WO2016006262A1 - 心臓細胞培養材料 - Google Patents
心臓細胞培養材料 Download PDFInfo
- Publication number
- WO2016006262A1 WO2016006262A1 PCT/JP2015/050028 JP2015050028W WO2016006262A1 WO 2016006262 A1 WO2016006262 A1 WO 2016006262A1 JP 2015050028 W JP2015050028 W JP 2015050028W WO 2016006262 A1 WO2016006262 A1 WO 2016006262A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- heart
- cells
- vcam
- cell
- fibroblasts
- Prior art date
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/33—Fibroblasts
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0657—Cardiomyocytes; Heart cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/177—Receptors; Cell surface antigens; Cell surface determinants
- A61K38/1774—Immunoglobulin superfamily (e.g. CD2, CD4, CD8, ICAM molecules, B7 molecules, Fc-receptors, MHC-molecules)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/36—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix
- A61L27/38—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix containing added animal cells
- A61L27/3804—Materials for grafts or prostheses or for coating grafts or prostheses containing ingredients of undetermined constitution or reaction products thereof, e.g. transplant tissue, natural bone, extracellular matrix containing added animal cells characterised by specific cells or progenitors thereof, e.g. fibroblasts, connective tissue cells, kidney cells
- A61L27/3826—Muscle cells, e.g. smooth muscle cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms, e.g. protozoa; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0656—Adult fibroblasts
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/10—Cells modified by introduction of foreign genetic material
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2430/00—Materials or treatment for tissue regeneration
- A61L2430/20—Materials or treatment for tissue regeneration for reconstruction of the heart, e.g. heart valves
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/50—Cell markers; Cell surface determinants
- C12N2501/58—Adhesion molecules, e.g. ICAM, VCAM, CD18 (ligand), CD11 (ligand), CD49 (ligand)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/998—Proteins not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2502/00—Coculture with; Conditioned medium produced by
- C12N2502/13—Coculture with; Conditioned medium produced by connective tissue cells; generic mesenchyme cells, e.g. so-called "embryonic fibroblasts"
- C12N2502/1323—Adult fibroblasts
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2502/00—Coculture with; Conditioned medium produced by
- C12N2502/13—Coculture with; Conditioned medium produced by connective tissue cells; generic mesenchyme cells, e.g. so-called "embryonic fibroblasts"
- C12N2502/1329—Cardiomyocytes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2533/00—Supports or coatings for cell culture, characterised by material
- C12N2533/50—Proteins
Definitions
- the present invention relates to a heart cell culture material and a cell culture substrate obtained by coating the wall surface and / or bottom surface of a culture substrate having a wall surface and / or a bottom surface with the heart cell culture material.
- the present invention also relates to an artificial organ material obtained by culturing heart cells using the heart cell culture material and a method for producing the same.
- Fibroblasts are present in almost all vertebrates, and when the tissue is injured by trauma or ischemia, the damaged area is replaced by fibrotic tissue through fibroblast proliferation and abundant extracellular matrix attachment .
- various heart diseases such as myocardial infarction and cardiomyopathy, when many cardiomyocytes are lost, fibrous tissue is also replaced in that area, causing cardiac remodeling and excessive hemodynamic stress and neurohumorality. Leads to heart failure with stimulation.
- Neurohumoral factors such as angiotensin II and endothelin-1 are well known to contribute to the promotion of cardiac remodeling through elevated blood pressure, cardiomyocyte apoptosis and local inflammation, but cardiac fibroblasts are those factors. It has been reported to secrete.
- Cardiac fibroblasts are also known to play an important role in heart development.
- the cellular processes that interconnect in cardiac fibroblasts form a network of collagen, fibroblasts and muscle cells.
- Cardiomyocyte proliferation is an important process for the formation of thick ventricular walls, but embryonic cardiac fibroblasts have also been reported to promote myocardial mitotic activity through ⁇ 1 integrin signaling. To date, no substance has been known to produce the effect of becoming a cardiac fibroblast dominant.
- cardiac fibroblasts act on heart development and pathogenesis in various ways, the importance of understanding the interaction and basic mechanisms between cardiomyocytes and cardiac fibroblasts was widely recognized.
- the unclear characteristics of cardiac fibroblasts are a bottleneck, and it is necessary to clarify the functions and molecular biological properties of cardiac fibroblasts.
- Heart tissue engineering is a promising method not only for regenerative medicine but also as a tissue model.
- heart tissue based on cell sheets using temperature-responsive culture dishes has been developed.
- heart cell sheet layers of cardiomyocytes, fibroblasts, and endothelial cells from newborn rats on various types of vascular beds have three-dimensionally constructed vascularized viable heart tissue It was reported that this can be done (Non-Patent Documents 1 to 3). Since tissue engineering based on cell sheets does not require any scaffold, a certain amount of extracellular matrix is required to construct the cell sheets.
- Non-patent document 4 a specific amount of fibroblasts built a heart cell sheet when using purified embryonic stem cell-derived cardiomyocytes It is important to do this.
- Non-patent Document 5 cell-cell interactions between cardiomyocytes and non-myocytes are important in cardiac physiology and development. It would be essential to select appropriate fibroblasts to influence the function of the cardiac tissue and to build heart tissue in vitro as a tissue model.
- cardiac fibroblasts have specific functions for cardiomyocytes compared to other fibroblasts and related molecular mechanisms.
- cardiac fibroblasts play an important role in the development or healing of heart development and heart disease
- cardiac fibroblasts that specifically act on cardiac cells such as cardiomyocytes can be used as other fibroblasts. It is necessary to sort and collect.
- Fibroblasts were previously thought of as a homogeneous cell type, but recent studies have a very diverse phenotype, depending on the load on existing organs, tissues, or cells. Has become clear. However, since fibroblasts are not well understood and are the only cells that are classified morphologically, only fibroblasts with a specific function are selected. It is difficult.
- VCAM-1 and ⁇ 4 integrin Kwee et al. Reported that VCAM-1 is expressed in the epicardium, myocardial cells, ventricular septum, etc. on embryonic day 11.5. It has been reported that ⁇ 4 integrin is expressed in almost the same region as VCAM-1, but not expressed in the ventricular septum (Non-patent Document 6). In addition, in VCAM-1-deficient embryos, placental formation was inhibited by embryonic day 11.5, resulting in embryonic lethality, and malformations due to a decrease in the dense layer of ventricular myocardium and ventricular septum were reported. Yang et al.
- Non-patent Document 7 Also reported epicardial defects in ⁇ 4 integrin null embryos on embryonic day 11.5 (Non-patent Document 7). Therefore, VCAM-1 / ⁇ 4 integrin is thought to contribute mainly to the formation of heart cells and epicardium during embryogenesis.
- Shimizu T et al., Fabrication of pulsatile cardiac tissue grafts using a novel 3-dimensional cell sheet manipulation technique and temperature-responsive cell culture surfaces. Circulation research. 2002; 90: e40 Sekiya S, et al., Bioengineered cardiac cell sheet grafts have intrinsic angiogenic potential. Biochemical and biophysical research communications. 2006; 341: 573-582 Shimizu T, et al., Cell sheet engineering for myocardial tissue reconstruction. Biomaterials. 2003; 24: 2309-2316 Matsuura K, et al., Hagiwara N, Zandstra PW, Okano T. Creation of mouse embryonic stem cell-derived cardiac cell sheets. Biomaterials.
- the present invention provides a heart cell culture material that specifically acts on heart cells, and a cell culture substrate in which the wall surface and / or bottom surface of a culture substrate having a wall surface and / or a bottom surface is coated with the heart cell culture material.
- the purpose is to do.
- it aims at providing the artificial organ material obtained by culturing a heart cell using the said heart cell culture material, and its manufacturing method.
- heart cell culture it has been clarified that a functional heart tissue is satisfactorily constructed by using a heart cell culture material containing VCAM-1 protein. Therefore, the heart cell culture material can be coated on the wall surface and / or bottom surface of a culture substrate having a wall surface and / or bottom surface and used as a cell culture substrate. Heart cells cultured using the heart cell culture material can be used as artificial organ materials.
- the present invention is as follows.
- [1] A cardiac cell culture material containing VCAM-1 protein.
- [2] The cardiac cell culture according to [1], wherein the VCAM-1 protein is a cell expressing VCAM-1, a recombinant VCAM-1 protein, or a VCAM-1 protein separated and purified from animal material material.
- [3] The heart cell culture material according to [1] or [2], which is used for culture for constructing heart tissue.
- the heart cell culture material according to [2] or [3], wherein the cell expressing the VCAM-1 protein is a fibroblast expressing the VCAM-1 protein.
- [5] The heart cell culture material according to [4], wherein the fibroblast is derived from a heart.
- [6] The heart cell culture material according to [4] or [5], wherein the fibroblast is derived from the epicardium.
- a method for producing an artificial organ material comprising the step of culturing heart cells together with the heart cell culture material according to [1] to [6].
- a reagent for screening cardiac cell culture material comprising an anti-VCAM-1 antibody.
- heart cell culture material of the present invention By culturing heart cells using the heart cell culture material of the present invention, it is possible to construct a functional heart tissue that can be used in regenerative medicine and tissue models.
- the heart cell culture material can be coated on the wall surface and / or bottom surface of a culture substrate having a wall surface and / or bottom surface and used as a cell culture substrate.
- heart cells or heart tissue obtained by culturing can be used as an artificial organ material.
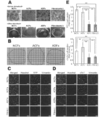
- cTnT emits green fluorescence (FITC: excitation wavelength 488 nm, fluorescence wavelength 530 nm), vimentin emits red fluorescence (cy3: excitation wavelength 512 nm, fluorescence wavelength 552 nm), nucleus is Hoechst 33258 (blue) (Hoechst) 33258: Staining was performed at an excitation wavelength of 352 nm and a fluorescence wavelength of 461 nm. Confocal microscopy observations suggested that there were more cTnT (+) cells in cells co-cultured with NCF or ACF compared to cells in co-culture with fibroblasts (-) or ADF.
- YFP emits green (yellow) fluorescence (YFP: excitation wavelength 514 nm, fluorescence wavelength 527 nm), cTnT emits red fluorescence (cy3: excitation wavelength 512 nm, fluorescence wavelength 552 nm), nucleus is Hoechst 33258 (blue) ) (Hoechst 33258: excitation wavelength 352 nm, fluorescence wavelength 461 nm).
- B Immunofluorescence staining on each cell culture dish on day 5 of culture using a confocal microscope.
- YFP emits green (yellow) fluorescence
- cTnT emits red fluorescence
- nucleus is Hoechst 33258 (blue)
- Hoechst 33258 excitation wavelength 352 nm, fluorescence wavelength 461 nm.
- (c) Number of cardiomyocytes in each culture dish. The bar graph shows an increase in the number of cells of YFP (+) and cTnT (+) (the value of fibroblasts (-) on the first day of culture was taken as 1).
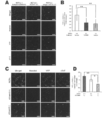
- cTnT emits green fluorescence (FITC: excitation wavelength 488 nm, fluorescence wavelength 530 nm), Ki67 emits red fluorescence (cy3: excitation wavelength 512 nm, fluorescence wavelength 552 nm), nucleus is Hoechst 33258 (blue) (Hoechst) 33258: Staining was performed at an excitation wavelength of 352 nm and a fluorescence wavelength of 461 nm.
- Ki67 (+) or phosphorylated histone 3 (phosphor S10; Phh3) (+) percentage of cardiomyocytes (N 4, ** P ⁇ 0.01) in each culture dish.
- C Immunofluorescence staining observation of phosphorylated histone 3 (phosphor S10; Phh3) positive cardiomyocytes in each culture dish using a confocal microscope.
- cTnT emits green fluorescence (FITC: excitation wavelength 488 nm, fluorescence wavelength 530 nm)
- phosphorylated histone 3 (phosphor S10; Phh3) emits red fluorescence (cy3: excitation wavelength 512 nm, fluorescence wavelength 552 nm)
- the nuclei were stained with Hoechst 33258 (blue) (Hoechst 33258: excitation wavelength 352 nm, fluorescence wavelength 461 nm).
- YFP emits green (yellow) fluorescence
- YFP excitation wavelength 514 nm, fluorescence wavelength 527 nm
- cTnT emits red fluorescence
- nucleus is Hoechst 33258 (blue) )
- Hoechst 33258 excitation wavelength 352 nm, fluorescence wavelength 461 nm.
- H Bar graph shows the increase in the number of YFP (+) and cTnT (+) cells between day 1 and day 5. The number of YFP (+) and cTnT (+) cells on the first day was taken as 1.
- A Comprehensive gene cluster analysis of ADF and NCF (photo). This genetic heat map shows significant differences between ADF and NCF. This map was classified into two groups. The first group consisted of ADF only, and the second group consisted of NCF only.
- C-D Expression level of VCAM-1 protein in NCF and ADF by Western blot analysis.
- E Immunofluorescent staining of VCAM-1 receptor ( ⁇ 4 ⁇ 1) on mESC-derived cardiomyocytes.
- F Western blot analysis of VCAM-1 receptor in mESC-derived cardiomyocytes.
- the following transient overexpressing cell lysate was used as a positive control: Jurkat whole cell lysate. Identification of cardiac growth factor by immunofluorescence staining analysis (photo).
- A-B Observation of immunofluorescence staining for the effect of neutralizing antibody on day 5 cardiomyocytes.
- YFP emits green (yellow) fluorescence
- YFP excitation wavelength 514 nm, fluorescence wavelength 527 nm
- cTnT emits red fluorescence
- nucleus is Hoechst 33258 (blue) )
- Hoechst 33258 excitation wavelength 352 nm, fluorescence wavelength 461 nm
- C-D Observation of immunofluorescent staining of the effect of VCAM-1 soluble protein on cardiomyocytes on day 5 of.
- YFP emits green (yellow) fluorescence
- cTnT emits red fluorescence
- nucleus is Hoechst 33258 (blue) )
- Hoechst 33258 excitation wavelength 352 nm, fluorescence wavelength 461 nm.
- a cardiomyocyte proliferative effect was obtained by culturing with VCAM-1 soluble protein (10 ⁇ g / mL).
- the present invention relates to a heart cell culture material containing VCAM-1.
- the “heart cell culture material” may be any material used for culturing heart cells.
- it may be a reagent added to the culture medium, and it can be used as a material for coating the bottom surface or wall surface of a culture substrate such as a culture container such as a petri dish or flask, but is not limited thereto.
- VCAM-1 vascular cell adhesion molecule-1
- vascular cell adhesion molecule-1 is a known protein as a cell adhesion molecule expressed in vascular endothelial cells and the like.
- accession number NM_001078 of NCBI National Center for Biotechnology Information
- the VCAM-1 protein in the present invention includes one or several amino acids in the VCAM-1 protein, for example, 1-20, 1-15, in addition to VCAM-1 and soluble VCAM-1 expressed on the cell surface. These include various variants having an amino acid sequence in which 1 to 10, 1 to 5 amino acids are deleted, substituted or added, and having an activity equivalent to that of the VCAM-1 protein.
- VCAM-1 in this invention what isolate
- VCAM-1 in the present invention cells expressing VCAM-1 may be used.
- a known cell sorting method may be used. Examples thereof include, but are not limited to, cell sorting methods such as flow cytometry using an anti-VCAM-1 antibody, magnetic bead method, affinity column method, or panning method.
- the anti-VCAM-1 antibody is not particularly limited, but a commercially available antibody may be used, or an antibody prepared by a known method using VCAM-1 as an antigen may be used.
- a monoclonal antibody or a polyclonal antibody may be used as long as cells expressing VCAM-1 can be selected. From the viewpoint of specificity, it is preferable to use a monoclonal antibody.
- the method for screening a heart cell culture material in the present invention comprises a step of obtaining cells, a step of cell sorting using anti-VCAM-1 antibodies, and expressing VCAM-1 as a result of cell sorting. Collecting only the cells determined to be present.
- the type of cell expressing VCAM-1 is not limited as long as VCAM-1 is expressed, but fibroblasts are preferably used.
- the fibroblasts include all cells that eventually become fibroblasts or myofibroblasts. In other words, cells that are in the process of differentiation or maturation, and cannot be identified as fibroblasts or myofibroblasts at that time, but eventually become fibroblasts or myofibroblasts. For example, it is included in the scope of fibroblasts in the present invention.
- fibroblasts there is no limitation on the origin of fibroblasts, and pluripotent stem cells such as ES cells, iPS cells or muse cells, and adult stem cells such as mesenchymal stem cells may be differentiated and used, or primary cells collected from animals
- heart-derived fibroblasts it is preferable to use heart-derived fibroblasts, and epicardial fibroblasts are particularly preferable.
- cell sorting can be omitted by selecting cells known to express VCAM-1.
- the animal from which the fibroblasts are derived can be appropriately selected according to the animal from which the cells to be co-cultured are derived.
- Examples include humans: laboratory animals such as mice, rats, guinea pigs, hamsters, pigs, monkeys, and rabbits: pets such as dogs, cats, and birds: domestic animals such as cows, horses, sheep, and goats.
- fibroblasts are collected from animals, those from any period of fetus, newborn, child, and adult may be used, and there is no particular limitation.
- the heart cell culture material in the present invention includes physiological saline, cell culture solution, cell preservation solution, etc. for maintenance or preservation of VCAM-1 protein or cells expressing VCAM-1 protein.
- a composition may be used, and there is no limitation on what is included in the composition as long as the function of VCAM-1 is not impaired.
- the heart cell culture material in the present invention may be in a liquid or gel form, or may be frozen or lyophilized, and there is no limitation on the state thereof.
- fibroblasts can be included regardless of the expression of VCAM-1 protein.
- Such fibroblasts include all cells that eventually become fibroblasts or myofibroblasts.
- CD31 vascular endothelial cell marker
- VCAM-1 protein expressing cell (cell (number): CD31 expressing cell (cell number) 5: 5 to 8: 2 is more preferred, and 6: 4 to 8: 2 is even more preferred.
- the present invention relates to an artificial organ material obtained by culturing heart cells together with the above heart cell culture material or a method for producing the same. That is, the heart cell culture material in the present invention can be constructed together with heart cells to construct a functional heart tissue that can be used in regenerative medicine and tissue models.
- the heart tissue can be used as an artificial organ material.
- the artificial organ material may be in any form. For example, it may be made into a sheet shape and attached to an injured part of an organ such as the heart, or a sheet may be laminated, or it may be made into a lump shape with a certain thickness using a scaffold and transplanted to an organ defect part. Good.
- Examples of the material of the scaffold include, but are not limited to, hydroxyapatite, atelocollagen, gel, and the like. Moreover, it is possible to perform cell transplantation or use in academic research or the like in the state of cultured cells without using a specific shape. Furthermore, an artificial organ can be produced from the artificial organ material using a 3D printer. The prepared artificial organ can be used not only for transplantation but also for safety pharmacology tests and preclinical studies.
- “constructing heart tissue” means at least one function of the heart that promotes division of heart cells or exhibits uniform pulsation throughout the tissue and can be used as a regenerative medicine or tissue model. It means building a prepared organization.
- the function possessed by the heart includes all of the heart functions known so far, such as autonomic pulsation ability, contraction and relaxation ability, stimulus conduction ability, and hormone secretion ability.
- the cardiac function is not limited to that which only the heart has. For example, the ability to contract and relax also has muscle cells, but other cells have equivalent functions, which does not affect the definition of cardiac function in the present invention.
- the level of the function of the heart is not limited, and any function may be used as long as it meets the purpose of use of the heart tissue. For example, for the purpose of producing an artificial heart, it is necessary to have the ability to contract and relax so that blood can be pumped throughout the body. It is only necessary that the ability can be detected by some means.
- the heart cells used in the artificial organ material or the method for producing the same in the present invention include all cells constituting the heart such as cardiomyocytes, smooth muscle cells, pacemaker cells, and vascular endothelial cells.
- the origin of the heart cells can be appropriately set according to the purpose of use as an artificial organ material.
- human-derived heart cells may be used for transplantation to humans, and mouse-derived ones may be used for the purpose of building a tissue model in mouse experiments.
- heart cells may be used at any time of the fetus, newborn, child, and adult, and are not particularly limited.
- the heart cells in the present invention are preferably prepared from pluripotent stem cells such as ES cells, iPS cells or muse cells, and adult stem cells such as mesenchymal stem cells.
- “Culture” in the present invention is not limited to the culture conditions as long as the heart cell culture material and the heart cells are present in a single culture vessel or are immersed in the same culture solution, It can be performed by a known cell culture method.
- the heart cell culture material is a cell expressing VCAM-1 protein
- the mixing ratio of the cell (cell number) expressing VCAM-1 protein to the heart cell is preferably 3 to 20%, preferably 6 to 18% is more preferable, and 9 to 16% is most preferable.
- the culture solution used for culturing can be appropriately set depending on the type of cells to be cultured.
- DMEM, ⁇ -MEM, RPMI-1640, etc. can be used.
- Nutritional substances such as FCS and FBS, antibiotics and the like may be added to the culture solution.
- the number of days until the desired number of cells and functions are provided may be appropriately set. For example, 1 day, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 8 days, 9 Examples include days, 10 days, 11 days, 12 days, 13 days, 14 days, 1 month, 2 months, 3 months, 4 months, 5 months, 6 months, and the like.
- the culture temperature can be appropriately set according to the type of cells to be cultured, and is, for example, 10 to 60 ° C, preferably 20 to 50 ° C, more preferably 30 to 40 ° C.
- the production method of the present invention may further include a step of collecting the cultured cells.
- the “cultured cells” may include both fibroblasts and heart cells, or may include only heart cells.
- the cells may be recovered by detaching with a protease such as trypsin, but using a temperature-responsive culture dish capable of detaching cells while retaining the extracellular matrix, It is preferable to detach and collect cells by temperature change.
- Example 1 Materials and methods ⁇ Animals and reagents> Wild type C57BL / 6 mice were purchased from Japan SLC (Shizuoka). B6 Cg-Tg (CAG-DsRed * MST) 1Nagy / J mice were purchased from Jackson Laboratory (Bar Harbor, Maine, USA). All experimental protocols were approved by the Tokyo Women's Medical University Animal Experiment Committee.
- the antibodies described below were used for immunocytochemistry, Western blot and flow cytometry analysis: rabbit polyclonal anti-discoidin domain receptor tyrosine kinase 2 (DDR2) (GeneTex, Irvine, CA); guinea pig monoclonal anti-vimentin (Progen, Heidelberg, Germany); mouse monoclonal anti-NG2 (Millipore, Temecula, CA); rabbit polyclonal anti- ⁇ smooth muscle actin (Abcam, Cambridge, UK); mouse monoclonal anti-cardiac troponin T (cTnT) (Thermo Scientific, Rockford, IL); mouse Monoclonal anti-cytokeratin 11 (EXBIO, Nad Safinou, CZ); Rabbit polyclonal anti-Ki67 (Abcam, Cambridge, UK); Rabbit polyclonal anti-histone H3 (phosphor S10) (Abcam, Cambridge, UK); Rat monoclonal anti-integrin ⁇ 4 / ⁇ 1 (Abcam, Cambridge, UK).
- VCAM-1 / CD106 Fc chimera Recombinant mouse VCAM-1 / CD106 Fc chimera (R & D systems, Minneapolis, Minn.) All reagents were purchased from Sigma-Aldrich unless otherwise specified. Secondary antibodies were purchased from Jackson ImmunoResearch Laboratories (West Grove, PA).
- ⁇ Mouse ES cell culture> For the maintenance, cardiomyocyte differentiation, and purification of mouse ES cells (mESC) expressing neomycin phosphotransferase gene under the control of ⁇ -myosin heavy chain promoter (Matsuura K, et al., Biomaterials. 2011; 32: 7355-7362). Briefly, trypsinized ES cells were seeded in spinner flasks (Integra Biosciences, Zizers, Switzerland) at 5 ⁇ 10 4 cells / mL (125 mL / flask) for cardiomyocyte induction and cardiomyocyte purification. The cells were cultured in DMEM supplemented with 10% FBS for 10 days, and then differentiated cells were treated with neomycin for 8 days.
- spinner flasks Integra Biosciences, Zizers, Switzerland
- Fibroblasts were obtained from wild type C57BL / 6 mice (newborn, 1 day old; adult, 10-12 weeks old).
- Neonatal mouse cardiac fibroblasts NCF were obtained from the heart of a neonatal mouse (1 day old) according to the method described in the previous report (Matsuura K, et al., Biomaterials. 2011; 32: 7355-7362). It was. NCF passaged 3 times was used for the experiment.
- Adult mouse cardiac fibroblasts (ACF) were obtained from the heart of adult mice (10-12 weeks old) by the following explant culture method. First, the heart was washed with PBS ( ⁇ ) and cut to about 5 mm 2 .
- the cut pieces were covered with a sterilized cover glass and cultured in a 10 cm culture dish with 10% FBS-added DMEM. Two weeks after the start of culture, the cells were detached with 0.25% trypsin / EDTA and subcultured in another 10 cm culture dish. ACF passaged three times was used for the experiment.
- Adult mouse dorsal fibroblasts (ADF) were obtained from dorsal skin tissue of adult mice (10-12 weeks old). First, the skin tissue was collected and treated with Dispase I [1000 U / mL] (Aidia Co., Ltd.) overnight at 4 ° C. It was then cut to about 1mm 2 the organization.
- the cut pieces were covered with a sterilized cover glass and cultured in a 10 cm culture dish with 10% FBS-added DMEM. Two weeks after the start of culture, the cells were detached with 0.25% trypsin / EDTA and subcultured in another 10 cm culture dish. ADF passaged 3 times was used for the experiment. In some experiments, NCF and ADF were obtained from B6.Cg-Tg (CAG-DsRed * MST) 1Nagy / J mice (newborn: 1 day old, adult: 10 weeks old) in the same manner as above. It was.
- ⁇ Cell sheet production> Before seeding the cells, the surface of a temperature-responsive culture dish (UpCell; cell seed) was coated with FBS for 2 hours. mESC-derived cardiomyocytes and each type of fibroblast were co-cultured in 10% FBS-added DMEM at a ratio of 8: 2 (3.2 ⁇ 10 5 cells / cm 2 ). After 5 days in culture, the cells were cultured at 20 ° C. in order to peel off the cell sheet. Sample bright field images were obtained with Nikon ECLIPSE Ti.
- BD Cytofix / Cytoperm Buffer (BD Pharmingen, Franklin Lakes, NJ); BD Perm / Wash Buffer (10X) (BD Pharmingen, Franklin Lakes, NJ); BD CytopermPlus Buffer (10X) ( BD Pharmingen, Franklin Lakes, NJ); BrdU (10 mg / mL) (BD Pharmingen, Franklin Lakes, NJ); DNase (BD Pharmingen, Franklin Lakes, NJ).
- RNA extraction and comprehensive gene analysis> Total RNA was extracted using TRIzol (Invitrogen, Carlsbad, CA) according to the manufacturer's instructions attached to the product. Total RNA was further purified using Qiagen RNeasy Mini Kit (QIAGEN, Valencia, CA) according to the manufacturer's instructions attached to the product. RNA quantity and quality was determined using the Nanodrop ND-1000 spectrophotometer (Thermo Fisher Scientific Inc., Waltham, Mass.) And Agilent Bioanalyzer (Agilent Technologies, Palo Alto, Calif.) As recommended.
- the cDNA product was then used as a template in in vitro transcription to produce fluorescent cRNA.
- the cDNA product was mixed with transcription master mix in the presence of T7 RNA polymerase and Cy3 labeled CTP and incubated at 40 ° C. for 2 hours.
- the labeled cRNA was purified using QIAGEN's RNeasy mini spin columns and extracted with 30 ⁇ l of nuclease-free water. After amplification and labeling, the amount of cRNA and cyanine incorporation was determined using a Nanodrop® ND-1000 spectrophotometer and an Agilent® Bioanalyzer.
- Cy3-labeled cRNA ⁇ 1.65 ⁇ g was fragmented and hybridized to Agilent Mouse GE 4 ⁇ 44 Kv2 Microarray (Design ID: 026026655) at 65 ° C. for 17 hours. After washing, the microarray was scanned using an Agilent DNA microarray scanner. For microarray data analysis, the intensity value of each scanned feature was quantified using Agilent feature extraction software version 0.7 10.7.3.1 with background reduction.
- ⁇ Quantitative real-time PCR analysis> Complementary DNA was prepared from total RNA using the High Capacity cDNA Reverse Transcription Kit (Applied biosystems). VPCR1 Gene Expression Assays (life Technologies) were used as PCR-related primers. Each RT-PCR was performed on an iCycler (BIO-RAD) for 10 minutes at 25 ° C., 120 minutes at 37 ° C., and 5 seconds at 85 ° C. From each sample, a cDNA template (1 ⁇ g) was used. TaqMan probe real-time PCR experiments were performed using TaqMan Gene Expression Assays (Applied biosystems). All experiments were performed in triplicate.
- ⁇ Neutralizing antibody assay> The following antibodies and culture dishes were used for neutralizing antibody assays: anti-VCAM-1 (LifeSpan Biosciences, Seattle, WA); goat IgG isotype control (LifeSpan Biosciences, Seattle, WA). Cell culture insert 24-well plate. Pore size 0.4 ⁇ m translucent High Density PET Membrane (BD Pharmingen, Franklin Lakes, NJ). After pretreatment with 10 ⁇ g / mL antibody for 30 minutes, fibroblasts were seeded on the upper layer of the insert culture dish (2.4 ⁇ 10 5 cells). mESC-derived myocardium was seeded in the lower layer (4.8 ⁇ 10 5 cells). The culture medium containing 10 ⁇ g / mL antibody was changed daily until the fifth day.
- the optimal ratio of cardiomyocytes to fibroblasts is 8: 2 ( Biomaterials. 2011; 32: 7355-7362), mESC-derived cardiomyocytes and three types of fibroblasts (ACF, ADF and NCF) were used to produce a heart cell sheet in a temperature-responsive culture dish UpCell.
- ACF fibroblasts
- ADF fibroblasts
- the number of YFP (+) cells and the number of cardiac troponin T (cTnT) (+) cells indicate mESC-derived cardiomyocytes.
- the number of cells in the cell sheet co-cultured with ACF or NCF was higher than that in the cell sheet co-cultured with ADF.
- the number of myocardium in the cell sheet co-cultured with ADF was comparable to that cultured under conditions without fibroblasts.
- the number of cardiomyocytes on the 5th day was the same as that on the 1st day under the condition of coculture with ADF or cardiomyocytes alone.
- Time-lapse image analysis of YFP (+) cardiomyocytes and DsRed mouse-derived fibroblasts showed that in NCF co-culture, cardiomyocytes migrated and proliferated to form a mutual network configuration. In contrast, in ADF co-culture, cardiomyocyte proliferation was small and a network configuration was not formed.
- cardiomyocytes between each condition were confirmed by immunofluorescent staining.
- FIGS. 4A-D the percentage of cardiomyocytes of Ki67 (+) cells and phosphorylated histone 3 (PHH3) (+) under NCF co-culture was cultured with ADF co-culture or with cardiomyocytes alone. Significantly higher than those.
- the BrdU incorporation assay also showed a significant increase in the percentage of cardiomyocytes proliferated under NCF co-culture compared to those under ADF co-culture or cultured with cardiomyocytes alone (FIGS. 4E and F). ).
- mESC-derived cardiomyocytes and NCF were cultured using cell culture inserts.
- NCF was cultured in the upper layer and cardiomyocytes were cultured in the lower layer.
- the number of cardiomyocytes on day 5 was significantly higher than that on day 1 (FIG. 4G).
- the increase in the number of cardiomyocytes between day 1 and day 5 ( ⁇ 1.8 times) (FIG. 4H) was lower than that under co-culture conditions ( ⁇ 2.5 times).
- ⁇ VCAM-1-dependent cardiomyocyte proliferation in co-culture with cardiac fibroblasts Since integrin ⁇ 4 ⁇ 1 is known as the major co-receptor of VCAM-1, the expression of integrin ⁇ 4 ⁇ 1 in mESC-derived cardiomyocytes was examined. As shown in FIGS. 5E and F, almost all mESC-derived cardiomyocytes showed integrin ⁇ 4 ⁇ 1 expression. Next, it was elucidated whether VCAM-1 contributed to cardiac fibroblast-induced cardiomyocyte proliferation using neutralizing antibodies. After NCF was pretreated with anti-VCAM-1 antibody, NCF and mESC-derived cardiomyocytes were cultured using cell culture inserts. Anti-VCAM-1 antibody treatment significantly suppressed cardiac fibroblast-induced cardiomyocyte proliferation (FIGS.
- VCAM-1 VCAM-1 recombinant protein
- FIGS. 6C and D VCAM-1 treatment increased the number of cardiomyocytes compared to the control.
- VCAM-1 positive cells In order to confirm the importance of VCAM-1 positive cells in the construction of a functional heart cell sheet, the ratio of VCAM-1 positive cells in living cardiac fibroblasts was measured. Cardiac fibroblasts were dissected from newborn mice (1 day old) of mouse C57 / BL6, and skin fibroblasts were dissected from adult mice (10-12 weeks of age) and collected. Each fibroblast was cultured to adhere to passage number 3 to obtain a cell volume of 1 ⁇ 10 7 cells per condition. Passage number 3 is the same as the culture condition of the myocardial tissue prepared from the cell sheet.
- FIGS. 7A-C The results of cardiac fibroblasts (NCF) are shown in FIGS. 7A-C.
- the ratio of VCAM-1 positive cells in NCF was found to be about 60% (FIG. 7A: 66.57%, FIG. 7B: 58.95%, FIG. 7C: 54.73%).
- the proportion of VCAM-1 positive cells in skin fibroblasts (ADF) was about 5%, and the proportion of VCAM-1 positive cells in NCF was found to be significantly higher than that of ADF. (P ⁇ 0.001).
- VCAM-1-positive cardiac fibroblasts are thought to originate from epicardial cells from a developmental perspective. Fibroblasts are classified from a embryological perspective, and functional tissues are classified. We have suggested that it is effective to perform functional classification instead of morphological classification as a cell source for construction.
- CD31 vascular endothelial cell marker
- EMT epithelial-mesenchymal transition
- EndMT endothelial-mesenchymal transition
- kidney fibroblasts that differentiate from vascular endothelial cells via EndMT as well as cardiac fibroblasts express CD31 (J Am Soc Nephrol 19: 2282-2287, 2008), and NCF expresses CD31. It can be one of the grounds for expression.
- cardiac fibroblasts not dermal fibroblasts, promoted the proliferation of mouse embryonic stem cell (mESC) -derived cardiomyocytes and contributed to the construction of more functional cardiac cell sheets . Furthermore, cardiac fibroblasts express VCAM-1 more abundantly than dermal fibroblasts, and the VCAM-1 in cardiac fibroblasts is functionally biologically engineered with the proliferation of cardiac cells Have been shown to play an important role in the construction of the cardiac tissue.
- mESC mouse embryonic stem cell
- a functional heart tissue is satisfactorily constructed by culturing using the heart cell culture material in the present invention.
- Heart cells obtained by the culture can be used as regenerative medicine such as transplantation or artificial organ materials such as heart tissue models.
Abstract
Description
しかし、線維芽細胞は、その機能が良く分かっておらず、形態学的に分類されているほぼ唯一の細胞であるため、線維芽細胞の中から、ある特定の機能を持つものだけを選別することは困難である。
[1]VCAM-1蛋白質を含む、心臓細胞培養材料。
[2]上記VCAM-1蛋白質が、動物材料から分離精製したVCAM-1、VCAM-1組み換え蛋白質、又は、VCAM-1蛋白質を発現している細胞である、[1]に記載の心臓細胞培養材料。
[3]心臓組織を構築するための培養に使用されるものである、[1]または[2]に記載の心臓細胞培養材料。
[4]前記VCAM-1蛋白質を発現している細胞が、VCAM-1蛋白質を発現している線維芽細胞である、[2]または[3]に記載の心臓細胞培養材料。
[5]前記線維芽細胞が、心臓由来のものである、[4]に記載の心臓細胞培養材料。
[6]前記線維芽細胞が、心外膜由来のものである、[4]または[5]に記載の心臓細胞培養材料。
[7] [1]~[6]に記載の心臓細胞培養材料で、壁面及び/又は底面を有する培養基材の壁面及び/又は底面をコーティングした、細胞培養基材。
[8] [1]~[6]に記載の心臓細胞培養材料と共に、心臓細胞を培養することにより得られる、人工臓器材料。
[9] [1]~[6]に記載の心臓細胞培養材料と共に、心臓細胞を培養するステップを含む、人工臓器材料の製造方法。
[10] 抗VCAM-1抗体を含む、心臓細胞培養材料スクリーニング用試薬。
[11] VCAM-1蛋白質を発現している心臓由来線維芽細胞。
[12] 培養した細胞を培養基材から剥離し回収するステップをさらに含む、[9]に記載の人工臓器材料の製造方法。
[13] 前記培養基材が温度応答性培養皿であり、前記剥離が温度変化により行われる、[12]に記載の人工臓器材料の製造方法。
抗VCAM-1抗体は、特に制限されないが、市販のものを用いてもよいし、VCAM-1を抗原として既知の方法により作製したものを用いてもよい。また、VCAM-1を発現している細胞を選別できる限りにおいて、モノクローナル抗体又はポリクローナル抗体のいずれを用いてもよいが、特異性の観点から、モノクローナル抗体を用いるのが好ましい。
培養期間は、所望の細胞数や機能が備わるまでの日数を適宜設定すればよいが、たとえば、1日、2日、3日、4日、5日、6日、7日、8日、9日、10日、11日、12日、13日、14日、1ヵ月、2か月、3か月、4か月、5か月、6か月等の期間が挙げられる。培養温度は、培養する細胞の種類に合わせて適宜設定可能であるが、たとえば、10~60℃、好ましくは20~50℃、より好ましくは30~40℃である。
1.材料と方法
<動物と試薬>
ワイルドタイプのC57BL/6マウスは、日本SLC(静岡)から購入した。B6 Cg-Tg(CAG-DsRed*MST) 1Nagy/Jマウスは、ジャクソンラボラトリー(米国メイン州バーハーバー)から購入した。全ての実験プロトコルは、東京女子医科大学の動物実験委員会に承認された。後述する抗体は、免疫細胞化学、ウエスタンブロット及びフローサイトメトリー解析に用いた:ウサギポリクローナル抗ディスコイジンドメインレセプターチロシンキナーゼ2(DDR2)(GeneTex, Irvine, CA);モルモットモノクローナル抗ビメンチン(Progen, Heidelberg, Germany);マウスモノクローナル抗NG2(Millipore, Temecula, CA);ウサギポリクローナル抗α平滑筋アクチン(Abcam, Cambridge, UK);マウスモノクローナル抗心筋型トロポニンT (cTnT) (Thermo Scientific, Rockford, IL);マウスモノクローナル抗サイトケラチン11 (EXBIO, Nad Safinou, CZ);ウサギポリクローナル抗Ki67 (Abcam, Cambridge, UK);ウサギポリクローナル抗ヒストンH3 (phosphor S10) (Abcam, Cambridge, UK);ラットモノクローナル抗インテグリンα4/β1 (Abcam, Cambridge, UK)。組み換えマウスVCAM-1/CD106 Fc キメラ (R&D systems, Minneapolis, MN)特に指定がない限り、全ての試薬はシグマアルドリッチから購入した。二次抗体はJackson ImmunoResearch Laboratories (West Grove, PA)から購入した。
α-ミオシン重鎖プロモーターの制御下でのネオマイシンホスフォトランスフェラーゼ遺伝子を発現するマウスES細胞(mESC)の維持、心筋細胞分化、及び精製については、既報(Matsuura K,et al., Biomaterials. 2011;32:7355-7362)に記載の方法に従った。簡潔に言うと、心筋細胞への誘導と心筋細胞精製のために、トリプシン処理したES細胞をスピナーフラスコ(Integra Biosciences, Zizers, Switzerland)に5 × 104細胞/mL (計125 mL/フラスコ)播種し、10% FBS添加DMEMで10日間培養し、その後分化した細胞を8日間ネオマイシンで処理した。
線維芽細胞は ワイルドタイプのC57BL/6マウス(新生仔, 1日齢; 成体, 10-12週齢)から得た。
新生仔マウス心臓線維芽細胞(NCF)は、新生仔マウス(1日齢)の心臓から、既報(Matsuura K,et al., Biomaterials. 2011;32:7355-7362)に記載の方法に従って得られた。3回継代したNCFが実験に使用された。
成体マウス心臓線維芽細胞(ACF)は、成体マウス(10-12週齢)の心臓から、下記の外植片培養方法により得られた。まず、心臓をPBS(-)で洗浄し、約5 mm2にカットした。カット片は滅菌したカバーガラスで覆い、10% FBS添加DMEMで10 cm培養皿にて培養した。培養開始から2週間後、細胞を0.25% トリプシン/EDTAで剥離し、他の10cm培養皿で継代培養した。3回継代したACFが実験に使用された。
成体マウス背部線維芽細胞(ADF)は、成体マウス(10-12週齢)の背部皮膚組織から得られた。まず、皮膚組織を採取し、ディスパーゼI [1000 U / mL] (エーディア株式会社) で一晩4℃にて処理した。次に、組織を約1mm2にカットした。カット片は、滅菌したカバーガラスで覆い、10% FBS添加DMEMで10 cm培養皿にて培養した。培養開始から2週間後、細胞を0.25% トリプシン/EDTAで剥離し、他の10cm培養皿で継代培養した。3回継代したADFが実験に使用された。
いくつかの実験では、NCFとADFは、B6.Cg-Tg (CAG-DsRed*MST) 1Nagy/J mice (新生仔:1日齢、成体: 10週齢)から、上記と同様の方法で得られた。
細胞を播種する前に、温度応答性培養皿(UpCell; セルシード)の表面を、FBSで2時間コートした。mESC由来心筋細胞とそれぞれの種類の線維芽細胞とは、8:2の割合で、10% FBS添加DMEMにて共培養した(3.2 x105細胞/cm2)。培養5日後、細胞シートを剥離するために、細胞を20℃で培養した。サンプルの明視野像はNikon ECLIPSE Tiにより得られた。
細胞シートの電気生理学的解析は、既報(Matsuura K,et al., Biomaterials. 2011;32:7355-7362)に記載のとおり、多電極アレイ(multi-electrode array:MED)システム(Alpha MED Sciences, Osaka, Japan)による細胞外活動電位測定から得られた。
細胞を、既報(Matsuura K,et al., Biomaterials. 2011;32:7355-7362)に記載のとおり、4%パラホルムアルデヒドで固定し、免疫染色を行った。サンプルの染色像がImageXpress Ultra Confocal High Content Screening System (Molecular Devices, CA, USA)により得られた。画像解析データはMetaExpress software (Molecular Devices, CA, USA)によって得られた。
培養細胞(5×105細胞)は、細胞培養液中の最終濃度が10 μMでBrdU染色された。FACS解析用のBrdU 染色は、BrdU Flow Kits Instruction Manual (BD Pharmingen, Franklin Lakes, NJ)に記載のとおりに行われた。簡潔に言うと、細胞は固定され、BD Cytofix/Cytoperm バッファーにて細胞膜透過処理された。それから取り込まれたBrdUをDNaseに曝した。BrdU染色は、APC-anti-BrdU antibody (BD Pharmingen, Franklin Lakes, NJ)を使用して行われた。サンプルはGallios (Beckman Coulter, Brea, CA)で解析された。下記の試薬が解析に使用された:BD Cytofix/Cytoperm Buffer (BD Pharmingen, Franklin Lakes,NJ); BD Perm/Wash Buffer (10X) (BD Pharmingen, Franklin Lakes, NJ); BD CytopermPlus Buffer (10X) (BD Pharmingen, Franklin Lakes, NJ); BrdU (10 mg/mL) (BD Pharmingen, Franklin Lakes, NJ); DNase (BD Pharmingen, Franklin Lakes, NJ)。
サンプルは5日間、5% CO2、37℃の条件下にて、BZ-9000 Fluorescence Microscope (Keyence, Osaka, Japan)により観察された。
トータルRNAは、TRIzol(Invitrogen, Carlsbad, CA)を製品に添付されたメーカー指示書に従って使用して、抽出した。トータルRNAは、Qiagen RNeasy Mini Kit (QIAGEN, Valencia, CA)を製品に添付されたメーカー指示書に従って使用して、さらに精製した。
RNAの量と質は、Nanodrop ND-1000 spectrophotometer (Thermo Fisher Scientific Inc., Waltham, MA) とAgilent Bioanalyzer (Agilent Technologies, Palo Alto, CA)を推奨どおりに使用して決定した。
転写物の変化は比較法により定量された。我々は、この実験において、シグナル強度2倍以上の変化を遺伝子発現の顕著な変化と定義した。
相補的DNAは、High Capacity cDNA Reverse Transcription Kit (Applied biosystems)を用いて、トータルRNAから作製された。PCR関連プライマーとしては、VCAM1 Gene Expression Assays(life Technologies)が使用された。各々のRT-PCRは、iCycler (BIO-RAD)にて、10分25 ℃、120分37 ℃、及び5秒85℃にて行われた。各々の試料から、cDNAテンプレート(1μg)が使用された。TaqManプローブリアルタイムPCR実験は、TaqMan Gene Expression Assays (Applied biosystems)を用いて行われた。全ての実験は三重測定で行われた。試料は7300 Real Time PCR System (Applied Biosystems)にて40回のサイクルで、以下のように行われた。50℃2分、95℃10分、その後95℃15秒及び60℃1分の40サイクルが続いた。相対的な定量は、内在性コントロールとしてのGapDH遺伝子を使用した定量的リアルタイムPCRのためのΔΔCT法に従って計算された。
NCF及びADFは、Laemmliサンプルバッファー(BIO-RAD, CA, USA)、プロテアーゼインヒビター(Boehringer Mannheim, Indianapolis, IN)、及び2-メルカプトエタノール(Wako Pure Chemical Industries, Japan)において溶解した。試料は4%-12% Bis-Tris Gels (Life Technologies, MD, United States)にて分離し、iBlot 7-Minute Blotting System (Life technologies, MD, United States)を使用してiBlot Transfer Stack, nitrocellulose, regular-size (Life technologies, MD, United States)に電気的にトランスファーを行い、そして化学発光解析のためにAmersham ECL Prime Western Blotting Detection Reagent (GE Healthcare, PA, United States)を使用して処理した。バンド強度はLAS4000 (Fujifilm, Tokyo, Japan)及びNIH image software (version 1.46r)を使用して解析された。下記の一過性過剰発現細胞溶解物がポジティブコントロールとして使用された。Col11a1を発現するK562 (ヒト赤骨髄芽球様白血病細胞系)(Abcam, CB, UK); Vcam-1を発現するSol8 (SantaCruz, CA, USA); β1/CD29を発現するITGB1 293T (Abnova, Taipei, Taiwan); integrin α4β1を発現するJurkat全細胞溶解物(SantaCruz, CA, USA)。
下記の抗体及び培養皿は、中和抗体アッセイに使用された:抗VCAM-1 (LifeSpan Biosciences, Seattle, WA); ヤギIgG アイソタイプコントロール(LifeSpan Biosciences, Seattle, WA)。セルカルチャーインサート24ウェルプレート。ポアサイズ0.4μm半透明High Density PET Membrane (BD Pharmingen, Franklin Lakes, NJ)。
10μg/mLの抗体で30分間前処理した後、線維芽細胞はインサートカルチャーディッシュの上層に播種された(2.4 x105 cells)。mESC由来心筋は下層に播種された(4.8 x105 cells)。10μg/mLの抗体を含む培養液は、5日目まで毎日交換された。
全てのデータは平均 ± SDで表された。異なる群間の変動有意性は、一元配置分散分析により決定した。その後、2つの群間の差は、Statcel ソフトウェアを使用してTukey-Kramer Multiple Comparison Testにより決定した。0.05より低いp値は有意に異なると見なされた。
<mESC由来心筋細胞及び線維芽細胞を使用した細胞シート作製>
まず我々は、共培養実験に使用する予定の細胞の特性を評価した。位相差像は、新生仔マウスの心臓、成体マウスの心臓、または成体マウスの皮膚組織から分離した細胞は線維芽細胞様の形態を示すことを明らかにした(図1A)。線維芽細胞に特異的な抗体がないので、DDR2(CD167b)、ビメンチン、αSMAなどの線維芽細胞において発現することが知られている蛋白質の発現を調べてみた。図1B-Eが示すとおり、ほとんどすべての各々のタイプの細胞がDDR2、ビメンチン、αSMAを発現していたが、カルポニン(平滑筋細胞マーカー)、サイトケラチン(上皮細胞マーカー)、及びNG2(ペリサイトマーカー)は発現していなかった。これらの発見に基づいて、我々はこれらの細胞を、続く実験において線維芽細胞として使用した。
心臓由来の線維芽細胞と共培養した細胞シートにおける心筋細胞数と、皮膚由来の線維芽細胞と共培養した細胞シートにおける心筋細胞数が異なる原因を調べるために、共培養開始から1日目と5日目における心筋細胞数を検査した(図3A-C)。一日目においては、心筋細胞数は各条件間で同じであったことから、各々のタイプの線維芽細胞は播種後の心筋細胞の初期接着には影響しないことが示唆された。ACF又はNCFとの共培養において、5日目におけるYFP(+)とcTnT(+)の心筋細胞数は、有意に1日目よりも多かった。一方、ADFと共培養又は心筋細胞のみの条件下では、5日目の心筋細胞数は1日目と同じであった。YFP(+)心筋細胞及びDsRedマウス由来線維芽細胞の低速度撮影像解析は、NCF共培養において、心筋細胞が移動して増殖し、相互ネットワーク構成を形成することを示した。これに対してADF共培養においては、心筋細胞の増殖は少なく、ネットワーク構成を形成しなかった。これらの発見は、皮膚由来ではなく心臓由来の線維芽細胞が共培養条件下でmESC由来の心筋細胞の増殖を誘導しているかもしれないことを示唆している。
上記の効果関連のものを左右する因子を同定するために、NCFとADFとの間でマイクロアレイ解析を使用して、網羅的遺伝子解析を行った。図5Aに示されるように、遺伝子発現において多くの異なる遺伝子がNCFとADFとの間で観察された。NCFでは、500以上の遺伝子の発現が、ADFと比較して10倍以上促進されていることが示された。リストの中で心血管関連遺伝子を選択すると、20遺伝子が残った。さらに、ノックアウトマウスモデルにおいて、心臓の発生に異常をきたし、胎生致死フェノタイプを示すことが報告された遺伝子であり、可溶性因子と接着因子の2つのフォームで作用する遺伝子を選択したところ、Vcam-1が残った。定量的RT-PCRとウエスタンブロット解析により、ADF共培養と比べて、NCF共培養では、Vcam-1発現が促進されていることを確認した(図5B-D)。
インテグリンα4β1はVCAM-1の主要な補助受容体として知られているので、mESC由来心筋細胞におけるインテグリンα4β1の発現を調べた。図5E及びFに示すとおり、ほぼ全てのmESC由来心筋細胞はインテグリンα4β1の発現を示した。
次に、VCAM-1が心臓線維芽細胞誘導性心筋細胞増殖に寄与しているかどうかを、中和抗体を使用して解明した。NCFを抗VCAM-1抗体により前処理した後、NCFとmESC由来心筋細胞をセルカルチャーインサートを使用して培養した。抗VCAM-1抗体処理は、心臓線維芽細胞誘導性の心筋細胞数の増殖を、有意に抑制した(図6A及びB)。
最後に、VCAM-1の心筋細胞増殖に対する直接的な効果を解明した。培養開始から1日後、心筋細胞にVCAM-1組み換え蛋白質を添加し、5日目まで続けた。図6C及びDに示されるように、VCAM-1処理により心筋細胞数がコントロールと比べて増加した。これらの発見は、心臓由来線維芽細胞は心筋細胞におけるVCAM-1とインテグリンα4β1経路で心筋細胞増殖を誘導するかもしれないことを、示唆している。
マウスC57/BL6の新生児マウス(1日齢)から心臓線維芽細胞を、成体マウス(10-12週齢)から皮膚線維芽細胞を解剖し、回収した。各々の線維芽細胞を継代数3まで接着培養し、1条件につき1×107細胞の細胞量を得た。なお、継代数3は、細胞シートにより作製された上記心筋組織の培養条件と同一である。
両者の線維芽細胞をGoat policlonal anti-VCAM-1抗体(R&D systems, Minneapolis, MN)で一次免疫蛍光染色し、Alexa Fluor 488 Donkey anti-goat IgG(Life Technologies, MD, United States)で二次免疫蛍光染色を行った。その後、Gallios(Beckman Coulter, Brea, CA)でFACS解析を行い、VCAM-1陽性細胞率を測定した(N = 3)。優位差の算出は、Student's t-testにより算出した。
Claims (9)
- VCAM-1蛋白質を含む、心臓細胞培養材料。
- 上記VCAM-1蛋白質が、動物材料から分離精製したVCAM-1蛋白質、VCAM-1組み換え蛋白質、又は、VCAM-1蛋白質を発現している細胞である、請求項1に記載の心臓細胞培養材料。
- 心臓組織を構築するための培養に使用されるものである、請求項1または2に記載の心臓細胞培養材料。
- 前記VCAM-1蛋白質を発現している細胞が、VCAM-1蛋白質を発現している線維芽細胞である、請求項2または3に記載の心臓細胞培養材料。
- 前記線維芽細胞が、心臓由来のものである、請求項4に記載の心臓細胞培養材料。
- 前記線維芽細胞が、心外膜由来のものである、請求項4または5に記載の心臓細胞培養材料。
- 請求項1~6に記載の心臓細胞培養材料で、壁面及び/又は底面を有する培養基材の壁面及び/又は底面をコーティングした、細胞培養基材。
- 請求項1~6に記載の心臓細胞培養材料と共に、心臓細胞を培養することにより得られる、人工臓器材料。
- 請求項1~6に記載の心臓細胞培養材料と共に、心臓細胞を培養するステップを含む、人工臓器材料の製造方法。
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP15819754.1A EP3168297B1 (en) | 2014-07-11 | 2015-01-05 | Cardiac cell culture material |
| AU2015286462A AU2015286462B2 (en) | 2014-07-11 | 2015-01-05 | Cardiac cell culture material |
| CA2954743A CA2954743C (en) | 2014-07-11 | 2015-01-05 | Cardiac cell culture material |
| KR1020177003798A KR102263824B1 (ko) | 2014-07-11 | 2015-01-05 | 심장 세포 배양 재료 |
| CN201580037730.1A CN106661552B (zh) | 2014-07-11 | 2015-01-05 | 心脏细胞培养材料 |
| JP2015138613A JP6241893B2 (ja) | 2014-07-11 | 2015-07-10 | 心臓細胞培養材料 |
| US15/401,832 US20170112880A1 (en) | 2014-07-11 | 2017-01-09 | Cardiac cell culture material |
| JP2017208721A JP2018029611A (ja) | 2014-07-11 | 2017-10-30 | 心臓細胞培養材料 |
| US16/106,723 US11801267B2 (en) | 2014-07-11 | 2018-08-21 | Cardiac cell culture material |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014-142804 | 2014-07-11 | ||
| JP2014142804 | 2014-07-11 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/401,832 Continuation-In-Part US20170112880A1 (en) | 2014-07-11 | 2017-01-09 | Cardiac cell culture material |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016006262A1 true WO2016006262A1 (ja) | 2016-01-14 |
Family
ID=55063910
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/050028 WO2016006262A1 (ja) | 2014-07-11 | 2015-01-05 | 心臓細胞培養材料 |
Country Status (8)
| Country | Link |
|---|---|
| US (2) | US20170112880A1 (ja) |
| EP (1) | EP3168297B1 (ja) |
| JP (2) | JP6241893B2 (ja) |
| KR (1) | KR102263824B1 (ja) |
| CN (2) | CN111876371A (ja) |
| AU (1) | AU2015286462B2 (ja) |
| CA (1) | CA2954743C (ja) |
| WO (1) | WO2016006262A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109890398A (zh) * | 2017-02-24 | 2019-06-14 | 玛土撤拉有限公司 | 可用于治疗心脏疾病并含有成纤维细胞的注射用组合物以及用于生产用于治疗用途的成纤维细胞的方法 |
| WO2020045547A1 (ja) | 2018-08-29 | 2020-03-05 | 株式会社メトセラ | 線維芽細胞の製造方法及びg-csf陽性線維芽細胞集団 |
| WO2021177387A1 (ja) | 2020-03-04 | 2021-09-10 | 株式会社メトセラ | エリスロポエチン産生能が亢進された線維芽細胞 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102200341B1 (ko) * | 2018-12-21 | 2021-01-08 | 고려대학교 산학협력단 | 지방줄기세포 시트를 함유하는 심장조직 모사 심장이식용 시트 및 이의 제조방법 |
| WO2020203850A1 (ja) | 2019-03-29 | 2020-10-08 | 国立大学法人長崎大学 | 培養組織及びその製造方法 |
| CN113498434A (zh) * | 2020-01-22 | 2021-10-12 | 京东方科技集团股份有限公司 | 间充质干细胞膜片及其用途 |
| KR102283340B1 (ko) * | 2020-03-27 | 2021-07-30 | 서울대학교산학협력단 | 심근 직분화를 위한 심장 모사 세포 배양장치 및 이를 이용한 세포 분화 방법 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2003901668A0 (en) * | 2003-03-28 | 2003-05-01 | Medvet Science Pty. Ltd. | Non-haemopoietic precursor cells |
| CA2453438C (en) * | 2001-07-12 | 2016-04-05 | Geron Corporation | Cells of the cardiomyocyte lineage produced from human pluripotent stem cells |
| WO2005001079A2 (en) * | 2003-06-27 | 2005-01-06 | Ethicon, Incorporated | Soft tissue repair and regeneration using postpartum-derived cells |
| WO2005035738A1 (en) * | 2003-10-07 | 2005-04-21 | Biomaster Inc. | Cell differentiation of adipose-derived precursor cells |
| EP1876233A1 (en) * | 2005-01-24 | 2008-01-09 | Japan Health Sciences Foundation | Cells capable of differentiating into cardiac muscle cells |
| CA2695374A1 (en) * | 2007-08-15 | 2009-02-19 | Amunix, Inc. | Compositions and methods for modifying properties of biologically active polypeptides |
| US9051550B2 (en) * | 2009-04-09 | 2015-06-09 | Arizona Board Of Regents, On Behalf Of The University Of Arizona | Cellular seeding and co-culture of a three dimensional fibroblast construct |
| US20120045487A1 (en) * | 2009-04-29 | 2012-02-23 | The Regents Of The University Of Michigan | Multiphasic microfibers for spatially guided cell growth |
| US8323972B2 (en) * | 2009-09-30 | 2012-12-04 | Advanced Technologies And Regenerative Medicine, Llc | Mammary artery derived cells and methods of use in tissue repair and regeneration |
| WO2012162741A1 (en) * | 2011-06-01 | 2012-12-06 | Monash University | Enrichment of cardiomyocytes |
| US10017739B2 (en) | 2012-09-06 | 2018-07-10 | Duke University | Methods of expanding and assessing B cells and using expanded B cells to treat disease |
-
2015
- 2015-01-05 CN CN202010584861.4A patent/CN111876371A/zh active Pending
- 2015-01-05 CN CN201580037730.1A patent/CN106661552B/zh active Active
- 2015-01-05 KR KR1020177003798A patent/KR102263824B1/ko active IP Right Grant
- 2015-01-05 WO PCT/JP2015/050028 patent/WO2016006262A1/ja active Application Filing
- 2015-01-05 AU AU2015286462A patent/AU2015286462B2/en active Active
- 2015-01-05 EP EP15819754.1A patent/EP3168297B1/en active Active
- 2015-01-05 CA CA2954743A patent/CA2954743C/en active Active
- 2015-07-10 JP JP2015138613A patent/JP6241893B2/ja active Active
-
2017
- 2017-01-09 US US15/401,832 patent/US20170112880A1/en not_active Abandoned
- 2017-10-30 JP JP2017208721A patent/JP2018029611A/ja active Pending
-
2018
- 2018-08-21 US US16/106,723 patent/US11801267B2/en active Active
Non-Patent Citations (4)
| Title |
|---|
| KATSUHISA MATSUURA ET AL.: "Tissue Engineering Chiryo 6 Kekkan Shinkin no Saisei Chiryo", BIOMEDICINE & THERAPEUTICS, vol. 43, no. 6, 2009, pages 646 - 650, XP008185244 * |
| MASUMOTO H. ET AL.: "Pluripotent stem cell - engineered cell sheets reassembled with defined cardiovascular populations ameliorate reduction in infarct heart function through cardiomyocyte-mediated neovascularization", STEM CELLS, vol. 30, no. 6, 2012, pages 1196 - 1205, XP055298190 * |
| MATSUURA K. ET AL.: "Creation of mouse embryonic stem cell -derived cardiac cell sheets", BIOMATERIALS, vol. 32, no. 30, 2011, pages 7355 - 7362, XP028261564, DOI: doi:10.1016/j.biomaterials.2011.05.042 * |
| UOSAKI H. ET AL.: "Efficient and scalable purification of cardiomyocytes from human embryonic and induced pluripotent stem cells by VCAM1 surface expression", PLOS ONE, vol. 6, no. 8, 2011, pages e23657, XP055124317, DOI: doi:10.1371/journal.pone.0023657 * |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109890398A (zh) * | 2017-02-24 | 2019-06-14 | 玛土撤拉有限公司 | 可用于治疗心脏疾病并含有成纤维细胞的注射用组合物以及用于生产用于治疗用途的成纤维细胞的方法 |
| KR20190117468A (ko) | 2017-02-24 | 2019-10-16 | 가부시키가이샤 메토세라 | 섬유아세포를 포함하고, 심장 질환들의 치료를 위하여 사용될 수 있는 주사용 조성물, 및 치료 용도를 위하여 섬유아세포를 생산하는 방법 |
| EP3476395A4 (en) * | 2017-02-24 | 2020-02-26 | Metcela Inc. | COMPOSITION FOR INJECTION THAT CAN BE USED FOR THE TREATMENT OF CARDIAC DISEASES AND CONTAINING FIBROBLASTS, AND METHOD FOR PRODUCING FIBROBLASTS FOR THERAPEUTIC USE |
| CN109890398B (zh) * | 2017-02-24 | 2024-01-16 | 玛土撤拉有限公司 | 可用于治疗心脏疾病并含有成纤维细胞的注射用组合物以及用于生产用于治疗用途的成纤维细胞的方法 |
| US20210346436A1 (en) * | 2017-02-24 | 2021-11-11 | Metcela Inc. | Composition for injection which can be used for treatment of heart diseases and contains fibroblasts, and method for producing fibroblast for therapy use |
| US11096969B2 (en) | 2017-02-24 | 2021-08-24 | Metcela, Inc. | Composition for injection which can be used for treatment of heart diseases and contains fibroblasts, and method for producing fibroblast for therapy use |
| KR20210049892A (ko) | 2018-08-29 | 2021-05-06 | 가부시키가이샤 메토세라 | 섬유아세포의 제조 방법 및 g-csf 양성 섬유아세포 집단 |
| CN112639082A (zh) * | 2018-08-29 | 2021-04-09 | 玛土撤拉有限公司 | 成纤维细胞的制备方法及g-csf阳性成纤维细胞群 |
| JPWO2020045547A1 (ja) * | 2018-08-29 | 2021-02-15 | 株式会社メトセラ | 線維芽細胞の製造方法及びg−csf陽性線維芽細胞集団 |
| WO2020045547A1 (ja) | 2018-08-29 | 2020-03-05 | 株式会社メトセラ | 線維芽細胞の製造方法及びg-csf陽性線維芽細胞集団 |
| JP7428389B2 (ja) | 2018-08-29 | 2024-02-06 | 株式会社メトセラ | 線維芽細胞の製造方法及びg-csf陽性線維芽細胞集団 |
| WO2021177387A1 (ja) | 2020-03-04 | 2021-09-10 | 株式会社メトセラ | エリスロポエチン産生能が亢進された線維芽細胞 |
| JPWO2021177387A1 (ja) * | 2020-03-04 | 2021-09-10 | ||
| KR20220148184A (ko) | 2020-03-04 | 2022-11-04 | 가부시키가이샤 메토세라 | 에리트로포이에틴 산생능이 항진된 섬유 아세포 |
| JP7229618B2 (ja) | 2020-03-04 | 2023-02-28 | 株式会社メトセラ | エリスロポエチン産生能が亢進された線維芽細胞 |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2954743A1 (en) | 2016-01-14 |
| CA2954743C (en) | 2022-03-15 |
| US11801267B2 (en) | 2023-10-31 |
| US20170112880A1 (en) | 2017-04-27 |
| EP3168297B1 (en) | 2020-09-16 |
| CN106661552B (zh) | 2020-07-07 |
| JP2016027797A (ja) | 2016-02-25 |
| EP3168297A1 (en) | 2017-05-17 |
| KR102263824B1 (ko) | 2021-06-10 |
| US20180369288A1 (en) | 2018-12-27 |
| JP2018029611A (ja) | 2018-03-01 |
| KR20170023188A (ko) | 2017-03-02 |
| EP3168297A4 (en) | 2018-03-07 |
| AU2015286462A1 (en) | 2017-03-02 |
| JP6241893B2 (ja) | 2017-12-06 |
| CN111876371A (zh) | 2020-11-03 |
| CN106661552A (zh) | 2017-05-10 |
| AU2015286462B2 (en) | 2021-04-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6241893B2 (ja) | 心臓細胞培養材料 | |
| US7727762B2 (en) | Method of inducing the differentiation of stem cells into myocardial cells | |
| Benton et al. | Advancing science and technology via 3D culture on basement membrane matrix | |
| Royce et al. | Human neoplastic submandibular intercalated duct cells express an acinar phenotype when cultured on a basement membrane matrix | |
| KR20100040276A (ko) | 심근 세포의 세포괴의 제조 방법 및 상기 심근 세포괴의 용도 | |
| CA2882802C (en) | Method for producing retinal pigment epithelial cell sheet | |
| Hausman et al. | Stromal vascular cells and adipogenesis: cells within adipose depots regulate adipogenesis | |
| KR20070001101A (ko) | 포유동물의 골수 세포 또는 제대혈 유래 세포와 지방조직을 이용한 심근 세포의 유도 | |
| Iwamiya et al. | Cardiac fibroblast-derived VCAM-1 enhances cardiomyocyte proliferation for fabrication of bioengineered cardiac tissue | |
| KR20190112090A (ko) | 다능성줄기세포의 분화 제어 방법 | |
| Shimada et al. | Efficient expansion of mouse primary tenocytes using a novel collagen gel culture method | |
| JP2012125207A (ja) | 角膜実質細胞培養用スキャフォールド | |
| Kihara et al. | Laminin-221-derived recombinant fragment facilitates isolation of cultured skeletal myoblasts | |
| JP6587877B2 (ja) | 表皮幹細胞を分離するためのマーカー及び三次元培養表皮の製造方法 | |
| Iwamiya et al. | Regenerative Therapy | |
| Girardi | TGFbeta signalling pathway in muscle regeneration: an important regulator of muscle cell fusion | |
| Koskimäki | Maturation and morphology of human pluripotent stem cell derived cardiomyocytes and vascular structures in 3D cardiovascular construct: Towards modelling myocardial ischemia | |
| Elhendawi et al. | Kidney organoids | |
| US20200095557A1 (en) | Cell spheroids containing capillary structures and methods of using same | |
| Li | Cardiac Fibroblasts in Heart Function, Disease, and Therapy: Insights from 3D | |
| Zhao | Establishment of in vitro Three-Dimensional Human Brown Adipose Tissue-like Constructs | |
| Dawson | Cardiac Tissue Engineering |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15819754 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2954743 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015819754 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015819754 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20177003798 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2015286462 Country of ref document: AU Date of ref document: 20150105 Kind code of ref document: A |