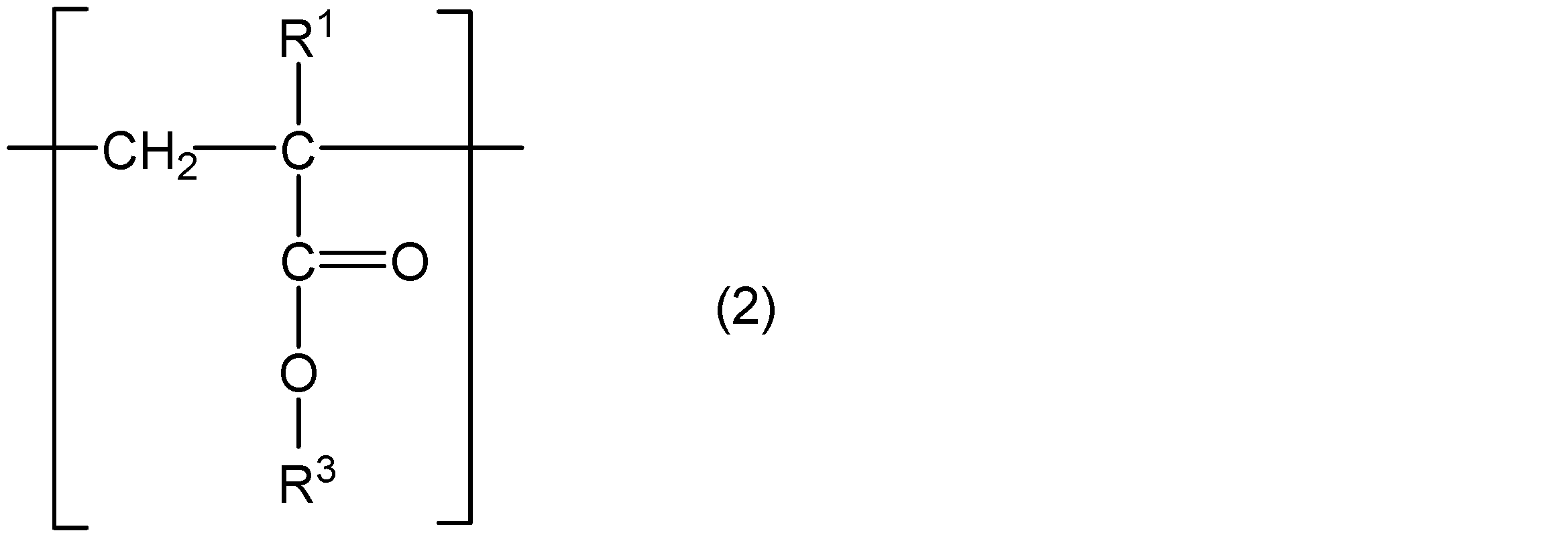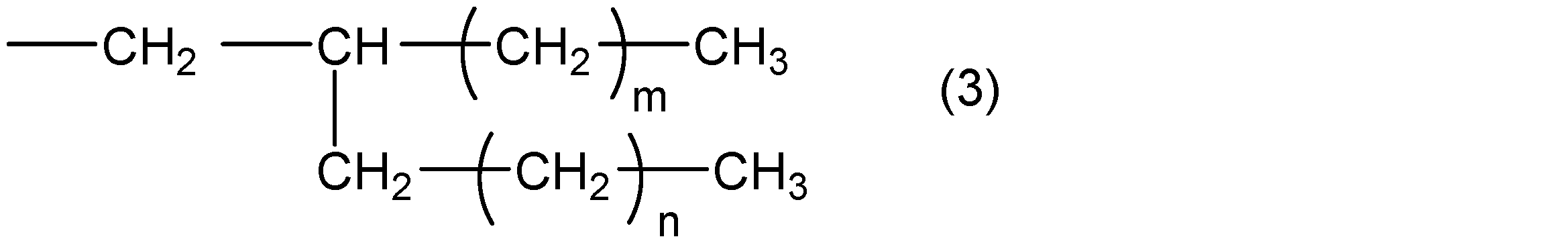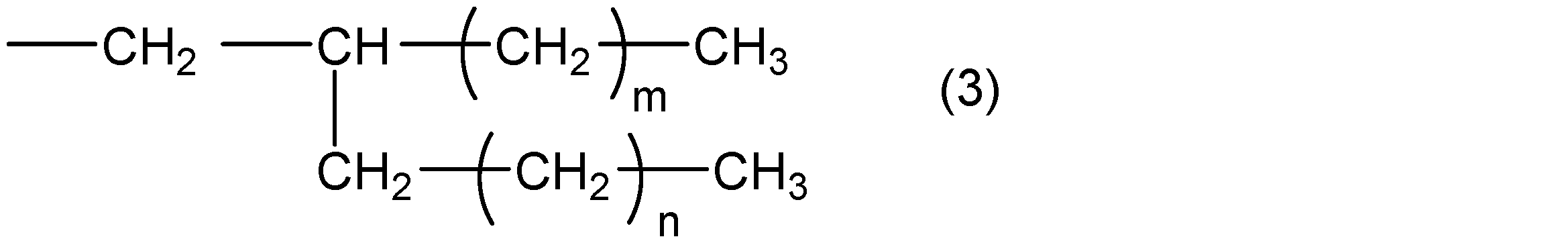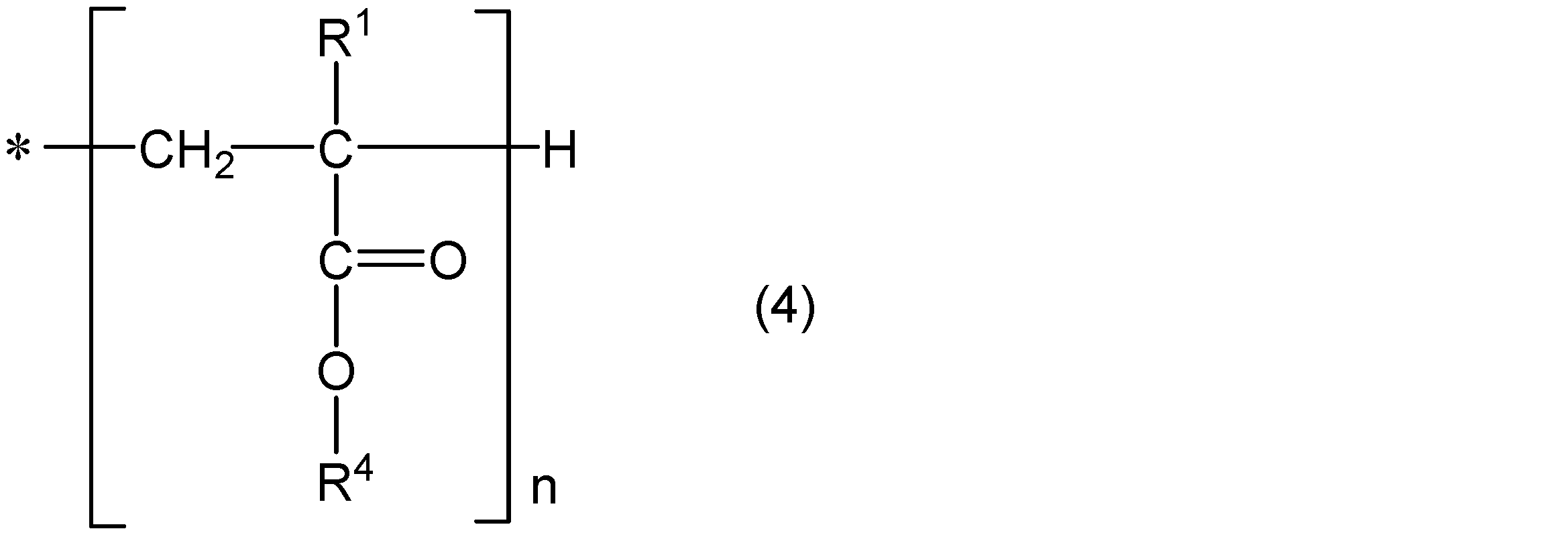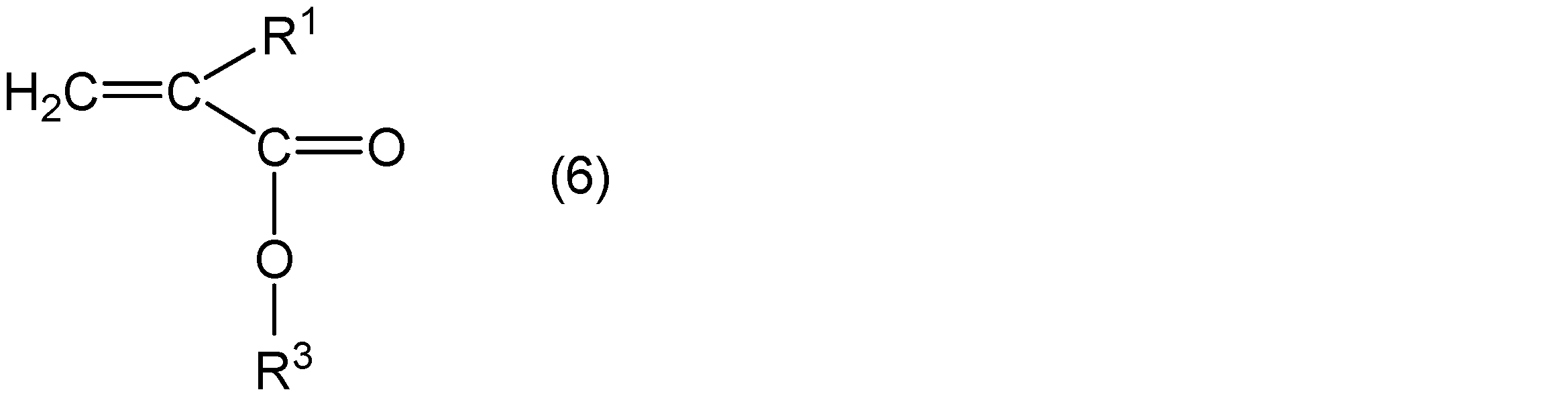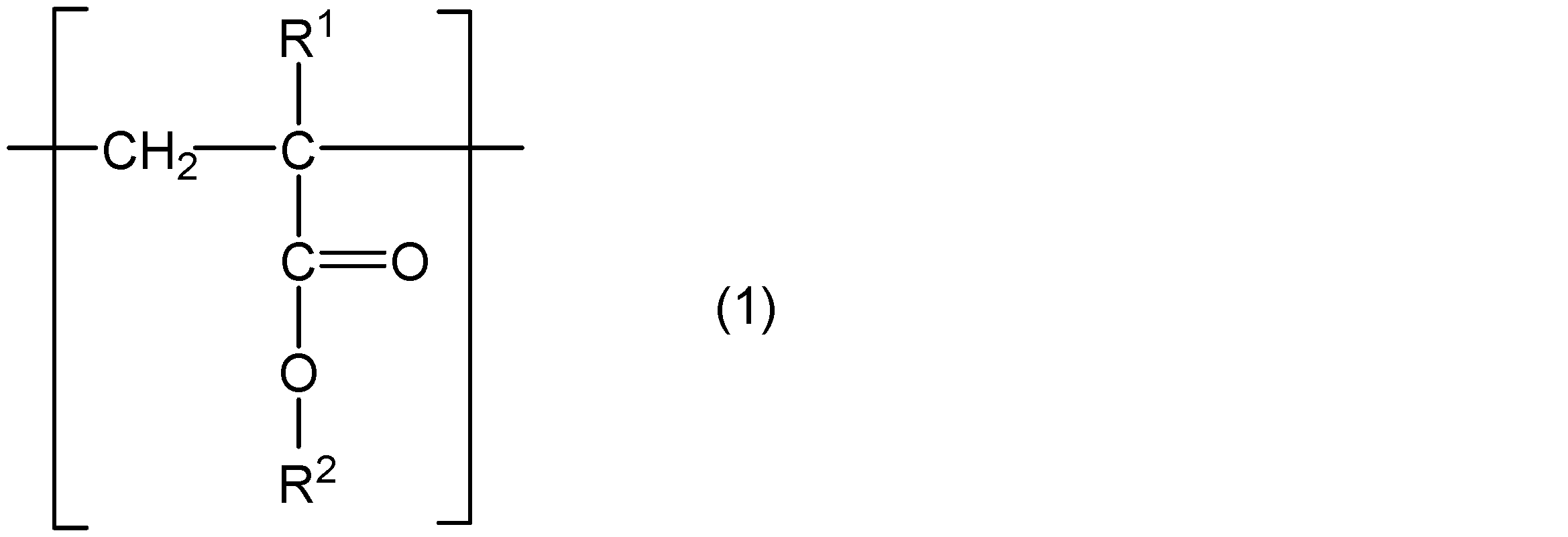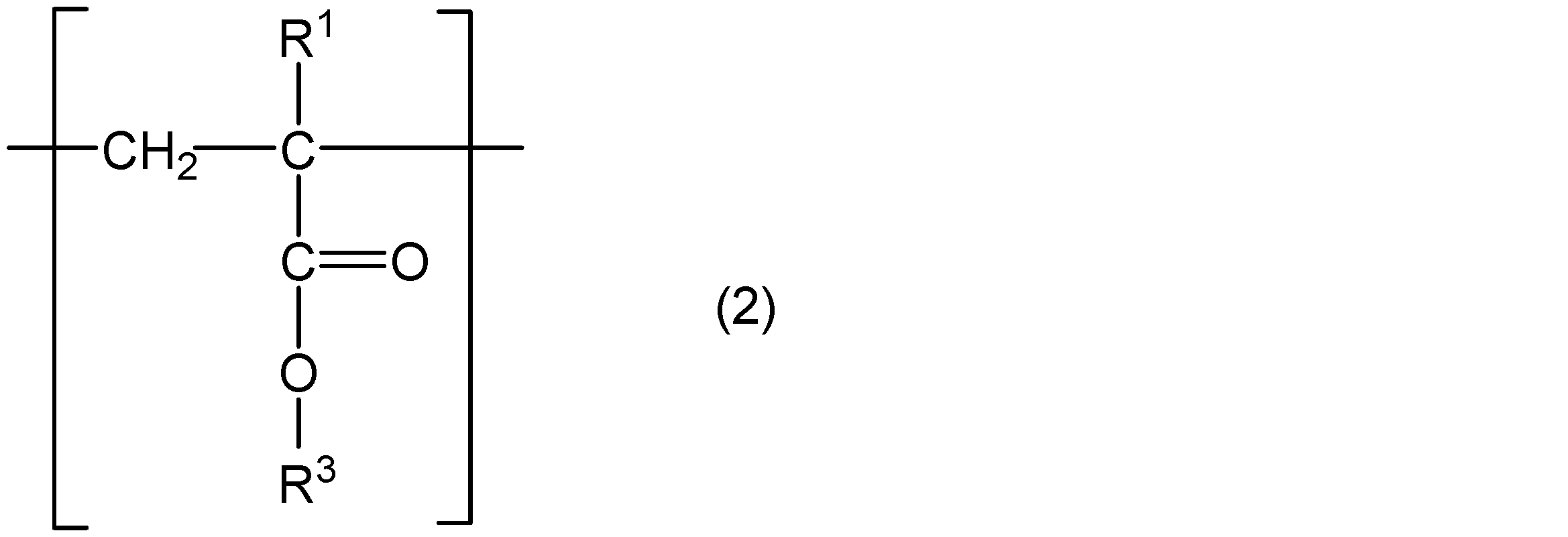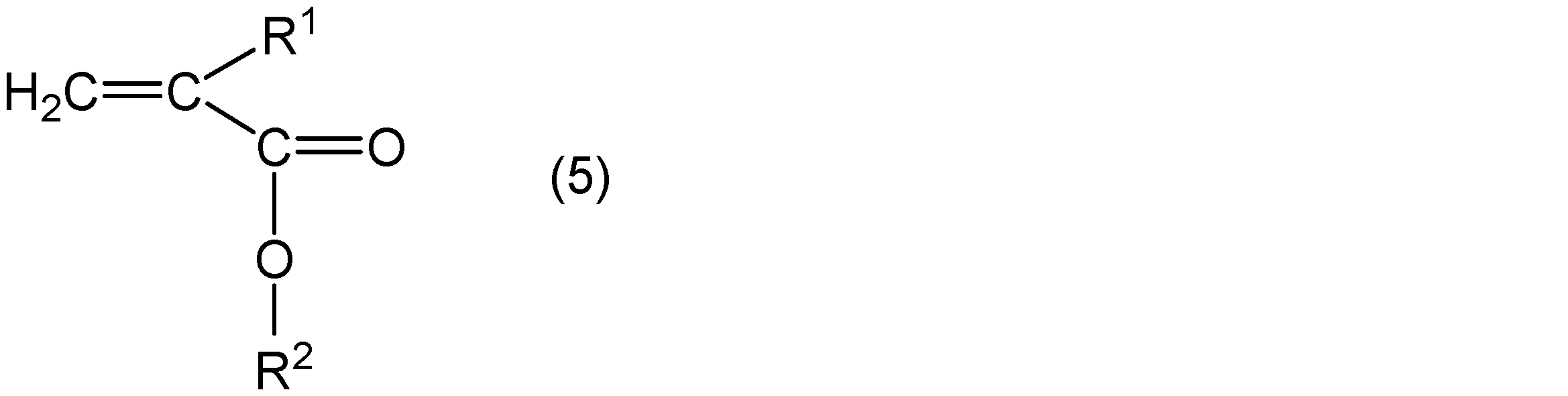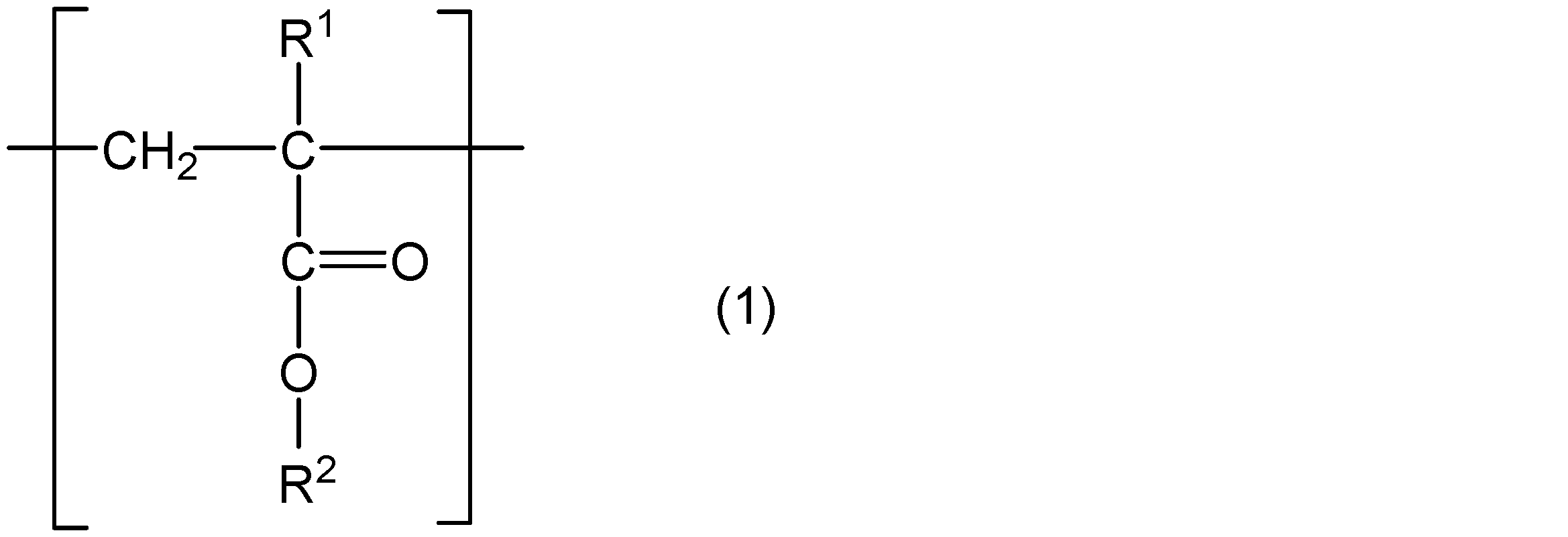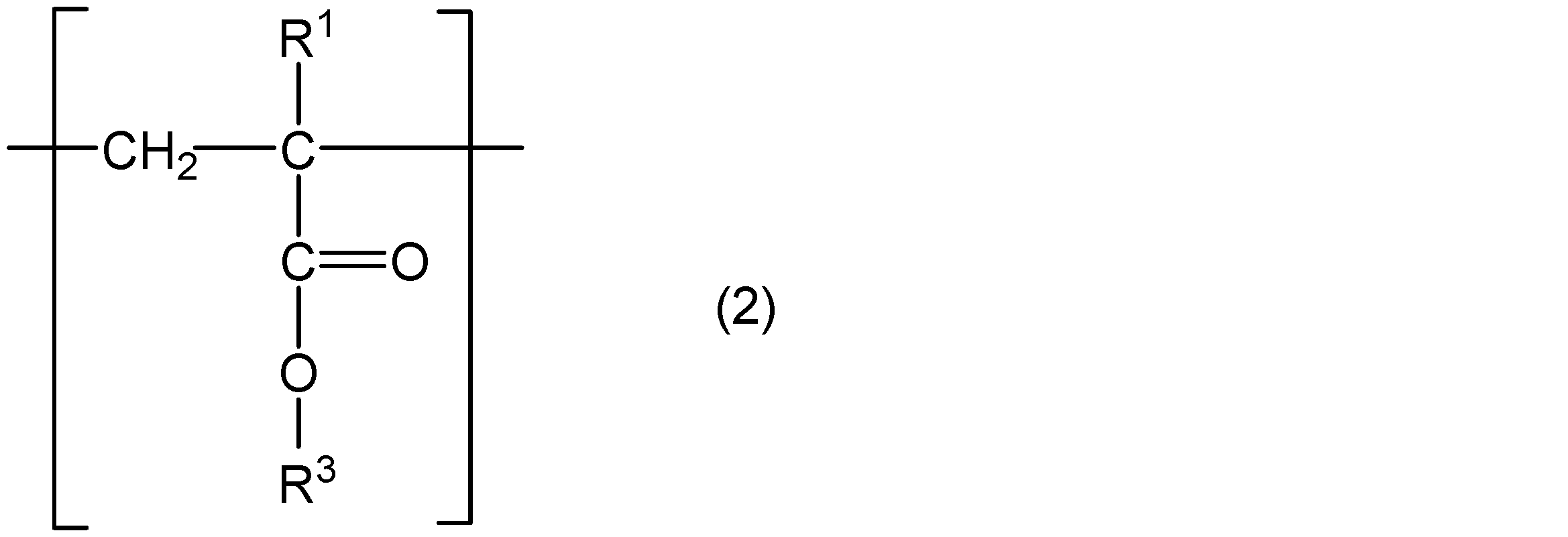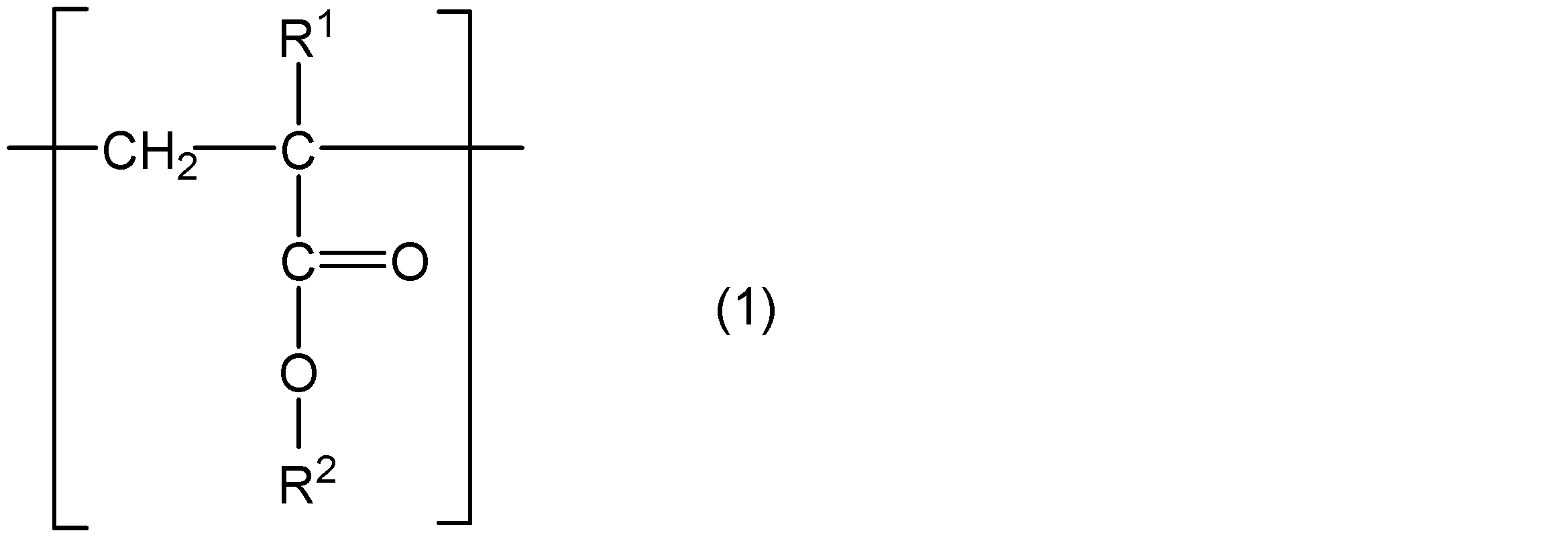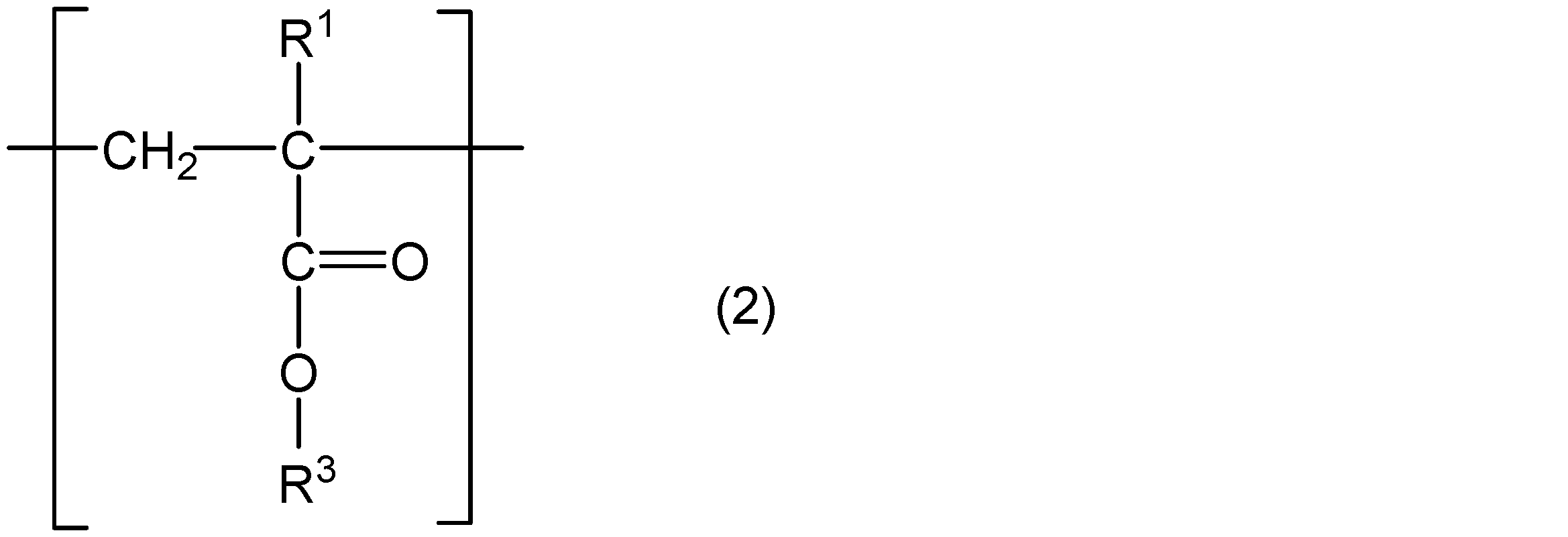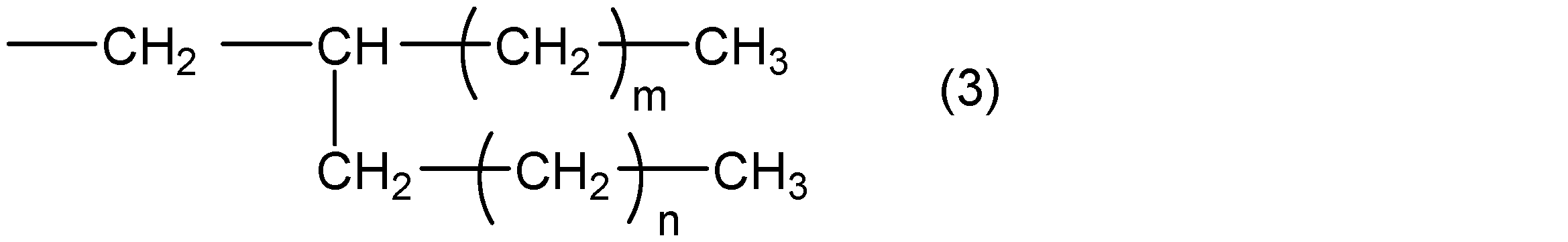WO2014017553A1 - ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 - Google Patents
ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 Download PDFInfo
- Publication number
- WO2014017553A1 WO2014017553A1 PCT/JP2013/070085 JP2013070085W WO2014017553A1 WO 2014017553 A1 WO2014017553 A1 WO 2014017553A1 JP 2013070085 W JP2013070085 W JP 2013070085W WO 2014017553 A1 WO2014017553 A1 WO 2014017553A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- viscosity index
- meth
- acrylate
- index improver
- poly
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M145/00—Lubricating compositions characterised by the additive being a macromolecular compound containing oxygen
- C10M145/02—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- C10M145/10—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds containing monomers having an unsaturated radical bound to a carboxyl radical, e.g. acrylate
- C10M145/12—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds containing monomers having an unsaturated radical bound to a carboxyl radical, e.g. acrylate monocarboxylic
- C10M145/14—Acrylate; Methacrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/12—Esters of monohydric alcohols or phenols
- C08F220/16—Esters of monohydric alcohols or phenols of phenols or of alcohols containing two or more carbon atoms
- C08F220/18—Esters of monohydric alcohols or phenols of phenols or of alcohols containing two or more carbon atoms with acrylic or methacrylic acids
- C08F220/1818—C13or longer chain (meth)acrylate, e.g. stearyl (meth)acrylate
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M101/00—Lubricating compositions characterised by the base-material being a mineral or fatty oil
- C10M101/02—Petroleum fractions
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M135/00—Lubricating compositions characterised by the additive being an organic non-macromolecular compound containing sulfur, selenium or tellurium
- C10M135/12—Thio-acids; Thiocyanates; Derivatives thereof
- C10M135/14—Thio-acids; Thiocyanates; Derivatives thereof having a carbon-to-sulfur double bond
- C10M135/18—Thio-acids; Thiocyanates; Derivatives thereof having a carbon-to-sulfur double bond thiocarbamic type, e.g. containing the groups
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M169/00—Lubricating compositions characterised by containing as components a mixture of at least two types of ingredient selected from base-materials, thickeners or additives, covered by the preceding groups, each of these compounds being essential
- C10M169/04—Mixtures of base-materials and additives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2203/00—Organic non-macromolecular hydrocarbon compounds and hydrocarbon fractions as ingredients in lubricant compositions
- C10M2203/10—Petroleum or coal fractions, e.g. tars, solvents, bitumen
- C10M2203/102—Aliphatic fractions
- C10M2203/1025—Aliphatic fractions used as base material
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2209/00—Organic macromolecular compounds containing oxygen as ingredients in lubricant compositions
- C10M2209/02—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- C10M2209/08—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds containing monomers having an unsaturated radical bound to a carboxyl radical, e.g. acrylate type
- C10M2209/084—Acrylate; Methacrylate
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant compositions
- C10M2215/02—Amines, e.g. polyalkylene polyamines; Quaternary amines
- C10M2215/06—Amines, e.g. polyalkylene polyamines; Quaternary amines having amino groups bound to carbon atoms of six-membered aromatic rings
- C10M2215/064—Di- and triaryl amines
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant compositions
- C10M2215/08—Amides
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2215/00—Organic non-macromolecular compounds containing nitrogen as ingredients in lubricant compositions
- C10M2215/28—Amides; Imides
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2219/00—Organic non-macromolecular compounds containing sulfur, selenium or tellurium as ingredients in lubricant compositions
- C10M2219/02—Sulfur-containing compounds obtained by sulfurisation with sulfur or sulfur-containing compounds
- C10M2219/024—Sulfur-containing compounds obtained by sulfurisation with sulfur or sulfur-containing compounds of esters, e.g. fats
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2219/00—Organic non-macromolecular compounds containing sulfur, selenium or tellurium as ingredients in lubricant compositions
- C10M2219/04—Organic non-macromolecular compounds containing sulfur, selenium or tellurium as ingredients in lubricant compositions containing sulfur-to-oxygen bonds, i.e. sulfones, sulfoxides
- C10M2219/046—Overbasedsulfonic acid salts
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2219/00—Organic non-macromolecular compounds containing sulfur, selenium or tellurium as ingredients in lubricant compositions
- C10M2219/10—Heterocyclic compounds containing sulfur, selenium or tellurium compounds in the ring
- C10M2219/104—Heterocyclic compounds containing sulfur, selenium or tellurium compounds in the ring containing sulfur and carbon with nitrogen or oxygen in the ring
- C10M2219/106—Thiadiazoles
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2223/00—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions
- C10M2223/02—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions having no phosphorus-to-carbon bonds
- C10M2223/04—Phosphate esters
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2010/00—Metal present as such or in compounds
- C10N2010/04—Groups 2 or 12
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2020/00—Specified physical or chemical properties or characteristics, i.e. function, of component of lubricating compositions
- C10N2020/01—Physico-chemical properties
- C10N2020/04—Molecular weight; Molecular weight distribution
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/02—Pour-point; Viscosity index
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/06—Oiliness; Film-strength; Anti-wear; Resistance to extreme pressure
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/08—Resistance to extreme temperature
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/54—Fuel economy
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/68—Shear stability
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/04—Oil-bath; Gear-boxes; Automatic transmissions; Traction drives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/25—Internal-combustion engines
Definitions
- the present invention relates to a poly (meth) acrylate viscosity index improver, and a lubricant additive and a lubricant composition containing the viscosity index improver.
- a lubricating oil used for an internal combustion engine such as an automobile engine
- lubricating oil for an internal combustion engine also referred to as “lubricating oil for an internal combustion engine” or “engine oil”
- a lubricating base oil is used as one means for improving fuel efficiency.
- a method for increasing the viscosity index of a lubricating oil by adding a viscosity index improver is known.
- lubricating oil such as ATF, MTF, CVTF, etc.
- driving system oil a lubricating oil
- One method is to reduce the viscosity resistance by reducing the viscosity of the transmission lubricant.
- the viscosity of the transmission lubricating oil is lowered, other problems such as oil leakage and seizure may occur.
- a method using a viscosity index improver there is a method using a viscosity index improver.
- the viscosity index of the transmission lubricant is increased by using a viscosity index improver, and the increase in the viscosity in the low temperature region is suppressed while the viscosity in the high temperature region is maintained.
- viscosity index improver various types have been proposed so far, and in particular, the use of poly (meth) acrylate viscosity index improvers has been proposed (for example, Patent Documents 1 to 7). reference).
- viscosity index improver greatly influences the fuel efficiency, it is desirable that the viscosity index improver is excellent in shear stability.
- conventional poly (meth) acrylate-based viscosity index improvers do not necessarily satisfy all of fuel economy, low-temperature fluidity and shear stability.
- an object of the present invention is to provide a viscosity index improver capable of achieving both fuel saving and low temperature fluidity, and a lubricating oil additive and a lubricating oil composition containing the viscosity index improver. It is in.
- Another object of the present invention is to sufficiently reduce the high shear viscosity at 100 ° C. while maintaining a high shear viscosity at 150 ° C., and to sufficiently ensure low temperature fluidity, and
- An object of the present invention is to provide a viscosity index improver exhibiting high shear stability, and a lubricant additive and a lubricant composition containing the viscosity index improver.
- Another object of the present invention is to provide a lubricating oil with a sufficient friction loss reducing effect and to ensure low-temperature fluidity, and to improve the viscosity index, and to improve the viscosity index. It is providing the lubricating oil additive and lubricating oil composition containing an agent.
- the inventors of the present invention have a specific structure, and poly (meth) acrylate having a weight average molecular weight Mw and a ratio Mw / Mn between the weight average molecular weight Mw and the number average molecular weight Mn satisfying a specific condition.
- the system viscosity index improver can sufficiently reduce the high shear viscosity at 100 ° C. while maintaining the high shear viscosity at 150 ° C., and can secure low-temperature fluidity, and has high shear stability. As a result, the present invention has been completed.
- the present invention comprises a core part, a polymer chain containing a structural unit represented by the following general formula (1) and a structural unit represented by the following general formula (2), and one end of the polymer chain is a core part.
- the weight average molecular weight Mw is 100,000 or more
- the ratio Mw / Mn of the weight average molecular weight Mw to the number average molecular weight Mn is 1.6 or less.
- a poly (meth) acrylate viscosity index improver hereinafter referred to as “first poly (meth) acrylate viscosity index improver” is provided.
- R 1 represents hydrogen or a methyl group
- R 2 represents a group represented by the following general formula (3)
- R 3 represents a straight chain or a branched chain having 5 or less carbon atoms.
- m and n are integers that satisfy m ⁇ 5, n ⁇ 4, and m + n ⁇ 31.
- the inventors of the present invention have poly (meta) having a specific structure and a weight average molecular weight and a ratio Mw / Mn between the weight average molecular weight Mw and the number average molecular weight Mn satisfying a specific condition. It has been found that an acrylate-based viscosity index improver can impart a friction loss reducing effect and can secure low-temperature fluidity and is excellent in shear stability, leading to the completion of the present invention.
- the present invention comprises a core part, a polymer chain containing a structural unit represented by the following general formula (1) and a structural unit represented by the following general formula (2), and one end of the polymer chain is a core part.
- the weight average molecular weight Mw is less than 100,000
- the ratio Mw / Mn of the weight average molecular weight Mw to the number average molecular weight Mn is 1.6 or less.
- a poly (meth) acrylate viscosity index improver hereinafter referred to as “second poly (meth) acrylate viscosity index improver” is provided.
- R 1 represents hydrogen or a methyl group
- R 2 represents a group represented by the following general formula (3)
- R 3 represents a straight chain or a branched chain having 5 or less carbon atoms.
- m and n are integers that satisfy m ⁇ 5, n ⁇ 4, and m + n ⁇ 31.
- the present invention also provides a lubricating oil additive containing at least one selected from the first poly (meth) acrylate viscosity index improver and the second poly (meth) acrylate viscosity index improver. .
- the present invention also includes a lubricating base oil and at least one selected from the first poly (meth) acrylate viscosity index improver and the second poly (meth) acrylate viscosity index improver.
- a lubricating oil composition is provided.
- a viscosity index improver capable of achieving both fuel economy and low temperature fluidity, and a lubricant additive and a lubricant composition containing the viscosity index improver.
- the high shear viscosity at 100 ° C. can be sufficiently reduced, and low-temperature fluidity can be sufficiently ensured, and high shear can be achieved.
- Viscosity index improvers exhibiting stability, and lubricating oil additives and lubricating oil compositions containing the viscosity index improvers can be provided.
- a viscosity index improver that can impart sufficient friction loss reduction effect to the lubricating oil and can secure low-temperature fluidity and is excellent in shear stability, and the viscosity index improver.
- Lubricating oil additives and lubricating oil compositions can be provided.
- the poly (meth) acrylate viscosity index improver is a polymerization including a core, a structural unit represented by the following general formula (1), and a structural unit represented by the following general formula (2). And three or more arm portions made of chains.
- the poly (meth) acrylate viscosity index improver has a weight average molecular weight Mw (hereinafter, simply referred to as “Mw” in some cases) of 100,000 or more, and a weight average molecular weight Mw and a number average molecular weight Mn (hereinafter, depending on circumstances).
- Mw / Mn The ratio Mw / Mn (hereinafter simply referred to as “Mw / Mn”) is 1.6 or less.
- R 1 represents hydrogen or a methyl group
- R 2 represents a group represented by the following general formula (3)
- R 3 represents a straight chain or a branched chain having 5 or less carbon atoms.
- m and n are integers that satisfy m ⁇ 5, n ⁇ 4, and m + n ⁇ 31.
- R 1 may be either hydrogen or a methyl group, but is preferably a methyl group.
- R 2 m is preferably 5 to 16, and n is 4 to 15, more preferably m is 6 to 15, and n is 6 to 10, and m is 7 from the viewpoint of viscosity reduction. More preferably, those having ⁇ 10 and n is 6 ⁇ 9.
- R 1 and R 2 may be the same or different among the structural units.
- the polymer chain constituting the arm portion includes the structural unit represented by the general formula (1) and the structural unit represented by the general formula (2).
- the structural unit represented by the general formula (1) is preferably contained in an amount of 20 to 80% by mass, more preferably 20 to 70% by mass, based on the total amount of the structural units contained in the polymer chain. More preferably, it is contained by mass%.
- the polymer chain preferably contains 20-80% by mass of the structural unit represented by the general formula (2) based on the total amount of the structural unit contained in the polymer chain, from the viewpoint of fuel economy. More preferably, it is contained in an amount of 30 to 80% by mass, and further preferably 50 to 80% by mass.
- the polymer chain is a combination of the structural unit represented by the general formula (1) and the structural unit represented by the general formula (2), and is based on the total amount of the structural units contained in the polymer chain. It is preferable to contain at least 80% by mass, more preferably at least 80% by mass, still more preferably at least 90% by mass, and most preferably at least 100% by mass.
- R 1 and R 3 may be the same or different among the structural units.
- the structural unit in which R 3 is a methyl group is based on the total amount of structural units contained in the polymer chain.
- the content is preferably 5 to 50% by mass, more preferably 10 to 45% by mass, and still more preferably 20 to 45% by mass.
- the structural unit in which R 3 is an alkyl group having 18 carbon atoms is preferably contained in an amount of 5 to 50% by mass based on the total amount of structural units contained in the polymer chain. More preferably, it is contained in an amount of 45% by mass, and more preferably 20-40% by mass.
- the polymer chain may contain only the structural unit represented by the general formula (1) and the structural unit represented by the general formula (2), or may further contain other structural units. Good. Moreover, one end is couple
- R 1 represents hydrogen or a methyl group
- R 4 represents a group represented by the above general formula (3), or a straight chain or having 1 to 18 carbon atoms having a branch having 5 or less carbon atoms.
- An alkyl group is shown, and n is an integer selected so that Mw and Mw / Mn satisfy the above conditions.
- n is an integer of 400 to 2000, for example.
- the weight average molecular weight Mw per arm part is appropriately selected so that the Mw of the poly (meth) acrylate viscosity index improver satisfies the above conditions, but is preferably 10,000 or more, and 15,000. More preferably, it is more preferably 18,000 or more.
- the number average molecular weight Mn per arm is appropriately selected so that the Mw / Mn of the poly (meth) acrylate viscosity index improver satisfies the above-mentioned conditions, but is preferably 8,000 or more. Is more preferably 15,000 or more, and further preferably 15,000 or more.
- the weight average molecular weight Mw of the poly (meth) acrylate viscosity index improver is 100,000 or more, preferably 125,000 or more, more preferably 150,000 or more from the viewpoint of fuel saving performance. Preferably, it is more preferably 175,000 or more.
- the upper limit of Mw is not particularly limited, but Mw is, for example, 500,000 or less.
- the number average molecular weight Mn of the poly (meth) acrylate viscosity index improver is appropriately selected so that Mw / Mn satisfies the above conditions.
- Mn is preferably 75,000 or more, more preferably 94,000 or more, and further preferably 110,000 or more.
- the upper limit of Mn is not particularly limited, Mn is, for example, 300,000 or less.
- Mw / Mn of the poly (meth) acrylate viscosity index improver is 1.6 or less, but from the viewpoint of fuel saving characteristics, it is preferably 1.5 or less, more preferably 1.4 or less. Preferably, it is 1.2 or less. Further, Mw / Mn is preferably 1.00 or more, more preferably 1.01 or more, and further preferably 1.02 or more, from the viewpoint of fuel saving characteristics.
- weight average molecular weight Mw “weight average molecular weight Mw”, “number average molecular weight Mn” and “ratio Mw / Mn of weight average molecular weight Mw and number average molecular weight Mn” are Mw, Mn and Mw obtained by GPC analysis. / Mn (polystyrene (standard sample) conversion value). Mw / Mn of the poly (meth) acrylate viscosity index improver and Mw and Mn per arm part can be measured, for example, as follows.
- the sample solution is analyzed using a GPC apparatus (Waters Alliance 2695).
- the analysis is carried out using a column having a solvent flow rate of 1 ml / min, an analyzable molecular weight of 10,000 to 256,000, and a refractive index as a detector.
- the relationship between the column retention time and the molecular weight is determined using a polystyrene standard with a clear molecular weight, a calibration curve is separately prepared, and the molecular weight is determined from the obtained retention time.
- the molecular weight of the arm (Mw and Mn) can be calculated by dividing the obtained molecular weight (Mw and Mn) by the number of functional groups of the initiator.
- the core part is derived from a compound having three or more functional groups that react with the carbon-carbon double bond of the acryloyl group.
- the compound having three or more functional groups that react with the carbon-carbon double bond of the acryloyl group include 1,1,1-tris (2-bromoisobutyloxymethylene) ethane, pentaerythritol tetrakis (2-bromoiso). Butyrate) and dipentaerythritol hexakis (2-bromoisobutyrate).
- the number of arm portions of the poly (meth) acrylate viscosity index improver corresponds to the number of the functional groups.
- the number of arm parts that is, the number of functional groups is preferably 2 to 12, more preferably 2 to 8, and still more preferably 3 to 6.
- a polymerization catalyst is added to the mixed solution containing an alkyl (meth) acrylate, an initiator, and a solvent, and alkyl ( A method of polymerizing (meth) acrylate is mentioned.
- alkyl (meth) acrylate an alkyl (meth) acrylate represented by the following general formula (5) and an alkyl (meth) acrylate represented by the following general formula (6) can be used.
- R 1 represents hydrogen or a methyl group
- R 2 represents a group represented by the general formula (3)
- R 3 represents a straight chain or a branch having 5 or less carbon atoms.
- R 1 is preferably a methyl group.
- R 2 is preferably m of 5 to 16 and n of 4 to 15, more preferably m of 6 to 15, and n of 6 to 10, m of 7 to 10, and n of 6 to 10. What is 9 is still more preferable.
- the alkyl (meth) acrylate represented by the general formula (5) and the alkyl (meth) acrylate represented by the general formula (6) can be used.
- the content of the alkyl (meth) acrylate represented by the general formula (5) is preferably 20 to 80% by mass and more preferably 20 to 70% by mass based on the total amount of the alkyl (meth) acrylate.
- the content is preferably 20 to 50% by mass.
- the content of the alkyl (meth) acrylate represented by the general formula (5) is preferably 20 to 80% by mass, and 30 to 80% by mass based on the total amount of the alkyl (meth) acrylate. Is more preferable, and 50 to 80% by mass is even more preferable.
- the alkyl (meth) acrylate represented by the general formula (6) one kind of the alkyl (meth) acrylate represented by the general formula (6) may be used alone, or two or more kinds may be mixed and used. However, it is preferable to use a mixture of two or more.
- the content of methyl (meth) acrylate in which R 2 is a methyl group is preferably 5 to 50% by mass based on the total amount of alkyl (meth) acrylate. More preferably, it is more preferably 20% to 45% by weight.
- the content of alkyl (meth) acrylate in which R 2 is an alkyl group having 18 carbon atoms is preferably 5 to 50% by mass, based on the total amount of alkyl (meth) acrylate, and is 10 to 45% by mass. More preferred is 20 to 40% by mass.
- the initiator one derived from a compound having three or more functional groups that react with the carbon-carbon double bond of the acryloyl group can be used.
- 1,1,1-tris (2-bromoisobutyloxy Methylene) ethane, pentaerythritol tetrakis (2-bromoisobutyrate), dipentaerythritol hexakis (2-bromoisobutyrate) can be used.
- solvent for example, highly refined mineral oil, anisole, and toluene can be used.
- highly refined mineral oil can be exemplified.
- polymerization catalyst examples include copper (II) bromide, tris (2-pyridylmethyl) amine, azobisisobutyronitrile, tin (II) 2-ethylhexanoate, tris [2- (dimethylamino) ethyl]. Amines can be used.
- Preferred examples of the polymerization catalyst include copper (II) bromide, tris (2-pyridylmethyl) amine, azobisisobutyronitrile, and tin (II) 2-ethylhexanoate. You may use these polymerization catalysts individually by 1 type or in mixture of 2 or more types.
- the reaction temperature for polymerizing the alkyl (meth) acrylate can be appropriately selected.
- a preferable reaction temperature is 60 to 100 ° C.
- the Mw / Mn of the resulting poly (meth) acrylate viscosity index improver tends to be 1.6 or less.
- Mw / Mn tends to be 1.0 to 1.2
- Mw / Mn is 1.2 to 1.4.
- Mw / Mn tends to be 1.4 to 1.6.
- the reaction time depends on the reaction conditions such as the raw material alkyl (meth) acrylate, polymerization reagent, solvent and initiator, reaction temperature, and the like, and the desired poly (meth) acrylate Mw and Mw / Mn. It can be selected as appropriate.
- a preferable reaction time is 8 to 16 hours.
- the polymerization of the alkyl (meth) acrylate is preferably performed in a nitrogen atmosphere.
- the lubricating oil additive according to the second embodiment of the present invention comprises a core, a structural unit represented by the general formula (1), and a polymer chain including the structural unit represented by the general formula (2). And an arm part in which one end of the polymer chain is bonded to the core part, the weight average molecular weight Mw is 100,000 or more, and the ratio Mw / Mn between the weight average molecular weight Mw and the number average molecular weight Mn is A poly (meth) acrylate viscosity index improver that is 1.6 or less is contained.
- the poly (meth) acrylate-based viscosity index improver in the present embodiment is the same as the viscosity index improver in the first embodiment, and a duplicate description is omitted here.
- the lubricating oil additive may consist only of the above poly (meth) acrylate viscosity index improver, or a mixture of the viscosity index improver and other additives (ie, additive composition). It may be.
- the lubricating oil additive is a mixture of the viscosity index improver and other additives, the mixing ratio is not particularly limited and can be appropriately selected according to the application.
- additives include viscosity index improvers other than the above poly (meth) acrylate viscosity index improvers, antioxidants, antiwear agents (or extreme pressure agents), corrosion inhibitors, rust inhibitors, viscosity indexes Examples thereof include additives such as improvers, pour point depressants, demulsifiers, metal deactivators, antifoaming agents, and ashless friction modifiers. These additives can be used individually by 1 type or in combination of 2 or more types.
- poly (meth) acrylate viscosity index improver other than the above poly (meth) acrylate viscosity index improver
- polyisobutene viscosity index improver examples thereof include an improver, an ethylene-propylene copolymer viscosity index improver, and a styrene-butadiene hydrogenated copolymer viscosity index improver.
- antioxidants examples include ashless antioxidants such as phenols and amines, and metal antioxidants such as zinc, copper, and molybdenum.
- phenolic antioxidants examples include 4,4′-methylenebis (2,6-di-tert-butylphenol), 4,4′-bis (2,6-di-tert-butylphenol), 4,4 ′.
- amine antioxidants include known amine antioxidants that are generally used for lubricating oils such as aromatic amine compounds, alkyldiphenylamines, alkylnaphthylamines, phenyl- ⁇ -naphthylamines, alkylphenyl- ⁇ -naphthylamines, and the like. Agents.
- corrosion inhibitor examples include benzotriazole, tolyltriazole, thiadiazole, or imidazole compounds.
- rust preventive examples include petroleum sulfonate, alkylbenzene sulfonate, dinonylnaphthalene sulfonate, alkenyl succinic acid ester, and polyhydric alcohol ester.
- metal deactivator examples include imidazoline, pyrimidine derivatives, alkylthiadiazole, mercaptobenzothiazole, benzotriazole or derivatives thereof, 1,3,4-thiadiazole polysulfide, 1,3,4-thiadiazolyl-2,5-bis.
- metal deactivator examples include dialkyldithiocarbamate, 2- (alkyldithio) benzimidazole, and ⁇ - (o-carboxybenzylthio) propiononitrile.
- antifoaming agents examples include silicone oils having a kinematic viscosity at 25 ° C. of 1,000 to 100,000 mm 2 / s, alkenyl succinic acid derivatives, esters of polyhydroxy aliphatic alcohols and long-chain fatty acids, and methyl salicylates. o-hydroxybenzyl alcohol and the like.
- any compound usually used as an ashless friction modifier for lubricating oils can be used.
- ashless friction modifiers such as amine compounds, fatty acid esters, fatty acid amides, fatty acids, aliphatic alcohols and aliphatic ethers having at least one linear alkyl group or linear alkenyl group in the molecule.
- Various ashless friction modifiers exemplified in International Publication No. 2005/037967 such as nitrogen-containing compounds and acid-modified derivatives thereof described in JP-A-2009-286831, can also be used.
- the lubricating oil additive according to this embodiment may further contain a solvent.
- a solvent highly refined mineral oil, solvent refined base oil, and synthetic oil can be used. Among these, it is preferable to use highly refined mineral oil.
- the lubricating oil additive contains a solvent, the content of the solvent is preferably 5 to 75% by mass, more preferably 30 to 60% by mass, based on the total amount of the lubricating oil additive.
- the lubricating oil composition according to the third embodiment comprises a lubricating base oil, a polymer chain containing a structural unit represented by the general formula (1) and a structural unit represented by the general formula (2), and An arm part in which the polymer chain is bonded to the core part, the weight average molecular weight Mw is 100,000 or more, and the ratio Mw / Mn of the weight average molecular weight Mw to the number average molecular weight Mn is 1.6.
- a poly (meth) acrylate viscosity index improver which is:
- the lubricating oil composition according to the present embodiment includes a mode including the lubricating base oil and the lubricating oil additive according to the second embodiment.
- the poly (meth) acrylate-based viscosity index improver in the present embodiment is the same as the poly (meth) acrylate-based viscosity index improver in the first embodiment and the second embodiment, and is included in the lubricating oil composition.
- Other additives and solvents to be obtained are the same as the other additives and solvents in the second embodiment, and redundant description is omitted here.
- the lubricating base oil is not particularly limited, and a lubricating base oil used for ordinary lubricating oil can be used. Specifically, a mineral oil base oil, a synthetic oil base oil, or a mixture of two or more kinds of lubricant base oils selected from these can be used.
- mineral oil base oils include, for example, solvent oil removal, solvent extraction, hydrocracking, solvent removal of lubricating oil fractions obtained by distillation under reduced pressure of atmospheric residual oil obtained by atmospheric distillation of crude oil.
- examples thereof include those refined by performing one or more treatments such as dewaxing and hydrorefining, or base oils produced by a method of isomerizing wax isomerized mineral oil or GTL wax (gas-tuly wax).
- Synthetic oil-based lubricating oils include, for example, polybutene or hydrides thereof; poly- ⁇ -olefins such as 1-octene oligomers and 1-decene oligomers or hydrides thereof; ditridecyl glutarate, di-2-ethylhexyl adipate, diisodecyl Diesters such as adipate, ditridecyl adipate, di-2-ethylhexyl sebacate; polyol esters such as trimethylolpropane caprylate, trimethylolpropane pelargonate, pentaerythritol-2-ethylhexanoate, pentaerythritol pelargonate; alkylnaphthalene And aromatic synthetic oils such as alkylbenzene or mixtures thereof.
- the kinematic viscosity of the lubricating base oil at 100 ° C. is preferably 2.5 to 10.0 mm 2 / s, more preferably 3.0 to 8.0 mm 2 / s, still more preferably 3.5 to 6.0 mm 2. / S.
- the viscosity index of the lubricating base oil is preferably 90 to 165, more preferably 100 to 155, and still more preferably 120 to 150.
- the saturated content of the lubricating base oil by chromatographic analysis is preferably 80% or more, more preferably, in order to facilitate the effects of additives such as the poly (meth) acrylate viscosity index improver according to the first embodiment. 85% or more, more preferably 90% or more, and most preferably 95% or more.
- the content of the poly (meth) acrylate viscosity index improver according to the first embodiment is preferably 0.1 to 20.0% by mass, more preferably 0.5 to 15 based on the total amount of the lubricating oil composition. 0.0 mass%, more preferably 1.0 to 10.0 mass%.
- the content is equal to or higher than the lower limit value, it is easy to obtain a sufficient addition effect.
- the content is equal to or lower than the upper limit value, shear stability is increased and fuel consumption sustainability is improved.
- the kinematic viscosity at 100 ° C. of the lubricating oil composition is preferably 3.0 to 16.3 mm 2 / s, more preferably 3.5 to 12.5 mm 2 / s, and still more preferably 4.0 to 9.3 mm 2. / S.
- the kinematic viscosity at 100 ° C. in the present invention means the kinematic viscosity at 100 ° C. defined in JIS K-2283-1993.
- the viscosity index of the lubricating oil composition is preferably 150 to 250, more preferably 160 to 240, and still more preferably 170 to 230.
- the viscosity index is equal to or higher than the lower limit, fuel economy can be further improved while maintaining the HTHS viscosity, and the low temperature viscosity is easily lowered.
- the viscosity index is less than or equal to the above upper limit, low temperature fluidity, solubility of additives, and compatibility with sealing materials can be ensured.
- the viscosity index in the present invention means a viscosity index defined in JIS K 2283-1993.
- the HTHS viscosity at 150 ° C. of the lubricating oil composition is preferably 1.7 mPa ⁇ s or more, more preferably 2.0 mPa ⁇ s or more, still more preferably 2.3 mPa ⁇ s or more, and most preferably 2.6 mPa ⁇ s or more. It is.
- the HTHS viscosity at 150 ° C. is equal to or higher than the lower limit, evaporation of the lubricating oil composition can be suppressed, and lubricity can be ensured.
- the HTHS viscosity at 100 ° C. is not more than the above upper limit value, higher fuel economy can be obtained.
- the HTHS viscosity at 150 ° C. or 100 ° C. means the high temperature high shear viscosity at 150 ° C. or 100 ° C. as defined in ASTM D-4683.
- the MRV viscosity of the lubricating oil composition at ⁇ 40 ° C. is preferably 60,000 mPa ⁇ s or less, more preferably 40,000 mPa ⁇ s or less, and still more preferably 30,000 mPa ⁇ s or less.
- the MRV viscosity at ⁇ 40 ° C. is not more than the above upper limit value, the pumping characteristics at low temperature are excellent.
- the MRV viscosity at ⁇ 40 ° C. means the MRV viscosity at ⁇ 40 ° C. defined by ASTM D-4684.
- the shear stability of the lubricating oil composition is evaluated by, for example, the viscosity reduction rate.
- the viscosity reduction rate of the lubricating oil composition is preferably 5.2% or less, more preferably 5.1% or less, and still more preferably 5.0% or less. When the viscosity reduction rate is less than or equal to the above upper limit, fuel economy is excellent.
- the viscosity reduction rate referred to in the present invention means the viscosity reduction rate in the ultrasonic shear test, and specifically, in accordance with JASO M347-95 (automatic transmission oil shear stability test method), only the sample volume. It means the rate of decrease in thickening due to the viscosity index improver when evaluated under increased conditions.
- PSSI Permanent Shear Stability Index
- the viscosity index improver according to the first embodiment described above, the lubricating oil additive according to the second embodiment, and the lubricating oil composition according to the third embodiment include a lubricating oil for an internal combustion engine, a drive system lubricating oil, and the like. Although it can be used in a wide range of fields, it is particularly useful in the field of lubricating oils for internal combustion engines.
- the fuel of the internal combustion engine may be either gasoline or diesel fuel.
- the poly (meth) acrylate viscosity index improver is a polymerization comprising a core, a structural unit represented by the following general formula (1), and a structural unit represented by the following general formula (2) And three or more arm portions made of chains.
- the poly (meth) acrylate viscosity index improver has a weight average molecular weight Mw (hereinafter, simply referred to as “Mw” in some cases) of less than 100,000, and a weight average molecular weight Mw and a number average molecular weight Mn (hereinafter, depending on circumstances).
- Mw / Mn The ratio Mw / Mn (hereinafter simply referred to as “Mw / Mn”) is 1.6 or less.
- R 1 represents hydrogen or a methyl group
- R 2 represents a group represented by the following general formula (3)
- R 3 represents a straight chain or a branched chain having 5 or less carbon atoms.
- m and n are integers that satisfy m ⁇ 5, n ⁇ 4, and m + n ⁇ 31.
- R 1 may be either hydrogen or a methyl group, but is preferably a methyl group.
- R 2 m is preferably 5 to 16, and n is 4 to 15, more preferably m is 6 to 15, and n is 6 to 10, and m is 7 from the viewpoint of viscosity reduction. More preferably, those having ⁇ 10 and n is 6 ⁇ 9.
- R 1 and R 2 may be the same or different among the structural units.
- the polymer chain constituting the arm portion includes the structural unit represented by the general formula (1) and the structural unit represented by the general formula (2).
- the structural unit represented by the general formula (1) is preferably contained in an amount of 20 to 80% by mass, more preferably 20 to 70% by mass, based on the total amount of the structural units contained in the polymer chain. More preferably, it is contained by mass%.
- the polymer chain preferably contains 20-80% by mass of the structural unit represented by the general formula (2) based on the total amount of the structural unit contained in the polymer chain, from the viewpoint of fuel economy. More preferably, it is contained in an amount of 30 to 80% by mass, and further preferably 50 to 80% by mass.
- the polymer chain is a combination of the structural unit represented by the general formula (1) and the structural unit represented by the general formula (2), and is based on the total amount of the structural units contained in the polymer chain. It is preferable to contain at least 80% by mass, more preferably at least 80% by mass, still more preferably at least 90% by mass, and most preferably at least 100% by mass.
- R 1 and R 3 may be the same or different among the structural units.
- the structural unit in which R 3 is a methyl group is based on the total amount of structural units contained in the polymer chain.
- the content is preferably 5 to 50% by mass, more preferably 10 to 45% by mass, and still more preferably 20 to 45% by mass.
- the structural unit in which R 3 is an alkyl group having 18 carbon atoms is preferably contained in an amount of 5 to 50% by mass based on the total amount of structural units contained in the polymer chain. More preferably, it is contained in an amount of 45% by mass, and more preferably 20-40% by mass.
- the polymer chain may contain only the structural unit represented by the general formula (1) and the structural unit represented by the general formula (2), or may further contain other structural units. Good. Moreover, one end is couple
- R 1 represents hydrogen or a methyl group
- R 4 represents a group represented by the above general formula (3), or a straight chain or having 1 to 18 carbon atoms having a branch having 5 or less carbon atoms.
- An alkyl group is shown, and n is an integer selected so that Mw and Mw / Mn satisfy the above conditions.
- n is an integer of 40 to 450, for example.
- the weight average molecular weight Mw per arm is appropriately selected so that the Mw of the poly (meth) acrylate viscosity index improver satisfies the above-mentioned conditions, but is preferably 33,000 or less, and 30,000. Or less, more preferably 27,000 or less.
- the number average molecular weight Mn per arm is appropriately selected so that the Mw / Mn of the poly (meth) acrylate viscosity index improver satisfies the above-mentioned conditions, but is preferably 2,000 or more. Is more preferably 8,000 or more, and further preferably 8,000 or more.
- the weight average molecular weight Mw of the poly (meth) acrylate viscosity index improver is less than 100,000, and is preferably 90,000 or less, more preferably 80,000 or less, from the viewpoint of shear stability. Preferably, it is 60,000 or less.
- the lower limit of Mw is not particularly limited, but Mw is, for example, 10,000 or more.
- the number average molecular weight Mn of the poly (meth) acrylate viscosity index improver is appropriately selected so that Mw / Mn satisfies the above conditions.
- Mn is preferably 6,000 or more, more preferably 10,000 or more, and further preferably 12,500 or more, from the viewpoint of fuel saving characteristics.
- the upper limit of Mn is not particularly limited, but Mn is, for example, 60,000 or less.
- Mw / Mn is 1.6 or less, but from the viewpoint of fuel economy, it is preferably 1.5 or less, more preferably 1.4 or less, and further preferably 1.3 or less. preferable. Further, Mw / Mn is preferably 1.0 or more, more preferably 1.01 or more, and further preferably 1.02 or more, from the viewpoint of fuel economy.
- weight average molecular weight Mw “weight average molecular weight Mw”, “number average molecular weight Mn” and “ratio Mw / Mn of weight average molecular weight Mw and number average molecular weight Mn” are Mw, Mn and Mw obtained by GPC analysis. / Mn (polystyrene (standard sample) conversion value). Mw / Mn of the poly (meth) acrylate viscosity index improver and Mw and Mn per arm part can be measured, for example, as follows.
- the sample solution is analyzed using a GPC apparatus (Waters Alliance 2695).
- the analysis is carried out using a column having a solvent flow rate of 1 ml / min, an analyzable molecular weight of 10,000 to 256,000, and a refractive index as a detector.
- the relationship between the column retention time and the molecular weight is determined using a polystyrene standard with a clear molecular weight, a calibration curve is separately prepared, and the molecular weight is determined from the obtained retention time.
- the molecular weight of the arm (Mw and Mn) can be calculated by dividing the obtained molecular weight (Mw and Mn) by the number of functional groups of the initiator.
- the core part is derived from a compound having three or more functional groups that react with the carbon-carbon double bond of the acryloyl group.
- the compound having three or more functional groups that react with the carbon-carbon double bond of the acryloyl group include 1,1,1-tris (2-bromoisobutyloxymethylene) ethane, pentaerythritol tetrakis (2-bromoiso). Butyrate) and dipentaerythritol hexakis (2-bromoisobutyrate).
- the number of arm portions of the poly (meth) acrylate viscosity index improver corresponds to the number of the functional groups.
- the number of arm parts that is, the number of functional groups is preferably 2 to 12, more preferably 2 to 8, and still more preferably 3 to 6.
- a polymerization catalyst is added to the mixed solution containing an alkyl (meth) acrylate, an initiator, and a solvent, and alkyl ( A method of polymerizing (meth) acrylate is mentioned.
- alkyl (meth) acrylate an alkyl (meth) acrylate represented by the following general formula (5) and an alkyl (meth) acrylate represented by the following general formula (6) can be used.
- R 1 represents hydrogen or a methyl group
- R 2 represents a group represented by the general formula (3)
- R 3 represents a straight chain or a branch having 5 or less carbon atoms.
- R 1 is preferably a methyl group.
- R 2 is preferably m of 5 to 16 and n of 4 to 15, more preferably m of 6 to 15, and n of 6 to 10, m of 7 to 10, and n of 6 to 10. What is 9 is still more preferable.
- the alkyl (meth) acrylate represented by the general formula (5) and the alkyl (meth) acrylate represented by the general formula (6) can be used.
- the content of the alkyl (meth) acrylate represented by the general formula (5) is preferably 20 to 80% by mass and more preferably 20 to 70% by mass based on the total amount of the alkyl (meth) acrylate.
- the content is preferably 20 to 50% by mass.
- the content of the alkyl (meth) acrylate represented by the general formula (5) is preferably 20 to 80% by mass, and 30 to 80% by mass based on the total amount of the alkyl (meth) acrylate. Is more preferable, and 50 to 80% by mass is even more preferable.
- the alkyl (meth) acrylate represented by the general formula (6) one kind of the alkyl (meth) acrylate represented by the general formula (6) may be used alone, or two or more kinds may be mixed and used. However, it is preferable to use a mixture of two or more.
- the content of methyl (meth) acrylate in which R 2 is a methyl group is preferably 5 to 50% by mass based on the total amount of alkyl (meth) acrylate. More preferably, it is more preferably 20% to 45% by weight.
- the content of alkyl (meth) acrylate in which R 2 is an alkyl group having 18 carbon atoms is preferably 5 to 50% by mass, based on the total amount of alkyl (meth) acrylate, and is 10 to 45% by mass. More preferred is 20 to 40% by mass.
- the initiator one derived from a compound having three or more functional groups that react with the carbon-carbon double bond of the acryloyl group can be used.
- 1,1,1-tris (2-bromoisobutyloxy Methylene) ethane, pentaerythritol tetrakis (2-bromoisobutyrate), dipentaerythritol hexakis (2-bromoisobutyrate) can be used.
- solvent for example, highly refined mineral oil, anisole, and toluene can be used.
- highly refined mineral oil can be exemplified.
- polymerization catalyst examples include copper (II) bromide, tris (2-pyridylmethyl) amine, azobisisobutyronitrile, tin (II) 2-ethylhexanoate, tris [2- (dimethylamino) ethyl]. Amines can be used.
- Preferred examples of the polymerization catalyst include copper (II) bromide, tris (2-pyridylmethyl) amine, azobisisobutyronitrile, and tin (II) 2-ethylhexanoate. You may use these polymerization catalysts individually by 1 type or in mixture of 2 or more types.
- the reaction temperature for polymerizing the alkyl (meth) acrylate can be appropriately selected.
- a preferable reaction temperature is 60 to 100 ° C.
- the Mw / Mn of the resulting poly (meth) acrylate viscosity index improver tends to be 1.6 or less.
- Mw / Mn tends to be 1.0 to 1.2
- Mw / Mn is 1.2 to 1.4.
- Mw / Mn tends to be 1.4 to 1.6.
- the reaction time depends on the reaction conditions such as the raw material alkyl (meth) acrylate, polymerization reagent, solvent and initiator, reaction temperature, and the like, and the desired poly (meth) acrylate Mw and Mw / Mn. It can be selected as appropriate.
- a preferable reaction time is 8 to 16 hours.
- the polymerization of the alkyl (meth) acrylate is preferably performed in a nitrogen atmosphere.
- the lubricating oil additive according to the fifth embodiment of the present invention includes a core, a structural unit represented by the general formula (1), and a polymer chain including the structural unit represented by the general formula (2). And an arm part in which one end of the polymer chain is bonded to the core part, the weight average molecular weight Mw is less than 100,000, and the ratio Mw / Mn between the weight average molecular weight Mw and the number average molecular weight Mn is A poly (meth) acrylate viscosity index improver that is 1.6 or less is contained.
- the poly (meth) acrylate-based viscosity index improver in the present embodiment is the same as the viscosity index improver in the fourth embodiment, and a duplicate description is omitted here.
- the lubricating oil additive may consist only of the above poly (meth) acrylate viscosity index improver, or a mixture of the viscosity index improver and other additives (ie, additive composition). It may be.
- the lubricating oil additive is a mixture of the viscosity index improver and other additives, the mixing ratio is not particularly limited and can be appropriately selected according to the application.
- the lubricating oil additive according to this embodiment may further contain a solvent.
- the other additives, the solvent, and the content of the solvent are the same as the other additives, the solvent, and the content of the solvent in the second embodiment, and redundant descriptions are omitted here.
- the lubricating oil composition according to the sixth embodiment comprises a lubricating base oil, a polymer chain containing a structural unit represented by the general formula (1) and a structural unit represented by the general formula (2), and The polymer chain has an arm part bonded to the core part, the weight average molecular weight Mw is less than 100,000, and the ratio Mw / Mn between the weight average molecular weight Mw and the number average molecular weight Mn is 1.6.
- a poly (meth) acrylate viscosity index improver which is:
- the lubricating oil composition according to the present embodiment includes a mode containing the lubricating base oil and the lubricating oil additive according to the fifth embodiment.
- the poly (meth) acrylate-based viscosity index improver in the present embodiment is the same as the poly (meth) acrylate-based viscosity index improver in the fourth and fifth embodiments, and is included in the lubricating oil composition.
- Other additives and solvents to be obtained are the same as the other additives and solvents in the fifth embodiment, and redundant description is omitted here.
- Lubricating oil base oil is the same as the lubricating oil base oil in the third embodiment, and redundant description is omitted here.
- the content of the poly (meth) acrylate viscosity index improver according to the fourth embodiment is preferably 0.1 to 20.0 mass%, more preferably 0.5 to 15 based on the total amount of the lubricating oil composition. 0.0 mass%, more preferably 1.0 to 10.0 mass%.
- the content is equal to or higher than the lower limit value, it is easy to obtain a sufficient addition effect.
- the content is equal to or lower than the upper limit value, shear stability is increased and fuel consumption sustainability is improved.
- the kinematic viscosity at 100 ° C. of the lubricating oil composition is preferably 2.0 to 16.3 mm 2 / s, more preferably 2.5 to 12.5 mm 2 / s, still more preferably 3.0 to 10.0 mm 2. / S.
- the kinematic viscosity at 100 ° C. in the present invention means the kinematic viscosity at 100 ° C. defined in JIS K-2283-1993.
- the viscosity index of the lubricating oil composition is preferably 130 to 250, more preferably 140 to 240, and still more preferably 160 to 230.
- the viscosity index is equal to or higher than the lower limit, fuel economy can be further improved while maintaining the HTHS viscosity, and the low temperature viscosity is easily lowered.
- the viscosity index is less than or equal to the above upper limit, low temperature fluidity, solubility of additives, and compatibility with sealing materials can be ensured.
- the viscosity index in the present invention means a viscosity index defined in JIS K 2283-1993.
- the BF viscosity at ⁇ 40 ° C. of the lubricating oil composition is preferably 20,000 mPa ⁇ s or less, more preferably 18,000 mPa ⁇ s or less, and further preferably 16,000 mPa ⁇ s or less.
- the BF viscosity at ⁇ 40 ° C. is not more than the above upper limit value, the low temperature fluidity is excellent, and the lubricating oil easily flows at low temperatures.
- the BF viscosity at ⁇ 40 ° C. means the BF viscosity at ⁇ 40 ° C. defined by JPI-5S-26-99.
- the shear rate of the lubricating oil composition is preferably 8% or less, more preferably 5% or less, and even more preferably 2% or less. When the shear rate is less than or equal to the above upper limit, the viscosity of the formulated oil can be further reduced.
- the shear rate in the present invention was evaluated by a mechanical shearing method using a KRL tapered roller bearing (test method: CEC L45-A-99) in order to simulate the shear stability in a real gear. Means shear rate. More specifically, a Group II base oil prepared with a viscosity index improver of 2% by mass is operated continuously for 120 hours in accordance with the above test method.
- the shear rate is the rate of decrease in kinematic viscosity at 100 ° C. before and after the test (the value (%) obtained by dividing the difference from the kinematic viscosity before and after the test by the kinematic viscosity before the test).
- the viscosity index improver according to the fourth embodiment described above, the lubricating oil additive according to the fifth embodiment, and the lubricating oil composition according to the sixth embodiment include a lubricating oil for an internal combustion engine, a drive system lubricating oil, and the like. Although it can be used in a wide range of fields, it is particularly useful in the field of drive system lubricants.
- the driving device in this case may be any of an automatic transmission (AT), a continuously variable automatic transmission (CVT), and a stepped transmission (TM).
- Example 1-1 A poly (meth) acrylate viscosity index improver was synthesized under the following conditions (referred to as “synthesis condition 1-1”).
- kinematic viscosity 4.2mm 2 / s
- 117g were added, and a homogeneous solution under stirring. This solution was cooled to 0 ° C. in an ice bath, and vacuum degassing / nitrogen purging of the reaction system was performed 5 times using a diaphragm pump.
- the weight average molecular weight Mw and the number average molecular weight Mn were measured by GPC analysis. As a result, the weight average molecular weight Mw was 241,000, the number average molecular weight Mn was 165,000, and Mw / Mn was 1.46.
- the procedure of GPC analysis is as follows.
- Tetrahydrofuran was used as a solvent and diluted to prepare a solution having a sample concentration of 2% by mass.
- the sample solution was analyzed using a GPC apparatus (Waters Alliance 2695). The analysis was carried out using a column with a solvent flow rate of 1 ml / min and an analyzable molecular weight of 10,000 to 256,000 and using the refractive index as a detector. The relationship between the column retention time and the molecular weight was determined using a polystyrene standard with a clear molecular weight, a calibration curve was prepared separately, and the molecular weight was determined from the obtained retention time.
- Example 1-7 A poly (meth) acrylate viscosity index improver was synthesized under the following conditions (referred to as “synthesis condition 1-2”).
- C1-MA methyl methacrylate
- A2 2-octyl 18 g of dodecyl methacrylate
- C18-MA stearyl methacrylate
- This solution was cooled to 0 ° C. in an ice bath, and vacuum degassing / nitrogen purging of the reaction system was performed 5 times using a diaphragm pump. Furthermore, a complex solution in which 0.004 g of copper (II) bromide and 0.005 g of tris (2-pyridylmethyl) amine as a polymerization catalyst were dissolved in 2.0 g of anisole from a sample introduction port under a nitrogen flow, and 2-ethyl A solution in which 0.17 g of tin (II) hexanoate was dissolved in 3 g of highly refined mineral oil was added, followed by polymerization by stirring at a solution temperature of 70 ° C. for 12 hours under a nitrogen atmosphere. A solution containing a poly (meth) acrylate viscosity index improver was obtained.
- the obtained poly (meth) acrylate viscosity index improver was subjected to GPC analysis in the same manner as in Example 1-1.
- the weight average molecular weight Mw was 252,000
- the number average molecular weight Mn was 227,000
- Mw / Mn was 1.11.
- C1-MA methyl methacrylate
- A2 2-octyl 18 g of dodecyl methacrylate
- C18-MA stearyl methacrylate
- CDTBA cumyldithiobenzoic acid
- This solution was cooled to 0 ° C. in an ice bath, and vacuum degassing / nitrogen purging of the reaction system was performed 5 times using a diaphragm pump. Furthermore, after introducing 0.003 g of azobisisobutyronitrile (AIBN) as an initiator from a sample inlet under a nitrogen flow, polymerization was carried out at a solution temperature of 90 ° C. for 12 hours in a nitrogen atmosphere, and poly (meta ) A solution containing an acrylate viscosity index improver was obtained.
- AIBN azobisisobutyronitrile
- the obtained poly (meth) acrylate viscosity index improver was subjected to GPC analysis in the same manner as in Example 1-1.
- the weight average molecular weight Mw was 285,000
- the number average molecular weight Mn was 172,000
- Mw / Mn was 1.65.
- a 300 ml four-necked reaction flask equipped with a stirring blade (with a vacuum seal), a Dimroth cooler, a three-way cock for introducing nitrogen, and a dropping funnel for introducing sample is charged with 60 g of highly purified mineral oil as a solvent, and an oil at 85 ° C. The mixture was stirred for 1 hour while purging with nitrogen in the bath.
- methyl methacrylate (C1-MA) as a raw material monomer
- 18 g of stearyl methacrylate (C18-MA) and dodecyl methacrylate (R 1 in formula (6) is a methyl group
- R 2 is a dodecyl group ( A straight-chain alkyl group having 12 carbon atoms, hereinafter referred to as “C12-MA”)
- a raw material in which 0.035 g of azobisisobutyronitrile (AIBN) is mixed as an initiator was dropped into the reaction flask over 120 minutes. Thereafter, polymerization was carried out for 8 hours while maintaining stirring at 85 ° C.
- AIBN azobisisobutyronitrile
- the obtained poly (meth) acrylate viscosity index improver was subjected to GPC analysis in the same manner as in Example 1-1.
- the weight average molecular weight Mw was 108,000
- the number average molecular weight Mn was 44,000
- Mw / Mn was 2.44.
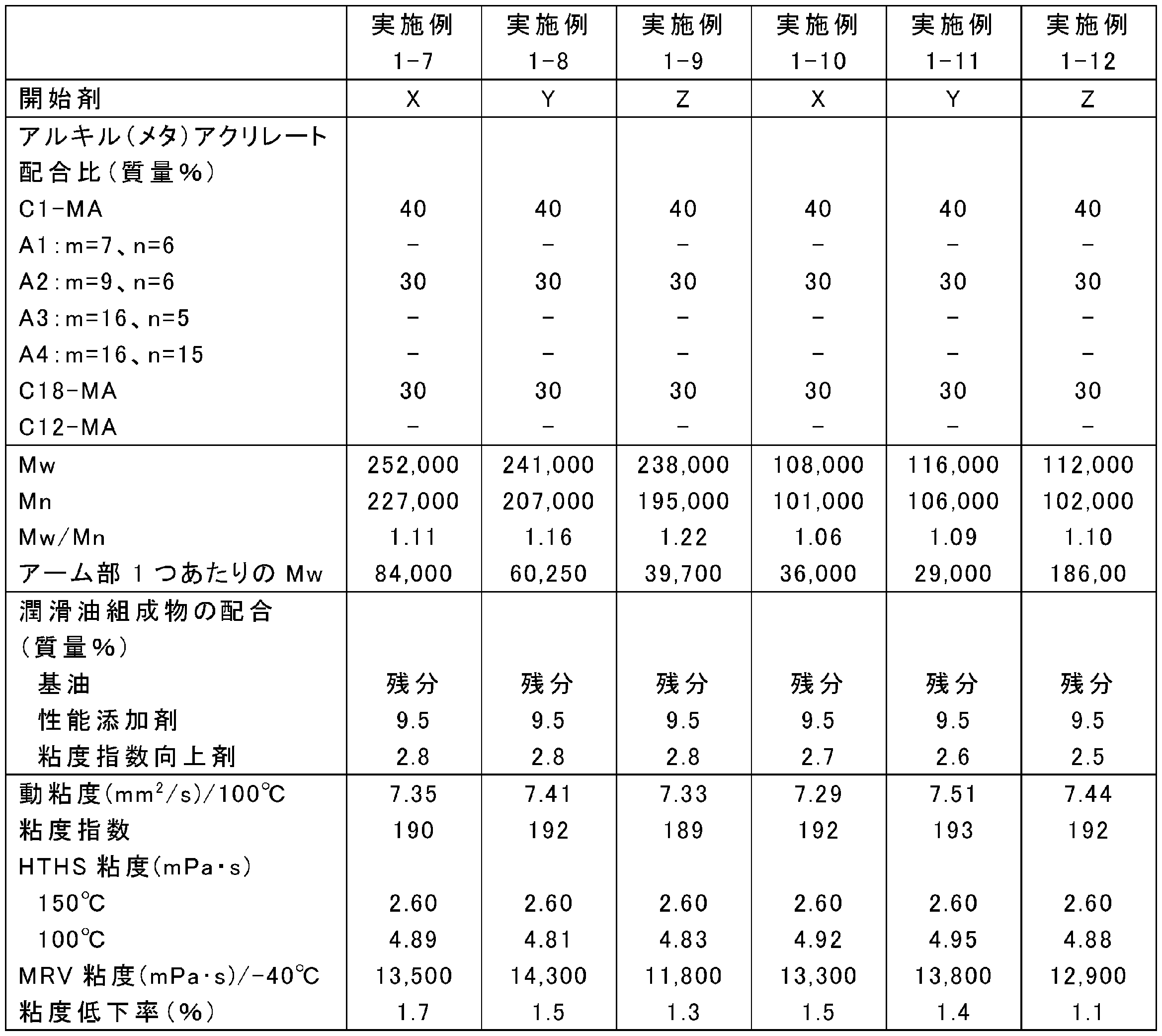
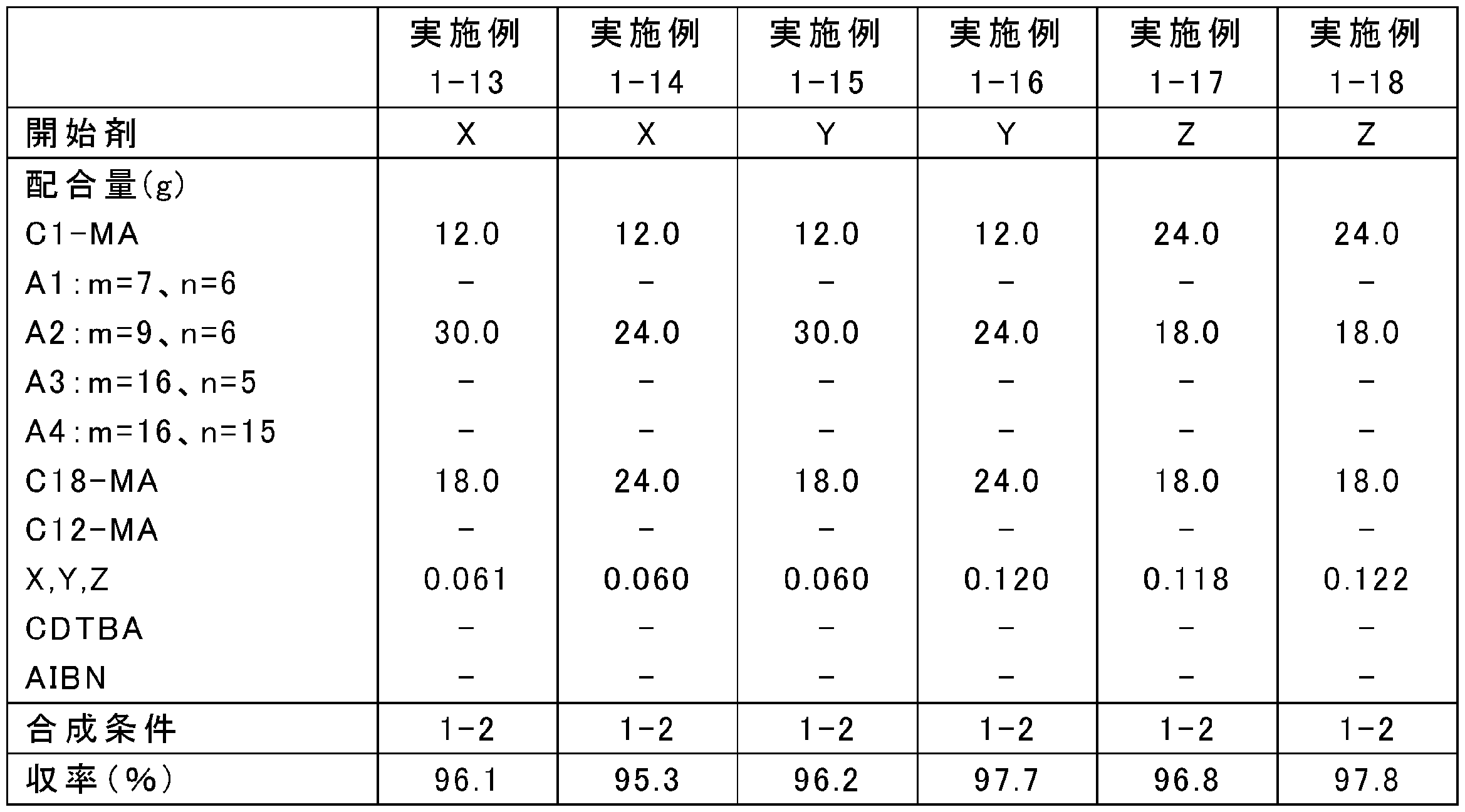
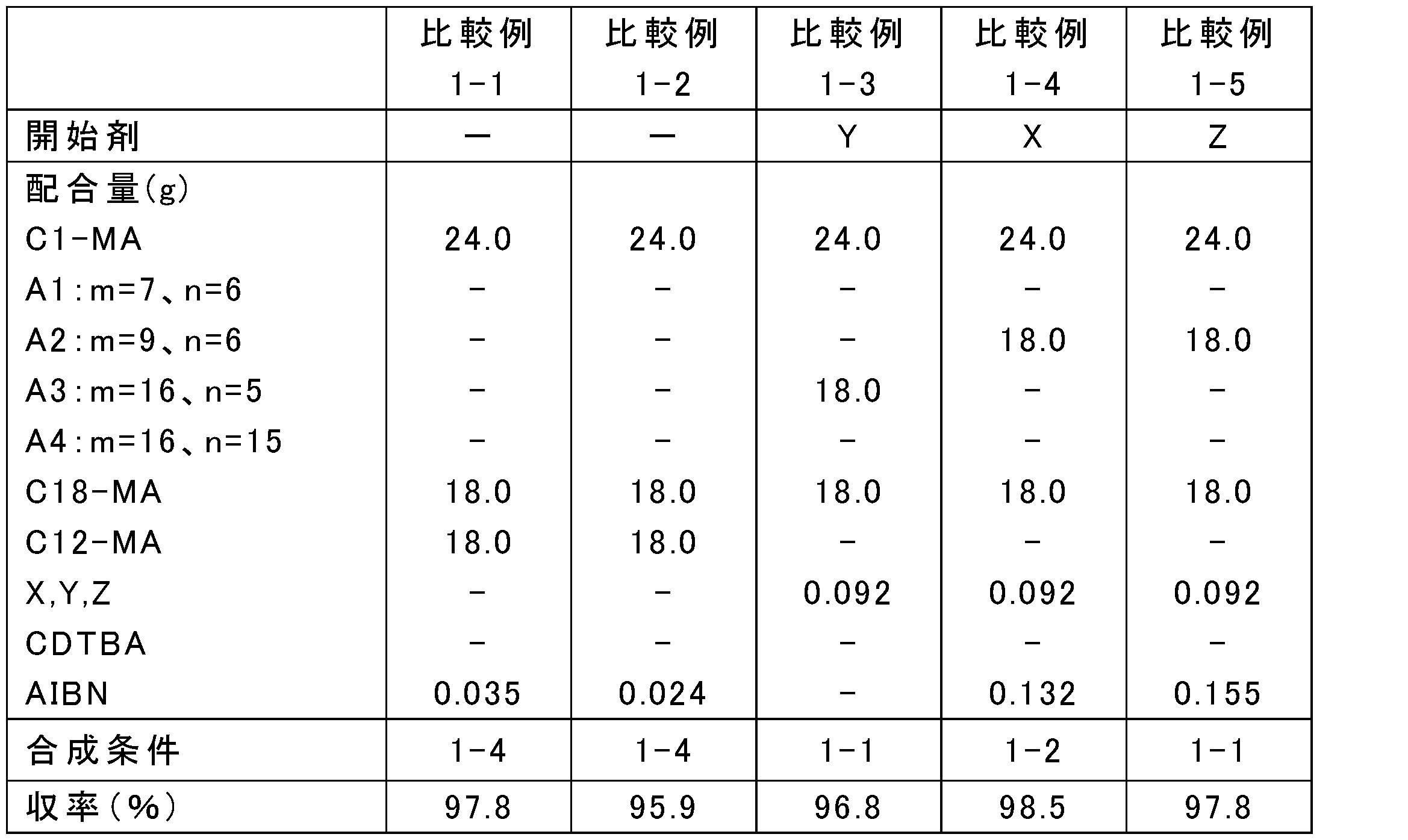
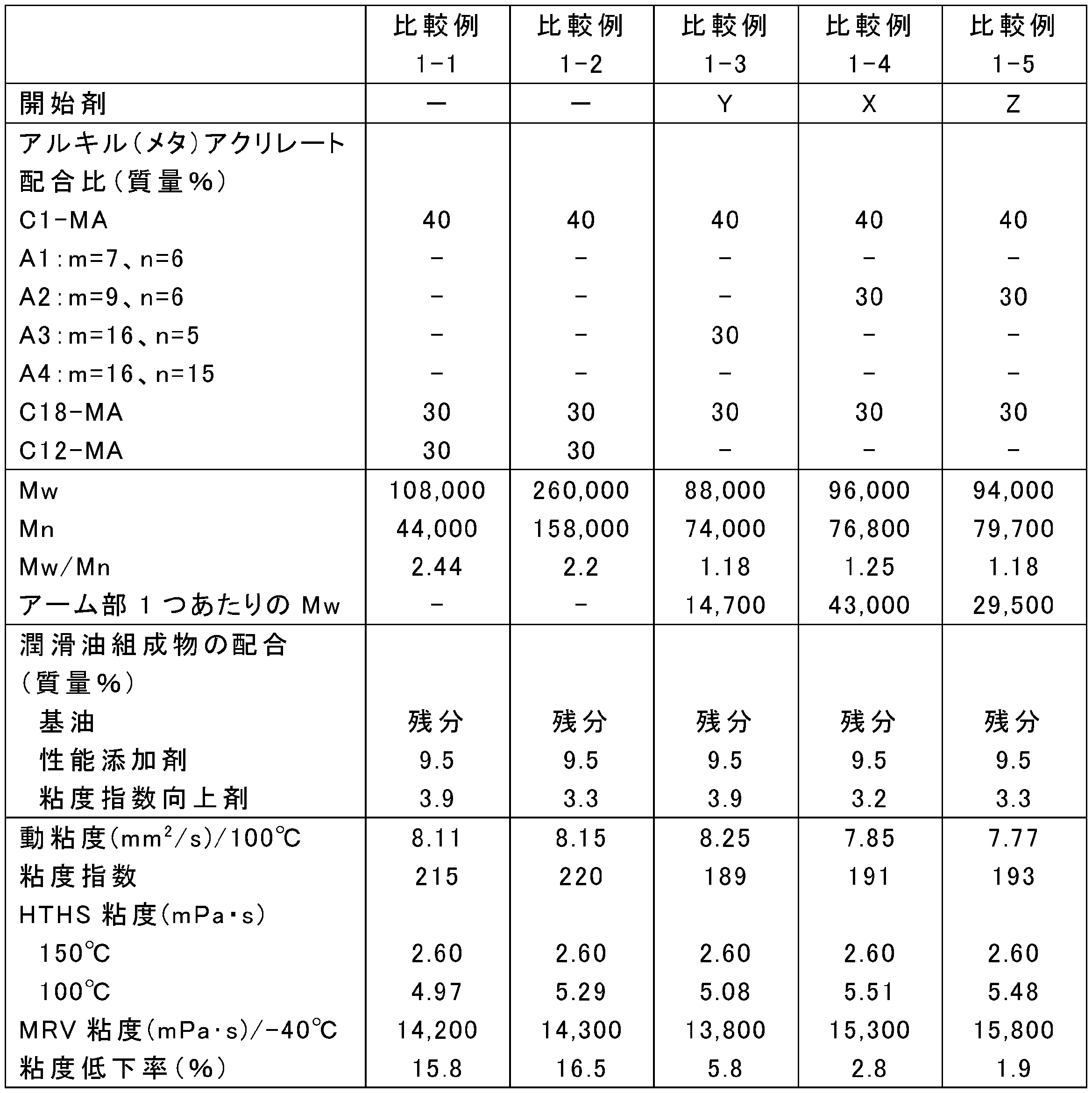
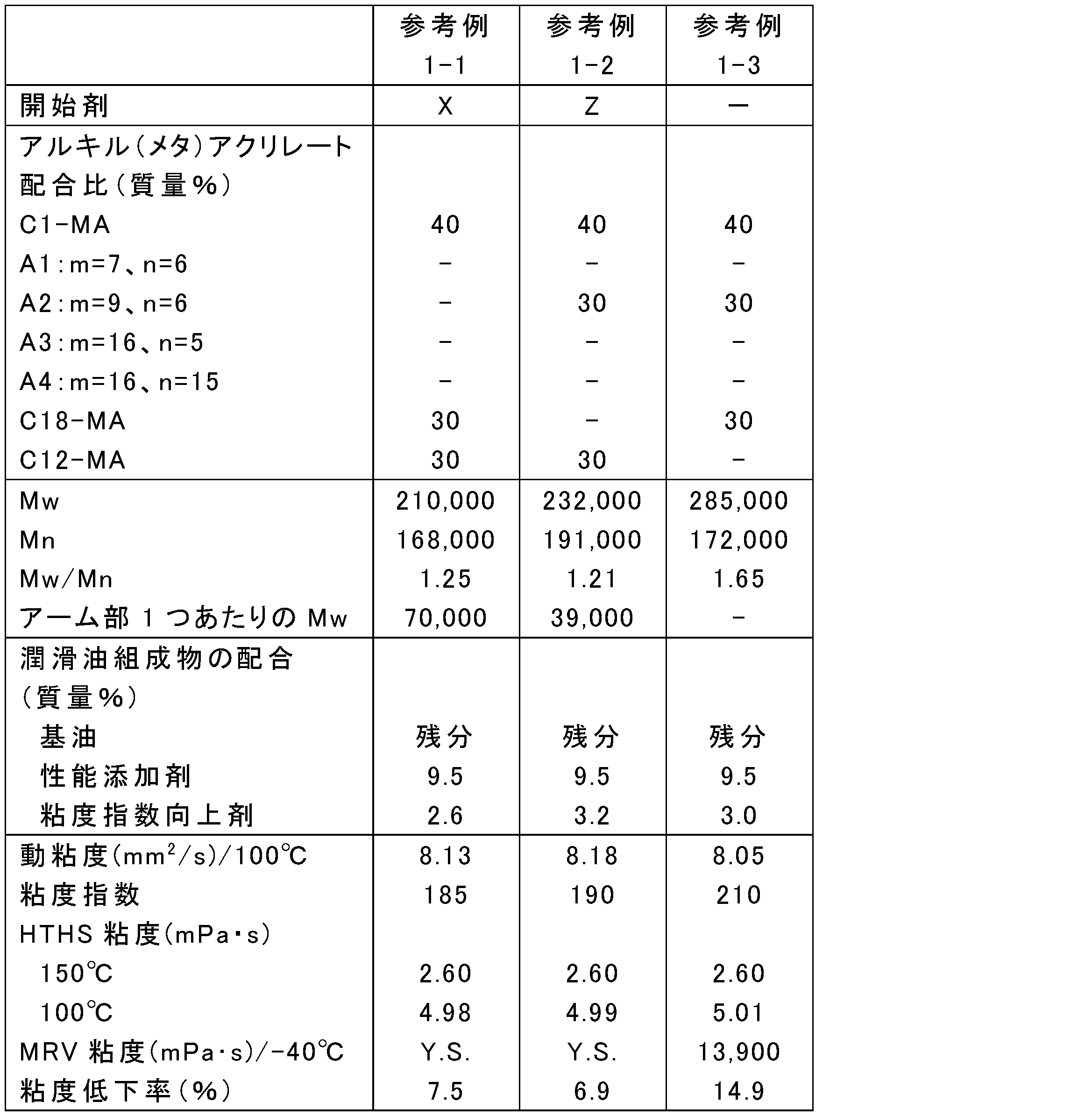
- the blending amount of the raw materials was changed as shown in Tables 1, 3, 5, 7, 9, and 11, and the rest was the same as in any one of the above synthesis conditions 1-1 to 1-4, and poly (meth) acrylate
- a system viscosity index improver was synthesized.
- Y represents pentaerythritol tetrakis (2-bromoisobutyrate) as a tetrafunctional initiator
- Z represents dipentaerythritol hexakis (2-bromoisobutyrate) as a hexafunctional initiator.
- Tables 2, 4, 6, 8, 10, and 12 show Mw, Mn, and Mw / Mn of the obtained poly (meth) acrylate viscosity index improvers.
- ⁇ Preparation of lubricating oil composition Poly (meth) acrylate viscosity index improvers obtained in Examples 1-1 to 1-24, Comparative Examples 1-1 to 1-5 and Reference Examples 1-1 to 1-3, respectively, and metal (calcium Performance additives including sulfonate-based detergent, ashless dispersant (succinimide), friction modifier (glycerin monooleate) and antiwear agent (zinc dithiophosphate), highly refined mineral oil (Group III base oil, 100 ° C. Kinematic viscosity: 4.2 mm 2 / s, VI: 125) were blended at the ratios shown in Tables 2, 4, 6, 8, 10, and 12 to prepare lubricating oil compositions.
- the viscosity reduction rate of each of the lubricating oil compositions of Examples 1-1 to 1-24, Comparative Examples 1-1 to 1-5, and Reference Examples 1-1 to 1-3 was measured as follows. That is, in accordance with JASO M347-95 (Automatic Transmission Oil Shear Stability Test Method), the rate of decrease in thickening by the viscosity index improver when evaluated under the condition that only the sample volume was increased was measured. More specifically, after adjusting the output with the standard oil A defined in the ASTM test method, a shear test was performed under the conditions of an amplitude of 28 ⁇ m, a vibration frequency of 10 KHz, an irradiation time of 10 minutes, and a sample volume of 50 mL. The permanent shear stability index PSSI of the polymer calculated based on the measured kinematic viscosity was calculated. The results are shown in Tables 2, 4, 6, 8, 10, and 12.
- Example 2-1 A poly (meth) acrylate viscosity index improver was synthesized under the following conditions (referred to as “synthesis condition 2-1”).
- the weight average molecular weight Mw and the number average molecular weight Mn were measured by GPC analysis. As a result, the weight average molecular weight Mw was 97,000, the number average molecular weight Mn was 66,000, and Mw / Mn was 1.46.
- the procedure of GPC analysis is as follows.
- Tetrahydrofuran was used as a solvent and diluted to prepare a solution having a sample concentration of 2% by mass.
- the sample solution was analyzed using a GPC apparatus (Waters Alliance 2695). The analysis was carried out using a column with a solvent flow rate of 1 ml / min and an analyzable molecular weight of 10,000 to 256,000 and using the refractive index as a detector. The relationship between the column retention time and the molecular weight was determined using a polystyrene standard with a clear molecular weight, a calibration curve was prepared separately, and the molecular weight was determined from the obtained retention time.
- Example 2-7 A poly (meth) acrylate viscosity index improver was synthesized under the following conditions (referred to as “synthesis condition 2-2”).
- C1-MA methyl methacrylate
- A2 2-octyl 18 g of dodecyl methacrylate
- C18-MA stearyl methacrylate
- This solution was cooled to 0 ° C. in an ice bath, and vacuum degassing / nitrogen purging of the reaction system was performed 5 times using a diaphragm pump. Furthermore, a complex solution in which 0.004 g of copper (II) bromide and 0.005 g of tris (2-pyridylmethyl) amine as a polymerization catalyst were dissolved in 2.0 g of anisole from a sample introduction port under a nitrogen flow, and 2-ethyl A solution in which 0.17 g of tin (II) hexanoate was dissolved in 3 g of highly refined mineral oil was added, followed by polymerization by stirring at a solution temperature of 70 ° C. for 12 hours under a nitrogen atmosphere. A solution containing a poly (meth) acrylate viscosity index improver was obtained.
- the obtained poly (meth) acrylate viscosity index improver was subjected to GPC analysis in the same manner as in Example 2-1.
- the weight average molecular weight Mw was 95,000
- the number average molecular weight Mn was 86,000
- Mw / Mn was 1.11.
- C1-MA methyl methacrylate
- A2 2-octyl 18 g of dodecyl methacrylate
- C18-MA stearyl methacrylate
- CDTBA cumyldithiobenzoic acid
- This solution was cooled to 0 ° C. in an ice bath, and vacuum degassing / nitrogen purging of the reaction system was performed 5 times using a diaphragm pump. Furthermore, after introducing 0.003 g of azobisisobutyronitrile (AIBN) as an initiator from a sample inlet under a nitrogen flow, polymerization was carried out at a solution temperature of 90 ° C. for 12 hours in a nitrogen atmosphere, and poly (meta ) A solution containing an acrylate viscosity index improver was obtained.
- AIBN azobisisobutyronitrile
- the obtained poly (meth) acrylate viscosity index improver was subjected to GPC analysis in the same manner as in Example 2-1.
- the weight average molecular weight Mw was 65,000
- the number average molecular weight Mn was 78,000
- Mw / Mn was 1.65.
- a 300 ml four-necked reaction flask equipped with a stirring blade (with a vacuum seal), a Dimroth cooler, a three-way cock for introducing nitrogen, and a dropping funnel for introducing sample is charged with 60 g of highly purified mineral oil as a solvent, and an oil at 85 ° C. The mixture was stirred for 1 hour while purging with nitrogen in the bath.
- methyl methacrylate (C1-MA) as a raw material monomer
- 18 g of stearyl methacrylate (C18-MA) and dodecyl methacrylate (R 1 in formula (6) is a methyl group
- R 2 is a dodecyl group ( A straight-chain alkyl group having 12 carbon atoms, hereinafter referred to as “C12-MA”)
- a raw material in which 0.035 g of azobisisobutyronitrile (AIBN) is mixed as an initiator was dropped into the reaction flask over 120 minutes. Thereafter, polymerization was carried out for 8 hours while maintaining stirring at 85 ° C.
- AIBN azobisisobutyronitrile
- the obtained poly (meth) acrylate viscosity index improver was subjected to GPC analysis in the same manner as in Example 2-1.
- the weight average molecular weight Mw was 78,000
- the number average molecular weight Mn was 32,000
- Mw / Mn was 2.44.
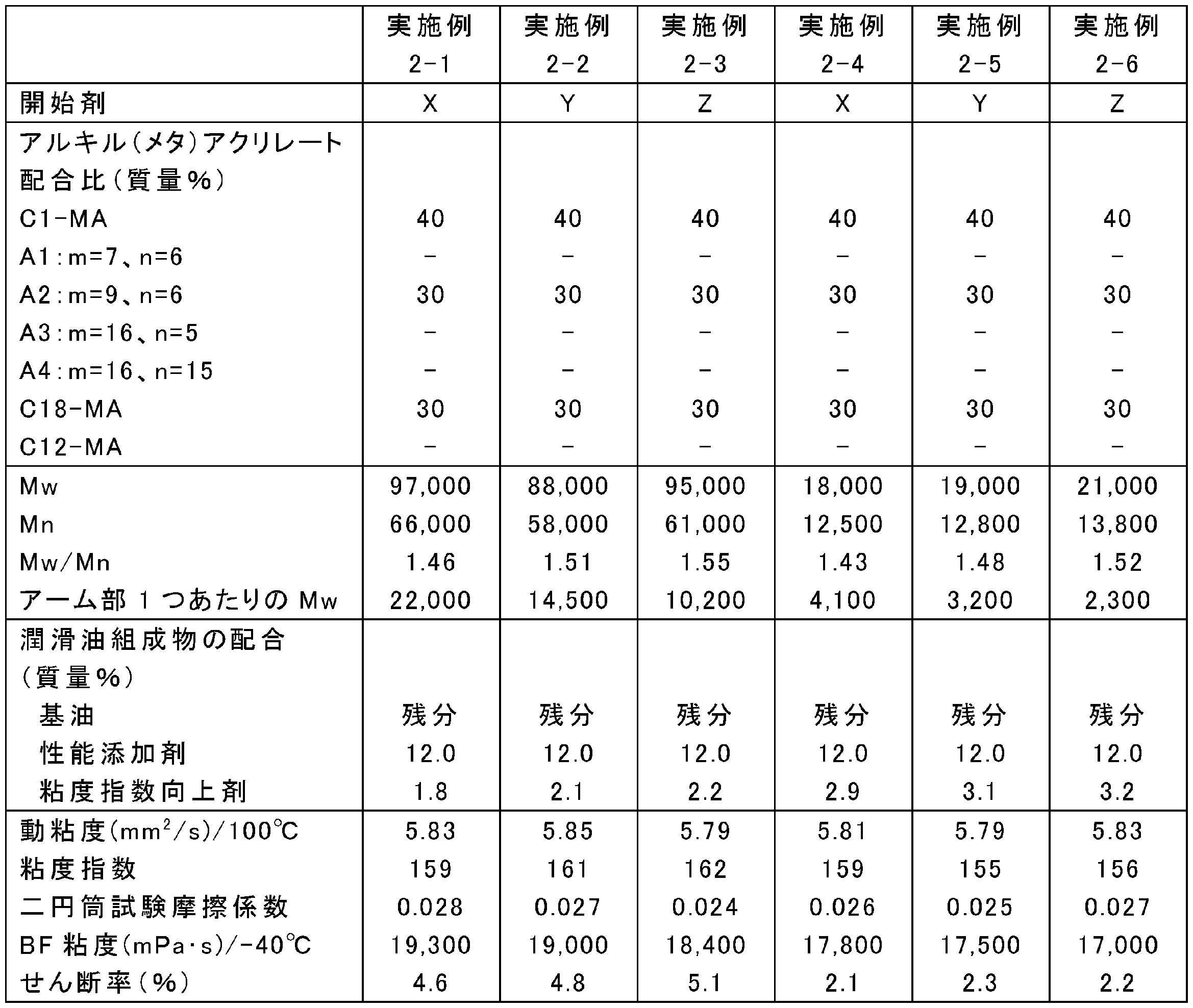
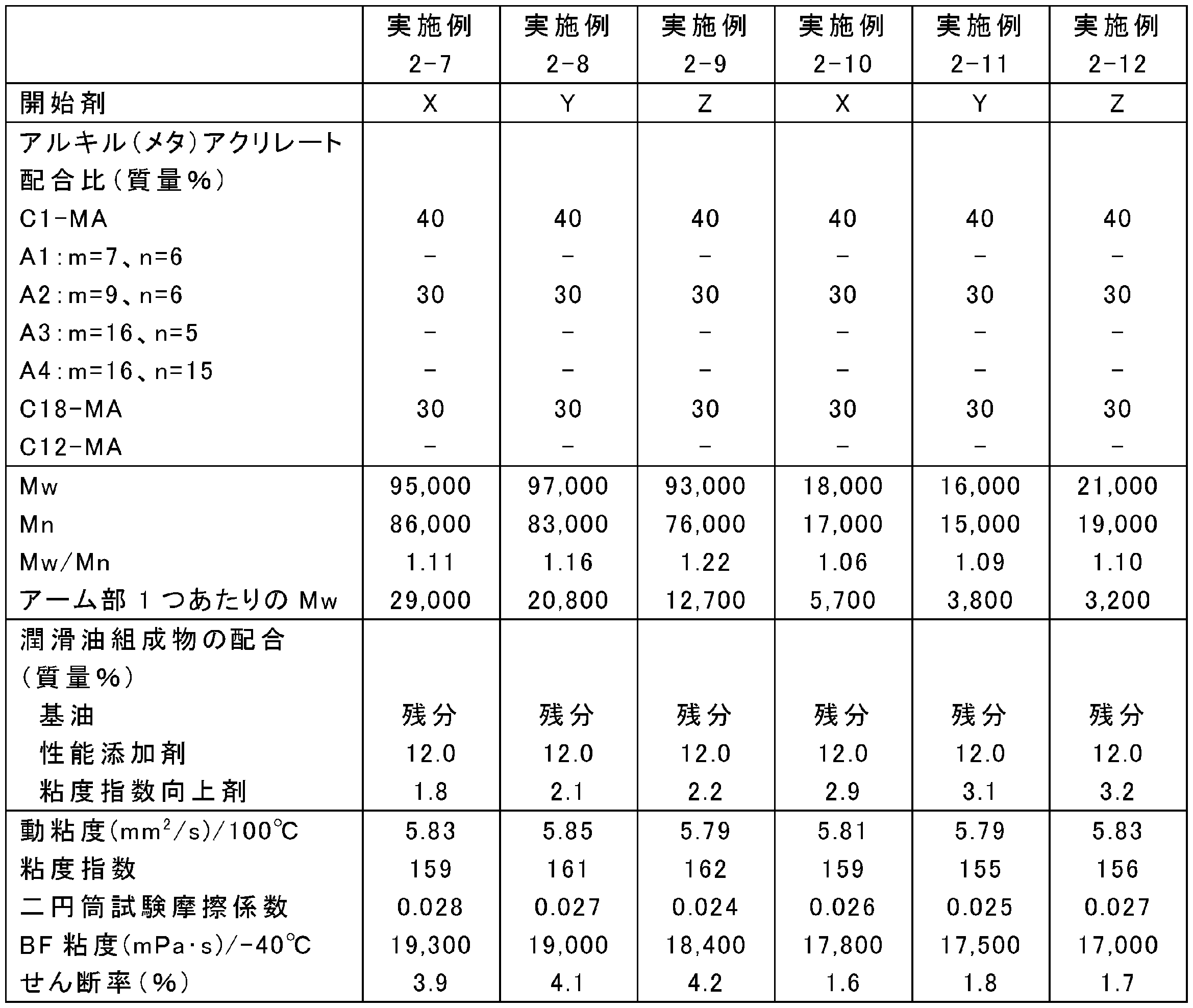
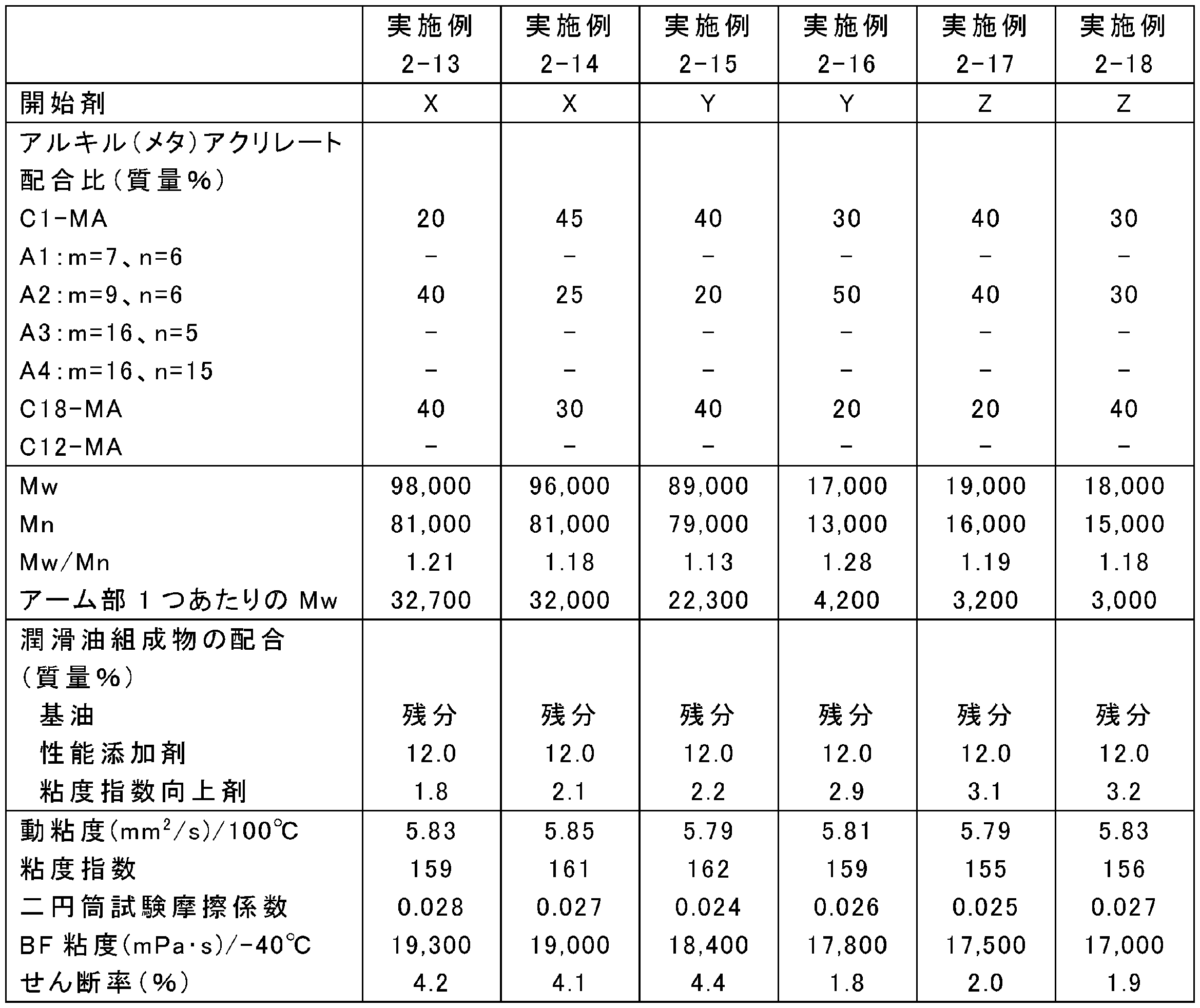
- the blending amounts of the raw materials were changed as shown in Tables 13, 15, 17, 19, 21, 23, and 25. Otherwise, the same as in any of the above synthesis conditions 2-1 to 2-4, poly (meta ) An acrylate viscosity index improver was synthesized.
- Y represents pentaerythritol tetrakis (2-bromoisobutyrate) as a tetrafunctional initiator
- Z represents dipentaerythritol hexakis (2-bromoisobutyrate) as a hexafunctional initiator.
- Tables 14, 16, 18, 20, 22, 24, and 26 show Mw, Mn, and Mw / Mn of the obtained poly (meth) acrylate viscosity index improvers.
- ⁇ Preparation of lubricating oil composition A poly (meth) acrylate viscosity index improver obtained in Examples 2-1 to 2-34 and Comparative Examples 2-1 to 2-6, and a metal (TBN 300 mg KOH / g calcium sulfonate) detergent, Ashless residue (succinimide), friction modifier (oleylamide), antiwear agent (phosphoric acid), antioxidant (diphenylamine), metal deactivator (thiadiazole), and sulfur-based additive (sulfurized ester) ) And highly refined mineral oil (Group II base oil, kinematic viscosity at 100 ° C .: 3.3 mm 2 / s, VI: 110), Tables 14, 16, 18, 20, 22, 24, 26 The lubricating oil composition was prepared by blending at the ratio shown in the following.
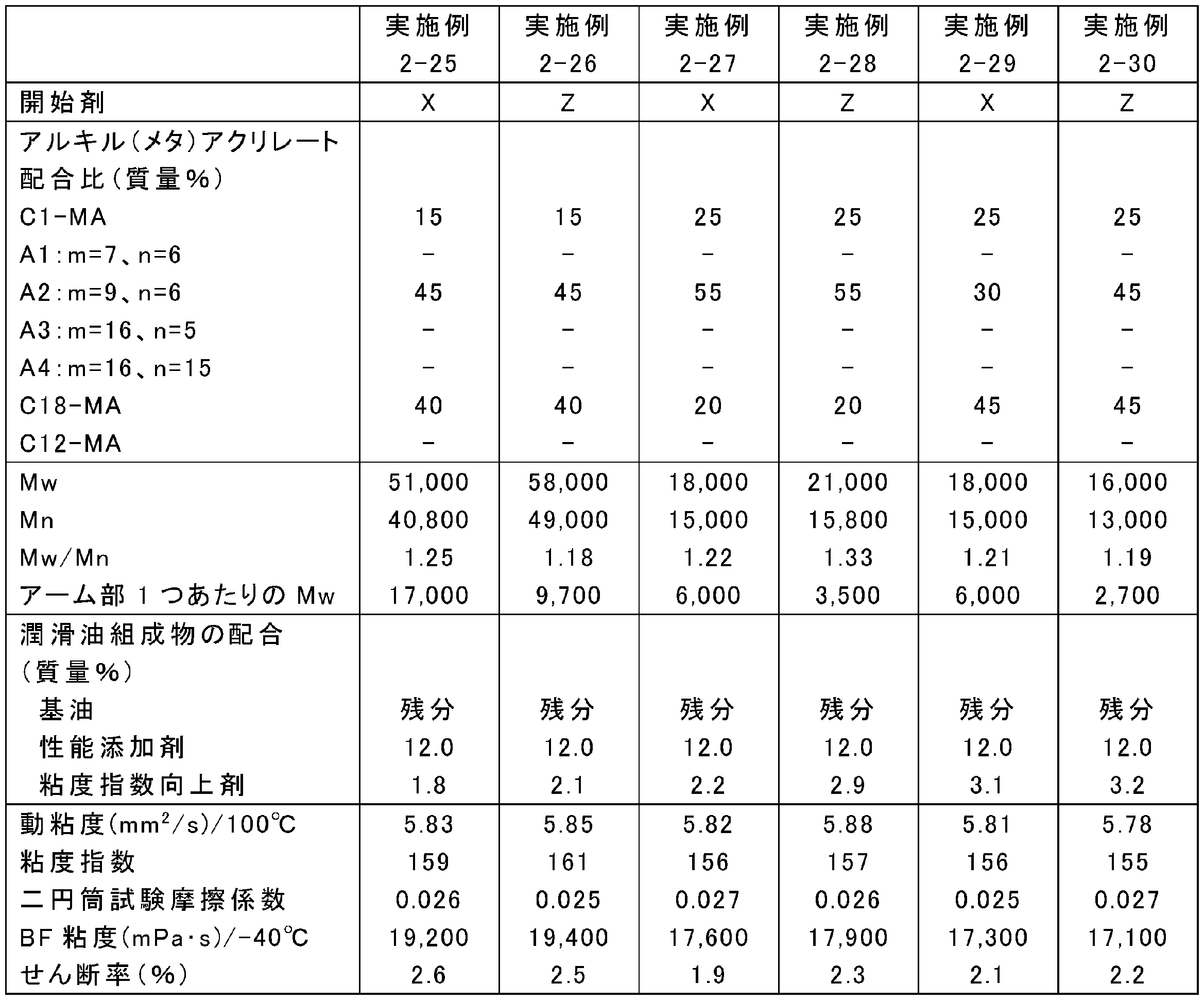
- the friction characteristics of the lubricating oil compositions of Examples 2-1 to 2-34 and Comparative Examples 2-1 to 2-6 were measured using a two-cylinder rolling and sliding friction tester. Evaluated by coefficient. Specifically, the friction coefficient for 10 minutes from the start of the test was averaged under the conditions of a test temperature of 80 ° C., a load of 142 N, a surface pressure of 0.48 GPa, a peripheral speed of 1.0 m / s, and a slip ratio of 5.1%. The results are shown in Tables 14, 16, 18, 20, 22, 24 and 26.
Abstract
Description
第1実施形態に係るポリ(メタ)アクリレート系粘度指数向上剤は、コア部と、下記一般式(1)で表される構造単位及び下記一般式(2)で表される構造単位を含む重合鎖からなるアーム部の3つ以上と、を有する。該ポリ(メタ)アクリレート系粘度指数向上剤の重量平均分子量Mw(以下、場合により単に「Mw」という。)は100,000以上であり、重量平均分子量Mwと数平均分子量Mn(以下、場合により単に「Mw」という。)との比Mw/Mn(以下、場合により単に「Mw/Mn」という。)は1.6以下である。
本発明の第2実施形態に係る潤滑油添加剤は、コア部と、上記一般式(1)で表される構造単位及び上記一般式(2)で表される構造単位を含む重合鎖からなり且つ該重合鎖の一端がコア部に結合しているアーム部と、を有し、重量平均分子量Mwが100,000以上であり、重量平均分子量Mwと数平均分子量Mnとの比Mw/Mnが1.6以下であるポリ(メタ)アクリレート系粘度指数向上剤を含有する。なお、本実施形態におけるポリ(メタ)アクリレート系粘度指数向上剤は、上記第1実施形態における粘度指数向上剤と同様であり、ここでは重複する説明を省略する。
第3実施形態に係る潤滑油組成物は、潤滑油基油と、上記一般式(1)で表される構造単位及び上記一般式(2)で表される構造単位を含む重合鎖からなり且つ該重合鎖がコア部に結合しているアーム部と、を有し、重量平均分子量Mwが100,000以上であり、重量平均分子量Mwと数平均分子量Mnとの比Mw/Mnが1.6以下であるポリ(メタ)アクリレート系粘度指数向上剤と、を含有する。ここで、本実施形態に係る潤滑油組成物には、潤滑油基油と上記第2実施形態に係る潤滑油添加剤とを含有する態様が包含される。本実施形態におけるポリ(メタ)アクリレート系粘度指数向上剤は上記第1実施形態及び第2実施形態におけるポリ(メタ)アクリレート系粘度指数向上剤と同様であり、また、潤滑油組成物に含まれ得る他の添加剤及び溶剤は第2実施形態における他の添加剤及び溶剤と同様であり、ここでは重複する説明を省略する。
第4実施形態に係るポリ(メタ)アクリレート系粘度指数向上剤は、コア部と、下記一般式(1)で表される構造単位及び下記一般式(2)で表される構造単位を含む重合鎖からなるアーム部の3つ以上と、を有する。該ポリ(メタ)アクリレート系粘度指数向上剤の重量平均分子量Mw(以下、場合により単に「Mw」という。)は100,000未満であり、重量平均分子量Mwと数平均分子量Mn(以下、場合により単に「Mw」という。)との比Mw/Mn(以下、場合により単に「Mw/Mn」という。)は1.6以下である。
本発明の第5実施形態に係る潤滑油添加剤は、コア部と、上記一般式(1)で表される構造単位及び上記一般式(2)で表される構造単位を含む重合鎖からなり且つ該重合鎖の一端がコア部に結合しているアーム部と、を有し、重量平均分子量Mwが100,000未満であり、重量平均分子量Mwと数平均分子量Mnとの比Mw/Mnが1.6以下であるポリ(メタ)アクリレート系粘度指数向上剤を含有する。なお、本実施形態におけるポリ(メタ)アクリレート系粘度指数向上剤は、上記第4実施形態における粘度指数向上剤と同様であり、ここでは重複する説明を省略する。
第6実施形態に係る潤滑油組成物は、潤滑油基油と、上記一般式(1)で表される構造単位及び上記一般式(2)で表される構造単位を含む重合鎖からなり且つ該重合鎖がコア部に結合しているアーム部と、を有し、重量平均分子量Mwが100,000未満であり、重量平均分子量Mwと数平均分子量Mnとの比Mw/Mnが1.6以下であるポリ(メタ)アクリレート系粘度指数向上剤と、を含有する。ここで、本実施形態に係る潤滑油組成物には、潤滑油基油と上記第5実施形態に係る潤滑油添加剤とを含有する態様が包含される。本実施形態におけるポリ(メタ)アクリレート系粘度指数向上剤は上記第4実施形態及び第5実施形態におけるポリ(メタ)アクリレート系粘度指数向上剤と同様であり、また、潤滑油組成物に含まれ得る他の添加剤及び溶剤は第5実施形態における他の添加剤及び溶剤と同様であり、ここでは重複する説明を省略する。
下記の条件(「合成条件1-1」とする)でポリ(メタ)アクリレート系粘度指数向上剤を合成した。
下記の条件(「合成条件1-2」とする)でポリ(メタ)アクリレート系粘度指数向上剤を合成した。
下記の条件(「合成条件1-3」とする)でポリ(メタ)アクリレート系粘度指数向上剤を合成した。
下記の条件(「合成条件1-4」とする)でポリ(メタ)アクリレート系粘度指数向上剤を合成した。
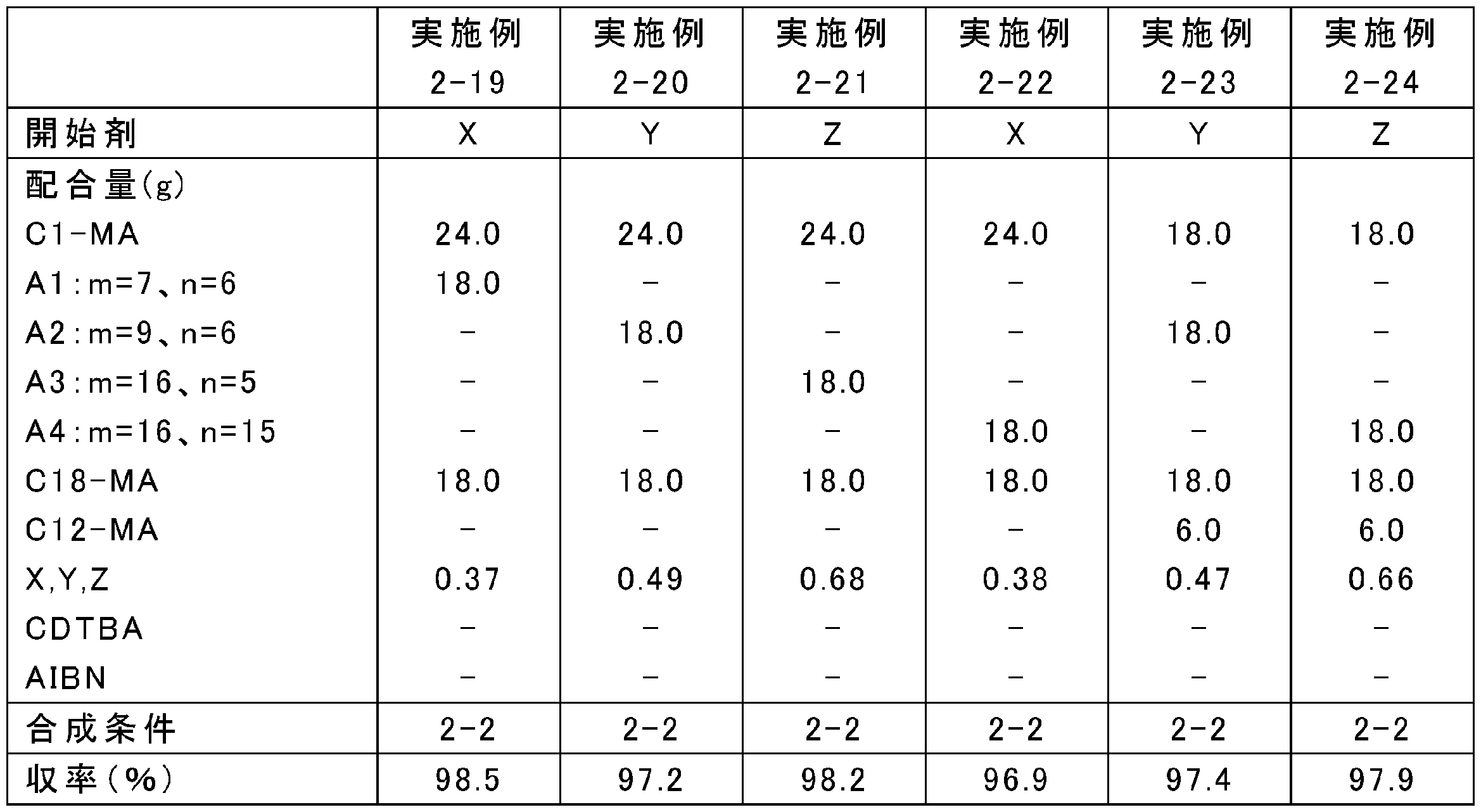
原料の配合量を表1、3、5、7、9、11に示すとおりに変更し、それ以外は上記の合成条件1-1~1-4のいずれかと同様にして、ポリ(メタ)アクリレート系粘度指数向上剤を合成した。なお、表中、Yは4官能開始剤であるペンタエリスリトールテトラキス(2-ブロモイソブチレート)、Zは6官能開始剤であるジペンタエリスリトールヘキサキス(2-ブロモイソブチレート)を表す。また、A1:m=7、n=6等は式(5)中のR1がメチル基、R2が式(3)でm=7、n=6である化合物等を表す。得られたポリ(メタ)アクリレート系粘度指数向上剤のMw、Mn及びMw/Mnを表2、4、6、8、10、12に示す。
実施例1-1~1-24、比較例1-1~1-5及び参考例1-1~1-3でそれぞれ得られたポリ(メタ)アクリレート系粘度指数向上剤と、金属系(カルシウムスルホネート系)清浄剤、無灰分散剤(コハク酸イミド)、摩擦調整剤(グリセリンモノオレート)及び摩耗防止剤(亜鉛ジチオフォスフェート)を含む性能添加剤と、高度精製鉱油(GroupIII基油、100℃における動粘度:4.2mm2/s、VI:125)とを、表2、4、6、8、10、12に示す割合で配合し、潤滑油組成物を調製した。
実施例1-1~1-24、比較例1-1~1-5及び参考例1-1~1-3の各潤滑油組成物について、100℃における動粘度、粘度指数、100℃及び150℃におけるHTHS粘度、並びに-40℃におけるMRV粘度を、それぞれ下記に準拠した方法により測定した。結果を表2、4、6、8、10、12に示す。なお、表中、MRV粘度の項目における「Y.S.」は降伏応力を表し、規格外れを意味する。
動粘度:JIS K-2283-1993
粘度指数:JIS K 2283-1993
HTHS粘度:ASTM D-4683
MRV粘度:ASTM D-4684
下記の条件(「合成条件2-1」とする)でポリ(メタ)アクリレート系粘度指数向上剤を合成した。
下記の条件(「合成条件2-2」とする)でポリ(メタ)アクリレート系粘度指数向上剤を合成した。
下記の条件(「合成条件2-3」とする)でポリ(メタ)アクリレート系粘度指数向上剤を合成した。
下記の条件(「合成条件2-4」とする)でポリ(メタ)アクリレート系粘度指数向上剤を合成した。
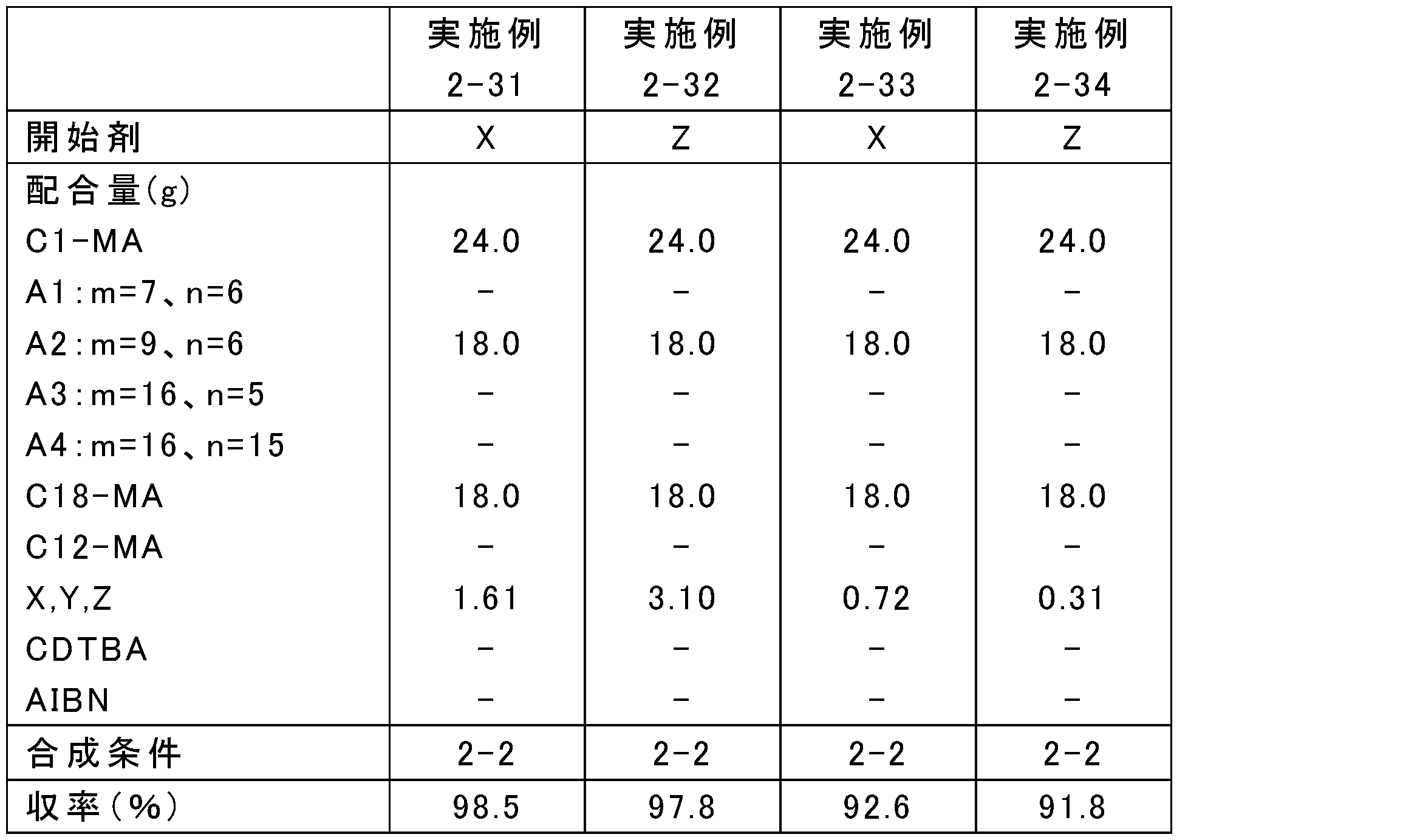
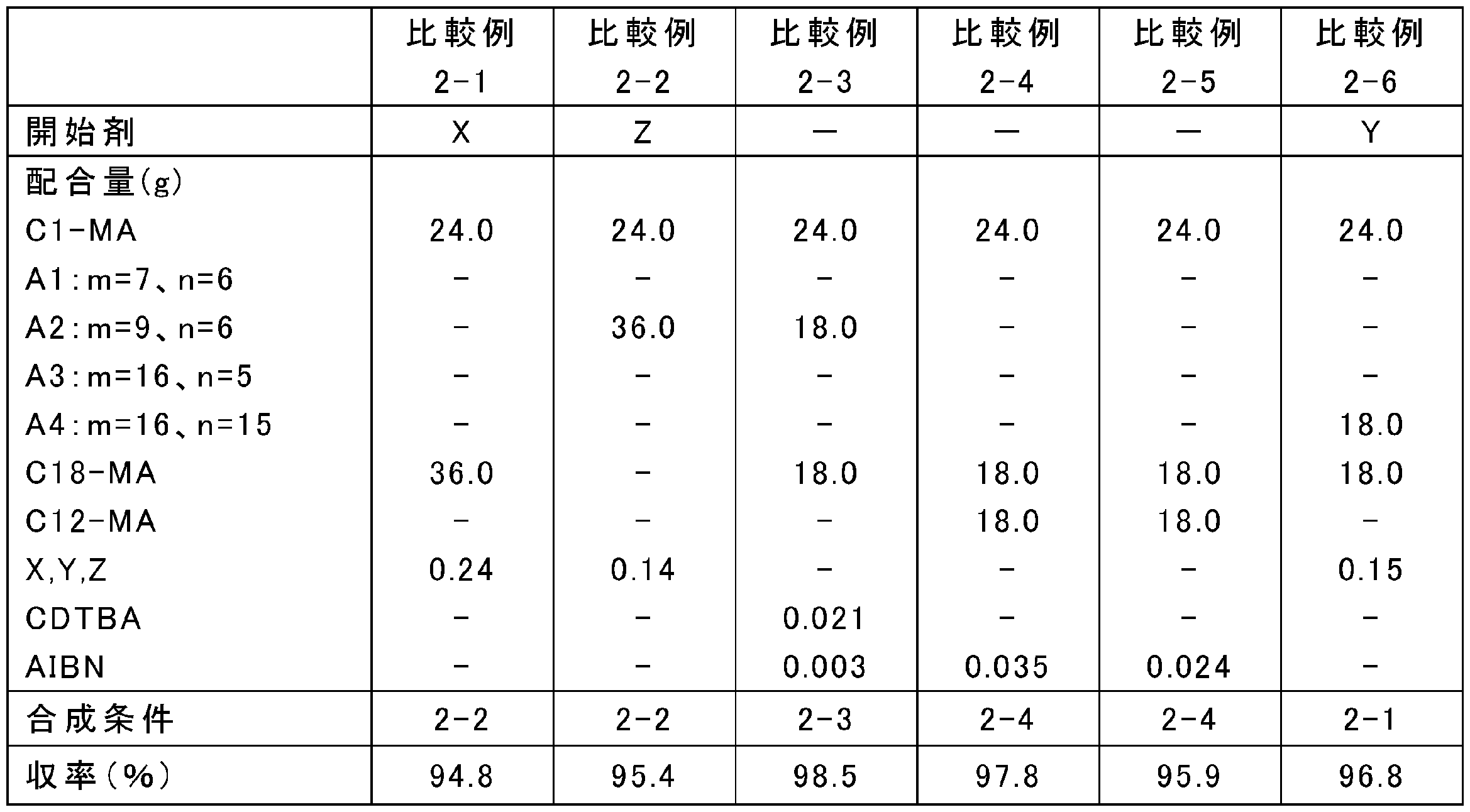
原料の配合量を表13、15、17、19、21、23、25に示すとおりに変更し、それ以外は上記の合成条件2-1~2-4のいずれかと同様にして、ポリ(メタ)アクリレート系粘度指数向上剤を合成した。なお、表中、Yは4官能開始剤であるペンタエリスリトールテトラキス(2-ブロモイソブチレート)、Zは6官能開始剤であるジペンタエリスリトールヘキサキス(2-ブロモイソブチレート)を表す。また、A1:m=7、n=6等は式(5)中のR1がメチル基、R2が式(3)でm=7、n=6である化合物等を表す。得られたポリ(メタ)アクリレート系粘度指数向上剤のMw、Mn及びMw/Mnを表14、16、18、20、22、24、26に示す。
実施例2-1~2-34及び比較例2-1~2-6でそれぞれ得られたポリ(メタ)アクリレート系粘度指数向上剤と、金属系(TBN300mgKOH/gのカルシウムスルホネート系)清浄剤、無灰分残剤(コハク酸イミド)、摩擦調整剤(オレイルアミド)、摩耗防止剤(リン酸)、酸化防止剤(ジフェニルアミン)、金属不活性化剤(チアジアゾール)、及び硫黄系添加剤(硫化エステル)を含む性能添加剤と、高度精製鉱油(GroupII基油、100℃における動粘度:3.3mm2/s、VI:110)とを、表14、16、18、20、22、24、26に示す割合で配合し、潤滑油組成物を調製した。
実施例2-1~2-34及び比較例2-1~2-6の各潤滑油組成物について、100℃における動粘度、粘度指数、及び-40℃におけるBF粘度を、それぞれ下記に準拠した方法により測定した。結果を表14、16、18、20、22、24、26に示す。
動粘度:JIS K-2283-1993
粘度指数:JIS K 2283-1993
BF粘度:JPI-5S-26-99
Claims (4)
- 請求項1又は2に記載のポリ(メタ)アクリレート系粘度指数向上剤を含有する潤滑油添加剤。
- 潤滑油基油と、請求項1又は2に記載のポリ(メタ)アクリレート系粘度指数向上剤と、を含有する潤滑油組成物。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/412,848 US20150322370A1 (en) | 2012-07-24 | 2013-07-24 | Poly(meth)acrylate-based viscosity index improver, lubricant additive and lubricant composition containing viscosity index improver |
| EP13823715.1A EP2878660A4 (en) | 2012-07-24 | 2013-07-24 | POLY (METH) ACRYLATE-BASED VISCOSITY INDEX ENHANCER, LUBRICANT ADDITION AND LUBRICANT COMPOSITION WITH THE VISCOSITY INDEX ENHANCER |
| CN201380033985.1A CN104411810A (zh) | 2012-07-24 | 2013-07-24 | 聚(甲基)丙烯酸酯系粘度指数改进剂、以及含有该粘度指数改进剂的润滑油添加剂及润滑油组合物 |
| KR20147034254A KR20150037750A (ko) | 2012-07-24 | 2013-07-24 | 폴리(메타)아크릴레이트계 점도 지수 향상제, 및 당해 점도 지수 향상제를 함유하는 윤활유 첨가제 및 윤활유 조성물 |
Applications Claiming Priority (20)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012-163619 | 2012-07-24 | ||
| JP2012-163622 | 2012-07-24 | ||
| JP2012-163624 | 2012-07-24 | ||
| JP2012163624 | 2012-07-24 | ||
| JP2012163622 | 2012-07-24 | ||
| JP2012163619 | 2012-07-24 | ||
| JP2013079832 | 2013-04-05 | ||
| JP2013079829 | 2013-04-05 | ||
| JP2013079828 | 2013-04-05 | ||
| JP2013079816 | 2013-04-05 | ||
| JP2013-079832 | 2013-04-05 | ||
| JP2013-079828 | 2013-04-05 | ||
| JP2013-079830 | 2013-04-05 | ||
| JP2013-079829 | 2013-04-05 | ||
| JP2013079830 | 2013-04-05 | ||
| JP2013-079816 | 2013-04-05 | ||
| JP2013-142019 | 2013-07-05 | ||
| JP2013142019A JP6077954B2 (ja) | 2013-07-05 | 2013-07-05 | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 |
| JP2013142035A JP6077955B2 (ja) | 2013-07-05 | 2013-07-05 | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 |
| JP2013-142035 | 2013-07-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014017553A1 true WO2014017553A1 (ja) | 2014-01-30 |
Family
ID=49997362
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/070085 WO2014017553A1 (ja) | 2012-07-24 | 2013-07-24 | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20150322370A1 (ja) |
| EP (1) | EP2878660A4 (ja) |
| KR (1) | KR20150037750A (ja) |
| CN (1) | CN104411810A (ja) |
| WO (1) | WO2014017553A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3192857A1 (en) | 2016-01-13 | 2017-07-19 | Basf Se | Use of poly(meth)acrylate copolymers with branched c17 alkyl chains in lubricant oil compositions |
| WO2017122707A1 (ja) * | 2016-01-12 | 2017-07-20 | 株式会社クラレ | (メタ)アクリレート系重合体 |
| WO2017122721A1 (ja) * | 2016-01-12 | 2017-07-20 | Jxエネルギー株式会社 | 潤滑油組成物 |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3116962B1 (en) * | 2014-03-10 | 2019-10-30 | 3M Innovative Properties Company | Conformable coating composition |
| JP6420964B2 (ja) * | 2014-03-31 | 2018-11-07 | 出光興産株式会社 | 内燃機関用潤滑油組成物 |
| WO2016043195A1 (ja) | 2014-09-17 | 2016-03-24 | 株式会社日本触媒 | 粘度指数向上剤及びその製造方法、並びに潤滑油組成物 |
| WO2018056316A1 (ja) | 2016-09-21 | 2018-03-29 | 株式会社日本触媒 | 粘度指数向上剤および潤滑油組成物 |
| CN112694936B (zh) * | 2019-10-23 | 2022-11-15 | 中国石油化工股份有限公司 | 一种柴油机油组合物及其制备方法 |
| CN113249158B (zh) * | 2020-02-13 | 2022-09-27 | 中国石油化工股份有限公司 | 降凝剂及其制备方法、用途 |
| CN113493717B (zh) * | 2020-03-18 | 2023-01-13 | 中国石油化工股份有限公司 | 汽油机油组合物及其制备方法 |
| CN113493714B (zh) * | 2020-03-18 | 2023-01-13 | 中国石油化工股份有限公司 | 甲醇发动机润滑油组合物及其制备方法 |
Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH03100099A (ja) | 1989-09-09 | 1991-04-25 | Roehm Gmbh | 分散作用を有する粘度指数改善剤及びその製法 |
| JPH06145258A (ja) | 1992-03-20 | 1994-05-24 | Rohm & Haas Co | 無灰分散剤ポリメタクリレート重合体 |
| JPH0748421A (ja) | 1993-04-30 | 1995-02-21 | Rohm & Haas Co | 潤滑油用の流動点降下剤として有用なコポリマー |
| JPH0762372A (ja) | 1993-07-23 | 1995-03-07 | Rohm & Haas Co | 作動液用の粘度指数改良剤として有用なコポリマーを製造する方法 |
| JP2000063439A (ja) * | 1998-08-10 | 2000-02-29 | Ethyl Corp | 優れた低温特性を有する(メタ)アクリレ―トコポリマ― |
| JP2002302687A (ja) | 2001-04-06 | 2002-10-18 | Sanyo Chem Ind Ltd | 粘度指数向上剤および潤滑油組成物 |
| JP2004124080A (ja) | 2002-08-02 | 2004-04-22 | Sanyo Chem Ind Ltd | 粘度指数向上剤及び潤滑油組成物 |
| WO2005037967A1 (ja) | 2003-10-16 | 2005-04-28 | Nippon Oil Corporation | 潤滑油添加剤及び潤滑油組成物 |
| JP2005187736A (ja) | 2003-12-26 | 2005-07-14 | Sanyo Chem Ind Ltd | 潤滑油組成物 |
| JP2008518052A (ja) * | 2004-10-25 | 2008-05-29 | ザ ルブリゾル コーポレイション | 星形重合体およびそれらの組成物 |
| JP2009506179A (ja) * | 2005-08-31 | 2009-02-12 | エボニック ローマックス アディティヴス ゲゼルシャフト ミット ベシュレンクテル ハフツング | 油溶性ポリマー |
| JP2009534521A (ja) * | 2006-04-24 | 2009-09-24 | ザ ルブリゾル コーポレイション | 星形ポリマー潤滑組成物 |
| JP2009286831A (ja) | 2008-05-27 | 2009-12-10 | Nippon Oil Corp | 金属ベルト式無段変速機用潤滑油組成物 |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007127615A2 (en) * | 2006-04-24 | 2007-11-08 | The Lubrizol Corporation | Star polymer lubricating composition |
| EP2021387B1 (en) * | 2006-05-08 | 2018-02-21 | The Lubrizol Corporation | Novel polymers and methods of controlling viscosity |
-
2013
- 2013-07-24 EP EP13823715.1A patent/EP2878660A4/en not_active Withdrawn
- 2013-07-24 CN CN201380033985.1A patent/CN104411810A/zh active Pending
- 2013-07-24 US US14/412,848 patent/US20150322370A1/en not_active Abandoned
- 2013-07-24 KR KR20147034254A patent/KR20150037750A/ko not_active Application Discontinuation
- 2013-07-24 WO PCT/JP2013/070085 patent/WO2014017553A1/ja active Application Filing
Patent Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH03100099A (ja) | 1989-09-09 | 1991-04-25 | Roehm Gmbh | 分散作用を有する粘度指数改善剤及びその製法 |
| JPH06145258A (ja) | 1992-03-20 | 1994-05-24 | Rohm & Haas Co | 無灰分散剤ポリメタクリレート重合体 |
| JPH0748421A (ja) | 1993-04-30 | 1995-02-21 | Rohm & Haas Co | 潤滑油用の流動点降下剤として有用なコポリマー |
| JPH0762372A (ja) | 1993-07-23 | 1995-03-07 | Rohm & Haas Co | 作動液用の粘度指数改良剤として有用なコポリマーを製造する方法 |
| JP2000063439A (ja) * | 1998-08-10 | 2000-02-29 | Ethyl Corp | 優れた低温特性を有する(メタ)アクリレ―トコポリマ― |
| JP2002302687A (ja) | 2001-04-06 | 2002-10-18 | Sanyo Chem Ind Ltd | 粘度指数向上剤および潤滑油組成物 |
| JP2004124080A (ja) | 2002-08-02 | 2004-04-22 | Sanyo Chem Ind Ltd | 粘度指数向上剤及び潤滑油組成物 |
| WO2005037967A1 (ja) | 2003-10-16 | 2005-04-28 | Nippon Oil Corporation | 潤滑油添加剤及び潤滑油組成物 |
| JP2005187736A (ja) | 2003-12-26 | 2005-07-14 | Sanyo Chem Ind Ltd | 潤滑油組成物 |
| JP2008518052A (ja) * | 2004-10-25 | 2008-05-29 | ザ ルブリゾル コーポレイション | 星形重合体およびそれらの組成物 |
| JP2009506179A (ja) * | 2005-08-31 | 2009-02-12 | エボニック ローマックス アディティヴス ゲゼルシャフト ミット ベシュレンクテル ハフツング | 油溶性ポリマー |
| JP2009534521A (ja) * | 2006-04-24 | 2009-09-24 | ザ ルブリゾル コーポレイション | 星形ポリマー潤滑組成物 |
| JP2009286831A (ja) | 2008-05-27 | 2009-12-10 | Nippon Oil Corp | 金属ベルト式無段変速機用潤滑油組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2878660A4 * |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017122707A1 (ja) * | 2016-01-12 | 2017-07-20 | 株式会社クラレ | (メタ)アクリレート系重合体 |
| WO2017122721A1 (ja) * | 2016-01-12 | 2017-07-20 | Jxエネルギー株式会社 | 潤滑油組成物 |
| US10759888B2 (en) | 2016-01-12 | 2020-09-01 | Kuraray Co., Ltd. | (Meth) acrylate polymers |
| EP3192857A1 (en) | 2016-01-13 | 2017-07-19 | Basf Se | Use of poly(meth)acrylate copolymers with branched c17 alkyl chains in lubricant oil compositions |
| RU2729517C2 (ru) * | 2016-01-13 | 2020-08-07 | Басф Се | Поли(мет)акрилатные сополимеры с разветвленными с17 алкильными цепями и их применение в композициях смазочного масла |
| US10975325B2 (en) | 2016-01-13 | 2021-04-13 | Basf Se | Poly(meth)acrylate copolymers with branched C17 alkyl chains and their use in lubricant oil compositions |
Also Published As
| Publication number | Publication date |
|---|---|
| US20150322370A1 (en) | 2015-11-12 |
| EP2878660A4 (en) | 2015-08-26 |
| KR20150037750A (ko) | 2015-04-08 |
| EP2878660A1 (en) | 2015-06-03 |
| CN104411810A (zh) | 2015-03-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2014017553A1 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| EP2474601B1 (en) | Lubricant oil composition | |
| WO2014017558A1 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| WO2014017554A1 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| US10815445B2 (en) | Lubricating oil composition, lubricating method, and transmission | |
| WO2012128350A1 (ja) | 粘度指数向上剤、潤滑油用添加剤および潤滑油組成物 | |
| JP2007284564A (ja) | 自動変速機用潤滑油組成物 | |
| US9783757B2 (en) | Poly(meth)acrylate-based viscosity index improver, lubricant additive and lubricant composition containing viscosity index improver | |
| JP6043245B2 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| JP6702611B2 (ja) | 潤滑油組成物、潤滑方法、及び変速機 | |
| JP6077954B2 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| JP6077955B2 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| JP6088924B2 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| US10844311B2 (en) | Lubricating oil composition, lubricating method, and transmission | |
| JP6077956B2 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| JP6018982B2 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| JP2018184518A (ja) | 内燃機関用潤滑油組成物 | |
| JP6113004B2 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| JP6018981B2 (ja) | ポリ(メタ)アクリレート系粘度指数向上剤、並びに該粘度指数向上剤を含有する潤滑油添加剤及び潤滑油組成物 | |
| JP2000144166A (ja) | 内燃機関用潤滑油組成物 | |
| JP2018188549A (ja) | 潤滑油組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13823715 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013823715 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20147034254 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14412848 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |