WO2016068278A1 - 水晶体硬化抑制剤 - Google Patents
水晶体硬化抑制剤 Download PDFInfo
- Publication number
- WO2016068278A1 WO2016068278A1 PCT/JP2015/080673 JP2015080673W WO2016068278A1 WO 2016068278 A1 WO2016068278 A1 WO 2016068278A1 JP 2015080673 W JP2015080673 W JP 2015080673W WO 2016068278 A1 WO2016068278 A1 WO 2016068278A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- lens
- salt
- formula
- acid
- Prior art date
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5383—1,4-Oxazines, e.g. morpholine ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/195—Carboxylic acids, e.g. valproic acid having an amino group
- A61K31/197—Carboxylic acids, e.g. valproic acid having an amino group the amino and the carboxyl groups being attached to the same acyclic carbon chain, e.g. gamma-aminobutyric acid [GABA], beta-alanine, epsilon-aminocaproic acid, pantothenic acid
- A61K31/198—Alpha-aminoacids, e.g. alanine, edetic acids [EDTA]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0048—Eye, e.g. artificial tears
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/12—Ophthalmic agents for cataracts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2300/00—Mixtures or combinations of active ingredients, wherein at least one active ingredient is fully defined in groups A61K31/00 - A61K41/00
Definitions
- the present invention relates to a lens hardening inhibitor containing pirenoxine or a salt thereof and / or a thiopronin or a salt thereof, and a therapeutic and / or preventive agent for a disease involving lens hardening.
- the lens is an organ that is in front of the eyeball and refracts light rays from outside to form an image on the retina. It plays the role of a convex lens in the camera and adjusts the focus of the eye by making the lens thicker when looking closer and thinner when looking far.
- the lens is connected to a muscle called the ciliary body and is supported by the chin zonule, and the lens thickness is controlled by the contraction and relaxation of the ciliary muscle and the relaxation and contraction of the chin zonule. Yes.
- the lens decreases and the lens becomes clouded with aging, an eye disease occurs. For example, when the lens loses its elasticity and hardens, the lens thickness cannot be adjusted properly and presbyopia occurs. In addition, when the lens becomes clouded, light is scattered at that portion, and the object becomes hazy or blurred (cataract).
- pirenoxine product name: catalin
- glutathione product name: tathion
- thiopronin product name: thiola
- salivary gland hormone product name: parotin
- Non-Patent Document 1 corneal damage accompanied by oxidative DNA damage and lacrimal gland dysfunction occur in rats exposed to mainstream smoke.
- smoking treatment we succeeded in causing lens hardening in experimental animals, and further showed that the effect of the drug on the elasticity of the lens can be evaluated using this model (Patent Document 1).
- an object of the present invention is to search for a substance having an inhibitory action on lens hardening, and thus to provide a novel and effective therapeutic agent and / or preventive agent for diseases involving lens hardening such as presbyopia. It is.
- the present inventor has conducted intensive research to solve the above-mentioned problems, and as a result, found that pirenoxine or thiopronin has an effect of suppressing the hardening of the lens, and has completed the present invention.
- R 1 to R 4 are the same or different and each represents a hydrogen atom, a halogen atom, a hydroxy group, a sulfanyl group, a lower alkyl group, a lower acyl group, a lower alkoxy group, a carboxyl group, a carbamoyl group, or a carbonylamino acid group. Or a salt thereof, and / or formula (II):
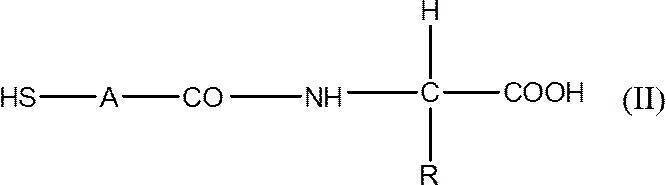
- R represents a hydrogen atom, or a lower alkyl group which may be substituted with an amino group, a hydroxy group, a sulfanyl group or a carboxyl group, and A represents a lower alkylene group, or A lens hardening inhibitor comprising the salt.
- the compound represented by the formula (I) is 1-hydroxy-5-oxo-5H-pyrido [3,2- ⁇ ] phenoxazine-3-carboxylic acid.
- the agent according to [1] comprising a compound represented by the formula (II) or a salt thereof.
- [5] The agent according to [4], wherein the compound represented by the formula (II) is N- (2-mercaptopropionyl) glycine.
- [6] The agent according to any one of [1]-[5], which is a therapeutic and / or prophylactic agent for a disease involving lens hardening.
- [7] The agent according to [6], wherein the disease involving lens hardening is presbyopia.
- the present invention it is possible to treat and / or prevent a disease involving hardening of the lens, such as presbyopia, for which no effective therapeutic and / or preventive drug has existed.
- FIG. 1 shows the effects of mainstream smoke exposure on rat body weight (left graph, unit (g)) and tear volume (right graph, unit (mm / min)).
- NT left bar graph of each column in the right graph
- Smoking right bar graph of each column in the right graph
- FIG. 2 shows the effects of mainstream smoke exposure on the corneal state of rats, and the efficacy of pirenoxine and thiopronin.
- NT indicates a non-smoking treatment group
- Smoking indicates a smoking treatment group
- S + Tiopronin indicates a smoking treatment + 0.1% thiopronin eye drop group
- S + Catalin indicates a smoking treatment + 0.005% pirenoxine eye drop group.
- FIG. 3 shows that lens hardening in rats due to mainstream smoke exposure is suppressed by pirenoxine or thiopronin administration.
- NT indicates a non-smoking treatment group
- Smoking indicates a smoking treatment group
- S + 0.1% Tiopronin indicates a smoking treatment + 0.1% thiopronin ophthalmic group
- S + Catalin indicates a smoking treatment + 0.005% pirenoxine ophthalmic group. . **; p ⁇ 0.01.
- the present invention provides a lens hardening inhibitor comprising pirenoxine or a salt thereof and / or thiopronin or a salt thereof.
- Pignoxins means pirenoxine (1-hydroxy-5-oxo-5H-pyrido [3,2- ⁇ ] phenoxazine-3-carboxylic acid) and its analog compounds, specifically, It means a compound represented by the formula (I).
- R 1 to R 4 are the same or different and each represents a hydrogen atom, a halogen atom, a hydroxy group, a sulfanyl group, a lower alkyl group, a lower acyl group, a lower alkoxy group, a carboxyl group, a carbamoyl group, or a carbonylamino acid group.
- the compound represented by the formula (I) may be referred to as the compound (I).
- Halogen atom includes fluorine atom, chlorine atom, bromine atom and iodine atom.
- “Lower alkyl group” means a linear or branched alkyl group having 1 to 3 carbon atoms, and examples thereof include methyl, ethyl, propyl, and isopropyl.
- “Lower acyl group” means a linear or branched acyl group having 1 to 3 carbon atoms, and includes formyl, acetyl, and propionyl.
- “Lower alkoxy group” means a linear or branched alkoxy group having 1 to 3 carbon atoms, and includes methoxy, ethoxy, propoxy, and isopropoxy.
- Carbonyl amino acid group means a group in which a carbonyl group and an amino group of an amino acid are amide-bonded, and examples thereof include carbonyl glycine, carbonyl alanine, carbonyl threonine, and carbonyl glutamic acid.
- R 1 to R 4 are preferably all hydrogen atoms.
- the compounds in which R 1 -R 4 are all hydrogen atoms are pirenoxine (1-hydroxy-5-oxo-5H-pyrido [3,2- ⁇ ] phenoxazine-3-carboxylic acid) It is a known compound known by name, and has been used as a therapeutic agent for early senile cataract because it has anti-oxidant action and protein insolubility-inhibiting action and prevents turbidity of the lens.
- Ophthalmic solution 0.005% (Santen Pharmaceutical Co., Ltd.), product name: Catalin K ophthalmic solution (Senju Pharmaceutical Co., Ltd.), product name: Catalin ophthalmic solution (Senju Pharmaceutical Co., Ltd., etc.).
- pirenoxine has an effect of suppressing the hardening of the lens.
- Thiopronins mean thiopronin (N- (2-mercaptopropionyl) glycine) and its analog compounds, specifically, compounds represented by the following formula (II).
- R represents a hydrogen atom or a lower alkyl group which may be substituted with an amino group, a hydroxy group, a sulfanyl group or a carboxyl group, and A represents a lower alkylene group.
- R represents a hydrogen atom or a lower alkyl group which may be substituted with an amino group, a hydroxy group, a sulfanyl group or a carboxyl group, and A represents a lower alkylene group.
- the “lower alkyl group optionally substituted with an amino group, hydroxy group, sulfanyl group or carboxyl group” means a linear or branched group optionally substituted with an amino group, hydroxy group, sulfanyl group or carboxyl group And an alkyl group having 1 to 4 carbon atoms such as methyl, mercaptomethyl, 4-aminobutyl, carboxymethyl, 1-hydroxyethyl and the like. Mercaptomethyl is preferred.
- the “lower alkylene group” means a linear or branched alkylene group having 1 to 3 carbon atoms, and includes methylene, ethylidene, ethylene, propylidene, isopropylidene, propylene, trimethylene and the like. Ethylidene is preferred.
- R is preferably a hydrogen atom and A is preferably ethylidene.
- the compound in which R is a hydrogen atom and A is ethylidene in the formula (II) is a known compound known as thiopronin (N- (2-mercaptopropionyl) glycine) and promotes the activity of the liver enzyme system Since it has an action, an inhibitory action on lens protein aggregation, etc., it has been conventionally used as a liver function improving drug for chronic liver disease, a therapeutic agent for early senile cortical cataract, etc. Corporation)]. However, it has not been known so far that thiopronin has an effect of suppressing the hardening of the lens.
- Compound (I) or (II) may be in the form of a salt with an inorganic base, an organic base, an inorganic acid, an organic acid, or the like.
- the salt with the inorganic base include, for example, alkali metal salts such as sodium salt and potassium salt; alkaline earth metal salts such as calcium salt and magnesium salt; and aluminum salt and ammonium salt.
- the salt with the organic base include salts with trimethylamine, triethylamine, pyridine, picoline, ethanolamine, diethanolamine, triethanolamine, dicyclohexylamine, and N, N′-dibenzylethylenediamine.
- Examples of the salt with the inorganic acid include salts with hydrochloric acid, hydrobromic acid, nitric acid, sulfuric acid, and phosphoric acid.
- Examples of the salt with the organic acid include formic acid, acetic acid, trifluoroacetic acid, fumaric acid, oxalic acid, tartaric acid, maleic acid, citric acid, succinic acid, malic acid, methanesulfonic acid, benzenesulfonic acid, p- And a salt with toluenesulfonic acid. Of these salts, pharmacologically acceptable salts are preferred.
- preferred salts thereof include alkali metal salts.
- compound (II) ⁇ has an isomer such as an optical isomer, a stereoisomer, a positional isomer, a rotational isomer, etc.
- an isomer or a mixture of isomers is included in compound (II) Is done.
- compound (II) ⁇ has an optical isomer
- an optical isomer resolved from a racemate is also encompassed in compound (II).
- Each of these isomers can be obtained as a single product by a known synthesis method or separation method (concentration, solvent extraction, column chromatography, recrystallization, etc.).
- Compound (I) or (II) may be crystalline or amorphous.
- compound (I) or (II) is a crystal, it is included in compound (I) I or (II) regardless of whether it is a single crystal form or a mixture of crystal forms.
- the crystal can be produced by crystallization by applying a crystallization method known per se.
- Compound (I) or (II) ⁇ ⁇ may be a solvate (for example, hydrate etc.) or a non-solvate, and both are included in compound (I) or (II). .
- Compound (I) or (II) may be labeled with an isotope (eg, 3 H, 14 C, 35 S, etc.).
- an isotope eg, 3 H, 14 C, 35 S, etc.
- Compound (I) can be produced, for example, according to the method described in Japanese Patent Publication No. 55-10570.
- Compound (II) can be produced according to the method described in, for example, Japanese Patent Publication No. 56-5388.
- compound (I) is pirenoxine
- compound (II) is thiopronin
- thiopronin-containing pharmaceutical composition can also be used.
- compound (I) or (II) ⁇ ⁇ ⁇ can suppress lens hardening, it is effective for the treatment and / or prevention of diseases involving lens hardening.
- “inhibition of hardening of the lens” includes not only suppressing the hardening of the lens and maintaining the hardness of the lens, but also recovering the hardening of the lens and imparting elasticity.
- the “disease involving lens hardening” means any disease in which hardening of the lens contributes to its onset / progression or causes hardening of the lens with the onset / progress.
- Examples of such diseases include presbyopia, hyperopia, cataracts, etc., and preferred examples include presbyopia, cataracts (provided that compound (I) ⁇ is pirenoxine and compound (II) is thiopronin). Except for use as a treatment for cataracts).
- the lens adjusts the focus of the eye by changing its thickness. Therefore, a disease state in which lens hardening is involved is that the lens is hardened and loses elasticity, making it difficult to adjust the focus of the eye.
- Compound (I) or (II) ⁇ ⁇ has low toxicity, and as a pharmaceutical composition in which a pharmaceutically acceptable additive is mixed as it is or according to a method known per se, human and other mammalian moths (eg, rats, rabbits, sheep) , Pigs, cattle, cats, dogs, monkeys, etc.) and can be safely administered orally or parenterally.
- human and other mammalian moths eg, rats, rabbits, sheep
- pirenoxine and thiopronin are already on the market as pharmaceuticals, and knowledge about safety is accumulated.
- Compound ⁇ ⁇ ⁇ (I) or a salt thereof contained in the lens sclerosis inhibitor or the therapeutic and / or prophylactic agent for diseases involving lens sclerosis of the present invention (hereinafter sometimes collectively referred to as ⁇ the agent of the present invention '')
- the amount of the compound (II) or a salt ⁇ ⁇ thereof (hereinafter sometimes collectively referred to as the ⁇ compound of the present invention '') is particularly sufficient if it is sufficient to suppress lens hardening and does not exhibit cytotoxicity.
- the content of the compound of the present invention can be appropriately increased or decreased depending on the purpose of use, dosage form, degree of disease state, and the like.
- the above compound (II) or a salt thereof can also be used in combination.
- all the active ingredients may be blended in the same preparation, or each active ingredient may be blended separately in two or more preparations. What is necessary is just to set so that the total content of an active ingredient may become said range when using two or more active ingredients together.
- the dosage form of the agent of the present invention is not particularly limited, and various dosage forms suitable for oral administration or parenteral administration can be appropriately selected.
- preparations for parenteral administration for example, eye drops, ointments, lotions, creams, injections, suppositories, etc. are used, preferably dosage forms suitable for topical administration, such as eye drops Examples thereof include aqueous preparations (aqueous eye drops, non-aqueous eye drops, suspension eye drops, emulsion eye drops, etc.), ointments, lotions, creams and the like.
- a substrate can be appropriately used.
- the base material used for the eye drops include phosphate buffer, Hanks buffer, physiological saline, perfusate, artificial tears and the like.
- a pharmaceutically acceptable additive for example, a preparation for topical ocular administration, for example, a buffer, an isotonic agent, a solubilizing agent, a preservative.
- a buffer for example, a buffer, an isotonic agent, a solubilizing agent, a preservative.
- Viscosity base, chelating agent, cooling agent, pH adjuster, antioxidant and the like can be appropriately selected and added.
- the buffer include a phosphate buffer, a borate buffer, a citrate buffer, a tartrate buffer, an acetate buffer, and an amino acid.
- isotonic agent examples include saccharides such as sorbitol, glucose and mannitol, polyhydric alcohols such as glycerin and propylene glycol, salts such as sodium chloride, boric acid and the like.
- solubilizers include polyoxyethylene sorbitan monooleate (for example, polysorbate 80), polyoxyethylene hydrogenated castor oil, nonionic surfactants such as tyloxapol and pluronic, and polyhydric alcohols such as glycerin and macrogol. It is done.
- preservative examples include quaternary ammonium salts such as benzalkonium chloride, benzethonium chloride, cetylpyridinium chloride, paraoxybenzoic acid such as methyl paraoxybenzoate, ethyl paraoxybenzoate, propyl paraoxybenzoate, and butyl paraoxybenzoate.
- quaternary ammonium salts such as benzalkonium chloride, benzethonium chloride, cetylpyridinium chloride, paraoxybenzoic acid such as methyl paraoxybenzoate, ethyl paraoxybenzoate, propyl paraoxybenzoate, and butyl paraoxybenzoate.
- esters examples, benzyl alcohol, sorbic acid and its salts (sodium salt, potassium salt, etc.), thimerosal (trade name), chlorobutanol, sodium dehydroacetate and the like.
- Examples of the viscous base include water-soluble polymers such as polyvinyl pyrrolidone, polyethylene glycol, and polyvinyl alcohol, and celluloses such as hydroxyethyl cellulose, methyl cellulose, hydroxypropyl methyl cellulose, and sodium carboxymethyl cellulose.
- Examples of chelating agents include sodium edetate and citric acid.
- Examples of the refreshing agent include l-menthol, borneol, camphor, and eucalyptus oil.
- Examples of the pH adjuster include sodium hydroxide, potassium hydroxide, sodium carbonate, sodium bicarbonate, boric acid or a salt thereof (borax), hydrochloric acid, citric acid or a salt thereof (sodium citrate, sodium dihydrogen citrate).
- Etc. phosphoric acid or salts thereof (disodium hydrogen phosphate, potassium dihydrogen phosphate, etc.), acetic acid or salts thereof (sodium acetate, ammonium acetate, etc.), tartaric acid or salts thereof (sodium tartrate, etc.), and the like.
- the antioxidant include sodium bisulfite, dry sodium sulfite, sodium pyrosulfite, and concentrated mixed tocopherol.
- Compound (I) ⁇ is generally insoluble or sparingly soluble in water, and is made soluble in water in the form of a salt or in the presence of a base and used as an aqueous solution, but generally when used as an aqueous solution or other organic solvent solution Is unstable and easily decomposed, and is particularly inferior in heat and / or light stability. Therefore, it is desirable to formulate it as a solid preparation such as a tablet or granule and dissolve it in a solvent before use.
- the pH of the solvent is usually adjusted to about 3.0 to about 7.0, preferably about 5.0 to about 7.0. Alternatively, it can also be prepared as a finely divided suspension without dissolving in water.
- the pH of the agent of the present invention can be generally adjusted to about 5.0 to about 8.5, preferably about 6.0 to about 8.0.
- These liquid preparations are preferably subjected to sterilization such as filtration sterilization using a membrane filter or the like.
- the agent of the present invention may further contain an ointment base.
- an ointment base Generally fats and oils, wax, a hydrocarbon compound, etc. as a hydrophobic base can be used. Specific examples include mineral bases such as yellow petrolatum, white petrolatum, paraffin, liquid paraffin, plastibase, and silicone, and animal and plant bases such as beeswax and animal and vegetable fats and oils.
- the agent of the present invention is a preparation for oral administration, for example, tablet candy (including sugar-coated tablets and film-coated tablets), pills, granules, powders, capsules (including soft capsules), syrups, It can be formulated into an emulsion, suspension or the like.
- tablet candy including sugar-coated tablets and film-coated tablets
- pills granules, powders, capsules (including soft capsules), syrups
- It can be formulated into an emulsion, suspension or the like.
- These preparations can be produced by a method known per se, for example, the method described in the 14th revised Japanese Pharmacopoeia, General Rules for Preparations, and the above-mentioned pharmaceutically acceptable additives that can be used for preparations for topical ophthalmic administration.
- excipients, lubricants, binders, disintegrants, water-soluble polymers, basic inorganic salts and the like that are usually used in the pharmaceutical field may be contained.
- excipient examples include lactose, sucrose, D-mannitol, starch, corn starch, crystalline cellulose, light anhydrous silicic acid, titanium oxide and the like.
- lubricant examples include magnesium stearate, sucrose fatty acid ester, polyethylene glycol, talc, stearic acid and the like.
- binder examples include hydroxypropylcellulose, hydroxypropylmethylcellulose, crystalline cellulose, starch, polyvinylpyrrolidone, gum arabic powder, gelatin, pullulan, and low-substituted hydroxypropylcellulose.
- Disintegrants include: (1) Crospovidone, (2) Croscarmellose sodium (FMC-Asahi Kasei), Carmellose calcium (Gotoku Pharmaceutical) and other super disintegrants, (3) Carboxymethyl starch sodium ( Examples include Matsutani Chemical Co., Ltd.), (4) Low-substituted hydroxypropyl cellulose (eg, Shin-Etsu Chemical Co., Ltd.), and (5) Corn starch.
- the “crospovidone” is crosslinked with a chemical name of 1-ethenyl-2-pyrrolidinone homopolymer, including those called polyvinylpolypyrrolidone (PVPP) and 1-vinyl-2-pyrrolidinone homopolymer.
- water-soluble polymers examples include ethanol-soluble water-soluble polymers [eg, cellulose derivatives such as hydroxypropylcellulose (hereinafter sometimes referred to as HPC), polyvinylpyrrolidone, etc.], ethanol-insoluble water-soluble polymers [eg, , Hydroxypropylmethylcellulose (hereinafter sometimes referred to as HPMC), cellulose derivatives such as methylcellulose and sodium carboxymethylcellulose, sodium polyacrylate, polyvinyl alcohol, sodium alginate, guar gum and the like.
- the basic inorganic salt include basic inorganic salts of sodium, potassium, magnesium and / or calcium. Preferred is a basic inorganic salt of magnesium and / or calcium. More preferred is a basic inorganic salt of magnesium.
- Examples of the basic inorganic salt of sodium include sodium carbonate, sodium hydrogen carbonate, disodium hydrogen phosphate and the like.
- Examples of the basic inorganic salt of potassium include potassium carbonate and potassium hydrogen carbonate.
- Examples of the basic inorganic salt of magnesium include heavy magnesium carbonate, magnesium carbonate, magnesium oxide, magnesium hydroxide, magnesium magnesium silicate, magnesium silicate, magnesium aluminate, synthetic hydrotalcite [Mg 6 Al 2 ( OH) 16 ⁇ CO 3 ⁇ 4H 2 O] and alumina hydroxide / magnesium, preferably heavy magnesium carbonate, magnesium carbonate, magnesium oxide, magnesium hydroxide and the like.
- Examples of the basic inorganic salt of calcium include precipitated calcium carbonate and calcium hydroxide.
- the pharmaceutical composition can be encapsulated in liposomes in order to facilitate transport into the cells.
- Preferred liposomes are positively-charged liposomes, positively-charged cholesterol, membrane-permeable peptide-bonded liposomes, etc. (Nakanishi Mamoru et al., Protein Nucleic Acid Enzyme, 44: 1590-1596 1999 (1999), Futaki Shiro, Chemistry and Biology, 43: 649- 653 (2005), Clinical Cancer Research 59: 4325-4333 (1999)).
- the agent of the present invention has other active ingredients such as an antiallergic or antihistamine component, a decongestant component, a local anesthetic component, a vitamin component, an effective amino acid, as long as it does not cause an unfavorable interaction with the compound of the present invention.
- Other amino acid components such as valine, leucine, isoleucine, serine, threonine, methionine, proline, phenylalanine, tyrosine, tryptophan, aspartic acid, glutamic acid, lysine, histidine, citrulline, ornithine, cystine, taurine, glycine You may do it.
- active ingredients various drugs known per se can be appropriately used.
- other active ingredients may be formulated separately from the agent of the present invention and administered to the same subject at the same time or at a time difference, or by the same route or different routes.
- the dose of the agent of the present invention varies depending on the subject (animal species) and its age, weight, symptom, dosage form, administration route, etc., for example, pirenoxine or a salt thereof as an active ingredient, and as a therapeutic agent for presbyopia
- pirenoxine or a salt thereof as an active ingredient
- a therapeutic agent for presbyopia When used in human form in the form of eye drops, administer a solution of pirenoxine concentration 0.001-0.01% by weight 1-2 drops at a time, once or 2-6 times a day, preferably 3-5 times. Can do.
- an equivalent amount can be administered. It should be noted that the amount applied and the number of times can be appropriately increased or decreased depending on the degree of symptoms.
- thiopronin or a salt thereof is an active ingredient and is orally administered to a human as a presbyopia therapeutic agent
- the amount of thiopronin is 50-1000 mg, preferably 100-500 mg, once a day or 2-3 Can be administered in divided doses. In the case of parenteral administration, an amount equivalent to this can be administered. It should be noted that the amount applied and the number of times can be appropriately increased or decreased depending on the degree of symptoms.
- a contact lens such as a non-oxygen permeable hard contact lens, an oxygen permeable hard contact lens, or a soft contact lens is mounted.
- each preparation may be administered to the same subject at the same time or at a time difference, or by the same route or different routes.
- Example 1 Smoking treatment by mainstream smoke exposure
- Male 6-8 week old Sprague-Dawley (SD) rats were divided into 4 groups of 4 each, each group being non-smoking treated group (NT), smoking treated Group (Smoking), smoking treatment + 0.1% thiopronin ophthalmic group (S + 0.1% Tiopronin), smoking treatment + 0.005% pirenoxine ophthalmic group (S + Catalin).
- Thiopronin Tiora Tablets 100, manufactured by Mylan Pharmaceutical Co., Ltd. was prepared so that the amount of the active ingredient was 0.1 w / v%, and an eye drop was prepared.
- Example 2 Lens hardness measurement after smoking treatment
- the lens hardness of the lens extracted from the eyeball in Example 1 was measured by the following method.
- the lens hardness was measured by combining an electronic balance and a height gauge.
- a lens whose minor axis length was measured in advance was placed on an electronic balance, and the weight was adjusted to zero.
- the height gauge handle was operated from above the lens so that the tip was in contact with the lens. Furthermore, the tip was lowered about 5-10% of the short axis length by operating the handle, and pressure was applied to the crystalline lens.
- the change in weight at this time was measured with an electronic balance, and the weight was divided by the moving distance indicated by the height gauge as the hardness. It shows that it is so hard that a value is large.
- Figure 3 shows the lenses of the non-smoking treatment group (NT), smoking treatment group (Smoking), smoking treatment + 0.1% thiopronin ophthalmic group (S + 0.1% Tiopronin), smoking treatment + 0.005% pirenoxine ophthalmic group (S + Catalin)
- NT non-smoking treatment group
- S + 0.1% Tiopronin smoking treatment + 0.1% thiopronin ophthalmic group
- S + Catalin The measurement result of hardness is shown. Exposure to mainstream smoke increased the lens hardness (Smoking), and this increase in hardness was significantly suppressed by administration of pirenoxine (S + Catalin) or thiopronin (S + 0.1% Tiopronin).
- the compound of the present invention has an excellent lens hardening inhibitory effect, effective treatment and / or prevention for diseases associated with lens hardening such as presbyopia, for which no effective therapeutic and / or preventive drug has existed so far. Means can be provided.
Abstract
Description
ヒトでは、水晶体は、毛様体と呼ばれる筋肉とつながり、チン小帯で支えられており、毛様筋の収縮・弛緩、及びチン小帯の弛緩・収縮により、水晶体の厚さを制御している。
その要因の1つとして、加齢による水晶体の弾力性低下の正確な発生機序が不明であり、そのため、水晶体硬化(老視)のモデル動物が作製されていなかったことが挙げられる。
[1] 式 (I):
[2] 式 (I) で表される化合物若しくはその塩を含有してなる、[1] に記載の剤。
[3] 式 (I) で表される化合物が1-ヒドロキシ-5-オキソ-5H-ピリド[3,2-α]フェノキサジン-3-カルボン酸である、[2] に記載の剤。
[4] 式 (II) で表される化合物若しくはその塩を含有してなる、[1] に記載の剤。
[5] 式 (II) で表される化合物が、N-(2-メルカプトプロピオニル)グリシンである、[4] に記載の剤。
[6] 水晶体硬化が関与する疾患の治療及び/又は予防剤である、[1]-[5] のいずれかに記載の剤。
[7] 水晶体硬化が関与する疾患が老視である、[6] に記載の剤。
[8] 点眼剤又は眼軟膏剤である、[1]-[7] のいずれかに記載の剤。
本明細書中では、以下、式 (I) で表される化合物を化合物 (I) と称する場合がある。
本明細書中では、式 (II) で表される化合物を化合物 (II) と称する場合がある。
緩衝剤としては、例えば、リン酸緩衝剤、ホウ酸緩衝剤、クエン酸緩衝剤、酒石酸緩衝剤、酢酸緩衝剤、アミノ酸などが挙げられる。
等張化剤としては、ソルビトール、グルコース、マンニトールなどの糖類、グリセリン、プロピレングリコールなどの多価アルコール類、塩化ナトリウムなどの塩類、ホウ酸などが挙げられる。
溶解補助剤としては、ポリオキシエチレンソルビタンモノオレート (例えば、ポリソルベート80)、ポリオキシエチレン硬化ヒマシ油、チロキサポール、プルロニックなどの非イオン性界面活性剤、グリセリン、マクロゴールなどの多価アルコールなどが挙げられる。
防腐剤としては、例えば、塩化ベンザルコニウム、塩化ベンゼトニウム、塩化セチルピリジニウムなどの第四級アンモニウム塩類、パラオキシ安息香酸メチル、パラオキシ安息香酸エチル、パラオキシ安息香酸プロピル、パラオキシ安息香酸ブチルなどのパラオキシ安息香酸エステル類、ベンジルアルコール、ソルビン酸及びその塩 (ナトリウム塩、カリウム塩など)、チメロサール (商品名)、クロロブタノール、デヒドロ酢酸ナトリウムなどが挙げられる。
粘性基剤としては、ポリビニルピロリドン、ポリエチレングリコール、ポリビニルアルコールなどの水溶性高分子、ヒドロキシエチルセルロース、メチルセルロース、ヒドロキシプロピルメチルセルロース、カルボキシメチルセルロースナトリウムなどのセルロース類などが挙げられる。
キレート剤としては、エデト酸ナトリウム、クエン酸などが挙げられる。
清涼化剤としては、l-メントール、ボルネオール、カンフル、ユーカリ油などが挙げられる。
pH調整剤としては、例えば、水酸化ナトリウム、水酸化カリウム、炭酸ナトリウム、炭酸水素ナトリウム、ホウ酸又はその塩 (ホウ砂)、塩酸、クエン酸又はその塩 (クエン酸ナトリウム、クエン酸二水素ナトリウム等)、リン酸又はその塩 (リン酸水素二ナトリウム、リン酸二水素カリウム等)、酢酸又はその塩 (酢酸ナトリウム、酢酸アンモニウム等)、酒石酸又はその塩 (酒石酸ナトリウム等)等が挙げられる。
抗酸化剤としては、例えば、亜硫酸水素ナトリウム、乾燥亜硫酸ナトリウム、ピロ亜硫酸ナトリウム、濃縮混合トコフェロール等が挙げられる。
滑沢剤としては、例えば、ステアリン酸マグネシウム、ショ糖脂肪酸エステル、ポリエチレングリコール、タルク、ステアリン酸等が挙げられる。
結合剤としては、例えば、ヒドロキシプロピルセルロース、ヒドロキシプロピルメチルセルロース、結晶セルロース、デンプン、ポリビニルピロリドン、アラビアゴム末、ゼラチン、プルラン、低置換度ヒドロキシプロピルセルロース等が挙げられる。
崩壊剤としては、(1) クロスポビドン、(2) クロスカルメロースナトリウム (FMC-旭化成)、カルメロースカルシウム (五徳薬品) 等スーパー崩壊剤と称される崩壊剤、(3) カルボキシメチルスターチナトリウム (例、松谷化学 (株) 製)、(4) 低置換度ヒドロキシプロピルセルロース (例、信越化学 (株) 製)、(5) コーンスターチ等が挙げられる。該「クロスポビドン」としては、ポリビニルポリピロリドン (PVPP)、1-ビニル-2-ピロリジノンホモポリマーと称されているものも含め、1-エテニル-2-ピロリジノンホモポリマーという化学名を有し架橋されている重合物のいずれであってもよく、具体例としては、コリドンCL (BASF社製)、ポリプラスドンXL (ISP社製)、ポリプラスドンXL-10 (ISP社製)、ポリプラスドンINF-10 (ISP社製)等である。
水溶性高分子としては、例えば、エタノール可溶性水溶性高分子 [例えば、ヒドロキシプロピルセルロース (以下、HPCと記載することがある) 等のセルロース誘導体、ポリビニルピロリドン等]、エタノール不溶性水溶性高分子 [例えば、ヒドロキシプロピルメチルセルロース (以下、HPMCと記載することがある)、メチルセルロース、カルボキシメチルセルロースナトリウム等のセルロース誘導体、ポリアクリル酸ナトリウム、ポリビニルアルコール、アルギン酸ナトリウム、グアーガム等] 等が挙げられる。
塩基性無機塩としては、例えば、ナトリウム、カリウム、マグネシウム及び/又はカルシウムの塩基性無機塩が挙げられる。好ましくはマグネシウム及び/又はカルシウムの塩基性無機塩である。さらに好ましくはマグネシウムの塩基性無機塩である。該ナトリウムの塩基性無機塩としては、例えば、炭酸ナトリウム、炭酸水素ナトリウム、リン酸水素二ナトリウム等が挙げられる。該カリウムの塩基性無機塩としては、例えば、炭酸カリウム、炭酸水素カリウム等が挙げられる。該マグネシウムの塩基性無機塩としては、例えば、重質炭酸マグネシウム、炭酸マグネシウム、酸化マグネシウム、水酸化マグネシウム、メタ珪酸アルミン酸マグネシウム、珪酸マグネシウム、アルミン酸マグネシウム、合成ヒドロタルサイト [Mg6Al2(OH)16・CO3・4H2O] 及び水酸化アルミナ・マグネシウム、好ましくは、重質炭酸マグネシウム、炭酸マグネシウム、酸化マグネシウム、水酸化マグネシウム等が挙げられる。該カルシウムの塩基性無機塩としては、例えば、沈降炭酸カルシウム、水酸化カルシウム等が挙げられる。
雄の6-8週齢のSprague-Dawley (SD) ラットを、各群4匹づつ4群に分け、それぞれを、非喫煙処理群 (NT)、喫煙処理群 (Smoking)、喫煙処理+0.1% チオプロニン点眼群 (S+0.1% Tiopronin)、喫煙処理+0.005% ピレノキシン点眼群 (S+Catalin) とした。
チオプロニン (チオラ錠100、マイラン製薬株式会社製) は、有効成分量が0.1 w/v%となるように調製し、点眼剤を作製した。0.005% ピレノキシン点眼群には、カタリン点眼用0.005% (千寿製薬株式会社製) を用いた。点眼は、各眼に1回につき5 μLずつ点眼を行った。下記の喫煙処理前に1回、処理後に3回の点眼を、12日間行った。
喫煙処理は、Higuchiらの方法 (Free Radic. Biol. Med., 2011, Vol. 51, pages 2210-2216) を参考にして、以下のように行った。ラットを入れた喫煙チャンバー内にシリンジを使用して300 mLの主流煙を添加した。雄の6-8週齢SDラットを、実験用に作製した喫煙チャンバー (60 cm×40 cm×35 cm) 内に入れ (最大12匹まで)、チャンバー内にエアーポンプを用いて新鮮な空気を送り込んだ。タバコ (セブンスター (登録商標)) を50 mLシリンジに付け、主流煙を吸引しチャンバー内に吹き込んだ。この吸引操作を6回繰り返し、主流煙を合計300 mLチャンバー内に入れた後、新鮮な空気をポンプで送りながら30分間放置した。同様の操作をあと5回 (計6回) 繰り返し、ラットは1日当り合計3時間主流煙に曝露させた。その後、ラットを飼育室に戻した。12日間の喫煙処理を施したラット及び非喫煙群は、体重測定後、角膜、涙腺、水晶体の採取を行った。喫煙処理により、体重は低下傾向となり、涙液量も低下した (図1)。次に、採取した角膜の蛍光染色を行い、角膜の状態を確認した (図2)。喫煙処理群では、非喫煙群と比較して、蛍光染色スコアが有意に増大しており、角膜状態の悪化が認められた。喫煙処理されたラットのうち、点眼群 (S+Tiopronin、S+Catalin) と非点眼群 (Smoking) の蛍光スコアを比較した場合には、点眼による角膜状態の悪化は認められず、0.005% ピレノキシン点眼群 (S+Catalin) では、改善傾向が認められた。
実施例1で眼球から摘出した水晶体は、以下の方法によって水晶体硬度を測定した。水晶体硬度は電子天秤とハイトゲージを組み合わせて測定した。あらかじめ短軸長を測定した水晶体を電子天秤の上に置き、重さを0に合わせた。水晶体の上からハイトゲージのハンドルを操作して、先端部分が水晶体に接するようにした。さらにハンドルを操作して短軸長の5-10%程度先端を下げ、水晶体に圧力をかけた。この時の重さの変化を電子天秤で測定し、重さをハイトゲージに示された移動距離で割り硬度とした。値が大きいほど硬いことを示す。水晶体硬度の統計処理は、喫煙処理群に対する各群の有意差検定を、Dunnett法を用いて行った。
図3に、非喫煙処理群 (NT)、喫煙処理群 (Smoking)、喫煙処理+0.1% チオプロニン点眼群 (S+0.1% Tiopronin)、喫煙処理+0.005% ピレノキシン点眼群 (S+Catalin) の水晶体硬度の測定結果を示す。主流煙曝露により水晶体硬度は増大し (Smoking)、この硬度の増大はピレノキシンの投与 (S+Catalin)、又はチオプロニン (S+0.1% Tiopronin) の投与により有意に抑制された。
Claims (8)
- 式 (I) で表される化合物若しくはその塩を含有してなる、請求項1に記載の剤。
- 式 (I) で表される化合物が1-ヒドロキシ-5-オキソ-5H-ピリド[3,2-α]フェノキサジン-3-カルボン酸である、請求項2に記載の剤。
- 式 (II) で表される化合物若しくはその塩を含有してなる、請求項1に記載の剤。
- 式 (II) で表される化合物が、N-(2-メルカプトプロピオニル)グリシンである、請求項4に記載の剤。
- 水晶体硬化が関与する疾患の治療及び/又は予防剤である、請求項1-5のいずれか1項に記載の剤。
- 水晶体硬化が関与する疾患が老視である、請求項6に記載の剤。
- 点眼剤又は眼軟膏剤である、請求項1-7のいずれか1項に記載の剤。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016556651A JP6509244B2 (ja) | 2014-10-31 | 2015-10-30 | 水晶体硬化抑制剤 |
| CN201580058951.7A CN107106566A (zh) | 2014-10-31 | 2015-10-30 | 晶状体硬化抑制剂 |
| EP15854906.3A EP3213751A4 (en) | 2014-10-31 | 2015-10-30 | Phacosclerosis inhibitor |
| US15/521,803 US20170246176A1 (en) | 2014-10-31 | 2015-10-30 | Phacosclerosis inhibitor |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014-223294 | 2014-10-31 | ||
| JP2014223294 | 2014-10-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016068278A1 true WO2016068278A1 (ja) | 2016-05-06 |
Family
ID=55857613
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/080673 WO2016068278A1 (ja) | 2014-10-31 | 2015-10-30 | 水晶体硬化抑制剤 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20170246176A1 (ja) |
| EP (1) | EP3213751A4 (ja) |
| JP (1) | JP6509244B2 (ja) |
| CN (1) | CN107106566A (ja) |
| TW (1) | TW201625264A (ja) |
| WO (1) | WO2016068278A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108558906A (zh) * | 2018-05-21 | 2018-09-21 | 日照市普达医药科技有限公司 | 一种治疗白内障药物苯并吩噁嗪类化合物及其制备方法 |
| WO2022107735A1 (ja) | 2020-11-17 | 2022-05-27 | 株式会社林原 | 水晶体硬度調節剤 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11458104B1 (en) | 2018-06-21 | 2022-10-04 | Mission Pharmacal Company | Enteric coated tiopronin tablet |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH08337525A (ja) * | 1995-04-13 | 1996-12-24 | Kikkoman Corp | 白内障の予防または治療薬剤 |
| JP2007512352A (ja) * | 2003-11-20 | 2007-05-17 | オセラ・フアーマシユーチカルズ・インコーポレーテツド | 黄斑変性およびその他の眼科疾患の改善 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8697109B2 (en) * | 2000-08-16 | 2014-04-15 | Encore Health, Llc | Caged mercaptan and seleno-mercaptan compounds and methods of using them |
| US20060172972A1 (en) * | 2002-12-20 | 2006-08-03 | Chakshu Research Inc | Formulation and method for administration of ophthalmologically active agents |
| CN101102770A (zh) * | 2003-11-20 | 2008-01-09 | 奥特拉药物公司 | 白内障、黄斑变性和其它眼科疾病的改善 |
| AU2006270035A1 (en) * | 2005-07-15 | 2007-01-25 | Chakshu Research Inc. | Formulation and method for administration of ophthalmologically active agents |
| CN102688227A (zh) * | 2012-06-06 | 2012-09-26 | 正大青春宝药业有限公司 | 一种含冰片的丹酚酸a组合物眼用制剂及其制备方法 |
-
2015
- 2015-10-30 EP EP15854906.3A patent/EP3213751A4/en not_active Withdrawn
- 2015-10-30 WO PCT/JP2015/080673 patent/WO2016068278A1/ja active Application Filing
- 2015-10-30 TW TW104135763A patent/TW201625264A/zh unknown
- 2015-10-30 JP JP2016556651A patent/JP6509244B2/ja active Active
- 2015-10-30 CN CN201580058951.7A patent/CN107106566A/zh active Pending
- 2015-10-30 US US15/521,803 patent/US20170246176A1/en not_active Abandoned
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH08337525A (ja) * | 1995-04-13 | 1996-12-24 | Kikkoman Corp | 白内障の予防または治療薬剤 |
| JP2007512352A (ja) * | 2003-11-20 | 2007-05-17 | オセラ・フアーマシユーチカルズ・インコーポレーテツド | 黄斑変性およびその他の眼科疾患の改善 |
Non-Patent Citations (3)
| Title |
|---|
| AKIHIRO HIGUCHI ET AL.: "Research for the Prevention and Treatment of Presbyopia", JOURNAL OF THE EYE, vol. 31, no. 10, 30 October 2014 (2014-10-30), pages 1443 - 1448, XP009502671, ISSN: 0910-1810 * |
| KATSUHIKO NIIMI: "Hakunaisho", JAPANESE JOURNAL OF CLINICAL MEDICINE, vol. 49, no. 624, 29 July 1991 (1991-07-29), pages 1199 - 1200, XP009502673, ISSN: 0047-1852 * |
| See also references of EP3213751A4 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108558906A (zh) * | 2018-05-21 | 2018-09-21 | 日照市普达医药科技有限公司 | 一种治疗白内障药物苯并吩噁嗪类化合物及其制备方法 |
| WO2022107735A1 (ja) | 2020-11-17 | 2022-05-27 | 株式会社林原 | 水晶体硬度調節剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3213751A1 (en) | 2017-09-06 |
| TW201625264A (zh) | 2016-07-16 |
| JPWO2016068278A1 (ja) | 2017-08-10 |
| EP3213751A4 (en) | 2018-06-06 |
| JP6509244B2 (ja) | 2019-05-08 |
| CN107106566A (zh) | 2017-08-29 |
| US20170246176A1 (en) | 2017-08-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4933897B2 (ja) | 眼内移行性促進水性点眼剤 | |
| RU2406499C2 (ru) | Профилактическое или терапевтическое средство для лечения кератоконъюнктивитных нарушений | |
| US11951098B2 (en) | Therapeutic agent for meibomian dysfunction | |
| JP2005145961A (ja) | 角結膜障害の治療剤 | |
| EA034839B1 (ru) | Офтальмологический раствор | |
| JP7222042B2 (ja) | 5′-アデノシン二リン酸リボース(adpr)の使用方法 | |
| JP7248836B2 (ja) | ヘテロシクリデンアセトアミド誘導体含有医薬 | |
| JP6509244B2 (ja) | 水晶体硬化抑制剤 | |
| WO2016171152A1 (ja) | 角膜障害の治療剤、改善剤または予防剤 | |
| US7772258B2 (en) | Agent for treatment of allergic eye disease | |
| KR102633606B1 (ko) | 활성 성분으로서 티오트로피움을 포함하는, 근시 예방, 근시 치료, 및/또는 근시 진행 예방을 위한 제제 | |
| US20160303079A1 (en) | Use of indolyl and idolinyl hydroxamates for treating neurodegenerative disorders or cognitive decicits | |
| JP2005047909A (ja) | ピペリジン誘導体を有効成分とする掻痒治療剤 | |
| ES2860767T3 (es) | Mirabegrón para el tratamiento de enfermedades retinianas | |
| JP6596022B2 (ja) | 角膜上皮障害治療剤 | |
| TW201934542A (zh) | 含有FP促效藥及β阻斷藥的青光眼治療劑 | |
| TW201838629A (zh) | 用於預防近視、治療近視及/或預防近視增長之包含蕪地銨作爲活性成分的藥劑 | |
| JP2007291091A (ja) | 角結膜障害治療剤 | |
| JP5087233B2 (ja) | 角結膜障害の予防または治療剤 | |
| JP2006104199A (ja) | 角結膜障害治療剤 | |
| WO2007066678A1 (ja) | 角結膜障害治療剤 | |
| JP2006199688A (ja) | 角結膜障害治療剤 | |
| JP2005187458A (ja) | シロミラストまたはその塩を有効成分とする掻痒治療剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15854906 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016556651 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15521803 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015854906 Country of ref document: EP |