WO2007085572A2 - Mischungen von faserreaktiven farbstoffen, ihre herstellung und verwendung - Google Patents
Mischungen von faserreaktiven farbstoffen, ihre herstellung und verwendung Download PDFInfo
- Publication number
- WO2007085572A2 WO2007085572A2 PCT/EP2007/050560 EP2007050560W WO2007085572A2 WO 2007085572 A2 WO2007085572 A2 WO 2007085572A2 EP 2007050560 W EP2007050560 W EP 2007050560W WO 2007085572 A2 WO2007085572 A2 WO 2007085572A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- dye
- hydrogen
- independently
- vinylsulfone
- general formula
- Prior art date
Links
- 0 C*1/C=C/C(/Nc2nc(C)nc(Cl)n2)=C\C(C)(C)/C=C1 Chemical compound C*1/C=C/C(/Nc2nc(C)nc(Cl)n2)=C\C(C)(C)/C=C1 0.000 description 1
- BMVXCPBXGZKUPN-UHFFFAOYSA-N CCCCCCN Chemical compound CCCCCCN BMVXCPBXGZKUPN-UHFFFAOYSA-N 0.000 description 1
- HBQWSZQNYZVWNR-UHFFFAOYSA-N CCCc1nc(NC2=CC(C)(C)C=CC(C)(C)C=C2)nc(C)n1 Chemical compound CCCc1nc(NC2=CC(C)(C)C=CC(C)(C)C=C2)nc(C)n1 HBQWSZQNYZVWNR-UHFFFAOYSA-N 0.000 description 1
- DMRMPFXMHGWUJA-UHFFFAOYSA-N Cc1nc(NCCC=C)nc(NCCC=C)n1 Chemical compound Cc1nc(NCCC=C)nc(NCCC=C)n1 DMRMPFXMHGWUJA-UHFFFAOYSA-N 0.000 description 1
- UCJDCGANFAKTKA-UHFFFAOYSA-N Cc1ncnc(C)n1 Chemical compound Cc1ncnc(C)n1 UCJDCGANFAKTKA-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
- C09B67/0041—Blends of pigments; Mixtured crystals; Solid solutions mixtures containing one azo dye
- C09B67/0042—Mixtures containing two reactive dyes one of them being an azo dye
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B1/00—Dyes with anthracene nucleus not condensed with any other ring
- C09B1/16—Amino-anthraquinones
- C09B1/20—Preparation from starting materials already containing the anthracene nucleus
- C09B1/26—Dyes with amino groups substituted by hydrocarbon radicals
- C09B1/32—Dyes with amino groups substituted by hydrocarbon radicals substituted by aryl groups
- C09B1/34—Dyes with amino groups substituted by hydrocarbon radicals substituted by aryl groups sulfonated
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B45/00—Complex metal compounds of azo dyes
- C09B45/02—Preparation from dyes containing in o-position a hydroxy group and in o'-position hydroxy, alkoxy, carboxyl, amino or keto groups
- C09B45/04—Azo compounds in general
- C09B45/08—Copper compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
- C09B67/0041—Blends of pigments; Mixtured crystals; Solid solutions mixtures containing one azo dye
- C09B67/0042—Mixtures containing two reactive dyes one of them being an azo dye
- C09B67/0045—Mixtures containing two reactive dyes one of them being an azo dye both having the reactive group not directly attached to a heterocyclic system
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
- C09B67/0046—Mixtures of two or more azo dyes
- C09B67/0055—Mixtures of two or more disazo dyes
- C09B67/0057—Mixtures of two or more reactive disazo dyes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
- C09B67/0046—Mixtures of two or more azo dyes
- C09B67/0055—Mixtures of two or more disazo dyes
- C09B67/0057—Mixtures of two or more reactive disazo dyes
- C09B67/0059—Mixtures of two or more reactive disazo dyes all the reactive groups are not directly attached to a heterocyclic system
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/02—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using azo dyes
- D06P1/10—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using azo dyes containing metal
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06P—DYEING OR PRINTING TEXTILES; DYEING LEATHER, FURS OR SOLID MACROMOLECULAR SUBSTANCES IN ANY FORM
- D06P1/00—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed
- D06P1/38—General processes of dyeing or printing textiles, or general processes of dyeing leather, furs, or solid macromolecular substances in any form, classified according to the dyes, pigments, or auxiliary substances employed using reactive dyes
Definitions
- the present invention is in the field of reactive dyes and relates to dye mixtures suitable for dyeing and printing fiber materials having hydroxyl or amide groups.
- Fiber materials described, for example, EP 1 275 700 A2 and DE 10 2004 028 919 A1 are mentioned.
- these conventional dyes especially in deep black and navy shades, do not adequately meet the latest stringent demands on the fastness properties of the dyed or printed products.
- the present invention has now found dye mixtures whose dyeings surprisingly have significantly better fastness properties compared to the dye mixtures described in EP 1 275 700 A2 and DE 10 2004 028 919 A1, the chlorine fastness being particularly noteworthy. Furthermore, an improved build-up behavior of the mixtures according to the invention over the individual dyes of the mixture is achieved.
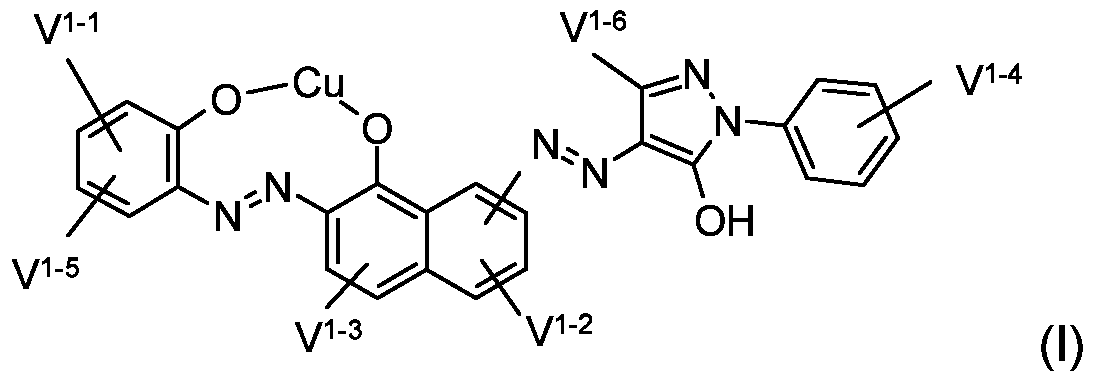
- the present invention relates to dye mixtures which are characterized in that they contain at least one dye of the general formula (I)
- V 1 "2 and V 1" 3 are independently hydrogen or -SO 3 M;
- V 1 "4 represents hydrogen, -SO 3 M, vinylsulfone or ethylsulfone which is substituted in the ⁇ -position by a substituent eliminable by the action of alkali;
- V, 1 I -6 D is hydrogen, (C r C 6 ) -alkyl, -COOM, -COO- (C r C 4 ) -alkyl I;
- M is hydrogen, alkali, ammonium or the equivalent of an alkaline earth metal ion
- R 2 "1 to R 2" 4 independently of one another represent hydrogen, -SO 3 M, (C 1 -C 4 ) -alkyl or -COOR 2 "5 ;
- W 2 represents -NR 2 6 D 2 ;
- R 2" 5 and R 2 " 6 independently of one another are hydrogen or (C 1 -C 4 ) -alkyl;
- D 2 is one of the following groups
- V 2 "1 and V 2" 2 are each independently hydrogen, -SO 3 M, vinylsulfone or
- V 2 "4 and V 2" 6 are independently hydrogen or -SO 3 M;
- W 3 "1 and W 3 '2 are independently hydrogen, (C r C4) alkyl, (C r C4) alkyl which is substituted by -OSO 3 M, or one of the following groups
- V 3 "1 to V 3" 12 independently of one another represent hydrogen, -SO 3 M, vinylsulfone or ethylsulfone which can be eliminated in the ⁇ -position by an action which can be eliminated by the action of alkali Substituted substituent; and n is 1, 2, 3 or 4; and M and HaI are as defined above; wherein the dye of the general formula (IM), if V 3 "1 to V 3" 12 independently of one another represent hydrogen or -SO 3 M, at least one structural element of the formula
- Y 4 "2 has the same meaning as Y 4" 1 or for one of the following groups
- W, 4 j is vinylsulfone or ethylsulfone which is substituted in the ⁇ position by a substituent eliminable by exposure to alkali or -NR 4 "1 D 4 ;
- D 4 is one of the following groups
- V 4 "1 to V 4" 4 and V 4 "9 to V 4" 10 independently of one another represent hydrogen or -SO 3 M;
- R 4 "1 is hydrogen, methyl or ethyl
- V 4" is 5 to V 4 "7 independently of one another hydrogen, -SO 3 M, vinylsulfone or
- V 4 "8 represents vinylsulfone or ethylsulfone substituted in ⁇ -position by an alkali-eliminable substituent, and and M is as defined above.
- (C 1 -C 4 ) -alkyl or (C 1 -C 6 ) -alkyl groups may be straight-chain or branched and are, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, i-butyl or sec-butyl.
- (C 1 -C 6 ) -alkyl groups can also be used, for example
- Pentyl or hexyl stand.
- Preferred (C 1 -C 4 ) -alkyl or (C 1 -C 6 ) -alkyl groups are
- (C 1 -C 6 ) -alkoxy groups which accordingly may be, for example, methoxy, ethoxy, n-propoxy or isopropoxy and methoxy being particularly preferred.
- alkali-eliminable substituents in the ⁇ -position of ethylsulfone are chlorine and -OSO 3 M, especially -OSO 3 M.
- alkali metals are in particular sodium, potassium and lithium.
- Alkaline earth metals whose equivalent can stand for M are in particular calcium and magnesium.
- M is preferably hydrogen, sodium and potassium.
- Halogen is in particular fluorine, chlorine and bromine.
- substituents substituted for vinylsulfone or ethylsulfone which is substituted in the ⁇ -position by an alkali-eliminable substituent may have both meanings, even if the respective dye is otherwise is identical.
- substituents may have the meanings vinyl sulfone and ß-sulfatoethylsulfone.
- the proportion of the particular dye with the vinylsulfonyl group is up to about 30 mol%, based on the respective total amount of dye.
- the dye mixtures according to the invention may also contain dyes of the general formulas (II) and (III) or (II) and (IV) or (III) and (IV).
- they may also contain dyes of the general formulas (M), (III) and (IV).
- Preferred dye mixtures according to the invention contain a dye of the general formula (Ia)
- V 1a 1 for vinylsulfone, sulfatoethylsulfone or chloroethylsulfone;
- V 1a "4 represents hydrogen, -SO 3 M, vinylsulfone, sulfatoethylsulfone or chloroethylsulfone;
- M is hydrogen, sodium or potassium, and at least one further dye of the general formulas (II), (III) or (IV).
- compositions according to the invention are those which contain at least one dye of the general formula (I) and at least one further dye of the general formula (IIb)
- R 2b 1 to R 2b "4 are each independently hydrogen, -SO 3 M, methyl or
- R 2b "5 and R 2b" 6 are independently hydrogen, methyl or ethyl
- V 2b 1 and V 2b "2 independently of one another represent hydrogen, -SO 3 M, vinylsulfone,
- V 2b "3 and V 2b" 5 are independently vinylsulfone, sulfatoethylsulfone or
- V 2b "4 and V 2b" 6 are independently hydrogen or -SO 3 M; and M is hydrogen, sodium or potassium.
- Preferred dyestuff mixtures according to the invention are furthermore those which comprise at least one dyestuff of the general formula (I) and at least one further dyestuff of the general formulas (MIb)
- W 1 and W 3b 3b "2 are independently hydrogen, (C r C4) alkyl, (C 1 -C 4) - alkyl substituted by -OSO 3 M, or one of the following groups
- ⁇ / 3b-4 D v3b-2 are independently a group of formulas
- M are hydrogen, sodium or potassium.
- preferred dye mixtures according to the invention are those which contain at least one dye of the general formula (I) and at least one further dye of the general formula (IVb) wherein
- Y 4b "2 has the same meaning as Y 413" 1 or one of the following groups
- V 413 "1 to V 413" 3 and V 413 "9 to V 413" 10 are each independently hydrogen or
- V 413 "5 bisV 413" 7 independently of one another hydrogen, SO 3 M, vinylsulfone,
- V 413 "8 vinylsulfone, sulfatoethylsulfone or chloroethylsulfone;
- R 4b "4 represents hydrogen, methyl or ethyl
- M is hydrogen, sodium or potassium; are included.
- Particularly preferred dye mixtures according to the invention are those which contain at least one dye of the general formula (Ia) and at least one further dye of either the general formula (IIb) or the general formula (MIb) or the general formula (IVb).
- dye mixtures according to the invention which contain a dye of the general formulas (Ic)
- V 1c 1 is vinylsulfone or sulfatoethylsulfone; V 1 c 4 f Ur -SO 3 M and
- M is hydrogen or sodium and at least one further dye of the general formulas (II), (IM) or (IV) included.
- Particularly preferred dye mixtures according to the invention are also those which contain at least one dye of the general formula (I) and at least one further dye of the general formula (Mc)
- R 2c 1 to R 2c 3 independently of one another represent hydrogen or methyl
- R 2c 4 is hydrogen or SO 3 M
- V 2c 1 and V 2c ⁇ 3 are independently hydrogen or SO 3 M;
- V 2c 2 is hydrogen, vinylsulfone or sulfatoethylsulfone
- V 2c-4 for vinylsulfone or sulfatoethylsulfone
- Particularly preferred dye mixtures according to the invention are furthermore those which contain at least one dye of the general formula (I) and at least one further dye of the general formula (IIIc)
- W # 3c-1 and independently represent (CH 2 ) 2 -OSO 3 M or one of the following groups
- V3 -4.
- V 3c 1 to V 3c "4 independently of one another represent hydrogen, SO 3 M, vinylsulfone or sulfatoethylsulfone;
- dye mixtures according to the invention are those which contain at least one dye of the general formula (I) and at least one further dye of the general formula (IVc)
- Y 4c "2 has the same meaning as Y 40" 1 or for one of the following groups
- V 40 "8 represents vinylsulfone or sulfatoethylsulfone
- y 4c - 4 y 4c9 and v 40 - 10 independently of one another denote hydrogen or -SO 3 M;
- M is hydrogen or sodium, are present.
- Very particularly preferred dye mixtures according to the invention are those which contain at least one dye of the general formula (Ic) and at least one further dye of either the general formula (Mc) or the general formula (MIc) or the general formula (IVc).
- the dye mixtures according to the invention contain the dyes of the general formulas (I) and (M) and / or (III) and / or (IV) in weight ratios which can vary within wide limits.
- dyes of the general formula (I) are present in amounts of from 90 to 10% by weight and the dyes of the general formulas (M) and / or (III) and / or (IV) in amounts of from 10 to 90% by weight. %.
- Dyes of the general formula (I) are preferably present in amounts of from 80 to 20% by weight and the dyes of the general formulas (M) and / or (III) and / or (IV) in amounts of from 20 to 80% by weight. %.
- dyes of the general formula (I) in amounts of from 65 to 35% by weight and the dyes of the general formulas (M) and / or (III) and / or (IV) in amounts of from 35 to 65% by weight. -%.
- dye mixtures according to the invention which contain dyes of the general formulas (Ia), (Mb), (Mc), (MIb), (MIc), (IVb) or (IVc).
- the dyestuff mixtures according to the invention can be prepared by processes known per se and customary, for example by mechanical mixing of the individual dyestuffs of the general formulas (I) and (M), (III) or (IV), whether in the form of dyestuff powders or granules or of synthetic solutions of the dyes of the general formulas (I) and (M), (III) or (IV) or of aqueous solutions of the dyes of the general formulas (I) and (M), (III) or (IV) in general.
- the dye solutions mentioned may still contain conventional auxiliaries.
- the dyes of the general formulas (I), (M), (III) and (IV) are known as individual substances and their preparation is described in the literature and known to the person skilled in the art. They are available in the market as individual substances or can be prepared by known methods.
- the dye mixtures according to the invention can be present as a preparation in solid or in liquid (dissolved) form.
- solid form they contain, as far as necessary, the customary in water-soluble and especially fiber-reactive dyes electrolyte salts, such as
- Sodium chloride, potassium chloride and sodium sulfate may further include those in Commercial dyes contain conventional auxiliaries, such as buffer substances, which are able to adjust a pH in aqueous solution between 3 and 7, such as sodium acetate, sodium citrate, sodium borate, sodium bicarbonate, sodium dihydrogen phosphate and disodium hydrogen phosphate, also dyeing aids, dedusting agents and small amounts of siccatives. If present in liquid aqueous solution (including the level of thickening agents common in printing pastes), they may also contain substances which will ensure the durability of these preparations, such as mildewproofing agents.
- the dye mixtures according to the invention may contain further fiber-reactive dyes in any desired amounts.
- they may contain dyes which serve to shade the dye mixture in an amount of up to 5% by weight.
- These further dyes may be added by conventional mixing or may also be prepared chemically in the same reaction mixture together with the synthesis of a dye mixture according to the invention and introduced into the dye mixture if one or more precursors of the further dye with one or more precursors of the dyes of the general formulas (US Pat. I) and / or (II) and / or (IM) and / or (IV) are identical.
- the present invention also relates to the use of the dye mixtures according to the invention for dyeing or printing hydroxyl- and / or carboxamido-containing materials or to a process for dyeing or printing a hydroxy- and / or carboxamido-containing material, which comprises applying a dye mixture to the material and fixing of the
- Dye mixture comprises by means of heat and / or by means of an alkaline agent on the material and which is characterized in that a dye mixture according to the invention is used. This navy blue to black dyeings or prints are obtained.
- Hydroxy-containing materials can be of natural or synthetic origin. Examples are cellulose fiber materials, such as preferably cotton, linen, hemp, jute and ramie fibers, regenerated products, such as preferably rayon and viscose rayon, chemically modified Cellulosic fibers such as aminated cellulose fibers, as well as polyvinyl alcohols.
- cellulose fiber materials such as preferably cotton, linen, hemp, jute and ramie fibers
- regenerated products such as preferably rayon and viscose rayon
- chemically modified Cellulosic fibers such as aminated cellulose fibers, as well as polyvinyl alcohols.
- carbonamido-containing materials are synthetic and natural polyamides and polyurethanes, for example wool and other animal hair, silk, leather, polyamide-6,6, polyamide-6, polyamide-11 and polyamide-4.
- the said hydroxyl- and / or carboxamido-containing materials may be in various forms.
- fabrics such as paper and leather
- films such as polyamide films
- a mass for example of polyamide and polyurethane
- fibers such as cellulose fibers.
- the fibers are preferably textile fibers, for example in the form of fabrics or yarns or in the form of strands or wound bodies.
- the dye mixtures according to the invention can be applied and fixed on the abovementioned materials, in particular on the fiber materials mentioned, according to the application techniques known for water-soluble, in particular, those known for fiber-reactive dyes.
- cellulose fibers by exhaustion both from short and long liquor, for example in the ratio of product to liquor from 1: 5 to 1: 100, preferably 1: 6 to 1: 30, using a variety of acid-binding agents and if necessary, neutral salts, such as sodium chloride or sodium sulfate, dyeings with very good color yields.
- acid-binding agents such as sodium chloride or sodium sulfate
- aqueous bath at temperatures between 40 and 105 0 C, optionally at a temperature up to 130 0 C under pressure, but preferably at 30 to 95 ° C, in particular 45 to 65 ° C, and optionally in the presence of conventional dyeing auxiliaries , It is possible to proceed in such a way that the material is introduced into the warm bath and this is gradually heated to the desired dyeing temperature and the dyeing process is completed at this temperature. If necessary, the neutral salts which accelerate the removal of the dyes can also be added to the bath only after the actual dyeing temperature has been reached.
- the dyestuff mixtures according to the invention are obtained by conventional printing processes for cellulosic fibers, the single phase, for example by printing with a sodium bicarbonate or other acid-binding agent containing printing paste and subsequent steaming at 100 to 103 0 C, or two-phase, for example by printing with neutral or weakly acidic Printing ink and subsequent fixing either by passing through a hot electrolyte-containing alkaline bath or by over-padding with an alkaline electrolyte-containing padding liquor and then lingering or steaming or treatment with dry heat of the alkaline padded material, can be carried out even strong prints with good contour and a clear white ground. The failure of the prints is only slightly dependent on changing fixing conditions.
- the acid-binding and fixing the dyes of the dye mixtures of the invention on the cellulose fibers effecting agents include, for example, water-soluble basic salts of alkali metals and also alkaline earth metals of inorganic or organic acids or compounds which release alkali in the heat, further alkali metal silicates.
- alkali metal hydroxides and alkali metal salts of weak to medium inorganic or organic acids may be mentioned, of the alkali metal compounds preferably the sodium and potassium compounds are meant.
- Such acid-binding agents are for example, sodium hydroxide, potassium hydroxide, sodium carbonate, sodium bicarbonate, potassium carbonate, sodium formate, sodium dihydrogenphosphate, disodium hydrogenphosphate, sodium trichloroacetate, trisodium phosphate or waterglass, or mixtures thereof, such as mixtures of caustic soda and waterglass.
- the dye mixtures according to the invention are distinguished on the cellulose fiber materials when used in the dyeing and printing process by an excellent color strength, which can be achieved partially in the presence of no or only very small alkali or alkaline earth metal compounds.
- no electrolyte salt is needed for a small depth of color, for an average color depth no more than 5 g / l of electrolyte salt and for large color depths no more than 10 g / l of electrolyte salt.
- a low color depth refers to the use of 2% by weight of dye based on the substrate to be dyed, an average color depth denotes the use of 2 to 4% by weight of dye based on the substrate to be dyed and a large color depth denotes the use of 4 to 10 wt .-% of the dye based on the substrate to be dyed.
- the dyeings and prints obtainable with the dye mixtures according to the invention have clear nuances and have good fastness to lightfastness on cellulosic fiber materials and, in particular, good wet fastness properties, such as washing, flexing, water, seawater, overdyeing and acidic as well as alkaline perspiration fastnesses. In addition, they have good plushing fastness, ironing and rubbing fastness. In particular, however, the very good chlorine fastness is to be emphasized.
- the dye mixtures according to the invention can also be used for the fiber-reactive dyeing of wool.
- This also includes felt-free or low-felt wool (see, for example, H. Rath, textbook of textile chemistry, Springer-Verlag, 3rd edition (1972), pp. 295-299, in particular the equipment according to the so-called Hercosett method (p J. Soc. Dyers and Colorists 1972, 93-99, and 1975, 33-44).
- Dyeing on wool is carried out in an usual and known manner from an acidic environment. So you can, for example, the dyebath acetic acid and / or ammonium sulfate or acetic acid and ammonium acetate or sodium acetate to obtain the desired pH.
- leveling agents such as based on a reaction product of cyanuric chloride with three times the molar amount of an aminobenzenesulfonic acid and / or a Aminonaphthalinsulfonklare or based on a reaction product of, for example, stearylamine with ethylene oxide.
- the dye mixture according to the invention is preferably first subjected to the exhaustion process from acid dyebath having a pH of about 3.5 to 5.5 under the control of the pH and the pH then, towards the end of the dyeing time, in the neutral and possibly weak shifted alkaline range to a pH of 8.5, especially to achieve high color depths, the full reactive binding between the dyes of the dye mixtures of the invention and the fiber bring about. At the same time, the non-reactively bound dye fraction is removed.
- the procedure described here also applies to the production of dyeings on fiber materials of other natural or synthetic polyamides and their mixtures with polyurethanes.
- the material to be dyed is introduced at a temperature of about 40 0 C in the bath, there for some time moved therein, the dyebath then adjusted to the desired weakly acid, preferably weakly acetic acid, pH and the actual color in a Temperature between 60 and 98 ° C performed.
- the dyeings can also be carried out at boiling temperature or in closed dyeing apparatus at temperatures up to 106 0 C. Since the water solubility of the dye mixtures according to the invention is very good, they can also be used with advantage in conventional continuous dyeing processes.
- the dye mixtures according to the invention can also be used in digital printing processes, in particular in digital textile printing. For this it is necessary to formulate the dye mixtures according to the invention in inks.
- Aqueous inks for digital printing which are characterized by a content of a dye mixture according to the invention, are also provided by the present invention.
- the inks according to the invention contain the dye mixture according to the invention preferably in amounts of from 0.1% by weight to 50% by weight, more preferably in amounts of from 1% by weight to 30% by weight and most preferably in amounts of 1% by weight % to 15% by weight based on the total weight of the ink.
- the inks may contain, in addition to the dye mixture according to the invention, further reactive dyes which are used in digital printing.
- a conductivity of 0.5 to 25 mS / m can be set by addition of electrolyte.
- electrolyte for example, lithium nitrate and potassium nitrate are suitable.
- the inks of the invention may contain organic solvents having a total content of 1-50%, preferably 5-30% by weight. Suitable organic solvents are, for example, alcohols, such as. For example, methanol, ethanol, 1-propanol, isopropanol, 1-butanol, tert. Butanol and pentyl alcohol; polyhydric alcohols, such as. B.
- polyethylene glycol and polypropylene glycol For example, polyethylene glycol and polypropylene glycol; Alkylene glycols having 1 to 8 alkylene groups, such as. For example, monoethylene glycol, diethylene glycol, triethylene glycol, tetraethylene glycol, thioglycol, thiodiglycol, butyl triglycol, hexylene glycol, propylene glycol, dipropylene glycol and tripropylene glycol; lower alkyl ethers of polyhydric alcohols, such as. For example, ethylene glycol monomethyl ether, ethylene glycol monoethyl ether, ethylene glycol monobutyl ether,
- Diethylene glycol monomethyl ether diethylene glycol monoethyl ether, diethylene glycol monobutyl ether, diethylene glycol monohexyl ether, triethylene glycol monomethyl ether, triethylene glycol monobutyl ether, tripropylene glycol monomethyl ether, tetraethylene glycol monomethyl ether, tetraethylene glycol monobutyl ether, tetraethylene glycol dimethyl ether, propylene glycol monomethyl ether, propylene glycol monoethyl ether, propylene glycol monobutyl ether and tripropylene glycol isopropyl ether; Polyalkylene glycol ethers, such as. For example, polyethylene glycol monomethyl ether, Polypropylene glycol glycerol ether, polyethylene glycol tridecyl ether, polyethylene glycol nonylphenyl ether;
- Amines such as Methylamine, ethylamine, thethylamine, diethylamine, dimethylamine, trimethylamine, dibutylamine, diethanolamine, triethanolamine, N-acetylethanolamine, N-formylethanolamine, ethylenediamine;
- Urea derivatives such as. B. urea,
- N-methylurea N, N'-epsilon-dimethylurea, ethyleneurea, 1, 1, 3,3-tetramethylurea
- Amides such as Dimethylformamide, dimethylacetamide and acetamide
- Ketones or keto alcohols such as. Acetone and diacetone alcohol
- cyclic ethers such as.
- the inks of the invention may contain the customary additives, such as viscosity moderators to adjust viscosities in the range of 1, 5 to 40.0 mPa.s in a temperature range from 20 to 50 0 C.
- Preferred inks have a viscosity of 1.5 to 20 mPas and particularly preferred inks have a viscosity of 1.5 to 15 mPas.
- Suitable viscosity moderators are theological additives, for example: polyvinylcaprolactam, polyvinylpyrrolidone and their co-polymers polyetherpolyol, associative thickener, polyurea, polyurethane, sodium alginates, modified galactomannans, polyetherurea, polyurethane, nonionic cellulose ethers.
- the inks according to the invention may contain surface-active substances for the adjustment of surface tensions of from 20 to 65 mN / m, which are disclosed in US Pat Depending on the method used (thermal or piezo technology) may be adjusted if necessary.
- Suitable surface-active substances are, for example, surfactants of all kinds, preferably nonionic surfactants, butyldiglycol and 1,2-hexanediol.
- the inks of the present invention may contain conventional additives such as fungicidal and bacterial growth inhibiting agents in amounts of from 0.01 to 1% by weight based on the total weight of the ink.
- the inks can be prepared in a conventional manner by mixing the components in water.
- the inks according to the invention are particularly suitable for use in inkjet printing processes for printing a wide variety of preprepared materials, such as silk, leather, wool, polyamide fibers and polyurethanes, and in particular cellulose-containing fiber materials of all kinds.
- preprepared materials such as silk, leather, wool, polyamide fibers and polyurethanes, and in particular cellulose-containing fiber materials of all kinds.
- blended fabrics can be printed, for example mixtures of cotton, silk or wool with polyester fibers or polyamide fibers.
- the aids In contrast to conventional textile printing, in which the printing ink already contains all the fixing chemicals and thickeners for a reactive dye, in the case of digital or ink-jet printing, the aids must be applied to the textile substrate in a separate pretreatment step.
- the pretreatment of the textile substrate such as cellulose and
- the textile fiber material is dried at 120 to 150 0 C and then fixed.
- the fixation of the ink jet prints prepared with reactive dyes can be carried out at room temperature, or with saturated steam, with superheated steam, with hot air, with microwaves, with infrared radiation, with laser or electron beams or with other suitable types of energy transfer.
- the post-treatment is carried out, which is the prerequisite for good fastness, high brilliance and a perfect white foundation.
- the prints made with the inks of the present invention have high color strength and high fiber-dye bonding stability in both acidic and alkaline regions, furthermore good fastness to light and very good wet fastness properties such as washing, water, seawater -, over-dyeing and perspiration fastness, as well as a good pleating fastness, ironing fastness and rubbing fastness.
- the following examples serve to illustrate the invention.
- the parts are by weight, the percentages are by weight unless otherwise stated. Parts by weight refer to parts by volume such as kilograms to liters.
- the compounds described by formula in the examples are written in the form of the sodium salts, since they are generally prepared and isolated in the form of their salts, preferably sodium or potassium salts, and used in the form of their salts for dyeing.
- the starting compounds mentioned in the following examples can be used in the form of the free acid or else in the form of their salts, preferably alkali metal salts, such as sodium or potassium salts, ie M is defined as indicated above.
- a dye mixture having a molar mixing ratio of the dye (I-2) to dye (111-1) of 60:40 having.
- the mixture contains electrolyte salts, such as sodium chloride and sodium sulfate, which originate from the respective dye synthesis and shows very good dyeing properties.
- electrolyte salts such as sodium chloride and sodium sulfate
- fiber-reactive shading component give deep and level navy blue dyeings having good fastness to light in a dyeing process common to fiber-reactive dyes.
- Example 2 500 parts of an aqueous solution with 70 parts of the below-mentioned dye of the formula (1-3)
- a dye mixture having a molar mixing ratio of the dye (1-3) to dye (11-3) of 52: 48.
- the mixture contains electrolyte salts, such as sodium chloride and sodium sulfate, which originate from the respective dye synthesis and shows very good dyeing properties. For example, it provides strong and level gray-blue dyeings on cellulosic fiber materials such as cotton or regenerated cellulose fibers in a pultrusion dyeing process which is customary for fiber-reactive dyes. Examples 3 to 17
- the following examples relate to further dye mixtures according to the invention which have very good performance properties and to the materials mentioned in the description, in particular cellulose fiber materials, according to the usual in the art application methods in dyeing and printing, preferably according to the usual in the art application and Fixing methods for fiber-reactive dyes, strong blue-gray dyeings and prints with good fastness properties and good color build.
- these blends in combination with a red-dyeing fiber-reactive component on natural and synthetic polyamide materials, such as wool or nylon 6-6, provide deep and level navy blue dyeings having good fastness to light in a dyeing process common to fiber-reactive dyes.
- a textile fabric consisting of mercerized cotton is padded with a liquor containing 35 g / l sodium carbonate, 50 g / l urea and 150 g / l of a low-viscosity sodium alginate solution (6%) and then dried.
- the fleet intake is 70%.
- the textile pretreated in this way is treated with an aqueous ink containing 8% of a dye mixture according to Example 1, 20% of 1, 2-propanediol, 0.01% of Mergal K9N and 71, 99% of water, with a drop-on-demand (bubble Jet) Ink-jet print head a pattern printed.
- the print is completely dried.
- the fixation takes place by means of saturated steam at 102 0 C for 8 minutes.
- the pressure is then rinsed warm, subjected to a fastness wash with hot water at 95 ° C., rinsed warm and then dried. This gives a gray-blue print with excellent use fastness.
- Example 19 A fabric consisting of mercerized cotton is padded with a liquor containing 35 g / l of sodium carbonate, 100 g / l of urea and 150 g / l of a low-viscosity sodium alginate solution (6%) and then dried. The fleet intake is 70%.
- the textile pretreated in this way is treated with an aqueous ink containing 8% of a dye mixture according to Example 2, 15% N-methylpyrrolidone, 0.01% Mergal K9N and 76.99% water, with a drop-on-demand (bubble jet). Ink-jet printhead a pattern printed. The print is completely dried. The fixation takes place by means of saturated steam at 102 0 C for 8 minutes. The pressure is then rinsed warm, subjected to a fastness wash with hot water at 95 ° C., rinsed warm and then dried. This gives a gray-blue print with excellent use fastness.
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/162,146 US20090041938A1 (en) | 2006-01-26 | 2007-01-19 | Mixtures of fibre reactive dyes production and use thereof |
| EP07704030A EP1984456A2 (de) | 2006-01-26 | 2007-01-19 | Mischungen von faserreaktiven farbstoffen, ihre herstellung und verwendung |
| JP2008551766A JP2009525355A (ja) | 2006-01-26 | 2007-01-19 | 繊維反応性染料の混合物、それらの調製及び使用 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102006003621.2 | 2006-01-26 | ||
| DE102006003621A DE102006003621A1 (de) | 2006-01-26 | 2006-01-26 | Mischungen von faserreaktiven Farbstoffen, ihre Herstellung und Verwendung |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2007085572A2 true WO2007085572A2 (de) | 2007-08-02 |
| WO2007085572A3 WO2007085572A3 (de) | 2008-01-03 |
Family
ID=38181163
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2007/050560 WO2007085572A2 (de) | 2006-01-26 | 2007-01-19 | Mischungen von faserreaktiven farbstoffen, ihre herstellung und verwendung |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US20090041938A1 (de) |
| EP (1) | EP1984456A2 (de) |
| JP (1) | JP2009525355A (de) |
| KR (1) | KR20080090474A (de) |
| CN (1) | CN101374913A (de) |
| DE (1) | DE102006003621A1 (de) |
| IN (1) | IN2008KO02064A (de) |
| TW (1) | TW200728410A (de) |
| WO (1) | WO2007085572A2 (de) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101177547B (zh) * | 2007-11-30 | 2010-06-09 | 上虞新晟化工工业有限公司 | 一种酸性灰染料组合物 |
| WO2010086243A3 (en) * | 2009-01-27 | 2010-10-21 | Dystar Colours Deutschland Gmbh | Fiber-reactive copper complex disazo dyes |
| CN102485802A (zh) * | 2010-12-03 | 2012-06-06 | 上海雅运纺织化工股份有限公司 | 藏青色活性染料组合物及其在纤维上的染色应用 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102321390B (zh) * | 2011-09-07 | 2013-11-06 | 上海雅运纺织化工股份有限公司 | 三原色活性染料组合物及其在纤维上的染色应用 |
| CN108723942B (zh) * | 2018-07-25 | 2023-06-30 | 泰州神舟传动科技有限公司 | 一种后桥生产用防偏距的高精度双主轴机床 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0668328A2 (de) * | 1994-02-19 | 1995-08-23 | Hoechst Aktiengesellschaft | Reaktivfarbstoffmischungen |
| WO2002010289A1 (de) * | 2000-07-29 | 2002-02-07 | Dystar Textilfarben Gmbh & Co. Deutschland Kg | Farbstoffmischung von wasserlöslichen faserreaktiven farbstoffen, verfahren zu ihrer herstellung und ihre verwendung |
| EP1234858A2 (de) * | 2001-02-27 | 2002-08-28 | Everlight USA, Inc. | Reaktivfarbstoffmischungen und deren Verwendung |
| EP1275700A2 (de) * | 2001-07-12 | 2003-01-15 | Ciba SC Holding AG | Verfahren zum Trichromie-Färben oder -Bedrucken von synthetischen Polyamidfasermaterialien |
| DE102004028919A1 (de) * | 2003-06-24 | 2005-01-27 | Everlight Usa, Inc. | Farbstoffzusammensetzungen und deren Verwendung |
| US20050087099A1 (en) * | 2003-10-28 | 2005-04-28 | Eastman Kodak Company | Ink jet ink set |
-
2006
- 2006-01-26 DE DE102006003621A patent/DE102006003621A1/de not_active Withdrawn
-
2007
- 2007-01-19 US US12/162,146 patent/US20090041938A1/en not_active Abandoned
- 2007-01-19 KR KR1020087018450A patent/KR20080090474A/ko not_active Application Discontinuation
- 2007-01-19 CN CNA2007800035047A patent/CN101374913A/zh active Pending
- 2007-01-19 WO PCT/EP2007/050560 patent/WO2007085572A2/de active Application Filing
- 2007-01-19 JP JP2008551766A patent/JP2009525355A/ja not_active Withdrawn
- 2007-01-19 EP EP07704030A patent/EP1984456A2/de not_active Withdrawn
- 2007-01-24 TW TW96102682A patent/TW200728410A/zh unknown
-
2008
- 2008-05-22 IN IN2064KO2008 patent/IN2008KO02064A/en unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0668328A2 (de) * | 1994-02-19 | 1995-08-23 | Hoechst Aktiengesellschaft | Reaktivfarbstoffmischungen |
| WO2002010289A1 (de) * | 2000-07-29 | 2002-02-07 | Dystar Textilfarben Gmbh & Co. Deutschland Kg | Farbstoffmischung von wasserlöslichen faserreaktiven farbstoffen, verfahren zu ihrer herstellung und ihre verwendung |
| EP1234858A2 (de) * | 2001-02-27 | 2002-08-28 | Everlight USA, Inc. | Reaktivfarbstoffmischungen und deren Verwendung |
| EP1275700A2 (de) * | 2001-07-12 | 2003-01-15 | Ciba SC Holding AG | Verfahren zum Trichromie-Färben oder -Bedrucken von synthetischen Polyamidfasermaterialien |
| DE102004028919A1 (de) * | 2003-06-24 | 2005-01-27 | Everlight Usa, Inc. | Farbstoffzusammensetzungen und deren Verwendung |
| US20050087099A1 (en) * | 2003-10-28 | 2005-04-28 | Eastman Kodak Company | Ink jet ink set |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101177547B (zh) * | 2007-11-30 | 2010-06-09 | 上虞新晟化工工业有限公司 | 一种酸性灰染料组合物 |
| WO2010086243A3 (en) * | 2009-01-27 | 2010-10-21 | Dystar Colours Deutschland Gmbh | Fiber-reactive copper complex disazo dyes |
| JP2012515823A (ja) * | 2009-01-27 | 2012-07-12 | ダイスター・カラーズ・ドイッチュラント・ゲゼルシャフト・ミト・ベシュレンクテル・ハフツング | 繊維反応性銅錯体ジスアゾ染料 |
| US9062208B2 (en) | 2009-01-27 | 2015-06-23 | Dystar Colours Distribution Gmbh | Fiber-reactive copper complex disazo dyes |
| CN102485802A (zh) * | 2010-12-03 | 2012-06-06 | 上海雅运纺织化工股份有限公司 | 藏青色活性染料组合物及其在纤维上的染色应用 |
| CN102485802B (zh) * | 2010-12-03 | 2014-01-01 | 上海雅运纺织化工股份有限公司 | 藏青色活性染料组合物及其在纤维上的染色应用 |
Also Published As
| Publication number | Publication date |
|---|---|
| TW200728410A (en) | 2007-08-01 |
| EP1984456A2 (de) | 2008-10-29 |
| US20090041938A1 (en) | 2009-02-12 |
| WO2007085572A3 (de) | 2008-01-03 |
| IN2008KO02064A (de) | 2009-01-16 |
| CN101374913A (zh) | 2009-02-25 |
| KR20080090474A (ko) | 2008-10-08 |
| DE102006003621A1 (de) | 2007-08-09 |
| JP2009525355A (ja) | 2009-07-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2035507B1 (de) | Farbstoffmischungen von faserreaktiven azofarbstoffen, ihre herstellung und ihre verwendung | |
| DE102008054404A1 (de) | Faserreaktive Azofarbstoffe und Farbstoffmischungen, Verfahren zu ihrer Herstellung und ihre Verwendung | |
| EP1508596B1 (de) | Farbstoffmischungen von faserreaktiven Azofarbstoffen, ihre Herstellung und ihre Verwendung | |
| DE102005047391A1 (de) | Farbstoffe und Farbstoffmischungen von faserreaktiven Azofarbstoffen, ihre Herstellung und ihre Verwendung | |
| EP2118214A2 (de) | Mischungen von faserreaktiven azofarbstoffen | |
| EP1737915A1 (de) | Mischungen von faserreaktiven azofarbstoffen, ihre herstellung und verwendung | |
| WO2006024639A1 (de) | Farbstoffmischungen von faserreaktiven azofarbstoffen, ihre herstellung und ihre verwendung | |
| EP1666540B1 (de) | Mischungen von faserreaktiven Azofarbstoffen, Verfahren zu deren Herstellung und ihre Verwendung | |
| EP1508598B1 (de) | Farbstoffmischungen von faserreaktiven Azofarbstoffen, ihre Herstellung und ihre Verwendung | |
| EP1984456A2 (de) | Mischungen von faserreaktiven farbstoffen, ihre herstellung und verwendung | |
| DE102009000417A1 (de) | Faserreaktive Azofarbstoffe, Verfahren zu ihrer Herstellung und ihre Verwendung | |
| EP1555300A1 (de) | Reaktive Azofarbstoffe, Verfahren zu ihrer Herstellung und ihre Verwendung | |
| TWI550027B (zh) | 纖維活性偶氮染料之混合物 | |
| DE102006003864A1 (de) | Wasserlösliche faserreaktive Farbstoffe, Verfahren zu ihrer Herstellung und ihre Verwendung | |
| EP1891161B1 (de) | Reaktivfarbstoffe, verfahren zu deren herstellung und ihre verwendung | |
| WO2006072548A2 (de) | Reaktive triphendioxazin-farbstoffe, verfahren zu ihrer herstellung und ihre verwendung | |
| WO2006136548A2 (de) | Farbstoffmischung von wasserlöslichen faserreaktiven farbstoffen, verfahren zu ihrer herstellung und ihre verwendung | |
| DE102009000423A1 (de) | Faserreaktive Kupferkomplex-Disazofarbstoffe | |
| EP2035508A2 (de) | Farbstoffmischungen und ihre verwendung zum bedrucken von fasermaterialien | |
| EP1877495A2 (de) | Reaktive azofarbstoffe, verfahren zu ihrer herstellung und ihre verwendung | |
| CN108473787B (zh) | 不含重金属的蓝色和海军蓝纤维活性染料混合物 | |
| WO2006103186A2 (de) | Reaktivfarbstoffe, verfahren zu deren herstellung und ihre verwendung |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2064/KOLNP/2008 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007704030 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008071219 Country of ref document: EG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008551766 Country of ref document: JP Ref document number: 200780003504.7 Country of ref document: CN Ref document number: 1020087018450 Country of ref document: KR |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12162146 Country of ref document: US |