WO2007114338A1 - 酸分泌抑制薬 - Google Patents
酸分泌抑制薬 Download PDFInfo
- Publication number
- WO2007114338A1 WO2007114338A1 PCT/JP2007/057102 JP2007057102W WO2007114338A1 WO 2007114338 A1 WO2007114338 A1 WO 2007114338A1 JP 2007057102 W JP2007057102 W JP 2007057102W WO 2007114338 A1 WO2007114338 A1 WO 2007114338A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- ring
- atom
- substituted
- compound
- Prior art date
Links
- 230000009858 acid secretion Effects 0.000 title claims abstract description 24
- 239000003112 inhibitor Substances 0.000 title claims description 29
- 150000001875 compounds Chemical class 0.000 claims abstract description 270
- 125000000217 alkyl group Chemical group 0.000 claims abstract description 112
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 70
- 125000001424 substituent group Chemical group 0.000 claims abstract description 67
- 125000004433 nitrogen atom Chemical group N* 0.000 claims abstract description 57
- 125000004429 atom Chemical group 0.000 claims abstract description 56
- 125000005843 halogen group Chemical group 0.000 claims abstract description 56
- 150000003839 salts Chemical class 0.000 claims abstract description 53
- 229920006395 saturated elastomer Polymers 0.000 claims abstract description 53
- 229910052799 carbon Inorganic materials 0.000 claims abstract description 48
- 229910052717 sulfur Inorganic materials 0.000 claims abstract description 38
- 125000004434 sulfur atom Chemical group 0.000 claims abstract description 37
- 125000004430 oxygen atom Chemical group O* 0.000 claims abstract description 34
- 125000001072 heteroaryl group Chemical group 0.000 claims abstract description 33
- 125000003118 aryl group Chemical group 0.000 claims abstract description 31
- 125000005842 heteroatom Chemical group 0.000 claims abstract description 30
- 125000004093 cyano group Chemical group *C#N 0.000 claims abstract description 26
- 125000004043 oxo group Chemical group O=* 0.000 claims abstract description 24
- 229940002612 prodrug Drugs 0.000 claims abstract description 21
- 239000000651 prodrug Substances 0.000 claims abstract description 21
- 125000004432 carbon atom Chemical group C* 0.000 claims abstract description 19
- 125000004202 aminomethyl group Chemical group [H]N([H])C([H])([H])* 0.000 claims abstract description 16
- 229940126409 proton pump inhibitor Drugs 0.000 claims abstract description 15
- 239000000612 proton pump inhibitor Substances 0.000 claims abstract description 15
- -1 aminoamino group Chemical group 0.000 claims description 202
- 239000000126 substance Substances 0.000 claims description 110
- 208000021302 gastroesophageal reflux disease Diseases 0.000 claims description 43
- 229910052736 halogen Inorganic materials 0.000 claims description 39
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 38
- 150000002367 halogens Chemical class 0.000 claims description 37
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 30
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 28
- 208000008469 Peptic Ulcer Diseases 0.000 claims description 24
- 201000006549 dyspepsia Diseases 0.000 claims description 23
- 239000003814 drug Substances 0.000 claims description 21
- 208000011906 peptic ulcer disease Diseases 0.000 claims description 19
- 125000003107 substituted aryl group Chemical group 0.000 claims description 18
- 230000035882 stress Effects 0.000 claims description 17
- 230000002496 gastric effect Effects 0.000 claims description 16
- 239000003795 chemical substances by application Substances 0.000 claims description 15
- 208000000689 peptic esophagitis Diseases 0.000 claims description 15
- 208000007882 Gastritis Diseases 0.000 claims description 13
- 208000005718 Stomach Neoplasms Diseases 0.000 claims description 13
- 239000002253 acid Substances 0.000 claims description 13
- 125000003277 amino group Chemical group 0.000 claims description 13
- 208000000718 duodenal ulcer Diseases 0.000 claims description 13
- 206010017758 gastric cancer Diseases 0.000 claims description 13
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 13
- 125000000168 pyrrolyl group Chemical group 0.000 claims description 13
- 125000006413 ring segment Chemical group 0.000 claims description 13
- 201000011549 stomach cancer Diseases 0.000 claims description 13
- 206010020601 Hyperchlorhydria Diseases 0.000 claims description 12
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical group C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 claims description 12
- 201000008629 Zollinger-Ellison syndrome Diseases 0.000 claims description 12
- 201000000052 gastrinoma Diseases 0.000 claims description 12
- 125000003545 alkoxy group Chemical group 0.000 claims description 11
- 208000017215 gastric mucosa-associated lymphoid tissue lymphoma Diseases 0.000 claims description 11
- 206010017866 Gastritis haemorrhagic Diseases 0.000 claims description 10
- 206010042220 Stress ulcer Diseases 0.000 claims description 10
- 230000037328 acute stress Effects 0.000 claims description 10
- 125000006615 aromatic heterocyclic group Chemical group 0.000 claims description 10
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 10
- 230000002265 prevention Effects 0.000 claims description 10
- 125000000714 pyrimidinyl group Chemical group 0.000 claims description 10
- 206010046274 Upper gastrointestinal haemorrhage Diseases 0.000 claims description 9
- 125000002883 imidazolyl group Chemical group 0.000 claims description 9
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 claims description 8
- 125000000547 substituted alkyl group Chemical group 0.000 claims description 8
- 125000004390 alkyl sulfonyl group Chemical group 0.000 claims description 7
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical group [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 7
- 229910052760 oxygen Inorganic materials 0.000 claims description 7
- 239000001301 oxygen Chemical group 0.000 claims description 7
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 7
- 238000010992 reflux Methods 0.000 claims description 7
- 208000023514 Barrett esophagus Diseases 0.000 claims description 6
- 208000023665 Barrett oesophagus Diseases 0.000 claims description 6
- 125000003806 alkyl carbonyl amino group Chemical group 0.000 claims description 6
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 claims description 5
- 229910052700 potassium Inorganic materials 0.000 claims description 5
- 239000011591 potassium Substances 0.000 claims description 5
- 241000124008 Mammalia Species 0.000 claims description 4
- 125000004122 cyclic group Chemical group 0.000 claims description 4
- RNAMYOYQYRYFQY-UHFFFAOYSA-N 2-(4,4-difluoropiperidin-1-yl)-6-methoxy-n-(1-propan-2-ylpiperidin-4-yl)-7-(3-pyrrolidin-1-ylpropoxy)quinazolin-4-amine Chemical compound N1=C(N2CCC(F)(F)CC2)N=C2C=C(OCCCN3CCCC3)C(OC)=CC2=C1NC1CCN(C(C)C)CC1 RNAMYOYQYRYFQY-UHFFFAOYSA-N 0.000 claims description 2
- 230000001629 suppression Effects 0.000 claims description 2
- 210000003238 esophagus Anatomy 0.000 claims 1
- 230000000694 effects Effects 0.000 abstract description 16
- 230000002401 inhibitory effect Effects 0.000 abstract description 16
- 230000000767 anti-ulcer Effects 0.000 abstract 1
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 195
- 239000000243 solution Substances 0.000 description 110
- 238000006243 chemical reaction Methods 0.000 description 82
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 81
- 239000000203 mixture Substances 0.000 description 79
- 230000002829 reductive effect Effects 0.000 description 73
- 239000002904 solvent Substances 0.000 description 64
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 63
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical group C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 56
- 235000002639 sodium chloride Nutrition 0.000 description 56
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 50
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 49
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 49
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 49
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 47
- 229910052794 bromium Inorganic materials 0.000 description 47
- 229910052801 chlorine Inorganic materials 0.000 description 47
- 229910052731 fluorine Inorganic materials 0.000 description 47
- 229910052740 iodine Inorganic materials 0.000 description 47
- 239000000284 extract Substances 0.000 description 45
- 239000011630 iodine Substances 0.000 description 45
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 45
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 44
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 44
- 239000000460 chlorine Substances 0.000 description 44
- 239000011737 fluorine Substances 0.000 description 44
- 150000001721 carbon Chemical group 0.000 description 40
- 238000000034 method Methods 0.000 description 40
- 235000017557 sodium bicarbonate Nutrition 0.000 description 40
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 40
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 39
- 238000005160 1H NMR spectroscopy Methods 0.000 description 37
- 239000011541 reaction mixture Substances 0.000 description 36
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 33
- 239000013078 crystal Substances 0.000 description 31
- 238000010898 silica gel chromatography Methods 0.000 description 31
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 30
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical group C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 29
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 27
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 27
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 27
- RAXXELZNTBOGNW-UHFFFAOYSA-N 1H-imidazole Chemical compound C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 25
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 25
- 239000002585 base Substances 0.000 description 24
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 24
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 24
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 23
- OAYLNYINCPYISS-UHFFFAOYSA-N ethyl acetate;hexane Chemical compound CCCCCC.CCOC(C)=O OAYLNYINCPYISS-UHFFFAOYSA-N 0.000 description 22
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 21
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 20
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 20
- 238000003756 stirring Methods 0.000 description 19
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 18
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 17
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 17
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 16
- 239000003921 oil Substances 0.000 description 16
- 235000019198 oils Nutrition 0.000 description 16
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 16
- OISVCGZHLKNMSJ-UHFFFAOYSA-N 2,6-dimethylpyridine Chemical compound CC1=CC=CC(C)=N1 OISVCGZHLKNMSJ-UHFFFAOYSA-N 0.000 description 15
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 15
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 15
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 15
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 15
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 15
- 238000002360 preparation method Methods 0.000 description 15
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 14
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 14
- 208000025865 Ulcer Diseases 0.000 description 14
- 239000012312 sodium hydride Substances 0.000 description 14
- 229910000104 sodium hydride Inorganic materials 0.000 description 14
- 231100000397 ulcer Toxicity 0.000 description 14
- 229940079593 drug Drugs 0.000 description 13
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 12
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 12
- 125000002088 tosyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C([H])([H])[H])S(*)(=O)=O 0.000 description 12
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 11
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 11
- 239000007787 solid Substances 0.000 description 11
- JLTDJTHDQAWBAV-UHFFFAOYSA-N N,N-dimethylaniline Chemical compound CN(C)C1=CC=CC=C1 JLTDJTHDQAWBAV-UHFFFAOYSA-N 0.000 description 10
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 10
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 10
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 10
- 229910000027 potassium carbonate Inorganic materials 0.000 description 10
- 235000011181 potassium carbonates Nutrition 0.000 description 10
- 238000005481 NMR spectroscopy Methods 0.000 description 9
- 150000004982 aromatic amines Chemical class 0.000 description 9
- 239000010410 layer Substances 0.000 description 9
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 9
- 125000003356 phenylsulfanyl group Chemical group [*]SC1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 9
- 239000012279 sodium borohydride Substances 0.000 description 9
- 229910000033 sodium borohydride Inorganic materials 0.000 description 9
- 239000003826 tablet Substances 0.000 description 9
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 8
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 8
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 8
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical group C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 8
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 8
- 239000003153 chemical reaction reagent Substances 0.000 description 8
- 239000000470 constituent Substances 0.000 description 8
- 238000001816 cooling Methods 0.000 description 8
- 150000002170 ethers Chemical class 0.000 description 8
- NUJOXMJBOLGQSY-UHFFFAOYSA-N manganese dioxide Chemical compound O=[Mn]=O NUJOXMJBOLGQSY-UHFFFAOYSA-N 0.000 description 8
- 238000004519 manufacturing process Methods 0.000 description 8
- NQMRYBIKMRVZLB-UHFFFAOYSA-N methylamine hydrochloride Chemical compound [Cl-].[NH3+]C NQMRYBIKMRVZLB-UHFFFAOYSA-N 0.000 description 8
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 8
- 239000012046 mixed solvent Substances 0.000 description 8
- 238000007254 oxidation reaction Methods 0.000 description 8
- 239000000843 powder Substances 0.000 description 8
- 239000000047 product Substances 0.000 description 8
- 230000035484 reaction time Effects 0.000 description 8
- 229910000029 sodium carbonate Inorganic materials 0.000 description 8
- 102000004190 Enzymes Human genes 0.000 description 7
- 108090000790 Enzymes Proteins 0.000 description 7
- 102100032709 Potassium-transporting ATPase alpha chain 2 Human genes 0.000 description 7
- 108010083204 Proton Pumps Proteins 0.000 description 7
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 7
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 description 7
- 230000002378 acidificating effect Effects 0.000 description 7
- 150000001408 amides Chemical class 0.000 description 7
- 229920002678 cellulose Polymers 0.000 description 7
- 239000001913 cellulose Substances 0.000 description 7
- 235000010980 cellulose Nutrition 0.000 description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 7
- 229940088598 enzyme Drugs 0.000 description 7
- 239000000706 filtrate Substances 0.000 description 7
- 125000001153 fluoro group Chemical group F* 0.000 description 7
- 229910017053 inorganic salt Inorganic materials 0.000 description 7
- 239000011777 magnesium Chemical class 0.000 description 7
- 229910052749 magnesium Inorganic materials 0.000 description 7
- 125000003170 phenylsulfonyl group Chemical group C1(=CC=CC=C1)S(=O)(=O)* 0.000 description 7
- 230000002980 postoperative effect Effects 0.000 description 7
- PAMIQIKDUOTOBW-UHFFFAOYSA-N 1-methylpiperidine Chemical compound CN1CCCCC1 PAMIQIKDUOTOBW-UHFFFAOYSA-N 0.000 description 6
- HIXDQWDOVZUNNA-UHFFFAOYSA-N 2-(3,4-dimethoxyphenyl)-5-hydroxy-7-methoxychromen-4-one Chemical compound C=1C(OC)=CC(O)=C(C(C=2)=O)C=1OC=2C1=CC=C(OC)C(OC)=C1 HIXDQWDOVZUNNA-UHFFFAOYSA-N 0.000 description 6
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 6
- NTYJJOPFIAHURM-UHFFFAOYSA-N Histamine Chemical compound NCCC1=CN=CN1 NTYJJOPFIAHURM-UHFFFAOYSA-N 0.000 description 6
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical class [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- 229930006000 Sucrose Natural products 0.000 description 6
- 230000009471 action Effects 0.000 description 6
- 150000001447 alkali salts Chemical class 0.000 description 6
- 239000003242 anti bacterial agent Substances 0.000 description 6
- 229940088710 antibiotic agent Drugs 0.000 description 6
- SIPUZPBQZHNSDW-UHFFFAOYSA-N bis(2-methylpropyl)aluminum Chemical compound CC(C)C[Al]CC(C)C SIPUZPBQZHNSDW-UHFFFAOYSA-N 0.000 description 6
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 6
- 229910000024 caesium carbonate Inorganic materials 0.000 description 6
- 239000003638 chemical reducing agent Substances 0.000 description 6
- 239000012230 colorless oil Substances 0.000 description 6
- 150000003983 crown ethers Chemical class 0.000 description 6
- HPYNZHMRTTWQTB-UHFFFAOYSA-N dimethylpyridine Natural products CC1=CC=CN=C1C HPYNZHMRTTWQTB-UHFFFAOYSA-N 0.000 description 6
- 230000032050 esterification Effects 0.000 description 6
- 238000005886 esterification reaction Methods 0.000 description 6
- 125000006125 ethylsulfonyl group Chemical group 0.000 description 6
- 230000002140 halogenating effect Effects 0.000 description 6
- 125000000623 heterocyclic group Chemical group 0.000 description 6
- 229930195733 hydrocarbon Natural products 0.000 description 6
- 150000002430 hydrocarbons Chemical class 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 229910052751 metal Inorganic materials 0.000 description 6
- 239000002184 metal Substances 0.000 description 6
- 239000000041 non-steroidal anti-inflammatory agent Substances 0.000 description 6
- 239000008194 pharmaceutical composition Substances 0.000 description 6
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 6
- 239000011734 sodium Substances 0.000 description 6
- 229910052708 sodium Inorganic materials 0.000 description 6
- 239000005720 sucrose Substances 0.000 description 6
- 239000011593 sulfur Chemical group 0.000 description 6
- AKEJUJNQAAGONA-UHFFFAOYSA-N sulfur trioxide Chemical compound O=S(=O)=O AKEJUJNQAAGONA-UHFFFAOYSA-N 0.000 description 6
- 150000003512 tertiary amines Chemical class 0.000 description 6
- RMVRSNDYEFQCLF-UHFFFAOYSA-N thiophenol Chemical compound SC1=CC=CC=C1 RMVRSNDYEFQCLF-UHFFFAOYSA-N 0.000 description 6
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 6
- 229920003169 water-soluble polymer Polymers 0.000 description 6
- AVFZOVWCLRSYKC-UHFFFAOYSA-N 1-methylpyrrolidine Chemical compound CN1CCCC1 AVFZOVWCLRSYKC-UHFFFAOYSA-N 0.000 description 5
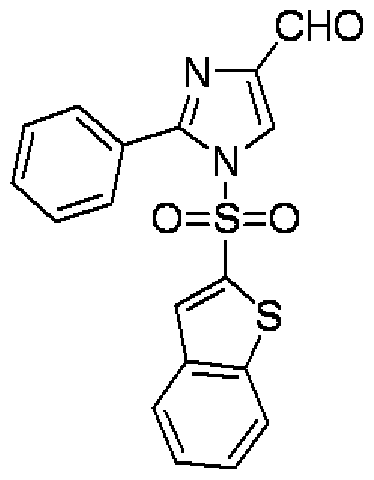
- ZQEXIXXJFSQPNA-UHFFFAOYSA-N 1h-imidazole-5-carbaldehyde Chemical compound O=CC1=CNC=N1 ZQEXIXXJFSQPNA-UHFFFAOYSA-N 0.000 description 5
- 125000003349 3-pyridyl group Chemical group N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 description 5
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 5
- 125000000339 4-pyridyl group Chemical group N1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 5
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 5
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 5
- SVYKKECYCPFKGB-UHFFFAOYSA-N N,N-dimethylcyclohexylamine Chemical compound CN(C)C1CCCCC1 SVYKKECYCPFKGB-UHFFFAOYSA-N 0.000 description 5
- AHVYPIQETPWLSZ-UHFFFAOYSA-N N-methyl-pyrrolidine Natural products CN1CC=CC1 AHVYPIQETPWLSZ-UHFFFAOYSA-N 0.000 description 5
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 5
- 239000002202 Polyethylene glycol Substances 0.000 description 5
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 5
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical group [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 5
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 5
- 239000005557 antagonist Substances 0.000 description 5
- 239000007864 aqueous solution Substances 0.000 description 5
- 239000011575 calcium Substances 0.000 description 5
- 229910052791 calcium Inorganic materials 0.000 description 5
- 239000002775 capsule Substances 0.000 description 5
- 125000001309 chloro group Chemical group Cl* 0.000 description 5
- 210000004211 gastric acid Anatomy 0.000 description 5
- 239000008187 granular material Substances 0.000 description 5
- 229910052739 hydrogen Inorganic materials 0.000 description 5
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 5
- 235000010355 mannitol Nutrition 0.000 description 5
- CEAJFNBWKBTRQE-UHFFFAOYSA-N methanamine;methanol Chemical compound NC.OC CEAJFNBWKBTRQE-UHFFFAOYSA-N 0.000 description 5
- WCPAKWJPBJAGKN-UHFFFAOYSA-N oxadiazole Chemical group C1=CON=N1 WCPAKWJPBJAGKN-UHFFFAOYSA-N 0.000 description 5
- 229920001223 polyethylene glycol Polymers 0.000 description 5
- 239000012321 sodium triacetoxyborohydride Substances 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 5
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 description 4
- 206010063655 Erosive oesophagitis Diseases 0.000 description 4
- ULGZDMOVFRHVEP-RWJQBGPGSA-N Erythromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@@](C)(O)[C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 ULGZDMOVFRHVEP-RWJQBGPGSA-N 0.000 description 4
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 4
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 4
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 4
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 4
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 4
- JRNVZBWKYDBUCA-UHFFFAOYSA-N N-chlorosuccinimide Chemical compound ClN1C(=O)CCC1=O JRNVZBWKYDBUCA-UHFFFAOYSA-N 0.000 description 4
- LQZMLBORDGWNPD-UHFFFAOYSA-N N-iodosuccinimide Chemical compound IN1C(=O)CCC1=O LQZMLBORDGWNPD-UHFFFAOYSA-N 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 4
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 4
- 229920002472 Starch Polymers 0.000 description 4
- 208000007107 Stomach Ulcer Diseases 0.000 description 4
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 4
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 4
- 108030003004 Triphosphatases Proteins 0.000 description 4
- 125000003342 alkenyl group Chemical group 0.000 description 4
- 229940069428 antacid Drugs 0.000 description 4
- 239000003159 antacid agent Substances 0.000 description 4
- 125000005605 benzo group Chemical group 0.000 description 4
- 125000004618 benzofuryl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 4
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 description 4
- 239000011230 binding agent Substances 0.000 description 4
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 4
- 125000000259 cinnolinyl group Chemical group N1=NC(=CC2=CC=CC=C12)* 0.000 description 4
- 238000002425 crystallisation Methods 0.000 description 4
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 4
- 201000010099 disease Diseases 0.000 description 4
- 239000007884 disintegrant Substances 0.000 description 4
- 239000002702 enteric coating Substances 0.000 description 4
- 238000009505 enteric coating Methods 0.000 description 4
- MVEAAGBEUOMFRX-UHFFFAOYSA-N ethyl acetate;hydrochloride Chemical compound Cl.CCOC(C)=O MVEAAGBEUOMFRX-UHFFFAOYSA-N 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 229940093915 gynecological organic acid Drugs 0.000 description 4
- IKDUDTNKRLTJSI-UHFFFAOYSA-N hydrazine hydrate Chemical compound O.NN IKDUDTNKRLTJSI-UHFFFAOYSA-N 0.000 description 4
- 239000001257 hydrogen Substances 0.000 description 4
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- 150000007529 inorganic bases Chemical class 0.000 description 4
- 239000000543 intermediate Substances 0.000 description 4
- 125000000904 isoindolyl group Chemical group C=1(NC=C2C=CC=CC12)* 0.000 description 4
- 210000000111 lower esophageal sphincter Anatomy 0.000 description 4
- 239000000314 lubricant Substances 0.000 description 4
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 4
- 239000001095 magnesium carbonate Substances 0.000 description 4
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 4
- 230000002503 metabolic effect Effects 0.000 description 4
- 150000007522 mineralic acids Chemical class 0.000 description 4
- 239000002808 molecular sieve Substances 0.000 description 4
- 150000007524 organic acids Chemical class 0.000 description 4
- 235000005985 organic acids Nutrition 0.000 description 4
- 125000002971 oxazolyl group Chemical group 0.000 description 4
- 239000007800 oxidant agent Substances 0.000 description 4
- 239000000546 pharmaceutical excipient Substances 0.000 description 4
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical compound OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 4
- 235000013809 polyvinylpolypyrrolidone Nutrition 0.000 description 4
- 229920000523 polyvinylpolypyrrolidone Polymers 0.000 description 4
- 229960003975 potassium Drugs 0.000 description 4
- 229940126535 potassium competitive acid blocker Drugs 0.000 description 4
- RPDAUEIUDPHABB-UHFFFAOYSA-N potassium ethoxide Chemical compound [K+].CC[O-] RPDAUEIUDPHABB-UHFFFAOYSA-N 0.000 description 4
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 4
- 125000004309 pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 4
- 230000028327 secretion Effects 0.000 description 4
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 4
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 4
- 239000008107 starch Substances 0.000 description 4
- 235000019698 starch Nutrition 0.000 description 4
- 210000002784 stomach Anatomy 0.000 description 4
- 239000000829 suppository Substances 0.000 description 4
- 239000000725 suspension Substances 0.000 description 4
- 238000013268 sustained release Methods 0.000 description 4
- 239000012730 sustained-release form Substances 0.000 description 4
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 4
- OXRSCXFOUBLJAR-TYYBGVCCSA-N (e)-but-2-enedioic acid;methanamine Chemical compound NC.OC(=O)\C=C\C(O)=O OXRSCXFOUBLJAR-TYYBGVCCSA-N 0.000 description 3
- UGUHFDPGDQDVGX-UHFFFAOYSA-N 1,2,3-thiadiazole Chemical group C1=CSN=N1 UGUHFDPGDQDVGX-UHFFFAOYSA-N 0.000 description 3
- YGTAZGSLCXNBQL-UHFFFAOYSA-N 1,2,4-thiadiazole Chemical group C=1N=CSN=1 YGTAZGSLCXNBQL-UHFFFAOYSA-N 0.000 description 3
- MBIZXFATKUQOOA-UHFFFAOYSA-N 1,3,4-thiadiazole Chemical group C1=NN=CS1 MBIZXFATKUQOOA-UHFFFAOYSA-N 0.000 description 3
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 3
- 125000002941 2-furyl group Chemical group O1C([*])=C([H])C([H])=C1[H] 0.000 description 3
- YNJSNEKCXVFDKW-UHFFFAOYSA-N 3-(5-amino-1h-indol-3-yl)-2-azaniumylpropanoate Chemical compound C1=C(N)C=C2C(CC(N)C(O)=O)=CNC2=C1 YNJSNEKCXVFDKW-UHFFFAOYSA-N 0.000 description 3
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 3
- 125000003682 3-furyl group Chemical group O1C([H])=C([*])C([H])=C1[H] 0.000 description 3
- 125000004207 3-methoxyphenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(OC([H])([H])[H])=C1[H] 0.000 description 3
- YYROPELSRYBVMQ-UHFFFAOYSA-N 4-toluenesulfonyl chloride Chemical compound CC1=CC=C(S(Cl)(=O)=O)C=C1 YYROPELSRYBVMQ-UHFFFAOYSA-N 0.000 description 3
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- 206010002243 Anastomotic ulcer Diseases 0.000 description 3
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- 108091006146 Channels Proteins 0.000 description 3
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 3
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- 206010030216 Oesophagitis Diseases 0.000 description 3
- 229930182555 Penicillin Natural products 0.000 description 3
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 3
- LXKFVPKKRVKNIJ-MCDZGGTQSA-N [K].C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O Chemical compound [K].C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O LXKFVPKKRVKNIJ-MCDZGGTQSA-N 0.000 description 3
- 239000004480 active ingredient Substances 0.000 description 3
- 239000013543 active substance Substances 0.000 description 3
- 238000005917 acylation reaction Methods 0.000 description 3
- 239000000654 additive Substances 0.000 description 3
- 125000004414 alkyl thio group Chemical group 0.000 description 3
- 238000005804 alkylation reaction Methods 0.000 description 3
- 235000001014 amino acid Nutrition 0.000 description 3
- 150000001413 amino acids Chemical class 0.000 description 3
- 230000003042 antagnostic effect Effects 0.000 description 3
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- 239000003086 colorant Substances 0.000 description 3
- 230000002860 competitive effect Effects 0.000 description 3
- 230000008025 crystallization Effects 0.000 description 3
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 3
- 239000003405 delayed action preparation Substances 0.000 description 3
- 208000035475 disorder Diseases 0.000 description 3
- 208000006881 esophagitis Diseases 0.000 description 3
- UREBWPXBXRYXRJ-UHFFFAOYSA-N ethyl acetate;methanol Chemical compound OC.CCOC(C)=O UREBWPXBXRYXRJ-UHFFFAOYSA-N 0.000 description 3
- 239000004088 foaming agent Substances 0.000 description 3
- 239000003205 fragrance Substances 0.000 description 3
- 125000002541 furyl group Chemical group 0.000 description 3
- 229960001340 histamine Drugs 0.000 description 3
- 238000005984 hydrogenation reaction Methods 0.000 description 3
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 3
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 3
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 3
- 230000006872 improvement Effects 0.000 description 3
- 125000001041 indolyl group Chemical group 0.000 description 3
- 230000005764 inhibitory process Effects 0.000 description 3
- 231100000053 low toxicity Toxicity 0.000 description 3
- FDZZZRQASAIRJF-UHFFFAOYSA-M malachite green Chemical compound [Cl-].C1=CC(N(C)C)=CC=C1C(C=1C=CC=CC=1)=C1C=CC(=[N+](C)C)C=C1 FDZZZRQASAIRJF-UHFFFAOYSA-M 0.000 description 3
- 229940107698 malachite green Drugs 0.000 description 3
- 230000003287 optical effect Effects 0.000 description 3
- PFPYHYZFFJJQFD-UHFFFAOYSA-N oxalic anhydride Chemical compound O=C1OC1=O PFPYHYZFFJJQFD-UHFFFAOYSA-N 0.000 description 3
- 230000003647 oxidation Effects 0.000 description 3
- 229940049954 penicillin Drugs 0.000 description 3
- 125000006239 protecting group Chemical group 0.000 description 3
- 125000001042 pteridinyl group Chemical group N1=C(N=CC2=NC=CN=C12)* 0.000 description 3
- 125000003373 pyrazinyl group Chemical group 0.000 description 3
- 125000002206 pyridazin-3-yl group Chemical group [H]C1=C([H])C([H])=C(*)N=N1 0.000 description 3
- 125000004940 pyridazin-4-yl group Chemical group N1=NC=C(C=C1)* 0.000 description 3
- 125000005493 quinolyl group Chemical group 0.000 description 3
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 3
- 229940044551 receptor antagonist Drugs 0.000 description 3
- 239000002464 receptor antagonist Substances 0.000 description 3
- 238000006722 reduction reaction Methods 0.000 description 3
- 239000000932 sedative agent Substances 0.000 description 3
- 159000000000 sodium salts Chemical class 0.000 description 3
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 3
- 235000019345 sodium thiosulphate Nutrition 0.000 description 3
- 239000007901 soft capsule Substances 0.000 description 3
- 239000011975 tartaric acid Substances 0.000 description 3
- 235000002906 tartaric acid Nutrition 0.000 description 3
- 150000003536 tetrazoles Chemical group 0.000 description 3
- 229940124597 therapeutic agent Drugs 0.000 description 3
- 125000000335 thiazolyl group Chemical group 0.000 description 3
- 125000003396 thiol group Chemical class [H]S* 0.000 description 3
- DSNBHJFQCNUKMA-SCKDECHMSA-N thromboxane A2 Chemical compound OC(=O)CCC\C=C/C[C@@H]1[C@@H](/C=C/[C@@H](O)CCCCC)O[C@@H]2O[C@H]1C2 DSNBHJFQCNUKMA-SCKDECHMSA-N 0.000 description 3
- JWZZKOKVBUJMES-UHFFFAOYSA-N (+-)-Isoprenaline Chemical compound CC(C)NCC(O)C1=CC=C(O)C(O)=C1 JWZZKOKVBUJMES-UHFFFAOYSA-N 0.000 description 2
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 2
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical group C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 2
- 125000001399 1,2,3-triazolyl group Chemical group N1N=NC(=C1)* 0.000 description 2
- BBVIDBNAYOIXOE-UHFFFAOYSA-N 1,2,4-oxadiazole Chemical group C=1N=CON=1 BBVIDBNAYOIXOE-UHFFFAOYSA-N 0.000 description 2
- FYADHXFMURLYQI-UHFFFAOYSA-N 1,2,4-triazine Chemical group C1=CN=NC=N1 FYADHXFMURLYQI-UHFFFAOYSA-N 0.000 description 2
- FKASFBLJDCHBNZ-UHFFFAOYSA-N 1,3,4-oxadiazole Chemical group C1=NN=CO1 FKASFBLJDCHBNZ-UHFFFAOYSA-N 0.000 description 2
- JIHQDMXYYFUGFV-UHFFFAOYSA-N 1,3,5-triazine Chemical group C1=NC=NC=N1 JIHQDMXYYFUGFV-UHFFFAOYSA-N 0.000 description 2
- YNGDWRXWKFWCJY-UHFFFAOYSA-N 1,4-Dihydropyridine Chemical group C1C=CNC=C1 YNGDWRXWKFWCJY-UHFFFAOYSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- GPWNWKWQOLEVEQ-UHFFFAOYSA-N 2,4-diaminopyrimidine-5-carbaldehyde Chemical compound NC1=NC=C(C=O)C(N)=N1 GPWNWKWQOLEVEQ-UHFFFAOYSA-N 0.000 description 2
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 2
- 125000001216 2-naphthoyl group Chemical group C1=C(C=CC2=CC=CC=C12)C(=O)* 0.000 description 2
- 125000001622 2-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C(*)C([H])=C([H])C2=C1[H] 0.000 description 2
- ZCUJYXPAKHMBAZ-UHFFFAOYSA-N 2-phenyl-1h-imidazole Chemical compound C1=CNC(C=2C=CC=CC=2)=N1 ZCUJYXPAKHMBAZ-UHFFFAOYSA-N 0.000 description 2
- LFKJFIOTRHYONM-UHFFFAOYSA-N 2-phenyl-1h-imidazole-5-carbaldehyde Chemical compound N1C(C=O)=CN=C1C1=CC=CC=C1 LFKJFIOTRHYONM-UHFFFAOYSA-N 0.000 description 2
- WRMNZCZEMHIOCP-UHFFFAOYSA-N 2-phenylethanol Chemical compound OCCC1=CC=CC=C1 WRMNZCZEMHIOCP-UHFFFAOYSA-N 0.000 description 2
- FPLYMSUAUOSKLM-UHFFFAOYSA-N 2-phenylsulfanyl-1h-imidazole Chemical compound C=1C=CC=CC=1SC1=NC=CN1 FPLYMSUAUOSKLM-UHFFFAOYSA-N 0.000 description 2
- 125000000389 2-pyrrolyl group Chemical group [H]N1C([*])=C([H])C([H])=C1[H] 0.000 description 2
- KGWNRZLPXLBMPS-UHFFFAOYSA-N 2h-1,3-oxazine Chemical group C1OC=CC=N1 KGWNRZLPXLBMPS-UHFFFAOYSA-N 0.000 description 2
- NTYABNDBNKVWOO-UHFFFAOYSA-N 2h-1,3-thiazine Chemical group C1SC=CC=N1 NTYABNDBNKVWOO-UHFFFAOYSA-N 0.000 description 2
- YHWMFDLNZGIJSD-UHFFFAOYSA-N 2h-1,4-oxazine Chemical group C1OC=CN=C1 YHWMFDLNZGIJSD-UHFFFAOYSA-N 0.000 description 2
- BCHZICNRHXRCHY-UHFFFAOYSA-N 2h-oxazine Chemical group N1OC=CC=C1 BCHZICNRHXRCHY-UHFFFAOYSA-N 0.000 description 2
- AGIJRRREJXSQJR-UHFFFAOYSA-N 2h-thiazine Chemical group N1SC=CC=C1 AGIJRRREJXSQJR-UHFFFAOYSA-N 0.000 description 2
- QMDFJHAAWUGVKQ-UHFFFAOYSA-N 2h-thiopyran Chemical group C1SC=CC=C1 QMDFJHAAWUGVKQ-UHFFFAOYSA-N 0.000 description 2
- FTAHXMZRJCZXDL-UHFFFAOYSA-N 3-piperideine Chemical group C1CC=CCN1 FTAHXMZRJCZXDL-UHFFFAOYSA-N 0.000 description 2
- 125000001397 3-pyrrolyl group Chemical group [H]N1C([H])=C([*])C([H])=C1[H] 0.000 description 2
- YPELFRMCRYSPKZ-UHFFFAOYSA-N 4-amino-5-chloro-2-ethoxy-N-({4-[(4-fluorophenyl)methyl]morpholin-2-yl}methyl)benzamide Chemical compound CCOC1=CC(N)=C(Cl)C=C1C(=O)NCC1OCCN(CC=2C=CC(F)=CC=2)C1 YPELFRMCRYSPKZ-UHFFFAOYSA-N 0.000 description 2
- KDDQRKBRJSGMQE-UHFFFAOYSA-N 4-thiazolyl Chemical compound [C]1=CSC=N1 KDDQRKBRJSGMQE-UHFFFAOYSA-N 0.000 description 2
- NSPMIYGKQJPBQR-UHFFFAOYSA-N 4H-1,2,4-triazole Chemical group C=1N=CNN=1 NSPMIYGKQJPBQR-UHFFFAOYSA-N 0.000 description 2
- SUBDBMMJDZJVOS-UHFFFAOYSA-N 5-methoxy-2-{[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]sulfinyl}-1H-benzimidazole Chemical compound N=1C2=CC(OC)=CC=C2NC=1S(=O)CC1=NC=C(C)C(OC)=C1C SUBDBMMJDZJVOS-UHFFFAOYSA-N 0.000 description 2
- CWDWFSXUQODZGW-UHFFFAOYSA-N 5-thiazolyl Chemical group [C]1=CN=CS1 CWDWFSXUQODZGW-UHFFFAOYSA-N 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 2
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- KPYSYYIEGFHWSV-UHFFFAOYSA-N Baclofen Chemical compound OC(=O)CC(CN)C1=CC=C(Cl)C=C1 KPYSYYIEGFHWSV-UHFFFAOYSA-N 0.000 description 2
- ZYUZLEUJKZZXNN-UHFFFAOYSA-N C1=CC(CC(N)C(O)=O)=CC=C1OS(=O)(=O)C1=CC=C(C=CC=C2)C2=C1 Chemical group C1=CC(CC(N)C(O)=O)=CC=C1OS(=O)(=O)C1=CC=C(C=CC=C2)C2=C1 ZYUZLEUJKZZXNN-UHFFFAOYSA-N 0.000 description 2
- 229920000623 Cellulose acetate phthalate Polymers 0.000 description 2
- 229920002261 Corn starch Polymers 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- 206010013710 Drug interaction Diseases 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 108010002352 Interleukin-1 Proteins 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 206010025323 Lymphomas Diseases 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- 241001024304 Mino Species 0.000 description 2
- HSHXDCVZWHOWCS-UHFFFAOYSA-N N'-hexadecylthiophene-2-carbohydrazide Chemical compound CCCCCCCCCCCCCCCCNNC(=O)c1cccs1 HSHXDCVZWHOWCS-UHFFFAOYSA-N 0.000 description 2
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 2
- LFTLOKWAGJYHHR-UHFFFAOYSA-N N-methylmorpholine N-oxide Chemical compound CN1(=O)CCOCC1 LFTLOKWAGJYHHR-UHFFFAOYSA-N 0.000 description 2
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 2
- 241000283973 Oryctolagus cuniculus Species 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- NPYPAHLBTDXSSS-UHFFFAOYSA-N Potassium ion Chemical compound [K+] NPYPAHLBTDXSSS-UHFFFAOYSA-N 0.000 description 2
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 2
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 2
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 2
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 2
- 239000000556 agonist Substances 0.000 description 2
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 2
- 229910052783 alkali metal Inorganic materials 0.000 description 2
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 2
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 2
- 125000003282 alkyl amino group Chemical group 0.000 description 2
- 125000004448 alkyl carbonyl group Chemical group 0.000 description 2
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 2
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- 150000003863 ammonium salts Chemical class 0.000 description 2
- 229960003022 amoxicillin Drugs 0.000 description 2
- LSQZJLSUYDQPKJ-NJBDSQKTSA-N amoxicillin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@H]3SC([C@@H](N3C2=O)C(O)=O)(C)C)=CC=C(O)C=C1 LSQZJLSUYDQPKJ-NJBDSQKTSA-N 0.000 description 2
- 230000001458 anti-acid effect Effects 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 2
- 125000003710 aryl alkyl group Chemical group 0.000 description 2
- 125000004658 aryl carbonyl amino group Chemical group 0.000 description 2
- 125000005129 aryl carbonyl group Chemical group 0.000 description 2
- 125000004391 aryl sulfonyl group Chemical group 0.000 description 2
- 125000005110 aryl thio group Chemical group 0.000 description 2
- 125000004104 aryloxy group Chemical group 0.000 description 2
- 229960000794 baclofen Drugs 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 229960004916 benidipine Drugs 0.000 description 2
- QZVNQOLPLYWLHQ-ZEQKJWHPSA-N benidipine Chemical compound C1([C@H]2C(=C(C)NC(C)=C2C(=O)OC)C(=O)O[C@H]2CN(CC=3C=CC=CC=3)CCC2)=CC=CC([N+]([O-])=O)=C1 QZVNQOLPLYWLHQ-ZEQKJWHPSA-N 0.000 description 2
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzaldehyde Chemical compound O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 2
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 2
- SESFRYSPDFLNCH-UHFFFAOYSA-N benzyl benzoate Chemical compound C=1C=CC=CC=1C(=O)OCC1=CC=CC=C1 SESFRYSPDFLNCH-UHFFFAOYSA-N 0.000 description 2
- 125000000440 benzylamino group Chemical group [H]N(*)C([H])([H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 2
- 230000003115 biocidal effect Effects 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 159000000007 calcium salts Chemical class 0.000 description 2
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 description 2
- 229940081734 cellulose acetate phthalate Drugs 0.000 description 2
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 2
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 2
- 238000004587 chromatography analysis Methods 0.000 description 2
- KRVSOGSZCMJSLX-UHFFFAOYSA-L chromic acid Substances O[Cr](O)(=O)=O KRVSOGSZCMJSLX-UHFFFAOYSA-L 0.000 description 2
- 229960002626 clarithromycin Drugs 0.000 description 2
- AGOYDEPGAOXOCK-KCBOHYOISA-N clarithromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@](C)([C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)OC)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 AGOYDEPGAOXOCK-KCBOHYOISA-N 0.000 description 2
- 238000004891 communication Methods 0.000 description 2
- ORTQZVOHEJQUHG-UHFFFAOYSA-L copper(II) chloride Chemical compound Cl[Cu]Cl ORTQZVOHEJQUHG-UHFFFAOYSA-L 0.000 description 2
- 239000008120 corn starch Substances 0.000 description 2
- 229960000913 crospovidone Drugs 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- HGCIXCUEYOPUTN-UHFFFAOYSA-N cyclohexene Chemical compound C1CCC=CC1 HGCIXCUEYOPUTN-UHFFFAOYSA-N 0.000 description 2
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 2
- PAFZNILMFXTMIY-UHFFFAOYSA-N cyclohexylamine Chemical compound NC1CCCCC1 PAFZNILMFXTMIY-UHFFFAOYSA-N 0.000 description 2
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 2
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 2
- 230000003412 degenerative effect Effects 0.000 description 2
- 238000010511 deprotection reaction Methods 0.000 description 2
- 229960003529 diazepam Drugs 0.000 description 2
- AAOVKJBEBIDNHE-UHFFFAOYSA-N diazepam Chemical compound N=1CC(=O)N(C)C2=CC=C(Cl)C=C2C=1C1=CC=CC=C1 AAOVKJBEBIDNHE-UHFFFAOYSA-N 0.000 description 2
- 230000029087 digestion Effects 0.000 description 2
- 229940124568 digestive agent Drugs 0.000 description 2
- 125000004925 dihydropyridyl group Chemical group N1(CC=CC=C1)* 0.000 description 2
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 2
- 125000006222 dimethylaminomethyl group Chemical group [H]C([H])([H])N(C([H])([H])[H])C([H])([H])* 0.000 description 2
- FGXWKSZFVQUSTL-UHFFFAOYSA-N domperidone Chemical compound C12=CC=CC=C2NC(=O)N1CCCN(CC1)CCC1N1C2=CC=C(Cl)C=C2NC1=O FGXWKSZFVQUSTL-UHFFFAOYSA-N 0.000 description 2
- 229960001253 domperidone Drugs 0.000 description 2
- 238000006911 enzymatic reaction Methods 0.000 description 2
- 230000008029 eradication Effects 0.000 description 2
- 230000003628 erosive effect Effects 0.000 description 2
- 229960003276 erythromycin Drugs 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 239000010419 fine particle Substances 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 235000003599 food sweetener Nutrition 0.000 description 2
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 2
- 239000001530 fumaric acid Substances 0.000 description 2
- 125000000524 functional group Chemical group 0.000 description 2
- 125000003838 furazanyl group Chemical group 0.000 description 2
- AWJWCTOOIBYHON-UHFFFAOYSA-N furo[3,4-b]pyrazine-5,7-dione Chemical compound C1=CN=C2C(=O)OC(=O)C2=N1 AWJWCTOOIBYHON-UHFFFAOYSA-N 0.000 description 2
- 230000027119 gastric acid secretion Effects 0.000 description 2
- 230000005176 gastrointestinal motility Effects 0.000 description 2
- 150000008282 halocarbons Chemical class 0.000 description 2
- 229920001519 homopolymer Polymers 0.000 description 2
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 2
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 2
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 2
- HRRXCXABAPSOCP-UHFFFAOYSA-N ilaprazole Chemical compound COC1=CC=NC(CS(=O)C=2NC3=CC(=CC=C3N=2)N2C=CC=C2)=C1C HRRXCXABAPSOCP-UHFFFAOYSA-N 0.000 description 2
- 229950008491 ilaprazole Drugs 0.000 description 2
- UTCSSFWDNNEEBH-UHFFFAOYSA-N imidazo[1,2-a]pyridine Chemical compound C1=CC=CC2=NC=CN21 UTCSSFWDNNEEBH-UHFFFAOYSA-N 0.000 description 2
- UVNXNSUKKOLFBM-UHFFFAOYSA-N imidazo[2,1-b][1,3,4]thiadiazole Chemical compound N1=CSC2=NC=CN21 UVNXNSUKKOLFBM-UHFFFAOYSA-N 0.000 description 2
- 125000002632 imidazolidinyl group Chemical group 0.000 description 2
- 125000002636 imidazolinyl group Chemical group 0.000 description 2
- CGIGDMFJXJATDK-UHFFFAOYSA-N indomethacin Chemical compound CC1=C(CC(O)=O)C2=CC(OC)=CC=C2N1C(=O)C1=CC=C(Cl)C=C1 CGIGDMFJXJATDK-UHFFFAOYSA-N 0.000 description 2
- 229910052816 inorganic phosphate Inorganic materials 0.000 description 2
- 230000000968 intestinal effect Effects 0.000 description 2
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 2
- 125000005956 isoquinolyl group Chemical group 0.000 description 2
- ZLTPDFXIESTBQG-UHFFFAOYSA-N isothiazole Chemical group C=1C=NSC=1 ZLTPDFXIESTBQG-UHFFFAOYSA-N 0.000 description 2
- 125000001786 isothiazolyl group Chemical group 0.000 description 2
- 239000007951 isotonicity adjuster Substances 0.000 description 2
- 125000000842 isoxazolyl group Chemical group 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- SIXIIKVOZAGHPV-UHFFFAOYSA-N lansoprazole Chemical compound CC1=C(OCC(F)(F)F)C=CN=C1CS(=O)C1=NC2=CC=C[CH]C2=N1 SIXIIKVOZAGHPV-UHFFFAOYSA-N 0.000 description 2
- 229960003174 lansoprazole Drugs 0.000 description 2
- 239000012280 lithium aluminium hydride Substances 0.000 description 2
- 229940031703 low substituted hydroxypropyl cellulose Drugs 0.000 description 2
- 239000003120 macrolide antibiotic agent Substances 0.000 description 2
- 229910001629 magnesium chloride Inorganic materials 0.000 description 2
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 2
- 239000000347 magnesium hydroxide Substances 0.000 description 2
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 2
- 239000000395 magnesium oxide Substances 0.000 description 2
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 2
- 159000000003 magnesium salts Chemical class 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 2
- 239000011976 maleic acid Substances 0.000 description 2
- 239000001630 malic acid Substances 0.000 description 2
- 235000011090 malic acid Nutrition 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 229910001507 metal halide Inorganic materials 0.000 description 2
- 150000005309 metal halides Chemical class 0.000 description 2
- 229920003145 methacrylic acid copolymer Polymers 0.000 description 2
- 229940098779 methanesulfonic acid Drugs 0.000 description 2
- 125000005948 methanesulfonyloxy group Chemical group 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- 125000000250 methylamino group Chemical group [H]N(*)C([H])([H])[H] 0.000 description 2
- 229960004085 mosapride Drugs 0.000 description 2
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 2
- 125000004593 naphthyridinyl group Chemical group N1=C(C=CC2=CC=CN=C12)* 0.000 description 2
- 229910017604 nitric acid Inorganic materials 0.000 description 2
- 150000002825 nitriles Chemical class 0.000 description 2
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 2
- 229960000381 omeprazole Drugs 0.000 description 2
- 239000006191 orally-disintegrating tablet Substances 0.000 description 2
- 150000007530 organic bases Chemical class 0.000 description 2
- 235000006408 oxalic acid Nutrition 0.000 description 2
- LSQZJLSUYDQPKJ-UHFFFAOYSA-N p-Hydroxyampicillin Natural products O=C1N2C(C(O)=O)C(C)(C)SC2C1NC(=O)C(N)C1=CC=C(O)C=C1 LSQZJLSUYDQPKJ-UHFFFAOYSA-N 0.000 description 2
- 230000036407 pain Effects 0.000 description 2
- 125000006678 phenoxycarbonyl group Chemical group 0.000 description 2
- 230000004962 physiological condition Effects 0.000 description 2
- 239000000049 pigment Substances 0.000 description 2
- 239000006187 pill Substances 0.000 description 2
- 125000003386 piperidinyl group Chemical group 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 239000001253 polyvinylpolypyrrolidone Substances 0.000 description 2
- 239000001103 potassium chloride Substances 0.000 description 2
- 235000011164 potassium chloride Nutrition 0.000 description 2
- 229910001414 potassium ion Inorganic materials 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 230000003449 preventive effect Effects 0.000 description 2
- WGYKZJWCGVVSQN-UHFFFAOYSA-O propan-1-aminium Chemical compound CCC[NH3+] WGYKZJWCGVVSQN-UHFFFAOYSA-O 0.000 description 2
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 2
- 235000018102 proteins Nutrition 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 125000002755 pyrazolinyl group Chemical group 0.000 description 2
- PBMFSQRYOILNGV-UHFFFAOYSA-N pyridazine Chemical group C1=CC=NN=C1 PBMFSQRYOILNGV-UHFFFAOYSA-N 0.000 description 2
- 125000002098 pyridazinyl group Chemical group 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-O pyridinium Chemical compound C1=CC=[NH+]C=C1 JUJWROOIHBZHMG-UHFFFAOYSA-O 0.000 description 2
- 125000004076 pyridyl group Chemical group 0.000 description 2
- 125000000246 pyrimidin-2-yl group Chemical group [H]C1=NC(*)=NC([H])=C1[H] 0.000 description 2
- 125000004527 pyrimidin-4-yl group Chemical group N1=CN=C(C=C1)* 0.000 description 2
- 125000004528 pyrimidin-5-yl group Chemical group N1=CN=CC(=C1)* 0.000 description 2
- ZSKGQVFRTSEPJT-UHFFFAOYSA-N pyrrole-2-carboxaldehyde Chemical compound O=CC1=CC=CN1 ZSKGQVFRTSEPJT-UHFFFAOYSA-N 0.000 description 2
- 125000001422 pyrrolinyl group Chemical group 0.000 description 2
- 238000001953 recrystallisation Methods 0.000 description 2
- 230000002441 reversible effect Effects 0.000 description 2
- 229940125723 sedative agent Drugs 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 239000001632 sodium acetate Substances 0.000 description 2
- 235000017281 sodium acetate Nutrition 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- VWDWKYIASSYTQR-UHFFFAOYSA-N sodium nitrate Chemical compound [Na+].[O-][N+]([O-])=O VWDWKYIASSYTQR-UHFFFAOYSA-N 0.000 description 2
- 239000012453 solvate Substances 0.000 description 2
- 238000000638 solvent extraction Methods 0.000 description 2
- 229960002920 sorbitol Drugs 0.000 description 2
- 230000001954 sterilising effect Effects 0.000 description 2
- 238000004659 sterilization and disinfection Methods 0.000 description 2
- 239000000021 stimulant Substances 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- 125000000446 sulfanediyl group Chemical group *S* 0.000 description 2
- 238000001356 surgical procedure Methods 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- 230000009747 swallowing Effects 0.000 description 2
- 239000003765 sweetening agent Substances 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- IKBKZGMPCYNSLU-RGVLZGJSSA-N tegaserod Chemical compound C1=C(OC)C=C2C(/C=N/NC(=N)NCCCCC)=CNC2=C1 IKBKZGMPCYNSLU-RGVLZGJSSA-N 0.000 description 2
- 229960002876 tegaserod Drugs 0.000 description 2
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 2
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 125000004853 tetrahydropyridinyl group Chemical group N1(CCCC=C1)* 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 150000008334 thiadiazines Chemical group 0.000 description 2
- 150000004867 thiadiazoles Chemical group 0.000 description 2
- YUKQRDCYNOVPGJ-UHFFFAOYSA-N thioacetamide Chemical compound CC(N)=S YUKQRDCYNOVPGJ-UHFFFAOYSA-N 0.000 description 2
- BRNULMACUQOKMR-UHFFFAOYSA-N thiomorpholine Chemical group C1CSCCN1 BRNULMACUQOKMR-UHFFFAOYSA-N 0.000 description 2
- URAYPUMNDPQOKB-UHFFFAOYSA-N triacetin Chemical compound CC(=O)OCC(OC(C)=O)COC(C)=O URAYPUMNDPQOKB-UHFFFAOYSA-N 0.000 description 2
- 150000003852 triazoles Chemical group 0.000 description 2
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- YFTHZRPMJXBUME-UHFFFAOYSA-N tripropylamine Chemical compound CCCN(CCC)CCC YFTHZRPMJXBUME-UHFFFAOYSA-N 0.000 description 2
- NOOLISFMXDJSKH-UTLUCORTSA-N (+)-Neomenthol Chemical compound CC(C)[C@@H]1CC[C@@H](C)C[C@@H]1O NOOLISFMXDJSKH-UTLUCORTSA-N 0.000 description 1
- HMJIYCCIJYRONP-UHFFFAOYSA-N (+-)-Isradipine Chemical compound COC(=O)C1=C(C)NC(C)=C(C(=O)OC(C)C)C1C1=CC=CC2=NON=C12 HMJIYCCIJYRONP-UHFFFAOYSA-N 0.000 description 1
- FTLYMKDSHNWQKD-UHFFFAOYSA-N (2,4,5-trichlorophenyl)boronic acid Chemical compound OB(O)C1=CC(Cl)=C(Cl)C=C1Cl FTLYMKDSHNWQKD-UHFFFAOYSA-N 0.000 description 1
- IAJNFGSAQDDBSS-UHFFFAOYSA-N (2-ethylphenyl) butanoate Chemical compound CCCC(=O)OC1=CC=CC=C1CC IAJNFGSAQDDBSS-UHFFFAOYSA-N 0.000 description 1
- CCIWVEMVBWEMCY-RCFOMQFPSA-N (2s)-1-[(3as,4s,7as)-4-hydroxy-4-(2-methoxyphenyl)-7,7-diphenyl-1,3,3a,5,6,7a-hexahydroisoindol-2-yl]-2-(2-methoxyphenyl)propan-1-one Chemical compound COC1=CC=CC=C1[C@H](C)C(=O)N1C[C@H](C(CC[C@@]2(O)C=3C(=CC=CC=3)OC)(C=3C=CC=CC=3)C=3C=CC=CC=3)[C@H]2C1 CCIWVEMVBWEMCY-RCFOMQFPSA-N 0.000 description 1
- NPSVXOVMLVOMDD-SXRVEDALSA-N (2s)-2-[[(3s,6s,9s,12s)-12-[[(2s)-4-[[(2r,3r,4r,5s,6r)-3-acetamido-4,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]amino]-2-amino-4-oxobutanoyl]amino]-6-benzyl-9-(1h-indol-3-ylmethyl)-5,8,11,14-tetraoxo-1,4,7,10-tetrazacyclotetradecane-3-carbonyl]amino]-4-methyl Chemical compound C([C@H](N)C(=O)N[C@H]1CC(=O)NC[C@H](NC(=O)[C@H](CC=2C=CC=CC=2)NC(=O)[C@H](CC=2C3=CC=CC=C3NC=2)NC1=O)C(=O)N[C@@H](CC(C)C)C(O)=O)C(=O)N[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1NC(C)=O NPSVXOVMLVOMDD-SXRVEDALSA-N 0.000 description 1
- MOLOJNHYNHBPCW-ZETCQYMHSA-N (2s)-2-amino-4-[2-(1-aminoethylideneamino)ethylsulfanyl]butanoic acid Chemical compound CC(=N)NCCSCC[C@H](N)C(O)=O MOLOJNHYNHBPCW-ZETCQYMHSA-N 0.000 description 1
- MFOOVZCXWVAWOV-RUZDIDTESA-N (3r)-5-[2-(8-azaspiro[4.5]decane-8-carbonyl)-4,6-dimethylanilino]-3-naphthalen-1-yl-5-oxopentanoic acid Chemical compound C=1C(C)=CC(C)=C(NC(=O)C[C@H](CC(O)=O)C=2C3=CC=CC=C3C=CC=2)C=1C(=O)N(CC1)CCC21CCCC2 MFOOVZCXWVAWOV-RUZDIDTESA-N 0.000 description 1
- YFZOSOXDZQOKGB-UHFFFAOYSA-N (4-phenyl-5-phenylsulfanyl-1,3-thiazol-2-yl)methanamine Chemical compound S1C(CN)=NC(C=2C=CC=CC=2)=C1SC1=CC=CC=C1 YFZOSOXDZQOKGB-UHFFFAOYSA-N 0.000 description 1
- FFTVPQUHLQBXQZ-KVUCHLLUSA-N (4s,4as,5ar,12ar)-4,7-bis(dimethylamino)-1,10,11,12a-tetrahydroxy-3,12-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide Chemical compound C1C2=C(N(C)C)C=CC(O)=C2C(O)=C2[C@@H]1C[C@H]1[C@H](N(C)C)C(=O)C(C(N)=O)=C(O)[C@@]1(O)C2=O FFTVPQUHLQBXQZ-KVUCHLLUSA-N 0.000 description 1
- ZWIXEQBDJMFCMN-DHHNQDMHSA-N (7r,10r,13s,16r)-13-cyclopropyl-7-[(4-fluorophenyl)methyl]-10,11,16-trimethyl-17-oxa-5,8,11,14-tetrazabicyclo[16.4.0]docosa-1(22),18,20-triene-6,9,12-trione;hydrate;hydrochloride Chemical compound O.Cl.C1([C@@H]2NC[C@H](OC3=CC=CC=C3CCCNC(=O)[C@@H](CC=3C=CC(F)=CC=3)NC(=O)[C@@H](C)N(C)C2=O)C)CC1 ZWIXEQBDJMFCMN-DHHNQDMHSA-N 0.000 description 1
- IDFPQEHZYBXIFO-GFCCVEGCSA-N (R)-(4-fluoro-2-propylphenyl)-(1H-imidazol-2-yl)methanol Chemical compound CCCc1cc(F)ccc1[C@@H](O)c1ncc[nH]1 IDFPQEHZYBXIFO-GFCCVEGCSA-N 0.000 description 1
- PVHUJELLJLJGLN-INIZCTEOSA-N (S)-nitrendipine Chemical compound CCOC(=O)C1=C(C)NC(C)=C(C(=O)OC)[C@@H]1C1=CC=CC([N+]([O-])=O)=C1 PVHUJELLJLJGLN-INIZCTEOSA-N 0.000 description 1
- KYVBNYUBXIEUFW-UHFFFAOYSA-N 1,1,3,3-tetramethylguanidine Chemical compound CN(C)C(=N)N(C)C KYVBNYUBXIEUFW-UHFFFAOYSA-N 0.000 description 1
- UDGKZGLPXCRRAM-UHFFFAOYSA-N 1,2,5-thiadiazole Chemical compound C=1C=NSN=1 UDGKZGLPXCRRAM-UHFFFAOYSA-N 0.000 description 1
- ABADUMLIAZCWJD-UHFFFAOYSA-N 1,3-dioxole Chemical compound C1OC=CO1 ABADUMLIAZCWJD-UHFFFAOYSA-N 0.000 description 1
- 150000000192 1,4-thiazines Chemical group 0.000 description 1
- 125000000183 1,4-thiazinyl group Chemical group S1C(C=NC=C1)* 0.000 description 1
- LQIAZOCLNBBZQK-UHFFFAOYSA-N 1-(1,2-Diphosphanylethyl)pyrrolidin-2-one Chemical compound PCC(P)N1CCCC1=O LQIAZOCLNBBZQK-UHFFFAOYSA-N 0.000 description 1
- BXQOBMZMKYGXJN-UHFFFAOYSA-N 1-(2,6-difluorophenyl)sulfonyl-2-phenylimidazole-4-carbaldehyde Chemical compound FC1=CC=CC(F)=C1S(=O)(=O)N1C(C=2C=CC=CC=2)=NC(C=O)=C1 BXQOBMZMKYGXJN-UHFFFAOYSA-N 0.000 description 1
- QYIGOGBGVKONDY-UHFFFAOYSA-N 1-(2-bromo-5-chlorophenyl)-3-methylpyrazole Chemical compound N1=C(C)C=CN1C1=CC(Cl)=CC=C1Br QYIGOGBGVKONDY-UHFFFAOYSA-N 0.000 description 1
- TYBKOOHPCMWWKY-UHFFFAOYSA-N 1-(4-nitrophenyl)-2-(4-nitrophenyl)sulfanylimidazole Chemical compound C1=CC([N+](=O)[O-])=CC=C1SC1=NC=CN1C1=CC=C([N+]([O-])=O)C=C1 TYBKOOHPCMWWKY-UHFFFAOYSA-N 0.000 description 1
- JQSAYKKFZOSZGJ-UHFFFAOYSA-N 1-[bis(4-fluorophenyl)methyl]-4-[(2,3,4-trimethoxyphenyl)methyl]piperazine Chemical compound COC1=C(OC)C(OC)=CC=C1CN1CCN(C(C=2C=CC(F)=CC=2)C=2C=CC(F)=CC=2)CC1 JQSAYKKFZOSZGJ-UHFFFAOYSA-N 0.000 description 1
- FKIIVBOPAHICHQ-UHFFFAOYSA-N 1-benzothiophene-2-sulfonyl chloride Chemical compound C1=CC=C2SC(S(=O)(=O)Cl)=CC2=C1 FKIIVBOPAHICHQ-UHFFFAOYSA-N 0.000 description 1
- RCMLKQSZCSYLLS-UHFFFAOYSA-N 1-hydroxy-2,3-dihydroindole Chemical compound C1=CC=C2N(O)CCC2=C1 RCMLKQSZCSYLLS-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- 125000001088 1-naphthoyl group Chemical group C1(=CC=CC2=CC=CC=C12)C(=O)* 0.000 description 1
- ALWXETURCOIGIZ-UHFFFAOYSA-N 1-nitropropylbenzene Chemical compound CCC([N+]([O-])=O)C1=CC=CC=C1 ALWXETURCOIGIZ-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- ZGABDPXDUGYGQE-UHFFFAOYSA-N 1-piperidin-1-ylpiperazine Chemical compound C1CCCCN1N1CCNCC1 ZGABDPXDUGYGQE-UHFFFAOYSA-N 0.000 description 1
- 125000001462 1-pyrrolyl group Chemical group [*]N1C([H])=C([H])C([H])=C1[H] 0.000 description 1
- VFTFKUDGYRBSAL-UHFFFAOYSA-N 15-crown-5 Chemical compound C1COCCOCCOCCOCCO1 VFTFKUDGYRBSAL-UHFFFAOYSA-N 0.000 description 1
- CHNYVNOFAWYUEG-UHFFFAOYSA-N 1h-pyrrole-3-carbaldehyde Chemical compound O=CC=1C=CNC=1 CHNYVNOFAWYUEG-UHFFFAOYSA-N 0.000 description 1
- PVJZBZSCGJAWNG-UHFFFAOYSA-N 2,4,6-trimethylbenzenesulfonyl chloride Chemical compound CC1=CC(C)=C(S(Cl)(=O)=O)C(C)=C1 PVJZBZSCGJAWNG-UHFFFAOYSA-N 0.000 description 1
- QXWAUQMMMIMLTO-UHFFFAOYSA-N 2,6-difluorobenzenesulfonyl chloride Chemical compound FC1=CC=CC(F)=C1S(Cl)(=O)=O QXWAUQMMMIMLTO-UHFFFAOYSA-N 0.000 description 1
- SGTNSNPWRIOYBX-UHFFFAOYSA-N 2-(3,4-dimethoxyphenyl)-5-{[2-(3,4-dimethoxyphenyl)ethyl](methyl)amino}-2-(propan-2-yl)pentanenitrile Chemical compound C1=C(OC)C(OC)=CC=C1CCN(C)CCCC(C#N)(C(C)C)C1=CC=C(OC)C(OC)=C1 SGTNSNPWRIOYBX-UHFFFAOYSA-N 0.000 description 1
- NSVFSAJIGAJDMR-UHFFFAOYSA-N 2-[benzyl(phenyl)amino]ethyl 5-(5,5-dimethyl-2-oxido-1,3,2-dioxaphosphinan-2-yl)-2,6-dimethyl-4-(3-nitrophenyl)-1,4-dihydropyridine-3-carboxylate Chemical compound CC=1NC(C)=C(C(=O)OCCN(CC=2C=CC=CC=2)C=2C=CC=CC=2)C(C=2C=C(C=CC=2)[N+]([O-])=O)C=1P1(=O)OCC(C)(C)CO1 NSVFSAJIGAJDMR-UHFFFAOYSA-N 0.000 description 1
- YGZFYDFBHIDIBH-UHFFFAOYSA-N 2-[bis(2-hydroxyethyl)amino]icosan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCC(CO)N(CCO)CCO YGZFYDFBHIDIBH-UHFFFAOYSA-N 0.000 description 1
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- 125000004174 2-benzimidazolyl group Chemical group [H]N1C(*)=NC2=C([H])C([H])=C([H])C([H])=C12 0.000 description 1
- KKMOSYLWYLMHAL-UHFFFAOYSA-N 2-bromo-6-nitroaniline Chemical compound NC1=C(Br)C=CC=C1[N+]([O-])=O KKMOSYLWYLMHAL-UHFFFAOYSA-N 0.000 description 1
- REXUYBKPWIPONM-UHFFFAOYSA-N 2-bromoacetonitrile Chemical compound BrCC#N REXUYBKPWIPONM-UHFFFAOYSA-N 0.000 description 1
- 125000004198 2-fluorophenyl group Chemical group [H]C1=C([H])C(F)=C(*)C([H])=C1[H] 0.000 description 1
- JSGQQGNVTXXNQN-UHFFFAOYSA-N 2-hexadecoxy-2-oxoacetic acid Chemical compound CCCCCCCCCCCCCCCCOC(=O)C(O)=O JSGQQGNVTXXNQN-UHFFFAOYSA-N 0.000 description 1
- 125000006040 2-hexenyl group Chemical group 0.000 description 1
- XWKFPIODWVPXLX-UHFFFAOYSA-N 2-methyl-5-methylpyridine Natural products CC1=CC=C(C)N=C1 XWKFPIODWVPXLX-UHFFFAOYSA-N 0.000 description 1
- BSKHPKMHTQYZBB-UHFFFAOYSA-N 2-methylpyridine Chemical compound CC1=CC=CC=N1 BSKHPKMHTQYZBB-UHFFFAOYSA-N 0.000 description 1
- FOQYDURHXZVLFT-UHFFFAOYSA-N 2-phenyl-2-pyridin-2-ylethanethioamide Chemical compound C=1C=CC=NC=1C(C(=S)N)C1=CC=CC=C1 FOQYDURHXZVLFT-UHFFFAOYSA-N 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- UVDDGALZIHEYOK-UHFFFAOYSA-N 2-phenylsulfanylacetaldehyde Chemical compound O=CCSC1=CC=CC=C1 UVDDGALZIHEYOK-UHFFFAOYSA-N 0.000 description 1
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 1
- QMEQBOSUJUOXMX-UHFFFAOYSA-N 2h-oxadiazine Chemical group N1OC=CC=N1 QMEQBOSUJUOXMX-UHFFFAOYSA-N 0.000 description 1
- AEDQNOLIADXSBB-UHFFFAOYSA-N 3-(dodecylazaniumyl)propanoate Chemical compound CCCCCCCCCCCCNCCC(O)=O AEDQNOLIADXSBB-UHFFFAOYSA-N 0.000 description 1
- ONNMDRQRSGKZCN-UHFFFAOYSA-N 3-aminopropyl(butyl)phosphinic acid Chemical compound CCCCP(O)(=O)CCCN ONNMDRQRSGKZCN-UHFFFAOYSA-N 0.000 description 1
- JHJKSEKUZNJKGO-UHFFFAOYSA-N 3-methoxybenzenesulfonyl chloride Chemical compound COC1=CC=CC(S(Cl)(=O)=O)=C1 JHJKSEKUZNJKGO-UHFFFAOYSA-N 0.000 description 1
- 125000005977 3-phenylpropyloxy group Chemical group 0.000 description 1
- 239000003185 4 aminobutyric acid B receptor stimulating agent Substances 0.000 description 1
- 239000003148 4 aminobutyric acid receptor blocking agent Substances 0.000 description 1
- 239000003477 4 aminobutyric acid receptor stimulating agent Substances 0.000 description 1
- XASKKMXCSTYBPB-UHFFFAOYSA-N 4-(1-thiomorpholin-4-ylazepan-2-yl)morpholine Chemical compound O1CCN(CC1)C1N(CCCCC1)N1CCSCC1 XASKKMXCSTYBPB-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N 4-hydroxybenzoic acid Chemical class OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 125000006418 4-methylphenylsulfonyl group Chemical group 0.000 description 1
- OBKXEAXTFZPCHS-UHFFFAOYSA-M 4-phenylbutyrate Chemical compound [O-]C(=O)CCCC1=CC=CC=C1 OBKXEAXTFZPCHS-UHFFFAOYSA-M 0.000 description 1
- PJJGZPJJTHBVMX-UHFFFAOYSA-N 5,7-Dihydroxyisoflavone Chemical compound C=1C(O)=CC(O)=C(C2=O)C=1OC=C2C1=CC=CC=C1 PJJGZPJJTHBVMX-UHFFFAOYSA-N 0.000 description 1
- 125000006618 5- to 10-membered aromatic heterocyclic group Chemical group 0.000 description 1
- ZBFDAUIVDSSISP-UHFFFAOYSA-N 5-methoxy-2-[(4-methoxy-3,5-dimethyl-2-pyridinyl)methylsulfinyl]-1H-imidazo[4,5-b]pyridine Chemical compound N=1C2=NC(OC)=CC=C2NC=1S(=O)CC1=NC=C(C)C(OC)=C1C ZBFDAUIVDSSISP-UHFFFAOYSA-N 0.000 description 1
- OEDUIFSDODUDRK-UHFFFAOYSA-N 5-phenyl-1h-pyrazole Chemical compound N1N=CC=C1C1=CC=CC=C1 OEDUIFSDODUDRK-UHFFFAOYSA-N 0.000 description 1
- WFNBZLITOZFWMW-UHFFFAOYSA-N 5-phenylsulfanyl-1,3-thiazole Chemical compound C1(=CC=CC=C1)SC1=CN=CS1 WFNBZLITOZFWMW-UHFFFAOYSA-N 0.000 description 1
- PVXPPJIGRGXGCY-DJHAAKORSA-N 6-O-alpha-D-glucopyranosyl-alpha-D-fructofuranose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1OC[C@@H]1[C@@H](O)[C@H](O)[C@](O)(CO)O1 PVXPPJIGRGXGCY-DJHAAKORSA-N 0.000 description 1
- JRKLRIAIMIKGHT-UHFFFAOYSA-N 8-bromoquinoline-4-carbaldehyde Chemical compound C1=CN=C2C(Br)=CC=CC2=C1C=O JRKLRIAIMIKGHT-UHFFFAOYSA-N 0.000 description 1
- GSDSWSVVBLHKDQ-UHFFFAOYSA-N 9-fluoro-3-methyl-10-(4-methylpiperazin-1-yl)-7-oxo-2,3-dihydro-7H-[1,4]oxazino[2,3,4-ij]quinoline-6-carboxylic acid Chemical compound FC1=CC(C(C(C(O)=O)=C2)=O)=C3N2C(C)COC3=C1N1CCN(C)CC1 GSDSWSVVBLHKDQ-UHFFFAOYSA-N 0.000 description 1
- ZKHQWZAMYRWXGA-KQYNXXCUSA-J ATP(4-) Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](COP([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O)[C@@H](O)[C@H]1O ZKHQWZAMYRWXGA-KQYNXXCUSA-J 0.000 description 1
- 108091006112 ATPases Proteins 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- IKHGUXGNUITLKF-UHFFFAOYSA-N Acetaldehyde Chemical compound CC=O IKHGUXGNUITLKF-UHFFFAOYSA-N 0.000 description 1
- 102000057290 Adenosine Triphosphatases Human genes 0.000 description 1
- ZKHQWZAMYRWXGA-UHFFFAOYSA-N Adenosine triphosphate Natural products C1=NC=2C(N)=NC=NC=2N1C1OC(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)C(O)C1O ZKHQWZAMYRWXGA-UHFFFAOYSA-N 0.000 description 1
- WLDHEUZGFKACJH-ZRUFZDNISA-K Amaranth Chemical compound [Na+].[Na+].[Na+].C12=CC=C(S([O-])(=O)=O)C=C2C=C(S([O-])(=O)=O)C(O)=C1\N=N\C1=CC=C(S([O-])(=O)=O)C2=CC=CC=C12 WLDHEUZGFKACJH-ZRUFZDNISA-K 0.000 description 1
- 102000013142 Amylases Human genes 0.000 description 1
- 108010065511 Amylases Proteins 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- NCUCGYYHUFIYNU-UHFFFAOYSA-N Aranidipine Chemical compound COC(=O)C1=C(C)NC(C)=C(C(=O)OCC(C)=O)C1C1=CC=CC=C1[N+]([O-])=O NCUCGYYHUFIYNU-UHFFFAOYSA-N 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 1
- 208000023275 Autoimmune disease Diseases 0.000 description 1
- 229920003084 Avicel® PH-102 Polymers 0.000 description 1
- ZKFQEACEUNWPMT-UHFFFAOYSA-N Azelnidipine Chemical compound CC(C)OC(=O)C1=C(C)NC(N)=C(C(=O)OC2CN(C2)C(C=2C=CC=CC=2)C=2C=CC=CC=2)C1C1=CC=CC([N+]([O-])=O)=C1 ZKFQEACEUNWPMT-UHFFFAOYSA-N 0.000 description 1
- IUWLTSZHVYHOHY-FJXQXJEOSA-M C1=C(NC=N1)C[C@@H](C(=O)O)[N-]C(=O)CC[NH-].[Zn+2] Chemical compound C1=C(NC=N1)C[C@@H](C(=O)O)[N-]C(=O)CC[NH-].[Zn+2] IUWLTSZHVYHOHY-FJXQXJEOSA-M 0.000 description 1
- 229940127291 Calcium channel antagonist Drugs 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 206010007269 Carcinogenicity Diseases 0.000 description 1
- 206010048610 Cardiotoxicity Diseases 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 235000008733 Citrus aurantifolia Nutrition 0.000 description 1
- 235000005979 Citrus limon Nutrition 0.000 description 1
- 244000131522 Citrus pyriformis Species 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229910021592 Copper(II) chloride Inorganic materials 0.000 description 1
- NOOLISFMXDJSKH-UHFFFAOYSA-N DL-menthol Natural products CC(C)C1CCC(C)CC1O NOOLISFMXDJSKH-UHFFFAOYSA-N 0.000 description 1
- 239000004287 Dehydroacetic acid Substances 0.000 description 1
- XBPCUCUWBYBCDP-UHFFFAOYSA-N Dicyclohexylamine Chemical compound C1CCCCC1NC1CCCCC1 XBPCUCUWBYBCDP-UHFFFAOYSA-N 0.000 description 1
- 241000790917 Dioxys <bee> Species 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 239000004386 Erythritol Substances 0.000 description 1
- UNXHWFMMPAWVPI-UHFFFAOYSA-N Erythritol Natural products OCC(O)C(O)CO UNXHWFMMPAWVPI-UHFFFAOYSA-N 0.000 description 1
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- 229920003163 Eudragit® NE 30 D Polymers 0.000 description 1
- 229920003157 Eudragit® RL 30 D Polymers 0.000 description 1
- 229920003161 Eudragit® RS 30 D Polymers 0.000 description 1
- 229920003134 Eudragit® polymer Polymers 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 229920001917 Ficoll Polymers 0.000 description 1
- 235000016623 Fragaria vesca Nutrition 0.000 description 1
- 240000009088 Fragaria x ananassa Species 0.000 description 1
- 235000011363 Fragaria x ananassa Nutrition 0.000 description 1
- XQLWNAFCTODIRK-UHFFFAOYSA-N Gallopamil Chemical compound C1=C(OC)C(OC)=CC=C1CCN(C)CCCC(C#N)(C(C)C)C1=CC(OC)=C(OC)C(OC)=C1 XQLWNAFCTODIRK-UHFFFAOYSA-N 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- CEAZRRDELHUEMR-URQXQFDESA-N Gentamicin Chemical compound O1[C@H](C(C)NC)CC[C@@H](N)[C@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](NC)[C@@](C)(O)CO2)O)[C@H](N)C[C@@H]1N CEAZRRDELHUEMR-URQXQFDESA-N 0.000 description 1
- 229930182566 Gentamicin Natural products 0.000 description 1
- 241001071795 Gentiana Species 0.000 description 1
- 101800001586 Ghrelin Proteins 0.000 description 1
- 102000012004 Ghrelin Human genes 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- 239000004378 Glycyrrhizin Substances 0.000 description 1
- 229920002907 Guar gum Polymers 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 206010019196 Head injury Diseases 0.000 description 1
- 208000032843 Hemorrhage Diseases 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- HEFNNWSXXWATRW-UHFFFAOYSA-N Ibuprofen Chemical compound CC(C)CC1=CC=C(C(C)C(O)=O)C=C1 HEFNNWSXXWATRW-UHFFFAOYSA-N 0.000 description 1
- 102000000589 Interleukin-1 Human genes 0.000 description 1
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 1
- 229920003085 Kollidon® CL Polymers 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- KMZQAVXSMUKBPD-DJWKRKHSSA-N Lafutidine Chemical compound C=1C=COC=1C[S+]([O-])CC(=O)NC\C=C/COC(N=CC=1)=CC=1CN1CCCCC1 KMZQAVXSMUKBPD-DJWKRKHSSA-N 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- 108010092101 MEN 11420 Proteins 0.000 description 1
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 1
- BYBLEWFAAKGYCD-UHFFFAOYSA-N Miconazole Chemical compound ClC1=CC(Cl)=CC=C1COC(C=1C(=CC(Cl)=CC=1)Cl)CN1C=NC=C1 BYBLEWFAAKGYCD-UHFFFAOYSA-N 0.000 description 1
- 208000034486 Multi-organ failure Diseases 0.000 description 1
- 208000010718 Multiple Organ Failure Diseases 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- WUKZPHOXUVCQOR-UHFFFAOYSA-N N-(1-azabicyclo[2.2.2]octan-3-yl)-6-chloro-4-methyl-3-oxo-1,4-benzoxazine-8-carboxamide Chemical compound C1N(CC2)CCC2C1NC(=O)C1=CC(Cl)=CC2=C1OCC(=O)N2C WUKZPHOXUVCQOR-UHFFFAOYSA-N 0.000 description 1
- QQQIECGTIMUVDS-UHFFFAOYSA-N N-[[4-[2-(dimethylamino)ethoxy]phenyl]methyl]-3,4-dimethoxybenzamide Chemical compound C1=C(OC)C(OC)=CC=C1C(=O)NCC1=CC=C(OCCN(C)C)C=C1 QQQIECGTIMUVDS-UHFFFAOYSA-N 0.000 description 1
- ZBBHBTPTTSWHBA-UHFFFAOYSA-N Nicardipine Chemical compound COC(=O)C1=C(C)NC(C)=C(C(=O)OCCN(C)CC=2C=CC=CC=2)C1C1=CC=CC([N+]([O-])=O)=C1 ZBBHBTPTTSWHBA-UHFFFAOYSA-N 0.000 description 1
- FAIIFDPAEUKBEP-UHFFFAOYSA-N Nilvadipine Chemical compound COC(=O)C1=C(C#N)NC(C)=C(C(=O)OC(C)C)C1C1=CC=CC([N+]([O-])=O)=C1 FAIIFDPAEUKBEP-UHFFFAOYSA-N 0.000 description 1
- 229940123921 Nitric oxide synthase inhibitor Drugs 0.000 description 1
- JCXJVPUVTGWSNB-UHFFFAOYSA-N Nitrogen dioxide Chemical compound O=[N]=O JCXJVPUVTGWSNB-UHFFFAOYSA-N 0.000 description 1
- 239000008913 Normacol Substances 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- IQPSEEYGBUAQFF-UHFFFAOYSA-N Pantoprazole Chemical compound COC1=CC=NC(CS(=O)C=2NC3=CC=C(OC(F)F)C=C3N=2)=C1OC IQPSEEYGBUAQFF-UHFFFAOYSA-N 0.000 description 1
- 241000233805 Phoenix Species 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- 239000004373 Pullulan Substances 0.000 description 1
- 229920001218 Pullulan Polymers 0.000 description 1
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- ALLWOAVDORUJLA-UHFFFAOYSA-N Rebamipida Chemical compound C=1C(=O)NC2=CC=CC=C2C=1CC(C(=O)O)NC(=O)C1=CC=C(Cl)C=C1 ALLWOAVDORUJLA-UHFFFAOYSA-N 0.000 description 1
- 206010074268 Reproductive toxicity Diseases 0.000 description 1
- 241000709400 Ruba Species 0.000 description 1
- 244000000231 Sesamum indicum Species 0.000 description 1
- 229920001800 Shellac Polymers 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- 244000228451 Stevia rebaudiana Species 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 1
- LINDOXZENKYESA-UHFFFAOYSA-N TMG Natural products CNC(N)=NC LINDOXZENKYESA-UHFFFAOYSA-N 0.000 description 1
- 239000004098 Tetracycline Substances 0.000 description 1
- 206010053615 Thermal burn Diseases 0.000 description 1
- WKDDRNSBRWANNC-UHFFFAOYSA-N Thienamycin Natural products C1C(SCCN)=C(C(O)=O)N2C(=O)C(C(O)C)C21 WKDDRNSBRWANNC-UHFFFAOYSA-N 0.000 description 1
- 235000011941 Tilia x europaea Nutrition 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- 102000004357 Transferases Human genes 0.000 description 1
- 108090000992 Transferases Proteins 0.000 description 1
- 102100029613 Transient receptor potential cation channel subfamily V member 1 Human genes 0.000 description 1
- 108050004388 Transient receptor potential cation channel subfamily V member 1 Proteins 0.000 description 1
- DOOTYTYQINUNNV-UHFFFAOYSA-N Triethyl citrate Chemical compound CCOC(=O)CC(O)(C(=O)OCC)CC(=O)OCC DOOTYTYQINUNNV-UHFFFAOYSA-N 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 108010067973 Valinomycin Proteins 0.000 description 1
- 235000010724 Wisteria floribunda Nutrition 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- GTUNMKRGRHOANR-UHFFFAOYSA-N [B].[Ca] Chemical compound [B].[Ca] GTUNMKRGRHOANR-UHFFFAOYSA-N 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- VJHCJDRQFCCTHL-UHFFFAOYSA-N acetic acid 2,3,4,5,6-pentahydroxyhexanal Chemical compound CC(O)=O.OCC(O)C(O)C(O)C(O)C=O VJHCJDRQFCCTHL-UHFFFAOYSA-N 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- IYKJEILNJZQJPU-UHFFFAOYSA-N acetic acid;butanedioic acid Chemical compound CC(O)=O.OC(=O)CCC(O)=O IYKJEILNJZQJPU-UHFFFAOYSA-N 0.000 description 1
- MOQOOKGPCBQMCY-UHFFFAOYSA-N acetic acid;hexane Chemical compound CC(O)=O.CCCCCC MOQOOKGPCBQMCY-UHFFFAOYSA-N 0.000 description 1
- WJGAPUXHSQQWQF-UHFFFAOYSA-N acetic acid;hydrochloride Chemical compound Cl.CC(O)=O WJGAPUXHSQQWQF-UHFFFAOYSA-N 0.000 description 1
- 229960001138 acetylsalicylic acid Drugs 0.000 description 1
- 230000007059 acute toxicity Effects 0.000 description 1
- 231100000403 acute toxicity Toxicity 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 125000004466 alkoxycarbonylamino group Chemical group 0.000 description 1
- 125000005194 alkoxycarbonyloxy group Chemical group 0.000 description 1
- 125000005196 alkyl carbonyloxy group Chemical group 0.000 description 1
- 125000004656 alkyl sulfonylamino group Chemical group 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 125000005530 alkylenedioxy group Chemical group 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- JSWZEAMFRNKZNL-UHFFFAOYSA-N alosetron Chemical compound N1C=NC(CN2C(C3=C(N(C4=CC=CC=C43)C)CC2)=O)=C1C JSWZEAMFRNKZNL-UHFFFAOYSA-N 0.000 description 1
- 229960003550 alosetron Drugs 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- WMGSQTMJHBYJMQ-UHFFFAOYSA-N aluminum;magnesium;silicate Chemical compound [Mg+2].[Al+3].[O-][Si]([O-])([O-])[O-] WMGSQTMJHBYJMQ-UHFFFAOYSA-N 0.000 description 1
- LCWAOCHOPBSGMU-UHFFFAOYSA-J aluminum;magnesium;sodium;hydrogen carbonate;oxygen(2-);silicon;trihydroxide Chemical compound [OH-].[OH-].[OH-].[O-2].[Na+].[Mg+2].[Al+3].[Si].OC([O-])=O LCWAOCHOPBSGMU-UHFFFAOYSA-J 0.000 description 1
- 229940024554 amdinocillin Drugs 0.000 description 1
- 229960004821 amikacin Drugs 0.000 description 1
- LKCWBDHBTVXHDL-RMDFUYIESA-N amikacin Chemical compound O([C@@H]1[C@@H](N)C[C@H]([C@@H]([C@H]1O)O[C@@H]1[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O1)O)NC(=O)[C@@H](O)CCN)[C@H]1O[C@H](CN)[C@@H](O)[C@H](O)[C@H]1O LKCWBDHBTVXHDL-RMDFUYIESA-N 0.000 description 1
- 239000002647 aminoglycoside antibiotic agent Substances 0.000 description 1
- 150000003927 aminopyridines Chemical class 0.000 description 1
- 229960000528 amlodipine Drugs 0.000 description 1
- HTIQEAQVCYTUBX-UHFFFAOYSA-N amlodipine Chemical compound CCOC(=O)C1=C(COCCN)NC(C)=C(C(=O)OC)C1C1=CC=CC=C1Cl HTIQEAQVCYTUBX-UHFFFAOYSA-N 0.000 description 1
- 239000011609 ammonium molybdate Substances 0.000 description 1
- APUPEJJSWDHEBO-UHFFFAOYSA-P ammonium molybdate Chemical compound [NH4+].[NH4+].[O-][Mo]([O-])(=O)=O APUPEJJSWDHEBO-UHFFFAOYSA-P 0.000 description 1
- 235000018660 ammonium molybdate Nutrition 0.000 description 1
- 229940010552 ammonium molybdate Drugs 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- 125000000748 anthracen-2-yl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C([H])=C([*])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 239000003420 antiserotonin agent Substances 0.000 description 1
- 239000003699 antiulcer agent Substances 0.000 description 1
- ATALOFNDEOCMKK-OITMNORJSA-N aprepitant Chemical compound O([C@@H]([C@@H]1C=2C=CC(F)=CC=2)O[C@H](C)C=2C=C(C=C(C=2)C(F)(F)F)C(F)(F)F)CCN1CC1=NNC(=O)N1 ATALOFNDEOCMKK-OITMNORJSA-N 0.000 description 1
- 150000003974 aralkylamines Chemical class 0.000 description 1
- 229950007556 aranidipine Drugs 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 150000001491 aromatic compounds Chemical group 0.000 description 1
- 125000001691 aryl alkyl amino group Chemical group 0.000 description 1
- 125000004659 aryl alkyl thio group Chemical group 0.000 description 1
- 125000002102 aryl alkyloxo group Chemical group 0.000 description 1
- 125000005199 aryl carbonyloxy group Chemical group 0.000 description 1
- 125000005161 aryl oxy carbonyl group Chemical group 0.000 description 1
- 125000005135 aryl sulfinyl group Chemical group 0.000 description 1
- 125000004657 aryl sulfonyl amino group Chemical group 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 229950005951 azasetron Drugs 0.000 description 1
- 229950004646 azelnidipine Drugs 0.000 description 1
- 159000000009 barium salts Chemical class 0.000 description 1
- 229960002992 barnidipine Drugs 0.000 description 1
- VXMOONUMYLCFJD-DHLKQENFSA-N barnidipine Chemical compound C1([C@@H]2C(=C(C)NC(C)=C2C(=O)OC)C(=O)O[C@@H]2CN(CC=3C=CC=CC=3)CC2)=CC=CC([N+]([O-])=O)=C1 VXMOONUMYLCFJD-DHLKQENFSA-N 0.000 description 1
- 229960000686 benzalkonium chloride Drugs 0.000 description 1
- UREZNYTWGJKWBI-UHFFFAOYSA-M benzethonium chloride Chemical compound [Cl-].C1=CC(C(C)(C)CC(C)(C)C)=CC=C1OCCOCC[N+](C)(C)CC1=CC=CC=C1 UREZNYTWGJKWBI-UHFFFAOYSA-M 0.000 description 1
- 229960001950 benzethonium chloride Drugs 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 125000004604 benzisothiazolyl group Chemical group S1N=C(C2=C1C=CC=C2)* 0.000 description 1
- 125000004603 benzisoxazolyl group Chemical group O1N=C(C2=C1C=CC=C2)* 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 229960002903 benzyl benzoate Drugs 0.000 description 1
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 1
- 229960003665 bepridil Drugs 0.000 description 1
- UIEATEWHFDRYRU-UHFFFAOYSA-N bepridil Chemical compound C1CCCN1C(COCC(C)C)CN(C=1C=CC=CC=1)CC1=CC=CC=C1 UIEATEWHFDRYRU-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 229960003588 bevantolol Drugs 0.000 description 1
- HXLAFSUPPDYFEO-UHFFFAOYSA-N bevantolol Chemical compound C1=C(OC)C(OC)=CC=C1CCNCC(O)COC1=CC=CC(C)=C1 HXLAFSUPPDYFEO-UHFFFAOYSA-N 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 125000006267 biphenyl group Chemical group 0.000 description 1
- 150000001621 bismuth Chemical class 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 239000012267 brine Substances 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- ODWXUNBKCRECNW-UHFFFAOYSA-M bromocopper(1+) Chemical compound Br[Cu+] ODWXUNBKCRECNW-UHFFFAOYSA-M 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 239000000480 calcium channel blocker Substances 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- 230000006315 carbonylation Effects 0.000 description 1
- 238000005810 carbonylation reaction Methods 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 125000002057 carboxymethyl group Chemical group [H]OC(=O)C([H])([H])[*] 0.000 description 1
- 230000007670 carcinogenicity Effects 0.000 description 1
- 231100000260 carcinogenicity Toxicity 0.000 description 1
- 231100000259 cardiotoxicity Toxicity 0.000 description 1
- 229950008138 carmellose Drugs 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- QYIYFLOTGYLRGG-GPCCPHFNSA-N cefaclor Chemical compound C1([C@H](C(=O)N[C@@H]2C(N3C(=C(Cl)CS[C@@H]32)C(O)=O)=O)N)=CC=CC=C1 QYIYFLOTGYLRGG-GPCCPHFNSA-N 0.000 description 1
- 229960005361 cefaclor Drugs 0.000 description 1
- OKBVVJOGVLARMR-QSWIMTSFSA-N cefixime Chemical compound S1C(N)=NC(C(=N\OCC(O)=O)\C(=O)N[C@@H]2C(N3C(=C(C=C)CS[C@@H]32)C(O)=O)=O)=C1 OKBVVJOGVLARMR-QSWIMTSFSA-N 0.000 description 1
- 229960002129 cefixime Drugs 0.000 description 1
- RZEKVGVHFLEQIL-UHFFFAOYSA-N celecoxib Chemical compound C1=CC(C)=CC=C1C1=CC(C(F)(F)F)=NN1C1=CC=C(S(N)(=O)=O)C=C1 RZEKVGVHFLEQIL-UHFFFAOYSA-N 0.000 description 1
- 229960000590 celecoxib Drugs 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 208000026106 cerebrovascular disease Diseases 0.000 description 1
- UKTAZPQNNNJVKR-KJGYPYNMSA-N chembl2368925 Chemical compound C1=CC=C2C(C(O[C@@H]3C[C@@H]4C[C@H]5C[C@@H](N4CC5=O)C3)=O)=CNC2=C1 UKTAZPQNNNJVKR-KJGYPYNMSA-N 0.000 description 1
- BWWVAEOLVKTZFQ-ISVUSNJMSA-N chembl530 Chemical compound N(/[C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)=C\N1CCCCCC1 BWWVAEOLVKTZFQ-ISVUSNJMSA-N 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- ANTSCNMPPGJYLG-UHFFFAOYSA-N chlordiazepoxide Chemical compound O=N=1CC(NC)=NC2=CC=C(Cl)C=C2C=1C1=CC=CC=C1 ANTSCNMPPGJYLG-UHFFFAOYSA-N 0.000 description 1
- 229960004782 chlordiazepoxide Drugs 0.000 description 1
- 229960004926 chlorobutanol Drugs 0.000 description 1
- QQVDYSUDFZZPSU-UHFFFAOYSA-M chloromethylidene(dimethyl)azanium;chloride Chemical compound [Cl-].C[N+](C)=CCl QQVDYSUDFZZPSU-UHFFFAOYSA-M 0.000 description 1
- JIVPVXMEBJLZRO-UHFFFAOYSA-N chlorthalidone Chemical compound C1=C(Cl)C(S(=O)(=O)N)=CC(C2(O)C3=CC=CC=C3C(=O)N2)=C1 JIVPVXMEBJLZRO-UHFFFAOYSA-N 0.000 description 1
- 235000012000 cholesterol Nutrition 0.000 description 1
- 230000007665 chronic toxicity Effects 0.000 description 1
- 231100000160 chronic toxicity Toxicity 0.000 description 1
- 229960001380 cimetidine Drugs 0.000 description 1
- AQIXAKUUQRKLND-UHFFFAOYSA-N cimetidine Chemical compound N#C/N=C(/NC)NCCSCC=1N=CNC=1C AQIXAKUUQRKLND-UHFFFAOYSA-N 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 239000011247 coating layer Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- FCFNRCROJUBPLU-UHFFFAOYSA-N compound M126 Natural products CC(C)C1NC(=O)C(C)OC(=O)C(C(C)C)NC(=O)C(C(C)C)OC(=O)C(C(C)C)NC(=O)C(C)OC(=O)C(C(C)C)NC(=O)C(C(C)C)OC(=O)C(C(C)C)NC(=O)C(C)OC(=O)C(C(C)C)NC(=O)C(C(C)C)OC1=O FCFNRCROJUBPLU-UHFFFAOYSA-N 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- GBRBMTNGQBKBQE-UHFFFAOYSA-L copper;diiodide Chemical compound I[Cu]I GBRBMTNGQBKBQE-UHFFFAOYSA-L 0.000 description 1
- 235000005687 corn oil Nutrition 0.000 description 1
- 239000002285 corn oil Substances 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- LDHQCZJRKDOVOX-NSCUHMNNSA-N crotonic acid Chemical compound C\C=C\C(O)=O LDHQCZJRKDOVOX-NSCUHMNNSA-N 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 1
- LEVWYRKDKASIDU-IMJSIDKUSA-N cystine group Chemical group C([C@@H](C(=O)O)N)SSC[C@@H](C(=O)O)N LEVWYRKDKASIDU-IMJSIDKUSA-N 0.000 description 1
- 229950007605 dapitant Drugs 0.000 description 1
- 235000019258 dehydroacetic acid Nutrition 0.000 description 1
- 229940061632 dehydroacetic acid Drugs 0.000 description 1
- JEQRBTDTEKWZBW-UHFFFAOYSA-N dehydroacetic acid Chemical compound CC(=O)C1=C(O)OC(C)=CC1=O JEQRBTDTEKWZBW-UHFFFAOYSA-N 0.000 description 1
- PGRHXDWITVMQBC-UHFFFAOYSA-N dehydroacetic acid Natural products CC(=O)C1C(=O)OC(C)=CC1=O PGRHXDWITVMQBC-UHFFFAOYSA-N 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- GDVKFRBCXAPAQJ-UHFFFAOYSA-A dialuminum;hexamagnesium;carbonate;hexadecahydroxide Chemical compound [OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[OH-].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Mg+2].[Al+3].[Al+3].[O-]C([O-])=O GDVKFRBCXAPAQJ-UHFFFAOYSA-A 0.000 description 1
- 229940111205 diastase Drugs 0.000 description 1
- 229960001259 diclofenac Drugs 0.000 description 1
- DCOPUUMXTXDBNB-UHFFFAOYSA-N diclofenac Chemical compound OC(=O)CC1=CC=CC=C1NC1=C(Cl)C=CC=C1Cl DCOPUUMXTXDBNB-UHFFFAOYSA-N 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 235000015872 dietary supplement Nutrition 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 230000001079 digestive effect Effects 0.000 description 1
- IJKVHSBPTUYDLN-UHFFFAOYSA-N dihydroxy(oxo)silane Chemical compound O[Si](O)=O IJKVHSBPTUYDLN-UHFFFAOYSA-N 0.000 description 1
- HSUGRBWQSSZJOP-RTWAWAEBSA-N diltiazem Chemical compound C1=CC(OC)=CC=C1[C@H]1[C@@H](OC(C)=O)C(=O)N(CCN(C)C)C2=CC=CC=C2S1 HSUGRBWQSSZJOP-RTWAWAEBSA-N 0.000 description 1
- 229960004166 diltiazem Drugs 0.000 description 1
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 1
- KCIDZIIHRGYJAE-YGFYJFDDSA-L dipotassium;[(2r,3r,4s,5r,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl] phosphate Chemical compound [K+].[K+].OC[C@H]1O[C@H](OP([O-])([O-])=O)[C@H](O)[C@@H](O)[C@H]1O KCIDZIIHRGYJAE-YGFYJFDDSA-L 0.000 description 1
- BNIILDVGGAEEIG-UHFFFAOYSA-L disodium hydrogen phosphate Chemical compound [Na+].[Na+].OP([O-])([O-])=O BNIILDVGGAEEIG-UHFFFAOYSA-L 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 229960003413 dolasetron Drugs 0.000 description 1
- 239000003210 dopamine receptor blocking agent Substances 0.000 description 1
- 239000008298 dragée Substances 0.000 description 1
- 230000000857 drug effect Effects 0.000 description 1
- 230000008406 drug-drug interaction Effects 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 229950003102 efonidipine Drugs 0.000 description 1
- 239000012055 enteric layer Substances 0.000 description 1
- 235000019414 erythritol Nutrition 0.000 description 1
- UNXHWFMMPAWVPI-ZXZARUISSA-N erythritol Chemical compound OC[C@H](O)[C@H](O)CO UNXHWFMMPAWVPI-ZXZARUISSA-N 0.000 description 1
- 229940009714 erythritol Drugs 0.000 description 1
- SUBDBMMJDZJVOS-DEOSSOPVSA-N esomeprazole Chemical compound C([S@](=O)C1=NC2=CC=C(C=C2N1)OC)C1=NC=C(C)C(OC)=C1C SUBDBMMJDZJVOS-DEOSSOPVSA-N 0.000 description 1
- 229960004770 esomeprazole Drugs 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- WHQLQYRFIHPMNA-UHFFFAOYSA-N ethyl acetate;oxolane Chemical compound C1CCOC1.CCOC(C)=O WHQLQYRFIHPMNA-UHFFFAOYSA-N 0.000 description 1
- 125000004494 ethyl ester group Chemical group 0.000 description 1
- 125000000031 ethylamino group Chemical group [H]C([H])([H])C([H])([H])N([H])[*] 0.000 description 1
- XFBVBWWRPKNWHW-UHFFFAOYSA-N etodolac Chemical compound C1COC(CC)(CC(O)=O)C2=N[C]3C(CC)=CC=CC3=C21 XFBVBWWRPKNWHW-UHFFFAOYSA-N 0.000 description 1
- 229960005293 etodolac Drugs 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000010408 film Substances 0.000 description 1
- 239000007941 film coated tablet Substances 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000004508 fractional distillation Methods 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 229960000457 gallopamil Drugs 0.000 description 1
- 210000001156 gastric mucosa Anatomy 0.000 description 1
- 201000005917 gastric ulcer Diseases 0.000 description 1
- 229940045140 gaviscon Drugs 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 231100000025 genetic toxicology Toxicity 0.000 description 1
- 230000001738 genotoxic effect Effects 0.000 description 1
- 229960002518 gentamicin Drugs 0.000 description 1
- GNKDKYIHGQKHHM-RJKLHVOGSA-N ghrelin Chemical compound C([C@H](NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)CN)COC(=O)CCCCCCC)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1N=CNC=1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C1=CC=CC=C1 GNKDKYIHGQKHHM-RJKLHVOGSA-N 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- 239000001087 glyceryl triacetate Substances 0.000 description 1
- 235000013773 glyceryl triacetate Nutrition 0.000 description 1
- LPLVUJXQOOQHMX-UHFFFAOYSA-N glycyrrhetinic acid glycoside Natural products C1CC(C2C(C3(CCC4(C)CCC(C)(CC4C3=CC2=O)C(O)=O)C)(C)CC2)(C)C2C(C)(C)C1OC1OC(C(O)=O)C(O)C(O)C1OC1OC(C(O)=O)C(O)C(O)C1O LPLVUJXQOOQHMX-UHFFFAOYSA-N 0.000 description 1
- UYRUBYNTXSDKQT-UHFFFAOYSA-N glycyrrhizic acid Natural products CC1(C)C(CCC2(C)C1CCC3(C)C2C(=O)C=C4C5CC(C)(CCC5(C)CCC34C)C(=O)O)OC6OC(C(O)C(O)C6OC7OC(O)C(O)C(O)C7C(=O)O)C(=O)O UYRUBYNTXSDKQT-UHFFFAOYSA-N 0.000 description 1
- 229960004949 glycyrrhizic acid Drugs 0.000 description 1
- 235000019410 glycyrrhizin Nutrition 0.000 description 1
- LPLVUJXQOOQHMX-QWBHMCJMSA-N glycyrrhizinic acid Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@H](O[C@@H]1O[C@@H]1C([C@H]2[C@]([C@@H]3[C@@]([C@@]4(CC[C@@]5(C)CC[C@@](C)(C[C@H]5C4=CC3=O)C(O)=O)C)(C)CC2)(C)CC1)(C)C)C(O)=O)[C@@H]1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O LPLVUJXQOOQHMX-QWBHMCJMSA-N 0.000 description 1
- MFWNKCLOYSRHCJ-BTTYYORXSA-N granisetron Chemical compound C1=CC=C2C(C(=O)N[C@H]3C[C@H]4CCC[C@@H](C3)N4C)=NN(C)C2=C1 MFWNKCLOYSRHCJ-BTTYYORXSA-N 0.000 description 1
- 229960003727 granisetron Drugs 0.000 description 1
- 239000000665 guar gum Substances 0.000 description 1
- 235000010417 guar gum Nutrition 0.000 description 1
- 229960002154 guar gum Drugs 0.000 description 1
- 125000003707 hexyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 229920001477 hydrophilic polymer Polymers 0.000 description 1
- 229960001545 hydrotalcite Drugs 0.000 description 1
- 229910001701 hydrotalcite Inorganic materials 0.000 description 1
- 230000033444 hydroxylation Effects 0.000 description 1
- 238000005805 hydroxylation reaction Methods 0.000 description 1
- 229920003063 hydroxymethyl cellulose Polymers 0.000 description 1
- 229940031574 hydroxymethyl cellulose Drugs 0.000 description 1
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 description 1
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 description 1
- UWNADWZGEHDQAB-UHFFFAOYSA-N i-Pr2C2H4i-Pr2 Natural products CC(C)CCC(C)C UWNADWZGEHDQAB-UHFFFAOYSA-N 0.000 description 1
- 229960001680 ibuprofen Drugs 0.000 description 1
- 125000005946 imidazo[1,2-a]pyridyl group Chemical group 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- ZSKVGTPCRGIANV-ZXFLCMHBSA-N imipenem Chemical compound C1C(SCC\N=C\N)=C(C(O)=O)N2C(=O)[C@H]([C@H](O)C)[C@H]21 ZSKVGTPCRGIANV-ZXFLCMHBSA-N 0.000 description 1
- 229960002182 imipenem Drugs 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 229960000905 indomethacin Drugs 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000002198 insoluble material Substances 0.000 description 1
- 210000002011 intestinal secretion Anatomy 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000000555 isopropenyl group Chemical group [H]\C([H])=C(\*)C([H])([H])[H] 0.000 description 1
- 125000003965 isoxazolidinyl group Chemical group 0.000 description 1
- 229960004427 isradipine Drugs 0.000 description 1
- 229960005302 itopride Drugs 0.000 description 1
- 229950001743 itriglumide Drugs 0.000 description 1
- 229940095358 konsyl Drugs 0.000 description 1
- 229960004340 lacidipine Drugs 0.000 description 1
- GKQPCPXONLDCMU-CCEZHUSRSA-N lacidipine Chemical compound CCOC(=O)C1=C(C)NC(C)=C(C(=O)OCC)C1C1=CC=CC=C1\C=C\C(=O)OC(C)(C)C GKQPCPXONLDCMU-CCEZHUSRSA-N 0.000 description 1
- 229960003303 lafutidine Drugs 0.000 description 1
- 229950005286 lanepitant Drugs 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 229960004294 lercanidipine Drugs 0.000 description 1
- ZDXUKAKRHYTAKV-UHFFFAOYSA-N lercanidipine Chemical compound COC(=O)C1=C(C)NC(C)=C(C(=O)OC(C)(C)CN(C)CCC(C=2C=CC=CC=2)C=2C=CC=CC=2)C1C1=CC=CC([N+]([O-])=O)=C1 ZDXUKAKRHYTAKV-UHFFFAOYSA-N 0.000 description 1
- 231100001231 less toxic Toxicity 0.000 description 1
- 239000004571 lime Substances 0.000 description 1
- 229950007692 lomerizine Drugs 0.000 description 1
- 230000005923 long-lasting effect Effects 0.000 description 1
- 229940099076 maalox Drugs 0.000 description 1
- 229960003511 macrogol Drugs 0.000 description 1
- 229940041033 macrolides Drugs 0.000 description 1
- HCWCAKKEBCNQJP-UHFFFAOYSA-N magnesium orthosilicate Chemical compound [Mg+2].[Mg+2].[O-][Si]([O-])([O-])[O-] HCWCAKKEBCNQJP-UHFFFAOYSA-N 0.000 description 1
- 239000000391 magnesium silicate Substances 0.000 description 1
- 229910052919 magnesium silicate Inorganic materials 0.000 description 1
- 235000019792 magnesium silicate Nutrition 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 235000010449 maltitol Nutrition 0.000 description 1
- 239000000845 maltitol Substances 0.000 description 1
- VQHSOMBJVWLPSR-WUJBLJFYSA-N maltitol Chemical compound OC[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O VQHSOMBJVWLPSR-WUJBLJFYSA-N 0.000 description 1
- 229940035436 maltitol Drugs 0.000 description 1
- 229960003963 manidipine Drugs 0.000 description 1
- ANEBWFXPVPTEET-UHFFFAOYSA-N manidipine Chemical compound COC(=O)C1=C(C)NC(C)=C(C(=O)OCCN2CCN(CC2)C(C=2C=CC=CC=2)C=2C=CC=CC=2)C1C1=CC=CC([N+]([O-])=O)=C1 ANEBWFXPVPTEET-UHFFFAOYSA-N 0.000 description 1
- 229960001855 mannitol Drugs 0.000 description 1
- 230000000873 masking effect Effects 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 229960003464 mefenamic acid Drugs 0.000 description 1
- 229940041616 menthol Drugs 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229910000000 metal hydroxide Inorganic materials 0.000 description 1
- 150000004692 metal hydroxides Chemical class 0.000 description 1
- 229940117841 methacrylic acid copolymer Drugs 0.000 description 1
- WVJKHCGMRZGIJH-UHFFFAOYSA-N methanetriamine Chemical compound NC(N)N WVJKHCGMRZGIJH-UHFFFAOYSA-N 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- 238000006063 methoxycarbonylation reaction Methods 0.000 description 1
- WLBNVSIQCFHAQB-UHFFFAOYSA-N methyl 1h-pyrrole-3-carboxylate Chemical compound COC(=O)C=1C=CNC=1 WLBNVSIQCFHAQB-UHFFFAOYSA-N 0.000 description 1
- VKQFCGNPDRICFG-UHFFFAOYSA-N methyl 2-methylpropyl 2,6-dimethyl-4-(2-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylate Chemical compound COC(=O)C1=C(C)NC(C)=C(C(=O)OCC(C)C)C1C1=CC=CC=C1[N+]([O-])=O VKQFCGNPDRICFG-UHFFFAOYSA-N 0.000 description 1
- 125000006533 methyl amino methyl group Chemical group [H]N(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- LQEXPHILFYMEJR-UHFFFAOYSA-N methylazanium dichloride Chemical compound Cl.Cl.NC.NC LQEXPHILFYMEJR-UHFFFAOYSA-N 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 125000002816 methylsulfanyl group Chemical group [H]C([H])([H])S[*] 0.000 description 1
- TTWJBBZEZQICBI-UHFFFAOYSA-N metoclopramide Chemical compound CCN(CC)CCNC(=O)C1=CC(Cl)=C(N)C=C1OC TTWJBBZEZQICBI-UHFFFAOYSA-N 0.000 description 1
- 229960004503 metoclopramide Drugs 0.000 description 1
- 229960000282 metronidazole Drugs 0.000 description 1
- VAOCPAMSLUNLGC-UHFFFAOYSA-N metronidazole Chemical compound CC1=NC=C([N+]([O-])=O)N1CCO VAOCPAMSLUNLGC-UHFFFAOYSA-N 0.000 description 1
- 229960002509 miconazole Drugs 0.000 description 1
- 230000003228 microsomal effect Effects 0.000 description 1
- 229960004023 minocycline Drugs 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 125000005322 morpholin-1-yl group Chemical group 0.000 description 1
- RPAWVEMNAJPPEL-UHFFFAOYSA-N morpholine;thiomorpholine Chemical compound C1COCCN1.C1CSCCN1 RPAWVEMNAJPPEL-UHFFFAOYSA-N 0.000 description 1
- 208000029744 multiple organ dysfunction syndrome Diseases 0.000 description 1
- PSHKMPUSSFXUIA-UHFFFAOYSA-N n,n-dimethylpyridin-2-amine Chemical compound CN(C)C1=CC=CC=N1 PSHKMPUSSFXUIA-UHFFFAOYSA-N 0.000 description 1
- CVXJAPZTZWLRBP-MUUNZHRXSA-N n-[(2r)-1-[acetyl-[(2-methoxyphenyl)methyl]amino]-3-(1h-indol-3-yl)propan-2-yl]-2-(4-piperidin-1-ylpiperidin-1-yl)acetamide Chemical compound COC1=CC=CC=C1CN(C(C)=O)C[C@H](NC(=O)CN1CCC(CC1)N1CCCCC1)CC1=CNC2=CC=CC=C12 CVXJAPZTZWLRBP-MUUNZHRXSA-N 0.000 description 1
- YUTIXVXZQIQWGY-UHFFFAOYSA-N n-[4-[6-[4-(trifluoromethyl)phenyl]pyrimidin-4-yl]oxy-1,3-benzothiazol-2-yl]acetamide Chemical compound C1=CC=C2SC(NC(=O)C)=NC2=C1OC(N=CN=1)=CC=1C1=CC=C(C(F)(F)F)C=C1 YUTIXVXZQIQWGY-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000005146 naphthylsulfonyl group Chemical group C1(=CC=CC2=CC=CC=C12)S(=O)(=O)* 0.000 description 1
- 125000005029 naphthylthio group Chemical group C1(=CC=CC2=CC=CC=C12)S* 0.000 description 1
- CMWTZPSULFXXJA-VIFPVBQESA-N naproxen Chemical group C1=C([C@H](C)C(O)=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-VIFPVBQESA-N 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 230000001613 neoplastic effect Effects 0.000 description 1
- 229950000640 nepadutant Drugs 0.000 description 1
- 229960001783 nicardipine Drugs 0.000 description 1
- 229960001597 nifedipine Drugs 0.000 description 1
- HYIMSNHJOBLJNT-UHFFFAOYSA-N nifedipine Chemical compound COC(=O)C1=C(C)NC(C)=C(C(=O)OC)C1C1=CC=CC=C1[N+]([O-])=O HYIMSNHJOBLJNT-UHFFFAOYSA-N 0.000 description 1
- 229960005366 nilvadipine Drugs 0.000 description 1
- 229960000227 nisoldipine Drugs 0.000 description 1
- 229960005425 nitrendipine Drugs 0.000 description 1
- 239000000236 nitric oxide synthase inhibitor Substances 0.000 description 1
- RGHXWDVNBYKJQH-UHFFFAOYSA-N nitroacetic acid Chemical compound OC(=O)C[N+]([O-])=O RGHXWDVNBYKJQH-UHFFFAOYSA-N 0.000 description 1
- MCSAJNNLRCFZED-UHFFFAOYSA-N nitroethane Chemical compound CC[N+]([O-])=O MCSAJNNLRCFZED-UHFFFAOYSA-N 0.000 description 1
- DLWSRGHNJVLJAH-UHFFFAOYSA-N nitroflurbiprofen Chemical compound FC1=CC(C(C(=O)OCCCCO[N+]([O-])=O)C)=CC=C1C1=CC=CC=C1 DLWSRGHNJVLJAH-UHFFFAOYSA-N 0.000 description 1
- SGXXNSQHWDMGGP-IZZDOVSWSA-N nizatidine Chemical compound [O-][N+](=O)\C=C(/NC)NCCSCC1=CSC(CN(C)C)=N1 SGXXNSQHWDMGGP-IZZDOVSWSA-N 0.000 description 1
- 229960004872 nizatidine Drugs 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 230000005311 nuclear magnetism Effects 0.000 description 1
- 125000003261 o-tolyl group Chemical group [H]C1=C([H])C(*)=C(C([H])=C1[H])C([H])([H])[H] 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 229960001699 ofloxacin Drugs 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 125000005740 oxycarbonyl group Chemical group [*:1]OC([*:2])=O 0.000 description 1
- 125000000636 p-nitrophenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)[N+]([O-])=O 0.000 description 1
- 125000001037 p-tolyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 1
- CPZBLNMUGSZIPR-NVXWUHKLSA-N palonosetron Chemical compound C1N(CC2)CCC2[C@@H]1N1C(=O)C(C=CC=C2CCC3)=C2[C@H]3C1 CPZBLNMUGSZIPR-NVXWUHKLSA-N 0.000 description 1
- 229960002131 palonosetron Drugs 0.000 description 1
- 229960005019 pantoprazole Drugs 0.000 description 1
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 1
- 235000019371 penicillin G benzathine Nutrition 0.000 description 1
- 229940056360 penicillin g Drugs 0.000 description 1
- 125000004894 pentylamino group Chemical group C(CCCC)N* 0.000 description 1
- 125000005981 pentynyl group Chemical group 0.000 description 1
- 230000001175 peptic effect Effects 0.000 description 1
- 238000011170 pharmaceutical development Methods 0.000 description 1
- LIGACIXOYTUXAW-UHFFFAOYSA-N phenacyl bromide Chemical compound BrCC(=O)C1=CC=CC=C1 LIGACIXOYTUXAW-UHFFFAOYSA-N 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N phenylbenzene Natural products C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 1
- 125000004344 phenylpropyl group Chemical group 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- ACVYVLVWPXVTIT-UHFFFAOYSA-N phosphinic acid Chemical compound O[PH2]=O ACVYVLVWPXVTIT-UHFFFAOYSA-N 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 125000005633 phthalidyl group Chemical group 0.000 description 1
- 229960002292 piperacillin Drugs 0.000 description 1
- WCMIIGXFCMNQDS-IDYPWDAWSA-M piperacillin sodium Chemical compound [Na+].O=C1C(=O)N(CC)CCN1C(=O)N[C@H](C=1C=CC=CC=1)C(=O)N[C@@H]1C(=O)N2[C@@H](C([O-])=O)C(C)(C)S[C@@H]21 WCMIIGXFCMNQDS-IDYPWDAWSA-M 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 150000003053 piperidines Chemical class 0.000 description 1
- QYSPLQLAKJAUJT-UHFFFAOYSA-N piroxicam Chemical compound OC=1C2=CC=CC=C2S(=O)(=O)N(C)C=1C(=O)NC1=CC=CC=N1 QYSPLQLAKJAUJT-UHFFFAOYSA-N 0.000 description 1
- 229960002702 piroxicam Drugs 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- SUWYPNNPLSRNPS-UNTSEYQFSA-N plaunotol Chemical compound CC(C)=CCC\C(C)=C\CC\C(CO)=C\CC\C(C)=C\CO SUWYPNNPLSRNPS-UNTSEYQFSA-N 0.000 description 1
- SUWYPNNPLSRNPS-UHFFFAOYSA-N plaunotol Natural products CC(C)=CCCC(C)=CCCC(CO)=CCCC(C)=CCO SUWYPNNPLSRNPS-UHFFFAOYSA-N 0.000 description 1
- 229950009291 plaunotol Drugs 0.000 description 1
- 229950004693 polaprezinc Drugs 0.000 description 1
- 108700035912 polaprezinc Proteins 0.000 description 1
- 229920001495 poly(sodium acrylate) polymer Polymers 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 229940068984 polyvinyl alcohol Drugs 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 1
- 239000011736 potassium bicarbonate Substances 0.000 description 1
- 235000015497 potassium bicarbonate Nutrition 0.000 description 1
- 229910000028 potassium bicarbonate Inorganic materials 0.000 description 1
- CHKVPAROMQMJNQ-UHFFFAOYSA-M potassium bisulfate Chemical compound [K+].OS([O-])(=O)=O CHKVPAROMQMJNQ-UHFFFAOYSA-M 0.000 description 1
- 229910000343 potassium bisulfate Inorganic materials 0.000 description 1
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 1
- 229940086066 potassium hydrogencarbonate Drugs 0.000 description 1
- FYRHIOVKTDQVFC-UHFFFAOYSA-M potassium phthalimide Chemical compound [K+].C1=CC=C2C(=O)[N-]C(=O)C2=C1 FYRHIOVKTDQVFC-UHFFFAOYSA-M 0.000 description 1
- 159000000001 potassium salts Chemical class 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 229940088417 precipitated calcium carbonate Drugs 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 239000001294 propane Substances 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 125000004742 propyloxycarbonyl group Chemical group 0.000 description 1
- 239000003223 protective agent Substances 0.000 description 1
- 235000019423 pullulan Nutrition 0.000 description 1
- DXKXZXHDDYMNSU-UHFFFAOYSA-N pyrazole-1-carbaldehyde Chemical compound O=CN1C=CC=N1 DXKXZXHDDYMNSU-UHFFFAOYSA-N 0.000 description 1
- CDRNYKLYADJTMN-UHFFFAOYSA-N pyridine-3-sulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CN=C1 CDRNYKLYADJTMN-UHFFFAOYSA-N 0.000 description 1
- UDJFFSGCRRMVFH-UHFFFAOYSA-N pyrido[2,3-d]pyrimidine Chemical compound N1=CN=CC2=CC=CN=C21 UDJFFSGCRRMVFH-UHFFFAOYSA-N 0.000 description 1
- WQGWDDDVZFFDIG-UHFFFAOYSA-N pyrogallol Chemical compound OC1=CC=CC(O)=C1O WQGWDDDVZFFDIG-UHFFFAOYSA-N 0.000 description 1
- HNJBEVLQSNELDL-UHFFFAOYSA-N pyrrolidin-2-one Chemical compound O=C1CCCN1 HNJBEVLQSNELDL-UHFFFAOYSA-N 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 150000007660 quinolones Chemical class 0.000 description 1
- 229960004157 rabeprazole Drugs 0.000 description 1
- YREYEVIYCVEVJK-UHFFFAOYSA-N rabeprazole Chemical compound COCCCOC1=CC=NC(CS(=O)C=2NC3=CC=CC=C3N=2)=C1C YREYEVIYCVEVJK-UHFFFAOYSA-N 0.000 description 1
- NTHPAPBPFQJABD-LLVKDONJSA-N ramosetron Chemical compound C12=CC=CC=C2N(C)C=C1C(=O)[C@H]1CC(NC=N2)=C2CC1 NTHPAPBPFQJABD-LLVKDONJSA-N 0.000 description 1
- 229950001588 ramosetron Drugs 0.000 description 1
- VMXUWOKSQNHOCA-LCYFTJDESA-N ranitidine Chemical compound [O-][N+](=O)/C=C(/NC)NCCSCC1=CC=C(CN(C)C)O1 VMXUWOKSQNHOCA-LCYFTJDESA-N 0.000 description 1
- 229960000620 ranitidine Drugs 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 229950004535 rebamipide Drugs 0.000 description 1
- HELXLJCILKEWJH-NCGAPWICSA-N rebaudioside A Chemical compound O([C@H]1[C@H](O)[C@@H](CO)O[C@H]([C@@H]1O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O HELXLJCILKEWJH-NCGAPWICSA-N 0.000 description 1
- 238000006268 reductive amination reaction Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 231100000372 reproductive toxicity Toxicity 0.000 description 1
- 230000007696 reproductive toxicity Effects 0.000 description 1
- WBISVCLTLBMTDS-UHFFFAOYSA-N s-phenyl ethanethioate Chemical compound CC(=O)SC1=CC=CC=C1 WBISVCLTLBMTDS-UHFFFAOYSA-N 0.000 description 1
- 229940085605 saccharin sodium Drugs 0.000 description 1
- 239000012266 salt solution Substances 0.000 description 1
- 125000003548 sec-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000000580 secretagogue effect Effects 0.000 description 1
- 230000001624 sedative effect Effects 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 239000004208 shellac Substances 0.000 description 1
- ZLGIYFNHBLSMPS-ATJNOEHPSA-N shellac Chemical compound OCCCCCC(O)C(O)CCCCCCCC(O)=O.C1C23[C@H](C(O)=O)CCC2[C@](C)(CO)[C@@H]1C(C(O)=O)=C[C@@H]3O ZLGIYFNHBLSMPS-ATJNOEHPSA-N 0.000 description 1
- 229940113147 shellac Drugs 0.000 description 1
- 235000013874 shellac Nutrition 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 239000004317 sodium nitrate Substances 0.000 description 1
- 235000010344 sodium nitrate Nutrition 0.000 description 1
- NNMHYFLPFNGQFZ-UHFFFAOYSA-M sodium polyacrylate Chemical compound [Na+].[O-]C(=O)C=C NNMHYFLPFNGQFZ-UHFFFAOYSA-M 0.000 description 1
- PODWXQQNRWNDGD-UHFFFAOYSA-L sodium thiosulfate pentahydrate Chemical compound O.O.O.O.O.[Na+].[Na+].[O-]S([S-])(=O)=O PODWXQQNRWNDGD-UHFFFAOYSA-L 0.000 description 1
- 239000004071 soot Substances 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 235000010356 sorbitol Nutrition 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- MNQYNQBOVCBZIQ-JQOFMKNESA-A sucralfate Chemical compound O[Al](O)OS(=O)(=O)O[C@@H]1[C@@H](OS(=O)(=O)O[Al](O)O)[C@H](OS(=O)(=O)O[Al](O)O)[C@@H](COS(=O)(=O)O[Al](O)O)O[C@H]1O[C@@]1(COS(=O)(=O)O[Al](O)O)[C@@H](OS(=O)(=O)O[Al](O)O)[C@H](OS(=O)(=O)O[Al](O)O)[C@@H](OS(=O)(=O)O[Al](O)O)O1 MNQYNQBOVCBZIQ-JQOFMKNESA-A 0.000 description 1
- 229960004291 sucralfate Drugs 0.000 description 1
- 150000005846 sugar alcohols Chemical class 0.000 description 1
- 239000007940 sugar coated tablet Substances 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 239000008399 tap water Substances 0.000 description 1
- 235000020679 tap water Nutrition 0.000 description 1
- 229950008375 tenatoprazole Drugs 0.000 description 1
- 229960002180 tetracycline Drugs 0.000 description 1
- 229930101283 tetracycline Natural products 0.000 description 1
- 235000019364 tetracycline Nutrition 0.000 description 1
- 229940072172 tetracycline antibiotic Drugs 0.000 description 1
- 150000003522 tetracyclines Chemical class 0.000 description 1
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical compound C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 239000000892 thaumatin Substances 0.000 description 1
- 235000010436 thaumatin Nutrition 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 150000007979 thiazole derivatives Chemical class 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- CFOAUYCPAUGDFF-UHFFFAOYSA-N tosmic Chemical compound CC1=CC=C(S(=O)(=O)C[N+]#[C-])C=C1 CFOAUYCPAUGDFF-UHFFFAOYSA-N 0.000 description 1
- LDHQCZJRKDOVOX-UHFFFAOYSA-N trans-crotonic acid Natural products CC=CC(O)=O LDHQCZJRKDOVOX-UHFFFAOYSA-N 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 229960002622 triacetin Drugs 0.000 description 1
- 239000001069 triethyl citrate Substances 0.000 description 1
- VMYFZRTXGLUXMZ-UHFFFAOYSA-N triethyl citrate Natural products CCOC(=O)C(O)(C(=O)OCC)C(=O)OCC VMYFZRTXGLUXMZ-UHFFFAOYSA-N 0.000 description 1
- 235000013769 triethyl citrate Nutrition 0.000 description 1
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 description 1
- UJMBCXLDXJUMFB-UHFFFAOYSA-K trisodium;5-oxo-1-(4-sulfonatophenyl)-4-[(4-sulfonatophenyl)diazenyl]-4h-pyrazole-3-carboxylate Chemical compound [Na+].[Na+].[Na+].[O-]C(=O)C1=NN(C=2C=CC(=CC=2)S([O-])(=O)=O)C(=O)C1N=NC1=CC=C(S([O-])(=O)=O)C=C1 UJMBCXLDXJUMFB-UHFFFAOYSA-K 0.000 description 1
- 210000002438 upper gastrointestinal tract Anatomy 0.000 description 1
- 229960005486 vaccine Drugs 0.000 description 1
- FCFNRCROJUBPLU-DNDCDFAISA-N valinomycin Chemical compound CC(C)[C@@H]1NC(=O)[C@H](C)OC(=O)[C@@H](C(C)C)NC(=O)[C@@H](C(C)C)OC(=O)[C@H](C(C)C)NC(=O)[C@H](C)OC(=O)[C@@H](C(C)C)NC(=O)[C@@H](C(C)C)OC(=O)[C@H](C(C)C)NC(=O)[C@H](C)OC(=O)[C@@H](C(C)C)NC(=O)[C@@H](C(C)C)OC1=O FCFNRCROJUBPLU-DNDCDFAISA-N 0.000 description 1
- 229960001722 verapamil Drugs 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 239000008215 water for injection Substances 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/30—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D207/34—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D207/36—Oxygen or sulfur atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/415—1,2-Diazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/4164—1,3-Diazoles
- A61K31/417—Imidazole-alkylamines, e.g. histamine, phentolamine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/4164—1,3-Diazoles
- A61K31/4178—1,3-Diazoles not condensed 1,3-diazoles and containing further heterocyclic rings, e.g. pilocarpine, nitrofurantoin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/426—1,3-Thiazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/4439—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. omeprazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/06—Anti-spasmodics, e.g. drugs for colics, esophagic dyskinesia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/12—Antidiarrhoeals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/04—Antihaemorrhagics; Procoagulants; Haemostatic agents; Antifibrinolytic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/12—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/64—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with substituted hydrocarbon radicals attached to ring carbon atoms, e.g. histidine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/66—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D233/84—Sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/02—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings
- C07D277/20—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D277/32—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D277/36—Sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
Definitions
- the present invention relates to a compound having an acid secretion inhibitory activity.
- proton pump inhibitors represented by omebrazole that suppress the secretion of gastric acid are widely used in clinical settings.
- existing proton pump inhibitors have problems in terms of effects and side effects.
- existing proton pump inhibitors are unstable under acidic conditions and are therefore often formulated as enteric preparations. In such cases, it takes several hours to develop the action.
- existing proton pump inhibitors are concerned about drug-drug interactions with drugs such as diazepam, which vary in therapeutic effects based on metabolic enzyme polymorphism, and improvements are desired.
- Patent Document 1 describes a thiazole derivative as a compound having a proton pump inhibitory action.
- Patent Document 2 discloses a compound having the formula
- rl is carboxy, protected carboxy, carboxy (lower) alkyl, protected carboxy (lower) alkyl, carboxy (lower) alkenyl or protected carboxy (lower) alkenyl
- r2 is hydrogen; Lower alkyl; having aminoimino or protected aminoimino, or may be heterocyclic (lower) alkyl; heterocyclic (lower) alkenyl; or heterocyclic carbonyl
- r3 is hydrogen or lower alkyl
- r4 is asil
- r5 is hydrogen
- A lower alkylene
- Z is S or NH.
- rl is carboxy, protected carboxy, carboxy (lower) alkyl, protected carboxy (lower) alkyl, carboxy (lower) alkenyl or protected carboxy (lower) alkenyl
- r3 is hydrogen or lower alkyl
- r4 is asil
- r5 is hydrogen
- A is lower alkylene
- Z is S or NH
- Patent Document 3 discloses a formula.
- Patent Document 4 discloses a pyridyl or imidazolyl derivative having a protein “isoprenyl” transferase inhibitory action.
- Patent Document 1 EP-A-0259085
- Patent Document 2 JP-A-8-119936
- Patent Document 3 International Publication No. 2004/103968 Pamphlet
- Patent Document 4 US2002 / 0193596
- an object of the present invention is to provide a compound having an excellent acid secretion inhibitory action (particularly a proton pump inhibitory action) that has improved these problems.
- ring A may have 1 to 4 heteroatoms selected from a nitrogen atom, an oxygen atom, and a sulfur atom in addition to a carbon atom as a ring-constituting atom. Or a 6-membered ring group, wherein the ring atoms X and X are carbon atoms or nitrogen atoms, respectively.
- the ring-constituting atom X represents a carbon atom, a nitrogen atom, an oxygen atom or a sulfur atom, R 1
- R 2 represents an optionally substituted alkyl group, an optionally substituted aryl group or Represents an optionally substituted heteroaryl group
- R 3 represents a ring structure other than X, X and X.
- a substituent on a synthetic atom which may be an aminoamino group which may be substituted with one or two lower alkyl groups, and ring A is further selected from a lower alkyl group, a halogen atom, a cyano group and an oxo group;
- each symbol has the same meaning as described above, and the pyrrole ring may further have a substituent selected from a lower alkyl group, a halogen atom, a cyano group and an oxo group]
- their salts hereinafter abbreviated as Compound (I)
- Compound (I) have an unexpectedly very strong acid secretion inhibitory action (proton pump inhibitory action) and are sufficiently satisfactory as a pharmaceutical.
- the present invention was completed based on these findings.
- ring A may have 1 to 4 heteroatoms selected from a nitrogen atom, an oxygen atom and a sulfur atom in addition to a carbon atom as a ring-constituting atom. Or a 6-membered ring group, wherein the ring atoms X and X are carbon atoms or nitrogen atoms, respectively.
- the ring-constituting atom X represents a carbon atom, a nitrogen atom, an oxygen atom or a sulfur atom, R 1
- R 2 represents an optionally substituted alkyl group, an optionally substituted aryl group or Represents an optionally substituted heteroaryl group
- R 3 represents a ring structure other than X, X and X.
- a substituent on a synthetic atom which may be an aminoamino group which may be substituted with one or two lower alkyl groups, and ring A is further selected from a lower alkyl group, a halogen atom, a cyano group and an oxo group;
- pyrrole ring may further have a substituent selected from a lower alkyl group, a halogen atom, a cyano group and an oxo group]
- An acid secretion inhibitor comprising a salt thereof, or a prodrug thereof,
- ring C may have 1 to 4 heteroatoms selected from a nitrogen atom, an oxygen atom and a sulfur atom in addition to a carbon atom as a ring-constituting atom, saturated or unsaturated.
- Ring D may have 1 to 4 heteroatoms selected from nitrogen, oxygen and sulfur atoms in addition to carbon atoms as ring constituent atoms, saturated or unsaturated
- R la and R lb are each an optionally substituted aryl group or substituted, and may be a heteroaryl group
- R 2a and R 2b are each substituted
- R 6a , R 7a , R 6b, and R 7b are each unsubstituted, substituted, substituted, substituted, aryl, or substituted, substituted, hetero, or heteroaryl.
- R 3a and one or two optionally substituted aminomethyl group R 3b are each a lower alkyl group, R 8b is unsubstituted force ⁇ or hydrogen atom, a lower alkyl group, a halogen atom , Represents a cyano group or an oxo group] (provided that the formula
- ring C may have 1 to 4 heteroatoms selected from a nitrogen atom, an oxygen atom and a sulfur atom in addition to a carbon atom as a ring-constituting atom, saturated or unsaturated.
- Ring D may have 1 to 4 heteroatoms selected from nitrogen, oxygen and sulfur atoms in addition to carbon atoms as ring constituent atoms, saturated or unsaturated
- R la and R lb are each an optionally substituted aryl group or substituted, and may be a heteroaryl group
- R 2a and R 2b are each substituted
- R 6a , R 7a , R 6b, and R 7b are each unsubstituted, substituted, substituted, substituted, aryl, or substituted, substituted, hetero, or heteroaryl.
- the ⁇ amino group, one or two optionally substituted aminomethyl group R 3a and R 3b are each a lower alkyl group, the force ⁇ also R 8b is unsubstituted Represents a hydrogen atom, a lower alkyl group, a halogen atom, a cyano group or an oxo group].
- R la is (i) halogen, (ii) hydroxy, (iii) cyan, (iv) C alkyl optionally substituted with halogen, (V) C alkoxy optionally substituted with halogen. (Vi) C
- 1-6 Amino group optionally substituted with 1-6 alkyl, (vii) oxo, (viii) canolevamoyl, (ix) monono C alkyl one strength rubermoyl, (X) di-C alkyl one strength ruber moyl, (xi ) C
- R 2a is an optionally substituted alkyl group, an optionally substituted aryl group or an optionally substituted heteroaryl group
- R 3a is a lower alkyl group of 1 or 2
- a medicament comprising the compound according to [9] above or a prodrug thereof,
- the present invention also provides:
- ring A may have 1 to 4 heteroatoms selected from a nitrogen atom, an oxygen atom and a sulfur atom in addition to a carbon atom as a ring-constituting atom, saturated or unsaturated.
- a 5- or 6-membered ring group, and the ring atoms X and X are each a carbon atom or a nitrogen atom.
- ring atom X represents a carbon atom, a nitrogen atom, an oxygen atom or a sulfur atom, R :
- R 2 represents an optionally substituted alkyl group, an optionally substituted aryl group or Represents an optionally substituted heteroaryl group
- R 3 represents a ring structure other than X, X and X.
- a substituent on a synthetic atom which may be an aminoamino group which may be substituted with one or two lower alkyl groups, and ring A is further selected from a lower alkyl group, a halogen atom, a cyano group and an oxo group;
- each symbol has the same meaning as described above, and the pyrrole ring may further have a substituent selected from a lower alkyl group, a halogen atom, a cyan group, and an oxo group].
- ring C may have 1 to 4 heteroatoms selected from a nitrogen atom, an oxygen atom and a sulfur atom in addition to a carbon atom as a ring-constituting atom, saturated or unsaturated.
- Ring D may have 1 to 4 heteroatoms selected from nitrogen, oxygen and sulfur atoms in addition to carbon atoms as ring constituent atoms, saturated or unsaturated
- R la and R lb are each an optionally substituted aryl group or substituted, and may be a heteroaryl group
- R 2a and R 2b are each substituted
- R 6a , R 7a , R 6b, and R 7b are each unsubstituted, substituted, substituted, substituted, aryl, or substituted, substituted, hetero, or heteroaryl.
- R da and R db are each an aminomethyl group which may be substituted with one or two lower alkyl groups
- R 8b is an unsubstituted or hydrogen atom, lower An alkyl group, a halogen atom, a cyano group or an oxo group]
- a peptic ulcer, Zollinger-Ellison syndrome, gastritis, reflux esophagitis, symptomatic gastroesophageal reverse 3 ⁇ 4i ⁇ t3 ⁇ 4E comprising the proton pump inhibitor described in [15] above Symptomatic gastroesophageal Reflux Disease (symptomatic jr ERD)), NUD (Non Ulcer Dyspepsia), gastric cancer, gastric MALT lymphoma, ulcers caused by nonsteroidal anti-inflammatory drugs or postoperative stress and treatment and prevention of ulcers; or Inhibitor of upper gastrointestinal bleeding due to peptic ulcer, acute stress ulcer, hemorrhagic gastritis or invasive stress,
- ring C may have 1 to 4 heteroatoms selected from a nitrogen atom, an oxygen atom and a sulfur atom in addition to a carbon atom as a ring-constituting atom, saturated or unsaturated.
- Ring D may have 1 to 4 heteroatoms selected from nitrogen, oxygen and sulfur atoms in addition to carbon atoms as ring constituent atoms, saturated or unsaturated
- R la and R lb are each an optionally substituted aryl group or substituted, and may be a heteroaryl group
- R 2a and R 2b are each substituted
- R 6a , R 7a , R 6b, and R 7b are each unsubstituted, substituted, substituted, substituted, aryl, or substituted, substituted, hetero, or heteroaryl.
- R 3a and one or two optionally substituted aminomethyl group R 3b are each a lower alkyl group, R 8b is unsubstituted force ⁇ or hydrogen atom, a lower alkyl group, a halogen atom , Represents a cyano group or an oxo group] (provided that the formula
- a medicament comprising the compound according to [21] or a prodrug thereof,
- a peptic ulcer, Zollinger-Ellison syndrome, gastritis, reflux characterized by administering an effective amount of the compound or prodrug thereof described in [21] to a mammal Esophagitis, Symptomatic Gastroeso phageal Reflux Disease (Symptomatic GERD), NUD (Non Ulcer Dys pepsia), gastric cancer, gastric MALT lymphoma, ulcers caused by non-steroidal anti-inflammatory agents or postoperative Methods of treating or preventing gastric hyperacidity and ulcers due to stress; or methods of suppressing peptic ulcers, acute stress ulcers, hemorrhagic gastritis or upper gastrointestinal bleeding due to invasive stress, and
- the present invention relates to the use of the compound described in [21] above or a prodrug thereof for producing an inhibitor of upper gastrointestinal bleeding due to stress.
- Compound (I) exhibits an excellent proton pump inhibitory action.
- conventional proton pump inhibitors such as omebrazol and lansoprazole bind covalently to cystine residues of H + ZK + _ATPase and irreversibly inhibit enzyme activity
- compound (I) Tonpump (H + / K + -ATPase) activity is reversibly inhibited by a K + antagonistic inhibition mode, and as a result, acid secretion is suppressed. Therefore, potassium ion competitive acid blocker ( ⁇ tassium ⁇ competitive acid blocker: P—CAB), sometimes referred to as Acid Pump Antagonist (APA).
- APA Acid Pump Antagonist
- the present invention relates to peptic ulcers (eg, gastric ulcers, duodenal ulcers, anastomotic ulcers, ulcers caused by non-steroidal anti-inflammatory agents, ulcers due to postoperative stress, etc.), Zollinger-Ellison (Zollinger-Ellison) ) Syndrome, gastritis, erosive esophagitis, reflux esophagitis, symptomatic Gastroesophage al Reflux Disease (Symptomatic GERD),: Barrett esophagus, Functional Dyspepsia; NUD (Non Ulcer Dyspepsia))), clinically useful preventive and therapeutic agents for gastric cancer, gastric MALT lymphoma, or hyperacidity; or upper gastrointestinal bleeding due to extirpable ulcer, acute stress ulcer, hemorrhagic gastritis or invasive
- Compound (I) is useful as a pharmaceutical because it is less toxic and has excellent water solubility, pharmacokinetics, and drug efficacy. Furthermore, since compound (I) is stable under acidic conditions, it can be administered orally as a normal tablet or the like without an enteric preparation. For this reason, since preparations, such as a tablet, can be made small, it has the advantage that it becomes easy to take to a sick person with weak swallowing force, especially an elderly person and a dwarf. In addition, since there is no sustained release effect as in the case of enteric preparations, the improvement of symptoms such as pain that causes rapid onset of gastric acid secretion suppression is rapid.
- ring A may have 1 to 4 heteroatoms selected from a nitrogen atom, an oxygen atom and a sulfur atom in addition to a carbon atom as a ring-constituting atom, saturated or unsaturated. Represents a 5- or 6-membered cyclic group.
- ring A examples include thiophene ring, furan ring, pyrrole ring, imidazole ring, pyrazole ring, isothiazole ring, thiazole ring, isoxazole ring, oxazole ring, pyrrolidine ring, pyrroline ring, imidazolidine ring, imidazoline ring, villa Zolidine ring, pyrazoline ring, furazane ring, oxadiazole ring (eg, 1, 2, 3-oxadiazole ring, 1, 2, 4-oxadiazole ring, 1, 3, 4-oxadiazole ring), thiadiazole ring (eg, 1 , 2, 3-thiadiazole ring, 1, 2, 4-thiadiazole ring, 1, 3, 4-thiadiazole ring), triazole ring (eg, 1, 2, 3-triazole ring, 1, 2, 4-triazole ring) ), Tetrazole ring, tetrahydrofuran
- the ring member atom (X) of ring A to which the substituent group to be bonded is bonded is a carbon atom or a nitrogen atom.
- the ring member atom (X) adjacent to X is a carbon atom, nitrogen atom, oxygen atom or sulfur atom.
- R 6 may have a substituent (R 6 ) selected from an alkyl group, a halogen atom and a cyan group.
- R 6 substituents
- Examples of the “lower alkyl group” represented by R 6 include C alkyl groups such as methylol, ethyl, propyl, isopropyl, butyl, isobutyl, sec butyl, tert butyl and the like.
- halogen atom represented by R 6 include a fluorine atom, a chlorine atom, a bromine atom, and an iodine atom.
- the aryl group in the “optionally substituted aryl group” represented by R 1 includes phenyl, 1 naphthyl, 2 naphthyl, 2 biphenylyl, 3 biphenylyl, 4-biphenylolinole, 2-anthryl.
- C aryl group such as
- Substituents for the aryl group include (1) halogen atoms (eg, fluorine atoms, chlorine atoms, fluorine atoms, iodine atoms, etc.), (2) nitro, (3) cyano, (4) hydroxy, (5) It may have 1 to 5 (preferably 1 to 3) halogen atoms (eg, fluorine, chlorine, bromine, iodine), C alkoxy (eg, methoxy, ethoxy, propoxy, isopropoxy, Butoxy, iso
- alkylthio eg, benzylthio, phenethylthio, diphenylmethylthio, 1_naphthylmethylthio, 2_naphthylmethylthio, 2,2_diphenylethylthio, 3-phenylpropylthio, 4-phenylbutylthio, 5-phenyl Dirupentylthio, etc.
- arylenoamino eg, pheninoreamino, 1_naphthinoleamino, 2-naphthylamino
- mono-C aralkylamino eg, benzylamino
- di-C anorequinoleamino
- Di-C arylamine e.g., Diphenyl
- alkyl carbonyl eg, acetyl, propionyl, etc.
- arylcarbonyl eg, benzoyl, 1-naphthoyl, 2-naphthoyl, etc.
- (22) force noroxyl e.g., noroxycarbonyl, ethoxycanole
- (23) C alkoxycarbonyl eg, methoxycarbonyl, ethoxycanole
- Rurubamoyl eg, phenylcarbamoyl, 1_naphthylcarbamoyl, 2_naphthylcarbamoyl, etc.
- C alkylsulfonyl eg, methylsulfonyl, ethyl
- killsulphonylamino eg, methylsulphonylaminoethyl sulphonylamino
- C arylsulylamino eg, phenylsulphonylamino, 2-naphthylsulphoni
- Bonyloxy (eg, methoxycarbonyloxy, ethoxycarbonyloxy, propoxycarbonyloxy, butoxycarbonyloxy, etc.), (43) mono-C alkyl-carb
- Vamoyloxy eg, methylcarbamoyloxy, ethylcarbamoyloxy, etc.
- Di-C alkyl alkyl rubamoyloxy eg, dimethylcarbamoyloxy
- C cycloalkyl eg, cyclopropyl, ethylene dioxy, etc.
- Oral pill cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, etc.), (50) 1 to 5 (preferably 1 to 3) halogen atoms (eg, fluorine, chlorine, bromine, iodine) C alkyl group (eg, methyl, ethyl, n-propyl, isopropyl)
- halogen atoms eg, fluorine, chlorine, bromine, iodine
- C alkyl group eg, methyl, ethyl, n-propyl, isopropyl
- 1 to 5 preferably 1 to 3) halogen atoms (eg, fluorine, chlorine, bromine, iodine) C aryl group (eg phenyl), (54) 1 to 5 (preferably 1 to 3)
- Halogen atoms eg, fluorine, chlorine, bromine, iodine
- 7-16 kills eg, benzyl, phenethyl, etc.
- the number of substituents which can be substituted at the substitutable position is 1 to 5, preferably 1 to 3.
- heteroaryl group in the “substituted, optionally, heteroaryl group” represented by R 1 examples include pyrrolyl (eg, 1—, 2 or 3 pyrrolyl), pyrazolyl (eg, 1—, 3—, 4—or 5 pyrazolyl), imidazolyl (eg, 1—, 2—, 4 or 5—imidazolyl), tria ⁇ (NO, ⁇ 3 (J, 1, 2, 3— -1, 1, 3-Tri ⁇ 1-1, 1, 3, 1-1, 1-2, 1-1, 1, 2-3 , 4—Tri-Insole 1-Nore 3-Inole, 1, 2, 4 ⁇ ⁇ 1-Nore 4-Inole, 1, 2, 4 ⁇ -Inol ⁇ -Inole 5—Inole), Tetrazolyl (eg, Tetrazole 1-— , 2 or 5—yl), furyl (eg, 2 or 3—furyl), cenyl (eg, 2 or 3 cheryl), oxazolyl (
- Examples of the substituent of the heteroaryl group include the same substituents that the aryl group in R 1 may have.
- the substituent is present at a substitutable position, and the number of substituents is 1 to 5, preferably 1 to 3.
- alkyl group in the “optionally substituted alkyl group” represented by R 2 examples include methyl, ethyl, n_propyl, isopropyl, n-butyl, isobutyl, sec-butyl, t ert-butinole, n-pentinole. , Sec pentinoles, isopentinoles, neopentinoles, C-alkyl groups such as n-hexyl and isohexyl.
- the substituent of the alkyl group includes (1) a halogen atom (eg, fluorine atom, chlorine atom, odor Elementary atoms, iodine atoms, etc.), (2) nitro, (3) cyan, (4) hydroxy, (5) 1 to 3 halogen atoms (eg, fluorine, chlorine, bromine, iodine) C alkoxy (e.g.,
- alkylthio eg, methylthio, difluoromethylthio, trifluoromethylthio, ethylthio, propylthio, isopropylthio, butylthio, 4, 4, 4 trifluorobutylthio, pentylthio, hexylthio, etc.
- C arylthio eg, phenylthio
- Methoxycanole poninole Methoxycanole poninole, ethoxycanoleboninole, propoxycanoleboninole, tert_butoxycarbonyl, etc.
- C aryloxycarbonyl eg, phenoxycarbonyl, etc.
- C arylsulfinyl eg, phenylsulfinyl, 1_naphthylsulfinyl, 2
- C cycloalkyl eg, cyclopropyl, cyclobutyl, cyclopentyl, cycl
- the number of substituents is 1 to 3.
- R 2 ' is substituted, it may also be, Ariru group "or” substituted with Re, also good les, Te Roariru group to "as is being” substituted in the R 1 Examples thereof include the same as the “optionally aryl group” and the “optionally substituted heteroaryl group”.
- R 3 is a substituent on a ring member atom other than X, X and X, and is a lower alkyl group.
- the lower alkyl group in the “aminomethyl group optionally substituted with one or two lower alkyl groups” includes C alkyl such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, etc. Groups and the like.
- R 3 - CH- NR 4 R 4 and R 5 in R 5 may be the same or different and each represents a hydrogen
- a lower alkyl group such as an atom or a C alkyl group.
- R 3 includes aminomethyl group (one CH 2 —NH 2), methylaminomethyl group (
- Ring A is located at a substitutable position in addition to the substituents represented by —SO 2 R 2 and R 3.
- substituents examples include lower alkyl groups (eg, C alkyl groups such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl, see-butyl, tert_butyl, etc.), halogen atoms (eg, fluorine atoms, chlorine atoms). , Bromine atom, iodine
- ring B may have 1 to 4 heteroatoms selected from a nitrogen atom, an oxygen atom, and a sulfur atom in addition to a carbon atom as a ring-constituting atom. Or a 6-membered ring group, wherein the ring atoms X and X are carbon atoms or nitrogen atoms, respectively.
- ring atoms X and X are each a carbon atom, nitrogen atom, oxygen atom or
- X is (
- An atom having a substituent R 3 on the atom, and the other may be a carbon atom, a nitrogen atom, an oxygen atom or a sulfur atom, or a carbon atom or a nitrogen atom.
- R 1 represents a substituted or unsubstituted or aryl group or a substituted or unsubstituted or heteroaryl group
- R 2 represents an optionally substituted alkyl group or a substituted group.
- R 3 is an aminomethyl (—CH—NR 4 R 5 ) group (R) which may be substituted with one or two lower alkyl groups. 4 and R 5 are the same or
- the ring B may have "1 to 4 heteroatoms selected from a nitrogen atom, an oxygen atom and a sulfur atom in addition to a carbon atom as a ring-constituting atom.
- Examples of the “unsaturated 5- or 6-membered ring group” include those similar to the 5- or 6-membered ring group exemplified for ring A.
- Ring atoms X and X each represent a carbon atom or a nitrogen atom, and the ring atoms
- X and X each represent a carbon atom, a nitrogen atom, an oxygen atom or a sulfur atom.
- X is a carbon atom or a nitrogen atom, a lower alkyl group, halogen atom and
- R 6 may have a substituent (R 6 ) selected from a cyano group.
- R 6 include the same as those exemplified for the compound (I).
- X is a carbon atom or a nitrogen atom, a lower alkyl group, halogen atom and
- R 7 may have a substituent (R 7 ) selected from a cyano group.
- lower alkyl group and “halogen atom” in the above are the same as those for R 6 .
- Preferred embodiments of the compounds (I) and ( ⁇ ) are the following compounds (la) and (lb).
- ring C represents a nitrogen atom, an oxygen atom, or sulfur in addition to a carbon atom as a ring-constituting atom. It may have 1 to 4 heteroatoms selected from atoms, a saturated or unsaturated 5-membered ring group
- ring D is a nitrogen atom, oxygen atom and sulfur other than carbon atoms as ring constituent atoms It may have 1 to 4 heteroatoms selected from atoms, a saturated or unsaturated 6-membered cyclic group
- R la and R lb each may be an optionally substituted aryl group or a substituted
- R 2a and R 2b are each substituted, R, may, alkyl, substituted, ally, aryl, or substituted,
- a heteroaryl group, R 6a , R 7a , R 6b and R 7b are each an unsubstituted force, or a hydrogen atom, a lower alkyl group, a halogen atom
- ring C examples include thiophene ring, furan ring, pyrrole ring, imidazole ring, pyrazole ring, isothiazole ring, thiazole ring, isoxazole ring, oxazole ring, pyrrolidine ring, pyrroline ring.
- a pyrrole ring, an imidazole ring or a thiazole ring is preferred.
- the ring-constituting atom of ring C to which the group represented by -SO _R la is bonded the configuration represented by R 2a
- the ring atom of ring C to which the substituent is bonded and the ring atom of ring C to which the substituent represented by R da is bonded are a carbon atom or a nitrogen atom.
- the ring constituent atom of ring C to which the substituent represented by R 6a or R 7a is bonded represents a carbon atom, a nitrogen atom, an oxygen atom or a sulfur atom.
- the ring-constituting atom of ring C to which the substituent represented by R 6a or R 7a is bonded is an oxygen atom or a sulfur atom, it cannot have the substituent represented by R 6a or R 7a .
- Substituent means unsubstituted.
- R 6a or R 7a is a force that is unsubstituted, or A hydrogen atom, a lower alkyl group, a halogen atom or a cyan group;
- the compound in which R 6a or R 7a is unsubstituted is R 6a or R 7 ring-constituting atoms of the ring C which is a substituent represented by a binding is a compound the double bond is present between the ring-constituting atom adjacent a with the nitrogen atom a nitrogen atom.
- Examples of the “lower alkyl group” represented by R 6a or R 7a include C-alkyl such as methylol, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl and the like.
- halogen atom represented by R 6a or R 7a
- examples of the “halogen atom” represented by R 6a or R 7a include a fluorine atom, a chlorine atom, a bromine atom, and an iodine atom.
- R la , R 2a and R 3a include the same groups as those exemplified for R 1 R 2 and R 3 described above.
- ring D examples include pyridine ring, pyrazine ring, pyrimidine ring, pyridazine ring, piperidine ring, piperazine ring, monorephorin ring, thiomorpholine ring, pyran ring, thiopyran ring, triazine.
- Ring eg, 1, 2, 4_triazine ring, 1, 3, 5_triazine ring
- oxazine ring eg, 1,4_oxazine ring, 1, 3_oxazine ring
- thiazine ring eg, 1, 4_thiazine ring, 1,3_thiazine ring
- oxaziazine ring thiadiazine ring
- tetrahydran pyran ring tetrahydropyridine ring (eg, 1, 2, 3, 6-tetrahydropyridine ring)
- dihydropyridine ring eg, 1) , 4-dihydropyridine ring
- cyclohexane ring cyclohexene ring, cyclohexagen ring, benzene ring and the like.
- a benzene ring A pyridine ring or a pyrimidine ring.
- the ring atom of ring D to which the substituent is bonded and the ring atom of ring D to which the substituent represented by R 3b is bonded are a carbon atom or a nitrogen atom.
- the ring constituent atom of ring D to which the substituents represented by R 6b , R 7b and R 8b are bonded represents a carbon atom, a nitrogen atom, an oxygen atom or a sulfur atom.
- the ring represented by R 6b , R 7b, or R 8b is bonded to the ring D when the ring-constituting atom is an oxygen atom or a sulfur atom, respectively, it has the substituent represented by R 6b , R 7b, or R 8b Meaning that these substituents are unsubstituted.
- R 6b , R 7b or R 8b when the ring constituting atom of ring D to which the substituent represented by R 6b , R 7b or R 8b is bonded is a carbon atom or a nitrogen atom, R 6b , R 7b or R 8b is unsubstituted. Or a hydrogen atom, a lower alkyl group, a halogen atom or a cyano group.
- R 6b the compound R 7b or R 8b is unsubstituted, R 6b, adjacent nitrogen atom ring members ring D substituent represented by R 7b or R 8b is attached is a nitrogen atom It is a compound in which a double bond exists between atoms.
- lower alkyl group and “halogen atom” represented by R 6b , R 7b or R 8b are the same as the “lower alkyl group” and “halogen atom” represented by R 6a or R 7a described above. Can be mentioned.
- R lb , R 2b and R 3b are Examples thereof are the same as those exemplified for R 2 and R 3 .
- pyrrole ring, imidazole ring or thiazole ring derivatives represented by compounds (la— :!) to (Ia — 12) and (la — 21) to (la — 24) are preferred.
- the benzene ring, pyridine ring or pyrimidine ring derivatives represented by the compounds (lb- :!) to (Ib-28) are preferred.
- R la and R lb include (i) halogen (eg, fluorine, chlorine, bromine, iodine), (ii) hydroxy, (iii) cyan, (iv) halogen (eg, fluorine, chlorine, bromine, iodine) ) Optionally substituted with 1 to 5 (preferably 1 to 3) C alkyl (eg, methyl, ethyl,
- halogen eg, fluorine, chlorine, bromine, iodine
- C alkoxy eg, methoxy, ethoxy, propoxy, di
- Vamoyl eg, dimethylcarbamoyl, jetylcarbamoyl, ethylmethylcarbamoyl, etc.
- Xi C alkylsulfonyl (eg, methylsulfonyl, ethylsulfonyl, etc.)
- Prime ring group or condensed ring group thereof eg, condensed ring group of 5- to 6-membered aromatic heterocyclic group and benzene ring or 5- to 6-membered aromatic heterocyclic ring
- C such as phenyl, 1 or 2 naphthyl
- Aryl group 2 or 3 phenyl, 2 or 3 furyl, 2 or 3 pyro
- Ril 2—, 4 or 5 oxazolyl, 2—, 4 or 5 thiazolyl, 2—, 4 or 5—imidazolyl, 3—, 4 or 5—isoxazolyl, 3—, 4 or 5—isothiazolyl, 3— , 4 or 5 pyrazolyl, 2—, 3 or 4 pyridyl, 2—, 4 or 5_pyrimidinyl, 3_ or 4_pyridazinyl, 2 to 6-membered aromatic heterocyclic groups such as 2-pyrazur; 3_benzofuryl, 2_ or 3_benzocenyl, 1_ or 3_isoindolyl, 2_benzimidazolyl, 2_benzoxazolyl, 3_benzoisoxazolyl, 2_benzothiazolyl, 3_benzoisothiazolyl, 2_, 3 _ Or 4 _ quinolyl, 1_, 3_ or 4_ isoquinolyl, 3_ or 4_
- halogen eg, fluorine, chlorine, bromine, iodine
- hydroxy e.g. cyan
- halogen eg, fluorine, chlorine, bromine, iodine
- C alkyl eg, methyl, ethyl, propyl, isopropyl
- halogens eg, fluorine, chlorine, bromine, iodine
- C alkoxy eg, methoxy, ethoxy, propoxy, isopropoxy, butoxy
- 1-6 alkyl-carbonylamino eg, acetylamino
- a phenyl group eg, 2-, 3- or 4-pyridyl
- benzozenyl groups eg 2- or 3-benzozoenyl
- halogens eg fluorine, chlorine, bromine, iodine
- halogens eg fluorine, chlorine
- Bromine, iodine C alkyl optionally substituted with 1 to 5 (preferably 1 to 3) (eg, methyl)
- halogen eg, fluorine, chlorine, bromine, iodine
- C alkoxy eg, methoxy, ethoxy
- R 2 , R 2a and R 2b include: [1] (i) halogen (eg, fluorine, chlorine, bromine, iodine), (ii) hydroxy, (iii) cyano, (iv) halogen (eg, fluorine, chlorine, bromine, iodine) ) Optionally substituted with 1 to 5 (preferably 1 to 3) C-alkyl (eg, methinole, eth Nore, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, hexyl, etc.), (V) halogen (eg, fluorine, chlorine, bromine, iodine):! ⁇ 5 (preferably Or 1 to 3 optionally substituted
- C alkyl eg, methylol, ethyl, propyl, isopropyl, buty
- Amino group optionally substituted with Nore, isobutyl, sec-butyl, tert-butyl, pentyl, hexyl, etc.), (vii) force rubamoyl, (viii) mono-C alkyl-force rubamoyl (
- Moyl eg, dimethylcarbamoyl, jetylcarbamoyl, ethylmethylcarbamoyl, etc.
- X C alkylsulfonyl (eg, methylsulfonyl, ethylsulfonyl, etc.)
- C aryl group e.g., phenyl
- ⁇ 5 preferably 1 to 3 substituents.
- Halogen eg, fluorine, chlorine, bromine,
- Til isobutyl, sec-butyl, tert-butyl, pentyl, hexyl, etc.
- halogen eg, fluorine, chlorine, bromine, iodine:! ⁇ 5 (preferably 1 to 3)
- C alkoxy eg, methoxy, ethoxy, propoxy, isopropoxy, butoxy, i
- 5- to 6-membered aromatic heterocyclic group or its condensed ring group (eg, 5- to 6-membered aromatic heterocyclic group) which may be substituted with 1 to 3 substituents selected from ninoleamino (eg, acetylamino) And a benzene ring or a condensed ring group of a 5- to 6-membered aromatic heterocyclic ring) (for example, 2_ or 3_ Enyl, 2 or 3 furyl, 2 or 3 pyrrolyl, 2—, 4 or 5 oxazolinole, 2—, 4 or ⁇ or 5 thiazolinole, 2—, 4 or ⁇ 5 imidazolinole, 3—, 4 or 5—iso Oxazolinole, 3—, 4 or 5—isothiazolinole, 3—, 4 or 5—virazolyl, 2_, 3_ or 4_pyridyl, 2_, 4_ or 5_pyrimidinyl, 3_ or
- halogen eg, fluorine, chlorine, bromine, iodine
- hydroxy e.g., hydroxy
- halogen eg, fluorine, chlorine, bromine, iodine
- C alkyl eg., methyl, ethyl, propyl, isopropyl, buty
- substituents selected from ninoleamino (eg, acetylamino) C aryl group (eg, phenyl group) which may be substituted with [2] (i) halogen (eg, fluorine group)
- halogen eg, fluorine, chlorine, bromine, iodine
- C alkoxy eg, methoxy
- Amino group optionally substituted with sopropyl, butyl, isobutyl, sec-butyl, tert_butyl, pentyl, hexyl, etc.), (vii) rubamoyl, (viii) mono C alkyl
- rukyl-strength rubamoyl eg, dimethylcarbamoyl, jetylcarbamoyl, ethylmethylcarbamoyl, etc.
- C alkylsulfonyl eg, methylsulfonyl, ethyl
- substituents selected from: 1 with 2 or 3 cenyl or 2—, 3 or 4 pyridyl, or [3] halogen (eg, fluorine, chlorine, bromine, iodine) ⁇ 5 (preferably 1 to 3) optionally substituted C alkyl (eg methyl, ethyl)
- halogen atoms eg, fluorine, chlorine, bromine, iodine
- halogen atoms eg, fluorine, chlorine, bromine, iodine
- Halogen eg, fluorine, chlorine, bromine, iodine:! To 5 (preferably 1 to 3) optionally substituted C alkyl (eg, methinole, ethyl, propiyl)
- a phenyl group which may be substituted with 1 to 5 (preferably 1 to 3) substituents selected from Nore, isopropyl, butyl, isobutyl, see butyl, tert_butyl, pentyl, hexinole, etc.
- C alkyl optionally substituted with 1 to 5 (preferably 1 to 3) halogen (eg, fluorine, chlorine, bromine, iodine) (eg, methylol, ethyl, propyl, iso)
- R 2 , R 2a , and R 2b are preferably a phenyl group, a 2_fluorophenyl group, or 2-methylphenyl group; and C alkyl (eg, methylol, ethyl, propyl, iso
- R 3 in the above compounds (1) (1,), (la), (lb), (la— :!) to (Ia — 36) and (lb —:!
- R 3a , R 3b are aminomethyl groups (one CH 2 —NH 2), methinoreamino
- R 6 , R 6a , R 6b are [1] unsubstituted (eg, compounds (Ia_7), (Ia_9), (Ia_14), (la-18) to (Ia-20), (la-22) to (la— 24), (Ia_26), (Ia_28), (I a_31), (Ia_32), (Ia_35), (Ia_36), (Ib_8), (Ib_15), (Ib_19), (lb—20), (lb—36 ), (Ib_37), (Ib_39), (Ib_41), etc.), [2] hydrogen atoms, [3] C alkyl groups such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec but
- R 7 , R 7a and R 7b are [1] unsubstituted (eg, compounds (la-5), (la-12), (la16) to (Ia-18), (Ia-20) to (Ia-22) ), (Ia—24), (Ia—25), (Ia—27), (la 32), (Ia—36), (lb—4), (lb—11), (lb—23), ( lb—24), (lb—26), (lb—2 8), (lb—31), (lb—33), etc.), [2] hydrogen atoms, [3] methyl, ethyl, propyl, isopropyl, C alkyl groups such as butyl, isobutyl, sec butyl, tert butyl,
- a halogen atom eg, fluorine, chlorine, bromine, iodine
- a cyan group e.g, fluorine, chlorine, bromine, iodine
- R 8 and R 8b are:
- Examples of the salt of compound (I) include metal salts, ammonium salts, salts with organic bases, salts with inorganic acids, salts with organic acids, salts with basic or acidic amino acids, and the like.
- Preferable examples of the metal salt include alkali metal salts such as sodium salt and potassium salt; alkaline earth metal salts such as calcium salt, magnesium salt and sodium salt; aluminum salt and the like.
- Preferable examples of the salt with an organic base include, for example, trimethylamine, triethylamine, pyridine, picoline, 2,6-lutidine, ethanolamine, diethanolamine, triethanolamine, cyclohexylamine, dicyclohexylamine, And salts with N, N′-dibenzenole ethylenediamine and the like.
- Preferable examples of the salt with inorganic acid include salts with hydrochloric acid, hydrobromic acid, nitric acid, sulfuric acid, phosphoric acid and the like.
- salts with organic acids include formic acid, acetic acid, trifluoroacetic acid, phthalic acid, fumaric acid, oxalic acid, tartaric acid, maleic acid, succinic acid, succinic acid, malic acid, methanesulfonic acid, benzenesulfuric acid. And salts with phonic acid, p_toluenesulfonic acid and the like.
- salt with basic amino acid include salts with arginine, lysine, onolenitine and the like, and preferable examples of the salt with acidic amino acid include aspartic acid, glutamic acid and the like. Of salt.
- inorganic compounds such as alkali metal salts (eg, sodium salts, potassium salts, etc.), alkaline earth metal salts (eg, calcium salts, magnesium salts, barium salts, etc.), etc. Salt, ammonium salt, etc.
- the compound has a basic functional group, for example, a salt with an inorganic acid such as hydrochloric acid, hydrobromic acid, nitric acid, sulfuric acid, phosphoric acid, or acetic acid, phthalic acid, fumaric acid, oxalic acid And salts with organic acids such as tartaric acid, maleic acid, succinic acid, succinic acid, methanesulfonic acid and p-toluenesulfonic acid.
- an inorganic acid such as hydrochloric acid, hydrobromic acid, nitric acid, sulfuric acid, phosphoric acid, or acetic acid, phthalic acid, fumaric acid, oxalic acid
- organic acids such as tartaric acid, maleic acid, succinic acid, succinic acid, methanesulfonic acid and p-toluenesulfonic acid.
- compounds (II) to (XXI) in the formula may form a salt.
- Examples of such a salt include the same salts as the salt of compound (I).
- n represents an integer of 0, 1, and 2.
- the compound can be converted to a compound of n 2 by oxidation using an appropriate oxidizing agent in each compound. it can.
- the compound obtained in each step can be used for the next reaction as it is in the reaction solution or as a crude product, but can be isolated from the reaction mixture according to a conventional method and recrystallized. It can be easily purified by separation means such as crystallization, distillation, chromatography and the like.
- X 1 is a hydrogen atom or halogen (eg, fluorine, chlorine
- This reaction is advantageously performed using a solvent inert to the reaction.
- a solvent is not particularly limited as long as the reaction proceeds, but hydrocarbons such as benzene and toluene and ethers such as tetrahydrofuran, N, N-dimethylformamide, N, N-dimethylacetamide and the like. Solvents such as amides or mixed solvents thereof are preferred.
- the use of a base is effective.
- the base include inorganic bases such as sodium hydride, sodium hydroxide and potassium hydroxide, basic salts such as sodium carbonate, potassium carbonate, cesium carbonate and sodium hydrogen carbonate, potassium ethoxide, potassium tert-butoxide, Metal bases such as sodium methoxide and sodium ethoxide, aromatic amines such as pyridine and lutidine, triethylamine, tripropylamine, tributylamine, cyclohexyldimethylamine, 4-dimethylaminopyridine, N, And tertiary amines such as N-dimethylaniline, N-methylbiperidine, N-methylpyrrolidine, N-methylmorpholine.
- the amount of these bases to be used is about 1 to about 10 mol, preferably about 1 to about 5 mol, per 1 mol of compound (11).
- This reaction can also be carried out in the presence of crown ethers or halogenating agents.
- crown ethers include 15-crown 1-ether and 18-crown 1-ether.
- Halogenating agents include N-iodosuccinimide, N-bromosuccinimide, N-chlorosuccinimide, bromine.
- Etc The amount of the crown ether and the halogenating agent to be used is about 1 to about 10 monolayers, preferably about 1 to about 5 moles, per 1 mole of the compound (II).
- reaction time varies depending on the reagent and solvent to be used, it is generally about 30 min to about 24 hr, preferably about 30 min to about 8 hr.
- the reaction temperature is generally about 0 ° C to about 100 ° C, preferably about 10 ° C to about 50 ° C.
- This reaction is also carried out by a method similar to or similar to Organic Letters (Org. Lett.), 6 ⁇ , page 4587 (2004), Synlett, page 1254 (2004). I can do it.
- compound (IV) is compounded from lithium aluminum hydride, diisobutylaluminum hydride, sodium borohydride, hydrogenated It can manufacture by reducing using reducing agents, such as boron calcium.
- reducing agents such as boron calcium.
- diisobutylaluminum hydride is particularly preferable.
- the amount of these reducing agents to be used is about 0.75 to about 10 equivalents, preferably about 1 to about 5 equivalents, per 1 mol of compound (IV).
- This reaction is advantageously performed using a solvent inert to the reaction.
- a solvent is not particularly limited as long as the reaction proceeds, but solvents such as hydrocarbons such as benzene and toluene and ethers such as tetrahydrofuran and jetyl ether or a mixed solvent thereof are preferable. Masle.
- reaction time varies depending on the reagent and solvent to be used, it is generally about 30 min to about 24 hr, preferably about 30 min to about 8 hr.
- the reaction temperature is usually about _78 ° C to about 100 ° C, preferably about _78 ° C to about 25 ° C.
- Compound (VI) (wherein each symbol is as defined above) includes compound (V) and chromic acid 'pyridine complex, pyridinium chromatochromate, manganese dioxide, sulfur trioxide' pyridine A complex can be synthesized by reacting with an oxidizing agent such as tetra_n_propylammonium perruthenate. Oxidation As the agent, manganese dioxide, sulfur trioxide 'pyridine complex or tetra-n-propynoleammonium perruthenate is preferred. This oxidation reaction can be performed, for example, according to the method described in Synthesis, page 639 (1994).
- each reaction step for deriving compound (I) from the above compound (II) if desired, protection or deprotection reaction, acylation reaction, alkylation reaction, hydrogenation reaction, oxidation reaction, reduction reaction,
- the carbon chain extension reaction, the substituent exchange reaction, etc. may be conducted either alone or in combination of two or more thereof, and various intermediates may be used. After introducing a compound included in compound (I), These reactions may be combined to induce further to the desired mode.
- X 1 represents a hydrogen atom or a leaving group such as halogen (eg, fluorine, chlorine, bromine, iodine)). It can be manufactured by reacting.
- halogen eg, fluorine, chlorine, bromine, iodine
- This reaction is advantageously performed using a solvent inert to the reaction.
- solvents are not particularly limited as long as the reaction proceeds, but hydrocarbons such as benzene and toluene.
- ethers such as tetrahydrofuran, solvents such as N, N-dimethylformamide, amides such as N, N-dimethylacetamide, or a mixed solvent thereof are preferable.
- the use of a base is effective.
- the base include inorganic bases such as sodium hydride, sodium hydroxide and potassium hydroxide, basic salts such as sodium carbonate, potassium carbonate, cesium carbonate and sodium bicarbonate, potassium ethoxide and potassium tert-butoxide.
- Metal bases such as sodium methoxide and sodium ethoxide, aromatic amines such as pyridine and lutidine, triethinoleamine, tripropinoleamine, tributylamine, cyclohexyldimethylamine, 4-dimethylamino And tertiary amines such as pyridine, N, N-dimethylaniline, N-methylpiperidine, N-methylpyrrolidine, N-methylmorpholine.
- the amount of these bases to be used is about 1 to about 10 moles, preferably about 1 to about 5 moles per mole of Compound (VIII).
- This reaction can also be carried out in the presence of a crown ether or a halogenating agent.
- crown ethers include 15-crown 1-ether and 18-crown 1-ether.
- Halogenating agents include N-iodosuccinimide, N-bromosuccinimide, N-chlorosuccinimide, bromine.
- Etc. The amount of the crown ether and the halogenating agent to be used is about:! To about 10 moles, preferably about 1 to about 5 moles per mole of Compound (VIII).
- reaction time varies depending on the reagent and solvent to be used, it is generally about 30 min to about 24 hr, preferably about 30 min to about 8 hr.
- the reaction temperature is generally about 0 ° C to about 100 ° C, preferably about 10 ° C to about 50 ° C.
- Compound (IX) includes compound (X) and formula (XI)
- X 2 represents a leaving group such as halogen (eg, fluorine, chlorine, bromine, iodine), methanesulfonyloxy, etc.)
- halogen eg, fluorine, chlorine, bromine, iodine
- methanesulfonyloxy etc.
- solvents are not particularly limited as long as the reaction proceeds, but hydrocarbons such as benzene and toluene and ethers such as tetrahydrofuran, N, N-dimethylformamide, N, N-dimethylenoacetamide.
- a solvent such as amides such as N-methylbiperidone or a mixed solvent thereof is preferable.
- the use of a base is effective.
- the base include inorganic bases such as sodium hydride, sodium hydroxide and potassium hydroxide, basic salts such as sodium carbonate, potassium carbonate, cesium carbonate and sodium bicarbonate, potassium ethoxide and potassium tert-butoxide.
- Metal bases such as sodium methoxide and sodium ethoxide, aromatic amines such as pyridine and lutidine, triethinoleamine, tripropinoleamine, tributylamine, cyclohexyldimethylamine, 4-dimethylamino And tertiary amines such as pyridine, N, N-dimethylaniline, N-methylpiperidine, N-methylpyrrolidine, N-methylmorpholine.
- the amount of these bases to be used is about 1 to about 10 mol, preferably about 1 to about 5 mol, per 1 mol of compound (X).
- reaction time varies depending on the reagent and solvent to be used, it is generally about 30 min to about 24 hr, preferably about 30 min to about 8 hr.
- the reaction temperature is usually about 0 ° C to about 200 ° C, preferably about 10 ° C to about 100 ° C.
- compound (IX) is obtained by converting compound (IX) into lithium aluminum hydride, diisobutylaluminum hydride, sodium borohydride, bis (borohydride ) It can be produced by reducing with a reducing agent such as calcium.
- a reducing agent such as calcium.
- diisobutylaluminum hydride is particularly preferable.
- the amount of the reducing agent to be used is about 0.75 to about 10 equivalents, preferably about 1 to about 5 equivalents, per 1 mol of compound (IX).
- This reaction is advantageously performed using a solvent inert to the reaction.
- a solvent is not particularly limited as long as the reaction proceeds, but solvents such as hydrocarbons such as benzene and toluene and ethers such as tetrahydrofuran and jetyl ether or a mixed solvent thereof are preferable. .
- reaction time varies depending on the reagent and solvent used, it is generally preferred to be about 30 minutes to about 24 hours. Or about 30 minutes to about 8 hours.
- the reaction temperature is usually about -78 ° C to about 100 ° C, preferably about -78 ° C to about 25 ° C.
- each reaction step for deriving the compound (la) from the compound (VIII) or (X), or in each reaction step for deriving the compound (la) from the compound (XIV) or (XV) described later it may be carried out as desired. More intermediates by protecting or deprotecting reaction, acylation reaction, alkylation reaction, hydrogenation reaction, oxidation reaction, reduction reaction, carbon chain extension reaction, substituent exchange reaction, etc. Alternatively, after the compound included in the compound (la) is derived, the above-described reactions may be combined to induce further to a desired mode.
- the compounds (XIII) and (la) can also be produced by the following method.
- Compound (XIV) (in the formulas, each symbol is as defined above) may be a commercially available product, or a method known per se, for example, Journal of Medicinal Chemistry ⁇ Med. Chem. ), 43 ⁇ , 2165 (2000), Tetrahedron, 46 ⁇ , 7587 (1990) and Tetrahedron, 57 ⁇ , 7813 (2001), etc. Or according to a method according to these.
- Compound (XIII) (in the formulas, each symbol is as defined above) is the same as or similar to the method for producing compound (IX) from compound (VIII) with respect to compound (XIV).
- Compound (la) can also be produced by the following method.
- Compound (XVII) (wherein each symbol is as defined above, X 3 represents a leaving group such as halogen (eg, fluorine, chlorine, bromine, iodine)), compound (XVI) is chlorine, It can be obtained by treating with halogens such as bromine and iodine or metal halides such as copper bromide ( ⁇ ) and copper (II) chloride.
- halogen eg, fluorine, chlorine, bromine, iodine
- compound (XVI) is chlorine, It can be obtained by treating with halogens such as bromine and iodine or metal halides such as copper bromide ( ⁇ ) and copper (II) chloride.
- the amount of halogen or metal halide used is about 1 mole and about 5 moles, preferably about 1 mole and about 2 moles per mole of Compound (XVI).
- This reaction is advantageously performed in the absence of a solvent or in the presence of a solvent inert to the reaction.
- the solvent is not particularly limited as long as the reaction proceeds.
- This reaction can also be carried out in the presence of an acid or a base.
- Examples of the acid include inorganic acids such as hydrochloric acid and hydrobromic acid
- examples of the base include metal hydroxides such as sodium hydroxide, potassium hydroxide and lithium hydroxide, sodium carbonate, Basic salts such as potassium carbonate, cesium carbonate, sodium hydrogencarbonate, sodium acetate, aromatic amines such as pyridine and lutidine, triethylamine, tripropylamine, tributylamine, cyclohexyldimethylamine, 4-dimethyl And tertiary amines such as aminopyridine, N, N-dimethylaniline, N-methylbiperidine, N_methylpyrrolidine, N_methylmorpholine, and the like.
- the amount of the acid used is approximately 0.01 to approximately 3 mol, preferably approximately 0.01 to approximately 1 mol, with respect to 1 mol of the compound (XVI).
- the amount of the base to be used is about 1 to about 10 mol, preferably about 1 to about 3 mol, per 1 mol of compound (XVI).
- reaction time varies depending on the reagent and solvent to be used, it is generally about 5 min to about 24 hr, preferably about 10 min to about 5 hr.
- the reaction temperature is usually about 20 ° C to about 150 ° C, preferably about 0 ° C to about 100 ° C.
- the commercially available product may be used as it is, or may be obtained by a method known per se or a method analogous thereto.
- the amount of compound (XVIII) to be used is about 0.5 to about 3 mol, preferably about 0.8 to about 2 mol, per 1 mol of compound (XVII).
- This reaction is advantageously performed in the absence of a solvent or in the presence of a solvent inert to the reaction.
- the solvent is not particularly limited as long as the reaction proceeds.
- halogenated hydrocarbons aliphatic hydrocarbons, aromatic hydrocarbons, ethers, amides, alcohols, nitriles, or two of these A mixture of the above is used.
- This reaction can also be carried out in the presence of a base if desired.
- the base include basic salts such as sodium carbonate, potassium carbonate, cesium carbonate, sodium hydrogen carbonate and sodium acetate, aromatic amines such as pyridine and lutidine, triethylamine, tripropynoleamine, tributynoreamine and cyclohexene.
- aromatic amines such as pyridine and lutidine, triethylamine, tripropynoleamine, tributynoreamine and cyclohexene.
- tertiary amines such as xyldimethylamine, 4-dimethylaminopyridin, ⁇ , ⁇ _dimethylaniline, ⁇ methylbiperidine, ⁇ methylpyrrolidine, ⁇ methylmorpholine.
- the amount of the base to be used is about 1 to about 30 mol, preferably about 1 to about 10 mol, per 1 mol of compound (XVII).
- reaction time varies depending on the reagent and solvent to be used, it is generally about 5 min to about 72 hr, preferably about 0.5 to about 30 hr.
- the reaction temperature is usually about 5 ° C to about 200 ° C, preferably about 5 ° C to about 150 ° C.
- Compound (XIX) (wherein the symbols in the formula have the same meanings as described above) is the same as or similar to the method for producing compound (IX) from compound (VIII) with respect to compound (XX). Line It can be manufactured.
- Compound (la) (wherein the symbols in the formulas have the same meanings as defined above) represents the compound of formula (XXI) with respect to compound (XIX)
- X 4 represents a leaving group such as halogen (eg, fluorine, chlorine, bromine, iodine), methanesulfonyloxy, etc.) It can be produced by reacting.
- halogen eg, fluorine, chlorine, bromine, iodine
- methanesulfonyloxy etc.
- This reaction is advantageously performed using a solvent inert to the reaction.
- solvents are not particularly limited as long as the reaction proceeds, but hydrocarbons such as benzene and toluene and ethers such as tetrahydrofuran, N, N-dimethylformamide, N, N-dimethylacetate.
- Solvents such as amides such as amides or mixed solvents thereof are preferred.
- the use of a base is effective.
- the base include inorganic bases such as sodium hydride, sodium hydroxide and potassium hydroxide, basic salts such as sodium carbonate, potassium carbonate, cesium carbonate and sodium hydrogen carbonate, potassium ethoxide, potassium tert-butoxide, Metal bases such as sodium methoxide and sodium ethoxide, aromatic amines such as pyridine and lutidine, triethinoreamine, tripropinoleamine, tributylamine, cyclohexyldimethylamine, 4-dimethylaminopyridine And tertiary amines such as N, N-dimethylaniline, N-methylpiperidine, N-methylpyrrolidine, and N-methylmorpholine.
- the amount of these bases to be used is about 1 to about 10 monomers, preferably about 1 to about 5 mol, per 1 mol of compound (XIX).
- reaction time varies depending on the reagent and solvent to be used, it is generally about 30 min to about 24 hr, preferably about 30 min to about 8 hr.
- the reaction temperature is usually about ⁇ 20 ° C. to about 100 ° C., preferably about 0 ° C. to about 50 ° C.
- a protection or deprotection reaction In each reaction step for deriving the compound (la) from the compound (XVI) or (XX), a protection or deprotection reaction, an acylation reaction, an alkylation reaction, a hydrogenation reaction, An oxidation reaction, a reduction reaction, a carbon chain extension reaction, a substituent exchange reaction, etc., each of which may be used alone or in combination of two or more thereof, may be routed through various intermediates. After guiding, the above reactions may be combined to induce further to the desired mode.
- Compound (I) can be isolated and purified by a known means such as phase transfer, concentration, solvent extraction, fractional distillation, liquid conversion, crystallization, recrystallization, chromatography and the like.
- compound (I) When compound (I) is obtained as a free compound, it can be converted to the target salt by a method known per se or a method analogous thereto, and conversely, when it is obtained as a salt, it is known per se. It can be converted to a free form or other desired salt by the method described above or a method analogous thereto.
- Compound (I) may be used as a prodrug.
- a prodrug of compound (I) is a compound that is converted to compound (I) by a reaction with an enzyme, gastric acid or the like under physiological conditions in vivo, that is, it is enzymatically oxidized, reduced, hydrolyzed, etc. ) Refers to a compound that undergoes hydrolysis or the like by gastric acid or the like and changes to compound (I).
- a prodrug of the compound (I) a compound in which the amino group of the compound (I) is acylated, alkylated, and phosphorylated (for example, the amino group of the compound (I) is eicosanoylated, alanylated, pentylamino.
- prodrug of compound (I) changes to compound (I) under physiological conditions as described in Hirokawa Shoten, 1990, “Pharmaceutical Development”, 7th Molecular Design, pages 163 to 198. There may be.
- compound (I) has an isomer such as an optical isomer, a stereoisomer, a positional isomer, a rotational isomer, etc.
- an isomer such as an optical isomer, a stereoisomer, a positional isomer, a rotational isomer, etc.
- either isomer or mixture is encompassed in compound (I).
- the optical isomer resolved from the racemate is also encompassed in compound (I).
- Each of these isomers can be obtained as a single product by a known synthesis method or separation method (concentration, solvent extraction, column chromatography, recrystallization, etc.).
- Compound (I) is included in compound (I) whether it is a crystal or a single crystal form or a mixture of crystal forms. Crystals can be produced by crystallization by applying a crystallization method known per se.
- Compound (I) is included in Compound (I), whether it is a solvate (eg, a hydrate) or a non-solvate.
- a solvate eg, a hydrate
- Compounds labeled with isotopes are also included in Compound (I).
- the compound (I) of the present invention or a prodrug thereof (hereinafter sometimes abbreviated as the compound of the present invention) has a proton pump inhibitory action and effectively suppresses secretion of gastric acid.
- it has low toxicity (for example, acute toxicity, chronic toxicity, genotoxicity, reproductive toxicity, cardiotoxicity, drug interaction, carcinogenicity, etc.), high water solubility, stability, pharmacokinetics (absorbability, Distribution, metabolism , Excretion, etc.), and is also useful as a medicine because of its excellent drug efficacy.
- the compound of the present invention can be used in mammals (eg, humans, monkeys, hidges, rabbits, horses, dogs, cats, rabbits, rats, mice, etc.) peptic ulcers (eg, gastric ulcers, gastric ulcers due to postoperative stress, Duodenal ulcers, anastomotic ulcers, ulcers caused by non-steroidal anti-inflammatory agents, etc.); gastritis; erosive esophagitis; non-erosive esophagitis; reflux esophagitis such as erosive reflux esophagitis; non-erosive Symptomatic gastric diet such as gastroesophageal reflux disease or gastroesophageal reflux disease without esophagitis
- mammals eg, humans, monkeys, hidges, rabbits, horses, dogs, cats, rabbits, rats, mice, etc.
- peptic ulcers eg, gastric ulcers, gastric ulcers due to postoperative stress, Duodenal ulcers, ana
- Symptomatic Liastroesophageal Reflux Disease Symptomatic GERD
- Functional Dyspepsia including NUD (Non Ulcer Dyspepsia)
- Gastric cancer Interleukin _ 1 gene polymorphism promotes interleukin _ 1 j3 production Gastric MALT lymphoma; Zollinger-Ellison syndrome; hyperacidity; peptic ulcer, acute stress ulcer, hemorrhagic gastritis or invasive stress (major surgery requiring intensive management after surgery) And upper gastrointestinal bleeding due to cerebrovascular disorders requiring intensive care, head trauma, multiple organ failure, and extensive burns); Airway Disorders; Asthma treatment and prevention It is useful for administration before anesthesia, for eradication of Helicopacter pylori or for assistance in sterilization.
- GERD Symptomatic gastroesophageal reflux disease
- the content of the compound of the present invention in the pharmaceutical composition of the present invention is about 0.01% or 100% by weight of the total composition.
- the dose varies depending on the administration subject, administration route, disease, etc., but for example, when administered orally to an adult (60 kg) as an anti-ulcer agent, the effective component is about 0.5 to about 1500 mg. / ⁇ , preferably about 5 to about 150 mg / ⁇ .
- the compound of the present invention may be administered once a day or divided into 2 to 3 times a day.
- the compound of the present invention has low toxicity as it is or according to a method known per se, a pharmaceutical composition mixed with a pharmacologically acceptable carrier, such as tablets (including sugar-coated tablets and film-coated tablets), powders, granules. , Capsules (including soft capsules), orally disintegrating tablets, orally disintegrating films, solutions, injections, suppositories, sustained-release agents, patch preparations, Can be safely administered orally or parenterally (eg, topical, rectal, intravenous, etc.)
- a pharmaceutical composition mixed with a pharmacologically acceptable carrier such as tablets (including sugar-coated tablets and film-coated tablets), powders, granules. , Capsules (including soft capsules), orally disintegrating tablets, orally disintegrating films, solutions, injections, suppositories, sustained-release agents, patch preparations, Can be safely administered orally or parenterally (eg, topical, rectal, intravenous, etc.)
- Examples of the pharmacologically acceptable carrier that may be used in the production of the pharmaceutical composition of the present invention include various organic or inorganic carrier substances that are commonly used as pharmaceutical materials, such as excipients in solid formulations, Lubricants, binders, disintegrants, water-soluble polymers, basic inorganic salts; solvents, solubilizers, suspending agents, isotonic agents, buffers, soothing agents, etc. in liquid preparations.
- additives such as ordinary preservatives, antioxidants, coloring agents, sweeteners, sour agents, foaming agents, and fragrances can be used as necessary.
- excipient examples include lactose, sucrose, D-mannitol, starch, corn starch, crystalline cellulose, light anhydrous key acid, titanium oxide and the like.
- lubricant examples include magnesium stearate, sucrose fatty acid ester, polyethylene glycol, talc, stearic acid and the like.
- binder examples include hydroxypropylcellulose, hydroxypropylmethylcellulose, crystalline cellulose, starch, polyvinylpyrrolidone, gum arabic powder, gelatin, pullulan, low-substituted hydroxypropylcellulose, and the like.
- Examples of the “disintegrant” include (1) crospovidone, (2) croscanolemellose sodium (FMC—Asahi Kasei), carmellose calcium (Gotoku Pharmaceutical) and other superdisintegrants, and (3) carboxy Examples include methyl starch sodium (eg, Matsutani Chemical Co., Ltd.), (4) low-substituted hydroxypropyl cellulose (eg, Shin-Etsu Chemical Co., Ltd.), and (5) corn starch.
- the “crospovidone” includes polyvinylpolypyrrolidone (PVPP), 1-vininole-2-pyrrolidinone homopolymer, and the chemical name 1 ethenyl-2-pyrrolidinone homopolymer.
- polymers that can be used include Kollidon CL (manufactured by BASF), Polyplastidone XL (manufactured by ISP), Polyplastidone XL-1 10 (manufactured by ISP), Polyplastidone INF-10 (manufactured by ISP).
- water-soluble polymer examples include, for example, ethanol-soluble water-soluble polymers [eg, cellulose derivatives such as hydroxypropylcellulose (hereinafter sometimes referred to as HPC), polybulurpyrrolidone, etc.], ethanol Insoluble water-soluble polymers [for example, hydroxypropyl methylcellulose (hereinafter sometimes referred to as HPMC), methylcellulose, strength Cellulose derivatives such as sodium ruboxymethylcellulose, sodium polyacrylate, polyvinyl alcohol, sodium alginate, guar gum and the like].
- HPC hydroxypropylcellulose
- HPMC hydroxypropyl methylcellulose
- methylcellulose strength Cellulose derivatives such as sodium ruboxymethylcellulose, sodium polyacrylate, polyvinyl alcohol, sodium alginate, guar gum and the like.
- basic inorganic salt examples include basic inorganic salts of sodium, potassium, magnesium and / or calcium. Preferred are basic inorganic salts of magnesium and Z or calcium.
- the basic inorganic salt of magnesium is a basic inorganic salt of magnesium.
- the basic inorganic salt of sodium include sodium carbonate, sodium hydrogen carbonate, disodium hydrogen phosphate and the like.
- Examples of the basic inorganic salt of potassium include potassium carbonate and potassium hydrogen carbonate.
- Examples of the basic inorganic salt of magnesium include heavy magnesium carbonate, magnesium carbonate, magnesium oxide, magnesium hydroxide, magnesium aluminate metasilicate, magnesium silicate, magnesium aluminate, synthetic hydrotalcite [Mg Al (OH )-CO ⁇ 4 ⁇ 0) and hydroxylation
- Alumina preferably heavy magnesium carbonate, magnesium carbonate, magnesium oxide, magnesium hydroxide and the like.
- Examples of the basic inorganic salt of calcium include precipitated calcium carbonate and calcium hydroxide.
- solvent examples include water for injection, alcohol, propylene glycol, macrogol, sesame oil, corn oil, olive oil and the like.
- dissolution aid examples include polyethylene glycol, propylene glycol, D-mannitol, benzyl benzoate, ethanol, trisaminomethane, cholesterol, triethanolamine, sodium carbonate, sodium citrate and the like.
- the “suspending agent” examples include a surfactant lj such as stearyl triethanolamine, sodium lauryl sulfate, laurylaminopropionic acid, lecithin, benzalkonium chloride, benzethonium chloride, glyceryl monostearate;
- a surfactant lj such as stearyl triethanolamine, sodium lauryl sulfate, laurylaminopropionic acid, lecithin, benzalkonium chloride, benzethonium chloride, glyceryl monostearate
- hydrophilic polymers such as polybulol alcohol, polyvinylol pyrrolidone, canoleboxy methylenoresenorelose sodium, methinoresenorelose, hydroxymethinoresenorelose, hydroxyethinoresenorelose, hydroxypropinoresenorelose Etc.
- isotonic agent examples include glucose, D-sorbitol, sodium chloride, glycerin, D-mannitol and the like.
- buffering agent examples include buffer solutions of phosphate, acetate, carbonate, kenate and the like. Etc.
- Examples of the “soothing agent” include benzinole alcohol.
- preservative examples include p-hydroxybenzoates, chlorobutanol, benzyl alcohol, phenethyl alcohol, dehydroacetic acid, sorbic acid and the like.
- antioxidant for example, sulfite, ascorbic acid, human cofrol and the like can be mentioned.
- Examples of the “colorant” include edible pigments such as edible yellow No. 5, edible red No. 2, and edible blue No. 2; edible lake pigments, bengara and the like.
- sweetening agent examples include saccharin sodium, dipotassium glycyrrhizin, aspartame, stevia, thaumatin and the like.
- sour agent examples include citrate (anhydrous citrate), tartaric acid, malic acid and the like.
- Examples of the “foaming agent” include sodium bicarbonate.
- any of a synthetic product and a natural product may be used, for example, lemon, lime, orange, menthol, strawberry and the like.
- the compound of the present invention is compression-molded according to a method known per se, for example, by adding a carrier such as an excipient, a disintegrant, a binder or a lubricant, and then, if necessary, masking of taste, entericity or For long-lasting purposes, it can be made into an orally administered preparation by coating by a method known per se.
- a carrier such as an excipient, a disintegrant, a binder or a lubricant
- an intermediate layer can be provided between the enteric layer and the drug-containing layer by a method known per se for the purpose of separating both layers.
- a core containing crystalline cellulose and lactose is coated with the compound of the present invention and, if necessary, a basic inorganic salt, and further coated with a coating layer containing a water-soluble polymer.
- a composition and the resulting composition is coated with a polyethylene glycol-containing enteric coating layer, then coated with a triethyl citrate-containing enteric coating layer, and further coated with a polyethylene glycol-containing enteric coating layer, Finally, it is coated with mannitol to obtain fine particles, and the resulting fine particles and additives are mixed and manufactured.
- enteric coating layer examples include cellulose acetate phthalate (CAP), hydroxypropylmethylcellulose phthalate, hydroxymethylcellulose acetate succinate, methacrylic acid copolymer [for example, Eudragit L30D-5 5 (Product Name; manufactured by Laem Co., Ltd.), Kollicoat MAE30DP (trade name; manufactured by BASF), polyquid PA 30 (trade name; manufactured by Sanyo Kasei Co., Ltd.), etc.], water-based enteric polymer bases such as carboxymethylethyl cellulose and shellac; Sustained release bases such as methacrylic acid copolymers [eg Eudragit NE30D (trade name), Eudragit RL30D (trade name), Eudragit RS30D (trade name), etc.]; water-soluble polymers; triethyl taenoate, polyethylene glycol , Acetylene monoglyceride, triacetin, castor oil and other plasticizers It includes a layer formed of
- additives examples include water-soluble sugar alcohols (eg, sorbitol, mannitol and maltitol, reduced starch saccharified product, xylitol, reduced palatinose, erythritol, etc.), crystalline cellulose (eg, Serus KG 801, Avicenore PH 101, Avicel PH 102, Ryohi, Senore PH 301, Ryohi, Senore Pti 302, Abi'Senore RC-591 (ligated senore mouth carmellose sodium), etc.), low substitution degree Hydroxypropyl cellulose (eg, LH-22, LH-32, LH-23, LH-33 (Shin-Etsu Chemical Co., Ltd.) and mixtures thereof), etc., and binders, acidulants, foaming agents, sweetness, etc. Agents, fragrances, lubricants, colorants, stabilizers, excipients, disintegrants, and the like
- the compound of the present invention may be used in combination with 1 to 3 other active ingredients.
- anti-helicopacter pylori active substances examples include anti-helicopacter pylori active substances, imidazole compounds, bismuth salts, quinolone compounds, and the like.
- anti-helicopacter pylori active substance examples include penicillin antibiotics (eg, amoxicillin, benzylpenicillin, piperacillin, mecillinam, etc.), cefme antibiotics (eg, cefixime, cefaclor etc.), macrolides Antibiotics (eg, erythromycin, clarithromycin, etc.), tetracycline antibiotics (eg, tetracycline, minocycline, streptomycin, etc.), aminoglycoside antibiotics (eg, gentamicin, amikacin, etc.), imipenem, etc. Can be mentioned. Of these, penicillin antibiotics and macrolide antibiotics are preferred.
- Examples of the "imidazole compound” include metronidazole, miconazole and the like.
- bismuth salt examples include bismuth acetate, bismuth citrate and the like.
- quinolone compound examples include ofloxacin, cyproxacin and the like.
- the compound (I) of the present invention or a salt thereof a penicillin antibiotic (eg, amoxicillin, etc.) and an erythromycin antibiotic (eg, clarithromycin, etc.) Preferably used.
- a penicillin antibiotic eg, amoxicillin, etc.
- an erythromycin antibiotic eg, clarithromycin, etc.
- the compounds of the present invention can enhance the antibacterial action of other antibiotics by regulating action and the like of the gastric P H, the auxiliary of based rather sterilization effect the action of antibiotics in combination It also plays a role.
- the “other active ingredient” and the compound (I) of the present invention or a salt thereof are mixed according to a method known per se, and a single pharmaceutical composition (eg, tablet, powder, granule, capsule (soft (Including pills), liquids, injections, suppositories, sustained-release preparations, etc.), each of which can be formulated separately and formulated separately, with the same subject at the same time or with a time difference May be administered.
- a single pharmaceutical composition eg, tablet, powder, granule, capsule (soft (Including pills), liquids, injections, suppositories, sustained-release preparations, etc.
- the compound of the present invention is a gastrointestinal motility-promoting drug, a drug acting on the lower esophageal sphincter (eg, a transient lower esophageal sphincter relaxation inhibitor, etc.), C1C 2 channel opener (C1C-2 channel opener) ( Intestinal fluid secretagogues), histamine H receptor antagonists, antacids, sedatives, digestion of the stomach
- NSAIDs non-steroidal anti-inflammatory drugs
- gastrointestinal motility promoter examples include domperidone, metocloblamide, mosapride, itopride, tegaserod and the like.
- Examples of the “drug that acts on the lower esophageal sphincter” include GABA-B receptor agonists such as baclofen and its optically active substance.
- C1C — 2 channel opener intestinal fluid secretion promoter
- rubiprostone intestinal fluid secretion promoter
- histamine H receptor antagonist examples include cimetidine, ranitidine, famot Gin, oral xanthidine, nizatidine, lafutidine and the like.
- Examples of the “antacid” include sodium hydrogen carbonate, aluminum hydroxide and the like.
- Examples of the “sedative” include diazepam, chlordiazepoxide and the like.
- Examples of the “healthy stomach digestive agent” include gentian, sempri, diastase and the like.
- non-steroidal anti-inflammatory agent examples include aspirin, indomethacin, ibuprofen, mefenamic acid, diclofenac, etodolac, piroxicam, celecoxib and the like.
- Gastrointestinal motility stimulants drugs that act on the lower esophageal sphincter, C1C-1 2-channel openers (intestinal secretion promoters), histamine H receptor antagonists, antacids, sedatives, and stomach digestives
- a non-steroidal anti-inflammatory agent and the compound (I) of the present invention or a salt thereof are mixed according to a method known per se, and a single pharmaceutical composition (eg, tablet, powder, granule, capsule (soft capsule Including liquid preparations, injections, suppositories, sustained-release preparations, etc.), which may be used together and may be formulated separately and administered to the same subject at the same time or with a time difference .
- a single pharmaceutical composition eg, tablet, powder, granule, capsule (soft capsule Including liquid preparations, injections, suppositories, sustained-release preparations, etc.
- the compound of the present invention may also be used in combination with the following drugs.
- proton pump inhibitors eg, omeprazole, esomeprazole, huntophunol, pantoprazole, rabeprazole, tenatof. Razorole (tenatoprazole), ilaprazole (ilaprazole) and lansoprazo
- oral antacid mixtures eg, Maalox, Aludrox and Gaviscon;
- (Hi) mucosal protective agents e.g. polaprezinc, ecabe so dium, reno mipide (rebamipide li, tef. Renon eprenone), cetraxa te, sucralfate, Chloropylline (copper) and plaunotol;
- anti-gastric 1J eg, anti-gastrin vaccine, itriglumide and Z-360;
- 5-HT antagonists eg dolasetron, palonosetron tron), alosetron, azasetron, ramosetron, mitrazapine, granisetron, tro etron;
- GABA agonists eg, baclofen and AZD_3355;
- GABA antagonists eg, GAS-360 and SGS-742;
- Calcium channel blockers eg, aranidipine, lac idipine, falodipine, azelnidipine, clinidipine, clinidipine, lomerizine, diltiazem, sword gall opamil), efonidipine, nisoldipine, amlodipine, lercanidipine, bevantolol, benitolol, nicardipine, isradipine, benidipine, benidipine (verapamil), nitrendipine, barnidipine, pronophenone, manidipine, bepridil, nifedipine, nilvadipine, nodivadipine, nodivadipine, imodipine f asudil);
- aranidipine lac idipine, falodipine, azelnidipine, clinidipine, clinidipine, lomerizine, diltiaze
- Dopamine antagonists eg, metoclopramide, domperidone and levosnolech. Lito Qevosulpiride);
- tachykun (NK) antagonists in particular NK_3, NK-2 and NK_1 Antagonists, eg, nepadutant, saledutant, talenetant, (hi R, 9R) _ 7 _ [3,5 _Bis (trifluoromethyl) benzyl] _ 8, 9, 10
- nitric oxide synthase inhibitor eg, GW—274150, tilargine, P54, guanidioethyldisulfide and nitroflurbiprofen;
- ghrelin agonists eg, force promorelin and TZP-101
- AchE release stimulants eg, Z_338 and KW_5092.
- the above drugs (i) to (xvi) and the compound (I) or a salt thereof of the present invention are mixed according to a method known per se, and a single pharmaceutical composition (for example, tablet, powder, granule, capsule) (Including soft capsules), liquids, injections, suppositories, sustained-release preparations, etc.) may be formulated and used together.
- a single pharmaceutical composition for example, tablet, powder, granule, capsule
- liquids liquids
- “Room temperature” in the following Reference Examples and Examples usually indicates about 10 ° C. to about 35 ° C., but is not particularly strictly limited. The mixing ratio of the liquid indicates a volume ratio. “%” Indicates weight percent unless otherwise specified. However, the yield is mol / mol%.
- Silica gel column chromatography was performed using MERCK silica gel 60 (0.063-0.200 mm) or Fuji Silica Chemical Co., Ltd. Chromatorex (trade name) NH (denoted as basic silica gel column chromatography).
- iH—NMR spectrum uses tetramethylsilane as internal standard, Varian Gemini—200 (200 MHz), Mercury—300 (300 MHz), Smmeter, Bmker AVANCE AV300 (300 MHz) and JNM—AL400 (400 MHz) nuclear magnetism Using a resonance device ⁇ IEOL DATUM (JEOL Datum) Measured. The following abbreviations are used to express the measurement results.
- s singlet
- d doublet
- dd double do ublet
- dt doublet triplet
- t triplet
- q quartet
- m Manople plet (multiplet)
- br Frozen (broad)
- brs Broad singlet
- J coupling constant
- Hz Hertz.
- the extract was washed with a saturated aqueous sodium hydrogen carbonate solution, water and saturated brine in that order, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure.
- N-dimethylformamide 10 mL was added dropwise. After completion of the dropwise addition, the reaction solution was stirred at room temperature for 30 minutes and cooled with ice-cold p-toluenesulfonyl chloride (3.16 g). In N, N dimethylformamide (l OmL). After completion of the dropwise addition, the reaction solution was stirred at room temperature for 1 hour and concentrated under reduced pressure.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Communicable Diseases (AREA)
- Hematology (AREA)
- Oncology (AREA)
- Diabetes (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Thiazole And Isothizaole Compounds (AREA)
- Pyrrole Compounds (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/225,851 US7994205B2 (en) | 2006-03-31 | 2007-03-30 | Aryl-or heteroaryl-sulfonyl compounds as acid secretion inhibitors |
| EP07740538A EP2005957B1 (en) | 2006-03-31 | 2007-03-30 | Acid secretion inhibitor |
| CA002647862A CA2647862A1 (en) | 2006-03-31 | 2007-03-30 | Acid secretion inhibitor |
| JP2008508655A JP5207964B2 (ja) | 2006-03-31 | 2007-03-30 | 酸分泌抑制薬 |
| US13/204,093 US8686010B2 (en) | 2006-03-31 | 2011-08-05 | Aryl- or heteroaryl-sulfonyl compounds as acid secretion inhibitors |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006-100651 | 2006-03-31 | ||
| JP2006100651 | 2006-03-31 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/225,851 A-371-Of-International US7994205B2 (en) | 2006-03-31 | 2007-03-30 | Aryl-or heteroaryl-sulfonyl compounds as acid secretion inhibitors |
| US13/204,093 Division US8686010B2 (en) | 2006-03-31 | 2011-08-05 | Aryl- or heteroaryl-sulfonyl compounds as acid secretion inhibitors |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2007114338A1 true WO2007114338A1 (ja) | 2007-10-11 |
Family
ID=38563605
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2007/057102 WO2007114338A1 (ja) | 2006-03-31 | 2007-03-30 | 酸分泌抑制薬 |
Country Status (5)
| Country | Link |
|---|---|
| US (2) | US7994205B2 (ja) |
| EP (1) | EP2005957B1 (ja) |
| JP (1) | JP5207964B2 (ja) |
| CA (1) | CA2647862A1 (ja) |
| WO (1) | WO2007114338A1 (ja) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009041447A1 (ja) | 2007-09-28 | 2009-04-02 | Takeda Pharmaceutical Company Limited | 5員複素環化合物 |
| WO2009041705A3 (en) * | 2007-09-28 | 2009-06-25 | Takeda Pharmaceutical | 5-membered heterocyclic compounds as proton pump inhibitors |
| WO2010110378A1 (ja) * | 2009-03-26 | 2010-09-30 | 武田薬品工業株式会社 | ピラゾール化合物 |
| WO2011004882A1 (ja) | 2009-07-09 | 2011-01-13 | ラクオリア創薬株式会社 | 消化管運動異常が関与する疾患を治療するためのアシッドポンプ拮抗剤 |
| US8592597B2 (en) | 2008-08-27 | 2013-11-26 | Takeda Pharmaceutical Company Limited | Pyrrole compounds |
| WO2014003199A1 (en) | 2012-06-27 | 2014-01-03 | Takeda Pharmaceutical Company Limited | Liquid preparations of amines and organic acids stabilized by salts |
| US8933105B2 (en) | 2007-02-28 | 2015-01-13 | Takeda Pharmaceutical Company Limited | Pyrrole compounds |
| EP3088397A1 (en) * | 2007-04-11 | 2016-11-02 | Canbas Co. Ltd. | Compounds that abrogate the cell cycle g2 checkpoint for use in the treatment of cancer |
| JP2019509308A (ja) * | 2016-03-25 | 2019-04-04 | デウン ファーマシューティカル カンパニー リミテッド | 1−(5−(2,4−ジフルオロフェニル)−1−((3−フルオロフェニル)スルホニル)−4−メトキシ−1h−ピロール−3−イル)−n−メチルメタンアミン塩の新規な結晶形 |
| JP2019509320A (ja) * | 2016-03-25 | 2019-04-04 | デウン ファーマシューティカル カンパニー リミテッド | 1−(5−(2,4−ジフルオロフェニル)−1−((3−フルオロフェニル)スルホニル)−4−メトキシ−1h−ピロール−3−イル)−n−メチルメタンアミンの新規な酸付加塩 |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7994205B2 (en) | 2006-03-31 | 2011-08-09 | Takeda Pharmaceutical Company Limited | Aryl-or heteroaryl-sulfonyl compounds as acid secretion inhibitors |
| EP2963019B1 (en) | 2013-02-28 | 2020-08-12 | Takeda Pharmaceutical Company Limited | Method for producing pyridine-3-sulfonyl chloride |
| WO2015004610A1 (en) | 2013-07-11 | 2015-01-15 | Adamed Sp. Z O.O. | 1,5-dihydropyrrol-2-one derivatives as inhibitors of p53-mdm2/mdm4 protein-protein interaction |
| KR102084185B1 (ko) * | 2013-08-29 | 2020-03-04 | 주식회사 대웅제약 | 테트라히드로사이클로펜타피롤 유도체 및 이의 제조방법 |
| CA2947134A1 (en) | 2014-06-12 | 2015-12-17 | Adamed Sp. Z O.O. | Compounds comprising 1,1',2,5'-tetrahydrospiro[indole-3,2'-pyrrole]-2,5'-dione system as inhibitors p53-mdm2 protein-protein interaction |
| CN105985278A (zh) * | 2015-01-27 | 2016-10-05 | 江苏柯菲平医药股份有限公司 | 吡咯磺酰类衍生物、其制备方法及其在医药上的应用 |
| GB201602934D0 (en) | 2016-02-19 | 2016-04-06 | Cancer Res Inst Royal | Compounds |
| WO2017164575A1 (en) * | 2016-03-25 | 2017-09-28 | Daewoong Pharmaceutical Co.,Ltd. | Novel acid addition salt of 1-(5-(2,4-difluorophenyl)-1-((3- fluorophenyl)sulfonyl)-4-methoxy-1h-pyrrol-3-yl)-n- methylmethanamine |
| CN108203430A (zh) * | 2016-12-16 | 2018-06-26 | 天地人和生物科技有限公司 | 一种新型可逆性质子泵抑制剂及其制备方法和用途 |
| KR102126576B1 (ko) * | 2018-09-19 | 2020-06-24 | 주식회사 대웅제약 | 4-메톡시 피롤 유도체의 제조 방법 |
| KR20210156235A (ko) | 2020-06-17 | 2021-12-24 | 일동제약(주) | 신규한 산 분비 억제제 및 이의 용도 |
| CN111943932B (zh) * | 2020-08-06 | 2023-07-14 | 四川国康药业有限公司 | 一种可以治疗消化性溃疡的3-吡啶磺酰-1-n-杂吡咯衍生物及其制备方法和用途 |
| CN113620930B (zh) * | 2021-07-12 | 2022-08-16 | 南京烁慧医药科技有限公司 | 一种含磺酰胺结构的化合物及其制备方法和应用、一种药物组合物及应用 |
| CN113735777B (zh) * | 2021-08-18 | 2023-05-02 | 上海药坦药物研究开发有限公司 | 一种制备环硫脲化合物的方法 |
| WO2023113458A1 (en) * | 2021-12-15 | 2023-06-22 | Ildong Pharmaceutical Co., Ltd. | Novel salt of 1-sulfonyl pyrrole derivative, preparation method thereof and pharmaceutical composition comprising thereof |
| WO2023113474A1 (ko) * | 2021-12-15 | 2023-06-22 | 일동제약(주) | 1-설포닐 피롤 유도체의 신규 염, 이의 제조 방법 및 이를 포함하는 약학 조성물 |
| WO2023230179A1 (en) * | 2022-05-24 | 2023-11-30 | Case Western Reserve University | Compositions and methods for detecting and treating cancer |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0259085A1 (en) | 1986-08-29 | 1988-03-09 | Pfizer Inc. | 2-Guanidino-4-arylthiazoles for treatment of peptic ulcers |
| JPH08119936A (ja) | 1994-10-18 | 1996-05-14 | Fujisawa Pharmaceut Co Ltd | 複素環式誘導体 |
| JPH0948778A (ja) * | 1995-08-04 | 1997-02-18 | Sagami Chem Res Center | 2−置換−3−アルコキシ−5−(ピロール−2−イル)フラン誘導体 |
| WO1999042463A1 (en) * | 1998-02-23 | 1999-08-26 | Warner-Lambert Company | Substituted quinoxaline derivatives as interleukin-8 receptor antagonists |
| US20020193596A1 (en) | 1995-11-06 | 2002-12-19 | University Of Pittsburgh | Inhibitors of protein isoprenyl transferases |
| WO2004007478A2 (en) | 2002-07-16 | 2004-01-22 | Aventis Pharma Deutschland Gmbh | Derivatives of 3-guanidinocarbonyl-1-heteroaryl-pyrrole, process for their preparation and their pharmaceutical use |
| WO2004103968A1 (en) | 2003-05-26 | 2004-12-02 | Aponetics Ag | Sulfopyrroles |
| JP2006100651A (ja) | 2004-09-30 | 2006-04-13 | Renesas Technology Corp | 半導体装置の製造方法 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3285991A (en) | 1963-03-26 | 1966-11-15 | Union Carbide Corp | Polyepoxides cured with the reaction product of a dihydroxydiphenylsul-fone, an amine, and an aldehyde |
| US4847289A (en) | 1987-06-08 | 1989-07-11 | Merck & Co., Inc. | Thiophene sulfonamide antiglaucoma agents |
| MX9205392A (es) | 1991-10-29 | 1993-04-01 | Du Pont | Triazolcarbozamidas herbicidas y procedimiento para su obtencion. |
| WO1996010019A1 (fr) * | 1994-09-26 | 1996-04-04 | Shionogi & Co., Ltd. | Derive imidazole |
| AU7597596A (en) | 1995-11-06 | 1997-05-29 | University Of Pittsburgh | Inhibitors of protein isoprenyl transferases |
| SK86699A3 (en) | 1996-12-23 | 2000-11-07 | Du Pont Pharm Co | Nitrogen containing heteroaromatics as factor xa inhibitors |
| US7226732B2 (en) | 2001-07-16 | 2007-06-05 | Cepheid | Methods, apparatus, and computer programs for verifying the integrity of a probe |
| CA2493618A1 (en) * | 2002-08-01 | 2004-02-12 | Nitromed, Inc. | Nitrosated proton pump inhibitors, compositions and methods of use |
| CA2532800C (en) | 2003-07-23 | 2013-06-18 | Exelixis, Inc. | Anaplastic lymphoma kinase modulators and methods of use |
| JP5173192B2 (ja) | 2004-09-30 | 2013-03-27 | 武田薬品工業株式会社 | プロトンポンプ阻害薬 |
| US7994205B2 (en) | 2006-03-31 | 2011-08-09 | Takeda Pharmaceutical Company Limited | Aryl-or heteroaryl-sulfonyl compounds as acid secretion inhibitors |
-
2007
- 2007-03-30 US US12/225,851 patent/US7994205B2/en active Active
- 2007-03-30 WO PCT/JP2007/057102 patent/WO2007114338A1/ja active Application Filing
- 2007-03-30 CA CA002647862A patent/CA2647862A1/en not_active Abandoned
- 2007-03-30 EP EP07740538A patent/EP2005957B1/en active Active
- 2007-03-30 JP JP2008508655A patent/JP5207964B2/ja active Active
-
2011
- 2011-08-05 US US13/204,093 patent/US8686010B2/en active Active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0259085A1 (en) | 1986-08-29 | 1988-03-09 | Pfizer Inc. | 2-Guanidino-4-arylthiazoles for treatment of peptic ulcers |
| JPH08119936A (ja) | 1994-10-18 | 1996-05-14 | Fujisawa Pharmaceut Co Ltd | 複素環式誘導体 |
| JPH0948778A (ja) * | 1995-08-04 | 1997-02-18 | Sagami Chem Res Center | 2−置換−3−アルコキシ−5−(ピロール−2−イル)フラン誘導体 |
| US20020193596A1 (en) | 1995-11-06 | 2002-12-19 | University Of Pittsburgh | Inhibitors of protein isoprenyl transferases |
| WO1999042463A1 (en) * | 1998-02-23 | 1999-08-26 | Warner-Lambert Company | Substituted quinoxaline derivatives as interleukin-8 receptor antagonists |
| WO2004007478A2 (en) | 2002-07-16 | 2004-01-22 | Aventis Pharma Deutschland Gmbh | Derivatives of 3-guanidinocarbonyl-1-heteroaryl-pyrrole, process for their preparation and their pharmaceutical use |
| WO2004103968A1 (en) | 2003-05-26 | 2004-12-02 | Aponetics Ag | Sulfopyrroles |
| JP2006100651A (ja) | 2004-09-30 | 2006-04-13 | Renesas Technology Corp | 半導体装置の製造方法 |
Non-Patent Citations (19)
| Title |
|---|
| "Jikken Kagaku Koza (Courses in Experimental Chemistry", vol. 21, 1991, MARUZEN PRESS, pages: 106 - 124 |
| "Jikken Kagaku Koza", vol. 14-III, 1978, MARUZEN PRESS, pages: 1380 - 1385 |
| "Molecule Design", vol. 7, 1990, HIROKAWA PUBLISHING CO, article "Pharmaceutical Research and Development", pages: 163 - 198 |
| HALAZY S., TETRAHEDRON LETTERS, vol. 25, no. 14, 1984, pages 1421 - 1424, XP003017186 * |
| J. HETEROCYCLIC. CHEM, vol. 13, 1976, pages 1145 |
| J. MED. CHEM., vol. 35, 1992, pages 4195 |
| J. MED. CHEM., vol. 43, 2000, pages 2165 |
| J. ORG. CHEM., vol. 39, 1974, pages 1290 |
| J. ORG. CHEM., vol. 46, 1981, pages 2596 |
| J.C.S. CHEM. COMMUN., 1983, pages 26 |
| ORG. LETT., vol. 3, 2001, pages 1261 |
| ORG. LETT., vol. 6, 2004, pages 4587 |
| See also references of EP2005957A4 |
| SYNLETT, 2004, pages 1254 |
| SYNTHESIS, 1994, pages 639 |
| TETRAHEDRON, vol. 46, 1990, pages 7587 |
| TETRAHEDRON, vol. 57, 2001, pages 7813 |
| THEODORA W. GREENE; PETER G. M. WUTS: "Protective Groups in Organic Synthesis", 1999, WILEY-INTERSCIENCE |
| WALLMARK, BIOCHIM. BIOPHYS. ACTA, vol. 728, 1983, pages 31 |
Cited By (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8933105B2 (en) | 2007-02-28 | 2015-01-13 | Takeda Pharmaceutical Company Limited | Pyrrole compounds |
| EP3567035A1 (en) * | 2007-04-11 | 2019-11-13 | Canbas Co. Ltd. | N-substituted 2,5-dioxo-azoline compounds for use in the treatment of cancer |
| EP3088397A1 (en) * | 2007-04-11 | 2016-11-02 | Canbas Co. Ltd. | Compounds that abrogate the cell cycle g2 checkpoint for use in the treatment of cancer |
| WO2009041705A3 (en) * | 2007-09-28 | 2009-06-25 | Takeda Pharmaceutical | 5-membered heterocyclic compounds as proton pump inhibitors |
| JP2010540412A (ja) * | 2007-09-28 | 2010-12-24 | 武田薬品工業株式会社 | プロトンポンプ阻害剤としての5員複素環化合物 |
| EP3162798A1 (en) | 2007-09-28 | 2017-05-03 | Takeda Pharmaceutical Company Limited | 5-membered heterocyclic compound |
| US8334301B2 (en) | 2007-09-28 | 2012-12-18 | Takeda Pharmaceutical Company Limited | 5-Membered heterocyclic compound |
| WO2009041447A1 (ja) | 2007-09-28 | 2009-04-02 | Takeda Pharmaceutical Company Limited | 5員複素環化合物 |
| US8592597B2 (en) | 2008-08-27 | 2013-11-26 | Takeda Pharmaceutical Company Limited | Pyrrole compounds |
| US8993598B2 (en) | 2008-08-27 | 2015-03-31 | Takeda Pharmaceutical Company Limited | Pyrrole compounds |
| US8969387B2 (en) | 2008-08-27 | 2015-03-03 | Takeda Pharmaceutical Company Limited | Pyrrole compounds |
| JP5552481B2 (ja) * | 2009-03-26 | 2014-07-16 | 武田薬品工業株式会社 | ピラゾール化合物 |
| US8895588B2 (en) | 2009-03-26 | 2014-11-25 | Takeda Pharmaceutical Company Limited | Pyrazole compound |
| EA019853B1 (ru) * | 2009-03-26 | 2014-06-30 | Такеда Фармасьютикал Компани Лимитед | Производные 1-(пиридин-3-ил)-3-метиламинометил-5-фенилсульфонилпиразола |
| KR101656906B1 (ko) | 2009-03-26 | 2016-09-12 | 다케다 야쿠힌 고교 가부시키가이샤 | 피라졸 화합물 |
| KR20110130483A (ko) | 2009-03-26 | 2011-12-05 | 다케다 야쿠힌 고교 가부시키가이샤 | 피라졸 화합물 |
| WO2010110378A1 (ja) * | 2009-03-26 | 2010-09-30 | 武田薬品工業株式会社 | ピラゾール化合物 |
| WO2011004882A1 (ja) | 2009-07-09 | 2011-01-13 | ラクオリア創薬株式会社 | 消化管運動異常が関与する疾患を治療するためのアシッドポンプ拮抗剤 |
| WO2014003199A1 (en) | 2012-06-27 | 2014-01-03 | Takeda Pharmaceutical Company Limited | Liquid preparations of amines and organic acids stabilized by salts |
| JP2019509308A (ja) * | 2016-03-25 | 2019-04-04 | デウン ファーマシューティカル カンパニー リミテッド | 1−(5−(2,4−ジフルオロフェニル)−1−((3−フルオロフェニル)スルホニル)−4−メトキシ−1h−ピロール−3−イル)−n−メチルメタンアミン塩の新規な結晶形 |
| JP2019509320A (ja) * | 2016-03-25 | 2019-04-04 | デウン ファーマシューティカル カンパニー リミテッド | 1−(5−(2,4−ジフルオロフェニル)−1−((3−フルオロフェニル)スルホニル)−4−メトキシ−1h−ピロール−3−イル)−n−メチルメタンアミンの新規な酸付加塩 |
Also Published As
| Publication number | Publication date |
|---|---|
| US7994205B2 (en) | 2011-08-09 |
| EP2005957A1 (en) | 2008-12-24 |
| CA2647862A1 (en) | 2007-10-11 |
| US20110288040A1 (en) | 2011-11-24 |
| JP5207964B2 (ja) | 2013-06-12 |
| US8686010B2 (en) | 2014-04-01 |
| US20090118335A1 (en) | 2009-05-07 |
| JPWO2007114338A1 (ja) | 2009-08-20 |
| EP2005957B1 (en) | 2012-05-30 |
| EP2005957A4 (en) | 2010-11-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2007114338A1 (ja) | 酸分泌抑制薬 | |
| JP5638515B2 (ja) | ピロール化合物 | |
| KR101559597B1 (ko) | 5―원 헤테로시클릭 화합물 | |
| WO2006036024A1 (ja) | プロトンポンプ阻害薬 | |
| JP5552481B2 (ja) | ピラゾール化合物 | |
| JP2003313186A (ja) | ベンズイミダゾール化合物、その製造法およびその用途 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07740538 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2008508655 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2647862 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12225851 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007740538 Country of ref document: EP |