WO2010079467A2 - Delivery particles - Google Patents
Delivery particles Download PDFInfo
- Publication number
- WO2010079467A2 WO2010079467A2 PCT/IB2010/052121 IB2010052121W WO2010079467A2 WO 2010079467 A2 WO2010079467 A2 WO 2010079467A2 IB 2010052121 W IB2010052121 W IB 2010052121W WO 2010079467 A2 WO2010079467 A2 WO 2010079467A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- acid
- water
- poly
- oil
- Prior art date
Links
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J13/00—Colloid chemistry, e.g. the production of colloidal materials or their solutions, not otherwise provided for; Making microcapsules or microballoons
- B01J13/02—Making microcapsules or microballoons
- B01J13/06—Making microcapsules or microballoons by phase separation
- B01J13/14—Polymerisation; cross-linking
- B01J13/16—Interfacial polymerisation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/11—Encapsulated compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/92—Oils, fats or waxes; Derivatives thereof, e.g. hydrogenation products thereof
- A61K8/922—Oils, fats or waxes; Derivatives thereof, e.g. hydrogenation products thereof of vegetable origin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q15/00—Anti-perspirants or body deodorants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/10—Washing or bathing preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/02—Preparations for cleaning the hair
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/12—Preparations containing hair conditioners
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J13/00—Colloid chemistry, e.g. the production of colloidal materials or their solutions, not otherwise provided for; Making microcapsules or microballoons
- B01J13/02—Making microcapsules or microballoons
- B01J13/06—Making microcapsules or microballoons by phase separation
- B01J13/14—Polymerisation; cross-linking
- B01J13/18—In situ polymerisation with all reactants being present in the same phase
- B01J13/185—In situ polymerisation with all reactants being present in the same phase in an organic phase
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0097—Dye preparations of special physical nature; Tablets, films, extrusion, microcapsules, sheets, pads, bags with dyes
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D17/00—Detergent materials or soaps characterised by their shape or physical properties
- C11D17/0008—Detergent materials or soaps characterised by their shape or physical properties aqueous liquid non soap compositions
- C11D17/0013—Liquid compositions with insoluble particles in suspension
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D17/00—Detergent materials or soaps characterised by their shape or physical properties
- C11D17/0008—Detergent materials or soaps characterised by their shape or physical properties aqueous liquid non soap compositions
- C11D17/003—Colloidal solutions, e.g. gels; Thixotropic solutions or pastes
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D17/00—Detergent materials or soaps characterised by their shape or physical properties
- C11D17/0039—Coated compositions or coated components in the compositions, (micro)capsules
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/50—Perfumes
- C11D3/502—Protected perfumes
- C11D3/505—Protected perfumes encapsulated or adsorbed on a carrier, e.g. zeolite or clay
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/41—Particular ingredients further characterized by their size
- A61K2800/412—Microsized, i.e. having sizes between 0.1 and 100 microns
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/56—Compounds, absorbed onto or entrapped into a solid carrier, e.g. encapsulated perfumes, inclusion compounds, sustained release forms
Definitions
- This invention relates to capsule manufacturing processes and microcapsules produced by such processes as well as compositions comprising such microcapsules and methods of making and using such compositions
- U S Pat Nos 2,730,456, 2,800,457, and 2,800,458 describe methods for capsule formation
- Other useful methods for microcapsule manufacture are U S Pat Nos 4,001 , 140, 4,081 ,376 and 4,089,802 desc ⁇ bing a reaction between urea and formaldehyde, U S Pat No 4, 100,103 describing reaction between melamrne and formaldehyde, British Pat No 2,062,570 desc ⁇ bing a process for producing microcapsules having walls produced by polymerization of melamine and formaldehyde in the presence of a styrenesulfonic acid Microcapsules are also taught in U S Pat Nos 2,730,457 and 4, 197,346 Forming microcapsules from urea-formaldehyde resin and/or melamine formaldehyde resin is disclosed in U S Pat Nos 4,001, 140, 4,081 ,376, 4,089,802, 4,100,103, 4,105,823,
- Interfacial polymerization is a process wherein a microcapsule wall of a polyamide, an epoxy resin, a polyurethane, a polyurea or the like is formed at an interface between two phases
- U S Pat No 4,622,267 discloses an interfacial polymerization technique for preparation of microcapsules
- the core mate ⁇ al is initially dissolved in a solvent and an aliphatic diisocyanate soluble in the solvent mixture is added Subsequently, a nonsolvent for the aliphatic diisocyanate is added until the turbidity point is just barely reached
- This organic phase is then emulsified in an aqueous solution, and a reactive amine is added to the aqueous phase The amine diffuses to the interface, where it reacts with the diisocyanate to form polymeric polyurethane shells
- U S Pat No 4,547,429 U S Pat No 4,547,429
- U S Patent 5,292,835 teaches polymerizing esters of acrylic acid or methacrylic acid with polyfunctional monomers Specifically illustrated are reactions of polyvinylpyrrolidone with acrylates such as butanediol diacrylate or methylmethacrylate together with a free radical initiator
- the core material which is to be encapsulated is typically emulsified or dispersed in a suitable dispersion medium
- This medium is typically aqueous but involves the formation of a polymer rich phase
- this medium is a solution of the intended capsule wall material
- the solvent characteristics of the medium are changed such as to cause phase separation of the wall material
- the wall material is thereby contained in a liquid phase which is also dispersed in the same medium as the intended capsule core material
- the liquid wall material phase deposits itself as a continuous coating about the dispersed droplets of the internal phase or capsule core material
- the wall material is then solidified This process is commonly known as coacervation
- U S Patent No 4,046,750 teaches an ionene modified polymeric bead Dimethylamino substituted acrylic polymer is cross-linked and then the formed beads are reacted with a mixture of ditertiary amine and a dihalide to attach ionene segments to the tertiary amine centers on the beads Insoluble cationic modified beads are formed These cationic beads are useful in affinity chromatography
- Microcapsule Technologies in WO 01/41915 teaches coating formed anionic microcapsules by adding compounds with a cationic charge in a controlled manner For example polyurea capsules are introduced to a solution of vinylpyrrohdone to coat the capsules and render them with cationic character Similarly melamine microcapsules are taught to be coated with a homogenous solution of hydroxypropyl guar to impact cationic character Gelatin capsules are illustrated coated with hydroxyethyl cellulose followed by epichlorhyd ⁇ n to render them cationic
- U S publication 2005/01 12152 also teaches applying a second coating of a cationic material over an acrylamide and melamine formaldehyde based wall Cationic polymer coated capsules are taught prepared by mixing uncoated fragrance containing capsules with a cationic polymeric deposition aid
- capsules manufactured using the aforementioned methods and raw materials have several drawbacks which include (1) they cannot be formulated in certain classes of products due to st ⁇ ct formulation limits, (2) they have high permeabilities when incorporated into products that contain high levels of surfactant, solvents, and/or water, which results in the premature benefit agent release, (3) they can only effectively encapsulate a limited breadth of benefit agents, and (4) they either are so stable that they do not release the benefit agent in use or have insufficient mechanical stability to withstand the processes required to incorporate them in and/or make a consumer product and (5) they do not adequately deposit on the situs that is being treated with consumer product that contains capsules.
- Capsules made according to the invention can be made to better control permeability characte ⁇ stics. Capsules made according to the invention are surprisingly better able to contain liquid contents without leakage over time. The capsules can be made less leaky than those made by comparable prior art processes. Alternatively permeability in certain applications is desired Through selection of wall mate ⁇ al and control of length of time of cross-linking or temperature of cross-linking, capsules can be made with differing permeability profiles from extremely tight with little to no leakage to capsules that have measurable permeability useful where a measurable release rate over time is desired.
- the capsules according to the invention are useful with a wide va ⁇ ety of capsule contents ("core materials") including, by way of illustration and without limitation, perfumes; b ⁇ ghteners; insect repellants; silicones; waxes; flavors, vitamins; fab ⁇ c softening agents; skin care agents; enzymes; probiotics; dye polymer conjugate, dye clay conjugate; perfume delivery system,, sensates in one aspect a cooling agent; attractants, in one aspect a pheromone; antibacterial agents; dyes, pigments; bleaches; flavorants, sweeteners, waxes; pharmaceuticals, fertilizers; herbicides and mixtures thereof.
- core materials including, by way of illustration and without limitation, perfumes; b ⁇ ghteners; insect repellants; silicones; waxes; flavors, vitamins; fab ⁇ c softening agents; skin care agents; enzymes; probiotics; dye polymer conjugate, dye clay conjugate; perfume delivery system,, sensates in one aspect a cooling agent; attractants, in one aspect
- the microcapsule core materials can include mate ⁇ als which alter rheology or flow characte ⁇ stics, or extend shelf life or product stability.
- Essential oils as core materials can include, for example, by way of illustration wintergreen oil, cinnamon oil, clove oil, lemon oil, lime oil, orange oil, peppermint oil and the like.
- Dyes can include fluorans, lactones, indolyl red, I6B, leuco dyes, all by way of illustration and not limitation
- the core mate ⁇ al should be dispersible or sufficiently soluble in the capsule internal phase mate ⁇ al namely in the internal phase oil or soluble or dispersible in the monomers or oligomers solubihzed or dispersed in the internal phase oil
- the core material should be dispersible or sufficiently soluble in the water phase
- the invention is particularly useful to encapsulate volatile fragrances and flavorants
- the core mate ⁇ al should be soluble or dispersible in the water phase so as to form a dispersion in water that can be emulsified into the oil phase
- capsules according to the invention are also able to be fashioned with thermoplastic polymeric materials resulting in low leakage heat sensitive capsules that could be opened with heat in addition to conventional techniques such as pressure, scraping, friction, shearing, impact, or other energy input
- the capsules according to the invention can also be useful in applications with thermal print heads, or lasers, or other heating or impact elements
- light sensitive capsules are also feasible
- a low permeability capsule according to the invention can be fashioned Where measurable release is desired, more permeable capsules can also be fashioned Where adhesion on surfaces such as textiles and anionic substrates is desired, cationic capsules can also be fashioned
- microcapsules are obtained through either oil in water (OAV) or water in oil (W/O) emulsifications
- microcapsules are obtained by steps comprising dispersing an oil soluble amine modified polyfunctional polyvinyl monomer (or oligomer) and an oil soluble bi- or polyfunctional vinyl monomer or oligomer along with a free radical initiator such as an azo or peroxy initiator and an organic acid into an internal phase oil which is a non-solvent for the water phase
- the phase in excess is water with OAV emulsification
- W/O emulsifications the phase in excess or continuous phase is oil
- the term internal phase oil is used for convenience and simplicity to refer to the oil phase and to refer to the type of oils conventionally used as the internal phase or contents of microcapsules in conventional microencapsulation (with the W/O emulsifications taught herein, the oil however ends up being the continuous
- microcapsules are obtained by steps comprising dispersing an oil soluble amine modified ethoxylated t ⁇ methylol propane t ⁇ acrylate and an oil soluble diethylene glycol dimethacrylate along with a free radical initiator such as an azo initiator and an organic acid into an internal oil phase, heating for time and temperature sufficient to ohgome ⁇ ze the amine modified ethoxylated t ⁇ methylol propane t ⁇ acrylate and a diethylene glycol dimethacrylate forming a pre-polymer, then, adding to the oil phase a water phase comprising a dispersion of water, and an emulsifier, and an optional second initiator The water phase is then emulsified into the oil phase (W/O) and heated for a time and temperature sufficient to decompose at least one of the free radical initiators in either or both of the oil and water phases, thereby forming microcapsule wall material at the interface of the water and oil phases
- microcapsules are obtained by steps comprising dispersing an oil soluble amine modified polyfunctional polyvinyl monomer and an oil soluble bi- or polyfunctional vinyl monomer or oligomer along with a free radical azo initiator and an organic acid into an internal phase, then, heating for a time and temperature sufficient to ohgome ⁇ ze or further oligome ⁇ ze the amine modified polyfunctional polyvinyl monomer or oligomer and oil soluble bi- or polyfunctional vinyl monomer forming a pre-polymer, then, adding to the oil phase a water phase in excess comprising a dispersion in water of an emulsifier and an optional second initiator, and adding an emulsifying agent, emulsifying the oil phase into the water phase, then heating for a time and temperature sufficient to decompose the free radical initiators in either or both of the oil and water phases; thereby forming microcapsule wall material at the interface of the water and
- microcapsules are obtained by steps comprising dispersing an oil soluble amine modified ethoxylated trimethylol propane triacrylate and an oil soluble diethylene glycol dimethacrylate along with a free radical initiator such as an initiator and an organic acid into an internal phase oil; heating for time and temperature sufficient to oligomerize the amine modified ethoxylated trimethylol propane triacrylate and a diethylene glycol dimethacrylate forming a pre-polymer; then, adding to the internal phase oil a water phase comprising a dispersion of water, and an emulsifier, emulsifying the oil phase into the water phase; then heating for a time and temperature sufficient to decompose the free radical initiators in the oil and water phases; thereby forming microcapsule wall material at the interface of the water and oil phases.
- the free radical initiator can be included in one or both of the oil and water phase.
- the invention comprises microcapsules obtained by steps comprising dispersing an oil soluble amine modified polyfunctional polyvinyl monomer or oligomer and an oil soluble bi- or polyfunctional vinyl monomer or oligomer along with a free radical initiator and an organic acid into an internal phase oil.
- a first heating step is used comprising, heating for a time and temperature sufficient to oligomerize or further oligomerize the amine modified polyfunctional polyvinyl monomer or oligomer and oil soluble bi- or polyfunctional vinyl monomer oligomer forming a pre-polymer.
- a water phase comprising a dispersion in water of an emulsifier and a free radical initiator is added to the internal phase oil.
- the water phase is emulsified into the oil phase.
- a second heating step is used comprising, heating for a time and temperature sufficient to decompose the free radical initiators in the oil and water phases thereby forming microcapsule wall material at the interface of and oil phases.
- a third heating step comprising heating to a temperature equal to or greater than the second heating step temperature is used for a time sufficient to polymerize the wall material.
- the free radical initiator is preferably selected from an azo or peroxy initiator. Oligomerization in one embodiment is accomplished by heating, in the first heating step, to at least 55°C for at least one hour to form the prepolymer.
- the initiator in the oil phase decomposes at a first temperature and the initiator in the water phase decomposes at a second temperature.
- the invention comprises microcapsules obtained by steps comprising dispersing an oil soluble amine modified polyfunctional polyvinyl monomer or oligomer and an oil soluble bi- or polyfunctional vinyl monomer or oligomer along with a free radical initiator and an organic acid into an internal phase oil, a first heating step comprising, heating for a time and temperature sufficient to oligome ⁇ ze or further oligomenze the amine modified polyfunctional polyvinyl monomer or oligomer and oil soluble bi- or polyfunctional vinyl monomer or oligomer forming a pre-polymer, adding to the internal phase oil a water phase in excess compnsing a dispersion in water of a polyacrylic or polymethacrylic acid, and a free radical initiator, and adding an emulsifying agent, emulsifying the oil phase into
- the oligome ⁇ zation is accomplished by heating, in the first heating step, is to at least 55°C for at least one hour to form the prepolymer and the third heating step is to at least 90 0 C for at least three hours
- the second heating step composed heating to a temperature equal to or greater than the first step, preferably greater
- the second step temperature could involve dropping the temperature slightly less than the first step, if only prolonged heating is needed to degrade any remaining free radical initiator
- microcapsules are obtained by steps comprising dispersing an oil soluble amine modified ethoxylated t ⁇ methylol propane tnacrylate and an oil soluble diethylene glycol dimethacrylate along with free radical initiator and an organic acid into an internal phase oil
- a first heating step is used comprising, heating for time and temperature sufficient to oligomenze the amine modified ethoxylated t ⁇ methylol propane tnacrylate and a diethylene glycol dimethacrylate forming a pre-polymer, then adding to the internal phase oil a water phase compnsing a dispersion of water, and a polyacrylic or polymethacrylic acid, and adding an emulsifying agent, and emulsifying the water phase into the oil phase
- a second heating step then comprises, heating for a time and temperature sufficient to decompose at least a portion of the free radical initiator in the oil phase, thereby forming microcapsule wall material at the interface of the water and oil phases, and
- the initiators in the oil phase and the water phase can be the same or different
- microcapsules are obtained by steps composing dispersing an oil soluble amine modified polyfunctional polyvinyl monomer or oligomer and an oil soluble bi- or polyfunctional vinyl monomer or oligomer along with a free radical initiator and an organic acid into an internal phase oil, a first heating step comprising heating for a time and temperature sufficient to decompose at least some portion of the free radical initiator and thereby ohgome ⁇ ze or further ohgome ⁇ ze the amine modified polyfunctional polyvinyl monomer or oligomer and oil soluble bi- or polyfunctional vinyl monomer or oligomer forming a pre-polymer
- Added to the internal phase oil is a water phase comprising a dispersion in water of an emulsifier, and emulsifying the water phase into the oil phase forming droplets of the water phase dispersed in the oil phase
- a second heating step comp ⁇ sing heating for a time and temperature sufficient to decompose the remaining portion of free radical initiator thereby
- the oligome ⁇ zation is accomplished by heating, in the first heating step, to at least 55 0 C for at least one hour to form the prepolymer
- the third heating step comprises heating to at least 9O 0 C for at least three hours
- the initiator is added in addition to the water phase dispersion of anionic emulsifier, and the initiator in the oil phase decomposes at a first temperature and the initiator in the water phase decomposes at a second temperature
- the initiators in the oil and water phases can be the same or different
- microcapsules are obtained by steps comp ⁇ sing providing an internal phase oil and a water phase containing a free radical initiator in at least one of said phases, and dispersing an oil soluble amine modified polyfunctional polyvinyl monomer or oligomer and an oil soluble bi- or polyfunctional vinyl monomer or oligomer and an organic acid into the internal phase oil, then adding to the internal phase oil the water phase which further comprises a dispersion in water of an emulsifier, and emulsifying the water phase into the oil phase forming droplets of the water phase in the oil phase
- a first heating step comprises, heating for a time and temperature sufficient to decompose the free radical initiator in at least the oil or water phase, and sufficient to oligome ⁇ ze or further oligome ⁇ ze the amine modified polyfunctional polyvinyl monomer or oligomer and oil soluble bi- or polyfunctional vinyl monomer or oligomer forming a pre- polymer and thereby forming microcapsule wall material at the interface of the water and oil phases
- a second heating step comprises heating to a temperature equal to or greater than the first heating step temperature for a time sufficient to polymerize the wall material
- the second heating step comprises heating to at least 90 0 C for at least three hours
- initiator can be added to both the oil and water phases
- the initiator in the oil phase can decompose at a first temperature
- the initiator in the water phase can decompose at a second temperature
- the initiators in the oil and water phases can be the same or different
- the cationic or nonionic emulsifier comprises a water-soluble or water-dispersible material and optionally a water phase initiator, where the first composition initiator and the water phase initiator is an energy-activated initiator
- the reaction product of the first composition and second composition results in the formation of a population of microcapsules having a microcapsule wall of low permeance to the core material and having a zeta potential of -5 millivolts or greater, the resulting microcapsules having strong adherence to anionic surfaces
- consumer product means baby care, personal care, fabric & home care, family care, feminine care, health care, snack and/or beverage products or devices intended to be used or consumed in the form in which it is sold, and not intended for subsequent commercial manufacture or modification
- Such products include but are not limited to diapers, bibs, wipes, products for and/or methods relating to treating hair (human, dog, and/or cat), including, bleaching, coloring, dyeing, conditioning, shampooing, styling, deodorants and antiperspirants, personal cleansing, cosmetics, skin care including application of creams, lotions, and other topically applied products for consumer use, and shaving products, products for and/or methods relating to treating fabrics, hard surfaces and any other surfaces in the area of fabric and home care, including air care, car care, dishwashing, fabric conditioning (including softening), laundry detergency, laundry and rinse additive and/or care, hard surface cleaning and/or treatment, and other cleaning for consumer or institutional use, products and/or methods relating to bath tissue, facial tissue, paper handker
- cleaning composition includes, unless otherwise indicated, granular or powder-form all-purpose or “heavy-duty” washing agents, especially cleaning detergents, liquid, gel or paste-form all-purpose washing agents, especially the so-called heavy- duty liquid types, liquid fine-fabric detergents, hand dishwashing agents or light duty dishwashing agents, especially those of the high-foaming type, machine dishwashing agents, including the va ⁇ ous tablet, granular, liquid and rinse-aid types for household and institutional use, liquid cleaning and disinfecting agents, including antibacte ⁇ al hand- wash types, cleaning bars, mouthwashes, denture cleaners, dentif ⁇ ce, car or carpet shampoos, bathroom cleaners, hair shampoos and hair-rinses, shower gels and foam baths and metal cleaners, as well as cleaning auxiliaries such as foam substrates, films, and combinations thereof, bleach additives and "stain- stick” or pre-treat types, substrate-laden products such as dryer added sheets, dry and wetted wipes and pads
- fabric care composition includes, unless otherwise indicated, fab ⁇ c softening compositions, fabric enhancing compositions, fabric freshening compositions and combinations there of
- the term "personal care composition” includes, unless otherwise indicated, any personal care composition that can be applied to the keratinaceous surfaces of the body including the skin and/or hair
- the personal care compositions can be, for example, formulated as bars, liquids, emulsions, shampoos, gels, powders, sticks, hair conditioners (rinse off and leave in), hair tonics, pastes, hair colorants, sprays, mousses and/or other styling products, as well as shave prep products, and devices used for shaving
- fluid includes liquid, gel, paste and gas product forms
- solid means granular, powder, bar and tablet product forms
- situs includes paper products, fabrics, garments, hard surfaces, hair and skin
- microcapsule encompass perfume microcapsules
- test methods disclosed in the Test Methods Section of the present application should be used to determine the respective values of the parameters of Applicants' inventions
- component or composition levels are in reference to the active portion of that component or composition, and are exclusive of impurities, for example, residual solvents or by-products, which may be present in commercially available sources of such components or compositions
- the present invention teaches novel processes for microencapsulation involving water in oil, or oil in water emulsifications, and microcapsules obtained by such processes
- the present invention teaches a low permeability microcapsule particle comp ⁇ sing a core mate ⁇ al and a wall material at least partially surrounding, and preferably completely surrounding a core mate ⁇ al
- the a first composition may comprise an oil soluble or dispersible primary, secondary, or tertiary amine with a multifunctional acrylate or methacrylate monomer or oligomer and an oil soluble acid and an initiator
- a second composition is an emulsifier and comp ⁇ ses a water soluble or water dispersible polymer or copolymer, usually at least one water phase initiator and one or more of an alkali or alkali salt
- water phase initiator it is meant that the initiator is soluble or dispersible in water
- the amines can include by way of illustration and not limitation amine modified vinyl monomers including amine modified acrylates or methacrylates such as mono or diacrylate amines, mono or dimethacrylate amines, amine modified polyetheracrylates and amine modified polyethermethacrylates, aminoalkyl acrylates or aminoalkyl methacrylate
- the amines can include primary, secondary or tertiary amines and can include tertiary butyl aminethylmethacrylate, diethylaminoethyl methacrylate, or dimethylaminoethyl methacrylate
- the present invention in one embodiment is a process of obtaining microcapsules by dispersing an oil soluble amine modified polyfunctional polyvinyl monomer or oligomer and an oil soluble bi- or polyfunctional vinyl monomer or oligomer along with a free radical initiator, such as an azo or peroxy initiator, and an organic acid into an internal phase oil
- a free radical initiator such as an azo or peroxy initiator
- an organic acid such as an azo or peroxy initiator
- This dispersion is heated for a time and temperature sufficient to ohgome ⁇ ze or further oligome ⁇ ze the amine modified polyfunctional polyvinyl monomer and oil soluble bi- or polyfunctional vinyl monomer or oligomer to form a prepolymer
- a water phase is added comprising a dispersion in water of an emulsifier or an initiator
- the water phase in one embodiment is emulsified into the oil phase (W/O)
- the dispersion is then heated for a time and
- the emulsion is usually milled to a size of from about 2 microns to about 80 microns, from about 5 microns to about 50 micronsm, or even from about 10 microns to about 30 microns Larger sizes for particular applications are also feasible
- the invention teaches novel processes for microencapsulation using water in oil, or alternatively oil in water emulsifications
- the capsules by the process of the invention enable a low permeability or controlled permeability capsule to be fashioned Permeability can be controlled through wall mate ⁇ al selection, through control of degree of cross-linking, by controlling temperature of cross-linking, by controlling length of time of cross-linking, or with UV initiated systems by controlling intensity of UV light and duration
- the present invention is a process of obtaining microcapsules by dispersing an oil soluble amine modified polyfunctional polyvinyl monomer or oligomer and an oil soluble bi- or polyfunctional vinyl monomer or oligomer along with a free radical initiator, such as a peroxy or azo initiator, and an organic acid into an internal phase oil
- a free radical initiator such as a peroxy or azo initiator
- an organic acid such as a peroxy or azo initiator
- This dispersion is heated for a time and temperature sufficient to ohgome ⁇ ze or further ohgome ⁇ ze the amine modified polyfunctional polyvinyl monomer and oil soluble bi- or polyfunctional vinyl monomer to form a prepolymer
- a water phase is added in excess comprising a dispersion in water of an emulsifier and optionally, a free radical initiator
- the oil phase in this embodiment is emulsified into the water phase (OAV)
- the dispersion is then heated for
- Preferred amine modified polyfunctional polyvinyl monomers include amine modified ethoxylated t ⁇ methylol propane t ⁇ acrylate, ethoxylated aliphatic , acrylated amines, such as diacrylate amines, t ⁇ acrylate amines dimethacrylate amines, amine modified polyetheracrylates and amine modified polyethermethacrylates
- Preferred bi- or polyfunctional vinyl monomers include by way of illustration and not limitation, allyl methacrylate, methylene glycol dimethacrylate, ethylene glycol dimethacrylate, diethylene glycol dimethacrylate, aliphatic or aromatic urethane diacrylates, difunctional urethane acrylates, ethoxylated aliphatic difunctional urethane methacrylates, aliphatic or aromatic urethane dimethacrylates, epoxy acrylates, epoxymethacrylates, tetraethylene glycol dimethacrylate, polyethylene glycol dimethacrylate, 1 ,3 butylene glycol diacrylate, 1 ,4- butanediol dimethacrylate, 1 ,4-butaneidiol diacrylate, diethylene glycol diacrylate, 1 ,6 hexanediol diacrylate, 1 ,6 hexanediol dimethacrylate, neopentyl glycol diacrylate
- the organic acid can be selected from various acids such as carboxy acids, with monoalkyl maleates such as monomethyl, monoethyl or monobutyl maleate being preferred, with monobutyl maleate being most preferred
- organic acids that can be usefully employed in the invention include, organic sulfonic acids such as alkyl benezene sulfonic acid, more particularly linear alkyl benzene sulfonic acid, t ⁇ decylbenzene sulfonic acid, more particularly linear t ⁇ alkyl benzene sulfonic acid such as linear t ⁇ decyl benzene sulfonic acid, alkyldiphenyloxide sulfonic acid, preferably dodecyl diphenyl oxidedisulfonic acid, more particularly branched C12 diphenyl oxide disulfonic acid, alkylbenzene sulfonic acid, more particularly, dodecyl benzene sulfonic acid, dialkyl n
- Suitable emulsifiers for use herein are anionic, cationic, or nonionic emulsifiers
- amphote ⁇ c emulsifiers and zwitte ⁇ onic emulsifiers can find applicability
- the emulsifier is cationic the cationic or nonionic emulsifier comp ⁇ ses a water soluble or water dispersible material and optionally a water phase initiator
- the first composition initiator and the water phase initiator is an energy-activated initiator
- the reaction product of the first composition and second composition results in the formation of a population of microcapsules having a microcapsule wall of low permeance to the core material and having a zeta potential of -5 millivolts or greater
- the resulting microcapsules have adherence to anionic surfaces
- the emulsifier generally has a molecular weight greater than about 100
- Cationic emulsifiers include amine polymers with primary, secondary or tertiary functionality
- the cationic emulsifier is selected from palmitamidopropyltrimonium chloride (Va ⁇ soft PATCTM, available from Degussa Evonik, Essen, Germany), distearyl dimonium chloride, cetylt ⁇ methylammonium chloride, quaternary ammonium compounds, fatty amines, aliphatic ammonium halides, alkyldimethylbenzylammonium halides, alkyldimethylethylammonium halides, polyethyleneimine, poly(2-dimethylamino)ethyl methacrylate) methyl chloride quaternary salt, poly(l -vinylpyrrohdone-co-2-dimethylaminoethyl methacrylate), poly(acrylamide-co-diallyl
- the cationic emulsifiers include quaternary ammonium compounds with a long-chain aliphatic radical, e g distearyldiammonium chloride, and fatty amines
- catioruc emulsifiers which may be mentioned are alkyldimethylbenzylammonium halides, alkyldimethylethylammonium halides, etc
- Preferred emulsifiers are those which significantly reduce the interfacial tension between the aqueous phase and dispersed phase, and thereby reduce the tendency for droplet coalescence
- the emulsifier is nonionic
- the nonionic emulsifier is selected from polyalkylene glycol ether, condensation products of alkyl phenols, aliphatic alcohols, or fatty acids with alkylene oxide, ethoxylated alkyl phenols, ethoxylated arylphenols, ethoxylated polyaryl phenols, carboxyhc esters solubihzed with a polyol, polyvinyl alcohol, polyvinyl acetate, or copolymers of polyvinyl alcohol polyvinyl acetate, polyacrylamide, poly(7V-isopropylacrylamide), poly(2-hydroxypropyl methacrylate), poly(2- ethyl-2-oxazoline), poly(2-isopropenyl-2-oxazoline-co-methyl methacrylate), poly(methyl vinyl ether), and polyvinyl alcohol-co-ethylene).
- Especially useful polyvinylalcohols include polyvinyl alcohols of molecular weight 13000 to 186000 daltons, preferably from 13000 to about 23000 daltons, or even from 146000 to 186000 daltons.
- the polyvinyl alcohol can be partially or fully hydrolyzed.
- Polyvinyl alcohol partially hydrolyzed in the range of 85 to 95% hydrolyzed is preferred. Partially hydrolyzed polyvinylalcohol at 88% hydrolysis or less was useful, with about 88% hydrolysis being more preferred.
- An exemplary zwitterionic emulsifier is cocoamidopropyl betaine.
- the amount of emulsifier is anywhere from about 0.1 to about 40 percent by weight of all constituents, more preferably from 0.5 to about 10 percent, most preferably 0.5 to 5 percent by weight. Typically emulsifier is employed at 0.2 to about 10% by weight based on percentage of the total formulation.
- the primary, secondary or tertiary amine acrylate or methacrylate and the multi-functional acrylate or methacrylate monomers are used in a relative ratio by weight of from about 0.1 :99.9 to about 10:90 preferably from about 0.5:99.5 to about 5:95, and most preferably 1 :99 to about 3:97.
- the ratio of the amine to the multifunctional acrylate is in the range of from 0.1 :99.9 to 10:90. Therefore the amine can be in the range of from 0.1 to 10% by weight as compared to the multifunctional acrylate, or even from 0.5 to 5%, or most preferably 1 to 3% by weight.
- the amine modified polyfunctional polyvinyl monomer and the oil soluble bi- or poly functional vinyl monomers are the larger constituents by weight used in a relative ratio of from about 0.5: 1 to about 1 :3 preferably from about 1 : 1 to about 1 :2.
- the average molecular weight of the monomers initially is in the hundreds of daltons.
- For the oligomer molecular weights are in the thousands to tens of thousands of daltons.
- Prepolymers accordingly are higher molecular weight still.
- Prepolymers are an intermediate block of oligomers and monomers eventually forming a polymer.
- the monomer or oligomers should be selected to be soluble or dispersible in the oil phase.
- the largest constitutents are typically solvent, 10 to 70 weight percent, preferably 35 to 65 weight percent oil phase solvent and oil; 10 to 70 weight percent, preferably 35 to 65 weight percent water; 0.1 to 20 weight percent, usually 0.5 to 8 weight percent preferably 2 to 6 weight percent, bi- or polyfunctional vinyl monomer or oligomer; oil to 20 weight percent, usually 0.5 to 8 weight percent, preferably 2 to about 4 weight percent, amine modified amine modified polyfunctional monomer or oligomer Initiator is 10% or less, usually about 1 % or less, preferably 0 5% by weight or less and more preferably 0 1 % or less
- the amount of the respective solvent or oil can be increased or decreased as needed for rheology and depending on whether an W/O or OAV system is desired
- Preferred free radical initiators include peroxy initiators, azo initiators, peroxides, and compounds such as 2,2'-azobismethylbutyronit ⁇ le, dibenzoyl peroxide More particularly, and without limitation the free radical initiator can be selected from the group of initiators comprising an azo or peroxy initiator, such as peroxide, dialkyl peroxide, alkyl peroxide, peroxyester, peroxycarbonate, peroxyketone and peroxydicarbonate, 2, 2'-azobis (isobutylnitrile), 2,2'-azobis(2,4-dimethylpentanenit ⁇ le), 2,2'-azobis (2,4-dimethylvaleronit ⁇ le), 2,2'-azobis(2-methylpropanenitnle), 2,2'-azobis (methylbutyronit ⁇ le), l ,l '-azobis (cyclohexanecarbonit ⁇ le), l ,l '-azobis(cyanocyclohexane), benzoy
- Internal phase oils, or oil phase, or oil solvent or “nonsolvent for the water phase,” used interchangeably for purposes hereof can be selected from solvents and the solvents can include by way of illustration and not limitation, ethyldiphenylmethane, butyl biphenyl ethane, benzylxylene, alkyl biphenyls such as propylbiphenyl and butylbiphenyl, dialkyl phthalates e g dibutyl phthalate, dioctylphthalate, dinonyl phthalate and dit ⁇ decylphthalate, 2,2,4-t ⁇ methyl- 1,3-pentanediol dnsobutyrate, alkyl benzenes such as dodecyl benzene, alkyl or aralkyl benzoates such as benzyl benzoate, diaryl ethers, di(aralkyl)ethers and aryl aralkyl ethers,
- the capsule core may comp ⁇ se a partitioning modifier selected from the group consisting of oil soluble mate ⁇ als that have a ClogP greater than from about 4, or from about 5, or from about 7, or even from about 11 and/or mate ⁇ als that also have a density higher than 1 gram per cubic centimeter
- suitable partitioning modifier may comprise a material selected from the group consisting of mate ⁇ als include Mono, di- and t ⁇ -esters of C4-C 2 4 fatty acids and glycerine, fatty acid esters of poly glycerol oligomers, polyalphaolefins, silicone oil, crosslinked silicones comp ⁇ sing polyether substituted structural units and acrylate crosslinks, polyglycertol ether silicone crosspolymers, alkyl substituted cellulose, hydroxypropyl cellulose , fatty esters of acrylic or methacrylic acid that have side chain crystallizing groups, copolymers of ethylene, including ethylene and vinyl
- the microencapsulation process in certain of the embodiments is believed to rely on the organic acid for formation of a changed species that drives the wall material to the oil water interface
- Charged species may also be formed through the use of an oil-soluble organic acidic acrylate or methacrylate with an inorganic water-soluble base, or an oil-soluble organic amine acrylate or methacrylate with an inorganic water-soluble base
- Oil-soluble acids or bases may also be utilized, as appropriate, for neutralization of the acrylate or methacrylate acids or bases
- the size of the capsules can be controlled by adjusting the speed of agitation Smaller size dispersions are achieved through faster agitation resulting in smaller capsules
- Emulsifying agents or protective colloids can be conveniently employed to facilitate dispersion
- Such materials for example include carboxylated or partially hydrolyzed polyvinyl alcohol, methyl cellulose, and various latex materials, stearates, lecithins, and various surfactants
- microcapsules according to the invention can be used to microencapsulate various core materials such as chromogens and dyes, flavorants, perfumes, sweeteners, fragrances, oils, waxes, silicone oils, softening agents, vitamins, fats, pigments, cleaning oils, pharmaceuticals, pharmaceutical oils, perfume oils, mold inhibitors, antimicrobial agents, adhesives, phase change materials, scents, fertilizers, nutrients, and herbicides by way of illustration and without limitation
- Microencapsulation can facilitate processing by increasing particle size or by converting liquids into free flowing solids
- the largest volume applications of microcapsules are in imaging systems such as carbonless papers
- the microcapsule wall can serve the purpose of extending shelf life, stabilize and protect the core material, mask strong flavors, or protect contents so that they are available to participate in reactions such as imaging or adhesive formation when the capsule wall is ruptured, sheared, fractured, broken or melted
- the core matenal can be a minor or major constituent of the material encapsulated by the microcapsules If the core material can function as the oil or water solvent in the capsules, it is possible to make the core matenal the major or total matenal encapsulated Usually however, the core material is from 0 01 to 99 weight percent of the capsule internal contents, preferably 0 01 to about 65 by weight of the capsule internal contents, and more preferably from 0 1 to about 45% by weight of the capsule internal contents With certain especially potent materials, the core can be at just trace quantities
- a first composition is prepared as an oil phase #1
- the temperature of this oil phase is brought to a wall pre-reaction temperature
- a nitrogen blanket is preferably employed and the solution mixed with high shear agitation to disperse the droplets Gradually the temperature is increased to create a first composition reaction product
- a second oil phase is prepared and may be held at a pre-reaction temperature of the initiator
- the two oil solutions are allowed to pre-react and are combined
- the mixtures are stirred and held at the pre-reaction temperature for sufficient time to pre-react the wall material
- the water phase is added to the oil solutions
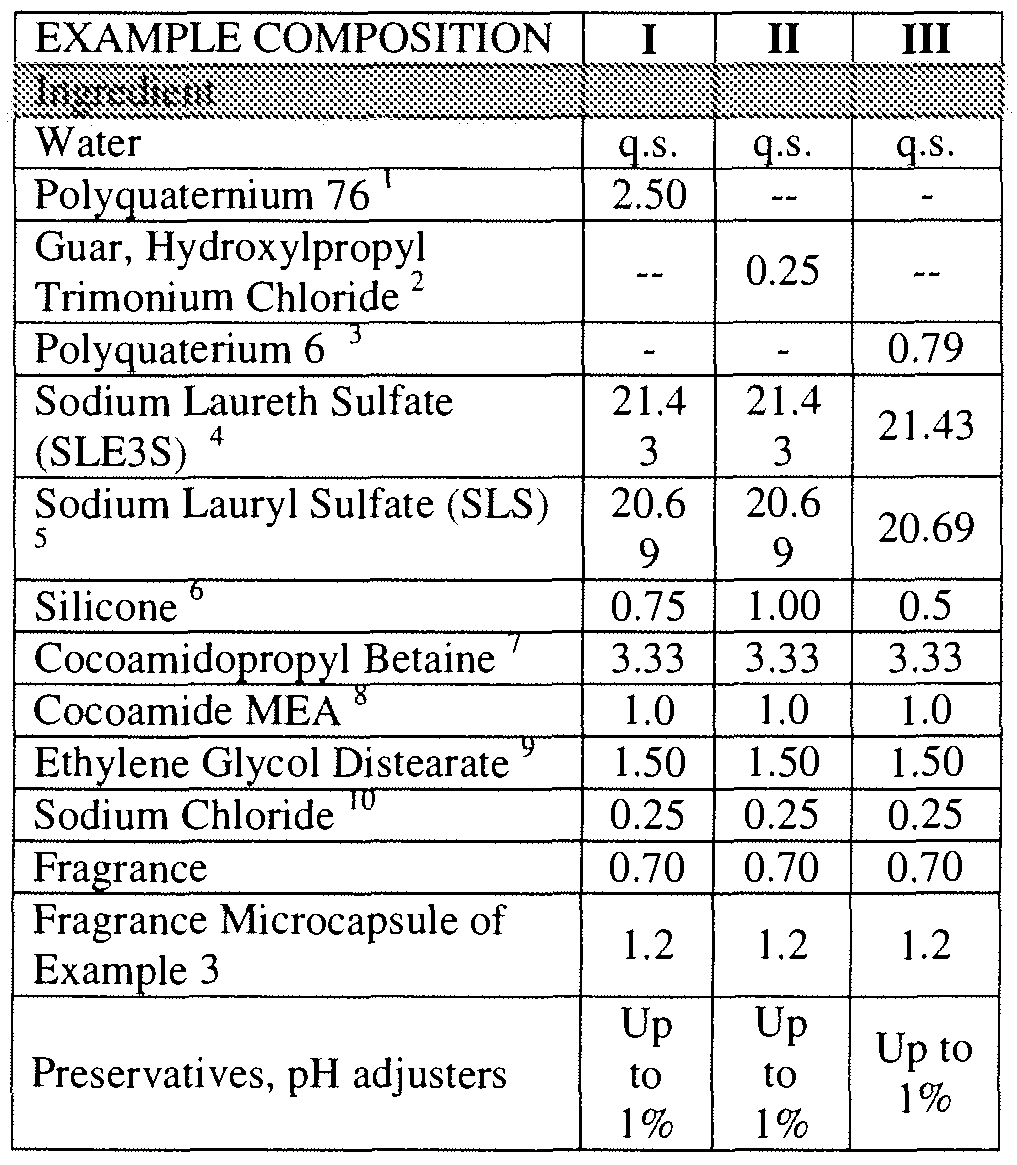
- Non-Ionic Capsule Type Polyvinyl Alcohol emulsifier
- Microcapsule particles according to the invention by selection of curing conditions, wall materials, initiators, and concentration can select for a desired permeance level allowing formation of capsules with more targeted release profiles appropriate to the end use application.
- the process of the invention enables manufacture of capsules with different permeability levels. Permeability is conveniently expressed as release of less than a certain quantity of core material over a given time frame. For example, low permeability would be release of less than 1.0 mg/ml at 48 hours extraction time, or less than 2 mg/ml at 1 week extraction time or less than 5 mg/ml at four weeks extraction time.
- the desired end use application often will dictate the target release rate deemed acceptable to meet the needs of the application. Slurry/Aggolmerate
- a slurry that may comprise any of the particles disclosed in the present specification is disclosed Said slurry may be combined with an adjunct ingredient to form a composition, for example, a consumer product
- one or more processing aids are selected from the group consisting of water, aggregate inhibiting materials such as divalent salts, particle suspending polymers, and mixtures thereof
- aggregate inhibiting materials include salts that can have a charge-shielding effect around the particle, such as magnesium chlo ⁇ de, calcium chloride, magnesium bromide, magnesium sulfate, and mixtures thereof
- particle suspending polymers include polymers such as xanthan gum, carrageenan gum, guar gum, shellac, alginates, chitosan, cellulosic materials such as carboxymethyl cellulose, hydroxypropyl methyl cellulose, cationically charged cellulosic materials, polyacrylic acid, polyvinyl alcohol, hydrogenated castor oil, ethylene glycol distearate, and mixtures thereof
- said slurry may comprise one or more processing aids, selected from the group consisting of water, aggregate inhibiting materials such as divalent salts, particle suspending polymers such as xanthan gum, guar gum, caboxy methyl cellulose
- said one or more earners may be selected from the group consisting of polar solvents, including but not limited to, water, ethylene glycol, propylene glycol, polyethylene glycol, glycerol, nonpolar solvents, including but not limited to, mineral oil, perfume raw materials, silicone oils, hydrocarbon paraffin oils, and mixtures thereof
- said slurry may comp ⁇ se a deposition aid that may comprise a polymer selected from the group comp ⁇ sing polysaccharides, in one aspect, cationically modified starch and/or cationically modified guar, polysiloxanes, poly diallyl dimethyl ammonium hahdes, copolymers of poly diallyl dimethyl ammonium chloride and polyvinyl pyrrohdone, a composition comprising polyethylene glycol and polyvinyl pyrrohdone, acrylamides, imidazoles, lmidazolinium hahdes, polyvinyl amine, copolymers of poly vinyl amine and N-vinyl formamide, polyvinylformamide, polyvinyl alcohol, polyvinyl alcohol crosslinked with boric acid, polyacrylic acid, polyglycerol ether silicone crosspolymers, polyacrylic acids, polyacrylates, copolymers of polyvinylamine and polvyinylalco
- an agglomerate that comprises said particles and a second material is disclosed
- said second mate ⁇ al may comp ⁇ se a material selected from the group consisting of silicas, cit ⁇ c acid, sodium carbonate, sodium sulfate, sodium chloride, and binders such as sodium silicates, modified celluloses, polyethylene glycols, polyacrylates, polyacrylic acids, zeolites and mixtures thereof
- a composition comprising an adjunct ingredient and a population of microcapsule particles comprising an oil soluble or dispersible core mate ⁇ al and a non-anionic wall material at least partially surrounding the core mate ⁇ al, the microcapsule wall mate ⁇ al comprising the reaction product of a first composition in the presence of a second composition comprising an emulsifier which is non-anionic, the first composition comprising a reaction product of i) an oil soluble or dispersible amine acrylate or methacrylate with ⁇ ) a multifunctional acrylate or methacrylate monomer or oligomer, and in) a soluble acid and an initiator, wherein the soluble acid and the amine acrylate are in a molar proportion from 3 1 to 1 3 and together have a percent by weight as compared to the weight of the wall mate ⁇ al of from 0 1 to 20% , the non-anionic emulsifier comprising a water soluble or dispersible material at a pH from 4 to 12, and optional
- the emulsifier may be cationic
- the cationic emulsifier may be selected from palmitamidopropylt ⁇ monium chloride, distearyl dimonium chloride, cetylt ⁇ methylammonium chloride, quaternary ammonium compounds, fatty amines, aliphatic ammonium halides, alkyldimethylbenzylammonium halides, alkyldimethylethylammonium halides, polyethyleneimine, ⁇ oly(2-dimethylamino)ethyl methacrylate) methyl chlo ⁇ de quaternary salt, poly(l -vinylpyrrolidone-co-2-dimethylaminoethyl methacrylate), poly(acrylamide-co- diallyldimethylammonium chlo ⁇ de), ⁇ oly(allylamine), poly[bis(2-chloroethyl) ether-alt- 1 ,3- bis[3-(dimethylamino
- the soluble acid and amine acrylate may have a percent by weight as compared to the weight of the wall mate ⁇ al of from 1 to 5 percent
- the soluble acid and amine acrylate may have a molar proportion of from 1 25/1 to 1/1 25
- the non-anionic emulsifier may comprise a water soluble or dispersible material at a pH of from 8-10
- the emulsifier may be nonionic
- the nonionic emulsifier may be selected from polyalkylene glycol ether, condensation products of alkyl phenols, aliphatic alcohols, or fatty acids with alkylene oxide, ethoxylated alkyl phenols, ethoxylated arylphenols, ethoxylated polyaryl phenols, carboxylic esters solubihzed with a polyol, polyvinyl alcohol, partially hydrolyzed polyvinyl alcohol, polyvinyl acetate, copolymers of polyvinyl alcohol, copolymers of polyvinyl acetate, polyacrylamide, poly(N-isopropylacrylamide), poly(2-hydroxypropyl methacrylate), poly(2-ethyl-2-oxazohne), ⁇ oly(2-isopropenyl-2-oxazohne-co-methyl methacrylate), poly(methyl vinyl ether), and polyvinyl alcohol-co-ethylene)
- the soluble acid and amine acrylate may have a percent by weight as compared to the weight of the wall mate ⁇ al of from 1 to 5 percent
- the soluble acid and amine acrylate may have a molar proportion of from 1 25/1 to 1/1 25
- the emulsifier may have a molecular weight greater than about 100, and may be selected from polymers with primary, secondary or tertiary amine functionality
- the emulsifier may have a molecular weight greater than 100 and may be selected from polymers with hydroxyl, ether, ester, ketone or amide functionality
- said population of microcapsule particles may have a capsule retention value on cotton fiber of at least 1 mg oil
- the emulsifier may be cationic
- the cationic emulsifier may be selected from palmitamidopropylt ⁇ monium chlo ⁇ de, distearyl dimonium chloride, cetylt ⁇ methylammonium chloride, quaternary ammonium compounds, fatty amines, aliphatic ammonium hahdes, alkyldimethylbenzylammonium hahdes, alkyldimethylethylammonium hahdes, polyethyleneimine, poly(2-dimethylamino)ethyl methacrylate) methyl chloride quaternary salt, poly( 1 -vinylpyrrolidone-co ⁇ -dimethylaminoethyl methacrylate), poly(acry lamide-co- diallyldimethylammonium chloride), poly(allylamine), poly[bis(2-chloroethyl) ether-alt- 1,3- bis[3-(dimethylamino)propyl]urea]
- the soluble acid and amine acrylate may have a percent by weight as compared to the weight of the wall material of from 1 to 5 percent
- the soluble acid and amine acrylate may have a molar proportion of from 1 25/1 to 1/1 25
- the non-anionic emulsifier may comp ⁇ se a water soluble or dispersible material at a pH of from 8-10
- the emulsifier may be nonionic
- the nonionic emulsifier may be selected from polyalkylene glycol ether, condensation products of alkyl phenols, aliphatic alcohols, or fatty acids with alkylene oxide, ethoxylated alkyl phenols, ethoxylated arylphenols, ethoxylated polyaryl phenols, carboxyhc esters solubihzed with a polyol, polyvinyl alcohol, polyvinyl acetate, copolymers of polyvinyl alcohol, copolymers of polyvinyl acetate, polyacrylamide, poly(N-isopropylacrylamide), poly(2-hydroxypropyl methacrylate), poly(2-ethyl-2-oxazohne), poly(2-isopropenyl-2-oxazohne-co-methyl methacrylate), poly(methyl vinyl ether), and polyvinyl alcohol-co-ethylene)
- the soluble acid and amine acrylate may have a percent by weight as compared to the weight of the wall mate ⁇ al of from 1 to 5 percent
- the soluble acid and amine acrylate may have a molar proportion of from 1 25/1 to 1/1 25
- the emulsifier may have a molecular weight greater than about 100, and may be selected from polymers with primary, secondary or tertiary amine functionality
- the emulsifier may have a molecular weight greater than 100 and may be selected from polymers having a hydroxyl, ether, ester, ketone or amide functionality
- the nonionic emulsifier may be partially hydrolyzed polyvinyl alcohol in the range of from 85 to 95% hydrolyzed
- said population of microcapsules may have a capsule retention value on cotton fiber of at least 1 mg oil
- said particles may be contained in a slurry that is combined with said adjunct
- said slurry may comp ⁇ se one or more processing aids, selected from the group consisting of water, aggregate inhibiting mate ⁇ als such as divalent salts, particle suspending polymers such as xanthan gum, guar gum, caboxy methyl cellulose
- said particles may be contained in an agglomerate that is combined with said adjunct
- said agglomerate may comprise materials selected from the group consisting of silicas, citric acid, sodium carbonate, sodium sulfate, sodium chlo ⁇ de, and binders such as sodium silicates, modified celluloses, polyethylene glycols, polyacrylates, polyacrylic acids, zeolites and mixtures thereof
- said adjunct may be selected from the group consisting of polymers, in one aspect, a cationic polymer, surfactants, builders, chelating agents, dye transfer inhibiting agents, dispersants, enzymes, enzyme stabilizers, catalytic materials, bleach activators, polymeric dispersing agents, clay soil removal/anti-redeposition agents, b ⁇ ghteners, dye polymer conjugates, dye clay conjugates, suds suppressors, dyes, bleach catalysts, additional perfume and/or perfume delivery systems, structure elasticizing agents, fab ⁇ c softeners, earners, hydrotropes, processing aids, rheology modifiers, structurants, thickeners, pigments, water and mixtures thereof
- said composition may comp ⁇ se a material selected from the group consisting of dyes, perfume, optical b ⁇ ghteners, rheology modifiers, structurants, thickeners, deposition aids, and mixtures thereof
- said composition may comprise a deposition aid that may comprise a polymer selected from the group comprising polysaccha ⁇ des, in one aspect, cationically modified starch and/or cationically modified guar, polysiloxanes, poly diallyl dimethyl ammonium hahdes, copolymers of poly diallyl dimethyl ammonium chlo ⁇ de and polyvinyl pyrrohdone, a composition comp ⁇ sing polyethylene glycol and polyvinyl pyrrohdone, acrylamides, imidazoles, lmidazohnium hahdes, polyvinyl amine, copolymers of poly vinyl amine and N-vinyl formamide, polyvinylformamide, polyvinyl alcohol, polyvinyl alcohol crosslinked with boric acid, polyacrylic acid, polyglycerol ether silicone crosspolymers, polyacrylic acids, polyacrylates, copolymers of polyvinylamine and polvyinylalcohol
- At least 75% of said particles may have a fracture strength of from about 0 2 MPa to about 30 MPa, from about 0 6 MPa to about 10MPa, from about 1 0 MPa to about 5 MPa, or from about 1 2 MPa to about 3MPa
- said composition comprises a rheology modifier, thickener and/or structurant having a high shear viscosity, at 20 sec-1 shear rate and at 21°C, of from 1 to 7000 cps and a viscosity at low shear (0 5 sec-1 shear rate at 21°C) of greater than 1000 cps, or even 1000 cps to 200,000 cps
- rheology modifiers impart to the aqueous liquid composition a high shear viscosity, at 20 sec-1 and at 21°C, of from 50 to 3000 cps and a viscosity at low shear (0 5 sec- 1 shear rate at 21°C) of greater than 1000 cps, or even 1000 cps to 200,000 cps
- suitable rheology modifiers, thickeners and/or structurants may be selected from the group consisting of polyacrylates, polymethacrylates, polycarboxylates
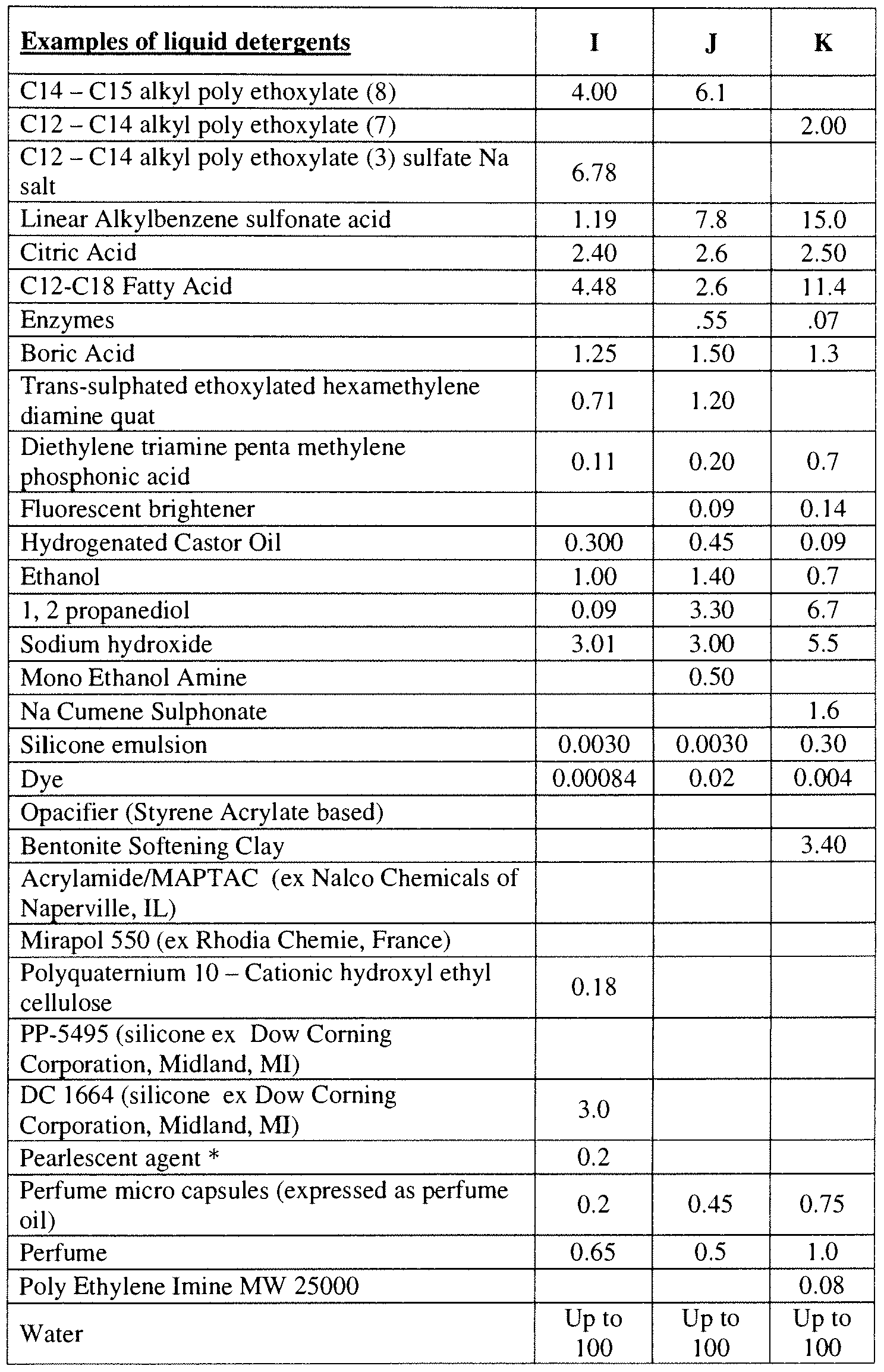
- said composition may be a fluid detergent and that may comprise, based on total fluid detergent weight, less than about less then about 80% water, less than about 60% to about 2% water, from about 45% to about 7% water, from about 35% to about 9% water
- said composition may have a viscosity of from about 10 cps to about 999 cps, or even from about 100 cps to about 800 cps at a shear rate of 1 sec -1
- said composition may be a gel comprising, based on total gel weight, less than about 45% water less than about 45% to about 2% water, from about 45% to about 7% water, from about 35% to about 9% water and may have a neat viscosity of from about 1 ,000 cps to about 10,000 cps or even from about 1,200 cps to about 8,000 cps,
- said composition may be a fluid fab ⁇ c enhancer, a solid fab ⁇ c enhancer, a fluid shampoo, a solid shampoo, hair conditioner, body wash, solid antiperspirant, fluid antiperspirant, solid deodorant, fluid deodorant, fluid detergent, solid detergent, fluid hard surface cleaner, solid hard surface cleaner, or a unit dose detergent comp ⁇ sing a detergent and a water soluble film encapsulating said detergent
- compositions disclosed herein may have any combination of materials and/or characteristics disclosed herein
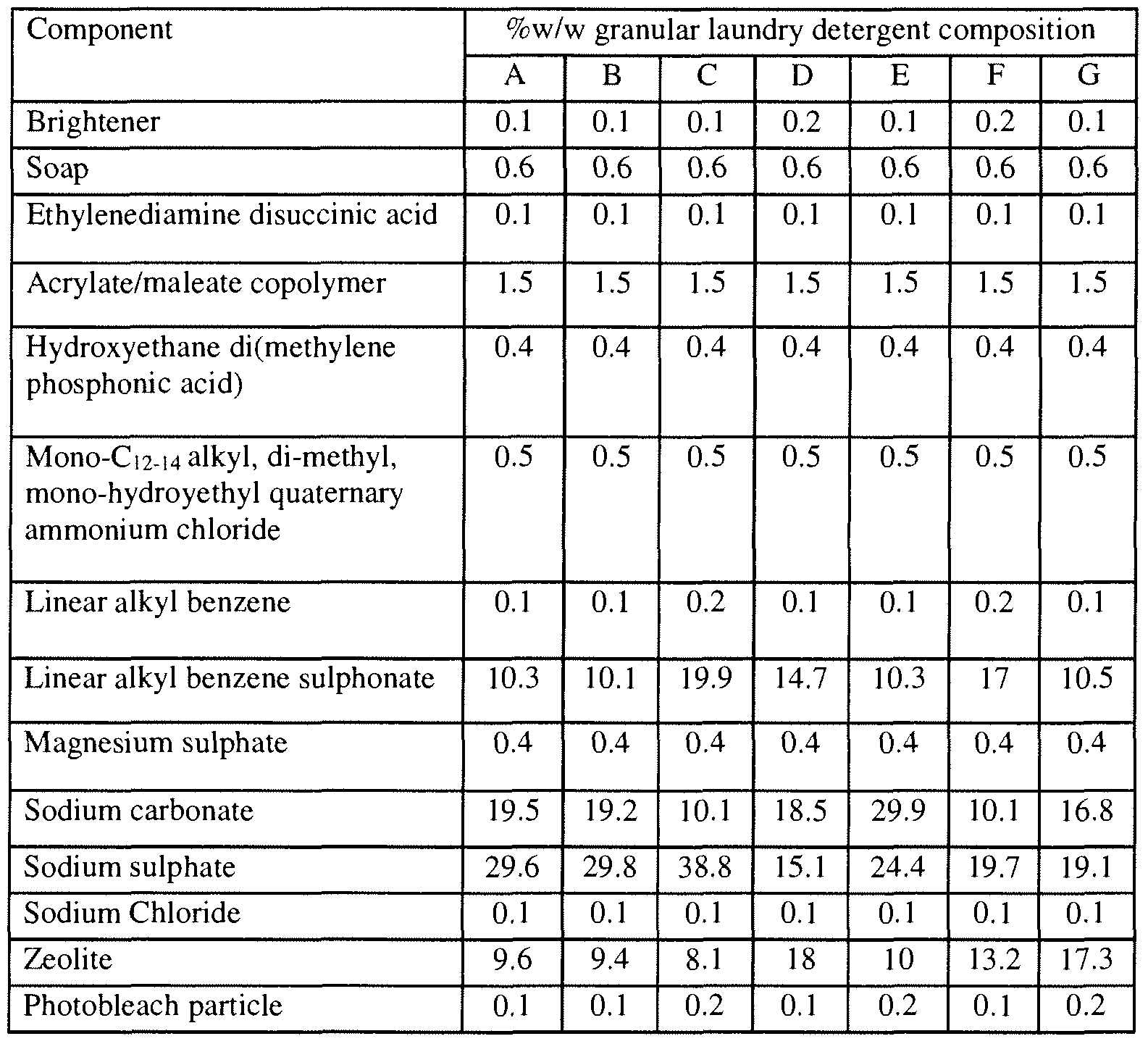
- aspects of the invention include the use of the particles of the present invention in laundry detergent compositions (e g , ⁇ DETM), hard surface cleaners (e g , MR CLEANTM), automatic dishwashing liquids (e g , CASCADETM), and floor cleaners (e g , SWIFFERTM)
- cleaning compositions may include those described in U S Pat Nos 4,515,705, 4,537,706, 4,537,707, 4,550,862, 4,561,998, 4,597,898, 4,968,451 , 5,565, 145, 5,929,022, 6,294,514, and 6,376,445
- the cleaning compositions disclosed herein are typically formulated such that, du ⁇ ng use in aqueous cleaning operations, the wash water will have a pH of between about 6 5 and about 12, or between about 7 5 and 10 5
- Liquid dishwashing product formulations typically have a pH between about 6 8 and about 9 0
- Cleaning products are typically formulated to have a pH of from about 7 to
- compositions of the present invention can be applied to the skin and/or hair
- the compositions can be, for example, formulated as bars, liquids, emulsions, shampoos, gels, powders, sticks, hair conditioners ( ⁇ nse off and leave in), hair tonics, pastes, hair colorants, sprays, mousses and/or other styling products Personal Care Compositions
- the consumer products disclosed herein may be personal care compositions comprising any aspect of the particles described in the present specification
- Such compositions may be in solid or fluid form
- Such compositions can be applied to the skin and/or hair or in other embodiments used to treat and/clean a situs
- the compositions can be, for example, formulated as bars, liquids, emulsions, shampoos, gels, powders, sticks, hair conditioners (rinse off and leave in), hair tonics, pastes, hair colorants, sprays, mousses and other styling products
- the particle is incorporated into a personal care composition suitable for use before, during or after hair removal
- the personal care composition of the present invention can be used in combination with various hair removal applications (prior to, concurrently with, and/or after), including but not limited to shaving (wet or dry shaving, via electric razors, via powered or manual razors which can be reuseable or disposable, and combinations thereof), epilation, electrolysis, wax or depilatories as well as energy delivery devices to help regulate hair growth
- the hair removal composition can be an aerosol, such as an aerosol shave preparation which can be a foam, gel, or post foaming gel, or a non-aerosol shave preparation such as generally available in the market
- the shave preparation is an emulsion which can be in the form of a cream or lotion, or the shave preparation can be a gel, which most commonly consists of polymer thickened surfactant systems
- the particle is incorporated into a shaving aid which can be incorporated into a shaving razor cartridge
- shaving aids are also commonly referred to as lubricating strips Suitable shaving aids and/or lubricating strips are disclosed in U S Patents 7,069,658, 6,944,952, 6,594,904, 6,182,365, 6,185,822, 6,298,558 and 5,1 13,585, and U S Design Patent D424,745
- the shaving aid comprises from about 50% to about 95% of a lub ⁇ cious water soluble polymer, selected from the group consisting of polyethylene oxide, polyvinyl pyrrohdone, polyacrylamide, modified hydroxyalkyl cellulose, polyvinyl imidazoline, polyvinyl alcohol, polysulfone, polyhydroxyethyl-methacrylate, and mixture thereof
- the shaving aid may also include from about 1% to about 50% of a non-soluble polymer selected from the group consisting of polyethylene, polypropylene, poly
- compositions of the present inventions may include the following components: A. Detersive Surfactant
- the composition of the present invention may include a detersive surfactant.
- the detersive surfactant component may comprise anionic detersive surfactant, zwitterionic or amphoteric detersive surfactant, or a combination thereof.
- the concentration of the anionic surfactant component in the composition should be sufficient to provide the desired cleaning and lather performance, and generally range from about 5% to about 50%.
- Anionic surfactants suitable for use in the compositions are the alkyl and alkyl ether sulfates.
- Other suitable anionic detersive surfactants are the water-soluble salts of organic, sulfuric acid reaction products conforming to the formula [ RI -SO3-M ] where Rl is a straight or branched chain, saturated, aliphatic hydrocarbon radical having from about 8 to about 24, or about 10 to about 18, carbon atoms; and M is a cation described hereinbefore.
- anionic detersive surfactants are the reaction products of fatty acids esterified with isethionic acid and neutralized with sodium hydroxide where, for example, the fatty acids are derived from coconut oil or palm kernel oil; sodium or potassium salts of fatty acid amides of methyl tauride in which the fatty acids, for example, are derived from coconut oil or palm kernel oil.
- Other similar anionic surfactants are described in U.S. Pat. Nos. 2,486,921 ; 2,486,922; and 2,396,278.
- anionic detersive surfactants suitable for use in the compositions are the succinnates, examples of which include disodium N-octadecylsulfosuccinnate; disodium lauryl sulfosuccinate; diammonium lauryl sulfosuccinate; tetrasodium N-(l ,2-dicarboxyethyl)-N- octadecylsulfosuccinnate; diamyl ester of sodium sulfosuccinic acid; dihexyl ester of sodium sulfosuccinic acid; and dioctyl esters of sodium sulfosuccinic acid.
- succinnates examples of which include disodium N-octadecylsulfosuccinnate; disodium lauryl sulfosuccinate; diammonium lauryl sulfosuccinate; tetrasodium N-(l
- Suitable anionic detersive surfactants include olefin sulfonates having about 10 to about 24 carbon atoms.
- the olefin sulfonates can contain minor amounts of other materials, such as alkene disulfonates depending upon the reaction conditions, proportion of reactants, the nature of the starting olefins and impurities in the olefin stock and side reactions during the sulfonation process
- alpha-olefin sulfonate mixture is described in U S Patent 3,332,880
- anionic detersive surfactants suitable for use in the compositions is the beta-alkyloxy alkane sulfonates These surfactants conform to the formula
- Rl is a straight chain alkyl group having from about 6 to about 20 carbon atoms
- R ⁇ is a lower alkyl group having from about 1 to about 3 carbon atoms, or even 1 carbon atom
- M is a water-soluble cation as described hereinbefore
- the composition of the present invention may comprise a cationic surfactant system
- the cationic surfactant system can be one cationic surfactant or a mixture of two or more cationic surfactants If present, the cationic surfactant system is included in the composition at a level by weight of from about 0 1 % to about 10%, from about 0 5% to about 8%, from about 1 % to about 5%, or even from about 1 4% to about 4%, in view of balance among ease-to- ⁇ nse feel, rheology and wet conditioning benefits
- a vanety of cationic surfactants including mono- and di-alkyl chain cationic surfactants can be used in the compositions of the present invention
- suitable matenals include mono-alkyl chain cationic surfactants in view of the desired gel matrix and wet conditioning benefits
- the mono-alkyl cationic surfactants are those having one long alkyl chain which has from 12 to 22 carbon atoms, from 16 to 22 carbon atoms, or a C 18 -C 22 alkyl group, in view of providing balanced wet conditioning benefits
- the remaining groups attached to nitrogen are independently selected from an alkyl group of from 1 to about 4 carbon atoms or an alkoxy, polyoxyalkylene, alkylamido, hydroxyalkyl, aryl or alkylaryl group having up to about 4 carbon atoms
- Such mono-alkyl cationic surfactants include, for example, mono-alkyl quaternary ammonium salts and mono-alkyl amines
- Mono-long alkyl quaternized ammonium salts useful herein are those having the formula (ID (H) R 75
- R 7 l_i ⁇ _ R - X ⁇ R 77 wherein one of R 75 , R 76 , R 77 and R 78 is selected from an alkyl group of from 12 to 30 carbon atoms or an aromatic, alkoxy, polyoxyalkylene, alkylamido, hydroxyalkyl, aryl or alkylaryl group having up to about 30 carbon atoms, the remainder of R 75 , R 76 , R 77 and R 78 are independently selected from an alkyl group of from 1 to about 4 carbon atoms or an alkoxy, polyoxyalkylene, alkylamido, hydroxyalkyl, aryl or alkylaryl group having up to about 4 carbon atoms, and X is a salt-forming anion such as those selected from halogen, (e g chloride, bromide), acetate, citrate, lactate, glycolate, phosphate, nitrate, sulfonate, sulfate, alkylsul
- Suitable mono-long alkyl quaternized ammonium salt cationic surfactants include behenyl tnmethyl ammonium salt, stearyl t ⁇ methyl ammonium salt, cetyl t ⁇ methyl ammonium salt, and hydrogenated tallow alkyl t ⁇ methyl ammonium salt Among them, highly useful materials are behenyl tnmethyl ammonium salt and stearyl tnmethyl ammonium salt
- Mono-alkyl amines are also suitable as cationic surfactants
- Primary, secondary, and tertiary fatty amines are useful Particularly useful are tertiary amido amines having an alkyl group of from about 12 to about 22 carbons
- Exemplary tertiary amido amines include stearamidopropyldimethylamine, stearamidopropyldiethylamine, stearamidoethyldiethylamine, stearamidoethyldimethylamine, pal ⁇ utamidopropyldimethylamine, palmitamidopropyldiethylamine, palmitamidoethyldiethylamine, palmitamidoethyldimethylamine, behenamidopropyldimethylamine, behenamidopropyldiethylamine, behenamidoethyldiethylamine, behenamidoethyldimethylamine, arachid
- di-alkyl chain cationic surfactants include, for example, dialkyl (14-18) dimethyl ammonium chlo ⁇ de, ditallow alkyl dimethyl ammonium chlo ⁇ de, dihydrogenated tallow alkyl dimethyl ammonium chloride, distearyl dimethyl ammonium chloride, and dicetyl dimethyl ammonium chloride C High Melting Point Fatty Compound
- the composition of the present invention may include a high melting point fatty compound
- the high melting point fatty compound useful herein has a melting point of 25°C or higher, and is selected from the group consisting of fatty alcohols, fatty acids, fatty alcohol de ⁇ vatives, fatty acid de ⁇ vatives, and mixtures thereof It is understood by the artisan that the compounds disclosed in this section of the specification can in some instances fall into more than one classification, e g , some fatty alcohol derivatives can also be classified as fatty acid derivatives However, a given classification is not intended to be a limitation on that particular compound, but is done so for convenience of classification and nomenclature Further, it is understood by the artisan that, depending on the number and position of double bonds, and length and position of the branches, certain compounds having certain required carbon atoms may have a melting point of less than 25°C Such compounds of low melting point are not intended to be included in this section
- fatty alcohols are used in one aspect the present invention
- the fatty alcohols useful herein are those having from about 14 to about 30 carbon atoms, or even from about 16 to about 22 carbon atoms These fatty alcohols are saturated and can be straight or branched chain alcohols
- fatty alcohols include, for example, cetyl alcohol, stearyl alcohol, behenyl alcohol, and mixtures thereof
- High melting point fatty compounds of a single compound of high pu ⁇ ty are typically used
- single compounds of pure fatty alcohols selected from the group of pure cetyl alcohol, stearyl alcohol, and behenyl alcohol are employed By "pure” herein, what is meant is that the compound has a purity of at least about 90%, or even at least about 95%
- the high melting point fatty compound is included in the composition at a level of from about 0 1 % to about 40%, from about 1% to about 30%, from about 1 5% to about 16% by weight of the composition, or even from about 1 5% to about 8% in view of providing improved conditioning benefits such as slippery feel during the application to wet hair, softness and moistu ⁇ zed feel on dry hair D Cationic Polymers
- compositions of the present invention may contain a cationic polymer
- Concentrations of the cationic polymer in the composition typically range from about 0 05% to about 3%, in another embodiment from about 0 075% to about 2 0%, and in yet another embodiment from about 0 1 % to about 1 0%
- Suitable cationic polymers will have cationic charge densities of at least about 0 5 meq/gm, in another embodiment at least about 0 9 meq/gm, in another embodiment at least about 1 2 meq/gm, in yet another embodiment at least about 1 5 meq/gm, but in one embodiment also less than about 7 meq/gm, and in another embodiment less than about 5 meq/gm, at the pH of intended use of the composition, which pH will generally range from about pH 3 to about pH 9, in one embodiment between about pH 4 and about pH 8
- cationic charge density refers to the ratio of the number of positive charges on the polymer to the molecular weight of the polymer The average molecular weight
- Suitable cationic polymers for use in the compositions of the present invention contain cationic nitrogen-containing moieties such as quaternary ammonium or cationic protonated amino moieties
- the cationic protonated amines can be primary, secondary, or tertiary amines (in one aspect, secondary or tertiary), depending upon the particular species and the selected pH of the composition
- Any anionic counte ⁇ on can be used in association with the cationic polymers so long as the polymers remain soluble in water, in the composition, or in a coacervate phase of the composition, and so long as the counte ⁇ ons are physically and chemically compatible with the essential components of the composition or do not otherwise unduly impair product performance, stability or aesthetics
- Non limiting examples of such counte ⁇ ons include halides (e g , chlo ⁇ de, fluoride, bromide, iodide), sulfate and methyl sulfate
- Non limiting examples of suitable canonic polymers include copolymers of vinyl monomers having canonic protonated amine or quaternary ammonium functionalities with water soluble spacer monomers such as acrylamide, methacrylamide, alkyl and dialkyl acrylamides, alkyl and dialkyl methacrylamides, alkyl acrylate, alkyl methacrylate, vinyl caprolactone or vinyl pyrrolidone
- Suitable canonic protonated amino and quaternary ammonium monomers for inclusion in the canonic polymers of the composition herein, include vinyl compounds substituted with dialkylaminoalkyl acrylate, dialkylaminoalkyl methacrylate, monoalkylaminoalkyl acrylate, monoalkylaminoalkyl methacrylate, t ⁇ alkyl methacryloxyalkyl ammonium salt, t ⁇ alkyl acryloxyalkyl ammonium salt, diallyl quaternary ammonium salts, and vinyl quaternary ammonium monomers having cyclic cationic nitrogen-containing ⁇ ngs such as py ⁇ dinium, lirudazolium, and quaternized pyrrolidone, e g , alkyl vinyl lmidazolium, alkyl vinyl py ⁇ dinium, alkyl vinyl pyrrolidone salts
- Suitable cationic polymers for use in the compositions include copolymers of 1- vinyl-2-pyrrolidone and l -vinyl-3-methylimidazolium salt (e g , chlonde salt) (referred to in the industry by the Cosmetic, Toiletry, and Fragrance Association, "CTFA", as Polyquaternium-16), copolymers of l -vinyl-2-pyrrolidone and dimethylaminoethyl methacrylate (referred to in the industry by CTFA as Polyquaternium- 1 1), cationic diallyl quaternary ammonium-containing polymers, including, for example, dimethyldiallylammonium chlonde homopolymer, copolymers of acrylamide and dimethyldiallylammonium chloride (referred to in the industry by CTFA as Polyquaternium 6 and Polyquaternium 7, respectively), amphoteric copolymers of acrylic acid including copolymers of acrylic acid and dimethyldiallylammonium chlo
- R 1 is hydrogen, methyl or ethyl
- each of R 2 , R 3 and R 4 are independently hydrogen or a short chain alkyl having from about 1 to about 8 carbon atoms, from about 1 to about 5 carbon atoms, or even from about 1 to about 2 carbon atoms
- n is an integer having a value of from about 1 to about 8, or even from about 1 to about 4
- X is a countenon
- the nitrogen attached to R 2 , R 3 and R 4 may be a protonated amine (primary, secondary or tertiary), but is in one aspect, a quaternary ammonium wherein each of R 2 , R 3 and R 4 are alkyl groups a non limiting example of which is polymethacrylamidopropyl tnmonium chloride, available under the trade name Polycare ® 133, from Rhone-Poulenc, Cranberry, N J 1 U S A
- Suitable cationic polymers for use in the composition include polysaccharide polymers, such as cationic cellulose derivatives and cationic starch derivatives Suitable cationic polysaccharide polymers include those which conform to the formula
- A is an anhydroglucose residual group, such as a starch or cellulose anhydroglucose residual
- R is an alkylene oxyalkylene, polyoxyalkylene, or hydroxy alky lene group, or combination thereof
- R 1 , R 2 , and R 3 independently are alkyl, aryl, alkylaryl, arylalkyl, alkoxyalkyl, or alkoxyaryl groups, each group containing up to about 18 carbon atoms, and the total number of carbon atoms for each cationic moiety (i e , the sum of carbon atoms in Rl , R2 and R3) is typically about 20 or less
- X is an anionic countenon as described in hereinbefore
- Useful cationic cellulose polymers include salts of hydroxyethyl cellulose reacted with tnmethyl ammonium substituted epoxide, referred to in the industry (CTFA) as Polyquaternium 10 and available from Amerchol Corp (Edison, N J , USA) in their UcareTM Polymer LR, UcareTM Polymer JR, and UcareTM Polymer KG series of polymers
- CTFA tnmethyl ammonium substituted epoxide
- Other suitable types of cationic cellulose include the polymeric quaternary ammonium salts of hydroxyethyl cellulose reacted with lauryl dimethyl ammonium-substituted epoxide referred to in the industry (CTFA) as Polyquaternium 24 These materials are available from Amerchol Corp under the trade name UcareTM Polymer LM-200
- Suitable cationic polymers include cationic guar gum derivatives, such as guar hydroxypropylt ⁇ monium chloride, specific examples of which include the Jaguar se ⁇ es commercially available from Rhone-Poulenc Incorporated and the N-Hance series commercially available from Aqualon Division of Hercules, Inc
- suitable cationic polymers include quaternary nitrogen-containing cellulose ethers, some examples of which are described in U S Pat No 3,962,418
- Other suitable polymers include synthetic polymers such as those disclosed in U S Publication No 2007/0207109Al
- suitable cationic polymers include copolymers of ethe ⁇ fied cellulose, guar and starch, some examples of which are described in U S Pat No 3,958,581 When used, the cationic polymers herein are either soluble in the composition or are soluble in a complex coacervate phase in the composition formed by the cationic polymer and the anionic, amphoteric and/or zwitte ⁇
- composition of the present invention may include a nonionic polymer
- Polyalkylene glycols having a molecular weight of more than about 1000 are useful herein Useful are those having the following general formula wherein R is selected from the group consisting of H, methyl, and mixtures thereof
- Polyethylene glycol polymers useful herein are PEG-2M (also known as Polyox WSR ® N-IO, which is available from Union Carbide and as PEG-2,000), PEG-5M (also known as Polyox WSR ® N-35 and Polyox WSR ® N-80, available from Union Carbide and as PEG-5,000 and Polyethylene Glycol 300,000), PEG-7M (also known as Polyox WSR ® N-750 available from Union Carbide), PEG-9M (also known as Polyox WSR ® N-3333 available from Union Carbide), and PEG- 14 M (also known as Polyox WSR ® N-3000 available from Union Carbide) F Conditioning agents
- Conditioning agents include any material which is used to give a particular conditioning benefit to hair and/or skin
- suitable conditioning agents are those which deliver one or more benefits relating to shine, softness, compatibility, antistatic properties, wet- handling, damage, manageability, body, and greasiness
- the conditioning agents useful in the compositions of the present invention typically comprise a water insoluble, water dispersible, non-volatile, liquid that forms emulsified, liquid particles
- Suitable conditioning agents for use in the composition are those conditioning agents characterized generally as silicones (e g , silicone oils, cationic silicones, silicone gums, high refractive silicones, and silicone resins), organic conditioning oils (e g , hydrocarbon oils, polyolefins, and fatty esters) or combinations thereof, or those conditioning agents which otherwise form liquid, dispersed particles in the aqueous surfactant matrix herein
- Such conditioning agents should be physically and chemically compatible with the essential components of the composition, and should not otherwise
- the concentration of the conditioning agent in the composition should be sufficient to provide the desired conditioning benefits, and as will be apparent to one of ordinary skill in the art Such concentration can vary with the conditioning agent, the conditioning performance desired, the average size of the conditioning agent particles, the type and concentration of other components, and other like factors 1 Silicones
- the conditioning agent of the compositions of the present invention can be an insoluble silicone conditioning agent
- the silicone conditioning agent particles may comp ⁇ se volatile silicone, non-volatile silicones, or combinations thereof
- non-volatile silicones conditioning agents are employed If volatile silicones are present, it will typically be incidental to their use as a solvent or carrier for commercially available forms of non-volatile silicone materials ingredients, such as silicone gums and resins
- the silicone conditioning agent particles may comprise a silicone fluid conditioning agent and may also comp ⁇ se other ingredients, such as a silicone resin to improve silicone fluid deposition efficiency or enhance glossiness of the hair
- the concentration of the silicone conditioning agent typically ranges from about 0 01 % to about 10%, from about 0 1% to about 8%, from about 0 1 % to about 5%, or even from about 0 2% to about 3%
- suitable silicone conditioning agents, and optional suspending agents for the silicone are described in U S Reissue Pat No 34,584, U S Pat No 5, 104,646, and U S Pat No 5, 106,609
- the silicone conditioning agents for use in the compositions of the present invention typically have a viscosity, as measured at 25 0 C, from about 20 centistokes to about 2,000,000 centistokes ("cst"), from about 1,000 cst to about 1 ,800,000 cst, from about 50,000cst to about 1 ,500,000 cst, or even from about 100,000 cst to about 1,500,000 csk
- the dispersed silicone conditioning agent particles typically have a number average particle diameter ranging from about 0 Ol ⁇ m to about 50 ⁇ m
- the number average particle diameters typically range from about O Ol ⁇ m to about 4 ⁇ m, from about O Ol ⁇ m to about 2 ⁇ m, or even from about O Ol ⁇ m to about 0 5 ⁇ m
- the number average particle diameters typically range from about 4 ⁇ m to about 50 ⁇ m, from about 6 ⁇ m to about 30 ⁇ m, from about 9 ⁇ m to about 20 ⁇ m, or even from about 12 ⁇ m to about 18 ⁇ m a Silicone oils
- Silicone fluids may include silicone oils, which are flowable silicone mate ⁇ als having a viscosity, as measured at 25°C, less than 1 ,000,000 cst, from about 5 cst to about 1 ,000,000 cst, or even from about 100 cst to about 600,000 cst
- Suitable silicone oils for use in the compositions of the present invention include polyalkyl siloxanes, polyaryl siloxanes, polyalkylaryl siloxanes, polyether siloxane copolymers, and mixtures thereof
- Other insoluble, non-volatile silicone fluids having hair conditioning properties may also be used b Amino and Cationic silicones
- compositions of the present invention may include an aminosihcone
- Aminosilicones are silicones containing at least one primary amine, secondary amine, tertiary amine, or a quaternary ammonium group
- Useful aminosilicones may have less than about 0 5% nitrogen by weight of the aminosihcone, less than about 0 2%, or even less than about 0 1 %
- Higher levels of nitrogen (amine functional groups) in the amino silicone tend to result in less fnction reduction, and consequently less conditioning benefit from the aminosihcone It should be understood that in some product forms, higher levels of nitrogen are acceptable in accordance with the present invention
- the aminosilicones used m the present invention have a particle size of less than about 50 ⁇ once incorporated into the final composition
- the particle size measurement is taken from dispersed droplets in the final composition Particle size may be measured by means of a laser light scattering technique, using a Ho ⁇ ba model LA-930 Laser Scatte ⁇ ng Particle Size Distribution Analyzer (Ho ⁇ ba Instruments, Inc )
- the aminosilicone typically has a viscosity of from about 1 ,000 cst (centistokes) to about 1 ,000,000 cst, from about 10,000 to about 700,000 cst, from about 50,000 cst to about 500,000 cst, or even from about 100,000 cst to about 400,000 cst
- This embodiment may also comprise a low viscosity fluid, such as, for example, those materials described below in Section F (I)
- the viscosity of arranosilicones discussed herein is measured at 25 0 C
- the aminosilicone typically has a viscosity of from about 1,000 cst to about 100,000 cst, from about 2,000 cst to about 50,000 cst, from about 4,000 cst to about 40,000 cst, or even from about 6,000 cst to about 30,000 cs
- the aminosilicone typically is contained in the composition of the present invention at a level by weight of from about 0 05% to about 20%, from about 0 1% to about 10%, and or even from about 0 3% to about 5% c Silicone gums
- silicone fluids suitable for use in the compositions of the present invention are the insoluble silicone gums These gums are polyorganosiloxane materials having a viscosity, as measured at 25 0 C, of greater than or equal to 1,000,000 csk
- Specific non-1 imiting examples of silicone gums for use in the compositions of the present invention include polydimethylsiloxane, (polydimethylsiloxane) (methylvinylsiloxane) copolymer, poly(dimethylsiloxane) (diphenyl siloxane)(methylvinylsiloxane) copolymer and mixtures thereof d High refractive index silicones
- non-volatile, insoluble silicone fluid conditioning agents that are suitable for use in the compositions of the present invention are those known as "high refractive index silicones," having a refractive index of at least about 1 46, at least about 1 48, m at least about 1 52, or even at least about 1 55
- the refractive index of the polysiloxane fluid will generally be less than about 1 70, typically less than about 1 60
- polysiloxane "fluid” includes oils as well as gums
- the high refractive index polysiloxane fluid includes those represented by general Formula (III) above, as well as cyclic polysiloxanes such as those represented by Formula (VIII) below
- R is as defined above, and n is a number from about 3 to about 7, or even from about 3 to about 5.
- Silicone fluids suitable for use in the compositions of the present invention are disclosed in U.S. Pat. No. 2,826,551 , U.S. Pat. No. 3,964,500, and U.S. Pat. No. 4,364,837. e. Silicone resins
- Silicone resins may be included in the conditioning agent of the compositions of the present invention. These resins are highly cross-linked polymeric siloxane systems. The cross- linking is introduced through the incorporation of trifunctional and tetrafunctional silanes with monofunctional or difunctional, or both, silanes during manufacture of the silicone resin.
- Silicone materials and silicone resins in particular can conveniently be identified according to a shorthand nomenclature system known to those of ordinary skill in the art as "MDTQ" nomenclature. Under this system, the silicone is described according to presence of various siloxane monomer units which make up the silicone. Briefly, the symbol M denotes the monofunctional unit (CH 3 ) 3 SiOos; D denotes the difunctional unit (CH 3 ⁇ SiO; T denotes the trifunctional unit (CH 3 )SiO) 5 ; and Q denotes the quadra- or tetra-functional unit SiC> 2 . Primes of the unit symbols (e.g. M', D', T', and Q') denote substituents other than methyl, and must be specifically defined for each occurrence.
- silicone resins for use in the compositions of the present invention include, but are not limited to MQ, MT, MTQ, MDT and MDTQ resins.
- Methyl is a highly suitable silicone substituent.
- silicone resins are typically MQ resins, wherein the M:Q ratio is typically from about 0.5: 1.0 to about 1.5: 1.0 and the average molecular weight of the silicone resin is typically from about 1000 to about 10,000. f. Modified silicones or silicone copolymers
- modified silicones or silicone copolymers are also useful herein.
- examples of these include silicone-based quaternary ammonium compounds (Kennan quats) disclosed in U.S. Patent Nos. 6,607,717 and 6,482,969; end-terminal quaternary siloxanes; silicone aminopolyalkyleneoxide block copolymers disclosed in U.S. Patent Nos.
- sihcone-based quaternary ammonium compounds may be combined with the silicone polymers desc ⁇ bed in US Patent Nos 7,041 ,767 and 7,217,777 and US Application number 2007/0041929Al
- compositions of the present invention may also comprise from about 0 05% to about 3%, from about 0 08% to about 1 5%, or even from about 0 1 % to about 1 %, of at least one organic conditioning oil as the conditioning agent, either alone or in combination with other conditioning agents, such as the silicones (described herein)
- Suitable conditioning oils include hydrocarbon oils, polyolefins, and fatty esters
- Suitable hydrocarbon oils include, but are not limited to, hydrocarbon oils having at least about 10 carbon atoms, such as cyclic hydrocarbons, straight chain aliphatic hydrocarbons (saturated or unsaturated), and branched chain aliphatic hydrocarbons (saturated or unsaturated), including polymers and mixtures thereof
- Straight chain hydrocarbon oils are typically from about C 12 to about C 19
- Suitable polyolefins include liquid polyolefins, liquid poly- ⁇ -o
- conditioning agents described by the Procter & Gamble Company in U S Pat Nos 5,674,478, and 5,750, 122
- compositions of the present invention may also contain an anti-dandruff agent
- anti-dandruff actives include antimicrobial actives, py ⁇ dinethione salts, azoles, selenium sulfide, particulate sulfur, keratolytic acid, salicylic acid, octopirox (piroctone olamine), coal tar, and combinations thereof
- the anti- dandruff actives typically are py ⁇ dinethione salts
- Such anti-dandruff particulate should be physically and chemically compatible with the essential components of the composition, and should not otherwise unduly impair product stability, aesthetics or performance
- Py ⁇ dinethione anti-dandruff agents are desc ⁇ bed, for example, in U S Pat No 2,809,971 , U S Pat No 3,236,733, U S Pat No 3,753,196, U S Pat No 3,761 ,418, U S Pat No 4,345,080, U S Pat No 4,323,683, U S Pat No 4,379,753, and U S Pat No 4,470,982 It is contemplated that when ZPT is used as the anti-dandruff particulate in the compositions herein, that the growth or re-growth of hair may be stimulated or regulated, or both, or that hair loss may be reduced or inhibited, or that hair may appear thicker or fuller H Humectant
- compositions of the present invention may contain a humectant
- humectants herein are selected from the group consisting of polyhyd ⁇ c alcohols, water soluble alkoxylated nonionic polymers, and mixtures thereof
- the humectants, when used herein, are typically used at levels of from about 0 1 % to about 20%, or even from about 0 5% to about 5% I Suspending Agent
- compositions of the present invention may further comprise a suspending agent at concentrations effective for suspending water-insoluble material in dispersed form in the compositions or for modifying the viscosity of the composition Such concentrations range from about 0 1 % to about 10%, or even from about 0 3% to about 5 0%
- Suspending agents useful herein include anionic polymers and nonionic polymers
- vinyl polymers such as cross linked acrylic acid polymers with the CTFA name Carbomer, cellulose derivatives and modified cellulose polymers such as methyl cellulose, ethyl cellulose, hydroxyethyl cellulose, hydroxypropyl methyl cellulose, nitro cellulose, sodium cellulose sulfate, sodium carboxymethyl cellulose, crystalline cellulose, cellulose powder, polyvinylpyrrolidone, polyvinyl alcohol, guar gum, hydroxypropyl guar gum, xanthan gum, arabia gum, tragacanth, galactan, carob gum, guar gum, karaya gum, carrageenan, pectin, agar, quince seed (Cydonia oblonga Mill), starch (rice, corn, potato, wheat), algae colloids (algae extract), microbiological polymers such as dextran, succinoglucan, pulleran, star
- viscosity modifiers highly useful herein include Carbomers with trade names Carbopol ® 934, Carbopol ® 940, Carbopol ® 950, Carbopol ® 980, and Carbopol ® 981 , all available from B F Good ⁇ ch Company, acrylates/steareth-20 methacrylate copolymer with trade name ACRYSOLTM 22 available from Rohm and Hass, nonoxynyl hydroxyethylcellulose with trade name AmercellTM POLYMER HM- 1500 available from Amerchol, methylcellulose with trade name BENECEL ® , hydroxyethyl cellulose with trade name NATROSOL ® , hydroxypropyl cellulose with trade name KLUCEL ® , cetyl hydroxyethyl cellulose with trade name POLYSURF ® 67, all supplied by Hercules, ethylene oxide and/or propylene oxide based polymers with trade names CARBOW AX ® PEGs, POLYSURF
- suspending agents include crystalline suspending agents which can be categorized as acyl de ⁇ vatives, long chain amine oxides, and mixtures thereof These suspending agents are desc ⁇ bed in U S Pat No 4,741 ,855
- suspending agents include ethylene glycol esters of fatty acids in one aspect having from about 16 to about 22 carbon atoms
- useful suspending agents include ethylene glycol stearates, both mono and distearate, but in one aspect, the distearate containing less than about 7% of the mono stearate
- suitable suspending agents include alkanol amides of fatty acids, having from about 16 to about 22 carbon atoms, or even about 16 to 18 carbon atoms, examples of which include stea ⁇ c monoethanolamide, stea ⁇ c diethanolarrude, stearic monoisopropanolamide and stea ⁇ c monoethanolamide stearate
- Other long chain acyl de ⁇ vatives include long chain esters of long chain fatty acids (e g , stearyl stearate, cetyl palmitate, etc ), long chain esters of long chain alkanol amides (e g , stearamide diethanolamide distearate, stearamide monoethanol
- acyl derivatives suitable for use as suspending agents include N, N- dihydrocarbyl amido benzoic acid and soluble salts thereof (e g , Na, K), particularly N 1 N- di(hydrogenated) C
- Suitable long chain amine oxides for use as suspending agents include alkyl dimethyl amine oxides, e g , stearyl dimethyl amine oxide
- suspending agents include primary amines having a fatty alkyl moiety having at least about 16 carbon atoms, examples of which include palmitamine or stearamine, and secondary amines having two fatty alkyl moieties each having at least about 12 carbon atoms, examples of which include dipalmitoylamine or di(hydrogenated tallow)amine
- suitable suspending agents include di(hydrogenated tallow)phthahc acid amide, and crosslinked maleic anhydnde-methyl vinyl ether copolymer J Aqueous Carrier
- compositions of the present invention can be in the form of pourable liquids (under ambient conditions) Such compositions will therefore typically comprise an aqueous earner, which is present at a level of from about 20% to about 95%, or even from about 60% to about 85%
- the aqueous earner may compnse water, or a miscible mixture of water and organic solvent, and in one aspect may comprise water with minimal or no significant concentrations of organic solvent, except as otherwise incidentally incorporated into the composition as minor ingredients of other essential or optional components
- the earner useful in the present invention includes water and water solutions of lower alkyl alcohols and polyhydric alcohols
- the lower alkyl alcohols useful herein are monohyd ⁇ c alcohols having 1 to 6 carbons, in one aspect, ethanol and isopropanol
- the polyhydric alcohols useful herein include propylene glycol, hexylene glycol, glycenn, and propane diol K Dispersed Particles
- compositions may optionally comprise particles
- the particles may be dispersed water-insoluble particles
- the particles may be inorganic, synthetic, or semi-synthetic In one embodiment, the particles have an average mean particle size of less than about 300 ⁇ m L Gel Matrix
- the above cationic surfactants together with high melting point fatty compounds and an aqueous carrier, may form a gel matrix in the composition of the present invention
- the gel matrix is suitable for providing various conditioning benefits such as slippery feel during the application to wet hair and softness and moistu ⁇ zed feel on dry hair
- the cationic surfactant and the high melting point fatty compound are contained at a level such that the weight ratio of the cationic surfactant to the high melting point fatty compound is in the range of, from about 1 1 to about 1 10, or even from about 1 1 to about 1 6 M Skin Care Actives
- the composition may comprise at least one skin care active, useful for regulating and/or improving the condition and/or appearance of mammalian skin
- the skin care active may be soluble in oil or water, and may be present primarily in the oil phase and/or in the aqueous phase
- Suitable actives include, but are not limited to, vitamins, peptides, sugar amines, sunscreens, oil control agents, tanning actives, anti-acne actives, desquamation actives, anti- celluhte actives, chelating agents, skin lightening agents, flavonoids, protease inhibitors, non- vitamin antioxidants and radical scavengers, hair growth regulators, anti-wrinkle actives, anti- atrophy actives, minerals, phytosterols and/or plant hormones, tyrosinase inhibitors, antiinflammatory agents, N-acyl amino acid compounds, antimicrobials, and antifungals
- the composition may comprise from about 0 001% to about 10%, alternatively from about 0 01 % to about 5%, of at least one vitamin
- vitamins means vitamins, provitamins, and their salts, isomers and denvatives
- suitable vitamins include vitamin B compounds (including B 1 compounds, B2 compounds, B3 compounds such as niacinamide, niacinnicotinic acid, tocopheryl nicotinate, Ci-Cis nicotinic acid esters, and nicotinyl alcohol, B5 compounds, such as panthenol or "pro-B5", pantothenic acid, pantothenyl, B6 compounds, such as pyroxidine, py ⁇ doxal, py ⁇ doxamine, carnitine, thiamine, riboflavin), vitamin A compounds, and all natural and/or synthetic analogs of Vitamin A, including retinoids, retinol, retinyl acetate, retiny
- composition may comprise one or more peptides
- peptide refers to peptides containing ten or fewer amino acids, their denvatives, isomers, and complexes with other species such as metal ions (for example, copper, zinc, manganese, and magnesium)
- metal ions for example, copper, zinc, manganese, and magnesium
- peptide refers to both naturally occurring and synthesized peptides
- the peptides are di-, tn-, tetra-, penta-, and hexa-peptides, their salts, isomers, denvatives, and mixtures thereof
- useful peptide derivatives include, but are not limited to, peptides derived from soy proteins, carnosine (beta-alanine-histidine), palmitoyl- lysine-threonine (pal-KT) and palmitoyl-lysine-threonine-threonine-lysine-se ⁇ ne (
- the composition may comprise a sugar amine, also known as amino sugars, and their salts, isomers, tautomers and derivatives
- Sugar amines can be synthetic or natural in ongin and can be used as pure compounds or as mixtures of compounds ⁇ e g , extracts from natural sources or mixtures of synthetic materials)
- glucosamine is generally found in many shellfish and can also be derived from fungal sources
- sugar amines include glucosamine, N-acetyl glucosamine, mannosamine, N-acetyl mannosamine, galactosamine, N- acetyl galactosamine, their isomers ⁇ e g , stereoisomers), and their salts (e g , HCl salt)
- Other sugar amine compounds useful in skin care compositions include those descnbed in U S Patent No 6,159,485, issued to Yu, et al
- the composition may comprise from about 0.01 % to about 15%, alternatively from about 0.1% to about
- the composition may comprise one or more sunscreen actives (or sunscreen agents) and/or ultraviolet light absorbers.
- suitable sunscreen actives include oil-soluble sunscreens, insoluble sunscreens, and water-soluble sunscreens.
- the composition may comprise from about 1 % to about 20%, or, alternatively, from about 2% to about 10%, by weight of the composition, of the sunscreen active and/or ultraviolet light absorber. Exact amounts will vary depending upon the chosen sunscreen active and/or ultraviolet light absorber and the desired Sun Protection Factor (SPF), and are within the knowledge and judgment of one of skill in the art.
- SPF Sun Protection Factor
- Non-limiting examples of suitable oil-soluble sunscreens include benzophenone-3, bis- ethylhexyloxyphenol methoxyphenyl triazine, butyl methoxydibenzoyl-methane, diethylamino hydroxy-benzoyl hexyl benzoate, drometrizole trisiloxane, ethylhexyl methoxy-cinnamate, ethylhexyl salicylate, ethylhexyl triazone, octocrylene, homosalate, polysilicone-15, and derivatives and mixtures thereof.
- Non-limiting examples of suitable insoluble sunscreens include methylene bis- benzotriazolyl tetramethylbutyl-phenol, titanium dioxide, zinc cerium oxide, zinc oxide, and derivatives and mixtures thereof.
- Non-limiting examples of suitable water-soluble sunscreens include phenylbenzimidazole sulfonic acid (PBSA), terephthalylidene dicamphor sulfonic acid, (MexorylTM SX), benzophenone-4, benzophenone-5, benzylidene camphor sulfonic acid, cinnamidopropyl-trimonium chloride, methoxycinnamido-propyl ethyldimonium chloride ether, disodium bisethylphenyl triaminotriazine stilbenedisulfonate, disodium distyrylbiphenyl disulfonate, disodium phenyl dibenzimidazole tetrasulfonate, methoxycinnamido-propyl hydroxysultaine, methoxycinnamido-propyl laurdimonium tosylate, PEG-25 PABA (p- aminobenzoic
- the composition may comprise one or more compounds for regulating the production of skin oil, or sebum, and for improving the appearance of oily skin.
- suitable oil control agents include salicylic acid, dehydroacetic acid, benzoyl peroxide, vitamin B3 compounds (for example, niacinamide or tocopheryl nicotinate), their isomers, esters, salts and derivatives, and mixtures thereof.
- the compositions may comprise from about 0.0001 % to about 15%, alternatively from about 0.01 % to about 10%, alternatively from about 0.1 % to about 5%, and alternatively from about 0.2% to about 2%, of an oil control agent.
- the composition may comprise a tanning active.
- the compositions may comprise from about 0.1 % to about 20%, from about 2% to about 7%, or, alternatively, from about 3% to about 6%, by weight of the composition, of a tanning active.
- a suitable tanning active includes dihydroxyacetone, which is also known as DHA or l ,3-dihydroxy-2-propanone.
- the composition may comprise a safe and effective amount of one or more anti-acne actives.
- useful anti-acne actives include resorcinol, sulfur, salicylic acid, erythromycin, zinc, and benzoyl peroxide. Suitable anti-acne actives are described in further detail in U. S. Patent No. 5,607,980.
- the composition may comprise a safe and effective amount of a desquamation active such as from about 0.01 % to about 10%, from about 0.5% to about 5%, or, alternatively, from about 0.1 % to about 2%, by weight of the composition.
- the desquamation actives tend to improve the texture of the skin (e.g., smoothness).
- a suitable desquamation system may comprise sulfhydryl compounds and zwitterionic surfactants and is described in U.S. Pat. No. 5,681,852.
- Another suitable desquamation system may comprise salicylic acid and zwitterionic surfactants and is described in U.S. Pat. No. 5,652,228.
- composition may comprise a safe and effective amount of an anti-cellulite agent.
- Suitable agents may include, but are not limited to, xanthine compounds (e.g., caffeine, theophylline, theobromine, and aminophylline).
- Skin care compositions may comprise a safe and effective amount of a chelating agent such as from about 0.1 % to about 10% or from about 1% to about 5% of the composition.
- a chelating agent such as from about 0.1 % to about 10% or from about 1% to about 5% of the composition.
- Exemplary chelators are disclosed in U.S. Patent No. 5,487,884.
- a suitable chelator is furildioxime and derivatives.
- the composition may comprise a skin lightening agent.
- the compositions may comprise from about 0.1 % to about 10%, from about 0.2% to about 5%, or, alternatively, from about 0.5% to about 2%, by weight of the composition, of a skin lightening agent.
- Suitable skin lightening agents include kojic acid, arbutin, tranexamic acid, ascorbic acid and derivatives (e.g., magnesium ascorbyl phosphate or sodium ascorbyl phosphate or other salts of ascorbyl phosphate), ascorbyl glucoside, and the like.
- composition compositions may comprise a flavonoid
- the flavonoid can be synthetic materials or obtained as extracts from natural sources, which also further may be de ⁇ vatized Examples of classes of suitable flavonoids are disclosed in U S Patent 6,235,773
- compositions may comprise protease inhibitors including, but are not limited to, hexamidine compounds, vanillin acetate, menthyl anthranilate, soybean trypsin inhibitor, Bowman-Birk inhibitor, and mixtures thereof
- protease inhibitors including, but are not limited to, hexamidine compounds, vanillin acetate, menthyl anthranilate, soybean trypsin inhibitor, Bowman-Birk inhibitor, and mixtures thereof
- Skin care compositions can include hexamidine compounds, its salts, and derivatives
- hexaminide compound means a compound having the formula
- R 1 and R 2 are optional or are organic acids (e g , sulfonic acids, etc )
- a particularly suitable hexamidine compound is hexamidine diisethionate
- composition may other optional components such as non-vitamin antioxidants and radical scavengers, hair growth regulators, anti-wrinkle actives, anti-atrophy actives, minerals, phytosterols and/or plant hormones, tyrosinase inhibitors, anti-inflammatory agents, N-acyl amino acid compounds, antimicrobial or antifungal actives, and other useful skin care actives, which are described in further detail in U S application publication No US 2006/0275237 Al and US 2004/ 0175347 Al N Color Cosmetics
- the silicones of the present invention may also be used in cosmetic compositions, i e , in products suitable for use in, on, or around the eyes, eyebrows, face, neck, chest, lips, hands, feet, or nails
- Exemplary cosmetic products include eye liners, eye shadows, eyebrow pencils, mascaras, eye makeup removers, false eyelashes, under-eye concealers, eye creams, concealers, correctors, primers, blushes, bronzers, highlighters, shimmers, foundations, powders, sunscreens, brushes, face creams, lip primers, lip pencils, lipsticks, lip glosses, lip balms, lip stains, lip creams, and lotions
- Examples of cosmetic products are found in U S Pat No 6,325,995 directed to an exemplary lip product, and U S Pat No 6,696,049 directed to an exemplary face product, and U S Pat No 6,503,495
- the silicones of the present invention may be combined with materials commonly found in these compositions, such as alkyl dimethicone copoly
- compositions of the present invention may contain also vitamins and amino acids such as water soluble vitamins such as vitamin B l , B2, B6, B 12, C, pantothenic acid, pantothenyl ethyl ether, panthenol, biotin, and their derivatives, water soluble amino acids such as asparagine, alanin, indole, glutamic acid and their salts, water insoluble vitamins such as vitamin A, D, E, and their salts and/or derivatives, water insoluble amino acids such as tyrosine, tryptamine, viscosity modifiers, dyes, nonvolatile solvents or diluents (water soluble and insoluble), pearlescent aids, foam boosters, additional surfactants or nonionic co-surfactants, pediculocides, pH adjusting agents, perfumes, preservatives, chelants, proteins, skin active agents, sunscreens, UV absorbers, vitamins, niacinamide, caffeine and minoxidil
- compositions of the present invention may also contain pigment matenals such as inorganic, nitroso, monoazo, disazo, carotenoid, t ⁇ phenyl methane, t ⁇ aryl methane, xanthene, quinohne, oxazine, azine, anthraquinone, indigoid, thionindigoid, quinac ⁇ done, phthalocianine, botanical, natural colors, including water soluble components such as those having C I Names
- the compositions of the present invention may also contain antimicrobial agents which are useful as cosmetic biocides
- compositions of the present invention may also contain chelating agents
- compositions of the present invention may include oxidative dye compounds in the form of primary intermediates (developers) or couplers
- the compounds suitable for use in the inventive compositions (including those optionally added), in so far as they are bases, may be used as free bases or in the form of their physiologically compatible salts with organic or inorganic acids, such as hydrochloric, hydrobromic, citnc, acetic, lactic, succinic, tartaric, or sulfunc acids, or, in so far as they have aromatic hydroxyl groups, in the form of their salts with bases, such as alkali phenolates
- Suitable developers for use in the compositions described herein include, but are not limited to, p- phenylenediamine derivatives, e g benzene- 1 ,4-diamine (commonly known as p- phenylenediamine), 2-chloro-benzene- l ,4-diamine, N-phenyl-benzene-l ,4-diamine, N-(2- ethoxyethyl)benzene-l ,4-diamine, 2-[(4-amino-phenyl)-(2-hydroxy-ethyl)-amino]-ethanol
- N,N-bis(2-hydroxyethyl)-p-phenylenediamine (2,5-diamino-phenyl)- methanol, 2-(2,5-diamino-phenyl)-ethanol, N-(4-aminophenyl)benzene- l ,4-diamine, 2,6- dimethyl-benzene-l ,4-diamine, 2-isopropyl-benzene- l ,4-diamine, l-[(4-aminophenyl)amino]- propan-2-ol, 2-propyl-benzene- 1 ,4-diamine, 1 ,3-bis[(4-aminophenyl)(2- hydroxyethyl)amino]propan-2-ol, 1 ,4-diamine, 2-methoxy-benzene- 1 ,4-diamine, l-(2,5-diaminophenyl)
- Additional developers are selected from the group consisting of N-O-furylmethyObenzene- M- diamine, N-thiophen-3-ylmethyl-benzene- l ,4-diamine, ⁇ r -(2-furylmethyl)benzene-l ,4-diamine, N-thiophen-2-ylmethyl-benzene- 1,4-diamine, 3-(2,5-diamino-phenyl)-N-ethyl-acrylamide, 2-[3- (3-a ⁇ uno-phenylamino)-propenyl]-benzene- 1 ,4-diamine, 2-[3-(4-amino-phenyla ⁇ uno)-propenyl]- benzene- 1 ,4-diamine , 2-(6-methyl-py ⁇ din-2-y l)-benzene- 1 ,4-diamine , 2-py ⁇ din-2-yl-benzene- 1 ,
- developers include but are not limited to: p-phenylenediamine derivatives such as: 2-methyl-benzene-l,4-diamine; benzene- 1 ,4-diamine; l-(2,5-diamino- phenyl)-ethanol; 2-(methoxymethyl)benzene- 1,4-diamine; N-(2-methoxyethyl)benzene- 1,4- diamine; l-(2,5-diaminophenyl)ethane-l ,2-diol; l,3-bis(N-(2-hydroxyethyl)-N-(4-amino- phenyl)amino)-2-propanol; 2,2'-[ 1 ,2-ethanediyl-bis-(oxy-2, 1 -ethanediyloxy)]-bis-benzene- 1 ,4- diamine; N,N-bis(2-hydroxyethyl)-p
- developers include 2-methyl-benzene-l ,4-diamine, 2- (methoxymethyl)benzene- 1 ,4-diamine, benzene- 1 ,4-diamine, N,N-bis(2-hydroxyethyl)-p- phenylenediamine, 4-amino-phenol, 4-methylarruno-phenol, 4-arruno-3-methyl-phenol, 2-amino- phenol, 2-arruno-5-methyl-phenol, 2-arruno-5-ethyl-phenol, 2-arruno-6-methyl-phenol, 1 -methyl - lH-pyrazole-4,5-diamine, 2-(4,5-diamino- lH-pyrazol-l-yl)ethanol, 2,5-diaminotoluene, 2,5- diaminophenylethyl alcohol, salts thereof, and mixtures thereof
- Suitable couplers for use in the compositions described herein include, but are not limited to phenols, resorcinols, naphthols, m-aminophenols, m-phenylenediamines, and heterocyclic compounds, and de ⁇ vatives thereof such as 2-amino-5-ethyl-phenol, naphthalene- 1 ,7-diol, benzene- 1 ,3-diol, 4-chlorobenzene-l,3-diol, naphthalen- 1 -ol, 2-methyl-na ⁇ hthalen- l-ol, naphthalene- 1 ,5-diol, naphthalene-2,7-diol, benzene- 1,4-diol, 2-methyl-benzene-l,3-diol, 7- amino-4-hydroxy-naphthalene-2-sulfonic acid, l,2,3,4-te
- couplers include but are not limited to phenol, resorcinol, and naphthol de ⁇ vatives such as 2-ammo-5-ethyl-phenol, naphthalene- 1,7-diol, benzene- 1,3-diol, 4- chlorobenzene-l ,3-diol, naphthalen-1-ol, 2-methyl-naphthalen-l-ol, naphthalene- 1,5-diol, naphthalene-2,7-diol, benzene- 1,4-diol, 2-methyl-benzene- l ,3-diol, and 2-isopropyl-5- methylphenol, 1,2,4-t ⁇ hydroxybenzene, l-acetoxy-2-methylnaphthalene, and mixtures thereof, m-phenylenediamine derivatives such as benzene- 1,3-diamine, 2-(2,4-diamin
- couplers include 2-amino-5-ethyl-phenol, benzene- 1,3-diol, 4- chlorobenzene-l,3-diol, 4,6-dichlorobenzene- l,3-diol, 2-methyl-benzene- l ,3-diol, 2-amino-4-(2'- hydroxyethyl)aminoanisole, 2,4-diaminobenzyl alcohol, 2,4-diaminophenylethyl alcohol, m- phenylenediamine, 5-amino-2-methyl-phenol, 3-amino-2,6-dimethylphenol, 2,4- diaminophenoxyethanol, 1-naphthol, 2-methyl-naphthol, 3-aminophenol, 3-amino-2- methylphenol, 4-hydroxy- l,2-methylenedioxybenzene, 4-a ⁇ uno-l ,2-methylenedioxybenzene, 4-
- developers and couplers include 5 -methoxy methyl - 2-arrunophenol, 5-ethyl-2-arruno ⁇ henol, 5-phenyl-2-aminophenol, 5-cyanoethyl-2-aminophenol, salts thereof, and mixtures thereof Any of the developers and couplers described above may be combined to form a mixture of developers and couplers.
- the hair dye compositions of the present invention will generally comprise from about 0.001 % to about 10% by weight of the dyeing composition of developer and coupler dyes.
- compositions providing low intensity dyeing such as natural blond to light brown hair shades generally comprise from about 0.001 % to about 5%, in some embodiments, from about 0.1 % to about 2%, in certain embodiments, from about 0.2% to about 1 % by weight of dyeing composition of developers and couplers.
- Darker shades such as browns and black typically comprise from 0.001 % to about 10% by weight, in some embodiments, from about 0.05% to about 7% by weight, in certain embodiments, from about 1 % to about 5% of developers and couplers.
- Developer compounds are generally used in approximately equimolar quantities with respect to coupler compounds. The developer compound may, however, be present in a greater or lesser quantity with respect to the coupler compound.
- the inventive compositions may also comprise compatible direct dyes, in an amount sufficient to provide coloring, particularly with regard to intensity. Typically, such an amount will range from about 0.05% to about 4%, by weight of the dye composition.
- Suitable direct dyes include but are not limited to: Acid Yellow 1 ; Acid Orange 3; Disperse Red 17; Basic Brown 17; Acid Black 52; Acid Black 1 ; Disperse Violet 4; 4-nitro-o-phenyIenediamine; 2-nitro-p- phenylenediamine; Picramic Acid; HC Red No. 13; l ,4-bis-(2'-hydroxyethyI)-amino-2- nitrobenzene; HC Yellow No. 5; HC Red No. 7; HC Blue No. 2; HC Yellow No.
- Preferred direct dyes include but are not limited to: Disperse Black 9; HC Yellow 2; HC Yellow 4; HC Yellow 15; 4-nitro-o-phenylenediamine; 2- amino-6-chloro-4-nitrophenol; HC Red 3; Disperse Violet 1 ; HC Blue 2; Disperse Blue 3; Disperse Blue 377; Basic Red 51 ; Basic Orange 31 ; Basic Yellow 87; and mixtures thereof.
- the inventive compositions may comprise an oxidizing agent, present in an amount sufficient to bleach melanin pigment in hair and/or cause formation of dye chromophores from oxidative dye precursors (including developers and/or couplers when present).
- Inorganic peroxygen materials capable of yielding hydrogen peroxide in an aqueous medium are preferred and include but are not limited to: hydrogen peroxide; inorganic alkali metal peroxides (e.g. sodium periodate and sodium peroxide); organic peroxides (e.g. urea peroxide, melamine peroxide); inorganic perhydrate salt bleaching compounds (e.g.
- the oxidizaing agents of the present invention are selected from percarbonates (such as sodium percarbonate, ammonium percarbonate and potassium percarbonate); and persulphates (such as sodium persulphate, ammonium persulphate, and potassium persulphate).
- the oxidizaing agents of the present invention are selected from sodium percarbonate and ammonium persulfate.
- inventive compositions may comprise a pH modifier and/or buffering agent in an amount that is sufficiently effective to adjust the pH of the composition to fall within a range from about 3 to about 13, in some embodiments from about 8 to about 12, and even from about 8 to about 1 1.
- the pH range for the carbonate ion source as described herein below is from 8.5 to 9.5, preferably from 8 to 9.
- Suitable pH modifiers and/or buffering agents for use herein include, but are not limited to: ammonia, alkanolamines such as monoethanolamine, diethanolamine, triethanolamine, monopropanolamine, dipropanolamine, tripropanolamine, tripropanol amine, 2-amino-2-methyl-l-propanol, and 2-amino-2-hydroxymethyl-l ,3,-propandiol and guanidium salts, alkali metal and ammonium hydroxides and carbonates, preferably sodium hydroxide and ammonium carbonate, and acidulents such as inorganic and inorganic acids, e.g., phosphoric acid, acetic acid, ascorbic acid, citric acid or tartaric acid, hydrochloric acid, and mixtures thereof.
- alkanolamines such as monoethanolamine, diethanolamine, triethanolamine, monopropanolamine, dipropanolamine, tripropanolamine, tripropanol amine,
- compositions of the present invention may further comprise in an embodiment at least one source of peroxymonocarbonate ions, preferably formed in situ from a source of hydrogen peroxide and a carbonate ion source.
- the compositions thus also may comprise at least a source of carbonate ions or carbamate ions or hydrocarbonate ions or any mixture thereof. Any source of these ions may be utilized.
- Suitable sources for use herein include sodium, potassium, guanidine, arginine, lithium, calcium, magnesium, barium, ammonium salts of carbonate, carbamate and hydrocarbonate ions and mixtures thereof such as sodium carbonate, sodium hydrogen carbonate, potassium carbonate, potassium hydrogen carbonate, guanidine carbonate, guanidine hydrogen carbonate, lithium carbonate, calcium carbonate, magnesium carbonate, barium carbonate, ammonium carbonate, ammonium hydrogen carbonate and mixtures thereof.
- Percarbonate salts may also be utilized to provide both the source of carbonate ions and oxidizing agent
- Suitable sources of carbonate ions, carbamate and hydrocarbonate ions include sodium hydrogen carbonate, potassium hydrogen carbonate, ammonium carbamate and mixtures thereof
- the inventive compositions may comp ⁇ se a radical scavenger, in a sufficient amount to reduce damage to the hair du ⁇ ng an oxidative bleaching or colo ⁇ ng process
- the radical scavenger is preferably selected such that it is not an identical species as the alkalizing agent
- the radical scavenger is a species that can react with a carbonate radical to convert the carbonate radical by a se ⁇ es of fast reactions to a less reactive species
- Suitable radical scavengers may be selected from the classes of alkanolamines, amino sugars, amino acids and mixtures thereof, and may include, but are not limited to monoethanolamine, 3-arruno- l-propanol, 4-amino-l-butanol,5- amino-1-pentanol, l-amino-2-pro ⁇ anol, 1 -arruno-2-butanol, l-amino-2-pentanol, l-amino-3- pentanol
- the inventive composition may comp ⁇ se chelants in an amount sufficient to reduce the amount of metals available to interact with formulation components, particularly oxidizing agents, more particularly peroxides
- Suitable chelants for use herein include but are not limited to diamine - N,N'-dipolyacid, monoamine monoamide-N,N'-dipolyacid, and N,N'-bis(2- hydroxybenzyl)ethylenediamine-N,N'-diacetic acid chelants (preferably EDDS (ethylenediaminedisuccinic acid)), carboxyhc acids (preferably aminocarboxylic acids), phosphonic acids (preferably aminophosphonic acids) and polyphospho ⁇ c acids (in particular straight polyphospho ⁇ c acids), their salts and de ⁇ vatives Foaming agents
- the inventive composition may be delivered in the form of a foam
- a foaming agent such as surfactants (e g , anionic, non-ionic, cationic and amphoteric), proteins (e.g., enzymes), cellulosic materials, polymeric materials and mixtures thereof.
- Suitable polymeric materials include hydrophilic polymers, such as, agar-agar, polyvinyl alcohol, sodium alginate and sodium dodecyl sulphate-poly(ethylene oxide).
- a preferred polymeric material is a hydrophobically-modified alkali soluble emulsion polymer synthesized through an emulsion polymerization process from an acid/acrylate copolymer backbone and a monomer that connects the hydrophobic groups as side chains.
- An example of such a material is AculynTM 22, commercially available from Rohm Haas, which is synthesized from acrylic acid, acrylate esters and a steareth-20 methacrylate ester.
- Another preferred polymer is an anionic alkali-soluble polymer emulsion synthesized from acid and acrylate comonomers through emulsion polymerization.
- An example of such a material is AculynTM 33, commercially available from Rohm Haas.
- Other foaming agents include cetyl hydroxyethylcellulose, PEG 7M, hydroxypropyl methylcellulose, Carbomer and polyquaternium-55. Mixtures of these materials may be used.
- foam means a hair colorant composition which after being passed through a manually-actuable, non-aerosol dispenser has a foam specific volume from about 6 to about 14 ml/g, such as about 7.5 ml/g to about 12ml/g, or even from about 8 to about 10.5 ml/g.
- Acceptable foam characteristics in hair colorant composition are exemplified by foam that holds its shape and stays in a consistent form.
- the minimum time for this is at least long enough to transfer from a user's hand to the desired location on the hair, e.g. the foam substantially maintains its shape for at least 15 seconds, for example at least 20, or at least 30 seconds. It could be longer if a quantity of foam, e.g. a bowl full by a hair dresser, is generated and spreading on the head only starts once the bowl full is readily made.
- any movement of the user's hand causes the foam to run, drip or otherwise move from the user's hand before the foam reaches the desired location and is considered undesirable.
- the foam is suitable when formed when the composition is used with a manually-actable, non- aerosol dispenser where the composition is mixed with air such that the ratio of air to composition is from about 1 :6 to about 1 : 15, from about 1 :8 to about 1 : 12, or about 1 : 10.
- a suitable manually-actuable, non-aerosol dispenser structure include the dimensions of the dip tube, dimensions of the air ingress into the mixing chamber, mixing chamber dimensions, including the ingress and egress orifices from the mixing chamber, dispensing channel dimensions, porous elements (such as screens or meshes) and dispensing head orifice
- undecyl-based surfactant is blended with the other components of the shampoo compositions, according to standard methods known in the art
- the typical procedure used for a clarifying shampoo would be to combine the undecyl sulfate paste or undeceth sulfate paste or mixtures thereof with water, add the desired water soluble co-surfactant and finish the composition by the addition preservatives, pH control agents, perfume, and salts to obtain the target physical properties
- a water insoluble co-surfactant is desired the surfactant and water mixture can be heated to a suitable temperature to facilitate its incorporation
- a rheology modifier it can be added to the surfactant mixture p ⁇ or the finishing step
- the surfactant paste is combined with the co-surfactant as above and diluted with water to a target level commensurate to achieving the final activity
- Rheology modifiers can be added at this point followed by conditioning agents, e g sucrose polyesters, silicones or silicone emulsions or other oils, cationic polymers from polymer premixes, perfumes, pearlizing agents or opacifiers, perfumes, and preservatives Approp ⁇ ate mixing steps to insure homogeneity are used as needed
- the product is finished by the addition of pH control agents, hydrotropes, and salts to the desired physical properties
- the hair conditioners can be prepared by any conventional method well known in the art They are suitably made as follows deionized water is heated to 85° C and cationic surfactants and high melting point fatty compounds are mixed in If necessary, cationic surfactants and fatty alcohols can be pre-melted at 85° C before addition to the water The water is maintained at a temperature of about 85° C until the components are homogenized, and no solids are observed The mixture is then cooled to about 55° C and maintained at this temperature, to form a gel matrix Silicones, or a blend of silicones and a low viscosity fluid, or an aqueous dispersion of a silicone is added to the gel matrix When included, poly alpha-olefin oils, polypropylene glycols, and/or polysorbates are also added to the gel matrix When included, other additional components such as perfumes and preservatives are added with agitation The gel matrix is maintained at about 50° C du ⁇ ng this time with constant stirring to assure homogenization After it is homogenized,
- the present invention can also be used in a compact hair care formulation
- a compact formula is a formula which delivers the same benefit to the consumer at a lower usage level
- Compact formulations and methods of making compact formulations are described in US Application Publication No 2009/0221463A l
- adjuncts While not essential for the purposes of the present invention, the non-limiting list of adjuncts illustrated hereinafter are suitable for use in the instant compositions and may be desirably incorporated in certain embodiments of the invention, for example to assist or enhance performance, for treatment of the substrate to be cleaned, or to modify the aesthetics of the composition as is the case with perfumes, colorants, dyes or the like It is understood that such adjuncts are in addition to the components that are supplied via Applicants' agglomerate/particle The precise nature of these additional components, and levels of incorporation thereof, will depend on the physical form of the composition and the nature of the operation for which it is to be used Suitable adjunct mate ⁇ als include, but are not limited to, polymers, for example cationic polymers, surfactants, builders, chelating agents, dye transfer inhibiting agents, dispersants, enzymes, and enzyme stabilizers, catalytic mate ⁇ als, bleach activators, polyme ⁇ c dispersing agents, clay soil removal/anti-redeposition agents, b ⁇ ghteners,
- adjunct ingredients are not essential to Applicants' cleaning and fabric care compositions
- certain embodiments of Applicants' compositions do not contain one or more of the following adjuncts materials bleach activators, surfactants, builders, chelating agents, dye transfer inhibiting agents, dispersants, enzymes, and enzyme stabilizers, catalytic metal complexes, polymeric dispersing agents, clay and soil removal/anti-redeposition agents, b ⁇ ghteners, suds suppressors, dyes, additional perfumes and perfume delivery systems, structure elasticizing agents, fab ⁇ c softeners, carriers, hydrotropes, processing aids and/or pigments
- one or more adjuncts may be present as detailed below
- compositions according to the present invention can comprise a surfactant or surfactant system wherein the surfactant can be selected from nonionic and/or anionic and/or cationic surfactants and/or ampholytic and/or zwitte ⁇ onic and/or semi-polar nonionic surfactants
- the surfactant is typically present at a level of from about 0 1 %, from about 1%, or even from about 5% by weight of the cleaning compositions to about 99 9%, to about 80%, to about 35%, or even to about 30% by weight of the cleaning compositions
- compositions of the present invention can comprise one or more detergent builders or builder systems When present, the compositions will typically compose at least about 1 % builder, or from about 5% or 10% to about 80%, 50%, or even 30% by weight, of said builder Builders include, but are not limited to, the alkali metal, ammonium and alkanolammonium salts of polyphosphates, alkali metal silicates, alkaline earth and alkali metal carbonates, aluminosilicate builders polycarboxylate compounds ether hydroxypolycarboxylates, copolymers of maleic anhyd ⁇ de with ethylene or vinyl methyl ether, l ,3,5-t ⁇ hydroxybenzene-2,4,6-t ⁇ sulphonic acid, and carboxymethyl-oxysuccinic acid, the various alkali metal, ammonium and substituted ammonium salts of polyacetic acids such as ethylenediamine tetraacetic acid and nit ⁇ lot ⁇ acetic acid, as well
- compositions herein may also optionally contain one or more copper, iron and/or manganese chelating agents If utilized, chelating agents will generally comprise from about 0 1 % by weight of the compositions herein to about 15%, or even from about 3 0% to about 15% by weight of the compositions herein
- compositions of the present invention may also include one or more dye transfer inhibiting agents Suitable polyme ⁇ c dye transfer inhibiting agents include, but are not limited to, polyvinylpyrrolidone polymers, polyamine N-oxide polymers, copolymers of N-vinylpyrrolidone and N-vinylimidazole, polyvinyloxazohdones and polyvinyhmidazoles or mixtures thereof When present in the compositions herein, the dye transfer inhibiting agents are present at levels from about 0 0001 %, from about 0 01 %, from about 0 05% by weight of the cleaning compositions to about 10%, about 2%, or even about 1 % by weight of the cleaning compositions Dispersants - The compositions of the present invention can also contain dispersants Suitable water-soluble organic materials are the homo- or co-polyme ⁇ c acids or their salts, m which the polycarboxylic acid may comprise at least two carboxyl radicals separated from each other by not
- Enzymes - The compositions can compose one or more detergent enzymes which provide cleaning performance and/or fabric care benefits
- suitable enzymes include, but are not limited to, hemicellulases, peroxidases, proteases, cellulases, xylanases, lipases, phosphohpases, esterases, cutinases, pectinases, keratanases, reductases, oxidases, phenoloxidases, lipoxygenases, hgninases, pullulanases, tannases, pentosanases, malanases, ⁇ - glucanases, arabinosidases, hyaluronidase, chondroitinase, laccase, and amylases, or mixtures thereof
- a typical combination is a cocktail of conventional applicable enzymes like protease, lipase, cutinase and/or cellulase in conjunction with amylase
- Enzyme Stabilizers - Enzymes for use in compositions for example, detergents can be stabilized by va ⁇ ous techniques
- the enzymes employed herein can be stabilized by the presence of water-soluble sources of calcium and/or magnesium ions in the finished compositions that provide such ions to the enzymes
- Catalytic Metal Complexes - Applicants' compositions may include catalytic metal complexes
- One type of metal-containing bleach catalyst is a catalyst system comp ⁇ sing a transition metal cation of defined bleach catalytic activity, such as copper, iron, titanium, ruthenium, tungsten, molybdenum, or manganese cations, an auxiliary metal cation having little or no bleach catalytic activity, such as zinc or aluminum cations, and a sequestrate having defined stability constants for the catalytic and auxiliary metal cations, particularly ethylenediaminetetraacetic acid, ethylenediaminetetra (methyl-enephosphonic acid) and water- soluble salts thereof
- Such catalysts are disclosed in U S patent 4,430,243
- compositions herein can be catalyzed by means of a manganese compound
- a manganese compound Such compounds and levels of use are well known in the art and include, for example, the manganese-based catalysts disclosed in U S patent 5,576,282
- Cobalt bleach catalysts useful herein are known, and are described, for example, in U S patents 5,597,936 and 5,595,967
- Such cobalt catalysts are readily prepared by known procedures, such as taught for example in U S patents 5,597,936, and 5,595,967
- compositions herein may also suitably include a transition metal complex of a macropolycyclic ⁇ gid ligand - abbreviated as "MRL"
- MRL macropolycyclic ⁇ gid ligand - abbreviated as "MRL”
- the compositions and cleaning processes herein can be adjusted to provide on the order of at least one part per hundred million of the benefit agent MRL species in the aqueous washing medium, and may provide from about 0 005 ppm to about 25 ppm, from about 0 05 ppm to about 10 ppm, or even from about 0 1 ppm to about 5 ppm, of the MRL in the wash liquor
- Suitable transition-metals in the instant transition-metal bleach catalyst include manganese, iron and chromium
- Suitable MRL' s herein are a special type of ultra - ⁇ gid hgand that is cross-b ⁇ dged such as 5, 12-diethyl-l ,5,8, 12-tetraazabicyclo[6 6 2]hexa-decane
- Suitable transition metal MRLs are readily prepared by known procedures, such as taught for example in U S patent 6,225,464
- compositions of the present invention can be formulated into any suitable form and prepared by any process chosen by the formulator, non-limiting examples of which are described in U S 5,879,584, U S 5,691 ,297, U S 5,574,005, U S 5,569,645, U S 5,565,422, U S 5,516,448, U S 5,489,392, U S 5,486,303 all of which are incorporated herein by reference
- a process for producing a composition comprising a) forming a microcapsule of selected permeability in accordance with a process disclosed in this specification, and b) combing said microcapsules with one or more adjunct ingredients, is disclosed
- said particles may be contained in a slurry that is combined with said adjunct
- said slurry may comprise one or more processing aids, selected from the group consisting of water, aggregate inhibiting materials such as divalent salts, particle suspending polymers such as xanthan gum, guar gum, caboxy methyl cellulose
- said particles may be contained in an agglomerate that is combined with said adjunct
- said agglomerate may comprise materials selected from the group consisting of silicas, citric acid, sodium carbonate, sodium sulfate, sodium chlo ⁇ de, and binders such as sodium silicates, modified celluloses, polyethylene glycols, polyacrylates, polyacrylic acids, zeolites and mixtures thereof
- undecyl-based surfactant is blended with the other components of the shampoo compositions, according to standard methods known in the art
- the typical procedure used for a clarifying shampoo would be to combine the undecyl sulfate paste or undeceth sulfate paste or mixtures thereof with water, add the desired water soluble co-surfactant and finish the composition by the addition preservatives, pH control agents, perfume, and salts to obtain the target physical properties
- a water insoluble co-surfactant is desired the surfactant and water mixture can be heated to a suitable temperature to facilitate its incorporation
- a rheology modifier it can be added to the surfactant mixture pnor the finishing step
- the surfactant paste is combined with the co-surfactant as above and diluted with water to a target level commensurate to achieving the final activity
- Rheology modifiers can be added at this point followed by conditioning agents, e g sucrose polyesters, silicones or silicone emulsions or other oils, cationic polymers from polymer premixes, perfumes, pearlizing agents or opacifiers, perfumes, and preservatives Appropriate mixing steps to insure homogeneity are used as needed
- the product is finished by the addition of pH control agents, hydrotropes, and salts to the desired physical properties
- the hair conditioners can be prepared by any conventional method well known in the art They are suitably made as follows deionized water is heated to 85° C and cationic surfactants and high melting point fatty compounds are mixed in If necessary, cationic surfactants and fatty alcohols can be pre-melted at 85° C before addition to the water The water is maintained at a temperature of about 85° C until the components are homogenized, and no solids are observed The mixture is then cooled to about 55° C and maintained at this temperature, to form a gel matrix Silicones, or a blend of silicones and a low viscosity fluid, or an aqueous dispersion of a silicone are added to the gel matrix When included, poly alpha-olefin oils, polypropylene glycols, and/or polysorbates are also added to the gel matrix When included, other additional components such as perfumes and preservatives are added with agitation The gel matrix is maintained at about 50° C during this time with constant stirring to assure homogenization After it is homogenized, it is
- a method of treating and/or cleaning a situs may comprise optionally washing and/or rinsing said situs, contacting said situs with any single or combination of compositions disclosed in the present specification, and optionally washing and/or rinsing said situs Typically at least a portion of the situs is contacted with an embodiment of Applicants' composition, in neat form or diluted in a liquor, for example, a wash liquor
- washing includes but is not limited to, scrubbing, and mechanical agitation
- Liquors that may comp ⁇ se the disclosed compositions may have a pH of from about 3 to about 1 1 5
- Such compositions are typically employed at concentrations of from about 500 ppm to about 15,000 ppm in solution
- the wash solvent is water
- the water temperature typically ranges from about 5 0 C to about 90 0 C and,
- Particle size is measured using an Accusizer 780A, made by Particle Sizing Systems, Santa Barbara CA The instrument is calibrated from 0 to 300 ⁇ using Duke particle size standards Samples for particle size evaluation are prepared by diluting about Ig of capsule slurry in about 5g of de-ionized water and further diluting about Ig of this solution in about 25g of water
- the Accusizer should be reading in excess of 9200 counts/second If the counts are less than 9200 additional sample should be added The accusizer will dilute the test sample until 9200 counts/second and initiate the evaluation After 2 minutes of testing the Accusizer will display the results, including volume- weighted median size
- Substrate Sample A sample of the substrate for evaluation (typically a fabric), is placed over the inner hoop of an embroidery hoop. The outer hoop is placed over the sample and the inner hoop, the outer hoop is tightened partially, the substrate is pulled tight in the hoop set by pulling the substrate edges, and the outer hoop is fully tightened to securely hold the substrate sample. Excess sample is cut from the edges of the hoop and discarded.
- the substrate is chosen to be cotton fabric, 250 thread count.
- Capsule Retention Bath The capsule retention test is done using a 2000 mL glass beaker as the bath in which the substrate sample is submerged.
- the beaker is filled to about 1500 mL for all testing.
- the substrate sample, suspended on an embroidery hoop is placed into the beaker, with the top of the sample just below the liquid level and the bottom of the sample just above a magnetic stir bar that is run to keep movement in the fluid.
- Sample Pre -Rinse A substrate sample is suspended in de-ionized water in the capsule retention bath for 10 minutes to rinse away any fiber fragments, dust, dirt, or water- soluble surface treatments that may exist on the substrate surface. After 10 minutes in the retention bath, the sample is removed and air-dried for 10 minutes.
- Microcapsule Retention Microcapsules are added to a fresh 1500 rnL of de-ionized for the microcapsule retention test. The total weight of microcapsules (dry basis) is about 50% of the substrate weight (0.65g capsules, 1.25g substrate). The substrate is re- suspended in the retention bath for another 10 minutes. The sample is removed, allowed to dry on the hoop for about 1 hour and is then removed and allowed to air-dry at least 4 hours total.
- Extraction Samples After the retention sample has been air-dried, 2-1" by 3.125" samples are cut from it, using a steel template and razor blade. This provides replicate test samples for the subsequent extraction testing.
- Example one drop of perfume oil weighs 14.0 mg.
- the concentration for this standard solution would be 14 mg/15 ml.
- Example 2 the concentration for this standard solution would be 7 mg/15 ml.
- the concentration for this standard solution would be 1.4 mg/15 ml. lllllSllfrransfer a small amount of the standard solution to an auto-sampler vial and cap for GC analysis.
- a first oil phase consisting of 5Og cedar oil, 0.65g TBAEMA, and 0.52g Beta-C, is mixed for about 1 hour before the addition of 26g CN997. The solution is allowed to mix until needed later in the batch.
- a second oil solution consisting of 20Og of cedar oil, 1.56g Vazo-52 and 0.52g Vazo-67 is added to a jacketed steel reactor.
- the reactor is held at 35 0 C, and the oil solution is mixed at 1000 rpm with a 2" 4-tip flat blade mixer.
- a nitrogen blanket is applied to the reactor at a rate of 300 cc/min.
- the solution is heated to 75 0 C in 45 minutes and held at 75 0 C for 35 minutes, before cooling to 55 0 C in 75 minutes.
- the first oil phase is added, and the combined oils are mixed for another 70 minutes at 55 0 C.
- a water phase containing 30g of Colloid 351 , 1.1 g 20% NaOH, 60Og water, and 1.56g Vazo-68WSP, is prepared and mixed until the Vazo is dissolved.
- the water-phase pH is measured and the solution mixed until needed for batch preparation.
- the water phase pH for this batch is 4.58.
- mixing is stopped and the water phase is added to the batch via funnel (to prevent premature mixing of the phases)
- Mixing is restarted at an approp ⁇ ate speed to produce an emulsion with the desired size characteristics In this particular case, mixing is done at 3000 rpm for 20 minutes and 2000 rpm for 40 minutes
- Example 2 to 25 in Table 1 are similarly prepared Batch preparation methods are similar to those described in Example 1 with the exceptions stated in Table 1 Water phase pH is adjusted up or down as necessary using either 20% NaOH or concentrated HCl For examples 6 and 7, milling is continued throughout the batch due to emulsion instability du ⁇ ng capsule wall formation
- Example 15 and 16 are identical capsules, but for Example 16 the capsule batch pH is adjusted to pH 4
- Example 19 and 20 are similar
- Example 20 the capsule retention study conditions are first adjusted to pH 4
- Example 25 is a melamine-formaldehyde wall capsule, shown for comparative purposes
- Capsule test data are shown in Table 1
- Table 1 The table includes capsule leakage data (free-oil, and 4-week hexane leakage), Zeta potential for each batch (a measure of capsule surface charge), and a measure of relative capsule retention
- capsule retention on cotton fiber is usefully at least 1 mg oil determined following the capsule retention procedure set forth herein
- the invention makes possible capsule retention values of at least 1 mg oil, or even at least 4 mg oil, or even at least 8 mg oil, or even at least 10 mg oil
- Selected microcapsules from the above examples are formulated into a leave-on-conditioner formulation as follows to 98 0 grams of leave-on-conditioner (with a typical formulation given below) is added an appropriate amount of microcapsule slurry of examples 1 through 24, to deliver an encapsulated oil usage level of 05wt%
- the microcapsules are added on top of the conditioner formulation, then the contents are mixed using a SpeedMixer by Hauschild DAC 400FVZ, at 1000 RPM for 1 minute
- a typical composition of a leave-on conditioner formulation is given in the following table
- a subset of the capsules from the above examples is formulated into a ⁇ nse-off Shampoo formulation as follows: to 90.0 grams of shampoo formulation (with a typical formulation given below) is added an appropriate amount of microcapsule slurry of examples 1 through 24, to deliver an encapsulated oil usage level of 0.5wt%. The microcapsules and water are added on top of the shampoo formulation, then the contents are mixed using a SpeedMixer by Hauschild
- CD 1.6 meq./gram; 10% active ; Supplier Rhodia
- Sodium Chloride USP food grade
- supplier Morton note that salt is an adjustable ingredient, higher or lower levels may be added to achieve target viscosity.
- Cetyl alcohol Konol TM series available from Shin Nihon Rika
- Methylchloroisothiazolinone/Methylisothiazolinone Kathon TM CG available from
- N-Hance 3269 (with MoI. W. of -500,000 and 0.8meq/g) available from Aqulaon/Hercules
- composition may comprise one or more other skin care actives, their salts and derivatives, as disclosed herein, in amounts also disclosed herein as would be deemed suitable by one of skill in the art
- a suitable container combine the ingredients of Phase A
- combine the ingredients of Phase B Heat each phase to 73°C-78°C while mixing each phase using a suitable mixer (e g , Anchor blade, propeller blade, or IKA T25) until each reaches a substantially constant desired temperature and is homogenous
- a suitable mixer e g , Anchor blade, propeller blade, or IKA T25
- Phase B Heat each phase to 73°C-78°C while mixing each phase using a suitable mixer (e g , Anchor blade, propeller blade, or IKA T25) until each reaches a substantially constant desired temperature and is homogenous
- Phase B e.g , Anchor blade, propeller blade, or IKA T25
- Phase A Continue mixing until batch is uniform Pour product into suitable containers at 73-78 0 C and store at room temperature Alternatively, continuing to stir the mixture as temperature decreases results in lower observed hardness values at 21 and 33 0 C
- EXAMPLE 29 Microcapsules in Single Unit Dose Personal Care Product
- surfactant/polymer liquid processing composition is prepared at the indicated weight percentages as described in Table 1 below
- a target weight of 300 grams of the above composition is prepared with the use of a conventional overhead stirrer (IKA® RW20DZM Stirrer available from IKA® Works, Inc , Wilmington, DE) and a hot plate (Corning Incorporated Life Sciences, Lowell, MA)
- a conventional overhead stirrer IKA® RW20DZM Stirrer available from IKA® Works, Inc , Wilmington, DE
- a hot plate Corning Incorporated Life Sciences, Lowell, MA
- the cationic polymer when present, is then slowly added with constant stirring until homogenous
- the polyvinyl alcohol is weighed into a suitable container and slowly added to the main mixture in small increments using a spatula while continuing to stir while avoiding the formation of visible lumps
- the mixing speed is adjusted to minimize foam formation
- the mixture is slowly heated to 8O 0 C after which surfactants are added
- the mixture is then heated to 85 0 C while continuing to stir and then allowed to cool to room temperature
- Additional distilled water is added to
- a porous dissolvable solid substrate (also referred to in the examples herein as “substrate”) is prepared from the above liquid processing mixture as described in Table 2 below Table 2
- 300 grams of the processing mixture is stored within a convection oven for greater than two hours at 7O 0 C to pre-heat the processing mixture
- the mixture is then transferred into a pre- heated 5 quart stainless steel bowl (by placing into 7O 0 C oven for greater than 15 minutes) of a KITCHENAID® Mixer Model K5SS (available from Hobart Corporation, Troy, OH) fitted with a fiat beater attachment and with a water bath attachment comp ⁇ sing tap water at 70-75 0 C
- the mixture is vigorously aerated at a maximum speed setting of 10 until a wet density of approximately 0 26 grams/cm 3 is achieved (time recorded in table)
- the density is measured by weighing a filling a cup with a known volume and evenly scraping off the top of the cup with a spatula
- the resulting aerated mixture is then spread with a spatula into square 160 mm x 160 mm aluminum molds with a depth of 6 5 mm with the excess wet foam being
- Each of the resulting 160 mm x 160 mm square substrates is cut into nine 43 mm x 43 mm squares (with rounded edges) using a cutting die and a Samco SB20 cutting machine (each square representing surface area of approximately 16 9 cm 2 )
- the resulting smaller substrates are then equilibrated overnight ( 14 hours) in a constant environment room kept at 7O 0 F and 50% relative humidity within large zip-lock bags that are left open to the room atmosphere
- the substrate is mounted on a stainless steel easel that rests at about a 60 degree angle and with notches holding the substrate from sliding downward and with a hole in plate so that the substrate can easily be removed from the mount by pushing from the easel It is important that the top surface of the substrate (the side that is exposed to the air in the drying oven and opposite the side that is in direct contact with the aluminum mold during the drying process) is facing away from the easel
- a small glass bottle with a pump spray is filled with the primary fragrance oil 1 a and then sprayed onto the surface of the substrate from a distance of 2 to 3 inches
- the substrate is then removed from the easel and returned to the weigh boat on the balance with the top side facing upwards
- the weight of perfume applied is recorded and in the instance that the target weight is not achieved, either another spray amount is applied or a Kim wipe to absorb excess perfume away from the substrate This iterative process is repeated until the target weight range is achieved
- the amount of fragrance I a applied is recorded in the below table
- the resulting substrate resting on the small weigh boat
- Examples VI to IX can be made as follows all ingredients except the fragrance, linalool, and dihydromyrcenol are combined in a suitable container and heated to about 85°C to form a homogenous liquid The solution is then cooled to about 62°C and then the fragrance, linalool, and dihydromyrcenol are added The mixture is then poured into an approp ⁇ ate container and allowed to solidify up cooling to ambient temperature
- Example X can be made as follows all the ingredients except the propellant are combined in an appropriate aerosol container The container is then sealed with an appropriate aerosol delivery valve Next air in the container is removed by applying a vacuum to the valve and then propellant is added to container through the valve Finally an appropriate actuator is connected to the valve to allow dispensing of the product EXAMPLE 31.
- Aminosilicone- 1 (AMD): having an amine content of 0.12-0.15m mol/g and a viscosity of 3,000-8,00OmPa-S, which is water insoluble
- Aminosilicone-2 (TAS): having an amine content of 0.04-0.06m mol/g and a viscosity of 10,000-16,00OmPa-S, which is water insoluble Method of Preparation
- compositions of "Ex I" through “Ex VI” are prepared as follows Cationic surfactants, high melting point fatty compounds are added to water with agitation, and heated to about 8O 0 C The mixture is cooled down to about 5O 0 C to form a gel matrix earner Separately, slurries of perfume microcapsules and silicones are mixed with agitation at room temperature to form a premix The premix is added to the gel matrix earner with agitation If included, other ingredients such as preservatives are added with agitation Then the compositions are cooled down to room temperature
- Cationic surfactants high melting point fatty compounds are added to water with agitation, and heated to about 8O 0 C The mixture is cooled down to about 5O 0 C to form a gel matnx earner Then, silicones are added with agitation Separately, slurries of perfume microcapsules, and if included, other ingredients such as preservatives are added with agitation Then the compositions are cooled down to room temperature
- Non-limiting examples of product formulations containing purified perfume microcapsules of the aforementioned examples are summarized in the following table.
- Non-limiting examples of product formulations containing purified perfume microcapsules of the aforementioned examples are summarized in the following table.
- Non-limiting examples of product formulations containing purified perfume microcapsules of the aforementioned examples are summarized in the following table.
- Core/wall ratio can range from 80/20 up to 90/10 and average particle diameter can range from 5 ⁇ m to 50 ⁇ m, and can be pu ⁇ fied via any of the aforementioned examples
- Exemplary microcapsules that are suitable for formulating are provided in examples 1 through 25
- Non-limiting examples of product formulations containing purified perfume microcapsules of the aforementioned examples are summa ⁇ zed in the following table
- MiCa-TiO 2 Prestige Silk Silver Star ex Eckart or BiOCl (Biron Silver CO - Merck ) or pre- crystalhzed EGDS (Tegopearl N 100 ex Degussa, expressed as pure EGDS) **Microcapsule added as 25-35% active slurry (aqueous solution).
- Core/wall ratio can range from 80/20 up to 90/10 and average particle diameter can range from 5 ⁇ m to 50 ⁇ m, and can be purified via any of the aforementioned examples.
- Exemplary microcapsules that are suitable for formulating are provided in examples 1 through 25.
- Microcapsule added as 25-35% active slurry (aqueous solution) Core/wall ratio can range from 80/20 up to 90/10 and average particle diameter can range from 5 ⁇ m to 50 ⁇ m, and can be purified via any of the aforementioned examples Exemplary microcapsules that are suitable for formulating are provided in examples 1 through 25
- Core/wall ratio can range from 80/20 up to 90/10 and average particle diameter can range from 5 ⁇ m to 50 ⁇ m, and can be pu ⁇ fied via any of the aforementioned examples
- Exemplary microcapsules that are suitable for formulating are provided in examples 1 through 25 ** Low water liquid detergent in Polyvinylalcohol unidose/sachet
- Core/wall ratio can range from 80/20 up to 90/10 and average particle diameter can range from 5 ⁇ m to 50 ⁇ m, and can be pu ⁇ fied via any of the aforementioned examples
- Exemplary microcapsules that are suitable for formulating are provided in examples 1 through 25
- EXAMPLES 37 Liquid Unit Dose
- the following are examples of unit dose executions wherein the liquid composition is enclosed within a PVA film
- Microcapsule added as 25-35% active slurry (aqueous solution) Core/wall ratio can range from 80/20 up to 90/10 and average particle diameter can range from 5 ⁇ m to 50 ⁇ m, and can be punfied via any of the aforementioned examples Exemplary microcapsules that are suitable for formulating are provided in examples 1 through 25
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Dispersion Chemistry (AREA)
- Wood Science & Technology (AREA)
- Engineering & Computer Science (AREA)
- Dermatology (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Cosmetics (AREA)
- Manufacturing Of Micro-Capsules (AREA)
- Detergent Compositions (AREA)
- Fats And Perfumes (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Polymerisation Methods In General (AREA)
- Disinfection, Sterilisation Or Deodorisation Of Air (AREA)
- Chemical Or Physical Treatment Of Fibers (AREA)
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MX2012012549A MX356208B (en) | 2010-04-28 | 2010-05-12 | Delivery particles. |
| CA2800456A CA2800456C (en) | 2010-04-28 | 2010-05-12 | Delivery particles |
| EP20179227.2A EP3733827A1 (en) | 2010-04-28 | 2010-05-12 | Delivery particles |
| MX2016014810A MX358448B (en) | 2010-04-28 | 2010-05-12 | Delivery particles. |
| JP2013506763A JP2013531079A (en) | 2010-04-28 | 2010-05-12 | Delivery particle |
| EP10721567A EP2563894A2 (en) | 2010-04-28 | 2010-05-12 | Delivery particles |
| RU2012144736/04A RU2566753C2 (en) | 2010-04-28 | 2010-05-12 | Particles for delivery |
| CN201080066442.6A CN102858940B (en) | 2010-04-28 | 2010-05-12 | Delivery particles |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US32896210P | 2010-04-28 | 2010-04-28 | |
| US61/328,962 | 2010-04-28 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| WO2010079467A2 true WO2010079467A2 (en) | 2010-07-15 |
| WO2010079467A3 WO2010079467A3 (en) | 2011-05-05 |
| WO2010079467A4 WO2010079467A4 (en) | 2011-06-30 |
Family
ID=42304396
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2010/052121 WO2010079467A2 (en) | 2010-04-28 | 2010-05-12 | Delivery particles |
Country Status (8)
| Country | Link |
|---|---|
| US (2) | US9993793B2 (en) |
| EP (3) | EP3733827A1 (en) |
| JP (4) | JP2013531079A (en) |
| CN (2) | CN103589530B (en) |
| CA (1) | CA2800456C (en) |
| MX (2) | MX358448B (en) |
| RU (1) | RU2566753C2 (en) |
| WO (1) | WO2010079467A2 (en) |
Cited By (47)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011163337A1 (en) | 2010-06-22 | 2011-12-29 | The Procter & Gamble Company | Perfume systems |
| WO2012003316A1 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Process for making films from nonwoven webs |
| WO2012003319A2 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Filaments comprising an active agent nonwoven webs and methods for making same |
| WO2012003360A2 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Detergent product and method for making same |
| WO2012003300A2 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Filaments comprising a non-perfume active agent nonwoven webs and methods for making same |
| WO2012003367A2 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Method for delivering an active agent |
| WO2012138696A2 (en) | 2011-04-07 | 2012-10-11 | The Procter & Gamble Company | Shampoo compositions with increased deposition of polyacrylate microcapsules |
| WO2012138710A2 (en) | 2011-04-07 | 2012-10-11 | The Procter & Gamble Company | Personal cleansing compositions with increased deposition of polyacrylate microcapsules |
| WO2012177357A1 (en) | 2011-06-23 | 2012-12-27 | The Procter & Gamble Company | Perfume systems |
| WO2013002786A1 (en) | 2011-06-29 | 2013-01-03 | Solae | Baked food compositions comprising soy whey proteins that have been isolated from processing streams |
| FR2985273A1 (en) | 2012-01-04 | 2013-07-05 | Procter & Gamble | FIBROUS STRUCTURES CONTAINING ASSETS AND HAVING MULTIPLE REGIONS |
| US8636918B2 (en) | 2011-08-05 | 2014-01-28 | Ecolab Usa Inc. | Cleaning composition containing a polysaccharide hybrid polymer composition and methods of controlling hard water scale |
| US8674021B2 (en) | 2006-07-21 | 2014-03-18 | Akzo Nobel N.V. | Sulfonated graft copolymers |
| US8679366B2 (en) | 2011-08-05 | 2014-03-25 | Ecolab Usa Inc. | Cleaning composition containing a polysaccharide graft polymer composition and methods of controlling hard water scale |
| JP2014510140A (en) * | 2011-04-07 | 2014-04-24 | ザ プロクター アンド ギャンブル カンパニー | Conditioner composition with increased adhesion of polyacrylate microcapsules |
| US8841246B2 (en) | 2011-08-05 | 2014-09-23 | Ecolab Usa Inc. | Cleaning composition containing a polysaccharide hybrid polymer composition and methods of improving drainage |
| US8853144B2 (en) | 2011-08-05 | 2014-10-07 | Ecolab Usa Inc. | Cleaning composition containing a polysaccharide graft polymer composition and methods of improving drainage |
| US8945314B2 (en) | 2012-07-30 | 2015-02-03 | Ecolab Usa Inc. | Biodegradable stability binding agent for a solid detergent |
| WO2015041791A1 (en) * | 2013-09-23 | 2015-03-26 | The Procter & Gamble Company | Particles |
| WO2014189906A3 (en) * | 2013-05-20 | 2015-04-09 | The Procter & Gamble Company | Encapsulates |
| US9051406B2 (en) | 2011-11-04 | 2015-06-09 | Akzo Nobel Chemicals International B.V. | Graft dendrite copolymers, and methods for producing the same |
| FR3014456A1 (en) | 2013-12-09 | 2015-06-12 | Procter & Gamble | |
| US9109068B2 (en) | 2005-07-21 | 2015-08-18 | Akzo Nobel N.V. | Hybrid copolymer compositions |
| WO2016090624A1 (en) * | 2014-12-12 | 2016-06-16 | The Procter & Gamble Company | Liquid cleaning composition |
| EP3087139A4 (en) * | 2013-12-23 | 2017-12-06 | Arlanxeo Singapore Pte. Ltd. | Ultra pure rubber |
| US9988526B2 (en) | 2011-11-04 | 2018-06-05 | Akzo Nobel Chemicals International B.V. | Hybrid dendrite copolymers, compositions thereof and methods for producing the same |
| WO2018140432A1 (en) | 2017-01-27 | 2018-08-02 | The Procter & Gamble Company | Active agent-containing articles that exhibit consumer acceptable article in-use properties |
| WO2018140472A1 (en) | 2017-01-27 | 2018-08-02 | The Procter & Gamble Company | Active agent-containing articles that exhibit consumer acceptable article in-use properties |
| WO2018140454A1 (en) | 2017-01-27 | 2018-08-02 | The Procter & Gamble Company | Active agent-containing articles and product-shipping assemblies for containing the same |
| WO2018140431A1 (en) | 2017-01-27 | 2018-08-02 | The Procter & Gamble Company | Active agent-containing articles that exhibit consumer acceptable article in-use properties |
| US10053652B2 (en) | 2014-05-15 | 2018-08-21 | Ecolab Usa Inc. | Bio-based pot and pan pre-soak |
| EP3369845A1 (en) | 2012-01-04 | 2018-09-05 | The Procter & Gamble Company | Active containing fibrous structures with multiple regions having differing densities |
| WO2018236700A1 (en) * | 2017-06-20 | 2018-12-27 | The Procter & Gamble Company | Multi composition systems comprising a bleaching agent and encapsulates |
| EP3087138A4 (en) * | 2013-12-23 | 2019-04-17 | Arlanxeo Singapore Pte. Ltd. | Rubbers with tunable levels of metal containing anti-agglomerants |
| US10266796B2 (en) | 2014-12-17 | 2019-04-23 | The Procter & Gamble Company | Detergent composition |
| WO2019141530A1 (en) * | 2018-01-17 | 2019-07-25 | Unilever Plc | Laundry detergent |
| US10662398B2 (en) | 2014-12-17 | 2020-05-26 | The Procter & Gamble Company | Detergent composition |
| EP3719192A1 (en) | 2012-01-04 | 2020-10-07 | The Procter & Gamble Company | Fibrous structures comprising particles and methods for making same |
| EP3736320A1 (en) * | 2019-05-08 | 2020-11-11 | The Procter & Gamble Company | Particles for through the wash laundry softening |
| EP3375855B1 (en) | 2017-03-16 | 2021-04-21 | The Procter & Gamble Company | Fabric softener composition comprising encapsulated benefit agent |
| WO2021113568A1 (en) | 2019-12-05 | 2021-06-10 | The Procter & Gamble Company | Method of making a cleaning composition |
| WO2021113567A1 (en) | 2019-12-05 | 2021-06-10 | The Procter & Gamble Company | Cleaning composition |
| US11065593B2 (en) | 2015-09-03 | 2021-07-20 | Tagra Biotechnologies Ltd. | Microcapsules encapsulating a reflective agent |
| US11085008B2 (en) | 2015-06-30 | 2021-08-10 | The Procter & Gamble Company | Methods for making compositions containing multiple populations of microcapsules |
| US11773351B2 (en) | 2015-06-30 | 2023-10-03 | The Procter & Gamble Company | Compositions containing multiple populations of microcapsules |
| US11820960B2 (en) | 2015-06-30 | 2023-11-21 | The Procter & Gamble Company | Compositions containing multiple populations of microcapsules |
| WO2024012811A1 (en) * | 2022-07-14 | 2024-01-18 | Givaudan Sa | Improvements in or relating to organic compounds |
Families Citing this family (115)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6460029B1 (en) | 1998-12-23 | 2002-10-01 | Microsoft Corporation | System for improving search text |
| US9993793B2 (en) | 2010-04-28 | 2018-06-12 | The Procter & Gamble Company | Delivery particles |
| US9186642B2 (en) | 2010-04-28 | 2015-11-17 | The Procter & Gamble Company | Delivery particle |
| US20110269657A1 (en) * | 2010-04-28 | 2011-11-03 | Jiten Odhavji Dihora | Delivery particles |
| US20120322709A1 (en) | 2011-05-26 | 2012-12-20 | Jianjun Justin Li | Compositions comprising an efficient perfume bloom |
| WO2013107586A2 (en) * | 2012-01-17 | 2013-07-25 | Unilever Plc | Benefit delivery particle, process for preparing said particle, compositions comprising said particles and a method for treating substrates |
| EP2895142B1 (en) | 2012-09-14 | 2017-04-19 | The Procter & Gamble Company | Aerosol antiperspirant compositions, products and methods |
| JP2015535207A (en) | 2012-11-09 | 2015-12-10 | ザ プロクター アンド ギャンブルカンパニー | Spraying equipment |
| WO2014082057A2 (en) | 2012-11-26 | 2014-05-30 | The Procter & Gamble Company | Packaged antiperspirant compositions |
| JP6189531B2 (en) | 2013-06-19 | 2017-08-30 | ザ プロクター アンド ギャンブル カンパニー | Absorbent article containing fragrance or odor control composition |
| CN105324135A (en) | 2013-06-19 | 2016-02-10 | 宝洁公司 | Absorbent article comprising complexed or encapsulated reactive compounds |
| CN105324102B (en) * | 2013-06-27 | 2019-01-29 | 宝洁公司 | Personal care product |
| US11186424B2 (en) | 2013-07-16 | 2021-11-30 | The Procter & Gamble Company | Antiperspirant spray devices and compositions |
| US20150023886A1 (en) | 2013-07-16 | 2015-01-22 | The Procter & Gamble Company | Antiperspirant Spray Devices and Compositions |
| US10787283B2 (en) | 2013-07-16 | 2020-09-29 | The Procter & Gamble Company | Antiperspirant spray devices and compositions |
| JP6490929B2 (en) * | 2013-09-12 | 2019-03-27 | ライオン株式会社 | Liquid detergent for textile products |
| US10092485B2 (en) * | 2013-10-04 | 2018-10-09 | Encapsys, Llc | Benefit agent delivery particle |
| US20150159001A1 (en) * | 2013-12-05 | 2015-06-11 | The Goodyear Tire & Rubber Company | Rubber composition and pneumatic tire |
| SG11201605113UA (en) | 2013-12-23 | 2016-07-28 | Arlanxeo Singapore Pte Ltd | Highly pure halogenated rubbers |
| US10081711B2 (en) | 2013-12-23 | 2018-09-25 | Arlanxeo Singapore Pte. Ltd. | Anti-agglomerants for the rubber industry |
| RU2708245C2 (en) | 2013-12-23 | 2019-12-05 | Арланксео Сингапур Пте. Лтд. | NOVEL ANTI-AGGLOMERATORS FOR ELASTOMERIC COPOLYMERS OF ETHYLENE AND α-OLEFIN |
| WO2015112487A1 (en) | 2014-01-21 | 2015-07-30 | The Procter & Gamble Company | Package for antiperspirant compositions |
| JP6462120B2 (en) | 2014-06-17 | 2019-01-30 | ザ プロクター アンド ギャンブル カンパニー | Composition for reducing hair curling |
| US10385200B2 (en) | 2014-06-30 | 2019-08-20 | Arlanxeo Singapore Pte. Ltd. | Anti-agglomerants for the rubber industry |
| WO2016003948A1 (en) * | 2014-06-30 | 2016-01-07 | The Procter & Gamble Company | Personal care compositions and methods |
| US9809783B2 (en) | 2014-08-27 | 2017-11-07 | The Procter & Gamble Company | Detergent composition comprising an acrylamide/maptac cationic polymer |
| US9714396B2 (en) | 2014-10-16 | 2017-07-25 | Encapsys Llc | Controlled release dual walled microcapsules |
| US9714397B2 (en) | 2014-10-16 | 2017-07-25 | Encapsys Llc | Controlled release microcapsules |
| US10485739B2 (en) | 2014-10-16 | 2019-11-26 | Encapsys Llc | High strength microcapsules |
| MX365260B (en) | 2014-11-10 | 2019-05-27 | Procter & Gamble | Personal care compositions. |
| CN104450252A (en) * | 2014-11-19 | 2015-03-25 | 苏州佑君环境科技有限公司 | Environment-friendly purification washing paste and preparation method thereof |
| JP6412269B2 (en) | 2014-12-05 | 2018-10-24 | ザ プロクター アンド ギャンブル カンパニー | Composition for reducing curly hair |
| US10117819B2 (en) | 2014-12-05 | 2018-11-06 | The Procter And Gamble Company | Composition for hair frizz reduction |
| WO2016093882A1 (en) | 2014-12-08 | 2016-06-16 | Kinnos Inc. | Additive compositions for pigmented disinfection and methods thereof |
| EP3034589A1 (en) * | 2014-12-17 | 2016-06-22 | The Procter and Gamble Company | Detergent composition |
| EP3034592A1 (en) * | 2014-12-17 | 2016-06-22 | The Procter and Gamble Company | Method of automatic dishwashing |
| WO2016096323A1 (en) * | 2014-12-19 | 2016-06-23 | Henkel Ag & Co. Kgaa | Pearly liquid detergent composition |
| CN105796375B (en) * | 2014-12-31 | 2018-07-13 | 罗允俊 | Portable clearing article |
| EP3061500B1 (en) * | 2015-02-25 | 2019-07-10 | Symrise AG | Stable dispersions |
| EP3265182A1 (en) * | 2015-03-03 | 2018-01-10 | The Procter and Gamble Company | Hair conditioning compositions with microcapsules |
| WO2016141171A1 (en) * | 2015-03-03 | 2016-09-09 | The Procter & Gamble Company | Hair conditioning compositions with microcapsules |
| DE102015205802A1 (en) * | 2015-03-31 | 2016-10-06 | Henkel Ag & Co. Kgaa | Detergent composition with bleach catalyst and perfume capsules |
| US10660835B2 (en) | 2015-04-02 | 2020-05-26 | The Procter And Gamble Company | Method for hair frizz reduction |
| US10632054B2 (en) | 2015-04-02 | 2020-04-28 | The Procter And Gamble Company | Method for hair frizz reduction |
| CN107530248B (en) | 2015-04-23 | 2021-03-12 | 宝洁公司 | Concentrated personal cleansing compositions and methods |
| CN107530245B (en) | 2015-04-23 | 2021-05-04 | 宝洁公司 | Concentrated personal cleansing compositions and uses |
| CN107530249B (en) | 2015-04-23 | 2021-09-07 | 宝洁公司 | Concentrated personal cleansing compositions and methods |
| MX369415B (en) | 2015-04-23 | 2019-11-07 | Procter & Gamble | Concentrated personal cleansing compositions. |
| US10245221B2 (en) | 2015-05-07 | 2019-04-02 | Celeb LLC | Stabilized color depositing shampoo |
| US9889080B2 (en) | 2015-05-07 | 2018-02-13 | Celeb LLC | Color depositing shampoo |
| EP3101099A1 (en) * | 2015-06-05 | 2016-12-07 | The Procter and Gamble Company | Compacted liquid laundry detergent composition |
| EP3101104B1 (en) * | 2015-06-05 | 2019-04-24 | The Procter and Gamble Company | Compacted liquid laundry detergent composition |
| CN106479710A (en) * | 2015-08-25 | 2017-03-08 | 石海旺 | A kind of multifuctional cleaning agent |
| EP3390483B1 (en) * | 2015-10-27 | 2022-05-18 | The Procter & Gamble Company | Encapsulation |
| WO2017091421A1 (en) | 2015-11-27 | 2017-06-01 | The Procter & Gamble Company | Multi-component fragrance dispensing apparatus |
| WO2017096154A1 (en) | 2015-12-04 | 2017-06-08 | The Procter & Gamble Company | Hair care regimen using compositions comprising moisture control materials |
| US10258555B2 (en) | 2015-12-04 | 2019-04-16 | The Procter And Gamble Company | Composition for hair frizz reduction |
| JP6938514B2 (en) * | 2016-01-14 | 2021-09-22 | アイエスピー インヴェストメンツ エルエルシー | Microcapsules whose shell is fragile and how to adjust and use them |
| US10344251B2 (en) | 2016-02-12 | 2019-07-09 | Kinnos, Inc. | Compositions and methods for use in surface decontamination |
| MY191406A (en) * | 2016-02-16 | 2022-06-24 | Lion Corp | Detergent composition |
| CN105670809A (en) * | 2016-02-24 | 2016-06-15 | 奉化市瑶琴生物科技有限公司 | Portable household cleaning agent and preparing and use methods thereof |
| US10538720B2 (en) | 2016-03-08 | 2020-01-21 | The Procter & Gamble Company | Particles including enzyme |
| CN105754740A (en) * | 2016-03-23 | 2016-07-13 | 奉化市瑶琴生物科技有限公司 | Convenient biological household detergent with mite killing and sterilization functions as well as preparation and application methods of household detergent |
| WO2017173050A1 (en) | 2016-04-01 | 2017-10-05 | The Procter & Gamble Company | Composition for fast dry of hair |
| US11491089B2 (en) | 2016-05-03 | 2022-11-08 | International Flavors & Fragrances Inc. | Reloadable microcapsules |
| WO2018026625A1 (en) * | 2016-08-01 | 2018-02-08 | The Procter & Gamble Company | Sprayable freshening product comprising suspended particles and methods of freshening the air or a surface with the same |
| US11097031B2 (en) | 2016-08-01 | 2021-08-24 | The Procter & Gamble Company | Phase-stable, sprayable freshening compositions comprising suspended particles |
| US10836985B2 (en) | 2016-08-09 | 2020-11-17 | Takasago International Corporation | Solid composition comprising free and encapsulated fragrances |
| EP3528777B1 (en) | 2016-10-21 | 2021-09-01 | The Procter & Gamble Company | Skin cleansing compositions and methods |
| US11185486B2 (en) | 2016-10-21 | 2021-11-30 | The Procter And Gamble Company | Personal cleansing compositions and methods |
| PL3535370T3 (en) * | 2016-11-01 | 2020-12-28 | The Procter & Gamble Company | Methods of using leuco colorants as bluing agents in laundry care compositions |
| US10870816B2 (en) | 2016-11-18 | 2020-12-22 | The Procter & Gamble Company | Fabric treatment compositions having low calculated cationic charge density polymers and fabric softening actives and methods for providing a benefit |
| US10675231B2 (en) * | 2017-02-17 | 2020-06-09 | The Procter & Gamble Company | Packaged personal cleansing product |
| PL3596192T3 (en) * | 2017-03-16 | 2024-07-15 | The Procter & Gamble Company | Benefit agent containing delivery particle slurries |
| EP3595779A1 (en) | 2017-03-16 | 2020-01-22 | The Procter and Gamble Company | Consumer product compositions comprising microcapsules |
| EP3375856B1 (en) | 2017-03-16 | 2021-09-01 | The Procter & Gamble Company | Fabric softener composition comprising encapsulated benefit agent |
| US10385297B2 (en) | 2017-03-16 | 2019-08-20 | The Procter & Gamble Company | Methods for making encapsulate-containing product compositions |
| JP2022009062A (en) * | 2017-03-16 | 2022-01-14 | ザ プロクター アンド ギャンブル カンパニー | Beneficial agent-containing delivery particle |
| US10385296B2 (en) | 2017-03-16 | 2019-08-20 | The Procter & Gamble Company | Methods for making encapsulate-containing product compositions |
| WO2018169532A1 (en) * | 2017-03-16 | 2018-09-20 | The Procter & Gamble Company | Benefit agent containing delivery particle |
| US10611988B2 (en) | 2017-03-16 | 2020-04-07 | The Procter & Gamble Company | Methods for making encapsulate-containing product compositions |
| CN110418669A (en) | 2017-03-16 | 2019-11-05 | 宝洁公司 | Consumer product compositions comprising microcapsules |
| US10980723B2 (en) | 2017-04-10 | 2021-04-20 | The Procter And Gamble Company | Non-aqueous composition for hair frizz reduction |
| JP2018176047A (en) * | 2017-04-10 | 2018-11-15 | パナソニックIpマネジメント株式会社 | Microcapsule and method for producing microcapsule, and cosmetics and method for producing cosmetics |
| EP3403640A1 (en) * | 2017-05-18 | 2018-11-21 | The Procter & Gamble Company | Conditioner compositions with increased deposition of polyacrylate microcapsules |
| JP6873556B2 (en) * | 2017-05-26 | 2021-05-19 | 花王株式会社 | Goods for hard surface modification |
| CN114788791A (en) | 2017-06-23 | 2022-07-26 | 宝洁公司 | Compositions and methods for improving the appearance of skin |
| JP7203044B2 (en) * | 2017-12-06 | 2023-01-12 | 花王株式会社 | fabric treatment composition |
| MX2020005977A (en) | 2017-12-06 | 2020-08-24 | Kao Corp | Soil release agent. |
| CN111448220B (en) | 2017-12-06 | 2023-03-31 | 花王株式会社 | Polysaccharide derivatives |
| AU2018381369B2 (en) | 2017-12-06 | 2023-11-30 | Kao Corporation | Composition |
| US11058616B2 (en) | 2017-12-18 | 2021-07-13 | The Procter & Gamble Company | Aerosol antiperspirant methods |
| JP2020515657A (en) | 2018-03-13 | 2020-05-28 | ザ プロクター アンド ギャンブル カンパニーThe Procter & Gamble Company | Consumer product composition containing microcapsules |
| WO2019177716A1 (en) | 2018-03-13 | 2019-09-19 | The Procter & Gamble Company | Consumer product compositions comprising microcapsules |
| WO2019177717A1 (en) | 2018-03-13 | 2019-09-19 | The Procter & Gamble Company | Consumer product compositions comprising microcapsules |
| CN108727995A (en) * | 2018-06-13 | 2018-11-02 | 合肥市旺友门窗有限公司 | A kind of door-window section bar high-performance group angle glue and preparation method thereof |
| CN112437657A (en) | 2018-07-03 | 2021-03-02 | 宝洁公司 | Method of treating skin conditions |
| EP3643290A1 (en) | 2018-10-24 | 2020-04-29 | The Procter & Gamble Company | Conditioner compositions with increased deposition of polyacrylate microcapsules |
| EP3643289A1 (en) | 2018-10-24 | 2020-04-29 | The Procter & Gamble Company | Conditioner compositions with increased deposition of polyacrylate microcapsules |
| EP3643292A1 (en) | 2018-10-24 | 2020-04-29 | The Procter & Gamble Company | Conditioner compositions with increased deposition of polyacrylate microcapsules |
| CN113382801B (en) * | 2019-01-25 | 2023-05-12 | 诺力昂化学品国际有限公司 | Spherical capsule based on glycol cellulose |
| BR112021018837A2 (en) * | 2019-03-26 | 2021-11-30 | Unilever Ip Holdings Bv | Liquid laundry composition |
| BR112021019949A2 (en) * | 2019-04-05 | 2021-12-07 | Unilever Ip Holdings Bv | Detergent composition for non-oxidative washing of fabric stains and method for non-oxidative washing of fabric stains |
| CN117050817A (en) | 2019-04-12 | 2023-11-14 | 埃科莱布美国股份有限公司 | Antimicrobial multipurpose cleaners and methods of making and using the same |
| MX2021013306A (en) * | 2019-05-06 | 2021-12-10 | Colgate Palmolive Co | Solid fabric care compositions and methods for the same. |
| WO2020249691A1 (en) | 2019-06-12 | 2020-12-17 | Nouryon Chemicals International B.V. | Process for the production of diacyl peroxides |
| ES2963356T3 (en) * | 2019-06-12 | 2024-03-26 | Nouryon Chemicals Int Bv | Method for isolating carboxylic acid from an aqueous side stream |
| US11976035B2 (en) | 2019-06-12 | 2024-05-07 | Nouryon Chemicals International B.V. | Process for the production of diacyl peroxides |
| CN111500380A (en) * | 2020-04-14 | 2020-08-07 | 广州市浪奇实业股份有限公司 | Composition capable of controlling release through ion concentration and preparation method thereof |
| US20230193160A1 (en) * | 2020-05-29 | 2023-06-22 | Isp Investments Llc | A suspending polymer composition, and method of use thereof |
| US10959933B1 (en) | 2020-06-01 | 2021-03-30 | The Procter & Gamble Company | Low pH skin care composition and methods of using the same |
| EP4157206A1 (en) | 2020-06-01 | 2023-04-05 | The Procter & Gamble Company | Method of improving penetration of a vitamin b3 compound into skin |
| CN117897136A (en) * | 2021-06-16 | 2024-04-16 | 埃斯普投资有限公司 | Suspension polymer compositions and methods of use thereof |
| CN113372486A (en) * | 2021-06-22 | 2021-09-10 | 西南石油大学 | Synthesis of temperature and pH dual stimulus response nano hydrogel and water-based drilling fluid |
| US20230015204A1 (en) | 2021-06-30 | 2023-01-19 | The Procter & Gamble Company | Spray devices for antiperspirant or deodorant compositions with a compressed gas propellant |
Family Cites Families (551)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US34584A (en) | 1862-03-04 | Improvement in rakes for harvesters | ||
| US424745A (en) | 1890-04-01 | Edwin d | ||
| US2062570A (en) | 1927-01-18 | 1936-12-01 | Ncr Co | Cash register |
| BE406221A (en) | 1933-11-15 | |||
| US2438091A (en) | 1943-09-06 | 1948-03-16 | American Cyanamid Co | Aspartic acid esters and their preparation |
| BE498391A (en) | 1944-10-16 | |||
| BE498392A (en) | 1945-11-09 | |||
| US2528378A (en) | 1947-09-20 | 1950-10-31 | John J Mccabe Jr | Metal salts of substituted quaternary hydroxy cycloimidinic acid metal alcoholates and process for preparation of same |
| US2658072A (en) | 1951-05-17 | 1953-11-03 | Monsanto Chemicals | Process of preparing amine sulfonates and products obtained thereof |
| US2800457A (en) | 1953-06-30 | 1957-07-23 | Ncr Co | Oil-containing microscopic capsules and method of making them |
| US2730456A (en) | 1953-06-30 | 1956-01-10 | Ncr Co | Manifold record material |
| BE530010A (en) | 1953-06-30 | |||
| US2730457A (en) | 1953-06-30 | 1956-01-10 | Ncr Co | Pressure responsive record materials |
| US2826551A (en) | 1954-01-04 | 1958-03-11 | Simoniz Co | Nontangling shampoo |
| US2809971A (en) | 1955-11-22 | 1957-10-15 | Olin Mathieson | Heavy-metal derivatives of 1-hydroxy-2-pyridinethiones and method of preparing same |
| US3429827A (en) | 1962-11-23 | 1969-02-25 | Moore Business Forms Inc | Method of encapsulation |
| US3236733A (en) | 1963-09-05 | 1966-02-22 | Vanderbilt Co R T | Method of combatting dandruff with pyridinethiones metal salts detergent compositions |
| US3332880A (en) | 1965-01-04 | 1967-07-25 | Procter & Gamble | Detergent composition |
| US3516941A (en) | 1966-07-25 | 1970-06-23 | Minnesota Mining & Mfg | Microcapsules and process of making |
| US3761418A (en) | 1967-09-27 | 1973-09-25 | Procter & Gamble | Detergent compositions containing particle deposition enhancing agents |
| JPS5212150B1 (en) | 1968-06-04 | 1977-04-05 | ||
| US3681248A (en) | 1969-06-13 | 1972-08-01 | Nat Patent Dev Corp | Water soluble entrapping of a fragrance |
| US3772215A (en) | 1969-06-13 | 1973-11-13 | Nat Patent Dev Corp | Water soluble entrapping of a fragrance |
| US3691140A (en) | 1970-03-09 | 1972-09-12 | Spencer Ferguson Silver | Acrylate copolymer microspheres |
| US4075134A (en) | 1970-06-08 | 1978-02-21 | The Dow Chemical Company | Emulsion polymerization method for preparing microspheres having liquid center and seamless rigid walls |
| US3965033A (en) | 1970-07-27 | 1976-06-22 | Fuji Photo Film Co., Ltd. | Process for the production of oil-containing microcapsules |
| US3886085A (en) | 1971-08-31 | 1975-05-27 | Fuji Photo Film Co Ltd | Process for producing fine oil-containing microcapsules having strong protective shells and microcapsules produced thereby |
| US3753196A (en) | 1971-10-05 | 1973-08-14 | Kulite Semiconductor Products | Transducers employing integral protective coatings and supports |
| US3826756A (en) | 1972-02-22 | 1974-07-30 | Loctite Corp | Process for preparing discrete particles of microencapsulated liquid anaerobic compositions |
| US4285720A (en) | 1972-03-15 | 1981-08-25 | Stauffer Chemical Company | Encapsulation process and capsules produced thereby |
| US3958581A (en) | 1972-05-17 | 1976-05-25 | L'oreal | Cosmetic composition containing a cationic polymer and divalent metal salt for strengthening the hair |
| US3898039A (en) | 1972-06-15 | 1975-08-05 | Tong Joe Lin | Article having fumigant containing substrate for diffusion promoting candle |
| CA1018893A (en) | 1972-12-11 | 1977-10-11 | Roger C. Birkofer | Mild thickened shampoo compositions with conditioning properties |
| FR2215864A5 (en) | 1973-01-25 | 1974-08-23 | Pepro | |
| US4062799A (en) | 1973-01-30 | 1977-12-13 | Fuji Photo Film Co., Ltd. | Method of forming microcapsule films having low porosity |
| GB1403139A (en) | 1973-09-18 | 1975-08-13 | Colgate Palmolive Co | Antiperspirant compositions |
| US3964500A (en) | 1973-12-26 | 1976-06-22 | Lever Brothers Company | Lusterizing shampoo containing a polysiloxane and a hair-bodying agent |
| JPS5843137B2 (en) | 1974-04-09 | 1983-09-24 | 富士写真フイルム株式会社 | capsule kahouhou |
| US4217914A (en) | 1974-05-16 | 1980-08-19 | L'oreal | Quaternized polymer for use as a cosmetic agent in cosmetic compositions for the hair and skin |
| US4422853A (en) | 1974-05-16 | 1983-12-27 | L'oreal | Hair dyeing compositions containing quaternized polymer |
| US4001140A (en) | 1974-07-10 | 1977-01-04 | Ncr Corporation | Capsule manufacture |
| DE2437090A1 (en) | 1974-08-01 | 1976-02-19 | Hoechst Ag | CLEANING SUPPLIES |
| US4046750A (en) | 1974-09-30 | 1977-09-06 | California Institute Of Technology | Ionene modified small polymeric beads |
| US4093556A (en) | 1975-07-02 | 1978-06-06 | Loctite Corporation | Process for microencapsulation of metallocenes |
| AT365448B (en) | 1975-07-04 | 1982-01-11 | Oreal | COSMETIC PREPARATION |
| US4197865A (en) | 1975-07-04 | 1980-04-15 | L'oreal | Treating hair with quaternized polymers |
| CH589464A5 (en) | 1975-07-11 | 1977-07-15 | Filtrox Maschinenbau Ag | |
| GB1507739A (en) | 1975-11-26 | 1978-04-19 | Wiggins Teape Ltd | Capsules |
| US4145184A (en) | 1975-11-28 | 1979-03-20 | The Procter & Gamble Company | Detergent composition containing encapsulated perfume |
| GB1546480A (en) | 1976-04-26 | 1979-05-23 | Fellows A N | Disinfectant and sterilizing preparations |
| US4100103A (en) | 1976-12-30 | 1978-07-11 | Ncr Corporation | Capsule manufacture |
| US4234627A (en) | 1977-02-04 | 1980-11-18 | The Procter & Gamble Company | Fabric conditioning compositions |
| LU76955A1 (en) | 1977-03-15 | 1978-10-18 | ||
| JPS545051A (en) | 1977-06-15 | 1979-01-16 | Norio Tanaka | Powdery perfume |
| JPS5425277A (en) | 1977-07-27 | 1979-02-26 | Fuji Photo Film Co Ltd | Method of producing microcapsule |
| US4166152B1 (en) | 1977-08-17 | 1999-05-18 | Minnesota Mining & Mfg | Tacky polymeric microspheres |
| FR2416008A1 (en) | 1978-02-02 | 1979-08-31 | Oreal | LIPOSOME LYOPHILISATES |
| JPS54107881A (en) | 1978-02-14 | 1979-08-24 | Fuji Photo Film Co Ltd | Preparation of minute capsule |
| US4197346A (en) | 1978-10-10 | 1980-04-08 | Appleton Papers Inc. | Self-contained pressure-sensitive record material and process of preparation |
| US4275055A (en) | 1979-06-22 | 1981-06-23 | Conair Corporation | Hair conditioner having a stabilized, pearlescent effect |
| US4507280A (en) | 1979-07-02 | 1985-03-26 | Clairol Incorporated | Hair conditioning composition and method for use |
| US4663158A (en) | 1979-07-02 | 1987-05-05 | Clairol Incorporated | Hair conditioning composition containing cationic polymer and amphoteric surfactant and method for use |
| JPS5651238A (en) | 1979-10-02 | 1981-05-08 | Fuji Photo Film Co Ltd | Production of microminiature capsule |
| US4379753A (en) | 1980-02-07 | 1983-04-12 | The Procter & Gamble Company | Hair care compositions |
| US4345080A (en) | 1980-02-07 | 1982-08-17 | The Procter & Gamble Company | Pyridinethione salts and hair care compositions |
| US4323683A (en) | 1980-02-07 | 1982-04-06 | The Procter & Gamble Company | Process for making pyridinethione salts |
| US4529586A (en) | 1980-07-11 | 1985-07-16 | Clairol Incorporated | Hair conditioning composition and process |
| US4423099A (en) | 1980-07-28 | 1983-12-27 | Ciba-Geigy Corporation | Membrane modified hydrogels |
| US4470982A (en) | 1980-12-22 | 1984-09-11 | The Procter & Gamble Company | Shampoo compositions |
| JPS5814942A (en) | 1981-07-17 | 1983-01-28 | Jujo Paper Co Ltd | Preparation of fine capsule |
| JPS5819261A (en) | 1981-07-27 | 1983-02-04 | 株式会社エンクラ−ビジネス | Aromatic composition |
| GR76237B (en) | 1981-08-08 | 1984-08-04 | Procter & Gamble | |
| US4446032A (en) | 1981-08-20 | 1984-05-01 | International Flavors & Fragrances Inc. | Liquid or solid fabric softener composition comprising microencapsulated fragrance suspension and process for preparing same |
| US4428869A (en) | 1981-08-20 | 1984-01-31 | International Flavors & Fragrances Inc. | Cologne consisting of microcapsule suspension |
| US4364837A (en) | 1981-09-08 | 1982-12-21 | Lever Brothers Company | Shampoo compositions comprising saccharides |
| US4444699A (en) | 1982-04-20 | 1984-04-24 | Appleton Papers Inc. | Capsule manufacture |
| US4561998A (en) | 1982-05-24 | 1985-12-31 | The Procter & Gamble Company | Near-neutral pH detergents containing anionic surfactant, cosurfactant and fatty acid |
| NL8203275A (en) | 1982-08-20 | 1984-03-16 | Naarden International Nv | METHOD FOR PREPARING AIR FRESHENING GELS, AND GELS GIVEN SO |
| US4550862A (en) | 1982-11-17 | 1985-11-05 | The Procter & Gamble Company | Liquid product pouring and measuring package with self draining feature |
| US4597898A (en) | 1982-12-23 | 1986-07-01 | The Proctor & Gamble Company | Detergent compositions containing ethoxylated amines having clay soil removal/anti-redeposition properties |
| JPS59139268A (en) | 1983-01-31 | 1984-08-10 | 株式会社 山村化学研究所 | Gradual release type aromatic composition |
| JPS59159175A (en) | 1983-03-02 | 1984-09-08 | Konishiroku Photo Ind Co Ltd | Pressure fixable microcapsule type toner |
| CH652035A5 (en) | 1983-03-23 | 1985-10-31 | Firmenich & Cie | Support element gelled vapor distribution devices agents sanitizers. |
| US4552811A (en) | 1983-07-26 | 1985-11-12 | Appleton Papers Inc. | Capsule manufacture |
| US4574110A (en) | 1983-07-28 | 1986-03-04 | Mitsui Toatsu Chemicals, Incorporated | Process for producing microcapsules and microcapsule slurry |
| AU561281B2 (en) | 1983-09-14 | 1987-05-07 | Three Bond Co. Ltd. | Acrylic photopolymerised micro-capsules |
| US4610927A (en) | 1983-09-14 | 1986-09-09 | Kureha Kagaku Kogyo Kabushiki Kaisha | Microcapsules containing a hydrophobic, volatile core substance and their production |
| DE3333654A1 (en) | 1983-09-17 | 1985-03-28 | Cassella Ag, 6000 Frankfurt | ENCLOSED, SLOWLY SOLUBLE IN WATER, THEIR PRODUCTION AND THEIR USE |
| US4515705A (en) | 1983-11-14 | 1985-05-07 | The Procter & Gamble Company | Compositions containing odor purified proteolytic enzymes and perfumes |
| DE3346601C2 (en) | 1983-12-23 | 1986-06-12 | Feldmühle AG, 4000 Düsseldorf | Microcapsules, process for their production and their use in pressure-sensitive carbonless copying papers |
| US4601863A (en) | 1984-02-09 | 1986-07-22 | Kanzaki Paper Manufacturing Co., Ltd. | Process for producing powder of microcapsules |
| NL8401362A (en) | 1984-04-27 | 1985-11-18 | Tno | METHOD OF COOPERATING PARTICULAR MATERIALS WITH A POLYMER IN ORDER TO ENABLE THE REGULATED DELIVERY OF THESE MATERIALS TO THE ENVIRONMENT AND THEREFORE OBTAINED COVERED MATERIAL. |
| US4537707A (en) | 1984-05-14 | 1985-08-27 | The Procter & Gamble Company | Liquid detergents containing boric acid and formate to stabilize enzymes |
| US4537706A (en) | 1984-05-14 | 1985-08-27 | The Procter & Gamble Company | Liquid detergents containing boric acid to stabilize enzymes |
| JPS6187734A (en) | 1984-10-03 | 1986-05-06 | Japan Synthetic Rubber Co Ltd | Production of hollow polymer particle |
| CA1261276A (en) | 1984-11-09 | 1989-09-26 | Mark B. Grote | Shampoo compositions |
| USRE34584E (en) | 1984-11-09 | 1994-04-12 | The Procter & Gamble Company | Shampoo compositions |
| JPS61244366A (en) | 1985-04-23 | 1986-10-30 | 小林 健郎 | Drug component gradual release powder |
| JPH0611396B2 (en) | 1985-07-17 | 1994-02-16 | 日本製紙株式会社 | Microcapsule manufacturing method |
| ES2001074A6 (en) | 1985-08-21 | 1988-04-16 | Clorox Co | Dry peracid based bleaching product. |
| US4863626A (en) | 1985-08-21 | 1989-09-05 | The Clorox Company | Encapsulated enzyme in dry bleach composition |
| JPS62116506A (en) | 1985-11-15 | 1987-05-28 | Sunstar Inc | Granular dentifrice composition having varying taste and flavor |
| US5145675A (en) | 1986-03-31 | 1992-09-08 | Advanced Polymer Systems, Inc. | Two step method for preparation of controlled release formulations |
| US4803947A (en) | 1986-01-15 | 1989-02-14 | Canon Kabushiki Kaisha | Apparatus for forming deposited film |
| FR2599975B1 (en) | 1986-06-16 | 1994-06-17 | Pointier Alain | PERFUME DIFFUSER, ODOR ABSORBENT, AND ASHTRAY EXTINGUISHER |
| JPS63112190A (en) | 1986-10-30 | 1988-05-17 | Fuji Photo Film Co Ltd | Recording material |
| US4754110A (en) | 1986-12-22 | 1988-06-28 | Lucerne Products, Inc. | Barrier sealing means for an electrical switch for resisting entry of foreign material into the switch body |
| US5370881A (en) | 1987-04-20 | 1994-12-06 | Fuisz Technologies Ltd. | Water-soluble delivery systems for hydrophobic liquids |
| GB8719091D0 (en) | 1987-08-12 | 1987-09-16 | Unilever Plc | Skin treatment composition |
| US5225278A (en) | 1987-08-26 | 1993-07-06 | Rohm And Haas Company | Process for microencapsulation |
| DE3856302T2 (en) | 1987-10-22 | 1999-09-09 | The Procter & Gamble Co. | Sunscreen containing chelating agents |
| US4911851A (en) | 1987-11-06 | 1990-03-27 | The Procter & Gamble Company | Detergent compatible, dryer released fabric softening/antistatic agents |
| GB9110408D0 (en) | 1989-08-24 | 1991-07-03 | Allied Colloids Ltd | Polymeric compositions |
| JP2634836B2 (en) | 1988-01-29 | 1997-07-30 | 大王製紙株式会社 | Manufacturing method of microcapsules |
| US4882220A (en) | 1988-02-02 | 1989-11-21 | Kanebo, Ltd. | Fibrous structures having a durable fragrance |
| CA1303437C (en) | 1988-02-29 | 1992-06-16 | Nobuo Kawahashi | Hollow polymer particles, process for production thereof, and use thereof as pigment |
| JPH01256965A (en) | 1988-04-07 | 1989-10-13 | Toray Ind Inc | Deodorizer and deodorant molding therefrom |
| GB8808359D0 (en) | 1988-04-09 | 1988-05-11 | Kitty Little Ltd | Deodorizing composition |
| US4919841A (en) | 1988-06-06 | 1990-04-24 | Lever Brothers Company | Wax encapsulated actives and emulsion process for their production |
| US4824707A (en) | 1988-06-27 | 1989-04-25 | Donald Spector | Decorative air freshener unit |
| JPH0236803A (en) | 1988-07-27 | 1990-02-06 | Tokyo Houraishiya:Kk | Clothes with deodorant or mark material containing deodorant and manufacture thereof |
| JPH0252661A (en) | 1988-08-12 | 1990-02-22 | Tokyo Houraishiya:Kk | Marking material having deodorant or deodorant exhalation function |
| US4968451A (en) | 1988-08-26 | 1990-11-06 | The Procter & Gamble Company | Soil release agents having allyl-derived sulfonated end caps |
| US5856409A (en) | 1989-04-07 | 1999-01-05 | Dow Corning Corporation | Method of making hydrophobic copolymers hydrophilic |
| JP2502146B2 (en) | 1989-04-12 | 1996-05-29 | 日本製紙株式会社 | Microcapsule manufacturing method |
| AU5741590A (en) | 1989-05-04 | 1990-11-29 | Southern Research Institute | Improved encapsulation process and products therefrom |
| US5188753A (en) | 1989-05-11 | 1993-02-23 | The Procter & Gamble Company | Detergent composition containing coated perfume particles |
| US5137646A (en) | 1989-05-11 | 1992-08-11 | The Procter & Gamble Company | Coated perfume particles in fabric softener or antistatic agents |
| WO1991001801A1 (en) | 1989-08-01 | 1991-02-21 | Kanebo, Ltd. | Microcapsule, treatment liquid containing microcapsules, and textile structure having microcapsules stuck thereto |
| US5104646A (en) | 1989-08-07 | 1992-04-14 | The Procter & Gamble Company | Vehicle systems for use in cosmetic compositions |
| US5106609A (en) | 1990-05-01 | 1992-04-21 | The Procter & Gamble Company | Vehicle systems for use in cosmetic compositions |
| US5071706A (en) | 1989-08-31 | 1991-12-10 | Eurand America, Incorporated | Oily, free-flowing, microcapsules |
| US5066419A (en) | 1990-02-20 | 1991-11-19 | The Procter & Gamble Company | Coated perfume particles |
| JP2798093B2 (en) | 1990-03-02 | 1998-09-17 | 日本ペイント株式会社 | Lubricating hydrophilic treatment agent and treatment method |
| US5105823A (en) | 1990-04-05 | 1992-04-21 | Blum Alvin S | Shielded replaceable lancet blade assembly |
| JPH0421513A (en) | 1990-05-11 | 1992-01-24 | Takeda Chem Ind Ltd | Silica gel bead coated with polymer composition |
| US5292835A (en) | 1990-05-16 | 1994-03-08 | Basf Aktiengesellschaft | Microcapsules |
| DK0462709T3 (en) | 1990-06-21 | 1996-02-05 | Revlon Consumer Prod Corp | Cosmetic preparations |
| US5234682A (en) | 1990-06-21 | 1993-08-10 | Revlon Consumer Products Corporation | Cosmetic compositions |
| JPH0482558A (en) | 1990-07-24 | 1992-03-16 | Kankyo Kagaku Kaihatsu Kk | Deodorizing and germ removing composition |
| GB9021061D0 (en) | 1990-09-27 | 1990-11-07 | Unilever Plc | Encapsulating method and products containing encapsulated material |
| US5113585A (en) | 1990-09-28 | 1992-05-19 | The Gillette Company | Shaving system |
| JP2555475B2 (en) | 1990-10-16 | 1996-11-20 | 工業技術院長 | Method for producing inorganic microspheres |
| JPH04156851A (en) | 1990-10-19 | 1992-05-29 | Toyo Ink Mfg Co Ltd | Deodorizing composition material |
| US5120349A (en) | 1990-12-07 | 1992-06-09 | Landec Labs, Inc. | Microcapsule having temperature-dependent permeability profile |
| US5176903A (en) | 1990-12-13 | 1993-01-05 | Revlon Consumer Products Corporation | Antiperspirant/deodorant containing microcapsules |
| DE4105672C1 (en) | 1991-02-22 | 1992-10-08 | Paul Ritzau Pari-Werk Gmbh, 8130 Starnberg, De | Oxygen distributor for inhalation therapy - has stirring chamber with agitator and apertures, with connector opening into chamber |
| ES2071418T3 (en) | 1991-04-24 | 1995-06-16 | Unilever Nv | PARTICLES ENCAPSULATED WITH WAX AND MANUFACTURING PROCEDURE THEREOF. |
| US5219887A (en) | 1991-06-07 | 1993-06-15 | Minnesota Mining And Manufacturing Company | Disinfecting shampoo composition for animals |
| JP2964181B2 (en) | 1991-07-11 | 1999-10-18 | 花王株式会社 | Bath composition |
| EP0523287A1 (en) | 1991-07-18 | 1993-01-20 | The Procter & Gamble Company | Perfume additives for fabric-softening compositions |
| US5232613A (en) | 1991-08-28 | 1993-08-03 | The Procter & Gamble Company | Process for preparing protected particles of water sensitive material |
| US5234611A (en) | 1991-08-28 | 1993-08-10 | The Procter & Gamble Company | Fabric softener, preferably liquid, with protected, dryer-activated, cyclodextrin/perfume complex |
| DE4130743A1 (en) | 1991-09-16 | 1993-03-18 | Bayer Ag | MICROCAPSULES MADE FROM ISOCYANATES WITH GROUPS CONTAINING POLYETHYLENE OXIDE |
| GB9120952D0 (en) | 1991-10-02 | 1991-11-13 | Unilever Plc | Perfume particles |
| WO1993008600A1 (en) | 1991-10-15 | 1993-04-29 | Velox Computer Technology, Inc. | Intrinsically controlled cooling container |
| WO1993013195A1 (en) | 1991-12-20 | 1993-07-08 | The Procter & Gamble Company | A process for preparing a perfume capsule composition |
| US5580578A (en) | 1992-01-27 | 1996-12-03 | Euro-Celtique, S.A. | Controlled release formulations coated with aqueous dispersions of acrylic polymers |
| JP3104394B2 (en) | 1992-05-13 | 2000-10-30 | 日本製紙株式会社 | Method for producing microcapsules |
| JP3401029B2 (en) | 1992-06-19 | 2003-04-28 | トッパン・フォームズ株式会社 | Improved microcapsules |
| US6325995B1 (en) | 1992-09-21 | 2001-12-04 | The Procter & Gamble Company | Lipsticks compositions containing association structures |
| JPH0641576U (en) | 1992-11-16 | 1994-06-03 | 株式会社ナカツカ | Rug |
| US6375872B1 (en) | 1992-11-17 | 2002-04-23 | Moore Business Forms | Microencapsulated adhesive |
| US5876755A (en) | 1993-02-22 | 1999-03-02 | Quest International Bv | Humidity resistant composition |
| CA2157178C (en) | 1993-03-01 | 2002-08-20 | Errol Hoffman Wahl | Concentrated biodegradable quaternary ammonium fabric softener compositions and compounds containing intermediate iodine value unsaturated fatty acid chains |
| FR2702961A1 (en) | 1993-03-26 | 1994-09-30 | Couvreur Continental | Device for slow, even and extended diffusion of a volatile substance such as an aromatic substance |
| DK0695169T3 (en) | 1993-04-22 | 2003-03-17 | Skyepharma Inc | Multivesicular liposomes with encapsulated cyclodextrin and pharmacologically active compounds and methods for using them |
| DE4321205B4 (en) | 1993-06-25 | 2006-06-29 | Basf Ag | Microcapsules, process for their preparation and their use |
| JPH0775666A (en) | 1993-07-14 | 1995-03-20 | Matsumoto Yushi Seiyaku Co Ltd | Gel aromatic |
| EP0711146B1 (en) | 1993-07-22 | 2000-09-06 | Pfizer Inc. | Osmotic device having a vapor-permeable coating |
| US5486303A (en) | 1993-08-27 | 1996-01-23 | The Procter & Gamble Company | Process for making high density detergent agglomerates using an anhydrous powder additive |
| US5366652A (en) | 1993-08-27 | 1994-11-22 | The Procter & Gamble Company | Process for making high density detergent agglomerates using an anhydrous powder additive |
| US5681852A (en) | 1993-11-12 | 1997-10-28 | The Procter & Gamble Company | Desquamation compositions |
| CN1100532C (en) | 1993-11-12 | 2003-02-05 | 普罗克特和甘保尔公司 | Desquamation compositions containing salicylic acid and zwitterionic compounds |
| US5782409A (en) | 1993-12-06 | 1998-07-21 | Paul; Leonard | Air freshening and deodorizing system |
| US5468473A (en) | 1994-02-09 | 1995-11-21 | Innova Products, Inc. | Antiperspirant for hands and feet |
| IT1276525B1 (en) | 1994-04-13 | 1997-10-31 | Webcraft Technologies Inc | DEVICE AND PROCEDURE FOR THE SELECTIVE EXPOSURE OF MICRO-ENCAPSULATED LIQUIDS. |
| US6531156B1 (en) | 1994-04-15 | 2003-03-11 | Temple University | Aqueous solven encapsulation method, apparatus and microcapsules |
| JPH07305049A (en) | 1994-05-12 | 1995-11-21 | Sekisui Chem Co Ltd | Inorganic curable composition and method for producing inorganic cured product |
| PE6995A1 (en) | 1994-05-25 | 1995-03-20 | Procter & Gamble | COMPOSITION INCLUDING A PROPOXYLATED POLYKYLENE OAMINE POLYKYLENE OAMINE POLYMER AS DIRT SEPARATION AGENT |
| US5637401A (en) | 1994-06-08 | 1997-06-10 | Fragrance Technology Trust | Odorant composition, delivery system and method |
| US5725869A (en) | 1994-06-20 | 1998-03-10 | Zeneca Limited | Microsphere reservoirs for controlled release application |
| CA2194161C (en) | 1994-07-01 | 2002-03-26 | Mingchih M. Tseng | Skin engaging member for razor blade assembly |
| US6594904B1 (en) | 1994-07-01 | 2003-07-22 | The Gillette Company | Shaving system |
| US6944952B1 (en) | 1994-07-01 | 2005-09-20 | The Gillette Company | Shaving system |
| US5879584A (en) | 1994-09-10 | 1999-03-09 | The Procter & Gamble Company | Process for manufacturing aqueous compositions comprising peracids |
| US5516448A (en) | 1994-09-20 | 1996-05-14 | The Procter & Gamble Company | Process for making a high density detergent composition which includes selected recycle streams for improved agglomerate |
| US5489392A (en) | 1994-09-20 | 1996-02-06 | The Procter & Gamble Company | Process for making a high density detergent composition in a single mixer/densifier with selected recycle streams for improved agglomerate properties |
| US5691297A (en) | 1994-09-20 | 1997-11-25 | The Procter & Gamble Company | Process for making a high density detergent composition by controlling agglomeration within a dispersion index |
| US6298558B1 (en) | 1994-10-31 | 2001-10-09 | The Gillette Company | Skin engaging member |
| US5573756A (en) | 1995-01-25 | 1996-11-12 | Banner Pharmacaps Inc. | Shampoo conditioner and softgel filled therewith |
| US5534179A (en) | 1995-02-03 | 1996-07-09 | Procter & Gamble | Detergent compositions comprising multiperacid-forming bleach activators |
| US5716938A (en) | 1995-02-07 | 1998-02-10 | Provitt; Robert Darnell | Mattress freshener and room deodorizer composition |
| US5574005A (en) | 1995-03-07 | 1996-11-12 | The Procter & Gamble Company | Process for producing detergent agglomerates from high active surfactant pastes having non-linear viscoelastic properties |
| US5569645A (en) | 1995-04-24 | 1996-10-29 | The Procter & Gamble Company | Low dosage detergent composition containing optimum proportions of agglomerates and spray dried granules for improved flow properties |
| US5702714A (en) | 1995-06-07 | 1997-12-30 | Goss; Louis | Skin conditioner |
| US6358577B1 (en) | 1995-06-07 | 2002-03-19 | Pechiney Emballage Flexible Europe | Membrane permeable to aromatic products |
| US5597936A (en) | 1995-06-16 | 1997-01-28 | The Procter & Gamble Company | Method for manufacturing cobalt catalysts |
| US5565422A (en) | 1995-06-23 | 1996-10-15 | The Procter & Gamble Company | Process for preparing a free-flowing particulate detergent composition having improved solubility |
| US5607980A (en) | 1995-07-24 | 1997-03-04 | The Procter & Gamble Company | Topical compositions having improved skin feel |
| US5576282A (en) | 1995-09-11 | 1996-11-19 | The Procter & Gamble Company | Color-safe bleach boosters, compositions and laundry methods employing same |
| US5750122A (en) | 1996-01-16 | 1998-05-12 | The Procter & Gamble Company | Compositions for treating hair or skin |
| US5674478A (en) | 1996-01-12 | 1997-10-07 | The Procter & Gamble Company | Hair conditioning compositions |
| US5648328A (en) | 1996-02-06 | 1997-07-15 | The Procter & Gamble Company | Process for producing a particulate laundry additive composition for perfume delivery |
| US5656584A (en) | 1996-02-06 | 1997-08-12 | The Procter & Gamble Company | Process for producing a particulate laundry additive composition for perfume delivery |
| US5723420A (en) | 1996-03-04 | 1998-03-03 | The Procter & Gamble Company | Personal cleansing bar compositions which contain a fragrance-releasing complex for improved fragrance delivery |
| JP3859723B2 (en) | 1996-03-04 | 2006-12-20 | オーエスアイ スペシャルティーズ インコーポレーテッド | Silicone amino polyalkylene oxide block copolymer |
| US5756436A (en) | 1996-03-27 | 1998-05-26 | The Procter & Gamble Company | Conditioning shampoo compositions containing select cationic conditioning polymers |
| MA24136A1 (en) | 1996-04-16 | 1997-12-31 | Procter & Gamble | MANUFACTURE OF SURFACE AGENTS. |
| FR2747669B1 (en) | 1996-04-22 | 1998-05-22 | Rhone Poulenc Chimie | PROCESS FOR THE PREPARATION OF HOLLOW SILICA PARTICLES |
| BR9709698A (en) | 1996-06-11 | 1999-08-10 | Procter & Gamble | Process for preparing a particulate laundry washing additive for providing perfumes containing improved physical properties |
| CA2258264A1 (en) | 1996-06-14 | 1997-12-18 | Emisphere Technologies, Inc. | Microencapsulated fragrances and method for preparation |
| US5783536A (en) | 1996-06-26 | 1998-07-21 | Lever Brothers Company, Division Of Conopco, Inc. | Bar composition comprising additive for delivering benefit agent |
| EP0820762A1 (en) | 1996-07-15 | 1998-01-28 | Unilever Plc | Perfume compositions |
| US5929022A (en) | 1996-08-01 | 1999-07-27 | The Procter & Gamble Company | Detergent compositions containing amine and specially selected perfumes |
| EP0829259A1 (en) | 1996-09-04 | 1998-03-18 | Warner-Lambert Company | Foam/gel with microbeads and/or fine particles |
| CA2265804A1 (en) | 1996-09-18 | 1998-03-26 | Athanasios Surutzidis | Process for making particulate laundry additive composition |
| AU4985497A (en) | 1996-10-25 | 1998-05-22 | Mccormick & Company, Inc. | Fat-coated encapsulation compositions and method for preparing the same |
| US6503495B1 (en) | 1996-10-31 | 2003-01-07 | The Procter & Gamble Company | Cosmetic compositions having improved wear and beauty |
| WO1998026808A2 (en) | 1996-12-17 | 1998-06-25 | The Procter & Gamble Company | Absorbent articles with odor control system |
| BR9713619A (en) | 1996-12-23 | 2000-04-11 | Quest Int | Particles containing organic polymer, process for producing them, and product and sunscreen product containing them |
| JPH10195478A (en) | 1996-12-27 | 1998-07-28 | Lion Corp | Cleansing agent composition |
| JP4021513B2 (en) | 1997-02-21 | 2007-12-12 | 水澤化学工業株式会社 | Alumina or alumina hydrate having ultra-low bulk density, high specific surface area, and high porosity, production method and use thereof |
| US5956848A (en) | 1997-02-27 | 1999-09-28 | The Gillette Company | Shaving system |
| JP4489190B2 (en) | 1997-03-07 | 2010-06-23 | ザ、プロクター、エンド、ギャンブル、カンパニー | Bleach composition containing metal bleach catalyst and bleach activator and / or organic percarboxylic acid |
| WO1998039335A1 (en) | 1997-03-07 | 1998-09-11 | The Procter & Gamble Company | Improved methods of making cross-bridged macropolycycles |
| BR9701287A (en) | 1997-03-14 | 1998-11-10 | Unilever Nv | Fabric treatment composition in washing and process to treat fabrics to provide them with dirt repellent properties |
| BR9808409A (en) | 1997-03-20 | 2000-05-16 | Procter & Gamble | Additive part for washing clothes with multiple surface coatings |
| US6248364B1 (en) | 1997-04-07 | 2001-06-19 | 3M Innovative Properties Company | Encapsulation process and encapsulated products |
| US5833971A (en) | 1997-06-16 | 1998-11-10 | Baldwin; Stanley | Perfumed rocks |
| US5800805A (en) | 1997-06-19 | 1998-09-01 | Church & Dwight Co., Inc | Aerosol deodorant product |
| US5945085A (en) | 1997-06-19 | 1999-08-31 | Church & Dwight Co., Inc. | Aerosol deodorant-antiperspirant product |
| US6451754B1 (en) | 1997-08-02 | 2002-09-17 | The Procter & Gamble Company | Process for preparing detergent tablet |
| US6376445B1 (en) | 1997-08-14 | 2002-04-23 | Procter & Gamble Company | Detergent compositions comprising a mannanase and a protease |
| IT1295289B1 (en) | 1997-10-07 | 1999-05-04 | Telos S R L | CO-GRINDED FOR COSMETIC AND DERMATOLOGICAL USE |
| WO1999021532A1 (en) | 1997-10-24 | 1999-05-06 | The Procter & Gamble Company | Cleansing and conditioning article for skin or hair having improved fragrance delivery |
| AU1042999A (en) | 1997-11-10 | 1999-05-31 | Quest International B.V. | Encapsulate of active material in alginate matrix |
| GB9803831D0 (en) | 1998-02-25 | 1998-04-22 | Cussons Int Ltd | Washing composition |
| CA2323181A1 (en) | 1998-03-16 | 1999-09-23 | The Procter & Gamble Company | Methods for regulating skin appearance |
| US6682749B1 (en) | 1998-04-03 | 2004-01-27 | Colgate-Palmolive Company | Low Residue cosmetic composition |
| US6498135B1 (en) | 1998-04-06 | 2002-12-24 | Procter & Gamble Company | Process for producing electrostatically coated non-particulate detergent product |
| US5962018A (en) | 1998-04-28 | 1999-10-05 | Avon Products, Inc. | Method of treating the skin with organic acids in anhydrous microsphere delivery systems |
| US6207782B1 (en) | 1998-05-28 | 2001-03-27 | Cromption Corporation | Hydrophilic siloxane latex emulsions |
| EP0965326B1 (en) | 1998-06-15 | 2007-07-25 | The Procter & Gamble Company | Perfume compositions |
| EP1100864A1 (en) | 1998-07-29 | 2001-05-23 | Reckitt Benckiser N.V. | Composition for use in a dishwashing machine |
| GB9818782D0 (en) | 1998-08-28 | 1998-10-21 | Crosfield Joseph & Sons | Granular compositions |
| DE19842766A1 (en) | 1998-09-18 | 2000-03-23 | Beiersdorf Ag | Emulsifier-free finely dispersed systems of the oil-in-water and water-in-oil type |
| US20070122481A1 (en) | 1998-11-02 | 2007-05-31 | Elan Corporation Plc | Modified Release Compositions Comprising a Fluorocytidine Derivative for the Treatment of Cancer |
| US20070160675A1 (en) | 1998-11-02 | 2007-07-12 | Elan Corporation, Plc | Nanoparticulate and controlled release compositions comprising a cephalosporin |
| US20080113025A1 (en) | 1998-11-02 | 2008-05-15 | Elan Pharma International Limited | Compositions comprising nanoparticulate naproxen and controlled release hydrocodone |
| US20080118556A1 (en) | 1998-11-02 | 2008-05-22 | Elan Corporation, Plc | Modified Release of Compositions Containing a Combination of Carbidopa, Levodopa and Entacapone |
| US20060240105A1 (en) | 1998-11-02 | 2006-10-26 | Elan Corporation, Plc | Multiparticulate modified release composition |
| US20090149479A1 (en) | 1998-11-02 | 2009-06-11 | Elan Pharma International Limited | Dosing regimen |
| US20080102121A1 (en) | 1998-11-02 | 2008-05-01 | Elan Pharma International Limited | Compositions comprising nanoparticulate meloxicam and controlled release hydrocodone |
| PL205109B1 (en) | 1998-11-02 | 2010-03-31 | Elan Pharma Int Ltd | Multiparticulate modified release composition |
| FR2785530B1 (en) | 1998-11-09 | 2000-12-15 | Oreal | COSMETIC COMPOSITION WITHOUT TRANSFER COMPRISING A DISPERSION OF POLYMER PARTICLES AND A SPECIFIC RHEOLOGICAL AGENT |
| US6294514B1 (en) | 1998-11-24 | 2001-09-25 | The Procter & Gamble Company | Process for preparing mono-long chain amine oxide surfactants with low nitrite, nitrosamine and low residual peroxide |
| EP1135392A2 (en) | 1998-11-30 | 2001-09-26 | The Procter & Gamble Company | Process for preparing cross-bridged tetraaza macrocycles |
| US6159485A (en) | 1999-01-08 | 2000-12-12 | Yugenic Limited Partnership | N-acetyl aldosamines, n-acetylamino acids and related n-acetyl compounds and their topical use |
| WO2000041528A2 (en) | 1999-01-14 | 2000-07-20 | Amcol International Corporation | Improved controlled release compositions and method |
| US6258857B1 (en) | 1999-02-04 | 2001-07-10 | Kyowa Industrial Co., Ltd. | Internal liquid composition contained as internal liquid in a releasing container and releasing container product |
| EP1034705A3 (en) | 1999-03-10 | 2001-02-28 | Haarmann & Reimer Gmbh | Encapsulating medium and method for encapsulating perfumes and flavours |
| FR2791992B1 (en) | 1999-04-12 | 2003-06-13 | Rhodia Chimie Sa | MULTI-PARTICLE VECTOR SYSTEM OF ACTIVE MATERIAL (S), ITS PREPARATION AND USES |
| US6767880B1 (en) | 1999-04-19 | 2004-07-27 | The Procter & Gamble Company | Liquid dishwashing detergent composition having polymeric particles |
| DE19918267A1 (en) | 1999-04-22 | 2000-10-26 | Henkel Kgaa | Surfactant-containing material, e.g., a washing-up liquid, which includes microcapsules in which incompatible or sensitive components are enclosed |
| DE19918265A1 (en) | 1999-04-22 | 2000-10-26 | Henkel Kgaa | Abrasive-containing aqueous liquid cleaning or scouring agent for hard surfaces contains microcapsules containing an ingredient to be protected e.g. a perfume |
| US6245733B1 (en) | 1999-04-30 | 2001-06-12 | James Mosbaugh | Porous agglomerations of fused microspheres |
| WO2000067716A1 (en) | 1999-05-07 | 2000-11-16 | The Procter & Gamble Company | Cosmetic compositions comprising cyclic oligosaccharide and fragrance |
| ES2247749T3 (en) | 1999-07-02 | 2006-03-01 | Cognis Ip Management Gmbh | MICROCAPSULES III. |
| DE19932144A1 (en) | 1999-07-09 | 2001-01-11 | Basf Ag | Microcapsule preparations and washing and cleaning agents containing microcapsules |
| WO2001005926A1 (en) | 1999-07-20 | 2001-01-25 | The Procter & Gamble Company | Improved encapsulated oil particles |
| USD424745S (en) | 1999-08-05 | 2000-05-09 | The Gillette Company | Shaving aid strip for razor cartridge |
| JP2001049287A (en) | 1999-08-06 | 2001-02-20 | Yoshihiro Kusaka | Perfume composition |
| EP1218482B1 (en) | 1999-10-04 | 2004-07-21 | Unilever N.V. | Detergent composition comprising fragance particle |
| US6348218B1 (en) | 1999-10-04 | 2002-02-19 | Invent Resources, Inc. | Self dosing skin preparation |
| US7442838B2 (en) | 1999-10-29 | 2008-10-28 | Ciba Specialty Chemicals Corp. | Polymorphic forms of sertraline hydrochloride |
| TWI260315B (en) | 1999-10-29 | 2006-08-21 | Ciba Sc Holding Ag | Polymorphic forms of sertraline hydrochloride |
| US20050119351A1 (en) | 1999-10-29 | 2005-06-02 | Van Der Schaaf Paul A. | Polymorphic forms of sertraline hydrochloride |
| US7838037B2 (en) * | 1999-11-17 | 2010-11-23 | Tagra Biotechnologies Ltd. | Method of microencapsulation |
| US6790814B1 (en) | 1999-12-03 | 2004-09-14 | Procter & Gamble Company | Delivery system having encapsulated porous carrier loaded with additives, particularly detergent additives such as perfumes |
| FR2801811B1 (en) | 1999-12-06 | 2002-05-03 | Gerard Habar | PROCESS FOR THE MANUFACTURE OF MICROCAPSULES CARRYING CATIONIC CHARGES |
| US6235274B1 (en) | 1999-12-21 | 2001-05-22 | International Flavors & Fragrances Inc. | Microparticles which controllably release olfactorily active substances, methods of using same and processes for preparing same |
| DE10000223A1 (en) | 2000-01-05 | 2001-07-12 | Basf Ag | Microcapsules which are useful in, e.g. detergent or skin care compositions, can release a fragrance from a hydrophobic core when the polymer coating of the capsule is broken down |
| US6780507B2 (en) | 2000-02-09 | 2004-08-24 | Analytical Research Systems, Inc. | Hydrocapsules and method of preparation thereof |
| CA2400970A1 (en) | 2000-02-25 | 2001-08-30 | James Mosbaugh | Method of shaping porous agglomerations of fused microspheres |
| DE10012063A1 (en) | 2000-03-14 | 2001-09-20 | Basf Ag | Soft plasticizer-free capsules for use in pharmaceuticals, cosmetics, detergents or plant protectants are made from a polymers obtained by polymerizing a vinyl ester in presence of a polyether substrate |
| DE10015468A1 (en) | 2000-03-29 | 2001-10-11 | Basf Ag | Hard capsules containing polymers and vinyl esters and polyethers, their use and production |
| JP2004500408A (en) | 2000-03-31 | 2004-01-08 | ザ、プロクター、エンド、ギャンブル、カンパニー | Leave-in hair cosmetic composition for increasing volume, comprising fluid-enclosed flexible microspheres |
| US20030104969A1 (en) | 2000-05-11 | 2003-06-05 | Caswell Debra Sue | Laundry system having unitized dosing |
| AU2001263062A1 (en) | 2000-05-11 | 2001-11-20 | The Procter And Gamble Company | Highly concentrated fabric softener compositions and articles containing such compositions |
| US7208463B2 (en) | 2000-06-02 | 2007-04-24 | The Procter & Gamble Company | Fragrance compositions |
| AU2001274208B2 (en) | 2000-06-05 | 2005-03-17 | Syngenta Limited | Novel microcapsules |
| DE10031132A1 (en) | 2000-06-30 | 2002-01-17 | Henkel Kgaa | Process for the production of active ingredient-containing capsules with an ultra-thin wall layer |
| US20020016269A1 (en) | 2000-07-06 | 2002-02-07 | The Procter & Gamble Co. | Particle perfume delivery system |
| US6696049B2 (en) | 2000-07-10 | 2004-02-24 | The Procter & Gamble Company | Cosmetic compositions |
| US7041767B2 (en) | 2000-07-27 | 2006-05-09 | Ge Bayer Silicones Gmbh & Co. Kg | Polysiloxane polymers, method for their production and the use thereof |
| ES2227271T3 (en) | 2000-07-27 | 2005-04-01 | GE BAYER SILICONES GMBH & CO. KG | POLYAMONIO-POLISILOXANE COMPOUNDS, PROCEDURE FOR PRODUCTION AND USE. |
| CA2385221C (en) | 2000-09-06 | 2011-01-11 | Appleton Papers Inc. | In situ microencapsulated adhesive |
| US7407650B2 (en) | 2000-10-27 | 2008-08-05 | The Procter & Gamble Company | Fragrance compositions |
| US7413731B2 (en) | 2000-10-27 | 2008-08-19 | The Procter And Gamble Company | Fragrance compositions |
| EP1201743A1 (en) | 2000-10-31 | 2002-05-02 | The Procter & Gamble Company | Detergent compositions |
| US7208465B2 (en) | 2000-11-03 | 2007-04-24 | The Procter & Gamble Company | Methods and compositions for improved fragrancing of a surface |
| DE10062585A1 (en) | 2000-12-15 | 2002-06-27 | Henkel Kgaa | Particulate additives for compositions used in dishwashing or textile washing machines, have the active component(s) enclosed in a shell which is especially of a polymeric material |
| DE10064878A1 (en) | 2000-12-27 | 2002-07-04 | Cognis Deutschland Gmbh | Encapsulation of emulsions |
| US6864223B2 (en) | 2000-12-27 | 2005-03-08 | Colgate-Palmolive Company | Thickened fabric conditioners |
| GB0104698D0 (en) | 2001-02-26 | 2001-04-11 | Syngenta Ltd | Process for producing microcapsules |
| DE10112441A1 (en) | 2001-03-15 | 2002-09-19 | Degussa | Silica by precipitation with a constant alkali number and its use |
| ATE288318T1 (en) | 2001-03-22 | 2005-02-15 | Cognis Iberia Sl | MICRO CAPSULES (VIII) |
| ATE289865T1 (en) | 2001-03-22 | 2005-03-15 | Cognis Iberia Sl | MICRO CAPSULES (VII) |
| WO2002076479A1 (en) | 2001-03-27 | 2002-10-03 | Bime Laboratories | Composition for treating acne vulgaris and fabrication method |
| US7230750B2 (en) | 2001-05-15 | 2007-06-12 | E Ink Corporation | Electrophoretic media and processes for the production thereof |
| EP1247568A1 (en) | 2001-04-03 | 2002-10-09 | Primacare S.L., c/o Cognis Iberica S.L. | Microcapsules (XIII) |
| GB2374082A (en) | 2001-04-04 | 2002-10-09 | Procter & Gamble | Particles for a detergent product |
| AU2002303583A1 (en) | 2001-05-04 | 2002-11-18 | The Procter And Gamble Company | Air freshening compositions, articles comprising same and methods for preparing same |
| JP2002326904A (en) | 2001-05-08 | 2002-11-15 | Shiseido Co Ltd | Powdery cosmetic |
| US7241835B2 (en) | 2001-05-16 | 2007-07-10 | General Electric Company | Cosmetic compositions comprising silicone gels |
| US20030203978A1 (en) | 2001-05-16 | 2003-10-30 | O'brien Michael Joseph | Cosmetic compositions comprising silicone gels comprising entrapped, occluded or encapsulated pigments |
| US20030031722A1 (en) | 2001-06-04 | 2003-02-13 | Hongjie Cao | Starch-oil composites for use in personal care applications |
| US7270828B2 (en) | 2001-06-20 | 2007-09-18 | The Procter & Gamble Company | Personal care composition comprising hydrophobic gel |
| GB0115681D0 (en) | 2001-06-27 | 2001-08-22 | Ciba Spec Chem Water Treat Ltd | Process for making polymeric particles |
| WO2003005986A1 (en) | 2001-07-11 | 2003-01-23 | Unilever Plc | Hair treatment compositions |
| US6881482B2 (en) | 2001-08-16 | 2005-04-19 | Southwest Research Institute | Microencapsulation using electromagnetic energy and core and shell materials with different dielectric constants and dissipation factors |
| ATE319807T1 (en) | 2001-08-20 | 2006-03-15 | Unilever Nv | PHOTO BLEACH SPECKS AND DETERGENTS CONTAINING THEM |
| US7211273B2 (en) | 2001-08-28 | 2007-05-01 | Unilever Home & Personal Care Usa, Division Of Conopco, Inc. | Detergent or personal care composition with oil capsules |
| GB0121176D0 (en) | 2001-08-31 | 2001-10-24 | Unilever Plc | Perfumed coloured speckle composition and particulate laundry detergent compositions containing it |
| US6982256B2 (en) | 2001-09-07 | 2006-01-03 | Boehringer Ingelheim Pharma Kg | Tolerance of 4-(4-(2-pyrrolylcarbonyl)-1-piperazinyl)-3-trifluoromethyl-benzoylguanidine in intravenous administration |
| JP4082558B2 (en) | 2001-09-21 | 2008-04-30 | 株式会社リコー | Friction drive |
| CN1655753A (en) | 2001-10-03 | 2005-08-17 | 宝洁公司 | Shampoo containing particles and a deposition aid |
| US20030108501A1 (en) | 2001-10-03 | 2003-06-12 | Hofrichter Brian David | Shampoo containing a cationic polymer and particles |
| US7186679B2 (en) | 2001-10-11 | 2007-03-06 | Hair Systems, Inc. | Stabilization of fragrances in salt mixtures by dual encapsulation and entrapment |
| US20030077378A1 (en) | 2001-10-22 | 2003-04-24 | Lou Wen Chin | Hydroxypropyl cellulose encapsulation material |
| US6607717B1 (en) | 2001-10-24 | 2003-08-19 | Dow Corning Corporation | Silicon based quaternary ammonium functional compositions and their applications |
| US6482969B1 (en) | 2001-10-24 | 2002-11-19 | Dow Corning Corporation | Silicon based quaternary ammonium functional compositions and methods for making them |
| MXPA04003821A (en) | 2001-11-02 | 2004-07-30 | Procter & Gamble | Composition containing a cationic polymer and water insoluble solid material. |
| FR2831802B1 (en) | 2001-11-08 | 2004-10-15 | Oreal | COSMETIC COMPOSITIONS CONTAINING AN AMINO SILICONE AND A THICKENING AGENT AND THEIR USES |
| WO2003043728A1 (en) | 2001-11-22 | 2003-05-30 | Firmenich Sa | Perfuming or flavouring microcapsules comprising a fireproofing agent |
| DE10163142A1 (en) | 2001-12-20 | 2003-07-10 | Henkel Kgaa | Polymeric fragrance capsules and their manufacture |
| US20030186834A1 (en) | 2002-01-09 | 2003-10-02 | Croda, Inc. | Immidazoline quats |
| US20040138088A1 (en) | 2002-01-09 | 2004-07-15 | Croda, Inc. | Immidazoline quats |
| US7799421B2 (en) | 2002-02-14 | 2010-09-21 | Akzo Nobel N.V. | Controlled release materials |
| US6806249B2 (en) | 2002-02-28 | 2004-10-19 | Unilever Home & Personal Care Usa, A Division Of Conopco | Perfume containing surfactant compositions having perfume burst when diluted |
| US6797683B2 (en) | 2002-03-04 | 2004-09-28 | Unilever Home & Personal Care Usa, Division Of Conopco, Inc. | Ordered liquid crystalline cleansing composition with benefit agent particles |
| JP4156851B2 (en) | 2002-03-22 | 2008-09-24 | オリンパス株式会社 | Microdissection device |
| US7419947B2 (en) | 2002-03-27 | 2008-09-02 | Novozymes A/S | Process for preparing granules with filamentous coatings |
| US7053034B2 (en) | 2002-04-10 | 2006-05-30 | Salvona, Llc | Targeted controlled delivery compositions activated by changes in pH or salt concentration |
| US20030194416A1 (en) | 2002-04-15 | 2003-10-16 | Adl Shefer | Moisture triggered release systems comprising aroma ingredients providing fragrance burst in response to moisture |
| US20040175404A1 (en) | 2002-04-15 | 2004-09-09 | Adi Shefer | Moisture triggered sealed release system |
| US20030199412A1 (en) | 2002-04-17 | 2003-10-23 | Gargi Gupta | Fragrance containing cleaning product |
| US20030216488A1 (en) | 2002-04-18 | 2003-11-20 | The Procter & Gamble Company | Compositions comprising a dispersant and microcapsules containing an active material |
| US20030215417A1 (en) | 2002-04-18 | 2003-11-20 | The Procter & Gamble Company | Malodor-controlling compositions comprising odor control agents and microcapsules containing an active material |
| US7192599B2 (en) | 2002-06-03 | 2007-03-20 | Mmp, Inc. | Mattifying oil-in-water emulsion |
| CN101157015B (en) | 2002-06-12 | 2011-11-02 | 创普太克公司 | Encapsulated active particles and methods for making and using the same |
| EP1517983B1 (en) | 2002-06-27 | 2007-01-03 | Unilever N.V. | Perfume composition |
| US7235261B2 (en) | 2002-06-27 | 2007-06-26 | Haarmann & Reimer Corporation | Controlled release encapsulation |
| ATE349229T1 (en) | 2002-07-11 | 2007-01-15 | Firmenich & Cie | COMPOSITION FOR THE CONTROLLED RELEASE OF PERFUME AND FLAVORS |
| US6846785B2 (en) | 2002-07-31 | 2005-01-25 | The Dial Corporation | Liquid soap with vitamin beads and method for making same |
| CA2489354C (en) | 2002-08-02 | 2013-09-03 | The Gillette Company | Clear personal care compositions containing visible capsules |
| JP4865225B2 (en) | 2002-08-14 | 2012-02-01 | ジボダン・ネーデルランド・サービシーズ・ビー・ブイ | Composition comprising encapsulated material |
| CA2499423A1 (en) | 2002-09-04 | 2004-03-18 | Niraj Vasisht | Microencapsulation of oxygen or water sensitive materials |
| GB0220580D0 (en) | 2002-09-04 | 2002-10-09 | Unilever Plc | Hair treatment composition |
| JP2004099743A (en) | 2002-09-10 | 2004-04-02 | T Hasegawa Co Ltd | Encapsulated perfume and its use |
| US7125835B2 (en) | 2002-10-10 | 2006-10-24 | International Flavors & Fragrances Inc | Encapsulated fragrance chemicals |
| BR0303954A (en) | 2002-10-10 | 2004-09-08 | Int Flavors & Fragrances Inc | Composition, fragrance, method for dividing an olfactory effective amount of fragrance into a non-rinse and non-rinse product |
| US7585824B2 (en) | 2002-10-10 | 2009-09-08 | International Flavors & Fragrances Inc. | Encapsulated fragrance chemicals |
| US20040071742A1 (en) | 2002-10-10 | 2004-04-15 | Popplewell Lewis Michael | Encapsulated fragrance chemicals |
| US6924261B2 (en) | 2002-11-01 | 2005-08-02 | Colgate-Palmolive Co. | Aqueous composition comprising oligomeric esterquats |
| US20040091445A1 (en) | 2002-11-01 | 2004-05-13 | The Procter & Gamble Company | Rinse-off personal care compositions comprising cationic perfume polymeric particles |
| US7524807B2 (en) | 2002-11-01 | 2009-04-28 | The Procter & Gamble Company | Rinse-off personal care compositions comprising anionic and/or nonionic perfume polymeric particles |
| US20040109920A1 (en) | 2002-12-04 | 2004-06-10 | Bioactives Llc | Coated carotenoid cyclodextrin complexes |
| BR0205449A (en) | 2002-12-09 | 2006-03-07 | Biolab Sanus Farmaceutica Ltda | pharmaceutical compositions of peptides secreted by snake venom glands, particularly from bothrops jararaca, vasopeptidase inhibitors, evasins, their analogues, derivatives and associated products for the development of applications and use in chronic degenerative diseases |
| WO2004060418A1 (en) | 2003-01-07 | 2004-07-22 | Hanjin Printing & Chemical Co., Ltd. | Deodorizing film of sanitary absorbent |
| GB0300808D0 (en) | 2003-01-14 | 2003-02-12 | Unilever Plc | Home and personal care compositions with lubricants |
| US9068234B2 (en) | 2003-01-21 | 2015-06-30 | Ptc Therapeutics, Inc. | Methods and agents for screening for compounds capable of modulating gene expression |
| US8460864B2 (en) | 2003-01-21 | 2013-06-11 | Ptc Therapeutics, Inc. | Methods for identifying compounds that modulate untranslated region-dependent gene expression and methods of using same |
| US8426194B2 (en) | 2003-01-21 | 2013-04-23 | Ptc Therapeutics, Inc. | Methods and agents for screening for compounds capable of modulating VEGF expression |
| EP1443058A1 (en) | 2003-01-29 | 2004-08-04 | Firmenich Sa | Polymeric particles and fragrance delivery systems |
| US20070031463A1 (en) | 2003-02-26 | 2007-02-08 | Spiros Fotinos | Methods and devices for releasing volatile substances |
| US7137570B2 (en) | 2003-02-28 | 2006-11-21 | American Covers, Inc. | Air freshener and method |
| US20050169793A1 (en) | 2003-02-28 | 2005-08-04 | American Covers, Inc. | Air freshener and method |
| US7293719B2 (en) | 2003-02-28 | 2007-11-13 | American Covers, Inc. | Air freshener and method |
| US20040175347A1 (en) | 2003-03-04 | 2004-09-09 | The Procter & Gamble Company | Regulation of mammalian keratinous tissue using hexamidine compositions |
| US20060127430A1 (en) | 2003-04-18 | 2006-06-15 | Bioderm Research | Controlled-Release of Cosmetic and Pharmaceutical Agents via Osmotic Nano-Diffusion from Zeolite Cage Complexes |
| US20040208902A1 (en) | 2003-04-18 | 2004-10-21 | Gupta Shyam K. | Controlled-release nano-diffusion delivery systems for cosmetic and pharmaceutical compositions |
| EP1471137B1 (en) | 2003-04-23 | 2013-08-07 | The Procter & Gamble Company | A composition comprising a surface deposition enhacing cationic polymer |
| DE10319738A1 (en) | 2003-04-30 | 2004-11-18 | Basf Ag | Process for the preparation of aqueous dispersions of polyelectrolyte complexes and their use for increasing the wet strength of paper, cardboard and cardboard |
| ES2285457T3 (en) | 2003-05-07 | 2007-11-16 | Firmenich Sa | SPRAY PERFUME WITH AN IMPROVED TENACITY. |
| US20050014674A1 (en) | 2003-05-28 | 2005-01-20 | L'oreal | Cosmetic compositions for making up and/or caring for skin |
| CA2533406C (en) | 2003-07-31 | 2013-01-22 | Sol-Gel Technologies Ltd. | Microcapsules loaded with active ingredients and a method for their preparation |
| EP1502646B1 (en) | 2003-08-01 | 2016-07-13 | The Procter & Gamble Company | Microcapsules |
| US20050038188A1 (en) | 2003-08-14 | 2005-02-17 | Dongchan Ahn | Silicones having improved chemical resistance and curable silicone compositions having improved migration resistance |
| US7226607B2 (en) * | 2003-09-11 | 2007-06-05 | The Procter & Gamble Company | Compositions comprising a dispersant and microcapsules containing an active material and a stabilizer |
| JP2007511625A (en) | 2003-10-29 | 2007-05-10 | フイルメニツヒ ソシエテ アノニム | Perfume delivery system based on inorganic nanoparticles |
| US20060263898A1 (en) | 2003-10-29 | 2006-11-23 | Walter Paget | Perfume delivery systems based on inorganic nano particles |
| EP1711231A1 (en) | 2003-10-31 | 2006-10-18 | Firmenich Sa | Fragrance delivery system for surface cleaners and conditioners |
| JP2007510754A (en) | 2003-11-12 | 2007-04-26 | フレキシトラル インコーポレイテッド | New oxynitriles |
| US20050112152A1 (en) | 2003-11-20 | 2005-05-26 | Popplewell Lewis M. | Encapsulated materials |
| US20050113282A1 (en) | 2003-11-20 | 2005-05-26 | Parekh Prabodh P. | Melamine-formaldehyde microcapsule slurries for fabric article freshening |
| US7105064B2 (en) | 2003-11-20 | 2006-09-12 | International Flavors & Fragrances Inc. | Particulate fragrance deposition on surfaces and malodour elimination from surfaces |
| EP1541121B1 (en) | 2003-12-11 | 2007-03-21 | Rohm And Haas Company | System and process for releasing encapsulated active ingredients |
| DE10358799A1 (en) | 2003-12-12 | 2005-07-14 | Basf Ag | Use of water-dispersible copolymers of hydroxyalkyl (meth) acrylates for the production of hard capsules |
| US20050129759A1 (en) | 2003-12-16 | 2005-06-16 | Milan Sojka | Sustained release compositions and controlled delivery method |
| JP2005194308A (en) | 2003-12-26 | 2005-07-21 | Mitsubishi Chemicals Corp | Sustained release perfume carrier and sustained release perfume |
| US7399324B2 (en) | 2004-01-29 | 2008-07-15 | Camovision Of Georgia, Llc | Active agent delivery device |
| US7311900B2 (en) | 2004-02-11 | 2007-12-25 | Belle-Aire Fregrances, Inc. | Gel/air freshener system |
| US7452547B2 (en) | 2004-03-31 | 2008-11-18 | Johnson&Johnson Consumer Co., Inc. | Product for treating the skin comprising a polyamine microcapsule wall and a skin lightening agent |
| EP1735006B1 (en) | 2004-04-05 | 2007-09-26 | Basf Aktiengesellschaft | Polymer particles containing active agents |
| US20050227907A1 (en) | 2004-04-13 | 2005-10-13 | Kaiping Lee | Stable fragrance microcapsule suspension and process for using same |
| US20050226900A1 (en) | 2004-04-13 | 2005-10-13 | Winton Brooks Clint D | Skin and hair treatment composition and process for using same resulting in controllably-releasable fragrance and/or malodour counteractant evolution |
| US7211556B2 (en) | 2004-04-15 | 2007-05-01 | Colgate-Palmolive Company | Fabric care composition comprising polymer encapsulated fabric or skin beneficiating ingredient |
| DE102004020400A1 (en) | 2004-04-23 | 2005-11-17 | Henkel Kgaa | Perfumed solids |
| ES2308064T3 (en) | 2004-04-29 | 2008-12-01 | Kao Corporation | PERFUME PARTICLES AND A PROCESS FOR PREPARATION. |
| US20050276831A1 (en) | 2004-06-10 | 2005-12-15 | Dihora Jiten O | Benefit agent containing delivery particle |
| ES2393725T3 (en) | 2004-08-10 | 2012-12-27 | Basf Se | Preparation of coarse particle microcapsules |
| EP1637188A1 (en) | 2004-08-20 | 2006-03-22 | Firmenich Sa | Improved liquid/sprayable compositions comprising fragranced aminoplast capsules |
| GB0419268D0 (en) | 2004-08-31 | 2004-09-29 | Givaudan Sa | Organic compounds |
| WO2006027664A2 (en) | 2004-09-08 | 2006-03-16 | Firmenich Sa | Process for producing nano-capsules containing a fragrance |
| WO2006038110A2 (en) | 2004-10-08 | 2006-04-13 | Firmenich Sa | Amphiphilic star block copolymers |
| EP1655344A1 (en) | 2004-11-05 | 2006-05-10 | The Procter & Gamble Company | Polymeric compositions for sustained release of volatile materials |
| EP1661977A1 (en) | 2004-11-29 | 2006-05-31 | The Procter & Gamble Company | Detergent compositions |
| RU2378332C2 (en) | 2004-12-21 | 2010-01-10 | Дегусса Гмбх | System for delivering fragrant substances |
| JPWO2006070578A1 (en) | 2004-12-28 | 2008-06-12 | クオリカプス株式会社 | Hard capsule band seal |
| DE102005002411A1 (en) | 2005-01-18 | 2006-07-27 | Basf Ag | Coarse-particled microcapsule preparation |
| US20060165740A1 (en) | 2005-01-24 | 2006-07-27 | Goldschmidt Chemical Corporation | Perfume delivery system |
| US20060166855A1 (en) | 2005-01-25 | 2006-07-27 | Howard Murad | Fragrance delivery system |
| FR2881048B1 (en) | 2005-01-27 | 2009-07-17 | Laggridore Soc Par Actions Sim | PERFUME COMPOSITION IN COMPACT FORM |
| FR2881959B1 (en) | 2005-02-14 | 2007-04-06 | Monique Cervilla | SCENTED COMPOSITION WITH CONTROLLED DISSEMINATION |
| US20070298061A1 (en) | 2005-02-25 | 2007-12-27 | Cadbury Adams Usa Llc | Process for manufacturing a delivery system for active components as part of an edible compostion |
| EP1702674A1 (en) | 2005-03-16 | 2006-09-20 | Cognis IP Management GmbH | Visible beads |
| US20060248665A1 (en) | 2005-05-06 | 2006-11-09 | Pluyter Johan G L | Encapsulated fragrance materials and methods for making same |
| US20070207174A1 (en) | 2005-05-06 | 2007-09-06 | Pluyter Johan G L | Encapsulated fragrance materials and methods for making same |
| US20060275237A1 (en) | 2005-05-09 | 2006-12-07 | Bissett Donald L | Skin care compositions containing idebenone |
| US20060258557A1 (en) | 2005-05-11 | 2006-11-16 | Popplewell Lewis M | Hard surface cleaning compositions and methods for making same |
| US7575633B2 (en) | 2005-05-17 | 2009-08-18 | Nordson Corporation | Fluid dispenser with positive displacement pump |
| US20060263311A1 (en) | 2005-05-19 | 2006-11-23 | Scavone Timothy A | Consumer noticeable improvement in wetness protection using solid antiperspirant compositions |
| US8147808B2 (en) | 2005-05-19 | 2012-04-03 | The Procter & Gamble Company | Consumer noticeable improvement in wetness protection using solid antiperspirant compositions |
| US20060292098A1 (en) | 2005-05-19 | 2006-12-28 | Scavone Timothy A | Consumer noticeable improvement in wetness protection |
| US8632755B2 (en) | 2005-05-19 | 2014-01-21 | The Procter & Gamble Company | Consumer noticeable improvement in wetness protection |
| WO2006127454A2 (en) | 2005-05-23 | 2006-11-30 | Appleton Papers Inc. | Oil-in-water capsule manufacture process and microcapsules produced by such process |
| WO2006132863A1 (en) | 2005-06-03 | 2006-12-14 | Isp Investments Inc. | Fragrance delivery system |
| US20070041929A1 (en) | 2005-06-16 | 2007-02-22 | Torgerson Peter M | Hair conditioning composition comprising silicone polymers containing quaternary groups |
| DE102005029777A1 (en) | 2005-06-24 | 2007-01-04 | Henkel Kgaa | Deodorant or antiperspirant stick in oil-in water dispersion, useful e.g. in cosmetic to reduce body smell comprises e.g. lipid/wax component, non-ionic oil-in-water emulsifying agent, non-ionic water-in-oil emulsifying agent and oil |
| FR2887886B1 (en) | 2005-07-01 | 2010-01-08 | Oreal | NEUTRALIZED CATIONIC POLYMER, COMPOSITION COMPRISING THE SAME AND COSMETIC TREATMENT METHOD |
| JP4916682B2 (en) | 2005-07-08 | 2012-04-18 | 花王株式会社 | Aqueous composition |
| EP1745770A1 (en) | 2005-07-13 | 2007-01-24 | L'Oréal | Bilayered cosmetic product, its uses and make up kit containing said product |
| US20100068163A1 (en) | 2005-07-13 | 2010-03-18 | L'oreal | Keratin Fibre Coating Composition Comprising a Liquid Fatty Phase and a Tackifying Resin |
| US20070286904A1 (en) | 2005-08-31 | 2007-12-13 | Popplewell Lewis M | Structured materials |
| US20070048339A1 (en) | 2005-08-31 | 2007-03-01 | Popplewell Lewis M | Structured materials |
| GB0518059D0 (en) | 2005-09-06 | 2005-10-12 | Dow Corning | Delivery system for releasing active ingredients |
| US20080213451A1 (en) | 2005-09-12 | 2008-09-04 | Miharu Ogura | Flavor Composition or Fragrance Composition, and Flavor-Improving Agents |
| US20070071781A1 (en) | 2005-09-19 | 2007-03-29 | Nelson Technologies, Inc. | Compositions for rupturable capsules and methods of making the same |
| US20070071978A1 (en) | 2005-09-23 | 2007-03-29 | Sojka Milan F | Cosmetic Composition Containing Thermoplastic Microspheres and Skin Beneficial Agents |
| WO2007040517A1 (en) | 2005-09-30 | 2007-04-12 | Avon Products, Inc. | Long-lasting cosmetic composition, method for using, and process for making |
| US20070078071A1 (en) | 2005-09-30 | 2007-04-05 | Kaiping Lee | Spray dry capsule products and methods for preparing and using same |
| JP2009514660A (en) | 2005-10-28 | 2009-04-09 | ソーラーブレ, インコーポレイテッド | Photo-responsive microencapsulated materials, compositions and methods for their use |
| US20090047434A1 (en) | 2005-11-11 | 2009-02-19 | Gil Trophardy | Flavor or Fragrance Capsules |
| GB0524659D0 (en) | 2005-12-02 | 2006-01-11 | Unilever Plc | Improvements relating to fabric treatment compositions |
| US20070134411A1 (en) | 2005-12-14 | 2007-06-14 | General Electric Company | Method for making compositions containing microcapsules and compositions made thereof |
| US20070138672A1 (en) | 2005-12-15 | 2007-06-21 | Kaiping Lee | Process for preparing a high stability microcapsule product and method for using same |
| US20070138671A1 (en) | 2005-12-15 | 2007-06-21 | Anastasiou Theodore J | Encapsulated active material with reduced formaldehyde potential |
| US20070138674A1 (en) | 2005-12-15 | 2007-06-21 | Theodore James Anastasiou | Encapsulated active material with reduced formaldehyde potential |
| US20070138673A1 (en) | 2005-12-15 | 2007-06-21 | Kaiping Lee | Process for Preparing a High Stability Microcapsule Product and Method for Using Same |
| US9427391B2 (en) | 2006-01-09 | 2016-08-30 | The Procter & Gamble Company | Personal care compositions containing cationic synthetic copolymer and a detersive surfactant |
| EP1808115B1 (en) | 2006-01-12 | 2017-03-22 | The Procter & Gamble Company | Cleaning implement |
| WO2007099469A2 (en) | 2006-02-28 | 2007-09-07 | The Procter & Gamble Company | Benefit agent containing delivery particle |
| EP1996692B2 (en) * | 2006-03-22 | 2020-04-01 | The Procter and Gamble Company | Liquid treatment unitized dose composition |
| US20080027575A1 (en) | 2006-04-21 | 2008-01-31 | Jones Stevan D | Modeling systems for health and beauty consumer goods |
| WO2007128326A1 (en) | 2006-05-03 | 2007-11-15 | Evonik Goldschmidt Gmbh | Perfume delivery system for cleaners |
| US20080032909A1 (en) * | 2006-05-05 | 2008-02-07 | De Buzzaccarini Francesco | Compact fluid laundry detergent composition |
| US20070286837A1 (en) | 2006-05-17 | 2007-12-13 | Torgerson Peter M | Hair care composition comprising an aminosilicone and a high viscosity silicone copolymer emulsion |
| US7985445B2 (en) | 2006-05-18 | 2011-07-26 | Appleton Papers Inc. | Water-in-oil capsule manufacture process and microcapsules produced by such process |
| WO2007135583A2 (en) | 2006-05-19 | 2007-11-29 | Firmenich Sa | One step spray-drying process |
| GB0610562D0 (en) | 2006-05-30 | 2006-07-05 | Givaudan Sa | Microcapsules |
| CN101466347A (en) | 2006-06-13 | 2009-06-24 | 吉万奥丹股份有限公司 | Encapsulation compositions |
| US7772175B2 (en) | 2006-06-20 | 2010-08-10 | The Procter & Gamble Company | Detergent compositions for cleaning and fabric care comprising a benefit agent, deposition polymer, surfactant and laundry adjuncts |
| CN101506344A (en) | 2006-06-30 | 2009-08-12 | 高露洁-棕榄公司 | Cationic polymer stabilized microcapsule composition |
| EP2043773B1 (en) | 2006-07-13 | 2009-12-16 | Basf Se | Polyelectrolyte-modified microcapsules |
| CN101528339B (en) | 2006-10-17 | 2013-03-27 | 巴斯夫欧洲公司 | Microcapsules |
| US20080107615A1 (en) | 2006-11-08 | 2008-05-08 | L'oreal | Detackified compositions |
| EP1923423B1 (en) | 2006-11-14 | 2013-07-03 | Sociedad Anónima Minera Catalano-Aragonesa | Process for the additivation of synthetic fibres, artificial fibres and polymers with special properties |
| KR20090079957A (en) | 2006-11-17 | 2009-07-22 | 시바 홀딩 인크 | Microcapsules, their use and processes for their manufacture |
| GB0623110D0 (en) | 2006-11-21 | 2006-12-27 | Ciba Sc Holding Ag | Microcapules, their use and processes for their manufacture |
| JP2010511002A (en) | 2006-11-28 | 2010-04-08 | ユニリーバー・ナームローゼ・ベンノートシヤープ | Hair treatment composition |
| US8129327B2 (en) | 2006-12-01 | 2012-03-06 | The Procter & Gamble Company | Packaging for high moisture bar soap |
| DE102006058253A1 (en) | 2006-12-08 | 2008-06-19 | Cavis Microcaps Gmbh | Microcapsule, useful as accumulator for materials e.g. in industrial processes, comprises a capsule core, which contains a gelable matrix substance and filler, which forms a porous structure in which free-release material is present |
| US7833960B2 (en) | 2006-12-15 | 2010-11-16 | International Flavors & Fragrances Inc. | Encapsulated active material containing nanoscaled material |
| JP2008156565A (en) | 2006-12-26 | 2008-07-10 | Lion Corp | Additive particle for washing, method for producing the same, and detergent for clothes containing the same |
| DE502007003130D1 (en) | 2007-01-02 | 2010-04-29 | Symrise Gmbh & Co Kg | Mixtures with 3- (4-methylcyclohex-3-enyl) butyraldehyde and 2,6-dimethyl-7-octene-2-ol |
| WO2008100601A2 (en) | 2007-02-15 | 2008-08-21 | The Procter & Gamble Company | Benefit agent delivery compositions |
| GB0703679D0 (en) | 2007-02-26 | 2007-04-04 | Quest Int Serv Bv | Perfume compositions |
| DE102007010109A1 (en) | 2007-02-28 | 2008-09-04 | Henkel Ag & Co. Kgaa | Particle forming carrier partially coated with a fine particulate active agent carrier matrix, useful e.g. in detergent composition and in a procedure for the fumigation of textiles |
| US8771713B2 (en) | 2007-03-12 | 2014-07-08 | Board Of Regents, The University Of Texas System | Method and process for the production of multi-coated recognitive and releasing systems |
| US8821899B2 (en) | 2007-03-12 | 2014-09-02 | Board Of Regents, The University Of Texas System | Method and process for the production of multi-coated recognitive and releasing systems |
| WO2008112826A1 (en) | 2007-03-12 | 2008-09-18 | Board Of Regents, The University Of Texas System | Method and process for the production of multi-coated recognitive and releasing systems |
| US8741316B2 (en) | 2007-03-12 | 2014-06-03 | Board Of Regents, The University Of Texas System | Highly porous, recognitive polymer systems |
| US8349300B2 (en) | 2007-04-19 | 2013-01-08 | The Procter & Gamble Company | Personal care compositions containing at least two cationic polymers and an anionic surfactant |
| WO2008129028A1 (en) | 2007-04-24 | 2008-10-30 | Unilever Plc | Perfumed dispensing device |
| FR2915990B1 (en) | 2007-05-07 | 2010-09-10 | Prodisis | POROUS CERAMICS FOR THE DISTRIBUTION OF FRAGRANCES |
| US8470762B2 (en) | 2007-05-31 | 2013-06-25 | Colgate-Palmolive Company | Fabric softening compositions comprising polymeric materials |
| GB0710369D0 (en) | 2007-06-01 | 2007-07-11 | Unilever Plc | Improvements relating to perfume particles |
| US20100040884A1 (en) | 2008-06-04 | 2010-02-18 | Appleton Papers Inc. | Benefit agent containing delivery particles |
| WO2008152543A1 (en) | 2007-06-11 | 2008-12-18 | The Procter & Gamble Company | Benefit agent containing delivery particle |
| US20080311064A1 (en) | 2007-06-12 | 2008-12-18 | Yabin Lei | Higher Performance Capsule Particles |
| EP2022780A2 (en) | 2007-06-19 | 2009-02-11 | Givaudan SA | Cysteine derivatives which counteract malodour |
| US20080317788A1 (en) | 2007-06-22 | 2008-12-25 | Rafael Louzan Garcia | Additive for Domestic Washing Processes |
| US7538077B2 (en) | 2007-06-26 | 2009-05-26 | Conopco, Inc. | Extruded personal washing bars with plate-like polymeric inclusions |
| US20090029900A1 (en) | 2007-07-27 | 2009-01-29 | The Procter & Gamble Company | Personal care article for sequentially dispensing compositions with distinct fragrance characters |
| US20090324660A1 (en) | 2007-07-27 | 2009-12-31 | Jonathan Robert Cetti | Personal-care article for sequentially dispensing compositions with different fragrances |
| US20090035365A1 (en) | 2007-07-30 | 2009-02-05 | Lewis Michael Popplewell | Density Controlled Capsule Particles and Methods of Making the Same |
| JP5101202B2 (en) | 2007-08-02 | 2012-12-19 | 太陽化学株式会社 | Spherical mesoporous material |
| CN101088567A (en) | 2007-08-03 | 2007-12-19 | 梁小江 | Moisture-proof deodorant and its prepn process |
| US20090053165A1 (en) | 2007-08-20 | 2009-02-26 | Mark Anthony Brown | Method for Treating Damaged Hair |
| GB0718300D0 (en) | 2007-09-20 | 2007-10-31 | Univ Leeds | Microcapsules and methods |
| GB0718532D0 (en) | 2007-09-22 | 2007-10-31 | Unilever Plc | Improvements relating to fabric treatment compositions |
| WO2009047127A1 (en) | 2007-10-12 | 2009-04-16 | Unilever Plc | Granular detergent compositions with contrasting lamellar visual cues |
| WO2009080695A1 (en) | 2007-12-21 | 2009-07-02 | Akzo Nobel N.V. | Compositions for protection and release of active materials |
| KR101415994B1 (en) | 2007-12-27 | 2014-07-08 | (주)아모레퍼시픽 | Double layered polymer capsules for the stabilization of carotenoids, the process for preparing the same, and the cosmetic composition containing the same |
| CA2711200A1 (en) | 2008-01-15 | 2009-07-23 | Basf Se | Scent-comprising microcapsules with improved release behavior |
| CN102186451A (en) | 2008-01-18 | 2011-09-14 | 宝洁公司 | Concentrated personal cleansing compositions |
| KR100982275B1 (en) | 2008-01-28 | 2010-09-15 | 이대희 | Composite for emitting fragrance including compound fatty acid silver and organic acid silver |
| FR2926978B1 (en) | 2008-02-06 | 2010-05-07 | Oreal | DEODORANT AND / OR ANTI-TRANSPIRANT COMPOSITION BASED ON INTERFERENTIAL MICRIOPARTICLES; METHOD OF MAKE-UP AND TREATMENT OF PERSPIRATION AND / OR BODILY ODORS IN PARTICULAR AXILLAR |
| WO2009101593A2 (en) | 2008-02-15 | 2009-08-20 | The Procter & Gamble Company | Delivery particle |
| GB0803538D0 (en) | 2008-02-27 | 2008-04-02 | Dow Corning | Deposition of lipophilic active material in surfactant containing compositions |
| BRPI0909220A2 (en) | 2008-03-26 | 2015-08-25 | Procter & Gamble | Release particle |
| US20090258042A1 (en) | 2008-04-14 | 2009-10-15 | Theodore James Anastasiou | Encapsulated Active Materials Containing Adjunct Crosslinkers |
| US20090274906A1 (en) | 2008-05-01 | 2009-11-05 | Appleton Papers Inc. | Particle with low permeance wall |
| US8067089B2 (en) * | 2008-05-01 | 2011-11-29 | Appleton Papers Inc. | Cationic microcapsule particles |
| US8071214B2 (en) * | 2008-05-01 | 2011-12-06 | Appleton Papers Inc. | Particle with selected permeance wall |
| DE102008044700A1 (en) | 2008-08-15 | 2010-02-18 | Dr. Scheller Cosmetics Ag | Cosmetic composition, useful as skin care composition, comprises cosmetically effective- and/or active-substance, which is present partially in the form of nanocapsule in a nanoparticular carrier structure, and an excipient |
| ES2609092T3 (en) | 2008-10-27 | 2017-04-18 | Unilever N.V. | Antiperspirant compositions |
| ES2610627T3 (en) | 2008-10-27 | 2017-04-28 | Unilever N.V. | Antiperspirant or deodorant compositions |
| ES2618314T3 (en) | 2008-10-27 | 2017-06-21 | Unilever N.V. | Antiperspirant compositions |
| EP2349551B2 (en) | 2008-11-07 | 2023-07-26 | The Procter & Gamble Company | Benefit agent containing delivery particle |
| US20100190673A1 (en) | 2009-01-29 | 2010-07-29 | Johan Smets | Encapsulates |
| US20100190674A1 (en) | 2009-01-29 | 2010-07-29 | Johan Smets | Encapsulates |
| EP2221039B1 (en) | 2009-02-18 | 2017-11-22 | Unilever Plc, A Company Registered In England And Wales under company no. 41424 of Unilever House | Antiperspirant compositions |
| CL2012000290A1 (en) | 2009-08-06 | 2013-01-25 | Unilever Nv | An antiperspirant or deodorant composition comprising i) an antiperspirant or deodorant active, ii) a liquid carrier for the antiperspirant or deodorant active and iii) a fragrance; a product comprising the composition and a method of hiding body odor for a prolonged period of time. |
| JP5017338B2 (en) | 2009-09-07 | 2012-09-05 | キヤノン株式会社 | Manufacturing method of organic transistor |
| AR078889A1 (en) | 2009-11-06 | 2011-12-07 | Procter & Gamble | ENCAPSULATES AND DETERGENT COMPOSITIONS THAT UNDERSTAND THEM |
| US20110152147A1 (en) | 2009-12-18 | 2011-06-23 | Johan Smets | Encapsulates |
| US9993793B2 (en) | 2010-04-28 | 2018-06-12 | The Procter & Gamble Company | Delivery particles |
| JP5081953B2 (en) | 2010-04-28 | 2012-11-28 | 本田技研工業株式会社 | Head-up display device for vehicle |
| US20110268778A1 (en) | 2010-04-28 | 2011-11-03 | Jiten Odhavji Dihora | Delivery particles |
| US9186642B2 (en) | 2010-04-28 | 2015-11-17 | The Procter & Gamble Company | Delivery particle |
| US20110269657A1 (en) | 2010-04-28 | 2011-11-03 | Jiten Odhavji Dihora | Delivery particles |
| WO2012138710A2 (en) | 2011-04-07 | 2012-10-11 | The Procter & Gamble Company | Personal cleansing compositions with increased deposition of polyacrylate microcapsules |
| CN103458858B (en) | 2011-04-07 | 2016-04-27 | 宝洁公司 | There is the shampoo Compositions of the deposition of the polyacrylate microcapsule of enhancing |
| CN103458871B (en) | 2011-04-07 | 2015-05-13 | 宝洁公司 | Conditioner compositions with increased deposition of polyacrylate microcapsules |
| US20120322709A1 (en) | 2011-05-26 | 2012-12-20 | Jianjun Justin Li | Compositions comprising an efficient perfume bloom |
| CA2885381A1 (en) | 2012-09-20 | 2014-03-27 | Jiten Odhavji Dihora | Spray drying microcapsules |
| CA2881393A1 (en) | 2012-09-20 | 2014-03-27 | The Procter & Gamble Company | Anhydrous compositions having microcapsules and non-volatile oils |
| EP2897579B1 (en) | 2012-09-20 | 2018-02-21 | The Procter and Gamble Company | Compositions and articles having a parent fragrance and microcapsules encapsulating a non-parent fragrance |
-
2010
- 2010-05-11 US US12/777,314 patent/US9993793B2/en active Active
- 2010-05-12 RU RU2012144736/04A patent/RU2566753C2/en active
- 2010-05-12 MX MX2016014810A patent/MX358448B/en unknown
- 2010-05-12 JP JP2013506763A patent/JP2013531079A/en active Pending
- 2010-05-12 MX MX2012012549A patent/MX356208B/en active IP Right Grant
- 2010-05-12 EP EP20179227.2A patent/EP3733827A1/en not_active Withdrawn
- 2010-05-12 CN CN201310489226.8A patent/CN103589530B/en active Active
- 2010-05-12 EP EP13188005.6A patent/EP2687590A3/en not_active Withdrawn
- 2010-05-12 WO PCT/IB2010/052121 patent/WO2010079467A2/en active Application Filing
- 2010-05-12 EP EP10721567A patent/EP2563894A2/en not_active Withdrawn
- 2010-05-12 CA CA2800456A patent/CA2800456C/en active Active
- 2010-05-12 CN CN201080066442.6A patent/CN102858940B/en active Active
-
2013
- 2013-10-03 US US14/045,670 patent/US9221028B2/en active Active
- 2013-10-22 JP JP2013219552A patent/JP6046588B2/en active Active
-
2015
- 2015-04-24 JP JP2015089636A patent/JP2015180733A/en active Pending
-
2017
- 2017-05-31 JP JP2017108562A patent/JP2017149990A/en active Pending
Non-Patent Citations (3)
| Title |
|---|
| HUBER: "Capsular Adhesives", TAPPI, vol. 49, no. 5, May 1966 (1966-05-01), pages 41A - 44A |
| See also references of EP2563894A2 |
| ZHANG, Z.; SUN, G: "Mechanical Properties of Melamine-Formaldehyde microcapsules", J. MICROENCAPSULATION, vol. 18, no. 5, 2001, pages 593 - 602, XP008139505 |
Cited By (75)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9109068B2 (en) | 2005-07-21 | 2015-08-18 | Akzo Nobel N.V. | Hybrid copolymer compositions |
| US9321873B2 (en) | 2005-07-21 | 2016-04-26 | Akzo Nobel N.V. | Hybrid copolymer compositions for personal care applications |
| US8674021B2 (en) | 2006-07-21 | 2014-03-18 | Akzo Nobel N.V. | Sulfonated graft copolymers |
| EP3085759A2 (en) | 2010-06-22 | 2016-10-26 | The Procter and Gamble Company | Perfume systems |
| WO2011163337A1 (en) | 2010-06-22 | 2011-12-29 | The Procter & Gamble Company | Perfume systems |
| EP3533908A1 (en) | 2010-07-02 | 2019-09-04 | The Procter & Gamble Company | Nonwoven web comprising one or more active agents |
| WO2012003360A2 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Detergent product and method for making same |
| WO2012003316A1 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Process for making films from nonwoven webs |
| WO2012003367A2 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Method for delivering an active agent |
| WO2012003319A2 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Filaments comprising an active agent nonwoven webs and methods for making same |
| WO2012003351A2 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Web material and method for making same |
| WO2012003300A2 (en) | 2010-07-02 | 2012-01-05 | The Procter & Gamble Company | Filaments comprising a non-perfume active agent nonwoven webs and methods for making same |
| JP2014510140A (en) * | 2011-04-07 | 2014-04-24 | ザ プロクター アンド ギャンブル カンパニー | Conditioner composition with increased adhesion of polyacrylate microcapsules |
| EP2694016B1 (en) | 2011-04-07 | 2017-05-24 | The Procter and Gamble Company | Shampoo compositions with increased deposition of polyacrylate microcapsules |
| WO2012138710A2 (en) | 2011-04-07 | 2012-10-11 | The Procter & Gamble Company | Personal cleansing compositions with increased deposition of polyacrylate microcapsules |
| JP2014510141A (en) * | 2011-04-07 | 2014-04-24 | ザ プロクター アンド ギャンブル カンパニー | Shampoo compositions with increased adhesion of polyacrylate microcapsules |
| JP2014510142A (en) * | 2011-04-07 | 2014-04-24 | ザ プロクター アンド ギャンブル カンパニー | Personal cleansing composition with increased deposition of polyacrylate microcapsules |
| JP2016128499A (en) * | 2011-04-07 | 2016-07-14 | ザ プロクター アンド ギャンブル カンパニー | Personal cleansing compositions with increased deposition of polyacrylate microcapsules |
| WO2012138696A2 (en) | 2011-04-07 | 2012-10-11 | The Procter & Gamble Company | Shampoo compositions with increased deposition of polyacrylate microcapsules |
| WO2012177357A1 (en) | 2011-06-23 | 2012-12-27 | The Procter & Gamble Company | Perfume systems |
| WO2013002786A1 (en) | 2011-06-29 | 2013-01-03 | Solae | Baked food compositions comprising soy whey proteins that have been isolated from processing streams |
| US8679366B2 (en) | 2011-08-05 | 2014-03-25 | Ecolab Usa Inc. | Cleaning composition containing a polysaccharide graft polymer composition and methods of controlling hard water scale |
| US8853144B2 (en) | 2011-08-05 | 2014-10-07 | Ecolab Usa Inc. | Cleaning composition containing a polysaccharide graft polymer composition and methods of improving drainage |
| US8636918B2 (en) | 2011-08-05 | 2014-01-28 | Ecolab Usa Inc. | Cleaning composition containing a polysaccharide hybrid polymer composition and methods of controlling hard water scale |
| US8841246B2 (en) | 2011-08-05 | 2014-09-23 | Ecolab Usa Inc. | Cleaning composition containing a polysaccharide hybrid polymer composition and methods of improving drainage |
| US9051406B2 (en) | 2011-11-04 | 2015-06-09 | Akzo Nobel Chemicals International B.V. | Graft dendrite copolymers, and methods for producing the same |
| US9988526B2 (en) | 2011-11-04 | 2018-06-05 | Akzo Nobel Chemicals International B.V. | Hybrid dendrite copolymers, compositions thereof and methods for producing the same |
| EP3369845A1 (en) | 2012-01-04 | 2018-09-05 | The Procter & Gamble Company | Active containing fibrous structures with multiple regions having differing densities |
| FR2985273A1 (en) | 2012-01-04 | 2013-07-05 | Procter & Gamble | FIBROUS STRUCTURES CONTAINING ASSETS AND HAVING MULTIPLE REGIONS |
| EP3719192A1 (en) | 2012-01-04 | 2020-10-07 | The Procter & Gamble Company | Fibrous structures comprising particles and methods for making same |
| US8945314B2 (en) | 2012-07-30 | 2015-02-03 | Ecolab Usa Inc. | Biodegradable stability binding agent for a solid detergent |
| JP2016525928A (en) * | 2013-05-20 | 2016-09-01 | ザ プロクター アンド ギャンブル カンパニー | Inclusion body |
| WO2014189906A3 (en) * | 2013-05-20 | 2015-04-09 | The Procter & Gamble Company | Encapsulates |
| WO2015041791A1 (en) * | 2013-09-23 | 2015-03-26 | The Procter & Gamble Company | Particles |
| CN105555940A (en) * | 2013-09-23 | 2016-05-04 | 宝洁公司 | Particles |
| EP4253649A2 (en) | 2013-12-09 | 2023-10-04 | The Procter & Gamble Company | Fibrous structures including an active agent and having a graphic printed thereon |
| DE112014005598B4 (en) | 2013-12-09 | 2022-06-09 | The Procter & Gamble Company | Fibrous structures including an active substance and with graphics printed on it |
| EP3805350A1 (en) | 2013-12-09 | 2021-04-14 | The Procter & Gamble Company | Fibrous structures including an active agent and having a graphic printed thereon |
| WO2015088826A1 (en) | 2013-12-09 | 2015-06-18 | The Procter & Gamble Company | Fibrous structures including an active agent and having a graphic printed thereon |
| FR3014456A1 (en) | 2013-12-09 | 2015-06-12 | Procter & Gamble | |
| EP3572572A1 (en) | 2013-12-09 | 2019-11-27 | The Procter & Gamble Company | Fibrous structures including an active agent and having a graphic printed thereon |
| RU2721556C2 (en) * | 2013-12-23 | 2020-05-20 | Арланксео Сингапур Пте. Лтд. | Especially pure rubber |
| EP3087138A4 (en) * | 2013-12-23 | 2019-04-17 | Arlanxeo Singapore Pte. Ltd. | Rubbers with tunable levels of metal containing anti-agglomerants |
| EP3087139A4 (en) * | 2013-12-23 | 2017-12-06 | Arlanxeo Singapore Pte. Ltd. | Ultra pure rubber |
| US10053652B2 (en) | 2014-05-15 | 2018-08-21 | Ecolab Usa Inc. | Bio-based pot and pan pre-soak |
| WO2016090624A1 (en) * | 2014-12-12 | 2016-06-16 | The Procter & Gamble Company | Liquid cleaning composition |
| US10662398B2 (en) | 2014-12-17 | 2020-05-26 | The Procter & Gamble Company | Detergent composition |
| US10266796B2 (en) | 2014-12-17 | 2019-04-23 | The Procter & Gamble Company | Detergent composition |
| US11820960B2 (en) | 2015-06-30 | 2023-11-21 | The Procter & Gamble Company | Compositions containing multiple populations of microcapsules |
| US11773351B2 (en) | 2015-06-30 | 2023-10-03 | The Procter & Gamble Company | Compositions containing multiple populations of microcapsules |
| US11085008B2 (en) | 2015-06-30 | 2021-08-10 | The Procter & Gamble Company | Methods for making compositions containing multiple populations of microcapsules |
| US11065593B2 (en) | 2015-09-03 | 2021-07-20 | Tagra Biotechnologies Ltd. | Microcapsules encapsulating a reflective agent |
| WO2018140432A1 (en) | 2017-01-27 | 2018-08-02 | The Procter & Gamble Company | Active agent-containing articles that exhibit consumer acceptable article in-use properties |
| EP3991962A1 (en) | 2017-01-27 | 2022-05-04 | The Procter & Gamble Company | Active agent-containing articles that exhibit consumer acceptable article in-use properties |
| DE112018000558T5 (en) | 2017-01-27 | 2019-10-10 | The Procter & Gamble Company | Active substance-containing articles which have acceptable consumer properties acceptable to the consumer |
| WO2018140472A1 (en) | 2017-01-27 | 2018-08-02 | The Procter & Gamble Company | Active agent-containing articles that exhibit consumer acceptable article in-use properties |
| DE112018000568T5 (en) | 2017-01-27 | 2019-10-17 | The Procter & Gamble Company | Active substance-containing articles and product shipping arrangements for enclosing the same |
| EP4197598A1 (en) | 2017-01-27 | 2023-06-21 | The Procter & Gamble Company | Active agent-containing articles that exhibit consumer acceptable article in-use properties |
| DE112018000565T5 (en) | 2017-01-27 | 2019-10-24 | The Procter & Gamble Company | Active substance-containing articles which have acceptable consumer properties acceptable to the consumer |
| WO2018140454A1 (en) | 2017-01-27 | 2018-08-02 | The Procter & Gamble Company | Active agent-containing articles and product-shipping assemblies for containing the same |
| EP3915643A1 (en) | 2017-01-27 | 2021-12-01 | The Procter & Gamble Company | Active agent-containing articles that exhibit consumer acceptable article in-use properties |
| EP3881900A1 (en) | 2017-01-27 | 2021-09-22 | The Procter & Gamble Company | Active agent-containing articles that exhibit consumer acceptable article in-use properties |
| WO2018140431A1 (en) | 2017-01-27 | 2018-08-02 | The Procter & Gamble Company | Active agent-containing articles that exhibit consumer acceptable article in-use properties |
| DE112018000563T5 (en) | 2017-01-27 | 2019-10-24 | The Procter & Gamble Company | Active substance-containing articles which have acceptable consumer properties acceptable to the consumer |
| EP3375855B1 (en) | 2017-03-16 | 2021-04-21 | The Procter & Gamble Company | Fabric softener composition comprising encapsulated benefit agent |
| WO2018236700A1 (en) * | 2017-06-20 | 2018-12-27 | The Procter & Gamble Company | Multi composition systems comprising a bleaching agent and encapsulates |
| CN110741071A (en) * | 2017-06-20 | 2020-01-31 | 宝洁公司 | Multiple composition system comprising bleach and encapsulate |
| EP3740555B1 (en) | 2018-01-17 | 2021-10-13 | Unilever IP Holdings B.V. | Laundry detergent |
| WO2019141530A1 (en) * | 2018-01-17 | 2019-07-25 | Unilever Plc | Laundry detergent |
| CN113748196A (en) * | 2019-05-08 | 2021-12-03 | 宝洁公司 | Granule for washing and softening of full laundry |
| WO2020227737A1 (en) * | 2019-05-08 | 2020-11-12 | The Procter & Gamble Company | Particles for through the wash laundry softening |
| EP3736320A1 (en) * | 2019-05-08 | 2020-11-11 | The Procter & Gamble Company | Particles for through the wash laundry softening |
| WO2021113567A1 (en) | 2019-12-05 | 2021-06-10 | The Procter & Gamble Company | Cleaning composition |
| WO2021113568A1 (en) | 2019-12-05 | 2021-06-10 | The Procter & Gamble Company | Method of making a cleaning composition |
| WO2024012811A1 (en) * | 2022-07-14 | 2024-01-18 | Givaudan Sa | Improvements in or relating to organic compounds |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2015180733A (en) | 2015-10-15 |
| EP2563894A2 (en) | 2013-03-06 |
| US9993793B2 (en) | 2018-06-12 |
| RU2012144736A (en) | 2014-06-10 |
| JP2013531079A (en) | 2013-08-01 |
| CN103589530B (en) | 2016-08-24 |
| CN102858940A (en) | 2013-01-02 |
| US20110269658A1 (en) | 2011-11-03 |
| RU2566753C2 (en) | 2015-10-27 |
| CA2800456C (en) | 2017-03-07 |
| WO2010079467A3 (en) | 2011-05-05 |
| JP2017149990A (en) | 2017-08-31 |
| US9221028B2 (en) | 2015-12-29 |
| EP2687590A3 (en) | 2016-07-27 |
| EP2687590A2 (en) | 2014-01-22 |
| US20140227328A1 (en) | 2014-08-14 |
| CN102858940B (en) | 2017-05-03 |
| WO2010079467A4 (en) | 2011-06-30 |
| EP3733827A1 (en) | 2020-11-04 |
| MX2012012549A (en) | 2012-11-23 |
| JP2014051670A (en) | 2014-03-20 |
| MX358448B (en) | 2018-08-20 |
| CN103589530A (en) | 2014-02-19 |
| CA2800456A1 (en) | 2010-07-15 |
| JP6046588B2 (en) | 2016-12-21 |
| MX356208B (en) | 2018-05-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11096875B2 (en) | Delivery particle | |
| US9221028B2 (en) | Delivery particles | |
| CA2795616C (en) | Microcapsule particles | |
| US20180360706A1 (en) | Delivery Particles |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080066442.6 Country of ref document: CN |
|
| ENP | Entry into the national phase |
Ref document number: 2800456 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 9172/DELNP/2012 Country of ref document: IN |
|
| REEP | Request for entry into the european phase |
Ref document number: 2010721567 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010721567 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2012/012549 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 2013506763 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2012144736 Country of ref document: RU Kind code of ref document: A |