WO2012111762A1 - 抗cd40抗体の高濃度製剤 - Google Patents
抗cd40抗体の高濃度製剤 Download PDFInfo
- Publication number
- WO2012111762A1 WO2012111762A1 PCT/JP2012/053687 JP2012053687W WO2012111762A1 WO 2012111762 A1 WO2012111762 A1 WO 2012111762A1 JP 2012053687 W JP2012053687 W JP 2012053687W WO 2012111762 A1 WO2012111762 A1 WO 2012111762A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- antibody
- solution preparation
- ser
- turbidity
- concentration
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39591—Stabilisation, fragmentation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/16—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing nitrogen, e.g. nitro-, nitroso-, azo-compounds, nitriles, cyanates
- A61K47/18—Amines; Amides; Ureas; Quaternary ammonium compounds; Amino acids; Oligopeptides having up to five amino acids
- A61K47/183—Amino acids, e.g. glycine, EDTA or aspartame
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2878—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the NGF-receptor/TNF-receptor superfamily, e.g. CD27, CD30, CD40, CD95
Definitions
- the present invention relates to a solution preparation containing a high concentration of antagonistic anti-CD40 antibody.
- CD40 is an antigen expressed on the surface of B cells, DCs, macrophages, endothelial cells and epithelial cells and their tumor cells.
- Anti-CD40 antibodies that act on CD40 are agonistic (also referred to as “agonistic antibodies”). ) And antagonistic (also referred to as “antagonistic antibodies”).
- agonistic antibodies also referred to as “agonistic antibodies”.
- antagonistic antibodies also referred to as “antagonistic antibodies”.
- activation of B cells is known. Examples of such an antibody include the agonistic anti-CD40 antibody described in Patent Document 1.
- CD40 plays an important role in the immune response
- inhibition of binding of CD40 and its ligand by an antagonistic antibody can develop immunosuppressive drugs during organ transplantation and therapeutics for autoimmune diseases. It is expected.
- an anti-CD40 antibody it is required that CD40L binds to human CD40 and that the antibody itself does not activate CD40.
- Patent Document 1 discloses a modified antibody of 4D11 (hereinafter also referred to as “4D11G4PE”). This modified antibody is used both in vitro and in vivo as compared with the original 4D11 antibody. In terms of avoiding the expression of agonist activity, it is expected as an anti-antigonal CD40 antibody that is particularly useful in immunosuppression during organ transplantation and in the treatment of autoimmune diseases (Patent Document 3).
- a freeze-dried preparation can be mentioned as described in Patent Document 4.
- lyophilized preparations at medical sites are cumbersome to prepare for administration, which is a burden for medical staff and there is a concern about the risk of bacterial contamination due to the procedure.
- solution preparations are simpler than pre-administration and thus reduce the risk of bacterial contamination, which is considered more desirable than lyophilized preparations in medical settings.
- an antibody when used for medical purposes, it is generally administered by intravenous or subcutaneous route.
- subcutaneous administration such as injections may be used for reasons such as ease of administration, but subcutaneous administration depends on the volume limit (usually 1.5 ml or less) and the required dosage (usually 50 mg or more). Therefore, it is often necessary to administer antibodies at high concentrations.
- the antibody is administered to a patient at 2 mg / kg weekly, assuming that the average body weight of the patient is 70 kg, the antibody dose is 140 mg and the antibody concentration for subcutaneous administration is approximately 100 mg / ml. (In this case 1.4 ml will contain about 140 mg of antibody).
- Non-Patent Document 1 shows that a high-concentration protein preparation increases in viscosity and increases in turbidity, and it is proposed that ionic strength affects the degree.
- Non-Patent Document 2 presents a method for stabilizing an antibody at a high concentration using spray-dry.
- Non-Patent Document 1 does not propose a radical solution for suppressing the viscosity and turbidity of a high concentration protein preparation. Also, in spray-dry described in Non-Patent Document 2, since preparation for administration is complicated as in the case of freeze-dried preparations, it is a burden for medical staff and there is a concern about the risk of contamination by bacteria due to the procedure.
- a solution preparation of anti-CD40 antibody a solution preparation of 4D11 antibody described in Patent Document 5 is known. Although the preparation has been confirmed to be stable in various tests, the antibody concentration is 10 mg / ml. Therefore, it is difficult to use as a solution preparation for subcutaneous administration.
- solution preparations containing high concentrations of antibodies provide superior improvements in clinical applications such as improved administration methods (eg, subcutaneous administration), extended administration intervals, and greatly contribute to improved therapeutic efficacy and patient compliance. It is expected.
- improved administration methods eg, subcutaneous administration
- extended administration intervals e.g., extended administration intervals
- problems such as turbidity and generation of insoluble foreign matter, but a general improvement method has not yet been found.
- an object of the present invention is to provide a high-concentration solution formulation of 4D11G4PE in which the occurrence of turbidity or insoluble foreign matter due to antibodies is suppressed to the same level as that of conventional low-concentration formulations. .
- the present inventors In the solution preparation containing a high concentration of anti-antigonal CD40 antibody, the present inventors have a specific concentration of a surfactant, and when the pH is in a specific range, the occurrence of turbidity caused by the antibody or insolubility The inventors have found that the generation of foreign substances can be suppressed to the same level as that of conventional low-concentration preparations, and have completed the present invention.
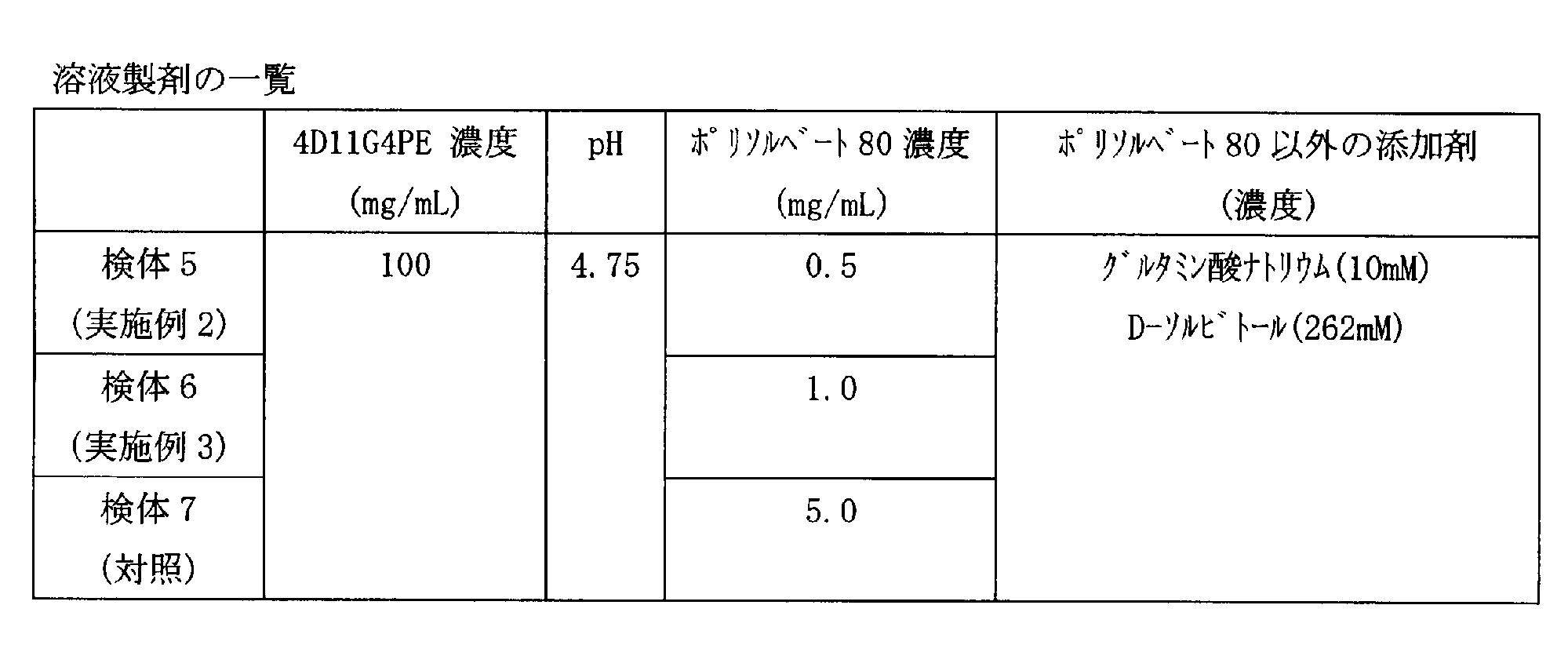
- the present invention includes the following [1] to [10] inventions as medically useful antibody pharmaceutical preparations: [1] pH comprising glutamic acid or a pharmaceutically acceptable salt thereof, about 0.5 mg / mL to about 2.0 mg / mL polysorbate 80, and about 50 mg / mL to about 200 mg / mL antibody
- the antibody is a heavy chain consisting of the amino acid sequence represented by the 27th Q to the 474th K in SEQ ID NO: 1, and SEQ ID NO: 2
- a solution preparation which is an antibody comprising a light chain consisting of the amino acid sequence represented by the 23rd A to the 235th C.
- [2] The solution formulation according to [1], wherein the antibody concentration is about 100 mg / mL or more and about 120 mg / mL or less.
- [3] The solution preparation according to [1] or [2], wherein glutamic acid or a pharmaceutically acceptable salt thereof is sodium glutamate.
- [4] The solution formulation according to any one of [1] to [3], wherein the concentration of polysorbate 80 is about 1.0 mg / mL.
- [5] The solution preparation according to any one of [1] to [4], wherein the antibody concentration is about 100 mg / mL.
- [6] The solution preparation according to any one of [1] to [5], which has a pH of about 4.75 to about 4.8.
- the solution preparation of the present invention can suppress the occurrence of turbidity or insoluble foreign matter due to the increase in concentration of the 4D11G4PE antibody to the same level as the low concentration solution preparation. Specifically, even when the solution preparation of the present invention is stored at 25 ° C. for 1 month or 3 months, the occurrence of turbidity or insoluble foreign matter due to the antibody during the storage period is less than that of the low concentration solution preparation. It can be suppressed to the same level.
- the “solution formulation” in the present invention is a formulation in which an antibody having a therapeutic effect is dissolved in a pharmaceutically acceptable solution.
- the solution include an aqueous solution and a non-aqueous solution.
- An aqueous solution is preferable, and water for injection is more preferable.
- the “antibody” used in the solution preparation of the present invention is an antagonistic anti-CD40 antibody, a heavy chain consisting of the amino acid sequence represented by the 27th Q to the 474th K in SEQ ID NO: 1, and SEQ ID NO: 2 is an antibody (also referred to as “4D11G4PE”) comprising a light chain consisting of the amino acid sequence represented by the 23rd A to the 235th C.
- SEQ ID NO: 1 and SEQ ID NO: 2 are shown below.
- the antibody 4D11G4PE used in the solution preparation of the present invention can be easily prepared by those skilled in the art using methods known in the art based on the amino acid sequences of the heavy and light chains.
- An exemplary method for producing the antibody is disclosed in Patent Document 3.
- the concentration of the antibody contained in the solution preparation of the present invention is about 50 mg / mL to about 200 mg / mL, preferably about 80 mg / mL to about 150 mg / mL, preferably about 100 to about 120 mg / mL.
- the solution preparation of the present invention contains glutamic acid or a pharmaceutically acceptable salt thereof as a “buffer”.
- glutamic acid or a pharmaceutically acceptable salt thereof used include glutamic acid, sodium glutamate, potassium glutamate, lysine glutamate, arginine glutamate, and glutamic acid hydrochloride. Preferred is glutamic acid or sodium glutamate.
- the buffer may be used alone or in combination of two or more.
- the concentration of glutamic acid used or a pharmaceutically acceptable salt thereof can be adjusted depending on its buffering capacity and the like, for example, about 1 mM to about 50 mM, preferably about 10 mM.
- the pH of the solution preparation of the present invention is adjusted to about 4.6 to about 5.0, preferably about 4.75 to about 4.8.
- the solution preparation of the present invention contains polysorbate 80 (polyoxyethylene sorbitan monooleate) as a “surfactant”.
- concentration of polysorbate 80 in the solution preparation of the present invention is about 0.5 mg / mL or more and about 2.0 mg / mL or less, preferably about 1.0 mg / mL.
- the solution preparation of the present invention may contain one or more pharmaceutically acceptable isotonic agents, stabilizers, preservatives, suspending agents, other additives such as emulsifiers, or combinations thereof.
- an “isotonic agent” may be used to adjust the osmotic pressure of the solution formulation of the present invention to have essentially the same osmotic pressure as human blood.
- the isotonic agent used is not particularly limited as long as the solution preparation has essentially the same osmotic pressure as human blood, and examples thereof include salts and sugars.
- Salt used as an isotonic agent is a compound produced in such a way that a cation and an anion neutralize the charge.
- the salt include sodium chloride, potassium chloride, calcium chloride, magnesium chloride, sodium bisulfite, calcium bromide and sodium bromide.
- “Sugar” used as an isotonic agent is a carbohydrate represented by Cn (H 2 O) m and derivatives thereof (for example, polyhydric alcohols obtained by reducing the carbonyl group of sugar, Aldoses and ketoses formed from polyhydric alcohols), and polymers purified from the carbohydrates and / or the derivatives.
- sugar examples include mannitol, inositol, glucose, sorbitol, fructose, lactose, xylose, mannose, maltose, raffinose, sucrose, and trehalose.
- the isotonic agent used as the solution preparation of the present invention is preferably a sugar, and more preferably a non-reducing sugar.
- the “reducing sugar” is a sugar having an aldehyde group or a ketone group in an aqueous solution and acting as a reducing agent, and the non-reducing sugar is a sugar that is not a reducing sugar.
- non-reducing sugars examples include sorbitol, mannitol, inositol, sucrose, and trehalose. Among these, sorbitol is preferable and D-sorbitol is more preferable.
- the concentration of the isotonic agent in the solution preparation of the present invention is not particularly limited as long as the solution preparation of the present invention is isotonic.
- the solution preparation contains other substances that have the ability to be isotonic, such as buffering agents and antioxidants, and the concentration of the isotonic agent in the solution preparation is combined with these substances. Is preferably adjusted so as to have 200 to 400 mOsm.
- the solution preparation of the present invention is typically stored in an aqueous solution state and preferably administered as it is.
- the solution preparation of the present invention may be a solution before preparing a lyophilized preparation, or a solution after dissolving and reconstituting the lyophilized preparation.
- the solution preparation of the present invention may be a solution (raw drug substance) that is a raw material for producing the solution preparation.
- the “turbidity” in the solution preparation of the present invention refers to fine particles having a size that cannot be visually identified as particles. Since the light transmittance is reduced because the light is scattered in the solution due to turbidity, the degree of turbidity can be evaluated by visual inspection.
- Turbidity rank “-” When the inspector observes at a brightness of 5000 lux, no turbidity is observed.
- Turbidity rank “ ⁇ ” When the inspector observes at a brightness of 5000 lux, a slight turbidity is recognized.
- Turbidity rank “+” Slight turbidity is observed when the inspector observes at a brightness of 5000 lux.
- Turbidity rank “2+” Turbidity is easily recognized when an inspector observes at a brightness of 5000 lux.
- Turbidity rank “3+” When an inspector observes at a brightness of 5000 lux, turbidity is easily recognized, and when observed at a brightness of 200 lux (under indoor diffused light), turbidity is recognized.
- Turbidity is caused by stress such as heat, light and vibration, or by the presence of other substances that destabilize antibody molecules.
- insoluble foreign matter refers to fine particles having a size that can be discriminated visually as particles.
- the degree of generation of insoluble foreign matter can be evaluated by confirming the size and / or number of fine particles by visual inspection.
- insoluble foreign substances are evaluated based on the following criteria.
- Insoluble foreign matter rank "-" When the inspector observes at a brightness of 5000 lux, no foreign matter is recognized.
- Insoluble foreign matter rank “ ⁇ ” When an inspector observes at a brightness of 5000 lux, a small number of suspended matters that are discriminated as minute foreign matters are faintly recognized.
- Insoluble foreign matter rank “+” When an inspector observes at a brightness of 5000 lux, a slight foreign matter is recognized.
- Insoluble foreign matter rank “++++” A foreign matter that can be easily detected is observed when the inspector observes at a brightness of 1000 lux.
- Insoluble foreign matter may have a high particle size and / or number of particles due to stress such as heat, light and vibration, or the presence of other substances that destabilize antibody molecules.
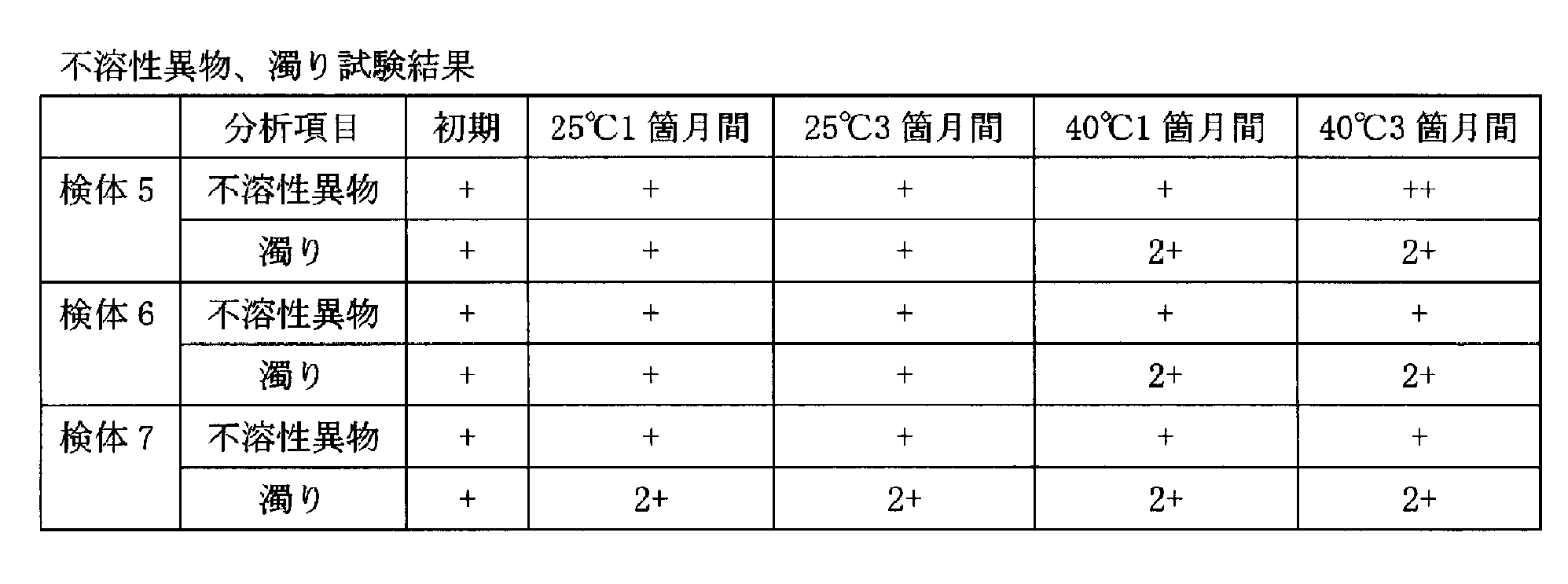
- both the rank of insoluble foreign matter and the turbidity rank in the solution preparation after storage for 1 month or 3 months at 25 ° C. are + or less.
- the solution preparation of the present invention preferably has an insoluble foreign substance rank and a turbidity rank both lower than + in a solution preparation after storage at 25 ° C. for 1 month, and insoluble in a solution preparation after storage at 25 ° C. for 3 months.
- the rank of foreign matter and the turbidity rank are both + or less, more preferably, the rank of insoluble foreign matter in the solution preparation after storage for 1 month at 25 ° C. is ⁇ , the rank of turbidity is + or less, and 25 ° C.
- the rank of insoluble foreign matter in the solution preparation after storage for 3 months is ⁇ or less, and the turbidity rank is + or less.
- Sample 1 is a low concentration (10 mg / mL) solution formulation of 4D11G4PE for intravenous administration.
- the additives other than the antibody and the concentration thereof in the specimen 1 are the same as those in the low concentration preparation of 4D11 described in Example 5 of Patent Document 5.
- Sample 1 was used as a comparison target of the high concentration preparation.
- 4D11G4PE was produced according to the description in Patent Document 3.
- a placebo solution not containing 4D11G4PE is prepared in advance, and the solution containing 4D11G4PE is replaced with the placebo solution by an ultrafiltration membrane (Millipore), and then 4D11G4PE is concentrated using the same ultrafiltration membrane. It was prepared by.
- Each sample was aseptically filtered using a 0.22 ⁇ m filter (Millipore) in a clean bench, and filled in a 5 mL glass vial (compatible with the Japanese Pharmacopoeia) 1 mL at a time in the clean bench.
- Thermal stability test Stored in an incubator (TABAI ESPEC) controlled at 25 ° C. for 1 month or 3 months.
- each specimen was stored in a low-temperature storage controlled at 5 ° C. until the start of analysis after each stress load.
- Thermal stability test Stored in an incubator (TABAI ESPEC) controlled at 25 ° C. or 40 ° C. for 1 month or 3 months.
- each specimen was stored in a low-temperature storage controlled at 5 ° C. until the start of analysis after each stress load.
- Thermal stability test It was stored for 1 month in an incubator (TABAI ESPEC) controlled at 25 ° C.
- each specimen was stored in a low-temperature storage controlled at 5 ° C. until the start of analysis after each stress load.
Abstract
Description
[1]グルタミン酸またはその製薬学的に許容される塩、約0.5mg/mL以上約2.0mg/mL以下のポリソルベート80、および約50mg/mL以上約200mg/mL以下の抗体を含む、pHが約4.6~約5.0である溶液製剤であって、該抗体が、配列番号1における27番目のQから474番目のKで表されるアミノ酸配列からなる重鎖、および配列番号2における23番目のAから235番目のCで表されるアミノ酸配列からなる軽鎖を含む抗体である、溶液製剤。
[2]抗体濃度が約100mg/mL以上約120mg/mL以下である、[1]に記載の溶液製剤。
[3]グルタミン酸またはその製薬学的に許容される塩がグルタミン酸ナトリウムである、[1]または[2]に記載の溶液製剤。
[4]ポリソルベート80の濃度が約1.0mg/mLである、[1]~[3]のいずれか1に記載の溶液製剤。
[5]抗体濃度が約100mg/mLである、[1]~[4]のいずれか1に記載の溶液製剤。
[6]pHが約4.75~約4.8である、[1]~[5]のいずれか1に記載の溶液製剤。
[7]更に等張化剤を含む、[1]~[6]のいずれか1に記載の溶液製剤。
[8]等張化剤が塩または糖である、[7]に記載の溶液製剤。
[9]等張化剤が非還元糖である、[8]に記載の溶液製剤。
[10]非還元糖がソルビトールである、[9]に記載の溶液製剤。
本発明の溶液製剤における「濁り」は、目視によって粒子であると判別することができない大きさの微粒子をいう。濁りにより溶液中で光が散乱されるために光の透過率が減少するため、濁りの程度を目視検査によって評価することが可能である。
濁りランク「-」:検査員が5000ルクスの明るさで観察したとき、濁りを認めない。
濁りランク「±」:検査員が5000ルクスの明るさで観察したとき、ごく僅かに濁りを認める。
濁りランク「+」:検査員が5000ルクスの明るさで観察したとき、僅かに濁りを認める。
濁りランク「2+」:検査員が5000ルクスの明るさで観察したとき、容易に濁りを認める。
濁りランク「3+」:検査員が5000ルクスの明るさで観察したとき、容易に濁りを認め、かつ200ルクスの明るさ(室内散光下)で観察したとき、濁りを認める。
本発明の溶液製剤における「不溶性異物」は、目視によって粒子であると判別することができる大きさの微粒子をいう。不溶性異物の生成は、微粒子の大きさおよび/または数を目視検査によって確認することで、その程度を評価することが可能である。
不溶性異物ランク「-」:検査員が5000ルクスの明るさで観察したとき、異物を認めない。
不溶性異物ランク「±」:検査員が5000ルクスの明るさで観察したとき、微小な異物と判別される少数の浮遊物をかすかに認める。
不溶性異物ランク「+」:検査員が5000ルクスの明るさで観察したとき、微小な異物と判別されるものをわずかに認める。
不溶性異物ランク「++」:検査員が5000ルクスの明るさで観察したとき、容易に検出できる異物を認めるが、1000ルクスの明るさではたやすく検出できる異物を認めない。
不溶性異物ランク「+++」:検査員が1000ルクスの明るさで観察したとき、たやすく検出できる異物を認める。
高濃度の抗体(4D11G4PE)を含有する溶液製剤において、不溶性異物および濁りに対してpHが与える影響を評価するために、検体1~4(検体1~3「対照」、検体4「実施例1」)を作製した(表1)。
4D11G4PEは特許文献3の記載に従って製造した。各検体について、あらかじめ4D11G4PEを含まないプラセボ溶液を作製し、4D11G4PEを含む溶液を限外ろ過膜(ミリポア社製)により該プラセボ溶液と置換した後、同限外ろ過膜を用いて4D11G4PEを濃縮することにより調製した。また、各検体における4D11G4PEの濃度を、OD280nm吸光度係数ε=1.4を用いて換算し、調整した。
本実験例において不溶性異物および濁りの評価を行うため、以下の条件に従い各検体にストレスを与えた。
不溶性異物および濁りは、先述の異物ランクおよび濁りランクに従ってそれぞれ分析を行った。
結果を表2に示す。
高濃度の抗体(4D11G4PE)を含有する溶液製剤において、不溶性異物および濁りに対して界面活性剤の濃度が与える影響を評価するために、検体5~7(実施例2及び3、対照)を作製した(表3)。
実験例1と同様の材料および方法を用いた。
本実験例において安定性評価を行うため、以下の条件に従い各製剤検体にストレスを与えた。
実験例1と同様の分析を行った。
結果を表4に示す。
高濃度の抗体(4D11G4PE)を含有する溶液製剤において、不溶性異物および濁りに対して抗体濃度が与える影響を評価するために、検体8(実施例4)を作製した(表5)。
実験例1と同様の材料および方法を用いた。
本実験例において安定性評価を行うため、以下の条件に従い各製剤検体にストレスを与えた。
実験例1と同様の分析を行った。
結果を表6に示す。
Claims (10)
- グルタミン酸またはその製薬学的に許容される塩、約0.5mg/mL以上約2.0mg/mL以下のポリソルベート80、および約50mg/mL以上約200mg/mL以下の抗体を含む、pHが約4.6~約5.0である溶液製剤であって、該抗体が、配列番号1における27番目のQから474番目のKで表されるアミノ酸配列からなる重鎖、および配列番号2における23番目のAから235番目のCで表されるアミノ酸配列からなる軽鎖を含む抗体である、溶液製剤。
- 抗体濃度が約100mg/mL以上約120mg/mL以下である、請求項1に記載の溶液製剤。
- グルタミン酸またはその製薬学的に許容される塩がグルタミン酸ナトリウムである、請求項1に記載の溶液製剤。
- ポリソルベート80の濃度が約1.0mg/mLである、請求項1に記載の溶液製剤。
- pHが約4.75~約4.8である、請求項1に記載の溶液製剤。
- pHが約4.75~約4.8である、請求項1~5のいずれか1項に記載の溶液製剤。
- 更に等張化剤を含む、請求項1~6のいずれか1項に記載の溶液製剤。
- 等張化剤が塩または糖である、請求項7に記載の溶液製剤。
- 等張化剤が非還元糖である、請求項8に記載の溶液製剤。
- 非還元糖がソルビトールである、請求項9に記載の溶液製剤。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ES12747045T ES2734076T3 (es) | 2011-02-17 | 2012-02-16 | Preparación farmacéutica de anticuerpo anti-CD40 muy concentrada |
| US14/000,103 US9125893B2 (en) | 2011-02-17 | 2012-02-16 | Highly concentrated anti-CD40 antibody pharmaceutical preparation |
| EP12747045.8A EP2676677B1 (en) | 2011-02-17 | 2012-02-16 | Highly concentrated anti-cd40 antibody pharmaceutical preparation |
| JP2012558014A JP5458188B2 (ja) | 2011-02-17 | 2012-02-16 | 抗cd40抗体の高濃度製剤 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011-031894 | 2011-02-17 | ||
| JP2011031894 | 2011-02-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012111762A1 true WO2012111762A1 (ja) | 2012-08-23 |
Family
ID=46672674
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/053687 WO2012111762A1 (ja) | 2011-02-17 | 2012-02-16 | 抗cd40抗体の高濃度製剤 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US9125893B2 (ja) |
| EP (1) | EP2676677B1 (ja) |
| JP (1) | JP5458188B2 (ja) |
| ES (1) | ES2734076T3 (ja) |
| WO (1) | WO2012111762A1 (ja) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016539947A (ja) * | 2013-11-29 | 2016-12-22 | アレス トレーディング ソシエテ アノニム | TNFR及びFc領域を含む融合タンパク質の液体製剤 |
| WO2017220989A1 (en) | 2016-06-20 | 2017-12-28 | Kymab Limited | Anti-pd-l1 and il-2 cytokines |
| US9974855B2 (en) | 2015-09-04 | 2018-05-22 | Primatope Therapeutics Inc. | Humanized anti-CD40 antibodies and methods of administering thereof |
| US9987356B2 (en) | 2011-03-11 | 2018-06-05 | Beth Israel Deaconess Medical Center, Inc. | Anti-CD40 antibodies and methods of administering thereof |
| WO2019241730A2 (en) | 2018-06-15 | 2019-12-19 | Flagship Pioneering Innovations V, Inc. | Increasing immune activity through modulation of postcellular signaling factors |
| WO2020227159A2 (en) | 2019-05-03 | 2020-11-12 | Flagship Pioneering Innovations V, Inc. | Methods of modulating immune activity |
| WO2021127217A1 (en) | 2019-12-17 | 2021-06-24 | Flagship Pioneering Innovations V, Inc. | Combination anti-cancer therapies with inducers of iron-dependent cellular disassembly |
| US11090367B2 (en) | 2016-11-09 | 2021-08-17 | The Brigham And Women's Hospital, Inc. | Restoration of tumor suppression using mRNA-based delivery system |
| US11142800B2 (en) | 2010-10-07 | 2021-10-12 | The General Hospital Corporation | Biomarkers of cancer |
| WO2022006179A1 (en) | 2020-06-29 | 2022-01-06 | Flagship Pioneering Innovations V, Inc. | Viruses engineered to promote thanotransmission and their use in treating cancer |
| EP4317972A2 (en) | 2018-02-06 | 2024-02-07 | The General Hospital Corporation | Repeat rna as biomarkers of tumor immune response |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SI1707627T1 (sl) * | 2003-12-25 | 2013-02-28 | Kyowa Hakko Kirin Co., Ltd. | Antagonistiäśen mutant anti-cd40 protitelesa |
| EP3229586A4 (en) | 2014-12-10 | 2018-10-24 | Regents of the University of Minnesota | Genetically modified cells, tissues, and organs for treating disease |
| CN116963774A (zh) | 2021-01-28 | 2023-10-27 | 瑞泽恩制药公司 | 用于治疗细胞因子释放综合征的组合物和方法 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1994001547A2 (en) | 1992-07-09 | 1994-01-20 | Cetus Oncology Corporation | A method for generation of antibodies to cell surface molecules |
| WO1998022136A2 (de) | 1996-11-19 | 1998-05-28 | Roche Diagnostics Gmbh | Stabile lyophilisierte pharmazeutische zubereitungen von mono- oder polyklonalen antikörpern |
| WO2002088186A1 (en) | 2001-04-27 | 2002-11-07 | Kirin Beer Kabushiki Kaisha | Anti-cd40 monoclonal antibody |
| WO2005063981A1 (ja) | 2003-12-25 | 2005-07-14 | Kirin Beer Kabushiki Kaisha | 抗cd40抗体の変異体 |
| WO2005063291A1 (ja) | 2003-12-25 | 2005-07-14 | Kirin Beer Kabushiki Kaisha | 抗体を含有する安定な水性医薬製剤 |
| JP2006511457A (ja) * | 2002-08-16 | 2006-04-06 | アボツト・バイオテクノロジー・リミテツド | Tnf−アルファ関連疾患の治療用ヒト抗体の製剤 |
| JP2010505852A (ja) * | 2006-10-06 | 2010-02-25 | アムジエン・インコーポレーテツド | 安定な抗体製剤 |
| JP2011031894A (ja) | 2009-07-30 | 2011-02-17 | Daisoo Trading Kk | パネルロック装置 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DK1682178T3 (da) * | 2003-11-04 | 2010-10-04 | Novartis Vaccines & Diagnostic | Fremgangsmåder til terapi af cancere der udtrykker CD-40-antigenet |
| WO2006096461A2 (en) * | 2005-03-08 | 2006-09-14 | Pharmacia & Upjohn Company Llc | Composition comprising an antibody against macrophage colony-stimulating factor (m-csf) and a chelating agent |
| MEP39508A (en) | 2006-04-21 | 2011-02-10 | Novartis Ag | Antagonist anti-cd40 antibody pharmaceutical compositions |
| CA2760185A1 (en) * | 2009-05-04 | 2010-11-11 | Abbott Biotechnology Ltd. | Stable high protein concentration formulations of human anti-tnf-alpha antibodies |
| AR083847A1 (es) * | 2010-11-15 | 2013-03-27 | Novartis Ag | Variantes de fc (fragmento constante) silenciosas de los anticuerpos anti-cd40 |
-
2012
- 2012-02-16 WO PCT/JP2012/053687 patent/WO2012111762A1/ja active Application Filing
- 2012-02-16 US US14/000,103 patent/US9125893B2/en not_active Expired - Fee Related
- 2012-02-16 EP EP12747045.8A patent/EP2676677B1/en active Active
- 2012-02-16 ES ES12747045T patent/ES2734076T3/es active Active
- 2012-02-16 JP JP2012558014A patent/JP5458188B2/ja not_active Expired - Fee Related
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1994001547A2 (en) | 1992-07-09 | 1994-01-20 | Cetus Oncology Corporation | A method for generation of antibodies to cell surface molecules |
| WO1998022136A2 (de) | 1996-11-19 | 1998-05-28 | Roche Diagnostics Gmbh | Stabile lyophilisierte pharmazeutische zubereitungen von mono- oder polyklonalen antikörpern |
| WO2002088186A1 (en) | 2001-04-27 | 2002-11-07 | Kirin Beer Kabushiki Kaisha | Anti-cd40 monoclonal antibody |
| JP2006511457A (ja) * | 2002-08-16 | 2006-04-06 | アボツト・バイオテクノロジー・リミテツド | Tnf−アルファ関連疾患の治療用ヒト抗体の製剤 |
| WO2005063981A1 (ja) | 2003-12-25 | 2005-07-14 | Kirin Beer Kabushiki Kaisha | 抗cd40抗体の変異体 |
| WO2005063291A1 (ja) | 2003-12-25 | 2005-07-14 | Kirin Beer Kabushiki Kaisha | 抗体を含有する安定な水性医薬製剤 |
| JP2010505852A (ja) * | 2006-10-06 | 2010-02-25 | アムジエン・インコーポレーテツド | 安定な抗体製剤 |
| JP2011031894A (ja) | 2009-07-30 | 2011-02-17 | Daisoo Trading Kk | パネルロック装置 |
Non-Patent Citations (3)
| Title |
|---|
| BHAS DANI ET AL., JOURNAL OF PHARMACEUTICAL SCIENCES, vol. 96, no. 6, 2007, pages 1504 - 1517 |
| SALINAS BA ET AL., JOURNAL OF PHARMACEUTICAL SCIENCES, vol. 99, no. 1, 2010, pages 82 - 93 |
| See also references of EP2676677A4 |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11142800B2 (en) | 2010-10-07 | 2021-10-12 | The General Hospital Corporation | Biomarkers of cancer |
| US10561728B2 (en) | 2011-03-11 | 2020-02-18 | Beth Israel Deaconess Medical Center, Inc. | Polynucleotides encoding anti-CD40 antibodies |
| US9987356B2 (en) | 2011-03-11 | 2018-06-05 | Beth Israel Deaconess Medical Center, Inc. | Anti-CD40 antibodies and methods of administering thereof |
| JP2016539947A (ja) * | 2013-11-29 | 2016-12-22 | アレス トレーディング ソシエテ アノニム | TNFR及びFc領域を含む融合タンパク質の液体製剤 |
| US9974855B2 (en) | 2015-09-04 | 2018-05-22 | Primatope Therapeutics Inc. | Humanized anti-CD40 antibodies and methods of administering thereof |
| US10772958B2 (en) | 2015-09-04 | 2020-09-15 | Primatope Therapeutics Inc. | Humanized anti-CD40 antibodies and methods of administering thereof |
| US11439706B2 (en) | 2015-09-04 | 2022-09-13 | Primatope Therapeutics Inc. | Polynucleotides encoding a humanized anti-CD40 antibody |
| WO2017220988A1 (en) | 2016-06-20 | 2017-12-28 | Kymab Limited | Multispecific antibodies for immuno-oncology |
| WO2017220990A1 (en) | 2016-06-20 | 2017-12-28 | Kymab Limited | Anti-pd-l1 antibodies |
| WO2017220989A1 (en) | 2016-06-20 | 2017-12-28 | Kymab Limited | Anti-pd-l1 and il-2 cytokines |
| US11090367B2 (en) | 2016-11-09 | 2021-08-17 | The Brigham And Women's Hospital, Inc. | Restoration of tumor suppression using mRNA-based delivery system |
| US11471515B2 (en) | 2016-11-09 | 2022-10-18 | The Brigham And Women's Hospital, Inc. | Restoration of tumor suppression using MRNA-based delivery system |
| EP4317972A2 (en) | 2018-02-06 | 2024-02-07 | The General Hospital Corporation | Repeat rna as biomarkers of tumor immune response |
| WO2019241730A2 (en) | 2018-06-15 | 2019-12-19 | Flagship Pioneering Innovations V, Inc. | Increasing immune activity through modulation of postcellular signaling factors |
| WO2020227159A2 (en) | 2019-05-03 | 2020-11-12 | Flagship Pioneering Innovations V, Inc. | Methods of modulating immune activity |
| WO2021127217A1 (en) | 2019-12-17 | 2021-06-24 | Flagship Pioneering Innovations V, Inc. | Combination anti-cancer therapies with inducers of iron-dependent cellular disassembly |
| WO2022006179A1 (en) | 2020-06-29 | 2022-01-06 | Flagship Pioneering Innovations V, Inc. | Viruses engineered to promote thanotransmission and their use in treating cancer |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2676677A9 (en) | 2014-05-07 |
| US9125893B2 (en) | 2015-09-08 |
| JP5458188B2 (ja) | 2014-04-02 |
| JPWO2012111762A1 (ja) | 2014-07-07 |
| EP2676677A1 (en) | 2013-12-25 |
| US20130323267A1 (en) | 2013-12-05 |
| EP2676677B1 (en) | 2019-05-22 |
| ES2734076T3 (es) | 2019-12-04 |
| EP2676677A4 (en) | 2015-02-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5458188B2 (ja) | 抗cd40抗体の高濃度製剤 | |
| KR102588846B1 (ko) | 고농도의 항-vegf 항체를 포함하는 안정한 단백질 용액 제제 | |
| CN106999591B (zh) | 一种抗pd-1抗体制剂及其在医药上的应用 | |
| US7056886B2 (en) | GLP-2 formulations | |
| WO2011161226A2 (en) | Novel antibody formulation | |
| EA028520B1 (ru) | Препараты этанерцепта, стабилизированные меглюмином | |
| EA029193B1 (ru) | Составы этанерцепта, отличающиеся заметным уменьшением содержания частиц довидимого диапазона | |
| KR20190078572A (ko) | 액상의 약학 조성물 | |
| RU2749342C1 (ru) | Фармацевтическая композиция на основе белкового комплекса il-15 и ее применения | |
| KR20220020821A (ko) | 이중 glp1/2 아고니스트의 약학적 비경구 조성물 | |
| JP6979427B2 (ja) | 可溶性Fc受容体の高濃度製剤 | |
| CN111375057A (zh) | 一种包含抗Her2单克隆抗体的药物配制剂 | |
| TW200826974A (en) | Stable lyophilized pharmaceutical preparation comprising antibody | |
| KR20210106476A (ko) | 고농도의 항-vegf 항체를 함유하는 단백질 용액 제형 | |
| EP3257524B1 (en) | Stabilized solution preparation of pharmaceutical glp-1r antibody fusion protein | |
| TW202128133A (zh) | 重組全人源抗tigit單株抗體製劑及其製備方法和用途 | |
| WO2012144579A1 (ja) | 溶液状抗体製剤 | |
| EP4070817A1 (en) | Liquid preparation containing anti-il-17 antibody | |
| CA3214867A1 (en) | Performance-enhancing excipients and methods of reducing viscosity and increasing stability of biologic formulations | |
| CN113827718A (zh) | 一种含有透明质酸酶的制剂及其应用 | |
| CN114652825A (zh) | 稳定的抗体制剂及其制备方法和应用 | |
| NZ714292B2 (en) | HIGHLY CONCENTRATED FORMULATIONS OF SOLUBLE Fc RECEPTORS | |
| TW202310873A (zh) | 含有抗人類tslp受體抗體之醫藥組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12747045 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2012558014 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14000103 Country of ref document: US Ref document number: 2012747045 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |