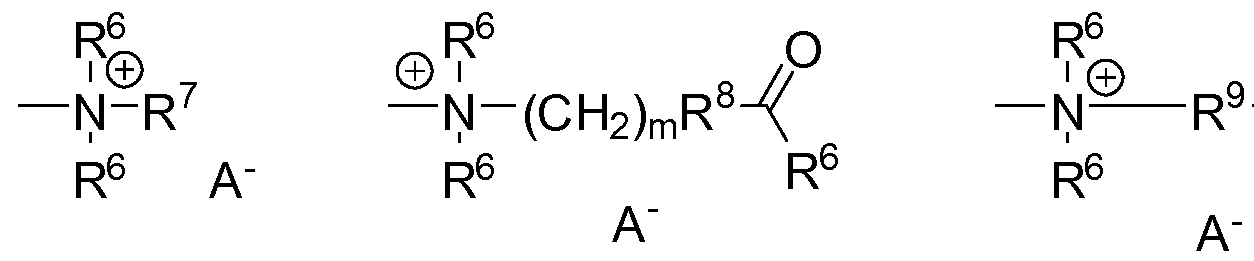WO2011032797A1 - Neuartige polysiloxane mit quaternären ammoniumgruppen und deren verwendung - Google Patents
Neuartige polysiloxane mit quaternären ammoniumgruppen und deren verwendung Download PDFInfo
- Publication number
- WO2011032797A1 WO2011032797A1 PCT/EP2010/061968 EP2010061968W WO2011032797A1 WO 2011032797 A1 WO2011032797 A1 WO 2011032797A1 EP 2010061968 W EP2010061968 W EP 2010061968W WO 2011032797 A1 WO2011032797 A1 WO 2011032797A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- independently
- branched
- different
- polysiloxanes
- formula
- Prior art date
Links
- JDVPNEBUTKFJQG-UHFFFAOYSA-N CCC(CC1C2)C2C(C)C1O Chemical compound CCC(CC1C2)C2C(C)C1O JDVPNEBUTKFJQG-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/38—Polysiloxanes modified by chemical after-treatment
- C08G77/382—Polysiloxanes modified by chemical after-treatment containing atoms other than carbon, hydrogen, oxygen or silicon
- C08G77/388—Polysiloxanes modified by chemical after-treatment containing atoms other than carbon, hydrogen, oxygen or silicon containing nitrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/38—Polysiloxanes modified by chemical after-treatment
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/37—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/643—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicon in the main chain
- D06M15/6436—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicon in the main chain containing amino groups
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/37—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/643—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicon in the main chain
- D06M15/647—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicon in the main chain containing polyether sequences
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2200/00—Functionality of the treatment composition and/or properties imparted to the textile material
- D06M2200/50—Modified hand or grip properties; Softening compositions
Definitions
- the invention relates to novel multi-branched polysiloxanes with quaternary ammonium groups. It further relates to the use of these polymers as plasticizers for fabrics such as tissue, tissue, non-wovens and / or fibers of natural and / or synthetic raw materials and / or leather.
- Plasticisers for fabrics, woven fabrics, knitted fabrics, non-wovens and / or fibers of natural and / or synthetic raw materials are fabrics which give textile materials a soft, supple feel.
- Particularly suitable are polysiloxanes with quaternary ammonium groups. Via electrostatic attractions, the ionic groups anchor the siloxane to the fiber. In this way the friction is reduced and the desired softening effect is achieved.
- the siloxane when applied in the form of microemulsions, the siloxane can penetrate the fiber, giving it inner softness and fullness.
- This invention has the object zugru hands to find polysiloxanes with quaternary ammonium groups, which can be prepared in good yields and beyond are particularly well suited as hydrophilic softeners for textiles and also can not be easily washed down by a textile.
- the object according to the invention is achieved by branched organomodified polysiloxanes which have more than three quaternary ammonium groups.
- the branched organomodified polysiloxanes according to the invention (referred to as acid oxa nates) give a very good hydrophilic soft feel and an increased permanence on textiles.
- a high elasticity and an improved anti-wrinkling property of such a product should be considered as another positive feature.
- branched organomodified polysiloxanes are able to solve the problem.
- the object is achieved by branched organomodified polysiloxanes having more than three quaternary ammonium groups.
- Another object of the invention are branched organomodified polysiloxanes of general formula I.
- R 1 independently of one another identical or different linear or branched alkyl, aryl or alkaryl radicals having 1 to 30
- organic radicals carry the ammonium functions
- radicals selected from the group:
- Nitrogen groups preferably selected anions from the inorganic or organic acids HA generally known in the state of the art, more preferably from acetic acid, lactic acid, aromatic carboxylic acids or HCl,
- R 6 independently the same or different
- R 7 independently of one another, identical or different
- Alkyl radicals having 1 to 30 carbon atoms preferably stearyl, hexadecyl, dodecyl, undecylene, octyl, ethyl or
- Methyl radicals, R 8 independently the same or different radicals selected from the group O, NH or NR 6 ,
- Polyether radicals in particular polyether radicals of the general formula III: - [CH 2 CH 2 O] n [CH 2 CH (CH 3) O] o [CH (CH 3) CH 2 O] pR
- n, o, p are independently 0 to 100
- R 11 independently of one another identical or different radicals from the group -COR 1 , R 1 or H
- R 3 independently the same or different
- Polyether radicals in particular identical or different
- Ph phenyl, with the proviso that the sum of g and h is greater than 1, preferably the sum of g and h is greater than or equal to 2,
- the sum of g and h is greater than or equal to 3 and the further proviso that the sum of b and e is greater than 3, preferably the sum of b and e is greater than or equal to 3.5, more preferably the sum of b and e is greater than or equal to 4;
- siloxane Polymers hereinafter also referred to as siloxane quat (s)
- siloxane quat differ from those of the prior art in that they have a siloxane backbone with more than one branching unit.
- Such products have a surprisingly good suitability as a softener for textiles.
- branching unit they are fluid and can be well formulated.
- Their softness is much better than purely side-modified siloxanes. In comparison to the ⁇ , ⁇ -modified linear siloxanes, they have a significantly improved permanence, so that they have an improved overall property profile.
- branched SiH siloxane is prepared by equilibrating tri- or tetraalkoxysilanes with cyclic siloxanes and ⁇ , ⁇ -SiH siloxanes and hydrolyzing the alkoxy groups in the presence of water.
- branched SiH-functional siloxanes are formed in the siloxane moiety.
- this process is described in detail in WO 2009/065644.
- the content of the above-cited patent literature relating to the preparation of branched SiH siloxanes is hereby incorporated by reference and is considered part of the disclosure of the present application.
- double bond-containing epoxides e.g., allyl glycidyl ether
- the epoxy-functional siloxanes thus prepared can then be reacted with tertiary amines to give the corresponding branched polysiloxanes of the invention having quaternary ammonium groups.
- Another object of the invention is therefore a process for the preparation of branched polysiloxanes having quaternary ammonium groups of general formula I, wherein in a first step a) a branched SiH siloxane is prepared by tri ⁇ or tetraalkoxysilanes with cyclic siloxanes and ⁇ , ⁇ - SiH siloxanes are equilibrated and in the presence of water, the alkoxy groups are hydrolyzed and in a second step during
- double bond-containing epoxies e.g.
- Another object of the invention is the use of the compounds of the invention, obtainable according to the said Process as an optionally permanent plasticizer for fabrics such as woven, knitted, nonwovens, tissue (paper fiber) and / or fibers of natural and / or synthetic raw materials and / or leather, wherein the plasticizer may optionally impart hydrophilic properties to the fabrics treated therewith ,
- fabrics such as woven, knitted, non-woven, tissue (paper) and / or natural fibers and / or synthetic raw materials and / or leather.
- fabrics such as woven, knitted, non-woven, tissue (paper) and / or natural fibers and / or synthetic raw materials and / or leather.
- such compounds have a high silicone character in order to produce a pleasant feel, and on the other hand they have a viscosity which makes it possible to formulate such compounds in aqueous form.
- the viscosity of the polysiloxanes of the formula I is in the range up to about 25,000 mPa * s at 25 ° C.
- the content of the compound of general formula I used in the aqueous formulations according to the invention is between 0.5 and 99 wt .-%, preferably between 3 and 70 wt .-%, in particular between 5 and 50 wt .-%, based on the entire formulation.
- the branched polysiloxanes according to the invention can be used in the form of concentrates, compounds / emulsion concentrates, formulations and liquors prepared therefrom and optionally applied. It is known to the person skilled in the art that the compounds are present in the form of a mixture with a distribution of the indices mentioned essentially regulated by statistical laws. Fleet stands for a mostly aqueous liquid in which textiles are washed, bleached, dyed or impregnated.
- liquor means the entirety of solvent (usually water) and all contained therein (dissolved, emulsified or dispersed) components such as dyes, emulsifiers and other auxiliaries.
- the total of the components dissolved in the liquor are commonly referred to as solids, the solids content indicating the residue after evaporation of the volatiles (at about 100 ° C-105 ° C).
- the amount of components of a liquor is usually given in g / l for liquids or% (based on the weight of the goods).
- a treatment liquor as the bath (mostly aqueous) in which (or with) the fabric is provided with one or more (surface-active) substances.
- Compound or emulsion concentrate contains 50-90 wt .-%, preferably 50-80 wt .-% of the polysiloxane compound containing as further constituents water and / or solvent selected from the group of glycols, unbranched and / or branched alcohols and / or alkyl ethers having 1 to 6 carbon atoms and optionally one or more non-ionic emulsifiers, for example an alcohol ethoxylate containing 3-25 ethylene oxide units.
- Compounds / emulsion concentrates are usually soluble or self-emulsifiable in water.
- Formulations and / or (aqueous) emulsions contain 5-20% by weight of the polysiloxane according to the invention, solvents, emulsifiers (also cationic or amphoteric), water.
- the solids content of these formulations or emulsions is usually about 10-40 wt .-%.
- the (application) liquors (application / finishing baths) are prepared by diluting in water in the manufacturing plants / equipment enterprises. Typical liquor concentrations in the padding application are, for example, 5-8 g formulation / emulsion per liter of liquor solution or application liquor.
- polysiloxanes with quaternary ammonium groups available on the market are not self-emulsifiable in water and can only be introduced into an aqueous formulation by adding excipients of emulsifiers and / or solvents.
- emulsifiers fatty alcohol ethoxylates with degrees of ethoxylation between 3 and 12 and in a ratio of plasticizer to fatty alcohol ethoxylate of 5: 1 to 1: 1 are typically used.
- the solvents used are, for example, high-boiling glycols such as dipropylene glycol or butyldiglycol.
- branched polysiloxanes of the invention having quaternary ammonium groups processes for their preparation and their use are described below by way of example, without the invention being restricted to these exemplary embodiments.
- index numbers reproduced in the formulas given here and the value ranges of the specified indices are therefore also understood as the average values of the possible statistical distribution of the actual structures present and / or their mixtures. This also applies to as such per se exactly reproduced structural formulas, such as for formula I.
- the SiH functions are reacted with a 3-5% by weight butanolic sodium butylate solution and the resulting hydrogen is collected in a burette. From the volume of hydrogen formed, the content of silicon bound hydrogen can be determined. Determination of the epoxy number:
- a quantity of sample is weighed into the Ehenmeyer flasks depending on the expected epoxy value. 40 ml of a 0.1 mol / L hydrochloric acid solution in dioxane are then pipetted in with the solid pipette. The amount of sample should be chosen such that the amount of hydrochloric acid used is present in significant excess relative to the epoxy functions. The flask is closed and swirled until the sample has completely dissolved. The samples are then allowed to react for 15 minutes at room temperature. Then, make up to a volume of about 100 ml with ethanol. After adding a few drops of the cresol indicator solution in THF is against 0.1 mol / L ethanolic potassium hydroxide solution titrated.
- 1st stage 28.5 g of methyltriethoxysilane, 1067 g of decamethyltetracyclosiloxane, and 105 g of an ⁇ , w-modified SiH siloxane having the formula HSiMe 2 O [SiMe 2 O] 8 SiMe 2 H were mixed with 1.2 g of trifluoromethanesulfonic acid and stirred for two hours at 40 ° C. Subsequently, 8.6 g of water were added and allowed to react for one hour.
- 2nd step 333 g of the SiH siloxane obtained in the first step were mixed with 13.6 g of 2 - [(allyloxy) methyl] oxirane (allylglycidyl ether) and heated to 70.degree. Subsequently, 10 ppm of Pt were added in the form of the Karstedt catalyst. It initiated an exothermic reaction in which the reaction heated to 84 ° C. It was then heated to 90 ° C and stirred for two hours. Thereafter, the volatile constituents of the reaction mixture were removed at 130 ° C in an oil pump vacuum. It a liquid, clear, slightly brownish colored epoxysiloxane having an epoxy oxygen content of 0.44 wt.% was obtained.
- 3rd stage 32 g of isopropanol, 1.4 g of acetic acid and 7.3 g of a coconut fatty acid-based amide amine (CAS: 61790-62-3) were mixed together at room temperature. Subsequently, 147 g of the epoxysiloxane obtained in the second stage were added slowly and the reaction mixture was heated to 80.degree. It was stirred for six hours. Subsequently, the volatiles were removed at 100 ° C in an oil pump vacuum to give a clear, yellowish colored product having a viscosity of 1,900 mPa * s and a nitrogen content of 0.19 wt .-%.
- a commercially available microemulsion of an unbranched, linear polysiloxane having quaternary ammonium groups Tegopren ® 6924, having a solids content of 20 wt .-%.
- Formulation 5 - not of the invention A commercially available dispersion of an organic plasticizer, Rewoquat ® WE 18, having a solids content of 7 wt .-%.
- This plasticizer is silicon-free and is a typical ester quat.
- the test method based on DIN 53924 was used to measure the rise height of water.
- the finished cotton test fabric is cut into five 25 cm long and 1.5 cm wide strips, marked laterally with a water-soluble pencil and fastened to a support vertically tightly but without tension.
- the holder is then placed in a water basin for five minutes so that 2 cm of the strip dip into the water.
- the water-soluble marking serves to better ascertain the rise height by the color of the paint when wetted with water.
- the washes were washed in a commercially available washing machine, Miele Novotronic W 918 with colored laundry without prewashing at 40 ° C with wfk standard detergent IECA-Base and 3 kg BW ballast fabric. Finally, the thus treated fabric was dried at room temperature for 12 hours.
- Polyether modification in Formulation 2 versus Formulation 1 shows improved water absorption of 88.3% versus 84.0% of the treated fabric for excellent grip.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Silicon Polymers (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MX2012002890A MX2012002890A (es) | 2009-09-15 | 2010-08-17 | Polisiloxanos nuevos teniendo grupos de amonio cuaternarios y uso de estos. |
| US13/389,854 US8916511B2 (en) | 2009-09-15 | 2010-08-17 | Polysiloxanes having quaternary ammonium groups and use thereof |
| CN201080041059.5A CN102549046B (zh) | 2009-09-15 | 2010-08-17 | 具有季铵基团的新型聚硅氧烷及其用途 |
| EP10744925A EP2478036A1 (de) | 2009-09-15 | 2010-08-17 | Neuartige polysiloxane mit quaternären ammoniumgruppen und deren verwendung |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102009029450.3 | 2009-09-15 | ||
| DE200910029450 DE102009029450A1 (de) | 2009-09-15 | 2009-09-15 | Neuartige Polysiloxane mit quaternären Ammoniumgruppen und deren Verwendung |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011032797A1 true WO2011032797A1 (de) | 2011-03-24 |
Family
ID=42734728
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2010/061968 WO2011032797A1 (de) | 2009-09-15 | 2010-08-17 | Neuartige polysiloxane mit quaternären ammoniumgruppen und deren verwendung |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US8916511B2 (de) |
| EP (1) | EP2478036A1 (de) |
| CN (1) | CN102549046B (de) |
| DE (1) | DE102009029450A1 (de) |
| MX (1) | MX2012002890A (de) |
| WO (1) | WO2011032797A1 (de) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013032493A1 (en) * | 2011-08-26 | 2013-03-07 | Colgate-Palmolive Company | Fabric wrinkle reduction composition |
| US20140134125A1 (en) * | 2011-06-30 | 2014-05-15 | Evonik Degussa Gmbh | Microemulsion of polysiloxanes containing quaternary ammonium groups, production and use thereof |
Families Citing this family (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102012202521A1 (de) | 2012-02-20 | 2013-08-22 | Evonik Goldschmidt Gmbh | Verzweigte Polysiloxane und deren Verwendung |
| DE102012203737A1 (de) | 2012-03-09 | 2013-09-12 | Evonik Goldschmidt Gmbh | Modifizierte Alkoxylierungsprodukte, die zumindest eine nicht-terminale Alkoxysilylgruppe aufweisen und mehrere Urethangruppen enthalten und deren Verwendung |
| BR112015014664B1 (pt) * | 2012-12-21 | 2021-02-23 | Colgate-Palmolive Company | condicionador de tecido |
| JP6046505B2 (ja) * | 2013-01-29 | 2016-12-14 | 株式会社ダイセル | シート状モールド及びその製造方法並びにその用途 |
| DE102013216777A1 (de) | 2013-08-23 | 2015-02-26 | Evonik Industries Ag | Bei Raumtemperatur härtbare Silikonharz-Zusammensetzungen |
| DE102013216781A1 (de) | 2013-08-23 | 2015-02-26 | Evonik Industries Ag | Beschichtungsmassen |
| DE102013216787A1 (de) | 2013-08-23 | 2015-02-26 | Evonik Degussa Gmbh | Guanidingruppen aufweisende semi-organische Siliciumgruppen enthaltende Verbindungen |
| DE102013216751A1 (de) | 2013-08-23 | 2015-02-26 | Evonik Industries Ag | Modifizierte Alkoxylierungsprodukte, die Alkoxysilylgruppen aufweisen und Urethangruppen enthalten und deren Verwendung |
| DE102014217790A1 (de) | 2014-09-05 | 2016-03-10 | Evonik Degussa Gmbh | Verfahren zur Herstellung von hydrosilylierbaren Eugenol-Polyethern und Eugenol-Polyethersiloxanen sowie deren Verwendung |
| US20160145483A1 (en) * | 2014-11-26 | 2016-05-26 | Schlumberger Technology Corporation | Well treatment |
| EP3029087A1 (de) | 2014-12-05 | 2016-06-08 | Evonik Degussa GmbH | Verfahren zur Herstellung von niedrigviskosen Polyethersiloxanen |
| EP3061442A1 (de) * | 2015-02-27 | 2016-08-31 | Evonik Degussa GmbH | Zusammensetzung enthaltend Rhamnolipid und Siloxan |
| EP3106033B1 (de) | 2015-06-16 | 2017-04-26 | Evonik Degussa GmbH | Biologisch abbaubares superspreitendes organomodifiziertes trisiloxan |
| DE102015222139A1 (de) * | 2015-11-10 | 2017-05-11 | Wacker Chemie Ag | Verfahren zur Imprägnierung von Textilien mit Zusammensetzungen enthaltend Alkoxypolysiloxane |
| EP3360917A1 (de) | 2017-02-14 | 2018-08-15 | CHT R. BEITLICH GmbH | Bodenkonditionierer für die reduzierung der sodizität und dispersion zur verbesserung der wassermobilität in verschiedenen böden |
| PL3401353T3 (pl) * | 2017-05-08 | 2021-12-13 | Evonik Operations Gmbh | Mieszaniny cyklicznych, rozgałęzionych siloksanów typu D/T i ich dalszych produktów |
| PL3774995T3 (pl) * | 2018-04-03 | 2023-11-06 | Evonik Operations Gmbh | Siloksany do obróbki tekstyliów oraz do zastosowania ich w formulacjach czyszczących i pielęgnacyjnych |
| EP3954740A1 (de) | 2020-08-14 | 2022-02-16 | Evonik Operations GmbH | Entschäumerzusammensetzung auf basis von organofunktionell modifizierten polysiloxanen |
| CN113197792B (zh) * | 2021-04-23 | 2022-03-15 | 广东赛安特新材料有限公司 | 一种聚硅氧烷季铵盐微乳液及其制备方法与应用 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE1493384A1 (de) | 1964-07-14 | 1969-01-30 | Union Carbide Corp | Verfahren zur Herstellung von Organosiliciumverbindungen |
| EP0294642A2 (de) | 1987-06-06 | 1988-12-14 | Th. Goldschmidt AG | Diquaternäre Polysiloxane, deren Herstellung und Verwendung in kosmetischen Zubereitungen |
| WO2003078504A1 (de) * | 2002-03-20 | 2003-09-25 | Ge Bayer Silicones Gmbh & Co. Kg | Verzweigte polyorganosiloxanpolymere |
| EP1561770A1 (de) * | 2004-02-09 | 2005-08-10 | Schill + Seilacher "Struktol" Aktiengesellschaft | Verzweigte Polyorganosiloxane mit quarternären Ammoniumgruppen |
| DE102004002208A1 (de) * | 2004-01-15 | 2005-08-18 | Rudolf Gmbh & Co. Kg Chemische Fabrik | Zubereitungen auf Basis von Organopolysiloxan-Polyammonium-Blockcopolymeren und deren Anwendung auf textilen Substraten |
| EP1887024A1 (de) * | 2006-07-31 | 2008-02-13 | Evonik Goldschmidt GmbH | Neuartige Polysiloxane mit quaternären Ammoniumgruppen, Verfahren zu deren Herstellung und deren Verwendung in reinigenden und pflegenden Formulierungen |
| WO2009065644A1 (de) | 2007-11-21 | 2009-05-28 | Evonik Goldschmidt Gmbh | Verfahren zur herstellung verzweigter sih-funktioneller polysiloxane und deren verwendung zur herstellung sic- und sioc-verknüpfter, verzweigter organomodifizierter polysiloxane |
Family Cites Families (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2760970B1 (fr) * | 1997-03-18 | 2000-03-10 | Oreal | Nanoemulsions a base de lipides amphiphiles non-ioniques et de silicones aminees et utilisations |
| FR2774902B1 (fr) * | 1998-02-13 | 2002-07-19 | Oreal | Composition cosmetique a base de polyurethannes associatifs et de silicones quaternaires |
| EP1000959B1 (de) * | 1998-11-14 | 2003-04-16 | Goldschmidt AG | Polyetherquatfunktionelle Polysiloxane |
| DE102005001039B4 (de) | 2005-01-07 | 2017-11-09 | Evonik Degussa Gmbh | Verfahren zur Herstellung von Äquilibrierungsprodukten von Organosiloxanen und die so erhältlichen Organopolysiloxane |
| DE102005001041A1 (de) | 2005-01-07 | 2006-07-20 | Goldschmidt Gmbh | Neuartige Siloxanblockcopolymere |
| DE102005004706A1 (de) | 2005-02-02 | 2006-08-10 | Goldschmidt Gmbh | UV-Licht absorbierende quaternäre Polysiloxane |
| DE102005004704A1 (de) | 2005-02-02 | 2006-08-10 | Goldschmidt Gmbh | Guanidinogruppen-haltige Siloxane und deren Verwendung für kosmetische Formulierungen |
| DE102005043742A1 (de) | 2005-09-14 | 2007-03-22 | Goldschmidt Gmbh | Verwendung von Epoxy-funktionellen Silanen als Haftungsadditiv für kationisch strahlenhärtende Silikontrennbeschichtungen |
| DE102005051939A1 (de) | 2005-10-29 | 2007-05-03 | Goldschmidt Gmbh | Verfahren zur Herstellung von organisch modifizierten Polyorganosiloxanen |
| DE102006008387A1 (de) | 2006-02-21 | 2007-08-30 | Goldschmidt Gmbh | Verfahren zur Herstellung von siloxanhaltigen Trennbeschichtungen |
| DE102006027339A1 (de) | 2006-06-13 | 2007-12-20 | Goldschmidt Gmbh | Kationisch strahlenhärtende Controlled Release Beschichtungsmassen |
| DE102006035511A1 (de) | 2006-07-31 | 2008-02-07 | Evonik Goldschmidt Gmbh | Neuartige Polysiloxane mit quaternären Ammoniumgruppen, Verfahren zu deren Herstellung und deren Verwendung als Textilweichmacher |
| DE102006041088A1 (de) | 2006-09-01 | 2008-03-06 | Evonik Goldschmidt Gmbh | Siliconhaltige, blockweise aufgebaute Pfropfmischpolymere |
| DE102006041971A1 (de) | 2006-09-07 | 2008-03-27 | Evonik Goldschmidt Gmbh | Verwendung von partikulären Emulgatoren in abhäsiven siloxanhaltigen Beschichtungsmassen |
| DE102006042338A1 (de) | 2006-09-08 | 2008-03-27 | Evonik Goldschmidt Gmbh | Verwendung von Urethan- oder Harnstoffgruppen enthaltenden Polyethern zur Stabilisierung von Polyurethanschäumen |
| US8302691B2 (en) * | 2007-01-19 | 2012-11-06 | Halliburton Energy Services, Inc. | Methods for increasing gas production from a subterranean formation |
| DE102007041028A1 (de) | 2007-08-29 | 2009-03-05 | Evonik Goldschmidt Gmbh | Verwendung estermodifizierter Organopolysiloxane zur Herstellung kosmetischer oder pharmazeutischer Kompositionen |
| DE102008000243A1 (de) | 2008-02-06 | 2009-08-13 | Evonik Goldschmidt Gmbh | Neuartige Kompatibilisierungsmittel zur Verbesserung der Lagerstabilität von Polyolmischungen |
| DE102008001788A1 (de) | 2008-05-15 | 2009-11-26 | Evonik Goldschmidt Gmbh | Verwendung organomodifizierter Siloxanblockcopolymere zur Herstellung kosmetischer oder pharmazeutischer Zusammensetzungen |
| DE102008001786A1 (de) | 2008-05-15 | 2009-11-26 | Evonik Goldschmidt Gmbh | Verwendung organomodifizierter Siloxanblockcopolymere als Pflegewirkstoff zur Pflege von menschlichen oder tierischen Körperteilen |
| DE102008040986A1 (de) | 2008-08-05 | 2010-02-11 | Evonik Goldschmidt Gmbh | Hydrophobierung von Bauelementen aus Mineralfasern |
| DE102008041020A1 (de) | 2008-08-06 | 2010-02-11 | Evonik Goldschmidt Gmbh | Verwendung von Polysiloxanen mit quaternären Ammoniumgruppen zum Schutz von tierischen oder menschlichen Haaren vor Hitzeschädigung |
| DE102008041601A1 (de) | 2008-08-27 | 2010-03-04 | Evonik Goldschmidt Gmbh | Verfahren zur Herstellung verzweigter SiH-funtioneller Polysiloxane und deren Verwendung zur Herstellung flüssiger, SiC- oder SiOC-verknüpfter, verzweigter organomodifizierter Polysiloxane |
| DE102008041754A1 (de) | 2008-09-02 | 2010-03-04 | Evonik Goldschmidt Gmbh | Enzympräparate |
| DE102008042381A1 (de) | 2008-09-26 | 2010-04-01 | Evonik Goldschmidt Gmbh | Emulgator-Systeme für kosmetische und pharmazeutische Öl-in-Wasser-Emulsionen |
| CN102159071A (zh) | 2008-10-17 | 2011-08-17 | 赢创高施米特有限公司 | 含有具有高有机硅特征的烷基聚硅氧烷佐剂的农用化学油组合物 |
| DE102009015211A1 (de) | 2009-03-31 | 2010-10-14 | Evonik Goldschmidt Gmbh | Selbstvernetzende Polysiloxane in Beschichtungen von Enzymimmobilisaten |
| WO2010112683A1 (fr) * | 2009-03-31 | 2010-10-07 | L'oreal | Composition capillaire de traitement des cheveux |
| DE102009002415A1 (de) | 2009-04-16 | 2010-10-21 | Evonik Goldschmidt Gmbh | Emulgator enthaltend glycerinmodifizierte Organopolysiloxane |
| DE102009028640A1 (de) | 2009-08-19 | 2011-02-24 | Evonik Goldschmidt Gmbh | Härtbare Masse enthaltend Urethangruppen aufweisende silylierte Polymere und deren Verwendung in Dicht- und Klebstoffen, Binde- und/oder Oberflächenmodifizierungsmitteln |
| DE102009028636A1 (de) | 2009-08-19 | 2011-02-24 | Evonik Goldschmidt Gmbh | Neuartige Urethangruppen enthaltende silylierte Präpolymere und Verfahren zu deren Herstellung |
| DE102010002178A1 (de) | 2010-02-22 | 2011-08-25 | Evonik Goldschmidt GmbH, 45127 | Verfahren zur Herstellung von Amin-Amid-funktionellen Siloxanen |
| DE102010002180A1 (de) | 2010-02-22 | 2011-08-25 | Evonik Goldschmidt GmbH, 45127 | Stickstoffhaltige silizium-organische Pfropfmischpolymere |
| DE102010031087A1 (de) | 2010-07-08 | 2012-01-12 | Evonik Goldschmidt Gmbh | Neuartige polyestermodifizierte Organopolysiloxane |
| DE102010038774A1 (de) | 2010-08-02 | 2012-02-02 | Evonik Goldschmidt Gmbh | Modifizierte Alkoxylierungsprodukte, die zumindest eine nicht-terminale Alkoxysilylgruppe aufweisen, mit erhöhter Lagerstabilität und erhöhter Dehnbarkeit der unter deren Verwendung hergestellten Polymere |
-
2009
- 2009-09-15 DE DE200910029450 patent/DE102009029450A1/de not_active Withdrawn
-
2010
- 2010-08-17 US US13/389,854 patent/US8916511B2/en not_active Expired - Fee Related
- 2010-08-17 CN CN201080041059.5A patent/CN102549046B/zh not_active Expired - Fee Related
- 2010-08-17 EP EP10744925A patent/EP2478036A1/de not_active Withdrawn
- 2010-08-17 MX MX2012002890A patent/MX2012002890A/es active IP Right Grant
- 2010-08-17 WO PCT/EP2010/061968 patent/WO2011032797A1/de active Application Filing
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE1493384A1 (de) | 1964-07-14 | 1969-01-30 | Union Carbide Corp | Verfahren zur Herstellung von Organosiliciumverbindungen |
| EP0294642A2 (de) | 1987-06-06 | 1988-12-14 | Th. Goldschmidt AG | Diquaternäre Polysiloxane, deren Herstellung und Verwendung in kosmetischen Zubereitungen |
| WO2003078504A1 (de) * | 2002-03-20 | 2003-09-25 | Ge Bayer Silicones Gmbh & Co. Kg | Verzweigte polyorganosiloxanpolymere |
| DE102004002208A1 (de) * | 2004-01-15 | 2005-08-18 | Rudolf Gmbh & Co. Kg Chemische Fabrik | Zubereitungen auf Basis von Organopolysiloxan-Polyammonium-Blockcopolymeren und deren Anwendung auf textilen Substraten |
| EP1561770A1 (de) * | 2004-02-09 | 2005-08-10 | Schill + Seilacher "Struktol" Aktiengesellschaft | Verzweigte Polyorganosiloxane mit quarternären Ammoniumgruppen |
| EP1887024A1 (de) * | 2006-07-31 | 2008-02-13 | Evonik Goldschmidt GmbH | Neuartige Polysiloxane mit quaternären Ammoniumgruppen, Verfahren zu deren Herstellung und deren Verwendung in reinigenden und pflegenden Formulierungen |
| WO2009065644A1 (de) | 2007-11-21 | 2009-05-28 | Evonik Goldschmidt Gmbh | Verfahren zur herstellung verzweigter sih-funktioneller polysiloxane und deren verwendung zur herstellung sic- und sioc-verknüpfter, verzweigter organomodifizierter polysiloxane |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20140134125A1 (en) * | 2011-06-30 | 2014-05-15 | Evonik Degussa Gmbh | Microemulsion of polysiloxanes containing quaternary ammonium groups, production and use thereof |
| US9138385B2 (en) * | 2011-06-30 | 2015-09-22 | Evonik Degussa Gmbh | Microemulsion of polysiloxanes containing quaternary ammonium groups, production and use thereof |
| WO2013032493A1 (en) * | 2011-08-26 | 2013-03-07 | Colgate-Palmolive Company | Fabric wrinkle reduction composition |
| CN103748204A (zh) * | 2011-08-26 | 2014-04-23 | 高露洁-棕榄公司 | 织物皱折降低组合物 |
| US10428295B2 (en) | 2011-08-26 | 2019-10-01 | Colgate-Palmolive Company | Fabric wrinkle reduction composition |
Also Published As
| Publication number | Publication date |
|---|---|
| US20120168664A1 (en) | 2012-07-05 |
| DE102009029450A1 (de) | 2011-03-24 |
| CN102549046A (zh) | 2012-07-04 |
| CN102549046B (zh) | 2015-02-18 |
| EP2478036A1 (de) | 2012-07-25 |
| US8916511B2 (en) | 2014-12-23 |
| MX2012002890A (es) | 2012-04-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2011032797A1 (de) | Neuartige polysiloxane mit quaternären ammoniumgruppen und deren verwendung | |
| EP2444447B1 (de) | Polysiloxane mit stickstoffhaltigen Gruppen | |
| EP1887023B1 (de) | Neuartige Polysiloxane mit quaternären Ammoniumgruppen, Verfahren zu deren Herstellung und deren Verwendung als Textilweichmacher | |
| EP2528660B1 (de) | Neuartige lineare polydimethylsiloxan-polyether-copolymere mit amino- und/oder quaternären ammoniumgruppen und deren verwendung | |
| EP1632521B1 (de) | Öl-in-Wasser-Emulsionen von Aminosiloxanen | |
| EP3774995B1 (de) | Siloxane zur behandlung von textilien und zur verwendung in reinigungs- und pflegeformulierungen | |
| EP0313867B1 (de) | Organopolysiloxane mit Buntesalzgruppen | |
| DE69003009T2 (de) | Verfahren zur Behandlung von Fasermaterialien. | |
| EP0130460B1 (de) | Organopolysiloxane mit Buntesalzgruppen, deren Herstellung und Verwendung zur Oberflächenbehandlung von anorganischen oder organischen Materialien | |
| EP1900765A1 (de) | Neue polycarboxyfunktionelle Organosiloxanylderivate und deren Verwendung zur Behandlung von Leder | |
| DE102007015372A1 (de) | Polysiloxan und Textilhilfsmittel enthaltend ein Polysiloxan | |
| DE102006040010A1 (de) | Verwendung von polyethermodifizierten Siloxanblockcopolymeren als hydrophile silikonhaltige Weichmacher für Gewebe, Non-wovens und/oder Fasern aus natürlichen und/oder synthetischen Rohstoffen | |
| WO2018146016A1 (de) | Polymere zur hydrophoben und oleophoben textilausrüstung | |
| EP1914261B1 (de) | Neue phosphatierte Organosiloxanylderivate und deren Verwendung zur Behandlung von Leder | |
| EP3303453B1 (de) | Polyurethan-organopolysiloxane | |
| EP0016907B1 (de) | Zubereitung zum Schrumpffestmachen von Wolle | |
| EP2260140B1 (de) | Verwendung von polyamino- und/oder polyammonium-polysiloxan-copolymer-verbindungen | |
| EP1625180B1 (de) | Polyorganosiloxan-zusammensetzungen zur behandlung von substraten | |
| DE102005056864B4 (de) | Zubereitungen auf Basis von ammonium- und polyethermodifizierten Organopolysiloxanen und deren Verwendung zur Veredlung textiler Substrate | |
| EP1595910A1 (de) | Derivatisierte, permanent quaternierte Stickstoffatome aufweisende, geradkettige oder verzweigte aminofunktionelle Organopolysiloxane | |
| DE10004321A1 (de) | Quarternäre Ammonium- und tertiäre Aminogruppen enthaltende Organosiliciumverbindungen, deren Herstellung und Verwendung | |
| EP4166710A1 (de) | Weichgriffzusammensetzung |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080041059.5 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10744925 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 292/KOLNP/2012 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13389854 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2010744925 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010744925 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2012/002890 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |