WO2016152540A1 - 内燃機関用潤滑油組成物及びガソリンエンジンの摩擦低減方法 - Google Patents
内燃機関用潤滑油組成物及びガソリンエンジンの摩擦低減方法 Download PDFInfo
- Publication number
- WO2016152540A1 WO2016152540A1 PCT/JP2016/057465 JP2016057465W WO2016152540A1 WO 2016152540 A1 WO2016152540 A1 WO 2016152540A1 JP 2016057465 W JP2016057465 W JP 2016057465W WO 2016152540 A1 WO2016152540 A1 WO 2016152540A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- lubricating oil
- oil composition
- internal combustion
- combustion engine
- boron
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M169/00—Lubricating compositions characterised by containing as components a mixture of at least two types of ingredient selected from base-materials, thickeners or additives, covered by the preceding groups, each of these compounds being essential
- C10M169/04—Mixtures of base-materials and additives
- C10M169/044—Mixtures of base-materials and additives the additives being a mixture of non-macromolecular and macromolecular compounds
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M133/00—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing nitrogen
- C10M133/02—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing nitrogen having a carbon chain of less than 30 atoms
- C10M133/04—Amines, e.g. polyalkylene polyamines; Quaternary amines
- C10M133/06—Amines, e.g. polyalkylene polyamines; Quaternary amines having amino groups bound to acyclic or cycloaliphatic carbon atoms
- C10M133/08—Amines, e.g. polyalkylene polyamines; Quaternary amines having amino groups bound to acyclic or cycloaliphatic carbon atoms containing hydroxy groups
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M133/00—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing nitrogen
- C10M133/52—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing nitrogen having a carbon chain of 30 or more atoms
- C10M133/54—Amines
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M139/00—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing atoms of elements not provided for in groups C10M127/00 - C10M137/00
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M141/00—Lubricating compositions characterised by the additive being a mixture of two or more compounds covered by more than one of the main groups C10M125/00 - C10M139/00, each of these compounds being essential
- C10M141/12—Lubricating compositions characterised by the additive being a mixture of two or more compounds covered by more than one of the main groups C10M125/00 - C10M139/00, each of these compounds being essential at least one of them being an organic compound containing atoms of elements not provided for in groups C10M141/02 - C10M141/10
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M145/00—Lubricating compositions characterised by the additive being a macromolecular compound containing oxygen
- C10M145/02—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- C10M145/10—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds containing monomers having an unsaturated radical bound to a carboxyl radical, e.g. acrylate
- C10M145/12—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds containing monomers having an unsaturated radical bound to a carboxyl radical, e.g. acrylate monocarboxylic
- C10M145/14—Acrylate; Methacrylate
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M145/00—Lubricating compositions characterised by the additive being a macromolecular compound containing oxygen
- C10M145/18—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- C10M145/24—Polyethers
- C10M145/26—Polyoxyalkylenes
- C10M145/38—Polyoxyalkylenes esterified
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2203/00—Organic non-macromolecular hydrocarbon compounds and hydrocarbon fractions as ingredients in lubricant compositions
- C10M2203/10—Petroleum or coal fractions, e.g. tars, solvents, bitumen
- C10M2203/102—Aliphatic fractions
- C10M2203/1025—Aliphatic fractions used as base material
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2207/00—Organic non-macromolecular hydrocarbon compounds containing hydrogen, carbon and oxygen as ingredients in lubricant compositions
- C10M2207/02—Hydroxy compounds
- C10M2207/023—Hydroxy compounds having hydroxy groups bound to carbon atoms of six-membered aromatic rings
- C10M2207/026—Hydroxy compounds having hydroxy groups bound to carbon atoms of six-membered aromatic rings with tertiary alkyl groups
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2207/00—Organic non-macromolecular hydrocarbon compounds containing hydrogen, carbon and oxygen as ingredients in lubricant compositions
- C10M2207/26—Overbased carboxylic acid salts
- C10M2207/262—Overbased carboxylic acid salts derived from hydroxy substituted aromatic acids, e.g. salicylates
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2209/00—Organic macromolecular compounds containing oxygen as ingredients in lubricant compositions
- C10M2209/02—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- C10M2209/08—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds containing monomers having an unsaturated radical bound to a carboxyl radical, e.g. acrylate type
- C10M2209/084—Acrylate; Methacrylate
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2209/00—Organic macromolecular compounds containing oxygen as ingredients in lubricant compositions
- C10M2209/10—Macromolecular compoundss obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- C10M2209/103—Polyethers, i.e. containing di- or higher polyoxyalkylene groups
- C10M2209/109—Polyethers, i.e. containing di- or higher polyoxyalkylene groups esterified
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant compositions
- C10M2215/02—Amines, e.g. polyalkylene polyamines; Quaternary amines
- C10M2215/04—Amines, e.g. polyalkylene polyamines; Quaternary amines having amino groups bound to acyclic or cycloaliphatic carbon atoms
- C10M2215/042—Amines, e.g. polyalkylene polyamines; Quaternary amines having amino groups bound to acyclic or cycloaliphatic carbon atoms containing hydroxy groups; Alkoxylated derivatives thereof
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant compositions
- C10M2215/086—Imides
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant compositions
- C10M2215/28—Amides; Imides
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2223/00—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions
- C10M2223/02—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions having no phosphorus-to-carbon bonds
- C10M2223/04—Phosphate esters
- C10M2223/045—Metal containing thio derivatives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2010/00—Metal present as such or in compounds
- C10N2010/04—Groups 2 or 12
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2020/00—Specified physical or chemical properties or characteristics, i.e. function, of component of lubricating compositions
- C10N2020/01—Physico-chemical properties
- C10N2020/04—Molecular weight; Molecular weight distribution
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/02—Pour-point; Viscosity index
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/04—Detergent property or dispersant property
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/06—Oiliness; Film-strength; Anti-wear; Resistance to extreme pressure
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/40—Low content or no content compositions
- C10N2030/45—Ash-less or low ash content
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/54—Fuel economy
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/25—Internal-combustion engines
- C10N2040/255—Gasoline engines
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2060/00—Chemical after-treatment of the constituents of the lubricating composition
- C10N2060/14—Chemical after-treatment of the constituents of the lubricating composition by boron or a compound containing boron
Definitions
- the present invention relates to a lubricating oil composition for an internal combustion engine.
- Patent Documents 1 to 5 have been proposed as methods for reducing friction with lubricating oil.
- Patent Document 1 proposes to use a molybdenum-based compound such as MoDTC, which is a typical friction reducing technique, as a friction reducing agent.
- Patent Documents 2 and 3 propose using, as a friction reducing agent, an organic compound having a hydroxyl group or an amino group and a boron-containing compound obtained by heating and stirring boric acid or a boric acid derivative in a high-temperature environment.
- Patent Document 4 proposes a lubricating oil composition in which a nonionic surfactant having an HLB value of 15 or more is blended with a lubricating base oil.
- Patent Document 5 proposes a lubricating oil composition used for a frictional driving force transmission device containing a specific amine compound.
- ashless friction reducing agents such as glycerol monooleate have been conventionally used as a method for obtaining a friction reducing effect in a low temperature region.
- an ashless friction reducing agent cannot obtain a friction reducing effect in a practical temperature range of 80 ° C. or higher.
- a lubricating oil composition for an internal combustion engine is required to have a high friction reducing effect and a low ash differentiation in a practical temperature range of 80 ° C. or higher from a low temperature range at the start of the engine. It is difficult to meet this requirement only by using a molybdenum friction reducer and an ashless friction reducer in combination. Accordingly, there is a demand for a lubricating oil composition using an ashless friction reducing agent that can provide a sufficient friction reducing effect even in a practical temperature range of 80 ° C. or higher from a low temperature range when the engine is started.
- an embodiment of the present invention provides a lubricating oil composition for an internal combustion engine comprising a surfactant having an alkylene oxide as a structural unit and having an HLB value of 7 or more and less than 15, and a lubricating base oil.
- the lubricating oil composition for an internal combustion engine of the present invention can improve the friction reduction effect in a practical temperature range of 80 ° C. or higher from a low temperature range assumed at the time of engine start, and thus improve fuel efficiency.
- the lubricating oil composition for an internal combustion engine includes a surfactant having an alkylene oxide as a constituent unit and having an HLB value of 7 or more and less than 15, and a lubricating base oil.
- the internal combustion engine lubricating oil composition of the present embodiment includes a surfactant having an alkylene oxide as a structural unit and having an HLB value of 7 or more and less than 15. Even if the surfactant has an HLB value in the above range, those having no alkylene oxide in the structural unit tend to have insufficient friction reduction. Moreover, even if it is a surfactant which has an alkylene oxide in a structural unit, the thing with an HLB value of less than 7 has bad adsorbability with respect to a metal surface, and friction reduction is inadequate. Moreover, even if it is surfactant which has an alkylene oxide in a structural unit, a thing with HLB value more than 15 is bad in solubility to a lubricating base oil, and it is very difficult to use it.
- the surfactant various surfactants can be used. From the viewpoint of reducing friction due to adsorption to the metal surface and stability, an amine compound or amide formed by bonding an alkylene oxide to a nitrogen atom. System compounds and the like are preferred. Among these, amine compounds are preferable, and tertiary amines are preferable among amine compounds. Examples of the tertiary amine surfactant include compounds represented by the following general formula (I). The compound represented by the general formula (I) is preferable in that the ash content is 0% by mass while having the above effects.
- R 1 and R 2 each independently represents an alkyl group having 4 to 18 carbon atoms or an alkenyl group having 4 to 18 carbon atoms.
- x represents 0 or 1
- a 1 O and A 2 O each independently represents an oxyalkylene group having 2 to 4 carbon atoms.
- n 1 and n 2 represent the average number of added moles of the oxyalkylene group, each independently represents an integer of 1 to 13, and n 1 + n 2 is 5 to 14.
- the alkyl group and alkenyl group of R 1 and R 2 may be linear, branched or cyclic, but is preferably linear.
- R 1 and R 2 are preferably alkenyl groups.
- x 0, the carbon number of the alkyl group and alkenyl group of R 1 is preferably 12-18.
- the oxyalkylene group of A 1 O and A 2 O preferably has 2 to 3 carbon atoms, and more preferably 2 carbon atoms.
- N 1 and n 2 are each independently preferably an integer of 2 to 10, more preferably 3 to 7.
- n 1 + n 2 is preferably 8 to 12, and more preferably 9 to 11.
- (A 1 O) n1 and (A 2 O) n2 may be formed by bonding oxyalkylene groups having different carbon numbers randomly or in blocks.
- (A 1 O) n1 and (A 2 O) n2 may be ones in which an ethylene oxide (EO) group and a propylene oxide (PO) group are bonded randomly or in blocks.
- EO ethylene oxide
- PO propylene oxide
- the same type may be used, or different types may be mixed and used.
- the same species refers to those in which R 1 , R 2 and the like in the general formula (I) are all the same.
- the term “heterologous” refers to a substance in which at least one of R 1 and R 2 in the general formula (I) is different. Moreover, even when different types are mixed, it is preferable to include many of the preferred embodiments.
- the ratio of the mass of the tertiary amine in which R 1 and R 2 are alkenyl groups to the total mass of the tertiary amine of the above general formula (I) [general formula in which R 1 and R 2 are alkenyl groups
- the mass of the tertiary amine of (I) / the total mass of the general formula (I) tertiary amine] is preferably 60% by mass or more, more preferably 70% by mass or more, and 80% by mass or more. More preferably it is. From the viewpoint of the stability of the effect, it is preferable to use the tertiary amine of the general formula (I) having the same carbon number for R 1 . If a tertiary amine of general formula (I) contain an R 2, it is preferable to further the number of carbon atoms in R 2 is used as all the same.
- polyoxyalkylene fatty acid esters are also suitable.
- the number of carbon atoms in the oxyalkylene group of the polyoxyalkylene fatty acid ester is preferably 2 to 4, more preferably 2 to 3, and still more preferably 2.
- the oxyalkylene group may be formed by bonding oxyalkylene groups having different carbon numbers randomly or in blocks.
- the average number of added moles of the oxyalkylene group is preferably an integer of 2 to 10, and more preferably 3 to 7.
- the structural unit derived from the fatty acid of the polyoxyalkylene fatty acid ester preferably has 8 to 28 carbon atoms, more preferably 14 to 22 carbon atoms, and still more preferably 16 to 20 carbon atoms. Examples of such polyoxyalkylene fatty acid esters include polyoxyethylene oleate and polyoxyethylene stearate.
- the surfactant preferably has a molecular weight in the range of 350 to 950 g / mol from the viewpoint of friction reduction and compatibility between friction reduction and cleanliness, and preferably in the range of 440 to 940 g / mol. Is more preferable.
- the molecular weight of the surfactant is measured by a mass spectrum by liquid chromatography mass spectrometry (LC / MS). Specifically, the range in which the peak of the mass-to-charge ratio (m / z) of the surfactant appears was regarded as the range of the molecular weight (g / mol) of the surfactant.
- the surfactant preferably has an ash content of 0% by mass.
- the surfactant is preferably contained in the lubricating oil composition for an internal combustion machine in an amount of 0.01 to 2.0% by mass, more preferably 0.1 to 1.5% by mass, More preferably, it is contained in an amount of ⁇ 1.0% by mass.
- the lubricating oil composition for an internal combustion machine of the present embodiment preferably further contains a boron-modified product of succinimide.
- a boron-modified succinimide together with the above surfactant, friction can be further reduced and cleanliness is improved in a practical temperature range of 80 ° C. or higher from a low temperature range assumed when the engine is started. be able to.
- Examples of the boron-modified succinimide include boronated alkenyl or alkyl succinic monoimide, or alkenyl or alkyl succinic bisimide.
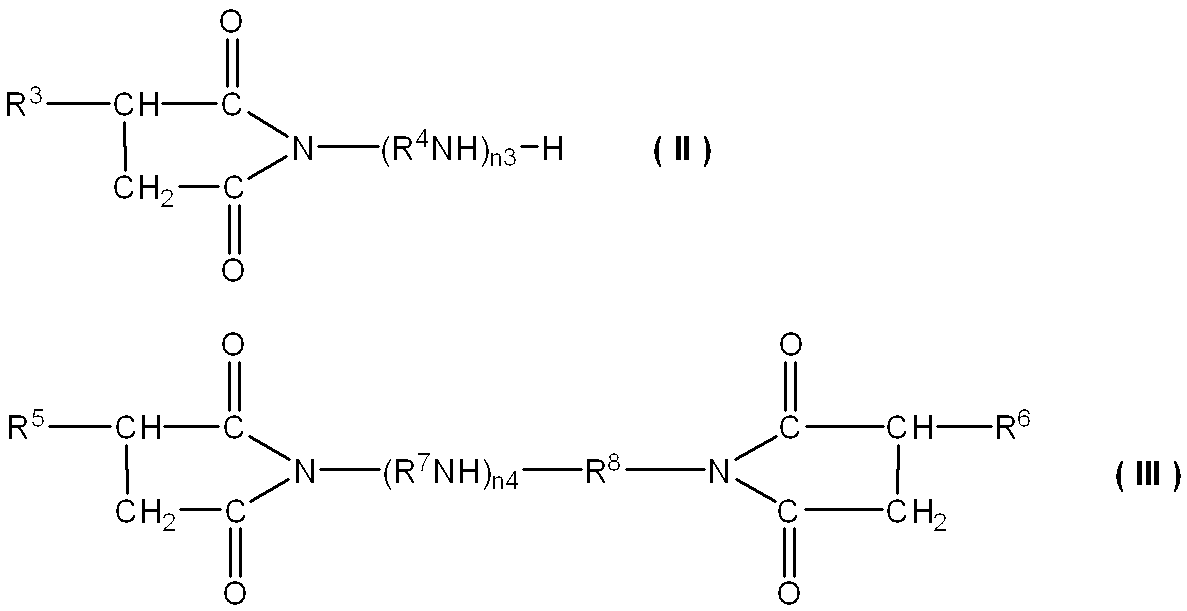
- Examples of the alkenyl or alkyl succinic acid monoimide include compounds represented by the following general formula (II).
- Examples of the alkenyl or alkyl succinic acid bisimide include compounds represented by the following general formula (III).
- R 3 , R 5 and R 6 are alkenyl groups or alkyl groups, and each have a weight average molecular weight of preferably 500 to 3,000, more preferably 1, 000 to 3,000.
- the weight average molecular weight of R 3 , R 5 and R 6 is 500 or more, the solubility in the lubricating base oil can be improved.
- R 5 and R 6 may be the same or different.
- R 4 , R 7 and R 8 are each an alkylene group having 2 to 5 carbon atoms, and R 7 and R 8 may be the same or different.
- n3 represents an integer of 1 to 10, and n4 represents 0 or an integer of 1 to 10.
- n3 is preferably 2 to 5, more preferably 2 to 4.
- n4 is preferably 1 to 6, more preferably 2 to 6.
- n4 is 1 or more, it is expected that the effect obtained by the present compound is appropriately exhibited.
- n4 is 6 or less, the solubility in the lubricating base oil is further improved.
- alkenyl group examples include a polybutenyl group, a polyisobutenyl group, and an ethylene-propylene copolymer
- examples of the alkyl group include hydrogenated groups thereof.
- Suitable alkenyl groups include polybutenyl or polyisobutenyl groups.
- the polybutenyl group a mixture of 1-butene and isobutene or a polymer obtained by polymerizing high-purity isobutene is preferably used.
- a representative example of a suitable alkyl group is a hydrogenated polybutenyl group or polyisobutenyl group.
- the boron-modified succinimide is obtained by, for example, reacting a polyolefin with maleic anhydride to obtain an alkenyl succinic anhydride (A), and further reacting a polyamine with a boron compound to obtain an intermediate (B).
- An intermediate (B) can be obtained by reacting alkenyl succinic anhydride (A) with intermediate (B) and imidizing.
- Monoimides or bisimides can be made by changing the ratio of alkenyl succinic anhydride or alkyl succinic anhydride to polyamine.
- the boron modified body of the said succinimide can be manufactured also by processing the alkenyl or alkyl succinic monoimide which does not contain boron, or an alkenyl or alkyl succinic acid bisimide with a boron compound.
- ⁇ -olefins having 2 to 8 carbon atoms can be mixed and used, but a mixture of isobutene and 1-butene is preferably used. be able to.
- polyamines include ethylenediamine, propylenediamine, butylenediamine, pentylenediamine, and other single diamines, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, pentaethylenehexamine, di (methylethylene) triamine, dibutylenetriamine, triethylene
- polyalkylene polyamines such as butylenetetramine and pentapentylenehexamine, and piperazine derivatives such as aminoethylpiperazine.
- Examples of the boron compound include boric acid, borates, and borate esters.
- Examples of boric acid include orthoboric acid, metaboric acid, and paraboric acid.
- Examples of the borate include ammonium borate such as ammonium metaborate, ammonium tetraborate, ammonium pentaborate, and ammonium octaborate.
- As borate esters monomethyl borate, dimethyl borate, trimethyl borate, monoethyl borate, diethyl borate, triethyl borate, monopropyl borate, dipropyl borate, tripropyl borate, monobutyl borate, Examples thereof include dibutyl borate and tributyl borate.
- the ratio of boron atom weight to nitrogen atom weight (B / N ratio) contained in the boron-modified succinimide is preferably 0.6 or more, and 0.7 or more. More preferably, it is 0.8 or more.
- the B / N ratio is not particularly limited, but is preferably 2.0 or less, more preferably 1.5 or less, and further preferably 1.3 or less.
- the boron-modified product of the succinimide preferably contains a large amount of tri-coordinated boron-modified product of succinimide, and specifically, a boron-modified product of tri-coordinated succinimide. Is preferably contained in a molar ratio of 0.50 or more, more preferably 0.60 or more, based on the total amount of boron-modified succinimide having three and four coordinates. More preferably, it is 65 or more.
- the ratio of the boron-modified 3-coordinate succinimide and the boron-modified 4-coordinated succinimide can be measured, for example, by 11 B-NMR measurement as a BF 3 ⁇ OEt 2 standard (0 ppm).
- 11 B-NMR measurement the peak of the boron-modified product of tricoordinate succinimide appears at 5 to 25 ppm, and the peak of the boron-modified product of tetracoordinate succinimide appears at ⁇ 10 to 5 ppm. Therefore, it is possible to calculate the ratio by calculating the integral value of each peak.
- the content of the boron-modified succinimide is preferably 0.1 to 15.0% by mass, and 0.2 to 10.0% by mass in the lubricating oil composition for an internal combustion engine. Is more preferably 0.5 to 5.0% by mass, and further preferably 0.5 to 2.0% by mass.
- the boron-modified content of the boron-modified succinimide is preferably 0.2% by mass or less in the lubricating oil composition for an internal combustion engine, and is 0.001 to 0.00. It is more preferably 05% by mass, and further preferably 0.005 to 0.03% by mass.
- the friction can be further reduced in a practical temperature range of 80 ° C. or higher from a low temperature range assumed at the time of engine start, and clean. It is easy to make the property better.
- the mass ratio of the surfactant content to the boron-modified product content of the succinimide is 100 or less. Preferably, it is preferably 20 or less, and more preferably 5 or less.
- the content of the surfactant relative to the content in terms of boron atom of the boron-modified product of the succinimide (the content in terms of boron atom of the boron-modified product of the succinimide / the surface activity).
- the mass ratio of the content of the agent is preferably 1 or less, more preferably 0.2 or less, and even more preferably 0.05 or less.
- the lubricating oil composition for an internal combustion machine of the present embodiment preferably further contains poly (meth) acrylate as a viscosity index improver.
- poly (meth) acrylate in addition to the improvement in fuel efficiency due to the friction reduction of the surfactant and the boron-modified succinimide, the fuel efficiency can be further improved.
- the monomer constituting the poly (meth) acrylate is an alkyl (meth) acrylate, and preferably a linear alkyl group having 1 to 18 carbon atoms or an alkyl (meth) acrylate having a branched alkyl group having 3 to 34 carbon atoms.
- alkyl (meth) acrylate for example, methyl (meth) acrylate, ethyl (meth) acrylate, propyl (meth) acrylate, butyl (meth) acrylate, pentyl (meth) acrylate, hexyl (meth) acrylate, Hexyl (meth) acrylate, heptyl (meth) acrylate, octyl (meth) acrylate, nonyl (meth) acrylate, decyl (meth) acrylate, dodecyl (meth) acrylate, tetra (meth) acrylate, hexa (meth) acrylate, octadecyl ( (Meth) acrylate and the like.
- the alkyl group of these monomers may be linear or branched.
- Examples of the alkyl (meth) acrylate having a branched alkyl group having 3 to 34 carbon atoms include isopropyl (meth) acrylate, 2-ethylhexyl (meth) acrylate, 3,5,5-trimethylhexyl (meth) acrylate, 2- Butyloctyl (meth) acrylate, 2-hexyldecyl (meth) acrylate, 2-octyldodecyl (meth) acrylate, 2-decyltetradecyl (meth) acrylate, 2-dodecylhexadecyl (meth) acrylate, 2-tetradecyloctadecyl (Meth) acrylate is mentioned.
- the poly (meth) acrylate preferably has a weight average molecular weight of 100,000 to 600,000, more preferably 15,000 to 300,000.
- weight average molecular weight refers to a molecular weight in terms of polystyrene determined by gel permeation chromatography (GPC) measurement.
- the poly (meth) acrylate preferably has an SSI of 30% or less, more preferably 1 to 28%. By making the said weight average molecular weight into the said range, SSI can be 30% or less.
- SSI means the shear stability index (Shear Stability Index), and indicates the ability to resist the decomposition of poly (meth) acrylate. The higher the SSI, the more unstable the polymer is to shear and the easier it is to degrade.
- Kv 0 is a value of 100 ° C. kinematic viscosity of a mixture obtained by adding poly (meth) acrylate to base oil.
- Kv 1 is the value of 100 ° C. kinematic viscosity after passing a mixture of base oil plus poly (meth) acrylate through a 30 cycle high shear Bosch diesel injector according to the procedure of ASTM D6278.
- Kv oil is the value of the 100 ° C. kinematic viscosity of the base oil.
- a base oil a Group II base oil having a kinematic viscosity of 100 ° C. of 5.35 mm 2 / s and a viscosity index of 105 is used.
- the content of the poly (meth) acrylate is preferably 1 to 15% by mass, more preferably 2 to 10% by mass in the lubricating oil composition for an internal combustion engine from the viewpoint of fuel economy. More preferably, it is 8 mass%.
- the lubricating oil composition for an internal combustion machine of the present embodiment preferably further contains a molybdenum compound from the viewpoint of reducing friction.
- the molybdenum compound include MoDTC (molybdenum dialkyldithiocarbamate) and MoDTP (molybdenum dialkyldithiophosphate).
- the content of the molybdenum compound is preferably 2.0% by mass or less, more preferably 0.1 to 1.0% by mass in the lubricating oil composition for internal combustion machines.
- ⁇ Lubricant base oil> examples of the lubricating base oil include mineral oil and / or synthetic oil.
- Mineral oils include paraffin-based mineral oils, intermediate-based mineral oils and naphthenic-based mineral oils obtained by ordinary refining methods such as solvent refining and hydrogenation refining; wax produced by the Fischer-Tropsch process (gas-tri-liquid wax) And wax isomerized oil produced by isomerizing wax such as mineral oil-based wax.
- synthetic oils include hydrocarbon synthetic oils and ether synthetic oils.
- hydrocarbon-based synthetic oil examples include polybutene, polyisobutylene, 1-octene oligomer, 1-decene oligomer, ⁇ -olefin oligomer such as ethylene-propylene copolymer or the hydride thereof, alkylbenzene, alkylnaphthalene and the like.
- ether synthetic oils include polyoxyalkylene glycol and polyphenyl ether.
- the lubricating base oil may be a single system using one of the above-mentioned mineral oils and synthetic oils, but is a mixture of two or more mineral oils or a mixture of two or more synthetic oils.
- a mixed system may be used, such as a mixture of one or more of mineral oil and synthetic oil.
- the lubricating base oil it is preferable to use one or more selected from mineral oils or synthetic oils classified into Group 3 and Group 4 in the base oil classification of the American Petroleum Institute.
- the content of the lubricating base oil is preferably 70% by mass or more and less than 100% by mass, more preferably 75% by mass or more and 95% by mass or less, and more preferably 80% by mass or more in the lubricating oil composition for an internal combustion engine. More preferably, it is 90 mass% or less.
- the internal combustion engine lubricating oil composition of the present embodiment may contain additives such as metal detergents, antioxidants, and antiwear agents.
- the content of these additives is preferably 0.01 to 5% by mass with respect to the total amount of the lubricating oil composition for an internal combustion machine.
- the lubricating oil composition for an internal combustion engine of the present embodiment has a 40 ° C. kinematic viscosity, a 100 ° C. kinematic viscosity, and a 150 ° C. HTHS viscosity from the viewpoint of reducing friction in a practical temperature range of 80 ° C. or higher from a low temperature range assuming engine starting.
- the 40 ° C. kinematic viscosity is preferably 20 to 40 mm 2 / s, and more preferably 30 to 35 mm 2 / s. 100 ° C.
- kinematic viscosity is preferably 3.0 ⁇ 12.5mm 2 / s, and more preferably 4.0 ⁇ 9.3mm 2 / s.
- the 150 ° C. HTHS viscosity is preferably 1.4 to 2.9 mPa ⁇ s, and more preferably 1.7 to 2.9 mPa ⁇ s.
- the kinematic viscosity was measured according to JIS K2283.
- the HTHS viscosity was measured according to ASTM D4683 using a TBS viscometer (Tapered Bearing Simulator Viscometer), oil temperature 100 ° C., shear rate 10 6 / s, rotation speed (motor) 3000 rpm, interval (rotor and stator The interval was measured under the condition of 3 ⁇ m.
- the lubricating oil composition for an internal combustion engine of the present embodiment can be suitably used for various internal combustion engines such as a four-wheeled vehicle and a two-wheeled vehicle. Moreover, among internal combustion engines, it can be particularly suitably used for gasoline engines.
- the internal combustion engine friction reducing method of the present embodiment is a method in which the above-described lubricating oil composition for an internal combustion engine of the present embodiment is added to the internal combustion engine.
- the friction reduction effect can be improved in a practical temperature range of 80 ° C. or higher from a low temperature range assumed at the time of engine start, and thus fuel economy is improved. Can be.
- the internal combustion engine is a gasoline engine, the effect can be made particularly good.
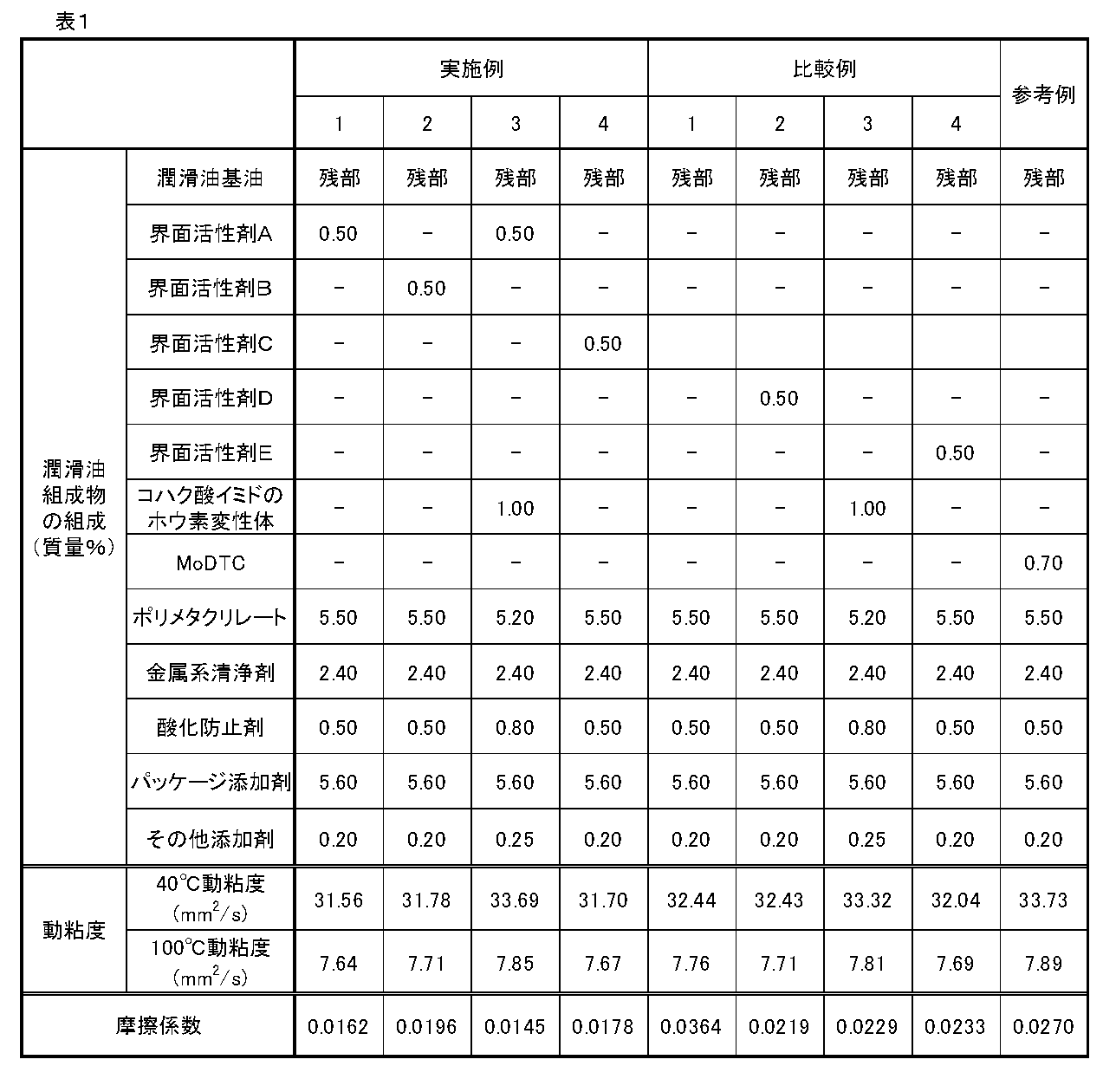
- Lubricating Oil Composition for Internal Combustion Machines Lubricating oil compositions for internal combustion machines of Examples, Comparative Examples, and Reference Examples were prepared at the composition ratios shown in Table 1.
- the lubricating oil compositions for internal combustion engines of the examples, comparative examples, and reference examples are all prepared by adjusting the HTHS viscosity at 150 ° C. to 2.6 mPa ⁇ s.
- HLB value 11.7. Peak appearance position of mass spectrum by liquid chromatography mass spectrometry: 400 to 850 m / z ( ⁇ molecular weight range: 400 to 850 g / mol).
- the lubricating oil compositions for internal combustion machines of Examples 1 to 4 are excellent in the friction reducing effect in a practical temperature range of 80 ° C. or higher while using an ashless friction reducing agent.
- the lubricating oil composition for an internal combustion engine of Example 3 comprising a combination of a surfactant having an alkylene oxide as a constituent unit and having an HLB value of 7 or more and less than 15 and a boron-modified succinimide, It can be confirmed that the effect is extremely excellent.
- the lubricating oil for an internal combustion engine of Example 3 comprising a combination of a surfactant having an alkylene oxide as a constituent unit and having an HLB value of 7 or more and less than 15 and a boron-modified succinimide. It can be confirmed that the composition can be further improved in cleanliness.
- the lubricating oil composition for an internal combustion engine of the present embodiment utilizes the characteristics that friction can be reduced and fuel economy can be improved in a practical temperature range of 80 ° C. or higher from a low temperature range assumed when the engine is started. It can be suitably used for various internal combustion engines such as a wheeled vehicle and a two-wheeled vehicle. Moreover, among internal combustion engines, it can be particularly suitably used for gasoline engines.
Abstract
Description
潤滑油による摩擦低減の手法としては、特許文献1~5が提案されている。
特許文献2及び3は、ヒドロキシル基又はアミノ基を有する有機化合物と、ホウ酸又はホウ酸誘導体を高温環境下で加熱攪拌して得られるホウ素含有化合物を摩擦低減剤として用いることを提案している。
特許文献4は、潤滑油基油に、HLB値が15以上である非イオン系界面活性剤を配合した潤滑油組成物を提案している。
特許文献5は、特定のアミン化合物を含有してなる摩擦式駆動力伝達装置に用いる潤滑油組成物を提案している。
しかし、特許文献1のように、モリブデン系摩擦低減剤を配合したのみでは、エンジン始動時等の低温領域では摩擦低減効果が得られず、省燃費性を十分に向上することができないものであった。
特許文献2~5に提案されている摩擦低減剤及び潤滑油組成物は、低温領域での摩擦低減については検討されていない。
現在、内燃機関用の潤滑油組成物には、エンジン始動時の低温領域から80℃以上の実用温度領域における高い摩擦低減効果と、低灰分化が要求されている。モリブデン系摩擦低減剤と無灰系摩擦低減剤とを併用するだけでは、該要求への対応が困難になっている。
そこで、エンジン始動時の低温領域から80℃以上の実用温度領域でも十分な摩擦低減効果が得られる無灰摩擦低減剤を用いた潤滑油組成物が求められている。
[内燃機用潤滑油組成物]
本実施形態の内燃機用潤滑油組成物は、アルキレンオキサイドを構成単位に有し、かつHLB値7以上15未満である界面活性剤、及び潤滑油基油を含むものである。
本実施形態の内燃機用潤滑油組成物は、アルキレンオキサイドを構成単位に有し、かつHLB値7以上15未満である界面活性剤を含む。
HLB値が上記範囲である界面活性剤であっても、アルキレンオキサイドを構成単位に有さないものは、摩擦低減が不十分な傾向にある。また、アルキレンオキサイドを構成単位に有する界面活性剤であっても、HLB値7未満のものは金属表面に対する吸着性が悪く、摩擦低減が不十分である。また、アルキレンオキサイドを構成単位に有する界面活性剤であっても、HLB値15超のものは潤滑油基油への溶解性が悪く、極めて使いにくい。
第3級アミンの界面活性剤としては、以下の一般式(I)に示す化合物が挙げられる。一般式(I)に示す化合物は、上記効果を有しつつ、灰分が0質量%である点で好適である。
(式中、R1及びR2はそれぞれ独立に、炭素数4~18のアルキル基、又は炭素数4~18のアルケニル基を示す。また、式中、xは0又は1を示し、かつx=0の時にyは1を示し、x=1の時にyは0を示す。また、式中、A1O及びA2Oはそれぞれ独立に、炭素数2~4のオキシアルキレン基を示す。また、式中、n1及びn2はオキシアルキレン基の平均付加モル数を示し、それぞれ独立に1~13の整数を示し、かつn1+n2は5~14である。)
また、n1及びn2は、それぞれ独立に2~10の整数であることが好ましく、3~7であることがより好ましい。n1+n2は8~12であることが好ましく、9~11であることがさらに好ましい。
また、異種のものを混合する場合でも、好適な実施態様のものを多く含むことが好ましい。例えば、上記一般式(I)の第3級アミンの全質量に対して、R1及びR2がアルケニル基である第3アミンの質量の割合[R1及びR2がアルケニル基である一般式(I)の第3アミンの質量/一般式(I)第3級アミンの全質量]が60質量%以上であることが好ましく、70質量%以上であることがより好ましく、80質量%以上であることがさらに好ましい。
また、効果の安定性の観点からは、一般式(I)の第3級アミンとして、R1の炭素数が全て同一のものを用いることが好ましい。一般式(I)の第3級アミンがR2を含む場合、さらにR2の炭素数が全て同一のものを用いることが好ましい。
ポリオキシアルキレン脂肪酸エステルのオキシアルキレン基の炭素数は、2~4であることが好ましく、2~3であることがより好ましく、2であることがさらに好ましい。該オキシアルキレン基は、炭素数の異なるオキシアルキレン基がランダム又はブロックに結合してなるものであってもよい。また、オキシアルキレン基の平均付加モル数は2~10の整数であることが好ましく、3~7であることがより好ましい。
ポリオキシアルキレン脂肪酸エステルの脂肪酸に由来する構成単位の炭素数は、8~28であることが好ましく、14~22であることがより好ましく、16~20であることがさらに好ましい。
このようなポリオキシアルキレン脂肪酸エステルとしては、ポリオキシエチレンオレイン酸エステル、ポリオキシエチレンステアリン酸エステル等が挙げられる。
なお、本実施形態において、界面活性剤の分子量は、液体クロマトグラフ質量分析法(LC/MS)による質量スペクトルにより測定したものである。具体的には、界面活性剤の質量電荷比(m/z)のピークが現れる範囲を、界面活性剤の分子量(g/mol)の範囲とみなした。
また、上記界面活性剤は、灰分が0質量%であることが好ましい。
上記界面活性剤の含有量を0.01質量%以上とすることにより、エンジン始動時を想定した低温領域から、80℃以上の実用温度領域において摩擦を低減することができる。また、界面活性剤の含有量を2.0質量%以下とすることにより、摩擦低減を維持するとともに、清浄性の低下を抑制しやすくできる。
本実施形態の内燃機用潤滑油組成物は、さらに、コハク酸イミドのホウ素変性体を含むことが好ましい。
上記界面活性剤とともにコハク酸イミドのホウ素変性体を含有することにより、エンジン始動時を想定した低温領域から、80℃以上の実用温度領域において、摩擦をより低減できるとともに、清浄性を良好にすることができる。
アルケニル又はアルキルコハク酸モノイミドとしては、下記一般式(II)で示される化合物が挙げられる。また、アルケニル又はアルキルコハク酸ビスイミドとしては、下記一般式(III)で示される化合物が挙げられる。
R3、R5及びR6の重量平均分子量が500以上であると、潤滑油基油への溶解性を良好にできる。また、3,000以下であると、本化合物により得られる効果を適切に発揮することが期待される。R5及びR6は同一でも異なっていてもよい。
R4、R7及びR8は、それぞれ炭素数2~5のアルキレン基であり、R7及びR8は同一でも異なっていてもよい。n3は1~10の整数を示し、n4は0又は1~10の整数を示す。ここで、n3は、好ましくは2~5、より好ましくは2~4である。n3が2以上であると、コハク酸イミドのホウ素変性体により得られる効果が得やすくなることが期待される。n3が5以下であると、潤滑油基油に対する溶解性がより一層良好となる。
一般式(III)において、n4は好ましくは1~6であり、より好ましくは2~6である。n4が1以上であると、本化合物により得られる効果を適切に発揮することが期待される。n4が6以下であると、潤滑油基油に対する溶解性がより一層良好となる。
また、上記コハク酸イミドのホウ素変性体は、ホウ素未含有のアルケニル又はアルキルコハク酸モノイミドや、アルケニル又はアルキルコハク酸ビスイミドを、ホウ素化合物で処理しても製造できる。
一方、ポリアミンとしては、エチレンジアミン、プロピレンジアミン、ブチレンジアミン、ペンチレンジアミン等の単一ジアミン、ジエチレントリアミン、トリエチレンテトラミン、テトラエチレンペンタミン、ペンタエチレンヘキサミン、ジ(メチルエチレン)トリアミン、ジブチレントリアミン、トリブチレンテトラミン、及びペンタペンチレンヘキサミン等のポリアルキレンポリアミン、アミノエチルピペラジン等のピペラジン誘導体を挙げることができる。
ホウ酸としては、オルトホウ酸、メタホウ酸及びパラホウ酸等が挙げられる。また、ホウ酸塩としては、メタホウ酸アンモニウム、四ホウ酸アンモニウム、五ホウ酸アンモニウム及び八ホウ酸アンモニウム等のホウ酸アンモニウム等が挙げられる。また、ホウ酸エステルとしては、ホウ酸モノメチル、ホウ酸ジメチル、ホウ酸トリメチル、ホウ酸モノエチル、ホウ酸ジエチル、ホウ酸トリエチル、ホウ酸モノプロピル、ホウ酸ジプロピル、ホウ酸トリプロピル、ホウ酸モノブチル、ホウ酸ジブチル及びホウ酸トリブチル等が挙げられる。
3配位のコハク酸イミドのホウ素変性体及び4配位のコハク酸イミドのホウ素変性体の割合は、例えば、BF3・OEt2標準(0ppm)として11B-NMR測定により測定可能である。この11B-NMR測定では、3配位のコハク酸イミドのホウ素変性体のピークが5~25ppmに出現し、4配位のコハク酸イミドのホウ素変性体のピークが-10~5ppmに出現するため、各ピークの積分値を算出することにより上記割合を算出することが可能である。
本実施形態の内燃機用潤滑油組成物は、さらに、粘度指数向上剤として、ポリ(メタ)アクリレートを含有することが好ましい。
ポリ(メタ)アクリレートを含有することにより、上記界面活性剤及び上記コハク酸イミドのホウ素変性体の摩擦低減による省燃費性の向上に加えて、さらに省燃費性を向上することができる。
アルキル(メタ)アクリレートを構成する好ましいモノマーとして、例えば、メチル(メタ)アクリレート、エチル(メタ)アクリレート、プロピル(メタ)アクリレート、ブチル(メタ)アクリレート、ペンチル(メタ)アクリレート、ヘキシル(メタ)アクリレート、ヘキシル(メタ)アクリレート、ヘプチル(メタ)アクリレート、オクチル(メタ)アクリレート、ノニル(メタ)アクリレート、デシル(メタ)アクリレート、ドデシル(メタ)アクリレート、テトラ(メタ)アクリレート、ヘキサ(メタ)アクリレート、オクタデシル(メタ)アクリレートなどが挙げられ、これらモノマーを2種類以上使用してコポリマーとしてもよい。これらモノマーのアルキル基は直鎖状でもよいし、分岐鎖状のものでもよい。
また、炭素数3~34の分岐アルキル基を有するアルキル(メタ)アクリレートとしては、イソプロピル(メタ)アクリレート、2-エチルヘキシル(メタ)アクリレート、3,5,5-トリメチルヘキシル(メタ)アクリレート、2-ブチルオクチル(メタ)アクリレート、2-ヘキシルデシル(メタ)アクリレート、2-オクチルドデシル(メタ)アクリレート、2-デシルテトラデシル(メタ)アクリレート、2-ドデシルヘキサデシル(メタ)アクリレート、2-テトラデシルオクタデシル(メタ)アクリレートが挙げられる。
なお、本実施形態において「重量平均分子量」は、ゲルパーミエーションクロマトグラフィー(GPC)測定によって求めたポリスチレン換算の分子量をいうものとする。
ここで、SSIとは、せん断安定性指数(Shear Stability Index)を意味し、ポリ(メタ)アクリレートの分解に抵抗する能力を示す。SSIが大きいほど、ポリマーはせん断に対して不安定で、より分解されやすい。
SSIは、ポリマーに由来するせん断による粘度低下をパーセンテージで示すもので、上記計算式により算出される。式中、Kv0は、基油にポリ(メタ)アクリレートを加えた混合物の100℃動粘度の値である。Kv1は、基油にポリ(メタ)アクリレートを加えた混合物を、ASTM D6278の手順にしたがって、30サイクル高剪断ボッシュ・ディーゼルインジェクターに通過させた後の100℃動粘度の値である。また、Kvoilは、基油の100℃動粘度の値である。なお、基油としては、100℃動粘度5.35mm2/s、粘度指数105のGroup II基油を使用する。
本実施形態の内燃機用潤滑油組成物は、摩擦低減の観点から、さらにモリブデン化合物を含有することが好ましい。
モリブデン化合物としては、MoDTC(ジアルキルジチオカルバミン酸モリブデン)、MoDTP(ジアルキルジチオリン酸モリブデン)等が挙げられる。
モリブデン化合物の含有量は、内燃機用潤滑油組成物中の2.0質量%以下であることが好ましく、0.1~1.0質量%であることがより好ましい。
潤滑油基油としては、鉱油及び/又は合成油が挙げられる。
鉱油としては、溶剤精製、水添精製等の通常の精製法により得られるパラフィン基系鉱油、中間基系鉱油及びナフテン基系鉱油等;フィッシャートロプシュプロセス等により製造されるワックス(ガストゥリキッドワックス)、鉱油系ワックス等のワックスを異性化することによって製造されるワックス異性化系油等が挙げられる。
合成油としては、炭化水素系合成油、エーテル系合成油等が挙げられる。炭化水素系合成油としては、ポリブテン、ポリイソブチレン、1-オクテンオリゴマー、1-デセンオリゴマー、エチレン-プロピレン共重合体等のα-オレフィンオリゴマー又はその水素化物、アルキルベンゼン、アルキルナフタレン等を挙げることができる。エーテル系合成油としては、ポリオキシアルキレングリコール、ポリフェニルエーテル等が挙げられる。
特に、潤滑油基油としては、米国石油協会の基油分類において、グループ3及びグループ4に分類される鉱油又は合成油から選ばれる1種以上を用いることが好ましい。
本実施形態の内燃機用潤滑油組成物は、金属系清浄剤、酸化防止剤、摩耗防止剤等の添加剤を含有してもよい。
これら添加剤の含有割合は、内燃機用潤滑油組成物の全量に対して、それぞれ0.01~5質量%であることが好ましい。
本実施形態の内燃機用潤滑油組成物は、エンジン始動時を想定した低温領域から、80℃以上の実用温度領域における摩擦低減の観点から、40℃動粘度、100℃動粘度及び150℃HTHS粘度が、それぞれ以下の範囲であることが好ましい。
40℃動粘度は、20~40mm2/sであることが好ましく、30~35mm2/sであることがより好ましい。
100℃動粘度は、3.0~12.5mm2/sであることが好ましく、4.0~9.3mm2/sであることがより好ましい。
150℃HTHS粘度は、1.4~2.9mPa・sであることが好ましく、1.7~2.9mPa・sであることがより好ましい。
なお、動粘度はJIS K2283に準拠して測定した。また、HTHS粘度は、ASTMD4683に準拠して、TBS粘度計(Tapered Bearing Simulator Viscometer)を用い、油温100℃、せん断速度106/s、回転数(モーター)3000rpm、間隔(ローターとステーターとの間隔)3μmの条件で測定した。
本実施形態の内燃機関用潤滑油組成物は、四輪自動車、二輪自動車等の各種の内燃機関用に好適に使用できる。また、内燃機関の中でも、ガソリンエンジン用に特に好適に使用できる。
本実施形態の内燃機関の摩擦低減方法は、内燃機関に、上述した本実施形態の内燃機用潤滑油組成物を添加するものである。
本実施形態の内燃機関の摩擦低減方法によれば、エンジン始動時を想定した低温領域から、80℃以上の実用温度領域において、摩擦低減効果を良好にすることができ、ひいては省燃費性を良好にできる。内燃機関がガソリンエンジンである場合、前記効果を特に良好にできる。
表1の組成比で実施例、比較例及び参考例の内燃機用潤滑油組成物を調製した。なお、実施例、比較例及び参考例の内燃機関用潤滑油組成物は、全て、150℃におけるHTHS粘度を2.6mPa・sに調整したものである。
実施例、比較例及び参考例の内燃機関用潤滑油組成物について、以下の測定及び評価を行った。結果を表1に示す。
2-1.動粘度
明細書本文の記載に従い、内燃機用潤滑油組成物の40℃動粘度及び100℃動粘度を測定した。
以下の条件で内燃機用潤滑油組成物の摩擦係数を測定した。
試験機:MTM(Mini Traction Machine)試験機、PCS Instruments社製
試験片:標準テストピース
ラビング(ならし)時間:2時間
油温:80℃
荷重:10N
測定速度:1.2m/s
<潤滑油基油>
100℃動粘度が4.07mm2/sの鉱油、粘度指数:131、%CA:-0.4、%CN:12.8、%CP:87.6
<界面活性剤A>
一般式(I)の第3級アミン(x=0、n1+n2=10、A1O及びA2Oの炭素数:2、R1:オレイル基)69質量%と、一般式(I)の第3級アミン(x=0、n1+n2=10、A1O及びA2Oの炭素数:2、R1:ステアリル基)31質量%との混合物(質量割合は液体クロマトグラフ質量分析法により測定)。HLB値:13.2。液体クロマトグラフ質量分析法による質量スペクトルのピーク出現位置:440~940m/z(≒分子量の範囲:440~940g/mol)。
なお、液体クロマトグラフ質量分析は、次の条件で行った。
<高速液体クロマトグラフ-質量分析の条件>
・検出器:フォトダイオードアレイ検出器、蒸発光散乱検出器
・カラム:Inertsil ODS(3.0×150mm、3μm)
・移動相:A)MeCN/(0.1%ギ酸+0.1%ギ酸アンモニウム)=80/20
B)THF
・質量分析:サーモフィッシャーサイエンティフィック社製イオントラップMS
・イオン源:Heated ESI positive、negative
・m/z範囲:150-1000
<界面活性剤B>
一般式(I)の第3級アミン(x=0、n1+n2=7、A1O及びA2Oの炭素数:2、R1:オレイル基)85質量%と、一般式(I)の第3級アミン(x=0、n1+n2=10、A1O及びA2Oの炭素数:2、R1:ステアリル基)15質量%との混合物(質量割合は液体クロマトグラフ質量分析法により測定)。HLB値:11.7。液体クロマトグラフ質量分析法による質量スペクトルのピーク出現位置:400~850m/z(≒分子量の範囲:400~850g/mol)。
<界面活性剤C>
ポリオキシアルキレン脂肪酸エステル(HLB値:11.1、オキシアルキレン基の炭素数:2、オキシアルキレン基の平均付加モル数:5、脂肪酸に由来する構成単位の炭素数:18)
<界面活性剤D>
商品名:エソミンO/12、ライオンアクゾ社製、物質名:ポリオキシエチレンオレイルアミン(一般式(I)の第3級アミン、x=0、n1+n2=2、A1O及びA2Oの炭素数:2)、HLB値6.5、重量平均分子量:356
<界面活性剤E>
商品名:INFINEUM-C9440、インフィニアム製、物質名:グリセロールモノオレート
ポリブテニルコハク酸ビスイミドのホウ素変性体、4配位と3配位の比率(3配位のピークの積分値/4配位のピークの積分値+3配位のピークの積分値):0.67、ホウ素原子量/窒素原子量:1.1、ホウ素含量:1.30質量%、窒素含量:1.23質量%
<MoDTC>
Mo含有量0.07質量%のMoDTC
<ポリメタクリレート>
重量平均分子量:23万、SSI:25.2%
<金属系清浄剤>
カルシウムサリシレート、カルシウム含有量:7.8質量%、過塩基性、塩基価224mgKOH/g
<酸化防止剤>
フェノール系酸化防止剤、商品名:IRGANOX-L135、BASF社製
<パッケージ添加剤>
ZnDTP、高分子ビスイミド、アミン系酸化防止剤を含むパッケージ添加剤
<その他添加剤>
金属不活性化剤 、流動点降下剤
実施例1、3、比較例1~3、参考例の内燃機用潤滑油組成物について、さらに清浄性の評価を行った。
<評点>
試験温度は300℃に設定し、その他の条件については、JPI-5S-55-99に準拠して測定した。JPI-5S-55-99に準拠して、試験後のガラス管を0点(黒色)~10点(無色)において0.5刻みで評価を行い、21段階にて評価した。数字が高い程、清浄性が良好であることを示す。
<付着量>
上記評点の試験において、試験後のガラス管に付着したラッカーの質量を測定した。付着量が少ないほど清浄性が良好であることを示す。
Claims (15)
- アルキレンオキサイドを構成単位に有し、かつHLB値7以上15未満である界面活性剤、及び潤滑油基油を含む内燃機関用潤滑油組成物。
- 前記界面活性剤は、分子量が350~950g/molの範囲内にある、請求項1に記載の内燃機関用潤滑油組成物。
- 前記界面活性剤がアミン系化合物である、請求項1又は2に記載の内燃機関用潤滑油組成物。
- 前記アミン系化合物が第3級アミンである、請求項3に記載の内燃機関用潤滑油組成物。
- 前記界面活性剤がポリオキシアルキレン脂肪酸エステルである、請求項1又は2に記載の内燃機関用潤滑油組成物。
- 前記潤滑油組成物中の前記界面活性剤の含有量が0.01~2.0質量%である、請求項1~5のいずれか1項に記載の内燃機関用潤滑油組成物。
- さらに、コハク酸イミドのホウ素変性体を含む、請求項1~6のいずれか1項に記載の内燃機関用潤滑油組成物。
- 前記潤滑油組成物中の前記コハク酸イミドのホウ素変性体のホウ素原子換算の含有量が0.2質量%以下である、請求項7に記載の内燃機関用潤滑油組成物。
- 前記コハク酸イミドのホウ素変性体の含有量に対する前記界面活性剤の含有量(前記コハク酸イミドのホウ素変性体の含有量/前記界面活性剤の含有量)の質量比が100以下である、請求項7又は8に記載の内燃機関用潤滑油組成物。
- 前記コハク酸イミドのホウ素変性体のホウ素原子換算の含有量に対する前記界面活性剤の含有量(前記コハク酸イミドのホウ素変性体のホウ素原子換算の含有量/前記界面活性剤の含有量)の質量比が20以下である、請求項7~9のいずれか1項に記載の内燃機関用潤滑油組成物。
- さらに、ポリ(メタ)アクリレートを含む、請求項1~10のいずれか1項に記載の内燃機関用潤滑油組成物。
- 前記潤滑油基油が、米国石油協会の基油分類において、グループ3及びグループ4に分類される鉱油又は合成油から選ばれる1種以上である請求項1~11のいずれか1項に記載の内燃機関用潤滑油組成物。
- 100℃の動粘度が、3.8~12.5mm2/sである、請求項1~12のいずれか1項に記載の内燃機関用潤滑油組成物。
- ガソリンエンジンに用いられる請求項1~13のいずれか1項に記載の内燃機関用潤滑油組成物。
- ガソリンエンジンに、請求項1~13のいずれか1項に記載の内燃機関用潤滑油組成物を添加するガソリンエンジンの摩擦低減方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/559,579 US20180044609A1 (en) | 2015-03-23 | 2016-03-09 | Lubricating oil composition for internal-combustion engine, and method for reducing friction in gasoline engine |
| EP16768443.0A EP3275979A4 (en) | 2015-03-23 | 2016-03-09 | Lubricating oil composition for internal-combustion engine, and method for reducing friction in gasoline engine |
| CN201680017222.1A CN107406794A (zh) | 2015-03-23 | 2016-03-09 | 内燃机用润滑油组合物和汽油发动机的降摩擦方法 |
| JP2017508200A JPWO2016152540A1 (ja) | 2015-03-23 | 2016-03-09 | 内燃機関用潤滑油組成物及びガソリンエンジンの摩擦低減方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015059666 | 2015-03-23 | ||
| JP2015-059666 | 2015-03-23 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016152540A1 true WO2016152540A1 (ja) | 2016-09-29 |
Family
ID=56977295
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/057465 WO2016152540A1 (ja) | 2015-03-23 | 2016-03-09 | 内燃機関用潤滑油組成物及びガソリンエンジンの摩擦低減方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20180044609A1 (ja) |
| EP (1) | EP3275979A4 (ja) |
| JP (1) | JPWO2016152540A1 (ja) |
| CN (1) | CN107406794A (ja) |
| WO (1) | WO2016152540A1 (ja) |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001139978A (ja) * | 1999-08-30 | 2001-05-22 | Tonen Corp | 内燃機関用潤滑油組成物 |
| JP2002294270A (ja) * | 2001-04-02 | 2002-10-09 | Tonengeneral Sekiyu Kk | 内燃機関用潤滑油組成物 |
| JP2004353456A (ja) * | 2003-05-27 | 2004-12-16 | Nissan Motor Co Ltd | 低摩擦摺動カム・フォロワの組合せ及びこれに用いる潤滑油組成物 |
| JP2005146010A (ja) * | 2003-11-11 | 2005-06-09 | Nippon Oil Corp | エンジン油用潤滑油組成物 |
| JP2008056876A (ja) * | 2006-09-04 | 2008-03-13 | Idemitsu Kosan Co Ltd | 内燃機関用潤滑油組成物 |
| JP2010209182A (ja) * | 2009-03-09 | 2010-09-24 | Cosmo Oil Lubricants Co Ltd | ガスエンジン用エンジン油組成物 |
| JP2013194097A (ja) * | 2012-03-16 | 2013-09-30 | Idemitsu Kosan Co Ltd | 潤滑油組成物、該潤滑油組成物を用いた摺動機構 |
| JP2014227424A (ja) * | 2013-05-17 | 2014-12-08 | 昭和シェル石油株式会社 | 内燃機関用潤滑油組成物 |
| JP5807833B1 (ja) * | 2015-05-16 | 2015-11-10 | 有限会社タービュランス・リミテッド | 潤滑油剤用の添加剤、及び潤滑油剤組成物 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4704217A (en) * | 1986-08-20 | 1987-11-03 | Texaco Inc. | Gasoline crankcase lubricant |
| EP0448129A3 (en) * | 1986-11-07 | 1991-10-23 | The Lubrizol Corporation | Sulfur-containing compositions, lubricant fuel and functional fluid compositions |
| CA2030481C (en) * | 1990-06-20 | 1998-08-11 | William B. Chamberlin, Iii | Lubricating oil compositions for meoh-fueled diesel engines |
| JP4931299B2 (ja) * | 2001-07-31 | 2012-05-16 | Jx日鉱日石エネルギー株式会社 | 潤滑油組成物 |
| US20070184991A1 (en) * | 2002-01-31 | 2007-08-09 | Winemiller Mark D | Lubricating oil compositions with improved friction properties |
| EP1482190B1 (en) * | 2003-05-27 | 2012-12-05 | Nissan Motor Company Limited | Rolling element |
| US9499763B2 (en) * | 2012-12-21 | 2016-11-22 | Afton Chemical Corporation | Additive compositions with plural friction modifiers |
-
2016
- 2016-03-09 EP EP16768443.0A patent/EP3275979A4/en not_active Withdrawn
- 2016-03-09 US US15/559,579 patent/US20180044609A1/en not_active Abandoned
- 2016-03-09 JP JP2017508200A patent/JPWO2016152540A1/ja active Pending
- 2016-03-09 CN CN201680017222.1A patent/CN107406794A/zh active Pending
- 2016-03-09 WO PCT/JP2016/057465 patent/WO2016152540A1/ja active Application Filing
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001139978A (ja) * | 1999-08-30 | 2001-05-22 | Tonen Corp | 内燃機関用潤滑油組成物 |
| JP2002294270A (ja) * | 2001-04-02 | 2002-10-09 | Tonengeneral Sekiyu Kk | 内燃機関用潤滑油組成物 |
| JP2004353456A (ja) * | 2003-05-27 | 2004-12-16 | Nissan Motor Co Ltd | 低摩擦摺動カム・フォロワの組合せ及びこれに用いる潤滑油組成物 |
| JP2005146010A (ja) * | 2003-11-11 | 2005-06-09 | Nippon Oil Corp | エンジン油用潤滑油組成物 |
| JP2008056876A (ja) * | 2006-09-04 | 2008-03-13 | Idemitsu Kosan Co Ltd | 内燃機関用潤滑油組成物 |
| JP2010209182A (ja) * | 2009-03-09 | 2010-09-24 | Cosmo Oil Lubricants Co Ltd | ガスエンジン用エンジン油組成物 |
| JP2013194097A (ja) * | 2012-03-16 | 2013-09-30 | Idemitsu Kosan Co Ltd | 潤滑油組成物、該潤滑油組成物を用いた摺動機構 |
| JP2014227424A (ja) * | 2013-05-17 | 2014-12-08 | 昭和シェル石油株式会社 | 内燃機関用潤滑油組成物 |
| JP5807833B1 (ja) * | 2015-05-16 | 2015-11-10 | 有限会社タービュランス・リミテッド | 潤滑油剤用の添加剤、及び潤滑油剤組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3275979A4 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN107406794A (zh) | 2017-11-28 |
| JPWO2016152540A1 (ja) | 2018-01-18 |
| EP3275979A1 (en) | 2018-01-31 |
| US20180044609A1 (en) | 2018-02-15 |
| EP3275979A4 (en) | 2018-08-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6114330B2 (ja) | 潤滑油組成物及び内燃機関の摩擦低減方法 | |
| CN107075405B (zh) | 润滑油组合物、和该润滑油组合物的制造方法 | |
| JP6334503B2 (ja) | 潤滑油組成物及びその製造方法 | |
| JP5280668B2 (ja) | 潤滑油組成物 | |
| US11072759B2 (en) | Lubricating oil composition, lubrication method, and transmission | |
| JP6302416B2 (ja) | 潤滑油組成物 | |
| WO2018074557A1 (ja) | 潤滑油組成物、潤滑方法、及び変速機 | |
| WO2004003118A1 (ja) | 潤滑油添加剤、これを含有する潤滑油組成物及びその製造方法 | |
| US20220169946A1 (en) | Lubricating oil composition | |
| JP5490434B2 (ja) | 熱処理油組成物 | |
| WO2016152540A1 (ja) | 内燃機関用潤滑油組成物及びガソリンエンジンの摩擦低減方法 | |
| JP2020164747A (ja) | 潤滑油組成物 | |
| CA3189299A1 (en) | Succinimide dispersants post-treated with heteroaromatic glycidyl ethers that exhibit good soot handling performance | |
| WO2017170401A1 (ja) | 潤滑油組成物 | |
| EP3950896A1 (en) | Lubricating oil composition | |
| JP6780826B2 (ja) | 潤滑油組成物及び内燃機関の摩擦低減方法 | |
| EP3536767A1 (en) | Lubricant composition | |
| WO2016170813A1 (ja) | 自動変速機油 | |
| JP2022184120A (ja) | 潤滑油用添加剤組成物及び潤滑油組成物 | |
| CA3193618A1 (en) | Lubricating oil containing alkyl phosphonic acid | |
| JP2018188549A (ja) | 潤滑油組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16768443 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2017508200 Country of ref document: JP Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2016768443 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15559579 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |