KR20120028362A - 6?(1h?이미다졸?1?일)?2?페닐퀴나졸린의 결정 형태 - Google Patents
6?(1h?이미다졸?1?일)?2?페닐퀴나졸린의 결정 형태 Download PDFInfo
- Publication number
- KR20120028362A KR20120028362A KR1020127000282A KR20127000282A KR20120028362A KR 20120028362 A KR20120028362 A KR 20120028362A KR 1020127000282 A KR1020127000282 A KR 1020127000282A KR 20127000282 A KR20127000282 A KR 20127000282A KR 20120028362 A KR20120028362 A KR 20120028362A
- Authority
- KR
- South Korea
- Prior art keywords
- imidazol
- spectrum
- phenylquinazolin
- xrpd
- dsc thermogram
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- CSZGXYBGYFNSCO-UHFFFAOYSA-N 6-imidazol-1-yl-2-phenylquinazoline Chemical compound C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 CSZGXYBGYFNSCO-UHFFFAOYSA-N 0.000 title claims abstract description 104
- 239000013078 crystal Substances 0.000 claims abstract description 59
- 238000000034 method Methods 0.000 claims abstract description 48
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 21
- 150000001875 compounds Chemical class 0.000 claims abstract description 13
- 238000000634 powder X-ray diffraction Methods 0.000 claims description 123
- 239000000047 product Substances 0.000 claims description 78
- 239000000203 mixture Substances 0.000 claims description 72
- 238000001228 spectrum Methods 0.000 claims description 67
- 238000006243 chemical reaction Methods 0.000 claims description 59
- 239000000725 suspension Substances 0.000 claims description 56
- 238000001157 Fourier transform infrared spectrum Methods 0.000 claims description 51
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 51
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 claims description 48
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 44
- 238000003756 stirring Methods 0.000 claims description 44
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 claims description 43
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 29
- 238000010992 reflux Methods 0.000 claims description 29
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 claims description 26
- 238000002844 melting Methods 0.000 claims description 25
- 230000008018 melting Effects 0.000 claims description 25
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 claims description 24
- 239000002904 solvent Substances 0.000 claims description 23
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 claims description 22
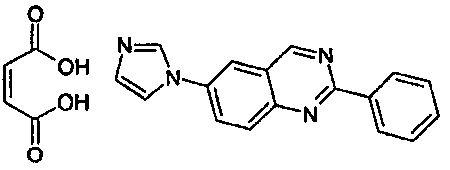
- CVRCPVCCSHDQPN-UHFFFAOYSA-N butanedioic acid;6-imidazol-1-yl-2-phenylquinazoline Chemical compound OC(=O)CCC(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 CVRCPVCCSHDQPN-UHFFFAOYSA-N 0.000 claims description 20
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 20
- 239000000243 solution Substances 0.000 claims description 19
- 230000000694 effects Effects 0.000 claims description 18
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 17
- QXFIIPBQSKGHJH-BTJKTKAUSA-N (z)-but-2-enedioic acid;6-imidazol-1-yl-2-phenylquinazoline Chemical compound OC(=O)\C=C/C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 QXFIIPBQSKGHJH-BTJKTKAUSA-N 0.000 claims description 14
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 claims description 14
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims description 14
- 238000002425 crystallisation Methods 0.000 claims description 13
- 230000008025 crystallization Effects 0.000 claims description 13
- 238000004519 manufacturing process Methods 0.000 claims description 13
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 claims description 12
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims description 12
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 claims description 12
- DOVFPQNKPMPDFJ-UHFFFAOYSA-N 6-imidazol-1-yl-2-phenylquinazoline;hydrate;dihydrochloride Chemical compound O.Cl.Cl.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 DOVFPQNKPMPDFJ-UHFFFAOYSA-N 0.000 claims description 10
- JMMWKPVZQRWMSS-UHFFFAOYSA-N isopropanol acetate Natural products CC(C)OC(C)=O JMMWKPVZQRWMSS-UHFFFAOYSA-N 0.000 claims description 10
- 229940011051 isopropyl acetate Drugs 0.000 claims description 10
- 230000008569 process Effects 0.000 claims description 10
- 238000011282 treatment Methods 0.000 claims description 10
- 230000007170 pathology Effects 0.000 claims description 9
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 claims description 7
- 239000000706 filtrate Substances 0.000 claims description 7
- 230000002757 inflammatory effect Effects 0.000 claims description 7
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 7
- QXFIIPBQSKGHJH-WLHGVMLRSA-N (e)-but-2-enedioic acid;6-imidazol-1-yl-2-phenylquinazoline Chemical compound OC(=O)\C=C\C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 QXFIIPBQSKGHJH-WLHGVMLRSA-N 0.000 claims description 6
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 claims description 6
- 208000002193 Pain Diseases 0.000 claims description 6
- 238000001914 filtration Methods 0.000 claims description 6
- DQSFYTNPZKLYPQ-UHFFFAOYSA-N 6-imidazol-1-yl-2-phenylquinazoline;phosphoric acid Chemical compound OP(O)(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 DQSFYTNPZKLYPQ-UHFFFAOYSA-N 0.000 claims description 5
- 238000000354 decomposition reaction Methods 0.000 claims description 5
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 5
- BUZVIRDRMZJESH-UHFFFAOYSA-N 6-imidazol-1-yl-2-phenylquinazoline;hydrochloride Chemical compound Cl.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 BUZVIRDRMZJESH-UHFFFAOYSA-N 0.000 claims description 4
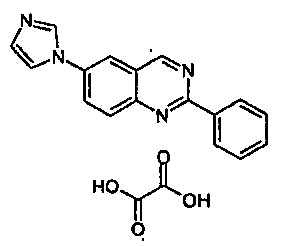
- MUGWMPDUWFYVOK-UHFFFAOYSA-N 6-imidazol-1-yl-2-phenylquinazoline;oxalic acid Chemical compound OC(=O)C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 MUGWMPDUWFYVOK-UHFFFAOYSA-N 0.000 claims description 4
- 238000001816 cooling Methods 0.000 claims description 4
- 238000001035 drying Methods 0.000 claims description 4
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 claims description 3
- 239000012296 anti-solvent Substances 0.000 claims description 3
- 230000001684 chronic effect Effects 0.000 claims description 3
- 238000004821 distillation Methods 0.000 claims description 3
- 208000000094 Chronic Pain Diseases 0.000 claims description 2
- 208000020401 Depressive disease Diseases 0.000 claims description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 claims description 2
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 claims description 2
- 208000005298 acute pain Diseases 0.000 claims description 2
- 239000012141 concentrate Substances 0.000 claims description 2
- 230000001747 exhibiting effect Effects 0.000 claims description 2
- 239000008101 lactose Substances 0.000 claims description 2
- 239000008096 xylene Substances 0.000 claims description 2
- 238000001938 differential scanning calorimetry curve Methods 0.000 claims 33
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 claims 2
- 229930195725 Mannitol Natural products 0.000 claims 2
- GWYFCOCPABKNJV-UHFFFAOYSA-N isovaleric acid Chemical compound CC(C)CC(O)=O GWYFCOCPABKNJV-UHFFFAOYSA-N 0.000 claims 2
- 239000000594 mannitol Substances 0.000 claims 2
- 235000010355 mannitol Nutrition 0.000 claims 2
- GSNUFIFRDBKVIE-UHFFFAOYSA-N DMF Natural products CC1=CC=C(C)O1 GSNUFIFRDBKVIE-UHFFFAOYSA-N 0.000 claims 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 claims 1
- 150000004683 dihydrates Chemical class 0.000 claims 1
- 239000000644 isotonic solution Substances 0.000 claims 1
- 238000010899 nucleation Methods 0.000 claims 1
- 229910052698 phosphorus Inorganic materials 0.000 claims 1
- 239000011574 phosphorus Substances 0.000 claims 1
- 229920000747 poly(lactic acid) Polymers 0.000 claims 1
- 239000004626 polylactic acid Substances 0.000 claims 1
- 150000003839 salts Chemical class 0.000 abstract description 38
- 239000012458 free base Substances 0.000 abstract description 22
- 239000012453 solvate Substances 0.000 abstract description 17
- 239000002585 base Substances 0.000 abstract description 8
- 150000004677 hydrates Chemical class 0.000 abstract description 5
- 230000001225 therapeutic effect Effects 0.000 abstract description 3
- 238000011170 pharmaceutical development Methods 0.000 abstract 1
- 238000002360 preparation method Methods 0.000 description 39
- 238000000113 differential scanning calorimetry Methods 0.000 description 23
- 238000002835 absorbance Methods 0.000 description 21
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 18
- 238000004090 dissolution Methods 0.000 description 16
- 239000000825 pharmaceutical preparation Substances 0.000 description 14
- 238000004458 analytical method Methods 0.000 description 13
- 235000019439 ethyl acetate Nutrition 0.000 description 13
- 229940126534 drug product Drugs 0.000 description 12
- 238000009472 formulation Methods 0.000 description 12
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 12
- KFNPURNIZVXVPJ-UHFFFAOYSA-N 6-imidazol-1-yl-2-phenylquinazoline;hydrate Chemical compound O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 KFNPURNIZVXVPJ-UHFFFAOYSA-N 0.000 description 11
- 239000004480 active ingredient Substances 0.000 description 11
- 238000003801 milling Methods 0.000 description 11
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 10
- 150000003890 succinate salts Chemical group 0.000 description 10
- 150000003892 tartrate salts Chemical group 0.000 description 10
- 238000010521 absorption reaction Methods 0.000 description 9
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 9
- 230000004580 weight loss Effects 0.000 description 9
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 8
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 8
- 230000008859 change Effects 0.000 description 8
- 125000002962 imidazol-1-yl group Chemical group [*]N1C([H])=NC([H])=C1[H] 0.000 description 8
- 238000005481 NMR spectroscopy Methods 0.000 description 7
- 239000007864 aqueous solution Substances 0.000 description 7
- 239000002775 capsule Substances 0.000 description 7
- GWYFCOCPABKNJV-UHFFFAOYSA-M isovalerate Chemical compound CC(C)CC([O-])=O GWYFCOCPABKNJV-UHFFFAOYSA-M 0.000 description 7
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical group OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 6
- BRUUBZYPEBMPOD-KMKQWAGMSA-N O.O.OC(=O)[C@H](O)[C@@H](O)C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 Chemical compound O.O.OC(=O)[C@H](O)[C@@H](O)C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 BRUUBZYPEBMPOD-KMKQWAGMSA-N 0.000 description 6
- URLKBWYHVLBVBO-UHFFFAOYSA-N Para-Xylene Chemical group CC1=CC=C(C)C=C1 URLKBWYHVLBVBO-UHFFFAOYSA-N 0.000 description 6
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 238000010438 heat treatment Methods 0.000 description 6
- 239000011976 maleic acid Substances 0.000 description 6
- 239000000463 material Substances 0.000 description 6
- 239000000843 powder Substances 0.000 description 6
- 239000007787 solid Substances 0.000 description 6
- FGQWBAYKGKVXRA-LREBCSMRSA-N (2r,3r)-2,3-dihydroxybutanedioic acid;6-imidazol-1-yl-2-phenylquinazoline Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 FGQWBAYKGKVXRA-LREBCSMRSA-N 0.000 description 5
- 238000005160 1H NMR spectroscopy Methods 0.000 description 5
- -1 2-phenylquinazoline succinate Chemical compound 0.000 description 5
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 5
- 241000700159 Rattus Species 0.000 description 5
- 230000035699 permeability Effects 0.000 description 5
- 229940095064 tartrate Drugs 0.000 description 5
- 238000002411 thermogravimetry Methods 0.000 description 5
- QCQCHGYLTSGIGX-GHXANHINSA-N 4-[[(3ar,5ar,5br,7ar,9s,11ar,11br,13as)-5a,5b,8,8,11a-pentamethyl-3a-[(5-methylpyridine-3-carbonyl)amino]-2-oxo-1-propan-2-yl-4,5,6,7,7a,9,10,11,11b,12,13,13a-dodecahydro-3h-cyclopenta[a]chrysen-9-yl]oxy]-2,2-dimethyl-4-oxobutanoic acid Chemical group N([C@@]12CC[C@@]3(C)[C@]4(C)CC[C@H]5C(C)(C)[C@@H](OC(=O)CC(C)(C)C(O)=O)CC[C@]5(C)[C@H]4CC[C@@H]3C1=C(C(C2)=O)C(C)C)C(=O)C1=CN=CC(C)=C1 QCQCHGYLTSGIGX-GHXANHINSA-N 0.000 description 4
- 230000000202 analgesic effect Effects 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 229910052757 nitrogen Inorganic materials 0.000 description 4
- 239000008363 phosphate buffer Substances 0.000 description 4
- RAXXELZNTBOGNW-UHFFFAOYSA-N 1H-imidazole Chemical compound C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 3
- BFUHAIPOEFXKSY-UACZKNEHSA-N O.OC(=O)[C@H](O)[C@@H](O)C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 Chemical compound O.OC(=O)[C@H](O)[C@@H](O)C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 BFUHAIPOEFXKSY-UACZKNEHSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- 238000009835 boiling Methods 0.000 description 3
- 238000012512 characterization method Methods 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 239000002270 dispersing agent Substances 0.000 description 3
- 210000001035 gastrointestinal tract Anatomy 0.000 description 3
- 239000012729 immediate-release (IR) formulation Substances 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 230000007935 neutral effect Effects 0.000 description 3
- 150000003891 oxalate salts Chemical group 0.000 description 3
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 3
- 229920000053 polysorbate 80 Polymers 0.000 description 3
- 238000012552 review Methods 0.000 description 3
- 235000012976 tarts Nutrition 0.000 description 3
- VDDAVZWCRBHDLQ-UHFFFAOYSA-N 2-phenylquinazoline Chemical compound C1=CC=CC=C1C1=NC=C(C=CC=C2)C2=N1 VDDAVZWCRBHDLQ-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 208000000112 Myalgia Diseases 0.000 description 2
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 229910019142 PO4 Inorganic materials 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical group OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 2
- 239000008186 active pharmaceutical agent Substances 0.000 description 2
- 230000001430 anti-depressive effect Effects 0.000 description 2
- 230000003110 anti-inflammatory effect Effects 0.000 description 2
- 239000000935 antidepressant agent Substances 0.000 description 2
- 229940005513 antidepressants Drugs 0.000 description 2
- 229940027991 antiseptic and disinfectant quinoline derivative Drugs 0.000 description 2
- 238000004807 desolvation Methods 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 238000001704 evaporation Methods 0.000 description 2
- 230000008020 evaporation Effects 0.000 description 2
- 230000004927 fusion Effects 0.000 description 2
- 230000002496 gastric effect Effects 0.000 description 2
- 239000010439 graphite Substances 0.000 description 2
- 229910002804 graphite Inorganic materials 0.000 description 2
- 238000002329 infrared spectrum Methods 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 229940049920 malate Drugs 0.000 description 2
- BJEPYKJPYRNKOW-UHFFFAOYSA-L malate(2-) Chemical compound [O-]C(=O)C(O)CC([O-])=O BJEPYKJPYRNKOW-UHFFFAOYSA-L 0.000 description 2
- 150000002688 maleic acid derivatives Chemical class 0.000 description 2
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 2
- 239000000155 melt Substances 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 208000013465 muscle pain Diseases 0.000 description 2
- 239000002547 new drug Substances 0.000 description 2
- 229940126701 oral medication Drugs 0.000 description 2
- 230000036407 pain Effects 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 2
- 239000010452 phosphate Substances 0.000 description 2
- 230000002980 postoperative effect Effects 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 229910052594 sapphire Inorganic materials 0.000 description 2
- 239000010980 sapphire Substances 0.000 description 2
- 230000035945 sensitivity Effects 0.000 description 2
- 238000001179 sorption measurement Methods 0.000 description 2
- 238000004611 spectroscopical analysis Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- KDYFGRWQOYBRFD-UHFFFAOYSA-N succinic acid Chemical compound OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 2
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- RNLYVRKMBPOHNI-UNDPIUDWSA-N (2s)-5-(diaminomethylideneamino)-n-[11-[4-[4-[4-[11-[[2-[4-[(2r)-2-hydroxypropyl]triazol-1-yl]acetyl]amino]undecanoyl]piperazin-1-yl]-6-[2-[2-(2-prop-2-ynoxyethoxy)ethoxy]ethylamino]-1,3,5-triazin-2-yl]piperazin-1-yl]-11-oxoundecyl]-2-[4-[(2s)-2-methylbut Chemical compound N1=NC(C[C@@H](C)CC)=CN1[C@@H](CCCN=C(N)N)C(=O)NCCCCCCCCCCC(=O)N1CCN(C=2N=C(N=C(NCCOCCOCCOCC#C)N=2)N2CCN(CC2)C(=O)CCCCCCCCCCNC(=O)CN2N=NC(C[C@@H](C)O)=C2)CC1 RNLYVRKMBPOHNI-UNDPIUDWSA-N 0.000 description 1
- MMCRTZBSRCXUJW-WLHGVMLRSA-N (e)-but-2-enedioic acid;quinazoline Chemical compound OC(=O)\C=C\C(O)=O.N1=CN=CC2=CC=CC=C21 MMCRTZBSRCXUJW-WLHGVMLRSA-N 0.000 description 1
- BRUUBZYPEBMPOD-UHFFFAOYSA-N 2,3-dihydroxybutanedioic acid;6-imidazol-1-yl-2-phenylquinazoline;dihydrate Chemical compound O.O.OC(=O)C(O)C(O)C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 BRUUBZYPEBMPOD-UHFFFAOYSA-N 0.000 description 1
- BFUHAIPOEFXKSY-UHFFFAOYSA-N 2,3-dihydroxybutanedioic acid;6-imidazol-1-yl-2-phenylquinazoline;hydrate Chemical compound O.OC(=O)C(O)C(O)C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 BFUHAIPOEFXKSY-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- YHLPVASKTHWKMQ-UHFFFAOYSA-N C(C=C1)C=CN1c(cc1)cc2c1nc(-c1ccccc1)nc2 Chemical compound C(C=C1)C=CN1c(cc1)cc2c1nc(-c1ccccc1)nc2 YHLPVASKTHWKMQ-UHFFFAOYSA-N 0.000 description 1
- 206010058019 Cancer Pain Diseases 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 206010009900 Colitis ulcerative Diseases 0.000 description 1
- 208000011231 Crohn disease Diseases 0.000 description 1
- 201000004624 Dermatitis Diseases 0.000 description 1
- 206010015150 Erythema Diseases 0.000 description 1
- 238000004566 IR spectroscopy Methods 0.000 description 1
- 102000009032 Imidazoline Receptors Human genes 0.000 description 1
- 108010049134 Imidazoline Receptors Proteins 0.000 description 1
- 102000010909 Monoamine Oxidase Human genes 0.000 description 1
- 108010062431 Monoamine oxidase Proteins 0.000 description 1
- CICSGUAUMYMOAO-ILKQHERXSA-N O.O.O.O.OC(=O)[C@H](O)[C@@H](O)C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 Chemical compound O.O.O.O.OC(=O)[C@H](O)[C@@H](O)C(O)=O.C1=NC=CN1C1=CC=C(N=C(N=C2)C=3C=CC=CC=3)C2=C1 CICSGUAUMYMOAO-ILKQHERXSA-N 0.000 description 1
- YZXJRJYHAXYNAK-QNSVNVJESA-N O[C@H](C(/C(/O)=[O]/c1cc2nc(-c3ccccc3)ncc2cc1-[n]1cncc1)O)C(O)=O Chemical compound O[C@H](C(/C(/O)=[O]/c1cc2nc(-c3ccccc3)ncc2cc1-[n]1cncc1)O)C(O)=O YZXJRJYHAXYNAK-QNSVNVJESA-N 0.000 description 1
- 206010065016 Post-traumatic pain Diseases 0.000 description 1
- 208000004550 Postoperative Pain Diseases 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 201000004681 Psoriasis Diseases 0.000 description 1
- 238000001069 Raman spectroscopy Methods 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 201000006704 Ulcerative Colitis Diseases 0.000 description 1
- 238000002441 X-ray diffraction Methods 0.000 description 1
- 238000000441 X-ray spectroscopy Methods 0.000 description 1
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 229940035676 analgesics Drugs 0.000 description 1
- 239000000730 antalgic agent Substances 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 239000008135 aqueous vehicle Substances 0.000 description 1
- 208000010668 atopic eczema Diseases 0.000 description 1
- 238000005102 attenuated total reflection Methods 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 229960000074 biopharmaceutical Drugs 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 238000007707 calorimetry Methods 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 230000001427 coherent effect Effects 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 235000008504 concentrate Nutrition 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- PCHPORCSPXIHLZ-UHFFFAOYSA-N diphenhydramine hydrochloride Chemical compound [Cl-].C=1C=CC=CC=1C(OCC[NH+](C)C)C1=CC=CC=C1 PCHPORCSPXIHLZ-UHFFFAOYSA-N 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 229940088679 drug related substance Drugs 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 231100000321 erythema Toxicity 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 238000005469 granulation Methods 0.000 description 1
- 230000003179 granulation Effects 0.000 description 1
- 238000000589 high-performance liquid chromatography-mass spectrometry Methods 0.000 description 1
- 230000036571 hydration Effects 0.000 description 1
- 238000006703 hydration reaction Methods 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- 229960003943 hypromellose Drugs 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 239000007972 injectable composition Substances 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 206010025135 lupus erythematosus Diseases 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 150000004682 monohydrates Chemical class 0.000 description 1
- 208000004296 neuralgia Diseases 0.000 description 1
- 208000021722 neuropathic pain Diseases 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- LYGJENNIWJXYER-UHFFFAOYSA-N nitromethane Chemical compound C[N+]([O-])=O LYGJENNIWJXYER-UHFFFAOYSA-N 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 201000008482 osteoarthritis Diseases 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 229940127557 pharmaceutical product Drugs 0.000 description 1
- 239000002831 pharmacologic agent Substances 0.000 description 1
- 238000011458 pharmacological treatment Methods 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 102000054765 polymorphisms of proteins Human genes 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 210000002345 respiratory system Anatomy 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000004467 single crystal X-ray diffraction Methods 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 238000009987 spinning Methods 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 239000001384 succinic acid Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 238000002460 vibrational spectroscopy Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/54—1,3-Diazines; Hydrogenated 1,3-diazines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/517—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with carbocyclic ring systems, e.g. quinazoline, perimidine
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pain & Pain Management (AREA)
- Neurosurgery (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Neurology (AREA)
- Biomedical Technology (AREA)
- Pest Control & Pesticides (AREA)
- Agronomy & Crop Science (AREA)
- Plant Pathology (AREA)
- Dentistry (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Epidemiology (AREA)
- Rheumatology (AREA)
- Psychiatry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
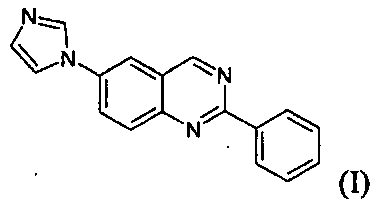
화학식(I)의 화합물은 5 가지의 상이한 결정 형태, 세 가지의 다형체 형태 및 두 가지의 수화물로 유리 염기로서 얻을 수 있다. 약제학적 개발을 위해서 허용되는 물리화학적 특성을 지니는 화학식(I)의 화합물의 일부 약제학적으로 허용되는 염이 얻어졌으며, 각각의 다형체 형태 및/또는 수화물이 특성화된다.
Description
Claims (27)
- 다음 특징:
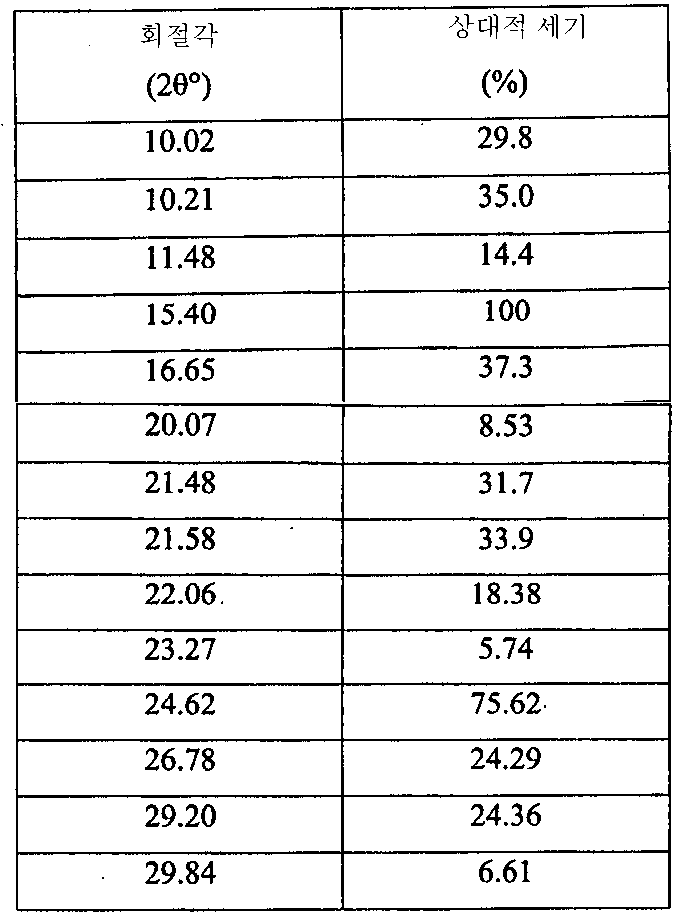
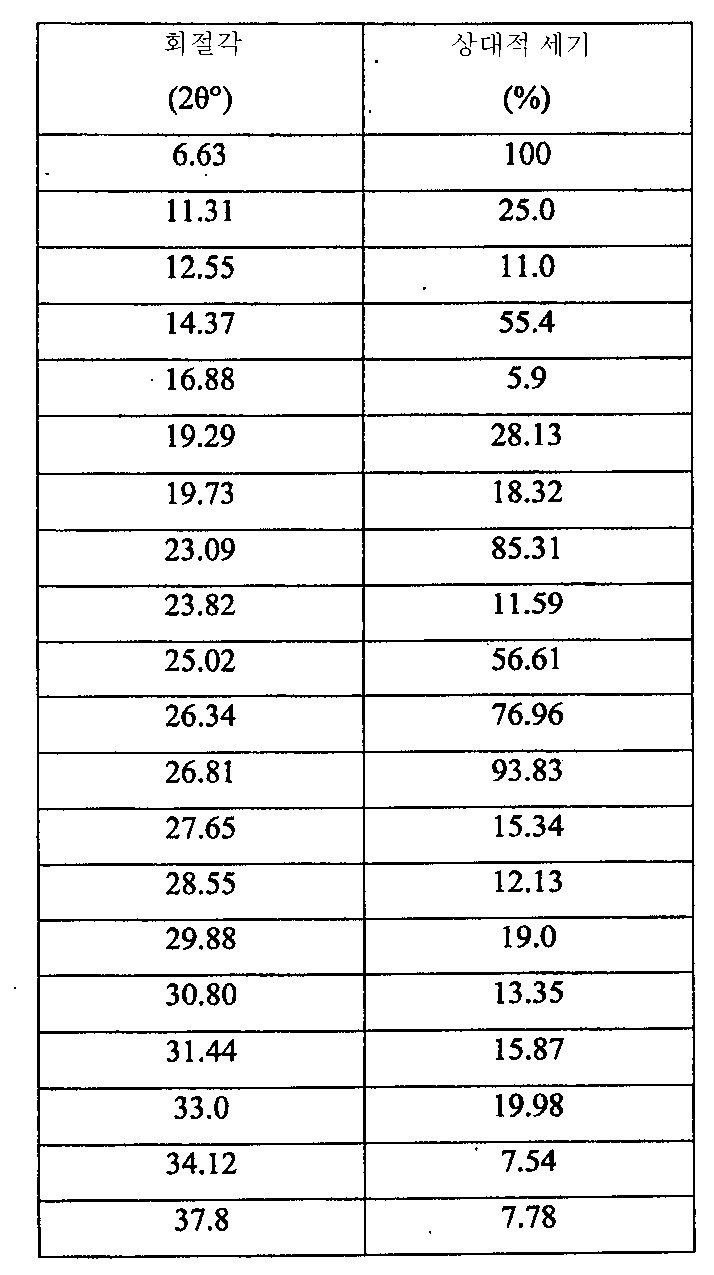
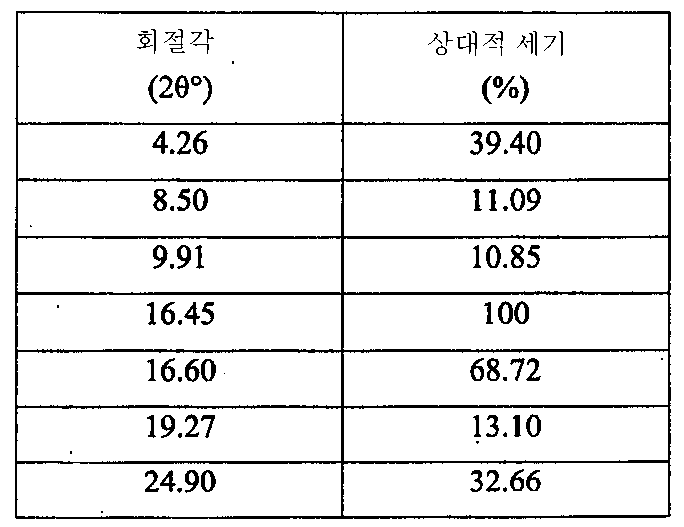
i) 도 1에 도시된 바와 같고/거나 표 1에 열거된 주 피크(main peak)를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 약 180℃에서 개시되는 융점 및 도 2에 도시된 DSC 온도기록도와 실질적으로 일치하는 DSC 온도기록도;
iii) 도 3에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;
중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린의 다형체 형태. - 다음 특징:
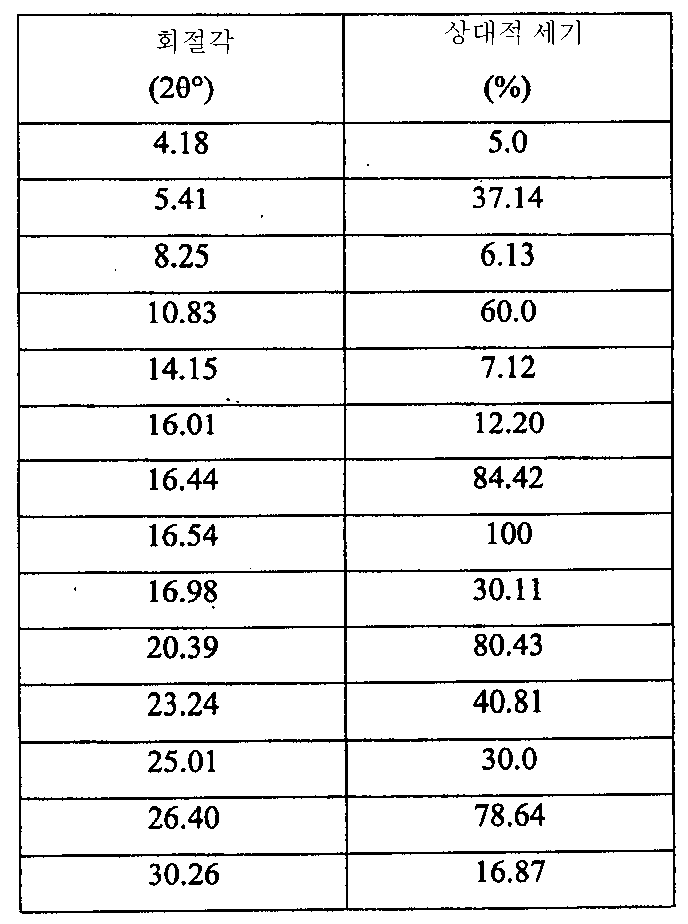
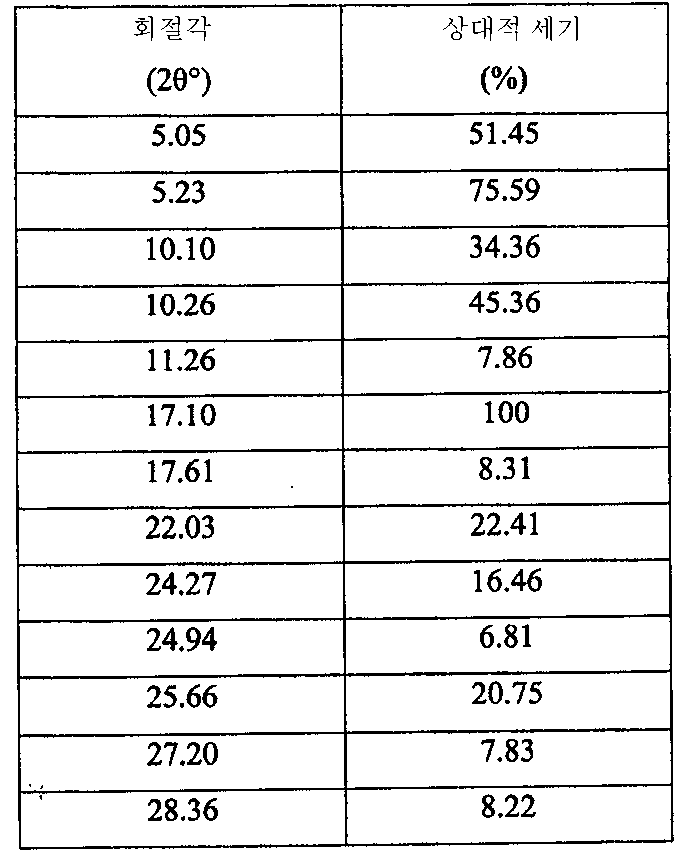
i) 도 4에 도시된 바와 같고/거나 표 2에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 약 160℃에서 개시되는 융점 및 도 5에 도시된 DSC 온도기록도와 실질적으로 일치하는 DSC 온도기록도;
iii) 도 6에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;
중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린의 다형체 형태. - 다음 특징:
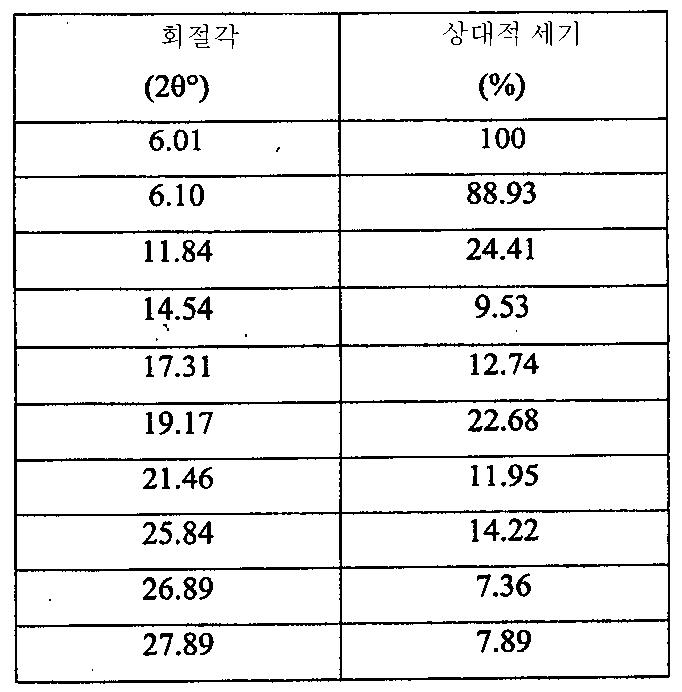
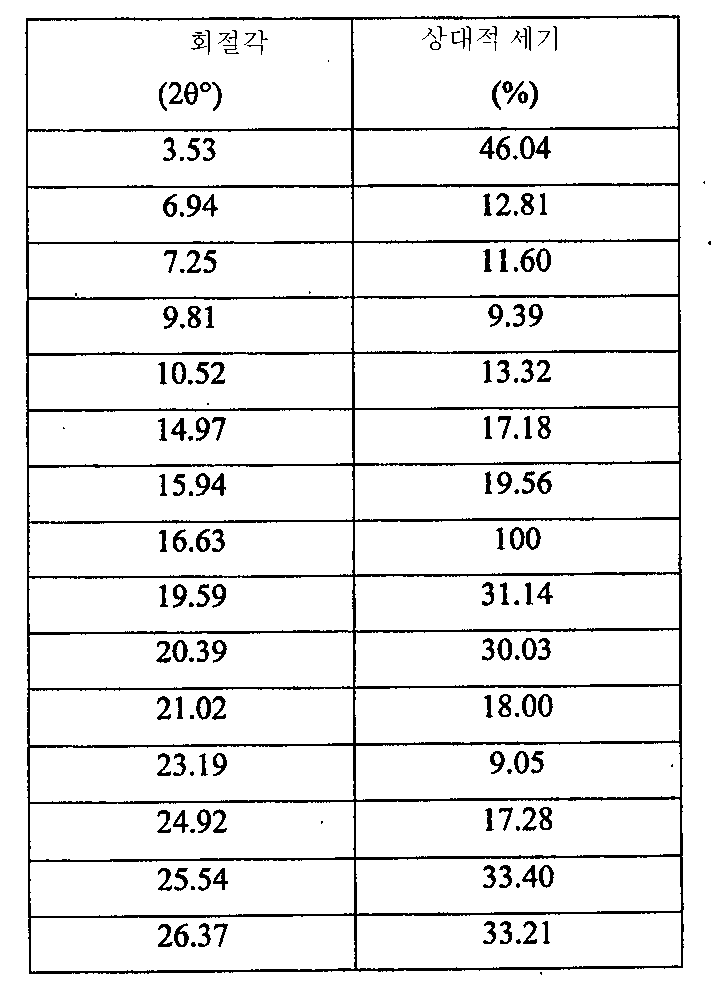
i) 도 7에 도시된 바와 같고/거나 표 3에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 약 162℃에서 개시되는 융점 및 도 8에 도시된 DSC 온도기록도와 실질적으로 일치하는 DSC 온도기록도;
iii) 도 9에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;
중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린의 다형체 형태. - 다음 특징:
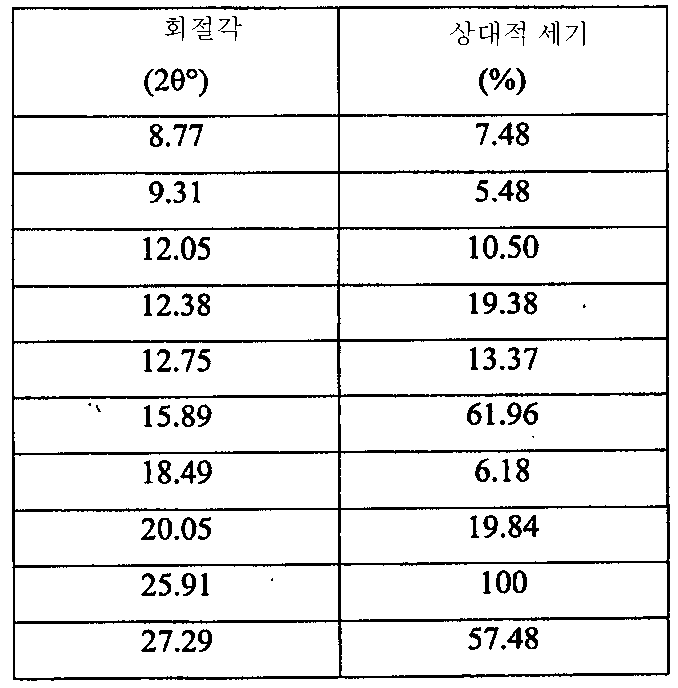
i) 도 10에 도시된 바와 같고/거나 표 4에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 약 40 내지 100℃의 범위에서 흡열 반응, 약 156℃에서 개시되는 흡열 반응, 약 178℃에서 개시되는 흡열 반응을 나타내는 도 11에 도시된 DSC 온도기록도와 실질적으로 일치하는 DSC 온도기록도;
iii) 도 12에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;
중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린의 수화된 형태. - 다음 특징:
i) 도 13에 도시된 바와 같고/거나 표 5에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 약 30 내지 80℃의 범위에서 흡열 반응, 약 163℃에서 개시되는 흡열 반응, 약 179℃에서 개시되는 흡열 반응을 나타내는 도 14에 도시된 DSC 온도기록도와 실질적으로 일치하는 DSC 온도기록도;
iii) 도 15에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;
중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린의 수화된 형태. - 다음 특징:
i) 도 17에 도시된 바와 같고/거나 표 6에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 약 144℃에서 개시되는 융점 및 도 18의 영역 130 내지 280℃와 일치하는 DSC 온도기록도;
iii) 도 19a에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;
중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린의 수화된 형태. - 다음 특징:
i) 도 20에 도시된 바와 같고/거나 표 7에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 약 240℃에서 분해와 함께 나타나는 융점;
중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 하이드로클로라이드의 결정 형태. - 셀 파라미터 a = 8.0152 (6) Å, b = 5.9038 (4) Å, c = 33.127 (3) Å, α: 90°, β = 93.280° (8), γ = 90°, V = 1565.0 (2) Å3, 공간 그룹(space group) P21/c을 나타내는 단사정계가 특징이며, 다음 특징:
i) 도 21a에 도시된 바와 같고/거나 표 8에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 22에 도시된 DSC 온도기록도와 일치되게, 약 150℃에서 개시되는 흡열 효과 및 약 183℃에서 개시되는 제 2 흡열 효과를 나타내는 DSC 온도기록도;
iii) 도 23에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 추가의 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 석시네이트의 결정 형태. - 다음 특징:
i) 도 24에 도시된 바와 같고/거나 표 9에 열거된 주 피크(main peak)를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 25에 도시된 DSC 온도기록도와 일치되게, 약 108℃에서 개시되는 흡열 효과 및 약 181℃에서 개시되는 제 2 흡열 효과를 나타내는 DSC 온도기록도;
iii) 도 26에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 석시네이트의 결정 형태. - 다음 특징:
i) 도 27에 도시된 바와 같고/거나 표 10에 열거된 주 피크(main peak)를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 28에 도시된 DSC 온도기록도와 일치되게, 약 119℃에서 개시되는 흡열 효과 및 약 184℃에서 개시되는 제 2 흡열 효과를 나타내는 DSC 온도기록도;
iii) 도 29에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 석시네이트의 결정 형태. - 다음 특징:
i) 도 31에 도시된 바와 같고/거나 표 11에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 32에 도시된 DSC 온도기록도와 일치되게, 약 60℃에서 개시되는 흡열 효과, 약 103℃에서 개시되는 발열 효과 및 약 190℃에서 개시되는 제 2 흡열 효과를 나타내는 DSC 온도기록도;
iii) 도 33에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 (L)-타르트레이트의 디하이드레이트 결정 형태. - 다음 특징:
i) 도 35에 도시된 바와 같고/거나 표 12에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 36에 도시된 DSC 온도기록도와 실질적으로 일치되게, 약 36 내지 100℃ 범위에서의 흡열 반응 및 약 187℃에서 개시되는 제 2 흡열 반응을 나타내는 DSC 온도기록도;
iii) 도 37에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 (L)-타르트레이트의 테트라하이드레이티드(tetrahydrated) 결정 형태. - 다음 특징:
i) 도 38에 도시된 바와 같고/거나 표 13에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 39에 도시된 DSC 온도기록도와 실질적으로 일치되게, 약 42℃에서 개시되는 흡열 반응 및 약 180℃에서 개시되는 제 2 흡열 반응을 나타내는 DSC 온도기록도;
iii) 도 40에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 (L)-타르트레이트의 모노하이드레이티드(monohydrated) 결정 형태. - 다음 특징:
i) 도 41에 도시된 바와 같고/거나 표 14에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 42에 도시된 DSC 온도기록도와 실질적으로 일치되게, 약 189℃에서의 흡열 효과를 나타내는 DSC 온도기록도;
iii) 도 43에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 (L)-타르트레이트, 형태 D의 결정 형태. - 셀 파라미터 a = 8.9412 (5) Å, b = 9.8081 (5) Å, c = 10.5922 (6) Å, α: 90.517° (4), β = 101.969° (5), γ = 99.132° (4), V = 896.34 (8) Å3, 공간 그룹 P-1을 나타내는 삼사정계가 특징이며, 다음 특징:
i) 도 47a에 도시된 바와 같고/거나 표 16에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 48에 도시된 DSC 온도기록도와 일치되게, 약 156℃에서 개시되는 흡열 효과 및 약 243℃에서 개시되는 제 2 흡열 효과를 나타내는 DSC 온도기록도;
iii) 도 49에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 추가의 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 말레에이트의 결정 형태. - 다음 특징:
i) 도 50에 도시된 바와 같고/거나 표 17에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 51에 도시된 DSC 온도기록도와 실질적으로 일치되게, 약 83℃에서의 피크로 나타나는 흡열 반응, 약 153℃에서 개시되는 흡열 반응 및 약 228℃에서 개시되는 흡열 반응을 나타내는 DSC 온도기록도;
iii) 도 52에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 말레에이트의 헤미하이드레이티드(hemihydrated) 결정 형태. - 셀 파라미터 a = 10.7980 (8) Å, b = 11.6643 (7) Å, c = 13.0888 (11) Å, α: 90°, β = 106.842° (8), γ = 90°, V = 1577.8 (2) Å3, 공간 그룹 P21/c을 나타내는 단사정계가 특징이며, 다음 특징:
i) 도 44a에 도시된 바와 같고/거나 표 15에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 45에 도시된 DSC 온도기록도와 일치되게, 약 199℃에서 개시되는 흡열 효과, 약 200℃에서 개시되는 흡열 효과 및 약 240℃에서 개시되는 제 3 흡열 효과를 나타내는 DSC 온도기록도;
iii) 도 46에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 푸마레이트의 결정 형태. - 다음 특징:
i) 도 53에 도시된 바와 같고/거나 표 18에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 242 내지 246℃의 융점;중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 포스페이트의 결정 형태. - 다음 특징:
i) 도 54에 도시된 바와 같고/거나 표 19에 열거된 주 피크를 포함하는 X-레이 분말 회절 스펙트럼(XRPD);
ii) 도 55에 도시된 DSC 온도기록도와 일치되게, 약 229℃에서 개시되는 흡열 효과를 나타내는 DSC 온도기록도;
iii) 도 56에 도시된 FT-IR 스펙트럼과 실질적으로 일치하는 FT-IR 스펙트럼;중 하나 이상이 특징인 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 옥살레이트의 결정 형태. - 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린을 제조하는 방법으로서,
40℃ 내지 환류 온도 사이로 구성된 온도에서 용매, 예컨대, 아세토니트릴, DMF, 메탄올, 에탄올 또는 DCM중에 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린을 용해시키고, 얻은 용액을 고온인 동안 여과하고, 여액을 역용매(anti-solvent), 예컨대, 에틸 아세테이트, 아세톤 또는 메틸 에틸 케톤에 첨가함을 포함하며; 사용된 용매가 메탄올 또는 에탄올인 경우에는, 상기 용매의 대부분이 50℃ 내지 환류 온도 사이로 구성된 온도에서 증류에 의해서 제거되어야 하고; 다형체의 결정화가 얻은 용액을 8 내지 36 시간 동안 교반함으로써 30℃ 내지 20℃ 사이로 구성된 온도에서 수행되는 방법. - 제 1항에 따른 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린을 제조하는 방법으로서,
용매, 예컨대, 아세토니트릴, 에틸 아세테이트, 이소프로필 아세테이트, TBME, 디에틸 에테르, 이소프로필 에테르 또는 헥산중에서, 15℃ 내지 35℃ 사이로 구성된 온도에서 24 시간 내지 7일로 구성된 시간 동안, 제 20항의 방법에 의해서 얻은 100mg 이상의 제 1항의 다형체의 씨딩의 존재하에, 제 1항 내지 제 5항에 기재된 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린의 결정 형태의 어떠한 조합의 혼합물을 현탁액으로 교반함을 포함하는 방법. - 제 2항에 따른 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린을 제조하는 방법으로서,
60℃ 내지 환류 온도 사이로 구성된 온도에서 이소프로필 아세테이트 또는 MEK 중에 생성물을 용해시키고, 얻은 용액을 여과하고, 용액을 60℃ 내지 환류 온도 사이로 구성된 온도에서 적절한 결정화 용적으로 농축시키고, 60℃ 내지 70℃에서 결정화 동안 용액을 교반하고, 70℃ 내지 25℃ 사이로 구성된 온도에서 결정화로부터 2 시간 이하의 시간 이내에 다형체를 여과함을 포함하는 방법. - 제 3항에 따른 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린을 제조하는 방법으로서,
자일렌으로부터 생성물을 결정화시키고, 이를 50℃ 내지 90℃ 사이로 구성된 온도에서 여과함을 포함하는 방법. - 제 6항에 따른 6-(1H-이미다졸-1-일)-2-페닐퀴나졸린 디하이드로클로라이드 모노하이드레이트를 제조하는 방법으로서,
50℃ 내지 환류 온도 사이로 구성된 온도에서 용매, 예컨대, 메탄올, 에탄올, 이소프로판올, 또는 물중에 염기를 용해시키고, 수성 HCl로 산성화시키고, 임의로 역용매, 예컨대, 아세톤, 디옥산 또는 테트라하이드로푸란을 첨가하고, 5℃로 냉각시키고, 적어도 3 내지 4 시간 동안 얻은 현탁액을 교반하고, 여과하고, 20 mmHg이하의 진공하에 20℃ 내지 30℃에서 건조시키는 것으로 이루어지는 방법. - 락토오스, 텍스트로오스, 만니톨, 폴리락트산 및 이들의 혼합물로 이루어진 군으로부터 선택된 하나 이상의 부형제 및 약리학적 활성 양의 제 1 항 내지 제 19항에 따른 화합물중 하나 이상을 포함하는 약제학적 제형.
- 약리학적 활성 양의 제 1 항 내지 제 19항에 따른 화합물중 하나 이상 및 염수 수용액(NaCl 0.9%), 만니톨의 등장성 용액 및 디메틸 설폭사이드로부터 선택된 적합한 용매를 포함하는 약제학적 제형.
- 만성 또는 급성 통증의 치료, 염증성 상태의 병리의 치료, 및 우울증의 치료에서 제 25항 및 제 26항에 따른 약제학적 조성물의 용도.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ITTO2009A000424 | 2009-06-04 | ||
| ITTO2009A000424A IT1395963B1 (it) | 2009-06-04 | 2009-06-04 | Forme cristalline di 6-(1h-imidazol-1-il)-2-fenil chinazolina e dei suoi sali |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020167022691A Division KR101939145B1 (ko) | 2009-06-04 | 2010-06-04 | 6―(1h―이미다졸―1―일)―2―페닐퀴나졸린의 결정 형태 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20120028362A true KR20120028362A (ko) | 2012-03-22 |
Family
ID=41527692
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020127000282A Ceased KR20120028362A (ko) | 2009-06-04 | 2010-06-04 | 6?(1h?이미다졸?1?일)?2?페닐퀴나졸린의 결정 형태 |
| KR1020167022691A Expired - Fee Related KR101939145B1 (ko) | 2009-06-04 | 2010-06-04 | 6―(1h―이미다졸―1―일)―2―페닐퀴나졸린의 결정 형태 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020167022691A Expired - Fee Related KR101939145B1 (ko) | 2009-06-04 | 2010-06-04 | 6―(1h―이미다졸―1―일)―2―페닐퀴나졸린의 결정 형태 |
Country Status (15)
| Country | Link |
|---|---|
| US (1) | US9174966B2 (ko) |
| EP (1) | EP2438058B1 (ko) |
| JP (1) | JP5739879B2 (ko) |
| KR (2) | KR20120028362A (ko) |
| CN (1) | CN102459237B (ko) |
| AU (1) | AU2010255376A1 (ko) |
| BR (1) | BRPI1009006B1 (ko) |
| CA (1) | CA2762307C (ko) |
| DK (1) | DK2438058T3 (ko) |
| ES (1) | ES2676646T3 (ko) |
| HU (1) | HUE037631T2 (ko) |
| IT (1) | IT1395963B1 (ko) |
| MX (1) | MX343234B (ko) |
| RU (1) | RU2557547C2 (ko) |
| WO (1) | WO2010140139A2 (ko) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ITTO20120103A1 (it) * | 2012-02-08 | 2013-08-09 | Rottapharm Spa | Forme cristalline di 6-(1h-imidazol-1-il)-2-fenil chinazolina e dei suoi sali |
| ITTO20120102A1 (it) * | 2012-02-08 | 2013-08-09 | Rottapharm Spa | Forme cristalline di 6-(1h-imidazol-1-il)-2-fenil chinazolina e dei suoi sali |
| SMT202000084T1 (it) * | 2013-03-06 | 2020-03-13 | Janssen Pharmaceutica Nv | Modulatori benzoimidazol-2-il pirimidinici del recettore h4 dell'istamina |
| PL3134407T3 (pl) * | 2014-04-22 | 2019-11-29 | Arqule Inc | Sole i polimorfy podstawionego związku imidazopirydynylo- aminopirydyny |
| EP3822264A4 (en) * | 2018-07-02 | 2021-11-10 | Jiangsu Hengrui Medicine Co., Ltd. | CRYSTALLINE FORMS OF AN OXYPYRIDINE AMIDE DERIVATIVE AND THEIR PREPARATION PROCESS |
| EP3735974A1 (en) | 2019-05-10 | 2020-11-11 | Rottapharm Biotech S.r.l. | Use of 2-phenyl-6-(1h-imidazol-1-yl)quinazoline for treating neurodegenerative diseases, preferably alzheimer's disease |
| BR112021024330A2 (pt) * | 2019-06-03 | 2022-01-11 | Biotheryx Inc | Sais cristalinos não higroscópicos de um composto de pirazol, e composições farmacêuticas e uso dos mesmos |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2066653B1 (en) * | 2006-08-03 | 2012-09-12 | Rottapharm S.p.A. | 6-1h-imidazo-quinazoline and quinolines derivatives, new potent analgesics and anti-inflammatory agents |
| US8999590B2 (en) * | 2007-07-25 | 2015-04-07 | Fuelcell Energy, Inc. | On-line monitoring assembly for detection of sulfur breakthrough in a desulfurizer assembly and sulfur breakthrough detection method |
| MX2010014394A (es) * | 2008-06-20 | 2011-05-19 | Rottapharm Spa | Derivados de 6-1h-imidazo-quinazolina y quinolinas, nuevos inhibidores de monoamina oxidasa y ligandos del receptor de imidazolina. |
-
2009
- 2009-06-04 IT ITTO2009A000424A patent/IT1395963B1/it active
-
2010
- 2010-06-04 CN CN201080024845.4A patent/CN102459237B/zh active Active
- 2010-06-04 CA CA2762307A patent/CA2762307C/en not_active Expired - Fee Related
- 2010-06-04 EP EP10731576.4A patent/EP2438058B1/en active Active
- 2010-06-04 AU AU2010255376A patent/AU2010255376A1/en not_active Abandoned
- 2010-06-04 HU HUE10731576A patent/HUE037631T2/hu unknown
- 2010-06-04 DK DK10731576.4T patent/DK2438058T3/en active
- 2010-06-04 KR KR1020127000282A patent/KR20120028362A/ko not_active Ceased
- 2010-06-04 WO PCT/IB2010/052496 patent/WO2010140139A2/en not_active Ceased
- 2010-06-04 RU RU2011153981/04A patent/RU2557547C2/ru active
- 2010-06-04 BR BRPI1009006-1A patent/BRPI1009006B1/pt not_active IP Right Cessation
- 2010-06-04 KR KR1020167022691A patent/KR101939145B1/ko not_active Expired - Fee Related
- 2010-06-04 MX MX2011012859A patent/MX343234B/es active IP Right Grant
- 2010-06-04 JP JP2012513729A patent/JP5739879B2/ja not_active Expired - Fee Related
- 2010-06-04 US US13/375,816 patent/US9174966B2/en active Active
- 2010-06-04 ES ES10731576.4T patent/ES2676646T3/es active Active
Also Published As
| Publication number | Publication date |
|---|---|
| HUE037631T2 (hu) | 2018-09-28 |
| US20120142713A1 (en) | 2012-06-07 |
| JP2012528849A (ja) | 2012-11-15 |
| CA2762307A1 (en) | 2010-12-09 |
| KR20160102325A (ko) | 2016-08-29 |
| AU2010255376A1 (en) | 2011-12-01 |
| CA2762307C (en) | 2017-12-12 |
| CN102459237A (zh) | 2012-05-16 |
| CN102459237B (zh) | 2015-05-27 |
| IT1395963B1 (it) | 2012-11-02 |
| ES2676646T3 (es) | 2018-07-23 |
| MX2011012859A (es) | 2012-05-23 |
| KR101939145B1 (ko) | 2019-01-16 |
| WO2010140139A2 (en) | 2010-12-09 |
| ITTO20090424A1 (it) | 2010-12-05 |
| RU2557547C2 (ru) | 2015-07-27 |
| US9174966B2 (en) | 2015-11-03 |
| DK2438058T3 (en) | 2018-06-06 |
| BRPI1009006B1 (pt) | 2019-09-24 |
| EP2438058A2 (en) | 2012-04-11 |
| RU2011153981A (ru) | 2013-07-20 |
| EP2438058B1 (en) | 2018-04-11 |
| WO2010140139A3 (en) | 2011-01-27 |
| JP5739879B2 (ja) | 2015-06-24 |
| BRPI1009006A2 (pt) | 2016-03-08 |
| MX343234B (es) | 2016-10-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102881316B1 (ko) | 암 치료를 위한 raf 억제제로서의 n-(3-(2-(2-하이드록시에톡시)-6-모르폴리노피리딘-4-일)-4-메틸페닐)-2 (트리플루오로메틸)이소니코틴아미드의 새로운 결정질 형태 | |
| CN105209437B (zh) | 心肌肌球蛋白激动剂的盐和制备盐的方法 | |
| US8022216B2 (en) | Maleate salts of (E)-N-{4-[3-chloro-4-(2-pyridinylmethoxy)anilino]-3-cyano-7-ethoxy-6-quinolinyl}-4-(dimethylamino)-2-butenamide and crystalline forms thereof | |
| KR101939145B1 (ko) | 6―(1h―이미다졸―1―일)―2―페닐퀴나졸린의 결정 형태 | |
| EP3430004B1 (en) | Solid state forms of nilotinib salts | |
| JP6816036B2 (ja) | ヒストン脱アセチル化阻害剤の結晶形態 | |
| WO2015001098A1 (en) | Crystalline forms of ponatinib hydrochloride | |
| CN105085387A (zh) | 贝曲西班盐及其制备方法和用途 | |
| CZ2016548A3 (cs) | Pevné formy eluxadolinu | |
| US20200308141A1 (en) | Solid state forms of neratinib and salts thereof | |
| EP4134369B1 (en) | Solid forms of tetrahydropyranyl amino-pyrrolopyrimidinone compounds | |
| JP7152122B2 (ja) | エダラボン塩 | |
| AU2011101558A4 (en) | Crystalline forms of 6-(1H-imadazol-1-YL)-2-phenylquinazoline | |
| KR20230026384A (ko) | 화합물의 결정 형태 | |
| TWI752916B (zh) | 1-(4-(2-((1-(3,4-二氟苯基)-1h-吡唑-3-基)甲氧基)乙基)哌-1-基)乙酮鹽 | |
| CN111499627A (zh) | 取代的咪唑并吡啶基-氨基吡啶化合物的盐和多晶型 | |
| KR20260003055A (ko) | 에베나마이드의 결정형 | |
| RU2807142C1 (ru) | Малеатные соли (E)-N-{ 4-[3-хлор-4-(2-пиридинилметокси)анилино]-3-циано-7-этокси-6-хинолинил} -4-(диметиламино)-2-бутенамида и их кристаллические формы | |
| TW201718538A (zh) | 福奈孚匹坦的結晶形式 | |
| JP2025532703A (ja) | 1-(8-ブロモピリド[2,3-e][1,2,4]トリアゾロ[4,3-a]ピラジン-4-イル)-N-メチルアゼチジン-3-アミンの塩および結晶形の製造方法、ならびに新規結晶形 | |
| ITTO20120102A1 (it) | Forme cristalline di 6-(1h-imidazol-1-il)-2-fenil chinazolina e dei suoi sali | |
| CN116322672A (zh) | 2-[3-({1-[2-(二甲基氨基)乙基]-2-(2,2-二甲基丙基)-1h-1,3-苯并二氮唑-5-基}磺酰基)氮杂环丁烷-1-基]乙-1-醇的稳定盐和晶型 | |
| ITTO20120103A1 (it) | Forme cristalline di 6-(1h-imidazol-1-il)-2-fenil chinazolina e dei suoi sali | |
| HU231012B1 (hu) | Lapatinib sók | |
| HK1234747B (zh) | 取代的咪唑并吡啶基-氨基吡啶化合物的盐和多晶型 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20120104 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| N231 | Notification of change of applicant | ||
| PN2301 | Change of applicant |
Patent event date: 20140702 Comment text: Notification of Change of Applicant Patent event code: PN23011R01D |
|
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20150604 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20160621 Patent event code: PE09021S01D |
|
| A107 | Divisional application of patent | ||
| PA0104 | Divisional application for international application |
Comment text: Divisional Application for International Patent Patent event code: PA01041R01D Patent event date: 20160819 |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20160823 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20160621 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |