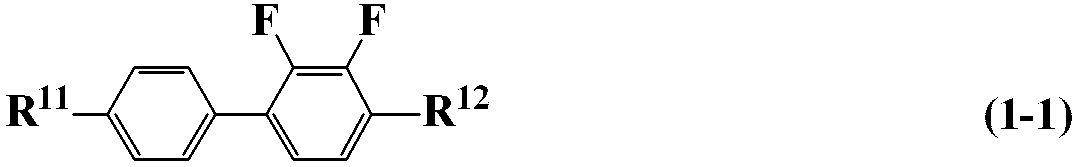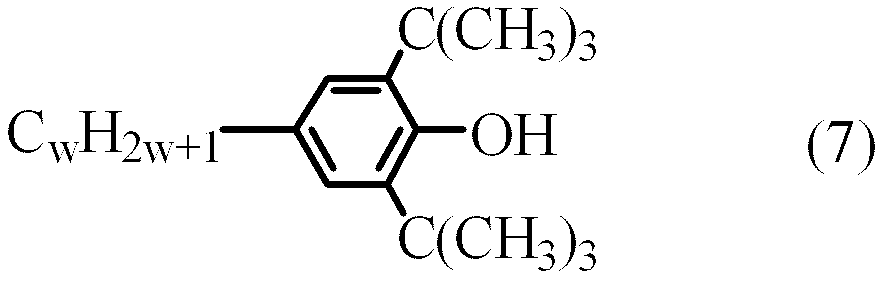WO2011132451A1 - 液晶組成物および液晶表示素子 - Google Patents
液晶組成物および液晶表示素子 Download PDFInfo
- Publication number
- WO2011132451A1 WO2011132451A1 PCT/JP2011/053496 JP2011053496W WO2011132451A1 WO 2011132451 A1 WO2011132451 A1 WO 2011132451A1 JP 2011053496 W JP2011053496 W JP 2011053496W WO 2011132451 A1 WO2011132451 A1 WO 2011132451A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- liquid crystal
- component
- carbons
- crystal composition
- compound
- Prior art date
Links
- 0 C*C(C1C(C)*c2ccc(C)cc2)C=C(**C(C(CC2)CCC2C(C)(C)*)=I)C([Si]C)=C1S Chemical compound C*C(C1C(C)*c2ccc(C)cc2)C=C(**C(C(CC2)CCC2C(C)(C)*)=I)C([Si]C)=C1S 0.000 description 3
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/42—Mixtures of liquid crystal compounds covered by two or more of the preceding groups C09K19/06 - C09K19/40
- C09K19/44—Mixtures of liquid crystal compounds covered by two or more of the preceding groups C09K19/06 - C09K19/40 containing compounds with benzene rings directly linked
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/12—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings at least two benzene rings directly linked, e.g. biphenyls
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/14—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings linked by a carbon chain
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3066—Cyclohexane rings in which the rings are linked by a chain containing carbon and oxygen atoms, e.g. esters or ethers
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/01—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour
- G02F1/13—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells
- G02F1/137—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells characterised by the electro-optical or magneto-optical effect, e.g. field-induced phase transition, orientation effect, guest-host interaction or dynamic scattering
- G02F1/139—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells characterised by the electro-optical or magneto-optical effect, e.g. field-induced phase transition, orientation effect, guest-host interaction or dynamic scattering based on orientation effects in which the liquid crystal remains transparent
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/12—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings at least two benzene rings directly linked, e.g. biphenyls
- C09K2019/121—Compounds containing phenylene-1,4-diyl (-Ph-)
- C09K2019/122—Ph-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3004—Cy-Cy
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3025—Cy-Ph-Ph-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3028—Cyclohexane rings in which at least two rings are linked by a carbon chain containing carbon to carbon single bonds
- C09K2019/3036—Cy-C2H4-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3028—Cyclohexane rings in which at least two rings are linked by a carbon chain containing carbon to carbon single bonds
- C09K2019/3037—Cy-Cy-C2H4-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3066—Cyclohexane rings in which the rings are linked by a chain containing carbon and oxygen atoms, e.g. esters or ethers
- C09K19/3068—Cyclohexane rings in which the rings are linked by a chain containing carbon and oxygen atoms, e.g. esters or ethers chain containing -COO- or -OCO- groups
- C09K2019/3078—Cy-Cy-COO-Ph-Cy
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2323/00—Functional layers of liquid crystal optical display excluding electroactive liquid crystal layer characterised by chemical composition
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2323/00—Functional layers of liquid crystal optical display excluding electroactive liquid crystal layer characterised by chemical composition
- C09K2323/03—Viewing layer characterised by chemical composition
Definitions
- the present invention mainly relates to a liquid crystal composition suitable for an AM (active matrix) device and the like, and an AM device containing the composition.
- a liquid crystal composition having a negative dielectric anisotropy, an IPS (in-plane switching) mode, VA (vertical alignment) mode, FFS (fringe field switching), or PSA (polymer sustained alignment) containing the composition. ) Mode elements.
- the classification based on the operation mode of the liquid crystal includes PC (phase change), TN (twisted nematic), STN (super twisted nematic), ECB (electrically controlled birefringence), OCB (optically compensated bend), IPS ( in-plane switching), VA (vertical alignment), FFS (fringe field switching), PSA (Polymer sustained alignment) mode, and the like.
- the classification based on the element drive system is PM (passive matrix) and AM (active matrix). PM is classified into static and multiplex, and AM is classified into TFT (thin film insulator), MIM (metal insulator metal), and the like. TFTs are classified into amorphous silicon and polycrystalline silicon. The latter is classified into a high temperature type and a low temperature type according to the manufacturing process.
- the classification based on the light source includes a reflection type using natural light, a transmission type using backlight, and a semi-transmission type using both natural light and backlight.
- the elements contain a liquid crystal composition having appropriate characteristics.
- This liquid crystal composition has a nematic phase.
- the general characteristics of the composition are improved.
- the relationships in the two general characteristics are summarized in Table 1 below.
- the general characteristics of the composition will be further described based on a commercially available AM device.
- the temperature range of the nematic phase is related to the temperature range in which the device can be used.
- a preferred upper limit temperature of the nematic phase is about 70 ° C. or more, and a preferred lower limit temperature of the nematic phase is about ⁇ 10 ° C. or less.
- the viscosity of the composition is related to the response time of the device. A short response time is preferred for displaying moving images on the device. Therefore, a small viscosity in the composition is preferred. Small viscosities at low temperatures are more preferred.
- the optical anisotropy of the composition is related to the contrast ratio of the device.
- the product ( ⁇ n ⁇ d) of the optical anisotropy ( ⁇ n) of the composition and the cell gap (d) of the device is designed to maximize the contrast ratio.
- the appropriate product value depends on the type of operation mode.
- the range is about 0.30 ⁇ m to about 0.40 ⁇ m for the VA mode device, and the range is about 0.20 ⁇ m to about 0.30 ⁇ m for the IPS mode or FFS mode device. In this case, a composition having a large optical anisotropy is preferable for a device having a small cell gap.
- the dielectric anisotropy having a large absolute value in the composition contributes to a low threshold voltage, a small power consumption and a large contrast ratio in the device. Therefore, a dielectric anisotropy having a large absolute value is preferable.
- a large specific resistance in the composition contributes to a large voltage holding ratio and a large contrast ratio in the device. Therefore, a composition having a large specific resistance not only at room temperature but also at a high temperature in the initial stage is preferable.
- a composition having a large specific resistance not only at room temperature but also at a high temperature after being used for a long time is preferable.
- the stability of the composition against ultraviolet rays and heat is related to the lifetime of the liquid crystal display device. When their stability is high, the lifetime of the device is long. Such characteristics are preferable for an AM device used in a liquid crystal projector, a liquid crystal television, and the like.
- a composition having a positive dielectric anisotropy is used for an AM device having a TN mode.
- a composition having negative dielectric anisotropy is used for an AM device having a VA mode.
- a composition having a positive or negative dielectric anisotropy is used in an AM device having an IPS mode or an FFS mode.
- a composition having a positive or negative dielectric anisotropy is used in an AM device having a PSA mode.
- Examples of liquid crystal compositions having negative dielectric anisotropy are disclosed in Patent Documents 1 to 4 described below.
- Desirable AM elements have such characteristics as a wide usable temperature range, a short response time, a large contrast ratio, a low threshold voltage, a large voltage holding ratio, and a long life. A shorter response time is desirable even at 1 millisecond. Therefore, desirable properties of the composition include a high maximum temperature of the nematic phase, a low minimum temperature of the nematic phase, a small viscosity, a suitable optical anisotropy, a large positive or negative dielectric anisotropy, a large specific resistance, and a high resistance to ultraviolet light. High stability, high heat stability, etc.
- One object of the present invention is to provide a high maximum temperature of the nematic phase, a low minimum temperature of the nematic phase, a small viscosity, a suitable optical anisotropy, a large negative dielectric anisotropy, a large specific resistance, and a high stability to ultraviolet light.
- the liquid crystal composition satisfies at least one characteristic in characteristics such as high stability to heat.
- Another object is a liquid crystal composition having an appropriate balance with respect to at least two properties, particularly a liquid crystal composition satisfying a high maximum temperature and a small viscosity.
- Another object is a liquid crystal display device containing such a composition.
- Another object is a composition having characteristics such as small optical anisotropy, or appropriate optical anisotropy that is large optical anisotropy, negative large dielectric anisotropy, and high stability to ultraviolet light,
- the AM element has characteristics such as a short response time, a large voltage holding ratio, a large contrast ratio, and a long lifetime.
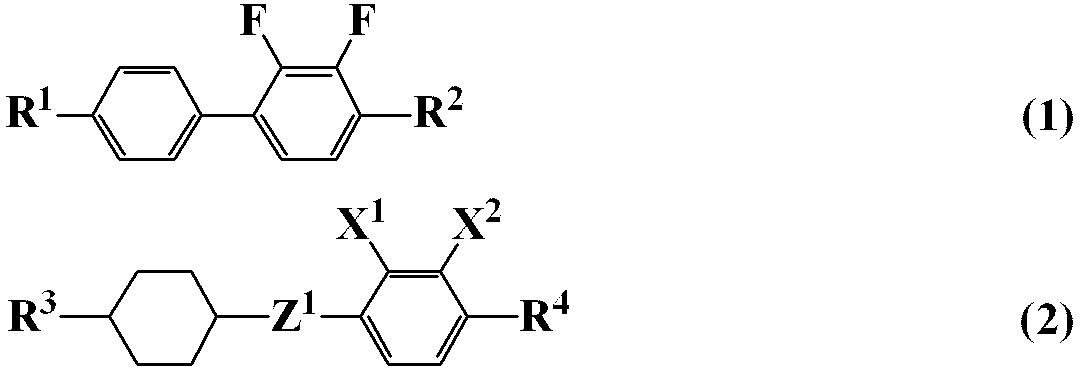
- R 1 , R 2 , R 3 , and R 4 are independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, or alkenyl having 2 to 12 carbons;
- X 1 and X 2 is independently fluorine or chlorine;
- Z 1 is ethylene or methyleneoxy.
- the advantages of the present invention are that the upper limit temperature of the nematic phase, the lower limit temperature of the nematic phase, the small viscosity, the appropriate optical anisotropy, the negative dielectric constant anisotropy, the large specific resistance, the high stability to ultraviolet rays, the heat It is a liquid crystal composition satisfying at least one of the properties such as high stability against the liquid crystal.
- One aspect of the present invention is a liquid crystal composition having an appropriate balance regarding at least two properties.
- Another aspect is a liquid crystal display device containing such a composition.
- Other aspects are compositions with suitable optical anisotropy, negatively large dielectric anisotropy, high stability to ultraviolet light, etc., and short response time, large voltage holding ratio, large contrast ratio, long lifetime An AM device having the above.
- liquid crystal composition of the present invention or the liquid crystal display device of the present invention may be abbreviated as “composition” or “device”, respectively.
- a liquid crystal display element is a general term for a liquid crystal display panel and a liquid crystal display module.
- Liquid crystal compound means a compound having a liquid crystal phase such as a nematic phase or a smectic phase, or a compound having no liquid crystal phase but useful as a component of a composition. This useful compound has a six-membered ring such as 1,4-cyclohexylene and 1,4-phenylene, and its molecular structure is rod-like. Optically active compounds and polymerizable compounds may be added to the composition.
- Compound (1) means one compound or two or more compounds represented by formula (1). The same applies to compounds represented by other formulas.
- “Arbitrary” means that not only the position but also the number can be freely selected.
- the upper limit temperature of the nematic phase may be abbreviated as “upper limit temperature”.
- the lower limit temperature of the nematic phase may be abbreviated as “lower limit temperature”.
- “High specific resistance” means that the composition has a large specific resistance not only at room temperature in the initial stage but also at a temperature close to the upper limit temperature of the nematic phase. It means having a large specific resistance even at a close temperature.
- “High voltage holding ratio” means that the device has a large voltage holding ratio not only at room temperature in the initial stage but also at a temperature close to the upper limit temperature of the nematic phase. It means having a large voltage holding ratio even at a temperature close to.
- the first component is one compound or two or more compounds.
- the “ratio of the first component” means the weight percentage (% by weight) of the first component based on the total weight of the liquid crystal composition. The same applies to the ratio of the second component.
- the ratio of the additive mixed with the composition means a percentage by weight (% by weight) or a percentage by weight (ppm) based on the total weight of the liquid crystal composition.
- R 3 is used for a plurality of compounds.
- the groups selected for R 3 of any two of these compounds may be the same or different.
- R 3 of compound (2) is ethyl and R 3 of compound (2-1) is ethyl.
- R 3 of compound (2) is ethyl and R 3 of compound (2-1) is propyl.
- This rule also applies to R 1 , R 2 , Z 1 and the like.
- the present invention includes the following items.
- R 1 , R 2 , R 3 , and R 4 are independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, or alkenyl having 2 to 12 carbons;
- X 1 and X 2 is independently fluorine or chlorine;
- Z 1 is ethylene or methyleneoxy.
- Item 2 The liquid crystal composition according to item 1, wherein the second component is at least one compound selected from the group of compounds represented by formula (2-1) or formula (2-2).
- R 3 and R 4 are independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, or alkenyl having 2 to 12 carbons.
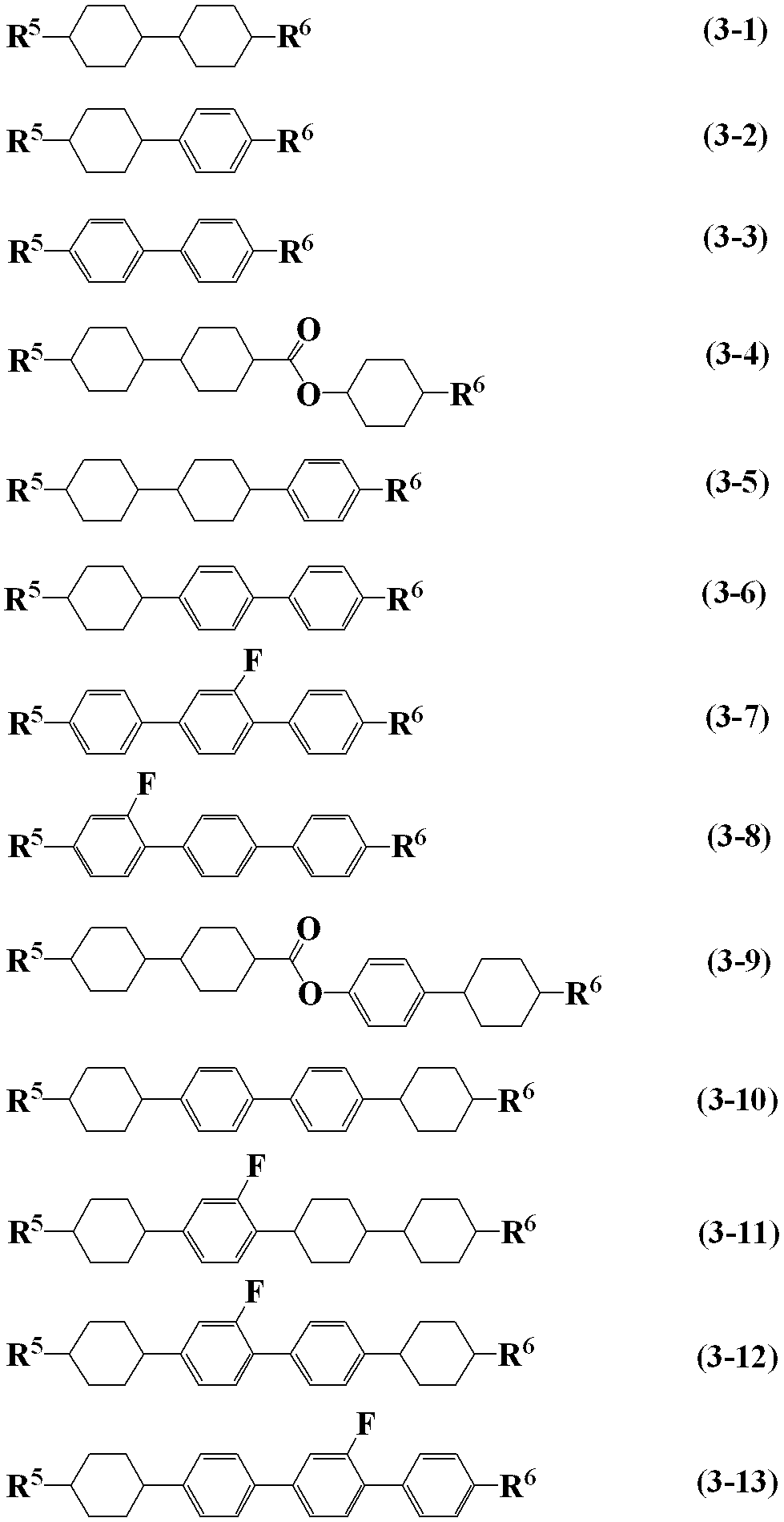
- Item 4 The liquid crystal composition according to any one of items 1 to 3, further comprising at least one compound selected from the group of compounds represented by formula (3) as a third component.
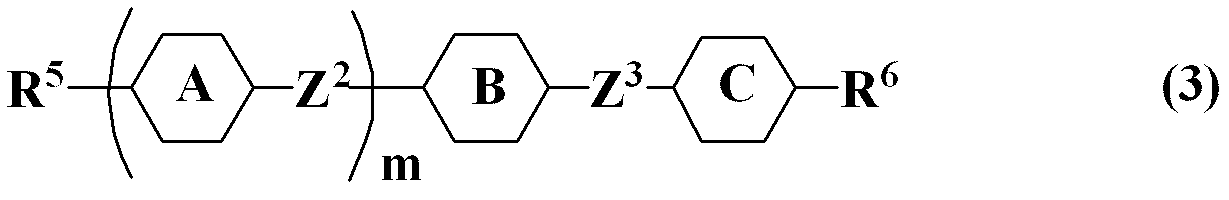
- R 5 and R 6 are each independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, alkenyl having 2 to 12 carbons, or carbon 2 having any hydrogen substituted with fluorine.
- Ring A, ring B, and ring C are independently 1,4-cyclohexylene, 1,4-phenylene, 2-fluoro-1,4-phenylene, or 3-fluoro- 1,4-phenylene;
- Z 2 and Z 3 are independently a single bond, ethylene, methyleneoxy, or carbonyloxy; m is 0, 1, or 2.
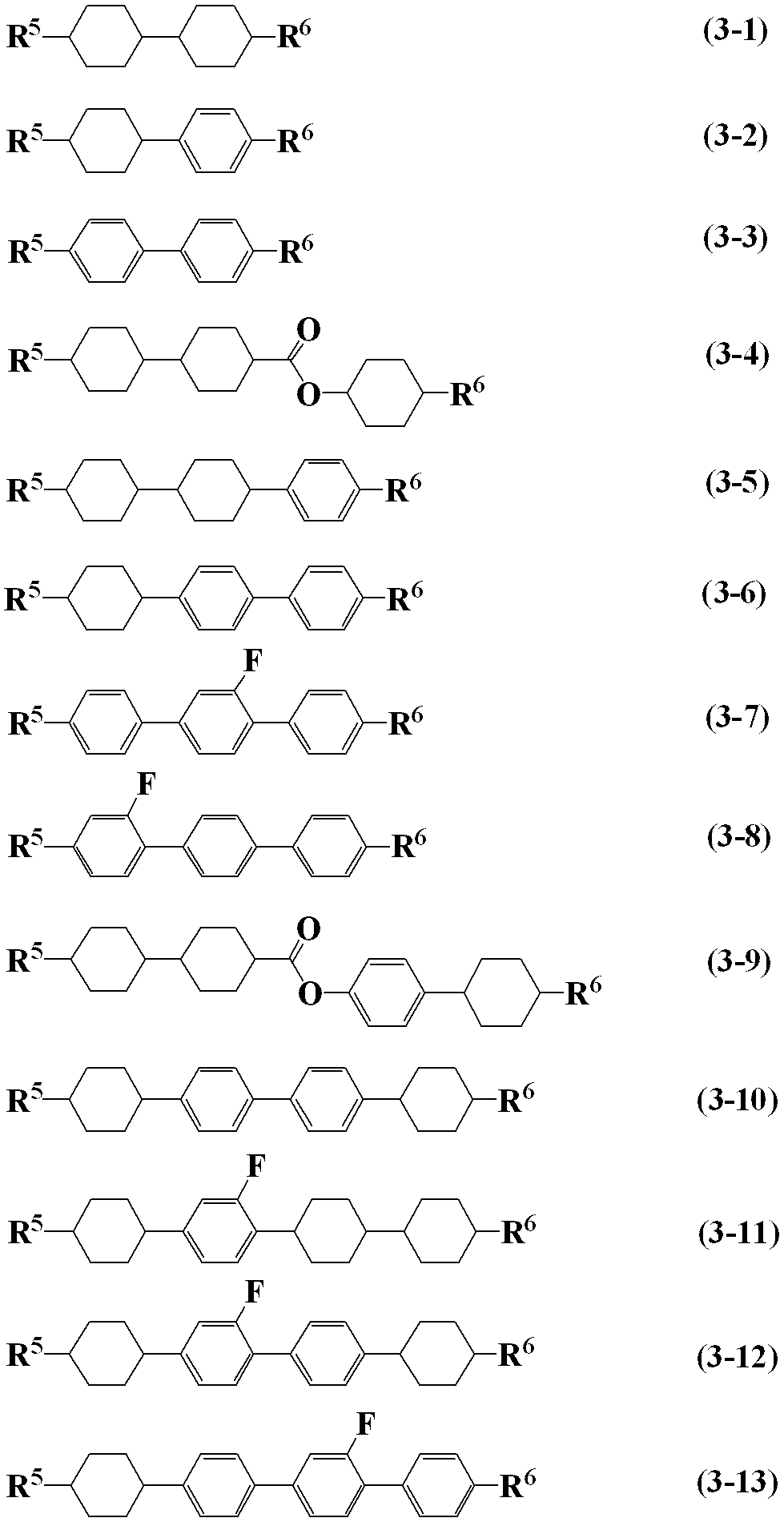
- Item 5 The liquid crystal composition according to item 4, wherein the third component is at least one compound selected from the group of compounds represented by formulas (3-1) to (3-13).
- R 5 and R 6 are each independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, alkenyl having 2 to 12 carbons, or carbon 2 having any hydrogen substituted with fluorine. To 12 alkenyl.
- the proportion of the first component is in the range of 5 wt% to 40 wt%
- the proportion of the second component is in the range of 5 wt% to 85 wt%
- the third component Item 8 The liquid crystal composition according to any one of items 4 to 7, wherein the ratio of is in the range of 10% by weight to 90% by weight.
- Item 9 The liquid crystal composition according to any one of items 1 to 8, further containing at least one compound selected from the group of compounds represented by formula (4) as a fourth component.
- R 7 and R 8 are independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, or alkenyl having 2 to 12 carbons;
- ring D and ring E are independently 1,4-cyclohexylene or 1,4-phenylene;
- X 3 and X 4 are independently fluorine or chlorine;
- Y 1 is hydrogen or methyl;
- Z 4 and Z 5 are independently Single bond, ethylene, or carbonyloxy;
- p is 1, 2, or 3
- q is 0 or 1 and the sum of p and q is 2 or 3.
- Item 10 The liquid crystal composition according to item 9, wherein the fourth component is at least one compound selected from the group of compounds represented by formula (4-1) to formula (4-4).
- R 7 and R 8 are independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, or alkenyl having 2 to 12 carbons.
- Item 13 The liquid crystal composition according to any one of items 9 to 12, wherein the ratio of the fourth component is in the range of 5% to 40% by weight based on the total weight of the liquid crystal composition.
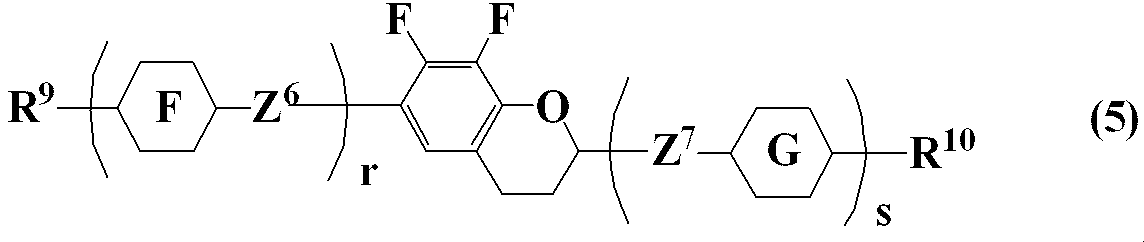
- Item 14 The liquid crystal composition according to any one of items 1 to 13, further containing at least one compound selected from the group of compounds represented by formula (5) as a fifth component.
- R 9 and R 10 are independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, or alkenyl having 2 to 12 carbons;
- ring F and ring G are independently 1,4-cyclohexylene or 1,4-phenylene;
- Z 6 and Z 7 are independently a single bond, ethylene, methyleneoxy, or carbonyloxy; r is 0, 1, or 2 , S is 0 or 1, and the sum of r and s is 1 or 2.
- Item 15 The liquid crystal composition according to item 14, wherein the fifth component is at least one compound selected from the group of compounds represented by formulas (5-1) to (5-5).
- R 9 and R 10 are independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, or alkenyl having 2 to 12 carbons.
- Item 17 The liquid crystal composition according to any one of items 14 to 16, wherein the ratio of the fifth component is in the range of 5% to 30% by weight based on the total weight of the liquid crystal composition.
- the upper limit temperature of the nematic phase is 70 ° C. or higher, the optical anisotropy (25 ° C.) at a wavelength of 589 nm is 0.08 or higher, and the dielectric anisotropy (25 ° C.) at a frequency of 1 kHz is ⁇ 2 or lower.
- Item 18 The liquid crystal composition according to any one of items 1 to 17.
- Item 19 A liquid crystal display device comprising the liquid crystal composition according to any one of items 1 to 18.
- Item 20 The liquid crystal display element according to item 19, wherein the operation mode of the liquid crystal display element is a VA mode, an IPS mode, an FFS mode, or a PSA mode, and the driving method of the liquid crystal display element is an active matrix method.
- the present invention includes the following items. 1) The above composition further containing an optically active compound, 2) the above composition further containing an additive such as an antioxidant, an ultraviolet absorber, and an antifoaming agent, and 3) the above composition.
- AM device 4) a device containing the above composition and having a mode of TN, ECB, OCB, IPS, FFS, VA, or PSA, 5) a transmissive device containing the above composition, 6) Use of the above composition as a composition having a nematic phase, 7) Use as an optically active composition by adding an optically active compound to the above composition.
- composition of the present invention will be described in the following order. First, the constitution of component compounds in the composition will be described. Second, the main characteristics of the component compounds and the main effects of the compounds on the composition will be explained. Third, the combination of components in the composition, the preferred ratio of the components, and the basis thereof will be described. Fourth, a preferred form of the component compound will be described. Fifth, specific examples of component compounds are shown. Sixth, additives that may be mixed into the composition will be described. Seventh, a method for synthesizing the component compounds will be described. Finally, the use of the composition will be described.
- composition A includes other liquid crystal compounds, additives, impurities, etc. May further be contained.
- the “other liquid crystal compound” is a liquid crystal compound different from the compound (1), the compound (2), the compound (3), the compound (4), and the compound (5). Such compounds are mixed into the composition for the purpose of further adjusting the properties.
- a smaller amount of cyano compound is preferable from the viewpoint of stability to heat or ultraviolet light.
- a more desirable ratio of the cyano compound is 0% by weight.
- Additives include optically active compounds, antioxidants, ultraviolet absorbers, dyes, antifoaming agents, polymerizable compounds, polymerization initiators, and the like. Impurities are compounds mixed in a process such as synthesis of component compounds. Even if this compound is a liquid crystal compound, it is classified as an impurity here.
- Composition B consists essentially of a compound selected from Compound (1), Compound (2), Compound (3), Compound (4), and Compound (5). “Substantially” means that the composition may contain additives and impurities, but does not contain a liquid crystal compound different from these compounds. Composition B has fewer components than composition A. From the viewpoint of reducing the cost, the composition B is preferable to the composition A. The composition A is preferable to the composition B from the viewpoint that the physical properties can be further adjusted by mixing other liquid crystal compounds.
- the main characteristics of the component compounds and the main effects of the compounds on the characteristics of the composition will be explained.
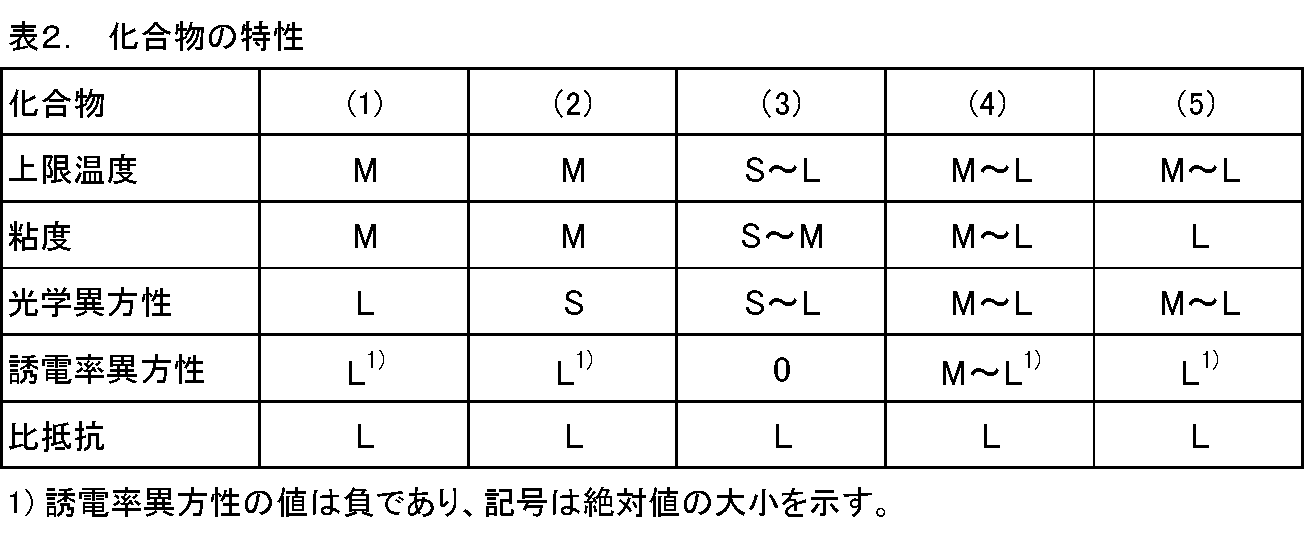
- the main characteristics of the component compounds are summarized in Table 2 based on the effects of the present invention.
- L represents large or high
- M represents moderate
- S represents small or low.
- L, M, and S are classifications based on a qualitative comparison among the component compounds, and 0 (zero) indicates that the value is almost zero.
- Compound (1) increases the absolute value of dielectric anisotropy and increases optical anisotropy.
- Compound (2) increases the absolute value of dielectric anisotropy and decreases the minimum temperature.
- Compound (3) decreases the viscosity, adjusts the appropriate optical anisotropy, increases the maximum temperature, and decreases the minimum temperature.
- Compound (4) increases the absolute value of dielectric anisotropy and decreases the minimum temperature.
- Compound (5) increases the absolute value of dielectric anisotropy and decreases the minimum temperature.
- first component + second component first component + second component + third component
- first component + second component + fourth component first component + second component + fifth Component
- first component + second component + third component + fourth component first component + second component + third component + fifth component
- first component + second component + fourth component + fifth component First component + second component + third component + fourth component + fifth component.
- a preferred combination of the components in the composition is first component + second component + third component for increasing the maximum temperature or decreasing the viscosity, and for increasing the absolute value of the dielectric anisotropy, the viscosity In order to reduce the minimum temperature or the lower limit temperature, the first component + the second component + the third component + the fourth component.
- a desirable ratio of the first component is approximately 5% by weight or more for increasing the absolute value of the dielectric anisotropy, and approximately 40% by weight or less for decreasing the minimum temperature.
- a more desirable ratio is in the range of approximately 10% by weight to approximately 35% by weight.
- a particularly preferred ratio is in the range of approximately 15% by weight to approximately 30% by weight.
- a desirable ratio of the second component is approximately 5% by weight or more for decreasing the viscosity, and approximately 85% by weight or less for decreasing the minimum temperature.
- a more desirable ratio is in the range of approximately 10% by weight to approximately 70% by weight for decreasing the viscosity.
- a particularly preferred ratio is in the range of approximately 15% by weight to approximately 50% by weight.
- a desirable ratio of the third component is approximately 10% by weight or more for increasing the absolute value of the dielectric anisotropy, and approximately 90% by weight or less for decreasing the minimum temperature.
- a more desirable ratio is in the range of approximately 30% by weight to approximately 80% by weight.
- a particularly preferred ratio is in the range of approximately 40% by weight to approximately 70% by weight.
- a desirable ratio of the fourth component is approximately 5% by weight or more for decreasing the viscosity, and approximately 40% by weight or less for decreasing the minimum temperature.
- a more desirable ratio is in the range of approximately 10% by weight to approximately 35% by weight.
- a particularly preferred ratio is in the range of approximately 15% by weight to approximately 30% by weight.
- a desirable ratio of the fifth component is approximately 5% by weight or more for increasing the absolute value of the dielectric anisotropy, and approximately 30% by weight or less for decreasing the minimum temperature.
- a more desirable ratio is in the range of approximately 10% by weight to approximately 25% by weight.
- a particularly preferred ratio is in the range of approximately 15% by weight to approximately 20% by weight.
- R 1 , R 2 , R 3 , R 4 , R 7 , R 8 , R 9 , and R 10 are independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, or 2 carbons 12 alkenyl
- R 5 and R 6 are independently alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, alkenyl having 2 to 12 carbons, or any hydrogen replaced with fluorine Alkenyl having 2 to 12 carbons.
- R 1 , R 2 , R 3 , R 4 , R 7 , R 8 , R 9 , or R 10 are alkyl having 1 to 12 carbons for decreasing the minimum temperature or decreasing the viscosity, or Alkenyl having 2 to 12 carbons. More desirable R 2 , R 4 , R 6 or R 8 is alkoxy having 1 to 12 carbons for increasing the absolute value of dielectric anisotropy. Desirable R 5 or R 6 is alkenyl having 2 to 12 carbons for decreasing the minimum temperature or decreasing the viscosity, and has 1 to 12 carbons for increasing the stability to ultraviolet light or heat. Alkyl.
- Preferred alkyl is methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl or octyl. More desirable alkyl is ethyl, propyl, butyl, pentyl, or heptyl for decreasing the viscosity.
- Preferred alkoxy is methoxy, ethoxy, propoxy, butoxy, pentyloxy, hexyloxy, or heptyloxy. More desirable alkoxy is methoxy or ethoxy for decreasing the viscosity.
- Preferred alkenyl is vinyl, 1-propenyl, 2-propenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, or 5-hexenyl. More desirable alkenyl is vinyl, 1-propenyl, 3-butenyl, or 3-pentenyl for decreasing the viscosity.
- the preferred configuration of —CH ⁇ CH— in these alkenyls depends on the position of the double bond.
- Trans is preferable in alkenyl such as 1-propenyl, 1-butenyl, 1-pentenyl, 1-hexenyl, 3-pentenyl and 3-hexenyl for decreasing the viscosity.
- Cis is preferred for alkenyl such as 2-butenyl, 2-pentenyl, and 2-hexenyl.
- linear alkenyl is preferable to branching.
- alkenyl in which any hydrogen is replaced by fluorine include 2,2-difluorovinyl, 3,3-difluoro-2-propenyl, 4,4-difluoro-3-butenyl, 5,5-difluoro-4- Pentenyl and 6,6-difluoro-5-hexenyl. Further preferred examples are 2,2-difluorovinyl and 4,4-difluoro-3-butenyl for decreasing the viscosity.
- Alkyl does not include cyclic alkyl.
- Alkoxy does not include cyclic alkoxy.
- Alkenyl does not include cyclic alkenyl.
- m is 0, 1, or 2. Desirable m is 1 or 2 for increasing the maximum temperature, and 0 for decreasing the viscosity.
- p is 1 to 3, q is 0 or 1, and the sum of p and q is 2 or 3.
- Preferred p is 2 for increasing the maximum temperature, and 1 for decreasing the viscosity.
- Preferred q is 1 for increasing the maximum temperature, and 0 for decreasing the minimum temperature.
- a preferable sum of p and q is 2 for decreasing the minimum temperature.
- r is 1 or 2
- s is 0 or 1
- Desirable r is 2 for increasing the maximum temperature. Desirable s is 0 for decreasing the minimum temperature.
- a preferable sum of r and s is 2 for increasing the maximum temperature.
- Ring A, Ring B, and Ring C are independently 1,4-cyclohexylene, 1,4-phenylene, 2-fluoro-1,4-phenylene, or 3-fluoro-1,4-phenylene.
- the two rings A may be the same or different.
- Desirable ring A, ring B or ring C is 1,4-cyclohexylene for increasing the maximum temperature, and 1,4-phenylene for increasing the optical anisotropy.
- Ring D and Ring E are independently 1,4-cyclohexylene, or 1,4-phenylene, and any two rings D when p is 2 or 3 are the same or different May be.
- Desirable ring D or ring E is 1,4-cyclohexylene for increasing the maximum temperature, and 1,4-phenylene for decreasing the minimum temperature.
- Ring F and Ring G are each independently 1,4-cyclohexylene or 1,4-phenylene, and when r is 2, the two rings F may be the same or different.
- Desirable ring F or ring G is 1,4-cyclohexylene for increasing the maximum temperature, and 1,4-phenylene for decreasing the minimum temperature.
- X 1 , X 2 , X 3 , and X 4 are independently fluorine or chlorine. Desirable X 1 , X 2 , X 3 or X 4 is fluorine for decreasing the viscosity.
- Y 1 is hydrogen or methyl.
- Preferred Y 1 is hydrogen for decreasing the viscosity, and methyl for increasing the stability to ultraviolet light, heat and the like.
- Z 1 is ethylene or methyleneoxy. Preferred Z 1 is ethylene for decreasing the viscosity.
- Z 2 , Z 3 , Z 6 , and Z 7 are each independently a single bond, ethylene, methyleneoxy, or carbonyloxy, and two Z 2 when m is 2 are the same or different The two Z 6 when r is 2 may be the same or different.
- Desirable Z 2 , Z 3 , Z 6 or Z 7 is a single bond or ethylene for decreasing the viscosity, and methyleneoxy or carbonyloxy for increasing the dielectric anisotropy.
- Z 4 and Z 5 are independently a single bond, ethylene, or carbonyloxy, and any two Z 4 s when p is 2 or 3 may be the same or different. Desirable Z 4 or Z 5 is a single bond or ethylene for decreasing the viscosity, and carbonyloxy for increasing the dielectric anisotropy.
- R 6 is alkyl having 1 to 12 carbons, alkoxy having 1 to 12 carbons, alkenyl having 2 to 12 carbons, or 2 to 12 carbons in which any hydrogen is replaced by fluorine.
- R 11 and R 13 are independently alkyl having 1 to 12 carbons and alkenyl having 2 to 12 carbons.
- R 12 is alkyl having 1 to 12 carbons and alkoxy having 1 to 12 carbons.
- trans is preferable to cis for the configuration of 1,4-cyclohexylene for increasing the maximum temperature.
- a preferred compound (1) is the compound (1-1). Desirable compounds (2) are the compound (2-1-1) and the compound (2-2-1). More desirable compound (2) is the compound (2-1-1). Desirable compounds (3) are from the compound (3-1-1) to the compound (3-13-1). More desirable compounds (3) are the compound (3-1-1) to the compound (3-5-1), and the compound (3-7-1) to the compound (3-13-1). Particularly preferred compounds (3) are the compound (3-1-1), the compound (3-3-1), the compound (3-5-1), the compound (3-7-1), the compound (3-8- 1), the compound (3-9-1), and the compound (3-13-1). Desirable compounds (4) are from the compound (4-1-1) to the compound (4-4-1). More desirable compounds (4) are the compound (4-1-1), the compound (4-3-1) and the compound (4-4-1). Particularly preferred compounds (4) are the compound (4-1-1) and the compound (4-3-1). Desirable compounds (5) are from the compound (5-1-1) to the compound (5-5-1). More desirable compound (5) is the compound (5-4-1).
- additives that may be mixed with the composition will be described.
- Such additives are optically active compounds, antioxidants, ultraviolet absorbers, dyes, antifoaming agents, polymerizable compounds, polymerization initiators, and the like.
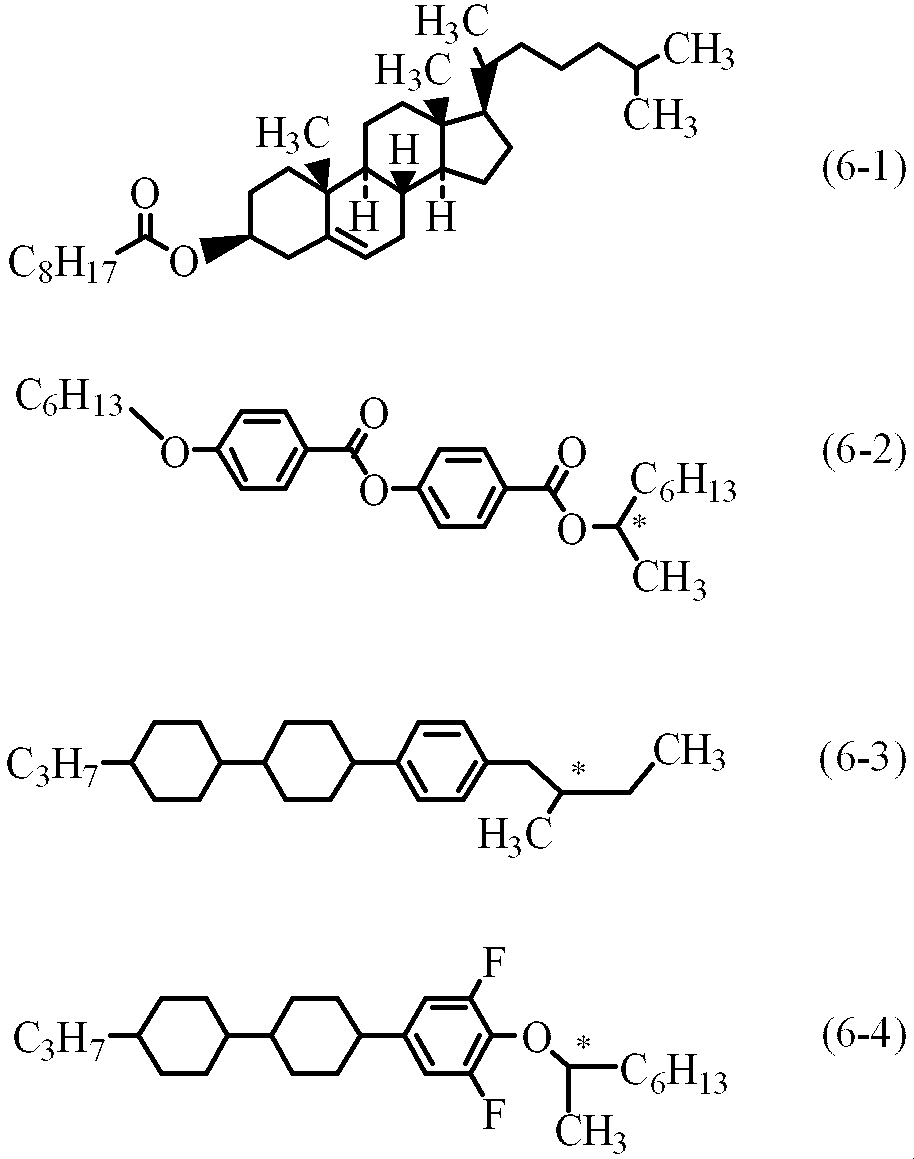
- An optically active compound is mixed with the composition for the purpose of inducing a helical structure of liquid crystal to give a twist angle. Examples of such a compound are the compound (6-1) to the compound (6-4).
- a desirable ratio of the optically active compound is approximately 5% by weight or less.
- a more desirable ratio is in the range of approximately 0.01% by weight to approximately 2% by weight.
- oxidation prevention An agent is mixed into the composition.
- Preferred examples of the antioxidant include a compound (7) where w is an integer of 1 to 9.
- preferred w is 1, 3, 5, 7, or 9. Further preferred w is 1 or 7. Since the compound (7) in which w is 1 has high volatility, it is effective in preventing a decrease in specific resistance due to heating in the atmosphere. Since the compound (7) in which w is 7 has low volatility, it is effective for maintaining a large voltage holding ratio not only at room temperature but also at a temperature close to the upper limit temperature of the nematic phase after using the device for a long time. .
- a desirable ratio of the antioxidant is approximately 50 ppm or more for achieving its effect, and is approximately 600 ppm or less for avoiding a decrease in the maximum temperature or avoiding an increase in the minimum temperature. A more desirable ratio is in the range of approximately 100 ppm to approximately 300 ppm.
- the ultraviolet absorber examples include benzophenone derivatives, benzoate derivatives, triazole derivatives and the like. Also preferred are light stabilizers such as sterically hindered amines.
- a desirable ratio in these absorbents and stabilizers is approximately 50 ppm or more for obtaining the effect thereof, and approximately 10,000 ppm or less for avoiding a decrease in the maximum temperature or avoiding an increase in the minimum temperature. A more desirable ratio is in the range of approximately 100 ppm to approximately 10,000 ppm.
- a dichroic dye such as an azo dye or an anthraquinone dye is mixed with the composition so as to be adapted to a GH (guest host) mode element.
- a preferred ratio of the dye is in the range of approximately 0.01% by weight to approximately 10% by weight.
- an antifoaming agent such as dimethyl silicone oil or methylphenyl silicone oil is mixed with the composition.
- a desirable ratio of the antifoaming agent is approximately 1 ppm or more for obtaining the effect thereof, and approximately 1000 ppm or less for preventing a poor display.
- a more desirable ratio is in the range of approximately 1 ppm to approximately 500 ppm.
- a polymerizable compound is mixed with the composition in order to adapt to a PSA (polymer-sustained alignment) mode element.
- Preferred examples of the polymerizable compound are compounds having a polymerizable group such as acrylate, methacrylate, vinyl compound, vinyloxy compound, propenyl ether, epoxy compound (oxirane, oxetane), vinyl ketone and the like. Particularly preferred examples are acrylate or methacrylate derivatives.
- a desirable ratio of the polymerizable compound is approximately 0.05% by weight or more for obtaining the effect thereof, and approximately 10% by weight or less for preventing a display defect. A more desirable ratio is in the range of approximately 0.1% by weight to approximately 2% by weight.
- the polymerizable compound is preferably polymerized by UV irradiation or the like in the presence of a suitable initiator such as a photopolymerization initiator.
- a suitable initiator such as a photopolymerization initiator.
- Appropriate conditions for polymerization, the appropriate type of initiator, and the appropriate amount are known to those skilled in the art and are described in the literature.
- Irgacure 651 registered trademark
- Irgacure 184 registered trademark
- Darocure 1173 registered trademark
- a preferred ratio of the photopolymerization initiator is in the range of about 0.1% to about 5% by weight of the polymerizable compound, and a particularly preferable ratio is in the range of about 1% to about 3% by weight.
- compositions mainly have a minimum temperature of about ⁇ 10 ° C. or lower, a maximum temperature of about 70 ° C. or higher, and an optical anisotropy in the range of about 0.07 to about 0.20.
- a device containing this composition has a large voltage holding ratio.
- This composition is suitable for an AM device.
- This composition is particularly suitable for a transmissive AM device.
- a composition having an optical anisotropy in the range of about 0.08 to about 0.25 may be prepared by controlling the ratio of the component compounds or by mixing other liquid crystal compounds.
- This composition can be used as a composition having a nematic phase, or can be used as an optically active composition by adding an optically active compound.
- This composition can be used for an AM device. Further, it can be used for PM elements.
- This composition can be used for an AM device and a PM device having modes such as PC, TN, STN, ECB, OCB, IPS, FFS, VA, and PSA.
- Use for an AM device having an IPS, FFS or VA mode is particularly preferable.
- These elements may be reflective, transmissive, or transflective. Use in a transmissive element is preferred. It can also be used for an amorphous silicon-TFT device or a polycrystalline silicon-TFT device.
- NCAP non-curvilinear-aligned-phase
- PD polymer-dispersed
- the composition and this compound are used as measurement objects.
- the object to be measured was a composition, it was measured as it was, and the obtained value was described.
- the smectic phase or crystal
- the ratio of the compound and the mother liquid crystal is 10% by weight: 90% by weight, 5% by weight: 95% by weight, 1% by weight: 99% by weight in this order. changed.
- the maximum temperature, optical anisotropy, viscosity and dielectric anisotropy values for the compound were determined.
- the components of the mother liquid crystals and their proportions are as follows.
- Measured characteristic values according to the following method. Many of them are the methods described in the Standard of Electronics Industry Association of Japan EIAJ ED-2521A, or a modified method thereof.
- NI Maximum temperature of nematic phase
- a sample was placed on a hot plate of a melting point measuring device equipped with a polarizing microscope and heated at a rate of 1 ° C./min. The temperature was measured when a part of the sample changed from a nematic phase to an isotropic liquid.
- the upper limit temperature of the nematic phase may be abbreviated as “upper limit temperature”.
- T C Minimum temperature of nematic phase
- Viscosity (bulk viscosity; ⁇ ; measured at 20 ° C .; mPa ⁇ s): An E-type viscometer was used for measurement.
- the dielectric constants ( ⁇ and ⁇ ) were measured as follows. 1) Measurement of dielectric constant ( ⁇ ): An ethanol (20 mL) solution of octadecyltriethoxysilane (0.16 mL) was applied to a well-cleaned glass substrate. The glass substrate was rotated with a spinner and then heated at 150 ° C. for 1 hour. A sample was put in a VA element in which the distance between two glass substrates (cell gap) was 4 ⁇ m, and the element was sealed with an adhesive that was cured with ultraviolet rays.
- Sine waves (0.5 V, 1 kHz) were applied to the device, and after 2 seconds, the dielectric constant ( ⁇ ) in the major axis direction of the liquid crystal molecules was measured.
- 2) Measurement of dielectric constant ( ⁇ ) A polyimide solution was applied to a well-cleaned glass substrate. After baking this glass substrate, the obtained alignment film was rubbed. A sample was put in a TN device in which the distance between two glass substrates (cell gap) was 9 ⁇ m and the twist angle was 80 degrees. Sine waves (0.5 V, 1 kHz) were applied to the device, and after 2 seconds, the dielectric constant ( ⁇ ) in the minor axis direction of the liquid crystal molecules was measured.
- Threshold voltage (Vth; measured at 25 ° C .; V): An LCD5100 luminance meter manufactured by Otsuka Electronics Co., Ltd. was used for measurement.
- the light source is a halogen lamp.
- a sample is placed in a normally black mode VA device in which the distance between two glass substrates (cell gap) is 4 ⁇ m and the rubbing direction is anti-parallel, and this device is used with a UV curing adhesive. And sealed.
- the voltage (60 Hz, rectangular wave) applied to this element was increased stepwise from 0V to 20V by 0.02V.
- the device was irradiated with light from the vertical direction, and the amount of light transmitted through the device was measured.
- a voltage-transmittance curve was created in which the transmittance was 100% when the light amount reached the maximum and the transmittance was 0% when the light amount was the minimum.
- the threshold voltage is a voltage when the transmittance reaches 10%.
- Voltage holding ratio (VHR-1; 25 ° C .;%):
- the TN device used for the measurement has a polyimide alignment film, and the distance between two glass substrates (cell gap) is 5 ⁇ m. This element was sealed with an adhesive polymerized by ultraviolet rays after putting a sample.
- the TN device was charged by applying a pulse voltage (60 microseconds at 5 V).
- the decaying voltage was measured for 16.7 milliseconds with a high-speed voltmeter, and the area A between the voltage curve and the horizontal axis in a unit cycle was determined.
- the area B is an area when it is not attenuated.
- the voltage holding ratio is a percentage of the area A with respect to the area B.
- Voltage holding ratio (VHR-2; 80 ° C .;%):
- the TN device used for the measurement has a polyimide alignment film, and the distance between two glass substrates (cell gap) is 5 ⁇ m. This element was sealed with an adhesive polymerized by ultraviolet rays after putting a sample.
- the TN device was charged by applying a pulse voltage (60 microseconds at 5 V).
- the decaying voltage was measured for 16.7 milliseconds with a high-speed voltmeter, and the area A between the voltage curve and the horizontal axis in a unit cycle was determined.
- the area B is an area when it is not attenuated.
- the voltage holding ratio is a percentage of the area A with respect to the area B.
- VHR-3 Voltage holding ratio
- the TN device used for measurement has a polyimide alignment film, and the cell gap is 5 ⁇ m.
- a sample was injected into this element and irradiated with light for 20 minutes.
- the light source is an ultra high pressure mercury lamp USH-500D (manufactured by USHIO), and the distance between the element and the light source is 20 cm.
- USH-500D ultra high pressure mercury lamp manufactured by USHIO
- the decreasing voltage was measured for 16.7 milliseconds.
- a composition having a large VHR-3 has a large stability to ultraviolet light.
- VHR-3 is preferably 90% or more, and more preferably 95% or more.
- VHR-4 Voltage holding ratio
- a TN device injected with a sample was heated in a thermostat at 80 ° C. for 500 hours, and then the voltage holding ratio was measured to evaluate the stability against heat. In measurement 4, the decaying voltage was measured for 16.7 milliseconds. .
- a composition having a large VHR-4 has a large stability to heat.
- Response time ( ⁇ ; measured at 25 ° C .; ms): An LCD5100 luminance meter manufactured by Otsuka Electronics Co., Ltd. was used for measurement.
- the light source is a halogen lamp.
- the low-pass filter was set to 5 kHz.
- a sample is placed in a normally black mode VA device in which the distance between two glass substrates (cell gap) is 4 ⁇ m and the rubbing direction is anti-parallel, and this device is used with a UV curing adhesive. And sealed.
- a rectangular wave 60 Hz, 10 V, 0.5 seconds
- the device was irradiated with light from the vertical direction, and the amount of light transmitted through the device was measured.
- the transmittance is 100% when the light amount is maximum, and the transmittance is 0% when the light amount is minimum.
- the response time is the time (fall time; millisecond) required to change the transmittance from 90% to 10%.
- GC-14B gas chromatograph manufactured by Shimadzu Corporation was used for measurement.
- the carrier gas is helium (2 mL / min).
- the sample vaporization chamber was set at 280 ° C, and the detector (FID) was set at 300 ° C.
- capillary column DB-1 (length 30 m, inner diameter 0.32 mm, film thickness 0.25 ⁇ m; stationary liquid phase is dimethylpolysiloxane; nonpolar) manufactured by Agilent Technologies Inc. was used.
- the column was held at 200 ° C. for 2 minutes and then heated to 280 ° C. at a rate of 5 ° C./min.
- a sample was prepared in an acetone solution (0.1% by weight), and 1 ⁇ L thereof was injected into the sample vaporization chamber.
- the recorder is a C-R5A Chromatopac manufactured by Shimadzu Corporation, or an equivalent product.
- the obtained gas chromatogram showed the peak retention time and peak area corresponding to the component compounds.
- capillary column As a solvent for diluting the sample, chloroform, hexane or the like may be used.
- the following capillary column may be used.
- HP-1 from Agilent Technologies Inc. (length 30 m, inner diameter 0.32 mm, film thickness 0.25 ⁇ m), Rtx-1 from Restek Corporation (length 30 m, inner diameter 0.32 mm, film thickness 0.25 ⁇ m), BP-1 (length 30 m, inner diameter 0.32 mm, film thickness 0.25 ⁇ m) manufactured by SGE International Pty.
- a capillary column CBP1-M50-025 length 50 m, inner diameter 0.25 mm, film thickness 0.25 ⁇ m
- Shimadzu Corporation may be used.
- the ratio of the liquid crystal compound contained in the composition may be calculated by the following method. Liquid crystalline compounds can be detected by gas chromatography. The area ratio of peaks in the gas chromatogram corresponds to the ratio (number of moles) of liquid crystal compounds. When the capillary column described above is used, the correction coefficient of each liquid crystal compound may be regarded as 1. Accordingly, the ratio (% by weight) of the liquid crystal compound is calculated from the peak area ratio.
- the present invention will be described in detail by examples.
- the present invention is not limited by the following examples.
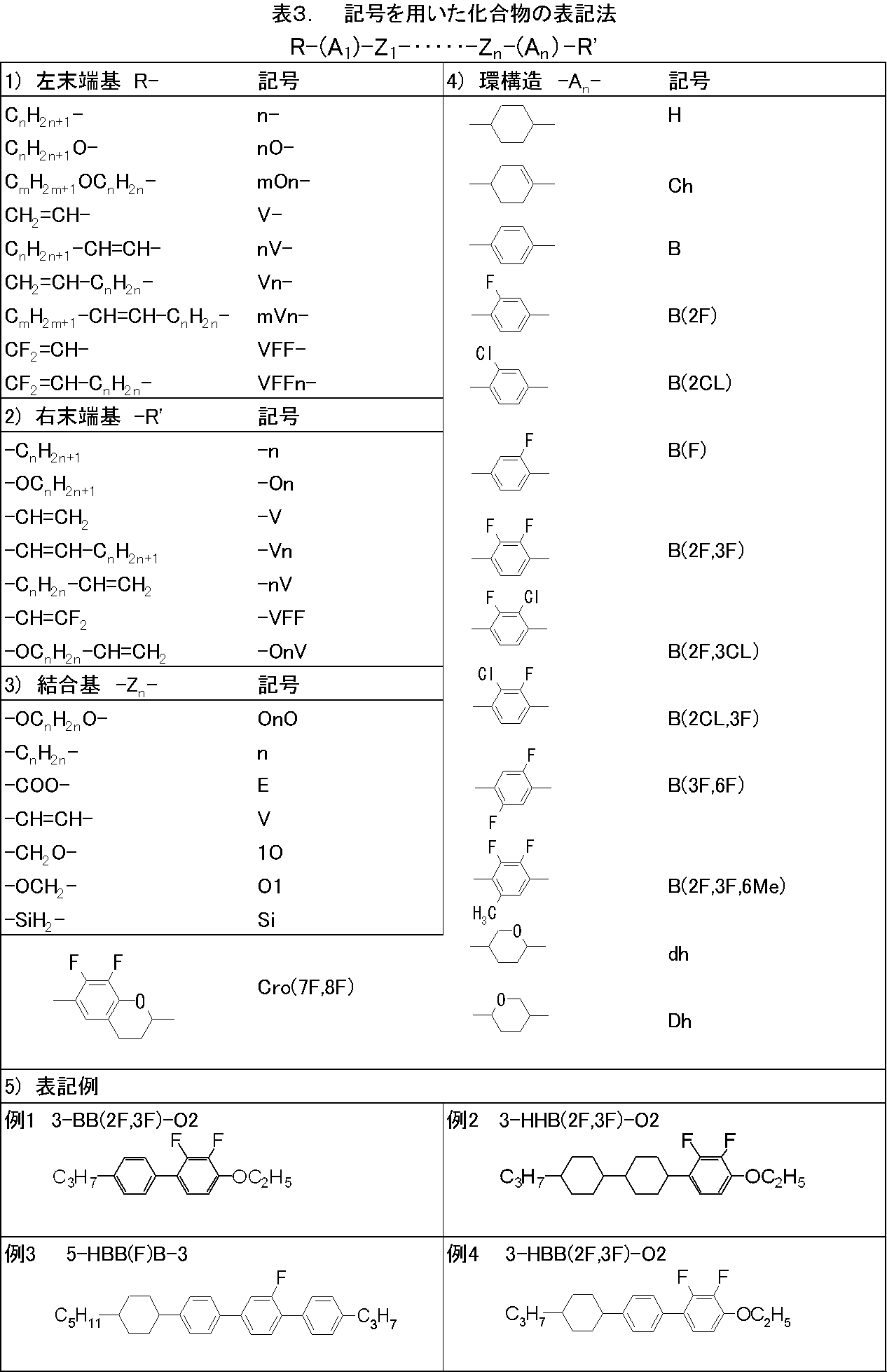
- the compounds in Comparative Examples and Examples were represented by symbols based on the definitions in Table 3 below.
- Table 3 the configuration regarding 1,4-cyclohexylene is trans.
- the number in parentheses after the symbol corresponds to the compound number.
- the symbol ( ⁇ ) means other liquid crystal compounds.
- the ratio (percentage) of the liquid crystal compound is a weight percentage (% by weight) based on the total weight of the liquid crystal composition, and the liquid crystal composition contains impurities.
- Example 7 was selected from the compositions disclosed in JP2009-035630A. This is because the composition is composed of a compound similar to the compound (1), the compound (3), the compound (4), and the compound (2).
- the components and properties of this composition are as follows.
- Example 1 A compound similar to compound (2) of Comparative Example 1 was replaced with compound (2-1-1).
- the components and properties of this composition are as follows. In Example 1, the upper limit temperature is higher, the lower limit temperature is lower, and the viscosity is lower than that of Comparative Example 1.
- Example 2 A compound similar to compound (2) of Comparative Example 1 was replaced with compound (2-2-1).
- the components and properties of this composition are as follows.
- the upper limit temperature is higher than that of Comparative Example 1
- the lower limit temperature is lower
- the absolute value of dielectric anisotropy is larger
- the viscosity is smaller.
- Example 3 was selected from the compositions disclosed in Japanese Patent Application Laid-Open No. 2008-285570. This is because the composition is composed of the compound (1), the compound (3), and the compound (4). The components and properties of this composition are as follows.
- Example 6 was selected from the compositions disclosed in Japanese Patent Application Laid-Open No. 2008-024815. This is because the composition is composed of the compound (1), the compound (3), and the compound (4). The components and properties of this composition are as follows.
- Example 3 was selected from the compositions disclosed in Japanese Patent Application Laid-Open No. 2003-327965. This is because the composition is composed of the compound (1), the compound (3), and the compound (4). This composition was prepared and measured by the method described above. The components and properties of this composition are as follows.
- compositions of Examples 1 to 11 have a lower viscosity than Comparative Examples 1 to 4. Therefore, the liquid crystal composition according to the present invention has more excellent characteristics than the liquid crystal compositions disclosed in Patent Documents 1 to 4.
- the liquid crystal composition satisfies at least one characteristic or has an appropriate balance regarding at least two characteristics.
- a liquid crystal display element containing such a composition becomes an AM element having a short response time, a large voltage holding ratio, a large contrast ratio, a long lifetime, and the like, and thus can be used for a liquid crystal projector, a liquid crystal television, and the like.
Abstract
Description
ここで、R1、R2、R3、およびR4は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、または炭素数2から12のアルケニルであり;X1およびX2は独立して、フッ素または塩素であり;Z1は、エチレンまたはメチレンオキシである。
ここで、R3およびR4は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、または炭素数2から12のアルケニルである。
ここで、R5およびR6は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、炭素数2から12のアルケニル、または任意の水素がフッ素で置き換えられた炭素数2から12のアルケニルであり;環A、環B、および環Cは独立して、1,4-シクロへキシレン、1,4-フェニレン、2-フルオロ-1,4-フェニレン、または3-フルオロ-1,4-フェニレンであり;Z2およびZ3は独立して、単結合、エチレン、メチレンオキシ、またはカルボニルオキシであり;mは、0、1、または2である。
ここで、R5およびR6は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、炭素数2から12のアルケニル、または任意の水素がフッ素で置き換えられた炭素数2から12のアルケニルである。
ここで、R7およびR8は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、または炭素数2から12のアルケニルであり;環Dおよび環Eは独立して、1,4-シクロへキシレンまたは1,4-フェニレンであり;X3およびX4は独立して、フッ素または塩素であり;Y1は、水素またはメチルであり;Z4およびZ5は独立して、単結合、エチレン、またはカルボニルオキシであり;pは、1、2、または3であり、qは0または1であり、そして、pとqの和が2または3である。
ここで、R7およびR8は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、または炭素数2から12のアルケニルである。
ここで、R9およびR10は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、または炭素数2から12のアルケニルであり;環Fおよび環Gは独立して、1,4-シクロヘキシレンまたは1,4-フェニレンであり;Z6およびZ7は独立して、単結合、エチレン、メチレンオキシ、またはカルボニルオキシであり;rは、0、1、または2であり、sは0または1であり、そして、rとsとの和が1または2である。
ここで、R9およびR10は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、または炭素数2から12のアルケニルである。
R1、R2、R3、R4、R7、R8、R9、およびR10は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、または炭素数2から12のアルケニルであり、R5およびR6は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、炭素数2から12のアルケニル、または任意の水素がフッ素で置き換えられた炭素数2から12のアルケニルである。
pは1から3であり、qは0または1であり、そして、pとqの和が2または3である。好ましいpは、上限温度を上げるために2であり、粘度を下げるために1である。好ましいqは上限温度を上げるために1であり、下限温度を下げるために0である。好ましいpとqの和は、下限温度を下げるために2である。
rは1または2であり、sは0または1であり、そして、rとsとの和が1または2である。好ましいrは上限温度を上げるために2である。好ましいsは下限温度を下げるために0である。好ましいrとsの和は、上限温度を上げるために2である。
環Dおよび環Eは独立して、1,4-シクロへキシレン、または1,4-フェニレンであり、pが2または3であるときの任意の2つの環Dは同じであっても、異なってもよい。好ましい環Dまたは環Eは、上限温度を上げるために1,4-シクロヘキシレンであり、下限温度を下げるために1,4-フェニレンである。
環Fおよび環Gは独立して、1,4-シクロヘキシレン、または1,4-フェニレンであり、rが2であるときの2つの環Fは同じであっても、異なってもよい。好ましい環Fまたは環Gは、上限温度を上げるために1,4-シクロヘキシレンであり、下限温度を下げるために1,4-フェニレンである。
Z2、Z3、Z6、およびZ7は独立して、単結合、エチレン、メチレンオキシ、またはカルボニルオキシであり、mが2であるときの2つのZ2は同じであっても、異なってもよく、rが2であるときの2つのZ6は同じであっても、異なってもよい。好ましいZ2、Z3、Z6、またはZ7は、粘度を下げるために、単結合またはエチレンであり、誘電率異方性を上げるために、メチレンオキシまたはカルボニルオキシである。
Z4およびZ5は独立して、単結合、エチレン、またはカルボニルオキシであり、pが2または3であるときの任意の2つのZ4は同じであっても、異なってもよい。好ましいZ4またはZ5は、粘度を下げるために単結合またはエチレンであり、誘電率異方性を上げるために、カルボニルオキシである。
下記の好ましい化合物において、R6は、炭素数1から12のアルキル、炭素数1から12のアルコキシ、炭素数2から12のアルケニル、または任意の水素がフッ素で置き換えられた炭素数2から12のアルケニルである。R11およびR13は独立して、炭素数1から12のアルキルおよび炭素数2から12のアルケニルである。R12は、炭素数1から12のアルキルおよび炭素数1から12のアルコキシである。これらの化合物において1,4-シクロヘキシレンに関する立体配置は、上限温度を上げるためにシスよりもトランスが好ましい。
1)誘電率(ε∥)の測定:よく洗浄したガラス基板にオクタデシルトリエトキシシラン(0.16mL)のエタノール(20mL)溶液を塗布した。ガラス基板をスピンナーで回転させたあと、150℃で1時間加熱した。2枚のガラス基板の間隔(セルギャップ)が4μmであるVA素子に試料を入れ、この素子を紫外線で硬化する接着剤で密閉した。この素子にサイン波(0.5V、1kHz)を印加し、2秒後に液晶分子の長軸方向における誘電率(ε∥)を測定した。
2)誘電率(ε⊥)の測定:よく洗浄したガラス基板にポリイミド溶液を塗布した。このガラス基板を焼成した後、得られた配向膜にラビング処理をした。2枚のガラス基板の間隔(セルギャップ)が9μmであり、ツイスト角が80度であるTN素子に試料を入れた。この素子にサイン波(0.5V、1kHz)を印加し、2秒後に液晶分子の短軸方向における誘電率(ε⊥)を測定した。
表3において、1,4-シクロヘキシレンに関する立体配置はトランスである。実施例において記号の後にあるかっこ内の番号は化合物の番号に対応する。(-)の記号はその他の液晶性化合物を意味する。液晶性化合物の割合(百分率)は、液晶組成物の全重量に基づいた重量百分率(重量%)であり、液晶組成物には不純物が含まれている。最後に、組成物の特性値をまとめた。
特開2009-035630号公報に開示された組成物の中から実施例7を選んだ。根拠はこの組成物が、化合物(1)、化合物(3)、化合物(4)、および化合物(2)に類似の化合物からなる組成物だからである。この組成物の成分および特性は下記のとおりである。
V2-BB(2F,3F)-O2 (1-1) 5%
3-HB(2F,3F)-O2 (2)類似 13%
5-HB(2F,3F)-O2 (2)類似 5%
3-HH-V (3-1-1) 24%
V2-BB-1 (3-3-1) 6%
V-HHB-1 (3-5-1) 5%
3-BB(F)B-2V (3-7-1) 5%
3-HHB(2F,3F)-O2 (4-1-1) 5%
2-HChB(2F,3F)-O2 (-) 7%
3-HChB(2F,3F)-O2 (-) 14%
V-HChB(2F,3F)-O2 (-) 8%
1O1-HBBH-5 (-) 3%
NI=86.3℃;Tc≦-20℃;Δn=0.111;η=15.9mPa・s;Δε=-2.7.
比較例1の化合物(2)に類似の化合物を、化合物(2-1-1)と置き換えた。この組成物の成分および特性は下記のとおりである。実施例1は比較例1に比べて上限温度が高く、下限温度が低く、粘度が小さくなっている。
V2-BB(2F,3F)-O2 (1-1) 5%
3-H2B(2F,3F)-O2 (2-1-1) 13%
5-H2B(2F,3F)-O2 (2-1-1) 5%
3-HH-V (3-1-1) 24%
V2-BB-1 (3-3-1) 6%
V-HHB-1 (3-5-1) 5%
3-BB(F)B-2V (3-7-1) 5%
3-HHB(2F,3F)-O2 (4-1-1) 5%
2-HChB(2F,3F)-O2 (-) 7%
3-HChB(2F,3F)-O2 (-) 14%
V-HChB(2F,3F)-O2 (-) 8%
1O1-HBBH-5 (-) 3%
NI=86.7℃;Tc≦-30℃;Δn=0.111;η=14.5mPa・s;Δε=-2.8;VHR-1=99.3%;VHR-2=98.2%;VHR-3=98.1%.
比較例1の化合物(2)に類似の化合物を、化合物(2-2-1)と置き換えた。この組成物の成分および特性は下記のとおりである。実施例2は比較例1に比べて上限温度が高く、下限温度が低く、誘電率異方性の絶対値が大きく、粘度が小さくなっている。
V2-BB(2F,3F)-O2 (1-1) 5%
3-H1OB(2F,3F)-O2 (2-2-1) 13%
5-H1OB(2F,3F)-O2 (2-2-1) 5%
3-HH-V (3-1-1) 24%
V2-BB-1 (3-3-1) 6%
V-HHB-1 (3-5-1) 5%
3-BB(F)B-2V (3-7-1) 5%
3-HHB(2F,3F)-O2 (4-1-1) 5%
2-HChB(2F,3F)-O2 (-) 7%
3-HChB(2F,3F)-O2 (-) 14%
V-HChB(2F,3F)-O2 (-) 8%
1O1-HBBH-5 (-) 3%
NI=87.1℃;Tc≦-30℃;Δn=0.112;η=15.1mPa・s;Δε=-3.3;VHR-1=99.1%;VHR-2=97.9%;VHR-3=97.9%
特開2008-285570号公報に開示された組成物の中から実施例3を選んだ。根拠はこの組成物が、化合物(1)、化合物(3)、および化合物(4)からなる組成物だからである。この組成物の成分および特性は下記のとおりである。
3-BB(2F,3F)-O2 (1-1) 10%
V2-BB(2F,3F)-O2 (1-1) 5%
3-HHB-1 (3-5-1) 5%
V-HHB-1 (3-5-1) 5%
5-HBB(F)B-3 (3-13-1) 5%
3-HHB(2F,3F)-O2 (4-1-1) 7%
5-HHB(2F,3F)-O2 (4-1-1) 7%
3-HH2B(2F,3F)-O2 (4-3-1) 6%
V-HBB(2F,3F)-O2 (4) 5%
3-HB(2F,3F)-O2 (-) 10%
5-HB(2F,3F)-O2 (-) 10%
3-HVH-1 (-) 7%
3-HVH-2 (-) 7%
3-HVHH-2 (-) 3%
V-HVHB-1 (-) 8%
NI=90.6℃;Tc≦-20℃;Δn=0.109;η=21.9mPa・s;Δε=-3.0.
特開2008-024815号公報に開示された組成物の中から実施例6を選んだ。根拠はこの組成物が、化合物(1)、化合物(3)、および化合物(4)からなる組成物だからである。この組成物の成分および特性は下記のとおりである。
3-BB(2F,3F)-O2 (1-1) 15%
5-BB(2F,3F)-O2 (1-1) 15%
5-HBB(F)B-2 (3-13-1) 12%
5-HBB(F)B-3 (3-13-1) 12%
2-BB(2F,3F)B-3 (4-4-1) 5%
2-BB(2F,3F)B-4 (4-4-1) 10%
5-BB(2F,3F)B-2 (4-4-1) 10%
3-B2B(2F,3F)-O2 (-) 11%
5-B2B(2F,3F)-O2 (-) 10%
NI=84.2℃;Tc≦-20℃;Δn=0.197;η=35.7mPa・s;Δε=-3.6.
特開2003-327965号公報に開示された組成物の中から例3を選んだ。根拠はこの組成物が、化合物(1)、化合物(3)、および化合物(4)からなる組成物だからである。本組成物を調合し、上述した方法により測定した。この組成物の成分および特性は下記のとおりである。
5-BB(2F,3F)-O4 (1-1) 5%
5-HH-V (3-1-1) 12%
3-HH-V1 (3-1-1) 12%
5-HB-3 (3-2-1) 7%
1V2-BB-1 (3-3-1) 7%
3-HBB-2 (3-6-1) 5%
2-HBB(2F,3F)-O2 (4) 11%
3-HBB(2F,3F)-O2 (4) 12%
2-BB(2F)B(2F,3F)-1 (4) 7%
3-BB(2F)B(2F,3F)-1 (4) 7%
5-HB(2F,3F)-O2 (-) 5%
5-HB(2F,3F)-O4 (-) 10%
NI=74.3℃;Tc≦-20℃;Δn=0.126;η=17.9mPa・s;Δε=-2.8.
3-BB(2F,3F)-O2 (1-1) 3%
V2-BB(2F,3F)-O2 (1-1) 6%
1V2-BB(2F,3F)-O2 (1-1) 4%
3-H2B(2F,3F)-O2 (2-1-1) 12%
5-H2B(2F,3F)-O2 (2-1-1) 12%
3-HH-V (3-1-1) 20%
3-HH-V1 (3-1-1) 8%
3-HB-O2 (3-2-1) 5%
1V2-BB-1 (3-3-1) 6%
3-HHEBH-3 (3-9-1) 5%
3-HHEBH-4 (3-9-1) 3%
3-HHEBH-5 (3-9-1) 3%
3-HB(F)HH-5 (3-11-1) 3%
5-HBB(F)B-2 (3-13-1) 5%
5-HBB(F)B-3 (3-13-1) 5%
NI=87.7℃;Tc≦-20℃;Δn=0.112;η=14.3mPa・s;Δε=-2.3;VHR-1=99.5%;VHR-2=98.3%;VHR-3=98.0%.
3-BB(2F,3F)-O2 (1-1) 6%
5-BB(2F,3F)-O2 (1-1) 3%
1V2-BB(2F,3F)-O2 (1-1) 5%
3-H2B(2F,3F)-O2 (2-1-1) 20%
3-HH-5 (3-1-1) 4%
3-HH-O1 (3-1-1) 4%
3-HH-V1 (3-1-1) 8%
3-HB-O1 (3-2-1) 5%
V-BB-1 (3-3-1) 7%
1V2-BB-1 (3-3-1) 5%
3-HHEH-5 (3-4-1) 5%
3-HHB-O1 (3-5-1) 5%
2-BB(F)B-5 (3-7-1) 3%
2-B(F)BB-2V (3-8-1) 3%
3-HHEBH-3 (3-9-1) 6%
3-HHEBH-4 (3-9-1) 2%
3-HHEBH-5 (3-9-1) 3%
5-HB(F)HH-V (3-11-1) 3%
3-HB(F)BH-3 (3-12-1) 3%
NI=87.7℃;Tc≦-20℃;Δn=0.120;η=14.4mPa・s;Δε=-2.3;VHR-1=99.6%;VHR-2=98.1%;VHR-3=98.2%.
2-BB(2F,3F)-O2 (1-1) 4%
5-BB(2F,3F)-O2 (1-1) 4%
8-BB(2F,3F)-O2 (1-1) 4%
V2-BB(2F,3F)-O2 (1-1) 4%
1V2-BB(2F,3F)-O2 (1-1) 6%
3-H2B(2F,3F)-O2 (2-1-1) 9%
5-H2B(2F,3F)-O2 (2-1-1) 9%
1-HH-2V1 (3-1-1) 5%
2-HH-2V1 (3-1-1) 5%
3-HH-2V1 (3-1-1) 5%
3-HH-VFF (3-1-1) 5%
V-BB-1 (3-3-1) 5%
V2-HHB-1 (3-5-1) 5%
3-HBB-2 (3-6-1) 3%
5-B(F)BB-2 (3-8-1) 7%
3-B(F)BB-2V (3-8-1) 5%
3-HHEBH-3 (3-9-1) 3%
3-HHEBH-5 (3-9-1) 3%
3-HBBH-3 (3-10-1) 3%
5-HB(F)HH-V (3-11-1) 3%
5-HBB(F)B-3 (3-13-1) 3%
NI=87.5℃;Tc≦-20℃;Δn=0.129;η=14.5mPa・s;Δε=-2.2;VHR-1=99.3%;VHR-2=98.1%;VHR-3=98.1%.
3-BB(2F,3F)-O2 (1-1) 5%
5-BB(2F,3F)-O2 (1-1) 3%
8-BB(2F,3F)-O2 (1-1) 5%
V2-BB(2F,3F)-O2 (1-1) 5%
3-H2B(2F,3F)-O2 (2-1-1) 5%
3-HH-V (3-1-1) 23%
V-BB-1 (3-3-1) 7%
1V2-BB-1 (3-3-1) 3%
3-HHB-1 (3-5-1) 7%
3-HHEBH-3 (3-9-1) 5%
5-HBB(F)B-3 (3-13-1) 3%
5-HHB(2F,3F)-O2 (4-1-1) 3%
3-HHB(2F,3F)-1 (4-1-1) 5%
3-HH2B(2F,3F)-O2 (4-3-1) 5%
5-HH2B(2F,3F)-O2 (4-3-1) 7%
2-BB(2F,3F)B-4 (4-4-1) 3%
3-HH1OB(2F,3F,6Me)-O2(4) 3%
1O1-HBBH-5 (-) 3%
NI=91.0℃;Tc≦-20℃;Δn=0.114;η=14.4mPa・s;Δε=-2.5;VHR-1=99.1%;VHR-2=98.0%;VHR-3=97.8%.
3-BB(2F,3F)-O2 (1-1) 3%
V2-BB(2F,3F)-O2 (1-1) 5%
1V2-BB(2F,3F)-O2 (1-1) 5%
3-H2B(2F,3F)-O2 (2-1-1) 5%
5-H2B(2F,3F)-O2 (2-1-1) 15%
3-HH-V (3-1-1) 20%
3-HH-V1 (3-1-1) 7%
3-HH-2V1 (3-1-1) 3%
V-BB-1 (3-3-1) 7%
3-HHB-O1 (3-5-1) 5%
3-HHEBH-3 (3-9-1) 4%
3-HHEBH-5 (3-9-1) 3%
5-HBB(F)B-2 (3-13-1) 5%
5-HBB(F)B-3 (3-13-1) 5%
5-HHB(2F,3CL)-O2 (4-2-1) 3%
5-HH2B(2F,3F)-O2 (4-3-1) 5%
NI=90.9℃;Tc≦-20℃;Δn=0.112;η=14.2mPa・s;Δε=-2.4;VHR-1=99.3%;VHR-2=98.3%;VHR-3=98.2%.
5-BB(2F,3F)-O2 (1-1) 3%
V2-BB(2F,3F)-O2 (1-1) 7%
5-H2B(2F,3F)-O2 (2-1-1) 9%
3-HH-V (3-1-1) 20%
3-HH-V1 (3-1-1) 7%
1-HH-2V1 (3-1-1) 4%
V-BB-1 (3-3-1) 8%
1V2-BB-1 (3-3-1) 5%
3-B(F)BB-2V (3-8-1) 3%
3-HHEBH-3 (3-9-1) 5%
3-HHEBH-5 (3-9-1) 4%
5-HB(F)BH-3 (3-12-1) 3%
5-HBB(F)B-2 (3-13-1) 5%
1V2-HHB(2F,3F)-O2 (4-1-1) 5%
5-HH2B(2F,3F)-O2 (4-3-1) 3%
5-H2Cro(7F,8F)-5 (5-1-1) 3%
3-HH2Cro(7F,8F)-5 (5-3-1) 3%
3-HH1OCro(7F,8F)-5 (5-4-1) 3%
NI=92.2℃;Tc≦-20℃;Δn=0.113;η=14.7mPa・s;Δε=-2.4;VHR-1=98.9%;VHR-2=97.8%;VHR-3=97.5%.
V2-BB(2F,3F)-O2 (1-1) 5%
1V2-BB(2F,3F)-O2 (1-1) 5%
3-H2B(2F,3F)-O2 (2-1-1) 5%
5-H2B(2F,3F)-O2 (2-1-1) 5%
3-HH-5 (3-1-1) 5%
3-HH-V (3-1-1) 10%
5-HH-V (3-1-1) 5%
3-HH-V1 (3-1-1) 6%
1-HH-2V1 (3-1-1) 6%
V-BB-1 (3-3-1) 5%
V2-BB-1 (3-3-1) 3%
1V2-BB-1 (3-3-1) 5%
3-HHEBH-3 (3-9-1) 5%
3-HHEBH-5 (3-9-1) 3%
3-HBBH-3 (3-10-1) 5%
5-HBB(F)B-3 (3-13-1) 5%
1V2-HHB(2F,3F)-O2 (4-1-1) 3%
5-HH2B(2F,3F)-O2 (4-3-1) 5%
5-H1OCro(7F,8F)-5 (5-2-1) 3%
5-HB1OCro(7F,8F)-5 (5-5-1) 3%
4O-Cro(7F,8F)H-3 (5) 3%
NI=92.5℃;Tc≦-20℃;Δn=0.112;η=14.8mPa・s;Δε=-2.5;VHR-1=98.8%;VHR-2=97.6%;VHR-3=97.6%.
V2-BB(2F,3F)-O2 (1-1) 6%
5-BB(2F,3F)-1 (1-1) 5%
V-H1OB(2F,3F)-O2 (2-2-1) 10%
3-H2B(2F,3CL)-O2 (2) 4%
5-H1OB(2F,3CL)-O2 (2) 4%
3-HH-V (3-1-1) 24%
3-HH-V1 (3-1-1) 7%
2-HH-2V1 (3-1-1) 3%
V-BB-1 (3-3-1) 7%
3-HHB-O1 (3-5-1) 5%
3-HHEBH-3 (3-9-1) 4%
3-HHEBH-4 (3-9-1) 3%
3-HHEBH-5 (3-9-1) 3%
5-HBB(F)B-2 (3-13-1) 5%
5-HBB(F)B-3 (3-13-1) 5%
5-HH2B(2F,3F)-O2 (4-3-1) 5%
NI=89.3℃;Tc≦-20℃;Δn=0.104;η=14.8mPa・s;Δε=-2.4;VHR-1=98.9%;VHR-2=97.7%;VHR-3=97.8%.
V2-BB(2F,3F)-O2 (1-1) 8%
1V2-BB(2F,3F)-O2 (1-1) 5%
5-H2B(2F,3F)-O2 (2-1-1) 5%
5-H1OB(2F,3F)-O2 (2-2-1) 5%
5-H2B(2CL,3F)-O2 (2) 5%
5-H1OB(2CL,3F)-O2 (2) 5%
3-HH-V (3-1-1) 23%
3-HH-V1 (3-1-1) 4%
1-HH-2V1 (3-1-1) 3%
2-HH-2V1 (3-1-1) 4%
V-BB-1 (3-3-1) 5%
V2-HHB-1 (3-5-1) 4%
3-HHEBH-3 (3-9-1) 5%
3-HHEBH-4 (3-9-1) 3%
3-HHEBH-5 (3-9-1) 3%
5-HBB(F)B-3 (3-13-1) 8%
5-HH2B(2F,3F)-O2 (4-3-1) 5%
NI=89.1℃;Tc≦-20℃;Δn=0.102;η=14.6mPa・s;Δε=-2.4;VHR-1=98.8%;VHR-2=97.8%;VHR-3=97.9%.
Claims (20)
- 液晶組成物の全重量に基づいて、第一成分の割合が15重量%から40重量%の範囲であり、第二成分の割合が15重量%から85重量%の範囲である請求項1または2のいずれか1項に記載の液晶組成物。
- 第三成分として式(3)で表される化合物の群から選択された少なくとも1つの化合物をさらに含有する、請求項1から3のいずれか1項に記載の液晶組成物。
ここで、R5およびR6は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、炭素数2から12のアルケニル、または任意の水素がフッ素で置き換えられた炭素数2から12のアルケニルであり;環A、環B、および環Cは独立して、1,4-シクロへキシレン、1,4-フェニレン、2-フルオロ-1,4-フェニレン、または3-フルオロ-1,4-フェニレンであり;Z2およびZ3は独立して、単結合、エチレン、メチレンオキシ、またはカルボニルオキシであり;mは、0、1、または2である。 - 第三成分が式(3-1)で表される化合物の群から選択された少なくとも1つの化合物である請求項5に記載の液晶組成物。
- 第三成分が式(3-13)で表される化合物の群から選択された少なくとも1つの化合物である請求項5に記載の液晶組成物。
- 液晶組成物の全重量に基づいて、第一成分の割合が5重量%から40重量%の範囲であり、第二成分の割合が5重量%から85重量%の範囲であり、そして第三成分の割合が10重量%から90重量%の範囲である請求項4から7のいずれか1項に記載の液晶組成物。
- 第四成分として式(4)で表される化合物の群から選択された少なくとも1つの化合物をさらに含有する、請求項1から8のいずれか1項に記載の液晶組成物。
ここで、R7およびR8は独立して、炭素数1から12のアルキル、炭素数1から12のアルコキシ、または炭素数2から12のアルケニルであり;環Dおよび環Eは独立して、1,4-シクロへキシレンまたは1,4-フェニレンであり;X3およびX4は独立して、フッ素または塩素であり;Y1は、水素またはメチルであり;Z4およびZ5は独立して、単結合、エチレン、またはカルボニルオキシであり;pは、1、2、または3であり、qは0または1であり、そして、pとqの和が2または3である。 - 第四成分が式(4-1)で表される化合物の群から選択された少なくとも1つの化合物である請求項10に記載の液晶組成物。
- 第四成分が式(4-3)で表される化合物の群から選択された少なくとも1つの化合物である請求項10に記載の液晶組成物。
- 液晶組成物の全重量に基づいて、第四成分の割合が5重量%から40重量%の範囲である請求項9から12のいずれか1項に記載の液晶組成物。
- 第五成分が式(5-4)で表される化合物の群から選択された少なくとも1つの化合物である請求項15に記載の液晶組成物。
- 液晶組成物の全重量に基づいて、第五成分の割合が5重量%から30重量%の範囲である請求項14から16のいずれか1項に記載の液晶組成物。
- ネマチック相の上限温度が70℃以上であり、波長589nmにおける光学異方性(25℃)が0.08以上であり、そして周波数1kHzにおける誘電率異方性(25℃)が-2以下である請求項1から17のいずれか1項に記載の液晶組成物。
- 請求項1から18のいずれか1項に記載の液晶組成物を含有する液晶表示素子。
- 液晶表示素子の動作モードが、VAモード、IPSモード、FFSモード、またはPSAモードであり、液晶表示素子の駆動方式がアクティブマトリックス方式である請求項19に記載の液晶表示素子。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020127027188A KR101842723B1 (ko) | 2010-04-22 | 2011-02-18 | 액정 조성물 및 액정 표시 소자 |
| CN201180016722.0A CN102834487B (zh) | 2010-04-22 | 2011-02-18 | 液晶组成物及液晶显示元件 |
| JP2012511576A JP5811087B2 (ja) | 2010-04-22 | 2011-02-18 | 液晶組成物および液晶表示素子 |
| EP11771791.8A EP2562233B1 (en) | 2010-04-22 | 2011-02-18 | Liquid crystal composition and liquid crystal display element |
| US13/634,545 US8795553B2 (en) | 2010-04-22 | 2011-02-18 | Liquid crystal composition and liquid crystal display device |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2010-098998 | 2010-04-22 | ||
| JP2010098998 | 2010-04-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011132451A1 true WO2011132451A1 (ja) | 2011-10-27 |
Family
ID=44833991
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2011/053496 WO2011132451A1 (ja) | 2010-04-22 | 2011-02-18 | 液晶組成物および液晶表示素子 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US8795553B2 (ja) |
| EP (1) | EP2562233B1 (ja) |
| JP (1) | JP5811087B2 (ja) |
| KR (1) | KR101842723B1 (ja) |
| CN (1) | CN102834487B (ja) |
| TW (1) | TWI494419B (ja) |
| WO (1) | WO2011132451A1 (ja) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2607451A1 (de) * | 2011-12-20 | 2013-06-26 | Merck Patent GmbH | Flüssigkristallines Medium |
| KR20140095326A (ko) * | 2013-01-24 | 2014-08-01 | 삼성디스플레이 주식회사 | 액정 표시 장치 및 이의 제조 방법 |
| WO2014155534A1 (ja) * | 2013-03-26 | 2014-10-02 | Dic株式会社 | 液晶組成物及びそれを使用した液晶表示素子 |
| JP2015157948A (ja) * | 2013-03-06 | 2015-09-03 | Dic株式会社 | ネマチック液晶組成物及びこれを用いた液晶表示素子 |
| WO2016013690A1 (ja) * | 2014-07-24 | 2016-01-28 | Dic株式会社 | 液晶組成物及びそれを使用した液晶表示素子 |
| US9683170B2 (en) | 2013-03-26 | 2017-06-20 | Dic Corporation | Liquid crystal composition and liquid crystal display device using the same |
| JPWO2016136344A1 (ja) * | 2015-02-25 | 2017-11-30 | Jnc株式会社 | 液晶組成物および液晶表示素子 |
| EP2703472B1 (de) * | 2012-08-31 | 2018-07-04 | Merck Patent GmbH | Flüssigkristallines Medium |
| JP2018127627A (ja) * | 2012-06-02 | 2018-08-16 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツングMerck Patent Gesellschaft mit beschraenkter Haftung | 液晶媒体 |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102088166B1 (ko) * | 2011-03-29 | 2020-03-12 | 메르크 파텐트 게엠베하 | 액정 매질 |
| TW201323588A (zh) * | 2011-12-05 | 2013-06-16 | Jnc Corp | 液晶組成物與其用途、及液晶顯示元件 |
| JP5679096B1 (ja) * | 2013-03-21 | 2015-03-04 | Dic株式会社 | 液晶組成物及びそれを使用した液晶表示素子 |
| CN105733607B (zh) * | 2013-03-26 | 2018-10-19 | Dic株式会社 | 液晶组合物以及使用其的液晶显示元件和液晶显示器 |
| CN105838387B (zh) * | 2013-03-26 | 2019-03-08 | Dic株式会社 | 液晶组合物、液晶显示元件和液晶显示器 |
| TWI629342B (zh) * | 2013-03-26 | 2018-07-11 | 迪愛生股份有限公司 | Liquid crystal composition and liquid crystal display element using same |
| TWI548725B (zh) * | 2013-03-26 | 2016-09-11 | Dainippon Ink & Chemicals | A liquid crystal composition, and a liquid crystal display device using the liquid crystal display device |
| TWI488948B (zh) * | 2013-03-26 | 2015-06-21 | Dainippon Ink & Chemicals | A liquid crystal composition, and a liquid crystal display device using the liquid crystal display device |
| KR102131117B1 (ko) * | 2014-02-07 | 2020-07-08 | 삼성디스플레이 주식회사 | 편광자, 편광자를 갖는 표시 장치, 및 편광자 제조 방법 |
| CN105940084A (zh) * | 2014-03-07 | 2016-09-14 | 捷恩智株式会社 | 液晶显示元件及液晶组合物 |
| EP3067405B1 (de) * | 2015-03-10 | 2019-03-27 | Merck Patent GmbH | Flüssigkristallines medium |
| CN107267157B (zh) * | 2016-04-08 | 2020-07-31 | 北京八亿时空液晶科技股份有限公司 | 一种负介电各向异性液晶组合物及其应用 |
| WO2018043144A1 (ja) * | 2016-09-01 | 2018-03-08 | Dic株式会社 | 液晶表示素子 |
| CN109437246B (zh) * | 2018-12-24 | 2021-04-30 | 杨志益 | 一种从脱硫废液中回收亚硫酸钠的方法 |
| CN112920810B (zh) * | 2019-12-30 | 2023-04-07 | 石家庄诚志永华显示材料有限公司 | 液晶组合物、液晶显示元件、液晶显示器 |
| CN114806598A (zh) * | 2022-05-20 | 2022-07-29 | 北京八亿时空液晶科技股份有限公司 | 一种具有高对比度的负性液晶组合物及其应用 |
Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3660505A (en) | 1969-08-27 | 1972-05-02 | Exxon Research Engineering Co | Hindered alkenyl phenols from quinone methide |
| JPS5970624A (ja) | 1982-10-13 | 1984-04-21 | Chisso Corp | トランス,トランス−4−アルキル−4′−アルキルビシクロヘキサン類 |
| JPH02503430A (ja) | 1988-03-10 | 1990-10-18 | メルク・パテント・ゲゼルシヤフト・ミツト・ベシユレンクテル・ハフツング | 2,3‐ジフルオロビフエニル化合物 |
| JPH02503441A (ja) | 1988-03-10 | 1990-10-18 | メルク・パテント・ゲゼルシヤフト・ミツト・ベシユレンクテル・ハフツング | ジフルオロベンゼン誘導体 |
| JP2001335586A (ja) * | 2000-05-30 | 2001-12-04 | Chisso Corp | シリコン誘導体、液晶組成物および液晶表示素子 |
| JP2003327965A (ja) | 2002-04-12 | 2003-11-19 | Merck Patent Gmbh | 液晶媒体およびこの媒体を含有する電気光学ディスプレイ |
| JP2005290349A (ja) | 2003-06-30 | 2005-10-20 | Dainippon Ink & Chem Inc | クロマン誘導体及びこの化合物を含有する液晶組成物 |
| JP2008024815A (ja) | 2006-07-20 | 2008-02-07 | Chisso Corp | 液晶組成物および液晶表示素子 |
| JP2008285570A (ja) | 2007-05-17 | 2008-11-27 | Chisso Corp | 液晶組成物および液晶表示素子 |
| JP2009035630A (ja) | 2007-08-01 | 2009-02-19 | Chisso Corp | 液晶組成物および液晶表示素子 |
| WO2009035630A2 (en) | 2007-09-12 | 2009-03-19 | Borgwarner Inc. | Boosting assist electric hybrid combination |
| WO2010016389A1 (ja) * | 2008-08-07 | 2010-02-11 | チッソ株式会社 | 液晶組成物および液晶表示素子 |
| WO2010072370A1 (de) * | 2008-12-22 | 2010-07-01 | Merck Patent Gmbh | Flüssigkristallines medium |
| WO2010119779A1 (ja) * | 2009-04-14 | 2010-10-21 | Dic株式会社 | 重合性化合物を含有する液晶組成物及びそれを使用した液晶表示素子 |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4320824B2 (ja) * | 1998-06-02 | 2009-08-26 | チッソ株式会社 | Δεが負の値を有するアルケニル化合物、液晶組成物および液晶表示素子 |
| US7226644B2 (en) | 2003-06-30 | 2007-06-05 | Dainippon Ink And Chemicals, Inc. | Chroman derivative and liquid-crystal composition containing the compound |
| WO2007108307A1 (ja) * | 2006-03-20 | 2007-09-27 | Chisso Corporation | 液晶組成物および液晶表示素子 |
| WO2008018248A1 (fr) * | 2006-08-07 | 2008-02-14 | Chisso Corporation | Composition de cristaux liquides et élément d'affichage à cristaux liquides |
| JP5374904B2 (ja) * | 2007-04-06 | 2013-12-25 | Jnc株式会社 | アルカジエニル基を有する化合物およびこれを用いた液晶組成物 |
| CN101868439B (zh) * | 2007-09-06 | 2014-02-05 | Jnc株式会社 | 具有侧向氟的4、5环液晶性化合物、液晶组成物以及液晶显示元件 |
| CN101980996B (zh) * | 2008-04-09 | 2013-06-26 | Jnc株式会社 | 含有侧向氟的3环液晶性化合物、液晶组成物及液晶显示元件 |
| TW201004931A (en) * | 2008-06-27 | 2010-02-01 | Chisso Corp | Liquid crystal compound having negative dielectric anisotropy, liquid crystal composition, and liquid crystal display device |
| CN101519594B (zh) * | 2009-04-01 | 2012-10-03 | 晶宝液晶有限公司 | 液晶混合物和含所述液晶混合物的液晶显示器件 |
-
2011
- 2011-02-18 US US13/634,545 patent/US8795553B2/en not_active Expired - Fee Related
- 2011-02-18 WO PCT/JP2011/053496 patent/WO2011132451A1/ja active Application Filing
- 2011-02-18 CN CN201180016722.0A patent/CN102834487B/zh active Active
- 2011-02-18 EP EP11771791.8A patent/EP2562233B1/en active Active
- 2011-02-18 JP JP2012511576A patent/JP5811087B2/ja active Active
- 2011-02-18 KR KR1020127027188A patent/KR101842723B1/ko active IP Right Grant
- 2011-02-24 TW TW100106233A patent/TWI494419B/zh active
Patent Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3660505A (en) | 1969-08-27 | 1972-05-02 | Exxon Research Engineering Co | Hindered alkenyl phenols from quinone methide |
| JPS5970624A (ja) | 1982-10-13 | 1984-04-21 | Chisso Corp | トランス,トランス−4−アルキル−4′−アルキルビシクロヘキサン類 |
| JPH02503430A (ja) | 1988-03-10 | 1990-10-18 | メルク・パテント・ゲゼルシヤフト・ミツト・ベシユレンクテル・ハフツング | 2,3‐ジフルオロビフエニル化合物 |
| JPH02503441A (ja) | 1988-03-10 | 1990-10-18 | メルク・パテント・ゲゼルシヤフト・ミツト・ベシユレンクテル・ハフツング | ジフルオロベンゼン誘導体 |
| JP2001335586A (ja) * | 2000-05-30 | 2001-12-04 | Chisso Corp | シリコン誘導体、液晶組成物および液晶表示素子 |
| JP2003327965A (ja) | 2002-04-12 | 2003-11-19 | Merck Patent Gmbh | 液晶媒体およびこの媒体を含有する電気光学ディスプレイ |
| JP2005290349A (ja) | 2003-06-30 | 2005-10-20 | Dainippon Ink & Chem Inc | クロマン誘導体及びこの化合物を含有する液晶組成物 |
| JP2008024815A (ja) | 2006-07-20 | 2008-02-07 | Chisso Corp | 液晶組成物および液晶表示素子 |
| JP2008285570A (ja) | 2007-05-17 | 2008-11-27 | Chisso Corp | 液晶組成物および液晶表示素子 |
| JP2009035630A (ja) | 2007-08-01 | 2009-02-19 | Chisso Corp | 液晶組成物および液晶表示素子 |
| WO2009035630A2 (en) | 2007-09-12 | 2009-03-19 | Borgwarner Inc. | Boosting assist electric hybrid combination |
| WO2010016389A1 (ja) * | 2008-08-07 | 2010-02-11 | チッソ株式会社 | 液晶組成物および液晶表示素子 |
| WO2010072370A1 (de) * | 2008-12-22 | 2010-07-01 | Merck Patent Gmbh | Flüssigkristallines medium |
| WO2010119779A1 (ja) * | 2009-04-14 | 2010-10-21 | Dic株式会社 | 重合性化合物を含有する液晶組成物及びそれを使用した液晶表示素子 |
Non-Patent Citations (4)
| Title |
|---|
| "Comprehensive Organic Synthesis", PERGAMON PRESS |
| "New Experimental Chemistry Course", MARUZEN CO., LTD. |
| "Organic Reactions", JOHN WILEY & SONS, INC. |
| "Organic Syntheses", JOHN WILEY & SONS, INC. |
Cited By (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102193673B1 (ko) | 2011-12-20 | 2020-12-21 | 메르크 파텐트 게엠베하 | 액정 매질 |
| JP2017171911A (ja) * | 2011-12-20 | 2017-09-28 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツングMerck Patent Gesellschaft mit beschraenkter Haftung | 液晶媒体 |
| US10072209B2 (en) | 2011-12-20 | 2018-09-11 | Merck Patent Gmbh | Liquid-crystalline media |
| CN106520141A (zh) * | 2011-12-20 | 2017-03-22 | 默克专利股份有限公司 | 液晶介质 |
| GB2514272B (en) * | 2011-12-20 | 2016-02-10 | Merck Patent Gmbh | Liquid-crystalline medium |
| US11377596B2 (en) | 2011-12-20 | 2022-07-05 | Merck Patent Gmbh | Liquid-crystalline media |
| GB2514272A (en) * | 2011-12-20 | 2014-11-19 | Merck Patent Gmbh | Liquid-crystalline medium |
| EP2607451A1 (de) * | 2011-12-20 | 2013-06-26 | Merck Patent GmbH | Flüssigkristallines Medium |
| KR102353429B1 (ko) * | 2011-12-20 | 2022-01-20 | 메르크 파텐트 게엠베하 | 액정 매질 |
| KR20200143322A (ko) * | 2011-12-20 | 2020-12-23 | 메르크 파텐트 게엠베하 | 액정 매질 |
| JP2013144796A (ja) * | 2011-12-20 | 2013-07-25 | Merck Patent Gmbh | 液晶媒体 |
| US9982194B2 (en) | 2011-12-20 | 2018-05-29 | Mereck Patent Gmbh | Liquid-crystalline media |
| JP2019163472A (ja) * | 2011-12-20 | 2019-09-26 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツングMerck Patent Gesellschaft mit beschraenkter Haftung | 液晶媒体 |
| CN106544041A (zh) * | 2011-12-20 | 2017-03-29 | 默克专利股份有限公司 | 液晶介质 |
| KR20130071393A (ko) * | 2011-12-20 | 2013-06-28 | 메르크 파텐트 게엠베하 | 액정 매질 |
| JP2017160436A (ja) * | 2011-12-20 | 2017-09-14 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツングMerck Patent Gesellschaft mit beschraenkter Haftung | 液晶媒体 |
| JP2018127627A (ja) * | 2012-06-02 | 2018-08-16 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツングMerck Patent Gesellschaft mit beschraenkter Haftung | 液晶媒体 |
| EP2703472B1 (de) * | 2012-08-31 | 2018-07-04 | Merck Patent GmbH | Flüssigkristallines Medium |
| KR102092042B1 (ko) * | 2013-01-24 | 2020-03-24 | 삼성디스플레이 주식회사 | 액정 표시 장치 및 이의 제조 방법 |
| KR20140095326A (ko) * | 2013-01-24 | 2014-08-01 | 삼성디스플레이 주식회사 | 액정 표시 장치 및 이의 제조 방법 |
| JP2014142585A (ja) * | 2013-01-24 | 2014-08-07 | Samsung Display Co Ltd | 液晶表示装置 |
| JP2015157948A (ja) * | 2013-03-06 | 2015-09-03 | Dic株式会社 | ネマチック液晶組成物及びこれを用いた液晶表示素子 |
| JP2015180727A (ja) * | 2013-03-06 | 2015-10-15 | Dic株式会社 | ネマチック液晶組成物及びこれを用いた液晶表示素子 |
| US9683170B2 (en) | 2013-03-26 | 2017-06-20 | Dic Corporation | Liquid crystal composition and liquid crystal display device using the same |
| WO2014155534A1 (ja) * | 2013-03-26 | 2014-10-02 | Dic株式会社 | 液晶組成物及びそれを使用した液晶表示素子 |
| US11466211B2 (en) | 2013-03-26 | 2022-10-11 | Dic Corporation | Liquid crystal composition and liquid crystal display element including the same |
| JP2016023292A (ja) * | 2014-07-24 | 2016-02-08 | Dic株式会社 | 液晶組成物及びそれを使用した液晶表示素子 |
| WO2016013690A1 (ja) * | 2014-07-24 | 2016-01-28 | Dic株式会社 | 液晶組成物及びそれを使用した液晶表示素子 |
| JPWO2016136344A1 (ja) * | 2015-02-25 | 2017-11-30 | Jnc株式会社 | 液晶組成物および液晶表示素子 |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2011132451A1 (ja) | 2013-07-18 |
| TWI494419B (zh) | 2015-08-01 |
| CN102834487B (zh) | 2015-04-22 |
| KR20130079343A (ko) | 2013-07-10 |
| KR101842723B1 (ko) | 2018-03-27 |
| JP5811087B2 (ja) | 2015-11-11 |
| EP2562233A1 (en) | 2013-02-27 |
| US8795553B2 (en) | 2014-08-05 |
| EP2562233A4 (en) | 2015-12-23 |
| EP2562233B1 (en) | 2019-07-17 |
| CN102834487A (zh) | 2012-12-19 |
| TW201213518A (en) | 2012-04-01 |
| US20130001469A1 (en) | 2013-01-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5811087B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5874641B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5790647B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5861633B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5636954B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5609649B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5531632B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5494486B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5500176B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5444723B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5522407B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5609650B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5609647B2 (ja) | 液晶組成物および液晶表示素子 | |
| WO2012053323A1 (ja) | 液晶組成物および液晶表示素子 | |
| WO2011040170A1 (ja) | 液晶組成物および液晶表示素子 | |
| WO2010029843A1 (ja) | 液晶組成物および液晶表示素子 | |
| WO2011158820A1 (ja) | 液晶組成物および液晶表示素子 | |
| WO2010016389A1 (ja) | 液晶組成物および液晶表示素子 | |
| WO2010032587A1 (ja) | 液晶組成物および液晶表示素子 | |
| WO2010016387A1 (ja) | 液晶組成物および液晶表示素子 | |
| WO2009125669A1 (ja) | 液晶組成物および液晶表示素子 | |
| JP5761021B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5228682B2 (ja) | 液晶組成物および液晶表示素子 | |
| JP5761022B2 (ja) | 液晶組成物および液晶表示素子 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201180016722.0 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11771791 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012511576 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011771791 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13634545 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 20127027188 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |