WO2004005276A1 - 1,3−ベンゾジオキソール−2−スピロシクロアルカン誘導体の製造法 - Google Patents
1,3−ベンゾジオキソール−2−スピロシクロアルカン誘導体の製造法 Download PDFInfo
- Publication number
- WO2004005276A1 WO2004005276A1 PCT/JP2003/008478 JP0308478W WO2004005276A1 WO 2004005276 A1 WO2004005276 A1 WO 2004005276A1 JP 0308478 W JP0308478 W JP 0308478W WO 2004005276 A1 WO2004005276 A1 WO 2004005276A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compound represented
- compound
- defined above
- substituted
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/06—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/357—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having two or more oxygen atoms in the same ring, e.g. crown ethers, guanadrel
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/02—Nasal agents, e.g. decongestants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/08—Bronchodilators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/16—Otologicals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/02—Non-specific cardiovascular stimulants, e.g. drugs for syncope, antihypotensives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/04—Inotropic agents, i.e. stimulants of cardiac contraction; Drugs for heart failure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C51/00—Preparation of carboxylic acids or their salts, halides or anhydrides
- C07C51/347—Preparation of carboxylic acids or their salts, halides or anhydrides by reactions not involving formation of carboxyl groups
- C07C51/377—Preparation of carboxylic acids or their salts, halides or anhydrides by reactions not involving formation of carboxyl groups by splitting-off hydrogen or functional groups; by hydrogenolysis of functional groups
Definitions
- the present invention has a phosphodiesterase (PDE) IV inhibitory effect, for example, inflammatory allergic diseases such as bronchial asthma, allergic rhinitis, nephritis, rheumatism, multiple sclerosis, Crohn's disease, psoriasis, systemic lupus erythema Autoimmune diseases such as Todes, depression, amnesia, central nervous system diseases such as dementia, heart failure, shock, visceral diseases associated with ischemia reperfusion caused by cerebral ischemic injury, etc.
- the present invention relates to a method for producing a 1,1-benzodioxol-2- (spirocycloaltin) derivative useful as a remedy for insulin-resistant diabetes, wound, AIDS and the like.
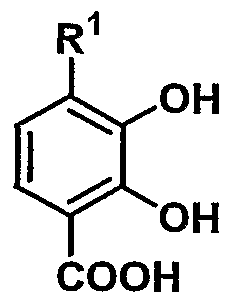
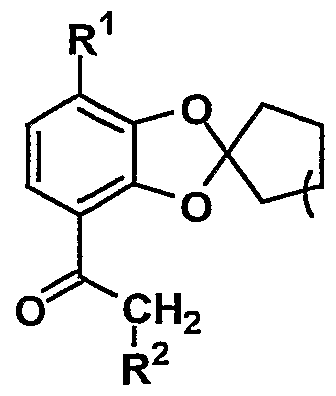
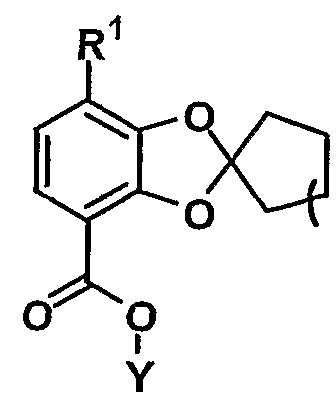
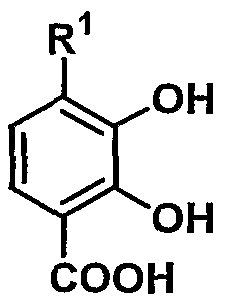
- R 1 represents hydroxy or substituted or unsubstituted lower alkoxy
- R 2 represents a substituted or unsubstituted aryl or a substituted or unsubstituted aromatic heterocyclic group
- n represents 1 to
- a compound containing a 1,3-benzodioxonore 1-2-spirocycloanolecan derivative represented by the following formula: has a PDE IV inhibitory effect, such as asthma, allergy, rheumatism, psoriasis, and cardiac muscle.
- An object of the present invention is to have a PDE IV inhibitory activity, for example, inflammatory allergic diseases such as bronchial asthma, allergic rhinitis, nephritis, rheumatism, multiple sclerosis, Crohn's disease, psoriasis, systemic lupus erythematosus, etc.
- inflammatory allergic diseases such as bronchial asthma, allergic rhinitis, nephritis, rheumatism, multiple sclerosis, Crohn's disease, psoriasis, systemic lupus erythematosus, etc.
- the present invention relates to the following (1) to (11).
- R 1 represents hydroxy or substituted or unsubstituted lower alkoxy
- R 1 and n are as defined above, and Y is lower alkyl, lower alkyl, lower alkynyl, substituted or unsubstituted aralkyl, substituted or unsubstituted aryl, or substituted or unsubstituted.
- R 2 represents a substituted or unsubstituted aryl or a substituted or unsubstituted aromatic heterocyclic group
- a base is added to the mixture, and a compound represented by the above formula (V) Wherein the compound represented by the formula (VI) is reacted with the compound represented by the formula (VI).
- n an integer of 1 to 6
- R 1 is as defined above, and the compound represented by the formula (II) is treated with hydrogen iodide.
- alkyl portion of lower alkyl and lower alkoxy examples include, for example, Straight-chain or branched alkyl having 1 to 10 carbon atoms, specifically methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butylinole, pentinole, neopentinole, hexinole , Heptinole, octyl, nonyl, decyl and the like.
- the lower alkenyl includes, for example, straight-chain or branched alkenyl having 2 to 8 carbon atoms, specifically, vinylol, aryl, 1-propenyl, butenyl, pentenyl, hexenyl, heptenyl, otathenyl And the like.
- the lower alkynyl includes, for example, straight-chain or branched C 2 to C 8 olenokininole, specifically, ethininole, propininole, buchenole, pentinole, hexinole, heptinole, and octinole. can give.
- aralkyl examples include aralkyl having 7 to 20 carbon atoms, specifically, benzyl, naphthylmethyl, triphenylmethyl and the like.
- aryl examples include aryl having 6 to 14 carbon atoms, specifically, phenyl, naphthyl, indenyl, and anthryl.
- aromatic heterocyclic group examples include a 5- or 6-membered monocyclic aromatic heterocyclic group containing at least one atom selected from a nitrogen atom, an oxygen atom and a sulfur atom, and 3 to 8 A condensed aromatic heterocyclic group containing at least one atom selected from a nitrogen atom, an oxygen atom and a sulfur atom, which is a bicyclic or tricyclic condensed member ring, and specifically, furyl.
- Substituents in the substituted lower alkoxy, substituted aralkyl, substituted aryl and substituted aromatic heterocyclic groups may be the same or different, for example, having 1 to 3 substitutions, lower alkynole, lower alkoxy, lower 'alkylthio, low Grade alkylamino, hydroxy, amino, halogen, nitro, cyano and the like.
- the lower alkyl moiety of the lower alkyl, lower alkoxy, lower alkylthio or lower alkylamino shown herein has the same meaning as the lower alkyl, and halogen means each atom of fluorine, chlorine, bromine and iodine.
- Compound (VI I) can be produced according to the following production method.
- Compound (VII) can be produced from compound (II) according to the following series of reaction steps.
- RR 2 , Y and n are as defined above, and X represents halogen, lower alkylsulfonyloxy or substituted or unsubstituted arylsulfonyloxy;
- the alkyl part of lower alkylsulfonyloxy and the aryl part of arylsulfonyloxy have the same meanings as the above-mentioned halogen, lower alkyl and aryl, respectively, and the substituent in substituted arylsulfuroxy is the above-mentioned substituted aryl. Is the same as the substituent of
- Compound (II) is a commercially available compound, or a known method [for example, Synthetic Communications, Vol. 16, p. 645 (1986), etc.] or a method similar thereto. Can be manufactured. Further, according to the method described in Production Method 2 described below, it is possible to produce more efficiently.
- Compound (III) can be produced by a commercially available method, or by a known method [for example, Synthesis (page 38) (1974) or the like] or a method analogous thereto.
- Compound (VI II) can be obtained as a commercial product, or can be obtained by a known method [for example, New Experimental Chemistry Course 14: Synthesis and Reaction of Organic Compounds (111), p. 1793 (1978)] or the like. Can be manufactured.
- Compound (VI) can be obtained as a commercially available product or a known method [eg, WO94 / 20455, Tetrahedron Lett., Vol. 37, p. 2565 (1996)], or It can be manufactured according to them.
- Compound (II) is used without solvent or in a solvent inert to the reaction, in an amount of 1 equivalent to a large excess, preferably 10 equivalents to 200 equivalents, of compound (III), usually at a temperature between 10 ° C and 150 ° C.
- the compound (IV) can be produced by reacting at a temperature of preferably 100 ° C to 150 ° C for 10 minutes to 48 hours.
- the solvent inert to the reaction is not particularly limited as long as it is a solvent inert to the reaction.
- aromatic solvents such as benzene, toluene, and xylene Hydrogen solvents
- aliphatic hydrocarbon solvents such as pentane, hexane, and cyclohexane, dichloromethane, dichloroethane, chlorophonolem, carbon tetrachloride, triphenylene, tonolen, dichlorobenzene, and cyclobenzene, Tetralin, dipheninoleether, dioxane, dimethoxetane, ethyleneglycoresinmethineoleatenore, getinoleatenore, diisopu Hexanone, methylethyl ketone, sulfolane, ethyl acetate, propyl acetate, butyl acetate, and the like, among which cyclopentan
- the compound (IV) obtained in the step 1 is combined with 1 to 10 equivalents, preferably 1 to 1.5 equivalents of the compound (VI II), in a solvent inert or in a solvent inert to the reaction, in an amount of 1 to ⁇ .
- the compound (V) is produced by reacting in the presence of 0 equivalent of a base, usually at a temperature between -10 ° C and 150 ° C, preferably at a temperature between 10 ° C and 70 ° C for 10 minutes to 48 hours. can do.
- Bases include, for example, triethylamine, triptylamin, disopropinoleetinoreamin, pyridine, 4-dimethinoreaminopyridine, 1,8-diazabicyclo [5.4.0] pindecene, 1,4 —Organic bases such as diazabicyclo [2.2.2] octane, N_methylmorpholine, sodium hydroxide, sodium hydroxide, lithium hydroxide, sodium hydroxide, magnesium hydroxide, hydroxide Cesium, sodium carbonate, potassium carbonate, cesium carbonate, calcium carbonate, magnesium carbonate, sodium bicarbonate, sodium bicarbonate, sodium phosphate, potassium phosphate, acetic acid Sodium, potassium acetate, potassium tert-butoxide, sodium tert-butoxide, sodium methoxide, sodium ethoxy Hydrogen Kana Application Benefits um, hydrogenated force Li um, inorganic bases and the like, such as calcium hydride, among others carbonate Na Application Benefits um,
- the solvent inert to the reaction is not particularly limited as long as it is a solvent inert to the reaction.
- aliphatic hydrocarbon solvents such as pentane, hexane, cyclohexane, benzene, toluene, xylene, etc.
- Aromatic hydrocarbon solvents tetralin, diphenyl ether, ethyl acetate, dichloromethane, black form, dichloroethane, carbon tetrachloride, trifluorotonolene, dichlorobenzene, black benzene , Pyridine, ethyl acetate, propynole acetate, butyl acetate, acetate, acetonitril, N, N-dimethylformamide, N, N-dimethylacetamide, 1-methyl1-2-pyro Ridon, 1, 3 —Dimethinoles 1 2 —Imidazolidinone, Dimethinoresnorelefoxide, Snoreholane, Dimethinoresnorehone, Teto Human mud furan, Jiokisan, dimethyl Tokisheta emissions, ethylene glycidyl Kozorejimechi ether, Jefferies Chino Les ether, diisopropyl Sopuro Pinor
- N N-dimethylformamide, N, N-dimethylacetamide, 1-methylino1-2-pyrrolidone, 1,3-dimethynole-1-N-imidazolidino, among others. And the like, and these can be used alone or in combination.
- any of the compounds (V) obtained in this step can be used in the next step.
- compound (V) is obtained as crystals
- compounds in which Y is n-butyl are more preferred. preferable.
- the solvent inert to the reaction is not particularly limited as long as it is inert to the reaction, and examples thereof include aliphatic hydrocarbon solvents such as pentane, hexane and cyclohexane, benzene, toluene, xylene and the like.
- Aromatic hydrocarbons Elemental solvents, tetralin, trifluorotoluene, benzene, diphenyl ether, pyridine, acetonitrile, N, N-dimethylformamide, N, N-dimethylacetamide, 1-methyl 1,2-pyrrolidone, 1,3-dimethyl_2-imidazolidinone, dimethylsulfoxide, ethynoleether, diisopropynoleatel, tetrahydrofuran, dioxane, dimethoxetane, ethylene glycol dimethyl ether Among them, getyl ether, tetrahydrofuran, hexane and the like are preferable, and these can be used alone or in combination.
- Examples of the base include n-butyllithium, sec-butyllithium, tert-butyllithium, lithium getylamide, lithium diisopropyl pyramide, lithium cyclohexinoleamide, lithium bis (trimethylsilyl) amide.
- Compound (II) are either obtained by the above-described good sea urchin commercially available or known methods, [for example Shinseti click 'grain Yunikesho lens (S ynt he tic Commun i cat i ons) N 16 Certificates, 645 pp. (1986) etc.] or according to them, but according to the following method, it is possible to produce more efficiently.
- the solvent is not particularly limited, but includes, for example, water, formic acid, acetic acid, propionic acid, butyric acid, trifluoroacetic acid, curoacetic acid, dichloroacetic acid, trichloroacetic acid, and acetonitride.
- Compound (I) can be obtained as a commercial product.
- the two methoxy groups at the 2-position and 3-position in compound (I) can be efficiently converted to a hydroxyl group with high selectivity.
- Compound (V) can also be produced by the following method.
- R Y and n are as defined above, Z represents halogen, and the halogen is as defined above
- Compound (IV) obtained in Step 1 of Production Method 1 is used without a solvent or in a solvent inert to the reaction with 1 equivalent to a large excess, preferably 1 equivalent to 10 equivalents of a halogenating agent, usually -10
- Compound (IX) can be obtained by treating at a temperature between ° C and 100 ° C for 10 minutes to 48 hours.
- Examples of the mouth-forming agent include thiol chloride, phosphorus oxychloride, phosphorus oxybromide, oxalyl dichloride, and phosgene.
- the solvent inert to the reaction is not particularly limited as long as it is inert to the reaction.
- examples thereof include aliphatic hydrocarbon solvents such as pentane, hexane and cyclohexane, benzene, toluene, and the like.
- Aromatic hydrocarbon solvents such as xylene, dichloromethane, dichloroethane, chlorophonolem, carbon tetrachloride, triphenylenelothenolene, dichlorobenzene, benzene, tetralamine, diphenylenoleatetenol, pyridin, ⁇ Cetonitrile, N, N-dimethylformamide, N, N-dimethylacetamide, 1-methyl-12-pyrrolidone, 1,3-dimethyl-2-imidazolidinone, getyl Ether, disopropynoleatene, tetrahydrofuran, dioxane, dimethoxetane, ethylene glycolone resin Ether, Mechiruechiruke tons, acetone tons, acetic Echiru, acetic propyl, acetic acid heptyl and the like, be used alone or as a mixture thereof it can.
- a catalyst amount to 10 equivalents, preferably 0.1 to 5 equivalents of a base such as pyridine or triethylamine, or Z, N, N-dimethylformamide or the like is added. May be.
- the solvent inert to the reaction is not particularly limited, as long as it is inert to the reaction.
- aliphatic hydrocarbon solvents such as pentane, hexane, and cyclohexane, benzene, toluene, and xylene Aromatic hydrocarbon solvents, such as dichloromethane, dichloroethane, chlorophonolem, carbon tetrachloride, triphenylene tonolenene, dichlorobenzene, cyclobenzene, tetralin, diphene Ninoleether, pyridine, acetotril, N, N-dimethylformamide, N, N-dimethinoreacetamide, 1-methyl_2-pyrrolidone, 1,3-dimethyl-2-imidazolidinone , Dimethyl sulfoxide, getyl ether, diisopropyl ether, tetrahydrofuran, dioxane, dimethoxet
- Examples of the base include pyridine, 4-dimethylaminopyridine, N, N-getylaniline, triethylamine, diisopropylethylamine, 1,8-diazabicyclo [5.4.0] indene, 1 , 4 diazabicyclo [2.2.2] octane, organic bases such as N-methinolemorpholine, sodium hydroxide, lithium hydroxide, lithium hydroxide, calcium hydroxide, magnesium hydroxide, hydroxide Cesium, sodium carbonate Lithium, calcium carbonate, cesium carbonate, calcium carbonate, magnesium carbonate, sodium bicarbonate, potassium bicarbonate, sodium phosphate, sodium phosphate, sodium acetate , Potassium acetate, potassium tert-butoxide, sodium tert-butoxide, sodium methoxide, sodium methoxide, sodium hydride, potassium hydride, And inorganic bases such as hydrogenated calcium.
- organic bases such as N-methinolemorpholine, sodium
- This step can also be performed subsequently without isolating the compound (IX) obtained in Step 5 in Step 5.
- Compound (IV) is mixed with 1 equivalent to a large excess, preferably 1 equivalent to 200 equivalents of compound (X) in the presence of 1 to 10 equivalents of a halogenating agent, in the absence of a solvent or a solvent inert to the reaction.
- the compound (V) can be produced by reacting the mixture at a temperature usually between -10 ° C and 100 ° C for 10 minutes to 48 hours.
- halogenating agent for example, thionyl chloride, phosphorus oxychloride, phosphorus oxybromide, oxalyl dichloride, phosgene and the like are used.
- the solvent inert to the reaction is not particularly limited as long as it is inert to the reaction.
- aliphatic hydrocarbon solvents such as pentane, hexane, and cyclohexane, benzene, toluene, and xylene
- Aromatic hydrocarbon solvents such as dichloromethane, dichloroethane, chlorophonolem, carbon tetrachloride, trifnoroleot / leene, dichlorobenzen, benzene benzene, tetralin, diphenylinoleether , Pyridine, acetonitrile, N, N-dimethinoleformamide, N, N-dimethylacetamide, 1-methyl-2-pyrrolidone, 1,3-dimethyl_2-imidazolidinone , Getinoleatenole, diisopropinoleatenole, tetrahydrofuran, dioxane, dim
- Step 8 Production of compound (V)
- Compound (IV) is used without a solvent or in a solvent inert to the reaction, in the presence of a catalytic amount to 10 equivalents, preferably 0.1 equivalent to 5 equivalents of a condensing agent, 1 equivalent to a large excess, preferably 1 equivalent.
- the compound (V) can be produced by reacting the compound (X) with an equivalent to 10 equivalents at a temperature usually between -10 ° C and 100 ° C for 10 minutes to 48 hours.
- an additive such as 1-hydroxybenzotriazole may be added.
- the solvent inert to the reaction is not particularly limited, as long as it is inert to the reaction.
- aliphatic hydrocarbon solvents such as pentane, hexane, and cyclohexane, benzene, toluene, and xylene
- Aromatic hydrocarbon solvents such as, dichloromethane, dichloroethane, chlorophorenolem, carbon tetrachloride, trifnoroleot / leene, dichlorobenzene, benzene, tetralamine, diphenylenethenole, pyridine , Astoni linole,
- condensing agent examples include condensing agents used for peptide synthesis. Examples thereof include N, N, and 1,2-dihexyl hexinole force / levodiimide, 1-ethyl-3- (3-dimethylaminopropyl) force rubodiimid. And the like.
- Intermediates and target compounds in the above-mentioned production methods can be isolated and purified by subjecting them to purification methods commonly used in organic synthetic chemistry, for example, filtration, extraction, washing, concentration, recrystallization, various types of chromatography, etc. .
- the intermediate can be subjected to the next reaction without purification.
- Step 3 (7-Methoxy-1,3, benzodioxol-2—spirocyclopentane) -1-4-Norevonic acid n—Ptinoleestenole
- the present invention has a PDE IV inhibitory effect, and has an inflammatory allergic disease such as bronchial asthma, allergic rhinitis, nephritis, rheumatism, multiple sclerosis, Crohn's disease, psoriasis, systemic lupus erythematosus, etc.
- Central nervous system diseases such as immune diseases, depression, amnesia, and dementia; organ diseases associated with ischemia reperfusion caused by heart failure, shock, cerebral ischemic injury, etc., and insulin resistant diabetes
- organ diseases associated with ischemia reperfusion caused by heart failure, shock, cerebral ischemic injury, etc. and insulin resistant diabetes
- insulin resistant diabetes Provided is a simple and large-scale method for producing 1,3-benzodioxo-l-2-spirocycloalkane derivatives and Z or intermediates thereof in a high yield, which is useful as a therapeutic agent for wounds, wounds, AIDS, etc. .
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Cardiology (AREA)
- Pulmonology (AREA)
- Heart & Thoracic Surgery (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Immunology (AREA)
- Diabetes (AREA)
- Dermatology (AREA)
- Epidemiology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Pain & Pain Management (AREA)
- Virology (AREA)
- Hospice & Palliative Care (AREA)
- Urology & Nephrology (AREA)
- Psychiatry (AREA)
- AIDS & HIV (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- Molecular Biology (AREA)
- Emergency Medicine (AREA)
- Endocrinology (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Tropical Medicine & Parasitology (AREA)
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP03762875A EP1535920A4 (en) | 2002-07-03 | 2003-07-03 | PROCESS FOR THE PREPARATION OF 1,3-BENZODIOXOLE-2-SPIRO-CYCLOALCAN DERIVATIVES |
| JP2004519253A JPWO2004005276A1 (ja) | 2002-07-03 | 2003-07-03 | 1,3−ベンゾジオキソール−2−スピロシクロアルカン誘導体の製造法 |
| AU2003252467A AU2003252467A1 (en) | 2002-07-03 | 2003-07-03 | Process for preparation of 1,3-benzodioxole-2-spiro- cycloalkane derivatives |
| CA002491464A CA2491464A1 (en) | 2002-07-03 | 2003-07-03 | Process for preparation of 1,3-benzodioxole-2-spiro- cycloalkane derivatives |
| US10/519,807 US20050245750A1 (en) | 2002-07-03 | 2003-07-03 | Process for preparing 1,3-benzodioxole-2-spirocycloalkane derivative |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2002-194273 | 2002-07-03 | ||
| JP2002194273 | 2002-07-03 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004005276A1 true WO2004005276A1 (ja) | 2004-01-15 |
Family
ID=30112298
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2003/008478 WO2004005276A1 (ja) | 2002-07-03 | 2003-07-03 | 1,3−ベンゾジオキソール−2−スピロシクロアルカン誘導体の製造法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20050245750A1 (ja) |
| EP (1) | EP1535920A4 (ja) |
| JP (1) | JPWO2004005276A1 (ja) |
| AU (1) | AU2003252467A1 (ja) |
| CA (1) | CA2491464A1 (ja) |
| WO (1) | WO2004005276A1 (ja) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004087149A1 (ja) * | 2003-03-31 | 2004-10-14 | Kyowa Hakko Kogyo Co. Ltd. | 医薬組成物 |
| WO2004087150A1 (ja) * | 2003-03-31 | 2004-10-14 | Kyowa Hakko Kogyo Co. Ltd. | 医薬組成物 |
| WO2004087147A1 (ja) * | 2003-03-31 | 2004-10-14 | Kyowa Hakko Kogyo Co., Ltd. | 肺疾患の治療および/または予防剤 |
| WO2004087151A1 (ja) * | 2003-03-31 | 2004-10-14 | Kyowa Hakko Kogyo Co., Ltd. | 医薬組成物 |
| WO2004087148A1 (ja) * | 2003-03-31 | 2004-10-14 | Kyowa Hakko Kogyo Co. Ltd. | 肺疾患の治療および/または予防剤 |
| WO2004096274A1 (ja) * | 2003-03-31 | 2004-11-11 | Kyowa Hakko Kogyo Co., Ltd. | 気道内投与剤 |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2012266514A1 (en) | 2011-06-06 | 2014-01-09 | Chiesi Farmaceutici S.P.A. | Derivatives of 1-phenyl-2-pyridinyl alkyl alcohols as phosphodiesterase inhibitors |
| PT3157930T (pt) * | 2014-06-23 | 2018-12-24 | Leo Pharma As | Métodos para a preparação de compostos heterocíclicos de 1,3-benzodioxole |
| RU2761337C2 (ru) | 2015-12-18 | 2021-12-07 | Юнион Терапьютикс А/С | Способы получения гетероциклических соединений 1, 3-бензодиоксола |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0771794A1 (en) * | 1995-05-19 | 1997-05-07 | Kyowa Hakko Kogyo Co., Ltd. | Oxygen-containing heterocyclic compounds |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6514996B2 (en) * | 1995-05-19 | 2003-02-04 | Kyowa Hakko Kogyo Co., Ltd. | Derivatives of benzofuran or benzodioxole |
-
2003
- 2003-07-03 JP JP2004519253A patent/JPWO2004005276A1/ja not_active Abandoned
- 2003-07-03 WO PCT/JP2003/008478 patent/WO2004005276A1/ja not_active Application Discontinuation
- 2003-07-03 US US10/519,807 patent/US20050245750A1/en not_active Abandoned
- 2003-07-03 CA CA002491464A patent/CA2491464A1/en not_active Abandoned
- 2003-07-03 EP EP03762875A patent/EP1535920A4/en not_active Withdrawn
- 2003-07-03 AU AU2003252467A patent/AU2003252467A1/en not_active Abandoned
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0771794A1 (en) * | 1995-05-19 | 1997-05-07 | Kyowa Hakko Kogyo Co., Ltd. | Oxygen-containing heterocyclic compounds |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004087149A1 (ja) * | 2003-03-31 | 2004-10-14 | Kyowa Hakko Kogyo Co. Ltd. | 医薬組成物 |
| WO2004087150A1 (ja) * | 2003-03-31 | 2004-10-14 | Kyowa Hakko Kogyo Co. Ltd. | 医薬組成物 |
| WO2004087147A1 (ja) * | 2003-03-31 | 2004-10-14 | Kyowa Hakko Kogyo Co., Ltd. | 肺疾患の治療および/または予防剤 |
| WO2004087151A1 (ja) * | 2003-03-31 | 2004-10-14 | Kyowa Hakko Kogyo Co., Ltd. | 医薬組成物 |
| WO2004087148A1 (ja) * | 2003-03-31 | 2004-10-14 | Kyowa Hakko Kogyo Co. Ltd. | 肺疾患の治療および/または予防剤 |
| WO2004096274A1 (ja) * | 2003-03-31 | 2004-11-11 | Kyowa Hakko Kogyo Co., Ltd. | 気道内投与剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20050245750A1 (en) | 2005-11-03 |
| AU2003252467A1 (en) | 2004-01-23 |
| EP1535920A4 (en) | 2007-03-21 |
| CA2491464A1 (en) | 2004-01-15 |
| EP1535920A1 (en) | 2005-06-01 |
| JPWO2004005276A1 (ja) | 2005-11-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6710642B2 (ja) | Hbv阻害剤としてのジヒドロピリミジン縮環誘導体 | |
| KR102240865B1 (ko) | Pde4 억제제의 제조 방법 | |
| JP4671104B2 (ja) | ピロロピリダジン誘導体 | |
| KR20090129479A (ko) | 화합물 2-히드록시-3-[5-(모르폴린-4-일메틸)피리딘-2-일]1h-인돌-5-카르보니트릴 701의 신규 제조 방법 | |
| JP2023027091A (ja) | アミノピリミジン誘導体を調製するための改善されたプロセス | |
| WO2004005276A1 (ja) | 1,3−ベンゾジオキソール−2−スピロシクロアルカン誘導体の製造法 | |
| CN112955426A (zh) | 制备光学富集的羟醛化合物的方法 | |
| JPH0640994A (ja) | 薬理活性のある新規テトラロン類 | |
| HU178354B (en) | Process for preparing 5-substituted 1,2-dihydro-3h-pyrrolo/1,2-a/-pyrrol-1-carboxylic acid derivatives from the corresponding nitriles | |
| Casaschi et al. | Palladium catalysed tandem cyclisation–anion capture. Part 7: Synthesis of derivatives of α-amino esters, nitrogen heterocycles and β-aryl/heteroaryl ethylamines via in situ generated vinylstannanes | |
| JP6174161B2 (ja) | 2−アミノニコチン酸ベンジルエステル誘導体の製造方法 | |
| FR2782515A1 (fr) | NOUVEAUX DERIVES DE L'INDANE-1-Ol, LEUR PROCEDE DE PREPARATION ET LES COMPOSITIONS PHARMACEUTIQUES QUI LES CONTIENNENT | |
| Fruit et al. | Syntheses and metalation of pyridazinecarboxamides and thiocarboxamides. Diazines. Part 32 | |
| JP2007099744A (ja) | テトラアザポルフィリン化合物の製造法 | |
| JP3880883B2 (ja) | ピリジン誘導体、その製造方法、及び除草剤中間体としての用途 | |
| JP4061333B2 (ja) | 2−(ピラゾール−1−イル)ピリジン誘導体 | |
| Ikemoto et al. | Unusual asymmetric oxidation of sulfide; the diastereoselective oxidation of prochiral sulfide-chiral acid salt with hydrogen peroxide without metal | |
| JP3066594B2 (ja) | アニリン誘導体及びその製造法 | |
| JP3224584B2 (ja) | 2,3−ジヒドロインドール−3,3−ジカルボン酸及び2,3−ジヒドロインドール−3−カルボン酸誘導体 | |
| JP4194856B2 (ja) | 2−フェニル−4−(1−ナフチル)イミダゾール | |
| CN116655520A (zh) | 6,6-二甲基-3-氮杂[3.1.0]己烷-2-羧酸类衍生物的制备方法 | |
| WO2009093463A1 (ja) | 神経細胞死抑制剤 | |
| WO2003045934A1 (en) | Process for producing 5-substituted 2(5h)-furanone | |
| JPH07278052A (ja) | 2,4−ペンタンジオン誘導体 | |
| JPH11246552A (ja) | アミン誘導体の製造法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2004519253 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10519807 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2491464 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003762875 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003762875 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2003762875 Country of ref document: EP |