WO2005061443A2 - Benzoylsubstituierte phenylalanin-amide - Google Patents
Benzoylsubstituierte phenylalanin-amide Download PDFInfo
- Publication number
- WO2005061443A2 WO2005061443A2 PCT/EP2004/014392 EP2004014392W WO2005061443A2 WO 2005061443 A2 WO2005061443 A2 WO 2005061443A2 EP 2004014392 W EP2004014392 W EP 2004014392W WO 2005061443 A2 WO2005061443 A2 WO 2005061443A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- formula
- aminocarbonyl
- alkoxy
- phenyl
- Prior art date
Links
- -1 Benzoyl-substituted phenylalanine amides Chemical class 0.000 title claims abstract description 1018
- 238000000034 method Methods 0.000 claims abstract description 26
- 150000003839 salts Chemical class 0.000 claims abstract description 16
- 239000001257 hydrogen Substances 0.000 claims description 109
- 229910052739 hydrogen Inorganic materials 0.000 claims description 109
- 125000000217 alkyl group Chemical group 0.000 claims description 83
- 150000002431 hydrogen Chemical group 0.000 claims description 77
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 claims description 60
- 229910052736 halogen Inorganic materials 0.000 claims description 56
- 150000002367 halogens Chemical class 0.000 claims description 56
- SNOOUWRIMMFWNE-UHFFFAOYSA-M sodium;6-[(3,4,5-trimethoxybenzoyl)amino]hexanoate Chemical compound [Na+].COC1=CC(C(=O)NCCCCCC([O-])=O)=CC(OC)=C1OC SNOOUWRIMMFWNE-UHFFFAOYSA-M 0.000 claims description 55
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 50
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 claims description 48
- 239000000203 mixture Substances 0.000 claims description 44
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 42
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 32
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 31
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 claims description 27
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 claims description 27
- ORTFAQDWJHRMNX-UHFFFAOYSA-N hydroxidooxidocarbon(.) Chemical group O[C]=O ORTFAQDWJHRMNX-UHFFFAOYSA-N 0.000 claims description 24
- 125000003545 alkoxy group Chemical group 0.000 claims description 21
- 125000004453 alkoxycarbonyl group Chemical group 0.000 claims description 21
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 20
- 235000010233 benzoic acid Nutrition 0.000 claims description 17
- 125000001424 substituent group Chemical group 0.000 claims description 16
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims description 15
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 15
- 125000000623 heterocyclic group Chemical group 0.000 claims description 14
- 239000005711 Benzoic acid Substances 0.000 claims description 13
- 235000008729 phenylalanine Nutrition 0.000 claims description 13
- 238000002360 preparation method Methods 0.000 claims description 13
- 230000008569 process Effects 0.000 claims description 12
- 230000009467 reduction Effects 0.000 claims description 12
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 claims description 11
- 150000001412 amines Chemical class 0.000 claims description 11
- 125000001188 haloalkyl group Chemical group 0.000 claims description 11
- 150000002994 phenylalanines Chemical class 0.000 claims description 11
- CIUQDSCDWFSTQR-UHFFFAOYSA-N [C]1=CC=CC=C1 Chemical compound [C]1=CC=CC=C1 CIUQDSCDWFSTQR-UHFFFAOYSA-N 0.000 claims description 7
- 239000003795 chemical substances by application Substances 0.000 claims description 7
- 239000004009 herbicide Substances 0.000 claims description 7
- 125000006517 heterocyclyl carbonyl group Chemical group 0.000 claims description 7
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 claims description 6
- 125000004457 alkyl amino carbonyl group Chemical group 0.000 claims description 6
- 125000004414 alkyl thio group Chemical group 0.000 claims description 6
- 125000000304 alkynyl group Chemical group 0.000 claims description 6
- 125000006297 carbonyl amino group Chemical group [H]N([*:2])C([*:1])=O 0.000 claims description 6
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 6
- 238000009472 formulation Methods 0.000 claims description 6
- 125000000468 ketone group Chemical group 0.000 claims description 6
- 125000000171 (C1-C6) haloalkyl group Chemical group 0.000 claims description 5
- 125000003342 alkenyl group Chemical group 0.000 claims description 5
- 125000004765 (C1-C4) haloalkyl group Chemical group 0.000 claims description 4
- 125000004390 alkyl sulfonyl group Chemical group 0.000 claims description 4
- 150000001559 benzoic acids Chemical class 0.000 claims description 4
- 125000004438 haloalkoxy group Chemical group 0.000 claims description 4
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 claims description 3
- 230000010933 acylation Effects 0.000 claims description 3
- 238000005917 acylation reaction Methods 0.000 claims description 3
- 150000001558 benzoic acid derivatives Chemical class 0.000 claims description 3
- 125000000051 benzyloxy group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])O* 0.000 claims description 3
- 125000004966 cyanoalkyl group Chemical group 0.000 claims description 3
- 125000001181 organosilyl group Chemical group [SiH3]* 0.000 claims description 3
- 125000006678 phenoxycarbonyl group Chemical group 0.000 claims description 3
- 230000008635 plant growth Effects 0.000 claims description 3
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 claims description 2
- 125000000262 haloalkenyl group Chemical group 0.000 claims description 2
- 125000004441 haloalkylsulfonyl group Chemical group 0.000 claims description 2
- 125000004995 haloalkylthio group Chemical group 0.000 claims description 2
- 239000000575 pesticide Substances 0.000 claims description 2
- 239000011814 protection agent Substances 0.000 claims description 2
- OBSIQMZKFXFYLV-QMMMGPOBSA-N L-phenylalanine amide group Chemical class N[C@@H](CC1=CC=CC=C1)C(=O)N OBSIQMZKFXFYLV-QMMMGPOBSA-N 0.000 claims 4
- 125000004737 (C1-C6) haloalkoxy group Chemical group 0.000 claims 2
- 125000004767 (C1-C4) haloalkoxy group Chemical group 0.000 claims 1
- 125000006700 (C1-C6) alkylthio group Chemical group 0.000 claims 1
- 150000001875 compounds Chemical class 0.000 abstract description 152
- 239000013067 intermediate product Substances 0.000 abstract description 3
- 239000000126 substance Substances 0.000 abstract 1
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 63
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 50
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 48
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 45
- 229910052731 fluorine Inorganic materials 0.000 description 41
- 239000011737 fluorine Substances 0.000 description 41
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 40
- 239000000460 chlorine Substances 0.000 description 40
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 39
- 229910052801 chlorine Inorganic materials 0.000 description 39
- 239000002904 solvent Substances 0.000 description 39
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 37
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 37
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 36
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 36
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 36
- 229910052783 alkali metal Inorganic materials 0.000 description 30
- 150000001340 alkali metals Chemical class 0.000 description 30
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 27
- 239000002585 base Substances 0.000 description 26
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 25
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 24
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 24
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 22
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 21
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 21
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 21
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 18
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 18
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 18
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 18
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 18
- IVSZLXZYQVIEFR-UHFFFAOYSA-N 1,3-Dimethylbenzene Natural products CC1=CC=CC(C)=C1 IVSZLXZYQVIEFR-UHFFFAOYSA-N 0.000 description 16
- 241000196324 Embryophyta Species 0.000 description 16
- URLKBWYHVLBVBO-UHFFFAOYSA-N Para-Xylene Chemical group CC1=CC=C(C)C=C1 URLKBWYHVLBVBO-UHFFFAOYSA-N 0.000 description 16
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 16
- 125000004448 alkyl carbonyl group Chemical group 0.000 description 16
- RDOXTESZEPMUJZ-UHFFFAOYSA-N anisole Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 16
- CTQNGGLPUBDAKN-UHFFFAOYSA-N o-dimethylbenzene Natural products CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 16
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 15
- 238000006243 chemical reaction Methods 0.000 description 15
- PAMIQIKDUOTOBW-UHFFFAOYSA-N 1-methylpiperidine Chemical compound CN1CCCCC1 PAMIQIKDUOTOBW-UHFFFAOYSA-N 0.000 description 14
- OISVCGZHLKNMSJ-UHFFFAOYSA-N 2,6-dimethylpyridine Chemical compound CC1=CC=CC(C)=N1 OISVCGZHLKNMSJ-UHFFFAOYSA-N 0.000 description 14
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 description 14
- 238000003776 cleavage reaction Methods 0.000 description 14
- FDPIMTJIUBPUKL-UHFFFAOYSA-N pentan-3-one Chemical compound CCC(=O)CC FDPIMTJIUBPUKL-UHFFFAOYSA-N 0.000 description 14
- 230000007017 scission Effects 0.000 description 14
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 14
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 12
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 12
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 12
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 12
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 12
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 12
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 12
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 12
- 239000004480 active ingredient Substances 0.000 description 12
- 229910000272 alkali metal oxide Inorganic materials 0.000 description 12
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 12
- 239000000243 solution Substances 0.000 description 12
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 11
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 11
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 11
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 10
- 150000001342 alkaline earth metals Chemical class 0.000 description 10
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 10
- 230000002363 herbicidal effect Effects 0.000 description 10
- 238000005684 Liebig rearrangement reaction Methods 0.000 description 9
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 9
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 9
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 9
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 9
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 9
- 239000003921 oil Substances 0.000 description 9
- 239000000047 product Substances 0.000 description 9
- 239000012312 sodium hydride Substances 0.000 description 9
- 229910000104 sodium hydride Inorganic materials 0.000 description 9
- 238000005160 1H NMR spectroscopy Methods 0.000 description 8
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 description 8
- 244000097706 Lagurus ovatus Species 0.000 description 8
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 8
- 239000002253 acid Substances 0.000 description 8
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 150000002576 ketones Chemical class 0.000 description 8
- UZKWTJUDCOPSNM-UHFFFAOYSA-N methoxybenzene Substances CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 8
- 239000003960 organic solvent Substances 0.000 description 8
- 150000003222 pyridines Chemical class 0.000 description 8
- 150000003254 radicals Chemical class 0.000 description 8
- CSDQQAQKBAQLLE-UHFFFAOYSA-N 4-(4-chlorophenyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine Chemical compound C1=CC(Cl)=CC=C1C1C(C=CS2)=C2CCN1 CSDQQAQKBAQLLE-UHFFFAOYSA-N 0.000 description 7
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 7
- 240000006122 Chenopodium album Species 0.000 description 7
- 235000011498 Chenopodium album var missouriense Nutrition 0.000 description 7
- 235000013328 Chenopodium album var. album Nutrition 0.000 description 7
- 235000014052 Chenopodium album var. microphyllum Nutrition 0.000 description 7
- 235000014050 Chenopodium album var. stevensii Nutrition 0.000 description 7
- 235000013012 Chenopodium album var. striatum Nutrition 0.000 description 7
- HOPRXXXSABQWAV-UHFFFAOYSA-N anhydrous collidine Natural products CC1=CC=NC(C)=C1C HOPRXXXSABQWAV-UHFFFAOYSA-N 0.000 description 7
- 238000009835 boiling Methods 0.000 description 7
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 7
- 229910052794 bromium Inorganic materials 0.000 description 7
- UTBIMNXEDGNJFE-UHFFFAOYSA-N collidine Natural products CC1=CC=C(C)C(C)=N1 UTBIMNXEDGNJFE-UHFFFAOYSA-N 0.000 description 7
- HPYNZHMRTTWQTB-UHFFFAOYSA-N dimethylpyridine Natural products CC1=CC=CN=C1C HPYNZHMRTTWQTB-UHFFFAOYSA-N 0.000 description 7
- 239000006185 dispersion Substances 0.000 description 7
- 150000002170 ethers Chemical class 0.000 description 7
- 150000008282 halocarbons Chemical class 0.000 description 7
- 150000002484 inorganic compounds Chemical class 0.000 description 7
- 229910010272 inorganic material Inorganic materials 0.000 description 7
- 238000002955 isolation Methods 0.000 description 7
- 229910000103 lithium hydride Inorganic materials 0.000 description 7
- 239000000395 magnesium oxide Substances 0.000 description 7
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 7
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 7
- 150000002825 nitriles Chemical class 0.000 description 7
- 150000007530 organic bases Chemical class 0.000 description 7
- NTTOTNSKUYCDAV-UHFFFAOYSA-N potassium hydride Chemical compound [KH] NTTOTNSKUYCDAV-UHFFFAOYSA-N 0.000 description 7
- 229910000105 potassium hydride Inorganic materials 0.000 description 7
- 239000011541 reaction mixture Substances 0.000 description 7
- 238000007142 ring opening reaction Methods 0.000 description 7
- 239000007858 starting material Substances 0.000 description 7
- GFYHSKONPJXCDE-UHFFFAOYSA-N sym-collidine Natural products CC1=CN=C(C)C(C)=C1 GFYHSKONPJXCDE-UHFFFAOYSA-N 0.000 description 7
- 150000003512 tertiary amines Chemical class 0.000 description 7
- 238000010626 work up procedure Methods 0.000 description 7
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 6
- 229910004013 NO 2 Inorganic materials 0.000 description 6
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 6
- 150000001298 alcohols Chemical class 0.000 description 6
- 229910001860 alkaline earth metal hydroxide Inorganic materials 0.000 description 6
- 229910000287 alkaline earth metal oxide Inorganic materials 0.000 description 6
- 125000003282 alkyl amino group Chemical group 0.000 description 6
- 229910000019 calcium carbonate Inorganic materials 0.000 description 6
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 6
- 239000000920 calcium hydroxide Substances 0.000 description 6
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 6
- BRPQOXSCLDDYGP-UHFFFAOYSA-N calcium oxide Chemical compound [O-2].[Ca+2] BRPQOXSCLDDYGP-UHFFFAOYSA-N 0.000 description 6
- 239000000292 calcium oxide Substances 0.000 description 6
- ODINCKMPIJJUCX-UHFFFAOYSA-N calcium oxide Inorganic materials [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 description 6
- 239000004359 castor oil Substances 0.000 description 6
- 235000019438 castor oil Nutrition 0.000 description 6
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 6
- XGZVUEUWXADBQD-UHFFFAOYSA-L lithium carbonate Chemical compound [Li+].[Li+].[O-]C([O-])=O XGZVUEUWXADBQD-UHFFFAOYSA-L 0.000 description 6
- 229910052808 lithium carbonate Inorganic materials 0.000 description 6
- FUJCRWPEOMXPAD-UHFFFAOYSA-N lithium oxide Chemical compound [Li+].[Li+].[O-2] FUJCRWPEOMXPAD-UHFFFAOYSA-N 0.000 description 6
- 229910001947 lithium oxide Inorganic materials 0.000 description 6
- PJGSXYOJTGTZAV-UHFFFAOYSA-N pinacolone Chemical compound CC(=O)C(C)(C)C PJGSXYOJTGTZAV-UHFFFAOYSA-N 0.000 description 6
- 229910000027 potassium carbonate Inorganic materials 0.000 description 6
- FVSKHRXBFJPNKK-UHFFFAOYSA-N propionitrile Chemical compound CCC#N FVSKHRXBFJPNKK-UHFFFAOYSA-N 0.000 description 6
- KKCBUQHMOMHUOY-UHFFFAOYSA-N sodium oxide Chemical compound [O-2].[Na+].[Na+] KKCBUQHMOMHUOY-UHFFFAOYSA-N 0.000 description 6
- 229910001948 sodium oxide Inorganic materials 0.000 description 6
- 125000004454 (C1-C6) alkoxycarbonyl group Chemical group 0.000 description 5
- 125000004916 (C1-C6) alkylcarbonyl group Chemical group 0.000 description 5
- KLDLRDSRCMJKGM-UHFFFAOYSA-N 3-[chloro-(2-oxo-1,3-oxazolidin-3-yl)phosphoryl]-1,3-oxazolidin-2-one Chemical compound C1COC(=O)N1P(=O)(Cl)N1CCOC1=O KLDLRDSRCMJKGM-UHFFFAOYSA-N 0.000 description 5
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 5
- 239000013543 active substance Substances 0.000 description 5
- 229910052799 carbon Inorganic materials 0.000 description 5
- 238000011097 chromatography purification Methods 0.000 description 5
- 239000012230 colorless oil Substances 0.000 description 5
- 239000012141 concentrate Substances 0.000 description 5
- 239000013078 crystal Substances 0.000 description 5
- 239000000839 emulsion Substances 0.000 description 5
- 125000001153 fluoro group Chemical group F* 0.000 description 5
- 150000002332 glycine derivatives Chemical class 0.000 description 5
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 5
- 229920006395 saturated elastomer Polymers 0.000 description 5
- 239000000741 silica gel Substances 0.000 description 5
- 229910002027 silica gel Inorganic materials 0.000 description 5
- 239000007787 solid Substances 0.000 description 5
- 239000000725 suspension Substances 0.000 description 5
- 125000004890 (C1-C6) alkylamino group Chemical group 0.000 description 4
- HSJKGGMUJITCBW-UHFFFAOYSA-N 3-hydroxybutanal Chemical compound CC(O)CC=O HSJKGGMUJITCBW-UHFFFAOYSA-N 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 4
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 4
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 4
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 4
- 230000003213 activating effect Effects 0.000 description 4
- 150000001408 amides Chemical class 0.000 description 4
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 239000003054 catalyst Substances 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 239000003638 chemical reducing agent Substances 0.000 description 4
- 230000012010 growth Effects 0.000 description 4
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 4
- 229910052757 nitrogen Inorganic materials 0.000 description 4
- LFGREXWGYUGZLY-UHFFFAOYSA-N phosphoryl Chemical group [P]=O LFGREXWGYUGZLY-UHFFFAOYSA-N 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- IZXIZTKNFFYFOF-UHFFFAOYSA-N 2-Oxazolidone Chemical class O=C1NCCO1 IZXIZTKNFFYFOF-UHFFFAOYSA-N 0.000 description 3
- CAAMSDWKXXPUJR-UHFFFAOYSA-N 3,5-dihydro-4H-imidazol-4-one Chemical class O=C1CNC=N1 CAAMSDWKXXPUJR-UHFFFAOYSA-N 0.000 description 3
- 240000005702 Galium aparine Species 0.000 description 3
- 150000007513 acids Chemical class 0.000 description 3
- 150000001450 anions Chemical class 0.000 description 3
- 125000003118 aryl group Chemical group 0.000 description 3
- 239000000969 carrier Substances 0.000 description 3
- 230000003197 catalytic effect Effects 0.000 description 3
- 150000001768 cations Chemical class 0.000 description 3
- 238000009833 condensation Methods 0.000 description 3
- 230000005494 condensation Effects 0.000 description 3
- 239000002270 dispersing agent Substances 0.000 description 3
- 239000008187 granular material Substances 0.000 description 3
- 229910052740 iodine Inorganic materials 0.000 description 3
- CRGZYKWWYNQGEC-UHFFFAOYSA-N magnesium;methanolate Chemical compound [Mg+2].[O-]C.[O-]C CRGZYKWWYNQGEC-UHFFFAOYSA-N 0.000 description 3
- DVSDBMFJEQPWNO-UHFFFAOYSA-N methyllithium Chemical compound C[Li] DVSDBMFJEQPWNO-UHFFFAOYSA-N 0.000 description 3
- 239000002480 mineral oil Substances 0.000 description 3
- 235000010446 mineral oil Nutrition 0.000 description 3
- 239000006072 paste Substances 0.000 description 3
- 239000012071 phase Substances 0.000 description 3
- NHKJPPKXDNZFBJ-UHFFFAOYSA-N phenyllithium Chemical compound [Li]C1=CC=CC=C1 NHKJPPKXDNZFBJ-UHFFFAOYSA-N 0.000 description 3
- 239000004033 plastic Substances 0.000 description 3
- 229920003023 plastic Polymers 0.000 description 3
- RPDAUEIUDPHABB-UHFFFAOYSA-N potassium ethoxide Chemical compound [K+].CC[O-] RPDAUEIUDPHABB-UHFFFAOYSA-N 0.000 description 3
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 125000006239 protecting group Chemical group 0.000 description 3
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 3
- 159000000000 sodium salts Chemical class 0.000 description 3
- 241000894007 species Species 0.000 description 3
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 3
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 3
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 2
- BDNKZNFMNDZQMI-UHFFFAOYSA-N 1,3-diisopropylcarbodiimide Chemical compound CC(C)N=C=NC(C)C BDNKZNFMNDZQMI-UHFFFAOYSA-N 0.000 description 2
- JVSFQJZRHXAUGT-UHFFFAOYSA-N 2,2-dimethylpropanoyl chloride Chemical compound CC(C)(C)C(Cl)=O JVSFQJZRHXAUGT-UHFFFAOYSA-N 0.000 description 2
- GRKMMLDAPZYEJP-UHFFFAOYSA-N 2-amino-3-[benzyl(formyl)amino]-3-phenylpropanoic acid Chemical compound C=1C=CC=CC=1C(C(N)C(O)=O)N(C=O)CC1=CC=CC=C1 GRKMMLDAPZYEJP-UHFFFAOYSA-N 0.000 description 2
- ZFFBIQMNKOJDJE-UHFFFAOYSA-N 2-bromo-1,2-diphenylethanone Chemical compound C=1C=CC=CC=1C(Br)C(=O)C1=CC=CC=C1 ZFFBIQMNKOJDJE-UHFFFAOYSA-N 0.000 description 2
- HDECRAPHCDXMIJ-UHFFFAOYSA-N 2-methylbenzenesulfonyl chloride Chemical compound CC1=CC=CC=C1S(Cl)(=O)=O HDECRAPHCDXMIJ-UHFFFAOYSA-N 0.000 description 2
- YOETUEMZNOLGDB-UHFFFAOYSA-N 2-methylpropyl carbonochloridate Chemical compound CC(C)COC(Cl)=O YOETUEMZNOLGDB-UHFFFAOYSA-N 0.000 description 2
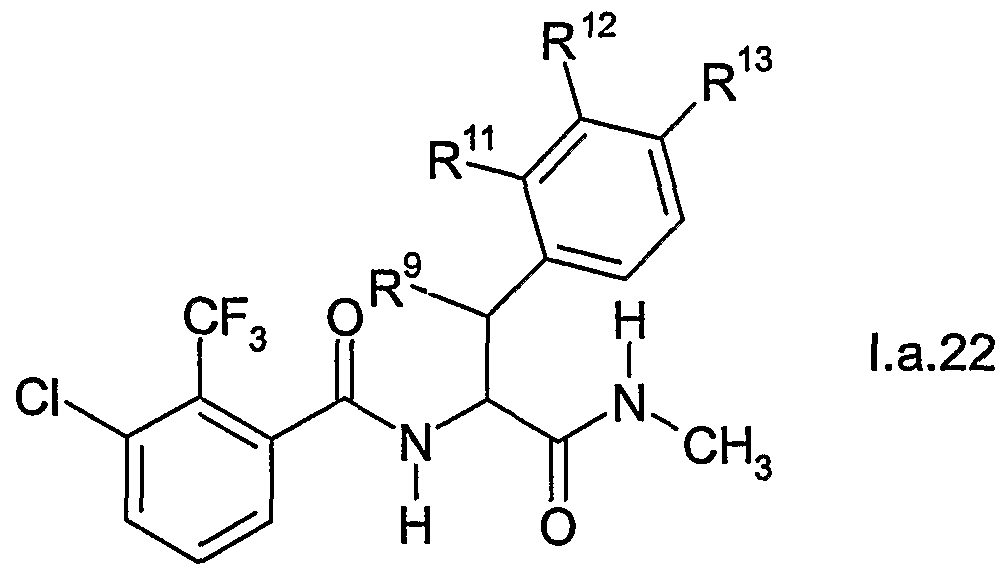
- DAVGYAZXHZUVIQ-UHFFFAOYSA-N 3-chloro-2-(trifluoromethyl)benzoic acid Chemical compound OC(=O)C1=CC=CC(Cl)=C1C(F)(F)F DAVGYAZXHZUVIQ-UHFFFAOYSA-N 0.000 description 2
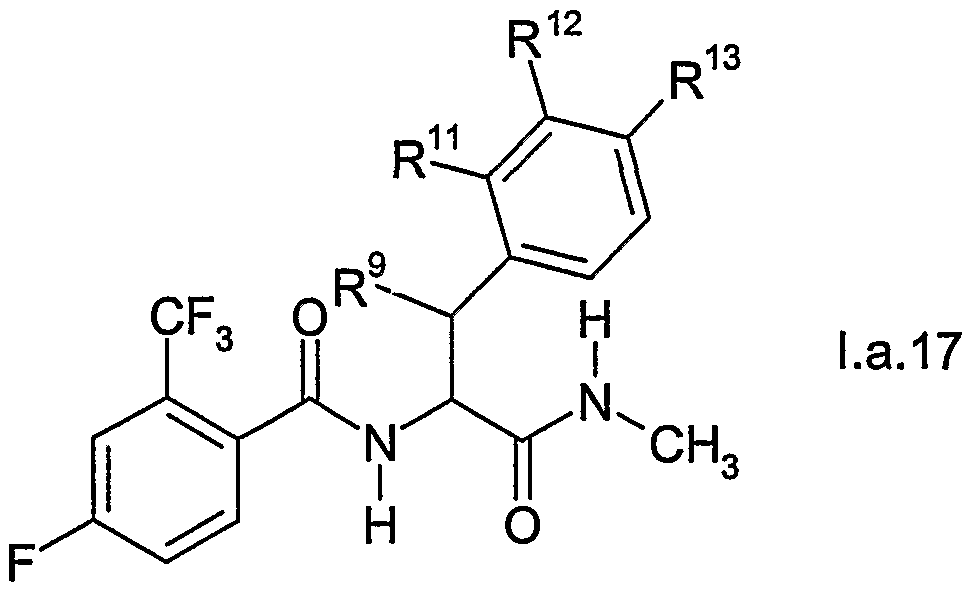
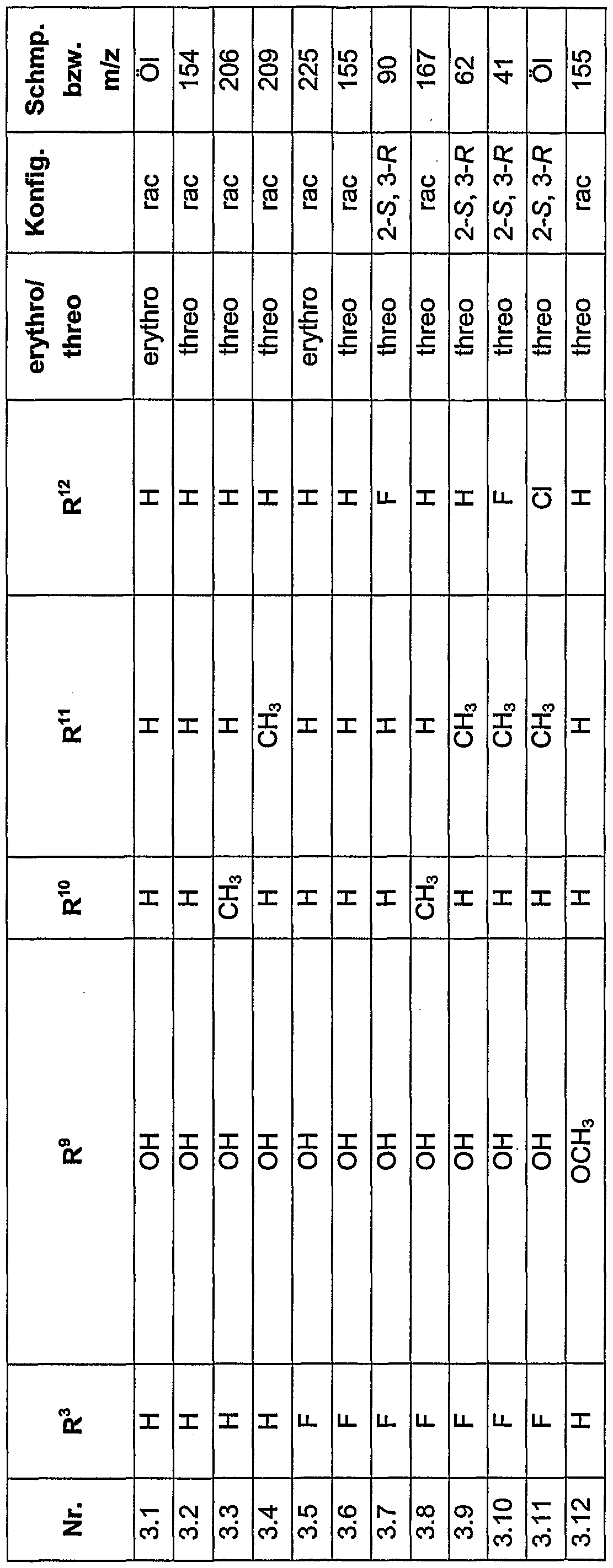
- IIEKHXMFOCJOQW-JKSUJKDBSA-N 4-fluoro-n-[(1r,2s)-1-hydroxy-3-(methylamino)-1-(2-methylphenyl)-3-oxopropan-2-yl]-2-(trifluoromethyl)benzamide Chemical compound N([C@H](C(=O)NC)[C@H](O)C=1C(=CC=CC=1)C)C(=O)C1=CC=C(F)C=C1C(F)(F)F IIEKHXMFOCJOQW-JKSUJKDBSA-N 0.000 description 2
- 239000005995 Aluminium silicate Substances 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- 235000021533 Beta vulgaris Nutrition 0.000 description 2
- 241000335053 Beta vulgaris Species 0.000 description 2
- 0 CNC(C(C(*)c1c(*=C)c(*)c(*)cc1)NC(c(ccc(F)c1C(F)(F)F)c1Cl)=O)=O Chemical compound CNC(C(C(*)c1c(*=C)c(*)c(*)cc1)NC(c(ccc(F)c1C(F)(F)F)c1Cl)=O)=O 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- 244000025254 Cannabis sativa Species 0.000 description 2
- 235000012766 Cannabis sativa ssp. sativa var. sativa Nutrition 0.000 description 2
- 235000012765 Cannabis sativa ssp. sativa var. spontanea Nutrition 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- 235000014375 Curcuma Nutrition 0.000 description 2
- 244000163122 Curcuma domestica Species 0.000 description 2
- QOSSAOTZNIDXMA-UHFFFAOYSA-N Dicylcohexylcarbodiimide Chemical compound C1CCCCC1N=C=NC1CCCCC1 QOSSAOTZNIDXMA-UHFFFAOYSA-N 0.000 description 2
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 2
- 235000010469 Glycine max Nutrition 0.000 description 2
- 244000068988 Glycine max Species 0.000 description 2
- 244000299507 Gossypium hirsutum Species 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- 239000012359 Methanesulfonyl chloride Substances 0.000 description 2
- 240000007594 Oryza sativa Species 0.000 description 2
- 235000007164 Oryza sativa Nutrition 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- 244000062793 Sorghum vulgare Species 0.000 description 2
- 235000021307 Triticum Nutrition 0.000 description 2
- 244000098338 Triticum aestivum Species 0.000 description 2
- 240000008042 Zea mays Species 0.000 description 2
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 2
- FLBYKICJJBFQPJ-ZETCQYMHSA-N [2-amino-6-[(2s)-2-amino-2-carboxyethyl]phenyl]-diazonioazanide Chemical class OC(=O)[C@@H](N)CC1=CC=CC(N)=C1N=[N+]=[N-] FLBYKICJJBFQPJ-ZETCQYMHSA-N 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 125000002252 acyl group Chemical group 0.000 description 2
- 150000001335 aliphatic alkanes Chemical class 0.000 description 2
- 150000004791 alkyl magnesium halides Chemical class 0.000 description 2
- 235000012211 aluminium silicate Nutrition 0.000 description 2
- 125000004682 aminothiocarbonyl group Chemical group NC(=S)* 0.000 description 2
- 150000008064 anhydrides Chemical class 0.000 description 2
- 125000004104 aryloxy group Chemical group 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 150000001541 aziridines Chemical class 0.000 description 2
- CSKNSYBAZOQPLR-UHFFFAOYSA-N benzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC=C1 CSKNSYBAZOQPLR-UHFFFAOYSA-N 0.000 description 2
- MUALRAIOVNYAIW-UHFFFAOYSA-N binap Chemical compound C1=CC=CC=C1P(C=1C(=C2C=CC=CC2=CC=1)C=1C2=CC=CC=C2C=CC=1P(C=1C=CC=CC=1)C=1C=CC=CC=1)C1=CC=CC=C1 MUALRAIOVNYAIW-UHFFFAOYSA-N 0.000 description 2
- 244000309464 bull Species 0.000 description 2
- YSMHTFWPDRJCMN-UHFFFAOYSA-N butan-2-yl carbonochloridate Chemical compound CCC(C)OC(Cl)=O YSMHTFWPDRJCMN-UHFFFAOYSA-N 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 235000009120 camo Nutrition 0.000 description 2
- 235000013877 carbamide Nutrition 0.000 description 2
- PFKFTWBEEFSNDU-UHFFFAOYSA-N carbonyldiimidazole Chemical compound C1=CN=CN1C(=O)N1C=CN=C1 PFKFTWBEEFSNDU-UHFFFAOYSA-N 0.000 description 2
- 235000005607 chanvre indien Nutrition 0.000 description 2
- 239000007795 chemical reaction product Substances 0.000 description 2
- FZFAMSAMCHXGEF-UHFFFAOYSA-N chloro formate Chemical compound ClOC=O FZFAMSAMCHXGEF-UHFFFAOYSA-N 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- AOGYCOYQMAVAFD-UHFFFAOYSA-N chlorocarbonic acid Chemical class OC(Cl)=O AOGYCOYQMAVAFD-UHFFFAOYSA-N 0.000 description 2
- 239000007859 condensation product Substances 0.000 description 2
- 244000038559 crop plants Species 0.000 description 2
- 239000012043 crude product Substances 0.000 description 2
- VFLDPWHFBUODDF-FCXRPNKRSA-N curcumin Chemical compound C1=C(O)C(OC)=CC(\C=C\C(=O)CC(=O)\C=C\C=2C=C(OC)C(O)=CC=2)=C1 VFLDPWHFBUODDF-FCXRPNKRSA-N 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 230000029087 digestion Effects 0.000 description 2
- BGRWYRAHAFMIBJ-UHFFFAOYSA-N diisopropylcarbodiimide Natural products CC(C)NC(=O)NC(C)C BGRWYRAHAFMIBJ-UHFFFAOYSA-N 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- IOHYSBURCJOBFE-DLBZAZTESA-N ethyl (2s,3r)-2-[[4-fluoro-2-(trifluoromethyl)benzoyl]amino]-3-hydroxy-3-(2-methylphenyl)propanoate Chemical compound N([C@H](C(=O)OCC)[C@H](O)C=1C(=CC=CC=1)C)C(=O)C1=CC=C(F)C=C1C(F)(F)F IOHYSBURCJOBFE-DLBZAZTESA-N 0.000 description 2
- IKVBHYDIOFTLJM-UHFFFAOYSA-N ethyl 1-benzyl-5-phenyl-4,5-dihydroimidazole-4-carboxylate Chemical compound C=1C=CC=CC=1C1C(C(=O)OCC)N=CN1CC1=CC=CC=C1 IKVBHYDIOFTLJM-UHFFFAOYSA-N 0.000 description 2
- XBMUPQDDPSNHJX-UHFFFAOYSA-N ethyl 2-amino-3-(2-methylphenyl)-3-oxopropanoate;hydrochloride Chemical compound Cl.CCOC(=O)C(N)C(=O)C1=CC=CC=C1C XBMUPQDDPSNHJX-UHFFFAOYSA-N 0.000 description 2
- RIFGWPKJUGCATF-UHFFFAOYSA-N ethyl chloroformate Chemical compound CCOC(Cl)=O RIFGWPKJUGCATF-UHFFFAOYSA-N 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 235000013312 flour Nutrition 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 230000035784 germination Effects 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- 239000011487 hemp Substances 0.000 description 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 2
- 230000007062 hydrolysis Effects 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- 150000002462 imidazolines Chemical class 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- ZXEKIIBDNHEJCQ-UHFFFAOYSA-N isobutanol Chemical compound CC(C)CO ZXEKIIBDNHEJCQ-UHFFFAOYSA-N 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 2
- 229910052744 lithium Inorganic materials 0.000 description 2
- AFRJJFRNGGLMDW-UHFFFAOYSA-N lithium amide Chemical compound [Li+].[NH2-] AFRJJFRNGGLMDW-UHFFFAOYSA-N 0.000 description 2
- YNESATAKKCNGOF-UHFFFAOYSA-N lithium bis(trimethylsilyl)amide Chemical compound [Li+].C[Si](C)(C)[N-][Si](C)(C)C YNESATAKKCNGOF-UHFFFAOYSA-N 0.000 description 2
- ZCSHNCUQKCANBX-UHFFFAOYSA-N lithium diisopropylamide Chemical compound [Li+].CC(C)[N-]C(C)C ZCSHNCUQKCANBX-UHFFFAOYSA-N 0.000 description 2
- CCERQOYLJJULMD-UHFFFAOYSA-M magnesium;carbanide;chloride Chemical compound [CH3-].[Mg+2].[Cl-] CCERQOYLJJULMD-UHFFFAOYSA-M 0.000 description 2
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 description 2
- WSFSSNUMVMOOMR-NJFSPNSNSA-N methanone Chemical compound O=[14CH2] WSFSSNUMVMOOMR-NJFSPNSNSA-N 0.000 description 2
- LSRUQLUVOKGZBJ-UHFFFAOYSA-N methyl 2-amino-3-[benzyl(formyl)amino]-3-phenylpropanoate Chemical compound C=1C=CC=CC=1C(C(N)C(=O)OC)N(C=O)CC1=CC=CC=C1 LSRUQLUVOKGZBJ-UHFFFAOYSA-N 0.000 description 2
- XHNMSVOAKPQBFD-UHFFFAOYSA-N methyl 3-[benzyl(formyl)amino]-2-[[4-fluoro-2-(trifluoromethyl)benzoyl]amino]-3-phenylpropanoate Chemical compound C=1C=CC=CC=1CN(C=O)C(C=1C=CC=CC=1)C(C(=O)OC)NC(=O)C1=CC=C(F)C=C1C(F)(F)F XHNMSVOAKPQBFD-UHFFFAOYSA-N 0.000 description 2
- XMJHPCRAQCTCFT-UHFFFAOYSA-N methyl chloroformate Chemical compound COC(Cl)=O XMJHPCRAQCTCFT-UHFFFAOYSA-N 0.000 description 2
- 244000005700 microbiome Species 0.000 description 2
- 235000010755 mineral Nutrition 0.000 description 2
- 239000011707 mineral Substances 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- GDJKEUNPXYLPHG-AWEZNQCLSA-N n-[(2s)-1-amino-1-oxo-3-phenylpropan-2-yl]benzamide Chemical class C([C@@H](C(=O)N)NC(=O)C=1C=CC=CC=1)C1=CC=CC=C1 GDJKEUNPXYLPHG-AWEZNQCLSA-N 0.000 description 2
- 150000002790 naphthalenes Chemical class 0.000 description 2
- 239000012074 organic phase Substances 0.000 description 2
- 150000002902 organometallic compounds Chemical class 0.000 description 2
- 150000002917 oxazolidines Chemical class 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- WLJVXDMOQOGPHL-UHFFFAOYSA-N phenylacetic acid Chemical compound OC(=O)CC1=CC=CC=C1 WLJVXDMOQOGPHL-UHFFFAOYSA-N 0.000 description 2
- 229920000137 polyphosphoric acid Polymers 0.000 description 2
- ZRLVQFQTCMUIRM-UHFFFAOYSA-N potassium;2-methylbutan-2-olate Chemical compound [K+].CCC(C)(C)[O-] ZRLVQFQTCMUIRM-UHFFFAOYSA-N 0.000 description 2
- CAEWJEXPFKNBQL-UHFFFAOYSA-N prop-2-enyl carbonochloridate Chemical compound ClC(=O)OCC=C CAEWJEXPFKNBQL-UHFFFAOYSA-N 0.000 description 2
- ODZPKZBBUMBTMG-UHFFFAOYSA-N sodium amide Chemical compound [NH2-].[Na+] ODZPKZBBUMBTMG-UHFFFAOYSA-N 0.000 description 2
- 239000002689 soil Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- YBBRCQOCSYXUOC-UHFFFAOYSA-N sulfuryl dichloride Chemical class ClS(Cl)(=O)=O YBBRCQOCSYXUOC-UHFFFAOYSA-N 0.000 description 2
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 2
- 150000003672 ureas Chemical class 0.000 description 2
- 235000013311 vegetables Nutrition 0.000 description 2
- 239000011701 zinc Substances 0.000 description 2
- 229910052725 zinc Inorganic materials 0.000 description 2
- JWZZKOKVBUJMES-UHFFFAOYSA-N (+-)-Isoprenaline Chemical compound CC(C)NCC(O)C1=CC=C(O)C(O)=C1 JWZZKOKVBUJMES-UHFFFAOYSA-N 0.000 description 1
- JIGYLPRAWVJJLD-QMMMGPOBSA-N (2s)-2-(n-aminoanilino)-3-hydroxypropanoic acid Chemical class OC[C@@H](C(O)=O)N(N)C1=CC=CC=C1 JIGYLPRAWVJJLD-QMMMGPOBSA-N 0.000 description 1
- LYBVTBCJUPMLBJ-HNNXBMFYSA-N (2s)-2-(n-benzylanilino)-3-hydroxypropanoic acid Chemical class C=1C=CC=CC=1N([C@@H](CO)C(O)=O)CC1=CC=CC=C1 LYBVTBCJUPMLBJ-HNNXBMFYSA-N 0.000 description 1
- RQKSXXDDFRCMJF-QMMMGPOBSA-N (2s)-2-(n-carbamoylanilino)-3-hydroxypropanoic acid Chemical class OC[C@@H](C(O)=O)N(C(=O)N)C1=CC=CC=C1 RQKSXXDDFRCMJF-QMMMGPOBSA-N 0.000 description 1
- 125000004760 (C1-C4) alkylsulfonylamino group Chemical group 0.000 description 1
- 125000004739 (C1-C6) alkylsulfonyl group Chemical group 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- LAXRNWSASWOFOT-UHFFFAOYSA-J (cymene)ruthenium dichloride dimer Chemical compound [Cl-].[Cl-].[Cl-].[Cl-].[Ru+2].[Ru+2].CC(C)C1=CC=C(C)C=C1.CC(C)C1=CC=C(C)C=C1 LAXRNWSASWOFOT-UHFFFAOYSA-J 0.000 description 1
- 125000006079 1,1,2-trimethyl-2-propenyl group Chemical group 0.000 description 1
- 125000004884 1,1,2-trimethylpropylcarbonyl group Chemical group CC(C(C)C)(C(=O)*)C 0.000 description 1
- 125000006059 1,1-dimethyl-2-butenyl group Chemical group 0.000 description 1
- 125000006033 1,1-dimethyl-2-propenyl group Chemical group 0.000 description 1
- 125000006060 1,1-dimethyl-3-butenyl group Chemical group 0.000 description 1
- 125000004876 1,1-dimethylbutylcarbonyl group Chemical group CC(CCC)(C(=O)*)C 0.000 description 1
- 125000004747 1,1-dimethylethoxycarbonyl group Chemical group CC(C)(OC(=O)*)C 0.000 description 1
- 125000004866 1,1-dimethylethylcarbonyl group Chemical group CC(C)(C(=O)*)C 0.000 description 1
- 125000004711 1,1-dimethylethylthio group Chemical group CC(C)(S*)C 0.000 description 1
- 125000004867 1,1-dimethylpropylcarbonyl group Chemical group CC(CC)(C(=O)*)C 0.000 description 1
- OTPDWCMLUKMQNO-UHFFFAOYSA-N 1,2,3,4-tetrahydropyrimidine Chemical compound C1NCC=CN1 OTPDWCMLUKMQNO-UHFFFAOYSA-N 0.000 description 1
- 125000001724 1,2,3-oxadiazol-4-yl group Chemical group [H]C1=C(*)N=NO1 0.000 description 1
- 125000004512 1,2,3-thiadiazol-4-yl group Chemical group S1N=NC(=C1)* 0.000 description 1
- 125000001607 1,2,3-triazol-1-yl group Chemical group [*]N1N=NC([H])=C1[H] 0.000 description 1
- 125000001359 1,2,3-triazol-4-yl group Chemical group [H]N1N=NC([*])=C1[H] 0.000 description 1
- 125000001766 1,2,4-oxadiazol-3-yl group Chemical group [H]C1=NC(*)=NO1 0.000 description 1
- 125000004515 1,2,4-thiadiazol-3-yl group Chemical group S1N=C(N=C1)* 0.000 description 1
- 125000004516 1,2,4-thiadiazol-5-yl group Chemical group S1N=CN=C1* 0.000 description 1
- YGTAZGSLCXNBQL-UHFFFAOYSA-N 1,2,4-thiadiazole Chemical class C=1N=CSN=1 YGTAZGSLCXNBQL-UHFFFAOYSA-N 0.000 description 1
- 125000003626 1,2,4-triazol-1-yl group Chemical group [*]N1N=C([H])N=C1[H] 0.000 description 1
- 125000001305 1,2,4-triazol-3-yl group Chemical group [H]N1N=C([*])N=C1[H] 0.000 description 1
- PAAZPARNPHGIKF-UHFFFAOYSA-N 1,2-dibromoethane Chemical compound BrCCBr PAAZPARNPHGIKF-UHFFFAOYSA-N 0.000 description 1
- 125000006061 1,2-dimethyl-1-butenyl group Chemical group 0.000 description 1
- 125000006034 1,2-dimethyl-1-propenyl group Chemical group 0.000 description 1
- 125000006062 1,2-dimethyl-2-butenyl group Chemical group 0.000 description 1
- 125000006035 1,2-dimethyl-2-propenyl group Chemical group 0.000 description 1
- 125000006063 1,2-dimethyl-3-butenyl group Chemical group 0.000 description 1
- 125000005918 1,2-dimethylbutyl group Chemical group 0.000 description 1
- 125000004877 1,2-dimethylbutylcarbonyl group Chemical group CC(C(CC)C)C(=O)* 0.000 description 1
- 125000004868 1,2-dimethylpropylcarbonyl group Chemical group CC(C(C)C)C(=O)* 0.000 description 1
- 125000004509 1,3,4-oxadiazol-2-yl group Chemical group O1C(=NN=C1)* 0.000 description 1
- 150000004869 1,3,4-thiadiazoles Chemical class 0.000 description 1
- 125000004317 1,3,5-triazin-2-yl group Chemical group [H]C1=NC(*)=NC([H])=N1 0.000 description 1
- 125000006064 1,3-dimethyl-1-butenyl group Chemical group 0.000 description 1
- 125000006065 1,3-dimethyl-2-butenyl group Chemical group 0.000 description 1
- 125000006066 1,3-dimethyl-3-butenyl group Chemical group 0.000 description 1
- 125000004878 1,3-dimethylbutylcarbonyl group Chemical group CC(CC(C)C)C(=O)* 0.000 description 1
- GCSBYWTVHSKTNC-UHFFFAOYSA-N 1,3-oxazolidin-5-one Chemical class O=C1CNCO1 GCSBYWTVHSKTNC-UHFFFAOYSA-N 0.000 description 1
- WGJCBBASTRWVJL-UHFFFAOYSA-N 1,3-thiazolidine-2-thione Chemical class SC1=NCCS1 WGJCBBASTRWVJL-UHFFFAOYSA-N 0.000 description 1
- XPIQQGXRDHXMGQ-UHFFFAOYSA-N 1-bromo-3-chloro-2-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=C(Cl)C=CC=C1Br XPIQQGXRDHXMGQ-UHFFFAOYSA-N 0.000 description 1
- 125000004973 1-butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000004972 1-butynyl group Chemical group [H]C([H])([H])C([H])([H])C#C* 0.000 description 1
- CMCBDXRRFKYBDG-UHFFFAOYSA-N 1-dodecoxydodecane Chemical compound CCCCCCCCCCCCOCCCCCCCCCCCC CMCBDXRRFKYBDG-UHFFFAOYSA-N 0.000 description 1
- 125000006080 1-ethyl-1-methyl-2-propenyl group Chemical group 0.000 description 1
- 125000004886 1-ethyl-1-methylpropylcarbonyl group Chemical group C(C)C(CC)(C(=O)*)C 0.000 description 1
- 125000006036 1-ethyl-1-propenyl group Chemical group 0.000 description 1
- 125000006074 1-ethyl-2-butenyl group Chemical group 0.000 description 1
- 125000006081 1-ethyl-2-methyl-1-propenyl group Chemical group 0.000 description 1
- 125000006082 1-ethyl-2-methyl-2-propenyl group Chemical group 0.000 description 1
- 125000004887 1-ethyl-2-methylpropylcarbonyl group Chemical group C(C)C(C(C)C)C(=O)* 0.000 description 1
- 125000004736 1-ethyl-2-methylpropylthio group Chemical group C(C)C(C(C)C)S* 0.000 description 1
- 125000006037 1-ethyl-2-propenyl group Chemical group 0.000 description 1
- 125000006075 1-ethyl-3-butenyl group Chemical group 0.000 description 1
- 125000006218 1-ethylbutyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004882 1-ethylbutylcarbonyl group Chemical group C(C)C(CCC)C(=O)* 0.000 description 1
- 125000004870 1-ethylpropylcarbonyl group Chemical group C(C)C(CC)C(=O)* 0.000 description 1
- HNEGJTWNOOWEMH-UHFFFAOYSA-N 1-fluoropropane Chemical group [CH2]CCF HNEGJTWNOOWEMH-UHFFFAOYSA-N 0.000 description 1
- 125000006039 1-hexenyl group Chemical group 0.000 description 1
- CXBDYQVECUFKRK-UHFFFAOYSA-N 1-methoxybutane Chemical compound CCCCOC CXBDYQVECUFKRK-UHFFFAOYSA-N 0.000 description 1
- 125000006025 1-methyl-1-butenyl group Chemical group 0.000 description 1
- 125000006044 1-methyl-1-pentenyl group Chemical group 0.000 description 1
- 125000006019 1-methyl-1-propenyl group Chemical group 0.000 description 1
- 125000006028 1-methyl-2-butenyl group Chemical group 0.000 description 1
- 125000006048 1-methyl-2-pentenyl group Chemical group 0.000 description 1
- 125000006021 1-methyl-2-propenyl group Chemical group 0.000 description 1
- 125000006030 1-methyl-3-butenyl group Chemical group 0.000 description 1
- 125000006055 1-methyl-4-pentenyl group Chemical group 0.000 description 1
- 125000006018 1-methyl-ethenyl group Chemical group 0.000 description 1
- 125000004679 1-methylbutylcarbonyl group Chemical group CC(CCC)C(=O)* 0.000 description 1
- 125000004743 1-methylethoxycarbonyl group Chemical group CC(C)OC(=O)* 0.000 description 1
- 125000004677 1-methylethylcarbonyl group Chemical group CC(C)C(=O)* 0.000 description 1
- 125000004872 1-methylpentylcarbonyl group Chemical group CC(CCCC)C(=O)* 0.000 description 1
- 125000004678 1-methylpropylcarbonyl group Chemical group CC(CC)C(=O)* 0.000 description 1
- 125000006023 1-pentenyl group Chemical group 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- 125000000530 1-propynyl group Chemical group [H]C([H])([H])C#C* 0.000 description 1
- 125000001462 1-pyrrolyl group Chemical group [*]N1C([H])=C([H])C([H])=C1[H] 0.000 description 1
- XUJLWPFSUCHPQL-UHFFFAOYSA-N 11-methyldodecan-1-ol Chemical compound CC(C)CCCCCCCCCCO XUJLWPFSUCHPQL-UHFFFAOYSA-N 0.000 description 1
- 125000000453 2,2,2-trichloroethyl group Chemical group [H]C([H])(*)C(Cl)(Cl)Cl 0.000 description 1
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- 125000004781 2,2-dichloro-2-fluoroethyl group Chemical group [H]C([H])(*)C(F)(Cl)Cl 0.000 description 1
- 125000004778 2,2-difluoroethyl group Chemical group [H]C([H])(*)C([H])(F)F 0.000 description 1
- 125000006067 2,2-dimethyl-3-butenyl group Chemical group 0.000 description 1
- 125000004879 2,2-dimethylbutylcarbonyl group Chemical group CC(CC(=O)*)(CC)C 0.000 description 1
- KBLAMUYRMZPYLS-UHFFFAOYSA-N 2,3-bis(2-methylpropyl)naphthalene-1-sulfonic acid Chemical compound C1=CC=C2C(S(O)(=O)=O)=C(CC(C)C)C(CC(C)C)=CC2=C1 KBLAMUYRMZPYLS-UHFFFAOYSA-N 0.000 description 1
- 125000006069 2,3-dimethyl-2-butenyl group Chemical group 0.000 description 1
- 125000006070 2,3-dimethyl-3-butenyl group Chemical group 0.000 description 1
- 125000004880 2,3-dimethylbutylcarbonyl group Chemical group CC(CC(=O)*)C(C)C 0.000 description 1
- CGNBQYFXGQHUQP-UHFFFAOYSA-N 2,3-dinitroaniline Chemical class NC1=CC=CC([N+]([O-])=O)=C1[N+]([O-])=O CGNBQYFXGQHUQP-UHFFFAOYSA-N 0.000 description 1
- MHKBMNACOMRIAW-UHFFFAOYSA-N 2,3-dinitrophenol Chemical class OC1=CC=CC([N+]([O-])=O)=C1[N+]([O-])=O MHKBMNACOMRIAW-UHFFFAOYSA-N 0.000 description 1
- GIAFURWZWWWBQT-UHFFFAOYSA-N 2-(2-aminoethoxy)ethanol Chemical compound NCCOCCO GIAFURWZWWWBQT-UHFFFAOYSA-N 0.000 description 1
- GWLPMMFYVKRJSB-UHFFFAOYSA-N 2-(2-azido-3-hydroxyphenyl)propanoic acid Chemical class N(=[N+]=[N-])C1=C(C=CC=C1O)C(C(=O)O)C GWLPMMFYVKRJSB-UHFFFAOYSA-N 0.000 description 1
- PAWQVTBBRAZDMG-UHFFFAOYSA-N 2-(3-bromo-2-fluorophenyl)acetic acid Chemical compound OC(=O)CC1=CC=CC(Br)=C1F PAWQVTBBRAZDMG-UHFFFAOYSA-N 0.000 description 1
- KHZFGSYKTZLZQV-UHFFFAOYSA-N 2-(5-hydroxy-6-hydroxyiminocyclohexa-2,4-dien-1-yl)propanoic acid Chemical class OC(=O)C(C)C1C=CC=C(O)C1=NO KHZFGSYKTZLZQV-UHFFFAOYSA-N 0.000 description 1
- QQZOPKMRPOGIEB-UHFFFAOYSA-N 2-Oxohexane Chemical compound CCCCC(C)=O QQZOPKMRPOGIEB-UHFFFAOYSA-N 0.000 description 1
- KLGTXQOZQXTQPQ-UHFFFAOYSA-N 2-benzyl-1,2-oxazolidin-3-one Chemical class O=C1CCON1CC1=CC=CC=C1 KLGTXQOZQXTQPQ-UHFFFAOYSA-N 0.000 description 1
- 125000005999 2-bromoethyl group Chemical group 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- 125000000069 2-butynyl group Chemical group [H]C([H])([H])C#CC([H])([H])* 0.000 description 1
- 125000004780 2-chloro-2,2-difluoroethyl group Chemical group [H]C([H])(*)C(F)(F)Cl 0.000 description 1
- 125000004794 2-chloro-2-fluoroethoxy group Chemical group ClC(CO*)F 0.000 description 1
- 125000004779 2-chloro-2-fluoroethyl group Chemical group [H]C([H])(*)C([H])(F)Cl 0.000 description 1
- VONWPEXRCLHKRJ-UHFFFAOYSA-N 2-chloro-n-phenylacetamide Chemical class ClCC(=O)NC1=CC=CC=C1 VONWPEXRCLHKRJ-UHFFFAOYSA-N 0.000 description 1
- 125000001340 2-chloroethyl group Chemical group [H]C([H])(Cl)C([H])([H])* 0.000 description 1
- KKZUMAMOMRDVKA-UHFFFAOYSA-N 2-chloropropane Chemical group [CH2]C(C)Cl KKZUMAMOMRDVKA-UHFFFAOYSA-N 0.000 description 1
- WBIQQQGBSDOWNP-UHFFFAOYSA-N 2-dodecylbenzenesulfonic acid Chemical compound CCCCCCCCCCCCC1=CC=CC=C1S(O)(=O)=O WBIQQQGBSDOWNP-UHFFFAOYSA-N 0.000 description 1
- 125000006076 2-ethyl-1-butenyl group Chemical group 0.000 description 1
- 125000006077 2-ethyl-2-butenyl group Chemical group 0.000 description 1
- 125000006078 2-ethyl-3-butenyl group Chemical group 0.000 description 1
- 125000006176 2-ethylbutyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(C([H])([H])*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004883 2-ethylbutylcarbonyl group Chemical group C(C)C(CC(=O)*)CC 0.000 description 1
- 125000004777 2-fluoroethyl group Chemical group [H]C([H])(F)C([H])([H])* 0.000 description 1
- 125000002941 2-furyl group Chemical group O1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000006040 2-hexenyl group Chemical group 0.000 description 1
- 125000006026 2-methyl-1-butenyl group Chemical group 0.000 description 1
- 125000006045 2-methyl-1-pentenyl group Chemical group 0.000 description 1
- 125000006020 2-methyl-1-propenyl group Chemical group 0.000 description 1
- 125000006029 2-methyl-2-butenyl group Chemical group 0.000 description 1
- 125000006049 2-methyl-2-pentenyl group Chemical group 0.000 description 1
- 125000006022 2-methyl-2-propenyl group Chemical group 0.000 description 1
- 125000006031 2-methyl-3-butenyl group Chemical group 0.000 description 1
- 125000006053 2-methyl-3-pentenyl group Chemical group 0.000 description 1
- 125000006056 2-methyl-4-pentenyl group Chemical group 0.000 description 1
- GPZXFICWCMCQPF-UHFFFAOYSA-N 2-methylbenzoyl chloride Chemical compound CC1=CC=CC=C1C(Cl)=O GPZXFICWCMCQPF-UHFFFAOYSA-N 0.000 description 1
- 125000004493 2-methylbut-1-yl group Chemical group CC(C*)CC 0.000 description 1
- 125000004680 2-methylbutylcarbonyl group Chemical group CC(CC(=O)*)CC 0.000 description 1
- 125000005916 2-methylpentyl group Chemical group 0.000 description 1
- 125000004873 2-methylpentylcarbonyl group Chemical group CC(CC(=O)*)CCC 0.000 description 1
- 125000006024 2-pentenyl group Chemical group 0.000 description 1
- QBGQIMOGHUXVKB-UHFFFAOYSA-N 2-phenyl-4,5,6,7-tetrahydroisoindole-1,3-dione Chemical class O=C1C(CCCC2)=C2C(=O)N1C1=CC=CC=C1 QBGQIMOGHUXVKB-UHFFFAOYSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 1
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 125000000389 2-pyrrolyl group Chemical group [H]N1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000000175 2-thienyl group Chemical group S1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- 125000004326 2H-pyran-2-yl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])(*)O1 0.000 description 1
- REEXLQXWNOSJKO-UHFFFAOYSA-N 2h-1$l^{4},2,3-benzothiadiazine 1-oxide Chemical class C1=CC=C2S(=O)NN=CC2=C1 REEXLQXWNOSJKO-UHFFFAOYSA-N 0.000 description 1
- JSIAIROWMJGMQZ-UHFFFAOYSA-N 2h-triazol-4-amine Chemical class NC1=CNN=N1 JSIAIROWMJGMQZ-UHFFFAOYSA-N 0.000 description 1
- 125000006071 3,3-dimethyl-1-butenyl group Chemical group 0.000 description 1
- 125000006072 3,3-dimethyl-2-butenyl group Chemical group 0.000 description 1
- 125000004881 3,3-dimethylbutylcarbonyl group Chemical group CC(CCC(=O)*)(C)C 0.000 description 1
- FOGYNLXERPKEGN-UHFFFAOYSA-N 3-(2-hydroxy-3-methoxyphenyl)-2-[2-methoxy-4-(3-sulfopropyl)phenoxy]propane-1-sulfonic acid Chemical compound COC1=CC=CC(CC(CS(O)(=O)=O)OC=2C(=CC(CCCS(O)(=O)=O)=CC=2)OC)=C1O FOGYNLXERPKEGN-UHFFFAOYSA-N 0.000 description 1
- 125000004975 3-butenyl group Chemical group C(CC=C)* 0.000 description 1
- 125000000474 3-butynyl group Chemical group [H]C#CC([H])([H])C([H])([H])* 0.000 description 1
- 125000003682 3-furyl group Chemical group O1C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 125000006041 3-hexenyl group Chemical group 0.000 description 1
- 125000006027 3-methyl-1-butenyl group Chemical group 0.000 description 1
- 125000006046 3-methyl-1-pentenyl group Chemical group 0.000 description 1
- 125000006050 3-methyl-2-pentenyl group Chemical group 0.000 description 1
- 125000006032 3-methyl-3-butenyl group Chemical group 0.000 description 1
- 125000006054 3-methyl-3-pentenyl group Chemical group 0.000 description 1
- 125000006057 3-methyl-4-pentenyl group Chemical group 0.000 description 1
- 125000003542 3-methylbutan-2-yl group Chemical group [H]C([H])([H])C([H])(*)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000004681 3-methylbutylcarbonyl group Chemical group CC(CCC(=O)*)C 0.000 description 1
- 125000005917 3-methylpentyl group Chemical group 0.000 description 1
- 125000004874 3-methylpentylcarbonyl group Chemical group CC(CCC(=O)*)CC 0.000 description 1
- 125000003349 3-pyridyl group Chemical group N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000001397 3-pyrrolyl group Chemical group [H]N1C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 125000001541 3-thienyl group Chemical group S1C([H])=C([*])C([H])=C1[H] 0.000 description 1
- JUHPDXOIGLHXTC-UHFFFAOYSA-N 4-fluoro-2-(trifluoromethyl)benzoic acid Chemical compound OC(=O)C1=CC=C(F)C=C1C(F)(F)F JUHPDXOIGLHXTC-UHFFFAOYSA-N 0.000 description 1
- OEYHURRIOWWRMD-UHFFFAOYSA-N 4-fluoro-2-(trifluoromethyl)benzoyl chloride Chemical compound FC1=CC=C(C(Cl)=O)C(C(F)(F)F)=C1 OEYHURRIOWWRMD-UHFFFAOYSA-N 0.000 description 1
- 125000006042 4-hexenyl group Chemical group 0.000 description 1
- 125000006047 4-methyl-1-pentenyl group Chemical group 0.000 description 1
- 125000006051 4-methyl-2-pentenyl group Chemical group 0.000 description 1
- 125000003119 4-methyl-3-pentenyl group Chemical group [H]\C(=C(/C([H])([H])[H])C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000006058 4-methyl-4-pentenyl group Chemical group 0.000 description 1
- 125000004875 4-methylpentylcarbonyl group Chemical group CC(CCCC(=O)*)C 0.000 description 1
- 125000000339 4-pyridyl group Chemical group N1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 125000004315 4H-pyran-2-yl group Chemical group [H]C1=C([H])C([H])([H])C([H])=C(*)O1 0.000 description 1
- 125000006043 5-hexenyl group Chemical group 0.000 description 1
- 125000004070 6 membered heterocyclic group Chemical group 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- 240000000321 Abutilon grandifolium Species 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 235000011624 Agave sisalana Nutrition 0.000 description 1
- 244000198134 Agave sisalana Species 0.000 description 1
- 235000005255 Allium cepa Nutrition 0.000 description 1
- 244000291564 Allium cepa Species 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 239000004254 Ammonium phosphate Substances 0.000 description 1
- 235000011446 Amygdalus persica Nutrition 0.000 description 1
- 244000099147 Ananas comosus Species 0.000 description 1
- 235000007119 Ananas comosus Nutrition 0.000 description 1
- 244000105624 Arachis hypogaea Species 0.000 description 1
- 235000010777 Arachis hypogaea Nutrition 0.000 description 1
- 244000003416 Asparagus officinalis Species 0.000 description 1
- 235000005340 Asparagus officinalis Nutrition 0.000 description 1
- 235000007320 Avena fatua Nutrition 0.000 description 1
- 244000075850 Avena orientalis Species 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 244000060924 Brassica campestris Species 0.000 description 1
- 235000005637 Brassica campestris Nutrition 0.000 description 1
- 235000011297 Brassica napobrassica Nutrition 0.000 description 1
- 244000178924 Brassica napobrassica Species 0.000 description 1
- 240000002791 Brassica napus Species 0.000 description 1
- 235000004977 Brassica sinapistrum Nutrition 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 1
- NWIJFBIDKOLVAQ-UHFFFAOYSA-N C1NN[S+]=C1 Chemical compound C1NN[S+]=C1 NWIJFBIDKOLVAQ-UHFFFAOYSA-N 0.000 description 1
- 125000003601 C2-C6 alkynyl group Chemical group 0.000 description 1
- 244000052707 Camellia sinensis Species 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 235000003255 Carthamus tinctorius Nutrition 0.000 description 1
- 244000020518 Carthamus tinctorius Species 0.000 description 1
- 244000068645 Carya illinoensis Species 0.000 description 1
- 235000009025 Carya illinoensis Nutrition 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 235000008733 Citrus aurantifolia Nutrition 0.000 description 1
- 235000005979 Citrus limon Nutrition 0.000 description 1
- 244000131522 Citrus pyriformis Species 0.000 description 1
- 235000009088 Citrus pyriformis Nutrition 0.000 description 1
- 235000005976 Citrus sinensis Nutrition 0.000 description 1
- 240000002319 Citrus sinensis Species 0.000 description 1
- 235000007460 Coffea arabica Nutrition 0.000 description 1
- 240000007154 Coffea arabica Species 0.000 description 1
- 241000228031 Coffea liberica Species 0.000 description 1
- 244000016593 Coffea robusta Species 0.000 description 1
- 235000002187 Coffea robusta Nutrition 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229920000742 Cotton Polymers 0.000 description 1
- 235000009849 Cucumis sativus Nutrition 0.000 description 1
- 240000008067 Cucumis sativus Species 0.000 description 1
- 244000052363 Cynodon dactylon Species 0.000 description 1
- NDUPDOJHUQKPAG-UHFFFAOYSA-N Dalapon Chemical compound CC(Cl)(Cl)C(O)=O NDUPDOJHUQKPAG-UHFFFAOYSA-N 0.000 description 1
- 244000000626 Daucus carota Species 0.000 description 1
- 235000002767 Daucus carota Nutrition 0.000 description 1
- 240000003133 Elaeis guineensis Species 0.000 description 1
- 235000001950 Elaeis guineensis Nutrition 0.000 description 1
- 241001289540 Fallopia convolvulus Species 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 1
- BDAGIHXWWSANSR-UHFFFAOYSA-M Formate Chemical compound [O-]C=O BDAGIHXWWSANSR-UHFFFAOYSA-M 0.000 description 1
- 235000016623 Fragaria vesca Nutrition 0.000 description 1
- 244000307700 Fragaria vesca Species 0.000 description 1
- 241000233866 Fungi Species 0.000 description 1
- 235000014751 Gossypium arboreum Nutrition 0.000 description 1
- 240000001814 Gossypium arboreum Species 0.000 description 1
- 240000000047 Gossypium barbadense Species 0.000 description 1
- 235000009429 Gossypium barbadense Nutrition 0.000 description 1
- 235000004341 Gossypium herbaceum Nutrition 0.000 description 1
- 240000002024 Gossypium herbaceum Species 0.000 description 1
- 235000009432 Gossypium hirsutum Nutrition 0.000 description 1
- 235000003222 Helianthus annuus Nutrition 0.000 description 1
- 244000020551 Helianthus annuus Species 0.000 description 1
- 244000043261 Hevea brasiliensis Species 0.000 description 1
- 241000209219 Hordeum Species 0.000 description 1
- 235000008694 Humulus lupulus Nutrition 0.000 description 1
- 244000025221 Humulus lupulus Species 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 244000017020 Ipomoea batatas Species 0.000 description 1
- 235000002678 Ipomoea batatas Nutrition 0.000 description 1
- 235000009496 Juglans regia Nutrition 0.000 description 1
- 240000007049 Juglans regia Species 0.000 description 1
- 239000005909 Kieselgur Substances 0.000 description 1
- 240000004322 Lens culinaris Species 0.000 description 1
- 235000010666 Lens esculenta Nutrition 0.000 description 1
- 235000019738 Limestone Nutrition 0.000 description 1
- 235000003403 Limnocharis flava Nutrition 0.000 description 1
- 235000004431 Linum usitatissimum Nutrition 0.000 description 1
- 240000006240 Linum usitatissimum Species 0.000 description 1
- 235000007688 Lycopersicon esculentum Nutrition 0.000 description 1
- 208000002720 Malnutrition Diseases 0.000 description 1
- 241000220225 Malus Species 0.000 description 1
- 240000003183 Manihot esculenta Species 0.000 description 1
- 235000004456 Manihot esculenta Nutrition 0.000 description 1
- 235000010624 Medicago sativa Nutrition 0.000 description 1
- 240000004658 Medicago sativa Species 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 241000234295 Musa Species 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- 241000208134 Nicotiana rustica Species 0.000 description 1
- 235000002637 Nicotiana tabacum Nutrition 0.000 description 1
- 244000061176 Nicotiana tabacum Species 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- IGFHQQFPSIBGKE-UHFFFAOYSA-N Nonylphenol Natural products CCCCCCCCCC1=CC=C(O)C=C1 IGFHQQFPSIBGKE-UHFFFAOYSA-N 0.000 description 1
- JZTPOMIFAFKKSK-UHFFFAOYSA-N O-phosphonohydroxylamine Chemical compound NOP(O)(O)=O JZTPOMIFAFKKSK-UHFFFAOYSA-N 0.000 description 1
- 235000002725 Olea europaea Nutrition 0.000 description 1
- 240000007817 Olea europaea Species 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 235000010627 Phaseolus vulgaris Nutrition 0.000 description 1
- 244000046052 Phaseolus vulgaris Species 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-L Phosphate ion(2-) Chemical compound OP([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-L 0.000 description 1
- 244000193463 Picea excelsa Species 0.000 description 1
- 235000008124 Picea excelsa Nutrition 0.000 description 1
- 235000005205 Pinus Nutrition 0.000 description 1
- 241000218602 Pinus <genus> Species 0.000 description 1
- 235000010582 Pisum sativum Nutrition 0.000 description 1
- 240000004713 Pisum sativum Species 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 244000007021 Prunus avium Species 0.000 description 1
- 235000010401 Prunus avium Nutrition 0.000 description 1
- 240000005809 Prunus persica Species 0.000 description 1
- 235000014443 Pyrus communis Nutrition 0.000 description 1
- 240000001987 Pyrus communis Species 0.000 description 1
- 244000281247 Ribes rubrum Species 0.000 description 1
- 235000016911 Ribes sativum Nutrition 0.000 description 1
- 240000000528 Ricinus communis Species 0.000 description 1
- 235000004443 Ricinus communis Nutrition 0.000 description 1
- 235000007201 Saccharum officinarum Nutrition 0.000 description 1
- 240000000111 Saccharum officinarum Species 0.000 description 1
- 235000010086 Setaria viridis var. viridis Nutrition 0.000 description 1
- 240000003768 Solanum lycopersicum Species 0.000 description 1
- 235000002595 Solanum tuberosum Nutrition 0.000 description 1
- 244000061456 Solanum tuberosum Species 0.000 description 1
- 235000007230 Sorghum bicolor Nutrition 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 229940100389 Sulfonylurea Drugs 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 1
- 235000006468 Thea sinensis Nutrition 0.000 description 1
- 244000299461 Theobroma cacao Species 0.000 description 1
- 235000009470 Theobroma cacao Nutrition 0.000 description 1
- 235000011941 Tilia x europaea Nutrition 0.000 description 1
- 206010044278 Trace element deficiency Diseases 0.000 description 1
- 235000015724 Trifolium pratense Nutrition 0.000 description 1
- 240000002913 Trifolium pratense Species 0.000 description 1
- 241000209140 Triticum Species 0.000 description 1
- 229920001807 Urea-formaldehyde Polymers 0.000 description 1
- 235000005373 Uvularia sessilifolia Nutrition 0.000 description 1
- 235000010749 Vicia faba Nutrition 0.000 description 1
- 240000006677 Vicia faba Species 0.000 description 1
- 235000014787 Vitis vinifera Nutrition 0.000 description 1
- 240000006365 Vitis vinifera Species 0.000 description 1
- 241000607479 Yersinia pestis Species 0.000 description 1
- 235000007244 Zea mays Nutrition 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- 241001148683 Zostera marina Species 0.000 description 1
- 241000235033 Zygosaccharomyces rouxii Species 0.000 description 1
- OMPDKLBVTGFBFW-UHFFFAOYSA-N [N+](=[N-])=C1C(=CC=CC1=O)C(C(=O)O)C Chemical class [N+](=[N-])=C1C(=CC=CC1=O)C(C(=O)O)C OMPDKLBVTGFBFW-UHFFFAOYSA-N 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 125000005089 alkenylaminocarbonyl group Chemical group 0.000 description 1
- 125000005090 alkenylcarbonyl group Chemical group 0.000 description 1
- 125000004183 alkoxy alkyl group Chemical group 0.000 description 1
- 125000004694 alkoxyaminocarbonyl group Chemical group 0.000 description 1
- 125000004666 alkoxyiminoalkyl group Chemical group 0.000 description 1
- 150000008055 alkyl aryl sulfonates Chemical class 0.000 description 1
- 125000005093 alkyl carbonyl alkyl group Chemical group 0.000 description 1
- 125000005196 alkyl carbonyloxy group Chemical group 0.000 description 1
- 125000005037 alkyl phenyl group Chemical group 0.000 description 1
- 125000005095 alkynylaminocarbonyl group Chemical group 0.000 description 1
- 125000005087 alkynylcarbonyl group Chemical group 0.000 description 1
- 125000005225 alkynyloxycarbonyl group Chemical group 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 235000001014 amino acid Nutrition 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 238000006399 aminohydroxylation reaction Methods 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 229910000148 ammonium phosphate Inorganic materials 0.000 description 1
- 235000019289 ammonium phosphates Nutrition 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 1
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 1
- 235000011130 ammonium sulphate Nutrition 0.000 description 1
- 229940051881 anilide analgesics and antipyretics Drugs 0.000 description 1
- 150000003931 anilides Chemical class 0.000 description 1
- 239000008346 aqueous phase Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 125000003435 aroyl group Chemical group 0.000 description 1
- 150000003935 benzaldehydes Chemical class 0.000 description 1
- YOUGRGFIHBUKRS-UHFFFAOYSA-N benzyl(trimethyl)azanium Chemical compound C[N+](C)(C)CC1=CC=CC=C1 YOUGRGFIHBUKRS-UHFFFAOYSA-N 0.000 description 1
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 1
- UORVGPXVDQYIDP-BJUDXGSMSA-N borane Chemical compound [10BH3] UORVGPXVDQYIDP-BJUDXGSMSA-N 0.000 description 1
- 229910000085 borane Inorganic materials 0.000 description 1
- 238000009395 breeding Methods 0.000 description 1
- 230000001488 breeding effect Effects 0.000 description 1
- 125000005997 bromomethyl group Chemical group 0.000 description 1
- 125000006251 butylcarbonyl group Chemical group 0.000 description 1
- 125000004744 butyloxycarbonyl group Chemical group 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Inorganic materials [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 1
- SMLHHBQGIJMAIM-UHFFFAOYSA-N calcium;2-dodecylbenzenesulfonic acid Chemical compound [Ca].CCCCCCCCCCCCC1=CC=CC=C1S(O)(=O)=O SMLHHBQGIJMAIM-UHFFFAOYSA-N 0.000 description 1
- 150000004657 carbamic acid derivatives Chemical class 0.000 description 1
- 125000001951 carbamoylamino group Chemical group C(N)(=O)N* 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 235000013339 cereals Nutrition 0.000 description 1
- 125000004775 chlorodifluoromethyl group Chemical group FC(F)(Cl)* 0.000 description 1
- 125000004773 chlorofluoromethyl group Chemical group [H]C(F)(Cl)* 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- 150000001851 cinnamic acid derivatives Chemical class 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 239000011280 coal tar Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- JEVCWSUVFOYBFI-UHFFFAOYSA-N cyanyl Chemical compound N#[C] JEVCWSUVFOYBFI-UHFFFAOYSA-N 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 238000006114 decarboxylation reaction Methods 0.000 description 1
- 238000010511 deprotection reaction Methods 0.000 description 1
- 125000004663 dialkyl amino group Chemical group 0.000 description 1
- 125000004473 dialkylaminocarbonyl group Chemical group 0.000 description 1
- 125000004671 dialkylaminothiocarbonyl group Chemical group 0.000 description 1
- MNNHAPBLZZVQHP-UHFFFAOYSA-N diammonium hydrogen phosphate Chemical compound [NH4+].[NH4+].OP([O-])([O-])=O MNNHAPBLZZVQHP-UHFFFAOYSA-N 0.000 description 1
- 150000004891 diazines Chemical class 0.000 description 1
- 125000004774 dichlorofluoromethyl group Chemical group FC(Cl)(Cl)* 0.000 description 1
- 125000004772 dichloromethyl group Chemical group [H]C(Cl)(Cl)* 0.000 description 1
- 239000002283 diesel fuel Substances 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-M dihydrogenphosphate Chemical compound OP(O)([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-M 0.000 description 1
- UAOMVDZJSHZZME-UHFFFAOYSA-N diisopropylamine Chemical compound CC(C)NC(C)C UAOMVDZJSHZZME-UHFFFAOYSA-N 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical class C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 229940060296 dodecylbenzenesulfonic acid Drugs 0.000 description 1
- 239000010459 dolomite Substances 0.000 description 1
- 229910000514 dolomite Inorganic materials 0.000 description 1
- 239000000428 dust Substances 0.000 description 1
- 238000010410 dusting Methods 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 238000010931 ester hydrolysis Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 229940052303 ethers for general anesthesia Drugs 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- 125000005745 ethoxymethyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])* 0.000 description 1
- JHQQRKVAVMTPHC-UHFFFAOYSA-N ethyl 2-[[4-fluoro-2-(trifluoromethyl)benzoyl]amino]-3-(2-methylphenyl)-3-oxopropanoate Chemical compound C=1C=CC=C(C)C=1C(=O)C(C(=O)OCC)NC(=O)C1=CC=C(F)C=C1C(F)(F)F JHQQRKVAVMTPHC-UHFFFAOYSA-N 0.000 description 1
- FPULFENIJDPZBX-UHFFFAOYSA-N ethyl 2-isocyanoacetate Chemical compound CCOC(=O)C[N+]#[C-] FPULFENIJDPZBX-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 125000006260 ethylaminocarbonyl group Chemical group [H]N(C(*)=O)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004672 ethylcarbonyl group Chemical group [H]C([H])([H])C([H])([H])C(*)=O 0.000 description 1
- LYCAIKOWRPUZTN-UHFFFAOYSA-N ethylene glycol Natural products OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 150000002191 fatty alcohols Chemical class 0.000 description 1
- 239000003337 fertilizer Substances 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 235000004426 flaxseed Nutrition 0.000 description 1
- 125000004785 fluoromethoxy group Chemical group [H]C([H])(F)O* 0.000 description 1
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 238000010353 genetic engineering Methods 0.000 description 1
- 235000002532 grape seed extract Nutrition 0.000 description 1
- 244000230342 green foxtail Species 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 125000000232 haloalkynyl group Chemical group 0.000 description 1
- 150000003977 halocarboxylic acids Chemical class 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 125000006343 heptafluoro propyl group Chemical group 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- 150000002391 heterocyclic compounds Chemical class 0.000 description 1
- 125000004415 heterocyclylalkyl group Chemical group 0.000 description 1
- 150000004761 hexafluorosilicates Chemical class 0.000 description 1
- 125000004871 hexylcarbonyl group Chemical group C(CCCCC)C(=O)* 0.000 description 1
- 239000003864 humus Substances 0.000 description 1
- YPGCWEMNNLXISK-UHFFFAOYSA-N hydratropic acid Chemical compound OC(=O)C(C)C1=CC=CC=C1 YPGCWEMNNLXISK-UHFFFAOYSA-N 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- 238000007327 hydrogenolysis reaction Methods 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 125000002962 imidazol-1-yl group Chemical group [*]N1C([H])=NC([H])=C1[H] 0.000 description 1
- 125000003037 imidazol-2-yl group Chemical group [H]N1C([*])=NC([H])=C1[H] 0.000 description 1
- 125000002140 imidazol-4-yl group Chemical group [H]N1C([H])=NC([*])=C1[H] 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- 150000002461 imidazolidines Chemical class 0.000 description 1
- 150000008624 imidazolidinones Chemical class 0.000 description 1
- 238000005470 impregnation Methods 0.000 description 1
- 239000000543 intermediate Substances 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001793 isothiazol-3-yl group Chemical group [H]C1=C([H])C(*)=NS1 0.000 description 1
- 125000004500 isothiazol-4-yl group Chemical group S1N=CC(=C1)* 0.000 description 1
- 125000004501 isothiazol-5-yl group Chemical group S1N=CC=C1* 0.000 description 1
- 125000004284 isoxazol-3-yl group Chemical group [H]C1=C([H])C(*)=NO1 0.000 description 1
- 125000004499 isoxazol-5-yl group Chemical group O1N=CC=C1* 0.000 description 1
- 239000003350 kerosene Substances 0.000 description 1
- 229920005610 lignin Polymers 0.000 description 1
- 239000004571 lime Substances 0.000 description 1
- 239000006028 limestone Substances 0.000 description 1
- 239000012280 lithium aluminium hydride Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 125000004458 methylaminocarbonyl group Chemical group [H]N(C(*)=O)C([H])([H])[H] 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000019713 millet Nutrition 0.000 description 1
- 125000002950 monocyclic group Chemical group 0.000 description 1
- PSHKMPUSSFXUIA-UHFFFAOYSA-N n,n-dimethylpyridin-2-amine Chemical compound CN(C)C1=CC=CC=N1 PSHKMPUSSFXUIA-UHFFFAOYSA-N 0.000 description 1
- MIYKHJXFICMPOJ-UHFFFAOYSA-N n-benzyl-1-phenylmethanimine Chemical compound C=1C=CC=CC=1CN=CC1=CC=CC=C1 MIYKHJXFICMPOJ-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- WNPVAXLJVUXYFU-UHFFFAOYSA-N n-cyclohex-2-en-1-ylidenehydroxylamine Chemical compound ON=C1CCCC=C1 WNPVAXLJVUXYFU-UHFFFAOYSA-N 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- CPGWSLFYXMRNDV-UHFFFAOYSA-N n-methyl-n-phenylcarbamoyl chloride Chemical compound ClC(=O)N(C)C1=CC=CC=C1 CPGWSLFYXMRNDV-UHFFFAOYSA-N 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- CWYZDPHNAGSFQB-UHFFFAOYSA-N n-propylbutan-1-amine Chemical class CCCCNCCC CWYZDPHNAGSFQB-UHFFFAOYSA-N 0.000 description 1
- PSZYNBSKGUBXEH-UHFFFAOYSA-N naphthalene-1-sulfonic acid Chemical class C1=CC=C2C(S(=O)(=O)O)=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-N 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005246 nonafluorobutyl group Chemical group FC(F)(F)C(F)(F)C(F)(F)C(F)(F)* 0.000 description 1
- 239000012875 nonionic emulsifier Substances 0.000 description 1
- 231100001184 nonphytotoxic Toxicity 0.000 description 1
- SNQQPOLDUKLAAF-UHFFFAOYSA-N nonylphenol Chemical compound CCCCCCCCCC1=CC=CC=C1O SNQQPOLDUKLAAF-UHFFFAOYSA-N 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 239000012038 nucleophile Substances 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- WOFPPJOZXUTRAU-UHFFFAOYSA-N octan-4-ol Chemical compound CCCCC(O)CCC WOFPPJOZXUTRAU-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 150000004866 oxadiazoles Chemical class 0.000 description 1
- 125000004287 oxazol-2-yl group Chemical group [H]C1=C([H])N=C(*)O1 0.000 description 1
- 125000004304 oxazol-5-yl group Chemical group O1C=NC=C1* 0.000 description 1
- 150000002918 oxazolines Chemical class 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 150000002924 oxiranes Chemical class 0.000 description 1
- 125000006340 pentafluoro ethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 1
- 125000003538 pentan-3-yl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004675 pentylcarbonyl group Chemical group C(CCCC)C(=O)* 0.000 description 1
- 125000001148 pentyloxycarbonyl group Chemical group 0.000 description 1
- 150000002989 phenols Chemical class 0.000 description 1
- 229960003424 phenylacetic acid Drugs 0.000 description 1
- 239000003279 phenylacetic acid Substances 0.000 description 1
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 1
- 150000002993 phenylalanine derivatives Chemical class 0.000 description 1
- 125000003884 phenylalkyl group Chemical group 0.000 description 1
- LEVJVKGPFAQPOI-UHFFFAOYSA-N phenylmethanone Chemical class O=[C]C1=CC=CC=C1 LEVJVKGPFAQPOI-UHFFFAOYSA-N 0.000 description 1
- UYWQUFXKFGHYNT-UHFFFAOYSA-N phenylmethyl ester of formic acid Natural products O=COCC1=CC=CC=C1 UYWQUFXKFGHYNT-UHFFFAOYSA-N 0.000 description 1
- XMGMFRIEKMMMSU-UHFFFAOYSA-N phenylmethylbenzene Chemical group C=1C=CC=CC=1[C]C1=CC=CC=C1 XMGMFRIEKMMMSU-UHFFFAOYSA-N 0.000 description 1
- 150000008048 phenylpyrazoles Chemical class 0.000 description 1
- 230000003032 phytopathogenic effect Effects 0.000 description 1
- SIOXPEMLGUPBBT-UHFFFAOYSA-N picolinic acid Chemical compound OC(=O)C1=CC=CC=N1 SIOXPEMLGUPBBT-UHFFFAOYSA-N 0.000 description 1
- 125000000587 piperidin-1-yl group Chemical group [H]C1([H])N(*)C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000004574 piperidin-2-yl group Chemical group N1C(CCCC1)* 0.000 description 1
- 125000004483 piperidin-3-yl group Chemical group N1CC(CCC1)* 0.000 description 1
- 125000004482 piperidin-4-yl group Chemical group N1CCC(CC1)* 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 229920000151 polyglycol Polymers 0.000 description 1
- 239000010695 polyglycol Substances 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- VCFOFCPAHBIEHH-UHFFFAOYSA-M potassium;2-methylbutanoate Chemical compound [K+].CCC(C)C([O-])=O VCFOFCPAHBIEHH-UHFFFAOYSA-M 0.000 description 1
- LBOHISOWGKIIKX-UHFFFAOYSA-M potassium;2-methylpropanoate Chemical compound [K+].CC(C)C([O-])=O LBOHISOWGKIIKX-UHFFFAOYSA-M 0.000 description 1
- 125000001844 prenyl group Chemical group [H]C([*])([H])C([H])=C(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005767 propoxymethyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])[#8]C([H])([H])* 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004673 propylcarbonyl group Chemical group 0.000 description 1
- 125000004742 propyloxycarbonyl group Chemical group 0.000 description 1
- 125000004353 pyrazol-1-yl group Chemical group [H]C1=NN(*)C([H])=C1[H] 0.000 description 1
- 125000004289 pyrazol-3-yl group Chemical group [H]N1N=C(*)C([H])=C1[H] 0.000 description 1
- 150000003217 pyrazoles Chemical class 0.000 description 1
- 125000002206 pyridazin-3-yl group Chemical group [H]C1=C([H])C([H])=C(*)N=N1 0.000 description 1
- 125000004940 pyridazin-4-yl group Chemical group N1=NC=C(C=C1)* 0.000 description 1
- 125000000246 pyrimidin-2-yl group Chemical group [H]C1=NC(*)=NC([H])=C1[H] 0.000 description 1
- 125000004527 pyrimidin-4-yl group Chemical group N1=CN=C(C=C1)* 0.000 description 1
- 125000004528 pyrimidin-5-yl group Chemical group N1=CN=CC(=C1)* 0.000 description 1
- LOAUVZALPPNFOQ-UHFFFAOYSA-N quinaldic acid Chemical compound C1=CC=CC2=NC(C(=O)O)=CC=C21 LOAUVZALPPNFOQ-UHFFFAOYSA-N 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 235000013526 red clover Nutrition 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 239000012266 salt solution Substances 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000003548 sec-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 125000005624 silicic acid group Chemical class 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 239000012279 sodium borohydride Substances 0.000 description 1
- 229910000033 sodium borohydride Inorganic materials 0.000 description 1
- BEOOHQFXGBMRKU-UHFFFAOYSA-N sodium cyanoborohydride Chemical compound [Na+].[B-]C#N BEOOHQFXGBMRKU-UHFFFAOYSA-N 0.000 description 1
- 238000009331 sowing Methods 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 229940124530 sulfonamide Drugs 0.000 description 1
- 150000003456 sulfonamides Chemical class 0.000 description 1
- 125000005537 sulfoxonium group Chemical group 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- DZLFLBLQUQXARW-UHFFFAOYSA-N tetrabutylammonium Chemical compound CCCC[N+](CCCC)(CCCC)CCCC DZLFLBLQUQXARW-UHFFFAOYSA-N 0.000 description 1
- 125000004192 tetrahydrofuran-2-yl group Chemical group [H]C1([H])OC([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000004187 tetrahydropyran-2-yl group Chemical group [H]C1([H])OC([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- CXWXQJXEFPUFDZ-UHFFFAOYSA-N tetralin Chemical compound C1=CC=C2CCCCC2=C1 CXWXQJXEFPUFDZ-UHFFFAOYSA-N 0.000 description 1
- QEMXHQIAXOOASZ-UHFFFAOYSA-N tetramethylammonium Chemical compound C[N+](C)(C)C QEMXHQIAXOOASZ-UHFFFAOYSA-N 0.000 description 1
- 125000004523 tetrazol-1-yl group Chemical group N1(N=NN=C1)* 0.000 description 1
- 125000004299 tetrazol-5-yl group Chemical group [H]N1N=NC(*)=N1 0.000 description 1
- 125000000437 thiazol-2-yl group Chemical group [H]C1=C([H])N=C(*)S1 0.000 description 1
- 125000004495 thiazol-4-yl group Chemical group S1C=NC(=C1)* 0.000 description 1
- 125000004496 thiazol-5-yl group Chemical group S1C=NC=C1* 0.000 description 1
- 150000003548 thiazolidines Chemical class 0.000 description 1
- 150000003573 thiols Chemical class 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- 150000003624 transition metals Chemical class 0.000 description 1
- 150000003918 triazines Chemical class 0.000 description 1
- 125000003866 trichloromethyl group Chemical group ClC(Cl)(Cl)* 0.000 description 1
- UORVGPXVDQYIDP-UHFFFAOYSA-N trihydridoboron Substances B UORVGPXVDQYIDP-UHFFFAOYSA-N 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 239000004562 water dispersible granule Substances 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 239000002023 wood Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/54—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C237/00—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups
- C07C237/28—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups having the carbon atom of at least one of the carboxamide groups bound to a carbon atom of a non-condensed six-membered aromatic ring of the carbon skeleton
- C07C237/42—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups having the carbon atom of at least one of the carboxamide groups bound to a carbon atom of a non-condensed six-membered aromatic ring of the carbon skeleton having nitrogen atoms of amino groups bound to the carbon skeleton of the acid part, further acylated
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/44—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing at least one carboxylic group or a thio analogue, or a derivative thereof, and a nitrogen atom attached to the same carbon skeleton by a single or double bond, this nitrogen atom not being a member of a derivative or of a thio analogue of a carboxylic group, e.g. amino-carboxylic acids
- A01N37/48—Nitro-carboxylic acids; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N41/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a sulfur atom bound to a hetero atom
- A01N41/02—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a sulfur atom bound to a hetero atom containing a sulfur-to-oxygen double bond
- A01N41/04—Sulfonic acids; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/02—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having no bond to a nitrogen atom
- A01N47/06—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having no bond to a nitrogen atom containing —O—CO—O— groups; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/10—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof
- A01N47/12—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof containing a —O—CO—N< group, or a thio analogue thereof, neither directly attached to a ring nor the nitrogen atom being a member of a heterocyclic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C233/00—Carboxylic acid amides
- C07C233/64—Carboxylic acid amides having carbon atoms of carboxamide groups bound to carbon atoms of six-membered aromatic rings
- C07C233/81—Carboxylic acid amides having carbon atoms of carboxamide groups bound to carbon atoms of six-membered aromatic rings having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by carboxyl groups
- C07C233/82—Carboxylic acid amides having carbon atoms of carboxamide groups bound to carbon atoms of six-membered aromatic rings having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by carboxyl groups with the substituted hydrocarbon radical bound to the nitrogen atom of the carboxamide group by an acyclic carbon atom
- C07C233/87—Carboxylic acid amides having carbon atoms of carboxamide groups bound to carbon atoms of six-membered aromatic rings having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by carboxyl groups with the substituted hydrocarbon radical bound to the nitrogen atom of the carboxamide group by an acyclic carbon atom of a carbon skeleton containing six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C237/00—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups
- C07C237/02—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups having the carbon atoms of the carboxamide groups bound to acyclic carbon atoms of the carbon skeleton
- C07C237/22—Carboxylic acid amides, the carbon skeleton of the acid part being further substituted by amino groups having the carbon atoms of the carboxamide groups bound to acyclic carbon atoms of the carbon skeleton having nitrogen atoms of amino groups bound to the carbon skeleton of the acid part, further acylated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C255/00—Carboxylic acid nitriles
- C07C255/49—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C255/58—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton containing cyano groups and singly-bound nitrogen atoms, not being further bound to other hetero atoms, bound to the carbon skeleton
- C07C255/60—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton containing cyano groups and singly-bound nitrogen atoms, not being further bound to other hetero atoms, bound to the carbon skeleton at least one of the singly-bound nitrogen atoms being acylated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C271/00—Derivatives of carbamic acids, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups
- C07C271/06—Esters of carbamic acids
- C07C271/08—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms
- C07C271/10—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atoms of the carbamate groups bound to hydrogen atoms or to acyclic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C271/00—Derivatives of carbamic acids, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups
- C07C271/06—Esters of carbamic acids
- C07C271/08—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms
- C07C271/10—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atoms of the carbamate groups bound to hydrogen atoms or to acyclic carbon atoms
- C07C271/12—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atoms of the carbamate groups bound to hydrogen atoms or to acyclic carbon atoms to hydrogen atoms or to carbon atoms of unsubstituted hydrocarbon radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C271/00—Derivatives of carbamic acids, i.e. compounds containing any of the groups, the nitrogen atom not being part of nitro or nitroso groups
- C07C271/06—Esters of carbamic acids
- C07C271/08—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms
- C07C271/26—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atom of at least one of the carbamate groups bound to a carbon atom of a six-membered aromatic ring
- C07C271/28—Esters of carbamic acids having oxygen atoms of carbamate groups bound to acyclic carbon atoms with the nitrogen atom of at least one of the carbamate groups bound to a carbon atom of a six-membered aromatic ring to a carbon atom of a non-condensed six-membered aromatic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C275/00—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups
- C07C275/04—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of urea groups bound to acyclic carbon atoms
- C07C275/20—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of urea groups bound to acyclic carbon atoms of an unsaturated carbon skeleton
- C07C275/24—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of urea groups bound to acyclic carbon atoms of an unsaturated carbon skeleton containing six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C275/00—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups
- C07C275/28—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of urea groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C275/42—Derivatives of urea, i.e. compounds containing any of the groups, the nitrogen atoms not being part of nitro or nitroso groups having nitrogen atoms of urea groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton being further substituted by carboxyl groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C309/00—Sulfonic acids; Halides, esters, or anhydrides thereof
- C07C309/63—Esters of sulfonic acids
- C07C309/64—Esters of sulfonic acids having sulfur atoms of esterified sulfo groups bound to acyclic carbon atoms
- C07C309/65—Esters of sulfonic acids having sulfur atoms of esterified sulfo groups bound to acyclic carbon atoms of a saturated carbon skeleton
- C07C309/66—Methanesulfonates
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/01—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms
- C07C311/02—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton
- C07C311/03—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton having the nitrogen atoms of the sulfonamide groups bound to hydrogen atoms or to acyclic carbon atoms
- C07C311/06—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton having the nitrogen atoms of the sulfonamide groups bound to hydrogen atoms or to acyclic carbon atoms to acyclic carbon atoms of hydrocarbon radicals substituted by carboxyl groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/01—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms
- C07C311/02—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton
- C07C311/08—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton having the nitrogen atom of at least one of the sulfonamide groups bound to a carbon atom of a six-membered aromatic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/01—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms
- C07C311/02—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton
- C07C311/09—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton the carbon skeleton being further substituted by at least two halogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/50—Compounds containing any of the groups, X being a hetero atom, Y being any atom
- C07C311/52—Y being a hetero atom
- C07C311/53—X and Y not being nitrogen atoms, e.g. N-sulfonylcarbamic acid
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/50—Compounds containing any of the groups, X being a hetero atom, Y being any atom
- C07C311/52—Y being a hetero atom
- C07C311/54—Y being a hetero atom either X or Y, but not both, being nitrogen atoms, e.g. N-sulfonylurea
- C07C311/55—Y being a hetero atom either X or Y, but not both, being nitrogen atoms, e.g. N-sulfonylurea having sulfur atoms of the sulfonylurea groups bound to acyclic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/50—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton
- C07C323/51—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atoms of the thio groups bound to acyclic carbon atoms of the carbon skeleton
- C07C323/52—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atoms of the thio groups bound to acyclic carbon atoms of the carbon skeleton the carbon skeleton being acyclic and saturated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/50—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton
- C07C323/51—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atoms of the thio groups bound to acyclic carbon atoms of the carbon skeleton
- C07C323/60—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atoms of the thio groups bound to acyclic carbon atoms of the carbon skeleton with the carbon atom of at least one of the carboxyl groups bound to nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/50—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton
- C07C323/62—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atom of at least one of the thio groups bound to a carbon atom of a six-membered aromatic ring of the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/50—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton
- C07C323/62—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atom of at least one of the thio groups bound to a carbon atom of a six-membered aromatic ring of the carbon skeleton
- C07C323/63—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atom of at least one of the thio groups bound to a carbon atom of a six-membered aromatic ring of the carbon skeleton the carbon skeleton being further substituted by nitrogen atoms, not being part of nitro or nitroso groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/78—Carbon atoms having three bonds to hetero atoms, with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D213/79—Acids; Esters
- C07D213/80—Acids; Esters in position 3
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/12—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/46—Two or more oxygen, sulphur or nitrogen atoms
- C07D239/60—Three or more oxygen or sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/08—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D309/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings
- C07D309/02—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
- C07D309/08—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings having no double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D317/00—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms
- C07D317/08—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms having the hetero atoms in positions 1 and 3
- C07D317/10—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms having the hetero atoms in positions 1 and 3 not condensed with other rings
- C07D317/32—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms having the hetero atoms in positions 1 and 3 not condensed with other rings with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/06—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to the ring carbon atoms
- C07D333/24—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic System
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/18—Compounds having one or more C—Si linkages as well as one or more C—O—Si linkages
- C07F7/1804—Compounds having Si-O-C linkages
Definitions
- the present invention relates to benzoyl-substituted phenylalanine amides of the formula
- R 1 is halogen, cyano, CRCE alkyl, CC 6 haloalkyl, C ⁇ -C 6 haloalkoxy, nitro, hydroxycarbonyl, C ⁇ -C6 alkoxycarbonyl, C ⁇ -C6 haloalkylthio, or phenyl;
- R 6 , R 7 is hydrogen, hydroxy or CC 6 alkoxy
- R 8 C ⁇ -C 6 -alkyl, -C 4 cyanoalkyl or C ⁇ -C 6 haloalkyl
- R 10 is hydrogen or C r C 6 alkyl
- R 11, R 12 is hydrogen, halogen, cyano, CRCE alkyl, CrC 6 -HalogenaIkyl, hydroxy, -C 6 alkoxy, -C 6 -haloalkoxy, hydroxy, nitro, hydroxy, C r C 4 alkyl, -C 6 alkoxy C 1 -C 4 -alkyl, tri (C 1 -C 6 -alkyl) silyloxyCrC 4 -alkyl, C 1 -C 4 -alkylthio, (hydroxycarbonyl) C 1 -C 6 -alkyl, (CC 6 -alkoxycarbonyl) C 1 -C 6 -alkyl, (hydroxycarbonyl) C 2 - C 6 -alkenyl, (C 1 -C 6 -alkoxycarbonyl) C 2 -C 6 -alkenyl, (hydroxycarbonyl) -CrC 4 -alkoxy, (C 1 -C 4 -
- R 13, R 14, R 15 is hydrogen, halogen, cyano, -C 6 alkyl, -C 6 -haloalkyl, -C 6 - alkoxy, CRCE haloalkoxy, hydroxy, nitro, -C 6 alkylthio or benzyl zyloxy;
- R 16 , R 17 , R 18 are hydrogen, C 1 -C 6 -alkyl, tri (C 1 -C 6 -alkyl) silyl, C 3 -C 6 -cycloalkyl, C 3 -C 6 -alkenyl, C 3 -C 6 -alkynyl, C 3 -C 6 haloalkenyl, C 3 -C 6 haloalkynyl, formyl, -C 6 alkylcarbonyl, C 3 -C 6 cycloalkylcarbonyl, C 2 -C 6 - alkenylcarbonyl, C 2 -C 6 alkynylcarbonyl, -C 6 alkoxycarbonyl, C 3 -C 6 - alkenyloxycarbonyl, C 3 -C 6 -AlkinyIoxycarbonyl, -C 6 alkylamino carbonyl, C 3 -Ce-alkenylaminocarbonyl, C3-
- R 19 is hydrogen, d-drAlkyl, QrCe cycloalkyl, C 3 -C 6 alkenyl, C 3 -C 6 alkynyl, C 3 -C 6 - haloalkenyl, C 3 -C 6 haloalkynyl, wherein the said alkyl and Cycloalkyl radicals may be partially or completely halogenated and / or may carry one to three of the following groups: cyano, hydroxy, C 3 -C 6 -cycloalkyl, C 1 -C 4 -alkoxy, C 1 -C 4 -alkylthio, di- (C 4 -alkyl) -amino, dd-alkylcarbonyl, hydroxycarbonyl, C 1 -C 4 -alkoxycarbonyl, aminocarbonyl, C 1 -C 4 -alkylaminocarbonyl, di (C 4 -alkyl) aminocarbonyl or dd
- R 20 CRCE alkyl, dC 6 -haloalkyl or phenyl, where the phenyl radical may be partially or fully halogenated and / or may carry one to three of the following groups: -C 6 alkyl, d-Ce-halo-alkyl or C 6 - alkoxy; and their agriculturally useful salts.
- the invention relates to processes and intermediates for the preparation of compounds of the formula I, compositions containing them and the use of these derivatives or agents for controlling harmful plants containing them.
- Benzoyl substituted amino acid amides having pharmaceutical activity are described i.a. in WO 97/05865, GB 2369117, JP 10/298151 and JP 03/294253.
- herbicidal agents were found which contain the compounds I and have a very good herbicidal activity.
- methods for the preparation of these compositions and methods for controlling undesired plant growth with the compounds I have been found.
- the compounds of the formula I contain two or more chiral centers and are then present as enantiomer or diastereomer mixtures.
- the invention provides both the pure enantiomers or diastereomers and mixtures thereof.
- the compounds of the formula I can also be present in the form of their agriculturally useful salts, wherein the type of salt generally does not matter.
- the salts of those cations or the acid addition salts of those acids come into consideration whose cations, or anions, do not adversely affect the herbicidal activity of the compounds I.
- the cations used are in particular ions of the alkali metals, preferably lithium, sodium and potassium, the alkaline earth metals, preferably calcium and magnesium, and the transition metals, preferably manganese, copper, zinc and iron, and ammonia.
- nium where, if desired, a 4 alkyl to four hydrogen atoms are replaced by CC, hydroxy-C C4 alkyl, CC 4 -alkoxy-CrC alkyl, hydroxy-CrC 4 may be replaced alkoxy-dC-alkyl, phenyl or benzyl, preferably ammonium, dimethylammonium, diisopropylammonium, tetramethylammonium, tetrabutylammonium, 2- (2-hydroxyeth-1-oxy) eth-1-ylammonium, di (2-hydroxyeth-1-yl) -ammonium, trimethylbenzylammonium, furthermore phosphonium ions, Sulfonium ions
- Anions of useful acid addition salts are primarily chloride, bromide, fluoride, hydrogen sulfate, sulfate, dihydrogen phosphate, hydrogen phosphate, nitrate, bicarbonate, carbonate, hexafluorosilicate, hexafluorophosphate, benzoate and the anions of dC 4 alkanoic acids, preferably formate, acetate, propionate and butyrate ,
- the organic moieties mentioned for the substituents R 1 -R 20 or as radicals on phenyl or heterocyclic rings represent collective terms for individual enumerations of the individual group members.
- All hydrocarbon chains eg all alkyl, alkenyl, alkynyl, cyanoalkyl, haloalkyl , Haloalkenyl, haloalkynyl, alkoxy, haloalkoxy, alkoxyalkyl, alkylcarbonyl, alkenylcarbonyl, alkynylcarbonyl, alkoxycarbonyl, aikeneyloxycarbonyl, alkynyloxycarbonyl, alkylamino, alkylaminocarbonyl, alkenylaminocarbonyl, alkynylaminocarbonyl -, Alkylsulfonylamino- carbonyl, dialkylaminocarbonyl, N-alkenyl-N-alkylaminocarbonyl, N
- halogenated substituents preferably carry one to five identical or different halogen atoms.
- the meaning halogen in each case stands for fluorine, chlorine, bromine or iodine.
- O-phenyl e.g. Methyl, ethyl, n-propyl, 1-methylethyl, n-butyl, 1-methylpropyl, 2-methylpropyl and 1, 1-dimethylethyl;
- d-Ce-alkyl and the alkyl moieties of d-Ce-alkylsulphonylaminocarbonyl N- (C 3 - C 6 -alkenyl) -N- (C 1 -C 6 alkyl) aminocarbonyl, (C3 -Ce alkynyl) - N- (C ⁇ -C6 alkyl) - aminocarbonyl, N- (C 1 -C 6 -alkoxy) -N- (C 1 -Ce-alkyl) aminocarbonyl, dC 6 - alkylcarbonyl-C ⁇ -C 6 alkyl, d-Ce-alkoxyimino-Ci-Ce-alkyl, N- (C 1 -C 6 -alkylamino) -imino-Cr-C 6 -alkyl, N- i-Ci-Ce-alkyl-aminoHmino-d-Ce-alkyl ,
- C 1 -C 6 -alkylcarbonyl and the alkylcarbonyl radicals of C 1 -C 6 -alkylcarbonyl-C 1 -C 6 -alkyl, phenylCrC 6 -alkylcarbonyl and heterocyclylCrC 6 -alkylcarbonyl: C 1 -C 4 -alkylcarbonyl, as mentioned above, and also, for example, pentylcarbonyl , 1-methylbutylcarbonyl, 2-methylbutylcarbonyl, 3-methylbutylcarbonyl, 2,2-
- C 3 -C 6 -cycloalkyl and the cycloalkyl moieties of C 3 -C 6 -cycloalkylcarbonyl monocyclic, saturated hydrocarbon having 3 to 6 ring members, such as cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl;
- (C 3 -C 6 alkenyl) -N- (Cr-C 6 -alkoxy) aminocarbonyl for example, 1-propenyl, 2-propenyl, 1-methylethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-methyl 1-propenyl, 2-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl, 1-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1-methyl-1 -butenyl, 2-methyl-1-butenyl, 3-methyl-1-butenyl, 1-methyl-2-butenyl, 2-methyl-2-butenyl, 3-methyl-2-butenyl, 1-methyl-3-butenyl .
- dC 4 cyanoalkyl eg cyanomethyl, 1-cyanoeth-1-yl, 2-cyanoeth-1-yl, 1-cyanoprop-1-yl, 2-cyanoprop-1-yl, 3-cyanoprop-1-yl, 1 Cyanoprop-2-yl, 2-cyanoprop-2-yl, 1-cyanobut-1-yl, 2-cyanobut-1-yl, 3-cyanobut-1-yl, 4-cyanobut-1-yl, 1-cyanobutyl 2-yl, 2-cyanobut-2-yl, 1-cyanobut-3-yl, 2-cyanobut-3-yl, 1
- C 3 -C 6 -haloalkynyl a C 3 -C 6 -alkynyl radical as mentioned above which is partially or completely substituted by fluorine, chlorine, bromine and / or iodine, for example 1,1-difluoro-prop-2-yn 1-yl, 3-iodo-prop-2-yn-1-yl, 4-fluorobut-2-yn-1-yl, 4-chlorobutyl
- dC 4 -haloalkoxy a dC-alkoxy radical as mentioned above which is partially or completely substituted by fluorine, chlorine, bromine and / or iodine, eg fluoromethoxy, difluoromethoxy, trifluoromethoxy, chlorodifluoromethoxy, bromodifluoromethoxy, 2-fluoroethoxy, 2-chloroethoxy, 2-bromomethoxy, 2-iodoethoxy, 2,2-difluoroethoxy, 2,2,2-trifluoroethoxy, 2-chloro-2-fluoroethoxy, 2-
- dC 6 -haloalkoxy dC 4 -haloalkoxy as mentioned above, and also, for example, 5-fluoropentoxy, 5-chloropentoxy, 5-bromopentoxy, 5-iodopentoxy, undecamluoropentoxy, 6-fluorohexoxy, 6-chlorohexoxy, 6-bromohexoxy, 6-iodohexoxy and dodecafluorohexoxy;
- Methylpropoxy) butyl 2- (2-methylpropoxy) butyl, 2- (1,1-dimethylethoxy) butyl, 3- (methoxy) butyl, 3- (methoxy) butyl, 3- (ethoxy) butyl, 3- (propoxy ) butyl, 3- (1-methylethoxy) butyl, 3- (butoxy) -butyl, 3- (1-methylpropoxy) butyl, 3- (2-methylpropoxy) butyl, 3- (1,1-dimethylethoxy) butyl, 4 - (methoxy) -butyl, 4- (ethoxy) butyl, 4- (propoxy) butyl, 4- (1-methylethoxy) butyl, 4- (butoxy) butyl, 4- (1-
- dC 4 -AIkylthio for example methylthio, ethylthio, propylthio, 1-methylethylthio, butyl tylthio, 1-methylpropylthio, 2-methylpropylthio and 1,1-dimethylethylthio;
- CC 6 -alkylthio C 1 -C 4 -alkylthio as mentioned above, and also, for example, pentylthio, 1-methylbutylthio, 2-methylbutylthio, 3-methylbutylthio, 2,2-dimethylpropylthio, 1-ethylpropylthio, hexylthio, 1,1-dimethylpropylthio, 1, 2 Dimethylpropylthio, 1-methylpentylthio, 2-methylpentylthio, 3-methylpentylthio, 4-methylpentylthio, 1,1-dimethylbutylthio, 1,2-dimethylbutylthio, 1,3-dimethylbutylthio, 2,2-dimethylbutylthio, 2,3-dimethylbutylthio , 3,3-dimethylbutylthio, 1-ethylbutylthio, 2-ethylbutylthio, 1,1,2-trimethylpropylthi
- C 1 -C 6 -alkylamino and the alkylamino radicals of N- (C 1 -C 6 -alkylamino) -iminoCr-C 6 -alkyl for example methylamino, ethylamino, propylamino, 1-methylethylamino, butylamino, 1-methylpropylamino, 2-methylpropylamino, 1,1-dimethylethylamino, pentylamino, 1-methylbutylamino, 2-methylbutylamino, 3-methylbutylamino, 2,2-dimethylpropylamino, 1-ethylpropylamino, hexylamino, 1,1-dimethylpropylamino, 1,2-dimethylpropylamino, 1-methylpentylamino, 2-
- Di- (C 1 -C 4 ) -alkyl) -amino for example N, N-dimethylamino, N, N-diethylamino, N, N-dipropylamino, N, N-di (1-methylethyl) -amino, N, N-dibutylamino, N, N-di- (1-methylpropyl) amino, N, N-di (2-methylpropyl) amino, N, N -di (1,1-dimethylethyl) amino, N-ethyl-N -methylamino, N-methyl-N-propylamino, N -methyl-N- (1-methylethyl) amino, N-butyl-N-methylamino, N -methyl-N- (1-methylpropyl) amino, N -methyl-N - (2-methylpropyl) amino, N- (1, 1-dimethyl-ethyl) -N-methylamino
- (d-Ce-Alkylamino) arbonyl (dd-AlkylaminoJcarbonyl as mentioned above, and also, for example, pentylaminocarbonyl, 1-methylbutylaminocarbonyl, 2-methylbutylaminocarbonyl, 3-methylbutylaminocarbonyl, 2,2-dimethylpropylaminocarbonyl, 1-ethylpropylaminocarbonyl, hexylaminocarbonyl, 1,1-dimethylpropylaminocarbonyl, 1,2-dimethylpropylaminocarbonyl, 1-methylpentylaminocarbonyl,
- Ethylbutylaminocarbonyl 1,1,2-trimethylpropylaminocarbonyl, 1,2,2-trimethyl-propylaminocarbonyl, 1-ethyl-1-methylpropylaminocarbonyl or 1-ethyl-2-methylpropylaminocarbonyl;
- Di- (C 1 -C 6 -alkyl) aminocarbonyl di (C 1 -C 4 -alkyl) aminocarbonyl, as mentioned above, and also, for example, N-methyl-N-pentylaminocarbonyl, N-methyl-N- (1-methylbutyl ) -aminocarbonyl, N-methyl-N- (2-methylbutyl) -aminocarbonyl, N-methyl-N- (3-methylbutyl) -aminocarbonyl, N-methyl-N- (2,2-dimethylpropyl) -aminocarbonyl, N- Methyl N- (1-ethyl-propyl) -aminocarbonyl, N-methyl-N-hexylaminocarbonyl, N-methyl-N- (1,1-dimethylpropyl) -aminocarbonyl, N-
- Di- (C 1 -C 6 -alkyl) -aminothiocarbonyl for example N, N-dimethylaminothiocarbonyl, N, N-diethylaminothiocarbonyl, N, N-di (1-methylethyl) aminothiocarbonyl, N, N-dipropylaminothiocarbonyl, N, N-dibutylaminothiocarbonyl , N, N-di (1-methylpropyl) -amino-thiocarbonyl, N, N-di- (2-methylpropyl) -aminothiocarbonyl, N, N-di- (1,1-dimethyl-ethyl) -aminothiocarbonyl, N Ethyl-N-methylaminothiocarbonyl, N-methyl-N-propylaminothiocarbonyl, N-methyl-N- (1-methylethyl) -aminothiocarbony
- N- (1-methylpropyl) -aminothiocarbonyl N-ethyl-N- (2-methylpropyl) -aminothiocarbonyl, N-ethyl-N- (1, 1-dimethylethyl) -aminothiocarbonyl, N- (1-methylethyl) -N- propylaminothiocarbonyl, N-butyl-N-propylaminothiocarbonyl, N- (1-methylpropyl) -N-propylaminothiocarbonyl, N- (2-methylpropyl) -N-propylamino-thiocarbonyl, N- (1, 1-dimethylethyl) -N-propylaminothiocarbonyl, N-butyl-N- (1-methylethyl) -aminothiocarbonyl, N- (1-methylethyl) -N- (1-methylpropyl) -aminothiocarbon
- Heterocyclyl as well as the Heterocyclylmaschine of Heterocyclyl-dC 6 -alkyl, Heterocyclylcarbonyl, HeterocyclylcarbonylCrC 6 -alkyl, Heterocyclyloxycarbonyl, Het- rocyclylaminocarbonyl, heterocyclylsulfonylaminocarbonyl, N- (C 1 -C 6 -alkyl) -N- (heterocyclyl) -aminocarbonyl and heterocyclylCrC 6 -alkylcarbonyl: a saturated, partially unsaturated or aromatic 5- or 6-membered heterocyclic ring containing one to four identical or different heteroatoms selected from the group oxygen, sulfur or nitrogen, and may be bonded via C or N, for example
- N-linked, 5-membered, saturated rings such as:
- C-linked, 5-membered, partially unsaturated rings such as: 2,3-dihydrofuran-2-yl, 2,3-dihydrofuran-3-yl, 2,5-dihydrofuran-2-yl, 2,5-dihydrofuran 3-yl, 4,5-dihydrofuran-2-yl, 4,5-dihydrofuran-3-yl, 2,3-dihydro-thien-2-yl, 2,3-dihydro-thien-3-yl, 2.5 Dihydroothien-2-yl, 2,5-dihydro-thien-3-yl, 4,5-dihydro-2-yl, 4,5-dihydro-thien-3-yl, 2,3-dihydro-1H-pyrrole-2 yl,
- N-linked, 5-membered, partially unsaturated rings such as:
- N-linked, 5-membered, aromatic rings such as:
- N-linked, 6-membered, saturated rings such as:
- Tetrahydropyrimidin-6-yl 1,2,3,4-tetrahydropyrazine-2-yl, 1,2,3,4-tetrahydropyrazine-5-yl, 1,2,3,4-tetrahydropyrimidin-2-yl, 1, 2,3,4-tetrahydropyrimidine ⁇ 4-yl, 1, 2,3,4-tetrahydropyrimidin-5-yl, 1, 2,3,4-tetrahydropyrimidin-6-yl, 2,3-dihydro-1, 4-thiazine-2-yl, 2,3-dihydro-1, 4-thiazine-3-yl, 2,3-dihydro-1, 4-thiazine-5-yl, 2,3-dihydro-1, 4 thiazine-6-yl, 2H-
- variables of the compounds of the formula I have the following meanings, these being considered on their own as well as in Combination with each other represent special embodiments of the compounds of formula I:
- R 1 is halogen, C 1 -C 4 -alkyl or C 1 -C 6 -haloalkyl; particularly preferably halogen or dC 6 -haloalkyl; especially preferably halogen or CC-haloalkyl; most preferably fluorine, chlorine or CF 3 ; means.
- R 2 is hydrogen, halogen, NO 2 , C 1 -C 4 -alkyl or C 1 -C 6 -haloalkyl; very preferably hydrogen, halogen, NO 2 or dC 6 -haloalkyl; particularly preferably hydrogen, halogen, NO 2 or dC 4 -haloalkyl; especially preferably hydrogen, fluorine, chlorine, NO 2 or CF 3 ; most preferably hydrogen, fluorine, chlorine or NO 2 ; very particularly preferably hydrogen, fluorine or NO 2 ; means.
- R 2 and R 3 are independently hydrogen, halogen, CC 4 alkyl or dC 6 haloalkyl; very preferably hydrogen, halogen or dC 6 -haloalkyl; particularly preferably hydrogen, halogen or C 1 -C 4 -haloalkyl; especially preferably hydrogen, fluorine, chlorine or CF 3 ; most preferably hydrogen, fluorine or chlorine; very particularly preferably hydrogen or fluorine; mean.
- R 4 is hydrogen, halogen, CC 4 alkyl or CC 4 -haloalkyl; particularly preferably hydrogen, halogen or CC 4 -alkyl; especially preferably hydrogen or halogen; most preferably hydrogen; means. Also preferred are the benzoyl-substituted phenylalanine amides of the formula I in which
- R 5 is hydrogen, halogen, C 1 -C 4 -alkyl or CC 4 -haloalkyl; particularly preferably hydrogen, halogen or C 1 -C 4 -alkyl; especially preferably hydrogen or halogen; most preferably hydrogen; means.
- R 6 is hydrogen
- R 7 is hydrogen or hydroxy; particularly preferably hydrogen; mean.
- R 8 is CrCe-alkyl or dC 6 -haloalkyl; particularly preferably CC 6 -alkyl; especially preferably C 1 -C 4 -alkyl; most preferably CH 3 ; means.
- R 9 is OR 16 or SR 17 ; particularly preferred OR 16 ; means.
- R 9 OR 16 or NR 18 R 19 ; particularly preferred NR 18 R 19 ; means.
- R 9 SR 16 or NR 18 R 19 particularly preferred SR 16 ; means.
- R 10 is hydrogen or dd-alkyl; preferably hydrogen or CH 3 ; especially preferably hydrogen; means.
- R 11 is hydrogen, halogen, C 1 -C 6 -alkyl, hydroxy, C 1 -C 6 -alkoxy, hydroxy-C 1 -C 4 -alkyl, C 1 -C 6 -alkoxy-C 1 -C 4 -alkyl, tri (C 1 -C 6 -alkyl) silyloxy-C 1 -C 4 -alkyl, hydroxycarbonyl) - dd-alkoxy, (CC 4 alkoxycarbonyl) CRC -alkoxy 4, [di (dC 4 - alkyl) aminocarbonyl] oxy-CrC 4 alkyl, -C 4 alkylsulfonylamino, dC 4 - haloalkylsulfonylamino, (CrC alkylcarbonyl ) amino or phenyl, where the phenyl radical may carry one to three radicals from the following group: halogen, nitro, C 1
- R 12 is hydrogen, halogen, CC 6 alkyl, CC 6 haloalkyl or (CC 4 alkylcarbonyl) amino; particularly preferably hydrogen, halogen, C 1 -C 6 -alkyl or (C 1 -C -alkylcarbonyl) -amino; especially preferably hydrogen, halogen, C 1 -C 4 -alkyl or (C 1 -C 4 -alkylcarbonyl) -amino; means.
- R 12 is hydrogen, halogen, CC 6 alkyl or dC 6 haloalkyl; particularly preferably hydrogen, halogen or C 1 -C 6 -alkyl; especially preferably hydrogen, halogen or C 1 -C 4 -alkyl; vorordenrlich preferably hydrogen or halogen; very particularly preferably hydrogen, fluorine or chlorine; means.
- R 13 , R 14 and R 15 are each independently hydrogen, halogen, cyano, CC 4 alkyl or CC 4 haloalkyl; particularly preferably hydrogen, halogen or cyano; especially preferably hydrogen, fluorine or chlorine; most preferably hydrogen; mean.
- the benzoyl phenylalanine amides are also preferably of the formula I, in which R 16, R 17 and R 18 are each independently hydrogen, dC 6 alkyl, C 3 -C 6 alkenyl, C 3 -C 6 -alkynyl, CC 6 alkylcarbonyl, C 2 - C ⁇ -alkenylcarbonyl, C 3 -C 6 cycloalkylcarbonyl, CC 6 alkoxycarbonyl, dC 6 - alkylaminocarbonyl, CrC 6 -Alkylsulfonylaminocarbonyl, di- (CrC 6 -alkyl) - aminocarbonyl, N- (CrC 6 - Alkoxy) -N- (C 1 -C 6 -alkyl) aminocarbonyl, di- (CC 6 -alkyl) -aminocarbonyl, C 1 -C 6 -alkoxyimino-C 1 -C 6
- R 16 , R 17 and R 18 are each independently hydrogen, C 1 -C 6 -alkyl, C 3 -C 6 -alkenyl, C 3 -C 6 -alkynyl, C 1 -C 6 -alkylcarbonyl, C 2 -C 6 -alkenylcarbonyl, C 3 -C 6 cycloalkylcarbonyl, CrC 6 alkoxycarbonyl, dC 6 - alkylaminocarbonyl, di- (CrC 6 alkyl) aminocarbonyl, N- (CrC 6 -alkoxy) -N- (CrC 6 - -alkyl) aminocarbonyl, di- ( C 1 -C 6 -alkyl) -aminothiocarbonyl, C 1 -C 6 -alkoxyimino-C 6 -alkyl, where the said alkyl, cycloalkyl or alkoxy radicals may be partially or completely halogenated and
- the benzoyl phenylalanine amides are also preferably of the formula I, in which R 16 and R 18 are each independently hydrogen, CrC 6 alkyl, C 3 -C 6 alkenyl, C 3 -C 6 -alkynyl, -C 6 alkylcarbonyl, CC 6 alkoxycarbonyl, CrC 6 alkylaminocarbonyl, di (CrC 6 alkyl) aminocarbonyl, N- (CrC 6 alkoxy) -N- (CrC 6 alkyl) amino carbonyl, wherein said alkyl, Alkoxy may be partially or fully halogenated and / or may carry one to three of the following groups: cyano, dC 4 alkoxy, dd-alkylaminocarbonyl or di- (dC 4 - alkyl) -aminocarbonyl; Phenyl-C 1 -C 6 -alkyl, phenylcarbonyl, phenylcarbony
- R 17 is hydrogen, C 1 -C 6 -alkylcarbonyl, CC 6 -alkoxycarbonyl, CC 6 -alkylaminocarbonyl, di- (C 1 -C 6 -alkyl) aminocarbonyl or N- (dC 6 - Alkoxy) -N- (C 1 -C 6 -alkyl) amino-carbonyl, where the said alkyl and alkoxy radicals may be partially or completely halogenated and / or may carry one to three of the following groups: cyano or C 1 -C 4 -alkoxy; particularly preferably hydrogen, dC 4 alkylcarbonyl, dC 4 alkoxycarbonyl, Crd-alkylaminocarbonyl, di- (CrC alkyl) aminocarbonyl or N- (dC - alkoxy) -N- (-C 4
- R 19 is hydrogen, C 1 -C 6 -alkyl, C 3 -C 6 -cycloalkyl, C 3 -C 6 -alkenyl or C 3 -C 6 -alkynyl, where the 4 last-mentioned radicals may be partially or completely halogenated and / or one to three of the may carry the following groups: cyano, hydroxy, C 3 -C 6 -cycloalkyl, dC 4 -alkoxy, CC 4 -alkylthio, di- (CC 4 -alkyl) -amino, C 1 -C 4 -alkylcarbonyl, hydroxycarbonyl, C 1 -C 4 -alkoxycarbonyl, aminocarbonyl , dd-alkylaminocarbonyl, di- (C 1 -C 4 -alkyl) -aminocarbonyl or C 1 -C 4 -alkylcarbonyloxy; Phenyl or phen
- R 20 is C 1 -C 6 -alkyl, C 1 -C 6 -haloalkyl or phenyl, where the phenyl radical may be partially or partially halogenated and / or may be substituted by C 1 -C 6 -alkyl; particularly preferably C 1 -C 4 -alkyl, dd-haloalkyl or phenyl; especially preferably methyl, trifluoromethyl or phenyl. means.
- R 1 is fluorine, chlorine or CF 3 ,
- R 2 and R 3 independently of one another are hydrogen, fluorine or chlorine, R 4 , R 5 , R 6 and R 7 are hydrogen, R 8 is dd-alkyl, particularly preferably CH 3 ;
- R 10 is hydrogen
- R 11 is hydrogen, halogen, cyano or C 1 -C 4 -alkyl, particularly preferably hydrogen, fluorine or CH 3 ;
- R 12 is hydrogen, halogen or cyano, particularly preferably hydrogen, fluorine or chlorine;
- R 13 , R 14 and R 15 independently of one another are hydrogen, fluorine or chlorine, particularly preferably hydrogen;
- R 16 and R 18 are independently hydrogen, dC 4 alkylcarbonyl, CC 4 - alkylaminocarbonyl, di- (CrC 4 alkyl) aminocarbonyl, phenylaminocarbonyl, N- (C1-C4 alkyl) -N- (phenyl) aminocarbonyl , SO 2 CH 3 , SO 2 CF 3 or SO 2 (C 6 H 5 );
- R 7 is hydrogen, dC alkylcarbonyl, CrC 4 -AIkoxycarbonyl, CC 4 - alkylaminocarbonyl, di- (CrC alky
- R 19 is hydrogen or C 1 -C 4 -alkyl; mean.
- R 3 is chlorine
- Chlorine and R 2 is fluorine.
- the compounds of the formula Ia8 in particular the compounds of the formulas Ia8.1 to Ia5.558, which differ from the corresponding the compounds of formula la1.1 to la1.558 differ in that R1 is chlorine and R 2 and R 3 is fluorine.
- R 3 is chlorine and R 2 is fluorine.
- the compounds of the formula Ia14 in particular the compounds of the formula Ia.14.1 to La.14.558, which differ from the corresponding compounds of the formula Ia1.1 to Ia1.558 in that R 1 is chlorine, R 2 is CF 3 and R 3 is fluorine.
- the compounds of the formula Ia15 in particular the compounds of the formula Ia1.1.1 to Ia5.558, which differ from the corresponding compounds of the formula Ia1.1 to Ia1.558 in that R 1 and R 3 are chlorine and R 2 stand for CF 3 .
- the compounds of the formula La.19 in particular the compounds of the formula Ia19.1 to Ia19.558, which differ from the corresponding compounds of the formula La.1.1 to Ia1.558 in that R 1 is CF 3 and R 2 is fluorine.
- the compounds of the formula Ia20 in particular the compounds of the formula Ia20.1 to Ia20558, which differ from the corresponding compounds of the formula Ia.1.1 to Ia1.558 in that R 1 is CF 3 and R 2 and R 3 is fluorine.
- the compounds of the formula Ia21 in particular the compounds of the formula Ia21.1 to Ia1.558, which differ from the corresponding compounds of the formula I.1.1 to Ia1.558 in that R 1 is CF 3 , R 2 is fluorine and R 3 is chlorine.
- the compounds of the formula Ia22 in particular the compounds of the formula Ia22.1 to Ia22.558, which differ from the corresponding compounds of the formula La.1.1 to La.1.558 in that R 1 is CF 3 and R 2 stands for chlorine.
- the compounds of the formula Ia23 in particular the compounds of the formula Ia23.1 to Ia23.558, which differ from the corresponding compounds of the formula Ia.1.1 to Ia1.558 in that R 1 is CF 3 , R 2 is chlorine and R 3 is fluorine.
- the compounds of the formula la24 in particular the compounds of the formula la24.1 to la24.558, which differ from the corresponding compounds of the formula La.1.1 to la 1.558 in that R 1 is CF 3 and R 2 and R 3 are chlorine.
- the compounds of the formula Ia27 in particular the compounds of the formula Ia27.1 to Ia27.558, which differ from the corresponding compounds of the formula Ia.1.1 to Ia1.558 in that R 1 and R 2 are each CF 3 and R 3 is chlorine.
- the compounds of the formula Ia29 in particular the compounds of the formulas Ia29.1 to Ia29.558, which differ from the corresponding compounds of the formulas La.1.1 to Ia1.558 in that R 1 is CF 3 and R 3 and R 4 are fluorine.
- benzoyl-substituted phenylalanine amides of the formula I can be obtained in various ways, for example by the following processes:
- a phenylalanine of the formula V is first reacted with benzoic acids or benzoic acid derivatives of the formula IV to give the corresponding benzoyl derivative of the formula III, which subsequently reacts with an amine of the formula II to give the desired benzoyl-substituted phenylalanine amide of the formula I:
- L is a nucleophilically displaceable leaving group, for example hydroxy or dC 6 -alkoxy.
- L 2 is a nucleophilically displaceable leaving group, for example, hydroxy, halogen, d-Ce-alkylcarbonyl, d-Ce-alkoxycarbonyl, dd-alkylsulfonyl, phosphoryl or iso-reyl.
- Suitable activating reagents are condensation agents such as, for example, polystyrene-bonded dicyclohexylcarbodiimide, diisopropylcarbodiimide, carbonyldiimidazole, chlorocarbonic acid esters such as methyl chloroformate, ethyl chloroformate, isoropyl chloroformate, isobutyl chloroformate, sec-butyl chloroformate or allyl chloroformate, pivaloyl chloride, polyphosphoric acid, propanephosphonic anhydride, bis (2-oxo-3-oxazolidinyl) phosphoryl chloride (BOPCI) or sulfonyl chlorides such as methanesulfonyl chloride, toluenesulfonyl chloride or benzenesulfonyl chloride.
- condensation agents such as, for example, polystyrene-bonded dicyclohexylcarbodiimide, diis
- Suitable solvents are aliphatic hydrocarbons such as pentane, hexane, cyclohexane and mixtures of C 5 -C 8 -alkanes, aromatic hydrocarbons such as benzene, toluene, o-, m- and p-xylene, halogenated hydrocarbons such as methylene chloride, chloroform and chlorobenzene, ethers such as diethyl ether , Diisopropyl ether, tert-butyl methyl ether, dioxane, anisole and tetrahydrofuran (THF), nitriles such as acetonitrile and propionitrile, ketones such as acetone, methyl ethyl ketone, diethyl ketone and tert-butyl methyl ketone, and dimethyl sulfoxide, dimethylformamide (DMF), dimethylacetamide (DMA) and N- Methylpyr
- Suitable bases are generally inorganic compounds such as alkali metal and alkaline earth metal hydroxides such as lithium hydroxide, sodium hydroxide, potassium hydroxide and calcium hydroxide, alkali metal and alkaline earth metal oxides such as lithium oxide, sodium oxide, calcium oxide and magnesium oxide, alkali metal and alkaline earth metal hydrides such as lithium hydride, sodium hydride, potassium hydride and Calcium hydride, alkali metal and alkaline earth metal carbonates such as lithium carbonate, potassium carbonate and calcium carbonate and alkali metal bicarbonates such as sodium bicarbonate, also organic bases, eg tertiary amines such as trimethylamine, triethylamine, diisopropylethylamine, N-methylmorpholine, and N-methylpiperidine, pyridine, substituted pyridines such as collidine, lutidine and 4-dimethylaminopyridine and bicyclic amines into consideration. Particularly preferred are sodium hydroxide, triethyl
- the bases are generally used in equimolar amounts. But they can also be used in excess or optionally as a solvent.
- the starting materials are generally reacted with one another in equimolar amounts. It may be advantageous to use IV in an excess relative to V.
- reaction mixtures are worked up in a customary manner, for example by mixing with water, separating the phases and optionally chromatographic purification of the crude products.
- the intermediate and end products fall z. T. in the form of viscous oils, which are freed or purified under reduced pressure and at moderately elevated temperature of volatile fractions. If the intermediate and end products are obtained as solids, the purification can also be carried out by recrystallization or digestion.
- Suitable solvents are aliphatic hydrocarbons such as pentane, hexane, cyclohexane and mixtures of C 5 -C 8 -alkanes, aromatic hydrocarbons such as benzene, toluene, o-, m- and p-xylene, halogenated hydrocarbons such as methylene chloride, chloroform and chlorobenzene, ethers such as diethyl ether , Diisopropyl ether, tert-butyl methyl ether, dioxane, anisole and tetrahydrofuran (THF), nitriles such as acetonitrile and propionitrile, ketones such as acetone, methyl ethyl ketone, diethyl ketone and tert-butyl methyl ketone, and dimethyl sulfoxide, dimethylformamide (DMF), dimethylacetamide (DMA) and N- Methylpyr
- Bases generally include inorganic compounds such as alkali metal and alkaline earth metal hydroxides such as lithium hydroxide, sodium hydroxide, potassium hydroxide and calcium hydroxide, alkali metal and alkaline earth metal oxides such as lithium oxide, sodium oxide, calcium oxide and magnesium oxide, alkali metal and alkaline earth metal hydrides such as lithium hydride, sodium hydride, potassium hydride and calcium hydride, Alkali metal and alkaline earth metal carbonates such as lithium carbonate, potassium carbonate and calcium carbonate and alkali metal bicarbonates such as sodium bicarbonate, as well as organic bases, for example tertiary amines such as trimethylamine, triethylamine, diisopropylethylamine, N-methylmorpholine, and N-methylpiperidine, pyridine, substituted pyridines such as collidine, lutidine and 4-Dimethylamin ⁇ pyridin and bicyclic amines into consideration.
- sodium hydroxide triethyl
- the bases are generally used in equimolar amounts. But they can also be used in excess or optionally as a solvent.
- the starting materials are generally reacted with one another in equimolar amounts. It may be advantageous to use IV in an excess relative to V.
- the phenylalanines of the formula V can first be reacted with amines of the formula II to give the corresponding amides, which then react with benzoic acid (derivatives) n of the formula IV to give the desired benzoyl-substituted phenylalanine amides of the formula I.
- R 9 OR 16 : by condensation of glycinenolate equivalents with benzaldehydes (Hvidt, T. et al., Tetrahedron Lett 27 (33), 3807-3810 (1986); Saeed, A. et al., Tetrahedron 48 (12 ), 2507-2514 (1992); Kikuchi, J. et al., Chem. Lett. (3), 553-556 (1993); Soloshonok, VA et al., Tetrahedron Lett. 35 (17), 2713-2716 (1994); Soloshonok, VA; et al.; Tetrahedron 52 (1), 245-254 (1996); Rozenberg, V. et al., Angew. Chem.
- R 9 SR 17 : by cleavage of 2-acylamino-3-thioalkyl-phenylalanine derivatives (Villeneuve, G. et al., J. Chem. Soc. Perkin Trans 1 (16), 1897-1904 (1993)) by ring-opening of thiazolidinethiones (Cook, AH et al., J. Chem. Soc. 1337 (1948).)
- R 9 NR 18 R 19 : by ring opening of substituted imidazolinones (Kavrakova, IK et al., Org. Prep. Proced. Int. 28 (3), 333-338 (1996))
- R 9 OR 16 : by condensation of glycinenolate equivalents with aldehydes: Nicolaou, KC et al., J. Am. Chem. Soc. 124 (35), 10451-10455 (2002); Carrara, G. et al., Gazz. Chim. Ital. 82, 325 (1952); Fuganti, C. et al., J. Org. Chem. 51 (7), 1126-1128 (1986); Boger, D.L. et al., J. Org. Chem. 62 (14), 4721-4736 (1997); Honig, H. et al., Tetrahedron (46), 3841 (1990); Kanemasa, S. et al., Tetrahedron Lett. 34 (4), 677-680 (1993); US 4873359)
- R 9 SR 17 : by ring opening of substituted thiazolidines (Nagai, U. et al., Heterocycles 28 (2), 589-592 (1989)) by ring opening of substituted aziridines with thiols (Legters, J. et al., Recl Trav. Chim. Pays-Bas 111 (1), 16-21 (1992))
- R 9 NR 18 R 19 : by reduction of substituted 2-azido-3-amino-phenylalanine derivatives (Lee SH, Tetrahedron 57 (11), 2139-2145 (2001)) by ring opening of substituted imidazolines (Zhou, XT et al., Tetrahedron Asymmetr. 10 (5), 855-862 (1999); Hayashi, T. et al., Tetrahedron Lett.
- the benzoic acids (derivatives) of the formula IV required for the preparation of the benzoyl derivatives of the formula III can be purchased or can be prepared analogously to regulations known from the literature by a Grignard-Redition from the corresponding halide [eg A. Mannschuk et al., Angew , Chem. 100, 299 (1988)].
- Suitable activating reagents are condensing agents, e.g. polystyrene-bonded dicyclohexylcarbodiimide, diisopropylcarbodiimide, carbonyldiimidazole, chlorocarbonic acid esters such as methyl chloroformate, ethyl chloroformate, isoropyl chloroformate, isobutyl chloroformate, sec-butyl chloroformate or allyl chloroformate, pivaloyl chloride, polyphosphoric acid, propanephosphonic anhydride, bis (2-oxo-3-oxazolidinyl) phosphoryl chloride (BOPCI) or sulfonyl chlorides such as methanesulfonyl chloride, toluenesulfonyl chloride or benzenesulfonyl chloride.
- condensing agents e.g. polystyrene-bonded dicyclohexylcarbodi
- Suitable solvents are aliphatic hydrocarbons such as pentane, hexane, cyclohexane and mixtures of C 5 -C 8 alkanes, aromatic hydrocarbons such as benzene, toluene, o-, m- and p-xylene, halogenated hydrocarbons such as methylene chloride, chloroform and chlorobenzene, ethers such as Diethyl ether, diisopropyl ether, tert-butyl methyl ether, dioxane, anisole and tetrahydrofuran (THF), nitriles such as acetonitrile and propionitrile, ketones such as acetone, methyl ethyl ketone, diethyl ketone and tert.
- aromatic hydrocarbons such as benzene, toluene, o-, m- and p-xylene
- halogenated hydrocarbons such as m
- Butyl methyl ketone alcohols such as methanol, ethanol, n-propanol, isopropanol, n-butanol and tert-butanol, and dimethyl sulfoxide, dimethylformamide (DMF), dimethylacetamide (DMA) and N-methylpyrrolidone (NMP) or in water, are particularly preferred Methylene chloride, THF, methanol, ethanol and water.
- DMF dimethylformamide
- DMA dimethylacetamide
- NMP N-methylpyrrolidone
- the bases used are generally inorganic compounds such as alkali metal and alkaline earth metal hydroxides such as lithium hydroxide, sodium hydroxide, potassium hydroxide and calcium hydroxide, alkali metal and alkaline earth metal oxides such as lithium oxide, sodium oxide, calcium oxide and magnesium oxide, alkali metal and alkaline earth metal hydrides such as lithium hydride, sodium hydride, potassium hydride and calcium hydride, alkali metal and alkaline earth metal carbonates such as lithium carbonate, potassium carbonate and calcium carbonate and alkali metal bicarbonates such as sodium bicarbonate, as well as organic bases, for example tertiary amines such as trimethylamine, triethylamine, diisopropylethylamine, N-methylmorpholine, and N-methylpiperidine, pyridine, substituted pyridines such as collidine, lutidine and 4-dimethylaminopyridine and bicyclic amines into consideration. Particularly preferred are sodium hydroxide, triethy
- the bases are generally used in catalytic amounts, but they can also be used equimolar, in excess or optionally as a solvent.
- the starting materials are generally reacted with one another in equimolar amounts. It may be advantageous to use II in an excess relative to III.
- the workup and isolation of the products can be done in a conventional manner.
- Suitable solvents are aliphatic hydrocarbons such as pentane, hexane, cyclohexane and mixtures of C 5 -C 8 -alkanes, aromatic hydrocarbons such as benzene, toluene, o-, m- and p-xylene, halogenated hydrocarbons such as methylene chloride, chloroform and chlorobenzene, Ether, such as diethyl ether, diisopropyl ether, tert.
- aliphatic hydrocarbons such as pentane, hexane, cyclohexane and mixtures of C 5 -C 8 -alkanes
- aromatic hydrocarbons such as benzene, toluene, o-, m- and p-xylene
- halogenated hydrocarbons such as methylene chloride, chloroform and chlorobenzene
- Ether such as diethyl ether, diisopropyl
- nitriles such as acetonitrile and propionitrile
- ketones such as acetone, methyl ethyl ketone, diethyl ketone and tert-butyl
- the reaction may optionally be carried out in the presence of a base.
- bases are generally inorganic compounds such as alkali metal and alkaline earth metal hydroxides such as lithium hydroxide, sodium hydroxide, potassium hydroxide and calcium hydroxide, alkali metal and alkaline earth metal oxides such as lithium oxide, sodium oxide, calcium oxide and magnesium oxide, alkali metal and alkaline earth metal hydrides such as lithium hydride, sodium hydride, potassium hydride and calcium hydride, alkali metal and alkaline earth metal carbonates such as lithium carbonate, potassium carbonate and calcium carbonate and alkali metal hydrogen carbonates such as sodium bicarbonate, as well as organic bases, eg tertiary amines such as trimethylamine, triethylamine, diisopropylethylamine, N-methylmorpholine, and N-methylpiperidine, pyridine, substituted pyridines such as collidine, lutidine and 4-
- Dimethylaminopyridine and bicyclic amines into consideration are particularly preferred.
- Particularly preferred are sodium hydroxide, triethylamine, ethyldiisopropylamine, N-methylmorpholine and pyridine.
- the bases are generally used in catalytic amounts, but they can also be used equimolar, in excess or optionally as a solvent.
- the starting materials are generally reacted with one another in equimolar amounts. It may be advantageous to use II in an excess relative to IM.
- the workup and isolation of the products can be done in a conventional manner.
- the amines of the formula II required for the preparation of the benzoyl-substituted phenylalanine amides of the formula I can be purchased.
- acylated glycine derivatives of the formula VIII where the acyl group is a releasable protective group such as benzyloxycarbonyl (see Villa with ⁇ benzyl) or tert-butyloxycarbonyl (cf
- L 1 is a nucleophilically displaceable leaving group, for example for hydroxy or dC 6 - alkoxy.
- L 2 is a nucleophilically displaceable leaving group, for example, hydroxy, halogen, d-Ce-alkylcarbonyl, CrC 6 alkoxycarbonyl, dd-alkylsulfonyl, phosphoryl or Iso ureyl.
- Suitable solvents are aliphatic hydrocarbons such as pentane, hexane, cyclohexane and mixtures of C 5 -C 8 alkanes, aromatic hydrocarbons such as toluene, o-, m- and p-xylene, ethers such as diethyl ether, diisopropyl ether, tert-butyl methyl ether, dioxane, anisole and tetrahydrofuran, and also dimethyl sulfoxide, dimethylformamide and dimethylacetamide, particularly preferably diethyl ether, dioxane and tetrahydrofuran.
- aliphatic hydrocarbons such as pentane, hexane, cyclohexane and mixtures of C 5 -C 8 alkanes
- aromatic hydrocarbons such as toluene, o-, m- and p-xylene
- ethers such as diethyl
- the bases used are generally inorganic compounds such as alkali metal and alkaline earth metal hydrides such as lithium hydride, sodium hydride, potassium hydride and calcium hydride, alkali metal azides such as lithium hexamethyldisilazide, organometallic compounds, in particular alkali metal alkyls such as methyllithium, butyllithium and phenyllithium, and alkali metal and alkaline earth metal alkoxides such as sodium methoxide, sodium ethanolate, potassium ethoxide, potassium tert-butoxide, potassium tert-pentoxide and dimethoxy magnesium, also organic bases, eg tertiary amines such as trimethylamine, triethylamine, diisopropylethylamine and N-methylpiperidine, pyridine, substituted pyridines such as collidine, lutidine and 4-dimethylaminopyridine and bicyclic amines into consideration. Particularly preferred are sodium
- the bases are generally used in equimolar amounts, but they can also be used catalytically, in excess or optionally as a solvent.
- the starting materials are generally reacted with one another in equimolar amounts. It may be advantageous to use the base and / or the heterocyclylcarbonyl compounds VII in an excess based on the glycine derivatives VIII.
- the workup and isolation of the products can be done in a conventional manner.
- the glycine derivatives of the formula VIII required for the preparation of the compounds I can be purchased, are known in the literature [e.g. H. Pessoa-Mahana et al., Synth. Comm. 32, 1437 (2002) or can be prepared according to the cited literature.
- L 1 is a nucleophilically displaceable leaving group, for example for hydroxy or CC 6 - alkoxy.
- L 3 is a nucleophilically displaceable leaving group, for example halogen, hydroxy, or d-Ce-alkoxy.
- Suitable solvents are aliphatic hydrocarbons such as pentane, hexane, cyclohexane and mixtures of C 5 -C 8 -alkanes, aromatic hydrocarbons such as toluene, o-, m- and p-xylene, halogenated hydrocarbons such as methylene chloride, chloroform and chlorobenzene, ethers such as diethyl ether, diisopropyl ether , tert-butyl methyl ether, dioxane, anisole and tetrahydrofuran, nitriles such as acetonitrile and propinonitrile, ketones such as acetone, methyl ethyl ketone, diethyl ketone and tert-butyl methyl ketone, alcohols such as methanol, ethanol, n-propanol, isopropanol, n-butanol and tert. Butano
- Suitable bases are generally inorganic compounds such as alkali metal and alkaline earth metal hydroxides such as lithium hydroxide, sodium hydroxide, potassium hydroxide and calcium hydroxide, alkali metal and alkaline earth metal oxides such as lithium oxide, sodium oxide, calcium oxide and magnesium oxide, alkali metal and alkaline earth metal hydrides such as lithium hydride, sodium hydride, potassium hydride and Calcium hydride, alkali metal amides such as lithium amide, sodium amide and potassium amide, alkali metal and alkaline earth metal carbonates such as lithium carbonate, potassium carbonate and calcium carbonate and alkali metal hydrogencarbonates such as sodium hydrogencarbonate, organometallic compounds, in particular alkali metal alkyls such as methyllithium, butyl lithium and phenyllithium, alkylmagnesium halides such as methylmagnesium chloride and alkali metal and alkaline earth metal alkoxides such as sodium methoxide, sodium ethanolate, potassium ethanolate
- tertiary amines such as trimethylamine, triethylamine, diisopropylethylamine and N-methylpiperidine, pyridine, substituted pyridines such as collidine, lutidine and 4-dimethylaminopyridine and bicyclic amines into consideration.
- Particularly preferred are sodium hydroxide, sodium hydride and triethylamine.
- the bases are generally used in equimolar amounts, but they can also be used catalytically, in excess or, if appropriate, as solvent.
- the starting materials are generally reacted with one another in equimolar amounts. It may be advantageous to use the base and / or IX in an excess based on III or I.
- the workup and isolation of the products can be done in a conventional manner.
- the required compounds of formula VIII can be purchased.
- L 1 is a nucleophilically displaceable leaving group, for example for hydroxy or dC 6 - alkoxy.
- L 2 is a nucleophilically displaceable leaving group, for example hydroxy, halogen, C 1 -C 6 -alkylcarbonyl, C 1 -C 6 -alkoxycarbonyl, C 1 -C 6 -alkylsulfonyl, phosphoryl or isopropyl.
- L 4 is a nucleophilically displaceable leaving group, for example for hydroxy or CC 6 - alkoxy.
- acylation of the aminomalonyl compounds of the formula XI with benzoic acid (derivatives) n of the formula IV to give corresponding N-acylaminomalonyl compounds of the formula X is usually carried out analogously to the reaction of the phenylalanines of the formula V with benzoic acid (derivatives ) n of the formula IV to the corresponding benzoyl derivatives of the formula IM.
- L 4 in the case of the N-acylaminomalonyl compounds of the formula X is C 1 -C 6 -oxy, it is advantageous to first prepare L 4 by ester hydrolysis [eg Hellmann, H. et al., Liebigs Ann. Chem. 631, 175-179 (1960)] into a hydroxy group.
- Suitable solvents are aliphatic hydrocarbons such as pentane, hexane, cyclohexane and mixtures of C 5 -C 8 -alkanes, aromatic hydrocarbons such as toluene, o-, m- and p-xylene, halogenated hydrocarbons such as methylene chloride, chloroform and chlorobenzene, ethers such as diethyl ether, Diisopropyl ether, tert-butyl methyl ether, dioxane, anisole and tetrahydrofuran, nitriles such as acetonitrile and propionitrile, ketones such as acetone, methyl ethyl ketone, diethyl ketone and tert-butyl methyl ketone, alcohols such as methanol, ethanol, n-propanol, isopropanol, n-butyl Butanol and tert-butan
- the bases used are generally inorganic compounds such as alkali metal and alkaline earth metal hydroxides such as lithium hydroxide, sodium hydroxide, potassium hydroxide and calcium hydroxide, alkali metal and alkaline earth metal oxides such as lithium oxide, sodium oxide, calcium oxide and magnesium oxide, alkali metal and alkaline earth metal hydrides such as lithium hydride, sodium hydride, potassium hydride and calcium hydride, alkali metal amides such as lithium amide, sodium amide and potassium amide, alkali metal and alkaline earth metal carbonates such as lithium carbonate, potassium carbonate and calcium carbonate and alkali metal hydrogencarbonates such as sodium bicarbonate, organometallic compounds, in particular Alkali metal alkyls such as methyllithium, butyllithium and phenyllithium, alkylmagnesium halides such as methylmagnesium chloride, and alkali metal and alkaline earth metal alcoholates such as sodium methoxide, sodium ethanolate, potassium ethanolate,
- the bases are generally used in catalytic amounts, but they can also be used equimolar, in excess or optionally as a solvent.
- the starting materials are generally reacted with one another in equimolar amounts. It may be advantageous to use the base in an excess based on X.
- the workup and isolation of the products can be done in a conventional manner.
- the required aminomalonyl compounds of formula XI can be purchased or are known in the literature [e.g. US 4904674; Hellmann, H. et al., Liebigs Ann. Chem. 631, 175-179 (1960)] or can be prepared according to the cited literature.
- L 1 is a nucleophilically displaceable leaving group, for example for hydroxy or dC 6 - alkoxy.
- L 2 is a nucleophilically displaceable leaving group, for example hydroxy, halogen, d-QrAlkylcarbonyl, dC 6 alkoxycarbonyl, CC 6 alkylsulfonyl, phosphoryl or Iso-reyl.
- the acylation of the keto compounds of the formula XIII with benzoic acid (derivatives) n of the formula IV to N-acyl keto compounds of the formula XII is usually carried out analogously to the reaction of the phenylalanines of the formula V mentioned under process A with benzoic acid (derivatives) n of the formula IV to the corresponding benzoyl derivatives of the formula
- Suitable solvents are aliphatic hydrocarbons such as pentane, hexane, cyclohexane and mixtures of C 5 -C 8 -alkanes, aromatic hydrocarbons such as toluene, o-, m- and p-xylene, halogenated hydrocarbons such as methylene chloride, chloroform and chlorobenzene, ethers such as diethyl ether, Diisopropyl ether, tert-butyl methyl ether, dioxane, anisole and tetrahydrofuran, nitriles such as acetonitrile and propionitrile, ketones such as acetone, methyl ethyl ketone, diethyl ketone and tert-butyl methyl ketone, alcohols such as methanol, ethanol, n-propanol, isopropanol, n-butanol and tert-butanol,
- Suitable reducing agents are e.g. Sodium borohydride, zinc borohydride, sodium cyanoborohydride, lithium triethylborohydride (Superhydrid®), lithium tri-sec.butylborohydride (L-Selectrid®), lithium aluminum hydride or borane [cf. e.g. WO 00/20424; Marchi, C. et al., Tetrahedron 58 (28), 5699 (2002); Blank, S. et al., Liebigs Ann. Chem. (8), 889-896 (1993); Kuwano, R. et al., J. Org. Chem. 63 (10), 3499-3503 (1998); Clariana, J. et al., Tetrahedron 55 (23), 7331-7344 (1999)].
- the reduction can also be carried out in the presence of hydrogen and a catalyst.
- Suitable catalysts are, for example, [Ru (BINAP) Cl 2 ] or Pd / C [cf. Noyori, R. et al., J. Am. Chem. Soc. 111 (25), 9134-9135 (1989); Bolhofer, A. et al., J. Am. Chem. Soc. 75, 4469 (1953)].
- the reduction can also take place the presence of a microorganism.
- a microorganism e.g. Saccharomyces Rouxii [cf. Soukup, M. et al., Helv. Chim. Acta 70, 232 (1987)].
- N-acyl keto compounds of the formula XII and the respective reducing agent are generally reacted with one another in equimolar amounts. It may be advantageous to use the reducing agent in an excess relative to XII.
- the workup and isolation of the products can be done in a conventional manner.
- R 1 to R 6 and R 9 to R 15 have the meanings mentioned under claim 1 and L 1 represents a nucleophilically displaceable leaving group such as hydroxy or dC 6 - alkoxy, are also an object of the present invention.
- benzoyl derivatives of the formula III correspond to those of the radicals R 1 to R 6 and R 9 to R 15 of the formula I. Particular preference is given to benzoyl derivatives of the formula III in which
- R 1 is fluorine, chlorine or CF 3 ,
- R 2 and R 3 independently of one another hydrogen, fluorine or chlorine
- R 4 , R 5 and R 6 are hydrogen, R 9 is OR 16 , SR 17 or NR 18 R 19 ;
- R 10 is hydrogen
- R 11 is hydrogen, fluorine or CH 3 ;
- R 12 is hydrogen, fluorine or chlorine
- R 13 , R 4 and R 15 are hydrogen;
- R 16 and R 18 independently of one another are hydrogen, CC-alkylcarbonyl, CC 4 -alkylaminocarbonyl, di (CrC 4 -alkyl) aminocarbonyl, phenylaminocarbonyl, N- (C 1 -C 4 -alkyl) -N- (phenyl) -aminocarbonyl, SO 2 CH 3 , SO 2 CF 3 or SO 2 (C 6 H 5 );
- R 17 is hydrogen, CC 4 -alkylcarbonyl, CrC alkoxycarbonyl, CC 4 - alkylaminocarbonyl, di- (CrC 4 alkyl) aminocarbonyl, N- (dC 4 alkoxy) -N- (CrC 4 - alkyl) aminocarbonyl; and
- R 19 is hydrogen or CC 4 alkyl; mean.
- the benzoyl-substituted phenylalanine amides of the formula I and their agriculturally useful salts are suitable - both as isomer mixtures and in the form of pure isomers - as herbicides.
- the compounds of the formula I containing herbicidal agents control plant growth on non-crop areas very well, especially at high application rates. In crops such as wheat, rice, corn, soybeans and cotton, they act against weeds and grass weeds without significantly damaging the crops. This effect occurs especially at low application rates.
- the compounds of the formula I or herbicidal compositions containing them can be used in a further number of crop plants for the removal of unwanted plants.
- the following cultures may be considered:
- the compounds of formula I may also be used in cultures tolerant to the action of herbicides by breeding, including genetic engineering.
- the compounds of the formula I or the herbicidal compositions containing them can be used, for example, in the form of directly sprayable aqueous solutions, powders, suspensions, also high-percentage aqueous, oily or other suspensions or dispersions, emulsions, oil dispersions, pastes, dusts, scattering agents or granules by spraying, atomizing, dusting, scattering or pouring.
- the forms of application depend on the intended use; In any case, they should ensure the finest possible distribution of the active compounds according to the invention.
- the herbicidal compositions contain a herbicidally effective amount of at least one compound of the formula I or an agriculturally useful salt of I and auxiliaries customary for the formulation of crop protection agents.
- Substantially suitable as inert auxiliaries are: mineral oil fractions of medium to high boiling point such as kerosene and diesel oil, furthermore coal tar oils and oils of vegetable or animal origin, aliphatic, cyclic and aromatic hydrocarbons, e.g. Paraffins, tetrahydronaphthalene, alkylated naphthalenes and their derivatives, alkylated benzenes and their derivatives, alcohols such as methanol, ethanol, propanol, butanol and cyclohexanol, ketones such as cyclohexanone, strong polar solvents, e.g. Amines such as N-methylpyrrolidone and water.
- mineral oil fractions of medium to high boiling point such as kerosene and diesel oil, furthermore coal tar oils and oils of vegetable or animal origin, aliphatic, cyclic and aromatic hydrocarbons, e.g. Paraffins, tetrahydronaphthalene, al
- Aqueous application forms can be prepared from emulsion concentrates, suspensions, pastes, wettable powders or water-dispersible granules by adding water.
- the substrates as such or dissolved in an oil or solvent, can be homogenized in water by means of wetting agents, tackifiers, dispersants or emulsifiers.
- concentrates consisting of active substance, wetting, adhesion, dispersing or emulsifying agent and possibly solvent or oil, which are suitable for dilution with water.
- Suitable surface-active substances are the alkali metal, alkaline earth metal, ammonium salts of aromatic sulfonic acids, for example lignin, phenol, naphthalene and dibutylnaphthalenesulfonic acid, and of fatty acids, alkyl and alkylaryl sulfonates, alkyl, lauryl ether and fatty alcohol sulfates, and salts of sulfated hexa-, hepta- and octadecanols and also of fatty alcohol glycol ethers, condensation products of sulfonated naphthalene and its derivatives with formaldehyde, condensation products of naphthalene or naphthalenesulfonic acids with phenol and formaldehyde, polyoxyethyl tocylphenol ether, ethoxylated isooctyl, octyl or nonylphenol , Alkyl tocylphenol
- Granules e.g. Coating, impregnation and homogeneous granules can be prepared by binding the active compounds to solid carriers.
- Solid carriers are mineral soils such as silicic acids, silica gels, silicates, talc, kaolin, limestone, lime, chalk, bolus, loess, clay, dolomite, diatomaceous earth, calcium and magnesium sulfate, magnesium oxide, ground plastics, fertilizers such as ammonium sulfate, ammonium phosphate, ammonium nitrate, Ureas and vegetable products such as cereal flour, tree bark, wood and nut shell flour, cellulose powder or other solid carriers.
- the concentrations of the compounds of the formula I in the ready-to-use formulations can be varied within wide limits.
- the formulations contain from about 0.001 to 98 wt .-%, preferably 0.01 to 95 wt .-%, of at least one active ingredient.
- the active ingredients are used in a purity of 90% to 100%, preferably 95% to 100% (according to NMR spectrum).
- the application of the compounds of the formula I or of the herbicidal compositions can be carried out in the preemergence or postemergence process. If the active ingredients are less compatible with certain crops, application techniques may be used in which the herbicidal agents are sprayed with the help of the sprayers so as not to hit the leaves of the sensitive crops, while the active ingredients on the leaves below grow undesirable plants or the uncovered soil surface (post-directed, lay-by).
- the application rates of compound of the formula I amount depending on the control target, season, target plants and growth stage from 0.001 to 3.0, preferably 0.01 to 1.0 kg / ha of active substance (aS).
- the benzoyl-substituted phenylalanine amides of the formula I can be mixed with numerous representatives of other herbicidal or growth-regulating active ingredient groups and applied together.
- the culture vessels used were plastic flower pots with loamy sand with about 3.0% humus as substrate.
- the seeds of the test plants were sown separately by species.
- the active ingredients suspended or emulsified in water were applied directly after sowing by means of finely distributing nozzles.
- the jars were lightly rained to promote germination and growth and then covered with clear plastic hoods until the plants had grown. This cover causes a uniform germination of the test plants, if it was not affected by the active ingredients.
- test plants were grown depending on the growth form only to a stature height of 3 to 15 cm and only then treated with the suspended or emulsified in water agents.
- the test plants were either sown directly and grown in the same containers or they were first grown separately as seedlings and transplanted into the test containers a few days before the treatment.
- the application rate for postemergence treatment was 1.0, 0.5, 0.25, 0.125 and 0.0625 kg / ha a.s. (active substance).
- the plants were kept species-specific at temperatures of 10 to 25 ° C and 20 to 35 ° C.
- the trial period lasted for 2 to 4 weeks. During this time, the plants were cared for, and their response to each treatment was evaluated.
- the rating was based on a scale of 0 to 100. 100 means no emergence of the plants or complete destruction of at least the above-ground parts and 0 no damage or normal growth course.
- the plants used in the greenhouse experiments were composed of the following species:
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Environmental Sciences (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Zoology (AREA)
- Pest Control & Pesticides (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Cosmetics (AREA)
- Heterocyclic Compounds Containing Sulfur Atoms (AREA)
- Pyridine Compounds (AREA)
- Pyrane Compounds (AREA)
Abstract
Description
Claims
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP04803999.4A EP1697309B1 (de) | 2003-12-19 | 2004-12-17 | Benzoylsubstituierte phenylalanin-amide |
| BRPI0417780A BRPI0417780B1 (pt) | 2003-12-19 | 2004-12-17 | composto, processo para preparar o mesmo, agente, processos para preparar o mesmo, e para combater vegetação indesejada, e, uso de um composto |
| JP2006544353A JP4541363B2 (ja) | 2003-12-19 | 2004-12-17 | ベンゾイル置換フェニルアラニンアミド |
| EA200601105A EA200601105A1 (ru) | 2003-12-19 | 2004-12-17 | Замещённые бензоилом фенилаланин-амиды |
| CA002548442A CA2548442A1 (en) | 2003-12-19 | 2004-12-17 | Benzoyl-substituted phenylalanine amides |
| US10/581,444 US7687435B2 (en) | 2003-12-19 | 2004-12-17 | Benzoyl-substituted phenylalanineamides |
| MXPA06005989A MXPA06005989A (es) | 2003-12-19 | 2004-12-17 | Fenilalanina-amidas sustituidas por benzoilo. |
| AU2004303492A AU2004303492A1 (en) | 2003-12-19 | 2004-12-17 | Benzoyl-substituted phenylalanine amides |
| UAA200608125A UA81568C2 (en) | 2003-12-19 | 2004-12-17 | Benzoyl-substituted phenylalanine amides, methods and intermediate products for the production thereof, herbicidal composition and method for controlling undesired plants |
| IL175884A IL175884A0 (en) | 2003-12-19 | 2006-05-24 | Benzoyl-substituted phenylalnine amides |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE10360395.6 | 2003-12-19 | ||
| DE10360395 | 2003-12-19 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2005061443A2 true WO2005061443A2 (de) | 2005-07-07 |
| WO2005061443A3 WO2005061443A3 (de) | 2005-12-22 |
Family
ID=34706404
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2004/014392 WO2005061443A2 (de) | 2003-12-19 | 2004-12-17 | Benzoylsubstituierte phenylalanin-amide |
Country Status (19)
| Country | Link |
|---|---|
| US (1) | US7687435B2 (de) |
| EP (1) | EP1697309B1 (de) |
| JP (1) | JP4541363B2 (de) |
| KR (1) | KR20060123344A (de) |
| CN (1) | CN1894202A (de) |
| AR (1) | AR047334A1 (de) |
| AU (1) | AU2004303492A1 (de) |
| BR (1) | BRPI0417780B1 (de) |
| CA (1) | CA2548442A1 (de) |
| CO (1) | CO5700811A2 (de) |
| CR (1) | CR8475A (de) |
| EA (1) | EA200601105A1 (de) |
| IL (1) | IL175884A0 (de) |
| MX (1) | MXPA06005989A (de) |
| PE (1) | PE20051058A1 (de) |
| UA (1) | UA81568C2 (de) |
| UY (1) | UY28683A1 (de) |
| WO (1) | WO2005061443A2 (de) |
| ZA (1) | ZA200605927B (de) |
Cited By (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006029829A1 (de) * | 2004-09-16 | 2006-03-23 | Basf Aktiengesellschaft | Heteroaroylsubstituierte serin-amide als herbizide |
| WO2006029828A1 (de) * | 2004-09-16 | 2006-03-23 | Basf Aktiengesellschaft | Benzoylsubstituierte serin-amide |
| WO2006125688A1 (de) * | 2005-05-25 | 2006-11-30 | Basf Aktiengesellschaft | Benzoylsubstituierte serin-amide |
| WO2007093539A2 (de) * | 2006-02-16 | 2007-08-23 | Basf Se | Benzoylsubstituierte alanine |
| WO2008084073A1 (de) * | 2007-01-11 | 2008-07-17 | Basf Se | Heteroaroylsubstituierte serin-amide |
| WO2008084070A1 (de) * | 2007-01-11 | 2008-07-17 | Basf Se | Benzoylsubstituierte serin-amide |
| WO2010049414A1 (en) * | 2008-10-31 | 2010-05-06 | Basf Se | Method for improving plant health |
| US7879761B2 (en) | 2005-05-25 | 2011-02-01 | Basf Aktiengesellschaft | Heteroaroyl-substituted serineamides |
| WO2013039990A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013040021A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013040117A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2014151255A1 (en) | 2013-03-15 | 2014-09-25 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2015108982A2 (en) | 2014-01-15 | 2015-07-23 | Monsanto Technology Llc | Methods and compositions for weed control using epsps polynucleotides |
| US9121022B2 (en) | 2010-03-08 | 2015-09-01 | Monsanto Technology Llc | Method for controlling herbicide-resistant plants |
| US9416363B2 (en) | 2011-09-13 | 2016-08-16 | Monsanto Technology Llc | Methods and compositions for weed control |
| US9422557B2 (en) | 2011-09-13 | 2016-08-23 | Monsanto Technology Llc | Methods and compositions for weed control |
| US9422558B2 (en) | 2011-09-13 | 2016-08-23 | Monsanto Technology Llc | Methods and compositions for weed control |
| US9540642B2 (en) | 2013-11-04 | 2017-01-10 | The United States Of America, As Represented By The Secretary Of Agriculture | Compositions and methods for controlling arthropod parasite and pest infestations |
| US9777288B2 (en) | 2013-07-19 | 2017-10-03 | Monsanto Technology Llc | Compositions and methods for controlling leptinotarsa |
| US9850496B2 (en) | 2013-07-19 | 2017-12-26 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US10041068B2 (en) | 2013-01-01 | 2018-08-07 | A. B. Seeds Ltd. | Isolated dsRNA molecules and methods of using same for silencing target molecules of interest |
| US10240162B2 (en) | 2012-05-24 | 2019-03-26 | A.B. Seeds Ltd. | Compositions and methods for silencing gene expression |
| US10378012B2 (en) | 2014-07-29 | 2019-08-13 | Monsanto Technology Llc | Compositions and methods for controlling insect pests |
| US10557138B2 (en) | 2013-12-10 | 2020-02-11 | Beeologics, Inc. | Compositions and methods for virus control in Varroa mite and bees |
| US10612019B2 (en) | 2013-03-13 | 2020-04-07 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10609930B2 (en) | 2013-03-13 | 2020-04-07 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10655136B2 (en) | 2015-06-03 | 2020-05-19 | Monsanto Technology Llc | Methods and compositions for introducing nucleic acids into plants |
| US10683505B2 (en) | 2013-01-01 | 2020-06-16 | Monsanto Technology Llc | Methods of introducing dsRNA to plant seeds for modulating gene expression |
| US10760086B2 (en) | 2011-09-13 | 2020-09-01 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10801028B2 (en) | 2009-10-14 | 2020-10-13 | Beeologics Inc. | Compositions for controlling Varroa mites in bees |
| US10806146B2 (en) | 2011-09-13 | 2020-10-20 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10808249B2 (en) | 2011-09-13 | 2020-10-20 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10829828B2 (en) | 2011-09-13 | 2020-11-10 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10883103B2 (en) | 2015-06-02 | 2021-01-05 | Monsanto Technology Llc | Compositions and methods for delivery of a polynucleotide into a plant |
| US10888579B2 (en) | 2007-11-07 | 2021-01-12 | Beeologics Inc. | Compositions for conferring tolerance to viral disease in social insects, and the use thereof |
| US10968449B2 (en) | 2015-01-22 | 2021-04-06 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US10988764B2 (en) | 2014-06-23 | 2021-04-27 | Monsanto Technology Llc | Compositions and methods for regulating gene expression via RNA interference |
| US11091770B2 (en) | 2014-04-01 | 2021-08-17 | Monsanto Technology Llc | Compositions and methods for controlling insect pests |
| US11807857B2 (en) | 2014-06-25 | 2023-11-07 | Monsanto Technology Llc | Methods and compositions for delivering nucleic acids to plant cells and regulating gene expression |
Families Citing this family (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1987008A2 (de) * | 2006-02-16 | 2008-11-05 | Basf Se | Heteroaroylsubstituierte alanine |
| EP2024327A1 (de) * | 2006-05-19 | 2009-02-18 | Basf Se | Benzoylsubstituierte alanine |
| US8168631B2 (en) * | 2008-02-05 | 2012-05-01 | Allergan, Inc. | 3-(4-fluorophenyl)-3-hydroxy-2-amino-propionic acid amides and related compounds having analgesic activity |
| JP2012506891A (ja) * | 2008-10-31 | 2012-03-22 | ビーエーエスエフ ソシエタス・ヨーロピア | 植物の健康を改善する方法 |
| US11653588B2 (en) | 2018-10-26 | 2023-05-23 | Deere & Company | Yield map generation and control system |
| US11240961B2 (en) | 2018-10-26 | 2022-02-08 | Deere & Company | Controlling a harvesting machine based on a geo-spatial representation indicating where the harvesting machine is likely to reach capacity |
| US11467605B2 (en) | 2019-04-10 | 2022-10-11 | Deere & Company | Zonal machine control |
| US11672203B2 (en) | 2018-10-26 | 2023-06-13 | Deere & Company | Predictive map generation and control |
| US11079725B2 (en) | 2019-04-10 | 2021-08-03 | Deere & Company | Machine control using real-time model |
| US11589509B2 (en) | 2018-10-26 | 2023-02-28 | Deere & Company | Predictive machine characteristic map generation and control system |
| US11178818B2 (en) | 2018-10-26 | 2021-11-23 | Deere & Company | Harvesting machine control system with fill level processing based on yield data |
| US11641800B2 (en) | 2020-02-06 | 2023-05-09 | Deere & Company | Agricultural harvesting machine with pre-emergence weed detection and mitigation system |
| US11957072B2 (en) | 2020-02-06 | 2024-04-16 | Deere & Company | Pre-emergence weed detection and mitigation system |
| US11234366B2 (en) | 2019-04-10 | 2022-02-01 | Deere & Company | Image selection for machine control |
| US11778945B2 (en) | 2019-04-10 | 2023-10-10 | Deere & Company | Machine control using real-time model |
| US11477940B2 (en) | 2020-03-26 | 2022-10-25 | Deere & Company | Mobile work machine control based on zone parameter modification |
| US11825768B2 (en) | 2020-10-09 | 2023-11-28 | Deere & Company | Machine control using a predictive map |
| US11844311B2 (en) | 2020-10-09 | 2023-12-19 | Deere & Company | Machine control using a predictive map |
| US11946747B2 (en) | 2020-10-09 | 2024-04-02 | Deere & Company | Crop constituent map generation and control system |
| US11895948B2 (en) | 2020-10-09 | 2024-02-13 | Deere & Company | Predictive map generation and control based on soil properties |
| US11650587B2 (en) | 2020-10-09 | 2023-05-16 | Deere & Company | Predictive power map generation and control system |
| US11874669B2 (en) | 2020-10-09 | 2024-01-16 | Deere & Company | Map generation and control system |
| US11474523B2 (en) | 2020-10-09 | 2022-10-18 | Deere & Company | Machine control using a predictive speed map |
| US11849672B2 (en) | 2020-10-09 | 2023-12-26 | Deere & Company | Machine control using a predictive map |
| US11635765B2 (en) | 2020-10-09 | 2023-04-25 | Deere & Company | Crop state map generation and control system |
| US11864483B2 (en) | 2020-10-09 | 2024-01-09 | Deere & Company | Predictive map generation and control system |
| US11845449B2 (en) | 2020-10-09 | 2023-12-19 | Deere & Company | Map generation and control system |
| US11711995B2 (en) | 2020-10-09 | 2023-08-01 | Deere & Company | Machine control using a predictive map |
| US11927459B2 (en) | 2020-10-09 | 2024-03-12 | Deere & Company | Machine control using a predictive map |
| US11849671B2 (en) | 2020-10-09 | 2023-12-26 | Deere & Company | Crop state map generation and control system |
| US11871697B2 (en) | 2020-10-09 | 2024-01-16 | Deere & Company | Crop moisture map generation and control system |
| US11675354B2 (en) | 2020-10-09 | 2023-06-13 | Deere & Company | Machine control using a predictive map |
| US11889788B2 (en) | 2020-10-09 | 2024-02-06 | Deere & Company | Predictive biomass map generation and control |
| US11727680B2 (en) | 2020-10-09 | 2023-08-15 | Deere & Company | Predictive map generation based on seeding characteristics and control |
| US11592822B2 (en) | 2020-10-09 | 2023-02-28 | Deere & Company | Machine control using a predictive map |
| US11889787B2 (en) | 2020-10-09 | 2024-02-06 | Deere & Company | Predictive speed map generation and control system |
Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4605759A (en) | 1983-08-01 | 1986-08-12 | Mitsui Toatsu Chemicals, Inc. | Process for production of β-phenylserine |
| US4810817A (en) | 1985-10-21 | 1989-03-07 | W. R. Grace & Co. | Aspartyl-beta substituted phenylalanine dipeptides |
| US4873359A (en) | 1985-10-21 | 1989-10-10 | W. R. Grace & Co. - Conn. | Process for preparing as partyl-phenylalanine dipeptides |
| US4904674A (en) | 1989-08-16 | 1990-02-27 | Hoechst-Roussel Pharmaceuticals Inc. | 1-(Benzo[b]thienyl)-2-(thienyl)ethenes and related compounds useful as anti-inflammatory agents |
| JPH02172956A (ja) | 1988-12-23 | 1990-07-04 | Sumitomo Pharmaceut Co Ltd | エリスロ―3―(3,4―ジヒドロキシフェニル)セリン誘導体の新規製造法 |
| ES2021557A6 (es) | 1990-07-23 | 1991-11-01 | Aliana S A | Procedimiento de obtencion de 4-hidroxi-metil-n-2-piridinil-2h-tieno (2,3 c)-1,2 tiazina-3 carboxamida 1,1 dioxido. |
| JPH03294253A (ja) | 1990-04-12 | 1991-12-25 | Shionogi & Co Ltd | 消化管ホルモン拮抗物質 |
| WO1997005865A1 (en) | 1995-08-08 | 1997-02-20 | Fibrogen, Inc. | C-proteinase inhibitors for the treatment of disorders related to the overproduction of collagen |
| JPH10298151A (ja) | 1997-04-30 | 1998-11-10 | Japan Energy Corp | C型肝炎ウイルスプロテアーゼ阻害剤 |
| WO2000020424A1 (fr) | 1998-10-07 | 2000-04-13 | Otsuka Kagaku Kabushiki Kaisha | PROCEDE DE PREPARATION D'ESTERS β-HYDROXY |
| US6057473A (en) | 1996-05-21 | 2000-05-02 | The Scripps Research Institute | Synthesis of aryl serines |
| JP2001046076A (ja) | 1999-08-13 | 2001-02-20 | Daicel Chem Ind Ltd | (r)−2−アミノ−1−フェニルエタノールまたはそのハロゲン置換体の製造方法 |
| GB2369117A (en) | 2000-11-17 | 2002-05-22 | Warner Lambert Co | Bombesin receptor antagonists |
| WO2002083111A2 (en) | 2001-04-16 | 2002-10-24 | Tanabe Seiyaku Co., Ltd. | Imidazole, thiazole and oxazole derivatives and their use for the manufacture of a medicament for the treatment and/or prevention of pollakiuria or urinary incontinence |
| WO2003066576A1 (de) | 2002-02-06 | 2003-08-14 | Basf Aktiengesellschaft | Phenylalaninderivate als herbizide |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IT1062206B (it) * | 1974-02-01 | 1983-09-20 | Rotta Research Lab S P A A S | Derivati della tirosina attivi sulla muscolatura liscia |
| JPS6098995A (ja) * | 1983-11-04 | 1985-06-01 | Microbial Chem Res Found | 光学活性3−(3,4−ジヒドロキシフエニル)セリン,およびその誘導体の製造法 |
| HU190864B (en) * | 1983-12-08 | 1986-11-28 | Alkaloida Vegyeszeti Gyar | Process for production of phenil-alanin derivatives |
| JPH075461B2 (ja) * | 1986-04-10 | 1995-01-25 | 三共株式会社 | 副作用の軽減されたペネム型またはカルバペネム型抗生物質製剤 |
| JPH08245552A (ja) * | 1995-03-08 | 1996-09-24 | Sumitomo Chem Co Ltd | ジアミノカルボン酸誘導体およびその製造法 |
| MY153569A (en) * | 1998-01-20 | 2015-02-27 | Mitsubishi Tanabe Pharma Corp | Inhibitors of ?4 mediated cell adhesion |
| US20040229955A1 (en) * | 2003-01-08 | 2004-11-18 | Andersen Niels H. | Antibacterial agents |
-
2004
- 2004-12-17 CN CNA2004800378537A patent/CN1894202A/zh active Pending
- 2004-12-17 BR BRPI0417780A patent/BRPI0417780B1/pt not_active IP Right Cessation
- 2004-12-17 US US10/581,444 patent/US7687435B2/en not_active Expired - Fee Related
- 2004-12-17 CA CA002548442A patent/CA2548442A1/en not_active Abandoned
- 2004-12-17 KR KR1020067011990A patent/KR20060123344A/ko not_active Application Discontinuation
- 2004-12-17 AR ARP040104766A patent/AR047334A1/es unknown
- 2004-12-17 JP JP2006544353A patent/JP4541363B2/ja not_active Expired - Fee Related
- 2004-12-17 EP EP04803999.4A patent/EP1697309B1/de not_active Not-in-force
- 2004-12-17 UY UY28683A patent/UY28683A1/es unknown
- 2004-12-17 MX MXPA06005989A patent/MXPA06005989A/es not_active Application Discontinuation
- 2004-12-17 WO PCT/EP2004/014392 patent/WO2005061443A2/de active Application Filing
- 2004-12-17 EA EA200601105A patent/EA200601105A1/ru unknown
- 2004-12-17 UA UAA200608125A patent/UA81568C2/xx unknown
- 2004-12-17 AU AU2004303492A patent/AU2004303492A1/en not_active Abandoned
-
2005
- 2005-01-03 PE PE2005000011A patent/PE20051058A1/es not_active Application Discontinuation
-
2006
- 2006-05-24 IL IL175884A patent/IL175884A0/en unknown
- 2006-06-13 CO CO06057507A patent/CO5700811A2/es not_active Application Discontinuation
- 2006-06-22 CR CR8475A patent/CR8475A/es not_active Application Discontinuation
- 2006-07-18 ZA ZA200605927A patent/ZA200605927B/en unknown
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4605759A (en) | 1983-08-01 | 1986-08-12 | Mitsui Toatsu Chemicals, Inc. | Process for production of β-phenylserine |
| US4810817A (en) | 1985-10-21 | 1989-03-07 | W. R. Grace & Co. | Aspartyl-beta substituted phenylalanine dipeptides |
| US4873359A (en) | 1985-10-21 | 1989-10-10 | W. R. Grace & Co. - Conn. | Process for preparing as partyl-phenylalanine dipeptides |
| JPH02172956A (ja) | 1988-12-23 | 1990-07-04 | Sumitomo Pharmaceut Co Ltd | エリスロ―3―(3,4―ジヒドロキシフェニル)セリン誘導体の新規製造法 |
| US4904674A (en) | 1989-08-16 | 1990-02-27 | Hoechst-Roussel Pharmaceuticals Inc. | 1-(Benzo[b]thienyl)-2-(thienyl)ethenes and related compounds useful as anti-inflammatory agents |
| JPH03294253A (ja) | 1990-04-12 | 1991-12-25 | Shionogi & Co Ltd | 消化管ホルモン拮抗物質 |
| ES2021557A6 (es) | 1990-07-23 | 1991-11-01 | Aliana S A | Procedimiento de obtencion de 4-hidroxi-metil-n-2-piridinil-2h-tieno (2,3 c)-1,2 tiazina-3 carboxamida 1,1 dioxido. |
| WO1997005865A1 (en) | 1995-08-08 | 1997-02-20 | Fibrogen, Inc. | C-proteinase inhibitors for the treatment of disorders related to the overproduction of collagen |
| US6057473A (en) | 1996-05-21 | 2000-05-02 | The Scripps Research Institute | Synthesis of aryl serines |
| JPH10298151A (ja) | 1997-04-30 | 1998-11-10 | Japan Energy Corp | C型肝炎ウイルスプロテアーゼ阻害剤 |
| WO2000020424A1 (fr) | 1998-10-07 | 2000-04-13 | Otsuka Kagaku Kabushiki Kaisha | PROCEDE DE PREPARATION D'ESTERS β-HYDROXY |
| JP2001046076A (ja) | 1999-08-13 | 2001-02-20 | Daicel Chem Ind Ltd | (r)−2−アミノ−1−フェニルエタノールまたはそのハロゲン置換体の製造方法 |
| GB2369117A (en) | 2000-11-17 | 2002-05-22 | Warner Lambert Co | Bombesin receptor antagonists |
| WO2002083111A2 (en) | 2001-04-16 | 2002-10-24 | Tanabe Seiyaku Co., Ltd. | Imidazole, thiazole and oxazole derivatives and their use for the manufacture of a medicament for the treatment and/or prevention of pollakiuria or urinary incontinence |
| WO2003066576A1 (de) | 2002-02-06 | 2003-08-14 | Basf Aktiengesellschaft | Phenylalaninderivate als herbizide |
Non-Patent Citations (88)
| Title |
|---|
| A. MANNSCHUK ET AL., ANGEW. CHEM., vol. 100, 1988, pages 299 |
| ALBRECHT, M. ET AL., SYNTHESIS, 2001, pages 468 - 472 |
| ALBRECHT, M. ET AL., SYNTHESIS, no. 3, 2001, pages 468 - 472 |
| ALKER, D. ET AL., TETRAHEDRON, vol. 54, no. 22, 1998, pages 6089 - 6098 |
| ARRAULT, A. ET AL., TETRAHEDRON LETT., vol. 43, no. 22, 2002, pages 4041 - 4044 |
| BERGMANN, E. D. ET AL., J CHEM SOC, 1951, pages 2673 |
| BEULSHAUSEN, T. ET AL., LIEBIGS ANN. CHEM., 1991, pages 1207 - 1209 |
| BLANK, S. ET AL., LIEBIGS ANN. CHEM., 1993, pages 889 - 896 |
| BLASER, D. ET AL., LIEBIGS ANN. CHEM., 1991, pages 1067 - 1078 |
| BOGER, D. L. ET AL., J. ORG. CHEM., vol. 62, no. 14, 1997, pages 4721 - 4736 |
| BOLHOFER, A. ET AL., J. AM. CHEM. SOC., vol. 75, 1953, pages 4469 |
| BOLHOFER, A., J. AM. CHEM. SOC., vol. 75, 1953, pages 4469 |
| BOTO, A ET AL., TETRAHEDRON LETTERS, vol. 39, no. 44, 1988, pages 8167 - 8170 |
| BOUKHRIS, S. ET AL., TETRAHEDRON LETT., vol. 40, no. 9, 1999, pages 1669 - 1672 |
| CADDICK, S., TETRAHEDRON, vol. 57, no. 30, 2001, pages 6615 - 6626 |
| CARRARA, G. ET AL., GAZZ. CHIM. ITAL., vol. 82, 1952, pages 325 |
| CASTRO, A. ET AL., J. ORG. CHEM., vol. 35, no. 8, 1970, pages 2815 - 2816 |
| CHANG, Y.-T. ET AL., J. AM. CHEM. SOC., vol. 75, 1953, pages 89 |
| CLARIANA, J. ET AL., TETRAHEDRON, vol. 55, no. 23, 1999, pages 7331 - 7344 |
| CLARK, J. E. ET AL., SYTHESIS, 1991, pages 891 - 894 |
| CLARK, J. E. ET AL., SYTHESIS, no. 10, 1991, pages 891 - 894 |
| COREY, E. J. ET AL., TETRAHEDRON LETT., vol. 32, no. 25, 1991, pages 2857 - 2860 |
| DAVIS, A. C. ET AL., J. CHEM. SOC., 1951, pages 3479 |
| DAVIS, C ET AL., J. CHEM. SOC., 1951, pages 3479 |
| DAVIS, F. A. ET AL., J. ORG. CHEM., vol. 59, no. 12, 1994, pages 3243 - 3245 |
| EWING W. ET AL., TETRAHEDRON LETT., vol. 30, no. 29, 1989, pages 3757 - 3760 |
| FUGANTI, C. ET AL., J. ORG. CHEM., vol. 51, no. 7, 1986, pages 1126 - 1128 |
| GELDERN, T. ET AL., J. OF MED. CHEM., vol. 39, no. 4, 1996, pages 957 - 967 |
| GIRARD A, TETRAHEDRON LETT., vol. 37, no. 44, 1996, pages 7967 - 7970 |
| GIRARD, A. ET AL., TETRAHEDRON LETT., vol. 37, no. 44, 1996, pages 7967 - 7970 |
| GUAN ET AL., J. COMB. CHEM., vol. 2, 2000, pages 297 |
| GUPTA, A. ET AL., J. CHEM. SOC. PERKIN TRANS., vol. 2, 1990, pages 1911 |
| H. PESSOA-MAHANA ET AL., SYNTH. COMM., vol. 32, 2002, pages 1437 |
| HELLMANN, H. ET AL., LIEBIGS ANN. CHEM., vol. 631, 1960, pages 175 - 179 |
| HERBERT, R. B. ET AL., CAN. J. CHEM., vol. 72, no. 1, 1994, pages 114 - 117 |
| HONIG, H. ET AL., TETRAHEDRON, 1990, pages 3841 |
| HUTTON, C. A., ORG. LETT., vol. 1, no. 2, 1999, pages 295 - 297 |
| HVIDT, T. ET AL., TETRAHEDRON LETT., vol. 27, no. 33, 1986, pages 3807 - 3810 |
| INOUE, H. ET AL., CHEM. PHAR. BULL., vol. 41, no. 9, 1993, pages 1521 - 1523 |
| IRIUCHIJIMA, S. ET AL., J. AM. CHEM. SOC., vol. 96, 1974, pages 4280 |
| ITO, S. ET AL., J. OF BIOL. CHEM., vol. 256, no. 15, 1981, pages 7834 - 4783 |
| J. M. ANDRES, TETRAHEDRON, vol. 56, 2000, pages 1523 |
| J.-F. ROUSSEAU ET AL., J. ORG. CHEM., vol. 63, 1998, pages 2731 - 2737 |
| JUNG, M. E. ET AL., SYNLETT, 1995, pages 563 - 564 |
| JUNG, M. E. ET AL., TETRAHEDRON LETT., vol. 30, no. 48, 1989, pages 6637 - 6640 |
| JURSIC, B. S. ET AL., SYNTH COMMUN, vol. 31, no. 4, 2001, pages 555 - 564 |
| KANEMASA, S. ET AL., TETRAHEDRON LETT., vol. 34, no. 4, 1993, pages 677 - 680 |
| KAWAHATA, N. H. ET AL., TETRAHEDRON LETT., vol. 43, no. 40, 2002, pages 7221 - 7223 |
| KIKUCHI, J. ET AL., CHEM. LETT., no. 3, 1993, pages 553 - 556 |
| KIM, . H. ET AL., TETRAHEDRON LETT., vol. 42, no. 48, 2001, pages 8401 - 8403 |
| KUWANO, R. ET AL., J. ORG .CHEM., vol. 63, no. 10, 1998, pages 3499 - 3503 |
| LEE, Y. ET AL., J. AM. CHEM. SOC., vol. 121, no. 36, 1999, pages 8407 - 8408 |
| LI, Y. Q. ET AL., TETRAHEDRON LETT., vol. 40, no. 51, 1999, pages 9097 - 9100 |
| LOOKER ET AL., J. ORG. CHEM., vol. 22, 1957, pages 1233 |
| LOWN, J. W. ET AL., CAN. J. CHEM., vol. 51, 1973, pages 856 |
| MAEDA, S ET AL., CHEM. & PHARM. BULL., vol. 32, no. 7, 1984, pages 2536 - 2543 |
| MARCHI, C. ET AL., TETRAHEDRON, vol. 58, no. 28, 2002, pages 5699 |
| MARTIN, S. F. ET AL., TETRAHEDRON LETT., vol. 39, no. 12, 1998, pages 1517 - 1520 |
| NICOLAOU, K. C. ET AL., ANGEW.N CHEM. INT. EDIT., vol. 37, no. 19, 1998, pages 2714 - 2716 |
| NICOLAOU, K. C. ET AL., J. AM. CHEM. SOC., vol. 124, no. 35, 2002, pages 10451 - 10455 |
| NOJORI R., J. AM. CHEM. SOC., vol. 111, no. 25, 1989, pages 9134 - 9135 |
| NOYORI, R. ET AL., J. AM. CHEM. SOC., vol. 111, no. 25, 1989, pages 9134 - 9135 |
| PARK, H. ET AL., J. ORG. CHEM., vol. 66, no. 21, 2001, pages 7223 - 7226 |
| PAULSEN, H. ET AL., LIEBIGS ANN. CHEM., 1987, pages 565 |
| PERICH, J. W.; JOHNS, R. B., J. ORG. CHEM., vol. 53, no. 17, 1988, pages 4103 - 4105 |
| POUPARDIN, O. ET AL., TETRAHEDRON LETT., vol. 42, no. 8, 2001, pages 1523 - 1526 |
| ROZENBERG, V. ET AL., ANGEW. CHEM., vol. 106, no. 1, 1994, pages 106 - 108 |
| SAEED, A. ET AL., TETRAHEDRON, vol. 48, no. 12, 1992, pages 2507 - 2514 |
| SCHMIDT U., SYNTHESIS, 1992, pages 1248 - 1254 |
| SHENGDE, W. ET AL., SYNTH. COMMUN., vol. 16, no. 12, 1986, pages 1479 |
| SINGH, J. ET AL., TETRAHEDRONLETTERS, vol. 34, no. 2, 1993, pages 211 - 214 |
| SOLLADIECAVALLO, A. ET AL., GAZZ. CHIM. ITAL., vol. 126, no. 3, 1996, pages 173 - 178 |
| SOLLADIECAVALLO, A. ET AL., TETRAHEDRON LETT., vol. 39, no. 15, 1998, pages 2191 - 2194 |
| SOLOSHONOK, V. A. ET AL., TETRAHEDRON LETT., vol. 35, no. 17, 1994, pages 2713 - 2716 |
| SOLOSHONOK, V. A. ET AL., TETRAHEDRON, vol. 52, no. 1, 1996, pages 245 - 254 |
| SOMLAI, C. ET AL., SYNTHESIS, 1992, pages 285 - 287 |
| SOUKUP, M. ET AL., HELV. CHIM. ACTA, vol. 70, 1987, pages 232 |
| SUZUKI, M. ET AL., J. ORG. CHEM., vol. 38, no. 20, 1973, pages 3571 - 3575 |
| SUZUKI, M. ET AL., SYNTHETIC COMMUNICATIONS, vol. 2, no. 4, 1972, pages 237 - 242 |
| TAKAHASHI, K. ET AL., J. ORG. CHEM., vol. 50, no. 18, 1985, pages 3414 - 3415 |
| TROAST, D. ET AL., ORG. LETT, vol. 4, no. 6, 2002, pages 991 - 994 |
| TROAST, D. ET AL., ORG. LETT., vol. 4, no. 6, 2002, pages 991 - 994 |
| VINOGRAD, L. ET AL., ZHURNAL ORGANICHESKOI KHIMII, vol. 16, no. 12, 1980, pages 2594 - 2599 |
| WU, S. D. ET AL., SYTHETIC COMMUN., vol. 16, no. 12, 1986, pages 1479 - 1484 |
| YADAV, L. D. S. ET AL., INDIAN J. CHEM B., vol. 41, no. 3, 2002, pages 593 - 595 |
| YOKOKAWA, F. ET AL., TETRAHEDRON LETT., vol. 42, no. 34, 2001, pages 5903 - 5908 |
| ZHDANKIN, V. V. ET AL., TETRAHEDRON LETT., vol. 41, no. 28, 2000, pages 5299 - 5302 |
| ZHOU, C. Y. ET AL., SYTHETIC COMMUN., vol. 17, no. 11, 1987, pages 1377 - 1382 |
Cited By (60)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006029828A1 (de) * | 2004-09-16 | 2006-03-23 | Basf Aktiengesellschaft | Benzoylsubstituierte serin-amide |
| WO2006029829A1 (de) * | 2004-09-16 | 2006-03-23 | Basf Aktiengesellschaft | Heteroaroylsubstituierte serin-amide als herbizide |
| EA013646B1 (ru) * | 2005-05-25 | 2010-06-30 | Басф Акциенгезельшафт | Замещённые бензоилом серин-амиды |
| WO2006125688A1 (de) * | 2005-05-25 | 2006-11-30 | Basf Aktiengesellschaft | Benzoylsubstituierte serin-amide |
| US7879761B2 (en) | 2005-05-25 | 2011-02-01 | Basf Aktiengesellschaft | Heteroaroyl-substituted serineamides |
| US7786046B2 (en) | 2005-05-25 | 2010-08-31 | Basf Aktiengesellschaft | Benzoyl-substituted serineamides |
| WO2007093539A2 (de) * | 2006-02-16 | 2007-08-23 | Basf Se | Benzoylsubstituierte alanine |
| WO2007093539A3 (de) * | 2006-02-16 | 2007-10-11 | Basf Ag | Benzoylsubstituierte alanine |
| JP2010515705A (ja) * | 2007-01-11 | 2010-05-13 | ビーエーエスエフ ソシエタス・ヨーロピア | ヘテロアロイル置換セリンアミド |
| WO2008084070A1 (de) * | 2007-01-11 | 2008-07-17 | Basf Se | Benzoylsubstituierte serin-amide |
| US8133851B2 (en) | 2007-01-11 | 2012-03-13 | Basf Se | Heteroaroyl-substituted serine amides |
| WO2008084073A1 (de) * | 2007-01-11 | 2008-07-17 | Basf Se | Heteroaroylsubstituierte serin-amide |
| US10888579B2 (en) | 2007-11-07 | 2021-01-12 | Beeologics Inc. | Compositions for conferring tolerance to viral disease in social insects, and the use thereof |
| WO2010049414A1 (en) * | 2008-10-31 | 2010-05-06 | Basf Se | Method for improving plant health |
| US10801028B2 (en) | 2009-10-14 | 2020-10-13 | Beeologics Inc. | Compositions for controlling Varroa mites in bees |
| US9121022B2 (en) | 2010-03-08 | 2015-09-01 | Monsanto Technology Llc | Method for controlling herbicide-resistant plants |
| US11812738B2 (en) | 2010-03-08 | 2023-11-14 | Monsanto Technology Llc | Polynucleotide molecules for gene regulation in plants |
| US9988634B2 (en) | 2010-03-08 | 2018-06-05 | Monsanto Technology Llc | Polynucleotide molecules for gene regulation in plants |
| EP3434780A1 (de) | 2011-09-13 | 2019-01-30 | Monsanto Technology LLC | Verfahren und zusammensetzungen zur unkrautbekämpfung |
| EP3382027A2 (de) | 2011-09-13 | 2018-10-03 | Monsanto Technology LLC | Verfahren und zusammensetzungen zur unkrautbekämpfung |
| US9422557B2 (en) | 2011-09-13 | 2016-08-23 | Monsanto Technology Llc | Methods and compositions for weed control |
| US9422558B2 (en) | 2011-09-13 | 2016-08-23 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013039990A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| US9416363B2 (en) | 2011-09-13 | 2016-08-16 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013040021A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10829828B2 (en) | 2011-09-13 | 2020-11-10 | Monsanto Technology Llc | Methods and compositions for weed control |
| EP3296402A2 (de) | 2011-09-13 | 2018-03-21 | Monsanto Technology LLC | Verfahren und zusammensetzungen zur unkrautbekämpfung |
| US10808249B2 (en) | 2011-09-13 | 2020-10-20 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10760086B2 (en) | 2011-09-13 | 2020-09-01 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013040117A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10806146B2 (en) | 2011-09-13 | 2020-10-20 | Monsanto Technology Llc | Methods and compositions for weed control |
| EP3434779A1 (de) | 2011-09-13 | 2019-01-30 | Monsanto Technology LLC | Verfahren und zusammensetzungen zur unkrautbekämpfung |
| US10240162B2 (en) | 2012-05-24 | 2019-03-26 | A.B. Seeds Ltd. | Compositions and methods for silencing gene expression |
| US10240161B2 (en) | 2012-05-24 | 2019-03-26 | A.B. Seeds Ltd. | Compositions and methods for silencing gene expression |
| US10934555B2 (en) | 2012-05-24 | 2021-03-02 | Monsanto Technology Llc | Compositions and methods for silencing gene expression |
| US10041068B2 (en) | 2013-01-01 | 2018-08-07 | A. B. Seeds Ltd. | Isolated dsRNA molecules and methods of using same for silencing target molecules of interest |
| US10683505B2 (en) | 2013-01-01 | 2020-06-16 | Monsanto Technology Llc | Methods of introducing dsRNA to plant seeds for modulating gene expression |
| US10609930B2 (en) | 2013-03-13 | 2020-04-07 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10612019B2 (en) | 2013-03-13 | 2020-04-07 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2014151255A1 (en) | 2013-03-15 | 2014-09-25 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10568328B2 (en) | 2013-03-15 | 2020-02-25 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10597676B2 (en) | 2013-07-19 | 2020-03-24 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US9856495B2 (en) | 2013-07-19 | 2018-01-02 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US11377667B2 (en) | 2013-07-19 | 2022-07-05 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US9777288B2 (en) | 2013-07-19 | 2017-10-03 | Monsanto Technology Llc | Compositions and methods for controlling leptinotarsa |
| US9850496B2 (en) | 2013-07-19 | 2017-12-26 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US10100306B2 (en) | 2013-11-04 | 2018-10-16 | Monsanto Technology Llc | Compositions and methods for controlling arthropod parasite and pest infestations |
| US9540642B2 (en) | 2013-11-04 | 2017-01-10 | The United States Of America, As Represented By The Secretary Of Agriculture | Compositions and methods for controlling arthropod parasite and pest infestations |
| US10927374B2 (en) | 2013-11-04 | 2021-02-23 | Monsanto Technology Llc | Compositions and methods for controlling arthropod parasite and pest infestations |
| US10557138B2 (en) | 2013-12-10 | 2020-02-11 | Beeologics, Inc. | Compositions and methods for virus control in Varroa mite and bees |
| US10334848B2 (en) | 2014-01-15 | 2019-07-02 | Monsanto Technology Llc | Methods and compositions for weed control using EPSPS polynucleotides |
| WO2015108982A2 (en) | 2014-01-15 | 2015-07-23 | Monsanto Technology Llc | Methods and compositions for weed control using epsps polynucleotides |
| US11091770B2 (en) | 2014-04-01 | 2021-08-17 | Monsanto Technology Llc | Compositions and methods for controlling insect pests |
| US10988764B2 (en) | 2014-06-23 | 2021-04-27 | Monsanto Technology Llc | Compositions and methods for regulating gene expression via RNA interference |
| US11807857B2 (en) | 2014-06-25 | 2023-11-07 | Monsanto Technology Llc | Methods and compositions for delivering nucleic acids to plant cells and regulating gene expression |
| US11124792B2 (en) | 2014-07-29 | 2021-09-21 | Monsanto Technology Llc | Compositions and methods for controlling insect pests |
| US10378012B2 (en) | 2014-07-29 | 2019-08-13 | Monsanto Technology Llc | Compositions and methods for controlling insect pests |
| US10968449B2 (en) | 2015-01-22 | 2021-04-06 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US10883103B2 (en) | 2015-06-02 | 2021-01-05 | Monsanto Technology Llc | Compositions and methods for delivery of a polynucleotide into a plant |
| US10655136B2 (en) | 2015-06-03 | 2020-05-19 | Monsanto Technology Llc | Methods and compositions for introducing nucleic acids into plants |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1697309A2 (de) | 2006-09-06 |
| CO5700811A2 (es) | 2006-11-30 |
| MXPA06005989A (es) | 2006-08-23 |
| EP1697309B1 (de) | 2014-04-23 |
| KR20060123344A (ko) | 2006-12-01 |
| JP2007534648A (ja) | 2007-11-29 |
| ZA200605927B (en) | 2008-05-28 |
| WO2005061443A3 (de) | 2005-12-22 |
| AR047334A1 (es) | 2006-01-18 |
| JP4541363B2 (ja) | 2010-09-08 |
| CA2548442A1 (en) | 2005-07-07 |
| CN1894202A (zh) | 2007-01-10 |
| BRPI0417780A (pt) | 2007-03-20 |
| PE20051058A1 (es) | 2006-01-19 |
| US7687435B2 (en) | 2010-03-30 |
| IL175884A0 (en) | 2006-10-05 |
| UY28683A1 (es) | 2005-08-31 |
| UA81568C2 (en) | 2008-01-10 |
| CR8475A (es) | 2006-12-07 |
| EA200601105A1 (ru) | 2006-12-29 |
| US20070142230A1 (en) | 2007-06-21 |
| BRPI0417780B1 (pt) | 2016-03-01 |
| AU2004303492A1 (en) | 2005-07-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7687435B2 (en) | Benzoyl-substituted phenylalanineamides | |
| EP1791825B1 (de) | Benzoylsubstituierte serin-amide | |
| US20070060480A1 (en) | Heteroaroyl-substituted phenylalanineamides | |
| US20070270312A1 (en) | Heteroaroyl-Substituted Serineamides | |
| EP0958292A1 (de) | 4-(3-heterocyclyl-1-benzoyl)pyrazole und ihre verwendung als herbizide | |
| EP1177194B1 (de) | 4-(3',4'-heterocyclylbenzoyl)pyrazole als herbizide | |
| EP1163240B1 (de) | Tricyclische benzoylpyrazol-derivate als herbizide | |
| EP1080089B1 (de) | Pyrazolyldioxothiochromanoyl-derivate | |
| EP1112256B1 (de) | Cyclohexenonchinolinoyl-derivate als herbizide mittel | |
| WO2000064912A1 (de) | Phosphorhaltige benzoyl-derivate und ihre verwendung als herbizide | |
| WO2000034270A1 (de) | Thiochromanoylpyrazolon-derivate | |
| WO2000059911A2 (de) | Tricyclische cyclohexandionderivate | |
| WO2000035903A2 (de) | Thiochromanoylcyclohexenon-derivate |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200480037853.7 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DPEN | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 175884 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2006/005989 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007142230 Country of ref document: US Ref document number: 10581444 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2548442 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004803999 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 06057507 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006544353 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020067011990 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004303492 Country of ref document: AU |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: CR2006-008475 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200601105 Country of ref document: EA |
|
| ENP | Entry into the national phase |
Ref document number: 2004303492 Country of ref document: AU Date of ref document: 20041217 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004303492 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006/05927 Country of ref document: ZA Ref document number: 200605927 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2637/CHENP/2006 Country of ref document: IN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004803999 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020067011990 Country of ref document: KR |
|
| ENP | Entry into the national phase |
Ref document number: PI0417780 Country of ref document: BR |
|
| WWP | Wipo information: published in national office |
Ref document number: 10581444 Country of ref document: US |