent stem cells manufactured using a current good manufacturing practice-compliant process differentiate into clinically relevant cells from three germ layers | |
| WO2016148216A1 (ja) | 肝細胞及び肝非実質細胞、並びにそれらの調製方法 | |
| WO2008116213A1 (en) | Dermal derived human stem cells and compositions and methods thereof | |
| WO2018100433A1 (en) | Methods for isolating human cardiac ventricular progenitor cells | |
| US20200231933A1 (en) | Human pluripotent stem cell derived endocardial endothelium | |
| JP6139054B2 (ja) | 細胞培養基材、およびそれを用いた細胞培養方法並びに多能性幹細胞の分化誘導方法 | |
| TW202043459A (zh) | 調節萬能性幹細胞之潛能的方法及應用 | |
| US11492596B2 (en) | Methods for differentiating cells into hepatic stellate cells | |
| JPWO2018025975A1 (ja) | インビトロで多能性幹細胞を分化誘導する方法 | |
| WO2018225703A1 (ja) | 分化誘導細胞の調製方法 | |
| CN112739208A (zh) | 人类多能成体干细胞 | |
| WO2017150294A1 (ja) | 多能性幹細胞様スフェロイドの製造方法および多能性幹細胞様スフェロイド | |
| WO2022254961A1 (ja) | 幹細胞を作製するための細胞培養補助剤、及び幹細胞の作製方法 | |
| Collins-Hooper et al. | Efficient myogenic reprogramming of adult white fat stem cells and bone marrow stem cells by freshly isolated skeletal muscle fibers | |
| Besser | Engineering a Compartmentalized Hydrogel Platform for In Vitro Assembly of Neuromuscular Junctions | |
| Brizi | Engineering Complex Kidney Structures for Disease Modelling, Drug Testing, and Studying Kidney Development | |
| Soong | Development of a novel technology to engineer heart muscle for contractile and paracrine support in heart failure |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16841594 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2017537771 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15755293 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2016841594 Country of ref document: EP |